User login
Management of Seizure Emergencies
Bearing Surfaces in Hip Arthroplasty
Confused about mammography guidelines? 7 questions answered
Some clinicians were reconsidering the need for an annual mammogram even before the US Preventive Services Task Force (USPSTF) issued new guidelines late last year.1
Andrew M. Kaunitz, MD, is one of those clinicians. In an editorial in the December issue of OBG Management, he was bold enough to declare: “My plan is to be more acquiescent when a woman says ‘No’ to an annual mammogram.”2
Among the evidence he cited to justify that acquiescence was a recent article in the Journal of the American Medical Association that expressed concern about the high number of early cancers—including ductal carcinoma in situ—that are detected by mammography and treated even though many are unlikely to progress or ever become clinically significant.3 This phenomenon—termed “over-diagnosis”—is one of the risks of breast cancer screening.
Dr. Kaunitz is professor and associate chairman of obstetrics and gynecology at the University of Florida College of Medicine–Jacksonville. He also serves on the OBG Management Board of Editors.
Although the USPSTF is the only official body to revise its recommendations on breast cancer screening so far, more changes seem likely. This article aims to sift through the static on the airwaves of late and offer concrete recommendations for practice. In the process, it addresses seven questions:
- How did USPSTF guidelines change?
- Why did they change?
- Why did the changes attract so much attention?
- What is ACOG’s position?
- What do thought leaders make of the new guidelines?
- Are the USPSTF recommendations likely to affect insurance coverage for mammography?
- What should you tell your patients about breast cancer screening?
1. How did USPSTF guidelines change?
In an article published November 16, the USPSTF made a number of revisions to earlier breast cancer screening guidelines for women at average risk of the disease:
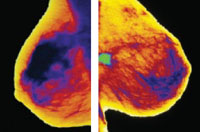
Approximately 39 million women undergo mammography each year in the United States, costing the health-care system more than $5 billion.
- Routine screening mammography is no longer recommended in women 40 to 49 years old. Rather, the decision about when to begin regular screening should be individualized and should “take into account patient context, including the patient’s values regarding specific benefits and harms” (Grade C recommendation).
- Screening mammography in women 50 to 74 years old should be biennial rather than annual (Grade B recommendation).
- Breast self-examination (BSE) is not recommended for any age group (Grade D recommendation).1
2. Why did the USPSTF guidelines change?
The changes were based on new data and analysis in the following areas:
- Mortality among women 40 to 49 years old. Although mammography screening reduces breast cancer mortality by 15% in this age group, the USPSTF concluded that “there is moderate certainty that the net benefit is small” in this population.1,4
- The effectiveness of BSE in decreasing breast cancer mortality among women of any age. Studies of BSE published since 2002 found no significant differences in breast cancer mortality between women who perform BSE and those who don’t.4
- The magnitude of harms of screening with mammography. Mammography screening in women 40 to 49 years old involves a significant risk of harms.4 Although the USPSTF observed that the benefits of mammography in women 40 to 49 years old appear to be equivalent to the benefits of mammography among women 50 to 59 years old, it concluded that the harms outweigh benefits in the younger women.
Harms cited by the USPSTF include:
- radiation exposure
- pain during the procedure
- anxiety and distress
- an increased rate of false-positive results
- greater need for additional imaging and biopsies.4
The USPSTF conceded that the radiation exposure from a mammogram is minimal, but questioned whether cumulative exposure in young women might be problematic. It also noted that “many women experience pain during the procedure (range, 1% to 77%), but few would consider this a deterrent from future screening.”4
As for false-positive results, the group observed: “Data from the [Breast Cancer Screening Consortium (BCSC)] for regularly screened women…indicate that false-positive mammography results are common in all age groups but are most common among women aged 40 to 49 years (97.8 per 1,000 women per screening round).”4
“The BCSC results indicate that for every case of invasive breast cancer detected by mammography screening in women aged 40 to 49 years, 556 women have mammography, 47 have additional imaging, and five have biopsies.”4
It is the significant rate of false positives that creates the need for additional screening, diagnostic imaging, and biopsy. These additional imaging and invasive procedures increase anxiety and distress among many women. The USPSTF concluded that these harms outweighed the benefits of mammography screening in women 40 to 49 years old.
After publication of the new US Preventive Services Task Force (USPSTF) breast cancer screening guidelines late last year, it was only a matter of hours before official bodies and professional organizations began to weigh in on the changes, and the verdict was unanimous—disagreement. Among those chiming in were the American Cancer Society (ACS), the American College of Obstetricians and Gynecologists (ACOG), the American College of Radiology, the American Society of Breast Surgeons, the Society for Breast Imaging (SBI), and Susan G. Komen for the Cure, among others. Here are excerpts from their statements.
American Cancer Society
The ACS immediately refuted the USPSTF recommendations:
The American Cancer Society continues to recommend annual screening using mammography and clinical breast examination for all women beginning at age 40. Our experts make this recommendation having reviewed virtually all the same data reviewed by the USPSTF, but also additional data that the USPSTF did not consider….[T]he American Cancer Society’s medical staff and volunteer experts overwhelmingly believe the benefits of screening women aged 40 to 49 outweigh its limitations.7
ACOG
The College reaffirmed its support for screening mammography every 1 to 2 years in women 40 to 49 years old and every year for women 50 and older, as well as breast self-examination for women of all ages:
At this time, The American College of Obstetricians and Gynecologists recommends that Fellows continue to follow current College guidelines for breast cancer screening. Evaluation of the new USPSTF recommendations is under way. Should the College update its guidelines in the future, Fellows would be alerted and such revised guidelines would be published in Obstetrics & Gynecology.5
American College of Radiology
The College minced no words in opposing the changes:
If cost-cutting US Preventive Services Task Force (USPSTF) mammography recommendations are adopted as policy, two decades of decline in breast cancer mortality could be reversed and countless American women may die needlessly from breast cancer each year.
These new recommendations seem to reflect a conscious decision to ration care. If Medicare and private insurers adopt these incredibly flawed USPSTF recommendations as a rationale for refusing women coverage of these life-saving exams, it could have deadly effects for American women,” said Carol H. Lee, MD, chair of the American College of Radiology Breast Imaging Commission.8
American Society of Breast Surgeons
The organization released a statement describing its position as “strongly opposed” to the USPSTF recommendations:
We believe there is sufficient data to support annual mammography screening for women age 40 and older. We also believe the breast cancer survival rate of women between 40 and 50 will improve from the increased use of digital mammographic screening, which is superior to older plain film techniques in detecting breast cancer in that age group.
While we recognize that there will be a number of benign biopsies, we also recognize that mammography is the optimal screening tool for the early diagnosis of breast cancer in terms of cost-effectiveness, practical use, and accuracy.9
Society for Breast Imaging
In its statement, the SBI noted the confusion caused by revision of the USPSTF guidelines, calling it “unnecessary and potentially deadly”:
Mammography has been shown unequivocally to save lives and is primarily responsible for the 30% decline in breast cancer mortality in the United States over the past 20 years. The USPSTF conclusion—that women under age 50 should not undergo routine screening—conflicts with their own report, which confirms a benefit of mammography to women age 40–49 that is statistically significant.
We strongly urge women and their physicians to adhere to the American Cancer Society recommendations of yearly screening beginning at age 40.10
Susan G. Komen for the Cure
This public advocacy group issued a statement in late November acknowledging “mass confusion and justifiable outrage” in the aftermath of the USPSTF changes:
”We have worked so hard to build public trust and urge people to get screened,” said Nancy G. Brinker, founder of Susan G. Komen for the Cure, “and now they hear that maybe they shouldn’t bother. That is dangerous….Let me say this as clearly as I can: Mammography saves lives, even this report says that. Keep doing what you are doing. And always, talk with your doctor.” Brinker also noted that Komen for the Cure was not changing its guidelines, continuing to recommend annual mammograms beginning at age 40.11
3. Why have the guidelines captured so much media attention?
Most of the controversy that has arisen since publication of the new guidelines has centered on the recommendation against screening mammography in women 40 to 49 years old. A number of media outlets have highlighted women whose breast cancer was detected by screening mammography when they were in their 40s, and many survivors with a similar history have spoken out against the new recommendations.
In addition, the American Cancer Society (ACS), the American College of Radiology, Susan G. Komen for the Cure, and other groups have publicly opposed the new guidelines. (See “Among professional organizations, a resounding chorus of disagreement”)
4. What is ACOG’s position on the new recommendations?
The American College of Obstetricians and Gynecologists (ACOG) was quick to weigh in on the new USPSTF guidelines, emphasizing that the College’s recommendations have not changed. They include:
- screening mammography every 1 to 2 years for women 40 to 49 years old
- screening mammography every year for women 50 years and older
- BSE for all women.
ACOG did note, however, that “the College is continuing to evaluate in detail the new USPSTF recommendations and the new evidence considered by the USPSTF.”5
5. What do thought leaders make of the USPSTF changes?
Although the USPSTF guidelines sparked a firestorm of media coverage, the change did not come as a shock to leaders in the ObGyn specialty.

Legitimate concerns about screening mammography have increasingly been raised by experts in the field.
ANDREW M. KAUNITZ, MD “I was not surprised,” said Dr. Kaunitz. “As I pointed out in my editorial in OBG Management, legitimate concerns about screening mammography have increasingly been raised by experts in the field.2 Proposals to stop routinely screening women in their 40s were made earlier in this decade, but were met with major pushback from the ACS, breast cancer advocacy organizations, and medical specialty groups. These same groups are now pushing back against the new USPSTF guidelines,” he added.
Robert L. Barbieri, MD, was not taken aback by the guidelines themselves, but he was surprised by the manner and timing of their release. Dr. Barbieri is Kate Macy Ladd professor of obstetrics, gynecology, and reproductive biology at Harvard Medical School and chief of obstetrics and gynecology at Brigham and Women’s Hospital in Boston. He serves as editor-in-chief of OBG Management.
“I was surprised that the USPSTF did not weigh the potential impact of its analysis on the key stakeholders: patients, disease-based coalitions such as the American Cancer Society and Susan G. Komen for the Cure, and professional societies such as the American College of Radiology and ACOG,” he said. “If I were supervising the process, I would have asked for a comment period before releasing the report. I would have included the comments from key stakeholders in an appendix to the report.”
Are other organizations—besides the USPSTF—likely to change their recommendations for mammography screening in the near future? In the case of ACOG, Dr. Barbieri doesn’t think so.
“I don’t think ACOG will change the age at which to initiate screening,” he said. “I believe it will stick to its recommendation to start screening at 40 and continue every 1 to 2 years from 40 to 50 years of age. However, I could see ACOG becoming a bit more flexible on the question of whether screening should take place at 1- or 2-year intervals after age 50.”
Dr. Kaunitz sees things differently.
“It seems possible that, going forward, the College will give Fellows and their patients permission to implement the new guidelines without mandating their implementation. For example, if women in their 40s wish to defer screening, that would be OK, as would biennial screening for women in their 50s and 60s.”
6. Are the USPSTF recommendations likely to affect insurance coverage?
In a press release issued soon after the new guidelines were published, US Health and Human Services Secretary Kathleen Sebelius addressed Americans directly to reaffirm her support for mammography in women 40 to 49 years old: “There is no question that the US Preventive Services Task Force recommendations have caused a great deal of confusion and worry among women and their families,” her statement read.6 She made it clear that the new recommendations are unlikely to affect federal coverage of mammography.
“The US Preventive Services Task Force is an outside independent panel of doctors and scientists who make recommendations. They do not set federal policy and they don’t determine what services are covered by the federal government,” she said.6
But Dr. Barbieri thinks some changes in insurance coverage are inevitable.
“Any claims that the new guidelines do not represent a major change would be disingenuous,” he said. Because the USPSTF rated its recommendation against mammography for women 40 to 49 years old as grade ‘C,’ that change in guidelines is likely to trigger at least some change in coverage.
“In reality, the ‘C’ rating will require many insurance companies—by their own rule—to stop reimbursing for this screening test,” he said. “The ‘C’ rating means that the test has little benefit.”
ACOG also deems it likely that insurance coverage may be affected for some women.
“Fellows should be aware that the new USPSTF recommendations against routine screening mammography for women aged 40–49 (a grade C recommendation) has implications for insurance coverage, as some insurers will cover only preventive services rated as an ‘A’ or a ‘B’ by the USPSTF. Fellows should counsel their patients that insurance coverage for ‘routine screening’ mammography may become variable and that patients should address this question with their insurers. These recommendations do not apply to high-risk women or patients with clinical findings, and they should be managed accordingly.”5
7. What should you tell your patients?
With all the media attention devoted to the change in guidelines, it’s little surprise that patients are asking questions.
“Patients are aware of the USPSTF report,” said Dr. Barbieri. “They are largely ignoring the recommendations and sticking with annual mammograms.”
“I think, as always, women are looking to their ObGyn for guidance,” added Dr. Kaunitz.
So what are these clinicians telling patients about mammography screening?
As he was to begin with, Dr. Kaunitz is acquiescent if patients prefer to defer mammography screening to their 50s.
“Because it seems that insurance coverage, over the short term, is unlikely to restrict current access to mammograms,” said Dr. Kaunitz, “my evolving philosophy is that the new USPSTF guidelines, along with ACOG and other existing guidelines, give ObGyns and their patients permission to:
- proceed or not proceed with mammograms for women in their 40s, with the decision based on issues such as patient preference, family history of breast cancer, and body mass index (BMI)
- be flexible regarding 1- to 2-year screening intervals among women in their 50s, 60s, and 70s, with the decision based on issues such as patient preference, use or non-use of estrogen-progestin hormone therapy, family history of breast cancer, and BMI.”
Dr. Barbieri believes some effort to integrate the ACOG and USPSTF recommendations is called for. “Accordingly,” he said, “I suggest the following:

I suggest actively recommending biennial mammography for women 40 to 75 years old. Offer annual mammography to women 40 to 75 years old if they prefer that option.
ROBERT L. BARBIERI, MD
- Actively recommend biennial mammography for women 40 to 75 years old. Offer annual mammography to women 40 to 75 years old if they prefer that option.
- Aggressively search for high-risk women, with high risk defined as a lifetime risk of breast cancer exceeding 15%. Among the variables contributing to high-risk status are a history of thoracic radiotherapy, a strong family history of breast cancer, and BRCA mutation. For these women, I would recommend annual mammography and biennial MRI of the breasts.
- Perform annual or biennial clinical breast exam.
- Obtain imaging for any woman who has a palpable breast lump, and resect or biopsy the lump even if that imaging is negative.”
1. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151:716-726.
2. Kaunitz AM. I’ve been rethinking my zeal for breast cancer screening. OBG Management. 2009;21(12):6-8.
3. Esserman L, Shieh Y, Thompson I. Rethinking screening for breast cancer and prostate cancer. JAMA. 2009;302:1685-1692.
4. Nelson HD, Tyne K, Nalk A, Bougatsos C, Chan BK, Humphrey L. Screening for breast cancer: an update for the US Preventive Services Task Force. Ann Intern Med. 2009;151:727-737.
5. American College of Obstetricians and Gynecologists. Response of the American College of Obstetricians and Gynecologists to new breast cancer screening recommendations from the US Preventive Services Task Force. Available at: http://www.acog.org/from_home/Misc/uspstfResponse.cfm. Accessed Nov. 25, 2009.
6. US Department of Health and Human Services. Secretary Sebelius statement on new breast cancer recommendations [news release]. Nov. 18, 2009. Available at: http://www.hhs.gov/news/press/2009pres/11/20091118a.html. Accessed Dec. 4, 2009.
7. American Cancer Society responds to changes to USPSTF mammography guidelines [news release]. American Cancer Society. Nov. 16, 2009. Available at: http://www.cancer.org/docroot/MED/content/MED_2_1x_American_Cancer_Society_
Responds_to_Changes_to_USPSTF_Mammography_Guidelines.asp. Accessed Dec. 4, 2009.
8. American College of Radiology. USPSTF mammography recommendations will result in countless unnecessary breast cancer deaths each year [news release]. Nov. 16, 2009. Available at: www.acr.org/MainMenuCategories/media_room/FeaturedCategories/PressReleases/USPSTFMammoRecs.aspx. Accessed Dec. 4, 2009.
9. American Society of Breast Surgeons. Society responds to USPSTF changes in mammography guidelines [news release]. Available at: http://www.breastsurgeons.org/news/article.php?id=57. Accessed Dec. 4, 2009.
10. Society of Breast Imaging. Official Society of Breast Imaging response to the announcement by HHS Secretary Sebelius regarding USPSTF mammography recommendations. Available at:http://www.sbi-online.org/associations/8199/files/OFFICIAL%20SOCIETY%20OF%20
BREAST%20IMAGING%20RESPONSE%20TO%20THE%20ANNOUNCEMENT%20
BY%20HHS%20SECRETARY%20
SEBELIUS%20REGARDING%20USPSTF%20MAMMOGRAPHY%20RECOMMENDATIONS.pdf. Accessed Dec. 4, 2009.
11. Susan G. Komen for the Cure founder, Nancy G. Brinker, calls new mammography guidelines a “set back”; makes call to action [news release]. Nov. 23, 2009. Available at: http://ww5.komen.org/KomenNewsArticle.aspx?id=6442451516. Accessed Dec. 7, 2009.
Some clinicians were reconsidering the need for an annual mammogram even before the US Preventive Services Task Force (USPSTF) issued new guidelines late last year.1
Andrew M. Kaunitz, MD, is one of those clinicians. In an editorial in the December issue of OBG Management, he was bold enough to declare: “My plan is to be more acquiescent when a woman says ‘No’ to an annual mammogram.”2
Among the evidence he cited to justify that acquiescence was a recent article in the Journal of the American Medical Association that expressed concern about the high number of early cancers—including ductal carcinoma in situ—that are detected by mammography and treated even though many are unlikely to progress or ever become clinically significant.3 This phenomenon—termed “over-diagnosis”—is one of the risks of breast cancer screening.
Dr. Kaunitz is professor and associate chairman of obstetrics and gynecology at the University of Florida College of Medicine–Jacksonville. He also serves on the OBG Management Board of Editors.
Although the USPSTF is the only official body to revise its recommendations on breast cancer screening so far, more changes seem likely. This article aims to sift through the static on the airwaves of late and offer concrete recommendations for practice. In the process, it addresses seven questions:
- How did USPSTF guidelines change?
- Why did they change?
- Why did the changes attract so much attention?
- What is ACOG’s position?
- What do thought leaders make of the new guidelines?
- Are the USPSTF recommendations likely to affect insurance coverage for mammography?
- What should you tell your patients about breast cancer screening?
1. How did USPSTF guidelines change?
In an article published November 16, the USPSTF made a number of revisions to earlier breast cancer screening guidelines for women at average risk of the disease:

Approximately 39 million women undergo mammography each year in the United States, costing the health-care system more than $5 billion.
- Routine screening mammography is no longer recommended in women 40 to 49 years old. Rather, the decision about when to begin regular screening should be individualized and should “take into account patient context, including the patient’s values regarding specific benefits and harms” (Grade C recommendation).
- Screening mammography in women 50 to 74 years old should be biennial rather than annual (Grade B recommendation).
- Breast self-examination (BSE) is not recommended for any age group (Grade D recommendation).1
2. Why did the USPSTF guidelines change?
The changes were based on new data and analysis in the following areas:
- Mortality among women 40 to 49 years old. Although mammography screening reduces breast cancer mortality by 15% in this age group, the USPSTF concluded that “there is moderate certainty that the net benefit is small” in this population.1,4
- The effectiveness of BSE in decreasing breast cancer mortality among women of any age. Studies of BSE published since 2002 found no significant differences in breast cancer mortality between women who perform BSE and those who don’t.4
- The magnitude of harms of screening with mammography. Mammography screening in women 40 to 49 years old involves a significant risk of harms.4 Although the USPSTF observed that the benefits of mammography in women 40 to 49 years old appear to be equivalent to the benefits of mammography among women 50 to 59 years old, it concluded that the harms outweigh benefits in the younger women.
Harms cited by the USPSTF include:
- radiation exposure
- pain during the procedure
- anxiety and distress
- an increased rate of false-positive results
- greater need for additional imaging and biopsies.4
The USPSTF conceded that the radiation exposure from a mammogram is minimal, but questioned whether cumulative exposure in young women might be problematic. It also noted that “many women experience pain during the procedure (range, 1% to 77%), but few would consider this a deterrent from future screening.”4
As for false-positive results, the group observed: “Data from the [Breast Cancer Screening Consortium (BCSC)] for regularly screened women…indicate that false-positive mammography results are common in all age groups but are most common among women aged 40 to 49 years (97.8 per 1,000 women per screening round).”4
“The BCSC results indicate that for every case of invasive breast cancer detected by mammography screening in women aged 40 to 49 years, 556 women have mammography, 47 have additional imaging, and five have biopsies.”4
It is the significant rate of false positives that creates the need for additional screening, diagnostic imaging, and biopsy. These additional imaging and invasive procedures increase anxiety and distress among many women. The USPSTF concluded that these harms outweighed the benefits of mammography screening in women 40 to 49 years old.
After publication of the new US Preventive Services Task Force (USPSTF) breast cancer screening guidelines late last year, it was only a matter of hours before official bodies and professional organizations began to weigh in on the changes, and the verdict was unanimous—disagreement. Among those chiming in were the American Cancer Society (ACS), the American College of Obstetricians and Gynecologists (ACOG), the American College of Radiology, the American Society of Breast Surgeons, the Society for Breast Imaging (SBI), and Susan G. Komen for the Cure, among others. Here are excerpts from their statements.
American Cancer Society
The ACS immediately refuted the USPSTF recommendations:
The American Cancer Society continues to recommend annual screening using mammography and clinical breast examination for all women beginning at age 40. Our experts make this recommendation having reviewed virtually all the same data reviewed by the USPSTF, but also additional data that the USPSTF did not consider….[T]he American Cancer Society’s medical staff and volunteer experts overwhelmingly believe the benefits of screening women aged 40 to 49 outweigh its limitations.7
ACOG
The College reaffirmed its support for screening mammography every 1 to 2 years in women 40 to 49 years old and every year for women 50 and older, as well as breast self-examination for women of all ages:
At this time, The American College of Obstetricians and Gynecologists recommends that Fellows continue to follow current College guidelines for breast cancer screening. Evaluation of the new USPSTF recommendations is under way. Should the College update its guidelines in the future, Fellows would be alerted and such revised guidelines would be published in Obstetrics & Gynecology.5
American College of Radiology
The College minced no words in opposing the changes:
If cost-cutting US Preventive Services Task Force (USPSTF) mammography recommendations are adopted as policy, two decades of decline in breast cancer mortality could be reversed and countless American women may die needlessly from breast cancer each year.
These new recommendations seem to reflect a conscious decision to ration care. If Medicare and private insurers adopt these incredibly flawed USPSTF recommendations as a rationale for refusing women coverage of these life-saving exams, it could have deadly effects for American women,” said Carol H. Lee, MD, chair of the American College of Radiology Breast Imaging Commission.8
American Society of Breast Surgeons
The organization released a statement describing its position as “strongly opposed” to the USPSTF recommendations:
We believe there is sufficient data to support annual mammography screening for women age 40 and older. We also believe the breast cancer survival rate of women between 40 and 50 will improve from the increased use of digital mammographic screening, which is superior to older plain film techniques in detecting breast cancer in that age group.
While we recognize that there will be a number of benign biopsies, we also recognize that mammography is the optimal screening tool for the early diagnosis of breast cancer in terms of cost-effectiveness, practical use, and accuracy.9
Society for Breast Imaging
In its statement, the SBI noted the confusion caused by revision of the USPSTF guidelines, calling it “unnecessary and potentially deadly”:
Mammography has been shown unequivocally to save lives and is primarily responsible for the 30% decline in breast cancer mortality in the United States over the past 20 years. The USPSTF conclusion—that women under age 50 should not undergo routine screening—conflicts with their own report, which confirms a benefit of mammography to women age 40–49 that is statistically significant.
We strongly urge women and their physicians to adhere to the American Cancer Society recommendations of yearly screening beginning at age 40.10
Susan G. Komen for the Cure
This public advocacy group issued a statement in late November acknowledging “mass confusion and justifiable outrage” in the aftermath of the USPSTF changes:
”We have worked so hard to build public trust and urge people to get screened,” said Nancy G. Brinker, founder of Susan G. Komen for the Cure, “and now they hear that maybe they shouldn’t bother. That is dangerous….Let me say this as clearly as I can: Mammography saves lives, even this report says that. Keep doing what you are doing. And always, talk with your doctor.” Brinker also noted that Komen for the Cure was not changing its guidelines, continuing to recommend annual mammograms beginning at age 40.11
3. Why have the guidelines captured so much media attention?
Most of the controversy that has arisen since publication of the new guidelines has centered on the recommendation against screening mammography in women 40 to 49 years old. A number of media outlets have highlighted women whose breast cancer was detected by screening mammography when they were in their 40s, and many survivors with a similar history have spoken out against the new recommendations.
In addition, the American Cancer Society (ACS), the American College of Radiology, Susan G. Komen for the Cure, and other groups have publicly opposed the new guidelines. (See “Among professional organizations, a resounding chorus of disagreement”)
4. What is ACOG’s position on the new recommendations?
The American College of Obstetricians and Gynecologists (ACOG) was quick to weigh in on the new USPSTF guidelines, emphasizing that the College’s recommendations have not changed. They include:
- screening mammography every 1 to 2 years for women 40 to 49 years old
- screening mammography every year for women 50 years and older
- BSE for all women.
ACOG did note, however, that “the College is continuing to evaluate in detail the new USPSTF recommendations and the new evidence considered by the USPSTF.”5
5. What do thought leaders make of the USPSTF changes?
Although the USPSTF guidelines sparked a firestorm of media coverage, the change did not come as a shock to leaders in the ObGyn specialty.

Legitimate concerns about screening mammography have increasingly been raised by experts in the field.
ANDREW M. KAUNITZ, MD “I was not surprised,” said Dr. Kaunitz. “As I pointed out in my editorial in OBG Management, legitimate concerns about screening mammography have increasingly been raised by experts in the field.2 Proposals to stop routinely screening women in their 40s were made earlier in this decade, but were met with major pushback from the ACS, breast cancer advocacy organizations, and medical specialty groups. These same groups are now pushing back against the new USPSTF guidelines,” he added.
Robert L. Barbieri, MD, was not taken aback by the guidelines themselves, but he was surprised by the manner and timing of their release. Dr. Barbieri is Kate Macy Ladd professor of obstetrics, gynecology, and reproductive biology at Harvard Medical School and chief of obstetrics and gynecology at Brigham and Women’s Hospital in Boston. He serves as editor-in-chief of OBG Management.
“I was surprised that the USPSTF did not weigh the potential impact of its analysis on the key stakeholders: patients, disease-based coalitions such as the American Cancer Society and Susan G. Komen for the Cure, and professional societies such as the American College of Radiology and ACOG,” he said. “If I were supervising the process, I would have asked for a comment period before releasing the report. I would have included the comments from key stakeholders in an appendix to the report.”
Are other organizations—besides the USPSTF—likely to change their recommendations for mammography screening in the near future? In the case of ACOG, Dr. Barbieri doesn’t think so.
“I don’t think ACOG will change the age at which to initiate screening,” he said. “I believe it will stick to its recommendation to start screening at 40 and continue every 1 to 2 years from 40 to 50 years of age. However, I could see ACOG becoming a bit more flexible on the question of whether screening should take place at 1- or 2-year intervals after age 50.”
Dr. Kaunitz sees things differently.
“It seems possible that, going forward, the College will give Fellows and their patients permission to implement the new guidelines without mandating their implementation. For example, if women in their 40s wish to defer screening, that would be OK, as would biennial screening for women in their 50s and 60s.”
6. Are the USPSTF recommendations likely to affect insurance coverage?
In a press release issued soon after the new guidelines were published, US Health and Human Services Secretary Kathleen Sebelius addressed Americans directly to reaffirm her support for mammography in women 40 to 49 years old: “There is no question that the US Preventive Services Task Force recommendations have caused a great deal of confusion and worry among women and their families,” her statement read.6 She made it clear that the new recommendations are unlikely to affect federal coverage of mammography.
“The US Preventive Services Task Force is an outside independent panel of doctors and scientists who make recommendations. They do not set federal policy and they don’t determine what services are covered by the federal government,” she said.6
But Dr. Barbieri thinks some changes in insurance coverage are inevitable.
“Any claims that the new guidelines do not represent a major change would be disingenuous,” he said. Because the USPSTF rated its recommendation against mammography for women 40 to 49 years old as grade ‘C,’ that change in guidelines is likely to trigger at least some change in coverage.
“In reality, the ‘C’ rating will require many insurance companies—by their own rule—to stop reimbursing for this screening test,” he said. “The ‘C’ rating means that the test has little benefit.”
ACOG also deems it likely that insurance coverage may be affected for some women.
“Fellows should be aware that the new USPSTF recommendations against routine screening mammography for women aged 40–49 (a grade C recommendation) has implications for insurance coverage, as some insurers will cover only preventive services rated as an ‘A’ or a ‘B’ by the USPSTF. Fellows should counsel their patients that insurance coverage for ‘routine screening’ mammography may become variable and that patients should address this question with their insurers. These recommendations do not apply to high-risk women or patients with clinical findings, and they should be managed accordingly.”5
7. What should you tell your patients?
With all the media attention devoted to the change in guidelines, it’s little surprise that patients are asking questions.
“Patients are aware of the USPSTF report,” said Dr. Barbieri. “They are largely ignoring the recommendations and sticking with annual mammograms.”
“I think, as always, women are looking to their ObGyn for guidance,” added Dr. Kaunitz.
So what are these clinicians telling patients about mammography screening?
As he was to begin with, Dr. Kaunitz is acquiescent if patients prefer to defer mammography screening to their 50s.
“Because it seems that insurance coverage, over the short term, is unlikely to restrict current access to mammograms,” said Dr. Kaunitz, “my evolving philosophy is that the new USPSTF guidelines, along with ACOG and other existing guidelines, give ObGyns and their patients permission to:
- proceed or not proceed with mammograms for women in their 40s, with the decision based on issues such as patient preference, family history of breast cancer, and body mass index (BMI)
- be flexible regarding 1- to 2-year screening intervals among women in their 50s, 60s, and 70s, with the decision based on issues such as patient preference, use or non-use of estrogen-progestin hormone therapy, family history of breast cancer, and BMI.”
Dr. Barbieri believes some effort to integrate the ACOG and USPSTF recommendations is called for. “Accordingly,” he said, “I suggest the following:

I suggest actively recommending biennial mammography for women 40 to 75 years old. Offer annual mammography to women 40 to 75 years old if they prefer that option.
ROBERT L. BARBIERI, MD
- Actively recommend biennial mammography for women 40 to 75 years old. Offer annual mammography to women 40 to 75 years old if they prefer that option.
- Aggressively search for high-risk women, with high risk defined as a lifetime risk of breast cancer exceeding 15%. Among the variables contributing to high-risk status are a history of thoracic radiotherapy, a strong family history of breast cancer, and BRCA mutation. For these women, I would recommend annual mammography and biennial MRI of the breasts.
- Perform annual or biennial clinical breast exam.
- Obtain imaging for any woman who has a palpable breast lump, and resect or biopsy the lump even if that imaging is negative.”
Some clinicians were reconsidering the need for an annual mammogram even before the US Preventive Services Task Force (USPSTF) issued new guidelines late last year.1
Andrew M. Kaunitz, MD, is one of those clinicians. In an editorial in the December issue of OBG Management, he was bold enough to declare: “My plan is to be more acquiescent when a woman says ‘No’ to an annual mammogram.”2
Among the evidence he cited to justify that acquiescence was a recent article in the Journal of the American Medical Association that expressed concern about the high number of early cancers—including ductal carcinoma in situ—that are detected by mammography and treated even though many are unlikely to progress or ever become clinically significant.3 This phenomenon—termed “over-diagnosis”—is one of the risks of breast cancer screening.
Dr. Kaunitz is professor and associate chairman of obstetrics and gynecology at the University of Florida College of Medicine–Jacksonville. He also serves on the OBG Management Board of Editors.
Although the USPSTF is the only official body to revise its recommendations on breast cancer screening so far, more changes seem likely. This article aims to sift through the static on the airwaves of late and offer concrete recommendations for practice. In the process, it addresses seven questions:
- How did USPSTF guidelines change?
- Why did they change?
- Why did the changes attract so much attention?
- What is ACOG’s position?
- What do thought leaders make of the new guidelines?
- Are the USPSTF recommendations likely to affect insurance coverage for mammography?
- What should you tell your patients about breast cancer screening?
1. How did USPSTF guidelines change?
In an article published November 16, the USPSTF made a number of revisions to earlier breast cancer screening guidelines for women at average risk of the disease:

Approximately 39 million women undergo mammography each year in the United States, costing the health-care system more than $5 billion.
- Routine screening mammography is no longer recommended in women 40 to 49 years old. Rather, the decision about when to begin regular screening should be individualized and should “take into account patient context, including the patient’s values regarding specific benefits and harms” (Grade C recommendation).
- Screening mammography in women 50 to 74 years old should be biennial rather than annual (Grade B recommendation).
- Breast self-examination (BSE) is not recommended for any age group (Grade D recommendation).1
2. Why did the USPSTF guidelines change?
The changes were based on new data and analysis in the following areas:
- Mortality among women 40 to 49 years old. Although mammography screening reduces breast cancer mortality by 15% in this age group, the USPSTF concluded that “there is moderate certainty that the net benefit is small” in this population.1,4
- The effectiveness of BSE in decreasing breast cancer mortality among women of any age. Studies of BSE published since 2002 found no significant differences in breast cancer mortality between women who perform BSE and those who don’t.4
- The magnitude of harms of screening with mammography. Mammography screening in women 40 to 49 years old involves a significant risk of harms.4 Although the USPSTF observed that the benefits of mammography in women 40 to 49 years old appear to be equivalent to the benefits of mammography among women 50 to 59 years old, it concluded that the harms outweigh benefits in the younger women.
Harms cited by the USPSTF include:
- radiation exposure
- pain during the procedure
- anxiety and distress
- an increased rate of false-positive results
- greater need for additional imaging and biopsies.4
The USPSTF conceded that the radiation exposure from a mammogram is minimal, but questioned whether cumulative exposure in young women might be problematic. It also noted that “many women experience pain during the procedure (range, 1% to 77%), but few would consider this a deterrent from future screening.”4
As for false-positive results, the group observed: “Data from the [Breast Cancer Screening Consortium (BCSC)] for regularly screened women…indicate that false-positive mammography results are common in all age groups but are most common among women aged 40 to 49 years (97.8 per 1,000 women per screening round).”4
“The BCSC results indicate that for every case of invasive breast cancer detected by mammography screening in women aged 40 to 49 years, 556 women have mammography, 47 have additional imaging, and five have biopsies.”4
It is the significant rate of false positives that creates the need for additional screening, diagnostic imaging, and biopsy. These additional imaging and invasive procedures increase anxiety and distress among many women. The USPSTF concluded that these harms outweighed the benefits of mammography screening in women 40 to 49 years old.
After publication of the new US Preventive Services Task Force (USPSTF) breast cancer screening guidelines late last year, it was only a matter of hours before official bodies and professional organizations began to weigh in on the changes, and the verdict was unanimous—disagreement. Among those chiming in were the American Cancer Society (ACS), the American College of Obstetricians and Gynecologists (ACOG), the American College of Radiology, the American Society of Breast Surgeons, the Society for Breast Imaging (SBI), and Susan G. Komen for the Cure, among others. Here are excerpts from their statements.
American Cancer Society
The ACS immediately refuted the USPSTF recommendations:
The American Cancer Society continues to recommend annual screening using mammography and clinical breast examination for all women beginning at age 40. Our experts make this recommendation having reviewed virtually all the same data reviewed by the USPSTF, but also additional data that the USPSTF did not consider….[T]he American Cancer Society’s medical staff and volunteer experts overwhelmingly believe the benefits of screening women aged 40 to 49 outweigh its limitations.7
ACOG
The College reaffirmed its support for screening mammography every 1 to 2 years in women 40 to 49 years old and every year for women 50 and older, as well as breast self-examination for women of all ages:
At this time, The American College of Obstetricians and Gynecologists recommends that Fellows continue to follow current College guidelines for breast cancer screening. Evaluation of the new USPSTF recommendations is under way. Should the College update its guidelines in the future, Fellows would be alerted and such revised guidelines would be published in Obstetrics & Gynecology.5
American College of Radiology
The College minced no words in opposing the changes:
If cost-cutting US Preventive Services Task Force (USPSTF) mammography recommendations are adopted as policy, two decades of decline in breast cancer mortality could be reversed and countless American women may die needlessly from breast cancer each year.
These new recommendations seem to reflect a conscious decision to ration care. If Medicare and private insurers adopt these incredibly flawed USPSTF recommendations as a rationale for refusing women coverage of these life-saving exams, it could have deadly effects for American women,” said Carol H. Lee, MD, chair of the American College of Radiology Breast Imaging Commission.8
American Society of Breast Surgeons
The organization released a statement describing its position as “strongly opposed” to the USPSTF recommendations:
We believe there is sufficient data to support annual mammography screening for women age 40 and older. We also believe the breast cancer survival rate of women between 40 and 50 will improve from the increased use of digital mammographic screening, which is superior to older plain film techniques in detecting breast cancer in that age group.
While we recognize that there will be a number of benign biopsies, we also recognize that mammography is the optimal screening tool for the early diagnosis of breast cancer in terms of cost-effectiveness, practical use, and accuracy.9
Society for Breast Imaging
In its statement, the SBI noted the confusion caused by revision of the USPSTF guidelines, calling it “unnecessary and potentially deadly”:
Mammography has been shown unequivocally to save lives and is primarily responsible for the 30% decline in breast cancer mortality in the United States over the past 20 years. The USPSTF conclusion—that women under age 50 should not undergo routine screening—conflicts with their own report, which confirms a benefit of mammography to women age 40–49 that is statistically significant.
We strongly urge women and their physicians to adhere to the American Cancer Society recommendations of yearly screening beginning at age 40.10
Susan G. Komen for the Cure
This public advocacy group issued a statement in late November acknowledging “mass confusion and justifiable outrage” in the aftermath of the USPSTF changes:
”We have worked so hard to build public trust and urge people to get screened,” said Nancy G. Brinker, founder of Susan G. Komen for the Cure, “and now they hear that maybe they shouldn’t bother. That is dangerous….Let me say this as clearly as I can: Mammography saves lives, even this report says that. Keep doing what you are doing. And always, talk with your doctor.” Brinker also noted that Komen for the Cure was not changing its guidelines, continuing to recommend annual mammograms beginning at age 40.11
3. Why have the guidelines captured so much media attention?
Most of the controversy that has arisen since publication of the new guidelines has centered on the recommendation against screening mammography in women 40 to 49 years old. A number of media outlets have highlighted women whose breast cancer was detected by screening mammography when they were in their 40s, and many survivors with a similar history have spoken out against the new recommendations.
In addition, the American Cancer Society (ACS), the American College of Radiology, Susan G. Komen for the Cure, and other groups have publicly opposed the new guidelines. (See “Among professional organizations, a resounding chorus of disagreement”)
4. What is ACOG’s position on the new recommendations?
The American College of Obstetricians and Gynecologists (ACOG) was quick to weigh in on the new USPSTF guidelines, emphasizing that the College’s recommendations have not changed. They include:
- screening mammography every 1 to 2 years for women 40 to 49 years old
- screening mammography every year for women 50 years and older
- BSE for all women.
ACOG did note, however, that “the College is continuing to evaluate in detail the new USPSTF recommendations and the new evidence considered by the USPSTF.”5
5. What do thought leaders make of the USPSTF changes?
Although the USPSTF guidelines sparked a firestorm of media coverage, the change did not come as a shock to leaders in the ObGyn specialty.

Legitimate concerns about screening mammography have increasingly been raised by experts in the field.
ANDREW M. KAUNITZ, MD “I was not surprised,” said Dr. Kaunitz. “As I pointed out in my editorial in OBG Management, legitimate concerns about screening mammography have increasingly been raised by experts in the field.2 Proposals to stop routinely screening women in their 40s were made earlier in this decade, but were met with major pushback from the ACS, breast cancer advocacy organizations, and medical specialty groups. These same groups are now pushing back against the new USPSTF guidelines,” he added.
Robert L. Barbieri, MD, was not taken aback by the guidelines themselves, but he was surprised by the manner and timing of their release. Dr. Barbieri is Kate Macy Ladd professor of obstetrics, gynecology, and reproductive biology at Harvard Medical School and chief of obstetrics and gynecology at Brigham and Women’s Hospital in Boston. He serves as editor-in-chief of OBG Management.
“I was surprised that the USPSTF did not weigh the potential impact of its analysis on the key stakeholders: patients, disease-based coalitions such as the American Cancer Society and Susan G. Komen for the Cure, and professional societies such as the American College of Radiology and ACOG,” he said. “If I were supervising the process, I would have asked for a comment period before releasing the report. I would have included the comments from key stakeholders in an appendix to the report.”
Are other organizations—besides the USPSTF—likely to change their recommendations for mammography screening in the near future? In the case of ACOG, Dr. Barbieri doesn’t think so.
“I don’t think ACOG will change the age at which to initiate screening,” he said. “I believe it will stick to its recommendation to start screening at 40 and continue every 1 to 2 years from 40 to 50 years of age. However, I could see ACOG becoming a bit more flexible on the question of whether screening should take place at 1- or 2-year intervals after age 50.”
Dr. Kaunitz sees things differently.
“It seems possible that, going forward, the College will give Fellows and their patients permission to implement the new guidelines without mandating their implementation. For example, if women in their 40s wish to defer screening, that would be OK, as would biennial screening for women in their 50s and 60s.”
6. Are the USPSTF recommendations likely to affect insurance coverage?
In a press release issued soon after the new guidelines were published, US Health and Human Services Secretary Kathleen Sebelius addressed Americans directly to reaffirm her support for mammography in women 40 to 49 years old: “There is no question that the US Preventive Services Task Force recommendations have caused a great deal of confusion and worry among women and their families,” her statement read.6 She made it clear that the new recommendations are unlikely to affect federal coverage of mammography.
“The US Preventive Services Task Force is an outside independent panel of doctors and scientists who make recommendations. They do not set federal policy and they don’t determine what services are covered by the federal government,” she said.6
But Dr. Barbieri thinks some changes in insurance coverage are inevitable.
“Any claims that the new guidelines do not represent a major change would be disingenuous,” he said. Because the USPSTF rated its recommendation against mammography for women 40 to 49 years old as grade ‘C,’ that change in guidelines is likely to trigger at least some change in coverage.
“In reality, the ‘C’ rating will require many insurance companies—by their own rule—to stop reimbursing for this screening test,” he said. “The ‘C’ rating means that the test has little benefit.”
ACOG also deems it likely that insurance coverage may be affected for some women.
“Fellows should be aware that the new USPSTF recommendations against routine screening mammography for women aged 40–49 (a grade C recommendation) has implications for insurance coverage, as some insurers will cover only preventive services rated as an ‘A’ or a ‘B’ by the USPSTF. Fellows should counsel their patients that insurance coverage for ‘routine screening’ mammography may become variable and that patients should address this question with their insurers. These recommendations do not apply to high-risk women or patients with clinical findings, and they should be managed accordingly.”5
7. What should you tell your patients?
With all the media attention devoted to the change in guidelines, it’s little surprise that patients are asking questions.
“Patients are aware of the USPSTF report,” said Dr. Barbieri. “They are largely ignoring the recommendations and sticking with annual mammograms.”
“I think, as always, women are looking to their ObGyn for guidance,” added Dr. Kaunitz.
So what are these clinicians telling patients about mammography screening?
As he was to begin with, Dr. Kaunitz is acquiescent if patients prefer to defer mammography screening to their 50s.
“Because it seems that insurance coverage, over the short term, is unlikely to restrict current access to mammograms,” said Dr. Kaunitz, “my evolving philosophy is that the new USPSTF guidelines, along with ACOG and other existing guidelines, give ObGyns and their patients permission to:
- proceed or not proceed with mammograms for women in their 40s, with the decision based on issues such as patient preference, family history of breast cancer, and body mass index (BMI)
- be flexible regarding 1- to 2-year screening intervals among women in their 50s, 60s, and 70s, with the decision based on issues such as patient preference, use or non-use of estrogen-progestin hormone therapy, family history of breast cancer, and BMI.”
Dr. Barbieri believes some effort to integrate the ACOG and USPSTF recommendations is called for. “Accordingly,” he said, “I suggest the following:

I suggest actively recommending biennial mammography for women 40 to 75 years old. Offer annual mammography to women 40 to 75 years old if they prefer that option.
ROBERT L. BARBIERI, MD
- Actively recommend biennial mammography for women 40 to 75 years old. Offer annual mammography to women 40 to 75 years old if they prefer that option.
- Aggressively search for high-risk women, with high risk defined as a lifetime risk of breast cancer exceeding 15%. Among the variables contributing to high-risk status are a history of thoracic radiotherapy, a strong family history of breast cancer, and BRCA mutation. For these women, I would recommend annual mammography and biennial MRI of the breasts.
- Perform annual or biennial clinical breast exam.
- Obtain imaging for any woman who has a palpable breast lump, and resect or biopsy the lump even if that imaging is negative.”
1. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151:716-726.
2. Kaunitz AM. I’ve been rethinking my zeal for breast cancer screening. OBG Management. 2009;21(12):6-8.
3. Esserman L, Shieh Y, Thompson I. Rethinking screening for breast cancer and prostate cancer. JAMA. 2009;302:1685-1692.
4. Nelson HD, Tyne K, Nalk A, Bougatsos C, Chan BK, Humphrey L. Screening for breast cancer: an update for the US Preventive Services Task Force. Ann Intern Med. 2009;151:727-737.
5. American College of Obstetricians and Gynecologists. Response of the American College of Obstetricians and Gynecologists to new breast cancer screening recommendations from the US Preventive Services Task Force. Available at: http://www.acog.org/from_home/Misc/uspstfResponse.cfm. Accessed Nov. 25, 2009.
6. US Department of Health and Human Services. Secretary Sebelius statement on new breast cancer recommendations [news release]. Nov. 18, 2009. Available at: http://www.hhs.gov/news/press/2009pres/11/20091118a.html. Accessed Dec. 4, 2009.
7. American Cancer Society responds to changes to USPSTF mammography guidelines [news release]. American Cancer Society. Nov. 16, 2009. Available at: http://www.cancer.org/docroot/MED/content/MED_2_1x_American_Cancer_Society_
Responds_to_Changes_to_USPSTF_Mammography_Guidelines.asp. Accessed Dec. 4, 2009.
8. American College of Radiology. USPSTF mammography recommendations will result in countless unnecessary breast cancer deaths each year [news release]. Nov. 16, 2009. Available at: www.acr.org/MainMenuCategories/media_room/FeaturedCategories/PressReleases/USPSTFMammoRecs.aspx. Accessed Dec. 4, 2009.
9. American Society of Breast Surgeons. Society responds to USPSTF changes in mammography guidelines [news release]. Available at: http://www.breastsurgeons.org/news/article.php?id=57. Accessed Dec. 4, 2009.
10. Society of Breast Imaging. Official Society of Breast Imaging response to the announcement by HHS Secretary Sebelius regarding USPSTF mammography recommendations. Available at:http://www.sbi-online.org/associations/8199/files/OFFICIAL%20SOCIETY%20OF%20
BREAST%20IMAGING%20RESPONSE%20TO%20THE%20ANNOUNCEMENT%20
BY%20HHS%20SECRETARY%20
SEBELIUS%20REGARDING%20USPSTF%20MAMMOGRAPHY%20RECOMMENDATIONS.pdf. Accessed Dec. 4, 2009.
11. Susan G. Komen for the Cure founder, Nancy G. Brinker, calls new mammography guidelines a “set back”; makes call to action [news release]. Nov. 23, 2009. Available at: http://ww5.komen.org/KomenNewsArticle.aspx?id=6442451516. Accessed Dec. 7, 2009.
1. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;151:716-726.
2. Kaunitz AM. I’ve been rethinking my zeal for breast cancer screening. OBG Management. 2009;21(12):6-8.
3. Esserman L, Shieh Y, Thompson I. Rethinking screening for breast cancer and prostate cancer. JAMA. 2009;302:1685-1692.
4. Nelson HD, Tyne K, Nalk A, Bougatsos C, Chan BK, Humphrey L. Screening for breast cancer: an update for the US Preventive Services Task Force. Ann Intern Med. 2009;151:727-737.
5. American College of Obstetricians and Gynecologists. Response of the American College of Obstetricians and Gynecologists to new breast cancer screening recommendations from the US Preventive Services Task Force. Available at: http://www.acog.org/from_home/Misc/uspstfResponse.cfm. Accessed Nov. 25, 2009.
6. US Department of Health and Human Services. Secretary Sebelius statement on new breast cancer recommendations [news release]. Nov. 18, 2009. Available at: http://www.hhs.gov/news/press/2009pres/11/20091118a.html. Accessed Dec. 4, 2009.
7. American Cancer Society responds to changes to USPSTF mammography guidelines [news release]. American Cancer Society. Nov. 16, 2009. Available at: http://www.cancer.org/docroot/MED/content/MED_2_1x_American_Cancer_Society_
Responds_to_Changes_to_USPSTF_Mammography_Guidelines.asp. Accessed Dec. 4, 2009.
8. American College of Radiology. USPSTF mammography recommendations will result in countless unnecessary breast cancer deaths each year [news release]. Nov. 16, 2009. Available at: www.acr.org/MainMenuCategories/media_room/FeaturedCategories/PressReleases/USPSTFMammoRecs.aspx. Accessed Dec. 4, 2009.
9. American Society of Breast Surgeons. Society responds to USPSTF changes in mammography guidelines [news release]. Available at: http://www.breastsurgeons.org/news/article.php?id=57. Accessed Dec. 4, 2009.
10. Society of Breast Imaging. Official Society of Breast Imaging response to the announcement by HHS Secretary Sebelius regarding USPSTF mammography recommendations. Available at:http://www.sbi-online.org/associations/8199/files/OFFICIAL%20SOCIETY%20OF%20
BREAST%20IMAGING%20RESPONSE%20TO%20THE%20ANNOUNCEMENT%20
BY%20HHS%20SECRETARY%20
SEBELIUS%20REGARDING%20USPSTF%20MAMMOGRAPHY%20RECOMMENDATIONS.pdf. Accessed Dec. 4, 2009.
11. Susan G. Komen for the Cure founder, Nancy G. Brinker, calls new mammography guidelines a “set back”; makes call to action [news release]. Nov. 23, 2009. Available at: http://ww5.komen.org/KomenNewsArticle.aspx?id=6442451516. Accessed Dec. 7, 2009.
PRENATAL COUNSELING
Three important areas of research into stillbirth have evolved over the past year, furthering our understanding of the phenomenon and our ability to provide comprehensive, evidence-based care:
- Genetic studies. Karyotype analysis is useful in determining the cause of stillbirth, especially when analysis is based on a sample of amniotic fluid that was obtained before delivery. And array-based comparative genomic hybridization, which yields information on the chromosome count as well as micro-duplications and deletions, can be performed on nondividing cells.
- Risk factors. Further investigation implicates advanced maternal age, obesity, and African-American race.
- Classification. Paring down the more than three dozen systems that exist for classification of stillbirth was the main challenge addressed by an international consensus group in 2009 and the focus of a separate analysis.
The individual studies that contribute to our knowledge base in these areas are discussed in more detail in the articles that follow.
Stillbirth is broadly defined as fetal demise after 20 weeks’ gestation and with a fetal weight exceeding 350 g. In the United States, stillbirth occurs in 1 of every 160 live births (6 stillbirths for every 1,000 live births). Although the rate of neonatal demise has decreased over the past decade, the rate of stillbirth has declined less strikingly.
For an analysis of karyotype, amniotic fluid is best
Korteweg FJ, Bouman K, Erwich JJ, et al. Cytogenetic analysis after evaluation of 750 fetal deaths: proposal for diagnosis workup. Obstet Gynecol. 2008;111:865–874.
ACOG Practice Bulletin #102: Management of stillbirth. Obstet Gynecol. 2009;113:748–760.
When stillbirth occurs, determination of the cause of death fulfills several goals:
- It informs counseling of the parents, who must come to terms with the loss
- It aids in determining the risk of recurrence, which informs family planning
- It furthers research into stillbirth and facilitates the comparison of national and international data.
Chromosomal anomaly is one potential cause of stillbirth. Its frequency depends on the presence of structural malformation. For example, Korteweg and colleagues found a rate of chromosomal anomaly of 4.6% among stillbirths involving fetuses without structural abnormality, but the rate rose to 38% when anatomic malformation was present. The distribution of chromosomes among stillbirths mirrored the pattern seen in live births, including 45, X and trisomies of chromosome 21, 13, and 18.
The utility of karyotype assessment when ultrasonography (US) has not identified structural malformation has been debated. Given the 5% incidence of chromosomal anomaly in the absence of structural abnormality, and the limitations of US in detecting subtle dysmorphology, a karyotype seems advisable to assess all stillbirths.
Comparison of methods points to superiority of amniocentesis
Because fewer than 20% of skin biopsies result in a useful culture, postmortem skin biopsy for karyotype assessment is unreliable. Korteweg and colleagues evaluated other methods of obtaining cells for examination and found that a successful karyotype is most likely with predelivery amniocentesis (85%), followed by umbilical cord culture (32.1%). A karyotype of cells from fascia lata and skin biopsy yielded poor results, especially in the setting of maceration. Placental biopsy is likely to provide an adequate karyotype (71% probability) but findings may be confounded by confined placental mosaicism.1
ACOG also advocates predelivery amniocentesis
In its 2009 practice bulletin, ACOG supported inclusion of amniocentesis in the assessment of stillbirth and preparation for delivery. Once an epidural is placed, amniocentesis provides cells for karyotype assessment, polymerase chain reaction (PCR) for viral studies, and any other metabolic or specific genetic studies that may be indicated by fetopsy.
If amniocentesis is not performed, ACOG recommends umbilical cord culture as an alternative. Because nondividing cells can be utilized in fluorescence in situ hybridization (FISH) for chromosome 13, 18, 21, X, and Y, this method should be considered in any case involving culture failure (TABLE).2
TABLE
Genetic components of stillbirth assessment
| Type of assessment | Steps |
|---|---|
| Inspection of fetus and placenta | Measure head circumference and length of fetus |
| Weigh fetus and placenta | |
| Photograph fetus and placenta, including frontal and profile shots of whole body, face, extremities, palms, and any abnormality | |
| Document findings | |
| Cytologic analysis | Obtain consent from parents |
| Obtain acceptable specimens using one of the following sterile techniques: | |
| • Amniocentesis at the time of prenatal diagnosis of demise • Placental block (1 x 1 cm) taken from below the cord-insertion site on the unfixed placenta • Umbilical cord segment (1.5 cm) • Internal fetal tissue specimen, e.g., costochondral junction or patella (not skin) | |
| Preserve specimens in a sterile culture medium of lactated Ringer’s solution at room temperature during transfer to laboratory | |
| Fetopsy | Obtain parental consent; if no consent is given, send placenta for pathologic analysis |
| Perform autopsy and pathologic assessment of the placenta | |
| Consider whole-body fetal radiographs | |
| Source: ACOG Practice Bulletin #102 | |
Perform predelivery amniocentesis whenever possible at the time of diagnosis of demise to obtain a cell sample for karyotype analysis to determine the cause of death.
Array-based comparative genomic hybridization makes assessment of nondividing cells possible
Raca G, Artzer A, Thorson L, et al. Array-based comparative hybridization (aCGH) in the genetic evaluation of stillbirth. Am J Med Genet A. 2009;149A:2437–2443.
Array-based comparative genomic hybridization (aCGH) makes it possible to assess the chromosome count and perform a high-resolution search for microduplications and deletions. With known segments of the genome printed on slides, the clinical scientist can analyze DNA from nondividing cells from a stillbirth. The ability to use nondividing cells is important because no cell culture is required. (Cell culture is often difficult to obtain after stillbirth.) Depending on the array selected, the resolution can be as fine as a single nucleotide polymorphism.
aCGH can inform preconception counseling
Raca and colleagues used a range of arrays to assess 15 stillbirths that involved two or more malformations. Chromosomal abnormalities, including trisomy 21 and an unbalanced translocation, were detected by aCGH in two infants. Identification of these abnormalities helped inform counseling of the parents:
- In the case of trisomy 21, parental karyotypes revealed a nontranslocation event, making it possible to assure the parents that the risk of recurrence is low
- The unbalanced translocation resulted from a balanced chromosome translocation in the mother and was associated with a significant risk of recurrence (in this case, FISH would not have helped because chromosomes 13, 18, 21, X, and Y were not involved).
Limitations of aCGH
One limitation is an inability to detect polyploidy such as triploidy or tetraploidy. This problem can be circumvented through the use of a FISH preparation prior to aCGH.
In most centers, parental blood samples are drawn at the time of aCGH studies. Because aCGH offers greater resolution of chromosome regions, an increasing number of benign variations (i.e., present in one parent) are being identified. As aCGH technology advances, we are accumulating data on copy-number variations.
A large clinical trial is needed to assess the full potential of aCGH in this setting.
Use of array-basic comparative genomic hybridization to assess cells from a stillborn fetus can help determine the cause of death and inform counseling of the parents about the risk of recurrence.
Risk factors for stillbirth include
advanced maternal age, obesity, and black race
ACOG Practice Bulletin #102: Management of stillbirth. Obstet Gynecol. 2009;113:748–760.
Willinger M, Ko CW, Reddy UM. Racial disparities in stillbirth risk across gestation in the United States. Am J Obstet Gynecol. 2009;201:469.e1–469.e8.
Fretts RC. The study of stillbirth. Am J Obstet Gynecol. 2009;201:429–430.
Women who have diseases such as insulin-dependent diabetes and systemic lupus erythematosus have long been recognized as having a six- to 20-fold increase in the risk of stillbirth, compared with the general population. However, each of these disorders accounts for 2% and less than 1% of the pregnant population, respectively, so their overall contribution to stillbirth is small. Larger portions of the population have a lower—but still significant—risk of stillbirth:
- women older than 35 years
- women who have a body mass index (BMI) above 30
- non-Hispanic black women.
Each of these categories represents 15% or more of the typical obstetric population, and each group faces a risk of stillbirth approaching 1%. The ACOG practice bulletin and the study by Willinger and colleagues address these risks in detail.
Advanced maternal age is particularly risky among nulliparous women
Advanced maternal age (>35 years) is associated with increased rates of chromosomal abnormality and maternal morbidity, such as hypertension, that are known to raise the risk of stillbirth. Even when these and other variables associated with advanced maternal age, such as placenta previa, diabetes, and multiple gestation, are controlled, however, the increased risk of stillbirth remains.
Advanced maternal age in a first pregnancy carries a particularly elevated risk. For example, the risk of stillbirth in a 40-year-old nulliparous woman is more than twice the risk in a 40-year-old multiparous woman (1 in every 116 pregnancies vs 1 in every 304).3
The increased risk of stillbirth associated with advanced maternal age is present at all gestational ages, though it becomes most profound at 37 to 42 weeks’ gestation, notably for:
- women 35 to 39 years old (1 in every 382 pregnancies; relative risk [RR] of 1.32, compared with women <35 years old; 95% confidence interval [CI], 1.22, 1.43)
- women >40 years old (1 in every 267 pregnancies; RR, 1.88; 95% CI, 1.64, 2.16).
These numbers remain significant even after controlling for medical conditions.3
The utility of antepartum surveillance and induction of labor for delivery is unclear, given the risk of iatrogenic prematurity.
Risk of stillbirth is doubled among obese and markedly obese women
Although the number of adults who are overweight (BMI 25–30) has remained fairly constant over the past 20 years (30% to 35% of the population), the percentage of women of reproductive age who are obese (BMI >30) has risen markedly. Obesity is now present in 35% of the population, and marked obesity (BMI >40) affects an additional 6%. Both obese and markedly obese women face a twofold relative risk of stillbirth, compared with women of normal weight. The rate of stillbirth in this population is 12 to 18 for every 1,000 births—a 1.2% to 1.8% risk.
Although obesity-related stillbirth likely has multiple causes, the risk remains elevated even after exclusion of confounding factors such as smoking, gestational diabetes, and preeclampsia.
Race is an independent contributor
Racial differences in the rate of stillbirth remain despite a decrease in the overall stillbirth rate over the past 20 years ( FIGURE ). In 2003, the rate of stillbirth was 5 for every 1,000 births among non-Hispanic whites, 5.5 among Hispanics, and 12 among non-Hispanic blacks. In other words, the risk of stillbirth was 1 in 202, 1 in 183, and 1 in 87 births for white, Hispanic, and black women, respectively.
Willinger and colleagues utilized data from the National Center for Health Statistics and assessed 2001–2002 birth and infant death datasets for 36 states, examining the stillbirth hazard risk for more than 5 million singleton pregnancies. Stillbirth peaked at 20 to 23 weeks and 39 to 41 weeks’ gestation, as expected. However, at 20 to 23 weeks, the risk of stillbirth among non-Hispanic black women was more than twice the rate for non-Hispanic white women (RR, 2.8). Although it then declined as term approached, it remained greater than that of non-Hispanic white women (RR, 1.6).
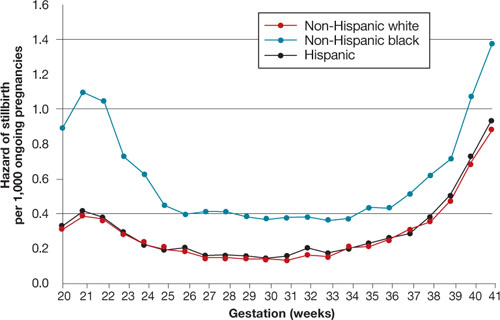
FIGURE Racial disparities in the risk of stillbirth
Hazard of stillbirth for singleton pregnancies by gestational age and race and ethnicity, 2001–2002. SOURCE: Willinger et al. Greater acceptance and use of induction of labor at term among whites merits attention
In an editorial accompanying the study by Willinger and colleagues, Fretts pointed out the higher rate of induction of labor at term among white women that has been observed in at least three studies of vital statistics. (Willinger and colleagues also pointed out this difference.) The acceptance and use of labor induction at term—and the lower stillbirth rate—among white women warrants further investigation.
Education appears to reduce the risk of stillbirth to a greater degree among whites than it does among blacks. Again, nulliparity and advanced maternal age were important contributors to the risk of stillbirth across all three races.
Counsel African-American gravidas and women older than 35 years that their risk of stillbirth is elevated.
Obese women should be advised to lose weight before conception if at all possible to reduce the risk of stillbirth.
Needed: Standardized analysis
and documentation of stillbirth
Reddy UM, Goldberg R, Silver R, et al. Stillbirth classification—developing an international consensus for research: executive summary of a National Institute of Child Health and Human Development workshop. Obstet Gynecol. 2009;114:901–914.
Flenady V, Frøen JF, Pinar H, et al. An evaluation of classification systems for stillbirth. BMC Pregnancy Childbirth. 2009;9:24.
Further guidance for the clinical management of stillbirth will come from investigations of the underlying pathologies and associated risk factors. Key to development of this guidance is the involvement of obstetricians in documenting the antenatal record and delivery information. Also needed is a standardized system for recording this information. More than three dozen systems have been developed to classify stillbirth, at the expense of uniformity of content.
An international consensus group published guidelines on how to describe the cause of death in research endeavors, recognizing the need to maintain the ability to attach a level of uncertainty. In addition, Flenady and colleagues compared the most widely used systems in clinical practice, assigning the highest score for components such as ease of use, inter observer variability, and proportion of unexplained stillbirths to CODAC [cause of death and two associated causes]. This system assigns a primary cause of death from a specified list of choices and allows inclusion of two possible contributing causes.
Both the international consensus classification and the CODAC scoring system are accessible through links embedded within the articles. Both systems require the establishment of standardized evaluation and review of stillbirth that should include obstetricians, pathologists, and geneticists.
Because assessment and classification of stillbirth are fundamental to its prevention, as well as a critical part of clinical practice, ObGyns should become familiar with the international consensus classification and CODAC scoring systems and adopt a standardized approach to assessment and documentation.
1. Rodgers CS, Creasy MR, Fitchett M, Maliszewska CT, Pratt NR, Waters JJ. Solid tissue culture for cytogenetic analysis: a collaborative survey for the Association of Clinical Cytogeneticists. J Clin Pathol. 1996;49:638-641.
2. Rivasi F, Schirosi L, Bettelli S, et al. FISH analysis in cell touch preparations and cytological specimens from formalin-fixed fetal autopsies. Diagn Cytopathol. 2008;36:633-636.
3. Reddy UM, Ko CW, Willinger M. Maternal age and the risk of stillbirth throughout pregnancy in the United States. Am J Obstet Gynecol. 2006;195:764-770.
4. MacDorman MF, Mathews TJ. NCHS Data Brief #9: Recent trends in infant mortality in the United States. Atlanta, Ga: National Center for Health Statistics; October 2008. Available at: http://www.cdc.gov/nchs/data/databriefs/db09.htm. Accessed Dec. 15, 2009.
Three important areas of research into stillbirth have evolved over the past year, furthering our understanding of the phenomenon and our ability to provide comprehensive, evidence-based care:
- Genetic studies. Karyotype analysis is useful in determining the cause of stillbirth, especially when analysis is based on a sample of amniotic fluid that was obtained before delivery. And array-based comparative genomic hybridization, which yields information on the chromosome count as well as micro-duplications and deletions, can be performed on nondividing cells.
- Risk factors. Further investigation implicates advanced maternal age, obesity, and African-American race.
- Classification. Paring down the more than three dozen systems that exist for classification of stillbirth was the main challenge addressed by an international consensus group in 2009 and the focus of a separate analysis.
The individual studies that contribute to our knowledge base in these areas are discussed in more detail in the articles that follow.
Stillbirth is broadly defined as fetal demise after 20 weeks’ gestation and with a fetal weight exceeding 350 g. In the United States, stillbirth occurs in 1 of every 160 live births (6 stillbirths for every 1,000 live births). Although the rate of neonatal demise has decreased over the past decade, the rate of stillbirth has declined less strikingly.
For an analysis of karyotype, amniotic fluid is best
Korteweg FJ, Bouman K, Erwich JJ, et al. Cytogenetic analysis after evaluation of 750 fetal deaths: proposal for diagnosis workup. Obstet Gynecol. 2008;111:865–874.
ACOG Practice Bulletin #102: Management of stillbirth. Obstet Gynecol. 2009;113:748–760.
When stillbirth occurs, determination of the cause of death fulfills several goals:
- It informs counseling of the parents, who must come to terms with the loss
- It aids in determining the risk of recurrence, which informs family planning
- It furthers research into stillbirth and facilitates the comparison of national and international data.
Chromosomal anomaly is one potential cause of stillbirth. Its frequency depends on the presence of structural malformation. For example, Korteweg and colleagues found a rate of chromosomal anomaly of 4.6% among stillbirths involving fetuses without structural abnormality, but the rate rose to 38% when anatomic malformation was present. The distribution of chromosomes among stillbirths mirrored the pattern seen in live births, including 45, X and trisomies of chromosome 21, 13, and 18.
The utility of karyotype assessment when ultrasonography (US) has not identified structural malformation has been debated. Given the 5% incidence of chromosomal anomaly in the absence of structural abnormality, and the limitations of US in detecting subtle dysmorphology, a karyotype seems advisable to assess all stillbirths.
Comparison of methods points to superiority of amniocentesis
Because fewer than 20% of skin biopsies result in a useful culture, postmortem skin biopsy for karyotype assessment is unreliable. Korteweg and colleagues evaluated other methods of obtaining cells for examination and found that a successful karyotype is most likely with predelivery amniocentesis (85%), followed by umbilical cord culture (32.1%). A karyotype of cells from fascia lata and skin biopsy yielded poor results, especially in the setting of maceration. Placental biopsy is likely to provide an adequate karyotype (71% probability) but findings may be confounded by confined placental mosaicism.1
ACOG also advocates predelivery amniocentesis
In its 2009 practice bulletin, ACOG supported inclusion of amniocentesis in the assessment of stillbirth and preparation for delivery. Once an epidural is placed, amniocentesis provides cells for karyotype assessment, polymerase chain reaction (PCR) for viral studies, and any other metabolic or specific genetic studies that may be indicated by fetopsy.
If amniocentesis is not performed, ACOG recommends umbilical cord culture as an alternative. Because nondividing cells can be utilized in fluorescence in situ hybridization (FISH) for chromosome 13, 18, 21, X, and Y, this method should be considered in any case involving culture failure (TABLE).2
TABLE
Genetic components of stillbirth assessment
| Type of assessment | Steps |
|---|---|
| Inspection of fetus and placenta | Measure head circumference and length of fetus |
| Weigh fetus and placenta | |
| Photograph fetus and placenta, including frontal and profile shots of whole body, face, extremities, palms, and any abnormality | |
| Document findings | |
| Cytologic analysis | Obtain consent from parents |
| Obtain acceptable specimens using one of the following sterile techniques: | |
| • Amniocentesis at the time of prenatal diagnosis of demise • Placental block (1 x 1 cm) taken from below the cord-insertion site on the unfixed placenta • Umbilical cord segment (1.5 cm) • Internal fetal tissue specimen, e.g., costochondral junction or patella (not skin) | |
| Preserve specimens in a sterile culture medium of lactated Ringer’s solution at room temperature during transfer to laboratory | |
| Fetopsy | Obtain parental consent; if no consent is given, send placenta for pathologic analysis |
| Perform autopsy and pathologic assessment of the placenta | |
| Consider whole-body fetal radiographs | |
| Source: ACOG Practice Bulletin #102 | |
Perform predelivery amniocentesis whenever possible at the time of diagnosis of demise to obtain a cell sample for karyotype analysis to determine the cause of death.
Array-based comparative genomic hybridization makes assessment of nondividing cells possible
Raca G, Artzer A, Thorson L, et al. Array-based comparative hybridization (aCGH) in the genetic evaluation of stillbirth. Am J Med Genet A. 2009;149A:2437–2443.
Array-based comparative genomic hybridization (aCGH) makes it possible to assess the chromosome count and perform a high-resolution search for microduplications and deletions. With known segments of the genome printed on slides, the clinical scientist can analyze DNA from nondividing cells from a stillbirth. The ability to use nondividing cells is important because no cell culture is required. (Cell culture is often difficult to obtain after stillbirth.) Depending on the array selected, the resolution can be as fine as a single nucleotide polymorphism.
aCGH can inform preconception counseling
Raca and colleagues used a range of arrays to assess 15 stillbirths that involved two or more malformations. Chromosomal abnormalities, including trisomy 21 and an unbalanced translocation, were detected by aCGH in two infants. Identification of these abnormalities helped inform counseling of the parents:
- In the case of trisomy 21, parental karyotypes revealed a nontranslocation event, making it possible to assure the parents that the risk of recurrence is low
- The unbalanced translocation resulted from a balanced chromosome translocation in the mother and was associated with a significant risk of recurrence (in this case, FISH would not have helped because chromosomes 13, 18, 21, X, and Y were not involved).
Limitations of aCGH
One limitation is an inability to detect polyploidy such as triploidy or tetraploidy. This problem can be circumvented through the use of a FISH preparation prior to aCGH.
In most centers, parental blood samples are drawn at the time of aCGH studies. Because aCGH offers greater resolution of chromosome regions, an increasing number of benign variations (i.e., present in one parent) are being identified. As aCGH technology advances, we are accumulating data on copy-number variations.
A large clinical trial is needed to assess the full potential of aCGH in this setting.
Use of array-basic comparative genomic hybridization to assess cells from a stillborn fetus can help determine the cause of death and inform counseling of the parents about the risk of recurrence.
Risk factors for stillbirth include
advanced maternal age, obesity, and black race
ACOG Practice Bulletin #102: Management of stillbirth. Obstet Gynecol. 2009;113:748–760.
Willinger M, Ko CW, Reddy UM. Racial disparities in stillbirth risk across gestation in the United States. Am J Obstet Gynecol. 2009;201:469.e1–469.e8.
Fretts RC. The study of stillbirth. Am J Obstet Gynecol. 2009;201:429–430.
Women who have diseases such as insulin-dependent diabetes and systemic lupus erythematosus have long been recognized as having a six- to 20-fold increase in the risk of stillbirth, compared with the general population. However, each of these disorders accounts for 2% and less than 1% of the pregnant population, respectively, so their overall contribution to stillbirth is small. Larger portions of the population have a lower—but still significant—risk of stillbirth:
- women older than 35 years
- women who have a body mass index (BMI) above 30
- non-Hispanic black women.
Each of these categories represents 15% or more of the typical obstetric population, and each group faces a risk of stillbirth approaching 1%. The ACOG practice bulletin and the study by Willinger and colleagues address these risks in detail.
Advanced maternal age is particularly risky among nulliparous women
Advanced maternal age (>35 years) is associated with increased rates of chromosomal abnormality and maternal morbidity, such as hypertension, that are known to raise the risk of stillbirth. Even when these and other variables associated with advanced maternal age, such as placenta previa, diabetes, and multiple gestation, are controlled, however, the increased risk of stillbirth remains.
Advanced maternal age in a first pregnancy carries a particularly elevated risk. For example, the risk of stillbirth in a 40-year-old nulliparous woman is more than twice the risk in a 40-year-old multiparous woman (1 in every 116 pregnancies vs 1 in every 304).3
The increased risk of stillbirth associated with advanced maternal age is present at all gestational ages, though it becomes most profound at 37 to 42 weeks’ gestation, notably for:
- women 35 to 39 years old (1 in every 382 pregnancies; relative risk [RR] of 1.32, compared with women <35 years old; 95% confidence interval [CI], 1.22, 1.43)
- women >40 years old (1 in every 267 pregnancies; RR, 1.88; 95% CI, 1.64, 2.16).
These numbers remain significant even after controlling for medical conditions.3
The utility of antepartum surveillance and induction of labor for delivery is unclear, given the risk of iatrogenic prematurity.
Risk of stillbirth is doubled among obese and markedly obese women
Although the number of adults who are overweight (BMI 25–30) has remained fairly constant over the past 20 years (30% to 35% of the population), the percentage of women of reproductive age who are obese (BMI >30) has risen markedly. Obesity is now present in 35% of the population, and marked obesity (BMI >40) affects an additional 6%. Both obese and markedly obese women face a twofold relative risk of stillbirth, compared with women of normal weight. The rate of stillbirth in this population is 12 to 18 for every 1,000 births—a 1.2% to 1.8% risk.
Although obesity-related stillbirth likely has multiple causes, the risk remains elevated even after exclusion of confounding factors such as smoking, gestational diabetes, and preeclampsia.
Race is an independent contributor
Racial differences in the rate of stillbirth remain despite a decrease in the overall stillbirth rate over the past 20 years ( FIGURE ). In 2003, the rate of stillbirth was 5 for every 1,000 births among non-Hispanic whites, 5.5 among Hispanics, and 12 among non-Hispanic blacks. In other words, the risk of stillbirth was 1 in 202, 1 in 183, and 1 in 87 births for white, Hispanic, and black women, respectively.
Willinger and colleagues utilized data from the National Center for Health Statistics and assessed 2001–2002 birth and infant death datasets for 36 states, examining the stillbirth hazard risk for more than 5 million singleton pregnancies. Stillbirth peaked at 20 to 23 weeks and 39 to 41 weeks’ gestation, as expected. However, at 20 to 23 weeks, the risk of stillbirth among non-Hispanic black women was more than twice the rate for non-Hispanic white women (RR, 2.8). Although it then declined as term approached, it remained greater than that of non-Hispanic white women (RR, 1.6).

FIGURE Racial disparities in the risk of stillbirth
Hazard of stillbirth for singleton pregnancies by gestational age and race and ethnicity, 2001–2002. SOURCE: Willinger et al. Greater acceptance and use of induction of labor at term among whites merits attention
In an editorial accompanying the study by Willinger and colleagues, Fretts pointed out the higher rate of induction of labor at term among white women that has been observed in at least three studies of vital statistics. (Willinger and colleagues also pointed out this difference.) The acceptance and use of labor induction at term—and the lower stillbirth rate—among white women warrants further investigation.
Education appears to reduce the risk of stillbirth to a greater degree among whites than it does among blacks. Again, nulliparity and advanced maternal age were important contributors to the risk of stillbirth across all three races.
Counsel African-American gravidas and women older than 35 years that their risk of stillbirth is elevated.
Obese women should be advised to lose weight before conception if at all possible to reduce the risk of stillbirth.
Needed: Standardized analysis
and documentation of stillbirth
Reddy UM, Goldberg R, Silver R, et al. Stillbirth classification—developing an international consensus for research: executive summary of a National Institute of Child Health and Human Development workshop. Obstet Gynecol. 2009;114:901–914.
Flenady V, Frøen JF, Pinar H, et al. An evaluation of classification systems for stillbirth. BMC Pregnancy Childbirth. 2009;9:24.
Further guidance for the clinical management of stillbirth will come from investigations of the underlying pathologies and associated risk factors. Key to development of this guidance is the involvement of obstetricians in documenting the antenatal record and delivery information. Also needed is a standardized system for recording this information. More than three dozen systems have been developed to classify stillbirth, at the expense of uniformity of content.
An international consensus group published guidelines on how to describe the cause of death in research endeavors, recognizing the need to maintain the ability to attach a level of uncertainty. In addition, Flenady and colleagues compared the most widely used systems in clinical practice, assigning the highest score for components such as ease of use, inter observer variability, and proportion of unexplained stillbirths to CODAC [cause of death and two associated causes]. This system assigns a primary cause of death from a specified list of choices and allows inclusion of two possible contributing causes.
Both the international consensus classification and the CODAC scoring system are accessible through links embedded within the articles. Both systems require the establishment of standardized evaluation and review of stillbirth that should include obstetricians, pathologists, and geneticists.
Because assessment and classification of stillbirth are fundamental to its prevention, as well as a critical part of clinical practice, ObGyns should become familiar with the international consensus classification and CODAC scoring systems and adopt a standardized approach to assessment and documentation.
Three important areas of research into stillbirth have evolved over the past year, furthering our understanding of the phenomenon and our ability to provide comprehensive, evidence-based care:
- Genetic studies. Karyotype analysis is useful in determining the cause of stillbirth, especially when analysis is based on a sample of amniotic fluid that was obtained before delivery. And array-based comparative genomic hybridization, which yields information on the chromosome count as well as micro-duplications and deletions, can be performed on nondividing cells.
- Risk factors. Further investigation implicates advanced maternal age, obesity, and African-American race.
- Classification. Paring down the more than three dozen systems that exist for classification of stillbirth was the main challenge addressed by an international consensus group in 2009 and the focus of a separate analysis.
The individual studies that contribute to our knowledge base in these areas are discussed in more detail in the articles that follow.
Stillbirth is broadly defined as fetal demise after 20 weeks’ gestation and with a fetal weight exceeding 350 g. In the United States, stillbirth occurs in 1 of every 160 live births (6 stillbirths for every 1,000 live births). Although the rate of neonatal demise has decreased over the past decade, the rate of stillbirth has declined less strikingly.
For an analysis of karyotype, amniotic fluid is best
Korteweg FJ, Bouman K, Erwich JJ, et al. Cytogenetic analysis after evaluation of 750 fetal deaths: proposal for diagnosis workup. Obstet Gynecol. 2008;111:865–874.
ACOG Practice Bulletin #102: Management of stillbirth. Obstet Gynecol. 2009;113:748–760.
When stillbirth occurs, determination of the cause of death fulfills several goals:
- It informs counseling of the parents, who must come to terms with the loss
- It aids in determining the risk of recurrence, which informs family planning
- It furthers research into stillbirth and facilitates the comparison of national and international data.
Chromosomal anomaly is one potential cause of stillbirth. Its frequency depends on the presence of structural malformation. For example, Korteweg and colleagues found a rate of chromosomal anomaly of 4.6% among stillbirths involving fetuses without structural abnormality, but the rate rose to 38% when anatomic malformation was present. The distribution of chromosomes among stillbirths mirrored the pattern seen in live births, including 45, X and trisomies of chromosome 21, 13, and 18.
The utility of karyotype assessment when ultrasonography (US) has not identified structural malformation has been debated. Given the 5% incidence of chromosomal anomaly in the absence of structural abnormality, and the limitations of US in detecting subtle dysmorphology, a karyotype seems advisable to assess all stillbirths.
Comparison of methods points to superiority of amniocentesis
Because fewer than 20% of skin biopsies result in a useful culture, postmortem skin biopsy for karyotype assessment is unreliable. Korteweg and colleagues evaluated other methods of obtaining cells for examination and found that a successful karyotype is most likely with predelivery amniocentesis (85%), followed by umbilical cord culture (32.1%). A karyotype of cells from fascia lata and skin biopsy yielded poor results, especially in the setting of maceration. Placental biopsy is likely to provide an adequate karyotype (71% probability) but findings may be confounded by confined placental mosaicism.1
ACOG also advocates predelivery amniocentesis
In its 2009 practice bulletin, ACOG supported inclusion of amniocentesis in the assessment of stillbirth and preparation for delivery. Once an epidural is placed, amniocentesis provides cells for karyotype assessment, polymerase chain reaction (PCR) for viral studies, and any other metabolic or specific genetic studies that may be indicated by fetopsy.
If amniocentesis is not performed, ACOG recommends umbilical cord culture as an alternative. Because nondividing cells can be utilized in fluorescence in situ hybridization (FISH) for chromosome 13, 18, 21, X, and Y, this method should be considered in any case involving culture failure (TABLE).2
TABLE
Genetic components of stillbirth assessment
| Type of assessment | Steps |
|---|---|
| Inspection of fetus and placenta | Measure head circumference and length of fetus |
| Weigh fetus and placenta | |
| Photograph fetus and placenta, including frontal and profile shots of whole body, face, extremities, palms, and any abnormality | |
| Document findings | |
| Cytologic analysis | Obtain consent from parents |
| Obtain acceptable specimens using one of the following sterile techniques: | |
| • Amniocentesis at the time of prenatal diagnosis of demise • Placental block (1 x 1 cm) taken from below the cord-insertion site on the unfixed placenta • Umbilical cord segment (1.5 cm) • Internal fetal tissue specimen, e.g., costochondral junction or patella (not skin) | |
| Preserve specimens in a sterile culture medium of lactated Ringer’s solution at room temperature during transfer to laboratory | |
| Fetopsy | Obtain parental consent; if no consent is given, send placenta for pathologic analysis |
| Perform autopsy and pathologic assessment of the placenta | |
| Consider whole-body fetal radiographs | |
| Source: ACOG Practice Bulletin #102 | |
Perform predelivery amniocentesis whenever possible at the time of diagnosis of demise to obtain a cell sample for karyotype analysis to determine the cause of death.
Array-based comparative genomic hybridization makes assessment of nondividing cells possible
Raca G, Artzer A, Thorson L, et al. Array-based comparative hybridization (aCGH) in the genetic evaluation of stillbirth. Am J Med Genet A. 2009;149A:2437–2443.
Array-based comparative genomic hybridization (aCGH) makes it possible to assess the chromosome count and perform a high-resolution search for microduplications and deletions. With known segments of the genome printed on slides, the clinical scientist can analyze DNA from nondividing cells from a stillbirth. The ability to use nondividing cells is important because no cell culture is required. (Cell culture is often difficult to obtain after stillbirth.) Depending on the array selected, the resolution can be as fine as a single nucleotide polymorphism.
aCGH can inform preconception counseling
Raca and colleagues used a range of arrays to assess 15 stillbirths that involved two or more malformations. Chromosomal abnormalities, including trisomy 21 and an unbalanced translocation, were detected by aCGH in two infants. Identification of these abnormalities helped inform counseling of the parents:
- In the case of trisomy 21, parental karyotypes revealed a nontranslocation event, making it possible to assure the parents that the risk of recurrence is low
- The unbalanced translocation resulted from a balanced chromosome translocation in the mother and was associated with a significant risk of recurrence (in this case, FISH would not have helped because chromosomes 13, 18, 21, X, and Y were not involved).
Limitations of aCGH
One limitation is an inability to detect polyploidy such as triploidy or tetraploidy. This problem can be circumvented through the use of a FISH preparation prior to aCGH.
In most centers, parental blood samples are drawn at the time of aCGH studies. Because aCGH offers greater resolution of chromosome regions, an increasing number of benign variations (i.e., present in one parent) are being identified. As aCGH technology advances, we are accumulating data on copy-number variations.
A large clinical trial is needed to assess the full potential of aCGH in this setting.
Use of array-basic comparative genomic hybridization to assess cells from a stillborn fetus can help determine the cause of death and inform counseling of the parents about the risk of recurrence.
Risk factors for stillbirth include
advanced maternal age, obesity, and black race
ACOG Practice Bulletin #102: Management of stillbirth. Obstet Gynecol. 2009;113:748–760.
Willinger M, Ko CW, Reddy UM. Racial disparities in stillbirth risk across gestation in the United States. Am J Obstet Gynecol. 2009;201:469.e1–469.e8.
Fretts RC. The study of stillbirth. Am J Obstet Gynecol. 2009;201:429–430.
Women who have diseases such as insulin-dependent diabetes and systemic lupus erythematosus have long been recognized as having a six- to 20-fold increase in the risk of stillbirth, compared with the general population. However, each of these disorders accounts for 2% and less than 1% of the pregnant population, respectively, so their overall contribution to stillbirth is small. Larger portions of the population have a lower—but still significant—risk of stillbirth:
- women older than 35 years
- women who have a body mass index (BMI) above 30
- non-Hispanic black women.
Each of these categories represents 15% or more of the typical obstetric population, and each group faces a risk of stillbirth approaching 1%. The ACOG practice bulletin and the study by Willinger and colleagues address these risks in detail.
Advanced maternal age is particularly risky among nulliparous women
Advanced maternal age (>35 years) is associated with increased rates of chromosomal abnormality and maternal morbidity, such as hypertension, that are known to raise the risk of stillbirth. Even when these and other variables associated with advanced maternal age, such as placenta previa, diabetes, and multiple gestation, are controlled, however, the increased risk of stillbirth remains.
Advanced maternal age in a first pregnancy carries a particularly elevated risk. For example, the risk of stillbirth in a 40-year-old nulliparous woman is more than twice the risk in a 40-year-old multiparous woman (1 in every 116 pregnancies vs 1 in every 304).3
The increased risk of stillbirth associated with advanced maternal age is present at all gestational ages, though it becomes most profound at 37 to 42 weeks’ gestation, notably for:
- women 35 to 39 years old (1 in every 382 pregnancies; relative risk [RR] of 1.32, compared with women <35 years old; 95% confidence interval [CI], 1.22, 1.43)
- women >40 years old (1 in every 267 pregnancies; RR, 1.88; 95% CI, 1.64, 2.16).
These numbers remain significant even after controlling for medical conditions.3
The utility of antepartum surveillance and induction of labor for delivery is unclear, given the risk of iatrogenic prematurity.
Risk of stillbirth is doubled among obese and markedly obese women
Although the number of adults who are overweight (BMI 25–30) has remained fairly constant over the past 20 years (30% to 35% of the population), the percentage of women of reproductive age who are obese (BMI >30) has risen markedly. Obesity is now present in 35% of the population, and marked obesity (BMI >40) affects an additional 6%. Both obese and markedly obese women face a twofold relative risk of stillbirth, compared with women of normal weight. The rate of stillbirth in this population is 12 to 18 for every 1,000 births—a 1.2% to 1.8% risk.
Although obesity-related stillbirth likely has multiple causes, the risk remains elevated even after exclusion of confounding factors such as smoking, gestational diabetes, and preeclampsia.
Race is an independent contributor
Racial differences in the rate of stillbirth remain despite a decrease in the overall stillbirth rate over the past 20 years ( FIGURE ). In 2003, the rate of stillbirth was 5 for every 1,000 births among non-Hispanic whites, 5.5 among Hispanics, and 12 among non-Hispanic blacks. In other words, the risk of stillbirth was 1 in 202, 1 in 183, and 1 in 87 births for white, Hispanic, and black women, respectively.
Willinger and colleagues utilized data from the National Center for Health Statistics and assessed 2001–2002 birth and infant death datasets for 36 states, examining the stillbirth hazard risk for more than 5 million singleton pregnancies. Stillbirth peaked at 20 to 23 weeks and 39 to 41 weeks’ gestation, as expected. However, at 20 to 23 weeks, the risk of stillbirth among non-Hispanic black women was more than twice the rate for non-Hispanic white women (RR, 2.8). Although it then declined as term approached, it remained greater than that of non-Hispanic white women (RR, 1.6).

FIGURE Racial disparities in the risk of stillbirth
Hazard of stillbirth for singleton pregnancies by gestational age and race and ethnicity, 2001–2002. SOURCE: Willinger et al. Greater acceptance and use of induction of labor at term among whites merits attention
In an editorial accompanying the study by Willinger and colleagues, Fretts pointed out the higher rate of induction of labor at term among white women that has been observed in at least three studies of vital statistics. (Willinger and colleagues also pointed out this difference.) The acceptance and use of labor induction at term—and the lower stillbirth rate—among white women warrants further investigation.
Education appears to reduce the risk of stillbirth to a greater degree among whites than it does among blacks. Again, nulliparity and advanced maternal age were important contributors to the risk of stillbirth across all three races.
Counsel African-American gravidas and women older than 35 years that their risk of stillbirth is elevated.
Obese women should be advised to lose weight before conception if at all possible to reduce the risk of stillbirth.
Needed: Standardized analysis
and documentation of stillbirth
Reddy UM, Goldberg R, Silver R, et al. Stillbirth classification—developing an international consensus for research: executive summary of a National Institute of Child Health and Human Development workshop. Obstet Gynecol. 2009;114:901–914.
Flenady V, Frøen JF, Pinar H, et al. An evaluation of classification systems for stillbirth. BMC Pregnancy Childbirth. 2009;9:24.
Further guidance for the clinical management of stillbirth will come from investigations of the underlying pathologies and associated risk factors. Key to development of this guidance is the involvement of obstetricians in documenting the antenatal record and delivery information. Also needed is a standardized system for recording this information. More than three dozen systems have been developed to classify stillbirth, at the expense of uniformity of content.
An international consensus group published guidelines on how to describe the cause of death in research endeavors, recognizing the need to maintain the ability to attach a level of uncertainty. In addition, Flenady and colleagues compared the most widely used systems in clinical practice, assigning the highest score for components such as ease of use, inter observer variability, and proportion of unexplained stillbirths to CODAC [cause of death and two associated causes]. This system assigns a primary cause of death from a specified list of choices and allows inclusion of two possible contributing causes.
Both the international consensus classification and the CODAC scoring system are accessible through links embedded within the articles. Both systems require the establishment of standardized evaluation and review of stillbirth that should include obstetricians, pathologists, and geneticists.
Because assessment and classification of stillbirth are fundamental to its prevention, as well as a critical part of clinical practice, ObGyns should become familiar with the international consensus classification and CODAC scoring systems and adopt a standardized approach to assessment and documentation.
1. Rodgers CS, Creasy MR, Fitchett M, Maliszewska CT, Pratt NR, Waters JJ. Solid tissue culture for cytogenetic analysis: a collaborative survey for the Association of Clinical Cytogeneticists. J Clin Pathol. 1996;49:638-641.
2. Rivasi F, Schirosi L, Bettelli S, et al. FISH analysis in cell touch preparations and cytological specimens from formalin-fixed fetal autopsies. Diagn Cytopathol. 2008;36:633-636.
3. Reddy UM, Ko CW, Willinger M. Maternal age and the risk of stillbirth throughout pregnancy in the United States. Am J Obstet Gynecol. 2006;195:764-770.
4. MacDorman MF, Mathews TJ. NCHS Data Brief #9: Recent trends in infant mortality in the United States. Atlanta, Ga: National Center for Health Statistics; October 2008. Available at: http://www.cdc.gov/nchs/data/databriefs/db09.htm. Accessed Dec. 15, 2009.
1. Rodgers CS, Creasy MR, Fitchett M, Maliszewska CT, Pratt NR, Waters JJ. Solid tissue culture for cytogenetic analysis: a collaborative survey for the Association of Clinical Cytogeneticists. J Clin Pathol. 1996;49:638-641.
2. Rivasi F, Schirosi L, Bettelli S, et al. FISH analysis in cell touch preparations and cytological specimens from formalin-fixed fetal autopsies. Diagn Cytopathol. 2008;36:633-636.
3. Reddy UM, Ko CW, Willinger M. Maternal age and the risk of stillbirth throughout pregnancy in the United States. Am J Obstet Gynecol. 2006;195:764-770.
4. MacDorman MF, Mathews TJ. NCHS Data Brief #9: Recent trends in infant mortality in the United States. Atlanta, Ga: National Center for Health Statistics; October 2008. Available at: http://www.cdc.gov/nchs/data/databriefs/db09.htm. Accessed Dec. 15, 2009.
A guide for clinicians: Bariatric surgery and the ObGyn patient
CASE: New-onset abdominal pain and a history of bariatric surgery
A 28-year-old woman who is 14 weeks pregnant (G5P1A4) complains of abdominal pain, nausea, and vomiting for the past 3 days. Four years ago, she underwent a successful Roux-en-Y gastric bypass, and her body mass index (BMI) decreased from 61.1 to 34.8 as a result. Today, after unremarkable ultrasonography at an outside hospital, she is transferred to your facility, where she is noted to be afebrile and tachycardic (133 bpm) with a very tender abdomen and a white blood cell (WBC) count of 12.2×103/μL.
Could her symptoms be related to her history of gastric bypass? How should you manage her condition?
This case illustrates why it is imperative that Ob Gyns have a basic understanding of bariatric surgery—particularly the types of procedures being performed today and the complications that may arise.
In this case, urgent intervention is called for, given the elevated WBC count and extreme tenderness of the abdomen. This patient was taken to surgery without waiting for additional imaging studies. Operative findings included intussusception with necrosis, a late complication of the Roux-en-Y procedure, necessitating resection of 60 cm of small intestine ( FIGURE 1 ). Although fetal heart sounds remained encouraging overnight, fetal demise occurred the next day. The patient’s physical recovery was otherwise unremarkable.
This scenario is not as unlikely as you might imagine. A similar episode was described in 2004.1 The outcome: maternal and fetal death.
FIGURE 1 Life-threatening intestinal obstruction
Patchy and continuous necrosis is evident following reduction of an obstruction-causing intussusception.
A boom in bariatric surgery
The incidence of surgical weight-loss procedures increased sixfold over a recent 7-year time span.2 In 2008 alone, more than 200,000 people underwent this type of operation—at least half of them women of reproductive age.2,3 In my experience at the University Hospitals of Cleveland Bariatric Program, which I directed, 85% of patients were women.4 This percentage is similar to the gender breakdown in other bariatric programs.
Because you are called on to provide both primary and specialty care, you are very likely to encounter women who have undergone bariatric surgery or who are considering it. As the prevalence of morbid obesity continues to rise, so will the need for familiarity with surgical weight-loss reduction.
In this article, I offer an overview of bariatric surgery, focusing on:
- the different procedures performed today
- the potential complications associated with the most common operations
- the need for nutritional assessment.
In the process, I discuss the health benefits of bariatric surgery, including greater fertility and improved pregnancy outcomes.
How we talk about obesity
The language of obesity centers on a basic metric: the body mass index (BMI).5 This index was devised in the 19th century by Adolphe Quetelet to describe total body fat.
Consider its use to describe two markedly overweight individuals. One stands 6 ft and weighs 500 lb; the other stands 5 ft and weighs 400 lb. It is obvious which person is heavier; the BMI quantifies who is actually bigger. The first individual has a BMI of 67.8, whereas the shorter person has a BMI of 78, despite weighing 100 lb less.
The formula for determining BMI is:
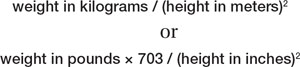
Among the imperfections of the BMI are its failure to differentiate between genders and its marked inaccuracy in persons who have significant muscle mass and limited body fat, such as body builders. However, it does make it possible to compare patients of different height and weight, thereby facilitating attempts to prognosticate the risk of illness and identify candidates for bariatric surgery ( TABLE 1 ).
The 1991 National Institutes of Health (NIH) Consensus Conference on Gastrointestinal Surgery for Severe Obesity recommended that bariatric surgery (restrictive and bypass procedures) be considered for patients who have a BMI of 40 or above and for patients who have a BMI of 35 or above with high-risk comorbidity.6
TABLE 1
How size is classified using the body mass index
| BMI | Classification | Risk of illness and comorbidity |
|---|---|---|
| <18.5 | Underweight | Increased |
| 18.5–24.9 | Normal | Average |
| 25–29.9 | Overweight | Increased |
| 30–34.9 | Obese (class I) | Moderate |
| 35–39.9 | Obese (class II) | Severe |
| ≥40 | Morbid obesity (class III) | Very severe |
| ≥50 | “Severe morbid obesity” | !!! Severe |
Most agree that bariatric surgery originated nearly 60 years ago with the jejunoileal bypass, a purely malabsorptive procedure in which the proximal jejunum was anastomosed to the distal ileum.7-9 This operation was based on the observation of dramatic weight loss following major small-bowel resection. The procedure was thought to allow obese patients to eat with abandon, thanks to the limited absorptive ability of the small intestine’s mucosal surface, which would impede digestion. However, the operation was fraught with complications, including protein calorie malnutrition, bacterial overgrowth, and even death. In addition, despite the dramatic reduction in the absorptive surface of the small intestine, some patients eventually regained weight.
One may still encounter patients who underwent jejunoileal bypass many years ago. One particular complication of this procedure is worth mentioning: hepatic cirrhosis. Any patient who has a history of jejunoileal bypass should have her liver function assessed before undergoing procedures that require general anesthesia.
At present, bariatric surgery may incorporate a component of malabsorption, but no other procedures are purely malabsorptive ( TABLE 2 ).
TABLE 2
Bariatric procedures for weight reduction, past and present
| Mechanism of action | Procedure |
|---|---|
| Malabsorptive | Jejunoileal bypass* |
| Restrictive | Vertical banded gastroplasty Gastric banding (Lap-Band) Laparoscopic sleeve gastrectomy Magenstrasse and Mill operation |
| Combination (malabsorptive and restrictive) | Roux-en-Y gastric bypass Biliopancreatic diversion Duodenal switch |
| Other | Gastric pacing† |
| * No longer performed | |
| †Experimental | |
Restrictive procedures
Vertical banded gastroplasty (VBG). This procedure is one of the earliest successful restrictive procedures ( FIGURE 2 ). Although it was originally performed via laparotomy, it is easily carried out laparoscopically. However, a risk of complication, such as severe reflux, and a low long-term success rate have decreased its widespread application.
Laparoscopic adjustable silicone band. The most common restrictive procedure performed today is the laparoscopic adjustable silicone band (Lap-Band) ( FIGURE 2 ). Among its benefits are:
- easy insertion, especially in comparison with the gastric bypass
- ability to adjust the amount of restriction (not possible with VBG)
- elimination of the need for supplemental nutrition
- reversibility.
Long-term results from a US population are not available because FDA approval did not occur until 2001. However, some centers outside the United States have reported unfavorable long-term outcomes. (See “Lap-Band complications may be pervasive,”.)
Laparoscopic sleeve gastrectomy (LSG). This operation was originally devised as the first stage of a duodenal switch. However, weight loss with this stage alone made LSG a bariatric procedure in its own right ( FIGURE 2 ).
Although this operation is categorized as restrictive, gastric sleeve resection eliminates the gastric fundus, the major site where ghrelin—“the hunger hormone”—is produced. This hormonal alteration may contribute to weight loss that occurs after LSG.
Magenstrasse and Mill operation. This operation converts the stomach into a long tube similar to the gastric sleeve. The greater curvature of the stomach is separated from the newly created tube but remains attached to the rest of the stomach at the antrum. (The greater curvature of the stomach is resected in the gastric sleeve procedure.)
Many insurers refuse to reimburse for restrictive operations other than the VBG and Lap-Band.
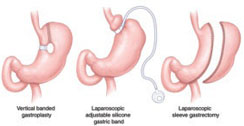
FIGURE 2 Three common surgeries
These restrictive procedures decrease the size of the stomach reservoir without impairing absorption of calories.
Combination procedures
Biliopancreatic diversion, duodenal switch. Despite the risk of protein malnutrition, these two procedures attempt to produce selective malabsorption of fat and carbohydrate as a means of treating the most severely obese patients. These operations carry the highest mortality rate and are therefore less likely to be offered routinely.
Roux-en-Y gastric bypass (RYGB). In the United States, the RYGB is performed more frequently than other procedures that combine malabsorption and restriction. It is considered by most to be the “gold standard” bariatric operation ( FIGURE 3 ).
The RYGB may induce weight loss through one or more of the following mechanisms:
- a decrease in the size of the gastric pouch. After the procedure, this pouch has a capacity of about 30 cc (size of a thumb)
- the small diameter of the gastrojejunal anastomosis, which is usually 1 cm
- glucose intolerance. About 50% of patients experience dumping symptoms when they ingest refined carbohydrates
- subclinical malabsorption. Food does not encounter the majority of digestive enzymes until it reaches the jejunojejunostomy anastomosis
- suppression of ghrelin production. Ghrelin is a hormone produced primarily in the gastric fundus and secondarily in the duodenum, both of which are bypassed with the RYGB. Plasma ghrelin levels peak before meals and are suppressed with food intake. After RYGB, patients demonstrate a flattening of this pattern, which appears to correlate with a suppression of appetite.10
FIGURE 3 Roux-en-Y gastric bypass
A gastric pouch is created at the top of the stomach and connected directly to the jejunum, bypassing the rest of the stomach and duodenum.
When is bariatric surgery indicated?
The NIH Consensus Conference identified patients who might be candidates for bariatric surgery, as I noted:
- patients who have a BMI of 40 or above, whether or not they have comorbidity
- patients who have a BMI of 35 or above and comorbidity.6
These recommendations have been adopted by third-party payers.
Potential comorbidities include hypertension, coronary artery disease (CAD), lipid abnormality, diabetes, obstructive sleep apnea (OSA), and severe osteoarthritis, among others.
When identified together, several of these comorbidities constitute metabolic syndrome, a risk factor for CAD. More than 50 million Americans are thought to exhibit this syndrome, which is diagnosed when three or more of the following are present:
- waist circumference ≥40 in (102 cm) in men; ≥35 in (88 cm) in women
- triglycerides ≥150 mg/dL
- high-density lipoprotein cholesterol <40 mg/dL in men; <50 mg/dL in women
- blood pressure ≥130/85 mm Hg
- fasting glucose ≥100 mg/dL.
Postsurgery weight loss has many benefits
Obstructive sleep apnea may resolve
Although this condition is known to be associated with obesity, it is drastically underdiagnosed.11 In our program, only 21% of patients had been given a diagnosis of OSA at the time of initial evaluation for surgery. After we obtained a polysomnogram (sleep study) for each of them, however, the true prevalence was determined to be 91%.
OSA can have serious consequences if it is untreated, and it may increase the risks associated with surgery and general anesthesia. By identifying and treating OSA before gastric bypass (RYGB), we were able to eliminate the respiratory ICU stay—a benefit that should be applicable to nonbariatric surgeries in the morbidly obese patient as well.12
With successful bariatric surgery, OSA abnormalities identified during polysomnography are dramatically improved or eliminated. Treatment with continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BIPAP) can usually be discontinued 6 months to 1 year after the dramatic weight loss associated with RYGB.
Cancer death rate may be reduced
Three consequences of morbid obesity not generally thought to be indications for bariatric surgery are:
- obesity-associated cancer
- decreased longevity
- infertility.
However, each of these conditions may be improved through successful bariatric surgery.
In a prospective study of more than 900,000 adults in the United States who were followed for 16 years, overweight or obesity was thought to account for 14% of cancer deaths in men and 20% of cancer deaths in women.13 Death rates for persons who had a BMI of at least 40 were 52% higher in men and 62% higher in women than they were in people who had a BMI below that threshold. BMI was significantly associated with cancer of the esophagus, colon and rectum, liver, gallbladder, pancreas, and kidney, as well as with non-Hodgkin’s lymphoma and multiple myeloma.
A trend toward an increasing risk of death with higher BMI was observed for cancer of the stomach (men), prostate, breast (women), uterus, ovary, and cervix.
The benefits of bariatric surgery in regard to cancer incidence and longevity were revealed in a 15-year, nonrandomized, prospective study involving more than 4,000 patients.14 At 10.9 years of follow-up, with a follow-up rate of 99.9%, this investigation concluded that bariatric surgery reduces mortality attributable to cardiovascular disease and cancer.14 Most surprising was the finding of a reduced incidence of cancer in patients who underwent bariatric surgery, compared with matched controls.14 An editorial accompanying this study summed it up in the headline: “The missing link—lose weight, live longer.”15
The Framingham risk score estimates the 10-year risk of CAD. This score is reduced by more than 50% after successful gastric bypass surgery.16
After surgery, fertility improves and pregnancy has better outcomes
From your practice, you’re certainly aware of the detrimental effects that obesity has on fertility, pregnancy, and fetal health.17-22 Although neither pregnancy nor impaired fertility is a primary indication for surgical weight reduction in an obese woman, bariatric surgery can improve the likelihood of fertility and successful pregnancy.
Advise patients to delay pregnancy after bariatric surgery. Although none of the bariatric operations performed today are contraindications to pregnancy, we caution all women to delay pregnancy—using two forms of birth control—until weight loss has stabilized. This usually takes about 12 to 18 months after surgery.
In our program, we identified 28 women who became pregnant following gastric bypass. Although we had cautioned all women to avoid early pregnancy, 10 became pregnant within 1 year of the bypass. Among these women, the rate of miscarriage was 40%, compared with 17% among the 18 women who waited more than 1 year to conceive. These percentages merit concern despite the small sample size.
Dao and colleagues concluded that the timing of pregnancy after gastric bypass is irrelevant.23 However, critical review of their data reveals an average weight gain of 4 lb in the group of women who became pregnant within a year after surgery (range, -70 to +45 lb), compared with 34 lb in the group that waited at least 1 year to conceive (range, +13 to +75 lb). As it was among our own patients, miscarriage was more common in the early group (24% vs 8% in the late group). Although these data are not statistically significant, they should arouse concern. I believe the most prudent advice to give women following bariatric surgery is to delay pregnancy until weight loss has stabilized.
The World Health Organization has estimated that there are 1 billion overweight adults on the planet. The problem of obesity, however, is concentrated in the United States. For well over a decade, the problem has escalated, with the US obesity rate referred to as “epidemic” and “a crisis”—among both adults and children.24
Among adults 20 years and older, the prevalence of obesity in the United States is 32%, and the prevalence of morbid obesity is almost 5%. Among adult women 20 to 39 years old, the prevalence of obesity and morbid obesity is 29% and 8%, respectively.27
Complications of bariatric surgery
Complications following bariatric surgery are intrinsic to the specific procedure performed. Because RYGB and the Lap-Band procedure constitute the overwhelming majority of bariatric operations, the most important complications associated with these surgeries will be addressed.
After RYGB, greatest risk is intestinal obstruction
Some of the early postoperative complications following RYGB are unlikely to be seen by physicians other than bariatric surgeons. They include anastomotic leaks, peritonitis, and bleeding. Pulmonary emboli should be managed as usual during the postoperative course. The amount of heparin necessary to achieve therapeutic anticoagulation may be quite substantial, but this fact should not deter the treating physician.
Nausea is almost universally present during the first few days after RYGB. When nausea and increasing food intolerance occur later (after the patient has demonstrated that food can be tolerated), it may indicate stenosis of the gastrojejunal anastomosis. The optimal study to evaluate these symptoms is a barium upper gastrointestinal (GI) swallow study. Stenosis can usually be managed by endoscopic dilatation.24
Intestinal obstruction. One of the most serious problems following RYGB is intestinal obstruction, which may be caused by internal hernia, adhesion, intussusception (not rare in the gastric bypass patient), and other less common causes ( FIGURE 1 ). Intestinal obstruction may occur early or years after the procedure.
In the RYGB patient, obstruction manifests with prominent pain, but distension and vomiting are usually absent, and plain abdominal radiographs are generally normal. The reason for this unusual presentation? The biliopancreatic limb tends to be the segment involved in the blockage, thereby creating a closed-loop obstruction. The most efficient way to diagnose this potentially life-threatening problem is by computed tomography (CT) ( FIGURE 4 ).
FIGURE 4 CT facilitates diagnosis of closed-loop obstruction
These computed tomography images reveal obstruction of the remnant stomach, duodenum, and proximal jejunum (the biliopancreatic limb) in a patient who has undergone Roux-en-Y gastric bypass.
Lap-Band complications may be pervasive
The Lap-Band procedure is often touted as having very low morbidity and mortality. However, data from outside the United States, where longer follow-up has been conducted, have cast a shadow of doubt on the supposed benefits of this restrictive procedure.
One study involving nearly 900 patients and 12 years of follow-up identified a rate of late complication of nearly 28% (2.3%, band erosion; 15.5%, pouch dilatation; 8.9%, port problems) and a rate of major reoperation of nearly 30%. The failure rate increased over time, reaching 65% at 10 years.
Despite these concerns, there is widespread enthusiasm among patients and bariatric surgeons who are proponents of this procedure.
A life-threatening complication. Any physician who might care for a patient who has undergone a Lap-Band procedure should be aware of one potentially life-threatening consequence: a slipped band. This complication can lead to incarceration of the stomach—usually the fundus—which, if left untreated, causes gastric infarction.
The main symptom of band slippage is pain. Any patient who has a Lap-Band and who reports significant upper abdominal pain must be evaluated to exclude slippage. Evaluation usually consists of a swallow study. Surgical correction is mandatory and urgent.
Like complications of surgery, the nutritional demands following bariatric surgery depend on the type of procedure that is performed.
After Lap-Band and other purely restrictive procedures,
nutritional levels remain stable
With restrictive procedures, such as the Lap-Band operation, the GI tract is essentially unaffected. Consequently, vitamin and micronutrient absorption remains intact. In these patients, we usually recommend a daily multivitamin. We assess serum levels to determine whether calcium and vitamin D supplementation might be necessary, which is especially important in female patients.
Close monitoring is needed after RYGB
After RYGB and other combined procedures, the question of nutritional supplementation becomes more complex.25 Bypass of the duodenum and upper jejunum affect absorption of iron, vitamin B12, calcium, and vitamin D. Serum calcium assessment is inadequate to determine the adequacy of calcium and vitamin D supplementation.
We evaluate parathyroid hormone and alkaline phosphatase levels in these patients. Any increase in these levels in the presence of normal serum calcium suggests the induction of secondary hyperparathyroidism and the need for additional calcium and vitamin D.26
Our recommendations for all patients following RYGB are a daily multivitamin, vitamin B12, and supplemental calcium and vitamin D. Vitamin B1 (thiamine) deficiency is extremely rare unless prolonged vomiting has been present. If B1 deficiency is suspected, administer thiamine before giving glucose because glucose can make symptoms worse.
Most women also need supplemental iron, which can be provided alone or in combination with the multivitamin, as in prenatal vitamins.
After RYGB, we measure vitamin and iron levels at 2 weeks, 3 months, 6 months, 12 months, 18 months, and then yearly thereafter unless a major abnormality is identified.
1. Moore KA, Ouyang DW, Whang EE. Maternal and fetal deaths after gastric bypass surgery for morbid obesity. N Engl J Med. 2004;351:721-722.
2. Bariatric Surgery in Women of Reproductive Age: Special Concerns for Pregnancy. Structured Abstract. Rockville, Md: Agency for Healthcare Research and Quality; November 2008. Available at: www.ahrq.gov/Clinic/tp/barireptp.htm. Accessed Dec. 8, 2009.
3. American Society for Metabolic and Bariatric Surgery. Metabolic and bariatric surgery fact sheet. Available at: www.asmbs.org/Newsite07/media/asmbs_fs_surgery.pdf. Accessed Dec. 8, 2009.
4. Stellato TA, Hallowell PT, Crouse C, Schuster M, Petrozzi MC. Two-day length of stay following open Roux-en-Y gastric bypass: is it feasible, safe and reasonable? Obes Surg. 2004;14:27-34.
5. Oria HE, Carrasquilla C, Cunningham P, et al. American Society for Bariatric Surgery Standards Committee, 2004–2005. Guidelines for weight calculations and follow-up in bariatric surgery. Surg Obes Relat Dis. 2005;1:67-68.
6. Gastrointestinal surgery for severe obesity: National Institutes of Health Consensus Development Conference Statement. Am J Clin Nutr. 1992;55(Suppl 2):615S-619S.
7. MacDonald KG, Jr. Overview of the epidemiology of obesity and the early history of procedures to remedy morbid obesity. Arch Surg. 2003;138:357-360.
8. Buchwald H. Overview of bariatric surgery. J Am Coll Surg. 2002;194:367-375.
9. DeMaria EJ. Bariatric surgery for morbid obesity. N Engl J Med. 2007;356:2176-2183.
10. Cummings DE, Weigle DS, Frayo RS, et al. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med. 2002;346:1623-1630.
11. Hallowell PT, Stellato TA, Schuster M, et al. Potentially life-threatening sleep apnea is unrecognized without aggressive evaluation. Am J Surg. 2007;193:364-367.
12. Hallowell PT, Stellato TA, Petrozzi MC, et al. Eliminating respiratory intensive care unit stay after gastric bypass surgery. Surgery. 2007;142:608-612.
13. Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003;348:1625-1638.
14. Sjöström L, Narbro K, Sjöström CD, et al. Swedish Obese Subjects Study. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741-752.
15. Bray GA. The missing link—lose weight, live longer. N Engl J Med. 2007;357:818-820.
16. Kligman MD, Dexter DJ, Omer S, Park AE. Shrinking cardiovascular risk through bariatric surgery: application of Framingham risk score in gastric bypass. Surgery. 2008;143:533-538.
17. Linné Y. Effects of obesity on women’s reproduction and complications during pregnancy. Obes Rev. 2004;5:137-143.
18. Pasquali R, Pelusi C, Genghini S, Cacciari M, Gambineri A. Obesity and reproductive disorders in women. Hum Reprod Update. 2003;9:359-372.
19. Baeten JM, Bukusi EA, Lambe M. Pregnancy complications and outcomes among overweight and obese nulliparous women. Am J Public Health. 2001;91:436-440.
20. Lashen H, Fear K, Sturdee DW. Obesity is associated with increased risk of first trimester and recurrent miscarriage: matched case-control study. Hum Reprod. 2004;19:1644-1646.
21. Cnattingius S, Bergström R, Lipworth L, Kramer MS. Prepregnancy weight and the risk of adverse pregnancy outcomes. N Engl J Med. 1998;338:147-152.
22. Pathi A, Esen U, Hildreth A. A comparison of complications of pregnancy and delivery in morbidly obese and non-obese women. J Obstet Gynaecol. 2006;26:527-530.
23. Dao T, Kuhn J, Ehmer D, Fisher T, McCarty T. Pregnancy outcomes after gastric-bypass surgery. Am J Surg. 2006;192:762-766.
24. Stellato TA, Crouse C, Hallowell PT. Bariatric surgery: creating new challenges for the endoscopist. Gastrointest Endosc. 2003;57:86-94.
25. Poitou Bernert C, Ciangura C, Coupaye M, Czernichow S, Bouillot JL, Basdevant A. Nutritional deficiency after gastric bypass: diagnosis, prevention and treatment. Diabetes Metab. 2007;33:13-24.
26. Jin J, Robinson AV, Hallowell PT, Jasper JJ, Stellato TA, Wilhelm SM. Increases in parathyroid hormone (PTH) after gastric bypass surgery appear to be of a secondary nature. Surgery. 2007;142:914-920.
27. Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA. 2006;295:1549-1555.
CASE: New-onset abdominal pain and a history of bariatric surgery
A 28-year-old woman who is 14 weeks pregnant (G5P1A4) complains of abdominal pain, nausea, and vomiting for the past 3 days. Four years ago, she underwent a successful Roux-en-Y gastric bypass, and her body mass index (BMI) decreased from 61.1 to 34.8 as a result. Today, after unremarkable ultrasonography at an outside hospital, she is transferred to your facility, where she is noted to be afebrile and tachycardic (133 bpm) with a very tender abdomen and a white blood cell (WBC) count of 12.2×103/μL.
Could her symptoms be related to her history of gastric bypass? How should you manage her condition?
This case illustrates why it is imperative that Ob Gyns have a basic understanding of bariatric surgery—particularly the types of procedures being performed today and the complications that may arise.
In this case, urgent intervention is called for, given the elevated WBC count and extreme tenderness of the abdomen. This patient was taken to surgery without waiting for additional imaging studies. Operative findings included intussusception with necrosis, a late complication of the Roux-en-Y procedure, necessitating resection of 60 cm of small intestine ( FIGURE 1 ). Although fetal heart sounds remained encouraging overnight, fetal demise occurred the next day. The patient’s physical recovery was otherwise unremarkable.
This scenario is not as unlikely as you might imagine. A similar episode was described in 2004.1 The outcome: maternal and fetal death.

FIGURE 1 Life-threatening intestinal obstruction
Patchy and continuous necrosis is evident following reduction of an obstruction-causing intussusception.
A boom in bariatric surgery
The incidence of surgical weight-loss procedures increased sixfold over a recent 7-year time span.2 In 2008 alone, more than 200,000 people underwent this type of operation—at least half of them women of reproductive age.2,3 In my experience at the University Hospitals of Cleveland Bariatric Program, which I directed, 85% of patients were women.4 This percentage is similar to the gender breakdown in other bariatric programs.
Because you are called on to provide both primary and specialty care, you are very likely to encounter women who have undergone bariatric surgery or who are considering it. As the prevalence of morbid obesity continues to rise, so will the need for familiarity with surgical weight-loss reduction.
In this article, I offer an overview of bariatric surgery, focusing on:
- the different procedures performed today
- the potential complications associated with the most common operations
- the need for nutritional assessment.
In the process, I discuss the health benefits of bariatric surgery, including greater fertility and improved pregnancy outcomes.
How we talk about obesity
The language of obesity centers on a basic metric: the body mass index (BMI).5 This index was devised in the 19th century by Adolphe Quetelet to describe total body fat.
Consider its use to describe two markedly overweight individuals. One stands 6 ft and weighs 500 lb; the other stands 5 ft and weighs 400 lb. It is obvious which person is heavier; the BMI quantifies who is actually bigger. The first individual has a BMI of 67.8, whereas the shorter person has a BMI of 78, despite weighing 100 lb less.
The formula for determining BMI is:
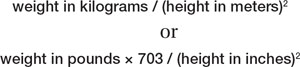
Among the imperfections of the BMI are its failure to differentiate between genders and its marked inaccuracy in persons who have significant muscle mass and limited body fat, such as body builders. However, it does make it possible to compare patients of different height and weight, thereby facilitating attempts to prognosticate the risk of illness and identify candidates for bariatric surgery ( TABLE 1 ).
The 1991 National Institutes of Health (NIH) Consensus Conference on Gastrointestinal Surgery for Severe Obesity recommended that bariatric surgery (restrictive and bypass procedures) be considered for patients who have a BMI of 40 or above and for patients who have a BMI of 35 or above with high-risk comorbidity.6
TABLE 1
How size is classified using the body mass index
| BMI | Classification | Risk of illness and comorbidity |
|---|---|---|
| <18.5 | Underweight | Increased |
| 18.5–24.9 | Normal | Average |
| 25–29.9 | Overweight | Increased |
| 30–34.9 | Obese (class I) | Moderate |
| 35–39.9 | Obese (class II) | Severe |
| ≥40 | Morbid obesity (class III) | Very severe |
| ≥50 | “Severe morbid obesity” | !!! Severe |
Most agree that bariatric surgery originated nearly 60 years ago with the jejunoileal bypass, a purely malabsorptive procedure in which the proximal jejunum was anastomosed to the distal ileum.7-9 This operation was based on the observation of dramatic weight loss following major small-bowel resection. The procedure was thought to allow obese patients to eat with abandon, thanks to the limited absorptive ability of the small intestine’s mucosal surface, which would impede digestion. However, the operation was fraught with complications, including protein calorie malnutrition, bacterial overgrowth, and even death. In addition, despite the dramatic reduction in the absorptive surface of the small intestine, some patients eventually regained weight.
One may still encounter patients who underwent jejunoileal bypass many years ago. One particular complication of this procedure is worth mentioning: hepatic cirrhosis. Any patient who has a history of jejunoileal bypass should have her liver function assessed before undergoing procedures that require general anesthesia.
At present, bariatric surgery may incorporate a component of malabsorption, but no other procedures are purely malabsorptive ( TABLE 2 ).
TABLE 2
Bariatric procedures for weight reduction, past and present
| Mechanism of action | Procedure |
|---|---|
| Malabsorptive | Jejunoileal bypass* |
| Restrictive | Vertical banded gastroplasty Gastric banding (Lap-Band) Laparoscopic sleeve gastrectomy Magenstrasse and Mill operation |
| Combination (malabsorptive and restrictive) | Roux-en-Y gastric bypass Biliopancreatic diversion Duodenal switch |
| Other | Gastric pacing† |
| * No longer performed | |
| †Experimental | |
Restrictive procedures
Vertical banded gastroplasty (VBG). This procedure is one of the earliest successful restrictive procedures ( FIGURE 2 ). Although it was originally performed via laparotomy, it is easily carried out laparoscopically. However, a risk of complication, such as severe reflux, and a low long-term success rate have decreased its widespread application.
Laparoscopic adjustable silicone band. The most common restrictive procedure performed today is the laparoscopic adjustable silicone band (Lap-Band) ( FIGURE 2 ). Among its benefits are:
- easy insertion, especially in comparison with the gastric bypass
- ability to adjust the amount of restriction (not possible with VBG)
- elimination of the need for supplemental nutrition
- reversibility.
Long-term results from a US population are not available because FDA approval did not occur until 2001. However, some centers outside the United States have reported unfavorable long-term outcomes. (See “Lap-Band complications may be pervasive,”.)
Laparoscopic sleeve gastrectomy (LSG). This operation was originally devised as the first stage of a duodenal switch. However, weight loss with this stage alone made LSG a bariatric procedure in its own right ( FIGURE 2 ).
Although this operation is categorized as restrictive, gastric sleeve resection eliminates the gastric fundus, the major site where ghrelin—“the hunger hormone”—is produced. This hormonal alteration may contribute to weight loss that occurs after LSG.
Magenstrasse and Mill operation. This operation converts the stomach into a long tube similar to the gastric sleeve. The greater curvature of the stomach is separated from the newly created tube but remains attached to the rest of the stomach at the antrum. (The greater curvature of the stomach is resected in the gastric sleeve procedure.)
Many insurers refuse to reimburse for restrictive operations other than the VBG and Lap-Band.

FIGURE 2 Three common surgeries
These restrictive procedures decrease the size of the stomach reservoir without impairing absorption of calories.
Combination procedures
Biliopancreatic diversion, duodenal switch. Despite the risk of protein malnutrition, these two procedures attempt to produce selective malabsorption of fat and carbohydrate as a means of treating the most severely obese patients. These operations carry the highest mortality rate and are therefore less likely to be offered routinely.
Roux-en-Y gastric bypass (RYGB). In the United States, the RYGB is performed more frequently than other procedures that combine malabsorption and restriction. It is considered by most to be the “gold standard” bariatric operation ( FIGURE 3 ).
The RYGB may induce weight loss through one or more of the following mechanisms:
- a decrease in the size of the gastric pouch. After the procedure, this pouch has a capacity of about 30 cc (size of a thumb)
- the small diameter of the gastrojejunal anastomosis, which is usually 1 cm
- glucose intolerance. About 50% of patients experience dumping symptoms when they ingest refined carbohydrates
- subclinical malabsorption. Food does not encounter the majority of digestive enzymes until it reaches the jejunojejunostomy anastomosis
- suppression of ghrelin production. Ghrelin is a hormone produced primarily in the gastric fundus and secondarily in the duodenum, both of which are bypassed with the RYGB. Plasma ghrelin levels peak before meals and are suppressed with food intake. After RYGB, patients demonstrate a flattening of this pattern, which appears to correlate with a suppression of appetite.10

FIGURE 3 Roux-en-Y gastric bypass
A gastric pouch is created at the top of the stomach and connected directly to the jejunum, bypassing the rest of the stomach and duodenum.
When is bariatric surgery indicated?
The NIH Consensus Conference identified patients who might be candidates for bariatric surgery, as I noted:
- patients who have a BMI of 40 or above, whether or not they have comorbidity
- patients who have a BMI of 35 or above and comorbidity.6
These recommendations have been adopted by third-party payers.
Potential comorbidities include hypertension, coronary artery disease (CAD), lipid abnormality, diabetes, obstructive sleep apnea (OSA), and severe osteoarthritis, among others.
When identified together, several of these comorbidities constitute metabolic syndrome, a risk factor for CAD. More than 50 million Americans are thought to exhibit this syndrome, which is diagnosed when three or more of the following are present:
- waist circumference ≥40 in (102 cm) in men; ≥35 in (88 cm) in women
- triglycerides ≥150 mg/dL
- high-density lipoprotein cholesterol <40 mg/dL in men; <50 mg/dL in women
- blood pressure ≥130/85 mm Hg
- fasting glucose ≥100 mg/dL.
Postsurgery weight loss has many benefits
Obstructive sleep apnea may resolve
Although this condition is known to be associated with obesity, it is drastically underdiagnosed.11 In our program, only 21% of patients had been given a diagnosis of OSA at the time of initial evaluation for surgery. After we obtained a polysomnogram (sleep study) for each of them, however, the true prevalence was determined to be 91%.
OSA can have serious consequences if it is untreated, and it may increase the risks associated with surgery and general anesthesia. By identifying and treating OSA before gastric bypass (RYGB), we were able to eliminate the respiratory ICU stay—a benefit that should be applicable to nonbariatric surgeries in the morbidly obese patient as well.12
With successful bariatric surgery, OSA abnormalities identified during polysomnography are dramatically improved or eliminated. Treatment with continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BIPAP) can usually be discontinued 6 months to 1 year after the dramatic weight loss associated with RYGB.
Cancer death rate may be reduced
Three consequences of morbid obesity not generally thought to be indications for bariatric surgery are:
- obesity-associated cancer
- decreased longevity
- infertility.
However, each of these conditions may be improved through successful bariatric surgery.
In a prospective study of more than 900,000 adults in the United States who were followed for 16 years, overweight or obesity was thought to account for 14% of cancer deaths in men and 20% of cancer deaths in women.13 Death rates for persons who had a BMI of at least 40 were 52% higher in men and 62% higher in women than they were in people who had a BMI below that threshold. BMI was significantly associated with cancer of the esophagus, colon and rectum, liver, gallbladder, pancreas, and kidney, as well as with non-Hodgkin’s lymphoma and multiple myeloma.
A trend toward an increasing risk of death with higher BMI was observed for cancer of the stomach (men), prostate, breast (women), uterus, ovary, and cervix.
The benefits of bariatric surgery in regard to cancer incidence and longevity were revealed in a 15-year, nonrandomized, prospective study involving more than 4,000 patients.14 At 10.9 years of follow-up, with a follow-up rate of 99.9%, this investigation concluded that bariatric surgery reduces mortality attributable to cardiovascular disease and cancer.14 Most surprising was the finding of a reduced incidence of cancer in patients who underwent bariatric surgery, compared with matched controls.14 An editorial accompanying this study summed it up in the headline: “The missing link—lose weight, live longer.”15
The Framingham risk score estimates the 10-year risk of CAD. This score is reduced by more than 50% after successful gastric bypass surgery.16
After surgery, fertility improves and pregnancy has better outcomes
From your practice, you’re certainly aware of the detrimental effects that obesity has on fertility, pregnancy, and fetal health.17-22 Although neither pregnancy nor impaired fertility is a primary indication for surgical weight reduction in an obese woman, bariatric surgery can improve the likelihood of fertility and successful pregnancy.
Advise patients to delay pregnancy after bariatric surgery. Although none of the bariatric operations performed today are contraindications to pregnancy, we caution all women to delay pregnancy—using two forms of birth control—until weight loss has stabilized. This usually takes about 12 to 18 months after surgery.
In our program, we identified 28 women who became pregnant following gastric bypass. Although we had cautioned all women to avoid early pregnancy, 10 became pregnant within 1 year of the bypass. Among these women, the rate of miscarriage was 40%, compared with 17% among the 18 women who waited more than 1 year to conceive. These percentages merit concern despite the small sample size.
Dao and colleagues concluded that the timing of pregnancy after gastric bypass is irrelevant.23 However, critical review of their data reveals an average weight gain of 4 lb in the group of women who became pregnant within a year after surgery (range, -70 to +45 lb), compared with 34 lb in the group that waited at least 1 year to conceive (range, +13 to +75 lb). As it was among our own patients, miscarriage was more common in the early group (24% vs 8% in the late group). Although these data are not statistically significant, they should arouse concern. I believe the most prudent advice to give women following bariatric surgery is to delay pregnancy until weight loss has stabilized.
The World Health Organization has estimated that there are 1 billion overweight adults on the planet. The problem of obesity, however, is concentrated in the United States. For well over a decade, the problem has escalated, with the US obesity rate referred to as “epidemic” and “a crisis”—among both adults and children.24
Among adults 20 years and older, the prevalence of obesity in the United States is 32%, and the prevalence of morbid obesity is almost 5%. Among adult women 20 to 39 years old, the prevalence of obesity and morbid obesity is 29% and 8%, respectively.27
Complications of bariatric surgery
Complications following bariatric surgery are intrinsic to the specific procedure performed. Because RYGB and the Lap-Band procedure constitute the overwhelming majority of bariatric operations, the most important complications associated with these surgeries will be addressed.
After RYGB, greatest risk is intestinal obstruction
Some of the early postoperative complications following RYGB are unlikely to be seen by physicians other than bariatric surgeons. They include anastomotic leaks, peritonitis, and bleeding. Pulmonary emboli should be managed as usual during the postoperative course. The amount of heparin necessary to achieve therapeutic anticoagulation may be quite substantial, but this fact should not deter the treating physician.
Nausea is almost universally present during the first few days after RYGB. When nausea and increasing food intolerance occur later (after the patient has demonstrated that food can be tolerated), it may indicate stenosis of the gastrojejunal anastomosis. The optimal study to evaluate these symptoms is a barium upper gastrointestinal (GI) swallow study. Stenosis can usually be managed by endoscopic dilatation.24
Intestinal obstruction. One of the most serious problems following RYGB is intestinal obstruction, which may be caused by internal hernia, adhesion, intussusception (not rare in the gastric bypass patient), and other less common causes ( FIGURE 1 ). Intestinal obstruction may occur early or years after the procedure.
In the RYGB patient, obstruction manifests with prominent pain, but distension and vomiting are usually absent, and plain abdominal radiographs are generally normal. The reason for this unusual presentation? The biliopancreatic limb tends to be the segment involved in the blockage, thereby creating a closed-loop obstruction. The most efficient way to diagnose this potentially life-threatening problem is by computed tomography (CT) ( FIGURE 4 ).

FIGURE 4 CT facilitates diagnosis of closed-loop obstruction
These computed tomography images reveal obstruction of the remnant stomach, duodenum, and proximal jejunum (the biliopancreatic limb) in a patient who has undergone Roux-en-Y gastric bypass.
Lap-Band complications may be pervasive
The Lap-Band procedure is often touted as having very low morbidity and mortality. However, data from outside the United States, where longer follow-up has been conducted, have cast a shadow of doubt on the supposed benefits of this restrictive procedure.
One study involving nearly 900 patients and 12 years of follow-up identified a rate of late complication of nearly 28% (2.3%, band erosion; 15.5%, pouch dilatation; 8.9%, port problems) and a rate of major reoperation of nearly 30%. The failure rate increased over time, reaching 65% at 10 years.
Despite these concerns, there is widespread enthusiasm among patients and bariatric surgeons who are proponents of this procedure.
A life-threatening complication. Any physician who might care for a patient who has undergone a Lap-Band procedure should be aware of one potentially life-threatening consequence: a slipped band. This complication can lead to incarceration of the stomach—usually the fundus—which, if left untreated, causes gastric infarction.
The main symptom of band slippage is pain. Any patient who has a Lap-Band and who reports significant upper abdominal pain must be evaluated to exclude slippage. Evaluation usually consists of a swallow study. Surgical correction is mandatory and urgent.
Like complications of surgery, the nutritional demands following bariatric surgery depend on the type of procedure that is performed.
After Lap-Band and other purely restrictive procedures,
nutritional levels remain stable
With restrictive procedures, such as the Lap-Band operation, the GI tract is essentially unaffected. Consequently, vitamin and micronutrient absorption remains intact. In these patients, we usually recommend a daily multivitamin. We assess serum levels to determine whether calcium and vitamin D supplementation might be necessary, which is especially important in female patients.
Close monitoring is needed after RYGB
After RYGB and other combined procedures, the question of nutritional supplementation becomes more complex.25 Bypass of the duodenum and upper jejunum affect absorption of iron, vitamin B12, calcium, and vitamin D. Serum calcium assessment is inadequate to determine the adequacy of calcium and vitamin D supplementation.
We evaluate parathyroid hormone and alkaline phosphatase levels in these patients. Any increase in these levels in the presence of normal serum calcium suggests the induction of secondary hyperparathyroidism and the need for additional calcium and vitamin D.26
Our recommendations for all patients following RYGB are a daily multivitamin, vitamin B12, and supplemental calcium and vitamin D. Vitamin B1 (thiamine) deficiency is extremely rare unless prolonged vomiting has been present. If B1 deficiency is suspected, administer thiamine before giving glucose because glucose can make symptoms worse.
Most women also need supplemental iron, which can be provided alone or in combination with the multivitamin, as in prenatal vitamins.
After RYGB, we measure vitamin and iron levels at 2 weeks, 3 months, 6 months, 12 months, 18 months, and then yearly thereafter unless a major abnormality is identified.
CASE: New-onset abdominal pain and a history of bariatric surgery
A 28-year-old woman who is 14 weeks pregnant (G5P1A4) complains of abdominal pain, nausea, and vomiting for the past 3 days. Four years ago, she underwent a successful Roux-en-Y gastric bypass, and her body mass index (BMI) decreased from 61.1 to 34.8 as a result. Today, after unremarkable ultrasonography at an outside hospital, she is transferred to your facility, where she is noted to be afebrile and tachycardic (133 bpm) with a very tender abdomen and a white blood cell (WBC) count of 12.2×103/μL.
Could her symptoms be related to her history of gastric bypass? How should you manage her condition?
This case illustrates why it is imperative that Ob Gyns have a basic understanding of bariatric surgery—particularly the types of procedures being performed today and the complications that may arise.
In this case, urgent intervention is called for, given the elevated WBC count and extreme tenderness of the abdomen. This patient was taken to surgery without waiting for additional imaging studies. Operative findings included intussusception with necrosis, a late complication of the Roux-en-Y procedure, necessitating resection of 60 cm of small intestine ( FIGURE 1 ). Although fetal heart sounds remained encouraging overnight, fetal demise occurred the next day. The patient’s physical recovery was otherwise unremarkable.
This scenario is not as unlikely as you might imagine. A similar episode was described in 2004.1 The outcome: maternal and fetal death.

FIGURE 1 Life-threatening intestinal obstruction
Patchy and continuous necrosis is evident following reduction of an obstruction-causing intussusception.
A boom in bariatric surgery
The incidence of surgical weight-loss procedures increased sixfold over a recent 7-year time span.2 In 2008 alone, more than 200,000 people underwent this type of operation—at least half of them women of reproductive age.2,3 In my experience at the University Hospitals of Cleveland Bariatric Program, which I directed, 85% of patients were women.4 This percentage is similar to the gender breakdown in other bariatric programs.
Because you are called on to provide both primary and specialty care, you are very likely to encounter women who have undergone bariatric surgery or who are considering it. As the prevalence of morbid obesity continues to rise, so will the need for familiarity with surgical weight-loss reduction.
In this article, I offer an overview of bariatric surgery, focusing on:
- the different procedures performed today
- the potential complications associated with the most common operations
- the need for nutritional assessment.
In the process, I discuss the health benefits of bariatric surgery, including greater fertility and improved pregnancy outcomes.
How we talk about obesity
The language of obesity centers on a basic metric: the body mass index (BMI).5 This index was devised in the 19th century by Adolphe Quetelet to describe total body fat.
Consider its use to describe two markedly overweight individuals. One stands 6 ft and weighs 500 lb; the other stands 5 ft and weighs 400 lb. It is obvious which person is heavier; the BMI quantifies who is actually bigger. The first individual has a BMI of 67.8, whereas the shorter person has a BMI of 78, despite weighing 100 lb less.
The formula for determining BMI is:
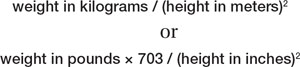
Among the imperfections of the BMI are its failure to differentiate between genders and its marked inaccuracy in persons who have significant muscle mass and limited body fat, such as body builders. However, it does make it possible to compare patients of different height and weight, thereby facilitating attempts to prognosticate the risk of illness and identify candidates for bariatric surgery ( TABLE 1 ).
The 1991 National Institutes of Health (NIH) Consensus Conference on Gastrointestinal Surgery for Severe Obesity recommended that bariatric surgery (restrictive and bypass procedures) be considered for patients who have a BMI of 40 or above and for patients who have a BMI of 35 or above with high-risk comorbidity.6
TABLE 1
How size is classified using the body mass index
| BMI | Classification | Risk of illness and comorbidity |
|---|---|---|
| <18.5 | Underweight | Increased |
| 18.5–24.9 | Normal | Average |
| 25–29.9 | Overweight | Increased |
| 30–34.9 | Obese (class I) | Moderate |
| 35–39.9 | Obese (class II) | Severe |
| ≥40 | Morbid obesity (class III) | Very severe |
| ≥50 | “Severe morbid obesity” | !!! Severe |
Most agree that bariatric surgery originated nearly 60 years ago with the jejunoileal bypass, a purely malabsorptive procedure in which the proximal jejunum was anastomosed to the distal ileum.7-9 This operation was based on the observation of dramatic weight loss following major small-bowel resection. The procedure was thought to allow obese patients to eat with abandon, thanks to the limited absorptive ability of the small intestine’s mucosal surface, which would impede digestion. However, the operation was fraught with complications, including protein calorie malnutrition, bacterial overgrowth, and even death. In addition, despite the dramatic reduction in the absorptive surface of the small intestine, some patients eventually regained weight.
One may still encounter patients who underwent jejunoileal bypass many years ago. One particular complication of this procedure is worth mentioning: hepatic cirrhosis. Any patient who has a history of jejunoileal bypass should have her liver function assessed before undergoing procedures that require general anesthesia.
At present, bariatric surgery may incorporate a component of malabsorption, but no other procedures are purely malabsorptive ( TABLE 2 ).
TABLE 2
Bariatric procedures for weight reduction, past and present
| Mechanism of action | Procedure |
|---|---|
| Malabsorptive | Jejunoileal bypass* |
| Restrictive | Vertical banded gastroplasty Gastric banding (Lap-Band) Laparoscopic sleeve gastrectomy Magenstrasse and Mill operation |
| Combination (malabsorptive and restrictive) | Roux-en-Y gastric bypass Biliopancreatic diversion Duodenal switch |
| Other | Gastric pacing† |
| * No longer performed | |
| †Experimental | |
Restrictive procedures
Vertical banded gastroplasty (VBG). This procedure is one of the earliest successful restrictive procedures ( FIGURE 2 ). Although it was originally performed via laparotomy, it is easily carried out laparoscopically. However, a risk of complication, such as severe reflux, and a low long-term success rate have decreased its widespread application.
Laparoscopic adjustable silicone band. The most common restrictive procedure performed today is the laparoscopic adjustable silicone band (Lap-Band) ( FIGURE 2 ). Among its benefits are:
- easy insertion, especially in comparison with the gastric bypass
- ability to adjust the amount of restriction (not possible with VBG)
- elimination of the need for supplemental nutrition
- reversibility.
Long-term results from a US population are not available because FDA approval did not occur until 2001. However, some centers outside the United States have reported unfavorable long-term outcomes. (See “Lap-Band complications may be pervasive,”.)
Laparoscopic sleeve gastrectomy (LSG). This operation was originally devised as the first stage of a duodenal switch. However, weight loss with this stage alone made LSG a bariatric procedure in its own right ( FIGURE 2 ).
Although this operation is categorized as restrictive, gastric sleeve resection eliminates the gastric fundus, the major site where ghrelin—“the hunger hormone”—is produced. This hormonal alteration may contribute to weight loss that occurs after LSG.
Magenstrasse and Mill operation. This operation converts the stomach into a long tube similar to the gastric sleeve. The greater curvature of the stomach is separated from the newly created tube but remains attached to the rest of the stomach at the antrum. (The greater curvature of the stomach is resected in the gastric sleeve procedure.)
Many insurers refuse to reimburse for restrictive operations other than the VBG and Lap-Band.

FIGURE 2 Three common surgeries
These restrictive procedures decrease the size of the stomach reservoir without impairing absorption of calories.
Combination procedures
Biliopancreatic diversion, duodenal switch. Despite the risk of protein malnutrition, these two procedures attempt to produce selective malabsorption of fat and carbohydrate as a means of treating the most severely obese patients. These operations carry the highest mortality rate and are therefore less likely to be offered routinely.
Roux-en-Y gastric bypass (RYGB). In the United States, the RYGB is performed more frequently than other procedures that combine malabsorption and restriction. It is considered by most to be the “gold standard” bariatric operation ( FIGURE 3 ).
The RYGB may induce weight loss through one or more of the following mechanisms:
- a decrease in the size of the gastric pouch. After the procedure, this pouch has a capacity of about 30 cc (size of a thumb)
- the small diameter of the gastrojejunal anastomosis, which is usually 1 cm
- glucose intolerance. About 50% of patients experience dumping symptoms when they ingest refined carbohydrates
- subclinical malabsorption. Food does not encounter the majority of digestive enzymes until it reaches the jejunojejunostomy anastomosis
- suppression of ghrelin production. Ghrelin is a hormone produced primarily in the gastric fundus and secondarily in the duodenum, both of which are bypassed with the RYGB. Plasma ghrelin levels peak before meals and are suppressed with food intake. After RYGB, patients demonstrate a flattening of this pattern, which appears to correlate with a suppression of appetite.10

FIGURE 3 Roux-en-Y gastric bypass
A gastric pouch is created at the top of the stomach and connected directly to the jejunum, bypassing the rest of the stomach and duodenum.
When is bariatric surgery indicated?
The NIH Consensus Conference identified patients who might be candidates for bariatric surgery, as I noted:
- patients who have a BMI of 40 or above, whether or not they have comorbidity
- patients who have a BMI of 35 or above and comorbidity.6
These recommendations have been adopted by third-party payers.
Potential comorbidities include hypertension, coronary artery disease (CAD), lipid abnormality, diabetes, obstructive sleep apnea (OSA), and severe osteoarthritis, among others.
When identified together, several of these comorbidities constitute metabolic syndrome, a risk factor for CAD. More than 50 million Americans are thought to exhibit this syndrome, which is diagnosed when three or more of the following are present:
- waist circumference ≥40 in (102 cm) in men; ≥35 in (88 cm) in women
- triglycerides ≥150 mg/dL
- high-density lipoprotein cholesterol <40 mg/dL in men; <50 mg/dL in women
- blood pressure ≥130/85 mm Hg
- fasting glucose ≥100 mg/dL.
Postsurgery weight loss has many benefits
Obstructive sleep apnea may resolve
Although this condition is known to be associated with obesity, it is drastically underdiagnosed.11 In our program, only 21% of patients had been given a diagnosis of OSA at the time of initial evaluation for surgery. After we obtained a polysomnogram (sleep study) for each of them, however, the true prevalence was determined to be 91%.
OSA can have serious consequences if it is untreated, and it may increase the risks associated with surgery and general anesthesia. By identifying and treating OSA before gastric bypass (RYGB), we were able to eliminate the respiratory ICU stay—a benefit that should be applicable to nonbariatric surgeries in the morbidly obese patient as well.12
With successful bariatric surgery, OSA abnormalities identified during polysomnography are dramatically improved or eliminated. Treatment with continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BIPAP) can usually be discontinued 6 months to 1 year after the dramatic weight loss associated with RYGB.
Cancer death rate may be reduced
Three consequences of morbid obesity not generally thought to be indications for bariatric surgery are:
- obesity-associated cancer
- decreased longevity
- infertility.
However, each of these conditions may be improved through successful bariatric surgery.
In a prospective study of more than 900,000 adults in the United States who were followed for 16 years, overweight or obesity was thought to account for 14% of cancer deaths in men and 20% of cancer deaths in women.13 Death rates for persons who had a BMI of at least 40 were 52% higher in men and 62% higher in women than they were in people who had a BMI below that threshold. BMI was significantly associated with cancer of the esophagus, colon and rectum, liver, gallbladder, pancreas, and kidney, as well as with non-Hodgkin’s lymphoma and multiple myeloma.
A trend toward an increasing risk of death with higher BMI was observed for cancer of the stomach (men), prostate, breast (women), uterus, ovary, and cervix.
The benefits of bariatric surgery in regard to cancer incidence and longevity were revealed in a 15-year, nonrandomized, prospective study involving more than 4,000 patients.14 At 10.9 years of follow-up, with a follow-up rate of 99.9%, this investigation concluded that bariatric surgery reduces mortality attributable to cardiovascular disease and cancer.14 Most surprising was the finding of a reduced incidence of cancer in patients who underwent bariatric surgery, compared with matched controls.14 An editorial accompanying this study summed it up in the headline: “The missing link—lose weight, live longer.”15
The Framingham risk score estimates the 10-year risk of CAD. This score is reduced by more than 50% after successful gastric bypass surgery.16
After surgery, fertility improves and pregnancy has better outcomes
From your practice, you’re certainly aware of the detrimental effects that obesity has on fertility, pregnancy, and fetal health.17-22 Although neither pregnancy nor impaired fertility is a primary indication for surgical weight reduction in an obese woman, bariatric surgery can improve the likelihood of fertility and successful pregnancy.
Advise patients to delay pregnancy after bariatric surgery. Although none of the bariatric operations performed today are contraindications to pregnancy, we caution all women to delay pregnancy—using two forms of birth control—until weight loss has stabilized. This usually takes about 12 to 18 months after surgery.
In our program, we identified 28 women who became pregnant following gastric bypass. Although we had cautioned all women to avoid early pregnancy, 10 became pregnant within 1 year of the bypass. Among these women, the rate of miscarriage was 40%, compared with 17% among the 18 women who waited more than 1 year to conceive. These percentages merit concern despite the small sample size.
Dao and colleagues concluded that the timing of pregnancy after gastric bypass is irrelevant.23 However, critical review of their data reveals an average weight gain of 4 lb in the group of women who became pregnant within a year after surgery (range, -70 to +45 lb), compared with 34 lb in the group that waited at least 1 year to conceive (range, +13 to +75 lb). As it was among our own patients, miscarriage was more common in the early group (24% vs 8% in the late group). Although these data are not statistically significant, they should arouse concern. I believe the most prudent advice to give women following bariatric surgery is to delay pregnancy until weight loss has stabilized.
The World Health Organization has estimated that there are 1 billion overweight adults on the planet. The problem of obesity, however, is concentrated in the United States. For well over a decade, the problem has escalated, with the US obesity rate referred to as “epidemic” and “a crisis”—among both adults and children.24
Among adults 20 years and older, the prevalence of obesity in the United States is 32%, and the prevalence of morbid obesity is almost 5%. Among adult women 20 to 39 years old, the prevalence of obesity and morbid obesity is 29% and 8%, respectively.27
Complications of bariatric surgery
Complications following bariatric surgery are intrinsic to the specific procedure performed. Because RYGB and the Lap-Band procedure constitute the overwhelming majority of bariatric operations, the most important complications associated with these surgeries will be addressed.
After RYGB, greatest risk is intestinal obstruction
Some of the early postoperative complications following RYGB are unlikely to be seen by physicians other than bariatric surgeons. They include anastomotic leaks, peritonitis, and bleeding. Pulmonary emboli should be managed as usual during the postoperative course. The amount of heparin necessary to achieve therapeutic anticoagulation may be quite substantial, but this fact should not deter the treating physician.
Nausea is almost universally present during the first few days after RYGB. When nausea and increasing food intolerance occur later (after the patient has demonstrated that food can be tolerated), it may indicate stenosis of the gastrojejunal anastomosis. The optimal study to evaluate these symptoms is a barium upper gastrointestinal (GI) swallow study. Stenosis can usually be managed by endoscopic dilatation.24
Intestinal obstruction. One of the most serious problems following RYGB is intestinal obstruction, which may be caused by internal hernia, adhesion, intussusception (not rare in the gastric bypass patient), and other less common causes ( FIGURE 1 ). Intestinal obstruction may occur early or years after the procedure.
In the RYGB patient, obstruction manifests with prominent pain, but distension and vomiting are usually absent, and plain abdominal radiographs are generally normal. The reason for this unusual presentation? The biliopancreatic limb tends to be the segment involved in the blockage, thereby creating a closed-loop obstruction. The most efficient way to diagnose this potentially life-threatening problem is by computed tomography (CT) ( FIGURE 4 ).

FIGURE 4 CT facilitates diagnosis of closed-loop obstruction
These computed tomography images reveal obstruction of the remnant stomach, duodenum, and proximal jejunum (the biliopancreatic limb) in a patient who has undergone Roux-en-Y gastric bypass.
Lap-Band complications may be pervasive
The Lap-Band procedure is often touted as having very low morbidity and mortality. However, data from outside the United States, where longer follow-up has been conducted, have cast a shadow of doubt on the supposed benefits of this restrictive procedure.
One study involving nearly 900 patients and 12 years of follow-up identified a rate of late complication of nearly 28% (2.3%, band erosion; 15.5%, pouch dilatation; 8.9%, port problems) and a rate of major reoperation of nearly 30%. The failure rate increased over time, reaching 65% at 10 years.
Despite these concerns, there is widespread enthusiasm among patients and bariatric surgeons who are proponents of this procedure.
A life-threatening complication. Any physician who might care for a patient who has undergone a Lap-Band procedure should be aware of one potentially life-threatening consequence: a slipped band. This complication can lead to incarceration of the stomach—usually the fundus—which, if left untreated, causes gastric infarction.
The main symptom of band slippage is pain. Any patient who has a Lap-Band and who reports significant upper abdominal pain must be evaluated to exclude slippage. Evaluation usually consists of a swallow study. Surgical correction is mandatory and urgent.
Like complications of surgery, the nutritional demands following bariatric surgery depend on the type of procedure that is performed.
After Lap-Band and other purely restrictive procedures,
nutritional levels remain stable
With restrictive procedures, such as the Lap-Band operation, the GI tract is essentially unaffected. Consequently, vitamin and micronutrient absorption remains intact. In these patients, we usually recommend a daily multivitamin. We assess serum levels to determine whether calcium and vitamin D supplementation might be necessary, which is especially important in female patients.
Close monitoring is needed after RYGB
After RYGB and other combined procedures, the question of nutritional supplementation becomes more complex.25 Bypass of the duodenum and upper jejunum affect absorption of iron, vitamin B12, calcium, and vitamin D. Serum calcium assessment is inadequate to determine the adequacy of calcium and vitamin D supplementation.
We evaluate parathyroid hormone and alkaline phosphatase levels in these patients. Any increase in these levels in the presence of normal serum calcium suggests the induction of secondary hyperparathyroidism and the need for additional calcium and vitamin D.26
Our recommendations for all patients following RYGB are a daily multivitamin, vitamin B12, and supplemental calcium and vitamin D. Vitamin B1 (thiamine) deficiency is extremely rare unless prolonged vomiting has been present. If B1 deficiency is suspected, administer thiamine before giving glucose because glucose can make symptoms worse.
Most women also need supplemental iron, which can be provided alone or in combination with the multivitamin, as in prenatal vitamins.
After RYGB, we measure vitamin and iron levels at 2 weeks, 3 months, 6 months, 12 months, 18 months, and then yearly thereafter unless a major abnormality is identified.
1. Moore KA, Ouyang DW, Whang EE. Maternal and fetal deaths after gastric bypass surgery for morbid obesity. N Engl J Med. 2004;351:721-722.
2. Bariatric Surgery in Women of Reproductive Age: Special Concerns for Pregnancy. Structured Abstract. Rockville, Md: Agency for Healthcare Research and Quality; November 2008. Available at: www.ahrq.gov/Clinic/tp/barireptp.htm. Accessed Dec. 8, 2009.
3. American Society for Metabolic and Bariatric Surgery. Metabolic and bariatric surgery fact sheet. Available at: www.asmbs.org/Newsite07/media/asmbs_fs_surgery.pdf. Accessed Dec. 8, 2009.
4. Stellato TA, Hallowell PT, Crouse C, Schuster M, Petrozzi MC. Two-day length of stay following open Roux-en-Y gastric bypass: is it feasible, safe and reasonable? Obes Surg. 2004;14:27-34.
5. Oria HE, Carrasquilla C, Cunningham P, et al. American Society for Bariatric Surgery Standards Committee, 2004–2005. Guidelines for weight calculations and follow-up in bariatric surgery. Surg Obes Relat Dis. 2005;1:67-68.
6. Gastrointestinal surgery for severe obesity: National Institutes of Health Consensus Development Conference Statement. Am J Clin Nutr. 1992;55(Suppl 2):615S-619S.
7. MacDonald KG, Jr. Overview of the epidemiology of obesity and the early history of procedures to remedy morbid obesity. Arch Surg. 2003;138:357-360.
8. Buchwald H. Overview of bariatric surgery. J Am Coll Surg. 2002;194:367-375.
9. DeMaria EJ. Bariatric surgery for morbid obesity. N Engl J Med. 2007;356:2176-2183.
10. Cummings DE, Weigle DS, Frayo RS, et al. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med. 2002;346:1623-1630.
11. Hallowell PT, Stellato TA, Schuster M, et al. Potentially life-threatening sleep apnea is unrecognized without aggressive evaluation. Am J Surg. 2007;193:364-367.
12. Hallowell PT, Stellato TA, Petrozzi MC, et al. Eliminating respiratory intensive care unit stay after gastric bypass surgery. Surgery. 2007;142:608-612.
13. Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003;348:1625-1638.
14. Sjöström L, Narbro K, Sjöström CD, et al. Swedish Obese Subjects Study. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741-752.
15. Bray GA. The missing link—lose weight, live longer. N Engl J Med. 2007;357:818-820.
16. Kligman MD, Dexter DJ, Omer S, Park AE. Shrinking cardiovascular risk through bariatric surgery: application of Framingham risk score in gastric bypass. Surgery. 2008;143:533-538.
17. Linné Y. Effects of obesity on women’s reproduction and complications during pregnancy. Obes Rev. 2004;5:137-143.
18. Pasquali R, Pelusi C, Genghini S, Cacciari M, Gambineri A. Obesity and reproductive disorders in women. Hum Reprod Update. 2003;9:359-372.
19. Baeten JM, Bukusi EA, Lambe M. Pregnancy complications and outcomes among overweight and obese nulliparous women. Am J Public Health. 2001;91:436-440.
20. Lashen H, Fear K, Sturdee DW. Obesity is associated with increased risk of first trimester and recurrent miscarriage: matched case-control study. Hum Reprod. 2004;19:1644-1646.
21. Cnattingius S, Bergström R, Lipworth L, Kramer MS. Prepregnancy weight and the risk of adverse pregnancy outcomes. N Engl J Med. 1998;338:147-152.
22. Pathi A, Esen U, Hildreth A. A comparison of complications of pregnancy and delivery in morbidly obese and non-obese women. J Obstet Gynaecol. 2006;26:527-530.
23. Dao T, Kuhn J, Ehmer D, Fisher T, McCarty T. Pregnancy outcomes after gastric-bypass surgery. Am J Surg. 2006;192:762-766.
24. Stellato TA, Crouse C, Hallowell PT. Bariatric surgery: creating new challenges for the endoscopist. Gastrointest Endosc. 2003;57:86-94.
25. Poitou Bernert C, Ciangura C, Coupaye M, Czernichow S, Bouillot JL, Basdevant A. Nutritional deficiency after gastric bypass: diagnosis, prevention and treatment. Diabetes Metab. 2007;33:13-24.
26. Jin J, Robinson AV, Hallowell PT, Jasper JJ, Stellato TA, Wilhelm SM. Increases in parathyroid hormone (PTH) after gastric bypass surgery appear to be of a secondary nature. Surgery. 2007;142:914-920.
27. Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA. 2006;295:1549-1555.
1. Moore KA, Ouyang DW, Whang EE. Maternal and fetal deaths after gastric bypass surgery for morbid obesity. N Engl J Med. 2004;351:721-722.
2. Bariatric Surgery in Women of Reproductive Age: Special Concerns for Pregnancy. Structured Abstract. Rockville, Md: Agency for Healthcare Research and Quality; November 2008. Available at: www.ahrq.gov/Clinic/tp/barireptp.htm. Accessed Dec. 8, 2009.
3. American Society for Metabolic and Bariatric Surgery. Metabolic and bariatric surgery fact sheet. Available at: www.asmbs.org/Newsite07/media/asmbs_fs_surgery.pdf. Accessed Dec. 8, 2009.
4. Stellato TA, Hallowell PT, Crouse C, Schuster M, Petrozzi MC. Two-day length of stay following open Roux-en-Y gastric bypass: is it feasible, safe and reasonable? Obes Surg. 2004;14:27-34.
5. Oria HE, Carrasquilla C, Cunningham P, et al. American Society for Bariatric Surgery Standards Committee, 2004–2005. Guidelines for weight calculations and follow-up in bariatric surgery. Surg Obes Relat Dis. 2005;1:67-68.
6. Gastrointestinal surgery for severe obesity: National Institutes of Health Consensus Development Conference Statement. Am J Clin Nutr. 1992;55(Suppl 2):615S-619S.
7. MacDonald KG, Jr. Overview of the epidemiology of obesity and the early history of procedures to remedy morbid obesity. Arch Surg. 2003;138:357-360.
8. Buchwald H. Overview of bariatric surgery. J Am Coll Surg. 2002;194:367-375.
9. DeMaria EJ. Bariatric surgery for morbid obesity. N Engl J Med. 2007;356:2176-2183.
10. Cummings DE, Weigle DS, Frayo RS, et al. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med. 2002;346:1623-1630.
11. Hallowell PT, Stellato TA, Schuster M, et al. Potentially life-threatening sleep apnea is unrecognized without aggressive evaluation. Am J Surg. 2007;193:364-367.
12. Hallowell PT, Stellato TA, Petrozzi MC, et al. Eliminating respiratory intensive care unit stay after gastric bypass surgery. Surgery. 2007;142:608-612.
13. Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003;348:1625-1638.
14. Sjöström L, Narbro K, Sjöström CD, et al. Swedish Obese Subjects Study. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741-752.
15. Bray GA. The missing link—lose weight, live longer. N Engl J Med. 2007;357:818-820.
16. Kligman MD, Dexter DJ, Omer S, Park AE. Shrinking cardiovascular risk through bariatric surgery: application of Framingham risk score in gastric bypass. Surgery. 2008;143:533-538.
17. Linné Y. Effects of obesity on women’s reproduction and complications during pregnancy. Obes Rev. 2004;5:137-143.
18. Pasquali R, Pelusi C, Genghini S, Cacciari M, Gambineri A. Obesity and reproductive disorders in women. Hum Reprod Update. 2003;9:359-372.
19. Baeten JM, Bukusi EA, Lambe M. Pregnancy complications and outcomes among overweight and obese nulliparous women. Am J Public Health. 2001;91:436-440.
20. Lashen H, Fear K, Sturdee DW. Obesity is associated with increased risk of first trimester and recurrent miscarriage: matched case-control study. Hum Reprod. 2004;19:1644-1646.
21. Cnattingius S, Bergström R, Lipworth L, Kramer MS. Prepregnancy weight and the risk of adverse pregnancy outcomes. N Engl J Med. 1998;338:147-152.
22. Pathi A, Esen U, Hildreth A. A comparison of complications of pregnancy and delivery in morbidly obese and non-obese women. J Obstet Gynaecol. 2006;26:527-530.
23. Dao T, Kuhn J, Ehmer D, Fisher T, McCarty T. Pregnancy outcomes after gastric-bypass surgery. Am J Surg. 2006;192:762-766.
24. Stellato TA, Crouse C, Hallowell PT. Bariatric surgery: creating new challenges for the endoscopist. Gastrointest Endosc. 2003;57:86-94.
25. Poitou Bernert C, Ciangura C, Coupaye M, Czernichow S, Bouillot JL, Basdevant A. Nutritional deficiency after gastric bypass: diagnosis, prevention and treatment. Diabetes Metab. 2007;33:13-24.
26. Jin J, Robinson AV, Hallowell PT, Jasper JJ, Stellato TA, Wilhelm SM. Increases in parathyroid hormone (PTH) after gastric bypass surgery appear to be of a secondary nature. Surgery. 2007;142:914-920.
27. Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM. Prevalence of overweight and obesity in the United States, 1999–2004. JAMA. 2006;295:1549-1555.
Testing the BRIEF Health Literacy Screening Tool
Surgical Management of Spontaneous Rupture of Umbilical Hernia Using Biological Mesh
Do Diabetic Foot Clinics Spread MRSA?
Diagnosing and Treating Hansen's Disease
According to data from the World Health Organization (WHO), the number of new cases of Hansen’s disease (HD, or leprosy) identified worldwide declined by about 20% from 2003 to 2004. At least 116 of 122 countries once considered leprosy—endemic have achieved a prevalence rate of less than one case per 10,000 population.1,2
Meanwhile, the number of active HD infections in the United States catalogued through the CDC and the National Hansen’s Disease Program (NHDP) rose from 76 cases in 2000 to 166 in 2005, with an additional 137 cases in 2006 (the most recent year for which complete figures are available).3,4 Currently, 6,500 US residents are known to have HD, of whom 3,300 require care for active disease.3
Accurate diagnosis and straightforward, aggressive treatment can mean certain cure for this dreaded disease, as it is responsive to a combination of available drugs. For primary care providers in the US, familiarity with this condition can play a role in eliminating it altogether.
Background and Etiology
HD is believed to have originated in East Africa with the pseudonym of leprosy, from the Greek lepi, meaning “fish scales.” Although HD is one of the oldest known human infections, it is not well understood by many Western health care providers. Once considered highly contagious and easily transmissible, HD has proven to be neither: 95% of the human population is not susceptible to the responsible pathogen.3 HD retains its reputation as a devastating illness when left untreated, however, with sequelae often including significant sensory and motor dysfunction.5
Leprosy is a chronic bacterial disease caused by Mycobacterium leprae. Mycobacterium is the same rod-shaped, acid-fast bacillus (AFB) implicated in pulmonary tuberculosis, cutaneous tuberculosis, and numerous cutaneous nontuberculoid infections.6,7
HD transmission is believed to occur almost exclusively by nasal droplet, inhaled by way of others’ nares or lungs. Research continues in possible transmission by skin-to-skin contact.8,9
Leprosy is particularly challenging for immunocompromised persons, such as those with HIV/AIDS or tuberculosis. According to data from the NHDP, HD in the US is found primarily among undocumented immigrants who live in close quarters. Cases considered “home-grown” rather than “imported”5 are most commonly diagnosed in Hawaii and in a swath from Texas to Georgia and south toward Florida. Pockets of HD incidence are also found in California, New York, and Massachusetts.4
Clinicians’ lack of familiarity with HD and its relative rarity in the US help explain why diagnosis and treatment are commonly delayed. As a patient’s disease progresses from an insidious, gradual, and relatively painless onset, the primary care provider may be grappling with an extensive differential diagnosis (see “Differential Diagnosis for Hansen’s Disease [Leprosy],”10,11 below). Even after confirming a diagnosis of HD, the provider must contend with a lack of information and misinformation about HD among staff members, the patient’s family, and society at large, as well as the associated stigma. Patient education and effective treatment are of comparable importance.
Patient Presentation and History
A 40-year-old Latino man presented to a primary care provider with a well-demarcated, erythematous patch on his right upper back that had raised outer edging and faint flaking. The patch was surrounded by several smaller but similar asymmetrical macules. The patient denied pruritus but stated that the area “felt funny.” He said the lesion might have been spreading during the previous several months, but he could not be certain because of its location.
The man denied any medication use except for a topical antibiotic ointment he had applied to the affected area with no improvement. His provider made a diagnosis of tinea corporis and prescribed an antifungal cream, naftifine. After two weeks without results, the patient was referred to a dermatologist.
During the assessment, it became apparent that the patient was experiencing numbness in his fingertips and general muscle weakness in his arms. A KOH prep was difficult to obtain, as the lesion site was somewhat smooth. Any dermatophytes that may have been present had been eliminated by the naftifine, and KOH results were negative.
A 5.0-mm punch biopsy from the largest lesion was ordered. A preliminary diagnosis of sarcoidosis (chronic inflammation with an unusual skin manifestation) was made, with serious consideration given to early cutaneous T-cell lymphoma (mycosis fungoides). A high-potency steroid was prescribed for five days’ use, twice daily.
On his return to the dermatologist, the patient reported that the site was no better. He was informed that the dermatologist and the pathologist, both with considerable experience, had detected no bacilli or dermal nerve infiltration in the tissue sample but had observed abundant lymphocytic infiltration. This finding, coupled with the patient’s decreased sensation, muscle fatigue, and status as a recent immigrant from a South American country, led to an initial diagnosis of borderline leprosy (BB).
Physical Examination and Disease Progression
If the first provider suspected a fungal infection during the initial examination, he should have completed a KOH scraping to confirm that suspicion. While superficial fungal infections are extremely common, particularly in the southeastern US, a sampling of scale with KOH 5% to 20% and the absence of dermatophytes under the microscope might have permitted a quick rule-out.12 Additionally, dermatophytic infections are very responsive to current medications within days, not weeks.
Thus, the key to an accurate diagnosis of leprosy begins with a physical examination that takes into account both chronic unresolved cutaneous lesions and neurologic changes in sensitivity.
A great majority of patients with HD have no noteworthy clinical symptoms until the first skin manifestations. While sensory neuropathy is prominent in all patients, additional findings (depending on the type of leprosy involved) may include plantar ulcers, chronic nasal congestion with early evidence of cartilage loss over the nasal bridge, chronic nosebleeds, cranial nerve palsies, and eye paralysis.12 In rare cases, hypogonadism may develop, or the clinical team may discover amyloidosis, an accumulation of insoluble proteins in the tissues that impairs function in various organs; this condition is usually detected by skin biopsy. If early or indeterminate leprosy is identified, neither form persists; rather, the condition will progress to one of the following:
• Borderline lepromatous leprosy (BL) and lepromatous leprosy (LL) offer very high resistance to treatment and are considered malignant forms of HD. Findings in patients with BL or LL include multiple lesions in various crazy-quilt presentations: macules, papules, plaques, nodules, masses, and patches of deep, erythematous infiltration across the extremities, face, and trunk.12 Loss of all hair except on the scalp is possible. Ulcers may form on the palms of the hands and soles of the feet.
In both BL and LL, results on AFB smears will reflect moderately high or very high concentrations of the pathogen in the patient’s system. Thus, these forms of HD are defined as multibacillary. If either is left untreated, the accumulation of proteins attributed to amyloidosis and secondary infections that develop eventually lead to death.
• Borderline leprosy (BB) has already been described in the case patient, with five or six ill-defined lesions and mild impairment of the peripheral nervous system. Aggressive treatment of this form of HD is important. A strong immune system provides no guarantee of successful treatment, and therapy must forestall a likely continuum to more serious forms.13
• Borderline tuberculoid leprosy (BT) and tuberculoid leprosy (TT) are both forms of paucibacillary HD. They offer little resistance to treatment in the patient with a healthy immune system. Lesions are generally scant (fewer than five; see figure), and the AFB smear reveals 0 to 10 bacteria in 100 fields.12
For the primary care provider, a thorough physical examination also requires familiarity with reasonable differentials. Hallmarks of conditions less likely to be leprosy include nausea, vomiting, or diarrhea, fever and chills, rectal bleeding and unexplained weight loss, headaches, and shortness of breath.
Diagnostic Testing
To help gauge progression of the disease and assess effectiveness of therapy, it is common practice to conduct a neurosensory test at each office visit. This is performed by touching the patient’s extremities and the periphery of lesions with a simple nylon monofilament.13 The patient’s response or lack of response is documented and changes are noted for future therapeutic decision making.
In patients with suspected HD, the NHDP recommends that a full-thickness 4.0-mm elliptical or punch biopsy be performed at the periphery of the largest or most active lesion and that slides be sent to the NHDP in formalin or paraffin for review.14 Experienced NHDP pathologists may order additional stains. A detailed protocol can be found at www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm.
Proficiency in performing slit smears, a technique to obtain tissue fluid, is also helpful.15 Measurement of the number of bacteria in a set number of fields allows for calculation of the host’s bacterial index, making it possible to determine an appropriate treatment level.
The preferred sites for the slit smear are active lesions, if possible from both ear lobes plus two additional locations. After alcohol swabbing, a tight, bloodless pinch is made, with a 3.0-mm incision that yields blood and fluid. The resulting culture is examined for AFB, which are found abundantly in lepromatous leprosy (whereas tuberculoid leprosy, by definition, should yield none or few). This smear may be repeated at three- to six-month intervals to determine effectiveness of therapy. It is considered superior to a nasal culture, which often yields false-negative results.16
After the Diagnosis
In the US, HD is a nationally notifiable disease: Health care providers are required by law to inform the CDC of the known particulars while complying with HIPAA requirements.17
That the case patient was a recent immigrant from a country with a high incidence of HD, coupled with a loss of sensation in his extremities, led to the correct diagnosis. His lack of response to antifungals or anti-inflammatory agents, though frustrating to both patient and provider, was helpful in excluding two common conditions in the differential diagnosis. Other possibilites that were considered ultimately did not ring true:
Vitiligo, a loss of pigment that produces white patches resembling borderline leprosy but lacks the raised edging18;
Granuloma annulare, a lightly raised, ring-shaped lesion that is usually found on the extremities19; and
Lupus erythematosus, in which patients almost universally experience pruritus and burning, whereas lack of sensation is key to a diagnosis of leprosy.20
Treatment and Management
Health care providers must appear confident to instill confidence in their patients. Any provider with the knowledge and acuity to make a diagnosis of leprosy should be adequately equipped to manage the patient’s care. However, the hesitant or uncertain provider should consider referral to dermatology or to infectious disease for continued management.
The NHDP recognizes the WHO classification of leprosy as multibacillary or paucibacillary (observing a “simple clinical rule” of six or more lesions representing multibacillary HD and five or fewer, paucibacillary HD1), with additional consideration to the three major disease subtypes mentioned earlier: TT, which is treatable; BB, which is considered unstable; and LL, which is resistant to treatment. NHDP and WHO recommendations for treatment differ slightly but address the same goals: complete elimination of the pathogen, preservation of muscle and nerve function, prevention of secondary infections, and management of the adverse effects of medication.1,21 Adhering to universal precautions is important.
In the US, the multidrug treatment regimen for paucibacillary (TT or BT) leprosy comprises one year of oral rifampin 600 mg/d and oral dapsone 100 mg/d21; WHO, in deference to cost containment in developing countries, recommends a six-month regimen of these agents (ie, rifampin 600 mg/mo and dapsone 100 mg/d).1 A third drug sometimes given to allay active neuritis is oral clofazimine 50 mg/d for one year21 (per WHO recommendations, a monthly dose of 300 mg, plus 50 mg/d1). In patients with preexisting or subsequent anemia, dapsone dosing should be lowered to 50 mg, the minimally effective level. Of note, clofazimine is no longer commercially available in the US and is held by the NHDP as an investigational new drug for treatment of US patients.21
According to US recommendations,21 use of this three-agent regimen is extended to two years for patients with multibacillary (LL, BL, BB) leprosy. For these patients, WHO recommends its regimen for 12 months.1
Rifampin is a powerful antibiotic used for effective treatment of tuberculosis, methicillin-resistant Staphylococcus aureus, gonorrhea, Listeria, and Haemophilus influenzae. However, its use has been shown to reduce the efficacy of anticoagulants, oral contraceptives, and prednisone, possibly requiring therapeutic adjustments.22 Recommended alternatives for rifampin include (in the order of preference) minocycline 100 mg/d, ofloxacin 400 mg/d, levofloxacin 500 mg/d, or twice-daily clarithromycin 500 mg.13
Patient compliance with the regimen is facilitated by convenient blister packs that contain standard weekly and monthly doses of these medications, all clearly marked. Many local health departments make these medications available at no cost to patients.
Patient Education
Informing patients what to expect regarding their medications, necessary lab work, and avoiding transmission of HD can ease worry, protect others from illness, and increase the likelihood of an excellent prognosis. Patients with lepromatous (multibacillary) HD should be urged to limit their interactions with others at work or school (particularly preventing exposure to their nasal excretions) until a low bacterial index is achieved. For close contacts of patients recently diagnosed with HD, single-dose rifampin has been shown to provide effective prevention for two years.23
Emphasizing adherence to daily multidrug therapy minimizes the chance that bacterial resistance will develop. Patients should be given complete information about the individual agents used:
Rifampin 600 mg is the maximum daily dose for an adult. Hepatotoxicity is a concern, and liver enzyme levels (ie, aspartate aminotransferase, alanine aminotransferase) should be measured at three-month intervals. It is important to obtain a history of previous liver disease, exposure to hepatitis, and drug and alcohol use. In otherwise healthy adults, rifampin has the curious effect of turning bodily fluids such as urine and tears bright red. Forewarning patients of this benign development may prevent anxious reactions. Contact lenses may be irreversibly stained.24
Dapsone, for many years the first-line monotherapy for leprosy, is a bacteriostatic, sulfa-based antibiotic that only prevents the multiplication of bacteria, but it is still worthy of respect. Before dapsone is initiated, a screening test for glucose-6-phosphate dehydrogenase deficiency should be performed to determine the risk for dapsone-associated hemolysis.24 Dapsone use is contraindicated in women who are breastfeeding. In patients with severe heart conditions, dapsone can reduce oxygen flow, as evidenced by a bluish discoloration of the lips and fingertips. Mild anemia, headache, gastrointestinal upset and nausea are all common adverse effects.
Clofazimine, a soft gelatin capsule, may cause gray-blue or red hyperpigmentation in individuals who are light-skinned; this effect reverses once the drug is discontinued. Nausea, vomiting, and abdominal pain have been reported. This slow-release bactericidal drug has an extremely long half-life of 70 days. It provides anti-inflammatory action similar to that of prednisone without the latter agent’s long-term adverse effects.24
The Patient’s Outcome
The case patient was the second with HD seen in this dermatology practice. A man who presented on an earlier occasion, possibly an undocumented worker in fear of deportation, seemed concerned about the reporting process, even though he was assured that no personal information would be released. He did not return for follow-up, and attempts to reach him proved fruitless.
The case patient, by contrast, was pleasant and cooperative. He appeared to understand his condition, the required treatment and its possible adverse effects, and the need for continued lab work and regular follow-up appointments. A day laborer in a large factory, he was particularly careful to avoid transmission of his illness to his family or co-workers.
Within two months of beginning multidrug therapy, he experienced quick clearance of the lesions on his back. His bacterial index approached 0 by the time he underwent the second smear. Although he complained of loose stools and classic orange-red bodily fluid throughout the entire year of treatment, he resisted the temptation to discontinue the regimen.
He has experienced no relapses in six years and is considered cured.
Conclusion
Only one in every 20 people is susceptible to infection with M leprae, but cases of leprosy continue to be reported in the US, and illness is of particular concern among immunocompromised individuals. Treatment delay is common because of the condition’s relative rarity in the US, but therapy with standard antibiotics is extremely effective. Lengthy isolation from family and community is not often needed, and relapses are rare. Fully restored health is the rule rather than the exception for patients treated for Hansen’s disease.
1. World Health Organization. Global strategy for further reducing the leprosy burden and sustaining leprosy control activities 2006-2010: operational guidelines. www.who.int/lep/resources/SEAGLP20062.pdf. Accessed November 17, 2009.
2. World Health Organization Communicable -Diseases Department. Leprosy: frequently as-ked questions. www.searo.who.int/EN/Section10/Section373_11716.htm. Accessed November 17, 2009.
3. National Hansen’s Disease (Leprosy) Program. www.hrsa.gov/hansens. Accessed November 23, 2009.
4. Health Resources and Services Administration, US Department of Health and Human Services, National Hansen’s Disease Program. A summary of Hansen’s disease in the United States—2006. ftp://ftp.hrsa.gov/hansens/2006RegistryReport.pdf. Accessed November 17, 2009.
5. Boggild AK, Correia JD, Keystone JS, Kain KC. Leprosy in Toronto: an analysis of 184 imported cases. CMAJ. 2004;170(1):55-59.
6. Cheesbrough M. Bacterial pathogens. In: District Laboratory Practice in Tropical Countries. Part 2. 2nd ed. Cambridge, UK: Cambridge University Press. 2006;157-234.
7. Sasaki S, Takeshita F, Okuda K, Ishii N. Mycobacterium leprae and leprosy: a compendium. Microbiol Immunol. 2001;45(11):729–736.
8. Job CK, Jayakumar J, Kearney M, Gillis TP. Transmission of leprosy: a study of skin and nasal secretions of household contacts of leprosy patients using PCR. Am J Trop Med Hyg. 2008; 78(3):518-521.
9. Girdhar BK. Skin to skin transmission of leprosy. Indian J Dermatol Venereol Leprol. 2005;71(4):223-225.
10. National Leprosy Eradication Programme, Directorate General of Health Services (Government of India). Differential diagnosis for leprosy (2009). http://nlep.nic.in/pdf/Annex-%20III% 20Differential%20diagnosis.pdf. September 14, 2009.
11. Kim EC. Hansen disease: differential diagnoses and workup (2006). http://emedicine.medscape .com/article/1213853-diagnosis. Accessed November 17, 2009.
12. James WD, Berger TG, Elston DM. Hansen’s disease. In: Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Philadelphia, PA: Saunders. 2005;343-366.
13. Leprosy. In: Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 2nd ed. Philadelphia, PA: WB Saunders; 2005.
14. National Hansen’s Disease Program. Skin biopsy in the diagnosis of Hansen’s disease. www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm. Accessed November 17, 2009.
15. Bhushan P, Sardana K, Koranne RV, et al. Diagnosing multibacillary leprosy: a comparative evaluation of diagnostic accuracy of slit-skin smear, bacterial index of granuloma and WHO operational classification. Indian J Dermatol Venereol Leprol. 2008;74(4):322-326.
16. Wolff K, Johnson R, Suurmond R. Leprosy. In: Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. Chicago, IL: McGraw-Hill Professional; 2005:655-661.
17. Centers for Disease Control and Prevention. Summary of notifiable diseases—United States, 2007. MMWR Morb Mortal Wkly Rep. 2009;56 (53):3, 10, 22.
18. American Academy of Dermatology. Vitiligo. www.aad.org/public/publications/pamphlets/com mon_vitilgo.html. Accessed November 17, 2009.
19. American Academy of Dermatology. Granuloma annulare. www.aad.org/public/publica tions/pamphlets/common_granuloma.html. Accessed November 17, 2009.
20. American Academy of Dermatology. Lupus and the skin. www.aad.org/public/publications/pamphlets/common_lupus.html. Accessed November 17, 2009.
21. National Hansen’s Disease Program. Recommended treatment regimens. www.hrsa.gov/han sens/clinical/regimens.htm. Accessed November 17, 2009.
22. Chen J, Raymond K. Roles of rifampicin in drug-drug interactions: underlying molecular mechanisms involving the nuclear pregnane X receptor. Ann Clin Microbiol Antimicrob. 2006 Feb 15;5:3.
23. Moet FJ, Pahan D, Oskam L, Richardus JH; COLEP Study Group. Effectiveness of single dose rifampicin in preventing leprosy in close contacts of patients with newly diagnosed leprosy: cluster randomised controlled trial. BMJ. 2008;336 (7647):761-764.
24. World Health Organization. WHO model prescribing information: drugs used in leprosy. http://apps.who.int/medicinedocs/fr/d/Jh2988e/ 14.2.html. Accessed November 17, 2009.
25. Bruce S, Schroeder TL, Ellner K, et al. Armadillo exposure and Hansen’s disease: an epidemiologic survey in southern Texas. J Am Acad Dermatol. 2000;43(2 pt 1):223-228.
26. Clark BM, Murray CK, Horvath LL, et al. Case-control study of armadillo contact and Hansen’s disease. Am J Trop Med Hyg. 2008;78(6):962-967.
According to data from the World Health Organization (WHO), the number of new cases of Hansen’s disease (HD, or leprosy) identified worldwide declined by about 20% from 2003 to 2004. At least 116 of 122 countries once considered leprosy—endemic have achieved a prevalence rate of less than one case per 10,000 population.1,2
Meanwhile, the number of active HD infections in the United States catalogued through the CDC and the National Hansen’s Disease Program (NHDP) rose from 76 cases in 2000 to 166 in 2005, with an additional 137 cases in 2006 (the most recent year for which complete figures are available).3,4 Currently, 6,500 US residents are known to have HD, of whom 3,300 require care for active disease.3
Accurate diagnosis and straightforward, aggressive treatment can mean certain cure for this dreaded disease, as it is responsive to a combination of available drugs. For primary care providers in the US, familiarity with this condition can play a role in eliminating it altogether.
Background and Etiology
HD is believed to have originated in East Africa with the pseudonym of leprosy, from the Greek lepi, meaning “fish scales.” Although HD is one of the oldest known human infections, it is not well understood by many Western health care providers. Once considered highly contagious and easily transmissible, HD has proven to be neither: 95% of the human population is not susceptible to the responsible pathogen.3 HD retains its reputation as a devastating illness when left untreated, however, with sequelae often including significant sensory and motor dysfunction.5
Leprosy is a chronic bacterial disease caused by Mycobacterium leprae. Mycobacterium is the same rod-shaped, acid-fast bacillus (AFB) implicated in pulmonary tuberculosis, cutaneous tuberculosis, and numerous cutaneous nontuberculoid infections.6,7
HD transmission is believed to occur almost exclusively by nasal droplet, inhaled by way of others’ nares or lungs. Research continues in possible transmission by skin-to-skin contact.8,9
Leprosy is particularly challenging for immunocompromised persons, such as those with HIV/AIDS or tuberculosis. According to data from the NHDP, HD in the US is found primarily among undocumented immigrants who live in close quarters. Cases considered “home-grown” rather than “imported”5 are most commonly diagnosed in Hawaii and in a swath from Texas to Georgia and south toward Florida. Pockets of HD incidence are also found in California, New York, and Massachusetts.4
Clinicians’ lack of familiarity with HD and its relative rarity in the US help explain why diagnosis and treatment are commonly delayed. As a patient’s disease progresses from an insidious, gradual, and relatively painless onset, the primary care provider may be grappling with an extensive differential diagnosis (see “Differential Diagnosis for Hansen’s Disease [Leprosy],”10,11 below). Even after confirming a diagnosis of HD, the provider must contend with a lack of information and misinformation about HD among staff members, the patient’s family, and society at large, as well as the associated stigma. Patient education and effective treatment are of comparable importance.
Patient Presentation and History
A 40-year-old Latino man presented to a primary care provider with a well-demarcated, erythematous patch on his right upper back that had raised outer edging and faint flaking. The patch was surrounded by several smaller but similar asymmetrical macules. The patient denied pruritus but stated that the area “felt funny.” He said the lesion might have been spreading during the previous several months, but he could not be certain because of its location.
The man denied any medication use except for a topical antibiotic ointment he had applied to the affected area with no improvement. His provider made a diagnosis of tinea corporis and prescribed an antifungal cream, naftifine. After two weeks without results, the patient was referred to a dermatologist.
During the assessment, it became apparent that the patient was experiencing numbness in his fingertips and general muscle weakness in his arms. A KOH prep was difficult to obtain, as the lesion site was somewhat smooth. Any dermatophytes that may have been present had been eliminated by the naftifine, and KOH results were negative.
A 5.0-mm punch biopsy from the largest lesion was ordered. A preliminary diagnosis of sarcoidosis (chronic inflammation with an unusual skin manifestation) was made, with serious consideration given to early cutaneous T-cell lymphoma (mycosis fungoides). A high-potency steroid was prescribed for five days’ use, twice daily.
On his return to the dermatologist, the patient reported that the site was no better. He was informed that the dermatologist and the pathologist, both with considerable experience, had detected no bacilli or dermal nerve infiltration in the tissue sample but had observed abundant lymphocytic infiltration. This finding, coupled with the patient’s decreased sensation, muscle fatigue, and status as a recent immigrant from a South American country, led to an initial diagnosis of borderline leprosy (BB).
Physical Examination and Disease Progression
If the first provider suspected a fungal infection during the initial examination, he should have completed a KOH scraping to confirm that suspicion. While superficial fungal infections are extremely common, particularly in the southeastern US, a sampling of scale with KOH 5% to 20% and the absence of dermatophytes under the microscope might have permitted a quick rule-out.12 Additionally, dermatophytic infections are very responsive to current medications within days, not weeks.
Thus, the key to an accurate diagnosis of leprosy begins with a physical examination that takes into account both chronic unresolved cutaneous lesions and neurologic changes in sensitivity.
A great majority of patients with HD have no noteworthy clinical symptoms until the first skin manifestations. While sensory neuropathy is prominent in all patients, additional findings (depending on the type of leprosy involved) may include plantar ulcers, chronic nasal congestion with early evidence of cartilage loss over the nasal bridge, chronic nosebleeds, cranial nerve palsies, and eye paralysis.12 In rare cases, hypogonadism may develop, or the clinical team may discover amyloidosis, an accumulation of insoluble proteins in the tissues that impairs function in various organs; this condition is usually detected by skin biopsy. If early or indeterminate leprosy is identified, neither form persists; rather, the condition will progress to one of the following:
• Borderline lepromatous leprosy (BL) and lepromatous leprosy (LL) offer very high resistance to treatment and are considered malignant forms of HD. Findings in patients with BL or LL include multiple lesions in various crazy-quilt presentations: macules, papules, plaques, nodules, masses, and patches of deep, erythematous infiltration across the extremities, face, and trunk.12 Loss of all hair except on the scalp is possible. Ulcers may form on the palms of the hands and soles of the feet.
In both BL and LL, results on AFB smears will reflect moderately high or very high concentrations of the pathogen in the patient’s system. Thus, these forms of HD are defined as multibacillary. If either is left untreated, the accumulation of proteins attributed to amyloidosis and secondary infections that develop eventually lead to death.
• Borderline leprosy (BB) has already been described in the case patient, with five or six ill-defined lesions and mild impairment of the peripheral nervous system. Aggressive treatment of this form of HD is important. A strong immune system provides no guarantee of successful treatment, and therapy must forestall a likely continuum to more serious forms.13
• Borderline tuberculoid leprosy (BT) and tuberculoid leprosy (TT) are both forms of paucibacillary HD. They offer little resistance to treatment in the patient with a healthy immune system. Lesions are generally scant (fewer than five; see figure), and the AFB smear reveals 0 to 10 bacteria in 100 fields.12
For the primary care provider, a thorough physical examination also requires familiarity with reasonable differentials. Hallmarks of conditions less likely to be leprosy include nausea, vomiting, or diarrhea, fever and chills, rectal bleeding and unexplained weight loss, headaches, and shortness of breath.
Diagnostic Testing
To help gauge progression of the disease and assess effectiveness of therapy, it is common practice to conduct a neurosensory test at each office visit. This is performed by touching the patient’s extremities and the periphery of lesions with a simple nylon monofilament.13 The patient’s response or lack of response is documented and changes are noted for future therapeutic decision making.
In patients with suspected HD, the NHDP recommends that a full-thickness 4.0-mm elliptical or punch biopsy be performed at the periphery of the largest or most active lesion and that slides be sent to the NHDP in formalin or paraffin for review.14 Experienced NHDP pathologists may order additional stains. A detailed protocol can be found at www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm.
Proficiency in performing slit smears, a technique to obtain tissue fluid, is also helpful.15 Measurement of the number of bacteria in a set number of fields allows for calculation of the host’s bacterial index, making it possible to determine an appropriate treatment level.
The preferred sites for the slit smear are active lesions, if possible from both ear lobes plus two additional locations. After alcohol swabbing, a tight, bloodless pinch is made, with a 3.0-mm incision that yields blood and fluid. The resulting culture is examined for AFB, which are found abundantly in lepromatous leprosy (whereas tuberculoid leprosy, by definition, should yield none or few). This smear may be repeated at three- to six-month intervals to determine effectiveness of therapy. It is considered superior to a nasal culture, which often yields false-negative results.16
After the Diagnosis
In the US, HD is a nationally notifiable disease: Health care providers are required by law to inform the CDC of the known particulars while complying with HIPAA requirements.17
That the case patient was a recent immigrant from a country with a high incidence of HD, coupled with a loss of sensation in his extremities, led to the correct diagnosis. His lack of response to antifungals or anti-inflammatory agents, though frustrating to both patient and provider, was helpful in excluding two common conditions in the differential diagnosis. Other possibilites that were considered ultimately did not ring true:
Vitiligo, a loss of pigment that produces white patches resembling borderline leprosy but lacks the raised edging18;
Granuloma annulare, a lightly raised, ring-shaped lesion that is usually found on the extremities19; and
Lupus erythematosus, in which patients almost universally experience pruritus and burning, whereas lack of sensation is key to a diagnosis of leprosy.20
Treatment and Management
Health care providers must appear confident to instill confidence in their patients. Any provider with the knowledge and acuity to make a diagnosis of leprosy should be adequately equipped to manage the patient’s care. However, the hesitant or uncertain provider should consider referral to dermatology or to infectious disease for continued management.
The NHDP recognizes the WHO classification of leprosy as multibacillary or paucibacillary (observing a “simple clinical rule” of six or more lesions representing multibacillary HD and five or fewer, paucibacillary HD1), with additional consideration to the three major disease subtypes mentioned earlier: TT, which is treatable; BB, which is considered unstable; and LL, which is resistant to treatment. NHDP and WHO recommendations for treatment differ slightly but address the same goals: complete elimination of the pathogen, preservation of muscle and nerve function, prevention of secondary infections, and management of the adverse effects of medication.1,21 Adhering to universal precautions is important.
In the US, the multidrug treatment regimen for paucibacillary (TT or BT) leprosy comprises one year of oral rifampin 600 mg/d and oral dapsone 100 mg/d21; WHO, in deference to cost containment in developing countries, recommends a six-month regimen of these agents (ie, rifampin 600 mg/mo and dapsone 100 mg/d).1 A third drug sometimes given to allay active neuritis is oral clofazimine 50 mg/d for one year21 (per WHO recommendations, a monthly dose of 300 mg, plus 50 mg/d1). In patients with preexisting or subsequent anemia, dapsone dosing should be lowered to 50 mg, the minimally effective level. Of note, clofazimine is no longer commercially available in the US and is held by the NHDP as an investigational new drug for treatment of US patients.21
According to US recommendations,21 use of this three-agent regimen is extended to two years for patients with multibacillary (LL, BL, BB) leprosy. For these patients, WHO recommends its regimen for 12 months.1
Rifampin is a powerful antibiotic used for effective treatment of tuberculosis, methicillin-resistant Staphylococcus aureus, gonorrhea, Listeria, and Haemophilus influenzae. However, its use has been shown to reduce the efficacy of anticoagulants, oral contraceptives, and prednisone, possibly requiring therapeutic adjustments.22 Recommended alternatives for rifampin include (in the order of preference) minocycline 100 mg/d, ofloxacin 400 mg/d, levofloxacin 500 mg/d, or twice-daily clarithromycin 500 mg.13
Patient compliance with the regimen is facilitated by convenient blister packs that contain standard weekly and monthly doses of these medications, all clearly marked. Many local health departments make these medications available at no cost to patients.
Patient Education
Informing patients what to expect regarding their medications, necessary lab work, and avoiding transmission of HD can ease worry, protect others from illness, and increase the likelihood of an excellent prognosis. Patients with lepromatous (multibacillary) HD should be urged to limit their interactions with others at work or school (particularly preventing exposure to their nasal excretions) until a low bacterial index is achieved. For close contacts of patients recently diagnosed with HD, single-dose rifampin has been shown to provide effective prevention for two years.23
Emphasizing adherence to daily multidrug therapy minimizes the chance that bacterial resistance will develop. Patients should be given complete information about the individual agents used:
Rifampin 600 mg is the maximum daily dose for an adult. Hepatotoxicity is a concern, and liver enzyme levels (ie, aspartate aminotransferase, alanine aminotransferase) should be measured at three-month intervals. It is important to obtain a history of previous liver disease, exposure to hepatitis, and drug and alcohol use. In otherwise healthy adults, rifampin has the curious effect of turning bodily fluids such as urine and tears bright red. Forewarning patients of this benign development may prevent anxious reactions. Contact lenses may be irreversibly stained.24
Dapsone, for many years the first-line monotherapy for leprosy, is a bacteriostatic, sulfa-based antibiotic that only prevents the multiplication of bacteria, but it is still worthy of respect. Before dapsone is initiated, a screening test for glucose-6-phosphate dehydrogenase deficiency should be performed to determine the risk for dapsone-associated hemolysis.24 Dapsone use is contraindicated in women who are breastfeeding. In patients with severe heart conditions, dapsone can reduce oxygen flow, as evidenced by a bluish discoloration of the lips and fingertips. Mild anemia, headache, gastrointestinal upset and nausea are all common adverse effects.
Clofazimine, a soft gelatin capsule, may cause gray-blue or red hyperpigmentation in individuals who are light-skinned; this effect reverses once the drug is discontinued. Nausea, vomiting, and abdominal pain have been reported. This slow-release bactericidal drug has an extremely long half-life of 70 days. It provides anti-inflammatory action similar to that of prednisone without the latter agent’s long-term adverse effects.24
The Patient’s Outcome
The case patient was the second with HD seen in this dermatology practice. A man who presented on an earlier occasion, possibly an undocumented worker in fear of deportation, seemed concerned about the reporting process, even though he was assured that no personal information would be released. He did not return for follow-up, and attempts to reach him proved fruitless.
The case patient, by contrast, was pleasant and cooperative. He appeared to understand his condition, the required treatment and its possible adverse effects, and the need for continued lab work and regular follow-up appointments. A day laborer in a large factory, he was particularly careful to avoid transmission of his illness to his family or co-workers.
Within two months of beginning multidrug therapy, he experienced quick clearance of the lesions on his back. His bacterial index approached 0 by the time he underwent the second smear. Although he complained of loose stools and classic orange-red bodily fluid throughout the entire year of treatment, he resisted the temptation to discontinue the regimen.
He has experienced no relapses in six years and is considered cured.
Conclusion
Only one in every 20 people is susceptible to infection with M leprae, but cases of leprosy continue to be reported in the US, and illness is of particular concern among immunocompromised individuals. Treatment delay is common because of the condition’s relative rarity in the US, but therapy with standard antibiotics is extremely effective. Lengthy isolation from family and community is not often needed, and relapses are rare. Fully restored health is the rule rather than the exception for patients treated for Hansen’s disease.
According to data from the World Health Organization (WHO), the number of new cases of Hansen’s disease (HD, or leprosy) identified worldwide declined by about 20% from 2003 to 2004. At least 116 of 122 countries once considered leprosy—endemic have achieved a prevalence rate of less than one case per 10,000 population.1,2
Meanwhile, the number of active HD infections in the United States catalogued through the CDC and the National Hansen’s Disease Program (NHDP) rose from 76 cases in 2000 to 166 in 2005, with an additional 137 cases in 2006 (the most recent year for which complete figures are available).3,4 Currently, 6,500 US residents are known to have HD, of whom 3,300 require care for active disease.3
Accurate diagnosis and straightforward, aggressive treatment can mean certain cure for this dreaded disease, as it is responsive to a combination of available drugs. For primary care providers in the US, familiarity with this condition can play a role in eliminating it altogether.
Background and Etiology
HD is believed to have originated in East Africa with the pseudonym of leprosy, from the Greek lepi, meaning “fish scales.” Although HD is one of the oldest known human infections, it is not well understood by many Western health care providers. Once considered highly contagious and easily transmissible, HD has proven to be neither: 95% of the human population is not susceptible to the responsible pathogen.3 HD retains its reputation as a devastating illness when left untreated, however, with sequelae often including significant sensory and motor dysfunction.5
Leprosy is a chronic bacterial disease caused by Mycobacterium leprae. Mycobacterium is the same rod-shaped, acid-fast bacillus (AFB) implicated in pulmonary tuberculosis, cutaneous tuberculosis, and numerous cutaneous nontuberculoid infections.6,7
HD transmission is believed to occur almost exclusively by nasal droplet, inhaled by way of others’ nares or lungs. Research continues in possible transmission by skin-to-skin contact.8,9
Leprosy is particularly challenging for immunocompromised persons, such as those with HIV/AIDS or tuberculosis. According to data from the NHDP, HD in the US is found primarily among undocumented immigrants who live in close quarters. Cases considered “home-grown” rather than “imported”5 are most commonly diagnosed in Hawaii and in a swath from Texas to Georgia and south toward Florida. Pockets of HD incidence are also found in California, New York, and Massachusetts.4
Clinicians’ lack of familiarity with HD and its relative rarity in the US help explain why diagnosis and treatment are commonly delayed. As a patient’s disease progresses from an insidious, gradual, and relatively painless onset, the primary care provider may be grappling with an extensive differential diagnosis (see “Differential Diagnosis for Hansen’s Disease [Leprosy],”10,11 below). Even after confirming a diagnosis of HD, the provider must contend with a lack of information and misinformation about HD among staff members, the patient’s family, and society at large, as well as the associated stigma. Patient education and effective treatment are of comparable importance.
Patient Presentation and History
A 40-year-old Latino man presented to a primary care provider with a well-demarcated, erythematous patch on his right upper back that had raised outer edging and faint flaking. The patch was surrounded by several smaller but similar asymmetrical macules. The patient denied pruritus but stated that the area “felt funny.” He said the lesion might have been spreading during the previous several months, but he could not be certain because of its location.
The man denied any medication use except for a topical antibiotic ointment he had applied to the affected area with no improvement. His provider made a diagnosis of tinea corporis and prescribed an antifungal cream, naftifine. After two weeks without results, the patient was referred to a dermatologist.
During the assessment, it became apparent that the patient was experiencing numbness in his fingertips and general muscle weakness in his arms. A KOH prep was difficult to obtain, as the lesion site was somewhat smooth. Any dermatophytes that may have been present had been eliminated by the naftifine, and KOH results were negative.
A 5.0-mm punch biopsy from the largest lesion was ordered. A preliminary diagnosis of sarcoidosis (chronic inflammation with an unusual skin manifestation) was made, with serious consideration given to early cutaneous T-cell lymphoma (mycosis fungoides). A high-potency steroid was prescribed for five days’ use, twice daily.
On his return to the dermatologist, the patient reported that the site was no better. He was informed that the dermatologist and the pathologist, both with considerable experience, had detected no bacilli or dermal nerve infiltration in the tissue sample but had observed abundant lymphocytic infiltration. This finding, coupled with the patient’s decreased sensation, muscle fatigue, and status as a recent immigrant from a South American country, led to an initial diagnosis of borderline leprosy (BB).
Physical Examination and Disease Progression
If the first provider suspected a fungal infection during the initial examination, he should have completed a KOH scraping to confirm that suspicion. While superficial fungal infections are extremely common, particularly in the southeastern US, a sampling of scale with KOH 5% to 20% and the absence of dermatophytes under the microscope might have permitted a quick rule-out.12 Additionally, dermatophytic infections are very responsive to current medications within days, not weeks.
Thus, the key to an accurate diagnosis of leprosy begins with a physical examination that takes into account both chronic unresolved cutaneous lesions and neurologic changes in sensitivity.
A great majority of patients with HD have no noteworthy clinical symptoms until the first skin manifestations. While sensory neuropathy is prominent in all patients, additional findings (depending on the type of leprosy involved) may include plantar ulcers, chronic nasal congestion with early evidence of cartilage loss over the nasal bridge, chronic nosebleeds, cranial nerve palsies, and eye paralysis.12 In rare cases, hypogonadism may develop, or the clinical team may discover amyloidosis, an accumulation of insoluble proteins in the tissues that impairs function in various organs; this condition is usually detected by skin biopsy. If early or indeterminate leprosy is identified, neither form persists; rather, the condition will progress to one of the following:
• Borderline lepromatous leprosy (BL) and lepromatous leprosy (LL) offer very high resistance to treatment and are considered malignant forms of HD. Findings in patients with BL or LL include multiple lesions in various crazy-quilt presentations: macules, papules, plaques, nodules, masses, and patches of deep, erythematous infiltration across the extremities, face, and trunk.12 Loss of all hair except on the scalp is possible. Ulcers may form on the palms of the hands and soles of the feet.
In both BL and LL, results on AFB smears will reflect moderately high or very high concentrations of the pathogen in the patient’s system. Thus, these forms of HD are defined as multibacillary. If either is left untreated, the accumulation of proteins attributed to amyloidosis and secondary infections that develop eventually lead to death.
• Borderline leprosy (BB) has already been described in the case patient, with five or six ill-defined lesions and mild impairment of the peripheral nervous system. Aggressive treatment of this form of HD is important. A strong immune system provides no guarantee of successful treatment, and therapy must forestall a likely continuum to more serious forms.13
• Borderline tuberculoid leprosy (BT) and tuberculoid leprosy (TT) are both forms of paucibacillary HD. They offer little resistance to treatment in the patient with a healthy immune system. Lesions are generally scant (fewer than five; see figure), and the AFB smear reveals 0 to 10 bacteria in 100 fields.12
For the primary care provider, a thorough physical examination also requires familiarity with reasonable differentials. Hallmarks of conditions less likely to be leprosy include nausea, vomiting, or diarrhea, fever and chills, rectal bleeding and unexplained weight loss, headaches, and shortness of breath.
Diagnostic Testing
To help gauge progression of the disease and assess effectiveness of therapy, it is common practice to conduct a neurosensory test at each office visit. This is performed by touching the patient’s extremities and the periphery of lesions with a simple nylon monofilament.13 The patient’s response or lack of response is documented and changes are noted for future therapeutic decision making.
In patients with suspected HD, the NHDP recommends that a full-thickness 4.0-mm elliptical or punch biopsy be performed at the periphery of the largest or most active lesion and that slides be sent to the NHDP in formalin or paraffin for review.14 Experienced NHDP pathologists may order additional stains. A detailed protocol can be found at www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm.
Proficiency in performing slit smears, a technique to obtain tissue fluid, is also helpful.15 Measurement of the number of bacteria in a set number of fields allows for calculation of the host’s bacterial index, making it possible to determine an appropriate treatment level.
The preferred sites for the slit smear are active lesions, if possible from both ear lobes plus two additional locations. After alcohol swabbing, a tight, bloodless pinch is made, with a 3.0-mm incision that yields blood and fluid. The resulting culture is examined for AFB, which are found abundantly in lepromatous leprosy (whereas tuberculoid leprosy, by definition, should yield none or few). This smear may be repeated at three- to six-month intervals to determine effectiveness of therapy. It is considered superior to a nasal culture, which often yields false-negative results.16
After the Diagnosis
In the US, HD is a nationally notifiable disease: Health care providers are required by law to inform the CDC of the known particulars while complying with HIPAA requirements.17
That the case patient was a recent immigrant from a country with a high incidence of HD, coupled with a loss of sensation in his extremities, led to the correct diagnosis. His lack of response to antifungals or anti-inflammatory agents, though frustrating to both patient and provider, was helpful in excluding two common conditions in the differential diagnosis. Other possibilites that were considered ultimately did not ring true:
Vitiligo, a loss of pigment that produces white patches resembling borderline leprosy but lacks the raised edging18;
Granuloma annulare, a lightly raised, ring-shaped lesion that is usually found on the extremities19; and
Lupus erythematosus, in which patients almost universally experience pruritus and burning, whereas lack of sensation is key to a diagnosis of leprosy.20
Treatment and Management
Health care providers must appear confident to instill confidence in their patients. Any provider with the knowledge and acuity to make a diagnosis of leprosy should be adequately equipped to manage the patient’s care. However, the hesitant or uncertain provider should consider referral to dermatology or to infectious disease for continued management.
The NHDP recognizes the WHO classification of leprosy as multibacillary or paucibacillary (observing a “simple clinical rule” of six or more lesions representing multibacillary HD and five or fewer, paucibacillary HD1), with additional consideration to the three major disease subtypes mentioned earlier: TT, which is treatable; BB, which is considered unstable; and LL, which is resistant to treatment. NHDP and WHO recommendations for treatment differ slightly but address the same goals: complete elimination of the pathogen, preservation of muscle and nerve function, prevention of secondary infections, and management of the adverse effects of medication.1,21 Adhering to universal precautions is important.
In the US, the multidrug treatment regimen for paucibacillary (TT or BT) leprosy comprises one year of oral rifampin 600 mg/d and oral dapsone 100 mg/d21; WHO, in deference to cost containment in developing countries, recommends a six-month regimen of these agents (ie, rifampin 600 mg/mo and dapsone 100 mg/d).1 A third drug sometimes given to allay active neuritis is oral clofazimine 50 mg/d for one year21 (per WHO recommendations, a monthly dose of 300 mg, plus 50 mg/d1). In patients with preexisting or subsequent anemia, dapsone dosing should be lowered to 50 mg, the minimally effective level. Of note, clofazimine is no longer commercially available in the US and is held by the NHDP as an investigational new drug for treatment of US patients.21
According to US recommendations,21 use of this three-agent regimen is extended to two years for patients with multibacillary (LL, BL, BB) leprosy. For these patients, WHO recommends its regimen for 12 months.1
Rifampin is a powerful antibiotic used for effective treatment of tuberculosis, methicillin-resistant Staphylococcus aureus, gonorrhea, Listeria, and Haemophilus influenzae. However, its use has been shown to reduce the efficacy of anticoagulants, oral contraceptives, and prednisone, possibly requiring therapeutic adjustments.22 Recommended alternatives for rifampin include (in the order of preference) minocycline 100 mg/d, ofloxacin 400 mg/d, levofloxacin 500 mg/d, or twice-daily clarithromycin 500 mg.13
Patient compliance with the regimen is facilitated by convenient blister packs that contain standard weekly and monthly doses of these medications, all clearly marked. Many local health departments make these medications available at no cost to patients.
Patient Education
Informing patients what to expect regarding their medications, necessary lab work, and avoiding transmission of HD can ease worry, protect others from illness, and increase the likelihood of an excellent prognosis. Patients with lepromatous (multibacillary) HD should be urged to limit their interactions with others at work or school (particularly preventing exposure to their nasal excretions) until a low bacterial index is achieved. For close contacts of patients recently diagnosed with HD, single-dose rifampin has been shown to provide effective prevention for two years.23
Emphasizing adherence to daily multidrug therapy minimizes the chance that bacterial resistance will develop. Patients should be given complete information about the individual agents used:
Rifampin 600 mg is the maximum daily dose for an adult. Hepatotoxicity is a concern, and liver enzyme levels (ie, aspartate aminotransferase, alanine aminotransferase) should be measured at three-month intervals. It is important to obtain a history of previous liver disease, exposure to hepatitis, and drug and alcohol use. In otherwise healthy adults, rifampin has the curious effect of turning bodily fluids such as urine and tears bright red. Forewarning patients of this benign development may prevent anxious reactions. Contact lenses may be irreversibly stained.24
Dapsone, for many years the first-line monotherapy for leprosy, is a bacteriostatic, sulfa-based antibiotic that only prevents the multiplication of bacteria, but it is still worthy of respect. Before dapsone is initiated, a screening test for glucose-6-phosphate dehydrogenase deficiency should be performed to determine the risk for dapsone-associated hemolysis.24 Dapsone use is contraindicated in women who are breastfeeding. In patients with severe heart conditions, dapsone can reduce oxygen flow, as evidenced by a bluish discoloration of the lips and fingertips. Mild anemia, headache, gastrointestinal upset and nausea are all common adverse effects.
Clofazimine, a soft gelatin capsule, may cause gray-blue or red hyperpigmentation in individuals who are light-skinned; this effect reverses once the drug is discontinued. Nausea, vomiting, and abdominal pain have been reported. This slow-release bactericidal drug has an extremely long half-life of 70 days. It provides anti-inflammatory action similar to that of prednisone without the latter agent’s long-term adverse effects.24
The Patient’s Outcome
The case patient was the second with HD seen in this dermatology practice. A man who presented on an earlier occasion, possibly an undocumented worker in fear of deportation, seemed concerned about the reporting process, even though he was assured that no personal information would be released. He did not return for follow-up, and attempts to reach him proved fruitless.
The case patient, by contrast, was pleasant and cooperative. He appeared to understand his condition, the required treatment and its possible adverse effects, and the need for continued lab work and regular follow-up appointments. A day laborer in a large factory, he was particularly careful to avoid transmission of his illness to his family or co-workers.
Within two months of beginning multidrug therapy, he experienced quick clearance of the lesions on his back. His bacterial index approached 0 by the time he underwent the second smear. Although he complained of loose stools and classic orange-red bodily fluid throughout the entire year of treatment, he resisted the temptation to discontinue the regimen.
He has experienced no relapses in six years and is considered cured.
Conclusion
Only one in every 20 people is susceptible to infection with M leprae, but cases of leprosy continue to be reported in the US, and illness is of particular concern among immunocompromised individuals. Treatment delay is common because of the condition’s relative rarity in the US, but therapy with standard antibiotics is extremely effective. Lengthy isolation from family and community is not often needed, and relapses are rare. Fully restored health is the rule rather than the exception for patients treated for Hansen’s disease.
1. World Health Organization. Global strategy for further reducing the leprosy burden and sustaining leprosy control activities 2006-2010: operational guidelines. www.who.int/lep/resources/SEAGLP20062.pdf. Accessed November 17, 2009.
2. World Health Organization Communicable -Diseases Department. Leprosy: frequently as-ked questions. www.searo.who.int/EN/Section10/Section373_11716.htm. Accessed November 17, 2009.
3. National Hansen’s Disease (Leprosy) Program. www.hrsa.gov/hansens. Accessed November 23, 2009.
4. Health Resources and Services Administration, US Department of Health and Human Services, National Hansen’s Disease Program. A summary of Hansen’s disease in the United States—2006. ftp://ftp.hrsa.gov/hansens/2006RegistryReport.pdf. Accessed November 17, 2009.
5. Boggild AK, Correia JD, Keystone JS, Kain KC. Leprosy in Toronto: an analysis of 184 imported cases. CMAJ. 2004;170(1):55-59.
6. Cheesbrough M. Bacterial pathogens. In: District Laboratory Practice in Tropical Countries. Part 2. 2nd ed. Cambridge, UK: Cambridge University Press. 2006;157-234.
7. Sasaki S, Takeshita F, Okuda K, Ishii N. Mycobacterium leprae and leprosy: a compendium. Microbiol Immunol. 2001;45(11):729–736.
8. Job CK, Jayakumar J, Kearney M, Gillis TP. Transmission of leprosy: a study of skin and nasal secretions of household contacts of leprosy patients using PCR. Am J Trop Med Hyg. 2008; 78(3):518-521.
9. Girdhar BK. Skin to skin transmission of leprosy. Indian J Dermatol Venereol Leprol. 2005;71(4):223-225.
10. National Leprosy Eradication Programme, Directorate General of Health Services (Government of India). Differential diagnosis for leprosy (2009). http://nlep.nic.in/pdf/Annex-%20III% 20Differential%20diagnosis.pdf. September 14, 2009.
11. Kim EC. Hansen disease: differential diagnoses and workup (2006). http://emedicine.medscape .com/article/1213853-diagnosis. Accessed November 17, 2009.
12. James WD, Berger TG, Elston DM. Hansen’s disease. In: Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Philadelphia, PA: Saunders. 2005;343-366.
13. Leprosy. In: Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 2nd ed. Philadelphia, PA: WB Saunders; 2005.
14. National Hansen’s Disease Program. Skin biopsy in the diagnosis of Hansen’s disease. www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm. Accessed November 17, 2009.
15. Bhushan P, Sardana K, Koranne RV, et al. Diagnosing multibacillary leprosy: a comparative evaluation of diagnostic accuracy of slit-skin smear, bacterial index of granuloma and WHO operational classification. Indian J Dermatol Venereol Leprol. 2008;74(4):322-326.
16. Wolff K, Johnson R, Suurmond R. Leprosy. In: Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. Chicago, IL: McGraw-Hill Professional; 2005:655-661.
17. Centers for Disease Control and Prevention. Summary of notifiable diseases—United States, 2007. MMWR Morb Mortal Wkly Rep. 2009;56 (53):3, 10, 22.
18. American Academy of Dermatology. Vitiligo. www.aad.org/public/publications/pamphlets/com mon_vitilgo.html. Accessed November 17, 2009.
19. American Academy of Dermatology. Granuloma annulare. www.aad.org/public/publica tions/pamphlets/common_granuloma.html. Accessed November 17, 2009.
20. American Academy of Dermatology. Lupus and the skin. www.aad.org/public/publications/pamphlets/common_lupus.html. Accessed November 17, 2009.
21. National Hansen’s Disease Program. Recommended treatment regimens. www.hrsa.gov/han sens/clinical/regimens.htm. Accessed November 17, 2009.
22. Chen J, Raymond K. Roles of rifampicin in drug-drug interactions: underlying molecular mechanisms involving the nuclear pregnane X receptor. Ann Clin Microbiol Antimicrob. 2006 Feb 15;5:3.
23. Moet FJ, Pahan D, Oskam L, Richardus JH; COLEP Study Group. Effectiveness of single dose rifampicin in preventing leprosy in close contacts of patients with newly diagnosed leprosy: cluster randomised controlled trial. BMJ. 2008;336 (7647):761-764.
24. World Health Organization. WHO model prescribing information: drugs used in leprosy. http://apps.who.int/medicinedocs/fr/d/Jh2988e/ 14.2.html. Accessed November 17, 2009.
25. Bruce S, Schroeder TL, Ellner K, et al. Armadillo exposure and Hansen’s disease: an epidemiologic survey in southern Texas. J Am Acad Dermatol. 2000;43(2 pt 1):223-228.
26. Clark BM, Murray CK, Horvath LL, et al. Case-control study of armadillo contact and Hansen’s disease. Am J Trop Med Hyg. 2008;78(6):962-967.
1. World Health Organization. Global strategy for further reducing the leprosy burden and sustaining leprosy control activities 2006-2010: operational guidelines. www.who.int/lep/resources/SEAGLP20062.pdf. Accessed November 17, 2009.
2. World Health Organization Communicable -Diseases Department. Leprosy: frequently as-ked questions. www.searo.who.int/EN/Section10/Section373_11716.htm. Accessed November 17, 2009.
3. National Hansen’s Disease (Leprosy) Program. www.hrsa.gov/hansens. Accessed November 23, 2009.
4. Health Resources and Services Administration, US Department of Health and Human Services, National Hansen’s Disease Program. A summary of Hansen’s disease in the United States—2006. ftp://ftp.hrsa.gov/hansens/2006RegistryReport.pdf. Accessed November 17, 2009.
5. Boggild AK, Correia JD, Keystone JS, Kain KC. Leprosy in Toronto: an analysis of 184 imported cases. CMAJ. 2004;170(1):55-59.
6. Cheesbrough M. Bacterial pathogens. In: District Laboratory Practice in Tropical Countries. Part 2. 2nd ed. Cambridge, UK: Cambridge University Press. 2006;157-234.
7. Sasaki S, Takeshita F, Okuda K, Ishii N. Mycobacterium leprae and leprosy: a compendium. Microbiol Immunol. 2001;45(11):729–736.
8. Job CK, Jayakumar J, Kearney M, Gillis TP. Transmission of leprosy: a study of skin and nasal secretions of household contacts of leprosy patients using PCR. Am J Trop Med Hyg. 2008; 78(3):518-521.
9. Girdhar BK. Skin to skin transmission of leprosy. Indian J Dermatol Venereol Leprol. 2005;71(4):223-225.
10. National Leprosy Eradication Programme, Directorate General of Health Services (Government of India). Differential diagnosis for leprosy (2009). http://nlep.nic.in/pdf/Annex-%20III% 20Differential%20diagnosis.pdf. September 14, 2009.
11. Kim EC. Hansen disease: differential diagnoses and workup (2006). http://emedicine.medscape .com/article/1213853-diagnosis. Accessed November 17, 2009.
12. James WD, Berger TG, Elston DM. Hansen’s disease. In: Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Philadelphia, PA: Saunders. 2005;343-366.
13. Leprosy. In: Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 2nd ed. Philadelphia, PA: WB Saunders; 2005.
14. National Hansen’s Disease Program. Skin biopsy in the diagnosis of Hansen’s disease. www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm. Accessed November 17, 2009.
15. Bhushan P, Sardana K, Koranne RV, et al. Diagnosing multibacillary leprosy: a comparative evaluation of diagnostic accuracy of slit-skin smear, bacterial index of granuloma and WHO operational classification. Indian J Dermatol Venereol Leprol. 2008;74(4):322-326.
16. Wolff K, Johnson R, Suurmond R. Leprosy. In: Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. Chicago, IL: McGraw-Hill Professional; 2005:655-661.
17. Centers for Disease Control and Prevention. Summary of notifiable diseases—United States, 2007. MMWR Morb Mortal Wkly Rep. 2009;56 (53):3, 10, 22.
18. American Academy of Dermatology. Vitiligo. www.aad.org/public/publications/pamphlets/com mon_vitilgo.html. Accessed November 17, 2009.
19. American Academy of Dermatology. Granuloma annulare. www.aad.org/public/publica tions/pamphlets/common_granuloma.html. Accessed November 17, 2009.
20. American Academy of Dermatology. Lupus and the skin. www.aad.org/public/publications/pamphlets/common_lupus.html. Accessed November 17, 2009.
21. National Hansen’s Disease Program. Recommended treatment regimens. www.hrsa.gov/han sens/clinical/regimens.htm. Accessed November 17, 2009.
22. Chen J, Raymond K. Roles of rifampicin in drug-drug interactions: underlying molecular mechanisms involving the nuclear pregnane X receptor. Ann Clin Microbiol Antimicrob. 2006 Feb 15;5:3.
23. Moet FJ, Pahan D, Oskam L, Richardus JH; COLEP Study Group. Effectiveness of single dose rifampicin in preventing leprosy in close contacts of patients with newly diagnosed leprosy: cluster randomised controlled trial. BMJ. 2008;336 (7647):761-764.
24. World Health Organization. WHO model prescribing information: drugs used in leprosy. http://apps.who.int/medicinedocs/fr/d/Jh2988e/ 14.2.html. Accessed November 17, 2009.
25. Bruce S, Schroeder TL, Ellner K, et al. Armadillo exposure and Hansen’s disease: an epidemiologic survey in southern Texas. J Am Acad Dermatol. 2000;43(2 pt 1):223-228.
26. Clark BM, Murray CK, Horvath LL, et al. Case-control study of armadillo contact and Hansen’s disease. Am J Trop Med Hyg. 2008;78(6):962-967.
Diagnosing and Treating Hansen's Disease
According to data from the World Health Organization (WHO), the number of new cases of Hansen’s disease (HD, or leprosy) identified worldwide declined by about 20% from 2003 to 2004. At least 116 of 122 countries once considered leprosy—endemic have achieved a prevalence rate of less than one case per 10,000 population.1,2
Meanwhile, the number of active HD infections in the United States catalogued through the CDC and the National Hansen’s Disease Program (NHDP) rose from 76 cases in 2000 to 166 in 2005, with an additional 137 cases in 2006 (the most recent year for which complete figures are available).3,4 Currently, 6,500 US residents are known to have HD, of whom 3,300 require care for active disease.3
Accurate diagnosis and straightforward, aggressive treatment can mean certain cure for this dreaded disease, as it is responsive to a combination of available drugs. For primary care providers in the US, familiarity with this condition can play a role in eliminating it altogether.
Background and Etiology
HD is believed to have originated in East Africa with the pseudonym of leprosy, from the Greek lepi, meaning “fish scales.” Although HD is one of the oldest known human infections, it is not well understood by many Western health care providers. Once considered highly contagious and easily transmissible, HD has proven to be neither: 95% of the human population is not susceptible to the responsible pathogen.3 HD retains its reputation as a devastating illness when left untreated, however, with sequelae often including significant sensory and motor dysfunction.5
Leprosy is a chronic bacterial disease caused by Mycobacterium leprae. Mycobacterium is the same rod-shaped, acid-fast bacillus (AFB) implicated in pulmonary tuberculosis, cutaneous tuberculosis, and numerous cutaneous nontuberculoid infections.6,7
HD transmission is believed to occur almost exclusively by nasal droplet, inhaled by way of others’ nares or lungs. Research continues in possible transmission by skin-to-skin contact.8,9
Leprosy is particularly challenging for immunocompromised persons, such as those with HIV/AIDS or tuberculosis. According to data from the NHDP, HD in the US is found primarily among undocumented immigrants who live in close quarters. Cases considered “home-grown” rather than “imported”5 are most commonly diagnosed in Hawaii and in a swath from Texas to Georgia and south toward Florida. Pockets of HD incidence are also found in California, New York, and Massachusetts.4
Clinicians’ lack of familiarity with HD and its relative rarity in the US help explain why diagnosis and treatment are commonly delayed. As a patient’s disease progresses from an insidious, gradual, and relatively painless onset, the primary care provider may be grappling with an extensive differential diagnosis (see “Differential Diagnosis for Hansen’s Disease [Leprosy],”10,11 below). Even after confirming a diagnosis of HD, the provider must contend with a lack of information and misinformation about HD among staff members, the patient’s family, and society at large, as well as the associated stigma. Patient education and effective treatment are of comparable importance.
Patient Presentation and History
A 40-year-old Latino man presented to a primary care provider with a well-demarcated, erythematous patch on his right upper back that had raised outer edging and faint flaking. The patch was surrounded by several smaller but similar asymmetrical macules. The patient denied pruritus but stated that the area “felt funny.” He said the lesion might have been spreading during the previous several months, but he could not be certain because of its location.
The man denied any medication use except for a topical antibiotic ointment he had applied to the affected area with no improvement. His provider made a diagnosis of tinea corporis and prescribed an antifungal cream, naftifine. After two weeks without results, the patient was referred to a dermatologist.
During the assessment, it became apparent that the patient was experiencing numbness in his fingertips and general muscle weakness in his arms. A KOH prep was difficult to obtain, as the lesion site was somewhat smooth. Any dermatophytes that may have been present had been eliminated by the naftifine, and KOH results were negative.
A 5.0-mm punch biopsy from the largest lesion was ordered. A preliminary diagnosis of sarcoidosis (chronic inflammation with an unusual skin manifestation) was made, with serious consideration given to early cutaneous T-cell lymphoma (mycosis fungoides). A high-potency steroid was prescribed for five days’ use, twice daily.
On his return to the dermatologist, the patient reported that the site was no better. He was informed that the dermatologist and the pathologist, both with considerable experience, had detected no bacilli or dermal nerve infiltration in the tissue sample but had observed abundant lymphocytic infiltration. This finding, coupled with the patient’s decreased sensation, muscle fatigue, and status as a recent immigrant from a South American country, led to an initial diagnosis of borderline leprosy (BB).
Physical Examination and Disease Progression
If the first provider suspected a fungal infection during the initial examination, he should have completed a KOH scraping to confirm that suspicion. While superficial fungal infections are extremely common, particularly in the southeastern US, a sampling of scale with KOH 5% to 20% and the absence of dermatophytes under the microscope might have permitted a quick rule-out.12 Additionally, dermatophytic infections are very responsive to current medications within days, not weeks.
Thus, the key to an accurate diagnosis of leprosy begins with a physical examination that takes into account both chronic unresolved cutaneous lesions and neurologic changes in sensitivity.
A great majority of patients with HD have no noteworthy clinical symptoms until the first skin manifestations. While sensory neuropathy is prominent in all patients, additional findings (depending on the type of leprosy involved) may include plantar ulcers, chronic nasal congestion with early evidence of cartilage loss over the nasal bridge, chronic nosebleeds, cranial nerve palsies, and eye paralysis.12 In rare cases, hypogonadism may develop, or the clinical team may discover amyloidosis, an accumulation of insoluble proteins in the tissues that impairs function in various organs; this condition is usually detected by skin biopsy. If early or indeterminate leprosy is identified, neither form persists; rather, the condition will progress to one of the following:
• Borderline lepromatous leprosy (BL) and lepromatous leprosy (LL) offer very high resistance to treatment and are considered malignant forms of HD. Findings in patients with BL or LL include multiple lesions in various crazy-quilt presentations: macules, papules, plaques, nodules, masses, and patches of deep, erythematous infiltration across the extremities, face, and trunk.12 Loss of all hair except on the scalp is possible. Ulcers may form on the palms of the hands and soles of the feet.
In both BL and LL, results on AFB smears will reflect moderately high or very high concentrations of the pathogen in the patient’s system. Thus, these forms of HD are defined as multibacillary. If either is left untreated, the accumulation of proteins attributed to amyloidosis and secondary infections that develop eventually lead to death.
• Borderline leprosy (BB) has already been described in the case patient, with five or six ill-defined lesions and mild impairment of the peripheral nervous system. Aggressive treatment of this form of HD is important. A strong immune system provides no guarantee of successful treatment, and therapy must forestall a likely continuum to more serious forms.13
• Borderline tuberculoid leprosy (BT) and tuberculoid leprosy (TT) are both forms of paucibacillary HD. They offer little resistance to treatment in the patient with a healthy immune system. Lesions are generally scant (fewer than five; see figure), and the AFB smear reveals 0 to 10 bacteria in 100 fields.12
For the primary care provider, a thorough physical examination also requires familiarity with reasonable differentials. Hallmarks of conditions less likely to be leprosy include nausea, vomiting, or diarrhea, fever and chills, rectal bleeding and unexplained weight loss, headaches, and shortness of breath.
Diagnostic Testing
To help gauge progression of the disease and assess effectiveness of therapy, it is common practice to conduct a neurosensory test at each office visit. This is performed by touching the patient’s extremities and the periphery of lesions with a simple nylon monofilament.13 The patient’s response or lack of response is documented and changes are noted for future therapeutic decision making.
In patients with suspected HD, the NHDP recommends that a full-thickness 4.0-mm elliptical or punch biopsy be performed at the periphery of the largest or most active lesion and that slides be sent to the NHDP in formalin or paraffin for review.14 Experienced NHDP pathologists may order additional stains. A detailed protocol can be found at www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm.
Proficiency in performing slit smears, a technique to obtain tissue fluid, is also helpful.15 Measurement of the number of bacteria in a set number of fields allows for calculation of the host’s bacterial index, making it possible to determine an appropriate treatment level.
The preferred sites for the slit smear are active lesions, if possible from both ear lobes plus two additional locations. After alcohol swabbing, a tight, bloodless pinch is made, with a 3.0-mm incision that yields blood and fluid. The resulting culture is examined for AFB, which are found abundantly in lepromatous leprosy (whereas tuberculoid leprosy, by definition, should yield none or few). This smear may be repeated at three- to six-month intervals to determine effectiveness of therapy. It is considered superior to a nasal culture, which often yields false-negative results.16
After the Diagnosis
In the US, HD is a nationally notifiable disease: Health care providers are required by law to inform the CDC of the known particulars while complying with HIPAA requirements.17
That the case patient was a recent immigrant from a country with a high incidence of HD, coupled with a loss of sensation in his extremities, led to the correct diagnosis. His lack of response to antifungals or anti-inflammatory agents, though frustrating to both patient and provider, was helpful in excluding two common conditions in the differential diagnosis. Other possibilites that were considered ultimately did not ring true:
Vitiligo, a loss of pigment that produces white patches resembling borderline leprosy but lacks the raised edging18;
Granuloma annulare, a lightly raised, ring-shaped lesion that is usually found on the extremities19; and
Lupus erythematosus, in which patients almost universally experience pruritus and burning, whereas lack of sensation is key to a diagnosis of leprosy.20
Treatment and Management
Health care providers must appear confident to instill confidence in their patients. Any provider with the knowledge and acuity to make a diagnosis of leprosy should be adequately equipped to manage the patient’s care. However, the hesitant or uncertain provider should consider referral to dermatology or to infectious disease for continued management.
The NHDP recognizes the WHO classification of leprosy as multibacillary or paucibacillary (observing a “simple clinical rule” of six or more lesions representing multibacillary HD and five or fewer, paucibacillary HD1), with additional consideration to the three major disease subtypes mentioned earlier: TT, which is treatable; BB, which is considered unstable; and LL, which is resistant to treatment. NHDP and WHO recommendations for treatment differ slightly but address the same goals: complete elimination of the pathogen, preservation of muscle and nerve function, prevention of secondary infections, and management of the adverse effects of medication.1,21 Adhering to universal precautions is important.
In the US, the multidrug treatment regimen for paucibacillary (TT or BT) leprosy comprises one year of oral rifampin 600 mg/d and oral dapsone 100 mg/d21; WHO, in deference to cost containment in developing countries, recommends a six-month regimen of these agents (ie, rifampin 600 mg/mo and dapsone 100 mg/d).1 A third drug sometimes given to allay active neuritis is oral clofazimine 50 mg/d for one year21 (per WHO recommendations, a monthly dose of 300 mg, plus 50 mg/d1). In patients with preexisting or subsequent anemia, dapsone dosing should be lowered to 50 mg, the minimally effective level. Of note, clofazimine is no longer commercially available in the US and is held by the NHDP as an investigational new drug for treatment of US patients.21
According to US recommendations,21 use of this three-agent regimen is extended to two years for patients with multibacillary (LL, BL, BB) leprosy. For these patients, WHO recommends its regimen for 12 months.1
Rifampin is a powerful antibiotic used for effective treatment of tuberculosis, methicillin-resistant Staphylococcus aureus, gonorrhea, Listeria, and Haemophilus influenzae. However, its use has been shown to reduce the efficacy of anticoagulants, oral contraceptives, and prednisone, possibly requiring therapeutic adjustments.22 Recommended alternatives for rifampin include (in the order of preference) minocycline 100 mg/d, ofloxacin 400 mg/d, levofloxacin 500 mg/d, or twice-daily clarithromycin 500 mg.13
Patient compliance with the regimen is facilitated by convenient blister packs that contain standard weekly and monthly doses of these medications, all clearly marked. Many local health departments make these medications available at no cost to patients.
Patient Education
Informing patients what to expect regarding their medications, necessary lab work, and avoiding transmission of HD can ease worry, protect others from illness, and increase the likelihood of an excellent prognosis. Patients with lepromatous (multibacillary) HD should be urged to limit their interactions with others at work or school (particularly preventing exposure to their nasal excretions) until a low bacterial index is achieved. For close contacts of patients recently diagnosed with HD, single-dose rifampin has been shown to provide effective prevention for two years.23
Emphasizing adherence to daily multidrug therapy minimizes the chance that bacterial resistance will develop. Patients should be given complete information about the individual agents used:
Rifampin 600 mg is the maximum daily dose for an adult. Hepatotoxicity is a concern, and liver enzyme levels (ie, aspartate aminotransferase, alanine aminotransferase) should be measured at three-month intervals. It is important to obtain a history of previous liver disease, exposure to hepatitis, and drug and alcohol use. In otherwise healthy adults, rifampin has the curious effect of turning bodily fluids such as urine and tears bright red. Forewarning patients of this benign development may prevent anxious reactions. Contact lenses may be irreversibly stained.24
Dapsone, for many years the first-line monotherapy for leprosy, is a bacteriostatic, sulfa-based antibiotic that only prevents the multiplication of bacteria, but it is still worthy of respect. Before dapsone is initiated, a screening test for glucose-6-phosphate dehydrogenase deficiency should be performed to determine the risk for dapsone-associated hemolysis.24 Dapsone use is contraindicated in women who are breastfeeding. In patients with severe heart conditions, dapsone can reduce oxygen flow, as evidenced by a bluish discoloration of the lips and fingertips. Mild anemia, headache, gastrointestinal upset and nausea are all common adverse effects.
Clofazimine, a soft gelatin capsule, may cause gray-blue or red hyperpigmentation in individuals who are light-skinned; this effect reverses once the drug is discontinued. Nausea, vomiting, and abdominal pain have been reported. This slow-release bactericidal drug has an extremely long half-life of 70 days. It provides anti-inflammatory action similar to that of prednisone without the latter agent’s long-term adverse effects.24
The Patient’s Outcome
The case patient was the second with HD seen in this dermatology practice. A man who presented on an earlier occasion, possibly an undocumented worker in fear of deportation, seemed concerned about the reporting process, even though he was assured that no personal information would be released. He did not return for follow-up, and attempts to reach him proved fruitless.
The case patient, by contrast, was pleasant and cooperative. He appeared to understand his condition, the required treatment and its possible adverse effects, and the need for continued lab work and regular follow-up appointments. A day laborer in a large factory, he was particularly careful to avoid transmission of his illness to his family or co-workers.
Within two months of beginning multidrug therapy, he experienced quick clearance of the lesions on his back. His bacterial index approached 0 by the time he underwent the second smear. Although he complained of loose stools and classic orange-red bodily fluid throughout the entire year of treatment, he resisted the temptation to discontinue the regimen.
He has experienced no relapses in six years and is considered cured.
Conclusion
Only one in every 20 people is susceptible to infection with M leprae, but cases of leprosy continue to be reported in the US, and illness is of particular concern among immunocompromised individuals. Treatment delay is common because of the condition’s relative rarity in the US, but therapy with standard antibiotics is extremely effective. Lengthy isolation from family and community is not often needed, and relapses are rare. Fully restored health is the rule rather than the exception for patients treated for Hansen’s disease.
1. World Health Organization. Global strategy for further reducing the leprosy burden and sustaining leprosy control activities 2006-2010: operational guidelines. www.who.int/lep/resources/SEAGLP20062.pdf. Accessed November 17, 2009.
2. World Health Organization Communicable -Diseases Department. Leprosy: frequently as-ked questions. www.searo.who.int/EN/Section10/Section373_11716.htm. Accessed November 17, 2009.
3. National Hansen’s Disease (Leprosy) Program. www.hrsa.gov/hansens. Accessed November 23, 2009.
4. Health Resources and Services Administration, US Department of Health and Human Services, National Hansen’s Disease Program. A summary of Hansen’s disease in the United States—2006. ftp://ftp.hrsa.gov/hansens/2006RegistryReport.pdf. Accessed November 17, 2009.
5. Boggild AK, Correia JD, Keystone JS, Kain KC. Leprosy in Toronto: an analysis of 184 imported cases. CMAJ. 2004;170(1):55-59.
6. Cheesbrough M. Bacterial pathogens. In: District Laboratory Practice in Tropical Countries. Part 2. 2nd ed. Cambridge, UK: Cambridge University Press. 2006;157-234.
7. Sasaki S, Takeshita F, Okuda K, Ishii N. Mycobacterium leprae and leprosy: a compendium. Microbiol Immunol. 2001;45(11):729–736.
8. Job CK, Jayakumar J, Kearney M, Gillis TP. Transmission of leprosy: a study of skin and nasal secretions of household contacts of leprosy patients using PCR. Am J Trop Med Hyg. 2008; 78(3):518-521.
9. Girdhar BK. Skin to skin transmission of leprosy. Indian J Dermatol Venereol Leprol. 2005;71(4):223-225.
10. National Leprosy Eradication Programme, Directorate General of Health Services (Government of India). Differential diagnosis for leprosy (2009). http://nlep.nic.in/pdf/Annex-%20III% 20Differential%20diagnosis.pdf. September 14, 2009.
11. Kim EC. Hansen disease: differential diagnoses and workup (2006). http://emedicine.medscape .com/article/1213853-diagnosis. Accessed November 17, 2009.
12. James WD, Berger TG, Elston DM. Hansen’s disease. In: Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Philadelphia, PA: Saunders. 2005;343-366.
13. Leprosy. In: Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 2nd ed. Philadelphia, PA: WB Saunders; 2005.
14. National Hansen’s Disease Program. Skin biopsy in the diagnosis of Hansen’s disease. www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm. Accessed November 17, 2009.
15. Bhushan P, Sardana K, Koranne RV, et al. Diagnosing multibacillary leprosy: a comparative evaluation of diagnostic accuracy of slit-skin smear, bacterial index of granuloma and WHO operational classification. Indian J Dermatol Venereol Leprol. 2008;74(4):322-326.
16. Wolff K, Johnson R, Suurmond R. Leprosy. In: Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. Chicago, IL: McGraw-Hill Professional; 2005:655-661.
17. Centers for Disease Control and Prevention. Summary of notifiable diseases—United States, 2007. MMWR Morb Mortal Wkly Rep. 2009;56 (53):3, 10, 22.
18. American Academy of Dermatology. Vitiligo. www.aad.org/public/publications/pamphlets/com mon_vitilgo.html. Accessed November 17, 2009.
19. American Academy of Dermatology. Granuloma annulare. www.aad.org/public/publica tions/pamphlets/common_granuloma.html. Accessed November 17, 2009.
20. American Academy of Dermatology. Lupus and the skin. www.aad.org/public/publications/pamphlets/common_lupus.html. Accessed November 17, 2009.
21. National Hansen’s Disease Program. Recommended treatment regimens. www.hrsa.gov/han sens/clinical/regimens.htm. Accessed November 17, 2009.
22. Chen J, Raymond K. Roles of rifampicin in drug-drug interactions: underlying molecular mechanisms involving the nuclear pregnane X receptor. Ann Clin Microbiol Antimicrob. 2006 Feb 15;5:3.
23. Moet FJ, Pahan D, Oskam L, Richardus JH; COLEP Study Group. Effectiveness of single dose rifampicin in preventing leprosy in close contacts of patients with newly diagnosed leprosy: cluster randomised controlled trial. BMJ. 2008;336 (7647):761-764.
24. World Health Organization. WHO model prescribing information: drugs used in leprosy. http://apps.who.int/medicinedocs/fr/d/Jh2988e/ 14.2.html. Accessed November 17, 2009.
25. Bruce S, Schroeder TL, Ellner K, et al. Armadillo exposure and Hansen’s disease: an epidemiologic survey in southern Texas. J Am Acad Dermatol. 2000;43(2 pt 1):223-228.
26. Clark BM, Murray CK, Horvath LL, et al. Case-control study of armadillo contact and Hansen’s disease. Am J Trop Med Hyg. 2008;78(6):962-967.
According to data from the World Health Organization (WHO), the number of new cases of Hansen’s disease (HD, or leprosy) identified worldwide declined by about 20% from 2003 to 2004. At least 116 of 122 countries once considered leprosy—endemic have achieved a prevalence rate of less than one case per 10,000 population.1,2
Meanwhile, the number of active HD infections in the United States catalogued through the CDC and the National Hansen’s Disease Program (NHDP) rose from 76 cases in 2000 to 166 in 2005, with an additional 137 cases in 2006 (the most recent year for which complete figures are available).3,4 Currently, 6,500 US residents are known to have HD, of whom 3,300 require care for active disease.3
Accurate diagnosis and straightforward, aggressive treatment can mean certain cure for this dreaded disease, as it is responsive to a combination of available drugs. For primary care providers in the US, familiarity with this condition can play a role in eliminating it altogether.
Background and Etiology
HD is believed to have originated in East Africa with the pseudonym of leprosy, from the Greek lepi, meaning “fish scales.” Although HD is one of the oldest known human infections, it is not well understood by many Western health care providers. Once considered highly contagious and easily transmissible, HD has proven to be neither: 95% of the human population is not susceptible to the responsible pathogen.3 HD retains its reputation as a devastating illness when left untreated, however, with sequelae often including significant sensory and motor dysfunction.5
Leprosy is a chronic bacterial disease caused by Mycobacterium leprae. Mycobacterium is the same rod-shaped, acid-fast bacillus (AFB) implicated in pulmonary tuberculosis, cutaneous tuberculosis, and numerous cutaneous nontuberculoid infections.6,7
HD transmission is believed to occur almost exclusively by nasal droplet, inhaled by way of others’ nares or lungs. Research continues in possible transmission by skin-to-skin contact.8,9
Leprosy is particularly challenging for immunocompromised persons, such as those with HIV/AIDS or tuberculosis. According to data from the NHDP, HD in the US is found primarily among undocumented immigrants who live in close quarters. Cases considered “home-grown” rather than “imported”5 are most commonly diagnosed in Hawaii and in a swath from Texas to Georgia and south toward Florida. Pockets of HD incidence are also found in California, New York, and Massachusetts.4
Clinicians’ lack of familiarity with HD and its relative rarity in the US help explain why diagnosis and treatment are commonly delayed. As a patient’s disease progresses from an insidious, gradual, and relatively painless onset, the primary care provider may be grappling with an extensive differential diagnosis (see “Differential Diagnosis for Hansen’s Disease [Leprosy],”10,11 below). Even after confirming a diagnosis of HD, the provider must contend with a lack of information and misinformation about HD among staff members, the patient’s family, and society at large, as well as the associated stigma. Patient education and effective treatment are of comparable importance.
Patient Presentation and History
A 40-year-old Latino man presented to a primary care provider with a well-demarcated, erythematous patch on his right upper back that had raised outer edging and faint flaking. The patch was surrounded by several smaller but similar asymmetrical macules. The patient denied pruritus but stated that the area “felt funny.” He said the lesion might have been spreading during the previous several months, but he could not be certain because of its location.
The man denied any medication use except for a topical antibiotic ointment he had applied to the affected area with no improvement. His provider made a diagnosis of tinea corporis and prescribed an antifungal cream, naftifine. After two weeks without results, the patient was referred to a dermatologist.
During the assessment, it became apparent that the patient was experiencing numbness in his fingertips and general muscle weakness in his arms. A KOH prep was difficult to obtain, as the lesion site was somewhat smooth. Any dermatophytes that may have been present had been eliminated by the naftifine, and KOH results were negative.
A 5.0-mm punch biopsy from the largest lesion was ordered. A preliminary diagnosis of sarcoidosis (chronic inflammation with an unusual skin manifestation) was made, with serious consideration given to early cutaneous T-cell lymphoma (mycosis fungoides). A high-potency steroid was prescribed for five days’ use, twice daily.
On his return to the dermatologist, the patient reported that the site was no better. He was informed that the dermatologist and the pathologist, both with considerable experience, had detected no bacilli or dermal nerve infiltration in the tissue sample but had observed abundant lymphocytic infiltration. This finding, coupled with the patient’s decreased sensation, muscle fatigue, and status as a recent immigrant from a South American country, led to an initial diagnosis of borderline leprosy (BB).
Physical Examination and Disease Progression
If the first provider suspected a fungal infection during the initial examination, he should have completed a KOH scraping to confirm that suspicion. While superficial fungal infections are extremely common, particularly in the southeastern US, a sampling of scale with KOH 5% to 20% and the absence of dermatophytes under the microscope might have permitted a quick rule-out.12 Additionally, dermatophytic infections are very responsive to current medications within days, not weeks.
Thus, the key to an accurate diagnosis of leprosy begins with a physical examination that takes into account both chronic unresolved cutaneous lesions and neurologic changes in sensitivity.
A great majority of patients with HD have no noteworthy clinical symptoms until the first skin manifestations. While sensory neuropathy is prominent in all patients, additional findings (depending on the type of leprosy involved) may include plantar ulcers, chronic nasal congestion with early evidence of cartilage loss over the nasal bridge, chronic nosebleeds, cranial nerve palsies, and eye paralysis.12 In rare cases, hypogonadism may develop, or the clinical team may discover amyloidosis, an accumulation of insoluble proteins in the tissues that impairs function in various organs; this condition is usually detected by skin biopsy. If early or indeterminate leprosy is identified, neither form persists; rather, the condition will progress to one of the following:
• Borderline lepromatous leprosy (BL) and lepromatous leprosy (LL) offer very high resistance to treatment and are considered malignant forms of HD. Findings in patients with BL or LL include multiple lesions in various crazy-quilt presentations: macules, papules, plaques, nodules, masses, and patches of deep, erythematous infiltration across the extremities, face, and trunk.12 Loss of all hair except on the scalp is possible. Ulcers may form on the palms of the hands and soles of the feet.
In both BL and LL, results on AFB smears will reflect moderately high or very high concentrations of the pathogen in the patient’s system. Thus, these forms of HD are defined as multibacillary. If either is left untreated, the accumulation of proteins attributed to amyloidosis and secondary infections that develop eventually lead to death.
• Borderline leprosy (BB) has already been described in the case patient, with five or six ill-defined lesions and mild impairment of the peripheral nervous system. Aggressive treatment of this form of HD is important. A strong immune system provides no guarantee of successful treatment, and therapy must forestall a likely continuum to more serious forms.13
• Borderline tuberculoid leprosy (BT) and tuberculoid leprosy (TT) are both forms of paucibacillary HD. They offer little resistance to treatment in the patient with a healthy immune system. Lesions are generally scant (fewer than five; see figure), and the AFB smear reveals 0 to 10 bacteria in 100 fields.12
For the primary care provider, a thorough physical examination also requires familiarity with reasonable differentials. Hallmarks of conditions less likely to be leprosy include nausea, vomiting, or diarrhea, fever and chills, rectal bleeding and unexplained weight loss, headaches, and shortness of breath.
Diagnostic Testing
To help gauge progression of the disease and assess effectiveness of therapy, it is common practice to conduct a neurosensory test at each office visit. This is performed by touching the patient’s extremities and the periphery of lesions with a simple nylon monofilament.13 The patient’s response or lack of response is documented and changes are noted for future therapeutic decision making.
In patients with suspected HD, the NHDP recommends that a full-thickness 4.0-mm elliptical or punch biopsy be performed at the periphery of the largest or most active lesion and that slides be sent to the NHDP in formalin or paraffin for review.14 Experienced NHDP pathologists may order additional stains. A detailed protocol can be found at www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm.
Proficiency in performing slit smears, a technique to obtain tissue fluid, is also helpful.15 Measurement of the number of bacteria in a set number of fields allows for calculation of the host’s bacterial index, making it possible to determine an appropriate treatment level.
The preferred sites for the slit smear are active lesions, if possible from both ear lobes plus two additional locations. After alcohol swabbing, a tight, bloodless pinch is made, with a 3.0-mm incision that yields blood and fluid. The resulting culture is examined for AFB, which are found abundantly in lepromatous leprosy (whereas tuberculoid leprosy, by definition, should yield none or few). This smear may be repeated at three- to six-month intervals to determine effectiveness of therapy. It is considered superior to a nasal culture, which often yields false-negative results.16
After the Diagnosis
In the US, HD is a nationally notifiable disease: Health care providers are required by law to inform the CDC of the known particulars while complying with HIPAA requirements.17
That the case patient was a recent immigrant from a country with a high incidence of HD, coupled with a loss of sensation in his extremities, led to the correct diagnosis. His lack of response to antifungals or anti-inflammatory agents, though frustrating to both patient and provider, was helpful in excluding two common conditions in the differential diagnosis. Other possibilites that were considered ultimately did not ring true:
Vitiligo, a loss of pigment that produces white patches resembling borderline leprosy but lacks the raised edging18;
Granuloma annulare, a lightly raised, ring-shaped lesion that is usually found on the extremities19; and
Lupus erythematosus, in which patients almost universally experience pruritus and burning, whereas lack of sensation is key to a diagnosis of leprosy.20
Treatment and Management
Health care providers must appear confident to instill confidence in their patients. Any provider with the knowledge and acuity to make a diagnosis of leprosy should be adequately equipped to manage the patient’s care. However, the hesitant or uncertain provider should consider referral to dermatology or to infectious disease for continued management.
The NHDP recognizes the WHO classification of leprosy as multibacillary or paucibacillary (observing a “simple clinical rule” of six or more lesions representing multibacillary HD and five or fewer, paucibacillary HD1), with additional consideration to the three major disease subtypes mentioned earlier: TT, which is treatable; BB, which is considered unstable; and LL, which is resistant to treatment. NHDP and WHO recommendations for treatment differ slightly but address the same goals: complete elimination of the pathogen, preservation of muscle and nerve function, prevention of secondary infections, and management of the adverse effects of medication.1,21 Adhering to universal precautions is important.
In the US, the multidrug treatment regimen for paucibacillary (TT or BT) leprosy comprises one year of oral rifampin 600 mg/d and oral dapsone 100 mg/d21; WHO, in deference to cost containment in developing countries, recommends a six-month regimen of these agents (ie, rifampin 600 mg/mo and dapsone 100 mg/d).1 A third drug sometimes given to allay active neuritis is oral clofazimine 50 mg/d for one year21 (per WHO recommendations, a monthly dose of 300 mg, plus 50 mg/d1). In patients with preexisting or subsequent anemia, dapsone dosing should be lowered to 50 mg, the minimally effective level. Of note, clofazimine is no longer commercially available in the US and is held by the NHDP as an investigational new drug for treatment of US patients.21
According to US recommendations,21 use of this three-agent regimen is extended to two years for patients with multibacillary (LL, BL, BB) leprosy. For these patients, WHO recommends its regimen for 12 months.1
Rifampin is a powerful antibiotic used for effective treatment of tuberculosis, methicillin-resistant Staphylococcus aureus, gonorrhea, Listeria, and Haemophilus influenzae. However, its use has been shown to reduce the efficacy of anticoagulants, oral contraceptives, and prednisone, possibly requiring therapeutic adjustments.22 Recommended alternatives for rifampin include (in the order of preference) minocycline 100 mg/d, ofloxacin 400 mg/d, levofloxacin 500 mg/d, or twice-daily clarithromycin 500 mg.13
Patient compliance with the regimen is facilitated by convenient blister packs that contain standard weekly and monthly doses of these medications, all clearly marked. Many local health departments make these medications available at no cost to patients.
Patient Education
Informing patients what to expect regarding their medications, necessary lab work, and avoiding transmission of HD can ease worry, protect others from illness, and increase the likelihood of an excellent prognosis. Patients with lepromatous (multibacillary) HD should be urged to limit their interactions with others at work or school (particularly preventing exposure to their nasal excretions) until a low bacterial index is achieved. For close contacts of patients recently diagnosed with HD, single-dose rifampin has been shown to provide effective prevention for two years.23
Emphasizing adherence to daily multidrug therapy minimizes the chance that bacterial resistance will develop. Patients should be given complete information about the individual agents used:
Rifampin 600 mg is the maximum daily dose for an adult. Hepatotoxicity is a concern, and liver enzyme levels (ie, aspartate aminotransferase, alanine aminotransferase) should be measured at three-month intervals. It is important to obtain a history of previous liver disease, exposure to hepatitis, and drug and alcohol use. In otherwise healthy adults, rifampin has the curious effect of turning bodily fluids such as urine and tears bright red. Forewarning patients of this benign development may prevent anxious reactions. Contact lenses may be irreversibly stained.24
Dapsone, for many years the first-line monotherapy for leprosy, is a bacteriostatic, sulfa-based antibiotic that only prevents the multiplication of bacteria, but it is still worthy of respect. Before dapsone is initiated, a screening test for glucose-6-phosphate dehydrogenase deficiency should be performed to determine the risk for dapsone-associated hemolysis.24 Dapsone use is contraindicated in women who are breastfeeding. In patients with severe heart conditions, dapsone can reduce oxygen flow, as evidenced by a bluish discoloration of the lips and fingertips. Mild anemia, headache, gastrointestinal upset and nausea are all common adverse effects.
Clofazimine, a soft gelatin capsule, may cause gray-blue or red hyperpigmentation in individuals who are light-skinned; this effect reverses once the drug is discontinued. Nausea, vomiting, and abdominal pain have been reported. This slow-release bactericidal drug has an extremely long half-life of 70 days. It provides anti-inflammatory action similar to that of prednisone without the latter agent’s long-term adverse effects.24
The Patient’s Outcome
The case patient was the second with HD seen in this dermatology practice. A man who presented on an earlier occasion, possibly an undocumented worker in fear of deportation, seemed concerned about the reporting process, even though he was assured that no personal information would be released. He did not return for follow-up, and attempts to reach him proved fruitless.
The case patient, by contrast, was pleasant and cooperative. He appeared to understand his condition, the required treatment and its possible adverse effects, and the need for continued lab work and regular follow-up appointments. A day laborer in a large factory, he was particularly careful to avoid transmission of his illness to his family or co-workers.
Within two months of beginning multidrug therapy, he experienced quick clearance of the lesions on his back. His bacterial index approached 0 by the time he underwent the second smear. Although he complained of loose stools and classic orange-red bodily fluid throughout the entire year of treatment, he resisted the temptation to discontinue the regimen.
He has experienced no relapses in six years and is considered cured.
Conclusion
Only one in every 20 people is susceptible to infection with M leprae, but cases of leprosy continue to be reported in the US, and illness is of particular concern among immunocompromised individuals. Treatment delay is common because of the condition’s relative rarity in the US, but therapy with standard antibiotics is extremely effective. Lengthy isolation from family and community is not often needed, and relapses are rare. Fully restored health is the rule rather than the exception for patients treated for Hansen’s disease.
According to data from the World Health Organization (WHO), the number of new cases of Hansen’s disease (HD, or leprosy) identified worldwide declined by about 20% from 2003 to 2004. At least 116 of 122 countries once considered leprosy—endemic have achieved a prevalence rate of less than one case per 10,000 population.1,2
Meanwhile, the number of active HD infections in the United States catalogued through the CDC and the National Hansen’s Disease Program (NHDP) rose from 76 cases in 2000 to 166 in 2005, with an additional 137 cases in 2006 (the most recent year for which complete figures are available).3,4 Currently, 6,500 US residents are known to have HD, of whom 3,300 require care for active disease.3
Accurate diagnosis and straightforward, aggressive treatment can mean certain cure for this dreaded disease, as it is responsive to a combination of available drugs. For primary care providers in the US, familiarity with this condition can play a role in eliminating it altogether.
Background and Etiology
HD is believed to have originated in East Africa with the pseudonym of leprosy, from the Greek lepi, meaning “fish scales.” Although HD is one of the oldest known human infections, it is not well understood by many Western health care providers. Once considered highly contagious and easily transmissible, HD has proven to be neither: 95% of the human population is not susceptible to the responsible pathogen.3 HD retains its reputation as a devastating illness when left untreated, however, with sequelae often including significant sensory and motor dysfunction.5
Leprosy is a chronic bacterial disease caused by Mycobacterium leprae. Mycobacterium is the same rod-shaped, acid-fast bacillus (AFB) implicated in pulmonary tuberculosis, cutaneous tuberculosis, and numerous cutaneous nontuberculoid infections.6,7
HD transmission is believed to occur almost exclusively by nasal droplet, inhaled by way of others’ nares or lungs. Research continues in possible transmission by skin-to-skin contact.8,9
Leprosy is particularly challenging for immunocompromised persons, such as those with HIV/AIDS or tuberculosis. According to data from the NHDP, HD in the US is found primarily among undocumented immigrants who live in close quarters. Cases considered “home-grown” rather than “imported”5 are most commonly diagnosed in Hawaii and in a swath from Texas to Georgia and south toward Florida. Pockets of HD incidence are also found in California, New York, and Massachusetts.4
Clinicians’ lack of familiarity with HD and its relative rarity in the US help explain why diagnosis and treatment are commonly delayed. As a patient’s disease progresses from an insidious, gradual, and relatively painless onset, the primary care provider may be grappling with an extensive differential diagnosis (see “Differential Diagnosis for Hansen’s Disease [Leprosy],”10,11 below). Even after confirming a diagnosis of HD, the provider must contend with a lack of information and misinformation about HD among staff members, the patient’s family, and society at large, as well as the associated stigma. Patient education and effective treatment are of comparable importance.
Patient Presentation and History
A 40-year-old Latino man presented to a primary care provider with a well-demarcated, erythematous patch on his right upper back that had raised outer edging and faint flaking. The patch was surrounded by several smaller but similar asymmetrical macules. The patient denied pruritus but stated that the area “felt funny.” He said the lesion might have been spreading during the previous several months, but he could not be certain because of its location.
The man denied any medication use except for a topical antibiotic ointment he had applied to the affected area with no improvement. His provider made a diagnosis of tinea corporis and prescribed an antifungal cream, naftifine. After two weeks without results, the patient was referred to a dermatologist.
During the assessment, it became apparent that the patient was experiencing numbness in his fingertips and general muscle weakness in his arms. A KOH prep was difficult to obtain, as the lesion site was somewhat smooth. Any dermatophytes that may have been present had been eliminated by the naftifine, and KOH results were negative.
A 5.0-mm punch biopsy from the largest lesion was ordered. A preliminary diagnosis of sarcoidosis (chronic inflammation with an unusual skin manifestation) was made, with serious consideration given to early cutaneous T-cell lymphoma (mycosis fungoides). A high-potency steroid was prescribed for five days’ use, twice daily.
On his return to the dermatologist, the patient reported that the site was no better. He was informed that the dermatologist and the pathologist, both with considerable experience, had detected no bacilli or dermal nerve infiltration in the tissue sample but had observed abundant lymphocytic infiltration. This finding, coupled with the patient’s decreased sensation, muscle fatigue, and status as a recent immigrant from a South American country, led to an initial diagnosis of borderline leprosy (BB).
Physical Examination and Disease Progression
If the first provider suspected a fungal infection during the initial examination, he should have completed a KOH scraping to confirm that suspicion. While superficial fungal infections are extremely common, particularly in the southeastern US, a sampling of scale with KOH 5% to 20% and the absence of dermatophytes under the microscope might have permitted a quick rule-out.12 Additionally, dermatophytic infections are very responsive to current medications within days, not weeks.
Thus, the key to an accurate diagnosis of leprosy begins with a physical examination that takes into account both chronic unresolved cutaneous lesions and neurologic changes in sensitivity.
A great majority of patients with HD have no noteworthy clinical symptoms until the first skin manifestations. While sensory neuropathy is prominent in all patients, additional findings (depending on the type of leprosy involved) may include plantar ulcers, chronic nasal congestion with early evidence of cartilage loss over the nasal bridge, chronic nosebleeds, cranial nerve palsies, and eye paralysis.12 In rare cases, hypogonadism may develop, or the clinical team may discover amyloidosis, an accumulation of insoluble proteins in the tissues that impairs function in various organs; this condition is usually detected by skin biopsy. If early or indeterminate leprosy is identified, neither form persists; rather, the condition will progress to one of the following:
• Borderline lepromatous leprosy (BL) and lepromatous leprosy (LL) offer very high resistance to treatment and are considered malignant forms of HD. Findings in patients with BL or LL include multiple lesions in various crazy-quilt presentations: macules, papules, plaques, nodules, masses, and patches of deep, erythematous infiltration across the extremities, face, and trunk.12 Loss of all hair except on the scalp is possible. Ulcers may form on the palms of the hands and soles of the feet.
In both BL and LL, results on AFB smears will reflect moderately high or very high concentrations of the pathogen in the patient’s system. Thus, these forms of HD are defined as multibacillary. If either is left untreated, the accumulation of proteins attributed to amyloidosis and secondary infections that develop eventually lead to death.
• Borderline leprosy (BB) has already been described in the case patient, with five or six ill-defined lesions and mild impairment of the peripheral nervous system. Aggressive treatment of this form of HD is important. A strong immune system provides no guarantee of successful treatment, and therapy must forestall a likely continuum to more serious forms.13
• Borderline tuberculoid leprosy (BT) and tuberculoid leprosy (TT) are both forms of paucibacillary HD. They offer little resistance to treatment in the patient with a healthy immune system. Lesions are generally scant (fewer than five; see figure), and the AFB smear reveals 0 to 10 bacteria in 100 fields.12
For the primary care provider, a thorough physical examination also requires familiarity with reasonable differentials. Hallmarks of conditions less likely to be leprosy include nausea, vomiting, or diarrhea, fever and chills, rectal bleeding and unexplained weight loss, headaches, and shortness of breath.
Diagnostic Testing
To help gauge progression of the disease and assess effectiveness of therapy, it is common practice to conduct a neurosensory test at each office visit. This is performed by touching the patient’s extremities and the periphery of lesions with a simple nylon monofilament.13 The patient’s response or lack of response is documented and changes are noted for future therapeutic decision making.
In patients with suspected HD, the NHDP recommends that a full-thickness 4.0-mm elliptical or punch biopsy be performed at the periphery of the largest or most active lesion and that slides be sent to the NHDP in formalin or paraffin for review.14 Experienced NHDP pathologists may order additional stains. A detailed protocol can be found at www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm.
Proficiency in performing slit smears, a technique to obtain tissue fluid, is also helpful.15 Measurement of the number of bacteria in a set number of fields allows for calculation of the host’s bacterial index, making it possible to determine an appropriate treatment level.
The preferred sites for the slit smear are active lesions, if possible from both ear lobes plus two additional locations. After alcohol swabbing, a tight, bloodless pinch is made, with a 3.0-mm incision that yields blood and fluid. The resulting culture is examined for AFB, which are found abundantly in lepromatous leprosy (whereas tuberculoid leprosy, by definition, should yield none or few). This smear may be repeated at three- to six-month intervals to determine effectiveness of therapy. It is considered superior to a nasal culture, which often yields false-negative results.16
After the Diagnosis
In the US, HD is a nationally notifiable disease: Health care providers are required by law to inform the CDC of the known particulars while complying with HIPAA requirements.17
That the case patient was a recent immigrant from a country with a high incidence of HD, coupled with a loss of sensation in his extremities, led to the correct diagnosis. His lack of response to antifungals or anti-inflammatory agents, though frustrating to both patient and provider, was helpful in excluding two common conditions in the differential diagnosis. Other possibilites that were considered ultimately did not ring true:
Vitiligo, a loss of pigment that produces white patches resembling borderline leprosy but lacks the raised edging18;
Granuloma annulare, a lightly raised, ring-shaped lesion that is usually found on the extremities19; and
Lupus erythematosus, in which patients almost universally experience pruritus and burning, whereas lack of sensation is key to a diagnosis of leprosy.20
Treatment and Management
Health care providers must appear confident to instill confidence in their patients. Any provider with the knowledge and acuity to make a diagnosis of leprosy should be adequately equipped to manage the patient’s care. However, the hesitant or uncertain provider should consider referral to dermatology or to infectious disease for continued management.
The NHDP recognizes the WHO classification of leprosy as multibacillary or paucibacillary (observing a “simple clinical rule” of six or more lesions representing multibacillary HD and five or fewer, paucibacillary HD1), with additional consideration to the three major disease subtypes mentioned earlier: TT, which is treatable; BB, which is considered unstable; and LL, which is resistant to treatment. NHDP and WHO recommendations for treatment differ slightly but address the same goals: complete elimination of the pathogen, preservation of muscle and nerve function, prevention of secondary infections, and management of the adverse effects of medication.1,21 Adhering to universal precautions is important.
In the US, the multidrug treatment regimen for paucibacillary (TT or BT) leprosy comprises one year of oral rifampin 600 mg/d and oral dapsone 100 mg/d21; WHO, in deference to cost containment in developing countries, recommends a six-month regimen of these agents (ie, rifampin 600 mg/mo and dapsone 100 mg/d).1 A third drug sometimes given to allay active neuritis is oral clofazimine 50 mg/d for one year21 (per WHO recommendations, a monthly dose of 300 mg, plus 50 mg/d1). In patients with preexisting or subsequent anemia, dapsone dosing should be lowered to 50 mg, the minimally effective level. Of note, clofazimine is no longer commercially available in the US and is held by the NHDP as an investigational new drug for treatment of US patients.21
According to US recommendations,21 use of this three-agent regimen is extended to two years for patients with multibacillary (LL, BL, BB) leprosy. For these patients, WHO recommends its regimen for 12 months.1
Rifampin is a powerful antibiotic used for effective treatment of tuberculosis, methicillin-resistant Staphylococcus aureus, gonorrhea, Listeria, and Haemophilus influenzae. However, its use has been shown to reduce the efficacy of anticoagulants, oral contraceptives, and prednisone, possibly requiring therapeutic adjustments.22 Recommended alternatives for rifampin include (in the order of preference) minocycline 100 mg/d, ofloxacin 400 mg/d, levofloxacin 500 mg/d, or twice-daily clarithromycin 500 mg.13
Patient compliance with the regimen is facilitated by convenient blister packs that contain standard weekly and monthly doses of these medications, all clearly marked. Many local health departments make these medications available at no cost to patients.
Patient Education
Informing patients what to expect regarding their medications, necessary lab work, and avoiding transmission of HD can ease worry, protect others from illness, and increase the likelihood of an excellent prognosis. Patients with lepromatous (multibacillary) HD should be urged to limit their interactions with others at work or school (particularly preventing exposure to their nasal excretions) until a low bacterial index is achieved. For close contacts of patients recently diagnosed with HD, single-dose rifampin has been shown to provide effective prevention for two years.23
Emphasizing adherence to daily multidrug therapy minimizes the chance that bacterial resistance will develop. Patients should be given complete information about the individual agents used:
Rifampin 600 mg is the maximum daily dose for an adult. Hepatotoxicity is a concern, and liver enzyme levels (ie, aspartate aminotransferase, alanine aminotransferase) should be measured at three-month intervals. It is important to obtain a history of previous liver disease, exposure to hepatitis, and drug and alcohol use. In otherwise healthy adults, rifampin has the curious effect of turning bodily fluids such as urine and tears bright red. Forewarning patients of this benign development may prevent anxious reactions. Contact lenses may be irreversibly stained.24
Dapsone, for many years the first-line monotherapy for leprosy, is a bacteriostatic, sulfa-based antibiotic that only prevents the multiplication of bacteria, but it is still worthy of respect. Before dapsone is initiated, a screening test for glucose-6-phosphate dehydrogenase deficiency should be performed to determine the risk for dapsone-associated hemolysis.24 Dapsone use is contraindicated in women who are breastfeeding. In patients with severe heart conditions, dapsone can reduce oxygen flow, as evidenced by a bluish discoloration of the lips and fingertips. Mild anemia, headache, gastrointestinal upset and nausea are all common adverse effects.
Clofazimine, a soft gelatin capsule, may cause gray-blue or red hyperpigmentation in individuals who are light-skinned; this effect reverses once the drug is discontinued. Nausea, vomiting, and abdominal pain have been reported. This slow-release bactericidal drug has an extremely long half-life of 70 days. It provides anti-inflammatory action similar to that of prednisone without the latter agent’s long-term adverse effects.24
The Patient’s Outcome
The case patient was the second with HD seen in this dermatology practice. A man who presented on an earlier occasion, possibly an undocumented worker in fear of deportation, seemed concerned about the reporting process, even though he was assured that no personal information would be released. He did not return for follow-up, and attempts to reach him proved fruitless.
The case patient, by contrast, was pleasant and cooperative. He appeared to understand his condition, the required treatment and its possible adverse effects, and the need for continued lab work and regular follow-up appointments. A day laborer in a large factory, he was particularly careful to avoid transmission of his illness to his family or co-workers.
Within two months of beginning multidrug therapy, he experienced quick clearance of the lesions on his back. His bacterial index approached 0 by the time he underwent the second smear. Although he complained of loose stools and classic orange-red bodily fluid throughout the entire year of treatment, he resisted the temptation to discontinue the regimen.
He has experienced no relapses in six years and is considered cured.
Conclusion
Only one in every 20 people is susceptible to infection with M leprae, but cases of leprosy continue to be reported in the US, and illness is of particular concern among immunocompromised individuals. Treatment delay is common because of the condition’s relative rarity in the US, but therapy with standard antibiotics is extremely effective. Lengthy isolation from family and community is not often needed, and relapses are rare. Fully restored health is the rule rather than the exception for patients treated for Hansen’s disease.
1. World Health Organization. Global strategy for further reducing the leprosy burden and sustaining leprosy control activities 2006-2010: operational guidelines. www.who.int/lep/resources/SEAGLP20062.pdf. Accessed November 17, 2009.
2. World Health Organization Communicable -Diseases Department. Leprosy: frequently as-ked questions. www.searo.who.int/EN/Section10/Section373_11716.htm. Accessed November 17, 2009.
3. National Hansen’s Disease (Leprosy) Program. www.hrsa.gov/hansens. Accessed November 23, 2009.
4. Health Resources and Services Administration, US Department of Health and Human Services, National Hansen’s Disease Program. A summary of Hansen’s disease in the United States—2006. ftp://ftp.hrsa.gov/hansens/2006RegistryReport.pdf. Accessed November 17, 2009.
5. Boggild AK, Correia JD, Keystone JS, Kain KC. Leprosy in Toronto: an analysis of 184 imported cases. CMAJ. 2004;170(1):55-59.
6. Cheesbrough M. Bacterial pathogens. In: District Laboratory Practice in Tropical Countries. Part 2. 2nd ed. Cambridge, UK: Cambridge University Press. 2006;157-234.
7. Sasaki S, Takeshita F, Okuda K, Ishii N. Mycobacterium leprae and leprosy: a compendium. Microbiol Immunol. 2001;45(11):729–736.
8. Job CK, Jayakumar J, Kearney M, Gillis TP. Transmission of leprosy: a study of skin and nasal secretions of household contacts of leprosy patients using PCR. Am J Trop Med Hyg. 2008; 78(3):518-521.
9. Girdhar BK. Skin to skin transmission of leprosy. Indian J Dermatol Venereol Leprol. 2005;71(4):223-225.
10. National Leprosy Eradication Programme, Directorate General of Health Services (Government of India). Differential diagnosis for leprosy (2009). http://nlep.nic.in/pdf/Annex-%20III% 20Differential%20diagnosis.pdf. September 14, 2009.
11. Kim EC. Hansen disease: differential diagnoses and workup (2006). http://emedicine.medscape .com/article/1213853-diagnosis. Accessed November 17, 2009.
12. James WD, Berger TG, Elston DM. Hansen’s disease. In: Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Philadelphia, PA: Saunders. 2005;343-366.
13. Leprosy. In: Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 2nd ed. Philadelphia, PA: WB Saunders; 2005.
14. National Hansen’s Disease Program. Skin biopsy in the diagnosis of Hansen’s disease. www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm. Accessed November 17, 2009.
15. Bhushan P, Sardana K, Koranne RV, et al. Diagnosing multibacillary leprosy: a comparative evaluation of diagnostic accuracy of slit-skin smear, bacterial index of granuloma and WHO operational classification. Indian J Dermatol Venereol Leprol. 2008;74(4):322-326.
16. Wolff K, Johnson R, Suurmond R. Leprosy. In: Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. Chicago, IL: McGraw-Hill Professional; 2005:655-661.
17. Centers for Disease Control and Prevention. Summary of notifiable diseases—United States, 2007. MMWR Morb Mortal Wkly Rep. 2009;56 (53):3, 10, 22.
18. American Academy of Dermatology. Vitiligo. www.aad.org/public/publications/pamphlets/com mon_vitilgo.html. Accessed November 17, 2009.
19. American Academy of Dermatology. Granuloma annulare. www.aad.org/public/publica tions/pamphlets/common_granuloma.html. Accessed November 17, 2009.
20. American Academy of Dermatology. Lupus and the skin. www.aad.org/public/publications/pamphlets/common_lupus.html. Accessed November 17, 2009.
21. National Hansen’s Disease Program. Recommended treatment regimens. www.hrsa.gov/han sens/clinical/regimens.htm. Accessed November 17, 2009.
22. Chen J, Raymond K. Roles of rifampicin in drug-drug interactions: underlying molecular mechanisms involving the nuclear pregnane X receptor. Ann Clin Microbiol Antimicrob. 2006 Feb 15;5:3.
23. Moet FJ, Pahan D, Oskam L, Richardus JH; COLEP Study Group. Effectiveness of single dose rifampicin in preventing leprosy in close contacts of patients with newly diagnosed leprosy: cluster randomised controlled trial. BMJ. 2008;336 (7647):761-764.
24. World Health Organization. WHO model prescribing information: drugs used in leprosy. http://apps.who.int/medicinedocs/fr/d/Jh2988e/ 14.2.html. Accessed November 17, 2009.
25. Bruce S, Schroeder TL, Ellner K, et al. Armadillo exposure and Hansen’s disease: an epidemiologic survey in southern Texas. J Am Acad Dermatol. 2000;43(2 pt 1):223-228.
26. Clark BM, Murray CK, Horvath LL, et al. Case-control study of armadillo contact and Hansen’s disease. Am J Trop Med Hyg. 2008;78(6):962-967.
1. World Health Organization. Global strategy for further reducing the leprosy burden and sustaining leprosy control activities 2006-2010: operational guidelines. www.who.int/lep/resources/SEAGLP20062.pdf. Accessed November 17, 2009.
2. World Health Organization Communicable -Diseases Department. Leprosy: frequently as-ked questions. www.searo.who.int/EN/Section10/Section373_11716.htm. Accessed November 17, 2009.
3. National Hansen’s Disease (Leprosy) Program. www.hrsa.gov/hansens. Accessed November 23, 2009.
4. Health Resources and Services Administration, US Department of Health and Human Services, National Hansen’s Disease Program. A summary of Hansen’s disease in the United States—2006. ftp://ftp.hrsa.gov/hansens/2006RegistryReport.pdf. Accessed November 17, 2009.
5. Boggild AK, Correia JD, Keystone JS, Kain KC. Leprosy in Toronto: an analysis of 184 imported cases. CMAJ. 2004;170(1):55-59.
6. Cheesbrough M. Bacterial pathogens. In: District Laboratory Practice in Tropical Countries. Part 2. 2nd ed. Cambridge, UK: Cambridge University Press. 2006;157-234.
7. Sasaki S, Takeshita F, Okuda K, Ishii N. Mycobacterium leprae and leprosy: a compendium. Microbiol Immunol. 2001;45(11):729–736.
8. Job CK, Jayakumar J, Kearney M, Gillis TP. Transmission of leprosy: a study of skin and nasal secretions of household contacts of leprosy patients using PCR. Am J Trop Med Hyg. 2008; 78(3):518-521.
9. Girdhar BK. Skin to skin transmission of leprosy. Indian J Dermatol Venereol Leprol. 2005;71(4):223-225.
10. National Leprosy Eradication Programme, Directorate General of Health Services (Government of India). Differential diagnosis for leprosy (2009). http://nlep.nic.in/pdf/Annex-%20III% 20Differential%20diagnosis.pdf. September 14, 2009.
11. Kim EC. Hansen disease: differential diagnoses and workup (2006). http://emedicine.medscape .com/article/1213853-diagnosis. Accessed November 17, 2009.
12. James WD, Berger TG, Elston DM. Hansen’s disease. In: Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Philadelphia, PA: Saunders. 2005;343-366.
13. Leprosy. In: Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 2nd ed. Philadelphia, PA: WB Saunders; 2005.
14. National Hansen’s Disease Program. Skin biopsy in the diagnosis of Hansen’s disease. www.hrsa.gov/hansens/clinical/diagnostics/biopsy.htm. Accessed November 17, 2009.
15. Bhushan P, Sardana K, Koranne RV, et al. Diagnosing multibacillary leprosy: a comparative evaluation of diagnostic accuracy of slit-skin smear, bacterial index of granuloma and WHO operational classification. Indian J Dermatol Venereol Leprol. 2008;74(4):322-326.
16. Wolff K, Johnson R, Suurmond R. Leprosy. In: Fitzpatrick’s Color Atlas and Synopsis of Clinical Dermatology. Chicago, IL: McGraw-Hill Professional; 2005:655-661.
17. Centers for Disease Control and Prevention. Summary of notifiable diseases—United States, 2007. MMWR Morb Mortal Wkly Rep. 2009;56 (53):3, 10, 22.
18. American Academy of Dermatology. Vitiligo. www.aad.org/public/publications/pamphlets/com mon_vitilgo.html. Accessed November 17, 2009.
19. American Academy of Dermatology. Granuloma annulare. www.aad.org/public/publica tions/pamphlets/common_granuloma.html. Accessed November 17, 2009.
20. American Academy of Dermatology. Lupus and the skin. www.aad.org/public/publications/pamphlets/common_lupus.html. Accessed November 17, 2009.
21. National Hansen’s Disease Program. Recommended treatment regimens. www.hrsa.gov/han sens/clinical/regimens.htm. Accessed November 17, 2009.
22. Chen J, Raymond K. Roles of rifampicin in drug-drug interactions: underlying molecular mechanisms involving the nuclear pregnane X receptor. Ann Clin Microbiol Antimicrob. 2006 Feb 15;5:3.
23. Moet FJ, Pahan D, Oskam L, Richardus JH; COLEP Study Group. Effectiveness of single dose rifampicin in preventing leprosy in close contacts of patients with newly diagnosed leprosy: cluster randomised controlled trial. BMJ. 2008;336 (7647):761-764.
24. World Health Organization. WHO model prescribing information: drugs used in leprosy. http://apps.who.int/medicinedocs/fr/d/Jh2988e/ 14.2.html. Accessed November 17, 2009.
25. Bruce S, Schroeder TL, Ellner K, et al. Armadillo exposure and Hansen’s disease: an epidemiologic survey in southern Texas. J Am Acad Dermatol. 2000;43(2 pt 1):223-228.
26. Clark BM, Murray CK, Horvath LL, et al. Case-control study of armadillo contact and Hansen’s disease. Am J Trop Med Hyg. 2008;78(6):962-967.