User login
Emergent Diagnosis of the Unknown Rash: An Algorithmic Approach
Cementless Femoral Fixation in Total Hip Arthroplasty
Laparoscopic myomectomy: 8 pearls
Myomectomy is the surgery of choice for women who have symptomatic fibroids and who wish to retain their uterus. And laparoscopic myomectomy is preferable to the abdominal approach in many ways, offering: 1-4
- faster recovery
- a shorter hospital stay
- diminished blood loss
- decreased adhesion formation
- a comparable or higher rate of pregnancy.
Nevertheless, laparoscopic myomectomy is a technically challenging procedure with surgeon-specific limitations. The biggest challenge: appropriately suturing the hysterotomy site.
In this article, I share my experience with laparoscopic myomectomy and offer 8 pearls that may contribute to a successful outcome.
1. Don't settle on laparoscopy prematurely
Given its advantages over the abdominal route, laparoscopic myomectomy should be the preferred approach in the treatment of symptomatic uterine fibroids ( FIGURE 1 ). However, not all patients are appropriate candidates for laparoscopy. Several guidelines have recommended a maximum number and size of fibroids for laparoscopic removal, but practice varies widely, and experienced surgeons successfully take on cases that are well beyond the limits set by most published guidelines.5-7
At our institution, we do not have firm guidelines in place for the number and size of fibroids that can be removed laparoscopically. Other variables enter into decision-making and counseling, among them any medical comorbidity or history of uterine surgery the patient may have, as well as her desires in regard to childbearing and uterine retention.
Hysterectomy may be the most straightforward option for women who have symptomatic fibroids and who have completed childbearing. However, myomectomy is also appropriate as long as the patient is aware of the risk that fibroids may recur and the potential for further surgery. When the patient is in her late 40s or early 50s, the likelihood of fibroid recurrence may be lower than it is in the general population.
In my practice, submucosal and intracavitary fibroids smaller than 4 cm and more than 5 mm away from the uterine serosa are generally removed hysteroscopically, an approach beyond the scope of this article. In women who have completed childbearing but who wish to conserve the uterus, we deliberately enter the uterine cavity laparoscopically because this strategy allows for efficient removal of submucosal and intracavitary fibroids.
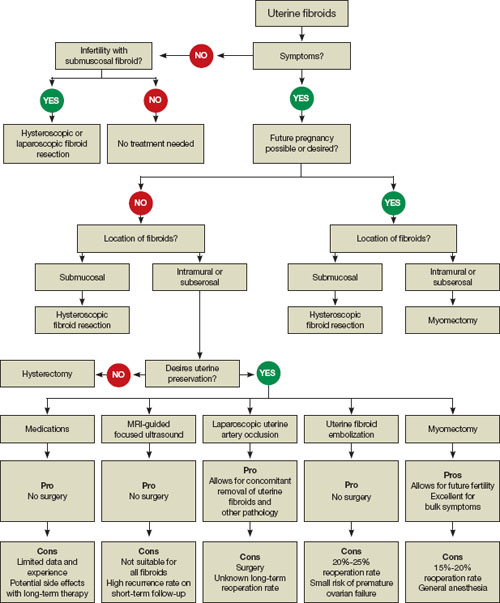
FIGURE 1 When and how to treat uterine fibroids
2. Estimate the duration of surgery
When the patient has fibroids that are intramural or subserosal, our general rule of thumb is to determine her suitability for laparoscopic myomectomy, based on the estimated duration of the operation. A surgeon's ability to calculate the length of the operation for a particular patient increases with experience.
We tend to recommend the laparoscopic approach when the procedure is expected to take less than 3 hours to complete. More than 95% of our patients fall into this category.
When we anticipate a prolonged operating time, we discuss the option of hand-assisted laparoscopic myomectomy. This approach involves two or three 5-mm trocar punctures high on the abdomen in conjunction with a suprapubic incision, 6 to 7 cm in length with a hand port in place. Prospective studies have demonstrated a significantly longer recovery with minilaparotomy than with laparoscopy, but these trials compared uteri of similar size.4,8 We expect the laparoscopic approach to confer fewer advantages when operative time is prolonged significantly.
In our practice, we consider one or more of the following conditions appropriate for hand-assisted laparoscopic myomectomy:
- a very large uterus (i.e., heavier than 1,500 to 2,000 g). In these cases, operating times can be excessive because of the need for extensive suturing and morcellation, and bleeding may increase as a result
- more than 20 fibroids on magnetic resonance imaging (MRI). It can be a challenge to locate all of the fibroids; multiple uterine incisions may be necessary
- a medical comorbidity that renders the patient unable to tolerate prolonged anesthesia. For example, we operated on a patient who had Ehlers-Danlos syndrome and who needed to avoid a prolonged operation due to fragile bones and joint laxity.
Of necessity, these guidelines will vary from practice to practice, and gynecologic surgeons who are just beginning to perform laparoscopic myomectomy should not include multiple fibroids or a large uterus among their initial cases. Instead, perform the first few cases in patients who have not had abdominal surgery and who have a symptomatic intramural or subserosal fibroid that is close to the uterine fundus and no larger than 6 cm in diameter.
3. Consider preoperative MRI
Preoperative imaging greatly supplements the clinical examination and facilitates identification of the number, location, and characteristics of the fibroids. Pelvic ultrasonography (US) is appropriate for most patients. We prefer preoperative MRI of the pelvis in the following scenarios:
- uterus larger than 12 weeks (280 g) on clinical examination
- identification of multiple fibroids via US
- history suggestive of adenomyosis.
MRI facilitates preoperative planning by accurately delineating the size and location of the fibroids, and by distinguishing between an adenomyoma and fibroid in most cases.9 For an example of its utility, see "How MRI can guide treatment: 3 cases."
4. Preoperative medical therapy may be indicated
When given preoperatively, gonadotropin-releasing hormone (GnRH) agonists have been shown to reduce blood loss and shorten operative time. The exception: cases involving hypoechoic fibroids, because the cleavage plane may be more difficult to identify, prolonging operative time.10
We generally prefer to use a GnRH agonist in two clinical scenarios: 1) anemia and 2) a uterus that extends above the umbilicus. In the second scenario, the GnRH agonist helps reduce the uterus to a more manageable size.
Aromatase inhibitors show great promise as preoperative agents because there is no initial flare. In addition, because fibroids have a higher concentration of aromatase activity than the surrounding myometrium, a low dosage of an aromatase inhibitor is effective and does not cause significant menopausal symptoms.
A recent comparative study found that fibroid shrinkage was greater after 3 months of letrozole (2.5 mg/day) than after use of a GnRH agonist.11 Total myoma volume decreased by 45.6% in the letrozole group, compared with 33.2% in the group that received a GnRH agonist (P =.02).11
Aromatase inhibitors have also been successfully used during the initial period of GnRH agonist therapy to prevent the symptoms of flare.12 However, because clinical experience is limited, the long-term efficacy and safety of aromatase inhibitors in premenopausal women is unknown.
Findings A 40-year-old nulliparous woman seeks treatment for menometrorrhagia and dysmenorrhea but wants to conserve her uterus. MRI reveals a 4.5-cm submucosal fibroid (arrow) that extends all the way to the uterine serosa, with no evidence of adenomyosis. Her thyroid-stimulating hormone (TSH) level is normal, as is an endometrial biopsy.

CASE 1
Outcome We decide to proceed with laparoscopic myomectomy because a hysteroscopic approach would carry a risk of uterine rupture.
Findings A 36-year-old nulliparous woman complains of significant "bulk" symptoms (heaviness, urinary frequency, and abdominal bloating). She has a visible mass that extends four finger-breadths above the umbilicus. Pelvic MRI reveals multiple intramural fibroids in a uterus estimated to weigh roughly 2,850 g. The patient is given a 3-month course of a GnRH agonist.
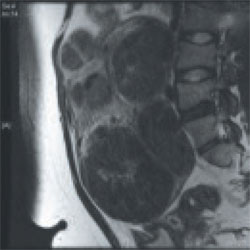
CASE 2
Outcome After treatment with the GnRH agonist, the patient undergoes hand-assisted, laparoscopic, multiple myomectomy. She is discharged home the following day and resumes normal activities within two weeks.
Findings A 32-year-old nulliparous patient seeks treatment for menomenorrhagia and symptoms of bulk and expresses a desire for uterine conservation. Pelvic MRI reveals two distinct intramural fibroids, 6 cm and 9 cm in size.

CASE 3
Outcome The patient undergoes laparoscopic myomectomy without preoperative treatment with a GnRH agonist and is discharged home the same day without postoperative complication. (Although the uterus had two large fibroids, we did not use a GnRH agonist because the uterus was well below the belly button.)
5. Use careful surgical technique
Pay attention to set-up, initial entry
Although we lack definitive data on the practical utility of preoperative, intravenous (IV) antibiotics, we administer cefazolin prophylactically, switching to IV clindamycin if there is a documented allergy to penicillin.
In addition, a uterine manipulator is helpful when the patient has a small or medium-sized uterus. A variety of manipulators are available, but we generally use the VCare manipulator (ConMed Corp) because it is easy to insert and provides excellent uterine mobility.
Initial entry is at the umbilicus, unless the uterus extends above the umbilicus, in which case we enter in the left upper quadrant. We generally place the camera through the umbilical port and use two parallel operative ports on the left side of the patient, where the primary surgeon stands. A detailed description of our laparoscopic entry technique was published recently.13
Place the first trocar two finger-breadths medial and superior to the iliac spine and the second trocar 8 cm cephalad to the first port ( FIGURE 2 ). In a large uterus, trocars may have to be placed higher on the abdomen. A third operative port may be added on the right side, if it is needed.
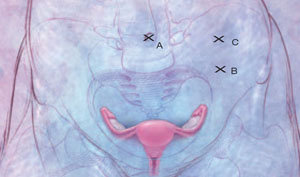
FIGURE 2 Laparoscopic port placement
Place the camera through the umbilical port (A) and operate through two additional ports on the left-hand side of the patient, where the primary surgeon stands. Place the first operative port two finger-breadths medial and superior to the iliac spine (B) and the second port 8 cm cephalad to the first (C).
Incise the uterus
Infiltrate the uterus with dilute vasopressin (20 U in 60 mL of saline), taking care to administer no more than 10 U at a time to minimize the potential for cardiovascular side effects such as bradycardia and hypertension.14 In the past, we periodically encountered episodes of bradycardia when we used 20 U in 40 mL of saline, but we have not had that problem since we changed to a more dilute vasopressin and used no more than 10 U at a time. It may be that an even smaller amount of vasopressin is just as effective, but we do not yet have sufficient data on myomectomy to determine whether that is the case.
Inject the vasopressin subserosally and along the planned hysterotomy. The fibroid itself contains no blood vessels, but the blood supply to the fibroid generally assumes a coronal pattern around it.15 Therefore, it is important to inject the vasopressin into the correct subserosal plane.
We prefer to make a horizontal hysterotomy using the Harmonic Scalpel (Ethicon Endo-Surgery), but other energy sources, such as a monopolar hook or bipolar spatula, are also appropriate.
We choose a horizontal incision because of the ipsilateral port placement we use for suturing. Surgeons who use a midline or contralateral port for suturing may find it easier to repair a vertical hysterotomy. The pattern of blood vessels along the uterus is heterogeneous and variable, and there is no evidence that blood loss or other outcomes are affected by the direction of the uterine incision.15
Once the uterus has been incised, it is important to work efficiently because bleeding will probably continue until the hysterotomy site is completely closed.
Extract the fibroid ("rock and roll")
Extract the fibroid from the uterus by applying generous traction using a tenaculum, and by applying counter-traction using an atraumatic grasper and the Harmonic Scalpel, as needed. We try to limit the use of thermal energy during this step.
The most important aspect of fibroid extraction is ensuring entry into the correct plane. Appropriate entry makes it possible to remove most fibroids without the need for sharp or thermal dissection.
If you are not sure whether you have entered the correct plane, it is better to cut into the fibroid rather than remain too shallow. If you do not enter all the way into the correct plane, you run the risk of pulling and tearing uterine muscle fibers and causing bleeding.
We describe the technique of fibroid extraction as "rock and roll" because it is generally easier to grab the fibroid near the hysterotomy and roll it out rather than pull on the portion of the fibroid that protrudes from the uterus (see the image, for example).
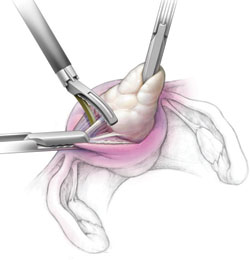
During laparoscopic myomectomy, extract the fibroid by applying generous traction with the tenaculum and counter-traction with an atraumatic grasper and ultrasonic shears. Once you have entered the correct surgical plane, grasp the fibroid near the hysterotomy and simply roll it out of the uterus.
Pregnancy outcomes after laparoscopic myomectomy are generally favorable, with a pregnancy rate that is comparable to or even higher than the rate associated with abdominal myomectomy.1-4
Uneventful vaginal deliveries following laparoscopic myomectomy have been reported in several case series, but so have a number of cases of gravid uterine rupture.5 In a recent study involving 2,050 laparoscopic myomectomies, investigators tracked 386 post-myomectomy pregnancies, 309 deliveries in all, of which 68 were vaginal deliveries.25 It found one case of uterine rupture documented at 33 weeks in a woman who had undergone adenomyomectomy.25
Overall, the literature suggests that uterine rupture after laparoscopic myomectomy is a rare event, occurring in fewer than 1% of pregnancies. Some surgeons use a somewhat arbitrary rule of thumb requiring cesarean delivery if the uterine cavity is entered at myomectomy. This practice is not based on hard evidence, but it does make intuitive sense. If the uterine cavity is entered during myomectomy, it creates a transmural defect that may be more difficult to repair and could carry a higher risk of rupture.
Uterine rupture has also occurred several years after removal of a pedunculated fibroid, suggesting that the use of electrosurgery may weaken the uterine muscle and increase the risk of rupture.
In general—and regardless of the depth of the hysterotomy—it is advisable to counsel patients who have undergone laparoscopic myomectomy that the uterus heals with a scar that may be slightly weaker than the normal myometrium and that elective cesarean delivery may be the optimal strategy. However, a trial of labor is a reasonable alternative, provided the patient receives careful surveillance in a hospital setting.
Close the myometrium
In the event of inadvertent entry into the uterine cavity, close the endometrial defect using running 2-0 polyglactin 910 suture, taking care to avoid suture entry into the uterine cavity. Tie this suture using intracorporeal knot-tying.
Close the hysterotomy in layers using 14 × 14 cm bidirectional barbed 0 PDO suture on a 36-mm, half-circle, taper-point needle. If the hysterotomy is longer than 8 cm, we prefer to use 24 × 24 cm suture.
Tack the first needle into the opposite anterior abdominal wall to help prevent tangling of the suture. Close the deepest layer using the first needle and the more superficial layer and serosa using the second needle. Then cut the needles. Because of the uniform tension and bidirectional nature of the barbed suture, no knots are required.
We began using Quill bidirectional barbed suture (Angiotech) in March 2008.16 Since then, we have completed almost 300 laparoscopic cases using this material, including approximately 100 laparoscopic myomectomies. We compiled data on our first year of experience with this material ( TABLE ), during which we had no major complications related to use of the suture, no conversions to laparotomy, and no returns to the OR to address bleeding or complications arising from the use of bidirectional barbed suture.
The original version of barbed suture included a 6-cm segment of regular, smooth suture. If suturing extends to include this segment, apply a LapraTy clip (Ethicon). This use of LapraTy is off-label because the clip is intended for use with Vicryl 2/0, 3/0, and 4/0 (manufacturer). Nevertheless, our clinical experience with this approach has been favorable.16,17
When closing the uterus, use as many layers as necessary to eliminate all dead space within the myometrium. Sometimes, as many as five layers are needed to close a deep myometrial defect, but a two- or three-layer closure is most common.
TABLE
1 year of experience with laparoscopic myomectomy using bidirectional barbed suture
| Variable | Mean ± standard deviation |
|---|---|
| Duration of surgery (min) | 125.47 ± 55.30 |
| Estimated blood loss (mL) | 158.68 ± 252.35 |
| Number of fibroids removed | 4.01 ± 4.21 |
| Weight of fibroids (g) | 252.07 ± 196.43 |
| Hospital stay (days) | 0.73 ± 0.36 |
| Data represent the author's experience with 55 consecutive laparoscopic myomectomy cases between March 2008 and March 2009. | |
Ward off adhesions
We generally cover the hysterotomy site with an adhesion barrier such as Interceed (Gynecare). Although no adhesion barrier is ideal, Interceed has proved to be effective in this clinical scenario.18 Make sure that the hysterotomy site is completely hemostatic at the time the barrier is applied.
Morcellate with caution
We generally use a 12- to 15-mm electronic morcellator for fibroid removal. Morcellation through the umbilicus is often feasible and prevents the need for a large peripheral incision, which may be less cosmetically pleasing to the patient and potentially more painful than a 15-mm umbilical incision.
We place a 5-mm optic through a peripheral port on the ipsilateral side of the surgeon because it allows the surgeon to operate away from the camera, causing less disorientation. Morcellation is inherently dangerous because of the risk of injury to internal organs such as bowel and blood vessels. The best way to prevent such injuries is to:
- keep the rotating blade in view at all times
- stay on the surface of the fibroid during morcellation (avoid coring)
- hold the morcellator steady during morcellation, i.e., do not move it forward while it is active.
6. NSAIDs and few restrictions are the norm postoperatively
We discharge almost all of our patients postoperatively on the day of laparoscopic myomectomy. Patients who have several medical comorbidities may need to stay overnight, however. We have not yet had to readmit a patient after a day-of-procedure discharge, and patients generally recover fairly rapidly.
We are prospectively evaluating our patients' return to daily activities. Most have resumed normal preoperative activities within 10 days. We recommend the scheduled use of nonsteroidal anti-inflammatory medications (NSAIDs), such as 800 mg of ibuprofen every 6 to 8 hours, for the first 3 to 5 days after surgery.
We encourage patients to remain active after surgery, with no weight-lifting restrictions. Instead, we instruct patients to live by the rule, "If it hurts, don't do it." We do prescribe narcotics, but we instruct patients to limit their use as much as possible. We give IV ketorolac perioperatively.
7. Uterine artery occlusion may prevent recurrence
One treatment option that we occasionally use in conjunction with laparoscopic myomectomy is laparoscopic uterine artery occlusion (LUAO) because it can significantly reduce the recurrence rate of uterine fibroids.19 LUAO is especially valuable in the setting of multiple, small uterine fibroids ("bag of marbles") and other cases where it is unlikely that all fibroids will be removed during myomectomy.
At present, we perform LUAO only in patients who have completed childbearing, although early evidence suggests that pregnancy may be relatively safe after uterine artery occlusion.19,20 More data and longer follow-up are required before LUAO should be offered to all women of reproductive age.
8. Consider single-incision laparoscopic myomectomy
This approach has been touted as offering an improved cosmetic outcome and, possibly, less postoperative pain, although these potential benefits have yet to be demonstrated in a well-designed prospective trial.21
We have performed three cases of single-incision myomectomy for an intramural fibroid and have demonstrated this approach to be feasible (article in press). Barbed suture is especially valuable in single-incision surgery because intracorporeal knot-tying can be more challenging when there is only one incision. However, limitations include:
- a lack of triangulation
- instrument crowding at the umbilicus
- difficulty suturing using traditional or barbed suture.
Proper documentation is key
Current Procedural Terminology (CPT) offers two coding options when you've performed a laparoscopic myomectomy:
- 58545 (laparoscopy, surgical, myomectomy, excision; 1 to 4 intramural myomas with total weight of 250 g or less and/or removal of surface myomas)
- 58546 (laparoscopy, surgical, myomectomy, excision; 5 or more intramural myomas and/or intramural myomas with total weight greater than 250 g).
Which code you submit can, of course, make a difference in how much you're reimbursed: 58545 carries 24.21 relative value units (RVU); 58546, 30.59 RVU. The documentation that you present will, therefore, be key in getting paid for the work you've performed.
First, look at the description of 58545. You have two documentation options:
- You removed between one and four intramural myomas (International Classification of Diseases [ICD-9] 218.1; intramural leiomyoma of uterus) whose total weight was =250 g
- You encountered surface myomas (ICD9 218.2; subserous leiomyoma of uterus) and removed all of them, weight aside.
Second, to bill the higher-paying code (58546), you must clearly document removal of intramural myomas only. Again, your work must meet either of two criteria:
- Total weight of all intramural myomas removed is >250 g
- You removed five or more intramural myomas.
You can determine the total weight of the excised tissue 1) in the operating room, if a scale is available, or 2) from the pathology report. A caution: The tissue that you've removed will shrink after it arrives in pathology, and this shrinkage may make a difference when, for example, fewer than five myomas were removed and their total weight is close to 250 g.
Last, estimating the weight of myomas by ultrasonography before surgery is not considered acceptable documentation of weight by most payers.—Melanie Witt, RN, CPC, COBGC, MA
Ms. Witt is an independent coding and documentation consultant and former program manager, department of coding and nomenclature, American College of Obstetricians and Gynecologists.
Therefore, it may be challenging to apply single-port surgery to more complex pathology, such as very large fibroids and severe pelvic adhesive disease.
Single-incision surgery may offer marginal cosmetic benefit for some patients. When we surveyed our patients informally, however, most of them expressed satisfaction with the cosmetic appearance of peripheral laparoscopic port incisions.
Another potential limitation of single-incision surgery is the cost associated with disposable, articulating instruments and single-port access devices. Although robotic surgery is a feasible approach to both multi-port22,23 and single-port surgery,24 prospective data are lacking, and cost remains an issue. It is possible that future developments in robotic surgery may facilitate suture-intensive, single-incision cases such as myomectomy and sacrocolpopexy. Well-designed prospective trials are urgently needed.
1. Seracchioli R, Rossi S, Govoni F, et al. Fertility and obstetric outcome after laparoscopic myomectomy of large myomata: a randomized comparison with abdominal myomectomy. Hum Reprod. 2000;15(12):2663-2668.
2. Rossetti A, Sizzi O, Soranna L, Cucinelli F, Mancuso S, Lanzone A. Long-term results of laparoscopic myomectomy: recurrence rate in comparison with abdominal myomectomy. Hum Reprod. 2001;16(4):770-774.
3. Stringer NH, Walker JC, Meyer PM. Comparison of 49 laparoscopic myomectomies with 49 open myomectomies. J Am Assoc Gynecol Laparosc. 1997;4(4):457-464.
4. Palomba S, Zupi E, Falbo A, et al. A multicenter randomized, controlled study comparing laparoscopic versus minilaparotomic myomectomy: reproductive outcomes. Fertil Steril. 2007;88(4):933-941.
5. Hurst BS, Matthews ML, Marshburn PB. Laparoscopic myomectomy for symptomatic uterine myomas. Fertil Steril. 2005;83:1-23.
6. Agdi M, Tulandi T. Endoscopic management of uterine fibroids. Best Pract Res Clin Obstet Gynaecol. 2008;22(4):707-716.
7. Sinha R, Hegde A, Mahajan C, Dubey N, Sundaram M. Laparoscopic myomectomy: do size, number, and location of the myomas form limiting factors for laparoscopic myomectomy? J Minim Invasive Gynecol. 2008;15(3):292-300.
8. Malzoni M, Tinelli R, Cosentino F, et al. Laparoscopy versus minilaparotomy in women with symptomatic uterine myomas: short-term and fertility results [published online ahead of print March 12, 2009; corrected proof March 16, 2009]. Fertil Steril. 2009;doi:10.1016/j.fertnstert.2008.12.127.
9. Dudiak CM, Turner DA, Patel SK, Archie JT, Silver B, Norusis M. Uterine leiomyomas in the infertile patient: preoperative localization with MR imaging versus US and hysterosalpingography. Radiology. 1988;167(3):627-630.
10. Zullo F, Pellicano M, De Stefano R, Zupi E, Mastrantonio P. A prospective randomized study to evaluate leuprolide acetate treatment before laparoscopic myomectomy: efficacy and ultrasonographic predictors. Am J Obstet Gynecol. 1998;178(1 Pt 1):108-112.
11. Parsanezhad ME, Azmoon M, Alborzi S, et al. A randomized, controlled clinical trial comparing the effects of aromatase inhibitor (letrozole) and gonadotropin-releasing hormone agonist (triptorelin) on uterine leiomyoma volume and hormonal status. Fertil Steril. 2010;93(1):192-198.
12. Bedaiwy MA, Mousa NA, Casper RF. Aromatase inhibitors prevent the estrogen rise associated with the flare effect of gonadotropins in patients treated with GnRH agonists. Fertil Steril. 2009;91(suppl 4):1574-1577.
13. Vellinga TT, De Alwis S, Suzuki Y, Einarsson JI. Laparoscopic entry: the modified Alwis method and more. Rev Obstet Gynecol. 2009;2(3):193-198.
14. Frishman G. Vasopressin: if some is good, is more better? Obstet Gynecol. 2009;113(2 Pt 2):476-477.
15. Discepola F, Valenti DA, Reinhold C, Tulandi T. Analysis of arterial blood vessels surrounding the myoma: relevance to myomectomy. Obstet Gynecol. 2007;110(6):1301-1303.
16. Greenberg JA, Einarsson JI. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy. J Minim Invasive Gynecol. 2008;15(5):621-623.
17. Einarsson JI, Vellinga TT, Twijnstra AR, Suzuki Y, Greenberg JA. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy; an evaluation of safety and clinical outcomes [abstract]. J Minim Invasive Gynecol. 2009;16(6)(suppl 1):S28-S29.Abstract 95.
18. Mais V, Ajossa S, Piras B, Guerriero S, Marongiu D, Melis GB. Prevention of de-novo adhesion formation after laparoscopic myomectomy: a randomized trial to evaluate the effectiveness of an oxidized regenerated cellulose absorbable barrier. Hum Reprod. 1995;10(12):3133-3135.
19. Alborzi S, Ghannadan E, Alborzi S, Alborzi M. A comparison of combined laparoscopic uterine artery ligation and myomectomy versus laparoscopic myomectomy in treatment of symptomatic myoma. Fertil Steril. 2009;92(2):742-747.
20. Holub Z, Mara M, Kuzel D, Jabor A, Maskova J, Eim J. Pregnancy outcomes after uterine artery occlusion: prospective multicentric study. Fertil Steril. 2008;90(5):1886-1891.
21. Curcillo PG, King SA, Podolsky ER, Rottman SJ. Single Port Access (SPA) minimal access surgery through a single incision. Surg Technol Int. 2009;18:19-25.
22. Nezhat C, Lavie O, Hsu S, Watson J, Barnett O, Lemyre M. Robotic-assisted laparoscopic myomectomy compared with standard laparoscopic myomectomy—a retrospective matched control study. Fertil Steril. 2009;91(2):556-559.
23. Bedient CE, Magrina JF, Noble BN, Kho RM. Comparison of robotic and laparoscopic myomectomy. Am J Obstet Gynecol. 2009;201(6):566.e1-5.
24. Escobar PF, Fader AN, Paraiso MF, Kaouk JH, Falcone T. Robotic-assisted laparoendoscopic single-site surgery in gynecology: initial report and technique. J Minim Invasive Gynecol. 2009;16(5):589-591.
25. Sizzi O, Rossetti A, Malzoni M, et al. Italian multicenter study on complications of laparoscopic myomectomy. J Minim Invasive Gynecol. 2007;14(4):453-462.
Myomectomy is the surgery of choice for women who have symptomatic fibroids and who wish to retain their uterus. And laparoscopic myomectomy is preferable to the abdominal approach in many ways, offering: 1-4
- faster recovery
- a shorter hospital stay
- diminished blood loss
- decreased adhesion formation
- a comparable or higher rate of pregnancy.
Nevertheless, laparoscopic myomectomy is a technically challenging procedure with surgeon-specific limitations. The biggest challenge: appropriately suturing the hysterotomy site.
In this article, I share my experience with laparoscopic myomectomy and offer 8 pearls that may contribute to a successful outcome.
1. Don't settle on laparoscopy prematurely
Given its advantages over the abdominal route, laparoscopic myomectomy should be the preferred approach in the treatment of symptomatic uterine fibroids ( FIGURE 1 ). However, not all patients are appropriate candidates for laparoscopy. Several guidelines have recommended a maximum number and size of fibroids for laparoscopic removal, but practice varies widely, and experienced surgeons successfully take on cases that are well beyond the limits set by most published guidelines.5-7
At our institution, we do not have firm guidelines in place for the number and size of fibroids that can be removed laparoscopically. Other variables enter into decision-making and counseling, among them any medical comorbidity or history of uterine surgery the patient may have, as well as her desires in regard to childbearing and uterine retention.
Hysterectomy may be the most straightforward option for women who have symptomatic fibroids and who have completed childbearing. However, myomectomy is also appropriate as long as the patient is aware of the risk that fibroids may recur and the potential for further surgery. When the patient is in her late 40s or early 50s, the likelihood of fibroid recurrence may be lower than it is in the general population.
In my practice, submucosal and intracavitary fibroids smaller than 4 cm and more than 5 mm away from the uterine serosa are generally removed hysteroscopically, an approach beyond the scope of this article. In women who have completed childbearing but who wish to conserve the uterus, we deliberately enter the uterine cavity laparoscopically because this strategy allows for efficient removal of submucosal and intracavitary fibroids.

FIGURE 1 When and how to treat uterine fibroids
2. Estimate the duration of surgery
When the patient has fibroids that are intramural or subserosal, our general rule of thumb is to determine her suitability for laparoscopic myomectomy, based on the estimated duration of the operation. A surgeon's ability to calculate the length of the operation for a particular patient increases with experience.
We tend to recommend the laparoscopic approach when the procedure is expected to take less than 3 hours to complete. More than 95% of our patients fall into this category.
When we anticipate a prolonged operating time, we discuss the option of hand-assisted laparoscopic myomectomy. This approach involves two or three 5-mm trocar punctures high on the abdomen in conjunction with a suprapubic incision, 6 to 7 cm in length with a hand port in place. Prospective studies have demonstrated a significantly longer recovery with minilaparotomy than with laparoscopy, but these trials compared uteri of similar size.4,8 We expect the laparoscopic approach to confer fewer advantages when operative time is prolonged significantly.
In our practice, we consider one or more of the following conditions appropriate for hand-assisted laparoscopic myomectomy:
- a very large uterus (i.e., heavier than 1,500 to 2,000 g). In these cases, operating times can be excessive because of the need for extensive suturing and morcellation, and bleeding may increase as a result
- more than 20 fibroids on magnetic resonance imaging (MRI). It can be a challenge to locate all of the fibroids; multiple uterine incisions may be necessary
- a medical comorbidity that renders the patient unable to tolerate prolonged anesthesia. For example, we operated on a patient who had Ehlers-Danlos syndrome and who needed to avoid a prolonged operation due to fragile bones and joint laxity.
Of necessity, these guidelines will vary from practice to practice, and gynecologic surgeons who are just beginning to perform laparoscopic myomectomy should not include multiple fibroids or a large uterus among their initial cases. Instead, perform the first few cases in patients who have not had abdominal surgery and who have a symptomatic intramural or subserosal fibroid that is close to the uterine fundus and no larger than 6 cm in diameter.
3. Consider preoperative MRI
Preoperative imaging greatly supplements the clinical examination and facilitates identification of the number, location, and characteristics of the fibroids. Pelvic ultrasonography (US) is appropriate for most patients. We prefer preoperative MRI of the pelvis in the following scenarios:
- uterus larger than 12 weeks (280 g) on clinical examination
- identification of multiple fibroids via US
- history suggestive of adenomyosis.
MRI facilitates preoperative planning by accurately delineating the size and location of the fibroids, and by distinguishing between an adenomyoma and fibroid in most cases.9 For an example of its utility, see "How MRI can guide treatment: 3 cases."
4. Preoperative medical therapy may be indicated
When given preoperatively, gonadotropin-releasing hormone (GnRH) agonists have been shown to reduce blood loss and shorten operative time. The exception: cases involving hypoechoic fibroids, because the cleavage plane may be more difficult to identify, prolonging operative time.10
We generally prefer to use a GnRH agonist in two clinical scenarios: 1) anemia and 2) a uterus that extends above the umbilicus. In the second scenario, the GnRH agonist helps reduce the uterus to a more manageable size.
Aromatase inhibitors show great promise as preoperative agents because there is no initial flare. In addition, because fibroids have a higher concentration of aromatase activity than the surrounding myometrium, a low dosage of an aromatase inhibitor is effective and does not cause significant menopausal symptoms.
A recent comparative study found that fibroid shrinkage was greater after 3 months of letrozole (2.5 mg/day) than after use of a GnRH agonist.11 Total myoma volume decreased by 45.6% in the letrozole group, compared with 33.2% in the group that received a GnRH agonist (P =.02).11
Aromatase inhibitors have also been successfully used during the initial period of GnRH agonist therapy to prevent the symptoms of flare.12 However, because clinical experience is limited, the long-term efficacy and safety of aromatase inhibitors in premenopausal women is unknown.
Findings A 40-year-old nulliparous woman seeks treatment for menometrorrhagia and dysmenorrhea but wants to conserve her uterus. MRI reveals a 4.5-cm submucosal fibroid (arrow) that extends all the way to the uterine serosa, with no evidence of adenomyosis. Her thyroid-stimulating hormone (TSH) level is normal, as is an endometrial biopsy.

CASE 1
Outcome We decide to proceed with laparoscopic myomectomy because a hysteroscopic approach would carry a risk of uterine rupture.
Findings A 36-year-old nulliparous woman complains of significant "bulk" symptoms (heaviness, urinary frequency, and abdominal bloating). She has a visible mass that extends four finger-breadths above the umbilicus. Pelvic MRI reveals multiple intramural fibroids in a uterus estimated to weigh roughly 2,850 g. The patient is given a 3-month course of a GnRH agonist.

CASE 2
Outcome After treatment with the GnRH agonist, the patient undergoes hand-assisted, laparoscopic, multiple myomectomy. She is discharged home the following day and resumes normal activities within two weeks.
Findings A 32-year-old nulliparous patient seeks treatment for menomenorrhagia and symptoms of bulk and expresses a desire for uterine conservation. Pelvic MRI reveals two distinct intramural fibroids, 6 cm and 9 cm in size.

CASE 3
Outcome The patient undergoes laparoscopic myomectomy without preoperative treatment with a GnRH agonist and is discharged home the same day without postoperative complication. (Although the uterus had two large fibroids, we did not use a GnRH agonist because the uterus was well below the belly button.)
5. Use careful surgical technique
Pay attention to set-up, initial entry
Although we lack definitive data on the practical utility of preoperative, intravenous (IV) antibiotics, we administer cefazolin prophylactically, switching to IV clindamycin if there is a documented allergy to penicillin.
In addition, a uterine manipulator is helpful when the patient has a small or medium-sized uterus. A variety of manipulators are available, but we generally use the VCare manipulator (ConMed Corp) because it is easy to insert and provides excellent uterine mobility.
Initial entry is at the umbilicus, unless the uterus extends above the umbilicus, in which case we enter in the left upper quadrant. We generally place the camera through the umbilical port and use two parallel operative ports on the left side of the patient, where the primary surgeon stands. A detailed description of our laparoscopic entry technique was published recently.13
Place the first trocar two finger-breadths medial and superior to the iliac spine and the second trocar 8 cm cephalad to the first port ( FIGURE 2 ). In a large uterus, trocars may have to be placed higher on the abdomen. A third operative port may be added on the right side, if it is needed.

FIGURE 2 Laparoscopic port placement
Place the camera through the umbilical port (A) and operate through two additional ports on the left-hand side of the patient, where the primary surgeon stands. Place the first operative port two finger-breadths medial and superior to the iliac spine (B) and the second port 8 cm cephalad to the first (C).
Incise the uterus
Infiltrate the uterus with dilute vasopressin (20 U in 60 mL of saline), taking care to administer no more than 10 U at a time to minimize the potential for cardiovascular side effects such as bradycardia and hypertension.14 In the past, we periodically encountered episodes of bradycardia when we used 20 U in 40 mL of saline, but we have not had that problem since we changed to a more dilute vasopressin and used no more than 10 U at a time. It may be that an even smaller amount of vasopressin is just as effective, but we do not yet have sufficient data on myomectomy to determine whether that is the case.
Inject the vasopressin subserosally and along the planned hysterotomy. The fibroid itself contains no blood vessels, but the blood supply to the fibroid generally assumes a coronal pattern around it.15 Therefore, it is important to inject the vasopressin into the correct subserosal plane.
We prefer to make a horizontal hysterotomy using the Harmonic Scalpel (Ethicon Endo-Surgery), but other energy sources, such as a monopolar hook or bipolar spatula, are also appropriate.
We choose a horizontal incision because of the ipsilateral port placement we use for suturing. Surgeons who use a midline or contralateral port for suturing may find it easier to repair a vertical hysterotomy. The pattern of blood vessels along the uterus is heterogeneous and variable, and there is no evidence that blood loss or other outcomes are affected by the direction of the uterine incision.15
Once the uterus has been incised, it is important to work efficiently because bleeding will probably continue until the hysterotomy site is completely closed.
Extract the fibroid ("rock and roll")
Extract the fibroid from the uterus by applying generous traction using a tenaculum, and by applying counter-traction using an atraumatic grasper and the Harmonic Scalpel, as needed. We try to limit the use of thermal energy during this step.
The most important aspect of fibroid extraction is ensuring entry into the correct plane. Appropriate entry makes it possible to remove most fibroids without the need for sharp or thermal dissection.
If you are not sure whether you have entered the correct plane, it is better to cut into the fibroid rather than remain too shallow. If you do not enter all the way into the correct plane, you run the risk of pulling and tearing uterine muscle fibers and causing bleeding.
We describe the technique of fibroid extraction as "rock and roll" because it is generally easier to grab the fibroid near the hysterotomy and roll it out rather than pull on the portion of the fibroid that protrudes from the uterus (see the image, for example).

During laparoscopic myomectomy, extract the fibroid by applying generous traction with the tenaculum and counter-traction with an atraumatic grasper and ultrasonic shears. Once you have entered the correct surgical plane, grasp the fibroid near the hysterotomy and simply roll it out of the uterus.
Pregnancy outcomes after laparoscopic myomectomy are generally favorable, with a pregnancy rate that is comparable to or even higher than the rate associated with abdominal myomectomy.1-4
Uneventful vaginal deliveries following laparoscopic myomectomy have been reported in several case series, but so have a number of cases of gravid uterine rupture.5 In a recent study involving 2,050 laparoscopic myomectomies, investigators tracked 386 post-myomectomy pregnancies, 309 deliveries in all, of which 68 were vaginal deliveries.25 It found one case of uterine rupture documented at 33 weeks in a woman who had undergone adenomyomectomy.25
Overall, the literature suggests that uterine rupture after laparoscopic myomectomy is a rare event, occurring in fewer than 1% of pregnancies. Some surgeons use a somewhat arbitrary rule of thumb requiring cesarean delivery if the uterine cavity is entered at myomectomy. This practice is not based on hard evidence, but it does make intuitive sense. If the uterine cavity is entered during myomectomy, it creates a transmural defect that may be more difficult to repair and could carry a higher risk of rupture.
Uterine rupture has also occurred several years after removal of a pedunculated fibroid, suggesting that the use of electrosurgery may weaken the uterine muscle and increase the risk of rupture.
In general—and regardless of the depth of the hysterotomy—it is advisable to counsel patients who have undergone laparoscopic myomectomy that the uterus heals with a scar that may be slightly weaker than the normal myometrium and that elective cesarean delivery may be the optimal strategy. However, a trial of labor is a reasonable alternative, provided the patient receives careful surveillance in a hospital setting.
Close the myometrium
In the event of inadvertent entry into the uterine cavity, close the endometrial defect using running 2-0 polyglactin 910 suture, taking care to avoid suture entry into the uterine cavity. Tie this suture using intracorporeal knot-tying.
Close the hysterotomy in layers using 14 × 14 cm bidirectional barbed 0 PDO suture on a 36-mm, half-circle, taper-point needle. If the hysterotomy is longer than 8 cm, we prefer to use 24 × 24 cm suture.
Tack the first needle into the opposite anterior abdominal wall to help prevent tangling of the suture. Close the deepest layer using the first needle and the more superficial layer and serosa using the second needle. Then cut the needles. Because of the uniform tension and bidirectional nature of the barbed suture, no knots are required.
We began using Quill bidirectional barbed suture (Angiotech) in March 2008.16 Since then, we have completed almost 300 laparoscopic cases using this material, including approximately 100 laparoscopic myomectomies. We compiled data on our first year of experience with this material ( TABLE ), during which we had no major complications related to use of the suture, no conversions to laparotomy, and no returns to the OR to address bleeding or complications arising from the use of bidirectional barbed suture.
The original version of barbed suture included a 6-cm segment of regular, smooth suture. If suturing extends to include this segment, apply a LapraTy clip (Ethicon). This use of LapraTy is off-label because the clip is intended for use with Vicryl 2/0, 3/0, and 4/0 (manufacturer). Nevertheless, our clinical experience with this approach has been favorable.16,17
When closing the uterus, use as many layers as necessary to eliminate all dead space within the myometrium. Sometimes, as many as five layers are needed to close a deep myometrial defect, but a two- or three-layer closure is most common.
TABLE
1 year of experience with laparoscopic myomectomy using bidirectional barbed suture
| Variable | Mean ± standard deviation |
|---|---|
| Duration of surgery (min) | 125.47 ± 55.30 |
| Estimated blood loss (mL) | 158.68 ± 252.35 |
| Number of fibroids removed | 4.01 ± 4.21 |
| Weight of fibroids (g) | 252.07 ± 196.43 |
| Hospital stay (days) | 0.73 ± 0.36 |
| Data represent the author's experience with 55 consecutive laparoscopic myomectomy cases between March 2008 and March 2009. | |
Ward off adhesions
We generally cover the hysterotomy site with an adhesion barrier such as Interceed (Gynecare). Although no adhesion barrier is ideal, Interceed has proved to be effective in this clinical scenario.18 Make sure that the hysterotomy site is completely hemostatic at the time the barrier is applied.
Morcellate with caution
We generally use a 12- to 15-mm electronic morcellator for fibroid removal. Morcellation through the umbilicus is often feasible and prevents the need for a large peripheral incision, which may be less cosmetically pleasing to the patient and potentially more painful than a 15-mm umbilical incision.
We place a 5-mm optic through a peripheral port on the ipsilateral side of the surgeon because it allows the surgeon to operate away from the camera, causing less disorientation. Morcellation is inherently dangerous because of the risk of injury to internal organs such as bowel and blood vessels. The best way to prevent such injuries is to:
- keep the rotating blade in view at all times
- stay on the surface of the fibroid during morcellation (avoid coring)
- hold the morcellator steady during morcellation, i.e., do not move it forward while it is active.
6. NSAIDs and few restrictions are the norm postoperatively
We discharge almost all of our patients postoperatively on the day of laparoscopic myomectomy. Patients who have several medical comorbidities may need to stay overnight, however. We have not yet had to readmit a patient after a day-of-procedure discharge, and patients generally recover fairly rapidly.
We are prospectively evaluating our patients' return to daily activities. Most have resumed normal preoperative activities within 10 days. We recommend the scheduled use of nonsteroidal anti-inflammatory medications (NSAIDs), such as 800 mg of ibuprofen every 6 to 8 hours, for the first 3 to 5 days after surgery.
We encourage patients to remain active after surgery, with no weight-lifting restrictions. Instead, we instruct patients to live by the rule, "If it hurts, don't do it." We do prescribe narcotics, but we instruct patients to limit their use as much as possible. We give IV ketorolac perioperatively.
7. Uterine artery occlusion may prevent recurrence
One treatment option that we occasionally use in conjunction with laparoscopic myomectomy is laparoscopic uterine artery occlusion (LUAO) because it can significantly reduce the recurrence rate of uterine fibroids.19 LUAO is especially valuable in the setting of multiple, small uterine fibroids ("bag of marbles") and other cases where it is unlikely that all fibroids will be removed during myomectomy.
At present, we perform LUAO only in patients who have completed childbearing, although early evidence suggests that pregnancy may be relatively safe after uterine artery occlusion.19,20 More data and longer follow-up are required before LUAO should be offered to all women of reproductive age.
8. Consider single-incision laparoscopic myomectomy
This approach has been touted as offering an improved cosmetic outcome and, possibly, less postoperative pain, although these potential benefits have yet to be demonstrated in a well-designed prospective trial.21
We have performed three cases of single-incision myomectomy for an intramural fibroid and have demonstrated this approach to be feasible (article in press). Barbed suture is especially valuable in single-incision surgery because intracorporeal knot-tying can be more challenging when there is only one incision. However, limitations include:
- a lack of triangulation
- instrument crowding at the umbilicus
- difficulty suturing using traditional or barbed suture.
Proper documentation is key
Current Procedural Terminology (CPT) offers two coding options when you've performed a laparoscopic myomectomy:
- 58545 (laparoscopy, surgical, myomectomy, excision; 1 to 4 intramural myomas with total weight of 250 g or less and/or removal of surface myomas)
- 58546 (laparoscopy, surgical, myomectomy, excision; 5 or more intramural myomas and/or intramural myomas with total weight greater than 250 g).
Which code you submit can, of course, make a difference in how much you're reimbursed: 58545 carries 24.21 relative value units (RVU); 58546, 30.59 RVU. The documentation that you present will, therefore, be key in getting paid for the work you've performed.
First, look at the description of 58545. You have two documentation options:
- You removed between one and four intramural myomas (International Classification of Diseases [ICD-9] 218.1; intramural leiomyoma of uterus) whose total weight was =250 g
- You encountered surface myomas (ICD9 218.2; subserous leiomyoma of uterus) and removed all of them, weight aside.
Second, to bill the higher-paying code (58546), you must clearly document removal of intramural myomas only. Again, your work must meet either of two criteria:
- Total weight of all intramural myomas removed is >250 g
- You removed five or more intramural myomas.
You can determine the total weight of the excised tissue 1) in the operating room, if a scale is available, or 2) from the pathology report. A caution: The tissue that you've removed will shrink after it arrives in pathology, and this shrinkage may make a difference when, for example, fewer than five myomas were removed and their total weight is close to 250 g.
Last, estimating the weight of myomas by ultrasonography before surgery is not considered acceptable documentation of weight by most payers.—Melanie Witt, RN, CPC, COBGC, MA
Ms. Witt is an independent coding and documentation consultant and former program manager, department of coding and nomenclature, American College of Obstetricians and Gynecologists.
Therefore, it may be challenging to apply single-port surgery to more complex pathology, such as very large fibroids and severe pelvic adhesive disease.
Single-incision surgery may offer marginal cosmetic benefit for some patients. When we surveyed our patients informally, however, most of them expressed satisfaction with the cosmetic appearance of peripheral laparoscopic port incisions.
Another potential limitation of single-incision surgery is the cost associated with disposable, articulating instruments and single-port access devices. Although robotic surgery is a feasible approach to both multi-port22,23 and single-port surgery,24 prospective data are lacking, and cost remains an issue. It is possible that future developments in robotic surgery may facilitate suture-intensive, single-incision cases such as myomectomy and sacrocolpopexy. Well-designed prospective trials are urgently needed.
Myomectomy is the surgery of choice for women who have symptomatic fibroids and who wish to retain their uterus. And laparoscopic myomectomy is preferable to the abdominal approach in many ways, offering: 1-4
- faster recovery
- a shorter hospital stay
- diminished blood loss
- decreased adhesion formation
- a comparable or higher rate of pregnancy.
Nevertheless, laparoscopic myomectomy is a technically challenging procedure with surgeon-specific limitations. The biggest challenge: appropriately suturing the hysterotomy site.
In this article, I share my experience with laparoscopic myomectomy and offer 8 pearls that may contribute to a successful outcome.
1. Don't settle on laparoscopy prematurely
Given its advantages over the abdominal route, laparoscopic myomectomy should be the preferred approach in the treatment of symptomatic uterine fibroids ( FIGURE 1 ). However, not all patients are appropriate candidates for laparoscopy. Several guidelines have recommended a maximum number and size of fibroids for laparoscopic removal, but practice varies widely, and experienced surgeons successfully take on cases that are well beyond the limits set by most published guidelines.5-7
At our institution, we do not have firm guidelines in place for the number and size of fibroids that can be removed laparoscopically. Other variables enter into decision-making and counseling, among them any medical comorbidity or history of uterine surgery the patient may have, as well as her desires in regard to childbearing and uterine retention.
Hysterectomy may be the most straightforward option for women who have symptomatic fibroids and who have completed childbearing. However, myomectomy is also appropriate as long as the patient is aware of the risk that fibroids may recur and the potential for further surgery. When the patient is in her late 40s or early 50s, the likelihood of fibroid recurrence may be lower than it is in the general population.
In my practice, submucosal and intracavitary fibroids smaller than 4 cm and more than 5 mm away from the uterine serosa are generally removed hysteroscopically, an approach beyond the scope of this article. In women who have completed childbearing but who wish to conserve the uterus, we deliberately enter the uterine cavity laparoscopically because this strategy allows for efficient removal of submucosal and intracavitary fibroids.

FIGURE 1 When and how to treat uterine fibroids
2. Estimate the duration of surgery
When the patient has fibroids that are intramural or subserosal, our general rule of thumb is to determine her suitability for laparoscopic myomectomy, based on the estimated duration of the operation. A surgeon's ability to calculate the length of the operation for a particular patient increases with experience.
We tend to recommend the laparoscopic approach when the procedure is expected to take less than 3 hours to complete. More than 95% of our patients fall into this category.
When we anticipate a prolonged operating time, we discuss the option of hand-assisted laparoscopic myomectomy. This approach involves two or three 5-mm trocar punctures high on the abdomen in conjunction with a suprapubic incision, 6 to 7 cm in length with a hand port in place. Prospective studies have demonstrated a significantly longer recovery with minilaparotomy than with laparoscopy, but these trials compared uteri of similar size.4,8 We expect the laparoscopic approach to confer fewer advantages when operative time is prolonged significantly.
In our practice, we consider one or more of the following conditions appropriate for hand-assisted laparoscopic myomectomy:
- a very large uterus (i.e., heavier than 1,500 to 2,000 g). In these cases, operating times can be excessive because of the need for extensive suturing and morcellation, and bleeding may increase as a result
- more than 20 fibroids on magnetic resonance imaging (MRI). It can be a challenge to locate all of the fibroids; multiple uterine incisions may be necessary
- a medical comorbidity that renders the patient unable to tolerate prolonged anesthesia. For example, we operated on a patient who had Ehlers-Danlos syndrome and who needed to avoid a prolonged operation due to fragile bones and joint laxity.
Of necessity, these guidelines will vary from practice to practice, and gynecologic surgeons who are just beginning to perform laparoscopic myomectomy should not include multiple fibroids or a large uterus among their initial cases. Instead, perform the first few cases in patients who have not had abdominal surgery and who have a symptomatic intramural or subserosal fibroid that is close to the uterine fundus and no larger than 6 cm in diameter.
3. Consider preoperative MRI
Preoperative imaging greatly supplements the clinical examination and facilitates identification of the number, location, and characteristics of the fibroids. Pelvic ultrasonography (US) is appropriate for most patients. We prefer preoperative MRI of the pelvis in the following scenarios:
- uterus larger than 12 weeks (280 g) on clinical examination
- identification of multiple fibroids via US
- history suggestive of adenomyosis.
MRI facilitates preoperative planning by accurately delineating the size and location of the fibroids, and by distinguishing between an adenomyoma and fibroid in most cases.9 For an example of its utility, see "How MRI can guide treatment: 3 cases."
4. Preoperative medical therapy may be indicated
When given preoperatively, gonadotropin-releasing hormone (GnRH) agonists have been shown to reduce blood loss and shorten operative time. The exception: cases involving hypoechoic fibroids, because the cleavage plane may be more difficult to identify, prolonging operative time.10
We generally prefer to use a GnRH agonist in two clinical scenarios: 1) anemia and 2) a uterus that extends above the umbilicus. In the second scenario, the GnRH agonist helps reduce the uterus to a more manageable size.
Aromatase inhibitors show great promise as preoperative agents because there is no initial flare. In addition, because fibroids have a higher concentration of aromatase activity than the surrounding myometrium, a low dosage of an aromatase inhibitor is effective and does not cause significant menopausal symptoms.
A recent comparative study found that fibroid shrinkage was greater after 3 months of letrozole (2.5 mg/day) than after use of a GnRH agonist.11 Total myoma volume decreased by 45.6% in the letrozole group, compared with 33.2% in the group that received a GnRH agonist (P =.02).11
Aromatase inhibitors have also been successfully used during the initial period of GnRH agonist therapy to prevent the symptoms of flare.12 However, because clinical experience is limited, the long-term efficacy and safety of aromatase inhibitors in premenopausal women is unknown.
Findings A 40-year-old nulliparous woman seeks treatment for menometrorrhagia and dysmenorrhea but wants to conserve her uterus. MRI reveals a 4.5-cm submucosal fibroid (arrow) that extends all the way to the uterine serosa, with no evidence of adenomyosis. Her thyroid-stimulating hormone (TSH) level is normal, as is an endometrial biopsy.

CASE 1
Outcome We decide to proceed with laparoscopic myomectomy because a hysteroscopic approach would carry a risk of uterine rupture.
Findings A 36-year-old nulliparous woman complains of significant "bulk" symptoms (heaviness, urinary frequency, and abdominal bloating). She has a visible mass that extends four finger-breadths above the umbilicus. Pelvic MRI reveals multiple intramural fibroids in a uterus estimated to weigh roughly 2,850 g. The patient is given a 3-month course of a GnRH agonist.

CASE 2
Outcome After treatment with the GnRH agonist, the patient undergoes hand-assisted, laparoscopic, multiple myomectomy. She is discharged home the following day and resumes normal activities within two weeks.
Findings A 32-year-old nulliparous patient seeks treatment for menomenorrhagia and symptoms of bulk and expresses a desire for uterine conservation. Pelvic MRI reveals two distinct intramural fibroids, 6 cm and 9 cm in size.

CASE 3
Outcome The patient undergoes laparoscopic myomectomy without preoperative treatment with a GnRH agonist and is discharged home the same day without postoperative complication. (Although the uterus had two large fibroids, we did not use a GnRH agonist because the uterus was well below the belly button.)
5. Use careful surgical technique
Pay attention to set-up, initial entry
Although we lack definitive data on the practical utility of preoperative, intravenous (IV) antibiotics, we administer cefazolin prophylactically, switching to IV clindamycin if there is a documented allergy to penicillin.
In addition, a uterine manipulator is helpful when the patient has a small or medium-sized uterus. A variety of manipulators are available, but we generally use the VCare manipulator (ConMed Corp) because it is easy to insert and provides excellent uterine mobility.
Initial entry is at the umbilicus, unless the uterus extends above the umbilicus, in which case we enter in the left upper quadrant. We generally place the camera through the umbilical port and use two parallel operative ports on the left side of the patient, where the primary surgeon stands. A detailed description of our laparoscopic entry technique was published recently.13
Place the first trocar two finger-breadths medial and superior to the iliac spine and the second trocar 8 cm cephalad to the first port ( FIGURE 2 ). In a large uterus, trocars may have to be placed higher on the abdomen. A third operative port may be added on the right side, if it is needed.

FIGURE 2 Laparoscopic port placement
Place the camera through the umbilical port (A) and operate through two additional ports on the left-hand side of the patient, where the primary surgeon stands. Place the first operative port two finger-breadths medial and superior to the iliac spine (B) and the second port 8 cm cephalad to the first (C).
Incise the uterus
Infiltrate the uterus with dilute vasopressin (20 U in 60 mL of saline), taking care to administer no more than 10 U at a time to minimize the potential for cardiovascular side effects such as bradycardia and hypertension.14 In the past, we periodically encountered episodes of bradycardia when we used 20 U in 40 mL of saline, but we have not had that problem since we changed to a more dilute vasopressin and used no more than 10 U at a time. It may be that an even smaller amount of vasopressin is just as effective, but we do not yet have sufficient data on myomectomy to determine whether that is the case.
Inject the vasopressin subserosally and along the planned hysterotomy. The fibroid itself contains no blood vessels, but the blood supply to the fibroid generally assumes a coronal pattern around it.15 Therefore, it is important to inject the vasopressin into the correct subserosal plane.
We prefer to make a horizontal hysterotomy using the Harmonic Scalpel (Ethicon Endo-Surgery), but other energy sources, such as a monopolar hook or bipolar spatula, are also appropriate.
We choose a horizontal incision because of the ipsilateral port placement we use for suturing. Surgeons who use a midline or contralateral port for suturing may find it easier to repair a vertical hysterotomy. The pattern of blood vessels along the uterus is heterogeneous and variable, and there is no evidence that blood loss or other outcomes are affected by the direction of the uterine incision.15
Once the uterus has been incised, it is important to work efficiently because bleeding will probably continue until the hysterotomy site is completely closed.
Extract the fibroid ("rock and roll")
Extract the fibroid from the uterus by applying generous traction using a tenaculum, and by applying counter-traction using an atraumatic grasper and the Harmonic Scalpel, as needed. We try to limit the use of thermal energy during this step.
The most important aspect of fibroid extraction is ensuring entry into the correct plane. Appropriate entry makes it possible to remove most fibroids without the need for sharp or thermal dissection.
If you are not sure whether you have entered the correct plane, it is better to cut into the fibroid rather than remain too shallow. If you do not enter all the way into the correct plane, you run the risk of pulling and tearing uterine muscle fibers and causing bleeding.
We describe the technique of fibroid extraction as "rock and roll" because it is generally easier to grab the fibroid near the hysterotomy and roll it out rather than pull on the portion of the fibroid that protrudes from the uterus (see the image, for example).

During laparoscopic myomectomy, extract the fibroid by applying generous traction with the tenaculum and counter-traction with an atraumatic grasper and ultrasonic shears. Once you have entered the correct surgical plane, grasp the fibroid near the hysterotomy and simply roll it out of the uterus.
Pregnancy outcomes after laparoscopic myomectomy are generally favorable, with a pregnancy rate that is comparable to or even higher than the rate associated with abdominal myomectomy.1-4
Uneventful vaginal deliveries following laparoscopic myomectomy have been reported in several case series, but so have a number of cases of gravid uterine rupture.5 In a recent study involving 2,050 laparoscopic myomectomies, investigators tracked 386 post-myomectomy pregnancies, 309 deliveries in all, of which 68 were vaginal deliveries.25 It found one case of uterine rupture documented at 33 weeks in a woman who had undergone adenomyomectomy.25
Overall, the literature suggests that uterine rupture after laparoscopic myomectomy is a rare event, occurring in fewer than 1% of pregnancies. Some surgeons use a somewhat arbitrary rule of thumb requiring cesarean delivery if the uterine cavity is entered at myomectomy. This practice is not based on hard evidence, but it does make intuitive sense. If the uterine cavity is entered during myomectomy, it creates a transmural defect that may be more difficult to repair and could carry a higher risk of rupture.
Uterine rupture has also occurred several years after removal of a pedunculated fibroid, suggesting that the use of electrosurgery may weaken the uterine muscle and increase the risk of rupture.
In general—and regardless of the depth of the hysterotomy—it is advisable to counsel patients who have undergone laparoscopic myomectomy that the uterus heals with a scar that may be slightly weaker than the normal myometrium and that elective cesarean delivery may be the optimal strategy. However, a trial of labor is a reasonable alternative, provided the patient receives careful surveillance in a hospital setting.
Close the myometrium
In the event of inadvertent entry into the uterine cavity, close the endometrial defect using running 2-0 polyglactin 910 suture, taking care to avoid suture entry into the uterine cavity. Tie this suture using intracorporeal knot-tying.
Close the hysterotomy in layers using 14 × 14 cm bidirectional barbed 0 PDO suture on a 36-mm, half-circle, taper-point needle. If the hysterotomy is longer than 8 cm, we prefer to use 24 × 24 cm suture.
Tack the first needle into the opposite anterior abdominal wall to help prevent tangling of the suture. Close the deepest layer using the first needle and the more superficial layer and serosa using the second needle. Then cut the needles. Because of the uniform tension and bidirectional nature of the barbed suture, no knots are required.
We began using Quill bidirectional barbed suture (Angiotech) in March 2008.16 Since then, we have completed almost 300 laparoscopic cases using this material, including approximately 100 laparoscopic myomectomies. We compiled data on our first year of experience with this material ( TABLE ), during which we had no major complications related to use of the suture, no conversions to laparotomy, and no returns to the OR to address bleeding or complications arising from the use of bidirectional barbed suture.
The original version of barbed suture included a 6-cm segment of regular, smooth suture. If suturing extends to include this segment, apply a LapraTy clip (Ethicon). This use of LapraTy is off-label because the clip is intended for use with Vicryl 2/0, 3/0, and 4/0 (manufacturer). Nevertheless, our clinical experience with this approach has been favorable.16,17
When closing the uterus, use as many layers as necessary to eliminate all dead space within the myometrium. Sometimes, as many as five layers are needed to close a deep myometrial defect, but a two- or three-layer closure is most common.
TABLE
1 year of experience with laparoscopic myomectomy using bidirectional barbed suture
| Variable | Mean ± standard deviation |
|---|---|
| Duration of surgery (min) | 125.47 ± 55.30 |
| Estimated blood loss (mL) | 158.68 ± 252.35 |
| Number of fibroids removed | 4.01 ± 4.21 |
| Weight of fibroids (g) | 252.07 ± 196.43 |
| Hospital stay (days) | 0.73 ± 0.36 |
| Data represent the author's experience with 55 consecutive laparoscopic myomectomy cases between March 2008 and March 2009. | |
Ward off adhesions
We generally cover the hysterotomy site with an adhesion barrier such as Interceed (Gynecare). Although no adhesion barrier is ideal, Interceed has proved to be effective in this clinical scenario.18 Make sure that the hysterotomy site is completely hemostatic at the time the barrier is applied.
Morcellate with caution
We generally use a 12- to 15-mm electronic morcellator for fibroid removal. Morcellation through the umbilicus is often feasible and prevents the need for a large peripheral incision, which may be less cosmetically pleasing to the patient and potentially more painful than a 15-mm umbilical incision.
We place a 5-mm optic through a peripheral port on the ipsilateral side of the surgeon because it allows the surgeon to operate away from the camera, causing less disorientation. Morcellation is inherently dangerous because of the risk of injury to internal organs such as bowel and blood vessels. The best way to prevent such injuries is to:
- keep the rotating blade in view at all times
- stay on the surface of the fibroid during morcellation (avoid coring)
- hold the morcellator steady during morcellation, i.e., do not move it forward while it is active.
6. NSAIDs and few restrictions are the norm postoperatively
We discharge almost all of our patients postoperatively on the day of laparoscopic myomectomy. Patients who have several medical comorbidities may need to stay overnight, however. We have not yet had to readmit a patient after a day-of-procedure discharge, and patients generally recover fairly rapidly.
We are prospectively evaluating our patients' return to daily activities. Most have resumed normal preoperative activities within 10 days. We recommend the scheduled use of nonsteroidal anti-inflammatory medications (NSAIDs), such as 800 mg of ibuprofen every 6 to 8 hours, for the first 3 to 5 days after surgery.
We encourage patients to remain active after surgery, with no weight-lifting restrictions. Instead, we instruct patients to live by the rule, "If it hurts, don't do it." We do prescribe narcotics, but we instruct patients to limit their use as much as possible. We give IV ketorolac perioperatively.
7. Uterine artery occlusion may prevent recurrence
One treatment option that we occasionally use in conjunction with laparoscopic myomectomy is laparoscopic uterine artery occlusion (LUAO) because it can significantly reduce the recurrence rate of uterine fibroids.19 LUAO is especially valuable in the setting of multiple, small uterine fibroids ("bag of marbles") and other cases where it is unlikely that all fibroids will be removed during myomectomy.
At present, we perform LUAO only in patients who have completed childbearing, although early evidence suggests that pregnancy may be relatively safe after uterine artery occlusion.19,20 More data and longer follow-up are required before LUAO should be offered to all women of reproductive age.
8. Consider single-incision laparoscopic myomectomy
This approach has been touted as offering an improved cosmetic outcome and, possibly, less postoperative pain, although these potential benefits have yet to be demonstrated in a well-designed prospective trial.21
We have performed three cases of single-incision myomectomy for an intramural fibroid and have demonstrated this approach to be feasible (article in press). Barbed suture is especially valuable in single-incision surgery because intracorporeal knot-tying can be more challenging when there is only one incision. However, limitations include:
- a lack of triangulation
- instrument crowding at the umbilicus
- difficulty suturing using traditional or barbed suture.
Proper documentation is key
Current Procedural Terminology (CPT) offers two coding options when you've performed a laparoscopic myomectomy:
- 58545 (laparoscopy, surgical, myomectomy, excision; 1 to 4 intramural myomas with total weight of 250 g or less and/or removal of surface myomas)
- 58546 (laparoscopy, surgical, myomectomy, excision; 5 or more intramural myomas and/or intramural myomas with total weight greater than 250 g).
Which code you submit can, of course, make a difference in how much you're reimbursed: 58545 carries 24.21 relative value units (RVU); 58546, 30.59 RVU. The documentation that you present will, therefore, be key in getting paid for the work you've performed.
First, look at the description of 58545. You have two documentation options:
- You removed between one and four intramural myomas (International Classification of Diseases [ICD-9] 218.1; intramural leiomyoma of uterus) whose total weight was =250 g
- You encountered surface myomas (ICD9 218.2; subserous leiomyoma of uterus) and removed all of them, weight aside.
Second, to bill the higher-paying code (58546), you must clearly document removal of intramural myomas only. Again, your work must meet either of two criteria:
- Total weight of all intramural myomas removed is >250 g
- You removed five or more intramural myomas.
You can determine the total weight of the excised tissue 1) in the operating room, if a scale is available, or 2) from the pathology report. A caution: The tissue that you've removed will shrink after it arrives in pathology, and this shrinkage may make a difference when, for example, fewer than five myomas were removed and their total weight is close to 250 g.
Last, estimating the weight of myomas by ultrasonography before surgery is not considered acceptable documentation of weight by most payers.—Melanie Witt, RN, CPC, COBGC, MA
Ms. Witt is an independent coding and documentation consultant and former program manager, department of coding and nomenclature, American College of Obstetricians and Gynecologists.
Therefore, it may be challenging to apply single-port surgery to more complex pathology, such as very large fibroids and severe pelvic adhesive disease.
Single-incision surgery may offer marginal cosmetic benefit for some patients. When we surveyed our patients informally, however, most of them expressed satisfaction with the cosmetic appearance of peripheral laparoscopic port incisions.
Another potential limitation of single-incision surgery is the cost associated with disposable, articulating instruments and single-port access devices. Although robotic surgery is a feasible approach to both multi-port22,23 and single-port surgery,24 prospective data are lacking, and cost remains an issue. It is possible that future developments in robotic surgery may facilitate suture-intensive, single-incision cases such as myomectomy and sacrocolpopexy. Well-designed prospective trials are urgently needed.
1. Seracchioli R, Rossi S, Govoni F, et al. Fertility and obstetric outcome after laparoscopic myomectomy of large myomata: a randomized comparison with abdominal myomectomy. Hum Reprod. 2000;15(12):2663-2668.
2. Rossetti A, Sizzi O, Soranna L, Cucinelli F, Mancuso S, Lanzone A. Long-term results of laparoscopic myomectomy: recurrence rate in comparison with abdominal myomectomy. Hum Reprod. 2001;16(4):770-774.
3. Stringer NH, Walker JC, Meyer PM. Comparison of 49 laparoscopic myomectomies with 49 open myomectomies. J Am Assoc Gynecol Laparosc. 1997;4(4):457-464.
4. Palomba S, Zupi E, Falbo A, et al. A multicenter randomized, controlled study comparing laparoscopic versus minilaparotomic myomectomy: reproductive outcomes. Fertil Steril. 2007;88(4):933-941.
5. Hurst BS, Matthews ML, Marshburn PB. Laparoscopic myomectomy for symptomatic uterine myomas. Fertil Steril. 2005;83:1-23.
6. Agdi M, Tulandi T. Endoscopic management of uterine fibroids. Best Pract Res Clin Obstet Gynaecol. 2008;22(4):707-716.
7. Sinha R, Hegde A, Mahajan C, Dubey N, Sundaram M. Laparoscopic myomectomy: do size, number, and location of the myomas form limiting factors for laparoscopic myomectomy? J Minim Invasive Gynecol. 2008;15(3):292-300.
8. Malzoni M, Tinelli R, Cosentino F, et al. Laparoscopy versus minilaparotomy in women with symptomatic uterine myomas: short-term and fertility results [published online ahead of print March 12, 2009; corrected proof March 16, 2009]. Fertil Steril. 2009;doi:10.1016/j.fertnstert.2008.12.127.
9. Dudiak CM, Turner DA, Patel SK, Archie JT, Silver B, Norusis M. Uterine leiomyomas in the infertile patient: preoperative localization with MR imaging versus US and hysterosalpingography. Radiology. 1988;167(3):627-630.
10. Zullo F, Pellicano M, De Stefano R, Zupi E, Mastrantonio P. A prospective randomized study to evaluate leuprolide acetate treatment before laparoscopic myomectomy: efficacy and ultrasonographic predictors. Am J Obstet Gynecol. 1998;178(1 Pt 1):108-112.
11. Parsanezhad ME, Azmoon M, Alborzi S, et al. A randomized, controlled clinical trial comparing the effects of aromatase inhibitor (letrozole) and gonadotropin-releasing hormone agonist (triptorelin) on uterine leiomyoma volume and hormonal status. Fertil Steril. 2010;93(1):192-198.
12. Bedaiwy MA, Mousa NA, Casper RF. Aromatase inhibitors prevent the estrogen rise associated with the flare effect of gonadotropins in patients treated with GnRH agonists. Fertil Steril. 2009;91(suppl 4):1574-1577.
13. Vellinga TT, De Alwis S, Suzuki Y, Einarsson JI. Laparoscopic entry: the modified Alwis method and more. Rev Obstet Gynecol. 2009;2(3):193-198.
14. Frishman G. Vasopressin: if some is good, is more better? Obstet Gynecol. 2009;113(2 Pt 2):476-477.
15. Discepola F, Valenti DA, Reinhold C, Tulandi T. Analysis of arterial blood vessels surrounding the myoma: relevance to myomectomy. Obstet Gynecol. 2007;110(6):1301-1303.
16. Greenberg JA, Einarsson JI. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy. J Minim Invasive Gynecol. 2008;15(5):621-623.
17. Einarsson JI, Vellinga TT, Twijnstra AR, Suzuki Y, Greenberg JA. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy; an evaluation of safety and clinical outcomes [abstract]. J Minim Invasive Gynecol. 2009;16(6)(suppl 1):S28-S29.Abstract 95.
18. Mais V, Ajossa S, Piras B, Guerriero S, Marongiu D, Melis GB. Prevention of de-novo adhesion formation after laparoscopic myomectomy: a randomized trial to evaluate the effectiveness of an oxidized regenerated cellulose absorbable barrier. Hum Reprod. 1995;10(12):3133-3135.
19. Alborzi S, Ghannadan E, Alborzi S, Alborzi M. A comparison of combined laparoscopic uterine artery ligation and myomectomy versus laparoscopic myomectomy in treatment of symptomatic myoma. Fertil Steril. 2009;92(2):742-747.
20. Holub Z, Mara M, Kuzel D, Jabor A, Maskova J, Eim J. Pregnancy outcomes after uterine artery occlusion: prospective multicentric study. Fertil Steril. 2008;90(5):1886-1891.
21. Curcillo PG, King SA, Podolsky ER, Rottman SJ. Single Port Access (SPA) minimal access surgery through a single incision. Surg Technol Int. 2009;18:19-25.
22. Nezhat C, Lavie O, Hsu S, Watson J, Barnett O, Lemyre M. Robotic-assisted laparoscopic myomectomy compared with standard laparoscopic myomectomy—a retrospective matched control study. Fertil Steril. 2009;91(2):556-559.
23. Bedient CE, Magrina JF, Noble BN, Kho RM. Comparison of robotic and laparoscopic myomectomy. Am J Obstet Gynecol. 2009;201(6):566.e1-5.
24. Escobar PF, Fader AN, Paraiso MF, Kaouk JH, Falcone T. Robotic-assisted laparoendoscopic single-site surgery in gynecology: initial report and technique. J Minim Invasive Gynecol. 2009;16(5):589-591.
25. Sizzi O, Rossetti A, Malzoni M, et al. Italian multicenter study on complications of laparoscopic myomectomy. J Minim Invasive Gynecol. 2007;14(4):453-462.
1. Seracchioli R, Rossi S, Govoni F, et al. Fertility and obstetric outcome after laparoscopic myomectomy of large myomata: a randomized comparison with abdominal myomectomy. Hum Reprod. 2000;15(12):2663-2668.
2. Rossetti A, Sizzi O, Soranna L, Cucinelli F, Mancuso S, Lanzone A. Long-term results of laparoscopic myomectomy: recurrence rate in comparison with abdominal myomectomy. Hum Reprod. 2001;16(4):770-774.
3. Stringer NH, Walker JC, Meyer PM. Comparison of 49 laparoscopic myomectomies with 49 open myomectomies. J Am Assoc Gynecol Laparosc. 1997;4(4):457-464.
4. Palomba S, Zupi E, Falbo A, et al. A multicenter randomized, controlled study comparing laparoscopic versus minilaparotomic myomectomy: reproductive outcomes. Fertil Steril. 2007;88(4):933-941.
5. Hurst BS, Matthews ML, Marshburn PB. Laparoscopic myomectomy for symptomatic uterine myomas. Fertil Steril. 2005;83:1-23.
6. Agdi M, Tulandi T. Endoscopic management of uterine fibroids. Best Pract Res Clin Obstet Gynaecol. 2008;22(4):707-716.
7. Sinha R, Hegde A, Mahajan C, Dubey N, Sundaram M. Laparoscopic myomectomy: do size, number, and location of the myomas form limiting factors for laparoscopic myomectomy? J Minim Invasive Gynecol. 2008;15(3):292-300.
8. Malzoni M, Tinelli R, Cosentino F, et al. Laparoscopy versus minilaparotomy in women with symptomatic uterine myomas: short-term and fertility results [published online ahead of print March 12, 2009; corrected proof March 16, 2009]. Fertil Steril. 2009;doi:10.1016/j.fertnstert.2008.12.127.
9. Dudiak CM, Turner DA, Patel SK, Archie JT, Silver B, Norusis M. Uterine leiomyomas in the infertile patient: preoperative localization with MR imaging versus US and hysterosalpingography. Radiology. 1988;167(3):627-630.
10. Zullo F, Pellicano M, De Stefano R, Zupi E, Mastrantonio P. A prospective randomized study to evaluate leuprolide acetate treatment before laparoscopic myomectomy: efficacy and ultrasonographic predictors. Am J Obstet Gynecol. 1998;178(1 Pt 1):108-112.
11. Parsanezhad ME, Azmoon M, Alborzi S, et al. A randomized, controlled clinical trial comparing the effects of aromatase inhibitor (letrozole) and gonadotropin-releasing hormone agonist (triptorelin) on uterine leiomyoma volume and hormonal status. Fertil Steril. 2010;93(1):192-198.
12. Bedaiwy MA, Mousa NA, Casper RF. Aromatase inhibitors prevent the estrogen rise associated with the flare effect of gonadotropins in patients treated with GnRH agonists. Fertil Steril. 2009;91(suppl 4):1574-1577.
13. Vellinga TT, De Alwis S, Suzuki Y, Einarsson JI. Laparoscopic entry: the modified Alwis method and more. Rev Obstet Gynecol. 2009;2(3):193-198.
14. Frishman G. Vasopressin: if some is good, is more better? Obstet Gynecol. 2009;113(2 Pt 2):476-477.
15. Discepola F, Valenti DA, Reinhold C, Tulandi T. Analysis of arterial blood vessels surrounding the myoma: relevance to myomectomy. Obstet Gynecol. 2007;110(6):1301-1303.
16. Greenberg JA, Einarsson JI. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy. J Minim Invasive Gynecol. 2008;15(5):621-623.
17. Einarsson JI, Vellinga TT, Twijnstra AR, Suzuki Y, Greenberg JA. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy; an evaluation of safety and clinical outcomes [abstract]. J Minim Invasive Gynecol. 2009;16(6)(suppl 1):S28-S29.Abstract 95.
18. Mais V, Ajossa S, Piras B, Guerriero S, Marongiu D, Melis GB. Prevention of de-novo adhesion formation after laparoscopic myomectomy: a randomized trial to evaluate the effectiveness of an oxidized regenerated cellulose absorbable barrier. Hum Reprod. 1995;10(12):3133-3135.
19. Alborzi S, Ghannadan E, Alborzi S, Alborzi M. A comparison of combined laparoscopic uterine artery ligation and myomectomy versus laparoscopic myomectomy in treatment of symptomatic myoma. Fertil Steril. 2009;92(2):742-747.
20. Holub Z, Mara M, Kuzel D, Jabor A, Maskova J, Eim J. Pregnancy outcomes after uterine artery occlusion: prospective multicentric study. Fertil Steril. 2008;90(5):1886-1891.
21. Curcillo PG, King SA, Podolsky ER, Rottman SJ. Single Port Access (SPA) minimal access surgery through a single incision. Surg Technol Int. 2009;18:19-25.
22. Nezhat C, Lavie O, Hsu S, Watson J, Barnett O, Lemyre M. Robotic-assisted laparoscopic myomectomy compared with standard laparoscopic myomectomy—a retrospective matched control study. Fertil Steril. 2009;91(2):556-559.
23. Bedient CE, Magrina JF, Noble BN, Kho RM. Comparison of robotic and laparoscopic myomectomy. Am J Obstet Gynecol. 2009;201(6):566.e1-5.
24. Escobar PF, Fader AN, Paraiso MF, Kaouk JH, Falcone T. Robotic-assisted laparoendoscopic single-site surgery in gynecology: initial report and technique. J Minim Invasive Gynecol. 2009;16(5):589-591.
25. Sizzi O, Rossetti A, Malzoni M, et al. Italian multicenter study on complications of laparoscopic myomectomy. J Minim Invasive Gynecol. 2007;14(4):453-462.
UPDATE: CERVICAL DISEASE
“Astonishment fatigue.” That phenomenon may be responsible for clinicians’ muted reaction to new ACOG guidelines on cervical cancer screening, which were released late last year. 1 Through a coincidence of timing, the new guidelines hit the airwaves just after the U. S. Preventive Services Task Force announced controversial changes to its recommendations on mammography. As a result, the cervical cytology guidelines seemed to dissolve into the stratosphere.
Or, perhaps, the cervical cancer screening guidelines slipped by with little fanfare because they were soundly based in evidence and, therefore, widely accepted among ObGyns. Even if that is the case, the medical community may not be familiar with the specific data behind the guideline changes. In this article, I discuss the evidence driving all major changes to the guidelines based on Level-A evidence. Changes based on Level-B or -C evidence are listed in TABLE 1 .
Hold off on cervical cancer screening until the patient is 21 years old
Screening before age 21 should be avoided because it may lead to unnecessary and harmful evaluation and treatment in women at very low risk of cancer.1
How different is this from the 2003 ACOG recommendation to begin screening within 3 years of first intercourse or at age 21, whichever comes first?
Very, very different. In fact, it is the most dramatic change in the 2009 screening recommendations.
It is even more striking in comparison with ACOG’s earlier recommendation—which prevailed from the late 1970s through 2002—to begin cervical screening at age 18 or at the onset of intercourse, whichever comes first.
The median age of first intercourse in the United States is 16 years. Until this latest change in guidelines, most young women began cervical screening during adolescence.
What’s wrong with screening adolescents? Don’t they acquire human papillomavirus (HPV)? (Yes.) And once they do, aren’t they at risk of cervical cancer? (Yes.)
Several variables support the delay of screening to age 21:
- the transience of most HPV infections
- the typically long natural history of carcinogenesis in the few young women in whom HPV might persist
- the adverse consequences of over-screening and over-management of adolescents who have cervical intraepithelial neoplasia (CIN).
Let’s look more closely at these variables.
TABLE 1
Other ACOG cervical disease guidelines are based on Level-B and Level-C evidence*
| Recommendation | Level of evidence | Comment |
|---|---|---|
| Test sexually active adolescents (i.e., females 21 years or younger) for sexually transmitted infection, and counsel them about safe sexual practices and contraception | B | These measures can be carried out without cervical cytology and, in the asymptomatic patient, without the introduction of a speculum |
| It is reasonable to discontinue cervical cancer screening in any woman 65 to 70 years old who has had three or more consecutive negative Pap tests and no abnormal tests in the past 10 years | B | |
| Continue annual screening for at least 20 years in any woman who has been treated for CIN 2, CIN 3, or cancer. This population remains at risk of persistent or recurrent disease for at least 20 years after treatment and after initial posttreatment surveillance | B | |
| Continue to screen any woman who has had a total hysterectomy if she has a history of CIN 2 or CIN 3 or if a negative history cannot be documented. This screening should continue even after initial post-treatment surveillance | B | Although the screening interval may ultimately be extended, we lack reliable data to support or refute the discontinuation of screening in this population |
| Inform the patient that annual gynecologic examination may still be appropriate even if cervical cytology is not assessed at each visit | C | |
| Screen any woman who has been immunized against HPV 16 and 18 as though she has not been immunized | C | |
| * Level-B recommendations are based on limited and inconsistent scientific evidence. Level-C recommendations are based primarily on consensus and expert opinion. | ||
HPV is common but usually resolves on its own
It’s common for young women to acquire HPV shortly after they become sexually active, but their immune system clears most infections within 1 or 2 years without the virus producing neoplastic changes.1
HPV detection peaks in the late teens and early 20s, when approximately 25% of women test positive for the virus, resulting in high rates of low-grade squamous intraepithelial lesions (LSIL) and HPV-positive, atypical squamous cells of undetermined significance (ASC-US).2 These findings are mostly transient.2
Detection of CIN 3 does not peak until a woman reaches her late 20s, and the median detection of microinvasive cancer does not peak until she reaches her early 40s. These facts indicate that adolescents have the lowest risk of incipient cervical cancer but the highest risk of undergoing unnecessary procedures for HPV-related events—events that are highly likely to resolve without treatment.
From 1998 to 2006, an average of 14 cervical cancers occurred annually in women 15 to 19 years old, an incidence of only 1 or 2 cases of cervical cancer for every 1 million women in that age group ( TABLE 2 ).
TABLE 2
Incidence of invasive cervical carcinoma: United States, 1998-2003
| Age (y) | Average annual count | Incidence (95% CI) | Incidence as a percentage | Median age at diagnosis |
|---|---|---|---|---|
| All ages | 10,846 | 8.9 (8.8–9.0) | 100 | 47 |
| 0–14 | 0 | 0 | 0 | Not applicable (NA) |
| 15–19 | 14 | 0.2 (0.1–0.2) | 0.1 | NA |
| 20–24 | 123 | 1.6 (1.5–1.7) | 1.1 | NA |
| 25–29 | 543 | 6.9 (6.7–7.2) | 5.0 | NA |
| 30–34 | 1,045 | 12.3 (12.0–12.6) | 9.6 | NA |
| 35–39 | 1,350 | 14.6 (14.3–14.9) | 12.5 | NA |
| 40–44 | 1,534 | 16.3 (15.9–16.6) | 14.1 | NA |
| 45–49 | 1,323 | 15.4 (15.0–15.7) | 12.2 | NA |
| 50–59 | 1,958 | 14.5 (14.2–14.7) | 18.0 | NA |
| 60–69 | 1,352 | 14.8 (14.5–15.1) | 12.5 | NA |
| 70–79 | 1,008 | 12.9 (12.6–13.3) | 9.3 | NA |
| ≥80 | 595 | 11.2 (10.9–11.6) | 5.5 | NA |
| Source: Watson et al5 | ||||
In teens, screening does not reduce mortality
Even this low rate of cervical cancer might justify the screening of adolescents, provided such screening was shown to reduce the incidence of and mortality from cervical cancer in that age group. However, all data point to the opposite conclusion:
- The incidence of cervical cancer in this age group has not changed since the years between 1973 and 1977, a period that preceded the recommendation to begin screening at age 18 or first intercourse
- No data demonstrate a benefit of screening in women younger than 21 years in regard to future rates of CIN 2 and 3—or even that screening women 20 to 24 years old reduces the rate of cervical cancer in women 30 years or younger3
- CIN 2 and 3 do occur in adolescents, and the fear of delaying their diagnosis has driven much of the opposition to the guideline change—specifically, the omission of the option to begin screening within 3 years after first intercourse; however, even when high-grade CIN develops, spontaneous regression is common in this age group (e.g., 65% rate of regression of CIN 2 after 18 months; 75% after 36 months)
- When CIN 3 develops and persists, more than 10 years are typically required for the lesion to acquire the capacity to become invasive.1,2
In addition, extensive data suggest that screening adolescents may be harmful. Adverse psychological effects related to cervical cancer screening, evaluation of abnormal results, and treatment of CIN have been reported, including negative effects on sexual function and a higher risk of preterm and low-birth-weight infants.1
Virtually all studies of pregnancy outcomes following loop electrosurgical excision procedure (LEEP) have demonstrated a doubling or tripling of the rate of preterm birth.
Screening women 21 years or younger for cervical cancer may be harmful and lacks proven benefit. Screening should not begin until the patient is 21, regardless of the age of first intercourse.
Extend the screening interval to 2 years for women 21 to 29 years old
Both liquid-based and conventional methods of cervical cytology are acceptable for screening; hence, screening frequency should not vary based on the method used.1
The 2003 ACOG guidelines recommended annual cervical screening of women in their 20s using either conventional or liquid-based cytology. In contrast, in 2002, the American Cancer Society (ACS) recommended annual screening when the conventional Pap test was used, and a 2-year interval when screening involved liquid-based cytology. With ACOG’s latest recommendation—a 2-year interval for women 21 to 29 years old, regardless of test method—the College moves in line with the ACS in regard to liquid-based cytology. It also acknowledges more recent evidence that liquid-based cytology is no more sensitive than conventional cytology.1
Liquid-based cytology does have a number of other unquestionable advantages, however:
- It offers the convenience of being able to test for HPV, Neisseria gonorrhoeae, and Chlamydia trachomatis directly from the residual sample
- It produces fewer unsatisfactory cytology results than conventional cytology
- Cytotechnologists find liquid-based cytology easier to read.
More than 90% of Pap tests in the United States utilize liquid-based cytology, and that percentage is not likely to diminish.
Women 21 to 29 years old should have a Pap test every 2 years, regardless of the method used.
Some women 30 years and older can be screened every 3 years
Cervical cytology screening is recommended every 3 years for women age 30 years and older if:
- they have had three consecutive negative cervical cytology screening test results and have no history of CIN 2 or CIN 3, are not HIV-infected, are not immunocompromised, and were not exposed to diethylstilbestrol in utero or
- they have received negative test cotest results on both cervical cytology screening and HPV DNA testing and are considered low risk.1
A study of the detection of squamous cell cervical cancer (SCC) within 3.5 years of one, two, or three consecutive normal Pap tests demonstrated that the incidence of cervical cancer increases to 3 to 5 cases for every 100,000 woman-years in each of the subsequent 2 years. Some experts argue that this relatively low increase—the equivalent of the incidence of breast cancer in men—supports extension of the screening interval to 3 years after three consecutive normal Pap results.
Clinicians have generally been hesitant to widen the screening interval, despite ACS and ACOG recommendations for 2- or 3-year screening among women who have had three consecutive normal results. Many of these clinicians may find it difficult to dismiss even this low number of excess cancers (3 to 5 cases for every 100,000 woman-years) when more frequent or better screening would likely prevent them. As a result, the extension of screening intervals on the basis of negative cytology alone may continue to meet resistance from clinicians and their patients.
Wider intervals reduce the risk of unnecessary treatment
The extension of screening intervals, whether it is based on cytology alone or cytology combined with HPV testing, benefits most women by reducing the likelihood that transient, HPV-induced events will be detected and treated even though they are not destined to become CIN 3, adenocarcinoma in situ, or cervical cancer.
At the same time, however, extending the screening interval to 3 years in the setting of “opportunistic” screening—the screening approach used in the United States—may lead to irregular screening for many women at intervals beyond the recommended 3 years, thereby reducing the protective effect of a program based on cytology alone. Approximately 10% of cervical cancers occur in women who have not had a Pap test in the preceding 5 years.
Cotesting may be the solution
There is no question that extending cytology-only screening beyond 3 years significantly increases the risk of cervical cancer. However, among women tested for HPV, the risk of CIN 3 or greater does not begin to rise until at least 6 years following a negative test result, providing a margin of safety that would protect most women who miss the recommended 3-year screening interval.
Earlier this year, Ronco and colleagues published the results of a large primary cervical screening trial involving more than 94,000 women who were randomly assigned to screening with cytology alone or cotesting (i.e., cytology plus HPV testing).4 In the cytology-only group, women were referred to colposcopy for a Pap result of ASC-US or higher-grade findings. In the cotesting group, they were referred to colposcopy if the HPV or Pap test (or both) was positive. A second screening was performed an average of 3 years later, and the incidences of CIN 2, CIN 3, and cancer at each screening were compared between groups.
The number of cancers detected in the initial round of screening did not differ between groups. In the second round of screening, no cancers were found in the cotesting group, compared with nine cancers in the cytology-only group. The authors attributed this difference to the detection and treatment of twice as many cases of CIN 3 in the initial round of screening among women undergoing cotesting, compared with those tested with cytology alone.4
In addition, women in the cotesting group had an extremely low rate of CIN 3 in the second round of screening (2 cases for every 10,000 women). Investigators also noted that a high proportion of invasive cancers detected in the cytology group during the second round of screening were adenocarcinomas, consistent with reports from earlier studies that found cytology to be less effective in detecting adenocarcinomas than in detecting SCC.4
Although HPV testing was previously shown to outperform cytology in reducing the risk of cervical cancer in a low-resource country (India), this is the first study to do so in a developed country with a well-screened population and a low incidence of cervical cancer.4
Although ACOG guidelines encourage the extension of screening intervals to 3 years for women 30 years or older who have had three consecutive normal Pap tests, many clinicians have been reluctant to take this step. Cotesting with HPV and Pap tests should provide the reassurance necessary for these clinicians to adopt the wider screening intervals.
Keep annual screening for women who have a history of CIN, HIV, or certain chronic conditions
The recommendations to screen women every 2 years until age 30 and to extend the screening interval to 3 years thereafter, provided three consecutive Pap tests are normal or cotesting is negative, apply only to women at average risk of cervical cancer. Conditions that indicate elevated risk include:
- HIV infection
- immunosuppression for other reasons, e.g., organ transplant
- in utero exposure to diethylstilbestrol
- history of CIN 2, CIN 3, or cancer.
Two Pap tests are recommended in the first year after diagnosis of HIV infection, followed by annual screening. It can be presumed that women who have chronic immunosuppression should be managed similarly.
As for women known to have been exposed to diethylstilbestrol in utero, no specific recommendation is given other than “more frequent screening.”1
The relatively recent documentation that women with a history of CIN 2 or 3 (and probably adenocarcinoma in situ) remain at risk of developing cervical cancer for at least 20 years after treatment warrants annual screening for at least 20 years. The increased reassurance that no CIN 3 or greater is missed when cotesting is negative for both cytology and HPV testing might argue for extension of the screening interval for women who have negative cotest results and who have completed recommended posttreatment follow-up. However, at this time, we lack data on long-term follow-up of women who have been treated for cervical neoplasia and who have negative cotest results. Therefore, such a recommendation cannot be made at this time.
Do not increase the screening interval beyond annual testing for women who are HIV-positive, who are immunosuppressed, who were exposed in utero to diethylstilbestrol, or who have been treated for CIN 2 or 3 or adenocarcinoma in situ.
Routine cytology testing should be discontinued after total hysterectomy for benign indications, provided the woman has no history of high-grade cervical intraepithelial neoplasia or adenocarcinoma in situ.1 This recommendation has not changed since the 2003 ACOG guidelines on cervical cancer screening were published, and it is consistent with guidelines from the U.S. Preventive Services Task Force and the American Cancer Society.
1. Cervical cytology screening. ACOG Practice Bulletin #109. Obstet Gynecol. 2009;114(6):1409-1420.
2. Moscicki AB, Cox JT. Practice Improvement in Cervical Screening and Management (PICSM): symposium on management of cervical abnormalities in adolescents and young women. J Low Gen Tract Dis. 2010;14(1):73-80.
3. Sasieni P, Castanon A, Cuzick J. Effectiveness of cervical screening with age: population-based case-control study of prospectively recorded data. BMJ. 2009;339:b2968.-
4. Ronco G, Giorgi-Rossi P, Carozzi F, et al. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomized controlled trial. http://www.thelancet.com/oncology. Published online January 19, 2010. DOI:10.1016/S1470–2045(09)70360-2.
5. Watson M, Saraiya M, Bernard V, et al. Burden of cervical cancer in the United States, 1998–2003. Cancer. 2008;113(suppl 10):2855-2864.
6. Schiffman M, Castle PE. The promise of global cervical-cancer prevention. N Engl J Med. 2005;353(20):2101-2104.
“Astonishment fatigue.” That phenomenon may be responsible for clinicians’ muted reaction to new ACOG guidelines on cervical cancer screening, which were released late last year. 1 Through a coincidence of timing, the new guidelines hit the airwaves just after the U. S. Preventive Services Task Force announced controversial changes to its recommendations on mammography. As a result, the cervical cytology guidelines seemed to dissolve into the stratosphere.
Or, perhaps, the cervical cancer screening guidelines slipped by with little fanfare because they were soundly based in evidence and, therefore, widely accepted among ObGyns. Even if that is the case, the medical community may not be familiar with the specific data behind the guideline changes. In this article, I discuss the evidence driving all major changes to the guidelines based on Level-A evidence. Changes based on Level-B or -C evidence are listed in TABLE 1 .
Hold off on cervical cancer screening until the patient is 21 years old
Screening before age 21 should be avoided because it may lead to unnecessary and harmful evaluation and treatment in women at very low risk of cancer.1
How different is this from the 2003 ACOG recommendation to begin screening within 3 years of first intercourse or at age 21, whichever comes first?
Very, very different. In fact, it is the most dramatic change in the 2009 screening recommendations.
It is even more striking in comparison with ACOG’s earlier recommendation—which prevailed from the late 1970s through 2002—to begin cervical screening at age 18 or at the onset of intercourse, whichever comes first.
The median age of first intercourse in the United States is 16 years. Until this latest change in guidelines, most young women began cervical screening during adolescence.
What’s wrong with screening adolescents? Don’t they acquire human papillomavirus (HPV)? (Yes.) And once they do, aren’t they at risk of cervical cancer? (Yes.)
Several variables support the delay of screening to age 21:
- the transience of most HPV infections
- the typically long natural history of carcinogenesis in the few young women in whom HPV might persist
- the adverse consequences of over-screening and over-management of adolescents who have cervical intraepithelial neoplasia (CIN).
Let’s look more closely at these variables.
TABLE 1
Other ACOG cervical disease guidelines are based on Level-B and Level-C evidence*
| Recommendation | Level of evidence | Comment |
|---|---|---|
| Test sexually active adolescents (i.e., females 21 years or younger) for sexually transmitted infection, and counsel them about safe sexual practices and contraception | B | These measures can be carried out without cervical cytology and, in the asymptomatic patient, without the introduction of a speculum |
| It is reasonable to discontinue cervical cancer screening in any woman 65 to 70 years old who has had three or more consecutive negative Pap tests and no abnormal tests in the past 10 years | B | |
| Continue annual screening for at least 20 years in any woman who has been treated for CIN 2, CIN 3, or cancer. This population remains at risk of persistent or recurrent disease for at least 20 years after treatment and after initial posttreatment surveillance | B | |
| Continue to screen any woman who has had a total hysterectomy if she has a history of CIN 2 or CIN 3 or if a negative history cannot be documented. This screening should continue even after initial post-treatment surveillance | B | Although the screening interval may ultimately be extended, we lack reliable data to support or refute the discontinuation of screening in this population |
| Inform the patient that annual gynecologic examination may still be appropriate even if cervical cytology is not assessed at each visit | C | |
| Screen any woman who has been immunized against HPV 16 and 18 as though she has not been immunized | C | |
| * Level-B recommendations are based on limited and inconsistent scientific evidence. Level-C recommendations are based primarily on consensus and expert opinion. | ||
HPV is common but usually resolves on its own
It’s common for young women to acquire HPV shortly after they become sexually active, but their immune system clears most infections within 1 or 2 years without the virus producing neoplastic changes.1
HPV detection peaks in the late teens and early 20s, when approximately 25% of women test positive for the virus, resulting in high rates of low-grade squamous intraepithelial lesions (LSIL) and HPV-positive, atypical squamous cells of undetermined significance (ASC-US).2 These findings are mostly transient.2
Detection of CIN 3 does not peak until a woman reaches her late 20s, and the median detection of microinvasive cancer does not peak until she reaches her early 40s. These facts indicate that adolescents have the lowest risk of incipient cervical cancer but the highest risk of undergoing unnecessary procedures for HPV-related events—events that are highly likely to resolve without treatment.
From 1998 to 2006, an average of 14 cervical cancers occurred annually in women 15 to 19 years old, an incidence of only 1 or 2 cases of cervical cancer for every 1 million women in that age group ( TABLE 2 ).
TABLE 2
Incidence of invasive cervical carcinoma: United States, 1998-2003
| Age (y) | Average annual count | Incidence (95% CI) | Incidence as a percentage | Median age at diagnosis |
|---|---|---|---|---|
| All ages | 10,846 | 8.9 (8.8–9.0) | 100 | 47 |
| 0–14 | 0 | 0 | 0 | Not applicable (NA) |
| 15–19 | 14 | 0.2 (0.1–0.2) | 0.1 | NA |
| 20–24 | 123 | 1.6 (1.5–1.7) | 1.1 | NA |
| 25–29 | 543 | 6.9 (6.7–7.2) | 5.0 | NA |
| 30–34 | 1,045 | 12.3 (12.0–12.6) | 9.6 | NA |
| 35–39 | 1,350 | 14.6 (14.3–14.9) | 12.5 | NA |
| 40–44 | 1,534 | 16.3 (15.9–16.6) | 14.1 | NA |
| 45–49 | 1,323 | 15.4 (15.0–15.7) | 12.2 | NA |
| 50–59 | 1,958 | 14.5 (14.2–14.7) | 18.0 | NA |
| 60–69 | 1,352 | 14.8 (14.5–15.1) | 12.5 | NA |
| 70–79 | 1,008 | 12.9 (12.6–13.3) | 9.3 | NA |
| ≥80 | 595 | 11.2 (10.9–11.6) | 5.5 | NA |
| Source: Watson et al5 | ||||
In teens, screening does not reduce mortality
Even this low rate of cervical cancer might justify the screening of adolescents, provided such screening was shown to reduce the incidence of and mortality from cervical cancer in that age group. However, all data point to the opposite conclusion:
- The incidence of cervical cancer in this age group has not changed since the years between 1973 and 1977, a period that preceded the recommendation to begin screening at age 18 or first intercourse
- No data demonstrate a benefit of screening in women younger than 21 years in regard to future rates of CIN 2 and 3—or even that screening women 20 to 24 years old reduces the rate of cervical cancer in women 30 years or younger3
- CIN 2 and 3 do occur in adolescents, and the fear of delaying their diagnosis has driven much of the opposition to the guideline change—specifically, the omission of the option to begin screening within 3 years after first intercourse; however, even when high-grade CIN develops, spontaneous regression is common in this age group (e.g., 65% rate of regression of CIN 2 after 18 months; 75% after 36 months)
- When CIN 3 develops and persists, more than 10 years are typically required for the lesion to acquire the capacity to become invasive.1,2
In addition, extensive data suggest that screening adolescents may be harmful. Adverse psychological effects related to cervical cancer screening, evaluation of abnormal results, and treatment of CIN have been reported, including negative effects on sexual function and a higher risk of preterm and low-birth-weight infants.1
Virtually all studies of pregnancy outcomes following loop electrosurgical excision procedure (LEEP) have demonstrated a doubling or tripling of the rate of preterm birth.
Screening women 21 years or younger for cervical cancer may be harmful and lacks proven benefit. Screening should not begin until the patient is 21, regardless of the age of first intercourse.
Extend the screening interval to 2 years for women 21 to 29 years old
Both liquid-based and conventional methods of cervical cytology are acceptable for screening; hence, screening frequency should not vary based on the method used.1
The 2003 ACOG guidelines recommended annual cervical screening of women in their 20s using either conventional or liquid-based cytology. In contrast, in 2002, the American Cancer Society (ACS) recommended annual screening when the conventional Pap test was used, and a 2-year interval when screening involved liquid-based cytology. With ACOG’s latest recommendation—a 2-year interval for women 21 to 29 years old, regardless of test method—the College moves in line with the ACS in regard to liquid-based cytology. It also acknowledges more recent evidence that liquid-based cytology is no more sensitive than conventional cytology.1
Liquid-based cytology does have a number of other unquestionable advantages, however:
- It offers the convenience of being able to test for HPV, Neisseria gonorrhoeae, and Chlamydia trachomatis directly from the residual sample
- It produces fewer unsatisfactory cytology results than conventional cytology
- Cytotechnologists find liquid-based cytology easier to read.
More than 90% of Pap tests in the United States utilize liquid-based cytology, and that percentage is not likely to diminish.
Women 21 to 29 years old should have a Pap test every 2 years, regardless of the method used.
Some women 30 years and older can be screened every 3 years
Cervical cytology screening is recommended every 3 years for women age 30 years and older if:
- they have had three consecutive negative cervical cytology screening test results and have no history of CIN 2 or CIN 3, are not HIV-infected, are not immunocompromised, and were not exposed to diethylstilbestrol in utero or
- they have received negative test cotest results on both cervical cytology screening and HPV DNA testing and are considered low risk.1
A study of the detection of squamous cell cervical cancer (SCC) within 3.5 years of one, two, or three consecutive normal Pap tests demonstrated that the incidence of cervical cancer increases to 3 to 5 cases for every 100,000 woman-years in each of the subsequent 2 years. Some experts argue that this relatively low increase—the equivalent of the incidence of breast cancer in men—supports extension of the screening interval to 3 years after three consecutive normal Pap results.
Clinicians have generally been hesitant to widen the screening interval, despite ACS and ACOG recommendations for 2- or 3-year screening among women who have had three consecutive normal results. Many of these clinicians may find it difficult to dismiss even this low number of excess cancers (3 to 5 cases for every 100,000 woman-years) when more frequent or better screening would likely prevent them. As a result, the extension of screening intervals on the basis of negative cytology alone may continue to meet resistance from clinicians and their patients.
Wider intervals reduce the risk of unnecessary treatment
The extension of screening intervals, whether it is based on cytology alone or cytology combined with HPV testing, benefits most women by reducing the likelihood that transient, HPV-induced events will be detected and treated even though they are not destined to become CIN 3, adenocarcinoma in situ, or cervical cancer.
At the same time, however, extending the screening interval to 3 years in the setting of “opportunistic” screening—the screening approach used in the United States—may lead to irregular screening for many women at intervals beyond the recommended 3 years, thereby reducing the protective effect of a program based on cytology alone. Approximately 10% of cervical cancers occur in women who have not had a Pap test in the preceding 5 years.
Cotesting may be the solution
There is no question that extending cytology-only screening beyond 3 years significantly increases the risk of cervical cancer. However, among women tested for HPV, the risk of CIN 3 or greater does not begin to rise until at least 6 years following a negative test result, providing a margin of safety that would protect most women who miss the recommended 3-year screening interval.
Earlier this year, Ronco and colleagues published the results of a large primary cervical screening trial involving more than 94,000 women who were randomly assigned to screening with cytology alone or cotesting (i.e., cytology plus HPV testing).4 In the cytology-only group, women were referred to colposcopy for a Pap result of ASC-US or higher-grade findings. In the cotesting group, they were referred to colposcopy if the HPV or Pap test (or both) was positive. A second screening was performed an average of 3 years later, and the incidences of CIN 2, CIN 3, and cancer at each screening were compared between groups.
The number of cancers detected in the initial round of screening did not differ between groups. In the second round of screening, no cancers were found in the cotesting group, compared with nine cancers in the cytology-only group. The authors attributed this difference to the detection and treatment of twice as many cases of CIN 3 in the initial round of screening among women undergoing cotesting, compared with those tested with cytology alone.4
In addition, women in the cotesting group had an extremely low rate of CIN 3 in the second round of screening (2 cases for every 10,000 women). Investigators also noted that a high proportion of invasive cancers detected in the cytology group during the second round of screening were adenocarcinomas, consistent with reports from earlier studies that found cytology to be less effective in detecting adenocarcinomas than in detecting SCC.4
Although HPV testing was previously shown to outperform cytology in reducing the risk of cervical cancer in a low-resource country (India), this is the first study to do so in a developed country with a well-screened population and a low incidence of cervical cancer.4
Although ACOG guidelines encourage the extension of screening intervals to 3 years for women 30 years or older who have had three consecutive normal Pap tests, many clinicians have been reluctant to take this step. Cotesting with HPV and Pap tests should provide the reassurance necessary for these clinicians to adopt the wider screening intervals.
Keep annual screening for women who have a history of CIN, HIV, or certain chronic conditions
The recommendations to screen women every 2 years until age 30 and to extend the screening interval to 3 years thereafter, provided three consecutive Pap tests are normal or cotesting is negative, apply only to women at average risk of cervical cancer. Conditions that indicate elevated risk include:
- HIV infection
- immunosuppression for other reasons, e.g., organ transplant
- in utero exposure to diethylstilbestrol
- history of CIN 2, CIN 3, or cancer.
Two Pap tests are recommended in the first year after diagnosis of HIV infection, followed by annual screening. It can be presumed that women who have chronic immunosuppression should be managed similarly.
As for women known to have been exposed to diethylstilbestrol in utero, no specific recommendation is given other than “more frequent screening.”1
The relatively recent documentation that women with a history of CIN 2 or 3 (and probably adenocarcinoma in situ) remain at risk of developing cervical cancer for at least 20 years after treatment warrants annual screening for at least 20 years. The increased reassurance that no CIN 3 or greater is missed when cotesting is negative for both cytology and HPV testing might argue for extension of the screening interval for women who have negative cotest results and who have completed recommended posttreatment follow-up. However, at this time, we lack data on long-term follow-up of women who have been treated for cervical neoplasia and who have negative cotest results. Therefore, such a recommendation cannot be made at this time.
Do not increase the screening interval beyond annual testing for women who are HIV-positive, who are immunosuppressed, who were exposed in utero to diethylstilbestrol, or who have been treated for CIN 2 or 3 or adenocarcinoma in situ.
Routine cytology testing should be discontinued after total hysterectomy for benign indications, provided the woman has no history of high-grade cervical intraepithelial neoplasia or adenocarcinoma in situ.1 This recommendation has not changed since the 2003 ACOG guidelines on cervical cancer screening were published, and it is consistent with guidelines from the U.S. Preventive Services Task Force and the American Cancer Society.
“Astonishment fatigue.” That phenomenon may be responsible for clinicians’ muted reaction to new ACOG guidelines on cervical cancer screening, which were released late last year. 1 Through a coincidence of timing, the new guidelines hit the airwaves just after the U. S. Preventive Services Task Force announced controversial changes to its recommendations on mammography. As a result, the cervical cytology guidelines seemed to dissolve into the stratosphere.
Or, perhaps, the cervical cancer screening guidelines slipped by with little fanfare because they were soundly based in evidence and, therefore, widely accepted among ObGyns. Even if that is the case, the medical community may not be familiar with the specific data behind the guideline changes. In this article, I discuss the evidence driving all major changes to the guidelines based on Level-A evidence. Changes based on Level-B or -C evidence are listed in TABLE 1 .
Hold off on cervical cancer screening until the patient is 21 years old
Screening before age 21 should be avoided because it may lead to unnecessary and harmful evaluation and treatment in women at very low risk of cancer.1
How different is this from the 2003 ACOG recommendation to begin screening within 3 years of first intercourse or at age 21, whichever comes first?
Very, very different. In fact, it is the most dramatic change in the 2009 screening recommendations.
It is even more striking in comparison with ACOG’s earlier recommendation—which prevailed from the late 1970s through 2002—to begin cervical screening at age 18 or at the onset of intercourse, whichever comes first.
The median age of first intercourse in the United States is 16 years. Until this latest change in guidelines, most young women began cervical screening during adolescence.
What’s wrong with screening adolescents? Don’t they acquire human papillomavirus (HPV)? (Yes.) And once they do, aren’t they at risk of cervical cancer? (Yes.)
Several variables support the delay of screening to age 21:
- the transience of most HPV infections
- the typically long natural history of carcinogenesis in the few young women in whom HPV might persist
- the adverse consequences of over-screening and over-management of adolescents who have cervical intraepithelial neoplasia (CIN).
Let’s look more closely at these variables.
TABLE 1
Other ACOG cervical disease guidelines are based on Level-B and Level-C evidence*
| Recommendation | Level of evidence | Comment |
|---|---|---|
| Test sexually active adolescents (i.e., females 21 years or younger) for sexually transmitted infection, and counsel them about safe sexual practices and contraception | B | These measures can be carried out without cervical cytology and, in the asymptomatic patient, without the introduction of a speculum |
| It is reasonable to discontinue cervical cancer screening in any woman 65 to 70 years old who has had three or more consecutive negative Pap tests and no abnormal tests in the past 10 years | B | |
| Continue annual screening for at least 20 years in any woman who has been treated for CIN 2, CIN 3, or cancer. This population remains at risk of persistent or recurrent disease for at least 20 years after treatment and after initial posttreatment surveillance | B | |
| Continue to screen any woman who has had a total hysterectomy if she has a history of CIN 2 or CIN 3 or if a negative history cannot be documented. This screening should continue even after initial post-treatment surveillance | B | Although the screening interval may ultimately be extended, we lack reliable data to support or refute the discontinuation of screening in this population |
| Inform the patient that annual gynecologic examination may still be appropriate even if cervical cytology is not assessed at each visit | C | |
| Screen any woman who has been immunized against HPV 16 and 18 as though she has not been immunized | C | |
| * Level-B recommendations are based on limited and inconsistent scientific evidence. Level-C recommendations are based primarily on consensus and expert opinion. | ||
HPV is common but usually resolves on its own
It’s common for young women to acquire HPV shortly after they become sexually active, but their immune system clears most infections within 1 or 2 years without the virus producing neoplastic changes.1
HPV detection peaks in the late teens and early 20s, when approximately 25% of women test positive for the virus, resulting in high rates of low-grade squamous intraepithelial lesions (LSIL) and HPV-positive, atypical squamous cells of undetermined significance (ASC-US).2 These findings are mostly transient.2
Detection of CIN 3 does not peak until a woman reaches her late 20s, and the median detection of microinvasive cancer does not peak until she reaches her early 40s. These facts indicate that adolescents have the lowest risk of incipient cervical cancer but the highest risk of undergoing unnecessary procedures for HPV-related events—events that are highly likely to resolve without treatment.
From 1998 to 2006, an average of 14 cervical cancers occurred annually in women 15 to 19 years old, an incidence of only 1 or 2 cases of cervical cancer for every 1 million women in that age group ( TABLE 2 ).
TABLE 2
Incidence of invasive cervical carcinoma: United States, 1998-2003
| Age (y) | Average annual count | Incidence (95% CI) | Incidence as a percentage | Median age at diagnosis |
|---|---|---|---|---|
| All ages | 10,846 | 8.9 (8.8–9.0) | 100 | 47 |
| 0–14 | 0 | 0 | 0 | Not applicable (NA) |
| 15–19 | 14 | 0.2 (0.1–0.2) | 0.1 | NA |
| 20–24 | 123 | 1.6 (1.5–1.7) | 1.1 | NA |
| 25–29 | 543 | 6.9 (6.7–7.2) | 5.0 | NA |
| 30–34 | 1,045 | 12.3 (12.0–12.6) | 9.6 | NA |
| 35–39 | 1,350 | 14.6 (14.3–14.9) | 12.5 | NA |
| 40–44 | 1,534 | 16.3 (15.9–16.6) | 14.1 | NA |
| 45–49 | 1,323 | 15.4 (15.0–15.7) | 12.2 | NA |
| 50–59 | 1,958 | 14.5 (14.2–14.7) | 18.0 | NA |
| 60–69 | 1,352 | 14.8 (14.5–15.1) | 12.5 | NA |
| 70–79 | 1,008 | 12.9 (12.6–13.3) | 9.3 | NA |
| ≥80 | 595 | 11.2 (10.9–11.6) | 5.5 | NA |
| Source: Watson et al5 | ||||
In teens, screening does not reduce mortality
Even this low rate of cervical cancer might justify the screening of adolescents, provided such screening was shown to reduce the incidence of and mortality from cervical cancer in that age group. However, all data point to the opposite conclusion:
- The incidence of cervical cancer in this age group has not changed since the years between 1973 and 1977, a period that preceded the recommendation to begin screening at age 18 or first intercourse
- No data demonstrate a benefit of screening in women younger than 21 years in regard to future rates of CIN 2 and 3—or even that screening women 20 to 24 years old reduces the rate of cervical cancer in women 30 years or younger3
- CIN 2 and 3 do occur in adolescents, and the fear of delaying their diagnosis has driven much of the opposition to the guideline change—specifically, the omission of the option to begin screening within 3 years after first intercourse; however, even when high-grade CIN develops, spontaneous regression is common in this age group (e.g., 65% rate of regression of CIN 2 after 18 months; 75% after 36 months)
- When CIN 3 develops and persists, more than 10 years are typically required for the lesion to acquire the capacity to become invasive.1,2
In addition, extensive data suggest that screening adolescents may be harmful. Adverse psychological effects related to cervical cancer screening, evaluation of abnormal results, and treatment of CIN have been reported, including negative effects on sexual function and a higher risk of preterm and low-birth-weight infants.1
Virtually all studies of pregnancy outcomes following loop electrosurgical excision procedure (LEEP) have demonstrated a doubling or tripling of the rate of preterm birth.
Screening women 21 years or younger for cervical cancer may be harmful and lacks proven benefit. Screening should not begin until the patient is 21, regardless of the age of first intercourse.
Extend the screening interval to 2 years for women 21 to 29 years old
Both liquid-based and conventional methods of cervical cytology are acceptable for screening; hence, screening frequency should not vary based on the method used.1
The 2003 ACOG guidelines recommended annual cervical screening of women in their 20s using either conventional or liquid-based cytology. In contrast, in 2002, the American Cancer Society (ACS) recommended annual screening when the conventional Pap test was used, and a 2-year interval when screening involved liquid-based cytology. With ACOG’s latest recommendation—a 2-year interval for women 21 to 29 years old, regardless of test method—the College moves in line with the ACS in regard to liquid-based cytology. It also acknowledges more recent evidence that liquid-based cytology is no more sensitive than conventional cytology.1
Liquid-based cytology does have a number of other unquestionable advantages, however:
- It offers the convenience of being able to test for HPV, Neisseria gonorrhoeae, and Chlamydia trachomatis directly from the residual sample
- It produces fewer unsatisfactory cytology results than conventional cytology
- Cytotechnologists find liquid-based cytology easier to read.
More than 90% of Pap tests in the United States utilize liquid-based cytology, and that percentage is not likely to diminish.
Women 21 to 29 years old should have a Pap test every 2 years, regardless of the method used.
Some women 30 years and older can be screened every 3 years
Cervical cytology screening is recommended every 3 years for women age 30 years and older if:
- they have had three consecutive negative cervical cytology screening test results and have no history of CIN 2 or CIN 3, are not HIV-infected, are not immunocompromised, and were not exposed to diethylstilbestrol in utero or
- they have received negative test cotest results on both cervical cytology screening and HPV DNA testing and are considered low risk.1
A study of the detection of squamous cell cervical cancer (SCC) within 3.5 years of one, two, or three consecutive normal Pap tests demonstrated that the incidence of cervical cancer increases to 3 to 5 cases for every 100,000 woman-years in each of the subsequent 2 years. Some experts argue that this relatively low increase—the equivalent of the incidence of breast cancer in men—supports extension of the screening interval to 3 years after three consecutive normal Pap results.
Clinicians have generally been hesitant to widen the screening interval, despite ACS and ACOG recommendations for 2- or 3-year screening among women who have had three consecutive normal results. Many of these clinicians may find it difficult to dismiss even this low number of excess cancers (3 to 5 cases for every 100,000 woman-years) when more frequent or better screening would likely prevent them. As a result, the extension of screening intervals on the basis of negative cytology alone may continue to meet resistance from clinicians and their patients.
Wider intervals reduce the risk of unnecessary treatment
The extension of screening intervals, whether it is based on cytology alone or cytology combined with HPV testing, benefits most women by reducing the likelihood that transient, HPV-induced events will be detected and treated even though they are not destined to become CIN 3, adenocarcinoma in situ, or cervical cancer.
At the same time, however, extending the screening interval to 3 years in the setting of “opportunistic” screening—the screening approach used in the United States—may lead to irregular screening for many women at intervals beyond the recommended 3 years, thereby reducing the protective effect of a program based on cytology alone. Approximately 10% of cervical cancers occur in women who have not had a Pap test in the preceding 5 years.
Cotesting may be the solution
There is no question that extending cytology-only screening beyond 3 years significantly increases the risk of cervical cancer. However, among women tested for HPV, the risk of CIN 3 or greater does not begin to rise until at least 6 years following a negative test result, providing a margin of safety that would protect most women who miss the recommended 3-year screening interval.
Earlier this year, Ronco and colleagues published the results of a large primary cervical screening trial involving more than 94,000 women who were randomly assigned to screening with cytology alone or cotesting (i.e., cytology plus HPV testing).4 In the cytology-only group, women were referred to colposcopy for a Pap result of ASC-US or higher-grade findings. In the cotesting group, they were referred to colposcopy if the HPV or Pap test (or both) was positive. A second screening was performed an average of 3 years later, and the incidences of CIN 2, CIN 3, and cancer at each screening were compared between groups.
The number of cancers detected in the initial round of screening did not differ between groups. In the second round of screening, no cancers were found in the cotesting group, compared with nine cancers in the cytology-only group. The authors attributed this difference to the detection and treatment of twice as many cases of CIN 3 in the initial round of screening among women undergoing cotesting, compared with those tested with cytology alone.4
In addition, women in the cotesting group had an extremely low rate of CIN 3 in the second round of screening (2 cases for every 10,000 women). Investigators also noted that a high proportion of invasive cancers detected in the cytology group during the second round of screening were adenocarcinomas, consistent with reports from earlier studies that found cytology to be less effective in detecting adenocarcinomas than in detecting SCC.4
Although HPV testing was previously shown to outperform cytology in reducing the risk of cervical cancer in a low-resource country (India), this is the first study to do so in a developed country with a well-screened population and a low incidence of cervical cancer.4
Although ACOG guidelines encourage the extension of screening intervals to 3 years for women 30 years or older who have had three consecutive normal Pap tests, many clinicians have been reluctant to take this step. Cotesting with HPV and Pap tests should provide the reassurance necessary for these clinicians to adopt the wider screening intervals.
Keep annual screening for women who have a history of CIN, HIV, or certain chronic conditions
The recommendations to screen women every 2 years until age 30 and to extend the screening interval to 3 years thereafter, provided three consecutive Pap tests are normal or cotesting is negative, apply only to women at average risk of cervical cancer. Conditions that indicate elevated risk include:
- HIV infection
- immunosuppression for other reasons, e.g., organ transplant
- in utero exposure to diethylstilbestrol
- history of CIN 2, CIN 3, or cancer.
Two Pap tests are recommended in the first year after diagnosis of HIV infection, followed by annual screening. It can be presumed that women who have chronic immunosuppression should be managed similarly.
As for women known to have been exposed to diethylstilbestrol in utero, no specific recommendation is given other than “more frequent screening.”1
The relatively recent documentation that women with a history of CIN 2 or 3 (and probably adenocarcinoma in situ) remain at risk of developing cervical cancer for at least 20 years after treatment warrants annual screening for at least 20 years. The increased reassurance that no CIN 3 or greater is missed when cotesting is negative for both cytology and HPV testing might argue for extension of the screening interval for women who have negative cotest results and who have completed recommended posttreatment follow-up. However, at this time, we lack data on long-term follow-up of women who have been treated for cervical neoplasia and who have negative cotest results. Therefore, such a recommendation cannot be made at this time.
Do not increase the screening interval beyond annual testing for women who are HIV-positive, who are immunosuppressed, who were exposed in utero to diethylstilbestrol, or who have been treated for CIN 2 or 3 or adenocarcinoma in situ.
Routine cytology testing should be discontinued after total hysterectomy for benign indications, provided the woman has no history of high-grade cervical intraepithelial neoplasia or adenocarcinoma in situ.1 This recommendation has not changed since the 2003 ACOG guidelines on cervical cancer screening were published, and it is consistent with guidelines from the U.S. Preventive Services Task Force and the American Cancer Society.
1. Cervical cytology screening. ACOG Practice Bulletin #109. Obstet Gynecol. 2009;114(6):1409-1420.
2. Moscicki AB, Cox JT. Practice Improvement in Cervical Screening and Management (PICSM): symposium on management of cervical abnormalities in adolescents and young women. J Low Gen Tract Dis. 2010;14(1):73-80.
3. Sasieni P, Castanon A, Cuzick J. Effectiveness of cervical screening with age: population-based case-control study of prospectively recorded data. BMJ. 2009;339:b2968.-
4. Ronco G, Giorgi-Rossi P, Carozzi F, et al. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomized controlled trial. http://www.thelancet.com/oncology. Published online January 19, 2010. DOI:10.1016/S1470–2045(09)70360-2.
5. Watson M, Saraiya M, Bernard V, et al. Burden of cervical cancer in the United States, 1998–2003. Cancer. 2008;113(suppl 10):2855-2864.
6. Schiffman M, Castle PE. The promise of global cervical-cancer prevention. N Engl J Med. 2005;353(20):2101-2104.
1. Cervical cytology screening. ACOG Practice Bulletin #109. Obstet Gynecol. 2009;114(6):1409-1420.
2. Moscicki AB, Cox JT. Practice Improvement in Cervical Screening and Management (PICSM): symposium on management of cervical abnormalities in adolescents and young women. J Low Gen Tract Dis. 2010;14(1):73-80.
3. Sasieni P, Castanon A, Cuzick J. Effectiveness of cervical screening with age: population-based case-control study of prospectively recorded data. BMJ. 2009;339:b2968.-
4. Ronco G, Giorgi-Rossi P, Carozzi F, et al. Efficacy of human papillomavirus testing for the detection of invasive cervical cancers and cervical intraepithelial neoplasia: a randomized controlled trial. http://www.thelancet.com/oncology. Published online January 19, 2010. DOI:10.1016/S1470–2045(09)70360-2.
5. Watson M, Saraiya M, Bernard V, et al. Burden of cervical cancer in the United States, 1998–2003. Cancer. 2008;113(suppl 10):2855-2864.
6. Schiffman M, Castle PE. The promise of global cervical-cancer prevention. N Engl J Med. 2005;353(20):2101-2104.
Effects of Exenatide on Diabetes Management and Weight Loss in a VA Population
Slow Walking Speed May Predict Cardiovascular Death
Grand Rounds: Girl, 6, With Rapid Heart Rate
A 6-year-old girl was brought by her parents to the emergency department (ED) with an elevated heart rate. According to the parents, the girl was carrying her younger sister when they both fell, landing on their buttocks. The child told them that her heart was beating fast, and the parents said she appeared to be on the verge of fainting.
They stated that their daughter was healthy and active; they denied previous episodes of shortness of breath, headache, weakness, tachycardia, syncope, or fatigue with exercise. Her caffeine intake, they claimed, was limited to one small cup of soda they allowed her each week.
Initial evaluation in the ED revealed an anxious child with tachycardia and shortness of breath. She presented with a temperature of 98.3°F (36.8°C); pulse, 210 beats/min; respirations, 33 breaths/min; blood pressure, 100/72 mm Hg; weight, 78 lb; height, 45 in; and BMI, 27.1. ECG revealed a heart rate exceeding 210 beats/min, and a pediatric cardiologist made a diagnosis of supraventricular tachycardia (SVT).
The pediatric cardiologist prescribed an adenosine IV drip, which successfully stabilized the child’s heart to sinus rhythm. After three hours in the ED, the patient was discharged with a stable heart rate of 100 beats/min. (It is well known that heart rate regulation changes significantly during development; this is most obvious in higher basal rates in infants and children, compared with adults.1)
The parents were advised to administer atenolol 12.5 mg (one tablet) twice daily and to make a follow-up appointment with a pediatric electrophysiologist. (Although atenolol is not currently FDA approved for this use, a multicenter prospective randomized controlled trial comparing digoxin with beta-blockers for the treatment of SVT in children is presently under way.2)
At that appointment, the pediatric electrophysiologist provided information to the parents regarding the therapeutic options for SVT. The parents continued to administer atenolol to the child, as was deemed necessary until any accessory electrical pathway could be identified and, if so, an ablation procedure could be performed. They were uncertain how to proceed so long as their daughter experienced no recurrent episodes of SVT while receiving pharmacologic therapy.
However, six months after the initial episode, the child (then age 7) presented to the ED once again with recurrent SVT. The pediatric cardiologist ordered an adenosine IV drip, which resulted in successful conversion to sinus rhythm. The parents were instructed to increase the child’s atenolol dosage to 25 mg twice a day.
Six months later, after extensive research and consultation, the parents agreed to an ablation procedure in order to prevent further episodes of SVT. Upon their informed consent, the child was sent to a cardiac catheterization laboratory for an electrophysiology study (EPS), which confirmed the presence of an accessory pathway, as well as the diagnosis of atrioventricular reciprocating tachycardia (AVRT). The procedure was followed by radiofrequency catheter ablation to correct the 7-year-old patient’s accessory pathway–mediated reentry tachycardia.
Discussion
SVT, also known as paroxysmal supraventricular tachycardia (PSVT), is one of the most common symptomatic pediatric arrhythmias, affecting between one in 25,000 and one in 250 children.3 It is defined as rapid heart rhythm (140 to 240 beats/min) that is caused by the presence of additional electrical connections and/or congenital muscle fibers between the atrium and the ventricle or within the atrioventricular (AV) node that did not, for unknown reasons, separate completely during development.4 SVT can be triggered by physical or psychological stress automaticity.3
Approximately 50% of children with SVT present with a first episode before age 1. SVT usually occurs in early childhood, between ages 6 and 9.4 Almost 90% of pediatric patients with SVT are diagnosed with a reentry mechanism.3 The symptoms experienced may be resolved pharmacologically or by means of an invasive therapy. Serious sequelae associated with SVT include heart failure and cardiac arrest.
For children with rare and mildly symptomatic episodes in whom SVT is easily terminated, the SVT may not warrant treatment. However, it may be advisable to offer medical therapy or transcatheter ablation as therapeutic options for children with episodes that are difficult to terminate, occur frequently, or occur during participation in athletics.4
Pathophysiology
SVT generally presents as one of three types: AVRT, which is also known as Wolff-Parkinson-White syndrome; atrioventricular nodal reentry tachycardia (AVNRT); and automatic tachycardia (AT).
AVRT, the most common type of SVT, comprises about 90% of pediatric cases. It is defined by the presence of one or more accessory conduction pathways that are anatomically separated from the normal cardiac conduction system.5 AVRT may be orthodromic (that is, the arrhythmia circuit proceeds down the AV node and retrograde up the accessory conduction pathway) or antedromic (ie, proceeding down the accessory pathway and up the AV node5; see figure.6,7)
AVNRT, considered the second most common type of SVT in children, accounts for about 10% of pediatric cases. AVNRT is caused by an interaction between the two types of pathways within the AV node—one with a fast conduction time and a short refractory period, and the other with a slow conduction time and a long refractory period. AVNRT occurs when the antegrade conduction block in the fast pathway results in conduction over the slow pathway and back up the fast pathway, forming a microreentrant circuit.5
AT is the result of rapid depolarization from an automatic focus originating within the atria but outside the sinus node.3
Patient Presentation and History
The typical presentation of AVRT in children of school age includes palpitations, chest pain or tightness, dizziness, anxiety, decrease in exercise tolerance, easy fatigability, and/or shortness of breath.3 Onset is described as abrupt, while termination of SVT is described as slower because the catecholamine levels are typically elevated.4
The frequency and duration of SVT can vary greatly, from a few minutes to a few hours; it can occur as regularly as daily or as uncommonly as once or twice per year.4 Additionally, SVT symptoms can go unrecognized until a cardiac dysfunction develops. As for the patient in the case study, no apparent factor in her history was identified that may have induced SVT.
The differential diagnosis for SVT is broad, including sinus tachycardia, multifocal atrial tachycardia, and SVT with aberrancy.8 Additional considerations include stress, anxiety, hyperthyroidism, electrolyte abnormalities, and dehydration—any of which can present with a tachycardia response.4 Furthermore, clinicians are often unlikely to diagnose a child with any cardiac problem because chest pain is more commonly a presenting symptom of a pulmonary or musculoskeletal condition than a cardiac problem.3
Physical Examination
SVT can be diagnosed based on medical history and physical examination. During the physical examination, providers will assess the patient’s blood pressure and pulse, auscultate heart and lung sounds, assess the veins in the patient’s neck for different types of pulsations, and conduct cardiac maneuvers, including the Valsalva maneuver and carotid sinus massage.9,10
Laboratory Work-up and Diagnosis
Three specific tests help clinicians monitor and evaluate a patient’s conduction system. ECG is important to assess the heart rhythm both at baseline and when symptoms are occurring, if possible.3 Ambulatory ECG (ie, Holter monitoring, event recorders) record the patient’s heart rhythm on a continuous basis.
An EPS, which is performed to classify the mechanism of SVT, is conducted by inserting one or more electrocatheters into the heart by way of the femoral vein or other peripheral vessel.3 Pacing and sensing electrodes at the ends of the catheters record local intracardiac electrical activity and timing information, providing a detailed analysis of the heart’s electrical activity. The EPS is critical to determine the presence of one or more extra electrical pathways within the heart and to localize it by region.3,11 An ablation procedure may follow.
Management Options
SVT can be treated pharmacologically or nonpharmacologically. First-line pharmacologic options include certain beta-blockers (including atenolol and propranolol), digoxin, and calcium channel blockers. Second-line pharmacologic treatments include amiodarone, flecainide, and sotalol,4 all of which are contraindicated in children younger than 1 year because of these patients’ hemodynamic dependency on the heart and inability to generate stroke volume.3 Pharmacologic treatment of SVT is associated with a 68% success rate in children4 (see Table 14).
For children in whom pharmacologic treatment is ineffective, an ablation procedure may be performed. Radiofrequency catheter ablation is currently considered first-line therapy for AVRT and AVNRT.12 In this invasive procedure, intracardiac electrical mapping is performed and the initiating focus of the arrhythmia or the accessory electrical pathway that has been identified within the heart is destroyed by radiofrequency energy, delivered by electrocatheter. Ablations performed during the acute phase of SVT have a 95% success rate.3,13
Cryoablation is a relatively new treatment in which liquid nitrous oxide is used to cool the catheter to subfreezing temperatures, enabling it to destroy the myocardial tissue by freezing.3,14 The advantage of cryoablation is the option of reversible cooling, which allows the electrophysiologist to test the area first, confirming the accuracy of the apparent location accessory pathway.15
Noninvasive, nonpharmacologic interventions that increase the refractoriness of the AV node may be successful in terminating the tachyarrhythmia during episodes of SVT (see Table 23,9,13,16). They are used to terminate and diagnose tachydysrhythmias, increase parasympathetic tone, and slow conduction through the AV node.3
Patient Education
It is very important for health care providers to relieve parents’ and caregivers’ stress, anxiety, and uncertainty by educating them about the child’s cardiac condition of SVT. Information to convey include an understanding of what SVT is, what may cause it, what triggers the patient should avoid, what treatments are available and appropriate (including the maneuvers shown in Table 2), and what outcomes may be expected. An excellent patient/family education handout from the Children’s Hospitals and Clinics of Minnesota17 is available at www.childrensmn.org/Manuals/PFS/Condill/018303.pdf.
Follow-Up
Primary care providers must emphasize the importance of monitoring the patient’s progress, based on the severity of his or her SVT symptoms. The provider may choose to monitor the patient for a few weeks or a few months, assessing the frequency of arrhythmia recurrence and the heart rate, to adjust or substitute medications based on repeat ECG or Holter evaluations, and to plan further therapy, should the condition worsen.5
The Case Patient
One month after undergoing radiofrequency catheter ablation, the child presented to the pediatric cardiologist for follow-up. Since the procedure, she had been without any symptoms referable to the cardiovascular system. She denied experiencing any fast heart rate, palpitations, chest pain, shortness of breath, or dizziness. ECG demonstrated normal sinus rhythm.
Two years after undergoing radiofrequency ablation, the child is functioning at a normal activity level with no recurrence of SVT episodes.
Conclusion
The purpose of this case study is to improve primary care providers’ understanding of SVT in children and to convey the importance of identifying the condition in a timely manner and referring patients to a pediatric cardiologist or electrophysiologist. For most children affected by SVT, a regimen of pharmacologic and/or nonpharmacologic treatment—supported by detailed education for their parents and caregivers—can allow them to live a healthy, normal life.
1. Kudielka BM, Buske-Kirschbaum A, Hellhammer DH, Kirschbaum C. Differential heart rate reactivity and recovery after psychosocial stress (TSST) in healthy children, younger adults, and elderly adults: the impact of age and gender. Int J Behav Med. 2004;11(2):116-121.
2. Multicenter Study of Antiarrhythmic Medications for Treatment of Infants With Supraventricular Tachycardia. www.clinicaltrials.gov/ct2/results?term=NCT00390546. Accessed January 26, 2010.
3. Schlechte EA, Boramanand N, Funk M. Supraventricular tachycardia in the pediatric primary care setting: age-related presentation, diagnosis, and management. J Pediatr Health Care. 2008;22(5): 289-299.
4. Salerno JC, Seslar SP. Supraventricular tachycardia. Arch Pediatr Adolesc Med. 2009;163(3): 268-274.
5. Doniger SJ, Sharieff GQ. Pediatric dysrhythmias. Pediatr Clin North Am. 2006;53(1):85-105, vi.
6. Mavroudis C, Deal BJ, Backer CL, Tsao S. Arrhythmia surgery in patients with and without congenital heart disease. Ann Thorac Surg. 2008;86(3):857-868.
7. Wang PJ, Estes NAM III. Supraventricular tachycardia. Circulation. 2002;106(25):e206-e208.
8. Buttaro TM, Trybulski J, Bailey PP, Sandberg-Cook J. Primary Care: A Collaborative Practice. 3rd ed. St. Louis, MO: Mosby Elsevier; 2008.
9. Wen ZC, Chen SA, Tai CT, et al. Electrophysiological mechanisms and determinants of vagal maneuvers for termination of paroxysmal supraventricular tachycardia. Circulation.1998;98(24):2716-2723.
10. Julian MR. Treatment of paroxysmal supraventricular tachycardia using instrument-assisted manipulation of the fourth rib: a 6-year case study. J Manipulative Physiol Ther. 2008;31(5):389-391.
11. Calkins H, Kumar VKA, Francis J. Radiofrequency catheter ablation of supraventricular tachycardia. Indian Pacing Electrophysiol J. 2002;2(2):45-49.
12. Nakagawa H, Jackman WM. Catheter ablation of paroxysmal supraventricular tachycardia. Circulation. 2007;116(21):2465-2478.
13. Kugler JD, Danford DA, Houston K, Felix G; Pediatric Radiofrequency Ablation Registry of the Pediatric Electrophysiology Society. Pediatric radiofrequency catheter ablation registry success, fluoroscopy time, and complication rate for supraventricular tachycardia: comparison of early and recent eras. J Cardiovasc Electrophysiol. 2002;13(4):336-341.
14. Chun TU, Van Hare GF. Advances in the approach to treatment of supraventricular tachycardia in the pediatric population. Curr Cardiol Rep. 2004; 6(5):322-326.
15. Friedman PL, Dubuc M, Green MS, et al. Catheter cryoablation of supraventricular tachycardia: results of the multicenter prospective “frosty” trial. Heart Rhythm. 2004;1(2):129-138.
16. Bosen DM. Atrio-ventricular nodal reentry tachycardia in children. Dimens Crit Care Nurs. 2002; 21(4):134-139.
17. Children’s Hospitals and Clinics of Minnesota. Patient and family education: supraventricular tachycardia (2009). www.childrensmn.org/Manuals/PFS/Condill/018303.pdf. Accessed January 26, 2010.
A 6-year-old girl was brought by her parents to the emergency department (ED) with an elevated heart rate. According to the parents, the girl was carrying her younger sister when they both fell, landing on their buttocks. The child told them that her heart was beating fast, and the parents said she appeared to be on the verge of fainting.
They stated that their daughter was healthy and active; they denied previous episodes of shortness of breath, headache, weakness, tachycardia, syncope, or fatigue with exercise. Her caffeine intake, they claimed, was limited to one small cup of soda they allowed her each week.
Initial evaluation in the ED revealed an anxious child with tachycardia and shortness of breath. She presented with a temperature of 98.3°F (36.8°C); pulse, 210 beats/min; respirations, 33 breaths/min; blood pressure, 100/72 mm Hg; weight, 78 lb; height, 45 in; and BMI, 27.1. ECG revealed a heart rate exceeding 210 beats/min, and a pediatric cardiologist made a diagnosis of supraventricular tachycardia (SVT).
The pediatric cardiologist prescribed an adenosine IV drip, which successfully stabilized the child’s heart to sinus rhythm. After three hours in the ED, the patient was discharged with a stable heart rate of 100 beats/min. (It is well known that heart rate regulation changes significantly during development; this is most obvious in higher basal rates in infants and children, compared with adults.1)
The parents were advised to administer atenolol 12.5 mg (one tablet) twice daily and to make a follow-up appointment with a pediatric electrophysiologist. (Although atenolol is not currently FDA approved for this use, a multicenter prospective randomized controlled trial comparing digoxin with beta-blockers for the treatment of SVT in children is presently under way.2)
At that appointment, the pediatric electrophysiologist provided information to the parents regarding the therapeutic options for SVT. The parents continued to administer atenolol to the child, as was deemed necessary until any accessory electrical pathway could be identified and, if so, an ablation procedure could be performed. They were uncertain how to proceed so long as their daughter experienced no recurrent episodes of SVT while receiving pharmacologic therapy.
However, six months after the initial episode, the child (then age 7) presented to the ED once again with recurrent SVT. The pediatric cardiologist ordered an adenosine IV drip, which resulted in successful conversion to sinus rhythm. The parents were instructed to increase the child’s atenolol dosage to 25 mg twice a day.
Six months later, after extensive research and consultation, the parents agreed to an ablation procedure in order to prevent further episodes of SVT. Upon their informed consent, the child was sent to a cardiac catheterization laboratory for an electrophysiology study (EPS), which confirmed the presence of an accessory pathway, as well as the diagnosis of atrioventricular reciprocating tachycardia (AVRT). The procedure was followed by radiofrequency catheter ablation to correct the 7-year-old patient’s accessory pathway–mediated reentry tachycardia.
Discussion
SVT, also known as paroxysmal supraventricular tachycardia (PSVT), is one of the most common symptomatic pediatric arrhythmias, affecting between one in 25,000 and one in 250 children.3 It is defined as rapid heart rhythm (140 to 240 beats/min) that is caused by the presence of additional electrical connections and/or congenital muscle fibers between the atrium and the ventricle or within the atrioventricular (AV) node that did not, for unknown reasons, separate completely during development.4 SVT can be triggered by physical or psychological stress automaticity.3
Approximately 50% of children with SVT present with a first episode before age 1. SVT usually occurs in early childhood, between ages 6 and 9.4 Almost 90% of pediatric patients with SVT are diagnosed with a reentry mechanism.3 The symptoms experienced may be resolved pharmacologically or by means of an invasive therapy. Serious sequelae associated with SVT include heart failure and cardiac arrest.
For children with rare and mildly symptomatic episodes in whom SVT is easily terminated, the SVT may not warrant treatment. However, it may be advisable to offer medical therapy or transcatheter ablation as therapeutic options for children with episodes that are difficult to terminate, occur frequently, or occur during participation in athletics.4
Pathophysiology
SVT generally presents as one of three types: AVRT, which is also known as Wolff-Parkinson-White syndrome; atrioventricular nodal reentry tachycardia (AVNRT); and automatic tachycardia (AT).
AVRT, the most common type of SVT, comprises about 90% of pediatric cases. It is defined by the presence of one or more accessory conduction pathways that are anatomically separated from the normal cardiac conduction system.5 AVRT may be orthodromic (that is, the arrhythmia circuit proceeds down the AV node and retrograde up the accessory conduction pathway) or antedromic (ie, proceeding down the accessory pathway and up the AV node5; see figure.6,7)
AVNRT, considered the second most common type of SVT in children, accounts for about 10% of pediatric cases. AVNRT is caused by an interaction between the two types of pathways within the AV node—one with a fast conduction time and a short refractory period, and the other with a slow conduction time and a long refractory period. AVNRT occurs when the antegrade conduction block in the fast pathway results in conduction over the slow pathway and back up the fast pathway, forming a microreentrant circuit.5
AT is the result of rapid depolarization from an automatic focus originating within the atria but outside the sinus node.3
Patient Presentation and History
The typical presentation of AVRT in children of school age includes palpitations, chest pain or tightness, dizziness, anxiety, decrease in exercise tolerance, easy fatigability, and/or shortness of breath.3 Onset is described as abrupt, while termination of SVT is described as slower because the catecholamine levels are typically elevated.4
The frequency and duration of SVT can vary greatly, from a few minutes to a few hours; it can occur as regularly as daily or as uncommonly as once or twice per year.4 Additionally, SVT symptoms can go unrecognized until a cardiac dysfunction develops. As for the patient in the case study, no apparent factor in her history was identified that may have induced SVT.
The differential diagnosis for SVT is broad, including sinus tachycardia, multifocal atrial tachycardia, and SVT with aberrancy.8 Additional considerations include stress, anxiety, hyperthyroidism, electrolyte abnormalities, and dehydration—any of which can present with a tachycardia response.4 Furthermore, clinicians are often unlikely to diagnose a child with any cardiac problem because chest pain is more commonly a presenting symptom of a pulmonary or musculoskeletal condition than a cardiac problem.3
Physical Examination
SVT can be diagnosed based on medical history and physical examination. During the physical examination, providers will assess the patient’s blood pressure and pulse, auscultate heart and lung sounds, assess the veins in the patient’s neck for different types of pulsations, and conduct cardiac maneuvers, including the Valsalva maneuver and carotid sinus massage.9,10
Laboratory Work-up and Diagnosis
Three specific tests help clinicians monitor and evaluate a patient’s conduction system. ECG is important to assess the heart rhythm both at baseline and when symptoms are occurring, if possible.3 Ambulatory ECG (ie, Holter monitoring, event recorders) record the patient’s heart rhythm on a continuous basis.
An EPS, which is performed to classify the mechanism of SVT, is conducted by inserting one or more electrocatheters into the heart by way of the femoral vein or other peripheral vessel.3 Pacing and sensing electrodes at the ends of the catheters record local intracardiac electrical activity and timing information, providing a detailed analysis of the heart’s electrical activity. The EPS is critical to determine the presence of one or more extra electrical pathways within the heart and to localize it by region.3,11 An ablation procedure may follow.
Management Options
SVT can be treated pharmacologically or nonpharmacologically. First-line pharmacologic options include certain beta-blockers (including atenolol and propranolol), digoxin, and calcium channel blockers. Second-line pharmacologic treatments include amiodarone, flecainide, and sotalol,4 all of which are contraindicated in children younger than 1 year because of these patients’ hemodynamic dependency on the heart and inability to generate stroke volume.3 Pharmacologic treatment of SVT is associated with a 68% success rate in children4 (see Table 14).
For children in whom pharmacologic treatment is ineffective, an ablation procedure may be performed. Radiofrequency catheter ablation is currently considered first-line therapy for AVRT and AVNRT.12 In this invasive procedure, intracardiac electrical mapping is performed and the initiating focus of the arrhythmia or the accessory electrical pathway that has been identified within the heart is destroyed by radiofrequency energy, delivered by electrocatheter. Ablations performed during the acute phase of SVT have a 95% success rate.3,13
Cryoablation is a relatively new treatment in which liquid nitrous oxide is used to cool the catheter to subfreezing temperatures, enabling it to destroy the myocardial tissue by freezing.3,14 The advantage of cryoablation is the option of reversible cooling, which allows the electrophysiologist to test the area first, confirming the accuracy of the apparent location accessory pathway.15
Noninvasive, nonpharmacologic interventions that increase the refractoriness of the AV node may be successful in terminating the tachyarrhythmia during episodes of SVT (see Table 23,9,13,16). They are used to terminate and diagnose tachydysrhythmias, increase parasympathetic tone, and slow conduction through the AV node.3
Patient Education
It is very important for health care providers to relieve parents’ and caregivers’ stress, anxiety, and uncertainty by educating them about the child’s cardiac condition of SVT. Information to convey include an understanding of what SVT is, what may cause it, what triggers the patient should avoid, what treatments are available and appropriate (including the maneuvers shown in Table 2), and what outcomes may be expected. An excellent patient/family education handout from the Children’s Hospitals and Clinics of Minnesota17 is available at www.childrensmn.org/Manuals/PFS/Condill/018303.pdf.
Follow-Up
Primary care providers must emphasize the importance of monitoring the patient’s progress, based on the severity of his or her SVT symptoms. The provider may choose to monitor the patient for a few weeks or a few months, assessing the frequency of arrhythmia recurrence and the heart rate, to adjust or substitute medications based on repeat ECG or Holter evaluations, and to plan further therapy, should the condition worsen.5
The Case Patient
One month after undergoing radiofrequency catheter ablation, the child presented to the pediatric cardiologist for follow-up. Since the procedure, she had been without any symptoms referable to the cardiovascular system. She denied experiencing any fast heart rate, palpitations, chest pain, shortness of breath, or dizziness. ECG demonstrated normal sinus rhythm.
Two years after undergoing radiofrequency ablation, the child is functioning at a normal activity level with no recurrence of SVT episodes.
Conclusion
The purpose of this case study is to improve primary care providers’ understanding of SVT in children and to convey the importance of identifying the condition in a timely manner and referring patients to a pediatric cardiologist or electrophysiologist. For most children affected by SVT, a regimen of pharmacologic and/or nonpharmacologic treatment—supported by detailed education for their parents and caregivers—can allow them to live a healthy, normal life.
A 6-year-old girl was brought by her parents to the emergency department (ED) with an elevated heart rate. According to the parents, the girl was carrying her younger sister when they both fell, landing on their buttocks. The child told them that her heart was beating fast, and the parents said she appeared to be on the verge of fainting.
They stated that their daughter was healthy and active; they denied previous episodes of shortness of breath, headache, weakness, tachycardia, syncope, or fatigue with exercise. Her caffeine intake, they claimed, was limited to one small cup of soda they allowed her each week.
Initial evaluation in the ED revealed an anxious child with tachycardia and shortness of breath. She presented with a temperature of 98.3°F (36.8°C); pulse, 210 beats/min; respirations, 33 breaths/min; blood pressure, 100/72 mm Hg; weight, 78 lb; height, 45 in; and BMI, 27.1. ECG revealed a heart rate exceeding 210 beats/min, and a pediatric cardiologist made a diagnosis of supraventricular tachycardia (SVT).
The pediatric cardiologist prescribed an adenosine IV drip, which successfully stabilized the child’s heart to sinus rhythm. After three hours in the ED, the patient was discharged with a stable heart rate of 100 beats/min. (It is well known that heart rate regulation changes significantly during development; this is most obvious in higher basal rates in infants and children, compared with adults.1)
The parents were advised to administer atenolol 12.5 mg (one tablet) twice daily and to make a follow-up appointment with a pediatric electrophysiologist. (Although atenolol is not currently FDA approved for this use, a multicenter prospective randomized controlled trial comparing digoxin with beta-blockers for the treatment of SVT in children is presently under way.2)
At that appointment, the pediatric electrophysiologist provided information to the parents regarding the therapeutic options for SVT. The parents continued to administer atenolol to the child, as was deemed necessary until any accessory electrical pathway could be identified and, if so, an ablation procedure could be performed. They were uncertain how to proceed so long as their daughter experienced no recurrent episodes of SVT while receiving pharmacologic therapy.
However, six months after the initial episode, the child (then age 7) presented to the ED once again with recurrent SVT. The pediatric cardiologist ordered an adenosine IV drip, which resulted in successful conversion to sinus rhythm. The parents were instructed to increase the child’s atenolol dosage to 25 mg twice a day.
Six months later, after extensive research and consultation, the parents agreed to an ablation procedure in order to prevent further episodes of SVT. Upon their informed consent, the child was sent to a cardiac catheterization laboratory for an electrophysiology study (EPS), which confirmed the presence of an accessory pathway, as well as the diagnosis of atrioventricular reciprocating tachycardia (AVRT). The procedure was followed by radiofrequency catheter ablation to correct the 7-year-old patient’s accessory pathway–mediated reentry tachycardia.
Discussion
SVT, also known as paroxysmal supraventricular tachycardia (PSVT), is one of the most common symptomatic pediatric arrhythmias, affecting between one in 25,000 and one in 250 children.3 It is defined as rapid heart rhythm (140 to 240 beats/min) that is caused by the presence of additional electrical connections and/or congenital muscle fibers between the atrium and the ventricle or within the atrioventricular (AV) node that did not, for unknown reasons, separate completely during development.4 SVT can be triggered by physical or psychological stress automaticity.3
Approximately 50% of children with SVT present with a first episode before age 1. SVT usually occurs in early childhood, between ages 6 and 9.4 Almost 90% of pediatric patients with SVT are diagnosed with a reentry mechanism.3 The symptoms experienced may be resolved pharmacologically or by means of an invasive therapy. Serious sequelae associated with SVT include heart failure and cardiac arrest.
For children with rare and mildly symptomatic episodes in whom SVT is easily terminated, the SVT may not warrant treatment. However, it may be advisable to offer medical therapy or transcatheter ablation as therapeutic options for children with episodes that are difficult to terminate, occur frequently, or occur during participation in athletics.4
Pathophysiology
SVT generally presents as one of three types: AVRT, which is also known as Wolff-Parkinson-White syndrome; atrioventricular nodal reentry tachycardia (AVNRT); and automatic tachycardia (AT).
AVRT, the most common type of SVT, comprises about 90% of pediatric cases. It is defined by the presence of one or more accessory conduction pathways that are anatomically separated from the normal cardiac conduction system.5 AVRT may be orthodromic (that is, the arrhythmia circuit proceeds down the AV node and retrograde up the accessory conduction pathway) or antedromic (ie, proceeding down the accessory pathway and up the AV node5; see figure.6,7)
AVNRT, considered the second most common type of SVT in children, accounts for about 10% of pediatric cases. AVNRT is caused by an interaction between the two types of pathways within the AV node—one with a fast conduction time and a short refractory period, and the other with a slow conduction time and a long refractory period. AVNRT occurs when the antegrade conduction block in the fast pathway results in conduction over the slow pathway and back up the fast pathway, forming a microreentrant circuit.5
AT is the result of rapid depolarization from an automatic focus originating within the atria but outside the sinus node.3
Patient Presentation and History
The typical presentation of AVRT in children of school age includes palpitations, chest pain or tightness, dizziness, anxiety, decrease in exercise tolerance, easy fatigability, and/or shortness of breath.3 Onset is described as abrupt, while termination of SVT is described as slower because the catecholamine levels are typically elevated.4
The frequency and duration of SVT can vary greatly, from a few minutes to a few hours; it can occur as regularly as daily or as uncommonly as once or twice per year.4 Additionally, SVT symptoms can go unrecognized until a cardiac dysfunction develops. As for the patient in the case study, no apparent factor in her history was identified that may have induced SVT.
The differential diagnosis for SVT is broad, including sinus tachycardia, multifocal atrial tachycardia, and SVT with aberrancy.8 Additional considerations include stress, anxiety, hyperthyroidism, electrolyte abnormalities, and dehydration—any of which can present with a tachycardia response.4 Furthermore, clinicians are often unlikely to diagnose a child with any cardiac problem because chest pain is more commonly a presenting symptom of a pulmonary or musculoskeletal condition than a cardiac problem.3
Physical Examination
SVT can be diagnosed based on medical history and physical examination. During the physical examination, providers will assess the patient’s blood pressure and pulse, auscultate heart and lung sounds, assess the veins in the patient’s neck for different types of pulsations, and conduct cardiac maneuvers, including the Valsalva maneuver and carotid sinus massage.9,10
Laboratory Work-up and Diagnosis
Three specific tests help clinicians monitor and evaluate a patient’s conduction system. ECG is important to assess the heart rhythm both at baseline and when symptoms are occurring, if possible.3 Ambulatory ECG (ie, Holter monitoring, event recorders) record the patient’s heart rhythm on a continuous basis.
An EPS, which is performed to classify the mechanism of SVT, is conducted by inserting one or more electrocatheters into the heart by way of the femoral vein or other peripheral vessel.3 Pacing and sensing electrodes at the ends of the catheters record local intracardiac electrical activity and timing information, providing a detailed analysis of the heart’s electrical activity. The EPS is critical to determine the presence of one or more extra electrical pathways within the heart and to localize it by region.3,11 An ablation procedure may follow.
Management Options
SVT can be treated pharmacologically or nonpharmacologically. First-line pharmacologic options include certain beta-blockers (including atenolol and propranolol), digoxin, and calcium channel blockers. Second-line pharmacologic treatments include amiodarone, flecainide, and sotalol,4 all of which are contraindicated in children younger than 1 year because of these patients’ hemodynamic dependency on the heart and inability to generate stroke volume.3 Pharmacologic treatment of SVT is associated with a 68% success rate in children4 (see Table 14).
For children in whom pharmacologic treatment is ineffective, an ablation procedure may be performed. Radiofrequency catheter ablation is currently considered first-line therapy for AVRT and AVNRT.12 In this invasive procedure, intracardiac electrical mapping is performed and the initiating focus of the arrhythmia or the accessory electrical pathway that has been identified within the heart is destroyed by radiofrequency energy, delivered by electrocatheter. Ablations performed during the acute phase of SVT have a 95% success rate.3,13
Cryoablation is a relatively new treatment in which liquid nitrous oxide is used to cool the catheter to subfreezing temperatures, enabling it to destroy the myocardial tissue by freezing.3,14 The advantage of cryoablation is the option of reversible cooling, which allows the electrophysiologist to test the area first, confirming the accuracy of the apparent location accessory pathway.15
Noninvasive, nonpharmacologic interventions that increase the refractoriness of the AV node may be successful in terminating the tachyarrhythmia during episodes of SVT (see Table 23,9,13,16). They are used to terminate and diagnose tachydysrhythmias, increase parasympathetic tone, and slow conduction through the AV node.3
Patient Education
It is very important for health care providers to relieve parents’ and caregivers’ stress, anxiety, and uncertainty by educating them about the child’s cardiac condition of SVT. Information to convey include an understanding of what SVT is, what may cause it, what triggers the patient should avoid, what treatments are available and appropriate (including the maneuvers shown in Table 2), and what outcomes may be expected. An excellent patient/family education handout from the Children’s Hospitals and Clinics of Minnesota17 is available at www.childrensmn.org/Manuals/PFS/Condill/018303.pdf.
Follow-Up
Primary care providers must emphasize the importance of monitoring the patient’s progress, based on the severity of his or her SVT symptoms. The provider may choose to monitor the patient for a few weeks or a few months, assessing the frequency of arrhythmia recurrence and the heart rate, to adjust or substitute medications based on repeat ECG or Holter evaluations, and to plan further therapy, should the condition worsen.5
The Case Patient
One month after undergoing radiofrequency catheter ablation, the child presented to the pediatric cardiologist for follow-up. Since the procedure, she had been without any symptoms referable to the cardiovascular system. She denied experiencing any fast heart rate, palpitations, chest pain, shortness of breath, or dizziness. ECG demonstrated normal sinus rhythm.
Two years after undergoing radiofrequency ablation, the child is functioning at a normal activity level with no recurrence of SVT episodes.
Conclusion
The purpose of this case study is to improve primary care providers’ understanding of SVT in children and to convey the importance of identifying the condition in a timely manner and referring patients to a pediatric cardiologist or electrophysiologist. For most children affected by SVT, a regimen of pharmacologic and/or nonpharmacologic treatment—supported by detailed education for their parents and caregivers—can allow them to live a healthy, normal life.
1. Kudielka BM, Buske-Kirschbaum A, Hellhammer DH, Kirschbaum C. Differential heart rate reactivity and recovery after psychosocial stress (TSST) in healthy children, younger adults, and elderly adults: the impact of age and gender. Int J Behav Med. 2004;11(2):116-121.
2. Multicenter Study of Antiarrhythmic Medications for Treatment of Infants With Supraventricular Tachycardia. www.clinicaltrials.gov/ct2/results?term=NCT00390546. Accessed January 26, 2010.
3. Schlechte EA, Boramanand N, Funk M. Supraventricular tachycardia in the pediatric primary care setting: age-related presentation, diagnosis, and management. J Pediatr Health Care. 2008;22(5): 289-299.
4. Salerno JC, Seslar SP. Supraventricular tachycardia. Arch Pediatr Adolesc Med. 2009;163(3): 268-274.
5. Doniger SJ, Sharieff GQ. Pediatric dysrhythmias. Pediatr Clin North Am. 2006;53(1):85-105, vi.
6. Mavroudis C, Deal BJ, Backer CL, Tsao S. Arrhythmia surgery in patients with and without congenital heart disease. Ann Thorac Surg. 2008;86(3):857-868.
7. Wang PJ, Estes NAM III. Supraventricular tachycardia. Circulation. 2002;106(25):e206-e208.
8. Buttaro TM, Trybulski J, Bailey PP, Sandberg-Cook J. Primary Care: A Collaborative Practice. 3rd ed. St. Louis, MO: Mosby Elsevier; 2008.
9. Wen ZC, Chen SA, Tai CT, et al. Electrophysiological mechanisms and determinants of vagal maneuvers for termination of paroxysmal supraventricular tachycardia. Circulation.1998;98(24):2716-2723.
10. Julian MR. Treatment of paroxysmal supraventricular tachycardia using instrument-assisted manipulation of the fourth rib: a 6-year case study. J Manipulative Physiol Ther. 2008;31(5):389-391.
11. Calkins H, Kumar VKA, Francis J. Radiofrequency catheter ablation of supraventricular tachycardia. Indian Pacing Electrophysiol J. 2002;2(2):45-49.
12. Nakagawa H, Jackman WM. Catheter ablation of paroxysmal supraventricular tachycardia. Circulation. 2007;116(21):2465-2478.
13. Kugler JD, Danford DA, Houston K, Felix G; Pediatric Radiofrequency Ablation Registry of the Pediatric Electrophysiology Society. Pediatric radiofrequency catheter ablation registry success, fluoroscopy time, and complication rate for supraventricular tachycardia: comparison of early and recent eras. J Cardiovasc Electrophysiol. 2002;13(4):336-341.
14. Chun TU, Van Hare GF. Advances in the approach to treatment of supraventricular tachycardia in the pediatric population. Curr Cardiol Rep. 2004; 6(5):322-326.
15. Friedman PL, Dubuc M, Green MS, et al. Catheter cryoablation of supraventricular tachycardia: results of the multicenter prospective “frosty” trial. Heart Rhythm. 2004;1(2):129-138.
16. Bosen DM. Atrio-ventricular nodal reentry tachycardia in children. Dimens Crit Care Nurs. 2002; 21(4):134-139.
17. Children’s Hospitals and Clinics of Minnesota. Patient and family education: supraventricular tachycardia (2009). www.childrensmn.org/Manuals/PFS/Condill/018303.pdf. Accessed January 26, 2010.
1. Kudielka BM, Buske-Kirschbaum A, Hellhammer DH, Kirschbaum C. Differential heart rate reactivity and recovery after psychosocial stress (TSST) in healthy children, younger adults, and elderly adults: the impact of age and gender. Int J Behav Med. 2004;11(2):116-121.
2. Multicenter Study of Antiarrhythmic Medications for Treatment of Infants With Supraventricular Tachycardia. www.clinicaltrials.gov/ct2/results?term=NCT00390546. Accessed January 26, 2010.
3. Schlechte EA, Boramanand N, Funk M. Supraventricular tachycardia in the pediatric primary care setting: age-related presentation, diagnosis, and management. J Pediatr Health Care. 2008;22(5): 289-299.
4. Salerno JC, Seslar SP. Supraventricular tachycardia. Arch Pediatr Adolesc Med. 2009;163(3): 268-274.
5. Doniger SJ, Sharieff GQ. Pediatric dysrhythmias. Pediatr Clin North Am. 2006;53(1):85-105, vi.
6. Mavroudis C, Deal BJ, Backer CL, Tsao S. Arrhythmia surgery in patients with and without congenital heart disease. Ann Thorac Surg. 2008;86(3):857-868.
7. Wang PJ, Estes NAM III. Supraventricular tachycardia. Circulation. 2002;106(25):e206-e208.
8. Buttaro TM, Trybulski J, Bailey PP, Sandberg-Cook J. Primary Care: A Collaborative Practice. 3rd ed. St. Louis, MO: Mosby Elsevier; 2008.
9. Wen ZC, Chen SA, Tai CT, et al. Electrophysiological mechanisms and determinants of vagal maneuvers for termination of paroxysmal supraventricular tachycardia. Circulation.1998;98(24):2716-2723.
10. Julian MR. Treatment of paroxysmal supraventricular tachycardia using instrument-assisted manipulation of the fourth rib: a 6-year case study. J Manipulative Physiol Ther. 2008;31(5):389-391.
11. Calkins H, Kumar VKA, Francis J. Radiofrequency catheter ablation of supraventricular tachycardia. Indian Pacing Electrophysiol J. 2002;2(2):45-49.
12. Nakagawa H, Jackman WM. Catheter ablation of paroxysmal supraventricular tachycardia. Circulation. 2007;116(21):2465-2478.
13. Kugler JD, Danford DA, Houston K, Felix G; Pediatric Radiofrequency Ablation Registry of the Pediatric Electrophysiology Society. Pediatric radiofrequency catheter ablation registry success, fluoroscopy time, and complication rate for supraventricular tachycardia: comparison of early and recent eras. J Cardiovasc Electrophysiol. 2002;13(4):336-341.
14. Chun TU, Van Hare GF. Advances in the approach to treatment of supraventricular tachycardia in the pediatric population. Curr Cardiol Rep. 2004; 6(5):322-326.
15. Friedman PL, Dubuc M, Green MS, et al. Catheter cryoablation of supraventricular tachycardia: results of the multicenter prospective “frosty” trial. Heart Rhythm. 2004;1(2):129-138.
16. Bosen DM. Atrio-ventricular nodal reentry tachycardia in children. Dimens Crit Care Nurs. 2002; 21(4):134-139.
17. Children’s Hospitals and Clinics of Minnesota. Patient and family education: supraventricular tachycardia (2009). www.childrensmn.org/Manuals/PFS/Condill/018303.pdf. Accessed January 26, 2010.
