User login
What you should know about heterotopic pregnancy
The authors report no financial relationships relevant to this article.
CASE 1: Pelvic pain and few signs of intrauterine pregnancy
A 24-year-old woman, para 1-0-0-1, visits the hospital emergency department complaining of pelvic pain. She says the pain arose suddenly and reports that she had a positive urine pregnancy test earlier in the week. When asked about her obstetric history, she reports vaginal delivery of an 8 lb, 8 oz infant at 38 weeks’ gestation 2 years earlier. Her human chorionic gonadotropin (hCG) level is 3,000 mIU/mL, but ultrasonography (US) reveals no evidence of pregnancy. She is discharged with instructions to follow up with her physician in 2 days.
When her abdominal pain worsens, she returns to the emergency department. Physical examination reveals significant tenderness of the abdomen and moderate to severe tenderness of the cervix upon motion. Transvaginal US shows a uterus of normal size with a 5-mm endometrial lining and no gestational sac. The patient’s abdomen is full of fluid, with large, hypodense areas adjacent to the uterus bilaterally but larger on the right. The preoperative diagnosis: ruptured ectopic pregnancy.
During the diagnostic laparoscopy that follows, approximately 500 mL of blood is discovered in the abdomen and pelvis, and a gestational sac is found to be densely adherent to the right pelvic sidewall, where the ureter nears the uterine vessels. The sac, which has partially separated from the sidewall, is bleeding.
The surgeon peels the sac off the sidewall and controls bleeding with electrocautery and liquid thrombin. The final pathology report describes the tissue as an organizing blood clot with trophoblasts, consistent with ectopic pregnancy.
At a follow-up visit 3 weeks later, the patient reports persistent symptoms of pregnancy. Repeat US reveals a twin intrauterine pregnancy with two sacs, only one of which has a heartbeat. One week later, US shows confluence of the sacs, with a single viable fetus at 8 weeks and 2 days of gestation.
Could heterotopic pregnancy have been diagnosed earlier?
This case illustrates challenges inherent in the diagnosis of heterotopic pregnancy, which is much more common today than it was when it was first described 300 years ago. Incidence has increased from approximately 1 in 30,000 pregnancies to 1 in 2,600 pregnancies annually. When assisted reproductive technologies (ART) are used, the incidence may be as high as 1 in 100 pregnancies.1
The rising incidence suggests that the diagnosis of intrauterine pregnancy can no longer be used to exclude the presence of ectopic pregnancy, and vice versa. Instead, steps must be taken to rule out both when a woman exhibits pain and signs of pregnancy.
In this article, we discuss the causes, diagnosis, and treatment of heterotopic pregnancy, including the necessity of a high index of suspicion, the unreliability of US imaging in 50% of cases, and the need to avoid curettage in the treatment of ectopic pregnancy until an empty uterus can be confirmed.
Duverney was the first to report heterotopic pregnancy, in 1708, after finding an intrauterine pregnancy during the autopsy of a woman who had died from a ruptured ectopic pregnancy.1 It was 165 years, however, before the first review of the phenomenon was written.2 By 1970, only 479 such cases had been reported.20
Determining incidence remains a challenge—except that it is rising
In 1948, DeVoe and Pratt calculated the incidence of heterotopic pregnancy by multiplying the incidence of two-ovum pregnancy by the incidence of ectopic pregnancy, reaching an estimate of 1 in 30,000 pregnancies annually.2
In 1965, Rothman and Shapiro found that only about 500 cases of heterotopic pregnancy had been reported cumulatively.7 They reasoned that, if the incidence of fraternal twins is 1 in 110 and the incidence of ectopic pregnancy is 1 in 250, heterotopic pregnancy should occur at a rate of 1 in 27,500 gestations.7 They also speculated that many heterotopic pregnancies go undiagnosed because of early pregnancy loss.7
In 1971, Payne and colleagues hypothesized that ovulation-induction agents increase the incidence of heterotopic pregnancy, and McLain and associates reached a similar conclusion in 1987.9 The incidence of multiple pregnancy after oral ovulation induction is 8% to 10%, and it is 20% to 50% with injectable agents.9 In 1994, Crabtree and associates reported that both abdominal and heterotopic pregnancies appear to be increasing in incidence.17
Today, the incidence of heterotopic pregnancy is thought to be about 1 in 2,600 pregnancies annually—primarily because of assisted reproduction.2 The calculated risk of heterotopic pregnancy is 1 in 119, and it rises to 1 in 45 with embryo transfer.8
Why is heterotopic pregnancy on the increase?
One reason may be the increase in ectopic pregnancy. Among the factors contributing to the rising incidence of ectopic pregnancy are:
- pelvic adhesive disease
- effects of diethylstilbestrol (DES) on the genital tract
- antibiotic-induced tubal disease
- use of an intrauterine device (IUD)
- voluntary restriction of family size
- iatrogenic curettage of intrauterine pregnancy during surgery for ectopic pregnancy
- pregnancy termination
- history of surgery to treat infertility, ectopic pregnancy, or tubal adhesions
- improvement in the assay used to measure gonadotropin
- improvement in ultrasonography.2-4
A previous ectopic pregnancy is a risk factor for ectopic pregnancy as well as for heterotopic pregnancy.5
Pelvic infection, antibiotic-induced tubal disease, previous ectopic pregnancy, pelvic adhesions, and tubal surgery with cauterization of the tubes and subsequent bowel adhesions distort the fallopian tubes and may render them unable to propel a migrated embryo into the uterine cavity.2,6 Ectopic pregnancy may result from internal migration of a fertilized ovum or transperitoneal migration of sperm.7
DES exposure can distort the uterine cavity.8 Congenital and acquired uterine malformations increase the risk of ectopic pregnancy.3
In addition, ovulation-inducing drugs and ovarian stimulation increase the number of eggs available for conception, with a greater risk of multiple gestation and heterotopic pregnancy.3,9,10
The greatest increase in heterotopic pregnancy has been seen with ART involving the transfer of multiple embryos into the uterus, as well as gamete intrafallopian transfer, also known as GIFT.10,11 When five or more embryos are transferred, the risk of heterotopic pregnancy increases to 1 in 45 pregnancies annually.11 Inadvertent placement of the catheter tip near the tube, excessive force or volume during transfer, and retrograde migration of an embryo because of uterine contraction may also increase the risk of heterotopic pregnancy.8
A heterotopic pregnancy in spontaneous conceptual cycles without ART is relatively rare, even in a woman who has risk factors for ectopic pregnancy.12
How does heterotopic pregnancy develop?
Possibilities include the following, according to Wolf and colleagues:
- fertilization of two ova from a single coitus
- superimposition of an intrauterine pregnancy over an existing ectopic pregnancy (also known as superfetation).13
The appearance of cardiac activity may be discordant in heterotopic pregnancy, according to Hirsh and associates, suggesting that superfetation is indeed a mechanism in its development, with one pregnancy conceived earlier than the other.14
Diagnosis requires a high index of suspicion
The timely detection of heterotopic pregnancy necessitates vigilance.10,15 The TABLE lists signs and symptoms of this condition, which include abdominal pain, an adnexal mass, peritoneal irritation, an enlarged uterus, and absence of vaginal bleeding.6 In contrast to ectopic pregnancy, there is no vaginal bleeding with heterotopic pregnancy because an intact intrauterine pregnancy is present.8
TABLE
Signs and symptoms of heterotopic pregnancy
| Pain after spontaneous or induced abortion |
| Two corpora lutea detected during ultrasonography or laparotomy |
| Persistent Hegar’s sign or Chadwick’s sign after laparotomy for ectopic pregnancy |
| Absence of vaginal bleeding after laparotomy for ectopic pregnancy |
| Lateral location of a gestational sac identified via ultrasonography |
| Fluid in the uterus |
| Discordant appearance of fetal cardiac activity |
| Unpredictable quantitative human chorionic gonadotropin levels |
CASE 2: Ovarian “cyst” turns out to be a gestation
A 28-year-old woman, para 2-0-0-2, visits the emergency department complaining of acute abdominal pain, and is given two diagnoses: urinary tract infection and incomplete abortion at 5 weeks’ gestation. She is treated for the infection and discharged, to be followed up with treatment by her private ObGyn for the incomplete abortion.
Three days later she returns, reporting cramping and increased pain. Ultrasonography reveals intrauterine fetal demise at 8 weeks and 6 days of gestation, along with a hemorrhagic mass in the cul-de-sac—most likely a ruptured hemorrhagic ovarian cyst. Her history includes two cesarean deliveries and treatment with clonidine for hypertension. Her blood pressure is normal, and her abdomen is diffusely tender, with bowel sounds present. The preoperative diagnosis: incomplete abortion and a ruptured hemorrhagic cyst.
During exploratory laparotomy, left salpingo-oophorectomy is performed, and a hemorrhagic ovarian cyst is removed, with evacuation of hemoperitoneum, followed by suction curettage. Almost no tissue is present in the uterine cavity at the time of surgery. The final pathology report determines that the hemorrhagic cyst contained organizing clotted blood with trophoblasts, consistent with ectopic pregnancy. In addition, the uterine contents included scant tissue with hypersecretory endometrium. The physician theorizes that the collapsed gestational sac may have passed out of the patient’s uterus after US or during preoperative preparation.
The patient does well postoperatively and is discharged home.
Should ectopic pregnancy have been suspected earlier?
When a patient experiences pain after spontaneous or induced abortion, ectopic pregnancy should be suspected.2 In addition, women who exhibit signs or symptoms of ectopic pregnancy or continuing pregnancy after an inconclusive or negative US should be assessed thoroughly to exclude ectopic pregnancy.5
Conversely, if symptoms of pregnancy persist or worsen after laparotomy for ectopic pregnancy, the surgeon should suspect another pregnancy.7 Even when a patient who is being treated for infertility exhibits signs and symptoms of ectopic pregnancy, concurrent intrauterine pregnancy must be ruled out.9
A persistent Hegar’s sign or Chadwick’s sign means that a pregnancy is still present.7 In addition, the absence of vaginal bleeding after surgery for ectopic pregnancy may indicate the presence of gestation.6
Few heterotopic pregnancies are identified before surgery
Only 10% of heterotopic pregnancies are detected preoperatively.3 One third of intrauterine gestations in a heterotopic pregnancy spontaneously abort.11
Don’t assume that the presence of an intrauterine gestation excludes the possibility of ectopic pregnancy when the patient experiences abdominal pain.16
Imaging is helpful but not foolproof
Identifying a heterotopic pregnancy before surgery is an imaging challenge. Even when US is employed, the diagnosis is missed in 50% of cases—and even transvaginal US has low sensitivity.17,18 One reason may be the discordant appearance of fetal cardiac activity in coexisting intrauterine and ectopic pregnancies. Alternatively, the gestational sac may be anembryonic.19
A gestational sac is a sonolucent structure with a double decidual sac sign—i.e., an echogenic ring surrounding the sac. A pseudogestational sac containing fluid or blood can mimic a gestational sac.5 One helpful diagnostic sign of heterotopic pregnancy during US examination is a lateral location of one of the gestational sacs.16
If two corpora lutea are present on US—or even at laparotomy or laparoscopy—suspect heterotopic pregnancy.7
Obstetric imaging should include views of the adnexae
The adnexa and surrounding structures are usually not imaged during obstetric US because the focus is on the intrauterine gestation.5 In addition, ultrasonographers are frequently misled by the presence of fluid in the uterus.
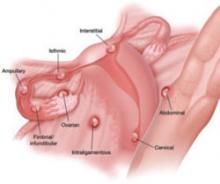
It is important for the ultrasonographer to examine the entire pelvic region for pregnancy (FIGURE), especially in women who have been treated with ART or who have pelvic inflammatory disease or a history of pelvic surgery.11 The adnexae should be assessed during every obstetric US, especially in women who are at risk of ectopic pregnancy.5
FIGURE When a pregnancy wanders, there are many possibilities for where
Ectopic pregnancy can arise as a result of internal migration of a fertilized ovum or transperitoneal migration of sperm. It may be observed at a number of possible sites within the pelvis.
Serial beta-hCG levels aren’t helpful
In heterotopic pregnancy, both gestations produce hCG, so the assessment of serial serum quantitative beta-hCG levels is not informative.11
When treating ectopic pregnancy, hold off on curettage
Ectopic pregnancy and extrauterine gestation are life-threatening emergencies that require timely diagnosis and treatment.5,19 The traditional treatment for ectopic pregnancy is laparotomy or laparoscopy with removal of the ectopic pregnancy, followed by dilation and curettage (D&C). The curettage removes the decidual cast and clots and is intended to prevent postoperative bleeding. However, curettage could destroy any intrauterine pregnancy that is not yet diagnosed. Therefore, D&C should be withheld until the uterus is confirmed to be empty.10
1. Childs AJ, Royek AB, Leigh TB, Gallup PG. Triplet heterotopic pregnancy after gonadotropin stimulation and intrauterine insemination diagnosed at laparoscopy: a case report. South Med J. 2005;98:833-835.
2. Richards SR, Stempel LE, Carlton BD. Heterotopic pregnancy: reappraisal of incidence. Am J Obstet Gynecol. 1982;142:928-930.
3. Laband SJ, Cherny WB, Finberg HJ. Heterotopic pregnancy: report of four cases. Am J Obstet Gynecol. 1988;158:437-438.
4. Snyder T, delCastillo J, Graff J, Hoxsey R, Hefti M. Heterotopic pregnancy after in vitro fertilization and ovulatory drugs. Ann Emerg Med. 1988;17:846-849.
5. DeFrancesch F, DiLeo L, Martinez J. Heterotopic pregnancy: discovery of ectopic pregnancy after elective abortion. South Med J. 1999;92:330-332.
6. Rizk B, Tan SL, Morcos S, et al. Heterotopic pregnancies after in vitro fertilization and embryo transfer. Am J Obstet Gynecol. 1991;164(1 Pt 1):161-164.
7. Rothman A, Shapiro J. Heterotopic pregnancy after homolateral salpingo-oophorectomy. Report of a case. Obstet Gynecol. 1965;26:718-720.
8. Sherer DM, Scibetta JJ, Sanko SR. Heterotopic quadruplet gestation with laparoscopic resection of ruptured interstitial pregnancy and subsequent successful outcome of triplets. Am J Obstet Gynecol. 1995;172(1 Pt 1):216-217.
9. Payne S, Duge J, Bradbury W. Ectopic pregnancy concomitant with twin intrauterine pregnancy. A case report. Obstet Gynecol. 1971;38:905-906.
10. Gamberdella FR, Marrs RP. Heterotopic pregnancy associated with assisted reproductive technology. Am J Obstet Gynecol. 1989;160:1520-1524.
11. Beyer DA, Dumesic DA. Heterotopic pregnancy: an emerging diagnostic challenge. OBG Management. 2002;14(10):36-46.
12. Somers MP, Spears M, Maynard AS, Syverud SA. Ruptured heterotopic pregnancy presenting with relative bradycardia in a woman not receiving reproductive assistance. Ann Emerg Med. 2004;43:382-385.
13. Wolf GC, Kritzer L, DeBold C. Heterotopic pregnancy: midtrimester management. Obstet Gynecol. 1979;54:756-758.
14. Hirsch E, Cohen L, Hecht BR. Heterotopic pregnancy with discordant ultrasonic appearance of fetal cardiac activity. Obstet Gynecol. 1992;79(5 Pt 2):824-825.
15. McLain PL, Kirkwood CR. Ovarian and intrauterine heterotopic pregnancy following clomiphene ovulation induction: report of a healthy live birth. J Fam Pract. 1987;24:76-79.
16. Luckas MJM, Fishwick K, Martin-Hierro K, Shaw GC, Walkinshaw SA. Survival of intrauterine twins and an interstitial singleton fetus from a heterotopic in vitro fertilisation–embryo transfer pregnancy. Br J Obstet Gynaecol. 1997;104:751-752.
17. Crabtree KE, Collet B, Kilpatrick SJ. Puerperal presentation of a living abdominal pregnancy. Obstet Gynecol. 1994;84(4 Pt 2):646-648.
18. Cheng PJ, Chueh HY, Qiu JT. Heterotopic pregnancy in a natural conception cycle presenting as hematometra. Obstet Gynecol. 2004;104(5 Pt 2):1195-1198.
19. Fa EM, Gerscovich EO. High resolution ultrasound in the diagnosis of heterotopic pregnancy: combined transabdominal and transvaginal approach. Br J Obstet Gynaecol. 1993;100:871-872.
20. Smith DJ, Siddique FH. A case of heterotopic pregnancy. Am J Obstet Gynecol. 1970;108:1289-1290.
The authors report no financial relationships relevant to this article.
CASE 1: Pelvic pain and few signs of intrauterine pregnancy
A 24-year-old woman, para 1-0-0-1, visits the hospital emergency department complaining of pelvic pain. She says the pain arose suddenly and reports that she had a positive urine pregnancy test earlier in the week. When asked about her obstetric history, she reports vaginal delivery of an 8 lb, 8 oz infant at 38 weeks’ gestation 2 years earlier. Her human chorionic gonadotropin (hCG) level is 3,000 mIU/mL, but ultrasonography (US) reveals no evidence of pregnancy. She is discharged with instructions to follow up with her physician in 2 days.
When her abdominal pain worsens, she returns to the emergency department. Physical examination reveals significant tenderness of the abdomen and moderate to severe tenderness of the cervix upon motion. Transvaginal US shows a uterus of normal size with a 5-mm endometrial lining and no gestational sac. The patient’s abdomen is full of fluid, with large, hypodense areas adjacent to the uterus bilaterally but larger on the right. The preoperative diagnosis: ruptured ectopic pregnancy.
During the diagnostic laparoscopy that follows, approximately 500 mL of blood is discovered in the abdomen and pelvis, and a gestational sac is found to be densely adherent to the right pelvic sidewall, where the ureter nears the uterine vessels. The sac, which has partially separated from the sidewall, is bleeding.
The surgeon peels the sac off the sidewall and controls bleeding with electrocautery and liquid thrombin. The final pathology report describes the tissue as an organizing blood clot with trophoblasts, consistent with ectopic pregnancy.
At a follow-up visit 3 weeks later, the patient reports persistent symptoms of pregnancy. Repeat US reveals a twin intrauterine pregnancy with two sacs, only one of which has a heartbeat. One week later, US shows confluence of the sacs, with a single viable fetus at 8 weeks and 2 days of gestation.
Could heterotopic pregnancy have been diagnosed earlier?
This case illustrates challenges inherent in the diagnosis of heterotopic pregnancy, which is much more common today than it was when it was first described 300 years ago. Incidence has increased from approximately 1 in 30,000 pregnancies to 1 in 2,600 pregnancies annually. When assisted reproductive technologies (ART) are used, the incidence may be as high as 1 in 100 pregnancies.1
The rising incidence suggests that the diagnosis of intrauterine pregnancy can no longer be used to exclude the presence of ectopic pregnancy, and vice versa. Instead, steps must be taken to rule out both when a woman exhibits pain and signs of pregnancy.
In this article, we discuss the causes, diagnosis, and treatment of heterotopic pregnancy, including the necessity of a high index of suspicion, the unreliability of US imaging in 50% of cases, and the need to avoid curettage in the treatment of ectopic pregnancy until an empty uterus can be confirmed.
Duverney was the first to report heterotopic pregnancy, in 1708, after finding an intrauterine pregnancy during the autopsy of a woman who had died from a ruptured ectopic pregnancy.1 It was 165 years, however, before the first review of the phenomenon was written.2 By 1970, only 479 such cases had been reported.20
Determining incidence remains a challenge—except that it is rising
In 1948, DeVoe and Pratt calculated the incidence of heterotopic pregnancy by multiplying the incidence of two-ovum pregnancy by the incidence of ectopic pregnancy, reaching an estimate of 1 in 30,000 pregnancies annually.2
In 1965, Rothman and Shapiro found that only about 500 cases of heterotopic pregnancy had been reported cumulatively.7 They reasoned that, if the incidence of fraternal twins is 1 in 110 and the incidence of ectopic pregnancy is 1 in 250, heterotopic pregnancy should occur at a rate of 1 in 27,500 gestations.7 They also speculated that many heterotopic pregnancies go undiagnosed because of early pregnancy loss.7
In 1971, Payne and colleagues hypothesized that ovulation-induction agents increase the incidence of heterotopic pregnancy, and McLain and associates reached a similar conclusion in 1987.9 The incidence of multiple pregnancy after oral ovulation induction is 8% to 10%, and it is 20% to 50% with injectable agents.9 In 1994, Crabtree and associates reported that both abdominal and heterotopic pregnancies appear to be increasing in incidence.17
Today, the incidence of heterotopic pregnancy is thought to be about 1 in 2,600 pregnancies annually—primarily because of assisted reproduction.2 The calculated risk of heterotopic pregnancy is 1 in 119, and it rises to 1 in 45 with embryo transfer.8
Why is heterotopic pregnancy on the increase?
One reason may be the increase in ectopic pregnancy. Among the factors contributing to the rising incidence of ectopic pregnancy are:
- pelvic adhesive disease
- effects of diethylstilbestrol (DES) on the genital tract
- antibiotic-induced tubal disease
- use of an intrauterine device (IUD)
- voluntary restriction of family size
- iatrogenic curettage of intrauterine pregnancy during surgery for ectopic pregnancy
- pregnancy termination
- history of surgery to treat infertility, ectopic pregnancy, or tubal adhesions
- improvement in the assay used to measure gonadotropin
- improvement in ultrasonography.2-4
A previous ectopic pregnancy is a risk factor for ectopic pregnancy as well as for heterotopic pregnancy.5
Pelvic infection, antibiotic-induced tubal disease, previous ectopic pregnancy, pelvic adhesions, and tubal surgery with cauterization of the tubes and subsequent bowel adhesions distort the fallopian tubes and may render them unable to propel a migrated embryo into the uterine cavity.2,6 Ectopic pregnancy may result from internal migration of a fertilized ovum or transperitoneal migration of sperm.7
DES exposure can distort the uterine cavity.8 Congenital and acquired uterine malformations increase the risk of ectopic pregnancy.3
In addition, ovulation-inducing drugs and ovarian stimulation increase the number of eggs available for conception, with a greater risk of multiple gestation and heterotopic pregnancy.3,9,10
The greatest increase in heterotopic pregnancy has been seen with ART involving the transfer of multiple embryos into the uterus, as well as gamete intrafallopian transfer, also known as GIFT.10,11 When five or more embryos are transferred, the risk of heterotopic pregnancy increases to 1 in 45 pregnancies annually.11 Inadvertent placement of the catheter tip near the tube, excessive force or volume during transfer, and retrograde migration of an embryo because of uterine contraction may also increase the risk of heterotopic pregnancy.8
A heterotopic pregnancy in spontaneous conceptual cycles without ART is relatively rare, even in a woman who has risk factors for ectopic pregnancy.12
How does heterotopic pregnancy develop?
Possibilities include the following, according to Wolf and colleagues:
- fertilization of two ova from a single coitus
- superimposition of an intrauterine pregnancy over an existing ectopic pregnancy (also known as superfetation).13
The appearance of cardiac activity may be discordant in heterotopic pregnancy, according to Hirsh and associates, suggesting that superfetation is indeed a mechanism in its development, with one pregnancy conceived earlier than the other.14
Diagnosis requires a high index of suspicion
The timely detection of heterotopic pregnancy necessitates vigilance.10,15 The TABLE lists signs and symptoms of this condition, which include abdominal pain, an adnexal mass, peritoneal irritation, an enlarged uterus, and absence of vaginal bleeding.6 In contrast to ectopic pregnancy, there is no vaginal bleeding with heterotopic pregnancy because an intact intrauterine pregnancy is present.8
TABLE
Signs and symptoms of heterotopic pregnancy
| Pain after spontaneous or induced abortion |
| Two corpora lutea detected during ultrasonography or laparotomy |
| Persistent Hegar’s sign or Chadwick’s sign after laparotomy for ectopic pregnancy |
| Absence of vaginal bleeding after laparotomy for ectopic pregnancy |
| Lateral location of a gestational sac identified via ultrasonography |
| Fluid in the uterus |
| Discordant appearance of fetal cardiac activity |
| Unpredictable quantitative human chorionic gonadotropin levels |
CASE 2: Ovarian “cyst” turns out to be a gestation
A 28-year-old woman, para 2-0-0-2, visits the emergency department complaining of acute abdominal pain, and is given two diagnoses: urinary tract infection and incomplete abortion at 5 weeks’ gestation. She is treated for the infection and discharged, to be followed up with treatment by her private ObGyn for the incomplete abortion.
Three days later she returns, reporting cramping and increased pain. Ultrasonography reveals intrauterine fetal demise at 8 weeks and 6 days of gestation, along with a hemorrhagic mass in the cul-de-sac—most likely a ruptured hemorrhagic ovarian cyst. Her history includes two cesarean deliveries and treatment with clonidine for hypertension. Her blood pressure is normal, and her abdomen is diffusely tender, with bowel sounds present. The preoperative diagnosis: incomplete abortion and a ruptured hemorrhagic cyst.
During exploratory laparotomy, left salpingo-oophorectomy is performed, and a hemorrhagic ovarian cyst is removed, with evacuation of hemoperitoneum, followed by suction curettage. Almost no tissue is present in the uterine cavity at the time of surgery. The final pathology report determines that the hemorrhagic cyst contained organizing clotted blood with trophoblasts, consistent with ectopic pregnancy. In addition, the uterine contents included scant tissue with hypersecretory endometrium. The physician theorizes that the collapsed gestational sac may have passed out of the patient’s uterus after US or during preoperative preparation.
The patient does well postoperatively and is discharged home.
Should ectopic pregnancy have been suspected earlier?
When a patient experiences pain after spontaneous or induced abortion, ectopic pregnancy should be suspected.2 In addition, women who exhibit signs or symptoms of ectopic pregnancy or continuing pregnancy after an inconclusive or negative US should be assessed thoroughly to exclude ectopic pregnancy.5
Conversely, if symptoms of pregnancy persist or worsen after laparotomy for ectopic pregnancy, the surgeon should suspect another pregnancy.7 Even when a patient who is being treated for infertility exhibits signs and symptoms of ectopic pregnancy, concurrent intrauterine pregnancy must be ruled out.9
A persistent Hegar’s sign or Chadwick’s sign means that a pregnancy is still present.7 In addition, the absence of vaginal bleeding after surgery for ectopic pregnancy may indicate the presence of gestation.6
Few heterotopic pregnancies are identified before surgery
Only 10% of heterotopic pregnancies are detected preoperatively.3 One third of intrauterine gestations in a heterotopic pregnancy spontaneously abort.11
Don’t assume that the presence of an intrauterine gestation excludes the possibility of ectopic pregnancy when the patient experiences abdominal pain.16
Imaging is helpful but not foolproof
Identifying a heterotopic pregnancy before surgery is an imaging challenge. Even when US is employed, the diagnosis is missed in 50% of cases—and even transvaginal US has low sensitivity.17,18 One reason may be the discordant appearance of fetal cardiac activity in coexisting intrauterine and ectopic pregnancies. Alternatively, the gestational sac may be anembryonic.19
A gestational sac is a sonolucent structure with a double decidual sac sign—i.e., an echogenic ring surrounding the sac. A pseudogestational sac containing fluid or blood can mimic a gestational sac.5 One helpful diagnostic sign of heterotopic pregnancy during US examination is a lateral location of one of the gestational sacs.16
If two corpora lutea are present on US—or even at laparotomy or laparoscopy—suspect heterotopic pregnancy.7
Obstetric imaging should include views of the adnexae
The adnexa and surrounding structures are usually not imaged during obstetric US because the focus is on the intrauterine gestation.5 In addition, ultrasonographers are frequently misled by the presence of fluid in the uterus.
It is important for the ultrasonographer to examine the entire pelvic region for pregnancy (FIGURE), especially in women who have been treated with ART or who have pelvic inflammatory disease or a history of pelvic surgery.11 The adnexae should be assessed during every obstetric US, especially in women who are at risk of ectopic pregnancy.5
FIGURE When a pregnancy wanders, there are many possibilities for where
Ectopic pregnancy can arise as a result of internal migration of a fertilized ovum or transperitoneal migration of sperm. It may be observed at a number of possible sites within the pelvis.
Serial beta-hCG levels aren’t helpful
In heterotopic pregnancy, both gestations produce hCG, so the assessment of serial serum quantitative beta-hCG levels is not informative.11
When treating ectopic pregnancy, hold off on curettage
Ectopic pregnancy and extrauterine gestation are life-threatening emergencies that require timely diagnosis and treatment.5,19 The traditional treatment for ectopic pregnancy is laparotomy or laparoscopy with removal of the ectopic pregnancy, followed by dilation and curettage (D&C). The curettage removes the decidual cast and clots and is intended to prevent postoperative bleeding. However, curettage could destroy any intrauterine pregnancy that is not yet diagnosed. Therefore, D&C should be withheld until the uterus is confirmed to be empty.10
The authors report no financial relationships relevant to this article.
CASE 1: Pelvic pain and few signs of intrauterine pregnancy
A 24-year-old woman, para 1-0-0-1, visits the hospital emergency department complaining of pelvic pain. She says the pain arose suddenly and reports that she had a positive urine pregnancy test earlier in the week. When asked about her obstetric history, she reports vaginal delivery of an 8 lb, 8 oz infant at 38 weeks’ gestation 2 years earlier. Her human chorionic gonadotropin (hCG) level is 3,000 mIU/mL, but ultrasonography (US) reveals no evidence of pregnancy. She is discharged with instructions to follow up with her physician in 2 days.
When her abdominal pain worsens, she returns to the emergency department. Physical examination reveals significant tenderness of the abdomen and moderate to severe tenderness of the cervix upon motion. Transvaginal US shows a uterus of normal size with a 5-mm endometrial lining and no gestational sac. The patient’s abdomen is full of fluid, with large, hypodense areas adjacent to the uterus bilaterally but larger on the right. The preoperative diagnosis: ruptured ectopic pregnancy.
During the diagnostic laparoscopy that follows, approximately 500 mL of blood is discovered in the abdomen and pelvis, and a gestational sac is found to be densely adherent to the right pelvic sidewall, where the ureter nears the uterine vessels. The sac, which has partially separated from the sidewall, is bleeding.
The surgeon peels the sac off the sidewall and controls bleeding with electrocautery and liquid thrombin. The final pathology report describes the tissue as an organizing blood clot with trophoblasts, consistent with ectopic pregnancy.
At a follow-up visit 3 weeks later, the patient reports persistent symptoms of pregnancy. Repeat US reveals a twin intrauterine pregnancy with two sacs, only one of which has a heartbeat. One week later, US shows confluence of the sacs, with a single viable fetus at 8 weeks and 2 days of gestation.
Could heterotopic pregnancy have been diagnosed earlier?
This case illustrates challenges inherent in the diagnosis of heterotopic pregnancy, which is much more common today than it was when it was first described 300 years ago. Incidence has increased from approximately 1 in 30,000 pregnancies to 1 in 2,600 pregnancies annually. When assisted reproductive technologies (ART) are used, the incidence may be as high as 1 in 100 pregnancies.1
The rising incidence suggests that the diagnosis of intrauterine pregnancy can no longer be used to exclude the presence of ectopic pregnancy, and vice versa. Instead, steps must be taken to rule out both when a woman exhibits pain and signs of pregnancy.
In this article, we discuss the causes, diagnosis, and treatment of heterotopic pregnancy, including the necessity of a high index of suspicion, the unreliability of US imaging in 50% of cases, and the need to avoid curettage in the treatment of ectopic pregnancy until an empty uterus can be confirmed.
Duverney was the first to report heterotopic pregnancy, in 1708, after finding an intrauterine pregnancy during the autopsy of a woman who had died from a ruptured ectopic pregnancy.1 It was 165 years, however, before the first review of the phenomenon was written.2 By 1970, only 479 such cases had been reported.20
Determining incidence remains a challenge—except that it is rising
In 1948, DeVoe and Pratt calculated the incidence of heterotopic pregnancy by multiplying the incidence of two-ovum pregnancy by the incidence of ectopic pregnancy, reaching an estimate of 1 in 30,000 pregnancies annually.2
In 1965, Rothman and Shapiro found that only about 500 cases of heterotopic pregnancy had been reported cumulatively.7 They reasoned that, if the incidence of fraternal twins is 1 in 110 and the incidence of ectopic pregnancy is 1 in 250, heterotopic pregnancy should occur at a rate of 1 in 27,500 gestations.7 They also speculated that many heterotopic pregnancies go undiagnosed because of early pregnancy loss.7
In 1971, Payne and colleagues hypothesized that ovulation-induction agents increase the incidence of heterotopic pregnancy, and McLain and associates reached a similar conclusion in 1987.9 The incidence of multiple pregnancy after oral ovulation induction is 8% to 10%, and it is 20% to 50% with injectable agents.9 In 1994, Crabtree and associates reported that both abdominal and heterotopic pregnancies appear to be increasing in incidence.17
Today, the incidence of heterotopic pregnancy is thought to be about 1 in 2,600 pregnancies annually—primarily because of assisted reproduction.2 The calculated risk of heterotopic pregnancy is 1 in 119, and it rises to 1 in 45 with embryo transfer.8
Why is heterotopic pregnancy on the increase?
One reason may be the increase in ectopic pregnancy. Among the factors contributing to the rising incidence of ectopic pregnancy are:
- pelvic adhesive disease
- effects of diethylstilbestrol (DES) on the genital tract
- antibiotic-induced tubal disease
- use of an intrauterine device (IUD)
- voluntary restriction of family size
- iatrogenic curettage of intrauterine pregnancy during surgery for ectopic pregnancy
- pregnancy termination
- history of surgery to treat infertility, ectopic pregnancy, or tubal adhesions
- improvement in the assay used to measure gonadotropin
- improvement in ultrasonography.2-4
A previous ectopic pregnancy is a risk factor for ectopic pregnancy as well as for heterotopic pregnancy.5
Pelvic infection, antibiotic-induced tubal disease, previous ectopic pregnancy, pelvic adhesions, and tubal surgery with cauterization of the tubes and subsequent bowel adhesions distort the fallopian tubes and may render them unable to propel a migrated embryo into the uterine cavity.2,6 Ectopic pregnancy may result from internal migration of a fertilized ovum or transperitoneal migration of sperm.7
DES exposure can distort the uterine cavity.8 Congenital and acquired uterine malformations increase the risk of ectopic pregnancy.3
In addition, ovulation-inducing drugs and ovarian stimulation increase the number of eggs available for conception, with a greater risk of multiple gestation and heterotopic pregnancy.3,9,10
The greatest increase in heterotopic pregnancy has been seen with ART involving the transfer of multiple embryos into the uterus, as well as gamete intrafallopian transfer, also known as GIFT.10,11 When five or more embryos are transferred, the risk of heterotopic pregnancy increases to 1 in 45 pregnancies annually.11 Inadvertent placement of the catheter tip near the tube, excessive force or volume during transfer, and retrograde migration of an embryo because of uterine contraction may also increase the risk of heterotopic pregnancy.8
A heterotopic pregnancy in spontaneous conceptual cycles without ART is relatively rare, even in a woman who has risk factors for ectopic pregnancy.12
How does heterotopic pregnancy develop?
Possibilities include the following, according to Wolf and colleagues:
- fertilization of two ova from a single coitus
- superimposition of an intrauterine pregnancy over an existing ectopic pregnancy (also known as superfetation).13
The appearance of cardiac activity may be discordant in heterotopic pregnancy, according to Hirsh and associates, suggesting that superfetation is indeed a mechanism in its development, with one pregnancy conceived earlier than the other.14
Diagnosis requires a high index of suspicion
The timely detection of heterotopic pregnancy necessitates vigilance.10,15 The TABLE lists signs and symptoms of this condition, which include abdominal pain, an adnexal mass, peritoneal irritation, an enlarged uterus, and absence of vaginal bleeding.6 In contrast to ectopic pregnancy, there is no vaginal bleeding with heterotopic pregnancy because an intact intrauterine pregnancy is present.8
TABLE
Signs and symptoms of heterotopic pregnancy
| Pain after spontaneous or induced abortion |
| Two corpora lutea detected during ultrasonography or laparotomy |
| Persistent Hegar’s sign or Chadwick’s sign after laparotomy for ectopic pregnancy |
| Absence of vaginal bleeding after laparotomy for ectopic pregnancy |
| Lateral location of a gestational sac identified via ultrasonography |
| Fluid in the uterus |
| Discordant appearance of fetal cardiac activity |
| Unpredictable quantitative human chorionic gonadotropin levels |
CASE 2: Ovarian “cyst” turns out to be a gestation
A 28-year-old woman, para 2-0-0-2, visits the emergency department complaining of acute abdominal pain, and is given two diagnoses: urinary tract infection and incomplete abortion at 5 weeks’ gestation. She is treated for the infection and discharged, to be followed up with treatment by her private ObGyn for the incomplete abortion.
Three days later she returns, reporting cramping and increased pain. Ultrasonography reveals intrauterine fetal demise at 8 weeks and 6 days of gestation, along with a hemorrhagic mass in the cul-de-sac—most likely a ruptured hemorrhagic ovarian cyst. Her history includes two cesarean deliveries and treatment with clonidine for hypertension. Her blood pressure is normal, and her abdomen is diffusely tender, with bowel sounds present. The preoperative diagnosis: incomplete abortion and a ruptured hemorrhagic cyst.
During exploratory laparotomy, left salpingo-oophorectomy is performed, and a hemorrhagic ovarian cyst is removed, with evacuation of hemoperitoneum, followed by suction curettage. Almost no tissue is present in the uterine cavity at the time of surgery. The final pathology report determines that the hemorrhagic cyst contained organizing clotted blood with trophoblasts, consistent with ectopic pregnancy. In addition, the uterine contents included scant tissue with hypersecretory endometrium. The physician theorizes that the collapsed gestational sac may have passed out of the patient’s uterus after US or during preoperative preparation.
The patient does well postoperatively and is discharged home.
Should ectopic pregnancy have been suspected earlier?
When a patient experiences pain after spontaneous or induced abortion, ectopic pregnancy should be suspected.2 In addition, women who exhibit signs or symptoms of ectopic pregnancy or continuing pregnancy after an inconclusive or negative US should be assessed thoroughly to exclude ectopic pregnancy.5
Conversely, if symptoms of pregnancy persist or worsen after laparotomy for ectopic pregnancy, the surgeon should suspect another pregnancy.7 Even when a patient who is being treated for infertility exhibits signs and symptoms of ectopic pregnancy, concurrent intrauterine pregnancy must be ruled out.9
A persistent Hegar’s sign or Chadwick’s sign means that a pregnancy is still present.7 In addition, the absence of vaginal bleeding after surgery for ectopic pregnancy may indicate the presence of gestation.6
Few heterotopic pregnancies are identified before surgery
Only 10% of heterotopic pregnancies are detected preoperatively.3 One third of intrauterine gestations in a heterotopic pregnancy spontaneously abort.11
Don’t assume that the presence of an intrauterine gestation excludes the possibility of ectopic pregnancy when the patient experiences abdominal pain.16
Imaging is helpful but not foolproof
Identifying a heterotopic pregnancy before surgery is an imaging challenge. Even when US is employed, the diagnosis is missed in 50% of cases—and even transvaginal US has low sensitivity.17,18 One reason may be the discordant appearance of fetal cardiac activity in coexisting intrauterine and ectopic pregnancies. Alternatively, the gestational sac may be anembryonic.19
A gestational sac is a sonolucent structure with a double decidual sac sign—i.e., an echogenic ring surrounding the sac. A pseudogestational sac containing fluid or blood can mimic a gestational sac.5 One helpful diagnostic sign of heterotopic pregnancy during US examination is a lateral location of one of the gestational sacs.16
If two corpora lutea are present on US—or even at laparotomy or laparoscopy—suspect heterotopic pregnancy.7
Obstetric imaging should include views of the adnexae
The adnexa and surrounding structures are usually not imaged during obstetric US because the focus is on the intrauterine gestation.5 In addition, ultrasonographers are frequently misled by the presence of fluid in the uterus.
It is important for the ultrasonographer to examine the entire pelvic region for pregnancy (FIGURE), especially in women who have been treated with ART or who have pelvic inflammatory disease or a history of pelvic surgery.11 The adnexae should be assessed during every obstetric US, especially in women who are at risk of ectopic pregnancy.5
FIGURE When a pregnancy wanders, there are many possibilities for where
Ectopic pregnancy can arise as a result of internal migration of a fertilized ovum or transperitoneal migration of sperm. It may be observed at a number of possible sites within the pelvis.
Serial beta-hCG levels aren’t helpful
In heterotopic pregnancy, both gestations produce hCG, so the assessment of serial serum quantitative beta-hCG levels is not informative.11
When treating ectopic pregnancy, hold off on curettage
Ectopic pregnancy and extrauterine gestation are life-threatening emergencies that require timely diagnosis and treatment.5,19 The traditional treatment for ectopic pregnancy is laparotomy or laparoscopy with removal of the ectopic pregnancy, followed by dilation and curettage (D&C). The curettage removes the decidual cast and clots and is intended to prevent postoperative bleeding. However, curettage could destroy any intrauterine pregnancy that is not yet diagnosed. Therefore, D&C should be withheld until the uterus is confirmed to be empty.10
1. Childs AJ, Royek AB, Leigh TB, Gallup PG. Triplet heterotopic pregnancy after gonadotropin stimulation and intrauterine insemination diagnosed at laparoscopy: a case report. South Med J. 2005;98:833-835.
2. Richards SR, Stempel LE, Carlton BD. Heterotopic pregnancy: reappraisal of incidence. Am J Obstet Gynecol. 1982;142:928-930.
3. Laband SJ, Cherny WB, Finberg HJ. Heterotopic pregnancy: report of four cases. Am J Obstet Gynecol. 1988;158:437-438.
4. Snyder T, delCastillo J, Graff J, Hoxsey R, Hefti M. Heterotopic pregnancy after in vitro fertilization and ovulatory drugs. Ann Emerg Med. 1988;17:846-849.
5. DeFrancesch F, DiLeo L, Martinez J. Heterotopic pregnancy: discovery of ectopic pregnancy after elective abortion. South Med J. 1999;92:330-332.
6. Rizk B, Tan SL, Morcos S, et al. Heterotopic pregnancies after in vitro fertilization and embryo transfer. Am J Obstet Gynecol. 1991;164(1 Pt 1):161-164.
7. Rothman A, Shapiro J. Heterotopic pregnancy after homolateral salpingo-oophorectomy. Report of a case. Obstet Gynecol. 1965;26:718-720.
8. Sherer DM, Scibetta JJ, Sanko SR. Heterotopic quadruplet gestation with laparoscopic resection of ruptured interstitial pregnancy and subsequent successful outcome of triplets. Am J Obstet Gynecol. 1995;172(1 Pt 1):216-217.
9. Payne S, Duge J, Bradbury W. Ectopic pregnancy concomitant with twin intrauterine pregnancy. A case report. Obstet Gynecol. 1971;38:905-906.
10. Gamberdella FR, Marrs RP. Heterotopic pregnancy associated with assisted reproductive technology. Am J Obstet Gynecol. 1989;160:1520-1524.
11. Beyer DA, Dumesic DA. Heterotopic pregnancy: an emerging diagnostic challenge. OBG Management. 2002;14(10):36-46.
12. Somers MP, Spears M, Maynard AS, Syverud SA. Ruptured heterotopic pregnancy presenting with relative bradycardia in a woman not receiving reproductive assistance. Ann Emerg Med. 2004;43:382-385.
13. Wolf GC, Kritzer L, DeBold C. Heterotopic pregnancy: midtrimester management. Obstet Gynecol. 1979;54:756-758.
14. Hirsch E, Cohen L, Hecht BR. Heterotopic pregnancy with discordant ultrasonic appearance of fetal cardiac activity. Obstet Gynecol. 1992;79(5 Pt 2):824-825.
15. McLain PL, Kirkwood CR. Ovarian and intrauterine heterotopic pregnancy following clomiphene ovulation induction: report of a healthy live birth. J Fam Pract. 1987;24:76-79.
16. Luckas MJM, Fishwick K, Martin-Hierro K, Shaw GC, Walkinshaw SA. Survival of intrauterine twins and an interstitial singleton fetus from a heterotopic in vitro fertilisation–embryo transfer pregnancy. Br J Obstet Gynaecol. 1997;104:751-752.
17. Crabtree KE, Collet B, Kilpatrick SJ. Puerperal presentation of a living abdominal pregnancy. Obstet Gynecol. 1994;84(4 Pt 2):646-648.
18. Cheng PJ, Chueh HY, Qiu JT. Heterotopic pregnancy in a natural conception cycle presenting as hematometra. Obstet Gynecol. 2004;104(5 Pt 2):1195-1198.
19. Fa EM, Gerscovich EO. High resolution ultrasound in the diagnosis of heterotopic pregnancy: combined transabdominal and transvaginal approach. Br J Obstet Gynaecol. 1993;100:871-872.
20. Smith DJ, Siddique FH. A case of heterotopic pregnancy. Am J Obstet Gynecol. 1970;108:1289-1290.
1. Childs AJ, Royek AB, Leigh TB, Gallup PG. Triplet heterotopic pregnancy after gonadotropin stimulation and intrauterine insemination diagnosed at laparoscopy: a case report. South Med J. 2005;98:833-835.
2. Richards SR, Stempel LE, Carlton BD. Heterotopic pregnancy: reappraisal of incidence. Am J Obstet Gynecol. 1982;142:928-930.
3. Laband SJ, Cherny WB, Finberg HJ. Heterotopic pregnancy: report of four cases. Am J Obstet Gynecol. 1988;158:437-438.
4. Snyder T, delCastillo J, Graff J, Hoxsey R, Hefti M. Heterotopic pregnancy after in vitro fertilization and ovulatory drugs. Ann Emerg Med. 1988;17:846-849.
5. DeFrancesch F, DiLeo L, Martinez J. Heterotopic pregnancy: discovery of ectopic pregnancy after elective abortion. South Med J. 1999;92:330-332.
6. Rizk B, Tan SL, Morcos S, et al. Heterotopic pregnancies after in vitro fertilization and embryo transfer. Am J Obstet Gynecol. 1991;164(1 Pt 1):161-164.
7. Rothman A, Shapiro J. Heterotopic pregnancy after homolateral salpingo-oophorectomy. Report of a case. Obstet Gynecol. 1965;26:718-720.
8. Sherer DM, Scibetta JJ, Sanko SR. Heterotopic quadruplet gestation with laparoscopic resection of ruptured interstitial pregnancy and subsequent successful outcome of triplets. Am J Obstet Gynecol. 1995;172(1 Pt 1):216-217.
9. Payne S, Duge J, Bradbury W. Ectopic pregnancy concomitant with twin intrauterine pregnancy. A case report. Obstet Gynecol. 1971;38:905-906.
10. Gamberdella FR, Marrs RP. Heterotopic pregnancy associated with assisted reproductive technology. Am J Obstet Gynecol. 1989;160:1520-1524.
11. Beyer DA, Dumesic DA. Heterotopic pregnancy: an emerging diagnostic challenge. OBG Management. 2002;14(10):36-46.
12. Somers MP, Spears M, Maynard AS, Syverud SA. Ruptured heterotopic pregnancy presenting with relative bradycardia in a woman not receiving reproductive assistance. Ann Emerg Med. 2004;43:382-385.
13. Wolf GC, Kritzer L, DeBold C. Heterotopic pregnancy: midtrimester management. Obstet Gynecol. 1979;54:756-758.
14. Hirsch E, Cohen L, Hecht BR. Heterotopic pregnancy with discordant ultrasonic appearance of fetal cardiac activity. Obstet Gynecol. 1992;79(5 Pt 2):824-825.
15. McLain PL, Kirkwood CR. Ovarian and intrauterine heterotopic pregnancy following clomiphene ovulation induction: report of a healthy live birth. J Fam Pract. 1987;24:76-79.
16. Luckas MJM, Fishwick K, Martin-Hierro K, Shaw GC, Walkinshaw SA. Survival of intrauterine twins and an interstitial singleton fetus from a heterotopic in vitro fertilisation–embryo transfer pregnancy. Br J Obstet Gynaecol. 1997;104:751-752.
17. Crabtree KE, Collet B, Kilpatrick SJ. Puerperal presentation of a living abdominal pregnancy. Obstet Gynecol. 1994;84(4 Pt 2):646-648.
18. Cheng PJ, Chueh HY, Qiu JT. Heterotopic pregnancy in a natural conception cycle presenting as hematometra. Obstet Gynecol. 2004;104(5 Pt 2):1195-1198.
19. Fa EM, Gerscovich EO. High resolution ultrasound in the diagnosis of heterotopic pregnancy: combined transabdominal and transvaginal approach. Br J Obstet Gynaecol. 1993;100:871-872.
20. Smith DJ, Siddique FH. A case of heterotopic pregnancy. Am J Obstet Gynecol. 1970;108:1289-1290.
PELVIC FLOOR DYSFUNCTION
The authors report no financial relationships relevant to this article.
Overactive bladder (OAB)—urinary urgency, with or without incontinence, usually with frequency and nocturia1—is a common problem among women who seek care from an ObGyn. In fact, the condition is estimated to carry a health-care cost in excess of $12 billion annually in the United States.2
A recent community-based survey in Norway estimated the prevalence of urinary incontinence there to be 27% in women between the ages of 65 and 69 years and 35% to 40% in those 80 years or older.3 A population-based study in the United States suggested an even higher rate of urinary incontinence here: greater than 50% in women 60 years or older, with 1) urge urinary incontinence (UUI) predominating4 and 2) the prevalence particularly high among older women who are homebound or who live in a long-term care facility.5
OAB can undermine quality of life in several ways: social isolation, anxiety, poor sleep, higher risk of fracture after a fall,6 reduced ability to function, and poor self-perception. Despite these harmful effects, many women delay seeking care for OAB because they are embarrassed to talk about it with their physician.
Treatment by generalists is feasible—but there is a catch
It’s possible to treat most patients with OAB without referral to a specialist. Two common concerns, however, may set up a roadblock to successful management: the adverse effects associated with some agents and suboptimal control of symptoms.
In this Update, we review recent findings about 1) the potential that anticholinergic therapy has for impairing cognitive function in the older population of women and 2) the important role that concomitant behavioral therapy plays in the long-term success of, and patients’ satisfaction with, treatment of OAB.
Behavioral therapy for OAB: Is it worth all the effort?
Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc. 2000;48:370–374.
The authors of this article followed a randomized clinical trial of older women that compared behavioral and drug therapy for OAB. In the trial, biofeedback-assisted behavioral training (comprising anorectal biofeedback, urge strategies, pelvic muscle biofeedback, and practitioner-directed review with optimization) was compared with treatment with oxybutynin, between 2.5 and 15 mg/day. Both biofeedback-assisted behavioral therapy and the drug regimen were found effective, although neither treatment provided an entirely satisfactory result for all patients. (For a brief description of what constitutes behavioral treatment, see “6 tenets of behavioral therapy for urge urinary incontinence.”)
Second phase of the trial. To determine if treatment satisfaction could be enhanced, the investigators performed a modified crossover study to determine whether combination therapy—biofeedback-assisted behavioral training plus oxybutynin—added any benefit over treatment with behavioral therapy or drug therapy alone. Eligibility was determined by age (55 years or older), demonstrated UUI for at least 3 months, and incomplete dryness or incomplete satisfaction with the outcome of 8 weeks of single-intervention treatment (with either treatment) during the initial phase of the trial.
This subgroup was offered an additional 8 weeks of combination therapy. The primary outcome measure was a reduction in the frequency of episodes of incontinence episodes as recorded by subjects in a bladder diary.
Of 197 women who participated in the original randomized clinical trial, 35—27 who completed drug therapy and 8 who completed behavioral treatment—elected to receive combination therapy. Those 35 subjects did not differ in any of the multiple baseline variables; mean age was 69.3 years (standard deviation [SD], ±7.9 years).
Among subjects originally assigned to behavioral therapy alone, overall reduction in incontinence increased from a mean of 57.5% to a mean of 88.5% after combined therapy (P=.034). Subjects originally assigned to drug therapy alone demonstrated an improvement from 72.7% reduction in incontinence to a mean 84.3% overall reduction with combined therapy (P=.001).
These data suggest that combined therapy can be more effective than behavioral therapy or drug therapy alone. The impact of this study is limited, however, by the relatively low percentage (12.7%) of patients who had received behavioral therapy and chose to add drug therapy, compared with the 41.5% who moved from drug therapy alone to add behavioral therapy.
Furthermore, subjects were self-selected: They chose to continue with an additional 8 weeks of therapy after their initial suboptimal outcome. It is possible that some subjects who were neither totally continent nor completely satisfied with initial therapy chose not to continue with the crossover segment of the trial because it posed too great a burden or because they were discouraged with the initial degree of improvement.
Generalizing these results to all older women with UUI is difficult. The authors point out, however, that, in practice, patients may be more likely than not to choose combination therapy in the hope of shortening the duration of medical therapy. Although it isn’t known whether providing combination therapy from the outset would have yielded better outcomes than either single therapy did, the authors hypothesize that initial combination therapy may result in greater improvement because patients have a high level of motivation and expectation of improvement at the beginning of treatment.
Importance of this article. The investigators demonstrated that a combination of behavioral and drug therapies can provide increased effectiveness in patients for whom each treatment alone led to suboptimal satisfaction. Furthermore, by targeting women older than 55 years, the investigators were able to demonstrate this effectiveness in a group for whom pelvic-floor training may be more difficult than it is for younger women.
It will be interesting to see if future research will 1) validate these findings and 2) determine whether combined therapy can reduce the duration of drug therapy in this older population through behavioral modification and pelvic floor reeducation.
Fluid management
This first-step therapy can involve providing a handout to the patient that details techniques she can use to monitor and control her fluid intake in a manner that addresses her problem. Among such steps:
- avoiding caffeine and artificial sweeteners
- tracking her diet to identify any other bladder irritants
- limiting fluids before times she is more likely to be incontinent—during a long drive, for example, or, in the case of nocturia, after the evening meal.
Scheduled voiding
With scheduled, or prompted, voiding, the patient empties her bladder at a set interval—usually, every 1.5 to 2 hours. If nocturia, or the more severe enuresis, is a problem, the patient can be prompted by an alarm clock or (if she is institutionalized) by nursing staff. Combining scheduled voiding with fluid management principles helps the patient avoid reaching a bladder volume at which an episode of incontinence becomes more likely.
Bladder training
This is a modification of scheduled voiding that attempts to establish a normal voiding interval in patients who have significant frequency but a small voided volume. It imposes a regimented voiding schedule that gradually (over 7 to 10 days) extends the duration between voids.
Pelvic floor-muscle exercises
The focus here is on using pelvic-floor muscles to prevent incontinence. The muscles are strengthened by having the patient perform Kegel exercises (named for Arnold H. Kegel, MD, who, in 1948, recognized the role of pelvic floor-muscle rehabilitation in the treatment of incontinence). The exercises involve simultaneous 1) contraction of the pelvic and periurethral musculature and 2) relaxation of other muscles, including abdominal muscles, which can increase pressure on the bladder.
Once the patient learns to perform Kegel exercises, she can use them to suppress urgency: Instead of hurrying to the bathroom when urgency arises, she is encouraged to sit down, relax, and contract the pelvic-floor muscles repeatedly until the urge to void diminishes. Once it does, the patient proceeds to the toilet to void normally.
Pelvic exam
By self-exam, the patient can identify and familiarize herself with her purposeful contractions of the pelvic-floor musculature and thereby strengthen those muscles with effective exercise.
Biofeedback
Direct feedback about contractions of the pelvic-floor muscles—by a display of data on a gauge or computer monitor, gathered using an intravaginal or anorectal sensor or probe—allows a patient who is exercising those muscles to better target her efforts and maximize their effectiveness.
Combining behavioral therapy and an anticholinergic medication for urge urinary incontinence may yield a superior result after either modality alone has been disappointing by the patient’s account of success.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
Does oxybutynin for UUI further erode cognition in elderly women who are cognitively impaired?
Lackner TE, Wyman JF, McCarthy TC, Monigold M, Davey C. Randomized, placebo-controlled trial of the cognitive effect, safety, and tolerability of oral extended-release oxybutynin in cognitively impaired nursing home residents with urge urinary incontinence. J Am Geriatr Soc. 2008;56:862–870.
Although anticholinergic therapy is modestly effective against UUI in nursing home residents, past studies have suggested that such treatment can impair, or further impair, cognition in this population—a concern that may lead to underuse. This double-blinded, randomized, placebo-controlled trial compared short-term oral extended-release oxybutynin with placebo.
Consequently, the authors sought to determine the cognitive effect, safety, and tolerability of 5 mg/day oral extended-release oxybutynin (the most commonly prescribed dosage) in cognitively impaired older nursing home residents who have UUI.
Subjects were eligible if they:
- were 65 years or older
- had UUI
- lived in a nursing home longer than 3 months
- had cognitive impairment.
Women already being treated for urinary incontinence, those who had an indwelling Foley catheter or urinary retention, and those who were bed-bound or incommunicative were excluded.
Fifty women, mean age 88.6 years (SD, ±6.2), from 12 nursing home facilities, agreed to participate. They were further stratified based on the score of a Mini-Mental State Exam (MMSE): 13 had severe cognitive impairment (MMSE score, 5–10) and 37 had mild or moderate impairment (score, 11–23).
Subjects were randomized to 4 weeks’ treatment with either 5 mg/day oral extended-release oxybutynin or one placebo tablet daily. A nurse practitioner who was blinded to randomization collected all data. The Confusion Assessment Method (CAM) algorithm, MMSE, and Severe Impairment Battery (SIB) were used to assess cognitive decline. The Brief Agitation Rating Scale (BARS) assessed agitation.
No baseline differences were noted with regard to: age; demographic, functional, and neuropsychiatric characteristics; clinical factors predisposing to delirium; and serum anticholinergic activity. Adherence was similar in the treatment (97%) and placebo (97.4%) groups.
Finding: Cognitive impairment. Treatment and placebo groups in the baseline mild-or-moderate stratum (by MMSE) showed equivalent mean changes in CAM scores at all time points. Because of the small sample size, however, CAM score equivalence could not be definitively determined for the groups in the severe impairment stratum. Evaluation of mean MMSE and BARS scores showed no significant changes between groups.
Finding: Tolerability. Excellent tolerability was noted in the treatment group: 96% of subjects completed the trial (compared with 92% of the placebo group). No difference in the rate of adverse events was noted between treatment and placebo groups; of adverse events recorded, 90% were judged “mild” by the investigators. Constipation and dry mouth were most common.
Finding: Falls. More than half—54%—of subjects in both groups experienced at least one fall during the trial or during the preceding or following 3 months. Despite this, no difference in the rate of falls between the treatment and placebo groups was noted. Furthermore, regression analysis revealed no treatment or period effect on falls per month across the time of observation.
Conclusions. Treatment with 5 mg/day oral extended-release oxybutynin in older patients with some cognitive impairment is well tolerated, the study’s findings suggest, with minimal risk of further cognitive decline or delirium over the short term. The potential that long-term therapy has to harm cognitive function remains, however; data on long-term treatment are needed to illuminate that area.
The authors also address the importance of dosing, especially over time, and discuss the lower potential of newer-generation anticholinergics to produce cognitive impairment.
A limited number of articles in the medical literature address anticholinergics in an older population, specifically, and only a few of those evaluated the effects of the drugs on cognitive function. By investigating patients who had an existing cognitive impairment, the authors of this article were able to target a cohort at risk of further cognitive impairment from medication use—thereby giving further weight to their findings of no significant effect.
Main strengths and limitations of the study. The investigators used validated, standardized cognitive tests that were administered by a uniform blinded evaluator in a randomized, controlled trial. The study was limited, however, because patients were evaluated only over a relatively short period (1 month) and because the efficacy of therapy was not addressed.
Further studies of anticholinergic medications, using the same rigorous scientific approach that these investigators applied, are needed to address 1) the long-term efficacy of oxybutynin and similar agents and 2) the cognitive effects of long-term treatment in this older population.
Further impairment is unlikely over the short term when a cognitively impaired nursing home patient who has urge urinary incontinence is treated with 5 mg/day oral extended-release oxybutynin.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
1. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Am J Obstet Gynecol. 2002;187:116-126.
2. Hu TW, Wagner TH, Bentkover JD, et al. Estimated economic costs of overactive bladder in the United States. Urology. 2003;61:1123-1128.
3. Hannestad YS, Rortveit G, Sandvik H, Hunskaar S. A community-based epidemiological survey of female urinary incontinence: the Norwegian EPINCONT study. Epidemiology of Incontinence in the County of Nord-Trøndelag. J Clin Epidemiol. 2000;53:1150-1157.
4. Melville JL, Katon W, Delaney K, Newton K. Urinary incontinence in US women: a population-based study. Arch Intern Med. 2005;165:537-542.
5. Fantl JA, Newman DK, Colling J, et al. Managing Acute and Chronic Urinary Incontinence. Clinical Practice Guideline. Quick Reference Guide for Clinicians, No. 2, 1996 Update. Rockville, MD: US Department of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research. AHCPR Pub. No. 96-0686. January 1996. Available at: http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat6.chapter.32554. Accessed September 11, 2009.
6. Brown JS, Vittinghoff E, Wyman JF, et al. Urinary incontinence: does it increase risk for falls and fractures? Study of Osteoporotic Fractures Research Group. J Am Geriatr Soc. 2000;48:721-725.
The authors report no financial relationships relevant to this article.
Overactive bladder (OAB)—urinary urgency, with or without incontinence, usually with frequency and nocturia1—is a common problem among women who seek care from an ObGyn. In fact, the condition is estimated to carry a health-care cost in excess of $12 billion annually in the United States.2
A recent community-based survey in Norway estimated the prevalence of urinary incontinence there to be 27% in women between the ages of 65 and 69 years and 35% to 40% in those 80 years or older.3 A population-based study in the United States suggested an even higher rate of urinary incontinence here: greater than 50% in women 60 years or older, with 1) urge urinary incontinence (UUI) predominating4 and 2) the prevalence particularly high among older women who are homebound or who live in a long-term care facility.5
OAB can undermine quality of life in several ways: social isolation, anxiety, poor sleep, higher risk of fracture after a fall,6 reduced ability to function, and poor self-perception. Despite these harmful effects, many women delay seeking care for OAB because they are embarrassed to talk about it with their physician.
Treatment by generalists is feasible—but there is a catch
It’s possible to treat most patients with OAB without referral to a specialist. Two common concerns, however, may set up a roadblock to successful management: the adverse effects associated with some agents and suboptimal control of symptoms.
In this Update, we review recent findings about 1) the potential that anticholinergic therapy has for impairing cognitive function in the older population of women and 2) the important role that concomitant behavioral therapy plays in the long-term success of, and patients’ satisfaction with, treatment of OAB.
Behavioral therapy for OAB: Is it worth all the effort?
Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc. 2000;48:370–374.
The authors of this article followed a randomized clinical trial of older women that compared behavioral and drug therapy for OAB. In the trial, biofeedback-assisted behavioral training (comprising anorectal biofeedback, urge strategies, pelvic muscle biofeedback, and practitioner-directed review with optimization) was compared with treatment with oxybutynin, between 2.5 and 15 mg/day. Both biofeedback-assisted behavioral therapy and the drug regimen were found effective, although neither treatment provided an entirely satisfactory result for all patients. (For a brief description of what constitutes behavioral treatment, see “6 tenets of behavioral therapy for urge urinary incontinence.”)
Second phase of the trial. To determine if treatment satisfaction could be enhanced, the investigators performed a modified crossover study to determine whether combination therapy—biofeedback-assisted behavioral training plus oxybutynin—added any benefit over treatment with behavioral therapy or drug therapy alone. Eligibility was determined by age (55 years or older), demonstrated UUI for at least 3 months, and incomplete dryness or incomplete satisfaction with the outcome of 8 weeks of single-intervention treatment (with either treatment) during the initial phase of the trial.
This subgroup was offered an additional 8 weeks of combination therapy. The primary outcome measure was a reduction in the frequency of episodes of incontinence episodes as recorded by subjects in a bladder diary.
Of 197 women who participated in the original randomized clinical trial, 35—27 who completed drug therapy and 8 who completed behavioral treatment—elected to receive combination therapy. Those 35 subjects did not differ in any of the multiple baseline variables; mean age was 69.3 years (standard deviation [SD], ±7.9 years).
Among subjects originally assigned to behavioral therapy alone, overall reduction in incontinence increased from a mean of 57.5% to a mean of 88.5% after combined therapy (P=.034). Subjects originally assigned to drug therapy alone demonstrated an improvement from 72.7% reduction in incontinence to a mean 84.3% overall reduction with combined therapy (P=.001).
These data suggest that combined therapy can be more effective than behavioral therapy or drug therapy alone. The impact of this study is limited, however, by the relatively low percentage (12.7%) of patients who had received behavioral therapy and chose to add drug therapy, compared with the 41.5% who moved from drug therapy alone to add behavioral therapy.
Furthermore, subjects were self-selected: They chose to continue with an additional 8 weeks of therapy after their initial suboptimal outcome. It is possible that some subjects who were neither totally continent nor completely satisfied with initial therapy chose not to continue with the crossover segment of the trial because it posed too great a burden or because they were discouraged with the initial degree of improvement.
Generalizing these results to all older women with UUI is difficult. The authors point out, however, that, in practice, patients may be more likely than not to choose combination therapy in the hope of shortening the duration of medical therapy. Although it isn’t known whether providing combination therapy from the outset would have yielded better outcomes than either single therapy did, the authors hypothesize that initial combination therapy may result in greater improvement because patients have a high level of motivation and expectation of improvement at the beginning of treatment.
Importance of this article. The investigators demonstrated that a combination of behavioral and drug therapies can provide increased effectiveness in patients for whom each treatment alone led to suboptimal satisfaction. Furthermore, by targeting women older than 55 years, the investigators were able to demonstrate this effectiveness in a group for whom pelvic-floor training may be more difficult than it is for younger women.
It will be interesting to see if future research will 1) validate these findings and 2) determine whether combined therapy can reduce the duration of drug therapy in this older population through behavioral modification and pelvic floor reeducation.
Fluid management
This first-step therapy can involve providing a handout to the patient that details techniques she can use to monitor and control her fluid intake in a manner that addresses her problem. Among such steps:
- avoiding caffeine and artificial sweeteners
- tracking her diet to identify any other bladder irritants
- limiting fluids before times she is more likely to be incontinent—during a long drive, for example, or, in the case of nocturia, after the evening meal.
Scheduled voiding
With scheduled, or prompted, voiding, the patient empties her bladder at a set interval—usually, every 1.5 to 2 hours. If nocturia, or the more severe enuresis, is a problem, the patient can be prompted by an alarm clock or (if she is institutionalized) by nursing staff. Combining scheduled voiding with fluid management principles helps the patient avoid reaching a bladder volume at which an episode of incontinence becomes more likely.
Bladder training
This is a modification of scheduled voiding that attempts to establish a normal voiding interval in patients who have significant frequency but a small voided volume. It imposes a regimented voiding schedule that gradually (over 7 to 10 days) extends the duration between voids.
Pelvic floor-muscle exercises
The focus here is on using pelvic-floor muscles to prevent incontinence. The muscles are strengthened by having the patient perform Kegel exercises (named for Arnold H. Kegel, MD, who, in 1948, recognized the role of pelvic floor-muscle rehabilitation in the treatment of incontinence). The exercises involve simultaneous 1) contraction of the pelvic and periurethral musculature and 2) relaxation of other muscles, including abdominal muscles, which can increase pressure on the bladder.
Once the patient learns to perform Kegel exercises, she can use them to suppress urgency: Instead of hurrying to the bathroom when urgency arises, she is encouraged to sit down, relax, and contract the pelvic-floor muscles repeatedly until the urge to void diminishes. Once it does, the patient proceeds to the toilet to void normally.
Pelvic exam
By self-exam, the patient can identify and familiarize herself with her purposeful contractions of the pelvic-floor musculature and thereby strengthen those muscles with effective exercise.
Biofeedback
Direct feedback about contractions of the pelvic-floor muscles—by a display of data on a gauge or computer monitor, gathered using an intravaginal or anorectal sensor or probe—allows a patient who is exercising those muscles to better target her efforts and maximize their effectiveness.
Combining behavioral therapy and an anticholinergic medication for urge urinary incontinence may yield a superior result after either modality alone has been disappointing by the patient’s account of success.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
Does oxybutynin for UUI further erode cognition in elderly women who are cognitively impaired?
Lackner TE, Wyman JF, McCarthy TC, Monigold M, Davey C. Randomized, placebo-controlled trial of the cognitive effect, safety, and tolerability of oral extended-release oxybutynin in cognitively impaired nursing home residents with urge urinary incontinence. J Am Geriatr Soc. 2008;56:862–870.
Although anticholinergic therapy is modestly effective against UUI in nursing home residents, past studies have suggested that such treatment can impair, or further impair, cognition in this population—a concern that may lead to underuse. This double-blinded, randomized, placebo-controlled trial compared short-term oral extended-release oxybutynin with placebo.
Consequently, the authors sought to determine the cognitive effect, safety, and tolerability of 5 mg/day oral extended-release oxybutynin (the most commonly prescribed dosage) in cognitively impaired older nursing home residents who have UUI.
Subjects were eligible if they:
- were 65 years or older
- had UUI
- lived in a nursing home longer than 3 months
- had cognitive impairment.
Women already being treated for urinary incontinence, those who had an indwelling Foley catheter or urinary retention, and those who were bed-bound or incommunicative were excluded.
Fifty women, mean age 88.6 years (SD, ±6.2), from 12 nursing home facilities, agreed to participate. They were further stratified based on the score of a Mini-Mental State Exam (MMSE): 13 had severe cognitive impairment (MMSE score, 5–10) and 37 had mild or moderate impairment (score, 11–23).
Subjects were randomized to 4 weeks’ treatment with either 5 mg/day oral extended-release oxybutynin or one placebo tablet daily. A nurse practitioner who was blinded to randomization collected all data. The Confusion Assessment Method (CAM) algorithm, MMSE, and Severe Impairment Battery (SIB) were used to assess cognitive decline. The Brief Agitation Rating Scale (BARS) assessed agitation.
No baseline differences were noted with regard to: age; demographic, functional, and neuropsychiatric characteristics; clinical factors predisposing to delirium; and serum anticholinergic activity. Adherence was similar in the treatment (97%) and placebo (97.4%) groups.
Finding: Cognitive impairment. Treatment and placebo groups in the baseline mild-or-moderate stratum (by MMSE) showed equivalent mean changes in CAM scores at all time points. Because of the small sample size, however, CAM score equivalence could not be definitively determined for the groups in the severe impairment stratum. Evaluation of mean MMSE and BARS scores showed no significant changes between groups.
Finding: Tolerability. Excellent tolerability was noted in the treatment group: 96% of subjects completed the trial (compared with 92% of the placebo group). No difference in the rate of adverse events was noted between treatment and placebo groups; of adverse events recorded, 90% were judged “mild” by the investigators. Constipation and dry mouth were most common.
Finding: Falls. More than half—54%—of subjects in both groups experienced at least one fall during the trial or during the preceding or following 3 months. Despite this, no difference in the rate of falls between the treatment and placebo groups was noted. Furthermore, regression analysis revealed no treatment or period effect on falls per month across the time of observation.
Conclusions. Treatment with 5 mg/day oral extended-release oxybutynin in older patients with some cognitive impairment is well tolerated, the study’s findings suggest, with minimal risk of further cognitive decline or delirium over the short term. The potential that long-term therapy has to harm cognitive function remains, however; data on long-term treatment are needed to illuminate that area.
The authors also address the importance of dosing, especially over time, and discuss the lower potential of newer-generation anticholinergics to produce cognitive impairment.
A limited number of articles in the medical literature address anticholinergics in an older population, specifically, and only a few of those evaluated the effects of the drugs on cognitive function. By investigating patients who had an existing cognitive impairment, the authors of this article were able to target a cohort at risk of further cognitive impairment from medication use—thereby giving further weight to their findings of no significant effect.
Main strengths and limitations of the study. The investigators used validated, standardized cognitive tests that were administered by a uniform blinded evaluator in a randomized, controlled trial. The study was limited, however, because patients were evaluated only over a relatively short period (1 month) and because the efficacy of therapy was not addressed.
Further studies of anticholinergic medications, using the same rigorous scientific approach that these investigators applied, are needed to address 1) the long-term efficacy of oxybutynin and similar agents and 2) the cognitive effects of long-term treatment in this older population.
Further impairment is unlikely over the short term when a cognitively impaired nursing home patient who has urge urinary incontinence is treated with 5 mg/day oral extended-release oxybutynin.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
The authors report no financial relationships relevant to this article.
Overactive bladder (OAB)—urinary urgency, with or without incontinence, usually with frequency and nocturia1—is a common problem among women who seek care from an ObGyn. In fact, the condition is estimated to carry a health-care cost in excess of $12 billion annually in the United States.2
A recent community-based survey in Norway estimated the prevalence of urinary incontinence there to be 27% in women between the ages of 65 and 69 years and 35% to 40% in those 80 years or older.3 A population-based study in the United States suggested an even higher rate of urinary incontinence here: greater than 50% in women 60 years or older, with 1) urge urinary incontinence (UUI) predominating4 and 2) the prevalence particularly high among older women who are homebound or who live in a long-term care facility.5
OAB can undermine quality of life in several ways: social isolation, anxiety, poor sleep, higher risk of fracture after a fall,6 reduced ability to function, and poor self-perception. Despite these harmful effects, many women delay seeking care for OAB because they are embarrassed to talk about it with their physician.
Treatment by generalists is feasible—but there is a catch
It’s possible to treat most patients with OAB without referral to a specialist. Two common concerns, however, may set up a roadblock to successful management: the adverse effects associated with some agents and suboptimal control of symptoms.
In this Update, we review recent findings about 1) the potential that anticholinergic therapy has for impairing cognitive function in the older population of women and 2) the important role that concomitant behavioral therapy plays in the long-term success of, and patients’ satisfaction with, treatment of OAB.
Behavioral therapy for OAB: Is it worth all the effort?
Burgio KL, Locher JL, Goode PS. Combined behavioral and drug therapy for urge incontinence in older women. J Am Geriatr Soc. 2000;48:370–374.
The authors of this article followed a randomized clinical trial of older women that compared behavioral and drug therapy for OAB. In the trial, biofeedback-assisted behavioral training (comprising anorectal biofeedback, urge strategies, pelvic muscle biofeedback, and practitioner-directed review with optimization) was compared with treatment with oxybutynin, between 2.5 and 15 mg/day. Both biofeedback-assisted behavioral therapy and the drug regimen were found effective, although neither treatment provided an entirely satisfactory result for all patients. (For a brief description of what constitutes behavioral treatment, see “6 tenets of behavioral therapy for urge urinary incontinence.”)
Second phase of the trial. To determine if treatment satisfaction could be enhanced, the investigators performed a modified crossover study to determine whether combination therapy—biofeedback-assisted behavioral training plus oxybutynin—added any benefit over treatment with behavioral therapy or drug therapy alone. Eligibility was determined by age (55 years or older), demonstrated UUI for at least 3 months, and incomplete dryness or incomplete satisfaction with the outcome of 8 weeks of single-intervention treatment (with either treatment) during the initial phase of the trial.
This subgroup was offered an additional 8 weeks of combination therapy. The primary outcome measure was a reduction in the frequency of episodes of incontinence episodes as recorded by subjects in a bladder diary.
Of 197 women who participated in the original randomized clinical trial, 35—27 who completed drug therapy and 8 who completed behavioral treatment—elected to receive combination therapy. Those 35 subjects did not differ in any of the multiple baseline variables; mean age was 69.3 years (standard deviation [SD], ±7.9 years).
Among subjects originally assigned to behavioral therapy alone, overall reduction in incontinence increased from a mean of 57.5% to a mean of 88.5% after combined therapy (P=.034). Subjects originally assigned to drug therapy alone demonstrated an improvement from 72.7% reduction in incontinence to a mean 84.3% overall reduction with combined therapy (P=.001).
These data suggest that combined therapy can be more effective than behavioral therapy or drug therapy alone. The impact of this study is limited, however, by the relatively low percentage (12.7%) of patients who had received behavioral therapy and chose to add drug therapy, compared with the 41.5% who moved from drug therapy alone to add behavioral therapy.
Furthermore, subjects were self-selected: They chose to continue with an additional 8 weeks of therapy after their initial suboptimal outcome. It is possible that some subjects who were neither totally continent nor completely satisfied with initial therapy chose not to continue with the crossover segment of the trial because it posed too great a burden or because they were discouraged with the initial degree of improvement.
Generalizing these results to all older women with UUI is difficult. The authors point out, however, that, in practice, patients may be more likely than not to choose combination therapy in the hope of shortening the duration of medical therapy. Although it isn’t known whether providing combination therapy from the outset would have yielded better outcomes than either single therapy did, the authors hypothesize that initial combination therapy may result in greater improvement because patients have a high level of motivation and expectation of improvement at the beginning of treatment.
Importance of this article. The investigators demonstrated that a combination of behavioral and drug therapies can provide increased effectiveness in patients for whom each treatment alone led to suboptimal satisfaction. Furthermore, by targeting women older than 55 years, the investigators were able to demonstrate this effectiveness in a group for whom pelvic-floor training may be more difficult than it is for younger women.
It will be interesting to see if future research will 1) validate these findings and 2) determine whether combined therapy can reduce the duration of drug therapy in this older population through behavioral modification and pelvic floor reeducation.
Fluid management
This first-step therapy can involve providing a handout to the patient that details techniques she can use to monitor and control her fluid intake in a manner that addresses her problem. Among such steps:
- avoiding caffeine and artificial sweeteners
- tracking her diet to identify any other bladder irritants
- limiting fluids before times she is more likely to be incontinent—during a long drive, for example, or, in the case of nocturia, after the evening meal.
Scheduled voiding
With scheduled, or prompted, voiding, the patient empties her bladder at a set interval—usually, every 1.5 to 2 hours. If nocturia, or the more severe enuresis, is a problem, the patient can be prompted by an alarm clock or (if she is institutionalized) by nursing staff. Combining scheduled voiding with fluid management principles helps the patient avoid reaching a bladder volume at which an episode of incontinence becomes more likely.
Bladder training
This is a modification of scheduled voiding that attempts to establish a normal voiding interval in patients who have significant frequency but a small voided volume. It imposes a regimented voiding schedule that gradually (over 7 to 10 days) extends the duration between voids.
Pelvic floor-muscle exercises
The focus here is on using pelvic-floor muscles to prevent incontinence. The muscles are strengthened by having the patient perform Kegel exercises (named for Arnold H. Kegel, MD, who, in 1948, recognized the role of pelvic floor-muscle rehabilitation in the treatment of incontinence). The exercises involve simultaneous 1) contraction of the pelvic and periurethral musculature and 2) relaxation of other muscles, including abdominal muscles, which can increase pressure on the bladder.
Once the patient learns to perform Kegel exercises, she can use them to suppress urgency: Instead of hurrying to the bathroom when urgency arises, she is encouraged to sit down, relax, and contract the pelvic-floor muscles repeatedly until the urge to void diminishes. Once it does, the patient proceeds to the toilet to void normally.
Pelvic exam
By self-exam, the patient can identify and familiarize herself with her purposeful contractions of the pelvic-floor musculature and thereby strengthen those muscles with effective exercise.
Biofeedback
Direct feedback about contractions of the pelvic-floor muscles—by a display of data on a gauge or computer monitor, gathered using an intravaginal or anorectal sensor or probe—allows a patient who is exercising those muscles to better target her efforts and maximize their effectiveness.
Combining behavioral therapy and an anticholinergic medication for urge urinary incontinence may yield a superior result after either modality alone has been disappointing by the patient’s account of success.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
Does oxybutynin for UUI further erode cognition in elderly women who are cognitively impaired?
Lackner TE, Wyman JF, McCarthy TC, Monigold M, Davey C. Randomized, placebo-controlled trial of the cognitive effect, safety, and tolerability of oral extended-release oxybutynin in cognitively impaired nursing home residents with urge urinary incontinence. J Am Geriatr Soc. 2008;56:862–870.
Although anticholinergic therapy is modestly effective against UUI in nursing home residents, past studies have suggested that such treatment can impair, or further impair, cognition in this population—a concern that may lead to underuse. This double-blinded, randomized, placebo-controlled trial compared short-term oral extended-release oxybutynin with placebo.
Consequently, the authors sought to determine the cognitive effect, safety, and tolerability of 5 mg/day oral extended-release oxybutynin (the most commonly prescribed dosage) in cognitively impaired older nursing home residents who have UUI.
Subjects were eligible if they:
- were 65 years or older
- had UUI
- lived in a nursing home longer than 3 months
- had cognitive impairment.
Women already being treated for urinary incontinence, those who had an indwelling Foley catheter or urinary retention, and those who were bed-bound or incommunicative were excluded.
Fifty women, mean age 88.6 years (SD, ±6.2), from 12 nursing home facilities, agreed to participate. They were further stratified based on the score of a Mini-Mental State Exam (MMSE): 13 had severe cognitive impairment (MMSE score, 5–10) and 37 had mild or moderate impairment (score, 11–23).
Subjects were randomized to 4 weeks’ treatment with either 5 mg/day oral extended-release oxybutynin or one placebo tablet daily. A nurse practitioner who was blinded to randomization collected all data. The Confusion Assessment Method (CAM) algorithm, MMSE, and Severe Impairment Battery (SIB) were used to assess cognitive decline. The Brief Agitation Rating Scale (BARS) assessed agitation.
No baseline differences were noted with regard to: age; demographic, functional, and neuropsychiatric characteristics; clinical factors predisposing to delirium; and serum anticholinergic activity. Adherence was similar in the treatment (97%) and placebo (97.4%) groups.
Finding: Cognitive impairment. Treatment and placebo groups in the baseline mild-or-moderate stratum (by MMSE) showed equivalent mean changes in CAM scores at all time points. Because of the small sample size, however, CAM score equivalence could not be definitively determined for the groups in the severe impairment stratum. Evaluation of mean MMSE and BARS scores showed no significant changes between groups.
Finding: Tolerability. Excellent tolerability was noted in the treatment group: 96% of subjects completed the trial (compared with 92% of the placebo group). No difference in the rate of adverse events was noted between treatment and placebo groups; of adverse events recorded, 90% were judged “mild” by the investigators. Constipation and dry mouth were most common.
Finding: Falls. More than half—54%—of subjects in both groups experienced at least one fall during the trial or during the preceding or following 3 months. Despite this, no difference in the rate of falls between the treatment and placebo groups was noted. Furthermore, regression analysis revealed no treatment or period effect on falls per month across the time of observation.
Conclusions. Treatment with 5 mg/day oral extended-release oxybutynin in older patients with some cognitive impairment is well tolerated, the study’s findings suggest, with minimal risk of further cognitive decline or delirium over the short term. The potential that long-term therapy has to harm cognitive function remains, however; data on long-term treatment are needed to illuminate that area.
The authors also address the importance of dosing, especially over time, and discuss the lower potential of newer-generation anticholinergics to produce cognitive impairment.
A limited number of articles in the medical literature address anticholinergics in an older population, specifically, and only a few of those evaluated the effects of the drugs on cognitive function. By investigating patients who had an existing cognitive impairment, the authors of this article were able to target a cohort at risk of further cognitive impairment from medication use—thereby giving further weight to their findings of no significant effect.
Main strengths and limitations of the study. The investigators used validated, standardized cognitive tests that were administered by a uniform blinded evaluator in a randomized, controlled trial. The study was limited, however, because patients were evaluated only over a relatively short period (1 month) and because the efficacy of therapy was not addressed.
Further studies of anticholinergic medications, using the same rigorous scientific approach that these investigators applied, are needed to address 1) the long-term efficacy of oxybutynin and similar agents and 2) the cognitive effects of long-term treatment in this older population.
Further impairment is unlikely over the short term when a cognitively impaired nursing home patient who has urge urinary incontinence is treated with 5 mg/day oral extended-release oxybutynin.—JOHN P. JUDD, MD, AND CINDY L. AMUNDSEN, MD
1. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Am J Obstet Gynecol. 2002;187:116-126.
2. Hu TW, Wagner TH, Bentkover JD, et al. Estimated economic costs of overactive bladder in the United States. Urology. 2003;61:1123-1128.
3. Hannestad YS, Rortveit G, Sandvik H, Hunskaar S. A community-based epidemiological survey of female urinary incontinence: the Norwegian EPINCONT study. Epidemiology of Incontinence in the County of Nord-Trøndelag. J Clin Epidemiol. 2000;53:1150-1157.
4. Melville JL, Katon W, Delaney K, Newton K. Urinary incontinence in US women: a population-based study. Arch Intern Med. 2005;165:537-542.
5. Fantl JA, Newman DK, Colling J, et al. Managing Acute and Chronic Urinary Incontinence. Clinical Practice Guideline. Quick Reference Guide for Clinicians, No. 2, 1996 Update. Rockville, MD: US Department of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research. AHCPR Pub. No. 96-0686. January 1996. Available at: http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat6.chapter.32554. Accessed September 11, 2009.
6. Brown JS, Vittinghoff E, Wyman JF, et al. Urinary incontinence: does it increase risk for falls and fractures? Study of Osteoporotic Fractures Research Group. J Am Geriatr Soc. 2000;48:721-725.
1. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Am J Obstet Gynecol. 2002;187:116-126.
2. Hu TW, Wagner TH, Bentkover JD, et al. Estimated economic costs of overactive bladder in the United States. Urology. 2003;61:1123-1128.
3. Hannestad YS, Rortveit G, Sandvik H, Hunskaar S. A community-based epidemiological survey of female urinary incontinence: the Norwegian EPINCONT study. Epidemiology of Incontinence in the County of Nord-Trøndelag. J Clin Epidemiol. 2000;53:1150-1157.
4. Melville JL, Katon W, Delaney K, Newton K. Urinary incontinence in US women: a population-based study. Arch Intern Med. 2005;165:537-542.
5. Fantl JA, Newman DK, Colling J, et al. Managing Acute and Chronic Urinary Incontinence. Clinical Practice Guideline. Quick Reference Guide for Clinicians, No. 2, 1996 Update. Rockville, MD: US Department of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research. AHCPR Pub. No. 96-0686. January 1996. Available at: http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat6.chapter.32554. Accessed September 11, 2009.
6. Brown JS, Vittinghoff E, Wyman JF, et al. Urinary incontinence: does it increase risk for falls and fractures? Study of Osteoporotic Fractures Research Group. J Am Geriatr Soc. 2000;48:721-725.
How to avert postoperative wound complication—and treat it when it occurs
The authors report no financial relationships relevant to this article.
Despite advances in medicine and surgery over the past century, postoperative wound complication remains a serious challenge. When a complication occurs, it translates into prolonged hospitalization, lost time from work, and greater cost to the patient and the health-care system.
Prevention of wound complication begins well before surgery. Requirements include:
- understanding of wound healing (see below) and the classification of wounds (TABLE 1)
- thorough assessment of the patient for risk factors for impaired wound healing, such as diabetes or use of corticosteroid medication (TABLE 2)
- antibiotic prophylaxis, if indicated (TABLE 3)
- good surgical technique, gentle tissue handling, and meticulous hemostasis
- placement of a drain, when appropriate
- awareness of technology that can enhance healing
- close monitoring in the postoperative period
- intervention at the first hint of abnormality.
In this article, we describe predisposing factors and preventive techniques and measures, and outline the most common wound complications, from seroma to dehiscence, including effective management strategies.
It was pioneering Scottish surgeon John Hunter who noted that “injury alone has in all cases the tendency to produce the disposition and means of a cure.”15
Unlike the tissue regeneration that occurs primarily in lower animals, human wound healing is mediated by collagen deposition, or scarring, which provides structural support to the wound. This scarring process may itself cause a variety of clinical problems.
Wound healing is characterized by overlapping, largely interdependent phases, with no clear demarcation between them. Failure in one phase may have a negative impact on the overall outcome.
In general, wound healing involves two phases: inflammation and proliferation. Within these phases, the following processes occur: scar maturation, wound contraction, and epithelialization. These repair mechanisms are activated in response to tissue injury even when it is surgically induced.
Inflammatory phase
The initial response to tissue injury is inflammation, which is mediated by various amines, enzymes, and other substances. This inflammation can be further broken down into vascular and cellular responses.
Inflammation triggers increased blood flow and migration of neutrophils, monocytes, macrophages, and other cells into the wound.
The first burst of blood acts to cleanse the wound of foreign debris. It is followed by vasoconstriction, which is mediated by thromboxane 2, to decrease blood loss. Vasodilation then occurs once histamine and serotonin are released, permitting increased blood flow to the wound. The surge in blood flow accounts for the increased warmth and redness of the wound. Vasodilation also increases capillary permeability, allowing the migration of red blood cells, platelets, leukocytes, plasma, and other tissue fluids into the interstitium of the wound. This migration accounts for wound edema.
In the cellular response, which is facilitated by increased blood flow, cell migration occurs as part of an immune response. Neutrophils, the first cells to enter the wound, engage in phagocytosis of bacteria and debris. Subsequently, there is migration of monocytes, macrophages, and other cells. This nonspecific immune response is sustained by prostaglandins, aided by complement factors and cytokines. A specific immune response follows, aimed at destroying specific antigens, and involves both B- and T-lymphocytes.
Proliferative phase
Proliferation is characterized by the infiltration of endothelial cells and fibroblasts and subsequent collagen deposition along a previously formed fibrin network. This new, highly vascularized tissue assumes a granular appearance—hence, the term “granulation tissue.”
Collagen that is deposited in the wound undergoes maturation and remodeling, increasing the tensile strength of the wound. The process continues for months after the initial insult.
All wounds undergo some degree of contraction, but the process is more relevant in wounds that remain open or involve significant tissue loss.
Last, the external covering of the wound is restored by epithelialization.
TABLE 1
Classification of surgical wounds
| CLASS I – Clean wounds (infection rate <5%) |
|
| CLASS II – Clean–contaminated wounds (infection rate 2–10%) |
|
| CLASS III – Contaminated wounds (infection rate 15–20%) |
|
| CLASS IV – Dirty or infected wounds (infection rate >30%) |
|
| SOURCE: Centers for Disease Control and Prevention and the American College of Surgeons |
TABLE 2
Risk factors for poor wound healing and dehiscence
| Poor wound healing |
|
| Abdominal wound dehiscence |
|
| SOURCE: Carlson,11 Cliby12. |
Conditions and drugs that impair healing
Preexisting medical conditions may limit healing, especially conditions associated with diminished delivery of oxygen and nutrients to healing tissues.
Diabetes can damage the vasculature and may impair healing if the blood glucose level is markedly elevated in the perioperative period. Such an elevation impedes transport of vitamin C, a key component of collagen synthesis.
Malignancy and immunosuppressive disorders may prevent optimal healing by compromising the immune response.
Bacterial vaginosis, a common polymicrobial infection involving aerobic and anaerobic bacteria, appears to be associated with postoperative febrile morbidity and surgical-site infection, particularly after hysterectomy.1 Current guidelines recommend that medical therapy for bacterial vaginosis be instituted at least 4 days before surgery and continued postoperatively.
Because steroids, NSAIDs, and chemotherapy agents impede wound healing, and anticoagulants may interfere with granulation, it is crucial to review the patient’s medications well in advance of surgery.
Nutrition plays a critical role
The importance of nutrition cannot be overstated. A significant percentage of patients are thought to have some degree of nutritional deficiency preoperatively. This deficiency may alter the inflammatory response, impair collagen synthesis, and reduce the tensile strength of the wound.
Because healing requires energy, deficits in carbohydrates may limit protein utilization, and deficiencies of vitamins and micronutrients can also interfere with healing.2
Obesity, too, increases the risk of postoperative wound complication. Markedly obese patients have a thick, avascular, subcutaneous layer of fat that compromises healing.3
Meticulous technique required
Good surgical technique and appropriate use of antibiotics are critical components of successful wound healing.
When placing the incision, avoid the moist, bacteria-laden subpannicular crease in the markedly obese.
During a procedure, handle tissue gently, keep it moist, and make minimal use of electrocautery to reduce tissue injury and promote healing. Keep operating time and blood loss to a minimum, and debride the wound of any foreign material and devitalized tissue.
Multiple studies have demonstrated that judicious use of prophylactic antibiotics significantly decreases the incidence of wound infection, particularly in relation to hysterectomy and vaginal procedures and when entry into bowel is anticipated.4,5 A number of prophylactic regimens are given in TABLE 3.
Meticulous hemostasis at the time of closure is imperative. When complete hemostasis cannot be confirmed, place a small drain in the subcutaneous space (or subfascial space, if there is oozing on the muscle bed) and apply a pressure dressing to help prevent hematoma. Although a drain is not a substitute for precise hemostasis or careful surgical technique, it may be helpful when there is concern about oozing or a “wet” surface, or when the patient is markedly obese.
Some practitioners have expressed concern over the risk of bacterial migration and infection with placement of a drain, but others, including us, advocate use of a drain in the subcutaneous space to help remove residual blood, fluid, and other debris to prevent the formation of dead space and infection and promote wound closure and healing. In a small study, Gallup and associates demonstrated a decreased incidence of wound breakdown when a drain was placed.6
A closed-suction drain, such as a Jackson-Pratt or Hemovac model, helps minimize wound complication when it is placed in the subcutaneous layer. (Avoid a rubber Penrose drain because it may allow bacteria to enter the wound.) It is imperative that the drain exit the body via a separate site and not through the incision itself. We advocate removal when less than 30 mL of fluid accumulates in the reservoir over 24 hours.
TABLE 3
3 prophylactic antibiotic regimens
| Procedure | Antibiotic | Single intravenous dose |
|---|---|---|
| Hysterectomy and urogynecologic procedures, including those that involve mesh | Cefazolin | 1 g or 2 g |
| Clindamycin plus gentamicin, a quinolone, or aztreonam | 600 mg plus 1.5 mg/kg, 400 mg, or 1 g, respectively | |
| Metronidazole plus gentamicin or a quinolone | 500 mg plus 1.5 mg/kg or 400 mg, respectively | |
| SOURCE: American College of Obstetricians and Gynecologists5 | ||
Fluid within the wound does not always indicate infection
Wound collections are not necessarily indicative of infection; collections of fluid within the wound may represent a serous transudate, blood, pus, or a combination of these. If the fluid is not addressed, however, fulminant infection may be the result.
Seroma is usually painless
A seroma is a collection of wound exudates within the dead space. Seroma typically involves thin, pink, watery discharge and minimal edge separation. In some cases, there may be surrounding edema but generally little to no tenderness.
When a seroma is detected, remove the staples or stitches in the area of concern and explore the wound. It is essential to ensure fascial integrity, as serous wound drainage may be a sign of impending evisceration. After these measures are taken, cleanse and lightly pack the wound to permit drainage.
Hematoma requires identification of the source of bleeding
Hematoma represents blood or a blood clot within the tissues beneath the skin. It may be caused by persistent bleeding of a vessel, although the pressure within the wound and the pressure produced by the dressing often provide tamponade on the bleeding source, in which case the hematoma forms with no active bleeding.
Hematoma is usually caused by small bleeding vessels that were not apparent at the time of surgery or were not cauterized or ligated at the time of closure. For this reason, it is important to achieve good hemostasis and a “dry” wound before closing the skin.
When hematoma is suspected, open the wound enough to permit adequate exposure and identify the source of bleeding. Evacuate as much blood and clot as possible because blood is an ideal medium for bacterial growth. If active bleeding is found, use a silver nitrate applicator or handheld cautery pen to accomplish hemostasis at bedside. If bleeding is more severe, or the source cannot be visualized, consider returning to the operating room for more extensive exploration.
Once hemostasis is achieved, irrigate the wound copiously and institute local wound care.
How common is infection?
Before it is possible to address this question, it is necessary to clarify the terminology of infection. Contamination and colonization are different entities. The first refers to the presence of bacteria without multiplication. The latter describes the multiplication of bacteria in the absence of a host response. When infection is present, bacterial proliferation produces clinical signs and symptoms.
Postoperative abdominal wound infection occurs in about 5% of cases but may be more common in procedures that involve a greater level of contamination.7 One study found a 12% incidence of wound infection, but the rate declined to 8% when antibiotic prophylaxis was instituted.4
Several other studies have examined determinants of infection. For example, a large Cochrane review found no real differences in infection rate by preoperative skin preparation technique or agent, but it did observe that one study had demonstrated the superiority of chlorhexidine to other cleansing agents.8
Cruse and Foord also noted the slight superiority of chlorhexidine, as well as the efficacy of clipping abdominal hair immediately before surgery.7
When identifying organisms, look for the usual suspects
The offending pathogens in infection are usually endogenous flora found on the patient’s skin and within hollow organs (vagina, bowel). The organisms most commonly responsible for infection are Staphylococcus (aureus, epidermidis), enterococci, and Escherichia coli. However, the bacteria identified in the wound may not be the causative organism.
Most infections typically become clinically apparent between the fifth and 10th postoperative days, often after the patient has been discharged, although they may appear much earlier or much later. One of us (Dr. Perkins) had a patient who presented with a suppurative infection after undergoing hysterectomy for endometrial carcinoma 5 months earlier.
Cellulitis is common
Wound cellulitis, a common, nonsuppurative infection of skin and underlying connective tissue, is generally not severe. The wound assumes a brawny, reddish brown appearance associated with edema, warmth, and erythema. Fever is not always present.
It is important to remember that cellulitis may surround a deeper infection. Although needle aspiration of the leading edge has been advocated, it yields a positive culture in only 20% to 40% of cases.
In the absence of purulent drainage, treat cellulitis with antibiotics, utilizing sulfamethoxazole-trimethroprim, a cephalosporin, or augmented penicillin, and apply warm packs to the wound.
If purulent drainage is seen, or the patient fails to improve significantly within 24 hours, suspect an abscess or resistant organism.
- During preoperative evaluation, assess the patient for risk factors, comorbid conditions, and medications that can impair healing
- If the patient is morbidly obese and planning to undergo an elective, nonurgent procedure, consider instituting a plan for preoperative weight loss
- Advise smokers to “kick the habit” 1 or 2 months before surgery
- Avoid using electrocautery in the “coagulation current” setting when incising the fascia
- When approximating the fascia, take wide bites of tissue (1.5 to 2 cm from the edge)
- Avoid excessive suture tension when closing the fascial layer (“Approximate, don’t strangulate”)
- Obtain good hemostasis before closing the wound; consider placing a drain in an obese patient
- In a high-risk patient who has multiple risk factors, consider retention sutures
- Minimize or avoid abdominal distention during the postoperative period with:
- Assess the wound for infection early, and treat infection promptly
- Remember to administer prophylactic antibiotics
Most wound infections are superficial
Approximately 75% of all wound infections involve the skin and subcutaneous tissue layers. Superficial infection is more likely to occur when there is an undrained hematoma, excessively tight sutures, tissue trauma, or a retained foreign material. Edema, erythema, and pain and tenderness may be more pronounced than with cellulitis. A low-grade fever may be present, and incisional discharge typically occurs.
Drainage is the cornerstone of management and requires the removal of staples or sutures from the area. Local exploration is mandatory, and fascial integrity must be confirmed. If a pocket of pus is found, open the wound liberally to determine the extent of the pocket and permit as much evacuation as possible. Wound culture is optional. Institute local wound care and consider adjuvant antibiotics in selected cases.
Ensure fascial integrity
Any infection that arises immediately adjacent to the fascia may have an intra-abdominal component, although that is unlikely. Extensive exploration is warranted to assess fascial integrity.
If intra-abdominal infection is suspected, order appropriate imaging.
Patients who have deep infection usually exhibit frank, purulent discharge; fever; and severe pain. Marked separation of wound edges is often present as well, as is an elevated white blood cell count.
As with superficial infection, the key to therapy is liberal exploration, drainage of the abscess cavity, and mechanical wound debridement. Irrigate the wound copiously using a dilute mixture of saline and hydrogen peroxide to remove any remaining debris. Avoid povidone-iodine solution because it inhibits normal tissue granulation.
The wound may be left open to heal by secondary intention, or it may be closed secondarily after 3 to 6 days, provided there is no evidence of infection and a healthy granulating bed is present.
Consider adjuvant antibiotics, especially when the patient is immunocompromised.
If the wound has pronounced edema and unusual discoloration, consider a serious infection such as necrotizing fasciitis.
Wound dehiscence raises risk of evisceration
Dehiscence of the abdominal incision occurs when the various layers separate. Dehiscence may be extrafascial (superficial disruption of the skin and subcutaneous tissue only) or may involve all layers, including peritoneum (complete fascial dehiscence or burst abdomen).
When bowel or omentum extrudes, the term evisceration is appropriate.
In several reviews of the literature, the incidence of dehiscence ranged from 0.4% in earlier studies to 1% to 3% in later reviews.9-12 Despite advances in preoperative and postoperative care, suture materials, surgical technique, and antibiotics, fascial dehiscence remains a serious problem in abdominal surgery.
What causes wound disruption?
To a great extent, abdominal wound breakdown is a function of surgical technique and method of closure. Although the conventional wisdom is that dehiscence occurs less frequently with a transverse incision than with a vertical one, this assumption is being challenged. A small study by Hendrix and associates found no differences in the rate of dehiscence by type of incision.13 That finding suggests that the incidence of dehiscence is inversely related to the strength of closure.
Selection of the appropriate suture material also is important. In addition, use of electrocautery in the “cutting current” mode when the abdomen is opened causes less tissue injury than “coagulation current.” The latter has a greater thermal effect, thereby weakening the fascial layer.
Patient characteristics that influence wound integrity include comorbidities such as diabetes and malignancy, recent corticosteroid administration, and malnutrition.
Although infection may accompany superficial wound separation, its role in complete dehiscence is unclear.
Conditions that cause abdominal distention, such as severe coughing, vomiting, ileus, and ascites, may contribute to dehiscence, particularly when the closure method is less than satisfactory.
Some authors have found a greater incidence of wound disruption when multiple risk factors are present. In patients who had eight or more risk factors, wound disruption was universal.11,12
Management entails debridement, irrigation, and closure
When extrafascial dehiscence occurs, mechanical debridement and irrigation are usually the only measures necessary before deciding how to close the wound—even if infection is present. Remove all foreign material and excise any devitalized tissue.
As for the method of closure, the choice is usually between secondary closure and leaving the wound open to heal by secondary intention. An alternative to the latter is wound closure after several days, once a healthy granulating bed develops.
Dodson and colleagues described a technique of superficial wound closure that can be performed at the bedside using local anesthesia, with little discomfort to the patient.14 Wound separation caused by a small hematoma or sterile seroma especially lends itself to this type of immediate closure.
Vacuum-assisted closure
The vacuum-assisted wound closure system is a device that speeds healing and reduces the risk of complication. It consists of a sponge dressing that can be sized to fit an open wound and connected to an apparatus that generates negative pressure. The device enhances healing by removing excess fluid and debris and decreasing wound edema.
Argenta and associates reported successful use of this system to expedite healing in three cases of wound failure.16 It can be employed in the home-health setting by nurses trained in its use.
Human acellular dermal matrix
Occasionally, breakdown of a wound creates marked fascial defects that preclude secondary closure. Synthetic materials—both absorbable and nonabsorbable varieties—have been employed to bridge the defect, but their use sometimes leads to adhesions, infection, and cutaneous fistula. These risks are of special concern when the wound is already contaminated or otherwise compromised.
One alternative is human acellular dermal matrix (AlloDerm, LifeCell Corp). Tung and colleagues described its use for repair of a fascial defect in a previously irradiated cancer patient whose postoperative course was complicated by pelvic infection.17 This dermal matrix, a basement membrane taken from cadaveric skin, promotes neovascularization and is thought to be associated with a lower incidence of infection and adhesions than is traditional mesh. It is widely used in the burn setting and in the repair of ventral hernia, but is a relatively new addition to the management of fascial defects associated with wound breakdown.
Growth factors
Wound healing is regulated by a number of entities, including cytokines and growth factors, so it is no surprise that research has turned its focus on them. In a preliminary study, investigators found that separated abdominal wounds closed faster when recombinant human platelet-derived growth factor BB was topically administered than they did when they were left open to close by secondary intention.18
Although their use is not commonplace in wound management, research suggests that growth factors may one day be helpful adjuncts in the care of wound complications.
Complete fascial dehiscence is a “catastrophic” complication
Complete dehiscence of the fascia and extrusion of intra-abdominal contents is a serious catastrophic complication that is associated with a mortality rate of about 20%. It typically occurs between the third and seventh postoperative days, although later occurrences have been reported.
Warning signs of impending evisceration include serous drainage in the absence of obvious infection, and a “popping” sensation on the part of the patient—a feeling that something is “giving way.”
If evisceration occurs, cover exposed bowel with packs soaked in saline or povidone-iodine and prepare the patient for emergency surgery. Institute both hydration and broad-spectrum antibiotics.
Before replacing the abdominal contents, thoroughly irrigate the peritoneal cavity and inspect the bowel carefully, excising any necrotic tissue.
Reapproximate the fascia using interrupted #1 or #2 monofilament suture. Also consider placing retention sutures, particularly when the patient has multiple risk factors for wound complications (FIGURE). Leave the wound open, prepared for later closure.
If the abdomen cannot be closed because of peritonitis or bowel edema, or there is an insufficient amount of fascia remaining, approximate the abdominal wall using bridging sutures over a gauze pack as a temporizing measure until reconstruction can be performed. Consultation with a plastic surgeon or trauma specialist is recommended.
FIGURE Consider retention sutures for high-risk patients
Retention sutures are placed in interrupted fashion to support the primary suture line and are carried through the full thickness of the tissue, from the abdominal wall skin through the fascia and, if possible, the peritoneum. A rubber bolster placed across each suture keeps the suture from cutting into the skin (inset).
Necrotizing fasciitis: Worst of the worst
Necrotizing fasciitis is a dangerous, synergistic, bacterial infection involving the fascia, subcutaneous tissue, and skin. The culprits are multiple bacterial pathogens that include Streptococcus pyogenes, staphylococcal species, gram-negative aerobes, and anaerobes. The infection typically originates at a localized area, spreads along the fascial planes, and ultimately causes septic thrombosis of the vessels penetrating the skin and deeper layers. The result is necrosis. The associated mortality rate is approximately 20%.
The patient who has necrotizing fasciitis typically displays severe pain; anesthetic, edematous skin; purple, necrotic wound edges; hemorrhagic bullae; and crepitus.
Frank necrosis subsequently develops, with surrounding inflammation and edema, and leads to systemic toxicity, with fever, hemodynamic abnormality, and shock. In advanced stages, gangrene is present.
Laboratory evaluation includes a white blood cell count. Biopsy also is recommended. If necrotizing fasciitis is present, biopsy will reveal necrosis and thrombi of vessels passing through the fascia.
Treatment of necrotizing fasciitis requires intravenous, broad-spectrum antibiotics, including penicillin, that are adjusted according to the findings of the wound culture and sensitivity test. Cardiovascular and fluid-volume support is critical, as is wide surgical debridement of all necrosed skin and fascia. The latter, in fact, is the cornerstone of therapy.
1. Lin L, Song J, Kimber N, et al. The role of bacterial vaginosis in infection after major gynecologic surgery. Infect Dis Obstet Gynecol. 1999;7:169-174.
2. Williams JZ, Barbul A. Nutrition and wound healing. Surg Clin North Am. 2003;83:571-596.
3. Perkins JD, Jackson RA. Risks and remedies when your surgical patient is obese. OBG Management. 2007;19(10)34-54.
4. Kamat AA, Brancazio L, Gibson M. Wound infection in gynecologic surgery. Infect Dis Obstet Gynecol. 2000;8:230-234.
5. American College of Obstetricians and Gynecologists. Antibiotic prophylaxis for gynecologic procedures. ACOG Practice Bulletin #104. Washington, DC: ACOG; 2009.
6. Gallup DC, Gallup DG, Nolan TE, Smith RP, Messing MF, Kline KL. Use of a subcutaneous closed drainage system and antibiotics in obese gynecologic patients. Am J Obstet Gynecol. 1996;175:358-362.
7. Cruse PJE, Foord R. The epidemiology of wound infection. A 10-year prospective study of 62,639 wounds. Surg Clin North Am. 1980;60:27-40.
8. Edwards PS, Lipp A, Holmes A. Preoperative skin antiseptics for preventing surgical wound infections after clean surgery. Cochrane Database Syst Rev. 2004;(3):CD003949.-
9. Baggish MS, Lee WK. Abdominal wound disruption. Obstet Gynecol. 1975;46:530-534.
10. Poole GV, Jr. Mechanical factors in abdominal wound closure: the prevention of fascial dehiscence. Surgery. 1985;97:631-640.
11. Carlson MA. Acute wound failure. Surg Clin North Am. 1997;77:607-636.
12. Cliby WA. Abdominal incision wound breakdown. Clin Obstet Gynecol. 2002;45:507-517.
13. Hendrix SL, Schimp V, Martin J, Singh A, Kruger M, McNeeley SG. The legendary superior strength of the Pfannenstiel incision: a myth? Am J Obstet Gynecol. 2000;182:1446-1451.
14. Dodson MK, Magann EF, Sullivan DL, Meeks GR. Extrafascial wound dehiscence: deep en bloc closure versus superficial skin closure. Obstet Gynecol. 1994;83:142-145.
15. Brunicardi FC, Andersen DK, Billiar TR, Dunn DL, Hunter JG, Pollock RE. Chapter 8: Wound healing. In: Schwartz’s Principles of Surgery. 8th ed. New York: McGraw-Hill; 2005.
16. Argenta PA, Rahaman J, Gretz HF, 3rd, Nezhat F, Cohen CJ. Vacuum-assisted closure in the treatment of complex gynecologic wound failures. Obstet Gynecol. 2002;99:497-501.
17. Tung CS, Zighelboim I, Scott B, Anderson ML. Human acellular dermal matrix for closure of a contaminated gynecologic wound. Gynecol Oncol. 2006;103:354-356.
18. Shackelford DP, Fackler E, Hoffman MK, Atkinson S. Use of topical recombinant human platelet-derived growth factor BB in abdominal wound separation. Am J Obstet Gynecol. 2002;186:701-704.
The authors report no financial relationships relevant to this article.
Despite advances in medicine and surgery over the past century, postoperative wound complication remains a serious challenge. When a complication occurs, it translates into prolonged hospitalization, lost time from work, and greater cost to the patient and the health-care system.
Prevention of wound complication begins well before surgery. Requirements include:
- understanding of wound healing (see below) and the classification of wounds (TABLE 1)
- thorough assessment of the patient for risk factors for impaired wound healing, such as diabetes or use of corticosteroid medication (TABLE 2)
- antibiotic prophylaxis, if indicated (TABLE 3)
- good surgical technique, gentle tissue handling, and meticulous hemostasis
- placement of a drain, when appropriate
- awareness of technology that can enhance healing
- close monitoring in the postoperative period
- intervention at the first hint of abnormality.
In this article, we describe predisposing factors and preventive techniques and measures, and outline the most common wound complications, from seroma to dehiscence, including effective management strategies.
It was pioneering Scottish surgeon John Hunter who noted that “injury alone has in all cases the tendency to produce the disposition and means of a cure.”15
Unlike the tissue regeneration that occurs primarily in lower animals, human wound healing is mediated by collagen deposition, or scarring, which provides structural support to the wound. This scarring process may itself cause a variety of clinical problems.
Wound healing is characterized by overlapping, largely interdependent phases, with no clear demarcation between them. Failure in one phase may have a negative impact on the overall outcome.
In general, wound healing involves two phases: inflammation and proliferation. Within these phases, the following processes occur: scar maturation, wound contraction, and epithelialization. These repair mechanisms are activated in response to tissue injury even when it is surgically induced.
Inflammatory phase
The initial response to tissue injury is inflammation, which is mediated by various amines, enzymes, and other substances. This inflammation can be further broken down into vascular and cellular responses.
Inflammation triggers increased blood flow and migration of neutrophils, monocytes, macrophages, and other cells into the wound.
The first burst of blood acts to cleanse the wound of foreign debris. It is followed by vasoconstriction, which is mediated by thromboxane 2, to decrease blood loss. Vasodilation then occurs once histamine and serotonin are released, permitting increased blood flow to the wound. The surge in blood flow accounts for the increased warmth and redness of the wound. Vasodilation also increases capillary permeability, allowing the migration of red blood cells, platelets, leukocytes, plasma, and other tissue fluids into the interstitium of the wound. This migration accounts for wound edema.
In the cellular response, which is facilitated by increased blood flow, cell migration occurs as part of an immune response. Neutrophils, the first cells to enter the wound, engage in phagocytosis of bacteria and debris. Subsequently, there is migration of monocytes, macrophages, and other cells. This nonspecific immune response is sustained by prostaglandins, aided by complement factors and cytokines. A specific immune response follows, aimed at destroying specific antigens, and involves both B- and T-lymphocytes.
Proliferative phase
Proliferation is characterized by the infiltration of endothelial cells and fibroblasts and subsequent collagen deposition along a previously formed fibrin network. This new, highly vascularized tissue assumes a granular appearance—hence, the term “granulation tissue.”
Collagen that is deposited in the wound undergoes maturation and remodeling, increasing the tensile strength of the wound. The process continues for months after the initial insult.
All wounds undergo some degree of contraction, but the process is more relevant in wounds that remain open or involve significant tissue loss.
Last, the external covering of the wound is restored by epithelialization.
TABLE 1
Classification of surgical wounds
| CLASS I – Clean wounds (infection rate <5%) |
|
| CLASS II – Clean–contaminated wounds (infection rate 2–10%) |
|
| CLASS III – Contaminated wounds (infection rate 15–20%) |
|
| CLASS IV – Dirty or infected wounds (infection rate >30%) |
|
| SOURCE: Centers for Disease Control and Prevention and the American College of Surgeons |
TABLE 2
Risk factors for poor wound healing and dehiscence
| Poor wound healing |
|
| Abdominal wound dehiscence |
|
| SOURCE: Carlson,11 Cliby12. |
Conditions and drugs that impair healing
Preexisting medical conditions may limit healing, especially conditions associated with diminished delivery of oxygen and nutrients to healing tissues.
Diabetes can damage the vasculature and may impair healing if the blood glucose level is markedly elevated in the perioperative period. Such an elevation impedes transport of vitamin C, a key component of collagen synthesis.
Malignancy and immunosuppressive disorders may prevent optimal healing by compromising the immune response.
Bacterial vaginosis, a common polymicrobial infection involving aerobic and anaerobic bacteria, appears to be associated with postoperative febrile morbidity and surgical-site infection, particularly after hysterectomy.1 Current guidelines recommend that medical therapy for bacterial vaginosis be instituted at least 4 days before surgery and continued postoperatively.
Because steroids, NSAIDs, and chemotherapy agents impede wound healing, and anticoagulants may interfere with granulation, it is crucial to review the patient’s medications well in advance of surgery.
Nutrition plays a critical role
The importance of nutrition cannot be overstated. A significant percentage of patients are thought to have some degree of nutritional deficiency preoperatively. This deficiency may alter the inflammatory response, impair collagen synthesis, and reduce the tensile strength of the wound.
Because healing requires energy, deficits in carbohydrates may limit protein utilization, and deficiencies of vitamins and micronutrients can also interfere with healing.2
Obesity, too, increases the risk of postoperative wound complication. Markedly obese patients have a thick, avascular, subcutaneous layer of fat that compromises healing.3
Meticulous technique required
Good surgical technique and appropriate use of antibiotics are critical components of successful wound healing.
When placing the incision, avoid the moist, bacteria-laden subpannicular crease in the markedly obese.
During a procedure, handle tissue gently, keep it moist, and make minimal use of electrocautery to reduce tissue injury and promote healing. Keep operating time and blood loss to a minimum, and debride the wound of any foreign material and devitalized tissue.
Multiple studies have demonstrated that judicious use of prophylactic antibiotics significantly decreases the incidence of wound infection, particularly in relation to hysterectomy and vaginal procedures and when entry into bowel is anticipated.4,5 A number of prophylactic regimens are given in TABLE 3.
Meticulous hemostasis at the time of closure is imperative. When complete hemostasis cannot be confirmed, place a small drain in the subcutaneous space (or subfascial space, if there is oozing on the muscle bed) and apply a pressure dressing to help prevent hematoma. Although a drain is not a substitute for precise hemostasis or careful surgical technique, it may be helpful when there is concern about oozing or a “wet” surface, or when the patient is markedly obese.
Some practitioners have expressed concern over the risk of bacterial migration and infection with placement of a drain, but others, including us, advocate use of a drain in the subcutaneous space to help remove residual blood, fluid, and other debris to prevent the formation of dead space and infection and promote wound closure and healing. In a small study, Gallup and associates demonstrated a decreased incidence of wound breakdown when a drain was placed.6
A closed-suction drain, such as a Jackson-Pratt or Hemovac model, helps minimize wound complication when it is placed in the subcutaneous layer. (Avoid a rubber Penrose drain because it may allow bacteria to enter the wound.) It is imperative that the drain exit the body via a separate site and not through the incision itself. We advocate removal when less than 30 mL of fluid accumulates in the reservoir over 24 hours.
TABLE 3
3 prophylactic antibiotic regimens
| Procedure | Antibiotic | Single intravenous dose |
|---|---|---|
| Hysterectomy and urogynecologic procedures, including those that involve mesh | Cefazolin | 1 g or 2 g |
| Clindamycin plus gentamicin, a quinolone, or aztreonam | 600 mg plus 1.5 mg/kg, 400 mg, or 1 g, respectively | |
| Metronidazole plus gentamicin or a quinolone | 500 mg plus 1.5 mg/kg or 400 mg, respectively | |
| SOURCE: American College of Obstetricians and Gynecologists5 | ||
Fluid within the wound does not always indicate infection
Wound collections are not necessarily indicative of infection; collections of fluid within the wound may represent a serous transudate, blood, pus, or a combination of these. If the fluid is not addressed, however, fulminant infection may be the result.
Seroma is usually painless
A seroma is a collection of wound exudates within the dead space. Seroma typically involves thin, pink, watery discharge and minimal edge separation. In some cases, there may be surrounding edema but generally little to no tenderness.
When a seroma is detected, remove the staples or stitches in the area of concern and explore the wound. It is essential to ensure fascial integrity, as serous wound drainage may be a sign of impending evisceration. After these measures are taken, cleanse and lightly pack the wound to permit drainage.
Hematoma requires identification of the source of bleeding
Hematoma represents blood or a blood clot within the tissues beneath the skin. It may be caused by persistent bleeding of a vessel, although the pressure within the wound and the pressure produced by the dressing often provide tamponade on the bleeding source, in which case the hematoma forms with no active bleeding.
Hematoma is usually caused by small bleeding vessels that were not apparent at the time of surgery or were not cauterized or ligated at the time of closure. For this reason, it is important to achieve good hemostasis and a “dry” wound before closing the skin.
When hematoma is suspected, open the wound enough to permit adequate exposure and identify the source of bleeding. Evacuate as much blood and clot as possible because blood is an ideal medium for bacterial growth. If active bleeding is found, use a silver nitrate applicator or handheld cautery pen to accomplish hemostasis at bedside. If bleeding is more severe, or the source cannot be visualized, consider returning to the operating room for more extensive exploration.
Once hemostasis is achieved, irrigate the wound copiously and institute local wound care.
How common is infection?
Before it is possible to address this question, it is necessary to clarify the terminology of infection. Contamination and colonization are different entities. The first refers to the presence of bacteria without multiplication. The latter describes the multiplication of bacteria in the absence of a host response. When infection is present, bacterial proliferation produces clinical signs and symptoms.
Postoperative abdominal wound infection occurs in about 5% of cases but may be more common in procedures that involve a greater level of contamination.7 One study found a 12% incidence of wound infection, but the rate declined to 8% when antibiotic prophylaxis was instituted.4
Several other studies have examined determinants of infection. For example, a large Cochrane review found no real differences in infection rate by preoperative skin preparation technique or agent, but it did observe that one study had demonstrated the superiority of chlorhexidine to other cleansing agents.8
Cruse and Foord also noted the slight superiority of chlorhexidine, as well as the efficacy of clipping abdominal hair immediately before surgery.7
When identifying organisms, look for the usual suspects
The offending pathogens in infection are usually endogenous flora found on the patient’s skin and within hollow organs (vagina, bowel). The organisms most commonly responsible for infection are Staphylococcus (aureus, epidermidis), enterococci, and Escherichia coli. However, the bacteria identified in the wound may not be the causative organism.
Most infections typically become clinically apparent between the fifth and 10th postoperative days, often after the patient has been discharged, although they may appear much earlier or much later. One of us (Dr. Perkins) had a patient who presented with a suppurative infection after undergoing hysterectomy for endometrial carcinoma 5 months earlier.
Cellulitis is common
Wound cellulitis, a common, nonsuppurative infection of skin and underlying connective tissue, is generally not severe. The wound assumes a brawny, reddish brown appearance associated with edema, warmth, and erythema. Fever is not always present.
It is important to remember that cellulitis may surround a deeper infection. Although needle aspiration of the leading edge has been advocated, it yields a positive culture in only 20% to 40% of cases.
In the absence of purulent drainage, treat cellulitis with antibiotics, utilizing sulfamethoxazole-trimethroprim, a cephalosporin, or augmented penicillin, and apply warm packs to the wound.
If purulent drainage is seen, or the patient fails to improve significantly within 24 hours, suspect an abscess or resistant organism.
- During preoperative evaluation, assess the patient for risk factors, comorbid conditions, and medications that can impair healing
- If the patient is morbidly obese and planning to undergo an elective, nonurgent procedure, consider instituting a plan for preoperative weight loss
- Advise smokers to “kick the habit” 1 or 2 months before surgery
- Avoid using electrocautery in the “coagulation current” setting when incising the fascia
- When approximating the fascia, take wide bites of tissue (1.5 to 2 cm from the edge)
- Avoid excessive suture tension when closing the fascial layer (“Approximate, don’t strangulate”)
- Obtain good hemostasis before closing the wound; consider placing a drain in an obese patient
- In a high-risk patient who has multiple risk factors, consider retention sutures
- Minimize or avoid abdominal distention during the postoperative period with:
- Assess the wound for infection early, and treat infection promptly
- Remember to administer prophylactic antibiotics
Most wound infections are superficial
Approximately 75% of all wound infections involve the skin and subcutaneous tissue layers. Superficial infection is more likely to occur when there is an undrained hematoma, excessively tight sutures, tissue trauma, or a retained foreign material. Edema, erythema, and pain and tenderness may be more pronounced than with cellulitis. A low-grade fever may be present, and incisional discharge typically occurs.
Drainage is the cornerstone of management and requires the removal of staples or sutures from the area. Local exploration is mandatory, and fascial integrity must be confirmed. If a pocket of pus is found, open the wound liberally to determine the extent of the pocket and permit as much evacuation as possible. Wound culture is optional. Institute local wound care and consider adjuvant antibiotics in selected cases.
Ensure fascial integrity
Any infection that arises immediately adjacent to the fascia may have an intra-abdominal component, although that is unlikely. Extensive exploration is warranted to assess fascial integrity.
If intra-abdominal infection is suspected, order appropriate imaging.
Patients who have deep infection usually exhibit frank, purulent discharge; fever; and severe pain. Marked separation of wound edges is often present as well, as is an elevated white blood cell count.
As with superficial infection, the key to therapy is liberal exploration, drainage of the abscess cavity, and mechanical wound debridement. Irrigate the wound copiously using a dilute mixture of saline and hydrogen peroxide to remove any remaining debris. Avoid povidone-iodine solution because it inhibits normal tissue granulation.
The wound may be left open to heal by secondary intention, or it may be closed secondarily after 3 to 6 days, provided there is no evidence of infection and a healthy granulating bed is present.
Consider adjuvant antibiotics, especially when the patient is immunocompromised.
If the wound has pronounced edema and unusual discoloration, consider a serious infection such as necrotizing fasciitis.
Wound dehiscence raises risk of evisceration
Dehiscence of the abdominal incision occurs when the various layers separate. Dehiscence may be extrafascial (superficial disruption of the skin and subcutaneous tissue only) or may involve all layers, including peritoneum (complete fascial dehiscence or burst abdomen).
When bowel or omentum extrudes, the term evisceration is appropriate.
In several reviews of the literature, the incidence of dehiscence ranged from 0.4% in earlier studies to 1% to 3% in later reviews.9-12 Despite advances in preoperative and postoperative care, suture materials, surgical technique, and antibiotics, fascial dehiscence remains a serious problem in abdominal surgery.
What causes wound disruption?
To a great extent, abdominal wound breakdown is a function of surgical technique and method of closure. Although the conventional wisdom is that dehiscence occurs less frequently with a transverse incision than with a vertical one, this assumption is being challenged. A small study by Hendrix and associates found no differences in the rate of dehiscence by type of incision.13 That finding suggests that the incidence of dehiscence is inversely related to the strength of closure.
Selection of the appropriate suture material also is important. In addition, use of electrocautery in the “cutting current” mode when the abdomen is opened causes less tissue injury than “coagulation current.” The latter has a greater thermal effect, thereby weakening the fascial layer.
Patient characteristics that influence wound integrity include comorbidities such as diabetes and malignancy, recent corticosteroid administration, and malnutrition.
Although infection may accompany superficial wound separation, its role in complete dehiscence is unclear.
Conditions that cause abdominal distention, such as severe coughing, vomiting, ileus, and ascites, may contribute to dehiscence, particularly when the closure method is less than satisfactory.
Some authors have found a greater incidence of wound disruption when multiple risk factors are present. In patients who had eight or more risk factors, wound disruption was universal.11,12
Management entails debridement, irrigation, and closure
When extrafascial dehiscence occurs, mechanical debridement and irrigation are usually the only measures necessary before deciding how to close the wound—even if infection is present. Remove all foreign material and excise any devitalized tissue.
As for the method of closure, the choice is usually between secondary closure and leaving the wound open to heal by secondary intention. An alternative to the latter is wound closure after several days, once a healthy granulating bed develops.
Dodson and colleagues described a technique of superficial wound closure that can be performed at the bedside using local anesthesia, with little discomfort to the patient.14 Wound separation caused by a small hematoma or sterile seroma especially lends itself to this type of immediate closure.
Vacuum-assisted closure
The vacuum-assisted wound closure system is a device that speeds healing and reduces the risk of complication. It consists of a sponge dressing that can be sized to fit an open wound and connected to an apparatus that generates negative pressure. The device enhances healing by removing excess fluid and debris and decreasing wound edema.
Argenta and associates reported successful use of this system to expedite healing in three cases of wound failure.16 It can be employed in the home-health setting by nurses trained in its use.
Human acellular dermal matrix
Occasionally, breakdown of a wound creates marked fascial defects that preclude secondary closure. Synthetic materials—both absorbable and nonabsorbable varieties—have been employed to bridge the defect, but their use sometimes leads to adhesions, infection, and cutaneous fistula. These risks are of special concern when the wound is already contaminated or otherwise compromised.
One alternative is human acellular dermal matrix (AlloDerm, LifeCell Corp). Tung and colleagues described its use for repair of a fascial defect in a previously irradiated cancer patient whose postoperative course was complicated by pelvic infection.17 This dermal matrix, a basement membrane taken from cadaveric skin, promotes neovascularization and is thought to be associated with a lower incidence of infection and adhesions than is traditional mesh. It is widely used in the burn setting and in the repair of ventral hernia, but is a relatively new addition to the management of fascial defects associated with wound breakdown.
Growth factors
Wound healing is regulated by a number of entities, including cytokines and growth factors, so it is no surprise that research has turned its focus on them. In a preliminary study, investigators found that separated abdominal wounds closed faster when recombinant human platelet-derived growth factor BB was topically administered than they did when they were left open to close by secondary intention.18
Although their use is not commonplace in wound management, research suggests that growth factors may one day be helpful adjuncts in the care of wound complications.
Complete fascial dehiscence is a “catastrophic” complication
Complete dehiscence of the fascia and extrusion of intra-abdominal contents is a serious catastrophic complication that is associated with a mortality rate of about 20%. It typically occurs between the third and seventh postoperative days, although later occurrences have been reported.
Warning signs of impending evisceration include serous drainage in the absence of obvious infection, and a “popping” sensation on the part of the patient—a feeling that something is “giving way.”
If evisceration occurs, cover exposed bowel with packs soaked in saline or povidone-iodine and prepare the patient for emergency surgery. Institute both hydration and broad-spectrum antibiotics.
Before replacing the abdominal contents, thoroughly irrigate the peritoneal cavity and inspect the bowel carefully, excising any necrotic tissue.
Reapproximate the fascia using interrupted #1 or #2 monofilament suture. Also consider placing retention sutures, particularly when the patient has multiple risk factors for wound complications (FIGURE). Leave the wound open, prepared for later closure.
If the abdomen cannot be closed because of peritonitis or bowel edema, or there is an insufficient amount of fascia remaining, approximate the abdominal wall using bridging sutures over a gauze pack as a temporizing measure until reconstruction can be performed. Consultation with a plastic surgeon or trauma specialist is recommended.
FIGURE Consider retention sutures for high-risk patients
Retention sutures are placed in interrupted fashion to support the primary suture line and are carried through the full thickness of the tissue, from the abdominal wall skin through the fascia and, if possible, the peritoneum. A rubber bolster placed across each suture keeps the suture from cutting into the skin (inset).
Necrotizing fasciitis: Worst of the worst
Necrotizing fasciitis is a dangerous, synergistic, bacterial infection involving the fascia, subcutaneous tissue, and skin. The culprits are multiple bacterial pathogens that include Streptococcus pyogenes, staphylococcal species, gram-negative aerobes, and anaerobes. The infection typically originates at a localized area, spreads along the fascial planes, and ultimately causes septic thrombosis of the vessels penetrating the skin and deeper layers. The result is necrosis. The associated mortality rate is approximately 20%.
The patient who has necrotizing fasciitis typically displays severe pain; anesthetic, edematous skin; purple, necrotic wound edges; hemorrhagic bullae; and crepitus.
Frank necrosis subsequently develops, with surrounding inflammation and edema, and leads to systemic toxicity, with fever, hemodynamic abnormality, and shock. In advanced stages, gangrene is present.
Laboratory evaluation includes a white blood cell count. Biopsy also is recommended. If necrotizing fasciitis is present, biopsy will reveal necrosis and thrombi of vessels passing through the fascia.
Treatment of necrotizing fasciitis requires intravenous, broad-spectrum antibiotics, including penicillin, that are adjusted according to the findings of the wound culture and sensitivity test. Cardiovascular and fluid-volume support is critical, as is wide surgical debridement of all necrosed skin and fascia. The latter, in fact, is the cornerstone of therapy.
The authors report no financial relationships relevant to this article.
Despite advances in medicine and surgery over the past century, postoperative wound complication remains a serious challenge. When a complication occurs, it translates into prolonged hospitalization, lost time from work, and greater cost to the patient and the health-care system.
Prevention of wound complication begins well before surgery. Requirements include:
- understanding of wound healing (see below) and the classification of wounds (TABLE 1)
- thorough assessment of the patient for risk factors for impaired wound healing, such as diabetes or use of corticosteroid medication (TABLE 2)
- antibiotic prophylaxis, if indicated (TABLE 3)
- good surgical technique, gentle tissue handling, and meticulous hemostasis
- placement of a drain, when appropriate
- awareness of technology that can enhance healing
- close monitoring in the postoperative period
- intervention at the first hint of abnormality.
In this article, we describe predisposing factors and preventive techniques and measures, and outline the most common wound complications, from seroma to dehiscence, including effective management strategies.
It was pioneering Scottish surgeon John Hunter who noted that “injury alone has in all cases the tendency to produce the disposition and means of a cure.”15
Unlike the tissue regeneration that occurs primarily in lower animals, human wound healing is mediated by collagen deposition, or scarring, which provides structural support to the wound. This scarring process may itself cause a variety of clinical problems.
Wound healing is characterized by overlapping, largely interdependent phases, with no clear demarcation between them. Failure in one phase may have a negative impact on the overall outcome.
In general, wound healing involves two phases: inflammation and proliferation. Within these phases, the following processes occur: scar maturation, wound contraction, and epithelialization. These repair mechanisms are activated in response to tissue injury even when it is surgically induced.
Inflammatory phase
The initial response to tissue injury is inflammation, which is mediated by various amines, enzymes, and other substances. This inflammation can be further broken down into vascular and cellular responses.
Inflammation triggers increased blood flow and migration of neutrophils, monocytes, macrophages, and other cells into the wound.
The first burst of blood acts to cleanse the wound of foreign debris. It is followed by vasoconstriction, which is mediated by thromboxane 2, to decrease blood loss. Vasodilation then occurs once histamine and serotonin are released, permitting increased blood flow to the wound. The surge in blood flow accounts for the increased warmth and redness of the wound. Vasodilation also increases capillary permeability, allowing the migration of red blood cells, platelets, leukocytes, plasma, and other tissue fluids into the interstitium of the wound. This migration accounts for wound edema.
In the cellular response, which is facilitated by increased blood flow, cell migration occurs as part of an immune response. Neutrophils, the first cells to enter the wound, engage in phagocytosis of bacteria and debris. Subsequently, there is migration of monocytes, macrophages, and other cells. This nonspecific immune response is sustained by prostaglandins, aided by complement factors and cytokines. A specific immune response follows, aimed at destroying specific antigens, and involves both B- and T-lymphocytes.
Proliferative phase
Proliferation is characterized by the infiltration of endothelial cells and fibroblasts and subsequent collagen deposition along a previously formed fibrin network. This new, highly vascularized tissue assumes a granular appearance—hence, the term “granulation tissue.”
Collagen that is deposited in the wound undergoes maturation and remodeling, increasing the tensile strength of the wound. The process continues for months after the initial insult.
All wounds undergo some degree of contraction, but the process is more relevant in wounds that remain open or involve significant tissue loss.
Last, the external covering of the wound is restored by epithelialization.
TABLE 1
Classification of surgical wounds
| CLASS I – Clean wounds (infection rate <5%) |
|
| CLASS II – Clean–contaminated wounds (infection rate 2–10%) |
|
| CLASS III – Contaminated wounds (infection rate 15–20%) |
|
| CLASS IV – Dirty or infected wounds (infection rate >30%) |
|
| SOURCE: Centers for Disease Control and Prevention and the American College of Surgeons |
TABLE 2
Risk factors for poor wound healing and dehiscence
| Poor wound healing |
|
| Abdominal wound dehiscence |
|
| SOURCE: Carlson,11 Cliby12. |
Conditions and drugs that impair healing
Preexisting medical conditions may limit healing, especially conditions associated with diminished delivery of oxygen and nutrients to healing tissues.
Diabetes can damage the vasculature and may impair healing if the blood glucose level is markedly elevated in the perioperative period. Such an elevation impedes transport of vitamin C, a key component of collagen synthesis.
Malignancy and immunosuppressive disorders may prevent optimal healing by compromising the immune response.
Bacterial vaginosis, a common polymicrobial infection involving aerobic and anaerobic bacteria, appears to be associated with postoperative febrile morbidity and surgical-site infection, particularly after hysterectomy.1 Current guidelines recommend that medical therapy for bacterial vaginosis be instituted at least 4 days before surgery and continued postoperatively.
Because steroids, NSAIDs, and chemotherapy agents impede wound healing, and anticoagulants may interfere with granulation, it is crucial to review the patient’s medications well in advance of surgery.
Nutrition plays a critical role
The importance of nutrition cannot be overstated. A significant percentage of patients are thought to have some degree of nutritional deficiency preoperatively. This deficiency may alter the inflammatory response, impair collagen synthesis, and reduce the tensile strength of the wound.
Because healing requires energy, deficits in carbohydrates may limit protein utilization, and deficiencies of vitamins and micronutrients can also interfere with healing.2
Obesity, too, increases the risk of postoperative wound complication. Markedly obese patients have a thick, avascular, subcutaneous layer of fat that compromises healing.3
Meticulous technique required
Good surgical technique and appropriate use of antibiotics are critical components of successful wound healing.
When placing the incision, avoid the moist, bacteria-laden subpannicular crease in the markedly obese.
During a procedure, handle tissue gently, keep it moist, and make minimal use of electrocautery to reduce tissue injury and promote healing. Keep operating time and blood loss to a minimum, and debride the wound of any foreign material and devitalized tissue.
Multiple studies have demonstrated that judicious use of prophylactic antibiotics significantly decreases the incidence of wound infection, particularly in relation to hysterectomy and vaginal procedures and when entry into bowel is anticipated.4,5 A number of prophylactic regimens are given in TABLE 3.
Meticulous hemostasis at the time of closure is imperative. When complete hemostasis cannot be confirmed, place a small drain in the subcutaneous space (or subfascial space, if there is oozing on the muscle bed) and apply a pressure dressing to help prevent hematoma. Although a drain is not a substitute for precise hemostasis or careful surgical technique, it may be helpful when there is concern about oozing or a “wet” surface, or when the patient is markedly obese.
Some practitioners have expressed concern over the risk of bacterial migration and infection with placement of a drain, but others, including us, advocate use of a drain in the subcutaneous space to help remove residual blood, fluid, and other debris to prevent the formation of dead space and infection and promote wound closure and healing. In a small study, Gallup and associates demonstrated a decreased incidence of wound breakdown when a drain was placed.6
A closed-suction drain, such as a Jackson-Pratt or Hemovac model, helps minimize wound complication when it is placed in the subcutaneous layer. (Avoid a rubber Penrose drain because it may allow bacteria to enter the wound.) It is imperative that the drain exit the body via a separate site and not through the incision itself. We advocate removal when less than 30 mL of fluid accumulates in the reservoir over 24 hours.
TABLE 3
3 prophylactic antibiotic regimens
| Procedure | Antibiotic | Single intravenous dose |
|---|---|---|
| Hysterectomy and urogynecologic procedures, including those that involve mesh | Cefazolin | 1 g or 2 g |
| Clindamycin plus gentamicin, a quinolone, or aztreonam | 600 mg plus 1.5 mg/kg, 400 mg, or 1 g, respectively | |
| Metronidazole plus gentamicin or a quinolone | 500 mg plus 1.5 mg/kg or 400 mg, respectively | |
| SOURCE: American College of Obstetricians and Gynecologists5 | ||
Fluid within the wound does not always indicate infection
Wound collections are not necessarily indicative of infection; collections of fluid within the wound may represent a serous transudate, blood, pus, or a combination of these. If the fluid is not addressed, however, fulminant infection may be the result.
Seroma is usually painless
A seroma is a collection of wound exudates within the dead space. Seroma typically involves thin, pink, watery discharge and minimal edge separation. In some cases, there may be surrounding edema but generally little to no tenderness.
When a seroma is detected, remove the staples or stitches in the area of concern and explore the wound. It is essential to ensure fascial integrity, as serous wound drainage may be a sign of impending evisceration. After these measures are taken, cleanse and lightly pack the wound to permit drainage.
Hematoma requires identification of the source of bleeding
Hematoma represents blood or a blood clot within the tissues beneath the skin. It may be caused by persistent bleeding of a vessel, although the pressure within the wound and the pressure produced by the dressing often provide tamponade on the bleeding source, in which case the hematoma forms with no active bleeding.
Hematoma is usually caused by small bleeding vessels that were not apparent at the time of surgery or were not cauterized or ligated at the time of closure. For this reason, it is important to achieve good hemostasis and a “dry” wound before closing the skin.
When hematoma is suspected, open the wound enough to permit adequate exposure and identify the source of bleeding. Evacuate as much blood and clot as possible because blood is an ideal medium for bacterial growth. If active bleeding is found, use a silver nitrate applicator or handheld cautery pen to accomplish hemostasis at bedside. If bleeding is more severe, or the source cannot be visualized, consider returning to the operating room for more extensive exploration.
Once hemostasis is achieved, irrigate the wound copiously and institute local wound care.
How common is infection?
Before it is possible to address this question, it is necessary to clarify the terminology of infection. Contamination and colonization are different entities. The first refers to the presence of bacteria without multiplication. The latter describes the multiplication of bacteria in the absence of a host response. When infection is present, bacterial proliferation produces clinical signs and symptoms.
Postoperative abdominal wound infection occurs in about 5% of cases but may be more common in procedures that involve a greater level of contamination.7 One study found a 12% incidence of wound infection, but the rate declined to 8% when antibiotic prophylaxis was instituted.4
Several other studies have examined determinants of infection. For example, a large Cochrane review found no real differences in infection rate by preoperative skin preparation technique or agent, but it did observe that one study had demonstrated the superiority of chlorhexidine to other cleansing agents.8
Cruse and Foord also noted the slight superiority of chlorhexidine, as well as the efficacy of clipping abdominal hair immediately before surgery.7
When identifying organisms, look for the usual suspects
The offending pathogens in infection are usually endogenous flora found on the patient’s skin and within hollow organs (vagina, bowel). The organisms most commonly responsible for infection are Staphylococcus (aureus, epidermidis), enterococci, and Escherichia coli. However, the bacteria identified in the wound may not be the causative organism.
Most infections typically become clinically apparent between the fifth and 10th postoperative days, often after the patient has been discharged, although they may appear much earlier or much later. One of us (Dr. Perkins) had a patient who presented with a suppurative infection after undergoing hysterectomy for endometrial carcinoma 5 months earlier.
Cellulitis is common
Wound cellulitis, a common, nonsuppurative infection of skin and underlying connective tissue, is generally not severe. The wound assumes a brawny, reddish brown appearance associated with edema, warmth, and erythema. Fever is not always present.
It is important to remember that cellulitis may surround a deeper infection. Although needle aspiration of the leading edge has been advocated, it yields a positive culture in only 20% to 40% of cases.
In the absence of purulent drainage, treat cellulitis with antibiotics, utilizing sulfamethoxazole-trimethroprim, a cephalosporin, or augmented penicillin, and apply warm packs to the wound.
If purulent drainage is seen, or the patient fails to improve significantly within 24 hours, suspect an abscess or resistant organism.
- During preoperative evaluation, assess the patient for risk factors, comorbid conditions, and medications that can impair healing
- If the patient is morbidly obese and planning to undergo an elective, nonurgent procedure, consider instituting a plan for preoperative weight loss
- Advise smokers to “kick the habit” 1 or 2 months before surgery
- Avoid using electrocautery in the “coagulation current” setting when incising the fascia
- When approximating the fascia, take wide bites of tissue (1.5 to 2 cm from the edge)
- Avoid excessive suture tension when closing the fascial layer (“Approximate, don’t strangulate”)
- Obtain good hemostasis before closing the wound; consider placing a drain in an obese patient
- In a high-risk patient who has multiple risk factors, consider retention sutures
- Minimize or avoid abdominal distention during the postoperative period with:
- Assess the wound for infection early, and treat infection promptly
- Remember to administer prophylactic antibiotics
Most wound infections are superficial
Approximately 75% of all wound infections involve the skin and subcutaneous tissue layers. Superficial infection is more likely to occur when there is an undrained hematoma, excessively tight sutures, tissue trauma, or a retained foreign material. Edema, erythema, and pain and tenderness may be more pronounced than with cellulitis. A low-grade fever may be present, and incisional discharge typically occurs.
Drainage is the cornerstone of management and requires the removal of staples or sutures from the area. Local exploration is mandatory, and fascial integrity must be confirmed. If a pocket of pus is found, open the wound liberally to determine the extent of the pocket and permit as much evacuation as possible. Wound culture is optional. Institute local wound care and consider adjuvant antibiotics in selected cases.
Ensure fascial integrity
Any infection that arises immediately adjacent to the fascia may have an intra-abdominal component, although that is unlikely. Extensive exploration is warranted to assess fascial integrity.
If intra-abdominal infection is suspected, order appropriate imaging.
Patients who have deep infection usually exhibit frank, purulent discharge; fever; and severe pain. Marked separation of wound edges is often present as well, as is an elevated white blood cell count.
As with superficial infection, the key to therapy is liberal exploration, drainage of the abscess cavity, and mechanical wound debridement. Irrigate the wound copiously using a dilute mixture of saline and hydrogen peroxide to remove any remaining debris. Avoid povidone-iodine solution because it inhibits normal tissue granulation.
The wound may be left open to heal by secondary intention, or it may be closed secondarily after 3 to 6 days, provided there is no evidence of infection and a healthy granulating bed is present.
Consider adjuvant antibiotics, especially when the patient is immunocompromised.
If the wound has pronounced edema and unusual discoloration, consider a serious infection such as necrotizing fasciitis.
Wound dehiscence raises risk of evisceration
Dehiscence of the abdominal incision occurs when the various layers separate. Dehiscence may be extrafascial (superficial disruption of the skin and subcutaneous tissue only) or may involve all layers, including peritoneum (complete fascial dehiscence or burst abdomen).
When bowel or omentum extrudes, the term evisceration is appropriate.
In several reviews of the literature, the incidence of dehiscence ranged from 0.4% in earlier studies to 1% to 3% in later reviews.9-12 Despite advances in preoperative and postoperative care, suture materials, surgical technique, and antibiotics, fascial dehiscence remains a serious problem in abdominal surgery.
What causes wound disruption?
To a great extent, abdominal wound breakdown is a function of surgical technique and method of closure. Although the conventional wisdom is that dehiscence occurs less frequently with a transverse incision than with a vertical one, this assumption is being challenged. A small study by Hendrix and associates found no differences in the rate of dehiscence by type of incision.13 That finding suggests that the incidence of dehiscence is inversely related to the strength of closure.
Selection of the appropriate suture material also is important. In addition, use of electrocautery in the “cutting current” mode when the abdomen is opened causes less tissue injury than “coagulation current.” The latter has a greater thermal effect, thereby weakening the fascial layer.
Patient characteristics that influence wound integrity include comorbidities such as diabetes and malignancy, recent corticosteroid administration, and malnutrition.
Although infection may accompany superficial wound separation, its role in complete dehiscence is unclear.
Conditions that cause abdominal distention, such as severe coughing, vomiting, ileus, and ascites, may contribute to dehiscence, particularly when the closure method is less than satisfactory.
Some authors have found a greater incidence of wound disruption when multiple risk factors are present. In patients who had eight or more risk factors, wound disruption was universal.11,12
Management entails debridement, irrigation, and closure
When extrafascial dehiscence occurs, mechanical debridement and irrigation are usually the only measures necessary before deciding how to close the wound—even if infection is present. Remove all foreign material and excise any devitalized tissue.
As for the method of closure, the choice is usually between secondary closure and leaving the wound open to heal by secondary intention. An alternative to the latter is wound closure after several days, once a healthy granulating bed develops.
Dodson and colleagues described a technique of superficial wound closure that can be performed at the bedside using local anesthesia, with little discomfort to the patient.14 Wound separation caused by a small hematoma or sterile seroma especially lends itself to this type of immediate closure.
Vacuum-assisted closure
The vacuum-assisted wound closure system is a device that speeds healing and reduces the risk of complication. It consists of a sponge dressing that can be sized to fit an open wound and connected to an apparatus that generates negative pressure. The device enhances healing by removing excess fluid and debris and decreasing wound edema.
Argenta and associates reported successful use of this system to expedite healing in three cases of wound failure.16 It can be employed in the home-health setting by nurses trained in its use.
Human acellular dermal matrix
Occasionally, breakdown of a wound creates marked fascial defects that preclude secondary closure. Synthetic materials—both absorbable and nonabsorbable varieties—have been employed to bridge the defect, but their use sometimes leads to adhesions, infection, and cutaneous fistula. These risks are of special concern when the wound is already contaminated or otherwise compromised.
One alternative is human acellular dermal matrix (AlloDerm, LifeCell Corp). Tung and colleagues described its use for repair of a fascial defect in a previously irradiated cancer patient whose postoperative course was complicated by pelvic infection.17 This dermal matrix, a basement membrane taken from cadaveric skin, promotes neovascularization and is thought to be associated with a lower incidence of infection and adhesions than is traditional mesh. It is widely used in the burn setting and in the repair of ventral hernia, but is a relatively new addition to the management of fascial defects associated with wound breakdown.
Growth factors
Wound healing is regulated by a number of entities, including cytokines and growth factors, so it is no surprise that research has turned its focus on them. In a preliminary study, investigators found that separated abdominal wounds closed faster when recombinant human platelet-derived growth factor BB was topically administered than they did when they were left open to close by secondary intention.18
Although their use is not commonplace in wound management, research suggests that growth factors may one day be helpful adjuncts in the care of wound complications.
Complete fascial dehiscence is a “catastrophic” complication
Complete dehiscence of the fascia and extrusion of intra-abdominal contents is a serious catastrophic complication that is associated with a mortality rate of about 20%. It typically occurs between the third and seventh postoperative days, although later occurrences have been reported.
Warning signs of impending evisceration include serous drainage in the absence of obvious infection, and a “popping” sensation on the part of the patient—a feeling that something is “giving way.”
If evisceration occurs, cover exposed bowel with packs soaked in saline or povidone-iodine and prepare the patient for emergency surgery. Institute both hydration and broad-spectrum antibiotics.
Before replacing the abdominal contents, thoroughly irrigate the peritoneal cavity and inspect the bowel carefully, excising any necrotic tissue.
Reapproximate the fascia using interrupted #1 or #2 monofilament suture. Also consider placing retention sutures, particularly when the patient has multiple risk factors for wound complications (FIGURE). Leave the wound open, prepared for later closure.
If the abdomen cannot be closed because of peritonitis or bowel edema, or there is an insufficient amount of fascia remaining, approximate the abdominal wall using bridging sutures over a gauze pack as a temporizing measure until reconstruction can be performed. Consultation with a plastic surgeon or trauma specialist is recommended.
FIGURE Consider retention sutures for high-risk patients
Retention sutures are placed in interrupted fashion to support the primary suture line and are carried through the full thickness of the tissue, from the abdominal wall skin through the fascia and, if possible, the peritoneum. A rubber bolster placed across each suture keeps the suture from cutting into the skin (inset).
Necrotizing fasciitis: Worst of the worst
Necrotizing fasciitis is a dangerous, synergistic, bacterial infection involving the fascia, subcutaneous tissue, and skin. The culprits are multiple bacterial pathogens that include Streptococcus pyogenes, staphylococcal species, gram-negative aerobes, and anaerobes. The infection typically originates at a localized area, spreads along the fascial planes, and ultimately causes septic thrombosis of the vessels penetrating the skin and deeper layers. The result is necrosis. The associated mortality rate is approximately 20%.
The patient who has necrotizing fasciitis typically displays severe pain; anesthetic, edematous skin; purple, necrotic wound edges; hemorrhagic bullae; and crepitus.
Frank necrosis subsequently develops, with surrounding inflammation and edema, and leads to systemic toxicity, with fever, hemodynamic abnormality, and shock. In advanced stages, gangrene is present.
Laboratory evaluation includes a white blood cell count. Biopsy also is recommended. If necrotizing fasciitis is present, biopsy will reveal necrosis and thrombi of vessels passing through the fascia.
Treatment of necrotizing fasciitis requires intravenous, broad-spectrum antibiotics, including penicillin, that are adjusted according to the findings of the wound culture and sensitivity test. Cardiovascular and fluid-volume support is critical, as is wide surgical debridement of all necrosed skin and fascia. The latter, in fact, is the cornerstone of therapy.
1. Lin L, Song J, Kimber N, et al. The role of bacterial vaginosis in infection after major gynecologic surgery. Infect Dis Obstet Gynecol. 1999;7:169-174.
2. Williams JZ, Barbul A. Nutrition and wound healing. Surg Clin North Am. 2003;83:571-596.
3. Perkins JD, Jackson RA. Risks and remedies when your surgical patient is obese. OBG Management. 2007;19(10)34-54.
4. Kamat AA, Brancazio L, Gibson M. Wound infection in gynecologic surgery. Infect Dis Obstet Gynecol. 2000;8:230-234.
5. American College of Obstetricians and Gynecologists. Antibiotic prophylaxis for gynecologic procedures. ACOG Practice Bulletin #104. Washington, DC: ACOG; 2009.
6. Gallup DC, Gallup DG, Nolan TE, Smith RP, Messing MF, Kline KL. Use of a subcutaneous closed drainage system and antibiotics in obese gynecologic patients. Am J Obstet Gynecol. 1996;175:358-362.
7. Cruse PJE, Foord R. The epidemiology of wound infection. A 10-year prospective study of 62,639 wounds. Surg Clin North Am. 1980;60:27-40.
8. Edwards PS, Lipp A, Holmes A. Preoperative skin antiseptics for preventing surgical wound infections after clean surgery. Cochrane Database Syst Rev. 2004;(3):CD003949.-
9. Baggish MS, Lee WK. Abdominal wound disruption. Obstet Gynecol. 1975;46:530-534.
10. Poole GV, Jr. Mechanical factors in abdominal wound closure: the prevention of fascial dehiscence. Surgery. 1985;97:631-640.
11. Carlson MA. Acute wound failure. Surg Clin North Am. 1997;77:607-636.
12. Cliby WA. Abdominal incision wound breakdown. Clin Obstet Gynecol. 2002;45:507-517.
13. Hendrix SL, Schimp V, Martin J, Singh A, Kruger M, McNeeley SG. The legendary superior strength of the Pfannenstiel incision: a myth? Am J Obstet Gynecol. 2000;182:1446-1451.
14. Dodson MK, Magann EF, Sullivan DL, Meeks GR. Extrafascial wound dehiscence: deep en bloc closure versus superficial skin closure. Obstet Gynecol. 1994;83:142-145.
15. Brunicardi FC, Andersen DK, Billiar TR, Dunn DL, Hunter JG, Pollock RE. Chapter 8: Wound healing. In: Schwartz’s Principles of Surgery. 8th ed. New York: McGraw-Hill; 2005.
16. Argenta PA, Rahaman J, Gretz HF, 3rd, Nezhat F, Cohen CJ. Vacuum-assisted closure in the treatment of complex gynecologic wound failures. Obstet Gynecol. 2002;99:497-501.
17. Tung CS, Zighelboim I, Scott B, Anderson ML. Human acellular dermal matrix for closure of a contaminated gynecologic wound. Gynecol Oncol. 2006;103:354-356.
18. Shackelford DP, Fackler E, Hoffman MK, Atkinson S. Use of topical recombinant human platelet-derived growth factor BB in abdominal wound separation. Am J Obstet Gynecol. 2002;186:701-704.
1. Lin L, Song J, Kimber N, et al. The role of bacterial vaginosis in infection after major gynecologic surgery. Infect Dis Obstet Gynecol. 1999;7:169-174.
2. Williams JZ, Barbul A. Nutrition and wound healing. Surg Clin North Am. 2003;83:571-596.
3. Perkins JD, Jackson RA. Risks and remedies when your surgical patient is obese. OBG Management. 2007;19(10)34-54.
4. Kamat AA, Brancazio L, Gibson M. Wound infection in gynecologic surgery. Infect Dis Obstet Gynecol. 2000;8:230-234.
5. American College of Obstetricians and Gynecologists. Antibiotic prophylaxis for gynecologic procedures. ACOG Practice Bulletin #104. Washington, DC: ACOG; 2009.
6. Gallup DC, Gallup DG, Nolan TE, Smith RP, Messing MF, Kline KL. Use of a subcutaneous closed drainage system and antibiotics in obese gynecologic patients. Am J Obstet Gynecol. 1996;175:358-362.
7. Cruse PJE, Foord R. The epidemiology of wound infection. A 10-year prospective study of 62,639 wounds. Surg Clin North Am. 1980;60:27-40.
8. Edwards PS, Lipp A, Holmes A. Preoperative skin antiseptics for preventing surgical wound infections after clean surgery. Cochrane Database Syst Rev. 2004;(3):CD003949.-
9. Baggish MS, Lee WK. Abdominal wound disruption. Obstet Gynecol. 1975;46:530-534.
10. Poole GV, Jr. Mechanical factors in abdominal wound closure: the prevention of fascial dehiscence. Surgery. 1985;97:631-640.
11. Carlson MA. Acute wound failure. Surg Clin North Am. 1997;77:607-636.
12. Cliby WA. Abdominal incision wound breakdown. Clin Obstet Gynecol. 2002;45:507-517.
13. Hendrix SL, Schimp V, Martin J, Singh A, Kruger M, McNeeley SG. The legendary superior strength of the Pfannenstiel incision: a myth? Am J Obstet Gynecol. 2000;182:1446-1451.
14. Dodson MK, Magann EF, Sullivan DL, Meeks GR. Extrafascial wound dehiscence: deep en bloc closure versus superficial skin closure. Obstet Gynecol. 1994;83:142-145.
15. Brunicardi FC, Andersen DK, Billiar TR, Dunn DL, Hunter JG, Pollock RE. Chapter 8: Wound healing. In: Schwartz’s Principles of Surgery. 8th ed. New York: McGraw-Hill; 2005.
16. Argenta PA, Rahaman J, Gretz HF, 3rd, Nezhat F, Cohen CJ. Vacuum-assisted closure in the treatment of complex gynecologic wound failures. Obstet Gynecol. 2002;99:497-501.
17. Tung CS, Zighelboim I, Scott B, Anderson ML. Human acellular dermal matrix for closure of a contaminated gynecologic wound. Gynecol Oncol. 2006;103:354-356.
18. Shackelford DP, Fackler E, Hoffman MK, Atkinson S. Use of topical recombinant human platelet-derived growth factor BB in abdominal wound separation. Am J Obstet Gynecol. 2002;186:701-704.