User login
Plantar Fibromatosis
Barbed suture, now in the toolbox of minimally invasive gyn surgery
The authors report no financial relationships relevant to this article.
Barbed suture is a relatively new technology that has the potential to greatly facilitate laparoscopic suturing. Two barbed sutures, each a different type, are available, or soon will be:
- the Quill bidirectional barbed suture (Angiotech Pharmaceuticals) was approved by the US Food and Drug Administration (FDA) in 2004 for soft-tissue approximation and has been sold in the United States since 2007 ( FIGURE 1A )1
- the V-Loc 180 unidirectional barbed suture (Covidien) won FDA approval in March for soft-tissue approximation. It should be available in October ( FIGURE 1B ).
(Note: We have performed laparoscopic surgery using the Quill suture, but not the V-Loc 180, both as part of clinical research and in practice.)
In the bidirectional suture, barbs are cut into suture in a direction opposite to the needle; the barbs change direction at the midpoint of the suture, and needles are swaged onto both ends ( FIGURE 1A ).2
The anchoring of bidirectional barbed suture resists migration and can be conceptualized as a “continuous interrupted” suture without knots; it has been shown to have tissue-holding performance that is at least equal to knot-anchored suture.3,4
A suture with bidirectional barbs offers several advantages:
- Because it self-anchors and is balanced by the countervailing barbs, no knots are required
- It self-anchors every 1 mm of tissue, yielding more consistent wound opposition; this may result in a more “watertight” seal
- Because it is knotless, it can securely reapproximate tissues in less time, at less cost, and with less aggravation.5,6
Note: Whether these characteristics apply to unidirectional barbed suture remains to be determined.
FIGURE 1 Two types of barbed suture
Bidirectional suture, in which barbs are cut into suture and change direction at the midpoint.
Unidirectional suture has a loop at the distal end to facilitate initial suture fastening.
Specifications
Because the effective diameter of suture is decreased when barbs are cut into it, barbed suture is rated one suture size smaller than smooth suture. A 0 barbed suture, for example, equals a 2-0 smooth suture.
In the current version of the Quill suture, there is a segment of approximately 7 cm of smooth suture proximal to each needle. A second-generation Quill will be released this year, with barbs extending all the way to the needles. Bidirectional barbed suture materials currently include polydioxanone (PDO), poliglecaprone 25 (Monoderm), nylon, and polypropylene.
The V-Loc 180 is a unidirectional barbed polyglyconate (Maxon) suture with a loop at the distal end to facilitate initial fastening.
We have performed more than 200 laparoscopic surgeries using Quill suture since March 2008; most involved total laparoscopic hysterectomy and laparoscopic myomectomy. We have also used Quill suture for laparoscopic sacrocolpopexy and uterosacral ligament suspension.
We have had outstanding clinical results: no cuff evisceration and only minor cases of vaginal bleeding or spotting in the immediate postoperative period.7 In one case, reoperation was necessary after laparoscopic myomectomy, but the repeat surgery was performed to treat small-bowel obstruction that was not directly related to the hysterotomy repair.
Technique
For vaginal-cuff closure, we use 0 PDO suture on a 36-mm half-circle taper-point needle. We initially used half of a 14×14-cm suture with a LapraTy clip on the distal end. We began the repair at the distal end of the vaginal cuff, taking care to incorporate the uterosacral ligament into the initial bite, and continuing proximally until the other uterosacral ligament was incorporated into the repair.
More recently, we have been using the 7×7-cm 0 PDO because it is now available with the 36-mm half-circle taper-point needle. Regardless of suture material, it is important to obtain a full-thickness bite, with a 1-cm margin on the vaginal mucosa on each bite.
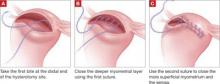
With the shorter suture, we begin closure in the middle of the cuff and take each needle to the opposite end of the cuff ( FIGURE 2 ). We incorporate the uterosacral ligament into the final bite on each side, and we cut the suture without a knot or a LapraTy clip.
FIGURE 2 Closing the vaginal cuff with bidirectional barbed suture For myomectomy closure, we use 14×14-cm 0 PDO suture on a 36-mm half-circle taper-point needle. If the hysterotomy is longer than 8 cm, we prefer to use 24×24-cm suture. We tack the first needle to the opposite anterior abdominal wall to avoid having the suture become entangled. We close the deepest layer using the first needle; we use the second needle to close the more superficial layer and the serosa, if possible ( FIGURE 3 ). If the suture is used beyond the barbed portion, we cut the needles and apply a LapraTy clip. Sometimes, three or four layers are needed to close a deep myometrial defect. 2/0 Monoderm can also be used for the serosa, continuously or as a baseball stitch.
FIGURE 3 Myomectomy closure with bidirectional barbed suture
1. Department of Health and Human Services letter approving Section 510(k) premarket notification for Quill suture. Available at http://www.accessdata.fda.gov/cdrh_docs/pdf4/k042075.pdf. Accessed August 13, 2009.
2. Leung JC. Barbed suture technology: recent advances. In: Medical Textiles 2004, Conference Proceedings; Oct. 26–27, 2004; pp. 62–80; Pittsburgh.
3. Rashid RM, Sartori M, White LE, Villa MT, Yoo SS, Alam M. Breaking strength of barbed polypropylene sutures: rater-blinded, controlled comparison with nonbarbed sutures of various calibers. Arch Dermatol. 2007;143:869-872.
4. Rodeheaver GT, Piñeros-Fernandez A, Salopek LS, et al. Barbed sutures for wound closure: in vivo wound security, tissue compatibility and cosmesis measurements. In: Society for Biomaterials 30th Annual Meeting Transactions; 2004; transaction 229, p. 232.
5. Greenberg JA, Einarsson JI. Use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy. J Minim Invasive Gynecol. 2008;15:621-623.
6. Moran ME, Marsh C, Perrotti M. Bidirectional-barbed sutured knotless running anastomosis v classic Van Velthoven suturing in a model system. J Endourol. 2007;21:1175-1178.
7. Einarsson JI, Vellinga T, Twijnstra A, Suzuki Y, Greenberg JA. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy; an evaluation of safety and clinical outcomes. Submitted as an abstract to the 38th Global Congress in Minimally Invasive Gynecology, 2009.
The authors report no financial relationships relevant to this article.
Barbed suture is a relatively new technology that has the potential to greatly facilitate laparoscopic suturing. Two barbed sutures, each a different type, are available, or soon will be:
- the Quill bidirectional barbed suture (Angiotech Pharmaceuticals) was approved by the US Food and Drug Administration (FDA) in 2004 for soft-tissue approximation and has been sold in the United States since 2007 ( FIGURE 1A )1
- the V-Loc 180 unidirectional barbed suture (Covidien) won FDA approval in March for soft-tissue approximation. It should be available in October ( FIGURE 1B ).
(Note: We have performed laparoscopic surgery using the Quill suture, but not the V-Loc 180, both as part of clinical research and in practice.)
In the bidirectional suture, barbs are cut into suture in a direction opposite to the needle; the barbs change direction at the midpoint of the suture, and needles are swaged onto both ends ( FIGURE 1A ).2
The anchoring of bidirectional barbed suture resists migration and can be conceptualized as a “continuous interrupted” suture without knots; it has been shown to have tissue-holding performance that is at least equal to knot-anchored suture.3,4
A suture with bidirectional barbs offers several advantages:
- Because it self-anchors and is balanced by the countervailing barbs, no knots are required
- It self-anchors every 1 mm of tissue, yielding more consistent wound opposition; this may result in a more “watertight” seal
- Because it is knotless, it can securely reapproximate tissues in less time, at less cost, and with less aggravation.5,6
Note: Whether these characteristics apply to unidirectional barbed suture remains to be determined.
FIGURE 1 Two types of barbed suture
Bidirectional suture, in which barbs are cut into suture and change direction at the midpoint.
Unidirectional suture has a loop at the distal end to facilitate initial suture fastening.
Specifications
Because the effective diameter of suture is decreased when barbs are cut into it, barbed suture is rated one suture size smaller than smooth suture. A 0 barbed suture, for example, equals a 2-0 smooth suture.
In the current version of the Quill suture, there is a segment of approximately 7 cm of smooth suture proximal to each needle. A second-generation Quill will be released this year, with barbs extending all the way to the needles. Bidirectional barbed suture materials currently include polydioxanone (PDO), poliglecaprone 25 (Monoderm), nylon, and polypropylene.
The V-Loc 180 is a unidirectional barbed polyglyconate (Maxon) suture with a loop at the distal end to facilitate initial fastening.
We have performed more than 200 laparoscopic surgeries using Quill suture since March 2008; most involved total laparoscopic hysterectomy and laparoscopic myomectomy. We have also used Quill suture for laparoscopic sacrocolpopexy and uterosacral ligament suspension.
We have had outstanding clinical results: no cuff evisceration and only minor cases of vaginal bleeding or spotting in the immediate postoperative period.7 In one case, reoperation was necessary after laparoscopic myomectomy, but the repeat surgery was performed to treat small-bowel obstruction that was not directly related to the hysterotomy repair.
Technique
For vaginal-cuff closure, we use 0 PDO suture on a 36-mm half-circle taper-point needle. We initially used half of a 14×14-cm suture with a LapraTy clip on the distal end. We began the repair at the distal end of the vaginal cuff, taking care to incorporate the uterosacral ligament into the initial bite, and continuing proximally until the other uterosacral ligament was incorporated into the repair.
More recently, we have been using the 7×7-cm 0 PDO because it is now available with the 36-mm half-circle taper-point needle. Regardless of suture material, it is important to obtain a full-thickness bite, with a 1-cm margin on the vaginal mucosa on each bite.
With the shorter suture, we begin closure in the middle of the cuff and take each needle to the opposite end of the cuff ( FIGURE 2 ). We incorporate the uterosacral ligament into the final bite on each side, and we cut the suture without a knot or a LapraTy clip.
FIGURE 2 Closing the vaginal cuff with bidirectional barbed suture For myomectomy closure, we use 14×14-cm 0 PDO suture on a 36-mm half-circle taper-point needle. If the hysterotomy is longer than 8 cm, we prefer to use 24×24-cm suture. We tack the first needle to the opposite anterior abdominal wall to avoid having the suture become entangled. We close the deepest layer using the first needle; we use the second needle to close the more superficial layer and the serosa, if possible ( FIGURE 3 ). If the suture is used beyond the barbed portion, we cut the needles and apply a LapraTy clip. Sometimes, three or four layers are needed to close a deep myometrial defect. 2/0 Monoderm can also be used for the serosa, continuously or as a baseball stitch.
FIGURE 3 Myomectomy closure with bidirectional barbed suture
The authors report no financial relationships relevant to this article.
Barbed suture is a relatively new technology that has the potential to greatly facilitate laparoscopic suturing. Two barbed sutures, each a different type, are available, or soon will be:
- the Quill bidirectional barbed suture (Angiotech Pharmaceuticals) was approved by the US Food and Drug Administration (FDA) in 2004 for soft-tissue approximation and has been sold in the United States since 2007 ( FIGURE 1A )1
- the V-Loc 180 unidirectional barbed suture (Covidien) won FDA approval in March for soft-tissue approximation. It should be available in October ( FIGURE 1B ).
(Note: We have performed laparoscopic surgery using the Quill suture, but not the V-Loc 180, both as part of clinical research and in practice.)
In the bidirectional suture, barbs are cut into suture in a direction opposite to the needle; the barbs change direction at the midpoint of the suture, and needles are swaged onto both ends ( FIGURE 1A ).2
The anchoring of bidirectional barbed suture resists migration and can be conceptualized as a “continuous interrupted” suture without knots; it has been shown to have tissue-holding performance that is at least equal to knot-anchored suture.3,4
A suture with bidirectional barbs offers several advantages:
- Because it self-anchors and is balanced by the countervailing barbs, no knots are required
- It self-anchors every 1 mm of tissue, yielding more consistent wound opposition; this may result in a more “watertight” seal
- Because it is knotless, it can securely reapproximate tissues in less time, at less cost, and with less aggravation.5,6
Note: Whether these characteristics apply to unidirectional barbed suture remains to be determined.
FIGURE 1 Two types of barbed suture
Bidirectional suture, in which barbs are cut into suture and change direction at the midpoint.
Unidirectional suture has a loop at the distal end to facilitate initial suture fastening.
Specifications
Because the effective diameter of suture is decreased when barbs are cut into it, barbed suture is rated one suture size smaller than smooth suture. A 0 barbed suture, for example, equals a 2-0 smooth suture.
In the current version of the Quill suture, there is a segment of approximately 7 cm of smooth suture proximal to each needle. A second-generation Quill will be released this year, with barbs extending all the way to the needles. Bidirectional barbed suture materials currently include polydioxanone (PDO), poliglecaprone 25 (Monoderm), nylon, and polypropylene.
The V-Loc 180 is a unidirectional barbed polyglyconate (Maxon) suture with a loop at the distal end to facilitate initial fastening.
We have performed more than 200 laparoscopic surgeries using Quill suture since March 2008; most involved total laparoscopic hysterectomy and laparoscopic myomectomy. We have also used Quill suture for laparoscopic sacrocolpopexy and uterosacral ligament suspension.
We have had outstanding clinical results: no cuff evisceration and only minor cases of vaginal bleeding or spotting in the immediate postoperative period.7 In one case, reoperation was necessary after laparoscopic myomectomy, but the repeat surgery was performed to treat small-bowel obstruction that was not directly related to the hysterotomy repair.
Technique
For vaginal-cuff closure, we use 0 PDO suture on a 36-mm half-circle taper-point needle. We initially used half of a 14×14-cm suture with a LapraTy clip on the distal end. We began the repair at the distal end of the vaginal cuff, taking care to incorporate the uterosacral ligament into the initial bite, and continuing proximally until the other uterosacral ligament was incorporated into the repair.
More recently, we have been using the 7×7-cm 0 PDO because it is now available with the 36-mm half-circle taper-point needle. Regardless of suture material, it is important to obtain a full-thickness bite, with a 1-cm margin on the vaginal mucosa on each bite.
With the shorter suture, we begin closure in the middle of the cuff and take each needle to the opposite end of the cuff ( FIGURE 2 ). We incorporate the uterosacral ligament into the final bite on each side, and we cut the suture without a knot or a LapraTy clip.
FIGURE 2 Closing the vaginal cuff with bidirectional barbed suture For myomectomy closure, we use 14×14-cm 0 PDO suture on a 36-mm half-circle taper-point needle. If the hysterotomy is longer than 8 cm, we prefer to use 24×24-cm suture. We tack the first needle to the opposite anterior abdominal wall to avoid having the suture become entangled. We close the deepest layer using the first needle; we use the second needle to close the more superficial layer and the serosa, if possible ( FIGURE 3 ). If the suture is used beyond the barbed portion, we cut the needles and apply a LapraTy clip. Sometimes, three or four layers are needed to close a deep myometrial defect. 2/0 Monoderm can also be used for the serosa, continuously or as a baseball stitch.
FIGURE 3 Myomectomy closure with bidirectional barbed suture
1. Department of Health and Human Services letter approving Section 510(k) premarket notification for Quill suture. Available at http://www.accessdata.fda.gov/cdrh_docs/pdf4/k042075.pdf. Accessed August 13, 2009.
2. Leung JC. Barbed suture technology: recent advances. In: Medical Textiles 2004, Conference Proceedings; Oct. 26–27, 2004; pp. 62–80; Pittsburgh.
3. Rashid RM, Sartori M, White LE, Villa MT, Yoo SS, Alam M. Breaking strength of barbed polypropylene sutures: rater-blinded, controlled comparison with nonbarbed sutures of various calibers. Arch Dermatol. 2007;143:869-872.
4. Rodeheaver GT, Piñeros-Fernandez A, Salopek LS, et al. Barbed sutures for wound closure: in vivo wound security, tissue compatibility and cosmesis measurements. In: Society for Biomaterials 30th Annual Meeting Transactions; 2004; transaction 229, p. 232.
5. Greenberg JA, Einarsson JI. Use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy. J Minim Invasive Gynecol. 2008;15:621-623.
6. Moran ME, Marsh C, Perrotti M. Bidirectional-barbed sutured knotless running anastomosis v classic Van Velthoven suturing in a model system. J Endourol. 2007;21:1175-1178.
7. Einarsson JI, Vellinga T, Twijnstra A, Suzuki Y, Greenberg JA. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy; an evaluation of safety and clinical outcomes. Submitted as an abstract to the 38th Global Congress in Minimally Invasive Gynecology, 2009.
1. Department of Health and Human Services letter approving Section 510(k) premarket notification for Quill suture. Available at http://www.accessdata.fda.gov/cdrh_docs/pdf4/k042075.pdf. Accessed August 13, 2009.
2. Leung JC. Barbed suture technology: recent advances. In: Medical Textiles 2004, Conference Proceedings; Oct. 26–27, 2004; pp. 62–80; Pittsburgh.
3. Rashid RM, Sartori M, White LE, Villa MT, Yoo SS, Alam M. Breaking strength of barbed polypropylene sutures: rater-blinded, controlled comparison with nonbarbed sutures of various calibers. Arch Dermatol. 2007;143:869-872.
4. Rodeheaver GT, Piñeros-Fernandez A, Salopek LS, et al. Barbed sutures for wound closure: in vivo wound security, tissue compatibility and cosmesis measurements. In: Society for Biomaterials 30th Annual Meeting Transactions; 2004; transaction 229, p. 232.
5. Greenberg JA, Einarsson JI. Use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy. J Minim Invasive Gynecol. 2008;15:621-623.
6. Moran ME, Marsh C, Perrotti M. Bidirectional-barbed sutured knotless running anastomosis v classic Van Velthoven suturing in a model system. J Endourol. 2007;21:1175-1178.
7. Einarsson JI, Vellinga T, Twijnstra A, Suzuki Y, Greenberg JA. The use of bidirectional barbed suture in laparoscopic myomectomy and total laparoscopic hysterectomy; an evaluation of safety and clinical outcomes. Submitted as an abstract to the 38th Global Congress in Minimally Invasive Gynecology, 2009.
What’s the best approach to managing chronic pain?
The authors report no financial relationships relevant to this article.
This article is adapted from the December 2008 installment of The Journal of Family Practice’s “Guideline Update” series. The Journal of Family Practice is an NLM-indexed publication of Quadrant HealthCom Inc., publisher of OBG Management.
- What are the critical steps in the assessment of a patient who suffers chronic pain?
- What are the four biologic mechanisms of pain?
- When is referral to a pain specialist recommended?
Answers to these questions are summarized below, and in the 2008 edition of Assessment and Management of Chronic Pain, a practice guideline developed and first published in 2005 by the Institute for Clinical Systems Improvement (ICSI), which also funded the work. ICSI is a collaboration of 57 medical groups sponsored by six Minnesota health plans. A third edition of the guideline, released in August 2008, summarizes current evidence about the assessment and treatment of chronic pain in mature adolescents (16 to 18 years old) and adults.
A distinct challenge to clinicians
Chronic pain—a persistent, life-altering condition—is one of the most challenging disorders for primary care physicians to treat. Unlike the case with acute pain, for which we seek to cure the underlying biologic condition, the goal of chronic pain management is to improve function in the face of pain that may never completely resolve.
Achieving that goal, according to the new guideline, requires a patient-centered, multifaceted approach—often involving a health-care team that includes specialists in behavioral health and physical rehabilitation—that is coordinated by a primary care physician. An effective treatment plan must address biopsychosocial factors as well as spiritual and cultural issues. Patients must be taught self-management skills focused on fitness, stress reduction, and maintaining a healthy lifestyle.
Grade A recommendations
- Develop a physician–patient partnership. This should include a plan of care and realistic goal-setting.
- Begin physical rehabilitation and psychosocial management. This includes an exercise fitness program, cognitive-behavioral therapy, and self-management.
Grade B recommendations
- Obtain a general history, including psychological assessment and spirituality evaluation, and identify barriers to treatment.
- Obtain a thorough pain history.
- Perform a physical examination, including a focused musculoskeletal and neurologic evaluation.
- Perform diagnostic testing as indicated. X-rays, computed tomography, magnetic resonance imaging, electromyography, and nerve conduction studies can help differentiate the biological mechanisms of pain.
- Teach patients to use pain scales for self-reporting.
Grade C recommendations
- Categorize the 4 biological mechanisms of pain (inflammatory, mechanical, musculoskeletal, or neuropathic).
- Consider the following pharmacologic options for Level-I care:
- Consider the following Level-I therapeutic procedures:
- Consider the following Level-II interventions:
Medications may be part of the treatment plan but should not be the sole focus, according to the guideline. Opioids are an option when other therapies fail.
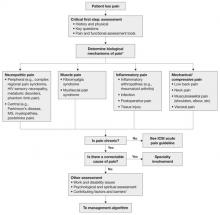
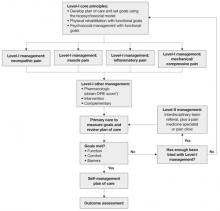
The updated ICSI guideline also addresses the effects of various therapies, the role of psychosocial factors, and the identification of barriers to treatment. The comprehensive guideline, which has 172 references and nine appendices, also features two easy-to-use algorithms. One addresses the assessment of chronic pain ( FIGURE 1 ) and the other deals with chronic pain management ( FIGURE 2 ).
Both algorithms identify Level-I and Level-II strategies that can be readily adapted to primary care practice. They are extremely helpful to physicians who are evaluating and developing a care plan for a patient who has chronic pain.
FIGURE 1 Chronic pain assessment
HIV, human immunodeficiency virus; ICSI, Institute for Clinical Systems Improvement; MS, multiple sclerosis.
*Pain types and contributing factors are not mutually exclusive. Patients frequently have more than one type of pain, as well as overlapping contributing factors.
Source: Institute for Clinical Systems Improvement. Reprinted with permission.
4 objectives
This latest guideline was developed to:
- improve the treatment of adult chronic-pain patients by encouraging physicians to complete an appropriate biopsychosocial assessment (and reassessment)
- improve patients’ function by recommending development and use of a comprehensive treatment plan that includes a multispecialty team
- improve the use of Level-I and Level-II treatment approaches to chronic pain
- provide guidance on the most effective use of nonopioid and opioid medications in the treatment of chronic pain.
With these objectives in mind, the ICSI work group conducted a comprehensive literature review, giving priority to randomized controlled trials (RCTs), meta-analyses, and systematic reviews. The work group used a seven-tier grading system to rate the evidence and a three-category system for the worksheets in the guideline appendices.
For this article, we converted evidence ratings in the guideline into so-called strength-of-recommendation taxonomy, or SORT.1
What aspects of practice have changed?
In addition to reflecting the latest research, the new guideline contains a number of clarifications. For example: The update states that medications are not the “sole” focus of treatment and should be used, when necessary, as part of an overall approach to pain management. (The previous version noted that medications were not the “primary” focus.)
The management algorithm ( FIGURE 2 ) now leads with “core principles”—a term suggesting greater importance than the former term, “general management,” implied. Clinical highlights, a synthesis of key recommendations, have been revised to better align with the guideline’s main components—assessment, functional goals, patient-centered/biopsychosocial care planning, Level-I versus Level-II approaches, and medication and patient selection.
Other changes in the guideline may contribute to clinicians’ understanding of chronic pain and its complex presentation. The guideline now includes a statement about allodynia and hyperalgesia to indicate that both may play an important role in any pain syndrome—not just in complex regional pain syndrome. Information about fibromyalgia symptoms and myofascial pain has been added. The definitions page now has an entry for “biopsychosocial model,” as well as language designed to stress the differences between untreated acute pain and ongoing chronic pain.
FIGURE 2 Chronic pain management
* DIRE, diagnosis, intractability, risk, efficacy.
Source: Institute for Clinical System Improvement. Reprinted with permission.
A limitation, an improvement
A limitation of the guideline is the lack of studies addressing the effectiveness of a comprehensive, multidisciplinary treatment approach to chronic pain management; most studies consider single-therapy management. An improvement, on the other hand, is that the evidence levels for each strategy are now listed within the section describing it—a notable change that makes it easier to identify the quality of individual recommendations.
As has been the case in the past, this latest edition of the guideline offers a number of tools for physicians. The assessment and management algorithms walk clinicians through decision-making. In addition, the following nine appendices provide practical guidance to physicians in various aspects of patient evaluation and care:
- Brief Pain Inventory (Short Form)
- Patient Health Questionnaire (PHQ-9)
- Functional Ability Questionnaire
- Personal Care Plan for Chronic Pain
- DIRE (diagnosis, intractability, risk, efficacy) Score: Patient Selection for Chronic Opioid Analgesia
- Opioid Agreement Form
- Opioid Analgesics
- Pharmaceutical Interventions for Neuropathic Pain
- Neuropathic Pain Treatment Diagram.
As noted, the source document for this guideline is: Assessment and Management of Chronic Pain. 3rd ed. Bloomington (Minn): Institute for Clinical Systems Improvement (ICSI); 2008 July.
The complete guideline is available at: pain__chronic__assessment_and_management_of__guideline_.html " target="_blank"> http://www.icsi.org/pain__chronic__assessment_and_management_of_14399/
pain__chronic__assessment_and_management_of__guideline_.html . (Accessed August 18, 2009.)
Reference
1. Ebell M, Siwek J, Weiss BD, et al. Strength of recommendation taxonomy (SORT): a patient-centered approach to grading evidence in the medical literature. J Fam Pract. 2004;53:111-120.
The authors report no financial relationships relevant to this article.
This article is adapted from the December 2008 installment of The Journal of Family Practice’s “Guideline Update” series. The Journal of Family Practice is an NLM-indexed publication of Quadrant HealthCom Inc., publisher of OBG Management.
- What are the critical steps in the assessment of a patient who suffers chronic pain?
- What are the four biologic mechanisms of pain?
- When is referral to a pain specialist recommended?
Answers to these questions are summarized below, and in the 2008 edition of Assessment and Management of Chronic Pain, a practice guideline developed and first published in 2005 by the Institute for Clinical Systems Improvement (ICSI), which also funded the work. ICSI is a collaboration of 57 medical groups sponsored by six Minnesota health plans. A third edition of the guideline, released in August 2008, summarizes current evidence about the assessment and treatment of chronic pain in mature adolescents (16 to 18 years old) and adults.
A distinct challenge to clinicians
Chronic pain—a persistent, life-altering condition—is one of the most challenging disorders for primary care physicians to treat. Unlike the case with acute pain, for which we seek to cure the underlying biologic condition, the goal of chronic pain management is to improve function in the face of pain that may never completely resolve.
Achieving that goal, according to the new guideline, requires a patient-centered, multifaceted approach—often involving a health-care team that includes specialists in behavioral health and physical rehabilitation—that is coordinated by a primary care physician. An effective treatment plan must address biopsychosocial factors as well as spiritual and cultural issues. Patients must be taught self-management skills focused on fitness, stress reduction, and maintaining a healthy lifestyle.
Grade A recommendations
- Develop a physician–patient partnership. This should include a plan of care and realistic goal-setting.
- Begin physical rehabilitation and psychosocial management. This includes an exercise fitness program, cognitive-behavioral therapy, and self-management.
Grade B recommendations
- Obtain a general history, including psychological assessment and spirituality evaluation, and identify barriers to treatment.
- Obtain a thorough pain history.
- Perform a physical examination, including a focused musculoskeletal and neurologic evaluation.
- Perform diagnostic testing as indicated. X-rays, computed tomography, magnetic resonance imaging, electromyography, and nerve conduction studies can help differentiate the biological mechanisms of pain.
- Teach patients to use pain scales for self-reporting.
Grade C recommendations
- Categorize the 4 biological mechanisms of pain (inflammatory, mechanical, musculoskeletal, or neuropathic).
- Consider the following pharmacologic options for Level-I care:
- Consider the following Level-I therapeutic procedures:
- Consider the following Level-II interventions:
Medications may be part of the treatment plan but should not be the sole focus, according to the guideline. Opioids are an option when other therapies fail.
The updated ICSI guideline also addresses the effects of various therapies, the role of psychosocial factors, and the identification of barriers to treatment. The comprehensive guideline, which has 172 references and nine appendices, also features two easy-to-use algorithms. One addresses the assessment of chronic pain ( FIGURE 1 ) and the other deals with chronic pain management ( FIGURE 2 ).
Both algorithms identify Level-I and Level-II strategies that can be readily adapted to primary care practice. They are extremely helpful to physicians who are evaluating and developing a care plan for a patient who has chronic pain.
FIGURE 1 Chronic pain assessment
HIV, human immunodeficiency virus; ICSI, Institute for Clinical Systems Improvement; MS, multiple sclerosis.
*Pain types and contributing factors are not mutually exclusive. Patients frequently have more than one type of pain, as well as overlapping contributing factors.
Source: Institute for Clinical Systems Improvement. Reprinted with permission.
4 objectives
This latest guideline was developed to:
- improve the treatment of adult chronic-pain patients by encouraging physicians to complete an appropriate biopsychosocial assessment (and reassessment)
- improve patients’ function by recommending development and use of a comprehensive treatment plan that includes a multispecialty team
- improve the use of Level-I and Level-II treatment approaches to chronic pain
- provide guidance on the most effective use of nonopioid and opioid medications in the treatment of chronic pain.
With these objectives in mind, the ICSI work group conducted a comprehensive literature review, giving priority to randomized controlled trials (RCTs), meta-analyses, and systematic reviews. The work group used a seven-tier grading system to rate the evidence and a three-category system for the worksheets in the guideline appendices.
For this article, we converted evidence ratings in the guideline into so-called strength-of-recommendation taxonomy, or SORT.1
What aspects of practice have changed?
In addition to reflecting the latest research, the new guideline contains a number of clarifications. For example: The update states that medications are not the “sole” focus of treatment and should be used, when necessary, as part of an overall approach to pain management. (The previous version noted that medications were not the “primary” focus.)
The management algorithm ( FIGURE 2 ) now leads with “core principles”—a term suggesting greater importance than the former term, “general management,” implied. Clinical highlights, a synthesis of key recommendations, have been revised to better align with the guideline’s main components—assessment, functional goals, patient-centered/biopsychosocial care planning, Level-I versus Level-II approaches, and medication and patient selection.
Other changes in the guideline may contribute to clinicians’ understanding of chronic pain and its complex presentation. The guideline now includes a statement about allodynia and hyperalgesia to indicate that both may play an important role in any pain syndrome—not just in complex regional pain syndrome. Information about fibromyalgia symptoms and myofascial pain has been added. The definitions page now has an entry for “biopsychosocial model,” as well as language designed to stress the differences between untreated acute pain and ongoing chronic pain.
FIGURE 2 Chronic pain management
* DIRE, diagnosis, intractability, risk, efficacy.
Source: Institute for Clinical System Improvement. Reprinted with permission.
A limitation, an improvement
A limitation of the guideline is the lack of studies addressing the effectiveness of a comprehensive, multidisciplinary treatment approach to chronic pain management; most studies consider single-therapy management. An improvement, on the other hand, is that the evidence levels for each strategy are now listed within the section describing it—a notable change that makes it easier to identify the quality of individual recommendations.
As has been the case in the past, this latest edition of the guideline offers a number of tools for physicians. The assessment and management algorithms walk clinicians through decision-making. In addition, the following nine appendices provide practical guidance to physicians in various aspects of patient evaluation and care:
- Brief Pain Inventory (Short Form)
- Patient Health Questionnaire (PHQ-9)
- Functional Ability Questionnaire
- Personal Care Plan for Chronic Pain
- DIRE (diagnosis, intractability, risk, efficacy) Score: Patient Selection for Chronic Opioid Analgesia
- Opioid Agreement Form
- Opioid Analgesics
- Pharmaceutical Interventions for Neuropathic Pain
- Neuropathic Pain Treatment Diagram.
As noted, the source document for this guideline is: Assessment and Management of Chronic Pain. 3rd ed. Bloomington (Minn): Institute for Clinical Systems Improvement (ICSI); 2008 July.
The complete guideline is available at: pain__chronic__assessment_and_management_of__guideline_.html " target="_blank"> http://www.icsi.org/pain__chronic__assessment_and_management_of_14399/
pain__chronic__assessment_and_management_of__guideline_.html . (Accessed August 18, 2009.)
The authors report no financial relationships relevant to this article.
This article is adapted from the December 2008 installment of The Journal of Family Practice’s “Guideline Update” series. The Journal of Family Practice is an NLM-indexed publication of Quadrant HealthCom Inc., publisher of OBG Management.
- What are the critical steps in the assessment of a patient who suffers chronic pain?
- What are the four biologic mechanisms of pain?
- When is referral to a pain specialist recommended?
Answers to these questions are summarized below, and in the 2008 edition of Assessment and Management of Chronic Pain, a practice guideline developed and first published in 2005 by the Institute for Clinical Systems Improvement (ICSI), which also funded the work. ICSI is a collaboration of 57 medical groups sponsored by six Minnesota health plans. A third edition of the guideline, released in August 2008, summarizes current evidence about the assessment and treatment of chronic pain in mature adolescents (16 to 18 years old) and adults.
A distinct challenge to clinicians
Chronic pain—a persistent, life-altering condition—is one of the most challenging disorders for primary care physicians to treat. Unlike the case with acute pain, for which we seek to cure the underlying biologic condition, the goal of chronic pain management is to improve function in the face of pain that may never completely resolve.
Achieving that goal, according to the new guideline, requires a patient-centered, multifaceted approach—often involving a health-care team that includes specialists in behavioral health and physical rehabilitation—that is coordinated by a primary care physician. An effective treatment plan must address biopsychosocial factors as well as spiritual and cultural issues. Patients must be taught self-management skills focused on fitness, stress reduction, and maintaining a healthy lifestyle.
Grade A recommendations
- Develop a physician–patient partnership. This should include a plan of care and realistic goal-setting.
- Begin physical rehabilitation and psychosocial management. This includes an exercise fitness program, cognitive-behavioral therapy, and self-management.
Grade B recommendations
- Obtain a general history, including psychological assessment and spirituality evaluation, and identify barriers to treatment.
- Obtain a thorough pain history.
- Perform a physical examination, including a focused musculoskeletal and neurologic evaluation.
- Perform diagnostic testing as indicated. X-rays, computed tomography, magnetic resonance imaging, electromyography, and nerve conduction studies can help differentiate the biological mechanisms of pain.
- Teach patients to use pain scales for self-reporting.
Grade C recommendations
- Categorize the 4 biological mechanisms of pain (inflammatory, mechanical, musculoskeletal, or neuropathic).
- Consider the following pharmacologic options for Level-I care:
- Consider the following Level-I therapeutic procedures:
- Consider the following Level-II interventions:
Medications may be part of the treatment plan but should not be the sole focus, according to the guideline. Opioids are an option when other therapies fail.
The updated ICSI guideline also addresses the effects of various therapies, the role of psychosocial factors, and the identification of barriers to treatment. The comprehensive guideline, which has 172 references and nine appendices, also features two easy-to-use algorithms. One addresses the assessment of chronic pain ( FIGURE 1 ) and the other deals with chronic pain management ( FIGURE 2 ).
Both algorithms identify Level-I and Level-II strategies that can be readily adapted to primary care practice. They are extremely helpful to physicians who are evaluating and developing a care plan for a patient who has chronic pain.
FIGURE 1 Chronic pain assessment
HIV, human immunodeficiency virus; ICSI, Institute for Clinical Systems Improvement; MS, multiple sclerosis.
*Pain types and contributing factors are not mutually exclusive. Patients frequently have more than one type of pain, as well as overlapping contributing factors.
Source: Institute for Clinical Systems Improvement. Reprinted with permission.
4 objectives
This latest guideline was developed to:
- improve the treatment of adult chronic-pain patients by encouraging physicians to complete an appropriate biopsychosocial assessment (and reassessment)
- improve patients’ function by recommending development and use of a comprehensive treatment plan that includes a multispecialty team
- improve the use of Level-I and Level-II treatment approaches to chronic pain
- provide guidance on the most effective use of nonopioid and opioid medications in the treatment of chronic pain.
With these objectives in mind, the ICSI work group conducted a comprehensive literature review, giving priority to randomized controlled trials (RCTs), meta-analyses, and systematic reviews. The work group used a seven-tier grading system to rate the evidence and a three-category system for the worksheets in the guideline appendices.
For this article, we converted evidence ratings in the guideline into so-called strength-of-recommendation taxonomy, or SORT.1
What aspects of practice have changed?
In addition to reflecting the latest research, the new guideline contains a number of clarifications. For example: The update states that medications are not the “sole” focus of treatment and should be used, when necessary, as part of an overall approach to pain management. (The previous version noted that medications were not the “primary” focus.)
The management algorithm ( FIGURE 2 ) now leads with “core principles”—a term suggesting greater importance than the former term, “general management,” implied. Clinical highlights, a synthesis of key recommendations, have been revised to better align with the guideline’s main components—assessment, functional goals, patient-centered/biopsychosocial care planning, Level-I versus Level-II approaches, and medication and patient selection.
Other changes in the guideline may contribute to clinicians’ understanding of chronic pain and its complex presentation. The guideline now includes a statement about allodynia and hyperalgesia to indicate that both may play an important role in any pain syndrome—not just in complex regional pain syndrome. Information about fibromyalgia symptoms and myofascial pain has been added. The definitions page now has an entry for “biopsychosocial model,” as well as language designed to stress the differences between untreated acute pain and ongoing chronic pain.
FIGURE 2 Chronic pain management
* DIRE, diagnosis, intractability, risk, efficacy.
Source: Institute for Clinical System Improvement. Reprinted with permission.
A limitation, an improvement
A limitation of the guideline is the lack of studies addressing the effectiveness of a comprehensive, multidisciplinary treatment approach to chronic pain management; most studies consider single-therapy management. An improvement, on the other hand, is that the evidence levels for each strategy are now listed within the section describing it—a notable change that makes it easier to identify the quality of individual recommendations.
As has been the case in the past, this latest edition of the guideline offers a number of tools for physicians. The assessment and management algorithms walk clinicians through decision-making. In addition, the following nine appendices provide practical guidance to physicians in various aspects of patient evaluation and care:
- Brief Pain Inventory (Short Form)
- Patient Health Questionnaire (PHQ-9)
- Functional Ability Questionnaire
- Personal Care Plan for Chronic Pain
- DIRE (diagnosis, intractability, risk, efficacy) Score: Patient Selection for Chronic Opioid Analgesia
- Opioid Agreement Form
- Opioid Analgesics
- Pharmaceutical Interventions for Neuropathic Pain
- Neuropathic Pain Treatment Diagram.
As noted, the source document for this guideline is: Assessment and Management of Chronic Pain. 3rd ed. Bloomington (Minn): Institute for Clinical Systems Improvement (ICSI); 2008 July.
The complete guideline is available at: pain__chronic__assessment_and_management_of__guideline_.html " target="_blank"> http://www.icsi.org/pain__chronic__assessment_and_management_of_14399/
pain__chronic__assessment_and_management_of__guideline_.html . (Accessed August 18, 2009.)
Reference
1. Ebell M, Siwek J, Weiss BD, et al. Strength of recommendation taxonomy (SORT): a patient-centered approach to grading evidence in the medical literature. J Fam Pract. 2004;53:111-120.
Reference
1. Ebell M, Siwek J, Weiss BD, et al. Strength of recommendation taxonomy (SORT): a patient-centered approach to grading evidence in the medical literature. J Fam Pract. 2004;53:111-120.
Guidelines confirm safety of pregnancy in women who have epilepsy—with caveats
“Good evidence shows that valproate is linked to an increased risk for fetal malformations and decreased thinking skills in children, whether used by itself or with other medications,” said lead guideline author Cynthia Harden, MD, director of the Epilepsy Division at the University of Miami’s Miller School of Medicine and member of the AAN.
The guidelines also suggest that, if possible, women who have epilepsy avoid taking more than one epilepsy drug during pregnancy because the use of multiple antiseizure medications increases the risk of birth defects.
In addition, the guidelines recommend that physicians avoid prescribing the epilepsy drugs phenytoin and phenobarbital during pregnancy. When a fetus is exposed to one of these drugs, cognitive development may be impaired.
Safe pregnancy is likely—if no seizures occur
Aside from the risks known to be associated with valproate, phenytoin, phenobarbital, and polytherapy, pregnancy in women who have well-controlled epilepsy appears to be relatively safe.
“Overall, what we found should be very reassuring to every woman with epilepsy planning to become pregnant,” said Harden.
“These guidelines show that women with epilepsy are not at a substantially increased risk of having a cesarean section, late-pregnancy bleeding, or premature contractions or premature labor and delivery. Also, if a woman is seizure-free 9 months before she becomes pregnant, it’s likely that she will not have any seizures during the pregnancy.”
However, a just-published study suggests that the presence of seizures during pregnancy confers some degree of risk, according to data from Yi-Hua Chen, PhD, and colleagues, of Taipei Medical University and General Cathay Hospital in Taiwan.4
Chen and associates performed a retrospective cross-sectional study that linked two nationwide population-based data sets from Taiwan. The study focused on 1,016 women who had epilepsy and who delivered singleton infants from 2001 to 2003; these women had been diagnosed with epilepsy within 2 years prior to their index delivery. Women who had epilepsy were further stratified into two groups: those who did and those who did not have seizures during pregnancy. They were compared with 8,128 women who had no chronic disease.
Women who experienced seizures during pregnancy were more likely to give birth to preterm, small, or low-birth-weight babies than were women who did not have epilepsy. In addition, women who experienced seizures during pregnancy were more likely to give birth to a small-for-gestational-age infant than were women who had epilepsy but who did not have seizures.
Some previous studies had reported a link between adverse pregnancy outcomes and a mother’s epilepsy, but others found no association, Chen and colleagues observed.
“Our study further illuminates these conflicting data to suggest that it is the seizures themselves that seem to contribute greatly to the increased risk of infants being delivered preterm, of low birthweight, and small for gestational age. For women who remained seizure-free throughout pregnancy, null or mild risk was identified, compared with unaffected women,” they wrote.
- It is estimated that approximately 500,000 women of childbearing age in the United States have epilepsy, and that 3 to 5 of every 1,000 births are to women who have epilepsy.
- Most people who have epilepsy have well-controlled seizures and are otherwise healthy.
- The birth rate in women who have epilepsy is slightly lower than it is in women who do not have the disease.10
- Most women who have epilepsy have uneventful pregnancies and deliver healthy infants with no complications.11
- Epilepsy can be associated with reproductive endocrine disorders, including polycystic ovary syndrome, hypothalamic amenorrhea, or functional hyperprolactinemia, possibly through the effects of antiepileptic drugs.12
- The children of women who have idiopathic epilepsy have a slightly elevated risk ( 13
As a safeguard, measure blood levels
of antiseizure drugs
The guidelines from the AAN and AES recommend that pregnant women who have epilepsy consider having their blood tested regularly.
“Levels of seizure medications in the blood tend to drop during pregnancy, so checking these levels and adjusting the medication doses should help to keep the levels in the effective range and the pregnant woman seizure-free,” said Harden.
Antiepileptic drugs should be administered at the lowest dosage and lowest plasma level that protects against tonic-clonic seizures and other complex partial seizures.5
to protect the fetus
Here is a summary of the main recommendations in the guidelines from the American Academy of Neurology and American Epilepsy Society:
Avoid certain drugs; discourage smoking
- Avoid first-trimester exposure to the antiepileptic drug valproate because of its link to an increased risk of fetal malformation and cognitive impairment in children. Also avoid epilepsy drug polytherapy during the first trimester.
- Besides avoiding valproate and antiepileptic drug polytherapy during the first trimester, women who have epilepsy should avoid these regimens throughout pregnancy to prevent adverse cognitive outcomes in the infant.
- Avoid prescribing phenytoin and phenobarbital during pregnancy.
- Women who take antiepileptic drugs are probably at increased risk of delivering a small-for-gestational-age baby and, possibly, delivering a newborn with an Apgar score below 7 at 1 minute.
- Women who have epilepsy and who smoke may increase the risk that they will develop premature contractions, premature labor, and premature delivery.
Monitor levels of some drugs
- Monitor levels of lamotrigine, carbamazepine, and phenytoin during pregnancy. Also monitor levels of levetiracetam and oxcarbazepine (a monohydroxy derivative). Blood levels of antiepileptic drugs tend to drop during pregnancy, and the dosage may need to be adjusted.
Seizure-free pregnancy is possible
- Counsel women who have epilepsy that remaining free from seizures for at least 9 months before pregnancy greatly increases the likelihood that they will remain seizure-free during pregnancy.
Folic acid may be beneficial
- Consider giving women who have epilepsy at least 0.4 mg of folic acid daily before they become pregnant, as it appears likely to lower the risk of major congenital malformation. It is unclear whether a higher daily dosage offers greater protective benefits.
Counsel the mother about breastfeeding concerns
- Women who have epilepsy and who choose to breastfeed should be counseled that primidone and levetiracetam probably pass into breast milk in significant amounts. In addition, gabapentin, lamotrigine, and topiramate may pass into breast milk in significant amounts. In contrast, valproate, phenobarbital, phenytoin, and carbamazepine probably do not pass into breast milk in clinically important amounts.
Guidelines were based on a review of the literature
The guidelines were developed after a review of all scientific studies available on each topic and were published in the online issue of the journal Epilepsia.1-3 Their development was supported in part by the Milken Family Foundation.
“For too long, women living with epilepsy have feared the added risk of premature birth and other consequences of both their epilepsy and their medications,” said Howard R. Soule, PhD, chief science officer for the Milken Family Foundation. “The results of this project will help relieve the worries of these women and their families.”
Do not withdraw antiepileptic drugs during pregnancy
Some physicians attempt to discontinue an antiepileptic drug when a woman has gone 2 years without experiencing a seizure.5 In this scenario, the likelihood that seizures will recur within 6 and 12 months is 12% and 32%, respectively.6 Because of the risk that seizures will recur, and the increased likelihood of adverse outcomes associated with seizures during pregnancy, antiepileptic medication should not be discontinued during gestation.
Nor should a woman attempt to transition from one antiepileptic drug to another during pregnancy solely for the purpose of reducing teratogenicity.5 Doing so could precipitate seizures and exposes the fetus to the potentially hazardous effects of an additional antiseizure medication during a critical period. Moreover, there may be no advantage associated with switching drugs once a pregnancy has been established.7
Screen for malformations rigorously
Comprehensive screening for fetal anomalies early in the pregnancy is recommended for two main reasons:
- If a malformation is identified, the mother has the option of terminating the pregnancy
- Even if the patient decides not to terminate a gestation in which fetal anomaly has been identified, the information may help the practitioner determine the best mode and place of delivery.
Cesarean delivery may be warranted if the mother
has had recent seizures
Although most women who have epilepsy can expect to have a normal vaginal delivery, elective cesarean should be considered if the mother has experienced frequent seizures during the third trimester, or if she has a history of stress-related status epilepticus.5
Tonic-clonic seizures occur during labor in 1% to 2% of women who have epilepsy, and in an additional 1% to 2% of women in the 24 hours immediately following delivery.
Plasma levels of antiepileptic medication should be monitored during the third trimester and delivery to ensure that the medication is given in adequate strength to prevent seizures. In addition, the patient should be counseled about the importance of taking her medication consistently during this period.5
If the patient experiences a seizure during labor and delivery, treat her promptly with an intravenous benzodiazepine, preferably lorazepam.8 However, be aware that phenobarbital, primidone, and benzodiazepines remain in neonatal plasma for several days after delivery and may cause sedation or neonatal withdrawal syndrome.9
Do not give magnesium sulfate for epileptic seizures unless the seizures first appear during the third trimester or immediate postpartum period and could be associated with eclampsia. In such cases, treat the eclampsia and evaluate the patient for other potential causes of the seizures.5
1. Harden CL, Hopp J, Ting TY, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): I. Obstetrical complications and change in seizure frequency: Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1229-1236.
2. Harden CL, Meador KJ, Pennell PB, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): II. Teratogenesis and perinatal outcomes: Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1237-1246.
3. Harden CL, Pennell PB, Koppel BS, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): III. Vitamin K, folic acid, blood levels, and breast-feeding. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1247-1255.
4. Chen YH, Chiou HY, Lin HC, Lin HL. Affect of seizures during gestation on pregnancy outcomes in women with epilepsy. Arch Neurol. 2009;66:979-984.
5. Schachter SC. Management of epilepsy and pregnancy. ©2009. http://www.UpToDate.com. Available at: http://www.uptodate.com/patients/content/topic.do?topicKey=~JqqkRi44op3f4Y. Accessed Aug. 17, 2009.
6. EURAP study group. Seizure control and treatment in pregnancy: observations from the EURAP epilepsy pregnancy registry. Neurology. 2006;66:354-360.
7. Practice parameter: management issues for women with epilepsy (summary statement). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 1998;51:944-948.
8. Yerby MS. Problems and management of the pregnant woman with epilepsy. Epilepsia. 1987;28 Suppl 3:S29-S36.
9. Kuhnz W, Koch S, Helge H, Nau H. Primidone and phenobarbital during lactation period in epileptic women: total and free drug serum levels in the nursed infants and their effects on neonatal behavior. Dev Pharmacol Ther. 1988;11:147-154.
10. Artama M, Isojarvi JI, Raitanen J, Auvinen A. Birth rate among patients with epilepsy: a nationwide population-based cohort study in Finland. Am J Epidemiol. 2004;159:1057-1063.
11. Crawford PM. Managing epilepsy in women of childbearing age. Drug Saf. 2009;32:293-307.
12. Bauer J, Cooper-Mahkorn D. Reproductive dysfunction in women with epilepsy: menstrual cycle abnormalities, fertility, and polycystic ovary syndrome. Int Rev Neurobiol. 2008;83:135-155.
13. Dam M. Is epilepsy hereditary? Available at: http://www.epilepsy.dk/Handbook/Hereditary-uk.asp. Accessed Aug. 18, 2009.
“Good evidence shows that valproate is linked to an increased risk for fetal malformations and decreased thinking skills in children, whether used by itself or with other medications,” said lead guideline author Cynthia Harden, MD, director of the Epilepsy Division at the University of Miami’s Miller School of Medicine and member of the AAN.
The guidelines also suggest that, if possible, women who have epilepsy avoid taking more than one epilepsy drug during pregnancy because the use of multiple antiseizure medications increases the risk of birth defects.
In addition, the guidelines recommend that physicians avoid prescribing the epilepsy drugs phenytoin and phenobarbital during pregnancy. When a fetus is exposed to one of these drugs, cognitive development may be impaired.
Safe pregnancy is likely—if no seizures occur
Aside from the risks known to be associated with valproate, phenytoin, phenobarbital, and polytherapy, pregnancy in women who have well-controlled epilepsy appears to be relatively safe.
“Overall, what we found should be very reassuring to every woman with epilepsy planning to become pregnant,” said Harden.
“These guidelines show that women with epilepsy are not at a substantially increased risk of having a cesarean section, late-pregnancy bleeding, or premature contractions or premature labor and delivery. Also, if a woman is seizure-free 9 months before she becomes pregnant, it’s likely that she will not have any seizures during the pregnancy.”
However, a just-published study suggests that the presence of seizures during pregnancy confers some degree of risk, according to data from Yi-Hua Chen, PhD, and colleagues, of Taipei Medical University and General Cathay Hospital in Taiwan.4
Chen and associates performed a retrospective cross-sectional study that linked two nationwide population-based data sets from Taiwan. The study focused on 1,016 women who had epilepsy and who delivered singleton infants from 2001 to 2003; these women had been diagnosed with epilepsy within 2 years prior to their index delivery. Women who had epilepsy were further stratified into two groups: those who did and those who did not have seizures during pregnancy. They were compared with 8,128 women who had no chronic disease.
Women who experienced seizures during pregnancy were more likely to give birth to preterm, small, or low-birth-weight babies than were women who did not have epilepsy. In addition, women who experienced seizures during pregnancy were more likely to give birth to a small-for-gestational-age infant than were women who had epilepsy but who did not have seizures.
Some previous studies had reported a link between adverse pregnancy outcomes and a mother’s epilepsy, but others found no association, Chen and colleagues observed.
“Our study further illuminates these conflicting data to suggest that it is the seizures themselves that seem to contribute greatly to the increased risk of infants being delivered preterm, of low birthweight, and small for gestational age. For women who remained seizure-free throughout pregnancy, null or mild risk was identified, compared with unaffected women,” they wrote.
- It is estimated that approximately 500,000 women of childbearing age in the United States have epilepsy, and that 3 to 5 of every 1,000 births are to women who have epilepsy.
- Most people who have epilepsy have well-controlled seizures and are otherwise healthy.
- The birth rate in women who have epilepsy is slightly lower than it is in women who do not have the disease.10
- Most women who have epilepsy have uneventful pregnancies and deliver healthy infants with no complications.11
- Epilepsy can be associated with reproductive endocrine disorders, including polycystic ovary syndrome, hypothalamic amenorrhea, or functional hyperprolactinemia, possibly through the effects of antiepileptic drugs.12
- The children of women who have idiopathic epilepsy have a slightly elevated risk ( 13
As a safeguard, measure blood levels
of antiseizure drugs
The guidelines from the AAN and AES recommend that pregnant women who have epilepsy consider having their blood tested regularly.
“Levels of seizure medications in the blood tend to drop during pregnancy, so checking these levels and adjusting the medication doses should help to keep the levels in the effective range and the pregnant woman seizure-free,” said Harden.
Antiepileptic drugs should be administered at the lowest dosage and lowest plasma level that protects against tonic-clonic seizures and other complex partial seizures.5
to protect the fetus
Here is a summary of the main recommendations in the guidelines from the American Academy of Neurology and American Epilepsy Society:
Avoid certain drugs; discourage smoking
- Avoid first-trimester exposure to the antiepileptic drug valproate because of its link to an increased risk of fetal malformation and cognitive impairment in children. Also avoid epilepsy drug polytherapy during the first trimester.
- Besides avoiding valproate and antiepileptic drug polytherapy during the first trimester, women who have epilepsy should avoid these regimens throughout pregnancy to prevent adverse cognitive outcomes in the infant.
- Avoid prescribing phenytoin and phenobarbital during pregnancy.
- Women who take antiepileptic drugs are probably at increased risk of delivering a small-for-gestational-age baby and, possibly, delivering a newborn with an Apgar score below 7 at 1 minute.
- Women who have epilepsy and who smoke may increase the risk that they will develop premature contractions, premature labor, and premature delivery.
Monitor levels of some drugs
- Monitor levels of lamotrigine, carbamazepine, and phenytoin during pregnancy. Also monitor levels of levetiracetam and oxcarbazepine (a monohydroxy derivative). Blood levels of antiepileptic drugs tend to drop during pregnancy, and the dosage may need to be adjusted.
Seizure-free pregnancy is possible
- Counsel women who have epilepsy that remaining free from seizures for at least 9 months before pregnancy greatly increases the likelihood that they will remain seizure-free during pregnancy.
Folic acid may be beneficial
- Consider giving women who have epilepsy at least 0.4 mg of folic acid daily before they become pregnant, as it appears likely to lower the risk of major congenital malformation. It is unclear whether a higher daily dosage offers greater protective benefits.
Counsel the mother about breastfeeding concerns
- Women who have epilepsy and who choose to breastfeed should be counseled that primidone and levetiracetam probably pass into breast milk in significant amounts. In addition, gabapentin, lamotrigine, and topiramate may pass into breast milk in significant amounts. In contrast, valproate, phenobarbital, phenytoin, and carbamazepine probably do not pass into breast milk in clinically important amounts.
Guidelines were based on a review of the literature
The guidelines were developed after a review of all scientific studies available on each topic and were published in the online issue of the journal Epilepsia.1-3 Their development was supported in part by the Milken Family Foundation.
“For too long, women living with epilepsy have feared the added risk of premature birth and other consequences of both their epilepsy and their medications,” said Howard R. Soule, PhD, chief science officer for the Milken Family Foundation. “The results of this project will help relieve the worries of these women and their families.”
Do not withdraw antiepileptic drugs during pregnancy
Some physicians attempt to discontinue an antiepileptic drug when a woman has gone 2 years without experiencing a seizure.5 In this scenario, the likelihood that seizures will recur within 6 and 12 months is 12% and 32%, respectively.6 Because of the risk that seizures will recur, and the increased likelihood of adverse outcomes associated with seizures during pregnancy, antiepileptic medication should not be discontinued during gestation.
Nor should a woman attempt to transition from one antiepileptic drug to another during pregnancy solely for the purpose of reducing teratogenicity.5 Doing so could precipitate seizures and exposes the fetus to the potentially hazardous effects of an additional antiseizure medication during a critical period. Moreover, there may be no advantage associated with switching drugs once a pregnancy has been established.7
Screen for malformations rigorously
Comprehensive screening for fetal anomalies early in the pregnancy is recommended for two main reasons:
- If a malformation is identified, the mother has the option of terminating the pregnancy
- Even if the patient decides not to terminate a gestation in which fetal anomaly has been identified, the information may help the practitioner determine the best mode and place of delivery.
Cesarean delivery may be warranted if the mother
has had recent seizures
Although most women who have epilepsy can expect to have a normal vaginal delivery, elective cesarean should be considered if the mother has experienced frequent seizures during the third trimester, or if she has a history of stress-related status epilepticus.5
Tonic-clonic seizures occur during labor in 1% to 2% of women who have epilepsy, and in an additional 1% to 2% of women in the 24 hours immediately following delivery.
Plasma levels of antiepileptic medication should be monitored during the third trimester and delivery to ensure that the medication is given in adequate strength to prevent seizures. In addition, the patient should be counseled about the importance of taking her medication consistently during this period.5
If the patient experiences a seizure during labor and delivery, treat her promptly with an intravenous benzodiazepine, preferably lorazepam.8 However, be aware that phenobarbital, primidone, and benzodiazepines remain in neonatal plasma for several days after delivery and may cause sedation or neonatal withdrawal syndrome.9
Do not give magnesium sulfate for epileptic seizures unless the seizures first appear during the third trimester or immediate postpartum period and could be associated with eclampsia. In such cases, treat the eclampsia and evaluate the patient for other potential causes of the seizures.5
“Good evidence shows that valproate is linked to an increased risk for fetal malformations and decreased thinking skills in children, whether used by itself or with other medications,” said lead guideline author Cynthia Harden, MD, director of the Epilepsy Division at the University of Miami’s Miller School of Medicine and member of the AAN.
The guidelines also suggest that, if possible, women who have epilepsy avoid taking more than one epilepsy drug during pregnancy because the use of multiple antiseizure medications increases the risk of birth defects.
In addition, the guidelines recommend that physicians avoid prescribing the epilepsy drugs phenytoin and phenobarbital during pregnancy. When a fetus is exposed to one of these drugs, cognitive development may be impaired.
Safe pregnancy is likely—if no seizures occur
Aside from the risks known to be associated with valproate, phenytoin, phenobarbital, and polytherapy, pregnancy in women who have well-controlled epilepsy appears to be relatively safe.
“Overall, what we found should be very reassuring to every woman with epilepsy planning to become pregnant,” said Harden.
“These guidelines show that women with epilepsy are not at a substantially increased risk of having a cesarean section, late-pregnancy bleeding, or premature contractions or premature labor and delivery. Also, if a woman is seizure-free 9 months before she becomes pregnant, it’s likely that she will not have any seizures during the pregnancy.”
However, a just-published study suggests that the presence of seizures during pregnancy confers some degree of risk, according to data from Yi-Hua Chen, PhD, and colleagues, of Taipei Medical University and General Cathay Hospital in Taiwan.4
Chen and associates performed a retrospective cross-sectional study that linked two nationwide population-based data sets from Taiwan. The study focused on 1,016 women who had epilepsy and who delivered singleton infants from 2001 to 2003; these women had been diagnosed with epilepsy within 2 years prior to their index delivery. Women who had epilepsy were further stratified into two groups: those who did and those who did not have seizures during pregnancy. They were compared with 8,128 women who had no chronic disease.
Women who experienced seizures during pregnancy were more likely to give birth to preterm, small, or low-birth-weight babies than were women who did not have epilepsy. In addition, women who experienced seizures during pregnancy were more likely to give birth to a small-for-gestational-age infant than were women who had epilepsy but who did not have seizures.
Some previous studies had reported a link between adverse pregnancy outcomes and a mother’s epilepsy, but others found no association, Chen and colleagues observed.
“Our study further illuminates these conflicting data to suggest that it is the seizures themselves that seem to contribute greatly to the increased risk of infants being delivered preterm, of low birthweight, and small for gestational age. For women who remained seizure-free throughout pregnancy, null or mild risk was identified, compared with unaffected women,” they wrote.
- It is estimated that approximately 500,000 women of childbearing age in the United States have epilepsy, and that 3 to 5 of every 1,000 births are to women who have epilepsy.
- Most people who have epilepsy have well-controlled seizures and are otherwise healthy.
- The birth rate in women who have epilepsy is slightly lower than it is in women who do not have the disease.10
- Most women who have epilepsy have uneventful pregnancies and deliver healthy infants with no complications.11
- Epilepsy can be associated with reproductive endocrine disorders, including polycystic ovary syndrome, hypothalamic amenorrhea, or functional hyperprolactinemia, possibly through the effects of antiepileptic drugs.12
- The children of women who have idiopathic epilepsy have a slightly elevated risk ( 13
As a safeguard, measure blood levels
of antiseizure drugs
The guidelines from the AAN and AES recommend that pregnant women who have epilepsy consider having their blood tested regularly.
“Levels of seizure medications in the blood tend to drop during pregnancy, so checking these levels and adjusting the medication doses should help to keep the levels in the effective range and the pregnant woman seizure-free,” said Harden.
Antiepileptic drugs should be administered at the lowest dosage and lowest plasma level that protects against tonic-clonic seizures and other complex partial seizures.5
to protect the fetus
Here is a summary of the main recommendations in the guidelines from the American Academy of Neurology and American Epilepsy Society:
Avoid certain drugs; discourage smoking
- Avoid first-trimester exposure to the antiepileptic drug valproate because of its link to an increased risk of fetal malformation and cognitive impairment in children. Also avoid epilepsy drug polytherapy during the first trimester.
- Besides avoiding valproate and antiepileptic drug polytherapy during the first trimester, women who have epilepsy should avoid these regimens throughout pregnancy to prevent adverse cognitive outcomes in the infant.
- Avoid prescribing phenytoin and phenobarbital during pregnancy.
- Women who take antiepileptic drugs are probably at increased risk of delivering a small-for-gestational-age baby and, possibly, delivering a newborn with an Apgar score below 7 at 1 minute.
- Women who have epilepsy and who smoke may increase the risk that they will develop premature contractions, premature labor, and premature delivery.
Monitor levels of some drugs
- Monitor levels of lamotrigine, carbamazepine, and phenytoin during pregnancy. Also monitor levels of levetiracetam and oxcarbazepine (a monohydroxy derivative). Blood levels of antiepileptic drugs tend to drop during pregnancy, and the dosage may need to be adjusted.
Seizure-free pregnancy is possible
- Counsel women who have epilepsy that remaining free from seizures for at least 9 months before pregnancy greatly increases the likelihood that they will remain seizure-free during pregnancy.
Folic acid may be beneficial
- Consider giving women who have epilepsy at least 0.4 mg of folic acid daily before they become pregnant, as it appears likely to lower the risk of major congenital malformation. It is unclear whether a higher daily dosage offers greater protective benefits.
Counsel the mother about breastfeeding concerns
- Women who have epilepsy and who choose to breastfeed should be counseled that primidone and levetiracetam probably pass into breast milk in significant amounts. In addition, gabapentin, lamotrigine, and topiramate may pass into breast milk in significant amounts. In contrast, valproate, phenobarbital, phenytoin, and carbamazepine probably do not pass into breast milk in clinically important amounts.
Guidelines were based on a review of the literature
The guidelines were developed after a review of all scientific studies available on each topic and were published in the online issue of the journal Epilepsia.1-3 Their development was supported in part by the Milken Family Foundation.
“For too long, women living with epilepsy have feared the added risk of premature birth and other consequences of both their epilepsy and their medications,” said Howard R. Soule, PhD, chief science officer for the Milken Family Foundation. “The results of this project will help relieve the worries of these women and their families.”
Do not withdraw antiepileptic drugs during pregnancy
Some physicians attempt to discontinue an antiepileptic drug when a woman has gone 2 years without experiencing a seizure.5 In this scenario, the likelihood that seizures will recur within 6 and 12 months is 12% and 32%, respectively.6 Because of the risk that seizures will recur, and the increased likelihood of adverse outcomes associated with seizures during pregnancy, antiepileptic medication should not be discontinued during gestation.
Nor should a woman attempt to transition from one antiepileptic drug to another during pregnancy solely for the purpose of reducing teratogenicity.5 Doing so could precipitate seizures and exposes the fetus to the potentially hazardous effects of an additional antiseizure medication during a critical period. Moreover, there may be no advantage associated with switching drugs once a pregnancy has been established.7
Screen for malformations rigorously
Comprehensive screening for fetal anomalies early in the pregnancy is recommended for two main reasons:
- If a malformation is identified, the mother has the option of terminating the pregnancy
- Even if the patient decides not to terminate a gestation in which fetal anomaly has been identified, the information may help the practitioner determine the best mode and place of delivery.
Cesarean delivery may be warranted if the mother
has had recent seizures
Although most women who have epilepsy can expect to have a normal vaginal delivery, elective cesarean should be considered if the mother has experienced frequent seizures during the third trimester, or if she has a history of stress-related status epilepticus.5
Tonic-clonic seizures occur during labor in 1% to 2% of women who have epilepsy, and in an additional 1% to 2% of women in the 24 hours immediately following delivery.
Plasma levels of antiepileptic medication should be monitored during the third trimester and delivery to ensure that the medication is given in adequate strength to prevent seizures. In addition, the patient should be counseled about the importance of taking her medication consistently during this period.5
If the patient experiences a seizure during labor and delivery, treat her promptly with an intravenous benzodiazepine, preferably lorazepam.8 However, be aware that phenobarbital, primidone, and benzodiazepines remain in neonatal plasma for several days after delivery and may cause sedation or neonatal withdrawal syndrome.9
Do not give magnesium sulfate for epileptic seizures unless the seizures first appear during the third trimester or immediate postpartum period and could be associated with eclampsia. In such cases, treat the eclampsia and evaluate the patient for other potential causes of the seizures.5
1. Harden CL, Hopp J, Ting TY, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): I. Obstetrical complications and change in seizure frequency: Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1229-1236.
2. Harden CL, Meador KJ, Pennell PB, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): II. Teratogenesis and perinatal outcomes: Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1237-1246.
3. Harden CL, Pennell PB, Koppel BS, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): III. Vitamin K, folic acid, blood levels, and breast-feeding. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1247-1255.
4. Chen YH, Chiou HY, Lin HC, Lin HL. Affect of seizures during gestation on pregnancy outcomes in women with epilepsy. Arch Neurol. 2009;66:979-984.
5. Schachter SC. Management of epilepsy and pregnancy. ©2009. http://www.UpToDate.com. Available at: http://www.uptodate.com/patients/content/topic.do?topicKey=~JqqkRi44op3f4Y. Accessed Aug. 17, 2009.
6. EURAP study group. Seizure control and treatment in pregnancy: observations from the EURAP epilepsy pregnancy registry. Neurology. 2006;66:354-360.
7. Practice parameter: management issues for women with epilepsy (summary statement). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 1998;51:944-948.
8. Yerby MS. Problems and management of the pregnant woman with epilepsy. Epilepsia. 1987;28 Suppl 3:S29-S36.
9. Kuhnz W, Koch S, Helge H, Nau H. Primidone and phenobarbital during lactation period in epileptic women: total and free drug serum levels in the nursed infants and their effects on neonatal behavior. Dev Pharmacol Ther. 1988;11:147-154.
10. Artama M, Isojarvi JI, Raitanen J, Auvinen A. Birth rate among patients with epilepsy: a nationwide population-based cohort study in Finland. Am J Epidemiol. 2004;159:1057-1063.
11. Crawford PM. Managing epilepsy in women of childbearing age. Drug Saf. 2009;32:293-307.
12. Bauer J, Cooper-Mahkorn D. Reproductive dysfunction in women with epilepsy: menstrual cycle abnormalities, fertility, and polycystic ovary syndrome. Int Rev Neurobiol. 2008;83:135-155.
13. Dam M. Is epilepsy hereditary? Available at: http://www.epilepsy.dk/Handbook/Hereditary-uk.asp. Accessed Aug. 18, 2009.
1. Harden CL, Hopp J, Ting TY, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): I. Obstetrical complications and change in seizure frequency: Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1229-1236.
2. Harden CL, Meador KJ, Pennell PB, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): II. Teratogenesis and perinatal outcomes: Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1237-1246.
3. Harden CL, Pennell PB, Koppel BS, et al. Management issues for women with epilepsy—focus on pregnancy (evidence-based review): III. Vitamin K, folic acid, blood levels, and breast-feeding. Report of the Quality Standards Subcommittee and Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsia. 2009;50:1247-1255.
4. Chen YH, Chiou HY, Lin HC, Lin HL. Affect of seizures during gestation on pregnancy outcomes in women with epilepsy. Arch Neurol. 2009;66:979-984.
5. Schachter SC. Management of epilepsy and pregnancy. ©2009. http://www.UpToDate.com. Available at: http://www.uptodate.com/patients/content/topic.do?topicKey=~JqqkRi44op3f4Y. Accessed Aug. 17, 2009.
6. EURAP study group. Seizure control and treatment in pregnancy: observations from the EURAP epilepsy pregnancy registry. Neurology. 2006;66:354-360.
7. Practice parameter: management issues for women with epilepsy (summary statement). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 1998;51:944-948.
8. Yerby MS. Problems and management of the pregnant woman with epilepsy. Epilepsia. 1987;28 Suppl 3:S29-S36.
9. Kuhnz W, Koch S, Helge H, Nau H. Primidone and phenobarbital during lactation period in epileptic women: total and free drug serum levels in the nursed infants and their effects on neonatal behavior. Dev Pharmacol Ther. 1988;11:147-154.
10. Artama M, Isojarvi JI, Raitanen J, Auvinen A. Birth rate among patients with epilepsy: a nationwide population-based cohort study in Finland. Am J Epidemiol. 2004;159:1057-1063.
11. Crawford PM. Managing epilepsy in women of childbearing age. Drug Saf. 2009;32:293-307.
12. Bauer J, Cooper-Mahkorn D. Reproductive dysfunction in women with epilepsy: menstrual cycle abnormalities, fertility, and polycystic ovary syndrome. Int Rev Neurobiol. 2008;83:135-155.
13. Dam M. Is epilepsy hereditary? Available at: http://www.epilepsy.dk/Handbook/Hereditary-uk.asp. Accessed Aug. 18, 2009.
UPDATE ON TECHNOLOGY: VESSEL-SEALING DEVICES
When Harry Reich performed the first laparoscopically assisted vaginal hysterectomy in 1989, he advocated the use of sutures for control of the uterine vessels. Monopolar, bipolar, and laser instruments available at that time were inherently risky to use along the pelvic sidewall because of their potential for 1) considerable thermal energy spread beyond the area of treatment (bipolar, NG:YAG laser, monopolar) and 2) unreliable hemostasis (CO2 laser).
Minimally invasive surgical practice has driven meaningful advances in instrumentation and technique over the past 20 years. The constraints inherent in laparoscopic surgery, although somewhat mitigated by robotics, have generated a proliferation of technologies to obtain reliable hemostasis. Every device now on the market claims to “seal” vessels. In this article, I review the mechanism of action of these instruments and compare their strengths and weaknesses, based on high-quality scientific evidence.
Two studies highlight vessel ligation
Newcomb WL, Hope WW, Schmeizer TM, et al. Comparison of blood vessel sealing among new electrosurgical and ultrasonic devices. Surg Endosc. 2009;23:90–96.
Lamberton GR, Hsi RS, Jin DH, et al. Prospective comparison of four laparoscopic vessel ligation devices. J Endourol. 2008;22:2307–2312.
Many energy-delivery systems are available for the gynecologic surgeon; any of them can be used effectively and safely under most circumstances. Before we can make an informed choice about which system is best for our own practice, however, we need to be aware of the strengths and limitations of the systems overall ( TABLE ).
These two studies focus on the following devices:
- Gyrus PK Tissue Management System, PKS Cutting Forceps, and Plasma Trissector (all from Gyrus Medical). These are bipolar electrosurgical devices designed to deliver high current and very low voltage to tissue. Tissue impedance is continuously monitored between the jaws of the instrument, and energy delivery is adjusted accordingly. These systems deliver electrosurgical energy through a series of rapid pulses, thereby allowing the tissue to cool briefly and limiting the heating of adjacent tissue. Protein in the vessel walls is denatured and forms a coagulum, which occludes the lumen.
- Harmonic Scalpel (Ethicon EndoSurgery). This device uses a high-frequency ultrasonic transducer (55,000 cycles/second) to create mechanical vibration of one of the two jaws. The device can be used to vaporize tissue (cut) or achieve hemostasis by coagulation. As with the other devices, protein is denatured and vessels are occluded by formation of a coagulum.
- Ligamax 5 Endoscopic Multiple Clip Applier (Ethicon). This device is a sterile, single-patient-use, 5-mm, endoscopic, multiple-clip applicator that delivers 15 medium or large titanium clips that close to 8.8 mm, the same clip size as the 10-mm applicator. Ligamax 5 has long, thin angled jaws (8.4 mm) that extend beyond vessels and ducts to improve visibility. It includes a long, 33-cm shaft for additional reach, and an anti-clip drop-ratchet mechanism for control over clip closure.
- EnSeal Tissue Sealing and Hemostasis System and EnSeal PTC (SurgRx). EnSeal utilizes nanotechnology to control the energy at the electrode–tissue interface. The jaws contain a temperature-sensitive matrix with embedded conductive carbon spherules designed to “sense” tissue characteristics. It uses extremely high jaw compression to create uniform tissue effects. It does not require a dedicated electrosurgical unit for use; an adapter can be purchased that permits use with most generators.
- LigaSure V (Valleylab). LigaSure is a bipolar electrosurgical device designed to deliver high current and very low voltage to tissue. It monitors tissue impedance between the jaws of the instrument and continuously adjusts the delivery of energy.
TABLE
Rating vessel-sealing devices: 5 measures of success
| Device | Safety: Minimal thermal spread | Reliability: Efficacy on vessels ≤7 mm | Efficiency: Treatment time | Consistency: Independent of user | Utility: Multiple uses |
|---|---|---|---|---|---|
| Harmonic Scalpel | Excellent | Poor | Excellent | Poor | Excellent |
| Gyrus PK | Poor | Poor | Excellent | Fair | Fair |
| LigaSure V | Good | Excellent | Good | Excellent | Fair |
| EnSeal | Fair | Excellent | Poor | Excellent | Poor |
| These ratings were devised by the author based on data from independent studies in living tissue models. | |||||
FIGURE How energy-based vessel-sealing works
(A) When the carotid artery of an animal model was sealed using a standard bipolar forceps, the lumen remained open and a proximal thrombus developed. (B) When LigaSure was used, the artery was fused and the lumen obliterated with one 5-second application. (C and D) When a renal artery was sealed with LigaSure, the lumen of the vessel walls fused completely.
What the studies found
Newcomb and associates compared blood-vessel-sealing ability of the following devices:
- Gyrus 5-mm PKS Cutting Forceps
- Gyrus Plasma Trissector
- Harmonic Scalpel
- EnSeal Tissue Sealing and Hemostasis System
- LigaSure V, using the LigaSure Generator (Valleylab)
- LigaSure V, using the Force Triad Generator (Valleylab)
- Ligamax 5 Endoscopic Multiple Clip Applier.
The authors assessed mean seal times and burst pressures. Used on medium and large vessels, the Harmonic Scalpel and Gyrus products had significant failure rates: 8% to 22% for the Harmonic Scalpel and 41% to 92% for the pulsed, bipolar Gyrus systems.
The shortest sealing times for medium to large vessels were achieved with the LigaSure V using the Force Triad Generator. The Gyrus systems were the fastest devices when vessels were 2 to 3 mm in diameter.
There were no seal failures with the Ligamax, EnSeal, or LigaSure products.
Second study involved repeated applications
Lamberton and colleagues tested 5-mm laparoscopic devices under controlled temperature and humidity using a laparoscopic simulator, focusing on the following systems:
- LigaSure V
- Gyrus PK
- Harmonic Scalpel
- EnSeal Tissue Sealing and Hemostasis System.
The authors used 5-mm bovine arteries to assess sealing time, burst pressure, lateral thermal spread, and smoke production, as well as both subjective and objective effects on visibility.
Each device was used 10 times to determine burst pressure, lateral thermal spread, visibility, and smoke production. The devices were applied 20 times to measure time to seal, based on the devices’ preprogrammed endpoints.
The Harmonic Scalpel produced the lowest thermal spread and least smoke, but also had the lowest mean burst pressure.
The Gyrus PK generated the most smoke and had variable burst pressure. Although it had the fastest sealing times, in three of 10 trials there was a completely open arterial lumen following transection. In addition, 50% of applications involved burst pressures below 50 mm Hg.
Maximum temperatures 2 mm from the device were 49.9°C for the Harmonic Scalpel, 55.5°C for LigaSure, 58.9°C for EnSeal, and 64.5°C for Gyrus PK.
LigaSure was the highest-rated device overall, with the highest burst pressure and fastest sealing time.
EnSeal was the slowest and had variable burst pressures.
Clinical implications of the trials
These studies—neither of which was industry-sponsored—suggest that larger vessels cannot be controlled consistently and effectively using the Harmonic Scalpel or Gyrus systems. The Harmonic Scalpel is most user-dependent.
So what’s the bottom line? In the end, according to Andrew Brill, MD, past president of AAGL, “it’s not the wand, it’s the magician.”
for laparoscopic surgery
When OBG Management surveyed a number of laparoscopic experts and members of our Virtual Board of Editors about their preferred energy sources for laparoscopic surgery, the responses were strikingly similar. The consensus? The best device depends on the case at hand, the skill of the surgeon, economic concerns, and other variables.
As Keith Isaacson, MD, put it, “Because tissue may be thick or thin, moist or desiccated, vascular or avascular, the ideal instrument to achieve a pure cut varies. Because a vessel may be large or small, contain a large amount of collagen or very little collagen, or be on tension or relaxed, the ideal instrument for vessel sealing also varies, depending on the surgical situation.”
All of the devices are roughly equivalent, he added. “Fortunately, almost all of the commercially available energy sources that utilize bipolar radiofrequency or ultrasonic energy will perform our desired function if the surgeon understands the technology and utilizes the instruments properly.”
Here is a summary of recommendations made by the experts we interviewed.
Andrew I. Brill, MD
Director of Minimally Invasive Gynecology,
California Pacific Medical Center,
San Francisco, Calif
Having been actively engaged in advanced laparoscopic surgical training for more than 20 years, I have extensive experience with all of these novel devices. I critically assess any new energy-based device for its ergonomic handedness, propensity for sticking to tissue and production of plume, ability to manipulate and dissect tissue, efficiency in desiccated or fatty tissues, discoloration of tissue by carbon, response to tissue tension, and reliability for hemostasis. Because no device is perfectly suited to all procedures, I customarily rely upon several devices to satisfy my technical needs.
All advanced bipolar energy devices—LigaSure, PK Cutting Forceps, and EnSeal—can be safely used to coagulate and cut all vascular pedicles during hysterectomy and salpingo-oophorectomy. These devices perform best when tissue tension is reduced to maximize vessel sealing. Despite the fact that these devices provide audible feedback to signal the electrosurgical endpoint, I also gauge tissue color, retraction, and the emission of steam before advancing the cutting blade.
The tapered-tip design of the PK Cutting Forceps offers some comparative advantage for fine tissue dissection, but I find that hemostasis is more consistently achieved with less thermal spread using the EnSeal or LigaSure device.
I commonly utilize the Harmonic Scalpel in lieu of any electrosurgical device, understanding that it requires more finesse to achieve equivalent results.
When I anticipate that I will need to manipulate uterine pedicles during a difficult laparoscopic hysterectomy, I employ electrosurgery using the EnSeal device for its superior tissue sealing. A 3-mm curved device by EnSeal, akin to a Pean clamp, works as an exceptionally agile instrument for hysterectomy without sacrificing hemostasis.
Only the LigaSure Advance provides both coaptive coagulation and spark cutting via an electrode on the tip of one blade; therefore, an adjunctive mechanical or energy-based device must be employed to perform culdotomy during total laparoscopic hysterectomy when EnSeal or the PK Cutting Forceps is used.
The ability to efficiently and hemostatically cut through tissue of variable mass with minimal plume, predictable thermal margins, and the retention of tissue color make the Harmonic Scalpel my first choice for laparoscopic resection of endometriosis, extensive adhesiolysis, myomectomy, cervical amputation during supracervical hysterectomy, and culdotomy during total hysterectomy. Although I have similarly utilized the unique capacity of the spatula, J-hook, and needle electrodes by Plasmacision (Gyrus) to cut tissue using bipolar electrosurgery, all have afforded less technical control, not as much hemostasis, and wider thermal margins.
Use of any energy-based device does not preclude the need for skill. Before adding any of these devices to your surgical armamentarium, appropriate training should be acquired in a skills lab using living tissue or with a laparoscopic trainer using a tissue surrogate.
Dr. Brill reports no financial relationships relevant to this review.
Jon I. Einarsson, MD, MPH
Director of Minimally Invasive Gynecologic Surgery,
Brigham and Women’s Hospital,
Assistant Professor of Obstetrics,
Gynecology, and Reproductive Biology,
Harvard Medical School, Boston, Mass
I prefer the Harmonic Scalpel and a Kelly RoBi Bipolar Grasper (Karl Storz). I have been using these instruments for approximately 10 years. I use this combination for all operative laparoscopy cases, including hysterectomy, myomectomy, sacrocolpopexy, resection of endometriosis, and lysis of adhesions.
The Harmonic Scalpel is a versatile instrument that can perform dissection, cutting, and coagulation very efficiently. However, there are better vessel sealers out there, such as the LigaSure and EnSeal devices. In this era of cost containment, it is important to limit the use of multiple disposable energy sources in a single case. By adding the reusable bipolar grasper, vessel sealing becomes efficient and reliable without added cost. In addition, the bipolar grasper functions as a dissector and retractor, thereby minimizing instrument changes.
I watch the bubbles that form during desiccation with the bipolar grasper. Once these bubbles start to “die down,” I know that the tissue is desiccated and safe to transect. It is important to remember that thermal spread with a traditional bipolar grasper is significant (up to 13 mm). For that reason, when I desiccate the uterine vessels with the bipolar grasper, I stay above the rim of the colpotomy ring in order to maintain a safe distance from the ureter during this step.
The Harmonic Scalpel offers several advantages over monopolar electrosurgery for cutting, such as decreased smoke formation and avoidance of complications such as insulation failure, direct coupling, and capacitative coupling.
Dr. Einarsson reports no financial relationships relevant to this review.
Carl F. Giesler, MD
Associate Professor and Director of
Minimally Invasive Surgery,
Department of Obstetrics and Gynecology,
Baylor College of Medicine,
Waco, Tex
I was introduced to laparoscopy as a resident in 1974, and have been teaching laparoscopic surgical techniques to general surgeons and gynecologists since 1988. I began performing laparoscopic hysterectomy in 1991. As an educator and trainer of other laparoscopic surgeons, I have used all available laparoscopic energy sources.
I became an early adapter and user of ultrasonic mechanical energy when it was introduced for laparoscopy in 1995, because of its safety for adjacent tissues and its single-instrument, multiple-task properties.
This energy source was originally named the Ultrasonic Coagulating Shears by Ultracision, but Ethicon EndoSurgery purchased the product in 1995 and changed its name to the Harmonic Scalpel. The current generation is called the Harmonic ACE, and I use it as my primary laparoscopic energy source for all procedures, from management of unruptured ectopic pregnancy to total laparoscopic hysterectomy and extensive adhesiolyis in previously operated abdomens.
The Harmonic ACE allows me to confidently dissect adhesions over loops of bowel, as well as over the ureter, because of its limited lateral thermal spread. It allows nearly bloodless dissection of the retroperitoneal space and space around the bladder. The cavitation effect created by the rapidly moving instrument tip (55,500 cycles/second) creates tissue separation along the areolar tissue planes, where most blood vessels are the size of capillaries and where larger blood vessels are easily identified and controlled by coaptation, coagulation, and cutting.
The protein coagulum is produced at a temperature of 54°C. The sides and back of the active blade have minimal thermal effect because tissue is not held tightly against the moving blade. Minimal char is observed because of the low tissue temperature associated with coagulum formation. Minimal sticking of tissue occurs because the rapid motion of the instrument tip dislodges the tissue.
The ease of use, multiple functions for a single instrument, tissue safety, and minimal residual traumatized tissue are the reasons I prefer the mechanical energy of the Harmonic ACE as my primary laparoscopic energy source.
Dr. Giesler serves as a speaker for Ethicon EndoSurgery.
Cheryl Iglesia, MD
Director, Urogynecology and Reconstructive Pelvic Surgery,
Washington Hospital Center,
Associate Professor, Departments of ObGyn and Urology,
Georgetown University, Washington, DC.
Member, OBG Management Board of Editors
I prefer the Harmonic ACE for straight-stick laparoscopic hysterectomy cases and the PK Cutting Forceps for robotic hysterectomy and sacrocolpopexy.
The Harmonic ACE is fast and has little thermal spread; the PK Cutting Forceps is the only energy source available with reticulating arms for the robot.
I have been using the Harmonic ACE for more than 10 years and the PK Cutting Forceps since 2006.
Dr. Iglesia reports no financial relationships relevant to this review.
Keith Isaacson, MD
Director of Minimally Invasive Gynecologic
Surgery and Infertility,
Director of the AAGL/SRS
Fellowship in Minimally Invasive Gynecologic Surgery,
Newton Wellesley Hospital,
Associate Professor of Obstetrics, Gynecology,
and Reproductive Biology,
Harvard Medical School, Boston, Mass
I have used every bipolar and harmonic tool that is commercially available, including the recently released EnSeal. I like them all. The tools I have chosen were selected in conjunction with our purchasing department and determined to be the optimal tools for the lowest cost and highest efficiency. Our goal is to use as few disposables as possible, but to be as efficient as possible.
The instruments I utilize most frequently are the reusable 5-mm bipolar instruments from Karl Storz (RoBi) and ERBE Medical. The RoBi set includes a variety of tips, ranging from a dissecting Maryland-type tip to a flat, broad-based tip similar to the Kleppinger design. These are all plugged into the ERBE VIO generator. With this assortment of tools, I can seal a vessel of almost any size, whether it be thin-walled, such as a large vein, or a thick-walled artery.
To cut tissue, I prefer the Harmonic Scalpel and the bipolar Laparoscopic Spatula (Gyrus). I use the Harmonic Scalpel during laparoscopic hysterectomy, and the Spatula during laparoscopic myomectomy. Both devices cut tissue well while simultaneously sealing small vessels.
I have been using these instruments for approximately 10 years.
Dr. Isaacson reports that he is a consultant for Karl Storz in hysteroscopy.
Noor Ahmed, MB, ChB, MD
Consultant Gynecologist,
Dundee, Scotland.
Member, OBG Management Virtual
Board of Editors
I am a consultant gynecologist with an interest in fertility, endometriosis surgery, and general gynecologic surgery. Most of my surgical work is performed laparoscopically or vaginally.
My first experience with vessel-sealing technology was in 2000, when I began using the LigaSure Max for difficult vaginal hysterectomies. I was impressed by its ability to clamp and seal and, in later versions, its ability to cut, as well. Since then, I have been using various versions of LigaSure vessel- and tissue-sealing technology for many gynecologic procedures.
I have had some exposure to other vessel- and tissue-sealing technologies, but prefer LigaSure because:
1. It provides an experience similar to that of conventional clamp, cut, and suture through the control offered by its unique combination of pressure and energy, creating vessel and tissue fusion that does not rely on a proximal thrombus and produces minimal thermal spread.
2. The feedback-controlled response system automatically discontinues energy delivery when sealing is complete.
3. It works with clamps of various sizes and shapes, using the same hardware (generator) and saving money and space. Moreover, the nursing staff has to develop familiarity with one machine only.
For laparoscopic procedures, I use LigaSure Lap, LigaSure Atlas Sealer/Divider and, most commonly, the 5-mm LigaSure V Sealer/Divider.
Dr. Ahmed reports no financial relationships relevant to this review.
Michael Kirwin, MD
General Obstetrics and Gynecology, Freehold, NJ.
Member, OBG Management Virtual Board of Editors
With advanced laparoscopic cases, particularly hysterectomy, I use the Harmonic Scalpel and PK Cutting Forceps. I have used the Harmonic Scalpel regularly for approximately 4 years, and it works well for dissection and, particularly, for transecting the cervix in supracervical cases.
Over the past year, I have also begun to use the Gyrus forceps, especially when I expect to encounter larger vessels that require coagulation. Both instruments can be used in 5-mm ports. Both seem relatively reliable and easy for the OR team to use. I would hope that an articulation feature can be added in the future.
Dr. Kirwin reports no financial relationships relevant to this review.
When Harry Reich performed the first laparoscopically assisted vaginal hysterectomy in 1989, he advocated the use of sutures for control of the uterine vessels. Monopolar, bipolar, and laser instruments available at that time were inherently risky to use along the pelvic sidewall because of their potential for 1) considerable thermal energy spread beyond the area of treatment (bipolar, NG:YAG laser, monopolar) and 2) unreliable hemostasis (CO2 laser).
Minimally invasive surgical practice has driven meaningful advances in instrumentation and technique over the past 20 years. The constraints inherent in laparoscopic surgery, although somewhat mitigated by robotics, have generated a proliferation of technologies to obtain reliable hemostasis. Every device now on the market claims to “seal” vessels. In this article, I review the mechanism of action of these instruments and compare their strengths and weaknesses, based on high-quality scientific evidence.
Two studies highlight vessel ligation
Newcomb WL, Hope WW, Schmeizer TM, et al. Comparison of blood vessel sealing among new electrosurgical and ultrasonic devices. Surg Endosc. 2009;23:90–96.
Lamberton GR, Hsi RS, Jin DH, et al. Prospective comparison of four laparoscopic vessel ligation devices. J Endourol. 2008;22:2307–2312.
Many energy-delivery systems are available for the gynecologic surgeon; any of them can be used effectively and safely under most circumstances. Before we can make an informed choice about which system is best for our own practice, however, we need to be aware of the strengths and limitations of the systems overall ( TABLE ).
These two studies focus on the following devices:
- Gyrus PK Tissue Management System, PKS Cutting Forceps, and Plasma Trissector (all from Gyrus Medical). These are bipolar electrosurgical devices designed to deliver high current and very low voltage to tissue. Tissue impedance is continuously monitored between the jaws of the instrument, and energy delivery is adjusted accordingly. These systems deliver electrosurgical energy through a series of rapid pulses, thereby allowing the tissue to cool briefly and limiting the heating of adjacent tissue. Protein in the vessel walls is denatured and forms a coagulum, which occludes the lumen.
- Harmonic Scalpel (Ethicon EndoSurgery). This device uses a high-frequency ultrasonic transducer (55,000 cycles/second) to create mechanical vibration of one of the two jaws. The device can be used to vaporize tissue (cut) or achieve hemostasis by coagulation. As with the other devices, protein is denatured and vessels are occluded by formation of a coagulum.
- Ligamax 5 Endoscopic Multiple Clip Applier (Ethicon). This device is a sterile, single-patient-use, 5-mm, endoscopic, multiple-clip applicator that delivers 15 medium or large titanium clips that close to 8.8 mm, the same clip size as the 10-mm applicator. Ligamax 5 has long, thin angled jaws (8.4 mm) that extend beyond vessels and ducts to improve visibility. It includes a long, 33-cm shaft for additional reach, and an anti-clip drop-ratchet mechanism for control over clip closure.
- EnSeal Tissue Sealing and Hemostasis System and EnSeal PTC (SurgRx). EnSeal utilizes nanotechnology to control the energy at the electrode–tissue interface. The jaws contain a temperature-sensitive matrix with embedded conductive carbon spherules designed to “sense” tissue characteristics. It uses extremely high jaw compression to create uniform tissue effects. It does not require a dedicated electrosurgical unit for use; an adapter can be purchased that permits use with most generators.
- LigaSure V (Valleylab). LigaSure is a bipolar electrosurgical device designed to deliver high current and very low voltage to tissue. It monitors tissue impedance between the jaws of the instrument and continuously adjusts the delivery of energy.
TABLE
Rating vessel-sealing devices: 5 measures of success
| Device | Safety: Minimal thermal spread | Reliability: Efficacy on vessels ≤7 mm | Efficiency: Treatment time | Consistency: Independent of user | Utility: Multiple uses |
|---|---|---|---|---|---|
| Harmonic Scalpel | Excellent | Poor | Excellent | Poor | Excellent |
| Gyrus PK | Poor | Poor | Excellent | Fair | Fair |
| LigaSure V | Good | Excellent | Good | Excellent | Fair |
| EnSeal | Fair | Excellent | Poor | Excellent | Poor |
| These ratings were devised by the author based on data from independent studies in living tissue models. | |||||
FIGURE How energy-based vessel-sealing works
(A) When the carotid artery of an animal model was sealed using a standard bipolar forceps, the lumen remained open and a proximal thrombus developed. (B) When LigaSure was used, the artery was fused and the lumen obliterated with one 5-second application. (C and D) When a renal artery was sealed with LigaSure, the lumen of the vessel walls fused completely.
What the studies found
Newcomb and associates compared blood-vessel-sealing ability of the following devices:
- Gyrus 5-mm PKS Cutting Forceps
- Gyrus Plasma Trissector
- Harmonic Scalpel
- EnSeal Tissue Sealing and Hemostasis System
- LigaSure V, using the LigaSure Generator (Valleylab)
- LigaSure V, using the Force Triad Generator (Valleylab)
- Ligamax 5 Endoscopic Multiple Clip Applier.
The authors assessed mean seal times and burst pressures. Used on medium and large vessels, the Harmonic Scalpel and Gyrus products had significant failure rates: 8% to 22% for the Harmonic Scalpel and 41% to 92% for the pulsed, bipolar Gyrus systems.
The shortest sealing times for medium to large vessels were achieved with the LigaSure V using the Force Triad Generator. The Gyrus systems were the fastest devices when vessels were 2 to 3 mm in diameter.
There were no seal failures with the Ligamax, EnSeal, or LigaSure products.
Second study involved repeated applications
Lamberton and colleagues tested 5-mm laparoscopic devices under controlled temperature and humidity using a laparoscopic simulator, focusing on the following systems:
- LigaSure V
- Gyrus PK
- Harmonic Scalpel
- EnSeal Tissue Sealing and Hemostasis System.
The authors used 5-mm bovine arteries to assess sealing time, burst pressure, lateral thermal spread, and smoke production, as well as both subjective and objective effects on visibility.
Each device was used 10 times to determine burst pressure, lateral thermal spread, visibility, and smoke production. The devices were applied 20 times to measure time to seal, based on the devices’ preprogrammed endpoints.
The Harmonic Scalpel produced the lowest thermal spread and least smoke, but also had the lowest mean burst pressure.
The Gyrus PK generated the most smoke and had variable burst pressure. Although it had the fastest sealing times, in three of 10 trials there was a completely open arterial lumen following transection. In addition, 50% of applications involved burst pressures below 50 mm Hg.
Maximum temperatures 2 mm from the device were 49.9°C for the Harmonic Scalpel, 55.5°C for LigaSure, 58.9°C for EnSeal, and 64.5°C for Gyrus PK.
LigaSure was the highest-rated device overall, with the highest burst pressure and fastest sealing time.
EnSeal was the slowest and had variable burst pressures.
Clinical implications of the trials
These studies—neither of which was industry-sponsored—suggest that larger vessels cannot be controlled consistently and effectively using the Harmonic Scalpel or Gyrus systems. The Harmonic Scalpel is most user-dependent.
So what’s the bottom line? In the end, according to Andrew Brill, MD, past president of AAGL, “it’s not the wand, it’s the magician.”
for laparoscopic surgery
When OBG Management surveyed a number of laparoscopic experts and members of our Virtual Board of Editors about their preferred energy sources for laparoscopic surgery, the responses were strikingly similar. The consensus? The best device depends on the case at hand, the skill of the surgeon, economic concerns, and other variables.
As Keith Isaacson, MD, put it, “Because tissue may be thick or thin, moist or desiccated, vascular or avascular, the ideal instrument to achieve a pure cut varies. Because a vessel may be large or small, contain a large amount of collagen or very little collagen, or be on tension or relaxed, the ideal instrument for vessel sealing also varies, depending on the surgical situation.”
All of the devices are roughly equivalent, he added. “Fortunately, almost all of the commercially available energy sources that utilize bipolar radiofrequency or ultrasonic energy will perform our desired function if the surgeon understands the technology and utilizes the instruments properly.”
Here is a summary of recommendations made by the experts we interviewed.
Andrew I. Brill, MD
Director of Minimally Invasive Gynecology,
California Pacific Medical Center,
San Francisco, Calif
Having been actively engaged in advanced laparoscopic surgical training for more than 20 years, I have extensive experience with all of these novel devices. I critically assess any new energy-based device for its ergonomic handedness, propensity for sticking to tissue and production of plume, ability to manipulate and dissect tissue, efficiency in desiccated or fatty tissues, discoloration of tissue by carbon, response to tissue tension, and reliability for hemostasis. Because no device is perfectly suited to all procedures, I customarily rely upon several devices to satisfy my technical needs.
All advanced bipolar energy devices—LigaSure, PK Cutting Forceps, and EnSeal—can be safely used to coagulate and cut all vascular pedicles during hysterectomy and salpingo-oophorectomy. These devices perform best when tissue tension is reduced to maximize vessel sealing. Despite the fact that these devices provide audible feedback to signal the electrosurgical endpoint, I also gauge tissue color, retraction, and the emission of steam before advancing the cutting blade.
The tapered-tip design of the PK Cutting Forceps offers some comparative advantage for fine tissue dissection, but I find that hemostasis is more consistently achieved with less thermal spread using the EnSeal or LigaSure device.
I commonly utilize the Harmonic Scalpel in lieu of any electrosurgical device, understanding that it requires more finesse to achieve equivalent results.
When I anticipate that I will need to manipulate uterine pedicles during a difficult laparoscopic hysterectomy, I employ electrosurgery using the EnSeal device for its superior tissue sealing. A 3-mm curved device by EnSeal, akin to a Pean clamp, works as an exceptionally agile instrument for hysterectomy without sacrificing hemostasis.
Only the LigaSure Advance provides both coaptive coagulation and spark cutting via an electrode on the tip of one blade; therefore, an adjunctive mechanical or energy-based device must be employed to perform culdotomy during total laparoscopic hysterectomy when EnSeal or the PK Cutting Forceps is used.
The ability to efficiently and hemostatically cut through tissue of variable mass with minimal plume, predictable thermal margins, and the retention of tissue color make the Harmonic Scalpel my first choice for laparoscopic resection of endometriosis, extensive adhesiolysis, myomectomy, cervical amputation during supracervical hysterectomy, and culdotomy during total hysterectomy. Although I have similarly utilized the unique capacity of the spatula, J-hook, and needle electrodes by Plasmacision (Gyrus) to cut tissue using bipolar electrosurgery, all have afforded less technical control, not as much hemostasis, and wider thermal margins.
Use of any energy-based device does not preclude the need for skill. Before adding any of these devices to your surgical armamentarium, appropriate training should be acquired in a skills lab using living tissue or with a laparoscopic trainer using a tissue surrogate.
Dr. Brill reports no financial relationships relevant to this review.
Jon I. Einarsson, MD, MPH
Director of Minimally Invasive Gynecologic Surgery,
Brigham and Women’s Hospital,
Assistant Professor of Obstetrics,
Gynecology, and Reproductive Biology,
Harvard Medical School, Boston, Mass
I prefer the Harmonic Scalpel and a Kelly RoBi Bipolar Grasper (Karl Storz). I have been using these instruments for approximately 10 years. I use this combination for all operative laparoscopy cases, including hysterectomy, myomectomy, sacrocolpopexy, resection of endometriosis, and lysis of adhesions.
The Harmonic Scalpel is a versatile instrument that can perform dissection, cutting, and coagulation very efficiently. However, there are better vessel sealers out there, such as the LigaSure and EnSeal devices. In this era of cost containment, it is important to limit the use of multiple disposable energy sources in a single case. By adding the reusable bipolar grasper, vessel sealing becomes efficient and reliable without added cost. In addition, the bipolar grasper functions as a dissector and retractor, thereby minimizing instrument changes.
I watch the bubbles that form during desiccation with the bipolar grasper. Once these bubbles start to “die down,” I know that the tissue is desiccated and safe to transect. It is important to remember that thermal spread with a traditional bipolar grasper is significant (up to 13 mm). For that reason, when I desiccate the uterine vessels with the bipolar grasper, I stay above the rim of the colpotomy ring in order to maintain a safe distance from the ureter during this step.
The Harmonic Scalpel offers several advantages over monopolar electrosurgery for cutting, such as decreased smoke formation and avoidance of complications such as insulation failure, direct coupling, and capacitative coupling.
Dr. Einarsson reports no financial relationships relevant to this review.
Carl F. Giesler, MD
Associate Professor and Director of
Minimally Invasive Surgery,
Department of Obstetrics and Gynecology,
Baylor College of Medicine,
Waco, Tex
I was introduced to laparoscopy as a resident in 1974, and have been teaching laparoscopic surgical techniques to general surgeons and gynecologists since 1988. I began performing laparoscopic hysterectomy in 1991. As an educator and trainer of other laparoscopic surgeons, I have used all available laparoscopic energy sources.
I became an early adapter and user of ultrasonic mechanical energy when it was introduced for laparoscopy in 1995, because of its safety for adjacent tissues and its single-instrument, multiple-task properties.
This energy source was originally named the Ultrasonic Coagulating Shears by Ultracision, but Ethicon EndoSurgery purchased the product in 1995 and changed its name to the Harmonic Scalpel. The current generation is called the Harmonic ACE, and I use it as my primary laparoscopic energy source for all procedures, from management of unruptured ectopic pregnancy to total laparoscopic hysterectomy and extensive adhesiolyis in previously operated abdomens.
The Harmonic ACE allows me to confidently dissect adhesions over loops of bowel, as well as over the ureter, because of its limited lateral thermal spread. It allows nearly bloodless dissection of the retroperitoneal space and space around the bladder. The cavitation effect created by the rapidly moving instrument tip (55,500 cycles/second) creates tissue separation along the areolar tissue planes, where most blood vessels are the size of capillaries and where larger blood vessels are easily identified and controlled by coaptation, coagulation, and cutting.
The protein coagulum is produced at a temperature of 54°C. The sides and back of the active blade have minimal thermal effect because tissue is not held tightly against the moving blade. Minimal char is observed because of the low tissue temperature associated with coagulum formation. Minimal sticking of tissue occurs because the rapid motion of the instrument tip dislodges the tissue.
The ease of use, multiple functions for a single instrument, tissue safety, and minimal residual traumatized tissue are the reasons I prefer the mechanical energy of the Harmonic ACE as my primary laparoscopic energy source.
Dr. Giesler serves as a speaker for Ethicon EndoSurgery.
Cheryl Iglesia, MD
Director, Urogynecology and Reconstructive Pelvic Surgery,
Washington Hospital Center,
Associate Professor, Departments of ObGyn and Urology,
Georgetown University, Washington, DC.
Member, OBG Management Board of Editors
I prefer the Harmonic ACE for straight-stick laparoscopic hysterectomy cases and the PK Cutting Forceps for robotic hysterectomy and sacrocolpopexy.
The Harmonic ACE is fast and has little thermal spread; the PK Cutting Forceps is the only energy source available with reticulating arms for the robot.
I have been using the Harmonic ACE for more than 10 years and the PK Cutting Forceps since 2006.
Dr. Iglesia reports no financial relationships relevant to this review.
Keith Isaacson, MD
Director of Minimally Invasive Gynecologic
Surgery and Infertility,
Director of the AAGL/SRS
Fellowship in Minimally Invasive Gynecologic Surgery,
Newton Wellesley Hospital,
Associate Professor of Obstetrics, Gynecology,
and Reproductive Biology,
Harvard Medical School, Boston, Mass
I have used every bipolar and harmonic tool that is commercially available, including the recently released EnSeal. I like them all. The tools I have chosen were selected in conjunction with our purchasing department and determined to be the optimal tools for the lowest cost and highest efficiency. Our goal is to use as few disposables as possible, but to be as efficient as possible.
The instruments I utilize most frequently are the reusable 5-mm bipolar instruments from Karl Storz (RoBi) and ERBE Medical. The RoBi set includes a variety of tips, ranging from a dissecting Maryland-type tip to a flat, broad-based tip similar to the Kleppinger design. These are all plugged into the ERBE VIO generator. With this assortment of tools, I can seal a vessel of almost any size, whether it be thin-walled, such as a large vein, or a thick-walled artery.
To cut tissue, I prefer the Harmonic Scalpel and the bipolar Laparoscopic Spatula (Gyrus). I use the Harmonic Scalpel during laparoscopic hysterectomy, and the Spatula during laparoscopic myomectomy. Both devices cut tissue well while simultaneously sealing small vessels.
I have been using these instruments for approximately 10 years.
Dr. Isaacson reports that he is a consultant for Karl Storz in hysteroscopy.
Noor Ahmed, MB, ChB, MD
Consultant Gynecologist,
Dundee, Scotland.
Member, OBG Management Virtual
Board of Editors
I am a consultant gynecologist with an interest in fertility, endometriosis surgery, and general gynecologic surgery. Most of my surgical work is performed laparoscopically or vaginally.
My first experience with vessel-sealing technology was in 2000, when I began using the LigaSure Max for difficult vaginal hysterectomies. I was impressed by its ability to clamp and seal and, in later versions, its ability to cut, as well. Since then, I have been using various versions of LigaSure vessel- and tissue-sealing technology for many gynecologic procedures.
I have had some exposure to other vessel- and tissue-sealing technologies, but prefer LigaSure because:
1. It provides an experience similar to that of conventional clamp, cut, and suture through the control offered by its unique combination of pressure and energy, creating vessel and tissue fusion that does not rely on a proximal thrombus and produces minimal thermal spread.
2. The feedback-controlled response system automatically discontinues energy delivery when sealing is complete.
3. It works with clamps of various sizes and shapes, using the same hardware (generator) and saving money and space. Moreover, the nursing staff has to develop familiarity with one machine only.
For laparoscopic procedures, I use LigaSure Lap, LigaSure Atlas Sealer/Divider and, most commonly, the 5-mm LigaSure V Sealer/Divider.
Dr. Ahmed reports no financial relationships relevant to this review.
Michael Kirwin, MD
General Obstetrics and Gynecology, Freehold, NJ.
Member, OBG Management Virtual Board of Editors
With advanced laparoscopic cases, particularly hysterectomy, I use the Harmonic Scalpel and PK Cutting Forceps. I have used the Harmonic Scalpel regularly for approximately 4 years, and it works well for dissection and, particularly, for transecting the cervix in supracervical cases.
Over the past year, I have also begun to use the Gyrus forceps, especially when I expect to encounter larger vessels that require coagulation. Both instruments can be used in 5-mm ports. Both seem relatively reliable and easy for the OR team to use. I would hope that an articulation feature can be added in the future.
Dr. Kirwin reports no financial relationships relevant to this review.
When Harry Reich performed the first laparoscopically assisted vaginal hysterectomy in 1989, he advocated the use of sutures for control of the uterine vessels. Monopolar, bipolar, and laser instruments available at that time were inherently risky to use along the pelvic sidewall because of their potential for 1) considerable thermal energy spread beyond the area of treatment (bipolar, NG:YAG laser, monopolar) and 2) unreliable hemostasis (CO2 laser).
Minimally invasive surgical practice has driven meaningful advances in instrumentation and technique over the past 20 years. The constraints inherent in laparoscopic surgery, although somewhat mitigated by robotics, have generated a proliferation of technologies to obtain reliable hemostasis. Every device now on the market claims to “seal” vessels. In this article, I review the mechanism of action of these instruments and compare their strengths and weaknesses, based on high-quality scientific evidence.
Two studies highlight vessel ligation
Newcomb WL, Hope WW, Schmeizer TM, et al. Comparison of blood vessel sealing among new electrosurgical and ultrasonic devices. Surg Endosc. 2009;23:90–96.
Lamberton GR, Hsi RS, Jin DH, et al. Prospective comparison of four laparoscopic vessel ligation devices. J Endourol. 2008;22:2307–2312.
Many energy-delivery systems are available for the gynecologic surgeon; any of them can be used effectively and safely under most circumstances. Before we can make an informed choice about which system is best for our own practice, however, we need to be aware of the strengths and limitations of the systems overall ( TABLE ).
These two studies focus on the following devices:
- Gyrus PK Tissue Management System, PKS Cutting Forceps, and Plasma Trissector (all from Gyrus Medical). These are bipolar electrosurgical devices designed to deliver high current and very low voltage to tissue. Tissue impedance is continuously monitored between the jaws of the instrument, and energy delivery is adjusted accordingly. These systems deliver electrosurgical energy through a series of rapid pulses, thereby allowing the tissue to cool briefly and limiting the heating of adjacent tissue. Protein in the vessel walls is denatured and forms a coagulum, which occludes the lumen.
- Harmonic Scalpel (Ethicon EndoSurgery). This device uses a high-frequency ultrasonic transducer (55,000 cycles/second) to create mechanical vibration of one of the two jaws. The device can be used to vaporize tissue (cut) or achieve hemostasis by coagulation. As with the other devices, protein is denatured and vessels are occluded by formation of a coagulum.
- Ligamax 5 Endoscopic Multiple Clip Applier (Ethicon). This device is a sterile, single-patient-use, 5-mm, endoscopic, multiple-clip applicator that delivers 15 medium or large titanium clips that close to 8.8 mm, the same clip size as the 10-mm applicator. Ligamax 5 has long, thin angled jaws (8.4 mm) that extend beyond vessels and ducts to improve visibility. It includes a long, 33-cm shaft for additional reach, and an anti-clip drop-ratchet mechanism for control over clip closure.
- EnSeal Tissue Sealing and Hemostasis System and EnSeal PTC (SurgRx). EnSeal utilizes nanotechnology to control the energy at the electrode–tissue interface. The jaws contain a temperature-sensitive matrix with embedded conductive carbon spherules designed to “sense” tissue characteristics. It uses extremely high jaw compression to create uniform tissue effects. It does not require a dedicated electrosurgical unit for use; an adapter can be purchased that permits use with most generators.
- LigaSure V (Valleylab). LigaSure is a bipolar electrosurgical device designed to deliver high current and very low voltage to tissue. It monitors tissue impedance between the jaws of the instrument and continuously adjusts the delivery of energy.
TABLE
Rating vessel-sealing devices: 5 measures of success
| Device | Safety: Minimal thermal spread | Reliability: Efficacy on vessels ≤7 mm | Efficiency: Treatment time | Consistency: Independent of user | Utility: Multiple uses |
|---|---|---|---|---|---|
| Harmonic Scalpel | Excellent | Poor | Excellent | Poor | Excellent |
| Gyrus PK | Poor | Poor | Excellent | Fair | Fair |
| LigaSure V | Good | Excellent | Good | Excellent | Fair |
| EnSeal | Fair | Excellent | Poor | Excellent | Poor |
| These ratings were devised by the author based on data from independent studies in living tissue models. | |||||
FIGURE How energy-based vessel-sealing works
(A) When the carotid artery of an animal model was sealed using a standard bipolar forceps, the lumen remained open and a proximal thrombus developed. (B) When LigaSure was used, the artery was fused and the lumen obliterated with one 5-second application. (C and D) When a renal artery was sealed with LigaSure, the lumen of the vessel walls fused completely.
What the studies found
Newcomb and associates compared blood-vessel-sealing ability of the following devices:
- Gyrus 5-mm PKS Cutting Forceps
- Gyrus Plasma Trissector
- Harmonic Scalpel
- EnSeal Tissue Sealing and Hemostasis System
- LigaSure V, using the LigaSure Generator (Valleylab)
- LigaSure V, using the Force Triad Generator (Valleylab)
- Ligamax 5 Endoscopic Multiple Clip Applier.
The authors assessed mean seal times and burst pressures. Used on medium and large vessels, the Harmonic Scalpel and Gyrus products had significant failure rates: 8% to 22% for the Harmonic Scalpel and 41% to 92% for the pulsed, bipolar Gyrus systems.
The shortest sealing times for medium to large vessels were achieved with the LigaSure V using the Force Triad Generator. The Gyrus systems were the fastest devices when vessels were 2 to 3 mm in diameter.
There were no seal failures with the Ligamax, EnSeal, or LigaSure products.
Second study involved repeated applications
Lamberton and colleagues tested 5-mm laparoscopic devices under controlled temperature and humidity using a laparoscopic simulator, focusing on the following systems:
- LigaSure V
- Gyrus PK
- Harmonic Scalpel
- EnSeal Tissue Sealing and Hemostasis System.
The authors used 5-mm bovine arteries to assess sealing time, burst pressure, lateral thermal spread, and smoke production, as well as both subjective and objective effects on visibility.
Each device was used 10 times to determine burst pressure, lateral thermal spread, visibility, and smoke production. The devices were applied 20 times to measure time to seal, based on the devices’ preprogrammed endpoints.
The Harmonic Scalpel produced the lowest thermal spread and least smoke, but also had the lowest mean burst pressure.
The Gyrus PK generated the most smoke and had variable burst pressure. Although it had the fastest sealing times, in three of 10 trials there was a completely open arterial lumen following transection. In addition, 50% of applications involved burst pressures below 50 mm Hg.
Maximum temperatures 2 mm from the device were 49.9°C for the Harmonic Scalpel, 55.5°C for LigaSure, 58.9°C for EnSeal, and 64.5°C for Gyrus PK.
LigaSure was the highest-rated device overall, with the highest burst pressure and fastest sealing time.
EnSeal was the slowest and had variable burst pressures.
Clinical implications of the trials
These studies—neither of which was industry-sponsored—suggest that larger vessels cannot be controlled consistently and effectively using the Harmonic Scalpel or Gyrus systems. The Harmonic Scalpel is most user-dependent.
So what’s the bottom line? In the end, according to Andrew Brill, MD, past president of AAGL, “it’s not the wand, it’s the magician.”
for laparoscopic surgery
When OBG Management surveyed a number of laparoscopic experts and members of our Virtual Board of Editors about their preferred energy sources for laparoscopic surgery, the responses were strikingly similar. The consensus? The best device depends on the case at hand, the skill of the surgeon, economic concerns, and other variables.
As Keith Isaacson, MD, put it, “Because tissue may be thick or thin, moist or desiccated, vascular or avascular, the ideal instrument to achieve a pure cut varies. Because a vessel may be large or small, contain a large amount of collagen or very little collagen, or be on tension or relaxed, the ideal instrument for vessel sealing also varies, depending on the surgical situation.”
All of the devices are roughly equivalent, he added. “Fortunately, almost all of the commercially available energy sources that utilize bipolar radiofrequency or ultrasonic energy will perform our desired function if the surgeon understands the technology and utilizes the instruments properly.”
Here is a summary of recommendations made by the experts we interviewed.
Andrew I. Brill, MD
Director of Minimally Invasive Gynecology,
California Pacific Medical Center,
San Francisco, Calif
Having been actively engaged in advanced laparoscopic surgical training for more than 20 years, I have extensive experience with all of these novel devices. I critically assess any new energy-based device for its ergonomic handedness, propensity for sticking to tissue and production of plume, ability to manipulate and dissect tissue, efficiency in desiccated or fatty tissues, discoloration of tissue by carbon, response to tissue tension, and reliability for hemostasis. Because no device is perfectly suited to all procedures, I customarily rely upon several devices to satisfy my technical needs.
All advanced bipolar energy devices—LigaSure, PK Cutting Forceps, and EnSeal—can be safely used to coagulate and cut all vascular pedicles during hysterectomy and salpingo-oophorectomy. These devices perform best when tissue tension is reduced to maximize vessel sealing. Despite the fact that these devices provide audible feedback to signal the electrosurgical endpoint, I also gauge tissue color, retraction, and the emission of steam before advancing the cutting blade.
The tapered-tip design of the PK Cutting Forceps offers some comparative advantage for fine tissue dissection, but I find that hemostasis is more consistently achieved with less thermal spread using the EnSeal or LigaSure device.
I commonly utilize the Harmonic Scalpel in lieu of any electrosurgical device, understanding that it requires more finesse to achieve equivalent results.
When I anticipate that I will need to manipulate uterine pedicles during a difficult laparoscopic hysterectomy, I employ electrosurgery using the EnSeal device for its superior tissue sealing. A 3-mm curved device by EnSeal, akin to a Pean clamp, works as an exceptionally agile instrument for hysterectomy without sacrificing hemostasis.
Only the LigaSure Advance provides both coaptive coagulation and spark cutting via an electrode on the tip of one blade; therefore, an adjunctive mechanical or energy-based device must be employed to perform culdotomy during total laparoscopic hysterectomy when EnSeal or the PK Cutting Forceps is used.
The ability to efficiently and hemostatically cut through tissue of variable mass with minimal plume, predictable thermal margins, and the retention of tissue color make the Harmonic Scalpel my first choice for laparoscopic resection of endometriosis, extensive adhesiolysis, myomectomy, cervical amputation during supracervical hysterectomy, and culdotomy during total hysterectomy. Although I have similarly utilized the unique capacity of the spatula, J-hook, and needle electrodes by Plasmacision (Gyrus) to cut tissue using bipolar electrosurgery, all have afforded less technical control, not as much hemostasis, and wider thermal margins.
Use of any energy-based device does not preclude the need for skill. Before adding any of these devices to your surgical armamentarium, appropriate training should be acquired in a skills lab using living tissue or with a laparoscopic trainer using a tissue surrogate.
Dr. Brill reports no financial relationships relevant to this review.
Jon I. Einarsson, MD, MPH
Director of Minimally Invasive Gynecologic Surgery,
Brigham and Women’s Hospital,
Assistant Professor of Obstetrics,
Gynecology, and Reproductive Biology,
Harvard Medical School, Boston, Mass
I prefer the Harmonic Scalpel and a Kelly RoBi Bipolar Grasper (Karl Storz). I have been using these instruments for approximately 10 years. I use this combination for all operative laparoscopy cases, including hysterectomy, myomectomy, sacrocolpopexy, resection of endometriosis, and lysis of adhesions.
The Harmonic Scalpel is a versatile instrument that can perform dissection, cutting, and coagulation very efficiently. However, there are better vessel sealers out there, such as the LigaSure and EnSeal devices. In this era of cost containment, it is important to limit the use of multiple disposable energy sources in a single case. By adding the reusable bipolar grasper, vessel sealing becomes efficient and reliable without added cost. In addition, the bipolar grasper functions as a dissector and retractor, thereby minimizing instrument changes.
I watch the bubbles that form during desiccation with the bipolar grasper. Once these bubbles start to “die down,” I know that the tissue is desiccated and safe to transect. It is important to remember that thermal spread with a traditional bipolar grasper is significant (up to 13 mm). For that reason, when I desiccate the uterine vessels with the bipolar grasper, I stay above the rim of the colpotomy ring in order to maintain a safe distance from the ureter during this step.
The Harmonic Scalpel offers several advantages over monopolar electrosurgery for cutting, such as decreased smoke formation and avoidance of complications such as insulation failure, direct coupling, and capacitative coupling.
Dr. Einarsson reports no financial relationships relevant to this review.
Carl F. Giesler, MD
Associate Professor and Director of
Minimally Invasive Surgery,
Department of Obstetrics and Gynecology,
Baylor College of Medicine,
Waco, Tex
I was introduced to laparoscopy as a resident in 1974, and have been teaching laparoscopic surgical techniques to general surgeons and gynecologists since 1988. I began performing laparoscopic hysterectomy in 1991. As an educator and trainer of other laparoscopic surgeons, I have used all available laparoscopic energy sources.
I became an early adapter and user of ultrasonic mechanical energy when it was introduced for laparoscopy in 1995, because of its safety for adjacent tissues and its single-instrument, multiple-task properties.
This energy source was originally named the Ultrasonic Coagulating Shears by Ultracision, but Ethicon EndoSurgery purchased the product in 1995 and changed its name to the Harmonic Scalpel. The current generation is called the Harmonic ACE, and I use it as my primary laparoscopic energy source for all procedures, from management of unruptured ectopic pregnancy to total laparoscopic hysterectomy and extensive adhesiolyis in previously operated abdomens.
The Harmonic ACE allows me to confidently dissect adhesions over loops of bowel, as well as over the ureter, because of its limited lateral thermal spread. It allows nearly bloodless dissection of the retroperitoneal space and space around the bladder. The cavitation effect created by the rapidly moving instrument tip (55,500 cycles/second) creates tissue separation along the areolar tissue planes, where most blood vessels are the size of capillaries and where larger blood vessels are easily identified and controlled by coaptation, coagulation, and cutting.
The protein coagulum is produced at a temperature of 54°C. The sides and back of the active blade have minimal thermal effect because tissue is not held tightly against the moving blade. Minimal char is observed because of the low tissue temperature associated with coagulum formation. Minimal sticking of tissue occurs because the rapid motion of the instrument tip dislodges the tissue.
The ease of use, multiple functions for a single instrument, tissue safety, and minimal residual traumatized tissue are the reasons I prefer the mechanical energy of the Harmonic ACE as my primary laparoscopic energy source.
Dr. Giesler serves as a speaker for Ethicon EndoSurgery.
Cheryl Iglesia, MD
Director, Urogynecology and Reconstructive Pelvic Surgery,
Washington Hospital Center,
Associate Professor, Departments of ObGyn and Urology,
Georgetown University, Washington, DC.
Member, OBG Management Board of Editors
I prefer the Harmonic ACE for straight-stick laparoscopic hysterectomy cases and the PK Cutting Forceps for robotic hysterectomy and sacrocolpopexy.
The Harmonic ACE is fast and has little thermal spread; the PK Cutting Forceps is the only energy source available with reticulating arms for the robot.
I have been using the Harmonic ACE for more than 10 years and the PK Cutting Forceps since 2006.
Dr. Iglesia reports no financial relationships relevant to this review.
Keith Isaacson, MD
Director of Minimally Invasive Gynecologic
Surgery and Infertility,
Director of the AAGL/SRS
Fellowship in Minimally Invasive Gynecologic Surgery,
Newton Wellesley Hospital,
Associate Professor of Obstetrics, Gynecology,
and Reproductive Biology,
Harvard Medical School, Boston, Mass
I have used every bipolar and harmonic tool that is commercially available, including the recently released EnSeal. I like them all. The tools I have chosen were selected in conjunction with our purchasing department and determined to be the optimal tools for the lowest cost and highest efficiency. Our goal is to use as few disposables as possible, but to be as efficient as possible.
The instruments I utilize most frequently are the reusable 5-mm bipolar instruments from Karl Storz (RoBi) and ERBE Medical. The RoBi set includes a variety of tips, ranging from a dissecting Maryland-type tip to a flat, broad-based tip similar to the Kleppinger design. These are all plugged into the ERBE VIO generator. With this assortment of tools, I can seal a vessel of almost any size, whether it be thin-walled, such as a large vein, or a thick-walled artery.
To cut tissue, I prefer the Harmonic Scalpel and the bipolar Laparoscopic Spatula (Gyrus). I use the Harmonic Scalpel during laparoscopic hysterectomy, and the Spatula during laparoscopic myomectomy. Both devices cut tissue well while simultaneously sealing small vessels.
I have been using these instruments for approximately 10 years.
Dr. Isaacson reports that he is a consultant for Karl Storz in hysteroscopy.
Noor Ahmed, MB, ChB, MD
Consultant Gynecologist,
Dundee, Scotland.
Member, OBG Management Virtual
Board of Editors
I am a consultant gynecologist with an interest in fertility, endometriosis surgery, and general gynecologic surgery. Most of my surgical work is performed laparoscopically or vaginally.
My first experience with vessel-sealing technology was in 2000, when I began using the LigaSure Max for difficult vaginal hysterectomies. I was impressed by its ability to clamp and seal and, in later versions, its ability to cut, as well. Since then, I have been using various versions of LigaSure vessel- and tissue-sealing technology for many gynecologic procedures.
I have had some exposure to other vessel- and tissue-sealing technologies, but prefer LigaSure because:
1. It provides an experience similar to that of conventional clamp, cut, and suture through the control offered by its unique combination of pressure and energy, creating vessel and tissue fusion that does not rely on a proximal thrombus and produces minimal thermal spread.
2. The feedback-controlled response system automatically discontinues energy delivery when sealing is complete.
3. It works with clamps of various sizes and shapes, using the same hardware (generator) and saving money and space. Moreover, the nursing staff has to develop familiarity with one machine only.
For laparoscopic procedures, I use LigaSure Lap, LigaSure Atlas Sealer/Divider and, most commonly, the 5-mm LigaSure V Sealer/Divider.
Dr. Ahmed reports no financial relationships relevant to this review.
Michael Kirwin, MD
General Obstetrics and Gynecology, Freehold, NJ.
Member, OBG Management Virtual Board of Editors
With advanced laparoscopic cases, particularly hysterectomy, I use the Harmonic Scalpel and PK Cutting Forceps. I have used the Harmonic Scalpel regularly for approximately 4 years, and it works well for dissection and, particularly, for transecting the cervix in supracervical cases.
Over the past year, I have also begun to use the Gyrus forceps, especially when I expect to encounter larger vessels that require coagulation. Both instruments can be used in 5-mm ports. Both seem relatively reliable and easy for the OR team to use. I would hope that an articulation feature can be added in the future.
Dr. Kirwin reports no financial relationships relevant to this review.
Alcohol Abuse in Posttraumatic Stress Disorder: Identification and Intervention
Tackling Adverse Reactions to Local Anesthetics
Whose Pain Scale Is It, Anyway?
A Promising New Risk Score
Grand Rounds: Man, 29, With Apparent Throat Obstruction
A 29-year-old man presented to the emergency department (ED) with a chief complaint of food stuck in his throat. He reported that he had swallowed a piece of chicken and felt it get stuck. Drinking water to help it go down was unsuccessful.
The patient’s history was positive for childhood asthma and nine years of solid food dysphagia. There was no history of a caustic chemical ingestion or of drug-induced esophagitis. He denied having dyspepsia, heartburn, or chest pain. He was not taking any medications and had no allergies.
When his dysphagia symptoms began nine years ago, he was diagnosed with acid reflux disease, confirmed by an upper gastrointestinal (GI) tract x-ray. Since that time, he reported having to swallow liquid after every bite of food and said he suffered from severe anxiety over fear of choking.
Evaluation in the ED consisted of endoscopic examination by a gastroenterologist. In addition to dislodging a food bolus, the endoscope revealed a narrowed, ringed esophagus with mucosal changes throughout the length of the esophagus (see Figures 1 through 3). Esophageal biopsies were taken, and the esophagus was dilated successfully with a 40-Fr Maloney dilator. The endoscopist detected too much resistance to pass a larger dilator.
Biopsy results revealed eosinophilic esophagitis. The patient was given oral fluticasone propionate. At one-month follow-up, he reported feeling much better. Upper endoscopy revealed some improvement, and the gastroenterologist was able to pass both a 46- and a 48-Fr Maloney dilator with only mild resistance. (The largest Maloney dilator, a 60-Fr dilator, should easily pass through a normal esophagus, according to T. L. Sack, MD, oral communication, June 2009.)
Discussion
Eosinophilic esophagitis (EE) involves the infiltration of the esophageal mucosa with eosinophils, causing edema, inflammation, and eventually, thickening and stenotic changes of the esophageal mucosa.1
The normal esophageal mucosa contains lymphocytes, mast cells, and dendritic cells, which protect the esophagus from invading toxins and microorganisms. Eosinophils are not usually present, but when they are, they can have toxic effects on the esophageal mucosa.2 EE is associated with solid food dysphagia, a direct result of damage to the esophageal mucosa, and other causes that are not clearly understood.
Research findings suggest that symptoms of dysphagia may be caused by degranulating eosinophils and mast cells, which have an antagonistic effect on the muscarinic receptors and cause smooth muscle to contract.3,4 The proposed triggering mechanism of EE is an immunoglobulin E (IgE) immune–mediated response to an allergen.2 Based on results from IgE radioallergosorbent testing (RAST), aeroallergens are more likely than food to act as triggers.5
EE in the Adult Patient
Traditionally, EE has been a condition seen in the pediatric population, with symptoms of nausea, vomiting, and failure to thrive; however, it is becoming increasingly recognized among adults. The typical patient is a man in his 20s or 30s (although cases of EE have been reported among women and older adults) with acute and recurrent solid food dysphagia, with or without food impaction.4
Often the patient reports a history of environmental or food allergies, asthma, rhinitis, or eczema.2,4-6 Researchers have reported the presence of allergic symptoms in at least 50% of patients diagnosed with EE,2 and many patients experience exacerbations associated with seasonal changes.7
GERD may coexist with EE; however, no relationship has been identified between the two.8 EE should be considered in patients with gastrointestinal symptoms that persist despite at least four weeks’ treatment with a proton pump inhibitor (PPI).2
Dysphagia: Differential Diagnosis
Adult patients with esophageal dysphagia usually report the feeling of food getting stuck when they try to swallow.9 Dysphagia may result from a mechanical obstruction or a neuromuscular/motility condition. Patients with mechanical obstructions usually have difficulty swallowing solids, while those with motility disorders tend to have difficulty with both liquids and solids.1,9
Mechanical obstructions may include carcinomas (intrinsic and extrinsic), strictures, or Schatzki rings (small thin mucosal rings of unknown etiology located at the gastroesophageal junction).1,9 Progressive dysphagia to solids over a short period of time is often indicative of esophageal carcinoma. GERD, pill-induced trauma, previous ingestion of a caustic chemical, and radiation are common causes of esophageal stricture formation. For a list of medications that are particularly caustic to the esophageal mucosa, see the table.9,10
Neuromuscular manifestations of dysphagia include achalasia, diffuse esophageal spasm, nutcracker esophagus, and scleroderma.1 These are usually associated with progressive difficulty in swallowing.9
Evaluating the Patient
A thorough patient history can often reveal potential causes of dysphagia and eliminate others. This should include current medications, chronic medical conditions and details regarding their onset and duration, and symptoms associated with dysphagia.9
Physical examination should include palpation of the thyroid because of the potential for a thyroid mass to cause extrinsic compression of the esophagus, palpation of the abdomen for masses or organomegaly, and a complete neurologic evaluation.9
Laboratory tests should be ordered based on the information obtained from the history and physical. Testing may include thyroid studies to eliminate hypothyroid or hyperthyroid causes of dysphagia, and complete blood count (CBC) with differential to rule out inflammatory or infectious processes.9 While eosinophilia may be present in the differential, it is not a universally accepted marker for establishing the diagnosis of EE.2,5 Stools should be checked for occult blood, because a positive finding may suggest esophageal carcinoma.9
Diagnosis
In the primary care setting, a barium esophagram may be used during the initial workup to evaluate the anatomic structures of the esophagus and to differentiate between a mechanical obstruction and a neuromuscular disorder.1,9 This noninvasive test requires the patient to swallow a radiopaque liquid as x-rays are taken.
The gold standard for diagnosing EE, however, is upper endoscopy with biopsy of the esophageal mucosa.6 Endoscopic findings that indicate EE are atypical of GERD; they may include a narrowed, small-caliber esophagus, concentric mucosal rings, proximal stenosis, linear ulcerations, atrophic changes, and white papules associated with eosinophilic microabscesses.6
Although there is no consensus regarding the number of eosinophils that should be present for an accurate diagnosis of EE, microscopic interpretation of the biopsy from both the proximal and the distal esophageal epithelia5 usually shows 15 or more eosinophils per high-power field.2,11 It has been suggested that mucosal biopsies be taken along the entire length of the esophagus, as eosinophilic infiltration may extend from the proximal to the distal esophagus.2
GERD and trauma induced by medication use may also be associated with esophageal eosinophilic infiltration5; however, eosinophils are usually present only in the distal esophageal mucosa3 and are not as abundant as in EE.7 If endoscopy reveals persistent eosinophilia despite four to eight weeks’ treatment with a PPI, the diagnosis of EE is confirmed.2
Treatment
Treatment for EE is still under investigation. Research has examined the association between EE and food allergies or aeroallergens.4 Evaluation by an allergist using skin prick tests or RAST is recommended in the adult patient to help determine the source of the underlying inflammation.5,7 Eliminating any identified allergen should help alleviate symptoms.4
For patients in whom no source of inflammation can be identified, treatment with 1.0 to 2.0 mg/kg/d of oral prednisone for acute exacerbations has been shown to significantly improve symptoms and histology12; however, because of the associated risk for adverse systemic effects, long-term use is not recommended.
In many patients, the inhaled corticosteroid fluticasone has also proved successful in reducing EE—associated inflammation.6 Current evidence supports adult dosing between 880 and 1,760 mcg per day for six to eight weeks, administered with a metered-dose inhaler and no spacer. Fluticasone should be sprayed directly into the mouth and swallowed, after which the patient should take nothing by mouth for 30 minutes.13 Prolonged fluticasone use has been associated with esophageal candidiasis.2 There are currently no recommendations regarding its use as maintenance therapy.
Montelukast, a leukotriene receptor antagonist, has also been shown in some studies to reduce the inflammatory process11; however, one study team recently found it to have no therapeutic effect.13
PPIs may be effective for improving EE symptoms even in the absence of GERD because of the reduced gastric acid production,7 but they do not usually improve EE’s histologic features.3
Use of esophageal dilation in patients with EE is controversial because of an associated risk for perforation.14 If this intervention is to be performed, the patient should be treated in advance with oral corticosteroids to reduce esophageal inflammation.15,16 In addition, the endoscopist should start with small-sized dilators and carefully proceed to larger sizes.11 Critics of esophageal dilation argue that the procedure is only a temporary solution and does nothing for the underlying condition.4,8
Regarding endoscopic surveillance, an interval of at least four weeks between interventions is recommended.13
Role of the Primary Care Clinician
Undiagnosed EE can cause the patient discomfort, frustration, and anxiety, as seen in the case study. Many patients with undiagnosed EE have been exposed to unnecessary medical therapy and antireflux surgery.3 Without proper diagnosis and treatment, EE may worsen, causing complications associated with chronic inflammation (ie, esophageal fibrosis and strictures).2,6
The long-term prognosis of EE is unknown at this time.8 The disease is usually chronic, with periods of remission and exacerbation. With an understanding of EE and appropriate therapies, the primary care practitioner can team with the gastroenterologist to provide effective disease management through endoscopic surveillance and intervention for acute exacerbations. Guidelines recommend that patients be closely followed with regular office visits to reassess symptoms, compliance with therapy, and adverse effects, with the goal of preventing complications associated with EE.13
Conclusion
To effectively evaluate the patient who presents with dysphagia, the primary care provider should have a working knowledge of EE, as well as an understanding of the key elements in the history and physical examination to help ensure an accurate diagnosis. This will facilitate timely referral to a gastroenterologist for endoscopic evaluation, when indicated.
1. McQuaid KR. Gastrointestinal disorders. In: McPhee S, Papadakis M. CURRENT Medical Diagnosis & Treatment 2009. New York: McGraw-Hill: 2009:487-581.
2. Nurko S, Furuta GT. Eosinophilic esophagitis (2006). GI Motility Online. www.nature.com/gimo/contents/pt1/full/gimo49.html. Accessed July 27, 2009.
3. Parfitt JR, Gregor JC, Suskin NG, et al. Eosinophilic esophagitis in adults: distinguishing features from gastroesophageal reflux disease: a study of 41 patients. Mod Pathol. 2006;19(1):90-96.
4. Swoger JM, Weiler CR, Arora AS. Eosinophilic esophagitis: is it all allergies? Mayo Clin Proc. 2007;82(12):1541-1549.
5. Conus S, Simon HU. General laboratory diagnostics of eosinophilic GI diseases. Best Pract Res Clin Gastroenterol. 2008;22(3):441-453.
6. Remedios M, Campbell C, Jones DM, Kerlin P. Eosinophilic esophagitis in adults: clinical, endoscopic, histologic findings, and response to treatment with fluticasone propionate. Gastrointest Endosc. 2006;63(1):3-12.
7. Rothenberg ME. Eosinophilic gastrointestinal disorders (EGID). J Allergy Clin Immunol. 2004; 113(1):11-28.
8. Lucendo AJ, Carrion G, Navarro M, et al. Eosinophilic esophagitis in adults: an emerging disease. Dig Dis Sci. 2004;49(11-12):1884-1888.
9. Spieker MR. Evaluating dysphagia. Am Fam Physician. 2000;61(12):3639-3648.
10. Boyce HW. Drug-induced esophageal damage: diseases of medical progress. Gastrointest Endosc. 1998;47:547-550.
11. Potter JW, Saeian K, Staff D, et al. Eosinophilic esophagitis in adults: an emerging problem with unique esophageal features. Gastrointest Endosc. 2004;59(3):355-361.
12. Schaefer ET, Fitzgerald JF, Molleston JP, et al. Comparison of oral prednisone and topical fluticasone in the treatment of eosinophilic esophagitis: a randomized trial in children. Clin Gastroenterol Hepatol. 2008;6(2):165-173.
13. Furuta GT, Liacouras CA, Collins MH, et al; First International Gastrointestinal Eosinophil Research Symposium (FIGERS) Subcommittees. Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendation for diagnosis and treatment. Gastroenterology. 2007;133(4): 1342-1363.
14. Straumann A, Rossi L, Simon HU, et al. Fragility of the esophageal mucosa: a pathognomonic endoscopic sign of primary eosinophilic esophagitis. Gastrointest Endosc. 2003;57(3):407-412.
15. Hawari R, Pasricha PJ. Images in clinical medicine: eosinophilic esophagitis. N Engl J Med. 2007; 356(20):e20.
16. Leclercq P, Marting A, Gast P. Eosinophilic esophagitis. N Engl J Med. 2007;357(14):1446.
A 29-year-old man presented to the emergency department (ED) with a chief complaint of food stuck in his throat. He reported that he had swallowed a piece of chicken and felt it get stuck. Drinking water to help it go down was unsuccessful.
The patient’s history was positive for childhood asthma and nine years of solid food dysphagia. There was no history of a caustic chemical ingestion or of drug-induced esophagitis. He denied having dyspepsia, heartburn, or chest pain. He was not taking any medications and had no allergies.
When his dysphagia symptoms began nine years ago, he was diagnosed with acid reflux disease, confirmed by an upper gastrointestinal (GI) tract x-ray. Since that time, he reported having to swallow liquid after every bite of food and said he suffered from severe anxiety over fear of choking.
Evaluation in the ED consisted of endoscopic examination by a gastroenterologist. In addition to dislodging a food bolus, the endoscope revealed a narrowed, ringed esophagus with mucosal changes throughout the length of the esophagus (see Figures 1 through 3). Esophageal biopsies were taken, and the esophagus was dilated successfully with a 40-Fr Maloney dilator. The endoscopist detected too much resistance to pass a larger dilator.
Biopsy results revealed eosinophilic esophagitis. The patient was given oral fluticasone propionate. At one-month follow-up, he reported feeling much better. Upper endoscopy revealed some improvement, and the gastroenterologist was able to pass both a 46- and a 48-Fr Maloney dilator with only mild resistance. (The largest Maloney dilator, a 60-Fr dilator, should easily pass through a normal esophagus, according to T. L. Sack, MD, oral communication, June 2009.)
Discussion
Eosinophilic esophagitis (EE) involves the infiltration of the esophageal mucosa with eosinophils, causing edema, inflammation, and eventually, thickening and stenotic changes of the esophageal mucosa.1
The normal esophageal mucosa contains lymphocytes, mast cells, and dendritic cells, which protect the esophagus from invading toxins and microorganisms. Eosinophils are not usually present, but when they are, they can have toxic effects on the esophageal mucosa.2 EE is associated with solid food dysphagia, a direct result of damage to the esophageal mucosa, and other causes that are not clearly understood.
Research findings suggest that symptoms of dysphagia may be caused by degranulating eosinophils and mast cells, which have an antagonistic effect on the muscarinic receptors and cause smooth muscle to contract.3,4 The proposed triggering mechanism of EE is an immunoglobulin E (IgE) immune–mediated response to an allergen.2 Based on results from IgE radioallergosorbent testing (RAST), aeroallergens are more likely than food to act as triggers.5
EE in the Adult Patient
Traditionally, EE has been a condition seen in the pediatric population, with symptoms of nausea, vomiting, and failure to thrive; however, it is becoming increasingly recognized among adults. The typical patient is a man in his 20s or 30s (although cases of EE have been reported among women and older adults) with acute and recurrent solid food dysphagia, with or without food impaction.4
Often the patient reports a history of environmental or food allergies, asthma, rhinitis, or eczema.2,4-6 Researchers have reported the presence of allergic symptoms in at least 50% of patients diagnosed with EE,2 and many patients experience exacerbations associated with seasonal changes.7
GERD may coexist with EE; however, no relationship has been identified between the two.8 EE should be considered in patients with gastrointestinal symptoms that persist despite at least four weeks’ treatment with a proton pump inhibitor (PPI).2
Dysphagia: Differential Diagnosis
Adult patients with esophageal dysphagia usually report the feeling of food getting stuck when they try to swallow.9 Dysphagia may result from a mechanical obstruction or a neuromuscular/motility condition. Patients with mechanical obstructions usually have difficulty swallowing solids, while those with motility disorders tend to have difficulty with both liquids and solids.1,9
Mechanical obstructions may include carcinomas (intrinsic and extrinsic), strictures, or Schatzki rings (small thin mucosal rings of unknown etiology located at the gastroesophageal junction).1,9 Progressive dysphagia to solids over a short period of time is often indicative of esophageal carcinoma. GERD, pill-induced trauma, previous ingestion of a caustic chemical, and radiation are common causes of esophageal stricture formation. For a list of medications that are particularly caustic to the esophageal mucosa, see the table.9,10
Neuromuscular manifestations of dysphagia include achalasia, diffuse esophageal spasm, nutcracker esophagus, and scleroderma.1 These are usually associated with progressive difficulty in swallowing.9
Evaluating the Patient
A thorough patient history can often reveal potential causes of dysphagia and eliminate others. This should include current medications, chronic medical conditions and details regarding their onset and duration, and symptoms associated with dysphagia.9
Physical examination should include palpation of the thyroid because of the potential for a thyroid mass to cause extrinsic compression of the esophagus, palpation of the abdomen for masses or organomegaly, and a complete neurologic evaluation.9
Laboratory tests should be ordered based on the information obtained from the history and physical. Testing may include thyroid studies to eliminate hypothyroid or hyperthyroid causes of dysphagia, and complete blood count (CBC) with differential to rule out inflammatory or infectious processes.9 While eosinophilia may be present in the differential, it is not a universally accepted marker for establishing the diagnosis of EE.2,5 Stools should be checked for occult blood, because a positive finding may suggest esophageal carcinoma.9
Diagnosis
In the primary care setting, a barium esophagram may be used during the initial workup to evaluate the anatomic structures of the esophagus and to differentiate between a mechanical obstruction and a neuromuscular disorder.1,9 This noninvasive test requires the patient to swallow a radiopaque liquid as x-rays are taken.
The gold standard for diagnosing EE, however, is upper endoscopy with biopsy of the esophageal mucosa.6 Endoscopic findings that indicate EE are atypical of GERD; they may include a narrowed, small-caliber esophagus, concentric mucosal rings, proximal stenosis, linear ulcerations, atrophic changes, and white papules associated with eosinophilic microabscesses.6
Although there is no consensus regarding the number of eosinophils that should be present for an accurate diagnosis of EE, microscopic interpretation of the biopsy from both the proximal and the distal esophageal epithelia5 usually shows 15 or more eosinophils per high-power field.2,11 It has been suggested that mucosal biopsies be taken along the entire length of the esophagus, as eosinophilic infiltration may extend from the proximal to the distal esophagus.2
GERD and trauma induced by medication use may also be associated with esophageal eosinophilic infiltration5; however, eosinophils are usually present only in the distal esophageal mucosa3 and are not as abundant as in EE.7 If endoscopy reveals persistent eosinophilia despite four to eight weeks’ treatment with a PPI, the diagnosis of EE is confirmed.2
Treatment
Treatment for EE is still under investigation. Research has examined the association between EE and food allergies or aeroallergens.4 Evaluation by an allergist using skin prick tests or RAST is recommended in the adult patient to help determine the source of the underlying inflammation.5,7 Eliminating any identified allergen should help alleviate symptoms.4
For patients in whom no source of inflammation can be identified, treatment with 1.0 to 2.0 mg/kg/d of oral prednisone for acute exacerbations has been shown to significantly improve symptoms and histology12; however, because of the associated risk for adverse systemic effects, long-term use is not recommended.
In many patients, the inhaled corticosteroid fluticasone has also proved successful in reducing EE—associated inflammation.6 Current evidence supports adult dosing between 880 and 1,760 mcg per day for six to eight weeks, administered with a metered-dose inhaler and no spacer. Fluticasone should be sprayed directly into the mouth and swallowed, after which the patient should take nothing by mouth for 30 minutes.13 Prolonged fluticasone use has been associated with esophageal candidiasis.2 There are currently no recommendations regarding its use as maintenance therapy.
Montelukast, a leukotriene receptor antagonist, has also been shown in some studies to reduce the inflammatory process11; however, one study team recently found it to have no therapeutic effect.13
PPIs may be effective for improving EE symptoms even in the absence of GERD because of the reduced gastric acid production,7 but they do not usually improve EE’s histologic features.3
Use of esophageal dilation in patients with EE is controversial because of an associated risk for perforation.14 If this intervention is to be performed, the patient should be treated in advance with oral corticosteroids to reduce esophageal inflammation.15,16 In addition, the endoscopist should start with small-sized dilators and carefully proceed to larger sizes.11 Critics of esophageal dilation argue that the procedure is only a temporary solution and does nothing for the underlying condition.4,8
Regarding endoscopic surveillance, an interval of at least four weeks between interventions is recommended.13
Role of the Primary Care Clinician
Undiagnosed EE can cause the patient discomfort, frustration, and anxiety, as seen in the case study. Many patients with undiagnosed EE have been exposed to unnecessary medical therapy and antireflux surgery.3 Without proper diagnosis and treatment, EE may worsen, causing complications associated with chronic inflammation (ie, esophageal fibrosis and strictures).2,6
The long-term prognosis of EE is unknown at this time.8 The disease is usually chronic, with periods of remission and exacerbation. With an understanding of EE and appropriate therapies, the primary care practitioner can team with the gastroenterologist to provide effective disease management through endoscopic surveillance and intervention for acute exacerbations. Guidelines recommend that patients be closely followed with regular office visits to reassess symptoms, compliance with therapy, and adverse effects, with the goal of preventing complications associated with EE.13
Conclusion
To effectively evaluate the patient who presents with dysphagia, the primary care provider should have a working knowledge of EE, as well as an understanding of the key elements in the history and physical examination to help ensure an accurate diagnosis. This will facilitate timely referral to a gastroenterologist for endoscopic evaluation, when indicated.
A 29-year-old man presented to the emergency department (ED) with a chief complaint of food stuck in his throat. He reported that he had swallowed a piece of chicken and felt it get stuck. Drinking water to help it go down was unsuccessful.
The patient’s history was positive for childhood asthma and nine years of solid food dysphagia. There was no history of a caustic chemical ingestion or of drug-induced esophagitis. He denied having dyspepsia, heartburn, or chest pain. He was not taking any medications and had no allergies.
When his dysphagia symptoms began nine years ago, he was diagnosed with acid reflux disease, confirmed by an upper gastrointestinal (GI) tract x-ray. Since that time, he reported having to swallow liquid after every bite of food and said he suffered from severe anxiety over fear of choking.
Evaluation in the ED consisted of endoscopic examination by a gastroenterologist. In addition to dislodging a food bolus, the endoscope revealed a narrowed, ringed esophagus with mucosal changes throughout the length of the esophagus (see Figures 1 through 3). Esophageal biopsies were taken, and the esophagus was dilated successfully with a 40-Fr Maloney dilator. The endoscopist detected too much resistance to pass a larger dilator.
Biopsy results revealed eosinophilic esophagitis. The patient was given oral fluticasone propionate. At one-month follow-up, he reported feeling much better. Upper endoscopy revealed some improvement, and the gastroenterologist was able to pass both a 46- and a 48-Fr Maloney dilator with only mild resistance. (The largest Maloney dilator, a 60-Fr dilator, should easily pass through a normal esophagus, according to T. L. Sack, MD, oral communication, June 2009.)
Discussion
Eosinophilic esophagitis (EE) involves the infiltration of the esophageal mucosa with eosinophils, causing edema, inflammation, and eventually, thickening and stenotic changes of the esophageal mucosa.1
The normal esophageal mucosa contains lymphocytes, mast cells, and dendritic cells, which protect the esophagus from invading toxins and microorganisms. Eosinophils are not usually present, but when they are, they can have toxic effects on the esophageal mucosa.2 EE is associated with solid food dysphagia, a direct result of damage to the esophageal mucosa, and other causes that are not clearly understood.
Research findings suggest that symptoms of dysphagia may be caused by degranulating eosinophils and mast cells, which have an antagonistic effect on the muscarinic receptors and cause smooth muscle to contract.3,4 The proposed triggering mechanism of EE is an immunoglobulin E (IgE) immune–mediated response to an allergen.2 Based on results from IgE radioallergosorbent testing (RAST), aeroallergens are more likely than food to act as triggers.5
EE in the Adult Patient
Traditionally, EE has been a condition seen in the pediatric population, with symptoms of nausea, vomiting, and failure to thrive; however, it is becoming increasingly recognized among adults. The typical patient is a man in his 20s or 30s (although cases of EE have been reported among women and older adults) with acute and recurrent solid food dysphagia, with or without food impaction.4
Often the patient reports a history of environmental or food allergies, asthma, rhinitis, or eczema.2,4-6 Researchers have reported the presence of allergic symptoms in at least 50% of patients diagnosed with EE,2 and many patients experience exacerbations associated with seasonal changes.7
GERD may coexist with EE; however, no relationship has been identified between the two.8 EE should be considered in patients with gastrointestinal symptoms that persist despite at least four weeks’ treatment with a proton pump inhibitor (PPI).2
Dysphagia: Differential Diagnosis
Adult patients with esophageal dysphagia usually report the feeling of food getting stuck when they try to swallow.9 Dysphagia may result from a mechanical obstruction or a neuromuscular/motility condition. Patients with mechanical obstructions usually have difficulty swallowing solids, while those with motility disorders tend to have difficulty with both liquids and solids.1,9
Mechanical obstructions may include carcinomas (intrinsic and extrinsic), strictures, or Schatzki rings (small thin mucosal rings of unknown etiology located at the gastroesophageal junction).1,9 Progressive dysphagia to solids over a short period of time is often indicative of esophageal carcinoma. GERD, pill-induced trauma, previous ingestion of a caustic chemical, and radiation are common causes of esophageal stricture formation. For a list of medications that are particularly caustic to the esophageal mucosa, see the table.9,10
Neuromuscular manifestations of dysphagia include achalasia, diffuse esophageal spasm, nutcracker esophagus, and scleroderma.1 These are usually associated with progressive difficulty in swallowing.9
Evaluating the Patient
A thorough patient history can often reveal potential causes of dysphagia and eliminate others. This should include current medications, chronic medical conditions and details regarding their onset and duration, and symptoms associated with dysphagia.9
Physical examination should include palpation of the thyroid because of the potential for a thyroid mass to cause extrinsic compression of the esophagus, palpation of the abdomen for masses or organomegaly, and a complete neurologic evaluation.9
Laboratory tests should be ordered based on the information obtained from the history and physical. Testing may include thyroid studies to eliminate hypothyroid or hyperthyroid causes of dysphagia, and complete blood count (CBC) with differential to rule out inflammatory or infectious processes.9 While eosinophilia may be present in the differential, it is not a universally accepted marker for establishing the diagnosis of EE.2,5 Stools should be checked for occult blood, because a positive finding may suggest esophageal carcinoma.9
Diagnosis
In the primary care setting, a barium esophagram may be used during the initial workup to evaluate the anatomic structures of the esophagus and to differentiate between a mechanical obstruction and a neuromuscular disorder.1,9 This noninvasive test requires the patient to swallow a radiopaque liquid as x-rays are taken.
The gold standard for diagnosing EE, however, is upper endoscopy with biopsy of the esophageal mucosa.6 Endoscopic findings that indicate EE are atypical of GERD; they may include a narrowed, small-caliber esophagus, concentric mucosal rings, proximal stenosis, linear ulcerations, atrophic changes, and white papules associated with eosinophilic microabscesses.6
Although there is no consensus regarding the number of eosinophils that should be present for an accurate diagnosis of EE, microscopic interpretation of the biopsy from both the proximal and the distal esophageal epithelia5 usually shows 15 or more eosinophils per high-power field.2,11 It has been suggested that mucosal biopsies be taken along the entire length of the esophagus, as eosinophilic infiltration may extend from the proximal to the distal esophagus.2
GERD and trauma induced by medication use may also be associated with esophageal eosinophilic infiltration5; however, eosinophils are usually present only in the distal esophageal mucosa3 and are not as abundant as in EE.7 If endoscopy reveals persistent eosinophilia despite four to eight weeks’ treatment with a PPI, the diagnosis of EE is confirmed.2
Treatment
Treatment for EE is still under investigation. Research has examined the association between EE and food allergies or aeroallergens.4 Evaluation by an allergist using skin prick tests or RAST is recommended in the adult patient to help determine the source of the underlying inflammation.5,7 Eliminating any identified allergen should help alleviate symptoms.4
For patients in whom no source of inflammation can be identified, treatment with 1.0 to 2.0 mg/kg/d of oral prednisone for acute exacerbations has been shown to significantly improve symptoms and histology12; however, because of the associated risk for adverse systemic effects, long-term use is not recommended.
In many patients, the inhaled corticosteroid fluticasone has also proved successful in reducing EE—associated inflammation.6 Current evidence supports adult dosing between 880 and 1,760 mcg per day for six to eight weeks, administered with a metered-dose inhaler and no spacer. Fluticasone should be sprayed directly into the mouth and swallowed, after which the patient should take nothing by mouth for 30 minutes.13 Prolonged fluticasone use has been associated with esophageal candidiasis.2 There are currently no recommendations regarding its use as maintenance therapy.
Montelukast, a leukotriene receptor antagonist, has also been shown in some studies to reduce the inflammatory process11; however, one study team recently found it to have no therapeutic effect.13
PPIs may be effective for improving EE symptoms even in the absence of GERD because of the reduced gastric acid production,7 but they do not usually improve EE’s histologic features.3
Use of esophageal dilation in patients with EE is controversial because of an associated risk for perforation.14 If this intervention is to be performed, the patient should be treated in advance with oral corticosteroids to reduce esophageal inflammation.15,16 In addition, the endoscopist should start with small-sized dilators and carefully proceed to larger sizes.11 Critics of esophageal dilation argue that the procedure is only a temporary solution and does nothing for the underlying condition.4,8
Regarding endoscopic surveillance, an interval of at least four weeks between interventions is recommended.13
Role of the Primary Care Clinician
Undiagnosed EE can cause the patient discomfort, frustration, and anxiety, as seen in the case study. Many patients with undiagnosed EE have been exposed to unnecessary medical therapy and antireflux surgery.3 Without proper diagnosis and treatment, EE may worsen, causing complications associated with chronic inflammation (ie, esophageal fibrosis and strictures).2,6
The long-term prognosis of EE is unknown at this time.8 The disease is usually chronic, with periods of remission and exacerbation. With an understanding of EE and appropriate therapies, the primary care practitioner can team with the gastroenterologist to provide effective disease management through endoscopic surveillance and intervention for acute exacerbations. Guidelines recommend that patients be closely followed with regular office visits to reassess symptoms, compliance with therapy, and adverse effects, with the goal of preventing complications associated with EE.13
Conclusion
To effectively evaluate the patient who presents with dysphagia, the primary care provider should have a working knowledge of EE, as well as an understanding of the key elements in the history and physical examination to help ensure an accurate diagnosis. This will facilitate timely referral to a gastroenterologist for endoscopic evaluation, when indicated.
1. McQuaid KR. Gastrointestinal disorders. In: McPhee S, Papadakis M. CURRENT Medical Diagnosis & Treatment 2009. New York: McGraw-Hill: 2009:487-581.
2. Nurko S, Furuta GT. Eosinophilic esophagitis (2006). GI Motility Online. www.nature.com/gimo/contents/pt1/full/gimo49.html. Accessed July 27, 2009.
3. Parfitt JR, Gregor JC, Suskin NG, et al. Eosinophilic esophagitis in adults: distinguishing features from gastroesophageal reflux disease: a study of 41 patients. Mod Pathol. 2006;19(1):90-96.
4. Swoger JM, Weiler CR, Arora AS. Eosinophilic esophagitis: is it all allergies? Mayo Clin Proc. 2007;82(12):1541-1549.
5. Conus S, Simon HU. General laboratory diagnostics of eosinophilic GI diseases. Best Pract Res Clin Gastroenterol. 2008;22(3):441-453.
6. Remedios M, Campbell C, Jones DM, Kerlin P. Eosinophilic esophagitis in adults: clinical, endoscopic, histologic findings, and response to treatment with fluticasone propionate. Gastrointest Endosc. 2006;63(1):3-12.
7. Rothenberg ME. Eosinophilic gastrointestinal disorders (EGID). J Allergy Clin Immunol. 2004; 113(1):11-28.
8. Lucendo AJ, Carrion G, Navarro M, et al. Eosinophilic esophagitis in adults: an emerging disease. Dig Dis Sci. 2004;49(11-12):1884-1888.
9. Spieker MR. Evaluating dysphagia. Am Fam Physician. 2000;61(12):3639-3648.
10. Boyce HW. Drug-induced esophageal damage: diseases of medical progress. Gastrointest Endosc. 1998;47:547-550.
11. Potter JW, Saeian K, Staff D, et al. Eosinophilic esophagitis in adults: an emerging problem with unique esophageal features. Gastrointest Endosc. 2004;59(3):355-361.
12. Schaefer ET, Fitzgerald JF, Molleston JP, et al. Comparison of oral prednisone and topical fluticasone in the treatment of eosinophilic esophagitis: a randomized trial in children. Clin Gastroenterol Hepatol. 2008;6(2):165-173.
13. Furuta GT, Liacouras CA, Collins MH, et al; First International Gastrointestinal Eosinophil Research Symposium (FIGERS) Subcommittees. Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendation for diagnosis and treatment. Gastroenterology. 2007;133(4): 1342-1363.
14. Straumann A, Rossi L, Simon HU, et al. Fragility of the esophageal mucosa: a pathognomonic endoscopic sign of primary eosinophilic esophagitis. Gastrointest Endosc. 2003;57(3):407-412.
15. Hawari R, Pasricha PJ. Images in clinical medicine: eosinophilic esophagitis. N Engl J Med. 2007; 356(20):e20.
16. Leclercq P, Marting A, Gast P. Eosinophilic esophagitis. N Engl J Med. 2007;357(14):1446.
1. McQuaid KR. Gastrointestinal disorders. In: McPhee S, Papadakis M. CURRENT Medical Diagnosis & Treatment 2009. New York: McGraw-Hill: 2009:487-581.
2. Nurko S, Furuta GT. Eosinophilic esophagitis (2006). GI Motility Online. www.nature.com/gimo/contents/pt1/full/gimo49.html. Accessed July 27, 2009.
3. Parfitt JR, Gregor JC, Suskin NG, et al. Eosinophilic esophagitis in adults: distinguishing features from gastroesophageal reflux disease: a study of 41 patients. Mod Pathol. 2006;19(1):90-96.
4. Swoger JM, Weiler CR, Arora AS. Eosinophilic esophagitis: is it all allergies? Mayo Clin Proc. 2007;82(12):1541-1549.
5. Conus S, Simon HU. General laboratory diagnostics of eosinophilic GI diseases. Best Pract Res Clin Gastroenterol. 2008;22(3):441-453.
6. Remedios M, Campbell C, Jones DM, Kerlin P. Eosinophilic esophagitis in adults: clinical, endoscopic, histologic findings, and response to treatment with fluticasone propionate. Gastrointest Endosc. 2006;63(1):3-12.
7. Rothenberg ME. Eosinophilic gastrointestinal disorders (EGID). J Allergy Clin Immunol. 2004; 113(1):11-28.
8. Lucendo AJ, Carrion G, Navarro M, et al. Eosinophilic esophagitis in adults: an emerging disease. Dig Dis Sci. 2004;49(11-12):1884-1888.
9. Spieker MR. Evaluating dysphagia. Am Fam Physician. 2000;61(12):3639-3648.
10. Boyce HW. Drug-induced esophageal damage: diseases of medical progress. Gastrointest Endosc. 1998;47:547-550.
11. Potter JW, Saeian K, Staff D, et al. Eosinophilic esophagitis in adults: an emerging problem with unique esophageal features. Gastrointest Endosc. 2004;59(3):355-361.
12. Schaefer ET, Fitzgerald JF, Molleston JP, et al. Comparison of oral prednisone and topical fluticasone in the treatment of eosinophilic esophagitis: a randomized trial in children. Clin Gastroenterol Hepatol. 2008;6(2):165-173.
13. Furuta GT, Liacouras CA, Collins MH, et al; First International Gastrointestinal Eosinophil Research Symposium (FIGERS) Subcommittees. Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendation for diagnosis and treatment. Gastroenterology. 2007;133(4): 1342-1363.
14. Straumann A, Rossi L, Simon HU, et al. Fragility of the esophageal mucosa: a pathognomonic endoscopic sign of primary eosinophilic esophagitis. Gastrointest Endosc. 2003;57(3):407-412.
15. Hawari R, Pasricha PJ. Images in clinical medicine: eosinophilic esophagitis. N Engl J Med. 2007; 356(20):e20.
16. Leclercq P, Marting A, Gast P. Eosinophilic esophagitis. N Engl J Med. 2007;357(14):1446.