User login
Flexibility and speed mark the race to a COVID-19 vaccine
Extraordinary cooperation and accommodations are needed in the race to build a COVID-19 vaccine from scratch while chasing a pandemic, said members of industry and government who convened for an update on the vaccine clinical trial process.
The message came from representatives from the Food and Drug Administration, a think tank, and the nonprofit sector who provided some insight into the vaccine development process for COVID-19 at a press briefing hosted by the Commonwealth Fund.
Even in the best of times, vaccine development is not simple, said Litjen (L.J.) Tan, PhD, chief strategy officer for the Immunization Action Coalition. Ordinarily, the process can take from 10 to 20 years and cost well over a billion dollars. Many vaccines wind up being abandoned before phase 3 development just because the cost is prohibitive, he said.
Vaccines undergo extensive ongoing postmarketing surveillance even after approval, licensing, and distribution, noted Dr. Tan, adding that the development of a safe, effective vaccine is a “very complicated, significant process.”
In these extraordinary times, many adjustments to the usual trial trajectory are needed, agreed all participants. To speed the process, the traditional vaccine trajectory is being accelerated and compressed; changes may include simultaneous rather than sequential clinical trials that are run in parallel. These trials may be optimized for multiple target populations at the same time, and run in different countries, explained Dr. Tan.
“We are likely going to use something called adaptive trial designs” in which results are gathered during the trial and used to modify the trial according to prespecified rules, he said. “These trials shift to accommodate data as it comes up.”
Regulatory agencies are actively engaging in the process much earlier than usual, with input including how to incentivize scaling up production of vaccines and ensuring that vaccines will be fairly and equitably distributed across the globe, he added.
Esther Krofah is the executive director of FasterCures, a center within the nonprofit Milken Institute. Currently 86 different active COVID-19 vaccine projects are underway, she said, with 6 currently in clinical trials and about two dozen more expected to enter the clinical trial phase by the summer of 2020.
Many of these projects will involve a smaller biotech company or an academic research group with deep knowledge of a particular immune strategy partnering with a large pharmaceutical company that has economic capacity and global resources and reach, said Ms. Krofah.
From a policy perspective, she said, it’s important for the FDA to have surge capacity with “enough arms, legs, and staff to actively provide input into clinical design and protocols of studies.” The goal is to be able to review data in real-time and provide rapid feedback as studies are occurring so adaptive clinical trial design can be implemented.
As COVID-19 vaccine trials are rolled out, necessary compromises may include incorporation of real world evidence in later clinical trial stages. “There is a way to do randomized trials in the real world in situations that come up like this,” said Peter Marks, MD, the FDA’s director of the Center for Biologics Evaluation and Research. Long-term data about vaccine efficacy may be accrued over time, once a vaccine is being administered beyond the clinical trial stage, to see if efficacy wanes over time.
However, he said, “a lot will depend on where the vaccine is and what we encounter with whether there’s circulating virus or not” in terms of clinical trial design, including whether surrogate efficacy markers such as antibody production would be used.
Unvaccinated members of a population may be used as controls against a vaccinated group during an active outbreak, he said, a break from traditional trial design. “It’s not perfect. I’ll acknowledge right away there are certain people at the [National Institutes of Health] that would like to tell me that’s a horrible idea, but I think we are going to entertain and discuss all potential designs” for COVID-19 vaccine trials, he said. “We can’t out-of-hand dismiss any design here, whether real world based or evidence based.”
“This may be a little unusual hearing this from the FDA,” he said, “but this is possibly one of the most important things we are going to have to do in the next few years.” He added, “It’s unfortunately not unlikely that we will see a second wave, or maybe even a third wave, if we don’t get it right.”
He pointed out that there was no candidate vaccine in the pipeline when the pandemic blossomed and began its race around the globe. Current coronavirus candidates aren’t useful against COVID-19. “We know that there might be some complexities in development” related to the contributions of immune enhancement to the pathogenicity of the SARS-CoV-2 virus, he said.
The agency is currently working closely with sponsors of various candidate vaccines to take a hard look at preclinical data and trial design.
“Low efficacy could distract from capacity for more robust candidates to come forward,” he said. “We’d like to encourage people to have the absolute best vaccines. ... We hope to be able to facilitate rapid development of these,” he said.
Moving forward, a whole-government approach is necessary for development and delivery of the best vaccine. Ms. Krofah elaborated that the FDA and the Centers for Medicare & Medicaid will need to collaborate closely as studies evolve. In particular, CMS needs to be ready with reimbursement codes, recognizing that both public and private payers will likely be providing reimbursement for COVID-19 vaccinations.
“When these vaccines get approved, we are going to have to decide who is going to pay for them,” said Dr. Tan, who previously served as the director of medicine and public health at the American Medical Association. He noted that CPT coding comes through the AMA.
Dr. Marks stressed that ultimately, although treatments or prophylactic regimes against COVID-19 may be developed, “a vaccine is the most efficient way to protect large numbers of people.” Though there’s going to have to be a balancing act so speed doesn’t come at the expense of vaccine safety and efficacy. “We are very much hoping that we can find vaccines that have relatively high levels of efficacy,” he said, adding a hopeful point: “Right now there have not been mutations that have occurred … that would alter vaccine development programs in terms of the targets they’re going after.”
Dr. Marks offered a wildly optimistic – and then a more realistic – judgment as to when a successful vaccine might emerge from the development, trial, and approval process. After acknowledging that the FDA might consider an Emergency Use Authorization (EUA) if coronavirus activity is high when a candidate vaccine emerges, he said that “if everything goes perfectly,” an EUA for a vaccine might be issued within 9-12 months.
“The most likely timeline – and this is total speculation – is that it could be 12-18 months,” he said.
Ms. Krofah concurred, adding, “We’re seeing the big companies put a big bet on that timeline as well.” She cited Johnson & Johnson, which has committed to a $1 billion COVID-19 vaccine development program by the end of 2021.
Finally, when a vaccine does become available, who is first in line to receive it? Ms. Krofah said that it will be important for the public to know that there will be a tiering scheme for vaccine administration in the early days. Healthcare workers, emergency responders, and the particularly vulnerable may be among the first to receive protection, she said.
Extraordinary cooperation and accommodations are needed in the race to build a COVID-19 vaccine from scratch while chasing a pandemic, said members of industry and government who convened for an update on the vaccine clinical trial process.
The message came from representatives from the Food and Drug Administration, a think tank, and the nonprofit sector who provided some insight into the vaccine development process for COVID-19 at a press briefing hosted by the Commonwealth Fund.
Even in the best of times, vaccine development is not simple, said Litjen (L.J.) Tan, PhD, chief strategy officer for the Immunization Action Coalition. Ordinarily, the process can take from 10 to 20 years and cost well over a billion dollars. Many vaccines wind up being abandoned before phase 3 development just because the cost is prohibitive, he said.
Vaccines undergo extensive ongoing postmarketing surveillance even after approval, licensing, and distribution, noted Dr. Tan, adding that the development of a safe, effective vaccine is a “very complicated, significant process.”
In these extraordinary times, many adjustments to the usual trial trajectory are needed, agreed all participants. To speed the process, the traditional vaccine trajectory is being accelerated and compressed; changes may include simultaneous rather than sequential clinical trials that are run in parallel. These trials may be optimized for multiple target populations at the same time, and run in different countries, explained Dr. Tan.
“We are likely going to use something called adaptive trial designs” in which results are gathered during the trial and used to modify the trial according to prespecified rules, he said. “These trials shift to accommodate data as it comes up.”
Regulatory agencies are actively engaging in the process much earlier than usual, with input including how to incentivize scaling up production of vaccines and ensuring that vaccines will be fairly and equitably distributed across the globe, he added.
Esther Krofah is the executive director of FasterCures, a center within the nonprofit Milken Institute. Currently 86 different active COVID-19 vaccine projects are underway, she said, with 6 currently in clinical trials and about two dozen more expected to enter the clinical trial phase by the summer of 2020.
Many of these projects will involve a smaller biotech company or an academic research group with deep knowledge of a particular immune strategy partnering with a large pharmaceutical company that has economic capacity and global resources and reach, said Ms. Krofah.
From a policy perspective, she said, it’s important for the FDA to have surge capacity with “enough arms, legs, and staff to actively provide input into clinical design and protocols of studies.” The goal is to be able to review data in real-time and provide rapid feedback as studies are occurring so adaptive clinical trial design can be implemented.
As COVID-19 vaccine trials are rolled out, necessary compromises may include incorporation of real world evidence in later clinical trial stages. “There is a way to do randomized trials in the real world in situations that come up like this,” said Peter Marks, MD, the FDA’s director of the Center for Biologics Evaluation and Research. Long-term data about vaccine efficacy may be accrued over time, once a vaccine is being administered beyond the clinical trial stage, to see if efficacy wanes over time.
However, he said, “a lot will depend on where the vaccine is and what we encounter with whether there’s circulating virus or not” in terms of clinical trial design, including whether surrogate efficacy markers such as antibody production would be used.
Unvaccinated members of a population may be used as controls against a vaccinated group during an active outbreak, he said, a break from traditional trial design. “It’s not perfect. I’ll acknowledge right away there are certain people at the [National Institutes of Health] that would like to tell me that’s a horrible idea, but I think we are going to entertain and discuss all potential designs” for COVID-19 vaccine trials, he said. “We can’t out-of-hand dismiss any design here, whether real world based or evidence based.”
“This may be a little unusual hearing this from the FDA,” he said, “but this is possibly one of the most important things we are going to have to do in the next few years.” He added, “It’s unfortunately not unlikely that we will see a second wave, or maybe even a third wave, if we don’t get it right.”
He pointed out that there was no candidate vaccine in the pipeline when the pandemic blossomed and began its race around the globe. Current coronavirus candidates aren’t useful against COVID-19. “We know that there might be some complexities in development” related to the contributions of immune enhancement to the pathogenicity of the SARS-CoV-2 virus, he said.
The agency is currently working closely with sponsors of various candidate vaccines to take a hard look at preclinical data and trial design.
“Low efficacy could distract from capacity for more robust candidates to come forward,” he said. “We’d like to encourage people to have the absolute best vaccines. ... We hope to be able to facilitate rapid development of these,” he said.
Moving forward, a whole-government approach is necessary for development and delivery of the best vaccine. Ms. Krofah elaborated that the FDA and the Centers for Medicare & Medicaid will need to collaborate closely as studies evolve. In particular, CMS needs to be ready with reimbursement codes, recognizing that both public and private payers will likely be providing reimbursement for COVID-19 vaccinations.
“When these vaccines get approved, we are going to have to decide who is going to pay for them,” said Dr. Tan, who previously served as the director of medicine and public health at the American Medical Association. He noted that CPT coding comes through the AMA.
Dr. Marks stressed that ultimately, although treatments or prophylactic regimes against COVID-19 may be developed, “a vaccine is the most efficient way to protect large numbers of people.” Though there’s going to have to be a balancing act so speed doesn’t come at the expense of vaccine safety and efficacy. “We are very much hoping that we can find vaccines that have relatively high levels of efficacy,” he said, adding a hopeful point: “Right now there have not been mutations that have occurred … that would alter vaccine development programs in terms of the targets they’re going after.”
Dr. Marks offered a wildly optimistic – and then a more realistic – judgment as to when a successful vaccine might emerge from the development, trial, and approval process. After acknowledging that the FDA might consider an Emergency Use Authorization (EUA) if coronavirus activity is high when a candidate vaccine emerges, he said that “if everything goes perfectly,” an EUA for a vaccine might be issued within 9-12 months.
“The most likely timeline – and this is total speculation – is that it could be 12-18 months,” he said.
Ms. Krofah concurred, adding, “We’re seeing the big companies put a big bet on that timeline as well.” She cited Johnson & Johnson, which has committed to a $1 billion COVID-19 vaccine development program by the end of 2021.
Finally, when a vaccine does become available, who is first in line to receive it? Ms. Krofah said that it will be important for the public to know that there will be a tiering scheme for vaccine administration in the early days. Healthcare workers, emergency responders, and the particularly vulnerable may be among the first to receive protection, she said.
Extraordinary cooperation and accommodations are needed in the race to build a COVID-19 vaccine from scratch while chasing a pandemic, said members of industry and government who convened for an update on the vaccine clinical trial process.
The message came from representatives from the Food and Drug Administration, a think tank, and the nonprofit sector who provided some insight into the vaccine development process for COVID-19 at a press briefing hosted by the Commonwealth Fund.
Even in the best of times, vaccine development is not simple, said Litjen (L.J.) Tan, PhD, chief strategy officer for the Immunization Action Coalition. Ordinarily, the process can take from 10 to 20 years and cost well over a billion dollars. Many vaccines wind up being abandoned before phase 3 development just because the cost is prohibitive, he said.
Vaccines undergo extensive ongoing postmarketing surveillance even after approval, licensing, and distribution, noted Dr. Tan, adding that the development of a safe, effective vaccine is a “very complicated, significant process.”
In these extraordinary times, many adjustments to the usual trial trajectory are needed, agreed all participants. To speed the process, the traditional vaccine trajectory is being accelerated and compressed; changes may include simultaneous rather than sequential clinical trials that are run in parallel. These trials may be optimized for multiple target populations at the same time, and run in different countries, explained Dr. Tan.
“We are likely going to use something called adaptive trial designs” in which results are gathered during the trial and used to modify the trial according to prespecified rules, he said. “These trials shift to accommodate data as it comes up.”
Regulatory agencies are actively engaging in the process much earlier than usual, with input including how to incentivize scaling up production of vaccines and ensuring that vaccines will be fairly and equitably distributed across the globe, he added.
Esther Krofah is the executive director of FasterCures, a center within the nonprofit Milken Institute. Currently 86 different active COVID-19 vaccine projects are underway, she said, with 6 currently in clinical trials and about two dozen more expected to enter the clinical trial phase by the summer of 2020.
Many of these projects will involve a smaller biotech company or an academic research group with deep knowledge of a particular immune strategy partnering with a large pharmaceutical company that has economic capacity and global resources and reach, said Ms. Krofah.
From a policy perspective, she said, it’s important for the FDA to have surge capacity with “enough arms, legs, and staff to actively provide input into clinical design and protocols of studies.” The goal is to be able to review data in real-time and provide rapid feedback as studies are occurring so adaptive clinical trial design can be implemented.
As COVID-19 vaccine trials are rolled out, necessary compromises may include incorporation of real world evidence in later clinical trial stages. “There is a way to do randomized trials in the real world in situations that come up like this,” said Peter Marks, MD, the FDA’s director of the Center for Biologics Evaluation and Research. Long-term data about vaccine efficacy may be accrued over time, once a vaccine is being administered beyond the clinical trial stage, to see if efficacy wanes over time.
However, he said, “a lot will depend on where the vaccine is and what we encounter with whether there’s circulating virus or not” in terms of clinical trial design, including whether surrogate efficacy markers such as antibody production would be used.
Unvaccinated members of a population may be used as controls against a vaccinated group during an active outbreak, he said, a break from traditional trial design. “It’s not perfect. I’ll acknowledge right away there are certain people at the [National Institutes of Health] that would like to tell me that’s a horrible idea, but I think we are going to entertain and discuss all potential designs” for COVID-19 vaccine trials, he said. “We can’t out-of-hand dismiss any design here, whether real world based or evidence based.”
“This may be a little unusual hearing this from the FDA,” he said, “but this is possibly one of the most important things we are going to have to do in the next few years.” He added, “It’s unfortunately not unlikely that we will see a second wave, or maybe even a third wave, if we don’t get it right.”
He pointed out that there was no candidate vaccine in the pipeline when the pandemic blossomed and began its race around the globe. Current coronavirus candidates aren’t useful against COVID-19. “We know that there might be some complexities in development” related to the contributions of immune enhancement to the pathogenicity of the SARS-CoV-2 virus, he said.
The agency is currently working closely with sponsors of various candidate vaccines to take a hard look at preclinical data and trial design.
“Low efficacy could distract from capacity for more robust candidates to come forward,” he said. “We’d like to encourage people to have the absolute best vaccines. ... We hope to be able to facilitate rapid development of these,” he said.
Moving forward, a whole-government approach is necessary for development and delivery of the best vaccine. Ms. Krofah elaborated that the FDA and the Centers for Medicare & Medicaid will need to collaborate closely as studies evolve. In particular, CMS needs to be ready with reimbursement codes, recognizing that both public and private payers will likely be providing reimbursement for COVID-19 vaccinations.
“When these vaccines get approved, we are going to have to decide who is going to pay for them,” said Dr. Tan, who previously served as the director of medicine and public health at the American Medical Association. He noted that CPT coding comes through the AMA.
Dr. Marks stressed that ultimately, although treatments or prophylactic regimes against COVID-19 may be developed, “a vaccine is the most efficient way to protect large numbers of people.” Though there’s going to have to be a balancing act so speed doesn’t come at the expense of vaccine safety and efficacy. “We are very much hoping that we can find vaccines that have relatively high levels of efficacy,” he said, adding a hopeful point: “Right now there have not been mutations that have occurred … that would alter vaccine development programs in terms of the targets they’re going after.”
Dr. Marks offered a wildly optimistic – and then a more realistic – judgment as to when a successful vaccine might emerge from the development, trial, and approval process. After acknowledging that the FDA might consider an Emergency Use Authorization (EUA) if coronavirus activity is high when a candidate vaccine emerges, he said that “if everything goes perfectly,” an EUA for a vaccine might be issued within 9-12 months.
“The most likely timeline – and this is total speculation – is that it could be 12-18 months,” he said.
Ms. Krofah concurred, adding, “We’re seeing the big companies put a big bet on that timeline as well.” She cited Johnson & Johnson, which has committed to a $1 billion COVID-19 vaccine development program by the end of 2021.
Finally, when a vaccine does become available, who is first in line to receive it? Ms. Krofah said that it will be important for the public to know that there will be a tiering scheme for vaccine administration in the early days. Healthcare workers, emergency responders, and the particularly vulnerable may be among the first to receive protection, she said.
Researchers investigate impact of smoking on COVID-19 risk
but quitting smoking is likely to lower the risk of developing more severe or fatal cases of the infection, according to research from several recent papers.
Interest in how tobacco use affects COVID-19 infection rates stems from research showing that men at the epicenter of the outbreak in China having a higher early mortality rate. Early reports from China showed a case fatality rate of 4.7% for men, compared with 2.8% for women, according to the World Health Organization. The virus that causes COVID-19, severe acute respiratory syndrome coronavirus 2, is suspected to enter a cell using the ACE2 receptor. Since smoking up-regulates this receptor, one popular theory is that smoking can increase the risk of COVID-19 or exacerbate symptoms of an existing infection (Eur Respir J. 2020 Apr 8. doi: 10.1183/13993003.00688-2020). In China, about half of men are active smokers, compared with 2.7% of women (Transl Lung Cancer Res. 2019;8[Suppl 1]:S21-30), so this association would explain the severe cases and increased mortality in this group. In response to potential risk for public health, the World Health Organization, Centers for Disease Control and Prevention, the Attorney General of Massachusetts, and other organizations have warned that smoking may increase one’s risk of transmitting and developing COVID-19 or may worsen the infection.
“While it is easy to jump to the conclusion that more ACE2 means more susceptibility to severe infection, there is no evidence to support this,” Brandon Michael Henry, MD, of the cardiac intensive care unit and the Heart Institute at Cincinnati Children’s Hospital Medical Center, said in an interview. “Moreover, some would argue (including myself) that increased ACE2 may in fact be protective, as ACE2 decreases the levels of angiotensin-2 which likely plays a significant role in the pathophysiology of ARDS.”
Some researchers have examined the limited evidence of smoking on COVID-19 risk and come to preliminary conclusions. In a letter to the editor recently published in the European Journal of Internal Medicine, Dr. Henry and Giuseppe Lippi, MD, of the section of clinical biochemistry in the department of neuroscience, biomedicine, and movement at the University of Verona (Italy), performed a meta-analysis of papers examining smoking and COVID-19 up to March 9, 2020 and identified five articles with 1,399 COVID-19 cases (Eur J Intern Med. 2020 Mar 16. doi: 10.1016/j.ejim.2020.03.014).
“Given the fact that COVID-19 is a primarily respiratory illness, smoking was one of first risk factors we examined,” Dr. Henry said.
They noted that a study by Liu et al. in the Chinese Medical Journal was the only paper that showed a significant association between smoking status and COVID-19 case severity (Chin Med J [Engl]. 2020 Feb 28. doi: 10.1097/CM9.0000000000000775), while the four other studies showed no significant association. The pooled data of all five studies showed an association that was not statistically significant (odds ratio, 1.69; 95% confidence interval, 0.41-6.92; P = .254). When Dr. Lippi and Dr. Henry performed the analysis again after removing a paper by Guan et al. (N Engl J Med. 2020 Feb 28. doi: 10.1056/NEJMoa2002032) comprising 89.5% of patients in the pooled analysis, there was no significant association (OR, 4.35; 95% CI, 0.86-21.86; P = .129).
Constantine I. Vardavas, MD, FCCP, of the department of oral health policy and epidemiology at Harvard School of Dental Medicine, Boston, and Katerina Nikitara, of the University of Crete in Heraklion, Greece, also published a systematic review in Tobacco Induced Diseases of five studies evaluating smoking and COVID-19 (Tob Induc Dis. 2020. doi: 10.18332/tid/119324). Of the studies chosen for the review, four were shared with the paper by Dr. Lippi and Dr. Henry. They found “a higher percentage of smokers” made up severe COVID-19 cases, but acknowledged the majority of these were from the largest study by Guan et al. Overall, they calculated smokers carried a risk ratio of 1.4 (95% CI, 0.98-2.00) for developing severe COVID-19 symptoms, and were over twice as likely to be admitted to an ICU, require a mechanical ventilator, or die from COVID-19, compared with patients who did not smoke (RR, 2.4; 95% CI, 1.43-4.04).
“Although further research is warranted as the weight of the evidence increases, with the limited available data, and although the above results are unadjusted for other factors that may impact disease progression, smoking is most likely associated with the negative progression and adverse outcomes of COVID-19,” Dr. Vardavas and Ms. Nikitara concluded.
However, the association between smoking and severe disease was not significant, and it is not immediately clear how the analysis was performed based on the details in the editorial. “Both of our reports were limited by a lack of data adjusted for age, sex, and comorbidities which may influence any analysis on smoking,” Dr. Henry said.
Some researchers have proposed collecting information on smoking status and conducting further research on whether vaping devices like e-cigarettes also impact COVID-19 cases. An editorial by Samuel Brake and colleagues published in the Journal of Clinical Medicine proposed the ACE2-receptor binding site as an area of interest for COVID-19 and as a potential therapeutic target (J Clin Med. 2020 Mar 20. doi: 10.3390/jcm9030841).
Ultimately, whether smoking itself is associated with COVID-19 is still an open question. Nonetheless, encouraging patients to quit smoking should be a priority because long-term sequelae of smoking have been linked to worsened or fatal COVID-19 cases, said Dr. Henry.
“There is a lack of definitive data on smoking to date. Nonetheless, we do know that many illnesses associated with smoking, such as [chronic obstructive pulmonary disease, hypertension, and heart disease are all strong risk factors for severe and fatal COVID-19,” he said. “Thus, absolutely we should encourage the public to quit smoking, especially for older individuals and those with comorbidities.”
The papers by Lippi et al., Vardavas et al., and Brake et al. had no funding source, and the authors reported no relevant conflicts of interest.
but quitting smoking is likely to lower the risk of developing more severe or fatal cases of the infection, according to research from several recent papers.
Interest in how tobacco use affects COVID-19 infection rates stems from research showing that men at the epicenter of the outbreak in China having a higher early mortality rate. Early reports from China showed a case fatality rate of 4.7% for men, compared with 2.8% for women, according to the World Health Organization. The virus that causes COVID-19, severe acute respiratory syndrome coronavirus 2, is suspected to enter a cell using the ACE2 receptor. Since smoking up-regulates this receptor, one popular theory is that smoking can increase the risk of COVID-19 or exacerbate symptoms of an existing infection (Eur Respir J. 2020 Apr 8. doi: 10.1183/13993003.00688-2020). In China, about half of men are active smokers, compared with 2.7% of women (Transl Lung Cancer Res. 2019;8[Suppl 1]:S21-30), so this association would explain the severe cases and increased mortality in this group. In response to potential risk for public health, the World Health Organization, Centers for Disease Control and Prevention, the Attorney General of Massachusetts, and other organizations have warned that smoking may increase one’s risk of transmitting and developing COVID-19 or may worsen the infection.
“While it is easy to jump to the conclusion that more ACE2 means more susceptibility to severe infection, there is no evidence to support this,” Brandon Michael Henry, MD, of the cardiac intensive care unit and the Heart Institute at Cincinnati Children’s Hospital Medical Center, said in an interview. “Moreover, some would argue (including myself) that increased ACE2 may in fact be protective, as ACE2 decreases the levels of angiotensin-2 which likely plays a significant role in the pathophysiology of ARDS.”
Some researchers have examined the limited evidence of smoking on COVID-19 risk and come to preliminary conclusions. In a letter to the editor recently published in the European Journal of Internal Medicine, Dr. Henry and Giuseppe Lippi, MD, of the section of clinical biochemistry in the department of neuroscience, biomedicine, and movement at the University of Verona (Italy), performed a meta-analysis of papers examining smoking and COVID-19 up to March 9, 2020 and identified five articles with 1,399 COVID-19 cases (Eur J Intern Med. 2020 Mar 16. doi: 10.1016/j.ejim.2020.03.014).
“Given the fact that COVID-19 is a primarily respiratory illness, smoking was one of first risk factors we examined,” Dr. Henry said.
They noted that a study by Liu et al. in the Chinese Medical Journal was the only paper that showed a significant association between smoking status and COVID-19 case severity (Chin Med J [Engl]. 2020 Feb 28. doi: 10.1097/CM9.0000000000000775), while the four other studies showed no significant association. The pooled data of all five studies showed an association that was not statistically significant (odds ratio, 1.69; 95% confidence interval, 0.41-6.92; P = .254). When Dr. Lippi and Dr. Henry performed the analysis again after removing a paper by Guan et al. (N Engl J Med. 2020 Feb 28. doi: 10.1056/NEJMoa2002032) comprising 89.5% of patients in the pooled analysis, there was no significant association (OR, 4.35; 95% CI, 0.86-21.86; P = .129).
Constantine I. Vardavas, MD, FCCP, of the department of oral health policy and epidemiology at Harvard School of Dental Medicine, Boston, and Katerina Nikitara, of the University of Crete in Heraklion, Greece, also published a systematic review in Tobacco Induced Diseases of five studies evaluating smoking and COVID-19 (Tob Induc Dis. 2020. doi: 10.18332/tid/119324). Of the studies chosen for the review, four were shared with the paper by Dr. Lippi and Dr. Henry. They found “a higher percentage of smokers” made up severe COVID-19 cases, but acknowledged the majority of these were from the largest study by Guan et al. Overall, they calculated smokers carried a risk ratio of 1.4 (95% CI, 0.98-2.00) for developing severe COVID-19 symptoms, and were over twice as likely to be admitted to an ICU, require a mechanical ventilator, or die from COVID-19, compared with patients who did not smoke (RR, 2.4; 95% CI, 1.43-4.04).
“Although further research is warranted as the weight of the evidence increases, with the limited available data, and although the above results are unadjusted for other factors that may impact disease progression, smoking is most likely associated with the negative progression and adverse outcomes of COVID-19,” Dr. Vardavas and Ms. Nikitara concluded.
However, the association between smoking and severe disease was not significant, and it is not immediately clear how the analysis was performed based on the details in the editorial. “Both of our reports were limited by a lack of data adjusted for age, sex, and comorbidities which may influence any analysis on smoking,” Dr. Henry said.
Some researchers have proposed collecting information on smoking status and conducting further research on whether vaping devices like e-cigarettes also impact COVID-19 cases. An editorial by Samuel Brake and colleagues published in the Journal of Clinical Medicine proposed the ACE2-receptor binding site as an area of interest for COVID-19 and as a potential therapeutic target (J Clin Med. 2020 Mar 20. doi: 10.3390/jcm9030841).
Ultimately, whether smoking itself is associated with COVID-19 is still an open question. Nonetheless, encouraging patients to quit smoking should be a priority because long-term sequelae of smoking have been linked to worsened or fatal COVID-19 cases, said Dr. Henry.
“There is a lack of definitive data on smoking to date. Nonetheless, we do know that many illnesses associated with smoking, such as [chronic obstructive pulmonary disease, hypertension, and heart disease are all strong risk factors for severe and fatal COVID-19,” he said. “Thus, absolutely we should encourage the public to quit smoking, especially for older individuals and those with comorbidities.”
The papers by Lippi et al., Vardavas et al., and Brake et al. had no funding source, and the authors reported no relevant conflicts of interest.
but quitting smoking is likely to lower the risk of developing more severe or fatal cases of the infection, according to research from several recent papers.
Interest in how tobacco use affects COVID-19 infection rates stems from research showing that men at the epicenter of the outbreak in China having a higher early mortality rate. Early reports from China showed a case fatality rate of 4.7% for men, compared with 2.8% for women, according to the World Health Organization. The virus that causes COVID-19, severe acute respiratory syndrome coronavirus 2, is suspected to enter a cell using the ACE2 receptor. Since smoking up-regulates this receptor, one popular theory is that smoking can increase the risk of COVID-19 or exacerbate symptoms of an existing infection (Eur Respir J. 2020 Apr 8. doi: 10.1183/13993003.00688-2020). In China, about half of men are active smokers, compared with 2.7% of women (Transl Lung Cancer Res. 2019;8[Suppl 1]:S21-30), so this association would explain the severe cases and increased mortality in this group. In response to potential risk for public health, the World Health Organization, Centers for Disease Control and Prevention, the Attorney General of Massachusetts, and other organizations have warned that smoking may increase one’s risk of transmitting and developing COVID-19 or may worsen the infection.
“While it is easy to jump to the conclusion that more ACE2 means more susceptibility to severe infection, there is no evidence to support this,” Brandon Michael Henry, MD, of the cardiac intensive care unit and the Heart Institute at Cincinnati Children’s Hospital Medical Center, said in an interview. “Moreover, some would argue (including myself) that increased ACE2 may in fact be protective, as ACE2 decreases the levels of angiotensin-2 which likely plays a significant role in the pathophysiology of ARDS.”
Some researchers have examined the limited evidence of smoking on COVID-19 risk and come to preliminary conclusions. In a letter to the editor recently published in the European Journal of Internal Medicine, Dr. Henry and Giuseppe Lippi, MD, of the section of clinical biochemistry in the department of neuroscience, biomedicine, and movement at the University of Verona (Italy), performed a meta-analysis of papers examining smoking and COVID-19 up to March 9, 2020 and identified five articles with 1,399 COVID-19 cases (Eur J Intern Med. 2020 Mar 16. doi: 10.1016/j.ejim.2020.03.014).
“Given the fact that COVID-19 is a primarily respiratory illness, smoking was one of first risk factors we examined,” Dr. Henry said.
They noted that a study by Liu et al. in the Chinese Medical Journal was the only paper that showed a significant association between smoking status and COVID-19 case severity (Chin Med J [Engl]. 2020 Feb 28. doi: 10.1097/CM9.0000000000000775), while the four other studies showed no significant association. The pooled data of all five studies showed an association that was not statistically significant (odds ratio, 1.69; 95% confidence interval, 0.41-6.92; P = .254). When Dr. Lippi and Dr. Henry performed the analysis again after removing a paper by Guan et al. (N Engl J Med. 2020 Feb 28. doi: 10.1056/NEJMoa2002032) comprising 89.5% of patients in the pooled analysis, there was no significant association (OR, 4.35; 95% CI, 0.86-21.86; P = .129).
Constantine I. Vardavas, MD, FCCP, of the department of oral health policy and epidemiology at Harvard School of Dental Medicine, Boston, and Katerina Nikitara, of the University of Crete in Heraklion, Greece, also published a systematic review in Tobacco Induced Diseases of five studies evaluating smoking and COVID-19 (Tob Induc Dis. 2020. doi: 10.18332/tid/119324). Of the studies chosen for the review, four were shared with the paper by Dr. Lippi and Dr. Henry. They found “a higher percentage of smokers” made up severe COVID-19 cases, but acknowledged the majority of these were from the largest study by Guan et al. Overall, they calculated smokers carried a risk ratio of 1.4 (95% CI, 0.98-2.00) for developing severe COVID-19 symptoms, and were over twice as likely to be admitted to an ICU, require a mechanical ventilator, or die from COVID-19, compared with patients who did not smoke (RR, 2.4; 95% CI, 1.43-4.04).
“Although further research is warranted as the weight of the evidence increases, with the limited available data, and although the above results are unadjusted for other factors that may impact disease progression, smoking is most likely associated with the negative progression and adverse outcomes of COVID-19,” Dr. Vardavas and Ms. Nikitara concluded.
However, the association between smoking and severe disease was not significant, and it is not immediately clear how the analysis was performed based on the details in the editorial. “Both of our reports were limited by a lack of data adjusted for age, sex, and comorbidities which may influence any analysis on smoking,” Dr. Henry said.
Some researchers have proposed collecting information on smoking status and conducting further research on whether vaping devices like e-cigarettes also impact COVID-19 cases. An editorial by Samuel Brake and colleagues published in the Journal of Clinical Medicine proposed the ACE2-receptor binding site as an area of interest for COVID-19 and as a potential therapeutic target (J Clin Med. 2020 Mar 20. doi: 10.3390/jcm9030841).
Ultimately, whether smoking itself is associated with COVID-19 is still an open question. Nonetheless, encouraging patients to quit smoking should be a priority because long-term sequelae of smoking have been linked to worsened or fatal COVID-19 cases, said Dr. Henry.
“There is a lack of definitive data on smoking to date. Nonetheless, we do know that many illnesses associated with smoking, such as [chronic obstructive pulmonary disease, hypertension, and heart disease are all strong risk factors for severe and fatal COVID-19,” he said. “Thus, absolutely we should encourage the public to quit smoking, especially for older individuals and those with comorbidities.”
The papers by Lippi et al., Vardavas et al., and Brake et al. had no funding source, and the authors reported no relevant conflicts of interest.
Switching gears at high speed
Michigan hospitalists prepare for COVID-19 care
When March began, Valerie Vaughn, MD, split her time between caring for general inpatients at the University of Michigan’s hospitals in Ann Arbor and doing research on how to reduce overuse of antibiotics in hospitals nationwide.
By the time the month was over, she had helped create a new kind of hospital team focused on caring for patients with COVID-19, learned how to provide an intensive level of care for the sickest among them, trained hundreds of physicians in how to do the same, and created free online learning tools for physicians nationwide.
Call it switching gears while driving a race car. Changing horses in the middle of a raging river. Or going to medical boot camp. Whatever the metaphor, Dr. Vaughn and her colleagues did it.
And now they’re hoping that sharing what they learned will help others if their hospitals go through the same thing.
Near the epicenter
Michigan Medicine, the University of Michigan’s academic medical center, is a few dozen miles west of the Detroit hospitals that have become a national epicenter for COVID-19 cases. It’s gotten plenty of direct and transferred COVID-19 patients since mid-March.
When Dr. Vaughn’s boss, division of hospital medicine chief Vineet Chopra, MD, was tapped to lead the creation of an all-COVID unit, he asked Dr. Vaughn to work with him and the team of hospitalists, nurse practitioners, physician assistants, nurses, respiratory therapists, and other staff that had volunteered for the team.
They had 3 days to prepare.
The “SWAT team”, as Dr. Vaughn calls it, opened the RICU, or Regional Infectious Containment Unit, on March 16. They doubled the number of beds 2 weeks later.
By the end of March, the team had handed over the reins to a team of experienced intensive care professionals so the unit could focus on the sickest patients. And the RICU team had moved on to transforming other areas of the hospital, and training their staff, in the same way.
By early April, more than 200 beds across the University of Michigan’s hospitals were devoted to COVID-19 care. General medicine physicians who hadn’t practiced inside a hospital since their residency days – thanks to the ability to hand off to hospitalists – were being pulled into inpatient duty. Hospitalists were being pulled into caring for patients who would normally have been in the care of an intensive care team.
“What’s amazed me most is how much people have stepped up to the challenge,” says Dr. Vaughn. “As hard and uncomfortable as it is to do something you’re not typically doing, it can also be therapeutic to say how can I help, let me do something. Yes, they’re anxious, but they want to know how they can be as prepared as they can be, to be as helpful as possible to these patients.”
Dr. Chopra agrees. “The silver lining in all of this is that I have personally seen the best in us come to the surface. Nurses, physicians, pharmacists, and therapists have come together and have shown selflessness, kindness, empathy and resilience in profound ways.”
Making the leap
Even though they didn’t choose hospital medicine, or ICU medicine, as their specialty, physicians may greatly underestimate how useful they can be with a little just-in-time training and the help of residents, fellows, advanced practice providers, and experienced nurses and respiratory therapists.
That training is now available for free through Michigan Medicine’s new online COVID-19 CME portal. The session in “Inpatient Management of COVID-19 patients” provides an important overview for those who have never cared for a case, especially if they haven’t been on inpatient duty in a while. The ICU Bootcamp is for those who will be caring for sicker COVID-19 patients but haven’t practiced in an ICU for a while.
One of the most important roles of a COVID-19 inpatient physician, Dr. Vaughn notes, doesn’t involve new skills. Rather, it draws on the doctoring skills that general medicine and hospital medicine physicians have already honed: the ability to assess and treat the entire patient, to talk with families who can’t be with their loved ones, to humanize the experience for patients and their loved ones as much as possible, and to bring messages of love from the family back to the bedside.
By pairing a general medicine physician newly placed on inpatient duty with a resident, nurse practitioner, or physician assistant who can handle inpatient charting duties, the team can make the most of each kind of provider’s time. Administrators, too, can reduce the burden on the entire team by simplifying processes for what must be charted and recorded in the EMR.
“Hospitals facing a COVID-19 crunch need to make it easier for teams to focus on the medicine and the human connection” and to shorten the learning curve for those shifting into unfamiliar duties, she advises.
Other lessons learned
Placing COVID-19 patients on the same unit, and keeping non–COVID-19 patients in another area of the hospital, isn’t just a good idea for protecting uninfected patients, Dr. Vaughn notes. It’s also good for providers who are getting used to treating COVID-19 because they don’t have to shift between the needs of different types of patients as they go from room to room.
“The learning curve is steep, but after a couple of days taking care of these patients, you have a good feeling about how to care for them and a great sense of camaraderie with the rest of the team involved in caring for them,” she says. “Everyone jumps in to help because they know we’re in this as a team and that it’s OK for respiratory therapists to step up to help a physician who doesn’t know as much about ventilator care or for nurses to suggest medications based on what other physicians have used.”
The flattening of professional hierarchies long ingrained in hospitals may be a side effect of the tremendous and urgent sense of mission that has developed around responding to COVID-19, Dr. Vaughn notes.
Those stepping into new roles should invite their colleagues to alert them when they see them about to slip up on protective practices that might be new to them. Similarly, they should help each other resist the urge to rush into a COVID-19 patient’s room unprotected in order to help with an urgent situation. The safety of providers – to preserve their ability to care for the many more patients who will need them – must be paramount.
“To handle this pandemic, we need to all be all-in and working toward a common goal, without competing priorities,” she says. “We need to use everyone’s skill sets to the fullest, without creating burnout. We’re going to be different when all this is done.”
Avoiding provider burnout is harder than ever because team members caring for COVID-19 must stay apart from family at home and avoid in-person visits with loved ones and friends. Those who are switching to inpatient or ICU-level care should make a point of focusing on exercise, sleep, virtual connections with loved ones, and healthy eating in between shifts.
“You’re no good to anyone else if you’re not healthy,” Dr. Vaughn says. “Your mental and physical health have to come first because they enable you to help others.”
Paying attention to the appreciation that the community is showing health care workers can also brighten the day of a stressed COVID-19 inpatient clinician, she notes.
“All the little signs of love from the community – the thank you signs, sidewalk chalk drawings, hearts in people’s windows – really do help.”
This article is published courtesy of the University of Michigan Health Lab, where it appeared originally.
Michigan hospitalists prepare for COVID-19 care
Michigan hospitalists prepare for COVID-19 care
When March began, Valerie Vaughn, MD, split her time between caring for general inpatients at the University of Michigan’s hospitals in Ann Arbor and doing research on how to reduce overuse of antibiotics in hospitals nationwide.
By the time the month was over, she had helped create a new kind of hospital team focused on caring for patients with COVID-19, learned how to provide an intensive level of care for the sickest among them, trained hundreds of physicians in how to do the same, and created free online learning tools for physicians nationwide.
Call it switching gears while driving a race car. Changing horses in the middle of a raging river. Or going to medical boot camp. Whatever the metaphor, Dr. Vaughn and her colleagues did it.
And now they’re hoping that sharing what they learned will help others if their hospitals go through the same thing.
Near the epicenter
Michigan Medicine, the University of Michigan’s academic medical center, is a few dozen miles west of the Detroit hospitals that have become a national epicenter for COVID-19 cases. It’s gotten plenty of direct and transferred COVID-19 patients since mid-March.
When Dr. Vaughn’s boss, division of hospital medicine chief Vineet Chopra, MD, was tapped to lead the creation of an all-COVID unit, he asked Dr. Vaughn to work with him and the team of hospitalists, nurse practitioners, physician assistants, nurses, respiratory therapists, and other staff that had volunteered for the team.
They had 3 days to prepare.
The “SWAT team”, as Dr. Vaughn calls it, opened the RICU, or Regional Infectious Containment Unit, on March 16. They doubled the number of beds 2 weeks later.
By the end of March, the team had handed over the reins to a team of experienced intensive care professionals so the unit could focus on the sickest patients. And the RICU team had moved on to transforming other areas of the hospital, and training their staff, in the same way.
By early April, more than 200 beds across the University of Michigan’s hospitals were devoted to COVID-19 care. General medicine physicians who hadn’t practiced inside a hospital since their residency days – thanks to the ability to hand off to hospitalists – were being pulled into inpatient duty. Hospitalists were being pulled into caring for patients who would normally have been in the care of an intensive care team.
“What’s amazed me most is how much people have stepped up to the challenge,” says Dr. Vaughn. “As hard and uncomfortable as it is to do something you’re not typically doing, it can also be therapeutic to say how can I help, let me do something. Yes, they’re anxious, but they want to know how they can be as prepared as they can be, to be as helpful as possible to these patients.”
Dr. Chopra agrees. “The silver lining in all of this is that I have personally seen the best in us come to the surface. Nurses, physicians, pharmacists, and therapists have come together and have shown selflessness, kindness, empathy and resilience in profound ways.”
Making the leap
Even though they didn’t choose hospital medicine, or ICU medicine, as their specialty, physicians may greatly underestimate how useful they can be with a little just-in-time training and the help of residents, fellows, advanced practice providers, and experienced nurses and respiratory therapists.
That training is now available for free through Michigan Medicine’s new online COVID-19 CME portal. The session in “Inpatient Management of COVID-19 patients” provides an important overview for those who have never cared for a case, especially if they haven’t been on inpatient duty in a while. The ICU Bootcamp is for those who will be caring for sicker COVID-19 patients but haven’t practiced in an ICU for a while.
One of the most important roles of a COVID-19 inpatient physician, Dr. Vaughn notes, doesn’t involve new skills. Rather, it draws on the doctoring skills that general medicine and hospital medicine physicians have already honed: the ability to assess and treat the entire patient, to talk with families who can’t be with their loved ones, to humanize the experience for patients and their loved ones as much as possible, and to bring messages of love from the family back to the bedside.
By pairing a general medicine physician newly placed on inpatient duty with a resident, nurse practitioner, or physician assistant who can handle inpatient charting duties, the team can make the most of each kind of provider’s time. Administrators, too, can reduce the burden on the entire team by simplifying processes for what must be charted and recorded in the EMR.
“Hospitals facing a COVID-19 crunch need to make it easier for teams to focus on the medicine and the human connection” and to shorten the learning curve for those shifting into unfamiliar duties, she advises.
Other lessons learned
Placing COVID-19 patients on the same unit, and keeping non–COVID-19 patients in another area of the hospital, isn’t just a good idea for protecting uninfected patients, Dr. Vaughn notes. It’s also good for providers who are getting used to treating COVID-19 because they don’t have to shift between the needs of different types of patients as they go from room to room.
“The learning curve is steep, but after a couple of days taking care of these patients, you have a good feeling about how to care for them and a great sense of camaraderie with the rest of the team involved in caring for them,” she says. “Everyone jumps in to help because they know we’re in this as a team and that it’s OK for respiratory therapists to step up to help a physician who doesn’t know as much about ventilator care or for nurses to suggest medications based on what other physicians have used.”
The flattening of professional hierarchies long ingrained in hospitals may be a side effect of the tremendous and urgent sense of mission that has developed around responding to COVID-19, Dr. Vaughn notes.
Those stepping into new roles should invite their colleagues to alert them when they see them about to slip up on protective practices that might be new to them. Similarly, they should help each other resist the urge to rush into a COVID-19 patient’s room unprotected in order to help with an urgent situation. The safety of providers – to preserve their ability to care for the many more patients who will need them – must be paramount.
“To handle this pandemic, we need to all be all-in and working toward a common goal, without competing priorities,” she says. “We need to use everyone’s skill sets to the fullest, without creating burnout. We’re going to be different when all this is done.”
Avoiding provider burnout is harder than ever because team members caring for COVID-19 must stay apart from family at home and avoid in-person visits with loved ones and friends. Those who are switching to inpatient or ICU-level care should make a point of focusing on exercise, sleep, virtual connections with loved ones, and healthy eating in between shifts.
“You’re no good to anyone else if you’re not healthy,” Dr. Vaughn says. “Your mental and physical health have to come first because they enable you to help others.”
Paying attention to the appreciation that the community is showing health care workers can also brighten the day of a stressed COVID-19 inpatient clinician, she notes.
“All the little signs of love from the community – the thank you signs, sidewalk chalk drawings, hearts in people’s windows – really do help.”
This article is published courtesy of the University of Michigan Health Lab, where it appeared originally.
When March began, Valerie Vaughn, MD, split her time between caring for general inpatients at the University of Michigan’s hospitals in Ann Arbor and doing research on how to reduce overuse of antibiotics in hospitals nationwide.
By the time the month was over, she had helped create a new kind of hospital team focused on caring for patients with COVID-19, learned how to provide an intensive level of care for the sickest among them, trained hundreds of physicians in how to do the same, and created free online learning tools for physicians nationwide.
Call it switching gears while driving a race car. Changing horses in the middle of a raging river. Or going to medical boot camp. Whatever the metaphor, Dr. Vaughn and her colleagues did it.
And now they’re hoping that sharing what they learned will help others if their hospitals go through the same thing.
Near the epicenter
Michigan Medicine, the University of Michigan’s academic medical center, is a few dozen miles west of the Detroit hospitals that have become a national epicenter for COVID-19 cases. It’s gotten plenty of direct and transferred COVID-19 patients since mid-March.
When Dr. Vaughn’s boss, division of hospital medicine chief Vineet Chopra, MD, was tapped to lead the creation of an all-COVID unit, he asked Dr. Vaughn to work with him and the team of hospitalists, nurse practitioners, physician assistants, nurses, respiratory therapists, and other staff that had volunteered for the team.
They had 3 days to prepare.
The “SWAT team”, as Dr. Vaughn calls it, opened the RICU, or Regional Infectious Containment Unit, on March 16. They doubled the number of beds 2 weeks later.
By the end of March, the team had handed over the reins to a team of experienced intensive care professionals so the unit could focus on the sickest patients. And the RICU team had moved on to transforming other areas of the hospital, and training their staff, in the same way.
By early April, more than 200 beds across the University of Michigan’s hospitals were devoted to COVID-19 care. General medicine physicians who hadn’t practiced inside a hospital since their residency days – thanks to the ability to hand off to hospitalists – were being pulled into inpatient duty. Hospitalists were being pulled into caring for patients who would normally have been in the care of an intensive care team.
“What’s amazed me most is how much people have stepped up to the challenge,” says Dr. Vaughn. “As hard and uncomfortable as it is to do something you’re not typically doing, it can also be therapeutic to say how can I help, let me do something. Yes, they’re anxious, but they want to know how they can be as prepared as they can be, to be as helpful as possible to these patients.”
Dr. Chopra agrees. “The silver lining in all of this is that I have personally seen the best in us come to the surface. Nurses, physicians, pharmacists, and therapists have come together and have shown selflessness, kindness, empathy and resilience in profound ways.”
Making the leap
Even though they didn’t choose hospital medicine, or ICU medicine, as their specialty, physicians may greatly underestimate how useful they can be with a little just-in-time training and the help of residents, fellows, advanced practice providers, and experienced nurses and respiratory therapists.
That training is now available for free through Michigan Medicine’s new online COVID-19 CME portal. The session in “Inpatient Management of COVID-19 patients” provides an important overview for those who have never cared for a case, especially if they haven’t been on inpatient duty in a while. The ICU Bootcamp is for those who will be caring for sicker COVID-19 patients but haven’t practiced in an ICU for a while.
One of the most important roles of a COVID-19 inpatient physician, Dr. Vaughn notes, doesn’t involve new skills. Rather, it draws on the doctoring skills that general medicine and hospital medicine physicians have already honed: the ability to assess and treat the entire patient, to talk with families who can’t be with their loved ones, to humanize the experience for patients and their loved ones as much as possible, and to bring messages of love from the family back to the bedside.
By pairing a general medicine physician newly placed on inpatient duty with a resident, nurse practitioner, or physician assistant who can handle inpatient charting duties, the team can make the most of each kind of provider’s time. Administrators, too, can reduce the burden on the entire team by simplifying processes for what must be charted and recorded in the EMR.
“Hospitals facing a COVID-19 crunch need to make it easier for teams to focus on the medicine and the human connection” and to shorten the learning curve for those shifting into unfamiliar duties, she advises.
Other lessons learned
Placing COVID-19 patients on the same unit, and keeping non–COVID-19 patients in another area of the hospital, isn’t just a good idea for protecting uninfected patients, Dr. Vaughn notes. It’s also good for providers who are getting used to treating COVID-19 because they don’t have to shift between the needs of different types of patients as they go from room to room.
“The learning curve is steep, but after a couple of days taking care of these patients, you have a good feeling about how to care for them and a great sense of camaraderie with the rest of the team involved in caring for them,” she says. “Everyone jumps in to help because they know we’re in this as a team and that it’s OK for respiratory therapists to step up to help a physician who doesn’t know as much about ventilator care or for nurses to suggest medications based on what other physicians have used.”
The flattening of professional hierarchies long ingrained in hospitals may be a side effect of the tremendous and urgent sense of mission that has developed around responding to COVID-19, Dr. Vaughn notes.
Those stepping into new roles should invite their colleagues to alert them when they see them about to slip up on protective practices that might be new to them. Similarly, they should help each other resist the urge to rush into a COVID-19 patient’s room unprotected in order to help with an urgent situation. The safety of providers – to preserve their ability to care for the many more patients who will need them – must be paramount.
“To handle this pandemic, we need to all be all-in and working toward a common goal, without competing priorities,” she says. “We need to use everyone’s skill sets to the fullest, without creating burnout. We’re going to be different when all this is done.”
Avoiding provider burnout is harder than ever because team members caring for COVID-19 must stay apart from family at home and avoid in-person visits with loved ones and friends. Those who are switching to inpatient or ICU-level care should make a point of focusing on exercise, sleep, virtual connections with loved ones, and healthy eating in between shifts.
“You’re no good to anyone else if you’re not healthy,” Dr. Vaughn says. “Your mental and physical health have to come first because they enable you to help others.”
Paying attention to the appreciation that the community is showing health care workers can also brighten the day of a stressed COVID-19 inpatient clinician, she notes.
“All the little signs of love from the community – the thank you signs, sidewalk chalk drawings, hearts in people’s windows – really do help.”
This article is published courtesy of the University of Michigan Health Lab, where it appeared originally.
2019-2020 flu season ends with ‘very high’ activity in New Jersey
The 2019-2020 flu season is ending, but not without a revised map to reflect the COVID-induced new world order.
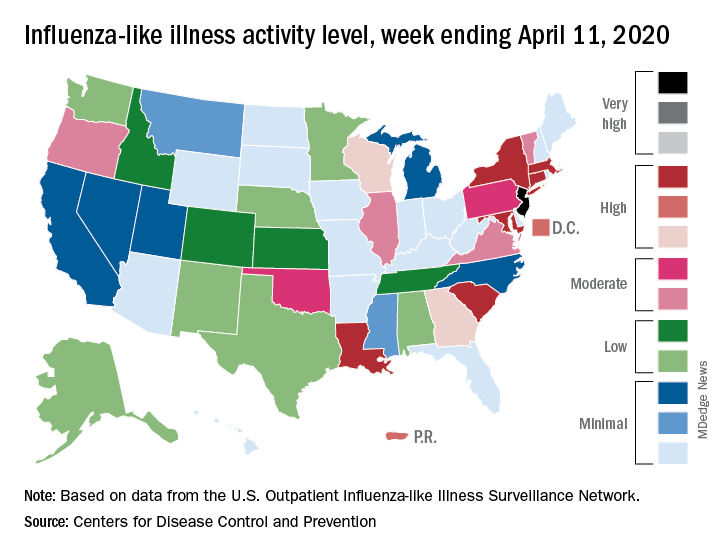
For the week ending April 11, those additions encompass only New Jersey at level 13 and New York City at level 12, the CDC reported April 17.
Eight states, plus the District of Columbia and Puerto Rico, were in the “high” range of flu activity, which runs from level 8 to level 10, for the same week. Those eight states included Connecticut, Georgia, Louisiana, Maryland, Massachusetts, New York, South Carolina, and Wisconsin.
The CDC’s influenza division included this note with its latest FluView report: “The COVID-19 pandemic is affecting healthcare seeking behavior. The number of persons and their reasons for seeking care in the outpatient and ED settings is changing. These changes impact data from ILINet [Outpatient Influenza-like Illness Surveillance Network] in ways that are difficult to differentiate from changes in illness levels, therefore ILINet data should be interpreted with caution.”
Outpatient visits for influenza-like illness made up 2.9% of all visits to health care providers for the week ending April 11, which is the 23rd consecutive week that it’s been at or above the national baseline level of 2.4%. Twenty-three weeks is longer than this has occurred during any flu season since the CDC started setting a baseline in 2007, according to ILINet data.
Mortality from pneumonia and influenza, at 11.7%, was well above the epidemic threshold of 7.0%, although, again, pneumonia mortality “is being driven primarily by an increase in non-influenza pneumonia deaths due to COVID-19,” the CDC wrote.
The total number of influenza-related deaths in children, with reports of two more added this week, is 168 for the season – higher than two of the last three seasons: 144 in 2018-2019, 188 in 2017-2018, and 110 in 2016-2017, according to the CDC.
The 2019-2020 flu season is ending, but not without a revised map to reflect the COVID-induced new world order.

For the week ending April 11, those additions encompass only New Jersey at level 13 and New York City at level 12, the CDC reported April 17.
Eight states, plus the District of Columbia and Puerto Rico, were in the “high” range of flu activity, which runs from level 8 to level 10, for the same week. Those eight states included Connecticut, Georgia, Louisiana, Maryland, Massachusetts, New York, South Carolina, and Wisconsin.
The CDC’s influenza division included this note with its latest FluView report: “The COVID-19 pandemic is affecting healthcare seeking behavior. The number of persons and their reasons for seeking care in the outpatient and ED settings is changing. These changes impact data from ILINet [Outpatient Influenza-like Illness Surveillance Network] in ways that are difficult to differentiate from changes in illness levels, therefore ILINet data should be interpreted with caution.”
Outpatient visits for influenza-like illness made up 2.9% of all visits to health care providers for the week ending April 11, which is the 23rd consecutive week that it’s been at or above the national baseline level of 2.4%. Twenty-three weeks is longer than this has occurred during any flu season since the CDC started setting a baseline in 2007, according to ILINet data.
Mortality from pneumonia and influenza, at 11.7%, was well above the epidemic threshold of 7.0%, although, again, pneumonia mortality “is being driven primarily by an increase in non-influenza pneumonia deaths due to COVID-19,” the CDC wrote.
The total number of influenza-related deaths in children, with reports of two more added this week, is 168 for the season – higher than two of the last three seasons: 144 in 2018-2019, 188 in 2017-2018, and 110 in 2016-2017, according to the CDC.
The 2019-2020 flu season is ending, but not without a revised map to reflect the COVID-induced new world order.

For the week ending April 11, those additions encompass only New Jersey at level 13 and New York City at level 12, the CDC reported April 17.
Eight states, plus the District of Columbia and Puerto Rico, were in the “high” range of flu activity, which runs from level 8 to level 10, for the same week. Those eight states included Connecticut, Georgia, Louisiana, Maryland, Massachusetts, New York, South Carolina, and Wisconsin.
The CDC’s influenza division included this note with its latest FluView report: “The COVID-19 pandemic is affecting healthcare seeking behavior. The number of persons and their reasons for seeking care in the outpatient and ED settings is changing. These changes impact data from ILINet [Outpatient Influenza-like Illness Surveillance Network] in ways that are difficult to differentiate from changes in illness levels, therefore ILINet data should be interpreted with caution.”
Outpatient visits for influenza-like illness made up 2.9% of all visits to health care providers for the week ending April 11, which is the 23rd consecutive week that it’s been at or above the national baseline level of 2.4%. Twenty-three weeks is longer than this has occurred during any flu season since the CDC started setting a baseline in 2007, according to ILINet data.
Mortality from pneumonia and influenza, at 11.7%, was well above the epidemic threshold of 7.0%, although, again, pneumonia mortality “is being driven primarily by an increase in non-influenza pneumonia deaths due to COVID-19,” the CDC wrote.
The total number of influenza-related deaths in children, with reports of two more added this week, is 168 for the season – higher than two of the last three seasons: 144 in 2018-2019, 188 in 2017-2018, and 110 in 2016-2017, according to the CDC.
Infectious disease experts say testing is key to reopening
The key to opening up the American economy rests on the ability to conduct mass testing, according to the Infectious Diseases Society of America (IDSA).
As policymakers weigh how to safely reopen parts of the United States, the IDSA, along with its HIV Medicine Association, issued a set of recommendations outlining the steps that would be necessary in order to begin easing physical distancing measures.
“A stepwise approach to reopening should reflect early diagnosis and enhanced surveillance for COVID-19 cases, linkage of cases to appropriate levels of care, isolation and/or quarantine, contact tracing, and data processing capabilities for state and local public health departments,” according to the recommendation document.
Some of the recommended steps include the following:
- Widespread testing and surveillance, including use of validated nucleic acid amplification assays and anti–SARS-CoV-2 antibody detection.
- The ability to diagnose, treat, and isolate individuals with COVID-19.
- Scaling up of health care capacity and supplies to manage recurrent episodic outbreaks.
- Maintaining a degree of physical distancing to prevent recurrent outbreaks, including use of masks, limiting gatherings, and continued distancing for susceptible adults.
“The recommendations stress that physical distancing policy changes must be based on relevant data and adequate public health resources and capacities and calls for a rolling and incremental approach to lifting these restrictions, ” Thomas File Jr., MD, IDSA president and a professor at Northeastern Ohio Universities, Rootstown, said during an April 17 press briefing.
The rolling approach “must reflect state and regional capacities for diagnosing, isolating, and treating people with the virus, tracing their contacts, protecting health care workers, and addressing the needs of populations disproportionately affected by COVID-19,” he continued.
In order to fully lift physical distancing restrictions, there would need to be effective treatments for COVID-19 and a protective vaccine that can be deployed to key at-risk populations, according to the recommendations.
During the call, Tina Q. Tan, MD, professor of pediatrics at Northwestern University, Chicago, and a member of the IDSA board of directors, said that easing social distancing requirements requires comprehensive data and that “one of the major missing data points” is the number of people who are currently infected or have been infected. She warned that easing restrictions too soon could have “disastrous consequences,” including an increase in spread of infection, hospitalization, and death rates, as well as overwhelming health care facilities.
“In order to reopen, we have to have the ability to safely, successfully, and rapidly diagnose and treat, as well as isolate, individuals with COVID-19, as well as track their contacts,” she said.
The implementation of more widespread, comprehensive testing would better enable targeting of resources, such as personal protective equipment, ICU beds, and ventilators, Dr. Tan said. “This is needed in order to ensure that, if there is an outbreak and it does occur again, the health care system and the first responders are ready for this,” she said.
The key to opening up the American economy rests on the ability to conduct mass testing, according to the Infectious Diseases Society of America (IDSA).
As policymakers weigh how to safely reopen parts of the United States, the IDSA, along with its HIV Medicine Association, issued a set of recommendations outlining the steps that would be necessary in order to begin easing physical distancing measures.
“A stepwise approach to reopening should reflect early diagnosis and enhanced surveillance for COVID-19 cases, linkage of cases to appropriate levels of care, isolation and/or quarantine, contact tracing, and data processing capabilities for state and local public health departments,” according to the recommendation document.
Some of the recommended steps include the following:
- Widespread testing and surveillance, including use of validated nucleic acid amplification assays and anti–SARS-CoV-2 antibody detection.
- The ability to diagnose, treat, and isolate individuals with COVID-19.
- Scaling up of health care capacity and supplies to manage recurrent episodic outbreaks.
- Maintaining a degree of physical distancing to prevent recurrent outbreaks, including use of masks, limiting gatherings, and continued distancing for susceptible adults.
“The recommendations stress that physical distancing policy changes must be based on relevant data and adequate public health resources and capacities and calls for a rolling and incremental approach to lifting these restrictions, ” Thomas File Jr., MD, IDSA president and a professor at Northeastern Ohio Universities, Rootstown, said during an April 17 press briefing.
The rolling approach “must reflect state and regional capacities for diagnosing, isolating, and treating people with the virus, tracing their contacts, protecting health care workers, and addressing the needs of populations disproportionately affected by COVID-19,” he continued.
In order to fully lift physical distancing restrictions, there would need to be effective treatments for COVID-19 and a protective vaccine that can be deployed to key at-risk populations, according to the recommendations.
During the call, Tina Q. Tan, MD, professor of pediatrics at Northwestern University, Chicago, and a member of the IDSA board of directors, said that easing social distancing requirements requires comprehensive data and that “one of the major missing data points” is the number of people who are currently infected or have been infected. She warned that easing restrictions too soon could have “disastrous consequences,” including an increase in spread of infection, hospitalization, and death rates, as well as overwhelming health care facilities.
“In order to reopen, we have to have the ability to safely, successfully, and rapidly diagnose and treat, as well as isolate, individuals with COVID-19, as well as track their contacts,” she said.
The implementation of more widespread, comprehensive testing would better enable targeting of resources, such as personal protective equipment, ICU beds, and ventilators, Dr. Tan said. “This is needed in order to ensure that, if there is an outbreak and it does occur again, the health care system and the first responders are ready for this,” she said.
The key to opening up the American economy rests on the ability to conduct mass testing, according to the Infectious Diseases Society of America (IDSA).
As policymakers weigh how to safely reopen parts of the United States, the IDSA, along with its HIV Medicine Association, issued a set of recommendations outlining the steps that would be necessary in order to begin easing physical distancing measures.
“A stepwise approach to reopening should reflect early diagnosis and enhanced surveillance for COVID-19 cases, linkage of cases to appropriate levels of care, isolation and/or quarantine, contact tracing, and data processing capabilities for state and local public health departments,” according to the recommendation document.
Some of the recommended steps include the following:
- Widespread testing and surveillance, including use of validated nucleic acid amplification assays and anti–SARS-CoV-2 antibody detection.
- The ability to diagnose, treat, and isolate individuals with COVID-19.
- Scaling up of health care capacity and supplies to manage recurrent episodic outbreaks.
- Maintaining a degree of physical distancing to prevent recurrent outbreaks, including use of masks, limiting gatherings, and continued distancing for susceptible adults.
“The recommendations stress that physical distancing policy changes must be based on relevant data and adequate public health resources and capacities and calls for a rolling and incremental approach to lifting these restrictions, ” Thomas File Jr., MD, IDSA president and a professor at Northeastern Ohio Universities, Rootstown, said during an April 17 press briefing.
The rolling approach “must reflect state and regional capacities for diagnosing, isolating, and treating people with the virus, tracing their contacts, protecting health care workers, and addressing the needs of populations disproportionately affected by COVID-19,” he continued.
In order to fully lift physical distancing restrictions, there would need to be effective treatments for COVID-19 and a protective vaccine that can be deployed to key at-risk populations, according to the recommendations.
During the call, Tina Q. Tan, MD, professor of pediatrics at Northwestern University, Chicago, and a member of the IDSA board of directors, said that easing social distancing requirements requires comprehensive data and that “one of the major missing data points” is the number of people who are currently infected or have been infected. She warned that easing restrictions too soon could have “disastrous consequences,” including an increase in spread of infection, hospitalization, and death rates, as well as overwhelming health care facilities.
“In order to reopen, we have to have the ability to safely, successfully, and rapidly diagnose and treat, as well as isolate, individuals with COVID-19, as well as track their contacts,” she said.
The implementation of more widespread, comprehensive testing would better enable targeting of resources, such as personal protective equipment, ICU beds, and ventilators, Dr. Tan said. “This is needed in order to ensure that, if there is an outbreak and it does occur again, the health care system and the first responders are ready for this,” she said.
How to sanitize N95 masks for reuse: NIH study
Exposing contaminated N95 respirators to vaporized hydrogen peroxide (VHP) or ultraviolet (UV) light appears to eliminate the SARS-CoV-2 virus from the material and preserve the integrity of the masks fit for up to three uses, a National Institutes of Health (NIH) study shows.
Dry heat (70° C) was also found to eliminate the virus on masks but was effective for two uses instead of three.
Robert Fischer, PhD, with the National Institute of Allergy and Infectious Diseases in Hamilton, Montana, and colleagues posted the findings on a preprint server on April 15. The paper has not yet been peer reviewed.
Four methods tested
Fischer and colleagues compared four methods for decontaminating the masks, which are designed for one-time use: UV radiation (260-285 nm); 70° C dry heat; 70% ethanol spray; and VHP.
For each method, the researchers compared the rate at which SARS-CoV-2 is inactivated on N95 filter fabric to that on stainless steel.
All four methods eliminated detectable SARS-CoV-2 virus from the fabric test samples, though the time needed for decontamination varied. VHP was the quickest, requiring 10 minutes. Dry heat and UV light each required approximately 60 minutes. Ethanol required an intermediate amount of time.
To test durability over three uses, the researchers treated intact, clean masks with the same decontamination method and assessed function via quantitative fit testing.
Volunteers from the Rocky Mountain laboratory wore the masks for 2 hours to test fit and seal.
The researchers found that masks that had been decontaminated with ethanol spray did not function effectively after decontamination, and they did not recommend use of that method.
By contrast, masks decontaminated with UV and VHP could be used up to three times and function properly. Masks decontaminated with dry heat could be used two times before function declined.
“Our results indicate that N95 respirators can be decontaminated and reused in times of shortage for up to three times for UV and HPV, and up to two times for dry heat,” the authors write. “However, utmost care should be given to ensure the proper functioning of the N95 respirator after each decontamination using readily available qualitative fit testing tools and to ensure that treatments are carried out for sufficient time to achieve desired risk-reduction.”
Reassurance for clinicians
The results will reassure clinicians, many of whom are already using these decontamination methods, Ravina Kullar, PharmD, MPH, an infectious disease expert with the Infectious Diseases Society of America, told Medscape Medical News.
Kullar, who is also an adjunct faculty member at the David Geffen School of Medicine of the University of California, Los Angeles, said the most widely used methods have been UV light and VPH.
UV light has been used for years to decontaminate rooms, she said. She also said that so far, supplies of hydrogen peroxide are adequate.
A shortcoming of the study, Kullar said, is that it tested the masks for only 2 hours, whereas in clinical practice, they are being worn for much longer periods.
After the study is peer reviewed, the Centers for Disease Control and Prevention (CDC) may update its recommendations, she said.
So far, she noted, the CDC has not approved any method for decontaminating masks, “but it has said that it does not object to using these sterilizers, disinfectants, devices, and air purifiers for effectively killing this virus.”
Safe, multiple use of the masks is critical in the COVID-19 crisis, she said.
“We have to look at other mechanisms to keep these N95 respirators in use when there’s such a shortage,” she said.
Integrity of the fit was an important factor in the study.
“All health care workers have to go through a fitting to have that mask fitted appropriately. That’s why these N95s are only approved for health care professionals, not the lay public,” she said.
The study was supported by the National Institutes of Health; the Defense Advanced Research Projects Agency; the University of California, Los Angeles; the US National Science Foundation; and the US Department of Defense.
This article first appeared on Medscape.com.
Exposing contaminated N95 respirators to vaporized hydrogen peroxide (VHP) or ultraviolet (UV) light appears to eliminate the SARS-CoV-2 virus from the material and preserve the integrity of the masks fit for up to three uses, a National Institutes of Health (NIH) study shows.
Dry heat (70° C) was also found to eliminate the virus on masks but was effective for two uses instead of three.
Robert Fischer, PhD, with the National Institute of Allergy and Infectious Diseases in Hamilton, Montana, and colleagues posted the findings on a preprint server on April 15. The paper has not yet been peer reviewed.
Four methods tested
Fischer and colleagues compared four methods for decontaminating the masks, which are designed for one-time use: UV radiation (260-285 nm); 70° C dry heat; 70% ethanol spray; and VHP.
For each method, the researchers compared the rate at which SARS-CoV-2 is inactivated on N95 filter fabric to that on stainless steel.
All four methods eliminated detectable SARS-CoV-2 virus from the fabric test samples, though the time needed for decontamination varied. VHP was the quickest, requiring 10 minutes. Dry heat and UV light each required approximately 60 minutes. Ethanol required an intermediate amount of time.
To test durability over three uses, the researchers treated intact, clean masks with the same decontamination method and assessed function via quantitative fit testing.
Volunteers from the Rocky Mountain laboratory wore the masks for 2 hours to test fit and seal.
The researchers found that masks that had been decontaminated with ethanol spray did not function effectively after decontamination, and they did not recommend use of that method.
By contrast, masks decontaminated with UV and VHP could be used up to three times and function properly. Masks decontaminated with dry heat could be used two times before function declined.
“Our results indicate that N95 respirators can be decontaminated and reused in times of shortage for up to three times for UV and HPV, and up to two times for dry heat,” the authors write. “However, utmost care should be given to ensure the proper functioning of the N95 respirator after each decontamination using readily available qualitative fit testing tools and to ensure that treatments are carried out for sufficient time to achieve desired risk-reduction.”
Reassurance for clinicians
The results will reassure clinicians, many of whom are already using these decontamination methods, Ravina Kullar, PharmD, MPH, an infectious disease expert with the Infectious Diseases Society of America, told Medscape Medical News.
Kullar, who is also an adjunct faculty member at the David Geffen School of Medicine of the University of California, Los Angeles, said the most widely used methods have been UV light and VPH.
UV light has been used for years to decontaminate rooms, she said. She also said that so far, supplies of hydrogen peroxide are adequate.
A shortcoming of the study, Kullar said, is that it tested the masks for only 2 hours, whereas in clinical practice, they are being worn for much longer periods.
After the study is peer reviewed, the Centers for Disease Control and Prevention (CDC) may update its recommendations, she said.
So far, she noted, the CDC has not approved any method for decontaminating masks, “but it has said that it does not object to using these sterilizers, disinfectants, devices, and air purifiers for effectively killing this virus.”
Safe, multiple use of the masks is critical in the COVID-19 crisis, she said.
“We have to look at other mechanisms to keep these N95 respirators in use when there’s such a shortage,” she said.
Integrity of the fit was an important factor in the study.
“All health care workers have to go through a fitting to have that mask fitted appropriately. That’s why these N95s are only approved for health care professionals, not the lay public,” she said.
The study was supported by the National Institutes of Health; the Defense Advanced Research Projects Agency; the University of California, Los Angeles; the US National Science Foundation; and the US Department of Defense.
This article first appeared on Medscape.com.
Exposing contaminated N95 respirators to vaporized hydrogen peroxide (VHP) or ultraviolet (UV) light appears to eliminate the SARS-CoV-2 virus from the material and preserve the integrity of the masks fit for up to three uses, a National Institutes of Health (NIH) study shows.
Dry heat (70° C) was also found to eliminate the virus on masks but was effective for two uses instead of three.
Robert Fischer, PhD, with the National Institute of Allergy and Infectious Diseases in Hamilton, Montana, and colleagues posted the findings on a preprint server on April 15. The paper has not yet been peer reviewed.
Four methods tested
Fischer and colleagues compared four methods for decontaminating the masks, which are designed for one-time use: UV radiation (260-285 nm); 70° C dry heat; 70% ethanol spray; and VHP.
For each method, the researchers compared the rate at which SARS-CoV-2 is inactivated on N95 filter fabric to that on stainless steel.
All four methods eliminated detectable SARS-CoV-2 virus from the fabric test samples, though the time needed for decontamination varied. VHP was the quickest, requiring 10 minutes. Dry heat and UV light each required approximately 60 minutes. Ethanol required an intermediate amount of time.
To test durability over three uses, the researchers treated intact, clean masks with the same decontamination method and assessed function via quantitative fit testing.
Volunteers from the Rocky Mountain laboratory wore the masks for 2 hours to test fit and seal.
The researchers found that masks that had been decontaminated with ethanol spray did not function effectively after decontamination, and they did not recommend use of that method.
By contrast, masks decontaminated with UV and VHP could be used up to three times and function properly. Masks decontaminated with dry heat could be used two times before function declined.
“Our results indicate that N95 respirators can be decontaminated and reused in times of shortage for up to three times for UV and HPV, and up to two times for dry heat,” the authors write. “However, utmost care should be given to ensure the proper functioning of the N95 respirator after each decontamination using readily available qualitative fit testing tools and to ensure that treatments are carried out for sufficient time to achieve desired risk-reduction.”
Reassurance for clinicians
The results will reassure clinicians, many of whom are already using these decontamination methods, Ravina Kullar, PharmD, MPH, an infectious disease expert with the Infectious Diseases Society of America, told Medscape Medical News.
Kullar, who is also an adjunct faculty member at the David Geffen School of Medicine of the University of California, Los Angeles, said the most widely used methods have been UV light and VPH.
UV light has been used for years to decontaminate rooms, she said. She also said that so far, supplies of hydrogen peroxide are adequate.
A shortcoming of the study, Kullar said, is that it tested the masks for only 2 hours, whereas in clinical practice, they are being worn for much longer periods.
After the study is peer reviewed, the Centers for Disease Control and Prevention (CDC) may update its recommendations, she said.
So far, she noted, the CDC has not approved any method for decontaminating masks, “but it has said that it does not object to using these sterilizers, disinfectants, devices, and air purifiers for effectively killing this virus.”
Safe, multiple use of the masks is critical in the COVID-19 crisis, she said.
“We have to look at other mechanisms to keep these N95 respirators in use when there’s such a shortage,” she said.
Integrity of the fit was an important factor in the study.
“All health care workers have to go through a fitting to have that mask fitted appropriately. That’s why these N95s are only approved for health care professionals, not the lay public,” she said.
The study was supported by the National Institutes of Health; the Defense Advanced Research Projects Agency; the University of California, Los Angeles; the US National Science Foundation; and the US Department of Defense.
This article first appeared on Medscape.com.
Cautionary tale spurs ‘world’s first’ COVID-19 psychiatric ward
There was no hand sanitizer on the hospital’s psychiatric ward for fear patients would drink it; they slept together on futons in communal rooms and the windows were sealed shut to prevent suicide attempts — all conditions that created the perfect environment for the rapid spread of a potentially deadly virus.
This scenario may sound like a something out of a horror film, but as reported last month by the UK newspaper The Independent, it was the reality in the psychiatric ward of South Korea’s Daenam Hospital after COVID-19 struck. Eventually health officials put the ward on lockdown, but it wasn’t long before all but two of the unit’s 103 patients were positive for the virus.
To avoid a similar catastrophe, staff at an Israeli hospital have created what they describe as the “world’s first” dedicated COVID-19 unit for psychiatric inpatients.
Clinicians at Israel’s national hospital, Sheba Medical Center Tel HaShomer in Tel Aviv, believe the 16-bed unit, which officially opened on March 26, will stop psychiatric inpatients with the virus — who may have trouble with social distancing — from spreading it to others on the ward.
“Psychiatric patients are going to get sick from coronavirus just like anybody else,” Mark Weiser, MD, head of the psychiatric division at the institution told Medscape Medical News. “But we’re concerned that, on a psychiatric ward, a patient who is COVID-19 positive can also be psychotic, manic, cognitively impaired, or have poor judgment … making it difficult for that patient to keep social distancing, and very quickly you’ll have an entire ward of patients infected.
“So the basic public health issue is how to prevent a single psychiatric patient who is hospitalized and COVID-19-positive from making everybody else sick,” he added.
Unique Challenges, Rapid Response
Adapting an existing psychiatric ward to one exclusively used by inpatients with COVID-19 required significant planning, coordination, and modifications to ensure the well-being of patients and staff.
A dedicated entrance for the exclusive use of infected psychiatric inpatients was also created.
In addition, two-way television cameras in patients’ rooms were installed to facilitate a constant flow of communication and enable therapeutic sessions and family visits. All of these modifications were completed in under a week.
“Under normal circumstances, we have cameras in the public areas of our wards, but in order to respect people’s privacy, we do not have cameras in their rooms.
“In this specific ward, on the other hand, we did put cameras in the rooms, so if a patient needs to be watched more closely, it could be done remotely without exposing staff to the virus. We have a person who’s watching the screens at all times, just to see what’s going on and see what patients are doing,” said Weiser.
Protective personal equipment (PPE) and clothing for staff was tailored to the unique challenges posed by the ward’s patient population.
“Of course, you need to wear clothes that are protective against the virus,” said Weiser. “But sometimes our patients can get agitated or even violent, so you’ve got protect against that as well.”
With this in mind, all personnel working on the ward must put on an extra layer of PPE as well as a tear-proof robe. The institution has also implemented a strict protocol that dictates the order in which PPE is donned and doffed.
“It’s got to be done in a very careful and very specific way,” said Weiser. “We have all of it organized with a poster that explains what should be taken off or put on, and in what order.”
For institutions considering setting up a similar unit, Weiser said close proximity to an active care hospital with the capacity to provide urgent care is key.
“We’re psychiatrists; we’re not great at treating acute respiratory problems. So patients with significant respiratory problems need a place to get appropriate care quickly,” he said.
In setting up the unit, there were still a few obstacles, Weiser noted. For instance, despite the many protective and safety measures undertaken by the institution, some of the hospital staff were concerned about their risk of contracting the virus.
To address these concerns, the hospital’s leadership brought in infectious disease experts to educate hospital personnel about the virus and transmission risk.
“They told our staff that given all the precautions we had taken, there was very little risk anyone else could become infected,” Weiser said.
Despite the many challenges, Weiser said he and his colleagues are thrilled with the dedicated ward and the positive reception it has received.
“My colleagues and the directors of psychiatric hospitals all around the country are very happy with this because now they’re not hospitalizing infected patients. They’re very happy for us to take care of this,” he said.
“No Easy Solutions”
Commenting on the initiative for Medscape Medical News, John M. Oldham, MD, chief of staff at Baylor College of Medicine’s Menninger Clinic in Houston, Texas, raised some questions.
“Is it really going to be the treatment unit or a quarantine unit? Because if you don’t have a comparable level of established, effective treatment for these patients, then you’re simply herding them off to a different place where they’re going to suffer both illnesses,” he cautioned.
Nevertheless, Oldham recognized that the issue of how to treat psychiatric patients who test positive for COVID-19 is complex.
“We’re still wrestling with that question here at Menninger. We have created an enclosed section of the inpatient area reserved for this possibility.
“If we have a patient who tests positive, we will immediately put that patient in one of these rooms in the quarantine section. Then we will use protective equipment for our staff to go and provide care for the patient,” he said.
However, he acknowledged that a psychiatric hospital is in no position to treat patients who develop severe illness from COVID-19.
“We’re certainly worried about it,” he said, “because how many inpatient general medical units are going to want to take a significantly symptomatic COVID-19 patient who was in the hospital for being acutely suicidal? There are no easy solutions.”
This article first appeared on Medscape.com.
There was no hand sanitizer on the hospital’s psychiatric ward for fear patients would drink it; they slept together on futons in communal rooms and the windows were sealed shut to prevent suicide attempts — all conditions that created the perfect environment for the rapid spread of a potentially deadly virus.
This scenario may sound like a something out of a horror film, but as reported last month by the UK newspaper The Independent, it was the reality in the psychiatric ward of South Korea’s Daenam Hospital after COVID-19 struck. Eventually health officials put the ward on lockdown, but it wasn’t long before all but two of the unit’s 103 patients were positive for the virus.
To avoid a similar catastrophe, staff at an Israeli hospital have created what they describe as the “world’s first” dedicated COVID-19 unit for psychiatric inpatients.
Clinicians at Israel’s national hospital, Sheba Medical Center Tel HaShomer in Tel Aviv, believe the 16-bed unit, which officially opened on March 26, will stop psychiatric inpatients with the virus — who may have trouble with social distancing — from spreading it to others on the ward.
“Psychiatric patients are going to get sick from coronavirus just like anybody else,” Mark Weiser, MD, head of the psychiatric division at the institution told Medscape Medical News. “But we’re concerned that, on a psychiatric ward, a patient who is COVID-19 positive can also be psychotic, manic, cognitively impaired, or have poor judgment … making it difficult for that patient to keep social distancing, and very quickly you’ll have an entire ward of patients infected.
“So the basic public health issue is how to prevent a single psychiatric patient who is hospitalized and COVID-19-positive from making everybody else sick,” he added.
Unique Challenges, Rapid Response
Adapting an existing psychiatric ward to one exclusively used by inpatients with COVID-19 required significant planning, coordination, and modifications to ensure the well-being of patients and staff.
A dedicated entrance for the exclusive use of infected psychiatric inpatients was also created.
In addition, two-way television cameras in patients’ rooms were installed to facilitate a constant flow of communication and enable therapeutic sessions and family visits. All of these modifications were completed in under a week.
“Under normal circumstances, we have cameras in the public areas of our wards, but in order to respect people’s privacy, we do not have cameras in their rooms.
“In this specific ward, on the other hand, we did put cameras in the rooms, so if a patient needs to be watched more closely, it could be done remotely without exposing staff to the virus. We have a person who’s watching the screens at all times, just to see what’s going on and see what patients are doing,” said Weiser.
Protective personal equipment (PPE) and clothing for staff was tailored to the unique challenges posed by the ward’s patient population.
“Of course, you need to wear clothes that are protective against the virus,” said Weiser. “But sometimes our patients can get agitated or even violent, so you’ve got protect against that as well.”
With this in mind, all personnel working on the ward must put on an extra layer of PPE as well as a tear-proof robe. The institution has also implemented a strict protocol that dictates the order in which PPE is donned and doffed.
“It’s got to be done in a very careful and very specific way,” said Weiser. “We have all of it organized with a poster that explains what should be taken off or put on, and in what order.”
For institutions considering setting up a similar unit, Weiser said close proximity to an active care hospital with the capacity to provide urgent care is key.
“We’re psychiatrists; we’re not great at treating acute respiratory problems. So patients with significant respiratory problems need a place to get appropriate care quickly,” he said.
In setting up the unit, there were still a few obstacles, Weiser noted. For instance, despite the many protective and safety measures undertaken by the institution, some of the hospital staff were concerned about their risk of contracting the virus.
To address these concerns, the hospital’s leadership brought in infectious disease experts to educate hospital personnel about the virus and transmission risk.
“They told our staff that given all the precautions we had taken, there was very little risk anyone else could become infected,” Weiser said.
Despite the many challenges, Weiser said he and his colleagues are thrilled with the dedicated ward and the positive reception it has received.
“My colleagues and the directors of psychiatric hospitals all around the country are very happy with this because now they’re not hospitalizing infected patients. They’re very happy for us to take care of this,” he said.
“No Easy Solutions”
Commenting on the initiative for Medscape Medical News, John M. Oldham, MD, chief of staff at Baylor College of Medicine’s Menninger Clinic in Houston, Texas, raised some questions.
“Is it really going to be the treatment unit or a quarantine unit? Because if you don’t have a comparable level of established, effective treatment for these patients, then you’re simply herding them off to a different place where they’re going to suffer both illnesses,” he cautioned.
Nevertheless, Oldham recognized that the issue of how to treat psychiatric patients who test positive for COVID-19 is complex.
“We’re still wrestling with that question here at Menninger. We have created an enclosed section of the inpatient area reserved for this possibility.
“If we have a patient who tests positive, we will immediately put that patient in one of these rooms in the quarantine section. Then we will use protective equipment for our staff to go and provide care for the patient,” he said.
However, he acknowledged that a psychiatric hospital is in no position to treat patients who develop severe illness from COVID-19.
“We’re certainly worried about it,” he said, “because how many inpatient general medical units are going to want to take a significantly symptomatic COVID-19 patient who was in the hospital for being acutely suicidal? There are no easy solutions.”
This article first appeared on Medscape.com.
There was no hand sanitizer on the hospital’s psychiatric ward for fear patients would drink it; they slept together on futons in communal rooms and the windows were sealed shut to prevent suicide attempts — all conditions that created the perfect environment for the rapid spread of a potentially deadly virus.
This scenario may sound like a something out of a horror film, but as reported last month by the UK newspaper The Independent, it was the reality in the psychiatric ward of South Korea’s Daenam Hospital after COVID-19 struck. Eventually health officials put the ward on lockdown, but it wasn’t long before all but two of the unit’s 103 patients were positive for the virus.
To avoid a similar catastrophe, staff at an Israeli hospital have created what they describe as the “world’s first” dedicated COVID-19 unit for psychiatric inpatients.
Clinicians at Israel’s national hospital, Sheba Medical Center Tel HaShomer in Tel Aviv, believe the 16-bed unit, which officially opened on March 26, will stop psychiatric inpatients with the virus — who may have trouble with social distancing — from spreading it to others on the ward.
“Psychiatric patients are going to get sick from coronavirus just like anybody else,” Mark Weiser, MD, head of the psychiatric division at the institution told Medscape Medical News. “But we’re concerned that, on a psychiatric ward, a patient who is COVID-19 positive can also be psychotic, manic, cognitively impaired, or have poor judgment … making it difficult for that patient to keep social distancing, and very quickly you’ll have an entire ward of patients infected.
“So the basic public health issue is how to prevent a single psychiatric patient who is hospitalized and COVID-19-positive from making everybody else sick,” he added.
Unique Challenges, Rapid Response
Adapting an existing psychiatric ward to one exclusively used by inpatients with COVID-19 required significant planning, coordination, and modifications to ensure the well-being of patients and staff.
A dedicated entrance for the exclusive use of infected psychiatric inpatients was also created.
In addition, two-way television cameras in patients’ rooms were installed to facilitate a constant flow of communication and enable therapeutic sessions and family visits. All of these modifications were completed in under a week.
“Under normal circumstances, we have cameras in the public areas of our wards, but in order to respect people’s privacy, we do not have cameras in their rooms.
“In this specific ward, on the other hand, we did put cameras in the rooms, so if a patient needs to be watched more closely, it could be done remotely without exposing staff to the virus. We have a person who’s watching the screens at all times, just to see what’s going on and see what patients are doing,” said Weiser.
Protective personal equipment (PPE) and clothing for staff was tailored to the unique challenges posed by the ward’s patient population.
“Of course, you need to wear clothes that are protective against the virus,” said Weiser. “But sometimes our patients can get agitated or even violent, so you’ve got protect against that as well.”
With this in mind, all personnel working on the ward must put on an extra layer of PPE as well as a tear-proof robe. The institution has also implemented a strict protocol that dictates the order in which PPE is donned and doffed.
“It’s got to be done in a very careful and very specific way,” said Weiser. “We have all of it organized with a poster that explains what should be taken off or put on, and in what order.”
For institutions considering setting up a similar unit, Weiser said close proximity to an active care hospital with the capacity to provide urgent care is key.
“We’re psychiatrists; we’re not great at treating acute respiratory problems. So patients with significant respiratory problems need a place to get appropriate care quickly,” he said.
In setting up the unit, there were still a few obstacles, Weiser noted. For instance, despite the many protective and safety measures undertaken by the institution, some of the hospital staff were concerned about their risk of contracting the virus.
To address these concerns, the hospital’s leadership brought in infectious disease experts to educate hospital personnel about the virus and transmission risk.
“They told our staff that given all the precautions we had taken, there was very little risk anyone else could become infected,” Weiser said.
Despite the many challenges, Weiser said he and his colleagues are thrilled with the dedicated ward and the positive reception it has received.
“My colleagues and the directors of psychiatric hospitals all around the country are very happy with this because now they’re not hospitalizing infected patients. They’re very happy for us to take care of this,” he said.
“No Easy Solutions”
Commenting on the initiative for Medscape Medical News, John M. Oldham, MD, chief of staff at Baylor College of Medicine’s Menninger Clinic in Houston, Texas, raised some questions.
“Is it really going to be the treatment unit or a quarantine unit? Because if you don’t have a comparable level of established, effective treatment for these patients, then you’re simply herding them off to a different place where they’re going to suffer both illnesses,” he cautioned.
Nevertheless, Oldham recognized that the issue of how to treat psychiatric patients who test positive for COVID-19 is complex.
“We’re still wrestling with that question here at Menninger. We have created an enclosed section of the inpatient area reserved for this possibility.
“If we have a patient who tests positive, we will immediately put that patient in one of these rooms in the quarantine section. Then we will use protective equipment for our staff to go and provide care for the patient,” he said.
However, he acknowledged that a psychiatric hospital is in no position to treat patients who develop severe illness from COVID-19.
“We’re certainly worried about it,” he said, “because how many inpatient general medical units are going to want to take a significantly symptomatic COVID-19 patient who was in the hospital for being acutely suicidal? There are no easy solutions.”
This article first appeared on Medscape.com.
COVID-19: How intensive care cardiology can inform the response
Because of their place at the interface between critical care and cardiovascular medicine, critical care cardiologists are in a good position to come up with novel approaches to adapting critical care systems to the current crisis. Health care and clinical resources have been severely strained in some places, and increasing evidence suggests that SARS-CoV-2 can cause injury to most organ systems. More than a quarter of hospitalized patients have cardiac injury, which can be a key reason for clinical deterioration.
An international group of critical care cardiologists led by Jason Katz, MD, of Duke University, Durham, N.C., offered suggestions for scalable models for critical care delivery in the context of COVID-19 in the Journal of the American College of Cardiology.
Critical care cardiology developed in response to changes in patient populations and their clinical needs. Respiratory insufficiency, heart failure, structural heart disease, and multisystem organ dysfunction became more common than patients with complicated acute MI, leading cardiologists in critical care cardiology to become more proficient in general critical care medicine, and to become leaders in forming collaborative multidisciplinary teams. The authors argued that COVID-19 is precipitating a similar need to adapt to the changing needs of patients.
“This pandemic should serve as a clarion call to our health care systems that we should continue to develop a nimble workforce that can adapt to change quickly during a crisis. We believe critical care cardiologists are well positioned to help serve society in this capacity,” the authors wrote.
Surge staging
They proposed four surge stages based in part on an American College of Chest Physicians–endorsed model (Chest 2014 Oct;146:e61S-74S), which regards a 25% capacity surge as minor. At the other end of the spectrum, a 200% surge is defined as a “disaster.” In minor surges (less than 25% increase), the traditional cardiac ICU (CICU) model can continue to be applied. During moderate (25%-100% increases) or major (100%-200%) surges, the critical care cardiologist should collaborate or consult within multiple health care teams. Physicians not trained in critical care can assist with care of intubated and critically ill patients under the supervision of a critical care cardiologist or under the supervision of a partnership between a non–cardiac critical care medicine provider and a cardiologist. The number of patients cared for by each team should increase in step with the size of the surge.
In disaster situations (more than 200% surge), there should be adaptive and dynamic staffing reorganization. The report included an illustration of a range of steps that can be taken, including alterations to staffing, regional care systems, resource management, and triage practices. Scoring systems such as Sequential Organ Failure Assessment may be useful for triaging, but the authors also suggest employment of validated cardiac disease–specific scores, because traditional ICU measures don’t always apply well to CICU populations.
At the hospital level, deferrals should be made for elective cardiac procedures that require CICU or postanesthesia care unit recovery periods. Semielective procedures should be considered after risk-benefit considerations when delays could lead to morbidity or mortality. Even some traditional emergency procedures may need to be reevaluated in the COVID-19 context: For example, some low-risk ST-segment elevation MI (STEMI) patients don’t require ICU care but are manageable in cardiac intermediate care beds instead. Historical triage practices should be reexamined to predict which STEMI patients will require ICU care.
Resource allocation
The CICU work flow will be affected as some of its beds are opened up to COVID-19 patients. Standard philosophies of concentrating intense resources will have to give way to a utilitarian approach that evaluates operations based on efficiency, equity, and justice. Physician-patient contact should be minimized using technological links when possible, and rounds might be reorganized to first examine patients without COVID-19, in order to minimize between-patient spread.
Military medicine, which is used to ramping up operations during times of crisis, has potential lessons for the current pandemic. In the face of mass casualties, military physicians often turn to the North Atlantic Treaty Organization triage system, which separates patients into four categories: immediate, requiring lifesaving intervention; delayed, requiring intervention within hours to days; minimal, where the patient is injured but ambulatory; and expectant patients who are deceased or too injured to save. Impersonal though this system may be, it may be required in the most severe scenarios when resources are scarce or absent.
The authors reported no relevant financial disclosures.
SOURCE: Katz J et al. J Am Coll Cardiol. 2020 Apr 15. doi: 10.1016/j.annonc.2020.02.01.
Because of their place at the interface between critical care and cardiovascular medicine, critical care cardiologists are in a good position to come up with novel approaches to adapting critical care systems to the current crisis. Health care and clinical resources have been severely strained in some places, and increasing evidence suggests that SARS-CoV-2 can cause injury to most organ systems. More than a quarter of hospitalized patients have cardiac injury, which can be a key reason for clinical deterioration.
An international group of critical care cardiologists led by Jason Katz, MD, of Duke University, Durham, N.C., offered suggestions for scalable models for critical care delivery in the context of COVID-19 in the Journal of the American College of Cardiology.
Critical care cardiology developed in response to changes in patient populations and their clinical needs. Respiratory insufficiency, heart failure, structural heart disease, and multisystem organ dysfunction became more common than patients with complicated acute MI, leading cardiologists in critical care cardiology to become more proficient in general critical care medicine, and to become leaders in forming collaborative multidisciplinary teams. The authors argued that COVID-19 is precipitating a similar need to adapt to the changing needs of patients.
“This pandemic should serve as a clarion call to our health care systems that we should continue to develop a nimble workforce that can adapt to change quickly during a crisis. We believe critical care cardiologists are well positioned to help serve society in this capacity,” the authors wrote.
Surge staging
They proposed four surge stages based in part on an American College of Chest Physicians–endorsed model (Chest 2014 Oct;146:e61S-74S), which regards a 25% capacity surge as minor. At the other end of the spectrum, a 200% surge is defined as a “disaster.” In minor surges (less than 25% increase), the traditional cardiac ICU (CICU) model can continue to be applied. During moderate (25%-100% increases) or major (100%-200%) surges, the critical care cardiologist should collaborate or consult within multiple health care teams. Physicians not trained in critical care can assist with care of intubated and critically ill patients under the supervision of a critical care cardiologist or under the supervision of a partnership between a non–cardiac critical care medicine provider and a cardiologist. The number of patients cared for by each team should increase in step with the size of the surge.
In disaster situations (more than 200% surge), there should be adaptive and dynamic staffing reorganization. The report included an illustration of a range of steps that can be taken, including alterations to staffing, regional care systems, resource management, and triage practices. Scoring systems such as Sequential Organ Failure Assessment may be useful for triaging, but the authors also suggest employment of validated cardiac disease–specific scores, because traditional ICU measures don’t always apply well to CICU populations.
At the hospital level, deferrals should be made for elective cardiac procedures that require CICU or postanesthesia care unit recovery periods. Semielective procedures should be considered after risk-benefit considerations when delays could lead to morbidity or mortality. Even some traditional emergency procedures may need to be reevaluated in the COVID-19 context: For example, some low-risk ST-segment elevation MI (STEMI) patients don’t require ICU care but are manageable in cardiac intermediate care beds instead. Historical triage practices should be reexamined to predict which STEMI patients will require ICU care.
Resource allocation
The CICU work flow will be affected as some of its beds are opened up to COVID-19 patients. Standard philosophies of concentrating intense resources will have to give way to a utilitarian approach that evaluates operations based on efficiency, equity, and justice. Physician-patient contact should be minimized using technological links when possible, and rounds might be reorganized to first examine patients without COVID-19, in order to minimize between-patient spread.
Military medicine, which is used to ramping up operations during times of crisis, has potential lessons for the current pandemic. In the face of mass casualties, military physicians often turn to the North Atlantic Treaty Organization triage system, which separates patients into four categories: immediate, requiring lifesaving intervention; delayed, requiring intervention within hours to days; minimal, where the patient is injured but ambulatory; and expectant patients who are deceased or too injured to save. Impersonal though this system may be, it may be required in the most severe scenarios when resources are scarce or absent.
The authors reported no relevant financial disclosures.
SOURCE: Katz J et al. J Am Coll Cardiol. 2020 Apr 15. doi: 10.1016/j.annonc.2020.02.01.
Because of their place at the interface between critical care and cardiovascular medicine, critical care cardiologists are in a good position to come up with novel approaches to adapting critical care systems to the current crisis. Health care and clinical resources have been severely strained in some places, and increasing evidence suggests that SARS-CoV-2 can cause injury to most organ systems. More than a quarter of hospitalized patients have cardiac injury, which can be a key reason for clinical deterioration.
An international group of critical care cardiologists led by Jason Katz, MD, of Duke University, Durham, N.C., offered suggestions for scalable models for critical care delivery in the context of COVID-19 in the Journal of the American College of Cardiology.
Critical care cardiology developed in response to changes in patient populations and their clinical needs. Respiratory insufficiency, heart failure, structural heart disease, and multisystem organ dysfunction became more common than patients with complicated acute MI, leading cardiologists in critical care cardiology to become more proficient in general critical care medicine, and to become leaders in forming collaborative multidisciplinary teams. The authors argued that COVID-19 is precipitating a similar need to adapt to the changing needs of patients.
“This pandemic should serve as a clarion call to our health care systems that we should continue to develop a nimble workforce that can adapt to change quickly during a crisis. We believe critical care cardiologists are well positioned to help serve society in this capacity,” the authors wrote.
Surge staging
They proposed four surge stages based in part on an American College of Chest Physicians–endorsed model (Chest 2014 Oct;146:e61S-74S), which regards a 25% capacity surge as minor. At the other end of the spectrum, a 200% surge is defined as a “disaster.” In minor surges (less than 25% increase), the traditional cardiac ICU (CICU) model can continue to be applied. During moderate (25%-100% increases) or major (100%-200%) surges, the critical care cardiologist should collaborate or consult within multiple health care teams. Physicians not trained in critical care can assist with care of intubated and critically ill patients under the supervision of a critical care cardiologist or under the supervision of a partnership between a non–cardiac critical care medicine provider and a cardiologist. The number of patients cared for by each team should increase in step with the size of the surge.
In disaster situations (more than 200% surge), there should be adaptive and dynamic staffing reorganization. The report included an illustration of a range of steps that can be taken, including alterations to staffing, regional care systems, resource management, and triage practices. Scoring systems such as Sequential Organ Failure Assessment may be useful for triaging, but the authors also suggest employment of validated cardiac disease–specific scores, because traditional ICU measures don’t always apply well to CICU populations.
At the hospital level, deferrals should be made for elective cardiac procedures that require CICU or postanesthesia care unit recovery periods. Semielective procedures should be considered after risk-benefit considerations when delays could lead to morbidity or mortality. Even some traditional emergency procedures may need to be reevaluated in the COVID-19 context: For example, some low-risk ST-segment elevation MI (STEMI) patients don’t require ICU care but are manageable in cardiac intermediate care beds instead. Historical triage practices should be reexamined to predict which STEMI patients will require ICU care.
Resource allocation
The CICU work flow will be affected as some of its beds are opened up to COVID-19 patients. Standard philosophies of concentrating intense resources will have to give way to a utilitarian approach that evaluates operations based on efficiency, equity, and justice. Physician-patient contact should be minimized using technological links when possible, and rounds might be reorganized to first examine patients without COVID-19, in order to minimize between-patient spread.
Military medicine, which is used to ramping up operations during times of crisis, has potential lessons for the current pandemic. In the face of mass casualties, military physicians often turn to the North Atlantic Treaty Organization triage system, which separates patients into four categories: immediate, requiring lifesaving intervention; delayed, requiring intervention within hours to days; minimal, where the patient is injured but ambulatory; and expectant patients who are deceased or too injured to save. Impersonal though this system may be, it may be required in the most severe scenarios when resources are scarce or absent.
The authors reported no relevant financial disclosures.
SOURCE: Katz J et al. J Am Coll Cardiol. 2020 Apr 15. doi: 10.1016/j.annonc.2020.02.01.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
HFpEF: Gender difference in sacubitril/valsartan response remains mystery
The explanation for the impressive clinical benefits of sacubitril/valsartan in women with heart failure with preserved ejection fraction in the PARAGON-HF trial – but not in the men – remains elusive, Jonathan W. Cunningham, MD, said at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
“We’ve all been trying to unravel the explanation for the differential effects between men and women in the primary trial. I don’t know that this NT-proBNP substudy gives a clear answer because we did see similar reduction in NT-proBNP in the men and women,” said Dr. Cunningham of Brigham and Women’s Hospital, Boston.
“Unfortunately, I think we’re still looking for the underlying physiological explanation for that very interesting interaction,” he added.
The PARAGON-HF trial included 4,796 patients with heart failure with preserved ejection fraction (HFpEF) who were randomized double-blind to sacubitril/valsartan (Entresto) or valsartan on top of background guideline-directed medical therapy and followed for a median of 34 months (N Engl J Med. 2019 Oct 24;381[17]:1609-20). The sacubitril/valsartan group’s 13% relative risk reduction in the primary composite endpoint of cardiovascular death and total heart failure hospitalizations fell tantalizingly short of statistical significance (P = 0.058).
In women, however, who comprised more than half of the study population, the benefit of sacubitril/valsartan was larger: a 27% relative risk reduction compared to valsartan alone. That’s a statistically significant difference in a prespecified subgroup analysis, but according to the rules of clinical trials and statistics it must be considered hypothesis-generating and nondefinitive, since the overall trial was negative. Men randomized to sacubitril/valsartan had a modest 3% increased risk of the primary endpoint compared to men on valsartan.
Because of the enormous unmet need for effective therapy for HFpEF, and the fact that HFpEF is more common in women than men, the search is on for an explanation that would account for the striking gender difference in outcome in PARAGON-HF. At ACC 2020, Dr. Cunningham presented a secondary analysis of the trial focusing on the relationships between baseline and on-treatment N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels and clinical outcomes.
Among the key findings was that the higher the baseline NT-proBNP, the greater the likelihood of the primary endpoint. Also, sacubitril/valsartan reduced NT-proBNP to a similar extent in men and women: For example, by 20% compared to valsartan in men and by 18% in women when measured 16 weeks after randomization. And reduction in NT-proBNP was associated with reduced risk of cardiovascular death and heart failure hospitalizations; indeed, 60% of participants in PARAGON-HF experienced a decrease in NT-proBNP, and they had a 23% lower event rate compared to patients whose NT-proBNP increased during the course of the study.
Another intriguing finding in the parent PARAGON-HF trial was that HFpEF patients with an LVEF of 45%-57% had a 22% lower rate of the primary endpoint than those with an LVEF of 58% or more. But as with the gender difference in clinical outcomes in response to sacubitril/valsartan, the difference in outcomes based on ejection fraction was not mediated by the drug’s impact on NT-proBNP, since sacubitril/valsartan reduced NT-proBNP to a similar degree in HFpEF patients with an LVEF above or below 57%.
The difference in outcomes by ejection fraction wasn’t entirely surprising, because those low-normal–range ejection fractions where sacubitril/valsartan had a favorable impact approach those characteristic of heart failure with reduced ejection fraction (HFrEF), and guidelines give sacubitril/valsartan a class I recommendation in patients with HFrEF on the strength of the medication’s demonstrated reduction in morbidity and mortality in the PARADIGM-HF trial.
Discussant Lee R. Goldberg, MD, predicted this analysis will have an impact on the design of future clinical trials in HFpEF, which up until now have required certain minimum NT-proBNP levels for participation.
“Maybe this is why so many of our trials in HFpEF have been unsuccessful. It’s a very heterogeneous population and perhaps NT-proBNP cutoffs are leading to a lot of mischief or heterogeneity that causes us some difficulty,” said Dr. Goldberg, professor of medicine and chief of the section of advanced heart failure and cardiac transplantation at the University of Pennsylvania, Philadelphia.
Dr. Cunningham reported having no financial conflicts regarding his study. The PARAGON-HF trial was funded by Novartis.
Simultaneously with Dr. Cunningham’s presentation at ACC 2020, the study results were published online (JACC Heart Fail. 2020 Mar 26; doi: 10.1016/j.jchf.2020.03.002.
SOURCE: Cunningham JW. ACC 2020, Abstract 412-08.
The explanation for the impressive clinical benefits of sacubitril/valsartan in women with heart failure with preserved ejection fraction in the PARAGON-HF trial – but not in the men – remains elusive, Jonathan W. Cunningham, MD, said at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
“We’ve all been trying to unravel the explanation for the differential effects between men and women in the primary trial. I don’t know that this NT-proBNP substudy gives a clear answer because we did see similar reduction in NT-proBNP in the men and women,” said Dr. Cunningham of Brigham and Women’s Hospital, Boston.
“Unfortunately, I think we’re still looking for the underlying physiological explanation for that very interesting interaction,” he added.
The PARAGON-HF trial included 4,796 patients with heart failure with preserved ejection fraction (HFpEF) who were randomized double-blind to sacubitril/valsartan (Entresto) or valsartan on top of background guideline-directed medical therapy and followed for a median of 34 months (N Engl J Med. 2019 Oct 24;381[17]:1609-20). The sacubitril/valsartan group’s 13% relative risk reduction in the primary composite endpoint of cardiovascular death and total heart failure hospitalizations fell tantalizingly short of statistical significance (P = 0.058).
In women, however, who comprised more than half of the study population, the benefit of sacubitril/valsartan was larger: a 27% relative risk reduction compared to valsartan alone. That’s a statistically significant difference in a prespecified subgroup analysis, but according to the rules of clinical trials and statistics it must be considered hypothesis-generating and nondefinitive, since the overall trial was negative. Men randomized to sacubitril/valsartan had a modest 3% increased risk of the primary endpoint compared to men on valsartan.
Because of the enormous unmet need for effective therapy for HFpEF, and the fact that HFpEF is more common in women than men, the search is on for an explanation that would account for the striking gender difference in outcome in PARAGON-HF. At ACC 2020, Dr. Cunningham presented a secondary analysis of the trial focusing on the relationships between baseline and on-treatment N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels and clinical outcomes.
Among the key findings was that the higher the baseline NT-proBNP, the greater the likelihood of the primary endpoint. Also, sacubitril/valsartan reduced NT-proBNP to a similar extent in men and women: For example, by 20% compared to valsartan in men and by 18% in women when measured 16 weeks after randomization. And reduction in NT-proBNP was associated with reduced risk of cardiovascular death and heart failure hospitalizations; indeed, 60% of participants in PARAGON-HF experienced a decrease in NT-proBNP, and they had a 23% lower event rate compared to patients whose NT-proBNP increased during the course of the study.
Another intriguing finding in the parent PARAGON-HF trial was that HFpEF patients with an LVEF of 45%-57% had a 22% lower rate of the primary endpoint than those with an LVEF of 58% or more. But as with the gender difference in clinical outcomes in response to sacubitril/valsartan, the difference in outcomes based on ejection fraction was not mediated by the drug’s impact on NT-proBNP, since sacubitril/valsartan reduced NT-proBNP to a similar degree in HFpEF patients with an LVEF above or below 57%.
The difference in outcomes by ejection fraction wasn’t entirely surprising, because those low-normal–range ejection fractions where sacubitril/valsartan had a favorable impact approach those characteristic of heart failure with reduced ejection fraction (HFrEF), and guidelines give sacubitril/valsartan a class I recommendation in patients with HFrEF on the strength of the medication’s demonstrated reduction in morbidity and mortality in the PARADIGM-HF trial.
Discussant Lee R. Goldberg, MD, predicted this analysis will have an impact on the design of future clinical trials in HFpEF, which up until now have required certain minimum NT-proBNP levels for participation.
“Maybe this is why so many of our trials in HFpEF have been unsuccessful. It’s a very heterogeneous population and perhaps NT-proBNP cutoffs are leading to a lot of mischief or heterogeneity that causes us some difficulty,” said Dr. Goldberg, professor of medicine and chief of the section of advanced heart failure and cardiac transplantation at the University of Pennsylvania, Philadelphia.
Dr. Cunningham reported having no financial conflicts regarding his study. The PARAGON-HF trial was funded by Novartis.
Simultaneously with Dr. Cunningham’s presentation at ACC 2020, the study results were published online (JACC Heart Fail. 2020 Mar 26; doi: 10.1016/j.jchf.2020.03.002.
SOURCE: Cunningham JW. ACC 2020, Abstract 412-08.
The explanation for the impressive clinical benefits of sacubitril/valsartan in women with heart failure with preserved ejection fraction in the PARAGON-HF trial – but not in the men – remains elusive, Jonathan W. Cunningham, MD, said at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
“We’ve all been trying to unravel the explanation for the differential effects between men and women in the primary trial. I don’t know that this NT-proBNP substudy gives a clear answer because we did see similar reduction in NT-proBNP in the men and women,” said Dr. Cunningham of Brigham and Women’s Hospital, Boston.
“Unfortunately, I think we’re still looking for the underlying physiological explanation for that very interesting interaction,” he added.
The PARAGON-HF trial included 4,796 patients with heart failure with preserved ejection fraction (HFpEF) who were randomized double-blind to sacubitril/valsartan (Entresto) or valsartan on top of background guideline-directed medical therapy and followed for a median of 34 months (N Engl J Med. 2019 Oct 24;381[17]:1609-20). The sacubitril/valsartan group’s 13% relative risk reduction in the primary composite endpoint of cardiovascular death and total heart failure hospitalizations fell tantalizingly short of statistical significance (P = 0.058).
In women, however, who comprised more than half of the study population, the benefit of sacubitril/valsartan was larger: a 27% relative risk reduction compared to valsartan alone. That’s a statistically significant difference in a prespecified subgroup analysis, but according to the rules of clinical trials and statistics it must be considered hypothesis-generating and nondefinitive, since the overall trial was negative. Men randomized to sacubitril/valsartan had a modest 3% increased risk of the primary endpoint compared to men on valsartan.
Because of the enormous unmet need for effective therapy for HFpEF, and the fact that HFpEF is more common in women than men, the search is on for an explanation that would account for the striking gender difference in outcome in PARAGON-HF. At ACC 2020, Dr. Cunningham presented a secondary analysis of the trial focusing on the relationships between baseline and on-treatment N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels and clinical outcomes.
Among the key findings was that the higher the baseline NT-proBNP, the greater the likelihood of the primary endpoint. Also, sacubitril/valsartan reduced NT-proBNP to a similar extent in men and women: For example, by 20% compared to valsartan in men and by 18% in women when measured 16 weeks after randomization. And reduction in NT-proBNP was associated with reduced risk of cardiovascular death and heart failure hospitalizations; indeed, 60% of participants in PARAGON-HF experienced a decrease in NT-proBNP, and they had a 23% lower event rate compared to patients whose NT-proBNP increased during the course of the study.
Another intriguing finding in the parent PARAGON-HF trial was that HFpEF patients with an LVEF of 45%-57% had a 22% lower rate of the primary endpoint than those with an LVEF of 58% or more. But as with the gender difference in clinical outcomes in response to sacubitril/valsartan, the difference in outcomes based on ejection fraction was not mediated by the drug’s impact on NT-proBNP, since sacubitril/valsartan reduced NT-proBNP to a similar degree in HFpEF patients with an LVEF above or below 57%.
The difference in outcomes by ejection fraction wasn’t entirely surprising, because those low-normal–range ejection fractions where sacubitril/valsartan had a favorable impact approach those characteristic of heart failure with reduced ejection fraction (HFrEF), and guidelines give sacubitril/valsartan a class I recommendation in patients with HFrEF on the strength of the medication’s demonstrated reduction in morbidity and mortality in the PARADIGM-HF trial.
Discussant Lee R. Goldberg, MD, predicted this analysis will have an impact on the design of future clinical trials in HFpEF, which up until now have required certain minimum NT-proBNP levels for participation.
“Maybe this is why so many of our trials in HFpEF have been unsuccessful. It’s a very heterogeneous population and perhaps NT-proBNP cutoffs are leading to a lot of mischief or heterogeneity that causes us some difficulty,” said Dr. Goldberg, professor of medicine and chief of the section of advanced heart failure and cardiac transplantation at the University of Pennsylvania, Philadelphia.
Dr. Cunningham reported having no financial conflicts regarding his study. The PARAGON-HF trial was funded by Novartis.
Simultaneously with Dr. Cunningham’s presentation at ACC 2020, the study results were published online (JACC Heart Fail. 2020 Mar 26; doi: 10.1016/j.jchf.2020.03.002.
SOURCE: Cunningham JW. ACC 2020, Abstract 412-08.
FROM ACC 2020
Obesity link to severe COVID-19, especially in patients aged under 60
It is becoming increasingly clear that obesity is one of the biggest risk factors for severe COVID-19 disease, particularly among younger patients.
Newly published data from New York show that, among those aged under 60 years, obesity was twice as likely to result in hospitalization for COVID-19 and also significantly increased the likelihood that a person would end up in intensive care.
“Obesity [in people younger than 60] appears to be a previously unrecognized risk factor for hospital admission and need for critical care. This has important and practical implications when nearly 40% of adults in the U.S. are obese with a body mass index [BMI] of [at least] 30,” wrote Jennifer Lighter, MD, of New York University Langone Health, and colleagues in their research letter published in Clinical Infectious Diseases.
Similar findings in a preprint publication, yet to be peer reviewed, from another New York hospital show that, with the exception of older age, obesity (BMI greater than 40 kg/m2) had the strongest association with hospitalization for COVID-19, increasing the risk more than 500%.
Meanwhile, a new French study shows a high frequency of obesity among patients admitted to one ICU for COVID-19; furthermore, disease severity increased with increasing BMI. One of the authors said in an interview that many of the presenting patients were younger, with their only risk factor being obesity.
“Patients with obesity should avoid any COVID-19 contamination by enforcing all prevention measures during the current pandemic,” wrote the authors, led by Arthur Simonnet, MD, Centre Hospitalier Universitaire de Lille (France).
They also stressed that COVID-19 patients “with severe obesity should be monitored more closely.”
Those with obesity are young and become very sick, very quickly
François Pattou, MD, PhD, coauthor of the French article published in Obesity said in an interview that, when patients with COVID-19 began to arrive at their ICU in Lille, there were young patients who did not have any other comorbidities.
“They were just obese,” he observed, adding that they seemed “to have a very specific disease, something different” from that seen before, with patients becoming very sick, very quickly.
In their study, they examined 124 consecutive patients admitted to intensive care with COVID-19 between Feb. 25 and April 5, 2020, and compared them with a historical control group of 306 patients admitted to the ICU at the same hospital for non–COVID-19-related severe acute respiratory disease in 2019.
By April 6, 60 patients with COVID-19 had been discharged from intensive care, 18 had died, and 46 remained in the unit. The majority (73%) were male, and their median age was 60 years. Obesity and severe obesity were significantly more prevalent among the patients with COVID-19, at 47.6% and 28.2% versus 25.2% and 10.8% among historical controls (P < .001 for trend).
A key finding was that those with a BMI greater than 35 had a more than 600% increased risk of requiring mechanical ventilation (odds ratio, 7.36; P = .021), compared with those with a BMI less than 25, even after adjusting for age, diabetes, and hypertension.
Obesity in under 60s at least doubles risk of admission in U.S.
The studies out of New York, one of which was stratified by age, paint a similar picture.
Dr. Lighter and colleagues found that, of the 3,615 individuals who tested positive for COVID-19 in their series, 775 (21%) had a BMI of 30-34 and 595 (16%) had a BMI of at least 35. Obesity wasn’t a predictor of admission to hospital or the ICU in those over the age of 60 years, but in those younger than 60 years, it was.
Those under age 60 with a BMI of 30-34 were twice as likely to be admitted to hospital (hazard ratio, 2.0; P < .0001) and critical care (HR, 1.8; P = .006), compared with those under age 60 with a BMI less than 30. Likewise, those under age 60 with a BMI of at least 35 were 2.2 (P < .0001) and 3.6 (P < .0001) times more likely to be admitted to acute and critical care.
“Unfortunately, obesity in people [less than] 60 years is a newly identified epidemiologic risk factor which may contribute to increased morbidity rates [with COVID-19] experienced in the U.S.,” they concluded.
And in the other U.S. study, Christopher M. Petrilli, MD, of New York University, and colleagues looked at 4,103 patients with COVID-19 treated between March 1 and April 2, 2020, and followed to April 7.
Just under half of patients (48.7%) were hospitalized, of whom 22.3% required mechanical ventilation and 14.6% died or were discharged to hospice. The research was published on medRxiv, showing that, apart from age, the strongest predictors of hospitalization were BMI greater than 40 (OR, 6.2) and heart failure (OR, 4.3).
“It is notable that the chronic condition with the strongest association with critical illness was obesity, with a substantially higher odds ratio than any cardiovascular or pulmonary disease,” they noted.
Inflammation is a possible culprit
Dr. Pattou believes that the culprit behind the increased risk of disease severity seen with obesity in COVID-19 is inflammation, mediated by fibrin deposits in the circulation, which his colleagues have seen on autopsy, and which “block oxygen passage through the blood.”
This may help explain why mechanical ventilation can be less successful in these patients. “The answer is to get rid of this inflammation,” Dr. Pattou observed.
Dr. Petrilli and colleagues also observed that obesity “is well-recognized to be a proinflammatory condition.”
And their findings showed “the importance of inflammatory markers in distinguishing future critical from noncritical illness,” they said, noting that, among these markers, early elevations in C-reactive protein and D-dimer “had the strongest association with mechanical ventilation or mortality.”
Livio Luzi, MD, of IRCCS MultiMedica, Milan, Italy, has previously written on the relationship between influenza and obesity, and discussed in an interview the potential lessons for the COVID-19 pandemic.
“Obesity is characterized by an impairment of immune response and by a low-grade chronic inflammation. Furthermore, obese subjects have an altered dynamic of pulmonary ventilation, with reduced diaphragmatic excursion,” Dr. Luzi said. These factors, alongside others, “may help to explain” the current results, and stress the importance of close monitoring of those with obesity and COVID-19.
No relevant financial relationships were declared.
This article first appeared on Medscape.com.
It is becoming increasingly clear that obesity is one of the biggest risk factors for severe COVID-19 disease, particularly among younger patients.
Newly published data from New York show that, among those aged under 60 years, obesity was twice as likely to result in hospitalization for COVID-19 and also significantly increased the likelihood that a person would end up in intensive care.
“Obesity [in people younger than 60] appears to be a previously unrecognized risk factor for hospital admission and need for critical care. This has important and practical implications when nearly 40% of adults in the U.S. are obese with a body mass index [BMI] of [at least] 30,” wrote Jennifer Lighter, MD, of New York University Langone Health, and colleagues in their research letter published in Clinical Infectious Diseases.
Similar findings in a preprint publication, yet to be peer reviewed, from another New York hospital show that, with the exception of older age, obesity (BMI greater than 40 kg/m2) had the strongest association with hospitalization for COVID-19, increasing the risk more than 500%.
Meanwhile, a new French study shows a high frequency of obesity among patients admitted to one ICU for COVID-19; furthermore, disease severity increased with increasing BMI. One of the authors said in an interview that many of the presenting patients were younger, with their only risk factor being obesity.
“Patients with obesity should avoid any COVID-19 contamination by enforcing all prevention measures during the current pandemic,” wrote the authors, led by Arthur Simonnet, MD, Centre Hospitalier Universitaire de Lille (France).
They also stressed that COVID-19 patients “with severe obesity should be monitored more closely.”
Those with obesity are young and become very sick, very quickly
François Pattou, MD, PhD, coauthor of the French article published in Obesity said in an interview that, when patients with COVID-19 began to arrive at their ICU in Lille, there were young patients who did not have any other comorbidities.
“They were just obese,” he observed, adding that they seemed “to have a very specific disease, something different” from that seen before, with patients becoming very sick, very quickly.
In their study, they examined 124 consecutive patients admitted to intensive care with COVID-19 between Feb. 25 and April 5, 2020, and compared them with a historical control group of 306 patients admitted to the ICU at the same hospital for non–COVID-19-related severe acute respiratory disease in 2019.
By April 6, 60 patients with COVID-19 had been discharged from intensive care, 18 had died, and 46 remained in the unit. The majority (73%) were male, and their median age was 60 years. Obesity and severe obesity were significantly more prevalent among the patients with COVID-19, at 47.6% and 28.2% versus 25.2% and 10.8% among historical controls (P < .001 for trend).
A key finding was that those with a BMI greater than 35 had a more than 600% increased risk of requiring mechanical ventilation (odds ratio, 7.36; P = .021), compared with those with a BMI less than 25, even after adjusting for age, diabetes, and hypertension.
Obesity in under 60s at least doubles risk of admission in U.S.
The studies out of New York, one of which was stratified by age, paint a similar picture.
Dr. Lighter and colleagues found that, of the 3,615 individuals who tested positive for COVID-19 in their series, 775 (21%) had a BMI of 30-34 and 595 (16%) had a BMI of at least 35. Obesity wasn’t a predictor of admission to hospital or the ICU in those over the age of 60 years, but in those younger than 60 years, it was.
Those under age 60 with a BMI of 30-34 were twice as likely to be admitted to hospital (hazard ratio, 2.0; P < .0001) and critical care (HR, 1.8; P = .006), compared with those under age 60 with a BMI less than 30. Likewise, those under age 60 with a BMI of at least 35 were 2.2 (P < .0001) and 3.6 (P < .0001) times more likely to be admitted to acute and critical care.
“Unfortunately, obesity in people [less than] 60 years is a newly identified epidemiologic risk factor which may contribute to increased morbidity rates [with COVID-19] experienced in the U.S.,” they concluded.
And in the other U.S. study, Christopher M. Petrilli, MD, of New York University, and colleagues looked at 4,103 patients with COVID-19 treated between March 1 and April 2, 2020, and followed to April 7.
Just under half of patients (48.7%) were hospitalized, of whom 22.3% required mechanical ventilation and 14.6% died or were discharged to hospice. The research was published on medRxiv, showing that, apart from age, the strongest predictors of hospitalization were BMI greater than 40 (OR, 6.2) and heart failure (OR, 4.3).
“It is notable that the chronic condition with the strongest association with critical illness was obesity, with a substantially higher odds ratio than any cardiovascular or pulmonary disease,” they noted.
Inflammation is a possible culprit
Dr. Pattou believes that the culprit behind the increased risk of disease severity seen with obesity in COVID-19 is inflammation, mediated by fibrin deposits in the circulation, which his colleagues have seen on autopsy, and which “block oxygen passage through the blood.”
This may help explain why mechanical ventilation can be less successful in these patients. “The answer is to get rid of this inflammation,” Dr. Pattou observed.
Dr. Petrilli and colleagues also observed that obesity “is well-recognized to be a proinflammatory condition.”
And their findings showed “the importance of inflammatory markers in distinguishing future critical from noncritical illness,” they said, noting that, among these markers, early elevations in C-reactive protein and D-dimer “had the strongest association with mechanical ventilation or mortality.”
Livio Luzi, MD, of IRCCS MultiMedica, Milan, Italy, has previously written on the relationship between influenza and obesity, and discussed in an interview the potential lessons for the COVID-19 pandemic.
“Obesity is characterized by an impairment of immune response and by a low-grade chronic inflammation. Furthermore, obese subjects have an altered dynamic of pulmonary ventilation, with reduced diaphragmatic excursion,” Dr. Luzi said. These factors, alongside others, “may help to explain” the current results, and stress the importance of close monitoring of those with obesity and COVID-19.
No relevant financial relationships were declared.
This article first appeared on Medscape.com.
It is becoming increasingly clear that obesity is one of the biggest risk factors for severe COVID-19 disease, particularly among younger patients.
Newly published data from New York show that, among those aged under 60 years, obesity was twice as likely to result in hospitalization for COVID-19 and also significantly increased the likelihood that a person would end up in intensive care.
“Obesity [in people younger than 60] appears to be a previously unrecognized risk factor for hospital admission and need for critical care. This has important and practical implications when nearly 40% of adults in the U.S. are obese with a body mass index [BMI] of [at least] 30,” wrote Jennifer Lighter, MD, of New York University Langone Health, and colleagues in their research letter published in Clinical Infectious Diseases.
Similar findings in a preprint publication, yet to be peer reviewed, from another New York hospital show that, with the exception of older age, obesity (BMI greater than 40 kg/m2) had the strongest association with hospitalization for COVID-19, increasing the risk more than 500%.
Meanwhile, a new French study shows a high frequency of obesity among patients admitted to one ICU for COVID-19; furthermore, disease severity increased with increasing BMI. One of the authors said in an interview that many of the presenting patients were younger, with their only risk factor being obesity.
“Patients with obesity should avoid any COVID-19 contamination by enforcing all prevention measures during the current pandemic,” wrote the authors, led by Arthur Simonnet, MD, Centre Hospitalier Universitaire de Lille (France).
They also stressed that COVID-19 patients “with severe obesity should be monitored more closely.”
Those with obesity are young and become very sick, very quickly
François Pattou, MD, PhD, coauthor of the French article published in Obesity said in an interview that, when patients with COVID-19 began to arrive at their ICU in Lille, there were young patients who did not have any other comorbidities.
“They were just obese,” he observed, adding that they seemed “to have a very specific disease, something different” from that seen before, with patients becoming very sick, very quickly.
In their study, they examined 124 consecutive patients admitted to intensive care with COVID-19 between Feb. 25 and April 5, 2020, and compared them with a historical control group of 306 patients admitted to the ICU at the same hospital for non–COVID-19-related severe acute respiratory disease in 2019.
By April 6, 60 patients with COVID-19 had been discharged from intensive care, 18 had died, and 46 remained in the unit. The majority (73%) were male, and their median age was 60 years. Obesity and severe obesity were significantly more prevalent among the patients with COVID-19, at 47.6% and 28.2% versus 25.2% and 10.8% among historical controls (P < .001 for trend).
A key finding was that those with a BMI greater than 35 had a more than 600% increased risk of requiring mechanical ventilation (odds ratio, 7.36; P = .021), compared with those with a BMI less than 25, even after adjusting for age, diabetes, and hypertension.
Obesity in under 60s at least doubles risk of admission in U.S.
The studies out of New York, one of which was stratified by age, paint a similar picture.
Dr. Lighter and colleagues found that, of the 3,615 individuals who tested positive for COVID-19 in their series, 775 (21%) had a BMI of 30-34 and 595 (16%) had a BMI of at least 35. Obesity wasn’t a predictor of admission to hospital or the ICU in those over the age of 60 years, but in those younger than 60 years, it was.
Those under age 60 with a BMI of 30-34 were twice as likely to be admitted to hospital (hazard ratio, 2.0; P < .0001) and critical care (HR, 1.8; P = .006), compared with those under age 60 with a BMI less than 30. Likewise, those under age 60 with a BMI of at least 35 were 2.2 (P < .0001) and 3.6 (P < .0001) times more likely to be admitted to acute and critical care.
“Unfortunately, obesity in people [less than] 60 years is a newly identified epidemiologic risk factor which may contribute to increased morbidity rates [with COVID-19] experienced in the U.S.,” they concluded.
And in the other U.S. study, Christopher M. Petrilli, MD, of New York University, and colleagues looked at 4,103 patients with COVID-19 treated between March 1 and April 2, 2020, and followed to April 7.
Just under half of patients (48.7%) were hospitalized, of whom 22.3% required mechanical ventilation and 14.6% died or were discharged to hospice. The research was published on medRxiv, showing that, apart from age, the strongest predictors of hospitalization were BMI greater than 40 (OR, 6.2) and heart failure (OR, 4.3).
“It is notable that the chronic condition with the strongest association with critical illness was obesity, with a substantially higher odds ratio than any cardiovascular or pulmonary disease,” they noted.
Inflammation is a possible culprit
Dr. Pattou believes that the culprit behind the increased risk of disease severity seen with obesity in COVID-19 is inflammation, mediated by fibrin deposits in the circulation, which his colleagues have seen on autopsy, and which “block oxygen passage through the blood.”
This may help explain why mechanical ventilation can be less successful in these patients. “The answer is to get rid of this inflammation,” Dr. Pattou observed.
Dr. Petrilli and colleagues also observed that obesity “is well-recognized to be a proinflammatory condition.”
And their findings showed “the importance of inflammatory markers in distinguishing future critical from noncritical illness,” they said, noting that, among these markers, early elevations in C-reactive protein and D-dimer “had the strongest association with mechanical ventilation or mortality.”
Livio Luzi, MD, of IRCCS MultiMedica, Milan, Italy, has previously written on the relationship between influenza and obesity, and discussed in an interview the potential lessons for the COVID-19 pandemic.
“Obesity is characterized by an impairment of immune response and by a low-grade chronic inflammation. Furthermore, obese subjects have an altered dynamic of pulmonary ventilation, with reduced diaphragmatic excursion,” Dr. Luzi said. These factors, alongside others, “may help to explain” the current results, and stress the importance of close monitoring of those with obesity and COVID-19.
No relevant financial relationships were declared.
This article first appeared on Medscape.com.