User login
Complications and death within 30 days after noncardiac surgery
Background: There have been advances in perioperative care and technology for adults, but at the same time the patient population is increasingly medically complex. We do not know the current mortality risk of noncardiac surgery in adults.
Study design: Prospective cohort study.
Setting: Twenty-eight academic centers in 14 countries in North America, South America, Asia, Europe, Africa, and Australia. At least four academic centers represented each of these continents, except Africa, with one center reporting there.
Synopsis: The VISION study included 40,004 inpatients, aged 45 years and older, followed for 30-day mortality after noncardiac surgery. One-third of surgeries were considered low risk. A startling 99.1% of patients completed the study. Mortality rate was 1.8%, with 71% of patients dying during the index admission and 29% dying after discharge.
Nine events were independently associated with postoperative death, but the top three – major bleeding, myocardial injury after noncardiac surgery (MINS), and sepsis – accounted for 45% of the attributable fraction. These, on average, occurred within 1-6 days after surgery. The other events (infection, kidney injury with dialysis, stroke, venous thromboembolism, new atrial fibrillation, and congestive heart failure) constituted less than 3% of the attributable fraction. Findings suggest that closer monitoring in the hospital and post discharge might improve survival after noncardiac surgery.
Limitations for hospitalists include that patients were younger and less medically complex than our typically comanaged patients: More than half of patients were aged 45-64, less than 10% had chronic kidney disease stage 3b or greater, and only 20% had diabetes mellitus.
Bottom line: Postoperative and postdischarge bleeding, myocardial injury after noncardiac surgery, and sepsis are major risk factors for 30-day mortality in adults undergoing noncardiac surgery. Closer postoperative monitoring for these conditions should be explored.
Citation: The Vision Study Investigators (Spence J et al.) Association between complications and death within 30 days after noncardiac surgery. CMAJ. 2019 Jul 29;191(30):E830-7.
Dr. Brouillette is a med-peds hospitalist at Maine Medical Center in Portland.
Background: There have been advances in perioperative care and technology for adults, but at the same time the patient population is increasingly medically complex. We do not know the current mortality risk of noncardiac surgery in adults.
Study design: Prospective cohort study.
Setting: Twenty-eight academic centers in 14 countries in North America, South America, Asia, Europe, Africa, and Australia. At least four academic centers represented each of these continents, except Africa, with one center reporting there.
Synopsis: The VISION study included 40,004 inpatients, aged 45 years and older, followed for 30-day mortality after noncardiac surgery. One-third of surgeries were considered low risk. A startling 99.1% of patients completed the study. Mortality rate was 1.8%, with 71% of patients dying during the index admission and 29% dying after discharge.
Nine events were independently associated with postoperative death, but the top three – major bleeding, myocardial injury after noncardiac surgery (MINS), and sepsis – accounted for 45% of the attributable fraction. These, on average, occurred within 1-6 days after surgery. The other events (infection, kidney injury with dialysis, stroke, venous thromboembolism, new atrial fibrillation, and congestive heart failure) constituted less than 3% of the attributable fraction. Findings suggest that closer monitoring in the hospital and post discharge might improve survival after noncardiac surgery.
Limitations for hospitalists include that patients were younger and less medically complex than our typically comanaged patients: More than half of patients were aged 45-64, less than 10% had chronic kidney disease stage 3b or greater, and only 20% had diabetes mellitus.
Bottom line: Postoperative and postdischarge bleeding, myocardial injury after noncardiac surgery, and sepsis are major risk factors for 30-day mortality in adults undergoing noncardiac surgery. Closer postoperative monitoring for these conditions should be explored.
Citation: The Vision Study Investigators (Spence J et al.) Association between complications and death within 30 days after noncardiac surgery. CMAJ. 2019 Jul 29;191(30):E830-7.
Dr. Brouillette is a med-peds hospitalist at Maine Medical Center in Portland.
Background: There have been advances in perioperative care and technology for adults, but at the same time the patient population is increasingly medically complex. We do not know the current mortality risk of noncardiac surgery in adults.
Study design: Prospective cohort study.
Setting: Twenty-eight academic centers in 14 countries in North America, South America, Asia, Europe, Africa, and Australia. At least four academic centers represented each of these continents, except Africa, with one center reporting there.
Synopsis: The VISION study included 40,004 inpatients, aged 45 years and older, followed for 30-day mortality after noncardiac surgery. One-third of surgeries were considered low risk. A startling 99.1% of patients completed the study. Mortality rate was 1.8%, with 71% of patients dying during the index admission and 29% dying after discharge.
Nine events were independently associated with postoperative death, but the top three – major bleeding, myocardial injury after noncardiac surgery (MINS), and sepsis – accounted for 45% of the attributable fraction. These, on average, occurred within 1-6 days after surgery. The other events (infection, kidney injury with dialysis, stroke, venous thromboembolism, new atrial fibrillation, and congestive heart failure) constituted less than 3% of the attributable fraction. Findings suggest that closer monitoring in the hospital and post discharge might improve survival after noncardiac surgery.
Limitations for hospitalists include that patients were younger and less medically complex than our typically comanaged patients: More than half of patients were aged 45-64, less than 10% had chronic kidney disease stage 3b or greater, and only 20% had diabetes mellitus.
Bottom line: Postoperative and postdischarge bleeding, myocardial injury after noncardiac surgery, and sepsis are major risk factors for 30-day mortality in adults undergoing noncardiac surgery. Closer postoperative monitoring for these conditions should be explored.
Citation: The Vision Study Investigators (Spence J et al.) Association between complications and death within 30 days after noncardiac surgery. CMAJ. 2019 Jul 29;191(30):E830-7.
Dr. Brouillette is a med-peds hospitalist at Maine Medical Center in Portland.
VA Ramps up Vaccinations as COVID-19 Cases Continue to Rise
Updated January 12, 2020
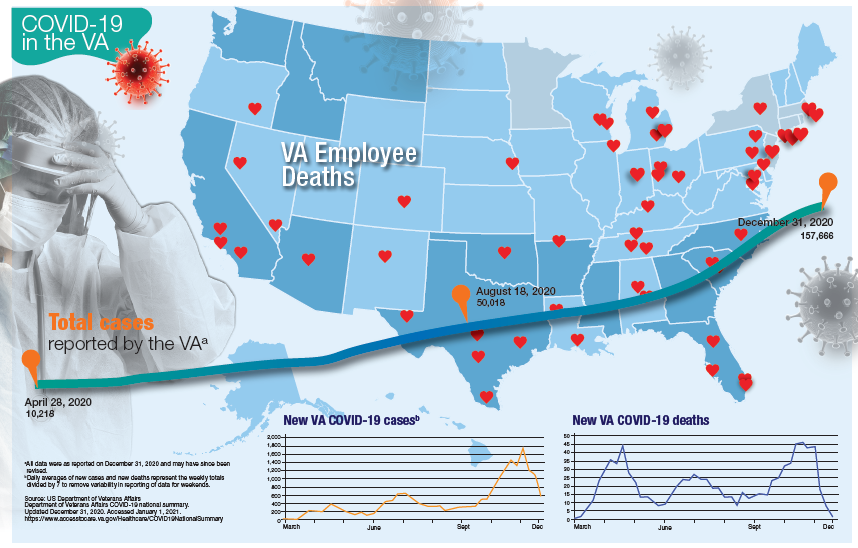
More than 181,000 veterans have contracted the COVID-19 virus and 7,385 have died, according to data released by the US Department of Veterans Affairs (VA) on January 12, 2020. The number of cases and deaths have increased sharply since November 2020. The VA also reports that it has administered at least 1 dose of the 2 approved vaccines to 33,875 veterans and 174,724 employees as of January 6.
Currently, the VA reports nearly 19,000 active cases of COVID-19, including 1,270 among VA employees. One hundred five VA employees have died from COVID-19.
Although facilities across the country are facing increased pressure as the number of cases rise, those in Southern California and Texas are reporting significant infection rates. Thirteen facilities have at least 300 active cases, including facilities in Loma Linda (418), Long Beach (381), Greater Los Angeles (361), and San Diego (274), all in California. In Texas, San Antonio (394), Dallas (370), Temple (338), and Houston (328) have all seen large numbers of active cases. Facilities in Columbia, South Carolina (420); Phoenix (407); Atlanta, Georgia (359); Cleveland, Ohio (352); and Orlando, (341) and Gainesville, Florida (340) also have reported significant numbers of cases.
While early on in the pandemic facilities in New York and New Jersey had reported the largest number of deaths, now nearly every facility has reported at least 1 death. Fourteen facilities have reported at least 100 deaths and 53 have reported between 50 and 99 deaths. The 7,385 VA COVID-19 deaths represent 2.0% of the 375,300 deaths reported in the US by Johns Hopkins University. VA has reported 0.8% of the total number of COVID-19 cases.
The VA also reports the demographic breakdown of its COVID-19 cases. Among the active cases, 56.9% are White, 18.3% Black, 9.4% Hispanic, and 1.4% Native American, Alaska Native, or Pacific Islander.

Updated January 12, 2020
More than 181,000 veterans have contracted the COVID-19 virus and 7,385 have died, according to data released by the US Department of Veterans Affairs (VA) on January 12, 2020. The number of cases and deaths have increased sharply since November 2020. The VA also reports that it has administered at least 1 dose of the 2 approved vaccines to 33,875 veterans and 174,724 employees as of January 6.
Currently, the VA reports nearly 19,000 active cases of COVID-19, including 1,270 among VA employees. One hundred five VA employees have died from COVID-19.
Although facilities across the country are facing increased pressure as the number of cases rise, those in Southern California and Texas are reporting significant infection rates. Thirteen facilities have at least 300 active cases, including facilities in Loma Linda (418), Long Beach (381), Greater Los Angeles (361), and San Diego (274), all in California. In Texas, San Antonio (394), Dallas (370), Temple (338), and Houston (328) have all seen large numbers of active cases. Facilities in Columbia, South Carolina (420); Phoenix (407); Atlanta, Georgia (359); Cleveland, Ohio (352); and Orlando, (341) and Gainesville, Florida (340) also have reported significant numbers of cases.
While early on in the pandemic facilities in New York and New Jersey had reported the largest number of deaths, now nearly every facility has reported at least 1 death. Fourteen facilities have reported at least 100 deaths and 53 have reported between 50 and 99 deaths. The 7,385 VA COVID-19 deaths represent 2.0% of the 375,300 deaths reported in the US by Johns Hopkins University. VA has reported 0.8% of the total number of COVID-19 cases.
The VA also reports the demographic breakdown of its COVID-19 cases. Among the active cases, 56.9% are White, 18.3% Black, 9.4% Hispanic, and 1.4% Native American, Alaska Native, or Pacific Islander.

Updated January 12, 2020
More than 181,000 veterans have contracted the COVID-19 virus and 7,385 have died, according to data released by the US Department of Veterans Affairs (VA) on January 12, 2020. The number of cases and deaths have increased sharply since November 2020. The VA also reports that it has administered at least 1 dose of the 2 approved vaccines to 33,875 veterans and 174,724 employees as of January 6.
Currently, the VA reports nearly 19,000 active cases of COVID-19, including 1,270 among VA employees. One hundred five VA employees have died from COVID-19.
Although facilities across the country are facing increased pressure as the number of cases rise, those in Southern California and Texas are reporting significant infection rates. Thirteen facilities have at least 300 active cases, including facilities in Loma Linda (418), Long Beach (381), Greater Los Angeles (361), and San Diego (274), all in California. In Texas, San Antonio (394), Dallas (370), Temple (338), and Houston (328) have all seen large numbers of active cases. Facilities in Columbia, South Carolina (420); Phoenix (407); Atlanta, Georgia (359); Cleveland, Ohio (352); and Orlando, (341) and Gainesville, Florida (340) also have reported significant numbers of cases.
While early on in the pandemic facilities in New York and New Jersey had reported the largest number of deaths, now nearly every facility has reported at least 1 death. Fourteen facilities have reported at least 100 deaths and 53 have reported between 50 and 99 deaths. The 7,385 VA COVID-19 deaths represent 2.0% of the 375,300 deaths reported in the US by Johns Hopkins University. VA has reported 0.8% of the total number of COVID-19 cases.
The VA also reports the demographic breakdown of its COVID-19 cases. Among the active cases, 56.9% are White, 18.3% Black, 9.4% Hispanic, and 1.4% Native American, Alaska Native, or Pacific Islander.
Over half of COVID-19 transmission may occur via asymptomatic people
As COVID-19 cases surge and vaccinations lag, health authorities continue to seek additional ways to mitigate the spread of the novel coronavirus.
Now, a modeling study estimates that more than half of transmissions come from pre-, never-, and asymptomatic individuals, indicating that symptom-based screening will have little effect on spread.
The Centers for Disease Control and Prevention study, published online Jan. 7 in JAMA Network Open, concludes that for optimal control, protective measures such as masking and social distancing should be supplemented with strategic testing of potentially exposed but asymptomatic individuals .
“In the absence of effective and widespread use of therapeutics or vaccines that can shorten or eliminate infectivity, successful control of SARS-CoV-2 cannot rely solely on identifying and isolating symptomatic cases; even if implemented effectively, this strategy would be insufficient,” CDC biologist Michael J. Johansson, PhD, and colleagues warn. “Multiple measures that effectively address transmission risk in the absence of symptoms are imperative to control SARS-CoV-2.”
According to the authors, the effectiveness of some current transmission prevention efforts has been disputed and subject to mixed messaging. Therefore, they decided to model the proportion of COVID-19 infections that are likely the result of individuals who show no symptoms and may be unknowingly infecting others.
“Unfortunately, there continues to be some skepticism about the value of community-wide mitigation efforts for preventing transmission such as masking, distancing, and hand hygiene, particularly for people without symptoms,” corresponding author Jay C. Butler, MD, said in an interview. “So we wanted to have a base assumption about how much transmission occurs from asymptomatic people to underscore the importance of mitigation measures and of creating immunity through vaccine delivery.”
Such a yardstick is especially germane in the context of the new, more transmissible variant. “It really puts [things] in a bigger box and underscores, boldfaces, and italicizes the need to change people’s behaviors and the importance of mitigation,” Dr. Butler said. It also highlights the advisability of targeted strategic testing in congregate settings, schools, and universities, which is already underway.
The analysis
Based on data from several COVID-19 studies from last year, the CDC’s analytical model assumes at baseline that infectiousness peaks at the median point of symptom onset, and that 30% of infected individuals never develop symptoms but are nevertheless 75% as infectious as those who develop overt symptoms.
The investigators then model multiple scenarios of transmission based pre- and never-symptomatic individuals, assuming different incubation and infectious periods, and varying numbers of days from point of infection to symptom onset.
When combined, the models predicts that 59% of all transmission would come from asymptomatic transmission – 35% from presymptomatic individuals and 24% from never-symptomatic individuals.
The findings complement those of an earlier CDC analysis, according to the authors.
The overall proportion of transmission from presymptomatic and never-symptomatic individuals is key to identifying mitigation measures that may be able to control SARS-CoV-2, the authors stated.
For example, they explain, if the infection reproduction number (R) in a particular setting is 2.0, a reduction in transmission of at least 50% is needed in order to reduce R to below 1.0. “Given that in some settings R is likely much greater than 2 and more than half of transmissions may come from individuals who are asymptomatic at the time of transmission, effective control must mitigate transmission risk from people without symptoms,” they wrote.
The authors acknowledge that the study applies a simplistic model to a complex and evolving phenomenon, and that the exact proportions of presymptomatic and never-symptomatic transmission and the incubation periods are not known. They also note symptoms and transmissions appear to vary across different population groups, with older individuals more likely than younger persons to experience symptoms, according to previous studies.
“Assume that everyone is potentially infected”
Other experts agree that expanded testing of asymptomatic individuals is important. “Screening for fever and isolation of symptomatic individuals is a common-sense approach to help prevent spread, but these measures are by no means adequate since it’s been clearly documented that individuals who are either asymptomatic or presymptomatic can still spread the virus,” said Brett Williams, MD, an infectious disease specialist and assistant professor of medicine at Rush University in Chicago.
“As we saw with the White House Rose Garden superspreader outbreak, testing does not reliably exclude infection either because the tested individual has not yet become positive or the test is falsely negative,” Dr. Williams, who was not involved in the CDC study, said in an interview. He further noted that when prevalence is as high as it currently is in the United States, the rate of false negatives will be high because a large proportion of those screened will be unknowingly infected.
At his center, all visitors and staff are screened with a temperature probe on entry, and since the earliest days of the pandemic, universal masking has been required. “Nationally there have been many instances of hospital break room outbreaks because of staff eating lunch together, and these outbreaks also demonstrate the incompleteness of symptomatic isolation,” Dr. Williams said.
For his part, virologist Frank Esper, MD, a pediatric infectious disease specialist at the Cleveland Clinic, said that while it’s been understood for some time that many infected people will not exhibit symptoms, “the question that remains is just how infectious are they?”
Dr. Esper’s takeaway from the modeling study is not so much that we need more screening of possibly exposed but asymptomatic people, but rather testing symptomatic people and tracing their contacts is not enough.
“We need to continue to assume that everyone is potentially infected whether they know it or not. And even though we have ramped up our testing to a much greater capacity than in the first wave, we need to continue to wear masks and socially distance because just identifying people who are sick and isolating or quarantining them is not going to be enough to contain the pandemic.”
And although assumption-based modeling is helpful, it cannot tell us “how many asymptomatic people are actually infected,” said Dr. Esper, who was not involved in the CDC study.
Dr. Esper also pointed out that the study estimates are based on data from early Chinese studies, but the virus has since changed. The new, more transmissible strain in the United States and elsewhere may involve not only more infections but also a longer presymptomatic stage. “So the CDC study may actually undershoot asymptomatic infections,” he said.
He also agreed with the authors that when it comes to infection, not all humans are equal. “Older people tend to be more symptomatic and become symptomatic more quickly so the asymptomatic rate is not the same across board from young people age 20 to older people.”
The bottom line, said David. A. Hirschwerk, MD, an infectious disease specialist at Northwell Health in Manhasset, N.Y., is that these data support the maintenance of protective measures we’ve been taking over the past months. “They support the concept that asymptomatic people are a significant source of transmission and that we need to adhere to mask wearing and social distancing, particularly indoors,” Dr. Hirschwerk, who was not involved in the analysis, said in an interview. “More testing would be better but it has to be fast and it has to be efficient, and there are a lot of challenges to overcome.”
The study was done as part of the CDC’s coronavirus disease 2019 response and was supported solely by federal base and response funding. The authors and commentators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As COVID-19 cases surge and vaccinations lag, health authorities continue to seek additional ways to mitigate the spread of the novel coronavirus.
Now, a modeling study estimates that more than half of transmissions come from pre-, never-, and asymptomatic individuals, indicating that symptom-based screening will have little effect on spread.
The Centers for Disease Control and Prevention study, published online Jan. 7 in JAMA Network Open, concludes that for optimal control, protective measures such as masking and social distancing should be supplemented with strategic testing of potentially exposed but asymptomatic individuals .
“In the absence of effective and widespread use of therapeutics or vaccines that can shorten or eliminate infectivity, successful control of SARS-CoV-2 cannot rely solely on identifying and isolating symptomatic cases; even if implemented effectively, this strategy would be insufficient,” CDC biologist Michael J. Johansson, PhD, and colleagues warn. “Multiple measures that effectively address transmission risk in the absence of symptoms are imperative to control SARS-CoV-2.”
According to the authors, the effectiveness of some current transmission prevention efforts has been disputed and subject to mixed messaging. Therefore, they decided to model the proportion of COVID-19 infections that are likely the result of individuals who show no symptoms and may be unknowingly infecting others.
“Unfortunately, there continues to be some skepticism about the value of community-wide mitigation efforts for preventing transmission such as masking, distancing, and hand hygiene, particularly for people without symptoms,” corresponding author Jay C. Butler, MD, said in an interview. “So we wanted to have a base assumption about how much transmission occurs from asymptomatic people to underscore the importance of mitigation measures and of creating immunity through vaccine delivery.”
Such a yardstick is especially germane in the context of the new, more transmissible variant. “It really puts [things] in a bigger box and underscores, boldfaces, and italicizes the need to change people’s behaviors and the importance of mitigation,” Dr. Butler said. It also highlights the advisability of targeted strategic testing in congregate settings, schools, and universities, which is already underway.
The analysis
Based on data from several COVID-19 studies from last year, the CDC’s analytical model assumes at baseline that infectiousness peaks at the median point of symptom onset, and that 30% of infected individuals never develop symptoms but are nevertheless 75% as infectious as those who develop overt symptoms.
The investigators then model multiple scenarios of transmission based pre- and never-symptomatic individuals, assuming different incubation and infectious periods, and varying numbers of days from point of infection to symptom onset.
When combined, the models predicts that 59% of all transmission would come from asymptomatic transmission – 35% from presymptomatic individuals and 24% from never-symptomatic individuals.
The findings complement those of an earlier CDC analysis, according to the authors.
The overall proportion of transmission from presymptomatic and never-symptomatic individuals is key to identifying mitigation measures that may be able to control SARS-CoV-2, the authors stated.
For example, they explain, if the infection reproduction number (R) in a particular setting is 2.0, a reduction in transmission of at least 50% is needed in order to reduce R to below 1.0. “Given that in some settings R is likely much greater than 2 and more than half of transmissions may come from individuals who are asymptomatic at the time of transmission, effective control must mitigate transmission risk from people without symptoms,” they wrote.
The authors acknowledge that the study applies a simplistic model to a complex and evolving phenomenon, and that the exact proportions of presymptomatic and never-symptomatic transmission and the incubation periods are not known. They also note symptoms and transmissions appear to vary across different population groups, with older individuals more likely than younger persons to experience symptoms, according to previous studies.
“Assume that everyone is potentially infected”
Other experts agree that expanded testing of asymptomatic individuals is important. “Screening for fever and isolation of symptomatic individuals is a common-sense approach to help prevent spread, but these measures are by no means adequate since it’s been clearly documented that individuals who are either asymptomatic or presymptomatic can still spread the virus,” said Brett Williams, MD, an infectious disease specialist and assistant professor of medicine at Rush University in Chicago.
“As we saw with the White House Rose Garden superspreader outbreak, testing does not reliably exclude infection either because the tested individual has not yet become positive or the test is falsely negative,” Dr. Williams, who was not involved in the CDC study, said in an interview. He further noted that when prevalence is as high as it currently is in the United States, the rate of false negatives will be high because a large proportion of those screened will be unknowingly infected.
At his center, all visitors and staff are screened with a temperature probe on entry, and since the earliest days of the pandemic, universal masking has been required. “Nationally there have been many instances of hospital break room outbreaks because of staff eating lunch together, and these outbreaks also demonstrate the incompleteness of symptomatic isolation,” Dr. Williams said.
For his part, virologist Frank Esper, MD, a pediatric infectious disease specialist at the Cleveland Clinic, said that while it’s been understood for some time that many infected people will not exhibit symptoms, “the question that remains is just how infectious are they?”
Dr. Esper’s takeaway from the modeling study is not so much that we need more screening of possibly exposed but asymptomatic people, but rather testing symptomatic people and tracing their contacts is not enough.
“We need to continue to assume that everyone is potentially infected whether they know it or not. And even though we have ramped up our testing to a much greater capacity than in the first wave, we need to continue to wear masks and socially distance because just identifying people who are sick and isolating or quarantining them is not going to be enough to contain the pandemic.”
And although assumption-based modeling is helpful, it cannot tell us “how many asymptomatic people are actually infected,” said Dr. Esper, who was not involved in the CDC study.
Dr. Esper also pointed out that the study estimates are based on data from early Chinese studies, but the virus has since changed. The new, more transmissible strain in the United States and elsewhere may involve not only more infections but also a longer presymptomatic stage. “So the CDC study may actually undershoot asymptomatic infections,” he said.
He also agreed with the authors that when it comes to infection, not all humans are equal. “Older people tend to be more symptomatic and become symptomatic more quickly so the asymptomatic rate is not the same across board from young people age 20 to older people.”
The bottom line, said David. A. Hirschwerk, MD, an infectious disease specialist at Northwell Health in Manhasset, N.Y., is that these data support the maintenance of protective measures we’ve been taking over the past months. “They support the concept that asymptomatic people are a significant source of transmission and that we need to adhere to mask wearing and social distancing, particularly indoors,” Dr. Hirschwerk, who was not involved in the analysis, said in an interview. “More testing would be better but it has to be fast and it has to be efficient, and there are a lot of challenges to overcome.”
The study was done as part of the CDC’s coronavirus disease 2019 response and was supported solely by federal base and response funding. The authors and commentators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
As COVID-19 cases surge and vaccinations lag, health authorities continue to seek additional ways to mitigate the spread of the novel coronavirus.
Now, a modeling study estimates that more than half of transmissions come from pre-, never-, and asymptomatic individuals, indicating that symptom-based screening will have little effect on spread.
The Centers for Disease Control and Prevention study, published online Jan. 7 in JAMA Network Open, concludes that for optimal control, protective measures such as masking and social distancing should be supplemented with strategic testing of potentially exposed but asymptomatic individuals .
“In the absence of effective and widespread use of therapeutics or vaccines that can shorten or eliminate infectivity, successful control of SARS-CoV-2 cannot rely solely on identifying and isolating symptomatic cases; even if implemented effectively, this strategy would be insufficient,” CDC biologist Michael J. Johansson, PhD, and colleagues warn. “Multiple measures that effectively address transmission risk in the absence of symptoms are imperative to control SARS-CoV-2.”
According to the authors, the effectiveness of some current transmission prevention efforts has been disputed and subject to mixed messaging. Therefore, they decided to model the proportion of COVID-19 infections that are likely the result of individuals who show no symptoms and may be unknowingly infecting others.
“Unfortunately, there continues to be some skepticism about the value of community-wide mitigation efforts for preventing transmission such as masking, distancing, and hand hygiene, particularly for people without symptoms,” corresponding author Jay C. Butler, MD, said in an interview. “So we wanted to have a base assumption about how much transmission occurs from asymptomatic people to underscore the importance of mitigation measures and of creating immunity through vaccine delivery.”
Such a yardstick is especially germane in the context of the new, more transmissible variant. “It really puts [things] in a bigger box and underscores, boldfaces, and italicizes the need to change people’s behaviors and the importance of mitigation,” Dr. Butler said. It also highlights the advisability of targeted strategic testing in congregate settings, schools, and universities, which is already underway.
The analysis
Based on data from several COVID-19 studies from last year, the CDC’s analytical model assumes at baseline that infectiousness peaks at the median point of symptom onset, and that 30% of infected individuals never develop symptoms but are nevertheless 75% as infectious as those who develop overt symptoms.
The investigators then model multiple scenarios of transmission based pre- and never-symptomatic individuals, assuming different incubation and infectious periods, and varying numbers of days from point of infection to symptom onset.
When combined, the models predicts that 59% of all transmission would come from asymptomatic transmission – 35% from presymptomatic individuals and 24% from never-symptomatic individuals.
The findings complement those of an earlier CDC analysis, according to the authors.
The overall proportion of transmission from presymptomatic and never-symptomatic individuals is key to identifying mitigation measures that may be able to control SARS-CoV-2, the authors stated.
For example, they explain, if the infection reproduction number (R) in a particular setting is 2.0, a reduction in transmission of at least 50% is needed in order to reduce R to below 1.0. “Given that in some settings R is likely much greater than 2 and more than half of transmissions may come from individuals who are asymptomatic at the time of transmission, effective control must mitigate transmission risk from people without symptoms,” they wrote.
The authors acknowledge that the study applies a simplistic model to a complex and evolving phenomenon, and that the exact proportions of presymptomatic and never-symptomatic transmission and the incubation periods are not known. They also note symptoms and transmissions appear to vary across different population groups, with older individuals more likely than younger persons to experience symptoms, according to previous studies.
“Assume that everyone is potentially infected”
Other experts agree that expanded testing of asymptomatic individuals is important. “Screening for fever and isolation of symptomatic individuals is a common-sense approach to help prevent spread, but these measures are by no means adequate since it’s been clearly documented that individuals who are either asymptomatic or presymptomatic can still spread the virus,” said Brett Williams, MD, an infectious disease specialist and assistant professor of medicine at Rush University in Chicago.
“As we saw with the White House Rose Garden superspreader outbreak, testing does not reliably exclude infection either because the tested individual has not yet become positive or the test is falsely negative,” Dr. Williams, who was not involved in the CDC study, said in an interview. He further noted that when prevalence is as high as it currently is in the United States, the rate of false negatives will be high because a large proportion of those screened will be unknowingly infected.
At his center, all visitors and staff are screened with a temperature probe on entry, and since the earliest days of the pandemic, universal masking has been required. “Nationally there have been many instances of hospital break room outbreaks because of staff eating lunch together, and these outbreaks also demonstrate the incompleteness of symptomatic isolation,” Dr. Williams said.
For his part, virologist Frank Esper, MD, a pediatric infectious disease specialist at the Cleveland Clinic, said that while it’s been understood for some time that many infected people will not exhibit symptoms, “the question that remains is just how infectious are they?”
Dr. Esper’s takeaway from the modeling study is not so much that we need more screening of possibly exposed but asymptomatic people, but rather testing symptomatic people and tracing their contacts is not enough.
“We need to continue to assume that everyone is potentially infected whether they know it or not. And even though we have ramped up our testing to a much greater capacity than in the first wave, we need to continue to wear masks and socially distance because just identifying people who are sick and isolating or quarantining them is not going to be enough to contain the pandemic.”
And although assumption-based modeling is helpful, it cannot tell us “how many asymptomatic people are actually infected,” said Dr. Esper, who was not involved in the CDC study.
Dr. Esper also pointed out that the study estimates are based on data from early Chinese studies, but the virus has since changed. The new, more transmissible strain in the United States and elsewhere may involve not only more infections but also a longer presymptomatic stage. “So the CDC study may actually undershoot asymptomatic infections,” he said.
He also agreed with the authors that when it comes to infection, not all humans are equal. “Older people tend to be more symptomatic and become symptomatic more quickly so the asymptomatic rate is not the same across board from young people age 20 to older people.”
The bottom line, said David. A. Hirschwerk, MD, an infectious disease specialist at Northwell Health in Manhasset, N.Y., is that these data support the maintenance of protective measures we’ve been taking over the past months. “They support the concept that asymptomatic people are a significant source of transmission and that we need to adhere to mask wearing and social distancing, particularly indoors,” Dr. Hirschwerk, who was not involved in the analysis, said in an interview. “More testing would be better but it has to be fast and it has to be efficient, and there are a lot of challenges to overcome.”
The study was done as part of the CDC’s coronavirus disease 2019 response and was supported solely by federal base and response funding. The authors and commentators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The importance of community pediatric hospital medicine
According to data from the American Academy of Pediatrics, over 2,000 physicians – or approximately 70% of all physicians practicing pediatric hospital medicine – do so in a community hospital. Like all areas of hospital medicine, community pediatric hospital medicine (CPHM) strives to fulfill one of our field’s central tenets – providing high-quality, evidence-based care to our patients.
A phrase often used among CPHM practitioners is that, “if you’ve seen one CPHM program, you’ve seen one CPHM program.” Every CPHM program is different. While this phrase may seem rather simplistic, it quite accurately portrays a unique aspect of our place in the hospital medicine field. CPHM programs usually require their practitioners to perform a broader range of roles and responsibilities than our colleagues who practice in university or children’s hospitals. Typically, these roles are aligned with the unique needs of each hospital within which we practice and the communities we serve. Factors such as the distance to a tertiary care referral center, access to subspecialists, availability and expertise of ancillary services for children, and the particular needs of each community further shape the role that CPHM practitioners may be asked to play.
In 2014, the AAP section on hospital medicine’s subcommittee on community hospitalists surveyed all CPHM programs to understand the unique roles that practitioners play within their institutions. Under the leadership of Clota Snow, MD, and Jacques Corriveau, MD, the aim was to contact every hospital in the country using the American Hospital Directory to see if they had a PHM program and to identify what roles the program was responsible for within their hospital.
Of the 535 programs identified, the primary responsibilities included inpatient care (85%), ED consultations (76%) and newborn nursery care (73%). Other common roles not typically associated with a university-based hospitalist’s responsibilities included delivery room attendance/neonatal resuscitations (44%), neonatal ICU management (47%) and subspecialty or surgical comanagement (52%). In some communities, even pediatric ICU management, sedation, and patient transport are part of our role. Because of the large breadth of roles that a CPHM practitioner may cover, we have often been referred to as “pediatric hospital-based generalists.”
Ideally, the presence of a pediatric hospitalist in a community hospital allows children to obtain high-quality, evidence-based care within their home communities. Most hospitalized children do not require direct access to subspecialists or all the pediatric-specific resources only available within a university or children’s hospital. Thus, if these resources are not required for the child’s care, CPHM practitioners can provide the care that a child needs in a setting that is less disruptive to the family and typically more cost effective.
CPHM physicians are often drawn to a career in a community hospital because it allows them to use their entire skill set to care for children with a wide variety of conditions. As they are often the only physicians in an adult hospital with a full understanding of the unique aspects of care that children require, it is important that they be comfortable in their role of managing the majority of pediatric care independently. Yet they also need to understand the limitations of their own ability, as well as their institution’s level of expertise in pediatric-specific care. They must be confident and vocal advocates for pediatric-specific needs throughout their institution and its numerous committees, and form close working relationships with colleagues and administrators in the different fields with whom we share care of our patients (e.g., ED, obstetrics, radiology, trauma, and other medical and surgical subspecialties).
CPHM physicians are particularly well suited to partner with local outpatient providers as well as tertiary care physicians to provide coordinated transitions between the inpatient and outpatient management of a child’s illness. In addition, a CPHM physician can often bring a unique and valuable perspective of the particular ethnic, cultural, and socioeconomic diversity of their community, as well as its available resources, to facilitate a greater level of engagement with the child’s needs and ultimate success of their care.
The 2014 survey of CPHM programs identified several major challenges to recruitment and career satisfaction as a CPHM physician. These include a lack of access to subspecialists, a lack of pediatric-specific ancillary services and the perception that our importance as community hospital providers was not valued as much in the PHM community as PHM physicians working in a university/children’s hospital setting. With the recent recognition of PHM as an official subspecialty by the American Board of Pediatrics, the concern has intensified within our field that a two-tiered system will develop with some PHM physicians being board certified and others not.
While the development of board subspecialization was not meant to limit the pool of providers available to staff community hospital sites, there is nowhere near the number of fellowship trained physicians to provide an adequate workforce to staff CPHM programs. This means that many CPHM physicians will not be board certified in pediatric hospital medicine but does not mean that CPHM programs will be unable to provide high-quality local care that benefits children and their families, including safe care for children who require the skills that an immediately available CPHM physician can provide.
Many pediatric residency programs do not currently provide their trainees with exposure to community hospital medicine. Further, with increased sub-specialization throughout pediatrics, fewer residents are developing the necessary skill set to perform roles integral to a caring for children in community hospitals such as stabilization of a critically ill child prior to transport and complex neonatal resuscitation.
A career in CPHM provides physicians with the opportunity to work together with a close-knit group to provide exceptional care to children and to advocate for the medical needs of children in their hospital and their community. The AAP’s subcommittee has made it a priority to engage physicians during all parts of their pediatric training about why a career in CPHM is exciting, fulfilling and a great life, as well as continuing to educate training programs at every level – as well as the larger PHM community – about why CPHM is a valuable and important part of pediatric medicine.
Dr. Welsh is a clinical associate professor of pediatrics at the Stanford (Calif.) University in the division of pediatric hospital medicine. He has practiced community pediatric hospital medicine for over 27 years in Washington state and the San Francisco Bay Area. He is the chair of the working group of the Future of Community Pediatric Hospital Medicine for the AAP section on hospital medicine’s subcommittee on community hospitalists.
According to data from the American Academy of Pediatrics, over 2,000 physicians – or approximately 70% of all physicians practicing pediatric hospital medicine – do so in a community hospital. Like all areas of hospital medicine, community pediatric hospital medicine (CPHM) strives to fulfill one of our field’s central tenets – providing high-quality, evidence-based care to our patients.
A phrase often used among CPHM practitioners is that, “if you’ve seen one CPHM program, you’ve seen one CPHM program.” Every CPHM program is different. While this phrase may seem rather simplistic, it quite accurately portrays a unique aspect of our place in the hospital medicine field. CPHM programs usually require their practitioners to perform a broader range of roles and responsibilities than our colleagues who practice in university or children’s hospitals. Typically, these roles are aligned with the unique needs of each hospital within which we practice and the communities we serve. Factors such as the distance to a tertiary care referral center, access to subspecialists, availability and expertise of ancillary services for children, and the particular needs of each community further shape the role that CPHM practitioners may be asked to play.
In 2014, the AAP section on hospital medicine’s subcommittee on community hospitalists surveyed all CPHM programs to understand the unique roles that practitioners play within their institutions. Under the leadership of Clota Snow, MD, and Jacques Corriveau, MD, the aim was to contact every hospital in the country using the American Hospital Directory to see if they had a PHM program and to identify what roles the program was responsible for within their hospital.
Of the 535 programs identified, the primary responsibilities included inpatient care (85%), ED consultations (76%) and newborn nursery care (73%). Other common roles not typically associated with a university-based hospitalist’s responsibilities included delivery room attendance/neonatal resuscitations (44%), neonatal ICU management (47%) and subspecialty or surgical comanagement (52%). In some communities, even pediatric ICU management, sedation, and patient transport are part of our role. Because of the large breadth of roles that a CPHM practitioner may cover, we have often been referred to as “pediatric hospital-based generalists.”
Ideally, the presence of a pediatric hospitalist in a community hospital allows children to obtain high-quality, evidence-based care within their home communities. Most hospitalized children do not require direct access to subspecialists or all the pediatric-specific resources only available within a university or children’s hospital. Thus, if these resources are not required for the child’s care, CPHM practitioners can provide the care that a child needs in a setting that is less disruptive to the family and typically more cost effective.
CPHM physicians are often drawn to a career in a community hospital because it allows them to use their entire skill set to care for children with a wide variety of conditions. As they are often the only physicians in an adult hospital with a full understanding of the unique aspects of care that children require, it is important that they be comfortable in their role of managing the majority of pediatric care independently. Yet they also need to understand the limitations of their own ability, as well as their institution’s level of expertise in pediatric-specific care. They must be confident and vocal advocates for pediatric-specific needs throughout their institution and its numerous committees, and form close working relationships with colleagues and administrators in the different fields with whom we share care of our patients (e.g., ED, obstetrics, radiology, trauma, and other medical and surgical subspecialties).
CPHM physicians are particularly well suited to partner with local outpatient providers as well as tertiary care physicians to provide coordinated transitions between the inpatient and outpatient management of a child’s illness. In addition, a CPHM physician can often bring a unique and valuable perspective of the particular ethnic, cultural, and socioeconomic diversity of their community, as well as its available resources, to facilitate a greater level of engagement with the child’s needs and ultimate success of their care.
The 2014 survey of CPHM programs identified several major challenges to recruitment and career satisfaction as a CPHM physician. These include a lack of access to subspecialists, a lack of pediatric-specific ancillary services and the perception that our importance as community hospital providers was not valued as much in the PHM community as PHM physicians working in a university/children’s hospital setting. With the recent recognition of PHM as an official subspecialty by the American Board of Pediatrics, the concern has intensified within our field that a two-tiered system will develop with some PHM physicians being board certified and others not.
While the development of board subspecialization was not meant to limit the pool of providers available to staff community hospital sites, there is nowhere near the number of fellowship trained physicians to provide an adequate workforce to staff CPHM programs. This means that many CPHM physicians will not be board certified in pediatric hospital medicine but does not mean that CPHM programs will be unable to provide high-quality local care that benefits children and their families, including safe care for children who require the skills that an immediately available CPHM physician can provide.
Many pediatric residency programs do not currently provide their trainees with exposure to community hospital medicine. Further, with increased sub-specialization throughout pediatrics, fewer residents are developing the necessary skill set to perform roles integral to a caring for children in community hospitals such as stabilization of a critically ill child prior to transport and complex neonatal resuscitation.
A career in CPHM provides physicians with the opportunity to work together with a close-knit group to provide exceptional care to children and to advocate for the medical needs of children in their hospital and their community. The AAP’s subcommittee has made it a priority to engage physicians during all parts of their pediatric training about why a career in CPHM is exciting, fulfilling and a great life, as well as continuing to educate training programs at every level – as well as the larger PHM community – about why CPHM is a valuable and important part of pediatric medicine.
Dr. Welsh is a clinical associate professor of pediatrics at the Stanford (Calif.) University in the division of pediatric hospital medicine. He has practiced community pediatric hospital medicine for over 27 years in Washington state and the San Francisco Bay Area. He is the chair of the working group of the Future of Community Pediatric Hospital Medicine for the AAP section on hospital medicine’s subcommittee on community hospitalists.
According to data from the American Academy of Pediatrics, over 2,000 physicians – or approximately 70% of all physicians practicing pediatric hospital medicine – do so in a community hospital. Like all areas of hospital medicine, community pediatric hospital medicine (CPHM) strives to fulfill one of our field’s central tenets – providing high-quality, evidence-based care to our patients.
A phrase often used among CPHM practitioners is that, “if you’ve seen one CPHM program, you’ve seen one CPHM program.” Every CPHM program is different. While this phrase may seem rather simplistic, it quite accurately portrays a unique aspect of our place in the hospital medicine field. CPHM programs usually require their practitioners to perform a broader range of roles and responsibilities than our colleagues who practice in university or children’s hospitals. Typically, these roles are aligned with the unique needs of each hospital within which we practice and the communities we serve. Factors such as the distance to a tertiary care referral center, access to subspecialists, availability and expertise of ancillary services for children, and the particular needs of each community further shape the role that CPHM practitioners may be asked to play.
In 2014, the AAP section on hospital medicine’s subcommittee on community hospitalists surveyed all CPHM programs to understand the unique roles that practitioners play within their institutions. Under the leadership of Clota Snow, MD, and Jacques Corriveau, MD, the aim was to contact every hospital in the country using the American Hospital Directory to see if they had a PHM program and to identify what roles the program was responsible for within their hospital.
Of the 535 programs identified, the primary responsibilities included inpatient care (85%), ED consultations (76%) and newborn nursery care (73%). Other common roles not typically associated with a university-based hospitalist’s responsibilities included delivery room attendance/neonatal resuscitations (44%), neonatal ICU management (47%) and subspecialty or surgical comanagement (52%). In some communities, even pediatric ICU management, sedation, and patient transport are part of our role. Because of the large breadth of roles that a CPHM practitioner may cover, we have often been referred to as “pediatric hospital-based generalists.”
Ideally, the presence of a pediatric hospitalist in a community hospital allows children to obtain high-quality, evidence-based care within their home communities. Most hospitalized children do not require direct access to subspecialists or all the pediatric-specific resources only available within a university or children’s hospital. Thus, if these resources are not required for the child’s care, CPHM practitioners can provide the care that a child needs in a setting that is less disruptive to the family and typically more cost effective.
CPHM physicians are often drawn to a career in a community hospital because it allows them to use their entire skill set to care for children with a wide variety of conditions. As they are often the only physicians in an adult hospital with a full understanding of the unique aspects of care that children require, it is important that they be comfortable in their role of managing the majority of pediatric care independently. Yet they also need to understand the limitations of their own ability, as well as their institution’s level of expertise in pediatric-specific care. They must be confident and vocal advocates for pediatric-specific needs throughout their institution and its numerous committees, and form close working relationships with colleagues and administrators in the different fields with whom we share care of our patients (e.g., ED, obstetrics, radiology, trauma, and other medical and surgical subspecialties).
CPHM physicians are particularly well suited to partner with local outpatient providers as well as tertiary care physicians to provide coordinated transitions between the inpatient and outpatient management of a child’s illness. In addition, a CPHM physician can often bring a unique and valuable perspective of the particular ethnic, cultural, and socioeconomic diversity of their community, as well as its available resources, to facilitate a greater level of engagement with the child’s needs and ultimate success of their care.
The 2014 survey of CPHM programs identified several major challenges to recruitment and career satisfaction as a CPHM physician. These include a lack of access to subspecialists, a lack of pediatric-specific ancillary services and the perception that our importance as community hospital providers was not valued as much in the PHM community as PHM physicians working in a university/children’s hospital setting. With the recent recognition of PHM as an official subspecialty by the American Board of Pediatrics, the concern has intensified within our field that a two-tiered system will develop with some PHM physicians being board certified and others not.
While the development of board subspecialization was not meant to limit the pool of providers available to staff community hospital sites, there is nowhere near the number of fellowship trained physicians to provide an adequate workforce to staff CPHM programs. This means that many CPHM physicians will not be board certified in pediatric hospital medicine but does not mean that CPHM programs will be unable to provide high-quality local care that benefits children and their families, including safe care for children who require the skills that an immediately available CPHM physician can provide.
Many pediatric residency programs do not currently provide their trainees with exposure to community hospital medicine. Further, with increased sub-specialization throughout pediatrics, fewer residents are developing the necessary skill set to perform roles integral to a caring for children in community hospitals such as stabilization of a critically ill child prior to transport and complex neonatal resuscitation.
A career in CPHM provides physicians with the opportunity to work together with a close-knit group to provide exceptional care to children and to advocate for the medical needs of children in their hospital and their community. The AAP’s subcommittee has made it a priority to engage physicians during all parts of their pediatric training about why a career in CPHM is exciting, fulfilling and a great life, as well as continuing to educate training programs at every level – as well as the larger PHM community – about why CPHM is a valuable and important part of pediatric medicine.
Dr. Welsh is a clinical associate professor of pediatrics at the Stanford (Calif.) University in the division of pediatric hospital medicine. He has practiced community pediatric hospital medicine for over 27 years in Washington state and the San Francisco Bay Area. He is the chair of the working group of the Future of Community Pediatric Hospital Medicine for the AAP section on hospital medicine’s subcommittee on community hospitalists.
Anaphylaxis cases after COVID-19 vaccine rising but still rare: CDC
Health care providers should be ready to treat rare cases of anaphylaxis following administration of COVID-19 vaccines, federal medical officials have urged. The officials also stressed the importance of continuing vaccinations, despite reports of the rare side effect.
There have been 29 cases of anaphylaxis to date following administration of a COVID-19 vaccine, officials from the Centers for Disease Control and Prevention said in a call with reporters on Jan. 6.
The severe allergic reaction, which appears to be rare, can happen with either the Pfizer-BioNTech vaccine or the rival Moderna product. The Food and Drug Administration granted emergency use authorizations for these two vaccines in December.
Even with the cases seen to date, the COVID-19 vaccines remain a “good value proposition,” Nancy Messonnier, MD, director of the CDC’s National Center for Immunization, said in the call.
There have been about 11.1 cases of anaphylaxis per million doses with the Pfizer-BioNTech COVID-19 vaccine, which is higher than the estimated 1.3 cases per million doses with influenza vaccines, she said. But the low risk of anaphylaxis must be balanced against the threat of COVID-19, which currently claims about 2,000 lives a day in the United States, she said. In addition, many people are reporting long-term complications with COVID-19 even if they recover.
Kept in context, the data on anaphylaxis should not scare people away from getting a COVID-19 vaccine, she added.
“Their risk from COVID and poor outcomes is still more than the risk of a severe outcome from the vaccine,” Dr. Messonnier said. “And fortunately, we know how to treat anaphylaxis.”
Dr. Messonnier urged health care workers administering COVID-19 vaccines to be prepared.
“Anybody administering vaccines needs not just to have the EpiPen available, but frankly, to know how to use it,” Dr. Messonnier said.
MMWR details
The CDC on Jan. 6 also provided an update on anaphylaxis in Morbidity and Mortality Weekly Report (MMWR).
The information included in the report was based on cases reported with the Pfizer-BioNTech vaccine – the first to get emergency use authorization from the FDA. On the call with reporters, CDC officials confirmed there have been additional reports since then and anaphylaxis has been reported with both the Pfizer-BioNTech and Moderna vaccines. CDC officials said they could not give a breakdown of how many cases were linked to each of these products at this time.
Between Dec. 14 and 23, 2020, monitoring by the Vaccine Adverse Event Reporting System detected 21 cases of anaphylaxis after administration of a reported 1,893,360 first doses of the Pfizer-BioNTech COVID-19 vaccine. Most reactions – 71% – occurred within 15 minutes of vaccination.
A version of this article first appeared on Medscape.com.
Health care providers should be ready to treat rare cases of anaphylaxis following administration of COVID-19 vaccines, federal medical officials have urged. The officials also stressed the importance of continuing vaccinations, despite reports of the rare side effect.
There have been 29 cases of anaphylaxis to date following administration of a COVID-19 vaccine, officials from the Centers for Disease Control and Prevention said in a call with reporters on Jan. 6.
The severe allergic reaction, which appears to be rare, can happen with either the Pfizer-BioNTech vaccine or the rival Moderna product. The Food and Drug Administration granted emergency use authorizations for these two vaccines in December.
Even with the cases seen to date, the COVID-19 vaccines remain a “good value proposition,” Nancy Messonnier, MD, director of the CDC’s National Center for Immunization, said in the call.
There have been about 11.1 cases of anaphylaxis per million doses with the Pfizer-BioNTech COVID-19 vaccine, which is higher than the estimated 1.3 cases per million doses with influenza vaccines, she said. But the low risk of anaphylaxis must be balanced against the threat of COVID-19, which currently claims about 2,000 lives a day in the United States, she said. In addition, many people are reporting long-term complications with COVID-19 even if they recover.
Kept in context, the data on anaphylaxis should not scare people away from getting a COVID-19 vaccine, she added.
“Their risk from COVID and poor outcomes is still more than the risk of a severe outcome from the vaccine,” Dr. Messonnier said. “And fortunately, we know how to treat anaphylaxis.”
Dr. Messonnier urged health care workers administering COVID-19 vaccines to be prepared.
“Anybody administering vaccines needs not just to have the EpiPen available, but frankly, to know how to use it,” Dr. Messonnier said.
MMWR details
The CDC on Jan. 6 also provided an update on anaphylaxis in Morbidity and Mortality Weekly Report (MMWR).
The information included in the report was based on cases reported with the Pfizer-BioNTech vaccine – the first to get emergency use authorization from the FDA. On the call with reporters, CDC officials confirmed there have been additional reports since then and anaphylaxis has been reported with both the Pfizer-BioNTech and Moderna vaccines. CDC officials said they could not give a breakdown of how many cases were linked to each of these products at this time.
Between Dec. 14 and 23, 2020, monitoring by the Vaccine Adverse Event Reporting System detected 21 cases of anaphylaxis after administration of a reported 1,893,360 first doses of the Pfizer-BioNTech COVID-19 vaccine. Most reactions – 71% – occurred within 15 minutes of vaccination.
A version of this article first appeared on Medscape.com.
Health care providers should be ready to treat rare cases of anaphylaxis following administration of COVID-19 vaccines, federal medical officials have urged. The officials also stressed the importance of continuing vaccinations, despite reports of the rare side effect.
There have been 29 cases of anaphylaxis to date following administration of a COVID-19 vaccine, officials from the Centers for Disease Control and Prevention said in a call with reporters on Jan. 6.
The severe allergic reaction, which appears to be rare, can happen with either the Pfizer-BioNTech vaccine or the rival Moderna product. The Food and Drug Administration granted emergency use authorizations for these two vaccines in December.
Even with the cases seen to date, the COVID-19 vaccines remain a “good value proposition,” Nancy Messonnier, MD, director of the CDC’s National Center for Immunization, said in the call.
There have been about 11.1 cases of anaphylaxis per million doses with the Pfizer-BioNTech COVID-19 vaccine, which is higher than the estimated 1.3 cases per million doses with influenza vaccines, she said. But the low risk of anaphylaxis must be balanced against the threat of COVID-19, which currently claims about 2,000 lives a day in the United States, she said. In addition, many people are reporting long-term complications with COVID-19 even if they recover.
Kept in context, the data on anaphylaxis should not scare people away from getting a COVID-19 vaccine, she added.
“Their risk from COVID and poor outcomes is still more than the risk of a severe outcome from the vaccine,” Dr. Messonnier said. “And fortunately, we know how to treat anaphylaxis.”
Dr. Messonnier urged health care workers administering COVID-19 vaccines to be prepared.
“Anybody administering vaccines needs not just to have the EpiPen available, but frankly, to know how to use it,” Dr. Messonnier said.
MMWR details
The CDC on Jan. 6 also provided an update on anaphylaxis in Morbidity and Mortality Weekly Report (MMWR).
The information included in the report was based on cases reported with the Pfizer-BioNTech vaccine – the first to get emergency use authorization from the FDA. On the call with reporters, CDC officials confirmed there have been additional reports since then and anaphylaxis has been reported with both the Pfizer-BioNTech and Moderna vaccines. CDC officials said they could not give a breakdown of how many cases were linked to each of these products at this time.
Between Dec. 14 and 23, 2020, monitoring by the Vaccine Adverse Event Reporting System detected 21 cases of anaphylaxis after administration of a reported 1,893,360 first doses of the Pfizer-BioNTech COVID-19 vaccine. Most reactions – 71% – occurred within 15 minutes of vaccination.
A version of this article first appeared on Medscape.com.
Children’s hospitals grapple with wave of mental illness
Krissy Williams, 15, had attempted suicide before, but never with pills.
The teen was diagnosed with schizophrenia when she was 9. People with this chronic mental health condition perceive reality differently and often experience hallucinations and delusions. She learned to manage these symptoms with a variety of services offered at home and at school.
But the pandemic upended those lifelines. She lost much of the support offered at school. She also lost regular contact with her peers. Her mother lost access to respite care – which allowed her to take a break.
On a Thursday in October, the isolation and sadness came to a head. As Krissy’s mother, Patricia Williams, called a mental crisis hotline for help, she said, Krissy stood on the deck of their Maryland home with a bottle of pain medication in one hand and water in the other.
Before Patricia could react, Krissy placed the pills in her mouth and swallowed.
Efforts to contain the spread of the novel coronavirus in the United States have led to drastic changes in the way children and teens learn, play and socialize. Tens of millions of students are attending school through some form of distance learning. Many extracurricular activities have been canceled. Playgrounds, zoos, and other recreational spaces have closed. Kids like Krissy have struggled to cope and the toll is becoming evident.
Government figures show the proportion of children who arrived in EDs with mental health issues increased 24% from mid-March through mid-October, compared with the same period in 2019. Among preteens and adolescents, it rose by 31%. Anecdotally, some hospitals said they are seeing more cases of severe depression and suicidal thoughts among children, particularly attempts to overdose.
The increased demand for intensive mental health care that has accompanied the pandemic has worsened issues that have long plagued the system. In some hospitals, the number of children unable to immediately get a bed in the psychiatric unit rose. Others reduced the number of beds or closed psychiatric units altogether to reduce the spread of COVID-19.
“It’s only a matter of time before a tsunami sort of reaches the shore of our service system, and it’s going to be overwhelmed with the mental health needs of kids,” said Jason Williams, PsyD, a psychologist and director of operations of the Pediatric Mental Health Institute at Children’s Hospital Colorado, Aurora.
“I think we’re just starting to see the tip of the iceberg, to be honest with you.”
Before COVID, more than 8 million kids between ages 3 and 17 were diagnosed with a mental or behavioral health condition, according to the most recent National Survey of Children’s Health. A separate survey from the Centers for Disease Control and Prevention found one in three high school students in 2019 reported feeling persistently sad and hopeless – a 40% increase from 2009.
The coronavirus pandemic appears to be adding to these difficulties. A review of 80 studies found forced isolation and loneliness among children correlated with an increased risk of depression.
“We’re all social beings, but they’re [teenagers] at the point in their development where their peers are their reality,” said Terrie Andrews, PhD, a psychologist and administrator of behavioral health at Wolfson Children’s Hospital in Jacksonville, Fla. “Their peers are their grounding mechanism.”
Children’s hospitals in Colorado, Missouri, and New York all reported an uptick in the number of patients who thought about or attempted suicide. Clinicians also mentioned spikes in children with severe depression and those with autism who are acting out.
The number of overdose attempts among children has caught the attention of clinicians at two facilities. Dr. Andrews said the facility gives out lockboxes for weapons and medication to the public – including parents who come in after children attempted to take their life using medication.
Children’s National Hospital in Washington, D.C., also has experienced an uptick, said Colby Tyson, MD, associate director of inpatient psychiatry. She’s seen children’s mental health deteriorate because of a likely increase in family conflict – often a consequence of the chaos caused by the pandemic. Without school, connections with peers or employment, families don’t have the opportunity to spend time away from one another and regroup, which can add stress to an already tense situation.
“That break is gone,” she said.
The higher demand for child mental health services caused by the pandemic has made finding a bed at an inpatient unit more difficult.
Now, some hospitals report running at full capacity and having more children “boarding,” or sleeping in EDs before being admitted to the psychiatric unit. Among them is the Pediatric Mental Health Institute at Children’s Hospital Colorado. Williams said the inpatient unit has been full since March. Some children now wait nearly 2 days for a bed, up from the 8-10 hours common before the pandemic.
Cincinnati Children’s Hospital Medical Center in Ohio is also running at full capacity, said clinicians, and had several days in which the unit was above capacity and placed kids instead in the emergency department waiting to be admitted. In Florida, Dr. Andrews said, up to 25 children have been held on surgical floors at Wolfson Children’s while waiting for a spot to open in the inpatient psychiatric unit. Their wait could last as long as 5 days, she said.
Multiple hospitals said the usual summer slump in child psychiatric admissions was missing last year. “We never saw that during the pandemic,” said Andrews. “We stayed completely busy the entire time.”
Some facilities have decided to reduce the number of beds available to maintain physical distancing, further constricting supply. Children’s National in D.C. cut five beds from its unit to maintain single occupancy in every room, said Adelaide Robb, MD, division chief of psychiatry and behavioral sciences.
The measures taken to curb the spread of COVID have also affected the way hospitalized children receive mental health services. In addition to providers wearing protective equipment, some hospitals like Cincinnati Children’s rearranged furniture and placed cues on the floor as reminders to stay 6 feet apart. The University of Pittsburgh Medical Center’s Western Psychiatric Hospital and other facilities encourage children to keep their masks on by offering rewards like extra computer time. Patients at Children’s National now eat in their rooms, a change from when they ate together.
Despite the need for distance, social interaction still represents an important part of mental health care for children, clinicians said. Facilities have come up with various ways to do so safely, including creating smaller pods for group therapy. Children at Cincinnati Children’s Hospital can play with toys, but only with ones that can be wiped clean afterward. No cards or board games, said Suzanne Sampang, MD, clinical medical director for child and adolescent psychiatry at the hospital.
“I think what’s different about psychiatric treatment is that, really, interaction is the treatment,” she said, “just as much as a medication.”
The added infection-control precautions pose challenges to forging therapeutic connections. Masks can complicate the ability to read a person’s face. Online meetings make it difficult to build trust between a patient and a therapist.
“There’s something about the real relationship in person that the best technology can’t give to you,” said Dr. Robb.
For now, Krissy Williams is relying on virtual platforms to receive some of her mental health services. Despite being hospitalized and suffering brain damage due to the overdose, she is now at home and in good spirits. She enjoys geometry, dancing on TikTok, and trying to beat her mother at Super Mario Bros. on the Wii. But being away from her friends, she said, has been a hard adjustment.
“When you’re used to something,” she said, “it’s not easy to change everything.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Krissy Williams, 15, had attempted suicide before, but never with pills.
The teen was diagnosed with schizophrenia when she was 9. People with this chronic mental health condition perceive reality differently and often experience hallucinations and delusions. She learned to manage these symptoms with a variety of services offered at home and at school.
But the pandemic upended those lifelines. She lost much of the support offered at school. She also lost regular contact with her peers. Her mother lost access to respite care – which allowed her to take a break.
On a Thursday in October, the isolation and sadness came to a head. As Krissy’s mother, Patricia Williams, called a mental crisis hotline for help, she said, Krissy stood on the deck of their Maryland home with a bottle of pain medication in one hand and water in the other.
Before Patricia could react, Krissy placed the pills in her mouth and swallowed.
Efforts to contain the spread of the novel coronavirus in the United States have led to drastic changes in the way children and teens learn, play and socialize. Tens of millions of students are attending school through some form of distance learning. Many extracurricular activities have been canceled. Playgrounds, zoos, and other recreational spaces have closed. Kids like Krissy have struggled to cope and the toll is becoming evident.
Government figures show the proportion of children who arrived in EDs with mental health issues increased 24% from mid-March through mid-October, compared with the same period in 2019. Among preteens and adolescents, it rose by 31%. Anecdotally, some hospitals said they are seeing more cases of severe depression and suicidal thoughts among children, particularly attempts to overdose.
The increased demand for intensive mental health care that has accompanied the pandemic has worsened issues that have long plagued the system. In some hospitals, the number of children unable to immediately get a bed in the psychiatric unit rose. Others reduced the number of beds or closed psychiatric units altogether to reduce the spread of COVID-19.
“It’s only a matter of time before a tsunami sort of reaches the shore of our service system, and it’s going to be overwhelmed with the mental health needs of kids,” said Jason Williams, PsyD, a psychologist and director of operations of the Pediatric Mental Health Institute at Children’s Hospital Colorado, Aurora.
“I think we’re just starting to see the tip of the iceberg, to be honest with you.”
Before COVID, more than 8 million kids between ages 3 and 17 were diagnosed with a mental or behavioral health condition, according to the most recent National Survey of Children’s Health. A separate survey from the Centers for Disease Control and Prevention found one in three high school students in 2019 reported feeling persistently sad and hopeless – a 40% increase from 2009.
The coronavirus pandemic appears to be adding to these difficulties. A review of 80 studies found forced isolation and loneliness among children correlated with an increased risk of depression.
“We’re all social beings, but they’re [teenagers] at the point in their development where their peers are their reality,” said Terrie Andrews, PhD, a psychologist and administrator of behavioral health at Wolfson Children’s Hospital in Jacksonville, Fla. “Their peers are their grounding mechanism.”
Children’s hospitals in Colorado, Missouri, and New York all reported an uptick in the number of patients who thought about or attempted suicide. Clinicians also mentioned spikes in children with severe depression and those with autism who are acting out.
The number of overdose attempts among children has caught the attention of clinicians at two facilities. Dr. Andrews said the facility gives out lockboxes for weapons and medication to the public – including parents who come in after children attempted to take their life using medication.
Children’s National Hospital in Washington, D.C., also has experienced an uptick, said Colby Tyson, MD, associate director of inpatient psychiatry. She’s seen children’s mental health deteriorate because of a likely increase in family conflict – often a consequence of the chaos caused by the pandemic. Without school, connections with peers or employment, families don’t have the opportunity to spend time away from one another and regroup, which can add stress to an already tense situation.
“That break is gone,” she said.
The higher demand for child mental health services caused by the pandemic has made finding a bed at an inpatient unit more difficult.
Now, some hospitals report running at full capacity and having more children “boarding,” or sleeping in EDs before being admitted to the psychiatric unit. Among them is the Pediatric Mental Health Institute at Children’s Hospital Colorado. Williams said the inpatient unit has been full since March. Some children now wait nearly 2 days for a bed, up from the 8-10 hours common before the pandemic.
Cincinnati Children’s Hospital Medical Center in Ohio is also running at full capacity, said clinicians, and had several days in which the unit was above capacity and placed kids instead in the emergency department waiting to be admitted. In Florida, Dr. Andrews said, up to 25 children have been held on surgical floors at Wolfson Children’s while waiting for a spot to open in the inpatient psychiatric unit. Their wait could last as long as 5 days, she said.
Multiple hospitals said the usual summer slump in child psychiatric admissions was missing last year. “We never saw that during the pandemic,” said Andrews. “We stayed completely busy the entire time.”
Some facilities have decided to reduce the number of beds available to maintain physical distancing, further constricting supply. Children’s National in D.C. cut five beds from its unit to maintain single occupancy in every room, said Adelaide Robb, MD, division chief of psychiatry and behavioral sciences.
The measures taken to curb the spread of COVID have also affected the way hospitalized children receive mental health services. In addition to providers wearing protective equipment, some hospitals like Cincinnati Children’s rearranged furniture and placed cues on the floor as reminders to stay 6 feet apart. The University of Pittsburgh Medical Center’s Western Psychiatric Hospital and other facilities encourage children to keep their masks on by offering rewards like extra computer time. Patients at Children’s National now eat in their rooms, a change from when they ate together.
Despite the need for distance, social interaction still represents an important part of mental health care for children, clinicians said. Facilities have come up with various ways to do so safely, including creating smaller pods for group therapy. Children at Cincinnati Children’s Hospital can play with toys, but only with ones that can be wiped clean afterward. No cards or board games, said Suzanne Sampang, MD, clinical medical director for child and adolescent psychiatry at the hospital.
“I think what’s different about psychiatric treatment is that, really, interaction is the treatment,” she said, “just as much as a medication.”
The added infection-control precautions pose challenges to forging therapeutic connections. Masks can complicate the ability to read a person’s face. Online meetings make it difficult to build trust between a patient and a therapist.
“There’s something about the real relationship in person that the best technology can’t give to you,” said Dr. Robb.
For now, Krissy Williams is relying on virtual platforms to receive some of her mental health services. Despite being hospitalized and suffering brain damage due to the overdose, she is now at home and in good spirits. She enjoys geometry, dancing on TikTok, and trying to beat her mother at Super Mario Bros. on the Wii. But being away from her friends, she said, has been a hard adjustment.
“When you’re used to something,” she said, “it’s not easy to change everything.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Krissy Williams, 15, had attempted suicide before, but never with pills.
The teen was diagnosed with schizophrenia when she was 9. People with this chronic mental health condition perceive reality differently and often experience hallucinations and delusions. She learned to manage these symptoms with a variety of services offered at home and at school.
But the pandemic upended those lifelines. She lost much of the support offered at school. She also lost regular contact with her peers. Her mother lost access to respite care – which allowed her to take a break.
On a Thursday in October, the isolation and sadness came to a head. As Krissy’s mother, Patricia Williams, called a mental crisis hotline for help, she said, Krissy stood on the deck of their Maryland home with a bottle of pain medication in one hand and water in the other.
Before Patricia could react, Krissy placed the pills in her mouth and swallowed.
Efforts to contain the spread of the novel coronavirus in the United States have led to drastic changes in the way children and teens learn, play and socialize. Tens of millions of students are attending school through some form of distance learning. Many extracurricular activities have been canceled. Playgrounds, zoos, and other recreational spaces have closed. Kids like Krissy have struggled to cope and the toll is becoming evident.
Government figures show the proportion of children who arrived in EDs with mental health issues increased 24% from mid-March through mid-October, compared with the same period in 2019. Among preteens and adolescents, it rose by 31%. Anecdotally, some hospitals said they are seeing more cases of severe depression and suicidal thoughts among children, particularly attempts to overdose.
The increased demand for intensive mental health care that has accompanied the pandemic has worsened issues that have long plagued the system. In some hospitals, the number of children unable to immediately get a bed in the psychiatric unit rose. Others reduced the number of beds or closed psychiatric units altogether to reduce the spread of COVID-19.
“It’s only a matter of time before a tsunami sort of reaches the shore of our service system, and it’s going to be overwhelmed with the mental health needs of kids,” said Jason Williams, PsyD, a psychologist and director of operations of the Pediatric Mental Health Institute at Children’s Hospital Colorado, Aurora.
“I think we’re just starting to see the tip of the iceberg, to be honest with you.”
Before COVID, more than 8 million kids between ages 3 and 17 were diagnosed with a mental or behavioral health condition, according to the most recent National Survey of Children’s Health. A separate survey from the Centers for Disease Control and Prevention found one in three high school students in 2019 reported feeling persistently sad and hopeless – a 40% increase from 2009.
The coronavirus pandemic appears to be adding to these difficulties. A review of 80 studies found forced isolation and loneliness among children correlated with an increased risk of depression.
“We’re all social beings, but they’re [teenagers] at the point in their development where their peers are their reality,” said Terrie Andrews, PhD, a psychologist and administrator of behavioral health at Wolfson Children’s Hospital in Jacksonville, Fla. “Their peers are their grounding mechanism.”
Children’s hospitals in Colorado, Missouri, and New York all reported an uptick in the number of patients who thought about or attempted suicide. Clinicians also mentioned spikes in children with severe depression and those with autism who are acting out.
The number of overdose attempts among children has caught the attention of clinicians at two facilities. Dr. Andrews said the facility gives out lockboxes for weapons and medication to the public – including parents who come in after children attempted to take their life using medication.
Children’s National Hospital in Washington, D.C., also has experienced an uptick, said Colby Tyson, MD, associate director of inpatient psychiatry. She’s seen children’s mental health deteriorate because of a likely increase in family conflict – often a consequence of the chaos caused by the pandemic. Without school, connections with peers or employment, families don’t have the opportunity to spend time away from one another and regroup, which can add stress to an already tense situation.
“That break is gone,” she said.
The higher demand for child mental health services caused by the pandemic has made finding a bed at an inpatient unit more difficult.
Now, some hospitals report running at full capacity and having more children “boarding,” or sleeping in EDs before being admitted to the psychiatric unit. Among them is the Pediatric Mental Health Institute at Children’s Hospital Colorado. Williams said the inpatient unit has been full since March. Some children now wait nearly 2 days for a bed, up from the 8-10 hours common before the pandemic.
Cincinnati Children’s Hospital Medical Center in Ohio is also running at full capacity, said clinicians, and had several days in which the unit was above capacity and placed kids instead in the emergency department waiting to be admitted. In Florida, Dr. Andrews said, up to 25 children have been held on surgical floors at Wolfson Children’s while waiting for a spot to open in the inpatient psychiatric unit. Their wait could last as long as 5 days, she said.
Multiple hospitals said the usual summer slump in child psychiatric admissions was missing last year. “We never saw that during the pandemic,” said Andrews. “We stayed completely busy the entire time.”
Some facilities have decided to reduce the number of beds available to maintain physical distancing, further constricting supply. Children’s National in D.C. cut five beds from its unit to maintain single occupancy in every room, said Adelaide Robb, MD, division chief of psychiatry and behavioral sciences.
The measures taken to curb the spread of COVID have also affected the way hospitalized children receive mental health services. In addition to providers wearing protective equipment, some hospitals like Cincinnati Children’s rearranged furniture and placed cues on the floor as reminders to stay 6 feet apart. The University of Pittsburgh Medical Center’s Western Psychiatric Hospital and other facilities encourage children to keep their masks on by offering rewards like extra computer time. Patients at Children’s National now eat in their rooms, a change from when they ate together.
Despite the need for distance, social interaction still represents an important part of mental health care for children, clinicians said. Facilities have come up with various ways to do so safely, including creating smaller pods for group therapy. Children at Cincinnati Children’s Hospital can play with toys, but only with ones that can be wiped clean afterward. No cards or board games, said Suzanne Sampang, MD, clinical medical director for child and adolescent psychiatry at the hospital.
“I think what’s different about psychiatric treatment is that, really, interaction is the treatment,” she said, “just as much as a medication.”
The added infection-control precautions pose challenges to forging therapeutic connections. Masks can complicate the ability to read a person’s face. Online meetings make it difficult to build trust between a patient and a therapist.
“There’s something about the real relationship in person that the best technology can’t give to you,” said Dr. Robb.
For now, Krissy Williams is relying on virtual platforms to receive some of her mental health services. Despite being hospitalized and suffering brain damage due to the overdose, she is now at home and in good spirits. She enjoys geometry, dancing on TikTok, and trying to beat her mother at Super Mario Bros. on the Wii. But being away from her friends, she said, has been a hard adjustment.
“When you’re used to something,” she said, “it’s not easy to change everything.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Study confirms key COVID-19 risk factors in children
Children and adolescents who receive positive COVID-19 test results are not only more likely to have been in close contact with someone with a confirmed case of the virus but also are less likely to have reported consistent mask use among students and staff inside the school they attended, reported Charlotte V. Hobbs, MD, and colleagues at the University of Mississippi, Jackson.
In partnership with the Centers for Disease Control and Prevention’s COVID-19 Response Team, Dr. Hobbs and colleagues conducted a case-control study of 397 children and adolescents under 18 years of age to assess school, community, and close contact exposures associated with pediatric COVID-19. Patients tested for COVID-19 at outpatient health centers or emergency departments affiliated with the University of Mississippi Medical Center between Sept. 1 and Nov. 5, 2020, were included in the study.
Nearly two-thirds reported that exposure came from family members
Of the total study participants observed, 82 (21%) were under 4 years of age; 214 (54%) were female; 217 (55%) were non-Hispanic black, and 145 (37%) were non-Hispanic white. More than half (53%) sought testing because of COVID-19 symptoms. Of those who tested positive, 66% reported having come into close contact with a COVID-19 case, and 64% reported that those contacts were family members, compared with 15% of contacts who were schoolmates and 27% who were child care classmates.
All participants completed in-person school or child care attendance less than 14 days before testing positive for the virus, including 62% of patients testing positive and 68% of those testing negative. The authors noted that school attendance itself was not found to be associated with any positive test results. In fact, parents in 64% of positive cases and 76% of negative cases reported mask wearing among children and staff inside places of learning.
Of those study participants testing positive who did come into close contact with someone with COVID-19, the contacts were more likely to be family members than school or child care classmates. Specifically, they were more likely, in the 2-week period preceding testing, to have attended gatherings with individuals outside their immediate households, including social events and activities with other children. Parents of students testing positive were also less likely to report consistent indoor mask use among their children older than 2 years and school staff members.
School attendance was not found to increase likelihood of testing positive
Attending in-person school or child care during the 2 weeks before the SARS-CoV-2 test was not associated with greater likelihood of testing positive, the study authors noted, adding that the majority of study respondents reported universal mask use inside school and child care facilities, consistent with Mississippi State Department of Health recommended guidelines.
Dr. Hobbs and colleagues reported at least four limitations of the study. They noted that the study participants may not be representative of youth in other geographic regions of the country. They considered the possibility of unmeasured confounding of participant behaviors that may not have been factored into the study. No attempt was made to verify parent claims of mask use at schools and child care programs. Lastly, they acknowledged that “case or control status might be subject to misclassification because of imperfect sensitivity or specificity of PCR-based testing.
As of Dec. 14, 2020, the CDC reported that 10.2% of all COVID-19 cases in the United States were in children and adolescents under the age of 18.
“Continued efforts to prevent transmission at schools and child care programs are important, as are assessments of various types of activities and exposures to identify risk factors for COVID-19 as children engage in classroom and social interactions.” Promoting behaviors to reduce exposures to the virus among youth in the household, the community, schools, and child care programs is important to preventing outbreaks of the virus at schools, the authors cautioned.
In a separate interview with this news organization, Karalyn Kinsella, MD, general pediatrician in a small group private practice in Cheshire, Conn., said, “What this report tells me is that COVID cases are more common when mask use is inconsistent in schools and at home and in schools that don’t properly adhere to CDC guidelines. Overall, so long as social distancing guidelines are followed, schools are pretty safe places for kids during this pandemic.”
This finding is important, since many families are keeping their children out of school over fears of contracting the virus, she added. Some of the consequences these children are suffering include a lack of social connection and structure, which in some cases is leading to worsening anxiety and depression, and for those with disabilities, such as those who receive physical therapy, occupational therapy, speech or have IEPs, they’re not getting the full benefit of the services that they would otherwise receive in person, she observed.
“I don’t think families really understand the risks of getting together with family or friends “in their bubble” or the risk of continuing sports participation. This is where the majority of COVID cases are coming from,” she said, adding that it is important to discuss this risk with them at appointments. So, when families ask us what we think of in-person learning, I think we should feel fairly confident that the benefit may outweigh the risk.”
Dr. Hobbs and colleagues, and Dr. Kinsella, had no conflicts of interest to report.
SOURCE: MMWR Morb Mortal Wkly Rep. 2020;69:1925-9. doi: 10.15585/mmwr.mm6950e3.
Children and adolescents who receive positive COVID-19 test results are not only more likely to have been in close contact with someone with a confirmed case of the virus but also are less likely to have reported consistent mask use among students and staff inside the school they attended, reported Charlotte V. Hobbs, MD, and colleagues at the University of Mississippi, Jackson.
In partnership with the Centers for Disease Control and Prevention’s COVID-19 Response Team, Dr. Hobbs and colleagues conducted a case-control study of 397 children and adolescents under 18 years of age to assess school, community, and close contact exposures associated with pediatric COVID-19. Patients tested for COVID-19 at outpatient health centers or emergency departments affiliated with the University of Mississippi Medical Center between Sept. 1 and Nov. 5, 2020, were included in the study.
Nearly two-thirds reported that exposure came from family members
Of the total study participants observed, 82 (21%) were under 4 years of age; 214 (54%) were female; 217 (55%) were non-Hispanic black, and 145 (37%) were non-Hispanic white. More than half (53%) sought testing because of COVID-19 symptoms. Of those who tested positive, 66% reported having come into close contact with a COVID-19 case, and 64% reported that those contacts were family members, compared with 15% of contacts who were schoolmates and 27% who were child care classmates.
All participants completed in-person school or child care attendance less than 14 days before testing positive for the virus, including 62% of patients testing positive and 68% of those testing negative. The authors noted that school attendance itself was not found to be associated with any positive test results. In fact, parents in 64% of positive cases and 76% of negative cases reported mask wearing among children and staff inside places of learning.
Of those study participants testing positive who did come into close contact with someone with COVID-19, the contacts were more likely to be family members than school or child care classmates. Specifically, they were more likely, in the 2-week period preceding testing, to have attended gatherings with individuals outside their immediate households, including social events and activities with other children. Parents of students testing positive were also less likely to report consistent indoor mask use among their children older than 2 years and school staff members.
School attendance was not found to increase likelihood of testing positive
Attending in-person school or child care during the 2 weeks before the SARS-CoV-2 test was not associated with greater likelihood of testing positive, the study authors noted, adding that the majority of study respondents reported universal mask use inside school and child care facilities, consistent with Mississippi State Department of Health recommended guidelines.
Dr. Hobbs and colleagues reported at least four limitations of the study. They noted that the study participants may not be representative of youth in other geographic regions of the country. They considered the possibility of unmeasured confounding of participant behaviors that may not have been factored into the study. No attempt was made to verify parent claims of mask use at schools and child care programs. Lastly, they acknowledged that “case or control status might be subject to misclassification because of imperfect sensitivity or specificity of PCR-based testing.
As of Dec. 14, 2020, the CDC reported that 10.2% of all COVID-19 cases in the United States were in children and adolescents under the age of 18.
“Continued efforts to prevent transmission at schools and child care programs are important, as are assessments of various types of activities and exposures to identify risk factors for COVID-19 as children engage in classroom and social interactions.” Promoting behaviors to reduce exposures to the virus among youth in the household, the community, schools, and child care programs is important to preventing outbreaks of the virus at schools, the authors cautioned.
In a separate interview with this news organization, Karalyn Kinsella, MD, general pediatrician in a small group private practice in Cheshire, Conn., said, “What this report tells me is that COVID cases are more common when mask use is inconsistent in schools and at home and in schools that don’t properly adhere to CDC guidelines. Overall, so long as social distancing guidelines are followed, schools are pretty safe places for kids during this pandemic.”
This finding is important, since many families are keeping their children out of school over fears of contracting the virus, she added. Some of the consequences these children are suffering include a lack of social connection and structure, which in some cases is leading to worsening anxiety and depression, and for those with disabilities, such as those who receive physical therapy, occupational therapy, speech or have IEPs, they’re not getting the full benefit of the services that they would otherwise receive in person, she observed.
“I don’t think families really understand the risks of getting together with family or friends “in their bubble” or the risk of continuing sports participation. This is where the majority of COVID cases are coming from,” she said, adding that it is important to discuss this risk with them at appointments. So, when families ask us what we think of in-person learning, I think we should feel fairly confident that the benefit may outweigh the risk.”
Dr. Hobbs and colleagues, and Dr. Kinsella, had no conflicts of interest to report.
SOURCE: MMWR Morb Mortal Wkly Rep. 2020;69:1925-9. doi: 10.15585/mmwr.mm6950e3.
Children and adolescents who receive positive COVID-19 test results are not only more likely to have been in close contact with someone with a confirmed case of the virus but also are less likely to have reported consistent mask use among students and staff inside the school they attended, reported Charlotte V. Hobbs, MD, and colleagues at the University of Mississippi, Jackson.
In partnership with the Centers for Disease Control and Prevention’s COVID-19 Response Team, Dr. Hobbs and colleagues conducted a case-control study of 397 children and adolescents under 18 years of age to assess school, community, and close contact exposures associated with pediatric COVID-19. Patients tested for COVID-19 at outpatient health centers or emergency departments affiliated with the University of Mississippi Medical Center between Sept. 1 and Nov. 5, 2020, were included in the study.
Nearly two-thirds reported that exposure came from family members
Of the total study participants observed, 82 (21%) were under 4 years of age; 214 (54%) were female; 217 (55%) were non-Hispanic black, and 145 (37%) were non-Hispanic white. More than half (53%) sought testing because of COVID-19 symptoms. Of those who tested positive, 66% reported having come into close contact with a COVID-19 case, and 64% reported that those contacts were family members, compared with 15% of contacts who were schoolmates and 27% who were child care classmates.
All participants completed in-person school or child care attendance less than 14 days before testing positive for the virus, including 62% of patients testing positive and 68% of those testing negative. The authors noted that school attendance itself was not found to be associated with any positive test results. In fact, parents in 64% of positive cases and 76% of negative cases reported mask wearing among children and staff inside places of learning.
Of those study participants testing positive who did come into close contact with someone with COVID-19, the contacts were more likely to be family members than school or child care classmates. Specifically, they were more likely, in the 2-week period preceding testing, to have attended gatherings with individuals outside their immediate households, including social events and activities with other children. Parents of students testing positive were also less likely to report consistent indoor mask use among their children older than 2 years and school staff members.
School attendance was not found to increase likelihood of testing positive
Attending in-person school or child care during the 2 weeks before the SARS-CoV-2 test was not associated with greater likelihood of testing positive, the study authors noted, adding that the majority of study respondents reported universal mask use inside school and child care facilities, consistent with Mississippi State Department of Health recommended guidelines.
Dr. Hobbs and colleagues reported at least four limitations of the study. They noted that the study participants may not be representative of youth in other geographic regions of the country. They considered the possibility of unmeasured confounding of participant behaviors that may not have been factored into the study. No attempt was made to verify parent claims of mask use at schools and child care programs. Lastly, they acknowledged that “case or control status might be subject to misclassification because of imperfect sensitivity or specificity of PCR-based testing.
As of Dec. 14, 2020, the CDC reported that 10.2% of all COVID-19 cases in the United States were in children and adolescents under the age of 18.
“Continued efforts to prevent transmission at schools and child care programs are important, as are assessments of various types of activities and exposures to identify risk factors for COVID-19 as children engage in classroom and social interactions.” Promoting behaviors to reduce exposures to the virus among youth in the household, the community, schools, and child care programs is important to preventing outbreaks of the virus at schools, the authors cautioned.
In a separate interview with this news organization, Karalyn Kinsella, MD, general pediatrician in a small group private practice in Cheshire, Conn., said, “What this report tells me is that COVID cases are more common when mask use is inconsistent in schools and at home and in schools that don’t properly adhere to CDC guidelines. Overall, so long as social distancing guidelines are followed, schools are pretty safe places for kids during this pandemic.”
This finding is important, since many families are keeping their children out of school over fears of contracting the virus, she added. Some of the consequences these children are suffering include a lack of social connection and structure, which in some cases is leading to worsening anxiety and depression, and for those with disabilities, such as those who receive physical therapy, occupational therapy, speech or have IEPs, they’re not getting the full benefit of the services that they would otherwise receive in person, she observed.
“I don’t think families really understand the risks of getting together with family or friends “in their bubble” or the risk of continuing sports participation. This is where the majority of COVID cases are coming from,” she said, adding that it is important to discuss this risk with them at appointments. So, when families ask us what we think of in-person learning, I think we should feel fairly confident that the benefit may outweigh the risk.”
Dr. Hobbs and colleagues, and Dr. Kinsella, had no conflicts of interest to report.
SOURCE: MMWR Morb Mortal Wkly Rep. 2020;69:1925-9. doi: 10.15585/mmwr.mm6950e3.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
IDSA panel updates guidelines on COVID molecular diagnostic tests
Saliva spit tests stack up well against the gold standard for molecular COVID-19 tests – the back-of-the-nose deep swab – without the discomfort and induced coughing or sneezing of the test taker, updated guidelines indicate.
In a press briefing on Jan. 6, the Infectious Diseases Society of America explained the findings of an expert panel that reviewed the literature since the IDSA released its first guidelines in May.
The panel found that saliva tests were especially effective if the test included instructions to cough or clear the throat before spitting into the tube, said panel chair Kimberly E. Hanson, MD, MHS, of University of Utah Health, Salt Lake City.
Throat swab alone less effective
Using a throat swab alone was less effective and missed more cases than the other methods, she said.
The IDSA has updated its recommendation: A saliva test or swabs from either the middle or front of the nose front are preferred to a throat swab alone.
A combination of saliva and swabs from the front and middle of the nose and throat together “looked pretty much equivalent” to the gold-standard deep swab, the panel found.
She acknowledged, however, that multiple swabs exacerbate already challenging supply issues.
Saliva samples do come with challenges, Dr. Hanson noted. A laboratory must validate that its systems can handle the stickier material. And asking a patient to cough necessitates more personal protective equipment for the health care professional.
Each center will have to tailor the specimen type it chooses, based on what resources it has available and the setting – whether in a hospital or a drive-through operation, for instance, she said.
Rapid testing vs. standard
Panel member Angela M. Caliendo, MD, PhD, of Brown University, Providence, R.I., said the panel preferred rapid polymerase chain reaction tests and standard, laboratory-based PCR tests over a rapid isothermal test.
The panel defined rapid tests as those for which results are available within an hour after a test provider has the specimen in hand. They excluded home tests for this category.
The only rapid isothermal test that had enough data on which to issue a recommendation was the ID NOW test (Abbott Labs), she noted.
Rapid PCR tests performed just as well as the standard laboratory-based tests, she said, with a high sensitivity of “97% on average and a very high specificity.”
But the rapid isothermal test had an average sensitivity of only about 80%, compared with the lab-based PCR test, Dr. Caliendo said, yielding a substantial number of false-negative results.
Testing centers will have to weigh the considerable advantages of having results in 15 minutes with a rapid isothermal test and being able to educate positive patients about immediate isolation against the potential for false negatives, which could send positive patients home thinking they don’t have the virus – and thus potentially spreading the disease.
And if a clinician gets a negative result with the rapid isothermal test, but has a strong suspicion the person has COVID or lives in an area with high prevalence, a backup test with a rapid PCR or laboratory-based test should be administered.
“You will miss a certain percentage of people using this rapid isothermal test,” she said.
However, Dr. Caliendo said, if the only available option is the isothermal test, “you should definitely use it because it’s certainly better than not testing at all.”
On a positive note, she said, all the varieties of tests have high specificity, so “you’re not going to see a lot of false-positive results.”
The guidelines back in May didn’t make recommendations on rapid tests, she said, because there weren’t enough data in the literature.
Dr. Caliendo noted that most of the available data were for symptomatic patients, but there are some data that show the amount of virus in the respiratory tract is similar for people with and without symptoms. The panel, therefore, expects that the performance of the various assays would be similar whether or not a person had symptoms.
Testing the immunocompromised
Dr. Hanson said the original recommendation in May was to do molecular testing for asymptomatic people who were awaiting a transplant or were waiting to start immunosuppressive therapy for cancer or an autoimmune disease. Now the current guidelines “make no recommendation for or against screening” in those cases.
Dr. Hanson added that the panel feels that patients awaiting bone marrow and solid organ transplants should have the testing because of the high risks that will result if patients have contracted the virus.
But for those with cancer or an autoimmune disease, the panel decided to leave it up to each physician to assess individual risk and determine whether the patient should be tested.
Home testing
The IDSA guidelines didn’t weigh in on home testing because the products are so new and studies so far have included fewer than 200 patients. But Dr. Caliendo said they clearly perform better earlier in the disease phase – the first 5-7 days – when the amount of the virus is higher.
Dr. Hanson and Dr. Caliendo also fielded a question about what the new virus variant, first discovered in the United Kingdom and now spreading to other countries (including the United States) means for diagnostic testing.
“So far we think with the majority of tests that are [emergency use] authorized, it doesn’t look like this new variant should really affect test performance,” Dr. Hanson said.
The variant has differences in the spike gene, and many of the current tests detect and identify SARS-CoV-2 without the spike gene so they wouldn’t be affected, she added.
Dr. Caliendo agreed: “I think the vast majority of our tests should be in good shape.”
Dr. Hanson and Dr. Caliendo disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Saliva spit tests stack up well against the gold standard for molecular COVID-19 tests – the back-of-the-nose deep swab – without the discomfort and induced coughing or sneezing of the test taker, updated guidelines indicate.
In a press briefing on Jan. 6, the Infectious Diseases Society of America explained the findings of an expert panel that reviewed the literature since the IDSA released its first guidelines in May.
The panel found that saliva tests were especially effective if the test included instructions to cough or clear the throat before spitting into the tube, said panel chair Kimberly E. Hanson, MD, MHS, of University of Utah Health, Salt Lake City.
Throat swab alone less effective
Using a throat swab alone was less effective and missed more cases than the other methods, she said.
The IDSA has updated its recommendation: A saliva test or swabs from either the middle or front of the nose front are preferred to a throat swab alone.
A combination of saliva and swabs from the front and middle of the nose and throat together “looked pretty much equivalent” to the gold-standard deep swab, the panel found.
She acknowledged, however, that multiple swabs exacerbate already challenging supply issues.
Saliva samples do come with challenges, Dr. Hanson noted. A laboratory must validate that its systems can handle the stickier material. And asking a patient to cough necessitates more personal protective equipment for the health care professional.
Each center will have to tailor the specimen type it chooses, based on what resources it has available and the setting – whether in a hospital or a drive-through operation, for instance, she said.
Rapid testing vs. standard
Panel member Angela M. Caliendo, MD, PhD, of Brown University, Providence, R.I., said the panel preferred rapid polymerase chain reaction tests and standard, laboratory-based PCR tests over a rapid isothermal test.
The panel defined rapid tests as those for which results are available within an hour after a test provider has the specimen in hand. They excluded home tests for this category.
The only rapid isothermal test that had enough data on which to issue a recommendation was the ID NOW test (Abbott Labs), she noted.
Rapid PCR tests performed just as well as the standard laboratory-based tests, she said, with a high sensitivity of “97% on average and a very high specificity.”
But the rapid isothermal test had an average sensitivity of only about 80%, compared with the lab-based PCR test, Dr. Caliendo said, yielding a substantial number of false-negative results.
Testing centers will have to weigh the considerable advantages of having results in 15 minutes with a rapid isothermal test and being able to educate positive patients about immediate isolation against the potential for false negatives, which could send positive patients home thinking they don’t have the virus – and thus potentially spreading the disease.
And if a clinician gets a negative result with the rapid isothermal test, but has a strong suspicion the person has COVID or lives in an area with high prevalence, a backup test with a rapid PCR or laboratory-based test should be administered.
“You will miss a certain percentage of people using this rapid isothermal test,” she said.
However, Dr. Caliendo said, if the only available option is the isothermal test, “you should definitely use it because it’s certainly better than not testing at all.”
On a positive note, she said, all the varieties of tests have high specificity, so “you’re not going to see a lot of false-positive results.”
The guidelines back in May didn’t make recommendations on rapid tests, she said, because there weren’t enough data in the literature.
Dr. Caliendo noted that most of the available data were for symptomatic patients, but there are some data that show the amount of virus in the respiratory tract is similar for people with and without symptoms. The panel, therefore, expects that the performance of the various assays would be similar whether or not a person had symptoms.
Testing the immunocompromised
Dr. Hanson said the original recommendation in May was to do molecular testing for asymptomatic people who were awaiting a transplant or were waiting to start immunosuppressive therapy for cancer or an autoimmune disease. Now the current guidelines “make no recommendation for or against screening” in those cases.
Dr. Hanson added that the panel feels that patients awaiting bone marrow and solid organ transplants should have the testing because of the high risks that will result if patients have contracted the virus.
But for those with cancer or an autoimmune disease, the panel decided to leave it up to each physician to assess individual risk and determine whether the patient should be tested.
Home testing
The IDSA guidelines didn’t weigh in on home testing because the products are so new and studies so far have included fewer than 200 patients. But Dr. Caliendo said they clearly perform better earlier in the disease phase – the first 5-7 days – when the amount of the virus is higher.
Dr. Hanson and Dr. Caliendo also fielded a question about what the new virus variant, first discovered in the United Kingdom and now spreading to other countries (including the United States) means for diagnostic testing.
“So far we think with the majority of tests that are [emergency use] authorized, it doesn’t look like this new variant should really affect test performance,” Dr. Hanson said.
The variant has differences in the spike gene, and many of the current tests detect and identify SARS-CoV-2 without the spike gene so they wouldn’t be affected, she added.
Dr. Caliendo agreed: “I think the vast majority of our tests should be in good shape.”
Dr. Hanson and Dr. Caliendo disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Saliva spit tests stack up well against the gold standard for molecular COVID-19 tests – the back-of-the-nose deep swab – without the discomfort and induced coughing or sneezing of the test taker, updated guidelines indicate.
In a press briefing on Jan. 6, the Infectious Diseases Society of America explained the findings of an expert panel that reviewed the literature since the IDSA released its first guidelines in May.
The panel found that saliva tests were especially effective if the test included instructions to cough or clear the throat before spitting into the tube, said panel chair Kimberly E. Hanson, MD, MHS, of University of Utah Health, Salt Lake City.
Throat swab alone less effective
Using a throat swab alone was less effective and missed more cases than the other methods, she said.
The IDSA has updated its recommendation: A saliva test or swabs from either the middle or front of the nose front are preferred to a throat swab alone.
A combination of saliva and swabs from the front and middle of the nose and throat together “looked pretty much equivalent” to the gold-standard deep swab, the panel found.
She acknowledged, however, that multiple swabs exacerbate already challenging supply issues.
Saliva samples do come with challenges, Dr. Hanson noted. A laboratory must validate that its systems can handle the stickier material. And asking a patient to cough necessitates more personal protective equipment for the health care professional.
Each center will have to tailor the specimen type it chooses, based on what resources it has available and the setting – whether in a hospital or a drive-through operation, for instance, she said.
Rapid testing vs. standard
Panel member Angela M. Caliendo, MD, PhD, of Brown University, Providence, R.I., said the panel preferred rapid polymerase chain reaction tests and standard, laboratory-based PCR tests over a rapid isothermal test.
The panel defined rapid tests as those for which results are available within an hour after a test provider has the specimen in hand. They excluded home tests for this category.
The only rapid isothermal test that had enough data on which to issue a recommendation was the ID NOW test (Abbott Labs), she noted.
Rapid PCR tests performed just as well as the standard laboratory-based tests, she said, with a high sensitivity of “97% on average and a very high specificity.”
But the rapid isothermal test had an average sensitivity of only about 80%, compared with the lab-based PCR test, Dr. Caliendo said, yielding a substantial number of false-negative results.
Testing centers will have to weigh the considerable advantages of having results in 15 minutes with a rapid isothermal test and being able to educate positive patients about immediate isolation against the potential for false negatives, which could send positive patients home thinking they don’t have the virus – and thus potentially spreading the disease.
And if a clinician gets a negative result with the rapid isothermal test, but has a strong suspicion the person has COVID or lives in an area with high prevalence, a backup test with a rapid PCR or laboratory-based test should be administered.
“You will miss a certain percentage of people using this rapid isothermal test,” she said.
However, Dr. Caliendo said, if the only available option is the isothermal test, “you should definitely use it because it’s certainly better than not testing at all.”
On a positive note, she said, all the varieties of tests have high specificity, so “you’re not going to see a lot of false-positive results.”
The guidelines back in May didn’t make recommendations on rapid tests, she said, because there weren’t enough data in the literature.
Dr. Caliendo noted that most of the available data were for symptomatic patients, but there are some data that show the amount of virus in the respiratory tract is similar for people with and without symptoms. The panel, therefore, expects that the performance of the various assays would be similar whether or not a person had symptoms.
Testing the immunocompromised
Dr. Hanson said the original recommendation in May was to do molecular testing for asymptomatic people who were awaiting a transplant or were waiting to start immunosuppressive therapy for cancer or an autoimmune disease. Now the current guidelines “make no recommendation for or against screening” in those cases.
Dr. Hanson added that the panel feels that patients awaiting bone marrow and solid organ transplants should have the testing because of the high risks that will result if patients have contracted the virus.
But for those with cancer or an autoimmune disease, the panel decided to leave it up to each physician to assess individual risk and determine whether the patient should be tested.
Home testing
The IDSA guidelines didn’t weigh in on home testing because the products are so new and studies so far have included fewer than 200 patients. But Dr. Caliendo said they clearly perform better earlier in the disease phase – the first 5-7 days – when the amount of the virus is higher.
Dr. Hanson and Dr. Caliendo also fielded a question about what the new virus variant, first discovered in the United Kingdom and now spreading to other countries (including the United States) means for diagnostic testing.
“So far we think with the majority of tests that are [emergency use] authorized, it doesn’t look like this new variant should really affect test performance,” Dr. Hanson said.
The variant has differences in the spike gene, and many of the current tests detect and identify SARS-CoV-2 without the spike gene so they wouldn’t be affected, she added.
Dr. Caliendo agreed: “I think the vast majority of our tests should be in good shape.”
Dr. Hanson and Dr. Caliendo disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Early use of high-titer plasma may prevent severe COVID-19
Administering convalescent plasma that has high levels of antibodies against SARS-CoV-2 within the first 3 days of symptoms was associated with significantly lower chances of progression to severe COVID-19, new evidence demonstrates.
In a trial of 160 older adults with COVID-19, half of whom were randomly assigned to receive plasma and half to receive placebo infusion, treatment with high-titer plasma lowered the relative risk for severe disease by 48% in an intent-to-treat analysis.
“We now have evidence, in the context of a small but well-designed study, that convalescent plasma with high titers of antibody against SARS-CoV-2 administered in the first 3 days of mild symptoms to infected elderly reduces progression of illness and the rate of severe presentations,” senior author Fernando Polack, MD, said in an interview.
“Not any plasma, not any time,” added Dr. Polack, an infectious disease specialist and scientific director at Fundacion INFANT and professor of pediatrics at the University of Buenos Aires. The key, he said, is to select plasma in the upper 28th percentile of IgG antibody concentrations and to administer therapy prior to disease progression.
The study was published online Jan. 6 in The New England Journal of Medicine.
“It’s a very good study and approaches a different population from the PlasmAr study,” Ventura Simonovich, MD, chief of the clinical pharmacology section, Medical Clinic Service, Hospital Italiano de Buenos Aires, said in an interview. “This is the first published randomized controlled trial that shows real benefit in this [older adult] population, the most vulnerable in this disease,” he said.
Dr. Simonovich, who was not affiliated with the current study, was lead author of the PlasmAr trial, which was published in The New England Journal of Medicine Nov. 24, 2020. In that trial, the researchers evaluated adults aged 18 years and older and found no significant benefit with convalescent plasma treatment over placebo for patients with COVID-19 and severe pneumonia.
“We know antibodies work best when given early and in high dose. This is one of the rare reports that validates it in the outpatient setting,” David Sullivan, MD, professor of molecular biology and immunology at Johns Hopkins Bloomberg School of Public Health, Baltimore, said in an interview when asked to comment.
Dr. Sullivan pointed out that most previous studies on convalescent plasma focused on patients with COVID-19 who had severe cases late in the disease course.
Regarding the current study, he said, “The striking thing is treating people within 3 days of illness.”
A more cautious interpretation may be warranted, one expert said. “The study demonstrates the benefit of early intervention. There was a dose-dependent effect, with higher titers providing a greater benefit,” Manoj Menon, MD, MPH, a hematologist and oncologist at the University of Washington, Seattle, said in an interview.
“Taken together, the findings have biologic plausibility and produce more data on the role of convalescent plasma to a relevant age cohort,” he added.
However, Dr. Menon said: “Given the limited sample size, I do not think this study, although well conducted, definitively addresses the role of convalescent plasma for COVID-19. But it does merit additional study.”
A search for clear answers
Treatments that target the early stages of COVID-19 “remain elusive. Few strategies provide benefit, several have failed, and others are being evaluated,” the researchers noted. “In hospitalized patients with COVID-19, the infusion of convalescent plasma against SARS-CoV-2 late in the course of illness has not shown clear benefits and, consequently, the most appropriate antibody concentrations for effective treatment are unclear.”
To learn more, Dr. Polack and colleagues included patients with PCR-confirmed COVID-19 who were aged 75 years or older, regardless of comorbidities. They also included patients aged 65-74 years who had at least one underlying condition. Participants were enrolled at clinical sites or geriatric units in Argentina. The mean age was 77 years, and 62% were women.
In an intent-to-treat analysis, the primary outcome – severe respiratory disease – occurred in 16% of the plasma recipients, vs. 31% of the group that received placebo. The relative risk was 0.52 (95% confidence interval, 0.29-0.94; P = .03).
The number needed to treat to avoid a severe respiratory disease episode was 7 (95% CI, 4-50).
Life-threatening respiratory disease, a secondary outcome, occurred in four people in the plasma group, compared with 10 in the placebo group. Two patients in the treatment group and four patients in the placebo group died.
The researchers also ran a modified intent-to-treat analysis that excluded six participants who experienced severe respiratory disease prior to receiving plasma or placebo. In this analysis, efficacy of plasma therapy increased to 60%.
“Again, this finding suggests that early intervention is critical for efficacy,” the investigators noted.
The investigators, who are based in Argentina, defined their primary endpoint as a respiratory rate of 30 or more breaths per minute and/or an oxygen saturation of less than 93% while breathing ambient air.
Dr. Sullivan pointed out that this is equivalent to the threshold commonly used for hospitalizing people with COVID-19 in the United States. “So it’s equivalent to avoiding hospitalizations. The take-home is high-titer plasma prevents respiratory distress, which equals hospitalization for us.”
Dr. Sullivan is conducting similar research in the United States regarding the use of plasma for treatment or prevention. He and colleagues are evaluating adults aged 18-90 years, “not just the ones at highest risk for going to the hospital,” he said. Enrollment is ongoing.
An inexpensive therapy with global potential?
“Although our trial lacked the statistical power to discern long-term outcomes, the convalescent plasma group appeared to have better outcomes than the placebo group with respect to all secondary endpoints,” the researchers wrote. “Our findings underscore the need to return to the classic approach of treating acute viral infections early, and they define IgG targets that facilitate donor selection.”
Dr. Polack said, “This is an inexpensive solution to mitigate the burden of severe illness in the population most vulnerable to the virus: the elderly. And it has the attraction of being applicable not only in industrialized countries but in many areas of the developing world.”
Convalescent plasma “is a potentially inexpensive alternative to monoclonal antibodies,” the researchers added. Furthermore, “early infusions of convalescent plasma can provide a bridge to recovery for at-risk patients until vaccines become widely available.”
Dr. Polack said the study findings did not surprise him. “We always thought that, as it has been the case in the past with many therapeutic strategies against respiratory and other viral infections, the earlier you treat, the better.
“We just hoped that within 72 hours of symptoms we would be treating early enough – remember that there is a 4- to 5-day incubation period that the virus leverages before the first symptom – and with enough antibody,” he added.
“We are glad it worked,” he said.
The study was supported by the Bill and Melinda Gates Foundation and by the Fundación INFANT Pandemic Fund. Dr. Polack, Dr. Simonovich, and Dr. Sullivan have disclosed various financial relationships industry.
A version of this article first appeared on Medscape.com.
Administering convalescent plasma that has high levels of antibodies against SARS-CoV-2 within the first 3 days of symptoms was associated with significantly lower chances of progression to severe COVID-19, new evidence demonstrates.
In a trial of 160 older adults with COVID-19, half of whom were randomly assigned to receive plasma and half to receive placebo infusion, treatment with high-titer plasma lowered the relative risk for severe disease by 48% in an intent-to-treat analysis.
“We now have evidence, in the context of a small but well-designed study, that convalescent plasma with high titers of antibody against SARS-CoV-2 administered in the first 3 days of mild symptoms to infected elderly reduces progression of illness and the rate of severe presentations,” senior author Fernando Polack, MD, said in an interview.
“Not any plasma, not any time,” added Dr. Polack, an infectious disease specialist and scientific director at Fundacion INFANT and professor of pediatrics at the University of Buenos Aires. The key, he said, is to select plasma in the upper 28th percentile of IgG antibody concentrations and to administer therapy prior to disease progression.
The study was published online Jan. 6 in The New England Journal of Medicine.
“It’s a very good study and approaches a different population from the PlasmAr study,” Ventura Simonovich, MD, chief of the clinical pharmacology section, Medical Clinic Service, Hospital Italiano de Buenos Aires, said in an interview. “This is the first published randomized controlled trial that shows real benefit in this [older adult] population, the most vulnerable in this disease,” he said.
Dr. Simonovich, who was not affiliated with the current study, was lead author of the PlasmAr trial, which was published in The New England Journal of Medicine Nov. 24, 2020. In that trial, the researchers evaluated adults aged 18 years and older and found no significant benefit with convalescent plasma treatment over placebo for patients with COVID-19 and severe pneumonia.
“We know antibodies work best when given early and in high dose. This is one of the rare reports that validates it in the outpatient setting,” David Sullivan, MD, professor of molecular biology and immunology at Johns Hopkins Bloomberg School of Public Health, Baltimore, said in an interview when asked to comment.
Dr. Sullivan pointed out that most previous studies on convalescent plasma focused on patients with COVID-19 who had severe cases late in the disease course.
Regarding the current study, he said, “The striking thing is treating people within 3 days of illness.”
A more cautious interpretation may be warranted, one expert said. “The study demonstrates the benefit of early intervention. There was a dose-dependent effect, with higher titers providing a greater benefit,” Manoj Menon, MD, MPH, a hematologist and oncologist at the University of Washington, Seattle, said in an interview.
“Taken together, the findings have biologic plausibility and produce more data on the role of convalescent plasma to a relevant age cohort,” he added.
However, Dr. Menon said: “Given the limited sample size, I do not think this study, although well conducted, definitively addresses the role of convalescent plasma for COVID-19. But it does merit additional study.”
A search for clear answers
Treatments that target the early stages of COVID-19 “remain elusive. Few strategies provide benefit, several have failed, and others are being evaluated,” the researchers noted. “In hospitalized patients with COVID-19, the infusion of convalescent plasma against SARS-CoV-2 late in the course of illness has not shown clear benefits and, consequently, the most appropriate antibody concentrations for effective treatment are unclear.”
To learn more, Dr. Polack and colleagues included patients with PCR-confirmed COVID-19 who were aged 75 years or older, regardless of comorbidities. They also included patients aged 65-74 years who had at least one underlying condition. Participants were enrolled at clinical sites or geriatric units in Argentina. The mean age was 77 years, and 62% were women.
In an intent-to-treat analysis, the primary outcome – severe respiratory disease – occurred in 16% of the plasma recipients, vs. 31% of the group that received placebo. The relative risk was 0.52 (95% confidence interval, 0.29-0.94; P = .03).
The number needed to treat to avoid a severe respiratory disease episode was 7 (95% CI, 4-50).
Life-threatening respiratory disease, a secondary outcome, occurred in four people in the plasma group, compared with 10 in the placebo group. Two patients in the treatment group and four patients in the placebo group died.
The researchers also ran a modified intent-to-treat analysis that excluded six participants who experienced severe respiratory disease prior to receiving plasma or placebo. In this analysis, efficacy of plasma therapy increased to 60%.
“Again, this finding suggests that early intervention is critical for efficacy,” the investigators noted.
The investigators, who are based in Argentina, defined their primary endpoint as a respiratory rate of 30 or more breaths per minute and/or an oxygen saturation of less than 93% while breathing ambient air.
Dr. Sullivan pointed out that this is equivalent to the threshold commonly used for hospitalizing people with COVID-19 in the United States. “So it’s equivalent to avoiding hospitalizations. The take-home is high-titer plasma prevents respiratory distress, which equals hospitalization for us.”
Dr. Sullivan is conducting similar research in the United States regarding the use of plasma for treatment or prevention. He and colleagues are evaluating adults aged 18-90 years, “not just the ones at highest risk for going to the hospital,” he said. Enrollment is ongoing.
An inexpensive therapy with global potential?
“Although our trial lacked the statistical power to discern long-term outcomes, the convalescent plasma group appeared to have better outcomes than the placebo group with respect to all secondary endpoints,” the researchers wrote. “Our findings underscore the need to return to the classic approach of treating acute viral infections early, and they define IgG targets that facilitate donor selection.”
Dr. Polack said, “This is an inexpensive solution to mitigate the burden of severe illness in the population most vulnerable to the virus: the elderly. And it has the attraction of being applicable not only in industrialized countries but in many areas of the developing world.”
Convalescent plasma “is a potentially inexpensive alternative to monoclonal antibodies,” the researchers added. Furthermore, “early infusions of convalescent plasma can provide a bridge to recovery for at-risk patients until vaccines become widely available.”
Dr. Polack said the study findings did not surprise him. “We always thought that, as it has been the case in the past with many therapeutic strategies against respiratory and other viral infections, the earlier you treat, the better.
“We just hoped that within 72 hours of symptoms we would be treating early enough – remember that there is a 4- to 5-day incubation period that the virus leverages before the first symptom – and with enough antibody,” he added.
“We are glad it worked,” he said.
The study was supported by the Bill and Melinda Gates Foundation and by the Fundación INFANT Pandemic Fund. Dr. Polack, Dr. Simonovich, and Dr. Sullivan have disclosed various financial relationships industry.
A version of this article first appeared on Medscape.com.
Administering convalescent plasma that has high levels of antibodies against SARS-CoV-2 within the first 3 days of symptoms was associated with significantly lower chances of progression to severe COVID-19, new evidence demonstrates.
In a trial of 160 older adults with COVID-19, half of whom were randomly assigned to receive plasma and half to receive placebo infusion, treatment with high-titer plasma lowered the relative risk for severe disease by 48% in an intent-to-treat analysis.
“We now have evidence, in the context of a small but well-designed study, that convalescent plasma with high titers of antibody against SARS-CoV-2 administered in the first 3 days of mild symptoms to infected elderly reduces progression of illness and the rate of severe presentations,” senior author Fernando Polack, MD, said in an interview.
“Not any plasma, not any time,” added Dr. Polack, an infectious disease specialist and scientific director at Fundacion INFANT and professor of pediatrics at the University of Buenos Aires. The key, he said, is to select plasma in the upper 28th percentile of IgG antibody concentrations and to administer therapy prior to disease progression.
The study was published online Jan. 6 in The New England Journal of Medicine.
“It’s a very good study and approaches a different population from the PlasmAr study,” Ventura Simonovich, MD, chief of the clinical pharmacology section, Medical Clinic Service, Hospital Italiano de Buenos Aires, said in an interview. “This is the first published randomized controlled trial that shows real benefit in this [older adult] population, the most vulnerable in this disease,” he said.
Dr. Simonovich, who was not affiliated with the current study, was lead author of the PlasmAr trial, which was published in The New England Journal of Medicine Nov. 24, 2020. In that trial, the researchers evaluated adults aged 18 years and older and found no significant benefit with convalescent plasma treatment over placebo for patients with COVID-19 and severe pneumonia.
“We know antibodies work best when given early and in high dose. This is one of the rare reports that validates it in the outpatient setting,” David Sullivan, MD, professor of molecular biology and immunology at Johns Hopkins Bloomberg School of Public Health, Baltimore, said in an interview when asked to comment.
Dr. Sullivan pointed out that most previous studies on convalescent plasma focused on patients with COVID-19 who had severe cases late in the disease course.
Regarding the current study, he said, “The striking thing is treating people within 3 days of illness.”
A more cautious interpretation may be warranted, one expert said. “The study demonstrates the benefit of early intervention. There was a dose-dependent effect, with higher titers providing a greater benefit,” Manoj Menon, MD, MPH, a hematologist and oncologist at the University of Washington, Seattle, said in an interview.
“Taken together, the findings have biologic plausibility and produce more data on the role of convalescent plasma to a relevant age cohort,” he added.
However, Dr. Menon said: “Given the limited sample size, I do not think this study, although well conducted, definitively addresses the role of convalescent plasma for COVID-19. But it does merit additional study.”
A search for clear answers
Treatments that target the early stages of COVID-19 “remain elusive. Few strategies provide benefit, several have failed, and others are being evaluated,” the researchers noted. “In hospitalized patients with COVID-19, the infusion of convalescent plasma against SARS-CoV-2 late in the course of illness has not shown clear benefits and, consequently, the most appropriate antibody concentrations for effective treatment are unclear.”
To learn more, Dr. Polack and colleagues included patients with PCR-confirmed COVID-19 who were aged 75 years or older, regardless of comorbidities. They also included patients aged 65-74 years who had at least one underlying condition. Participants were enrolled at clinical sites or geriatric units in Argentina. The mean age was 77 years, and 62% were women.
In an intent-to-treat analysis, the primary outcome – severe respiratory disease – occurred in 16% of the plasma recipients, vs. 31% of the group that received placebo. The relative risk was 0.52 (95% confidence interval, 0.29-0.94; P = .03).
The number needed to treat to avoid a severe respiratory disease episode was 7 (95% CI, 4-50).
Life-threatening respiratory disease, a secondary outcome, occurred in four people in the plasma group, compared with 10 in the placebo group. Two patients in the treatment group and four patients in the placebo group died.
The researchers also ran a modified intent-to-treat analysis that excluded six participants who experienced severe respiratory disease prior to receiving plasma or placebo. In this analysis, efficacy of plasma therapy increased to 60%.
“Again, this finding suggests that early intervention is critical for efficacy,” the investigators noted.
The investigators, who are based in Argentina, defined their primary endpoint as a respiratory rate of 30 or more breaths per minute and/or an oxygen saturation of less than 93% while breathing ambient air.
Dr. Sullivan pointed out that this is equivalent to the threshold commonly used for hospitalizing people with COVID-19 in the United States. “So it’s equivalent to avoiding hospitalizations. The take-home is high-titer plasma prevents respiratory distress, which equals hospitalization for us.”
Dr. Sullivan is conducting similar research in the United States regarding the use of plasma for treatment or prevention. He and colleagues are evaluating adults aged 18-90 years, “not just the ones at highest risk for going to the hospital,” he said. Enrollment is ongoing.
An inexpensive therapy with global potential?
“Although our trial lacked the statistical power to discern long-term outcomes, the convalescent plasma group appeared to have better outcomes than the placebo group with respect to all secondary endpoints,” the researchers wrote. “Our findings underscore the need to return to the classic approach of treating acute viral infections early, and they define IgG targets that facilitate donor selection.”
Dr. Polack said, “This is an inexpensive solution to mitigate the burden of severe illness in the population most vulnerable to the virus: the elderly. And it has the attraction of being applicable not only in industrialized countries but in many areas of the developing world.”
Convalescent plasma “is a potentially inexpensive alternative to monoclonal antibodies,” the researchers added. Furthermore, “early infusions of convalescent plasma can provide a bridge to recovery for at-risk patients until vaccines become widely available.”
Dr. Polack said the study findings did not surprise him. “We always thought that, as it has been the case in the past with many therapeutic strategies against respiratory and other viral infections, the earlier you treat, the better.
“We just hoped that within 72 hours of symptoms we would be treating early enough – remember that there is a 4- to 5-day incubation period that the virus leverages before the first symptom – and with enough antibody,” he added.
“We are glad it worked,” he said.
The study was supported by the Bill and Melinda Gates Foundation and by the Fundación INFANT Pandemic Fund. Dr. Polack, Dr. Simonovich, and Dr. Sullivan have disclosed various financial relationships industry.
A version of this article first appeared on Medscape.com.
No increase seen in children’s cumulative COVID-19 burden
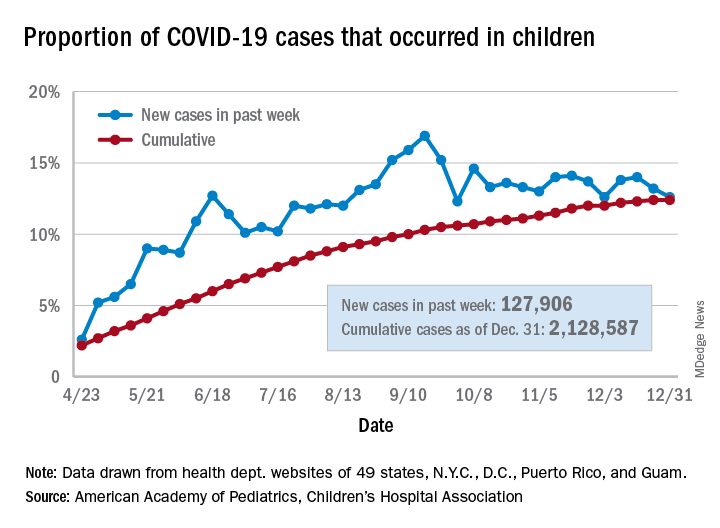
Children’s share of the cumulative COVID-19 burden remained at 12.4% for a second consecutive week, the AAP and CHA said in their weekly report. The last full week of 2020 also marked the second consecutive drop in new cases, although that may be holiday related.
There were almost 128,000 new cases of COVID-19 reported in children for the week, down from 179,000 cases the week before (Dec. 24) and down from the pandemic high of 182,000 reported 2 weeks earlier (Dec.17), based on data from 49 state health departments (excluding New York), along with the District of Columbia, New York City, Puerto Rico, and Guam.
Children’s proportion of new cases for the week, 12.6%, is at its lowest point since early October after dropping for the second week in a row. The cumulative rate of COVID-19 infection, however, is now 2,828 cases per 100,000 children, up from 2,658 the previous week, the AAP and CHA said.
State-level metrics show that North Dakota has the highest cumulative rate at 7,851 per 100,000 children and Hawaii the lowest at 828. Wyoming’s cumulative proportion of child cases, 20.3%, is the highest in the country, while Florida, which uses an age range of 0-14 years for children, is the lowest at 7.1%. California’s total of 268,000 cases is almost double the number of second-place Illinois (138,000), the AAP/CHA data show.
Cumulative child deaths from COVID-19 are up to 179 in the jurisdictions reporting such data (43 states and New York City). That represents just 0.6% of all coronavirus-related deaths and has changed little over the last several months – never rising higher than 0.7% or dropping below 0.6% since early July, according to the report.

Children’s share of the cumulative COVID-19 burden remained at 12.4% for a second consecutive week, the AAP and CHA said in their weekly report. The last full week of 2020 also marked the second consecutive drop in new cases, although that may be holiday related.
There were almost 128,000 new cases of COVID-19 reported in children for the week, down from 179,000 cases the week before (Dec. 24) and down from the pandemic high of 182,000 reported 2 weeks earlier (Dec.17), based on data from 49 state health departments (excluding New York), along with the District of Columbia, New York City, Puerto Rico, and Guam.
Children’s proportion of new cases for the week, 12.6%, is at its lowest point since early October after dropping for the second week in a row. The cumulative rate of COVID-19 infection, however, is now 2,828 cases per 100,000 children, up from 2,658 the previous week, the AAP and CHA said.
State-level metrics show that North Dakota has the highest cumulative rate at 7,851 per 100,000 children and Hawaii the lowest at 828. Wyoming’s cumulative proportion of child cases, 20.3%, is the highest in the country, while Florida, which uses an age range of 0-14 years for children, is the lowest at 7.1%. California’s total of 268,000 cases is almost double the number of second-place Illinois (138,000), the AAP/CHA data show.
Cumulative child deaths from COVID-19 are up to 179 in the jurisdictions reporting such data (43 states and New York City). That represents just 0.6% of all coronavirus-related deaths and has changed little over the last several months – never rising higher than 0.7% or dropping below 0.6% since early July, according to the report.

Children’s share of the cumulative COVID-19 burden remained at 12.4% for a second consecutive week, the AAP and CHA said in their weekly report. The last full week of 2020 also marked the second consecutive drop in new cases, although that may be holiday related.
There were almost 128,000 new cases of COVID-19 reported in children for the week, down from 179,000 cases the week before (Dec. 24) and down from the pandemic high of 182,000 reported 2 weeks earlier (Dec.17), based on data from 49 state health departments (excluding New York), along with the District of Columbia, New York City, Puerto Rico, and Guam.
Children’s proportion of new cases for the week, 12.6%, is at its lowest point since early October after dropping for the second week in a row. The cumulative rate of COVID-19 infection, however, is now 2,828 cases per 100,000 children, up from 2,658 the previous week, the AAP and CHA said.
State-level metrics show that North Dakota has the highest cumulative rate at 7,851 per 100,000 children and Hawaii the lowest at 828. Wyoming’s cumulative proportion of child cases, 20.3%, is the highest in the country, while Florida, which uses an age range of 0-14 years for children, is the lowest at 7.1%. California’s total of 268,000 cases is almost double the number of second-place Illinois (138,000), the AAP/CHA data show.
Cumulative child deaths from COVID-19 are up to 179 in the jurisdictions reporting such data (43 states and New York City). That represents just 0.6% of all coronavirus-related deaths and has changed little over the last several months – never rising higher than 0.7% or dropping below 0.6% since early July, according to the report.