User login
Updated gout guidelines: Don’t let kidney function dictate allopurinol dosing
ATLANTA – Soon-to-be-published gout guidelines from the American College of Rheumatology will recommend dosing allopurinol above 300 mg/day to get serum urate below 6 mg/dL, even in people with renal impairment.
It’s the same strong treat-to-target recommendation the group made in its last outing in 2012, but “we now have more evidence to support it,” said co–lead author, rheumatologist, and epidemiologist Tuhina Neogi, MD, PhD, a professor of medicine at Boston University.
She gave a sneak preview of the new guidelines, which will be published in 2020, at the ACR annual meeting. They are under review, but she said the “major recommendations will remain the same.”
“There will still be controversy that we have not yet proven that a threshold of 6 mg/dL is better than a threshold of 7 mg/dL, but we know that” at physiologic pH and temperature, monosodium urate starts to crystallize out at 6.8 mg/dL. “Serum urate is not a perfect measure or total body urate, so we need to get urate to below at least 6 mg/dL,” she said, and perhaps lower in some.
A popular alternative in primary care – where most gout is managed – is to treat to avoid symptoms. It “has no evidence,” and people “end up getting tophaceous gout with joint destruction. Suppressive colchicine therapy does not manage underlying hyperuricemia,” Dr. Neogi said.
With the symptom approach, “patients are often [profoundly] dismayed” when they find out they have large tophi and joint damage because they weren’t managed properly. “Primary care physicians [don’t often] see that because those patients don’t go back to them,” she said.
Dr. Neogi suspects that, for rheumatologists, the biggest surprise in the new guidelines will be a deemphasis on lifestyle and dietary factors. They can be triggers, but “gout is increasingly recognized as largely genetically determined,” and the impact of other factors on serum urate is low. Plus, “patients are embarrassed” by gout, and even less comfortable being honest with physicians “if they think we are blaming them,” she said.
The new document will recommend allopurinol as the definitive first-line option for hyperuricemia. Febuxostat (Uloric) was put on pretty much equal footing in 2012, but now “we acknowledge” that allopurinol dosing in head-to-head trials – 300 mg/day or 200 mg/day with renal impairment – was too low for most people, “so to say febuxostat is equivalent or superior isn’t really fair.” The substantially higher cost of febuxostat was also taken into consideration, she said.
The ACR will broaden the indications for urate lowering beyond frequent flares, tophi, and radiologic joint damage to include conditional, shared decision-making recommendations for people who have less than two flares per year, those with kidney stones, and people with a first flare if they are particularly susceptible to a second – namely those with serum urate at or above 9 mg/dL and people with stage 3 or worse chronic kidney disease, who are less able to tolerate NSAIDs and colchicine for symptom treatment.
The group will also relax its advice against treating asymptomatic hyperuricemia. Febuxostat trials have shown a reduction in incident gout, but the number needed to treat was large, so the ACR will recommend shared decision making.
Inadequate allopurinol dosing, meanwhile, has been the bête noire of rheumatology for years, but there is still reluctance among many to go above 300 mg/day. Dr. Neogi said it’s because of a decades-old concern, “unsupported by any evidence, that higher doses may be detrimental in people with renal insufficiency.” It’s frustrating, she said, because “there is good data supporting the safety of increasing the dose above 300 mg/day even in those with renal impairment,” and not doing so opens the door to entirely preventable complications.
As for allopurinol hypersensitivity – another reason people shy away from higher dosing, especially in the renally impaired – the trick is to start low and slowly titrate allopurinol up to the target urate range. Asian and black people, especially, should be screened beforehand for the HLA-B*58:01 genetic variant that increases the risk of severe reactions. Both will be strong recommendations in the new guidelines.
Dr. Neogi didn’t have any relevant industry disclosures.
ATLANTA – Soon-to-be-published gout guidelines from the American College of Rheumatology will recommend dosing allopurinol above 300 mg/day to get serum urate below 6 mg/dL, even in people with renal impairment.
It’s the same strong treat-to-target recommendation the group made in its last outing in 2012, but “we now have more evidence to support it,” said co–lead author, rheumatologist, and epidemiologist Tuhina Neogi, MD, PhD, a professor of medicine at Boston University.
She gave a sneak preview of the new guidelines, which will be published in 2020, at the ACR annual meeting. They are under review, but she said the “major recommendations will remain the same.”
“There will still be controversy that we have not yet proven that a threshold of 6 mg/dL is better than a threshold of 7 mg/dL, but we know that” at physiologic pH and temperature, monosodium urate starts to crystallize out at 6.8 mg/dL. “Serum urate is not a perfect measure or total body urate, so we need to get urate to below at least 6 mg/dL,” she said, and perhaps lower in some.
A popular alternative in primary care – where most gout is managed – is to treat to avoid symptoms. It “has no evidence,” and people “end up getting tophaceous gout with joint destruction. Suppressive colchicine therapy does not manage underlying hyperuricemia,” Dr. Neogi said.
With the symptom approach, “patients are often [profoundly] dismayed” when they find out they have large tophi and joint damage because they weren’t managed properly. “Primary care physicians [don’t often] see that because those patients don’t go back to them,” she said.
Dr. Neogi suspects that, for rheumatologists, the biggest surprise in the new guidelines will be a deemphasis on lifestyle and dietary factors. They can be triggers, but “gout is increasingly recognized as largely genetically determined,” and the impact of other factors on serum urate is low. Plus, “patients are embarrassed” by gout, and even less comfortable being honest with physicians “if they think we are blaming them,” she said.
The new document will recommend allopurinol as the definitive first-line option for hyperuricemia. Febuxostat (Uloric) was put on pretty much equal footing in 2012, but now “we acknowledge” that allopurinol dosing in head-to-head trials – 300 mg/day or 200 mg/day with renal impairment – was too low for most people, “so to say febuxostat is equivalent or superior isn’t really fair.” The substantially higher cost of febuxostat was also taken into consideration, she said.
The ACR will broaden the indications for urate lowering beyond frequent flares, tophi, and radiologic joint damage to include conditional, shared decision-making recommendations for people who have less than two flares per year, those with kidney stones, and people with a first flare if they are particularly susceptible to a second – namely those with serum urate at or above 9 mg/dL and people with stage 3 or worse chronic kidney disease, who are less able to tolerate NSAIDs and colchicine for symptom treatment.
The group will also relax its advice against treating asymptomatic hyperuricemia. Febuxostat trials have shown a reduction in incident gout, but the number needed to treat was large, so the ACR will recommend shared decision making.
Inadequate allopurinol dosing, meanwhile, has been the bête noire of rheumatology for years, but there is still reluctance among many to go above 300 mg/day. Dr. Neogi said it’s because of a decades-old concern, “unsupported by any evidence, that higher doses may be detrimental in people with renal insufficiency.” It’s frustrating, she said, because “there is good data supporting the safety of increasing the dose above 300 mg/day even in those with renal impairment,” and not doing so opens the door to entirely preventable complications.
As for allopurinol hypersensitivity – another reason people shy away from higher dosing, especially in the renally impaired – the trick is to start low and slowly titrate allopurinol up to the target urate range. Asian and black people, especially, should be screened beforehand for the HLA-B*58:01 genetic variant that increases the risk of severe reactions. Both will be strong recommendations in the new guidelines.
Dr. Neogi didn’t have any relevant industry disclosures.
ATLANTA – Soon-to-be-published gout guidelines from the American College of Rheumatology will recommend dosing allopurinol above 300 mg/day to get serum urate below 6 mg/dL, even in people with renal impairment.
It’s the same strong treat-to-target recommendation the group made in its last outing in 2012, but “we now have more evidence to support it,” said co–lead author, rheumatologist, and epidemiologist Tuhina Neogi, MD, PhD, a professor of medicine at Boston University.
She gave a sneak preview of the new guidelines, which will be published in 2020, at the ACR annual meeting. They are under review, but she said the “major recommendations will remain the same.”
“There will still be controversy that we have not yet proven that a threshold of 6 mg/dL is better than a threshold of 7 mg/dL, but we know that” at physiologic pH and temperature, monosodium urate starts to crystallize out at 6.8 mg/dL. “Serum urate is not a perfect measure or total body urate, so we need to get urate to below at least 6 mg/dL,” she said, and perhaps lower in some.
A popular alternative in primary care – where most gout is managed – is to treat to avoid symptoms. It “has no evidence,” and people “end up getting tophaceous gout with joint destruction. Suppressive colchicine therapy does not manage underlying hyperuricemia,” Dr. Neogi said.
With the symptom approach, “patients are often [profoundly] dismayed” when they find out they have large tophi and joint damage because they weren’t managed properly. “Primary care physicians [don’t often] see that because those patients don’t go back to them,” she said.
Dr. Neogi suspects that, for rheumatologists, the biggest surprise in the new guidelines will be a deemphasis on lifestyle and dietary factors. They can be triggers, but “gout is increasingly recognized as largely genetically determined,” and the impact of other factors on serum urate is low. Plus, “patients are embarrassed” by gout, and even less comfortable being honest with physicians “if they think we are blaming them,” she said.
The new document will recommend allopurinol as the definitive first-line option for hyperuricemia. Febuxostat (Uloric) was put on pretty much equal footing in 2012, but now “we acknowledge” that allopurinol dosing in head-to-head trials – 300 mg/day or 200 mg/day with renal impairment – was too low for most people, “so to say febuxostat is equivalent or superior isn’t really fair.” The substantially higher cost of febuxostat was also taken into consideration, she said.
The ACR will broaden the indications for urate lowering beyond frequent flares, tophi, and radiologic joint damage to include conditional, shared decision-making recommendations for people who have less than two flares per year, those with kidney stones, and people with a first flare if they are particularly susceptible to a second – namely those with serum urate at or above 9 mg/dL and people with stage 3 or worse chronic kidney disease, who are less able to tolerate NSAIDs and colchicine for symptom treatment.
The group will also relax its advice against treating asymptomatic hyperuricemia. Febuxostat trials have shown a reduction in incident gout, but the number needed to treat was large, so the ACR will recommend shared decision making.
Inadequate allopurinol dosing, meanwhile, has been the bête noire of rheumatology for years, but there is still reluctance among many to go above 300 mg/day. Dr. Neogi said it’s because of a decades-old concern, “unsupported by any evidence, that higher doses may be detrimental in people with renal insufficiency.” It’s frustrating, she said, because “there is good data supporting the safety of increasing the dose above 300 mg/day even in those with renal impairment,” and not doing so opens the door to entirely preventable complications.
As for allopurinol hypersensitivity – another reason people shy away from higher dosing, especially in the renally impaired – the trick is to start low and slowly titrate allopurinol up to the target urate range. Asian and black people, especially, should be screened beforehand for the HLA-B*58:01 genetic variant that increases the risk of severe reactions. Both will be strong recommendations in the new guidelines.
Dr. Neogi didn’t have any relevant industry disclosures.
REPORTING FROM ACR 2019
Bile acid diarrhea guideline highlights data shortage
The Canadian Association of Gastroenterology (CAG) recently co-published a clinical practice guideline for the management of bile acid diarrhea (BAD) in Clinical Gastroenterology and Hepatology and the Journal of the Canadian Association of Gastroenterology.
Given a minimal evidence base, 16 out of the 17 guideline recommendations are conditional, according to lead author Daniel C. Sadowski, MD, of Royal Alexandra Hospital, Edmonton, Alta., and colleagues. Considering the shortage of high-quality evidence, the panel called for more randomized clinical trials to address current knowledge gaps.
“BAD is an understudied, often underappreciated condition, and questions remain regarding its diagnosis and treatment,” the panelists wrote in Clinical Gastroenterology and Hepatology. “There have been guidelines on the management of chronic diarrhea from the American Gastroenterological Association, and the British Society of Gastroenterology, but diagnosis and management of BAD was not assessed extensively in these publications. The British Society of Gastroenterology updated guidelines on the investigation of chronic diarrhea in adults, published after the consensus meeting, addressed some issues related to BAD.”
For the current guideline, using available evidence and clinical experience, expert panelists from Canada, the United States, and the United Kingdom aimed to “provide a reasonable and practical approach to care for specialists.” The guideline was further reviewed by the CAG Practice Affairs and Clinical Affairs Committees and the CAG Board of Directors.
The guideline first puts BAD in clinical context, noting a chronic diarrhea prevalence rate of approximately 5%. According to the guideline, approximately 1 out of 4 of these patients with chronic diarrhea may have BAD and prevalence of BAD is likely higher among those with other conditions, such as terminal ileal disease.
While BAD may be relatively common, it isn’t necessarily easy to diagnose, the panelists noted.
“The diagnosis of BAD continues to be a challenge, although this may be improved in the future with the general availability of screening serologic tests and other diagnostic tests,” the panelists wrote. “Although a treatment trial with bile acid sequestrants therapy (BAST) often is used, this approach has not been studied adequately, and likely is imprecise, and may lead to both undertreatment and overtreatment.”
Instead, the panelists recommended testing for BAD with 75-selenium homocholic acid taurine (SeHCAT) or 7-alpha-hydroxy-4-cholesten-3-one.
After addressing treatable causes of BAD, the guideline recommends initial therapy with cholestyramine or, if this is poorly tolerated, switching to BAST. However, the panelists advised against BAST for patients with resection or ileal Crohn’s disease, for whom other antidiarrheal agents are more suitable. When appropriate, BAST should be given at the lowest effective dose, with periodic trials of on-demand, intermittent administration, the panelists recommended. When BAST is ineffective, the guideline recommends that clinicians review concurrent medications as a possible cause of BAD or reinvestigate.
Concluding the guideline, the panelists emphasized the need for more high-quality research.
“The group recognized that specific, high-certainty evidence was lacking in many areas and recommended further studies that would improve the data available in future methodologic evaluations,” the panelists wrote.
While improving diagnostic accuracy of BAD should be a major goal of such research, progress is currently limited by an integral shortcoming of diagnostic test accuracy (DTA) studies, the panelists wrote.
“The main challenge in conducting DTA studies for BAD is the lack of a widely accepted or universally agreed-upon reference standard because the condition is defined and classified based on pathophysiologic mechanisms and its response to treatment (BAST),” the panelists wrote. “In addition, the index tests (SeHCAT, C4, FGF19, fecal bile acid assay) provide a continuous measure of metabolic function. Hence, DTA studies are not the most appropriate study design.”
“Therefore, one of the research priorities in BAD is for the scientific and clinical communities to agree on a reference standard that best represents BAD (e.g., response to BAST), with full understanding that the reference standard is and likely will be imperfect.”
The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Review the AGA clinical practice guideline on the laboratory evaluation of functional diarrhea and diarrhea-predominan irritable bowel syndrome in adults at https:/www.gastrojournal.org/article/S0016-5085(19)41083-4/fulltext.
SOURCE: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
The Canadian Association of Gastroenterology (CAG) recently co-published a clinical practice guideline for the management of bile acid diarrhea (BAD) in Clinical Gastroenterology and Hepatology and the Journal of the Canadian Association of Gastroenterology.
Given a minimal evidence base, 16 out of the 17 guideline recommendations are conditional, according to lead author Daniel C. Sadowski, MD, of Royal Alexandra Hospital, Edmonton, Alta., and colleagues. Considering the shortage of high-quality evidence, the panel called for more randomized clinical trials to address current knowledge gaps.
“BAD is an understudied, often underappreciated condition, and questions remain regarding its diagnosis and treatment,” the panelists wrote in Clinical Gastroenterology and Hepatology. “There have been guidelines on the management of chronic diarrhea from the American Gastroenterological Association, and the British Society of Gastroenterology, but diagnosis and management of BAD was not assessed extensively in these publications. The British Society of Gastroenterology updated guidelines on the investigation of chronic diarrhea in adults, published after the consensus meeting, addressed some issues related to BAD.”
For the current guideline, using available evidence and clinical experience, expert panelists from Canada, the United States, and the United Kingdom aimed to “provide a reasonable and practical approach to care for specialists.” The guideline was further reviewed by the CAG Practice Affairs and Clinical Affairs Committees and the CAG Board of Directors.
The guideline first puts BAD in clinical context, noting a chronic diarrhea prevalence rate of approximately 5%. According to the guideline, approximately 1 out of 4 of these patients with chronic diarrhea may have BAD and prevalence of BAD is likely higher among those with other conditions, such as terminal ileal disease.
While BAD may be relatively common, it isn’t necessarily easy to diagnose, the panelists noted.
“The diagnosis of BAD continues to be a challenge, although this may be improved in the future with the general availability of screening serologic tests and other diagnostic tests,” the panelists wrote. “Although a treatment trial with bile acid sequestrants therapy (BAST) often is used, this approach has not been studied adequately, and likely is imprecise, and may lead to both undertreatment and overtreatment.”
Instead, the panelists recommended testing for BAD with 75-selenium homocholic acid taurine (SeHCAT) or 7-alpha-hydroxy-4-cholesten-3-one.
After addressing treatable causes of BAD, the guideline recommends initial therapy with cholestyramine or, if this is poorly tolerated, switching to BAST. However, the panelists advised against BAST for patients with resection or ileal Crohn’s disease, for whom other antidiarrheal agents are more suitable. When appropriate, BAST should be given at the lowest effective dose, with periodic trials of on-demand, intermittent administration, the panelists recommended. When BAST is ineffective, the guideline recommends that clinicians review concurrent medications as a possible cause of BAD or reinvestigate.
Concluding the guideline, the panelists emphasized the need for more high-quality research.
“The group recognized that specific, high-certainty evidence was lacking in many areas and recommended further studies that would improve the data available in future methodologic evaluations,” the panelists wrote.
While improving diagnostic accuracy of BAD should be a major goal of such research, progress is currently limited by an integral shortcoming of diagnostic test accuracy (DTA) studies, the panelists wrote.
“The main challenge in conducting DTA studies for BAD is the lack of a widely accepted or universally agreed-upon reference standard because the condition is defined and classified based on pathophysiologic mechanisms and its response to treatment (BAST),” the panelists wrote. “In addition, the index tests (SeHCAT, C4, FGF19, fecal bile acid assay) provide a continuous measure of metabolic function. Hence, DTA studies are not the most appropriate study design.”
“Therefore, one of the research priorities in BAD is for the scientific and clinical communities to agree on a reference standard that best represents BAD (e.g., response to BAST), with full understanding that the reference standard is and likely will be imperfect.”
The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Review the AGA clinical practice guideline on the laboratory evaluation of functional diarrhea and diarrhea-predominan irritable bowel syndrome in adults at https:/www.gastrojournal.org/article/S0016-5085(19)41083-4/fulltext.
SOURCE: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
The Canadian Association of Gastroenterology (CAG) recently co-published a clinical practice guideline for the management of bile acid diarrhea (BAD) in Clinical Gastroenterology and Hepatology and the Journal of the Canadian Association of Gastroenterology.
Given a minimal evidence base, 16 out of the 17 guideline recommendations are conditional, according to lead author Daniel C. Sadowski, MD, of Royal Alexandra Hospital, Edmonton, Alta., and colleagues. Considering the shortage of high-quality evidence, the panel called for more randomized clinical trials to address current knowledge gaps.
“BAD is an understudied, often underappreciated condition, and questions remain regarding its diagnosis and treatment,” the panelists wrote in Clinical Gastroenterology and Hepatology. “There have been guidelines on the management of chronic diarrhea from the American Gastroenterological Association, and the British Society of Gastroenterology, but diagnosis and management of BAD was not assessed extensively in these publications. The British Society of Gastroenterology updated guidelines on the investigation of chronic diarrhea in adults, published after the consensus meeting, addressed some issues related to BAD.”
For the current guideline, using available evidence and clinical experience, expert panelists from Canada, the United States, and the United Kingdom aimed to “provide a reasonable and practical approach to care for specialists.” The guideline was further reviewed by the CAG Practice Affairs and Clinical Affairs Committees and the CAG Board of Directors.
The guideline first puts BAD in clinical context, noting a chronic diarrhea prevalence rate of approximately 5%. According to the guideline, approximately 1 out of 4 of these patients with chronic diarrhea may have BAD and prevalence of BAD is likely higher among those with other conditions, such as terminal ileal disease.
While BAD may be relatively common, it isn’t necessarily easy to diagnose, the panelists noted.
“The diagnosis of BAD continues to be a challenge, although this may be improved in the future with the general availability of screening serologic tests and other diagnostic tests,” the panelists wrote. “Although a treatment trial with bile acid sequestrants therapy (BAST) often is used, this approach has not been studied adequately, and likely is imprecise, and may lead to both undertreatment and overtreatment.”
Instead, the panelists recommended testing for BAD with 75-selenium homocholic acid taurine (SeHCAT) or 7-alpha-hydroxy-4-cholesten-3-one.
After addressing treatable causes of BAD, the guideline recommends initial therapy with cholestyramine or, if this is poorly tolerated, switching to BAST. However, the panelists advised against BAST for patients with resection or ileal Crohn’s disease, for whom other antidiarrheal agents are more suitable. When appropriate, BAST should be given at the lowest effective dose, with periodic trials of on-demand, intermittent administration, the panelists recommended. When BAST is ineffective, the guideline recommends that clinicians review concurrent medications as a possible cause of BAD or reinvestigate.
Concluding the guideline, the panelists emphasized the need for more high-quality research.
“The group recognized that specific, high-certainty evidence was lacking in many areas and recommended further studies that would improve the data available in future methodologic evaluations,” the panelists wrote.
While improving diagnostic accuracy of BAD should be a major goal of such research, progress is currently limited by an integral shortcoming of diagnostic test accuracy (DTA) studies, the panelists wrote.
“The main challenge in conducting DTA studies for BAD is the lack of a widely accepted or universally agreed-upon reference standard because the condition is defined and classified based on pathophysiologic mechanisms and its response to treatment (BAST),” the panelists wrote. “In addition, the index tests (SeHCAT, C4, FGF19, fecal bile acid assay) provide a continuous measure of metabolic function. Hence, DTA studies are not the most appropriate study design.”
“Therefore, one of the research priorities in BAD is for the scientific and clinical communities to agree on a reference standard that best represents BAD (e.g., response to BAST), with full understanding that the reference standard is and likely will be imperfect.”
The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Review the AGA clinical practice guideline on the laboratory evaluation of functional diarrhea and diarrhea-predominan irritable bowel syndrome in adults at https:/www.gastrojournal.org/article/S0016-5085(19)41083-4/fulltext.
SOURCE: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: The Canadian Association of Gastroenterology recently published a clinical practice guideline for the management of bile acid diarrhea (BAD).
Major finding: BAD occurs in up to 35% of patients with chronic diarrhea or diarrhea-predominant irritable bowel syndrome.
Study details: A clinical practice guideline for the management of BAD.
Disclosures: The guideline was funded by unrestricted grants from Pendopharm and GE Healthcare Canada. The panelists disclosed relationships with AstraZeneca, AbbVie, Merck, Pfizer, and others.
Source: Sadowski DC et al. Clin Gastroenterol Hepatol. 2019 Sep 14. doi: 10.1016/j.cgh.2019.08.062.
AGA releases clinical practice update for pancreatic necrosis
The American Gastroenterological Association recently issued a clinical practice update for the management of pancreatic necrosis, including 15 recommendations based on a comprehensive literature review and the experiences of leading experts.
Recommendations range from the general, such as the need for a multidisciplinary approach, to the specific, such as the superiority of metal over plastic stents for endoscopic transmural drainage.
The expert review, which was conducted by lead author Todd H. Baron, MD, of the University of North Carolina in Chapel Hill and three other colleagues, was vetted by the AGA Institute Clinical Practice Updates Committee (CPUC) and the AGA Governing Board. In addition, the update underwent external peer review prior to publication in Gastroenterology.
In the update, the authors outlined the clinical landscape for pancreatic necrosis, including challenges posed by complex cases and a mortality rate as high as 30%.
“Successful management of these patients requires expert multidisciplinary care by gastroenterologists, surgeons, interventional radiologists, and specialists in critical care medicine, infectious disease, and nutrition,” the investigators wrote.
They went on to explain how management has evolved over the past 10 years.
“Whereby major surgical intervention and debridement was once the mainstay of therapy for patients with symptomatic necrotic collections, a minimally invasive approach focusing on percutaneous drainage and/or endoscopic drainage or debridement is now favored,” they wrote. They added that debridement is still generally agreed to be the best choice for cases of infected necrosis or patients with sterile necrosis “marked by abdominal pain, nausea, vomiting, and nutritional failure or with associated complications including gastrointestinal luminal obstruction, biliary obstruction, recurrent acute pancreatitis, fistulas, or persistent systemic inflammatory response syndrome (SIRS).”
Other elements of care, however, remain debated, the investigators noted, which has led to variations in multiple aspects of care, such as interventional timing, intravenous fluids, antibiotics, and nutrition. Within this framework, the present practice update is aimed at offering “concise best practice advice for the optimal management of patients with this highly morbid condition.”
Among these pieces of advice, the authors emphasized that routine prophylactic antibiotics and/or antifungals to prevent infected necrosis are unsupported by multiple clinical trials. When infection is suspected, the update recommends broad spectrum intravenous antibiotics, noting that, in most cases, it is unnecessary to perform CT-guided fine-needle aspiration for cultures and gram stain.
Regarding nutrition, the update recommends against “pancreatic rest”; instead, it calls for early oral intake and, if this is not possible, then initiation of total enteral nutrition. Although the authors deemed multiple routes of enteral feeding acceptable, they favored nasogastric or nasoduodenal tubes, when appropriate, because of ease of placement and maintenance. For prolonged total enteral nutrition or patients unable to tolerate nasoenteric feeding, the authors recommended endoscopic feeding tube placement with a percutaneous endoscopic gastrostomy tube for those who can tolerate gastric feeding or a percutaneous endoscopic jejunostomy tube for those who cannot or have a high risk of aspiration.
As described above, the update recommends debridement for cases of infected pancreatic necrosis. Ideally, this should be performed at least 4 weeks after onset, and avoided altogether within the first 2 weeks, because of associated risks of morbidity and mortality; instead, during this acute phase, percutaneous drainage may be considered.
For walled-off pancreatic necrosis, the authors recommended transmural drainage via endoscopic therapy because this mitigates risk of pancreatocutaneous fistula. Percutaneous drainage may be considered in addition to, or in absence of, endoscopic drainage, depending on clinical status.
The remainder of the update covers decisions related to stents, other minimally invasive techniques, open operative debridement, and disconnected left pancreatic remnants, along with discussions of key supporting clinical trials.
The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
SOURCE: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
The American Gastroenterological Association recently issued a clinical practice update for the management of pancreatic necrosis, including 15 recommendations based on a comprehensive literature review and the experiences of leading experts.
Recommendations range from the general, such as the need for a multidisciplinary approach, to the specific, such as the superiority of metal over plastic stents for endoscopic transmural drainage.
The expert review, which was conducted by lead author Todd H. Baron, MD, of the University of North Carolina in Chapel Hill and three other colleagues, was vetted by the AGA Institute Clinical Practice Updates Committee (CPUC) and the AGA Governing Board. In addition, the update underwent external peer review prior to publication in Gastroenterology.
In the update, the authors outlined the clinical landscape for pancreatic necrosis, including challenges posed by complex cases and a mortality rate as high as 30%.
“Successful management of these patients requires expert multidisciplinary care by gastroenterologists, surgeons, interventional radiologists, and specialists in critical care medicine, infectious disease, and nutrition,” the investigators wrote.
They went on to explain how management has evolved over the past 10 years.
“Whereby major surgical intervention and debridement was once the mainstay of therapy for patients with symptomatic necrotic collections, a minimally invasive approach focusing on percutaneous drainage and/or endoscopic drainage or debridement is now favored,” they wrote. They added that debridement is still generally agreed to be the best choice for cases of infected necrosis or patients with sterile necrosis “marked by abdominal pain, nausea, vomiting, and nutritional failure or with associated complications including gastrointestinal luminal obstruction, biliary obstruction, recurrent acute pancreatitis, fistulas, or persistent systemic inflammatory response syndrome (SIRS).”
Other elements of care, however, remain debated, the investigators noted, which has led to variations in multiple aspects of care, such as interventional timing, intravenous fluids, antibiotics, and nutrition. Within this framework, the present practice update is aimed at offering “concise best practice advice for the optimal management of patients with this highly morbid condition.”
Among these pieces of advice, the authors emphasized that routine prophylactic antibiotics and/or antifungals to prevent infected necrosis are unsupported by multiple clinical trials. When infection is suspected, the update recommends broad spectrum intravenous antibiotics, noting that, in most cases, it is unnecessary to perform CT-guided fine-needle aspiration for cultures and gram stain.
Regarding nutrition, the update recommends against “pancreatic rest”; instead, it calls for early oral intake and, if this is not possible, then initiation of total enteral nutrition. Although the authors deemed multiple routes of enteral feeding acceptable, they favored nasogastric or nasoduodenal tubes, when appropriate, because of ease of placement and maintenance. For prolonged total enteral nutrition or patients unable to tolerate nasoenteric feeding, the authors recommended endoscopic feeding tube placement with a percutaneous endoscopic gastrostomy tube for those who can tolerate gastric feeding or a percutaneous endoscopic jejunostomy tube for those who cannot or have a high risk of aspiration.
As described above, the update recommends debridement for cases of infected pancreatic necrosis. Ideally, this should be performed at least 4 weeks after onset, and avoided altogether within the first 2 weeks, because of associated risks of morbidity and mortality; instead, during this acute phase, percutaneous drainage may be considered.
For walled-off pancreatic necrosis, the authors recommended transmural drainage via endoscopic therapy because this mitigates risk of pancreatocutaneous fistula. Percutaneous drainage may be considered in addition to, or in absence of, endoscopic drainage, depending on clinical status.
The remainder of the update covers decisions related to stents, other minimally invasive techniques, open operative debridement, and disconnected left pancreatic remnants, along with discussions of key supporting clinical trials.
The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
SOURCE: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
The American Gastroenterological Association recently issued a clinical practice update for the management of pancreatic necrosis, including 15 recommendations based on a comprehensive literature review and the experiences of leading experts.
Recommendations range from the general, such as the need for a multidisciplinary approach, to the specific, such as the superiority of metal over plastic stents for endoscopic transmural drainage.
The expert review, which was conducted by lead author Todd H. Baron, MD, of the University of North Carolina in Chapel Hill and three other colleagues, was vetted by the AGA Institute Clinical Practice Updates Committee (CPUC) and the AGA Governing Board. In addition, the update underwent external peer review prior to publication in Gastroenterology.
In the update, the authors outlined the clinical landscape for pancreatic necrosis, including challenges posed by complex cases and a mortality rate as high as 30%.
“Successful management of these patients requires expert multidisciplinary care by gastroenterologists, surgeons, interventional radiologists, and specialists in critical care medicine, infectious disease, and nutrition,” the investigators wrote.
They went on to explain how management has evolved over the past 10 years.
“Whereby major surgical intervention and debridement was once the mainstay of therapy for patients with symptomatic necrotic collections, a minimally invasive approach focusing on percutaneous drainage and/or endoscopic drainage or debridement is now favored,” they wrote. They added that debridement is still generally agreed to be the best choice for cases of infected necrosis or patients with sterile necrosis “marked by abdominal pain, nausea, vomiting, and nutritional failure or with associated complications including gastrointestinal luminal obstruction, biliary obstruction, recurrent acute pancreatitis, fistulas, or persistent systemic inflammatory response syndrome (SIRS).”
Other elements of care, however, remain debated, the investigators noted, which has led to variations in multiple aspects of care, such as interventional timing, intravenous fluids, antibiotics, and nutrition. Within this framework, the present practice update is aimed at offering “concise best practice advice for the optimal management of patients with this highly morbid condition.”
Among these pieces of advice, the authors emphasized that routine prophylactic antibiotics and/or antifungals to prevent infected necrosis are unsupported by multiple clinical trials. When infection is suspected, the update recommends broad spectrum intravenous antibiotics, noting that, in most cases, it is unnecessary to perform CT-guided fine-needle aspiration for cultures and gram stain.
Regarding nutrition, the update recommends against “pancreatic rest”; instead, it calls for early oral intake and, if this is not possible, then initiation of total enteral nutrition. Although the authors deemed multiple routes of enteral feeding acceptable, they favored nasogastric or nasoduodenal tubes, when appropriate, because of ease of placement and maintenance. For prolonged total enteral nutrition or patients unable to tolerate nasoenteric feeding, the authors recommended endoscopic feeding tube placement with a percutaneous endoscopic gastrostomy tube for those who can tolerate gastric feeding or a percutaneous endoscopic jejunostomy tube for those who cannot or have a high risk of aspiration.
As described above, the update recommends debridement for cases of infected pancreatic necrosis. Ideally, this should be performed at least 4 weeks after onset, and avoided altogether within the first 2 weeks, because of associated risks of morbidity and mortality; instead, during this acute phase, percutaneous drainage may be considered.
For walled-off pancreatic necrosis, the authors recommended transmural drainage via endoscopic therapy because this mitigates risk of pancreatocutaneous fistula. Percutaneous drainage may be considered in addition to, or in absence of, endoscopic drainage, depending on clinical status.
The remainder of the update covers decisions related to stents, other minimally invasive techniques, open operative debridement, and disconnected left pancreatic remnants, along with discussions of key supporting clinical trials.
The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
SOURCE: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
FROM GASTROENTEROLOGY
Key clinical point: The American Gastroenterological Association has issued a clinical practice update for the management of pancreatic necrosis.
Major finding: N/A
Study details: A clinical practice update for the management of pancreatic necrosis.
Disclosures: The investigators disclosed relationships with Cook Endoscopy, Boston Scientific, Olympus, and others.
Source: Baron TH et al. Gastroenterology. 2019 Aug 31. doi: 10.1053/j.gastro.2019.07.064.
Guideline: Diagnosis and treatment of adults with community-acquired pneumonia
A new guideline has been published to update the 2007 guidelines for the management of adults with community-acquired pneumonia (CAP).
The practice guideline was jointly written by an ad hoc committee of the American Thoracic Society and Infectious Diseases Society of America. CAP refers to a pneumonia infection that was acquired by a patient in his or her community. Decisions about which antibiotics to use to treat this kind of infection are based on risk factors for resistant organisms and the severity of illness.
Pathogens
Traditionally, CAP is caused by common bacterial pathogens that include Streptococcus pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae, Staphylococcus aureus, Legionella species, Chlamydia pneumonia, and Moraxella catarrhalis. Risk factors for multidrug resistant pathogens such as methicillin-resistant S. aureus (MRSA) and Pseudomonas aeruginosa include previous infection with MRSA or P. aeruginosa, recent hospitalization, and requiring parenteral antibiotics in the last 90 days.
Defining severe community-acquired pneumonia
The health care–associated pneumonia, or HCAP, classification should no longer be used to determine empiric treatment. The recommendations for which antibiotics to use are linked to the severity of illness. Previously the site of treatment drove antibiotic selection, but since decision about the site of care can be affected by many considerations, the guidelines recommend using the CAP severity criteria. Severe CAP includes either one major or at least three minor criteria.
Major criteria are:
- Septic shock requiring vasopressors.
- Respiratory failure requiring mechanical ventilation.
Minor criteria are:
- Respiratory rate greater than or equal to 30 breaths/min.
- Ratio of arterial O2 partial pressure to fractional inspired O2 less than or equal to 250.
- Multilobar infiltrates.
- Confusion/disorientation.
- Uremia (blood urea nitrogen level greater than or equal to 20 mg/dL).
- Leukopenia (white blood cell count less than 4,000 cells/mcL).
- Thrombocytopenia (platelet count less than 100,000 mcL)
- Hypothermia (core temperature less than 36º C).
- Hypotension requiring aggressive fluid resuscitation.
Management and diagnostic testing
Clinicians should use the Pneumonia Severity Index (PSI) and clinical judgment to guide the site of treatment for patients. Gram stain, sputum, and blood culture should not be routinely obtained in an outpatient setting. Legionella antigen should not be routinely obtained unless indicated by epidemiological factors. During influenza season, a rapid influenza assay, preferably a nucleic acid amplification test, should be obtained to help guide treatment.
For patients with severe CAP or risk factors for MRSA or P. aeruginosa, gram stain and culture and Legionella antigen should be obtained to manage antibiotic choices. Also, blood cultures should be obtained for these patients.
Empiric antibiotic therapy should be initiated based on clinical judgment and radiographic confirmation of CAP. Serum procalcitonin should not be used to assess initiation of antibiotic therapy.
Empiric antibiotic therapy
Healthy adults without comorbidities should be treated with monotherapy of either:
- Amoxicillin 1 g three times daily.
- OR doxycycline 100 mg twice daily.
- OR a macrolide (azithromycin 500 mg on first day then 250 mg daily or clarithromycin 500 mg twice daily or clarithromycin extended release 1,000 mg daily) only in areas with pneumococcal resistance to macrolides less than 25%.
Adults with comorbidities such as chronic heart, lung, liver, or renal disease; diabetes mellitus; alcoholism; malignancy; or asplenia should be treated with:
- Amoxicillin/clavulanate 500 mg/125 mg three times daily, or amoxicillin/ clavulanate 875 mg/125 mg twice daily, or 2,000 mg/125 mg twice daily, or a cephalosporin (cefpodoxime 200 mg twice daily or cefuroxime 500 mg twice daily); and a macrolide (azithromycin 500 mg on first day then 250 mg daily, clarithromycin [500 mg twice daily or extended release 1,000 mg once daily]), or doxycycline 100 mg twice daily. (Some experts recommend that the first dose of doxycycline should be 200 mg.)
- OR monotherapy with respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily, or gemifloxacin 320 mg daily).
Inpatient pneumonia that is not severe, without risk factors for resistant organisms should be treated with:
- Beta-lactam (ampicillin 1 sulbactam 1.5-3 g every 6 h, cefotaxime 1-2 g every 8 h, ceftriaxone 1-2 g daily, or ceftaroline 600 mg every 12 h) and a macrolide (azithromycin 500 mg daily or clarithromycin 500 mg twice daily).
- OR monotherapy with a respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily).
If there is a contraindication for the use of both a macrolide and a fluoroquinolone, then doxycycline can be used instead.
Severe inpatient pneumonia without risk factors for resistant organisms should be treated with combination therapy of either (agents and doses the same as above):
- Beta-lactam and macrolide.
- OR fluoroquinolone and beta-lactam.
It is recommended to not routinely add anaerobic coverage for suspected aspiration pneumonia unless lung abscess or empyema is suspected. Clinicians should identify risk factors for MRSA or P. aeruginosa before adding additional agents.
Duration of antibiotic therapy is determined by the patient achieving clinical stability with no less than 5 days of antibiotics. In adults with symptom resolution within 5-7 days, no additional follow-up chest imaging is recommended. If patients test positive for influenza, then anti-influenza treatment such as oseltamivir should be used in addition to antibiotics regardless of length of influenza symptoms before presentation.
The bottom line
CAP treatment should be based on severity of illness and risk factors for resistant organisms. Blood and sputum cultures are recommended only for patients with severe pneumonia. There have been important changes in the recommendations for antibiotic treatment of CAP, with high-dose amoxicillin recommended for most patients with CAP who are treated as outpatients. Patients who exhibit clinical stability should be treated for at least 5 days and do not require follow up imaging studies.
For a podcast of this guideline, go to iTunes and download the Infectious Diseases Society of America guideline podcast.
Reference
Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-e67.
Tina Chuong, DO, is a second-year resident in the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Skolnik is professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington Jefferson Health.
A new guideline has been published to update the 2007 guidelines for the management of adults with community-acquired pneumonia (CAP).
The practice guideline was jointly written by an ad hoc committee of the American Thoracic Society and Infectious Diseases Society of America. CAP refers to a pneumonia infection that was acquired by a patient in his or her community. Decisions about which antibiotics to use to treat this kind of infection are based on risk factors for resistant organisms and the severity of illness.
Pathogens
Traditionally, CAP is caused by common bacterial pathogens that include Streptococcus pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae, Staphylococcus aureus, Legionella species, Chlamydia pneumonia, and Moraxella catarrhalis. Risk factors for multidrug resistant pathogens such as methicillin-resistant S. aureus (MRSA) and Pseudomonas aeruginosa include previous infection with MRSA or P. aeruginosa, recent hospitalization, and requiring parenteral antibiotics in the last 90 days.
Defining severe community-acquired pneumonia
The health care–associated pneumonia, or HCAP, classification should no longer be used to determine empiric treatment. The recommendations for which antibiotics to use are linked to the severity of illness. Previously the site of treatment drove antibiotic selection, but since decision about the site of care can be affected by many considerations, the guidelines recommend using the CAP severity criteria. Severe CAP includes either one major or at least three minor criteria.
Major criteria are:
- Septic shock requiring vasopressors.
- Respiratory failure requiring mechanical ventilation.
Minor criteria are:
- Respiratory rate greater than or equal to 30 breaths/min.
- Ratio of arterial O2 partial pressure to fractional inspired O2 less than or equal to 250.
- Multilobar infiltrates.
- Confusion/disorientation.
- Uremia (blood urea nitrogen level greater than or equal to 20 mg/dL).
- Leukopenia (white blood cell count less than 4,000 cells/mcL).
- Thrombocytopenia (platelet count less than 100,000 mcL)
- Hypothermia (core temperature less than 36º C).
- Hypotension requiring aggressive fluid resuscitation.
Management and diagnostic testing
Clinicians should use the Pneumonia Severity Index (PSI) and clinical judgment to guide the site of treatment for patients. Gram stain, sputum, and blood culture should not be routinely obtained in an outpatient setting. Legionella antigen should not be routinely obtained unless indicated by epidemiological factors. During influenza season, a rapid influenza assay, preferably a nucleic acid amplification test, should be obtained to help guide treatment.
For patients with severe CAP or risk factors for MRSA or P. aeruginosa, gram stain and culture and Legionella antigen should be obtained to manage antibiotic choices. Also, blood cultures should be obtained for these patients.
Empiric antibiotic therapy should be initiated based on clinical judgment and radiographic confirmation of CAP. Serum procalcitonin should not be used to assess initiation of antibiotic therapy.
Empiric antibiotic therapy
Healthy adults without comorbidities should be treated with monotherapy of either:
- Amoxicillin 1 g three times daily.
- OR doxycycline 100 mg twice daily.
- OR a macrolide (azithromycin 500 mg on first day then 250 mg daily or clarithromycin 500 mg twice daily or clarithromycin extended release 1,000 mg daily) only in areas with pneumococcal resistance to macrolides less than 25%.
Adults with comorbidities such as chronic heart, lung, liver, or renal disease; diabetes mellitus; alcoholism; malignancy; or asplenia should be treated with:
- Amoxicillin/clavulanate 500 mg/125 mg three times daily, or amoxicillin/ clavulanate 875 mg/125 mg twice daily, or 2,000 mg/125 mg twice daily, or a cephalosporin (cefpodoxime 200 mg twice daily or cefuroxime 500 mg twice daily); and a macrolide (azithromycin 500 mg on first day then 250 mg daily, clarithromycin [500 mg twice daily or extended release 1,000 mg once daily]), or doxycycline 100 mg twice daily. (Some experts recommend that the first dose of doxycycline should be 200 mg.)
- OR monotherapy with respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily, or gemifloxacin 320 mg daily).
Inpatient pneumonia that is not severe, without risk factors for resistant organisms should be treated with:
- Beta-lactam (ampicillin 1 sulbactam 1.5-3 g every 6 h, cefotaxime 1-2 g every 8 h, ceftriaxone 1-2 g daily, or ceftaroline 600 mg every 12 h) and a macrolide (azithromycin 500 mg daily or clarithromycin 500 mg twice daily).
- OR monotherapy with a respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily).
If there is a contraindication for the use of both a macrolide and a fluoroquinolone, then doxycycline can be used instead.
Severe inpatient pneumonia without risk factors for resistant organisms should be treated with combination therapy of either (agents and doses the same as above):
- Beta-lactam and macrolide.
- OR fluoroquinolone and beta-lactam.
It is recommended to not routinely add anaerobic coverage for suspected aspiration pneumonia unless lung abscess or empyema is suspected. Clinicians should identify risk factors for MRSA or P. aeruginosa before adding additional agents.
Duration of antibiotic therapy is determined by the patient achieving clinical stability with no less than 5 days of antibiotics. In adults with symptom resolution within 5-7 days, no additional follow-up chest imaging is recommended. If patients test positive for influenza, then anti-influenza treatment such as oseltamivir should be used in addition to antibiotics regardless of length of influenza symptoms before presentation.
The bottom line
CAP treatment should be based on severity of illness and risk factors for resistant organisms. Blood and sputum cultures are recommended only for patients with severe pneumonia. There have been important changes in the recommendations for antibiotic treatment of CAP, with high-dose amoxicillin recommended for most patients with CAP who are treated as outpatients. Patients who exhibit clinical stability should be treated for at least 5 days and do not require follow up imaging studies.
For a podcast of this guideline, go to iTunes and download the Infectious Diseases Society of America guideline podcast.
Reference
Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-e67.
Tina Chuong, DO, is a second-year resident in the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Skolnik is professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington Jefferson Health.
A new guideline has been published to update the 2007 guidelines for the management of adults with community-acquired pneumonia (CAP).
The practice guideline was jointly written by an ad hoc committee of the American Thoracic Society and Infectious Diseases Society of America. CAP refers to a pneumonia infection that was acquired by a patient in his or her community. Decisions about which antibiotics to use to treat this kind of infection are based on risk factors for resistant organisms and the severity of illness.
Pathogens
Traditionally, CAP is caused by common bacterial pathogens that include Streptococcus pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae, Staphylococcus aureus, Legionella species, Chlamydia pneumonia, and Moraxella catarrhalis. Risk factors for multidrug resistant pathogens such as methicillin-resistant S. aureus (MRSA) and Pseudomonas aeruginosa include previous infection with MRSA or P. aeruginosa, recent hospitalization, and requiring parenteral antibiotics in the last 90 days.
Defining severe community-acquired pneumonia
The health care–associated pneumonia, or HCAP, classification should no longer be used to determine empiric treatment. The recommendations for which antibiotics to use are linked to the severity of illness. Previously the site of treatment drove antibiotic selection, but since decision about the site of care can be affected by many considerations, the guidelines recommend using the CAP severity criteria. Severe CAP includes either one major or at least three minor criteria.
Major criteria are:
- Septic shock requiring vasopressors.
- Respiratory failure requiring mechanical ventilation.
Minor criteria are:
- Respiratory rate greater than or equal to 30 breaths/min.
- Ratio of arterial O2 partial pressure to fractional inspired O2 less than or equal to 250.
- Multilobar infiltrates.
- Confusion/disorientation.
- Uremia (blood urea nitrogen level greater than or equal to 20 mg/dL).
- Leukopenia (white blood cell count less than 4,000 cells/mcL).
- Thrombocytopenia (platelet count less than 100,000 mcL)
- Hypothermia (core temperature less than 36º C).
- Hypotension requiring aggressive fluid resuscitation.
Management and diagnostic testing
Clinicians should use the Pneumonia Severity Index (PSI) and clinical judgment to guide the site of treatment for patients. Gram stain, sputum, and blood culture should not be routinely obtained in an outpatient setting. Legionella antigen should not be routinely obtained unless indicated by epidemiological factors. During influenza season, a rapid influenza assay, preferably a nucleic acid amplification test, should be obtained to help guide treatment.
For patients with severe CAP or risk factors for MRSA or P. aeruginosa, gram stain and culture and Legionella antigen should be obtained to manage antibiotic choices. Also, blood cultures should be obtained for these patients.
Empiric antibiotic therapy should be initiated based on clinical judgment and radiographic confirmation of CAP. Serum procalcitonin should not be used to assess initiation of antibiotic therapy.
Empiric antibiotic therapy
Healthy adults without comorbidities should be treated with monotherapy of either:
- Amoxicillin 1 g three times daily.
- OR doxycycline 100 mg twice daily.
- OR a macrolide (azithromycin 500 mg on first day then 250 mg daily or clarithromycin 500 mg twice daily or clarithromycin extended release 1,000 mg daily) only in areas with pneumococcal resistance to macrolides less than 25%.
Adults with comorbidities such as chronic heart, lung, liver, or renal disease; diabetes mellitus; alcoholism; malignancy; or asplenia should be treated with:
- Amoxicillin/clavulanate 500 mg/125 mg three times daily, or amoxicillin/ clavulanate 875 mg/125 mg twice daily, or 2,000 mg/125 mg twice daily, or a cephalosporin (cefpodoxime 200 mg twice daily or cefuroxime 500 mg twice daily); and a macrolide (azithromycin 500 mg on first day then 250 mg daily, clarithromycin [500 mg twice daily or extended release 1,000 mg once daily]), or doxycycline 100 mg twice daily. (Some experts recommend that the first dose of doxycycline should be 200 mg.)
- OR monotherapy with respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily, or gemifloxacin 320 mg daily).
Inpatient pneumonia that is not severe, without risk factors for resistant organisms should be treated with:
- Beta-lactam (ampicillin 1 sulbactam 1.5-3 g every 6 h, cefotaxime 1-2 g every 8 h, ceftriaxone 1-2 g daily, or ceftaroline 600 mg every 12 h) and a macrolide (azithromycin 500 mg daily or clarithromycin 500 mg twice daily).
- OR monotherapy with a respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily).
If there is a contraindication for the use of both a macrolide and a fluoroquinolone, then doxycycline can be used instead.
Severe inpatient pneumonia without risk factors for resistant organisms should be treated with combination therapy of either (agents and doses the same as above):
- Beta-lactam and macrolide.
- OR fluoroquinolone and beta-lactam.
It is recommended to not routinely add anaerobic coverage for suspected aspiration pneumonia unless lung abscess or empyema is suspected. Clinicians should identify risk factors for MRSA or P. aeruginosa before adding additional agents.
Duration of antibiotic therapy is determined by the patient achieving clinical stability with no less than 5 days of antibiotics. In adults with symptom resolution within 5-7 days, no additional follow-up chest imaging is recommended. If patients test positive for influenza, then anti-influenza treatment such as oseltamivir should be used in addition to antibiotics regardless of length of influenza symptoms before presentation.
The bottom line
CAP treatment should be based on severity of illness and risk factors for resistant organisms. Blood and sputum cultures are recommended only for patients with severe pneumonia. There have been important changes in the recommendations for antibiotic treatment of CAP, with high-dose amoxicillin recommended for most patients with CAP who are treated as outpatients. Patients who exhibit clinical stability should be treated for at least 5 days and do not require follow up imaging studies.
For a podcast of this guideline, go to iTunes and download the Infectious Diseases Society of America guideline podcast.
Reference
Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-e67.
Tina Chuong, DO, is a second-year resident in the family medicine residency program at Abington (Pa.) Jefferson Health. Dr. Skolnik is professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington Jefferson Health.
Inpatient care declining among family physicians
and by 2017, only one of four FPs was practicing hospital medicine, according to the American Academy of Family Physicians.
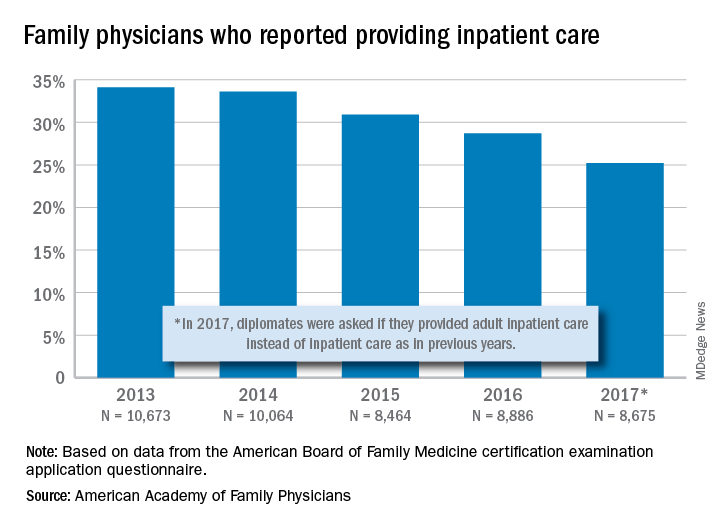
The share of family physicians who provided hospital care went from 34.1% in 2013 to 25.2% in 2017, for a relative decrease of 26% that left only a quarter of FPs seeing inpatients, based on data from the annual American Board of Family Medicine certification exam application questionnaire. For the 5-year period, 46,762 individuals were included in the study sample of FPs in direct patient care.
“As observed in other domains (prenatal care, home visits, nursing home care, and obstetric care), this study adds to the evidence demonstrating contracting scope of practice among FPs,” Anuradha Jetty, MPH, of the AAFP’s Robert Graham Center in Washington, D.C., and associates said in a recent Policy Brief published in the Journal of the American Board of Family Medicine.
Much of that contraction is occurring among new family physicians who can’t “find positions that allow them to use all their expertise,” the investigators said in a separate statement. The AAFP had previously reported that about 40% of family physicians had full hospital privileges in 2018, compared with 56% in 2012.
Many new FPs now work in large multispecialty practices or hospital systems, and “[some] of these employers dictate scope of practice, limiting family physicians to coordinating outpatient care and relying on subspecialists or hospitalists to provide inpatient care,” they noted.
and by 2017, only one of four FPs was practicing hospital medicine, according to the American Academy of Family Physicians.

The share of family physicians who provided hospital care went from 34.1% in 2013 to 25.2% in 2017, for a relative decrease of 26% that left only a quarter of FPs seeing inpatients, based on data from the annual American Board of Family Medicine certification exam application questionnaire. For the 5-year period, 46,762 individuals were included in the study sample of FPs in direct patient care.
“As observed in other domains (prenatal care, home visits, nursing home care, and obstetric care), this study adds to the evidence demonstrating contracting scope of practice among FPs,” Anuradha Jetty, MPH, of the AAFP’s Robert Graham Center in Washington, D.C., and associates said in a recent Policy Brief published in the Journal of the American Board of Family Medicine.
Much of that contraction is occurring among new family physicians who can’t “find positions that allow them to use all their expertise,” the investigators said in a separate statement. The AAFP had previously reported that about 40% of family physicians had full hospital privileges in 2018, compared with 56% in 2012.
Many new FPs now work in large multispecialty practices or hospital systems, and “[some] of these employers dictate scope of practice, limiting family physicians to coordinating outpatient care and relying on subspecialists or hospitalists to provide inpatient care,” they noted.
and by 2017, only one of four FPs was practicing hospital medicine, according to the American Academy of Family Physicians.

The share of family physicians who provided hospital care went from 34.1% in 2013 to 25.2% in 2017, for a relative decrease of 26% that left only a quarter of FPs seeing inpatients, based on data from the annual American Board of Family Medicine certification exam application questionnaire. For the 5-year period, 46,762 individuals were included in the study sample of FPs in direct patient care.
“As observed in other domains (prenatal care, home visits, nursing home care, and obstetric care), this study adds to the evidence demonstrating contracting scope of practice among FPs,” Anuradha Jetty, MPH, of the AAFP’s Robert Graham Center in Washington, D.C., and associates said in a recent Policy Brief published in the Journal of the American Board of Family Medicine.
Much of that contraction is occurring among new family physicians who can’t “find positions that allow them to use all their expertise,” the investigators said in a separate statement. The AAFP had previously reported that about 40% of family physicians had full hospital privileges in 2018, compared with 56% in 2012.
Many new FPs now work in large multispecialty practices or hospital systems, and “[some] of these employers dictate scope of practice, limiting family physicians to coordinating outpatient care and relying on subspecialists or hospitalists to provide inpatient care,” they noted.
AAD-NPF Pediatric psoriasis guideline advises on physical and mental care
Psoriasis management in children involves attention not only to treatment of the physical condition but also psychosocial wellness and quality of life, according to
Psoriasis affects approximately 1% of children, either alone or associated with comorbid conditions such as psoriatic arthritis (PsA), wrote Alan Menter, MD, of Baylor University Medical Center, Dallas, and coauthors of the guideline.
In the guideline, published in the Journal of the American Academy of Dermatology, the multidisciplinary work group identified screening tools to measure disease severity, strategies for management of comorbidities, and the safety and effectiveness of topical, systemic, and phototherapy treatments.
To assess disease severity, the work group recommended not only the use of body surface area (BSA), similar to measurement of severity in adults, but also the use of the Children’s Dermatology Life Quality Index, a 10-question quality of life survey, as BSA alone does not account for the potential negative impact of the disease on quality of life in terms of physical, emotional, social, and psychological function.
“For example, a child with psoriasis limited to the face or the entire scalp does not have severe disease based on BSA definitions, but if this involvement causes shame, social withdrawal, or bullying, it satisfies criteria for severe disease based on impact beyond the skin,” they said.
The work group stated that a variety of conditions may trigger or exacerbate psoriasis in children, including infections, cutaneous trauma, or physiological, emotional, and environmental stressors.
The majority of children with PsA develop joint inflammation before skin disease, the work group wrote. In addition, children with psoriasis are at increased risk for rheumatoid arthritis, so clinicians may need to distinguish between a combination of psoriasis and musculoskeletal issues and cases of either psoriatic or rheumatoid arthritis in young patients.
The cardiovascular risk factors associated with metabolic syndrome are greater in children with psoriasis, compared with children without psoriasis, the work group noted. In addition, pediatric psoriasis patients have a higher prevalence of obesity than children without psoriasis, and they recommended that children with psoriasis be monitored for the development of obesity, and that obese children with psoriasis should be referred for weight management.
The work group noted that data are insufficient in children to support the link between psoriasis and cardiovascular disease that has been documented in adults with psoriasis. However, “patients with pediatric psoriasis should have American Academy of Pediatrics (AAP)–recommended age-related cardiovascular screening regardless of the presence of signs or symptoms,” they said.
The guideline also recommends screening for dyslipidemia and hypertension according to AAP guidelines and educating pediatric psoriasis patients about the risk of diabetes and regularly screening for diabetes and insulin resistance in those who are obese. Overweight children with psoriasis may be screened at the provider’s discretion, they wrote. Patients with signs of inflammatory bowel disease, which also is associated with psoriasis in adults, should be considered for referral to a gastroenterologist, they noted.
Children with psoriasis should be screened regularly for mental health conditions regardless of age, and they should be asked about substance abuse, according to the guideline, and those with concerns should be referred for additional assessment and management.
The guideline divides treatment of psoriasis in children into three categories: topical, phototherapy and photochemotherapy, and systemic treatments (nonbiologic or biologic).
For topicals, the guideline recommendations include corticosteroids as an off-label therapy, as well as ultra-high-potency topical corticosteroids as monotherapy. Overall, “selection of a therapeutic routine (potency, delivery vehicle, frequency of application) should take into account sites of involvement, type and thickness of psoriasis, age of the patient, total BSA of application, anticipated occlusion, and disease acuity, among other patient-, disease-, and drug-related factors,” the authors wrote. Other topical options included in the recommendations: calcineurin inhibitors, topical vitamin D analogues, tazarotene (off label), anthralin, and coal tar.
Phototherapy has a history of use in psoriasis treatment and remains part of the current recommendations, although data in children are limited, and data on the use of phototherapy for pustular psoriasis in children are insufficient to make specific treatment and dosing recommendations, the work group noted. The researchers also noted that in-office phototherapy may not be feasible for many patients, but that in-home ultraviolet light equipment or natural sunlight in moderation could be recommended as an alternative.
The use of systemic, nonbiologic treatments for pediatric psoriasis should be “based on baseline severity of disease, subtype of psoriasis, speed of disease progression, lack of response to more conservative therapies such as topical agents and phototherapy (when appropriate), impaired physical or psychological functioning or [quality of life] due to disease extent, and the presence of comorbidities such as PsA,” the workgroup said.
Options for systemic treatment include methotrexate, cyclosporine (notably for pustular as well as plaque and erythrodermic psoriasis), and systemic retinoids. In addition, fumaric acid esters may be an option for children with moderate to severe psoriasis, with recommended clinical and laboratory monitoring.
The increasing safety and efficacy data on biologics in pediatric psoriasis patients support their consideration among first-line systemic treatments, the work group suggested. “Etanercept and ustekinumab are now [Food and Drug Administration] approved for patients with psoriasis 4 years and older and 12 years and older, respectively,” they said, and infliximab and adalimumab have been used off label in children.
The work group concluded that research and knowledge gaps about pediatric psoriasis persist and include mechanism of disease onset, development of comorbidities, and identification of ideal dosing for various treatments.
Finally, the work group emphasized the importance of collaboration between dermatologists and primary care providers for managing psoriasis in children, as well as the importance of patient education.
“Dermatologists should be mindful of the unique aspects of the emotional development of children and the social dynamics of having a visible difference,” they wrote. “Shared decision making with the patient (if age appropriate) and the caregivers is a useful approach, particularly as related to the use of off-label medications to treat severe disease,” they said.
“This is the first time that pediatric psoriasis has been discussed as an independent topic within the guideline,” said one of the guideline authors, Dawn M.R. Davis, MD, of the Mayo Clinic, Rochester, Minn., in an interview. “Children have unique physiology and psychosocial aspects to their care relative to adults. In addition, psoriasis has some clinical manifestations that are oftentimes distinctly seen in children,” she commented. “Creation of a guideline specific to children allows us to summarize the similarities and differences of disease presentation and management. It also allows an opportunity to clarify what research data (especially therapeutics) have been studied in children and their uses, safety profiles, and dosing,” she noted.
Psoriasis can be a psychosocially debilitating disease, she emphasized. “In children, for example, isolated or prominent facial involvement is common, which can be embarrassing and impact relationships.”
The take-home message for clinicians, Dr. Davis said, is to keep in mind the multisystemic nature of psoriasis. “It is not limited to the skin,” she said. “Treating a patient with psoriasis necessitates practicing whole-person care” and considering the multiple comorbidities that impact quality of life and overall health in children, as well as adults with psoriasis, she commented. “Dermatologists can empower patients and their caregivers by educating them on the multifocal, complex nature of the disease.” She added, “We have much to learn regarding psoriasis in the pediatric population. More research into therapeutics, topical and systemic, is necessary to optimize patient care.”
The guideline was based on studies published in the PubMed and MEDLINE databases from January 2011 through December 31, 2017.
Dr. Menter and Craig A. Elmets, MD, professor of dermatology, at the University of Alabama, Birmingham, were cochairs of the work group. The pediatric guideline is the latest in a multipart series of AAD-NPF guidelines on psoriasis being published this year in the Journal of the American Academy of Dermatology.
Many of the guideline authors, including lead author Dr. Menter, disclosed relationships with multiple companies; however, a minimum 51% of workgroup members had no relevant conflicts of interest in accordance with AAD policy. There was no funding source. Dr. Davis disclosed serving as an investigator for Regeneron, with no compensation.
SOURCE: Menter et al. J Am Acad Dermatol. 2019. doi: 10.1016/j.jaad.2019.08.049.
Psoriasis management in children involves attention not only to treatment of the physical condition but also psychosocial wellness and quality of life, according to
Psoriasis affects approximately 1% of children, either alone or associated with comorbid conditions such as psoriatic arthritis (PsA), wrote Alan Menter, MD, of Baylor University Medical Center, Dallas, and coauthors of the guideline.
In the guideline, published in the Journal of the American Academy of Dermatology, the multidisciplinary work group identified screening tools to measure disease severity, strategies for management of comorbidities, and the safety and effectiveness of topical, systemic, and phototherapy treatments.
To assess disease severity, the work group recommended not only the use of body surface area (BSA), similar to measurement of severity in adults, but also the use of the Children’s Dermatology Life Quality Index, a 10-question quality of life survey, as BSA alone does not account for the potential negative impact of the disease on quality of life in terms of physical, emotional, social, and psychological function.
“For example, a child with psoriasis limited to the face or the entire scalp does not have severe disease based on BSA definitions, but if this involvement causes shame, social withdrawal, or bullying, it satisfies criteria for severe disease based on impact beyond the skin,” they said.
The work group stated that a variety of conditions may trigger or exacerbate psoriasis in children, including infections, cutaneous trauma, or physiological, emotional, and environmental stressors.
The majority of children with PsA develop joint inflammation before skin disease, the work group wrote. In addition, children with psoriasis are at increased risk for rheumatoid arthritis, so clinicians may need to distinguish between a combination of psoriasis and musculoskeletal issues and cases of either psoriatic or rheumatoid arthritis in young patients.
The cardiovascular risk factors associated with metabolic syndrome are greater in children with psoriasis, compared with children without psoriasis, the work group noted. In addition, pediatric psoriasis patients have a higher prevalence of obesity than children without psoriasis, and they recommended that children with psoriasis be monitored for the development of obesity, and that obese children with psoriasis should be referred for weight management.
The work group noted that data are insufficient in children to support the link between psoriasis and cardiovascular disease that has been documented in adults with psoriasis. However, “patients with pediatric psoriasis should have American Academy of Pediatrics (AAP)–recommended age-related cardiovascular screening regardless of the presence of signs or symptoms,” they said.
The guideline also recommends screening for dyslipidemia and hypertension according to AAP guidelines and educating pediatric psoriasis patients about the risk of diabetes and regularly screening for diabetes and insulin resistance in those who are obese. Overweight children with psoriasis may be screened at the provider’s discretion, they wrote. Patients with signs of inflammatory bowel disease, which also is associated with psoriasis in adults, should be considered for referral to a gastroenterologist, they noted.
Children with psoriasis should be screened regularly for mental health conditions regardless of age, and they should be asked about substance abuse, according to the guideline, and those with concerns should be referred for additional assessment and management.
The guideline divides treatment of psoriasis in children into three categories: topical, phototherapy and photochemotherapy, and systemic treatments (nonbiologic or biologic).
For topicals, the guideline recommendations include corticosteroids as an off-label therapy, as well as ultra-high-potency topical corticosteroids as monotherapy. Overall, “selection of a therapeutic routine (potency, delivery vehicle, frequency of application) should take into account sites of involvement, type and thickness of psoriasis, age of the patient, total BSA of application, anticipated occlusion, and disease acuity, among other patient-, disease-, and drug-related factors,” the authors wrote. Other topical options included in the recommendations: calcineurin inhibitors, topical vitamin D analogues, tazarotene (off label), anthralin, and coal tar.
Phototherapy has a history of use in psoriasis treatment and remains part of the current recommendations, although data in children are limited, and data on the use of phototherapy for pustular psoriasis in children are insufficient to make specific treatment and dosing recommendations, the work group noted. The researchers also noted that in-office phototherapy may not be feasible for many patients, but that in-home ultraviolet light equipment or natural sunlight in moderation could be recommended as an alternative.
The use of systemic, nonbiologic treatments for pediatric psoriasis should be “based on baseline severity of disease, subtype of psoriasis, speed of disease progression, lack of response to more conservative therapies such as topical agents and phototherapy (when appropriate), impaired physical or psychological functioning or [quality of life] due to disease extent, and the presence of comorbidities such as PsA,” the workgroup said.
Options for systemic treatment include methotrexate, cyclosporine (notably for pustular as well as plaque and erythrodermic psoriasis), and systemic retinoids. In addition, fumaric acid esters may be an option for children with moderate to severe psoriasis, with recommended clinical and laboratory monitoring.
The increasing safety and efficacy data on biologics in pediatric psoriasis patients support their consideration among first-line systemic treatments, the work group suggested. “Etanercept and ustekinumab are now [Food and Drug Administration] approved for patients with psoriasis 4 years and older and 12 years and older, respectively,” they said, and infliximab and adalimumab have been used off label in children.
The work group concluded that research and knowledge gaps about pediatric psoriasis persist and include mechanism of disease onset, development of comorbidities, and identification of ideal dosing for various treatments.
Finally, the work group emphasized the importance of collaboration between dermatologists and primary care providers for managing psoriasis in children, as well as the importance of patient education.
“Dermatologists should be mindful of the unique aspects of the emotional development of children and the social dynamics of having a visible difference,” they wrote. “Shared decision making with the patient (if age appropriate) and the caregivers is a useful approach, particularly as related to the use of off-label medications to treat severe disease,” they said.
“This is the first time that pediatric psoriasis has been discussed as an independent topic within the guideline,” said one of the guideline authors, Dawn M.R. Davis, MD, of the Mayo Clinic, Rochester, Minn., in an interview. “Children have unique physiology and psychosocial aspects to their care relative to adults. In addition, psoriasis has some clinical manifestations that are oftentimes distinctly seen in children,” she commented. “Creation of a guideline specific to children allows us to summarize the similarities and differences of disease presentation and management. It also allows an opportunity to clarify what research data (especially therapeutics) have been studied in children and their uses, safety profiles, and dosing,” she noted.
Psoriasis can be a psychosocially debilitating disease, she emphasized. “In children, for example, isolated or prominent facial involvement is common, which can be embarrassing and impact relationships.”
The take-home message for clinicians, Dr. Davis said, is to keep in mind the multisystemic nature of psoriasis. “It is not limited to the skin,” she said. “Treating a patient with psoriasis necessitates practicing whole-person care” and considering the multiple comorbidities that impact quality of life and overall health in children, as well as adults with psoriasis, she commented. “Dermatologists can empower patients and their caregivers by educating them on the multifocal, complex nature of the disease.” She added, “We have much to learn regarding psoriasis in the pediatric population. More research into therapeutics, topical and systemic, is necessary to optimize patient care.”
The guideline was based on studies published in the PubMed and MEDLINE databases from January 2011 through December 31, 2017.
Dr. Menter and Craig A. Elmets, MD, professor of dermatology, at the University of Alabama, Birmingham, were cochairs of the work group. The pediatric guideline is the latest in a multipart series of AAD-NPF guidelines on psoriasis being published this year in the Journal of the American Academy of Dermatology.
Many of the guideline authors, including lead author Dr. Menter, disclosed relationships with multiple companies; however, a minimum 51% of workgroup members had no relevant conflicts of interest in accordance with AAD policy. There was no funding source. Dr. Davis disclosed serving as an investigator for Regeneron, with no compensation.
SOURCE: Menter et al. J Am Acad Dermatol. 2019. doi: 10.1016/j.jaad.2019.08.049.
Psoriasis management in children involves attention not only to treatment of the physical condition but also psychosocial wellness and quality of life, according to
Psoriasis affects approximately 1% of children, either alone or associated with comorbid conditions such as psoriatic arthritis (PsA), wrote Alan Menter, MD, of Baylor University Medical Center, Dallas, and coauthors of the guideline.
In the guideline, published in the Journal of the American Academy of Dermatology, the multidisciplinary work group identified screening tools to measure disease severity, strategies for management of comorbidities, and the safety and effectiveness of topical, systemic, and phototherapy treatments.
To assess disease severity, the work group recommended not only the use of body surface area (BSA), similar to measurement of severity in adults, but also the use of the Children’s Dermatology Life Quality Index, a 10-question quality of life survey, as BSA alone does not account for the potential negative impact of the disease on quality of life in terms of physical, emotional, social, and psychological function.
“For example, a child with psoriasis limited to the face or the entire scalp does not have severe disease based on BSA definitions, but if this involvement causes shame, social withdrawal, or bullying, it satisfies criteria for severe disease based on impact beyond the skin,” they said.
The work group stated that a variety of conditions may trigger or exacerbate psoriasis in children, including infections, cutaneous trauma, or physiological, emotional, and environmental stressors.
The majority of children with PsA develop joint inflammation before skin disease, the work group wrote. In addition, children with psoriasis are at increased risk for rheumatoid arthritis, so clinicians may need to distinguish between a combination of psoriasis and musculoskeletal issues and cases of either psoriatic or rheumatoid arthritis in young patients.
The cardiovascular risk factors associated with metabolic syndrome are greater in children with psoriasis, compared with children without psoriasis, the work group noted. In addition, pediatric psoriasis patients have a higher prevalence of obesity than children without psoriasis, and they recommended that children with psoriasis be monitored for the development of obesity, and that obese children with psoriasis should be referred for weight management.
The work group noted that data are insufficient in children to support the link between psoriasis and cardiovascular disease that has been documented in adults with psoriasis. However, “patients with pediatric psoriasis should have American Academy of Pediatrics (AAP)–recommended age-related cardiovascular screening regardless of the presence of signs or symptoms,” they said.
The guideline also recommends screening for dyslipidemia and hypertension according to AAP guidelines and educating pediatric psoriasis patients about the risk of diabetes and regularly screening for diabetes and insulin resistance in those who are obese. Overweight children with psoriasis may be screened at the provider’s discretion, they wrote. Patients with signs of inflammatory bowel disease, which also is associated with psoriasis in adults, should be considered for referral to a gastroenterologist, they noted.
Children with psoriasis should be screened regularly for mental health conditions regardless of age, and they should be asked about substance abuse, according to the guideline, and those with concerns should be referred for additional assessment and management.
The guideline divides treatment of psoriasis in children into three categories: topical, phototherapy and photochemotherapy, and systemic treatments (nonbiologic or biologic).
For topicals, the guideline recommendations include corticosteroids as an off-label therapy, as well as ultra-high-potency topical corticosteroids as monotherapy. Overall, “selection of a therapeutic routine (potency, delivery vehicle, frequency of application) should take into account sites of involvement, type and thickness of psoriasis, age of the patient, total BSA of application, anticipated occlusion, and disease acuity, among other patient-, disease-, and drug-related factors,” the authors wrote. Other topical options included in the recommendations: calcineurin inhibitors, topical vitamin D analogues, tazarotene (off label), anthralin, and coal tar.
Phototherapy has a history of use in psoriasis treatment and remains part of the current recommendations, although data in children are limited, and data on the use of phototherapy for pustular psoriasis in children are insufficient to make specific treatment and dosing recommendations, the work group noted. The researchers also noted that in-office phototherapy may not be feasible for many patients, but that in-home ultraviolet light equipment or natural sunlight in moderation could be recommended as an alternative.
The use of systemic, nonbiologic treatments for pediatric psoriasis should be “based on baseline severity of disease, subtype of psoriasis, speed of disease progression, lack of response to more conservative therapies such as topical agents and phototherapy (when appropriate), impaired physical or psychological functioning or [quality of life] due to disease extent, and the presence of comorbidities such as PsA,” the workgroup said.
Options for systemic treatment include methotrexate, cyclosporine (notably for pustular as well as plaque and erythrodermic psoriasis), and systemic retinoids. In addition, fumaric acid esters may be an option for children with moderate to severe psoriasis, with recommended clinical and laboratory monitoring.
The increasing safety and efficacy data on biologics in pediatric psoriasis patients support their consideration among first-line systemic treatments, the work group suggested. “Etanercept and ustekinumab are now [Food and Drug Administration] approved for patients with psoriasis 4 years and older and 12 years and older, respectively,” they said, and infliximab and adalimumab have been used off label in children.
The work group concluded that research and knowledge gaps about pediatric psoriasis persist and include mechanism of disease onset, development of comorbidities, and identification of ideal dosing for various treatments.
Finally, the work group emphasized the importance of collaboration between dermatologists and primary care providers for managing psoriasis in children, as well as the importance of patient education.
“Dermatologists should be mindful of the unique aspects of the emotional development of children and the social dynamics of having a visible difference,” they wrote. “Shared decision making with the patient (if age appropriate) and the caregivers is a useful approach, particularly as related to the use of off-label medications to treat severe disease,” they said.
“This is the first time that pediatric psoriasis has been discussed as an independent topic within the guideline,” said one of the guideline authors, Dawn M.R. Davis, MD, of the Mayo Clinic, Rochester, Minn., in an interview. “Children have unique physiology and psychosocial aspects to their care relative to adults. In addition, psoriasis has some clinical manifestations that are oftentimes distinctly seen in children,” she commented. “Creation of a guideline specific to children allows us to summarize the similarities and differences of disease presentation and management. It also allows an opportunity to clarify what research data (especially therapeutics) have been studied in children and their uses, safety profiles, and dosing,” she noted.
Psoriasis can be a psychosocially debilitating disease, she emphasized. “In children, for example, isolated or prominent facial involvement is common, which can be embarrassing and impact relationships.”
The take-home message for clinicians, Dr. Davis said, is to keep in mind the multisystemic nature of psoriasis. “It is not limited to the skin,” she said. “Treating a patient with psoriasis necessitates practicing whole-person care” and considering the multiple comorbidities that impact quality of life and overall health in children, as well as adults with psoriasis, she commented. “Dermatologists can empower patients and their caregivers by educating them on the multifocal, complex nature of the disease.” She added, “We have much to learn regarding psoriasis in the pediatric population. More research into therapeutics, topical and systemic, is necessary to optimize patient care.”
The guideline was based on studies published in the PubMed and MEDLINE databases from January 2011 through December 31, 2017.
Dr. Menter and Craig A. Elmets, MD, professor of dermatology, at the University of Alabama, Birmingham, were cochairs of the work group. The pediatric guideline is the latest in a multipart series of AAD-NPF guidelines on psoriasis being published this year in the Journal of the American Academy of Dermatology.
Many of the guideline authors, including lead author Dr. Menter, disclosed relationships with multiple companies; however, a minimum 51% of workgroup members had no relevant conflicts of interest in accordance with AAD policy. There was no funding source. Dr. Davis disclosed serving as an investigator for Regeneron, with no compensation.
SOURCE: Menter et al. J Am Acad Dermatol. 2019. doi: 10.1016/j.jaad.2019.08.049.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
EULAR releases recommendations for management of Sjögren’s syndrome
The European League Against Rheumatism has released its first recommendations for the management of Sjögren’s syndrome with topical and systemic treatments.
First author Manuel Ramos-Casals, MD, PhD, of Hospital Clinic de Barcelona Institut Clinic de Medicina i Dermatologia and colleagues on the multinational, multispecialty EULAR Sjögren Syndrome Task Force Group said the EULAR recommendations “synthesize current thinking” of management for Sjögren’s syndrome, which is complicated by individualized treatment that does not address the systemic disease and for which therapeutic decisions are often made based on expert opinion and personal experience because of a lack of data.
“Sjögren’s syndrome is generally treated through symptom management,” noted Sara McCoy, MD, who is director of the University of Wisconsin Health Sjögren’s Syndrome Clinic and was not involved in the recommendations, which were published in Annals of the Rheumatic Diseases. “Although we have measures to address these symptoms, we do not have any approved therapy that ameliorates the driving cause.”
There is limited high-quality evidence for many of the therapies discussed in the recommendations: In total, 9 randomized controlled trials, 18 prospective studies, and 5 case-control studies were included as evidence in the recommendations, and many involved a small number of patients.
The first overarching recommendation says that patients with Sjögren’s syndrome should be managed at centers of expertise using a multidisciplinary approach, the authors wrote. Two other overarching recommendations focus on how to manage certain patient cases. In cases of dryness, first-line therapy should include topical therapies for symptomatic relief, while clinicians should consider systemic therapies for patients with active systemic disease. All three overarching recommendations had a high level of agreement among task force members.
The task force also offered 12 specific recommendations on disease management, which take an “algorithmic approach” to Sjögren’s syndrome management that is guided by the EULAR Sjögren’s Syndrome Disease Activity Index (ESSDAI), Dr. McCoy said in an interview.
“The EULAR recommendations structure a clear approach to treatment of Sjögren’s manifestations, ranging from oral and ocular sicca management, to treatment of uncommon but severe systemic involvement of Sjögren’s,” she said.
Before treating patients for oral dryness symptoms, clinicians should evaluate salivary function at baseline using unstimulated whole salivary flows. First-line therapeutic approaches for oral dryness can include nonpharmacologic stimulation in cases of mild salivary gland dysfunction, pharmacologic stimulation in cases of moderate dysfunction, and substitution of saliva in cases of severe dysfunction, the authors said.
Ocular dryness should be assessed using ocular staining score followed by ocular surface–disease index, and once identified, it can be managed with artificial tears and ocular gels or ointments as first-line therapy. Topical immunosuppressive-containing drops and autologous serum eye drops are options for more severe or refractory ocular dryness.
Patients who have fatigue or pain associated with Sjögren’s syndrome should have their fatigue or pain scored with the EULAR Sjögren’s Syndrome Patient Reported Index (ESSPRI), and any concomitant disease should also be evaluated. Clinicians should consider acetaminophen and NSAIDs for treatment of acute musculoskeletal pain and other pain-modifying agents, such as hydroxychloroquine in cases of articular pain, despite a lack of evidence showing its efficacy in trials.
For systemic disease, the task force advised matching treatment to the organ(s) involved and using the ESSDAI to evaluate disease severity., They said that “patients presenting with at least moderate activity in one clinical domain, or with a global moderate disease activity score” of 5 or higher can be considered for systemic therapy.
Although many clinicians use glucocorticoids (GCs) for managing Sjögren’s syndrome, there is no evidence of the treatment’s efficacy in systemic disease. The task force recommended using glucocorticoids such as methylprednisolone at the minimum dose and withdrawing use “as soon as possible” in patients with inactive disease. “Based on the potential development of chronic damage in patients with uncontrolled systemic disease, some patients may require long-term therapy with GCs, especially those with severe organ impairments,” Dr. Ramos-Casals and colleagues wrote. “In these patients, the addition of immunosuppressive agents as GC-sparing agents is justified, always weighing the potential benefits and risks.” Because of a lack of head-to-head studies of different immunosuppressive agents, the task force did not recommend one particular agent over another.
One recommendation that contained a high level of evidence was for the use of B-cell targeted therapies for patients with severe, refractory systemic disease. Efficacy of rituximab was mixed in studies totaling more than 400 patients, but generally showed a positive outcome in at least one of the following areas: global response, organ-specific response, ESSDAI reduction, and prednisone reduction. The task force recommended that rituximab “may be considered” in “patients with severe, refractory systemic disease” and may be best for those with symptoms linked to cryoglobulinemic-associated vasculitis, with the potential of using belimumab (Benlysta) as rescue therapy. While systemic organ-specific therapeutic approaches vary, the general rule should be to treat with GCs first, followed by immunosuppressive agents and biologics as second-line therapy in cases refractory to GCs, either sequentially or in combination, the authors said.
For patients with B-cell lymphoma, clinicians should use individualized treatment based on the World Health Organization 2016 histologic subtype and the stage of the disease. While some clinicians employ a “watchful waiting” approach to treating low-grade hematologic neoplasia, “the decision to treat low-grade lymphomas or not must be discussed in a multidisciplinary committee, taking into account the fact that they are linked to the disease activity and are the ultimate stage of autoimmune B-cell activation,” the authors wrote.
The task force also defined a number of future research agenda goals for Sjögren’s syndrome treatment management, including developing personalized therapeutic approaches, finding predictors of biological response to agents used in treatment, and identifying biomarkers of poor outcomes.
In addition, EULAR acknowledged some research gaps exist in the guidelines for particular symptoms of Sjögren’s syndrome, such as in the case of fatigue. “The EULAR recommendations mention the common association of fatigue with Sjögren’s syndrome, though acknowledge that fatigue is more commonly seen in the same demographic cohort as Sjögren’s,” Dr. McCoy said. “Aside from exercise, there remains little available to address this common and burdensome symptom amongst Sjögren’s patients. Overall, this is a pertinent gap in knowledge in our Sjögren’s patients.”
The Sjögren’s syndrome recommendations were funded by EULAR. Many task force members reported relationships with industry in the form of advisory board and speaker’s bureau memberships, consultancies, and grants. Dr. McCoy reported serving on a data monitoring committee for Bristol-Myers Squibb unrelated to the data in the EULAR recommendations.
SOURCE: Ramos-Casals M et al. Ann Rheum Dis. 2019 Oct 31. doi: 10.1136/annrheumdis-2019-216114
The European League Against Rheumatism has released its first recommendations for the management of Sjögren’s syndrome with topical and systemic treatments.
First author Manuel Ramos-Casals, MD, PhD, of Hospital Clinic de Barcelona Institut Clinic de Medicina i Dermatologia and colleagues on the multinational, multispecialty EULAR Sjögren Syndrome Task Force Group said the EULAR recommendations “synthesize current thinking” of management for Sjögren’s syndrome, which is complicated by individualized treatment that does not address the systemic disease and for which therapeutic decisions are often made based on expert opinion and personal experience because of a lack of data.
“Sjögren’s syndrome is generally treated through symptom management,” noted Sara McCoy, MD, who is director of the University of Wisconsin Health Sjögren’s Syndrome Clinic and was not involved in the recommendations, which were published in Annals of the Rheumatic Diseases. “Although we have measures to address these symptoms, we do not have any approved therapy that ameliorates the driving cause.”
There is limited high-quality evidence for many of the therapies discussed in the recommendations: In total, 9 randomized controlled trials, 18 prospective studies, and 5 case-control studies were included as evidence in the recommendations, and many involved a small number of patients.
The first overarching recommendation says that patients with Sjögren’s syndrome should be managed at centers of expertise using a multidisciplinary approach, the authors wrote. Two other overarching recommendations focus on how to manage certain patient cases. In cases of dryness, first-line therapy should include topical therapies for symptomatic relief, while clinicians should consider systemic therapies for patients with active systemic disease. All three overarching recommendations had a high level of agreement among task force members.
The task force also offered 12 specific recommendations on disease management, which take an “algorithmic approach” to Sjögren’s syndrome management that is guided by the EULAR Sjögren’s Syndrome Disease Activity Index (ESSDAI), Dr. McCoy said in an interview.
“The EULAR recommendations structure a clear approach to treatment of Sjögren’s manifestations, ranging from oral and ocular sicca management, to treatment of uncommon but severe systemic involvement of Sjögren’s,” she said.
Before treating patients for oral dryness symptoms, clinicians should evaluate salivary function at baseline using unstimulated whole salivary flows. First-line therapeutic approaches for oral dryness can include nonpharmacologic stimulation in cases of mild salivary gland dysfunction, pharmacologic stimulation in cases of moderate dysfunction, and substitution of saliva in cases of severe dysfunction, the authors said.
Ocular dryness should be assessed using ocular staining score followed by ocular surface–disease index, and once identified, it can be managed with artificial tears and ocular gels or ointments as first-line therapy. Topical immunosuppressive-containing drops and autologous serum eye drops are options for more severe or refractory ocular dryness.
Patients who have fatigue or pain associated with Sjögren’s syndrome should have their fatigue or pain scored with the EULAR Sjögren’s Syndrome Patient Reported Index (ESSPRI), and any concomitant disease should also be evaluated. Clinicians should consider acetaminophen and NSAIDs for treatment of acute musculoskeletal pain and other pain-modifying agents, such as hydroxychloroquine in cases of articular pain, despite a lack of evidence showing its efficacy in trials.
For systemic disease, the task force advised matching treatment to the organ(s) involved and using the ESSDAI to evaluate disease severity., They said that “patients presenting with at least moderate activity in one clinical domain, or with a global moderate disease activity score” of 5 or higher can be considered for systemic therapy.
Although many clinicians use glucocorticoids (GCs) for managing Sjögren’s syndrome, there is no evidence of the treatment’s efficacy in systemic disease. The task force recommended using glucocorticoids such as methylprednisolone at the minimum dose and withdrawing use “as soon as possible” in patients with inactive disease. “Based on the potential development of chronic damage in patients with uncontrolled systemic disease, some patients may require long-term therapy with GCs, especially those with severe organ impairments,” Dr. Ramos-Casals and colleagues wrote. “In these patients, the addition of immunosuppressive agents as GC-sparing agents is justified, always weighing the potential benefits and risks.” Because of a lack of head-to-head studies of different immunosuppressive agents, the task force did not recommend one particular agent over another.
One recommendation that contained a high level of evidence was for the use of B-cell targeted therapies for patients with severe, refractory systemic disease. Efficacy of rituximab was mixed in studies totaling more than 400 patients, but generally showed a positive outcome in at least one of the following areas: global response, organ-specific response, ESSDAI reduction, and prednisone reduction. The task force recommended that rituximab “may be considered” in “patients with severe, refractory systemic disease” and may be best for those with symptoms linked to cryoglobulinemic-associated vasculitis, with the potential of using belimumab (Benlysta) as rescue therapy. While systemic organ-specific therapeutic approaches vary, the general rule should be to treat with GCs first, followed by immunosuppressive agents and biologics as second-line therapy in cases refractory to GCs, either sequentially or in combination, the authors said.
For patients with B-cell lymphoma, clinicians should use individualized treatment based on the World Health Organization 2016 histologic subtype and the stage of the disease. While some clinicians employ a “watchful waiting” approach to treating low-grade hematologic neoplasia, “the decision to treat low-grade lymphomas or not must be discussed in a multidisciplinary committee, taking into account the fact that they are linked to the disease activity and are the ultimate stage of autoimmune B-cell activation,” the authors wrote.
The task force also defined a number of future research agenda goals for Sjögren’s syndrome treatment management, including developing personalized therapeutic approaches, finding predictors of biological response to agents used in treatment, and identifying biomarkers of poor outcomes.
In addition, EULAR acknowledged some research gaps exist in the guidelines for particular symptoms of Sjögren’s syndrome, such as in the case of fatigue. “The EULAR recommendations mention the common association of fatigue with Sjögren’s syndrome, though acknowledge that fatigue is more commonly seen in the same demographic cohort as Sjögren’s,” Dr. McCoy said. “Aside from exercise, there remains little available to address this common and burdensome symptom amongst Sjögren’s patients. Overall, this is a pertinent gap in knowledge in our Sjögren’s patients.”
The Sjögren’s syndrome recommendations were funded by EULAR. Many task force members reported relationships with industry in the form of advisory board and speaker’s bureau memberships, consultancies, and grants. Dr. McCoy reported serving on a data monitoring committee for Bristol-Myers Squibb unrelated to the data in the EULAR recommendations.
SOURCE: Ramos-Casals M et al. Ann Rheum Dis. 2019 Oct 31. doi: 10.1136/annrheumdis-2019-216114
The European League Against Rheumatism has released its first recommendations for the management of Sjögren’s syndrome with topical and systemic treatments.
First author Manuel Ramos-Casals, MD, PhD, of Hospital Clinic de Barcelona Institut Clinic de Medicina i Dermatologia and colleagues on the multinational, multispecialty EULAR Sjögren Syndrome Task Force Group said the EULAR recommendations “synthesize current thinking” of management for Sjögren’s syndrome, which is complicated by individualized treatment that does not address the systemic disease and for which therapeutic decisions are often made based on expert opinion and personal experience because of a lack of data.
“Sjögren’s syndrome is generally treated through symptom management,” noted Sara McCoy, MD, who is director of the University of Wisconsin Health Sjögren’s Syndrome Clinic and was not involved in the recommendations, which were published in Annals of the Rheumatic Diseases. “Although we have measures to address these symptoms, we do not have any approved therapy that ameliorates the driving cause.”
There is limited high-quality evidence for many of the therapies discussed in the recommendations: In total, 9 randomized controlled trials, 18 prospective studies, and 5 case-control studies were included as evidence in the recommendations, and many involved a small number of patients.
The first overarching recommendation says that patients with Sjögren’s syndrome should be managed at centers of expertise using a multidisciplinary approach, the authors wrote. Two other overarching recommendations focus on how to manage certain patient cases. In cases of dryness, first-line therapy should include topical therapies for symptomatic relief, while clinicians should consider systemic therapies for patients with active systemic disease. All three overarching recommendations had a high level of agreement among task force members.
The task force also offered 12 specific recommendations on disease management, which take an “algorithmic approach” to Sjögren’s syndrome management that is guided by the EULAR Sjögren’s Syndrome Disease Activity Index (ESSDAI), Dr. McCoy said in an interview.
“The EULAR recommendations structure a clear approach to treatment of Sjögren’s manifestations, ranging from oral and ocular sicca management, to treatment of uncommon but severe systemic involvement of Sjögren’s,” she said.
Before treating patients for oral dryness symptoms, clinicians should evaluate salivary function at baseline using unstimulated whole salivary flows. First-line therapeutic approaches for oral dryness can include nonpharmacologic stimulation in cases of mild salivary gland dysfunction, pharmacologic stimulation in cases of moderate dysfunction, and substitution of saliva in cases of severe dysfunction, the authors said.
Ocular dryness should be assessed using ocular staining score followed by ocular surface–disease index, and once identified, it can be managed with artificial tears and ocular gels or ointments as first-line therapy. Topical immunosuppressive-containing drops and autologous serum eye drops are options for more severe or refractory ocular dryness.
Patients who have fatigue or pain associated with Sjögren’s syndrome should have their fatigue or pain scored with the EULAR Sjögren’s Syndrome Patient Reported Index (ESSPRI), and any concomitant disease should also be evaluated. Clinicians should consider acetaminophen and NSAIDs for treatment of acute musculoskeletal pain and other pain-modifying agents, such as hydroxychloroquine in cases of articular pain, despite a lack of evidence showing its efficacy in trials.
For systemic disease, the task force advised matching treatment to the organ(s) involved and using the ESSDAI to evaluate disease severity., They said that “patients presenting with at least moderate activity in one clinical domain, or with a global moderate disease activity score” of 5 or higher can be considered for systemic therapy.
Although many clinicians use glucocorticoids (GCs) for managing Sjögren’s syndrome, there is no evidence of the treatment’s efficacy in systemic disease. The task force recommended using glucocorticoids such as methylprednisolone at the minimum dose and withdrawing use “as soon as possible” in patients with inactive disease. “Based on the potential development of chronic damage in patients with uncontrolled systemic disease, some patients may require long-term therapy with GCs, especially those with severe organ impairments,” Dr. Ramos-Casals and colleagues wrote. “In these patients, the addition of immunosuppressive agents as GC-sparing agents is justified, always weighing the potential benefits and risks.” Because of a lack of head-to-head studies of different immunosuppressive agents, the task force did not recommend one particular agent over another.
One recommendation that contained a high level of evidence was for the use of B-cell targeted therapies for patients with severe, refractory systemic disease. Efficacy of rituximab was mixed in studies totaling more than 400 patients, but generally showed a positive outcome in at least one of the following areas: global response, organ-specific response, ESSDAI reduction, and prednisone reduction. The task force recommended that rituximab “may be considered” in “patients with severe, refractory systemic disease” and may be best for those with symptoms linked to cryoglobulinemic-associated vasculitis, with the potential of using belimumab (Benlysta) as rescue therapy. While systemic organ-specific therapeutic approaches vary, the general rule should be to treat with GCs first, followed by immunosuppressive agents and biologics as second-line therapy in cases refractory to GCs, either sequentially or in combination, the authors said.
For patients with B-cell lymphoma, clinicians should use individualized treatment based on the World Health Organization 2016 histologic subtype and the stage of the disease. While some clinicians employ a “watchful waiting” approach to treating low-grade hematologic neoplasia, “the decision to treat low-grade lymphomas or not must be discussed in a multidisciplinary committee, taking into account the fact that they are linked to the disease activity and are the ultimate stage of autoimmune B-cell activation,” the authors wrote.
The task force also defined a number of future research agenda goals for Sjögren’s syndrome treatment management, including developing personalized therapeutic approaches, finding predictors of biological response to agents used in treatment, and identifying biomarkers of poor outcomes.
In addition, EULAR acknowledged some research gaps exist in the guidelines for particular symptoms of Sjögren’s syndrome, such as in the case of fatigue. “The EULAR recommendations mention the common association of fatigue with Sjögren’s syndrome, though acknowledge that fatigue is more commonly seen in the same demographic cohort as Sjögren’s,” Dr. McCoy said. “Aside from exercise, there remains little available to address this common and burdensome symptom amongst Sjögren’s patients. Overall, this is a pertinent gap in knowledge in our Sjögren’s patients.”
The Sjögren’s syndrome recommendations were funded by EULAR. Many task force members reported relationships with industry in the form of advisory board and speaker’s bureau memberships, consultancies, and grants. Dr. McCoy reported serving on a data monitoring committee for Bristol-Myers Squibb unrelated to the data in the EULAR recommendations.
SOURCE: Ramos-Casals M et al. Ann Rheum Dis. 2019 Oct 31. doi: 10.1136/annrheumdis-2019-216114
FROM ANNALS OF THE RHEUMATIC DISEASES
ACP: Low-risk adults aged 50-75 should undergo regular screening for colorectal cancer
issued by the American College of Physicians.
Regular screening can be discontinued after age 75 years, Amir Qaseem, MD, president of clinical policy and the Center for Evidence Reviews at the American College of Physicians, and colleagues wrote in the Annals of Internal Medicine.
No one test is preferred over another, according to the guidance statement. Patients and physicians can select the test type together, based on individual needs and preferences, and each test carries its own screening interval. But regular testing has been proven time and again to reduce the risk of colorectal cancer mortality, and more people should have it, according to the guidance.
“Not enough people in the United States get screened for colorectal cancer,” ACP President Robert M. McLean, MD, said in a press statement. “Physicians should perform an individualized risk assessment for colorectal cancer in all adults. Doctors and patients should select the screening test based on a discussion of the benefits, harms, costs, availability, frequency, and patient preferences.”
The guidance is an attempt to balance existing guidelines authored by the U.S. Preventive Services Task Force and the Canadian Task Force on Preventive Health Care (CTFPHC), but it also was developed following critical review of those from the American Cancer Society and other organizations.
The ACP guidance is for adults at average risk for colorectal cancer who do not have symptoms; it does not apply to adults with a family history of colorectal cancer, a long-standing history of inflammatory bowel disease, genetic syndromes such as familial cancerous polyps, a personal history of previous colorectal cancer or benign polyps, or other risk factors.
The guidance was based on evaluations of stool-based tests, including the fecal immunochemical test (FIT), also called the immunochemical-based fecal occult blood test (FOBT), and direct visualization with endoscopic and radiologic tests, including flexible sigmoidoscopy, colonoscopy, and CT colonography. The guidance includes the following recommendations:
Clinicians should regularly screen for colorectal cancer in average-risk adults between the ages of 50 and 75 years.
This recommendation is in line with those made by the U.S. Preventive Services Task Force and the CTFPHC. Data suggest that regular screening reduces colorectal cancer–specific mortality in this age group, with those aged 65-75 years likely to garner the most benefit.
The absolute risk reduction increases with age and varies with test type. For every-other-year FOBT, it rises from 0.037% in those younger than 60 years to 0.20% in those aged 60 years or older. For flexible sigmoidoscopy, the risk reduction rises from 0.05% in the younger group to 0.29% in the older group.
Data from the CTFPHC show that the net benefit in those aged 50-59 years is small, however. This may influence the decision about when to start screening.
Clinicians should select the colorectal cancer screening test with the patient based on a discussion of benefits, harms, costs, availability, frequency, and patient preferences.
The FIT or FOBT should be performed every 2 years, colonoscopy every 10 years, and flexible sigmoidoscopy every 10 years, plus FIT every 2 years.
No data suggest a benefit of one test over another; however, “all screening tests are associated with potential benefits as well as harms,” the document states. “Clinical decisions need to be individualized using patient clinical characteristics, patient preferences, and screening test frequency and availability. Because many eligible patients have never been screened and some may not adhere to recommendations about subsequent screening or follow-up of positive findings on screening tests (such as colonoscopy after a positive result on a stool-based screening test), patient informed decision making and adherence are important factors in selection of a [colorectal cancer] screening test.”
Discussions with patients should include topics like the recommended frequency of each test, bowel preparation, anesthesia, transportation to and from testing site, time commitments, and the necessary steps if a test result is positive.
Clinicians should discontinue screening for colorectal cancer in average-risk adults older than 75 years or in adults with a life expectancy of 10 years or less.
While the benefit from screening increases with age, so do the risks for harm, especially serious harm. Data show the balance of harms and benefits reaching a tipping point at around 75 years of age. But again, this isn’t a universal recommendation, the statement says.
“Persons with no history of [colorectal cancer] screening may benefit from screening after age 75 years, whereas those who have received regular screening with negative results may not.”
Cessation of testing considers life expectancy after age 75 years. The average life expectancy for healthy 75-year-old men and women in the United States is 9.9 and 12 years, respectively. But among men and women with serious medical comorbidities, average life expectancy after age 70 years drops to 8.9 and 10.8 years, respectively.
“Therefore, most persons aged 75 years or older, as well as most adults who are younger than 75 years but have serious comorbid conditions [such as chronic renal failure], are unlikely to benefit from screening but would undergo unnecessary, burdensome, potentially harmful, and costly screening tests.”
As in any testing discussion, personal preferences are important, but not just to make patients feel comfortable about their choice. Mindset about colorectal cancer testing has a very big effect on compliance, the statement noted.
“For example, a biennial stool test is not a good screening strategy for patients who may be unwilling or unlikely to follow up every other year. In addition, given the tradeoffs between benefits and harms, some patients may want less intensive screening, such as screening that begins at a later age, stops at an earlier age, or recurs less frequently regardless of modality selected.”
SOURCE: Qassam A et al. Ann Intern Med. 2019;171:643-54.
Cost-effectiveness is one more factor in the colorectal screening discussion, Michael Pignone, MD, said in an accompanying editorial.
Two studies by Ladabaum et al. reported cost-effectiveness modeling for various CRC screening techniques: “Comparative effectiveness and cost effectiveness of a multitarget stool DNA test to screen for colorectal neoplasia” (Gastroenterology. 2019;151,:427-39.e6) and “Cost-effectiveness and national effects of initiating colorectal cancer screening for average-risk persons at age 45 years instead of 50 years” (Gastroenterology. 2019;157:137-48).
These reports concluded that annual stool testing is more effective – but also more costly – than biennial testing. However, the additional cost per unit of benefit (figured in quality-adjusted life-years) is about $33,000 per life-year gained – a reasonable cost. “Hence, annual testing is a viable screening option,” wrote Dr. Pignone.
Starting screening at age 45 years instead of 50 years also produced an additional cost per life-year, but again, it is reasonable at $33,900 for colonoscopy screening and $7,700 for stool testing.
“However, for the same amount of additional resources, increasing screening rates in 55- or 65-year-olds or improving the proportion of positive stool test results that are followed by colonoscopy from 60% to 90% would yield much more benefit in life-years gained than lowering the starting age to 45 years.”
Analyses such as these conditionally support earlier colorectal cancer screening only if the universal screening rate for 50- to 75-year-olds is more than 80%, he wrote. “They also reinforce the most important point in all of the major guidelines: Any recommended form of screening in the 50- to 75-year age range is likely to be very cost-effective (if not cost-saving) compared with no screening and should be strongly encouraged.”
Dr. Pignone is director of the program on cancer prevention and control at the LIVESTRONG Cancer Institutes at the University of Texas, Austin.
Cost-effectiveness is one more factor in the colorectal screening discussion, Michael Pignone, MD, said in an accompanying editorial.
Two studies by Ladabaum et al. reported cost-effectiveness modeling for various CRC screening techniques: “Comparative effectiveness and cost effectiveness of a multitarget stool DNA test to screen for colorectal neoplasia” (Gastroenterology. 2019;151,:427-39.e6) and “Cost-effectiveness and national effects of initiating colorectal cancer screening for average-risk persons at age 45 years instead of 50 years” (Gastroenterology. 2019;157:137-48).
These reports concluded that annual stool testing is more effective – but also more costly – than biennial testing. However, the additional cost per unit of benefit (figured in quality-adjusted life-years) is about $33,000 per life-year gained – a reasonable cost. “Hence, annual testing is a viable screening option,” wrote Dr. Pignone.
Starting screening at age 45 years instead of 50 years also produced an additional cost per life-year, but again, it is reasonable at $33,900 for colonoscopy screening and $7,700 for stool testing.
“However, for the same amount of additional resources, increasing screening rates in 55- or 65-year-olds or improving the proportion of positive stool test results that are followed by colonoscopy from 60% to 90% would yield much more benefit in life-years gained than lowering the starting age to 45 years.”
Analyses such as these conditionally support earlier colorectal cancer screening only if the universal screening rate for 50- to 75-year-olds is more than 80%, he wrote. “They also reinforce the most important point in all of the major guidelines: Any recommended form of screening in the 50- to 75-year age range is likely to be very cost-effective (if not cost-saving) compared with no screening and should be strongly encouraged.”
Dr. Pignone is director of the program on cancer prevention and control at the LIVESTRONG Cancer Institutes at the University of Texas, Austin.
Cost-effectiveness is one more factor in the colorectal screening discussion, Michael Pignone, MD, said in an accompanying editorial.
Two studies by Ladabaum et al. reported cost-effectiveness modeling for various CRC screening techniques: “Comparative effectiveness and cost effectiveness of a multitarget stool DNA test to screen for colorectal neoplasia” (Gastroenterology. 2019;151,:427-39.e6) and “Cost-effectiveness and national effects of initiating colorectal cancer screening for average-risk persons at age 45 years instead of 50 years” (Gastroenterology. 2019;157:137-48).
These reports concluded that annual stool testing is more effective – but also more costly – than biennial testing. However, the additional cost per unit of benefit (figured in quality-adjusted life-years) is about $33,000 per life-year gained – a reasonable cost. “Hence, annual testing is a viable screening option,” wrote Dr. Pignone.
Starting screening at age 45 years instead of 50 years also produced an additional cost per life-year, but again, it is reasonable at $33,900 for colonoscopy screening and $7,700 for stool testing.
“However, for the same amount of additional resources, increasing screening rates in 55- or 65-year-olds or improving the proportion of positive stool test results that are followed by colonoscopy from 60% to 90% would yield much more benefit in life-years gained than lowering the starting age to 45 years.”
Analyses such as these conditionally support earlier colorectal cancer screening only if the universal screening rate for 50- to 75-year-olds is more than 80%, he wrote. “They also reinforce the most important point in all of the major guidelines: Any recommended form of screening in the 50- to 75-year age range is likely to be very cost-effective (if not cost-saving) compared with no screening and should be strongly encouraged.”
Dr. Pignone is director of the program on cancer prevention and control at the LIVESTRONG Cancer Institutes at the University of Texas, Austin.
issued by the American College of Physicians.
Regular screening can be discontinued after age 75 years, Amir Qaseem, MD, president of clinical policy and the Center for Evidence Reviews at the American College of Physicians, and colleagues wrote in the Annals of Internal Medicine.
No one test is preferred over another, according to the guidance statement. Patients and physicians can select the test type together, based on individual needs and preferences, and each test carries its own screening interval. But regular testing has been proven time and again to reduce the risk of colorectal cancer mortality, and more people should have it, according to the guidance.
“Not enough people in the United States get screened for colorectal cancer,” ACP President Robert M. McLean, MD, said in a press statement. “Physicians should perform an individualized risk assessment for colorectal cancer in all adults. Doctors and patients should select the screening test based on a discussion of the benefits, harms, costs, availability, frequency, and patient preferences.”
The guidance is an attempt to balance existing guidelines authored by the U.S. Preventive Services Task Force and the Canadian Task Force on Preventive Health Care (CTFPHC), but it also was developed following critical review of those from the American Cancer Society and other organizations.
The ACP guidance is for adults at average risk for colorectal cancer who do not have symptoms; it does not apply to adults with a family history of colorectal cancer, a long-standing history of inflammatory bowel disease, genetic syndromes such as familial cancerous polyps, a personal history of previous colorectal cancer or benign polyps, or other risk factors.
The guidance was based on evaluations of stool-based tests, including the fecal immunochemical test (FIT), also called the immunochemical-based fecal occult blood test (FOBT), and direct visualization with endoscopic and radiologic tests, including flexible sigmoidoscopy, colonoscopy, and CT colonography. The guidance includes the following recommendations:
Clinicians should regularly screen for colorectal cancer in average-risk adults between the ages of 50 and 75 years.
This recommendation is in line with those made by the U.S. Preventive Services Task Force and the CTFPHC. Data suggest that regular screening reduces colorectal cancer–specific mortality in this age group, with those aged 65-75 years likely to garner the most benefit.
The absolute risk reduction increases with age and varies with test type. For every-other-year FOBT, it rises from 0.037% in those younger than 60 years to 0.20% in those aged 60 years or older. For flexible sigmoidoscopy, the risk reduction rises from 0.05% in the younger group to 0.29% in the older group.
Data from the CTFPHC show that the net benefit in those aged 50-59 years is small, however. This may influence the decision about when to start screening.
Clinicians should select the colorectal cancer screening test with the patient based on a discussion of benefits, harms, costs, availability, frequency, and patient preferences.
The FIT or FOBT should be performed every 2 years, colonoscopy every 10 years, and flexible sigmoidoscopy every 10 years, plus FIT every 2 years.
No data suggest a benefit of one test over another; however, “all screening tests are associated with potential benefits as well as harms,” the document states. “Clinical decisions need to be individualized using patient clinical characteristics, patient preferences, and screening test frequency and availability. Because many eligible patients have never been screened and some may not adhere to recommendations about subsequent screening or follow-up of positive findings on screening tests (such as colonoscopy after a positive result on a stool-based screening test), patient informed decision making and adherence are important factors in selection of a [colorectal cancer] screening test.”
Discussions with patients should include topics like the recommended frequency of each test, bowel preparation, anesthesia, transportation to and from testing site, time commitments, and the necessary steps if a test result is positive.
Clinicians should discontinue screening for colorectal cancer in average-risk adults older than 75 years or in adults with a life expectancy of 10 years or less.
While the benefit from screening increases with age, so do the risks for harm, especially serious harm. Data show the balance of harms and benefits reaching a tipping point at around 75 years of age. But again, this isn’t a universal recommendation, the statement says.
“Persons with no history of [colorectal cancer] screening may benefit from screening after age 75 years, whereas those who have received regular screening with negative results may not.”
Cessation of testing considers life expectancy after age 75 years. The average life expectancy for healthy 75-year-old men and women in the United States is 9.9 and 12 years, respectively. But among men and women with serious medical comorbidities, average life expectancy after age 70 years drops to 8.9 and 10.8 years, respectively.
“Therefore, most persons aged 75 years or older, as well as most adults who are younger than 75 years but have serious comorbid conditions [such as chronic renal failure], are unlikely to benefit from screening but would undergo unnecessary, burdensome, potentially harmful, and costly screening tests.”
As in any testing discussion, personal preferences are important, but not just to make patients feel comfortable about their choice. Mindset about colorectal cancer testing has a very big effect on compliance, the statement noted.
“For example, a biennial stool test is not a good screening strategy for patients who may be unwilling or unlikely to follow up every other year. In addition, given the tradeoffs between benefits and harms, some patients may want less intensive screening, such as screening that begins at a later age, stops at an earlier age, or recurs less frequently regardless of modality selected.”
SOURCE: Qassam A et al. Ann Intern Med. 2019;171:643-54.
issued by the American College of Physicians.
Regular screening can be discontinued after age 75 years, Amir Qaseem, MD, president of clinical policy and the Center for Evidence Reviews at the American College of Physicians, and colleagues wrote in the Annals of Internal Medicine.
No one test is preferred over another, according to the guidance statement. Patients and physicians can select the test type together, based on individual needs and preferences, and each test carries its own screening interval. But regular testing has been proven time and again to reduce the risk of colorectal cancer mortality, and more people should have it, according to the guidance.
“Not enough people in the United States get screened for colorectal cancer,” ACP President Robert M. McLean, MD, said in a press statement. “Physicians should perform an individualized risk assessment for colorectal cancer in all adults. Doctors and patients should select the screening test based on a discussion of the benefits, harms, costs, availability, frequency, and patient preferences.”
The guidance is an attempt to balance existing guidelines authored by the U.S. Preventive Services Task Force and the Canadian Task Force on Preventive Health Care (CTFPHC), but it also was developed following critical review of those from the American Cancer Society and other organizations.
The ACP guidance is for adults at average risk for colorectal cancer who do not have symptoms; it does not apply to adults with a family history of colorectal cancer, a long-standing history of inflammatory bowel disease, genetic syndromes such as familial cancerous polyps, a personal history of previous colorectal cancer or benign polyps, or other risk factors.
The guidance was based on evaluations of stool-based tests, including the fecal immunochemical test (FIT), also called the immunochemical-based fecal occult blood test (FOBT), and direct visualization with endoscopic and radiologic tests, including flexible sigmoidoscopy, colonoscopy, and CT colonography. The guidance includes the following recommendations:
Clinicians should regularly screen for colorectal cancer in average-risk adults between the ages of 50 and 75 years.
This recommendation is in line with those made by the U.S. Preventive Services Task Force and the CTFPHC. Data suggest that regular screening reduces colorectal cancer–specific mortality in this age group, with those aged 65-75 years likely to garner the most benefit.
The absolute risk reduction increases with age and varies with test type. For every-other-year FOBT, it rises from 0.037% in those younger than 60 years to 0.20% in those aged 60 years or older. For flexible sigmoidoscopy, the risk reduction rises from 0.05% in the younger group to 0.29% in the older group.
Data from the CTFPHC show that the net benefit in those aged 50-59 years is small, however. This may influence the decision about when to start screening.
Clinicians should select the colorectal cancer screening test with the patient based on a discussion of benefits, harms, costs, availability, frequency, and patient preferences.
The FIT or FOBT should be performed every 2 years, colonoscopy every 10 years, and flexible sigmoidoscopy every 10 years, plus FIT every 2 years.
No data suggest a benefit of one test over another; however, “all screening tests are associated with potential benefits as well as harms,” the document states. “Clinical decisions need to be individualized using patient clinical characteristics, patient preferences, and screening test frequency and availability. Because many eligible patients have never been screened and some may not adhere to recommendations about subsequent screening or follow-up of positive findings on screening tests (such as colonoscopy after a positive result on a stool-based screening test), patient informed decision making and adherence are important factors in selection of a [colorectal cancer] screening test.”
Discussions with patients should include topics like the recommended frequency of each test, bowel preparation, anesthesia, transportation to and from testing site, time commitments, and the necessary steps if a test result is positive.
Clinicians should discontinue screening for colorectal cancer in average-risk adults older than 75 years or in adults with a life expectancy of 10 years or less.
While the benefit from screening increases with age, so do the risks for harm, especially serious harm. Data show the balance of harms and benefits reaching a tipping point at around 75 years of age. But again, this isn’t a universal recommendation, the statement says.
“Persons with no history of [colorectal cancer] screening may benefit from screening after age 75 years, whereas those who have received regular screening with negative results may not.”
Cessation of testing considers life expectancy after age 75 years. The average life expectancy for healthy 75-year-old men and women in the United States is 9.9 and 12 years, respectively. But among men and women with serious medical comorbidities, average life expectancy after age 70 years drops to 8.9 and 10.8 years, respectively.
“Therefore, most persons aged 75 years or older, as well as most adults who are younger than 75 years but have serious comorbid conditions [such as chronic renal failure], are unlikely to benefit from screening but would undergo unnecessary, burdensome, potentially harmful, and costly screening tests.”
As in any testing discussion, personal preferences are important, but not just to make patients feel comfortable about their choice. Mindset about colorectal cancer testing has a very big effect on compliance, the statement noted.
“For example, a biennial stool test is not a good screening strategy for patients who may be unwilling or unlikely to follow up every other year. In addition, given the tradeoffs between benefits and harms, some patients may want less intensive screening, such as screening that begins at a later age, stops at an earlier age, or recurs less frequently regardless of modality selected.”
SOURCE: Qassam A et al. Ann Intern Med. 2019;171:643-54.
FROM THE ANNALS OF INTERNAL MEDICINE
First NCCN guideline on hematopoietic cell transplantation focuses on GVHD
Recommendations for the diagnosis and management of acute and chronic graft-versus-host disease (GVHD) are the central focus of the first National Comprehensive Cancer Network (NCCN) guideline on hematopoietic cell transplantation.
The guideline presents detailed recommendations for the evaluation of hematopoietic cell transplant (HCT) recipients, and an extensive section on the diagnosis and workup of GVHD, including information on staging and grading of acute GVHD, grading of chronic GVHD, treatment response criteria, and suggested systemic therapies for steroid-refractory disease.
“We wanted to both build on the commonly used approach to stage and treat graft-versus-host disease, and make sure that this information is readily available for physicians-in-training and young physicians who are learning about transplants,” said guideline committee chair Ayman Saad, MB BCh, of The Ohio State University Comprehensive Cancer Center, James Cancer Hospital and Solove Research Institute in Columbus, Ohio.
In an interview, Dr. Saad emphasized that an important goal of the guidelines is to encourage general oncologists to recognize early signs of GVHD and refer potential candidates to transplant centers for further evaluation.
“We also urge oncologists who may be caring for patients after HCT to familiarize themselves with the varied manifestations of GVHD – a very common and significant posttransplant complication – and to consult with transplant providers to optimize their ongoing care. The guidelines explain how to diagnose and treat this condition in order to achieve the best possible outcomes,” guideline panel member Alison W. Loren, MD, director of blood and marrow transplantation at Abraham Cancer Center, University of Pennsylvania, Philadelphia, said in a statement.
The guideline includes links to other NCCN guidelines for diseases where HCT is a common therapeutic option, including leukemias, myeloid malignancies, lymphomas, central nervous system cancers, and testicular cancer.
The HCT guideline includes:
- Pretransplant recipient evaluation with recommendations for clinical assessment and imaging.
- Diagnosis and workup of GVHD, with separate algorithms for suspected acute or chronic GVHD.
- Specific interventions for management of acute GVHD with corticosteroids or other systemic agents.
- Chronic GVHD diagnosis by organ site and symptoms, with a severity scoring system.
- Chronic GVHD steroid response definitions and criteria.
- Suggested systemic agents for steroid-refractory GVHD.
One feature that is unusual for an NCCN guideline document is a page of photographs to assist clinicians in diagnosing range-of-motion abnormalities in the shoulder, elbow, hand, and ankle of patients with suspected or confirmed GVHD. Dr. Saad said that future iterations of the guideline will include additional photos to help clinicians develop a visual repertoire of potential GVHD signs.
Future versions will also include a discussion section and more comprehensive information on other common complications following HCT transplant, as well as management of posttransplant relapse.
Ideally, the guideline will help clinicians document and justify clinical decisions surrounding HCT and GVHD management in discussions with third-party payers, Dr. Saad said.
“Sometimes we struggle with payers when we want to use a certain modality to treat GVHD, and they respond ‘that’s not approved,’ or ‘that’s not a common indication,’ et cetera,” he said. “What we’re trying to put here are the commonly used therapies that most, but not all, experts agree on, and we can use this to negotiate with payers.”
He also emphasized that the guideline is meant to be instructive rather than prescriptive and is not meant to hinder innovations that may emerge from clinical trials.
“We would appreciate any feedback from non-NCCN centers as well as experts at NCCN centers, and we’ll be more than happy to address any concerns or criticisms they have,” he said.
Dr. Saad reported financial relationships with Actinium and Incysus Biomedical. Dr. Loren has previously reported having no disclosures.
Recommendations for the diagnosis and management of acute and chronic graft-versus-host disease (GVHD) are the central focus of the first National Comprehensive Cancer Network (NCCN) guideline on hematopoietic cell transplantation.
The guideline presents detailed recommendations for the evaluation of hematopoietic cell transplant (HCT) recipients, and an extensive section on the diagnosis and workup of GVHD, including information on staging and grading of acute GVHD, grading of chronic GVHD, treatment response criteria, and suggested systemic therapies for steroid-refractory disease.
“We wanted to both build on the commonly used approach to stage and treat graft-versus-host disease, and make sure that this information is readily available for physicians-in-training and young physicians who are learning about transplants,” said guideline committee chair Ayman Saad, MB BCh, of The Ohio State University Comprehensive Cancer Center, James Cancer Hospital and Solove Research Institute in Columbus, Ohio.
In an interview, Dr. Saad emphasized that an important goal of the guidelines is to encourage general oncologists to recognize early signs of GVHD and refer potential candidates to transplant centers for further evaluation.
“We also urge oncologists who may be caring for patients after HCT to familiarize themselves with the varied manifestations of GVHD – a very common and significant posttransplant complication – and to consult with transplant providers to optimize their ongoing care. The guidelines explain how to diagnose and treat this condition in order to achieve the best possible outcomes,” guideline panel member Alison W. Loren, MD, director of blood and marrow transplantation at Abraham Cancer Center, University of Pennsylvania, Philadelphia, said in a statement.
The guideline includes links to other NCCN guidelines for diseases where HCT is a common therapeutic option, including leukemias, myeloid malignancies, lymphomas, central nervous system cancers, and testicular cancer.
The HCT guideline includes:
- Pretransplant recipient evaluation with recommendations for clinical assessment and imaging.
- Diagnosis and workup of GVHD, with separate algorithms for suspected acute or chronic GVHD.
- Specific interventions for management of acute GVHD with corticosteroids or other systemic agents.
- Chronic GVHD diagnosis by organ site and symptoms, with a severity scoring system.
- Chronic GVHD steroid response definitions and criteria.
- Suggested systemic agents for steroid-refractory GVHD.
One feature that is unusual for an NCCN guideline document is a page of photographs to assist clinicians in diagnosing range-of-motion abnormalities in the shoulder, elbow, hand, and ankle of patients with suspected or confirmed GVHD. Dr. Saad said that future iterations of the guideline will include additional photos to help clinicians develop a visual repertoire of potential GVHD signs.
Future versions will also include a discussion section and more comprehensive information on other common complications following HCT transplant, as well as management of posttransplant relapse.
Ideally, the guideline will help clinicians document and justify clinical decisions surrounding HCT and GVHD management in discussions with third-party payers, Dr. Saad said.
“Sometimes we struggle with payers when we want to use a certain modality to treat GVHD, and they respond ‘that’s not approved,’ or ‘that’s not a common indication,’ et cetera,” he said. “What we’re trying to put here are the commonly used therapies that most, but not all, experts agree on, and we can use this to negotiate with payers.”
He also emphasized that the guideline is meant to be instructive rather than prescriptive and is not meant to hinder innovations that may emerge from clinical trials.
“We would appreciate any feedback from non-NCCN centers as well as experts at NCCN centers, and we’ll be more than happy to address any concerns or criticisms they have,” he said.
Dr. Saad reported financial relationships with Actinium and Incysus Biomedical. Dr. Loren has previously reported having no disclosures.
Recommendations for the diagnosis and management of acute and chronic graft-versus-host disease (GVHD) are the central focus of the first National Comprehensive Cancer Network (NCCN) guideline on hematopoietic cell transplantation.
The guideline presents detailed recommendations for the evaluation of hematopoietic cell transplant (HCT) recipients, and an extensive section on the diagnosis and workup of GVHD, including information on staging and grading of acute GVHD, grading of chronic GVHD, treatment response criteria, and suggested systemic therapies for steroid-refractory disease.
“We wanted to both build on the commonly used approach to stage and treat graft-versus-host disease, and make sure that this information is readily available for physicians-in-training and young physicians who are learning about transplants,” said guideline committee chair Ayman Saad, MB BCh, of The Ohio State University Comprehensive Cancer Center, James Cancer Hospital and Solove Research Institute in Columbus, Ohio.
In an interview, Dr. Saad emphasized that an important goal of the guidelines is to encourage general oncologists to recognize early signs of GVHD and refer potential candidates to transplant centers for further evaluation.
“We also urge oncologists who may be caring for patients after HCT to familiarize themselves with the varied manifestations of GVHD – a very common and significant posttransplant complication – and to consult with transplant providers to optimize their ongoing care. The guidelines explain how to diagnose and treat this condition in order to achieve the best possible outcomes,” guideline panel member Alison W. Loren, MD, director of blood and marrow transplantation at Abraham Cancer Center, University of Pennsylvania, Philadelphia, said in a statement.
The guideline includes links to other NCCN guidelines for diseases where HCT is a common therapeutic option, including leukemias, myeloid malignancies, lymphomas, central nervous system cancers, and testicular cancer.
The HCT guideline includes:
- Pretransplant recipient evaluation with recommendations for clinical assessment and imaging.
- Diagnosis and workup of GVHD, with separate algorithms for suspected acute or chronic GVHD.
- Specific interventions for management of acute GVHD with corticosteroids or other systemic agents.
- Chronic GVHD diagnosis by organ site and symptoms, with a severity scoring system.
- Chronic GVHD steroid response definitions and criteria.
- Suggested systemic agents for steroid-refractory GVHD.
One feature that is unusual for an NCCN guideline document is a page of photographs to assist clinicians in diagnosing range-of-motion abnormalities in the shoulder, elbow, hand, and ankle of patients with suspected or confirmed GVHD. Dr. Saad said that future iterations of the guideline will include additional photos to help clinicians develop a visual repertoire of potential GVHD signs.
Future versions will also include a discussion section and more comprehensive information on other common complications following HCT transplant, as well as management of posttransplant relapse.
Ideally, the guideline will help clinicians document and justify clinical decisions surrounding HCT and GVHD management in discussions with third-party payers, Dr. Saad said.
“Sometimes we struggle with payers when we want to use a certain modality to treat GVHD, and they respond ‘that’s not approved,’ or ‘that’s not a common indication,’ et cetera,” he said. “What we’re trying to put here are the commonly used therapies that most, but not all, experts agree on, and we can use this to negotiate with payers.”
He also emphasized that the guideline is meant to be instructive rather than prescriptive and is not meant to hinder innovations that may emerge from clinical trials.
“We would appreciate any feedback from non-NCCN centers as well as experts at NCCN centers, and we’ll be more than happy to address any concerns or criticisms they have,” he said.
Dr. Saad reported financial relationships with Actinium and Incysus Biomedical. Dr. Loren has previously reported having no disclosures.
Updated international consensus recommendations on management of acute upper GI bleeding
Guidelines on the management of acute upper gastrointestinal bleeding have been updated, including recommendations on managing patients on antiplatelet or anticoagulant therapy and on use of endoscopy and new therapeutic approaches.
Writing in Annals of Internal Medicine, an international group of experts published an update to the 2010 International Consensus Recommendations on the Management of Patients With Nonvariceal Upper Gastrointestinal Bleeding, with a focus on resuscitation and risk assessment; pre-endoscopic, endoscopic, and pharmacologic management; and secondary prophylaxis.
Alan N. Barkun, MDCM, MSc, from McGill University, Montreal, and coauthors first recommended that fluid resuscitation should be initiated in patients with acute upper gastrointestinal bleeding and hemodynamic instability to avoid hemorrhagic shock and restore end-organ perfusion and tissue oxygenation while the bleeding is brought under control.
They acknowledged the uncertainty around whether colloid or crystalloid fluid should be used, but suggested routine use of colloids was not justified because they were more expensive and did not appear to increase survival.
On the question of whether the resuscitation should be aggressive or restrictive in its timing and rate, the group said there was not enough evidence to support a recommendation on this. “The important issue in patients with hemorrhagic shock due to trauma or UGIB [upper gastrointestinal bleeding] is to stop the bleeding while minimizing hemodynamic compromise,” they wrote.
They also advised blood transfusions in patients with a hemoglobin level below 80 g/L who did not have underlying cardiovascular disease, but suggested a higher hemoglobin threshold for those with underlying cardiovascular disease.
The second recommendation was that patients with a Glasgow Blatchford score of 1 or less were at very low risk for rebleeding and mortality, and these patients may therefore not need hospitalization or inpatient endoscopy. They advised against using the AIMS65 prognostic score for this purpose because it was designed to identify patients at high risk of death, not those at low risk for safe discharge.
In regard to endoscopic management, they advocated that all patients with acute upper gastrointestinal bleeding – whether low or high risk – undergo endoscopy within 24 hours of presentation. This was even more urgent in patients being treated with anticoagulants. “Because of the recognized benefits of early endoscopy, coagulopathy should be treated as necessary but endoscopy should not be delayed,” they wrote.
Patients with acutely bleeding ulcers with high-risk stigmata should undergo endoscopic therapy preferably with thermocoagulation or sclerosant injection, or with hemoclips depending on the bleeding location and patient characteristics.
The group also included two conditional recommendations, based on very-low-quality evidence, that patients with actively bleeding ulcers receive TC-325 hemostatic powder as temporizing therapy to stop the bleeding if conventional endoscopic therapies aren’t available or fail. However, they stressed that TC-325 should not be used as a single therapeutic strategy.
Because of a lack of efficacy data and low availability of expertise in the technology, the authors said they could not make a recommendation for or against using a Doppler endoscopic probe (DEP) to assess the need for further endoscopic therapy.
“The group generally agreed that although making a recommendation for or against using DEP to manage UGIB is premature, DEP has the potential to alter the usual approach to visually assessing bleeding lesion risk when evaluating the need for, and adequacy of, endoscopic hemostasis.”
The guidelines also addressed the issue of pharmacologic management of acute upper gastrointestinal bleeding. They strongly recommended that patients with bleeding ulcers and high-risk stigmata who have undergone successful endoscopic therapy should then receive an intravenous loading dose of proton pump inhibitor (PPI) therapy, followed by continuous intravenous infusion.
“Cost-effectiveness studies have suggested that high-dose intravenous PPIs after successful endoscopic hemostasis improve outcomes at a modest cost increase relative to non–high-dose intravenous or oral PPI strategies,” they wrote.
A second conditional recommendation, based on very-low-quality evidence, was that patients with a bleeding ulcer who were at high risk for rebleeding be also treated twice-daily with oral PPIs for 2 weeks, then once-daily. They also recommended patients on cardiovascular prophylaxis with single or dual antiplatelet therapy or anticoagulant therapy be given PPIs.
“The consensus group concluded that, for high-risk patients with an ongoing need for anticoagulants, the evidence suggests that the benefits of secondary prophylaxis outweigh the risks.”
The group was supported by a grant from CIHR Institute of Nutrition, Metabolism and Diabetes and from the Saudi Gastroenterology Association. Nine authors declared grants, personal fees, honoraria and other funding from the pharmaceutical and medical device sector outside the submitted work. No other conflicts of interest were declared.
SOURCE: Barkun A et al. Ann Intern Med 2019, October 22. doi: 10.7326/M19-1795.
These updated consensus guidelines provide a rigorous review of evidence on managing nonvariceal upper gastrointestinal bleeding. The recommendations for patients on anticoagulant or antiplatelet therapy will be particularly helpful because of increasing use of these medications. The advice on proton pump inhibitor therapy in patients on these drugs who have had previous ulcer bleeding can help allay concerns about possible integrations between PPIs and clopidogrel.
While the guidelines recommend using the Glasgow Blatchford scale to guide hospitalization decisions, prognostic scores are not commonly used in the emergency department, and many patients present with a Glasgow Blatchford score greater than 1, so this tool may have little impact on hospitalization rates. More studies are needed to compare clinical judgment with these prognostic scores.
Angel Lanas, MD, is from the University Clinic Hospital at the University of Zaragoza (Spain). These comments are adapted from an accompanying editorial (Ann Intern Med 2019, October 22. doi: 10.7326/M19-2789). Dr. Lanas declared unrelated personal fees from the pharmaceutical sector.
These updated consensus guidelines provide a rigorous review of evidence on managing nonvariceal upper gastrointestinal bleeding. The recommendations for patients on anticoagulant or antiplatelet therapy will be particularly helpful because of increasing use of these medications. The advice on proton pump inhibitor therapy in patients on these drugs who have had previous ulcer bleeding can help allay concerns about possible integrations between PPIs and clopidogrel.
While the guidelines recommend using the Glasgow Blatchford scale to guide hospitalization decisions, prognostic scores are not commonly used in the emergency department, and many patients present with a Glasgow Blatchford score greater than 1, so this tool may have little impact on hospitalization rates. More studies are needed to compare clinical judgment with these prognostic scores.
Angel Lanas, MD, is from the University Clinic Hospital at the University of Zaragoza (Spain). These comments are adapted from an accompanying editorial (Ann Intern Med 2019, October 22. doi: 10.7326/M19-2789). Dr. Lanas declared unrelated personal fees from the pharmaceutical sector.
These updated consensus guidelines provide a rigorous review of evidence on managing nonvariceal upper gastrointestinal bleeding. The recommendations for patients on anticoagulant or antiplatelet therapy will be particularly helpful because of increasing use of these medications. The advice on proton pump inhibitor therapy in patients on these drugs who have had previous ulcer bleeding can help allay concerns about possible integrations between PPIs and clopidogrel.
While the guidelines recommend using the Glasgow Blatchford scale to guide hospitalization decisions, prognostic scores are not commonly used in the emergency department, and many patients present with a Glasgow Blatchford score greater than 1, so this tool may have little impact on hospitalization rates. More studies are needed to compare clinical judgment with these prognostic scores.
Angel Lanas, MD, is from the University Clinic Hospital at the University of Zaragoza (Spain). These comments are adapted from an accompanying editorial (Ann Intern Med 2019, October 22. doi: 10.7326/M19-2789). Dr. Lanas declared unrelated personal fees from the pharmaceutical sector.
Guidelines on the management of acute upper gastrointestinal bleeding have been updated, including recommendations on managing patients on antiplatelet or anticoagulant therapy and on use of endoscopy and new therapeutic approaches.
Writing in Annals of Internal Medicine, an international group of experts published an update to the 2010 International Consensus Recommendations on the Management of Patients With Nonvariceal Upper Gastrointestinal Bleeding, with a focus on resuscitation and risk assessment; pre-endoscopic, endoscopic, and pharmacologic management; and secondary prophylaxis.
Alan N. Barkun, MDCM, MSc, from McGill University, Montreal, and coauthors first recommended that fluid resuscitation should be initiated in patients with acute upper gastrointestinal bleeding and hemodynamic instability to avoid hemorrhagic shock and restore end-organ perfusion and tissue oxygenation while the bleeding is brought under control.
They acknowledged the uncertainty around whether colloid or crystalloid fluid should be used, but suggested routine use of colloids was not justified because they were more expensive and did not appear to increase survival.
On the question of whether the resuscitation should be aggressive or restrictive in its timing and rate, the group said there was not enough evidence to support a recommendation on this. “The important issue in patients with hemorrhagic shock due to trauma or UGIB [upper gastrointestinal bleeding] is to stop the bleeding while minimizing hemodynamic compromise,” they wrote.
They also advised blood transfusions in patients with a hemoglobin level below 80 g/L who did not have underlying cardiovascular disease, but suggested a higher hemoglobin threshold for those with underlying cardiovascular disease.
The second recommendation was that patients with a Glasgow Blatchford score of 1 or less were at very low risk for rebleeding and mortality, and these patients may therefore not need hospitalization or inpatient endoscopy. They advised against using the AIMS65 prognostic score for this purpose because it was designed to identify patients at high risk of death, not those at low risk for safe discharge.
In regard to endoscopic management, they advocated that all patients with acute upper gastrointestinal bleeding – whether low or high risk – undergo endoscopy within 24 hours of presentation. This was even more urgent in patients being treated with anticoagulants. “Because of the recognized benefits of early endoscopy, coagulopathy should be treated as necessary but endoscopy should not be delayed,” they wrote.
Patients with acutely bleeding ulcers with high-risk stigmata should undergo endoscopic therapy preferably with thermocoagulation or sclerosant injection, or with hemoclips depending on the bleeding location and patient characteristics.
The group also included two conditional recommendations, based on very-low-quality evidence, that patients with actively bleeding ulcers receive TC-325 hemostatic powder as temporizing therapy to stop the bleeding if conventional endoscopic therapies aren’t available or fail. However, they stressed that TC-325 should not be used as a single therapeutic strategy.
Because of a lack of efficacy data and low availability of expertise in the technology, the authors said they could not make a recommendation for or against using a Doppler endoscopic probe (DEP) to assess the need for further endoscopic therapy.
“The group generally agreed that although making a recommendation for or against using DEP to manage UGIB is premature, DEP has the potential to alter the usual approach to visually assessing bleeding lesion risk when evaluating the need for, and adequacy of, endoscopic hemostasis.”
The guidelines also addressed the issue of pharmacologic management of acute upper gastrointestinal bleeding. They strongly recommended that patients with bleeding ulcers and high-risk stigmata who have undergone successful endoscopic therapy should then receive an intravenous loading dose of proton pump inhibitor (PPI) therapy, followed by continuous intravenous infusion.
“Cost-effectiveness studies have suggested that high-dose intravenous PPIs after successful endoscopic hemostasis improve outcomes at a modest cost increase relative to non–high-dose intravenous or oral PPI strategies,” they wrote.
A second conditional recommendation, based on very-low-quality evidence, was that patients with a bleeding ulcer who were at high risk for rebleeding be also treated twice-daily with oral PPIs for 2 weeks, then once-daily. They also recommended patients on cardiovascular prophylaxis with single or dual antiplatelet therapy or anticoagulant therapy be given PPIs.
“The consensus group concluded that, for high-risk patients with an ongoing need for anticoagulants, the evidence suggests that the benefits of secondary prophylaxis outweigh the risks.”
The group was supported by a grant from CIHR Institute of Nutrition, Metabolism and Diabetes and from the Saudi Gastroenterology Association. Nine authors declared grants, personal fees, honoraria and other funding from the pharmaceutical and medical device sector outside the submitted work. No other conflicts of interest were declared.
SOURCE: Barkun A et al. Ann Intern Med 2019, October 22. doi: 10.7326/M19-1795.
Guidelines on the management of acute upper gastrointestinal bleeding have been updated, including recommendations on managing patients on antiplatelet or anticoagulant therapy and on use of endoscopy and new therapeutic approaches.
Writing in Annals of Internal Medicine, an international group of experts published an update to the 2010 International Consensus Recommendations on the Management of Patients With Nonvariceal Upper Gastrointestinal Bleeding, with a focus on resuscitation and risk assessment; pre-endoscopic, endoscopic, and pharmacologic management; and secondary prophylaxis.
Alan N. Barkun, MDCM, MSc, from McGill University, Montreal, and coauthors first recommended that fluid resuscitation should be initiated in patients with acute upper gastrointestinal bleeding and hemodynamic instability to avoid hemorrhagic shock and restore end-organ perfusion and tissue oxygenation while the bleeding is brought under control.
They acknowledged the uncertainty around whether colloid or crystalloid fluid should be used, but suggested routine use of colloids was not justified because they were more expensive and did not appear to increase survival.
On the question of whether the resuscitation should be aggressive or restrictive in its timing and rate, the group said there was not enough evidence to support a recommendation on this. “The important issue in patients with hemorrhagic shock due to trauma or UGIB [upper gastrointestinal bleeding] is to stop the bleeding while minimizing hemodynamic compromise,” they wrote.
They also advised blood transfusions in patients with a hemoglobin level below 80 g/L who did not have underlying cardiovascular disease, but suggested a higher hemoglobin threshold for those with underlying cardiovascular disease.
The second recommendation was that patients with a Glasgow Blatchford score of 1 or less were at very low risk for rebleeding and mortality, and these patients may therefore not need hospitalization or inpatient endoscopy. They advised against using the AIMS65 prognostic score for this purpose because it was designed to identify patients at high risk of death, not those at low risk for safe discharge.
In regard to endoscopic management, they advocated that all patients with acute upper gastrointestinal bleeding – whether low or high risk – undergo endoscopy within 24 hours of presentation. This was even more urgent in patients being treated with anticoagulants. “Because of the recognized benefits of early endoscopy, coagulopathy should be treated as necessary but endoscopy should not be delayed,” they wrote.
Patients with acutely bleeding ulcers with high-risk stigmata should undergo endoscopic therapy preferably with thermocoagulation or sclerosant injection, or with hemoclips depending on the bleeding location and patient characteristics.
The group also included two conditional recommendations, based on very-low-quality evidence, that patients with actively bleeding ulcers receive TC-325 hemostatic powder as temporizing therapy to stop the bleeding if conventional endoscopic therapies aren’t available or fail. However, they stressed that TC-325 should not be used as a single therapeutic strategy.
Because of a lack of efficacy data and low availability of expertise in the technology, the authors said they could not make a recommendation for or against using a Doppler endoscopic probe (DEP) to assess the need for further endoscopic therapy.
“The group generally agreed that although making a recommendation for or against using DEP to manage UGIB is premature, DEP has the potential to alter the usual approach to visually assessing bleeding lesion risk when evaluating the need for, and adequacy of, endoscopic hemostasis.”
The guidelines also addressed the issue of pharmacologic management of acute upper gastrointestinal bleeding. They strongly recommended that patients with bleeding ulcers and high-risk stigmata who have undergone successful endoscopic therapy should then receive an intravenous loading dose of proton pump inhibitor (PPI) therapy, followed by continuous intravenous infusion.
“Cost-effectiveness studies have suggested that high-dose intravenous PPIs after successful endoscopic hemostasis improve outcomes at a modest cost increase relative to non–high-dose intravenous or oral PPI strategies,” they wrote.
A second conditional recommendation, based on very-low-quality evidence, was that patients with a bleeding ulcer who were at high risk for rebleeding be also treated twice-daily with oral PPIs for 2 weeks, then once-daily. They also recommended patients on cardiovascular prophylaxis with single or dual antiplatelet therapy or anticoagulant therapy be given PPIs.
“The consensus group concluded that, for high-risk patients with an ongoing need for anticoagulants, the evidence suggests that the benefits of secondary prophylaxis outweigh the risks.”
The group was supported by a grant from CIHR Institute of Nutrition, Metabolism and Diabetes and from the Saudi Gastroenterology Association. Nine authors declared grants, personal fees, honoraria and other funding from the pharmaceutical and medical device sector outside the submitted work. No other conflicts of interest were declared.
SOURCE: Barkun A et al. Ann Intern Med 2019, October 22. doi: 10.7326/M19-1795.
FROM ANNALS OF INTERNAL MEDICINE








