User login
FDA issues warnings to companies selling illegal Alzheimer’s treatments
The Food and Drug Administration has issued warning letters to 12 companies and advisory letters to 5 companies illegally selling more than 58 products claiming to treat Alzheimer’s disease.
The products, many of which are marketed as dietary supplements, are being sold in a variety of forms, including tablets, capsules, and oils. These drugs are either unapproved or mislabeled and claim to prevent, treat, or cure Alzheimer’s disease, as well as a number of other serious diseases and health conditions, in violation of the Federal Food, Drug, and Cosmetic Act.
“Alzheimer’s is a challenging disease that, unfortunately, has no cure. Any products making unproven drug claims could mislead consumers to believe that such therapies exist and keep them from accessing therapies that are known to help support the symptoms of the disease, or worse, as some fraudulent treatments can cause serious or even fatal injuries,” FDA Commissioner Scott Gottlieb, MD, said in a press release.
In an additional statement, Dr. Gottlieb detailed several new strategies for improving the safety and accuracy of dietary supplements, including efforts to more rapidly communicate to the public potential safety issues with dietary supplement products and to establish a flexible regulatory framework that promotes innovation and upholds product safety.
The Food and Drug Administration has issued warning letters to 12 companies and advisory letters to 5 companies illegally selling more than 58 products claiming to treat Alzheimer’s disease.
The products, many of which are marketed as dietary supplements, are being sold in a variety of forms, including tablets, capsules, and oils. These drugs are either unapproved or mislabeled and claim to prevent, treat, or cure Alzheimer’s disease, as well as a number of other serious diseases and health conditions, in violation of the Federal Food, Drug, and Cosmetic Act.
“Alzheimer’s is a challenging disease that, unfortunately, has no cure. Any products making unproven drug claims could mislead consumers to believe that such therapies exist and keep them from accessing therapies that are known to help support the symptoms of the disease, or worse, as some fraudulent treatments can cause serious or even fatal injuries,” FDA Commissioner Scott Gottlieb, MD, said in a press release.
In an additional statement, Dr. Gottlieb detailed several new strategies for improving the safety and accuracy of dietary supplements, including efforts to more rapidly communicate to the public potential safety issues with dietary supplement products and to establish a flexible regulatory framework that promotes innovation and upholds product safety.
The Food and Drug Administration has issued warning letters to 12 companies and advisory letters to 5 companies illegally selling more than 58 products claiming to treat Alzheimer’s disease.
The products, many of which are marketed as dietary supplements, are being sold in a variety of forms, including tablets, capsules, and oils. These drugs are either unapproved or mislabeled and claim to prevent, treat, or cure Alzheimer’s disease, as well as a number of other serious diseases and health conditions, in violation of the Federal Food, Drug, and Cosmetic Act.
“Alzheimer’s is a challenging disease that, unfortunately, has no cure. Any products making unproven drug claims could mislead consumers to believe that such therapies exist and keep them from accessing therapies that are known to help support the symptoms of the disease, or worse, as some fraudulent treatments can cause serious or even fatal injuries,” FDA Commissioner Scott Gottlieb, MD, said in a press release.
In an additional statement, Dr. Gottlieb detailed several new strategies for improving the safety and accuracy of dietary supplements, including efforts to more rapidly communicate to the public potential safety issues with dietary supplement products and to establish a flexible regulatory framework that promotes innovation and upholds product safety.
United States now over 100 measles cases for the year
according to the Centers for Disease Control and Prevention.
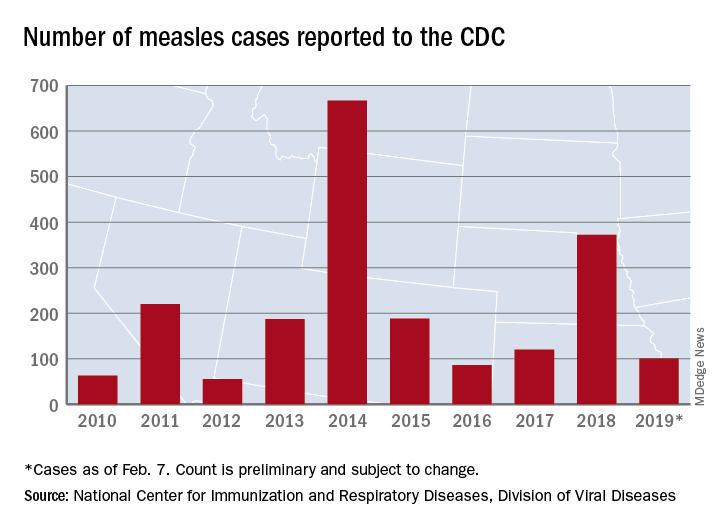
Just over half of the cases in 2019 have occurred in Clark County, Wash., which has reported 53 cases. That outbreak led Gov. Jay Inslee to declare a public health emergency for the entire state on Jan. 25.
The cases in Washington represent one of the five outbreaks – the CDC defines an outbreak as three or more cases – that have occurred so far this year, with three reported in New York State (Rockland County, Monroe County, and New York City) and one in Texas, which has been spread out over five counties, the CDC reported Feb. 11.
“These outbreaks are linked to travelers who brought measles back from other countries such as Israel and Ukraine, where large measles outbreaks are occurring,” the CDC noted. The other states with confirmed cases are California, Colorado, Connecticut, Georgia, Illinois, New Jersey, and Oregon.
In a video released Feb. 1, Surgeon General Jerome Adams stressed the importance of getting vaccinated and noted that an infected person can transmit the measles virus up to 4 days before he or she develops symptoms.
according to the Centers for Disease Control and Prevention.

Just over half of the cases in 2019 have occurred in Clark County, Wash., which has reported 53 cases. That outbreak led Gov. Jay Inslee to declare a public health emergency for the entire state on Jan. 25.
The cases in Washington represent one of the five outbreaks – the CDC defines an outbreak as three or more cases – that have occurred so far this year, with three reported in New York State (Rockland County, Monroe County, and New York City) and one in Texas, which has been spread out over five counties, the CDC reported Feb. 11.
“These outbreaks are linked to travelers who brought measles back from other countries such as Israel and Ukraine, where large measles outbreaks are occurring,” the CDC noted. The other states with confirmed cases are California, Colorado, Connecticut, Georgia, Illinois, New Jersey, and Oregon.
In a video released Feb. 1, Surgeon General Jerome Adams stressed the importance of getting vaccinated and noted that an infected person can transmit the measles virus up to 4 days before he or she develops symptoms.
according to the Centers for Disease Control and Prevention.

Just over half of the cases in 2019 have occurred in Clark County, Wash., which has reported 53 cases. That outbreak led Gov. Jay Inslee to declare a public health emergency for the entire state on Jan. 25.
The cases in Washington represent one of the five outbreaks – the CDC defines an outbreak as three or more cases – that have occurred so far this year, with three reported in New York State (Rockland County, Monroe County, and New York City) and one in Texas, which has been spread out over five counties, the CDC reported Feb. 11.
“These outbreaks are linked to travelers who brought measles back from other countries such as Israel and Ukraine, where large measles outbreaks are occurring,” the CDC noted. The other states with confirmed cases are California, Colorado, Connecticut, Georgia, Illinois, New Jersey, and Oregon.
In a video released Feb. 1, Surgeon General Jerome Adams stressed the importance of getting vaccinated and noted that an infected person can transmit the measles virus up to 4 days before he or she develops symptoms.
E-cig use reverses progress in reducing tobacco use in teens
A significant increase during 2017-2018 in e-cigarette use among U.S. youths has erased recent progress in reducing overall tobacco product use in this age group, a study from the Centers for Disease Control and Prevention has found.
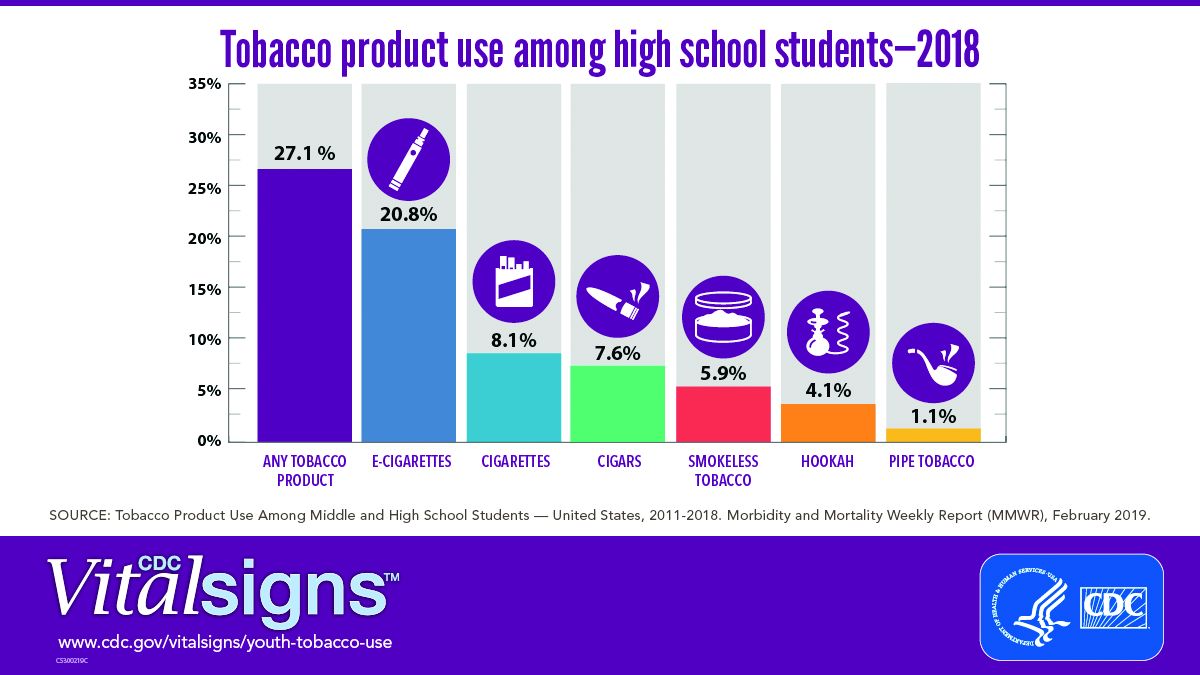
E-cigarettes are driving the trend. About 4 million high school students in the United States reported using any tobacco product in the last 30 days, and 3 million of them reported using e-cigarettes, according to a Vital Signs document published by the CDC on Feb. 11 in its Morbidity and Mortality Weekly Report.*
In addition, many high school students who use e-cigarettes use them often; 28% reported using the products at least 20 times in the past 28 days, up from 20% in 2017.
“Any use of any tobacco product is unsafe for teens,” Anne Schuchat, MD, principal deputy director of the CDC, said in a teleconference to present the findings. Nicotine is highly addictive and can harm brain development in youth, including capacity for learning, memory, and attention, she said.
The rise in e-cigarette use corresponds with the rise in marketing and availability of e-cigarette devices such as JUUL, which dispense nicotine via liquid refill pods available in flavors including strawberry and cotton candy, said Brian King, MPH, PhD, deputy director for research translation at the CDC’s Office on Smoking and Health.
“The advertising will lead a horse to water, the flavors will make them drink, and the nicotine will keep them coming back for more,” said Dr. King.
Approximately 27.1% of high school students and 7.2% of middle school students used a tobacco product in 2018, a significant increase from 2017 data, and with a major increase in e-cigarette use.
No change was noted in the use of other tobacco products, including cigarettes, from 2017 to 2018, according to the report. However, conventional cigarettes remained the most common companion product to e-cigarettes for youth who use two or more tobacco products (two in five high school students and one in three middle school students in 2018). From a demographic standpoint, e-cigarette use was highest among males, whites, and high school students.
Tobacco use in teens is trending in the direction of wiping out the progress made in recent years to reduce exposure to youths. The report noted, “The prevalence of e-cigarette use by U.S. high school students had peaked in 2015 before declining by 29% during 2015-2016 (from 16% to 11.3%); this decline was the first ever recorded for e-cigarette use among youths in the NYTS since monitoring began, and it was subsequently sustained during 2016-2017). However, current e-cigarette use increased by 77.8% among high school students and 48.5% among middle school students during 2017-2018, erasing the progress in reducing e-cigarette use, as well as any tobacco product use, that had occurred in prior years.”
The CDC and the Food and Drug Administration are taking action to curb the rise in e-cigarette use in youth in particular by seeking regulations to make the products less accessible, raising prices, and banning most flavorings, said Dr. Schuchat.
“We have targeted companies engaged in kid friendly marketing,” said Mitch Zeller, JD, director of the Center for Tobacco Products for the FDA.
In a statement published simultaneously with the Vital Signs study, FDA Commissioner Scott Gottlieb, MD, emphasized the link between e-cigarette use in teens and the potential for future tobacco use. “The kids using e-cigarettes are children who rejected conventional cigarettes, but don’t see the same stigma associated with the use of e-cigarettes. But now, having become exposed to nicotine through e-cigs, they will be more likely to smoke.” Dr. Gottlieb declared, “I will not allow a generation of children to become addicted to nicotine through e-cigarettes. We must stop the trends of youth e-cigarette use from continuing to build and will take whatever action is necessary to ensure these kids don’t become future smokers.” He reviewed steps taken in the past year by the FDA to counter tobacco use in teens but he warned of future actions that may need to be taken: “If these youth use trends continue, we’ll be forced to consider regulatory steps that could constrain or even foreclose the opportunities for currently addicted adult smokers to have the same level of access to these products that they now enjoy. I recognize that such a move could come with significant impacts to adult smokers.”
In the meantime, however, parents, teachers, community leaders, and health care providers are on the front lines and can make a difference in protecting youth and curbing nicotine use, Dr. King said.
One of the most important things clinicians can do is to ask young patients specifically about e-cigarette use, he emphasized. Learn and use the terminology the kids are using; ask, “Do you use JUUL?” If they are using these products, “make sure they know they are dangerous,” and can harm the developing brain, he said.
Although there are no currently approved medications to treat nicotine addiction in youth, research suggests that behavioral counseling, as well as reinforcement of the danger of nicotine from parents and other people of influence, can help, Dr. King said.
The Vital Signs report is based on data from the 2011-2018 National Youth Tobacco Survey, which assesses current use of cigarettes, cigars, smokeless tobacco, e-cigarettes, hookahs, pipe tobacco, and bidis among a nationally representative sample of middle and high school students in the United States. The findings were analyzed by the CDC, FDA, and the National Cancer Institute.
SOURCE: Gentzke AS et al. MMWR 2019 Feb 11. doi: 10.15585/mmwr.mm6806e1.
*Correction 2/13/2019 An earlier version of this article misstated the number of students using e-cigarettes as a proportion of all teen tobacco users.
A significant increase during 2017-2018 in e-cigarette use among U.S. youths has erased recent progress in reducing overall tobacco product use in this age group, a study from the Centers for Disease Control and Prevention has found.

E-cigarettes are driving the trend. About 4 million high school students in the United States reported using any tobacco product in the last 30 days, and 3 million of them reported using e-cigarettes, according to a Vital Signs document published by the CDC on Feb. 11 in its Morbidity and Mortality Weekly Report.*
In addition, many high school students who use e-cigarettes use them often; 28% reported using the products at least 20 times in the past 28 days, up from 20% in 2017.
“Any use of any tobacco product is unsafe for teens,” Anne Schuchat, MD, principal deputy director of the CDC, said in a teleconference to present the findings. Nicotine is highly addictive and can harm brain development in youth, including capacity for learning, memory, and attention, she said.
The rise in e-cigarette use corresponds with the rise in marketing and availability of e-cigarette devices such as JUUL, which dispense nicotine via liquid refill pods available in flavors including strawberry and cotton candy, said Brian King, MPH, PhD, deputy director for research translation at the CDC’s Office on Smoking and Health.
“The advertising will lead a horse to water, the flavors will make them drink, and the nicotine will keep them coming back for more,” said Dr. King.
Approximately 27.1% of high school students and 7.2% of middle school students used a tobacco product in 2018, a significant increase from 2017 data, and with a major increase in e-cigarette use.
No change was noted in the use of other tobacco products, including cigarettes, from 2017 to 2018, according to the report. However, conventional cigarettes remained the most common companion product to e-cigarettes for youth who use two or more tobacco products (two in five high school students and one in three middle school students in 2018). From a demographic standpoint, e-cigarette use was highest among males, whites, and high school students.
Tobacco use in teens is trending in the direction of wiping out the progress made in recent years to reduce exposure to youths. The report noted, “The prevalence of e-cigarette use by U.S. high school students had peaked in 2015 before declining by 29% during 2015-2016 (from 16% to 11.3%); this decline was the first ever recorded for e-cigarette use among youths in the NYTS since monitoring began, and it was subsequently sustained during 2016-2017). However, current e-cigarette use increased by 77.8% among high school students and 48.5% among middle school students during 2017-2018, erasing the progress in reducing e-cigarette use, as well as any tobacco product use, that had occurred in prior years.”
The CDC and the Food and Drug Administration are taking action to curb the rise in e-cigarette use in youth in particular by seeking regulations to make the products less accessible, raising prices, and banning most flavorings, said Dr. Schuchat.
“We have targeted companies engaged in kid friendly marketing,” said Mitch Zeller, JD, director of the Center for Tobacco Products for the FDA.
In a statement published simultaneously with the Vital Signs study, FDA Commissioner Scott Gottlieb, MD, emphasized the link between e-cigarette use in teens and the potential for future tobacco use. “The kids using e-cigarettes are children who rejected conventional cigarettes, but don’t see the same stigma associated with the use of e-cigarettes. But now, having become exposed to nicotine through e-cigs, they will be more likely to smoke.” Dr. Gottlieb declared, “I will not allow a generation of children to become addicted to nicotine through e-cigarettes. We must stop the trends of youth e-cigarette use from continuing to build and will take whatever action is necessary to ensure these kids don’t become future smokers.” He reviewed steps taken in the past year by the FDA to counter tobacco use in teens but he warned of future actions that may need to be taken: “If these youth use trends continue, we’ll be forced to consider regulatory steps that could constrain or even foreclose the opportunities for currently addicted adult smokers to have the same level of access to these products that they now enjoy. I recognize that such a move could come with significant impacts to adult smokers.”
In the meantime, however, parents, teachers, community leaders, and health care providers are on the front lines and can make a difference in protecting youth and curbing nicotine use, Dr. King said.
One of the most important things clinicians can do is to ask young patients specifically about e-cigarette use, he emphasized. Learn and use the terminology the kids are using; ask, “Do you use JUUL?” If they are using these products, “make sure they know they are dangerous,” and can harm the developing brain, he said.
Although there are no currently approved medications to treat nicotine addiction in youth, research suggests that behavioral counseling, as well as reinforcement of the danger of nicotine from parents and other people of influence, can help, Dr. King said.
The Vital Signs report is based on data from the 2011-2018 National Youth Tobacco Survey, which assesses current use of cigarettes, cigars, smokeless tobacco, e-cigarettes, hookahs, pipe tobacco, and bidis among a nationally representative sample of middle and high school students in the United States. The findings were analyzed by the CDC, FDA, and the National Cancer Institute.
SOURCE: Gentzke AS et al. MMWR 2019 Feb 11. doi: 10.15585/mmwr.mm6806e1.
*Correction 2/13/2019 An earlier version of this article misstated the number of students using e-cigarettes as a proportion of all teen tobacco users.
A significant increase during 2017-2018 in e-cigarette use among U.S. youths has erased recent progress in reducing overall tobacco product use in this age group, a study from the Centers for Disease Control and Prevention has found.

E-cigarettes are driving the trend. About 4 million high school students in the United States reported using any tobacco product in the last 30 days, and 3 million of them reported using e-cigarettes, according to a Vital Signs document published by the CDC on Feb. 11 in its Morbidity and Mortality Weekly Report.*
In addition, many high school students who use e-cigarettes use them often; 28% reported using the products at least 20 times in the past 28 days, up from 20% in 2017.
“Any use of any tobacco product is unsafe for teens,” Anne Schuchat, MD, principal deputy director of the CDC, said in a teleconference to present the findings. Nicotine is highly addictive and can harm brain development in youth, including capacity for learning, memory, and attention, she said.
The rise in e-cigarette use corresponds with the rise in marketing and availability of e-cigarette devices such as JUUL, which dispense nicotine via liquid refill pods available in flavors including strawberry and cotton candy, said Brian King, MPH, PhD, deputy director for research translation at the CDC’s Office on Smoking and Health.
“The advertising will lead a horse to water, the flavors will make them drink, and the nicotine will keep them coming back for more,” said Dr. King.
Approximately 27.1% of high school students and 7.2% of middle school students used a tobacco product in 2018, a significant increase from 2017 data, and with a major increase in e-cigarette use.
No change was noted in the use of other tobacco products, including cigarettes, from 2017 to 2018, according to the report. However, conventional cigarettes remained the most common companion product to e-cigarettes for youth who use two or more tobacco products (two in five high school students and one in three middle school students in 2018). From a demographic standpoint, e-cigarette use was highest among males, whites, and high school students.
Tobacco use in teens is trending in the direction of wiping out the progress made in recent years to reduce exposure to youths. The report noted, “The prevalence of e-cigarette use by U.S. high school students had peaked in 2015 before declining by 29% during 2015-2016 (from 16% to 11.3%); this decline was the first ever recorded for e-cigarette use among youths in the NYTS since monitoring began, and it was subsequently sustained during 2016-2017). However, current e-cigarette use increased by 77.8% among high school students and 48.5% among middle school students during 2017-2018, erasing the progress in reducing e-cigarette use, as well as any tobacco product use, that had occurred in prior years.”
The CDC and the Food and Drug Administration are taking action to curb the rise in e-cigarette use in youth in particular by seeking regulations to make the products less accessible, raising prices, and banning most flavorings, said Dr. Schuchat.
“We have targeted companies engaged in kid friendly marketing,” said Mitch Zeller, JD, director of the Center for Tobacco Products for the FDA.
In a statement published simultaneously with the Vital Signs study, FDA Commissioner Scott Gottlieb, MD, emphasized the link between e-cigarette use in teens and the potential for future tobacco use. “The kids using e-cigarettes are children who rejected conventional cigarettes, but don’t see the same stigma associated with the use of e-cigarettes. But now, having become exposed to nicotine through e-cigs, they will be more likely to smoke.” Dr. Gottlieb declared, “I will not allow a generation of children to become addicted to nicotine through e-cigarettes. We must stop the trends of youth e-cigarette use from continuing to build and will take whatever action is necessary to ensure these kids don’t become future smokers.” He reviewed steps taken in the past year by the FDA to counter tobacco use in teens but he warned of future actions that may need to be taken: “If these youth use trends continue, we’ll be forced to consider regulatory steps that could constrain or even foreclose the opportunities for currently addicted adult smokers to have the same level of access to these products that they now enjoy. I recognize that such a move could come with significant impacts to adult smokers.”
In the meantime, however, parents, teachers, community leaders, and health care providers are on the front lines and can make a difference in protecting youth and curbing nicotine use, Dr. King said.
One of the most important things clinicians can do is to ask young patients specifically about e-cigarette use, he emphasized. Learn and use the terminology the kids are using; ask, “Do you use JUUL?” If they are using these products, “make sure they know they are dangerous,” and can harm the developing brain, he said.
Although there are no currently approved medications to treat nicotine addiction in youth, research suggests that behavioral counseling, as well as reinforcement of the danger of nicotine from parents and other people of influence, can help, Dr. King said.
The Vital Signs report is based on data from the 2011-2018 National Youth Tobacco Survey, which assesses current use of cigarettes, cigars, smokeless tobacco, e-cigarettes, hookahs, pipe tobacco, and bidis among a nationally representative sample of middle and high school students in the United States. The findings were analyzed by the CDC, FDA, and the National Cancer Institute.
SOURCE: Gentzke AS et al. MMWR 2019 Feb 11. doi: 10.15585/mmwr.mm6806e1.
*Correction 2/13/2019 An earlier version of this article misstated the number of students using e-cigarettes as a proportion of all teen tobacco users.
FROM CDC VITAL SIGNS REPORT
Flu activity hits seasonal high
Influenza activity increased for the third consecutive week and has now reached its highest point for the 2018-2019 flu season, according to the Centers for Disease Control and Prevention.
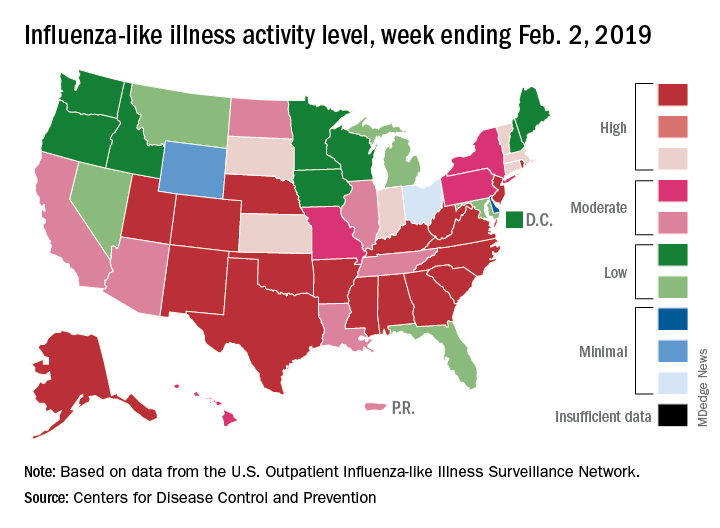
The proportion of outpatient visits for influenza-like illness (ILI) hit 4.3% for the week ending Feb. 2, which topped the previous high of 4.0% that was reached in late December (the national baseline rate is 2.2%). Outpatient ILI visits then dipped down to 3.1% after 2 weeks of decreases before rising again in mid-January, the CDC’s influenza division reported Feb. 8.
Season-high activity also was seen at the state level for the week ending Feb. 2. There were 18 states at level 10 on the CDC’s 1-10 scale of ILI activity, which was up from 16 the week before, and a total of 24 states were in the high range from 8-10, compared with 23 for the previous week. The geographic spread of influenza was reported as widespread in 47 states and Puerto Rico, the CDC said.
Four flu-related pediatric deaths were reported during the week ending Feb. 2, two of which occurred the previous week, which brings the total for the 2018-2019 season to 28, the CDC said.
There were 158 flu-related deaths among all ages during the week ending Jan. 26 – the latest for which such data are available – with reporting almost 75% complete. The previous week saw 177 overall flu deaths, with reporting for that week over 90% complete. During the corresponding weeks of the very severe 2017-2018 flu season, the overall death totals were 1,448 and 1,626, CDC data show.
Influenza activity increased for the third consecutive week and has now reached its highest point for the 2018-2019 flu season, according to the Centers for Disease Control and Prevention.

The proportion of outpatient visits for influenza-like illness (ILI) hit 4.3% for the week ending Feb. 2, which topped the previous high of 4.0% that was reached in late December (the national baseline rate is 2.2%). Outpatient ILI visits then dipped down to 3.1% after 2 weeks of decreases before rising again in mid-January, the CDC’s influenza division reported Feb. 8.
Season-high activity also was seen at the state level for the week ending Feb. 2. There were 18 states at level 10 on the CDC’s 1-10 scale of ILI activity, which was up from 16 the week before, and a total of 24 states were in the high range from 8-10, compared with 23 for the previous week. The geographic spread of influenza was reported as widespread in 47 states and Puerto Rico, the CDC said.
Four flu-related pediatric deaths were reported during the week ending Feb. 2, two of which occurred the previous week, which brings the total for the 2018-2019 season to 28, the CDC said.
There were 158 flu-related deaths among all ages during the week ending Jan. 26 – the latest for which such data are available – with reporting almost 75% complete. The previous week saw 177 overall flu deaths, with reporting for that week over 90% complete. During the corresponding weeks of the very severe 2017-2018 flu season, the overall death totals were 1,448 and 1,626, CDC data show.
Influenza activity increased for the third consecutive week and has now reached its highest point for the 2018-2019 flu season, according to the Centers for Disease Control and Prevention.

The proportion of outpatient visits for influenza-like illness (ILI) hit 4.3% for the week ending Feb. 2, which topped the previous high of 4.0% that was reached in late December (the national baseline rate is 2.2%). Outpatient ILI visits then dipped down to 3.1% after 2 weeks of decreases before rising again in mid-January, the CDC’s influenza division reported Feb. 8.
Season-high activity also was seen at the state level for the week ending Feb. 2. There were 18 states at level 10 on the CDC’s 1-10 scale of ILI activity, which was up from 16 the week before, and a total of 24 states were in the high range from 8-10, compared with 23 for the previous week. The geographic spread of influenza was reported as widespread in 47 states and Puerto Rico, the CDC said.
Four flu-related pediatric deaths were reported during the week ending Feb. 2, two of which occurred the previous week, which brings the total for the 2018-2019 season to 28, the CDC said.
There were 158 flu-related deaths among all ages during the week ending Jan. 26 – the latest for which such data are available – with reporting almost 75% complete. The previous week saw 177 overall flu deaths, with reporting for that week over 90% complete. During the corresponding weeks of the very severe 2017-2018 flu season, the overall death totals were 1,448 and 1,626, CDC data show.
FDA: 246 new reports on breast implant-associated lymphoma
The Food and Drug Administration has identified 457 unique cases of breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) and 9 related deaths since 2010, and received 246 new medical device reports (MDRs) regarding BIA-ALCL between September 2017 and September 2018, according to an update from the agency’s Center for Devices and Radiological Health.

That brings the total number of reports to 660; however, that number reflects duplicative cases, Binita Ashar, MD, a general surgeon and the director of the division of surgical devices at the center, said in a statement.
“These types of increases in the MDRs are to be expected and may include past cases that were not previously reported to the FDA,” Dr. Ashar said, addressing the high number of new reports. “The increased number of MDRs contributes to our evolving understanding of BIA-ALCL and represents a more thorough and comprehensive analysis.”
BIA-ALCL is a type of non-Hodgkin lymphoma and a known risk from breast implants that was first communicated by the FDA in 2011. Regular updates have been provided with respect to related medical device reports, cases, deaths, and known risks.
“We hope that this information prompts providers and patients to have important, informed conversations about breast implants and the risk of BIA-ALCL. At the same time, we remain committed to working in partnership with all stakeholders to continue to study, understand, and provide updates about this important public health issue,” Dr. Ashar said.
To that end, the center also issued a Letter to Health Care Providers to “encourage those who regularly treat patients, including primary care physicians and gynecologists, to learn about BIA-ALCL in patients with breast implants.”
Patients and providers are encouraged to file MDRs with the FDA via MedWatch, the FDA Safety Information and Adverse Event Reporting program, she said.
The Food and Drug Administration has identified 457 unique cases of breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) and 9 related deaths since 2010, and received 246 new medical device reports (MDRs) regarding BIA-ALCL between September 2017 and September 2018, according to an update from the agency’s Center for Devices and Radiological Health.

That brings the total number of reports to 660; however, that number reflects duplicative cases, Binita Ashar, MD, a general surgeon and the director of the division of surgical devices at the center, said in a statement.
“These types of increases in the MDRs are to be expected and may include past cases that were not previously reported to the FDA,” Dr. Ashar said, addressing the high number of new reports. “The increased number of MDRs contributes to our evolving understanding of BIA-ALCL and represents a more thorough and comprehensive analysis.”
BIA-ALCL is a type of non-Hodgkin lymphoma and a known risk from breast implants that was first communicated by the FDA in 2011. Regular updates have been provided with respect to related medical device reports, cases, deaths, and known risks.
“We hope that this information prompts providers and patients to have important, informed conversations about breast implants and the risk of BIA-ALCL. At the same time, we remain committed to working in partnership with all stakeholders to continue to study, understand, and provide updates about this important public health issue,” Dr. Ashar said.
To that end, the center also issued a Letter to Health Care Providers to “encourage those who regularly treat patients, including primary care physicians and gynecologists, to learn about BIA-ALCL in patients with breast implants.”
Patients and providers are encouraged to file MDRs with the FDA via MedWatch, the FDA Safety Information and Adverse Event Reporting program, she said.
The Food and Drug Administration has identified 457 unique cases of breast implant–associated anaplastic large cell lymphoma (BIA-ALCL) and 9 related deaths since 2010, and received 246 new medical device reports (MDRs) regarding BIA-ALCL between September 2017 and September 2018, according to an update from the agency’s Center for Devices and Radiological Health.

That brings the total number of reports to 660; however, that number reflects duplicative cases, Binita Ashar, MD, a general surgeon and the director of the division of surgical devices at the center, said in a statement.
“These types of increases in the MDRs are to be expected and may include past cases that were not previously reported to the FDA,” Dr. Ashar said, addressing the high number of new reports. “The increased number of MDRs contributes to our evolving understanding of BIA-ALCL and represents a more thorough and comprehensive analysis.”
BIA-ALCL is a type of non-Hodgkin lymphoma and a known risk from breast implants that was first communicated by the FDA in 2011. Regular updates have been provided with respect to related medical device reports, cases, deaths, and known risks.
“We hope that this information prompts providers and patients to have important, informed conversations about breast implants and the risk of BIA-ALCL. At the same time, we remain committed to working in partnership with all stakeholders to continue to study, understand, and provide updates about this important public health issue,” Dr. Ashar said.
To that end, the center also issued a Letter to Health Care Providers to “encourage those who regularly treat patients, including primary care physicians and gynecologists, to learn about BIA-ALCL in patients with breast implants.”
Patients and providers are encouraged to file MDRs with the FDA via MedWatch, the FDA Safety Information and Adverse Event Reporting program, she said.
Impella RP shows higher mortality in postapproval study
The Food and Drug Administration issued a letter on Feb. 4, 2019, to health care providers regarding interim results from a postapproval study for Abiomed’s Impella RP System because these results appear to have a higher mortality rate than was seen in premarket clinical studies.
As a condition of its approval, the FDA mandated Abiomed to perform a postapproval study (PAS); this study reflects use in a broader population than the premarket studies, which adhered to stricter inclusion and exclusion criteria.
Earlier in January, Abiomed submitted data to the FDA suggesting that differences in preimplant characteristics between patients in the PAS and those in the premarket clinical studies may explain the difference in mortality. Specifically, 16 of the 23 patients enrolled in the PAS would not have met the enrollment criteria for the premarket clinical studies because they were in cardiogenic shock for longer than 48 hours, experienced an in-hospital cardiac arrest, were treated with an intra-aortic balloon pump, or suffered a preimplant hypoxic or ischemic neurologic event.
“Although the FDA is concerned about the high mortality rate from the interim PAS results,” they wrote in the letter, which is available on the FDA website, “we believe that, when the device is used for the currently approved indication in appropriately selected patients, the benefits of the Impella RP system continue to outweigh the risks.”
The Food and Drug Administration issued a letter on Feb. 4, 2019, to health care providers regarding interim results from a postapproval study for Abiomed’s Impella RP System because these results appear to have a higher mortality rate than was seen in premarket clinical studies.
As a condition of its approval, the FDA mandated Abiomed to perform a postapproval study (PAS); this study reflects use in a broader population than the premarket studies, which adhered to stricter inclusion and exclusion criteria.
Earlier in January, Abiomed submitted data to the FDA suggesting that differences in preimplant characteristics between patients in the PAS and those in the premarket clinical studies may explain the difference in mortality. Specifically, 16 of the 23 patients enrolled in the PAS would not have met the enrollment criteria for the premarket clinical studies because they were in cardiogenic shock for longer than 48 hours, experienced an in-hospital cardiac arrest, were treated with an intra-aortic balloon pump, or suffered a preimplant hypoxic or ischemic neurologic event.
“Although the FDA is concerned about the high mortality rate from the interim PAS results,” they wrote in the letter, which is available on the FDA website, “we believe that, when the device is used for the currently approved indication in appropriately selected patients, the benefits of the Impella RP system continue to outweigh the risks.”
The Food and Drug Administration issued a letter on Feb. 4, 2019, to health care providers regarding interim results from a postapproval study for Abiomed’s Impella RP System because these results appear to have a higher mortality rate than was seen in premarket clinical studies.
As a condition of its approval, the FDA mandated Abiomed to perform a postapproval study (PAS); this study reflects use in a broader population than the premarket studies, which adhered to stricter inclusion and exclusion criteria.
Earlier in January, Abiomed submitted data to the FDA suggesting that differences in preimplant characteristics between patients in the PAS and those in the premarket clinical studies may explain the difference in mortality. Specifically, 16 of the 23 patients enrolled in the PAS would not have met the enrollment criteria for the premarket clinical studies because they were in cardiogenic shock for longer than 48 hours, experienced an in-hospital cardiac arrest, were treated with an intra-aortic balloon pump, or suffered a preimplant hypoxic or ischemic neurologic event.
“Although the FDA is concerned about the high mortality rate from the interim PAS results,” they wrote in the letter, which is available on the FDA website, “we believe that, when the device is used for the currently approved indication in appropriately selected patients, the benefits of the Impella RP system continue to outweigh the risks.”
FDA approves caplacizumab for aTTP
The Food and Drug Administration has approved caplacizumab (Cablivi) in combination with plasma exchange and immunosuppressive therapy for the treatment of adults with acquired thrombotic thrombocytopenic purpura (aTTP).
Caplacizumab is an anti–von Willebrand factor nanobody designed to inhibit the interaction between von Willebrand factor and platelets. The injection previously received orphan drug designation from the FDA and was approved under priority review.
The FDA’s approval of caplacizumab was based on results from the phase 3 HERCULES trial (N Engl J Med 2019 Jan 24;380:335-46).
The trial (NCT02553317) included 145 adults with aTTP. They were randomized to receive caplacizumab (n = 72) or placebo (n = 73), in addition to plasma exchange and immunosuppression.
The study’s primary endpoint was the time to platelet count response (normalization), which was defined as a platelet count of at least 150 x 109/L with subsequent stop of daily plasma exchange within 5 days.
There was a significant reduction in time to platelet count response in the caplacizumab arm, compared with the placebo arm – 2.69 days and 2.88 days, respectively. The platelet normalization rate ratio was 1.55 (P less than .01).
A secondary endpoint was the combination of aTTP-related death, aTTP recurrence, and at least one major thromboembolic event during study treatment. The incidence of this combined endpoint was 12% in the caplacizumab arm and 49% in the placebo arm (P less than .001).
The most common treatment-emergent adverse events (occurring in at least 15% of patients in the caplacizumab and placebo arms, respectively) were epistaxis (32% and 3%), headache (23% and 8%), urticaria (17% and 7%), and hypokalemia (9% and 19%).
During the treatment period, there were no deaths in the caplacizumab arm and three deaths in the placebo arm. There was one death (from cerebral ischemia) in the caplacizumab arm during the follow-up period, but it was considered unrelated to caplacizumab.
For more details on caplacizumab, see the full prescribing information.
The Food and Drug Administration has approved caplacizumab (Cablivi) in combination with plasma exchange and immunosuppressive therapy for the treatment of adults with acquired thrombotic thrombocytopenic purpura (aTTP).
Caplacizumab is an anti–von Willebrand factor nanobody designed to inhibit the interaction between von Willebrand factor and platelets. The injection previously received orphan drug designation from the FDA and was approved under priority review.
The FDA’s approval of caplacizumab was based on results from the phase 3 HERCULES trial (N Engl J Med 2019 Jan 24;380:335-46).
The trial (NCT02553317) included 145 adults with aTTP. They were randomized to receive caplacizumab (n = 72) or placebo (n = 73), in addition to plasma exchange and immunosuppression.
The study’s primary endpoint was the time to platelet count response (normalization), which was defined as a platelet count of at least 150 x 109/L with subsequent stop of daily plasma exchange within 5 days.
There was a significant reduction in time to platelet count response in the caplacizumab arm, compared with the placebo arm – 2.69 days and 2.88 days, respectively. The platelet normalization rate ratio was 1.55 (P less than .01).
A secondary endpoint was the combination of aTTP-related death, aTTP recurrence, and at least one major thromboembolic event during study treatment. The incidence of this combined endpoint was 12% in the caplacizumab arm and 49% in the placebo arm (P less than .001).
The most common treatment-emergent adverse events (occurring in at least 15% of patients in the caplacizumab and placebo arms, respectively) were epistaxis (32% and 3%), headache (23% and 8%), urticaria (17% and 7%), and hypokalemia (9% and 19%).
During the treatment period, there were no deaths in the caplacizumab arm and three deaths in the placebo arm. There was one death (from cerebral ischemia) in the caplacizumab arm during the follow-up period, but it was considered unrelated to caplacizumab.
For more details on caplacizumab, see the full prescribing information.
The Food and Drug Administration has approved caplacizumab (Cablivi) in combination with plasma exchange and immunosuppressive therapy for the treatment of adults with acquired thrombotic thrombocytopenic purpura (aTTP).
Caplacizumab is an anti–von Willebrand factor nanobody designed to inhibit the interaction between von Willebrand factor and platelets. The injection previously received orphan drug designation from the FDA and was approved under priority review.
The FDA’s approval of caplacizumab was based on results from the phase 3 HERCULES trial (N Engl J Med 2019 Jan 24;380:335-46).
The trial (NCT02553317) included 145 adults with aTTP. They were randomized to receive caplacizumab (n = 72) or placebo (n = 73), in addition to plasma exchange and immunosuppression.
The study’s primary endpoint was the time to platelet count response (normalization), which was defined as a platelet count of at least 150 x 109/L with subsequent stop of daily plasma exchange within 5 days.
There was a significant reduction in time to platelet count response in the caplacizumab arm, compared with the placebo arm – 2.69 days and 2.88 days, respectively. The platelet normalization rate ratio was 1.55 (P less than .01).
A secondary endpoint was the combination of aTTP-related death, aTTP recurrence, and at least one major thromboembolic event during study treatment. The incidence of this combined endpoint was 12% in the caplacizumab arm and 49% in the placebo arm (P less than .001).
The most common treatment-emergent adverse events (occurring in at least 15% of patients in the caplacizumab and placebo arms, respectively) were epistaxis (32% and 3%), headache (23% and 8%), urticaria (17% and 7%), and hypokalemia (9% and 19%).
During the treatment period, there were no deaths in the caplacizumab arm and three deaths in the placebo arm. There was one death (from cerebral ischemia) in the caplacizumab arm during the follow-up period, but it was considered unrelated to caplacizumab.
For more details on caplacizumab, see the full prescribing information.
FDA approves generic Advair Diskus
The Food and Drug Administration has approved a generic version of the Advair Diskus, a complex device-drug combination containing fluticasone propionate and salmeterol inhalation powder.
The generic device will be available in three strengths: fluticasone propionate 100 mcg/ salmeterol 50 mcg, fluticasone propionate 250 mcg/ salmeterol 50 mcg and fluticasone propionate 500 mcg/ salmeterol 50 mcg, according to the FDA announcement. It will be marketed by Mylan as Wixela Inhub and will launch in late February, according to a statement from Mylan.
Advair Diskus is among the most commonly used treatments for asthma and for chronic obstructive pulmonary disease (COPD), so it’s hoped this approval will increase access to the therapy, FDA officials said in a statement.
This approval is part of the FDA’s “longstanding commitment to advance access to lower cost, high quality generic alternatives,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a statement. “People living with asthma and COPD know too well the critical importance of having access to the treatment they need to feel better. Today’s approval will bring more competition to the market which will ultimately benefit the patients who rely on this drug.”
Wixela Inhub is indicated for twice-daily treatment of asthma in patients aged 4 years and older who are not adequately controlled by long-term asthma control treatments or whose disease warrants treatment with a combination of inhaled corticosteroids and long-acting beta agonists. It also is indicated for maintenance of COPD and reduction of COPD exacerbations.
The Food and Drug Administration has approved a generic version of the Advair Diskus, a complex device-drug combination containing fluticasone propionate and salmeterol inhalation powder.
The generic device will be available in three strengths: fluticasone propionate 100 mcg/ salmeterol 50 mcg, fluticasone propionate 250 mcg/ salmeterol 50 mcg and fluticasone propionate 500 mcg/ salmeterol 50 mcg, according to the FDA announcement. It will be marketed by Mylan as Wixela Inhub and will launch in late February, according to a statement from Mylan.
Advair Diskus is among the most commonly used treatments for asthma and for chronic obstructive pulmonary disease (COPD), so it’s hoped this approval will increase access to the therapy, FDA officials said in a statement.
This approval is part of the FDA’s “longstanding commitment to advance access to lower cost, high quality generic alternatives,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a statement. “People living with asthma and COPD know too well the critical importance of having access to the treatment they need to feel better. Today’s approval will bring more competition to the market which will ultimately benefit the patients who rely on this drug.”
Wixela Inhub is indicated for twice-daily treatment of asthma in patients aged 4 years and older who are not adequately controlled by long-term asthma control treatments or whose disease warrants treatment with a combination of inhaled corticosteroids and long-acting beta agonists. It also is indicated for maintenance of COPD and reduction of COPD exacerbations.
The Food and Drug Administration has approved a generic version of the Advair Diskus, a complex device-drug combination containing fluticasone propionate and salmeterol inhalation powder.
The generic device will be available in three strengths: fluticasone propionate 100 mcg/ salmeterol 50 mcg, fluticasone propionate 250 mcg/ salmeterol 50 mcg and fluticasone propionate 500 mcg/ salmeterol 50 mcg, according to the FDA announcement. It will be marketed by Mylan as Wixela Inhub and will launch in late February, according to a statement from Mylan.
Advair Diskus is among the most commonly used treatments for asthma and for chronic obstructive pulmonary disease (COPD), so it’s hoped this approval will increase access to the therapy, FDA officials said in a statement.
This approval is part of the FDA’s “longstanding commitment to advance access to lower cost, high quality generic alternatives,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in a statement. “People living with asthma and COPD know too well the critical importance of having access to the treatment they need to feel better. Today’s approval will bring more competition to the market which will ultimately benefit the patients who rely on this drug.”
Wixela Inhub is indicated for twice-daily treatment of asthma in patients aged 4 years and older who are not adequately controlled by long-term asthma control treatments or whose disease warrants treatment with a combination of inhaled corticosteroids and long-acting beta agonists. It also is indicated for maintenance of COPD and reduction of COPD exacerbations.
Flu activity ticks up for second week in a row
Influenza activity increased for a second straight week after a 2-week drop and by one measure has topped the high reached in late December, according to the Centers for Disease Control and Prevention.
For the week ending Jan. 26, 2019, there were 16 states at level 10 on the CDC’s 1-10 scale of influenza-like illness (ILI) activity, compared with 12 states during the week ending Dec. 29. With another seven states at levels 8 and 9, that makes 23 in the high range for the week ending Jan. 26, again putting it above the 19 reported for Dec. 29, the CDC’s influenza division reported Feb. 1.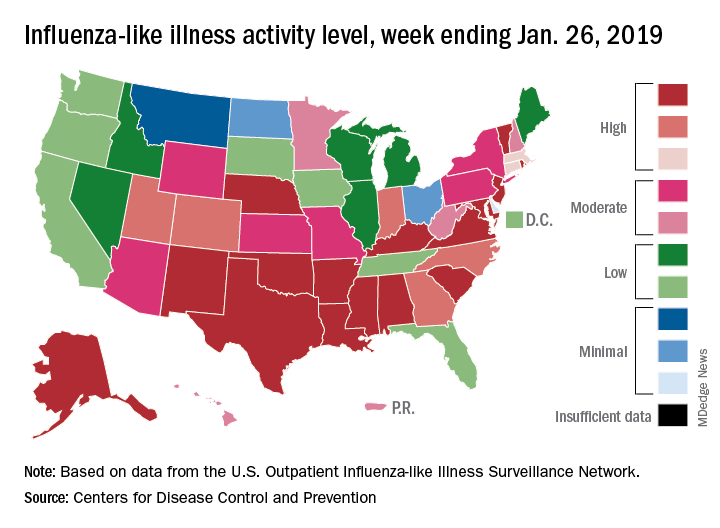
By another measure, however, that December peak in activity remains the seasonal high. The proportion of outpatient visits for ILI that week was 4.0%, compared with the 3.8% reported for Jan. 26. That’s up from 3.3% the week before and 3.1% the week before that, which in turn was the second week of a 2-week decline in activity in early January, CDC data show.
Two flu-related pediatric deaths were reported during the week ending Jan. 26, but both occurred the previous week. For the 2018-2019 flu season so far, a total of 24 pediatric flu deaths have been reported, the CDC said. At the same point in the 2017-2018 flu season, there had been 84 such deaths, according to the CDC’s Influenza-Associated Pediatric Mortality Surveillance System.
There were 143 overall flu-related deaths during the week of Jan. 19, which is the most recent week available. That is down from 189 the week before, but the Jan. 19 reporting is only 75% complete, data from the National Center for Health Statistics show.
Influenza activity increased for a second straight week after a 2-week drop and by one measure has topped the high reached in late December, according to the Centers for Disease Control and Prevention.
For the week ending Jan. 26, 2019, there were 16 states at level 10 on the CDC’s 1-10 scale of influenza-like illness (ILI) activity, compared with 12 states during the week ending Dec. 29. With another seven states at levels 8 and 9, that makes 23 in the high range for the week ending Jan. 26, again putting it above the 19 reported for Dec. 29, the CDC’s influenza division reported Feb. 1.
By another measure, however, that December peak in activity remains the seasonal high. The proportion of outpatient visits for ILI that week was 4.0%, compared with the 3.8% reported for Jan. 26. That’s up from 3.3% the week before and 3.1% the week before that, which in turn was the second week of a 2-week decline in activity in early January, CDC data show.
Two flu-related pediatric deaths were reported during the week ending Jan. 26, but both occurred the previous week. For the 2018-2019 flu season so far, a total of 24 pediatric flu deaths have been reported, the CDC said. At the same point in the 2017-2018 flu season, there had been 84 such deaths, according to the CDC’s Influenza-Associated Pediatric Mortality Surveillance System.
There were 143 overall flu-related deaths during the week of Jan. 19, which is the most recent week available. That is down from 189 the week before, but the Jan. 19 reporting is only 75% complete, data from the National Center for Health Statistics show.
Influenza activity increased for a second straight week after a 2-week drop and by one measure has topped the high reached in late December, according to the Centers for Disease Control and Prevention.
For the week ending Jan. 26, 2019, there were 16 states at level 10 on the CDC’s 1-10 scale of influenza-like illness (ILI) activity, compared with 12 states during the week ending Dec. 29. With another seven states at levels 8 and 9, that makes 23 in the high range for the week ending Jan. 26, again putting it above the 19 reported for Dec. 29, the CDC’s influenza division reported Feb. 1.
By another measure, however, that December peak in activity remains the seasonal high. The proportion of outpatient visits for ILI that week was 4.0%, compared with the 3.8% reported for Jan. 26. That’s up from 3.3% the week before and 3.1% the week before that, which in turn was the second week of a 2-week decline in activity in early January, CDC data show.
Two flu-related pediatric deaths were reported during the week ending Jan. 26, but both occurred the previous week. For the 2018-2019 flu season so far, a total of 24 pediatric flu deaths have been reported, the CDC said. At the same point in the 2017-2018 flu season, there had been 84 such deaths, according to the CDC’s Influenza-Associated Pediatric Mortality Surveillance System.
There were 143 overall flu-related deaths during the week of Jan. 19, which is the most recent week available. That is down from 189 the week before, but the Jan. 19 reporting is only 75% complete, data from the National Center for Health Statistics show.
FDA grants BI-1206 orphan designation for MCL
The Food and Drug Administration has granted orphan designation to BI-1206 for the treatment of mantle cell lymphoma (MCL).
BI-1206 is a monoclonal antibody being developed by BioInvent International.
The company says BI-1206 works by inhibiting FcgRIIB (CD32B), which is associated with poor prognosis in MCL and other non-Hodgkin lymphomas. By inhibiting FcgRIIB, BI-1206 is expected to enhance the activity of rituximab or other anti-CD20 monoclonal antibodies.
BioInvent is conducting a phase 1/2a study (NCT03571568) of BI-1206 in combination with rituximab in patients with indolent, relapsed/refractory B-cell non-Hodgkin lymphomas, including MCL. The first patient began receiving treatment with BI-1206 in September 2018.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases or disorders that affect fewer than 200,000 people in the United States. Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The Food and Drug Administration has granted orphan designation to BI-1206 for the treatment of mantle cell lymphoma (MCL).
BI-1206 is a monoclonal antibody being developed by BioInvent International.
The company says BI-1206 works by inhibiting FcgRIIB (CD32B), which is associated with poor prognosis in MCL and other non-Hodgkin lymphomas. By inhibiting FcgRIIB, BI-1206 is expected to enhance the activity of rituximab or other anti-CD20 monoclonal antibodies.
BioInvent is conducting a phase 1/2a study (NCT03571568) of BI-1206 in combination with rituximab in patients with indolent, relapsed/refractory B-cell non-Hodgkin lymphomas, including MCL. The first patient began receiving treatment with BI-1206 in September 2018.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases or disorders that affect fewer than 200,000 people in the United States. Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The Food and Drug Administration has granted orphan designation to BI-1206 for the treatment of mantle cell lymphoma (MCL).
BI-1206 is a monoclonal antibody being developed by BioInvent International.
The company says BI-1206 works by inhibiting FcgRIIB (CD32B), which is associated with poor prognosis in MCL and other non-Hodgkin lymphomas. By inhibiting FcgRIIB, BI-1206 is expected to enhance the activity of rituximab or other anti-CD20 monoclonal antibodies.
BioInvent is conducting a phase 1/2a study (NCT03571568) of BI-1206 in combination with rituximab in patients with indolent, relapsed/refractory B-cell non-Hodgkin lymphomas, including MCL. The first patient began receiving treatment with BI-1206 in September 2018.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases or disorders that affect fewer than 200,000 people in the United States. Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.