User login
Understanding Principles of High Reliability Organizations Through the Eyes of VIONE, A Clinical Program to Improve Patient Safety by Deprescribing Potentially Inappropriate Medications and Reducing Polypharmacy
High reliability organizations (HROs) incorporate continuous process improvement through leadership commitment to create a safety culture that works toward creating a zero-harm environment.1 The Veterans Health Administration (VHA) has set transformational goals for becoming an HRO. In this article, we describe VIONE, an expanding medication deprescribing clinical program, which exemplifies the translation of HRO principles into health care system models. Both VIONE and HRO are globally relevant.
Reducing medication errors and related adverse drug events are important for achieving zero harm. Preventable medical errors rank behind heart disease and cancer as the third leading cause of death in the US.2 The simultaneous use of multiple medications can lead to dangerous drug interactions, adverse outcomes, and challenges with adherence. When a person is taking multiple medicines, known as polypharmacy, it is more likely that some are potentially inappropriate medications (PIM). Current literature highlights the prevalence and dangers of polypharmacy, which ranks among the top 10 common causes of death in the US, as well as suggestions to address preventable adverse outcomes from polypharmacy and PIM.3-5
Deprescribing of PIM frequently results in better disease management with improved health outcomes and quality of life.4 Many health care settings lack standardized approaches or set expectations to proactively deprescribe PIM. There has been insufficient emphasis on how to make decisions for deprescribing medications when therapeutic benefits are not clear and/or when the adverse effects may outweigh the therapeutic benefits.5
It is imperative to provide practice guidance for deprescribing nonessential medications along with systems-based infrastructure to enable integrated and effective assessments during opportune moments in the health care continuum. Multimodal approaches that include education, risk stratification, population health management interventions, research and resource allocation can help transform organizational culture in health care facilities toward HRO models of care, aiming at zero harm to patients.
The practical lessons learned from VIONE implementation science experiences on various scales and under diverse circumstances, cumulative wisdom from hindsight, foresight and critical insights gathered during nationwide spread of VIONE over the past 3 years continues to propel us toward the desirable direction and core concepts of an HRO.
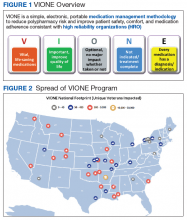
The VIONE program facilitates practical, real-time interventions that could be tailored to various health care settings, organizational needs, and available resources. VIONE implements an electronic Computerized Patient Record System (CPRS) tool to enable planned cessation of nonessential medications that are potentially harmful, inappropriate, not indicated, or not necessary. The VIONE tool supports systematic, individualized assessment and adjustment through 5 filters (Figure 1). It prompts providers to assign 1 of these filters intuitively and objectively. VIONE combines clinical evidence for best practices, an interprofessional team approach, patient engagement, adapted use of existing medical records systems, and HRO principles for effective implementation.
As a tool to support safer prescribing practices, VIONE aligns closely with HRO principles (Table 1) and core pillars (Table 2).6-8 A zero-harm safety culture necessitates that medications be used for correct reasons, over a correct duration of time, and following a correct schedule while monitoring for adverse outcomes. However, reality generally falls significantly short of this for a myriad of reasons, such as compromised health literacy, functional limitations, affordability, communication gaps, patients seen by multiple providers, and an accumulation of prescriptions due to comorbidities, symptom progression, and management of adverse effects. Through a sharpened focus on both precision medicine and competent prescription management, VIONE is a viable opportunity for investing in the zero-harm philosophy that is integral to an HRO.
Design and Implementation
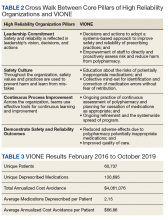
Initially launched in 2016 in a 15-bed inpatient, subacute rehabilitation unit within a VHA tertiary care facility, VIONE has been sustained and gradually expanded to 38 other VHA facility programs (Figure 2). Recognizing the potential value if adopted into widespread use, VIONE was a Gold Status winner in the VHA Under Secretary for Health Shark Tank-style competition in 2017 and was selected by the VHA Diffusion of Excellence as an innovation worthy of scale and spread through national dissemination.9 A toolkit for VIONE implementation, patient and provider brochures, VIONE vignette, and National Dialog template also have been created.10
Implementing VIONE in a new facility requires an actively engaged core team committed to patient safety and reduction of polypharmacy and PIM, interest and availability to lead project implementation strategies, along with meaningful local organizational support. The current structure for VIONE spread is as follows:
- Interested VHA participants review information and contact [email protected].
- The VIONE team orients implementing champions, mainly pharmacists, physicians, nurse practitioners, and physician assistants at a facility program level, offering guidance and available resources.
- Clinical Application Coordinators at Central Arkansas VA Healthcare System and participating facilities collaborate to add deprescribing menu options in CPRS and install the VIONE Polypharmacy Reminder Dialog template.
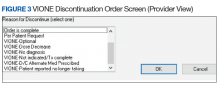
- Through close and ongoing collaborations, medical providers and clinical pharmacists proceed with deprescribing, aiming at planned cessation of nonessential and PIM, using the mnemonic prompt of VIONE. Vital and Important medications are continued and consolidated while a methodical plan is developed to deprescribe any medications that could lead to more harm than benefit and qualify based on the filters of Optional, Not indicated, and Every medicine has a diagnosis/reason. They select the proper discontinuation reasons in the CPRS medication menu (Figure 3) and document the rationale in the progress notes. It is highly encouraged that the collaborating pharmacists and health care providers add each other as cosigners and communicate effectively. Clinical pharmacy specialists also use the VIONE Polypharmacy Reminder Dialog Template (RDT) to document complete medication reviews with veterans to include deprescribing rationale and document shared decision making.
- A VIONE national dashboard captures deprescribing data in real time and automates reporting with daily updates that are readily accessible to all implementing facilities. Minimum data captured include the number of unique veterans impacted, number of medications deprescribed, cumulative cost avoidance to date, and number of prescriptions deprescribed per veteran. The dashboard facilitates real-time use of individual patient data and has also been designed to capture data from VHA administrative data portals and Corporate Data Warehouse.
Results
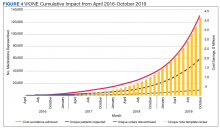
As of October 31, 2019, the assessment of polypharmacy using the VIONE tool across VHA sites has benefited > 60,000 unique veterans, of whom 49.2% were in urban areas, 47.7% in rural areas, and 3.1% in highly rural areas. Elderly male veterans comprised a clear majority. More than 128,000 medications have been deprescribed. The top classes of medications deprescribed are antihypertensives, over-the-counter medications, and antidiabetic medications. An annualized cost avoidance of > $4.0 million has been achieved. Cost avoidance is the cost of medications that otherwise would have continued to be filled and paid for by the VHA if they had not been deprescribed, projected for a maximum of 365 days. The calculation methodology can be summarized as follows:
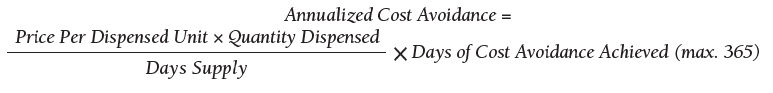
The calculations reported in Table 3 and Figure 4 are conservative and include only chronic outpatient prescriptions and do not account for medications deprescribed in inpatient units, nursing home, community living centers, or domiciliary populations. Data tracked separately from inpatient and community living center patient populations indicated an additional 25,536 deprescribed medications, across 28 VA facilities, impacting 7,076 veterans with an average 2.15 medications deprescribed per veteran. The additional achieved cost avoidance was $370,272 (based on $14.50 average cost per prescription). Medications restarted within 30 days of deprescribing are not included in these calculations.
The cost avoidance calculation further excludes the effects of VIONE implementation on many other types of interventions. These interventions include, but are not limited to, changing from aggressive care to end of life, comfort care when strongly indicated; reduced emergency department visits or invasive diagnostic and therapeutic approaches, when not indicated; medical supplies, antimicrobial preparations; labor costs related to packaging, mailing, and administering prescriptions; reduced/prevented clinical waste; reduced decompensation of systemic illnesses and subsequent health care needs precipitated by iatrogenic disturbances and prolonged convalescence; and overall changes to prescribing practices through purposeful and targeted interactions with colleagues across various disciplines and various hierarchical levels.
Discussion
The VIONE clinical program exemplifies the translation of HRO principles into health care system practices. VIONE offers a systematic approach to improve medication management with an emphasis on deprescribing nonessential medications across various health care settings, facilitating VHA efforts toward zero harm. It demonstrates close alignment with the key building blocks of an HRO. Effective VIONE incorporation into an organizational culture reflects leadership commitment to safety and reliability in their vision and actions. By empowering staff to proactively reduce inappropriate medications and thereby prevent patient harm, VIONE contributes to enhancing an enterprise-wide culture of safety, with fewer errors and greater reliability. As a standardized decision support tool for the ongoing practice of assessment and planned cessation of potentially inappropriate medications, VIONE illustrates how continuous process improvement can be a part of staff-engaged, veteran-centered, highly reliable care. The standardization of the VIONE tool promotes achievement and sustainment of desired HRO principles and practices within health care delivery systems.
Conclusions
The VIONE program was launched not as a cost savings or research program but as a practical, real-time bedside or ambulatory care intervention to improve patient safety. Its value is reflected in the overwhelming response from scholarly and well-engaged colleagues expressing serious interests in expanding collaborations and tailoring efforts to add more depth and breadth to VIONE related efforts.
Acknowledgments
The authors express their gratitude to Central Arkansas VA Healthcare System leadership, Clinical Applications Coordinators, and colleagues for their unconditional support, to the Diffusion of Excellence programs at US Department of Veterans Affairs Central Office for their endorsement, and to the many VHA participants who renew our optimism and energy as we continue this exciting journey. We also thank Bridget B. Kelly for her assistance in writing and editing of the manuscript.
1. Chassin MR, Jerod ML. High-reliability health care: getting there from here. The Joint Commission. Milbank Q. 2013;91(3):459-490.
2. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139.
3. Quinn KJ, Shah NH. A dataset quantifying polypharmacy in the United States. Sci Data. 2017;4:170167.
4. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
5. Steinman MA. Polypharmacy—time to get beyond numbers. JAMA Intern Med. 2016;176(4):482-483.
6. US Department of Veterans Affairs. High reliability. https://dvagov.sharepoint.com/sites/OHT-PMO/high-reliability/Pages/default.aspx. [Nonpublic source, not verified.]
7. Gordon S, Mendenhall P, O’Connor BB. Beyond the Checklist: What Else Health Care Can Learn from Aviation Teamwork and Safety. Ithaca, NY: Cornell University Press; 2013.
8. Institute of Medicine (US) Committee on Quality of Health Care in America; Kohn LT, Corrigan JM, Donaldson MS, eds. To Err Is Human: Building a Safer Health System. Washington, DC: The National Academies Press; 2000.
9. US Department of Veterans Affairs. Diffusion of Excellence. https://www.va.gov/HEALTHCAREEXCELLENCE/diffusion-of-excellence/. Updated August 10, 2018. Accessed June 26, 2019.
10. US Department of Veterans Affairs. VIONE program toolkit. https://www.vapulse.net/docs/DOC-259375. [Nonpublic source, not verified.]
High reliability organizations (HROs) incorporate continuous process improvement through leadership commitment to create a safety culture that works toward creating a zero-harm environment.1 The Veterans Health Administration (VHA) has set transformational goals for becoming an HRO. In this article, we describe VIONE, an expanding medication deprescribing clinical program, which exemplifies the translation of HRO principles into health care system models. Both VIONE and HRO are globally relevant.
Reducing medication errors and related adverse drug events are important for achieving zero harm. Preventable medical errors rank behind heart disease and cancer as the third leading cause of death in the US.2 The simultaneous use of multiple medications can lead to dangerous drug interactions, adverse outcomes, and challenges with adherence. When a person is taking multiple medicines, known as polypharmacy, it is more likely that some are potentially inappropriate medications (PIM). Current literature highlights the prevalence and dangers of polypharmacy, which ranks among the top 10 common causes of death in the US, as well as suggestions to address preventable adverse outcomes from polypharmacy and PIM.3-5
Deprescribing of PIM frequently results in better disease management with improved health outcomes and quality of life.4 Many health care settings lack standardized approaches or set expectations to proactively deprescribe PIM. There has been insufficient emphasis on how to make decisions for deprescribing medications when therapeutic benefits are not clear and/or when the adverse effects may outweigh the therapeutic benefits.5
It is imperative to provide practice guidance for deprescribing nonessential medications along with systems-based infrastructure to enable integrated and effective assessments during opportune moments in the health care continuum. Multimodal approaches that include education, risk stratification, population health management interventions, research and resource allocation can help transform organizational culture in health care facilities toward HRO models of care, aiming at zero harm to patients.
The practical lessons learned from VIONE implementation science experiences on various scales and under diverse circumstances, cumulative wisdom from hindsight, foresight and critical insights gathered during nationwide spread of VIONE over the past 3 years continues to propel us toward the desirable direction and core concepts of an HRO.
The VIONE program facilitates practical, real-time interventions that could be tailored to various health care settings, organizational needs, and available resources. VIONE implements an electronic Computerized Patient Record System (CPRS) tool to enable planned cessation of nonessential medications that are potentially harmful, inappropriate, not indicated, or not necessary. The VIONE tool supports systematic, individualized assessment and adjustment through 5 filters (Figure 1). It prompts providers to assign 1 of these filters intuitively and objectively. VIONE combines clinical evidence for best practices, an interprofessional team approach, patient engagement, adapted use of existing medical records systems, and HRO principles for effective implementation.
As a tool to support safer prescribing practices, VIONE aligns closely with HRO principles (Table 1) and core pillars (Table 2).6-8 A zero-harm safety culture necessitates that medications be used for correct reasons, over a correct duration of time, and following a correct schedule while monitoring for adverse outcomes. However, reality generally falls significantly short of this for a myriad of reasons, such as compromised health literacy, functional limitations, affordability, communication gaps, patients seen by multiple providers, and an accumulation of prescriptions due to comorbidities, symptom progression, and management of adverse effects. Through a sharpened focus on both precision medicine and competent prescription management, VIONE is a viable opportunity for investing in the zero-harm philosophy that is integral to an HRO.
Design and Implementation
Initially launched in 2016 in a 15-bed inpatient, subacute rehabilitation unit within a VHA tertiary care facility, VIONE has been sustained and gradually expanded to 38 other VHA facility programs (Figure 2). Recognizing the potential value if adopted into widespread use, VIONE was a Gold Status winner in the VHA Under Secretary for Health Shark Tank-style competition in 2017 and was selected by the VHA Diffusion of Excellence as an innovation worthy of scale and spread through national dissemination.9 A toolkit for VIONE implementation, patient and provider brochures, VIONE vignette, and National Dialog template also have been created.10
Implementing VIONE in a new facility requires an actively engaged core team committed to patient safety and reduction of polypharmacy and PIM, interest and availability to lead project implementation strategies, along with meaningful local organizational support. The current structure for VIONE spread is as follows:
- Interested VHA participants review information and contact [email protected].
- The VIONE team orients implementing champions, mainly pharmacists, physicians, nurse practitioners, and physician assistants at a facility program level, offering guidance and available resources.
- Clinical Application Coordinators at Central Arkansas VA Healthcare System and participating facilities collaborate to add deprescribing menu options in CPRS and install the VIONE Polypharmacy Reminder Dialog template.
- Through close and ongoing collaborations, medical providers and clinical pharmacists proceed with deprescribing, aiming at planned cessation of nonessential and PIM, using the mnemonic prompt of VIONE. Vital and Important medications are continued and consolidated while a methodical plan is developed to deprescribe any medications that could lead to more harm than benefit and qualify based on the filters of Optional, Not indicated, and Every medicine has a diagnosis/reason. They select the proper discontinuation reasons in the CPRS medication menu (Figure 3) and document the rationale in the progress notes. It is highly encouraged that the collaborating pharmacists and health care providers add each other as cosigners and communicate effectively. Clinical pharmacy specialists also use the VIONE Polypharmacy Reminder Dialog Template (RDT) to document complete medication reviews with veterans to include deprescribing rationale and document shared decision making.
- A VIONE national dashboard captures deprescribing data in real time and automates reporting with daily updates that are readily accessible to all implementing facilities. Minimum data captured include the number of unique veterans impacted, number of medications deprescribed, cumulative cost avoidance to date, and number of prescriptions deprescribed per veteran. The dashboard facilitates real-time use of individual patient data and has also been designed to capture data from VHA administrative data portals and Corporate Data Warehouse.
Results
As of October 31, 2019, the assessment of polypharmacy using the VIONE tool across VHA sites has benefited > 60,000 unique veterans, of whom 49.2% were in urban areas, 47.7% in rural areas, and 3.1% in highly rural areas. Elderly male veterans comprised a clear majority. More than 128,000 medications have been deprescribed. The top classes of medications deprescribed are antihypertensives, over-the-counter medications, and antidiabetic medications. An annualized cost avoidance of > $4.0 million has been achieved. Cost avoidance is the cost of medications that otherwise would have continued to be filled and paid for by the VHA if they had not been deprescribed, projected for a maximum of 365 days. The calculation methodology can be summarized as follows:
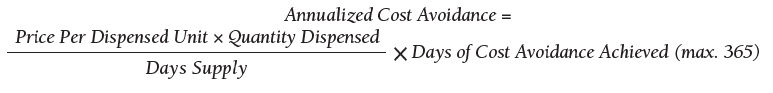
The calculations reported in Table 3 and Figure 4 are conservative and include only chronic outpatient prescriptions and do not account for medications deprescribed in inpatient units, nursing home, community living centers, or domiciliary populations. Data tracked separately from inpatient and community living center patient populations indicated an additional 25,536 deprescribed medications, across 28 VA facilities, impacting 7,076 veterans with an average 2.15 medications deprescribed per veteran. The additional achieved cost avoidance was $370,272 (based on $14.50 average cost per prescription). Medications restarted within 30 days of deprescribing are not included in these calculations.
The cost avoidance calculation further excludes the effects of VIONE implementation on many other types of interventions. These interventions include, but are not limited to, changing from aggressive care to end of life, comfort care when strongly indicated; reduced emergency department visits or invasive diagnostic and therapeutic approaches, when not indicated; medical supplies, antimicrobial preparations; labor costs related to packaging, mailing, and administering prescriptions; reduced/prevented clinical waste; reduced decompensation of systemic illnesses and subsequent health care needs precipitated by iatrogenic disturbances and prolonged convalescence; and overall changes to prescribing practices through purposeful and targeted interactions with colleagues across various disciplines and various hierarchical levels.
Discussion
The VIONE clinical program exemplifies the translation of HRO principles into health care system practices. VIONE offers a systematic approach to improve medication management with an emphasis on deprescribing nonessential medications across various health care settings, facilitating VHA efforts toward zero harm. It demonstrates close alignment with the key building blocks of an HRO. Effective VIONE incorporation into an organizational culture reflects leadership commitment to safety and reliability in their vision and actions. By empowering staff to proactively reduce inappropriate medications and thereby prevent patient harm, VIONE contributes to enhancing an enterprise-wide culture of safety, with fewer errors and greater reliability. As a standardized decision support tool for the ongoing practice of assessment and planned cessation of potentially inappropriate medications, VIONE illustrates how continuous process improvement can be a part of staff-engaged, veteran-centered, highly reliable care. The standardization of the VIONE tool promotes achievement and sustainment of desired HRO principles and practices within health care delivery systems.
Conclusions
The VIONE program was launched not as a cost savings or research program but as a practical, real-time bedside or ambulatory care intervention to improve patient safety. Its value is reflected in the overwhelming response from scholarly and well-engaged colleagues expressing serious interests in expanding collaborations and tailoring efforts to add more depth and breadth to VIONE related efforts.
Acknowledgments
The authors express their gratitude to Central Arkansas VA Healthcare System leadership, Clinical Applications Coordinators, and colleagues for their unconditional support, to the Diffusion of Excellence programs at US Department of Veterans Affairs Central Office for their endorsement, and to the many VHA participants who renew our optimism and energy as we continue this exciting journey. We also thank Bridget B. Kelly for her assistance in writing and editing of the manuscript.
High reliability organizations (HROs) incorporate continuous process improvement through leadership commitment to create a safety culture that works toward creating a zero-harm environment.1 The Veterans Health Administration (VHA) has set transformational goals for becoming an HRO. In this article, we describe VIONE, an expanding medication deprescribing clinical program, which exemplifies the translation of HRO principles into health care system models. Both VIONE and HRO are globally relevant.
Reducing medication errors and related adverse drug events are important for achieving zero harm. Preventable medical errors rank behind heart disease and cancer as the third leading cause of death in the US.2 The simultaneous use of multiple medications can lead to dangerous drug interactions, adverse outcomes, and challenges with adherence. When a person is taking multiple medicines, known as polypharmacy, it is more likely that some are potentially inappropriate medications (PIM). Current literature highlights the prevalence and dangers of polypharmacy, which ranks among the top 10 common causes of death in the US, as well as suggestions to address preventable adverse outcomes from polypharmacy and PIM.3-5
Deprescribing of PIM frequently results in better disease management with improved health outcomes and quality of life.4 Many health care settings lack standardized approaches or set expectations to proactively deprescribe PIM. There has been insufficient emphasis on how to make decisions for deprescribing medications when therapeutic benefits are not clear and/or when the adverse effects may outweigh the therapeutic benefits.5
It is imperative to provide practice guidance for deprescribing nonessential medications along with systems-based infrastructure to enable integrated and effective assessments during opportune moments in the health care continuum. Multimodal approaches that include education, risk stratification, population health management interventions, research and resource allocation can help transform organizational culture in health care facilities toward HRO models of care, aiming at zero harm to patients.
The practical lessons learned from VIONE implementation science experiences on various scales and under diverse circumstances, cumulative wisdom from hindsight, foresight and critical insights gathered during nationwide spread of VIONE over the past 3 years continues to propel us toward the desirable direction and core concepts of an HRO.
The VIONE program facilitates practical, real-time interventions that could be tailored to various health care settings, organizational needs, and available resources. VIONE implements an electronic Computerized Patient Record System (CPRS) tool to enable planned cessation of nonessential medications that are potentially harmful, inappropriate, not indicated, or not necessary. The VIONE tool supports systematic, individualized assessment and adjustment through 5 filters (Figure 1). It prompts providers to assign 1 of these filters intuitively and objectively. VIONE combines clinical evidence for best practices, an interprofessional team approach, patient engagement, adapted use of existing medical records systems, and HRO principles for effective implementation.
As a tool to support safer prescribing practices, VIONE aligns closely with HRO principles (Table 1) and core pillars (Table 2).6-8 A zero-harm safety culture necessitates that medications be used for correct reasons, over a correct duration of time, and following a correct schedule while monitoring for adverse outcomes. However, reality generally falls significantly short of this for a myriad of reasons, such as compromised health literacy, functional limitations, affordability, communication gaps, patients seen by multiple providers, and an accumulation of prescriptions due to comorbidities, symptom progression, and management of adverse effects. Through a sharpened focus on both precision medicine and competent prescription management, VIONE is a viable opportunity for investing in the zero-harm philosophy that is integral to an HRO.
Design and Implementation
Initially launched in 2016 in a 15-bed inpatient, subacute rehabilitation unit within a VHA tertiary care facility, VIONE has been sustained and gradually expanded to 38 other VHA facility programs (Figure 2). Recognizing the potential value if adopted into widespread use, VIONE was a Gold Status winner in the VHA Under Secretary for Health Shark Tank-style competition in 2017 and was selected by the VHA Diffusion of Excellence as an innovation worthy of scale and spread through national dissemination.9 A toolkit for VIONE implementation, patient and provider brochures, VIONE vignette, and National Dialog template also have been created.10
Implementing VIONE in a new facility requires an actively engaged core team committed to patient safety and reduction of polypharmacy and PIM, interest and availability to lead project implementation strategies, along with meaningful local organizational support. The current structure for VIONE spread is as follows:
- Interested VHA participants review information and contact [email protected].
- The VIONE team orients implementing champions, mainly pharmacists, physicians, nurse practitioners, and physician assistants at a facility program level, offering guidance and available resources.
- Clinical Application Coordinators at Central Arkansas VA Healthcare System and participating facilities collaborate to add deprescribing menu options in CPRS and install the VIONE Polypharmacy Reminder Dialog template.
- Through close and ongoing collaborations, medical providers and clinical pharmacists proceed with deprescribing, aiming at planned cessation of nonessential and PIM, using the mnemonic prompt of VIONE. Vital and Important medications are continued and consolidated while a methodical plan is developed to deprescribe any medications that could lead to more harm than benefit and qualify based on the filters of Optional, Not indicated, and Every medicine has a diagnosis/reason. They select the proper discontinuation reasons in the CPRS medication menu (Figure 3) and document the rationale in the progress notes. It is highly encouraged that the collaborating pharmacists and health care providers add each other as cosigners and communicate effectively. Clinical pharmacy specialists also use the VIONE Polypharmacy Reminder Dialog Template (RDT) to document complete medication reviews with veterans to include deprescribing rationale and document shared decision making.
- A VIONE national dashboard captures deprescribing data in real time and automates reporting with daily updates that are readily accessible to all implementing facilities. Minimum data captured include the number of unique veterans impacted, number of medications deprescribed, cumulative cost avoidance to date, and number of prescriptions deprescribed per veteran. The dashboard facilitates real-time use of individual patient data and has also been designed to capture data from VHA administrative data portals and Corporate Data Warehouse.
Results
As of October 31, 2019, the assessment of polypharmacy using the VIONE tool across VHA sites has benefited > 60,000 unique veterans, of whom 49.2% were in urban areas, 47.7% in rural areas, and 3.1% in highly rural areas. Elderly male veterans comprised a clear majority. More than 128,000 medications have been deprescribed. The top classes of medications deprescribed are antihypertensives, over-the-counter medications, and antidiabetic medications. An annualized cost avoidance of > $4.0 million has been achieved. Cost avoidance is the cost of medications that otherwise would have continued to be filled and paid for by the VHA if they had not been deprescribed, projected for a maximum of 365 days. The calculation methodology can be summarized as follows:
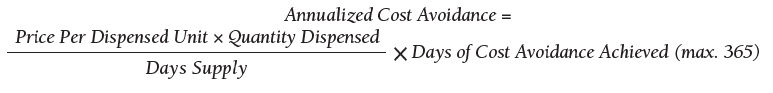
The calculations reported in Table 3 and Figure 4 are conservative and include only chronic outpatient prescriptions and do not account for medications deprescribed in inpatient units, nursing home, community living centers, or domiciliary populations. Data tracked separately from inpatient and community living center patient populations indicated an additional 25,536 deprescribed medications, across 28 VA facilities, impacting 7,076 veterans with an average 2.15 medications deprescribed per veteran. The additional achieved cost avoidance was $370,272 (based on $14.50 average cost per prescription). Medications restarted within 30 days of deprescribing are not included in these calculations.
The cost avoidance calculation further excludes the effects of VIONE implementation on many other types of interventions. These interventions include, but are not limited to, changing from aggressive care to end of life, comfort care when strongly indicated; reduced emergency department visits or invasive diagnostic and therapeutic approaches, when not indicated; medical supplies, antimicrobial preparations; labor costs related to packaging, mailing, and administering prescriptions; reduced/prevented clinical waste; reduced decompensation of systemic illnesses and subsequent health care needs precipitated by iatrogenic disturbances and prolonged convalescence; and overall changes to prescribing practices through purposeful and targeted interactions with colleagues across various disciplines and various hierarchical levels.
Discussion
The VIONE clinical program exemplifies the translation of HRO principles into health care system practices. VIONE offers a systematic approach to improve medication management with an emphasis on deprescribing nonessential medications across various health care settings, facilitating VHA efforts toward zero harm. It demonstrates close alignment with the key building blocks of an HRO. Effective VIONE incorporation into an organizational culture reflects leadership commitment to safety and reliability in their vision and actions. By empowering staff to proactively reduce inappropriate medications and thereby prevent patient harm, VIONE contributes to enhancing an enterprise-wide culture of safety, with fewer errors and greater reliability. As a standardized decision support tool for the ongoing practice of assessment and planned cessation of potentially inappropriate medications, VIONE illustrates how continuous process improvement can be a part of staff-engaged, veteran-centered, highly reliable care. The standardization of the VIONE tool promotes achievement and sustainment of desired HRO principles and practices within health care delivery systems.
Conclusions
The VIONE program was launched not as a cost savings or research program but as a practical, real-time bedside or ambulatory care intervention to improve patient safety. Its value is reflected in the overwhelming response from scholarly and well-engaged colleagues expressing serious interests in expanding collaborations and tailoring efforts to add more depth and breadth to VIONE related efforts.
Acknowledgments
The authors express their gratitude to Central Arkansas VA Healthcare System leadership, Clinical Applications Coordinators, and colleagues for their unconditional support, to the Diffusion of Excellence programs at US Department of Veterans Affairs Central Office for their endorsement, and to the many VHA participants who renew our optimism and energy as we continue this exciting journey. We also thank Bridget B. Kelly for her assistance in writing and editing of the manuscript.
1. Chassin MR, Jerod ML. High-reliability health care: getting there from here. The Joint Commission. Milbank Q. 2013;91(3):459-490.
2. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139.
3. Quinn KJ, Shah NH. A dataset quantifying polypharmacy in the United States. Sci Data. 2017;4:170167.
4. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
5. Steinman MA. Polypharmacy—time to get beyond numbers. JAMA Intern Med. 2016;176(4):482-483.
6. US Department of Veterans Affairs. High reliability. https://dvagov.sharepoint.com/sites/OHT-PMO/high-reliability/Pages/default.aspx. [Nonpublic source, not verified.]
7. Gordon S, Mendenhall P, O’Connor BB. Beyond the Checklist: What Else Health Care Can Learn from Aviation Teamwork and Safety. Ithaca, NY: Cornell University Press; 2013.
8. Institute of Medicine (US) Committee on Quality of Health Care in America; Kohn LT, Corrigan JM, Donaldson MS, eds. To Err Is Human: Building a Safer Health System. Washington, DC: The National Academies Press; 2000.
9. US Department of Veterans Affairs. Diffusion of Excellence. https://www.va.gov/HEALTHCAREEXCELLENCE/diffusion-of-excellence/. Updated August 10, 2018. Accessed June 26, 2019.
10. US Department of Veterans Affairs. VIONE program toolkit. https://www.vapulse.net/docs/DOC-259375. [Nonpublic source, not verified.]
1. Chassin MR, Jerod ML. High-reliability health care: getting there from here. The Joint Commission. Milbank Q. 2013;91(3):459-490.
2. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139.
3. Quinn KJ, Shah NH. A dataset quantifying polypharmacy in the United States. Sci Data. 2017;4:170167.
4. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
5. Steinman MA. Polypharmacy—time to get beyond numbers. JAMA Intern Med. 2016;176(4):482-483.
6. US Department of Veterans Affairs. High reliability. https://dvagov.sharepoint.com/sites/OHT-PMO/high-reliability/Pages/default.aspx. [Nonpublic source, not verified.]
7. Gordon S, Mendenhall P, O’Connor BB. Beyond the Checklist: What Else Health Care Can Learn from Aviation Teamwork and Safety. Ithaca, NY: Cornell University Press; 2013.
8. Institute of Medicine (US) Committee on Quality of Health Care in America; Kohn LT, Corrigan JM, Donaldson MS, eds. To Err Is Human: Building a Safer Health System. Washington, DC: The National Academies Press; 2000.
9. US Department of Veterans Affairs. Diffusion of Excellence. https://www.va.gov/HEALTHCAREEXCELLENCE/diffusion-of-excellence/. Updated August 10, 2018. Accessed June 26, 2019.
10. US Department of Veterans Affairs. VIONE program toolkit. https://www.vapulse.net/docs/DOC-259375. [Nonpublic source, not verified.]
Sequential CRT, immunotherapy nets high PFS in node-positive cervical cancer
Sequential chemoradiotherapy (CRT) and immunotherapy is safe, well tolerated, and efficacious among patients with locally advanced cervical cancer being treated with curative intent, a multicenter phase 1 trial suggests.
Less than 10% of patients treated with this sequence experienced a grade 3 toxicity. Meanwhile, more than 80% were alive and free of disease progression at 1 year.
“Despite standard CRT, most women with lymph node–positive cervical cancer experience disease recurrence,” note the investigators, led by Jyoti S. Mayadev, MD, associate professor in the department of radiation medicine and applied sciences, University of California, San Diego, in La Jolla. “Our study is potentially transformative in the standard treatment schema of locally advanced cervical cancer, with the prospect for immuno-oncology to add durable survival in patients with node-positive disease, a current unmet oncologic need.”
The investigators enrolled in the trial 32 women from Gynecology Oncology Cooperative Group member institutions who had stage IB2 to IVA cervical cancer with positive pelvic and/or para-aortic lymph nodes. Treatment consisted of six weekly doses of cisplatin, 40 mg/m2, concurrent with extended-field, 3-dimensional conformal radiotherapy, followed by the immune checkpoint inhibitor ipilimumab (Yervoy) every 21 days for four cycles.
Results reported in JAMA Oncology showed that all 32 patients completed CRT and 21 patients went on to receive ipilimumab. Among the latter, 86% completed all four planned cycles and the rest completed two cycles.
In the group receiving sequential CRT and ipilimumab, 9.5% experienced grade 3 toxicity (lipase increase in one case and dermatitis in another case). Both toxicities were self-limited.
With a 14.8-month median follow-up, the patients treated with CRT-ipilimumab had a 12-month overall survival rate of 90%, and a 12-month progression-free survival rate of 81% (median durations were not reached). Neither human papillomavirus genotype nor HLA subtype was associated with these outcomes.
Translational analyses showed that patients experienced an increase in peripheral blood T cells expressing programmed cell death 1 (PD-1) after CRT that was then sustained with ipilimumab therapy. “[T]he use of an immune checkpoint inhibitor could stimulate the antitumor activity of tumor-specific cytotoxic T cells and augment radiation-induced neoantigen load,” the investigators proposed.
“To our knowledge, this phase 1 study is the first to show tolerability with a signal of efficacy of an immune check-point inhibitor ... as a part of the definitive treatment of locally advanced cervical cancer,” they concluded. “Our findings show promise for the use of immunotherapy in the definitive setting of locally advanced, node-positive cervical cancer; patients with this cancer historically have a poor prognosis with standard therapy alone.”
Dr. Mayadev disclosed receiving a grant from the National Cancer Institute during the conduct of the study, personal fees from AstraZeneca, grants from NRG Oncology, and personal fees and nonfinancial support from the Gynecology Oncology Group Foundation outside the submitted work; receiving compensation for serving on the advisory board of Varian Medical Systems in 2018; and being a speaker for Samsung Medical Systems in 2017. The study was supported by the National Cancer Institute and by institutional funds.
SOURCE: Mayadev JS et al. JAMA Oncol. 2019 Nov 27. doi: 10.1001/jamaoncol.2019.3857.
Sequential chemoradiotherapy (CRT) and immunotherapy is safe, well tolerated, and efficacious among patients with locally advanced cervical cancer being treated with curative intent, a multicenter phase 1 trial suggests.
Less than 10% of patients treated with this sequence experienced a grade 3 toxicity. Meanwhile, more than 80% were alive and free of disease progression at 1 year.
“Despite standard CRT, most women with lymph node–positive cervical cancer experience disease recurrence,” note the investigators, led by Jyoti S. Mayadev, MD, associate professor in the department of radiation medicine and applied sciences, University of California, San Diego, in La Jolla. “Our study is potentially transformative in the standard treatment schema of locally advanced cervical cancer, with the prospect for immuno-oncology to add durable survival in patients with node-positive disease, a current unmet oncologic need.”
The investigators enrolled in the trial 32 women from Gynecology Oncology Cooperative Group member institutions who had stage IB2 to IVA cervical cancer with positive pelvic and/or para-aortic lymph nodes. Treatment consisted of six weekly doses of cisplatin, 40 mg/m2, concurrent with extended-field, 3-dimensional conformal radiotherapy, followed by the immune checkpoint inhibitor ipilimumab (Yervoy) every 21 days for four cycles.
Results reported in JAMA Oncology showed that all 32 patients completed CRT and 21 patients went on to receive ipilimumab. Among the latter, 86% completed all four planned cycles and the rest completed two cycles.
In the group receiving sequential CRT and ipilimumab, 9.5% experienced grade 3 toxicity (lipase increase in one case and dermatitis in another case). Both toxicities were self-limited.
With a 14.8-month median follow-up, the patients treated with CRT-ipilimumab had a 12-month overall survival rate of 90%, and a 12-month progression-free survival rate of 81% (median durations were not reached). Neither human papillomavirus genotype nor HLA subtype was associated with these outcomes.
Translational analyses showed that patients experienced an increase in peripheral blood T cells expressing programmed cell death 1 (PD-1) after CRT that was then sustained with ipilimumab therapy. “[T]he use of an immune checkpoint inhibitor could stimulate the antitumor activity of tumor-specific cytotoxic T cells and augment radiation-induced neoantigen load,” the investigators proposed.
“To our knowledge, this phase 1 study is the first to show tolerability with a signal of efficacy of an immune check-point inhibitor ... as a part of the definitive treatment of locally advanced cervical cancer,” they concluded. “Our findings show promise for the use of immunotherapy in the definitive setting of locally advanced, node-positive cervical cancer; patients with this cancer historically have a poor prognosis with standard therapy alone.”
Dr. Mayadev disclosed receiving a grant from the National Cancer Institute during the conduct of the study, personal fees from AstraZeneca, grants from NRG Oncology, and personal fees and nonfinancial support from the Gynecology Oncology Group Foundation outside the submitted work; receiving compensation for serving on the advisory board of Varian Medical Systems in 2018; and being a speaker for Samsung Medical Systems in 2017. The study was supported by the National Cancer Institute and by institutional funds.
SOURCE: Mayadev JS et al. JAMA Oncol. 2019 Nov 27. doi: 10.1001/jamaoncol.2019.3857.
Sequential chemoradiotherapy (CRT) and immunotherapy is safe, well tolerated, and efficacious among patients with locally advanced cervical cancer being treated with curative intent, a multicenter phase 1 trial suggests.
Less than 10% of patients treated with this sequence experienced a grade 3 toxicity. Meanwhile, more than 80% were alive and free of disease progression at 1 year.
“Despite standard CRT, most women with lymph node–positive cervical cancer experience disease recurrence,” note the investigators, led by Jyoti S. Mayadev, MD, associate professor in the department of radiation medicine and applied sciences, University of California, San Diego, in La Jolla. “Our study is potentially transformative in the standard treatment schema of locally advanced cervical cancer, with the prospect for immuno-oncology to add durable survival in patients with node-positive disease, a current unmet oncologic need.”
The investigators enrolled in the trial 32 women from Gynecology Oncology Cooperative Group member institutions who had stage IB2 to IVA cervical cancer with positive pelvic and/or para-aortic lymph nodes. Treatment consisted of six weekly doses of cisplatin, 40 mg/m2, concurrent with extended-field, 3-dimensional conformal radiotherapy, followed by the immune checkpoint inhibitor ipilimumab (Yervoy) every 21 days for four cycles.
Results reported in JAMA Oncology showed that all 32 patients completed CRT and 21 patients went on to receive ipilimumab. Among the latter, 86% completed all four planned cycles and the rest completed two cycles.
In the group receiving sequential CRT and ipilimumab, 9.5% experienced grade 3 toxicity (lipase increase in one case and dermatitis in another case). Both toxicities were self-limited.
With a 14.8-month median follow-up, the patients treated with CRT-ipilimumab had a 12-month overall survival rate of 90%, and a 12-month progression-free survival rate of 81% (median durations were not reached). Neither human papillomavirus genotype nor HLA subtype was associated with these outcomes.
Translational analyses showed that patients experienced an increase in peripheral blood T cells expressing programmed cell death 1 (PD-1) after CRT that was then sustained with ipilimumab therapy. “[T]he use of an immune checkpoint inhibitor could stimulate the antitumor activity of tumor-specific cytotoxic T cells and augment radiation-induced neoantigen load,” the investigators proposed.
“To our knowledge, this phase 1 study is the first to show tolerability with a signal of efficacy of an immune check-point inhibitor ... as a part of the definitive treatment of locally advanced cervical cancer,” they concluded. “Our findings show promise for the use of immunotherapy in the definitive setting of locally advanced, node-positive cervical cancer; patients with this cancer historically have a poor prognosis with standard therapy alone.”
Dr. Mayadev disclosed receiving a grant from the National Cancer Institute during the conduct of the study, personal fees from AstraZeneca, grants from NRG Oncology, and personal fees and nonfinancial support from the Gynecology Oncology Group Foundation outside the submitted work; receiving compensation for serving on the advisory board of Varian Medical Systems in 2018; and being a speaker for Samsung Medical Systems in 2017. The study was supported by the National Cancer Institute and by institutional funds.
SOURCE: Mayadev JS et al. JAMA Oncol. 2019 Nov 27. doi: 10.1001/jamaoncol.2019.3857.
FROM JAMA ONCOLOGY
Icosapent ethyl cost effective in REDUCE-IT analysis
PHILADELPHIA – The overall costs of icosapent ethyl were less than placebo, and the medication reduced cardiovascular events by 30% at a cost that fits well within acceptable quality-adjusted life-year (QALY) parameters, according to a cost-effectiveness analysis of the REDUCE-IT trial.
Days before the presentation of the analysis at the American Heart Association scientific sessions, a Food and Drug Administration advisory panel unanimously recommended approval of icosapent ethyl (Vascepa) for a new indication for reducing CV event risk. Icosapent ethyl, a highly purified form of the ethyl ester of eicosapentaenoic acid derived from fish oil, received FDA approval in 2012 for treatment of triglyceride levels of at least 500 mg/dL.
“What we found here is that icosapent ethyl is a dominant strategy,” said William S. Weintraub, MD, director of outcomes research at MedStar Heart & Vascular Institute in Washington, in reporting preliminary cost-analysis findings from REDUCE-IT (Reduction of Cardiovascular Events With Icosapent Ethyl – Intervention Trial). “It’s offering better outcomes at a lower cost.”
The dominant strategy was demonstrated by cost savings in 70% of simulations the cost-effectiveness analysis ran, Dr. Weintraub said.
“These are very impressive results,” said session moderator Seth S. Martin, MD, an internist and cardiologist at Johns Hopkins University, Baltimore. “We don’t often see dominant strategies for new drugs. This is very exciting.”
“Almost never,” Dr. Weintraub responded.
REDUCE-IT randomized 8,179 patients with a diagnosis of CVD or with diabetes and other risk factors who had been on statins and had triglycerides of 135-499 mg/dL to either 4 g of icosapent ethyl daily or placebo (N Engl J Med. 2019;380:11-22). Trial results showed the treatment group had an absolute risk reduction of 4.8% and a relative risk reduction of 25% of first CV events and a 30% relative risk reduction for total events, Dr. Weintraub said.
The analysis determined that the QALYs for icosapent ethyl versus those for placebo were 3.34 and 3.27, respectively, during the trial period and 11.61 and 11.35 over a lifetime. The mean costs for the two treatments were $27,576 and $28,205 during the trial period and $235,352 and $236,636 lifetime, respectively, Dr. Weintraub said.
An analysis of cost effectiveness showed that almost all of the estimates fell below the willingness-to-pay (WTP) threshold of $50,000 per QALY gained, Dr. Weintraub said. “In fact, some 70% plus are in what’s called quadrant two; that is, decreased cost and increased efficacy.”
The analysis also calculated the value of icosapent ethyl at three different WTP thresholds: up to $6 a day at a WTP of $50,000, up to $12 a day at $100,000, and up to $18 a day at $150,000. The analysis used the actual net pricing of $4.16 a day, Dr. Weintraub said. “That’s why we showed we have the dominant strategy,” he said.
Further cost-effectiveness analyses of the REDUCE-IT data will focus on subgroups, such as U.S. and non–U.S. patients and people with diabetes. He also emphasized the data he reported were preliminary. “We have a lot more work to do,” Dr. Weintraub said.
Dr. Weintraub reported having financial relationships with Amarin Pharma, which markets Vascepa, and AstraZeneca.
SOURCE: Weintraub WS. AHA 2019, Session FS.AOS.F1.
PHILADELPHIA – The overall costs of icosapent ethyl were less than placebo, and the medication reduced cardiovascular events by 30% at a cost that fits well within acceptable quality-adjusted life-year (QALY) parameters, according to a cost-effectiveness analysis of the REDUCE-IT trial.
Days before the presentation of the analysis at the American Heart Association scientific sessions, a Food and Drug Administration advisory panel unanimously recommended approval of icosapent ethyl (Vascepa) for a new indication for reducing CV event risk. Icosapent ethyl, a highly purified form of the ethyl ester of eicosapentaenoic acid derived from fish oil, received FDA approval in 2012 for treatment of triglyceride levels of at least 500 mg/dL.
“What we found here is that icosapent ethyl is a dominant strategy,” said William S. Weintraub, MD, director of outcomes research at MedStar Heart & Vascular Institute in Washington, in reporting preliminary cost-analysis findings from REDUCE-IT (Reduction of Cardiovascular Events With Icosapent Ethyl – Intervention Trial). “It’s offering better outcomes at a lower cost.”
The dominant strategy was demonstrated by cost savings in 70% of simulations the cost-effectiveness analysis ran, Dr. Weintraub said.
“These are very impressive results,” said session moderator Seth S. Martin, MD, an internist and cardiologist at Johns Hopkins University, Baltimore. “We don’t often see dominant strategies for new drugs. This is very exciting.”
“Almost never,” Dr. Weintraub responded.
REDUCE-IT randomized 8,179 patients with a diagnosis of CVD or with diabetes and other risk factors who had been on statins and had triglycerides of 135-499 mg/dL to either 4 g of icosapent ethyl daily or placebo (N Engl J Med. 2019;380:11-22). Trial results showed the treatment group had an absolute risk reduction of 4.8% and a relative risk reduction of 25% of first CV events and a 30% relative risk reduction for total events, Dr. Weintraub said.
The analysis determined that the QALYs for icosapent ethyl versus those for placebo were 3.34 and 3.27, respectively, during the trial period and 11.61 and 11.35 over a lifetime. The mean costs for the two treatments were $27,576 and $28,205 during the trial period and $235,352 and $236,636 lifetime, respectively, Dr. Weintraub said.
An analysis of cost effectiveness showed that almost all of the estimates fell below the willingness-to-pay (WTP) threshold of $50,000 per QALY gained, Dr. Weintraub said. “In fact, some 70% plus are in what’s called quadrant two; that is, decreased cost and increased efficacy.”
The analysis also calculated the value of icosapent ethyl at three different WTP thresholds: up to $6 a day at a WTP of $50,000, up to $12 a day at $100,000, and up to $18 a day at $150,000. The analysis used the actual net pricing of $4.16 a day, Dr. Weintraub said. “That’s why we showed we have the dominant strategy,” he said.
Further cost-effectiveness analyses of the REDUCE-IT data will focus on subgroups, such as U.S. and non–U.S. patients and people with diabetes. He also emphasized the data he reported were preliminary. “We have a lot more work to do,” Dr. Weintraub said.
Dr. Weintraub reported having financial relationships with Amarin Pharma, which markets Vascepa, and AstraZeneca.
SOURCE: Weintraub WS. AHA 2019, Session FS.AOS.F1.
PHILADELPHIA – The overall costs of icosapent ethyl were less than placebo, and the medication reduced cardiovascular events by 30% at a cost that fits well within acceptable quality-adjusted life-year (QALY) parameters, according to a cost-effectiveness analysis of the REDUCE-IT trial.
Days before the presentation of the analysis at the American Heart Association scientific sessions, a Food and Drug Administration advisory panel unanimously recommended approval of icosapent ethyl (Vascepa) for a new indication for reducing CV event risk. Icosapent ethyl, a highly purified form of the ethyl ester of eicosapentaenoic acid derived from fish oil, received FDA approval in 2012 for treatment of triglyceride levels of at least 500 mg/dL.
“What we found here is that icosapent ethyl is a dominant strategy,” said William S. Weintraub, MD, director of outcomes research at MedStar Heart & Vascular Institute in Washington, in reporting preliminary cost-analysis findings from REDUCE-IT (Reduction of Cardiovascular Events With Icosapent Ethyl – Intervention Trial). “It’s offering better outcomes at a lower cost.”
The dominant strategy was demonstrated by cost savings in 70% of simulations the cost-effectiveness analysis ran, Dr. Weintraub said.
“These are very impressive results,” said session moderator Seth S. Martin, MD, an internist and cardiologist at Johns Hopkins University, Baltimore. “We don’t often see dominant strategies for new drugs. This is very exciting.”
“Almost never,” Dr. Weintraub responded.
REDUCE-IT randomized 8,179 patients with a diagnosis of CVD or with diabetes and other risk factors who had been on statins and had triglycerides of 135-499 mg/dL to either 4 g of icosapent ethyl daily or placebo (N Engl J Med. 2019;380:11-22). Trial results showed the treatment group had an absolute risk reduction of 4.8% and a relative risk reduction of 25% of first CV events and a 30% relative risk reduction for total events, Dr. Weintraub said.
The analysis determined that the QALYs for icosapent ethyl versus those for placebo were 3.34 and 3.27, respectively, during the trial period and 11.61 and 11.35 over a lifetime. The mean costs for the two treatments were $27,576 and $28,205 during the trial period and $235,352 and $236,636 lifetime, respectively, Dr. Weintraub said.
An analysis of cost effectiveness showed that almost all of the estimates fell below the willingness-to-pay (WTP) threshold of $50,000 per QALY gained, Dr. Weintraub said. “In fact, some 70% plus are in what’s called quadrant two; that is, decreased cost and increased efficacy.”
The analysis also calculated the value of icosapent ethyl at three different WTP thresholds: up to $6 a day at a WTP of $50,000, up to $12 a day at $100,000, and up to $18 a day at $150,000. The analysis used the actual net pricing of $4.16 a day, Dr. Weintraub said. “That’s why we showed we have the dominant strategy,” he said.
Further cost-effectiveness analyses of the REDUCE-IT data will focus on subgroups, such as U.S. and non–U.S. patients and people with diabetes. He also emphasized the data he reported were preliminary. “We have a lot more work to do,” Dr. Weintraub said.
Dr. Weintraub reported having financial relationships with Amarin Pharma, which markets Vascepa, and AstraZeneca.
SOURCE: Weintraub WS. AHA 2019, Session FS.AOS.F1.
REPORTING FROM AHA 2019
DAPA-HF: Dapagliflozin benefits regardless of age, HF severity
PHILADELPHIA – The substantial benefits from adding dapagliflozin to guideline-directed medical therapy for patients with heart failure with reduced ejection fraction enrolled in the DAPA-HF trial applied to patients regardless of their age or baseline health status, a pair of new post hoc analyses suggest.
These findings emerged a day after a report that more fully delineated dapagliflozin’s consistent safety and efficacy in patients with heart failure with reduced ejection fraction (HFrEF) regardless of whether they also had type 2 diabetes. One of the new, post hoc analyses reported at the American Heart Association scientific sessions suggested that even the most elderly enrolled patients, 75 years and older, had a similar cut in mortality and acute heart failure exacerbations, compared with younger patients. A second post hoc analysis indicated that patients with severe heart failure symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with mild baseline symptoms, measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ).
The primary results from the DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial, first reported in August 2019, showed that among more than 4,700 patients with HFrEF randomized to receive the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) on top of standard HFrEF medications or placebo, those who received dapagliflozin had a statistically significant, 26% decrease in their incidence of the primary study endpoint over a median 18 months, regardless of diabetes status (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“These benefits were entirely consistent across the range of ages studied,” extending from patients younger than 55 years to those older than 75 years, John McMurray, MD, said at the meeting. “In many parts of the world, particularly North America and Western Europe, we have an increasingly elderly population. Many patients with heart failure are much older than in clinical trials,” he said.
“The thing of concern is whether elderly patients get as much benefit and tolerate treatment as well as younger patients,” said Dr. McMurray, professor of medical cardiology at the University of Glasgow.
“Dapagliflozin worked across all ages, including some very elderly patients enrolled in the trial,” said Mary Norine Walsh, MD, medical director of the heart failure and transplant program at St. Vincent Heart Center of Indiana in Indianapolis. “Many trials have not looked at age like this. I hope this is a new way to analyze trials to produce more information that can help patients,” she said in an interview.
Quality-of-life outcomes
The other new, post hoc analysis showed that patients with severe HF symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with milder baseline symptoms and less impaired function, measured by the KCCQ. Dapagliflozin treatment “improved cardiovascular death and worsening heart failure to a similar extent across the entire range of KCCQ at baseline,” Mikhail N. Kosiborod, MD, said in a separate talk at the meeting. In addition, dapagliflozin treatment increased the rate of small, moderate, and large clinically meaningful improvements in patients’ KCCQ scores across all key domains of the metric, which scores symptom frequency and severity, physical and social limitations, and quality of life, said Dr. Kosiborod, a cardiologist and professor of medicine at the University of Missouri–Kansas City.
After the first 8 months of treatment in the DAPA-HF trial, 58% of the 2,373 patients who received dapagliflozin had a clinically meaningful improvement in their total KCCQ symptom score of at least 5 points, compared with a 51% rate in the 2,371 patients in the control arm, a statistically significant difference. This meant that the number needed to treat with dapagliflozin was 14 patients to produce one additional patient with at least a 5-point KCCQ improvement compared with controls, a “very small” number needed to treat, Dr. Kosiborod said in an interview.
Addition of the KCCQ to the panel of assessments that patients underwent during DAPA-HF reflected an evolved approach to measuring efficacy outcomes in clinical trials by including patient-reported outcomes. Earlier in 2019, the Food and Drug Administration released draft guidance for heart failure drug development that explicitly called for efficacy endpoints in pivotal studies that measure how patients feel and function, and stating that these endpoints can be the basis for new drug approvals.
“To many patients, how they feel matters as much if not more than how long they live,” Dr. Kosiborod noted. The goals of heart failure treatments are not only to extend survival and reduced hospitalizations, but also to improve symptoms, function, and quality of life, he said.
“There is a lot of interest now in having outcomes in heart failure trials that are more meaningful to patients, like feeling better and being able to do more,” noted Dr. Walsh.
The DAPA-HF results also showed that patients had similar rates of reduction in death, heart failure hospitalization, or urgent clinical visits, regardless of how severely they were affected by their heart failure when they began dapagliflozin treatment. The researchers ran an analysis that divided the entire trial population into tertiles based on their KCCQ score on entering the study. Patients in the most severely-affected tertile had a 30% cut in their rate of death or acute heart failure exacerbation on dapagliflozin compared with placebo, while patients in the tertile with the mildest symptoms at baseline had a 38% reduction in their primary outcome incidence compared with controls who received placebo. Concurrently with Dr. Kosiborod’s report, the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044138).
Outcomes by age
Not surprisingly in DAPA-HF, the older patients were, the sicker, Dr. McMurray observed. Of the study’s 1,149 patients (24% of the study cohort) who were at least 75 years old, 62% had chronic kidney disease, compared with a 14% prevalence among the 636 patients younger than age 55. The 75-and-older group showed a steeper, 32% decline in incidence of the primary endpoint – a composite of cardiovascular (CV) death, HF hospitalization, or urgent HF visit requiring intravenous therapy – than in the other studied age groups: a 24% decline in those 65-74 years old, a 29% cut in those 55-64 years old, and a 13% drop in patients younger than 55 years old.
In addition, patients aged 75 years or greater were just as likely as the overall group to show at least a 5-point improvement in their KCCQ Total Symptom Score on dapagliflozin, as well as about the same reduced rate of deterioration compared with placebo as tracked with the KCCQ.
Patients “got as much benefit in terms of symptoms as well as morbidity and mortality,” Dr. McMurray concluded. Concurrently with the meeting report the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044133).
“These data are of critical importance, as improving patient-reported outcomes in heart failure, especially in highly symptomatic patients, is an important goal in drug development,” G. Michael Felker, MD, wrote in an editorial accompanying the two published analyses (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044578). These new analyses also highlight another attractive feature of dapagliflozin and, apparently, the entire class of SGLT2 inhibitors: They “ ‘play well with others’ when it comes to overlapping intolerances that often limit (either in reality or in perception) optimization of GDMT [guideline-directed medical therapy]. Although SGLT2 inhibitor therapy may lead to volume depletion and require adjustment of diuretics, the SGLT2 inhibitors generally lack some of the other dose-limiting adverse effects (such as renal dysfunction, hyperkalemia, and hypotension) that can make aggressive up-titration of GDMT problematic, particularly in older patients or those with more advanced disease,“ wrote Dr. Felker, professor of medicine at Duke University in Durham, N.C. “We stand at the beginning of a new era of ‘quadruple therapy’ for HFrEF with beta-blockers, an angiotensin receptor neprilysin inhibitor, mineralocorticoid receptor antagonists, and SGLT2 inhibitors,” he concluded.
A version of this article also appears on Medscape.com
In DAPA-HF, treatment with dapagliflozin met the three critical goals of heart failure management. When used on top of current guideline-directed medical therapy, the treatment reduced mortality, cut hospitalizations, and improved heart failure–related health status – all to a similar extent regardless of patients’ age or symptom severity at entry. These new, post hoc findings provide important, additional data supporting inhibition of sodium-glucose cotransporter (SGLT) 2 with dapagliflozin as the newest foundational pillar of treatment for heart failure with reduced ejection fraction (HFrEF).
The results of the analysis by baseline symptoms severity as measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ) showed similar treatment effects from dapagliflozin regardless of a patient’s baseline KCCQ score, suggesting that the prior report of a blunted effect of dapagliflozin in patients classified at baseline as being in New York Heart Association functional class III or IV compared with class I and II patients was likely a chance finding.
Both the analyses by age and by KCCQ scores were limited by their post hoc status using data collected in a single study. No evidence addresses whether these are class effects for all drugs in the SGLT2-inhibitor class, whether these findings from DAPA-HF are generalizable to real world practice, or whether treatment with dapagliflozin would have similar effects on outcomes if it had been used more often in combination with sacubitril/valsartan. In DAPA-HF, 11% of patients also received sacubitril/valsartan even though existing management guidelines recommend sacubitril/valsartan as the preferred agent for inhibiting the renin-angiotensin system.
It’s also unclear whether patient-reported outcomes such as those measured by the KCCQ will help in sequencing the introduction of drugs for HFrEF patients, or drug selection by patients, providers, payers, and in guidelines.
Carolyn S.P. Lam, MD, is professor of medicine at Duke-National University of Singapore. She has been a consultant to and has received research funding from AstraZeneca and several other companies. She made these comments as designated discussant for the two reports.
In DAPA-HF, treatment with dapagliflozin met the three critical goals of heart failure management. When used on top of current guideline-directed medical therapy, the treatment reduced mortality, cut hospitalizations, and improved heart failure–related health status – all to a similar extent regardless of patients’ age or symptom severity at entry. These new, post hoc findings provide important, additional data supporting inhibition of sodium-glucose cotransporter (SGLT) 2 with dapagliflozin as the newest foundational pillar of treatment for heart failure with reduced ejection fraction (HFrEF).
The results of the analysis by baseline symptoms severity as measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ) showed similar treatment effects from dapagliflozin regardless of a patient’s baseline KCCQ score, suggesting that the prior report of a blunted effect of dapagliflozin in patients classified at baseline as being in New York Heart Association functional class III or IV compared with class I and II patients was likely a chance finding.
Both the analyses by age and by KCCQ scores were limited by their post hoc status using data collected in a single study. No evidence addresses whether these are class effects for all drugs in the SGLT2-inhibitor class, whether these findings from DAPA-HF are generalizable to real world practice, or whether treatment with dapagliflozin would have similar effects on outcomes if it had been used more often in combination with sacubitril/valsartan. In DAPA-HF, 11% of patients also received sacubitril/valsartan even though existing management guidelines recommend sacubitril/valsartan as the preferred agent for inhibiting the renin-angiotensin system.
It’s also unclear whether patient-reported outcomes such as those measured by the KCCQ will help in sequencing the introduction of drugs for HFrEF patients, or drug selection by patients, providers, payers, and in guidelines.
Carolyn S.P. Lam, MD, is professor of medicine at Duke-National University of Singapore. She has been a consultant to and has received research funding from AstraZeneca and several other companies. She made these comments as designated discussant for the two reports.
In DAPA-HF, treatment with dapagliflozin met the three critical goals of heart failure management. When used on top of current guideline-directed medical therapy, the treatment reduced mortality, cut hospitalizations, and improved heart failure–related health status – all to a similar extent regardless of patients’ age or symptom severity at entry. These new, post hoc findings provide important, additional data supporting inhibition of sodium-glucose cotransporter (SGLT) 2 with dapagliflozin as the newest foundational pillar of treatment for heart failure with reduced ejection fraction (HFrEF).
The results of the analysis by baseline symptoms severity as measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ) showed similar treatment effects from dapagliflozin regardless of a patient’s baseline KCCQ score, suggesting that the prior report of a blunted effect of dapagliflozin in patients classified at baseline as being in New York Heart Association functional class III or IV compared with class I and II patients was likely a chance finding.
Both the analyses by age and by KCCQ scores were limited by their post hoc status using data collected in a single study. No evidence addresses whether these are class effects for all drugs in the SGLT2-inhibitor class, whether these findings from DAPA-HF are generalizable to real world practice, or whether treatment with dapagliflozin would have similar effects on outcomes if it had been used more often in combination with sacubitril/valsartan. In DAPA-HF, 11% of patients also received sacubitril/valsartan even though existing management guidelines recommend sacubitril/valsartan as the preferred agent for inhibiting the renin-angiotensin system.
It’s also unclear whether patient-reported outcomes such as those measured by the KCCQ will help in sequencing the introduction of drugs for HFrEF patients, or drug selection by patients, providers, payers, and in guidelines.
Carolyn S.P. Lam, MD, is professor of medicine at Duke-National University of Singapore. She has been a consultant to and has received research funding from AstraZeneca and several other companies. She made these comments as designated discussant for the two reports.
PHILADELPHIA – The substantial benefits from adding dapagliflozin to guideline-directed medical therapy for patients with heart failure with reduced ejection fraction enrolled in the DAPA-HF trial applied to patients regardless of their age or baseline health status, a pair of new post hoc analyses suggest.
These findings emerged a day after a report that more fully delineated dapagliflozin’s consistent safety and efficacy in patients with heart failure with reduced ejection fraction (HFrEF) regardless of whether they also had type 2 diabetes. One of the new, post hoc analyses reported at the American Heart Association scientific sessions suggested that even the most elderly enrolled patients, 75 years and older, had a similar cut in mortality and acute heart failure exacerbations, compared with younger patients. A second post hoc analysis indicated that patients with severe heart failure symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with mild baseline symptoms, measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ).
The primary results from the DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial, first reported in August 2019, showed that among more than 4,700 patients with HFrEF randomized to receive the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) on top of standard HFrEF medications or placebo, those who received dapagliflozin had a statistically significant, 26% decrease in their incidence of the primary study endpoint over a median 18 months, regardless of diabetes status (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“These benefits were entirely consistent across the range of ages studied,” extending from patients younger than 55 years to those older than 75 years, John McMurray, MD, said at the meeting. “In many parts of the world, particularly North America and Western Europe, we have an increasingly elderly population. Many patients with heart failure are much older than in clinical trials,” he said.
“The thing of concern is whether elderly patients get as much benefit and tolerate treatment as well as younger patients,” said Dr. McMurray, professor of medical cardiology at the University of Glasgow.
“Dapagliflozin worked across all ages, including some very elderly patients enrolled in the trial,” said Mary Norine Walsh, MD, medical director of the heart failure and transplant program at St. Vincent Heart Center of Indiana in Indianapolis. “Many trials have not looked at age like this. I hope this is a new way to analyze trials to produce more information that can help patients,” she said in an interview.
Quality-of-life outcomes
The other new, post hoc analysis showed that patients with severe HF symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with milder baseline symptoms and less impaired function, measured by the KCCQ. Dapagliflozin treatment “improved cardiovascular death and worsening heart failure to a similar extent across the entire range of KCCQ at baseline,” Mikhail N. Kosiborod, MD, said in a separate talk at the meeting. In addition, dapagliflozin treatment increased the rate of small, moderate, and large clinically meaningful improvements in patients’ KCCQ scores across all key domains of the metric, which scores symptom frequency and severity, physical and social limitations, and quality of life, said Dr. Kosiborod, a cardiologist and professor of medicine at the University of Missouri–Kansas City.
After the first 8 months of treatment in the DAPA-HF trial, 58% of the 2,373 patients who received dapagliflozin had a clinically meaningful improvement in their total KCCQ symptom score of at least 5 points, compared with a 51% rate in the 2,371 patients in the control arm, a statistically significant difference. This meant that the number needed to treat with dapagliflozin was 14 patients to produce one additional patient with at least a 5-point KCCQ improvement compared with controls, a “very small” number needed to treat, Dr. Kosiborod said in an interview.
Addition of the KCCQ to the panel of assessments that patients underwent during DAPA-HF reflected an evolved approach to measuring efficacy outcomes in clinical trials by including patient-reported outcomes. Earlier in 2019, the Food and Drug Administration released draft guidance for heart failure drug development that explicitly called for efficacy endpoints in pivotal studies that measure how patients feel and function, and stating that these endpoints can be the basis for new drug approvals.
“To many patients, how they feel matters as much if not more than how long they live,” Dr. Kosiborod noted. The goals of heart failure treatments are not only to extend survival and reduced hospitalizations, but also to improve symptoms, function, and quality of life, he said.
“There is a lot of interest now in having outcomes in heart failure trials that are more meaningful to patients, like feeling better and being able to do more,” noted Dr. Walsh.
The DAPA-HF results also showed that patients had similar rates of reduction in death, heart failure hospitalization, or urgent clinical visits, regardless of how severely they were affected by their heart failure when they began dapagliflozin treatment. The researchers ran an analysis that divided the entire trial population into tertiles based on their KCCQ score on entering the study. Patients in the most severely-affected tertile had a 30% cut in their rate of death or acute heart failure exacerbation on dapagliflozin compared with placebo, while patients in the tertile with the mildest symptoms at baseline had a 38% reduction in their primary outcome incidence compared with controls who received placebo. Concurrently with Dr. Kosiborod’s report, the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044138).
Outcomes by age
Not surprisingly in DAPA-HF, the older patients were, the sicker, Dr. McMurray observed. Of the study’s 1,149 patients (24% of the study cohort) who were at least 75 years old, 62% had chronic kidney disease, compared with a 14% prevalence among the 636 patients younger than age 55. The 75-and-older group showed a steeper, 32% decline in incidence of the primary endpoint – a composite of cardiovascular (CV) death, HF hospitalization, or urgent HF visit requiring intravenous therapy – than in the other studied age groups: a 24% decline in those 65-74 years old, a 29% cut in those 55-64 years old, and a 13% drop in patients younger than 55 years old.
In addition, patients aged 75 years or greater were just as likely as the overall group to show at least a 5-point improvement in their KCCQ Total Symptom Score on dapagliflozin, as well as about the same reduced rate of deterioration compared with placebo as tracked with the KCCQ.
Patients “got as much benefit in terms of symptoms as well as morbidity and mortality,” Dr. McMurray concluded. Concurrently with the meeting report the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044133).
“These data are of critical importance, as improving patient-reported outcomes in heart failure, especially in highly symptomatic patients, is an important goal in drug development,” G. Michael Felker, MD, wrote in an editorial accompanying the two published analyses (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044578). These new analyses also highlight another attractive feature of dapagliflozin and, apparently, the entire class of SGLT2 inhibitors: They “ ‘play well with others’ when it comes to overlapping intolerances that often limit (either in reality or in perception) optimization of GDMT [guideline-directed medical therapy]. Although SGLT2 inhibitor therapy may lead to volume depletion and require adjustment of diuretics, the SGLT2 inhibitors generally lack some of the other dose-limiting adverse effects (such as renal dysfunction, hyperkalemia, and hypotension) that can make aggressive up-titration of GDMT problematic, particularly in older patients or those with more advanced disease,“ wrote Dr. Felker, professor of medicine at Duke University in Durham, N.C. “We stand at the beginning of a new era of ‘quadruple therapy’ for HFrEF with beta-blockers, an angiotensin receptor neprilysin inhibitor, mineralocorticoid receptor antagonists, and SGLT2 inhibitors,” he concluded.
A version of this article also appears on Medscape.com
PHILADELPHIA – The substantial benefits from adding dapagliflozin to guideline-directed medical therapy for patients with heart failure with reduced ejection fraction enrolled in the DAPA-HF trial applied to patients regardless of their age or baseline health status, a pair of new post hoc analyses suggest.
These findings emerged a day after a report that more fully delineated dapagliflozin’s consistent safety and efficacy in patients with heart failure with reduced ejection fraction (HFrEF) regardless of whether they also had type 2 diabetes. One of the new, post hoc analyses reported at the American Heart Association scientific sessions suggested that even the most elderly enrolled patients, 75 years and older, had a similar cut in mortality and acute heart failure exacerbations, compared with younger patients. A second post hoc analysis indicated that patients with severe heart failure symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with mild baseline symptoms, measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ).
The primary results from the DAPA-HF (Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure) trial, first reported in August 2019, showed that among more than 4,700 patients with HFrEF randomized to receive the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) on top of standard HFrEF medications or placebo, those who received dapagliflozin had a statistically significant, 26% decrease in their incidence of the primary study endpoint over a median 18 months, regardless of diabetes status (N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“These benefits were entirely consistent across the range of ages studied,” extending from patients younger than 55 years to those older than 75 years, John McMurray, MD, said at the meeting. “In many parts of the world, particularly North America and Western Europe, we have an increasingly elderly population. Many patients with heart failure are much older than in clinical trials,” he said.
“The thing of concern is whether elderly patients get as much benefit and tolerate treatment as well as younger patients,” said Dr. McMurray, professor of medical cardiology at the University of Glasgow.
“Dapagliflozin worked across all ages, including some very elderly patients enrolled in the trial,” said Mary Norine Walsh, MD, medical director of the heart failure and transplant program at St. Vincent Heart Center of Indiana in Indianapolis. “Many trials have not looked at age like this. I hope this is a new way to analyze trials to produce more information that can help patients,” she said in an interview.
Quality-of-life outcomes
The other new, post hoc analysis showed that patients with severe HF symptoms at entry into the trial received about as much benefit from the addition of dapagliflozin as did patients with milder baseline symptoms and less impaired function, measured by the KCCQ. Dapagliflozin treatment “improved cardiovascular death and worsening heart failure to a similar extent across the entire range of KCCQ at baseline,” Mikhail N. Kosiborod, MD, said in a separate talk at the meeting. In addition, dapagliflozin treatment increased the rate of small, moderate, and large clinically meaningful improvements in patients’ KCCQ scores across all key domains of the metric, which scores symptom frequency and severity, physical and social limitations, and quality of life, said Dr. Kosiborod, a cardiologist and professor of medicine at the University of Missouri–Kansas City.
After the first 8 months of treatment in the DAPA-HF trial, 58% of the 2,373 patients who received dapagliflozin had a clinically meaningful improvement in their total KCCQ symptom score of at least 5 points, compared with a 51% rate in the 2,371 patients in the control arm, a statistically significant difference. This meant that the number needed to treat with dapagliflozin was 14 patients to produce one additional patient with at least a 5-point KCCQ improvement compared with controls, a “very small” number needed to treat, Dr. Kosiborod said in an interview.
Addition of the KCCQ to the panel of assessments that patients underwent during DAPA-HF reflected an evolved approach to measuring efficacy outcomes in clinical trials by including patient-reported outcomes. Earlier in 2019, the Food and Drug Administration released draft guidance for heart failure drug development that explicitly called for efficacy endpoints in pivotal studies that measure how patients feel and function, and stating that these endpoints can be the basis for new drug approvals.
“To many patients, how they feel matters as much if not more than how long they live,” Dr. Kosiborod noted. The goals of heart failure treatments are not only to extend survival and reduced hospitalizations, but also to improve symptoms, function, and quality of life, he said.
“There is a lot of interest now in having outcomes in heart failure trials that are more meaningful to patients, like feeling better and being able to do more,” noted Dr. Walsh.
The DAPA-HF results also showed that patients had similar rates of reduction in death, heart failure hospitalization, or urgent clinical visits, regardless of how severely they were affected by their heart failure when they began dapagliflozin treatment. The researchers ran an analysis that divided the entire trial population into tertiles based on their KCCQ score on entering the study. Patients in the most severely-affected tertile had a 30% cut in their rate of death or acute heart failure exacerbation on dapagliflozin compared with placebo, while patients in the tertile with the mildest symptoms at baseline had a 38% reduction in their primary outcome incidence compared with controls who received placebo. Concurrently with Dr. Kosiborod’s report, the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044138).
Outcomes by age
Not surprisingly in DAPA-HF, the older patients were, the sicker, Dr. McMurray observed. Of the study’s 1,149 patients (24% of the study cohort) who were at least 75 years old, 62% had chronic kidney disease, compared with a 14% prevalence among the 636 patients younger than age 55. The 75-and-older group showed a steeper, 32% decline in incidence of the primary endpoint – a composite of cardiovascular (CV) death, HF hospitalization, or urgent HF visit requiring intravenous therapy – than in the other studied age groups: a 24% decline in those 65-74 years old, a 29% cut in those 55-64 years old, and a 13% drop in patients younger than 55 years old.
In addition, patients aged 75 years or greater were just as likely as the overall group to show at least a 5-point improvement in their KCCQ Total Symptom Score on dapagliflozin, as well as about the same reduced rate of deterioration compared with placebo as tracked with the KCCQ.
Patients “got as much benefit in terms of symptoms as well as morbidity and mortality,” Dr. McMurray concluded. Concurrently with the meeting report the results appeared in an article online (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044133).
“These data are of critical importance, as improving patient-reported outcomes in heart failure, especially in highly symptomatic patients, is an important goal in drug development,” G. Michael Felker, MD, wrote in an editorial accompanying the two published analyses (Circulation. 2019 Nov 17. doi: 10.1161/CIRCULATIONAHA.119.044578). These new analyses also highlight another attractive feature of dapagliflozin and, apparently, the entire class of SGLT2 inhibitors: They “ ‘play well with others’ when it comes to overlapping intolerances that often limit (either in reality or in perception) optimization of GDMT [guideline-directed medical therapy]. Although SGLT2 inhibitor therapy may lead to volume depletion and require adjustment of diuretics, the SGLT2 inhibitors generally lack some of the other dose-limiting adverse effects (such as renal dysfunction, hyperkalemia, and hypotension) that can make aggressive up-titration of GDMT problematic, particularly in older patients or those with more advanced disease,“ wrote Dr. Felker, professor of medicine at Duke University in Durham, N.C. “We stand at the beginning of a new era of ‘quadruple therapy’ for HFrEF with beta-blockers, an angiotensin receptor neprilysin inhibitor, mineralocorticoid receptor antagonists, and SGLT2 inhibitors,” he concluded.
A version of this article also appears on Medscape.com
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
Atezolizumab/bevacizumab may offer benefit to patients with RCC
The combination of atezolizumab plus bevacizumab may offer some benefit to patients with advanced renal cell carcinoma, especially those who are positive for programmed death-ligand 1 (PD-L1), investigators report.
The overall response rate (ORR) among such patients was 60%, compared with 19% in PD-L1–negative patients, Bradley A. McGregor, MD, clinical director for the Lank Center of Genitourinary Oncology at Dana-Farber Cancer Institute in Boston, and colleagues reported in the Journal of Clinical Oncology.
The data were presented last summer at the American Society of Clinical Oncology Annual Meeting in Chicago.
The phase 2 study comprised 60 patients, 42 of whom had variant histology RCC, and 18 of whom had clear cell RCC (ccRCC ) with at least 20% sarcomatoid differentiation. All patients had advanced renal cell carcinoma of various histologies, including papillary (12), chromophobe (10), unclassified (9), TFE3 translocation (5), collecting duct (5), and medullary (1). Most (65%) had not received prior systemic therapy.
They all received infusions of atezolizumab 1,200 mg plus bevacizumab 15 mg/kg every 3 weeks. No dose modifications were allowed. Dose delays were allowed, and patients could also drop one agent and continue with the other. Treatment continued until disease progression, toxicity, or intolerable side effects.
The median number of cycles was 9.5, although the range was wide (1-42). At analysis, 15 were still on the treatment, but 45 had dropped out. Reasons were disease progression (34), death (1), toxicity (5), or unspecified (8). Six patients delayed bevacizumab doses, half because of adverse events.
After a median follow-up of 13 months, the ORR was 33%. Those with ccRCC with sarcomatoid differentiation responded best to the combination (ORR, 50%). Those with variant-histology RCC responded less robustly (ORR, 26%).
ORR varied by baseline risk category, being 33% in favorable-, 45% in intermediate-, and 11% in poor-risk patients. Median time to response was 2.7 months, median response duration was 8.9 months, and median progression-free survival was 8.3 months.
PD-L1 status was determined in 36 patients; 15 were positive. Among the positive patents, ORR was 60%, compared with 19% in PD-L1 negative patients. Response rates varied with tumor characteristics. Among patients with ccRCC with sarcomatoid differentiation, the ORR was 50% in PD-L1–positive patients and 29% in negative patients. In patients with variant histology RCC, the ORR was also better in PD-L1 positive patients (67% vs. 14%).
The most common treatment-related side effects were fatigue (35%), proteinuria (35%), musculoskeletal pain (33%), diarrhea (22%), rash (20%), hypertension (18%), pruritus (18%), thyroid dysfunction (17%), hepatitis (15%), fever (13%), and mucositis (12%). Thirty-four patients developed at least one grade 3 adverse event; there were no grade 4 or 5 toxicities. One patient died, presumably because of disease progression.
Quality of life scores were largely stable during treatment.
“The combination demonstrated responses across several subtypes of RCC, including collecting duct and medullary carcinoma, histologies that are often treated with cytotoxic chemotherapy,” the authors said. “This is notable given the generally poor prognosis and low response rate associated with variant histology RCC in trials to date.”
The study also suggests the PD-L1 status might be “intriguing as a biomarker for response to atezolizumab and bevacizumab in variant histology RCC. We plan to conduct additional correlative work, including genomic profiling and assessment of the immune microenvironment, to better elucidate markers of response and resistance,” the authors wrote.
SOURCE: McGregor BA et al. J Clin Oncol. 2019 Nov 13. doi: 10.1200/JCO.19.01882.
The combination of atezolizumab plus bevacizumab may offer some benefit to patients with advanced renal cell carcinoma, especially those who are positive for programmed death-ligand 1 (PD-L1), investigators report.
The overall response rate (ORR) among such patients was 60%, compared with 19% in PD-L1–negative patients, Bradley A. McGregor, MD, clinical director for the Lank Center of Genitourinary Oncology at Dana-Farber Cancer Institute in Boston, and colleagues reported in the Journal of Clinical Oncology.
The data were presented last summer at the American Society of Clinical Oncology Annual Meeting in Chicago.
The phase 2 study comprised 60 patients, 42 of whom had variant histology RCC, and 18 of whom had clear cell RCC (ccRCC ) with at least 20% sarcomatoid differentiation. All patients had advanced renal cell carcinoma of various histologies, including papillary (12), chromophobe (10), unclassified (9), TFE3 translocation (5), collecting duct (5), and medullary (1). Most (65%) had not received prior systemic therapy.
They all received infusions of atezolizumab 1,200 mg plus bevacizumab 15 mg/kg every 3 weeks. No dose modifications were allowed. Dose delays were allowed, and patients could also drop one agent and continue with the other. Treatment continued until disease progression, toxicity, or intolerable side effects.
The median number of cycles was 9.5, although the range was wide (1-42). At analysis, 15 were still on the treatment, but 45 had dropped out. Reasons were disease progression (34), death (1), toxicity (5), or unspecified (8). Six patients delayed bevacizumab doses, half because of adverse events.
After a median follow-up of 13 months, the ORR was 33%. Those with ccRCC with sarcomatoid differentiation responded best to the combination (ORR, 50%). Those with variant-histology RCC responded less robustly (ORR, 26%).
ORR varied by baseline risk category, being 33% in favorable-, 45% in intermediate-, and 11% in poor-risk patients. Median time to response was 2.7 months, median response duration was 8.9 months, and median progression-free survival was 8.3 months.
PD-L1 status was determined in 36 patients; 15 were positive. Among the positive patents, ORR was 60%, compared with 19% in PD-L1 negative patients. Response rates varied with tumor characteristics. Among patients with ccRCC with sarcomatoid differentiation, the ORR was 50% in PD-L1–positive patients and 29% in negative patients. In patients with variant histology RCC, the ORR was also better in PD-L1 positive patients (67% vs. 14%).
The most common treatment-related side effects were fatigue (35%), proteinuria (35%), musculoskeletal pain (33%), diarrhea (22%), rash (20%), hypertension (18%), pruritus (18%), thyroid dysfunction (17%), hepatitis (15%), fever (13%), and mucositis (12%). Thirty-four patients developed at least one grade 3 adverse event; there were no grade 4 or 5 toxicities. One patient died, presumably because of disease progression.
Quality of life scores were largely stable during treatment.
“The combination demonstrated responses across several subtypes of RCC, including collecting duct and medullary carcinoma, histologies that are often treated with cytotoxic chemotherapy,” the authors said. “This is notable given the generally poor prognosis and low response rate associated with variant histology RCC in trials to date.”
The study also suggests the PD-L1 status might be “intriguing as a biomarker for response to atezolizumab and bevacizumab in variant histology RCC. We plan to conduct additional correlative work, including genomic profiling and assessment of the immune microenvironment, to better elucidate markers of response and resistance,” the authors wrote.
SOURCE: McGregor BA et al. J Clin Oncol. 2019 Nov 13. doi: 10.1200/JCO.19.01882.
The combination of atezolizumab plus bevacizumab may offer some benefit to patients with advanced renal cell carcinoma, especially those who are positive for programmed death-ligand 1 (PD-L1), investigators report.
The overall response rate (ORR) among such patients was 60%, compared with 19% in PD-L1–negative patients, Bradley A. McGregor, MD, clinical director for the Lank Center of Genitourinary Oncology at Dana-Farber Cancer Institute in Boston, and colleagues reported in the Journal of Clinical Oncology.
The data were presented last summer at the American Society of Clinical Oncology Annual Meeting in Chicago.
The phase 2 study comprised 60 patients, 42 of whom had variant histology RCC, and 18 of whom had clear cell RCC (ccRCC ) with at least 20% sarcomatoid differentiation. All patients had advanced renal cell carcinoma of various histologies, including papillary (12), chromophobe (10), unclassified (9), TFE3 translocation (5), collecting duct (5), and medullary (1). Most (65%) had not received prior systemic therapy.
They all received infusions of atezolizumab 1,200 mg plus bevacizumab 15 mg/kg every 3 weeks. No dose modifications were allowed. Dose delays were allowed, and patients could also drop one agent and continue with the other. Treatment continued until disease progression, toxicity, or intolerable side effects.
The median number of cycles was 9.5, although the range was wide (1-42). At analysis, 15 were still on the treatment, but 45 had dropped out. Reasons were disease progression (34), death (1), toxicity (5), or unspecified (8). Six patients delayed bevacizumab doses, half because of adverse events.
After a median follow-up of 13 months, the ORR was 33%. Those with ccRCC with sarcomatoid differentiation responded best to the combination (ORR, 50%). Those with variant-histology RCC responded less robustly (ORR, 26%).
ORR varied by baseline risk category, being 33% in favorable-, 45% in intermediate-, and 11% in poor-risk patients. Median time to response was 2.7 months, median response duration was 8.9 months, and median progression-free survival was 8.3 months.
PD-L1 status was determined in 36 patients; 15 were positive. Among the positive patents, ORR was 60%, compared with 19% in PD-L1 negative patients. Response rates varied with tumor characteristics. Among patients with ccRCC with sarcomatoid differentiation, the ORR was 50% in PD-L1–positive patients and 29% in negative patients. In patients with variant histology RCC, the ORR was also better in PD-L1 positive patients (67% vs. 14%).
The most common treatment-related side effects were fatigue (35%), proteinuria (35%), musculoskeletal pain (33%), diarrhea (22%), rash (20%), hypertension (18%), pruritus (18%), thyroid dysfunction (17%), hepatitis (15%), fever (13%), and mucositis (12%). Thirty-four patients developed at least one grade 3 adverse event; there were no grade 4 or 5 toxicities. One patient died, presumably because of disease progression.
Quality of life scores were largely stable during treatment.
“The combination demonstrated responses across several subtypes of RCC, including collecting duct and medullary carcinoma, histologies that are often treated with cytotoxic chemotherapy,” the authors said. “This is notable given the generally poor prognosis and low response rate associated with variant histology RCC in trials to date.”
The study also suggests the PD-L1 status might be “intriguing as a biomarker for response to atezolizumab and bevacizumab in variant histology RCC. We plan to conduct additional correlative work, including genomic profiling and assessment of the immune microenvironment, to better elucidate markers of response and resistance,” the authors wrote.
SOURCE: McGregor BA et al. J Clin Oncol. 2019 Nov 13. doi: 10.1200/JCO.19.01882.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Depression linked to persistent opioid use after hysterectomy
VANCOUVER –
Women with depression had an 8% increased risk of perioperative opioid use but a 43% increased risk of persistent use, defined as at least one perioperative prescription followed by at least one prescription 90 days or longer after surgery.
Opioid prescriptions after surgery have been on the rise in recent years, and this has led to a focus on how chronic pain disorders are managed. But studies have shown that patients undergoing general surgery, both minor and major, are at increased risk of persistent opioid use, even after a single surgery, according to Erin Carey, MD, director of the division of minimally invasive gynecologic surgery at the University of North Carolina at Chapel Hill, who presented the research at the meeting sponsored by AAGL.
“We also know that preoperative depression has been linked to adverse outcomes after hysterectomy, both acute postoperative pain in the first 2 days after surgery, and increasing the risk of chronic postoperative pain,” Dr. Carey said.
That prompted her and her team to look at whether preoperative depression might influence the risk of new persistent opioid use after hysterectomy. They analyzed data from the IBM Watson/Truven Health Analytics MarketScan database of claims-based data, which collects information from a variety of sources, including electronic medical records and workplace records such as absences, disability, and long-term disability.
“So it does allow for long-term tracking, which makes it optimal for this type of study,” said Dr. Carey.
The study included 382,078 hysterectomies performed between 2001 and 2015 on women who had continuous prescription plans 180 days before to 180 days after the procedure, excluding anyone who had an opioid prescription in the previous 180 days; 60% of the procedures were minimally invasive. About 20% of women were considered to have depression before the procedure, based on a diagnosis (55%), an antidepressant prescription (22%), or both (23%).
There were some differences at baseline between the two populations: Women with preoperative depression were more likely to have a comorbid pain disorder, compared with patients without depression (20% vs. 14%), another psychiatric disorder (2% vs. less than 1%), and a Charlson comorbidity (12% vs. 9%). They also were less likely to undergo a minimally invasive procedure than women without depression (66% vs. 79%). There was an increase in the prevalence of depression over time, from 16% to 23%.
Overall, 74% of women were prescribed an opioid during the perioperative period; 17% were filled before the hysterectomy was performed. Preoperative fills also increased over time, from 4% in 2001 to 21% in 2015.
Women with preoperative depression were at a slightly greater risk for perioperative opioid use (risk ratio, 1.08), but a greater risk for persistent postoperative opioid use (11% vs. 8%; RR, 1.43). The heightened risk for opioid use was similar whether the surgery was performed on an outpatient or inpatient basis.
The presence of other comorbidities in women with diagnosed depression or prescribed antidepressants complicates the findings, according to Dr. Carey. “There may be additional chronic pain factors that are confounding this data, but it is consistent with other data that de novo postoperative opioid dependence may be a higher risk for these patients, so it’s important for us to look at that critically.”
Dr. Carey has been a consultant for Teleflex Medical and a speaker for Med-IQ.
VANCOUVER –
Women with depression had an 8% increased risk of perioperative opioid use but a 43% increased risk of persistent use, defined as at least one perioperative prescription followed by at least one prescription 90 days or longer after surgery.
Opioid prescriptions after surgery have been on the rise in recent years, and this has led to a focus on how chronic pain disorders are managed. But studies have shown that patients undergoing general surgery, both minor and major, are at increased risk of persistent opioid use, even after a single surgery, according to Erin Carey, MD, director of the division of minimally invasive gynecologic surgery at the University of North Carolina at Chapel Hill, who presented the research at the meeting sponsored by AAGL.
“We also know that preoperative depression has been linked to adverse outcomes after hysterectomy, both acute postoperative pain in the first 2 days after surgery, and increasing the risk of chronic postoperative pain,” Dr. Carey said.
That prompted her and her team to look at whether preoperative depression might influence the risk of new persistent opioid use after hysterectomy. They analyzed data from the IBM Watson/Truven Health Analytics MarketScan database of claims-based data, which collects information from a variety of sources, including electronic medical records and workplace records such as absences, disability, and long-term disability.
“So it does allow for long-term tracking, which makes it optimal for this type of study,” said Dr. Carey.
The study included 382,078 hysterectomies performed between 2001 and 2015 on women who had continuous prescription plans 180 days before to 180 days after the procedure, excluding anyone who had an opioid prescription in the previous 180 days; 60% of the procedures were minimally invasive. About 20% of women were considered to have depression before the procedure, based on a diagnosis (55%), an antidepressant prescription (22%), or both (23%).
There were some differences at baseline between the two populations: Women with preoperative depression were more likely to have a comorbid pain disorder, compared with patients without depression (20% vs. 14%), another psychiatric disorder (2% vs. less than 1%), and a Charlson comorbidity (12% vs. 9%). They also were less likely to undergo a minimally invasive procedure than women without depression (66% vs. 79%). There was an increase in the prevalence of depression over time, from 16% to 23%.
Overall, 74% of women were prescribed an opioid during the perioperative period; 17% were filled before the hysterectomy was performed. Preoperative fills also increased over time, from 4% in 2001 to 21% in 2015.
Women with preoperative depression were at a slightly greater risk for perioperative opioid use (risk ratio, 1.08), but a greater risk for persistent postoperative opioid use (11% vs. 8%; RR, 1.43). The heightened risk for opioid use was similar whether the surgery was performed on an outpatient or inpatient basis.
The presence of other comorbidities in women with diagnosed depression or prescribed antidepressants complicates the findings, according to Dr. Carey. “There may be additional chronic pain factors that are confounding this data, but it is consistent with other data that de novo postoperative opioid dependence may be a higher risk for these patients, so it’s important for us to look at that critically.”
Dr. Carey has been a consultant for Teleflex Medical and a speaker for Med-IQ.
VANCOUVER –
Women with depression had an 8% increased risk of perioperative opioid use but a 43% increased risk of persistent use, defined as at least one perioperative prescription followed by at least one prescription 90 days or longer after surgery.
Opioid prescriptions after surgery have been on the rise in recent years, and this has led to a focus on how chronic pain disorders are managed. But studies have shown that patients undergoing general surgery, both minor and major, are at increased risk of persistent opioid use, even after a single surgery, according to Erin Carey, MD, director of the division of minimally invasive gynecologic surgery at the University of North Carolina at Chapel Hill, who presented the research at the meeting sponsored by AAGL.
“We also know that preoperative depression has been linked to adverse outcomes after hysterectomy, both acute postoperative pain in the first 2 days after surgery, and increasing the risk of chronic postoperative pain,” Dr. Carey said.
That prompted her and her team to look at whether preoperative depression might influence the risk of new persistent opioid use after hysterectomy. They analyzed data from the IBM Watson/Truven Health Analytics MarketScan database of claims-based data, which collects information from a variety of sources, including electronic medical records and workplace records such as absences, disability, and long-term disability.
“So it does allow for long-term tracking, which makes it optimal for this type of study,” said Dr. Carey.
The study included 382,078 hysterectomies performed between 2001 and 2015 on women who had continuous prescription plans 180 days before to 180 days after the procedure, excluding anyone who had an opioid prescription in the previous 180 days; 60% of the procedures were minimally invasive. About 20% of women were considered to have depression before the procedure, based on a diagnosis (55%), an antidepressant prescription (22%), or both (23%).
There were some differences at baseline between the two populations: Women with preoperative depression were more likely to have a comorbid pain disorder, compared with patients without depression (20% vs. 14%), another psychiatric disorder (2% vs. less than 1%), and a Charlson comorbidity (12% vs. 9%). They also were less likely to undergo a minimally invasive procedure than women without depression (66% vs. 79%). There was an increase in the prevalence of depression over time, from 16% to 23%.
Overall, 74% of women were prescribed an opioid during the perioperative period; 17% were filled before the hysterectomy was performed. Preoperative fills also increased over time, from 4% in 2001 to 21% in 2015.
Women with preoperative depression were at a slightly greater risk for perioperative opioid use (risk ratio, 1.08), but a greater risk for persistent postoperative opioid use (11% vs. 8%; RR, 1.43). The heightened risk for opioid use was similar whether the surgery was performed on an outpatient or inpatient basis.
The presence of other comorbidities in women with diagnosed depression or prescribed antidepressants complicates the findings, according to Dr. Carey. “There may be additional chronic pain factors that are confounding this data, but it is consistent with other data that de novo postoperative opioid dependence may be a higher risk for these patients, so it’s important for us to look at that critically.”
Dr. Carey has been a consultant for Teleflex Medical and a speaker for Med-IQ.
REPORTING FROM THE AAGL GLOBAL CONGRESS
Frontline ibrutinib saves money over chemoimmunotherapy
Ibrutinib monotherapy was associated with lower total health care costs compared with chemoimmunotherapy in the frontline treatment of patients with chronic lymphocytic leukemia (CLL), according to a retrospective study.
“This study compared time to next treatment, health care resource utilization, and total direct costs among patients with CLL initiating front-line ibrutinib single agent or chemoimmunotherapy,” wrote Bruno Emond, of Analysis Group, Montreal, and colleagues. Their report is in Clinical Lymphoma, Myeloma & Leukemia.
The researchers retrospectively analyzed data from 1,161 patients with CLL who were started on ibrutinib monotherapy or chemoimmunotherapy from 2014 to 2017. Data were collected from the Optum Clinformatics Extended DataMart De-Identified Databases.
Between the two groups, differences in baseline characteristics were controlled for by way of inverse probability of treatment weighting. Two treatment periods were included in the study: the initial 6 months of treatment and entire duration of frontline therapy.
The team also conducted a subgroup analysis of patients treated with bendamustine and rituximab. This cohort was analyzed independently since the regimen is commonly used in clinical practice.
After analysis, the researchers found that ibrutinib monotherapy was associated with net monthly cost savings of $3,766 (P less than .0001), compared with chemoimmunotherapy and bendamustine/rituximab over the frontline therapy period.
Ibrutinib patients had fewer monthly days with outpatient services (rate ratio, 0.75; 95% confidence interval, 0.60-0.94; P = .0200), compared with those on chemoimmunotherapy; and were less likely to initiate a next line of treatment, compared with chemoimmunotherapy patients (hazard ratio, 0.54; 95% CI, 0.33-0.90; P = .0163).
“Cost savings and reductions in health care resource utilization were even more pronounced when considering only the first 6 months of front-line treatment,” the researchers wrote.
The researchers acknowledged that two key limitations of the study were the potential influence of unobserved confounding factors and the use of claims data, which could include errors and omissions.
“These results suggest that ibrutinib single-agent is associated with lower total costs driven by lower medical costs, despite higher pharmacy costs, compared with chemoimmunotherapy and bendamustine/rituximab,” they concluded.
The authors reported financial affiliations with Janssen Scientific Affairs, which funded the study, and other companies.
SOURCE: Emond B et al. Clin Lymphoma Myeloma Leuk. 2019 Aug 26. doi: 10.1016/j.clml.2019.08.004.
Ibrutinib monotherapy was associated with lower total health care costs compared with chemoimmunotherapy in the frontline treatment of patients with chronic lymphocytic leukemia (CLL), according to a retrospective study.
“This study compared time to next treatment, health care resource utilization, and total direct costs among patients with CLL initiating front-line ibrutinib single agent or chemoimmunotherapy,” wrote Bruno Emond, of Analysis Group, Montreal, and colleagues. Their report is in Clinical Lymphoma, Myeloma & Leukemia.
The researchers retrospectively analyzed data from 1,161 patients with CLL who were started on ibrutinib monotherapy or chemoimmunotherapy from 2014 to 2017. Data were collected from the Optum Clinformatics Extended DataMart De-Identified Databases.
Between the two groups, differences in baseline characteristics were controlled for by way of inverse probability of treatment weighting. Two treatment periods were included in the study: the initial 6 months of treatment and entire duration of frontline therapy.
The team also conducted a subgroup analysis of patients treated with bendamustine and rituximab. This cohort was analyzed independently since the regimen is commonly used in clinical practice.
After analysis, the researchers found that ibrutinib monotherapy was associated with net monthly cost savings of $3,766 (P less than .0001), compared with chemoimmunotherapy and bendamustine/rituximab over the frontline therapy period.
Ibrutinib patients had fewer monthly days with outpatient services (rate ratio, 0.75; 95% confidence interval, 0.60-0.94; P = .0200), compared with those on chemoimmunotherapy; and were less likely to initiate a next line of treatment, compared with chemoimmunotherapy patients (hazard ratio, 0.54; 95% CI, 0.33-0.90; P = .0163).
“Cost savings and reductions in health care resource utilization were even more pronounced when considering only the first 6 months of front-line treatment,” the researchers wrote.
The researchers acknowledged that two key limitations of the study were the potential influence of unobserved confounding factors and the use of claims data, which could include errors and omissions.
“These results suggest that ibrutinib single-agent is associated with lower total costs driven by lower medical costs, despite higher pharmacy costs, compared with chemoimmunotherapy and bendamustine/rituximab,” they concluded.
The authors reported financial affiliations with Janssen Scientific Affairs, which funded the study, and other companies.
SOURCE: Emond B et al. Clin Lymphoma Myeloma Leuk. 2019 Aug 26. doi: 10.1016/j.clml.2019.08.004.
Ibrutinib monotherapy was associated with lower total health care costs compared with chemoimmunotherapy in the frontline treatment of patients with chronic lymphocytic leukemia (CLL), according to a retrospective study.
“This study compared time to next treatment, health care resource utilization, and total direct costs among patients with CLL initiating front-line ibrutinib single agent or chemoimmunotherapy,” wrote Bruno Emond, of Analysis Group, Montreal, and colleagues. Their report is in Clinical Lymphoma, Myeloma & Leukemia.
The researchers retrospectively analyzed data from 1,161 patients with CLL who were started on ibrutinib monotherapy or chemoimmunotherapy from 2014 to 2017. Data were collected from the Optum Clinformatics Extended DataMart De-Identified Databases.
Between the two groups, differences in baseline characteristics were controlled for by way of inverse probability of treatment weighting. Two treatment periods were included in the study: the initial 6 months of treatment and entire duration of frontline therapy.
The team also conducted a subgroup analysis of patients treated with bendamustine and rituximab. This cohort was analyzed independently since the regimen is commonly used in clinical practice.
After analysis, the researchers found that ibrutinib monotherapy was associated with net monthly cost savings of $3,766 (P less than .0001), compared with chemoimmunotherapy and bendamustine/rituximab over the frontline therapy period.
Ibrutinib patients had fewer monthly days with outpatient services (rate ratio, 0.75; 95% confidence interval, 0.60-0.94; P = .0200), compared with those on chemoimmunotherapy; and were less likely to initiate a next line of treatment, compared with chemoimmunotherapy patients (hazard ratio, 0.54; 95% CI, 0.33-0.90; P = .0163).
“Cost savings and reductions in health care resource utilization were even more pronounced when considering only the first 6 months of front-line treatment,” the researchers wrote.
The researchers acknowledged that two key limitations of the study were the potential influence of unobserved confounding factors and the use of claims data, which could include errors and omissions.
“These results suggest that ibrutinib single-agent is associated with lower total costs driven by lower medical costs, despite higher pharmacy costs, compared with chemoimmunotherapy and bendamustine/rituximab,” they concluded.
The authors reported financial affiliations with Janssen Scientific Affairs, which funded the study, and other companies.
SOURCE: Emond B et al. Clin Lymphoma Myeloma Leuk. 2019 Aug 26. doi: 10.1016/j.clml.2019.08.004.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA
Early lenalidomide may delay progression of smoldering myeloma
Early treatment with lenalidomide may delay disease progression and prevent end-organ damage in patients with high-risk smoldering multiple myeloma (SMM), according to findings from a phase 3 trial.
While observation is the current standard of care in SMM, early therapy may represent a new standard for patients with high-risk disease, explained Sagar Lonial, MD, of Winship Cancer Institute, Emory University, Atlanta, and colleagues. Their findings were published in the Journal of Clinical Oncology.
The randomized, open-label, phase 3 study included 182 patients with intermediate- or high-risk SMM. Study patients were randomly allocated to receive either oral lenalidomide at 25 mg daily on days 1-21 of a 28-day cycle or observation.
Study subjects were stratified based on time since SMM diagnosis – 1 year or less vs. more than 1 year, and all patients in the lenalidomide arm received aspirin at 325 mg on days 1-28. Both interventions were maintained until unacceptable toxicity, disease progression, or withdrawal for other reasons.
The primary outcome was progression-free survival (PFS), measured from baseline to the development of symptomatic multiple myeloma (MM). The criteria for progression included evidence of end-organ damage in relation to MM and biochemical disease progression.
The researchers found that at 1 year PFS was 98% in the lenalidomide group and 89% in the observation group. At 2 years, PFS was 93% in the lenalidomide group and 76% in the observation group. PFS was 91% in the lenalidomide group and 66% in the observation group at 3 years (hazard ratio, 0.28; P = .002).
Among lenalidomide-treated patients, grade 3 or 4 hematologic and nonhematologic adverse events occurred in 36 patients (41%). Nonhematologic adverse events occurred in 25 patients (28%).
Frequent AEs among lenalidomide-treated patients included grade 4 decreased neutrophil count (4.5%), as well as grade 3 infections (20.5%), hypertension (9.1%), fatigue (6.8%), skin problems (5.7%), dyspnea (5.7%), and hypokalemia (3.4%). “In most cases, [adverse events] could be managed with dose modifications,” they wrote.
To reduce long-term toxicity, the researchers recommended a 2-year duration of therapy for patients at highest risk.
“Our results support the use of early intervention in patients with high-risk SMM – as defined by the 20/2/20 criteria where our magnitude of benefit was the greatest – rather than continued observation,” the researchers wrote.
The trial was funded by the National Cancer Institute. The authors reported financial affiliations with AbbVie, Aduro Biotech, Amgen, Bristol-Myers Squibb, Celgene, Juno Therapeutics, Kite Pharma, Sanofi, Takeda, and several other companies.
SOURCE: Lonial S et al. J Clin Oncol. 2019 Oct 25. doi: 10.1200/JCO.19.01740.
Early treatment with lenalidomide may delay disease progression and prevent end-organ damage in patients with high-risk smoldering multiple myeloma (SMM), according to findings from a phase 3 trial.
While observation is the current standard of care in SMM, early therapy may represent a new standard for patients with high-risk disease, explained Sagar Lonial, MD, of Winship Cancer Institute, Emory University, Atlanta, and colleagues. Their findings were published in the Journal of Clinical Oncology.
The randomized, open-label, phase 3 study included 182 patients with intermediate- or high-risk SMM. Study patients were randomly allocated to receive either oral lenalidomide at 25 mg daily on days 1-21 of a 28-day cycle or observation.
Study subjects were stratified based on time since SMM diagnosis – 1 year or less vs. more than 1 year, and all patients in the lenalidomide arm received aspirin at 325 mg on days 1-28. Both interventions were maintained until unacceptable toxicity, disease progression, or withdrawal for other reasons.
The primary outcome was progression-free survival (PFS), measured from baseline to the development of symptomatic multiple myeloma (MM). The criteria for progression included evidence of end-organ damage in relation to MM and biochemical disease progression.
The researchers found that at 1 year PFS was 98% in the lenalidomide group and 89% in the observation group. At 2 years, PFS was 93% in the lenalidomide group and 76% in the observation group. PFS was 91% in the lenalidomide group and 66% in the observation group at 3 years (hazard ratio, 0.28; P = .002).
Among lenalidomide-treated patients, grade 3 or 4 hematologic and nonhematologic adverse events occurred in 36 patients (41%). Nonhematologic adverse events occurred in 25 patients (28%).
Frequent AEs among lenalidomide-treated patients included grade 4 decreased neutrophil count (4.5%), as well as grade 3 infections (20.5%), hypertension (9.1%), fatigue (6.8%), skin problems (5.7%), dyspnea (5.7%), and hypokalemia (3.4%). “In most cases, [adverse events] could be managed with dose modifications,” they wrote.
To reduce long-term toxicity, the researchers recommended a 2-year duration of therapy for patients at highest risk.
“Our results support the use of early intervention in patients with high-risk SMM – as defined by the 20/2/20 criteria where our magnitude of benefit was the greatest – rather than continued observation,” the researchers wrote.
The trial was funded by the National Cancer Institute. The authors reported financial affiliations with AbbVie, Aduro Biotech, Amgen, Bristol-Myers Squibb, Celgene, Juno Therapeutics, Kite Pharma, Sanofi, Takeda, and several other companies.
SOURCE: Lonial S et al. J Clin Oncol. 2019 Oct 25. doi: 10.1200/JCO.19.01740.
Early treatment with lenalidomide may delay disease progression and prevent end-organ damage in patients with high-risk smoldering multiple myeloma (SMM), according to findings from a phase 3 trial.
While observation is the current standard of care in SMM, early therapy may represent a new standard for patients with high-risk disease, explained Sagar Lonial, MD, of Winship Cancer Institute, Emory University, Atlanta, and colleagues. Their findings were published in the Journal of Clinical Oncology.
The randomized, open-label, phase 3 study included 182 patients with intermediate- or high-risk SMM. Study patients were randomly allocated to receive either oral lenalidomide at 25 mg daily on days 1-21 of a 28-day cycle or observation.
Study subjects were stratified based on time since SMM diagnosis – 1 year or less vs. more than 1 year, and all patients in the lenalidomide arm received aspirin at 325 mg on days 1-28. Both interventions were maintained until unacceptable toxicity, disease progression, or withdrawal for other reasons.
The primary outcome was progression-free survival (PFS), measured from baseline to the development of symptomatic multiple myeloma (MM). The criteria for progression included evidence of end-organ damage in relation to MM and biochemical disease progression.
The researchers found that at 1 year PFS was 98% in the lenalidomide group and 89% in the observation group. At 2 years, PFS was 93% in the lenalidomide group and 76% in the observation group. PFS was 91% in the lenalidomide group and 66% in the observation group at 3 years (hazard ratio, 0.28; P = .002).
Among lenalidomide-treated patients, grade 3 or 4 hematologic and nonhematologic adverse events occurred in 36 patients (41%). Nonhematologic adverse events occurred in 25 patients (28%).
Frequent AEs among lenalidomide-treated patients included grade 4 decreased neutrophil count (4.5%), as well as grade 3 infections (20.5%), hypertension (9.1%), fatigue (6.8%), skin problems (5.7%), dyspnea (5.7%), and hypokalemia (3.4%). “In most cases, [adverse events] could be managed with dose modifications,” they wrote.
To reduce long-term toxicity, the researchers recommended a 2-year duration of therapy for patients at highest risk.
“Our results support the use of early intervention in patients with high-risk SMM – as defined by the 20/2/20 criteria where our magnitude of benefit was the greatest – rather than continued observation,” the researchers wrote.
The trial was funded by the National Cancer Institute. The authors reported financial affiliations with AbbVie, Aduro Biotech, Amgen, Bristol-Myers Squibb, Celgene, Juno Therapeutics, Kite Pharma, Sanofi, Takeda, and several other companies.
SOURCE: Lonial S et al. J Clin Oncol. 2019 Oct 25. doi: 10.1200/JCO.19.01740.
FROM JOURNAL OF CLINICAL ONCOLOGY
Will TP53-mutated AML respond to immunotherapy?
NATIONAL HARBOR, MD. – New research has shown increased immune infiltration in patients with TP53-mutated acute myeloid leukemia (AML).
Patients with TP53-mutated AML had higher levels of T-cell infiltration, immune checkpoint molecules, and interferon (IFN)–gamma signaling than patients with wild-type TP53.
These findings may indicate that patients with TP53-mutated AML will respond to T-cell targeting immunotherapies, but more investigation is needed, according to Sergio Rutella, MD, PhD, of Nottingham (England) Trent University.
Dr. Rutella described the findings at the annual meeting of the Society for Immunotherapy of Cancer.
He and his colleagues recently identified subgroups of AML, called “immune infiltrated” and “immune depleted,” that can predict chemotherapy resistance and response to flotetuzumab (ASH 2019, Abstract 460). However, the team has not determined the genetic drivers of immune infiltration in AML.*
With the current study, Dr. Rutella and his colleagues wanted to determine if TP53 mutations are associated with the AML immune milieu and see if TP53-mutated patients might benefit from immunotherapy.
Discovery cohort
The researchers first analyzed 147 patients with non-promyelocytic AML from the Cancer Genome Atlas. In total, 9% of these patients (n = 13) had TP53-mutated AML. The researchers assessed how 45 immune gene and biological activity signatures correlated with prognostic molecular lesions (TP53 mutations, FLT3-ITD, etc.) and clinical outcomes in this cohort.
The data showed that immune subtypes were associated with overall survival (OS). The median OS was 11.8 months in patients with immune-infiltrated AML, 16.4 months in patients with intermediate AML, and 25.8 months in patients with immune-depleted AML.
The inflammatory chemokine score (P = .011), IDO1 score (P = .027), IFN-gamma score (P = .036), and B7H3 score (P = .045) were all significantly associated with OS. In fact, these factors were all better predictors of OS than cytogenetic risk score (P = .049).
The IFN-gamma score, inflammatory chemokine score, and lymphoid score were all significantly higher in TP53-mutated patients than in patients with RUNX1 mutations, NPM1 mutations, FLT3-ITD (with or without NPM1 mutations), and TET2/DNMT3A/ASXL1 mutations (P values ranging from less than .0001 to .05).
Likewise, the tumor inflammation signature score was significantly higher among TP53-mutated patients than among patients with NPM1 mutations, FLT3-ITD (with or without NPM1 mutations), and TET2/DNMT3A/ASXL1 mutations (P values ranging from less than .0001 to .01).
Validation cohort and bone marrow samples
The researchers also looked at data from a validation cohort, which consisted of 140 patients with non-promyelocytic AML in the Beat AML Master Trial. Twelve percent of these patients (n = 17) had TP53 mutations.
Data in this cohort showed that CD3G messenger RNA (mRNA) was significantly higher in TP53-mutated AML than in TP53-wild-type AML (P = .04). The same was true for CD8A mRNA (P = .0002) and GZMB mRNA (P = .0005).
Likewise, IFN-gamma mRNA (P = .0052), IFIT2 mRNA (P = .0064), and IFIT3 mRNA (P = .003) were all significantly higher in patients with TP53-mutated AML.
Lastly, the researchers analyzed gene expression profiles of bone marrow samples from patients with AML, 36 with mutated TP53 and 24 with wild-type TP53.
The team found that IFN-gamma–induced genes (IFNG and IRF1), markers of T-cell infiltration (CD8A and CD3G) and senescence (EOMES, KLRD1, and HRAS), immune checkpoint molecules (IDO1, LAG3, PDL1, and VISTA), effector function molecules (GZMB, GZMK, and GZMM), and proinflammatory cytokines (IL17A and TNF) were all significantly overexpressed in TP53-mutated AML.
Among the top overexpressed genes in TP53-mutated AML were genes associated with IFN signaling and inflammation pathways – IL-33, IL-6, IFN-gamma, OASL, RIPK2, TNFAIP3, CSF1, and PTGER4. The IL-17 and TNF signaling pathways were the most enriched pathways in TP53-mutated AML.
“Our analysis of primary bone marrow samples showed that TP53-mutated samples are enriched in IL-17, TNF, and IFN signaling molecules, and show higher levels of T-cell infiltrations and immune checkpoints relative to their wild-type counterparts,” Dr. Rutella said.
“The in silico analysis indicated that TP53-mutated cases will show higher levels of T-cell infiltration, immune checkpoints, and IFN-gamma signaling, compared with AML subgroups without risk-defining molecular lesions,” he added. “This is speculative. Whether TP53-mutated AML can be amenable to respond to T-cell targeting immunotherapies is still to be determined.”
Dr. Rutella reported research support from NanoString Technologies, MacroGenics, and Kura Oncology.
SOURCE: Rutella S et al. SITC 2019. Abstract O3.
*This article was updated on 11/19/2019.
NATIONAL HARBOR, MD. – New research has shown increased immune infiltration in patients with TP53-mutated acute myeloid leukemia (AML).
Patients with TP53-mutated AML had higher levels of T-cell infiltration, immune checkpoint molecules, and interferon (IFN)–gamma signaling than patients with wild-type TP53.
These findings may indicate that patients with TP53-mutated AML will respond to T-cell targeting immunotherapies, but more investigation is needed, according to Sergio Rutella, MD, PhD, of Nottingham (England) Trent University.
Dr. Rutella described the findings at the annual meeting of the Society for Immunotherapy of Cancer.
He and his colleagues recently identified subgroups of AML, called “immune infiltrated” and “immune depleted,” that can predict chemotherapy resistance and response to flotetuzumab (ASH 2019, Abstract 460). However, the team has not determined the genetic drivers of immune infiltration in AML.*
With the current study, Dr. Rutella and his colleagues wanted to determine if TP53 mutations are associated with the AML immune milieu and see if TP53-mutated patients might benefit from immunotherapy.
Discovery cohort
The researchers first analyzed 147 patients with non-promyelocytic AML from the Cancer Genome Atlas. In total, 9% of these patients (n = 13) had TP53-mutated AML. The researchers assessed how 45 immune gene and biological activity signatures correlated with prognostic molecular lesions (TP53 mutations, FLT3-ITD, etc.) and clinical outcomes in this cohort.
The data showed that immune subtypes were associated with overall survival (OS). The median OS was 11.8 months in patients with immune-infiltrated AML, 16.4 months in patients with intermediate AML, and 25.8 months in patients with immune-depleted AML.
The inflammatory chemokine score (P = .011), IDO1 score (P = .027), IFN-gamma score (P = .036), and B7H3 score (P = .045) were all significantly associated with OS. In fact, these factors were all better predictors of OS than cytogenetic risk score (P = .049).
The IFN-gamma score, inflammatory chemokine score, and lymphoid score were all significantly higher in TP53-mutated patients than in patients with RUNX1 mutations, NPM1 mutations, FLT3-ITD (with or without NPM1 mutations), and TET2/DNMT3A/ASXL1 mutations (P values ranging from less than .0001 to .05).
Likewise, the tumor inflammation signature score was significantly higher among TP53-mutated patients than among patients with NPM1 mutations, FLT3-ITD (with or without NPM1 mutations), and TET2/DNMT3A/ASXL1 mutations (P values ranging from less than .0001 to .01).
Validation cohort and bone marrow samples
The researchers also looked at data from a validation cohort, which consisted of 140 patients with non-promyelocytic AML in the Beat AML Master Trial. Twelve percent of these patients (n = 17) had TP53 mutations.
Data in this cohort showed that CD3G messenger RNA (mRNA) was significantly higher in TP53-mutated AML than in TP53-wild-type AML (P = .04). The same was true for CD8A mRNA (P = .0002) and GZMB mRNA (P = .0005).
Likewise, IFN-gamma mRNA (P = .0052), IFIT2 mRNA (P = .0064), and IFIT3 mRNA (P = .003) were all significantly higher in patients with TP53-mutated AML.
Lastly, the researchers analyzed gene expression profiles of bone marrow samples from patients with AML, 36 with mutated TP53 and 24 with wild-type TP53.
The team found that IFN-gamma–induced genes (IFNG and IRF1), markers of T-cell infiltration (CD8A and CD3G) and senescence (EOMES, KLRD1, and HRAS), immune checkpoint molecules (IDO1, LAG3, PDL1, and VISTA), effector function molecules (GZMB, GZMK, and GZMM), and proinflammatory cytokines (IL17A and TNF) were all significantly overexpressed in TP53-mutated AML.
Among the top overexpressed genes in TP53-mutated AML were genes associated with IFN signaling and inflammation pathways – IL-33, IL-6, IFN-gamma, OASL, RIPK2, TNFAIP3, CSF1, and PTGER4. The IL-17 and TNF signaling pathways were the most enriched pathways in TP53-mutated AML.
“Our analysis of primary bone marrow samples showed that TP53-mutated samples are enriched in IL-17, TNF, and IFN signaling molecules, and show higher levels of T-cell infiltrations and immune checkpoints relative to their wild-type counterparts,” Dr. Rutella said.
“The in silico analysis indicated that TP53-mutated cases will show higher levels of T-cell infiltration, immune checkpoints, and IFN-gamma signaling, compared with AML subgroups without risk-defining molecular lesions,” he added. “This is speculative. Whether TP53-mutated AML can be amenable to respond to T-cell targeting immunotherapies is still to be determined.”
Dr. Rutella reported research support from NanoString Technologies, MacroGenics, and Kura Oncology.
SOURCE: Rutella S et al. SITC 2019. Abstract O3.
*This article was updated on 11/19/2019.
NATIONAL HARBOR, MD. – New research has shown increased immune infiltration in patients with TP53-mutated acute myeloid leukemia (AML).
Patients with TP53-mutated AML had higher levels of T-cell infiltration, immune checkpoint molecules, and interferon (IFN)–gamma signaling than patients with wild-type TP53.
These findings may indicate that patients with TP53-mutated AML will respond to T-cell targeting immunotherapies, but more investigation is needed, according to Sergio Rutella, MD, PhD, of Nottingham (England) Trent University.
Dr. Rutella described the findings at the annual meeting of the Society for Immunotherapy of Cancer.
He and his colleagues recently identified subgroups of AML, called “immune infiltrated” and “immune depleted,” that can predict chemotherapy resistance and response to flotetuzumab (ASH 2019, Abstract 460). However, the team has not determined the genetic drivers of immune infiltration in AML.*
With the current study, Dr. Rutella and his colleagues wanted to determine if TP53 mutations are associated with the AML immune milieu and see if TP53-mutated patients might benefit from immunotherapy.
Discovery cohort
The researchers first analyzed 147 patients with non-promyelocytic AML from the Cancer Genome Atlas. In total, 9% of these patients (n = 13) had TP53-mutated AML. The researchers assessed how 45 immune gene and biological activity signatures correlated with prognostic molecular lesions (TP53 mutations, FLT3-ITD, etc.) and clinical outcomes in this cohort.
The data showed that immune subtypes were associated with overall survival (OS). The median OS was 11.8 months in patients with immune-infiltrated AML, 16.4 months in patients with intermediate AML, and 25.8 months in patients with immune-depleted AML.
The inflammatory chemokine score (P = .011), IDO1 score (P = .027), IFN-gamma score (P = .036), and B7H3 score (P = .045) were all significantly associated with OS. In fact, these factors were all better predictors of OS than cytogenetic risk score (P = .049).
The IFN-gamma score, inflammatory chemokine score, and lymphoid score were all significantly higher in TP53-mutated patients than in patients with RUNX1 mutations, NPM1 mutations, FLT3-ITD (with or without NPM1 mutations), and TET2/DNMT3A/ASXL1 mutations (P values ranging from less than .0001 to .05).
Likewise, the tumor inflammation signature score was significantly higher among TP53-mutated patients than among patients with NPM1 mutations, FLT3-ITD (with or without NPM1 mutations), and TET2/DNMT3A/ASXL1 mutations (P values ranging from less than .0001 to .01).
Validation cohort and bone marrow samples
The researchers also looked at data from a validation cohort, which consisted of 140 patients with non-promyelocytic AML in the Beat AML Master Trial. Twelve percent of these patients (n = 17) had TP53 mutations.
Data in this cohort showed that CD3G messenger RNA (mRNA) was significantly higher in TP53-mutated AML than in TP53-wild-type AML (P = .04). The same was true for CD8A mRNA (P = .0002) and GZMB mRNA (P = .0005).
Likewise, IFN-gamma mRNA (P = .0052), IFIT2 mRNA (P = .0064), and IFIT3 mRNA (P = .003) were all significantly higher in patients with TP53-mutated AML.
Lastly, the researchers analyzed gene expression profiles of bone marrow samples from patients with AML, 36 with mutated TP53 and 24 with wild-type TP53.
The team found that IFN-gamma–induced genes (IFNG and IRF1), markers of T-cell infiltration (CD8A and CD3G) and senescence (EOMES, KLRD1, and HRAS), immune checkpoint molecules (IDO1, LAG3, PDL1, and VISTA), effector function molecules (GZMB, GZMK, and GZMM), and proinflammatory cytokines (IL17A and TNF) were all significantly overexpressed in TP53-mutated AML.
Among the top overexpressed genes in TP53-mutated AML were genes associated with IFN signaling and inflammation pathways – IL-33, IL-6, IFN-gamma, OASL, RIPK2, TNFAIP3, CSF1, and PTGER4. The IL-17 and TNF signaling pathways were the most enriched pathways in TP53-mutated AML.
“Our analysis of primary bone marrow samples showed that TP53-mutated samples are enriched in IL-17, TNF, and IFN signaling molecules, and show higher levels of T-cell infiltrations and immune checkpoints relative to their wild-type counterparts,” Dr. Rutella said.
“The in silico analysis indicated that TP53-mutated cases will show higher levels of T-cell infiltration, immune checkpoints, and IFN-gamma signaling, compared with AML subgroups without risk-defining molecular lesions,” he added. “This is speculative. Whether TP53-mutated AML can be amenable to respond to T-cell targeting immunotherapies is still to be determined.”
Dr. Rutella reported research support from NanoString Technologies, MacroGenics, and Kura Oncology.
SOURCE: Rutella S et al. SITC 2019. Abstract O3.
*This article was updated on 11/19/2019.
REPORTING FROM SITC 2019
CAR T-cell ‘cocktail’ may overcome antigen escape relapse
A chimeric antigen receptor (CAR) T-cell “cocktail” targeting both CD19 and CD22 could improve outcomes for patients with refractory or relapsed B-cell malignancies, according to investigators.
This dual approach, which appeared safe and effective, may be able to overcome antigen escape relapse, reported Na Wang, MD, of Huazhong University of Science and Technology in China, and colleagues.
The investigators tested this method in an open-label, single-arm pilot study involving 89 patients with refractory/relapsed B cell malignancies. Of these, 51 patients had B-cell acute lymphoblastic leukemia (B-ALL), while the remaining 38 had non-Hodgkin lymphoma (NHL). All patients had dual expression of CD19 and CD22 on malignant B cells, good performance status, and “essentially” normal organ function, the investigators reported in Blood.
Following lymphodepletion, patients were infused with CAR19 and CAR22 T cells, then evaluated for responses with imaging or bone marrow aspiration on a monthly basis for 6 months, then every 3 months thereafter.
After 30 days, most patients with ALL (96%) achieved a minimal residual disease-negative complete response or complete response with incomplete count recovery. After a median follow-up of 16.7 months, almost half of these responders relapsed (49%), median progression-free survival was 13.6 months, and overall survival was 31 months.
With a minimum follow-up of 3 months, half of the patients with NHL (50%) achieved complete responses, with the caveat that two patients who died of septic shock and severe cytokine release syndrome were excluded from this efficacy analysis. After a median follow-up of 14.4 months, in the NHL group, median progression-free survival was 9.9 months and overall survival was 18 months.
Across disease types, almost all patients (95.5%) experienced cytokine release syndrome, with more than three-quarters (77.6%) categorized as grade 1 or 2. CAR T cell-related encephalopathy syndrome (CRES) occurred in 13.5% of patients; most were low grade, apart from one case that was grade 4. In total, 12 patients died due to adverse events.
“The severe [adverse events] were mostly cytopenias and the most frequent fatal [adverse event] was lung infection, which was attributable in part to the high disease burden and heavy pretreatment of the enrolled patients,” the investigators wrote. “Nearly all the high-grade CRS and CRES were reversible and occurred in similar incidences as previously reported. Thus, the sequential infusion of CAR19/22 T-cell “cocktail” was an efficient and well-tolerated approach to circumvent antigen loss of CD19 or CD22.”
The investigators reported having no conflicts of interest.
SOURCE: Wang N et al. 2019 Oct 29. doi: 10.1182/blood.2019000017.
A chimeric antigen receptor (CAR) T-cell “cocktail” targeting both CD19 and CD22 could improve outcomes for patients with refractory or relapsed B-cell malignancies, according to investigators.
This dual approach, which appeared safe and effective, may be able to overcome antigen escape relapse, reported Na Wang, MD, of Huazhong University of Science and Technology in China, and colleagues.
The investigators tested this method in an open-label, single-arm pilot study involving 89 patients with refractory/relapsed B cell malignancies. Of these, 51 patients had B-cell acute lymphoblastic leukemia (B-ALL), while the remaining 38 had non-Hodgkin lymphoma (NHL). All patients had dual expression of CD19 and CD22 on malignant B cells, good performance status, and “essentially” normal organ function, the investigators reported in Blood.
Following lymphodepletion, patients were infused with CAR19 and CAR22 T cells, then evaluated for responses with imaging or bone marrow aspiration on a monthly basis for 6 months, then every 3 months thereafter.
After 30 days, most patients with ALL (96%) achieved a minimal residual disease-negative complete response or complete response with incomplete count recovery. After a median follow-up of 16.7 months, almost half of these responders relapsed (49%), median progression-free survival was 13.6 months, and overall survival was 31 months.
With a minimum follow-up of 3 months, half of the patients with NHL (50%) achieved complete responses, with the caveat that two patients who died of septic shock and severe cytokine release syndrome were excluded from this efficacy analysis. After a median follow-up of 14.4 months, in the NHL group, median progression-free survival was 9.9 months and overall survival was 18 months.
Across disease types, almost all patients (95.5%) experienced cytokine release syndrome, with more than three-quarters (77.6%) categorized as grade 1 or 2. CAR T cell-related encephalopathy syndrome (CRES) occurred in 13.5% of patients; most were low grade, apart from one case that was grade 4. In total, 12 patients died due to adverse events.
“The severe [adverse events] were mostly cytopenias and the most frequent fatal [adverse event] was lung infection, which was attributable in part to the high disease burden and heavy pretreatment of the enrolled patients,” the investigators wrote. “Nearly all the high-grade CRS and CRES were reversible and occurred in similar incidences as previously reported. Thus, the sequential infusion of CAR19/22 T-cell “cocktail” was an efficient and well-tolerated approach to circumvent antigen loss of CD19 or CD22.”
The investigators reported having no conflicts of interest.
SOURCE: Wang N et al. 2019 Oct 29. doi: 10.1182/blood.2019000017.
A chimeric antigen receptor (CAR) T-cell “cocktail” targeting both CD19 and CD22 could improve outcomes for patients with refractory or relapsed B-cell malignancies, according to investigators.
This dual approach, which appeared safe and effective, may be able to overcome antigen escape relapse, reported Na Wang, MD, of Huazhong University of Science and Technology in China, and colleagues.
The investigators tested this method in an open-label, single-arm pilot study involving 89 patients with refractory/relapsed B cell malignancies. Of these, 51 patients had B-cell acute lymphoblastic leukemia (B-ALL), while the remaining 38 had non-Hodgkin lymphoma (NHL). All patients had dual expression of CD19 and CD22 on malignant B cells, good performance status, and “essentially” normal organ function, the investigators reported in Blood.
Following lymphodepletion, patients were infused with CAR19 and CAR22 T cells, then evaluated for responses with imaging or bone marrow aspiration on a monthly basis for 6 months, then every 3 months thereafter.
After 30 days, most patients with ALL (96%) achieved a minimal residual disease-negative complete response or complete response with incomplete count recovery. After a median follow-up of 16.7 months, almost half of these responders relapsed (49%), median progression-free survival was 13.6 months, and overall survival was 31 months.
With a minimum follow-up of 3 months, half of the patients with NHL (50%) achieved complete responses, with the caveat that two patients who died of septic shock and severe cytokine release syndrome were excluded from this efficacy analysis. After a median follow-up of 14.4 months, in the NHL group, median progression-free survival was 9.9 months and overall survival was 18 months.
Across disease types, almost all patients (95.5%) experienced cytokine release syndrome, with more than three-quarters (77.6%) categorized as grade 1 or 2. CAR T cell-related encephalopathy syndrome (CRES) occurred in 13.5% of patients; most were low grade, apart from one case that was grade 4. In total, 12 patients died due to adverse events.
“The severe [adverse events] were mostly cytopenias and the most frequent fatal [adverse event] was lung infection, which was attributable in part to the high disease burden and heavy pretreatment of the enrolled patients,” the investigators wrote. “Nearly all the high-grade CRS and CRES were reversible and occurred in similar incidences as previously reported. Thus, the sequential infusion of CAR19/22 T-cell “cocktail” was an efficient and well-tolerated approach to circumvent antigen loss of CD19 or CD22.”
The investigators reported having no conflicts of interest.
SOURCE: Wang N et al. 2019 Oct 29. doi: 10.1182/blood.2019000017.
FROM BLOOD