User login
Ibrutinib-venetoclax found highly active in hard-to-treat CLL
The strategy of simultaneously inhibiting proliferation and reactivating apoptosis can eradicate chronic lymphocytic leukemia (CLL) in a large share of patients, suggest results from the phase 2 CLARITY trial.
“Both ibrutinib and venetoclax are active in CLL with improved survival; however, as monotherapies, both currently are given until disease progression,” wrote Peter Hillmen, MBChB, PhD, St. James’s University Hospital, Leeds, England, and his colleagues.
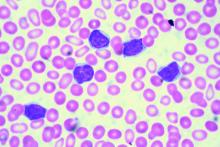
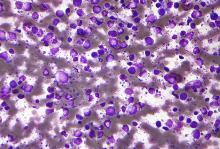
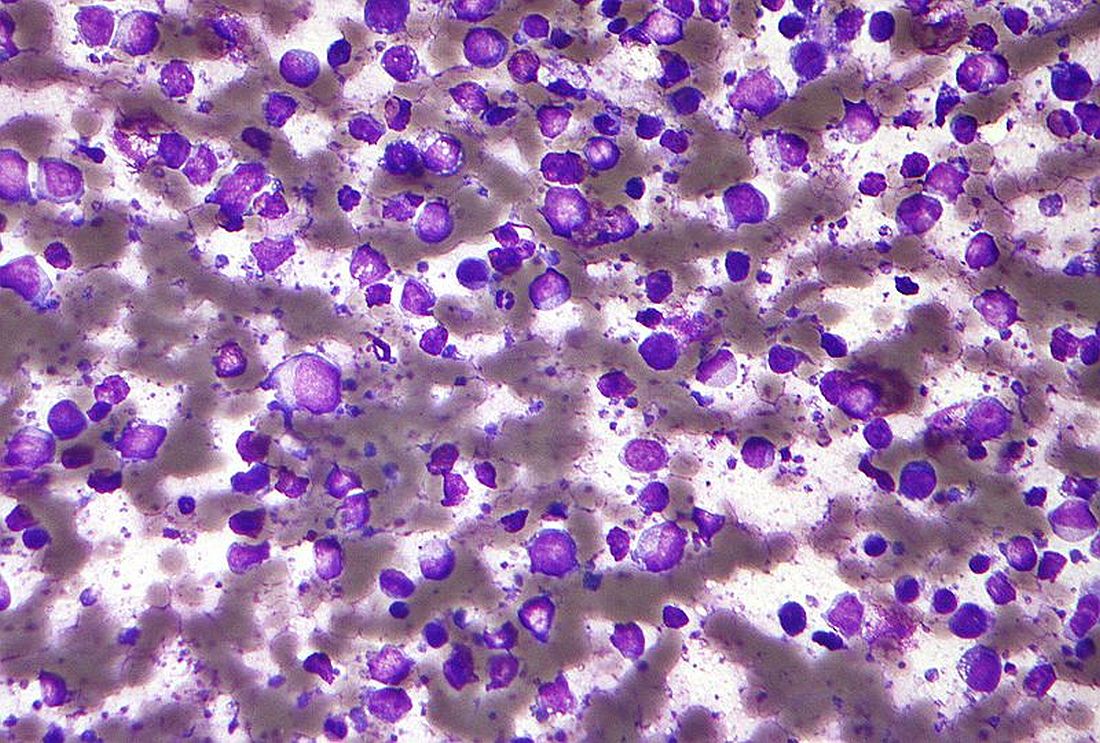
In the single-arm, open-label trial, the investigators treated 53 patients with relapsed or refractory CLL with combination ibrutinib (Imbruvica), a small-molecule inhibitor of Bruton’s tyrosine kinase, and venetoclax (Venclexta), a small molecule inhibitor of the anti-apoptotic protein Bcl-2. The primary endpoint was MRD negativity, defined as presence of fewer than one CLL cell in 10,000 leukocytes, after 12 months of combination therapy.
Results reported in the Journal of Clinical Oncology showed that the combination was highly active, with 53% of patients achieving MRD negativity in the blood and 36% achieving MRD negativity in the marrow.
Most patients, 89%, had a treatment response, and slightly more than half, 51%, achieved a complete remission. With a median 21.1-month follow-up, only a single patient experienced progression and all were still alive.
Adverse effects were generally manageable. Grade 3-4 adverse events of special interest included 34 cases of neutropenia and 1 case of biochemical tumor lysis syndrome that was managed by delaying venetoclax.
“We have demonstrated promising efficacy that indicates potent synergy between ibrutinib and venetoclax for inducing MRD-negative responses with manageable adverse effects,” the investigators wrote. “The observation that a significant proportion of patients experience MRD-negative remission indicates that this combination can be given for a limited period and then stopped after patients achieve a deep remission.”
Whether the combination leads to permanent disease eradication in certain patients is still unclear, the investigators added.
The trial was supported by Bloodwise under the Trials Acceleration Programme, by the National Institute for Health Research Leeds Clinical Research Facility, and by an unrestricted educational grant from Janssen-Cilag and AbbVie. Ibrutinib was provided free of charge by Janssen-Cilag, and venetoclax was provided free of charge by AbbVie. Dr. Hillman reported financial relationships with Janssen, AbbVie, Roche, Pharmacyclics, and Gilead Sciences.
SOURCE: Hillmen P et al. J Clin Oncol. 2019 Jul 11. doi: 10.1200/JCO.19.00894.
The strategy of simultaneously inhibiting proliferation and reactivating apoptosis can eradicate chronic lymphocytic leukemia (CLL) in a large share of patients, suggest results from the phase 2 CLARITY trial.
“Both ibrutinib and venetoclax are active in CLL with improved survival; however, as monotherapies, both currently are given until disease progression,” wrote Peter Hillmen, MBChB, PhD, St. James’s University Hospital, Leeds, England, and his colleagues.
In the single-arm, open-label trial, the investigators treated 53 patients with relapsed or refractory CLL with combination ibrutinib (Imbruvica), a small-molecule inhibitor of Bruton’s tyrosine kinase, and venetoclax (Venclexta), a small molecule inhibitor of the anti-apoptotic protein Bcl-2. The primary endpoint was MRD negativity, defined as presence of fewer than one CLL cell in 10,000 leukocytes, after 12 months of combination therapy.
Results reported in the Journal of Clinical Oncology showed that the combination was highly active, with 53% of patients achieving MRD negativity in the blood and 36% achieving MRD negativity in the marrow.
Most patients, 89%, had a treatment response, and slightly more than half, 51%, achieved a complete remission. With a median 21.1-month follow-up, only a single patient experienced progression and all were still alive.
Adverse effects were generally manageable. Grade 3-4 adverse events of special interest included 34 cases of neutropenia and 1 case of biochemical tumor lysis syndrome that was managed by delaying venetoclax.
“We have demonstrated promising efficacy that indicates potent synergy between ibrutinib and venetoclax for inducing MRD-negative responses with manageable adverse effects,” the investigators wrote. “The observation that a significant proportion of patients experience MRD-negative remission indicates that this combination can be given for a limited period and then stopped after patients achieve a deep remission.”
Whether the combination leads to permanent disease eradication in certain patients is still unclear, the investigators added.
The trial was supported by Bloodwise under the Trials Acceleration Programme, by the National Institute for Health Research Leeds Clinical Research Facility, and by an unrestricted educational grant from Janssen-Cilag and AbbVie. Ibrutinib was provided free of charge by Janssen-Cilag, and venetoclax was provided free of charge by AbbVie. Dr. Hillman reported financial relationships with Janssen, AbbVie, Roche, Pharmacyclics, and Gilead Sciences.
SOURCE: Hillmen P et al. J Clin Oncol. 2019 Jul 11. doi: 10.1200/JCO.19.00894.
The strategy of simultaneously inhibiting proliferation and reactivating apoptosis can eradicate chronic lymphocytic leukemia (CLL) in a large share of patients, suggest results from the phase 2 CLARITY trial.
“Both ibrutinib and venetoclax are active in CLL with improved survival; however, as monotherapies, both currently are given until disease progression,” wrote Peter Hillmen, MBChB, PhD, St. James’s University Hospital, Leeds, England, and his colleagues.
In the single-arm, open-label trial, the investigators treated 53 patients with relapsed or refractory CLL with combination ibrutinib (Imbruvica), a small-molecule inhibitor of Bruton’s tyrosine kinase, and venetoclax (Venclexta), a small molecule inhibitor of the anti-apoptotic protein Bcl-2. The primary endpoint was MRD negativity, defined as presence of fewer than one CLL cell in 10,000 leukocytes, after 12 months of combination therapy.
Results reported in the Journal of Clinical Oncology showed that the combination was highly active, with 53% of patients achieving MRD negativity in the blood and 36% achieving MRD negativity in the marrow.
Most patients, 89%, had a treatment response, and slightly more than half, 51%, achieved a complete remission. With a median 21.1-month follow-up, only a single patient experienced progression and all were still alive.
Adverse effects were generally manageable. Grade 3-4 adverse events of special interest included 34 cases of neutropenia and 1 case of biochemical tumor lysis syndrome that was managed by delaying venetoclax.
“We have demonstrated promising efficacy that indicates potent synergy between ibrutinib and venetoclax for inducing MRD-negative responses with manageable adverse effects,” the investigators wrote. “The observation that a significant proportion of patients experience MRD-negative remission indicates that this combination can be given for a limited period and then stopped after patients achieve a deep remission.”
Whether the combination leads to permanent disease eradication in certain patients is still unclear, the investigators added.
The trial was supported by Bloodwise under the Trials Acceleration Programme, by the National Institute for Health Research Leeds Clinical Research Facility, and by an unrestricted educational grant from Janssen-Cilag and AbbVie. Ibrutinib was provided free of charge by Janssen-Cilag, and venetoclax was provided free of charge by AbbVie. Dr. Hillman reported financial relationships with Janssen, AbbVie, Roche, Pharmacyclics, and Gilead Sciences.
SOURCE: Hillmen P et al. J Clin Oncol. 2019 Jul 11. doi: 10.1200/JCO.19.00894.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Statins crush early seizure risk poststroke
BANGKOK – Statin therapy, even when initiated only upon hospitalization for acute ischemic stroke, was associated with a striking reduction in the risk of early poststroke symptomatic seizure in a large observational study.
Using propensity-score matching to control for potential confounders, use of a statin during acute stroke management was associated with a “robust” 77% reduction in the risk of developing a symptomatic seizure within 7 days after hospital admission, Soichiro Matsubara, MD, reported at the International Epilepsy Congress.
This is an important finding because early symptomatic seizure (ESS) occurs in 2%-7% of patients following an acute ischemic stroke. Moreover, an Italian meta-analysis concluded that ESS was associated with a 4.4-fold increased risk of developing poststroke epilepsy (Epilepsia. 2016 Aug;57[8]:1205-14), noted Dr. Matsubara, a neurologist at the National Cerebral and Cardiovascular Center in Suita, Japan, as well as at Kumamoto (Japan) University.
He presented a study of 2,969 consecutive acute ischemic stroke patients with no history of epilepsy who were admitted to the Japanese comprehensive stroke center, of whom 2.2% experienced ESS. At physician discretion, 19% of the ESS cohort were on a statin during their acute stroke management, as were 55% of the no-ESS group. Four-fifths of patients on a statin initiated the drug only upon hospital admission.
Strokes tended to be more severe in the ESS group, with a median initial National Institutes of Health Stroke Scale score of 12.5, compared with 4 in the seizure-free patients. A cortical stroke lesion was evident upon imaging in 89% of the ESS group and 55% of no-ESS patients. Among ESS patients, 46% had a cardiometabolic stroke, compared with 34% of the no-ESS cohort. Mean C-reactive protein levels and white blood cell counts were significantly higher in the ESS cohort as well. Their median hospital length of stay was 25.5 days, versus 18 days in the no-ESS group, Dr. Matsubara said at the congress sponsored by the International League Against Epilepsy.
Of the 76 ESSs that occurred in 66 patients, 37% were focal awareness seizures, 35% were focal to bilateral tonic-clonic seizures, and 28% were focal impaired awareness seizures.
In a multivariate analysis adjusted for age, sex, body mass index, stroke subtype, and other potential confounders, statin therapy during acute management of stroke was independently associated with a 56% reduction in the relative risk of ESS. In contrast, a cortical stroke lesion was associated with a 2.83-fold increased risk.
Since this wasn’t a randomized trial of statin therapy, Dr. Matsubara and his coinvestigators felt the need to go further in analyzing the data. After extensive propensity score matching for atrial fibrillation, current smoking, systolic blood pressure, the presence or absence of a cortical stroke lesion, large vessel stenosis, and other possible confounders, they were left with two closely comparable groups: 886 statin-treated stroke patients and an equal number who were not on statin therapy during their acute stroke management. The key finding: The risk of ESS was reduced by a whopping 77% in the patients on statin therapy.
The neurologist observed that these new findings in acute ischemic stroke patients are consistent with an earlier study in a U.S. Veterans Affairs population, which demonstrated that statin therapy was associated with a significantly lower risk of new-onset geriatric epilepsy (J Am Geriatr Soc. 2009 Feb;57[2]:237-42).
As to the possible mechanism by which statins may protect against ESS, Dr. Matsubara noted that acute ischemic stroke causes toxic neuronal excitation because of blood-brain barrier disruption, ion channel dysfunction, altered gene expression, and increased release of neurotransmitters. In animal models, statins provide a neuroprotective effect by reducing glutamate levels, activating endothelial nitric oxide synthase, and inhibiting production of interleukin-6, tumor necrosis factor-alpha, and other inflammatory cytokines.
Asked about the intensity of the statin therapy, Dr. Matsubara replied that the target was typically an LDL cholesterol below 100 mg/dL.
He reported having no financial conflicts regarding the study, conducted free of commercial support.
SOURCE: Matsubara S et al. IEC 219, Abstract P002.
BANGKOK – Statin therapy, even when initiated only upon hospitalization for acute ischemic stroke, was associated with a striking reduction in the risk of early poststroke symptomatic seizure in a large observational study.
Using propensity-score matching to control for potential confounders, use of a statin during acute stroke management was associated with a “robust” 77% reduction in the risk of developing a symptomatic seizure within 7 days after hospital admission, Soichiro Matsubara, MD, reported at the International Epilepsy Congress.
This is an important finding because early symptomatic seizure (ESS) occurs in 2%-7% of patients following an acute ischemic stroke. Moreover, an Italian meta-analysis concluded that ESS was associated with a 4.4-fold increased risk of developing poststroke epilepsy (Epilepsia. 2016 Aug;57[8]:1205-14), noted Dr. Matsubara, a neurologist at the National Cerebral and Cardiovascular Center in Suita, Japan, as well as at Kumamoto (Japan) University.
He presented a study of 2,969 consecutive acute ischemic stroke patients with no history of epilepsy who were admitted to the Japanese comprehensive stroke center, of whom 2.2% experienced ESS. At physician discretion, 19% of the ESS cohort were on a statin during their acute stroke management, as were 55% of the no-ESS group. Four-fifths of patients on a statin initiated the drug only upon hospital admission.
Strokes tended to be more severe in the ESS group, with a median initial National Institutes of Health Stroke Scale score of 12.5, compared with 4 in the seizure-free patients. A cortical stroke lesion was evident upon imaging in 89% of the ESS group and 55% of no-ESS patients. Among ESS patients, 46% had a cardiometabolic stroke, compared with 34% of the no-ESS cohort. Mean C-reactive protein levels and white blood cell counts were significantly higher in the ESS cohort as well. Their median hospital length of stay was 25.5 days, versus 18 days in the no-ESS group, Dr. Matsubara said at the congress sponsored by the International League Against Epilepsy.
Of the 76 ESSs that occurred in 66 patients, 37% were focal awareness seizures, 35% were focal to bilateral tonic-clonic seizures, and 28% were focal impaired awareness seizures.
In a multivariate analysis adjusted for age, sex, body mass index, stroke subtype, and other potential confounders, statin therapy during acute management of stroke was independently associated with a 56% reduction in the relative risk of ESS. In contrast, a cortical stroke lesion was associated with a 2.83-fold increased risk.
Since this wasn’t a randomized trial of statin therapy, Dr. Matsubara and his coinvestigators felt the need to go further in analyzing the data. After extensive propensity score matching for atrial fibrillation, current smoking, systolic blood pressure, the presence or absence of a cortical stroke lesion, large vessel stenosis, and other possible confounders, they were left with two closely comparable groups: 886 statin-treated stroke patients and an equal number who were not on statin therapy during their acute stroke management. The key finding: The risk of ESS was reduced by a whopping 77% in the patients on statin therapy.
The neurologist observed that these new findings in acute ischemic stroke patients are consistent with an earlier study in a U.S. Veterans Affairs population, which demonstrated that statin therapy was associated with a significantly lower risk of new-onset geriatric epilepsy (J Am Geriatr Soc. 2009 Feb;57[2]:237-42).
As to the possible mechanism by which statins may protect against ESS, Dr. Matsubara noted that acute ischemic stroke causes toxic neuronal excitation because of blood-brain barrier disruption, ion channel dysfunction, altered gene expression, and increased release of neurotransmitters. In animal models, statins provide a neuroprotective effect by reducing glutamate levels, activating endothelial nitric oxide synthase, and inhibiting production of interleukin-6, tumor necrosis factor-alpha, and other inflammatory cytokines.
Asked about the intensity of the statin therapy, Dr. Matsubara replied that the target was typically an LDL cholesterol below 100 mg/dL.
He reported having no financial conflicts regarding the study, conducted free of commercial support.
SOURCE: Matsubara S et al. IEC 219, Abstract P002.
BANGKOK – Statin therapy, even when initiated only upon hospitalization for acute ischemic stroke, was associated with a striking reduction in the risk of early poststroke symptomatic seizure in a large observational study.
Using propensity-score matching to control for potential confounders, use of a statin during acute stroke management was associated with a “robust” 77% reduction in the risk of developing a symptomatic seizure within 7 days after hospital admission, Soichiro Matsubara, MD, reported at the International Epilepsy Congress.
This is an important finding because early symptomatic seizure (ESS) occurs in 2%-7% of patients following an acute ischemic stroke. Moreover, an Italian meta-analysis concluded that ESS was associated with a 4.4-fold increased risk of developing poststroke epilepsy (Epilepsia. 2016 Aug;57[8]:1205-14), noted Dr. Matsubara, a neurologist at the National Cerebral and Cardiovascular Center in Suita, Japan, as well as at Kumamoto (Japan) University.
He presented a study of 2,969 consecutive acute ischemic stroke patients with no history of epilepsy who were admitted to the Japanese comprehensive stroke center, of whom 2.2% experienced ESS. At physician discretion, 19% of the ESS cohort were on a statin during their acute stroke management, as were 55% of the no-ESS group. Four-fifths of patients on a statin initiated the drug only upon hospital admission.
Strokes tended to be more severe in the ESS group, with a median initial National Institutes of Health Stroke Scale score of 12.5, compared with 4 in the seizure-free patients. A cortical stroke lesion was evident upon imaging in 89% of the ESS group and 55% of no-ESS patients. Among ESS patients, 46% had a cardiometabolic stroke, compared with 34% of the no-ESS cohort. Mean C-reactive protein levels and white blood cell counts were significantly higher in the ESS cohort as well. Their median hospital length of stay was 25.5 days, versus 18 days in the no-ESS group, Dr. Matsubara said at the congress sponsored by the International League Against Epilepsy.
Of the 76 ESSs that occurred in 66 patients, 37% were focal awareness seizures, 35% were focal to bilateral tonic-clonic seizures, and 28% were focal impaired awareness seizures.
In a multivariate analysis adjusted for age, sex, body mass index, stroke subtype, and other potential confounders, statin therapy during acute management of stroke was independently associated with a 56% reduction in the relative risk of ESS. In contrast, a cortical stroke lesion was associated with a 2.83-fold increased risk.
Since this wasn’t a randomized trial of statin therapy, Dr. Matsubara and his coinvestigators felt the need to go further in analyzing the data. After extensive propensity score matching for atrial fibrillation, current smoking, systolic blood pressure, the presence or absence of a cortical stroke lesion, large vessel stenosis, and other possible confounders, they were left with two closely comparable groups: 886 statin-treated stroke patients and an equal number who were not on statin therapy during their acute stroke management. The key finding: The risk of ESS was reduced by a whopping 77% in the patients on statin therapy.
The neurologist observed that these new findings in acute ischemic stroke patients are consistent with an earlier study in a U.S. Veterans Affairs population, which demonstrated that statin therapy was associated with a significantly lower risk of new-onset geriatric epilepsy (J Am Geriatr Soc. 2009 Feb;57[2]:237-42).
As to the possible mechanism by which statins may protect against ESS, Dr. Matsubara noted that acute ischemic stroke causes toxic neuronal excitation because of blood-brain barrier disruption, ion channel dysfunction, altered gene expression, and increased release of neurotransmitters. In animal models, statins provide a neuroprotective effect by reducing glutamate levels, activating endothelial nitric oxide synthase, and inhibiting production of interleukin-6, tumor necrosis factor-alpha, and other inflammatory cytokines.
Asked about the intensity of the statin therapy, Dr. Matsubara replied that the target was typically an LDL cholesterol below 100 mg/dL.
He reported having no financial conflicts regarding the study, conducted free of commercial support.
SOURCE: Matsubara S et al. IEC 219, Abstract P002.
REPORTING FROM IEC 2019
Transdermal estradiol may modulate the relationship between sleep, cognition
LOS ANGELES – Estrogen therapy may have scored another goal in its comeback game, as a 7-year prospective study shows that a transdermal formulation preserves some measures of cognitive function and brain architecture in postmenopausal women.
In addition to performing better on subjective tests of memory, women using the estrogen patch experienced less cortical atrophy and were less likely to show amyloid on brain imaging. The observations were moderately associated with the improved sleep these women reported, Burcu Zeydan, MD, said at the Alzheimer’s Association International Conference.
“By 7 years, among the cognitive domains studied ... [less brain and cognitive change] correlated with lower global sleep score, meaning better sleep quality in the estradiol group,” said Dr. Zeydan, assistant professor of radiology at the Mayo Clinic in Rochester, Minn. “We previously found that preservation of dorsolateral prefrontal cortex over 7 years was associated with lower cortical beta-amyloid deposition on PET only in the estradiol group, pointing out the potential role of estrogen receptors in modulating this relationship.”
Dysregulated sleep is more common among women than men, particularly as menopause approaches and estrogen levels fluctuate, then decline, Dr. Zeydan said.
Dr. Zeydan reported the sleep substudy of KEEPS (the Kronos Early Estrogen Prevention Study), a randomized, double-blind, placebo-controlled, multisite trial that compared oral conjugated equine estrogen with transdermal estradiol. A control group received oral placebo and a placebo patch.*
Brain architecture was similar between the placebo and transdermal groups, but it was actually worse in some measures in the oral-estrogen group, compared with the placebo group. Women taking oral estrogen had more white matter hyperintensities, greater ventricle enlargement, and more cortical thinning. Those differences resolved after they stopped taking the oral formulation, bringing them into line with the transdermal and placebo groups.
The investigation also found that the transdermal group showed lower cerebral amyloid binding on PET scans relative to both placebo and oral estrogen.
“The relative preservation of dorsolateral prefrontal cortical volume in the [transdermal estradiol] group over 7 years indicates that hormone therapy may have long-term effects on the brain,” the team concluded. They noted that the original KEEPS study didn’t find any cognitive correlation with these changes.
The subanalysis looked at 69 women of the KEEPS cohort who had been followed for the full 7 years (4 years on treatment and 3 years off treatment). They were randomized to oral placebo and a placebo patch,* oral conjugated equine estrogen (0.45 mg/day), or transdermal estradiol (50 mcg/day). Participants in the active treatment groups received oral micronized progesterone 12 days each month. All had complete data on cognitive testing and brain imaging. Sleep quality was measured by the Pittsburgh Sleep Quality Index. Dr. Zeydan compared cognition and brain architecture findings in relation to the sleep score; lower scores mean better sleep.
The women were aged 42-58 years at baseline, and within 36 months from menopause. They had no history of menopausal hormone therapy or cardiovascular disease.
The investigators were particularly interested in how estrogen might have modulated the disturbed sleep patterns that often accompany perimenopause and early menopause, and whether the observed brain and cognitive changes tracked with sleep quality.
“During this time, 40% to 60% of women report problems sleeping, and estrogen decline seems to play an important role in sleep disturbances during this phase,” Dr. Zeydan said. “Although poor sleep quality is common in recently menopausal women, sleep quality improves with hormone therapy, as was previously demonstrated in KEEPS hormone therapy trial in recently menopausal women.”
By year 7, the cohort’s mean age was 61 years. The majority had at least some college education. The percentage who carried an apolipoprotein E epsilon-4 allele varied by group, with 15% positivity in the oral group, 48% in the transdermal group, and 16% in the placebo group.
Cognitive function was estimated with a global cognitive measure and four cognitive domain scores: verbal learning and memory, auditory attention and working memory, visual attention and executive function, and mental flexibility.
Higher attention and executive function scores were moderately correlated with a lower sleep score in the transdermal group (r = –0.54, a significant difference compared with the oral formulation). Lower sleep scores also showed a moderate correlation with preserved cortical volume of the dorsolateral prefrontal region (r = –0.47, also significantly different from the oral group).
Lower brain amyloid also positively correlated with better sleep. The correlation between sleep and global amyloid burden in the transdermal group was also moderate (r = 0.45), while the correlation in the oral group was significantly weaker (r = 0.18).
“We can say that sleep quality and transdermal estradiol during early postmenopausal years somehow interact to influence beta-amyloid deposition, preservation of dorsolateral prefrontal cortex volume, and attention and executive function,” Dr. Zeydan said.
Dr. Zeydan had no financial disclosures.
*Correction, 8/7/2019: An earlier version of this story did not make clear that participants in the control group received oral placebo and a placebo patch.
LOS ANGELES – Estrogen therapy may have scored another goal in its comeback game, as a 7-year prospective study shows that a transdermal formulation preserves some measures of cognitive function and brain architecture in postmenopausal women.
In addition to performing better on subjective tests of memory, women using the estrogen patch experienced less cortical atrophy and were less likely to show amyloid on brain imaging. The observations were moderately associated with the improved sleep these women reported, Burcu Zeydan, MD, said at the Alzheimer’s Association International Conference.
“By 7 years, among the cognitive domains studied ... [less brain and cognitive change] correlated with lower global sleep score, meaning better sleep quality in the estradiol group,” said Dr. Zeydan, assistant professor of radiology at the Mayo Clinic in Rochester, Minn. “We previously found that preservation of dorsolateral prefrontal cortex over 7 years was associated with lower cortical beta-amyloid deposition on PET only in the estradiol group, pointing out the potential role of estrogen receptors in modulating this relationship.”
Dysregulated sleep is more common among women than men, particularly as menopause approaches and estrogen levels fluctuate, then decline, Dr. Zeydan said.
Dr. Zeydan reported the sleep substudy of KEEPS (the Kronos Early Estrogen Prevention Study), a randomized, double-blind, placebo-controlled, multisite trial that compared oral conjugated equine estrogen with transdermal estradiol. A control group received oral placebo and a placebo patch.*
Brain architecture was similar between the placebo and transdermal groups, but it was actually worse in some measures in the oral-estrogen group, compared with the placebo group. Women taking oral estrogen had more white matter hyperintensities, greater ventricle enlargement, and more cortical thinning. Those differences resolved after they stopped taking the oral formulation, bringing them into line with the transdermal and placebo groups.
The investigation also found that the transdermal group showed lower cerebral amyloid binding on PET scans relative to both placebo and oral estrogen.
“The relative preservation of dorsolateral prefrontal cortical volume in the [transdermal estradiol] group over 7 years indicates that hormone therapy may have long-term effects on the brain,” the team concluded. They noted that the original KEEPS study didn’t find any cognitive correlation with these changes.
The subanalysis looked at 69 women of the KEEPS cohort who had been followed for the full 7 years (4 years on treatment and 3 years off treatment). They were randomized to oral placebo and a placebo patch,* oral conjugated equine estrogen (0.45 mg/day), or transdermal estradiol (50 mcg/day). Participants in the active treatment groups received oral micronized progesterone 12 days each month. All had complete data on cognitive testing and brain imaging. Sleep quality was measured by the Pittsburgh Sleep Quality Index. Dr. Zeydan compared cognition and brain architecture findings in relation to the sleep score; lower scores mean better sleep.
The women were aged 42-58 years at baseline, and within 36 months from menopause. They had no history of menopausal hormone therapy or cardiovascular disease.
The investigators were particularly interested in how estrogen might have modulated the disturbed sleep patterns that often accompany perimenopause and early menopause, and whether the observed brain and cognitive changes tracked with sleep quality.
“During this time, 40% to 60% of women report problems sleeping, and estrogen decline seems to play an important role in sleep disturbances during this phase,” Dr. Zeydan said. “Although poor sleep quality is common in recently menopausal women, sleep quality improves with hormone therapy, as was previously demonstrated in KEEPS hormone therapy trial in recently menopausal women.”
By year 7, the cohort’s mean age was 61 years. The majority had at least some college education. The percentage who carried an apolipoprotein E epsilon-4 allele varied by group, with 15% positivity in the oral group, 48% in the transdermal group, and 16% in the placebo group.
Cognitive function was estimated with a global cognitive measure and four cognitive domain scores: verbal learning and memory, auditory attention and working memory, visual attention and executive function, and mental flexibility.
Higher attention and executive function scores were moderately correlated with a lower sleep score in the transdermal group (r = –0.54, a significant difference compared with the oral formulation). Lower sleep scores also showed a moderate correlation with preserved cortical volume of the dorsolateral prefrontal region (r = –0.47, also significantly different from the oral group).
Lower brain amyloid also positively correlated with better sleep. The correlation between sleep and global amyloid burden in the transdermal group was also moderate (r = 0.45), while the correlation in the oral group was significantly weaker (r = 0.18).
“We can say that sleep quality and transdermal estradiol during early postmenopausal years somehow interact to influence beta-amyloid deposition, preservation of dorsolateral prefrontal cortex volume, and attention and executive function,” Dr. Zeydan said.
Dr. Zeydan had no financial disclosures.
*Correction, 8/7/2019: An earlier version of this story did not make clear that participants in the control group received oral placebo and a placebo patch.
LOS ANGELES – Estrogen therapy may have scored another goal in its comeback game, as a 7-year prospective study shows that a transdermal formulation preserves some measures of cognitive function and brain architecture in postmenopausal women.
In addition to performing better on subjective tests of memory, women using the estrogen patch experienced less cortical atrophy and were less likely to show amyloid on brain imaging. The observations were moderately associated with the improved sleep these women reported, Burcu Zeydan, MD, said at the Alzheimer’s Association International Conference.
“By 7 years, among the cognitive domains studied ... [less brain and cognitive change] correlated with lower global sleep score, meaning better sleep quality in the estradiol group,” said Dr. Zeydan, assistant professor of radiology at the Mayo Clinic in Rochester, Minn. “We previously found that preservation of dorsolateral prefrontal cortex over 7 years was associated with lower cortical beta-amyloid deposition on PET only in the estradiol group, pointing out the potential role of estrogen receptors in modulating this relationship.”
Dysregulated sleep is more common among women than men, particularly as menopause approaches and estrogen levels fluctuate, then decline, Dr. Zeydan said.
Dr. Zeydan reported the sleep substudy of KEEPS (the Kronos Early Estrogen Prevention Study), a randomized, double-blind, placebo-controlled, multisite trial that compared oral conjugated equine estrogen with transdermal estradiol. A control group received oral placebo and a placebo patch.*
Brain architecture was similar between the placebo and transdermal groups, but it was actually worse in some measures in the oral-estrogen group, compared with the placebo group. Women taking oral estrogen had more white matter hyperintensities, greater ventricle enlargement, and more cortical thinning. Those differences resolved after they stopped taking the oral formulation, bringing them into line with the transdermal and placebo groups.
The investigation also found that the transdermal group showed lower cerebral amyloid binding on PET scans relative to both placebo and oral estrogen.
“The relative preservation of dorsolateral prefrontal cortical volume in the [transdermal estradiol] group over 7 years indicates that hormone therapy may have long-term effects on the brain,” the team concluded. They noted that the original KEEPS study didn’t find any cognitive correlation with these changes.
The subanalysis looked at 69 women of the KEEPS cohort who had been followed for the full 7 years (4 years on treatment and 3 years off treatment). They were randomized to oral placebo and a placebo patch,* oral conjugated equine estrogen (0.45 mg/day), or transdermal estradiol (50 mcg/day). Participants in the active treatment groups received oral micronized progesterone 12 days each month. All had complete data on cognitive testing and brain imaging. Sleep quality was measured by the Pittsburgh Sleep Quality Index. Dr. Zeydan compared cognition and brain architecture findings in relation to the sleep score; lower scores mean better sleep.
The women were aged 42-58 years at baseline, and within 36 months from menopause. They had no history of menopausal hormone therapy or cardiovascular disease.
The investigators were particularly interested in how estrogen might have modulated the disturbed sleep patterns that often accompany perimenopause and early menopause, and whether the observed brain and cognitive changes tracked with sleep quality.
“During this time, 40% to 60% of women report problems sleeping, and estrogen decline seems to play an important role in sleep disturbances during this phase,” Dr. Zeydan said. “Although poor sleep quality is common in recently menopausal women, sleep quality improves with hormone therapy, as was previously demonstrated in KEEPS hormone therapy trial in recently menopausal women.”
By year 7, the cohort’s mean age was 61 years. The majority had at least some college education. The percentage who carried an apolipoprotein E epsilon-4 allele varied by group, with 15% positivity in the oral group, 48% in the transdermal group, and 16% in the placebo group.
Cognitive function was estimated with a global cognitive measure and four cognitive domain scores: verbal learning and memory, auditory attention and working memory, visual attention and executive function, and mental flexibility.
Higher attention and executive function scores were moderately correlated with a lower sleep score in the transdermal group (r = –0.54, a significant difference compared with the oral formulation). Lower sleep scores also showed a moderate correlation with preserved cortical volume of the dorsolateral prefrontal region (r = –0.47, also significantly different from the oral group).
Lower brain amyloid also positively correlated with better sleep. The correlation between sleep and global amyloid burden in the transdermal group was also moderate (r = 0.45), while the correlation in the oral group was significantly weaker (r = 0.18).
“We can say that sleep quality and transdermal estradiol during early postmenopausal years somehow interact to influence beta-amyloid deposition, preservation of dorsolateral prefrontal cortex volume, and attention and executive function,” Dr. Zeydan said.
Dr. Zeydan had no financial disclosures.
*Correction, 8/7/2019: An earlier version of this story did not make clear that participants in the control group received oral placebo and a placebo patch.
REPORTING FROM AAIC 2019
NOACs benefit early stage chronic kidney disease patients
Non–vitamin K oral anticoagulants (NOACs) significantly reduced the risk of stroke or systemic embolism compared to vitamin K antagonists (VKAs) for patients in the early stages of chronic kidney disease and comorbid atrial fibrillation, based on data from a meta-analysis of roughly 34,000 patients.
Chronic kidney disease increases the risk of complications including stroke, congestive heart failure, and death in patients who also have atrial fibrillation, but most trials of anticoagulant therapy to reduce the risk of such events have excluded these patients, wrote Jeffrey T. Ha, MBBS, of the George Institute for Global Health, Newtown, Australia, and colleagues.
To assess the benefits and harms of oral anticoagulants for multiple indications in chronic kidney disease patients, the researchers conducted a meta-analysis of 45 studies including 34,082 individuals. The findings were published in the Annals of Internal Medicine. The analysis included 8 trials of end stage kidney disease patients on dialysis; the remaining trials excluded patients with creatinine clearance less than 20 mL/min or an estimated glomerular filtration rate less than 15 mL/min per 1.73 m2. The interventional agents were rivaroxaban, dabigatran, apixaban, edoxaban, betrixaban, warfarin, and acenocoumarol.
A notable finding was the significant reduction in relative risk of stroke or systemic embolism (21%), hemorrhagic stroke (52%), and intracranial hemorrhage (51%) for early-stage chronic kidney disease patients with atrial fibrillation given NOACs, compared with those given VKAs.
The evidence for the superiority of NOACs over VKAs for reducing risk of venous thromboembolism (VTE) or VTE-related death was uncertain, as was the evidence to draw any conclusions about benefits and harms of either NOACs or VKAs for patients with advanced or end-stage kidney disease.
Across all trials, NOACs appeared to reduce the relative risk of major bleeding, compared with VKAs by roughly 25%, but the difference was not statistically significant, the researchers noted.
The findings were limited by the lack of evidence for oral anticoagulant use in patients with advanced chronic or end-stage kidney disease, as well as inability to assess differences among NOACs, the researchers noted. However, the results suggest that NOACs may be recommended over VKAs for the subgroup of early-stage chronic kidney disease patients with atrial fibrillation, they said.
Several additional trials are in progress, and future trials “should include not only participants with dialysis-dependent ESKD [end-stage kidney disease] but also those with CrCl [creatinine clearance of] less than 25 mL/min,” and compare NOACs with placebo as well, they noted.
Lead author Dr. Ha is supported by a University Postgraduate Award from University of New South Wales, Sydney, but had no financial conflicts to disclose; coauthors disclosed support from various organizations as well as pharmaceutical companies including Baxter, Amgen, Eli Lilly, Boehringer Ingelheim, Vifor Pharma, Janssen, Pfizer, Bristol-Myers Squibb, and GlaxoSmithKline.
SOURCE: Ha JT et al. Ann Intern Med. 2019 July 15. doi: 10.7326/M19-0087
The significant reduction in risk of hemorrhagic stroke, recurrent venous thromboembolism, and VTE-related deaths in patients with early-stage chronic kidney disease given a NOAC [non–vitamin K oral anticoagulants] in a meta-analysis supports clinical application, but is there a level of renal dysfunction for which clinicians should apply greater caution in extrapolating these findings? As the evidence supporting the safety and effectiveness of NOACs in the general population increases, there is a renewed interest in defining the role of anticoagulant therapy to prevent stroke and VTE in patients with chronic kidney disease and end-stage kidney disease. This interest is driven in part by uncertainty as to the benefits vs. harms of warfarin for patients with chronic kidney disease. The data in the meta-analysis by Ha and colleagues do not support any benefits for patients with end-stage disease, but the results of two ongoing clinical trials of patients with atrial fibrillation and end-stage kidney disease may offer insights.
Until the results of these trials become available, the decision to use anticoagulant therapy in patients with end-stage kidney disease will continue to require an individualized approach that balances potential benefits and harms.
Ainslie Hildebrand, MD, of University of Alberta, Edmonton; Christine Ribic, MD, of McMaster University, Hamilton, Ont.; and Deborah Zimmerman, MD, of the University of Ottawa, made these comments in an accompanying editorial (Ann Intern Med. 2019 July 15. doi:10.7326/M19-1504). Dr. Ribic disclosed grants from Pfizer, Leo Pharma, and Astellas Pharma. Dr. Hildebrand and Dr. Zimmerman had no financial conflicts to disclose.
The significant reduction in risk of hemorrhagic stroke, recurrent venous thromboembolism, and VTE-related deaths in patients with early-stage chronic kidney disease given a NOAC [non–vitamin K oral anticoagulants] in a meta-analysis supports clinical application, but is there a level of renal dysfunction for which clinicians should apply greater caution in extrapolating these findings? As the evidence supporting the safety and effectiveness of NOACs in the general population increases, there is a renewed interest in defining the role of anticoagulant therapy to prevent stroke and VTE in patients with chronic kidney disease and end-stage kidney disease. This interest is driven in part by uncertainty as to the benefits vs. harms of warfarin for patients with chronic kidney disease. The data in the meta-analysis by Ha and colleagues do not support any benefits for patients with end-stage disease, but the results of two ongoing clinical trials of patients with atrial fibrillation and end-stage kidney disease may offer insights.
Until the results of these trials become available, the decision to use anticoagulant therapy in patients with end-stage kidney disease will continue to require an individualized approach that balances potential benefits and harms.
Ainslie Hildebrand, MD, of University of Alberta, Edmonton; Christine Ribic, MD, of McMaster University, Hamilton, Ont.; and Deborah Zimmerman, MD, of the University of Ottawa, made these comments in an accompanying editorial (Ann Intern Med. 2019 July 15. doi:10.7326/M19-1504). Dr. Ribic disclosed grants from Pfizer, Leo Pharma, and Astellas Pharma. Dr. Hildebrand and Dr. Zimmerman had no financial conflicts to disclose.
The significant reduction in risk of hemorrhagic stroke, recurrent venous thromboembolism, and VTE-related deaths in patients with early-stage chronic kidney disease given a NOAC [non–vitamin K oral anticoagulants] in a meta-analysis supports clinical application, but is there a level of renal dysfunction for which clinicians should apply greater caution in extrapolating these findings? As the evidence supporting the safety and effectiveness of NOACs in the general population increases, there is a renewed interest in defining the role of anticoagulant therapy to prevent stroke and VTE in patients with chronic kidney disease and end-stage kidney disease. This interest is driven in part by uncertainty as to the benefits vs. harms of warfarin for patients with chronic kidney disease. The data in the meta-analysis by Ha and colleagues do not support any benefits for patients with end-stage disease, but the results of two ongoing clinical trials of patients with atrial fibrillation and end-stage kidney disease may offer insights.
Until the results of these trials become available, the decision to use anticoagulant therapy in patients with end-stage kidney disease will continue to require an individualized approach that balances potential benefits and harms.
Ainslie Hildebrand, MD, of University of Alberta, Edmonton; Christine Ribic, MD, of McMaster University, Hamilton, Ont.; and Deborah Zimmerman, MD, of the University of Ottawa, made these comments in an accompanying editorial (Ann Intern Med. 2019 July 15. doi:10.7326/M19-1504). Dr. Ribic disclosed grants from Pfizer, Leo Pharma, and Astellas Pharma. Dr. Hildebrand and Dr. Zimmerman had no financial conflicts to disclose.
Non–vitamin K oral anticoagulants (NOACs) significantly reduced the risk of stroke or systemic embolism compared to vitamin K antagonists (VKAs) for patients in the early stages of chronic kidney disease and comorbid atrial fibrillation, based on data from a meta-analysis of roughly 34,000 patients.
Chronic kidney disease increases the risk of complications including stroke, congestive heart failure, and death in patients who also have atrial fibrillation, but most trials of anticoagulant therapy to reduce the risk of such events have excluded these patients, wrote Jeffrey T. Ha, MBBS, of the George Institute for Global Health, Newtown, Australia, and colleagues.
To assess the benefits and harms of oral anticoagulants for multiple indications in chronic kidney disease patients, the researchers conducted a meta-analysis of 45 studies including 34,082 individuals. The findings were published in the Annals of Internal Medicine. The analysis included 8 trials of end stage kidney disease patients on dialysis; the remaining trials excluded patients with creatinine clearance less than 20 mL/min or an estimated glomerular filtration rate less than 15 mL/min per 1.73 m2. The interventional agents were rivaroxaban, dabigatran, apixaban, edoxaban, betrixaban, warfarin, and acenocoumarol.
A notable finding was the significant reduction in relative risk of stroke or systemic embolism (21%), hemorrhagic stroke (52%), and intracranial hemorrhage (51%) for early-stage chronic kidney disease patients with atrial fibrillation given NOACs, compared with those given VKAs.
The evidence for the superiority of NOACs over VKAs for reducing risk of venous thromboembolism (VTE) or VTE-related death was uncertain, as was the evidence to draw any conclusions about benefits and harms of either NOACs or VKAs for patients with advanced or end-stage kidney disease.
Across all trials, NOACs appeared to reduce the relative risk of major bleeding, compared with VKAs by roughly 25%, but the difference was not statistically significant, the researchers noted.
The findings were limited by the lack of evidence for oral anticoagulant use in patients with advanced chronic or end-stage kidney disease, as well as inability to assess differences among NOACs, the researchers noted. However, the results suggest that NOACs may be recommended over VKAs for the subgroup of early-stage chronic kidney disease patients with atrial fibrillation, they said.
Several additional trials are in progress, and future trials “should include not only participants with dialysis-dependent ESKD [end-stage kidney disease] but also those with CrCl [creatinine clearance of] less than 25 mL/min,” and compare NOACs with placebo as well, they noted.
Lead author Dr. Ha is supported by a University Postgraduate Award from University of New South Wales, Sydney, but had no financial conflicts to disclose; coauthors disclosed support from various organizations as well as pharmaceutical companies including Baxter, Amgen, Eli Lilly, Boehringer Ingelheim, Vifor Pharma, Janssen, Pfizer, Bristol-Myers Squibb, and GlaxoSmithKline.
SOURCE: Ha JT et al. Ann Intern Med. 2019 July 15. doi: 10.7326/M19-0087
Non–vitamin K oral anticoagulants (NOACs) significantly reduced the risk of stroke or systemic embolism compared to vitamin K antagonists (VKAs) for patients in the early stages of chronic kidney disease and comorbid atrial fibrillation, based on data from a meta-analysis of roughly 34,000 patients.
Chronic kidney disease increases the risk of complications including stroke, congestive heart failure, and death in patients who also have atrial fibrillation, but most trials of anticoagulant therapy to reduce the risk of such events have excluded these patients, wrote Jeffrey T. Ha, MBBS, of the George Institute for Global Health, Newtown, Australia, and colleagues.
To assess the benefits and harms of oral anticoagulants for multiple indications in chronic kidney disease patients, the researchers conducted a meta-analysis of 45 studies including 34,082 individuals. The findings were published in the Annals of Internal Medicine. The analysis included 8 trials of end stage kidney disease patients on dialysis; the remaining trials excluded patients with creatinine clearance less than 20 mL/min or an estimated glomerular filtration rate less than 15 mL/min per 1.73 m2. The interventional agents were rivaroxaban, dabigatran, apixaban, edoxaban, betrixaban, warfarin, and acenocoumarol.
A notable finding was the significant reduction in relative risk of stroke or systemic embolism (21%), hemorrhagic stroke (52%), and intracranial hemorrhage (51%) for early-stage chronic kidney disease patients with atrial fibrillation given NOACs, compared with those given VKAs.
The evidence for the superiority of NOACs over VKAs for reducing risk of venous thromboembolism (VTE) or VTE-related death was uncertain, as was the evidence to draw any conclusions about benefits and harms of either NOACs or VKAs for patients with advanced or end-stage kidney disease.
Across all trials, NOACs appeared to reduce the relative risk of major bleeding, compared with VKAs by roughly 25%, but the difference was not statistically significant, the researchers noted.
The findings were limited by the lack of evidence for oral anticoagulant use in patients with advanced chronic or end-stage kidney disease, as well as inability to assess differences among NOACs, the researchers noted. However, the results suggest that NOACs may be recommended over VKAs for the subgroup of early-stage chronic kidney disease patients with atrial fibrillation, they said.
Several additional trials are in progress, and future trials “should include not only participants with dialysis-dependent ESKD [end-stage kidney disease] but also those with CrCl [creatinine clearance of] less than 25 mL/min,” and compare NOACs with placebo as well, they noted.
Lead author Dr. Ha is supported by a University Postgraduate Award from University of New South Wales, Sydney, but had no financial conflicts to disclose; coauthors disclosed support from various organizations as well as pharmaceutical companies including Baxter, Amgen, Eli Lilly, Boehringer Ingelheim, Vifor Pharma, Janssen, Pfizer, Bristol-Myers Squibb, and GlaxoSmithKline.
SOURCE: Ha JT et al. Ann Intern Med. 2019 July 15. doi: 10.7326/M19-0087
FROM THE ANNALS OF INTERNAL MEDICINE
The costs and benefits of SGLT2 inhibitors & GLP-1 RAs
The options for treating type 2 diabetes without insulin have grown beyond metformin to include a long list of sodium-glucose cotransporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists that can be taken with or without metformin. These new drugs have cardiovascular and kidney benefits and help with weight loss, but they also carry risks and, according to some experts, their costs can be prohibitively expensive.
Given the medical community’s long-term experience with treating patients with metformin, and metformin’s lower cost, most of the physicians interviewed for this article advise using SGLT2 inhibitors and GLP-1 receptor agonists as second-line treatments. Others said that they would prefer to use the newer drugs as first-line therapies in select high-risk patients, but prior authorization hurdles created by insurance companies make that approach too burdensome.
“The economics of U.S. health care is stacked against many of our patients with diabetes in the current era,” Robert H. Hopkins Jr., MD, said in an interview.
Even when their insurance approves the drugs, patients still may not be able to afford the copay, explained Dr. Hopkins, professor of internal medicine and pediatrics and director of the division of general internal medicine at the University of Arkansas for Medical Sciences, Little Rock. “Sometimes patients can purchase drugs at a lower cost than the copay to purchase with the ‘drug coverage’ in their insurance plan – unfortunately, this is not the case with the newer diabetes medications we are discussing here.”
“SGLT2 inhibitors and GLP-1 agonists can cost several hundred dollars a month, and insurers often balk at paying for them. They’ll say, ‘Have you tried metformin?’ ” explained endocrinologist Victor Lawrence Roberts, MD, in a interview. “We have to work with insurance companies the best we can in a stepwise fashion.”
According to Dr. Roberts, 80% of his patients with diabetes struggle with the cost of medicine in general. “They’re either underinsured or not insured or their formulary is limited.
Douglas S. Paauw, MD, agreed in an interview that the newer drugs can be problematic on the insurance front.
“For some patients they aren’t affordable, especially for the uninsured if you can’t get them on an assistance program,” said Dr. Paauw, who is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the university.
Dr. Hopkins, who is on the Internal Medicine News board, noted that “unfortunately, the treatment of type 2 diabetes in patients who cannot achieve control with metformin, diet, weight control, and exercise is a story of the ‘haves’ and the ‘have nots.’ The ‘haves’ are those who have pharmacy benefits which make access to newer agents like SGLT2 inhibitors and GLP-1 agonists a possibility.”
“I have had very few of the ‘have nots’ who have been able to even consider these newer agents, which carry price tags of $600-$1,300 a month even with the availability of discounting coupons in the marketplace,” he added. “Most of these patients end up requiring a sulfonylurea or TZD [thiazolidinedione] as a second agent to achieve glycemic control. This makes it very difficult to achieve sufficient weight and metabolic control to avoid an eventual switch to insulin.”
Fatima Z. Syed, MD, an endocrine-trained general internist at DukeHealth in Durham, N.C., said she prescribes SGLT2 inhibitors and GLP-1 receptor agonists in combination with metformin. “I prescribe them frequently, but they are not first-line treatments,” she explained.
“Nothing replaces diet and exercise” as therapy for patients with type 2 diabetes, she added.
Neil S. Skolnik, MD, said that insurance companies were not preventing patients from using these drugs in his experience. He also provided an optimistic take on the accessibility of these drugs in the near future.
“Most insurance companies are now covering select SGLT2 inhibitors and GLP-1 receptor agonists for appropriate patients and those companies that currently do not will soon have to,” said Dr. Skolnik, who is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
“The outcomes [associated with use of the new drugs] are robust, the benefits are large, and are well worth the cost,” he added.
The side effects
While others praised these drugs for their beneficial effects, they also noted that the side effects of these drugs are serious and must be discussed with patients.
GLP-1 receptor agonists are linked to gastrointestinal symptoms, especially nausea, while SGLT2 inhibitors have been linked to kidney failure, ketoacidosis, and more. The Food and Drug Administration warned in 2018 that the SGLT2 inhibitors can cause a rare serious infection known as Fournier’s gangrene – necrotizing fasciitis of the perineum.
“We have to tell our patients to let us know right away if they get pain or swelling in the genital area,” Dr. Paauw, who is on the Internal Medicine News board, noted. “The chance that an infection could explode quickly is higher in those who take these drugs.”
Amputation risks also are associated with taking the SGLT2 inhibitor canagliflozin (Invokana). The FDA requires the manufacturer of this drug to include a black-box warning about the risk of “lower-limb amputations, most frequently of the toe and midfoot,” but also the leg. In approval trials, the risk doubled versus placebo.
These amputation risks “put a damper on some of the enthusiasm on behalf of physicians and patients ... for taking this drug,” noted Dr. Roberts, who is a professor of internal medicine at the University of Central Florida, Orlando.
While a manufacturer-funded study released last year found no link to amputations, the results weren’t powerful enough to rule out a moderately increased risk.
“[If] you are at high risk for having an amputation, we really have to take this risk very seriously,” said John B. Buse, MD, chief of the division of endocrinology at the University of North Carolina at Chapel Hill, in a presentation about the study at the 2018 annual scientific sessions of the American Diabetes Association.
The benefits
Despite these risks of adverse events, most interviewed agreed that the many benefits observed in those taking SGLT2 inhibitors or GLP-1 receptor agonists make them worth prescribing, at least to those who are able to afford them.
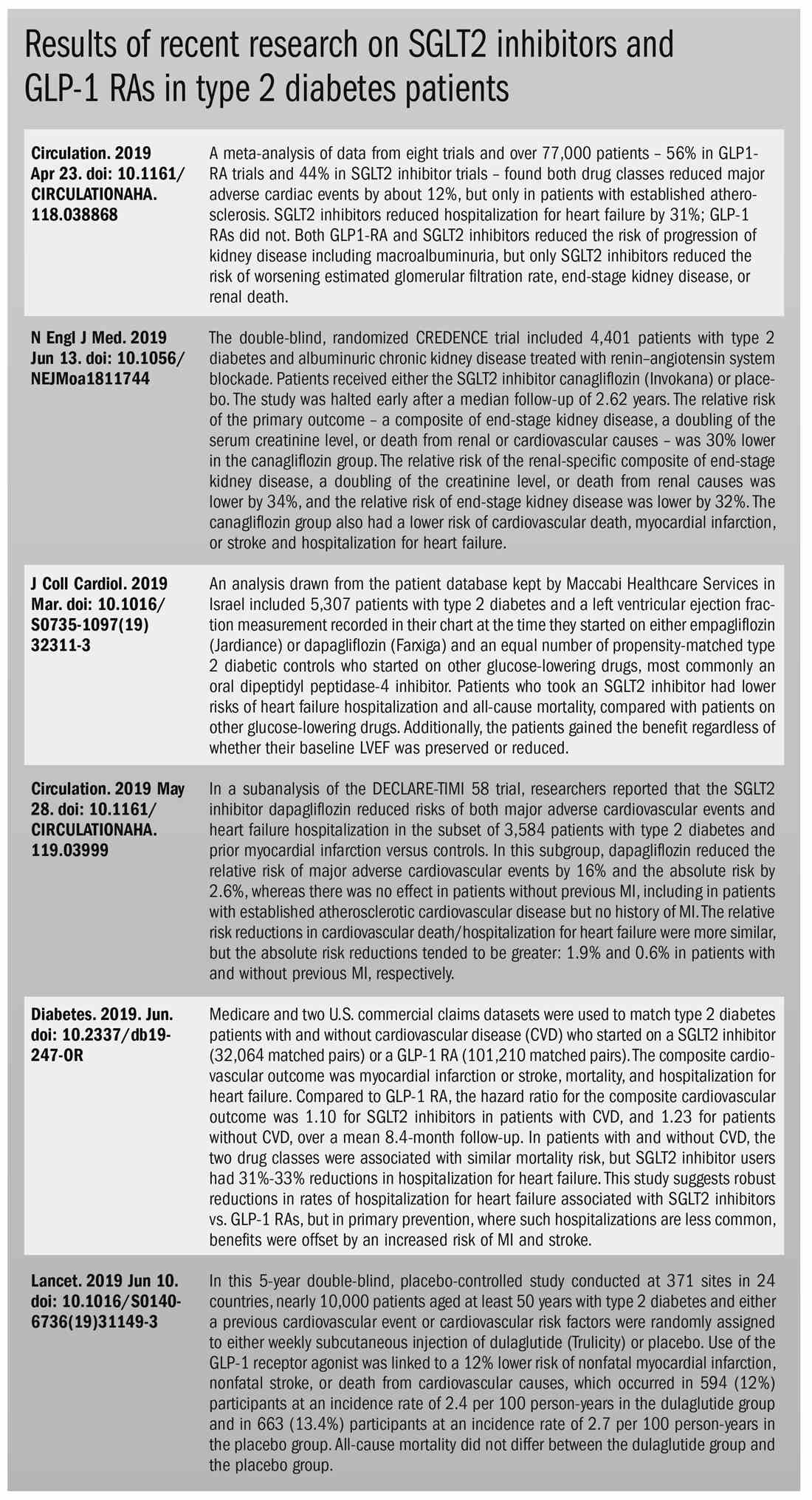
Both SGLT2 inhibitors and GLP-1 receptor agonists appear to have significant cardiovascular benefits. A 2019 meta-analysis and systematic review found that both drugs reduced major adverse cardiac events by about 12% (Circulation. 2019 Apr 23;139[17]:2022-31).
“They don’t cause hypoglycemia, they lower blood pressure, they don’t cause weight gain, and they might promote weight loss,” noted Dr. Paauw.
SGLT2 inhibitors also have shown signs of kidney benefits. The CREDENCE trial linked canagliflozin to a lowering of kidney disorders versus placebo (N Engl J Med. 2019 Jun 13;380[24]:2295-306). “The relative risk of the renal-specific composite of end-stage kidney disease, a doubling of the creatinine level, or death from renal causes was lower by 34% (hazard ratio, 0.66; 95% confidence interval, 0.53-0.81; P less than .001), and the relative risk of end-stage kidney disease was lower by 32% (HR, 0.68; 95% CI, 0.54-0.86; P = .002),” the trial investigators wrote.
“They showed very nicely that the drug improved the kidney function of those patients and reduced the kidney deterioration,” said Yehuda Handelsman, MD, an endocrinologist in Tarzana, Calif., who chaired the 2011 and 2015 American Association of Clinical Endocrinologists’ Comprehensive Diabetes Guidelines. The study was especially impressive, he added, because it included patients with low kidney function.
SGLT2 inhibitors’ “diuretic mechanism explains why there is a substantial reduction in heart failure hospitalizations in patients who take these drugs,” said cardiologist Marc E. Goldschmidt, MD, director of the Heart Success Program at Atlantic Health System’s Morristown (N.J.) Medical Center, in an interview. “Both the EMPA-REG Outcome and the CREDENCE trials demonstrated substantial benefit of this class of medications by showing a lower risk of cardiovascular death as well as death from any cause and a lower risk of hospitalization for heart failure."
Overall, the SGLT2 trial data have been very consistent with a benefit for cardiovascular risk reduction, particularly in regard to heart failure hospitalizations and even in potentially preventing heart failure in diabetics,” he added.
Dr. Skolnik, a columnist for Family Practice News, cited SGLT2 inhibitors and GLP-1 receptor agonists’ ability to slow renal disease progression, promote weight loss, and prevent poor cardiac outcomes.“These drugs should be used, in addition to metformin, in all patients with diabetes and vascular disease. These proven outcomes are far better than we ever were able to achieve previously and the strength of the evidence at this point is very strong,” said Dr. Skolnik. “In addition to the benefits of decreasing the development of cardiovascular disease, serious heart failure, and slowing progression of renal disease, these two classes of medication have additional benefits. Both classes help patients lose weight, which is very different from what was found with either sulfonylureas or insulin, which cause patients to gain weight. Also both the SGLT2 inhibitors and the GLP-1 RAs [receptor agonists] have a low incidence of hypoglycemia. For all these reasons, these have become important medications for us to use in primary care.”
Other recent trials offer “very powerful data” about SGLT2 inhibitors, Dr. Roberts said. That’s good news, since “our approach needs to be toward cardiovascular protection and preservation as well as managing blood sugar.”An Israeli trial, whose results were released in May 2019 at the annual meeting of the American College of Cardiology, found that, compared with other glucose-lowering drugs, taking an SGLT2 inhibitor was associated with lower risks of heart failure hospitalization and all-cause mortality (HR, 0.54; 95% CI, 0.44-0.65; P less than .001). This trial also offered a new detail: The patients gained the benefit regardless of whether their baseline left ventricular ejection fraction was preserved or reduced (J Coll Cardiol. 2019 Mar;73[9]:suppl 1). The SGLT2 inhibitors used in this trial included dapagliflozin (Farxiga) and empagliflozin (Jardiance).
In another study released this year, a subanalysis of the DECLARE-TIMI 58 trial, researchers reported that the SGLT2 inhibitor dapagliflozin reduced risks of both major adverse cardiovascular events and heart failure hospitalization in the subset of patients with type 2 diabetes and prior myocardial infarction versus controls (Circulation. 2019 May 28;139[22]:2516-27). The absolute risk reduction for major adverse cardiovascular events was 1.9% (HR, 0.81; 95% CI, 0.65-1.00; P = .046), while it was 0.6% for heart failure hospitalization (HR, 0.85; 95% CI, 0.72-1.00; P = .055).
These and other studies “speak volumes about the efficacy of managing blood sugar and addressing our biggest nemesis, which is cardiovascular disease,” Dr. Roberts said. “It’s irrefutable. The data [are] very good.”
Dr. Paauw said an SGLT2 inhibitor or GLP-1 receptor agonist is best reserved for use in select patients with cardiovascular risks and type 2 diabetes that need management beyond metformin.
For example, they might fit a 70-year-old with persistent hypertension who’s already taking a couple of blood pressure medications. “If they have another cardiovascular risk factor, the cardiovascular protection piece will be a bigger deal,” he said. Also, “it will probably help lower their blood pressure so they can avoid taking another blood pressure medicine.”
Trials of both GLP-1 receptor agonists and SGLT2 inhibitors have shown benefits “in improving [major adverse cardiac events], with the SGLT2 class showing substantial benefit in improving both heart failure and renal outcomes as well,” noted Dr. Skolnik. “It is in this context that one must address the question of whether the price of the medications are worthwhile. With such substantial benefit, there is no question in my mind that – for patients who have underlying cardiovascular illness, which includes patients with existent coronary disease, history of stroke, transient ischemic attack, or peripheral vascular disease – it is far and away worth it to prescribe these classes of medications.”
Indeed, the American Diabetes Association and the European Association for the Study of Diabetes’ most recent guidelines now call for a GLP-1 receptor agonist – instead of insulin – to be the first injectable used to treat type 2 diabetes (Diabetes Care 2018 Dec; 41[12]:2669-701).
“For the relatively small number of my patients who have been able to access and use these medications for months or longer, more have tolerated the GLP-1 agonists than SGLT2 inhibitors primarily due to urinary issues,” noted Dr. Hopkins.
Dipeptidyl peptidase–4 inhibitors are another option in patients with type 2 diabetes, but research suggests they may not be a top option for patients with cardiovascular risk. A 2018 review noted that cardiovascular outcome trials for alogliptin (Nesina), saxagliptin (Onglyza), and sitagliptin (Januvia) showed noninferiority but failed to demonstrate any superiority, compared with placebo in patients with type 2 diabetes mellitus and high cardiovascular risk (Circ Res. 2018 May 11;122[10]:1439-59).
The combination therapies
Many of the newer drugs are available as combinations with other types of diabetes drugs. In some cases, physicians create their own form of combination therapy by separately prescribing two or more diabetes drugs. Earlier this year, a study suggested the benefits of this kind of add-on therapy: Diabetes outcomes improved in patients who took the GLP-1 receptor agonist semaglutide and an SGLT2 inhibitor (Lancet Diabetes Endocrinol. 2019 Mar 1. doi: 10.1016/S2213-8587[19]30066-X).
Dr. Roberts suggested caution, however, when prescribing combination therapies. “My recommendation is always to begin with the individual medications to see if the patient tolerates the drugs and then decide which component needs to be titrated. It’s hard to titrate a combination drug, and it doesn’t leave a lot of flexibility. You never know which drug is doing what.
Dr. Handelsman said some patients may need to take three medications such as metformin, an SGLT2 inhibitor, and a GLP-1 receptor agonist.
“I don’t recommend using the combinations if you’re not familiar with the drugs ... These are relatively new pharmaceuticals, and most of us are on a learning curve as to how they fit into the armamentarium. If a drug is tolerated with a good response, you can certainly consider going to the combination tablets,” he added.
There is at least one drug that combines these three classes: The newly FDA-approved Qternmet XR, which combines dapagliflozin (an SGLT2 inhibitor), saxagliptin (a GLP-1 receptor agonist), and metformin. As of mid-June 2019, it was not yet available in the United States. Its sister drug Qtern, which combines dapagliflozin and saxagliptin, costs more than $500 a month with a free coupon, according to goodrx.com. In contrast, metformin is extremely inexpensive, costing just a few dollars a month for a common starting dose.
What about adding insulin?
“Both [SGLT2 inhibitors and GLP-1 receptor agonists] work very well with insulin,” Dr. Handelsman said. “There is a nice additive effect on the reduction of [hemoglobin] A1c. The only caution is that, although neither SGLT2 inhibitors nor GLP-1 receptor agonists cause hypoglycemia, in combination with insulin they do increase the risk of hypoglycemia. You may have to adjust the dose of insulin.”
Dr. Hopkins warned that cost becomes an even bigger issue when you add insulin into the mix.
“When insulin comes into the discussion, we are again stuck with astronomical costs which many struggle to afford,” he explained.
Indeed, the price tag on these drugs seems to be the biggest problem physicians have with them.
“The challenges in managing patients with diabetes aren’t the risks associated with the drugs. It’s dealing with their insurers,” noted Dr. Roberts.
Dr. Hopkins, Dr. Paauw, Dr. Roberts, and Dr. Syed reported no disclosures. Dr. Buse is an investigator for Johnson and Johnson. Dr. Goldschmidt is paid to speak by Novartis. Dr. Handelsman reported research grants, consulting work, and speaker honoraria from Amgen, Gilead, Lilly, Merck, Novo Nordisk, and others. Dr Skolnik reported nonfinancial support from AstraZeneca, Boehringer Ingelheim, Sanofi, and GlaxoSmithKline and personal fees from AstraZeneca, Boehringer Ingelheim, and Eli Lilly. He also serves on the advisory boards of AstraZeneca, Boehringer Ingelheim, Teva Pharmaceutical, Eli Lilly, Sanofi, Janssen Pharmaceuticals, Intarcia, Mylan, and GlaxoSmithKline.
Dr. Paauw and Dr. Skolnik are columnists for Family Practice News and Internal Medicine News.
M. Alexander Otto contributed to this report.
The options for treating type 2 diabetes without insulin have grown beyond metformin to include a long list of sodium-glucose cotransporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists that can be taken with or without metformin. These new drugs have cardiovascular and kidney benefits and help with weight loss, but they also carry risks and, according to some experts, their costs can be prohibitively expensive.
Given the medical community’s long-term experience with treating patients with metformin, and metformin’s lower cost, most of the physicians interviewed for this article advise using SGLT2 inhibitors and GLP-1 receptor agonists as second-line treatments. Others said that they would prefer to use the newer drugs as first-line therapies in select high-risk patients, but prior authorization hurdles created by insurance companies make that approach too burdensome.
“The economics of U.S. health care is stacked against many of our patients with diabetes in the current era,” Robert H. Hopkins Jr., MD, said in an interview.
Even when their insurance approves the drugs, patients still may not be able to afford the copay, explained Dr. Hopkins, professor of internal medicine and pediatrics and director of the division of general internal medicine at the University of Arkansas for Medical Sciences, Little Rock. “Sometimes patients can purchase drugs at a lower cost than the copay to purchase with the ‘drug coverage’ in their insurance plan – unfortunately, this is not the case with the newer diabetes medications we are discussing here.”
“SGLT2 inhibitors and GLP-1 agonists can cost several hundred dollars a month, and insurers often balk at paying for them. They’ll say, ‘Have you tried metformin?’ ” explained endocrinologist Victor Lawrence Roberts, MD, in a interview. “We have to work with insurance companies the best we can in a stepwise fashion.”
According to Dr. Roberts, 80% of his patients with diabetes struggle with the cost of medicine in general. “They’re either underinsured or not insured or their formulary is limited.
Douglas S. Paauw, MD, agreed in an interview that the newer drugs can be problematic on the insurance front.
“For some patients they aren’t affordable, especially for the uninsured if you can’t get them on an assistance program,” said Dr. Paauw, who is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the university.
Dr. Hopkins, who is on the Internal Medicine News board, noted that “unfortunately, the treatment of type 2 diabetes in patients who cannot achieve control with metformin, diet, weight control, and exercise is a story of the ‘haves’ and the ‘have nots.’ The ‘haves’ are those who have pharmacy benefits which make access to newer agents like SGLT2 inhibitors and GLP-1 agonists a possibility.”
“I have had very few of the ‘have nots’ who have been able to even consider these newer agents, which carry price tags of $600-$1,300 a month even with the availability of discounting coupons in the marketplace,” he added. “Most of these patients end up requiring a sulfonylurea or TZD [thiazolidinedione] as a second agent to achieve glycemic control. This makes it very difficult to achieve sufficient weight and metabolic control to avoid an eventual switch to insulin.”
Fatima Z. Syed, MD, an endocrine-trained general internist at DukeHealth in Durham, N.C., said she prescribes SGLT2 inhibitors and GLP-1 receptor agonists in combination with metformin. “I prescribe them frequently, but they are not first-line treatments,” she explained.
“Nothing replaces diet and exercise” as therapy for patients with type 2 diabetes, she added.
Neil S. Skolnik, MD, said that insurance companies were not preventing patients from using these drugs in his experience. He also provided an optimistic take on the accessibility of these drugs in the near future.
“Most insurance companies are now covering select SGLT2 inhibitors and GLP-1 receptor agonists for appropriate patients and those companies that currently do not will soon have to,” said Dr. Skolnik, who is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
“The outcomes [associated with use of the new drugs] are robust, the benefits are large, and are well worth the cost,” he added.
The side effects
While others praised these drugs for their beneficial effects, they also noted that the side effects of these drugs are serious and must be discussed with patients.
GLP-1 receptor agonists are linked to gastrointestinal symptoms, especially nausea, while SGLT2 inhibitors have been linked to kidney failure, ketoacidosis, and more. The Food and Drug Administration warned in 2018 that the SGLT2 inhibitors can cause a rare serious infection known as Fournier’s gangrene – necrotizing fasciitis of the perineum.
“We have to tell our patients to let us know right away if they get pain or swelling in the genital area,” Dr. Paauw, who is on the Internal Medicine News board, noted. “The chance that an infection could explode quickly is higher in those who take these drugs.”
Amputation risks also are associated with taking the SGLT2 inhibitor canagliflozin (Invokana). The FDA requires the manufacturer of this drug to include a black-box warning about the risk of “lower-limb amputations, most frequently of the toe and midfoot,” but also the leg. In approval trials, the risk doubled versus placebo.
These amputation risks “put a damper on some of the enthusiasm on behalf of physicians and patients ... for taking this drug,” noted Dr. Roberts, who is a professor of internal medicine at the University of Central Florida, Orlando.
While a manufacturer-funded study released last year found no link to amputations, the results weren’t powerful enough to rule out a moderately increased risk.
“[If] you are at high risk for having an amputation, we really have to take this risk very seriously,” said John B. Buse, MD, chief of the division of endocrinology at the University of North Carolina at Chapel Hill, in a presentation about the study at the 2018 annual scientific sessions of the American Diabetes Association.
The benefits
Despite these risks of adverse events, most interviewed agreed that the many benefits observed in those taking SGLT2 inhibitors or GLP-1 receptor agonists make them worth prescribing, at least to those who are able to afford them.
Both SGLT2 inhibitors and GLP-1 receptor agonists appear to have significant cardiovascular benefits. A 2019 meta-analysis and systematic review found that both drugs reduced major adverse cardiac events by about 12% (Circulation. 2019 Apr 23;139[17]:2022-31).
“They don’t cause hypoglycemia, they lower blood pressure, they don’t cause weight gain, and they might promote weight loss,” noted Dr. Paauw.
SGLT2 inhibitors also have shown signs of kidney benefits. The CREDENCE trial linked canagliflozin to a lowering of kidney disorders versus placebo (N Engl J Med. 2019 Jun 13;380[24]:2295-306). “The relative risk of the renal-specific composite of end-stage kidney disease, a doubling of the creatinine level, or death from renal causes was lower by 34% (hazard ratio, 0.66; 95% confidence interval, 0.53-0.81; P less than .001), and the relative risk of end-stage kidney disease was lower by 32% (HR, 0.68; 95% CI, 0.54-0.86; P = .002),” the trial investigators wrote.
“They showed very nicely that the drug improved the kidney function of those patients and reduced the kidney deterioration,” said Yehuda Handelsman, MD, an endocrinologist in Tarzana, Calif., who chaired the 2011 and 2015 American Association of Clinical Endocrinologists’ Comprehensive Diabetes Guidelines. The study was especially impressive, he added, because it included patients with low kidney function.
SGLT2 inhibitors’ “diuretic mechanism explains why there is a substantial reduction in heart failure hospitalizations in patients who take these drugs,” said cardiologist Marc E. Goldschmidt, MD, director of the Heart Success Program at Atlantic Health System’s Morristown (N.J.) Medical Center, in an interview. “Both the EMPA-REG Outcome and the CREDENCE trials demonstrated substantial benefit of this class of medications by showing a lower risk of cardiovascular death as well as death from any cause and a lower risk of hospitalization for heart failure."
Overall, the SGLT2 trial data have been very consistent with a benefit for cardiovascular risk reduction, particularly in regard to heart failure hospitalizations and even in potentially preventing heart failure in diabetics,” he added.
Dr. Skolnik, a columnist for Family Practice News, cited SGLT2 inhibitors and GLP-1 receptor agonists’ ability to slow renal disease progression, promote weight loss, and prevent poor cardiac outcomes.“These drugs should be used, in addition to metformin, in all patients with diabetes and vascular disease. These proven outcomes are far better than we ever were able to achieve previously and the strength of the evidence at this point is very strong,” said Dr. Skolnik. “In addition to the benefits of decreasing the development of cardiovascular disease, serious heart failure, and slowing progression of renal disease, these two classes of medication have additional benefits. Both classes help patients lose weight, which is very different from what was found with either sulfonylureas or insulin, which cause patients to gain weight. Also both the SGLT2 inhibitors and the GLP-1 RAs [receptor agonists] have a low incidence of hypoglycemia. For all these reasons, these have become important medications for us to use in primary care.”
Other recent trials offer “very powerful data” about SGLT2 inhibitors, Dr. Roberts said. That’s good news, since “our approach needs to be toward cardiovascular protection and preservation as well as managing blood sugar.”An Israeli trial, whose results were released in May 2019 at the annual meeting of the American College of Cardiology, found that, compared with other glucose-lowering drugs, taking an SGLT2 inhibitor was associated with lower risks of heart failure hospitalization and all-cause mortality (HR, 0.54; 95% CI, 0.44-0.65; P less than .001). This trial also offered a new detail: The patients gained the benefit regardless of whether their baseline left ventricular ejection fraction was preserved or reduced (J Coll Cardiol. 2019 Mar;73[9]:suppl 1). The SGLT2 inhibitors used in this trial included dapagliflozin (Farxiga) and empagliflozin (Jardiance).
In another study released this year, a subanalysis of the DECLARE-TIMI 58 trial, researchers reported that the SGLT2 inhibitor dapagliflozin reduced risks of both major adverse cardiovascular events and heart failure hospitalization in the subset of patients with type 2 diabetes and prior myocardial infarction versus controls (Circulation. 2019 May 28;139[22]:2516-27). The absolute risk reduction for major adverse cardiovascular events was 1.9% (HR, 0.81; 95% CI, 0.65-1.00; P = .046), while it was 0.6% for heart failure hospitalization (HR, 0.85; 95% CI, 0.72-1.00; P = .055).
These and other studies “speak volumes about the efficacy of managing blood sugar and addressing our biggest nemesis, which is cardiovascular disease,” Dr. Roberts said. “It’s irrefutable. The data [are] very good.”
Dr. Paauw said an SGLT2 inhibitor or GLP-1 receptor agonist is best reserved for use in select patients with cardiovascular risks and type 2 diabetes that need management beyond metformin.
For example, they might fit a 70-year-old with persistent hypertension who’s already taking a couple of blood pressure medications. “If they have another cardiovascular risk factor, the cardiovascular protection piece will be a bigger deal,” he said. Also, “it will probably help lower their blood pressure so they can avoid taking another blood pressure medicine.”
Trials of both GLP-1 receptor agonists and SGLT2 inhibitors have shown benefits “in improving [major adverse cardiac events], with the SGLT2 class showing substantial benefit in improving both heart failure and renal outcomes as well,” noted Dr. Skolnik. “It is in this context that one must address the question of whether the price of the medications are worthwhile. With such substantial benefit, there is no question in my mind that – for patients who have underlying cardiovascular illness, which includes patients with existent coronary disease, history of stroke, transient ischemic attack, or peripheral vascular disease – it is far and away worth it to prescribe these classes of medications.”
Indeed, the American Diabetes Association and the European Association for the Study of Diabetes’ most recent guidelines now call for a GLP-1 receptor agonist – instead of insulin – to be the first injectable used to treat type 2 diabetes (Diabetes Care 2018 Dec; 41[12]:2669-701).
“For the relatively small number of my patients who have been able to access and use these medications for months or longer, more have tolerated the GLP-1 agonists than SGLT2 inhibitors primarily due to urinary issues,” noted Dr. Hopkins.
Dipeptidyl peptidase–4 inhibitors are another option in patients with type 2 diabetes, but research suggests they may not be a top option for patients with cardiovascular risk. A 2018 review noted that cardiovascular outcome trials for alogliptin (Nesina), saxagliptin (Onglyza), and sitagliptin (Januvia) showed noninferiority but failed to demonstrate any superiority, compared with placebo in patients with type 2 diabetes mellitus and high cardiovascular risk (Circ Res. 2018 May 11;122[10]:1439-59).
The combination therapies
Many of the newer drugs are available as combinations with other types of diabetes drugs. In some cases, physicians create their own form of combination therapy by separately prescribing two or more diabetes drugs. Earlier this year, a study suggested the benefits of this kind of add-on therapy: Diabetes outcomes improved in patients who took the GLP-1 receptor agonist semaglutide and an SGLT2 inhibitor (Lancet Diabetes Endocrinol. 2019 Mar 1. doi: 10.1016/S2213-8587[19]30066-X).
Dr. Roberts suggested caution, however, when prescribing combination therapies. “My recommendation is always to begin with the individual medications to see if the patient tolerates the drugs and then decide which component needs to be titrated. It’s hard to titrate a combination drug, and it doesn’t leave a lot of flexibility. You never know which drug is doing what.
Dr. Handelsman said some patients may need to take three medications such as metformin, an SGLT2 inhibitor, and a GLP-1 receptor agonist.
“I don’t recommend using the combinations if you’re not familiar with the drugs ... These are relatively new pharmaceuticals, and most of us are on a learning curve as to how they fit into the armamentarium. If a drug is tolerated with a good response, you can certainly consider going to the combination tablets,” he added.
There is at least one drug that combines these three classes: The newly FDA-approved Qternmet XR, which combines dapagliflozin (an SGLT2 inhibitor), saxagliptin (a GLP-1 receptor agonist), and metformin. As of mid-June 2019, it was not yet available in the United States. Its sister drug Qtern, which combines dapagliflozin and saxagliptin, costs more than $500 a month with a free coupon, according to goodrx.com. In contrast, metformin is extremely inexpensive, costing just a few dollars a month for a common starting dose.
What about adding insulin?
“Both [SGLT2 inhibitors and GLP-1 receptor agonists] work very well with insulin,” Dr. Handelsman said. “There is a nice additive effect on the reduction of [hemoglobin] A1c. The only caution is that, although neither SGLT2 inhibitors nor GLP-1 receptor agonists cause hypoglycemia, in combination with insulin they do increase the risk of hypoglycemia. You may have to adjust the dose of insulin.”
Dr. Hopkins warned that cost becomes an even bigger issue when you add insulin into the mix.
“When insulin comes into the discussion, we are again stuck with astronomical costs which many struggle to afford,” he explained.
Indeed, the price tag on these drugs seems to be the biggest problem physicians have with them.
“The challenges in managing patients with diabetes aren’t the risks associated with the drugs. It’s dealing with their insurers,” noted Dr. Roberts.
Dr. Hopkins, Dr. Paauw, Dr. Roberts, and Dr. Syed reported no disclosures. Dr. Buse is an investigator for Johnson and Johnson. Dr. Goldschmidt is paid to speak by Novartis. Dr. Handelsman reported research grants, consulting work, and speaker honoraria from Amgen, Gilead, Lilly, Merck, Novo Nordisk, and others. Dr Skolnik reported nonfinancial support from AstraZeneca, Boehringer Ingelheim, Sanofi, and GlaxoSmithKline and personal fees from AstraZeneca, Boehringer Ingelheim, and Eli Lilly. He also serves on the advisory boards of AstraZeneca, Boehringer Ingelheim, Teva Pharmaceutical, Eli Lilly, Sanofi, Janssen Pharmaceuticals, Intarcia, Mylan, and GlaxoSmithKline.
Dr. Paauw and Dr. Skolnik are columnists for Family Practice News and Internal Medicine News.
M. Alexander Otto contributed to this report.

The options for treating type 2 diabetes without insulin have grown beyond metformin to include a long list of sodium-glucose cotransporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists that can be taken with or without metformin. These new drugs have cardiovascular and kidney benefits and help with weight loss, but they also carry risks and, according to some experts, their costs can be prohibitively expensive.
Given the medical community’s long-term experience with treating patients with metformin, and metformin’s lower cost, most of the physicians interviewed for this article advise using SGLT2 inhibitors and GLP-1 receptor agonists as second-line treatments. Others said that they would prefer to use the newer drugs as first-line therapies in select high-risk patients, but prior authorization hurdles created by insurance companies make that approach too burdensome.
“The economics of U.S. health care is stacked against many of our patients with diabetes in the current era,” Robert H. Hopkins Jr., MD, said in an interview.
Even when their insurance approves the drugs, patients still may not be able to afford the copay, explained Dr. Hopkins, professor of internal medicine and pediatrics and director of the division of general internal medicine at the University of Arkansas for Medical Sciences, Little Rock. “Sometimes patients can purchase drugs at a lower cost than the copay to purchase with the ‘drug coverage’ in their insurance plan – unfortunately, this is not the case with the newer diabetes medications we are discussing here.”
“SGLT2 inhibitors and GLP-1 agonists can cost several hundred dollars a month, and insurers often balk at paying for them. They’ll say, ‘Have you tried metformin?’ ” explained endocrinologist Victor Lawrence Roberts, MD, in a interview. “We have to work with insurance companies the best we can in a stepwise fashion.”
According to Dr. Roberts, 80% of his patients with diabetes struggle with the cost of medicine in general. “They’re either underinsured or not insured or their formulary is limited.
Douglas S. Paauw, MD, agreed in an interview that the newer drugs can be problematic on the insurance front.
“For some patients they aren’t affordable, especially for the uninsured if you can’t get them on an assistance program,” said Dr. Paauw, who is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the university.
Dr. Hopkins, who is on the Internal Medicine News board, noted that “unfortunately, the treatment of type 2 diabetes in patients who cannot achieve control with metformin, diet, weight control, and exercise is a story of the ‘haves’ and the ‘have nots.’ The ‘haves’ are those who have pharmacy benefits which make access to newer agents like SGLT2 inhibitors and GLP-1 agonists a possibility.”
“I have had very few of the ‘have nots’ who have been able to even consider these newer agents, which carry price tags of $600-$1,300 a month even with the availability of discounting coupons in the marketplace,” he added. “Most of these patients end up requiring a sulfonylurea or TZD [thiazolidinedione] as a second agent to achieve glycemic control. This makes it very difficult to achieve sufficient weight and metabolic control to avoid an eventual switch to insulin.”
Fatima Z. Syed, MD, an endocrine-trained general internist at DukeHealth in Durham, N.C., said she prescribes SGLT2 inhibitors and GLP-1 receptor agonists in combination with metformin. “I prescribe them frequently, but they are not first-line treatments,” she explained.
“Nothing replaces diet and exercise” as therapy for patients with type 2 diabetes, she added.
Neil S. Skolnik, MD, said that insurance companies were not preventing patients from using these drugs in his experience. He also provided an optimistic take on the accessibility of these drugs in the near future.
“Most insurance companies are now covering select SGLT2 inhibitors and GLP-1 receptor agonists for appropriate patients and those companies that currently do not will soon have to,” said Dr. Skolnik, who is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
“The outcomes [associated with use of the new drugs] are robust, the benefits are large, and are well worth the cost,” he added.
The side effects
While others praised these drugs for their beneficial effects, they also noted that the side effects of these drugs are serious and must be discussed with patients.
GLP-1 receptor agonists are linked to gastrointestinal symptoms, especially nausea, while SGLT2 inhibitors have been linked to kidney failure, ketoacidosis, and more. The Food and Drug Administration warned in 2018 that the SGLT2 inhibitors can cause a rare serious infection known as Fournier’s gangrene – necrotizing fasciitis of the perineum.
“We have to tell our patients to let us know right away if they get pain or swelling in the genital area,” Dr. Paauw, who is on the Internal Medicine News board, noted. “The chance that an infection could explode quickly is higher in those who take these drugs.”
Amputation risks also are associated with taking the SGLT2 inhibitor canagliflozin (Invokana). The FDA requires the manufacturer of this drug to include a black-box warning about the risk of “lower-limb amputations, most frequently of the toe and midfoot,” but also the leg. In approval trials, the risk doubled versus placebo.
These amputation risks “put a damper on some of the enthusiasm on behalf of physicians and patients ... for taking this drug,” noted Dr. Roberts, who is a professor of internal medicine at the University of Central Florida, Orlando.
While a manufacturer-funded study released last year found no link to amputations, the results weren’t powerful enough to rule out a moderately increased risk.
“[If] you are at high risk for having an amputation, we really have to take this risk very seriously,” said John B. Buse, MD, chief of the division of endocrinology at the University of North Carolina at Chapel Hill, in a presentation about the study at the 2018 annual scientific sessions of the American Diabetes Association.
The benefits
Despite these risks of adverse events, most interviewed agreed that the many benefits observed in those taking SGLT2 inhibitors or GLP-1 receptor agonists make them worth prescribing, at least to those who are able to afford them.
Both SGLT2 inhibitors and GLP-1 receptor agonists appear to have significant cardiovascular benefits. A 2019 meta-analysis and systematic review found that both drugs reduced major adverse cardiac events by about 12% (Circulation. 2019 Apr 23;139[17]:2022-31).
“They don’t cause hypoglycemia, they lower blood pressure, they don’t cause weight gain, and they might promote weight loss,” noted Dr. Paauw.
SGLT2 inhibitors also have shown signs of kidney benefits. The CREDENCE trial linked canagliflozin to a lowering of kidney disorders versus placebo (N Engl J Med. 2019 Jun 13;380[24]:2295-306). “The relative risk of the renal-specific composite of end-stage kidney disease, a doubling of the creatinine level, or death from renal causes was lower by 34% (hazard ratio, 0.66; 95% confidence interval, 0.53-0.81; P less than .001), and the relative risk of end-stage kidney disease was lower by 32% (HR, 0.68; 95% CI, 0.54-0.86; P = .002),” the trial investigators wrote.
“They showed very nicely that the drug improved the kidney function of those patients and reduced the kidney deterioration,” said Yehuda Handelsman, MD, an endocrinologist in Tarzana, Calif., who chaired the 2011 and 2015 American Association of Clinical Endocrinologists’ Comprehensive Diabetes Guidelines. The study was especially impressive, he added, because it included patients with low kidney function.
SGLT2 inhibitors’ “diuretic mechanism explains why there is a substantial reduction in heart failure hospitalizations in patients who take these drugs,” said cardiologist Marc E. Goldschmidt, MD, director of the Heart Success Program at Atlantic Health System’s Morristown (N.J.) Medical Center, in an interview. “Both the EMPA-REG Outcome and the CREDENCE trials demonstrated substantial benefit of this class of medications by showing a lower risk of cardiovascular death as well as death from any cause and a lower risk of hospitalization for heart failure."
Overall, the SGLT2 trial data have been very consistent with a benefit for cardiovascular risk reduction, particularly in regard to heart failure hospitalizations and even in potentially preventing heart failure in diabetics,” he added.
Dr. Skolnik, a columnist for Family Practice News, cited SGLT2 inhibitors and GLP-1 receptor agonists’ ability to slow renal disease progression, promote weight loss, and prevent poor cardiac outcomes.“These drugs should be used, in addition to metformin, in all patients with diabetes and vascular disease. These proven outcomes are far better than we ever were able to achieve previously and the strength of the evidence at this point is very strong,” said Dr. Skolnik. “In addition to the benefits of decreasing the development of cardiovascular disease, serious heart failure, and slowing progression of renal disease, these two classes of medication have additional benefits. Both classes help patients lose weight, which is very different from what was found with either sulfonylureas or insulin, which cause patients to gain weight. Also both the SGLT2 inhibitors and the GLP-1 RAs [receptor agonists] have a low incidence of hypoglycemia. For all these reasons, these have become important medications for us to use in primary care.”
Other recent trials offer “very powerful data” about SGLT2 inhibitors, Dr. Roberts said. That’s good news, since “our approach needs to be toward cardiovascular protection and preservation as well as managing blood sugar.”An Israeli trial, whose results were released in May 2019 at the annual meeting of the American College of Cardiology, found that, compared with other glucose-lowering drugs, taking an SGLT2 inhibitor was associated with lower risks of heart failure hospitalization and all-cause mortality (HR, 0.54; 95% CI, 0.44-0.65; P less than .001). This trial also offered a new detail: The patients gained the benefit regardless of whether their baseline left ventricular ejection fraction was preserved or reduced (J Coll Cardiol. 2019 Mar;73[9]:suppl 1). The SGLT2 inhibitors used in this trial included dapagliflozin (Farxiga) and empagliflozin (Jardiance).
In another study released this year, a subanalysis of the DECLARE-TIMI 58 trial, researchers reported that the SGLT2 inhibitor dapagliflozin reduced risks of both major adverse cardiovascular events and heart failure hospitalization in the subset of patients with type 2 diabetes and prior myocardial infarction versus controls (Circulation. 2019 May 28;139[22]:2516-27). The absolute risk reduction for major adverse cardiovascular events was 1.9% (HR, 0.81; 95% CI, 0.65-1.00; P = .046), while it was 0.6% for heart failure hospitalization (HR, 0.85; 95% CI, 0.72-1.00; P = .055).
These and other studies “speak volumes about the efficacy of managing blood sugar and addressing our biggest nemesis, which is cardiovascular disease,” Dr. Roberts said. “It’s irrefutable. The data [are] very good.”
Dr. Paauw said an SGLT2 inhibitor or GLP-1 receptor agonist is best reserved for use in select patients with cardiovascular risks and type 2 diabetes that need management beyond metformin.
For example, they might fit a 70-year-old with persistent hypertension who’s already taking a couple of blood pressure medications. “If they have another cardiovascular risk factor, the cardiovascular protection piece will be a bigger deal,” he said. Also, “it will probably help lower their blood pressure so they can avoid taking another blood pressure medicine.”
Trials of both GLP-1 receptor agonists and SGLT2 inhibitors have shown benefits “in improving [major adverse cardiac events], with the SGLT2 class showing substantial benefit in improving both heart failure and renal outcomes as well,” noted Dr. Skolnik. “It is in this context that one must address the question of whether the price of the medications are worthwhile. With such substantial benefit, there is no question in my mind that – for patients who have underlying cardiovascular illness, which includes patients with existent coronary disease, history of stroke, transient ischemic attack, or peripheral vascular disease – it is far and away worth it to prescribe these classes of medications.”
Indeed, the American Diabetes Association and the European Association for the Study of Diabetes’ most recent guidelines now call for a GLP-1 receptor agonist – instead of insulin – to be the first injectable used to treat type 2 diabetes (Diabetes Care 2018 Dec; 41[12]:2669-701).
“For the relatively small number of my patients who have been able to access and use these medications for months or longer, more have tolerated the GLP-1 agonists than SGLT2 inhibitors primarily due to urinary issues,” noted Dr. Hopkins.
Dipeptidyl peptidase–4 inhibitors are another option in patients with type 2 diabetes, but research suggests they may not be a top option for patients with cardiovascular risk. A 2018 review noted that cardiovascular outcome trials for alogliptin (Nesina), saxagliptin (Onglyza), and sitagliptin (Januvia) showed noninferiority but failed to demonstrate any superiority, compared with placebo in patients with type 2 diabetes mellitus and high cardiovascular risk (Circ Res. 2018 May 11;122[10]:1439-59).
The combination therapies
Many of the newer drugs are available as combinations with other types of diabetes drugs. In some cases, physicians create their own form of combination therapy by separately prescribing two or more diabetes drugs. Earlier this year, a study suggested the benefits of this kind of add-on therapy: Diabetes outcomes improved in patients who took the GLP-1 receptor agonist semaglutide and an SGLT2 inhibitor (Lancet Diabetes Endocrinol. 2019 Mar 1. doi: 10.1016/S2213-8587[19]30066-X).
Dr. Roberts suggested caution, however, when prescribing combination therapies. “My recommendation is always to begin with the individual medications to see if the patient tolerates the drugs and then decide which component needs to be titrated. It’s hard to titrate a combination drug, and it doesn’t leave a lot of flexibility. You never know which drug is doing what.
Dr. Handelsman said some patients may need to take three medications such as metformin, an SGLT2 inhibitor, and a GLP-1 receptor agonist.
“I don’t recommend using the combinations if you’re not familiar with the drugs ... These are relatively new pharmaceuticals, and most of us are on a learning curve as to how they fit into the armamentarium. If a drug is tolerated with a good response, you can certainly consider going to the combination tablets,” he added.
There is at least one drug that combines these three classes: The newly FDA-approved Qternmet XR, which combines dapagliflozin (an SGLT2 inhibitor), saxagliptin (a GLP-1 receptor agonist), and metformin. As of mid-June 2019, it was not yet available in the United States. Its sister drug Qtern, which combines dapagliflozin and saxagliptin, costs more than $500 a month with a free coupon, according to goodrx.com. In contrast, metformin is extremely inexpensive, costing just a few dollars a month for a common starting dose.
What about adding insulin?
“Both [SGLT2 inhibitors and GLP-1 receptor agonists] work very well with insulin,” Dr. Handelsman said. “There is a nice additive effect on the reduction of [hemoglobin] A1c. The only caution is that, although neither SGLT2 inhibitors nor GLP-1 receptor agonists cause hypoglycemia, in combination with insulin they do increase the risk of hypoglycemia. You may have to adjust the dose of insulin.”
Dr. Hopkins warned that cost becomes an even bigger issue when you add insulin into the mix.
“When insulin comes into the discussion, we are again stuck with astronomical costs which many struggle to afford,” he explained.
Indeed, the price tag on these drugs seems to be the biggest problem physicians have with them.
“The challenges in managing patients with diabetes aren’t the risks associated with the drugs. It’s dealing with their insurers,” noted Dr. Roberts.
Dr. Hopkins, Dr. Paauw, Dr. Roberts, and Dr. Syed reported no disclosures. Dr. Buse is an investigator for Johnson and Johnson. Dr. Goldschmidt is paid to speak by Novartis. Dr. Handelsman reported research grants, consulting work, and speaker honoraria from Amgen, Gilead, Lilly, Merck, Novo Nordisk, and others. Dr Skolnik reported nonfinancial support from AstraZeneca, Boehringer Ingelheim, Sanofi, and GlaxoSmithKline and personal fees from AstraZeneca, Boehringer Ingelheim, and Eli Lilly. He also serves on the advisory boards of AstraZeneca, Boehringer Ingelheim, Teva Pharmaceutical, Eli Lilly, Sanofi, Janssen Pharmaceuticals, Intarcia, Mylan, and GlaxoSmithKline.
Dr. Paauw and Dr. Skolnik are columnists for Family Practice News and Internal Medicine News.
M. Alexander Otto contributed to this report.

SC daratumumab deemed feasible for every multiple myeloma patient
CHICAGO – Subcutaneous (SC) daratumumab is noninferior to intravenous (IV) daratumumab for patients with relapsed or refractory multiple myeloma (MM), according findings from a phase 3 trial.
In the COLUMBA trial, SC daratumumab proved noninferior to IV daratumumab with regard to overall response rate and maximum trough concentration (Ctrough).
The safety profiles of the two formulations were similar, although patients who received SC daratumumab had a lower rate of infusion-related reactions. SC daratumumab also had a lower treatment burden.
“The COLUMBA study shows that [SC daratumumab] can be used in every myeloma patient [as a] single agent or, maybe in the future, in combination with the different backbones,” said Maria-Victoria Mateos, MD, PhD, of University Hospital of Salamanca (Spain).
Dr. Mateos presented results from the COLUMBA trial at the annual meeting of the American Society of Clinical Oncology.
Dr. Mateos cited a previous phase 1b study that had suggested that SC daratumumab might produce similar results as IV daratumumab (Blood. 2017;130:838) while providing a more convenient delivery method. She pointed out that infusions of IV daratumumab can last hours, while the SC formulation can be delivered in minutes.
The aim of the phase 3 COLUMBA study was to compare the IV and SC formulations head-to-head. The trial enrolled 522 patients with relapsed/refractory multiple myeloma. They were randomized to receive daratumumab SC (n = 263) or IV (n = 259).
The median patient age was 68 years (range, 33-92 years) in the IV arm and 65 years (range, 42-84 years) in the SC arm. Patients had received a median of four prior lines of therapy (range, 1-15 in the IV arm and 2-12 in the SC arm). Most patients were refractory to their last line of therapy – 85% in the IV arm and 80% in the SC arm – and most patients had standard-risk cytogenetics – 83% and 74%, respectively.
Treatment
Patients received SC daratumumab at 1,800 mg and IV daratumumab at 16 mg/kg. Both were given weekly for cycles 1-2, every 2 weeks for cycles 3-6, and every 4 weeks thereafter until disease progression.
The median duration of the first infusion was 421 minutes in the IV arm and 5 minutes in the SC arm. The median duration of the second infusion was 255 minutes and 5 minutes, respectively, and the median duration of subsequent infusions was 205 minutes and 5 minutes, respectively.
At a median follow-up of 7.46 months, 57% of patients in each arm had discontinued the study treatment. The most common reasons for discontinuation were progression – 44% of the IV arm and 43% of the SC arm – and adverse events (AEs) – 8% and 7%, respectively.
Safety
Dr. Mateos said the safety profiles of IV and SC daratumumab were comparable. However, infusion-related reactions were significantly less likely in the SC arm, occurring in 12.7% of those patients and 34.5% of patients in the IV arm (P less than .0001).
Grade 3 or higher treatment-emergent AEs occurred in 49% of patients in the IV arm and 46% of those in the SC arm. Rates of grade 5 AEs were 7% and 5%, respectively. The most common grade 3/4 AEs (in the IV and SC arms, respectively) were anemia (14% and 13%), thrombocytopenia (14% for both), neutropenia (8% and 13%), lymphopenia (6% and 5%), and hypertension (6% and 3%).
Efficacy
One of the study’s primary endpoints was overall response rate, which was 37.1% in the IV arm and 41.1% in the SC arm (relative risk, 1.11; 95% CI, 0.89-1.37; P less than .0001). This met the criteria for noninferiority, and overall response rates were comparable across all patient subgroups, Dr. Mateos noted.
The rates of complete response or stringent complete response were also comparable at 2.7% in the IV arm and 1.9% in the SC arm. Rates of very good partial response were 17.0% and 19.0%, respectively.
The study’s other primary endpoint was maximum Ctrough predose on day 1 of cycle 3. The ratio of maximum Ctrough for daratumumab SC over IV was 107.93% (90% CI, 95.74%-121.67%), which met the noninferiority criterion.
Survival outcomes were also similar between the IV and SC arms. The median progression-free survival was 6.1 months and 5.6 months, respectively (P = .9258). The rate of overall survival at 6 months was 83.0% and 87.5%, respectively (P = .6032).
Considering these results together, Dr. Mateos and colleagues concluded that SC daratumumab is noninferior to IV daratumumab.
“[SC daratumumab] has a reduced treatment burden due to a considerably shorter administration duration, and patients treated with [SC daratumumab] reported higher satisfaction with therapy,” Dr. Mateos said.
The results support the use of flat-dose 1,800-mg SC daratumumab, which is comparable with the IV formulation, she said.
The COLUMBA trial was sponsored by Janssen Research & Development. Dr. Mateos reported relationships with Amgen, Celgene, Janssen-Cilag, and Takeda.
SOURCE: Mateos MV et al. ASCO 2019, Abstract 8005.
CHICAGO – Subcutaneous (SC) daratumumab is noninferior to intravenous (IV) daratumumab for patients with relapsed or refractory multiple myeloma (MM), according findings from a phase 3 trial.
In the COLUMBA trial, SC daratumumab proved noninferior to IV daratumumab with regard to overall response rate and maximum trough concentration (Ctrough).
The safety profiles of the two formulations were similar, although patients who received SC daratumumab had a lower rate of infusion-related reactions. SC daratumumab also had a lower treatment burden.
“The COLUMBA study shows that [SC daratumumab] can be used in every myeloma patient [as a] single agent or, maybe in the future, in combination with the different backbones,” said Maria-Victoria Mateos, MD, PhD, of University Hospital of Salamanca (Spain).
Dr. Mateos presented results from the COLUMBA trial at the annual meeting of the American Society of Clinical Oncology.
Dr. Mateos cited a previous phase 1b study that had suggested that SC daratumumab might produce similar results as IV daratumumab (Blood. 2017;130:838) while providing a more convenient delivery method. She pointed out that infusions of IV daratumumab can last hours, while the SC formulation can be delivered in minutes.
The aim of the phase 3 COLUMBA study was to compare the IV and SC formulations head-to-head. The trial enrolled 522 patients with relapsed/refractory multiple myeloma. They were randomized to receive daratumumab SC (n = 263) or IV (n = 259).
The median patient age was 68 years (range, 33-92 years) in the IV arm and 65 years (range, 42-84 years) in the SC arm. Patients had received a median of four prior lines of therapy (range, 1-15 in the IV arm and 2-12 in the SC arm). Most patients were refractory to their last line of therapy – 85% in the IV arm and 80% in the SC arm – and most patients had standard-risk cytogenetics – 83% and 74%, respectively.
Treatment
Patients received SC daratumumab at 1,800 mg and IV daratumumab at 16 mg/kg. Both were given weekly for cycles 1-2, every 2 weeks for cycles 3-6, and every 4 weeks thereafter until disease progression.
The median duration of the first infusion was 421 minutes in the IV arm and 5 minutes in the SC arm. The median duration of the second infusion was 255 minutes and 5 minutes, respectively, and the median duration of subsequent infusions was 205 minutes and 5 minutes, respectively.
At a median follow-up of 7.46 months, 57% of patients in each arm had discontinued the study treatment. The most common reasons for discontinuation were progression – 44% of the IV arm and 43% of the SC arm – and adverse events (AEs) – 8% and 7%, respectively.
Safety
Dr. Mateos said the safety profiles of IV and SC daratumumab were comparable. However, infusion-related reactions were significantly less likely in the SC arm, occurring in 12.7% of those patients and 34.5% of patients in the IV arm (P less than .0001).
Grade 3 or higher treatment-emergent AEs occurred in 49% of patients in the IV arm and 46% of those in the SC arm. Rates of grade 5 AEs were 7% and 5%, respectively. The most common grade 3/4 AEs (in the IV and SC arms, respectively) were anemia (14% and 13%), thrombocytopenia (14% for both), neutropenia (8% and 13%), lymphopenia (6% and 5%), and hypertension (6% and 3%).
Efficacy
One of the study’s primary endpoints was overall response rate, which was 37.1% in the IV arm and 41.1% in the SC arm (relative risk, 1.11; 95% CI, 0.89-1.37; P less than .0001). This met the criteria for noninferiority, and overall response rates were comparable across all patient subgroups, Dr. Mateos noted.
The rates of complete response or stringent complete response were also comparable at 2.7% in the IV arm and 1.9% in the SC arm. Rates of very good partial response were 17.0% and 19.0%, respectively.
The study’s other primary endpoint was maximum Ctrough predose on day 1 of cycle 3. The ratio of maximum Ctrough for daratumumab SC over IV was 107.93% (90% CI, 95.74%-121.67%), which met the noninferiority criterion.
Survival outcomes were also similar between the IV and SC arms. The median progression-free survival was 6.1 months and 5.6 months, respectively (P = .9258). The rate of overall survival at 6 months was 83.0% and 87.5%, respectively (P = .6032).
Considering these results together, Dr. Mateos and colleagues concluded that SC daratumumab is noninferior to IV daratumumab.
“[SC daratumumab] has a reduced treatment burden due to a considerably shorter administration duration, and patients treated with [SC daratumumab] reported higher satisfaction with therapy,” Dr. Mateos said.
The results support the use of flat-dose 1,800-mg SC daratumumab, which is comparable with the IV formulation, she said.
The COLUMBA trial was sponsored by Janssen Research & Development. Dr. Mateos reported relationships with Amgen, Celgene, Janssen-Cilag, and Takeda.
SOURCE: Mateos MV et al. ASCO 2019, Abstract 8005.
CHICAGO – Subcutaneous (SC) daratumumab is noninferior to intravenous (IV) daratumumab for patients with relapsed or refractory multiple myeloma (MM), according findings from a phase 3 trial.
In the COLUMBA trial, SC daratumumab proved noninferior to IV daratumumab with regard to overall response rate and maximum trough concentration (Ctrough).
The safety profiles of the two formulations were similar, although patients who received SC daratumumab had a lower rate of infusion-related reactions. SC daratumumab also had a lower treatment burden.
“The COLUMBA study shows that [SC daratumumab] can be used in every myeloma patient [as a] single agent or, maybe in the future, in combination with the different backbones,” said Maria-Victoria Mateos, MD, PhD, of University Hospital of Salamanca (Spain).
Dr. Mateos presented results from the COLUMBA trial at the annual meeting of the American Society of Clinical Oncology.
Dr. Mateos cited a previous phase 1b study that had suggested that SC daratumumab might produce similar results as IV daratumumab (Blood. 2017;130:838) while providing a more convenient delivery method. She pointed out that infusions of IV daratumumab can last hours, while the SC formulation can be delivered in minutes.
The aim of the phase 3 COLUMBA study was to compare the IV and SC formulations head-to-head. The trial enrolled 522 patients with relapsed/refractory multiple myeloma. They were randomized to receive daratumumab SC (n = 263) or IV (n = 259).
The median patient age was 68 years (range, 33-92 years) in the IV arm and 65 years (range, 42-84 years) in the SC arm. Patients had received a median of four prior lines of therapy (range, 1-15 in the IV arm and 2-12 in the SC arm). Most patients were refractory to their last line of therapy – 85% in the IV arm and 80% in the SC arm – and most patients had standard-risk cytogenetics – 83% and 74%, respectively.
Treatment
Patients received SC daratumumab at 1,800 mg and IV daratumumab at 16 mg/kg. Both were given weekly for cycles 1-2, every 2 weeks for cycles 3-6, and every 4 weeks thereafter until disease progression.
The median duration of the first infusion was 421 minutes in the IV arm and 5 minutes in the SC arm. The median duration of the second infusion was 255 minutes and 5 minutes, respectively, and the median duration of subsequent infusions was 205 minutes and 5 minutes, respectively.
At a median follow-up of 7.46 months, 57% of patients in each arm had discontinued the study treatment. The most common reasons for discontinuation were progression – 44% of the IV arm and 43% of the SC arm – and adverse events (AEs) – 8% and 7%, respectively.
Safety
Dr. Mateos said the safety profiles of IV and SC daratumumab were comparable. However, infusion-related reactions were significantly less likely in the SC arm, occurring in 12.7% of those patients and 34.5% of patients in the IV arm (P less than .0001).
Grade 3 or higher treatment-emergent AEs occurred in 49% of patients in the IV arm and 46% of those in the SC arm. Rates of grade 5 AEs were 7% and 5%, respectively. The most common grade 3/4 AEs (in the IV and SC arms, respectively) were anemia (14% and 13%), thrombocytopenia (14% for both), neutropenia (8% and 13%), lymphopenia (6% and 5%), and hypertension (6% and 3%).
Efficacy
One of the study’s primary endpoints was overall response rate, which was 37.1% in the IV arm and 41.1% in the SC arm (relative risk, 1.11; 95% CI, 0.89-1.37; P less than .0001). This met the criteria for noninferiority, and overall response rates were comparable across all patient subgroups, Dr. Mateos noted.
The rates of complete response or stringent complete response were also comparable at 2.7% in the IV arm and 1.9% in the SC arm. Rates of very good partial response were 17.0% and 19.0%, respectively.
The study’s other primary endpoint was maximum Ctrough predose on day 1 of cycle 3. The ratio of maximum Ctrough for daratumumab SC over IV was 107.93% (90% CI, 95.74%-121.67%), which met the noninferiority criterion.
Survival outcomes were also similar between the IV and SC arms. The median progression-free survival was 6.1 months and 5.6 months, respectively (P = .9258). The rate of overall survival at 6 months was 83.0% and 87.5%, respectively (P = .6032).
Considering these results together, Dr. Mateos and colleagues concluded that SC daratumumab is noninferior to IV daratumumab.
“[SC daratumumab] has a reduced treatment burden due to a considerably shorter administration duration, and patients treated with [SC daratumumab] reported higher satisfaction with therapy,” Dr. Mateos said.
The results support the use of flat-dose 1,800-mg SC daratumumab, which is comparable with the IV formulation, she said.
The COLUMBA trial was sponsored by Janssen Research & Development. Dr. Mateos reported relationships with Amgen, Celgene, Janssen-Cilag, and Takeda.
SOURCE: Mateos MV et al. ASCO 2019, Abstract 8005.
REPORTING FROM ASCO 2019
Ibrutinib tops chlorambucil against CLL
AMSTERDAM – After 5 years, a large majority of patients with chronic lymphocytic leukemia treated with front-line ibrutinib (Imbruvica) have not experienced disease progression, and the median progression-free survival has still not been reached, long-term follow-up from the RESONATE-2 shows.
The 5-year estimated progression-free survival (PFS) rates were 70% for patients who had been randomized to receive ibrutinib monotherapy, compared with 12% for patients randomized to chlorambucil, reported Alessandra Tedeschi, MD, from Azienda Ospedaliera Niguarda Ca’ Granda in Milan.
Ibrutinib was also associated with a halving of risk for death, compared with chlorambucil, she said at the annual congress of the European Hematology Association.
“Importantly, the rate of progression during ibrutinib treatment was very low; only 8 – that is, 6% of patients” – experienced disease progression while receiving ibrutinib, she noted.
In the RESONATE-2 (PCYC-1115) trial, investigators enrolled 269 adults aged 65 years and older with previously untreated CLL/small lymphocytic lymphoma (SLL). Patients at the younger end of the age range (65-69 years) had to have comorbidities that would have made them ineligible for the FCR chemotherapy regimen (fludarabine, cyclophosphamide, and rituximab). Additionally, patients with the deleterious 17p deletion were excluded.
Patients were stratified by performance status and Rai stage and then randomized to receive either ibrutinib 420 mg once daily until disease progression or unacceptable toxicity (136 patients) or chlorambucil 0.5 mg/kg to a maximum of 0.8 mg/kg for up to 12 cycles (133 patients). The trial also had an extension study for patients who had disease progression as confirmed by an independent review committee or who had completed the RESONATE-2 trial. Of the 133 patients in the chlorambucil arm, 76 (57% of the intention-to-treat population) were crossed over to ibrutinib following disease progression.
The median duration of ibrutinib treatment was 57.1 months, with 73% of patients being on it for more than 3 years, 65% for more than 4 years, and 27% for more than 5 years. As of the data cutoff, 79 patients (58%) were continuing with ibrutinib on study.
At 5 years, 70% of ibrutinib-treated patients and 12% of chlorambucil-treated patients were estimated to be progression-free and alive (hazard ratio for PFS with ibrutinib 0.146 (95% confidence interval, 0.10-0.22). The benefit of ibrutinib was consistent for patients with high-risk genomic features, including the 11q deletion and unmutated immunoglobulin heavy-chain variable genes.
Estimated 5-year overall survival was also better with ibrutinib, at 83% vs. 68% (hazard ratio, 0.45; 95% CI, 0.266-0.761).
The most common grade 3 or greater adverse events occurring with ibrutinib were neutropenia (13%), pneumonia (12%), hypertension (8%), anemia (7%), hyponatremia (6%), atrial fibrillation (5%), and cataract (5%). The rates of most adverse events decreased over time, and dose reductions because of adverse events also diminished over time, from 5% of patients in the first year down to zero in years 4 through 5.
Patients responded to subsequent CLL therapies following ibrutinib discontinuation, including chemoimmunotherapy and other kinase inhibitors, Dr. Tedeschi said.
The trial was sponsored by Pharmacyclics with collaboration from Janssen Research & Development. Dr. Tedeschi reported advisory board activities with Janssen, AbbVie, and BeiGene.
SOURCE: Tedeschi A et al. EHA Congress, Abstract S107.
AMSTERDAM – After 5 years, a large majority of patients with chronic lymphocytic leukemia treated with front-line ibrutinib (Imbruvica) have not experienced disease progression, and the median progression-free survival has still not been reached, long-term follow-up from the RESONATE-2 shows.
The 5-year estimated progression-free survival (PFS) rates were 70% for patients who had been randomized to receive ibrutinib monotherapy, compared with 12% for patients randomized to chlorambucil, reported Alessandra Tedeschi, MD, from Azienda Ospedaliera Niguarda Ca’ Granda in Milan.
Ibrutinib was also associated with a halving of risk for death, compared with chlorambucil, she said at the annual congress of the European Hematology Association.
“Importantly, the rate of progression during ibrutinib treatment was very low; only 8 – that is, 6% of patients” – experienced disease progression while receiving ibrutinib, she noted.
In the RESONATE-2 (PCYC-1115) trial, investigators enrolled 269 adults aged 65 years and older with previously untreated CLL/small lymphocytic lymphoma (SLL). Patients at the younger end of the age range (65-69 years) had to have comorbidities that would have made them ineligible for the FCR chemotherapy regimen (fludarabine, cyclophosphamide, and rituximab). Additionally, patients with the deleterious 17p deletion were excluded.
Patients were stratified by performance status and Rai stage and then randomized to receive either ibrutinib 420 mg once daily until disease progression or unacceptable toxicity (136 patients) or chlorambucil 0.5 mg/kg to a maximum of 0.8 mg/kg for up to 12 cycles (133 patients). The trial also had an extension study for patients who had disease progression as confirmed by an independent review committee or who had completed the RESONATE-2 trial. Of the 133 patients in the chlorambucil arm, 76 (57% of the intention-to-treat population) were crossed over to ibrutinib following disease progression.
The median duration of ibrutinib treatment was 57.1 months, with 73% of patients being on it for more than 3 years, 65% for more than 4 years, and 27% for more than 5 years. As of the data cutoff, 79 patients (58%) were continuing with ibrutinib on study.
At 5 years, 70% of ibrutinib-treated patients and 12% of chlorambucil-treated patients were estimated to be progression-free and alive (hazard ratio for PFS with ibrutinib 0.146 (95% confidence interval, 0.10-0.22). The benefit of ibrutinib was consistent for patients with high-risk genomic features, including the 11q deletion and unmutated immunoglobulin heavy-chain variable genes.
Estimated 5-year overall survival was also better with ibrutinib, at 83% vs. 68% (hazard ratio, 0.45; 95% CI, 0.266-0.761).
The most common grade 3 or greater adverse events occurring with ibrutinib were neutropenia (13%), pneumonia (12%), hypertension (8%), anemia (7%), hyponatremia (6%), atrial fibrillation (5%), and cataract (5%). The rates of most adverse events decreased over time, and dose reductions because of adverse events also diminished over time, from 5% of patients in the first year down to zero in years 4 through 5.
Patients responded to subsequent CLL therapies following ibrutinib discontinuation, including chemoimmunotherapy and other kinase inhibitors, Dr. Tedeschi said.
The trial was sponsored by Pharmacyclics with collaboration from Janssen Research & Development. Dr. Tedeschi reported advisory board activities with Janssen, AbbVie, and BeiGene.
SOURCE: Tedeschi A et al. EHA Congress, Abstract S107.
AMSTERDAM – After 5 years, a large majority of patients with chronic lymphocytic leukemia treated with front-line ibrutinib (Imbruvica) have not experienced disease progression, and the median progression-free survival has still not been reached, long-term follow-up from the RESONATE-2 shows.
The 5-year estimated progression-free survival (PFS) rates were 70% for patients who had been randomized to receive ibrutinib monotherapy, compared with 12% for patients randomized to chlorambucil, reported Alessandra Tedeschi, MD, from Azienda Ospedaliera Niguarda Ca’ Granda in Milan.
Ibrutinib was also associated with a halving of risk for death, compared with chlorambucil, she said at the annual congress of the European Hematology Association.
“Importantly, the rate of progression during ibrutinib treatment was very low; only 8 – that is, 6% of patients” – experienced disease progression while receiving ibrutinib, she noted.
In the RESONATE-2 (PCYC-1115) trial, investigators enrolled 269 adults aged 65 years and older with previously untreated CLL/small lymphocytic lymphoma (SLL). Patients at the younger end of the age range (65-69 years) had to have comorbidities that would have made them ineligible for the FCR chemotherapy regimen (fludarabine, cyclophosphamide, and rituximab). Additionally, patients with the deleterious 17p deletion were excluded.
Patients were stratified by performance status and Rai stage and then randomized to receive either ibrutinib 420 mg once daily until disease progression or unacceptable toxicity (136 patients) or chlorambucil 0.5 mg/kg to a maximum of 0.8 mg/kg for up to 12 cycles (133 patients). The trial also had an extension study for patients who had disease progression as confirmed by an independent review committee or who had completed the RESONATE-2 trial. Of the 133 patients in the chlorambucil arm, 76 (57% of the intention-to-treat population) were crossed over to ibrutinib following disease progression.
The median duration of ibrutinib treatment was 57.1 months, with 73% of patients being on it for more than 3 years, 65% for more than 4 years, and 27% for more than 5 years. As of the data cutoff, 79 patients (58%) were continuing with ibrutinib on study.
At 5 years, 70% of ibrutinib-treated patients and 12% of chlorambucil-treated patients were estimated to be progression-free and alive (hazard ratio for PFS with ibrutinib 0.146 (95% confidence interval, 0.10-0.22). The benefit of ibrutinib was consistent for patients with high-risk genomic features, including the 11q deletion and unmutated immunoglobulin heavy-chain variable genes.
Estimated 5-year overall survival was also better with ibrutinib, at 83% vs. 68% (hazard ratio, 0.45; 95% CI, 0.266-0.761).
The most common grade 3 or greater adverse events occurring with ibrutinib were neutropenia (13%), pneumonia (12%), hypertension (8%), anemia (7%), hyponatremia (6%), atrial fibrillation (5%), and cataract (5%). The rates of most adverse events decreased over time, and dose reductions because of adverse events also diminished over time, from 5% of patients in the first year down to zero in years 4 through 5.
Patients responded to subsequent CLL therapies following ibrutinib discontinuation, including chemoimmunotherapy and other kinase inhibitors, Dr. Tedeschi said.
The trial was sponsored by Pharmacyclics with collaboration from Janssen Research & Development. Dr. Tedeschi reported advisory board activities with Janssen, AbbVie, and BeiGene.
SOURCE: Tedeschi A et al. EHA Congress, Abstract S107.
REPORTING FROM EHA CONGRESS
R2-CHOP doesn’t improve survival in DLBCL
LUGANO, Switzerland – Adding the immunomodulator lenalidomide (Revlimid) to standard chemotherapy for patients with newly diagnosed ABC-type diffuse large B-cell lymphoma (DLBCL) – the so-called R2-CHOP regimen – did not significantly improve either progression-free or overall survival, compared with R-CHOP alone, investigators in the phase 3 ROBUST trial found.
Among 570 patients with activated B-cell (ABC) type DLBCL followed for a median of 27.1 months, median progression-free survival (PFS) – the primary endpoint – had not been reached either for patients randomized to R-CHOP (rituximab, cyclophosphamide, vincristine, doxorubicin and prednisone) plus lenalidomide (R2-CHOP) or R-CHOP plus placebo.
The 1-year and 2-year PFS rate with R2-CHOP was 77%, compared with 75% for R-CHOP, and 2-year PFS rates were 67% and 64%, respectively, and neither comparison was statistically significant reported Umberto Vitolo, MD, from the Citta della Salute e della Scienzia Hospital and University in Turin, Italy.“The future direction is that promising preclinical data with next-generation immunomodulatory agents will be evaluated in future DLBCL clinical trials,” he said at the International Conference on Malignant Lymphoma.
The ROBUST trial is the latest in a long line of studies that failed to show improvement in outcomes with the addition of a novel agent to R-CHOP.
The rationale for adding lenalidomide to R-CHOP came from in-vitro studies showing antiproliferative and immunomodulatory action of lenalidomide against DLBCL, as well as two proof-of-concept clinical studies (REAL07 and MC078E) indicating efficacy against non–germinal center–like B (GCB) type DLBCL, Dr. Vitolo said.
In the ROBUST trial, investigators across 257 global sites enrolled 570 patients with ABC-type DLBCL, stratified them by International Prognostic Index (IPI) score (2 vs. 3 or greater), bulky disease (less than 7 cm vs. 7 cm or more), and age (younger than 65 years vs. 65 years and older) and randomly assigned them to receive R-CHOP with either oral lenalidomide 15 mg or placebo daily on days 1-14 of each 21-day cycle for six cycles.
All patients were required to have neutropenia prophylaxis according to local practice, with either a granulocyte- or granulocyte-macrophage colony-stimulating factor.
The efficacy analysis was by intention-to-treat and included 285 patients in each arm.
The investigators found no significant difference in the primary endpoint of PFS. Overall response rates (ORR) and complete response (CR) rates were high in both arms. The ORR was 91% in each arm, and the CR rate was 69% for R2-CHOP and 65% for R-CHOP.
Event-free survival (EFS) – a composite of first disease progression, death, or relapse after CR or start of second-line therapy – also did not differ significantly between the groups. The 1-year and 2-year EFS rates were 68% vs. 71% and 59% vs. 61%, respectively. The median EFS was not reached in either arm.
Similarly, overall survival did not differ between the groups. At 48 months of follow-up, 57 patients in the R2-CHOP arm and 62 patients in the R-CHOP arm had died. Respective 1- and 2-year overall survival rates were 91% vs. 90%, and 79% vs. 80%.
In the safety analysis, which included 283 patients in the R2-CHOP arm and 284 in the placebo/R-CHOP arm, there were no new safety signals observed. In all, 78% of patients in the lenalidomide arm and 71% in the placebo arm had at least one grade 3 or greater adverse events. The most common adverse events were hematologic, including neutropenia, febrile neutropenia, anemia, thrombocytopenia, and leukopenia.
The ROBUST study was funded by Celgene. Dr. Vitolo reported consulting and speaker’s bureau fees and research funding from the company.
SOURCE: Vitolo U et al. 15-ICML, Abstract 005.
LUGANO, Switzerland – Adding the immunomodulator lenalidomide (Revlimid) to standard chemotherapy for patients with newly diagnosed ABC-type diffuse large B-cell lymphoma (DLBCL) – the so-called R2-CHOP regimen – did not significantly improve either progression-free or overall survival, compared with R-CHOP alone, investigators in the phase 3 ROBUST trial found.
Among 570 patients with activated B-cell (ABC) type DLBCL followed for a median of 27.1 months, median progression-free survival (PFS) – the primary endpoint – had not been reached either for patients randomized to R-CHOP (rituximab, cyclophosphamide, vincristine, doxorubicin and prednisone) plus lenalidomide (R2-CHOP) or R-CHOP plus placebo.
The 1-year and 2-year PFS rate with R2-CHOP was 77%, compared with 75% for R-CHOP, and 2-year PFS rates were 67% and 64%, respectively, and neither comparison was statistically significant reported Umberto Vitolo, MD, from the Citta della Salute e della Scienzia Hospital and University in Turin, Italy.“The future direction is that promising preclinical data with next-generation immunomodulatory agents will be evaluated in future DLBCL clinical trials,” he said at the International Conference on Malignant Lymphoma.
The ROBUST trial is the latest in a long line of studies that failed to show improvement in outcomes with the addition of a novel agent to R-CHOP.
The rationale for adding lenalidomide to R-CHOP came from in-vitro studies showing antiproliferative and immunomodulatory action of lenalidomide against DLBCL, as well as two proof-of-concept clinical studies (REAL07 and MC078E) indicating efficacy against non–germinal center–like B (GCB) type DLBCL, Dr. Vitolo said.
In the ROBUST trial, investigators across 257 global sites enrolled 570 patients with ABC-type DLBCL, stratified them by International Prognostic Index (IPI) score (2 vs. 3 or greater), bulky disease (less than 7 cm vs. 7 cm or more), and age (younger than 65 years vs. 65 years and older) and randomly assigned them to receive R-CHOP with either oral lenalidomide 15 mg or placebo daily on days 1-14 of each 21-day cycle for six cycles.
All patients were required to have neutropenia prophylaxis according to local practice, with either a granulocyte- or granulocyte-macrophage colony-stimulating factor.
The efficacy analysis was by intention-to-treat and included 285 patients in each arm.
The investigators found no significant difference in the primary endpoint of PFS. Overall response rates (ORR) and complete response (CR) rates were high in both arms. The ORR was 91% in each arm, and the CR rate was 69% for R2-CHOP and 65% for R-CHOP.
Event-free survival (EFS) – a composite of first disease progression, death, or relapse after CR or start of second-line therapy – also did not differ significantly between the groups. The 1-year and 2-year EFS rates were 68% vs. 71% and 59% vs. 61%, respectively. The median EFS was not reached in either arm.
Similarly, overall survival did not differ between the groups. At 48 months of follow-up, 57 patients in the R2-CHOP arm and 62 patients in the R-CHOP arm had died. Respective 1- and 2-year overall survival rates were 91% vs. 90%, and 79% vs. 80%.
In the safety analysis, which included 283 patients in the R2-CHOP arm and 284 in the placebo/R-CHOP arm, there were no new safety signals observed. In all, 78% of patients in the lenalidomide arm and 71% in the placebo arm had at least one grade 3 or greater adverse events. The most common adverse events were hematologic, including neutropenia, febrile neutropenia, anemia, thrombocytopenia, and leukopenia.
The ROBUST study was funded by Celgene. Dr. Vitolo reported consulting and speaker’s bureau fees and research funding from the company.
SOURCE: Vitolo U et al. 15-ICML, Abstract 005.
LUGANO, Switzerland – Adding the immunomodulator lenalidomide (Revlimid) to standard chemotherapy for patients with newly diagnosed ABC-type diffuse large B-cell lymphoma (DLBCL) – the so-called R2-CHOP regimen – did not significantly improve either progression-free or overall survival, compared with R-CHOP alone, investigators in the phase 3 ROBUST trial found.
Among 570 patients with activated B-cell (ABC) type DLBCL followed for a median of 27.1 months, median progression-free survival (PFS) – the primary endpoint – had not been reached either for patients randomized to R-CHOP (rituximab, cyclophosphamide, vincristine, doxorubicin and prednisone) plus lenalidomide (R2-CHOP) or R-CHOP plus placebo.
The 1-year and 2-year PFS rate with R2-CHOP was 77%, compared with 75% for R-CHOP, and 2-year PFS rates were 67% and 64%, respectively, and neither comparison was statistically significant reported Umberto Vitolo, MD, from the Citta della Salute e della Scienzia Hospital and University in Turin, Italy.“The future direction is that promising preclinical data with next-generation immunomodulatory agents will be evaluated in future DLBCL clinical trials,” he said at the International Conference on Malignant Lymphoma.
The ROBUST trial is the latest in a long line of studies that failed to show improvement in outcomes with the addition of a novel agent to R-CHOP.
The rationale for adding lenalidomide to R-CHOP came from in-vitro studies showing antiproliferative and immunomodulatory action of lenalidomide against DLBCL, as well as two proof-of-concept clinical studies (REAL07 and MC078E) indicating efficacy against non–germinal center–like B (GCB) type DLBCL, Dr. Vitolo said.
In the ROBUST trial, investigators across 257 global sites enrolled 570 patients with ABC-type DLBCL, stratified them by International Prognostic Index (IPI) score (2 vs. 3 or greater), bulky disease (less than 7 cm vs. 7 cm or more), and age (younger than 65 years vs. 65 years and older) and randomly assigned them to receive R-CHOP with either oral lenalidomide 15 mg or placebo daily on days 1-14 of each 21-day cycle for six cycles.
All patients were required to have neutropenia prophylaxis according to local practice, with either a granulocyte- or granulocyte-macrophage colony-stimulating factor.
The efficacy analysis was by intention-to-treat and included 285 patients in each arm.
The investigators found no significant difference in the primary endpoint of PFS. Overall response rates (ORR) and complete response (CR) rates were high in both arms. The ORR was 91% in each arm, and the CR rate was 69% for R2-CHOP and 65% for R-CHOP.
Event-free survival (EFS) – a composite of first disease progression, death, or relapse after CR or start of second-line therapy – also did not differ significantly between the groups. The 1-year and 2-year EFS rates were 68% vs. 71% and 59% vs. 61%, respectively. The median EFS was not reached in either arm.
Similarly, overall survival did not differ between the groups. At 48 months of follow-up, 57 patients in the R2-CHOP arm and 62 patients in the R-CHOP arm had died. Respective 1- and 2-year overall survival rates were 91% vs. 90%, and 79% vs. 80%.
In the safety analysis, which included 283 patients in the R2-CHOP arm and 284 in the placebo/R-CHOP arm, there were no new safety signals observed. In all, 78% of patients in the lenalidomide arm and 71% in the placebo arm had at least one grade 3 or greater adverse events. The most common adverse events were hematologic, including neutropenia, febrile neutropenia, anemia, thrombocytopenia, and leukopenia.
The ROBUST study was funded by Celgene. Dr. Vitolo reported consulting and speaker’s bureau fees and research funding from the company.
SOURCE: Vitolo U et al. 15-ICML, Abstract 005.
REPORTING FROM 15-ICML
Sicker COPD patients may be more likely to initiate arformoterol
according to study to identify predictors of its use. In addition to being sicker, being treated by a pulmonologist rather than a primary care physician and being white were factors that increased a patient’s likelihood of receiving nebulized arformoterol.
Patients less likely to receive the nebulized version of this long-acting beta2 adrenoreceptor agonist (LABA) were African Americans, patients with psychiatric comorbidities, and patients eligible for both Medicare and Medicaid.
“Studies have shown that 40% to 71% of Medicare beneficiaries receive no maintenance treatment for COPD. Although a recent longitudinal study on Medicare populations reported that use of maintenance medications has been improving, in general, it is recognized that Medicare beneficiaries with COPD remain undertreated,” Todd P. Gilmer, PhD, from the department of family medicine and public health at the University of California, San Diego, and colleagues wrote.
The investigators identified patients with COPD using Medicare administrative data; of these patients, 11,887 were arformoterol users, and 450,178 were control patients who did not use arformoterol. Patients were included in the study if they had at least one claim for COPD medication and were continuously enrolled in Medicare Parts A, B, and D. The cohort consisted of mostly white women aged 70 years or older, and 47% were dual-eligible to receive both Medicare and Medicaid benefits. A subgroup of 1,778 arformoterol users were also identified for analysis who were hospitalized and discharged within 30 days of using arformoterol, as well as a subgroup of 21,910 control patients with hospitalizations.
The researchers found COPD-related hospitalization (odds ratio, 1.31; 95% confidence interval, 1.24-1.39; P less than .001), exacerbation (OR, 1.33; 95% CI, 1.26-1.41; P less than .001), use of a systemic corticosteroid (OR, 1.50; 95% CI, 1.43-1.57; P less than .001) or methylxanthine (OR, 1.37; 95% CI, 1.28-1.47; P less than .001), use of oxygen therapy (OR, 2.01; 95% CI, 1.93-2.09; P less than .001), pulmonologist care (OR, 1.40; 95% CI, 1.34-1.46; P less than .001), and respiratory therapist care (OR, 1.23; 95% CI, 1.11-1.36; P less than .001) strongly predicted arformoterol use, while racial/ethnic minority status, psychiatric comorbidity (OR, 0.65; 95% CI, 0.56-0.76; P less than .001), acquired immune deficiency syndrome (OR, 0.69; 95% CI, 0.52-0.94; P less than .01), and dual-eligibility for Medicare and Medicaid (OR, 0.73; 95% CI, 0.70-0.77; P less than .001) lowered the odds of arformoterol use (P less than .001). In the subgroup of patients with hospitalizations, COPD-related admission (OR, 1.83; 95% CI, 1.55-2.14; P less than .001), exacerbation (OR, 2.62; 95% CI, 1.88-3.63; P less than .001)m and inpatient care from a pulmonologist (OR, 1.78; 95% CI, 1.58-2.01; P less than .001) predicted arformoterol use.
“Given the results of this study, increasing access to nebulized maintenance therapy is warranted for select populations with COPD including racial/ethnic minorities, the dual-eligible, and those with certain comorbidities, such as psychiatric disorders,” Dr. Gilmer and colleagues wrote in their study. “Future studies are needed to explore the optimal time to initiate nebulized maintenance therapy, and the potential differential impact of early versus late initiation on patient outcomes.”
Researchers noted that, although their results may seem initially counterintuitive given that COPD has a higher prevalence in men, 56% of the beneficiaries in their Medicare data were women who were 65 years or older, and the results are consistent with other studies that show similar gender distribution findings for maintenance treatment patterns among COPD patients receiving Medicare.
“Since most Medicare beneficiaries with COPD are older than 70 years of age, the higher percentage of women than men in our two cohorts can be explained by the age distributions that ensued as a result of applying our various inclusion and exclusion criteria,” they said.
This study was funded by Sunovian. Dr. Gilmer and one coauthor are paid employees of University of California San Diego, which receives research funding from Advance Health Solutions. Another coauthor is an advisory board member for Advance Health Solutions and a consultant for GlaxoSmithKline, Boehringer-Ingelheim, Astra Zeneca, Novartis, and Pulmonix. Two other coauthors are paid employees of Advance Health Solutions, and another is a paid employee of Sunovion.
SOURCE: Gilmer TP et al. COPD. 2019 Jun 19. doi: 10.1080/15412555.2019.1618256.
according to study to identify predictors of its use. In addition to being sicker, being treated by a pulmonologist rather than a primary care physician and being white were factors that increased a patient’s likelihood of receiving nebulized arformoterol.
Patients less likely to receive the nebulized version of this long-acting beta2 adrenoreceptor agonist (LABA) were African Americans, patients with psychiatric comorbidities, and patients eligible for both Medicare and Medicaid.
“Studies have shown that 40% to 71% of Medicare beneficiaries receive no maintenance treatment for COPD. Although a recent longitudinal study on Medicare populations reported that use of maintenance medications has been improving, in general, it is recognized that Medicare beneficiaries with COPD remain undertreated,” Todd P. Gilmer, PhD, from the department of family medicine and public health at the University of California, San Diego, and colleagues wrote.
The investigators identified patients with COPD using Medicare administrative data; of these patients, 11,887 were arformoterol users, and 450,178 were control patients who did not use arformoterol. Patients were included in the study if they had at least one claim for COPD medication and were continuously enrolled in Medicare Parts A, B, and D. The cohort consisted of mostly white women aged 70 years or older, and 47% were dual-eligible to receive both Medicare and Medicaid benefits. A subgroup of 1,778 arformoterol users were also identified for analysis who were hospitalized and discharged within 30 days of using arformoterol, as well as a subgroup of 21,910 control patients with hospitalizations.
The researchers found COPD-related hospitalization (odds ratio, 1.31; 95% confidence interval, 1.24-1.39; P less than .001), exacerbation (OR, 1.33; 95% CI, 1.26-1.41; P less than .001), use of a systemic corticosteroid (OR, 1.50; 95% CI, 1.43-1.57; P less than .001) or methylxanthine (OR, 1.37; 95% CI, 1.28-1.47; P less than .001), use of oxygen therapy (OR, 2.01; 95% CI, 1.93-2.09; P less than .001), pulmonologist care (OR, 1.40; 95% CI, 1.34-1.46; P less than .001), and respiratory therapist care (OR, 1.23; 95% CI, 1.11-1.36; P less than .001) strongly predicted arformoterol use, while racial/ethnic minority status, psychiatric comorbidity (OR, 0.65; 95% CI, 0.56-0.76; P less than .001), acquired immune deficiency syndrome (OR, 0.69; 95% CI, 0.52-0.94; P less than .01), and dual-eligibility for Medicare and Medicaid (OR, 0.73; 95% CI, 0.70-0.77; P less than .001) lowered the odds of arformoterol use (P less than .001). In the subgroup of patients with hospitalizations, COPD-related admission (OR, 1.83; 95% CI, 1.55-2.14; P less than .001), exacerbation (OR, 2.62; 95% CI, 1.88-3.63; P less than .001)m and inpatient care from a pulmonologist (OR, 1.78; 95% CI, 1.58-2.01; P less than .001) predicted arformoterol use.
“Given the results of this study, increasing access to nebulized maintenance therapy is warranted for select populations with COPD including racial/ethnic minorities, the dual-eligible, and those with certain comorbidities, such as psychiatric disorders,” Dr. Gilmer and colleagues wrote in their study. “Future studies are needed to explore the optimal time to initiate nebulized maintenance therapy, and the potential differential impact of early versus late initiation on patient outcomes.”
Researchers noted that, although their results may seem initially counterintuitive given that COPD has a higher prevalence in men, 56% of the beneficiaries in their Medicare data were women who were 65 years or older, and the results are consistent with other studies that show similar gender distribution findings for maintenance treatment patterns among COPD patients receiving Medicare.
“Since most Medicare beneficiaries with COPD are older than 70 years of age, the higher percentage of women than men in our two cohorts can be explained by the age distributions that ensued as a result of applying our various inclusion and exclusion criteria,” they said.
This study was funded by Sunovian. Dr. Gilmer and one coauthor are paid employees of University of California San Diego, which receives research funding from Advance Health Solutions. Another coauthor is an advisory board member for Advance Health Solutions and a consultant for GlaxoSmithKline, Boehringer-Ingelheim, Astra Zeneca, Novartis, and Pulmonix. Two other coauthors are paid employees of Advance Health Solutions, and another is a paid employee of Sunovion.
SOURCE: Gilmer TP et al. COPD. 2019 Jun 19. doi: 10.1080/15412555.2019.1618256.
according to study to identify predictors of its use. In addition to being sicker, being treated by a pulmonologist rather than a primary care physician and being white were factors that increased a patient’s likelihood of receiving nebulized arformoterol.
Patients less likely to receive the nebulized version of this long-acting beta2 adrenoreceptor agonist (LABA) were African Americans, patients with psychiatric comorbidities, and patients eligible for both Medicare and Medicaid.
“Studies have shown that 40% to 71% of Medicare beneficiaries receive no maintenance treatment for COPD. Although a recent longitudinal study on Medicare populations reported that use of maintenance medications has been improving, in general, it is recognized that Medicare beneficiaries with COPD remain undertreated,” Todd P. Gilmer, PhD, from the department of family medicine and public health at the University of California, San Diego, and colleagues wrote.
The investigators identified patients with COPD using Medicare administrative data; of these patients, 11,887 were arformoterol users, and 450,178 were control patients who did not use arformoterol. Patients were included in the study if they had at least one claim for COPD medication and were continuously enrolled in Medicare Parts A, B, and D. The cohort consisted of mostly white women aged 70 years or older, and 47% were dual-eligible to receive both Medicare and Medicaid benefits. A subgroup of 1,778 arformoterol users were also identified for analysis who were hospitalized and discharged within 30 days of using arformoterol, as well as a subgroup of 21,910 control patients with hospitalizations.
The researchers found COPD-related hospitalization (odds ratio, 1.31; 95% confidence interval, 1.24-1.39; P less than .001), exacerbation (OR, 1.33; 95% CI, 1.26-1.41; P less than .001), use of a systemic corticosteroid (OR, 1.50; 95% CI, 1.43-1.57; P less than .001) or methylxanthine (OR, 1.37; 95% CI, 1.28-1.47; P less than .001), use of oxygen therapy (OR, 2.01; 95% CI, 1.93-2.09; P less than .001), pulmonologist care (OR, 1.40; 95% CI, 1.34-1.46; P less than .001), and respiratory therapist care (OR, 1.23; 95% CI, 1.11-1.36; P less than .001) strongly predicted arformoterol use, while racial/ethnic minority status, psychiatric comorbidity (OR, 0.65; 95% CI, 0.56-0.76; P less than .001), acquired immune deficiency syndrome (OR, 0.69; 95% CI, 0.52-0.94; P less than .01), and dual-eligibility for Medicare and Medicaid (OR, 0.73; 95% CI, 0.70-0.77; P less than .001) lowered the odds of arformoterol use (P less than .001). In the subgroup of patients with hospitalizations, COPD-related admission (OR, 1.83; 95% CI, 1.55-2.14; P less than .001), exacerbation (OR, 2.62; 95% CI, 1.88-3.63; P less than .001)m and inpatient care from a pulmonologist (OR, 1.78; 95% CI, 1.58-2.01; P less than .001) predicted arformoterol use.
“Given the results of this study, increasing access to nebulized maintenance therapy is warranted for select populations with COPD including racial/ethnic minorities, the dual-eligible, and those with certain comorbidities, such as psychiatric disorders,” Dr. Gilmer and colleagues wrote in their study. “Future studies are needed to explore the optimal time to initiate nebulized maintenance therapy, and the potential differential impact of early versus late initiation on patient outcomes.”
Researchers noted that, although their results may seem initially counterintuitive given that COPD has a higher prevalence in men, 56% of the beneficiaries in their Medicare data were women who were 65 years or older, and the results are consistent with other studies that show similar gender distribution findings for maintenance treatment patterns among COPD patients receiving Medicare.
“Since most Medicare beneficiaries with COPD are older than 70 years of age, the higher percentage of women than men in our two cohorts can be explained by the age distributions that ensued as a result of applying our various inclusion and exclusion criteria,” they said.
This study was funded by Sunovian. Dr. Gilmer and one coauthor are paid employees of University of California San Diego, which receives research funding from Advance Health Solutions. Another coauthor is an advisory board member for Advance Health Solutions and a consultant for GlaxoSmithKline, Boehringer-Ingelheim, Astra Zeneca, Novartis, and Pulmonix. Two other coauthors are paid employees of Advance Health Solutions, and another is a paid employee of Sunovion.
SOURCE: Gilmer TP et al. COPD. 2019 Jun 19. doi: 10.1080/15412555.2019.1618256.
FROM COPD: JOURNAL OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE
CNS-directed therapy appears more effective for synDLBCL
Controlling CNS disease is “paramount” in treating diffuse large B-cell lymphoma with synchronous CNS and systemic disease (synDLBCL), according to researchers.
In a retrospective study, the CNS was the most common site of relapse in patients with synDLBCL, and patients had better outcomes when they received CNS-directed therapy.
The 2-year progression-free survival rate was 50% in patients who received CNS-intensive therapy and 31% in those who received CNS-conservative therapy. The 2-year overall survival rate was 54% and 44%, respectively.
Dr. Joel C. Wight, of Austin Health in Heidelberg, Australia, and colleagues conducted this study and recounted their findings in the British Journal of Haematology.
The researchers retrospectively analyzed 80 patients with synDLBCL treated at 10 centers in Australia and the United Kingdom. Patients had DLBCL not otherwise specified (n = 67); high-grade B-cell lymphoma, including double-hit lymphoma (n = 12); or T-cell histiocyte-rich DLBCL (n = 1).
At baseline, all patients were treatment-naive, they had a median age of 64 years (range, 18-87 years), and 68% were male. Seventy percent of patients had high-risk disease according to the CNS International Prognostic Index (IPI), and 96% had non-CNS extranodal disease. The median number of extranodal sites outside the CNS was 2 (range, 0 to more than 10).
Patients were divided into those who received CNS-intensive therapy (n = 38) and those given CNS-conservative therapy (n = 42). The CNS-conservative group was significantly older (P less than .001), significantly more likely to have high-risk disease according to the National Comprehensive Cancer Network IPI (P = .009) or CNS IPI (P = .01) and significantly more likely to have leptomeningeal disease only (P less than .001).
Treatment
CNS-intensive therapy was defined as any established multiagent IV chemotherapy regimen with two or more CNS-penetrating drugs and cytarabine, with or without intrathecal chemotherapy and/or radiotherapy.
CNS-conservative therapy was defined as one or fewer IV CNS-penetrating chemotherapy agents in induction, with or without intrathecal chemotherapy and/or radiotherapy.
Systemic induction in the CNS-intensive group consisted of R-HyperCVAD (rituximab, hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with IV methotrexate and cytarabine) in 66% of patients and R-CODOX-M/IVAC (rituximab, hyperfractionated cyclophosphamide, vincristine, doxorubicin, methotrexate/ifosfamide, etoposide, cytarabine) in 24% of patients.
The most common systemic induction regimens in the CNS-conservative group were R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) or CHOP-like regimens, given to 83% of patients.
CNS-directed IV therapy was given to 100% of the CNS-intensive group and 60% of the CNS-conservative group. This consisted of IV methotrexate plus cytarabine (97%) or MATRix (methotrexate, cytarabine, and thiotepa; 3%) in the CNS-intensive group and high-dose methotrexate in the conservative group.
Intrathecal chemotherapy was given to 97% of the CNS-intensive group and 60% of the CNS-conservative group. CNS-directed radiation was given to 32% and 19%, respectively.
Thirteen patients in the CNS-intensive group and one in the CNS-conservative group underwent autologous transplant as consolidation.
Outcomes
Dose reductions were more frequent in the CNS-conservative group than in the CNS-intensive group, at 48% and 18% (P = .009), as was early cessation of chemotherapy, at 52% and 18% (P = .002). Rates of treatment-related mortality were similar, at 13% in the CNS-intensive group and 12% in the CNS-conservative group.
At the end of induction, the complete response rate was 69% in the CNS-intensive group and 51% in the CNS-conservative group (P = .16). Primary refractory disease was observed in 19% and 38% of patients, respectively (P = .07).
The CNS was the most common site of relapse or progression (n = 28). CNS progression or relapse occurred in 25% of the CNS-intensive group and 49% of the CNS-conservative group (P = .03).
The 2-year progression-free survival rate was 50% for the CNS-intensive group and 31% for the CNS-conservative group (P = .006). The 2-year overall survival rate was 54% and 44%, respectively (P = .037).
When patients were matched for induction outcomes, consolidative transplant did not improve survival.
“The most significant factor affecting survival was the ability to control the CNS disease, which was improved by the addition of IV cytarabine to [high-dose methotrexate],” the researchers wrote.
“Whilst the younger age and more intensive systemic treatment of the CNS-intensive group may have contributed to the improved survival, it is clear that CNS disease control was substantially improved by the addition of cytarabine with lower rates of CNS relapse/progression observed.”
The researchers noted that “adequate control of the CNS disease is paramount and is best achieved by intensive CNS-directed induction.”
There was no formal funding for this study, and the researchers did not provide financial disclosures.
SOURCE: Wight JC et al. Br J Haematol. 2019 Jun 24. doi: 10.1111/bjh.16064.
Controlling CNS disease is “paramount” in treating diffuse large B-cell lymphoma with synchronous CNS and systemic disease (synDLBCL), according to researchers.
In a retrospective study, the CNS was the most common site of relapse in patients with synDLBCL, and patients had better outcomes when they received CNS-directed therapy.
The 2-year progression-free survival rate was 50% in patients who received CNS-intensive therapy and 31% in those who received CNS-conservative therapy. The 2-year overall survival rate was 54% and 44%, respectively.
Dr. Joel C. Wight, of Austin Health in Heidelberg, Australia, and colleagues conducted this study and recounted their findings in the British Journal of Haematology.
The researchers retrospectively analyzed 80 patients with synDLBCL treated at 10 centers in Australia and the United Kingdom. Patients had DLBCL not otherwise specified (n = 67); high-grade B-cell lymphoma, including double-hit lymphoma (n = 12); or T-cell histiocyte-rich DLBCL (n = 1).
At baseline, all patients were treatment-naive, they had a median age of 64 years (range, 18-87 years), and 68% were male. Seventy percent of patients had high-risk disease according to the CNS International Prognostic Index (IPI), and 96% had non-CNS extranodal disease. The median number of extranodal sites outside the CNS was 2 (range, 0 to more than 10).
Patients were divided into those who received CNS-intensive therapy (n = 38) and those given CNS-conservative therapy (n = 42). The CNS-conservative group was significantly older (P less than .001), significantly more likely to have high-risk disease according to the National Comprehensive Cancer Network IPI (P = .009) or CNS IPI (P = .01) and significantly more likely to have leptomeningeal disease only (P less than .001).
Treatment
CNS-intensive therapy was defined as any established multiagent IV chemotherapy regimen with two or more CNS-penetrating drugs and cytarabine, with or without intrathecal chemotherapy and/or radiotherapy.
CNS-conservative therapy was defined as one or fewer IV CNS-penetrating chemotherapy agents in induction, with or without intrathecal chemotherapy and/or radiotherapy.
Systemic induction in the CNS-intensive group consisted of R-HyperCVAD (rituximab, hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with IV methotrexate and cytarabine) in 66% of patients and R-CODOX-M/IVAC (rituximab, hyperfractionated cyclophosphamide, vincristine, doxorubicin, methotrexate/ifosfamide, etoposide, cytarabine) in 24% of patients.
The most common systemic induction regimens in the CNS-conservative group were R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) or CHOP-like regimens, given to 83% of patients.
CNS-directed IV therapy was given to 100% of the CNS-intensive group and 60% of the CNS-conservative group. This consisted of IV methotrexate plus cytarabine (97%) or MATRix (methotrexate, cytarabine, and thiotepa; 3%) in the CNS-intensive group and high-dose methotrexate in the conservative group.
Intrathecal chemotherapy was given to 97% of the CNS-intensive group and 60% of the CNS-conservative group. CNS-directed radiation was given to 32% and 19%, respectively.
Thirteen patients in the CNS-intensive group and one in the CNS-conservative group underwent autologous transplant as consolidation.
Outcomes
Dose reductions were more frequent in the CNS-conservative group than in the CNS-intensive group, at 48% and 18% (P = .009), as was early cessation of chemotherapy, at 52% and 18% (P = .002). Rates of treatment-related mortality were similar, at 13% in the CNS-intensive group and 12% in the CNS-conservative group.
At the end of induction, the complete response rate was 69% in the CNS-intensive group and 51% in the CNS-conservative group (P = .16). Primary refractory disease was observed in 19% and 38% of patients, respectively (P = .07).
The CNS was the most common site of relapse or progression (n = 28). CNS progression or relapse occurred in 25% of the CNS-intensive group and 49% of the CNS-conservative group (P = .03).
The 2-year progression-free survival rate was 50% for the CNS-intensive group and 31% for the CNS-conservative group (P = .006). The 2-year overall survival rate was 54% and 44%, respectively (P = .037).
When patients were matched for induction outcomes, consolidative transplant did not improve survival.
“The most significant factor affecting survival was the ability to control the CNS disease, which was improved by the addition of IV cytarabine to [high-dose methotrexate],” the researchers wrote.
“Whilst the younger age and more intensive systemic treatment of the CNS-intensive group may have contributed to the improved survival, it is clear that CNS disease control was substantially improved by the addition of cytarabine with lower rates of CNS relapse/progression observed.”
The researchers noted that “adequate control of the CNS disease is paramount and is best achieved by intensive CNS-directed induction.”
There was no formal funding for this study, and the researchers did not provide financial disclosures.
SOURCE: Wight JC et al. Br J Haematol. 2019 Jun 24. doi: 10.1111/bjh.16064.
Controlling CNS disease is “paramount” in treating diffuse large B-cell lymphoma with synchronous CNS and systemic disease (synDLBCL), according to researchers.
In a retrospective study, the CNS was the most common site of relapse in patients with synDLBCL, and patients had better outcomes when they received CNS-directed therapy.
The 2-year progression-free survival rate was 50% in patients who received CNS-intensive therapy and 31% in those who received CNS-conservative therapy. The 2-year overall survival rate was 54% and 44%, respectively.
Dr. Joel C. Wight, of Austin Health in Heidelberg, Australia, and colleagues conducted this study and recounted their findings in the British Journal of Haematology.
The researchers retrospectively analyzed 80 patients with synDLBCL treated at 10 centers in Australia and the United Kingdom. Patients had DLBCL not otherwise specified (n = 67); high-grade B-cell lymphoma, including double-hit lymphoma (n = 12); or T-cell histiocyte-rich DLBCL (n = 1).
At baseline, all patients were treatment-naive, they had a median age of 64 years (range, 18-87 years), and 68% were male. Seventy percent of patients had high-risk disease according to the CNS International Prognostic Index (IPI), and 96% had non-CNS extranodal disease. The median number of extranodal sites outside the CNS was 2 (range, 0 to more than 10).
Patients were divided into those who received CNS-intensive therapy (n = 38) and those given CNS-conservative therapy (n = 42). The CNS-conservative group was significantly older (P less than .001), significantly more likely to have high-risk disease according to the National Comprehensive Cancer Network IPI (P = .009) or CNS IPI (P = .01) and significantly more likely to have leptomeningeal disease only (P less than .001).
Treatment
CNS-intensive therapy was defined as any established multiagent IV chemotherapy regimen with two or more CNS-penetrating drugs and cytarabine, with or without intrathecal chemotherapy and/or radiotherapy.
CNS-conservative therapy was defined as one or fewer IV CNS-penetrating chemotherapy agents in induction, with or without intrathecal chemotherapy and/or radiotherapy.
Systemic induction in the CNS-intensive group consisted of R-HyperCVAD (rituximab, hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone alternating with IV methotrexate and cytarabine) in 66% of patients and R-CODOX-M/IVAC (rituximab, hyperfractionated cyclophosphamide, vincristine, doxorubicin, methotrexate/ifosfamide, etoposide, cytarabine) in 24% of patients.
The most common systemic induction regimens in the CNS-conservative group were R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) or CHOP-like regimens, given to 83% of patients.
CNS-directed IV therapy was given to 100% of the CNS-intensive group and 60% of the CNS-conservative group. This consisted of IV methotrexate plus cytarabine (97%) or MATRix (methotrexate, cytarabine, and thiotepa; 3%) in the CNS-intensive group and high-dose methotrexate in the conservative group.
Intrathecal chemotherapy was given to 97% of the CNS-intensive group and 60% of the CNS-conservative group. CNS-directed radiation was given to 32% and 19%, respectively.
Thirteen patients in the CNS-intensive group and one in the CNS-conservative group underwent autologous transplant as consolidation.
Outcomes
Dose reductions were more frequent in the CNS-conservative group than in the CNS-intensive group, at 48% and 18% (P = .009), as was early cessation of chemotherapy, at 52% and 18% (P = .002). Rates of treatment-related mortality were similar, at 13% in the CNS-intensive group and 12% in the CNS-conservative group.
At the end of induction, the complete response rate was 69% in the CNS-intensive group and 51% in the CNS-conservative group (P = .16). Primary refractory disease was observed in 19% and 38% of patients, respectively (P = .07).
The CNS was the most common site of relapse or progression (n = 28). CNS progression or relapse occurred in 25% of the CNS-intensive group and 49% of the CNS-conservative group (P = .03).
The 2-year progression-free survival rate was 50% for the CNS-intensive group and 31% for the CNS-conservative group (P = .006). The 2-year overall survival rate was 54% and 44%, respectively (P = .037).
When patients were matched for induction outcomes, consolidative transplant did not improve survival.
“The most significant factor affecting survival was the ability to control the CNS disease, which was improved by the addition of IV cytarabine to [high-dose methotrexate],” the researchers wrote.
“Whilst the younger age and more intensive systemic treatment of the CNS-intensive group may have contributed to the improved survival, it is clear that CNS disease control was substantially improved by the addition of cytarabine with lower rates of CNS relapse/progression observed.”
The researchers noted that “adequate control of the CNS disease is paramount and is best achieved by intensive CNS-directed induction.”
There was no formal funding for this study, and the researchers did not provide financial disclosures.
SOURCE: Wight JC et al. Br J Haematol. 2019 Jun 24. doi: 10.1111/bjh.16064.
FROM BRITISH JOURNAL OF HAEMATOLOGY