User login
Allergy Testing in Dermatology and Beyond
Allergy testing typically refers to evaluation of a patient for suspected type I or type IV hypersensitivity.1,2 The possibility of type I hypersensitivity is raised in patients presenting with food allergies, allergic rhinitis, asthma, and immediate adverse reactions to medications, whereas type IV hypersensitivity is suspected in patients with eczematous eruptions, delayed adverse cutaneous reactions to medications, and failure of metallic implants (eg, metal joint replacements, cardiac stents) in conjunction with overlying skin rashes (Table 1).1-5 Type II (eg, pemphigus vulgaris) and type III (eg, IgA vasculitis) hypersensitivities are not evaluated with screening allergy tests.

Type I Sensitization
Type I hypersensitivity is an immediate hypersensitivity mediated predominantly by IgE activation of mast cells in the skin as well as the respiratory and gastric mucosa.1 Sensitization of an individual patient occurs when antigen-presenting cells induce a helper T cell (TH2) cytokine response leading to B-cell class switching and allergen-specific IgE production. Upon repeat exposure to the allergen, circulating antibodies then bind to high-affinity receptors on mast cells and basophils and initiate an allergic inflammatory response, leading to a clinical presentation of allergic rhinitis, urticaria, or immediate drug reactions. Confirming type I sensitization may be performed via serologic (in vitro) or skin testing (in vivo).5,6
Serologic Testing (In Vitro)
Serologic testing is a blood test that detects circulating IgE levels against specific allergens.5 The first such test, the radioallergosorbent test, was introduced in the 1970s but is not quantitative and is no longer used. Although common, it is inaccurate to describe current serum IgE (s-IgE) testing as radioallergosorbent testing. There are several US Food and Drug Administration-approved s-IgE assays in common use, and these tests may be helpful in elucidating relevant allergens and for tailoring therapy appropriately, which may consist of avoidance of certain foods or environmental agents and/or allergen immunotherapy.
Skin Testing (In Vivo)
Skin testing can be performed percutaneously (eg, percutaneous skin testing) or intradermally (eg, intradermal testing).6 Percutaneous skin testing is performed by placing a drop of allergen extract on the skin, after which a lancet is used to lightly scratch the skin; intradermal testing is performed by injecting a small amount of allergen extract into the dermis. In both cases, the skin is evaluated after 15 to 20 minutes for the presence and size of a cutaneous wheal. Medications with antihistaminergic activity must be discontinued prior to testing. Both s-IgE and skin testing assess for type I hypersensitivity, and factors such as extensive rash, concern for anaphylaxis, or inability to discontinue antihistamines may favor s-IgE testing versus skin testing. False-positive results can occur with both tests, and for this reason, test results should always be interpreted in conjunction with clinical examination and patient history to determine relevant allergies.
Type IV Sensitization
Type IV hypersensitivity is a delayed hypersensitivity mediated primarily by lymphocytes.2 Sensitization occurs when haptens bind to host proteins and are presented by epidermal and dermal dendritic cells to T lymphocytes in the skin. These lymphocytes then migrate to regional lymph nodes where antigen-specific T lymphocytes are produced and home back to the skin. Upon reexposure to the allergen, these memory T lymphocytes become activated and incite a delayed allergic response. Confirming type IV hypersensitivity primarily is accomplished via patch testing, though other testing modalities exist.
Skin Biopsy
Biopsy is sometimes performed in the workup of an individual presenting with allergic contact dermatitis (ACD) and typically will show spongiosis with normal stratum corneum and epidermal thickness in the setting of acute ACD and mild to marked acanthosis and parakeratosis in chronic ACD.7 The findings, however, are nonspecific and the differential of these histopathologic findings encompasses nummular dermatitis, atopic dermatitis, irritant contact dermatitis, and dyshidrotic eczema, among others. The presence of eosinophils and Langerhans cell microabscesses may provide supportive evidence for ACD over the other spongiotic dermatitides.7,8
Patch Testing
Patch testing is the gold standard in diagnosing type IV hypersensitivities resulting in a clinical presentation of ACD. Hundreds of allergens are commercially available for patch testing, and more commonly tested allergens fall into one of several categories, such as cosmetic preservatives, rubbers, metals, textiles, fragrances, adhesives, antibiotics, plants, and even corticosteroids. Of note, a common misconception is that ACD must result from new exposures; however, patients may develop ACD secondary to an exposure or product they have been using for many years without a problem.
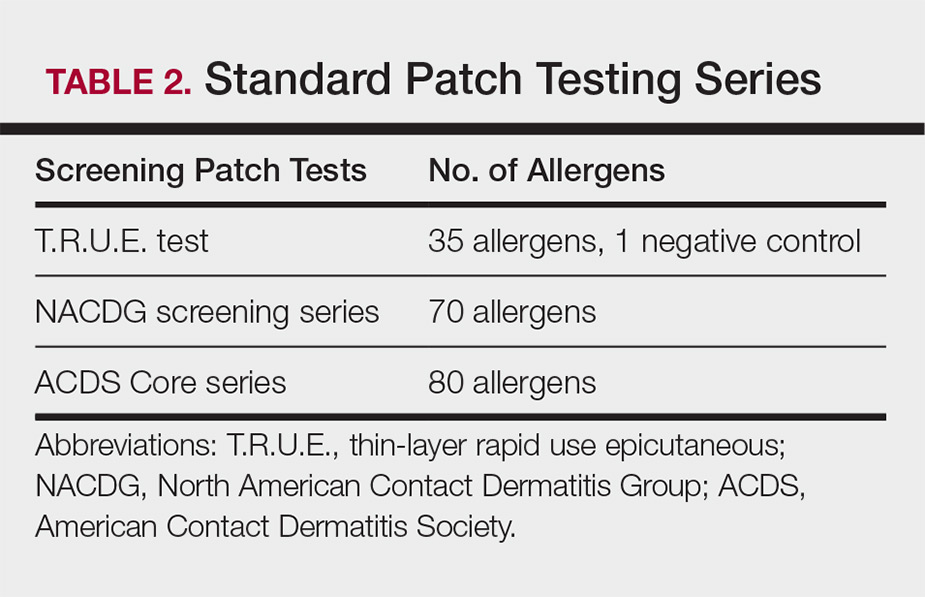
Three commonly used screening series are the thin-layer rapid use epicutaneous (T.R.U.E.) test (SmartPractice), North American Contact Dermatitis Group screening series, and American Contact Dermatitis Society Core 80 allergen series, which have some variation in the type and number of allergens included (Table 2). The T.R.U.E. test will miss a notable number of clinically relevant allergens in comparison to the North American Contact Dermatitis Group and American Contact Dermatitis Society Core series, and it may be of particularly low utility in identifying fragrance or preservative ACD.9
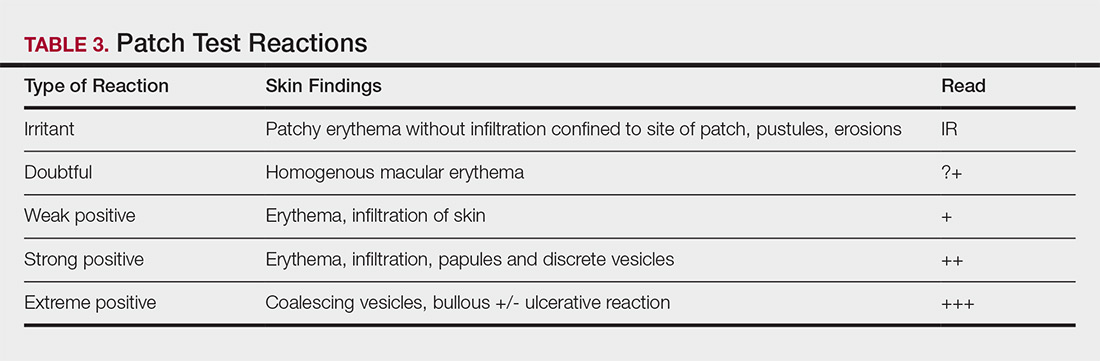
Allergens are placed on the back in chambers in a petrolatum or aqueous medium. The patches remain affixed for 48 hours, during which time the patient is asked to refrain from showering or exercising to prevent loss of patches. The patient's skin is then evaluated for reactions to allergens on 2 separate occasions: at the time of patch removal 48 hours after initial placement, then the areas of patches are marked for delayed readings at day 4 to day 7 after initial patch placement. Results are scored based on the degree of the inflammatory reaction (Table 3). Delayed readings beyond day 7 may be necessary for metals, specific preservatives (eg, dodecyl gallate, propolis), and neomycin.10
There is a wide spectrum of cutaneous disease that should prompt consideration of patch testing, including well-circumscribed eczematous dermatitis (eg, recurrent lip, hand, and foot dermatitis); patchy or diffuse eczema, especially if recently worsened and/or unresponsive to topical steroids; lichenoid eruptions, particularly of mucosal surfaces; mucous membrane eruptions (eg, stomatitis, vulvitis); and eczematous presentations that raise concern for airborne (photodistributed) or systemic contact dermatitis.11-13 Although further studies of efficacy and safety are ongoing, patch testing also may be useful in the diagnosis of nonimmediate cutaneous adverse drug reactions, especially fixed drug eruptions, acute generalized exanthematous pustulosis, systemic contact dermatitis from medications, and drug-induced hypersensitivity syndrome.3 Lastly, patients with type IV hypersensitivity to metals, adhesives, or antibiotics used in metallic orthopedic or cardiac implants may experience implant failure, regional contact dermatitis, or both, and benefit from patch testing prior to implant replacement to assess for potential allergens. Of the joints that fail, it is estimated that up to 5% are due to metal hypersensitivity.4
Throughout patch testing, patients may continue to manage their skin condition with oral antihistamines and topical steroids, though application to the site at which the patches are applied should be avoided throughout patch testing and during the week prior. According to expert consensus, immunosuppressive medications that are less likely to impact patch testing and therefore may be continued include low-dose methotrexate, oral prednisone less than 10 mg daily, biologic therapy, and low-dose cyclosporine (<2 mg/kg daily). Therapeutic interventions that are more likely to impact patch testing and should be avoided include phototherapy or extensive sun exposure within a week prior to testing, oral prednisone more than 10 mg daily, intramuscular triamcinolone within the preceding month, and high-dose cyclosporine (>2 mg/kg daily).14
An important component to successful patch testing is posttest patient counseling. Providers can create a safe list of products for patients by logging onto the American Contact Dermatitis Society website and accessing the Contact Allergen Management Program (CAMP).15 All relevant allergens found on patch testing may be selected and patient-specific identification codes generated. Once these codes are entered into the CAMP app on the patient's cellular device, a personalized, regularly updated list of safe products appears for many categories of products, including shampoos, sunscreens, moisturizers, cosmetic products, and laundry or dish detergents, among others. Of note, this app is not helpful for avoidance in patients with textile allergies. Patients should be counseled that improvement occurs with avoidance, which usually occurs within weeks but may slowly occur over time in some cases.
Lymphocyte Transformation Test (In Vitro)
The lymphocyte transformation test is an experimental in vitro test for type IV hypersensitivity. This serologic test utilizes allergens to stimulate memory T lymphocytes in vitro and measures the degree of response to the allergen. Although this test has generated excitement, particularly for the potential to safely evaluate for severe adverse cutaneous drug reactions, it currently is not the standard of care and is not utilized in the United States.16
Conclusion
Dermatologists play a vital role in the workup of suspected type IV hypersensitivities. Patch testing is an important but underutilized tool in the arsenal of allergy testing and may be indicated in a wide variety of cutaneous presentations, adverse reactions to medications, and implanted device failures. Identification and avoidance of a culprit allergen has the potential to lead to complete resolution of disease and notable improvement in quality of life for patients.
Acknowledgments
The author thanks Nina Botto, MD (San Francisco, California), for her mentorship in the arena of ACD as well as the Women's Dermatologic Society for the support they provided through the mentorship program.
- Oettgen H, Broide DH. Introduction to the mechanisms of allergic disease. In: Holgate ST, Church MK, Broide DH, et al, eds. Allergy. 4th ed. Philadelphia, PA: Elsevier Saunders; 2012:1-32.
- Werfel T, Kapp A. Atopic dermatitis and allergic contact dermatitis. In: Holgate ST, Church MK, Broide DH, et al, eds. Allergy. 4th ed. Philadelphia, PA: Elsevier Saunders; 2012:263-286.
- Zinn A, Gayam S, Chelliah MP, et al. Patch testing for nonimmediate cutaneous adverse drug reactions. J Am Acad Dermatol. 2018;78:421-423.
- Thyssen JP, Menne T, Schalock PC, et al. Pragmatic approach to the clinical work-up of patients with putative allergic disease to metallic orthopaedic implants before and after surgery. Br J Dermatol. 2011;164:473-478.
- Cox L. Overview of serological-specific IgE antibody testing in children. Curr Allergy Asthma Rep. 2011;11:447-453.
- Dolen WK. Skin testing and immunoassays for allergen-specific IgE. Clin Rev Allergy Immunol. 2001;21:229-239.
- Keeling BH, Gavino AC, Gavino AC. Skin biopsy, the allergists' tool: how to interpret a report. Curr Allergy Asthma Rep. 2015;15:62.
- Rosa G, Fernandez AP, Vij A, et al. Langerhans cell collections, but not eosinophils, are clues to a diagnosis of allergic contact dermatitis in appropriate skin biopsies. J Cutan Pathol. 2016;43:498-504.
- DeKoven JG, Warshaw EM, Belsito DV. North American Contact Dermatitis Group patch test results 2013-2014. Dermatitis. 2017;28:33-46.
- Davis MD, Bhate K, Rohlinger AL, et al. Delayed patch test reading after 5 days: the Mayo Clinic experience. J Am Acad Dermatol. 2008;59:225-233.
- Rajagopalan R, Anderson RT. The profile of a patient with contact dermatitis and a suspicion of contact allergy (history, physical characteristics, and dermatology-specific quality of life). Am J Contact Dermat. 1997;8:26-31.
- Huygens S, Goossens A. An update on airborne contact dermatitis. Contact Dermatitis. 2001;44:1-6.
- Salam TN, Fowler JF. Balsam-related systemic contact dermatitis. J Am Acad Dermatol. 2001;45:377-381.
- Fowler JF, Maibach HI, Zirwas M, et al. Effects of immunomodulatory agents on patch testing: expert opinion 2012. Dermatitis. 2012;23:301-303.
- ACDS CAMP. American Contact Dermatitis Society website. https://www.contactderm.org/i4a/pages/index.cfm?pageid=3489. Accessed November 14, 2018.
- Popple A, Williams J, Maxwell G, et al. The lymphocyte transformation test in allergic contact dermatitis: new opportunities. J Immunotoxicol. 2016;13:84-91.
Allergy testing typically refers to evaluation of a patient for suspected type I or type IV hypersensitivity.1,2 The possibility of type I hypersensitivity is raised in patients presenting with food allergies, allergic rhinitis, asthma, and immediate adverse reactions to medications, whereas type IV hypersensitivity is suspected in patients with eczematous eruptions, delayed adverse cutaneous reactions to medications, and failure of metallic implants (eg, metal joint replacements, cardiac stents) in conjunction with overlying skin rashes (Table 1).1-5 Type II (eg, pemphigus vulgaris) and type III (eg, IgA vasculitis) hypersensitivities are not evaluated with screening allergy tests.

Type I Sensitization
Type I hypersensitivity is an immediate hypersensitivity mediated predominantly by IgE activation of mast cells in the skin as well as the respiratory and gastric mucosa.1 Sensitization of an individual patient occurs when antigen-presenting cells induce a helper T cell (TH2) cytokine response leading to B-cell class switching and allergen-specific IgE production. Upon repeat exposure to the allergen, circulating antibodies then bind to high-affinity receptors on mast cells and basophils and initiate an allergic inflammatory response, leading to a clinical presentation of allergic rhinitis, urticaria, or immediate drug reactions. Confirming type I sensitization may be performed via serologic (in vitro) or skin testing (in vivo).5,6
Serologic Testing (In Vitro)
Serologic testing is a blood test that detects circulating IgE levels against specific allergens.5 The first such test, the radioallergosorbent test, was introduced in the 1970s but is not quantitative and is no longer used. Although common, it is inaccurate to describe current serum IgE (s-IgE) testing as radioallergosorbent testing. There are several US Food and Drug Administration-approved s-IgE assays in common use, and these tests may be helpful in elucidating relevant allergens and for tailoring therapy appropriately, which may consist of avoidance of certain foods or environmental agents and/or allergen immunotherapy.
Skin Testing (In Vivo)
Skin testing can be performed percutaneously (eg, percutaneous skin testing) or intradermally (eg, intradermal testing).6 Percutaneous skin testing is performed by placing a drop of allergen extract on the skin, after which a lancet is used to lightly scratch the skin; intradermal testing is performed by injecting a small amount of allergen extract into the dermis. In both cases, the skin is evaluated after 15 to 20 minutes for the presence and size of a cutaneous wheal. Medications with antihistaminergic activity must be discontinued prior to testing. Both s-IgE and skin testing assess for type I hypersensitivity, and factors such as extensive rash, concern for anaphylaxis, or inability to discontinue antihistamines may favor s-IgE testing versus skin testing. False-positive results can occur with both tests, and for this reason, test results should always be interpreted in conjunction with clinical examination and patient history to determine relevant allergies.
Type IV Sensitization
Type IV hypersensitivity is a delayed hypersensitivity mediated primarily by lymphocytes.2 Sensitization occurs when haptens bind to host proteins and are presented by epidermal and dermal dendritic cells to T lymphocytes in the skin. These lymphocytes then migrate to regional lymph nodes where antigen-specific T lymphocytes are produced and home back to the skin. Upon reexposure to the allergen, these memory T lymphocytes become activated and incite a delayed allergic response. Confirming type IV hypersensitivity primarily is accomplished via patch testing, though other testing modalities exist.
Skin Biopsy
Biopsy is sometimes performed in the workup of an individual presenting with allergic contact dermatitis (ACD) and typically will show spongiosis with normal stratum corneum and epidermal thickness in the setting of acute ACD and mild to marked acanthosis and parakeratosis in chronic ACD.7 The findings, however, are nonspecific and the differential of these histopathologic findings encompasses nummular dermatitis, atopic dermatitis, irritant contact dermatitis, and dyshidrotic eczema, among others. The presence of eosinophils and Langerhans cell microabscesses may provide supportive evidence for ACD over the other spongiotic dermatitides.7,8
Patch Testing
Patch testing is the gold standard in diagnosing type IV hypersensitivities resulting in a clinical presentation of ACD. Hundreds of allergens are commercially available for patch testing, and more commonly tested allergens fall into one of several categories, such as cosmetic preservatives, rubbers, metals, textiles, fragrances, adhesives, antibiotics, plants, and even corticosteroids. Of note, a common misconception is that ACD must result from new exposures; however, patients may develop ACD secondary to an exposure or product they have been using for many years without a problem.
Three commonly used screening series are the thin-layer rapid use epicutaneous (T.R.U.E.) test (SmartPractice), North American Contact Dermatitis Group screening series, and American Contact Dermatitis Society Core 80 allergen series, which have some variation in the type and number of allergens included (Table 2). The T.R.U.E. test will miss a notable number of clinically relevant allergens in comparison to the North American Contact Dermatitis Group and American Contact Dermatitis Society Core series, and it may be of particularly low utility in identifying fragrance or preservative ACD.9

Allergens are placed on the back in chambers in a petrolatum or aqueous medium. The patches remain affixed for 48 hours, during which time the patient is asked to refrain from showering or exercising to prevent loss of patches. The patient's skin is then evaluated for reactions to allergens on 2 separate occasions: at the time of patch removal 48 hours after initial placement, then the areas of patches are marked for delayed readings at day 4 to day 7 after initial patch placement. Results are scored based on the degree of the inflammatory reaction (Table 3). Delayed readings beyond day 7 may be necessary for metals, specific preservatives (eg, dodecyl gallate, propolis), and neomycin.10

There is a wide spectrum of cutaneous disease that should prompt consideration of patch testing, including well-circumscribed eczematous dermatitis (eg, recurrent lip, hand, and foot dermatitis); patchy or diffuse eczema, especially if recently worsened and/or unresponsive to topical steroids; lichenoid eruptions, particularly of mucosal surfaces; mucous membrane eruptions (eg, stomatitis, vulvitis); and eczematous presentations that raise concern for airborne (photodistributed) or systemic contact dermatitis.11-13 Although further studies of efficacy and safety are ongoing, patch testing also may be useful in the diagnosis of nonimmediate cutaneous adverse drug reactions, especially fixed drug eruptions, acute generalized exanthematous pustulosis, systemic contact dermatitis from medications, and drug-induced hypersensitivity syndrome.3 Lastly, patients with type IV hypersensitivity to metals, adhesives, or antibiotics used in metallic orthopedic or cardiac implants may experience implant failure, regional contact dermatitis, or both, and benefit from patch testing prior to implant replacement to assess for potential allergens. Of the joints that fail, it is estimated that up to 5% are due to metal hypersensitivity.4
Throughout patch testing, patients may continue to manage their skin condition with oral antihistamines and topical steroids, though application to the site at which the patches are applied should be avoided throughout patch testing and during the week prior. According to expert consensus, immunosuppressive medications that are less likely to impact patch testing and therefore may be continued include low-dose methotrexate, oral prednisone less than 10 mg daily, biologic therapy, and low-dose cyclosporine (<2 mg/kg daily). Therapeutic interventions that are more likely to impact patch testing and should be avoided include phototherapy or extensive sun exposure within a week prior to testing, oral prednisone more than 10 mg daily, intramuscular triamcinolone within the preceding month, and high-dose cyclosporine (>2 mg/kg daily).14
An important component to successful patch testing is posttest patient counseling. Providers can create a safe list of products for patients by logging onto the American Contact Dermatitis Society website and accessing the Contact Allergen Management Program (CAMP).15 All relevant allergens found on patch testing may be selected and patient-specific identification codes generated. Once these codes are entered into the CAMP app on the patient's cellular device, a personalized, regularly updated list of safe products appears for many categories of products, including shampoos, sunscreens, moisturizers, cosmetic products, and laundry or dish detergents, among others. Of note, this app is not helpful for avoidance in patients with textile allergies. Patients should be counseled that improvement occurs with avoidance, which usually occurs within weeks but may slowly occur over time in some cases.
Lymphocyte Transformation Test (In Vitro)
The lymphocyte transformation test is an experimental in vitro test for type IV hypersensitivity. This serologic test utilizes allergens to stimulate memory T lymphocytes in vitro and measures the degree of response to the allergen. Although this test has generated excitement, particularly for the potential to safely evaluate for severe adverse cutaneous drug reactions, it currently is not the standard of care and is not utilized in the United States.16
Conclusion
Dermatologists play a vital role in the workup of suspected type IV hypersensitivities. Patch testing is an important but underutilized tool in the arsenal of allergy testing and may be indicated in a wide variety of cutaneous presentations, adverse reactions to medications, and implanted device failures. Identification and avoidance of a culprit allergen has the potential to lead to complete resolution of disease and notable improvement in quality of life for patients.
Acknowledgments
The author thanks Nina Botto, MD (San Francisco, California), for her mentorship in the arena of ACD as well as the Women's Dermatologic Society for the support they provided through the mentorship program.
Allergy testing typically refers to evaluation of a patient for suspected type I or type IV hypersensitivity.1,2 The possibility of type I hypersensitivity is raised in patients presenting with food allergies, allergic rhinitis, asthma, and immediate adverse reactions to medications, whereas type IV hypersensitivity is suspected in patients with eczematous eruptions, delayed adverse cutaneous reactions to medications, and failure of metallic implants (eg, metal joint replacements, cardiac stents) in conjunction with overlying skin rashes (Table 1).1-5 Type II (eg, pemphigus vulgaris) and type III (eg, IgA vasculitis) hypersensitivities are not evaluated with screening allergy tests.

Type I Sensitization
Type I hypersensitivity is an immediate hypersensitivity mediated predominantly by IgE activation of mast cells in the skin as well as the respiratory and gastric mucosa.1 Sensitization of an individual patient occurs when antigen-presenting cells induce a helper T cell (TH2) cytokine response leading to B-cell class switching and allergen-specific IgE production. Upon repeat exposure to the allergen, circulating antibodies then bind to high-affinity receptors on mast cells and basophils and initiate an allergic inflammatory response, leading to a clinical presentation of allergic rhinitis, urticaria, or immediate drug reactions. Confirming type I sensitization may be performed via serologic (in vitro) or skin testing (in vivo).5,6
Serologic Testing (In Vitro)
Serologic testing is a blood test that detects circulating IgE levels against specific allergens.5 The first such test, the radioallergosorbent test, was introduced in the 1970s but is not quantitative and is no longer used. Although common, it is inaccurate to describe current serum IgE (s-IgE) testing as radioallergosorbent testing. There are several US Food and Drug Administration-approved s-IgE assays in common use, and these tests may be helpful in elucidating relevant allergens and for tailoring therapy appropriately, which may consist of avoidance of certain foods or environmental agents and/or allergen immunotherapy.
Skin Testing (In Vivo)
Skin testing can be performed percutaneously (eg, percutaneous skin testing) or intradermally (eg, intradermal testing).6 Percutaneous skin testing is performed by placing a drop of allergen extract on the skin, after which a lancet is used to lightly scratch the skin; intradermal testing is performed by injecting a small amount of allergen extract into the dermis. In both cases, the skin is evaluated after 15 to 20 minutes for the presence and size of a cutaneous wheal. Medications with antihistaminergic activity must be discontinued prior to testing. Both s-IgE and skin testing assess for type I hypersensitivity, and factors such as extensive rash, concern for anaphylaxis, or inability to discontinue antihistamines may favor s-IgE testing versus skin testing. False-positive results can occur with both tests, and for this reason, test results should always be interpreted in conjunction with clinical examination and patient history to determine relevant allergies.
Type IV Sensitization
Type IV hypersensitivity is a delayed hypersensitivity mediated primarily by lymphocytes.2 Sensitization occurs when haptens bind to host proteins and are presented by epidermal and dermal dendritic cells to T lymphocytes in the skin. These lymphocytes then migrate to regional lymph nodes where antigen-specific T lymphocytes are produced and home back to the skin. Upon reexposure to the allergen, these memory T lymphocytes become activated and incite a delayed allergic response. Confirming type IV hypersensitivity primarily is accomplished via patch testing, though other testing modalities exist.
Skin Biopsy
Biopsy is sometimes performed in the workup of an individual presenting with allergic contact dermatitis (ACD) and typically will show spongiosis with normal stratum corneum and epidermal thickness in the setting of acute ACD and mild to marked acanthosis and parakeratosis in chronic ACD.7 The findings, however, are nonspecific and the differential of these histopathologic findings encompasses nummular dermatitis, atopic dermatitis, irritant contact dermatitis, and dyshidrotic eczema, among others. The presence of eosinophils and Langerhans cell microabscesses may provide supportive evidence for ACD over the other spongiotic dermatitides.7,8
Patch Testing
Patch testing is the gold standard in diagnosing type IV hypersensitivities resulting in a clinical presentation of ACD. Hundreds of allergens are commercially available for patch testing, and more commonly tested allergens fall into one of several categories, such as cosmetic preservatives, rubbers, metals, textiles, fragrances, adhesives, antibiotics, plants, and even corticosteroids. Of note, a common misconception is that ACD must result from new exposures; however, patients may develop ACD secondary to an exposure or product they have been using for many years without a problem.
Three commonly used screening series are the thin-layer rapid use epicutaneous (T.R.U.E.) test (SmartPractice), North American Contact Dermatitis Group screening series, and American Contact Dermatitis Society Core 80 allergen series, which have some variation in the type and number of allergens included (Table 2). The T.R.U.E. test will miss a notable number of clinically relevant allergens in comparison to the North American Contact Dermatitis Group and American Contact Dermatitis Society Core series, and it may be of particularly low utility in identifying fragrance or preservative ACD.9

Allergens are placed on the back in chambers in a petrolatum or aqueous medium. The patches remain affixed for 48 hours, during which time the patient is asked to refrain from showering or exercising to prevent loss of patches. The patient's skin is then evaluated for reactions to allergens on 2 separate occasions: at the time of patch removal 48 hours after initial placement, then the areas of patches are marked for delayed readings at day 4 to day 7 after initial patch placement. Results are scored based on the degree of the inflammatory reaction (Table 3). Delayed readings beyond day 7 may be necessary for metals, specific preservatives (eg, dodecyl gallate, propolis), and neomycin.10

There is a wide spectrum of cutaneous disease that should prompt consideration of patch testing, including well-circumscribed eczematous dermatitis (eg, recurrent lip, hand, and foot dermatitis); patchy or diffuse eczema, especially if recently worsened and/or unresponsive to topical steroids; lichenoid eruptions, particularly of mucosal surfaces; mucous membrane eruptions (eg, stomatitis, vulvitis); and eczematous presentations that raise concern for airborne (photodistributed) or systemic contact dermatitis.11-13 Although further studies of efficacy and safety are ongoing, patch testing also may be useful in the diagnosis of nonimmediate cutaneous adverse drug reactions, especially fixed drug eruptions, acute generalized exanthematous pustulosis, systemic contact dermatitis from medications, and drug-induced hypersensitivity syndrome.3 Lastly, patients with type IV hypersensitivity to metals, adhesives, or antibiotics used in metallic orthopedic or cardiac implants may experience implant failure, regional contact dermatitis, or both, and benefit from patch testing prior to implant replacement to assess for potential allergens. Of the joints that fail, it is estimated that up to 5% are due to metal hypersensitivity.4
Throughout patch testing, patients may continue to manage their skin condition with oral antihistamines and topical steroids, though application to the site at which the patches are applied should be avoided throughout patch testing and during the week prior. According to expert consensus, immunosuppressive medications that are less likely to impact patch testing and therefore may be continued include low-dose methotrexate, oral prednisone less than 10 mg daily, biologic therapy, and low-dose cyclosporine (<2 mg/kg daily). Therapeutic interventions that are more likely to impact patch testing and should be avoided include phototherapy or extensive sun exposure within a week prior to testing, oral prednisone more than 10 mg daily, intramuscular triamcinolone within the preceding month, and high-dose cyclosporine (>2 mg/kg daily).14
An important component to successful patch testing is posttest patient counseling. Providers can create a safe list of products for patients by logging onto the American Contact Dermatitis Society website and accessing the Contact Allergen Management Program (CAMP).15 All relevant allergens found on patch testing may be selected and patient-specific identification codes generated. Once these codes are entered into the CAMP app on the patient's cellular device, a personalized, regularly updated list of safe products appears for many categories of products, including shampoos, sunscreens, moisturizers, cosmetic products, and laundry or dish detergents, among others. Of note, this app is not helpful for avoidance in patients with textile allergies. Patients should be counseled that improvement occurs with avoidance, which usually occurs within weeks but may slowly occur over time in some cases.
Lymphocyte Transformation Test (In Vitro)
The lymphocyte transformation test is an experimental in vitro test for type IV hypersensitivity. This serologic test utilizes allergens to stimulate memory T lymphocytes in vitro and measures the degree of response to the allergen. Although this test has generated excitement, particularly for the potential to safely evaluate for severe adverse cutaneous drug reactions, it currently is not the standard of care and is not utilized in the United States.16
Conclusion
Dermatologists play a vital role in the workup of suspected type IV hypersensitivities. Patch testing is an important but underutilized tool in the arsenal of allergy testing and may be indicated in a wide variety of cutaneous presentations, adverse reactions to medications, and implanted device failures. Identification and avoidance of a culprit allergen has the potential to lead to complete resolution of disease and notable improvement in quality of life for patients.
Acknowledgments
The author thanks Nina Botto, MD (San Francisco, California), for her mentorship in the arena of ACD as well as the Women's Dermatologic Society for the support they provided through the mentorship program.
- Oettgen H, Broide DH. Introduction to the mechanisms of allergic disease. In: Holgate ST, Church MK, Broide DH, et al, eds. Allergy. 4th ed. Philadelphia, PA: Elsevier Saunders; 2012:1-32.
- Werfel T, Kapp A. Atopic dermatitis and allergic contact dermatitis. In: Holgate ST, Church MK, Broide DH, et al, eds. Allergy. 4th ed. Philadelphia, PA: Elsevier Saunders; 2012:263-286.
- Zinn A, Gayam S, Chelliah MP, et al. Patch testing for nonimmediate cutaneous adverse drug reactions. J Am Acad Dermatol. 2018;78:421-423.
- Thyssen JP, Menne T, Schalock PC, et al. Pragmatic approach to the clinical work-up of patients with putative allergic disease to metallic orthopaedic implants before and after surgery. Br J Dermatol. 2011;164:473-478.
- Cox L. Overview of serological-specific IgE antibody testing in children. Curr Allergy Asthma Rep. 2011;11:447-453.
- Dolen WK. Skin testing and immunoassays for allergen-specific IgE. Clin Rev Allergy Immunol. 2001;21:229-239.
- Keeling BH, Gavino AC, Gavino AC. Skin biopsy, the allergists' tool: how to interpret a report. Curr Allergy Asthma Rep. 2015;15:62.
- Rosa G, Fernandez AP, Vij A, et al. Langerhans cell collections, but not eosinophils, are clues to a diagnosis of allergic contact dermatitis in appropriate skin biopsies. J Cutan Pathol. 2016;43:498-504.
- DeKoven JG, Warshaw EM, Belsito DV. North American Contact Dermatitis Group patch test results 2013-2014. Dermatitis. 2017;28:33-46.
- Davis MD, Bhate K, Rohlinger AL, et al. Delayed patch test reading after 5 days: the Mayo Clinic experience. J Am Acad Dermatol. 2008;59:225-233.
- Rajagopalan R, Anderson RT. The profile of a patient with contact dermatitis and a suspicion of contact allergy (history, physical characteristics, and dermatology-specific quality of life). Am J Contact Dermat. 1997;8:26-31.
- Huygens S, Goossens A. An update on airborne contact dermatitis. Contact Dermatitis. 2001;44:1-6.
- Salam TN, Fowler JF. Balsam-related systemic contact dermatitis. J Am Acad Dermatol. 2001;45:377-381.
- Fowler JF, Maibach HI, Zirwas M, et al. Effects of immunomodulatory agents on patch testing: expert opinion 2012. Dermatitis. 2012;23:301-303.
- ACDS CAMP. American Contact Dermatitis Society website. https://www.contactderm.org/i4a/pages/index.cfm?pageid=3489. Accessed November 14, 2018.
- Popple A, Williams J, Maxwell G, et al. The lymphocyte transformation test in allergic contact dermatitis: new opportunities. J Immunotoxicol. 2016;13:84-91.
- Oettgen H, Broide DH. Introduction to the mechanisms of allergic disease. In: Holgate ST, Church MK, Broide DH, et al, eds. Allergy. 4th ed. Philadelphia, PA: Elsevier Saunders; 2012:1-32.
- Werfel T, Kapp A. Atopic dermatitis and allergic contact dermatitis. In: Holgate ST, Church MK, Broide DH, et al, eds. Allergy. 4th ed. Philadelphia, PA: Elsevier Saunders; 2012:263-286.
- Zinn A, Gayam S, Chelliah MP, et al. Patch testing for nonimmediate cutaneous adverse drug reactions. J Am Acad Dermatol. 2018;78:421-423.
- Thyssen JP, Menne T, Schalock PC, et al. Pragmatic approach to the clinical work-up of patients with putative allergic disease to metallic orthopaedic implants before and after surgery. Br J Dermatol. 2011;164:473-478.
- Cox L. Overview of serological-specific IgE antibody testing in children. Curr Allergy Asthma Rep. 2011;11:447-453.
- Dolen WK. Skin testing and immunoassays for allergen-specific IgE. Clin Rev Allergy Immunol. 2001;21:229-239.
- Keeling BH, Gavino AC, Gavino AC. Skin biopsy, the allergists' tool: how to interpret a report. Curr Allergy Asthma Rep. 2015;15:62.
- Rosa G, Fernandez AP, Vij A, et al. Langerhans cell collections, but not eosinophils, are clues to a diagnosis of allergic contact dermatitis in appropriate skin biopsies. J Cutan Pathol. 2016;43:498-504.
- DeKoven JG, Warshaw EM, Belsito DV. North American Contact Dermatitis Group patch test results 2013-2014. Dermatitis. 2017;28:33-46.
- Davis MD, Bhate K, Rohlinger AL, et al. Delayed patch test reading after 5 days: the Mayo Clinic experience. J Am Acad Dermatol. 2008;59:225-233.
- Rajagopalan R, Anderson RT. The profile of a patient with contact dermatitis and a suspicion of contact allergy (history, physical characteristics, and dermatology-specific quality of life). Am J Contact Dermat. 1997;8:26-31.
- Huygens S, Goossens A. An update on airborne contact dermatitis. Contact Dermatitis. 2001;44:1-6.
- Salam TN, Fowler JF. Balsam-related systemic contact dermatitis. J Am Acad Dermatol. 2001;45:377-381.
- Fowler JF, Maibach HI, Zirwas M, et al. Effects of immunomodulatory agents on patch testing: expert opinion 2012. Dermatitis. 2012;23:301-303.
- ACDS CAMP. American Contact Dermatitis Society website. https://www.contactderm.org/i4a/pages/index.cfm?pageid=3489. Accessed November 14, 2018.
- Popple A, Williams J, Maxwell G, et al. The lymphocyte transformation test in allergic contact dermatitis: new opportunities. J Immunotoxicol. 2016;13:84-91.
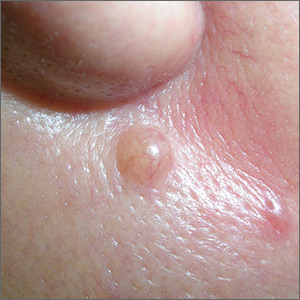
Mole near nose
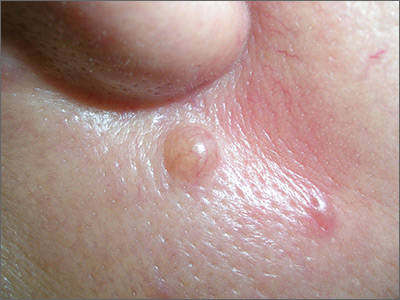
The differential diagnosis for this lesion included a benign intradermal nevus and a basal cell carcinoma. So the FP recommended a shave biopsy to be sure that it was not cancer. (See the Watch & Learn video on “Shave biopsy.”)
After obtaining patient consent, he injected the 1% lidocaine with epinephrine and waited 5 minutes for the epinephrine to begin to work. He performed the shave with a Dermablade, and used a cotton-tipped applicator to apply aluminum chloride to the site. He used a twisting motion and pressure to achieve hemostasis. He dressed the lesion with petrolatum and some gauze.
Dermatopathology showed that this mole was a benign intradermal nevus. The FP reassured the patient and recommended that she be careful to avoid sun exposure.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The differential diagnosis for this lesion included a benign intradermal nevus and a basal cell carcinoma. So the FP recommended a shave biopsy to be sure that it was not cancer. (See the Watch & Learn video on “Shave biopsy.”)
After obtaining patient consent, he injected the 1% lidocaine with epinephrine and waited 5 minutes for the epinephrine to begin to work. He performed the shave with a Dermablade, and used a cotton-tipped applicator to apply aluminum chloride to the site. He used a twisting motion and pressure to achieve hemostasis. He dressed the lesion with petrolatum and some gauze.
Dermatopathology showed that this mole was a benign intradermal nevus. The FP reassured the patient and recommended that she be careful to avoid sun exposure.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.

The differential diagnosis for this lesion included a benign intradermal nevus and a basal cell carcinoma. So the FP recommended a shave biopsy to be sure that it was not cancer. (See the Watch & Learn video on “Shave biopsy.”)
After obtaining patient consent, he injected the 1% lidocaine with epinephrine and waited 5 minutes for the epinephrine to begin to work. He performed the shave with a Dermablade, and used a cotton-tipped applicator to apply aluminum chloride to the site. He used a twisting motion and pressure to achieve hemostasis. He dressed the lesion with petrolatum and some gauze.
Dermatopathology showed that this mole was a benign intradermal nevus. The FP reassured the patient and recommended that she be careful to avoid sun exposure.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Craig Getting, Luis Aguilar-Montalva: Part I
Thomas Jefferson University in Philadelphia partnered with the Lantern Theater Company to offer a class that teaches staging, acting, and playwriting. The class is designed specifically for med students, current residents, attending physicians, and any other medical professionals. The class eliminates hierarchy, encourages artistic exploration, and can lead to increased empathy when dealing with patients and their families. Nick, Luis, and Craig recognize the similarities between medicine and art but also highlight what we can learn from different passions.
Apple Podcasts
Google Podcasts
Thomas Jefferson University in Philadelphia partnered with the Lantern Theater Company to offer a class that teaches staging, acting, and playwriting. The class is designed specifically for med students, current residents, attending physicians, and any other medical professionals. The class eliminates hierarchy, encourages artistic exploration, and can lead to increased empathy when dealing with patients and their families. Nick, Luis, and Craig recognize the similarities between medicine and art but also highlight what we can learn from different passions.
Apple Podcasts
Google Podcasts
Thomas Jefferson University in Philadelphia partnered with the Lantern Theater Company to offer a class that teaches staging, acting, and playwriting. The class is designed specifically for med students, current residents, attending physicians, and any other medical professionals. The class eliminates hierarchy, encourages artistic exploration, and can lead to increased empathy when dealing with patients and their families. Nick, Luis, and Craig recognize the similarities between medicine and art but also highlight what we can learn from different passions.
Apple Podcasts
Google Podcasts
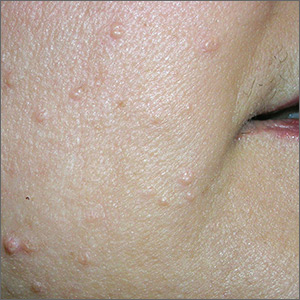
Multiple growths on face
Figure 1
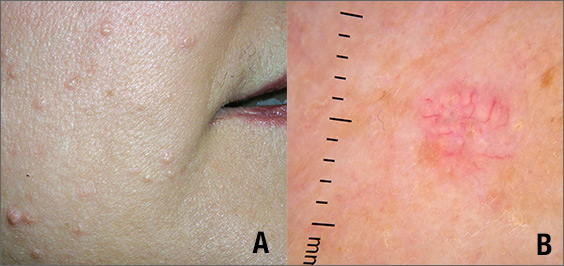
While the differential diagnosis for these lesions (FIGURE 1A) included basal cell carcinoma, the FP had reason to suspect that these papules were actually sebaceous hyperplasia.
The FP saw a pattern of crown-like vessels and popcorn-like structures (FIGURE 1B) when he examined the patient with his dermatoscope. None of the vessels crossed the midline, and the popcorn-like structures were hyperplastic sebaceous glands. The FP photographed the largest lesion with a dermatoscope attached to his smart phone and showed the reassuring pattern to the anxious patient. He explained to her why this was not skin cancer and how hyperplasia of sebaceous glands is often normal for aging facial skin. He also offered her treatment if she thought that the lesions were cosmetically unappealing.
The patient said that she would be grateful to have the largest lesion treated because it did bother her when she looked in the mirror. The FP treated this lesion using electrosurgery with a blunt tipped electrode, on a setting of 2.1, without anesthesia. He warned the patient before he applied the activated electrode to the skin, and the patient tolerated the procedure well. The sebaceous glands melted easily with the current. The result appeared gray and was expected to heal with minimal to no scarring. At a future visit, the patient said that she was happy with the result and asked if additional lesions could be treated with the same electrosurgical approach.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Figure 1

While the differential diagnosis for these lesions (FIGURE 1A) included basal cell carcinoma, the FP had reason to suspect that these papules were actually sebaceous hyperplasia.
The FP saw a pattern of crown-like vessels and popcorn-like structures (FIGURE 1B) when he examined the patient with his dermatoscope. None of the vessels crossed the midline, and the popcorn-like structures were hyperplastic sebaceous glands. The FP photographed the largest lesion with a dermatoscope attached to his smart phone and showed the reassuring pattern to the anxious patient. He explained to her why this was not skin cancer and how hyperplasia of sebaceous glands is often normal for aging facial skin. He also offered her treatment if she thought that the lesions were cosmetically unappealing.
The patient said that she would be grateful to have the largest lesion treated because it did bother her when she looked in the mirror. The FP treated this lesion using electrosurgery with a blunt tipped electrode, on a setting of 2.1, without anesthesia. He warned the patient before he applied the activated electrode to the skin, and the patient tolerated the procedure well. The sebaceous glands melted easily with the current. The result appeared gray and was expected to heal with minimal to no scarring. At a future visit, the patient said that she was happy with the result and asked if additional lesions could be treated with the same electrosurgical approach.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Figure 1

While the differential diagnosis for these lesions (FIGURE 1A) included basal cell carcinoma, the FP had reason to suspect that these papules were actually sebaceous hyperplasia.
The FP saw a pattern of crown-like vessels and popcorn-like structures (FIGURE 1B) when he examined the patient with his dermatoscope. None of the vessels crossed the midline, and the popcorn-like structures were hyperplastic sebaceous glands. The FP photographed the largest lesion with a dermatoscope attached to his smart phone and showed the reassuring pattern to the anxious patient. He explained to her why this was not skin cancer and how hyperplasia of sebaceous glands is often normal for aging facial skin. He also offered her treatment if she thought that the lesions were cosmetically unappealing.
The patient said that she would be grateful to have the largest lesion treated because it did bother her when she looked in the mirror. The FP treated this lesion using electrosurgery with a blunt tipped electrode, on a setting of 2.1, without anesthesia. He warned the patient before he applied the activated electrode to the skin, and the patient tolerated the procedure well. The sebaceous glands melted easily with the current. The result appeared gray and was expected to heal with minimal to no scarring. At a future visit, the patient said that she was happy with the result and asked if additional lesions could be treated with the same electrosurgical approach.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Think research is just for MD-PhDs? Think again
ATLANTA – You don’t have to hold an advanced research degree or secure National Institutes of Health funding in order to contribute to neurology research in a meaningful way.
That’s a key finding from an analysis of 244 neurology residency program graduates.
“Science as a whole is trying to get better,” lead study author Wyatt P. Bensken said in an interview at the annual meeting of the American Neurological Association. “If your goal is to be a clinician, that doesn’t mean you can’t contribute to research. If your goal is to see patients for 80% of your time, that doesn’t mean that other 20% – which is research – disqualifies you from being a physician-scientist.”
In an effort to better understand the current status of the physician-scientist workforce in the neurology field, Mr. Bensken and his colleagues identified neurology residency graduates from the top National Institute of Neurological Disorders and Stroke–funded institutions for 2003, 2004, and 2005 via program websites. Data points collected for each individual included complete NIH and other government funding history, number of post-residency publications by year, and the Hirsch-index, or h-index, which measures an individual’s research publication impact. The researchers conducted data analysis via visualization and ANOVA testing.
Mr. Bensken, a research collaborator with the NINDS who is also a PhD student at Case Western Reserve University in Cleveland, reported that 186 of the 244 neurology residency program graduates had demonstrated interest in research based on their publication activity findings. Specifically, 26 had obtained an R01 grant, 31 were non–R01-funded, and 129 were nonfunded. Of the 26 individuals who had obtained an R01, 15 (58%) were MD-PhDs, from a total of 50 MD‐PhDs in the cohort. In addition, 43 individuals had a K‐series award, with 18 going on to receive R01 funding.
Of those with non‐R01 funding or no funding, a number of individuals performed as well as R01‐funded individuals with respect to post‐residency publication rate and impact factor. However, the publication rate and impact factor were highest in the R01-funded group (6.4 and 28.6, respectively), followed by those in the non‐R01 group (3.0 and 15.9), and those in the nonfunded group (1.2 and 8.0). Further, the publications‐per‐research hour for the three groups revealed varied productivity levels. Specifically, those in the R01-funded group with 80% protected research time produced 3.2 publications per 1,000 research hours, while those in the non–R01-funded group with 40% protected research time produced 3.0 publications per 1,000 research hours. Meanwhile, those without R01 funding overall (those with non-RO1 funding and those without funding) performed at a higher per-hour rate, when estimating 10% or 15% protected time (4.9 and 3.3 publications per 1,000 research hours, respectively).
“I think this reinforces the notion that there are far more neurologists out there who aren’t trained as MD-PhDs, who aren’t receiving R01s, but who are making meaningful contributions,” Mr. Bensken said. “Our ultimate goal is to maximize the potential of everybody in this environment to contribute. If everyone was able to contribute what they could, I think research would be far more successful and far more impactful than it is now.”
The study was funded by the NINDS. Mr. Bensken reported having no financial disclosures.
SOURCE: Bensken WP et al. Ann Neurol. 2018;84[S22]:S72-3, Abstract S176.
ATLANTA – You don’t have to hold an advanced research degree or secure National Institutes of Health funding in order to contribute to neurology research in a meaningful way.
That’s a key finding from an analysis of 244 neurology residency program graduates.
“Science as a whole is trying to get better,” lead study author Wyatt P. Bensken said in an interview at the annual meeting of the American Neurological Association. “If your goal is to be a clinician, that doesn’t mean you can’t contribute to research. If your goal is to see patients for 80% of your time, that doesn’t mean that other 20% – which is research – disqualifies you from being a physician-scientist.”
In an effort to better understand the current status of the physician-scientist workforce in the neurology field, Mr. Bensken and his colleagues identified neurology residency graduates from the top National Institute of Neurological Disorders and Stroke–funded institutions for 2003, 2004, and 2005 via program websites. Data points collected for each individual included complete NIH and other government funding history, number of post-residency publications by year, and the Hirsch-index, or h-index, which measures an individual’s research publication impact. The researchers conducted data analysis via visualization and ANOVA testing.
Mr. Bensken, a research collaborator with the NINDS who is also a PhD student at Case Western Reserve University in Cleveland, reported that 186 of the 244 neurology residency program graduates had demonstrated interest in research based on their publication activity findings. Specifically, 26 had obtained an R01 grant, 31 were non–R01-funded, and 129 were nonfunded. Of the 26 individuals who had obtained an R01, 15 (58%) were MD-PhDs, from a total of 50 MD‐PhDs in the cohort. In addition, 43 individuals had a K‐series award, with 18 going on to receive R01 funding.
Of those with non‐R01 funding or no funding, a number of individuals performed as well as R01‐funded individuals with respect to post‐residency publication rate and impact factor. However, the publication rate and impact factor were highest in the R01-funded group (6.4 and 28.6, respectively), followed by those in the non‐R01 group (3.0 and 15.9), and those in the nonfunded group (1.2 and 8.0). Further, the publications‐per‐research hour for the three groups revealed varied productivity levels. Specifically, those in the R01-funded group with 80% protected research time produced 3.2 publications per 1,000 research hours, while those in the non–R01-funded group with 40% protected research time produced 3.0 publications per 1,000 research hours. Meanwhile, those without R01 funding overall (those with non-RO1 funding and those without funding) performed at a higher per-hour rate, when estimating 10% or 15% protected time (4.9 and 3.3 publications per 1,000 research hours, respectively).
“I think this reinforces the notion that there are far more neurologists out there who aren’t trained as MD-PhDs, who aren’t receiving R01s, but who are making meaningful contributions,” Mr. Bensken said. “Our ultimate goal is to maximize the potential of everybody in this environment to contribute. If everyone was able to contribute what they could, I think research would be far more successful and far more impactful than it is now.”
The study was funded by the NINDS. Mr. Bensken reported having no financial disclosures.
SOURCE: Bensken WP et al. Ann Neurol. 2018;84[S22]:S72-3, Abstract S176.
ATLANTA – You don’t have to hold an advanced research degree or secure National Institutes of Health funding in order to contribute to neurology research in a meaningful way.
That’s a key finding from an analysis of 244 neurology residency program graduates.
“Science as a whole is trying to get better,” lead study author Wyatt P. Bensken said in an interview at the annual meeting of the American Neurological Association. “If your goal is to be a clinician, that doesn’t mean you can’t contribute to research. If your goal is to see patients for 80% of your time, that doesn’t mean that other 20% – which is research – disqualifies you from being a physician-scientist.”
In an effort to better understand the current status of the physician-scientist workforce in the neurology field, Mr. Bensken and his colleagues identified neurology residency graduates from the top National Institute of Neurological Disorders and Stroke–funded institutions for 2003, 2004, and 2005 via program websites. Data points collected for each individual included complete NIH and other government funding history, number of post-residency publications by year, and the Hirsch-index, or h-index, which measures an individual’s research publication impact. The researchers conducted data analysis via visualization and ANOVA testing.
Mr. Bensken, a research collaborator with the NINDS who is also a PhD student at Case Western Reserve University in Cleveland, reported that 186 of the 244 neurology residency program graduates had demonstrated interest in research based on their publication activity findings. Specifically, 26 had obtained an R01 grant, 31 were non–R01-funded, and 129 were nonfunded. Of the 26 individuals who had obtained an R01, 15 (58%) were MD-PhDs, from a total of 50 MD‐PhDs in the cohort. In addition, 43 individuals had a K‐series award, with 18 going on to receive R01 funding.
Of those with non‐R01 funding or no funding, a number of individuals performed as well as R01‐funded individuals with respect to post‐residency publication rate and impact factor. However, the publication rate and impact factor were highest in the R01-funded group (6.4 and 28.6, respectively), followed by those in the non‐R01 group (3.0 and 15.9), and those in the nonfunded group (1.2 and 8.0). Further, the publications‐per‐research hour for the three groups revealed varied productivity levels. Specifically, those in the R01-funded group with 80% protected research time produced 3.2 publications per 1,000 research hours, while those in the non–R01-funded group with 40% protected research time produced 3.0 publications per 1,000 research hours. Meanwhile, those without R01 funding overall (those with non-RO1 funding and those without funding) performed at a higher per-hour rate, when estimating 10% or 15% protected time (4.9 and 3.3 publications per 1,000 research hours, respectively).
“I think this reinforces the notion that there are far more neurologists out there who aren’t trained as MD-PhDs, who aren’t receiving R01s, but who are making meaningful contributions,” Mr. Bensken said. “Our ultimate goal is to maximize the potential of everybody in this environment to contribute. If everyone was able to contribute what they could, I think research would be far more successful and far more impactful than it is now.”
The study was funded by the NINDS. Mr. Bensken reported having no financial disclosures.
SOURCE: Bensken WP et al. Ann Neurol. 2018;84[S22]:S72-3, Abstract S176.
REPORTING FROM ANA 2018
Key clinical point:
Major finding: Those in the R01-funded group with 80% protected research time produced 3.2 publications per 1,000 research hours, while those in the non–R01-funded group with 40% protected research time produced 3.0 publications per 1,000 research hours.
Study details: An analysis of 244 neurology residency program graduates.
Disclosures: The study was funded by the NINDS. Mr. Bensken reported having no financial disclosures.
Source: Bensken WP et al. Ann Neurol. 2018;84[S 22]:S72-3, Abstract S176.
Samuel Shem: Part II
Mr. Shem is the author of the satirical novel The House of God, which examined medical residency in the United States in the 1970s.
Mr. Shem is the author of the satirical novel The House of God, which examined medical residency in the United States in the 1970s.
Mr. Shem is the author of the satirical novel The House of God, which examined medical residency in the United States in the 1970s.
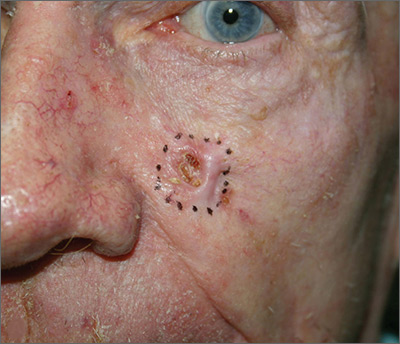
Growing lesion on cheek
Figure 1
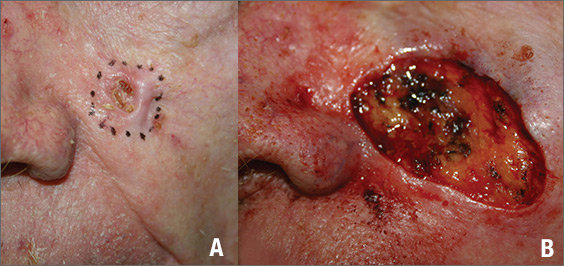
The FP suspected that this was a basal cell carcinoma (BCC) or squamous cell carcinoma. He leaned toward a BCC because of the pearly border on the edge, but knew that a biopsy diagnosis was needed before planning definitive treatment.
The FP recommended performing a shave biopsy that day. (See the Watch & Learn video on “Shave biopsy.”) After obtaining patient consent, he injected 1% lidocaine with epinephrine and waited for the epinephrine to work. He performed the shave biopsy with a Dermablade, and used a cotton-tipped applicator to vigorously apply aluminum chloride to the site. He used a twisting motion and pressure to achieve hemostasis. The bleeding stopped, and the FP dressed the lesion with petrolatum and some gauze. Dermatopathology revealed a sclerosing BCC.
The FP realized this was an aggressive tumor and referred the patient for Mohs surgery. The surgery required 4 excisions to get clean margins (FIGURE 1B). The usual 4- to 5-mm margins with an elliptical excision would not have removed the full tumor.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Figure 1

The FP suspected that this was a basal cell carcinoma (BCC) or squamous cell carcinoma. He leaned toward a BCC because of the pearly border on the edge, but knew that a biopsy diagnosis was needed before planning definitive treatment.
The FP recommended performing a shave biopsy that day. (See the Watch & Learn video on “Shave biopsy.”) After obtaining patient consent, he injected 1% lidocaine with epinephrine and waited for the epinephrine to work. He performed the shave biopsy with a Dermablade, and used a cotton-tipped applicator to vigorously apply aluminum chloride to the site. He used a twisting motion and pressure to achieve hemostasis. The bleeding stopped, and the FP dressed the lesion with petrolatum and some gauze. Dermatopathology revealed a sclerosing BCC.
The FP realized this was an aggressive tumor and referred the patient for Mohs surgery. The surgery required 4 excisions to get clean margins (FIGURE 1B). The usual 4- to 5-mm margins with an elliptical excision would not have removed the full tumor.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
Figure 1

The FP suspected that this was a basal cell carcinoma (BCC) or squamous cell carcinoma. He leaned toward a BCC because of the pearly border on the edge, but knew that a biopsy diagnosis was needed before planning definitive treatment.
The FP recommended performing a shave biopsy that day. (See the Watch & Learn video on “Shave biopsy.”) After obtaining patient consent, he injected 1% lidocaine with epinephrine and waited for the epinephrine to work. He performed the shave biopsy with a Dermablade, and used a cotton-tipped applicator to vigorously apply aluminum chloride to the site. He used a twisting motion and pressure to achieve hemostasis. The bleeding stopped, and the FP dressed the lesion with petrolatum and some gauze. Dermatopathology revealed a sclerosing BCC.
The FP realized this was an aggressive tumor and referred the patient for Mohs surgery. The surgery required 4 excisions to get clean margins (FIGURE 1B). The usual 4- to 5-mm margins with an elliptical excision would not have removed the full tumor.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Karnes J, Usatine R. Basal cell carcinoma. In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas of Family Medicine. 2nd ed. New York, NY: McGraw-Hill; 2013:989-998.
To learn more about the Color Atlas of Family Medicine, see: www.amazon.com/Color-Family-Medicine-Richard-Usatine/dp/0071769641/.
You can now get the second edition of the Color Atlas of Family Medicine as an app by clicking on this link: usatinemedia.com.
The price we pay for trying to see more and more patients
In the October 2018 issue of Medscape Business of Medicine, the question was asked, “How can you practice quality medicine if you’re being asked to see patients every 15 minutes or less?”
I’m pretty sure the answer is, “you can’t.”
Yet, this is what many doctors are asked to do just to make ends meet. The majority of everyday medicine is, and always will be, a thinking game. It takes time to piece together the clues from a history and exam and decide what tests and/or treatment are the next step.
This ain’t easy. Even the shortest residencies require a combined 7 years of medical school and postgrad training. Experience and learning makes us all faster, but then the number of things that you can handle in 15 minutes is minimal. And that doesn’t even include the time needed to answer patient or family questions (which can be quite a lot) write up or transmit test orders or a prescription, and, inevitably, document the entire encounter in a meaningful way.
I don’t see patients at such a breakneck speed in my office, and yet I still end up doing most of my dictations after (or before) office hours.
In spite of lip service by politicians and administrators to correct the issue, medicine still continues to penalize those services that require thinking. And this task is the center of being a good doctor – and always has been.
Procedures are more lucrative, but imagine how my colleagues in neurosurgery would react if they were given a similar time limit on cases: A new patient has to be on the table every 15-30 minutes, and in that time you have to open, do the surgery, close, meet with family, and document the whole thing. Then get back in the OR (scrub, first) before the next case. Doesn’t matter whether you’re doing a lumbar fusion, glioma resection, or carotid endarterectomy. Those are the time limits. You get 30 minutes for lunch and to return calls. The administrator said so.
And this is where medicine continues to go. Overhead costs keep rising, and, for most docs, the only way they know to keep up is to keep cramming more patients into the day. Nobody wants to practice shoddy, hurried medicine, but neither do they want to lose their jobs to the next hungry graduate or close down a practice they spent years building.
I wish I had an answer. In fact, I think most of us do, but not one that will make patients, administrators, and doctors all happy. So the spiral continues.
And that isn’t good for patients, the people at the center of this job.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
In the October 2018 issue of Medscape Business of Medicine, the question was asked, “How can you practice quality medicine if you’re being asked to see patients every 15 minutes or less?”
I’m pretty sure the answer is, “you can’t.”
Yet, this is what many doctors are asked to do just to make ends meet. The majority of everyday medicine is, and always will be, a thinking game. It takes time to piece together the clues from a history and exam and decide what tests and/or treatment are the next step.
This ain’t easy. Even the shortest residencies require a combined 7 years of medical school and postgrad training. Experience and learning makes us all faster, but then the number of things that you can handle in 15 minutes is minimal. And that doesn’t even include the time needed to answer patient or family questions (which can be quite a lot) write up or transmit test orders or a prescription, and, inevitably, document the entire encounter in a meaningful way.
I don’t see patients at such a breakneck speed in my office, and yet I still end up doing most of my dictations after (or before) office hours.
In spite of lip service by politicians and administrators to correct the issue, medicine still continues to penalize those services that require thinking. And this task is the center of being a good doctor – and always has been.
Procedures are more lucrative, but imagine how my colleagues in neurosurgery would react if they were given a similar time limit on cases: A new patient has to be on the table every 15-30 minutes, and in that time you have to open, do the surgery, close, meet with family, and document the whole thing. Then get back in the OR (scrub, first) before the next case. Doesn’t matter whether you’re doing a lumbar fusion, glioma resection, or carotid endarterectomy. Those are the time limits. You get 30 minutes for lunch and to return calls. The administrator said so.
And this is where medicine continues to go. Overhead costs keep rising, and, for most docs, the only way they know to keep up is to keep cramming more patients into the day. Nobody wants to practice shoddy, hurried medicine, but neither do they want to lose their jobs to the next hungry graduate or close down a practice they spent years building.
I wish I had an answer. In fact, I think most of us do, but not one that will make patients, administrators, and doctors all happy. So the spiral continues.
And that isn’t good for patients, the people at the center of this job.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
In the October 2018 issue of Medscape Business of Medicine, the question was asked, “How can you practice quality medicine if you’re being asked to see patients every 15 minutes or less?”
I’m pretty sure the answer is, “you can’t.”
Yet, this is what many doctors are asked to do just to make ends meet. The majority of everyday medicine is, and always will be, a thinking game. It takes time to piece together the clues from a history and exam and decide what tests and/or treatment are the next step.
This ain’t easy. Even the shortest residencies require a combined 7 years of medical school and postgrad training. Experience and learning makes us all faster, but then the number of things that you can handle in 15 minutes is minimal. And that doesn’t even include the time needed to answer patient or family questions (which can be quite a lot) write up or transmit test orders or a prescription, and, inevitably, document the entire encounter in a meaningful way.
I don’t see patients at such a breakneck speed in my office, and yet I still end up doing most of my dictations after (or before) office hours.
In spite of lip service by politicians and administrators to correct the issue, medicine still continues to penalize those services that require thinking. And this task is the center of being a good doctor – and always has been.
Procedures are more lucrative, but imagine how my colleagues in neurosurgery would react if they were given a similar time limit on cases: A new patient has to be on the table every 15-30 minutes, and in that time you have to open, do the surgery, close, meet with family, and document the whole thing. Then get back in the OR (scrub, first) before the next case. Doesn’t matter whether you’re doing a lumbar fusion, glioma resection, or carotid endarterectomy. Those are the time limits. You get 30 minutes for lunch and to return calls. The administrator said so.
And this is where medicine continues to go. Overhead costs keep rising, and, for most docs, the only way they know to keep up is to keep cramming more patients into the day. Nobody wants to practice shoddy, hurried medicine, but neither do they want to lose their jobs to the next hungry graduate or close down a practice they spent years building.
I wish I had an answer. In fact, I think most of us do, but not one that will make patients, administrators, and doctors all happy. So the spiral continues.
And that isn’t good for patients, the people at the center of this job.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
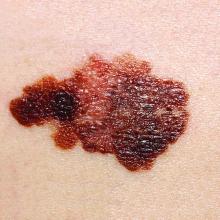
New pregnancy, genetic testing guidance added to AAD’s melanoma guidelines
Pregnancy does not necessarily increase a woman’s risk for melanoma, nor is it clear that becoming pregnant affects melanoma’s disease course, according to current evidence. This guidance is among several updates added to newly released guidelines for managing patients with primary cutaneous melanoma.
also addressed the burgeoning field of genetic testing for cancer in the guidelines, which were published online on Nov. 1. Although there may be a hereditary component to some melanomas, genetic testing may not be appropriate for all patients, and any formal genetic testing should be carried out only after individualized education and counseling, according to the updates.
However, the guidelines make it clear that all patients whose family history includes melanoma should be counseled about their genetic risk.
As with genetic testing, counseling regarding future pregnancies for women with melanoma, or a history of melanoma, should be personalized and account for individual history and melanoma risk, according to the new guidelines. Since evidence is lacking that pregnancy affects the course of melanoma, physicians caring for pregnant women with melanoma should first look at patient and the disease characteristics. The addition of detailed guidance regarding pregnancy reflects research showing that CM is the most common malignancy seen in pregnancy, amounting to nearly one-third of the malignancies that arise in pregnancy. “Although the incidence of CM is generally higher in men, it is higher in younger women than in men, most notably during women’s reproductive years,” wrote Susan M. Swetter, MD, and her guideline coauthors.
“Melanoma is the deadliest form of skin cancer, and we hope these guidelines will help dermatologists and other physicians enhance their delivery of life-saving treatment to patients,” Dr. Swetter said in a press release announcing the guideline updates. Dr. Swetter, professor of dermatology and director of the pigmented lesion and melanoma program at Stanford (Calif.) University Medical Center and Cancer Institute, led the working group that developed the guidelines. “In order to provide the best possible resource for practitioners, we reviewed the latest scientific data and addressed certain topics that weren’t covered in the AAD’s previous melanoma guidelines,” she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. Adjuvant topical therapies or radiation, say the guidelines, can be considered as second-line care, but only in limited situations in which surgery is not feasible. Staged excision techniques, such as Mohs surgery, also may be considered for certain types of melanoma and in certain body areas.
In an interview, Dr. Swetter also said that is critical that the updated guidelines have been harmonized with changes made in the American Joint Committee on Cancer’s 8th edition of its melanoma staging manual. Key points for dermatologists to understand that reporting of Breslow thickness to the nearest 1/10th decimal point (over the nearest 1/100th), such that a melanoma measuring 0.75-0.84 mm in thickness would be reported as 0.8 mm depth and one between 0.95-1.04 mm would be rounded to 1 mm.
The main changes regarding staging of thin (T1) melanoma – that is less than or equal to 1 mm – is that the 0.8 mm thickness is the threshold for a T1a melanoma (now classified as less than 0.8 mm without ulceration), whereas T1b is now 0.8 – 1.0 mm thickness with or w/out ulceration or less than 0.8 mm thickness with ulceration. A T1a melanoma generally is not considered appropriate for staging of the regional lymph nodes with sentinel lymph node biopsy (with exceptions noted in the guideline), whereas a T1b melanoma may be considered for SLNB staging – though rates of SLN positivity remain relatively low in the T1b group.”
Dr. Swetter also emphasized that histologic ulceration of the primary tumor was affirmed as an indicator of worse prognosis; mitotic rate, although removed from T1 staging, is still tracked by pathologists and still seen as an independent predictor of worse prognosis, according to the 8th edition, she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. “Mohs micrographic surgery and other staged excision techniques can provide exhaustive peripheral margin histologic assessment for melanoma in situ, lentigo maligna type and tissue sparing in anatomically constrained sites,” Dr. Swetter said. “Current data are insufficient to recommend Mohs surgery for invasive cutaneous melanoma, in which the use of surgical margins less than 1 cm has not been adequately studied,” she cautioned.
Reinforcing the importance of surgery as the primary treatment for melanoma, Dr. Swetter clarified that “Nonsurgical approaches (imiquimod and traditional forms of radiation therapy) should be considered [only] if surgery is impractical or contraindicated, and only for melanoma in situ, lentigo maligna type, as cure rates are lower.”
In terms of other therapies, the guideline working group found insufficient evidence to recommend electronic brachytherapy for melanoma.
Assessment of novel diagnostic and molecular imaging modalities was not the primary focus of the AAD guidelines, Dr. Swetter pointed out. Looking to the future, though, she added that the hope is “that these prebiopsy modalities can one day reduce unnecessary biopsies from being done” in the clinic.
Other knowledge gaps cited by the working group included several related to pathology, including determination of appropriate margin control in some lesion types, and the quest to reduce inter-reader variability in histopathologic diagnosis of melanocytic tissue samples. However, noted Dr. Swetter and her coauthors, the rapid pace of genomic medicine advances “may make many of the aforementioned issues obsolete” before the next guideline update.
In the interview, Dr. Swetter said that the guidelines reflect evolving thinking about melanoma in the context of a rapidly growing field. “Only in the last year have effective, more tolerable adjuvant therapies been [Food and Drug Administration] approved for patients with resected stage III melanoma, including patients with regional lymph node disease detected via sentinel lymph node biopsy. The hope is that less invasive procedures for melanoma will be performed in the future, and replaced by better drugs and novel techniques.”
Dr. Swetter reported that she had no relevant financial disclosures; several working group members reported multiple financial relationships with pharmaceutical, diagnostic, and imaging companies. Working group members were recused from discussion of guidelines where their particular relationships might pose a conflict of interest.
SOURCE: Swetter S. et al. J Am Acad Dermatol. 2011 Nov;65(5):1032-47.
Pregnancy does not necessarily increase a woman’s risk for melanoma, nor is it clear that becoming pregnant affects melanoma’s disease course, according to current evidence. This guidance is among several updates added to newly released guidelines for managing patients with primary cutaneous melanoma.
also addressed the burgeoning field of genetic testing for cancer in the guidelines, which were published online on Nov. 1. Although there may be a hereditary component to some melanomas, genetic testing may not be appropriate for all patients, and any formal genetic testing should be carried out only after individualized education and counseling, according to the updates.
However, the guidelines make it clear that all patients whose family history includes melanoma should be counseled about their genetic risk.
As with genetic testing, counseling regarding future pregnancies for women with melanoma, or a history of melanoma, should be personalized and account for individual history and melanoma risk, according to the new guidelines. Since evidence is lacking that pregnancy affects the course of melanoma, physicians caring for pregnant women with melanoma should first look at patient and the disease characteristics. The addition of detailed guidance regarding pregnancy reflects research showing that CM is the most common malignancy seen in pregnancy, amounting to nearly one-third of the malignancies that arise in pregnancy. “Although the incidence of CM is generally higher in men, it is higher in younger women than in men, most notably during women’s reproductive years,” wrote Susan M. Swetter, MD, and her guideline coauthors.
“Melanoma is the deadliest form of skin cancer, and we hope these guidelines will help dermatologists and other physicians enhance their delivery of life-saving treatment to patients,” Dr. Swetter said in a press release announcing the guideline updates. Dr. Swetter, professor of dermatology and director of the pigmented lesion and melanoma program at Stanford (Calif.) University Medical Center and Cancer Institute, led the working group that developed the guidelines. “In order to provide the best possible resource for practitioners, we reviewed the latest scientific data and addressed certain topics that weren’t covered in the AAD’s previous melanoma guidelines,” she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. Adjuvant topical therapies or radiation, say the guidelines, can be considered as second-line care, but only in limited situations in which surgery is not feasible. Staged excision techniques, such as Mohs surgery, also may be considered for certain types of melanoma and in certain body areas.
In an interview, Dr. Swetter also said that is critical that the updated guidelines have been harmonized with changes made in the American Joint Committee on Cancer’s 8th edition of its melanoma staging manual. Key points for dermatologists to understand that reporting of Breslow thickness to the nearest 1/10th decimal point (over the nearest 1/100th), such that a melanoma measuring 0.75-0.84 mm in thickness would be reported as 0.8 mm depth and one between 0.95-1.04 mm would be rounded to 1 mm.
The main changes regarding staging of thin (T1) melanoma – that is less than or equal to 1 mm – is that the 0.8 mm thickness is the threshold for a T1a melanoma (now classified as less than 0.8 mm without ulceration), whereas T1b is now 0.8 – 1.0 mm thickness with or w/out ulceration or less than 0.8 mm thickness with ulceration. A T1a melanoma generally is not considered appropriate for staging of the regional lymph nodes with sentinel lymph node biopsy (with exceptions noted in the guideline), whereas a T1b melanoma may be considered for SLNB staging – though rates of SLN positivity remain relatively low in the T1b group.”
Dr. Swetter also emphasized that histologic ulceration of the primary tumor was affirmed as an indicator of worse prognosis; mitotic rate, although removed from T1 staging, is still tracked by pathologists and still seen as an independent predictor of worse prognosis, according to the 8th edition, she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. “Mohs micrographic surgery and other staged excision techniques can provide exhaustive peripheral margin histologic assessment for melanoma in situ, lentigo maligna type and tissue sparing in anatomically constrained sites,” Dr. Swetter said. “Current data are insufficient to recommend Mohs surgery for invasive cutaneous melanoma, in which the use of surgical margins less than 1 cm has not been adequately studied,” she cautioned.
Reinforcing the importance of surgery as the primary treatment for melanoma, Dr. Swetter clarified that “Nonsurgical approaches (imiquimod and traditional forms of radiation therapy) should be considered [only] if surgery is impractical or contraindicated, and only for melanoma in situ, lentigo maligna type, as cure rates are lower.”
In terms of other therapies, the guideline working group found insufficient evidence to recommend electronic brachytherapy for melanoma.
Assessment of novel diagnostic and molecular imaging modalities was not the primary focus of the AAD guidelines, Dr. Swetter pointed out. Looking to the future, though, she added that the hope is “that these prebiopsy modalities can one day reduce unnecessary biopsies from being done” in the clinic.
Other knowledge gaps cited by the working group included several related to pathology, including determination of appropriate margin control in some lesion types, and the quest to reduce inter-reader variability in histopathologic diagnosis of melanocytic tissue samples. However, noted Dr. Swetter and her coauthors, the rapid pace of genomic medicine advances “may make many of the aforementioned issues obsolete” before the next guideline update.
In the interview, Dr. Swetter said that the guidelines reflect evolving thinking about melanoma in the context of a rapidly growing field. “Only in the last year have effective, more tolerable adjuvant therapies been [Food and Drug Administration] approved for patients with resected stage III melanoma, including patients with regional lymph node disease detected via sentinel lymph node biopsy. The hope is that less invasive procedures for melanoma will be performed in the future, and replaced by better drugs and novel techniques.”
Dr. Swetter reported that she had no relevant financial disclosures; several working group members reported multiple financial relationships with pharmaceutical, diagnostic, and imaging companies. Working group members were recused from discussion of guidelines where their particular relationships might pose a conflict of interest.
SOURCE: Swetter S. et al. J Am Acad Dermatol. 2011 Nov;65(5):1032-47.
Pregnancy does not necessarily increase a woman’s risk for melanoma, nor is it clear that becoming pregnant affects melanoma’s disease course, according to current evidence. This guidance is among several updates added to newly released guidelines for managing patients with primary cutaneous melanoma.
also addressed the burgeoning field of genetic testing for cancer in the guidelines, which were published online on Nov. 1. Although there may be a hereditary component to some melanomas, genetic testing may not be appropriate for all patients, and any formal genetic testing should be carried out only after individualized education and counseling, according to the updates.
However, the guidelines make it clear that all patients whose family history includes melanoma should be counseled about their genetic risk.
As with genetic testing, counseling regarding future pregnancies for women with melanoma, or a history of melanoma, should be personalized and account for individual history and melanoma risk, according to the new guidelines. Since evidence is lacking that pregnancy affects the course of melanoma, physicians caring for pregnant women with melanoma should first look at patient and the disease characteristics. The addition of detailed guidance regarding pregnancy reflects research showing that CM is the most common malignancy seen in pregnancy, amounting to nearly one-third of the malignancies that arise in pregnancy. “Although the incidence of CM is generally higher in men, it is higher in younger women than in men, most notably during women’s reproductive years,” wrote Susan M. Swetter, MD, and her guideline coauthors.
“Melanoma is the deadliest form of skin cancer, and we hope these guidelines will help dermatologists and other physicians enhance their delivery of life-saving treatment to patients,” Dr. Swetter said in a press release announcing the guideline updates. Dr. Swetter, professor of dermatology and director of the pigmented lesion and melanoma program at Stanford (Calif.) University Medical Center and Cancer Institute, led the working group that developed the guidelines. “In order to provide the best possible resource for practitioners, we reviewed the latest scientific data and addressed certain topics that weren’t covered in the AAD’s previous melanoma guidelines,” she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. Adjuvant topical therapies or radiation, say the guidelines, can be considered as second-line care, but only in limited situations in which surgery is not feasible. Staged excision techniques, such as Mohs surgery, also may be considered for certain types of melanoma and in certain body areas.
In an interview, Dr. Swetter also said that is critical that the updated guidelines have been harmonized with changes made in the American Joint Committee on Cancer’s 8th edition of its melanoma staging manual. Key points for dermatologists to understand that reporting of Breslow thickness to the nearest 1/10th decimal point (over the nearest 1/100th), such that a melanoma measuring 0.75-0.84 mm in thickness would be reported as 0.8 mm depth and one between 0.95-1.04 mm would be rounded to 1 mm.
The main changes regarding staging of thin (T1) melanoma – that is less than or equal to 1 mm – is that the 0.8 mm thickness is the threshold for a T1a melanoma (now classified as less than 0.8 mm without ulceration), whereas T1b is now 0.8 – 1.0 mm thickness with or w/out ulceration or less than 0.8 mm thickness with ulceration. A T1a melanoma generally is not considered appropriate for staging of the regional lymph nodes with sentinel lymph node biopsy (with exceptions noted in the guideline), whereas a T1b melanoma may be considered for SLNB staging – though rates of SLN positivity remain relatively low in the T1b group.”
Dr. Swetter also emphasized that histologic ulceration of the primary tumor was affirmed as an indicator of worse prognosis; mitotic rate, although removed from T1 staging, is still tracked by pathologists and still seen as an independent predictor of worse prognosis, according to the 8th edition, she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. “Mohs micrographic surgery and other staged excision techniques can provide exhaustive peripheral margin histologic assessment for melanoma in situ, lentigo maligna type and tissue sparing in anatomically constrained sites,” Dr. Swetter said. “Current data are insufficient to recommend Mohs surgery for invasive cutaneous melanoma, in which the use of surgical margins less than 1 cm has not been adequately studied,” she cautioned.
Reinforcing the importance of surgery as the primary treatment for melanoma, Dr. Swetter clarified that “Nonsurgical approaches (imiquimod and traditional forms of radiation therapy) should be considered [only] if surgery is impractical or contraindicated, and only for melanoma in situ, lentigo maligna type, as cure rates are lower.”
In terms of other therapies, the guideline working group found insufficient evidence to recommend electronic brachytherapy for melanoma.
Assessment of novel diagnostic and molecular imaging modalities was not the primary focus of the AAD guidelines, Dr. Swetter pointed out. Looking to the future, though, she added that the hope is “that these prebiopsy modalities can one day reduce unnecessary biopsies from being done” in the clinic.
Other knowledge gaps cited by the working group included several related to pathology, including determination of appropriate margin control in some lesion types, and the quest to reduce inter-reader variability in histopathologic diagnosis of melanocytic tissue samples. However, noted Dr. Swetter and her coauthors, the rapid pace of genomic medicine advances “may make many of the aforementioned issues obsolete” before the next guideline update.
In the interview, Dr. Swetter said that the guidelines reflect evolving thinking about melanoma in the context of a rapidly growing field. “Only in the last year have effective, more tolerable adjuvant therapies been [Food and Drug Administration] approved for patients with resected stage III melanoma, including patients with regional lymph node disease detected via sentinel lymph node biopsy. The hope is that less invasive procedures for melanoma will be performed in the future, and replaced by better drugs and novel techniques.”
Dr. Swetter reported that she had no relevant financial disclosures; several working group members reported multiple financial relationships with pharmaceutical, diagnostic, and imaging companies. Working group members were recused from discussion of guidelines where their particular relationships might pose a conflict of interest.
SOURCE: Swetter S. et al. J Am Acad Dermatol. 2011 Nov;65(5):1032-47.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY