User login
Circulating Tumor DNA Testing and Liquid Biopsy: The Future for Precision Medicine and Guided Targeted Therapy for Breast Cancer?
The current standard for breast cancer screening (for non–high-risk patients) is an annual or semiannual mammogram for women aged 40 and older.1 However, mammography-based screening can give false-positive or false-negative results. This can lead to excessive use of invasive tissue biopsies and unnecessary exposure to ionizing radiation—which can also become expensive and time-consuming for patients.2
Both normal and cancerous cells shed cell-free DNA (cfDNA) into the blood circulation.3 Circulating tumor DNA (ctDNA) are fragments of DNA derived from tumor cells that circulate in the blood together with cfDNA. The ctDNA originates directly from a tumor or from circulating tumor cells (and carries information from the tumor cell genome), whereas cfDNA enters the bloodstream after apoptosis or necrosis and carries genome-wide DNA information. The amount of ctDNA in the blood has been shown to be elevated in patients with cancer.3 Different cancers release varying levels of ctDNA; the amount of ctDNA released depends on the number of tumor cells that are in senescence vs undergoing apoptosis.4
The possibility of incorporating this biomarker obtained from a “liquid biopsy” is currently being studied and will hopefully become a standard of care for breast cancer screening and monitoring. The liquid biopsy detects ctDNA that has been released into the bloodstream from tumor regions and helps identify intratumoral heterogeneity and clonal evolution.5 Additionally, sequencing tumor DNA has opened new possibilities for precision oncology.6 Detection of somatic gene mutations, amplifications, and gene fusions helps to deliver targeted therapies.6 Analysis of potential somatic mutations in ctDNA, in combination with cfDNA levels, can help capture clinically relevant information beyond single genetic alterations and tumor fraction, potentially improving the accuracy of early detection and screening for breast cancer.
Recent advances in ctDNA testing technology have made it more accurate and reliable. ctDNA testing has several benefits, including early detection of cancer (detecting ctDNA at low levels)7; monitoring of tumor dynamics, therapeutic response, and residual disease8; as well as analysis of the evolution of genetic or epigenetic alterations characterizing the tumor.9 Its noninvasiveness, rapidity, and low cost allow for longitudinal monitoring of cancer in real time, potentially capturing tumor heterogeneity.10,11
The liquid biopsy potentially can give more options for therapeutic monitoring for breast cancer and may mirror clinically relevant genetic alterations that occur in all tumor tissues. Liquid biopsy offers many advantages. It allows for the detection of minimal residual disease and micrometastatic disease that may be difficult to detect with a traditional tissue biopsy.12 Liquid biopsy detects ctDNA that has been released into the bloodstream from multiple tumor regions and allows the possibility of identifying intratumoral heterogeneity and clonal evolution.5 It can also detect small quantitative variations within the blood, enabling real-time surveillance.
The liquid biopsy can offer earlier and easier access to some tumor-based genetic information at any given timepoint and can replace a tumor tissue biopsy in some cases, helping to avoid delays and complications of a solid tumor invasive biopsy procedure. This is especially relevant in the metastatic setting, in which ctDNA might be the only available genetic material from tumors.13 Tissue biopsy can only provide a static and spatially limited view of the disease at the time of sampling; ctDNA analysis could potentially reflect the genetic alterations that occur in all metastatic breast cancer sites over time.14,15 Furthermore, machine learning of multi-gene signatures, obtained from ctDNA, can possibly identify complex biological features, including measures of tumor proliferation and estrogen receptor signaling, similar to direct tumor tissue DNA or RNA profiling.16
ctDNA testing is currently being studied to monitor patients who have been diagnosed with breast cancer. Small retrospective studies have shown that detection of ctDNA in plasma, after patients have completed therapy for early-stage breast cancer, is associated with a very high risk of relapse.17
Ongoing studies are examining the tailoring of adjuvant treatment based on ctDNA. If these trials are successful, certain aspects of adjuvant treatment could be lessened, or omitted, for patients who have undetectable ctDNA or intensified for patients who have detectable ctDNA after definitive treatments. This could personalize treatment specifically to the patient.
The detection and persistence of ctDNA in the middle of neoadjuvant systemic therapy may have the potential to negatively predict response to treatment and identify patients who will not achieve pathologic complete response. This may have the potential to aid in clinical decision-making for treatment escalation in these nonresponders.18
Despite these distinct characteristics, the low levels of ctDNA found in early-stage disease, along with the lack of ctDNA shedding from some tumors, can further complicate or impede detection of recurrence in early-stage breast cancer. Testing is further complicated by hematologic genetic alterations.5 The limitation of ctDNA approaches is that these techniques only detect known mutations in certain genes, so patients without these mutations could be overlooked, limiting the application of this technology.19
Overall, ctDNA testing represents a promising area of research for the diagnosis, treatment, and monitoring of breast cancer. While more research is needed to fully understand its potential, the advances in this technology are certainly exciting and could lead to significant improvements in patient outcomes. It is hopeful that in the near future, ctDNA testing from liquid biopsy could become a standard of care in breast cancer screening, ultimately helping clinicians to personalize treatment therapies and improve patient outcomes when treating patients with breast cancer.
1. Oeffinger KC, Fontham ETH, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599-1614.
2. Zubor P, Kubatka P, Kajo K, et al. Why the gold standard approach by mammography demands extension by multiomics? Application of liquid biopsy miRNA profiles to breast cancer disease management. Int J Mol Sci. 2019;20(12):E2878.
3. Thierry AR, El Messaoudi S, Gahan PB, Anker P, Stroun M. Origins, structures, and functions of circulating DNA in oncology. Cancer Metastasis Rev. 2016;35(3):347-376.
4. Rostami A, Lambie M, Yu CW, Stambolic V, Waldron JN, Bratman SV. Senescence, necrosis, and apoptosis govern circulating cell-free DNA release kinetics. Cell Rep. 2020;31(13):107830.
5. De Rubis G, Rajeev Krishnan S, Bebawy M. Liquid biopsies in cancer diagnosis, monitoring, and prognosis. Trends Pharmacol Sci. 2019;40(3):172-186.
6. Mateo J, Chakravarty D, Dienstmann R, et al. A framework to rank genomic alterations as targets for cancer precision medicine: the ESMO Scale for Clinical Actionability of molecular Targets (ESCAT). Ann Oncol. 2018;29:1895-1902.
7. Wang J, Han X, Sun Y. DNA methylation signatures in circulating cell-free DNA as biomarkers for the early detection of cancer. Sci China Life Sci. 2017;60(4):356-362.
8. Dawson S-J, Tsui DWY, Murtaza M, et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N Engl J Med. 2013;368(13):1199-1209.
9. Diaz Jr LA, Bardelli A. Liquid biopsies: genotyping circulating tumor DNA. J Clin Oncol. 2014;32(6):579-586.
10. Oxnard GR, Paweletz CP, Kuang Y, et al. Noninvasive detection of response and resistance in EGFR-mutant lung cancer using quantitative next-generation genotyping of cell-free plasma DNA. Clin Cancer Res. 2014;20(6):1698-1705.
11. Jamal-Hanjani M, Wilson GA, Horswell S, et al. Detection of ubiquitous and heterogeneous mutations in cell-free DNA from patients with early-stage non-small-cell lung cancer. Ann Oncol. 2016;27(5):862-867.
12. Fiala C, Diamandis EP. Utility of circulating tumor DNA in cancer diagnostics with
13. Xia Y, Fan C, Hoadley KA, Parker JS, Perou CM. Genetic determinants of the molecular portraits of epithelial cancers. Nat Commun. 2019;10(1):5666.
14. Wan JCM, Massie C, Garcia-Corbacho J, et al. Liquid biopsies come of age: towards implementation of circulating tumour DNA. Nat Rev Cancer. 2017;17(4):223-238.
15. Boldrin E, Nardo G, Zulato E, et al. Detection of loss of heterozygosity in cfDNA of advanced EGFR- or KRAS-mutated non-small-cell lung cancer patients. Int J Mol Sci. 2019;21(1):66.
16. Prat A, Brasó-Maristany F, Martínez-Sáez O, et al. Circulating tumor DNA reveals complex biological features with clinical relevance in metastatic breast cancer. Nat Commun. 2023;14(1):1157.
17. Coombes RC, Page K, Salari R, et al. Personalized detection of circulating tumor DNA antedates breast cancer metastatic recurrence. Clin Cancer Res. 2019;25(14):4255-4263.
18. Zhou Q, Gampenrieder SP, Frantal S, et al. Persistence of ctDNA in patients with breast cancer during neoadjuvant treatment is a significant predictor of poor tumor response. Clin Cancer Res. 2022;28(4):697-707.
19. Lin C, Liu X, Zheng B, Ke R, Tzeng C-M. Liquid biopsy, ctDNA diagnosis through NGS. Life (Basel). 2021;11(9):890.
The current standard for breast cancer screening (for non–high-risk patients) is an annual or semiannual mammogram for women aged 40 and older.1 However, mammography-based screening can give false-positive or false-negative results. This can lead to excessive use of invasive tissue biopsies and unnecessary exposure to ionizing radiation—which can also become expensive and time-consuming for patients.2
Both normal and cancerous cells shed cell-free DNA (cfDNA) into the blood circulation.3 Circulating tumor DNA (ctDNA) are fragments of DNA derived from tumor cells that circulate in the blood together with cfDNA. The ctDNA originates directly from a tumor or from circulating tumor cells (and carries information from the tumor cell genome), whereas cfDNA enters the bloodstream after apoptosis or necrosis and carries genome-wide DNA information. The amount of ctDNA in the blood has been shown to be elevated in patients with cancer.3 Different cancers release varying levels of ctDNA; the amount of ctDNA released depends on the number of tumor cells that are in senescence vs undergoing apoptosis.4
The possibility of incorporating this biomarker obtained from a “liquid biopsy” is currently being studied and will hopefully become a standard of care for breast cancer screening and monitoring. The liquid biopsy detects ctDNA that has been released into the bloodstream from tumor regions and helps identify intratumoral heterogeneity and clonal evolution.5 Additionally, sequencing tumor DNA has opened new possibilities for precision oncology.6 Detection of somatic gene mutations, amplifications, and gene fusions helps to deliver targeted therapies.6 Analysis of potential somatic mutations in ctDNA, in combination with cfDNA levels, can help capture clinically relevant information beyond single genetic alterations and tumor fraction, potentially improving the accuracy of early detection and screening for breast cancer.
Recent advances in ctDNA testing technology have made it more accurate and reliable. ctDNA testing has several benefits, including early detection of cancer (detecting ctDNA at low levels)7; monitoring of tumor dynamics, therapeutic response, and residual disease8; as well as analysis of the evolution of genetic or epigenetic alterations characterizing the tumor.9 Its noninvasiveness, rapidity, and low cost allow for longitudinal monitoring of cancer in real time, potentially capturing tumor heterogeneity.10,11
The liquid biopsy potentially can give more options for therapeutic monitoring for breast cancer and may mirror clinically relevant genetic alterations that occur in all tumor tissues. Liquid biopsy offers many advantages. It allows for the detection of minimal residual disease and micrometastatic disease that may be difficult to detect with a traditional tissue biopsy.12 Liquid biopsy detects ctDNA that has been released into the bloodstream from multiple tumor regions and allows the possibility of identifying intratumoral heterogeneity and clonal evolution.5 It can also detect small quantitative variations within the blood, enabling real-time surveillance.
The liquid biopsy can offer earlier and easier access to some tumor-based genetic information at any given timepoint and can replace a tumor tissue biopsy in some cases, helping to avoid delays and complications of a solid tumor invasive biopsy procedure. This is especially relevant in the metastatic setting, in which ctDNA might be the only available genetic material from tumors.13 Tissue biopsy can only provide a static and spatially limited view of the disease at the time of sampling; ctDNA analysis could potentially reflect the genetic alterations that occur in all metastatic breast cancer sites over time.14,15 Furthermore, machine learning of multi-gene signatures, obtained from ctDNA, can possibly identify complex biological features, including measures of tumor proliferation and estrogen receptor signaling, similar to direct tumor tissue DNA or RNA profiling.16
ctDNA testing is currently being studied to monitor patients who have been diagnosed with breast cancer. Small retrospective studies have shown that detection of ctDNA in plasma, after patients have completed therapy for early-stage breast cancer, is associated with a very high risk of relapse.17
Ongoing studies are examining the tailoring of adjuvant treatment based on ctDNA. If these trials are successful, certain aspects of adjuvant treatment could be lessened, or omitted, for patients who have undetectable ctDNA or intensified for patients who have detectable ctDNA after definitive treatments. This could personalize treatment specifically to the patient.
The detection and persistence of ctDNA in the middle of neoadjuvant systemic therapy may have the potential to negatively predict response to treatment and identify patients who will not achieve pathologic complete response. This may have the potential to aid in clinical decision-making for treatment escalation in these nonresponders.18
Despite these distinct characteristics, the low levels of ctDNA found in early-stage disease, along with the lack of ctDNA shedding from some tumors, can further complicate or impede detection of recurrence in early-stage breast cancer. Testing is further complicated by hematologic genetic alterations.5 The limitation of ctDNA approaches is that these techniques only detect known mutations in certain genes, so patients without these mutations could be overlooked, limiting the application of this technology.19
Overall, ctDNA testing represents a promising area of research for the diagnosis, treatment, and monitoring of breast cancer. While more research is needed to fully understand its potential, the advances in this technology are certainly exciting and could lead to significant improvements in patient outcomes. It is hopeful that in the near future, ctDNA testing from liquid biopsy could become a standard of care in breast cancer screening, ultimately helping clinicians to personalize treatment therapies and improve patient outcomes when treating patients with breast cancer.
The current standard for breast cancer screening (for non–high-risk patients) is an annual or semiannual mammogram for women aged 40 and older.1 However, mammography-based screening can give false-positive or false-negative results. This can lead to excessive use of invasive tissue biopsies and unnecessary exposure to ionizing radiation—which can also become expensive and time-consuming for patients.2
Both normal and cancerous cells shed cell-free DNA (cfDNA) into the blood circulation.3 Circulating tumor DNA (ctDNA) are fragments of DNA derived from tumor cells that circulate in the blood together with cfDNA. The ctDNA originates directly from a tumor or from circulating tumor cells (and carries information from the tumor cell genome), whereas cfDNA enters the bloodstream after apoptosis or necrosis and carries genome-wide DNA information. The amount of ctDNA in the blood has been shown to be elevated in patients with cancer.3 Different cancers release varying levels of ctDNA; the amount of ctDNA released depends on the number of tumor cells that are in senescence vs undergoing apoptosis.4
The possibility of incorporating this biomarker obtained from a “liquid biopsy” is currently being studied and will hopefully become a standard of care for breast cancer screening and monitoring. The liquid biopsy detects ctDNA that has been released into the bloodstream from tumor regions and helps identify intratumoral heterogeneity and clonal evolution.5 Additionally, sequencing tumor DNA has opened new possibilities for precision oncology.6 Detection of somatic gene mutations, amplifications, and gene fusions helps to deliver targeted therapies.6 Analysis of potential somatic mutations in ctDNA, in combination with cfDNA levels, can help capture clinically relevant information beyond single genetic alterations and tumor fraction, potentially improving the accuracy of early detection and screening for breast cancer.
Recent advances in ctDNA testing technology have made it more accurate and reliable. ctDNA testing has several benefits, including early detection of cancer (detecting ctDNA at low levels)7; monitoring of tumor dynamics, therapeutic response, and residual disease8; as well as analysis of the evolution of genetic or epigenetic alterations characterizing the tumor.9 Its noninvasiveness, rapidity, and low cost allow for longitudinal monitoring of cancer in real time, potentially capturing tumor heterogeneity.10,11
The liquid biopsy potentially can give more options for therapeutic monitoring for breast cancer and may mirror clinically relevant genetic alterations that occur in all tumor tissues. Liquid biopsy offers many advantages. It allows for the detection of minimal residual disease and micrometastatic disease that may be difficult to detect with a traditional tissue biopsy.12 Liquid biopsy detects ctDNA that has been released into the bloodstream from multiple tumor regions and allows the possibility of identifying intratumoral heterogeneity and clonal evolution.5 It can also detect small quantitative variations within the blood, enabling real-time surveillance.
The liquid biopsy can offer earlier and easier access to some tumor-based genetic information at any given timepoint and can replace a tumor tissue biopsy in some cases, helping to avoid delays and complications of a solid tumor invasive biopsy procedure. This is especially relevant in the metastatic setting, in which ctDNA might be the only available genetic material from tumors.13 Tissue biopsy can only provide a static and spatially limited view of the disease at the time of sampling; ctDNA analysis could potentially reflect the genetic alterations that occur in all metastatic breast cancer sites over time.14,15 Furthermore, machine learning of multi-gene signatures, obtained from ctDNA, can possibly identify complex biological features, including measures of tumor proliferation and estrogen receptor signaling, similar to direct tumor tissue DNA or RNA profiling.16
ctDNA testing is currently being studied to monitor patients who have been diagnosed with breast cancer. Small retrospective studies have shown that detection of ctDNA in plasma, after patients have completed therapy for early-stage breast cancer, is associated with a very high risk of relapse.17
Ongoing studies are examining the tailoring of adjuvant treatment based on ctDNA. If these trials are successful, certain aspects of adjuvant treatment could be lessened, or omitted, for patients who have undetectable ctDNA or intensified for patients who have detectable ctDNA after definitive treatments. This could personalize treatment specifically to the patient.
The detection and persistence of ctDNA in the middle of neoadjuvant systemic therapy may have the potential to negatively predict response to treatment and identify patients who will not achieve pathologic complete response. This may have the potential to aid in clinical decision-making for treatment escalation in these nonresponders.18
Despite these distinct characteristics, the low levels of ctDNA found in early-stage disease, along with the lack of ctDNA shedding from some tumors, can further complicate or impede detection of recurrence in early-stage breast cancer. Testing is further complicated by hematologic genetic alterations.5 The limitation of ctDNA approaches is that these techniques only detect known mutations in certain genes, so patients without these mutations could be overlooked, limiting the application of this technology.19
Overall, ctDNA testing represents a promising area of research for the diagnosis, treatment, and monitoring of breast cancer. While more research is needed to fully understand its potential, the advances in this technology are certainly exciting and could lead to significant improvements in patient outcomes. It is hopeful that in the near future, ctDNA testing from liquid biopsy could become a standard of care in breast cancer screening, ultimately helping clinicians to personalize treatment therapies and improve patient outcomes when treating patients with breast cancer.
1. Oeffinger KC, Fontham ETH, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599-1614.
2. Zubor P, Kubatka P, Kajo K, et al. Why the gold standard approach by mammography demands extension by multiomics? Application of liquid biopsy miRNA profiles to breast cancer disease management. Int J Mol Sci. 2019;20(12):E2878.
3. Thierry AR, El Messaoudi S, Gahan PB, Anker P, Stroun M. Origins, structures, and functions of circulating DNA in oncology. Cancer Metastasis Rev. 2016;35(3):347-376.
4. Rostami A, Lambie M, Yu CW, Stambolic V, Waldron JN, Bratman SV. Senescence, necrosis, and apoptosis govern circulating cell-free DNA release kinetics. Cell Rep. 2020;31(13):107830.
5. De Rubis G, Rajeev Krishnan S, Bebawy M. Liquid biopsies in cancer diagnosis, monitoring, and prognosis. Trends Pharmacol Sci. 2019;40(3):172-186.
6. Mateo J, Chakravarty D, Dienstmann R, et al. A framework to rank genomic alterations as targets for cancer precision medicine: the ESMO Scale for Clinical Actionability of molecular Targets (ESCAT). Ann Oncol. 2018;29:1895-1902.
7. Wang J, Han X, Sun Y. DNA methylation signatures in circulating cell-free DNA as biomarkers for the early detection of cancer. Sci China Life Sci. 2017;60(4):356-362.
8. Dawson S-J, Tsui DWY, Murtaza M, et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N Engl J Med. 2013;368(13):1199-1209.
9. Diaz Jr LA, Bardelli A. Liquid biopsies: genotyping circulating tumor DNA. J Clin Oncol. 2014;32(6):579-586.
10. Oxnard GR, Paweletz CP, Kuang Y, et al. Noninvasive detection of response and resistance in EGFR-mutant lung cancer using quantitative next-generation genotyping of cell-free plasma DNA. Clin Cancer Res. 2014;20(6):1698-1705.
11. Jamal-Hanjani M, Wilson GA, Horswell S, et al. Detection of ubiquitous and heterogeneous mutations in cell-free DNA from patients with early-stage non-small-cell lung cancer. Ann Oncol. 2016;27(5):862-867.
12. Fiala C, Diamandis EP. Utility of circulating tumor DNA in cancer diagnostics with
13. Xia Y, Fan C, Hoadley KA, Parker JS, Perou CM. Genetic determinants of the molecular portraits of epithelial cancers. Nat Commun. 2019;10(1):5666.
14. Wan JCM, Massie C, Garcia-Corbacho J, et al. Liquid biopsies come of age: towards implementation of circulating tumour DNA. Nat Rev Cancer. 2017;17(4):223-238.
15. Boldrin E, Nardo G, Zulato E, et al. Detection of loss of heterozygosity in cfDNA of advanced EGFR- or KRAS-mutated non-small-cell lung cancer patients. Int J Mol Sci. 2019;21(1):66.
16. Prat A, Brasó-Maristany F, Martínez-Sáez O, et al. Circulating tumor DNA reveals complex biological features with clinical relevance in metastatic breast cancer. Nat Commun. 2023;14(1):1157.
17. Coombes RC, Page K, Salari R, et al. Personalized detection of circulating tumor DNA antedates breast cancer metastatic recurrence. Clin Cancer Res. 2019;25(14):4255-4263.
18. Zhou Q, Gampenrieder SP, Frantal S, et al. Persistence of ctDNA in patients with breast cancer during neoadjuvant treatment is a significant predictor of poor tumor response. Clin Cancer Res. 2022;28(4):697-707.
19. Lin C, Liu X, Zheng B, Ke R, Tzeng C-M. Liquid biopsy, ctDNA diagnosis through NGS. Life (Basel). 2021;11(9):890.
1. Oeffinger KC, Fontham ETH, Etzioni R, et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA. 2015;314(15):1599-1614.
2. Zubor P, Kubatka P, Kajo K, et al. Why the gold standard approach by mammography demands extension by multiomics? Application of liquid biopsy miRNA profiles to breast cancer disease management. Int J Mol Sci. 2019;20(12):E2878.
3. Thierry AR, El Messaoudi S, Gahan PB, Anker P, Stroun M. Origins, structures, and functions of circulating DNA in oncology. Cancer Metastasis Rev. 2016;35(3):347-376.
4. Rostami A, Lambie M, Yu CW, Stambolic V, Waldron JN, Bratman SV. Senescence, necrosis, and apoptosis govern circulating cell-free DNA release kinetics. Cell Rep. 2020;31(13):107830.
5. De Rubis G, Rajeev Krishnan S, Bebawy M. Liquid biopsies in cancer diagnosis, monitoring, and prognosis. Trends Pharmacol Sci. 2019;40(3):172-186.
6. Mateo J, Chakravarty D, Dienstmann R, et al. A framework to rank genomic alterations as targets for cancer precision medicine: the ESMO Scale for Clinical Actionability of molecular Targets (ESCAT). Ann Oncol. 2018;29:1895-1902.
7. Wang J, Han X, Sun Y. DNA methylation signatures in circulating cell-free DNA as biomarkers for the early detection of cancer. Sci China Life Sci. 2017;60(4):356-362.
8. Dawson S-J, Tsui DWY, Murtaza M, et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N Engl J Med. 2013;368(13):1199-1209.
9. Diaz Jr LA, Bardelli A. Liquid biopsies: genotyping circulating tumor DNA. J Clin Oncol. 2014;32(6):579-586.
10. Oxnard GR, Paweletz CP, Kuang Y, et al. Noninvasive detection of response and resistance in EGFR-mutant lung cancer using quantitative next-generation genotyping of cell-free plasma DNA. Clin Cancer Res. 2014;20(6):1698-1705.
11. Jamal-Hanjani M, Wilson GA, Horswell S, et al. Detection of ubiquitous and heterogeneous mutations in cell-free DNA from patients with early-stage non-small-cell lung cancer. Ann Oncol. 2016;27(5):862-867.
12. Fiala C, Diamandis EP. Utility of circulating tumor DNA in cancer diagnostics with
13. Xia Y, Fan C, Hoadley KA, Parker JS, Perou CM. Genetic determinants of the molecular portraits of epithelial cancers. Nat Commun. 2019;10(1):5666.
14. Wan JCM, Massie C, Garcia-Corbacho J, et al. Liquid biopsies come of age: towards implementation of circulating tumour DNA. Nat Rev Cancer. 2017;17(4):223-238.
15. Boldrin E, Nardo G, Zulato E, et al. Detection of loss of heterozygosity in cfDNA of advanced EGFR- or KRAS-mutated non-small-cell lung cancer patients. Int J Mol Sci. 2019;21(1):66.
16. Prat A, Brasó-Maristany F, Martínez-Sáez O, et al. Circulating tumor DNA reveals complex biological features with clinical relevance in metastatic breast cancer. Nat Commun. 2023;14(1):1157.
17. Coombes RC, Page K, Salari R, et al. Personalized detection of circulating tumor DNA antedates breast cancer metastatic recurrence. Clin Cancer Res. 2019;25(14):4255-4263.
18. Zhou Q, Gampenrieder SP, Frantal S, et al. Persistence of ctDNA in patients with breast cancer during neoadjuvant treatment is a significant predictor of poor tumor response. Clin Cancer Res. 2022;28(4):697-707.
19. Lin C, Liu X, Zheng B, Ke R, Tzeng C-M. Liquid biopsy, ctDNA diagnosis through NGS. Life (Basel). 2021;11(9):890.
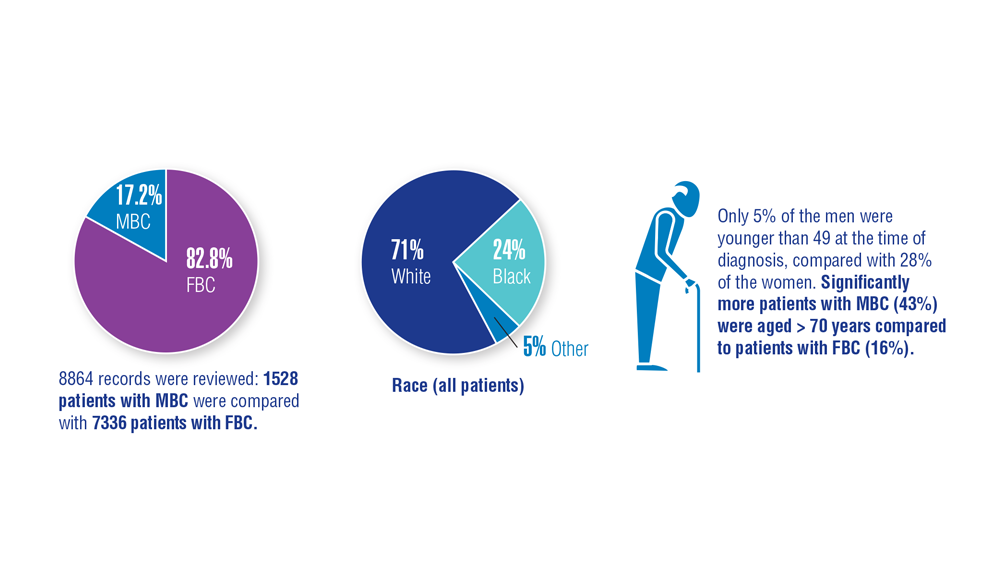
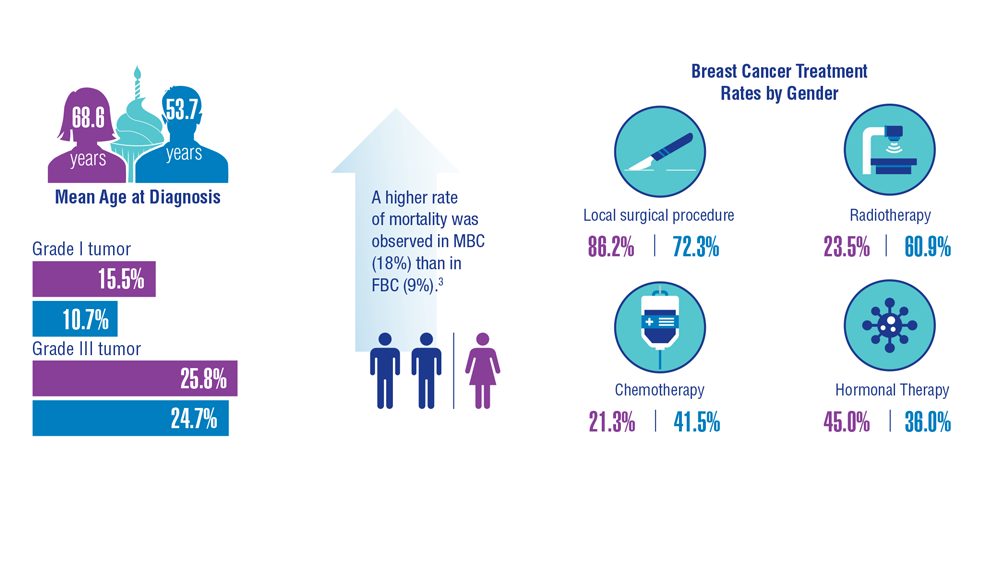
Gender Disparity in Breast Cancer Among US Veterans
1. Giordano SH, Cohen DS, Buzdar AU, Perkins G, Hortobagyi GN. Breast carcinoma in men: a population-based study. Cancer. 2004;101(1):51-57. doi:10.1002/cncr.20312
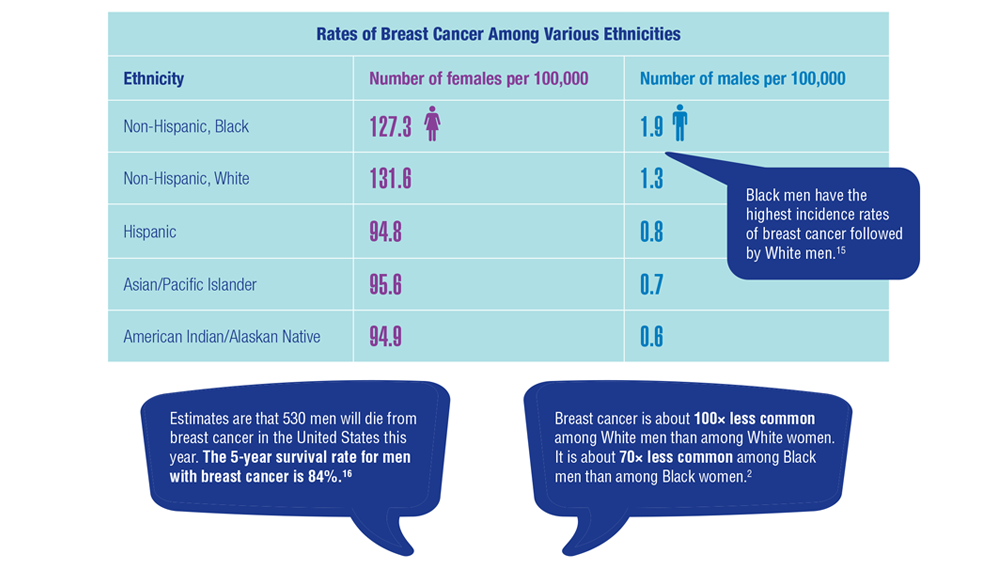
2. Key statistics for breast cancer in men. American Cancer Society. Updated January 12, 2022. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer-in-men/about/key-statistics.html
3. Aggarwal A, Adepoju B, Yacur M, Maron D, Sharma MH. Gender disparity in breast cancer: a veteran population-based comparison. Clin Breast Cancer. 2021;21(4):e471-e478. doi:10.1016/j.clbc.2021.01.013
4. Ravandi-Kashani F, Hayes TG. Male breast cancer: a review of the literature. Eur J Cancer. 1998;34(9):1341-1347. doi:10.1016/s0959-8049(98)00028-8
5. Giordano SH. A review of diagnosis and management of male breast cancer. Oncologist. 2005;10(7):471-479. doi:10.1634/theoncologist.10-7-471
6. Midding E, Halbach SM, Kowalski C, Weber R, Würstlein R, Ernstmann N. Men with a “woman's disease”: stigmatization of male breast cancer patients—a mixed methods analysis. Am J Mens Health. 2018;12(6):2194-2207. doi:10.1177/1557988318799025
7. Key statistics for breast cancer. American Cancer Society. Updated October 6, 2022. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html
8. Male breast cancer incidence and mortality, United States—2013-2017. Centers for Disease Control and Prevention. Updated October 1, 2020. Accessed December 14, 2022. https://www.cdc.gov/cancer/uscs/about/data-briefs/no19-male-breast-cancer-incidence-mortality-UnitedStates-2013-2017.htm
9. Anderson WF, Althuis MD, Brinton LA, Devesa SS. Is male breast cancer similar or different than female breast cancer? Breast Cancer Res Treat. 2004;83(1):77-86. doi:10.1023/B:BREA.0000010701.08825.2d 10. Pritzlaff M, Summerour P, McFarland R, et al. Male breast cancer in a multi-gene panel testing cohort: insights and unexpected results. Breast Cancer Res Treat. 2017;161(3):575-586. doi:10.1007/s10549-016-4085-4
11. Ottini L, Capalbo C, Rizzolo P, et al. HER2-positive male breast cancer: an update. Breast Cancer (Dove Med Press). 2010;2:45-58. doi:10.2147/BCTT.S6519
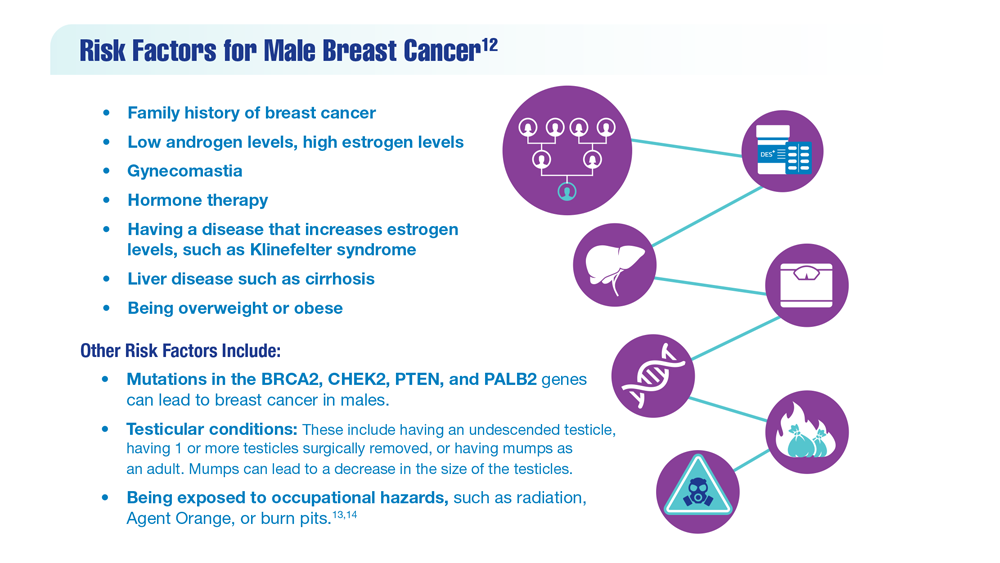
12. Risk factors for breast cancer in men. American Cancer Society. Updated April 27, 2018. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer-in-men/causes-risks-prevention/risk-factors.html
13. Palli D, Masala G, Mariani-Constantini R, et al. A gene–environment interaction between occupation and BRCA1/BRCA2 mutations in male breast cancer? Eur J Cancer. 2004;40(16):2472-2479. doi:10.1016/j.ejca.2004.07.012
14. Hansen J. Elevated risk for male breast cancer after occupational exposure to gasoline and vehicular combustion products. Am J Ind Med. 2000;37(4):349-352. doi:10.1002/(sici)1097-0274(200004)37:4<349::aid-ajim4>3.0.co;2-l
15. Sung H, DeSantis C, Jemal A. Subtype-specific breast cancer incidence rates in Black versus White men in the United States. JNCI Cancer Spectr. 2020;4(1):pkz091. doi:10.1093/jncics/pkz091
16. Breast cancer, male: statistics. Cancer.net. January 2022. Accessed December 14, 2022. https://www.cancer.net/cancer-types/breast-cancer-male/statistics
1. Giordano SH, Cohen DS, Buzdar AU, Perkins G, Hortobagyi GN. Breast carcinoma in men: a population-based study. Cancer. 2004;101(1):51-57. doi:10.1002/cncr.20312
2. Key statistics for breast cancer in men. American Cancer Society. Updated January 12, 2022. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer-in-men/about/key-statistics.html
3. Aggarwal A, Adepoju B, Yacur M, Maron D, Sharma MH. Gender disparity in breast cancer: a veteran population-based comparison. Clin Breast Cancer. 2021;21(4):e471-e478. doi:10.1016/j.clbc.2021.01.013
4. Ravandi-Kashani F, Hayes TG. Male breast cancer: a review of the literature. Eur J Cancer. 1998;34(9):1341-1347. doi:10.1016/s0959-8049(98)00028-8
5. Giordano SH. A review of diagnosis and management of male breast cancer. Oncologist. 2005;10(7):471-479. doi:10.1634/theoncologist.10-7-471
6. Midding E, Halbach SM, Kowalski C, Weber R, Würstlein R, Ernstmann N. Men with a “woman's disease”: stigmatization of male breast cancer patients—a mixed methods analysis. Am J Mens Health. 2018;12(6):2194-2207. doi:10.1177/1557988318799025
7. Key statistics for breast cancer. American Cancer Society. Updated October 6, 2022. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html
8. Male breast cancer incidence and mortality, United States—2013-2017. Centers for Disease Control and Prevention. Updated October 1, 2020. Accessed December 14, 2022. https://www.cdc.gov/cancer/uscs/about/data-briefs/no19-male-breast-cancer-incidence-mortality-UnitedStates-2013-2017.htm
9. Anderson WF, Althuis MD, Brinton LA, Devesa SS. Is male breast cancer similar or different than female breast cancer? Breast Cancer Res Treat. 2004;83(1):77-86. doi:10.1023/B:BREA.0000010701.08825.2d 10. Pritzlaff M, Summerour P, McFarland R, et al. Male breast cancer in a multi-gene panel testing cohort: insights and unexpected results. Breast Cancer Res Treat. 2017;161(3):575-586. doi:10.1007/s10549-016-4085-4
11. Ottini L, Capalbo C, Rizzolo P, et al. HER2-positive male breast cancer: an update. Breast Cancer (Dove Med Press). 2010;2:45-58. doi:10.2147/BCTT.S6519
12. Risk factors for breast cancer in men. American Cancer Society. Updated April 27, 2018. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer-in-men/causes-risks-prevention/risk-factors.html
13. Palli D, Masala G, Mariani-Constantini R, et al. A gene–environment interaction between occupation and BRCA1/BRCA2 mutations in male breast cancer? Eur J Cancer. 2004;40(16):2472-2479. doi:10.1016/j.ejca.2004.07.012
14. Hansen J. Elevated risk for male breast cancer after occupational exposure to gasoline and vehicular combustion products. Am J Ind Med. 2000;37(4):349-352. doi:10.1002/(sici)1097-0274(200004)37:4<349::aid-ajim4>3.0.co;2-l
15. Sung H, DeSantis C, Jemal A. Subtype-specific breast cancer incidence rates in Black versus White men in the United States. JNCI Cancer Spectr. 2020;4(1):pkz091. doi:10.1093/jncics/pkz091
16. Breast cancer, male: statistics. Cancer.net. January 2022. Accessed December 14, 2022. https://www.cancer.net/cancer-types/breast-cancer-male/statistics
1. Giordano SH, Cohen DS, Buzdar AU, Perkins G, Hortobagyi GN. Breast carcinoma in men: a population-based study. Cancer. 2004;101(1):51-57. doi:10.1002/cncr.20312
2. Key statistics for breast cancer in men. American Cancer Society. Updated January 12, 2022. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer-in-men/about/key-statistics.html
3. Aggarwal A, Adepoju B, Yacur M, Maron D, Sharma MH. Gender disparity in breast cancer: a veteran population-based comparison. Clin Breast Cancer. 2021;21(4):e471-e478. doi:10.1016/j.clbc.2021.01.013
4. Ravandi-Kashani F, Hayes TG. Male breast cancer: a review of the literature. Eur J Cancer. 1998;34(9):1341-1347. doi:10.1016/s0959-8049(98)00028-8
5. Giordano SH. A review of diagnosis and management of male breast cancer. Oncologist. 2005;10(7):471-479. doi:10.1634/theoncologist.10-7-471
6. Midding E, Halbach SM, Kowalski C, Weber R, Würstlein R, Ernstmann N. Men with a “woman's disease”: stigmatization of male breast cancer patients—a mixed methods analysis. Am J Mens Health. 2018;12(6):2194-2207. doi:10.1177/1557988318799025
7. Key statistics for breast cancer. American Cancer Society. Updated October 6, 2022. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html
8. Male breast cancer incidence and mortality, United States—2013-2017. Centers for Disease Control and Prevention. Updated October 1, 2020. Accessed December 14, 2022. https://www.cdc.gov/cancer/uscs/about/data-briefs/no19-male-breast-cancer-incidence-mortality-UnitedStates-2013-2017.htm
9. Anderson WF, Althuis MD, Brinton LA, Devesa SS. Is male breast cancer similar or different than female breast cancer? Breast Cancer Res Treat. 2004;83(1):77-86. doi:10.1023/B:BREA.0000010701.08825.2d 10. Pritzlaff M, Summerour P, McFarland R, et al. Male breast cancer in a multi-gene panel testing cohort: insights and unexpected results. Breast Cancer Res Treat. 2017;161(3):575-586. doi:10.1007/s10549-016-4085-4
11. Ottini L, Capalbo C, Rizzolo P, et al. HER2-positive male breast cancer: an update. Breast Cancer (Dove Med Press). 2010;2:45-58. doi:10.2147/BCTT.S6519
12. Risk factors for breast cancer in men. American Cancer Society. Updated April 27, 2018. Accessed December 14, 2022. https://www.cancer.org/cancer/breast-cancer-in-men/causes-risks-prevention/risk-factors.html
13. Palli D, Masala G, Mariani-Constantini R, et al. A gene–environment interaction between occupation and BRCA1/BRCA2 mutations in male breast cancer? Eur J Cancer. 2004;40(16):2472-2479. doi:10.1016/j.ejca.2004.07.012
14. Hansen J. Elevated risk for male breast cancer after occupational exposure to gasoline and vehicular combustion products. Am J Ind Med. 2000;37(4):349-352. doi:10.1002/(sici)1097-0274(200004)37:4<349::aid-ajim4>3.0.co;2-l
15. Sung H, DeSantis C, Jemal A. Subtype-specific breast cancer incidence rates in Black versus White men in the United States. JNCI Cancer Spectr. 2020;4(1):pkz091. doi:10.1093/jncics/pkz091
16. Breast cancer, male: statistics. Cancer.net. January 2022. Accessed December 14, 2022. https://www.cancer.net/cancer-types/breast-cancer-male/statistics
Cancer Data Trends 2023
Federal Practitioner and the Association of VA Hematology/Oncology (AVAHO) present the 2023 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- COVID-19 Outcomes in Veterans With Hematologic Cancers
- Promising New Approaches for Testicular and Prostate Cancer
- Screening Guideline Updates and New Treatments in Colon Cancer
- Exposure-Related Cancers: A Look at the PACT Act
- New Classifications and Emerging Treatments in Brain Cancer
- Gender Disparity in Breast Cancer Among US Veterans
- Lung Cancer Screening in Veterans
- Necessary Updates to Skin Cancer Risk Stratification
- Innovation in Cancer Treatment
Federal Practitioner and the Association of VA Hematology/Oncology (AVAHO) present the 2023 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- COVID-19 Outcomes in Veterans With Hematologic Cancers
- Promising New Approaches for Testicular and Prostate Cancer
- Screening Guideline Updates and New Treatments in Colon Cancer
- Exposure-Related Cancers: A Look at the PACT Act
- New Classifications and Emerging Treatments in Brain Cancer
- Gender Disparity in Breast Cancer Among US Veterans
- Lung Cancer Screening in Veterans
- Necessary Updates to Skin Cancer Risk Stratification
- Innovation in Cancer Treatment
Federal Practitioner and the Association of VA Hematology/Oncology (AVAHO) present the 2023 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- COVID-19 Outcomes in Veterans With Hematologic Cancers
- Promising New Approaches for Testicular and Prostate Cancer
- Screening Guideline Updates and New Treatments in Colon Cancer
- Exposure-Related Cancers: A Look at the PACT Act
- New Classifications and Emerging Treatments in Brain Cancer
- Gender Disparity in Breast Cancer Among US Veterans
- Lung Cancer Screening in Veterans
- Necessary Updates to Skin Cancer Risk Stratification
- Innovation in Cancer Treatment
PET-CT scans move more women with LABC up to stage IV
In women who have locally advanced breast cancer (LABC), staging defines the extent of the disease and guides therapy.
Researchers have found in the first large, randomized, controlled study on the subject that 18 F-labeled fluorodeoxyglucose positron emission tomography–computed tomography (PET-CT) finds more distant metastases and allows more accurate staging than usual staging, which is determined by a bone scan and computed tomography (CT) of the thorax/abdomen and pelvis.
Findings of the study, led by Ian S. Dayes, MD, MSc, with the department of oncology at McMaster University in Hamilton, Ont., were published online in the Journal of Clinical Oncology.
Scans indicate less aggressive treatment strategy
The authors of the new study wrote that women with LABC, who are at high risk of metastatic disease, have large tumors that “can involve the chest wall or skin, clinically fixed axillary lymph nodes, or infraclavicular, supraclavicular, or internal mammary lymphadenopathy.”
If staging does not detect metastases, treatment is centered on combined modality therapy with curative intent (neoadjuvant chemotherapy and surgery, followed by regional radiation). If metastases are found, the treatment goal changes to controlling the disease.
In this study, twice as many women saw their stage increase from stage IIB or III to stage IV when PET-CT was used instead of conventional staging, guiding their treatment toward less aggressive care to control, rather than attempt to cure, the disease.
The women included in this study had histological evidence of invasive ductal carcinoma of the breast and TNM stage III or IIb (T3N0, but not T2N1).
Methods and results
Between December 2016 and April 2022, consenting patients from six regional cancer centers in Ontario were randomly assigned to one of two groups: 184 patients were randomly assigned to whole-body PET-CT and 185 patients to conventional staging.
Overall, the authors wrote, 43 (23%) of PET-CT patients “were upstaged” to stage IV compared with 21 (11%) of the conventionally staged patients (relative risk, 2.4; 95% confidence interval [CI], 1.4-4.2, P = .002).
There were 33 patients in a subset with inflammatory breast cancer and, among them, 4 of 16 (25%) PET-CT patients were upstaged to stage IV compared with 4 of 17 (24%) conventionally staged patients.
In the patients who did not have inflammatory breast cancer, 39 of 168 (23%) PET-CT patients were upstaged compared with 17 (10%) of 168 in the conventionally staged group.
Journal of Clinical Oncology (JCO) Senior Deputy Editor Kathy D. Miller, MD, said that, “PET/CT staging identifies distant disease in more patients and changes goals of therapy. Further research is needed to determine the impact on patient outcome.”
Findings have already changed practice
Senior author, Mark Levine, MD, MSc, also with McMaster, said in an interview that the results of this study have already changed practice in Canada, and he expects the United States to follow suit.
Dr. Levine said the study is important “in terms of helping plan therapy and being very open and honest with patients as to their prognosis.”
The findings constitute level 1 evidence in favor of PET-CT. Already, in Canada, “because of the results of the study, people with stage III breast cancer can get a PET scan,” he said.
Dr. Levine said he expects this evidence also to clarify “wishy-washy” National Comprehensive Cancer Network guidelines on using PET scans for LABC in the United States when the guidelines are next updated.
“That will make it easier for payers in the United States,” he added.
Cost effectiveness, Dr. Levine said, is complicated, because on one hand PET scans are quite costly. But its use would lead to more women getting less aggressive and expensive therapy and surgery.
Dr. Levine noted that his team will be analyzing cost-effectiveness over the next year.
New questions with more in stage IV
In an editorial, Lajos Pusztai, MD, DPhil, scientific codirector of the breast center at Yale University in New Haven, Conn., noted that, “all good studies raise new questions” and this one is no exception.
He pointed out that the number of women with stage IV metastatic breast cancer (MBC) has been increasing over the past 2 decades because of more sensitive staging methods. At the same time the number of women with recurrent metastatic disease is decreasing, because adjuvant therapies have improved.
Findings highlight need for stage IV treatment studies
Dr. Pusztai noted that the patients who have de novo oligometastatic stage IV disease “are a unique subset among patients with MBC,” and the best treatment [for them] has not been established in randomized, controlled trials.
“Almost all randomized trials that targeted oligometastatic patients accrued mostly recurrent metastatic cancers; many included various cancer types, and none have tested the value of systemic multidrug regimens administered with curative intent,” he wrote.
If the health care systems adopt PET-CT for routine staging of locally advanced breast cancer, that will increase the diagnosis of de novo oligometastatic stage IV breast cancer, Dr. Pusztai said. That “underlines the importance of conducting studies for this unique subset of patients to establish level 1 evidence-based treatment strategies.”
Dr. Dayes has received honoraria from Verity Pharmaceuticals. One coauthor is employed by Point Biopharma. Other coauthors reported ties with AbbVie, Agendia, Genomic Health, InMode and Lutronic. Dr. Pusztai’s institution has received research funding from Merck, Genentech, Seagen, AstraZeneca, Bristol Myers Squibb, and Pfizer. He has received honoraria and travel expenses and has served in a consulting role for several pharmaceutical companies. Full disclosures are available on Open Payments.
In women who have locally advanced breast cancer (LABC), staging defines the extent of the disease and guides therapy.
Researchers have found in the first large, randomized, controlled study on the subject that 18 F-labeled fluorodeoxyglucose positron emission tomography–computed tomography (PET-CT) finds more distant metastases and allows more accurate staging than usual staging, which is determined by a bone scan and computed tomography (CT) of the thorax/abdomen and pelvis.
Findings of the study, led by Ian S. Dayes, MD, MSc, with the department of oncology at McMaster University in Hamilton, Ont., were published online in the Journal of Clinical Oncology.
Scans indicate less aggressive treatment strategy
The authors of the new study wrote that women with LABC, who are at high risk of metastatic disease, have large tumors that “can involve the chest wall or skin, clinically fixed axillary lymph nodes, or infraclavicular, supraclavicular, or internal mammary lymphadenopathy.”
If staging does not detect metastases, treatment is centered on combined modality therapy with curative intent (neoadjuvant chemotherapy and surgery, followed by regional radiation). If metastases are found, the treatment goal changes to controlling the disease.
In this study, twice as many women saw their stage increase from stage IIB or III to stage IV when PET-CT was used instead of conventional staging, guiding their treatment toward less aggressive care to control, rather than attempt to cure, the disease.
The women included in this study had histological evidence of invasive ductal carcinoma of the breast and TNM stage III or IIb (T3N0, but not T2N1).
Methods and results
Between December 2016 and April 2022, consenting patients from six regional cancer centers in Ontario were randomly assigned to one of two groups: 184 patients were randomly assigned to whole-body PET-CT and 185 patients to conventional staging.
Overall, the authors wrote, 43 (23%) of PET-CT patients “were upstaged” to stage IV compared with 21 (11%) of the conventionally staged patients (relative risk, 2.4; 95% confidence interval [CI], 1.4-4.2, P = .002).
There were 33 patients in a subset with inflammatory breast cancer and, among them, 4 of 16 (25%) PET-CT patients were upstaged to stage IV compared with 4 of 17 (24%) conventionally staged patients.
In the patients who did not have inflammatory breast cancer, 39 of 168 (23%) PET-CT patients were upstaged compared with 17 (10%) of 168 in the conventionally staged group.
Journal of Clinical Oncology (JCO) Senior Deputy Editor Kathy D. Miller, MD, said that, “PET/CT staging identifies distant disease in more patients and changes goals of therapy. Further research is needed to determine the impact on patient outcome.”
Findings have already changed practice
Senior author, Mark Levine, MD, MSc, also with McMaster, said in an interview that the results of this study have already changed practice in Canada, and he expects the United States to follow suit.
Dr. Levine said the study is important “in terms of helping plan therapy and being very open and honest with patients as to their prognosis.”
The findings constitute level 1 evidence in favor of PET-CT. Already, in Canada, “because of the results of the study, people with stage III breast cancer can get a PET scan,” he said.
Dr. Levine said he expects this evidence also to clarify “wishy-washy” National Comprehensive Cancer Network guidelines on using PET scans for LABC in the United States when the guidelines are next updated.
“That will make it easier for payers in the United States,” he added.
Cost effectiveness, Dr. Levine said, is complicated, because on one hand PET scans are quite costly. But its use would lead to more women getting less aggressive and expensive therapy and surgery.
Dr. Levine noted that his team will be analyzing cost-effectiveness over the next year.
New questions with more in stage IV
In an editorial, Lajos Pusztai, MD, DPhil, scientific codirector of the breast center at Yale University in New Haven, Conn., noted that, “all good studies raise new questions” and this one is no exception.
He pointed out that the number of women with stage IV metastatic breast cancer (MBC) has been increasing over the past 2 decades because of more sensitive staging methods. At the same time the number of women with recurrent metastatic disease is decreasing, because adjuvant therapies have improved.
Findings highlight need for stage IV treatment studies
Dr. Pusztai noted that the patients who have de novo oligometastatic stage IV disease “are a unique subset among patients with MBC,” and the best treatment [for them] has not been established in randomized, controlled trials.
“Almost all randomized trials that targeted oligometastatic patients accrued mostly recurrent metastatic cancers; many included various cancer types, and none have tested the value of systemic multidrug regimens administered with curative intent,” he wrote.
If the health care systems adopt PET-CT for routine staging of locally advanced breast cancer, that will increase the diagnosis of de novo oligometastatic stage IV breast cancer, Dr. Pusztai said. That “underlines the importance of conducting studies for this unique subset of patients to establish level 1 evidence-based treatment strategies.”
Dr. Dayes has received honoraria from Verity Pharmaceuticals. One coauthor is employed by Point Biopharma. Other coauthors reported ties with AbbVie, Agendia, Genomic Health, InMode and Lutronic. Dr. Pusztai’s institution has received research funding from Merck, Genentech, Seagen, AstraZeneca, Bristol Myers Squibb, and Pfizer. He has received honoraria and travel expenses and has served in a consulting role for several pharmaceutical companies. Full disclosures are available on Open Payments.
In women who have locally advanced breast cancer (LABC), staging defines the extent of the disease and guides therapy.
Researchers have found in the first large, randomized, controlled study on the subject that 18 F-labeled fluorodeoxyglucose positron emission tomography–computed tomography (PET-CT) finds more distant metastases and allows more accurate staging than usual staging, which is determined by a bone scan and computed tomography (CT) of the thorax/abdomen and pelvis.
Findings of the study, led by Ian S. Dayes, MD, MSc, with the department of oncology at McMaster University in Hamilton, Ont., were published online in the Journal of Clinical Oncology.
Scans indicate less aggressive treatment strategy
The authors of the new study wrote that women with LABC, who are at high risk of metastatic disease, have large tumors that “can involve the chest wall or skin, clinically fixed axillary lymph nodes, or infraclavicular, supraclavicular, or internal mammary lymphadenopathy.”
If staging does not detect metastases, treatment is centered on combined modality therapy with curative intent (neoadjuvant chemotherapy and surgery, followed by regional radiation). If metastases are found, the treatment goal changes to controlling the disease.
In this study, twice as many women saw their stage increase from stage IIB or III to stage IV when PET-CT was used instead of conventional staging, guiding their treatment toward less aggressive care to control, rather than attempt to cure, the disease.
The women included in this study had histological evidence of invasive ductal carcinoma of the breast and TNM stage III or IIb (T3N0, but not T2N1).
Methods and results
Between December 2016 and April 2022, consenting patients from six regional cancer centers in Ontario were randomly assigned to one of two groups: 184 patients were randomly assigned to whole-body PET-CT and 185 patients to conventional staging.
Overall, the authors wrote, 43 (23%) of PET-CT patients “were upstaged” to stage IV compared with 21 (11%) of the conventionally staged patients (relative risk, 2.4; 95% confidence interval [CI], 1.4-4.2, P = .002).
There were 33 patients in a subset with inflammatory breast cancer and, among them, 4 of 16 (25%) PET-CT patients were upstaged to stage IV compared with 4 of 17 (24%) conventionally staged patients.
In the patients who did not have inflammatory breast cancer, 39 of 168 (23%) PET-CT patients were upstaged compared with 17 (10%) of 168 in the conventionally staged group.
Journal of Clinical Oncology (JCO) Senior Deputy Editor Kathy D. Miller, MD, said that, “PET/CT staging identifies distant disease in more patients and changes goals of therapy. Further research is needed to determine the impact on patient outcome.”
Findings have already changed practice
Senior author, Mark Levine, MD, MSc, also with McMaster, said in an interview that the results of this study have already changed practice in Canada, and he expects the United States to follow suit.
Dr. Levine said the study is important “in terms of helping plan therapy and being very open and honest with patients as to their prognosis.”
The findings constitute level 1 evidence in favor of PET-CT. Already, in Canada, “because of the results of the study, people with stage III breast cancer can get a PET scan,” he said.
Dr. Levine said he expects this evidence also to clarify “wishy-washy” National Comprehensive Cancer Network guidelines on using PET scans for LABC in the United States when the guidelines are next updated.
“That will make it easier for payers in the United States,” he added.
Cost effectiveness, Dr. Levine said, is complicated, because on one hand PET scans are quite costly. But its use would lead to more women getting less aggressive and expensive therapy and surgery.
Dr. Levine noted that his team will be analyzing cost-effectiveness over the next year.
New questions with more in stage IV
In an editorial, Lajos Pusztai, MD, DPhil, scientific codirector of the breast center at Yale University in New Haven, Conn., noted that, “all good studies raise new questions” and this one is no exception.
He pointed out that the number of women with stage IV metastatic breast cancer (MBC) has been increasing over the past 2 decades because of more sensitive staging methods. At the same time the number of women with recurrent metastatic disease is decreasing, because adjuvant therapies have improved.
Findings highlight need for stage IV treatment studies
Dr. Pusztai noted that the patients who have de novo oligometastatic stage IV disease “are a unique subset among patients with MBC,” and the best treatment [for them] has not been established in randomized, controlled trials.
“Almost all randomized trials that targeted oligometastatic patients accrued mostly recurrent metastatic cancers; many included various cancer types, and none have tested the value of systemic multidrug regimens administered with curative intent,” he wrote.
If the health care systems adopt PET-CT for routine staging of locally advanced breast cancer, that will increase the diagnosis of de novo oligometastatic stage IV breast cancer, Dr. Pusztai said. That “underlines the importance of conducting studies for this unique subset of patients to establish level 1 evidence-based treatment strategies.”
Dr. Dayes has received honoraria from Verity Pharmaceuticals. One coauthor is employed by Point Biopharma. Other coauthors reported ties with AbbVie, Agendia, Genomic Health, InMode and Lutronic. Dr. Pusztai’s institution has received research funding from Merck, Genentech, Seagen, AstraZeneca, Bristol Myers Squibb, and Pfizer. He has received honoraria and travel expenses and has served in a consulting role for several pharmaceutical companies. Full disclosures are available on Open Payments.
FROM JOURNAL OF CLINICAL ONCOLOGY
Commentary: CDK4/6 Inhibitors, Breast Irradiation, and Aromatase Inhibitors in Breast Cancer Treatment, July 2023
After a median follow-up of 21.6 mo, the dalpiciclib group demonstrated a significantly longer median progression-free survival (PFS) compared with the placebo group (30.6 mo vs 18.2 mo; stratified hazard ratio [HR] 0.51; 95% CI 0.38-0.69; P < .0001). Overall, the dalpiciclib group demonstrated a manageable safety profile, although a higher percentage of grade 3/4 adverse events was noted with dalpiciclib than with placebo (90% vs 12%), as expected. Overall survival data for this CDK4/6 inhibitor are yet to come. These results suggest that dalpiciclib in combination with endocrine therapy is an alternative treatment for this group of patients, especially in countries where the traditionally approved CDK4/6 inhibitors (palbociclib, ribociclib, and abemaciclib) are not available.
The optimal sequencing of endocrine therapy (ET) after progression on CDK4/6 inhibitor–based therapy remains a challenge. In the phase 2 MAINTAIN trial, 119 patients (all of whom had HR+/HER2- metastatic breast cancer and who progressed on ET and CDK4/6 inhibitors) were randomly assigned to receive a different ET (fulvestrant or exemestane) from the previous ET they had received plus either the CDK4/6 inhibitor ribociclib or placebo. In the study by Kalinksky and colleagues, at a median follow-up of 18.2 mo, a significant improvement in PFS was observed in the switched ET-plus-ribociclib group compared with the switched ET-plus-placebo group (HR 0.57; P = .006). The phase 2 MAINTAIN trial is the first randomized trial to show the benefit of a CDK4/6 inhibitor after progression on another CDK4/6 inhibitor. It is important to note that the majority of patients in the MAINTAIN study previously received palbociclib in the first-line setting, which in recent studies has been demonstrated to be inferior to other CDK4/6 inhibitors. Therefore, it is important to confirm whether this will hold true upon progression from ribociclib or abemaciclib in the first-line setting. In addition, more data are needed to compare this approach with other ET treatment options, such as phosphoinositide 3-kinases inhibitors and oral selective estrogen receptor degraders.
There are several options for adjuvant radiation therapy for early-stage breast cancer. A meta-analysis of 14 randomized controlled trials and six comparative observational studies assessed the efficacy of whole breast irradiation (WBI) compared with partial breast irradiation (PBI) in 17,234 adults with early-stage breast cancer. Results of this meta-analysis showed that PBI was not significantly different from WBI, with similar rates of ipsilateral breast recurrence at 5 years (relative risk [RR] 1.34; 95% CI 0.83-2.18) and 10 years (RR 1.29; 95% CI 0.87-1.91), although patients undergoing PBI reported fewer acute adverse events compared with patients undergoing WBI (incidence rate ratio [IRR] 0.53; 95% CI 0.31-0.92) and acute grade ≥2 adverse events (IRR 0.21; 95% CI 0.07-0.62). These findings support using PBI as the adjuvant radiotherapy modality for select patients with favorable-risk early-stage breast cancer.
Another meta-analysis looked at assessing the survival benefit of adding CDK4/6 inhibitors to standard ET in older patients with advanced breast cancer. The study included 10 trials with 1985 older patients with advanced ER+ breast cancer who received ET with or without CDK4/6 inhibitors. The findings showed that adding CDK4/6 inhibitors to ET (letrozole or fulvestrant) significantly reduced the mortality risk by 21% (HR 0.79; 95% CI 0.69-0.91) and progression risk by 41% (HR 0.59; 95% CI 0.51-0.69) in older patients (age ≥ 65 years) with advanced breast cancer. Grade 3-4 neutropenia and diarrhea were similar in older patients. This study supports the use of CDK4/6 inhibitors as a reasonable treatment modality for older patients. More studies dedicated to the geriatric population are needed to help elaborate on the efficacy and tolerability of such agents in this population.
The phase 3 National Surgical Adjuvant Breast and Bowel Project B-42 (NSABP B-42) trial evaluated the role of extended letrozole therapy in postmenopausal breast cancer patients who were disease-free after 5 years of aromatase inhibitor–based therapy. The study included 3966 postmenopausal women with stage I-IIIA HR+ breast cancer who were randomly assigned to receive letrozole or placebo for 5 more years. After a median follow-up of 10.3 years, letrozole significantly improved disease-free survival (10-year absolute benefit 3.4%; HR 0.85; P = .01) compared with placebo, although there were no differences noted in overall survival between the groups (HR 0.97, P = .74). Furthermore, letrozole significantly reduced the breast cancer–free interval (HR 0.75, ,P = .003) and distant recurrence (HR 0.72, P = .01). There were no notable differences in toxicity, particularly rates of osteoporotic fractures and arterial thrombotic events, between the groups. Extended therapy with aromatase inhibitors beyond 5 years can be considered for select patients with early-stage breast cancer. Careful consideration of risks and benefits is needed to make these recommendations.
After a median follow-up of 21.6 mo, the dalpiciclib group demonstrated a significantly longer median progression-free survival (PFS) compared with the placebo group (30.6 mo vs 18.2 mo; stratified hazard ratio [HR] 0.51; 95% CI 0.38-0.69; P < .0001). Overall, the dalpiciclib group demonstrated a manageable safety profile, although a higher percentage of grade 3/4 adverse events was noted with dalpiciclib than with placebo (90% vs 12%), as expected. Overall survival data for this CDK4/6 inhibitor are yet to come. These results suggest that dalpiciclib in combination with endocrine therapy is an alternative treatment for this group of patients, especially in countries where the traditionally approved CDK4/6 inhibitors (palbociclib, ribociclib, and abemaciclib) are not available.
The optimal sequencing of endocrine therapy (ET) after progression on CDK4/6 inhibitor–based therapy remains a challenge. In the phase 2 MAINTAIN trial, 119 patients (all of whom had HR+/HER2- metastatic breast cancer and who progressed on ET and CDK4/6 inhibitors) were randomly assigned to receive a different ET (fulvestrant or exemestane) from the previous ET they had received plus either the CDK4/6 inhibitor ribociclib or placebo. In the study by Kalinksky and colleagues, at a median follow-up of 18.2 mo, a significant improvement in PFS was observed in the switched ET-plus-ribociclib group compared with the switched ET-plus-placebo group (HR 0.57; P = .006). The phase 2 MAINTAIN trial is the first randomized trial to show the benefit of a CDK4/6 inhibitor after progression on another CDK4/6 inhibitor. It is important to note that the majority of patients in the MAINTAIN study previously received palbociclib in the first-line setting, which in recent studies has been demonstrated to be inferior to other CDK4/6 inhibitors. Therefore, it is important to confirm whether this will hold true upon progression from ribociclib or abemaciclib in the first-line setting. In addition, more data are needed to compare this approach with other ET treatment options, such as phosphoinositide 3-kinases inhibitors and oral selective estrogen receptor degraders.
There are several options for adjuvant radiation therapy for early-stage breast cancer. A meta-analysis of 14 randomized controlled trials and six comparative observational studies assessed the efficacy of whole breast irradiation (WBI) compared with partial breast irradiation (PBI) in 17,234 adults with early-stage breast cancer. Results of this meta-analysis showed that PBI was not significantly different from WBI, with similar rates of ipsilateral breast recurrence at 5 years (relative risk [RR] 1.34; 95% CI 0.83-2.18) and 10 years (RR 1.29; 95% CI 0.87-1.91), although patients undergoing PBI reported fewer acute adverse events compared with patients undergoing WBI (incidence rate ratio [IRR] 0.53; 95% CI 0.31-0.92) and acute grade ≥2 adverse events (IRR 0.21; 95% CI 0.07-0.62). These findings support using PBI as the adjuvant radiotherapy modality for select patients with favorable-risk early-stage breast cancer.
Another meta-analysis looked at assessing the survival benefit of adding CDK4/6 inhibitors to standard ET in older patients with advanced breast cancer. The study included 10 trials with 1985 older patients with advanced ER+ breast cancer who received ET with or without CDK4/6 inhibitors. The findings showed that adding CDK4/6 inhibitors to ET (letrozole or fulvestrant) significantly reduced the mortality risk by 21% (HR 0.79; 95% CI 0.69-0.91) and progression risk by 41% (HR 0.59; 95% CI 0.51-0.69) in older patients (age ≥ 65 years) with advanced breast cancer. Grade 3-4 neutropenia and diarrhea were similar in older patients. This study supports the use of CDK4/6 inhibitors as a reasonable treatment modality for older patients. More studies dedicated to the geriatric population are needed to help elaborate on the efficacy and tolerability of such agents in this population.
The phase 3 National Surgical Adjuvant Breast and Bowel Project B-42 (NSABP B-42) trial evaluated the role of extended letrozole therapy in postmenopausal breast cancer patients who were disease-free after 5 years of aromatase inhibitor–based therapy. The study included 3966 postmenopausal women with stage I-IIIA HR+ breast cancer who were randomly assigned to receive letrozole or placebo for 5 more years. After a median follow-up of 10.3 years, letrozole significantly improved disease-free survival (10-year absolute benefit 3.4%; HR 0.85; P = .01) compared with placebo, although there were no differences noted in overall survival between the groups (HR 0.97, P = .74). Furthermore, letrozole significantly reduced the breast cancer–free interval (HR 0.75, ,P = .003) and distant recurrence (HR 0.72, P = .01). There were no notable differences in toxicity, particularly rates of osteoporotic fractures and arterial thrombotic events, between the groups. Extended therapy with aromatase inhibitors beyond 5 years can be considered for select patients with early-stage breast cancer. Careful consideration of risks and benefits is needed to make these recommendations.
After a median follow-up of 21.6 mo, the dalpiciclib group demonstrated a significantly longer median progression-free survival (PFS) compared with the placebo group (30.6 mo vs 18.2 mo; stratified hazard ratio [HR] 0.51; 95% CI 0.38-0.69; P < .0001). Overall, the dalpiciclib group demonstrated a manageable safety profile, although a higher percentage of grade 3/4 adverse events was noted with dalpiciclib than with placebo (90% vs 12%), as expected. Overall survival data for this CDK4/6 inhibitor are yet to come. These results suggest that dalpiciclib in combination with endocrine therapy is an alternative treatment for this group of patients, especially in countries where the traditionally approved CDK4/6 inhibitors (palbociclib, ribociclib, and abemaciclib) are not available.
The optimal sequencing of endocrine therapy (ET) after progression on CDK4/6 inhibitor–based therapy remains a challenge. In the phase 2 MAINTAIN trial, 119 patients (all of whom had HR+/HER2- metastatic breast cancer and who progressed on ET and CDK4/6 inhibitors) were randomly assigned to receive a different ET (fulvestrant or exemestane) from the previous ET they had received plus either the CDK4/6 inhibitor ribociclib or placebo. In the study by Kalinksky and colleagues, at a median follow-up of 18.2 mo, a significant improvement in PFS was observed in the switched ET-plus-ribociclib group compared with the switched ET-plus-placebo group (HR 0.57; P = .006). The phase 2 MAINTAIN trial is the first randomized trial to show the benefit of a CDK4/6 inhibitor after progression on another CDK4/6 inhibitor. It is important to note that the majority of patients in the MAINTAIN study previously received palbociclib in the first-line setting, which in recent studies has been demonstrated to be inferior to other CDK4/6 inhibitors. Therefore, it is important to confirm whether this will hold true upon progression from ribociclib or abemaciclib in the first-line setting. In addition, more data are needed to compare this approach with other ET treatment options, such as phosphoinositide 3-kinases inhibitors and oral selective estrogen receptor degraders.
There are several options for adjuvant radiation therapy for early-stage breast cancer. A meta-analysis of 14 randomized controlled trials and six comparative observational studies assessed the efficacy of whole breast irradiation (WBI) compared with partial breast irradiation (PBI) in 17,234 adults with early-stage breast cancer. Results of this meta-analysis showed that PBI was not significantly different from WBI, with similar rates of ipsilateral breast recurrence at 5 years (relative risk [RR] 1.34; 95% CI 0.83-2.18) and 10 years (RR 1.29; 95% CI 0.87-1.91), although patients undergoing PBI reported fewer acute adverse events compared with patients undergoing WBI (incidence rate ratio [IRR] 0.53; 95% CI 0.31-0.92) and acute grade ≥2 adverse events (IRR 0.21; 95% CI 0.07-0.62). These findings support using PBI as the adjuvant radiotherapy modality for select patients with favorable-risk early-stage breast cancer.
Another meta-analysis looked at assessing the survival benefit of adding CDK4/6 inhibitors to standard ET in older patients with advanced breast cancer. The study included 10 trials with 1985 older patients with advanced ER+ breast cancer who received ET with or without CDK4/6 inhibitors. The findings showed that adding CDK4/6 inhibitors to ET (letrozole or fulvestrant) significantly reduced the mortality risk by 21% (HR 0.79; 95% CI 0.69-0.91) and progression risk by 41% (HR 0.59; 95% CI 0.51-0.69) in older patients (age ≥ 65 years) with advanced breast cancer. Grade 3-4 neutropenia and diarrhea were similar in older patients. This study supports the use of CDK4/6 inhibitors as a reasonable treatment modality for older patients. More studies dedicated to the geriatric population are needed to help elaborate on the efficacy and tolerability of such agents in this population.
The phase 3 National Surgical Adjuvant Breast and Bowel Project B-42 (NSABP B-42) trial evaluated the role of extended letrozole therapy in postmenopausal breast cancer patients who were disease-free after 5 years of aromatase inhibitor–based therapy. The study included 3966 postmenopausal women with stage I-IIIA HR+ breast cancer who were randomly assigned to receive letrozole or placebo for 5 more years. After a median follow-up of 10.3 years, letrozole significantly improved disease-free survival (10-year absolute benefit 3.4%; HR 0.85; P = .01) compared with placebo, although there were no differences noted in overall survival between the groups (HR 0.97, P = .74). Furthermore, letrozole significantly reduced the breast cancer–free interval (HR 0.75, ,P = .003) and distant recurrence (HR 0.72, P = .01). There were no notable differences in toxicity, particularly rates of osteoporotic fractures and arterial thrombotic events, between the groups. Extended therapy with aromatase inhibitors beyond 5 years can be considered for select patients with early-stage breast cancer. Careful consideration of risks and benefits is needed to make these recommendations.
Commentary: Advances in HER2 advanced breast cancer, July 2023
The neoadjuvant setting provides a favorable environment to study de-escalation approaches as treatment response (via pathologic complete response [pCR] assessment) can be used as a surrogate marker for outcome. Studies have shown the effect of HER2-enriched subtype and high ERBB2 expression on pCR rates after receipt of a chemotherapy-free, dual HER2-targeted regimen.2 The prospective, multicenter, neoadjuvant phase 2 WSG-TP-II trial randomly assigned 207 patients with HR+/HER2+ early breast cancer to 12 weeks of endocrine therapy (ET)–trastuzumab-pertuzumab vs paclitaxel-trastuzumab-pertuzumab. The pCR rate was inferior in the ET arm compared with the paclitaxel arm (23.7% vs 56.4%; odds ratio 0.24; 95% CI 0.12-0.46; P < .001). In addition, an immunohistochemistry ERBB2 score of 3 or higher and ERBB2-enriched subtype were predictors of higher pCR rates in both arms (Gluz et al). This study not only supports a deescalated chemotherapy neoadjuvant strategy of paclitaxel + dual HER2 blockade but also suggests that a portion of patients may potentially be spared chemotherapy with very good results. The role of biomarkers is integral to patient selection for these approaches, and the evaluation of response in real-time will allow for the tailoring of therapy to achieve the best outcome.
Systemic staging for locally advanced breast cancer (LABC) is important for informing prognosis as well as aiding in development of an appropriate treatment plan for patients. The PETABC study included 369 patients with LABC (TNM stage III or IIB [T3N0]) with random assignment to 18F-labeled fluorodeoxyglucose PET-CT or conventional staging (bone scan, CT of chest/abdomen/pelvis), and was designed to assess the rate of upstaging with each imaging modality and effect on treatment (Dayes et al). In the PET-CT group, 23% (N = 43) of patients were upstaged to stage IV compared with 11% (N = 21) in the conventional-staging group (absolute difference 12.3%; 95% CI 3.9-19.9; P = .002). Fewer patients in the PET-CT group received combined modality treatment vs those patients in the conventional staging group (81% vs 89.2%; P = .03). These results support the consideration of PET-CT as a staging tool for LABC, and this is reflected in various clinical guidelines. Furthermore, the evolving role of other imaging techniques such as 18F-fluoroestradiol (18F-FES) PET-CT in detection of metastatic lesions related to estrogen receptor–positive breast cancer3 will continue to advance the field of imaging.
Additional References
- Rugo HS, Lerebours F, Ciruelos E, et al. Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): One cohort of a phase 2, multicentre, open-label, non-comparative study. Lancet Oncol. 2021;22:489-498. doi: 10.1016/S1470-2045(21)00034-6. Erratum in: Lancet Oncol. 2021;22(5):e184. doi: 10.1016/S1470-2045(21)00194-7
- Prat A, Pascual T, De Angelis C, et al. HER2-enriched subtype and ERBB2 expression in HER2-positive breast cancer treated with dual HER2 blockade. J Natl Cancer Inst. 2020;112:46-54. doi: 10.1093/jnci/djz042
- Ulaner GA, Jhaveri K, Chandarlapaty S, et al. Head-to-head evaluation of 18F-FES and 18F-FDG PET/CT in metastatic invasive lobular breast cancer. J Nucl Med. 2021;62:326-331. doi: 10.2967/jnumed.120.247882
The neoadjuvant setting provides a favorable environment to study de-escalation approaches as treatment response (via pathologic complete response [pCR] assessment) can be used as a surrogate marker for outcome. Studies have shown the effect of HER2-enriched subtype and high ERBB2 expression on pCR rates after receipt of a chemotherapy-free, dual HER2-targeted regimen.2 The prospective, multicenter, neoadjuvant phase 2 WSG-TP-II trial randomly assigned 207 patients with HR+/HER2+ early breast cancer to 12 weeks of endocrine therapy (ET)–trastuzumab-pertuzumab vs paclitaxel-trastuzumab-pertuzumab. The pCR rate was inferior in the ET arm compared with the paclitaxel arm (23.7% vs 56.4%; odds ratio 0.24; 95% CI 0.12-0.46; P < .001). In addition, an immunohistochemistry ERBB2 score of 3 or higher and ERBB2-enriched subtype were predictors of higher pCR rates in both arms (Gluz et al). This study not only supports a deescalated chemotherapy neoadjuvant strategy of paclitaxel + dual HER2 blockade but also suggests that a portion of patients may potentially be spared chemotherapy with very good results. The role of biomarkers is integral to patient selection for these approaches, and the evaluation of response in real-time will allow for the tailoring of therapy to achieve the best outcome.
Systemic staging for locally advanced breast cancer (LABC) is important for informing prognosis as well as aiding in development of an appropriate treatment plan for patients. The PETABC study included 369 patients with LABC (TNM stage III or IIB [T3N0]) with random assignment to 18F-labeled fluorodeoxyglucose PET-CT or conventional staging (bone scan, CT of chest/abdomen/pelvis), and was designed to assess the rate of upstaging with each imaging modality and effect on treatment (Dayes et al). In the PET-CT group, 23% (N = 43) of patients were upstaged to stage IV compared with 11% (N = 21) in the conventional-staging group (absolute difference 12.3%; 95% CI 3.9-19.9; P = .002). Fewer patients in the PET-CT group received combined modality treatment vs those patients in the conventional staging group (81% vs 89.2%; P = .03). These results support the consideration of PET-CT as a staging tool for LABC, and this is reflected in various clinical guidelines. Furthermore, the evolving role of other imaging techniques such as 18F-fluoroestradiol (18F-FES) PET-CT in detection of metastatic lesions related to estrogen receptor–positive breast cancer3 will continue to advance the field of imaging.
Additional References
- Rugo HS, Lerebours F, Ciruelos E, et al. Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): One cohort of a phase 2, multicentre, open-label, non-comparative study. Lancet Oncol. 2021;22:489-498. doi: 10.1016/S1470-2045(21)00034-6. Erratum in: Lancet Oncol. 2021;22(5):e184. doi: 10.1016/S1470-2045(21)00194-7
- Prat A, Pascual T, De Angelis C, et al. HER2-enriched subtype and ERBB2 expression in HER2-positive breast cancer treated with dual HER2 blockade. J Natl Cancer Inst. 2020;112:46-54. doi: 10.1093/jnci/djz042
- Ulaner GA, Jhaveri K, Chandarlapaty S, et al. Head-to-head evaluation of 18F-FES and 18F-FDG PET/CT in metastatic invasive lobular breast cancer. J Nucl Med. 2021;62:326-331. doi: 10.2967/jnumed.120.247882
The neoadjuvant setting provides a favorable environment to study de-escalation approaches as treatment response (via pathologic complete response [pCR] assessment) can be used as a surrogate marker for outcome. Studies have shown the effect of HER2-enriched subtype and high ERBB2 expression on pCR rates after receipt of a chemotherapy-free, dual HER2-targeted regimen.2 The prospective, multicenter, neoadjuvant phase 2 WSG-TP-II trial randomly assigned 207 patients with HR+/HER2+ early breast cancer to 12 weeks of endocrine therapy (ET)–trastuzumab-pertuzumab vs paclitaxel-trastuzumab-pertuzumab. The pCR rate was inferior in the ET arm compared with the paclitaxel arm (23.7% vs 56.4%; odds ratio 0.24; 95% CI 0.12-0.46; P < .001). In addition, an immunohistochemistry ERBB2 score of 3 or higher and ERBB2-enriched subtype were predictors of higher pCR rates in both arms (Gluz et al). This study not only supports a deescalated chemotherapy neoadjuvant strategy of paclitaxel + dual HER2 blockade but also suggests that a portion of patients may potentially be spared chemotherapy with very good results. The role of biomarkers is integral to patient selection for these approaches, and the evaluation of response in real-time will allow for the tailoring of therapy to achieve the best outcome.
Systemic staging for locally advanced breast cancer (LABC) is important for informing prognosis as well as aiding in development of an appropriate treatment plan for patients. The PETABC study included 369 patients with LABC (TNM stage III or IIB [T3N0]) with random assignment to 18F-labeled fluorodeoxyglucose PET-CT or conventional staging (bone scan, CT of chest/abdomen/pelvis), and was designed to assess the rate of upstaging with each imaging modality and effect on treatment (Dayes et al). In the PET-CT group, 23% (N = 43) of patients were upstaged to stage IV compared with 11% (N = 21) in the conventional-staging group (absolute difference 12.3%; 95% CI 3.9-19.9; P = .002). Fewer patients in the PET-CT group received combined modality treatment vs those patients in the conventional staging group (81% vs 89.2%; P = .03). These results support the consideration of PET-CT as a staging tool for LABC, and this is reflected in various clinical guidelines. Furthermore, the evolving role of other imaging techniques such as 18F-fluoroestradiol (18F-FES) PET-CT in detection of metastatic lesions related to estrogen receptor–positive breast cancer3 will continue to advance the field of imaging.
Additional References
- Rugo HS, Lerebours F, Ciruelos E, et al. Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): One cohort of a phase 2, multicentre, open-label, non-comparative study. Lancet Oncol. 2021;22:489-498. doi: 10.1016/S1470-2045(21)00034-6. Erratum in: Lancet Oncol. 2021;22(5):e184. doi: 10.1016/S1470-2045(21)00194-7
- Prat A, Pascual T, De Angelis C, et al. HER2-enriched subtype and ERBB2 expression in HER2-positive breast cancer treated with dual HER2 blockade. J Natl Cancer Inst. 2020;112:46-54. doi: 10.1093/jnci/djz042
- Ulaner GA, Jhaveri K, Chandarlapaty S, et al. Head-to-head evaluation of 18F-FES and 18F-FDG PET/CT in metastatic invasive lobular breast cancer. J Nucl Med. 2021;62:326-331. doi: 10.2967/jnumed.120.247882
Multiprong strategy makes clinical trials less White
CHICAGO – Clinical trials are so White. Only a small percentage of eligible patients participate in clinical trials in the first place, and very few come from racial and ethnic minority groups.
For example, according to the Food and Drug Administration, in trials that resulted in drug approvals from 2017 to 2020, only 2%-5% of participants were Black patients.
When clinical trials lack diverse patient populations, those who are left out have fewer opportunities to get new therapies. Moreover, the scope of the research is limited by smaller phenotypic and genotypic samples, and the trial results are applicable only to more homogeneous patient groups.
There has been a push to include more underrepresented patients in clinical trials. One group reported its success in doing so here at the annual meeting of the American Society of Clinical Oncology.
a period that included a pandemic-induced hiatus in clinical trials in general.
Alliance member Electra D. Paskett, PhD, from the College of Public Health at the Ohio State University in Columbus, presented accrual data from 117 trials led by the Alliance from 2014 to 2022.
During this period, accrual of racial and ethnic minority patients increased from 13.6% to 25.3% for cancer treatment trials and from 13% to 21.5% for cancer control trials.
Overall, the recruitment program resulted in an absolute increase from 13.5 % to 23.6% of underrepresented populations, which translated into a relative 74.8% improvement.
“We’re focusing now on monitoring accrual of women, rural populations, younger AYAs [adolescents and young adults] and older patients, and we’ll see what strategies we need to implement,” Dr. Packett told this news organization.
The Alliance has implemented a real-time accrual dashboard on its website that allows individual sites to review accrual by trial and overall for all of the identified underrepresented populations, she noted.
Program to increase underrepresented patient accrual
The impetus for the program to increase enrollment of underrepresented patients came from the goal set by Monica M. Bertagnolli, MD, group chair of the Alliance from 2011 to 2022 and currently the director of the U.S. National Cancer Institute.
“Our leader, Dr. Bertagnolli, set out a group-wide goal for accrual of underrepresented minorities to our trials of 20%, and that gave us permission to implement a whole host of new strategies,” Dr. Paskett said in an interview.
“These strategies follow the Accrual of Clinical Trials framework, which essentially says that the interaction between the patient and the provider for going on a clinical trial is not just an interaction between the patient and provider but recognizes, for example, that the provider has coworkers and they have norms and beliefs and attitudes, and the patient comes from a family with their own values. And then there are system-level barriers, and there are community barriers that all relate to this interaction about going on a trial,” Dr. Packett said.
What works?
The study was presented as a poster at the meeting. During the poster discussion session, comoderator Victoria S. Blinder, MD, from Memorial Sloan Kettering Cancer Center in New York, asked Dr. Paskett, “If you had a certain amount of money and you really wanted to use that resource to focus on one area, where would you put that resource?”
“I’m going to violate the rules of your question,” Dr. Paskett replied.
“You cannot change this problem by focusing on one thing, and that’s what we showed in our Alliance poster, and what I’ve said is based on over 30 years of work in this area,” she said.
She cited what she considered as the two most important components for improving accrual of underrepresented populations: a commitment by leadership to a recruitment goal, and the development of protocols with specific accrual goals for minority populations.
Still, those are only two components of a comprehensive program that includes the aforementioned accrual goal set by Dr. Bertagnolli, as well as the following:
- Funding of minority junior investigators and research that focuses on issues of concern to underrepresented populations.
- Establishment of work groups that focus on specific populations with the Alliance health disparities committee.
- Translation of informational materials for patients.
- Opening studies at National Cancer Institute Community. Oncology Research Program–designated minority underserved sites.
- Real-time monitoring of accrual demographics by the Alliance and at the trial site.
- Closing protocol enrollment to majority populations.
- Increasing the study sample sizes to enroll additional minority participants and to allow for subgroup analyses.
The study was funded by the National Institutes of Health. Dr. Packett and Dr. Blinder reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
CHICAGO – Clinical trials are so White. Only a small percentage of eligible patients participate in clinical trials in the first place, and very few come from racial and ethnic minority groups.
For example, according to the Food and Drug Administration, in trials that resulted in drug approvals from 2017 to 2020, only 2%-5% of participants were Black patients.
When clinical trials lack diverse patient populations, those who are left out have fewer opportunities to get new therapies. Moreover, the scope of the research is limited by smaller phenotypic and genotypic samples, and the trial results are applicable only to more homogeneous patient groups.
There has been a push to include more underrepresented patients in clinical trials. One group reported its success in doing so here at the annual meeting of the American Society of Clinical Oncology.
a period that included a pandemic-induced hiatus in clinical trials in general.
Alliance member Electra D. Paskett, PhD, from the College of Public Health at the Ohio State University in Columbus, presented accrual data from 117 trials led by the Alliance from 2014 to 2022.
During this period, accrual of racial and ethnic minority patients increased from 13.6% to 25.3% for cancer treatment trials and from 13% to 21.5% for cancer control trials.
Overall, the recruitment program resulted in an absolute increase from 13.5 % to 23.6% of underrepresented populations, which translated into a relative 74.8% improvement.
“We’re focusing now on monitoring accrual of women, rural populations, younger AYAs [adolescents and young adults] and older patients, and we’ll see what strategies we need to implement,” Dr. Packett told this news organization.
The Alliance has implemented a real-time accrual dashboard on its website that allows individual sites to review accrual by trial and overall for all of the identified underrepresented populations, she noted.
Program to increase underrepresented patient accrual
The impetus for the program to increase enrollment of underrepresented patients came from the goal set by Monica M. Bertagnolli, MD, group chair of the Alliance from 2011 to 2022 and currently the director of the U.S. National Cancer Institute.
“Our leader, Dr. Bertagnolli, set out a group-wide goal for accrual of underrepresented minorities to our trials of 20%, and that gave us permission to implement a whole host of new strategies,” Dr. Paskett said in an interview.
“These strategies follow the Accrual of Clinical Trials framework, which essentially says that the interaction between the patient and the provider for going on a clinical trial is not just an interaction between the patient and provider but recognizes, for example, that the provider has coworkers and they have norms and beliefs and attitudes, and the patient comes from a family with their own values. And then there are system-level barriers, and there are community barriers that all relate to this interaction about going on a trial,” Dr. Packett said.
What works?
The study was presented as a poster at the meeting. During the poster discussion session, comoderator Victoria S. Blinder, MD, from Memorial Sloan Kettering Cancer Center in New York, asked Dr. Paskett, “If you had a certain amount of money and you really wanted to use that resource to focus on one area, where would you put that resource?”
“I’m going to violate the rules of your question,” Dr. Paskett replied.
“You cannot change this problem by focusing on one thing, and that’s what we showed in our Alliance poster, and what I’ve said is based on over 30 years of work in this area,” she said.
She cited what she considered as the two most important components for improving accrual of underrepresented populations: a commitment by leadership to a recruitment goal, and the development of protocols with specific accrual goals for minority populations.
Still, those are only two components of a comprehensive program that includes the aforementioned accrual goal set by Dr. Bertagnolli, as well as the following:
- Funding of minority junior investigators and research that focuses on issues of concern to underrepresented populations.
- Establishment of work groups that focus on specific populations with the Alliance health disparities committee.
- Translation of informational materials for patients.
- Opening studies at National Cancer Institute Community. Oncology Research Program–designated minority underserved sites.
- Real-time monitoring of accrual demographics by the Alliance and at the trial site.
- Closing protocol enrollment to majority populations.
- Increasing the study sample sizes to enroll additional minority participants and to allow for subgroup analyses.
The study was funded by the National Institutes of Health. Dr. Packett and Dr. Blinder reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
CHICAGO – Clinical trials are so White. Only a small percentage of eligible patients participate in clinical trials in the first place, and very few come from racial and ethnic minority groups.
For example, according to the Food and Drug Administration, in trials that resulted in drug approvals from 2017 to 2020, only 2%-5% of participants were Black patients.
When clinical trials lack diverse patient populations, those who are left out have fewer opportunities to get new therapies. Moreover, the scope of the research is limited by smaller phenotypic and genotypic samples, and the trial results are applicable only to more homogeneous patient groups.
There has been a push to include more underrepresented patients in clinical trials. One group reported its success in doing so here at the annual meeting of the American Society of Clinical Oncology.
a period that included a pandemic-induced hiatus in clinical trials in general.
Alliance member Electra D. Paskett, PhD, from the College of Public Health at the Ohio State University in Columbus, presented accrual data from 117 trials led by the Alliance from 2014 to 2022.
During this period, accrual of racial and ethnic minority patients increased from 13.6% to 25.3% for cancer treatment trials and from 13% to 21.5% for cancer control trials.
Overall, the recruitment program resulted in an absolute increase from 13.5 % to 23.6% of underrepresented populations, which translated into a relative 74.8% improvement.
“We’re focusing now on monitoring accrual of women, rural populations, younger AYAs [adolescents and young adults] and older patients, and we’ll see what strategies we need to implement,” Dr. Packett told this news organization.
The Alliance has implemented a real-time accrual dashboard on its website that allows individual sites to review accrual by trial and overall for all of the identified underrepresented populations, she noted.
Program to increase underrepresented patient accrual
The impetus for the program to increase enrollment of underrepresented patients came from the goal set by Monica M. Bertagnolli, MD, group chair of the Alliance from 2011 to 2022 and currently the director of the U.S. National Cancer Institute.
“Our leader, Dr. Bertagnolli, set out a group-wide goal for accrual of underrepresented minorities to our trials of 20%, and that gave us permission to implement a whole host of new strategies,” Dr. Paskett said in an interview.
“These strategies follow the Accrual of Clinical Trials framework, which essentially says that the interaction between the patient and the provider for going on a clinical trial is not just an interaction between the patient and provider but recognizes, for example, that the provider has coworkers and they have norms and beliefs and attitudes, and the patient comes from a family with their own values. And then there are system-level barriers, and there are community barriers that all relate to this interaction about going on a trial,” Dr. Packett said.
What works?
The study was presented as a poster at the meeting. During the poster discussion session, comoderator Victoria S. Blinder, MD, from Memorial Sloan Kettering Cancer Center in New York, asked Dr. Paskett, “If you had a certain amount of money and you really wanted to use that resource to focus on one area, where would you put that resource?”
“I’m going to violate the rules of your question,” Dr. Paskett replied.
“You cannot change this problem by focusing on one thing, and that’s what we showed in our Alliance poster, and what I’ve said is based on over 30 years of work in this area,” she said.
She cited what she considered as the two most important components for improving accrual of underrepresented populations: a commitment by leadership to a recruitment goal, and the development of protocols with specific accrual goals for minority populations.
Still, those are only two components of a comprehensive program that includes the aforementioned accrual goal set by Dr. Bertagnolli, as well as the following:
- Funding of minority junior investigators and research that focuses on issues of concern to underrepresented populations.
- Establishment of work groups that focus on specific populations with the Alliance health disparities committee.
- Translation of informational materials for patients.
- Opening studies at National Cancer Institute Community. Oncology Research Program–designated minority underserved sites.
- Real-time monitoring of accrual demographics by the Alliance and at the trial site.
- Closing protocol enrollment to majority populations.
- Increasing the study sample sizes to enroll additional minority participants and to allow for subgroup analyses.
The study was funded by the National Institutes of Health. Dr. Packett and Dr. Blinder reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
AT ASCO 2023
CBSM phone app eases anxiety, depression in cancer patients
CHICAGO – One-third of patients with cancer also experience anxiety or depression, and an estimated 70% of the 18 million patients with cancer and cancer survivors in the US experience emotional symptoms, including fear of recurrence.
Despite many having these symptoms, few patients with cancer have access to psycho-oncologic support.
A digital cognitive-behavioral stress management (CBSM) application may help to ease some of the burden, reported Allison Ramiller, MPH, of Blue Note Therapeutics in San Francisco, which developed the app version of the program.
In addition, patients assigned to the CBSM app were twice as likely as control persons to report that their symptoms were “much” or “very much” improved after using the app for 12 weeks, Ms. Ramiller reported at an oral abstract session at the annual meeting of the American Society of Clinical Oncology (ASCO).
However, the investigators did not report baseline characteristics of patients in each of the study arms, which might have helped to clarify the depth of the effects they saw.
The CBSM program was developed by Michael H. Antoni, PhD, and colleagues in the University of Miami Health System. It is based on cognitive-behavioral therapy but also includes stress management and relaxation techniques to help patients cope with cancer-specific stress.
“”It has been clinically validated and shown to benefit patients with cancer,” Ms. Ramiller said. “However, access is a problem,” she said.
“There aren’t enough qualified, trained providers for the need, and patients with cancer encounter barriers to in-person participation, including things like transportation or financial barriers. So to overcome this, we developed a digitized version of CBSM,” she explained.
Impressive and elegant
“Everything about [the study] I thought was very impressive, very elegant, very nicely done,” said invited discussant Raymond U. Osarogiagbon, MBBS, FACP, chief scientist at Baptist Memorial Health Care Corp in Memphis, Tenn.
“They showed efficacy, they showed safety – very nice – user friendliness – very good. Certainly they look like they’re trying to address a highly important, unmet need in a very elegant way. Certainly, they pointed out it needs longer follow-up to see sustainability. We need to see will this work in other settings. Will this be cost-effective? You’ve gotta believe it probably will be,” he said.
CBSM has previously been shown to help patients with cancer reduce stress, improve general and cancer-specific quality of life at various stages of treatment, reduce symptom burden, and improve coping skills, Ms. Ramiller said.
To see whether these benefits could be conveyed digitally rather than in face-to-face encounters, Ms. Ramiller and colleagues worked with Dr. Antoni to develop the CBSM app.
Patients using the app received therapeutic content over 10 sessions with audio, video, and interactive tools that mimicked the sessions they would have received during in-person interventions.
They then compared the app against the control educational app in the randomized, decentralized RESTORE study.
High-quality control
Ms. Ramiller said that the control app set “a high bar.”
“The control also offered 10 interactive self-guided sessions. Both treatment apps were professionally designed and visually similar in styling, and they were presented as digital therapeutic-specific for cancer patients. And they were also in a match condition, meaning they received the same attention from study staff and cadence of reminders, but importantly, only the intervention app was based on CBSM,” she explained.
A total of 449 patients with cancers of stage I–III who were undergoing active systemic treatment or were planning to undergo such treatment within 6 months were randomly assigned to the CBSM app or the control app.
The CBSM app was superior to the control app for the primary outcome of anxiety reduction over baseline, as measured at 4, 8 and 12 weeks by the Patient-Reported Outcomes Measurement Information System Anxiety Scale (PROMIS-A) (beta = -.03; P = .019).
CBSM was also significantly better than the control app for the secondary endpoints of reducing symptoms of depression, as measured by the PROMIS-D scale (beta = -.02, P = .042), and also at increasing the percentage of patients who reported improvement in anxiety and depression symptoms on the Patient Global Impression of Change instrument (P < .001)
An extension study of the durability of the effects at 3 and 6 months is underway.
The investigators noted that the incremental cost of management of anxiety or depression is greater than $17,000 per patient per year.
“One of the big promises of a digital therapeutic like this is that it could potentially reduce costs,” Ms. Ramiller told the audience, but she acknowledged, “More work is really needed, however, to directly test the potential savings.”
The RESTORE study is funded by Blue Note Therapeutics. Dr. Osarogiagbon owns stock in Gilead, Lilly, and Pfizer, has received honoraria from Biodesix and Medscape, and has a consulting or advisory role for the American Cancer Society AstraZeneca, Genentech/Roche, LUNGevity, National Cancer Institute, and Triptych Health Partners.
A version of this article originally appeared on Medscape.com.
CHICAGO – One-third of patients with cancer also experience anxiety or depression, and an estimated 70% of the 18 million patients with cancer and cancer survivors in the US experience emotional symptoms, including fear of recurrence.
Despite many having these symptoms, few patients with cancer have access to psycho-oncologic support.
A digital cognitive-behavioral stress management (CBSM) application may help to ease some of the burden, reported Allison Ramiller, MPH, of Blue Note Therapeutics in San Francisco, which developed the app version of the program.
In addition, patients assigned to the CBSM app were twice as likely as control persons to report that their symptoms were “much” or “very much” improved after using the app for 12 weeks, Ms. Ramiller reported at an oral abstract session at the annual meeting of the American Society of Clinical Oncology (ASCO).
However, the investigators did not report baseline characteristics of patients in each of the study arms, which might have helped to clarify the depth of the effects they saw.
The CBSM program was developed by Michael H. Antoni, PhD, and colleagues in the University of Miami Health System. It is based on cognitive-behavioral therapy but also includes stress management and relaxation techniques to help patients cope with cancer-specific stress.
“”It has been clinically validated and shown to benefit patients with cancer,” Ms. Ramiller said. “However, access is a problem,” she said.
“There aren’t enough qualified, trained providers for the need, and patients with cancer encounter barriers to in-person participation, including things like transportation or financial barriers. So to overcome this, we developed a digitized version of CBSM,” she explained.
Impressive and elegant
“Everything about [the study] I thought was very impressive, very elegant, very nicely done,” said invited discussant Raymond U. Osarogiagbon, MBBS, FACP, chief scientist at Baptist Memorial Health Care Corp in Memphis, Tenn.
“They showed efficacy, they showed safety – very nice – user friendliness – very good. Certainly they look like they’re trying to address a highly important, unmet need in a very elegant way. Certainly, they pointed out it needs longer follow-up to see sustainability. We need to see will this work in other settings. Will this be cost-effective? You’ve gotta believe it probably will be,” he said.
CBSM has previously been shown to help patients with cancer reduce stress, improve general and cancer-specific quality of life at various stages of treatment, reduce symptom burden, and improve coping skills, Ms. Ramiller said.
To see whether these benefits could be conveyed digitally rather than in face-to-face encounters, Ms. Ramiller and colleagues worked with Dr. Antoni to develop the CBSM app.
Patients using the app received therapeutic content over 10 sessions with audio, video, and interactive tools that mimicked the sessions they would have received during in-person interventions.
They then compared the app against the control educational app in the randomized, decentralized RESTORE study.
High-quality control
Ms. Ramiller said that the control app set “a high bar.”
“The control also offered 10 interactive self-guided sessions. Both treatment apps were professionally designed and visually similar in styling, and they were presented as digital therapeutic-specific for cancer patients. And they were also in a match condition, meaning they received the same attention from study staff and cadence of reminders, but importantly, only the intervention app was based on CBSM,” she explained.
A total of 449 patients with cancers of stage I–III who were undergoing active systemic treatment or were planning to undergo such treatment within 6 months were randomly assigned to the CBSM app or the control app.
The CBSM app was superior to the control app for the primary outcome of anxiety reduction over baseline, as measured at 4, 8 and 12 weeks by the Patient-Reported Outcomes Measurement Information System Anxiety Scale (PROMIS-A) (beta = -.03; P = .019).
CBSM was also significantly better than the control app for the secondary endpoints of reducing symptoms of depression, as measured by the PROMIS-D scale (beta = -.02, P = .042), and also at increasing the percentage of patients who reported improvement in anxiety and depression symptoms on the Patient Global Impression of Change instrument (P < .001)
An extension study of the durability of the effects at 3 and 6 months is underway.
The investigators noted that the incremental cost of management of anxiety or depression is greater than $17,000 per patient per year.
“One of the big promises of a digital therapeutic like this is that it could potentially reduce costs,” Ms. Ramiller told the audience, but she acknowledged, “More work is really needed, however, to directly test the potential savings.”
The RESTORE study is funded by Blue Note Therapeutics. Dr. Osarogiagbon owns stock in Gilead, Lilly, and Pfizer, has received honoraria from Biodesix and Medscape, and has a consulting or advisory role for the American Cancer Society AstraZeneca, Genentech/Roche, LUNGevity, National Cancer Institute, and Triptych Health Partners.
A version of this article originally appeared on Medscape.com.
CHICAGO – One-third of patients with cancer also experience anxiety or depression, and an estimated 70% of the 18 million patients with cancer and cancer survivors in the US experience emotional symptoms, including fear of recurrence.
Despite many having these symptoms, few patients with cancer have access to psycho-oncologic support.
A digital cognitive-behavioral stress management (CBSM) application may help to ease some of the burden, reported Allison Ramiller, MPH, of Blue Note Therapeutics in San Francisco, which developed the app version of the program.
In addition, patients assigned to the CBSM app were twice as likely as control persons to report that their symptoms were “much” or “very much” improved after using the app for 12 weeks, Ms. Ramiller reported at an oral abstract session at the annual meeting of the American Society of Clinical Oncology (ASCO).
However, the investigators did not report baseline characteristics of patients in each of the study arms, which might have helped to clarify the depth of the effects they saw.
The CBSM program was developed by Michael H. Antoni, PhD, and colleagues in the University of Miami Health System. It is based on cognitive-behavioral therapy but also includes stress management and relaxation techniques to help patients cope with cancer-specific stress.
“”It has been clinically validated and shown to benefit patients with cancer,” Ms. Ramiller said. “However, access is a problem,” she said.
“There aren’t enough qualified, trained providers for the need, and patients with cancer encounter barriers to in-person participation, including things like transportation or financial barriers. So to overcome this, we developed a digitized version of CBSM,” she explained.
Impressive and elegant
“Everything about [the study] I thought was very impressive, very elegant, very nicely done,” said invited discussant Raymond U. Osarogiagbon, MBBS, FACP, chief scientist at Baptist Memorial Health Care Corp in Memphis, Tenn.
“They showed efficacy, they showed safety – very nice – user friendliness – very good. Certainly they look like they’re trying to address a highly important, unmet need in a very elegant way. Certainly, they pointed out it needs longer follow-up to see sustainability. We need to see will this work in other settings. Will this be cost-effective? You’ve gotta believe it probably will be,” he said.
CBSM has previously been shown to help patients with cancer reduce stress, improve general and cancer-specific quality of life at various stages of treatment, reduce symptom burden, and improve coping skills, Ms. Ramiller said.
To see whether these benefits could be conveyed digitally rather than in face-to-face encounters, Ms. Ramiller and colleagues worked with Dr. Antoni to develop the CBSM app.
Patients using the app received therapeutic content over 10 sessions with audio, video, and interactive tools that mimicked the sessions they would have received during in-person interventions.
They then compared the app against the control educational app in the randomized, decentralized RESTORE study.
High-quality control
Ms. Ramiller said that the control app set “a high bar.”
“The control also offered 10 interactive self-guided sessions. Both treatment apps were professionally designed and visually similar in styling, and they were presented as digital therapeutic-specific for cancer patients. And they were also in a match condition, meaning they received the same attention from study staff and cadence of reminders, but importantly, only the intervention app was based on CBSM,” she explained.
A total of 449 patients with cancers of stage I–III who were undergoing active systemic treatment or were planning to undergo such treatment within 6 months were randomly assigned to the CBSM app or the control app.
The CBSM app was superior to the control app for the primary outcome of anxiety reduction over baseline, as measured at 4, 8 and 12 weeks by the Patient-Reported Outcomes Measurement Information System Anxiety Scale (PROMIS-A) (beta = -.03; P = .019).
CBSM was also significantly better than the control app for the secondary endpoints of reducing symptoms of depression, as measured by the PROMIS-D scale (beta = -.02, P = .042), and also at increasing the percentage of patients who reported improvement in anxiety and depression symptoms on the Patient Global Impression of Change instrument (P < .001)
An extension study of the durability of the effects at 3 and 6 months is underway.
The investigators noted that the incremental cost of management of anxiety or depression is greater than $17,000 per patient per year.
“One of the big promises of a digital therapeutic like this is that it could potentially reduce costs,” Ms. Ramiller told the audience, but she acknowledged, “More work is really needed, however, to directly test the potential savings.”
The RESTORE study is funded by Blue Note Therapeutics. Dr. Osarogiagbon owns stock in Gilead, Lilly, and Pfizer, has received honoraria from Biodesix and Medscape, and has a consulting or advisory role for the American Cancer Society AstraZeneca, Genentech/Roche, LUNGevity, National Cancer Institute, and Triptych Health Partners.
A version of this article originally appeared on Medscape.com.
AT ASCO 2023
Breast cancer family history linked to better BC survival
TOPLINE:
METHODOLOGY:
- Investigators reviewed 28,649 Swedish women diagnosed with breast cancer from 1991 to 2019.
- Overall, 5,081 patients (17.7%) had at least one female first-degree relative previously diagnosed with breast cancer.
TAKEAWAYS:
- After adjusting for demographics, tumor characteristics, and treatments, a family history of breast cancer was associated with a lower risk of breast cancer–specific death in the full cohort (hazard ratio, 0.78) and in ER-negative women (HR, 0.57) within 5 years of diagnosis, after which point the association was no longer significant.
- The lower risk of death among women with a family history could mean that these women are more motivated and likely to get screened, potentially catching tumors earlier, and may be more likely to adhere to treatment recommendations.
- However, having a family history of early-onset breast cancer (before the age of 40) was associated with a higher risk of breast cancer–specific death (HR, 1.41).
IN PRACTICE:
Although the findings are reassuring for many women with breast cancer, “genetic testing of newly diagnosed patients with early-onset family history may provide useful information to aid treatment and future research,” the researchers concluded.
STUDY DETAILS:
The study was led by Yuqi Zhang, PhD, of the Karolinska Institutet, Stockholm, and published in JAMA Network Open.
LIMITATIONS:
- The main analysis did not include tumor characteristics only available within the last 20 years, including ERBB2 status.
- Relatively wide confidence intervals make the association between a family history of early-onset breast cancer and higher risk of breast cancer death somewhat uncertain.
DISCLOSURES:
- The work was funded by the Swedish Cancer Society and others.
- The investigators report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Investigators reviewed 28,649 Swedish women diagnosed with breast cancer from 1991 to 2019.
- Overall, 5,081 patients (17.7%) had at least one female first-degree relative previously diagnosed with breast cancer.
TAKEAWAYS:
- After adjusting for demographics, tumor characteristics, and treatments, a family history of breast cancer was associated with a lower risk of breast cancer–specific death in the full cohort (hazard ratio, 0.78) and in ER-negative women (HR, 0.57) within 5 years of diagnosis, after which point the association was no longer significant.
- The lower risk of death among women with a family history could mean that these women are more motivated and likely to get screened, potentially catching tumors earlier, and may be more likely to adhere to treatment recommendations.
- However, having a family history of early-onset breast cancer (before the age of 40) was associated with a higher risk of breast cancer–specific death (HR, 1.41).
IN PRACTICE:
Although the findings are reassuring for many women with breast cancer, “genetic testing of newly diagnosed patients with early-onset family history may provide useful information to aid treatment and future research,” the researchers concluded.
STUDY DETAILS:
The study was led by Yuqi Zhang, PhD, of the Karolinska Institutet, Stockholm, and published in JAMA Network Open.
LIMITATIONS:
- The main analysis did not include tumor characteristics only available within the last 20 years, including ERBB2 status.
- Relatively wide confidence intervals make the association between a family history of early-onset breast cancer and higher risk of breast cancer death somewhat uncertain.
DISCLOSURES:
- The work was funded by the Swedish Cancer Society and others.
- The investigators report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Investigators reviewed 28,649 Swedish women diagnosed with breast cancer from 1991 to 2019.
- Overall, 5,081 patients (17.7%) had at least one female first-degree relative previously diagnosed with breast cancer.
TAKEAWAYS:
- After adjusting for demographics, tumor characteristics, and treatments, a family history of breast cancer was associated with a lower risk of breast cancer–specific death in the full cohort (hazard ratio, 0.78) and in ER-negative women (HR, 0.57) within 5 years of diagnosis, after which point the association was no longer significant.
- The lower risk of death among women with a family history could mean that these women are more motivated and likely to get screened, potentially catching tumors earlier, and may be more likely to adhere to treatment recommendations.
- However, having a family history of early-onset breast cancer (before the age of 40) was associated with a higher risk of breast cancer–specific death (HR, 1.41).
IN PRACTICE:
Although the findings are reassuring for many women with breast cancer, “genetic testing of newly diagnosed patients with early-onset family history may provide useful information to aid treatment and future research,” the researchers concluded.
STUDY DETAILS:
The study was led by Yuqi Zhang, PhD, of the Karolinska Institutet, Stockholm, and published in JAMA Network Open.
LIMITATIONS:
- The main analysis did not include tumor characteristics only available within the last 20 years, including ERBB2 status.
- Relatively wide confidence intervals make the association between a family history of early-onset breast cancer and higher risk of breast cancer death somewhat uncertain.
DISCLOSURES:
- The work was funded by the Swedish Cancer Society and others.
- The investigators report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
T-DXd active in many solid tumors; ‘shift in thinking’
CHICAGO – Trastuzumab deruxtecan (T-DXd) (Enhertu) already has proven efficacy against HER2-expressing metastatic breast, gastroesophageal, and lung cancers.
including malignancies of the cervix, endometrium, ovaries, bladder, and other sites.
The findings come from the ongoing DESTINY-PanTumor02 trial. Among 267 patients with solid tumors at various organ sites, the investigator-assessed objective response rate among all patients was 37.1%, and ranged from as high as 57.5% for patients with endometrial cancers to as low as 4% for patients with pancreatic cancer, reported Funda Meric-Bernstam, MD, from the University of Texas MD Anderson Cancer Center, Houston.
For patients with tumors that had HER2 immunohistochemistry (IHC) scores of 3+, the highest level of HER2 expression, the overall response rate was 61.3%..
The responses were also durable, with a median duration of 11.8 months among all patients and 22.1 months among patients with IHC 3+ scores.
“Our data to date showed that T-DXd had clinically meaningful activity across a variety of tumor types,” she said in a briefing held prior to her presentation of the data at the annual meeting of the American Society of Clinical Oncology.
“HER2 expression has been around a long time. We think about this all the time in breast cancer and drugs are approved there, but HER2 is expressed in other tumors as well, and that really represents an unmet need, because we have limited options in this situation” commented ASCO expert Bradley Alexander McGregor, MD, from the Dana-Farber Cancer Institute, Boston, an invited discussant at the briefing.
“Aside from pancreatic cancer we saw really, really encouraging results with no new safety signals, so while early I think this really exciting and represents a shift in how we think about cancer care,” he added.
After the presentation, invited discussant Kohei Shitara, MD, of National Cancer Center Hospital East, Kashiwa, Japan, said that he agrees with authors that T-DXd is a potential new treatment option for patients with HER2-expressing solid tumors, and that the evidence suggests the potential for further tumor-agnostic development of the agent.
He cautioned, however, that there is a lack of concordance between local and central assessment of HER2 IHC, and that quality assurance will be required to ensure that the HER2 status of solid tumors is accurately characterized.
At a press briefing, Dr. Meric-Bernstam was asked how she envisioned using T-DXd in therapy for various HER2-expressing tumors.
“I think the activity we’ve seen is quite compelling, and one hopes that eventually this will be a drug that’s accessible for patients that are HER2-expressing across tumor types. Clearly, the activity is very compelling in some of the diseases to think about doing studies for earlier lines,” she said.
“The data indicate that there is tumor-agnostic activity across the board,” she said, but noted that tumors with epithelial components such as ovarian and breast cancers appear to have the highest responses to T-DXd therapy.
Briefing moderator Julie R. Gralow, MD, chief medical officer and executive vice president of ASCO, asked Dr. McGregor whether, in the light of this new data, oncologists should test more patients for HER2 expression.
“We have some cancers where we know HER2 expression is there. I think the good thing about HER2 testing is that it’s an IHC test, so this is something that can be easily done in local pathology [labs],” he said. As more evidence mounts of potential benefit of T-DXd in HER2 expressing tumors, clinicians will need to consider more routine HER2 testing.
A rendezvous with DESTINY
The DESTINY-PanTumor02 trial is a phase 2, open-label, multicenter study looking at T-DXd in patients with advanced solid tumors who are not eligible for therapy with curative intent.
All patients had disease progression after at least two prior lines of therapy, and had tumors with HER2 expression of IHC 3+ or 2+ either by local or central testing. Patients were allowed to have previously received HER2-targeting therapy. Patients also had to have good performance status (Eastern Cooperative Oncology Group/World Health Organization performance status 0 or 1).
The investigators planned to enroll 40 patients in each cohort, including patients with cervical, endometrial, ovarian, biliary tract, pancreatic, or bladder cancers, as well those with other tumors expressing HER2 who were not included in the other cohorts.
Under the protocol, cohorts in which none of the first 15 patients had objective responses would be closed, as happened with the pancreatic cancer cohort.
At a median follow-up of 9.7 months, an objective response was seen in 99 patients out of the 267 in the entire study population (ORR, 37.1%). This ORR consisted of 15 complete responses and 84 partial responses. An additional 123 patients had stable disease.
An analysis of ORR by HER2 expression showed that IHC 3+ expressing tumors had rates ranging from 84.6% in endometrial cancers, 75% in cervical cancer, 63.6% in ovarian cancers, and 56.3% in bladder cancers, down to zero in IHC 3+ expressing pancreatic cancer.
The T-DXd safety profile was consistent with that seen in other clinical trials, with most common adverse events being nausea, fatigue, neutropenia, anemia, diarrhea, and thrombocytopenia. There were 20 cases of interstitial lung disease, one of which was fatal.
The trial is ongoing, and investigators plan to report overall survival and progression-free survival results with additional follow-up.
DESTINY-PanTumor02 is funded by Daiichi Sankyo. Dr. Meric-Bernstam disclosed a consulting/advisory role with multiple pharmaceutical companies, research funding to her institution from Daiichi Sankyo and others, and travel expenses from ESMO and EORTC. Dr. McGregor disclosed a consulting/advisory role and institutional research funding with multiple companies, not including the study’s funder. Dr. Gralow disclosed a consulting or advisory role with Genentech and Roche.
A version of this article first appeared on Medscape.com.
CHICAGO – Trastuzumab deruxtecan (T-DXd) (Enhertu) already has proven efficacy against HER2-expressing metastatic breast, gastroesophageal, and lung cancers.
including malignancies of the cervix, endometrium, ovaries, bladder, and other sites.
The findings come from the ongoing DESTINY-PanTumor02 trial. Among 267 patients with solid tumors at various organ sites, the investigator-assessed objective response rate among all patients was 37.1%, and ranged from as high as 57.5% for patients with endometrial cancers to as low as 4% for patients with pancreatic cancer, reported Funda Meric-Bernstam, MD, from the University of Texas MD Anderson Cancer Center, Houston.
For patients with tumors that had HER2 immunohistochemistry (IHC) scores of 3+, the highest level of HER2 expression, the overall response rate was 61.3%..
The responses were also durable, with a median duration of 11.8 months among all patients and 22.1 months among patients with IHC 3+ scores.
“Our data to date showed that T-DXd had clinically meaningful activity across a variety of tumor types,” she said in a briefing held prior to her presentation of the data at the annual meeting of the American Society of Clinical Oncology.
“HER2 expression has been around a long time. We think about this all the time in breast cancer and drugs are approved there, but HER2 is expressed in other tumors as well, and that really represents an unmet need, because we have limited options in this situation” commented ASCO expert Bradley Alexander McGregor, MD, from the Dana-Farber Cancer Institute, Boston, an invited discussant at the briefing.
“Aside from pancreatic cancer we saw really, really encouraging results with no new safety signals, so while early I think this really exciting and represents a shift in how we think about cancer care,” he added.
After the presentation, invited discussant Kohei Shitara, MD, of National Cancer Center Hospital East, Kashiwa, Japan, said that he agrees with authors that T-DXd is a potential new treatment option for patients with HER2-expressing solid tumors, and that the evidence suggests the potential for further tumor-agnostic development of the agent.
He cautioned, however, that there is a lack of concordance between local and central assessment of HER2 IHC, and that quality assurance will be required to ensure that the HER2 status of solid tumors is accurately characterized.
At a press briefing, Dr. Meric-Bernstam was asked how she envisioned using T-DXd in therapy for various HER2-expressing tumors.
“I think the activity we’ve seen is quite compelling, and one hopes that eventually this will be a drug that’s accessible for patients that are HER2-expressing across tumor types. Clearly, the activity is very compelling in some of the diseases to think about doing studies for earlier lines,” she said.
“The data indicate that there is tumor-agnostic activity across the board,” she said, but noted that tumors with epithelial components such as ovarian and breast cancers appear to have the highest responses to T-DXd therapy.
Briefing moderator Julie R. Gralow, MD, chief medical officer and executive vice president of ASCO, asked Dr. McGregor whether, in the light of this new data, oncologists should test more patients for HER2 expression.
“We have some cancers where we know HER2 expression is there. I think the good thing about HER2 testing is that it’s an IHC test, so this is something that can be easily done in local pathology [labs],” he said. As more evidence mounts of potential benefit of T-DXd in HER2 expressing tumors, clinicians will need to consider more routine HER2 testing.
A rendezvous with DESTINY
The DESTINY-PanTumor02 trial is a phase 2, open-label, multicenter study looking at T-DXd in patients with advanced solid tumors who are not eligible for therapy with curative intent.
All patients had disease progression after at least two prior lines of therapy, and had tumors with HER2 expression of IHC 3+ or 2+ either by local or central testing. Patients were allowed to have previously received HER2-targeting therapy. Patients also had to have good performance status (Eastern Cooperative Oncology Group/World Health Organization performance status 0 or 1).
The investigators planned to enroll 40 patients in each cohort, including patients with cervical, endometrial, ovarian, biliary tract, pancreatic, or bladder cancers, as well those with other tumors expressing HER2 who were not included in the other cohorts.
Under the protocol, cohorts in which none of the first 15 patients had objective responses would be closed, as happened with the pancreatic cancer cohort.
At a median follow-up of 9.7 months, an objective response was seen in 99 patients out of the 267 in the entire study population (ORR, 37.1%). This ORR consisted of 15 complete responses and 84 partial responses. An additional 123 patients had stable disease.
An analysis of ORR by HER2 expression showed that IHC 3+ expressing tumors had rates ranging from 84.6% in endometrial cancers, 75% in cervical cancer, 63.6% in ovarian cancers, and 56.3% in bladder cancers, down to zero in IHC 3+ expressing pancreatic cancer.
The T-DXd safety profile was consistent with that seen in other clinical trials, with most common adverse events being nausea, fatigue, neutropenia, anemia, diarrhea, and thrombocytopenia. There were 20 cases of interstitial lung disease, one of which was fatal.
The trial is ongoing, and investigators plan to report overall survival and progression-free survival results with additional follow-up.
DESTINY-PanTumor02 is funded by Daiichi Sankyo. Dr. Meric-Bernstam disclosed a consulting/advisory role with multiple pharmaceutical companies, research funding to her institution from Daiichi Sankyo and others, and travel expenses from ESMO and EORTC. Dr. McGregor disclosed a consulting/advisory role and institutional research funding with multiple companies, not including the study’s funder. Dr. Gralow disclosed a consulting or advisory role with Genentech and Roche.
A version of this article first appeared on Medscape.com.
CHICAGO – Trastuzumab deruxtecan (T-DXd) (Enhertu) already has proven efficacy against HER2-expressing metastatic breast, gastroesophageal, and lung cancers.
including malignancies of the cervix, endometrium, ovaries, bladder, and other sites.
The findings come from the ongoing DESTINY-PanTumor02 trial. Among 267 patients with solid tumors at various organ sites, the investigator-assessed objective response rate among all patients was 37.1%, and ranged from as high as 57.5% for patients with endometrial cancers to as low as 4% for patients with pancreatic cancer, reported Funda Meric-Bernstam, MD, from the University of Texas MD Anderson Cancer Center, Houston.
For patients with tumors that had HER2 immunohistochemistry (IHC) scores of 3+, the highest level of HER2 expression, the overall response rate was 61.3%..
The responses were also durable, with a median duration of 11.8 months among all patients and 22.1 months among patients with IHC 3+ scores.
“Our data to date showed that T-DXd had clinically meaningful activity across a variety of tumor types,” she said in a briefing held prior to her presentation of the data at the annual meeting of the American Society of Clinical Oncology.
“HER2 expression has been around a long time. We think about this all the time in breast cancer and drugs are approved there, but HER2 is expressed in other tumors as well, and that really represents an unmet need, because we have limited options in this situation” commented ASCO expert Bradley Alexander McGregor, MD, from the Dana-Farber Cancer Institute, Boston, an invited discussant at the briefing.
“Aside from pancreatic cancer we saw really, really encouraging results with no new safety signals, so while early I think this really exciting and represents a shift in how we think about cancer care,” he added.
After the presentation, invited discussant Kohei Shitara, MD, of National Cancer Center Hospital East, Kashiwa, Japan, said that he agrees with authors that T-DXd is a potential new treatment option for patients with HER2-expressing solid tumors, and that the evidence suggests the potential for further tumor-agnostic development of the agent.
He cautioned, however, that there is a lack of concordance between local and central assessment of HER2 IHC, and that quality assurance will be required to ensure that the HER2 status of solid tumors is accurately characterized.
At a press briefing, Dr. Meric-Bernstam was asked how she envisioned using T-DXd in therapy for various HER2-expressing tumors.
“I think the activity we’ve seen is quite compelling, and one hopes that eventually this will be a drug that’s accessible for patients that are HER2-expressing across tumor types. Clearly, the activity is very compelling in some of the diseases to think about doing studies for earlier lines,” she said.
“The data indicate that there is tumor-agnostic activity across the board,” she said, but noted that tumors with epithelial components such as ovarian and breast cancers appear to have the highest responses to T-DXd therapy.
Briefing moderator Julie R. Gralow, MD, chief medical officer and executive vice president of ASCO, asked Dr. McGregor whether, in the light of this new data, oncologists should test more patients for HER2 expression.
“We have some cancers where we know HER2 expression is there. I think the good thing about HER2 testing is that it’s an IHC test, so this is something that can be easily done in local pathology [labs],” he said. As more evidence mounts of potential benefit of T-DXd in HER2 expressing tumors, clinicians will need to consider more routine HER2 testing.
A rendezvous with DESTINY
The DESTINY-PanTumor02 trial is a phase 2, open-label, multicenter study looking at T-DXd in patients with advanced solid tumors who are not eligible for therapy with curative intent.
All patients had disease progression after at least two prior lines of therapy, and had tumors with HER2 expression of IHC 3+ or 2+ either by local or central testing. Patients were allowed to have previously received HER2-targeting therapy. Patients also had to have good performance status (Eastern Cooperative Oncology Group/World Health Organization performance status 0 or 1).
The investigators planned to enroll 40 patients in each cohort, including patients with cervical, endometrial, ovarian, biliary tract, pancreatic, or bladder cancers, as well those with other tumors expressing HER2 who were not included in the other cohorts.
Under the protocol, cohorts in which none of the first 15 patients had objective responses would be closed, as happened with the pancreatic cancer cohort.
At a median follow-up of 9.7 months, an objective response was seen in 99 patients out of the 267 in the entire study population (ORR, 37.1%). This ORR consisted of 15 complete responses and 84 partial responses. An additional 123 patients had stable disease.
An analysis of ORR by HER2 expression showed that IHC 3+ expressing tumors had rates ranging from 84.6% in endometrial cancers, 75% in cervical cancer, 63.6% in ovarian cancers, and 56.3% in bladder cancers, down to zero in IHC 3+ expressing pancreatic cancer.
The T-DXd safety profile was consistent with that seen in other clinical trials, with most common adverse events being nausea, fatigue, neutropenia, anemia, diarrhea, and thrombocytopenia. There were 20 cases of interstitial lung disease, one of which was fatal.
The trial is ongoing, and investigators plan to report overall survival and progression-free survival results with additional follow-up.
DESTINY-PanTumor02 is funded by Daiichi Sankyo. Dr. Meric-Bernstam disclosed a consulting/advisory role with multiple pharmaceutical companies, research funding to her institution from Daiichi Sankyo and others, and travel expenses from ESMO and EORTC. Dr. McGregor disclosed a consulting/advisory role and institutional research funding with multiple companies, not including the study’s funder. Dr. Gralow disclosed a consulting or advisory role with Genentech and Roche.
A version of this article first appeared on Medscape.com.
AT ASCO 2023