User login
How physicians can provide better care to transgender patients
People who identify as transgender experience many health disparities, in addition to lack of access to quality care. The most commonly cited barrier is the lack of providers who are knowledgeable about transgender health care, according to past surveys.
Even those who do seek care often have unpleasant experiences. A 2015 survey conducted by the National Center for Transgender Equality found that 33% of those who saw a health care provider reported at least one unfavorable experience related to being transgender, such as being verbally harassed or refused treatment because of their gender identity. In fact, 23% of those surveyed say they did not seek health care they needed in the past year because of fear of being mistreated as a transgender person.
This interview has been edited for length and clarity.
Question: Surveys have shown that many people who identify as transgender will seek only transition care, not primary or preventive care. Why is that?
Dr. Brandt: My answer is multifactorial. Transgender patients do seek primary care – just not as readily. There’s a lot of misconceptions about health care needs for the LGBT community in general. For example, lesbian or bisexual women may be not as well informed about the need for Pap smears compared with their heterosexual counterparts. These misconceptions are further exacerbated in the transgender community.
The fact that a lot of patients seek only transition-related care, but not preventive services, such as primary care and gynecologic care, is also related to fears of discrimination and lack of education of providers. These patients are afraid when they walk into an office that they will be misgendered or their physician won’t be familiar with their health care needs.
What can clinics and clinicians do to create a safe and welcoming environment?
Dr. Brandt: It starts with educating office staff about terminology and gender identities.
A key feature of our EHR is the sexual orientation and gender identity platform, which asks questions about a patient’s gender identity, sexual orientation, sex assigned at birth, and organ inventory. These data are then found in the patient information tab and are just as relevant as their insurance status, age, and date of birth.
There are many ways a doctor’s office can signal to patients that they are inclusive. They can hang LGBTQ-friendly flags or symbols or a sign saying, “We have an anti-discrimination policy” in the waiting room. A welcoming environment can also be achieved by revising patient questionnaires or forms so that they aren’t gender-specific or binary.
Given that the patient may have limited contact with a primary care clinician, how do you prioritize what you address during the visit?
Dr. Brandt: Similar to cisgender patients, it depends initially on the age of the patient and the reason for the visit. The priorities of an otherwise healthy transgender patient in their 20s are going to be largely the same as for a cisgender patient of the same age. As patients age in the primary care world, you’re addressing more issues, such as colorectal screening, lipid disorders, and mammograms, and that doesn’t change. For the most part, the problems that you address should be specific for that age group.
It becomes more complicated when you add in factors such as hormone therapy and whether patients have had any type of gender-affirming surgery. Those things can change the usual recommendations for screening or risk assessment. We try to figure out what routine health maintenance and cancer screening a patient needs based on age and risk factors, in addition to hormone status and surgical state.
Do you think that many physicians are educated about the care of underserved populations such as transgender patients?
Dr. Brandt: Yes and no. We are definitely getting better at it. For example, the American College of Obstetricians and Gynecologists published a committee opinion highlighting transgender care. So organizations are starting to prioritize these populations and recognize that they are, in fact, underserved and they have special health care needs.
However, the knowledge gaps are still pretty big. I get calls daily from providers asking questions about how to manage patients on hormones, or how to examine a patient who has undergone a vaginoplasty. I hear a lot of horror stories from transgender patients who had their hormones stopped for absurd and medically misinformed reasons.
But I definitely think it’s getting better and it’s being addressed at all levels – the medical school level, the residency level, and the attending level. It just takes time to inform people and for people to get used to the health care needs of these patients.
What should physicians keep in mind when treating patients who identify as transgender?
Dr. Brandt: First and foremost, understanding the terminology and the difference between gender identity, sex, and sexual orientation. Being familiar with that language and being able to speak that language very comfortably and not being awkward about it is a really important thing for primary care physicians and indeed any physician who treats transgender patients.
Physicians should also be aware that any underserved population has higher rates of mental health issues, such as depression and anxiety. Obviously, that goes along with being underserved and the stigma and the disparities that exist for these patients. Having providers educate themselves about what those disparities are and how they impact a patient’s daily life and health is paramount to knowing how to treat patients.
What are your top health concerns for these patients and how do you address them?
Dr. Brandt: I think mental health and safety is probably the number one for me. About 41% of transgender adults have attempted suicide. That number is roughly 51% in transgender youth. That is an astonishing number. These patients have much higher rates of domestic violence, intimate partner violence, and sexual assault, especially trans women and trans women of color. So understanding those statistics is huge.
Obesity, smoking, and substance abuse are my next three. Again, those are things that should be addressed at any visit, regardless of the gender identity or sexual orientation of the patient, but those rates are particularly high in this population.
Fertility and long-term care for patients should be addressed. Many patients who identify as transgender are told they can’t have a family. As a primary care physician, you may see a patient before they are seen by an ob.gyn. or surgeon. Talking about what a patient’s long-term life goals are with fertility and family planning, and what that looks like for them, is a big thing for me. Other providers may not feel that’s a concern, but I believe it should be discussed before initiation of hormone therapy, which can significantly impact fertility in some patients.
Are there nuances to the physical examination that primary care physicians should be aware of when dealing with transmasculine patients vs. transfeminine patients?
Dr. Brandt: Absolutely. And this interview can’t cover the scope of those nuances. An example that comes to mind is the genital exam. For transgender women who have undergone a vaginoplasty, the pelvic exam can be very affirming. Whereas for transgender men, a gynecologic exam can significantly exacerbate dysphoria and there are ways to conduct the exam to limit this discomfort and avoid creating a traumatic experience for the patient. It’s important to be aware that the genital exam, or any type of genitourinary exam, can be either affirming or not affirming.
Sexually transmitted infections are up in the general population, and the trans population is at even higher risk. What should physicians think about when they assess this risk?
Dr. Brandt: It’s really important for primary care clinicians and for gynecologists to learn to be comfortable talking about sexual practices, because what people do behind closed doors is really a key to how to counsel patients about safe sex.
People are well aware of the need to have safe sex. However, depending on the type of sex that you’re having, what body parts go where, what is truly safe can vary and people may not know, for example, to wear a condom when sex toys are involved or that a transgender male on testosterone can become pregnant during penile-vaginal intercourse. Providers really should be very educated on the array of sexual practices that people have and how to counsel them about those. They should know how to ask patients the gender identity of their sexual partners, the sexual orientation of their partners, and what parts go where during sex.
Providers should also talk to patients about PrEP [pre-exposure prophylaxis], whether they identify as cisgender or transgender. My trans patients tend to be a lot more educated about PrEP than other patients. It’s something that many of the residents, even in a standard gynecologic clinic, for example, don’t talk to cisgender patients about because of the stigma surrounding HIV. Many providers still think that the only people who are at risk for HIV are men who have sex with men. And while those rates are higher in some populations, depending on sexual practices, those aren’t the only patients who qualify for PrEP.
Overall, in order to counsel patients about STIs and safe sexual practices, providers should learn to be comfortable talking about sex.
Do you have any strategies on how to make the appointment more successful in addressing those issues?
Dr. Brandt: Bedside manner is a hard thing to teach, and comfort in talking about sex, gender identity, and sexual orientation can vary – but there are a lot of continuing medical education courses that physicians can utilize through the World Professional Association for Transgender Health.
If providers start to notice an influx of patients who identify as transgender or if they want to start seeing transgender patients, it’s really important for them to have that training before they start interacting with patients. In all of medicine, we sort of learn as we go, but this patient population has been subjected to discrimination, violence, error, and misgendering. They have dealt with providers who didn’t understand their health care needs. While this field is evolving, knowing how to appropriately address a patient (using their correct name, pronouns, etc.) is an absolute must.
That needs to be part of a provider’s routine vernacular and not something that they sort of stumble through. You can scare a patient away as soon as they walk into the office with an uneducated front desk staff and things that are seen in the office. Seeking out those educational tools, being aware of your own deficits as a provider and the educational needs of your office, and addressing those needs is really key.
A version of this article first appeared on Medscape.com.
People who identify as transgender experience many health disparities, in addition to lack of access to quality care. The most commonly cited barrier is the lack of providers who are knowledgeable about transgender health care, according to past surveys.
Even those who do seek care often have unpleasant experiences. A 2015 survey conducted by the National Center for Transgender Equality found that 33% of those who saw a health care provider reported at least one unfavorable experience related to being transgender, such as being verbally harassed or refused treatment because of their gender identity. In fact, 23% of those surveyed say they did not seek health care they needed in the past year because of fear of being mistreated as a transgender person.
This interview has been edited for length and clarity.
Question: Surveys have shown that many people who identify as transgender will seek only transition care, not primary or preventive care. Why is that?
Dr. Brandt: My answer is multifactorial. Transgender patients do seek primary care – just not as readily. There’s a lot of misconceptions about health care needs for the LGBT community in general. For example, lesbian or bisexual women may be not as well informed about the need for Pap smears compared with their heterosexual counterparts. These misconceptions are further exacerbated in the transgender community.
The fact that a lot of patients seek only transition-related care, but not preventive services, such as primary care and gynecologic care, is also related to fears of discrimination and lack of education of providers. These patients are afraid when they walk into an office that they will be misgendered or their physician won’t be familiar with their health care needs.
What can clinics and clinicians do to create a safe and welcoming environment?
Dr. Brandt: It starts with educating office staff about terminology and gender identities.
A key feature of our EHR is the sexual orientation and gender identity platform, which asks questions about a patient’s gender identity, sexual orientation, sex assigned at birth, and organ inventory. These data are then found in the patient information tab and are just as relevant as their insurance status, age, and date of birth.
There are many ways a doctor’s office can signal to patients that they are inclusive. They can hang LGBTQ-friendly flags or symbols or a sign saying, “We have an anti-discrimination policy” in the waiting room. A welcoming environment can also be achieved by revising patient questionnaires or forms so that they aren’t gender-specific or binary.
Given that the patient may have limited contact with a primary care clinician, how do you prioritize what you address during the visit?
Dr. Brandt: Similar to cisgender patients, it depends initially on the age of the patient and the reason for the visit. The priorities of an otherwise healthy transgender patient in their 20s are going to be largely the same as for a cisgender patient of the same age. As patients age in the primary care world, you’re addressing more issues, such as colorectal screening, lipid disorders, and mammograms, and that doesn’t change. For the most part, the problems that you address should be specific for that age group.
It becomes more complicated when you add in factors such as hormone therapy and whether patients have had any type of gender-affirming surgery. Those things can change the usual recommendations for screening or risk assessment. We try to figure out what routine health maintenance and cancer screening a patient needs based on age and risk factors, in addition to hormone status and surgical state.
Do you think that many physicians are educated about the care of underserved populations such as transgender patients?
Dr. Brandt: Yes and no. We are definitely getting better at it. For example, the American College of Obstetricians and Gynecologists published a committee opinion highlighting transgender care. So organizations are starting to prioritize these populations and recognize that they are, in fact, underserved and they have special health care needs.
However, the knowledge gaps are still pretty big. I get calls daily from providers asking questions about how to manage patients on hormones, or how to examine a patient who has undergone a vaginoplasty. I hear a lot of horror stories from transgender patients who had their hormones stopped for absurd and medically misinformed reasons.
But I definitely think it’s getting better and it’s being addressed at all levels – the medical school level, the residency level, and the attending level. It just takes time to inform people and for people to get used to the health care needs of these patients.
What should physicians keep in mind when treating patients who identify as transgender?
Dr. Brandt: First and foremost, understanding the terminology and the difference between gender identity, sex, and sexual orientation. Being familiar with that language and being able to speak that language very comfortably and not being awkward about it is a really important thing for primary care physicians and indeed any physician who treats transgender patients.
Physicians should also be aware that any underserved population has higher rates of mental health issues, such as depression and anxiety. Obviously, that goes along with being underserved and the stigma and the disparities that exist for these patients. Having providers educate themselves about what those disparities are and how they impact a patient’s daily life and health is paramount to knowing how to treat patients.
What are your top health concerns for these patients and how do you address them?
Dr. Brandt: I think mental health and safety is probably the number one for me. About 41% of transgender adults have attempted suicide. That number is roughly 51% in transgender youth. That is an astonishing number. These patients have much higher rates of domestic violence, intimate partner violence, and sexual assault, especially trans women and trans women of color. So understanding those statistics is huge.
Obesity, smoking, and substance abuse are my next three. Again, those are things that should be addressed at any visit, regardless of the gender identity or sexual orientation of the patient, but those rates are particularly high in this population.
Fertility and long-term care for patients should be addressed. Many patients who identify as transgender are told they can’t have a family. As a primary care physician, you may see a patient before they are seen by an ob.gyn. or surgeon. Talking about what a patient’s long-term life goals are with fertility and family planning, and what that looks like for them, is a big thing for me. Other providers may not feel that’s a concern, but I believe it should be discussed before initiation of hormone therapy, which can significantly impact fertility in some patients.
Are there nuances to the physical examination that primary care physicians should be aware of when dealing with transmasculine patients vs. transfeminine patients?
Dr. Brandt: Absolutely. And this interview can’t cover the scope of those nuances. An example that comes to mind is the genital exam. For transgender women who have undergone a vaginoplasty, the pelvic exam can be very affirming. Whereas for transgender men, a gynecologic exam can significantly exacerbate dysphoria and there are ways to conduct the exam to limit this discomfort and avoid creating a traumatic experience for the patient. It’s important to be aware that the genital exam, or any type of genitourinary exam, can be either affirming or not affirming.
Sexually transmitted infections are up in the general population, and the trans population is at even higher risk. What should physicians think about when they assess this risk?
Dr. Brandt: It’s really important for primary care clinicians and for gynecologists to learn to be comfortable talking about sexual practices, because what people do behind closed doors is really a key to how to counsel patients about safe sex.
People are well aware of the need to have safe sex. However, depending on the type of sex that you’re having, what body parts go where, what is truly safe can vary and people may not know, for example, to wear a condom when sex toys are involved or that a transgender male on testosterone can become pregnant during penile-vaginal intercourse. Providers really should be very educated on the array of sexual practices that people have and how to counsel them about those. They should know how to ask patients the gender identity of their sexual partners, the sexual orientation of their partners, and what parts go where during sex.
Providers should also talk to patients about PrEP [pre-exposure prophylaxis], whether they identify as cisgender or transgender. My trans patients tend to be a lot more educated about PrEP than other patients. It’s something that many of the residents, even in a standard gynecologic clinic, for example, don’t talk to cisgender patients about because of the stigma surrounding HIV. Many providers still think that the only people who are at risk for HIV are men who have sex with men. And while those rates are higher in some populations, depending on sexual practices, those aren’t the only patients who qualify for PrEP.
Overall, in order to counsel patients about STIs and safe sexual practices, providers should learn to be comfortable talking about sex.
Do you have any strategies on how to make the appointment more successful in addressing those issues?
Dr. Brandt: Bedside manner is a hard thing to teach, and comfort in talking about sex, gender identity, and sexual orientation can vary – but there are a lot of continuing medical education courses that physicians can utilize through the World Professional Association for Transgender Health.
If providers start to notice an influx of patients who identify as transgender or if they want to start seeing transgender patients, it’s really important for them to have that training before they start interacting with patients. In all of medicine, we sort of learn as we go, but this patient population has been subjected to discrimination, violence, error, and misgendering. They have dealt with providers who didn’t understand their health care needs. While this field is evolving, knowing how to appropriately address a patient (using their correct name, pronouns, etc.) is an absolute must.
That needs to be part of a provider’s routine vernacular and not something that they sort of stumble through. You can scare a patient away as soon as they walk into the office with an uneducated front desk staff and things that are seen in the office. Seeking out those educational tools, being aware of your own deficits as a provider and the educational needs of your office, and addressing those needs is really key.
A version of this article first appeared on Medscape.com.
People who identify as transgender experience many health disparities, in addition to lack of access to quality care. The most commonly cited barrier is the lack of providers who are knowledgeable about transgender health care, according to past surveys.
Even those who do seek care often have unpleasant experiences. A 2015 survey conducted by the National Center for Transgender Equality found that 33% of those who saw a health care provider reported at least one unfavorable experience related to being transgender, such as being verbally harassed or refused treatment because of their gender identity. In fact, 23% of those surveyed say they did not seek health care they needed in the past year because of fear of being mistreated as a transgender person.
This interview has been edited for length and clarity.
Question: Surveys have shown that many people who identify as transgender will seek only transition care, not primary or preventive care. Why is that?
Dr. Brandt: My answer is multifactorial. Transgender patients do seek primary care – just not as readily. There’s a lot of misconceptions about health care needs for the LGBT community in general. For example, lesbian or bisexual women may be not as well informed about the need for Pap smears compared with their heterosexual counterparts. These misconceptions are further exacerbated in the transgender community.
The fact that a lot of patients seek only transition-related care, but not preventive services, such as primary care and gynecologic care, is also related to fears of discrimination and lack of education of providers. These patients are afraid when they walk into an office that they will be misgendered or their physician won’t be familiar with their health care needs.
What can clinics and clinicians do to create a safe and welcoming environment?
Dr. Brandt: It starts with educating office staff about terminology and gender identities.
A key feature of our EHR is the sexual orientation and gender identity platform, which asks questions about a patient’s gender identity, sexual orientation, sex assigned at birth, and organ inventory. These data are then found in the patient information tab and are just as relevant as their insurance status, age, and date of birth.
There are many ways a doctor’s office can signal to patients that they are inclusive. They can hang LGBTQ-friendly flags or symbols or a sign saying, “We have an anti-discrimination policy” in the waiting room. A welcoming environment can also be achieved by revising patient questionnaires or forms so that they aren’t gender-specific or binary.
Given that the patient may have limited contact with a primary care clinician, how do you prioritize what you address during the visit?
Dr. Brandt: Similar to cisgender patients, it depends initially on the age of the patient and the reason for the visit. The priorities of an otherwise healthy transgender patient in their 20s are going to be largely the same as for a cisgender patient of the same age. As patients age in the primary care world, you’re addressing more issues, such as colorectal screening, lipid disorders, and mammograms, and that doesn’t change. For the most part, the problems that you address should be specific for that age group.
It becomes more complicated when you add in factors such as hormone therapy and whether patients have had any type of gender-affirming surgery. Those things can change the usual recommendations for screening or risk assessment. We try to figure out what routine health maintenance and cancer screening a patient needs based on age and risk factors, in addition to hormone status and surgical state.
Do you think that many physicians are educated about the care of underserved populations such as transgender patients?
Dr. Brandt: Yes and no. We are definitely getting better at it. For example, the American College of Obstetricians and Gynecologists published a committee opinion highlighting transgender care. So organizations are starting to prioritize these populations and recognize that they are, in fact, underserved and they have special health care needs.
However, the knowledge gaps are still pretty big. I get calls daily from providers asking questions about how to manage patients on hormones, or how to examine a patient who has undergone a vaginoplasty. I hear a lot of horror stories from transgender patients who had their hormones stopped for absurd and medically misinformed reasons.
But I definitely think it’s getting better and it’s being addressed at all levels – the medical school level, the residency level, and the attending level. It just takes time to inform people and for people to get used to the health care needs of these patients.
What should physicians keep in mind when treating patients who identify as transgender?
Dr. Brandt: First and foremost, understanding the terminology and the difference between gender identity, sex, and sexual orientation. Being familiar with that language and being able to speak that language very comfortably and not being awkward about it is a really important thing for primary care physicians and indeed any physician who treats transgender patients.
Physicians should also be aware that any underserved population has higher rates of mental health issues, such as depression and anxiety. Obviously, that goes along with being underserved and the stigma and the disparities that exist for these patients. Having providers educate themselves about what those disparities are and how they impact a patient’s daily life and health is paramount to knowing how to treat patients.
What are your top health concerns for these patients and how do you address them?
Dr. Brandt: I think mental health and safety is probably the number one for me. About 41% of transgender adults have attempted suicide. That number is roughly 51% in transgender youth. That is an astonishing number. These patients have much higher rates of domestic violence, intimate partner violence, and sexual assault, especially trans women and trans women of color. So understanding those statistics is huge.
Obesity, smoking, and substance abuse are my next three. Again, those are things that should be addressed at any visit, regardless of the gender identity or sexual orientation of the patient, but those rates are particularly high in this population.
Fertility and long-term care for patients should be addressed. Many patients who identify as transgender are told they can’t have a family. As a primary care physician, you may see a patient before they are seen by an ob.gyn. or surgeon. Talking about what a patient’s long-term life goals are with fertility and family planning, and what that looks like for them, is a big thing for me. Other providers may not feel that’s a concern, but I believe it should be discussed before initiation of hormone therapy, which can significantly impact fertility in some patients.
Are there nuances to the physical examination that primary care physicians should be aware of when dealing with transmasculine patients vs. transfeminine patients?
Dr. Brandt: Absolutely. And this interview can’t cover the scope of those nuances. An example that comes to mind is the genital exam. For transgender women who have undergone a vaginoplasty, the pelvic exam can be very affirming. Whereas for transgender men, a gynecologic exam can significantly exacerbate dysphoria and there are ways to conduct the exam to limit this discomfort and avoid creating a traumatic experience for the patient. It’s important to be aware that the genital exam, or any type of genitourinary exam, can be either affirming or not affirming.
Sexually transmitted infections are up in the general population, and the trans population is at even higher risk. What should physicians think about when they assess this risk?
Dr. Brandt: It’s really important for primary care clinicians and for gynecologists to learn to be comfortable talking about sexual practices, because what people do behind closed doors is really a key to how to counsel patients about safe sex.
People are well aware of the need to have safe sex. However, depending on the type of sex that you’re having, what body parts go where, what is truly safe can vary and people may not know, for example, to wear a condom when sex toys are involved or that a transgender male on testosterone can become pregnant during penile-vaginal intercourse. Providers really should be very educated on the array of sexual practices that people have and how to counsel them about those. They should know how to ask patients the gender identity of their sexual partners, the sexual orientation of their partners, and what parts go where during sex.
Providers should also talk to patients about PrEP [pre-exposure prophylaxis], whether they identify as cisgender or transgender. My trans patients tend to be a lot more educated about PrEP than other patients. It’s something that many of the residents, even in a standard gynecologic clinic, for example, don’t talk to cisgender patients about because of the stigma surrounding HIV. Many providers still think that the only people who are at risk for HIV are men who have sex with men. And while those rates are higher in some populations, depending on sexual practices, those aren’t the only patients who qualify for PrEP.
Overall, in order to counsel patients about STIs and safe sexual practices, providers should learn to be comfortable talking about sex.
Do you have any strategies on how to make the appointment more successful in addressing those issues?
Dr. Brandt: Bedside manner is a hard thing to teach, and comfort in talking about sex, gender identity, and sexual orientation can vary – but there are a lot of continuing medical education courses that physicians can utilize through the World Professional Association for Transgender Health.
If providers start to notice an influx of patients who identify as transgender or if they want to start seeing transgender patients, it’s really important for them to have that training before they start interacting with patients. In all of medicine, we sort of learn as we go, but this patient population has been subjected to discrimination, violence, error, and misgendering. They have dealt with providers who didn’t understand their health care needs. While this field is evolving, knowing how to appropriately address a patient (using their correct name, pronouns, etc.) is an absolute must.
That needs to be part of a provider’s routine vernacular and not something that they sort of stumble through. You can scare a patient away as soon as they walk into the office with an uneducated front desk staff and things that are seen in the office. Seeking out those educational tools, being aware of your own deficits as a provider and the educational needs of your office, and addressing those needs is really key.
A version of this article first appeared on Medscape.com.
Contradictions abound in ‘The End of Mental Illness’
Daniel G. Amen, MD, is an American psychiatrist well-known for his eponymous clinics, television appearances, and series of books on mental health. One of his latest books, “The End of Mental Illness,” summarizes many of his views on the causes of and treatments for mental illnesses.
Dr. Amen’s approaches – such as his advocacy for the widespread use of single photon emission computed tomography (SPECT) imaging – are somewhat controversial and at times fall outside the mainstream of current psychiatric thought. So does “The End of Mental Illness” contain anything of value to the average practicing psychiatrist? (It should be noted that I listened to this as an audiobook and took notes as I listened. This does limit my ability to directly quote portions of the text, but I believe my notes are reliable.)
He begins the book by pointing out that the term “mental illness” might be better replaced with the term “brain illness.” With this shift in terminology, Dr. Amen introduces a theme that recurs throughout the book: That mental illnesses ultimately stem from various ways in which the brain can be harmed. While the suggested change in terminology might help reduce the stigma associated with psychiatric illnesses, Dr. Amen is surprisingly timid about implementing this term in his own book. He repeatedly refers to “brain health/mental health” issues instead of discarding the “mental” term altogether. Even his BRIGHT MINDS acronym for risk factors for mental illnesses includes the term “mind” instead of “brain.”
Continuing the theme of challenging terminology, Dr. Amen goes on to decry the weaknesses of the DSM system of nosology. This is a valid point, because under the current system, the same patient may receive differing diagnoses depending on which provider is seen and how certain symptoms are interpreted. Yet, here again, Dr. Amen does not seem to adhere to his own advice: He uses DSM terminology throughout the book, speaking of depression, anxiety, bipolar disorder, and ADHD. An oddity (which, admittedly, could have been the audiobook reader’s mistake rather than an error in the original text) is that the DSM is referred to as the “Diagnostic and Structural Manual” rather than the Diagnostic and Statistical Manual. He criticizes the DSM for its imprecision, pointing out the variety of symptom combinations that can produce the same diagnoses and how similar symptoms may overlap between differing diagnoses. Yet, his descriptions of common SPECT patterns (his preferred tool to assist in diagnosis) make it clear that here, too, there is a lot of overlap. As an example, ADHD was associated with at least three of the imaging patterns he described. It is also somewhat ironic how Dr. Amen obliquely criticizes the American Psychiatric Association for profiting from the use of the DSM, when SPECT imaging is expensive and profits his own organization.
Dr. Amen repeatedly asserts that psychiatry is unique among medical specialties for making diagnoses based on symptom clusters rather than direct visualization of the affected organ. Yet, psychiatry is not, in fact, unique in making diagnoses in this way. Some examples of diagnoses based on symptom clusters from other medical specialties are systemic lupus erythematosus, fibromyalgia, and chronic fatigue syndrome. Although he asserts that SPECT imaging better demonstrates the root cause of mental illnesses, it is unclear from his book whether this is actually the case.
The descriptions for the ways in which Dr. Amen uses SPECT (which, admittedly, are vague and presumably simplified for a general audience) suggest that he has made observations correlating specific imaging patterns with certain emotional/behavioral outcomes. However, the imaging patterns he describes in the book can be interpreted to represent multiple different mental conditions, making it clear that SPECT is not a laserlike diagnostic tool that produces a single, indisputable diagnosis. Accuracy with SPECT seems especially questionable in light of two case examples he shares where brain imaging was interpreted as representing illness, but the patients were not demonstrating any signs of mental dysfunction. In one case, Dr. Amen opined that the patient’s vibrant spiritual life “overrode” the sick brain, but if this is true,
Patient testimonials are provided, asserting that SPECT imaging helped them know “exactly” what treatment would help them. One cannot help but wonder whether part of the benefit of SPECT imaging is a placebo effect, boosting the confidence of patients that the treatment they are receiving is personalized and scientifically sound. A similar trend is currently seen more broadly in psychiatry with the widespread promotion of pharmacogenetic testing. Such testing may bolster patient confidence in their medication, but its value in improving patient outcomes has not been established.1
Dr. Amen outlines a brief history of mental health care, including differing approaches and therapies from the time of Sigmund Freud up to the present. His outline is somewhat critical of the perceived shortcomings of his psychiatric forebears, yet this seems entirely unnecessary. All scientific disciplines must start somewhere and build from limited knowledge to greater. Is it necessary to belittle Freud for not being able to do SPECT imaging in the 1800s?
Interestingly, Dr. Amen leaves cognitive-behavioral therapy (CBT), a landmark, evidence-based form of psychotherapy, out of his overview of the history of psychiatry. He does go on to mention CBT as part of the treatment offerings of the Amen Clinics, which could leave the lay reader with the incorrect impression that CBT is a treatment unique to Amen Clinics. Similarly, at one point Dr. Amen writes about “what I call automatic negative thoughts.” This phrasing could confuse readers who might not know that automatic thoughts are a concept endemic to CBT.
Dr. Amen writes repeatedly about the Amen Clinics 4 Circles, four key areas of life that can contribute to mental health. These areas are biological, psychological, social, and spiritual. While Amen Clinics may have come up with the term “4 Circles,” the biopsychosocial model of understanding illness was developed by George Engel, MD, in 1977, and current discussions of this model frequently incorporate a spiritual dimension as well.2
Dr. Amen’s writing at times mischaracterizes psychotropic medications in unhelpful ways. He speaks of psychotropic medications generally as being addictive. While this is certainly true for stimulants and benzodiazepines, most would agree that this does not apply to many other commonly used medications in psychiatry, including selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, antipsychotics, and mood stabilizers. He also paints with a broad brush when he states that anxiety medications can cause dementia. A concerning link has been demonstrated between benzodiazepine use and dementia,3 but SSRIs (which are considered first-line medications for anxiety) are not known to cause dementia and may actually delay progression from mild cognitive impairment to Alzheimer’s dementia.4 His mention of medication use affecting a patient’s insurability could have the unfortunate effect of scaring away suffering individuals from seeking help. The one category of psychiatric medication he does not seem concerned about is psychostimulants, which is odd – given the addictive, cardiovascular, and other risks associated with that medication class.
In contrast to his skepticism regarding many psychotropic medications, Dr. Amen expresses significant enthusiasm regarding nutraceutical use. While there has been research in this area supporting a role for some nutraceutical interventions, there is still a need for more rigorous studies.5 To support his endorsement of natural remedies, Dr. Amen mentions that Hippocrates recommended herbs and spices for many health conditions. But Hippocrates lived more than 2,000 years ago, and the state of medicine has advanced significantly since then.
Dr. Amen also mentions that 80% of the developing world relies upon natural or herbal remedies as the primary source of medicine. While he frames this statement as supporting his endorsement of such remedies, it could conversely be said that this is evidence of the need to make pharmacological interventions more widely available in the developing world.
Much of “The End of Mental Illness” is dedicated to reviewing specific risk factors that could cause harm to a person’s mental well-being. One example is head trauma. Dr. Amen documents at least one instance in which he was convinced that his patient had experienced head trauma, and questioned the patient again and again about possible brain injuries. One must wonder whether the positive results of such focused, repetitive questioning might be evidence of confirmation bias, as a search to confirm the preexisting belief of head trauma could lead to overlooking alternative explanations for a patient’s symptoms.
Another risk factor dwelt upon is exposure to toxins. One toxin Dr. Amen rightly recommends avoiding is tobacco smoke. Yet, his approach to advocate for a tobacco-free lifestyle is somewhat problematic. He lists chemicals contained in tobacco smoke, and then names unpleasant items that share those ingredients, such as paint. This smacks of the same sloppy logic manifested in social media memes decrying the use of vaccines by listing their ingredients alongside scary-sounding products that contain identical ingredients (for example, vaccines contain formaldehyde, which is used to embalm dead bodies!). This is analogous to saying that water is bad for you because it contains hydrogen, which is also an ingredient in atomic bombs.
Dr. Amen makes the blanket recommendation to avoid products containing “chemicals.” This is a difficult recommendation to interpret, since literally all matter is made of chemicals. It seems that Dr. Amen is leaning into the vague idea of a “chemical” as something artificially created in a lab, which must, therefore, be dangerous.
Along these lines, Dr. Amen suggests that if a person doesn’t know what is in a specific food item, it should not be eaten. Although this sounds reasonable on the surface, if people were told the names of the proteins and chemical compounds that make up many naturally occurring plants or meats, they would likely not recognize many of them. Dr. Amen dedicates space to list seemingly benign exposures – such as eating nonorganic produce, using two or more beauty products each day, or touching grocery store receipts – as possible “toxins.” By contrast, there is a certain irony in the absence of any mention of the risks associated with radiation from the SPECT imaging he staunchly advocates for. One potential risk of the book listing so many “toxins” to avoid is that patients could waste valuable time and energy eliminating exposures that pose little or no risk, rather than focusing efforts on well-established treatments.
In light of the observations and critiques offered above, one might come away with the impression that I would not recommend “The End of Mental Illness.” However, although one can nitpick details in the book, some of its bigger ideas make it worth commending to readers. Dr. Amen rightfully emphasizes the need for psychiatrists and patients to think more broadly about mental health issues beyond the use of pills. He justifiably criticizes the “15-minute med check” model of practice and the idea that medications are the end-all, be-all of treatment. He demonstrates an appropriate appreciation for the serious risks of reliance on benzodiazepines.6 Dr. Amen points out important contributions from Viktor Frankl, MD, to the field of psychiatry, which may go overlooked today. He also helpfully points out that bipolar disorder may often be misdiagnosed (although he attributes the misdiagnosis to traumatic brain injury, whereas other psychiatrists might say the misdiagnosis is due to borderline personality disorder).
Much of what Dr. Amen writes is sensible, and psychiatrists would do well to adopt the following steps he advocates for: Taking a comprehensive biopsychosocial-spiritual approach to the assessment and treatment of patients; thinking broadly in their differential diagnoses and not forgetting their medical training; understanding that medication alone is often not sufficient to make lasting, positive change in a person’s life; paying attention to healthy habits such as diet, exercise, sleep, and social activity; and knowing that CBT is a valuable tool that can change lives.
There is much to appreciate in “The End of Mental Illness,” especially the overarching idea that psychiatry isn’t just a symptom checklist and a prescription pad. Rather, achieving mental well-being often requires broader thinking and sustained lifestyle changes.
Although I did not agree with everything in the book, it did cause me to think and reflect on my own practice. I read “The End of Mental Illness” with colleagues in my department, and it stimulated a lively discussion. Isn’t that ultimately what a psychiatrist would want from a book like this – the opportunity to reflect, discuss, and potentially improve one’s own practice?
Dr. Weber is physician lead in the department of psychiatry at Intermountain Healthcare Budge Clinic, Logan (Utah) Psychiatry. He disclosed no relevant financial relationships.
References
1. JAMA Netw Open. 2020;3(12). doi: 10.1001/jamanetworkopen.2020.27909.
2. Curr Opin Psychiatry. 2014;27:358-63.
3. BMJ 2014. doi: 10.1136/bmj.g5205.
4. Am J Psychiatry. 2018 Mar 1;175:232-41.
5. Am J Psychiatry. 2016 Jun 1;173:575-87.
6. Current Psychiatry. 2018 Feb;17(2):22-7.
Daniel G. Amen, MD, is an American psychiatrist well-known for his eponymous clinics, television appearances, and series of books on mental health. One of his latest books, “The End of Mental Illness,” summarizes many of his views on the causes of and treatments for mental illnesses.
Dr. Amen’s approaches – such as his advocacy for the widespread use of single photon emission computed tomography (SPECT) imaging – are somewhat controversial and at times fall outside the mainstream of current psychiatric thought. So does “The End of Mental Illness” contain anything of value to the average practicing psychiatrist? (It should be noted that I listened to this as an audiobook and took notes as I listened. This does limit my ability to directly quote portions of the text, but I believe my notes are reliable.)
He begins the book by pointing out that the term “mental illness” might be better replaced with the term “brain illness.” With this shift in terminology, Dr. Amen introduces a theme that recurs throughout the book: That mental illnesses ultimately stem from various ways in which the brain can be harmed. While the suggested change in terminology might help reduce the stigma associated with psychiatric illnesses, Dr. Amen is surprisingly timid about implementing this term in his own book. He repeatedly refers to “brain health/mental health” issues instead of discarding the “mental” term altogether. Even his BRIGHT MINDS acronym for risk factors for mental illnesses includes the term “mind” instead of “brain.”
Continuing the theme of challenging terminology, Dr. Amen goes on to decry the weaknesses of the DSM system of nosology. This is a valid point, because under the current system, the same patient may receive differing diagnoses depending on which provider is seen and how certain symptoms are interpreted. Yet, here again, Dr. Amen does not seem to adhere to his own advice: He uses DSM terminology throughout the book, speaking of depression, anxiety, bipolar disorder, and ADHD. An oddity (which, admittedly, could have been the audiobook reader’s mistake rather than an error in the original text) is that the DSM is referred to as the “Diagnostic and Structural Manual” rather than the Diagnostic and Statistical Manual. He criticizes the DSM for its imprecision, pointing out the variety of symptom combinations that can produce the same diagnoses and how similar symptoms may overlap between differing diagnoses. Yet, his descriptions of common SPECT patterns (his preferred tool to assist in diagnosis) make it clear that here, too, there is a lot of overlap. As an example, ADHD was associated with at least three of the imaging patterns he described. It is also somewhat ironic how Dr. Amen obliquely criticizes the American Psychiatric Association for profiting from the use of the DSM, when SPECT imaging is expensive and profits his own organization.
Dr. Amen repeatedly asserts that psychiatry is unique among medical specialties for making diagnoses based on symptom clusters rather than direct visualization of the affected organ. Yet, psychiatry is not, in fact, unique in making diagnoses in this way. Some examples of diagnoses based on symptom clusters from other medical specialties are systemic lupus erythematosus, fibromyalgia, and chronic fatigue syndrome. Although he asserts that SPECT imaging better demonstrates the root cause of mental illnesses, it is unclear from his book whether this is actually the case.
The descriptions for the ways in which Dr. Amen uses SPECT (which, admittedly, are vague and presumably simplified for a general audience) suggest that he has made observations correlating specific imaging patterns with certain emotional/behavioral outcomes. However, the imaging patterns he describes in the book can be interpreted to represent multiple different mental conditions, making it clear that SPECT is not a laserlike diagnostic tool that produces a single, indisputable diagnosis. Accuracy with SPECT seems especially questionable in light of two case examples he shares where brain imaging was interpreted as representing illness, but the patients were not demonstrating any signs of mental dysfunction. In one case, Dr. Amen opined that the patient’s vibrant spiritual life “overrode” the sick brain, but if this is true,
Patient testimonials are provided, asserting that SPECT imaging helped them know “exactly” what treatment would help them. One cannot help but wonder whether part of the benefit of SPECT imaging is a placebo effect, boosting the confidence of patients that the treatment they are receiving is personalized and scientifically sound. A similar trend is currently seen more broadly in psychiatry with the widespread promotion of pharmacogenetic testing. Such testing may bolster patient confidence in their medication, but its value in improving patient outcomes has not been established.1
Dr. Amen outlines a brief history of mental health care, including differing approaches and therapies from the time of Sigmund Freud up to the present. His outline is somewhat critical of the perceived shortcomings of his psychiatric forebears, yet this seems entirely unnecessary. All scientific disciplines must start somewhere and build from limited knowledge to greater. Is it necessary to belittle Freud for not being able to do SPECT imaging in the 1800s?
Interestingly, Dr. Amen leaves cognitive-behavioral therapy (CBT), a landmark, evidence-based form of psychotherapy, out of his overview of the history of psychiatry. He does go on to mention CBT as part of the treatment offerings of the Amen Clinics, which could leave the lay reader with the incorrect impression that CBT is a treatment unique to Amen Clinics. Similarly, at one point Dr. Amen writes about “what I call automatic negative thoughts.” This phrasing could confuse readers who might not know that automatic thoughts are a concept endemic to CBT.
Dr. Amen writes repeatedly about the Amen Clinics 4 Circles, four key areas of life that can contribute to mental health. These areas are biological, psychological, social, and spiritual. While Amen Clinics may have come up with the term “4 Circles,” the biopsychosocial model of understanding illness was developed by George Engel, MD, in 1977, and current discussions of this model frequently incorporate a spiritual dimension as well.2
Dr. Amen’s writing at times mischaracterizes psychotropic medications in unhelpful ways. He speaks of psychotropic medications generally as being addictive. While this is certainly true for stimulants and benzodiazepines, most would agree that this does not apply to many other commonly used medications in psychiatry, including selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, antipsychotics, and mood stabilizers. He also paints with a broad brush when he states that anxiety medications can cause dementia. A concerning link has been demonstrated between benzodiazepine use and dementia,3 but SSRIs (which are considered first-line medications for anxiety) are not known to cause dementia and may actually delay progression from mild cognitive impairment to Alzheimer’s dementia.4 His mention of medication use affecting a patient’s insurability could have the unfortunate effect of scaring away suffering individuals from seeking help. The one category of psychiatric medication he does not seem concerned about is psychostimulants, which is odd – given the addictive, cardiovascular, and other risks associated with that medication class.
In contrast to his skepticism regarding many psychotropic medications, Dr. Amen expresses significant enthusiasm regarding nutraceutical use. While there has been research in this area supporting a role for some nutraceutical interventions, there is still a need for more rigorous studies.5 To support his endorsement of natural remedies, Dr. Amen mentions that Hippocrates recommended herbs and spices for many health conditions. But Hippocrates lived more than 2,000 years ago, and the state of medicine has advanced significantly since then.
Dr. Amen also mentions that 80% of the developing world relies upon natural or herbal remedies as the primary source of medicine. While he frames this statement as supporting his endorsement of such remedies, it could conversely be said that this is evidence of the need to make pharmacological interventions more widely available in the developing world.
Much of “The End of Mental Illness” is dedicated to reviewing specific risk factors that could cause harm to a person’s mental well-being. One example is head trauma. Dr. Amen documents at least one instance in which he was convinced that his patient had experienced head trauma, and questioned the patient again and again about possible brain injuries. One must wonder whether the positive results of such focused, repetitive questioning might be evidence of confirmation bias, as a search to confirm the preexisting belief of head trauma could lead to overlooking alternative explanations for a patient’s symptoms.
Another risk factor dwelt upon is exposure to toxins. One toxin Dr. Amen rightly recommends avoiding is tobacco smoke. Yet, his approach to advocate for a tobacco-free lifestyle is somewhat problematic. He lists chemicals contained in tobacco smoke, and then names unpleasant items that share those ingredients, such as paint. This smacks of the same sloppy logic manifested in social media memes decrying the use of vaccines by listing their ingredients alongside scary-sounding products that contain identical ingredients (for example, vaccines contain formaldehyde, which is used to embalm dead bodies!). This is analogous to saying that water is bad for you because it contains hydrogen, which is also an ingredient in atomic bombs.
Dr. Amen makes the blanket recommendation to avoid products containing “chemicals.” This is a difficult recommendation to interpret, since literally all matter is made of chemicals. It seems that Dr. Amen is leaning into the vague idea of a “chemical” as something artificially created in a lab, which must, therefore, be dangerous.
Along these lines, Dr. Amen suggests that if a person doesn’t know what is in a specific food item, it should not be eaten. Although this sounds reasonable on the surface, if people were told the names of the proteins and chemical compounds that make up many naturally occurring plants or meats, they would likely not recognize many of them. Dr. Amen dedicates space to list seemingly benign exposures – such as eating nonorganic produce, using two or more beauty products each day, or touching grocery store receipts – as possible “toxins.” By contrast, there is a certain irony in the absence of any mention of the risks associated with radiation from the SPECT imaging he staunchly advocates for. One potential risk of the book listing so many “toxins” to avoid is that patients could waste valuable time and energy eliminating exposures that pose little or no risk, rather than focusing efforts on well-established treatments.
In light of the observations and critiques offered above, one might come away with the impression that I would not recommend “The End of Mental Illness.” However, although one can nitpick details in the book, some of its bigger ideas make it worth commending to readers. Dr. Amen rightfully emphasizes the need for psychiatrists and patients to think more broadly about mental health issues beyond the use of pills. He justifiably criticizes the “15-minute med check” model of practice and the idea that medications are the end-all, be-all of treatment. He demonstrates an appropriate appreciation for the serious risks of reliance on benzodiazepines.6 Dr. Amen points out important contributions from Viktor Frankl, MD, to the field of psychiatry, which may go overlooked today. He also helpfully points out that bipolar disorder may often be misdiagnosed (although he attributes the misdiagnosis to traumatic brain injury, whereas other psychiatrists might say the misdiagnosis is due to borderline personality disorder).
Much of what Dr. Amen writes is sensible, and psychiatrists would do well to adopt the following steps he advocates for: Taking a comprehensive biopsychosocial-spiritual approach to the assessment and treatment of patients; thinking broadly in their differential diagnoses and not forgetting their medical training; understanding that medication alone is often not sufficient to make lasting, positive change in a person’s life; paying attention to healthy habits such as diet, exercise, sleep, and social activity; and knowing that CBT is a valuable tool that can change lives.
There is much to appreciate in “The End of Mental Illness,” especially the overarching idea that psychiatry isn’t just a symptom checklist and a prescription pad. Rather, achieving mental well-being often requires broader thinking and sustained lifestyle changes.
Although I did not agree with everything in the book, it did cause me to think and reflect on my own practice. I read “The End of Mental Illness” with colleagues in my department, and it stimulated a lively discussion. Isn’t that ultimately what a psychiatrist would want from a book like this – the opportunity to reflect, discuss, and potentially improve one’s own practice?
Dr. Weber is physician lead in the department of psychiatry at Intermountain Healthcare Budge Clinic, Logan (Utah) Psychiatry. He disclosed no relevant financial relationships.
References
1. JAMA Netw Open. 2020;3(12). doi: 10.1001/jamanetworkopen.2020.27909.
2. Curr Opin Psychiatry. 2014;27:358-63.
3. BMJ 2014. doi: 10.1136/bmj.g5205.
4. Am J Psychiatry. 2018 Mar 1;175:232-41.
5. Am J Psychiatry. 2016 Jun 1;173:575-87.
6. Current Psychiatry. 2018 Feb;17(2):22-7.
Daniel G. Amen, MD, is an American psychiatrist well-known for his eponymous clinics, television appearances, and series of books on mental health. One of his latest books, “The End of Mental Illness,” summarizes many of his views on the causes of and treatments for mental illnesses.
Dr. Amen’s approaches – such as his advocacy for the widespread use of single photon emission computed tomography (SPECT) imaging – are somewhat controversial and at times fall outside the mainstream of current psychiatric thought. So does “The End of Mental Illness” contain anything of value to the average practicing psychiatrist? (It should be noted that I listened to this as an audiobook and took notes as I listened. This does limit my ability to directly quote portions of the text, but I believe my notes are reliable.)
He begins the book by pointing out that the term “mental illness” might be better replaced with the term “brain illness.” With this shift in terminology, Dr. Amen introduces a theme that recurs throughout the book: That mental illnesses ultimately stem from various ways in which the brain can be harmed. While the suggested change in terminology might help reduce the stigma associated with psychiatric illnesses, Dr. Amen is surprisingly timid about implementing this term in his own book. He repeatedly refers to “brain health/mental health” issues instead of discarding the “mental” term altogether. Even his BRIGHT MINDS acronym for risk factors for mental illnesses includes the term “mind” instead of “brain.”
Continuing the theme of challenging terminology, Dr. Amen goes on to decry the weaknesses of the DSM system of nosology. This is a valid point, because under the current system, the same patient may receive differing diagnoses depending on which provider is seen and how certain symptoms are interpreted. Yet, here again, Dr. Amen does not seem to adhere to his own advice: He uses DSM terminology throughout the book, speaking of depression, anxiety, bipolar disorder, and ADHD. An oddity (which, admittedly, could have been the audiobook reader’s mistake rather than an error in the original text) is that the DSM is referred to as the “Diagnostic and Structural Manual” rather than the Diagnostic and Statistical Manual. He criticizes the DSM for its imprecision, pointing out the variety of symptom combinations that can produce the same diagnoses and how similar symptoms may overlap between differing diagnoses. Yet, his descriptions of common SPECT patterns (his preferred tool to assist in diagnosis) make it clear that here, too, there is a lot of overlap. As an example, ADHD was associated with at least three of the imaging patterns he described. It is also somewhat ironic how Dr. Amen obliquely criticizes the American Psychiatric Association for profiting from the use of the DSM, when SPECT imaging is expensive and profits his own organization.
Dr. Amen repeatedly asserts that psychiatry is unique among medical specialties for making diagnoses based on symptom clusters rather than direct visualization of the affected organ. Yet, psychiatry is not, in fact, unique in making diagnoses in this way. Some examples of diagnoses based on symptom clusters from other medical specialties are systemic lupus erythematosus, fibromyalgia, and chronic fatigue syndrome. Although he asserts that SPECT imaging better demonstrates the root cause of mental illnesses, it is unclear from his book whether this is actually the case.
The descriptions for the ways in which Dr. Amen uses SPECT (which, admittedly, are vague and presumably simplified for a general audience) suggest that he has made observations correlating specific imaging patterns with certain emotional/behavioral outcomes. However, the imaging patterns he describes in the book can be interpreted to represent multiple different mental conditions, making it clear that SPECT is not a laserlike diagnostic tool that produces a single, indisputable diagnosis. Accuracy with SPECT seems especially questionable in light of two case examples he shares where brain imaging was interpreted as representing illness, but the patients were not demonstrating any signs of mental dysfunction. In one case, Dr. Amen opined that the patient’s vibrant spiritual life “overrode” the sick brain, but if this is true,
Patient testimonials are provided, asserting that SPECT imaging helped them know “exactly” what treatment would help them. One cannot help but wonder whether part of the benefit of SPECT imaging is a placebo effect, boosting the confidence of patients that the treatment they are receiving is personalized and scientifically sound. A similar trend is currently seen more broadly in psychiatry with the widespread promotion of pharmacogenetic testing. Such testing may bolster patient confidence in their medication, but its value in improving patient outcomes has not been established.1
Dr. Amen outlines a brief history of mental health care, including differing approaches and therapies from the time of Sigmund Freud up to the present. His outline is somewhat critical of the perceived shortcomings of his psychiatric forebears, yet this seems entirely unnecessary. All scientific disciplines must start somewhere and build from limited knowledge to greater. Is it necessary to belittle Freud for not being able to do SPECT imaging in the 1800s?
Interestingly, Dr. Amen leaves cognitive-behavioral therapy (CBT), a landmark, evidence-based form of psychotherapy, out of his overview of the history of psychiatry. He does go on to mention CBT as part of the treatment offerings of the Amen Clinics, which could leave the lay reader with the incorrect impression that CBT is a treatment unique to Amen Clinics. Similarly, at one point Dr. Amen writes about “what I call automatic negative thoughts.” This phrasing could confuse readers who might not know that automatic thoughts are a concept endemic to CBT.
Dr. Amen writes repeatedly about the Amen Clinics 4 Circles, four key areas of life that can contribute to mental health. These areas are biological, psychological, social, and spiritual. While Amen Clinics may have come up with the term “4 Circles,” the biopsychosocial model of understanding illness was developed by George Engel, MD, in 1977, and current discussions of this model frequently incorporate a spiritual dimension as well.2
Dr. Amen’s writing at times mischaracterizes psychotropic medications in unhelpful ways. He speaks of psychotropic medications generally as being addictive. While this is certainly true for stimulants and benzodiazepines, most would agree that this does not apply to many other commonly used medications in psychiatry, including selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, antipsychotics, and mood stabilizers. He also paints with a broad brush when he states that anxiety medications can cause dementia. A concerning link has been demonstrated between benzodiazepine use and dementia,3 but SSRIs (which are considered first-line medications for anxiety) are not known to cause dementia and may actually delay progression from mild cognitive impairment to Alzheimer’s dementia.4 His mention of medication use affecting a patient’s insurability could have the unfortunate effect of scaring away suffering individuals from seeking help. The one category of psychiatric medication he does not seem concerned about is psychostimulants, which is odd – given the addictive, cardiovascular, and other risks associated with that medication class.
In contrast to his skepticism regarding many psychotropic medications, Dr. Amen expresses significant enthusiasm regarding nutraceutical use. While there has been research in this area supporting a role for some nutraceutical interventions, there is still a need for more rigorous studies.5 To support his endorsement of natural remedies, Dr. Amen mentions that Hippocrates recommended herbs and spices for many health conditions. But Hippocrates lived more than 2,000 years ago, and the state of medicine has advanced significantly since then.
Dr. Amen also mentions that 80% of the developing world relies upon natural or herbal remedies as the primary source of medicine. While he frames this statement as supporting his endorsement of such remedies, it could conversely be said that this is evidence of the need to make pharmacological interventions more widely available in the developing world.
Much of “The End of Mental Illness” is dedicated to reviewing specific risk factors that could cause harm to a person’s mental well-being. One example is head trauma. Dr. Amen documents at least one instance in which he was convinced that his patient had experienced head trauma, and questioned the patient again and again about possible brain injuries. One must wonder whether the positive results of such focused, repetitive questioning might be evidence of confirmation bias, as a search to confirm the preexisting belief of head trauma could lead to overlooking alternative explanations for a patient’s symptoms.
Another risk factor dwelt upon is exposure to toxins. One toxin Dr. Amen rightly recommends avoiding is tobacco smoke. Yet, his approach to advocate for a tobacco-free lifestyle is somewhat problematic. He lists chemicals contained in tobacco smoke, and then names unpleasant items that share those ingredients, such as paint. This smacks of the same sloppy logic manifested in social media memes decrying the use of vaccines by listing their ingredients alongside scary-sounding products that contain identical ingredients (for example, vaccines contain formaldehyde, which is used to embalm dead bodies!). This is analogous to saying that water is bad for you because it contains hydrogen, which is also an ingredient in atomic bombs.
Dr. Amen makes the blanket recommendation to avoid products containing “chemicals.” This is a difficult recommendation to interpret, since literally all matter is made of chemicals. It seems that Dr. Amen is leaning into the vague idea of a “chemical” as something artificially created in a lab, which must, therefore, be dangerous.
Along these lines, Dr. Amen suggests that if a person doesn’t know what is in a specific food item, it should not be eaten. Although this sounds reasonable on the surface, if people were told the names of the proteins and chemical compounds that make up many naturally occurring plants or meats, they would likely not recognize many of them. Dr. Amen dedicates space to list seemingly benign exposures – such as eating nonorganic produce, using two or more beauty products each day, or touching grocery store receipts – as possible “toxins.” By contrast, there is a certain irony in the absence of any mention of the risks associated with radiation from the SPECT imaging he staunchly advocates for. One potential risk of the book listing so many “toxins” to avoid is that patients could waste valuable time and energy eliminating exposures that pose little or no risk, rather than focusing efforts on well-established treatments.
In light of the observations and critiques offered above, one might come away with the impression that I would not recommend “The End of Mental Illness.” However, although one can nitpick details in the book, some of its bigger ideas make it worth commending to readers. Dr. Amen rightfully emphasizes the need for psychiatrists and patients to think more broadly about mental health issues beyond the use of pills. He justifiably criticizes the “15-minute med check” model of practice and the idea that medications are the end-all, be-all of treatment. He demonstrates an appropriate appreciation for the serious risks of reliance on benzodiazepines.6 Dr. Amen points out important contributions from Viktor Frankl, MD, to the field of psychiatry, which may go overlooked today. He also helpfully points out that bipolar disorder may often be misdiagnosed (although he attributes the misdiagnosis to traumatic brain injury, whereas other psychiatrists might say the misdiagnosis is due to borderline personality disorder).
Much of what Dr. Amen writes is sensible, and psychiatrists would do well to adopt the following steps he advocates for: Taking a comprehensive biopsychosocial-spiritual approach to the assessment and treatment of patients; thinking broadly in their differential diagnoses and not forgetting their medical training; understanding that medication alone is often not sufficient to make lasting, positive change in a person’s life; paying attention to healthy habits such as diet, exercise, sleep, and social activity; and knowing that CBT is a valuable tool that can change lives.
There is much to appreciate in “The End of Mental Illness,” especially the overarching idea that psychiatry isn’t just a symptom checklist and a prescription pad. Rather, achieving mental well-being often requires broader thinking and sustained lifestyle changes.
Although I did not agree with everything in the book, it did cause me to think and reflect on my own practice. I read “The End of Mental Illness” with colleagues in my department, and it stimulated a lively discussion. Isn’t that ultimately what a psychiatrist would want from a book like this – the opportunity to reflect, discuss, and potentially improve one’s own practice?
Dr. Weber is physician lead in the department of psychiatry at Intermountain Healthcare Budge Clinic, Logan (Utah) Psychiatry. He disclosed no relevant financial relationships.
References
1. JAMA Netw Open. 2020;3(12). doi: 10.1001/jamanetworkopen.2020.27909.
2. Curr Opin Psychiatry. 2014;27:358-63.
3. BMJ 2014. doi: 10.1136/bmj.g5205.
4. Am J Psychiatry. 2018 Mar 1;175:232-41.
5. Am J Psychiatry. 2016 Jun 1;173:575-87.
6. Current Psychiatry. 2018 Feb;17(2):22-7.
Gastrointestinal Symptoms and Lactic Acidosis in a Chronic Marijuana User
A 57-year-old woman with a history of traumatic brain injury, posttraumatic stress disorder, depression, migraines, hypothyroidism, and a hiatal hernia repair presented to the emergency department with a 1-day history of nausea, vomiting, and diffuse abdominal pain. She reported that her symptoms were relieved by hot showers. She also reported having similar symptoms and a previous gastric-emptying study that showed a slow-emptying stomach. Her history also consisted of frequent cannabis use for mood and appetite stimulation along with eliminating meat and fish from her diet, an increase in consumption of simple carbohydrates in the past year, and no alcohol use. Her medications included topiramate 100 mg and clonidine 0.3 mg nightly for migraines; levothyroxine 200 mcg daily for hypothyroidism; tizanidine 4 mg twice a day for muscle spasm; famotidine 40 mg twice a day as needed for gastric reflux; and bupropion 50 mg daily, citalopram 20 mg daily, and lamotrigine 25 mg nightly for mood.
The patient’s physical examination was notable for bradycardia (43 beats/min) and epigastric tenderness. Admission laboratory results were notable for an elevated lactic acid level of 4.8 (normal range, 0.50-2.20) mmol/L and a leukocytosis count of 10.8×109 cells/L. Serum alcohol level and blood cultures were negative. Liver function test, hemoglobin A1c, and lipase test were unremarkable. Her electrocardiogram showed an unchanged right bundle branch block. Chest X-ray, computed tomography (CT) of her abdomen/pelvis and echocardiogram were unremarkable.
What is your diagnosis?
How would you treat this patient?
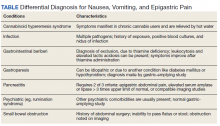
This patient was diagnosed with gastrointestinal beriberi. Because of her dietary changes, lactic acidosis, and bradycardia, thiamine deficiency was suspected after ruling out other possibilities on the differential diagnosis (Table). The patient’s symptoms resolved after administration of high-dose IV thiamine 500 mg 3 times daily for 4 days. Her white blood cell count and lactic acid level normalized. Unfortunately, thiamine levels were not obtained for the patient before treatment was initiated. After administration of IV thiamine, her plasma thiamine level was > 1,200 (normal range, 8-30) nmol/L.
Her differential diagnosis included infectious etiology. Given her leukocytosis and lactic acidosis, vancomycin and piperacillin/tazobactam were started on admission. One day later, her leukocytosis count doubled to 20.7×109 cells/L. However, after 48 hours of negative blood cultures, antibiotics were discontinued.
Small bowel obstruction was suspected due to the patient’s history of abdominal surgery but was ruled out with CT imaging. Similarly, pancreatitis was ruled out based on negative CT imaging and the patient’s normal lipase level. Gastroparesis also was considered because of the patient’s history of hypothyroidism, tobacco use, and her prior gastric-emptying study. The patient was treated for gastroparesis with a course of metoclopramide and erythromycin without improvement in symptoms. Additionally, gastroparesis would not explain the patient’s leukocytosis.
Cannabinoid hyperemesis syndrome (CHS) was suspected because the patient’s symptoms improved with cannabis discontinuation and hot showers.1 In chronic users, however, tetrahydrocannabinol levels have a half-life of 5 to 13 days.2 Although lactic acidosis and leukocytosis have been previously reported with cannabis use, it is unlikely that the patient would have such significant improvement within the first 4 days after discontinuation.1,3,4 Although the patient had many psychiatric comorbidities with previous hospitalizations describing concern for somatization disorder, her leukocytosis and elevated lactic acid levels were suggestive of an organic rather than a psychiatric etiology of her symptoms.
Discussion
Gastrointestinal beriberi has been reported in chronic cannabis users who present with nausea, vomiting, epigastric pain, leukocytosis, and lactic acidosis; all these symptoms rapidly improve after thiamine administration.5,6 The patient’s dietary change also eliminated her intake of vitamin B12, which compounded her condition. Thiamine deficiency produces lactic acidosis by disrupting pyruvate metabolism.7 Bradycardia also can be a sign of thiamine deficiency, although the patient’s use of clonidine for migraines is a confounder.8
Chronically ill patients are prone to nutritional deficiencies, including deficiencies of thiamine.7,9 Many patients with chronic illnesses also use cannabis to ameliorate physical and neuropsychiatric symptoms.2 Recent reports suggest cannabis users are prone to gastrointestinal beriberi and Wernicke encephalopathy.5,10 Treating gastrointestinal symptoms in these patients can be challenging to diagnose because gastrointestinal beriberi and CHS share many clinical manifestations.
The patient’s presentation is likely multifactorial resulting from the combination of gastrointestinal beriberi and CHS. However, thiamine deficiency seems to play the dominant role.
There is no standard treatment regimen for thiamine deficiency with neurologic deficits, and patients only retain about 10 to 15% of intramuscular (IM) injections of cyanocobalamin.11,12 The British Committee for Standards in Haematology recommends IM injections of 1,000 mcg of cyanocobalamin 3 times a week for 2 weeks and then reassess the need for continued treatment.13 The British Columbia guidelines also recommend IM injections of 1,000 mcg daily for 1 to 5 days before transitioning to oral repletion.14 European Neurology guidelines for the treatment of Wernicke encephalopathy recommend IV cyanocobalamin 200 mg 3 times daily.15 Low-level evidence with observational studies informs these decisions and is why there is variation.
The patient’s serum lactate and leukocytosis normalized 1 day after the administration of thiamine. Thiamine deficiency classically causes Wernicke encephalopathy and wet beriberi.16 The patient did not present with Wernicke encephalopathy’s triad: ophthalmoplegia, ataxia, or confusion. She also was euvolemic without signs or symptoms of wet beriberi.
Conclusions
Thiamine deficiency is principally a clinical diagnosis. Thiamine laboratory testing may not be readily available in all medical centers, and confirming a diagnosis of thiamine deficiency should not delay treatment when thiamine deficiency is suspected. This patient’s thiamine levels resulted a week after collection. The administration of thiamine before sampling also can alter the result as it did in this case. Additionally, laboratories may offer whole blood and serum testing. Whole blood testing is more accurate because most bioactive thiamine is found in red blood cells.17
1. Price SL, Fisher C, Kumar R, Hilgerson A. Cannabinoid hyperemesis syndrome as the underlying cause of intractable nausea and vomiting. J Am Osteopath Assoc. 2011;111(3):166-169. doi:10.7556/jaoa.2011.111.3.166
2. Sharma P, Murthy P, Bharath MM. Chemistry, metabolism, and toxicology of cannabis: clinical implications. Iran J Psychiatry. 2012;7(4):149-156.
3. Antill T, Jakkoju A, Dieguez J, Laskhmiprasad L. Lactic acidosis: a rare manifestation of synthetic marijuana intoxication. J La State Med Soc. 2015;167(3):155.
4. Sullivan S. Cannabinoid hyperemesis. Can J Gastroenterol. 2010;24(5):284-285. doi:10.1155/2010/481940
5. Duca J, Lum CJ, Lo AM. Elevated lactate secondary to gastrointestinal beriberi. J Gen Intern Med. 2016;31(1):133-136. doi:10.1007/s11606-015-3326-2
6. Prakash S. Gastrointestinal beriberi: a forme fruste of Wernicke’s encephalopathy? BMJ Case Rep. 2018;bcr2018224841. doi:10.1136/bcr-2018-224841
7. Friedenberg AS, Brandoff DE, Schiffman FJ. Type B lactic acidosis as a severe metabolic complication in lymphoma and leukemia: a case series from a single institution and literature review. Medicine (Baltimore). 2007;86(4):225-232. doi:10.1097/MD.0b013e318125759a
8. Liang CC. Bradycardia in thiamin deficiency and the role of glyoxylate. J Nutrition Sci Vitaminology. 1977;23(1):1-6. doi:10.3177/jnsv.23.1
9. Attaluri P, Castillo A, Edriss H, Nugent K. Thiamine deficiency: an important consideration in critically ill patients. Am J Med Sci. 2018;356(4):382-390. doi:10.1016/j.amjms.2018.06.015
10. Chaudhari A, Li ZY, Long A, Afshinnik A. Heavy cannabis use associated with Wernicke’s encephalopathy. Cureus. 2019;11(7):e5109. doi:10.7759/cureus.5109
11. Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013;368(2):149-160. doi:10.1056/NEJMcp1113996
12. Green R, Allen LH, Bjørke-Monsen A-L, et al. Vitamin B12 deficiency. Nat Rev Dis Primers. 2017;3(1):17040. doi:10.1038/nrdp.2017.40
13. Devalia V, Hamilton MS, Molloy AM. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
14. British Columbia Ministry of Health; Guidelines and Protocols and Advisory Committee. Guidelines and protocols cobalamin (vitamin B12) deficiency–investigation & management. Effective January 1, 2012. Revised May 1, 2013. Accessed March 10, 2021. https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/vitamin-b12
15. Galvin R, Brathen G, Ivashynka A, Hillbom M, Tanasescu R, Leone MA. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408-1418. doi:10.1111/j.1468-1331.2010.03153.x
16. Wiley KD, Gupta M. Vitamin B1 thiamine deficiency (beriberi). In: StatPearls. StatPearls Publishing LLC; 2019.
17. Jenco J, Krcmova LK, Solichova D, Solich P. Recent trends in determination of thiamine and its derivatives in clinical practice. J Chromatogra A. 2017;1510:1-12. doi:10.1016/j.chroma.2017.06.048
A 57-year-old woman with a history of traumatic brain injury, posttraumatic stress disorder, depression, migraines, hypothyroidism, and a hiatal hernia repair presented to the emergency department with a 1-day history of nausea, vomiting, and diffuse abdominal pain. She reported that her symptoms were relieved by hot showers. She also reported having similar symptoms and a previous gastric-emptying study that showed a slow-emptying stomach. Her history also consisted of frequent cannabis use for mood and appetite stimulation along with eliminating meat and fish from her diet, an increase in consumption of simple carbohydrates in the past year, and no alcohol use. Her medications included topiramate 100 mg and clonidine 0.3 mg nightly for migraines; levothyroxine 200 mcg daily for hypothyroidism; tizanidine 4 mg twice a day for muscle spasm; famotidine 40 mg twice a day as needed for gastric reflux; and bupropion 50 mg daily, citalopram 20 mg daily, and lamotrigine 25 mg nightly for mood.
The patient’s physical examination was notable for bradycardia (43 beats/min) and epigastric tenderness. Admission laboratory results were notable for an elevated lactic acid level of 4.8 (normal range, 0.50-2.20) mmol/L and a leukocytosis count of 10.8×109 cells/L. Serum alcohol level and blood cultures were negative. Liver function test, hemoglobin A1c, and lipase test were unremarkable. Her electrocardiogram showed an unchanged right bundle branch block. Chest X-ray, computed tomography (CT) of her abdomen/pelvis and echocardiogram were unremarkable.
What is your diagnosis?
How would you treat this patient?
This patient was diagnosed with gastrointestinal beriberi. Because of her dietary changes, lactic acidosis, and bradycardia, thiamine deficiency was suspected after ruling out other possibilities on the differential diagnosis (Table). The patient’s symptoms resolved after administration of high-dose IV thiamine 500 mg 3 times daily for 4 days. Her white blood cell count and lactic acid level normalized. Unfortunately, thiamine levels were not obtained for the patient before treatment was initiated. After administration of IV thiamine, her plasma thiamine level was > 1,200 (normal range, 8-30) nmol/L.
Her differential diagnosis included infectious etiology. Given her leukocytosis and lactic acidosis, vancomycin and piperacillin/tazobactam were started on admission. One day later, her leukocytosis count doubled to 20.7×109 cells/L. However, after 48 hours of negative blood cultures, antibiotics were discontinued.
Small bowel obstruction was suspected due to the patient’s history of abdominal surgery but was ruled out with CT imaging. Similarly, pancreatitis was ruled out based on negative CT imaging and the patient’s normal lipase level. Gastroparesis also was considered because of the patient’s history of hypothyroidism, tobacco use, and her prior gastric-emptying study. The patient was treated for gastroparesis with a course of metoclopramide and erythromycin without improvement in symptoms. Additionally, gastroparesis would not explain the patient’s leukocytosis.
Cannabinoid hyperemesis syndrome (CHS) was suspected because the patient’s symptoms improved with cannabis discontinuation and hot showers.1 In chronic users, however, tetrahydrocannabinol levels have a half-life of 5 to 13 days.2 Although lactic acidosis and leukocytosis have been previously reported with cannabis use, it is unlikely that the patient would have such significant improvement within the first 4 days after discontinuation.1,3,4 Although the patient had many psychiatric comorbidities with previous hospitalizations describing concern for somatization disorder, her leukocytosis and elevated lactic acid levels were suggestive of an organic rather than a psychiatric etiology of her symptoms.
Discussion
Gastrointestinal beriberi has been reported in chronic cannabis users who present with nausea, vomiting, epigastric pain, leukocytosis, and lactic acidosis; all these symptoms rapidly improve after thiamine administration.5,6 The patient’s dietary change also eliminated her intake of vitamin B12, which compounded her condition. Thiamine deficiency produces lactic acidosis by disrupting pyruvate metabolism.7 Bradycardia also can be a sign of thiamine deficiency, although the patient’s use of clonidine for migraines is a confounder.8
Chronically ill patients are prone to nutritional deficiencies, including deficiencies of thiamine.7,9 Many patients with chronic illnesses also use cannabis to ameliorate physical and neuropsychiatric symptoms.2 Recent reports suggest cannabis users are prone to gastrointestinal beriberi and Wernicke encephalopathy.5,10 Treating gastrointestinal symptoms in these patients can be challenging to diagnose because gastrointestinal beriberi and CHS share many clinical manifestations.
The patient’s presentation is likely multifactorial resulting from the combination of gastrointestinal beriberi and CHS. However, thiamine deficiency seems to play the dominant role.
There is no standard treatment regimen for thiamine deficiency with neurologic deficits, and patients only retain about 10 to 15% of intramuscular (IM) injections of cyanocobalamin.11,12 The British Committee for Standards in Haematology recommends IM injections of 1,000 mcg of cyanocobalamin 3 times a week for 2 weeks and then reassess the need for continued treatment.13 The British Columbia guidelines also recommend IM injections of 1,000 mcg daily for 1 to 5 days before transitioning to oral repletion.14 European Neurology guidelines for the treatment of Wernicke encephalopathy recommend IV cyanocobalamin 200 mg 3 times daily.15 Low-level evidence with observational studies informs these decisions and is why there is variation.
The patient’s serum lactate and leukocytosis normalized 1 day after the administration of thiamine. Thiamine deficiency classically causes Wernicke encephalopathy and wet beriberi.16 The patient did not present with Wernicke encephalopathy’s triad: ophthalmoplegia, ataxia, or confusion. She also was euvolemic without signs or symptoms of wet beriberi.
Conclusions
Thiamine deficiency is principally a clinical diagnosis. Thiamine laboratory testing may not be readily available in all medical centers, and confirming a diagnosis of thiamine deficiency should not delay treatment when thiamine deficiency is suspected. This patient’s thiamine levels resulted a week after collection. The administration of thiamine before sampling also can alter the result as it did in this case. Additionally, laboratories may offer whole blood and serum testing. Whole blood testing is more accurate because most bioactive thiamine is found in red blood cells.17
A 57-year-old woman with a history of traumatic brain injury, posttraumatic stress disorder, depression, migraines, hypothyroidism, and a hiatal hernia repair presented to the emergency department with a 1-day history of nausea, vomiting, and diffuse abdominal pain. She reported that her symptoms were relieved by hot showers. She also reported having similar symptoms and a previous gastric-emptying study that showed a slow-emptying stomach. Her history also consisted of frequent cannabis use for mood and appetite stimulation along with eliminating meat and fish from her diet, an increase in consumption of simple carbohydrates in the past year, and no alcohol use. Her medications included topiramate 100 mg and clonidine 0.3 mg nightly for migraines; levothyroxine 200 mcg daily for hypothyroidism; tizanidine 4 mg twice a day for muscle spasm; famotidine 40 mg twice a day as needed for gastric reflux; and bupropion 50 mg daily, citalopram 20 mg daily, and lamotrigine 25 mg nightly for mood.
The patient’s physical examination was notable for bradycardia (43 beats/min) and epigastric tenderness. Admission laboratory results were notable for an elevated lactic acid level of 4.8 (normal range, 0.50-2.20) mmol/L and a leukocytosis count of 10.8×109 cells/L. Serum alcohol level and blood cultures were negative. Liver function test, hemoglobin A1c, and lipase test were unremarkable. Her electrocardiogram showed an unchanged right bundle branch block. Chest X-ray, computed tomography (CT) of her abdomen/pelvis and echocardiogram were unremarkable.
What is your diagnosis?
How would you treat this patient?
This patient was diagnosed with gastrointestinal beriberi. Because of her dietary changes, lactic acidosis, and bradycardia, thiamine deficiency was suspected after ruling out other possibilities on the differential diagnosis (Table). The patient’s symptoms resolved after administration of high-dose IV thiamine 500 mg 3 times daily for 4 days. Her white blood cell count and lactic acid level normalized. Unfortunately, thiamine levels were not obtained for the patient before treatment was initiated. After administration of IV thiamine, her plasma thiamine level was > 1,200 (normal range, 8-30) nmol/L.
Her differential diagnosis included infectious etiology. Given her leukocytosis and lactic acidosis, vancomycin and piperacillin/tazobactam were started on admission. One day later, her leukocytosis count doubled to 20.7×109 cells/L. However, after 48 hours of negative blood cultures, antibiotics were discontinued.
Small bowel obstruction was suspected due to the patient’s history of abdominal surgery but was ruled out with CT imaging. Similarly, pancreatitis was ruled out based on negative CT imaging and the patient’s normal lipase level. Gastroparesis also was considered because of the patient’s history of hypothyroidism, tobacco use, and her prior gastric-emptying study. The patient was treated for gastroparesis with a course of metoclopramide and erythromycin without improvement in symptoms. Additionally, gastroparesis would not explain the patient’s leukocytosis.
Cannabinoid hyperemesis syndrome (CHS) was suspected because the patient’s symptoms improved with cannabis discontinuation and hot showers.1 In chronic users, however, tetrahydrocannabinol levels have a half-life of 5 to 13 days.2 Although lactic acidosis and leukocytosis have been previously reported with cannabis use, it is unlikely that the patient would have such significant improvement within the first 4 days after discontinuation.1,3,4 Although the patient had many psychiatric comorbidities with previous hospitalizations describing concern for somatization disorder, her leukocytosis and elevated lactic acid levels were suggestive of an organic rather than a psychiatric etiology of her symptoms.
Discussion
Gastrointestinal beriberi has been reported in chronic cannabis users who present with nausea, vomiting, epigastric pain, leukocytosis, and lactic acidosis; all these symptoms rapidly improve after thiamine administration.5,6 The patient’s dietary change also eliminated her intake of vitamin B12, which compounded her condition. Thiamine deficiency produces lactic acidosis by disrupting pyruvate metabolism.7 Bradycardia also can be a sign of thiamine deficiency, although the patient’s use of clonidine for migraines is a confounder.8
Chronically ill patients are prone to nutritional deficiencies, including deficiencies of thiamine.7,9 Many patients with chronic illnesses also use cannabis to ameliorate physical and neuropsychiatric symptoms.2 Recent reports suggest cannabis users are prone to gastrointestinal beriberi and Wernicke encephalopathy.5,10 Treating gastrointestinal symptoms in these patients can be challenging to diagnose because gastrointestinal beriberi and CHS share many clinical manifestations.
The patient’s presentation is likely multifactorial resulting from the combination of gastrointestinal beriberi and CHS. However, thiamine deficiency seems to play the dominant role.
There is no standard treatment regimen for thiamine deficiency with neurologic deficits, and patients only retain about 10 to 15% of intramuscular (IM) injections of cyanocobalamin.11,12 The British Committee for Standards in Haematology recommends IM injections of 1,000 mcg of cyanocobalamin 3 times a week for 2 weeks and then reassess the need for continued treatment.13 The British Columbia guidelines also recommend IM injections of 1,000 mcg daily for 1 to 5 days before transitioning to oral repletion.14 European Neurology guidelines for the treatment of Wernicke encephalopathy recommend IV cyanocobalamin 200 mg 3 times daily.15 Low-level evidence with observational studies informs these decisions and is why there is variation.
The patient’s serum lactate and leukocytosis normalized 1 day after the administration of thiamine. Thiamine deficiency classically causes Wernicke encephalopathy and wet beriberi.16 The patient did not present with Wernicke encephalopathy’s triad: ophthalmoplegia, ataxia, or confusion. She also was euvolemic without signs or symptoms of wet beriberi.
Conclusions
Thiamine deficiency is principally a clinical diagnosis. Thiamine laboratory testing may not be readily available in all medical centers, and confirming a diagnosis of thiamine deficiency should not delay treatment when thiamine deficiency is suspected. This patient’s thiamine levels resulted a week after collection. The administration of thiamine before sampling also can alter the result as it did in this case. Additionally, laboratories may offer whole blood and serum testing. Whole blood testing is more accurate because most bioactive thiamine is found in red blood cells.17
1. Price SL, Fisher C, Kumar R, Hilgerson A. Cannabinoid hyperemesis syndrome as the underlying cause of intractable nausea and vomiting. J Am Osteopath Assoc. 2011;111(3):166-169. doi:10.7556/jaoa.2011.111.3.166
2. Sharma P, Murthy P, Bharath MM. Chemistry, metabolism, and toxicology of cannabis: clinical implications. Iran J Psychiatry. 2012;7(4):149-156.
3. Antill T, Jakkoju A, Dieguez J, Laskhmiprasad L. Lactic acidosis: a rare manifestation of synthetic marijuana intoxication. J La State Med Soc. 2015;167(3):155.
4. Sullivan S. Cannabinoid hyperemesis. Can J Gastroenterol. 2010;24(5):284-285. doi:10.1155/2010/481940
5. Duca J, Lum CJ, Lo AM. Elevated lactate secondary to gastrointestinal beriberi. J Gen Intern Med. 2016;31(1):133-136. doi:10.1007/s11606-015-3326-2
6. Prakash S. Gastrointestinal beriberi: a forme fruste of Wernicke’s encephalopathy? BMJ Case Rep. 2018;bcr2018224841. doi:10.1136/bcr-2018-224841
7. Friedenberg AS, Brandoff DE, Schiffman FJ. Type B lactic acidosis as a severe metabolic complication in lymphoma and leukemia: a case series from a single institution and literature review. Medicine (Baltimore). 2007;86(4):225-232. doi:10.1097/MD.0b013e318125759a
8. Liang CC. Bradycardia in thiamin deficiency and the role of glyoxylate. J Nutrition Sci Vitaminology. 1977;23(1):1-6. doi:10.3177/jnsv.23.1
9. Attaluri P, Castillo A, Edriss H, Nugent K. Thiamine deficiency: an important consideration in critically ill patients. Am J Med Sci. 2018;356(4):382-390. doi:10.1016/j.amjms.2018.06.015
10. Chaudhari A, Li ZY, Long A, Afshinnik A. Heavy cannabis use associated with Wernicke’s encephalopathy. Cureus. 2019;11(7):e5109. doi:10.7759/cureus.5109
11. Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013;368(2):149-160. doi:10.1056/NEJMcp1113996
12. Green R, Allen LH, Bjørke-Monsen A-L, et al. Vitamin B12 deficiency. Nat Rev Dis Primers. 2017;3(1):17040. doi:10.1038/nrdp.2017.40
13. Devalia V, Hamilton MS, Molloy AM. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
14. British Columbia Ministry of Health; Guidelines and Protocols and Advisory Committee. Guidelines and protocols cobalamin (vitamin B12) deficiency–investigation & management. Effective January 1, 2012. Revised May 1, 2013. Accessed March 10, 2021. https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/vitamin-b12
15. Galvin R, Brathen G, Ivashynka A, Hillbom M, Tanasescu R, Leone MA. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408-1418. doi:10.1111/j.1468-1331.2010.03153.x
16. Wiley KD, Gupta M. Vitamin B1 thiamine deficiency (beriberi). In: StatPearls. StatPearls Publishing LLC; 2019.
17. Jenco J, Krcmova LK, Solichova D, Solich P. Recent trends in determination of thiamine and its derivatives in clinical practice. J Chromatogra A. 2017;1510:1-12. doi:10.1016/j.chroma.2017.06.048
1. Price SL, Fisher C, Kumar R, Hilgerson A. Cannabinoid hyperemesis syndrome as the underlying cause of intractable nausea and vomiting. J Am Osteopath Assoc. 2011;111(3):166-169. doi:10.7556/jaoa.2011.111.3.166
2. Sharma P, Murthy P, Bharath MM. Chemistry, metabolism, and toxicology of cannabis: clinical implications. Iran J Psychiatry. 2012;7(4):149-156.
3. Antill T, Jakkoju A, Dieguez J, Laskhmiprasad L. Lactic acidosis: a rare manifestation of synthetic marijuana intoxication. J La State Med Soc. 2015;167(3):155.
4. Sullivan S. Cannabinoid hyperemesis. Can J Gastroenterol. 2010;24(5):284-285. doi:10.1155/2010/481940
5. Duca J, Lum CJ, Lo AM. Elevated lactate secondary to gastrointestinal beriberi. J Gen Intern Med. 2016;31(1):133-136. doi:10.1007/s11606-015-3326-2
6. Prakash S. Gastrointestinal beriberi: a forme fruste of Wernicke’s encephalopathy? BMJ Case Rep. 2018;bcr2018224841. doi:10.1136/bcr-2018-224841
7. Friedenberg AS, Brandoff DE, Schiffman FJ. Type B lactic acidosis as a severe metabolic complication in lymphoma and leukemia: a case series from a single institution and literature review. Medicine (Baltimore). 2007;86(4):225-232. doi:10.1097/MD.0b013e318125759a
8. Liang CC. Bradycardia in thiamin deficiency and the role of glyoxylate. J Nutrition Sci Vitaminology. 1977;23(1):1-6. doi:10.3177/jnsv.23.1
9. Attaluri P, Castillo A, Edriss H, Nugent K. Thiamine deficiency: an important consideration in critically ill patients. Am J Med Sci. 2018;356(4):382-390. doi:10.1016/j.amjms.2018.06.015
10. Chaudhari A, Li ZY, Long A, Afshinnik A. Heavy cannabis use associated with Wernicke’s encephalopathy. Cureus. 2019;11(7):e5109. doi:10.7759/cureus.5109
11. Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013;368(2):149-160. doi:10.1056/NEJMcp1113996
12. Green R, Allen LH, Bjørke-Monsen A-L, et al. Vitamin B12 deficiency. Nat Rev Dis Primers. 2017;3(1):17040. doi:10.1038/nrdp.2017.40
13. Devalia V, Hamilton MS, Molloy AM. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
14. British Columbia Ministry of Health; Guidelines and Protocols and Advisory Committee. Guidelines and protocols cobalamin (vitamin B12) deficiency–investigation & management. Effective January 1, 2012. Revised May 1, 2013. Accessed March 10, 2021. https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/vitamin-b12
15. Galvin R, Brathen G, Ivashynka A, Hillbom M, Tanasescu R, Leone MA. EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol. 2010;17(12):1408-1418. doi:10.1111/j.1468-1331.2010.03153.x
16. Wiley KD, Gupta M. Vitamin B1 thiamine deficiency (beriberi). In: StatPearls. StatPearls Publishing LLC; 2019.
17. Jenco J, Krcmova LK, Solichova D, Solich P. Recent trends in determination of thiamine and its derivatives in clinical practice. J Chromatogra A. 2017;1510:1-12. doi:10.1016/j.chroma.2017.06.048
An international trip: Global experts weigh in on psychedelics
In 1967, when the United Nations Convention on Drugs classified psychedelics as schedule I substances, it effectively ended research into these agents as potential therapeutics for psychiatric disorders.
Psychedelics induce altered states of perception. They bind to the 5-hydroxytryptamine 2A (5-HT2A) receptor and include psilocybin, which is derived from “magic mushrooms”; N,N-dimethyltryptamine (DMT), a component of ayahuasca and mescaline (peyote cactus); and the synthesized compound D-lysergic acid diethylamide (LSD). Other agents, such as ketamine and 3,4-methylenedioxymethamphetamine (MDMA), also known as ecstasy, are sometimes considered psychedelics as well.
Before they were classified as schedule I agents, psychedelics had been shown to be particularly beneficial for patients with treatment-resistant conditions, including depression and posttraumatic stress disorder (PTSD), especially when administered in a supportive, therapeutic setting.
Now, after a hiatus of almost 50 years, there is renewed global interest in the scientific investigation of psychedelics. The attention was spurred in part by several exploratory studies of DMT in humans conducted in the 1990s by Rick Strassman, MD, and colleagues at the University of New Mexico, Albuquerque.
Around the same time, Franz X. Vollenweider, MD, and colleagues at the University of Zürich began researching psilocybin and its effects on human behavior. However, it was a 2006 study of psilocybin by a team of researchers at Johns Hopkins University, Baltimore, that is widely cited as a catalyst for the current renaissance in psychedelic research.
To provide a broad-based, international perspective on these agents, including their current legal status and indications, treatment regimens, safety, efficacy, and future considerations, this news organization interviewed nine expert researchers from around the globe.
Global legal status
In most, if not all, countries, it is still illegal to prescribe psychedelics in other than a research setting.
They can be used in research, but only with approval from the Food and Drug Administration under licensure from the Drug Enforcement Administration.
France lists all synthetic hallucinogens and hallucinogenic mushrooms as narcotic. As a result, possession, use, transportation, and collection are subject to criminal sanctions.
In France, NMDA antagonists such as ketamine and nitrous oxide are regarded as psychedelic molecules and can be used off label for various conditions or as part of research protocols authorized by the French public health code.
Although psychedelics are illegal under Mexican law, they are commonly used in indigenous communities as part of traditional rituals.
“The line between traditional consumption and psychedelic tourism is very thin,” José J. Mendoza Velásquez, MD, professor in the department of mental health, National Autonomous University of Mexico, Mexico City, said in an interview.
Psychedelics also are illegal in the United Kingdom, although government agencies have recently allowed research groups to investigate them. Psychedelics cannot be prescribed in Germany, Spain, or Italy. However, investigators in these countries can request permission from regulatory agencies to conduct research.
Brazil allows psychedelic substances to be researched, particularly ayahuasca, which has long traditional and religious roots in the country.
However, as in other countries, none of the classic psychedelics is regulated for therapeutic use in Brazil. It is widely expected that the Brazilian government will approve MDMA sometime in 2024 for use in the treatment of PTSD.
Potential indications
Psychedelics are currently under investigation as potential treatments for major depression, treatment-resistant depression, PTSD, pain management, and anorexia, among other conditions.
In France, Florian Ferreri, MD, PhD, at Hospital Saint-Antoine, Paris, is researching ketamine for treatment of patients with suicidal crisis/ideation and treatment-resistant depression.
In the United Kingdom, David Nutt, FMedSci, Edmond J. Safra Professor of Neuropsychopharmacology at Imperial College London, and his team have conducted studies of the use of psychedelics in conjunction with psychological support for patients with treatment-resistant depression, and they are currently exploring their use in the treatment of anorexia and various pain syndromes.
In Germany, Gerhard Gründer, MD, professor of psychiatry at the Central Institute of Mental Health, in Mannheim, noted that a study of psilocybin for treatment-resistant depression will launch sometime in 2021. In Italy, current research is focusing on MDMA and ketamine in the laboratory environment and in animal models for treating depression and drug abuse.
Researcher Helen Dolengevich-Segal, MD, a psychiatrist at Hospital Universitario del Henares, Madrid, noted that although research on esketamine for the treatment of severe depressive disorder with suicidal thoughts is underway, there is very limited published research from that country into the use of classic psychedelics for various psychiatric disorders, given their current illegal status.
Mexico’s Dr. Velásquez noted that although he is prohibited from prescribing psychedelics, he does have patients who take the drugs to augment medical treatment. For instance, he said, his patients frequently use psilocybin to help with severe depression, pain, and insomnia.
Environment is key
Most researchers agree that for psychedelics to be safe and effective, patient education and administration in a controlled environment by experienced clinicians are key to successful treatment.
Roland R. Griffiths, PhD, director of the Center for Psychedelic and Consciousness Research at Johns Hopkins, said that ongoing U.S. psilocybin research – primarily in major depressive disorder and psychological distress associated with life-threatening illness, drug addiction, anorexia nervosa, obsessive-compulsive disorder, and headache – generally includes one or two treatment sessions, each of which lasts 6-8 hours.
Such sessions typically involve oral administration of a moderately high dose of a psychedelic under what he characterizes as “psychologically supported conditions.”
For Dr. Griffiths, there are serious potential risks associated with the use of psilocybin and other psychedelics outside such environments.
“When taken in uncontrolled conditions, classic psychedelics can produce confusion and disorientation resulting in behavior dangerous to the participant and others, including life-threatening risk,” he said.
Dr. Gründer agreed.
“At the moment, I cannot imagine that you would go to the pharmacy with a prescription for psilocybin and get yourself a pill and then take it in a quiet little room,” he said. Dr. Dolengevich-Segal and Dr. Velásquez echoed these sentiments, noting the optimal location for administration is one that is quiet and secure and where patients feel safe.
Luís Fernando Tófoli, MD, PhD, professor of medical psychology and psychiatry at the University of Campinas, and Eduardo Schenberg, PhD, founder and CEO of Instituto Phaneros in São Paulo, Brazil, said more research is needed to determine the optimal therapeutic environment for individual agents.
“Most studies have a low number of participants (around 20 or 30), especially in neuroimaging, with high unblinding rates,” Dr. Schenberg said. “Therefore, novel methodological approaches are also necessary, as these substances do not easily fit into the traditional pharmacology epistemic model.”
Risks, abuse potential
The abuse potential of psychedelics is an ongoing concern for the public, researchers, and regulators, but the consensus among nearly all of these experts is that when administered by medical professionals in controlled settings, these drugs are associated with extremely low risk.
It is recreational use that presents an abuse concern, said Dr. Ferreri, but with the low doses used in psychiatry, the risk is “very limited or even nonexistent.”
Dr. Nutt said the abuse potential of psychedelics is so low that they can be used to treat addiction.
“Functionally, psychedelics are antiaddictive,” Dr. Nutt said. “The fact is, if you take them repeatedly, you develop tolerance, and the effect disappears. You can’t overcome it. But everyone believes they’re addictive because they’re scheduled drugs.”
Dr. Velásquez is something of an outlier. He believes the abuse potential with psychedelics is poorly understood and that some patients may develop tolerance, which is a potential gateway to dependence.
“Such is the case with LSD,” he said, “where this substance also favors tolerance to other psychedelic drugs such as psilocybin.”
Dosing also seems to play a key role in mitigating potential abuse, said Luca Pani, MD, professor of pharmacology and psychiatry at the University of Modena, Italy. Dr. Pani explained that with low doses and microdoses of psychedelics, the potential for abuse is eliminated.
Dr. Nutt, Dr. Pani, and Dr. Ferreri also noted the importance of medical supervision. For instance, said Dr. Ferreri, when administering ketamine, his team closely monitors both mental and physical parameters – heart rate and blood pressure, in particular – because the drug can have hypertensive effects.
Dr. Schenberg noted that ibogaine, a naturally occurring psychedelic frequently used by traditional communities in Africa in rituals and for healing purposes, could cause potentially fatal arrhythmias, so it’s critical that the treatment is administered in a hospital setting that has a cardiac unit.
Dr. Pani said there is a need for more research, especially regarding the molecular mechanisms behind the behavioral effects of low-dose psychedelic therapy and the potential risks of multiple treatments with the drugs.
“Although extensive toxicology has been conducted on a single active dose of psilocybin, which has been proven to be safe, further research is required to understand better the possible health risks, especially in relation to cardiac and lung tissue,” he said.
Psychologically challenging
The experts note that given the relative lack of experience with psychedelic therapy, preparing patients for potential adverse effects is paramount. This is particularly relevant in the research setting and highlights the need for adequate patient screening and aftercare.
Dr. Gründer and Dr. Dolengevich-Segal emphasized the importance of having qualified personnel available in the event that patients experience adverse psychological events during treatment.
For Dr. Gründer, the potential for psilocybin to cause patients to lose control, experience psychotic symptoms, or become paranoid warrants considerable preparation by treating physicians.
Patients occasionally experience fear and anxiety during treatment, though it’s usually short-lived, said Dr. Griffiths. Nevertheless, these experiences may open the door to greater insight. “A number of people report that these psychologically challenging states are a valuable part of the overall experience,” he said.
The situation is similar in Spain, where Dr. Dolengevich-Segal noted that typical treatment regimens have a strong focus on the patient’s experience as a therapeutic tool. As in the United Kingdom and the United States, her team guides patients to what they call a “peak experience,” which allows them to gain a better understanding of the trauma underlying their mental health problems.
Dr. Nutt said that in the United Kingdom, they haven’t seen adverse reactions in patients receiving psychedelic therapy, although sedatives such as benzodiazepines could be used to manage them. He added that at his center, two therapists are present at every treatment session, and all personnel are “trained medics or psychologists.”
Patient education
Preparing and educating patients about the therapy are critical, said Dr. Gründer, especially given the intense response psychedelic treatment often invokes.
Echoing Dr. Gründer, Dr. Tófoli said explaining the nature of psychedelic treatment to potential patients helps ease anxiety.
Dr. Griffiths noted that in the United States, study participants are not only educated about the potential effects of psychedelic agents but also undergo several hours of psychological preparation in advance of their first treatment session and are provided with psychological support after treatment.
There is also a strong emphasis on patient preparation and education in the United Kingdom, where patients meet with therapists before and after treatment. During these posttreatment debriefings, clinicians use the patients’ experience with psychedelics to help them gain insight into the underlying cause of their depression.
Dr. Schenberg noted that at his institution in São Paulo, there are online courses to teach clinicians about psychedelic therapy for psychiatric disorders. Next year, he added, a new training program in MDMA-assisted psychotherapy will begin.
Working out treatment protocols
Treatment protocols for psychedelics vary by agent and indication from country to country. For instance, Dr. Pani noted that current psychedelic research in Italy predominantly focuses more on microdosing, which involves administering 1% of the pharmacologically active dose to a maximum of 100 mcg, in contrast to low dosing or full dosing.
Therapeutic regimens in Brazil, said Dr. Schenberg, also differ by agent but share common elements. For instance, psychedelics are always administered in a research setting, and sessions include concomitant psychotherapy.
In Germany, investigators are working to determine optimal treatment regimen for psilocybin for resistant depression in a randomized three-arm study planned for 2021.
For Mexico’s Dr. Velásquez, treatment regimens are complex and varied. Either way, he said, patients always require long-term follow-up.
With ketamine therapy, Dr. Ferreri said his team administers the drug in 45- to 60-minute intravenous infusion sessions in a hospital room without light or sound stimulation. Regardless of the drug’s immediate effect, he said, the protocol is repeated within a 6-month period.
The question of the duration of treatment effect is important. Dr. Griffiths said research suggests that the positive effects of psilocybin are long lasting and that most individuals report positive changes in mood, attitude, and behavior that endure for months or even years after the session.
“Our research has shown that the benefits of these experiences can last as long as 14 months, often longer, and that many participants characterize their psilocybin experience as among the most profound and personally meaningful experiences of their lives,” said Dr. Griffiths.
Dr. Nutt agreed, noting that a single intense “trip” can improve mood for weeks, months, or even years. Nevertheless, he said, in his experience, approximately three-quarters of patients treated with psychedelics for major depression relapse within 3-9 months.
“Most get better,” he said, “but the majority of depression comes back over a period of months.”
Given the current illegal status of the drugs, he said it’s nearly impossible to provide patients with regular, subsequent treatment with psychedelics over time.
“My suspicion is that you might well have to dose four or five times over a couple of years to get people to escape from very severe depression,” said Dr. Nutt. “The longer they’ve been depressed, the harder it is for them to make a full recovery, because it’s more entrenched in the brain.”
All experts agree that exciting times are ahead for psychedelics as therapeutics for a wide range of psychiatric disorders.
“We can look forward to continued growth and expansion of this research,” said Dr. Griffiths, “including the refinement of protocols for a variety of therapeutic indications and to the development of a variety of new classic psychedelic compounds.”
A version of this article first appeared on Medscape.com.
In 1967, when the United Nations Convention on Drugs classified psychedelics as schedule I substances, it effectively ended research into these agents as potential therapeutics for psychiatric disorders.
Psychedelics induce altered states of perception. They bind to the 5-hydroxytryptamine 2A (5-HT2A) receptor and include psilocybin, which is derived from “magic mushrooms”; N,N-dimethyltryptamine (DMT), a component of ayahuasca and mescaline (peyote cactus); and the synthesized compound D-lysergic acid diethylamide (LSD). Other agents, such as ketamine and 3,4-methylenedioxymethamphetamine (MDMA), also known as ecstasy, are sometimes considered psychedelics as well.
Before they were classified as schedule I agents, psychedelics had been shown to be particularly beneficial for patients with treatment-resistant conditions, including depression and posttraumatic stress disorder (PTSD), especially when administered in a supportive, therapeutic setting.
Now, after a hiatus of almost 50 years, there is renewed global interest in the scientific investigation of psychedelics. The attention was spurred in part by several exploratory studies of DMT in humans conducted in the 1990s by Rick Strassman, MD, and colleagues at the University of New Mexico, Albuquerque.
Around the same time, Franz X. Vollenweider, MD, and colleagues at the University of Zürich began researching psilocybin and its effects on human behavior. However, it was a 2006 study of psilocybin by a team of researchers at Johns Hopkins University, Baltimore, that is widely cited as a catalyst for the current renaissance in psychedelic research.
To provide a broad-based, international perspective on these agents, including their current legal status and indications, treatment regimens, safety, efficacy, and future considerations, this news organization interviewed nine expert researchers from around the globe.
Global legal status
In most, if not all, countries, it is still illegal to prescribe psychedelics in other than a research setting.
They can be used in research, but only with approval from the Food and Drug Administration under licensure from the Drug Enforcement Administration.
France lists all synthetic hallucinogens and hallucinogenic mushrooms as narcotic. As a result, possession, use, transportation, and collection are subject to criminal sanctions.
In France, NMDA antagonists such as ketamine and nitrous oxide are regarded as psychedelic molecules and can be used off label for various conditions or as part of research protocols authorized by the French public health code.
Although psychedelics are illegal under Mexican law, they are commonly used in indigenous communities as part of traditional rituals.
“The line between traditional consumption and psychedelic tourism is very thin,” José J. Mendoza Velásquez, MD, professor in the department of mental health, National Autonomous University of Mexico, Mexico City, said in an interview.
Psychedelics also are illegal in the United Kingdom, although government agencies have recently allowed research groups to investigate them. Psychedelics cannot be prescribed in Germany, Spain, or Italy. However, investigators in these countries can request permission from regulatory agencies to conduct research.
Brazil allows psychedelic substances to be researched, particularly ayahuasca, which has long traditional and religious roots in the country.
However, as in other countries, none of the classic psychedelics is regulated for therapeutic use in Brazil. It is widely expected that the Brazilian government will approve MDMA sometime in 2024 for use in the treatment of PTSD.
Potential indications
Psychedelics are currently under investigation as potential treatments for major depression, treatment-resistant depression, PTSD, pain management, and anorexia, among other conditions.
In France, Florian Ferreri, MD, PhD, at Hospital Saint-Antoine, Paris, is researching ketamine for treatment of patients with suicidal crisis/ideation and treatment-resistant depression.
In the United Kingdom, David Nutt, FMedSci, Edmond J. Safra Professor of Neuropsychopharmacology at Imperial College London, and his team have conducted studies of the use of psychedelics in conjunction with psychological support for patients with treatment-resistant depression, and they are currently exploring their use in the treatment of anorexia and various pain syndromes.
In Germany, Gerhard Gründer, MD, professor of psychiatry at the Central Institute of Mental Health, in Mannheim, noted that a study of psilocybin for treatment-resistant depression will launch sometime in 2021. In Italy, current research is focusing on MDMA and ketamine in the laboratory environment and in animal models for treating depression and drug abuse.
Researcher Helen Dolengevich-Segal, MD, a psychiatrist at Hospital Universitario del Henares, Madrid, noted that although research on esketamine for the treatment of severe depressive disorder with suicidal thoughts is underway, there is very limited published research from that country into the use of classic psychedelics for various psychiatric disorders, given their current illegal status.
Mexico’s Dr. Velásquez noted that although he is prohibited from prescribing psychedelics, he does have patients who take the drugs to augment medical treatment. For instance, he said, his patients frequently use psilocybin to help with severe depression, pain, and insomnia.
Environment is key
Most researchers agree that for psychedelics to be safe and effective, patient education and administration in a controlled environment by experienced clinicians are key to successful treatment.
Roland R. Griffiths, PhD, director of the Center for Psychedelic and Consciousness Research at Johns Hopkins, said that ongoing U.S. psilocybin research – primarily in major depressive disorder and psychological distress associated with life-threatening illness, drug addiction, anorexia nervosa, obsessive-compulsive disorder, and headache – generally includes one or two treatment sessions, each of which lasts 6-8 hours.
Such sessions typically involve oral administration of a moderately high dose of a psychedelic under what he characterizes as “psychologically supported conditions.”
For Dr. Griffiths, there are serious potential risks associated with the use of psilocybin and other psychedelics outside such environments.
“When taken in uncontrolled conditions, classic psychedelics can produce confusion and disorientation resulting in behavior dangerous to the participant and others, including life-threatening risk,” he said.
Dr. Gründer agreed.
“At the moment, I cannot imagine that you would go to the pharmacy with a prescription for psilocybin and get yourself a pill and then take it in a quiet little room,” he said. Dr. Dolengevich-Segal and Dr. Velásquez echoed these sentiments, noting the optimal location for administration is one that is quiet and secure and where patients feel safe.
Luís Fernando Tófoli, MD, PhD, professor of medical psychology and psychiatry at the University of Campinas, and Eduardo Schenberg, PhD, founder and CEO of Instituto Phaneros in São Paulo, Brazil, said more research is needed to determine the optimal therapeutic environment for individual agents.
“Most studies have a low number of participants (around 20 or 30), especially in neuroimaging, with high unblinding rates,” Dr. Schenberg said. “Therefore, novel methodological approaches are also necessary, as these substances do not easily fit into the traditional pharmacology epistemic model.”
Risks, abuse potential
The abuse potential of psychedelics is an ongoing concern for the public, researchers, and regulators, but the consensus among nearly all of these experts is that when administered by medical professionals in controlled settings, these drugs are associated with extremely low risk.
It is recreational use that presents an abuse concern, said Dr. Ferreri, but with the low doses used in psychiatry, the risk is “very limited or even nonexistent.”
Dr. Nutt said the abuse potential of psychedelics is so low that they can be used to treat addiction.
“Functionally, psychedelics are antiaddictive,” Dr. Nutt said. “The fact is, if you take them repeatedly, you develop tolerance, and the effect disappears. You can’t overcome it. But everyone believes they’re addictive because they’re scheduled drugs.”
Dr. Velásquez is something of an outlier. He believes the abuse potential with psychedelics is poorly understood and that some patients may develop tolerance, which is a potential gateway to dependence.
“Such is the case with LSD,” he said, “where this substance also favors tolerance to other psychedelic drugs such as psilocybin.”
Dosing also seems to play a key role in mitigating potential abuse, said Luca Pani, MD, professor of pharmacology and psychiatry at the University of Modena, Italy. Dr. Pani explained that with low doses and microdoses of psychedelics, the potential for abuse is eliminated.
Dr. Nutt, Dr. Pani, and Dr. Ferreri also noted the importance of medical supervision. For instance, said Dr. Ferreri, when administering ketamine, his team closely monitors both mental and physical parameters – heart rate and blood pressure, in particular – because the drug can have hypertensive effects.
Dr. Schenberg noted that ibogaine, a naturally occurring psychedelic frequently used by traditional communities in Africa in rituals and for healing purposes, could cause potentially fatal arrhythmias, so it’s critical that the treatment is administered in a hospital setting that has a cardiac unit.
Dr. Pani said there is a need for more research, especially regarding the molecular mechanisms behind the behavioral effects of low-dose psychedelic therapy and the potential risks of multiple treatments with the drugs.
“Although extensive toxicology has been conducted on a single active dose of psilocybin, which has been proven to be safe, further research is required to understand better the possible health risks, especially in relation to cardiac and lung tissue,” he said.
Psychologically challenging
The experts note that given the relative lack of experience with psychedelic therapy, preparing patients for potential adverse effects is paramount. This is particularly relevant in the research setting and highlights the need for adequate patient screening and aftercare.
Dr. Gründer and Dr. Dolengevich-Segal emphasized the importance of having qualified personnel available in the event that patients experience adverse psychological events during treatment.
For Dr. Gründer, the potential for psilocybin to cause patients to lose control, experience psychotic symptoms, or become paranoid warrants considerable preparation by treating physicians.
Patients occasionally experience fear and anxiety during treatment, though it’s usually short-lived, said Dr. Griffiths. Nevertheless, these experiences may open the door to greater insight. “A number of people report that these psychologically challenging states are a valuable part of the overall experience,” he said.
The situation is similar in Spain, where Dr. Dolengevich-Segal noted that typical treatment regimens have a strong focus on the patient’s experience as a therapeutic tool. As in the United Kingdom and the United States, her team guides patients to what they call a “peak experience,” which allows them to gain a better understanding of the trauma underlying their mental health problems.
Dr. Nutt said that in the United Kingdom, they haven’t seen adverse reactions in patients receiving psychedelic therapy, although sedatives such as benzodiazepines could be used to manage them. He added that at his center, two therapists are present at every treatment session, and all personnel are “trained medics or psychologists.”
Patient education
Preparing and educating patients about the therapy are critical, said Dr. Gründer, especially given the intense response psychedelic treatment often invokes.
Echoing Dr. Gründer, Dr. Tófoli said explaining the nature of psychedelic treatment to potential patients helps ease anxiety.
Dr. Griffiths noted that in the United States, study participants are not only educated about the potential effects of psychedelic agents but also undergo several hours of psychological preparation in advance of their first treatment session and are provided with psychological support after treatment.
There is also a strong emphasis on patient preparation and education in the United Kingdom, where patients meet with therapists before and after treatment. During these posttreatment debriefings, clinicians use the patients’ experience with psychedelics to help them gain insight into the underlying cause of their depression.
Dr. Schenberg noted that at his institution in São Paulo, there are online courses to teach clinicians about psychedelic therapy for psychiatric disorders. Next year, he added, a new training program in MDMA-assisted psychotherapy will begin.
Working out treatment protocols
Treatment protocols for psychedelics vary by agent and indication from country to country. For instance, Dr. Pani noted that current psychedelic research in Italy predominantly focuses more on microdosing, which involves administering 1% of the pharmacologically active dose to a maximum of 100 mcg, in contrast to low dosing or full dosing.
Therapeutic regimens in Brazil, said Dr. Schenberg, also differ by agent but share common elements. For instance, psychedelics are always administered in a research setting, and sessions include concomitant psychotherapy.
In Germany, investigators are working to determine optimal treatment regimen for psilocybin for resistant depression in a randomized three-arm study planned for 2021.
For Mexico’s Dr. Velásquez, treatment regimens are complex and varied. Either way, he said, patients always require long-term follow-up.
With ketamine therapy, Dr. Ferreri said his team administers the drug in 45- to 60-minute intravenous infusion sessions in a hospital room without light or sound stimulation. Regardless of the drug’s immediate effect, he said, the protocol is repeated within a 6-month period.
The question of the duration of treatment effect is important. Dr. Griffiths said research suggests that the positive effects of psilocybin are long lasting and that most individuals report positive changes in mood, attitude, and behavior that endure for months or even years after the session.
“Our research has shown that the benefits of these experiences can last as long as 14 months, often longer, and that many participants characterize their psilocybin experience as among the most profound and personally meaningful experiences of their lives,” said Dr. Griffiths.
Dr. Nutt agreed, noting that a single intense “trip” can improve mood for weeks, months, or even years. Nevertheless, he said, in his experience, approximately three-quarters of patients treated with psychedelics for major depression relapse within 3-9 months.
“Most get better,” he said, “but the majority of depression comes back over a period of months.”
Given the current illegal status of the drugs, he said it’s nearly impossible to provide patients with regular, subsequent treatment with psychedelics over time.
“My suspicion is that you might well have to dose four or five times over a couple of years to get people to escape from very severe depression,” said Dr. Nutt. “The longer they’ve been depressed, the harder it is for them to make a full recovery, because it’s more entrenched in the brain.”
All experts agree that exciting times are ahead for psychedelics as therapeutics for a wide range of psychiatric disorders.
“We can look forward to continued growth and expansion of this research,” said Dr. Griffiths, “including the refinement of protocols for a variety of therapeutic indications and to the development of a variety of new classic psychedelic compounds.”
A version of this article first appeared on Medscape.com.
In 1967, when the United Nations Convention on Drugs classified psychedelics as schedule I substances, it effectively ended research into these agents as potential therapeutics for psychiatric disorders.
Psychedelics induce altered states of perception. They bind to the 5-hydroxytryptamine 2A (5-HT2A) receptor and include psilocybin, which is derived from “magic mushrooms”; N,N-dimethyltryptamine (DMT), a component of ayahuasca and mescaline (peyote cactus); and the synthesized compound D-lysergic acid diethylamide (LSD). Other agents, such as ketamine and 3,4-methylenedioxymethamphetamine (MDMA), also known as ecstasy, are sometimes considered psychedelics as well.
Before they were classified as schedule I agents, psychedelics had been shown to be particularly beneficial for patients with treatment-resistant conditions, including depression and posttraumatic stress disorder (PTSD), especially when administered in a supportive, therapeutic setting.
Now, after a hiatus of almost 50 years, there is renewed global interest in the scientific investigation of psychedelics. The attention was spurred in part by several exploratory studies of DMT in humans conducted in the 1990s by Rick Strassman, MD, and colleagues at the University of New Mexico, Albuquerque.
Around the same time, Franz X. Vollenweider, MD, and colleagues at the University of Zürich began researching psilocybin and its effects on human behavior. However, it was a 2006 study of psilocybin by a team of researchers at Johns Hopkins University, Baltimore, that is widely cited as a catalyst for the current renaissance in psychedelic research.
To provide a broad-based, international perspective on these agents, including their current legal status and indications, treatment regimens, safety, efficacy, and future considerations, this news organization interviewed nine expert researchers from around the globe.
Global legal status
In most, if not all, countries, it is still illegal to prescribe psychedelics in other than a research setting.
They can be used in research, but only with approval from the Food and Drug Administration under licensure from the Drug Enforcement Administration.
France lists all synthetic hallucinogens and hallucinogenic mushrooms as narcotic. As a result, possession, use, transportation, and collection are subject to criminal sanctions.
In France, NMDA antagonists such as ketamine and nitrous oxide are regarded as psychedelic molecules and can be used off label for various conditions or as part of research protocols authorized by the French public health code.
Although psychedelics are illegal under Mexican law, they are commonly used in indigenous communities as part of traditional rituals.
“The line between traditional consumption and psychedelic tourism is very thin,” José J. Mendoza Velásquez, MD, professor in the department of mental health, National Autonomous University of Mexico, Mexico City, said in an interview.
Psychedelics also are illegal in the United Kingdom, although government agencies have recently allowed research groups to investigate them. Psychedelics cannot be prescribed in Germany, Spain, or Italy. However, investigators in these countries can request permission from regulatory agencies to conduct research.
Brazil allows psychedelic substances to be researched, particularly ayahuasca, which has long traditional and religious roots in the country.
However, as in other countries, none of the classic psychedelics is regulated for therapeutic use in Brazil. It is widely expected that the Brazilian government will approve MDMA sometime in 2024 for use in the treatment of PTSD.
Potential indications
Psychedelics are currently under investigation as potential treatments for major depression, treatment-resistant depression, PTSD, pain management, and anorexia, among other conditions.
In France, Florian Ferreri, MD, PhD, at Hospital Saint-Antoine, Paris, is researching ketamine for treatment of patients with suicidal crisis/ideation and treatment-resistant depression.
In the United Kingdom, David Nutt, FMedSci, Edmond J. Safra Professor of Neuropsychopharmacology at Imperial College London, and his team have conducted studies of the use of psychedelics in conjunction with psychological support for patients with treatment-resistant depression, and they are currently exploring their use in the treatment of anorexia and various pain syndromes.
In Germany, Gerhard Gründer, MD, professor of psychiatry at the Central Institute of Mental Health, in Mannheim, noted that a study of psilocybin for treatment-resistant depression will launch sometime in 2021. In Italy, current research is focusing on MDMA and ketamine in the laboratory environment and in animal models for treating depression and drug abuse.
Researcher Helen Dolengevich-Segal, MD, a psychiatrist at Hospital Universitario del Henares, Madrid, noted that although research on esketamine for the treatment of severe depressive disorder with suicidal thoughts is underway, there is very limited published research from that country into the use of classic psychedelics for various psychiatric disorders, given their current illegal status.
Mexico’s Dr. Velásquez noted that although he is prohibited from prescribing psychedelics, he does have patients who take the drugs to augment medical treatment. For instance, he said, his patients frequently use psilocybin to help with severe depression, pain, and insomnia.
Environment is key
Most researchers agree that for psychedelics to be safe and effective, patient education and administration in a controlled environment by experienced clinicians are key to successful treatment.
Roland R. Griffiths, PhD, director of the Center for Psychedelic and Consciousness Research at Johns Hopkins, said that ongoing U.S. psilocybin research – primarily in major depressive disorder and psychological distress associated with life-threatening illness, drug addiction, anorexia nervosa, obsessive-compulsive disorder, and headache – generally includes one or two treatment sessions, each of which lasts 6-8 hours.
Such sessions typically involve oral administration of a moderately high dose of a psychedelic under what he characterizes as “psychologically supported conditions.”
For Dr. Griffiths, there are serious potential risks associated with the use of psilocybin and other psychedelics outside such environments.
“When taken in uncontrolled conditions, classic psychedelics can produce confusion and disorientation resulting in behavior dangerous to the participant and others, including life-threatening risk,” he said.
Dr. Gründer agreed.
“At the moment, I cannot imagine that you would go to the pharmacy with a prescription for psilocybin and get yourself a pill and then take it in a quiet little room,” he said. Dr. Dolengevich-Segal and Dr. Velásquez echoed these sentiments, noting the optimal location for administration is one that is quiet and secure and where patients feel safe.
Luís Fernando Tófoli, MD, PhD, professor of medical psychology and psychiatry at the University of Campinas, and Eduardo Schenberg, PhD, founder and CEO of Instituto Phaneros in São Paulo, Brazil, said more research is needed to determine the optimal therapeutic environment for individual agents.
“Most studies have a low number of participants (around 20 or 30), especially in neuroimaging, with high unblinding rates,” Dr. Schenberg said. “Therefore, novel methodological approaches are also necessary, as these substances do not easily fit into the traditional pharmacology epistemic model.”
Risks, abuse potential
The abuse potential of psychedelics is an ongoing concern for the public, researchers, and regulators, but the consensus among nearly all of these experts is that when administered by medical professionals in controlled settings, these drugs are associated with extremely low risk.
It is recreational use that presents an abuse concern, said Dr. Ferreri, but with the low doses used in psychiatry, the risk is “very limited or even nonexistent.”
Dr. Nutt said the abuse potential of psychedelics is so low that they can be used to treat addiction.
“Functionally, psychedelics are antiaddictive,” Dr. Nutt said. “The fact is, if you take them repeatedly, you develop tolerance, and the effect disappears. You can’t overcome it. But everyone believes they’re addictive because they’re scheduled drugs.”
Dr. Velásquez is something of an outlier. He believes the abuse potential with psychedelics is poorly understood and that some patients may develop tolerance, which is a potential gateway to dependence.
“Such is the case with LSD,” he said, “where this substance also favors tolerance to other psychedelic drugs such as psilocybin.”
Dosing also seems to play a key role in mitigating potential abuse, said Luca Pani, MD, professor of pharmacology and psychiatry at the University of Modena, Italy. Dr. Pani explained that with low doses and microdoses of psychedelics, the potential for abuse is eliminated.
Dr. Nutt, Dr. Pani, and Dr. Ferreri also noted the importance of medical supervision. For instance, said Dr. Ferreri, when administering ketamine, his team closely monitors both mental and physical parameters – heart rate and blood pressure, in particular – because the drug can have hypertensive effects.
Dr. Schenberg noted that ibogaine, a naturally occurring psychedelic frequently used by traditional communities in Africa in rituals and for healing purposes, could cause potentially fatal arrhythmias, so it’s critical that the treatment is administered in a hospital setting that has a cardiac unit.
Dr. Pani said there is a need for more research, especially regarding the molecular mechanisms behind the behavioral effects of low-dose psychedelic therapy and the potential risks of multiple treatments with the drugs.
“Although extensive toxicology has been conducted on a single active dose of psilocybin, which has been proven to be safe, further research is required to understand better the possible health risks, especially in relation to cardiac and lung tissue,” he said.
Psychologically challenging
The experts note that given the relative lack of experience with psychedelic therapy, preparing patients for potential adverse effects is paramount. This is particularly relevant in the research setting and highlights the need for adequate patient screening and aftercare.
Dr. Gründer and Dr. Dolengevich-Segal emphasized the importance of having qualified personnel available in the event that patients experience adverse psychological events during treatment.
For Dr. Gründer, the potential for psilocybin to cause patients to lose control, experience psychotic symptoms, or become paranoid warrants considerable preparation by treating physicians.
Patients occasionally experience fear and anxiety during treatment, though it’s usually short-lived, said Dr. Griffiths. Nevertheless, these experiences may open the door to greater insight. “A number of people report that these psychologically challenging states are a valuable part of the overall experience,” he said.
The situation is similar in Spain, where Dr. Dolengevich-Segal noted that typical treatment regimens have a strong focus on the patient’s experience as a therapeutic tool. As in the United Kingdom and the United States, her team guides patients to what they call a “peak experience,” which allows them to gain a better understanding of the trauma underlying their mental health problems.
Dr. Nutt said that in the United Kingdom, they haven’t seen adverse reactions in patients receiving psychedelic therapy, although sedatives such as benzodiazepines could be used to manage them. He added that at his center, two therapists are present at every treatment session, and all personnel are “trained medics or psychologists.”
Patient education
Preparing and educating patients about the therapy are critical, said Dr. Gründer, especially given the intense response psychedelic treatment often invokes.
Echoing Dr. Gründer, Dr. Tófoli said explaining the nature of psychedelic treatment to potential patients helps ease anxiety.
Dr. Griffiths noted that in the United States, study participants are not only educated about the potential effects of psychedelic agents but also undergo several hours of psychological preparation in advance of their first treatment session and are provided with psychological support after treatment.
There is also a strong emphasis on patient preparation and education in the United Kingdom, where patients meet with therapists before and after treatment. During these posttreatment debriefings, clinicians use the patients’ experience with psychedelics to help them gain insight into the underlying cause of their depression.
Dr. Schenberg noted that at his institution in São Paulo, there are online courses to teach clinicians about psychedelic therapy for psychiatric disorders. Next year, he added, a new training program in MDMA-assisted psychotherapy will begin.
Working out treatment protocols
Treatment protocols for psychedelics vary by agent and indication from country to country. For instance, Dr. Pani noted that current psychedelic research in Italy predominantly focuses more on microdosing, which involves administering 1% of the pharmacologically active dose to a maximum of 100 mcg, in contrast to low dosing or full dosing.
Therapeutic regimens in Brazil, said Dr. Schenberg, also differ by agent but share common elements. For instance, psychedelics are always administered in a research setting, and sessions include concomitant psychotherapy.
In Germany, investigators are working to determine optimal treatment regimen for psilocybin for resistant depression in a randomized three-arm study planned for 2021.
For Mexico’s Dr. Velásquez, treatment regimens are complex and varied. Either way, he said, patients always require long-term follow-up.
With ketamine therapy, Dr. Ferreri said his team administers the drug in 45- to 60-minute intravenous infusion sessions in a hospital room without light or sound stimulation. Regardless of the drug’s immediate effect, he said, the protocol is repeated within a 6-month period.
The question of the duration of treatment effect is important. Dr. Griffiths said research suggests that the positive effects of psilocybin are long lasting and that most individuals report positive changes in mood, attitude, and behavior that endure for months or even years after the session.
“Our research has shown that the benefits of these experiences can last as long as 14 months, often longer, and that many participants characterize their psilocybin experience as among the most profound and personally meaningful experiences of their lives,” said Dr. Griffiths.
Dr. Nutt agreed, noting that a single intense “trip” can improve mood for weeks, months, or even years. Nevertheless, he said, in his experience, approximately three-quarters of patients treated with psychedelics for major depression relapse within 3-9 months.
“Most get better,” he said, “but the majority of depression comes back over a period of months.”
Given the current illegal status of the drugs, he said it’s nearly impossible to provide patients with regular, subsequent treatment with psychedelics over time.
“My suspicion is that you might well have to dose four or five times over a couple of years to get people to escape from very severe depression,” said Dr. Nutt. “The longer they’ve been depressed, the harder it is for them to make a full recovery, because it’s more entrenched in the brain.”
All experts agree that exciting times are ahead for psychedelics as therapeutics for a wide range of psychiatric disorders.
“We can look forward to continued growth and expansion of this research,” said Dr. Griffiths, “including the refinement of protocols for a variety of therapeutic indications and to the development of a variety of new classic psychedelic compounds.”
A version of this article first appeared on Medscape.com.
Borderline personality disorder diagnosis: To tell or not to tell patients?
News of actor/comedian Pete Davidson expressing relief after finally receiving a diagnosis of borderline personality disorder (BPD) prompted a recent Twitter discussion among physicians regarding the ongoing debate on whether or not to tell a patient he or she has this diagnosis.
“I’ve heard from [many] trainees that they were told never to tell a patient they had BPD, but I can hardly think of anything more paternalistic and stigmatizing,” Amy Barnhorst, MD, vice chair of community psychiatry at University of California, Davis, tweeted.
“Most patients, when I explain it to them, have this kind of reaction – they feel relieved and understood,” she added.
“I was told that as well [not to tell] in one of my practicum placements,” one respondent who identified herself as a clinical/forensic psychologist tweeted back. “I said it anyway and the person was relieved there was a name for what they were living with.”
However, others disagreed with Dr. Barnhorst, noting that BPD is a very serious, stigmatizing, and challenging disorder to treat and, because of this, may cause patients to lose hope.
Still, Dr. Barnhorst stands by her position. Although “there is a negative stigma against a diagnosis of BPD,” that idea more often comes from the clinician instead of the patient, she said.
“I’ve never had a patient say, ‘how dare you call me that!’ like it was an insult,” she said in an interview. Not disclosing a diagnosis “is like you’re not trusting a patient to be a reasonable adult human about this.”
‘Hard diagnosis’
Although BPD is a “hard diagnosis, we would never withhold a diagnosis of cancer or liver disease or something else we knew patients didn’t want but that we were going to try and treat them for,” said Dr. Barnhorst.
BPD is linked to significant morbidity because of its common association with comorbid conditions, such as major depressive disorder, substance use disorders, and dysthymia. A history of self-harm is present in 70%-75% of these patients and some estimates suggest up to 9% of individuals with BPD die by suicide.
In an article published in Innovations in Clinical Neuroscience investigators discussed “ethical and clinical questions psychiatrists should consider” when treating BPD, including whether a diagnosis should be shared with a patient.
After such a diagnosis a patient may “react intensely in negative ways and these responses may be easily triggered,” the researchers wrote.
“A propensity that will likely cause psychiatrists anguish, however, is BPD patients’ increased likelihood of attempting suicide,” they added. Part of the problem has been that, in the past, it was thought that a BPD prognosis was untreatable. However, the researchers note that is no longer the case.
Still, Kaz Nelson, MD, associate professor of psychiatry and behavioral sciences at the University of Minnesota, Minneapolis, has labeled BPD a so-called “asterisk” disorder.
As she wrote in a recent blog, “We tell patients when they meet criteria for a medical diagnosis.* We show compassion and nonjudgmentalism to patients.* We do not discriminate against patients.*” However, the asterisk for each of these statements is: *Except for those with BPD.
Ongoing debate
Starting around the 1980s, the DSM listed personality disorders under the No. 2 Axis, which is for conditions with symptoms that are “not mitigatable,” said Dr. Nelson.
“It really started as well-meaning therapists who care about their patients who wanted to develop some precision in understanding people, and them starting to notice some patterns that can get in the way of optimal function,” she said in an interview.
The thought was not to disclose these diagnoses “because that was for you to understand, and for the patient to discover these patterns over time in the course of your work together,” Dr. Nelson added.
Although treatment for BPD used to be virtually nonexistent, there is now hope – especially with dialectic-behavior therapy (DBT), which uses mindfulness to teach patients how to control emotions and improve relationships.
According to the National Education Alliance for BPD, other useful treatments include mentalization-based therapy, transference-focused therapy, and “good psychiatric management.” Although there are currently no approved medications for BPD, some drugs are used to treat comorbid conditions such as depression or anxiety.
“We now know that people recover, and the whole paradigm has been turned on its head,” Dr. Nelson said. For example, “we no longer categorize these things as treatable or untreatable, which was a very positive move.”
So why is the field still debating the issue of diagnosis disclosure?
“To this day there are different psychiatrists and some medical school curricula that continue to teach that personality disorders are long-term, fixed, and nontreatable – and that it’s kind of disparaging to give this kind of diagnosis to a patient,” Dr. Nelson said.
Dr. Nelson, also the vice chair for education at the University of Minnesota, Minneapolis, medical school, reported that there “we acknowledge BPD’s painful history and that there are these misconceptions. They’re going to be on the front line of combating discrimination and the idea that if you see a patient with possible BPD coming you should run. That’s just unacceptable.”
Dr. Nelson noted that the idea of disclosing a BPD diagnosis is less controversial now than in the past, but “the whole thing is still under debate, and treatment guidelines [on BPD] are old and expired.”
Criteria for BPD were not updated when the DSM-5 was published in 2013, and that needs to be fixed, Dr. Nelson added. “In the meantime, we’re trying to get the word out that it’s okay to interact with people about the diagnosis, discuss treatment plans, and manage it as one would with any other psychiatric or medical illness.”
An evolution, not a debate
Paul Appelbaum, MD, past president of the American Psychiatric Association and current chair of the organization’s DSM steering committee, said in an interview that he hasn’t been involved in any recent debate on this issue.
“I think practice has changed to the point where the general practice is to discuss patient diagnoses with [patients] openly. Patients appreciate that and psychiatrists have come to see the advantages of it,” said Dr. Appelbaum, a professor of psychiatry, medicine, and law at Columbia University, New York.
Dr. Appelbaum noted that patients also increasingly have access to their medical records, “so the reality is that it’s no longer possible in many cases to withhold a diagnosis.”
he said. “Maybe not everyone is entirely on board yet but there has been a sea change in psychiatric practices.”
Asked whether there needs to be some type of guideline update or statement released by the APA regarding BPD, Dr. Appelbaum said he doesn’t think the overall issue is BPD specific but applies to all psychiatric diagnoses.
“To the extent that there are still practitioners today that are telling students or residents [not to disclose], I would guess that they were trained a very long time ago and have not adapted to the new world,” he said.
“I don’t want to speak for the APA, but speaking for myself: I certainly encourage residents that I teach to be open about a diagnosis. It’s not just clinically helpful in some cases, it’s also ethically required from the perspective of allowing patients to make appropriate decisions about their treatment. And arguably it’s legally required as well, as part of the informed consent requirement,” Dr. Appelbaum said.
Regarding DSM updates, he noted that the committee “looks to the field to propose to us additions or changes to the DSM that are warranted by data that have been gathered since the DSM-5 came out.” There is a process set up on the DSM’s website to review such proposals.
In addition, Dr. Appelbaum said that there have been discussions about using a new model “that focuses on dimensions rather than on discreet categories” in order to classify personality disorders.
“There’s a group out there that is formulating a proposal that they will submit to us” on this, he added. “That’s the major discussion that is going on right now and it would clearly have implications for borderline as well as all the other personality disorders.”
In a statement, the APA said practice guidelines for BPD are currently under review and that the organization does not have a “position statement” on BPD for clinicians. The last update to its guideline was in the early 2000s.
A version of this article first appeared on Medscape.com.
News of actor/comedian Pete Davidson expressing relief after finally receiving a diagnosis of borderline personality disorder (BPD) prompted a recent Twitter discussion among physicians regarding the ongoing debate on whether or not to tell a patient he or she has this diagnosis.
“I’ve heard from [many] trainees that they were told never to tell a patient they had BPD, but I can hardly think of anything more paternalistic and stigmatizing,” Amy Barnhorst, MD, vice chair of community psychiatry at University of California, Davis, tweeted.
“Most patients, when I explain it to them, have this kind of reaction – they feel relieved and understood,” she added.
“I was told that as well [not to tell] in one of my practicum placements,” one respondent who identified herself as a clinical/forensic psychologist tweeted back. “I said it anyway and the person was relieved there was a name for what they were living with.”
However, others disagreed with Dr. Barnhorst, noting that BPD is a very serious, stigmatizing, and challenging disorder to treat and, because of this, may cause patients to lose hope.
Still, Dr. Barnhorst stands by her position. Although “there is a negative stigma against a diagnosis of BPD,” that idea more often comes from the clinician instead of the patient, she said.
“I’ve never had a patient say, ‘how dare you call me that!’ like it was an insult,” she said in an interview. Not disclosing a diagnosis “is like you’re not trusting a patient to be a reasonable adult human about this.”
‘Hard diagnosis’
Although BPD is a “hard diagnosis, we would never withhold a diagnosis of cancer or liver disease or something else we knew patients didn’t want but that we were going to try and treat them for,” said Dr. Barnhorst.
BPD is linked to significant morbidity because of its common association with comorbid conditions, such as major depressive disorder, substance use disorders, and dysthymia. A history of self-harm is present in 70%-75% of these patients and some estimates suggest up to 9% of individuals with BPD die by suicide.
In an article published in Innovations in Clinical Neuroscience investigators discussed “ethical and clinical questions psychiatrists should consider” when treating BPD, including whether a diagnosis should be shared with a patient.
After such a diagnosis a patient may “react intensely in negative ways and these responses may be easily triggered,” the researchers wrote.
“A propensity that will likely cause psychiatrists anguish, however, is BPD patients’ increased likelihood of attempting suicide,” they added. Part of the problem has been that, in the past, it was thought that a BPD prognosis was untreatable. However, the researchers note that is no longer the case.
Still, Kaz Nelson, MD, associate professor of psychiatry and behavioral sciences at the University of Minnesota, Minneapolis, has labeled BPD a so-called “asterisk” disorder.
As she wrote in a recent blog, “We tell patients when they meet criteria for a medical diagnosis.* We show compassion and nonjudgmentalism to patients.* We do not discriminate against patients.*” However, the asterisk for each of these statements is: *Except for those with BPD.
Ongoing debate
Starting around the 1980s, the DSM listed personality disorders under the No. 2 Axis, which is for conditions with symptoms that are “not mitigatable,” said Dr. Nelson.
“It really started as well-meaning therapists who care about their patients who wanted to develop some precision in understanding people, and them starting to notice some patterns that can get in the way of optimal function,” she said in an interview.
The thought was not to disclose these diagnoses “because that was for you to understand, and for the patient to discover these patterns over time in the course of your work together,” Dr. Nelson added.
Although treatment for BPD used to be virtually nonexistent, there is now hope – especially with dialectic-behavior therapy (DBT), which uses mindfulness to teach patients how to control emotions and improve relationships.
According to the National Education Alliance for BPD, other useful treatments include mentalization-based therapy, transference-focused therapy, and “good psychiatric management.” Although there are currently no approved medications for BPD, some drugs are used to treat comorbid conditions such as depression or anxiety.
“We now know that people recover, and the whole paradigm has been turned on its head,” Dr. Nelson said. For example, “we no longer categorize these things as treatable or untreatable, which was a very positive move.”
So why is the field still debating the issue of diagnosis disclosure?
“To this day there are different psychiatrists and some medical school curricula that continue to teach that personality disorders are long-term, fixed, and nontreatable – and that it’s kind of disparaging to give this kind of diagnosis to a patient,” Dr. Nelson said.
Dr. Nelson, also the vice chair for education at the University of Minnesota, Minneapolis, medical school, reported that there “we acknowledge BPD’s painful history and that there are these misconceptions. They’re going to be on the front line of combating discrimination and the idea that if you see a patient with possible BPD coming you should run. That’s just unacceptable.”
Dr. Nelson noted that the idea of disclosing a BPD diagnosis is less controversial now than in the past, but “the whole thing is still under debate, and treatment guidelines [on BPD] are old and expired.”
Criteria for BPD were not updated when the DSM-5 was published in 2013, and that needs to be fixed, Dr. Nelson added. “In the meantime, we’re trying to get the word out that it’s okay to interact with people about the diagnosis, discuss treatment plans, and manage it as one would with any other psychiatric or medical illness.”
An evolution, not a debate
Paul Appelbaum, MD, past president of the American Psychiatric Association and current chair of the organization’s DSM steering committee, said in an interview that he hasn’t been involved in any recent debate on this issue.
“I think practice has changed to the point where the general practice is to discuss patient diagnoses with [patients] openly. Patients appreciate that and psychiatrists have come to see the advantages of it,” said Dr. Appelbaum, a professor of psychiatry, medicine, and law at Columbia University, New York.
Dr. Appelbaum noted that patients also increasingly have access to their medical records, “so the reality is that it’s no longer possible in many cases to withhold a diagnosis.”
he said. “Maybe not everyone is entirely on board yet but there has been a sea change in psychiatric practices.”
Asked whether there needs to be some type of guideline update or statement released by the APA regarding BPD, Dr. Appelbaum said he doesn’t think the overall issue is BPD specific but applies to all psychiatric diagnoses.
“To the extent that there are still practitioners today that are telling students or residents [not to disclose], I would guess that they were trained a very long time ago and have not adapted to the new world,” he said.
“I don’t want to speak for the APA, but speaking for myself: I certainly encourage residents that I teach to be open about a diagnosis. It’s not just clinically helpful in some cases, it’s also ethically required from the perspective of allowing patients to make appropriate decisions about their treatment. And arguably it’s legally required as well, as part of the informed consent requirement,” Dr. Appelbaum said.
Regarding DSM updates, he noted that the committee “looks to the field to propose to us additions or changes to the DSM that are warranted by data that have been gathered since the DSM-5 came out.” There is a process set up on the DSM’s website to review such proposals.
In addition, Dr. Appelbaum said that there have been discussions about using a new model “that focuses on dimensions rather than on discreet categories” in order to classify personality disorders.
“There’s a group out there that is formulating a proposal that they will submit to us” on this, he added. “That’s the major discussion that is going on right now and it would clearly have implications for borderline as well as all the other personality disorders.”
In a statement, the APA said practice guidelines for BPD are currently under review and that the organization does not have a “position statement” on BPD for clinicians. The last update to its guideline was in the early 2000s.
A version of this article first appeared on Medscape.com.
News of actor/comedian Pete Davidson expressing relief after finally receiving a diagnosis of borderline personality disorder (BPD) prompted a recent Twitter discussion among physicians regarding the ongoing debate on whether or not to tell a patient he or she has this diagnosis.
“I’ve heard from [many] trainees that they were told never to tell a patient they had BPD, but I can hardly think of anything more paternalistic and stigmatizing,” Amy Barnhorst, MD, vice chair of community psychiatry at University of California, Davis, tweeted.
“Most patients, when I explain it to them, have this kind of reaction – they feel relieved and understood,” she added.
“I was told that as well [not to tell] in one of my practicum placements,” one respondent who identified herself as a clinical/forensic psychologist tweeted back. “I said it anyway and the person was relieved there was a name for what they were living with.”
However, others disagreed with Dr. Barnhorst, noting that BPD is a very serious, stigmatizing, and challenging disorder to treat and, because of this, may cause patients to lose hope.
Still, Dr. Barnhorst stands by her position. Although “there is a negative stigma against a diagnosis of BPD,” that idea more often comes from the clinician instead of the patient, she said.
“I’ve never had a patient say, ‘how dare you call me that!’ like it was an insult,” she said in an interview. Not disclosing a diagnosis “is like you’re not trusting a patient to be a reasonable adult human about this.”
‘Hard diagnosis’
Although BPD is a “hard diagnosis, we would never withhold a diagnosis of cancer or liver disease or something else we knew patients didn’t want but that we were going to try and treat them for,” said Dr. Barnhorst.
BPD is linked to significant morbidity because of its common association with comorbid conditions, such as major depressive disorder, substance use disorders, and dysthymia. A history of self-harm is present in 70%-75% of these patients and some estimates suggest up to 9% of individuals with BPD die by suicide.
In an article published in Innovations in Clinical Neuroscience investigators discussed “ethical and clinical questions psychiatrists should consider” when treating BPD, including whether a diagnosis should be shared with a patient.
After such a diagnosis a patient may “react intensely in negative ways and these responses may be easily triggered,” the researchers wrote.
“A propensity that will likely cause psychiatrists anguish, however, is BPD patients’ increased likelihood of attempting suicide,” they added. Part of the problem has been that, in the past, it was thought that a BPD prognosis was untreatable. However, the researchers note that is no longer the case.
Still, Kaz Nelson, MD, associate professor of psychiatry and behavioral sciences at the University of Minnesota, Minneapolis, has labeled BPD a so-called “asterisk” disorder.
As she wrote in a recent blog, “We tell patients when they meet criteria for a medical diagnosis.* We show compassion and nonjudgmentalism to patients.* We do not discriminate against patients.*” However, the asterisk for each of these statements is: *Except for those with BPD.
Ongoing debate
Starting around the 1980s, the DSM listed personality disorders under the No. 2 Axis, which is for conditions with symptoms that are “not mitigatable,” said Dr. Nelson.
“It really started as well-meaning therapists who care about their patients who wanted to develop some precision in understanding people, and them starting to notice some patterns that can get in the way of optimal function,” she said in an interview.
The thought was not to disclose these diagnoses “because that was for you to understand, and for the patient to discover these patterns over time in the course of your work together,” Dr. Nelson added.
Although treatment for BPD used to be virtually nonexistent, there is now hope – especially with dialectic-behavior therapy (DBT), which uses mindfulness to teach patients how to control emotions and improve relationships.
According to the National Education Alliance for BPD, other useful treatments include mentalization-based therapy, transference-focused therapy, and “good psychiatric management.” Although there are currently no approved medications for BPD, some drugs are used to treat comorbid conditions such as depression or anxiety.
“We now know that people recover, and the whole paradigm has been turned on its head,” Dr. Nelson said. For example, “we no longer categorize these things as treatable or untreatable, which was a very positive move.”
So why is the field still debating the issue of diagnosis disclosure?
“To this day there are different psychiatrists and some medical school curricula that continue to teach that personality disorders are long-term, fixed, and nontreatable – and that it’s kind of disparaging to give this kind of diagnosis to a patient,” Dr. Nelson said.
Dr. Nelson, also the vice chair for education at the University of Minnesota, Minneapolis, medical school, reported that there “we acknowledge BPD’s painful history and that there are these misconceptions. They’re going to be on the front line of combating discrimination and the idea that if you see a patient with possible BPD coming you should run. That’s just unacceptable.”
Dr. Nelson noted that the idea of disclosing a BPD diagnosis is less controversial now than in the past, but “the whole thing is still under debate, and treatment guidelines [on BPD] are old and expired.”
Criteria for BPD were not updated when the DSM-5 was published in 2013, and that needs to be fixed, Dr. Nelson added. “In the meantime, we’re trying to get the word out that it’s okay to interact with people about the diagnosis, discuss treatment plans, and manage it as one would with any other psychiatric or medical illness.”
An evolution, not a debate
Paul Appelbaum, MD, past president of the American Psychiatric Association and current chair of the organization’s DSM steering committee, said in an interview that he hasn’t been involved in any recent debate on this issue.
“I think practice has changed to the point where the general practice is to discuss patient diagnoses with [patients] openly. Patients appreciate that and psychiatrists have come to see the advantages of it,” said Dr. Appelbaum, a professor of psychiatry, medicine, and law at Columbia University, New York.
Dr. Appelbaum noted that patients also increasingly have access to their medical records, “so the reality is that it’s no longer possible in many cases to withhold a diagnosis.”
he said. “Maybe not everyone is entirely on board yet but there has been a sea change in psychiatric practices.”
Asked whether there needs to be some type of guideline update or statement released by the APA regarding BPD, Dr. Appelbaum said he doesn’t think the overall issue is BPD specific but applies to all psychiatric diagnoses.
“To the extent that there are still practitioners today that are telling students or residents [not to disclose], I would guess that they were trained a very long time ago and have not adapted to the new world,” he said.
“I don’t want to speak for the APA, but speaking for myself: I certainly encourage residents that I teach to be open about a diagnosis. It’s not just clinically helpful in some cases, it’s also ethically required from the perspective of allowing patients to make appropriate decisions about their treatment. And arguably it’s legally required as well, as part of the informed consent requirement,” Dr. Appelbaum said.
Regarding DSM updates, he noted that the committee “looks to the field to propose to us additions or changes to the DSM that are warranted by data that have been gathered since the DSM-5 came out.” There is a process set up on the DSM’s website to review such proposals.
In addition, Dr. Appelbaum said that there have been discussions about using a new model “that focuses on dimensions rather than on discreet categories” in order to classify personality disorders.
“There’s a group out there that is formulating a proposal that they will submit to us” on this, he added. “That’s the major discussion that is going on right now and it would clearly have implications for borderline as well as all the other personality disorders.”
In a statement, the APA said practice guidelines for BPD are currently under review and that the organization does not have a “position statement” on BPD for clinicians. The last update to its guideline was in the early 2000s.
A version of this article first appeared on Medscape.com.
Antidepressants, TMS, and the risk of affective switch in bipolar depression
Because treatment resistance is a pervasive problem in bipolar depression, the use of neuromodulation treatments such as transcranial magnetic stimulation (TMS) is increasing for patients with this disorder.1-7 Patients with bipolar disorder tend to spend the majority of the time with depressive symptoms, which underscores the importance of providing effective treatment for bipolar depression, especially given the chronicity of this disease.2,3,5 Only a few medications are FDA-approved for treating bipolar depression (Table).
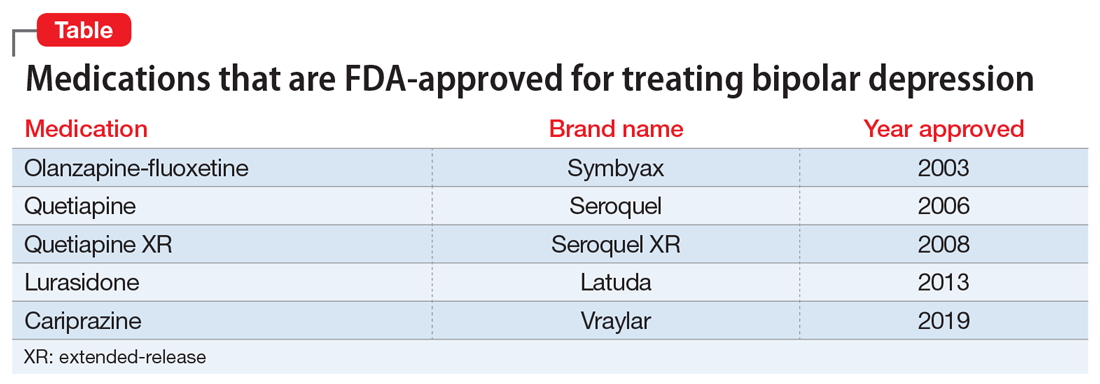
In this article, we describe the case of a patient with treatment-resistant bipolar depression undergoing adjunctive TMS treatment who experienced an affective switch from depression to mania. We also discuss evidence regarding the likelihood of treatment-emergent mania for antidepressants vs TMS in bipolar depression.
CASE
Ms. W, a 60-year-old White female with a history of bipolar I disorder and attention-deficit/hyperactivity disorder (ADHD), presented for TMS evaluation during a depressive episode. Throughout her life, she had experienced numerous manic episodes, but as she got older she noted an increasing frequency of depressive episodes. Over the course of her illness, she had completed adequate trials at therapeutic doses of many medications, including second-generation antipsychotics (SGAs) (aripiprazole, lurasidone, olanzapine, quetiapine), mood stabilizers (lamotrigine, lithium), and antidepressants (bupropion, venlafaxine, fluoxetine, mirtazapine, trazodone). A course of electroconvulsive therapy was not effective. Ms. W had a long-standing diagnosis of ADHD and had been treated with stimulants for >10 years, although it was unclear whether formal neuropsychological testing had been conducted to confirm this diagnosis. She had >10 suicide attempts and multiple psychiatric hospitalizations.
At her initial evaluation for TMS, Ms. W said she had depressive symptoms predominating for the past 2 years, including low mood, hopelessness, poor sleep, poor appetite, anhedonia, and suicidal ideation without a plan. At the time, she was taking clonazepam, 0.5 mg twice a day; lurasidone, 40 mg/d at bedtime; fluoxetine, 60 mg/d; trazodone, 50 mg/d at bedtime; and methylphenidate, 40 mg/d, and was participating in psychotherapy consistently.
After Ms. W and her clinicians discussed alternatives, risks, benefits, and adverse effects, she consented to adjunctive TMS treatment and provided written informed consent. The treatment plan was outlined as 6 weeks of daily TMS therapy (NeuroStar; Neuronetics, Malvern, PA), 1 treatment per day, 5 days a week. Her clinical status was assessed weekly using the Quick Inventory of Depressive Symptomatology (QIDS) for depression, Generalized Anxiety Disorder 7-item scale (GAD-7) for anxiety, and Young Mania Rating Scale (YMRS) for mania. The Figure shows the trends in Ms. W’s QIDS, GAD-7, and YMRS scores over the course of TMS treatment.
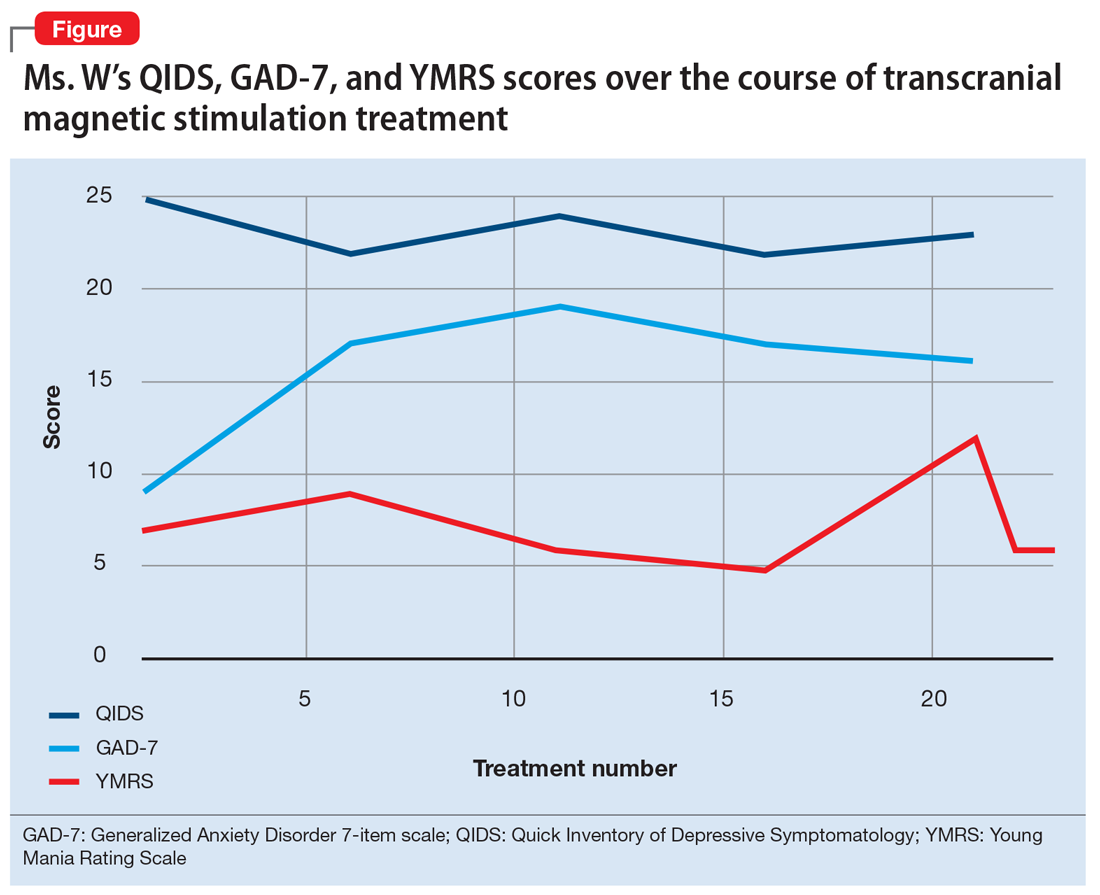
Prior to initiating TMS, her baseline scores were QIDS: 25, GAD-7: 9, and YMRS: 7, indicating very severe depression, mild anxiety, and the absence of mania. Ms. W’s psychotropic regimen remained unchanged throughout the course of her TMS treatment. After her motor threshold was determined, her TMS treatment began at 80% of motor threshold and was titrated up to 95% at the first treatment. By the second treatment, it was titrated up to 110%. By the third treatment, it was titrated up to 120% of motor threshold, which is the percentage used for the remaining treatments.
Initially, Ms. W reported some improvement in her depression, but this improvement was short-lived, and she continued to have elevated QIDS scores throughout treatment. By treatment #21, her QIDS and GAD-7 scores remained elevated, and her YMRS score had increased to 12. Due to this increase in YMRS score, the YMRS was repeated on the next 2 treatment days (#22 and #23), and her score was 6 on both days. When Ms. W presented for treatment #25, she was disorganized, irritable, and endorsed racing thoughts and decreased sleep. She was involuntarily hospitalized for mania, and TMS was discontinued. Unfortunately, she did not complete any clinical scales on that day. Upon admission to the hospital, Ms. W reported that at approximately the time of treatment #21, she had a fluctuation in her mood that consisted of increased goal-directed activity, decreased need for sleep, racing thoughts, and increased frivolous spending. She was treated with lithium, 300 mg twice a day. Lurasidone was increased to 80 mg/d at bedtime, and she continued clonazepam, trazodone, and methylphenidate at the previous doses. Over 14 days, Ms. W’s mania gradually resolved, and she was discharged home.
Continue to: Mixed evidence on the risk of switching
Mixed evidence on the risk of switching
Currently, several TMS devices are FDA-cleared for treating unipolar major depressive disorder, obsessive-compulsive disorder, and certain types of migraine. In March 2020, the FDA granted Breakthrough Device Designation for one TMS device, the NeuroStar Advanced Therapy System, for the treatment of bipolar depression.8 This designation created an expedited pathway for prioritized FDA review of the NeuroStar Advanced Therapy clinical trial program.
Few published clinical studies have evaluated using TMS to treat patients with bipolar depression.9-15 As with any antidepressant treatment for bipolar depression, there is a risk of affective switch from depression to mania when using TMS. Most of the literature available regarding the treatment of bipolar depression focuses on the risk of antidepressant medications to induce an affective switch. This risk depends on the class of the antidepressant,16 and there is a paucity of studies examining the risk of switch with TMS.
Interpretation of available literature is limited due to inconsistencies in the definition of an affective switch, the variable length of treatment with antidepressants, the use of concurrent medications such as mood stabilizers, and confounders such as the natural course of switching in bipolar disorder.17 Overall, the evidence for treatment-emergent mania related to antidepressant use is mixed, and the reported rate of treatment-emergent mania varies. In a systematic review and meta-analysis of >20 randomized controlled trials that included 1,316 patients with bipolar disorder who received antidepressants, Fornaro et al18 found that the incidence of treatment-emergent mania was 11.8%. It is generally recommended that if antidepressants are used to treat patients with bipolar disorder, they should be given with a traditional mood stabilizer to prevent affective switches, although whether mood stabilizers can prevent such switches is unproven.19
In a literature review by Xia et al,20 the affective switch rate in patients with bipolar depression who were treated with TMS was 3.1%, which was not statistically different from the affective switch rate with sham treatment.However, most of the patients included in this analysis were receiving other medications concurrently, and the length of treatment was 2 weeks, which is shorter than the average length of TMS treatment in clinical practice. In a recent literature review by Rachid,21 TMS was found to possibly induce manic episodes when used as monotherapy or in combination with antidepressants in patients with bipolar depression. To reduce the risk of treatment-emergent mania, current recommendations advise the use of a mood stabilizer for a minimum of 2 weeks before initiating TMS.1
In our case, Ms. W was receiving antidepressants (fluoxetine and trazodone), lurasidone (an SGA that is FDA-approved for bipolar depression), and methylphenidate before starting TMS treatment. Fluoxetine, trazodone, and methylphenidate may possibly contribute to an increased risk of an affective switch.1,22 Further studies are needed to clarify whether mood stabilizers or SGAs can prevent the development of mania in patients with bipolar depression who undergo TMS treatment.20
Continue to: Because bipolar depression poses...
Because bipolar depression poses a major clinical challenge,23,24 it is imperative to consider alternate treatments. When evaluating alternative treatment strategies, one may consider TMS in conjunction with a traditional mood stabilizer because this regimen may have a lower risk of treatment-emergent mania compared with antidepressants.1,25
Acknowledgment
The authors thank Dr. Sy Saeed for his expertise and guidance on this article.
Bottom Line
For patients with bipolar depression, treatment with transcranial magnetic stimulation in conjunction with a mood stabilizer may have lower rates of treatment-emergent mania than treatment with antidepressants.
Related Resources
- Transcranial magnetic stimulation: clinical applications for psychiatric practice. Bermudes RA, Lanocha K, Janicak PG, eds. American Psychiatric Association Publishing; 2017.
- Gold AK, Ornelas AC, Cirillo P, et al. Clinical applications of transcranial magnetic stimulation in bipolar disorder. Brain Behav. 2019;9(10):e01419. doi: 10.1002/brb3.1419
Drug Brand Names
Aripiprazole • Abilify
Bupropion • Wellbutrin
Cariprazine • Vraylar
Clonazepam • Klonopin
Fluoxetine • Prozac
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Methylphenidate • Ritalin, Concerta
Mirtazapine • Remeron
Olanzapine • Zyprexa
Olanzapine-fluoxetine • Symbyax
Quetiapine • Seroquel
Trazodone • Desyrel
Venlafaxine • Effexor
1. Aaronson ST, Croarkin PE. Transcranial magnetic stimulation for the treatment of other mood disorders. In: Bermudes RA, Lanocha K, Janicak PG, eds. Transcranial magnetic stimulation: clinical applications for psychiatric practice. American Psychiatric Association Publishing; 2017:127-156.
2. Geddes JR, Miklowitz DJ. Treatment of bipolar disorder. Lancet. 2013;381(9878):1672-1682.
3. Gitlin M. Treatment-resistant bipolar disorder. Molecular Psychiatry. 2006;11(3):227-240.
4. Harrison PJ, Geddes JR, Tunbridge EM. The emerging neurobiology of bipolar disorder. Trends Neurosci. 2018;41(1):18-30.
5. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the World Mental Health Survey Initiative. Arch Gen Psychiatry. 2011;68(3):241-251.
6. Myczkowski ML, Fernandes A, Moreno M, et al. Cognitive outcomes of TMS treatment in bipolar depression: safety data from a randomized controlled trial. J Affect Disord. 2018;235: 20-26.
7. Tavares DF, Myczkowski ML, Alberto RL, et al. Treatment of bipolar depression with deep TMS: results from a double-blind, randomized, parallel group, sham-controlled clinical trial. Neuropsychopharmacology. 2017;42(13):2593-2601.
8. Neuronetics. FDA grants NeuroStar® Advanced Therapy System Breakthrough Device Designation to treat bipolar depression. Accessed February 2, 2021. https://www.globenewswire.com/news-release/2020/03/06/1996447/0/en/FDA-Grants-NeuroStar-Advanced-Therapy-System-Breakthrough-Device-Designation-to-Treat-Bipolar-Depression.html
9. Cohen RB, Brunoni AR, Boggio PS, et al. Clinical predictors associated with duration of repetitive transcranial magnetic stimulation treatment for remission in bipolar depression: a naturalistic study. J Nerv Ment Dis. 2010;198(9):679-681.
10. Connolly KR, Helmer A, Cristancho MA, et al. Effectiveness of transcranial magnetic stimulation in clinical practice post-FDA approval in the United States: results observed with the first 100 consecutive cases of depression at an academic medical center. J Clin Psychiatry. 2012;73(4):e567-e573.
11. Dell’osso B, D’Urso N, Castellano F, et al. Long-term efficacy after acute augmentative repetitive transcranial magnetic stimulation in bipolar depression: a 1-year follow-up study. J ECT. 2011;27(2):141-144.
12. Dell’Osso B, Mundo E, D’Urso N, et al. Augmentative repetitive navigated transcranial magnetic stimulation (rTMS) in drug-resistant bipolar depression. Bipolar Disord. 2009;11(1):76-81.
13. Harel EV, Zangen A, Roth Y, et al. H-coil repetitive transcranial magnetic stimulation for the treatment of bipolar depression: an add-on, safety and feasibility study. World J Biol Psychiatry. 2011;12(2):119-126.
14. Nahas Z, Kozel FA, Li X, et al. Left prefrontal transcranial magnetic stimulation (TMS) treatment of depression in bipolar affective disorder: a pilot study of acute safety and efficacy. Bipolar Disord. 2003;5(1):40-47.
15. Tamas RL, Menkes D, El-Mallakh RS. Stimulating research: a prospective, randomized, double-blind, sham-controlled study of slow transcranial magnetic stimulation in depressed bipolar patients. J Neuropsychiatry Clin Neurosci. 2007;19(2):198-199.
16. Tundo A, Cavalieri P, Navari S, et al. Treating bipolar depression - antidepressants and alternatives: a critical review of the literature. Acta Neuropsychiatrica. 2011:23(3):94-105.
17. Gijsman HJ, Geddes JR, Rendell JM, et al. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry. 2004;161(9):1537-1547.
18. Fornaro M, Anastasia A, Novello S, et al. Incidence, prevalence and clinical correlates of antidepressant‐emergent mania in bipolar depression: a systematic review and meta‐analysis. Bipolar Disord. 2018;20(3):195-227.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society for Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262.
20. Xia G, Gajwani P, Muzina DJ, et al. Treatment-emergent mania in unipolar and bipolar depression: focus on repetitive transcranial magnetic stimulation. Int J Neuropsychopharmacol. 2008;11(1):119-130.
21. Rachid F. Repetitive transcranial magnetic stimulation and treatment-emergent mania and hypomania: a review of the literature. J Psychiatr Pract. 2017;23(2):150-159.
22. Victorin A, Rydén E, Thase M, et al. The risk of treatment-emergent mania with methylphenidate in bipolar disorder. Am J Psychiatry. 2017;174(4):341-348.
23. Hidalgo-Mazzei D, Berk M, Cipriani A, et al. Treatment-resistant and multi-therapy-resistant criteria for bipolar depression: consensus definition. Br J Psychiatry. 2019;214(1):27-35.
24. Baldessarini RJ, Vázquez GH, Tondo L. Bipolar depression: a major unsolved challenge. Int J Bipolar Disord. 2020;8(1):1.
25. Phillips AL, Burr RL, Dunner DL. Repetitive transcranial magnetic stimulation in the treatment of bipolar depression: Experience from a clinical setting. J Psychiatr Pract. 2020;26(1):37-45.
Because treatment resistance is a pervasive problem in bipolar depression, the use of neuromodulation treatments such as transcranial magnetic stimulation (TMS) is increasing for patients with this disorder.1-7 Patients with bipolar disorder tend to spend the majority of the time with depressive symptoms, which underscores the importance of providing effective treatment for bipolar depression, especially given the chronicity of this disease.2,3,5 Only a few medications are FDA-approved for treating bipolar depression (Table).

In this article, we describe the case of a patient with treatment-resistant bipolar depression undergoing adjunctive TMS treatment who experienced an affective switch from depression to mania. We also discuss evidence regarding the likelihood of treatment-emergent mania for antidepressants vs TMS in bipolar depression.
CASE
Ms. W, a 60-year-old White female with a history of bipolar I disorder and attention-deficit/hyperactivity disorder (ADHD), presented for TMS evaluation during a depressive episode. Throughout her life, she had experienced numerous manic episodes, but as she got older she noted an increasing frequency of depressive episodes. Over the course of her illness, she had completed adequate trials at therapeutic doses of many medications, including second-generation antipsychotics (SGAs) (aripiprazole, lurasidone, olanzapine, quetiapine), mood stabilizers (lamotrigine, lithium), and antidepressants (bupropion, venlafaxine, fluoxetine, mirtazapine, trazodone). A course of electroconvulsive therapy was not effective. Ms. W had a long-standing diagnosis of ADHD and had been treated with stimulants for >10 years, although it was unclear whether formal neuropsychological testing had been conducted to confirm this diagnosis. She had >10 suicide attempts and multiple psychiatric hospitalizations.
At her initial evaluation for TMS, Ms. W said she had depressive symptoms predominating for the past 2 years, including low mood, hopelessness, poor sleep, poor appetite, anhedonia, and suicidal ideation without a plan. At the time, she was taking clonazepam, 0.5 mg twice a day; lurasidone, 40 mg/d at bedtime; fluoxetine, 60 mg/d; trazodone, 50 mg/d at bedtime; and methylphenidate, 40 mg/d, and was participating in psychotherapy consistently.
After Ms. W and her clinicians discussed alternatives, risks, benefits, and adverse effects, she consented to adjunctive TMS treatment and provided written informed consent. The treatment plan was outlined as 6 weeks of daily TMS therapy (NeuroStar; Neuronetics, Malvern, PA), 1 treatment per day, 5 days a week. Her clinical status was assessed weekly using the Quick Inventory of Depressive Symptomatology (QIDS) for depression, Generalized Anxiety Disorder 7-item scale (GAD-7) for anxiety, and Young Mania Rating Scale (YMRS) for mania. The Figure shows the trends in Ms. W’s QIDS, GAD-7, and YMRS scores over the course of TMS treatment.

Prior to initiating TMS, her baseline scores were QIDS: 25, GAD-7: 9, and YMRS: 7, indicating very severe depression, mild anxiety, and the absence of mania. Ms. W’s psychotropic regimen remained unchanged throughout the course of her TMS treatment. After her motor threshold was determined, her TMS treatment began at 80% of motor threshold and was titrated up to 95% at the first treatment. By the second treatment, it was titrated up to 110%. By the third treatment, it was titrated up to 120% of motor threshold, which is the percentage used for the remaining treatments.
Initially, Ms. W reported some improvement in her depression, but this improvement was short-lived, and she continued to have elevated QIDS scores throughout treatment. By treatment #21, her QIDS and GAD-7 scores remained elevated, and her YMRS score had increased to 12. Due to this increase in YMRS score, the YMRS was repeated on the next 2 treatment days (#22 and #23), and her score was 6 on both days. When Ms. W presented for treatment #25, she was disorganized, irritable, and endorsed racing thoughts and decreased sleep. She was involuntarily hospitalized for mania, and TMS was discontinued. Unfortunately, she did not complete any clinical scales on that day. Upon admission to the hospital, Ms. W reported that at approximately the time of treatment #21, she had a fluctuation in her mood that consisted of increased goal-directed activity, decreased need for sleep, racing thoughts, and increased frivolous spending. She was treated with lithium, 300 mg twice a day. Lurasidone was increased to 80 mg/d at bedtime, and she continued clonazepam, trazodone, and methylphenidate at the previous doses. Over 14 days, Ms. W’s mania gradually resolved, and she was discharged home.
Continue to: Mixed evidence on the risk of switching
Mixed evidence on the risk of switching
Currently, several TMS devices are FDA-cleared for treating unipolar major depressive disorder, obsessive-compulsive disorder, and certain types of migraine. In March 2020, the FDA granted Breakthrough Device Designation for one TMS device, the NeuroStar Advanced Therapy System, for the treatment of bipolar depression.8 This designation created an expedited pathway for prioritized FDA review of the NeuroStar Advanced Therapy clinical trial program.
Few published clinical studies have evaluated using TMS to treat patients with bipolar depression.9-15 As with any antidepressant treatment for bipolar depression, there is a risk of affective switch from depression to mania when using TMS. Most of the literature available regarding the treatment of bipolar depression focuses on the risk of antidepressant medications to induce an affective switch. This risk depends on the class of the antidepressant,16 and there is a paucity of studies examining the risk of switch with TMS.
Interpretation of available literature is limited due to inconsistencies in the definition of an affective switch, the variable length of treatment with antidepressants, the use of concurrent medications such as mood stabilizers, and confounders such as the natural course of switching in bipolar disorder.17 Overall, the evidence for treatment-emergent mania related to antidepressant use is mixed, and the reported rate of treatment-emergent mania varies. In a systematic review and meta-analysis of >20 randomized controlled trials that included 1,316 patients with bipolar disorder who received antidepressants, Fornaro et al18 found that the incidence of treatment-emergent mania was 11.8%. It is generally recommended that if antidepressants are used to treat patients with bipolar disorder, they should be given with a traditional mood stabilizer to prevent affective switches, although whether mood stabilizers can prevent such switches is unproven.19
In a literature review by Xia et al,20 the affective switch rate in patients with bipolar depression who were treated with TMS was 3.1%, which was not statistically different from the affective switch rate with sham treatment.However, most of the patients included in this analysis were receiving other medications concurrently, and the length of treatment was 2 weeks, which is shorter than the average length of TMS treatment in clinical practice. In a recent literature review by Rachid,21 TMS was found to possibly induce manic episodes when used as monotherapy or in combination with antidepressants in patients with bipolar depression. To reduce the risk of treatment-emergent mania, current recommendations advise the use of a mood stabilizer for a minimum of 2 weeks before initiating TMS.1
In our case, Ms. W was receiving antidepressants (fluoxetine and trazodone), lurasidone (an SGA that is FDA-approved for bipolar depression), and methylphenidate before starting TMS treatment. Fluoxetine, trazodone, and methylphenidate may possibly contribute to an increased risk of an affective switch.1,22 Further studies are needed to clarify whether mood stabilizers or SGAs can prevent the development of mania in patients with bipolar depression who undergo TMS treatment.20
Continue to: Because bipolar depression poses...
Because bipolar depression poses a major clinical challenge,23,24 it is imperative to consider alternate treatments. When evaluating alternative treatment strategies, one may consider TMS in conjunction with a traditional mood stabilizer because this regimen may have a lower risk of treatment-emergent mania compared with antidepressants.1,25
Acknowledgment
The authors thank Dr. Sy Saeed for his expertise and guidance on this article.
Bottom Line
For patients with bipolar depression, treatment with transcranial magnetic stimulation in conjunction with a mood stabilizer may have lower rates of treatment-emergent mania than treatment with antidepressants.
Related Resources
- Transcranial magnetic stimulation: clinical applications for psychiatric practice. Bermudes RA, Lanocha K, Janicak PG, eds. American Psychiatric Association Publishing; 2017.
- Gold AK, Ornelas AC, Cirillo P, et al. Clinical applications of transcranial magnetic stimulation in bipolar disorder. Brain Behav. 2019;9(10):e01419. doi: 10.1002/brb3.1419
Drug Brand Names
Aripiprazole • Abilify
Bupropion • Wellbutrin
Cariprazine • Vraylar
Clonazepam • Klonopin
Fluoxetine • Prozac
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Methylphenidate • Ritalin, Concerta
Mirtazapine • Remeron
Olanzapine • Zyprexa
Olanzapine-fluoxetine • Symbyax
Quetiapine • Seroquel
Trazodone • Desyrel
Venlafaxine • Effexor
Because treatment resistance is a pervasive problem in bipolar depression, the use of neuromodulation treatments such as transcranial magnetic stimulation (TMS) is increasing for patients with this disorder.1-7 Patients with bipolar disorder tend to spend the majority of the time with depressive symptoms, which underscores the importance of providing effective treatment for bipolar depression, especially given the chronicity of this disease.2,3,5 Only a few medications are FDA-approved for treating bipolar depression (Table).

In this article, we describe the case of a patient with treatment-resistant bipolar depression undergoing adjunctive TMS treatment who experienced an affective switch from depression to mania. We also discuss evidence regarding the likelihood of treatment-emergent mania for antidepressants vs TMS in bipolar depression.
CASE
Ms. W, a 60-year-old White female with a history of bipolar I disorder and attention-deficit/hyperactivity disorder (ADHD), presented for TMS evaluation during a depressive episode. Throughout her life, she had experienced numerous manic episodes, but as she got older she noted an increasing frequency of depressive episodes. Over the course of her illness, she had completed adequate trials at therapeutic doses of many medications, including second-generation antipsychotics (SGAs) (aripiprazole, lurasidone, olanzapine, quetiapine), mood stabilizers (lamotrigine, lithium), and antidepressants (bupropion, venlafaxine, fluoxetine, mirtazapine, trazodone). A course of electroconvulsive therapy was not effective. Ms. W had a long-standing diagnosis of ADHD and had been treated with stimulants for >10 years, although it was unclear whether formal neuropsychological testing had been conducted to confirm this diagnosis. She had >10 suicide attempts and multiple psychiatric hospitalizations.
At her initial evaluation for TMS, Ms. W said she had depressive symptoms predominating for the past 2 years, including low mood, hopelessness, poor sleep, poor appetite, anhedonia, and suicidal ideation without a plan. At the time, she was taking clonazepam, 0.5 mg twice a day; lurasidone, 40 mg/d at bedtime; fluoxetine, 60 mg/d; trazodone, 50 mg/d at bedtime; and methylphenidate, 40 mg/d, and was participating in psychotherapy consistently.
After Ms. W and her clinicians discussed alternatives, risks, benefits, and adverse effects, she consented to adjunctive TMS treatment and provided written informed consent. The treatment plan was outlined as 6 weeks of daily TMS therapy (NeuroStar; Neuronetics, Malvern, PA), 1 treatment per day, 5 days a week. Her clinical status was assessed weekly using the Quick Inventory of Depressive Symptomatology (QIDS) for depression, Generalized Anxiety Disorder 7-item scale (GAD-7) for anxiety, and Young Mania Rating Scale (YMRS) for mania. The Figure shows the trends in Ms. W’s QIDS, GAD-7, and YMRS scores over the course of TMS treatment.

Prior to initiating TMS, her baseline scores were QIDS: 25, GAD-7: 9, and YMRS: 7, indicating very severe depression, mild anxiety, and the absence of mania. Ms. W’s psychotropic regimen remained unchanged throughout the course of her TMS treatment. After her motor threshold was determined, her TMS treatment began at 80% of motor threshold and was titrated up to 95% at the first treatment. By the second treatment, it was titrated up to 110%. By the third treatment, it was titrated up to 120% of motor threshold, which is the percentage used for the remaining treatments.
Initially, Ms. W reported some improvement in her depression, but this improvement was short-lived, and she continued to have elevated QIDS scores throughout treatment. By treatment #21, her QIDS and GAD-7 scores remained elevated, and her YMRS score had increased to 12. Due to this increase in YMRS score, the YMRS was repeated on the next 2 treatment days (#22 and #23), and her score was 6 on both days. When Ms. W presented for treatment #25, she was disorganized, irritable, and endorsed racing thoughts and decreased sleep. She was involuntarily hospitalized for mania, and TMS was discontinued. Unfortunately, she did not complete any clinical scales on that day. Upon admission to the hospital, Ms. W reported that at approximately the time of treatment #21, she had a fluctuation in her mood that consisted of increased goal-directed activity, decreased need for sleep, racing thoughts, and increased frivolous spending. She was treated with lithium, 300 mg twice a day. Lurasidone was increased to 80 mg/d at bedtime, and she continued clonazepam, trazodone, and methylphenidate at the previous doses. Over 14 days, Ms. W’s mania gradually resolved, and she was discharged home.
Continue to: Mixed evidence on the risk of switching
Mixed evidence on the risk of switching
Currently, several TMS devices are FDA-cleared for treating unipolar major depressive disorder, obsessive-compulsive disorder, and certain types of migraine. In March 2020, the FDA granted Breakthrough Device Designation for one TMS device, the NeuroStar Advanced Therapy System, for the treatment of bipolar depression.8 This designation created an expedited pathway for prioritized FDA review of the NeuroStar Advanced Therapy clinical trial program.
Few published clinical studies have evaluated using TMS to treat patients with bipolar depression.9-15 As with any antidepressant treatment for bipolar depression, there is a risk of affective switch from depression to mania when using TMS. Most of the literature available regarding the treatment of bipolar depression focuses on the risk of antidepressant medications to induce an affective switch. This risk depends on the class of the antidepressant,16 and there is a paucity of studies examining the risk of switch with TMS.
Interpretation of available literature is limited due to inconsistencies in the definition of an affective switch, the variable length of treatment with antidepressants, the use of concurrent medications such as mood stabilizers, and confounders such as the natural course of switching in bipolar disorder.17 Overall, the evidence for treatment-emergent mania related to antidepressant use is mixed, and the reported rate of treatment-emergent mania varies. In a systematic review and meta-analysis of >20 randomized controlled trials that included 1,316 patients with bipolar disorder who received antidepressants, Fornaro et al18 found that the incidence of treatment-emergent mania was 11.8%. It is generally recommended that if antidepressants are used to treat patients with bipolar disorder, they should be given with a traditional mood stabilizer to prevent affective switches, although whether mood stabilizers can prevent such switches is unproven.19
In a literature review by Xia et al,20 the affective switch rate in patients with bipolar depression who were treated with TMS was 3.1%, which was not statistically different from the affective switch rate with sham treatment.However, most of the patients included in this analysis were receiving other medications concurrently, and the length of treatment was 2 weeks, which is shorter than the average length of TMS treatment in clinical practice. In a recent literature review by Rachid,21 TMS was found to possibly induce manic episodes when used as monotherapy or in combination with antidepressants in patients with bipolar depression. To reduce the risk of treatment-emergent mania, current recommendations advise the use of a mood stabilizer for a minimum of 2 weeks before initiating TMS.1
In our case, Ms. W was receiving antidepressants (fluoxetine and trazodone), lurasidone (an SGA that is FDA-approved for bipolar depression), and methylphenidate before starting TMS treatment. Fluoxetine, trazodone, and methylphenidate may possibly contribute to an increased risk of an affective switch.1,22 Further studies are needed to clarify whether mood stabilizers or SGAs can prevent the development of mania in patients with bipolar depression who undergo TMS treatment.20
Continue to: Because bipolar depression poses...
Because bipolar depression poses a major clinical challenge,23,24 it is imperative to consider alternate treatments. When evaluating alternative treatment strategies, one may consider TMS in conjunction with a traditional mood stabilizer because this regimen may have a lower risk of treatment-emergent mania compared with antidepressants.1,25
Acknowledgment
The authors thank Dr. Sy Saeed for his expertise and guidance on this article.
Bottom Line
For patients with bipolar depression, treatment with transcranial magnetic stimulation in conjunction with a mood stabilizer may have lower rates of treatment-emergent mania than treatment with antidepressants.
Related Resources
- Transcranial magnetic stimulation: clinical applications for psychiatric practice. Bermudes RA, Lanocha K, Janicak PG, eds. American Psychiatric Association Publishing; 2017.
- Gold AK, Ornelas AC, Cirillo P, et al. Clinical applications of transcranial magnetic stimulation in bipolar disorder. Brain Behav. 2019;9(10):e01419. doi: 10.1002/brb3.1419
Drug Brand Names
Aripiprazole • Abilify
Bupropion • Wellbutrin
Cariprazine • Vraylar
Clonazepam • Klonopin
Fluoxetine • Prozac
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Lurasidone • Latuda
Methylphenidate • Ritalin, Concerta
Mirtazapine • Remeron
Olanzapine • Zyprexa
Olanzapine-fluoxetine • Symbyax
Quetiapine • Seroquel
Trazodone • Desyrel
Venlafaxine • Effexor
1. Aaronson ST, Croarkin PE. Transcranial magnetic stimulation for the treatment of other mood disorders. In: Bermudes RA, Lanocha K, Janicak PG, eds. Transcranial magnetic stimulation: clinical applications for psychiatric practice. American Psychiatric Association Publishing; 2017:127-156.
2. Geddes JR, Miklowitz DJ. Treatment of bipolar disorder. Lancet. 2013;381(9878):1672-1682.
3. Gitlin M. Treatment-resistant bipolar disorder. Molecular Psychiatry. 2006;11(3):227-240.
4. Harrison PJ, Geddes JR, Tunbridge EM. The emerging neurobiology of bipolar disorder. Trends Neurosci. 2018;41(1):18-30.
5. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the World Mental Health Survey Initiative. Arch Gen Psychiatry. 2011;68(3):241-251.
6. Myczkowski ML, Fernandes A, Moreno M, et al. Cognitive outcomes of TMS treatment in bipolar depression: safety data from a randomized controlled trial. J Affect Disord. 2018;235: 20-26.
7. Tavares DF, Myczkowski ML, Alberto RL, et al. Treatment of bipolar depression with deep TMS: results from a double-blind, randomized, parallel group, sham-controlled clinical trial. Neuropsychopharmacology. 2017;42(13):2593-2601.
8. Neuronetics. FDA grants NeuroStar® Advanced Therapy System Breakthrough Device Designation to treat bipolar depression. Accessed February 2, 2021. https://www.globenewswire.com/news-release/2020/03/06/1996447/0/en/FDA-Grants-NeuroStar-Advanced-Therapy-System-Breakthrough-Device-Designation-to-Treat-Bipolar-Depression.html
9. Cohen RB, Brunoni AR, Boggio PS, et al. Clinical predictors associated with duration of repetitive transcranial magnetic stimulation treatment for remission in bipolar depression: a naturalistic study. J Nerv Ment Dis. 2010;198(9):679-681.
10. Connolly KR, Helmer A, Cristancho MA, et al. Effectiveness of transcranial magnetic stimulation in clinical practice post-FDA approval in the United States: results observed with the first 100 consecutive cases of depression at an academic medical center. J Clin Psychiatry. 2012;73(4):e567-e573.
11. Dell’osso B, D’Urso N, Castellano F, et al. Long-term efficacy after acute augmentative repetitive transcranial magnetic stimulation in bipolar depression: a 1-year follow-up study. J ECT. 2011;27(2):141-144.
12. Dell’Osso B, Mundo E, D’Urso N, et al. Augmentative repetitive navigated transcranial magnetic stimulation (rTMS) in drug-resistant bipolar depression. Bipolar Disord. 2009;11(1):76-81.
13. Harel EV, Zangen A, Roth Y, et al. H-coil repetitive transcranial magnetic stimulation for the treatment of bipolar depression: an add-on, safety and feasibility study. World J Biol Psychiatry. 2011;12(2):119-126.
14. Nahas Z, Kozel FA, Li X, et al. Left prefrontal transcranial magnetic stimulation (TMS) treatment of depression in bipolar affective disorder: a pilot study of acute safety and efficacy. Bipolar Disord. 2003;5(1):40-47.
15. Tamas RL, Menkes D, El-Mallakh RS. Stimulating research: a prospective, randomized, double-blind, sham-controlled study of slow transcranial magnetic stimulation in depressed bipolar patients. J Neuropsychiatry Clin Neurosci. 2007;19(2):198-199.
16. Tundo A, Cavalieri P, Navari S, et al. Treating bipolar depression - antidepressants and alternatives: a critical review of the literature. Acta Neuropsychiatrica. 2011:23(3):94-105.
17. Gijsman HJ, Geddes JR, Rendell JM, et al. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry. 2004;161(9):1537-1547.
18. Fornaro M, Anastasia A, Novello S, et al. Incidence, prevalence and clinical correlates of antidepressant‐emergent mania in bipolar depression: a systematic review and meta‐analysis. Bipolar Disord. 2018;20(3):195-227.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society for Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262.
20. Xia G, Gajwani P, Muzina DJ, et al. Treatment-emergent mania in unipolar and bipolar depression: focus on repetitive transcranial magnetic stimulation. Int J Neuropsychopharmacol. 2008;11(1):119-130.
21. Rachid F. Repetitive transcranial magnetic stimulation and treatment-emergent mania and hypomania: a review of the literature. J Psychiatr Pract. 2017;23(2):150-159.
22. Victorin A, Rydén E, Thase M, et al. The risk of treatment-emergent mania with methylphenidate in bipolar disorder. Am J Psychiatry. 2017;174(4):341-348.
23. Hidalgo-Mazzei D, Berk M, Cipriani A, et al. Treatment-resistant and multi-therapy-resistant criteria for bipolar depression: consensus definition. Br J Psychiatry. 2019;214(1):27-35.
24. Baldessarini RJ, Vázquez GH, Tondo L. Bipolar depression: a major unsolved challenge. Int J Bipolar Disord. 2020;8(1):1.
25. Phillips AL, Burr RL, Dunner DL. Repetitive transcranial magnetic stimulation in the treatment of bipolar depression: Experience from a clinical setting. J Psychiatr Pract. 2020;26(1):37-45.
1. Aaronson ST, Croarkin PE. Transcranial magnetic stimulation for the treatment of other mood disorders. In: Bermudes RA, Lanocha K, Janicak PG, eds. Transcranial magnetic stimulation: clinical applications for psychiatric practice. American Psychiatric Association Publishing; 2017:127-156.
2. Geddes JR, Miklowitz DJ. Treatment of bipolar disorder. Lancet. 2013;381(9878):1672-1682.
3. Gitlin M. Treatment-resistant bipolar disorder. Molecular Psychiatry. 2006;11(3):227-240.
4. Harrison PJ, Geddes JR, Tunbridge EM. The emerging neurobiology of bipolar disorder. Trends Neurosci. 2018;41(1):18-30.
5. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the World Mental Health Survey Initiative. Arch Gen Psychiatry. 2011;68(3):241-251.
6. Myczkowski ML, Fernandes A, Moreno M, et al. Cognitive outcomes of TMS treatment in bipolar depression: safety data from a randomized controlled trial. J Affect Disord. 2018;235: 20-26.
7. Tavares DF, Myczkowski ML, Alberto RL, et al. Treatment of bipolar depression with deep TMS: results from a double-blind, randomized, parallel group, sham-controlled clinical trial. Neuropsychopharmacology. 2017;42(13):2593-2601.
8. Neuronetics. FDA grants NeuroStar® Advanced Therapy System Breakthrough Device Designation to treat bipolar depression. Accessed February 2, 2021. https://www.globenewswire.com/news-release/2020/03/06/1996447/0/en/FDA-Grants-NeuroStar-Advanced-Therapy-System-Breakthrough-Device-Designation-to-Treat-Bipolar-Depression.html
9. Cohen RB, Brunoni AR, Boggio PS, et al. Clinical predictors associated with duration of repetitive transcranial magnetic stimulation treatment for remission in bipolar depression: a naturalistic study. J Nerv Ment Dis. 2010;198(9):679-681.
10. Connolly KR, Helmer A, Cristancho MA, et al. Effectiveness of transcranial magnetic stimulation in clinical practice post-FDA approval in the United States: results observed with the first 100 consecutive cases of depression at an academic medical center. J Clin Psychiatry. 2012;73(4):e567-e573.
11. Dell’osso B, D’Urso N, Castellano F, et al. Long-term efficacy after acute augmentative repetitive transcranial magnetic stimulation in bipolar depression: a 1-year follow-up study. J ECT. 2011;27(2):141-144.
12. Dell’Osso B, Mundo E, D’Urso N, et al. Augmentative repetitive navigated transcranial magnetic stimulation (rTMS) in drug-resistant bipolar depression. Bipolar Disord. 2009;11(1):76-81.
13. Harel EV, Zangen A, Roth Y, et al. H-coil repetitive transcranial magnetic stimulation for the treatment of bipolar depression: an add-on, safety and feasibility study. World J Biol Psychiatry. 2011;12(2):119-126.
14. Nahas Z, Kozel FA, Li X, et al. Left prefrontal transcranial magnetic stimulation (TMS) treatment of depression in bipolar affective disorder: a pilot study of acute safety and efficacy. Bipolar Disord. 2003;5(1):40-47.
15. Tamas RL, Menkes D, El-Mallakh RS. Stimulating research: a prospective, randomized, double-blind, sham-controlled study of slow transcranial magnetic stimulation in depressed bipolar patients. J Neuropsychiatry Clin Neurosci. 2007;19(2):198-199.
16. Tundo A, Cavalieri P, Navari S, et al. Treating bipolar depression - antidepressants and alternatives: a critical review of the literature. Acta Neuropsychiatrica. 2011:23(3):94-105.
17. Gijsman HJ, Geddes JR, Rendell JM, et al. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry. 2004;161(9):1537-1547.
18. Fornaro M, Anastasia A, Novello S, et al. Incidence, prevalence and clinical correlates of antidepressant‐emergent mania in bipolar depression: a systematic review and meta‐analysis. Bipolar Disord. 2018;20(3):195-227.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society for Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262.
20. Xia G, Gajwani P, Muzina DJ, et al. Treatment-emergent mania in unipolar and bipolar depression: focus on repetitive transcranial magnetic stimulation. Int J Neuropsychopharmacol. 2008;11(1):119-130.
21. Rachid F. Repetitive transcranial magnetic stimulation and treatment-emergent mania and hypomania: a review of the literature. J Psychiatr Pract. 2017;23(2):150-159.
22. Victorin A, Rydén E, Thase M, et al. The risk of treatment-emergent mania with methylphenidate in bipolar disorder. Am J Psychiatry. 2017;174(4):341-348.
23. Hidalgo-Mazzei D, Berk M, Cipriani A, et al. Treatment-resistant and multi-therapy-resistant criteria for bipolar depression: consensus definition. Br J Psychiatry. 2019;214(1):27-35.
24. Baldessarini RJ, Vázquez GH, Tondo L. Bipolar depression: a major unsolved challenge. Int J Bipolar Disord. 2020;8(1):1.
25. Phillips AL, Burr RL, Dunner DL. Repetitive transcranial magnetic stimulation in the treatment of bipolar depression: Experience from a clinical setting. J Psychiatr Pract. 2020;26(1):37-45.
Steroid-induced psychiatric symptoms: What you need to know
Ms. N, age 30, presents to the emergency department for altered mental status, insomnia, and behavioral changes, which she has experienced for 1 week. On evaluation, she grabs a clinician’s hand and details her business ideas and life story with no prompting. Ms. N’s mental status examination is significant for hyperverbal speech with increased rate and volume; tangential thought process; and bright, expanded affect.
One week earlier, Ms. N was hospitalized for sudden-onset chest pain, weakness, and dizziness. She received 45 minutes of cardiopulmonary resuscitation prior to presentation and was found to have a ST-segment elevation myocardial infarction that required emergent left anterior descending coronary artery and right coronary artery percutaneous coronary intervention to place drug-eluting stents. Her recovery was complicated by acute cardiogenic shock, pulmonary edema, and hypoxic respiratory failure. Subsequently, she was intubated, admitted to the ICU, and received high-dose corticosteroids, including IV methylprednisolone, 40 mg every 12 hours, which was tapered prior to discharge. Her husband reports that since Ms. N came home, she has been more talkative and irritable, ruminating about past events, unable to sleep (<1 hour/night), and crying frequently. She has also been endorsing visual and auditory hallucinations, with increased praying and listening to religious music.
The frequent clinical use of steroids necessitates an understanding of these medications’ various adverse effects. The manifestations of steroid-induced psychiatric symptoms are broad and can involve affective, behavioral, and cognitive domains. While the current mechanism is unknown, this phenomenon may be related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity, as well as effects on brain regions such as the hippocampus and amygdala. The best interventions for steroid-induced psychiatric symptoms are awareness and early diagnosis. There are no FDA-approved treatments for steroid-induced psychiatric symptoms; initial measures should include tapering or discontinuing corticosteroids.
In this article, we review the literature on the incidence, characteristics, differential diagnoses, proposed mechanism, risk factors, and proposed treatments of steroid-induced psychiatric symptoms.
A wide range of presentations
Steroid use has increased over the past 2 decades, with 10% of medical and surgical inpatients and 1% to 3% of the general population taking long-term glucocorticoids.1 Even with topical application, steroid therapy is often systemically absorbed, and thus may lead to steroid-induced psychiatric symptoms. The incidence of steroid-induced psychiatric symptoms is difficult to assess because there can be a wide range of reactions that are dose- and time-related. Three reviews of a total of 122 cases reports found that an estimated 5% of patients treated with steroids experience severe psychiatric reactions.1-3
Steroid-induced psychopathology can include mood, behavioral, and/or cognitive impairments. Mania/hypomania is the most common overall psychiatric symptom; the most common mood manifestations are anxiety and depression.4,5 Other possible steroid-induced symptoms include psychosis, dementia, panic disorder, delirium, suicidal thinking and behavior, aggressive behavior, insomnia, agitation, depersonalization, and euphoria.5 The most common cognitive impairment is verbal or declarative memory deficit; others include distractibility and deficits in attention and psychomotor speed.5 These psychiatric symptoms can have a rapid onset, possibly within hours of starting steroids.1 However, studies have reported a median time to onset of 11.5 days; 39% of cases had onset during the first week and 62% within 2 weeks.3,6 After reducing or stopping the steroid, it may take days to weeks before symptoms start to subside.2
What to consider in the differential Dx
Psychiatric symptoms that are induced by steroids can mimic metabolic, neurologic, or toxic disorders. Other factors to consider include drug withdrawal/intoxication, infections, and paraneoplastic syndromes.4,5 Although there is no reported correlation between the location of neurologic lesions and the development of specific psychiatric symptoms, manic symptoms appear most commonly with lesions in the right frontal lobe. 4 Other factors to note include the presence of new-onset psychiatric illnesses such as bipolar, mood, or thought disorders,4 as well as psychosocial stressors that might be contributing to the patient’s presentation.5
Continue to: Proposed mechanisms
Proposed mechanisms
Although the exact mechanism by which steroids induce psychiatric symptoms is unknown, several mechanisms have been proposed. One hypothesis is that steroid-induced psychopathology is related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity.4,5,7 This may explain why many patients with major depressive disorder have elevated cortisol production and/or lack of suppression of cortisol secretion during a dexamethasone stimulation test, and why approximately one-half of patients with Cushing’s disease experience depressive symptoms.8 This is also likely why antipsychotics, which typically reduce cortisol, are efficacious treatments for some steroid-induced psychiatric symptoms.9
Cognitive impairments from steroid use may be related to these agents’ effects on certain brain regions. One such area is the hippocampus, an important mediator in the creation and maintenance of episodic and declarative memories.5,8,9 Acute glucocorticoid use is associated with decreased activity in the left hippocampus, reduced hippocampal glucose metabolism, and reduced cerebral blood flow in the posterior medial temporal lobe.10 Long-term glucocorticoid exposure is associated with smaller hippocampal volume and lower levels of temporal lobe N-acetylaspartate, a marker of neuronal viability.10 Because working memory depends on the prefrontal cortex and declarative memory relies on the hippocampus, deficits in these functions can be attributed to the effect of prolonged glucocorticoid exposure on glucocorticoid or mineralocorticoid receptors in the hippocampus, reduction of hippocampal volume, or elevated glutamate accumulation in that area.11 In addition, high cortisol levels inhibit brain-derived neurotrophic factor, which plays a crucial role in maintaining neural architecture in key brain regions such as the hippocampus and prefrontal cortex.11 There is also a correlation between the duration of prednisone treatment and atrophy of the right amygdala, which is an important regulator of mood and anxiety.11 Both the hippocampus and amygdala have dense collections of glucocorticoid receptors. This may explain why patients who receive high-dose corticosteroids can have reversible atrophy in the hypothalamus and amygdala, leading to deficits in emotional learning and the stress response.
Factors that increase risk
Several factors can increase the risk of steroid-induced psychopathology. The most significant is the dose; higher doses are more likely to produce psychiatric symptoms.1,5 Concurrent use of drugs that increase circulating levels of corticosteroids, such as inhibitors of the cytochrome P450 (CYP) enzyme (eg, clarithromycin), also increases the likelihood of developing psychiatric symptoms.1,5 Risk is also increased in patients with liver or renal dysfunction.1 Cerebral spinal fluid/serum albumin ratio, a marker of blood-brain barrier damage, and low serum complement levels were also reported to be independent risk factors,12 with the thought that increased permeability of the blood-brain barrier may allow hydrophobic steroid molecules to more easily penetrate the CNS, leading to increased neuropsychiatric effects. Hypoalbuminemia is another reported risk factor, perhaps because lower levels of serum albumin are related to higher levels of free and active glucocorticoids, which are normally inactive when bound to albumin.13 There also appears to be an increased prevalence of steroid-induced psychopathology in women, perhaps due to greater propensity in women to seek medical care or a higher prevalence of women with medical disorders that are treated with steroids.5 A previous history of psychiatric disorders may not increase risk.5
Several methods for reducing risk have been proposed, including using a divided-dosing regimens that may lower peak steroid plasma concentrations.13,14 However, the best prevention of steroid-induced psychiatric symptoms are awareness, early diagnosis, and intervention. Studies have suggested that N-methyl-
Treatment options
There are no FDA-approved medications for managing steroid-induced psychiatric symptoms.1,16 Treatment is based on evidence from case reports and a few small case series (Table2-5,17,18).

Continue to: When possible, initial treatment...
When possible, initial treatment should include discontinuing or tapering corticosteroids to <40 mg/d of prednisone-equivalent.1,4,10,18 Most studies have reported rapid reversal of deficits in declarative memory and of hippocampal volume loss once corticosteroids were tapered and discontinued.1,18 One study reported that >90% of patients recovered within 6 weeks, with patients with delirium recovering more quickly (mean: 5.4 days) than those with depression, mania, or psychosis (mean: 19.3 days).3 Another found that the vast majority (92%) of patients treated only with a steroid taper achieved clinical recovery, and 84% recovered with administration of antipsychotics without a steroid taper.3 In this study, all patients who received electroconvulsive therapy (ECT) recovered, as did those who received a steroid taper plus lithium or antipsychotics. Steroid tapering regimens are especially important for patients who have received long-term glucocorticoid treatment. Patients need to be closely monitored for signs of new or increased depression, delirium, or confusion during the taper. If these symptoms occur, the patient should be checked for adrenocortical insufficiency, which can be resolved by re-administering or increasing the dosage of the glucocorticoid.10
Mania. The treatment of mania/hypomania includes mood stabilizers (valproate, lithium, lamotrigine) and antipsychotics (quetiapine, olanzapine, haloperidol).2,4,5,10,14,18 Valproate has been reported to be an effective prophylactic of corticosteroid-induced mania,2 perhaps because it dampens neuronal hyperexcitability by attenuating NMDA receptors, blocking voltage-dependent sodium channels, and inhibiting the synthesis of cortical GABAergic steroids. Starting valproate while continuing corticosteroids (if necessary) may help lessen mania.2 Benzodiazepines also may be useful on a short-term basis.
Depression. Steroid-induced depression may be treated with sertraline or other first-line antidepressants.5,14 Consider ECT for patients with severe depression. Support for the use of antipsychotic medications stems from studies that reported steroids’ role in disrupting dopamine and 5HT2 activity. Lithium also has been used successfully to manage and prevent glucocorticoid-associated affective disorder.10,18 It can be used alone or in combination with selective serotonin reuptake inhibitors to alleviate depressive symptoms.10 Tricyclic antidepressants are generally avoided because their anticholinergic effects can exacerbate or worsen delirium.18 In general, ECT is an effective treatment for persistent and/or unresponsive steroid-induced depression,2,10 but may be difficult to use in patients with serious medical illnesses.
Agitation. Medications that have been proposed for treating steroid-induced agitation include benzodiazepines, haloperidol, and second-generation antipsychotics.5,17
Other considerations. Clinicians, patients, and families should discuss in detail the risks of steroid-induced psychiatric symptoms so an early diagnosis and appropriate intervention can be implemented. Before starting steroids, it is important to review the patient’s current medication list to ensure that steroid treatment is indicated, and to check for potential drug–drug interactions. In addition, the medical condition that is being treated with steroids also needs to be carefully reviewed, because certain illnesses are associated with the development of psychiatric symptoms. 5,10
Continue to: Young children...
Young children (age <6) and older adults appear to be at greater risk for cognitive and memory disturbances from steroid use.10 In addition, patients have individual levels of susceptibility to steroid-induced psychiatric symptoms that can vary over time. The risk for adverse effects may be elevated based on response to previous courses of glucocorticoid treatment.10 While gender, age, dosage, and duration of treatment influence risk, it is not possible to predict which patients will experience psychiatric effects during a given course of glucocorticoid therapy. Therefore, all patients should be considered to have the potential of developing such effects, and should be monitored during glucocorticoid treatment and withdrawal.
Goals for future research
To help reduce the severity of and cost associated with steroid-induced psychiatric symptoms,5,14 future studies should focus on controlled trials of preventative strategies. In particular, recent advances in genetic mapping may help identify involvement of certain genes or polymorphisms.5 Because current guidelines for the prevention and treatment of steroid-induced psychiatric symptoms are not evidence-based, controlled clinical trials are needed to elucidate the optimal management of such symptoms. There is much interplay between many of the proposed mechanisms of steroid-induced psychiatric symptoms, and future studies can help uncover a deeper understanding of the intricacies of this phenomenon.
CASE CONTINUED
Mrs. N is admitted for altered mental status. Medical workup includes MRI of the brain, MRI of the neck, cardiac echocardiogram, and EEG. There is no evidence of acute structural pathology. She is started on olanzapine, 10 mg/d at bedtime for manic and psychotic symptoms, and is discharged after 5 days. After 1 month, the outpatient psychiatrist gradually decreases and discontinues olanzapine as Mrs. N steadily returns to baseline. One year after discharge, Mrs. N continues to report resolution of her manic and psychotic symptoms.
Bottom Line
Steroids can induce a wide range of psychiatric symptoms, including mania/ hypomania, anxiety, and depression. Initial treatment typically includes tapering or discontinuing the steroid when possible. Other proposed treatments include certain antipsychotics, antidepressants, and other psychotropics, but the supporting evidence is largely anecdotal or based on case studies. Additional research is needed to elucidate the mechanism and treatment recommendations.
Related Resources
- Janes M, Kuster S, Goldson TM, et al. Steroid-induced psychosis. Proc (Bayl Univ Med Cent). 2019;32(4):614-615.
- Mayo Clinic. Prednisone and other corticosteroids. https://www.mayoclinic.org/steroids/art-20045692
Drug Brand Names
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Methylprednisolone injection • Solu-Medrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Phenytoin • Dilantin
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproate • Depakote
1. Dubovsky AN, Arvikar S, Stern TA, et al. The neuropsychiatric complications of glucocorticoid use: steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
2. Roxanas MG, Hunt GE. Rapid reversal of corticosteroid-induced mania with sodium valproate: a case series of 20 patients. Psychosomatics. 2012;53(6):575-581.
3. Lewis DA, Smith RE. Steroid‐induced psychiatric syndromes. A report of 14 cases and a review of the literature. J Affect Disord. 1983;5(4):319-332.
4. Warren KN, Katakam J, Espiridion ED. Acute-onset mania in a patient with non-small cell lung cancer. Cureus. 2019;11(8):e5436.
5. Kenna HA, Poon AW, de los Angeles CP, et al. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549-560.
6. Ling MH, Perry PJ, Tsuang MT. Side effects of corticosteroid therapy. Psychiatric aspects. Arch Gen. Psychiatry. 1981;38(4):471-477.
7. Ularntinon S, Tzuang D, Dahl G, et al. Concurrent treatment of steroid-related mood and psychotic symptoms with risperidone. Pediatrics. 2010;125(5):e1241-e1245.
8. Pokladinkova J, Meyboom RH, Vlcek J, et al. Intranasally administered corticosteroids and neuropsychiatric disturbances: a review of the international pharmacovigilance programme of the World Health Organization. Ann Allergy Asthma Immunol. 2008;101(1):67-73.
9. Walker EF, Trotman HD, Pearce BD, et al. Cortisol levels and risk for psychosis: initial findings from the North American prodrome longitudinal study. Biol Psychiatry. 2013;74(6):410-417.
10. Wolkowitz OM, Reus UI. Treatment of depression with antiglucocorticoid drugs. Psychosom Med. 1999;61(5):698-711.
11. Judd LL, Schettler PJ, Brown ES, et al. Adverse consequences of glucocorticoid medication: psychological, cognitive, and behavioral effects. Am J Psychiatry. 2014;171(10):1045-1051.
12. Appenzeller S, Cendes F, Costallat LT. Acute psychosis in systemic lupus erythematosus. Rheumatol Int. 2008;28(3):237-243.
13. Glynne-Jones R, Vernon CC, Bell G. Is steroid psychosis preventable by divided doses? Lancet. 1986;2(8520):1404.
14. Ismail MF, Lavelle C, Cassidy EM. Steroid-induced mental disorders in cancer patients: a systematic review. Future Oncol. 2017;13(29):2719-2731.
15. Magariños AM, McEwen BS. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience. 1995;69(1):89-98.
16. Brown BS, Stuard G, Liggin JDM, et al. Effect of phenytoin on mood and declarative memory during prescription corticosteroid therapy. Biol Psychiatry. 2005;57(5):543-548.
17. Desai S, Khanani S, Shad MU, et al. Attenutation of amygdala atrophy with lamotrigine in patients receiving corticosteroid therapy. J Clin Psychopharmacol. 2009;29(3):284-287.
18. Gable M, Depry D. Sustained corticosteroid-induced mania and psychosis despite cessation: a case study and brief literature review. Int J Psychiatry Med. 2015;50(4):398-404.
Ms. N, age 30, presents to the emergency department for altered mental status, insomnia, and behavioral changes, which she has experienced for 1 week. On evaluation, she grabs a clinician’s hand and details her business ideas and life story with no prompting. Ms. N’s mental status examination is significant for hyperverbal speech with increased rate and volume; tangential thought process; and bright, expanded affect.
One week earlier, Ms. N was hospitalized for sudden-onset chest pain, weakness, and dizziness. She received 45 minutes of cardiopulmonary resuscitation prior to presentation and was found to have a ST-segment elevation myocardial infarction that required emergent left anterior descending coronary artery and right coronary artery percutaneous coronary intervention to place drug-eluting stents. Her recovery was complicated by acute cardiogenic shock, pulmonary edema, and hypoxic respiratory failure. Subsequently, she was intubated, admitted to the ICU, and received high-dose corticosteroids, including IV methylprednisolone, 40 mg every 12 hours, which was tapered prior to discharge. Her husband reports that since Ms. N came home, she has been more talkative and irritable, ruminating about past events, unable to sleep (<1 hour/night), and crying frequently. She has also been endorsing visual and auditory hallucinations, with increased praying and listening to religious music.
The frequent clinical use of steroids necessitates an understanding of these medications’ various adverse effects. The manifestations of steroid-induced psychiatric symptoms are broad and can involve affective, behavioral, and cognitive domains. While the current mechanism is unknown, this phenomenon may be related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity, as well as effects on brain regions such as the hippocampus and amygdala. The best interventions for steroid-induced psychiatric symptoms are awareness and early diagnosis. There are no FDA-approved treatments for steroid-induced psychiatric symptoms; initial measures should include tapering or discontinuing corticosteroids.
In this article, we review the literature on the incidence, characteristics, differential diagnoses, proposed mechanism, risk factors, and proposed treatments of steroid-induced psychiatric symptoms.
A wide range of presentations
Steroid use has increased over the past 2 decades, with 10% of medical and surgical inpatients and 1% to 3% of the general population taking long-term glucocorticoids.1 Even with topical application, steroid therapy is often systemically absorbed, and thus may lead to steroid-induced psychiatric symptoms. The incidence of steroid-induced psychiatric symptoms is difficult to assess because there can be a wide range of reactions that are dose- and time-related. Three reviews of a total of 122 cases reports found that an estimated 5% of patients treated with steroids experience severe psychiatric reactions.1-3
Steroid-induced psychopathology can include mood, behavioral, and/or cognitive impairments. Mania/hypomania is the most common overall psychiatric symptom; the most common mood manifestations are anxiety and depression.4,5 Other possible steroid-induced symptoms include psychosis, dementia, panic disorder, delirium, suicidal thinking and behavior, aggressive behavior, insomnia, agitation, depersonalization, and euphoria.5 The most common cognitive impairment is verbal or declarative memory deficit; others include distractibility and deficits in attention and psychomotor speed.5 These psychiatric symptoms can have a rapid onset, possibly within hours of starting steroids.1 However, studies have reported a median time to onset of 11.5 days; 39% of cases had onset during the first week and 62% within 2 weeks.3,6 After reducing or stopping the steroid, it may take days to weeks before symptoms start to subside.2
What to consider in the differential Dx
Psychiatric symptoms that are induced by steroids can mimic metabolic, neurologic, or toxic disorders. Other factors to consider include drug withdrawal/intoxication, infections, and paraneoplastic syndromes.4,5 Although there is no reported correlation between the location of neurologic lesions and the development of specific psychiatric symptoms, manic symptoms appear most commonly with lesions in the right frontal lobe. 4 Other factors to note include the presence of new-onset psychiatric illnesses such as bipolar, mood, or thought disorders,4 as well as psychosocial stressors that might be contributing to the patient’s presentation.5
Continue to: Proposed mechanisms
Proposed mechanisms
Although the exact mechanism by which steroids induce psychiatric symptoms is unknown, several mechanisms have been proposed. One hypothesis is that steroid-induced psychopathology is related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity.4,5,7 This may explain why many patients with major depressive disorder have elevated cortisol production and/or lack of suppression of cortisol secretion during a dexamethasone stimulation test, and why approximately one-half of patients with Cushing’s disease experience depressive symptoms.8 This is also likely why antipsychotics, which typically reduce cortisol, are efficacious treatments for some steroid-induced psychiatric symptoms.9
Cognitive impairments from steroid use may be related to these agents’ effects on certain brain regions. One such area is the hippocampus, an important mediator in the creation and maintenance of episodic and declarative memories.5,8,9 Acute glucocorticoid use is associated with decreased activity in the left hippocampus, reduced hippocampal glucose metabolism, and reduced cerebral blood flow in the posterior medial temporal lobe.10 Long-term glucocorticoid exposure is associated with smaller hippocampal volume and lower levels of temporal lobe N-acetylaspartate, a marker of neuronal viability.10 Because working memory depends on the prefrontal cortex and declarative memory relies on the hippocampus, deficits in these functions can be attributed to the effect of prolonged glucocorticoid exposure on glucocorticoid or mineralocorticoid receptors in the hippocampus, reduction of hippocampal volume, or elevated glutamate accumulation in that area.11 In addition, high cortisol levels inhibit brain-derived neurotrophic factor, which plays a crucial role in maintaining neural architecture in key brain regions such as the hippocampus and prefrontal cortex.11 There is also a correlation between the duration of prednisone treatment and atrophy of the right amygdala, which is an important regulator of mood and anxiety.11 Both the hippocampus and amygdala have dense collections of glucocorticoid receptors. This may explain why patients who receive high-dose corticosteroids can have reversible atrophy in the hypothalamus and amygdala, leading to deficits in emotional learning and the stress response.
Factors that increase risk
Several factors can increase the risk of steroid-induced psychopathology. The most significant is the dose; higher doses are more likely to produce psychiatric symptoms.1,5 Concurrent use of drugs that increase circulating levels of corticosteroids, such as inhibitors of the cytochrome P450 (CYP) enzyme (eg, clarithromycin), also increases the likelihood of developing psychiatric symptoms.1,5 Risk is also increased in patients with liver or renal dysfunction.1 Cerebral spinal fluid/serum albumin ratio, a marker of blood-brain barrier damage, and low serum complement levels were also reported to be independent risk factors,12 with the thought that increased permeability of the blood-brain barrier may allow hydrophobic steroid molecules to more easily penetrate the CNS, leading to increased neuropsychiatric effects. Hypoalbuminemia is another reported risk factor, perhaps because lower levels of serum albumin are related to higher levels of free and active glucocorticoids, which are normally inactive when bound to albumin.13 There also appears to be an increased prevalence of steroid-induced psychopathology in women, perhaps due to greater propensity in women to seek medical care or a higher prevalence of women with medical disorders that are treated with steroids.5 A previous history of psychiatric disorders may not increase risk.5
Several methods for reducing risk have been proposed, including using a divided-dosing regimens that may lower peak steroid plasma concentrations.13,14 However, the best prevention of steroid-induced psychiatric symptoms are awareness, early diagnosis, and intervention. Studies have suggested that N-methyl-
Treatment options
There are no FDA-approved medications for managing steroid-induced psychiatric symptoms.1,16 Treatment is based on evidence from case reports and a few small case series (Table2-5,17,18).

Continue to: When possible, initial treatment...
When possible, initial treatment should include discontinuing or tapering corticosteroids to <40 mg/d of prednisone-equivalent.1,4,10,18 Most studies have reported rapid reversal of deficits in declarative memory and of hippocampal volume loss once corticosteroids were tapered and discontinued.1,18 One study reported that >90% of patients recovered within 6 weeks, with patients with delirium recovering more quickly (mean: 5.4 days) than those with depression, mania, or psychosis (mean: 19.3 days).3 Another found that the vast majority (92%) of patients treated only with a steroid taper achieved clinical recovery, and 84% recovered with administration of antipsychotics without a steroid taper.3 In this study, all patients who received electroconvulsive therapy (ECT) recovered, as did those who received a steroid taper plus lithium or antipsychotics. Steroid tapering regimens are especially important for patients who have received long-term glucocorticoid treatment. Patients need to be closely monitored for signs of new or increased depression, delirium, or confusion during the taper. If these symptoms occur, the patient should be checked for adrenocortical insufficiency, which can be resolved by re-administering or increasing the dosage of the glucocorticoid.10
Mania. The treatment of mania/hypomania includes mood stabilizers (valproate, lithium, lamotrigine) and antipsychotics (quetiapine, olanzapine, haloperidol).2,4,5,10,14,18 Valproate has been reported to be an effective prophylactic of corticosteroid-induced mania,2 perhaps because it dampens neuronal hyperexcitability by attenuating NMDA receptors, blocking voltage-dependent sodium channels, and inhibiting the synthesis of cortical GABAergic steroids. Starting valproate while continuing corticosteroids (if necessary) may help lessen mania.2 Benzodiazepines also may be useful on a short-term basis.
Depression. Steroid-induced depression may be treated with sertraline or other first-line antidepressants.5,14 Consider ECT for patients with severe depression. Support for the use of antipsychotic medications stems from studies that reported steroids’ role in disrupting dopamine and 5HT2 activity. Lithium also has been used successfully to manage and prevent glucocorticoid-associated affective disorder.10,18 It can be used alone or in combination with selective serotonin reuptake inhibitors to alleviate depressive symptoms.10 Tricyclic antidepressants are generally avoided because their anticholinergic effects can exacerbate or worsen delirium.18 In general, ECT is an effective treatment for persistent and/or unresponsive steroid-induced depression,2,10 but may be difficult to use in patients with serious medical illnesses.
Agitation. Medications that have been proposed for treating steroid-induced agitation include benzodiazepines, haloperidol, and second-generation antipsychotics.5,17
Other considerations. Clinicians, patients, and families should discuss in detail the risks of steroid-induced psychiatric symptoms so an early diagnosis and appropriate intervention can be implemented. Before starting steroids, it is important to review the patient’s current medication list to ensure that steroid treatment is indicated, and to check for potential drug–drug interactions. In addition, the medical condition that is being treated with steroids also needs to be carefully reviewed, because certain illnesses are associated with the development of psychiatric symptoms. 5,10
Continue to: Young children...
Young children (age <6) and older adults appear to be at greater risk for cognitive and memory disturbances from steroid use.10 In addition, patients have individual levels of susceptibility to steroid-induced psychiatric symptoms that can vary over time. The risk for adverse effects may be elevated based on response to previous courses of glucocorticoid treatment.10 While gender, age, dosage, and duration of treatment influence risk, it is not possible to predict which patients will experience psychiatric effects during a given course of glucocorticoid therapy. Therefore, all patients should be considered to have the potential of developing such effects, and should be monitored during glucocorticoid treatment and withdrawal.
Goals for future research
To help reduce the severity of and cost associated with steroid-induced psychiatric symptoms,5,14 future studies should focus on controlled trials of preventative strategies. In particular, recent advances in genetic mapping may help identify involvement of certain genes or polymorphisms.5 Because current guidelines for the prevention and treatment of steroid-induced psychiatric symptoms are not evidence-based, controlled clinical trials are needed to elucidate the optimal management of such symptoms. There is much interplay between many of the proposed mechanisms of steroid-induced psychiatric symptoms, and future studies can help uncover a deeper understanding of the intricacies of this phenomenon.
CASE CONTINUED
Mrs. N is admitted for altered mental status. Medical workup includes MRI of the brain, MRI of the neck, cardiac echocardiogram, and EEG. There is no evidence of acute structural pathology. She is started on olanzapine, 10 mg/d at bedtime for manic and psychotic symptoms, and is discharged after 5 days. After 1 month, the outpatient psychiatrist gradually decreases and discontinues olanzapine as Mrs. N steadily returns to baseline. One year after discharge, Mrs. N continues to report resolution of her manic and psychotic symptoms.
Bottom Line
Steroids can induce a wide range of psychiatric symptoms, including mania/ hypomania, anxiety, and depression. Initial treatment typically includes tapering or discontinuing the steroid when possible. Other proposed treatments include certain antipsychotics, antidepressants, and other psychotropics, but the supporting evidence is largely anecdotal or based on case studies. Additional research is needed to elucidate the mechanism and treatment recommendations.
Related Resources
- Janes M, Kuster S, Goldson TM, et al. Steroid-induced psychosis. Proc (Bayl Univ Med Cent). 2019;32(4):614-615.
- Mayo Clinic. Prednisone and other corticosteroids. https://www.mayoclinic.org/steroids/art-20045692
Drug Brand Names
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Methylprednisolone injection • Solu-Medrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Phenytoin • Dilantin
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproate • Depakote
Ms. N, age 30, presents to the emergency department for altered mental status, insomnia, and behavioral changes, which she has experienced for 1 week. On evaluation, she grabs a clinician’s hand and details her business ideas and life story with no prompting. Ms. N’s mental status examination is significant for hyperverbal speech with increased rate and volume; tangential thought process; and bright, expanded affect.
One week earlier, Ms. N was hospitalized for sudden-onset chest pain, weakness, and dizziness. She received 45 minutes of cardiopulmonary resuscitation prior to presentation and was found to have a ST-segment elevation myocardial infarction that required emergent left anterior descending coronary artery and right coronary artery percutaneous coronary intervention to place drug-eluting stents. Her recovery was complicated by acute cardiogenic shock, pulmonary edema, and hypoxic respiratory failure. Subsequently, she was intubated, admitted to the ICU, and received high-dose corticosteroids, including IV methylprednisolone, 40 mg every 12 hours, which was tapered prior to discharge. Her husband reports that since Ms. N came home, she has been more talkative and irritable, ruminating about past events, unable to sleep (<1 hour/night), and crying frequently. She has also been endorsing visual and auditory hallucinations, with increased praying and listening to religious music.
The frequent clinical use of steroids necessitates an understanding of these medications’ various adverse effects. The manifestations of steroid-induced psychiatric symptoms are broad and can involve affective, behavioral, and cognitive domains. While the current mechanism is unknown, this phenomenon may be related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity, as well as effects on brain regions such as the hippocampus and amygdala. The best interventions for steroid-induced psychiatric symptoms are awareness and early diagnosis. There are no FDA-approved treatments for steroid-induced psychiatric symptoms; initial measures should include tapering or discontinuing corticosteroids.
In this article, we review the literature on the incidence, characteristics, differential diagnoses, proposed mechanism, risk factors, and proposed treatments of steroid-induced psychiatric symptoms.
A wide range of presentations
Steroid use has increased over the past 2 decades, with 10% of medical and surgical inpatients and 1% to 3% of the general population taking long-term glucocorticoids.1 Even with topical application, steroid therapy is often systemically absorbed, and thus may lead to steroid-induced psychiatric symptoms. The incidence of steroid-induced psychiatric symptoms is difficult to assess because there can be a wide range of reactions that are dose- and time-related. Three reviews of a total of 122 cases reports found that an estimated 5% of patients treated with steroids experience severe psychiatric reactions.1-3
Steroid-induced psychopathology can include mood, behavioral, and/or cognitive impairments. Mania/hypomania is the most common overall psychiatric symptom; the most common mood manifestations are anxiety and depression.4,5 Other possible steroid-induced symptoms include psychosis, dementia, panic disorder, delirium, suicidal thinking and behavior, aggressive behavior, insomnia, agitation, depersonalization, and euphoria.5 The most common cognitive impairment is verbal or declarative memory deficit; others include distractibility and deficits in attention and psychomotor speed.5 These psychiatric symptoms can have a rapid onset, possibly within hours of starting steroids.1 However, studies have reported a median time to onset of 11.5 days; 39% of cases had onset during the first week and 62% within 2 weeks.3,6 After reducing or stopping the steroid, it may take days to weeks before symptoms start to subside.2
What to consider in the differential Dx
Psychiatric symptoms that are induced by steroids can mimic metabolic, neurologic, or toxic disorders. Other factors to consider include drug withdrawal/intoxication, infections, and paraneoplastic syndromes.4,5 Although there is no reported correlation between the location of neurologic lesions and the development of specific psychiatric symptoms, manic symptoms appear most commonly with lesions in the right frontal lobe. 4 Other factors to note include the presence of new-onset psychiatric illnesses such as bipolar, mood, or thought disorders,4 as well as psychosocial stressors that might be contributing to the patient’s presentation.5
Continue to: Proposed mechanisms
Proposed mechanisms
Although the exact mechanism by which steroids induce psychiatric symptoms is unknown, several mechanisms have been proposed. One hypothesis is that steroid-induced psychopathology is related to decreased levels of corticotropin, norepinephrine, and beta-endorphin immunoreactivity.4,5,7 This may explain why many patients with major depressive disorder have elevated cortisol production and/or lack of suppression of cortisol secretion during a dexamethasone stimulation test, and why approximately one-half of patients with Cushing’s disease experience depressive symptoms.8 This is also likely why antipsychotics, which typically reduce cortisol, are efficacious treatments for some steroid-induced psychiatric symptoms.9
Cognitive impairments from steroid use may be related to these agents’ effects on certain brain regions. One such area is the hippocampus, an important mediator in the creation and maintenance of episodic and declarative memories.5,8,9 Acute glucocorticoid use is associated with decreased activity in the left hippocampus, reduced hippocampal glucose metabolism, and reduced cerebral blood flow in the posterior medial temporal lobe.10 Long-term glucocorticoid exposure is associated with smaller hippocampal volume and lower levels of temporal lobe N-acetylaspartate, a marker of neuronal viability.10 Because working memory depends on the prefrontal cortex and declarative memory relies on the hippocampus, deficits in these functions can be attributed to the effect of prolonged glucocorticoid exposure on glucocorticoid or mineralocorticoid receptors in the hippocampus, reduction of hippocampal volume, or elevated glutamate accumulation in that area.11 In addition, high cortisol levels inhibit brain-derived neurotrophic factor, which plays a crucial role in maintaining neural architecture in key brain regions such as the hippocampus and prefrontal cortex.11 There is also a correlation between the duration of prednisone treatment and atrophy of the right amygdala, which is an important regulator of mood and anxiety.11 Both the hippocampus and amygdala have dense collections of glucocorticoid receptors. This may explain why patients who receive high-dose corticosteroids can have reversible atrophy in the hypothalamus and amygdala, leading to deficits in emotional learning and the stress response.
Factors that increase risk
Several factors can increase the risk of steroid-induced psychopathology. The most significant is the dose; higher doses are more likely to produce psychiatric symptoms.1,5 Concurrent use of drugs that increase circulating levels of corticosteroids, such as inhibitors of the cytochrome P450 (CYP) enzyme (eg, clarithromycin), also increases the likelihood of developing psychiatric symptoms.1,5 Risk is also increased in patients with liver or renal dysfunction.1 Cerebral spinal fluid/serum albumin ratio, a marker of blood-brain barrier damage, and low serum complement levels were also reported to be independent risk factors,12 with the thought that increased permeability of the blood-brain barrier may allow hydrophobic steroid molecules to more easily penetrate the CNS, leading to increased neuropsychiatric effects. Hypoalbuminemia is another reported risk factor, perhaps because lower levels of serum albumin are related to higher levels of free and active glucocorticoids, which are normally inactive when bound to albumin.13 There also appears to be an increased prevalence of steroid-induced psychopathology in women, perhaps due to greater propensity in women to seek medical care or a higher prevalence of women with medical disorders that are treated with steroids.5 A previous history of psychiatric disorders may not increase risk.5
Several methods for reducing risk have been proposed, including using a divided-dosing regimens that may lower peak steroid plasma concentrations.13,14 However, the best prevention of steroid-induced psychiatric symptoms are awareness, early diagnosis, and intervention. Studies have suggested that N-methyl-
Treatment options
There are no FDA-approved medications for managing steroid-induced psychiatric symptoms.1,16 Treatment is based on evidence from case reports and a few small case series (Table2-5,17,18).

Continue to: When possible, initial treatment...
When possible, initial treatment should include discontinuing or tapering corticosteroids to <40 mg/d of prednisone-equivalent.1,4,10,18 Most studies have reported rapid reversal of deficits in declarative memory and of hippocampal volume loss once corticosteroids were tapered and discontinued.1,18 One study reported that >90% of patients recovered within 6 weeks, with patients with delirium recovering more quickly (mean: 5.4 days) than those with depression, mania, or psychosis (mean: 19.3 days).3 Another found that the vast majority (92%) of patients treated only with a steroid taper achieved clinical recovery, and 84% recovered with administration of antipsychotics without a steroid taper.3 In this study, all patients who received electroconvulsive therapy (ECT) recovered, as did those who received a steroid taper plus lithium or antipsychotics. Steroid tapering regimens are especially important for patients who have received long-term glucocorticoid treatment. Patients need to be closely monitored for signs of new or increased depression, delirium, or confusion during the taper. If these symptoms occur, the patient should be checked for adrenocortical insufficiency, which can be resolved by re-administering or increasing the dosage of the glucocorticoid.10
Mania. The treatment of mania/hypomania includes mood stabilizers (valproate, lithium, lamotrigine) and antipsychotics (quetiapine, olanzapine, haloperidol).2,4,5,10,14,18 Valproate has been reported to be an effective prophylactic of corticosteroid-induced mania,2 perhaps because it dampens neuronal hyperexcitability by attenuating NMDA receptors, blocking voltage-dependent sodium channels, and inhibiting the synthesis of cortical GABAergic steroids. Starting valproate while continuing corticosteroids (if necessary) may help lessen mania.2 Benzodiazepines also may be useful on a short-term basis.
Depression. Steroid-induced depression may be treated with sertraline or other first-line antidepressants.5,14 Consider ECT for patients with severe depression. Support for the use of antipsychotic medications stems from studies that reported steroids’ role in disrupting dopamine and 5HT2 activity. Lithium also has been used successfully to manage and prevent glucocorticoid-associated affective disorder.10,18 It can be used alone or in combination with selective serotonin reuptake inhibitors to alleviate depressive symptoms.10 Tricyclic antidepressants are generally avoided because their anticholinergic effects can exacerbate or worsen delirium.18 In general, ECT is an effective treatment for persistent and/or unresponsive steroid-induced depression,2,10 but may be difficult to use in patients with serious medical illnesses.
Agitation. Medications that have been proposed for treating steroid-induced agitation include benzodiazepines, haloperidol, and second-generation antipsychotics.5,17
Other considerations. Clinicians, patients, and families should discuss in detail the risks of steroid-induced psychiatric symptoms so an early diagnosis and appropriate intervention can be implemented. Before starting steroids, it is important to review the patient’s current medication list to ensure that steroid treatment is indicated, and to check for potential drug–drug interactions. In addition, the medical condition that is being treated with steroids also needs to be carefully reviewed, because certain illnesses are associated with the development of psychiatric symptoms. 5,10
Continue to: Young children...
Young children (age <6) and older adults appear to be at greater risk for cognitive and memory disturbances from steroid use.10 In addition, patients have individual levels of susceptibility to steroid-induced psychiatric symptoms that can vary over time. The risk for adverse effects may be elevated based on response to previous courses of glucocorticoid treatment.10 While gender, age, dosage, and duration of treatment influence risk, it is not possible to predict which patients will experience psychiatric effects during a given course of glucocorticoid therapy. Therefore, all patients should be considered to have the potential of developing such effects, and should be monitored during glucocorticoid treatment and withdrawal.
Goals for future research
To help reduce the severity of and cost associated with steroid-induced psychiatric symptoms,5,14 future studies should focus on controlled trials of preventative strategies. In particular, recent advances in genetic mapping may help identify involvement of certain genes or polymorphisms.5 Because current guidelines for the prevention and treatment of steroid-induced psychiatric symptoms are not evidence-based, controlled clinical trials are needed to elucidate the optimal management of such symptoms. There is much interplay between many of the proposed mechanisms of steroid-induced psychiatric symptoms, and future studies can help uncover a deeper understanding of the intricacies of this phenomenon.
CASE CONTINUED
Mrs. N is admitted for altered mental status. Medical workup includes MRI of the brain, MRI of the neck, cardiac echocardiogram, and EEG. There is no evidence of acute structural pathology. She is started on olanzapine, 10 mg/d at bedtime for manic and psychotic symptoms, and is discharged after 5 days. After 1 month, the outpatient psychiatrist gradually decreases and discontinues olanzapine as Mrs. N steadily returns to baseline. One year after discharge, Mrs. N continues to report resolution of her manic and psychotic symptoms.
Bottom Line
Steroids can induce a wide range of psychiatric symptoms, including mania/ hypomania, anxiety, and depression. Initial treatment typically includes tapering or discontinuing the steroid when possible. Other proposed treatments include certain antipsychotics, antidepressants, and other psychotropics, but the supporting evidence is largely anecdotal or based on case studies. Additional research is needed to elucidate the mechanism and treatment recommendations.
Related Resources
- Janes M, Kuster S, Goldson TM, et al. Steroid-induced psychosis. Proc (Bayl Univ Med Cent). 2019;32(4):614-615.
- Mayo Clinic. Prednisone and other corticosteroids. https://www.mayoclinic.org/steroids/art-20045692
Drug Brand Names
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Methylprednisolone injection • Solu-Medrol
Olanzapine • Zyprexa
Paroxetine • Paxil
Phenytoin • Dilantin
Quetiapine • Seroquel
Risperidone • Risperdal
Sertraline • Zoloft
Valproate • Depakote
1. Dubovsky AN, Arvikar S, Stern TA, et al. The neuropsychiatric complications of glucocorticoid use: steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
2. Roxanas MG, Hunt GE. Rapid reversal of corticosteroid-induced mania with sodium valproate: a case series of 20 patients. Psychosomatics. 2012;53(6):575-581.
3. Lewis DA, Smith RE. Steroid‐induced psychiatric syndromes. A report of 14 cases and a review of the literature. J Affect Disord. 1983;5(4):319-332.
4. Warren KN, Katakam J, Espiridion ED. Acute-onset mania in a patient with non-small cell lung cancer. Cureus. 2019;11(8):e5436.
5. Kenna HA, Poon AW, de los Angeles CP, et al. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549-560.
6. Ling MH, Perry PJ, Tsuang MT. Side effects of corticosteroid therapy. Psychiatric aspects. Arch Gen. Psychiatry. 1981;38(4):471-477.
7. Ularntinon S, Tzuang D, Dahl G, et al. Concurrent treatment of steroid-related mood and psychotic symptoms with risperidone. Pediatrics. 2010;125(5):e1241-e1245.
8. Pokladinkova J, Meyboom RH, Vlcek J, et al. Intranasally administered corticosteroids and neuropsychiatric disturbances: a review of the international pharmacovigilance programme of the World Health Organization. Ann Allergy Asthma Immunol. 2008;101(1):67-73.
9. Walker EF, Trotman HD, Pearce BD, et al. Cortisol levels and risk for psychosis: initial findings from the North American prodrome longitudinal study. Biol Psychiatry. 2013;74(6):410-417.
10. Wolkowitz OM, Reus UI. Treatment of depression with antiglucocorticoid drugs. Psychosom Med. 1999;61(5):698-711.
11. Judd LL, Schettler PJ, Brown ES, et al. Adverse consequences of glucocorticoid medication: psychological, cognitive, and behavioral effects. Am J Psychiatry. 2014;171(10):1045-1051.
12. Appenzeller S, Cendes F, Costallat LT. Acute psychosis in systemic lupus erythematosus. Rheumatol Int. 2008;28(3):237-243.
13. Glynne-Jones R, Vernon CC, Bell G. Is steroid psychosis preventable by divided doses? Lancet. 1986;2(8520):1404.
14. Ismail MF, Lavelle C, Cassidy EM. Steroid-induced mental disorders in cancer patients: a systematic review. Future Oncol. 2017;13(29):2719-2731.
15. Magariños AM, McEwen BS. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience. 1995;69(1):89-98.
16. Brown BS, Stuard G, Liggin JDM, et al. Effect of phenytoin on mood and declarative memory during prescription corticosteroid therapy. Biol Psychiatry. 2005;57(5):543-548.
17. Desai S, Khanani S, Shad MU, et al. Attenutation of amygdala atrophy with lamotrigine in patients receiving corticosteroid therapy. J Clin Psychopharmacol. 2009;29(3):284-287.
18. Gable M, Depry D. Sustained corticosteroid-induced mania and psychosis despite cessation: a case study and brief literature review. Int J Psychiatry Med. 2015;50(4):398-404.
1. Dubovsky AN, Arvikar S, Stern TA, et al. The neuropsychiatric complications of glucocorticoid use: steroid psychosis revisited. Psychosomatics. 2012;53(2):103-115.
2. Roxanas MG, Hunt GE. Rapid reversal of corticosteroid-induced mania with sodium valproate: a case series of 20 patients. Psychosomatics. 2012;53(6):575-581.
3. Lewis DA, Smith RE. Steroid‐induced psychiatric syndromes. A report of 14 cases and a review of the literature. J Affect Disord. 1983;5(4):319-332.
4. Warren KN, Katakam J, Espiridion ED. Acute-onset mania in a patient with non-small cell lung cancer. Cureus. 2019;11(8):e5436.
5. Kenna HA, Poon AW, de los Angeles CP, et al. Psychiatric complications of treatment with corticosteroids: review with case report. Psychiatry Clin Neurosci. 2011;65(6):549-560.
6. Ling MH, Perry PJ, Tsuang MT. Side effects of corticosteroid therapy. Psychiatric aspects. Arch Gen. Psychiatry. 1981;38(4):471-477.
7. Ularntinon S, Tzuang D, Dahl G, et al. Concurrent treatment of steroid-related mood and psychotic symptoms with risperidone. Pediatrics. 2010;125(5):e1241-e1245.
8. Pokladinkova J, Meyboom RH, Vlcek J, et al. Intranasally administered corticosteroids and neuropsychiatric disturbances: a review of the international pharmacovigilance programme of the World Health Organization. Ann Allergy Asthma Immunol. 2008;101(1):67-73.
9. Walker EF, Trotman HD, Pearce BD, et al. Cortisol levels and risk for psychosis: initial findings from the North American prodrome longitudinal study. Biol Psychiatry. 2013;74(6):410-417.
10. Wolkowitz OM, Reus UI. Treatment of depression with antiglucocorticoid drugs. Psychosom Med. 1999;61(5):698-711.
11. Judd LL, Schettler PJ, Brown ES, et al. Adverse consequences of glucocorticoid medication: psychological, cognitive, and behavioral effects. Am J Psychiatry. 2014;171(10):1045-1051.
12. Appenzeller S, Cendes F, Costallat LT. Acute psychosis in systemic lupus erythematosus. Rheumatol Int. 2008;28(3):237-243.
13. Glynne-Jones R, Vernon CC, Bell G. Is steroid psychosis preventable by divided doses? Lancet. 1986;2(8520):1404.
14. Ismail MF, Lavelle C, Cassidy EM. Steroid-induced mental disorders in cancer patients: a systematic review. Future Oncol. 2017;13(29):2719-2731.
15. Magariños AM, McEwen BS. Stress-induced atrophy of apical dendrites of hippocampal CA3c neurons: involvement of glucocorticoid secretion and excitatory amino acid receptors. Neuroscience. 1995;69(1):89-98.
16. Brown BS, Stuard G, Liggin JDM, et al. Effect of phenytoin on mood and declarative memory during prescription corticosteroid therapy. Biol Psychiatry. 2005;57(5):543-548.
17. Desai S, Khanani S, Shad MU, et al. Attenutation of amygdala atrophy with lamotrigine in patients receiving corticosteroid therapy. J Clin Psychopharmacol. 2009;29(3):284-287.
18. Gable M, Depry D. Sustained corticosteroid-induced mania and psychosis despite cessation: a case study and brief literature review. Int J Psychiatry Med. 2015;50(4):398-404.
Rhythm and blues: Using heart rate to diagnose depression
Depression might be a disorder of the brain, but its harms aren’t confined to the cranium. Prolonged depression has been linked with a slew of health problems, from impaired immune function to gastrointestinal dysfunction. It’s also been linked with cardiovascular disease (CVD), even increasing the risk for heart attack and a disrupted heart rate. Now, researchers are exploring whether heart function could be a valuable biomarker in informing depression diagnosis and treatment.
Major depressive disorder has proved difficult to diagnose and treat, and biomarkers that indicate a depressive episode or suggest specific interventions would be an attractive solution to its clinically nebulous nature.
Currently, diagnosing depression relies on the patients effectively communicating their symptoms. If the patient does receive a diagnosis, treating it remains a matter of trial and error. It takes weeks to know whether a treatment is working, and in only one-third of cases does the condition go into remission after the patient is initially prescribed an antidepressant. Even after successful treatment, it’s challenging to identify who might be at risk for relapse, and when. Research now shows that cardiac biomarkers may be a way improve this picture. Clinicians could use changes in heart rate to both inform depression diagnosis and gauge a patient’s predicted response to treatment.
Given the increased risk for CVD among people with depression and the link between heart rate changes and CVD risk, these biomarkers could have implications for heart health, too. “We need more than just the current toolkit,” said Amit Shah, MD, a cardiologist and assistant professor of epidemiology at Emory University, Atlanta. “Ultimately, what we’re trying to do is develop interventions not only for depression but also for the associated physical health problems related to depression, in particular, cardiovascular disease,” he said. These overlapping interests – and the prospect of clinically considering both conditions in tandem – mean this research is “really worth its weight in gold,” added Dr. Shah.
The data on heart rate biomarkers
Patients with depression are often found to have lower heart rate variability (HRV) and higher heart rates. Scientists don’t know the mechanisms underpinning this relationship but think changes in the autonomic nervous system during depression, as well as stress generally, have a role.
Rébecca Robillard, PhD, is the head scientist of the Clinical Sleep Research Platform at the Royal’s Institute of Mental Health Research, Ottawa, Ont. In a 2019 study published in BMC Psychiatry, Dr. Robillard’s team used electrocardiogram recordings from sleep studies to see whether heart rate abnormalities were associated with depression. Using a profiling algorithm to analyze heart rate and HRV data, the team identified persons with depression with 80% accuracy among 174 people with sleep complaints.
Dr. Robillard said.
In another study, Stephan Claes, MD, PhD, psychiatrist and professor of psychiatry at Katholieke Universiteit Leuven, Belgium, and his group tested the biomarker potential of heart rate and HRV data that were continuously recorded over several days. They too used an algorithm to distinguish 16 people with treatment-resistant depression from 16 without depression. Within the depression group, they used the algorithm to distinguish patients who had received ketamine treatment from those who had not.
The algorithm could differentiate between the depressed and nondepressed groups with 90% accuracy. Those with depression had higher overall heart rates, particularly at night, and lower HRV. Dr. Claes noted that, unlike in other studies, “the most reliable parameter that we had for this prediction was the heart rate, not the HRV.” After treatment, heart rates improved, but HRV remained the same.
Although their study has not yet been peer reviewed and more research is needed, Dr. Claes said that increased heart rate, especially during the night, could eventually serve as a warning sign of depression relapse. “That would allow a quicker referral to care and better care because of earlier intervention,” he said.
Finding a signal amid the noise
But heart rate and HRV aren’t foolproof biomarkers. Some studies have found that antidepressant use lowers HRV and that HRV changes aren’t unique to depression. There’s the added complication that depression often overlaps with other mental disorders.
“I think we’ve been very disappointed about the success of using particular biomarkers for particular disorders, because the majority of mental disorders are very heterogeneous,” said Andrew Kemp, PhD, psychology professor at Swansea University, Swansea, Wales. “A particular biomarker will, at the end of the day, be just one particular aspect of the overall profile that clinicians will have on particular individuals.”
The clinical utility of a heart rate–depression connection may go both ways.
For instance, depression could serve as a warning sign for atrial fibrillation, according to research from Parveen K. Garg, MD, associate professor of clinical medicine at the University of Southern California, Los Angeles. In a study involving more than 6,000 people, Dr. Garg showed that higher scores on depression scales correlated with a higher risk for the occurrence of atrial fibrillation over a follow-up period of about 13 years.
Depression is associated with other heart conditions as well. “A lot of data seem to suggest that just the presence of depression can increase risk for a whole range of cardiovascular problems,” said Dr. Garg. Epidemiologic studies have found associations between depression and the development of coronary heart disease and a modest increased risk for stroke.
“Things going on in your brain also have effects on the rest of your body,” said Dr. Garg. “Just recognizing this link, that maybe mental illness has an effect on other illnesses or diseases that can affect other parts of your body – I think that’s something we can share now.”
A version of this article first appeared on Medscape.com.
Depression might be a disorder of the brain, but its harms aren’t confined to the cranium. Prolonged depression has been linked with a slew of health problems, from impaired immune function to gastrointestinal dysfunction. It’s also been linked with cardiovascular disease (CVD), even increasing the risk for heart attack and a disrupted heart rate. Now, researchers are exploring whether heart function could be a valuable biomarker in informing depression diagnosis and treatment.
Major depressive disorder has proved difficult to diagnose and treat, and biomarkers that indicate a depressive episode or suggest specific interventions would be an attractive solution to its clinically nebulous nature.
Currently, diagnosing depression relies on the patients effectively communicating their symptoms. If the patient does receive a diagnosis, treating it remains a matter of trial and error. It takes weeks to know whether a treatment is working, and in only one-third of cases does the condition go into remission after the patient is initially prescribed an antidepressant. Even after successful treatment, it’s challenging to identify who might be at risk for relapse, and when. Research now shows that cardiac biomarkers may be a way improve this picture. Clinicians could use changes in heart rate to both inform depression diagnosis and gauge a patient’s predicted response to treatment.
Given the increased risk for CVD among people with depression and the link between heart rate changes and CVD risk, these biomarkers could have implications for heart health, too. “We need more than just the current toolkit,” said Amit Shah, MD, a cardiologist and assistant professor of epidemiology at Emory University, Atlanta. “Ultimately, what we’re trying to do is develop interventions not only for depression but also for the associated physical health problems related to depression, in particular, cardiovascular disease,” he said. These overlapping interests – and the prospect of clinically considering both conditions in tandem – mean this research is “really worth its weight in gold,” added Dr. Shah.
The data on heart rate biomarkers
Patients with depression are often found to have lower heart rate variability (HRV) and higher heart rates. Scientists don’t know the mechanisms underpinning this relationship but think changes in the autonomic nervous system during depression, as well as stress generally, have a role.
Rébecca Robillard, PhD, is the head scientist of the Clinical Sleep Research Platform at the Royal’s Institute of Mental Health Research, Ottawa, Ont. In a 2019 study published in BMC Psychiatry, Dr. Robillard’s team used electrocardiogram recordings from sleep studies to see whether heart rate abnormalities were associated with depression. Using a profiling algorithm to analyze heart rate and HRV data, the team identified persons with depression with 80% accuracy among 174 people with sleep complaints.
Dr. Robillard said.
In another study, Stephan Claes, MD, PhD, psychiatrist and professor of psychiatry at Katholieke Universiteit Leuven, Belgium, and his group tested the biomarker potential of heart rate and HRV data that were continuously recorded over several days. They too used an algorithm to distinguish 16 people with treatment-resistant depression from 16 without depression. Within the depression group, they used the algorithm to distinguish patients who had received ketamine treatment from those who had not.
The algorithm could differentiate between the depressed and nondepressed groups with 90% accuracy. Those with depression had higher overall heart rates, particularly at night, and lower HRV. Dr. Claes noted that, unlike in other studies, “the most reliable parameter that we had for this prediction was the heart rate, not the HRV.” After treatment, heart rates improved, but HRV remained the same.
Although their study has not yet been peer reviewed and more research is needed, Dr. Claes said that increased heart rate, especially during the night, could eventually serve as a warning sign of depression relapse. “That would allow a quicker referral to care and better care because of earlier intervention,” he said.
Finding a signal amid the noise
But heart rate and HRV aren’t foolproof biomarkers. Some studies have found that antidepressant use lowers HRV and that HRV changes aren’t unique to depression. There’s the added complication that depression often overlaps with other mental disorders.
“I think we’ve been very disappointed about the success of using particular biomarkers for particular disorders, because the majority of mental disorders are very heterogeneous,” said Andrew Kemp, PhD, psychology professor at Swansea University, Swansea, Wales. “A particular biomarker will, at the end of the day, be just one particular aspect of the overall profile that clinicians will have on particular individuals.”
The clinical utility of a heart rate–depression connection may go both ways.
For instance, depression could serve as a warning sign for atrial fibrillation, according to research from Parveen K. Garg, MD, associate professor of clinical medicine at the University of Southern California, Los Angeles. In a study involving more than 6,000 people, Dr. Garg showed that higher scores on depression scales correlated with a higher risk for the occurrence of atrial fibrillation over a follow-up period of about 13 years.
Depression is associated with other heart conditions as well. “A lot of data seem to suggest that just the presence of depression can increase risk for a whole range of cardiovascular problems,” said Dr. Garg. Epidemiologic studies have found associations between depression and the development of coronary heart disease and a modest increased risk for stroke.
“Things going on in your brain also have effects on the rest of your body,” said Dr. Garg. “Just recognizing this link, that maybe mental illness has an effect on other illnesses or diseases that can affect other parts of your body – I think that’s something we can share now.”
A version of this article first appeared on Medscape.com.
Depression might be a disorder of the brain, but its harms aren’t confined to the cranium. Prolonged depression has been linked with a slew of health problems, from impaired immune function to gastrointestinal dysfunction. It’s also been linked with cardiovascular disease (CVD), even increasing the risk for heart attack and a disrupted heart rate. Now, researchers are exploring whether heart function could be a valuable biomarker in informing depression diagnosis and treatment.
Major depressive disorder has proved difficult to diagnose and treat, and biomarkers that indicate a depressive episode or suggest specific interventions would be an attractive solution to its clinically nebulous nature.
Currently, diagnosing depression relies on the patients effectively communicating their symptoms. If the patient does receive a diagnosis, treating it remains a matter of trial and error. It takes weeks to know whether a treatment is working, and in only one-third of cases does the condition go into remission after the patient is initially prescribed an antidepressant. Even after successful treatment, it’s challenging to identify who might be at risk for relapse, and when. Research now shows that cardiac biomarkers may be a way improve this picture. Clinicians could use changes in heart rate to both inform depression diagnosis and gauge a patient’s predicted response to treatment.
Given the increased risk for CVD among people with depression and the link between heart rate changes and CVD risk, these biomarkers could have implications for heart health, too. “We need more than just the current toolkit,” said Amit Shah, MD, a cardiologist and assistant professor of epidemiology at Emory University, Atlanta. “Ultimately, what we’re trying to do is develop interventions not only for depression but also for the associated physical health problems related to depression, in particular, cardiovascular disease,” he said. These overlapping interests – and the prospect of clinically considering both conditions in tandem – mean this research is “really worth its weight in gold,” added Dr. Shah.
The data on heart rate biomarkers
Patients with depression are often found to have lower heart rate variability (HRV) and higher heart rates. Scientists don’t know the mechanisms underpinning this relationship but think changes in the autonomic nervous system during depression, as well as stress generally, have a role.
Rébecca Robillard, PhD, is the head scientist of the Clinical Sleep Research Platform at the Royal’s Institute of Mental Health Research, Ottawa, Ont. In a 2019 study published in BMC Psychiatry, Dr. Robillard’s team used electrocardiogram recordings from sleep studies to see whether heart rate abnormalities were associated with depression. Using a profiling algorithm to analyze heart rate and HRV data, the team identified persons with depression with 80% accuracy among 174 people with sleep complaints.
Dr. Robillard said.
In another study, Stephan Claes, MD, PhD, psychiatrist and professor of psychiatry at Katholieke Universiteit Leuven, Belgium, and his group tested the biomarker potential of heart rate and HRV data that were continuously recorded over several days. They too used an algorithm to distinguish 16 people with treatment-resistant depression from 16 without depression. Within the depression group, they used the algorithm to distinguish patients who had received ketamine treatment from those who had not.
The algorithm could differentiate between the depressed and nondepressed groups with 90% accuracy. Those with depression had higher overall heart rates, particularly at night, and lower HRV. Dr. Claes noted that, unlike in other studies, “the most reliable parameter that we had for this prediction was the heart rate, not the HRV.” After treatment, heart rates improved, but HRV remained the same.
Although their study has not yet been peer reviewed and more research is needed, Dr. Claes said that increased heart rate, especially during the night, could eventually serve as a warning sign of depression relapse. “That would allow a quicker referral to care and better care because of earlier intervention,” he said.
Finding a signal amid the noise
But heart rate and HRV aren’t foolproof biomarkers. Some studies have found that antidepressant use lowers HRV and that HRV changes aren’t unique to depression. There’s the added complication that depression often overlaps with other mental disorders.
“I think we’ve been very disappointed about the success of using particular biomarkers for particular disorders, because the majority of mental disorders are very heterogeneous,” said Andrew Kemp, PhD, psychology professor at Swansea University, Swansea, Wales. “A particular biomarker will, at the end of the day, be just one particular aspect of the overall profile that clinicians will have on particular individuals.”
The clinical utility of a heart rate–depression connection may go both ways.
For instance, depression could serve as a warning sign for atrial fibrillation, according to research from Parveen K. Garg, MD, associate professor of clinical medicine at the University of Southern California, Los Angeles. In a study involving more than 6,000 people, Dr. Garg showed that higher scores on depression scales correlated with a higher risk for the occurrence of atrial fibrillation over a follow-up period of about 13 years.
Depression is associated with other heart conditions as well. “A lot of data seem to suggest that just the presence of depression can increase risk for a whole range of cardiovascular problems,” said Dr. Garg. Epidemiologic studies have found associations between depression and the development of coronary heart disease and a modest increased risk for stroke.
“Things going on in your brain also have effects on the rest of your body,” said Dr. Garg. “Just recognizing this link, that maybe mental illness has an effect on other illnesses or diseases that can affect other parts of your body – I think that’s something we can share now.”
A version of this article first appeared on Medscape.com.
Child abuse tied to persistent inflammation in later life
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New expert guidance on ketamine for resistant depression
An international panel of mood disorder experts has published guidance on how to safely and effectively use ketamine and esketamine to treat adults with treatment-resistant depression (TRD).
“Ketamine and esketamine are the first rapid-onset treatments for adults with TRD, and there was an international need for best-practice guidance on the deft and safe implementation of ketamine and esketamine at the point of care, as none previously existed,” first author Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, said in an interview.
“This need has only been amplified by the significant increase in the number of clinics and centers providing this treatment,” added Dr. McIntyre, head of the mood disorders psychopharmacology unit.
Their article was published online March 17 in the American Journal of Psychiatry.
Insufficient evidence of long-term efficacy
As reported by this news organization, the U.S. Food and Drug Administration (FDA) approved esketamine nasal spray (Spravato) for TRD in March 2019.
In August 2020, the FDA updated the approval to include adults with major depression and suicidal thoughts or actions.
To provide clinical guidance, Dr. McIntyre and colleagues synthesized the available literature on the efficacy, safety, and tolerability of ketamine and esketamine for TRD.
The evidence, they note, supports the rapid-onset (within 1-2 days) efficacy of esketamine and ketamine in TRD.
The strongest evidence of efficacy is for intranasal esketamine and intravenous ketamine. There is insufficient evidence for oral, subcutaneous, or intramuscular ketamine for TRD, they report.
Intranasal esketamine demonstrates efficacy, safety, and tolerability for up to 1 year in adults with TRD. Evidence for long-term efficacy, safety, and tolerability of intravenous ketamine for patients with TRD is insufficient, the group notes.
They also note that esketamine is approved in the United States for major depression in association with suicidal ideation or behavior and that it has been proven to reduce suicide completion.
Safety concerns with ketamine and esketamine identified in the literature include, but are not limited to, psychiatric, neurologic/cognitive, genitourinary, and hemodynamic effects.
Implementation checklist
The group has developed an “implementation checklist” for use of ketamine/esketamine in clinical practice.
Starting with patient selection, they note that appropriate patients are those with a confirmed diagnosis of TRD for whom psychosis and other conditions that would significantly affect the risk-benefit ratio have been ruled out.
They suggest that a physical examination and monitoring of vital signs be undertaken during treatment and during posttreatment surveillance. A urine drug screen should be considered if appropriate.
The group advises that esketamine and ketamine be administered only in settings with multidisciplinary personnel, including, but not limited to, those with expertise in the assessment of mood disorders.
Clinics should be equipped with appropriate cardiorespiratory monitoring and be capable of psychiatric assessment of dissociation and psychotomimetic effects.
Depressive symptoms should be measured, and the authors suggest assessing for anxiety, cognitive function, well-being, and psychosocial function.
Patients should be monitored immediately after treatment to ensure cardiorespiratory stability, clear sensorium, and attenuation of dissociative and psychotomimetic effects.
The United States and some other countries require a risk evaluation and mitigation strategy (REMS) when administering esketamine. Regarding the REMS, it is advised that all patients be monitored for a minimum of 2 hours before discharge.
Patients should arrange for reliable transportation for each appointment, and they should be advised not to operate motor vehicles or hazardous machinery without at least one night of sleep.
, only worsened by COVID-19,” said Dr. McIntyre.
“Clinicians of different professional backgrounds have been interested in ketamine/esketamine, and we are extraordinarily pleased to see our international guidelines published,” he added.
‘Extremely useful’
Reached for comment, Alan Schatzberg, MD, professor of psychiatry and behavioral sciences at Stanford (Calif.) University, said this document “puts a lot of information in one place as far as what we know and what we don’t know right now, and that’s helpful. I think it’s an attempt to have a kind of a somewhat objective review of the literature, and it’s in a good journal.”
The article, Dr. Schatzberg added, “could be extremely useful for someone who is considering whether ketamine is useful for a patient or what they can tell a patient about ketamine, that is, about how long they might need, is it going to work, will it continue to work, and the level of data we have either on benefits or side effects.”
The research had no specific funding. The original article contains a complete list of author disclosures. Dr. Schatzberg has received grant support from Janssen; has served as a consultant for Alkermes, Avanir, Brain Resource, Bracket, Compass, Delpor, Epiodyne, GLG, Jazz, Janssen Pharmaceuticals, Lundbeck/Takeda, McKinsey and Company, Merck, Myriad Genetics, Neuronetics, Owl Analytics, Pfizer, Sage, Sunovion, and Xhale; holds equity in Corcept (cofounder), Delpor, Dermira, Epiodyne, Gilead, Incyte Genetics, Intersect ENT, Madrigal, Merck, Owl Analytics, Seattle Genetics, Titan, and Xhale; and is listed as an inventor on patents for pharmacogenetics and antiglucocorticoid use in the prediction of antidepressant response.
A version of this article first appeared on Medscape.com.
An international panel of mood disorder experts has published guidance on how to safely and effectively use ketamine and esketamine to treat adults with treatment-resistant depression (TRD).
“Ketamine and esketamine are the first rapid-onset treatments for adults with TRD, and there was an international need for best-practice guidance on the deft and safe implementation of ketamine and esketamine at the point of care, as none previously existed,” first author Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, said in an interview.
“This need has only been amplified by the significant increase in the number of clinics and centers providing this treatment,” added Dr. McIntyre, head of the mood disorders psychopharmacology unit.
Their article was published online March 17 in the American Journal of Psychiatry.
Insufficient evidence of long-term efficacy
As reported by this news organization, the U.S. Food and Drug Administration (FDA) approved esketamine nasal spray (Spravato) for TRD in March 2019.
In August 2020, the FDA updated the approval to include adults with major depression and suicidal thoughts or actions.
To provide clinical guidance, Dr. McIntyre and colleagues synthesized the available literature on the efficacy, safety, and tolerability of ketamine and esketamine for TRD.
The evidence, they note, supports the rapid-onset (within 1-2 days) efficacy of esketamine and ketamine in TRD.
The strongest evidence of efficacy is for intranasal esketamine and intravenous ketamine. There is insufficient evidence for oral, subcutaneous, or intramuscular ketamine for TRD, they report.
Intranasal esketamine demonstrates efficacy, safety, and tolerability for up to 1 year in adults with TRD. Evidence for long-term efficacy, safety, and tolerability of intravenous ketamine for patients with TRD is insufficient, the group notes.
They also note that esketamine is approved in the United States for major depression in association with suicidal ideation or behavior and that it has been proven to reduce suicide completion.
Safety concerns with ketamine and esketamine identified in the literature include, but are not limited to, psychiatric, neurologic/cognitive, genitourinary, and hemodynamic effects.
Implementation checklist
The group has developed an “implementation checklist” for use of ketamine/esketamine in clinical practice.
Starting with patient selection, they note that appropriate patients are those with a confirmed diagnosis of TRD for whom psychosis and other conditions that would significantly affect the risk-benefit ratio have been ruled out.
They suggest that a physical examination and monitoring of vital signs be undertaken during treatment and during posttreatment surveillance. A urine drug screen should be considered if appropriate.
The group advises that esketamine and ketamine be administered only in settings with multidisciplinary personnel, including, but not limited to, those with expertise in the assessment of mood disorders.
Clinics should be equipped with appropriate cardiorespiratory monitoring and be capable of psychiatric assessment of dissociation and psychotomimetic effects.
Depressive symptoms should be measured, and the authors suggest assessing for anxiety, cognitive function, well-being, and psychosocial function.
Patients should be monitored immediately after treatment to ensure cardiorespiratory stability, clear sensorium, and attenuation of dissociative and psychotomimetic effects.
The United States and some other countries require a risk evaluation and mitigation strategy (REMS) when administering esketamine. Regarding the REMS, it is advised that all patients be monitored for a minimum of 2 hours before discharge.
Patients should arrange for reliable transportation for each appointment, and they should be advised not to operate motor vehicles or hazardous machinery without at least one night of sleep.
, only worsened by COVID-19,” said Dr. McIntyre.
“Clinicians of different professional backgrounds have been interested in ketamine/esketamine, and we are extraordinarily pleased to see our international guidelines published,” he added.
‘Extremely useful’
Reached for comment, Alan Schatzberg, MD, professor of psychiatry and behavioral sciences at Stanford (Calif.) University, said this document “puts a lot of information in one place as far as what we know and what we don’t know right now, and that’s helpful. I think it’s an attempt to have a kind of a somewhat objective review of the literature, and it’s in a good journal.”
The article, Dr. Schatzberg added, “could be extremely useful for someone who is considering whether ketamine is useful for a patient or what they can tell a patient about ketamine, that is, about how long they might need, is it going to work, will it continue to work, and the level of data we have either on benefits or side effects.”
The research had no specific funding. The original article contains a complete list of author disclosures. Dr. Schatzberg has received grant support from Janssen; has served as a consultant for Alkermes, Avanir, Brain Resource, Bracket, Compass, Delpor, Epiodyne, GLG, Jazz, Janssen Pharmaceuticals, Lundbeck/Takeda, McKinsey and Company, Merck, Myriad Genetics, Neuronetics, Owl Analytics, Pfizer, Sage, Sunovion, and Xhale; holds equity in Corcept (cofounder), Delpor, Dermira, Epiodyne, Gilead, Incyte Genetics, Intersect ENT, Madrigal, Merck, Owl Analytics, Seattle Genetics, Titan, and Xhale; and is listed as an inventor on patents for pharmacogenetics and antiglucocorticoid use in the prediction of antidepressant response.
A version of this article first appeared on Medscape.com.
An international panel of mood disorder experts has published guidance on how to safely and effectively use ketamine and esketamine to treat adults with treatment-resistant depression (TRD).
“Ketamine and esketamine are the first rapid-onset treatments for adults with TRD, and there was an international need for best-practice guidance on the deft and safe implementation of ketamine and esketamine at the point of care, as none previously existed,” first author Roger McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, said in an interview.
“This need has only been amplified by the significant increase in the number of clinics and centers providing this treatment,” added Dr. McIntyre, head of the mood disorders psychopharmacology unit.
Their article was published online March 17 in the American Journal of Psychiatry.
Insufficient evidence of long-term efficacy
As reported by this news organization, the U.S. Food and Drug Administration (FDA) approved esketamine nasal spray (Spravato) for TRD in March 2019.
In August 2020, the FDA updated the approval to include adults with major depression and suicidal thoughts or actions.
To provide clinical guidance, Dr. McIntyre and colleagues synthesized the available literature on the efficacy, safety, and tolerability of ketamine and esketamine for TRD.
The evidence, they note, supports the rapid-onset (within 1-2 days) efficacy of esketamine and ketamine in TRD.
The strongest evidence of efficacy is for intranasal esketamine and intravenous ketamine. There is insufficient evidence for oral, subcutaneous, or intramuscular ketamine for TRD, they report.
Intranasal esketamine demonstrates efficacy, safety, and tolerability for up to 1 year in adults with TRD. Evidence for long-term efficacy, safety, and tolerability of intravenous ketamine for patients with TRD is insufficient, the group notes.
They also note that esketamine is approved in the United States for major depression in association with suicidal ideation or behavior and that it has been proven to reduce suicide completion.
Safety concerns with ketamine and esketamine identified in the literature include, but are not limited to, psychiatric, neurologic/cognitive, genitourinary, and hemodynamic effects.
Implementation checklist
The group has developed an “implementation checklist” for use of ketamine/esketamine in clinical practice.
Starting with patient selection, they note that appropriate patients are those with a confirmed diagnosis of TRD for whom psychosis and other conditions that would significantly affect the risk-benefit ratio have been ruled out.
They suggest that a physical examination and monitoring of vital signs be undertaken during treatment and during posttreatment surveillance. A urine drug screen should be considered if appropriate.
The group advises that esketamine and ketamine be administered only in settings with multidisciplinary personnel, including, but not limited to, those with expertise in the assessment of mood disorders.
Clinics should be equipped with appropriate cardiorespiratory monitoring and be capable of psychiatric assessment of dissociation and psychotomimetic effects.
Depressive symptoms should be measured, and the authors suggest assessing for anxiety, cognitive function, well-being, and psychosocial function.
Patients should be monitored immediately after treatment to ensure cardiorespiratory stability, clear sensorium, and attenuation of dissociative and psychotomimetic effects.
The United States and some other countries require a risk evaluation and mitigation strategy (REMS) when administering esketamine. Regarding the REMS, it is advised that all patients be monitored for a minimum of 2 hours before discharge.
Patients should arrange for reliable transportation for each appointment, and they should be advised not to operate motor vehicles or hazardous machinery without at least one night of sleep.
, only worsened by COVID-19,” said Dr. McIntyre.
“Clinicians of different professional backgrounds have been interested in ketamine/esketamine, and we are extraordinarily pleased to see our international guidelines published,” he added.
‘Extremely useful’
Reached for comment, Alan Schatzberg, MD, professor of psychiatry and behavioral sciences at Stanford (Calif.) University, said this document “puts a lot of information in one place as far as what we know and what we don’t know right now, and that’s helpful. I think it’s an attempt to have a kind of a somewhat objective review of the literature, and it’s in a good journal.”
The article, Dr. Schatzberg added, “could be extremely useful for someone who is considering whether ketamine is useful for a patient or what they can tell a patient about ketamine, that is, about how long they might need, is it going to work, will it continue to work, and the level of data we have either on benefits or side effects.”
The research had no specific funding. The original article contains a complete list of author disclosures. Dr. Schatzberg has received grant support from Janssen; has served as a consultant for Alkermes, Avanir, Brain Resource, Bracket, Compass, Delpor, Epiodyne, GLG, Jazz, Janssen Pharmaceuticals, Lundbeck/Takeda, McKinsey and Company, Merck, Myriad Genetics, Neuronetics, Owl Analytics, Pfizer, Sage, Sunovion, and Xhale; holds equity in Corcept (cofounder), Delpor, Dermira, Epiodyne, Gilead, Incyte Genetics, Intersect ENT, Madrigal, Merck, Owl Analytics, Seattle Genetics, Titan, and Xhale; and is listed as an inventor on patents for pharmacogenetics and antiglucocorticoid use in the prediction of antidepressant response.
A version of this article first appeared on Medscape.com.