User login
Diabetes decision tool yields ‘modest’ benefit in low-resource clinics
a randomized trial in China showed.
The tool required clinicians to enter patient data into a computer in order to generate individualized treatment recommendations, adding to their administrative burdens. It also couldn’t tackle patients’ problems with access and affordability of medications.
Nevertheless, the model could curtail physician burnout and improve the quality of care in primary care clinics with limited resources, the researchers said in a paper published in the Annals of Internal Medicine.
They concluded that the findings support “widespread adoption” of the model in China and other low- or middle-income countries where diabetes is on the rise.
Co–principal investigator Jiang He, MD, PhD, chair of epidemiology at Tulane University, New Orleans, said the findings could apply to federally qualified health care (FQHC) clinics that treat underserved patients in the United States.
“At many FQHC clinics, nurse practitioners have to take care of patients with multiple chronic disease conditions. Team-based care with a computerized clinical decision support system will help them and improve patient care,” Dr. He said.
Small improvements
To conduct the trial, called Diabetes Complication Control in Community Clinics (D4C), Dr. He and colleagues randomly assigned 19 out of the 38 community health centers in Xiamen, China, to have a clinical decision support tool installed on the computers of primary care physicians and health coaches.
Starting in October 2016 the researchers recruited 11,132 patients aged 50 and older with uncontrolled diabetes and at least one comorbid condition, with 5,475 patients receiving team-based care with the CDSS and the remainder receiving team-based care alone.
The CDSS generated individualized risk factor summaries and treatment recommendations, including prescriptions based on Chinese and U.S. clinical guidelines. It incorporated data on patients’ insurance plans and local availability of drugs.
At all centers, primary care physicians received training in managing glycemia, blood pressure, and lipids. Nurses were certified as health coaches after receiving training on nutrition, lifestyle changes, and medication adherence. Patients met with their coaches for half an hour every 3 months, and diabetes specialists visited each clinic monthly for team meetings and consultations.
After 18 months, patients undergoing team-based care alone lowered their hemoglobin A1c by 0.6 percentage points (95% confidence interval, –0.7 to –0.5 percentage points), LDL cholesterol by 12.5 mg/dL (95% CI, –13.6 to –11.3 mg/dL), and systolic blood pressure by 7.5 mm Hg (95% CI, –8.4 to –6.6 mm Hg).
The group whose care teams used the CDSS further reduced A1c by 0.2 percentage points (95% CI, –0.3 to –0.1 percentage points), LDL cholesterol by 6.5 mg/dL (95% CI, –8.3 to -4.6 mg/dL), and blood pressure by 1.5 mm Hg (95% CI, –2.8 to –0.3 mm Hg).
All-cause mortality did not differ between the groups. Serious adverse events occurred in 9.1% of the CDSS group, compared with 10.9% of the group whose care team did not use the CDSS.
Addressing social needs
Experts who were not involved in the trial said the marginal impact of the CDSS was no surprise given the mixed results of such tools in previous studies.
However, the lackluster result “might be a shock to people investing a lot in clinical decision support,” said Elbert Huang, MD, MPH, director of the Center for Chronic Disease Research and Policy at the University of Chicago.
Anne Peters, MD, a professor of medicine at the University of Southern California, Los Angeles, said the administrative burden of entering each patient’s data into the system would slow down care and frustrate clinicians. “The system has to be smarter than this.”
On the other hand, the findings of the D4C trial align with other research showing that team-based care strategies are effective for diabetes management.
Dr. Huang noted that there is a “well-established history” of diabetes quality improvement programs, health coaches, buddy programs, and community health worker programs. He added that the new findings “might help to remind everyone of the importance of these programs, which are not always well supported.”
“The bottom line of the paper might be that investing in patient engagement programs might get us 90% of the way to our goal of improving diabetes care,” Dr. Huang said.
Still, Dr. Peters said the portion of patients in the trial who benefited from team-based care seemed “disturbingly low.” Just 16.9% of patients who received team-based care and CDSS and 13% of those who received team-based care alone improved in all three measures. “This system doesn’t get you to where you want to be by a long shot.”
She added that a team-based approach, particularly the use of health coaches, would be a “huge improvement” over fragmented care provided in much of the U.S. safety-net system.
Another team approach
Many systems are striving to improve diabetes management in response to payment incentives, Dr. Huang said.
In a separate retrospective analysis, published in Annals of Family Medicine, researchers at the Mayo Clinic, Rochester, Minn., reported quality improvement gains among primary care practices that adopted a team-based model called Enhanced Primary Care Diabetes (EPCD). The model deployed a range of strategies, such as empowering nurses to engage with patients outside of scheduled office visits and including pharmacists on care teams.
Mayo’s approach did not specifically target underserved populations. Rather, researchers evaluated the model’s impact on about 17,000 patients treated at 32 Mayo internal medicine and family medicine practices of varying sizes, resources, and community settings.
Among staff clinician practices using the EPCD model improved patients’ scores on a composite quality measure called D5, which incorporates glycemic control, blood pressure control, low-density lipoprotein control, tobacco abstinence, and aspirin use.
Following implementation, the portion of patients in those practices meeting the D5 indicator increased from 42.9% to 45.0% (incident rate ratio, 1.005; P = .001).
Meanwhile, the portion of patients meeting the indicator increased from 38.9% to 42.0% (IRR, 1.011; P = .003) at resident physician practices that used the EPCD model and decreased from 36.2% to 35.5% (IRR, 0.994; P < .001) at staff clinician practices that did not use the model.
In contrast to the team-based approach used in China, the EPCD protocol “is very complex, and it will be difficult to implement in low-resource settings,” Dr. He said.
The D4C trial was funded by the Xiamen Municipal Health Commission. The Mayo study was funded by a National Institutes of Diabetes and Digestive and Kidney Diseases grant. Dr. He, Dr. Peters, and Dr. Huang reported no relevant financial interests.
a randomized trial in China showed.
The tool required clinicians to enter patient data into a computer in order to generate individualized treatment recommendations, adding to their administrative burdens. It also couldn’t tackle patients’ problems with access and affordability of medications.
Nevertheless, the model could curtail physician burnout and improve the quality of care in primary care clinics with limited resources, the researchers said in a paper published in the Annals of Internal Medicine.
They concluded that the findings support “widespread adoption” of the model in China and other low- or middle-income countries where diabetes is on the rise.
Co–principal investigator Jiang He, MD, PhD, chair of epidemiology at Tulane University, New Orleans, said the findings could apply to federally qualified health care (FQHC) clinics that treat underserved patients in the United States.
“At many FQHC clinics, nurse practitioners have to take care of patients with multiple chronic disease conditions. Team-based care with a computerized clinical decision support system will help them and improve patient care,” Dr. He said.
Small improvements
To conduct the trial, called Diabetes Complication Control in Community Clinics (D4C), Dr. He and colleagues randomly assigned 19 out of the 38 community health centers in Xiamen, China, to have a clinical decision support tool installed on the computers of primary care physicians and health coaches.
Starting in October 2016 the researchers recruited 11,132 patients aged 50 and older with uncontrolled diabetes and at least one comorbid condition, with 5,475 patients receiving team-based care with the CDSS and the remainder receiving team-based care alone.
The CDSS generated individualized risk factor summaries and treatment recommendations, including prescriptions based on Chinese and U.S. clinical guidelines. It incorporated data on patients’ insurance plans and local availability of drugs.
At all centers, primary care physicians received training in managing glycemia, blood pressure, and lipids. Nurses were certified as health coaches after receiving training on nutrition, lifestyle changes, and medication adherence. Patients met with their coaches for half an hour every 3 months, and diabetes specialists visited each clinic monthly for team meetings and consultations.
After 18 months, patients undergoing team-based care alone lowered their hemoglobin A1c by 0.6 percentage points (95% confidence interval, –0.7 to –0.5 percentage points), LDL cholesterol by 12.5 mg/dL (95% CI, –13.6 to –11.3 mg/dL), and systolic blood pressure by 7.5 mm Hg (95% CI, –8.4 to –6.6 mm Hg).
The group whose care teams used the CDSS further reduced A1c by 0.2 percentage points (95% CI, –0.3 to –0.1 percentage points), LDL cholesterol by 6.5 mg/dL (95% CI, –8.3 to -4.6 mg/dL), and blood pressure by 1.5 mm Hg (95% CI, –2.8 to –0.3 mm Hg).
All-cause mortality did not differ between the groups. Serious adverse events occurred in 9.1% of the CDSS group, compared with 10.9% of the group whose care team did not use the CDSS.
Addressing social needs
Experts who were not involved in the trial said the marginal impact of the CDSS was no surprise given the mixed results of such tools in previous studies.
However, the lackluster result “might be a shock to people investing a lot in clinical decision support,” said Elbert Huang, MD, MPH, director of the Center for Chronic Disease Research and Policy at the University of Chicago.
Anne Peters, MD, a professor of medicine at the University of Southern California, Los Angeles, said the administrative burden of entering each patient’s data into the system would slow down care and frustrate clinicians. “The system has to be smarter than this.”
On the other hand, the findings of the D4C trial align with other research showing that team-based care strategies are effective for diabetes management.
Dr. Huang noted that there is a “well-established history” of diabetes quality improvement programs, health coaches, buddy programs, and community health worker programs. He added that the new findings “might help to remind everyone of the importance of these programs, which are not always well supported.”
“The bottom line of the paper might be that investing in patient engagement programs might get us 90% of the way to our goal of improving diabetes care,” Dr. Huang said.
Still, Dr. Peters said the portion of patients in the trial who benefited from team-based care seemed “disturbingly low.” Just 16.9% of patients who received team-based care and CDSS and 13% of those who received team-based care alone improved in all three measures. “This system doesn’t get you to where you want to be by a long shot.”
She added that a team-based approach, particularly the use of health coaches, would be a “huge improvement” over fragmented care provided in much of the U.S. safety-net system.
Another team approach
Many systems are striving to improve diabetes management in response to payment incentives, Dr. Huang said.
In a separate retrospective analysis, published in Annals of Family Medicine, researchers at the Mayo Clinic, Rochester, Minn., reported quality improvement gains among primary care practices that adopted a team-based model called Enhanced Primary Care Diabetes (EPCD). The model deployed a range of strategies, such as empowering nurses to engage with patients outside of scheduled office visits and including pharmacists on care teams.
Mayo’s approach did not specifically target underserved populations. Rather, researchers evaluated the model’s impact on about 17,000 patients treated at 32 Mayo internal medicine and family medicine practices of varying sizes, resources, and community settings.
Among staff clinician practices using the EPCD model improved patients’ scores on a composite quality measure called D5, which incorporates glycemic control, blood pressure control, low-density lipoprotein control, tobacco abstinence, and aspirin use.
Following implementation, the portion of patients in those practices meeting the D5 indicator increased from 42.9% to 45.0% (incident rate ratio, 1.005; P = .001).
Meanwhile, the portion of patients meeting the indicator increased from 38.9% to 42.0% (IRR, 1.011; P = .003) at resident physician practices that used the EPCD model and decreased from 36.2% to 35.5% (IRR, 0.994; P < .001) at staff clinician practices that did not use the model.
In contrast to the team-based approach used in China, the EPCD protocol “is very complex, and it will be difficult to implement in low-resource settings,” Dr. He said.
The D4C trial was funded by the Xiamen Municipal Health Commission. The Mayo study was funded by a National Institutes of Diabetes and Digestive and Kidney Diseases grant. Dr. He, Dr. Peters, and Dr. Huang reported no relevant financial interests.
a randomized trial in China showed.
The tool required clinicians to enter patient data into a computer in order to generate individualized treatment recommendations, adding to their administrative burdens. It also couldn’t tackle patients’ problems with access and affordability of medications.
Nevertheless, the model could curtail physician burnout and improve the quality of care in primary care clinics with limited resources, the researchers said in a paper published in the Annals of Internal Medicine.
They concluded that the findings support “widespread adoption” of the model in China and other low- or middle-income countries where diabetes is on the rise.
Co–principal investigator Jiang He, MD, PhD, chair of epidemiology at Tulane University, New Orleans, said the findings could apply to federally qualified health care (FQHC) clinics that treat underserved patients in the United States.
“At many FQHC clinics, nurse practitioners have to take care of patients with multiple chronic disease conditions. Team-based care with a computerized clinical decision support system will help them and improve patient care,” Dr. He said.
Small improvements
To conduct the trial, called Diabetes Complication Control in Community Clinics (D4C), Dr. He and colleagues randomly assigned 19 out of the 38 community health centers in Xiamen, China, to have a clinical decision support tool installed on the computers of primary care physicians and health coaches.
Starting in October 2016 the researchers recruited 11,132 patients aged 50 and older with uncontrolled diabetes and at least one comorbid condition, with 5,475 patients receiving team-based care with the CDSS and the remainder receiving team-based care alone.
The CDSS generated individualized risk factor summaries and treatment recommendations, including prescriptions based on Chinese and U.S. clinical guidelines. It incorporated data on patients’ insurance plans and local availability of drugs.
At all centers, primary care physicians received training in managing glycemia, blood pressure, and lipids. Nurses were certified as health coaches after receiving training on nutrition, lifestyle changes, and medication adherence. Patients met with their coaches for half an hour every 3 months, and diabetes specialists visited each clinic monthly for team meetings and consultations.
After 18 months, patients undergoing team-based care alone lowered their hemoglobin A1c by 0.6 percentage points (95% confidence interval, –0.7 to –0.5 percentage points), LDL cholesterol by 12.5 mg/dL (95% CI, –13.6 to –11.3 mg/dL), and systolic blood pressure by 7.5 mm Hg (95% CI, –8.4 to –6.6 mm Hg).
The group whose care teams used the CDSS further reduced A1c by 0.2 percentage points (95% CI, –0.3 to –0.1 percentage points), LDL cholesterol by 6.5 mg/dL (95% CI, –8.3 to -4.6 mg/dL), and blood pressure by 1.5 mm Hg (95% CI, –2.8 to –0.3 mm Hg).
All-cause mortality did not differ between the groups. Serious adverse events occurred in 9.1% of the CDSS group, compared with 10.9% of the group whose care team did not use the CDSS.
Addressing social needs
Experts who were not involved in the trial said the marginal impact of the CDSS was no surprise given the mixed results of such tools in previous studies.
However, the lackluster result “might be a shock to people investing a lot in clinical decision support,” said Elbert Huang, MD, MPH, director of the Center for Chronic Disease Research and Policy at the University of Chicago.
Anne Peters, MD, a professor of medicine at the University of Southern California, Los Angeles, said the administrative burden of entering each patient’s data into the system would slow down care and frustrate clinicians. “The system has to be smarter than this.”
On the other hand, the findings of the D4C trial align with other research showing that team-based care strategies are effective for diabetes management.
Dr. Huang noted that there is a “well-established history” of diabetes quality improvement programs, health coaches, buddy programs, and community health worker programs. He added that the new findings “might help to remind everyone of the importance of these programs, which are not always well supported.”
“The bottom line of the paper might be that investing in patient engagement programs might get us 90% of the way to our goal of improving diabetes care,” Dr. Huang said.
Still, Dr. Peters said the portion of patients in the trial who benefited from team-based care seemed “disturbingly low.” Just 16.9% of patients who received team-based care and CDSS and 13% of those who received team-based care alone improved in all three measures. “This system doesn’t get you to where you want to be by a long shot.”
She added that a team-based approach, particularly the use of health coaches, would be a “huge improvement” over fragmented care provided in much of the U.S. safety-net system.
Another team approach
Many systems are striving to improve diabetes management in response to payment incentives, Dr. Huang said.
In a separate retrospective analysis, published in Annals of Family Medicine, researchers at the Mayo Clinic, Rochester, Minn., reported quality improvement gains among primary care practices that adopted a team-based model called Enhanced Primary Care Diabetes (EPCD). The model deployed a range of strategies, such as empowering nurses to engage with patients outside of scheduled office visits and including pharmacists on care teams.
Mayo’s approach did not specifically target underserved populations. Rather, researchers evaluated the model’s impact on about 17,000 patients treated at 32 Mayo internal medicine and family medicine practices of varying sizes, resources, and community settings.
Among staff clinician practices using the EPCD model improved patients’ scores on a composite quality measure called D5, which incorporates glycemic control, blood pressure control, low-density lipoprotein control, tobacco abstinence, and aspirin use.
Following implementation, the portion of patients in those practices meeting the D5 indicator increased from 42.9% to 45.0% (incident rate ratio, 1.005; P = .001).
Meanwhile, the portion of patients meeting the indicator increased from 38.9% to 42.0% (IRR, 1.011; P = .003) at resident physician practices that used the EPCD model and decreased from 36.2% to 35.5% (IRR, 0.994; P < .001) at staff clinician practices that did not use the model.
In contrast to the team-based approach used in China, the EPCD protocol “is very complex, and it will be difficult to implement in low-resource settings,” Dr. He said.
The D4C trial was funded by the Xiamen Municipal Health Commission. The Mayo study was funded by a National Institutes of Diabetes and Digestive and Kidney Diseases grant. Dr. He, Dr. Peters, and Dr. Huang reported no relevant financial interests.
FROM ANNALS OF INTERNAL MEDICINE
Dapagliflozin reduces hospitalizations in patients with CKD
These findings add to a growing body of evidence supporting a range of positive benefits from dapagliflozin, including reduced risks of mortality, cardiovascular events, and kidney events, lead author Meir Schechter, MD, PhD, of the Hebrew University of Jerusalem and colleagues wrote in Annals of Internal Medicine.“Although cardiovascular and kidney outcomes with SGLT2 inhibitors have been studied extensively, there is a paucity of data evaluating the effects of SGLT2 inhibitors on hospitalizations for any cause.”
The findings are based on a post hoc analysis of the DAPA-CKD trial, which involved 4,304 patients with CKD in 21 countries. Patients were randomized in a 1:1 ratio to receive dapagliflozin 10 mg orally once a day or matching placebo. The present analysis quantified first hospitalizations for any cause, all hospitalizations, cause-specific hospitalizations, and several related outcomes.
After a median follow-up of 2.4 years, 28% of the population had been hospitalized a total of 2,072 times.
Compared with placebo, dapagliflozin significantly reduced risk of first hospitalization by 16% (hazard ratio, 0.84; 95% confidence interval, 0.75-0.94) and rate of all hospitalizations by 21% (rate ratio, 0.79; 95% CI, 0.70-0.89). These findings remained significant regardless of type 2 diabetes status, with significant benefits seen across reasons for admission, including renal/urinary disorders, cardiac disorders, neoplasms, and metabolism/nutrition disorders. In addition, dapagliflozin was associated with shorter mean time in hospital (2.3 vs. 2.8 days; P = .027) and longer time alive and out of hospital (354.9 vs. 351.7; P = .023).
“These findings highlight additional benefits of dapagliflozin beyond those seen for cardiovascular and kidney events, all-cause and cause-specific mortality, eGFR [estimated glomerular filtration rate] slope, and albuminuria and should be considered when evaluating the totality of evidence favoring provision of dapagliflozin to patients with CKD,” the investigators concluded.
Positive data, positive experiences
Shree Mulay, MD, a nephrologist in private practice in western Tennessee, said this study is “one of several other articles that already exist” demonstrating the broad benefits of SGLT2 inhibitors.
“The evidence is pretty substantial,” Dr. Mulay said in an interview. “I think SGLT2 inhibitors are the new statin of this era. ... I won’t be surprised if in the next year or 2 or 3 they truly become the standard of care.”
Dr. Mulay also speaks from experience working in both the chronic and acute setting, where he’s observed “some magical stuff happening” in patients started on SGLT2 inhibitors, especially those in heart failure who are fluid overloaded.
“It’s phenomenal stuff,” Dr. Mulay said. “You can really stabilize patients’ hemodynamics.”
In the private health care setting, he described widespread enthusiasm among nephrologists, although others still appear skeptical.
“It’s really our cardiology colleagues that I feel are underprescribing it,” Dr. Mulay said. “So, I’m kind of taking it on myself, when I see a heart failure patient, to go ahead and put them on this.”
It’s unclear why some cardiologists seem apprehensive, Dr. Mulay continued, although he suggested that unclear guidelines and a lack of first-hand experience may be to blame.
Nephrologists and cardiologists sometimes agree
In the academic arena, Leslie Gewin, MD, associate professor at Washington University in St. Louis and the John Cochran VA Hospital, also in St. Louis, has seen similar support for SGLT2 inhibitors among both nephrologists and cardiologists.
“We had a joint nephrology-cardiology medicine grand rounds at Wash U in St. Louis maybe 2 weeks ago,” Dr. Gewin said in an interview. “The cardiologists and nephrologists tag-teamed to present data about SGLT2 inhibitors, and we kind of joked that this was the one thing we both could get behind and support.”
Still, she has seen some reluctance among non-nephrology clinicians lacking SGLT2 experience, specifically when managing patients who have poor kidney function.
“There can be some hesitancy among physicians if the GFR is low,” Dr. Gewin said. “That’s where I’ve had to sort of push the envelope with non-nephrologists, saying: ‘Look, we feel pretty comfortable starting down to a GFR of about 20.’ ”
Early rises in creatinine may also spook providers, she noted.
“Sometimes, when we start SGLT2 inhibitors, the creatinine increases slightly, and the [primary care provider] gets concerned,” Dr. Gewin said. “We say: ‘No, this is expected. Don’t worry, hold the course, this is a good drug.’ ”
Like Dr. Mulay, Dr. Gewin said the present study offers further encouragement for the efficacy of this drug class. She also said sufficient data have been published to allay earlier concerns about potential safety signals, such as bone fractures and amputations.
“SGLT2 inhibitors seem to be a lot safer than what we initially had thought,” Dr. Gewin said. “That’s very encouraging.”
The study was funded by AstraZeneca. The investigators disclosed additional relationships with Bayer, Janssen, Gilead, and others. Dr. Gewin and Dr. Mulay disclosed no relevant conflicts of interest.
These findings add to a growing body of evidence supporting a range of positive benefits from dapagliflozin, including reduced risks of mortality, cardiovascular events, and kidney events, lead author Meir Schechter, MD, PhD, of the Hebrew University of Jerusalem and colleagues wrote in Annals of Internal Medicine.“Although cardiovascular and kidney outcomes with SGLT2 inhibitors have been studied extensively, there is a paucity of data evaluating the effects of SGLT2 inhibitors on hospitalizations for any cause.”
The findings are based on a post hoc analysis of the DAPA-CKD trial, which involved 4,304 patients with CKD in 21 countries. Patients were randomized in a 1:1 ratio to receive dapagliflozin 10 mg orally once a day or matching placebo. The present analysis quantified first hospitalizations for any cause, all hospitalizations, cause-specific hospitalizations, and several related outcomes.
After a median follow-up of 2.4 years, 28% of the population had been hospitalized a total of 2,072 times.
Compared with placebo, dapagliflozin significantly reduced risk of first hospitalization by 16% (hazard ratio, 0.84; 95% confidence interval, 0.75-0.94) and rate of all hospitalizations by 21% (rate ratio, 0.79; 95% CI, 0.70-0.89). These findings remained significant regardless of type 2 diabetes status, with significant benefits seen across reasons for admission, including renal/urinary disorders, cardiac disorders, neoplasms, and metabolism/nutrition disorders. In addition, dapagliflozin was associated with shorter mean time in hospital (2.3 vs. 2.8 days; P = .027) and longer time alive and out of hospital (354.9 vs. 351.7; P = .023).
“These findings highlight additional benefits of dapagliflozin beyond those seen for cardiovascular and kidney events, all-cause and cause-specific mortality, eGFR [estimated glomerular filtration rate] slope, and albuminuria and should be considered when evaluating the totality of evidence favoring provision of dapagliflozin to patients with CKD,” the investigators concluded.
Positive data, positive experiences
Shree Mulay, MD, a nephrologist in private practice in western Tennessee, said this study is “one of several other articles that already exist” demonstrating the broad benefits of SGLT2 inhibitors.
“The evidence is pretty substantial,” Dr. Mulay said in an interview. “I think SGLT2 inhibitors are the new statin of this era. ... I won’t be surprised if in the next year or 2 or 3 they truly become the standard of care.”
Dr. Mulay also speaks from experience working in both the chronic and acute setting, where he’s observed “some magical stuff happening” in patients started on SGLT2 inhibitors, especially those in heart failure who are fluid overloaded.
“It’s phenomenal stuff,” Dr. Mulay said. “You can really stabilize patients’ hemodynamics.”
In the private health care setting, he described widespread enthusiasm among nephrologists, although others still appear skeptical.
“It’s really our cardiology colleagues that I feel are underprescribing it,” Dr. Mulay said. “So, I’m kind of taking it on myself, when I see a heart failure patient, to go ahead and put them on this.”
It’s unclear why some cardiologists seem apprehensive, Dr. Mulay continued, although he suggested that unclear guidelines and a lack of first-hand experience may be to blame.
Nephrologists and cardiologists sometimes agree
In the academic arena, Leslie Gewin, MD, associate professor at Washington University in St. Louis and the John Cochran VA Hospital, also in St. Louis, has seen similar support for SGLT2 inhibitors among both nephrologists and cardiologists.
“We had a joint nephrology-cardiology medicine grand rounds at Wash U in St. Louis maybe 2 weeks ago,” Dr. Gewin said in an interview. “The cardiologists and nephrologists tag-teamed to present data about SGLT2 inhibitors, and we kind of joked that this was the one thing we both could get behind and support.”
Still, she has seen some reluctance among non-nephrology clinicians lacking SGLT2 experience, specifically when managing patients who have poor kidney function.
“There can be some hesitancy among physicians if the GFR is low,” Dr. Gewin said. “That’s where I’ve had to sort of push the envelope with non-nephrologists, saying: ‘Look, we feel pretty comfortable starting down to a GFR of about 20.’ ”
Early rises in creatinine may also spook providers, she noted.
“Sometimes, when we start SGLT2 inhibitors, the creatinine increases slightly, and the [primary care provider] gets concerned,” Dr. Gewin said. “We say: ‘No, this is expected. Don’t worry, hold the course, this is a good drug.’ ”
Like Dr. Mulay, Dr. Gewin said the present study offers further encouragement for the efficacy of this drug class. She also said sufficient data have been published to allay earlier concerns about potential safety signals, such as bone fractures and amputations.
“SGLT2 inhibitors seem to be a lot safer than what we initially had thought,” Dr. Gewin said. “That’s very encouraging.”
The study was funded by AstraZeneca. The investigators disclosed additional relationships with Bayer, Janssen, Gilead, and others. Dr. Gewin and Dr. Mulay disclosed no relevant conflicts of interest.
These findings add to a growing body of evidence supporting a range of positive benefits from dapagliflozin, including reduced risks of mortality, cardiovascular events, and kidney events, lead author Meir Schechter, MD, PhD, of the Hebrew University of Jerusalem and colleagues wrote in Annals of Internal Medicine.“Although cardiovascular and kidney outcomes with SGLT2 inhibitors have been studied extensively, there is a paucity of data evaluating the effects of SGLT2 inhibitors on hospitalizations for any cause.”
The findings are based on a post hoc analysis of the DAPA-CKD trial, which involved 4,304 patients with CKD in 21 countries. Patients were randomized in a 1:1 ratio to receive dapagliflozin 10 mg orally once a day or matching placebo. The present analysis quantified first hospitalizations for any cause, all hospitalizations, cause-specific hospitalizations, and several related outcomes.
After a median follow-up of 2.4 years, 28% of the population had been hospitalized a total of 2,072 times.
Compared with placebo, dapagliflozin significantly reduced risk of first hospitalization by 16% (hazard ratio, 0.84; 95% confidence interval, 0.75-0.94) and rate of all hospitalizations by 21% (rate ratio, 0.79; 95% CI, 0.70-0.89). These findings remained significant regardless of type 2 diabetes status, with significant benefits seen across reasons for admission, including renal/urinary disorders, cardiac disorders, neoplasms, and metabolism/nutrition disorders. In addition, dapagliflozin was associated with shorter mean time in hospital (2.3 vs. 2.8 days; P = .027) and longer time alive and out of hospital (354.9 vs. 351.7; P = .023).
“These findings highlight additional benefits of dapagliflozin beyond those seen for cardiovascular and kidney events, all-cause and cause-specific mortality, eGFR [estimated glomerular filtration rate] slope, and albuminuria and should be considered when evaluating the totality of evidence favoring provision of dapagliflozin to patients with CKD,” the investigators concluded.
Positive data, positive experiences
Shree Mulay, MD, a nephrologist in private practice in western Tennessee, said this study is “one of several other articles that already exist” demonstrating the broad benefits of SGLT2 inhibitors.
“The evidence is pretty substantial,” Dr. Mulay said in an interview. “I think SGLT2 inhibitors are the new statin of this era. ... I won’t be surprised if in the next year or 2 or 3 they truly become the standard of care.”
Dr. Mulay also speaks from experience working in both the chronic and acute setting, where he’s observed “some magical stuff happening” in patients started on SGLT2 inhibitors, especially those in heart failure who are fluid overloaded.
“It’s phenomenal stuff,” Dr. Mulay said. “You can really stabilize patients’ hemodynamics.”
In the private health care setting, he described widespread enthusiasm among nephrologists, although others still appear skeptical.
“It’s really our cardiology colleagues that I feel are underprescribing it,” Dr. Mulay said. “So, I’m kind of taking it on myself, when I see a heart failure patient, to go ahead and put them on this.”
It’s unclear why some cardiologists seem apprehensive, Dr. Mulay continued, although he suggested that unclear guidelines and a lack of first-hand experience may be to blame.
Nephrologists and cardiologists sometimes agree
In the academic arena, Leslie Gewin, MD, associate professor at Washington University in St. Louis and the John Cochran VA Hospital, also in St. Louis, has seen similar support for SGLT2 inhibitors among both nephrologists and cardiologists.
“We had a joint nephrology-cardiology medicine grand rounds at Wash U in St. Louis maybe 2 weeks ago,” Dr. Gewin said in an interview. “The cardiologists and nephrologists tag-teamed to present data about SGLT2 inhibitors, and we kind of joked that this was the one thing we both could get behind and support.”
Still, she has seen some reluctance among non-nephrology clinicians lacking SGLT2 experience, specifically when managing patients who have poor kidney function.
“There can be some hesitancy among physicians if the GFR is low,” Dr. Gewin said. “That’s where I’ve had to sort of push the envelope with non-nephrologists, saying: ‘Look, we feel pretty comfortable starting down to a GFR of about 20.’ ”
Early rises in creatinine may also spook providers, she noted.
“Sometimes, when we start SGLT2 inhibitors, the creatinine increases slightly, and the [primary care provider] gets concerned,” Dr. Gewin said. “We say: ‘No, this is expected. Don’t worry, hold the course, this is a good drug.’ ”
Like Dr. Mulay, Dr. Gewin said the present study offers further encouragement for the efficacy of this drug class. She also said sufficient data have been published to allay earlier concerns about potential safety signals, such as bone fractures and amputations.
“SGLT2 inhibitors seem to be a lot safer than what we initially had thought,” Dr. Gewin said. “That’s very encouraging.”
The study was funded by AstraZeneca. The investigators disclosed additional relationships with Bayer, Janssen, Gilead, and others. Dr. Gewin and Dr. Mulay disclosed no relevant conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
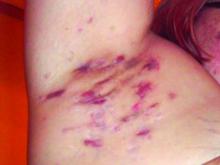
Consider quality of life, comorbidities in hidradenitis suppurativa
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Gestational hypertension-diabetes combo signals CVD risk
Women who develop transient hypertensive disorders during their pregnancy are at risk for developing subsequent cardiovascular disease (CVD), particularly if this experienced at the same time as gestational diabetes.
In a large population-based study, the adjusted hazard ratios for developing CVD following a gestational hypertensive disorder (GHTD) alone were 1.90 (95% confidence interval, 1.151-2.25) within 5 years and 1.41 (95% CI, 1.12-1.76) after 5 years or more.
When gestational diabetes was added into the mix, however, the risk for CVD after 5 years more than doubled (aHR, 2.43; 95% CI, 1.60-3.67). Risk in the earlier postpartum period was also raised by the combination, but this was not significant (aHR, 1.42; 95% CI, 0.78-2.58).
Having gestational diabetes by itself did not seem to increase the risk for later CVD in the analysis, despite being linked to higher heart disease risk in other studies.
“These are women coming out of a pregnancy – young women of reproductive age – so this is not a group that typically has cardiovascular events,” said Ravi Retnakaran, MD, in an interview, an investigator in the new study, which is published in JAMA Network Open.
“If they are somebody who has both disorders concurrently in their pregnancy, they may be at even greater risk than a woman with one or the other disorder,” added Dr. Retnakaran, who is professor of medicine at the University of Toronto and an endocrinologist at the Leadership Sinai Centre for Diabetes, Mount Sinai Hospital, also in Toronto. “In other words, amongst already high-risk patients. This is identifying a subset at maybe an even higher risk.”
It doesn’t mean that there is a huge absolute risk, Dr. Retnakaran said, but it is showing that there is a heightened risk such that women and their clinicians need to be aware of and potentially the need for greater preventative care in the future.
“It is allowing you to identify future lifetime risk of cardiovascular disease,” he said.
Study rationale and design
GHTD is “a forerunner of hypertension,” and gestational diabetes is “a precursor of diabetes” – each associated with a high risk of developing CVD in the years after pregnancy, the investigators said. While studies have looked at their individual contributions to future CVD risk, not many had looked to see what risks having both may confer in the postpregnancy years.
For the analysis, data on 886,295 women with GHTD (43,861), gestational diabetes (54,061), both (4,975), or neither (783,398) were obtained from several Canadian administrative health databases.
The mean age was around 30 years across the groups, with those with both conditions or gestational diabetes alone more likely to be older than those with GTHD alone or neither condition (32 vs. 29 years, respectively, P < .001).
After a total follow-up period of 12 years, 1,999 CVD events were recorded, most of them (1,162) 5 years after the pregnancy.
Pregnancy is a stress test for the heart
“We know that what we call adverse pregnancy outcomes – things like gestational hypertension, and gestational diabetes, and preeclampsia – are on the rise globally,” Natalie A. Bello, MD, director of hypertension research at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, commented in an interview.
“People who are younger and of childbearing age who are going into pregnancy now are less healthy than they perhaps were in the past,” Dr. Bello suggested, with more hypertension, more obesity, and people being less physically active. “We think that’s translating into some of the pregnancy complications.”
That’s concerning for a number of reasons, said Dr. Bello, who is also the cochair of the American College of Cardiology’s Cardio-Obstetrics Workgroup, and the biggest one perhaps is the stress that these may conditions may be placing on the heart.
“We know that when individuals have an adverse pregnancy outcome like gestational hypertension, or gestational diabetes, their risk for heart disease is increased in the future compared to someone who has an uncomplicated pregnancy,” she said. “So, we sort of say pregnancy is like a stress test for your heart.”
Dr. Bello added that “these situations, these adverse pregnancy outcomes are an indicator for us as physicians, but also they should be for patients as well, to sort of make sure they’re talking to their doctor about their risk factors and modifying them whenever possible.”
The population studied came from quite a racially, ethnically, and economically diverse area of Canada, Dr. Bello pointed out, although because of the nature of an administrative database there wasn’t information on individual level risk factors.
“We don’t know things like smoking, or if individuals were obese when they were pregnant. So, there are some limitations that should be noted,” she said.
Also, the results don’t mean that isolated gestational diabetes “isn’t something we need to be concerned about,” Dr. Bello observed, adding that the study may have been underpowered to look at this association. “It may just be that it will take a longer time for individuals who have gestational diabetes who don’t make lifestyle changes to develop diabetes, and then develop heart disease.”
The main message is that the women who have a co-occurrence of gestational hypertension and gestational diabetes are at particularly high risk of cardiovascular disease in the future,” said Dr. Retnakaran.
“The way to look at it from a patient standpoint is that we are all on different tracks in terms of our cardiometabolic destiny,” and that these data give “some understanding of what kind of tracks they are on for future risk,” Dr. Retnakaran said.
“A history of either gestational hypertension, and/or gestational diabetes should be really a warning sign for physicians and for patients that they have a higher risk of heart disease,” said Dr. Bello.
She added that this is a signal “that we need to do things to modify their risk, because we know that about 80% of heart disease is modifiable and preventable with proper risk factor management.”
The study was funded by the Ontario Ministry of Health and Long-Term Care. Dr. Retnakaran has received grants and personal fees from Novo Nordisk and Merck, grants from Boehringer Ingelheim, and personal fees from Eli Lily Takeda, and Sanofi. Dr. Bello had no conflicts of interest to disclose.
Women who develop transient hypertensive disorders during their pregnancy are at risk for developing subsequent cardiovascular disease (CVD), particularly if this experienced at the same time as gestational diabetes.
In a large population-based study, the adjusted hazard ratios for developing CVD following a gestational hypertensive disorder (GHTD) alone were 1.90 (95% confidence interval, 1.151-2.25) within 5 years and 1.41 (95% CI, 1.12-1.76) after 5 years or more.
When gestational diabetes was added into the mix, however, the risk for CVD after 5 years more than doubled (aHR, 2.43; 95% CI, 1.60-3.67). Risk in the earlier postpartum period was also raised by the combination, but this was not significant (aHR, 1.42; 95% CI, 0.78-2.58).
Having gestational diabetes by itself did not seem to increase the risk for later CVD in the analysis, despite being linked to higher heart disease risk in other studies.
“These are women coming out of a pregnancy – young women of reproductive age – so this is not a group that typically has cardiovascular events,” said Ravi Retnakaran, MD, in an interview, an investigator in the new study, which is published in JAMA Network Open.
“If they are somebody who has both disorders concurrently in their pregnancy, they may be at even greater risk than a woman with one or the other disorder,” added Dr. Retnakaran, who is professor of medicine at the University of Toronto and an endocrinologist at the Leadership Sinai Centre for Diabetes, Mount Sinai Hospital, also in Toronto. “In other words, amongst already high-risk patients. This is identifying a subset at maybe an even higher risk.”
It doesn’t mean that there is a huge absolute risk, Dr. Retnakaran said, but it is showing that there is a heightened risk such that women and their clinicians need to be aware of and potentially the need for greater preventative care in the future.
“It is allowing you to identify future lifetime risk of cardiovascular disease,” he said.
Study rationale and design
GHTD is “a forerunner of hypertension,” and gestational diabetes is “a precursor of diabetes” – each associated with a high risk of developing CVD in the years after pregnancy, the investigators said. While studies have looked at their individual contributions to future CVD risk, not many had looked to see what risks having both may confer in the postpregnancy years.
For the analysis, data on 886,295 women with GHTD (43,861), gestational diabetes (54,061), both (4,975), or neither (783,398) were obtained from several Canadian administrative health databases.
The mean age was around 30 years across the groups, with those with both conditions or gestational diabetes alone more likely to be older than those with GTHD alone or neither condition (32 vs. 29 years, respectively, P < .001).
After a total follow-up period of 12 years, 1,999 CVD events were recorded, most of them (1,162) 5 years after the pregnancy.
Pregnancy is a stress test for the heart
“We know that what we call adverse pregnancy outcomes – things like gestational hypertension, and gestational diabetes, and preeclampsia – are on the rise globally,” Natalie A. Bello, MD, director of hypertension research at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, commented in an interview.
“People who are younger and of childbearing age who are going into pregnancy now are less healthy than they perhaps were in the past,” Dr. Bello suggested, with more hypertension, more obesity, and people being less physically active. “We think that’s translating into some of the pregnancy complications.”
That’s concerning for a number of reasons, said Dr. Bello, who is also the cochair of the American College of Cardiology’s Cardio-Obstetrics Workgroup, and the biggest one perhaps is the stress that these may conditions may be placing on the heart.
“We know that when individuals have an adverse pregnancy outcome like gestational hypertension, or gestational diabetes, their risk for heart disease is increased in the future compared to someone who has an uncomplicated pregnancy,” she said. “So, we sort of say pregnancy is like a stress test for your heart.”
Dr. Bello added that “these situations, these adverse pregnancy outcomes are an indicator for us as physicians, but also they should be for patients as well, to sort of make sure they’re talking to their doctor about their risk factors and modifying them whenever possible.”
The population studied came from quite a racially, ethnically, and economically diverse area of Canada, Dr. Bello pointed out, although because of the nature of an administrative database there wasn’t information on individual level risk factors.
“We don’t know things like smoking, or if individuals were obese when they were pregnant. So, there are some limitations that should be noted,” she said.
Also, the results don’t mean that isolated gestational diabetes “isn’t something we need to be concerned about,” Dr. Bello observed, adding that the study may have been underpowered to look at this association. “It may just be that it will take a longer time for individuals who have gestational diabetes who don’t make lifestyle changes to develop diabetes, and then develop heart disease.”
The main message is that the women who have a co-occurrence of gestational hypertension and gestational diabetes are at particularly high risk of cardiovascular disease in the future,” said Dr. Retnakaran.
“The way to look at it from a patient standpoint is that we are all on different tracks in terms of our cardiometabolic destiny,” and that these data give “some understanding of what kind of tracks they are on for future risk,” Dr. Retnakaran said.
“A history of either gestational hypertension, and/or gestational diabetes should be really a warning sign for physicians and for patients that they have a higher risk of heart disease,” said Dr. Bello.
She added that this is a signal “that we need to do things to modify their risk, because we know that about 80% of heart disease is modifiable and preventable with proper risk factor management.”
The study was funded by the Ontario Ministry of Health and Long-Term Care. Dr. Retnakaran has received grants and personal fees from Novo Nordisk and Merck, grants from Boehringer Ingelheim, and personal fees from Eli Lily Takeda, and Sanofi. Dr. Bello had no conflicts of interest to disclose.
Women who develop transient hypertensive disorders during their pregnancy are at risk for developing subsequent cardiovascular disease (CVD), particularly if this experienced at the same time as gestational diabetes.
In a large population-based study, the adjusted hazard ratios for developing CVD following a gestational hypertensive disorder (GHTD) alone were 1.90 (95% confidence interval, 1.151-2.25) within 5 years and 1.41 (95% CI, 1.12-1.76) after 5 years or more.
When gestational diabetes was added into the mix, however, the risk for CVD after 5 years more than doubled (aHR, 2.43; 95% CI, 1.60-3.67). Risk in the earlier postpartum period was also raised by the combination, but this was not significant (aHR, 1.42; 95% CI, 0.78-2.58).
Having gestational diabetes by itself did not seem to increase the risk for later CVD in the analysis, despite being linked to higher heart disease risk in other studies.
“These are women coming out of a pregnancy – young women of reproductive age – so this is not a group that typically has cardiovascular events,” said Ravi Retnakaran, MD, in an interview, an investigator in the new study, which is published in JAMA Network Open.
“If they are somebody who has both disorders concurrently in their pregnancy, they may be at even greater risk than a woman with one or the other disorder,” added Dr. Retnakaran, who is professor of medicine at the University of Toronto and an endocrinologist at the Leadership Sinai Centre for Diabetes, Mount Sinai Hospital, also in Toronto. “In other words, amongst already high-risk patients. This is identifying a subset at maybe an even higher risk.”
It doesn’t mean that there is a huge absolute risk, Dr. Retnakaran said, but it is showing that there is a heightened risk such that women and their clinicians need to be aware of and potentially the need for greater preventative care in the future.
“It is allowing you to identify future lifetime risk of cardiovascular disease,” he said.
Study rationale and design
GHTD is “a forerunner of hypertension,” and gestational diabetes is “a precursor of diabetes” – each associated with a high risk of developing CVD in the years after pregnancy, the investigators said. While studies have looked at their individual contributions to future CVD risk, not many had looked to see what risks having both may confer in the postpregnancy years.
For the analysis, data on 886,295 women with GHTD (43,861), gestational diabetes (54,061), both (4,975), or neither (783,398) were obtained from several Canadian administrative health databases.
The mean age was around 30 years across the groups, with those with both conditions or gestational diabetes alone more likely to be older than those with GTHD alone or neither condition (32 vs. 29 years, respectively, P < .001).
After a total follow-up period of 12 years, 1,999 CVD events were recorded, most of them (1,162) 5 years after the pregnancy.
Pregnancy is a stress test for the heart
“We know that what we call adverse pregnancy outcomes – things like gestational hypertension, and gestational diabetes, and preeclampsia – are on the rise globally,” Natalie A. Bello, MD, director of hypertension research at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, commented in an interview.
“People who are younger and of childbearing age who are going into pregnancy now are less healthy than they perhaps were in the past,” Dr. Bello suggested, with more hypertension, more obesity, and people being less physically active. “We think that’s translating into some of the pregnancy complications.”
That’s concerning for a number of reasons, said Dr. Bello, who is also the cochair of the American College of Cardiology’s Cardio-Obstetrics Workgroup, and the biggest one perhaps is the stress that these may conditions may be placing on the heart.
“We know that when individuals have an adverse pregnancy outcome like gestational hypertension, or gestational diabetes, their risk for heart disease is increased in the future compared to someone who has an uncomplicated pregnancy,” she said. “So, we sort of say pregnancy is like a stress test for your heart.”
Dr. Bello added that “these situations, these adverse pregnancy outcomes are an indicator for us as physicians, but also they should be for patients as well, to sort of make sure they’re talking to their doctor about their risk factors and modifying them whenever possible.”
The population studied came from quite a racially, ethnically, and economically diverse area of Canada, Dr. Bello pointed out, although because of the nature of an administrative database there wasn’t information on individual level risk factors.
“We don’t know things like smoking, or if individuals were obese when they were pregnant. So, there are some limitations that should be noted,” she said.
Also, the results don’t mean that isolated gestational diabetes “isn’t something we need to be concerned about,” Dr. Bello observed, adding that the study may have been underpowered to look at this association. “It may just be that it will take a longer time for individuals who have gestational diabetes who don’t make lifestyle changes to develop diabetes, and then develop heart disease.”
The main message is that the women who have a co-occurrence of gestational hypertension and gestational diabetes are at particularly high risk of cardiovascular disease in the future,” said Dr. Retnakaran.
“The way to look at it from a patient standpoint is that we are all on different tracks in terms of our cardiometabolic destiny,” and that these data give “some understanding of what kind of tracks they are on for future risk,” Dr. Retnakaran said.
“A history of either gestational hypertension, and/or gestational diabetes should be really a warning sign for physicians and for patients that they have a higher risk of heart disease,” said Dr. Bello.
She added that this is a signal “that we need to do things to modify their risk, because we know that about 80% of heart disease is modifiable and preventable with proper risk factor management.”
The study was funded by the Ontario Ministry of Health and Long-Term Care. Dr. Retnakaran has received grants and personal fees from Novo Nordisk and Merck, grants from Boehringer Ingelheim, and personal fees from Eli Lily Takeda, and Sanofi. Dr. Bello had no conflicts of interest to disclose.
FROM JAMA NETWORK OPEN
The TikTok trend that triggered a diabetes drug shortage
Weight loss advice is everywhere you look on social media, but one trend sweeping TikTok has led to shortages of an important diabetes drug.
Ozempic, a weekly injection that helps boost insulin sensitivity in people with type 2 diabetes, also suppresses appetite, which leads to weight loss. Stories of celebrities using the drug off-label to lose a few pounds have led to an explosion of interest. And now people with diabetes – people whose lives could be saved by the drug – are having trouble finding it.
Kim Kardashian and Elon Musk
In the spring, Kim Kardashian pulled off a dramatic weight loss to fit into Marilyn Monroe’s dress for the Met Gala. Soon rumors began to circulate that she’d used Ozempic to do it. Just this week, new Twitter owner Elon Musk tweeted about his own use of Ozempic and its sibling drug, Wegovy.
Variety dubbed Ozempic “the worst kept secret in Hollywood – especially given that its most enthusiastic users are not prediabetic and do not require the drug.” The rich and famous are spending $1,200 to $1,500 a month to get access.
As so often happens, high-profile use sparked a trend. Videos on TikTok hashtagged #ozempic have amassed more than 275 million views, and #ozempicweightloss has more than 110 million.
This raises concerns about who, exactly, is watching these videos, and what message they’re receiving.
“Forty-two percent of Americans have obesity, and even more have overweight. That’s affecting our younger people and our adolescents,” says Caroline Apovian, MD, codirector of the Center for Weight Management and Wellness at the Brigham and Women’s Hospital in Boston. “They’re looking at TikTok and other social media outlets for help.”
A new study shows how damaging this can be: Researchers analyzed 1,000 videos with nutrition, food, and weight-related hashtags, with over 1 billion views combined. They found that nearly all included messages glorifying weight loss and thinness.
At last, an effective weight-loss drug
Ozempic is Danish drug company Novo Nordisk’s brand name for semaglutide, which works by mimicking a naturally occurring hormone known as GLP-1. It travels to your brain and helps you feel full on less food. That leads to weight loss. In one 68-week study, semaglutide helped people lose an average of 15% of their body weight. But it’s not a miracle drug: You still have to change your eating habits and stay physically active.
The FDA approved Ozempic to treat people with type 2 diabetes in 2017. Four years later, Novo Nordisk received the green light for a higher-dose version meant specifically for people with obesity. Wegovy is approved for use only if you have a BMI of at least 27 with one or more weight-related ailments, or a BMI of 30 or more with none.
“These drugs are dominating my practice, because they’re so effective,” says Amanda Velazquez, MD, director of obesity medicine at Cedars-Sinai Medical Center in Los Angeles. The drug is considered safe, “so the majority of patients are good candidates.”
More demand than supply
As word spread about how well Ozempic and Wegovy worked, social media posts helped drive even more people to seek out the drugs. Now demand is outpacing the supply – according to the FDA, starter doses of Ozempic will have limited availability through January.
“In Hollywood, people are losing 10 pounds, getting it for $1,500 a month, and depleting stores for people who have such severe obesity that they have congestive heart failure and diabetes,” Dr. Apovian says. “These are people who are going to die, and you’re taking it away just for cosmetic weight loss. That is deplorable.”
In addition to huge demand, Wegovy also had a disruption in its supply chain. Right now, it isn’t available at all in lower doses, which is helping to spike off-label demand for Ozempic. Novo Nordisk expects to have these problems sorted out by the end of the year, with distribution following soon after.
The price of access
With a list price of $1,350 a month, Wegovy costs as much as many mortgages. And Medicaid, Medicare, and many insurance companies don’t cover it. Although obesity is a disease, the insurance industry treats weight loss as more of a vanity issue – so even if you could find the drug, you might not be able to afford it.
“We’re seeing that roughly half the prescriptions we write aren’t being covered,” Dr. Apovian says. “And for the half that are covered, we have to do prior authorization, which takes days, and it’s laborious.” In some instances, she says, insurance companies withdraw authorization after 3 months if they don’t see enough weight coming off.
It’s not like you can take Wegovy for 3 months, lose some weight, and expect it to stay off, either. The medication requires a real commitment, potentially for life. That’s because once the semaglutide leaves your system, your appetite returns. In one study, people regained two-thirds of the weight they’d lost within a year of stopping.
Many see a double standard in the insurance companies’ refusal to cover a drug that could prevent serious illness or death.
“They’re saying it’s not cost-effective to give the 42% of Americans who have a BMI over 30 Wegovy. Did they say this when statins came out?” Dr. Apovian says. “Why are they doing this with antiobesity agents? It’s the culture. The culture isn’t ready to adopt obesity as the disease that it is.”
Unpleasant side effects
Let’s assume you’re one of the lucky ones – your insurance covers Wegovy, and you can actually find some. You might discover that using it is no walk in the park. Common side effects include gastrointestinal issues like nausea, vomiting, and diarrhea.
“The way we counteract that is to start very slowly at a low dose of these medications,” Dr. Apovian says. “We only go up when the patient doesn’t have nausea or it gets better.”
Elise Davenport was excited to try Wegovy. “I did my online research. I’m the type who’s interested in early adoption, tech gadgets and stuff,” says the 40-year-old technical writer. “I wanted to try it because I’d tried so many other things that failed, or hadn’t worked long-term.”
With a BMI over 30, Ms. Davenport qualified for the drug. She signed up for an online program that guaranteed insurance coverage and started taking it in October 2021. At first, the side effects were mild, just a touch of nausea and diarrhea. And the results were impressive. She found it easy to feel satisfied with smaller portions and lost her cravings for sugar and highly processed foods. The weight fell off, roughly 5 pounds a week.
It turns out, that’s too much, too fast. Dr. Apovian and Dr. Velazquez say their patients lose more like 2 pounds each week, with careful monitoring.
By early December, Ms. Davenport’s side effects were ramping up. Because of shortages in lower dosages, the online program wasn’t able to adjust hers right away. She felt nauseated all the time, bad enough that brushing her teeth made her vomit and she had to force herself to eat. Some weeks, she managed less than 500 calories a day. Her sleep patterns became erratic. And then her depression, which medication had kept under control for years, spiraled.
“I remember sitting on the floor of my bathroom crying, thinking I’d rather carry the extra weight,” she says. “I used to take a lot of enjoyment from food, and I had none of that anymore. It was such a joyless experience at that point.”
Eventually, her dosage was reduced and the symptoms let up, but her primary care doctor encouraged her to stop. By the time she did, in March, she’d lost 55 pounds. So far, she’s gained back about 10.
More than just weight loss
Even though Ms. Davenport’s experience wasn’t a good one, with better monitoring, she’d be willing to try again. For one thing, seeing how easy it was to eat less with medical help helped to undo years of shame.
“Our culture treats obesity like a moral failing. I realized I’d been made to feel that way by doctors and programs – that I wasn’t doing enough,” she says. “This drug made me realize there are legit physiological things going on in my body, things that are often excluded from the conversation.”
Dr. Apovian and Dr. Velazquez say their patients regularly discover similar things.
“Obesity is not a disease of willpower. Medications are not the easy way out,” Dr. Velazquez says. “This is a chronic, relapsing medical condition, and because of that, we should treat it how we treat diabetes, high blood pressure, all these other conditions. We’d never hold back medication for individuals coming in with high blood pressure, tell them to work on willpower and withhold drugs they’d qualify for.”
A version of this article first appeared on WebMD.com.
Weight loss advice is everywhere you look on social media, but one trend sweeping TikTok has led to shortages of an important diabetes drug.
Ozempic, a weekly injection that helps boost insulin sensitivity in people with type 2 diabetes, also suppresses appetite, which leads to weight loss. Stories of celebrities using the drug off-label to lose a few pounds have led to an explosion of interest. And now people with diabetes – people whose lives could be saved by the drug – are having trouble finding it.
Kim Kardashian and Elon Musk
In the spring, Kim Kardashian pulled off a dramatic weight loss to fit into Marilyn Monroe’s dress for the Met Gala. Soon rumors began to circulate that she’d used Ozempic to do it. Just this week, new Twitter owner Elon Musk tweeted about his own use of Ozempic and its sibling drug, Wegovy.
Variety dubbed Ozempic “the worst kept secret in Hollywood – especially given that its most enthusiastic users are not prediabetic and do not require the drug.” The rich and famous are spending $1,200 to $1,500 a month to get access.
As so often happens, high-profile use sparked a trend. Videos on TikTok hashtagged #ozempic have amassed more than 275 million views, and #ozempicweightloss has more than 110 million.
This raises concerns about who, exactly, is watching these videos, and what message they’re receiving.
“Forty-two percent of Americans have obesity, and even more have overweight. That’s affecting our younger people and our adolescents,” says Caroline Apovian, MD, codirector of the Center for Weight Management and Wellness at the Brigham and Women’s Hospital in Boston. “They’re looking at TikTok and other social media outlets for help.”
A new study shows how damaging this can be: Researchers analyzed 1,000 videos with nutrition, food, and weight-related hashtags, with over 1 billion views combined. They found that nearly all included messages glorifying weight loss and thinness.
At last, an effective weight-loss drug
Ozempic is Danish drug company Novo Nordisk’s brand name for semaglutide, which works by mimicking a naturally occurring hormone known as GLP-1. It travels to your brain and helps you feel full on less food. That leads to weight loss. In one 68-week study, semaglutide helped people lose an average of 15% of their body weight. But it’s not a miracle drug: You still have to change your eating habits and stay physically active.
The FDA approved Ozempic to treat people with type 2 diabetes in 2017. Four years later, Novo Nordisk received the green light for a higher-dose version meant specifically for people with obesity. Wegovy is approved for use only if you have a BMI of at least 27 with one or more weight-related ailments, or a BMI of 30 or more with none.
“These drugs are dominating my practice, because they’re so effective,” says Amanda Velazquez, MD, director of obesity medicine at Cedars-Sinai Medical Center in Los Angeles. The drug is considered safe, “so the majority of patients are good candidates.”
More demand than supply
As word spread about how well Ozempic and Wegovy worked, social media posts helped drive even more people to seek out the drugs. Now demand is outpacing the supply – according to the FDA, starter doses of Ozempic will have limited availability through January.
“In Hollywood, people are losing 10 pounds, getting it for $1,500 a month, and depleting stores for people who have such severe obesity that they have congestive heart failure and diabetes,” Dr. Apovian says. “These are people who are going to die, and you’re taking it away just for cosmetic weight loss. That is deplorable.”
In addition to huge demand, Wegovy also had a disruption in its supply chain. Right now, it isn’t available at all in lower doses, which is helping to spike off-label demand for Ozempic. Novo Nordisk expects to have these problems sorted out by the end of the year, with distribution following soon after.
The price of access
With a list price of $1,350 a month, Wegovy costs as much as many mortgages. And Medicaid, Medicare, and many insurance companies don’t cover it. Although obesity is a disease, the insurance industry treats weight loss as more of a vanity issue – so even if you could find the drug, you might not be able to afford it.
“We’re seeing that roughly half the prescriptions we write aren’t being covered,” Dr. Apovian says. “And for the half that are covered, we have to do prior authorization, which takes days, and it’s laborious.” In some instances, she says, insurance companies withdraw authorization after 3 months if they don’t see enough weight coming off.
It’s not like you can take Wegovy for 3 months, lose some weight, and expect it to stay off, either. The medication requires a real commitment, potentially for life. That’s because once the semaglutide leaves your system, your appetite returns. In one study, people regained two-thirds of the weight they’d lost within a year of stopping.
Many see a double standard in the insurance companies’ refusal to cover a drug that could prevent serious illness or death.
“They’re saying it’s not cost-effective to give the 42% of Americans who have a BMI over 30 Wegovy. Did they say this when statins came out?” Dr. Apovian says. “Why are they doing this with antiobesity agents? It’s the culture. The culture isn’t ready to adopt obesity as the disease that it is.”
Unpleasant side effects
Let’s assume you’re one of the lucky ones – your insurance covers Wegovy, and you can actually find some. You might discover that using it is no walk in the park. Common side effects include gastrointestinal issues like nausea, vomiting, and diarrhea.
“The way we counteract that is to start very slowly at a low dose of these medications,” Dr. Apovian says. “We only go up when the patient doesn’t have nausea or it gets better.”
Elise Davenport was excited to try Wegovy. “I did my online research. I’m the type who’s interested in early adoption, tech gadgets and stuff,” says the 40-year-old technical writer. “I wanted to try it because I’d tried so many other things that failed, or hadn’t worked long-term.”
With a BMI over 30, Ms. Davenport qualified for the drug. She signed up for an online program that guaranteed insurance coverage and started taking it in October 2021. At first, the side effects were mild, just a touch of nausea and diarrhea. And the results were impressive. She found it easy to feel satisfied with smaller portions and lost her cravings for sugar and highly processed foods. The weight fell off, roughly 5 pounds a week.
It turns out, that’s too much, too fast. Dr. Apovian and Dr. Velazquez say their patients lose more like 2 pounds each week, with careful monitoring.
By early December, Ms. Davenport’s side effects were ramping up. Because of shortages in lower dosages, the online program wasn’t able to adjust hers right away. She felt nauseated all the time, bad enough that brushing her teeth made her vomit and she had to force herself to eat. Some weeks, she managed less than 500 calories a day. Her sleep patterns became erratic. And then her depression, which medication had kept under control for years, spiraled.
“I remember sitting on the floor of my bathroom crying, thinking I’d rather carry the extra weight,” she says. “I used to take a lot of enjoyment from food, and I had none of that anymore. It was such a joyless experience at that point.”
Eventually, her dosage was reduced and the symptoms let up, but her primary care doctor encouraged her to stop. By the time she did, in March, she’d lost 55 pounds. So far, she’s gained back about 10.
More than just weight loss
Even though Ms. Davenport’s experience wasn’t a good one, with better monitoring, she’d be willing to try again. For one thing, seeing how easy it was to eat less with medical help helped to undo years of shame.
“Our culture treats obesity like a moral failing. I realized I’d been made to feel that way by doctors and programs – that I wasn’t doing enough,” she says. “This drug made me realize there are legit physiological things going on in my body, things that are often excluded from the conversation.”
Dr. Apovian and Dr. Velazquez say their patients regularly discover similar things.
“Obesity is not a disease of willpower. Medications are not the easy way out,” Dr. Velazquez says. “This is a chronic, relapsing medical condition, and because of that, we should treat it how we treat diabetes, high blood pressure, all these other conditions. We’d never hold back medication for individuals coming in with high blood pressure, tell them to work on willpower and withhold drugs they’d qualify for.”
A version of this article first appeared on WebMD.com.
Weight loss advice is everywhere you look on social media, but one trend sweeping TikTok has led to shortages of an important diabetes drug.
Ozempic, a weekly injection that helps boost insulin sensitivity in people with type 2 diabetes, also suppresses appetite, which leads to weight loss. Stories of celebrities using the drug off-label to lose a few pounds have led to an explosion of interest. And now people with diabetes – people whose lives could be saved by the drug – are having trouble finding it.
Kim Kardashian and Elon Musk
In the spring, Kim Kardashian pulled off a dramatic weight loss to fit into Marilyn Monroe’s dress for the Met Gala. Soon rumors began to circulate that she’d used Ozempic to do it. Just this week, new Twitter owner Elon Musk tweeted about his own use of Ozempic and its sibling drug, Wegovy.
Variety dubbed Ozempic “the worst kept secret in Hollywood – especially given that its most enthusiastic users are not prediabetic and do not require the drug.” The rich and famous are spending $1,200 to $1,500 a month to get access.
As so often happens, high-profile use sparked a trend. Videos on TikTok hashtagged #ozempic have amassed more than 275 million views, and #ozempicweightloss has more than 110 million.
This raises concerns about who, exactly, is watching these videos, and what message they’re receiving.
“Forty-two percent of Americans have obesity, and even more have overweight. That’s affecting our younger people and our adolescents,” says Caroline Apovian, MD, codirector of the Center for Weight Management and Wellness at the Brigham and Women’s Hospital in Boston. “They’re looking at TikTok and other social media outlets for help.”
A new study shows how damaging this can be: Researchers analyzed 1,000 videos with nutrition, food, and weight-related hashtags, with over 1 billion views combined. They found that nearly all included messages glorifying weight loss and thinness.
At last, an effective weight-loss drug
Ozempic is Danish drug company Novo Nordisk’s brand name for semaglutide, which works by mimicking a naturally occurring hormone known as GLP-1. It travels to your brain and helps you feel full on less food. That leads to weight loss. In one 68-week study, semaglutide helped people lose an average of 15% of their body weight. But it’s not a miracle drug: You still have to change your eating habits and stay physically active.
The FDA approved Ozempic to treat people with type 2 diabetes in 2017. Four years later, Novo Nordisk received the green light for a higher-dose version meant specifically for people with obesity. Wegovy is approved for use only if you have a BMI of at least 27 with one or more weight-related ailments, or a BMI of 30 or more with none.
“These drugs are dominating my practice, because they’re so effective,” says Amanda Velazquez, MD, director of obesity medicine at Cedars-Sinai Medical Center in Los Angeles. The drug is considered safe, “so the majority of patients are good candidates.”
More demand than supply
As word spread about how well Ozempic and Wegovy worked, social media posts helped drive even more people to seek out the drugs. Now demand is outpacing the supply – according to the FDA, starter doses of Ozempic will have limited availability through January.
“In Hollywood, people are losing 10 pounds, getting it for $1,500 a month, and depleting stores for people who have such severe obesity that they have congestive heart failure and diabetes,” Dr. Apovian says. “These are people who are going to die, and you’re taking it away just for cosmetic weight loss. That is deplorable.”
In addition to huge demand, Wegovy also had a disruption in its supply chain. Right now, it isn’t available at all in lower doses, which is helping to spike off-label demand for Ozempic. Novo Nordisk expects to have these problems sorted out by the end of the year, with distribution following soon after.
The price of access
With a list price of $1,350 a month, Wegovy costs as much as many mortgages. And Medicaid, Medicare, and many insurance companies don’t cover it. Although obesity is a disease, the insurance industry treats weight loss as more of a vanity issue – so even if you could find the drug, you might not be able to afford it.
“We’re seeing that roughly half the prescriptions we write aren’t being covered,” Dr. Apovian says. “And for the half that are covered, we have to do prior authorization, which takes days, and it’s laborious.” In some instances, she says, insurance companies withdraw authorization after 3 months if they don’t see enough weight coming off.
It’s not like you can take Wegovy for 3 months, lose some weight, and expect it to stay off, either. The medication requires a real commitment, potentially for life. That’s because once the semaglutide leaves your system, your appetite returns. In one study, people regained two-thirds of the weight they’d lost within a year of stopping.
Many see a double standard in the insurance companies’ refusal to cover a drug that could prevent serious illness or death.
“They’re saying it’s not cost-effective to give the 42% of Americans who have a BMI over 30 Wegovy. Did they say this when statins came out?” Dr. Apovian says. “Why are they doing this with antiobesity agents? It’s the culture. The culture isn’t ready to adopt obesity as the disease that it is.”
Unpleasant side effects
Let’s assume you’re one of the lucky ones – your insurance covers Wegovy, and you can actually find some. You might discover that using it is no walk in the park. Common side effects include gastrointestinal issues like nausea, vomiting, and diarrhea.
“The way we counteract that is to start very slowly at a low dose of these medications,” Dr. Apovian says. “We only go up when the patient doesn’t have nausea or it gets better.”
Elise Davenport was excited to try Wegovy. “I did my online research. I’m the type who’s interested in early adoption, tech gadgets and stuff,” says the 40-year-old technical writer. “I wanted to try it because I’d tried so many other things that failed, or hadn’t worked long-term.”
With a BMI over 30, Ms. Davenport qualified for the drug. She signed up for an online program that guaranteed insurance coverage and started taking it in October 2021. At first, the side effects were mild, just a touch of nausea and diarrhea. And the results were impressive. She found it easy to feel satisfied with smaller portions and lost her cravings for sugar and highly processed foods. The weight fell off, roughly 5 pounds a week.
It turns out, that’s too much, too fast. Dr. Apovian and Dr. Velazquez say their patients lose more like 2 pounds each week, with careful monitoring.
By early December, Ms. Davenport’s side effects were ramping up. Because of shortages in lower dosages, the online program wasn’t able to adjust hers right away. She felt nauseated all the time, bad enough that brushing her teeth made her vomit and she had to force herself to eat. Some weeks, she managed less than 500 calories a day. Her sleep patterns became erratic. And then her depression, which medication had kept under control for years, spiraled.
“I remember sitting on the floor of my bathroom crying, thinking I’d rather carry the extra weight,” she says. “I used to take a lot of enjoyment from food, and I had none of that anymore. It was such a joyless experience at that point.”
Eventually, her dosage was reduced and the symptoms let up, but her primary care doctor encouraged her to stop. By the time she did, in March, she’d lost 55 pounds. So far, she’s gained back about 10.
More than just weight loss
Even though Ms. Davenport’s experience wasn’t a good one, with better monitoring, she’d be willing to try again. For one thing, seeing how easy it was to eat less with medical help helped to undo years of shame.
“Our culture treats obesity like a moral failing. I realized I’d been made to feel that way by doctors and programs – that I wasn’t doing enough,” she says. “This drug made me realize there are legit physiological things going on in my body, things that are often excluded from the conversation.”
Dr. Apovian and Dr. Velazquez say their patients regularly discover similar things.
“Obesity is not a disease of willpower. Medications are not the easy way out,” Dr. Velazquez says. “This is a chronic, relapsing medical condition, and because of that, we should treat it how we treat diabetes, high blood pressure, all these other conditions. We’d never hold back medication for individuals coming in with high blood pressure, tell them to work on willpower and withhold drugs they’d qualify for.”
A version of this article first appeared on WebMD.com.
With type 1 diabetes delay possible, focus now on screening
The recent approval of teplizumab-mzwv (Tzield, Provention Bio) for the delay of type 1 diabetes by the Food and Drug Administration is expected to advance efforts to increase screening to cost effectively identify those at risk for the condition who would be eligible to receive the new treatment.
The anti-CD3 monoclonal antibody was approved Nov. 17 as the first disease-modifying therapy for impeding progression of type 1 diabetes. In a clinical trial, teplizumab delayed the onset of clinical (stage 3) type 1 diabetes by approximately 2 years, and longer in some cases.
It is administered by intravenous infusion once daily for 14 consecutive days and is expected to cost in the region of $200,000 for the course of treatment.
The specific indication is “to delay the onset of stage 3 type 1 diabetes in adults and pediatric patients 8 years and older who currently have stage 2 type 1 diabetes.” In stage 2 type 1 diabetes, the individual has two or more islet autoantibodies and abnormal glycemia but is as yet asymptomatic. It is associated with a nearly 100% lifetime risk of progression to clinical (stage 3) type 1 diabetes and a 75% risk of developing the condition within 5 years.
Currently, most people who are screened for type 1 diabetes autoantibodies are first-degree relatives of those with the condition through TrialNet, other local programs, or more recently, a $55 test offered by the research and advocacy organization JDRF.
But because 85%-90% of people who develop type 1 diabetes don’t have first-degree relatives with the condition, broader population screening will be necessary to identify eligible candidates for teplizumab.
During an investor call on Nov. 18, Provention Bio chief commercial officer Jason Hoitt said that among the company’s “strategic initiatives” were “advancing awareness and screening for autoantibodies in at-risk individuals, and ultimately, routine screening during pediatric well visits for the general population,” as well as “[health care provider] belief in teplizumab and desire to prescribe it for their patients.”
Without broad population-based screening, first-degree relatives of people with type 1 diabetes are likely to be the first to be screened and those with stage 2 identified for receipt of teplizumab. Today, that population is estimated at about 30,000 in the United States, Mr. Hoitt said, adding, “with this approval we hope that more stage 2 patients can be readily identified so the course of the disease can be changed.”
During the call, Mr. Hoitt also announced that the wholesale acquisition cost of Tzield would be $13,850 per vial, which translates to $193,900 per 14-vial continuous regimen, anticipated to be a sufficient dose for most patients. The company also launched a program called COMPASS to help patients navigate insurance reimbursement, as well as provide some with financial assistance.
Cost aside, JDRF CEO Aaron Kowalski, PhD, said in an interview that clinicians shouldn’t doubt the value of delaying type 1 diabetes onset, even if not completely preventing it. “This is the first drug ever to treat the underlying disease. There is this undercurrent that insulin is enough. Why would you undertake an additional risk of an immunotherapy? Type 1 is hard to live with. I think sometimes the clinical community doesn’t appreciate that insulin is not enough. It’s very difficult, and opening this door is important. ... We believe very strongly that the delay of onset of type 1 diabetes is clinically meaningful. We hear that from every family we’ve talked to. Clinicians should appreciate this and not discount it.”
How would screening happen?
While the path to universal screening for type 1 diabetes risk isn’t yet clear, quite a bit of thought and research has gone into it even before teplizumab and other immune-modulating agents showed promise in forestalling the condition.
Data from a universal screening program of schoolchildren implemented in Bavaria, Germany, and a screening program in Denver, suggest that even without such an intervention, identifying people at high risk for developing type 1 diabetes could be cost effective by allowing for education of the individual and family members about the signs of type 1 diabetes, thereby reducing the likelihood that the person would progress to developing diabetic ketoacidosis (DKA) prior to diagnosis.
Another study that used data from the United States and Western Europe, found that screening children for type 1 diabetes–associated islet autoantibodies at ages 2 and 6 years would identify most of those who go on to develop the disease by midadolescence.
However, using a genetic risk score at birth to identify those who would go on to autoantibody testing is potentially a more cost-effective approach, William A. Hagopian, MD, PhD, director of diabetes programs, Pacific Northwest Research Institute, Seattle, said in an interview.
The score – based on human leukocyte antigen haplotypes and their interactions as well as non-HLA genes – can stratify nearly 80% of childhood type 1 diabetes within the top 10% of all newborns. Thus, only the top 10% would then go on to receive the more expensive autoantibody testing.
“I’ve been working with U.K. colleagues for the past 3-4 years to develop a strategy using genetic risk scores followed by autoantibody screening. I feel strongly that that’s the cost-effective way to go. It’s relatively inexpensive, scalable, and can be applied commercially in newborn screening labs. To be successful an approach must be cost effective. Payors are willing to pay for newborn screening, but not so much on testing 100% of kids for antibodies,” Dr. Hagopian said.
He is now working with Washington State newborn screening labs to demonstrate feasibility of the approach using dried blood samples from actual neonatal screening after obtaining informed consent from the mothers in postpartum wards in several hospitals. Those found to be at high risk using the genetic risk score are contacted for follow-up with autoantibody screening. The program will continue for another year and a half. “I think it actually has a chance of being accepted into their regular program,” he said.
And then, he hopes, other states will follow, and eventually, the strategy will be added to the Recommended Uniform Screening Panel for universal newborn screening programs, as recommended by the Department of Health & Human Services.
“New newborn screenings for additional diseases are implemented regularly,” Dr. Hagopian said. “Most are far less common than type 1 diabetes. So even if our approach is less than 100% sensitive, this condition is a lot more common than the many inborn errors of metabolism, so we’re still going to be identifying a lot of cases. ... This is my hope for how universal type 1 diabetes screening will unfold. I see a way this may work quite well.”
A two-pronged approach to screening could work best
Meanwhile, JDRF, which supported the teplizumab research as well as others working in the space, is focusing on both genetic and autoantibody screening, Dr. Kowalski said.
“JDRF is working on both pathways – testing kids at birth for genetic predisposition and also antibody screening. We have huge programs focused on general population antibody screening.”
Dr. Kowalski said that, while the two-pronged approach certainly is worth exploring – and JDRF is doing that – he also thinks that universal autoantibody screening could be cost effective if done efficiently, such as with less expensive assays than the one used in TrialNet.
“We have programs where you do the genetic screening and keep an eye on people. We also have programs, like the one we’re funding in Germany, that are doing broad autoantibody screening of all kids. We’re hopeful that will be very cost effective if we move to cheaper assays.”
He noted that the proportion of children with new-onset type 1 diabetes who present in DKA rose from 40% pre–COVID-19 to 50% during the early days of the pandemic. On the other hand, “With screening you can get that to near zero, like they did in Bavaria. Here [in the United States], one ICU visit for DKA [costs] $100,000.”
While JDRF and others have been working on this for years, the new availability of teplizumab will be “multifold in helping things along. ... I think you’re going to see a lot of work on the cost-effectiveness of teplizumab. I think the case will be pretty straightforward that there’s huge upside to delaying the disease from a near-term and a long-term cost perspective. This is the first time we’ve had a drug out there with a price attached to it.”
But it may not happen quickly, Kowalski cautioned. “I feel there’s a ... series of events that has to happen to drive towards universal screening. Here in the U.S. it’s complicated because we have a very discrepant health care system with all these different payers, public and private.”
During the investor call, Mr. Hoitt said that Provention Bio is also exploring use of Tzield in younger patients and newly diagnosed patients, and the potential benefit of redosing or combining with other treatments.
Mr. Hoitt is an employee of Provention Bio. Dr. Kowalski is an employee of JDRF. Dr. Hagopian has reported receiving study funding from Janssen.
A version of this article first appeared on Medscape.com.
The recent approval of teplizumab-mzwv (Tzield, Provention Bio) for the delay of type 1 diabetes by the Food and Drug Administration is expected to advance efforts to increase screening to cost effectively identify those at risk for the condition who would be eligible to receive the new treatment.
The anti-CD3 monoclonal antibody was approved Nov. 17 as the first disease-modifying therapy for impeding progression of type 1 diabetes. In a clinical trial, teplizumab delayed the onset of clinical (stage 3) type 1 diabetes by approximately 2 years, and longer in some cases.
It is administered by intravenous infusion once daily for 14 consecutive days and is expected to cost in the region of $200,000 for the course of treatment.
The specific indication is “to delay the onset of stage 3 type 1 diabetes in adults and pediatric patients 8 years and older who currently have stage 2 type 1 diabetes.” In stage 2 type 1 diabetes, the individual has two or more islet autoantibodies and abnormal glycemia but is as yet asymptomatic. It is associated with a nearly 100% lifetime risk of progression to clinical (stage 3) type 1 diabetes and a 75% risk of developing the condition within 5 years.
Currently, most people who are screened for type 1 diabetes autoantibodies are first-degree relatives of those with the condition through TrialNet, other local programs, or more recently, a $55 test offered by the research and advocacy organization JDRF.
But because 85%-90% of people who develop type 1 diabetes don’t have first-degree relatives with the condition, broader population screening will be necessary to identify eligible candidates for teplizumab.
During an investor call on Nov. 18, Provention Bio chief commercial officer Jason Hoitt said that among the company’s “strategic initiatives” were “advancing awareness and screening for autoantibodies in at-risk individuals, and ultimately, routine screening during pediatric well visits for the general population,” as well as “[health care provider] belief in teplizumab and desire to prescribe it for their patients.”
Without broad population-based screening, first-degree relatives of people with type 1 diabetes are likely to be the first to be screened and those with stage 2 identified for receipt of teplizumab. Today, that population is estimated at about 30,000 in the United States, Mr. Hoitt said, adding, “with this approval we hope that more stage 2 patients can be readily identified so the course of the disease can be changed.”
During the call, Mr. Hoitt also announced that the wholesale acquisition cost of Tzield would be $13,850 per vial, which translates to $193,900 per 14-vial continuous regimen, anticipated to be a sufficient dose for most patients. The company also launched a program called COMPASS to help patients navigate insurance reimbursement, as well as provide some with financial assistance.
Cost aside, JDRF CEO Aaron Kowalski, PhD, said in an interview that clinicians shouldn’t doubt the value of delaying type 1 diabetes onset, even if not completely preventing it. “This is the first drug ever to treat the underlying disease. There is this undercurrent that insulin is enough. Why would you undertake an additional risk of an immunotherapy? Type 1 is hard to live with. I think sometimes the clinical community doesn’t appreciate that insulin is not enough. It’s very difficult, and opening this door is important. ... We believe very strongly that the delay of onset of type 1 diabetes is clinically meaningful. We hear that from every family we’ve talked to. Clinicians should appreciate this and not discount it.”
How would screening happen?
While the path to universal screening for type 1 diabetes risk isn’t yet clear, quite a bit of thought and research has gone into it even before teplizumab and other immune-modulating agents showed promise in forestalling the condition.
Data from a universal screening program of schoolchildren implemented in Bavaria, Germany, and a screening program in Denver, suggest that even without such an intervention, identifying people at high risk for developing type 1 diabetes could be cost effective by allowing for education of the individual and family members about the signs of type 1 diabetes, thereby reducing the likelihood that the person would progress to developing diabetic ketoacidosis (DKA) prior to diagnosis.
Another study that used data from the United States and Western Europe, found that screening children for type 1 diabetes–associated islet autoantibodies at ages 2 and 6 years would identify most of those who go on to develop the disease by midadolescence.
However, using a genetic risk score at birth to identify those who would go on to autoantibody testing is potentially a more cost-effective approach, William A. Hagopian, MD, PhD, director of diabetes programs, Pacific Northwest Research Institute, Seattle, said in an interview.
The score – based on human leukocyte antigen haplotypes and their interactions as well as non-HLA genes – can stratify nearly 80% of childhood type 1 diabetes within the top 10% of all newborns. Thus, only the top 10% would then go on to receive the more expensive autoantibody testing.
“I’ve been working with U.K. colleagues for the past 3-4 years to develop a strategy using genetic risk scores followed by autoantibody screening. I feel strongly that that’s the cost-effective way to go. It’s relatively inexpensive, scalable, and can be applied commercially in newborn screening labs. To be successful an approach must be cost effective. Payors are willing to pay for newborn screening, but not so much on testing 100% of kids for antibodies,” Dr. Hagopian said.
He is now working with Washington State newborn screening labs to demonstrate feasibility of the approach using dried blood samples from actual neonatal screening after obtaining informed consent from the mothers in postpartum wards in several hospitals. Those found to be at high risk using the genetic risk score are contacted for follow-up with autoantibody screening. The program will continue for another year and a half. “I think it actually has a chance of being accepted into their regular program,” he said.
And then, he hopes, other states will follow, and eventually, the strategy will be added to the Recommended Uniform Screening Panel for universal newborn screening programs, as recommended by the Department of Health & Human Services.
“New newborn screenings for additional diseases are implemented regularly,” Dr. Hagopian said. “Most are far less common than type 1 diabetes. So even if our approach is less than 100% sensitive, this condition is a lot more common than the many inborn errors of metabolism, so we’re still going to be identifying a lot of cases. ... This is my hope for how universal type 1 diabetes screening will unfold. I see a way this may work quite well.”
A two-pronged approach to screening could work best
Meanwhile, JDRF, which supported the teplizumab research as well as others working in the space, is focusing on both genetic and autoantibody screening, Dr. Kowalski said.
“JDRF is working on both pathways – testing kids at birth for genetic predisposition and also antibody screening. We have huge programs focused on general population antibody screening.”
Dr. Kowalski said that, while the two-pronged approach certainly is worth exploring – and JDRF is doing that – he also thinks that universal autoantibody screening could be cost effective if done efficiently, such as with less expensive assays than the one used in TrialNet.
“We have programs where you do the genetic screening and keep an eye on people. We also have programs, like the one we’re funding in Germany, that are doing broad autoantibody screening of all kids. We’re hopeful that will be very cost effective if we move to cheaper assays.”
He noted that the proportion of children with new-onset type 1 diabetes who present in DKA rose from 40% pre–COVID-19 to 50% during the early days of the pandemic. On the other hand, “With screening you can get that to near zero, like they did in Bavaria. Here [in the United States], one ICU visit for DKA [costs] $100,000.”
While JDRF and others have been working on this for years, the new availability of teplizumab will be “multifold in helping things along. ... I think you’re going to see a lot of work on the cost-effectiveness of teplizumab. I think the case will be pretty straightforward that there’s huge upside to delaying the disease from a near-term and a long-term cost perspective. This is the first time we’ve had a drug out there with a price attached to it.”
But it may not happen quickly, Kowalski cautioned. “I feel there’s a ... series of events that has to happen to drive towards universal screening. Here in the U.S. it’s complicated because we have a very discrepant health care system with all these different payers, public and private.”
During the investor call, Mr. Hoitt said that Provention Bio is also exploring use of Tzield in younger patients and newly diagnosed patients, and the potential benefit of redosing or combining with other treatments.
Mr. Hoitt is an employee of Provention Bio. Dr. Kowalski is an employee of JDRF. Dr. Hagopian has reported receiving study funding from Janssen.
A version of this article first appeared on Medscape.com.
The recent approval of teplizumab-mzwv (Tzield, Provention Bio) for the delay of type 1 diabetes by the Food and Drug Administration is expected to advance efforts to increase screening to cost effectively identify those at risk for the condition who would be eligible to receive the new treatment.
The anti-CD3 monoclonal antibody was approved Nov. 17 as the first disease-modifying therapy for impeding progression of type 1 diabetes. In a clinical trial, teplizumab delayed the onset of clinical (stage 3) type 1 diabetes by approximately 2 years, and longer in some cases.
It is administered by intravenous infusion once daily for 14 consecutive days and is expected to cost in the region of $200,000 for the course of treatment.
The specific indication is “to delay the onset of stage 3 type 1 diabetes in adults and pediatric patients 8 years and older who currently have stage 2 type 1 diabetes.” In stage 2 type 1 diabetes, the individual has two or more islet autoantibodies and abnormal glycemia but is as yet asymptomatic. It is associated with a nearly 100% lifetime risk of progression to clinical (stage 3) type 1 diabetes and a 75% risk of developing the condition within 5 years.
Currently, most people who are screened for type 1 diabetes autoantibodies are first-degree relatives of those with the condition through TrialNet, other local programs, or more recently, a $55 test offered by the research and advocacy organization JDRF.
But because 85%-90% of people who develop type 1 diabetes don’t have first-degree relatives with the condition, broader population screening will be necessary to identify eligible candidates for teplizumab.
During an investor call on Nov. 18, Provention Bio chief commercial officer Jason Hoitt said that among the company’s “strategic initiatives” were “advancing awareness and screening for autoantibodies in at-risk individuals, and ultimately, routine screening during pediatric well visits for the general population,” as well as “[health care provider] belief in teplizumab and desire to prescribe it for their patients.”
Without broad population-based screening, first-degree relatives of people with type 1 diabetes are likely to be the first to be screened and those with stage 2 identified for receipt of teplizumab. Today, that population is estimated at about 30,000 in the United States, Mr. Hoitt said, adding, “with this approval we hope that more stage 2 patients can be readily identified so the course of the disease can be changed.”
During the call, Mr. Hoitt also announced that the wholesale acquisition cost of Tzield would be $13,850 per vial, which translates to $193,900 per 14-vial continuous regimen, anticipated to be a sufficient dose for most patients. The company also launched a program called COMPASS to help patients navigate insurance reimbursement, as well as provide some with financial assistance.
Cost aside, JDRF CEO Aaron Kowalski, PhD, said in an interview that clinicians shouldn’t doubt the value of delaying type 1 diabetes onset, even if not completely preventing it. “This is the first drug ever to treat the underlying disease. There is this undercurrent that insulin is enough. Why would you undertake an additional risk of an immunotherapy? Type 1 is hard to live with. I think sometimes the clinical community doesn’t appreciate that insulin is not enough. It’s very difficult, and opening this door is important. ... We believe very strongly that the delay of onset of type 1 diabetes is clinically meaningful. We hear that from every family we’ve talked to. Clinicians should appreciate this and not discount it.”
How would screening happen?
While the path to universal screening for type 1 diabetes risk isn’t yet clear, quite a bit of thought and research has gone into it even before teplizumab and other immune-modulating agents showed promise in forestalling the condition.
Data from a universal screening program of schoolchildren implemented in Bavaria, Germany, and a screening program in Denver, suggest that even without such an intervention, identifying people at high risk for developing type 1 diabetes could be cost effective by allowing for education of the individual and family members about the signs of type 1 diabetes, thereby reducing the likelihood that the person would progress to developing diabetic ketoacidosis (DKA) prior to diagnosis.
Another study that used data from the United States and Western Europe, found that screening children for type 1 diabetes–associated islet autoantibodies at ages 2 and 6 years would identify most of those who go on to develop the disease by midadolescence.
However, using a genetic risk score at birth to identify those who would go on to autoantibody testing is potentially a more cost-effective approach, William A. Hagopian, MD, PhD, director of diabetes programs, Pacific Northwest Research Institute, Seattle, said in an interview.
The score – based on human leukocyte antigen haplotypes and their interactions as well as non-HLA genes – can stratify nearly 80% of childhood type 1 diabetes within the top 10% of all newborns. Thus, only the top 10% would then go on to receive the more expensive autoantibody testing.
“I’ve been working with U.K. colleagues for the past 3-4 years to develop a strategy using genetic risk scores followed by autoantibody screening. I feel strongly that that’s the cost-effective way to go. It’s relatively inexpensive, scalable, and can be applied commercially in newborn screening labs. To be successful an approach must be cost effective. Payors are willing to pay for newborn screening, but not so much on testing 100% of kids for antibodies,” Dr. Hagopian said.
He is now working with Washington State newborn screening labs to demonstrate feasibility of the approach using dried blood samples from actual neonatal screening after obtaining informed consent from the mothers in postpartum wards in several hospitals. Those found to be at high risk using the genetic risk score are contacted for follow-up with autoantibody screening. The program will continue for another year and a half. “I think it actually has a chance of being accepted into their regular program,” he said.
And then, he hopes, other states will follow, and eventually, the strategy will be added to the Recommended Uniform Screening Panel for universal newborn screening programs, as recommended by the Department of Health & Human Services.
“New newborn screenings for additional diseases are implemented regularly,” Dr. Hagopian said. “Most are far less common than type 1 diabetes. So even if our approach is less than 100% sensitive, this condition is a lot more common than the many inborn errors of metabolism, so we’re still going to be identifying a lot of cases. ... This is my hope for how universal type 1 diabetes screening will unfold. I see a way this may work quite well.”
A two-pronged approach to screening could work best
Meanwhile, JDRF, which supported the teplizumab research as well as others working in the space, is focusing on both genetic and autoantibody screening, Dr. Kowalski said.
“JDRF is working on both pathways – testing kids at birth for genetic predisposition and also antibody screening. We have huge programs focused on general population antibody screening.”
Dr. Kowalski said that, while the two-pronged approach certainly is worth exploring – and JDRF is doing that – he also thinks that universal autoantibody screening could be cost effective if done efficiently, such as with less expensive assays than the one used in TrialNet.
“We have programs where you do the genetic screening and keep an eye on people. We also have programs, like the one we’re funding in Germany, that are doing broad autoantibody screening of all kids. We’re hopeful that will be very cost effective if we move to cheaper assays.”
He noted that the proportion of children with new-onset type 1 diabetes who present in DKA rose from 40% pre–COVID-19 to 50% during the early days of the pandemic. On the other hand, “With screening you can get that to near zero, like they did in Bavaria. Here [in the United States], one ICU visit for DKA [costs] $100,000.”
While JDRF and others have been working on this for years, the new availability of teplizumab will be “multifold in helping things along. ... I think you’re going to see a lot of work on the cost-effectiveness of teplizumab. I think the case will be pretty straightforward that there’s huge upside to delaying the disease from a near-term and a long-term cost perspective. This is the first time we’ve had a drug out there with a price attached to it.”
But it may not happen quickly, Kowalski cautioned. “I feel there’s a ... series of events that has to happen to drive towards universal screening. Here in the U.S. it’s complicated because we have a very discrepant health care system with all these different payers, public and private.”
During the investor call, Mr. Hoitt said that Provention Bio is also exploring use of Tzield in younger patients and newly diagnosed patients, and the potential benefit of redosing or combining with other treatments.
Mr. Hoitt is an employee of Provention Bio. Dr. Kowalski is an employee of JDRF. Dr. Hagopian has reported receiving study funding from Janssen.
A version of this article first appeared on Medscape.com.
Both potatoes and beans reduced insulin resistance, weight in controlled study
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
FROM THE JOURNAL OF MEDICINAL FOOD
New genetic variant linked to maturity-onset diabetes of the young
A newly discovered genetic variant that is associated with type 2 diabetes (T2D) is responsible for almost 7% of all diabetes cases in Greenland, according to a whole-genome sequencing analysis of 448 Greenlandic Inuit individuals.
The variant, identified as c.1108G>T, “has the largest population impact of any previously reported variant” within the HNF1A gene – a gene that can cause maturity-onset diabetes of the young (MODY), reported senior author Torben Hansen, MD, PhD, of the University of Copenhagen, and colleagues in The Lancet Regional Health–Europe. The c.1108G>T variant does not cause MODY, but other variants within the HNF1A gene do. However, carriers of this variant, which is present in 1.9% of the Greenlandic Inuit population and has not been found elsewhere, have normal insulin sensitivity, but decreased beta-cell function and a more than fourfold risk of developing type 2 diabetes. “This adds to a previous discovery that about 11% of all diabetes in Greenlandic Inuit is explained by a mutation in the TBC1D4 variant,” Dr. Hansen told this publication. “Thus 1 in 5 patients diagnosed with type 2 diabetes in Greenland have a specific mutation explaining their diabetes. In European populations only about 1%-2% of patients diagnosed with type 2 diabetes have a known genetic etiology.”
The finding “provides new avenues to subgroup patients, detect diabetes in family members, and pursue precision treatment trials,” noted the authors, although they acknowledged that treatment choices for individuals with this variant still need to be explored. “We know from HNF1A-mutation carriers with European ancestry that they benefit from sulfonylurea treatment,” said Dr. Hansen. “However, we have not yet done treatment studies in Inuit.” The investigators noted that “it is not always the case that variants in HNF1A result in an increased insulin secretory response to sulfonylurea. ... Whether carriers of the c.1108G>T variant could benefit from treatment with sulfonylurea should be pursued within the context of a randomized clinical trial establishing both short- and long-term efficacy of sulfonylurea in these patients.”
A total of 4,497 study participants were randomly sampled from two cross-sectional cohorts in an adult Greenlandic population health survey. Among 448 participants who had whole genome sequencing, 14 known MODY genes were screened for both previously identified as well as novel variants. This identified the c.1108G>T variant, which was then genotyped in the full cohort in order to estimate an allele frequency of 1.3% in the general Greenlandic population, and 1.9% in the Inuit component. The variant was not found in genome sequences of other populations.
The researchers then tested the association of the variant with T2D and showed strong association with T2D (odds ratio, 4.35) and higher hemoglobin A1c levels.
“This is very well-conducted and exciting research that highlights the importance of studying the genetics of diverse populations,” said Miriam Udler, MD, PhD, director of the Massachusetts General Diabetes Genetics Clinic, and assistant professor at Harvard University, both in Boston. “This manuscript builds on prior work from the researchers identifying another genetic variant specific to the Greenlandic Inuit population in the gene TBC1D4,” she added. “About 3.8% of people in this population carry two copies of the TBC1D4 variant and have about a 10-fold increased risk of diabetes. Together the two variants affect 18% of Greenlanders with diabetes.”
With its fourfold increased risk of diabetes, the new variant falls into “an ever-growing category” of “intermediate risk” genetic variants, explained Dr. Udler – “meaning that they have a large impact on diabetes risk, but cannot fully predict whether someone will get diabetes. The contribution of additional risk factors is particularly important for ‘intermediate risk’ genetic variants,” she added. “Thus, clinically, we can tell patients who have variants such as HNF1A c.1108>T that they are at substantial increased risk of diabetes, but that many will not develop diabetes. And for those who do develop diabetes, we are not yet able to advise on particular therapeutic strategies.”
Still, she emphasized, the importance of studying diverse populations with specific genetic risk factors is the end-goal of precision medicine. “An active area of research is determining whether and how to return such information about ‘intermediate risk’ variants to patients who get clinical genetic testing for diabetes, since typically only variants that are very high risk ... are returned in clinical testing reports.” Dr. Udler added that “many more such “intermediate risk’ variants likely exist in all populations, but have yet to be characterized because they are less common than HNF1A c.1108>T; however, ongoing worldwide efforts to increase the sample sizes of human genetic studies will facilitate such discovery.”
The study was funded by Novo Nordisk Foundation, Independent Research Fund Denmark, and Karen Elise Jensen’s Foundation. Dr. Hansen and Dr. Udler had no disclosures.
A newly discovered genetic variant that is associated with type 2 diabetes (T2D) is responsible for almost 7% of all diabetes cases in Greenland, according to a whole-genome sequencing analysis of 448 Greenlandic Inuit individuals.
The variant, identified as c.1108G>T, “has the largest population impact of any previously reported variant” within the HNF1A gene – a gene that can cause maturity-onset diabetes of the young (MODY), reported senior author Torben Hansen, MD, PhD, of the University of Copenhagen, and colleagues in The Lancet Regional Health–Europe. The c.1108G>T variant does not cause MODY, but other variants within the HNF1A gene do. However, carriers of this variant, which is present in 1.9% of the Greenlandic Inuit population and has not been found elsewhere, have normal insulin sensitivity, but decreased beta-cell function and a more than fourfold risk of developing type 2 diabetes. “This adds to a previous discovery that about 11% of all diabetes in Greenlandic Inuit is explained by a mutation in the TBC1D4 variant,” Dr. Hansen told this publication. “Thus 1 in 5 patients diagnosed with type 2 diabetes in Greenland have a specific mutation explaining their diabetes. In European populations only about 1%-2% of patients diagnosed with type 2 diabetes have a known genetic etiology.”
The finding “provides new avenues to subgroup patients, detect diabetes in family members, and pursue precision treatment trials,” noted the authors, although they acknowledged that treatment choices for individuals with this variant still need to be explored. “We know from HNF1A-mutation carriers with European ancestry that they benefit from sulfonylurea treatment,” said Dr. Hansen. “However, we have not yet done treatment studies in Inuit.” The investigators noted that “it is not always the case that variants in HNF1A result in an increased insulin secretory response to sulfonylurea. ... Whether carriers of the c.1108G>T variant could benefit from treatment with sulfonylurea should be pursued within the context of a randomized clinical trial establishing both short- and long-term efficacy of sulfonylurea in these patients.”
A total of 4,497 study participants were randomly sampled from two cross-sectional cohorts in an adult Greenlandic population health survey. Among 448 participants who had whole genome sequencing, 14 known MODY genes were screened for both previously identified as well as novel variants. This identified the c.1108G>T variant, which was then genotyped in the full cohort in order to estimate an allele frequency of 1.3% in the general Greenlandic population, and 1.9% in the Inuit component. The variant was not found in genome sequences of other populations.
The researchers then tested the association of the variant with T2D and showed strong association with T2D (odds ratio, 4.35) and higher hemoglobin A1c levels.
“This is very well-conducted and exciting research that highlights the importance of studying the genetics of diverse populations,” said Miriam Udler, MD, PhD, director of the Massachusetts General Diabetes Genetics Clinic, and assistant professor at Harvard University, both in Boston. “This manuscript builds on prior work from the researchers identifying another genetic variant specific to the Greenlandic Inuit population in the gene TBC1D4,” she added. “About 3.8% of people in this population carry two copies of the TBC1D4 variant and have about a 10-fold increased risk of diabetes. Together the two variants affect 18% of Greenlanders with diabetes.”
With its fourfold increased risk of diabetes, the new variant falls into “an ever-growing category” of “intermediate risk” genetic variants, explained Dr. Udler – “meaning that they have a large impact on diabetes risk, but cannot fully predict whether someone will get diabetes. The contribution of additional risk factors is particularly important for ‘intermediate risk’ genetic variants,” she added. “Thus, clinically, we can tell patients who have variants such as HNF1A c.1108>T that they are at substantial increased risk of diabetes, but that many will not develop diabetes. And for those who do develop diabetes, we are not yet able to advise on particular therapeutic strategies.”
Still, she emphasized, the importance of studying diverse populations with specific genetic risk factors is the end-goal of precision medicine. “An active area of research is determining whether and how to return such information about ‘intermediate risk’ variants to patients who get clinical genetic testing for diabetes, since typically only variants that are very high risk ... are returned in clinical testing reports.” Dr. Udler added that “many more such “intermediate risk’ variants likely exist in all populations, but have yet to be characterized because they are less common than HNF1A c.1108>T; however, ongoing worldwide efforts to increase the sample sizes of human genetic studies will facilitate such discovery.”
The study was funded by Novo Nordisk Foundation, Independent Research Fund Denmark, and Karen Elise Jensen’s Foundation. Dr. Hansen and Dr. Udler had no disclosures.
A newly discovered genetic variant that is associated with type 2 diabetes (T2D) is responsible for almost 7% of all diabetes cases in Greenland, according to a whole-genome sequencing analysis of 448 Greenlandic Inuit individuals.
The variant, identified as c.1108G>T, “has the largest population impact of any previously reported variant” within the HNF1A gene – a gene that can cause maturity-onset diabetes of the young (MODY), reported senior author Torben Hansen, MD, PhD, of the University of Copenhagen, and colleagues in The Lancet Regional Health–Europe. The c.1108G>T variant does not cause MODY, but other variants within the HNF1A gene do. However, carriers of this variant, which is present in 1.9% of the Greenlandic Inuit population and has not been found elsewhere, have normal insulin sensitivity, but decreased beta-cell function and a more than fourfold risk of developing type 2 diabetes. “This adds to a previous discovery that about 11% of all diabetes in Greenlandic Inuit is explained by a mutation in the TBC1D4 variant,” Dr. Hansen told this publication. “Thus 1 in 5 patients diagnosed with type 2 diabetes in Greenland have a specific mutation explaining their diabetes. In European populations only about 1%-2% of patients diagnosed with type 2 diabetes have a known genetic etiology.”
The finding “provides new avenues to subgroup patients, detect diabetes in family members, and pursue precision treatment trials,” noted the authors, although they acknowledged that treatment choices for individuals with this variant still need to be explored. “We know from HNF1A-mutation carriers with European ancestry that they benefit from sulfonylurea treatment,” said Dr. Hansen. “However, we have not yet done treatment studies in Inuit.” The investigators noted that “it is not always the case that variants in HNF1A result in an increased insulin secretory response to sulfonylurea. ... Whether carriers of the c.1108G>T variant could benefit from treatment with sulfonylurea should be pursued within the context of a randomized clinical trial establishing both short- and long-term efficacy of sulfonylurea in these patients.”
A total of 4,497 study participants were randomly sampled from two cross-sectional cohorts in an adult Greenlandic population health survey. Among 448 participants who had whole genome sequencing, 14 known MODY genes were screened for both previously identified as well as novel variants. This identified the c.1108G>T variant, which was then genotyped in the full cohort in order to estimate an allele frequency of 1.3% in the general Greenlandic population, and 1.9% in the Inuit component. The variant was not found in genome sequences of other populations.
The researchers then tested the association of the variant with T2D and showed strong association with T2D (odds ratio, 4.35) and higher hemoglobin A1c levels.
“This is very well-conducted and exciting research that highlights the importance of studying the genetics of diverse populations,” said Miriam Udler, MD, PhD, director of the Massachusetts General Diabetes Genetics Clinic, and assistant professor at Harvard University, both in Boston. “This manuscript builds on prior work from the researchers identifying another genetic variant specific to the Greenlandic Inuit population in the gene TBC1D4,” she added. “About 3.8% of people in this population carry two copies of the TBC1D4 variant and have about a 10-fold increased risk of diabetes. Together the two variants affect 18% of Greenlanders with diabetes.”
With its fourfold increased risk of diabetes, the new variant falls into “an ever-growing category” of “intermediate risk” genetic variants, explained Dr. Udler – “meaning that they have a large impact on diabetes risk, but cannot fully predict whether someone will get diabetes. The contribution of additional risk factors is particularly important for ‘intermediate risk’ genetic variants,” she added. “Thus, clinically, we can tell patients who have variants such as HNF1A c.1108>T that they are at substantial increased risk of diabetes, but that many will not develop diabetes. And for those who do develop diabetes, we are not yet able to advise on particular therapeutic strategies.”
Still, she emphasized, the importance of studying diverse populations with specific genetic risk factors is the end-goal of precision medicine. “An active area of research is determining whether and how to return such information about ‘intermediate risk’ variants to patients who get clinical genetic testing for diabetes, since typically only variants that are very high risk ... are returned in clinical testing reports.” Dr. Udler added that “many more such “intermediate risk’ variants likely exist in all populations, but have yet to be characterized because they are less common than HNF1A c.1108>T; however, ongoing worldwide efforts to increase the sample sizes of human genetic studies will facilitate such discovery.”
The study was funded by Novo Nordisk Foundation, Independent Research Fund Denmark, and Karen Elise Jensen’s Foundation. Dr. Hansen and Dr. Udler had no disclosures.
FROM THE LANCET REGIONAL HEALTH–EUROPE
Will ICER review aid bid for Medicare to pay for obesity drugs?
A report from a well-respected nonprofit group may bolster efforts to have Medicare, the largest U.S. purchaser of prescription drugs, cover obesity medicines, for which there has been accumulating evidence of significant benefit.
The Institute for Clinical and Economic Review (ICER) released a report last month on obesity medicines, based on extensive review of research done to date and input from clinicians, drug-makers, and members of the public.
Of the treatments reviewed, the ICER report gave the best ratings to two Novo Nordisk products, a B+ for semaglutide (Wegovy) and a B for liraglutide (Saxenda), while also making the case for price cuts. At an annual U.S. net price estimated at $13,618, semaglutide exceeds what ICER considers typical cost-effectiveness thresholds. ICER suggested a benchmark annual price range for semaglutide of between $7,500 and $9,800.
The ICER report also directs insurers in general to provide more generous coverage of obesity medicines, with a specific recommendation for the U.S. Congress to pass a pending bill known as the Treat and Reduce Obesity Act of 2021. The bill would undo a restriction on weight-loss drugs in the Medicare Part D plans, which covered about 49 million people last year. Sen. Tom Carper (D-Del.) and Sen. Bill Cassidy, MD, (R-La.) have repeatedly introduced versions of the bill since 2013.
“In both chambers of Congress and with bipartisan support, we’ve pushed to expand Medicare coverage of additional therapies and medications to treat obesity,” Sen. Cassidy said in an email. “This report confirms what we’ve worked on for nearly a decade – our legislation will help improve lives.”
The current House version of the bill has the backing of more than a third of the members of that chamber, with 113 Democratic and 40 Republican cosponsors. The Senate version has 22 sponsors.
Changing views
The ICER report comes amid a broader change in how clinicians view obesity.
The American Academy of Pediatrics is readying a new Clinical Practice Guideline for the Evaluation and Treatment of Pediatric Obesity that will mark a major shift in approach. Aaron S. Kelly, PhD, a professor of pediatrics at the University of Minnesota, Minneapolis, described it as a “sea change,” with obesity now seen as “a chronic, refractory, relapsing disease,” for which watchful waiting is no longer appropriate.
But the field of obesity treatment looked quite different in the early 2000s when Congress worked on a plan to add a pharmacy benefit to Medicare.
The deliberate omission of obesity medicine in the Medicare Part D benefit reflected both the state of science at the time and U.S. experience with a dangerous weight-loss drug combo in the late 1990s.
Initial expectations for weight-loss pills were high after the Food and Drug Administration cleared dexfenfluramine HCl (Redux) in 1996, which was part of the popular fen-phen combination. “Newly Approved Diet Drug Promises to Help Millions of Obese Americans – But Is No Magic Bullet,” read a headline about the Redux approval in The Washington Post
When work began in the 2000s to create a Medicare pharmacy benefit, lawmakers and congressional staff had a pool of about $400 billion available to establish what became the Part D program, Joel White, a former House staffer who helped draft the law, told this news organization in an email exchange.
Given the state of obesity research at the time, it seemed to make sense to exclude weight-loss medications, wrote Mr. White. Mr. White is now chief executive of the consulting firm Horizon, which has clients in the drug industry including the Pharmaceutical Research and Manufacturers of America.
“Now we know that obesity is a chronic disease of epidemic proportions. Decades of research have produced a series of advances in the way we understand and treat obesity. While scientists and many who work directly with those impacted by this epidemic understand how treatments have advanced, the law lags behind,” Mr. White said.
XXXCurrent payment policies for obesity treatments are based on “outdated information and ongoing misperception,” he noted. “While Part D has been a resounding success, our Medicare approach to obesity is not.”
“In addition, it makes no sense that Medicare covers the most drastic procedure (bariatric surgery) but not less-invasive, effective treatments,” he added. “We should have long ago lifted restrictions based on advances in science and medicine.”
Overcoming the stigma
Scott Kahan, MD, MPH, agreed and hopes that the new ICER report will help more patients secure needed medications, raising a “call to arms” about the need for better coverage of obesity drugs.
Dr. Kahan is director of the National Center for Weight and Wellness, a private clinic in Washington, and chair of the clinical committee for The Obesity Society. He also served as a member of a policy roundtable that ICER convened as part of research on the report on obesity drugs. Dr. Kahan, who also serves on the faculty at the Johns Hopkins Bloomberg School of Public Health, Baltimore, has received fees from drug makers such as Eli Lilly.
The ICER report may help what Dr. Kahan described as well-founded caution about obesity treatments in general.
“When it comes to weight loss, there are all of these magical treatments that are sold on social media and traditional media. There are a lot of bad actors in terms of people calling themselves experts and gurus and promising all kinds of crazy stuff,” said Dr. Kahan.
And there are long-standing stigmas about obesity, he stressed.
“That underlies a lot of the backward policies, including poor coverage for medications and the noncoverage by Medicare,” Dr. Kahan said. “There’s a societal ingrained set of beliefs and misperceptions and biases. That takes time to unwind, and I think we’re on the way, but we’re not quite there yet.”
Lifestyle changes not enough to tackle obesity
AHIP (formerly America’s Health Insurance Plans) told this news organization its members consider ICER reports when making decisions about which products to cover. “And health plans already cover obesity treatments that they consider medically necessary,” said David Allen, an AHIP spokesperson.
“It is important to note that every treatment does not work for every patient, and many patients experience adverse events and may discontinue treatment,” he added in an email. “Health insurance providers play an important role in helping [health care] providers and patients identify the treatment options that are most likely to be effective as well as affordable.”
Separately, the nonprofit watchdog group Public Citizen cautioned against liraglutide on its Worst Pills, Best Pills website. In its view, the drug is minimally effective and has many dangerous adverse effects, which are even more frequent with the higher-dose weight-loss version (a lower-dose version is approved for type 2 diabetes).
“There is currently no medication that can be used safely to achieve weight loss effortlessly and without dangerous adverse effects,” the group said. “Rather than focus on losing weight by turning to risky drugs, overweight and obese adults seeking to achieve better health should make reasonable and sustainable changes to their lifestyle, such as eating a healthy diet and getting regular exercise.”
Yet, many people find there is little help available for making lifestyle changes, and some patients and physicians say these modifications by themselves are not enough.
“The vast majority of people with obesity cannot achieve sustained weight loss through diet and exercise alone,” said David Rind, MD, chief medical officer of ICER, in an Oct. 20 statement. “As such, obesity, and its resulting physical health, mental health, and social burdens, is not a choice or failing, but a medical condition.”
The focus should now be on assuring that effective medications “are priced in alignment with their benefits so that they are accessible and affordable across U.S. society,” Dr. Rind urges.
‘My own demise with a fork and knife’
ICER sought public feedback on a draft version of the report before finalizing it.
In their comments on ICER’s work, several pharmaceutical researchers and Novo Nordisk questioned the calculations used in making judgments about the value of obesity drugs. In a statement, Novo Nordisk told this news organization that the company’s view is that ICER’s modeling “does not adequately address the real-world complexities of obesity, and consequently underestimates the health and societal impact medical treatments can have.”
Commenters also dug into aspects of ICER’s calculations, including ones that consider quality-adjusted life-years (QALYs). ICER describes QALY as an academic standard for measuring how well all different types of medical treatments can extend or improve patients’ lives. In an explainer on its website, ICER says this metric has served as a fundamental component of cost-effectiveness analyses in the United States and around the world for more than 30 years.
ICER and drug makers have been at odds for some time, with PhRMA having criticized the nonprofit group. A 2020 Reuters article detailed public relations strategies used by firms paid by drug makers to raise questions about ICER’s work. Critics accuse it of allying with insurers.
ICER’s list of its recent financial supporters includes Blue Cross Blue Shield of Massachusetts and the Kaiser Foundation Health Plan, but also many other groups, such as the U.S. Department of Veterans Affairs, the American Academy of Neurology, and the American College of Rheumatology.
The public comments on the ICER report also include one from an unidentified woman who wrote of her past struggles to lose weight.
She said her health plan wouldn’t cover behavioral programs or semaglutide as a weight-loss drug but did cover it eventually because of signs that she had developed insulin resistance. The patient said the drug worked for her, whereas other approaches to control weight had failed.
“To put it simply, I now experience hunger and satiety in a way that I can only assume people with normal metabolism do. I am 49 years old and approaching the age where serious comorbidities associated with obesity begin to manifest,” the patient wrote.
“I no longer worry about bringing about my own demise with a fork and knife because of misfiring hunger cues.”
A version of this article first appeared on Medscape.com.
A report from a well-respected nonprofit group may bolster efforts to have Medicare, the largest U.S. purchaser of prescription drugs, cover obesity medicines, for which there has been accumulating evidence of significant benefit.
The Institute for Clinical and Economic Review (ICER) released a report last month on obesity medicines, based on extensive review of research done to date and input from clinicians, drug-makers, and members of the public.
Of the treatments reviewed, the ICER report gave the best ratings to two Novo Nordisk products, a B+ for semaglutide (Wegovy) and a B for liraglutide (Saxenda), while also making the case for price cuts. At an annual U.S. net price estimated at $13,618, semaglutide exceeds what ICER considers typical cost-effectiveness thresholds. ICER suggested a benchmark annual price range for semaglutide of between $7,500 and $9,800.
The ICER report also directs insurers in general to provide more generous coverage of obesity medicines, with a specific recommendation for the U.S. Congress to pass a pending bill known as the Treat and Reduce Obesity Act of 2021. The bill would undo a restriction on weight-loss drugs in the Medicare Part D plans, which covered about 49 million people last year. Sen. Tom Carper (D-Del.) and Sen. Bill Cassidy, MD, (R-La.) have repeatedly introduced versions of the bill since 2013.
“In both chambers of Congress and with bipartisan support, we’ve pushed to expand Medicare coverage of additional therapies and medications to treat obesity,” Sen. Cassidy said in an email. “This report confirms what we’ve worked on for nearly a decade – our legislation will help improve lives.”
The current House version of the bill has the backing of more than a third of the members of that chamber, with 113 Democratic and 40 Republican cosponsors. The Senate version has 22 sponsors.
Changing views
The ICER report comes amid a broader change in how clinicians view obesity.
The American Academy of Pediatrics is readying a new Clinical Practice Guideline for the Evaluation and Treatment of Pediatric Obesity that will mark a major shift in approach. Aaron S. Kelly, PhD, a professor of pediatrics at the University of Minnesota, Minneapolis, described it as a “sea change,” with obesity now seen as “a chronic, refractory, relapsing disease,” for which watchful waiting is no longer appropriate.
But the field of obesity treatment looked quite different in the early 2000s when Congress worked on a plan to add a pharmacy benefit to Medicare.
The deliberate omission of obesity medicine in the Medicare Part D benefit reflected both the state of science at the time and U.S. experience with a dangerous weight-loss drug combo in the late 1990s.
Initial expectations for weight-loss pills were high after the Food and Drug Administration cleared dexfenfluramine HCl (Redux) in 1996, which was part of the popular fen-phen combination. “Newly Approved Diet Drug Promises to Help Millions of Obese Americans – But Is No Magic Bullet,” read a headline about the Redux approval in The Washington Post
When work began in the 2000s to create a Medicare pharmacy benefit, lawmakers and congressional staff had a pool of about $400 billion available to establish what became the Part D program, Joel White, a former House staffer who helped draft the law, told this news organization in an email exchange.
Given the state of obesity research at the time, it seemed to make sense to exclude weight-loss medications, wrote Mr. White. Mr. White is now chief executive of the consulting firm Horizon, which has clients in the drug industry including the Pharmaceutical Research and Manufacturers of America.
“Now we know that obesity is a chronic disease of epidemic proportions. Decades of research have produced a series of advances in the way we understand and treat obesity. While scientists and many who work directly with those impacted by this epidemic understand how treatments have advanced, the law lags behind,” Mr. White said.
XXXCurrent payment policies for obesity treatments are based on “outdated information and ongoing misperception,” he noted. “While Part D has been a resounding success, our Medicare approach to obesity is not.”
“In addition, it makes no sense that Medicare covers the most drastic procedure (bariatric surgery) but not less-invasive, effective treatments,” he added. “We should have long ago lifted restrictions based on advances in science and medicine.”
Overcoming the stigma
Scott Kahan, MD, MPH, agreed and hopes that the new ICER report will help more patients secure needed medications, raising a “call to arms” about the need for better coverage of obesity drugs.
Dr. Kahan is director of the National Center for Weight and Wellness, a private clinic in Washington, and chair of the clinical committee for The Obesity Society. He also served as a member of a policy roundtable that ICER convened as part of research on the report on obesity drugs. Dr. Kahan, who also serves on the faculty at the Johns Hopkins Bloomberg School of Public Health, Baltimore, has received fees from drug makers such as Eli Lilly.
The ICER report may help what Dr. Kahan described as well-founded caution about obesity treatments in general.
“When it comes to weight loss, there are all of these magical treatments that are sold on social media and traditional media. There are a lot of bad actors in terms of people calling themselves experts and gurus and promising all kinds of crazy stuff,” said Dr. Kahan.
And there are long-standing stigmas about obesity, he stressed.
“That underlies a lot of the backward policies, including poor coverage for medications and the noncoverage by Medicare,” Dr. Kahan said. “There’s a societal ingrained set of beliefs and misperceptions and biases. That takes time to unwind, and I think we’re on the way, but we’re not quite there yet.”
Lifestyle changes not enough to tackle obesity
AHIP (formerly America’s Health Insurance Plans) told this news organization its members consider ICER reports when making decisions about which products to cover. “And health plans already cover obesity treatments that they consider medically necessary,” said David Allen, an AHIP spokesperson.
“It is important to note that every treatment does not work for every patient, and many patients experience adverse events and may discontinue treatment,” he added in an email. “Health insurance providers play an important role in helping [health care] providers and patients identify the treatment options that are most likely to be effective as well as affordable.”
Separately, the nonprofit watchdog group Public Citizen cautioned against liraglutide on its Worst Pills, Best Pills website. In its view, the drug is minimally effective and has many dangerous adverse effects, which are even more frequent with the higher-dose weight-loss version (a lower-dose version is approved for type 2 diabetes).
“There is currently no medication that can be used safely to achieve weight loss effortlessly and without dangerous adverse effects,” the group said. “Rather than focus on losing weight by turning to risky drugs, overweight and obese adults seeking to achieve better health should make reasonable and sustainable changes to their lifestyle, such as eating a healthy diet and getting regular exercise.”
Yet, many people find there is little help available for making lifestyle changes, and some patients and physicians say these modifications by themselves are not enough.
“The vast majority of people with obesity cannot achieve sustained weight loss through diet and exercise alone,” said David Rind, MD, chief medical officer of ICER, in an Oct. 20 statement. “As such, obesity, and its resulting physical health, mental health, and social burdens, is not a choice or failing, but a medical condition.”
The focus should now be on assuring that effective medications “are priced in alignment with their benefits so that they are accessible and affordable across U.S. society,” Dr. Rind urges.
‘My own demise with a fork and knife’
ICER sought public feedback on a draft version of the report before finalizing it.
In their comments on ICER’s work, several pharmaceutical researchers and Novo Nordisk questioned the calculations used in making judgments about the value of obesity drugs. In a statement, Novo Nordisk told this news organization that the company’s view is that ICER’s modeling “does not adequately address the real-world complexities of obesity, and consequently underestimates the health and societal impact medical treatments can have.”
Commenters also dug into aspects of ICER’s calculations, including ones that consider quality-adjusted life-years (QALYs). ICER describes QALY as an academic standard for measuring how well all different types of medical treatments can extend or improve patients’ lives. In an explainer on its website, ICER says this metric has served as a fundamental component of cost-effectiveness analyses in the United States and around the world for more than 30 years.
ICER and drug makers have been at odds for some time, with PhRMA having criticized the nonprofit group. A 2020 Reuters article detailed public relations strategies used by firms paid by drug makers to raise questions about ICER’s work. Critics accuse it of allying with insurers.
ICER’s list of its recent financial supporters includes Blue Cross Blue Shield of Massachusetts and the Kaiser Foundation Health Plan, but also many other groups, such as the U.S. Department of Veterans Affairs, the American Academy of Neurology, and the American College of Rheumatology.
The public comments on the ICER report also include one from an unidentified woman who wrote of her past struggles to lose weight.
She said her health plan wouldn’t cover behavioral programs or semaglutide as a weight-loss drug but did cover it eventually because of signs that she had developed insulin resistance. The patient said the drug worked for her, whereas other approaches to control weight had failed.
“To put it simply, I now experience hunger and satiety in a way that I can only assume people with normal metabolism do. I am 49 years old and approaching the age where serious comorbidities associated with obesity begin to manifest,” the patient wrote.
“I no longer worry about bringing about my own demise with a fork and knife because of misfiring hunger cues.”
A version of this article first appeared on Medscape.com.
A report from a well-respected nonprofit group may bolster efforts to have Medicare, the largest U.S. purchaser of prescription drugs, cover obesity medicines, for which there has been accumulating evidence of significant benefit.
The Institute for Clinical and Economic Review (ICER) released a report last month on obesity medicines, based on extensive review of research done to date and input from clinicians, drug-makers, and members of the public.
Of the treatments reviewed, the ICER report gave the best ratings to two Novo Nordisk products, a B+ for semaglutide (Wegovy) and a B for liraglutide (Saxenda), while also making the case for price cuts. At an annual U.S. net price estimated at $13,618, semaglutide exceeds what ICER considers typical cost-effectiveness thresholds. ICER suggested a benchmark annual price range for semaglutide of between $7,500 and $9,800.
The ICER report also directs insurers in general to provide more generous coverage of obesity medicines, with a specific recommendation for the U.S. Congress to pass a pending bill known as the Treat and Reduce Obesity Act of 2021. The bill would undo a restriction on weight-loss drugs in the Medicare Part D plans, which covered about 49 million people last year. Sen. Tom Carper (D-Del.) and Sen. Bill Cassidy, MD, (R-La.) have repeatedly introduced versions of the bill since 2013.
“In both chambers of Congress and with bipartisan support, we’ve pushed to expand Medicare coverage of additional therapies and medications to treat obesity,” Sen. Cassidy said in an email. “This report confirms what we’ve worked on for nearly a decade – our legislation will help improve lives.”
The current House version of the bill has the backing of more than a third of the members of that chamber, with 113 Democratic and 40 Republican cosponsors. The Senate version has 22 sponsors.
Changing views
The ICER report comes amid a broader change in how clinicians view obesity.
The American Academy of Pediatrics is readying a new Clinical Practice Guideline for the Evaluation and Treatment of Pediatric Obesity that will mark a major shift in approach. Aaron S. Kelly, PhD, a professor of pediatrics at the University of Minnesota, Minneapolis, described it as a “sea change,” with obesity now seen as “a chronic, refractory, relapsing disease,” for which watchful waiting is no longer appropriate.
But the field of obesity treatment looked quite different in the early 2000s when Congress worked on a plan to add a pharmacy benefit to Medicare.
The deliberate omission of obesity medicine in the Medicare Part D benefit reflected both the state of science at the time and U.S. experience with a dangerous weight-loss drug combo in the late 1990s.
Initial expectations for weight-loss pills were high after the Food and Drug Administration cleared dexfenfluramine HCl (Redux) in 1996, which was part of the popular fen-phen combination. “Newly Approved Diet Drug Promises to Help Millions of Obese Americans – But Is No Magic Bullet,” read a headline about the Redux approval in The Washington Post
When work began in the 2000s to create a Medicare pharmacy benefit, lawmakers and congressional staff had a pool of about $400 billion available to establish what became the Part D program, Joel White, a former House staffer who helped draft the law, told this news organization in an email exchange.
Given the state of obesity research at the time, it seemed to make sense to exclude weight-loss medications, wrote Mr. White. Mr. White is now chief executive of the consulting firm Horizon, which has clients in the drug industry including the Pharmaceutical Research and Manufacturers of America.
“Now we know that obesity is a chronic disease of epidemic proportions. Decades of research have produced a series of advances in the way we understand and treat obesity. While scientists and many who work directly with those impacted by this epidemic understand how treatments have advanced, the law lags behind,” Mr. White said.
XXXCurrent payment policies for obesity treatments are based on “outdated information and ongoing misperception,” he noted. “While Part D has been a resounding success, our Medicare approach to obesity is not.”
“In addition, it makes no sense that Medicare covers the most drastic procedure (bariatric surgery) but not less-invasive, effective treatments,” he added. “We should have long ago lifted restrictions based on advances in science and medicine.”
Overcoming the stigma
Scott Kahan, MD, MPH, agreed and hopes that the new ICER report will help more patients secure needed medications, raising a “call to arms” about the need for better coverage of obesity drugs.
Dr. Kahan is director of the National Center for Weight and Wellness, a private clinic in Washington, and chair of the clinical committee for The Obesity Society. He also served as a member of a policy roundtable that ICER convened as part of research on the report on obesity drugs. Dr. Kahan, who also serves on the faculty at the Johns Hopkins Bloomberg School of Public Health, Baltimore, has received fees from drug makers such as Eli Lilly.
The ICER report may help what Dr. Kahan described as well-founded caution about obesity treatments in general.
“When it comes to weight loss, there are all of these magical treatments that are sold on social media and traditional media. There are a lot of bad actors in terms of people calling themselves experts and gurus and promising all kinds of crazy stuff,” said Dr. Kahan.
And there are long-standing stigmas about obesity, he stressed.
“That underlies a lot of the backward policies, including poor coverage for medications and the noncoverage by Medicare,” Dr. Kahan said. “There’s a societal ingrained set of beliefs and misperceptions and biases. That takes time to unwind, and I think we’re on the way, but we’re not quite there yet.”
Lifestyle changes not enough to tackle obesity
AHIP (formerly America’s Health Insurance Plans) told this news organization its members consider ICER reports when making decisions about which products to cover. “And health plans already cover obesity treatments that they consider medically necessary,” said David Allen, an AHIP spokesperson.
“It is important to note that every treatment does not work for every patient, and many patients experience adverse events and may discontinue treatment,” he added in an email. “Health insurance providers play an important role in helping [health care] providers and patients identify the treatment options that are most likely to be effective as well as affordable.”
Separately, the nonprofit watchdog group Public Citizen cautioned against liraglutide on its Worst Pills, Best Pills website. In its view, the drug is minimally effective and has many dangerous adverse effects, which are even more frequent with the higher-dose weight-loss version (a lower-dose version is approved for type 2 diabetes).
“There is currently no medication that can be used safely to achieve weight loss effortlessly and without dangerous adverse effects,” the group said. “Rather than focus on losing weight by turning to risky drugs, overweight and obese adults seeking to achieve better health should make reasonable and sustainable changes to their lifestyle, such as eating a healthy diet and getting regular exercise.”
Yet, many people find there is little help available for making lifestyle changes, and some patients and physicians say these modifications by themselves are not enough.
“The vast majority of people with obesity cannot achieve sustained weight loss through diet and exercise alone,” said David Rind, MD, chief medical officer of ICER, in an Oct. 20 statement. “As such, obesity, and its resulting physical health, mental health, and social burdens, is not a choice or failing, but a medical condition.”
The focus should now be on assuring that effective medications “are priced in alignment with their benefits so that they are accessible and affordable across U.S. society,” Dr. Rind urges.
‘My own demise with a fork and knife’
ICER sought public feedback on a draft version of the report before finalizing it.
In their comments on ICER’s work, several pharmaceutical researchers and Novo Nordisk questioned the calculations used in making judgments about the value of obesity drugs. In a statement, Novo Nordisk told this news organization that the company’s view is that ICER’s modeling “does not adequately address the real-world complexities of obesity, and consequently underestimates the health and societal impact medical treatments can have.”
Commenters also dug into aspects of ICER’s calculations, including ones that consider quality-adjusted life-years (QALYs). ICER describes QALY as an academic standard for measuring how well all different types of medical treatments can extend or improve patients’ lives. In an explainer on its website, ICER says this metric has served as a fundamental component of cost-effectiveness analyses in the United States and around the world for more than 30 years.
ICER and drug makers have been at odds for some time, with PhRMA having criticized the nonprofit group. A 2020 Reuters article detailed public relations strategies used by firms paid by drug makers to raise questions about ICER’s work. Critics accuse it of allying with insurers.
ICER’s list of its recent financial supporters includes Blue Cross Blue Shield of Massachusetts and the Kaiser Foundation Health Plan, but also many other groups, such as the U.S. Department of Veterans Affairs, the American Academy of Neurology, and the American College of Rheumatology.
The public comments on the ICER report also include one from an unidentified woman who wrote of her past struggles to lose weight.
She said her health plan wouldn’t cover behavioral programs or semaglutide as a weight-loss drug but did cover it eventually because of signs that she had developed insulin resistance. The patient said the drug worked for her, whereas other approaches to control weight had failed.
“To put it simply, I now experience hunger and satiety in a way that I can only assume people with normal metabolism do. I am 49 years old and approaching the age where serious comorbidities associated with obesity begin to manifest,” the patient wrote.
“I no longer worry about bringing about my own demise with a fork and knife because of misfiring hunger cues.”
A version of this article first appeared on Medscape.com.
Night lights in the city link to increased risk of diabetes
Higher levels of exposure to outdoor artificial light at night are significantly linked with markers of diabetes and impaired glucose homeostasis, in a new national, cross-sectional study from China.
The results showed a 7% significant increase in diabetes prevalence per quintile exposure to artificial light at night (prevalence ratio, 1.07), report Ruizhi Zheng, PhD, of the Shanghai (China) Jiaotong University School of Medicine, and colleagues. People living in areas with the most exposure to light at night had a 28% higher prevalence of diabetes than those living in places with the lowest exposure (PR, 1.28), the researchers found.
The study was published online in Diabetologia.
Previous animal studies have shown that exposure to light at night may interfere with circadian rhythms and affect glucose homeostasis, the study team note. Other research has demonstrated that chronic exposure to moderate indoor light during sleep elevated the prevalence of diabetes in older adults, compared with those sleeping in a dim setting, the authors add.
“Our findings contribute to the growing literature suggesting that artificial light at night is detrimental to health and demonstrate that artificial light at night may be a potential novel risk factor for diabetes,” they write.
“Considering the coexistence of the diabetes epidemic and the widespread influence of light pollution at night, the positive associations indicate an urgent need for countries and governments to develop effective prevention and intervention policies and to protect people from the adverse health effects of light pollution at night,” the study authors stress.
Gareth Nye, PhD, senior lecturer at the University of Chester, England, agreed that prior research has found an association between metabolic conditions, such as diabetes, and artificial light at night, with most theories as to the cause focusing on the body’s natural circadian cycle.
He said that internal clocks regulate a variety of bodily processes, such as metabolism and hormone synthesis. They also affect sleep patterns by interfering with synthesis of the hormone melatonin, which is essential for sound sleep, Dr. Nye told the UK Science Media Centre.
However, he stressed that much more research is needed before any link can be considered definitive.
Outdoor night light exposure linked to fasting glucose, A1c
The Chinese researchers set out to approximate the relationships between diabetes prevalence and glucose homeostasis with chronic exposure to outdoor light at night.
They assessed 98,658 participants from the China Noncommunicable Disease Surveillance Study across 162 sites. The mean age of participants was 42.7 years. Female participants comprised 49.2% of the study cohort.
Diabetes was defined based on American Diabetes Association criteria. Satellite data were used to determine exposure to outdoor light at night in 2010. The associations between light exposure at night and indicators of glucose homeostasis were investigated.
Prevalence ratios were calculated and adjusted for sex, age, smoking status, education, body mass index, physical activity, household income, family history of diabetes, rural/urban areas, drinking status, and use of lipid-lowering prescription drugs (primarily statins) or antihypertensives.
The findings showed exposure levels to outdoor light at night were positively linked with 2-hour and fasting glucose concentrations, A1c, and insulin resistance (measured using homeostatic model assessment [HOMA]), but negatively related to β-cell function (measured using HOMA).
More research needed
“We advise caution against causal interpretation of the findings and call for further studies involving direct measurement of individual exposure to light at night,” the researchers conclude.
Dr. Nye agreed.
“One issue with this study is that the areas with the highest outdoor artificial light levels are likely to be those in urban areas and bigger cities. It has been known for a long time now that living in an urbanized area increases your risk of obesity through increased access to high-fat and convenience food, less physical activity levels due to transport links, and less social activities. The authors also state this and the fact participants tended to be older,” he noted.
Large datasets are used in this investigation, however, which generally increases the reliability of the data, he observed.
But it is also “unclear as to whether the population here was selected for this study or was retrospectively analyzed, which poses reliability issues, as does the selection of the representative sample, as it is not discussed,” he noted.
Ultimately, there is no confirmed evidence of the link, and until further work is done to directly link light exposure and diabetes in humans, “the link will remain an association only,” he concluded.
A version of this article first appeared on Medscape.com.
Higher levels of exposure to outdoor artificial light at night are significantly linked with markers of diabetes and impaired glucose homeostasis, in a new national, cross-sectional study from China.
The results showed a 7% significant increase in diabetes prevalence per quintile exposure to artificial light at night (prevalence ratio, 1.07), report Ruizhi Zheng, PhD, of the Shanghai (China) Jiaotong University School of Medicine, and colleagues. People living in areas with the most exposure to light at night had a 28% higher prevalence of diabetes than those living in places with the lowest exposure (PR, 1.28), the researchers found.
The study was published online in Diabetologia.
Previous animal studies have shown that exposure to light at night may interfere with circadian rhythms and affect glucose homeostasis, the study team note. Other research has demonstrated that chronic exposure to moderate indoor light during sleep elevated the prevalence of diabetes in older adults, compared with those sleeping in a dim setting, the authors add.
“Our findings contribute to the growing literature suggesting that artificial light at night is detrimental to health and demonstrate that artificial light at night may be a potential novel risk factor for diabetes,” they write.
“Considering the coexistence of the diabetes epidemic and the widespread influence of light pollution at night, the positive associations indicate an urgent need for countries and governments to develop effective prevention and intervention policies and to protect people from the adverse health effects of light pollution at night,” the study authors stress.
Gareth Nye, PhD, senior lecturer at the University of Chester, England, agreed that prior research has found an association between metabolic conditions, such as diabetes, and artificial light at night, with most theories as to the cause focusing on the body’s natural circadian cycle.
He said that internal clocks regulate a variety of bodily processes, such as metabolism and hormone synthesis. They also affect sleep patterns by interfering with synthesis of the hormone melatonin, which is essential for sound sleep, Dr. Nye told the UK Science Media Centre.
However, he stressed that much more research is needed before any link can be considered definitive.
Outdoor night light exposure linked to fasting glucose, A1c
The Chinese researchers set out to approximate the relationships between diabetes prevalence and glucose homeostasis with chronic exposure to outdoor light at night.
They assessed 98,658 participants from the China Noncommunicable Disease Surveillance Study across 162 sites. The mean age of participants was 42.7 years. Female participants comprised 49.2% of the study cohort.
Diabetes was defined based on American Diabetes Association criteria. Satellite data were used to determine exposure to outdoor light at night in 2010. The associations between light exposure at night and indicators of glucose homeostasis were investigated.
Prevalence ratios were calculated and adjusted for sex, age, smoking status, education, body mass index, physical activity, household income, family history of diabetes, rural/urban areas, drinking status, and use of lipid-lowering prescription drugs (primarily statins) or antihypertensives.
The findings showed exposure levels to outdoor light at night were positively linked with 2-hour and fasting glucose concentrations, A1c, and insulin resistance (measured using homeostatic model assessment [HOMA]), but negatively related to β-cell function (measured using HOMA).
More research needed
“We advise caution against causal interpretation of the findings and call for further studies involving direct measurement of individual exposure to light at night,” the researchers conclude.
Dr. Nye agreed.
“One issue with this study is that the areas with the highest outdoor artificial light levels are likely to be those in urban areas and bigger cities. It has been known for a long time now that living in an urbanized area increases your risk of obesity through increased access to high-fat and convenience food, less physical activity levels due to transport links, and less social activities. The authors also state this and the fact participants tended to be older,” he noted.
Large datasets are used in this investigation, however, which generally increases the reliability of the data, he observed.
But it is also “unclear as to whether the population here was selected for this study or was retrospectively analyzed, which poses reliability issues, as does the selection of the representative sample, as it is not discussed,” he noted.
Ultimately, there is no confirmed evidence of the link, and until further work is done to directly link light exposure and diabetes in humans, “the link will remain an association only,” he concluded.
A version of this article first appeared on Medscape.com.
Higher levels of exposure to outdoor artificial light at night are significantly linked with markers of diabetes and impaired glucose homeostasis, in a new national, cross-sectional study from China.
The results showed a 7% significant increase in diabetes prevalence per quintile exposure to artificial light at night (prevalence ratio, 1.07), report Ruizhi Zheng, PhD, of the Shanghai (China) Jiaotong University School of Medicine, and colleagues. People living in areas with the most exposure to light at night had a 28% higher prevalence of diabetes than those living in places with the lowest exposure (PR, 1.28), the researchers found.
The study was published online in Diabetologia.
Previous animal studies have shown that exposure to light at night may interfere with circadian rhythms and affect glucose homeostasis, the study team note. Other research has demonstrated that chronic exposure to moderate indoor light during sleep elevated the prevalence of diabetes in older adults, compared with those sleeping in a dim setting, the authors add.
“Our findings contribute to the growing literature suggesting that artificial light at night is detrimental to health and demonstrate that artificial light at night may be a potential novel risk factor for diabetes,” they write.
“Considering the coexistence of the diabetes epidemic and the widespread influence of light pollution at night, the positive associations indicate an urgent need for countries and governments to develop effective prevention and intervention policies and to protect people from the adverse health effects of light pollution at night,” the study authors stress.
Gareth Nye, PhD, senior lecturer at the University of Chester, England, agreed that prior research has found an association between metabolic conditions, such as diabetes, and artificial light at night, with most theories as to the cause focusing on the body’s natural circadian cycle.
He said that internal clocks regulate a variety of bodily processes, such as metabolism and hormone synthesis. They also affect sleep patterns by interfering with synthesis of the hormone melatonin, which is essential for sound sleep, Dr. Nye told the UK Science Media Centre.
However, he stressed that much more research is needed before any link can be considered definitive.
Outdoor night light exposure linked to fasting glucose, A1c
The Chinese researchers set out to approximate the relationships between diabetes prevalence and glucose homeostasis with chronic exposure to outdoor light at night.
They assessed 98,658 participants from the China Noncommunicable Disease Surveillance Study across 162 sites. The mean age of participants was 42.7 years. Female participants comprised 49.2% of the study cohort.
Diabetes was defined based on American Diabetes Association criteria. Satellite data were used to determine exposure to outdoor light at night in 2010. The associations between light exposure at night and indicators of glucose homeostasis were investigated.
Prevalence ratios were calculated and adjusted for sex, age, smoking status, education, body mass index, physical activity, household income, family history of diabetes, rural/urban areas, drinking status, and use of lipid-lowering prescription drugs (primarily statins) or antihypertensives.
The findings showed exposure levels to outdoor light at night were positively linked with 2-hour and fasting glucose concentrations, A1c, and insulin resistance (measured using homeostatic model assessment [HOMA]), but negatively related to β-cell function (measured using HOMA).
More research needed
“We advise caution against causal interpretation of the findings and call for further studies involving direct measurement of individual exposure to light at night,” the researchers conclude.
Dr. Nye agreed.
“One issue with this study is that the areas with the highest outdoor artificial light levels are likely to be those in urban areas and bigger cities. It has been known for a long time now that living in an urbanized area increases your risk of obesity through increased access to high-fat and convenience food, less physical activity levels due to transport links, and less social activities. The authors also state this and the fact participants tended to be older,” he noted.
Large datasets are used in this investigation, however, which generally increases the reliability of the data, he observed.
But it is also “unclear as to whether the population here was selected for this study or was retrospectively analyzed, which poses reliability issues, as does the selection of the representative sample, as it is not discussed,” he noted.
Ultimately, there is no confirmed evidence of the link, and until further work is done to directly link light exposure and diabetes in humans, “the link will remain an association only,” he concluded.
A version of this article first appeared on Medscape.com.