User login
Is there hope in the fight against aging?
For many years, it has been believed that the aging process is inevitable and that age-related diseases cannot be prevented or reversed. For example, the U.S. Food and Drug Administration does not recognize aging as an indication for drug approval because there are no markers to determine whether possible treatments have a significant impact on the hallmarks of aging.
The field of geroscience aims to find ways to change this by delaying the onset of age-related diseases or by extending the life span. Those mechanisms contribute to the vulnerability of older adults. The presentations focused on identifying biomarkers of aging and on the search for interventions to prevent and treat age-related diseases.
Perspectives from this meeting were published in a report.
An abridged glossary
- Senescent cells: These are old cells with irreversibly damaged DNA; they strongly resist apoptosis. Thus, they are not eliminated and continue to secrete pathogenic proinflammatory molecules.
- Senolytics: This is a class of compounds that promote the removal of senescent cells from the body.
- Autophagy: This is a process that promotes protein degradation, which is attenuated with aging and that impedes the aggregation of proteins harmful to cell function, particularly those of the central nervous system.
- Proteostasis: This is the dynamic regulation of protein homeostasis.
- Epigenetics: This is the field of biology that studies phenotype changes that are not caused by changes in DNA sequencing and that continue to affect cellular division.
- Metabolome: This refers to small molecules that make up the building blocks of all organismal features, from cell membranes to metabolic cycles to genes and proteins.
- Translational research: This involves applying primary research results to clinical research and vice versa.
Possible research topics
Senescence not only occurs with age but also drives aging. At the meeting, evidence was provided that senescent cells may exacerbate the clinical course of older adults in cases of infections (for example, COVID-19) as they lead to cytokine storms.
Experiments on old mice that have undergone genetic modification of senescent cells or the administration of “senolytic cocktails” composed of dasatinib plus quercetin protected the animals from the effects of viral infections. This finding corroborates the idea that factors involved in biological aging increase vulnerability and could be modified through treatment.
Alzheimer’s disease is an example of the effects of cellular senescence. Senescent cells develop a senescence-associated secretory phenotype that can be toxic to neighboring healthy cells and can allow senescence to propagate within tissues. This effect makes Alzheimer’s disease an essential focal point when studying the use of senolytics. In addition, agents that stimulate autophagy may be of interest for treating degenerative diseases.
Assessing therapeutic effects
It may be possible to assess the therapeutic effects of drug candidates using the following biomarkers.
- Growth hormone and type 1 insulin-like growth factor (IGF-1): Older adults are often prescribed growth hormone. However, recent data suggest that doing so is not advantageous to this patient population, because it antagonizes proteostasis and other cell maintenance mechanisms in older age. Experimental studies and studies conducted on centenarians suggest that low growth hormone and IGF-1 levels contribute to longevity and may be therapeutic biomarkers.
- Epigenetics: DNA methylation is a method that offers an “epigenetic clock” to compare biological age with chronologic age. Higher epigenetic age was associated with increased mortality risk, breast cancer, and nonalcoholic fatty liver disease. Therefore, it could also be a therapeutic biomarker.
- Metabolomics: Studying metabolomes facilitates the identification of the link between genetic polymorphisms and longevity, as most polymorphisms explain less than 0.5% of longevity variations.
- New translational strategy: It is common practice to treat each age-related disease individually. An alternative strategy would be to target the hallmarks of biological aging to prevent these diseases from developing. The rate of biological aging correlates with the speed of damage accumulation at the macromolecular, organelle, and cellular levels. It also affects the capacity of the body to repair this damage. The assessment of biomarkers would make possibile research into the effects of short- and long-term treatments that minimize damage and enhance resilience related to diseases common with aging.
New translational research
The report highlights two translational research models: the in-depth study of centenarians and the analysis of how immune aging makes older adults vulnerable to COVID-19. The impact of impaired immunity on aging became particularly evident during the pandemic. However, to home in on immunity as a therapeutic target and to better understand immune resilience, the specific nature of immune and biological deficits still need to be defined.
Metformin is among the therapeutic agents under investigation in cutting-edge clinical research. Its effect on aging will be studied in the Targeting Aging with Metformin (TAME) clinical trial. This trial is the first to study aging outcomes. The goal is to create a regulatory framework that future therapies can follow to achieve FDA approval.
There are three promising therapeutic platforms among the cutting-edge research studies. The first aims to produce adenosine triphosphate, levels of which decline dramatically with aging. The second aims to promote autophagy to remove cellular waste to treat neurodegenerative diseases. The third reprograms the epigenome to a younger state.
Research on mitochondrial dysfunction is relevant because it is highly involved in age-related diseases. Mitochondrial-derived peptides could potentially serve as biomarkers of mitochondrial function in aging studies and become promising therapeutic targets in age-related diseases. One of these peptides, humanin, has been demonstrated to exert protective effects on the heart, brain, and liver. Researchers observed that mitochondrial proteins are age-dependent and are suppressed by growth hormone and IGF-1. They also found that humanin levels are correlated with endothelial function. Data from animal studies have shown that sustained humanin levels are positively linked to longevity; these findings are mirrored in data from centenarians and their offspring, who have higher levels of humanin.
The formation of a Translational Geroscience Network composed of several scientists from various institutions should accelerate the application of this understanding. Despite the ongoing investigational and clinical studies, senolytics should not be regarded as extending life span or treating certain conditions, because their full safety profiles have not yet been elucidated.
Conclusion
Geroscience faces challenges in dealing with age-related problems. It is hoped that these challenges will be overcome through investigational and clinical studies on the mechanisms involved in aging. In-depth study of the interactions of underlying mechanisms of aging are needed to answer the following questions:
- Is there a hierarchical relationship among these mechanisms?
- Are there organ or cell-type differences in the interactions among these mechanisms?
- Is it possible to achieve a synergistic effect through combined interventions targeting several of the processes that drive aging?
It is complicated, but researchers are starting to see the light at the end of the tunnel.
This article was translated from the Medscape Portuguese edition and a version appeared on Medscape.com.
For many years, it has been believed that the aging process is inevitable and that age-related diseases cannot be prevented or reversed. For example, the U.S. Food and Drug Administration does not recognize aging as an indication for drug approval because there are no markers to determine whether possible treatments have a significant impact on the hallmarks of aging.
The field of geroscience aims to find ways to change this by delaying the onset of age-related diseases or by extending the life span. Those mechanisms contribute to the vulnerability of older adults. The presentations focused on identifying biomarkers of aging and on the search for interventions to prevent and treat age-related diseases.
Perspectives from this meeting were published in a report.
An abridged glossary
- Senescent cells: These are old cells with irreversibly damaged DNA; they strongly resist apoptosis. Thus, they are not eliminated and continue to secrete pathogenic proinflammatory molecules.
- Senolytics: This is a class of compounds that promote the removal of senescent cells from the body.
- Autophagy: This is a process that promotes protein degradation, which is attenuated with aging and that impedes the aggregation of proteins harmful to cell function, particularly those of the central nervous system.
- Proteostasis: This is the dynamic regulation of protein homeostasis.
- Epigenetics: This is the field of biology that studies phenotype changes that are not caused by changes in DNA sequencing and that continue to affect cellular division.
- Metabolome: This refers to small molecules that make up the building blocks of all organismal features, from cell membranes to metabolic cycles to genes and proteins.
- Translational research: This involves applying primary research results to clinical research and vice versa.
Possible research topics
Senescence not only occurs with age but also drives aging. At the meeting, evidence was provided that senescent cells may exacerbate the clinical course of older adults in cases of infections (for example, COVID-19) as they lead to cytokine storms.
Experiments on old mice that have undergone genetic modification of senescent cells or the administration of “senolytic cocktails” composed of dasatinib plus quercetin protected the animals from the effects of viral infections. This finding corroborates the idea that factors involved in biological aging increase vulnerability and could be modified through treatment.
Alzheimer’s disease is an example of the effects of cellular senescence. Senescent cells develop a senescence-associated secretory phenotype that can be toxic to neighboring healthy cells and can allow senescence to propagate within tissues. This effect makes Alzheimer’s disease an essential focal point when studying the use of senolytics. In addition, agents that stimulate autophagy may be of interest for treating degenerative diseases.
Assessing therapeutic effects
It may be possible to assess the therapeutic effects of drug candidates using the following biomarkers.
- Growth hormone and type 1 insulin-like growth factor (IGF-1): Older adults are often prescribed growth hormone. However, recent data suggest that doing so is not advantageous to this patient population, because it antagonizes proteostasis and other cell maintenance mechanisms in older age. Experimental studies and studies conducted on centenarians suggest that low growth hormone and IGF-1 levels contribute to longevity and may be therapeutic biomarkers.
- Epigenetics: DNA methylation is a method that offers an “epigenetic clock” to compare biological age with chronologic age. Higher epigenetic age was associated with increased mortality risk, breast cancer, and nonalcoholic fatty liver disease. Therefore, it could also be a therapeutic biomarker.
- Metabolomics: Studying metabolomes facilitates the identification of the link between genetic polymorphisms and longevity, as most polymorphisms explain less than 0.5% of longevity variations.
- New translational strategy: It is common practice to treat each age-related disease individually. An alternative strategy would be to target the hallmarks of biological aging to prevent these diseases from developing. The rate of biological aging correlates with the speed of damage accumulation at the macromolecular, organelle, and cellular levels. It also affects the capacity of the body to repair this damage. The assessment of biomarkers would make possibile research into the effects of short- and long-term treatments that minimize damage and enhance resilience related to diseases common with aging.
New translational research
The report highlights two translational research models: the in-depth study of centenarians and the analysis of how immune aging makes older adults vulnerable to COVID-19. The impact of impaired immunity on aging became particularly evident during the pandemic. However, to home in on immunity as a therapeutic target and to better understand immune resilience, the specific nature of immune and biological deficits still need to be defined.
Metformin is among the therapeutic agents under investigation in cutting-edge clinical research. Its effect on aging will be studied in the Targeting Aging with Metformin (TAME) clinical trial. This trial is the first to study aging outcomes. The goal is to create a regulatory framework that future therapies can follow to achieve FDA approval.
There are three promising therapeutic platforms among the cutting-edge research studies. The first aims to produce adenosine triphosphate, levels of which decline dramatically with aging. The second aims to promote autophagy to remove cellular waste to treat neurodegenerative diseases. The third reprograms the epigenome to a younger state.
Research on mitochondrial dysfunction is relevant because it is highly involved in age-related diseases. Mitochondrial-derived peptides could potentially serve as biomarkers of mitochondrial function in aging studies and become promising therapeutic targets in age-related diseases. One of these peptides, humanin, has been demonstrated to exert protective effects on the heart, brain, and liver. Researchers observed that mitochondrial proteins are age-dependent and are suppressed by growth hormone and IGF-1. They also found that humanin levels are correlated with endothelial function. Data from animal studies have shown that sustained humanin levels are positively linked to longevity; these findings are mirrored in data from centenarians and their offspring, who have higher levels of humanin.
The formation of a Translational Geroscience Network composed of several scientists from various institutions should accelerate the application of this understanding. Despite the ongoing investigational and clinical studies, senolytics should not be regarded as extending life span or treating certain conditions, because their full safety profiles have not yet been elucidated.
Conclusion
Geroscience faces challenges in dealing with age-related problems. It is hoped that these challenges will be overcome through investigational and clinical studies on the mechanisms involved in aging. In-depth study of the interactions of underlying mechanisms of aging are needed to answer the following questions:
- Is there a hierarchical relationship among these mechanisms?
- Are there organ or cell-type differences in the interactions among these mechanisms?
- Is it possible to achieve a synergistic effect through combined interventions targeting several of the processes that drive aging?
It is complicated, but researchers are starting to see the light at the end of the tunnel.
This article was translated from the Medscape Portuguese edition and a version appeared on Medscape.com.
For many years, it has been believed that the aging process is inevitable and that age-related diseases cannot be prevented or reversed. For example, the U.S. Food and Drug Administration does not recognize aging as an indication for drug approval because there are no markers to determine whether possible treatments have a significant impact on the hallmarks of aging.
The field of geroscience aims to find ways to change this by delaying the onset of age-related diseases or by extending the life span. Those mechanisms contribute to the vulnerability of older adults. The presentations focused on identifying biomarkers of aging and on the search for interventions to prevent and treat age-related diseases.
Perspectives from this meeting were published in a report.
An abridged glossary
- Senescent cells: These are old cells with irreversibly damaged DNA; they strongly resist apoptosis. Thus, they are not eliminated and continue to secrete pathogenic proinflammatory molecules.
- Senolytics: This is a class of compounds that promote the removal of senescent cells from the body.
- Autophagy: This is a process that promotes protein degradation, which is attenuated with aging and that impedes the aggregation of proteins harmful to cell function, particularly those of the central nervous system.
- Proteostasis: This is the dynamic regulation of protein homeostasis.
- Epigenetics: This is the field of biology that studies phenotype changes that are not caused by changes in DNA sequencing and that continue to affect cellular division.
- Metabolome: This refers to small molecules that make up the building blocks of all organismal features, from cell membranes to metabolic cycles to genes and proteins.
- Translational research: This involves applying primary research results to clinical research and vice versa.
Possible research topics
Senescence not only occurs with age but also drives aging. At the meeting, evidence was provided that senescent cells may exacerbate the clinical course of older adults in cases of infections (for example, COVID-19) as they lead to cytokine storms.
Experiments on old mice that have undergone genetic modification of senescent cells or the administration of “senolytic cocktails” composed of dasatinib plus quercetin protected the animals from the effects of viral infections. This finding corroborates the idea that factors involved in biological aging increase vulnerability and could be modified through treatment.
Alzheimer’s disease is an example of the effects of cellular senescence. Senescent cells develop a senescence-associated secretory phenotype that can be toxic to neighboring healthy cells and can allow senescence to propagate within tissues. This effect makes Alzheimer’s disease an essential focal point when studying the use of senolytics. In addition, agents that stimulate autophagy may be of interest for treating degenerative diseases.
Assessing therapeutic effects
It may be possible to assess the therapeutic effects of drug candidates using the following biomarkers.
- Growth hormone and type 1 insulin-like growth factor (IGF-1): Older adults are often prescribed growth hormone. However, recent data suggest that doing so is not advantageous to this patient population, because it antagonizes proteostasis and other cell maintenance mechanisms in older age. Experimental studies and studies conducted on centenarians suggest that low growth hormone and IGF-1 levels contribute to longevity and may be therapeutic biomarkers.
- Epigenetics: DNA methylation is a method that offers an “epigenetic clock” to compare biological age with chronologic age. Higher epigenetic age was associated with increased mortality risk, breast cancer, and nonalcoholic fatty liver disease. Therefore, it could also be a therapeutic biomarker.
- Metabolomics: Studying metabolomes facilitates the identification of the link between genetic polymorphisms and longevity, as most polymorphisms explain less than 0.5% of longevity variations.
- New translational strategy: It is common practice to treat each age-related disease individually. An alternative strategy would be to target the hallmarks of biological aging to prevent these diseases from developing. The rate of biological aging correlates with the speed of damage accumulation at the macromolecular, organelle, and cellular levels. It also affects the capacity of the body to repair this damage. The assessment of biomarkers would make possibile research into the effects of short- and long-term treatments that minimize damage and enhance resilience related to diseases common with aging.
New translational research
The report highlights two translational research models: the in-depth study of centenarians and the analysis of how immune aging makes older adults vulnerable to COVID-19. The impact of impaired immunity on aging became particularly evident during the pandemic. However, to home in on immunity as a therapeutic target and to better understand immune resilience, the specific nature of immune and biological deficits still need to be defined.
Metformin is among the therapeutic agents under investigation in cutting-edge clinical research. Its effect on aging will be studied in the Targeting Aging with Metformin (TAME) clinical trial. This trial is the first to study aging outcomes. The goal is to create a regulatory framework that future therapies can follow to achieve FDA approval.
There are three promising therapeutic platforms among the cutting-edge research studies. The first aims to produce adenosine triphosphate, levels of which decline dramatically with aging. The second aims to promote autophagy to remove cellular waste to treat neurodegenerative diseases. The third reprograms the epigenome to a younger state.
Research on mitochondrial dysfunction is relevant because it is highly involved in age-related diseases. Mitochondrial-derived peptides could potentially serve as biomarkers of mitochondrial function in aging studies and become promising therapeutic targets in age-related diseases. One of these peptides, humanin, has been demonstrated to exert protective effects on the heart, brain, and liver. Researchers observed that mitochondrial proteins are age-dependent and are suppressed by growth hormone and IGF-1. They also found that humanin levels are correlated with endothelial function. Data from animal studies have shown that sustained humanin levels are positively linked to longevity; these findings are mirrored in data from centenarians and their offspring, who have higher levels of humanin.
The formation of a Translational Geroscience Network composed of several scientists from various institutions should accelerate the application of this understanding. Despite the ongoing investigational and clinical studies, senolytics should not be regarded as extending life span or treating certain conditions, because their full safety profiles have not yet been elucidated.
Conclusion
Geroscience faces challenges in dealing with age-related problems. It is hoped that these challenges will be overcome through investigational and clinical studies on the mechanisms involved in aging. In-depth study of the interactions of underlying mechanisms of aging are needed to answer the following questions:
- Is there a hierarchical relationship among these mechanisms?
- Are there organ or cell-type differences in the interactions among these mechanisms?
- Is it possible to achieve a synergistic effect through combined interventions targeting several of the processes that drive aging?
It is complicated, but researchers are starting to see the light at the end of the tunnel.
This article was translated from the Medscape Portuguese edition and a version appeared on Medscape.com.
Guidance updated for congenital hypothyroidism screening, management
Congenital hypothyroidism is one of the most common preventable causes of intellectual disabilities worldwide, but newborn screening has not been established in all countries.
Additionally, screening alone is not enough to prevent adverse outcomes in children, write authors of a technical report published online in Pediatrics (Jan. 2023;151[1]:e2022060420).
Susan R. Rose, MD, with the division of endocrinology at Cincinnati Children’s Hospital Medical Center in Ohio, led the work group that updated guidance for screening and management of congenital hypothyroidism. The group worked in conjunction with the American Academy of Pediatrics Section on Endocrinology, the AAP Council on Genetics, the Pediatric Endocrine Society, and the American Thyroid Association.
In addition to screening, timely diagnosis, effective treatment, and follow-up are important.
Tests don’t always tell the full story with congenital hypothyroidism.
“Physicians need to consider hypothyroidism in the face of clinical symptoms, even if newborn screening thyroid test results are normal,” the authors write.
They add that newborn screening for congenital hypothyroidism followed by prompt levothyroxine therapy can prevent severe intellectual disability, psychomotor dysfunction, and impaired growth.
Incidence of congenital hypothyroidism ranges from approximately 1 in 2,000 to 1 in 4,000 newborn infants in countries that have newborn screening data, according to the report.
Following are highlights of the guidance:
Clinical signs
Symptoms and signs include large posterior fontanelle, lethargy, large tongue, prolonged jaundice, umbilical hernia, constipation, and/or hypothermia. With these signs, measuring serum thyroid-stimulating hormone (TSH) and free thyroxine (FT4) is indicated, regardless of screening results.
Newborn screening in first days
Population screening is cost effective when performed by state or other public health laboratories working with hospitals or birthing centers in their area, the authors write.
Multidisciplinary teams are best able to conduct comprehensive care when cases are detected.
The screening includes a dried blood spot from a heel stick on an approved paper card using appropriate collection methods. The blood spots are then sent to the laboratory. The preferred age for collecting the specimen is 48-72 hours of age.
That timing may be difficult, the authors note, as 90% of infants in the United States and Europe are discharged before 48 hours, but taking the specimen before discharge is important to avoid missing the early diagnosis.
“However, collection of the NBS [newborn screening] specimen before 48 hours of age, and particularly before 24 hours of age, necessitates the use of age-specific TSH reference ranges or repeat screening, particularly to avoid false-positive results,” the authors note.
If a newborn infant is transferred to another hospital, communication about the screening is critical.
Testing strategies
Three test strategies are used for screening: a primary TSH – reflex T4 measurement; primary T4 – reflex TSH measurement; and combined T4 and TSH measurement.
“All three test strategies detect moderate to severe primary congenital hypothyroidism with similar accuracy,” the authors write.
Most newborn screening programs in the United States and worldwide use a primary TSH test strategy.
Multiple births, same-sex twins
The incidence of congenital hypothyroidism appears to be higher with multiple births (1:876 in twin births and 1:575 in higher-order multiple births in one study). Another study showed the incidence of congenital hypothyroidism in same-sex twins to be 1 in 593, compared with 1 in 3,060 in different-sex twins.
“Most twin pairs (> 95%) are discordant for congenital hypothyroidism,” the authors write. “However, in monozygotic twins who share placental circulation, blood from a euthyroid fetal twin with normal thyroid hormone levels may cross to a fetal twin with congenital hypothyroidism, temporarily correcting the hypothyroidism and preventing its detection by newborn screening at 24-72 hours of life. Thus, all monozygotic twins, or same-sex twins for whom zygosity is unknown, should undergo repeat newborn screening around 2 weeks of age.”
Down syndrome
Congenital hypothyroidism incidence in infants with trisomy 21 (Down syndrome) is high and ranges from 1% to 12% in various reports. The infants tend to have lower T4 concentrations and higher TSH concentrations than do infants without trisomy. Down syndrome is associated with other comorbidities, including congenital heart disease, “that may further increase the risk of abnormal newborn screening results because of acute illness or excess iodine exposure,” the authors write.
Even infants with Down syndrome who don’t have congenital hypothyroidism are still at significant risk of developing primary hypothyroidism in their first year (approximately 7% in one prospective study).
“Therefore, in these infants, a second newborn screening should be performed at 2-4 weeks of life and serum TSH should be measured at 6 and 12 months of life,” the authors say.
Communication with primary care provider
Direct communication between the newborn screening program and the primary care physician is important for appropriate follow-up. Consulting a pediatric endocrinologist can speed diagnosis and management.
Serum confirmation after abnormal screening
The next step if any child’s screening results suggest congenital hypothyroidism is to perform a physical exam (for goiter, lingual thyroid gland, and/or physical signs of hypothyroidism) and to measure the concentrations of TSH and FT4 (or total T4) in the blood.
For confirmation of abnormal screening results, the authors say, measurement of FT4 is preferred over measuring total T4.
Interpreting serum confirmation
Some interpretations are clear cut: “Elevated TSH with low FT4 on the confirmatory serum testing indicates overt primary hypothyroidism,” the authors write.
But there are various other outcomes with more controversy.
Elevated TSH and normal FT4, for instance, is known as hyperthyrotropinemia or subclinical hypothyroidism and represents a mild primary thyroid abnormality.
In this scenario, there is controversy regarding the need for L-T4 therapy because there are few and conflicting studies regarding how mild congenital hypothyroidism affects cognitive development.
“[E]xpert opinion suggests that persistent TSH elevation > 10 mIU/L is an indication to initiate L-T4 treatment,” the authors write.
Normal TSH and low T4 is seen in patients with central hypothyroidism, prematurity, low birth weight, acute illness, or thyroxine-binding globulin deficiency.
“The concept that central hypothyroidism is usually mild appears unfounded: A study from the Netherlands found that mean pretreatment serum FT4 levels in central congenital hypothyroidism were similar to those of patients with moderately severe primary congenital hypothyroidism. Therefore, L-T4 treatment of central congenital hypothyroidism is indicated.”
Imaging
Routine thyroid imaging is controversial for patients with congenital hypothyroidism. In most cases, it won’t alter clinical management before age 3 years.
Thyroid ultrasonography can find thyroid tissue without radiation exposure and can be performed at any time after a congenital hypothyroidism diagnosis.
“Ultrasonography has lower sensitivity than scintigraphy for detecting ectopic thyroid tissue, the most common cause of congenital hypothyroidism, although its sensitivity is improved by the use of color Doppler,” the authors write.
Infants with normal thyroid imaging at birth may have transient hypothyroidism. In these patients, reevaluation of thyroid hormone therapy after 3 years of age to assess for persistent hypothyroidism may be beneficial.
Treatment
Congenital hypothyroidism is treated with enteral L-T4 at a starting dose of 10-15 mcg/kg per day, given once a day.
L-T4 tablets are the treatment of choice and generic tablets are fine for most children, the authors write, adding that a brand name formulation may be more consistent and better for children with severe congenital hypothyroidism.
An oral solution of L-T4 has been approved by the U.S. Food and Drug Administration for use in children.
“[H]owever, limited experience with its use showed that dosing may not be equivalent to dosing with tablet formulations,” the guidance states.
The goal of initial L-T4 therapy is to normalize serum FT4 and TSH levels as quickly as possible. The outlook is poorer for infants whose hypothyroidism is detected later in life, who receive inadequate doses of L-T4, or who have more severe forms.
Age-specific TSH reference ranges vary by laboratory, but recent studies indicate the top limit of normal TSH in infants in the first 3 months of life is 4.1-4.8 mIU/L.
“[T]herefore, TSH values above 5 mIU/L generally are abnormal if observed after 3 months of age. Whether overtreatment (defined by elevated serum FT4) is harmful remains unclear and evidence is conflicting,” the authors write.
Monitoring
In the near-term follow-up, close laboratory monitoring is necessary during L-T4 treatment to maintain blood TSH and FT4 in the target ranges. Studies support measuring those levels every 1-2 months in the first 6 months of life for children with congenital hypothyroidism, every 2-3 months in the second 6 months, and then every 3-4 months between 1 and 3 years of age.
In long-term follow-up, attention to behavioral and cognitive development is important, because children with congenital hypothyroidism may be at higher risk for neurocognitive and socioemotional dysfunction compared with their peers, even with adequate treatment of congenital hypothyroidism. Hearing deficits are reported in about 10% of children with congenital hypothyroidism.
Developmental outcomes
When L-T4 therapy is maintained and TSH and FT4 are within target range, growth and adult height are generally normal in children with congenital hypothyroidism.
In contrast, the neurodevelopmental prognosis is less certain when treatment starts late.
“[I]nfants with severe congenital hypothyroidism and intrauterine hypothyroidism (as indicated by retarded skeletal maturation at birth) may have low-to-normal intelligence,” the report states. “Similarly, although more than 80% of infants given L-T4 replacement therapy before 3 months of age have an intelligence [quotient] greater than 85, 77% of these infants show signs of cognitive impairment in arithmetic ability, speech, or fine motor coordination later in life.”
If a child is properly treated for congenital hypothyroidism but growth or development is abnormal, testing for other illness, hearing deficit, or other hormone deficiency is needed, the report states.
The authors report no relevant financial relationships.
Congenital hypothyroidism is one of the most common preventable causes of intellectual disabilities worldwide, but newborn screening has not been established in all countries.
Additionally, screening alone is not enough to prevent adverse outcomes in children, write authors of a technical report published online in Pediatrics (Jan. 2023;151[1]:e2022060420).
Susan R. Rose, MD, with the division of endocrinology at Cincinnati Children’s Hospital Medical Center in Ohio, led the work group that updated guidance for screening and management of congenital hypothyroidism. The group worked in conjunction with the American Academy of Pediatrics Section on Endocrinology, the AAP Council on Genetics, the Pediatric Endocrine Society, and the American Thyroid Association.
In addition to screening, timely diagnosis, effective treatment, and follow-up are important.
Tests don’t always tell the full story with congenital hypothyroidism.
“Physicians need to consider hypothyroidism in the face of clinical symptoms, even if newborn screening thyroid test results are normal,” the authors write.
They add that newborn screening for congenital hypothyroidism followed by prompt levothyroxine therapy can prevent severe intellectual disability, psychomotor dysfunction, and impaired growth.
Incidence of congenital hypothyroidism ranges from approximately 1 in 2,000 to 1 in 4,000 newborn infants in countries that have newborn screening data, according to the report.
Following are highlights of the guidance:
Clinical signs
Symptoms and signs include large posterior fontanelle, lethargy, large tongue, prolonged jaundice, umbilical hernia, constipation, and/or hypothermia. With these signs, measuring serum thyroid-stimulating hormone (TSH) and free thyroxine (FT4) is indicated, regardless of screening results.
Newborn screening in first days
Population screening is cost effective when performed by state or other public health laboratories working with hospitals or birthing centers in their area, the authors write.
Multidisciplinary teams are best able to conduct comprehensive care when cases are detected.
The screening includes a dried blood spot from a heel stick on an approved paper card using appropriate collection methods. The blood spots are then sent to the laboratory. The preferred age for collecting the specimen is 48-72 hours of age.
That timing may be difficult, the authors note, as 90% of infants in the United States and Europe are discharged before 48 hours, but taking the specimen before discharge is important to avoid missing the early diagnosis.
“However, collection of the NBS [newborn screening] specimen before 48 hours of age, and particularly before 24 hours of age, necessitates the use of age-specific TSH reference ranges or repeat screening, particularly to avoid false-positive results,” the authors note.
If a newborn infant is transferred to another hospital, communication about the screening is critical.
Testing strategies
Three test strategies are used for screening: a primary TSH – reflex T4 measurement; primary T4 – reflex TSH measurement; and combined T4 and TSH measurement.
“All three test strategies detect moderate to severe primary congenital hypothyroidism with similar accuracy,” the authors write.
Most newborn screening programs in the United States and worldwide use a primary TSH test strategy.
Multiple births, same-sex twins
The incidence of congenital hypothyroidism appears to be higher with multiple births (1:876 in twin births and 1:575 in higher-order multiple births in one study). Another study showed the incidence of congenital hypothyroidism in same-sex twins to be 1 in 593, compared with 1 in 3,060 in different-sex twins.
“Most twin pairs (> 95%) are discordant for congenital hypothyroidism,” the authors write. “However, in monozygotic twins who share placental circulation, blood from a euthyroid fetal twin with normal thyroid hormone levels may cross to a fetal twin with congenital hypothyroidism, temporarily correcting the hypothyroidism and preventing its detection by newborn screening at 24-72 hours of life. Thus, all monozygotic twins, or same-sex twins for whom zygosity is unknown, should undergo repeat newborn screening around 2 weeks of age.”
Down syndrome
Congenital hypothyroidism incidence in infants with trisomy 21 (Down syndrome) is high and ranges from 1% to 12% in various reports. The infants tend to have lower T4 concentrations and higher TSH concentrations than do infants without trisomy. Down syndrome is associated with other comorbidities, including congenital heart disease, “that may further increase the risk of abnormal newborn screening results because of acute illness or excess iodine exposure,” the authors write.
Even infants with Down syndrome who don’t have congenital hypothyroidism are still at significant risk of developing primary hypothyroidism in their first year (approximately 7% in one prospective study).
“Therefore, in these infants, a second newborn screening should be performed at 2-4 weeks of life and serum TSH should be measured at 6 and 12 months of life,” the authors say.
Communication with primary care provider
Direct communication between the newborn screening program and the primary care physician is important for appropriate follow-up. Consulting a pediatric endocrinologist can speed diagnosis and management.
Serum confirmation after abnormal screening
The next step if any child’s screening results suggest congenital hypothyroidism is to perform a physical exam (for goiter, lingual thyroid gland, and/or physical signs of hypothyroidism) and to measure the concentrations of TSH and FT4 (or total T4) in the blood.
For confirmation of abnormal screening results, the authors say, measurement of FT4 is preferred over measuring total T4.
Interpreting serum confirmation
Some interpretations are clear cut: “Elevated TSH with low FT4 on the confirmatory serum testing indicates overt primary hypothyroidism,” the authors write.
But there are various other outcomes with more controversy.
Elevated TSH and normal FT4, for instance, is known as hyperthyrotropinemia or subclinical hypothyroidism and represents a mild primary thyroid abnormality.
In this scenario, there is controversy regarding the need for L-T4 therapy because there are few and conflicting studies regarding how mild congenital hypothyroidism affects cognitive development.
“[E]xpert opinion suggests that persistent TSH elevation > 10 mIU/L is an indication to initiate L-T4 treatment,” the authors write.
Normal TSH and low T4 is seen in patients with central hypothyroidism, prematurity, low birth weight, acute illness, or thyroxine-binding globulin deficiency.
“The concept that central hypothyroidism is usually mild appears unfounded: A study from the Netherlands found that mean pretreatment serum FT4 levels in central congenital hypothyroidism were similar to those of patients with moderately severe primary congenital hypothyroidism. Therefore, L-T4 treatment of central congenital hypothyroidism is indicated.”
Imaging
Routine thyroid imaging is controversial for patients with congenital hypothyroidism. In most cases, it won’t alter clinical management before age 3 years.
Thyroid ultrasonography can find thyroid tissue without radiation exposure and can be performed at any time after a congenital hypothyroidism diagnosis.
“Ultrasonography has lower sensitivity than scintigraphy for detecting ectopic thyroid tissue, the most common cause of congenital hypothyroidism, although its sensitivity is improved by the use of color Doppler,” the authors write.
Infants with normal thyroid imaging at birth may have transient hypothyroidism. In these patients, reevaluation of thyroid hormone therapy after 3 years of age to assess for persistent hypothyroidism may be beneficial.
Treatment
Congenital hypothyroidism is treated with enteral L-T4 at a starting dose of 10-15 mcg/kg per day, given once a day.
L-T4 tablets are the treatment of choice and generic tablets are fine for most children, the authors write, adding that a brand name formulation may be more consistent and better for children with severe congenital hypothyroidism.
An oral solution of L-T4 has been approved by the U.S. Food and Drug Administration for use in children.
“[H]owever, limited experience with its use showed that dosing may not be equivalent to dosing with tablet formulations,” the guidance states.
The goal of initial L-T4 therapy is to normalize serum FT4 and TSH levels as quickly as possible. The outlook is poorer for infants whose hypothyroidism is detected later in life, who receive inadequate doses of L-T4, or who have more severe forms.
Age-specific TSH reference ranges vary by laboratory, but recent studies indicate the top limit of normal TSH in infants in the first 3 months of life is 4.1-4.8 mIU/L.
“[T]herefore, TSH values above 5 mIU/L generally are abnormal if observed after 3 months of age. Whether overtreatment (defined by elevated serum FT4) is harmful remains unclear and evidence is conflicting,” the authors write.
Monitoring
In the near-term follow-up, close laboratory monitoring is necessary during L-T4 treatment to maintain blood TSH and FT4 in the target ranges. Studies support measuring those levels every 1-2 months in the first 6 months of life for children with congenital hypothyroidism, every 2-3 months in the second 6 months, and then every 3-4 months between 1 and 3 years of age.
In long-term follow-up, attention to behavioral and cognitive development is important, because children with congenital hypothyroidism may be at higher risk for neurocognitive and socioemotional dysfunction compared with their peers, even with adequate treatment of congenital hypothyroidism. Hearing deficits are reported in about 10% of children with congenital hypothyroidism.
Developmental outcomes
When L-T4 therapy is maintained and TSH and FT4 are within target range, growth and adult height are generally normal in children with congenital hypothyroidism.
In contrast, the neurodevelopmental prognosis is less certain when treatment starts late.
“[I]nfants with severe congenital hypothyroidism and intrauterine hypothyroidism (as indicated by retarded skeletal maturation at birth) may have low-to-normal intelligence,” the report states. “Similarly, although more than 80% of infants given L-T4 replacement therapy before 3 months of age have an intelligence [quotient] greater than 85, 77% of these infants show signs of cognitive impairment in arithmetic ability, speech, or fine motor coordination later in life.”
If a child is properly treated for congenital hypothyroidism but growth or development is abnormal, testing for other illness, hearing deficit, or other hormone deficiency is needed, the report states.
The authors report no relevant financial relationships.
Congenital hypothyroidism is one of the most common preventable causes of intellectual disabilities worldwide, but newborn screening has not been established in all countries.
Additionally, screening alone is not enough to prevent adverse outcomes in children, write authors of a technical report published online in Pediatrics (Jan. 2023;151[1]:e2022060420).
Susan R. Rose, MD, with the division of endocrinology at Cincinnati Children’s Hospital Medical Center in Ohio, led the work group that updated guidance for screening and management of congenital hypothyroidism. The group worked in conjunction with the American Academy of Pediatrics Section on Endocrinology, the AAP Council on Genetics, the Pediatric Endocrine Society, and the American Thyroid Association.
In addition to screening, timely diagnosis, effective treatment, and follow-up are important.
Tests don’t always tell the full story with congenital hypothyroidism.
“Physicians need to consider hypothyroidism in the face of clinical symptoms, even if newborn screening thyroid test results are normal,” the authors write.
They add that newborn screening for congenital hypothyroidism followed by prompt levothyroxine therapy can prevent severe intellectual disability, psychomotor dysfunction, and impaired growth.
Incidence of congenital hypothyroidism ranges from approximately 1 in 2,000 to 1 in 4,000 newborn infants in countries that have newborn screening data, according to the report.
Following are highlights of the guidance:
Clinical signs
Symptoms and signs include large posterior fontanelle, lethargy, large tongue, prolonged jaundice, umbilical hernia, constipation, and/or hypothermia. With these signs, measuring serum thyroid-stimulating hormone (TSH) and free thyroxine (FT4) is indicated, regardless of screening results.
Newborn screening in first days
Population screening is cost effective when performed by state or other public health laboratories working with hospitals or birthing centers in their area, the authors write.
Multidisciplinary teams are best able to conduct comprehensive care when cases are detected.
The screening includes a dried blood spot from a heel stick on an approved paper card using appropriate collection methods. The blood spots are then sent to the laboratory. The preferred age for collecting the specimen is 48-72 hours of age.
That timing may be difficult, the authors note, as 90% of infants in the United States and Europe are discharged before 48 hours, but taking the specimen before discharge is important to avoid missing the early diagnosis.
“However, collection of the NBS [newborn screening] specimen before 48 hours of age, and particularly before 24 hours of age, necessitates the use of age-specific TSH reference ranges or repeat screening, particularly to avoid false-positive results,” the authors note.
If a newborn infant is transferred to another hospital, communication about the screening is critical.
Testing strategies
Three test strategies are used for screening: a primary TSH – reflex T4 measurement; primary T4 – reflex TSH measurement; and combined T4 and TSH measurement.
“All three test strategies detect moderate to severe primary congenital hypothyroidism with similar accuracy,” the authors write.
Most newborn screening programs in the United States and worldwide use a primary TSH test strategy.
Multiple births, same-sex twins
The incidence of congenital hypothyroidism appears to be higher with multiple births (1:876 in twin births and 1:575 in higher-order multiple births in one study). Another study showed the incidence of congenital hypothyroidism in same-sex twins to be 1 in 593, compared with 1 in 3,060 in different-sex twins.
“Most twin pairs (> 95%) are discordant for congenital hypothyroidism,” the authors write. “However, in monozygotic twins who share placental circulation, blood from a euthyroid fetal twin with normal thyroid hormone levels may cross to a fetal twin with congenital hypothyroidism, temporarily correcting the hypothyroidism and preventing its detection by newborn screening at 24-72 hours of life. Thus, all monozygotic twins, or same-sex twins for whom zygosity is unknown, should undergo repeat newborn screening around 2 weeks of age.”
Down syndrome
Congenital hypothyroidism incidence in infants with trisomy 21 (Down syndrome) is high and ranges from 1% to 12% in various reports. The infants tend to have lower T4 concentrations and higher TSH concentrations than do infants without trisomy. Down syndrome is associated with other comorbidities, including congenital heart disease, “that may further increase the risk of abnormal newborn screening results because of acute illness or excess iodine exposure,” the authors write.
Even infants with Down syndrome who don’t have congenital hypothyroidism are still at significant risk of developing primary hypothyroidism in their first year (approximately 7% in one prospective study).
“Therefore, in these infants, a second newborn screening should be performed at 2-4 weeks of life and serum TSH should be measured at 6 and 12 months of life,” the authors say.
Communication with primary care provider
Direct communication between the newborn screening program and the primary care physician is important for appropriate follow-up. Consulting a pediatric endocrinologist can speed diagnosis and management.
Serum confirmation after abnormal screening
The next step if any child’s screening results suggest congenital hypothyroidism is to perform a physical exam (for goiter, lingual thyroid gland, and/or physical signs of hypothyroidism) and to measure the concentrations of TSH and FT4 (or total T4) in the blood.
For confirmation of abnormal screening results, the authors say, measurement of FT4 is preferred over measuring total T4.
Interpreting serum confirmation
Some interpretations are clear cut: “Elevated TSH with low FT4 on the confirmatory serum testing indicates overt primary hypothyroidism,” the authors write.
But there are various other outcomes with more controversy.
Elevated TSH and normal FT4, for instance, is known as hyperthyrotropinemia or subclinical hypothyroidism and represents a mild primary thyroid abnormality.
In this scenario, there is controversy regarding the need for L-T4 therapy because there are few and conflicting studies regarding how mild congenital hypothyroidism affects cognitive development.
“[E]xpert opinion suggests that persistent TSH elevation > 10 mIU/L is an indication to initiate L-T4 treatment,” the authors write.
Normal TSH and low T4 is seen in patients with central hypothyroidism, prematurity, low birth weight, acute illness, or thyroxine-binding globulin deficiency.
“The concept that central hypothyroidism is usually mild appears unfounded: A study from the Netherlands found that mean pretreatment serum FT4 levels in central congenital hypothyroidism were similar to those of patients with moderately severe primary congenital hypothyroidism. Therefore, L-T4 treatment of central congenital hypothyroidism is indicated.”
Imaging
Routine thyroid imaging is controversial for patients with congenital hypothyroidism. In most cases, it won’t alter clinical management before age 3 years.
Thyroid ultrasonography can find thyroid tissue without radiation exposure and can be performed at any time after a congenital hypothyroidism diagnosis.
“Ultrasonography has lower sensitivity than scintigraphy for detecting ectopic thyroid tissue, the most common cause of congenital hypothyroidism, although its sensitivity is improved by the use of color Doppler,” the authors write.
Infants with normal thyroid imaging at birth may have transient hypothyroidism. In these patients, reevaluation of thyroid hormone therapy after 3 years of age to assess for persistent hypothyroidism may be beneficial.
Treatment
Congenital hypothyroidism is treated with enteral L-T4 at a starting dose of 10-15 mcg/kg per day, given once a day.
L-T4 tablets are the treatment of choice and generic tablets are fine for most children, the authors write, adding that a brand name formulation may be more consistent and better for children with severe congenital hypothyroidism.
An oral solution of L-T4 has been approved by the U.S. Food and Drug Administration for use in children.
“[H]owever, limited experience with its use showed that dosing may not be equivalent to dosing with tablet formulations,” the guidance states.
The goal of initial L-T4 therapy is to normalize serum FT4 and TSH levels as quickly as possible. The outlook is poorer for infants whose hypothyroidism is detected later in life, who receive inadequate doses of L-T4, or who have more severe forms.
Age-specific TSH reference ranges vary by laboratory, but recent studies indicate the top limit of normal TSH in infants in the first 3 months of life is 4.1-4.8 mIU/L.
“[T]herefore, TSH values above 5 mIU/L generally are abnormal if observed after 3 months of age. Whether overtreatment (defined by elevated serum FT4) is harmful remains unclear and evidence is conflicting,” the authors write.
Monitoring
In the near-term follow-up, close laboratory monitoring is necessary during L-T4 treatment to maintain blood TSH and FT4 in the target ranges. Studies support measuring those levels every 1-2 months in the first 6 months of life for children with congenital hypothyroidism, every 2-3 months in the second 6 months, and then every 3-4 months between 1 and 3 years of age.
In long-term follow-up, attention to behavioral and cognitive development is important, because children with congenital hypothyroidism may be at higher risk for neurocognitive and socioemotional dysfunction compared with their peers, even with adequate treatment of congenital hypothyroidism. Hearing deficits are reported in about 10% of children with congenital hypothyroidism.
Developmental outcomes
When L-T4 therapy is maintained and TSH and FT4 are within target range, growth and adult height are generally normal in children with congenital hypothyroidism.
In contrast, the neurodevelopmental prognosis is less certain when treatment starts late.
“[I]nfants with severe congenital hypothyroidism and intrauterine hypothyroidism (as indicated by retarded skeletal maturation at birth) may have low-to-normal intelligence,” the report states. “Similarly, although more than 80% of infants given L-T4 replacement therapy before 3 months of age have an intelligence [quotient] greater than 85, 77% of these infants show signs of cognitive impairment in arithmetic ability, speech, or fine motor coordination later in life.”
If a child is properly treated for congenital hypothyroidism but growth or development is abnormal, testing for other illness, hearing deficit, or other hormone deficiency is needed, the report states.
The authors report no relevant financial relationships.
FROM PEDIATRICS
Intermittent fasting can lead to type 2 diabetes remission
In a small randomized controlled trial of patients with type 2 diabetes in China, close to half of those who followed a novel intermittent fasting program for 3 months had diabetes remission (A1c less than 6.5% without taking antidiabetic drugs) that persisted for 1 year.
Importantly, “this study was performed under real-life conditions, and the intervention was delivered by trained nurses in primary care rather than by specialized staff at a research institute, making it a more practical and achievable way to manage” type 2 diabetes, the authors report.
Moreover, 65% of the patients in the intervention group who achieved diabetes remission had had diabetes for more than 6 years, which “suggests the possibility of remission for patients with longer duration” of diabetes, they note.
In addition, antidiabetic medication costs decreased by 77%, compared with baseline, in patients in the intermittent-fasting intervention group.
Although intermittent fasting has been studied for weight loss, it had not been investigated for effectiveness for diabetes remission.
These findings suggest that intermittent fasting “could be a paradigm shift in the management goals in diabetes care,” Xiao Yang and colleagues conclude in their study, published online in The Journal of Clinical Endocrinology & Metabolism.
“Type 2 diabetes is not necessarily a permanent, lifelong disease,” senior author Dongbo Liu, PhD, from the Hunan Agricultural University, Changsha, China, added in a press release from The Endocrine Society.
“Diabetes remission is possible if patients lose weight by changing their diet and exercise habits,” Dr. Liu said.
‘Excellent outcome’
Invited to comment, Amy E. Rothberg, MD, PhD, who was not involved with the research, agreed that the study indicates that intermittent fasting works for diabetes remission.
“We know that diabetes remission is possible with calorie restriction and subsequent weight loss, and intermittent fasting is just one of the many [dietary] approaches that may be suitable, appealing, and sustainable to some individuals, and usually results in calorie restriction and therefore weight loss,” she said.
The most studied types of intermittent fasting diets are alternate-day fasting, the 5:2 diet, and time-restricted consumption, Dr. Rothberg told this news organization.
This study presented a novel type of intermittent fasting, she noted. The intervention consisted of 6 cycles (3 months) of 5 fasting days followed by 10 ad libitum days, and then 3 months of follow-up (with no fasting days).
After 3 months of the intervention plus 3 months of follow-up, 47% of the 36 patients in the intervention group achieved diabetes remission (with a mean A1c of 5.66%), compared with only 2.8% of the 36 patients in the control group.
At 12 months, 44% of patients in the intervention group had sustained diabetes remission (with a mean A1c of 6.33%).
This was “an excellent outcome,” said Dr. Rothberg, professor of nutritional sciences, School of Public Health, University of Michigan, Ann Arbor, and a co-author of an international consensus statement that defined diabetes remission.
On average, patients in the intermittent fasting group lost 5.93 kg (13.0 lb) in 3 months, which was sustained over 12 months. “The large amount of weight reduction is key to continuing to achieve diabetes remission,” she noted.
This contrasted with an average weight loss of just 0.27 kg (0.6 lb) in the control group.
Participants who were prescribed fewer antidiabetic medications were more likely to achieve diabetes remission. The researchers acknowledge that the study was not blinded, and they did not record physical activity (although participants were encouraged to maintain their usual physical activity).
This was a small study, Dr. Rothberg acknowledged. The researchers did not specify which specific antidiabetic drugs patients were taking, and they did not determine waist or hip circumference or assess lipids.
The diet was culturally sensitive, appropriate, and feasible in this Chinese population and would not be generalizable to non-Asians.
Nevertheless, a similar approach could be used in any population if the diet is tailored to the individual, according to Dr. Rothberg. Importantly, patients would need to receive guidance from a dietician to make sure their diet comprises all the necessary micronutrients, vitamins, and minerals on fasting days, and they would need to maintain a relatively balanced diet and not gorge themselves on feast days.
“I think we should campaign widely about lifestyle approaches to achieve diabetes remission,” she urged.
72 patients with diabetes for an average of 6.6 years
“Despite a widespread public consensus that [type 2 diabetes] is irreversible and requires drug treatment escalation, there is some evidence of the possibility of remission,” Dr. Yang and colleagues write in their article.
They aimed to evaluate the effectiveness of intermittent fasting for diabetes remission and the durability of diabetes remission at 1 year.
Diabetes remission was defined having a stable A1c less than 6.5% for at least 3 months after discontinuing all antidiabetic medications, confirmed in at least annual A1c measurements (according to a 2021 consensus statement initiated by the American Diabetes Association).
Between 2019 and 2020, the researchers enrolled 72 participants aged 38-72 years who had had type 2 diabetes (duration 1 to 11 years) and a body mass index (BMI) of 19.1-30.4 kg/m2. Patients were randomized 1:1 to the intermittent fasting group or control group.
Baseline characteristics were similar in both groups. Patients were a mean age of 53 years and roughly 60% were men. They had a mean BMI of 24 kg/m2, a mean duration of diabetes of 6.6 years, and a mean A1c of 7.6%, and they were taking an average of 1.8 glucose-lowering medications.
On fasting days, patients in the intervention group received a Chinese Medical Nutrition Therapy kit that provided approximately 840 kcal/day (46% carbohydrates, 46% fat, 8% protein). The kit included a breakfast of a fruit and vegetable gruel, lunch of a solid beverage plus a nutritional rice composite, and dinner of a solid beverage and a meal replacement biscuit, which participants reconstituted by mixing with boiling water. They were allowed to consume noncaloric beverages.
On nonfasting days, patients chose foods ad libitum based on the 2017 Dietary Guidelines for Diabetes in China, which recommend approximately 50%-65% of total energy intake from carbohydrates, 15%-20% from protein, and 20%-30% from fat, and had greater than or equal to 5 g fiber per serving.
Patients in the control group chose foods ad libitum from the dietary guidelines during the entire study.
The study received funding from the National Natural Science Foundation of China. The authors have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
In a small randomized controlled trial of patients with type 2 diabetes in China, close to half of those who followed a novel intermittent fasting program for 3 months had diabetes remission (A1c less than 6.5% without taking antidiabetic drugs) that persisted for 1 year.
Importantly, “this study was performed under real-life conditions, and the intervention was delivered by trained nurses in primary care rather than by specialized staff at a research institute, making it a more practical and achievable way to manage” type 2 diabetes, the authors report.
Moreover, 65% of the patients in the intervention group who achieved diabetes remission had had diabetes for more than 6 years, which “suggests the possibility of remission for patients with longer duration” of diabetes, they note.
In addition, antidiabetic medication costs decreased by 77%, compared with baseline, in patients in the intermittent-fasting intervention group.
Although intermittent fasting has been studied for weight loss, it had not been investigated for effectiveness for diabetes remission.
These findings suggest that intermittent fasting “could be a paradigm shift in the management goals in diabetes care,” Xiao Yang and colleagues conclude in their study, published online in The Journal of Clinical Endocrinology & Metabolism.
“Type 2 diabetes is not necessarily a permanent, lifelong disease,” senior author Dongbo Liu, PhD, from the Hunan Agricultural University, Changsha, China, added in a press release from The Endocrine Society.
“Diabetes remission is possible if patients lose weight by changing their diet and exercise habits,” Dr. Liu said.
‘Excellent outcome’
Invited to comment, Amy E. Rothberg, MD, PhD, who was not involved with the research, agreed that the study indicates that intermittent fasting works for diabetes remission.
“We know that diabetes remission is possible with calorie restriction and subsequent weight loss, and intermittent fasting is just one of the many [dietary] approaches that may be suitable, appealing, and sustainable to some individuals, and usually results in calorie restriction and therefore weight loss,” she said.
The most studied types of intermittent fasting diets are alternate-day fasting, the 5:2 diet, and time-restricted consumption, Dr. Rothberg told this news organization.
This study presented a novel type of intermittent fasting, she noted. The intervention consisted of 6 cycles (3 months) of 5 fasting days followed by 10 ad libitum days, and then 3 months of follow-up (with no fasting days).
After 3 months of the intervention plus 3 months of follow-up, 47% of the 36 patients in the intervention group achieved diabetes remission (with a mean A1c of 5.66%), compared with only 2.8% of the 36 patients in the control group.
At 12 months, 44% of patients in the intervention group had sustained diabetes remission (with a mean A1c of 6.33%).
This was “an excellent outcome,” said Dr. Rothberg, professor of nutritional sciences, School of Public Health, University of Michigan, Ann Arbor, and a co-author of an international consensus statement that defined diabetes remission.
On average, patients in the intermittent fasting group lost 5.93 kg (13.0 lb) in 3 months, which was sustained over 12 months. “The large amount of weight reduction is key to continuing to achieve diabetes remission,” she noted.
This contrasted with an average weight loss of just 0.27 kg (0.6 lb) in the control group.
Participants who were prescribed fewer antidiabetic medications were more likely to achieve diabetes remission. The researchers acknowledge that the study was not blinded, and they did not record physical activity (although participants were encouraged to maintain their usual physical activity).
This was a small study, Dr. Rothberg acknowledged. The researchers did not specify which specific antidiabetic drugs patients were taking, and they did not determine waist or hip circumference or assess lipids.
The diet was culturally sensitive, appropriate, and feasible in this Chinese population and would not be generalizable to non-Asians.
Nevertheless, a similar approach could be used in any population if the diet is tailored to the individual, according to Dr. Rothberg. Importantly, patients would need to receive guidance from a dietician to make sure their diet comprises all the necessary micronutrients, vitamins, and minerals on fasting days, and they would need to maintain a relatively balanced diet and not gorge themselves on feast days.
“I think we should campaign widely about lifestyle approaches to achieve diabetes remission,” she urged.
72 patients with diabetes for an average of 6.6 years
“Despite a widespread public consensus that [type 2 diabetes] is irreversible and requires drug treatment escalation, there is some evidence of the possibility of remission,” Dr. Yang and colleagues write in their article.
They aimed to evaluate the effectiveness of intermittent fasting for diabetes remission and the durability of diabetes remission at 1 year.
Diabetes remission was defined having a stable A1c less than 6.5% for at least 3 months after discontinuing all antidiabetic medications, confirmed in at least annual A1c measurements (according to a 2021 consensus statement initiated by the American Diabetes Association).
Between 2019 and 2020, the researchers enrolled 72 participants aged 38-72 years who had had type 2 diabetes (duration 1 to 11 years) and a body mass index (BMI) of 19.1-30.4 kg/m2. Patients were randomized 1:1 to the intermittent fasting group or control group.
Baseline characteristics were similar in both groups. Patients were a mean age of 53 years and roughly 60% were men. They had a mean BMI of 24 kg/m2, a mean duration of diabetes of 6.6 years, and a mean A1c of 7.6%, and they were taking an average of 1.8 glucose-lowering medications.
On fasting days, patients in the intervention group received a Chinese Medical Nutrition Therapy kit that provided approximately 840 kcal/day (46% carbohydrates, 46% fat, 8% protein). The kit included a breakfast of a fruit and vegetable gruel, lunch of a solid beverage plus a nutritional rice composite, and dinner of a solid beverage and a meal replacement biscuit, which participants reconstituted by mixing with boiling water. They were allowed to consume noncaloric beverages.
On nonfasting days, patients chose foods ad libitum based on the 2017 Dietary Guidelines for Diabetes in China, which recommend approximately 50%-65% of total energy intake from carbohydrates, 15%-20% from protein, and 20%-30% from fat, and had greater than or equal to 5 g fiber per serving.
Patients in the control group chose foods ad libitum from the dietary guidelines during the entire study.
The study received funding from the National Natural Science Foundation of China. The authors have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
In a small randomized controlled trial of patients with type 2 diabetes in China, close to half of those who followed a novel intermittent fasting program for 3 months had diabetes remission (A1c less than 6.5% without taking antidiabetic drugs) that persisted for 1 year.
Importantly, “this study was performed under real-life conditions, and the intervention was delivered by trained nurses in primary care rather than by specialized staff at a research institute, making it a more practical and achievable way to manage” type 2 diabetes, the authors report.
Moreover, 65% of the patients in the intervention group who achieved diabetes remission had had diabetes for more than 6 years, which “suggests the possibility of remission for patients with longer duration” of diabetes, they note.
In addition, antidiabetic medication costs decreased by 77%, compared with baseline, in patients in the intermittent-fasting intervention group.
Although intermittent fasting has been studied for weight loss, it had not been investigated for effectiveness for diabetes remission.
These findings suggest that intermittent fasting “could be a paradigm shift in the management goals in diabetes care,” Xiao Yang and colleagues conclude in their study, published online in The Journal of Clinical Endocrinology & Metabolism.
“Type 2 diabetes is not necessarily a permanent, lifelong disease,” senior author Dongbo Liu, PhD, from the Hunan Agricultural University, Changsha, China, added in a press release from The Endocrine Society.
“Diabetes remission is possible if patients lose weight by changing their diet and exercise habits,” Dr. Liu said.
‘Excellent outcome’
Invited to comment, Amy E. Rothberg, MD, PhD, who was not involved with the research, agreed that the study indicates that intermittent fasting works for diabetes remission.
“We know that diabetes remission is possible with calorie restriction and subsequent weight loss, and intermittent fasting is just one of the many [dietary] approaches that may be suitable, appealing, and sustainable to some individuals, and usually results in calorie restriction and therefore weight loss,” she said.
The most studied types of intermittent fasting diets are alternate-day fasting, the 5:2 diet, and time-restricted consumption, Dr. Rothberg told this news organization.
This study presented a novel type of intermittent fasting, she noted. The intervention consisted of 6 cycles (3 months) of 5 fasting days followed by 10 ad libitum days, and then 3 months of follow-up (with no fasting days).
After 3 months of the intervention plus 3 months of follow-up, 47% of the 36 patients in the intervention group achieved diabetes remission (with a mean A1c of 5.66%), compared with only 2.8% of the 36 patients in the control group.
At 12 months, 44% of patients in the intervention group had sustained diabetes remission (with a mean A1c of 6.33%).
This was “an excellent outcome,” said Dr. Rothberg, professor of nutritional sciences, School of Public Health, University of Michigan, Ann Arbor, and a co-author of an international consensus statement that defined diabetes remission.
On average, patients in the intermittent fasting group lost 5.93 kg (13.0 lb) in 3 months, which was sustained over 12 months. “The large amount of weight reduction is key to continuing to achieve diabetes remission,” she noted.
This contrasted with an average weight loss of just 0.27 kg (0.6 lb) in the control group.
Participants who were prescribed fewer antidiabetic medications were more likely to achieve diabetes remission. The researchers acknowledge that the study was not blinded, and they did not record physical activity (although participants were encouraged to maintain their usual physical activity).
This was a small study, Dr. Rothberg acknowledged. The researchers did not specify which specific antidiabetic drugs patients were taking, and they did not determine waist or hip circumference or assess lipids.
The diet was culturally sensitive, appropriate, and feasible in this Chinese population and would not be generalizable to non-Asians.
Nevertheless, a similar approach could be used in any population if the diet is tailored to the individual, according to Dr. Rothberg. Importantly, patients would need to receive guidance from a dietician to make sure their diet comprises all the necessary micronutrients, vitamins, and minerals on fasting days, and they would need to maintain a relatively balanced diet and not gorge themselves on feast days.
“I think we should campaign widely about lifestyle approaches to achieve diabetes remission,” she urged.
72 patients with diabetes for an average of 6.6 years
“Despite a widespread public consensus that [type 2 diabetes] is irreversible and requires drug treatment escalation, there is some evidence of the possibility of remission,” Dr. Yang and colleagues write in their article.
They aimed to evaluate the effectiveness of intermittent fasting for diabetes remission and the durability of diabetes remission at 1 year.
Diabetes remission was defined having a stable A1c less than 6.5% for at least 3 months after discontinuing all antidiabetic medications, confirmed in at least annual A1c measurements (according to a 2021 consensus statement initiated by the American Diabetes Association).
Between 2019 and 2020, the researchers enrolled 72 participants aged 38-72 years who had had type 2 diabetes (duration 1 to 11 years) and a body mass index (BMI) of 19.1-30.4 kg/m2. Patients were randomized 1:1 to the intermittent fasting group or control group.
Baseline characteristics were similar in both groups. Patients were a mean age of 53 years and roughly 60% were men. They had a mean BMI of 24 kg/m2, a mean duration of diabetes of 6.6 years, and a mean A1c of 7.6%, and they were taking an average of 1.8 glucose-lowering medications.
On fasting days, patients in the intervention group received a Chinese Medical Nutrition Therapy kit that provided approximately 840 kcal/day (46% carbohydrates, 46% fat, 8% protein). The kit included a breakfast of a fruit and vegetable gruel, lunch of a solid beverage plus a nutritional rice composite, and dinner of a solid beverage and a meal replacement biscuit, which participants reconstituted by mixing with boiling water. They were allowed to consume noncaloric beverages.
On nonfasting days, patients chose foods ad libitum based on the 2017 Dietary Guidelines for Diabetes in China, which recommend approximately 50%-65% of total energy intake from carbohydrates, 15%-20% from protein, and 20%-30% from fat, and had greater than or equal to 5 g fiber per serving.
Patients in the control group chose foods ad libitum from the dietary guidelines during the entire study.
The study received funding from the National Natural Science Foundation of China. The authors have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
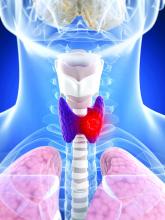
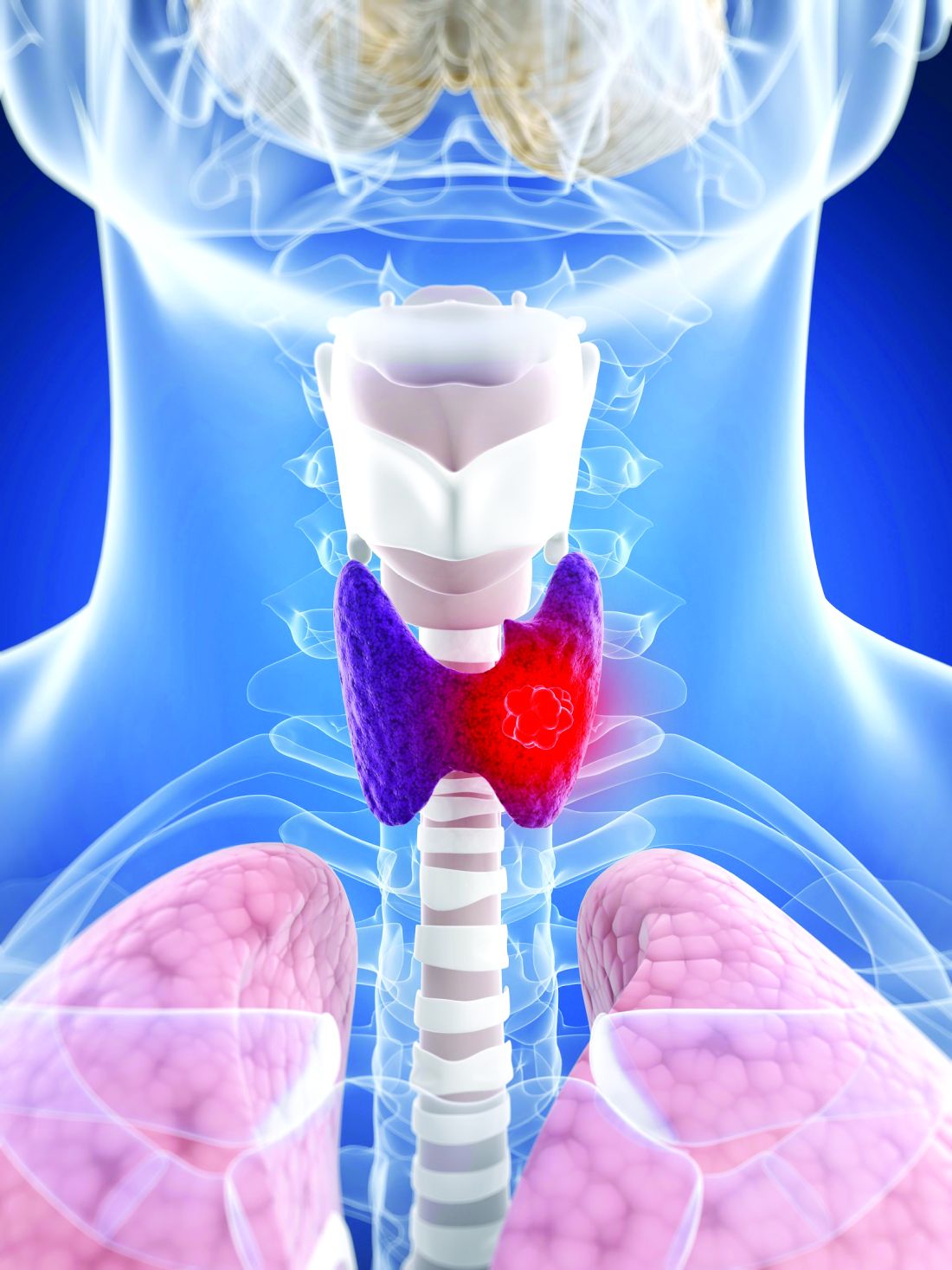
Lifestyle choices could curb genetic risk for thyroid cancer
A healthier lifestyle mitigated the impact of genetic factors on the risk of thyroid cancer, in a study based on data from more than 260,000 individuals.
Thyroid cancer has increased globally in recent years and ranks 9th among 36 cancers worldwide, at a considerable cost to health care systems, wrote Xiuming Feng of Guangxi Medical University, Nanning, Guangxi, China, and colleagues.
Both genetic and lifestyle factors are related to thyroid cancer; previous research suggests a heritability of about 50%, but data on the impact of modifiable lifestyle factors on thyroid cancer are limited, the researchers said.
In a prospective cohort study published in JAMA Network Open, the researchers used data from the UK Biobank and recruited adults aged 40-69 years during March 2006–October 2010. The final study population included 264,956 individuals of European descent. The median age of the participants was 57 years, and 52% were women.
Data on lifestyle behaviors were collected using interviews and questionnaires. The researchers constructed a total lifestyle score based on five variables: diet, physical activity, weight, smoking, and alcohol consumption. Each variable was assigned a score of 0 or 1, with 1 being favorable lifestyle behavior. Lifestyle was divided into three categories: unfavorable (scores 0-1), intermediate (score 2), and favorable (scores 3-5).
Each individual’s polygenic risk score (PRS) was categorized as low, intermediate, or high based on a meta–genome-wide association study of three cohorts.
The main outcome was the development of thyroid cancer.
The researchers identified 423 incident thyroid cancer cases over a median follow-up of 11.1 years.
Overall, higher PRSs were significantly associated with thyroid cancer (hazard ratio, 2.25; 95% confidence interval [CI], 1.91-2.64; P < .00001) as was an unfavorable lifestyle score (HR, 1.93; 95% CI, 1.50-2.49; P < .001 for trend).
An unfavorable lifestyle was significantly associated with thyroid cancer in the highest PRS group, and individuals with high PRS and unfavorable lifestyle had a nearly fivefold increased risk of thyroid cancer (HR, 4.89; 95% CI, 3.03-7.91; P < .001). By extension, “Adherence to a healthier lifestyle could decrease the incidence of thyroid cancer in individuals with a higher PRS,” the researchers wrote in their discussion.
The findings were limited by several factors, including the availability of only baseline lifestyle data, and lack of data on iodine intake, radiation exposure, experience, and family history, the researchers noted. Other limitations include the potential lack of generalizability to populations other than the individuals of European descent in the current study, they said.
However, the study is the first known to address the association among lifestyle, genetic factors, and risk of thyroid cancer, and was strengthened by the large study population, and the results suggest that lifestyle interventions may help reduce the risk of thyroid cancer in those with a genetic predisposition, they concluded.
Healthy living can make a difference
The incidence of thyroid cancer has increased annually, and exploring the possible risk factors could prevent the occurrence of thyroid cancer, corresponding author Xiaobo Yang, PhD, said in an interview.
Previous studies have reported that thyroid cancer is related to genetics and lifestyle, said Dr. Yang. “However, whether healthy lifestyle was associated with thyroid cancer risk and could attenuate the impact of genetic variants on thyroid cancer remains equivocal; therefore, it is crucial to determine the associations between genetic and lifestyle with thyroid cancer,” he said.
“To our surprise, we found that adherence to healthier lifestyle also could reduce the risk of thyroid cancer in those with high genetic predispositions,” said Dr. Yang. “The findings highlight the potential role of lifestyle interventions on thyroid cancer, especially in those with high genetic risk, because the heritability of thyroid cancer was very high, approximately 50%,” he said. “More attention should be paid to the role of healthier lifestyle in the prevention of cancer,” he added.
“Adherence to a healthier lifestyle could decrease the risk of thyroid cancer, which is the important message for clinicians,” said Dr. Yang. “It is not too soon to comment on implications for clinical practice, because many studies have maintained the consistent comment that healthier lifestyle could prevent the occurrence of cancer,” he said.
The relationship between sex-specific lifestyle factors such as smoking and alcohol use and thyroid cancer remains uncertain, and more research is needed to validate these associations, Dr. Yang said. More research also is needed to confirm the complex mechanism between lifestyle and genetics in thyroid cancer, he added.
The study was supported by the National Key R&D Program of China and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
A healthier lifestyle mitigated the impact of genetic factors on the risk of thyroid cancer, in a study based on data from more than 260,000 individuals.
Thyroid cancer has increased globally in recent years and ranks 9th among 36 cancers worldwide, at a considerable cost to health care systems, wrote Xiuming Feng of Guangxi Medical University, Nanning, Guangxi, China, and colleagues.
Both genetic and lifestyle factors are related to thyroid cancer; previous research suggests a heritability of about 50%, but data on the impact of modifiable lifestyle factors on thyroid cancer are limited, the researchers said.
In a prospective cohort study published in JAMA Network Open, the researchers used data from the UK Biobank and recruited adults aged 40-69 years during March 2006–October 2010. The final study population included 264,956 individuals of European descent. The median age of the participants was 57 years, and 52% were women.
Data on lifestyle behaviors were collected using interviews and questionnaires. The researchers constructed a total lifestyle score based on five variables: diet, physical activity, weight, smoking, and alcohol consumption. Each variable was assigned a score of 0 or 1, with 1 being favorable lifestyle behavior. Lifestyle was divided into three categories: unfavorable (scores 0-1), intermediate (score 2), and favorable (scores 3-5).
Each individual’s polygenic risk score (PRS) was categorized as low, intermediate, or high based on a meta–genome-wide association study of three cohorts.
The main outcome was the development of thyroid cancer.
The researchers identified 423 incident thyroid cancer cases over a median follow-up of 11.1 years.
Overall, higher PRSs were significantly associated with thyroid cancer (hazard ratio, 2.25; 95% confidence interval [CI], 1.91-2.64; P < .00001) as was an unfavorable lifestyle score (HR, 1.93; 95% CI, 1.50-2.49; P < .001 for trend).
An unfavorable lifestyle was significantly associated with thyroid cancer in the highest PRS group, and individuals with high PRS and unfavorable lifestyle had a nearly fivefold increased risk of thyroid cancer (HR, 4.89; 95% CI, 3.03-7.91; P < .001). By extension, “Adherence to a healthier lifestyle could decrease the incidence of thyroid cancer in individuals with a higher PRS,” the researchers wrote in their discussion.
The findings were limited by several factors, including the availability of only baseline lifestyle data, and lack of data on iodine intake, radiation exposure, experience, and family history, the researchers noted. Other limitations include the potential lack of generalizability to populations other than the individuals of European descent in the current study, they said.
However, the study is the first known to address the association among lifestyle, genetic factors, and risk of thyroid cancer, and was strengthened by the large study population, and the results suggest that lifestyle interventions may help reduce the risk of thyroid cancer in those with a genetic predisposition, they concluded.
Healthy living can make a difference
The incidence of thyroid cancer has increased annually, and exploring the possible risk factors could prevent the occurrence of thyroid cancer, corresponding author Xiaobo Yang, PhD, said in an interview.
Previous studies have reported that thyroid cancer is related to genetics and lifestyle, said Dr. Yang. “However, whether healthy lifestyle was associated with thyroid cancer risk and could attenuate the impact of genetic variants on thyroid cancer remains equivocal; therefore, it is crucial to determine the associations between genetic and lifestyle with thyroid cancer,” he said.
“To our surprise, we found that adherence to healthier lifestyle also could reduce the risk of thyroid cancer in those with high genetic predispositions,” said Dr. Yang. “The findings highlight the potential role of lifestyle interventions on thyroid cancer, especially in those with high genetic risk, because the heritability of thyroid cancer was very high, approximately 50%,” he said. “More attention should be paid to the role of healthier lifestyle in the prevention of cancer,” he added.
“Adherence to a healthier lifestyle could decrease the risk of thyroid cancer, which is the important message for clinicians,” said Dr. Yang. “It is not too soon to comment on implications for clinical practice, because many studies have maintained the consistent comment that healthier lifestyle could prevent the occurrence of cancer,” he said.
The relationship between sex-specific lifestyle factors such as smoking and alcohol use and thyroid cancer remains uncertain, and more research is needed to validate these associations, Dr. Yang said. More research also is needed to confirm the complex mechanism between lifestyle and genetics in thyroid cancer, he added.
The study was supported by the National Key R&D Program of China and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
A healthier lifestyle mitigated the impact of genetic factors on the risk of thyroid cancer, in a study based on data from more than 260,000 individuals.
Thyroid cancer has increased globally in recent years and ranks 9th among 36 cancers worldwide, at a considerable cost to health care systems, wrote Xiuming Feng of Guangxi Medical University, Nanning, Guangxi, China, and colleagues.
Both genetic and lifestyle factors are related to thyroid cancer; previous research suggests a heritability of about 50%, but data on the impact of modifiable lifestyle factors on thyroid cancer are limited, the researchers said.
In a prospective cohort study published in JAMA Network Open, the researchers used data from the UK Biobank and recruited adults aged 40-69 years during March 2006–October 2010. The final study population included 264,956 individuals of European descent. The median age of the participants was 57 years, and 52% were women.
Data on lifestyle behaviors were collected using interviews and questionnaires. The researchers constructed a total lifestyle score based on five variables: diet, physical activity, weight, smoking, and alcohol consumption. Each variable was assigned a score of 0 or 1, with 1 being favorable lifestyle behavior. Lifestyle was divided into three categories: unfavorable (scores 0-1), intermediate (score 2), and favorable (scores 3-5).
Each individual’s polygenic risk score (PRS) was categorized as low, intermediate, or high based on a meta–genome-wide association study of three cohorts.
The main outcome was the development of thyroid cancer.
The researchers identified 423 incident thyroid cancer cases over a median follow-up of 11.1 years.
Overall, higher PRSs were significantly associated with thyroid cancer (hazard ratio, 2.25; 95% confidence interval [CI], 1.91-2.64; P < .00001) as was an unfavorable lifestyle score (HR, 1.93; 95% CI, 1.50-2.49; P < .001 for trend).
An unfavorable lifestyle was significantly associated with thyroid cancer in the highest PRS group, and individuals with high PRS and unfavorable lifestyle had a nearly fivefold increased risk of thyroid cancer (HR, 4.89; 95% CI, 3.03-7.91; P < .001). By extension, “Adherence to a healthier lifestyle could decrease the incidence of thyroid cancer in individuals with a higher PRS,” the researchers wrote in their discussion.
The findings were limited by several factors, including the availability of only baseline lifestyle data, and lack of data on iodine intake, radiation exposure, experience, and family history, the researchers noted. Other limitations include the potential lack of generalizability to populations other than the individuals of European descent in the current study, they said.
However, the study is the first known to address the association among lifestyle, genetic factors, and risk of thyroid cancer, and was strengthened by the large study population, and the results suggest that lifestyle interventions may help reduce the risk of thyroid cancer in those with a genetic predisposition, they concluded.
Healthy living can make a difference
The incidence of thyroid cancer has increased annually, and exploring the possible risk factors could prevent the occurrence of thyroid cancer, corresponding author Xiaobo Yang, PhD, said in an interview.
Previous studies have reported that thyroid cancer is related to genetics and lifestyle, said Dr. Yang. “However, whether healthy lifestyle was associated with thyroid cancer risk and could attenuate the impact of genetic variants on thyroid cancer remains equivocal; therefore, it is crucial to determine the associations between genetic and lifestyle with thyroid cancer,” he said.
“To our surprise, we found that adherence to healthier lifestyle also could reduce the risk of thyroid cancer in those with high genetic predispositions,” said Dr. Yang. “The findings highlight the potential role of lifestyle interventions on thyroid cancer, especially in those with high genetic risk, because the heritability of thyroid cancer was very high, approximately 50%,” he said. “More attention should be paid to the role of healthier lifestyle in the prevention of cancer,” he added.
“Adherence to a healthier lifestyle could decrease the risk of thyroid cancer, which is the important message for clinicians,” said Dr. Yang. “It is not too soon to comment on implications for clinical practice, because many studies have maintained the consistent comment that healthier lifestyle could prevent the occurrence of cancer,” he said.
The relationship between sex-specific lifestyle factors such as smoking and alcohol use and thyroid cancer remains uncertain, and more research is needed to validate these associations, Dr. Yang said. More research also is needed to confirm the complex mechanism between lifestyle and genetics in thyroid cancer, he added.
The study was supported by the National Key R&D Program of China and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
Getting testosterone online is easy. Solid advice, not so much
When a secret shopper used telemedicine to request testosterone supplements, options were plentiful but good advice was scarce. As researchers showed, companies offered testosterone replacement therapy to the prospective buyer even though his stated T level was much higher than the cut-off for low testosterone levels.
Direct-to-consumer (DTC) delivery of testosterone or medications for erectile dysfunction has become routine. The many benefits of wide access include convenience and the ability to discuss sensitive topics in a safe environment. But as the new findings indicate, a lack of adherence to solid medical advice – such as guidance from the Endocrine Society and the American Urological Association – may be putting some people at risk for potentially harmful outcomes.
The Endocrine Society and the AUA state that only people with true testosterone deficiency should receive a T boost. Both groups discourage men who are planning to become parents in the near future from taking supplemental testosterone because of possible harms to their fertility.
“When guidelines are not being followed, there’s always the potential that we might not get the best outcomes for patients,” said Joshua A. Halpern, MD, a urologist at the Northwestern University Feinberg School of Medicine, Chicago, who led the study, which was published in JAMA Internal Medicine.
To conduct the research, the secret shopper approached seven different DTC testosterone websites with the same story for each: He was a 34-year-old man with low energy and libido who hoped to become a father soon. The customer noted that he had a testosterone level of 675 ng/dL, well above the 300 ng/dL the AUA considers low.
Despite these red flags – the normal T levels, the parenthood aspirations – representatives of almost every platform moved the shopper along toward receiving additional testosterone, with no attention to the possible harms to fertility. Only one platform declined to offer the testosterone because the shopper’s T levels were sufficient. In the other cases the secret shopper did not go forward with obtaining the medication, which would have required a prescription.
“Our goal with this study was to achieve a better understanding of what patients are experiencing and what’s out there,” Dr. Halpern said. While this study focused on cisgender patients, Dr. Halpern noted that it’s also important to understand the experiences of transgender patients who seek DTC hormonal therapy.
The research could help keep DTC companies more honest, according to Jesse N. Mills, MD, a urologist and men’s health specialist at the University of California, Los Angeles, who was not involved in the work. DTC platforms are financially incentivized to dispense medications regardless of need, unlike a traditional doctor who generally has no personal financial stake in a prescription.
“We need to keep the heat on DTC platforms,” Dr. Mills said, calling the article a “punchback” against the current DTC model for testosterone products. Dr. Mills said he is not opposed to telemedicine or DTC practices in general, adding that UCLA made a successful pivot to telemedicine during the pandemic.
“You can set up a lot of good care through video visits,” Dr. Mills said, as long as the system is ethical.
Dr. Halpern and Dr. Mills reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When a secret shopper used telemedicine to request testosterone supplements, options were plentiful but good advice was scarce. As researchers showed, companies offered testosterone replacement therapy to the prospective buyer even though his stated T level was much higher than the cut-off for low testosterone levels.
Direct-to-consumer (DTC) delivery of testosterone or medications for erectile dysfunction has become routine. The many benefits of wide access include convenience and the ability to discuss sensitive topics in a safe environment. But as the new findings indicate, a lack of adherence to solid medical advice – such as guidance from the Endocrine Society and the American Urological Association – may be putting some people at risk for potentially harmful outcomes.
The Endocrine Society and the AUA state that only people with true testosterone deficiency should receive a T boost. Both groups discourage men who are planning to become parents in the near future from taking supplemental testosterone because of possible harms to their fertility.
“When guidelines are not being followed, there’s always the potential that we might not get the best outcomes for patients,” said Joshua A. Halpern, MD, a urologist at the Northwestern University Feinberg School of Medicine, Chicago, who led the study, which was published in JAMA Internal Medicine.
To conduct the research, the secret shopper approached seven different DTC testosterone websites with the same story for each: He was a 34-year-old man with low energy and libido who hoped to become a father soon. The customer noted that he had a testosterone level of 675 ng/dL, well above the 300 ng/dL the AUA considers low.
Despite these red flags – the normal T levels, the parenthood aspirations – representatives of almost every platform moved the shopper along toward receiving additional testosterone, with no attention to the possible harms to fertility. Only one platform declined to offer the testosterone because the shopper’s T levels were sufficient. In the other cases the secret shopper did not go forward with obtaining the medication, which would have required a prescription.
“Our goal with this study was to achieve a better understanding of what patients are experiencing and what’s out there,” Dr. Halpern said. While this study focused on cisgender patients, Dr. Halpern noted that it’s also important to understand the experiences of transgender patients who seek DTC hormonal therapy.
The research could help keep DTC companies more honest, according to Jesse N. Mills, MD, a urologist and men’s health specialist at the University of California, Los Angeles, who was not involved in the work. DTC platforms are financially incentivized to dispense medications regardless of need, unlike a traditional doctor who generally has no personal financial stake in a prescription.
“We need to keep the heat on DTC platforms,” Dr. Mills said, calling the article a “punchback” against the current DTC model for testosterone products. Dr. Mills said he is not opposed to telemedicine or DTC practices in general, adding that UCLA made a successful pivot to telemedicine during the pandemic.
“You can set up a lot of good care through video visits,” Dr. Mills said, as long as the system is ethical.
Dr. Halpern and Dr. Mills reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When a secret shopper used telemedicine to request testosterone supplements, options were plentiful but good advice was scarce. As researchers showed, companies offered testosterone replacement therapy to the prospective buyer even though his stated T level was much higher than the cut-off for low testosterone levels.
Direct-to-consumer (DTC) delivery of testosterone or medications for erectile dysfunction has become routine. The many benefits of wide access include convenience and the ability to discuss sensitive topics in a safe environment. But as the new findings indicate, a lack of adherence to solid medical advice – such as guidance from the Endocrine Society and the American Urological Association – may be putting some people at risk for potentially harmful outcomes.
The Endocrine Society and the AUA state that only people with true testosterone deficiency should receive a T boost. Both groups discourage men who are planning to become parents in the near future from taking supplemental testosterone because of possible harms to their fertility.
“When guidelines are not being followed, there’s always the potential that we might not get the best outcomes for patients,” said Joshua A. Halpern, MD, a urologist at the Northwestern University Feinberg School of Medicine, Chicago, who led the study, which was published in JAMA Internal Medicine.
To conduct the research, the secret shopper approached seven different DTC testosterone websites with the same story for each: He was a 34-year-old man with low energy and libido who hoped to become a father soon. The customer noted that he had a testosterone level of 675 ng/dL, well above the 300 ng/dL the AUA considers low.
Despite these red flags – the normal T levels, the parenthood aspirations – representatives of almost every platform moved the shopper along toward receiving additional testosterone, with no attention to the possible harms to fertility. Only one platform declined to offer the testosterone because the shopper’s T levels were sufficient. In the other cases the secret shopper did not go forward with obtaining the medication, which would have required a prescription.
“Our goal with this study was to achieve a better understanding of what patients are experiencing and what’s out there,” Dr. Halpern said. While this study focused on cisgender patients, Dr. Halpern noted that it’s also important to understand the experiences of transgender patients who seek DTC hormonal therapy.
The research could help keep DTC companies more honest, according to Jesse N. Mills, MD, a urologist and men’s health specialist at the University of California, Los Angeles, who was not involved in the work. DTC platforms are financially incentivized to dispense medications regardless of need, unlike a traditional doctor who generally has no personal financial stake in a prescription.
“We need to keep the heat on DTC platforms,” Dr. Mills said, calling the article a “punchback” against the current DTC model for testosterone products. Dr. Mills said he is not opposed to telemedicine or DTC practices in general, adding that UCLA made a successful pivot to telemedicine during the pandemic.
“You can set up a lot of good care through video visits,” Dr. Mills said, as long as the system is ethical.
Dr. Halpern and Dr. Mills reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mind the geriatrician gap
These should be the best of times for geriatric medicine.
The baby boom has become a senior surge, bringing in a rapidly growing pool of aging patients for geriatricians to treat. According to the U.S. Census Bureau, more than 56 million adults aged 65 and older live in the United States. They account for about 17% of the nation’s population. That number is expected to hit 73 million by 2030 and 86 million by 2050.
The American Geriatrics Society estimates that 30% of older people require the attention of geriatricians. These clinicians excel in managing complex cases – patients with multiple comorbidities, such as coronary artery disease, dementia, and osteoporosis, who are taking a half dozen, and often more, medications.
. In the 2010s, geriatricians called for “25,000 [such specialists] by 2025.” As of 2021, 7123 certified geriatricians were practicing in the United States, according to the American Board of Medical Specialties.
The Health Resources and Services Administration, a federal agency that addresses medical workforce shortages, estimates that there will be 6,230 geriatricians by 2025, or approximately 1 for every 3,000 older adults requiring geriatric care. HRSA projects a shortage of 27,000 geriatricians by 2025.
The specialty has faced an uphill battle to attract fellows. This year, only 43% of the nation’s 177 geriatrics fellowship slots were filled, according to November’s National Resident Match Program report. Family medicine–based geriatrics achieved only a 32% fill rate, while internal medicine–based programs saw a rate of 45%.
“Our numbers are shrinking so we need another approach to make sure older adults get the care they need and deserve,” said G. Michael Harper, MD, president of the 6,000-member AGS.
But Dr. Harper, who practices at the University of California, San Francisco, and the San Francisco VA Medical Center, added a positive note: “We may be struggling to increase the number of board-certified geriatricians, but the field itself has made a lot of progress in terms of improving clinical care through advancements in science and in the ways we deliver care.”
Dr. Harper cited the Hospital Elder Life Program, a hospital model developed at the Harvard-affiliated Marcus Institute for Aging Research, which uses an interprofessional team and trained volunteers to prevent delirium and functional decline. HELP has been adopted by more than 200 hospitals worldwide and has been successful at returning older adults to their homes or previous living situations with maintained or improved ability to function, he said.
Mark Supiano, MD, professor and chief of geriatrics at the University of Utah, Salt Lake City, said the specialty has been in shortage mode since ABMS recognized it in 1988. He was in the initial cohort of fellowship-trained geriatricians, sitting for the first certifying exam in geriatrics offered that year.
“Back then, the demographic imperative of the aging of our society was on the horizon. We’re living it now. I knew enough to recognize it was coming and saw an opportunity,” Dr. Supiano said in an interview. “There was so much then that we didn’t know about how to understand aging or how to care for older adults that there really was such a knowledge gap.”
Dr. Supiano is an associate editor of Hazzard’s Geriatric Medicine and Gerontology (McGraw-Hill Education), which has more than doubled in pages and word count during his career.
Unfavorable finances
Katherine Thompson, MD, director of the geriatrics fellowship program at the University of Chicago and codirector of UChicago’s Successful Aging and Frailty Evaluation Clinic, said money is a major reason for the struggle. “I think probably the biggest driver is financial,” she said. “A lot of people are graduating medical school with really astronomical amounts of medical school loans.”
Geriatricians, like other doctors, carry a large debt – $200,000, on average, not counting undergraduate debt, according to the Association of American Medical Colleges.
But the typical geriatrician earns less than an internist or family medicine doctor who doesn’t undergo the additional year of training, Dr. Thompson said. “There’s not a lot of financial motivation to do this fellowship,” she said.
The jobs website Zippia reports that geriatricians earned roughly $165,000 per year on average in 2022. The average annual incomes in 2022 were $191,000 for pediatricians, $215,000 for family physicians, and $223,000 for internists, according to the site.
In other words, Dr. Harper said, “geriatrics is one of the few professions where you can actually do additional training and make less money.”
The reason for the pay issue is simple: Geriatricians treat patients covered by Medicare, whose reimbursement schedules lag behind those of commercial insurers. The Kaiser Family Foundation reported in 2020 that private insurance paid 143% of Medicare rates on average for physician services.
Dr. Harper said overall compensation for geriatricians has “not gained a lot of traction,” but they can earn comfortable livings.
Still, representation of the specialty on the American Medical Association’s Relative Value Scale Update Committee has led to approval by the Centers for Medicare & Medicaid Services of billing codes that pay geriatricians “for what they do. Examples include chronic care management, advance care planning, and dementia evaluation,” he said.
But the geriatrician gap goes beyond money.
Ageism, too, may play a role in residents not choosing geriatrics.
“Our culture is ageist. It definitely focuses on youth and looks at aging as being loss rather than just a change in what works well and what doesn’t work well,” said Mary Tinetti, MD, a geriatrician and researcher at Yale University, New Haven, Conn. “Ageism happens among physicians, just because they’re part of the broader society.”
Time for a new goal?
Dr. Tinetti said she’s optimistic that new ideas about geriatricians teaching other primary care clinicians about the tenets of geriatric medicine, which offer a wholistic approach to comorbidities, such as diabetes, atrial fibrillation, dementia, hypertension, hyperlipidemia, and polypharmacy problems faced by this population, especially those 85 and older.
She has called on her profession to abandon the goal of increasing the numbers of board-certified geriatricians – whom she refers to as big “G” geriatricians. She instead wants to develop a “small, elite workforce” that discovers and tests geriatrics principles through research, teaches these principles to all healthcare professions and to the public, and disseminates and implements the policies.
“We need a cadre of geriatricians who train all other clinicians in the care of older adults,” Dr. Tinetti said. “The goal is not more geriatricians but rather the preparation of all clinicians in the care of older adults.”
Dr. Thompson said geriatricians are teaching primary care specialists, nurses, social workers, and other health care providers the principles of age-friendly care. AGS has for the past 20 years led a program called the Geriatrics for Specialists Initiative to increase geriatrics knowledge and expertise of surgical and medical specialists.
Some specialties have taken the cue and have added geriatrics-related hyphens through additional training: geriatric-emergency, geriatric-general surgery, geriatric-hospitalists, and more.
HRSA runs programs to encourage physicians to train as geriatricians and geriatrics faculty, and it encourages the geriatrics interdisciplinary team approach.
Richard Olague, director of public affairs for HRSA, said his agency has invested over $160 million over the past 4 years in the education and training of geriatricians and other health care professionals who care for the elderly through its Geriatrics Workforce Enhancement Program and Geriatrics Academic Career Awards Program. In the academic year 2020-2021, the two programs trained 109 geriatricians; 456 other geriatric/gerontology providers and students; 44,450 other healthcare workforce professionals and students; and served 17,666 patients and 5,409 caregivers.
Dr. Harper, like his fellow geriatricians, tells young doctors that geriatrics is a fulfilling specialty.
“I get to care for the whole person and sometimes their families, too, and in the process form rich and meaningful relationships. And while I’m rarely in the position to cure, I always have the ability to care,” he said. “Sometimes that can mean being an advocate trying to make sure my patients receive the care they need, and other times it might mean protecting them from burdensome care that is unlikely to lead to any meaningful benefit. There is great reward in all of that.”
Dr. Supiano said geriatric patients are being helped by the Age-Friendly Health System initiative of the John A. Hartford Foundation and the Institute for Healthcare Improvement in partnership with the American Hospital Association and the Catholic Health Association of the United States. This is sort of a seal of approval for facilities committed to age-friendly care.
“When you go to your hospital, if they don’t have this age-friendly health system banner on the front door ... you either ask why that is not there, or you vote with your feet and go to another health system that is age friendly,” he said. “Geriatricians are eternal optimists.”
A version of this article first appeared on Medscape.com.
These should be the best of times for geriatric medicine.
The baby boom has become a senior surge, bringing in a rapidly growing pool of aging patients for geriatricians to treat. According to the U.S. Census Bureau, more than 56 million adults aged 65 and older live in the United States. They account for about 17% of the nation’s population. That number is expected to hit 73 million by 2030 and 86 million by 2050.
The American Geriatrics Society estimates that 30% of older people require the attention of geriatricians. These clinicians excel in managing complex cases – patients with multiple comorbidities, such as coronary artery disease, dementia, and osteoporosis, who are taking a half dozen, and often more, medications.
. In the 2010s, geriatricians called for “25,000 [such specialists] by 2025.” As of 2021, 7123 certified geriatricians were practicing in the United States, according to the American Board of Medical Specialties.
The Health Resources and Services Administration, a federal agency that addresses medical workforce shortages, estimates that there will be 6,230 geriatricians by 2025, or approximately 1 for every 3,000 older adults requiring geriatric care. HRSA projects a shortage of 27,000 geriatricians by 2025.
The specialty has faced an uphill battle to attract fellows. This year, only 43% of the nation’s 177 geriatrics fellowship slots were filled, according to November’s National Resident Match Program report. Family medicine–based geriatrics achieved only a 32% fill rate, while internal medicine–based programs saw a rate of 45%.
“Our numbers are shrinking so we need another approach to make sure older adults get the care they need and deserve,” said G. Michael Harper, MD, president of the 6,000-member AGS.
But Dr. Harper, who practices at the University of California, San Francisco, and the San Francisco VA Medical Center, added a positive note: “We may be struggling to increase the number of board-certified geriatricians, but the field itself has made a lot of progress in terms of improving clinical care through advancements in science and in the ways we deliver care.”
Dr. Harper cited the Hospital Elder Life Program, a hospital model developed at the Harvard-affiliated Marcus Institute for Aging Research, which uses an interprofessional team and trained volunteers to prevent delirium and functional decline. HELP has been adopted by more than 200 hospitals worldwide and has been successful at returning older adults to their homes or previous living situations with maintained or improved ability to function, he said.
Mark Supiano, MD, professor and chief of geriatrics at the University of Utah, Salt Lake City, said the specialty has been in shortage mode since ABMS recognized it in 1988. He was in the initial cohort of fellowship-trained geriatricians, sitting for the first certifying exam in geriatrics offered that year.
“Back then, the demographic imperative of the aging of our society was on the horizon. We’re living it now. I knew enough to recognize it was coming and saw an opportunity,” Dr. Supiano said in an interview. “There was so much then that we didn’t know about how to understand aging or how to care for older adults that there really was such a knowledge gap.”
Dr. Supiano is an associate editor of Hazzard’s Geriatric Medicine and Gerontology (McGraw-Hill Education), which has more than doubled in pages and word count during his career.
Unfavorable finances
Katherine Thompson, MD, director of the geriatrics fellowship program at the University of Chicago and codirector of UChicago’s Successful Aging and Frailty Evaluation Clinic, said money is a major reason for the struggle. “I think probably the biggest driver is financial,” she said. “A lot of people are graduating medical school with really astronomical amounts of medical school loans.”
Geriatricians, like other doctors, carry a large debt – $200,000, on average, not counting undergraduate debt, according to the Association of American Medical Colleges.
But the typical geriatrician earns less than an internist or family medicine doctor who doesn’t undergo the additional year of training, Dr. Thompson said. “There’s not a lot of financial motivation to do this fellowship,” she said.
The jobs website Zippia reports that geriatricians earned roughly $165,000 per year on average in 2022. The average annual incomes in 2022 were $191,000 for pediatricians, $215,000 for family physicians, and $223,000 for internists, according to the site.
In other words, Dr. Harper said, “geriatrics is one of the few professions where you can actually do additional training and make less money.”
The reason for the pay issue is simple: Geriatricians treat patients covered by Medicare, whose reimbursement schedules lag behind those of commercial insurers. The Kaiser Family Foundation reported in 2020 that private insurance paid 143% of Medicare rates on average for physician services.
Dr. Harper said overall compensation for geriatricians has “not gained a lot of traction,” but they can earn comfortable livings.
Still, representation of the specialty on the American Medical Association’s Relative Value Scale Update Committee has led to approval by the Centers for Medicare & Medicaid Services of billing codes that pay geriatricians “for what they do. Examples include chronic care management, advance care planning, and dementia evaluation,” he said.
But the geriatrician gap goes beyond money.
Ageism, too, may play a role in residents not choosing geriatrics.
“Our culture is ageist. It definitely focuses on youth and looks at aging as being loss rather than just a change in what works well and what doesn’t work well,” said Mary Tinetti, MD, a geriatrician and researcher at Yale University, New Haven, Conn. “Ageism happens among physicians, just because they’re part of the broader society.”
Time for a new goal?
Dr. Tinetti said she’s optimistic that new ideas about geriatricians teaching other primary care clinicians about the tenets of geriatric medicine, which offer a wholistic approach to comorbidities, such as diabetes, atrial fibrillation, dementia, hypertension, hyperlipidemia, and polypharmacy problems faced by this population, especially those 85 and older.
She has called on her profession to abandon the goal of increasing the numbers of board-certified geriatricians – whom she refers to as big “G” geriatricians. She instead wants to develop a “small, elite workforce” that discovers and tests geriatrics principles through research, teaches these principles to all healthcare professions and to the public, and disseminates and implements the policies.
“We need a cadre of geriatricians who train all other clinicians in the care of older adults,” Dr. Tinetti said. “The goal is not more geriatricians but rather the preparation of all clinicians in the care of older adults.”
Dr. Thompson said geriatricians are teaching primary care specialists, nurses, social workers, and other health care providers the principles of age-friendly care. AGS has for the past 20 years led a program called the Geriatrics for Specialists Initiative to increase geriatrics knowledge and expertise of surgical and medical specialists.
Some specialties have taken the cue and have added geriatrics-related hyphens through additional training: geriatric-emergency, geriatric-general surgery, geriatric-hospitalists, and more.
HRSA runs programs to encourage physicians to train as geriatricians and geriatrics faculty, and it encourages the geriatrics interdisciplinary team approach.
Richard Olague, director of public affairs for HRSA, said his agency has invested over $160 million over the past 4 years in the education and training of geriatricians and other health care professionals who care for the elderly through its Geriatrics Workforce Enhancement Program and Geriatrics Academic Career Awards Program. In the academic year 2020-2021, the two programs trained 109 geriatricians; 456 other geriatric/gerontology providers and students; 44,450 other healthcare workforce professionals and students; and served 17,666 patients and 5,409 caregivers.
Dr. Harper, like his fellow geriatricians, tells young doctors that geriatrics is a fulfilling specialty.
“I get to care for the whole person and sometimes their families, too, and in the process form rich and meaningful relationships. And while I’m rarely in the position to cure, I always have the ability to care,” he said. “Sometimes that can mean being an advocate trying to make sure my patients receive the care they need, and other times it might mean protecting them from burdensome care that is unlikely to lead to any meaningful benefit. There is great reward in all of that.”
Dr. Supiano said geriatric patients are being helped by the Age-Friendly Health System initiative of the John A. Hartford Foundation and the Institute for Healthcare Improvement in partnership with the American Hospital Association and the Catholic Health Association of the United States. This is sort of a seal of approval for facilities committed to age-friendly care.
“When you go to your hospital, if they don’t have this age-friendly health system banner on the front door ... you either ask why that is not there, or you vote with your feet and go to another health system that is age friendly,” he said. “Geriatricians are eternal optimists.”
A version of this article first appeared on Medscape.com.
These should be the best of times for geriatric medicine.
The baby boom has become a senior surge, bringing in a rapidly growing pool of aging patients for geriatricians to treat. According to the U.S. Census Bureau, more than 56 million adults aged 65 and older live in the United States. They account for about 17% of the nation’s population. That number is expected to hit 73 million by 2030 and 86 million by 2050.
The American Geriatrics Society estimates that 30% of older people require the attention of geriatricians. These clinicians excel in managing complex cases – patients with multiple comorbidities, such as coronary artery disease, dementia, and osteoporosis, who are taking a half dozen, and often more, medications.
. In the 2010s, geriatricians called for “25,000 [such specialists] by 2025.” As of 2021, 7123 certified geriatricians were practicing in the United States, according to the American Board of Medical Specialties.
The Health Resources and Services Administration, a federal agency that addresses medical workforce shortages, estimates that there will be 6,230 geriatricians by 2025, or approximately 1 for every 3,000 older adults requiring geriatric care. HRSA projects a shortage of 27,000 geriatricians by 2025.
The specialty has faced an uphill battle to attract fellows. This year, only 43% of the nation’s 177 geriatrics fellowship slots were filled, according to November’s National Resident Match Program report. Family medicine–based geriatrics achieved only a 32% fill rate, while internal medicine–based programs saw a rate of 45%.
“Our numbers are shrinking so we need another approach to make sure older adults get the care they need and deserve,” said G. Michael Harper, MD, president of the 6,000-member AGS.
But Dr. Harper, who practices at the University of California, San Francisco, and the San Francisco VA Medical Center, added a positive note: “We may be struggling to increase the number of board-certified geriatricians, but the field itself has made a lot of progress in terms of improving clinical care through advancements in science and in the ways we deliver care.”
Dr. Harper cited the Hospital Elder Life Program, a hospital model developed at the Harvard-affiliated Marcus Institute for Aging Research, which uses an interprofessional team and trained volunteers to prevent delirium and functional decline. HELP has been adopted by more than 200 hospitals worldwide and has been successful at returning older adults to their homes or previous living situations with maintained or improved ability to function, he said.
Mark Supiano, MD, professor and chief of geriatrics at the University of Utah, Salt Lake City, said the specialty has been in shortage mode since ABMS recognized it in 1988. He was in the initial cohort of fellowship-trained geriatricians, sitting for the first certifying exam in geriatrics offered that year.
“Back then, the demographic imperative of the aging of our society was on the horizon. We’re living it now. I knew enough to recognize it was coming and saw an opportunity,” Dr. Supiano said in an interview. “There was so much then that we didn’t know about how to understand aging or how to care for older adults that there really was such a knowledge gap.”
Dr. Supiano is an associate editor of Hazzard’s Geriatric Medicine and Gerontology (McGraw-Hill Education), which has more than doubled in pages and word count during his career.
Unfavorable finances
Katherine Thompson, MD, director of the geriatrics fellowship program at the University of Chicago and codirector of UChicago’s Successful Aging and Frailty Evaluation Clinic, said money is a major reason for the struggle. “I think probably the biggest driver is financial,” she said. “A lot of people are graduating medical school with really astronomical amounts of medical school loans.”
Geriatricians, like other doctors, carry a large debt – $200,000, on average, not counting undergraduate debt, according to the Association of American Medical Colleges.
But the typical geriatrician earns less than an internist or family medicine doctor who doesn’t undergo the additional year of training, Dr. Thompson said. “There’s not a lot of financial motivation to do this fellowship,” she said.
The jobs website Zippia reports that geriatricians earned roughly $165,000 per year on average in 2022. The average annual incomes in 2022 were $191,000 for pediatricians, $215,000 for family physicians, and $223,000 for internists, according to the site.
In other words, Dr. Harper said, “geriatrics is one of the few professions where you can actually do additional training and make less money.”
The reason for the pay issue is simple: Geriatricians treat patients covered by Medicare, whose reimbursement schedules lag behind those of commercial insurers. The Kaiser Family Foundation reported in 2020 that private insurance paid 143% of Medicare rates on average for physician services.
Dr. Harper said overall compensation for geriatricians has “not gained a lot of traction,” but they can earn comfortable livings.
Still, representation of the specialty on the American Medical Association’s Relative Value Scale Update Committee has led to approval by the Centers for Medicare & Medicaid Services of billing codes that pay geriatricians “for what they do. Examples include chronic care management, advance care planning, and dementia evaluation,” he said.
But the geriatrician gap goes beyond money.
Ageism, too, may play a role in residents not choosing geriatrics.
“Our culture is ageist. It definitely focuses on youth and looks at aging as being loss rather than just a change in what works well and what doesn’t work well,” said Mary Tinetti, MD, a geriatrician and researcher at Yale University, New Haven, Conn. “Ageism happens among physicians, just because they’re part of the broader society.”
Time for a new goal?
Dr. Tinetti said she’s optimistic that new ideas about geriatricians teaching other primary care clinicians about the tenets of geriatric medicine, which offer a wholistic approach to comorbidities, such as diabetes, atrial fibrillation, dementia, hypertension, hyperlipidemia, and polypharmacy problems faced by this population, especially those 85 and older.
She has called on her profession to abandon the goal of increasing the numbers of board-certified geriatricians – whom she refers to as big “G” geriatricians. She instead wants to develop a “small, elite workforce” that discovers and tests geriatrics principles through research, teaches these principles to all healthcare professions and to the public, and disseminates and implements the policies.
“We need a cadre of geriatricians who train all other clinicians in the care of older adults,” Dr. Tinetti said. “The goal is not more geriatricians but rather the preparation of all clinicians in the care of older adults.”
Dr. Thompson said geriatricians are teaching primary care specialists, nurses, social workers, and other health care providers the principles of age-friendly care. AGS has for the past 20 years led a program called the Geriatrics for Specialists Initiative to increase geriatrics knowledge and expertise of surgical and medical specialists.
Some specialties have taken the cue and have added geriatrics-related hyphens through additional training: geriatric-emergency, geriatric-general surgery, geriatric-hospitalists, and more.
HRSA runs programs to encourage physicians to train as geriatricians and geriatrics faculty, and it encourages the geriatrics interdisciplinary team approach.
Richard Olague, director of public affairs for HRSA, said his agency has invested over $160 million over the past 4 years in the education and training of geriatricians and other health care professionals who care for the elderly through its Geriatrics Workforce Enhancement Program and Geriatrics Academic Career Awards Program. In the academic year 2020-2021, the two programs trained 109 geriatricians; 456 other geriatric/gerontology providers and students; 44,450 other healthcare workforce professionals and students; and served 17,666 patients and 5,409 caregivers.
Dr. Harper, like his fellow geriatricians, tells young doctors that geriatrics is a fulfilling specialty.
“I get to care for the whole person and sometimes their families, too, and in the process form rich and meaningful relationships. And while I’m rarely in the position to cure, I always have the ability to care,” he said. “Sometimes that can mean being an advocate trying to make sure my patients receive the care they need, and other times it might mean protecting them from burdensome care that is unlikely to lead to any meaningful benefit. There is great reward in all of that.”
Dr. Supiano said geriatric patients are being helped by the Age-Friendly Health System initiative of the John A. Hartford Foundation and the Institute for Healthcare Improvement in partnership with the American Hospital Association and the Catholic Health Association of the United States. This is sort of a seal of approval for facilities committed to age-friendly care.
“When you go to your hospital, if they don’t have this age-friendly health system banner on the front door ... you either ask why that is not there, or you vote with your feet and go to another health system that is age friendly,” he said. “Geriatricians are eternal optimists.”
A version of this article first appeared on Medscape.com.
EHR alerts to both doc and patient may boost statin prescribing
Automated alerts to aid clinical decision-making are designed with the best of intentions but can be easy to ignore or overlook. But a randomized trial testing such electronic alerts or “nudges” for promoting statin prescribing may have identified a few design features that help their success, researchers say.
In the trial’s primary finding, for example, reminders displayed to primary care physicians in the electronic health record worked best when the system also reached out to the patient.
Reminders sent only to the clinician also boosted statin prescribing, but not as well, and nudging only the patient didn’t work at all, compared to a nudge-free usual care approach. The patient-only nudges consisted of text messages explaining why a statin prescription may figure in their upcoming appointment.
Nudge trustworthiness
Importantly, the clinician nudges were more than simply reminders to consider a statin prescription, Mitesh S. Patel, MD, MBA, Ascension Health, St. Louis, told this news organization. They also displayed the patient’s atherosclerotic cardiovascular disease (ASCVD) 10-year risk score and explained why a statin may be appropriate. He thinks that information, often left out of such clinical decision support alerts, increases physician trust in them.
In another key feature, Dr. Patel said, the EHR nudges themselves were actionable – that is, they were functional in ways that streamlined the prescribing process. In particular, they include checkbox shortcuts to prescribing statins at appropriate patient-specific dosages, making the entire process “faster and easier,” said Dr. Patel, who is senior author on the study published in JAMA Cardiology with lead author Srinath Adusumalli, MD, University of Pennsylvania, Philadelphia.
The timing may matter as well, he observed. In previous iterations of the study’s EHR nudge system, the nudge would appear “when you open the chart,” he said. “Now, it’s when you go to the orders section, which is when you’re going to be in the mindset of ordering prescriptions and tests.”
Prescription rates were higher with the doctor-patient nudges than with the doctor-only approach, Dr. Patel speculates, largely because the decision process for initiating statins is shared. “The most effective intervention is going to recognize that and try to bring the two groups together.”
Two text messages
The trial, with 158 participating physicians in 28 primary care practices, randomly assigned 4,131 patients to three intervention groups and one control group. Nudges were sent only to the physician, only to the patient, or to both physician and patient; and there was a no-nudge usual-care group.
Patient nudges consisted of two text messages, one 4 days and another 15 minutes before the appointment, announcing that prescription of a statin “to reduce the chance of a heart attack” would be discussed with the physician, the report states.
Statins are grossly underprescribed nationally, it notes, and that was reflected in prescription rates seen during the study’s initial 12-month, no-intervention period of observation. Rates ranged from only 4.7% up to 6% of patients across the four assignment groups.
During the subsequent 6-month intervention period, however, the rates climbed in the doctor-only and doctor-plus-patient nudge groups compared with usual care, by 5.5 (P = .01) and 7.2 (P = .001) absolute percentage points, respectively.
The overall cohort’s mean age was 65.5. About half were male, 29% were Black, 66% were White, and 22.6% already had a cardiovascular disease diagnosis. The analysis was adjusted for calendar month and preintervention statin prescribing rates. Further adjustment for demographics, insurance type, household income, and comorbidities yielded results similar to the primary analysis, the report states.
The results in context
“Although the differences in the combined clinician and patient and clinician-only arms were small, this outcome needs to be interpreted in the context of the population in which the study was performed,” an editorial accompanying the published report states.
For example, “the majority of untreated patients were candidates for primary, not secondary, prevention, making this group of patients particularly challenging for seeing large effect sizes of interventions.”
Moreover, “There was a high baseline prescription rate of statins in the statin-eligible population (approximately 70%) and a high rate of already established patients,” write Faraz S. Ahmad, MD, and Stephen D. Persell, MD, of Northwestern University, Chicago.
Among the approximately 30% of patients who had not previously been prescribed statins, the true target of the nudge interventions, the published trial report states, about 98% were not seeing the physician for the first time.
So “this may not have been the first opportunity to discuss statins,” they write. “It is possible that many of these patients were resistant to statins in the past, which could have created a ceiling effect for prescribing rates.”
Dr. Patel reports owning and receiving personal fees from Catalyst Health and serving on an advisory board for and receiving personal fees from Humana. Dr. Adusumalli reports having been employed by CVS Health. Dr. Ahmad reports receiving consulting fees from Teladoc Livongo and Pfizer. Dr. Persell discloses receiving grants from Omron Healthcare.
A version of this article first appeared on Medscape.com.
Automated alerts to aid clinical decision-making are designed with the best of intentions but can be easy to ignore or overlook. But a randomized trial testing such electronic alerts or “nudges” for promoting statin prescribing may have identified a few design features that help their success, researchers say.
In the trial’s primary finding, for example, reminders displayed to primary care physicians in the electronic health record worked best when the system also reached out to the patient.
Reminders sent only to the clinician also boosted statin prescribing, but not as well, and nudging only the patient didn’t work at all, compared to a nudge-free usual care approach. The patient-only nudges consisted of text messages explaining why a statin prescription may figure in their upcoming appointment.
Nudge trustworthiness
Importantly, the clinician nudges were more than simply reminders to consider a statin prescription, Mitesh S. Patel, MD, MBA, Ascension Health, St. Louis, told this news organization. They also displayed the patient’s atherosclerotic cardiovascular disease (ASCVD) 10-year risk score and explained why a statin may be appropriate. He thinks that information, often left out of such clinical decision support alerts, increases physician trust in them.
In another key feature, Dr. Patel said, the EHR nudges themselves were actionable – that is, they were functional in ways that streamlined the prescribing process. In particular, they include checkbox shortcuts to prescribing statins at appropriate patient-specific dosages, making the entire process “faster and easier,” said Dr. Patel, who is senior author on the study published in JAMA Cardiology with lead author Srinath Adusumalli, MD, University of Pennsylvania, Philadelphia.
The timing may matter as well, he observed. In previous iterations of the study’s EHR nudge system, the nudge would appear “when you open the chart,” he said. “Now, it’s when you go to the orders section, which is when you’re going to be in the mindset of ordering prescriptions and tests.”
Prescription rates were higher with the doctor-patient nudges than with the doctor-only approach, Dr. Patel speculates, largely because the decision process for initiating statins is shared. “The most effective intervention is going to recognize that and try to bring the two groups together.”
Two text messages
The trial, with 158 participating physicians in 28 primary care practices, randomly assigned 4,131 patients to three intervention groups and one control group. Nudges were sent only to the physician, only to the patient, or to both physician and patient; and there was a no-nudge usual-care group.
Patient nudges consisted of two text messages, one 4 days and another 15 minutes before the appointment, announcing that prescription of a statin “to reduce the chance of a heart attack” would be discussed with the physician, the report states.
Statins are grossly underprescribed nationally, it notes, and that was reflected in prescription rates seen during the study’s initial 12-month, no-intervention period of observation. Rates ranged from only 4.7% up to 6% of patients across the four assignment groups.
During the subsequent 6-month intervention period, however, the rates climbed in the doctor-only and doctor-plus-patient nudge groups compared with usual care, by 5.5 (P = .01) and 7.2 (P = .001) absolute percentage points, respectively.
The overall cohort’s mean age was 65.5. About half were male, 29% were Black, 66% were White, and 22.6% already had a cardiovascular disease diagnosis. The analysis was adjusted for calendar month and preintervention statin prescribing rates. Further adjustment for demographics, insurance type, household income, and comorbidities yielded results similar to the primary analysis, the report states.
The results in context
“Although the differences in the combined clinician and patient and clinician-only arms were small, this outcome needs to be interpreted in the context of the population in which the study was performed,” an editorial accompanying the published report states.
For example, “the majority of untreated patients were candidates for primary, not secondary, prevention, making this group of patients particularly challenging for seeing large effect sizes of interventions.”
Moreover, “There was a high baseline prescription rate of statins in the statin-eligible population (approximately 70%) and a high rate of already established patients,” write Faraz S. Ahmad, MD, and Stephen D. Persell, MD, of Northwestern University, Chicago.
Among the approximately 30% of patients who had not previously been prescribed statins, the true target of the nudge interventions, the published trial report states, about 98% were not seeing the physician for the first time.
So “this may not have been the first opportunity to discuss statins,” they write. “It is possible that many of these patients were resistant to statins in the past, which could have created a ceiling effect for prescribing rates.”
Dr. Patel reports owning and receiving personal fees from Catalyst Health and serving on an advisory board for and receiving personal fees from Humana. Dr. Adusumalli reports having been employed by CVS Health. Dr. Ahmad reports receiving consulting fees from Teladoc Livongo and Pfizer. Dr. Persell discloses receiving grants from Omron Healthcare.
A version of this article first appeared on Medscape.com.
Automated alerts to aid clinical decision-making are designed with the best of intentions but can be easy to ignore or overlook. But a randomized trial testing such electronic alerts or “nudges” for promoting statin prescribing may have identified a few design features that help their success, researchers say.
In the trial’s primary finding, for example, reminders displayed to primary care physicians in the electronic health record worked best when the system also reached out to the patient.
Reminders sent only to the clinician also boosted statin prescribing, but not as well, and nudging only the patient didn’t work at all, compared to a nudge-free usual care approach. The patient-only nudges consisted of text messages explaining why a statin prescription may figure in their upcoming appointment.
Nudge trustworthiness
Importantly, the clinician nudges were more than simply reminders to consider a statin prescription, Mitesh S. Patel, MD, MBA, Ascension Health, St. Louis, told this news organization. They also displayed the patient’s atherosclerotic cardiovascular disease (ASCVD) 10-year risk score and explained why a statin may be appropriate. He thinks that information, often left out of such clinical decision support alerts, increases physician trust in them.
In another key feature, Dr. Patel said, the EHR nudges themselves were actionable – that is, they were functional in ways that streamlined the prescribing process. In particular, they include checkbox shortcuts to prescribing statins at appropriate patient-specific dosages, making the entire process “faster and easier,” said Dr. Patel, who is senior author on the study published in JAMA Cardiology with lead author Srinath Adusumalli, MD, University of Pennsylvania, Philadelphia.
The timing may matter as well, he observed. In previous iterations of the study’s EHR nudge system, the nudge would appear “when you open the chart,” he said. “Now, it’s when you go to the orders section, which is when you’re going to be in the mindset of ordering prescriptions and tests.”
Prescription rates were higher with the doctor-patient nudges than with the doctor-only approach, Dr. Patel speculates, largely because the decision process for initiating statins is shared. “The most effective intervention is going to recognize that and try to bring the two groups together.”
Two text messages
The trial, with 158 participating physicians in 28 primary care practices, randomly assigned 4,131 patients to three intervention groups and one control group. Nudges were sent only to the physician, only to the patient, or to both physician and patient; and there was a no-nudge usual-care group.
Patient nudges consisted of two text messages, one 4 days and another 15 minutes before the appointment, announcing that prescription of a statin “to reduce the chance of a heart attack” would be discussed with the physician, the report states.
Statins are grossly underprescribed nationally, it notes, and that was reflected in prescription rates seen during the study’s initial 12-month, no-intervention period of observation. Rates ranged from only 4.7% up to 6% of patients across the four assignment groups.
During the subsequent 6-month intervention period, however, the rates climbed in the doctor-only and doctor-plus-patient nudge groups compared with usual care, by 5.5 (P = .01) and 7.2 (P = .001) absolute percentage points, respectively.
The overall cohort’s mean age was 65.5. About half were male, 29% were Black, 66% were White, and 22.6% already had a cardiovascular disease diagnosis. The analysis was adjusted for calendar month and preintervention statin prescribing rates. Further adjustment for demographics, insurance type, household income, and comorbidities yielded results similar to the primary analysis, the report states.
The results in context
“Although the differences in the combined clinician and patient and clinician-only arms were small, this outcome needs to be interpreted in the context of the population in which the study was performed,” an editorial accompanying the published report states.
For example, “the majority of untreated patients were candidates for primary, not secondary, prevention, making this group of patients particularly challenging for seeing large effect sizes of interventions.”
Moreover, “There was a high baseline prescription rate of statins in the statin-eligible population (approximately 70%) and a high rate of already established patients,” write Faraz S. Ahmad, MD, and Stephen D. Persell, MD, of Northwestern University, Chicago.
Among the approximately 30% of patients who had not previously been prescribed statins, the true target of the nudge interventions, the published trial report states, about 98% were not seeing the physician for the first time.
So “this may not have been the first opportunity to discuss statins,” they write. “It is possible that many of these patients were resistant to statins in the past, which could have created a ceiling effect for prescribing rates.”
Dr. Patel reports owning and receiving personal fees from Catalyst Health and serving on an advisory board for and receiving personal fees from Humana. Dr. Adusumalli reports having been employed by CVS Health. Dr. Ahmad reports receiving consulting fees from Teladoc Livongo and Pfizer. Dr. Persell discloses receiving grants from Omron Healthcare.
A version of this article first appeared on Medscape.com.
‘Game changer’: Thyroid cancer recurrence no higher with lobectomy
Patients with intermediate-risk papillary thyroid cancer and lymph node metastasis show no significant increase in tumor recurrence when undergoing lobectomy compared with a total thyroidectomy, new research shows.
“Results of this cohort study suggest that patients with ipsilateral clinical lateral neck metastasis (cN1b) papillary thyroid cancer who underwent lobectomy exhibited recurrence-free survival rates similar to those who underwent total thyroidectomy after controlling for major prognostic factors,” the authors conclude in the study published online in JAMA Surgery.
“These findings suggest that cN1b alone should not be an absolute indication for total thyroidectomy,” they note.
The study, involving the largest cohort to date to compare patients with intermediate-risk papillary thyroid cancer treated with lobectomy versus total thyroidectomy, “challenged the current guidelines and pushed the boundary of limited surgical treatment even further,” say Michelle B. Mulder, MD, and Quan-Yang Duh, MD, of the department of surgery, University of California, San Francisco, in an accompanying editorial.
“It can be a game changer if confirmed by future prospective and multicenter studies,” they add.
Guidelines still recommend total thyroidectomy with subsequent RAI
While lower-intensity treatment options, with a lower risk of complications, have gained favor in the treatment of low-risk papillary thyroid cancer, guidelines still recommend the consideration of total thyroidectomy and subsequent radioactive iodine ablation (RAI) for intermediate-risk cancers because of the higher chance of recurrence, particularly among those with clinically positive nodes.
However, data on the superiority of a total thyroidectomy, with or without RAI, versus lobectomy is inconsistent, prompting first author Siyuan Xu, MD, of the department of head and neck surgical oncology, National Cancer Center, Beijing, and colleagues to compare the risk of recurrence with the two approaches.
For the study, patients with intermediate-risk papillary thyroid cancer treated at the Chinese Academy of Medical Sciences Cancer Hospital in Beijing between January 2000 and December 2017, who had a lobectomy or total thyroidectomy, were paired 1:1 in a propensity score matching analysis.
Other than treatment type, the 265 pairs of patients were matched based on all other potential prognostic factors, including age, sex, primary tumor size, minor extrathyroidal extension, multifocality, number of lymph node metastases, and lymph node ratio.
Participants were a mean age of 37 years and 66% were female.
With a median follow-up of 60 months in the lobectomy group and 58 months in the total thyroidectomy group, structural recurrences occurred in 7.9% (21) and 6.4% (17) of patients, respectively, which was not significantly different.
The primary endpoint, 5-year rate of recurrence-free survival, was also not significantly different between the lobectomy (92.3%) and total thyroidectomy groups (93.7%) (adjusted hazard ratio, 1.10; P = .77).
In a further stratified analysis of patients treated with total thyroidectomy along with RAI (n = 75), the lack of a significant difference in recurrence-free survival versus lobectomy remained (aHR, 0.59; P = .46).
The results were similar in unadjusted as well as adjusted analyses, and a power analysis indicated that the study had a 90% power to detect a more than 4.9% difference in recurrence-free survival.
“Given the lower complication rate of lobectomy, a maximal 4.9% recurrence-free survival difference is acceptable, which enhances the reliability of the study results,” the authors say.
They conclude that “our findings call into question whether cN1b alone [ipsilateral clinical lateral neck metastasis papillary thyroid cancer] should be an absolute determinant for deciding the optimal extent of thyroid surgery for papillary thyroid cancer.”
With total thyroidectomy, RAI can be given
An important argument in favor of total thyroidectomy is that with the complete resection of thyroid tissue, RAI ablation can then be used for postoperative detection of residual or metastatic disease, as well as for treatment, the authors note.
Indeed, a study using the Surveillance, Epidemiology, and End Results (SEER) database showed RAI ablation is associated with a 29% reduction in the risk of death in patients with intermediate-risk papillary thyroid cancer, with a hazard risk of 0.71.
However, conflicting data from Memorial Sloan-Kettering Cancer Center, New York, suggests no significant benefit with total thyroidectomy and RAI ablation.
The current study’s analysis of patients treated with RAI, though limited in size, supports the latter study’s findings, the authors note.
“When we performed further stratified analyses in patients treated with total thyroidectomy plus RAI ablation and their counterparts, no significant difference was found, which conformed with [the] result from the whole cohort.”
“Certainly, the stratified comparison did not have enough power to examine the effect of RAI ablation on tumor recurrence subject to the limitation of sample size and case selection [and] further study is needed on this topic,” they write.
Some limitations warrant cautious interpretation
In their editorial, Dr. Mulder and Dr. Duh note that while some previous studies have shown similar outcomes relating to tumor size, thyroid hormone suppression therapy, and multifocality, “few have addressed lateral neck involvement.”
They suggest cautious interpretation, however, due to limitations, acknowledged by the authors, including the single-center nature of the study.
“Appropriate propensity matching may mitigate selection bias but cannot eliminate it entirely and their findings may not be replicated in other institutions by other surgeons,” they note.
Other limitations include that changes in clinical practice and patient selection were likely over the course of the study because of significant changes in American Thyroid Association (ATA) guidelines between 2009 and 2017, and characteristics including molecular genetic testing, which could have influenced final results, were not taken into consideration.
Furthermore, for patients with intermediate-risk cancer, modifications in postoperative follow-up are necessary following lobectomy versus total thyroidectomy; “the role of radioiodine is limited and the levels of thyroglobulin more complicated to interpret,” they note.
The study and editorial authors had no disclosures to report.
A version of this article first appeared on Medscape.com.
Patients with intermediate-risk papillary thyroid cancer and lymph node metastasis show no significant increase in tumor recurrence when undergoing lobectomy compared with a total thyroidectomy, new research shows.
“Results of this cohort study suggest that patients with ipsilateral clinical lateral neck metastasis (cN1b) papillary thyroid cancer who underwent lobectomy exhibited recurrence-free survival rates similar to those who underwent total thyroidectomy after controlling for major prognostic factors,” the authors conclude in the study published online in JAMA Surgery.
“These findings suggest that cN1b alone should not be an absolute indication for total thyroidectomy,” they note.
The study, involving the largest cohort to date to compare patients with intermediate-risk papillary thyroid cancer treated with lobectomy versus total thyroidectomy, “challenged the current guidelines and pushed the boundary of limited surgical treatment even further,” say Michelle B. Mulder, MD, and Quan-Yang Duh, MD, of the department of surgery, University of California, San Francisco, in an accompanying editorial.
“It can be a game changer if confirmed by future prospective and multicenter studies,” they add.
Guidelines still recommend total thyroidectomy with subsequent RAI
While lower-intensity treatment options, with a lower risk of complications, have gained favor in the treatment of low-risk papillary thyroid cancer, guidelines still recommend the consideration of total thyroidectomy and subsequent radioactive iodine ablation (RAI) for intermediate-risk cancers because of the higher chance of recurrence, particularly among those with clinically positive nodes.
However, data on the superiority of a total thyroidectomy, with or without RAI, versus lobectomy is inconsistent, prompting first author Siyuan Xu, MD, of the department of head and neck surgical oncology, National Cancer Center, Beijing, and colleagues to compare the risk of recurrence with the two approaches.
For the study, patients with intermediate-risk papillary thyroid cancer treated at the Chinese Academy of Medical Sciences Cancer Hospital in Beijing between January 2000 and December 2017, who had a lobectomy or total thyroidectomy, were paired 1:1 in a propensity score matching analysis.
Other than treatment type, the 265 pairs of patients were matched based on all other potential prognostic factors, including age, sex, primary tumor size, minor extrathyroidal extension, multifocality, number of lymph node metastases, and lymph node ratio.
Participants were a mean age of 37 years and 66% were female.
With a median follow-up of 60 months in the lobectomy group and 58 months in the total thyroidectomy group, structural recurrences occurred in 7.9% (21) and 6.4% (17) of patients, respectively, which was not significantly different.
The primary endpoint, 5-year rate of recurrence-free survival, was also not significantly different between the lobectomy (92.3%) and total thyroidectomy groups (93.7%) (adjusted hazard ratio, 1.10; P = .77).
In a further stratified analysis of patients treated with total thyroidectomy along with RAI (n = 75), the lack of a significant difference in recurrence-free survival versus lobectomy remained (aHR, 0.59; P = .46).
The results were similar in unadjusted as well as adjusted analyses, and a power analysis indicated that the study had a 90% power to detect a more than 4.9% difference in recurrence-free survival.
“Given the lower complication rate of lobectomy, a maximal 4.9% recurrence-free survival difference is acceptable, which enhances the reliability of the study results,” the authors say.
They conclude that “our findings call into question whether cN1b alone [ipsilateral clinical lateral neck metastasis papillary thyroid cancer] should be an absolute determinant for deciding the optimal extent of thyroid surgery for papillary thyroid cancer.”
With total thyroidectomy, RAI can be given
An important argument in favor of total thyroidectomy is that with the complete resection of thyroid tissue, RAI ablation can then be used for postoperative detection of residual or metastatic disease, as well as for treatment, the authors note.
Indeed, a study using the Surveillance, Epidemiology, and End Results (SEER) database showed RAI ablation is associated with a 29% reduction in the risk of death in patients with intermediate-risk papillary thyroid cancer, with a hazard risk of 0.71.
However, conflicting data from Memorial Sloan-Kettering Cancer Center, New York, suggests no significant benefit with total thyroidectomy and RAI ablation.
The current study’s analysis of patients treated with RAI, though limited in size, supports the latter study’s findings, the authors note.
“When we performed further stratified analyses in patients treated with total thyroidectomy plus RAI ablation and their counterparts, no significant difference was found, which conformed with [the] result from the whole cohort.”
“Certainly, the stratified comparison did not have enough power to examine the effect of RAI ablation on tumor recurrence subject to the limitation of sample size and case selection [and] further study is needed on this topic,” they write.
Some limitations warrant cautious interpretation
In their editorial, Dr. Mulder and Dr. Duh note that while some previous studies have shown similar outcomes relating to tumor size, thyroid hormone suppression therapy, and multifocality, “few have addressed lateral neck involvement.”
They suggest cautious interpretation, however, due to limitations, acknowledged by the authors, including the single-center nature of the study.
“Appropriate propensity matching may mitigate selection bias but cannot eliminate it entirely and their findings may not be replicated in other institutions by other surgeons,” they note.
Other limitations include that changes in clinical practice and patient selection were likely over the course of the study because of significant changes in American Thyroid Association (ATA) guidelines between 2009 and 2017, and characteristics including molecular genetic testing, which could have influenced final results, were not taken into consideration.
Furthermore, for patients with intermediate-risk cancer, modifications in postoperative follow-up are necessary following lobectomy versus total thyroidectomy; “the role of radioiodine is limited and the levels of thyroglobulin more complicated to interpret,” they note.
The study and editorial authors had no disclosures to report.
A version of this article first appeared on Medscape.com.
Patients with intermediate-risk papillary thyroid cancer and lymph node metastasis show no significant increase in tumor recurrence when undergoing lobectomy compared with a total thyroidectomy, new research shows.
“Results of this cohort study suggest that patients with ipsilateral clinical lateral neck metastasis (cN1b) papillary thyroid cancer who underwent lobectomy exhibited recurrence-free survival rates similar to those who underwent total thyroidectomy after controlling for major prognostic factors,” the authors conclude in the study published online in JAMA Surgery.
“These findings suggest that cN1b alone should not be an absolute indication for total thyroidectomy,” they note.
The study, involving the largest cohort to date to compare patients with intermediate-risk papillary thyroid cancer treated with lobectomy versus total thyroidectomy, “challenged the current guidelines and pushed the boundary of limited surgical treatment even further,” say Michelle B. Mulder, MD, and Quan-Yang Duh, MD, of the department of surgery, University of California, San Francisco, in an accompanying editorial.
“It can be a game changer if confirmed by future prospective and multicenter studies,” they add.
Guidelines still recommend total thyroidectomy with subsequent RAI
While lower-intensity treatment options, with a lower risk of complications, have gained favor in the treatment of low-risk papillary thyroid cancer, guidelines still recommend the consideration of total thyroidectomy and subsequent radioactive iodine ablation (RAI) for intermediate-risk cancers because of the higher chance of recurrence, particularly among those with clinically positive nodes.
However, data on the superiority of a total thyroidectomy, with or without RAI, versus lobectomy is inconsistent, prompting first author Siyuan Xu, MD, of the department of head and neck surgical oncology, National Cancer Center, Beijing, and colleagues to compare the risk of recurrence with the two approaches.
For the study, patients with intermediate-risk papillary thyroid cancer treated at the Chinese Academy of Medical Sciences Cancer Hospital in Beijing between January 2000 and December 2017, who had a lobectomy or total thyroidectomy, were paired 1:1 in a propensity score matching analysis.
Other than treatment type, the 265 pairs of patients were matched based on all other potential prognostic factors, including age, sex, primary tumor size, minor extrathyroidal extension, multifocality, number of lymph node metastases, and lymph node ratio.
Participants were a mean age of 37 years and 66% were female.
With a median follow-up of 60 months in the lobectomy group and 58 months in the total thyroidectomy group, structural recurrences occurred in 7.9% (21) and 6.4% (17) of patients, respectively, which was not significantly different.
The primary endpoint, 5-year rate of recurrence-free survival, was also not significantly different between the lobectomy (92.3%) and total thyroidectomy groups (93.7%) (adjusted hazard ratio, 1.10; P = .77).
In a further stratified analysis of patients treated with total thyroidectomy along with RAI (n = 75), the lack of a significant difference in recurrence-free survival versus lobectomy remained (aHR, 0.59; P = .46).
The results were similar in unadjusted as well as adjusted analyses, and a power analysis indicated that the study had a 90% power to detect a more than 4.9% difference in recurrence-free survival.
“Given the lower complication rate of lobectomy, a maximal 4.9% recurrence-free survival difference is acceptable, which enhances the reliability of the study results,” the authors say.
They conclude that “our findings call into question whether cN1b alone [ipsilateral clinical lateral neck metastasis papillary thyroid cancer] should be an absolute determinant for deciding the optimal extent of thyroid surgery for papillary thyroid cancer.”
With total thyroidectomy, RAI can be given
An important argument in favor of total thyroidectomy is that with the complete resection of thyroid tissue, RAI ablation can then be used for postoperative detection of residual or metastatic disease, as well as for treatment, the authors note.
Indeed, a study using the Surveillance, Epidemiology, and End Results (SEER) database showed RAI ablation is associated with a 29% reduction in the risk of death in patients with intermediate-risk papillary thyroid cancer, with a hazard risk of 0.71.
However, conflicting data from Memorial Sloan-Kettering Cancer Center, New York, suggests no significant benefit with total thyroidectomy and RAI ablation.
The current study’s analysis of patients treated with RAI, though limited in size, supports the latter study’s findings, the authors note.
“When we performed further stratified analyses in patients treated with total thyroidectomy plus RAI ablation and their counterparts, no significant difference was found, which conformed with [the] result from the whole cohort.”
“Certainly, the stratified comparison did not have enough power to examine the effect of RAI ablation on tumor recurrence subject to the limitation of sample size and case selection [and] further study is needed on this topic,” they write.
Some limitations warrant cautious interpretation
In their editorial, Dr. Mulder and Dr. Duh note that while some previous studies have shown similar outcomes relating to tumor size, thyroid hormone suppression therapy, and multifocality, “few have addressed lateral neck involvement.”
They suggest cautious interpretation, however, due to limitations, acknowledged by the authors, including the single-center nature of the study.
“Appropriate propensity matching may mitigate selection bias but cannot eliminate it entirely and their findings may not be replicated in other institutions by other surgeons,” they note.
Other limitations include that changes in clinical practice and patient selection were likely over the course of the study because of significant changes in American Thyroid Association (ATA) guidelines between 2009 and 2017, and characteristics including molecular genetic testing, which could have influenced final results, were not taken into consideration.
Furthermore, for patients with intermediate-risk cancer, modifications in postoperative follow-up are necessary following lobectomy versus total thyroidectomy; “the role of radioiodine is limited and the levels of thyroglobulin more complicated to interpret,” they note.
The study and editorial authors had no disclosures to report.
A version of this article first appeared on Medscape.com.
FROM JAMA SURGERY
Consider quality of life, comorbidities in hidradenitis suppurativa
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – , Robert G. Micheletti, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
For patients with HS, “the quality-of-life impact is profound, greater than any other systematically studied dermatologic condition,” said Dr. Micheletti, associate professor of dermatology at the Hospital of the University of Pennsylavnia, and chief of hospital dermatology, and chief of dermatology at Pennsylvania Hospital, Philadelphia.
Two key aspects of quality of life that affect HS patients are sexual health and overall pain, he said. The female-to-male ratio of HS is approximately 3:1, and data show that approximately 40% of female HS patients experience fertility issues and have unaddressed questions about HS and pregnancy, said Dr. Micheletti. Additionally, data from a systematic review showed that 50%-60% of patients with HS reported sexual dysfunction. Impaired sexual function is also associated with both overall impaired quality of life ratings and the presence of mood disorders, he noted.
Pain also has a significant impact on quality of life for HS patients. When these patients present in an emergency department, 70% report severe pain, and approximately 60% receive opioids, said Dr. Micheletti.
Data from a 2021 study showed that HS patients are significantly more likely to receive opioids compared with controls, and also more likely to be diagnosed with opioid use disorder than controls, especially if they are seen by nondermatologists, he noted.
For acute pain, Dr. Micheletti recommended starting with acetaminophen 500 mg every 4 to 6 hours as needed, and topical nonsteroidal anti-inflammatory drugs (NSAIDs). “It still makes sense to do topical care,” said Dr. Micheletti, but he added that he also prescribes medications for anxiety for these patients.
Patients with increased pain severity or refractory disease may benefit from systemic NSAIDs, or intralesional triamcinolone, he noted. Incision and draining of abscesses may provide temporary symptomatic relief, but keep in mind that lesions will recur, he noted.
For the most severe cases, Dr. Micheletti advised adding tramadol as a first-line opioid, or another short-acting opioid for breakthrough pain.
To manage patients with HS who have chronic pain, Dr. Micheletti recommended starting with HS disease–directed therapy, but also screening for pain severity and psychological comorbidities.
His strategies in these cases include nonpharmacological pain management in the form of physical therapy, wound care, and behavioral health. His algorithm for nociceptive pain is NSAIDs with or without acetaminophen; duloxetine or nortriptyline are other options. For neuropathic pain, gabapentin and/or duloxetine are top choices, but pregabalin, venlafaxine, and nortriptyline are on the list as well.
Topical NSAIDs or topical lidocaine may serve as add-ons to systemic therapy in more severe cases, or as first-line therapy for milder chronic pain, Dr. Micheletti noted. Patients who have failed treatment with at least two pharmacologic agents, suffer medically refractory HS with debilitating pain, or use opioids on an ongoing basis should be referred to a pain management specialist, he said.
Don’t forget lifestyle
Although data on the impact of diet on patients with HS are limited, “we know anecdotally that dairy and refined carbohydrates are associated with exacerbations,” said Dr. Micheletti.
In addition, many patients use complementary medicine “and they aren’t always telling us,” he emphasized. Smoking is prevalent among patients with HS, and is a risk factor for the disease in general, and for more severe and refractory disease, he added. Consequently, screening for tobacco smoking is recommended for patients with HS not only because of the impact on disease, but because it is a potentially modifiable cardiovascular risk factor, he explained.
Consider comorbidities
Cardiovascular disease is among several comorbidities associated with HS, said Dr. Micheletti. HS foundations in the United States and Canada recently published evidence-based recommendations for comorbidity screening. The recommendations included screening for 19 specific comorbidities: acne, dissecting cellulitis, pilonidal disease, pyoderma gangrenosum, depression, anxiety, suicide, smoking, substance abuse, polycystic ovary syndrome, obesity, dyslipidemia, diabetes mellitus, metabolic syndrome, hypertension, cardiovascular disease, inflammatory bowel disease, spondyloarthritis, and sexual dysfunction.
Dr. Micheletti highlighted cardiovascular comorbidities, and noted the association between HS and modifiable cardiovascular risk factors: smoking, obesity, diabetes mellitus, and dyslipidemia. “HS is also independently associated with cardiovascular disease leading to myocardial infarction, stroke, cardiovascular-associated death, and all-cause mortality compared to controls,” he said. Studies show an incidence rate ratio of 1.53 for major adverse cardiovascular events in patients with HS compared with controls, with the highest relative risk among those aged 18-29 years, he added.
Medical management
Depending on the patient, medical management of HS may involve antibiotics, hormonal agents, and biologics, said Dr. Micheletti. Some of the most commonly used antibiotic regimens for HS are those recommended in treatment guidelines, including doxycycline and a clindamycin/rifampin combination, he said. However, the use of trimethoprim-sulfamethoxazole or ciprofloxacin has been associated with increased antibiotic resistance and is not supported by available evidence, he noted.
Hormonal therapies may help some women with HS, said Dr. Micheletti. Options include spironolactone, metformin, or estrogen-containing hormonal contraceptives, he said.
When it comes to biologics, only 33% of HS patients meet criteria for their use (Hurley stage II or III, moderate or severe HS), he noted. However, research suggests “a huge gap” in the use of anti-TNF therapy even among patients for whom it is recommended, he said.
Of the TNF-alpha inhibitors, data on adalimumab, which is FDA-approved for HS, are the most recent. Adalimumab “is our gold standard biologic and our gateway biologic, for HS at this time,” Dr. Micheletti said.
However, those who respond to adalimumab “can continue to do better, but they can wax and wane and flare,” he cautioned. Infliximab, while not approved for HS, has been studied in patients with HS and is prescribed by some providers. Although no comparative studies have been done for infliximab versus adalimumab, “anecdotally, response to infliximab tends to be better, and it is the most effective biologic in common use for severe HS,” he noted.
Dr. Micheletti’s top treatment recommendations for using biologics start with considering biosimilars. Most patients on biosimilars do fine, but some patients who previously responded to infliximab will unpredictably lose efficacy or have reactions when switched to a biosimilar, he said.
Patients on biologics also may experience waning efficacy in the wake of an immune response stimulated by foreign antibodies, said Dr. Micheletti. “Anti-drug antibody formation is more likely to occur when treatment is interrupted,” he noted. Minimize the risk of antibody formation by paying attention to adherence issues and dosing frequency, he advised.
If patients fail both adalimumab and infliximab, Dr. Micheletti tells them not to lose hope, and that treatment is a trial-and-error process that may involve more than one therapy. Other biologics in active use for HS include ustekinumab, anakinra, secukinumab, brodalumab, golimumab, and JAK inhibitors, any of which might be effective in any given patient, he said.
Surgical solutions
For HS patients with chronic, recurring inflammation and drainage associated with a sinus tract, surgical deroofing may the best treatment option, Dr. Micheletti said. “Deroofing involves the use of a probe to trace the extent of the subcutaneous tract, followed by incision and removal of the tract ‘roof,’ ’’ he explained. The deroofing procedure involves local anesthesia and has a low morbidity rate, as well as a low recurrence rate and high levels of patient satisfaction, he said.
“The acute role for surgery is to remove active foci of inflammation and relieve pain,” which is achieved more effectively with deroofing, said Dr. Micheletti. By contrast, incision and drainage is associated with an almost 100% recurrence rate, he added.
When planning elective surgery for HS, Dr. Micheletti noted that holding infliximab for less than 4 weeks does not affect postoperative infection rates in patients with rheumatoid arthritis, and a recent randomized, controlled trial showed that adalimumab can be continued safely through HS surgeries.
In fact, “continuing TNF inhibitors through elective surgery does not increase infection risk and results in better disease control,” and dermatologists should work with surgery to balance infection and disease flare concerns in HS patients, he said.
Dr. Micheletti disclosed serving as a consultant or advisor for Adaptimmune and Vertex, and research funding from Amgen and Cabaletta Bio. MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Both potatoes and beans reduced insulin resistance, weight in controlled study
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
FROM THE JOURNAL OF MEDICINAL FOOD