User login
Cisplatin Ototoxicity: Evidence for Dosing Effect on the Risk of Hearing Shifts Among Head and Neck Cancer Patients Receiving Chemoradiation
Background: Between 2008 and 2014, 17,173 Veterans were treated with cisplatin-based chemotherapy. Many began treatment with pre-existing hearing loss and up to half likely sustained ototoxicity. Minor shifts in hearing, left untreated, can constrain effective provider-veteran
treatment partnerships, family and workplace communication, and limit quality of life. A better understanding of the risk factors and clinical presentation of ototoxicity is needed to inform ototoxicity monitoring programs and treatment decisions.
Methods: Data were examined in N = 21 head and neck cancer patients receiving concurrent chemoradiation therapy with cisplatin for whom audiometry data had been obtained prior to treatment and at 1 or more time points (35 days and 165 days) following the initial treatment.
Data were gathered as part of a larger prospective study on ototoxicity monitoring at the VA Portland HCS from 2014 to 2017. The primary outcome was a shift in the audiogram based on the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) Grades 1 or greater, which was evaluated in relation to cisplatin dosing (> 75 mg/m2 every 3 weeks (bolus) vs weekly dosing < 40 mg/m2). Effects of dosing on the risk of CTCAE grade 1 or greater ototoxic event were estimated using Bayesian analysis. Descriptive statistics characterize additional
factors (age, pre-existing hearing loss and ototoxic hearing shifts, cisplatin cumulative dose, radiation dose).
Results: Ototoxicity meeting CTCAE grade 1 was found at a rate of 23.8%. The estimated risk of ototoxicity did not vary by hearing monitoring method. Veterans receiving bolus-dose cisplatin chemoradiation are estimated to be at double the risk of ototoxicity than those receiving lower weekly dosing.
Implications: Evidence supports the view that CTCAE grade 1 or higher ototoxicity was associated with cisplatin dosing in this sample. High-dose regimens are more likely to cause ototoxicity and this increased risk does not appear to vary based on method of delivery of ototoxicity monitoring. Effects of cisplatin cumulative dose and radiation dose, as well as implications for ototoxicity monitoring, will be discussed.
Background: Between 2008 and 2014, 17,173 Veterans were treated with cisplatin-based chemotherapy. Many began treatment with pre-existing hearing loss and up to half likely sustained ototoxicity. Minor shifts in hearing, left untreated, can constrain effective provider-veteran
treatment partnerships, family and workplace communication, and limit quality of life. A better understanding of the risk factors and clinical presentation of ototoxicity is needed to inform ototoxicity monitoring programs and treatment decisions.
Methods: Data were examined in N = 21 head and neck cancer patients receiving concurrent chemoradiation therapy with cisplatin for whom audiometry data had been obtained prior to treatment and at 1 or more time points (35 days and 165 days) following the initial treatment.
Data were gathered as part of a larger prospective study on ototoxicity monitoring at the VA Portland HCS from 2014 to 2017. The primary outcome was a shift in the audiogram based on the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) Grades 1 or greater, which was evaluated in relation to cisplatin dosing (> 75 mg/m2 every 3 weeks (bolus) vs weekly dosing < 40 mg/m2). Effects of dosing on the risk of CTCAE grade 1 or greater ototoxic event were estimated using Bayesian analysis. Descriptive statistics characterize additional
factors (age, pre-existing hearing loss and ototoxic hearing shifts, cisplatin cumulative dose, radiation dose).
Results: Ototoxicity meeting CTCAE grade 1 was found at a rate of 23.8%. The estimated risk of ototoxicity did not vary by hearing monitoring method. Veterans receiving bolus-dose cisplatin chemoradiation are estimated to be at double the risk of ototoxicity than those receiving lower weekly dosing.
Implications: Evidence supports the view that CTCAE grade 1 or higher ototoxicity was associated with cisplatin dosing in this sample. High-dose regimens are more likely to cause ototoxicity and this increased risk does not appear to vary based on method of delivery of ototoxicity monitoring. Effects of cisplatin cumulative dose and radiation dose, as well as implications for ototoxicity monitoring, will be discussed.
Background: Between 2008 and 2014, 17,173 Veterans were treated with cisplatin-based chemotherapy. Many began treatment with pre-existing hearing loss and up to half likely sustained ototoxicity. Minor shifts in hearing, left untreated, can constrain effective provider-veteran
treatment partnerships, family and workplace communication, and limit quality of life. A better understanding of the risk factors and clinical presentation of ototoxicity is needed to inform ototoxicity monitoring programs and treatment decisions.
Methods: Data were examined in N = 21 head and neck cancer patients receiving concurrent chemoradiation therapy with cisplatin for whom audiometry data had been obtained prior to treatment and at 1 or more time points (35 days and 165 days) following the initial treatment.
Data were gathered as part of a larger prospective study on ototoxicity monitoring at the VA Portland HCS from 2014 to 2017. The primary outcome was a shift in the audiogram based on the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) Grades 1 or greater, which was evaluated in relation to cisplatin dosing (> 75 mg/m2 every 3 weeks (bolus) vs weekly dosing < 40 mg/m2). Effects of dosing on the risk of CTCAE grade 1 or greater ototoxic event were estimated using Bayesian analysis. Descriptive statistics characterize additional
factors (age, pre-existing hearing loss and ototoxic hearing shifts, cisplatin cumulative dose, radiation dose).
Results: Ototoxicity meeting CTCAE grade 1 was found at a rate of 23.8%. The estimated risk of ototoxicity did not vary by hearing monitoring method. Veterans receiving bolus-dose cisplatin chemoradiation are estimated to be at double the risk of ototoxicity than those receiving lower weekly dosing.
Implications: Evidence supports the view that CTCAE grade 1 or higher ototoxicity was associated with cisplatin dosing in this sample. High-dose regimens are more likely to cause ototoxicity and this increased risk does not appear to vary based on method of delivery of ototoxicity monitoring. Effects of cisplatin cumulative dose and radiation dose, as well as implications for ototoxicity monitoring, will be discussed.
Combining Chemotherapy and Trastuzimab in Patients With HER2/ Neu-Positive Metastatic Salivary Gland Carcinoma
Background: Salivary gland carcinoma is a rare type of cancer, and when metastasized is associated with poor prognosis. Response rates to standard chemotherapy are less (15-30%) and usually short-lived. Salivary gland tumors expressing HER2/neu have lately been recognized. We describe 2 cases of ERBB2 amplification identified by NGS testing with response to Anti Her-2 therapy.
Case 1: A 69-year-old male veteran was originally diagnosed with a left parotid gland carcinoma at pleomorphic adenoma in September 2015. Patient underwent surgery with a left parotidectomy 4 out of 85 lymph nodes were positive for metastasis (T3, N2b, Mx, G3, Stage IVA disease). Patient was offered adjuvant chemo/XRT but was lost to follow-up. In October 2016, patient presented with recurrent left parotid gland swelling staging workup revealed diffuse metastatic disease to liver and bone. Patient had a palliative resection of the left parotid mass tumor was consistent with carcinoma and was offered palliative chemotherapy with carbo platinum and Taxol. After 4 cycles of chemotherapy repeat PET CT scan revealed progression of disease with new sternal, T3 vertebral, right sacral and numerous liver and upper abdominal lymph nodes. Next generation sequencing (NGS) by Foundation One revealed ERBB2/HER2 mutation. Patient was started on second-line palliative chemotherapy utilizing docetaxel and Herceptin every 3 weeks. After 3 cycles restaging PET CT scan showed good partial response in bone and stable disease in the liver.
Case 2: A 73-year-old male presented on 4/16 with rapidly enlarging recurrent mass and bone pain one year after right radical neck dissection followed by adjuvant XRT. Pathology demonstrated salivary gland adenocarcinoma. PET/CT: extensive axial and appendicular skeletal metastasis with widespread bony pain. NGS testing of the original pathology with Foundation One revealed ERBB2 amplification as well as alteration of PTEN and TP53. Patient was offered Carboplatin + Herceptin which he received and tolerated well. Pain resolved after 1st cycle. Metabolic PR by PET/CT was noted after 4 cycles.
Conclusions: Adding trastuzumab to chemotherapy in patients with Her2/neu-positive metastatic salivary gland carcinoma gave promising results. Our institutional experience matches with few other case reports/series published to date. Validating this results with a randomized study would be a challenge, given the rarity of this disease.
Background: Salivary gland carcinoma is a rare type of cancer, and when metastasized is associated with poor prognosis. Response rates to standard chemotherapy are less (15-30%) and usually short-lived. Salivary gland tumors expressing HER2/neu have lately been recognized. We describe 2 cases of ERBB2 amplification identified by NGS testing with response to Anti Her-2 therapy.
Case 1: A 69-year-old male veteran was originally diagnosed with a left parotid gland carcinoma at pleomorphic adenoma in September 2015. Patient underwent surgery with a left parotidectomy 4 out of 85 lymph nodes were positive for metastasis (T3, N2b, Mx, G3, Stage IVA disease). Patient was offered adjuvant chemo/XRT but was lost to follow-up. In October 2016, patient presented with recurrent left parotid gland swelling staging workup revealed diffuse metastatic disease to liver and bone. Patient had a palliative resection of the left parotid mass tumor was consistent with carcinoma and was offered palliative chemotherapy with carbo platinum and Taxol. After 4 cycles of chemotherapy repeat PET CT scan revealed progression of disease with new sternal, T3 vertebral, right sacral and numerous liver and upper abdominal lymph nodes. Next generation sequencing (NGS) by Foundation One revealed ERBB2/HER2 mutation. Patient was started on second-line palliative chemotherapy utilizing docetaxel and Herceptin every 3 weeks. After 3 cycles restaging PET CT scan showed good partial response in bone and stable disease in the liver.
Case 2: A 73-year-old male presented on 4/16 with rapidly enlarging recurrent mass and bone pain one year after right radical neck dissection followed by adjuvant XRT. Pathology demonstrated salivary gland adenocarcinoma. PET/CT: extensive axial and appendicular skeletal metastasis with widespread bony pain. NGS testing of the original pathology with Foundation One revealed ERBB2 amplification as well as alteration of PTEN and TP53. Patient was offered Carboplatin + Herceptin which he received and tolerated well. Pain resolved after 1st cycle. Metabolic PR by PET/CT was noted after 4 cycles.
Conclusions: Adding trastuzumab to chemotherapy in patients with Her2/neu-positive metastatic salivary gland carcinoma gave promising results. Our institutional experience matches with few other case reports/series published to date. Validating this results with a randomized study would be a challenge, given the rarity of this disease.
Background: Salivary gland carcinoma is a rare type of cancer, and when metastasized is associated with poor prognosis. Response rates to standard chemotherapy are less (15-30%) and usually short-lived. Salivary gland tumors expressing HER2/neu have lately been recognized. We describe 2 cases of ERBB2 amplification identified by NGS testing with response to Anti Her-2 therapy.
Case 1: A 69-year-old male veteran was originally diagnosed with a left parotid gland carcinoma at pleomorphic adenoma in September 2015. Patient underwent surgery with a left parotidectomy 4 out of 85 lymph nodes were positive for metastasis (T3, N2b, Mx, G3, Stage IVA disease). Patient was offered adjuvant chemo/XRT but was lost to follow-up. In October 2016, patient presented with recurrent left parotid gland swelling staging workup revealed diffuse metastatic disease to liver and bone. Patient had a palliative resection of the left parotid mass tumor was consistent with carcinoma and was offered palliative chemotherapy with carbo platinum and Taxol. After 4 cycles of chemotherapy repeat PET CT scan revealed progression of disease with new sternal, T3 vertebral, right sacral and numerous liver and upper abdominal lymph nodes. Next generation sequencing (NGS) by Foundation One revealed ERBB2/HER2 mutation. Patient was started on second-line palliative chemotherapy utilizing docetaxel and Herceptin every 3 weeks. After 3 cycles restaging PET CT scan showed good partial response in bone and stable disease in the liver.
Case 2: A 73-year-old male presented on 4/16 with rapidly enlarging recurrent mass and bone pain one year after right radical neck dissection followed by adjuvant XRT. Pathology demonstrated salivary gland adenocarcinoma. PET/CT: extensive axial and appendicular skeletal metastasis with widespread bony pain. NGS testing of the original pathology with Foundation One revealed ERBB2 amplification as well as alteration of PTEN and TP53. Patient was offered Carboplatin + Herceptin which he received and tolerated well. Pain resolved after 1st cycle. Metabolic PR by PET/CT was noted after 4 cycles.
Conclusions: Adding trastuzumab to chemotherapy in patients with Her2/neu-positive metastatic salivary gland carcinoma gave promising results. Our institutional experience matches with few other case reports/series published to date. Validating this results with a randomized study would be a challenge, given the rarity of this disease.
Overview of Oral Oncolytic Processes at Iowa City VAHCS Oncology Clinic
Background: The availability and use of oral oncolytic agents to treat malignancy is increasing. The use of oral agents to treat cancer introduces new complexities with regard to patient compliance, toxicity management, and cost containment. Veterans Affairs (VA) oncology clinics are uniquely equipped to manage oral chemotherapy processes because of direct access to health care records, pharmacy dispensing information, and oncology providers. A recent evaluation of a 14-day oral chemotherapy dispensing protocol at 1 VA facility demonstrated that patients had increased medication possession ratios and decreased cost associated with their care when compared to standard 30-day dispensing practices.
Methods: The Iowa City VA Health Care System Oncology Clinic (IC VAHCS) has developed a process to aid in tracking dispensed prescriptions, scheduling followup telephone calls, evaluating laboratory monitoring, and identification of future oncology appointments. A pharmacist has developed the ability to extract administrative from the VA Corporate Data Warehouse (CDW) for clinic operational use to support local processes. The management of oral oncolytic therapy is spearheaded by the local oncology clinical pharmacy specialist, in collaboration with oncologists and oncology nurse case managers.
The current practice at the IC VAHCS will be detailed to aid other VA oncology clinics in developing similar processes at their local site.
Background: The availability and use of oral oncolytic agents to treat malignancy is increasing. The use of oral agents to treat cancer introduces new complexities with regard to patient compliance, toxicity management, and cost containment. Veterans Affairs (VA) oncology clinics are uniquely equipped to manage oral chemotherapy processes because of direct access to health care records, pharmacy dispensing information, and oncology providers. A recent evaluation of a 14-day oral chemotherapy dispensing protocol at 1 VA facility demonstrated that patients had increased medication possession ratios and decreased cost associated with their care when compared to standard 30-day dispensing practices.
Methods: The Iowa City VA Health Care System Oncology Clinic (IC VAHCS) has developed a process to aid in tracking dispensed prescriptions, scheduling followup telephone calls, evaluating laboratory monitoring, and identification of future oncology appointments. A pharmacist has developed the ability to extract administrative from the VA Corporate Data Warehouse (CDW) for clinic operational use to support local processes. The management of oral oncolytic therapy is spearheaded by the local oncology clinical pharmacy specialist, in collaboration with oncologists and oncology nurse case managers.
The current practice at the IC VAHCS will be detailed to aid other VA oncology clinics in developing similar processes at their local site.
Background: The availability and use of oral oncolytic agents to treat malignancy is increasing. The use of oral agents to treat cancer introduces new complexities with regard to patient compliance, toxicity management, and cost containment. Veterans Affairs (VA) oncology clinics are uniquely equipped to manage oral chemotherapy processes because of direct access to health care records, pharmacy dispensing information, and oncology providers. A recent evaluation of a 14-day oral chemotherapy dispensing protocol at 1 VA facility demonstrated that patients had increased medication possession ratios and decreased cost associated with their care when compared to standard 30-day dispensing practices.
Methods: The Iowa City VA Health Care System Oncology Clinic (IC VAHCS) has developed a process to aid in tracking dispensed prescriptions, scheduling followup telephone calls, evaluating laboratory monitoring, and identification of future oncology appointments. A pharmacist has developed the ability to extract administrative from the VA Corporate Data Warehouse (CDW) for clinic operational use to support local processes. The management of oral oncolytic therapy is spearheaded by the local oncology clinical pharmacy specialist, in collaboration with oncologists and oncology nurse case managers.
The current practice at the IC VAHCS will be detailed to aid other VA oncology clinics in developing similar processes at their local site.
Is Hypomagnesemia a Marker of Efficacy of Cetuximab in Locoregionally Advanced and Metastatic Head and Neck Cancer?
Background: Current NCCN guidelines recommend the use of cetuximab, an EGFR monoclonal antibody, in the treatment of head and neck (H&N) cancers in combination with radiation therapy as initial treatment of locally or regionally advanced disease in patients, who are ineligible
for platinum-based therapy. It is also the standard of care in the treatment of recurrent or persistent disease with distant metastases.
Objective: Hypomagnesemia is a common side effect of cetuximab. Previous studies demonstrated that magnesium reduction was a potential marker of efficacy and outcome in the treatment of advanced colorectal cancer. We hypothesize that hypomagnesemia is also a marker of efficacy of the anti-neoplastic treatment of H&N cancer.
Methods: We retrospectively reviewed the medical records of H&N cancer patients that were treated with cetuximab between January 1, 2006 and January 1, 2016 at the Stratton VA Medical Center. Included in the study were patients aged over 20 years with stage III or IV H&N cancer who received cetuximab. Exclusion criteria included prior magnesium supplementation, history of treatment with anti-EGFR therapy, malabsorption syndromes and genetic magnesium wasting syndrome.
Results: Of the 63 patients studied, 23 developed hypomagnesemia for an overall incidence of 36.5%. The median age of diagnosis was 65 years for the hypomagnesemia group and 66 years for the nonhypomagnesemia. The patients that developed hypomagnesemia had a median survival of 27 months (95% CI, 16.3-37.6) while those that maintained normal magnesium levels had a mean survival of 20 months (95% CI, 12.3-27.7) (P = .583).
Conclusions: To our knowledge, no study has examined the predictive value of hypomagnesemia for the overall survival of H&N cancer patients treated with cetuximab that develop hypomagnesemia vs those that don’t. While data from the colorectal cancer suggest that hypomagnesemia may be used as a surrogate of efficacy for cetuximab, our data negates such correlation. Further study is required to elicit the link between cetuximab and hypomagnesemia.
Background: Current NCCN guidelines recommend the use of cetuximab, an EGFR monoclonal antibody, in the treatment of head and neck (H&N) cancers in combination with radiation therapy as initial treatment of locally or regionally advanced disease in patients, who are ineligible
for platinum-based therapy. It is also the standard of care in the treatment of recurrent or persistent disease with distant metastases.
Objective: Hypomagnesemia is a common side effect of cetuximab. Previous studies demonstrated that magnesium reduction was a potential marker of efficacy and outcome in the treatment of advanced colorectal cancer. We hypothesize that hypomagnesemia is also a marker of efficacy of the anti-neoplastic treatment of H&N cancer.
Methods: We retrospectively reviewed the medical records of H&N cancer patients that were treated with cetuximab between January 1, 2006 and January 1, 2016 at the Stratton VA Medical Center. Included in the study were patients aged over 20 years with stage III or IV H&N cancer who received cetuximab. Exclusion criteria included prior magnesium supplementation, history of treatment with anti-EGFR therapy, malabsorption syndromes and genetic magnesium wasting syndrome.
Results: Of the 63 patients studied, 23 developed hypomagnesemia for an overall incidence of 36.5%. The median age of diagnosis was 65 years for the hypomagnesemia group and 66 years for the nonhypomagnesemia. The patients that developed hypomagnesemia had a median survival of 27 months (95% CI, 16.3-37.6) while those that maintained normal magnesium levels had a mean survival of 20 months (95% CI, 12.3-27.7) (P = .583).
Conclusions: To our knowledge, no study has examined the predictive value of hypomagnesemia for the overall survival of H&N cancer patients treated with cetuximab that develop hypomagnesemia vs those that don’t. While data from the colorectal cancer suggest that hypomagnesemia may be used as a surrogate of efficacy for cetuximab, our data negates such correlation. Further study is required to elicit the link between cetuximab and hypomagnesemia.
Background: Current NCCN guidelines recommend the use of cetuximab, an EGFR monoclonal antibody, in the treatment of head and neck (H&N) cancers in combination with radiation therapy as initial treatment of locally or regionally advanced disease in patients, who are ineligible
for platinum-based therapy. It is also the standard of care in the treatment of recurrent or persistent disease with distant metastases.
Objective: Hypomagnesemia is a common side effect of cetuximab. Previous studies demonstrated that magnesium reduction was a potential marker of efficacy and outcome in the treatment of advanced colorectal cancer. We hypothesize that hypomagnesemia is also a marker of efficacy of the anti-neoplastic treatment of H&N cancer.
Methods: We retrospectively reviewed the medical records of H&N cancer patients that were treated with cetuximab between January 1, 2006 and January 1, 2016 at the Stratton VA Medical Center. Included in the study were patients aged over 20 years with stage III or IV H&N cancer who received cetuximab. Exclusion criteria included prior magnesium supplementation, history of treatment with anti-EGFR therapy, malabsorption syndromes and genetic magnesium wasting syndrome.
Results: Of the 63 patients studied, 23 developed hypomagnesemia for an overall incidence of 36.5%. The median age of diagnosis was 65 years for the hypomagnesemia group and 66 years for the nonhypomagnesemia. The patients that developed hypomagnesemia had a median survival of 27 months (95% CI, 16.3-37.6) while those that maintained normal magnesium levels had a mean survival of 20 months (95% CI, 12.3-27.7) (P = .583).
Conclusions: To our knowledge, no study has examined the predictive value of hypomagnesemia for the overall survival of H&N cancer patients treated with cetuximab that develop hypomagnesemia vs those that don’t. While data from the colorectal cancer suggest that hypomagnesemia may be used as a surrogate of efficacy for cetuximab, our data negates such correlation. Further study is required to elicit the link between cetuximab and hypomagnesemia.
Two Somatic P53 Mutations, Two Histological Tumors From Single Founder Clone: A Conglomerate of Pituitary Corticotropinoma and Rhabdomyosarcoma
Background: Typically arising from skeletal muscle, rhabdomyosarcoma (RMS) rarely may be found in the head and neck. Here, we report a rare occurrence of fatal, aggressive RMS in association with pituitary corticotropinoma.
Case: A 52-year-old male veteran presented with 1-year history of hypertension, diabetes mellitus, vertebral fractures, central obesity, muscle wasting, and multiple pulmonary embolisms. Based on the clinical and biochemical findings, he was diagnosed with Cushing’s disease. Pituitary MRI demonstrated a hypoenhancing heterogeneous 16x15x12 mm sellar mass which was resected by trans-sphenoidal surgery (TSS). During TSS, 2 distinct tumor masses were identified—a firm sphenoidal mass with dural/bony invasion, and a soft adenomatous intrasellar mass contiguous through a small opening in the sellar bone.
Histopathologic analysis of the intrasellar component confirmed a pituitary corticotropinoma with strong ACTH staining and MIB-1 proliferative index (MIB-1) < 1%. By contrast, the tumor in the sphenoid sinus was a high grade, pleiomorphic RMS consisting of a malignant spindle cell neoplasm with occasional rhabdoid phenotype, brisk mitotic activity, high MIB index and areas of necrosis with strong positivity for desmin and MYF4. Molecular genetic profiling showed that corticotropinoma and RMS elements derived from the same cytogenetically abnormal founder clone, yet subsequently diverged as evidenced by distinct somatic p53 mutations in the corticotropinoma (c.764T>A, c.757A>C) and RMS sub clones (c.736A>G).
Pan CT, MRI brain and bone scan were negative for other primary/metastatic foci. A 18F-FDG PET study detected increased metabolic activity within the sphenoid mass. He was started on chemotherapy with etoposide, vincristine and cyclophosphamide, but the disease relentlessly
progressed with an extension to bilateral optic nerves, internal carotid arteries, ethmoid sinus and nasal cavity. The patient felt that he cannot fight a “losing battle,” opted for home hospice and eventually died in 2 months.
Conclusions: In nontextbook appearance of tumors, a high index of suspicion for unusual tumors like rhabdomyosarcoma arising within sella/sphenoid sinus, is needed for an early intervention. Analysis of genomic data in these tumors may suggest treatment matched to specific tumor mutations.
Background: Typically arising from skeletal muscle, rhabdomyosarcoma (RMS) rarely may be found in the head and neck. Here, we report a rare occurrence of fatal, aggressive RMS in association with pituitary corticotropinoma.
Case: A 52-year-old male veteran presented with 1-year history of hypertension, diabetes mellitus, vertebral fractures, central obesity, muscle wasting, and multiple pulmonary embolisms. Based on the clinical and biochemical findings, he was diagnosed with Cushing’s disease. Pituitary MRI demonstrated a hypoenhancing heterogeneous 16x15x12 mm sellar mass which was resected by trans-sphenoidal surgery (TSS). During TSS, 2 distinct tumor masses were identified—a firm sphenoidal mass with dural/bony invasion, and a soft adenomatous intrasellar mass contiguous through a small opening in the sellar bone.
Histopathologic analysis of the intrasellar component confirmed a pituitary corticotropinoma with strong ACTH staining and MIB-1 proliferative index (MIB-1) < 1%. By contrast, the tumor in the sphenoid sinus was a high grade, pleiomorphic RMS consisting of a malignant spindle cell neoplasm with occasional rhabdoid phenotype, brisk mitotic activity, high MIB index and areas of necrosis with strong positivity for desmin and MYF4. Molecular genetic profiling showed that corticotropinoma and RMS elements derived from the same cytogenetically abnormal founder clone, yet subsequently diverged as evidenced by distinct somatic p53 mutations in the corticotropinoma (c.764T>A, c.757A>C) and RMS sub clones (c.736A>G).
Pan CT, MRI brain and bone scan were negative for other primary/metastatic foci. A 18F-FDG PET study detected increased metabolic activity within the sphenoid mass. He was started on chemotherapy with etoposide, vincristine and cyclophosphamide, but the disease relentlessly
progressed with an extension to bilateral optic nerves, internal carotid arteries, ethmoid sinus and nasal cavity. The patient felt that he cannot fight a “losing battle,” opted for home hospice and eventually died in 2 months.
Conclusions: In nontextbook appearance of tumors, a high index of suspicion for unusual tumors like rhabdomyosarcoma arising within sella/sphenoid sinus, is needed for an early intervention. Analysis of genomic data in these tumors may suggest treatment matched to specific tumor mutations.
Background: Typically arising from skeletal muscle, rhabdomyosarcoma (RMS) rarely may be found in the head and neck. Here, we report a rare occurrence of fatal, aggressive RMS in association with pituitary corticotropinoma.
Case: A 52-year-old male veteran presented with 1-year history of hypertension, diabetes mellitus, vertebral fractures, central obesity, muscle wasting, and multiple pulmonary embolisms. Based on the clinical and biochemical findings, he was diagnosed with Cushing’s disease. Pituitary MRI demonstrated a hypoenhancing heterogeneous 16x15x12 mm sellar mass which was resected by trans-sphenoidal surgery (TSS). During TSS, 2 distinct tumor masses were identified—a firm sphenoidal mass with dural/bony invasion, and a soft adenomatous intrasellar mass contiguous through a small opening in the sellar bone.
Histopathologic analysis of the intrasellar component confirmed a pituitary corticotropinoma with strong ACTH staining and MIB-1 proliferative index (MIB-1) < 1%. By contrast, the tumor in the sphenoid sinus was a high grade, pleiomorphic RMS consisting of a malignant spindle cell neoplasm with occasional rhabdoid phenotype, brisk mitotic activity, high MIB index and areas of necrosis with strong positivity for desmin and MYF4. Molecular genetic profiling showed that corticotropinoma and RMS elements derived from the same cytogenetically abnormal founder clone, yet subsequently diverged as evidenced by distinct somatic p53 mutations in the corticotropinoma (c.764T>A, c.757A>C) and RMS sub clones (c.736A>G).
Pan CT, MRI brain and bone scan were negative for other primary/metastatic foci. A 18F-FDG PET study detected increased metabolic activity within the sphenoid mass. He was started on chemotherapy with etoposide, vincristine and cyclophosphamide, but the disease relentlessly
progressed with an extension to bilateral optic nerves, internal carotid arteries, ethmoid sinus and nasal cavity. The patient felt that he cannot fight a “losing battle,” opted for home hospice and eventually died in 2 months.
Conclusions: In nontextbook appearance of tumors, a high index of suspicion for unusual tumors like rhabdomyosarcoma arising within sella/sphenoid sinus, is needed for an early intervention. Analysis of genomic data in these tumors may suggest treatment matched to specific tumor mutations.
Thyroid-nodule size boosts serum thyroglobulin’s diagnostic value
BOSTON – Normalizing the serum thyroglobulin level by thyroid nodule size in patients surgically treated for a thyroid nodule produced a strongly significant link between the level of this marker and nodule malignancy in a review of nearly 200 patients treated at any of three Montreal centers.
After normalization, the serum thyroglobulin of patients with a malignant nodule averaged 51 mcg/L*cm, more than double the average 23 mcg/L*cm among patients with benign nodules, Neil Verma, MD, said at the World Congress on Thyroid Cancer.
But the senior investigator on the study said that, even if the MTNS+ gets a little more accurate by using a nodule size-normalized serum thyroglobulin level, the clinical utility of the MTNS+ will soon be completely eclipsed by widespread reliance on molecular tests, whereas the MTNS+ combines many clinical and conventional laboratory measures. It‘s only a matter of cost, said Richard J. Payne, MD, a head and neck surgeon at McGill.
Routine reimbursement for molecular diagnostic tests for the malignancy of thyroid nodules was discussed at a recent meeting of Canadian head and neck surgeons, who decided to lobby provincial governments to try to get it covered, according to Dr. Payne. “I’d be very surprised if we don’t have government coverage within 4-5 years,” in part because the cost for molecular testing will likely fall significantly in that time frame, he predicted.
The analysis reported by Dr. Verma included 196 patients with thyroid nodules who underwent a partial or total thyroidectomy at any of three McGill teaching hospitals during 2010-2015. He determined the benign or malignant status of their nodules based on their histology. The analysis he presented also showed that malignancy had no clear relationship to nodule size. Nodules that were less than 2 cm in diameter were about as likely to be malignant as were those that were 3 cm or larger in diameter, Dr. Verma reported.
Size-normalized serum thyroglobulin will now be incorporated into the MTNS+, which will be the fourth change to the original MTNS scoring system since it was developed more than a decade ago, noted Dr. Payne. But, while the MTNS+ allows better prediction of malignant potential than does the Bethesda system for evaluating nodule cytopathology in a fine-needle aspirate, it still falls short of molecular testing in its predictive accuracy, Dr. Payne said.
Dr. Verma and Dr. Payne had no disclosures.
[email protected]
On Twitter @mitchelzoler
BOSTON – Normalizing the serum thyroglobulin level by thyroid nodule size in patients surgically treated for a thyroid nodule produced a strongly significant link between the level of this marker and nodule malignancy in a review of nearly 200 patients treated at any of three Montreal centers.
After normalization, the serum thyroglobulin of patients with a malignant nodule averaged 51 mcg/L*cm, more than double the average 23 mcg/L*cm among patients with benign nodules, Neil Verma, MD, said at the World Congress on Thyroid Cancer.
But the senior investigator on the study said that, even if the MTNS+ gets a little more accurate by using a nodule size-normalized serum thyroglobulin level, the clinical utility of the MTNS+ will soon be completely eclipsed by widespread reliance on molecular tests, whereas the MTNS+ combines many clinical and conventional laboratory measures. It‘s only a matter of cost, said Richard J. Payne, MD, a head and neck surgeon at McGill.
Routine reimbursement for molecular diagnostic tests for the malignancy of thyroid nodules was discussed at a recent meeting of Canadian head and neck surgeons, who decided to lobby provincial governments to try to get it covered, according to Dr. Payne. “I’d be very surprised if we don’t have government coverage within 4-5 years,” in part because the cost for molecular testing will likely fall significantly in that time frame, he predicted.
The analysis reported by Dr. Verma included 196 patients with thyroid nodules who underwent a partial or total thyroidectomy at any of three McGill teaching hospitals during 2010-2015. He determined the benign or malignant status of their nodules based on their histology. The analysis he presented also showed that malignancy had no clear relationship to nodule size. Nodules that were less than 2 cm in diameter were about as likely to be malignant as were those that were 3 cm or larger in diameter, Dr. Verma reported.
Size-normalized serum thyroglobulin will now be incorporated into the MTNS+, which will be the fourth change to the original MTNS scoring system since it was developed more than a decade ago, noted Dr. Payne. But, while the MTNS+ allows better prediction of malignant potential than does the Bethesda system for evaluating nodule cytopathology in a fine-needle aspirate, it still falls short of molecular testing in its predictive accuracy, Dr. Payne said.
Dr. Verma and Dr. Payne had no disclosures.
[email protected]
On Twitter @mitchelzoler
BOSTON – Normalizing the serum thyroglobulin level by thyroid nodule size in patients surgically treated for a thyroid nodule produced a strongly significant link between the level of this marker and nodule malignancy in a review of nearly 200 patients treated at any of three Montreal centers.
After normalization, the serum thyroglobulin of patients with a malignant nodule averaged 51 mcg/L*cm, more than double the average 23 mcg/L*cm among patients with benign nodules, Neil Verma, MD, said at the World Congress on Thyroid Cancer.
But the senior investigator on the study said that, even if the MTNS+ gets a little more accurate by using a nodule size-normalized serum thyroglobulin level, the clinical utility of the MTNS+ will soon be completely eclipsed by widespread reliance on molecular tests, whereas the MTNS+ combines many clinical and conventional laboratory measures. It‘s only a matter of cost, said Richard J. Payne, MD, a head and neck surgeon at McGill.
Routine reimbursement for molecular diagnostic tests for the malignancy of thyroid nodules was discussed at a recent meeting of Canadian head and neck surgeons, who decided to lobby provincial governments to try to get it covered, according to Dr. Payne. “I’d be very surprised if we don’t have government coverage within 4-5 years,” in part because the cost for molecular testing will likely fall significantly in that time frame, he predicted.
The analysis reported by Dr. Verma included 196 patients with thyroid nodules who underwent a partial or total thyroidectomy at any of three McGill teaching hospitals during 2010-2015. He determined the benign or malignant status of their nodules based on their histology. The analysis he presented also showed that malignancy had no clear relationship to nodule size. Nodules that were less than 2 cm in diameter were about as likely to be malignant as were those that were 3 cm or larger in diameter, Dr. Verma reported.
Size-normalized serum thyroglobulin will now be incorporated into the MTNS+, which will be the fourth change to the original MTNS scoring system since it was developed more than a decade ago, noted Dr. Payne. But, while the MTNS+ allows better prediction of malignant potential than does the Bethesda system for evaluating nodule cytopathology in a fine-needle aspirate, it still falls short of molecular testing in its predictive accuracy, Dr. Payne said.
Dr. Verma and Dr. Payne had no disclosures.
[email protected]
On Twitter @mitchelzoler
AT WCTC 2017
Key clinical point:
Major finding: The average size-normalized serum thyroglobulin level was 51 mcg/L*cm in patients with malignant nodules and 23 mcg/L*cm with benign nodules.
Data source: Review of 196 patients who underwent partial or complete thyroidectomy at any of three Montreal centers.
Disclosures: Dr. Verma and Dr. Payne had no disclosures.
VIDEO: Less follow-up proposed for low-risk thyroid cancer
BOSTON – , Bryan R. Haugen, MD, suggested in a keynote lecture during the World Congress on Thyroid Cancer.
Traditionally, thyroid cancer specialists have monitored these patients for persistent or recurrent disease as often as every 6 or 12 months. “But what we’ve realized with recent assessments of response to treatment is that some patients do well without a recurrence over many years; so, the concept of doing less monitoring and less imaging, especially in patients with an excellent response [to their initial treatment], is being studied,” Dr. Haugen said in a video interview following his talk.
He estimated that perhaps two-thirds or as many as three-quarters of patients with differentiated thyroid cancer fall into the category of having low- or intermediate-risk disease with an excellent or good response to treatment, and hence they are potential candidates for eventually transitioning to less frequent follow-up.
During his talk, Dr. Haugen suggested that after several years with no sign of disease recurrence, lower-risk patients with an excellent treatment response may be able to stop undergoing regular monitoring, and those with a good treatment response may be able to safely have their monitoring intervals extended.
According to the most recent (2015) guidelines for differentiated thyroid cancer management from the American Thyroid Association, lower-risk patients with an excellent treatment response should have their serum thyroglobulin measured every 12-24 months and undergo an ultrasound examination every 3-5 years, while patients with a good response are targeted for serum thyroglobulin measurement annually with an ultrasound every 1-3 years (Thyroid. 2016 Jan;26[1]:1-133). Dr. Haugen chaired the expert panel that wrote these guidelines.
In another provocative suggestion, Dr. Haugen proposed that once well-responsive, lower-risk patients have remained disease free for several years, their less frequent follow-up monitoring could be continued by a primary care physician or another less specialized clinician.
At some time in the future, “a patient’s primary care physician could follow a simple tumor marker, thyroglobulin, maybe once every 5 years,” said Dr. Haugen, professor of medicine and head of the division of endocrinology, metabolism, and diabetes at the University of Colorado in Aurora. “At the University of Colorado, we use advanced-practice providers to do long-term follow-up” for lower-risk, treatment-responsive patients, he said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
BOSTON – , Bryan R. Haugen, MD, suggested in a keynote lecture during the World Congress on Thyroid Cancer.
Traditionally, thyroid cancer specialists have monitored these patients for persistent or recurrent disease as often as every 6 or 12 months. “But what we’ve realized with recent assessments of response to treatment is that some patients do well without a recurrence over many years; so, the concept of doing less monitoring and less imaging, especially in patients with an excellent response [to their initial treatment], is being studied,” Dr. Haugen said in a video interview following his talk.
He estimated that perhaps two-thirds or as many as three-quarters of patients with differentiated thyroid cancer fall into the category of having low- or intermediate-risk disease with an excellent or good response to treatment, and hence they are potential candidates for eventually transitioning to less frequent follow-up.
During his talk, Dr. Haugen suggested that after several years with no sign of disease recurrence, lower-risk patients with an excellent treatment response may be able to stop undergoing regular monitoring, and those with a good treatment response may be able to safely have their monitoring intervals extended.
According to the most recent (2015) guidelines for differentiated thyroid cancer management from the American Thyroid Association, lower-risk patients with an excellent treatment response should have their serum thyroglobulin measured every 12-24 months and undergo an ultrasound examination every 3-5 years, while patients with a good response are targeted for serum thyroglobulin measurement annually with an ultrasound every 1-3 years (Thyroid. 2016 Jan;26[1]:1-133). Dr. Haugen chaired the expert panel that wrote these guidelines.
In another provocative suggestion, Dr. Haugen proposed that once well-responsive, lower-risk patients have remained disease free for several years, their less frequent follow-up monitoring could be continued by a primary care physician or another less specialized clinician.
At some time in the future, “a patient’s primary care physician could follow a simple tumor marker, thyroglobulin, maybe once every 5 years,” said Dr. Haugen, professor of medicine and head of the division of endocrinology, metabolism, and diabetes at the University of Colorado in Aurora. “At the University of Colorado, we use advanced-practice providers to do long-term follow-up” for lower-risk, treatment-responsive patients, he said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
BOSTON – , Bryan R. Haugen, MD, suggested in a keynote lecture during the World Congress on Thyroid Cancer.
Traditionally, thyroid cancer specialists have monitored these patients for persistent or recurrent disease as often as every 6 or 12 months. “But what we’ve realized with recent assessments of response to treatment is that some patients do well without a recurrence over many years; so, the concept of doing less monitoring and less imaging, especially in patients with an excellent response [to their initial treatment], is being studied,” Dr. Haugen said in a video interview following his talk.
He estimated that perhaps two-thirds or as many as three-quarters of patients with differentiated thyroid cancer fall into the category of having low- or intermediate-risk disease with an excellent or good response to treatment, and hence they are potential candidates for eventually transitioning to less frequent follow-up.
During his talk, Dr. Haugen suggested that after several years with no sign of disease recurrence, lower-risk patients with an excellent treatment response may be able to stop undergoing regular monitoring, and those with a good treatment response may be able to safely have their monitoring intervals extended.
According to the most recent (2015) guidelines for differentiated thyroid cancer management from the American Thyroid Association, lower-risk patients with an excellent treatment response should have their serum thyroglobulin measured every 12-24 months and undergo an ultrasound examination every 3-5 years, while patients with a good response are targeted for serum thyroglobulin measurement annually with an ultrasound every 1-3 years (Thyroid. 2016 Jan;26[1]:1-133). Dr. Haugen chaired the expert panel that wrote these guidelines.
In another provocative suggestion, Dr. Haugen proposed that once well-responsive, lower-risk patients have remained disease free for several years, their less frequent follow-up monitoring could be continued by a primary care physician or another less specialized clinician.
At some time in the future, “a patient’s primary care physician could follow a simple tumor marker, thyroglobulin, maybe once every 5 years,” said Dr. Haugen, professor of medicine and head of the division of endocrinology, metabolism, and diabetes at the University of Colorado in Aurora. “At the University of Colorado, we use advanced-practice providers to do long-term follow-up” for lower-risk, treatment-responsive patients, he said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
AT WCTC 2017
Prehabilitation for lymphedema in head and neck cancer patients at a community cancer center
Lymphedema is the swelling of tissue caused by the accumulation of interstitial fluid in any area of the body where lymphatic flow has been compromised.1 Secondary lymphedema is an acquired abnormality in lymph drainage1,2 and is the type commonly seen in cancer patients. Secondary lymphedema can be described as external or internal. Internal lymphedema, swelling of deep structures and tissues, is very difficult to quantify.
Lymphedema in patients with head and neck cancers
Lymphedema is a complicating morbidity frequently seen in head and neck cancer patients who have undergone treatment with surgery, radiation, and chemotherapy. However, although it is one of the most prevalent side effects of treatment, it is both under-recognized and under-treated.3
In head and neck cancer patients, internal swelling may develop in the soft tissues of the upper aero-digestive tract,4 affecting articulation and swallowing. Currently, there does not seem to be an effective practical and reliable tool with which to measure internal lymphedema. In addition, it is generally accepted that there is no effective way to treat internal lymphedema. By contrast, external lymphedema is more readily observed, but both subjective and objective assessments are difficult. External swelling may occur in the face, jaw, and neck. However, the subjective scales currently available are insufficient to capture very important characteristics of external lymphedema.5 The Edge Task Force on Head and Neck Cancer in 2015 was not able to recommend any outcome measures for objectively quantifying external edema.6 Furthermore, objective measurements of head and neck lymphedema can be expensive and time consuming.
Extent and risk
A combination of both internal and external swelling is seen in more than 50% of patients.7 Risk factors include “throat” tumors, multicancer treatment approaches, higher total radiation dose, a greater number of radiation procedures, and radiation at the surgical site.5 More than 500,000 survivors of head and neck cancer in the United States are at risk of lymphedema.5 Although recent advances in treatment have reduced the incidence of other morbidities, 50% of patients who are treated for head and neck cancer may still develop lymphedema.1,8 The reported incidence in some centers may be much higher, with up to 75% of patients developing lymphedema following treatment.9
Measurement modalities for clinical evaluation
There is little current research into lymphedema of the head and neck, despite the high prevalence of the condition.8 According to Deng and colleagues, measurement of head and neck lymphedema is a challenge, which has an impact on clinical assessment, diagnosis, and treatment of this under-recognized, under-reported and under-addressed problem in head and neck cancer patients.10 In a review of the literature, Deng and colleagues identified three measurement modalities available for clinical evaluation: patient-reported outcomes, clinician-reported outcomes, and technology.10 One major factor, though, in detecting lymphedema, is physician awareness: physicians, health care professionals, and even some lymphedema therapists are not well educated about this problem.8
Treatment
The effectiveness of traditional lymphedema treatment is not well defined.8 Currently, complete decongestive therapy (CDT), is considered the standard of care for lymphedema. The National Lymphedema Network has stated that modifications of CDT, especially manual lymphatic drainage and modified compressive garments for external lymphedema, have been shown to be beneficial for the treatment of lymphedema in head and neck cancer patients.11 Most findings in lymphedema research, mainly in breast cancer patients, have shown that early intervention is the best management and yields the best outcomes. As with other chronic conditions, early identification and timely, appropriate treatment of lymphedema is critical to improve clinical outcomes, to decrease symptom burden and functional impairment, and to improve overall quality of life in head and neck cancer patients.10
Improving recognition and treatment
Head and neck oncologic treatment is increasingly offered outside the network of specialist academic hospitals, at hospitals serving more localized communities where the neediest, sickest patient groups may be receiving less than optimal care.3 This challenges community hospitals to provide optimal treatment, similar to that being offered at nationally recognized institutions. In January 2012, we implemented a prehabilitation program in our community hospital cancer center to provide early intervention for our patients based on the understanding that proper and prompt treatment for patients with early signs of lymphedema should be a priority.12 In this article, we outline how we implemented the program and the describe improvements we observed before and after the implementation of the program.
The prehabilitation program
The role of the nurse navigator
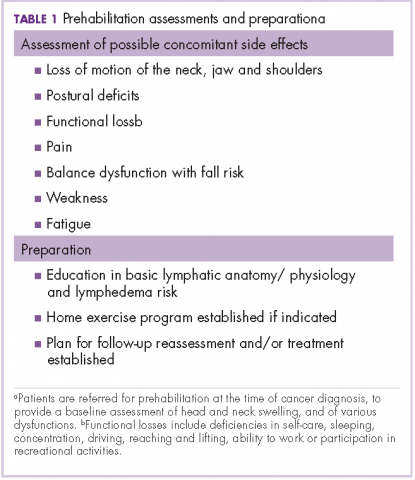
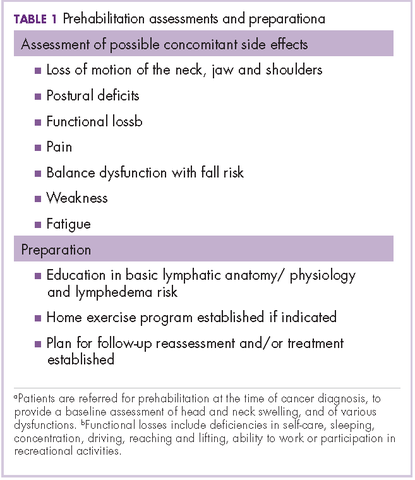
Before the introduction of the prehabilitation program, our pattern of practice was to refer patients to oncology rehabilitation for lymphedema management after they had completed their medical treatment with surgery, radiation, and chemotherapy. In 2012, that was changed to a prehabilitation model of care that was overseen by a head and neck nurse navigator. This focus on prehabilitation begins with patients being referred to oncology rehabilitation at the time of cancer diagnosis for baseline assessment of head and neck swelling. In addition, there is assessment of the many possible other side effects associated with head and neck cancer and its treatment, namely loss of range of motion of the neck, jaw (trismus), and/or shoulders, postural deficits, functional loss, pain, balance dysfunction with fall risk, weakness, and fatigue. Therapeutic interventions are initiated as needed and appropriate. This process also raises awareness of a condition that has been described as under-recognized and under-treated.3
The nurse navigator sits in on each radiation oncology consultation and aids in “navigating” patients through their treatment. The nurse ensures that each patient is referred to different ancillary services from the outset, such as seeing a dietician, social worker, physical/occupational therapist and certified lymphedema therapist, speech pathologist, and financial assistance advisor, if necessary (Table 1).
Assessment of lymphedema
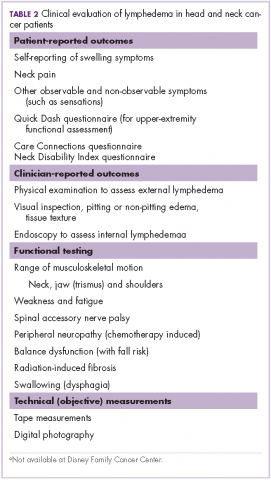
Measurement of head and neck lymphedema is a challenge.10 In our program, the physical therapy assessment also includes the evaluation of several other morbidities associated with head and neck cancer and its treatment, such as range of motion, weakness, fatigue, radiation fibrosis, balance dysfunction, and risk of falling (Table 2).
Patient-reported outcomes are essential to fully capture observable and unobservable symptoms (eg, sensations) as well as the functional impacts of lymphedema.10 In addition to lymphedema, there are many other morbidities that may be assessed on the basis of patient-reported outcome tools, such as upper extremity function with QuickDASH.13 At our clinic for head and neck cancer patients we use the Neck Disability Index (NDI)14 and Care Connections (CC)15 survey for the patient-reported outcomes. The Quick DASH, NDI, and CC tools all assess standard functional outcomes that are not specific to lymphedema, but are useful in documenting changes related to lymphedema. We initially used the CC survey and later transitioned to using the NDI. Neck pain is common with lymphedema in the head and neck region, and the NDI is a valid, reliable, responsive and internally consistent clinical tool to measure self-reported disability in patients with neck pain.16 These questionnaires were completed by the patients at their initial assessment, at reassessment, and at time of discharge.
Although objective criteria for external lymphedema have not been established, simple measurements such as using a tape measure to record neck circumference, allow a useful longitudinal assessment. Digital photography may be effective in the documentation and subjective evaluation of changes of external lymphedema.10,17 However, there are some limitations with photography because although external photographs (including digital photography and three-dimensional imaging) can capture some features, such as changes in contours, symmetry, and changes in skin quality and color, they do not detect changes in skin and soft tissue texture and compliance (Table 3).10
Impact on clinical outcomes
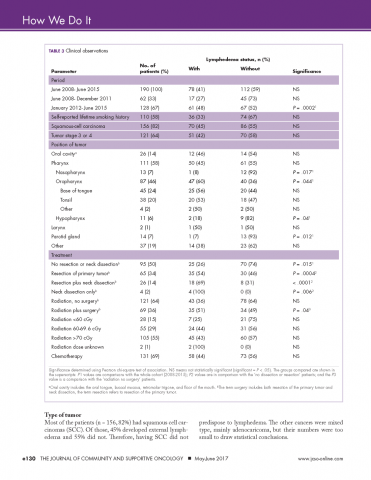
We retrospectively reviewed the medical records of 230 head and neck cancer patients who had been treated at our center between June 2008 and June 2015. Complete clinical data were available for 190 patients. The following information was extracted from each patient’s chart: whether they developed lymphedema, tumor stage, had surgery, radiation dose, type of chemotherapy given, their smoking history, if they had had a neck dissection and the primary site of the tumor (Table 3).
Incidence in different time periods. Of the 190 patients with complete records 78 (41%) were found to have lymphedema. These were all patients undergoing treatment for head and neck cancer during June 2008-June 2015. The prehabilitation program was initiated with the hiring of a nurse navigator for head and neck cancer, starting in January 2012. It is interesting to note that the incidence of lymphedema was 27% before the program was started, but after nurse navigator joined the team, the incidence increased significantly to 48% (P = .0002), in line with published expectations. This increase in recorded incidence may be attributable to the greater awareness of lymphedema intentionally fostered by the prehabilitation program.
Smoking history. Patients’ lifetime smoking history was retrieved from their medical records, based on their verbal admission of tobacco use. Most of the patients (n = 110) self-reported a history of smoking. Of those with a history of smoking, 36 (33%) developed external lymphedema after treatment for head and neck cancer, and 74 (67%) did not. However, this difference was not statistically significant. Hence, although smoking is a risk factor for head and neck cancer, it was not associated with the development of external lymphedema in our cohort of patients.
Type of tumor
Most of the patients (n = 156, 82%) had squamous cell carcinomas (SCC). Of those, 45% developed external lymphedema and 55% did not. Therefore, having SCC did not predispose to lymphedema. The other cancers were mixed type, mainly adenocaricoma, but their numbers were too small to draw statistical conclusions.
Stage of the tumor
About two thirds of the patients (n = 121, 64%) had stage 3 or 4 cancer. However, treatment of more advanced cancers was not associated with lymphedema development.
Site of the tumor
The literature suggests that patients with a primary tumor in the throat are at increased risk for lymphedema.5 The American Cancer Society has defined cancers of the oropharynx (throat) as including the base of the tongue (back third of the tongue), the soft palate, the tonsils, and the side and back walls of the throat.18 In our head and neck cancer cohort, patients with primary tumors of the oropharnyx were, perhaps, more susceptible to lymphedema (P = .044, Table 3). By contrast, in our cohort of patients, those with nasopharyngeal, hypopharyngeal, and parotid gland tumors were significantly less likely to develop lymphedema (Ps = .017, .04, .012, respectively).
No surgery
Half of our patients (n = 95) were not treated with surgery. In the patients who did not have surgery, 25 (26%) developed lymphedema, whereas 70 (74%) did not. Hence, although the incidence of lymphedema was significantly lower in patients who did not have surgery (P = .015), lymphedema did develop in patients who did not have a surgical procedure.
Resection of primary tumor without neck dissection
Of the 64 patients who had surgery, but without neck dissection, 35 (55%) developed external lymphedema. Compared with the no-surgery patients, the doubling of the incidence (from 26% to 55%) was highly significant (P = .0004). These findings are compatible with the literature reports that surgery increases the incidence of lymphedema, which is not surprising because surgery and subsequent scarring is known to compromise the lymphatic system.
Resection of primary tumor with neck dissection
The incidence of external lymphedema was increased to 69% when patients were subjected to both surgery and neck dissection. Compared with the June 2008-June 2015 cohort, there was a significant increase in the incidence of lymphedema in the neck dissection group (P = .007). Neck dissection involves the removal of lymph nodes and disruption of the lymphatic vessels, so it is not surprising that there is a higher incidence of external lymphedema. In our practice, neck dissections increased in frequency every year from June 2008 until December 2011, when 8 patients underwent neck dissections, 6 (75%) of whom developed lymphedema. Since January 2012, when the prehabilitation program was implemented, the number of neck dissections have declined, with more patients receiving chemoradiation and surgery being reserved for surgery. Hamoir and colleagues have reported that neck dissection is no longer justified unless there is clinically residual disease in the neck.19
Radiation
Lymphedema occurred in patients regardless of the dose of radiation received. Although the incidence of lymphedema seemed to be higher in patients who received more than 60 cGy, that difference was not statistically significant (Table 3). We had expected a relationship between radiation damage and greater lymphedema, but that was not evident in our patients.
Chemotherapy
The majority of patients (n = 131, 69%) received chemotherapy. The exposure to chemotherapy was not correlated with the risk of external lymphedema in our cohort of patients, with 58 of the 131 treated patients (44%) developing lymphedema, compared with 73 (56%) of treated patients who did not (Table 3).
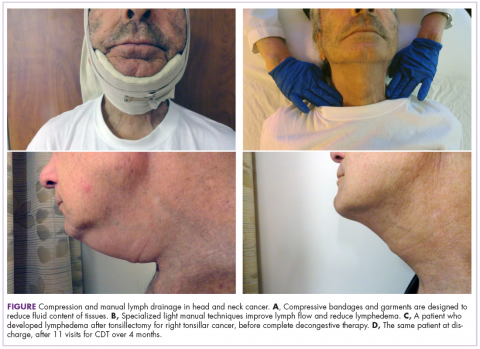
Complete decongestive therapy
All patients with documented lymphedema were evaluated for complete decongestive therapy (CDT). Contraindications to CDT included congestive heart failure, renal failure, acute infection, peripheral artery disease, upper-quadrant deep vein thrombosis, and carotid artery stenosis. Eligible patients were referred to a certified lymphedema therapist for CDT. As the program evolved, patients at risk for lymphedema were referred for CDT early on, usually at the time of diagnosis, to improve early identification and surveillance of lymphedema.
CDT included manual lymph drainage,
Patients’ responses to CDT were documented with digital photographs that were taken at each visit and, more recently, use of the NDI.
Communication and education
The head and neck cancer nurse navigator attends the cancer center’s multidisciplinary head and neck tumor board, which has representation from otolaryngology, diagnostic radiology, pathology, radiation oncology, medical oncology, reconstructive surgery, oncology rehabilitation (physical/occupational therapist), dietary services, speech pathology, social services and clinical research. This regular contact allows for earlier awareness about which patients are at greater risk for developing lymphedema, thus enabling early intervention (and patient education) in a timely manner.
Education of the patient, before cancer therapy, of the risks of lymphedema is very important. Before the implementation of the prehabilitation program, some patients did not fully comprehend what a painful and debilitating consequence of cancer treatment lymphedema could be.
Discussion
We introduced a prehabilitation program to detect and treat lymphedema in head and neck cancer patients in January 2012 part way through following an observation cohort from June2008 through June2015. Central to this, in our center, was the appointment of a nurse navigator whose primary focus was on head and neck cancer patients. We placed a high priority on the early detection and treatment of lymphedema because do so has been associated with better outcomes in other centers.
One immediate consequence of the inception of our program was the identification of more patients with external lymphedema. Our detected incidence rose significantly (P = .0002), from 27% in the period June 2008-December 20112010, before the program, to 48% during the January 2012-June 2015 period, after the inception of the program. This later incidence rate is in line with published incidence rates in most centers. However, it is still somewhat short of the 75% suggested in one center,9 which suggests we are either we are underdetecting lymphedema or there are differences in definition criteria or sensitivity levels for defining lymphedema.
There are currently no specific objective measures of lymphedema, so there is bound to be some variation in diagnosis rates. In our program, we rely heavily on the patient-reported outcome measures, the NDI instrument, and digital photography to detect and monitor lymphedema, starting with the pretreatment baseline values that are established for each patient.
The use of digital photography in our community hospital setting, which includes taking photographs before and after treatment and at each visit, motivates and encourages patients and provides a tool for clinical lymphedema therapists to visually document benefits of treatment. Patients’ motivation and compliance with their established home program for head and neck lymphedema self-management are essential. The elements of the home program may include self-manual lymph drainage, home-modified compression bandaging and garment wear, therapeutic exercises, and skin care. Patients with lymphedema who adhered closely with their therapy program were more than 8 times more likely to improve compared with noncompliant patients.17
Some groups of patients have a greater risk of developing lymphedema than others,5 so the development of an algorithm to predict lymphedema seemed possible. However, in our cohort of patients, only neck dissection, with its disruption of the lymphatic system of the neck, was strongly associated with external lymphedema (Table 3). It is important to note that some patients who did not undergo surgery developed lymphedema. In our patients, high doses of radiation alone did not seem to predispose to lymphedema. That suggests that no group of head and neck cancer patients should be ignored, which is why we did routine screening of all patients before, during, and after treatment.
Our protocol falls short in the detection of internal lymphedema. For example, information on swallowing gathered by our speech pathologists (in a different department) has not, so far, been included in our assessment. This is one opportunity to improve on our approach, especially because speech difficulties may be associated with internal lymphedema. In addition, we are not equipped for the requisite internal examinations. Unfortunately, there are no practical and successful treatments for patients suffering from internal swelling. This represents a challenge for the medical community to better meet this need. Therefore, although we are missing some assessments of internal lymphedema, this is of little therapeutic consequence at this time.
The increase in the detected incidence of external lymphedema points to a practice gap that has been resolved by the appointment of a dedicated nurse navigator who attends oncology reviews to share knowledge and information. Another educational effort has been made with the patients themselves to increase compliance and improve continuous care at home.
There is always room for improvement, however, either by feedback acquired from other institutions and hospitals or through the future introduction of more objective assessment techniques.
Conclusions
The introduction of the prehabilitation program at our center has coincided with a significantly improved detection rate for external lymphedema in head and neck cancer patients. It may be because the program emphasizes education about lymphedema that awareness of the condition has increased throughout the center. It is now widely recognized that all patients are at risk of lymphedema regardless of whether they fall into an acknowledged high-risk group. Our experience shows that there is no significant difference between treatment modalities apart from neck dissection. In our population, the use of this procedure is decreasing. External lymphedema can develop even in patients who do not have surgery. Therefore, there is no sound way to predict which patients are most likely to suffer from the accumulation of fluid in their head and neck after treatment for head and neck cancer. Thus, an assessment as described here, during and after treatment for all patients, is warranted. Patients are now being seen earlier as a part of the prehabilitation program, which facilitates access to complete decongestive treatment at an earlier stage, improves patient outcomes, and increases patient satisfaction with their treatment. Our prehabilitation program could serve as a model for other community hospital centers in achieving outcomes that are as good as those in academic centers.
Acknowledgments
The authors thank Irene Kadota and Heather Peters, from the Department of Radiation Oncology, and Julianne Courtenay, from the Department of Physical Therapy at the Disney Family Cancer Center, Burbank, California, for providing the original clinical data for analysis.
1. The National Lymphedema Medical Advisory Committee. The diagnosis and treatment of lymphedema. National Lymphedema Network. http://www.lymphnet.org/pdfDocs/nlntreatment.pdf. Updated February 2011. Accessed April 26, 2017.
2. McGarvey AC, Osmotherly PG, Hoffman GR, Chiarelli PE. Lymphedema following treatment for head and neck cancer: impact on patients, and beliefs of health professionals. Eur J Cancer Care (Engl). 2014;23(3):317-327.
3. Bhattacharyya N, Abemayor E. Patterns of hospital utilization for head and neck cancer care: changing demographics. JAMA Otolaryngol Head Neck Surg. 2015;141(4):307-312.
4. Deng J, Ridner SH, Dietrich MS, et al. Prevalence of secondary lymphedema in patients with head and neck cancer. J Pain Symptom Manage. 2012;43(2):244-252.
5. Deng J, Ridner SH, Dietrich MS, et al. Factors associated with external and internal lymphedema in patients with head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2012;84(3):e319-328.
6. Flores AM, Spinelli BA, Eden MM, Galantino ML. EDGE task force on head and neck cancer outcomes: a systematic review of outcomes measures for quantifying external lymphedema. Rehabil Oncol. 2015;33(2):15-23.
7. Ridner SH, Doersam J, Galford E. An update on lymphedema of the head and neck. http://www.lymphnet.org/pdfDocs/Vol_28-N2_Update_HN.pdf. Published April-June 2015. Accessed April 26, 2017.
8. Smith BG, Hutcheson KA, Little LG, et al. Lymphedema outcomes in patients with head and neck cancer. Otolaryngol Head Neck Surg. 2015;152(2);284-291.
9. Naqvi SHS, Karni RJ, Tan IC, et al. Int J Rad Oncol Biol Phys. 2016;4:927-928.
10. Deng J, Ridner SH, Aulino JM, Murphy BA. Assessment and measurement of head and neck lymphedema: state-of-the-science and future directions. Oral Oncol. 2015; 51(5):431-437.
11. Purcell A. Head and neck lymphedema management practices. J Lymphedema. 2013;8(2):8-15.
12. Paskett ED, Dean JA, Oliveri JM, Harrop JP. Cancer-related lymphedema risk factors, diagnosis treatment and impact: a review. J Clinl Oncol. 2012;30(30):3726-3733.
13. Quick DASH questionnaire. http://www.dash.iwh.on.ca/about-quickdash. [Last update not stated.] Accessed May 18, 2017.
14. Neck Disability Index questionnaire. www.aaos.org/uploadedFiles/NDI.pdf Accessed May 18, 2017.
15. Care Connections questionnaire. http://www.careconnections.com/. Accessed May 18, 2017.
16. Galantino ML, Eden MM, Spinelli BA, Flores AM. EDGE task force on head and neck cancer outcomes a systematic review of outcome measures for temporomandibular-related dysfunction. Rehabil Oncol. 2015;33(1):6-14.
17. Deng J, Ridner SH, Murphy BA. Lymphedema in patients with head and neck cancer. Oncol Nurs Forum. 2011;38(1):e1-e10.
18. What are oral cavity and oropharyngeal cancers? American Cancer Society. http://www.cancer.org/cancer/oralcavityandoropharyngealcancer/detailedguide/oral-cavity-and-oropharyngeal-cancer-what-is-oral-cavity-cancer. Last revised August 8, 2016. Accessed April 26, 2017.
19. Hamoir M, Schmitz S, Gregoire V. The role of neck dissection in squamous cell carcinoma of the head and neck. Curr Treat Options Oncol. 2014;15:611-624.
Lymphedema is the swelling of tissue caused by the accumulation of interstitial fluid in any area of the body where lymphatic flow has been compromised.1 Secondary lymphedema is an acquired abnormality in lymph drainage1,2 and is the type commonly seen in cancer patients. Secondary lymphedema can be described as external or internal. Internal lymphedema, swelling of deep structures and tissues, is very difficult to quantify.
Lymphedema in patients with head and neck cancers
Lymphedema is a complicating morbidity frequently seen in head and neck cancer patients who have undergone treatment with surgery, radiation, and chemotherapy. However, although it is one of the most prevalent side effects of treatment, it is both under-recognized and under-treated.3
In head and neck cancer patients, internal swelling may develop in the soft tissues of the upper aero-digestive tract,4 affecting articulation and swallowing. Currently, there does not seem to be an effective practical and reliable tool with which to measure internal lymphedema. In addition, it is generally accepted that there is no effective way to treat internal lymphedema. By contrast, external lymphedema is more readily observed, but both subjective and objective assessments are difficult. External swelling may occur in the face, jaw, and neck. However, the subjective scales currently available are insufficient to capture very important characteristics of external lymphedema.5 The Edge Task Force on Head and Neck Cancer in 2015 was not able to recommend any outcome measures for objectively quantifying external edema.6 Furthermore, objective measurements of head and neck lymphedema can be expensive and time consuming.
Extent and risk
A combination of both internal and external swelling is seen in more than 50% of patients.7 Risk factors include “throat” tumors, multicancer treatment approaches, higher total radiation dose, a greater number of radiation procedures, and radiation at the surgical site.5 More than 500,000 survivors of head and neck cancer in the United States are at risk of lymphedema.5 Although recent advances in treatment have reduced the incidence of other morbidities, 50% of patients who are treated for head and neck cancer may still develop lymphedema.1,8 The reported incidence in some centers may be much higher, with up to 75% of patients developing lymphedema following treatment.9
Measurement modalities for clinical evaluation
There is little current research into lymphedema of the head and neck, despite the high prevalence of the condition.8 According to Deng and colleagues, measurement of head and neck lymphedema is a challenge, which has an impact on clinical assessment, diagnosis, and treatment of this under-recognized, under-reported and under-addressed problem in head and neck cancer patients.10 In a review of the literature, Deng and colleagues identified three measurement modalities available for clinical evaluation: patient-reported outcomes, clinician-reported outcomes, and technology.10 One major factor, though, in detecting lymphedema, is physician awareness: physicians, health care professionals, and even some lymphedema therapists are not well educated about this problem.8
Treatment
The effectiveness of traditional lymphedema treatment is not well defined.8 Currently, complete decongestive therapy (CDT), is considered the standard of care for lymphedema. The National Lymphedema Network has stated that modifications of CDT, especially manual lymphatic drainage and modified compressive garments for external lymphedema, have been shown to be beneficial for the treatment of lymphedema in head and neck cancer patients.11 Most findings in lymphedema research, mainly in breast cancer patients, have shown that early intervention is the best management and yields the best outcomes. As with other chronic conditions, early identification and timely, appropriate treatment of lymphedema is critical to improve clinical outcomes, to decrease symptom burden and functional impairment, and to improve overall quality of life in head and neck cancer patients.10
Improving recognition and treatment
Head and neck oncologic treatment is increasingly offered outside the network of specialist academic hospitals, at hospitals serving more localized communities where the neediest, sickest patient groups may be receiving less than optimal care.3 This challenges community hospitals to provide optimal treatment, similar to that being offered at nationally recognized institutions. In January 2012, we implemented a prehabilitation program in our community hospital cancer center to provide early intervention for our patients based on the understanding that proper and prompt treatment for patients with early signs of lymphedema should be a priority.12 In this article, we outline how we implemented the program and the describe improvements we observed before and after the implementation of the program.
The prehabilitation program
The role of the nurse navigator
Before the introduction of the prehabilitation program, our pattern of practice was to refer patients to oncology rehabilitation for lymphedema management after they had completed their medical treatment with surgery, radiation, and chemotherapy. In 2012, that was changed to a prehabilitation model of care that was overseen by a head and neck nurse navigator. This focus on prehabilitation begins with patients being referred to oncology rehabilitation at the time of cancer diagnosis for baseline assessment of head and neck swelling. In addition, there is assessment of the many possible other side effects associated with head and neck cancer and its treatment, namely loss of range of motion of the neck, jaw (trismus), and/or shoulders, postural deficits, functional loss, pain, balance dysfunction with fall risk, weakness, and fatigue. Therapeutic interventions are initiated as needed and appropriate. This process also raises awareness of a condition that has been described as under-recognized and under-treated.3
The nurse navigator sits in on each radiation oncology consultation and aids in “navigating” patients through their treatment. The nurse ensures that each patient is referred to different ancillary services from the outset, such as seeing a dietician, social worker, physical/occupational therapist and certified lymphedema therapist, speech pathologist, and financial assistance advisor, if necessary (Table 1).
Assessment of lymphedema
Measurement of head and neck lymphedema is a challenge.10 In our program, the physical therapy assessment also includes the evaluation of several other morbidities associated with head and neck cancer and its treatment, such as range of motion, weakness, fatigue, radiation fibrosis, balance dysfunction, and risk of falling (Table 2).
Patient-reported outcomes are essential to fully capture observable and unobservable symptoms (eg, sensations) as well as the functional impacts of lymphedema.10 In addition to lymphedema, there are many other morbidities that may be assessed on the basis of patient-reported outcome tools, such as upper extremity function with QuickDASH.13 At our clinic for head and neck cancer patients we use the Neck Disability Index (NDI)14 and Care Connections (CC)15 survey for the patient-reported outcomes. The Quick DASH, NDI, and CC tools all assess standard functional outcomes that are not specific to lymphedema, but are useful in documenting changes related to lymphedema. We initially used the CC survey and later transitioned to using the NDI. Neck pain is common with lymphedema in the head and neck region, and the NDI is a valid, reliable, responsive and internally consistent clinical tool to measure self-reported disability in patients with neck pain.16 These questionnaires were completed by the patients at their initial assessment, at reassessment, and at time of discharge.
Although objective criteria for external lymphedema have not been established, simple measurements such as using a tape measure to record neck circumference, allow a useful longitudinal assessment. Digital photography may be effective in the documentation and subjective evaluation of changes of external lymphedema.10,17 However, there are some limitations with photography because although external photographs (including digital photography and three-dimensional imaging) can capture some features, such as changes in contours, symmetry, and changes in skin quality and color, they do not detect changes in skin and soft tissue texture and compliance (Table 3).10
Impact on clinical outcomes
We retrospectively reviewed the medical records of 230 head and neck cancer patients who had been treated at our center between June 2008 and June 2015. Complete clinical data were available for 190 patients. The following information was extracted from each patient’s chart: whether they developed lymphedema, tumor stage, had surgery, radiation dose, type of chemotherapy given, their smoking history, if they had had a neck dissection and the primary site of the tumor (Table 3).
Incidence in different time periods. Of the 190 patients with complete records 78 (41%) were found to have lymphedema. These were all patients undergoing treatment for head and neck cancer during June 2008-June 2015. The prehabilitation program was initiated with the hiring of a nurse navigator for head and neck cancer, starting in January 2012. It is interesting to note that the incidence of lymphedema was 27% before the program was started, but after nurse navigator joined the team, the incidence increased significantly to 48% (P = .0002), in line with published expectations. This increase in recorded incidence may be attributable to the greater awareness of lymphedema intentionally fostered by the prehabilitation program.
Smoking history. Patients’ lifetime smoking history was retrieved from their medical records, based on their verbal admission of tobacco use. Most of the patients (n = 110) self-reported a history of smoking. Of those with a history of smoking, 36 (33%) developed external lymphedema after treatment for head and neck cancer, and 74 (67%) did not. However, this difference was not statistically significant. Hence, although smoking is a risk factor for head and neck cancer, it was not associated with the development of external lymphedema in our cohort of patients.
Type of tumor
Most of the patients (n = 156, 82%) had squamous cell carcinomas (SCC). Of those, 45% developed external lymphedema and 55% did not. Therefore, having SCC did not predispose to lymphedema. The other cancers were mixed type, mainly adenocaricoma, but their numbers were too small to draw statistical conclusions.
Stage of the tumor
About two thirds of the patients (n = 121, 64%) had stage 3 or 4 cancer. However, treatment of more advanced cancers was not associated with lymphedema development.
Site of the tumor
The literature suggests that patients with a primary tumor in the throat are at increased risk for lymphedema.5 The American Cancer Society has defined cancers of the oropharynx (throat) as including the base of the tongue (back third of the tongue), the soft palate, the tonsils, and the side and back walls of the throat.18 In our head and neck cancer cohort, patients with primary tumors of the oropharnyx were, perhaps, more susceptible to lymphedema (P = .044, Table 3). By contrast, in our cohort of patients, those with nasopharyngeal, hypopharyngeal, and parotid gland tumors were significantly less likely to develop lymphedema (Ps = .017, .04, .012, respectively).
No surgery
Half of our patients (n = 95) were not treated with surgery. In the patients who did not have surgery, 25 (26%) developed lymphedema, whereas 70 (74%) did not. Hence, although the incidence of lymphedema was significantly lower in patients who did not have surgery (P = .015), lymphedema did develop in patients who did not have a surgical procedure.
Resection of primary tumor without neck dissection
Of the 64 patients who had surgery, but without neck dissection, 35 (55%) developed external lymphedema. Compared with the no-surgery patients, the doubling of the incidence (from 26% to 55%) was highly significant (P = .0004). These findings are compatible with the literature reports that surgery increases the incidence of lymphedema, which is not surprising because surgery and subsequent scarring is known to compromise the lymphatic system.
Resection of primary tumor with neck dissection
The incidence of external lymphedema was increased to 69% when patients were subjected to both surgery and neck dissection. Compared with the June 2008-June 2015 cohort, there was a significant increase in the incidence of lymphedema in the neck dissection group (P = .007). Neck dissection involves the removal of lymph nodes and disruption of the lymphatic vessels, so it is not surprising that there is a higher incidence of external lymphedema. In our practice, neck dissections increased in frequency every year from June 2008 until December 2011, when 8 patients underwent neck dissections, 6 (75%) of whom developed lymphedema. Since January 2012, when the prehabilitation program was implemented, the number of neck dissections have declined, with more patients receiving chemoradiation and surgery being reserved for surgery. Hamoir and colleagues have reported that neck dissection is no longer justified unless there is clinically residual disease in the neck.19
Radiation
Lymphedema occurred in patients regardless of the dose of radiation received. Although the incidence of lymphedema seemed to be higher in patients who received more than 60 cGy, that difference was not statistically significant (Table 3). We had expected a relationship between radiation damage and greater lymphedema, but that was not evident in our patients.
Chemotherapy
The majority of patients (n = 131, 69%) received chemotherapy. The exposure to chemotherapy was not correlated with the risk of external lymphedema in our cohort of patients, with 58 of the 131 treated patients (44%) developing lymphedema, compared with 73 (56%) of treated patients who did not (Table 3).
Complete decongestive therapy
All patients with documented lymphedema were evaluated for complete decongestive therapy (CDT). Contraindications to CDT included congestive heart failure, renal failure, acute infection, peripheral artery disease, upper-quadrant deep vein thrombosis, and carotid artery stenosis. Eligible patients were referred to a certified lymphedema therapist for CDT. As the program evolved, patients at risk for lymphedema were referred for CDT early on, usually at the time of diagnosis, to improve early identification and surveillance of lymphedema.
CDT included manual lymph drainage,
Patients’ responses to CDT were documented with digital photographs that were taken at each visit and, more recently, use of the NDI.
Communication and education
The head and neck cancer nurse navigator attends the cancer center’s multidisciplinary head and neck tumor board, which has representation from otolaryngology, diagnostic radiology, pathology, radiation oncology, medical oncology, reconstructive surgery, oncology rehabilitation (physical/occupational therapist), dietary services, speech pathology, social services and clinical research. This regular contact allows for earlier awareness about which patients are at greater risk for developing lymphedema, thus enabling early intervention (and patient education) in a timely manner.
Education of the patient, before cancer therapy, of the risks of lymphedema is very important. Before the implementation of the prehabilitation program, some patients did not fully comprehend what a painful and debilitating consequence of cancer treatment lymphedema could be.
Discussion
We introduced a prehabilitation program to detect and treat lymphedema in head and neck cancer patients in January 2012 part way through following an observation cohort from June2008 through June2015. Central to this, in our center, was the appointment of a nurse navigator whose primary focus was on head and neck cancer patients. We placed a high priority on the early detection and treatment of lymphedema because do so has been associated with better outcomes in other centers.
One immediate consequence of the inception of our program was the identification of more patients with external lymphedema. Our detected incidence rose significantly (P = .0002), from 27% in the period June 2008-December 20112010, before the program, to 48% during the January 2012-June 2015 period, after the inception of the program. This later incidence rate is in line with published incidence rates in most centers. However, it is still somewhat short of the 75% suggested in one center,9 which suggests we are either we are underdetecting lymphedema or there are differences in definition criteria or sensitivity levels for defining lymphedema.
There are currently no specific objective measures of lymphedema, so there is bound to be some variation in diagnosis rates. In our program, we rely heavily on the patient-reported outcome measures, the NDI instrument, and digital photography to detect and monitor lymphedema, starting with the pretreatment baseline values that are established for each patient.
The use of digital photography in our community hospital setting, which includes taking photographs before and after treatment and at each visit, motivates and encourages patients and provides a tool for clinical lymphedema therapists to visually document benefits of treatment. Patients’ motivation and compliance with their established home program for head and neck lymphedema self-management are essential. The elements of the home program may include self-manual lymph drainage, home-modified compression bandaging and garment wear, therapeutic exercises, and skin care. Patients with lymphedema who adhered closely with their therapy program were more than 8 times more likely to improve compared with noncompliant patients.17
Some groups of patients have a greater risk of developing lymphedema than others,5 so the development of an algorithm to predict lymphedema seemed possible. However, in our cohort of patients, only neck dissection, with its disruption of the lymphatic system of the neck, was strongly associated with external lymphedema (Table 3). It is important to note that some patients who did not undergo surgery developed lymphedema. In our patients, high doses of radiation alone did not seem to predispose to lymphedema. That suggests that no group of head and neck cancer patients should be ignored, which is why we did routine screening of all patients before, during, and after treatment.
Our protocol falls short in the detection of internal lymphedema. For example, information on swallowing gathered by our speech pathologists (in a different department) has not, so far, been included in our assessment. This is one opportunity to improve on our approach, especially because speech difficulties may be associated with internal lymphedema. In addition, we are not equipped for the requisite internal examinations. Unfortunately, there are no practical and successful treatments for patients suffering from internal swelling. This represents a challenge for the medical community to better meet this need. Therefore, although we are missing some assessments of internal lymphedema, this is of little therapeutic consequence at this time.
The increase in the detected incidence of external lymphedema points to a practice gap that has been resolved by the appointment of a dedicated nurse navigator who attends oncology reviews to share knowledge and information. Another educational effort has been made with the patients themselves to increase compliance and improve continuous care at home.
There is always room for improvement, however, either by feedback acquired from other institutions and hospitals or through the future introduction of more objective assessment techniques.
Conclusions
The introduction of the prehabilitation program at our center has coincided with a significantly improved detection rate for external lymphedema in head and neck cancer patients. It may be because the program emphasizes education about lymphedema that awareness of the condition has increased throughout the center. It is now widely recognized that all patients are at risk of lymphedema regardless of whether they fall into an acknowledged high-risk group. Our experience shows that there is no significant difference between treatment modalities apart from neck dissection. In our population, the use of this procedure is decreasing. External lymphedema can develop even in patients who do not have surgery. Therefore, there is no sound way to predict which patients are most likely to suffer from the accumulation of fluid in their head and neck after treatment for head and neck cancer. Thus, an assessment as described here, during and after treatment for all patients, is warranted. Patients are now being seen earlier as a part of the prehabilitation program, which facilitates access to complete decongestive treatment at an earlier stage, improves patient outcomes, and increases patient satisfaction with their treatment. Our prehabilitation program could serve as a model for other community hospital centers in achieving outcomes that are as good as those in academic centers.
Acknowledgments
The authors thank Irene Kadota and Heather Peters, from the Department of Radiation Oncology, and Julianne Courtenay, from the Department of Physical Therapy at the Disney Family Cancer Center, Burbank, California, for providing the original clinical data for analysis.
Lymphedema is the swelling of tissue caused by the accumulation of interstitial fluid in any area of the body where lymphatic flow has been compromised.1 Secondary lymphedema is an acquired abnormality in lymph drainage1,2 and is the type commonly seen in cancer patients. Secondary lymphedema can be described as external or internal. Internal lymphedema, swelling of deep structures and tissues, is very difficult to quantify.
Lymphedema in patients with head and neck cancers
Lymphedema is a complicating morbidity frequently seen in head and neck cancer patients who have undergone treatment with surgery, radiation, and chemotherapy. However, although it is one of the most prevalent side effects of treatment, it is both under-recognized and under-treated.3
In head and neck cancer patients, internal swelling may develop in the soft tissues of the upper aero-digestive tract,4 affecting articulation and swallowing. Currently, there does not seem to be an effective practical and reliable tool with which to measure internal lymphedema. In addition, it is generally accepted that there is no effective way to treat internal lymphedema. By contrast, external lymphedema is more readily observed, but both subjective and objective assessments are difficult. External swelling may occur in the face, jaw, and neck. However, the subjective scales currently available are insufficient to capture very important characteristics of external lymphedema.5 The Edge Task Force on Head and Neck Cancer in 2015 was not able to recommend any outcome measures for objectively quantifying external edema.6 Furthermore, objective measurements of head and neck lymphedema can be expensive and time consuming.
Extent and risk
A combination of both internal and external swelling is seen in more than 50% of patients.7 Risk factors include “throat” tumors, multicancer treatment approaches, higher total radiation dose, a greater number of radiation procedures, and radiation at the surgical site.5 More than 500,000 survivors of head and neck cancer in the United States are at risk of lymphedema.5 Although recent advances in treatment have reduced the incidence of other morbidities, 50% of patients who are treated for head and neck cancer may still develop lymphedema.1,8 The reported incidence in some centers may be much higher, with up to 75% of patients developing lymphedema following treatment.9
Measurement modalities for clinical evaluation
There is little current research into lymphedema of the head and neck, despite the high prevalence of the condition.8 According to Deng and colleagues, measurement of head and neck lymphedema is a challenge, which has an impact on clinical assessment, diagnosis, and treatment of this under-recognized, under-reported and under-addressed problem in head and neck cancer patients.10 In a review of the literature, Deng and colleagues identified three measurement modalities available for clinical evaluation: patient-reported outcomes, clinician-reported outcomes, and technology.10 One major factor, though, in detecting lymphedema, is physician awareness: physicians, health care professionals, and even some lymphedema therapists are not well educated about this problem.8
Treatment
The effectiveness of traditional lymphedema treatment is not well defined.8 Currently, complete decongestive therapy (CDT), is considered the standard of care for lymphedema. The National Lymphedema Network has stated that modifications of CDT, especially manual lymphatic drainage and modified compressive garments for external lymphedema, have been shown to be beneficial for the treatment of lymphedema in head and neck cancer patients.11 Most findings in lymphedema research, mainly in breast cancer patients, have shown that early intervention is the best management and yields the best outcomes. As with other chronic conditions, early identification and timely, appropriate treatment of lymphedema is critical to improve clinical outcomes, to decrease symptom burden and functional impairment, and to improve overall quality of life in head and neck cancer patients.10
Improving recognition and treatment
Head and neck oncologic treatment is increasingly offered outside the network of specialist academic hospitals, at hospitals serving more localized communities where the neediest, sickest patient groups may be receiving less than optimal care.3 This challenges community hospitals to provide optimal treatment, similar to that being offered at nationally recognized institutions. In January 2012, we implemented a prehabilitation program in our community hospital cancer center to provide early intervention for our patients based on the understanding that proper and prompt treatment for patients with early signs of lymphedema should be a priority.12 In this article, we outline how we implemented the program and the describe improvements we observed before and after the implementation of the program.
The prehabilitation program
The role of the nurse navigator
Before the introduction of the prehabilitation program, our pattern of practice was to refer patients to oncology rehabilitation for lymphedema management after they had completed their medical treatment with surgery, radiation, and chemotherapy. In 2012, that was changed to a prehabilitation model of care that was overseen by a head and neck nurse navigator. This focus on prehabilitation begins with patients being referred to oncology rehabilitation at the time of cancer diagnosis for baseline assessment of head and neck swelling. In addition, there is assessment of the many possible other side effects associated with head and neck cancer and its treatment, namely loss of range of motion of the neck, jaw (trismus), and/or shoulders, postural deficits, functional loss, pain, balance dysfunction with fall risk, weakness, and fatigue. Therapeutic interventions are initiated as needed and appropriate. This process also raises awareness of a condition that has been described as under-recognized and under-treated.3
The nurse navigator sits in on each radiation oncology consultation and aids in “navigating” patients through their treatment. The nurse ensures that each patient is referred to different ancillary services from the outset, such as seeing a dietician, social worker, physical/occupational therapist and certified lymphedema therapist, speech pathologist, and financial assistance advisor, if necessary (Table 1).
Assessment of lymphedema
Measurement of head and neck lymphedema is a challenge.10 In our program, the physical therapy assessment also includes the evaluation of several other morbidities associated with head and neck cancer and its treatment, such as range of motion, weakness, fatigue, radiation fibrosis, balance dysfunction, and risk of falling (Table 2).
Patient-reported outcomes are essential to fully capture observable and unobservable symptoms (eg, sensations) as well as the functional impacts of lymphedema.10 In addition to lymphedema, there are many other morbidities that may be assessed on the basis of patient-reported outcome tools, such as upper extremity function with QuickDASH.13 At our clinic for head and neck cancer patients we use the Neck Disability Index (NDI)14 and Care Connections (CC)15 survey for the patient-reported outcomes. The Quick DASH, NDI, and CC tools all assess standard functional outcomes that are not specific to lymphedema, but are useful in documenting changes related to lymphedema. We initially used the CC survey and later transitioned to using the NDI. Neck pain is common with lymphedema in the head and neck region, and the NDI is a valid, reliable, responsive and internally consistent clinical tool to measure self-reported disability in patients with neck pain.16 These questionnaires were completed by the patients at their initial assessment, at reassessment, and at time of discharge.
Although objective criteria for external lymphedema have not been established, simple measurements such as using a tape measure to record neck circumference, allow a useful longitudinal assessment. Digital photography may be effective in the documentation and subjective evaluation of changes of external lymphedema.10,17 However, there are some limitations with photography because although external photographs (including digital photography and three-dimensional imaging) can capture some features, such as changes in contours, symmetry, and changes in skin quality and color, they do not detect changes in skin and soft tissue texture and compliance (Table 3).10
Impact on clinical outcomes
We retrospectively reviewed the medical records of 230 head and neck cancer patients who had been treated at our center between June 2008 and June 2015. Complete clinical data were available for 190 patients. The following information was extracted from each patient’s chart: whether they developed lymphedema, tumor stage, had surgery, radiation dose, type of chemotherapy given, their smoking history, if they had had a neck dissection and the primary site of the tumor (Table 3).
Incidence in different time periods. Of the 190 patients with complete records 78 (41%) were found to have lymphedema. These were all patients undergoing treatment for head and neck cancer during June 2008-June 2015. The prehabilitation program was initiated with the hiring of a nurse navigator for head and neck cancer, starting in January 2012. It is interesting to note that the incidence of lymphedema was 27% before the program was started, but after nurse navigator joined the team, the incidence increased significantly to 48% (P = .0002), in line with published expectations. This increase in recorded incidence may be attributable to the greater awareness of lymphedema intentionally fostered by the prehabilitation program.
Smoking history. Patients’ lifetime smoking history was retrieved from their medical records, based on their verbal admission of tobacco use. Most of the patients (n = 110) self-reported a history of smoking. Of those with a history of smoking, 36 (33%) developed external lymphedema after treatment for head and neck cancer, and 74 (67%) did not. However, this difference was not statistically significant. Hence, although smoking is a risk factor for head and neck cancer, it was not associated with the development of external lymphedema in our cohort of patients.
Type of tumor
Most of the patients (n = 156, 82%) had squamous cell carcinomas (SCC). Of those, 45% developed external lymphedema and 55% did not. Therefore, having SCC did not predispose to lymphedema. The other cancers were mixed type, mainly adenocaricoma, but their numbers were too small to draw statistical conclusions.
Stage of the tumor
About two thirds of the patients (n = 121, 64%) had stage 3 or 4 cancer. However, treatment of more advanced cancers was not associated with lymphedema development.
Site of the tumor
The literature suggests that patients with a primary tumor in the throat are at increased risk for lymphedema.5 The American Cancer Society has defined cancers of the oropharynx (throat) as including the base of the tongue (back third of the tongue), the soft palate, the tonsils, and the side and back walls of the throat.18 In our head and neck cancer cohort, patients with primary tumors of the oropharnyx were, perhaps, more susceptible to lymphedema (P = .044, Table 3). By contrast, in our cohort of patients, those with nasopharyngeal, hypopharyngeal, and parotid gland tumors were significantly less likely to develop lymphedema (Ps = .017, .04, .012, respectively).
No surgery
Half of our patients (n = 95) were not treated with surgery. In the patients who did not have surgery, 25 (26%) developed lymphedema, whereas 70 (74%) did not. Hence, although the incidence of lymphedema was significantly lower in patients who did not have surgery (P = .015), lymphedema did develop in patients who did not have a surgical procedure.
Resection of primary tumor without neck dissection
Of the 64 patients who had surgery, but without neck dissection, 35 (55%) developed external lymphedema. Compared with the no-surgery patients, the doubling of the incidence (from 26% to 55%) was highly significant (P = .0004). These findings are compatible with the literature reports that surgery increases the incidence of lymphedema, which is not surprising because surgery and subsequent scarring is known to compromise the lymphatic system.
Resection of primary tumor with neck dissection
The incidence of external lymphedema was increased to 69% when patients were subjected to both surgery and neck dissection. Compared with the June 2008-June 2015 cohort, there was a significant increase in the incidence of lymphedema in the neck dissection group (P = .007). Neck dissection involves the removal of lymph nodes and disruption of the lymphatic vessels, so it is not surprising that there is a higher incidence of external lymphedema. In our practice, neck dissections increased in frequency every year from June 2008 until December 2011, when 8 patients underwent neck dissections, 6 (75%) of whom developed lymphedema. Since January 2012, when the prehabilitation program was implemented, the number of neck dissections have declined, with more patients receiving chemoradiation and surgery being reserved for surgery. Hamoir and colleagues have reported that neck dissection is no longer justified unless there is clinically residual disease in the neck.19
Radiation
Lymphedema occurred in patients regardless of the dose of radiation received. Although the incidence of lymphedema seemed to be higher in patients who received more than 60 cGy, that difference was not statistically significant (Table 3). We had expected a relationship between radiation damage and greater lymphedema, but that was not evident in our patients.
Chemotherapy
The majority of patients (n = 131, 69%) received chemotherapy. The exposure to chemotherapy was not correlated with the risk of external lymphedema in our cohort of patients, with 58 of the 131 treated patients (44%) developing lymphedema, compared with 73 (56%) of treated patients who did not (Table 3).
Complete decongestive therapy
All patients with documented lymphedema were evaluated for complete decongestive therapy (CDT). Contraindications to CDT included congestive heart failure, renal failure, acute infection, peripheral artery disease, upper-quadrant deep vein thrombosis, and carotid artery stenosis. Eligible patients were referred to a certified lymphedema therapist for CDT. As the program evolved, patients at risk for lymphedema were referred for CDT early on, usually at the time of diagnosis, to improve early identification and surveillance of lymphedema.
CDT included manual lymph drainage,
Patients’ responses to CDT were documented with digital photographs that were taken at each visit and, more recently, use of the NDI.
Communication and education
The head and neck cancer nurse navigator attends the cancer center’s multidisciplinary head and neck tumor board, which has representation from otolaryngology, diagnostic radiology, pathology, radiation oncology, medical oncology, reconstructive surgery, oncology rehabilitation (physical/occupational therapist), dietary services, speech pathology, social services and clinical research. This regular contact allows for earlier awareness about which patients are at greater risk for developing lymphedema, thus enabling early intervention (and patient education) in a timely manner.
Education of the patient, before cancer therapy, of the risks of lymphedema is very important. Before the implementation of the prehabilitation program, some patients did not fully comprehend what a painful and debilitating consequence of cancer treatment lymphedema could be.
Discussion
We introduced a prehabilitation program to detect and treat lymphedema in head and neck cancer patients in January 2012 part way through following an observation cohort from June2008 through June2015. Central to this, in our center, was the appointment of a nurse navigator whose primary focus was on head and neck cancer patients. We placed a high priority on the early detection and treatment of lymphedema because do so has been associated with better outcomes in other centers.
One immediate consequence of the inception of our program was the identification of more patients with external lymphedema. Our detected incidence rose significantly (P = .0002), from 27% in the period June 2008-December 20112010, before the program, to 48% during the January 2012-June 2015 period, after the inception of the program. This later incidence rate is in line with published incidence rates in most centers. However, it is still somewhat short of the 75% suggested in one center,9 which suggests we are either we are underdetecting lymphedema or there are differences in definition criteria or sensitivity levels for defining lymphedema.
There are currently no specific objective measures of lymphedema, so there is bound to be some variation in diagnosis rates. In our program, we rely heavily on the patient-reported outcome measures, the NDI instrument, and digital photography to detect and monitor lymphedema, starting with the pretreatment baseline values that are established for each patient.
The use of digital photography in our community hospital setting, which includes taking photographs before and after treatment and at each visit, motivates and encourages patients and provides a tool for clinical lymphedema therapists to visually document benefits of treatment. Patients’ motivation and compliance with their established home program for head and neck lymphedema self-management are essential. The elements of the home program may include self-manual lymph drainage, home-modified compression bandaging and garment wear, therapeutic exercises, and skin care. Patients with lymphedema who adhered closely with their therapy program were more than 8 times more likely to improve compared with noncompliant patients.17
Some groups of patients have a greater risk of developing lymphedema than others,5 so the development of an algorithm to predict lymphedema seemed possible. However, in our cohort of patients, only neck dissection, with its disruption of the lymphatic system of the neck, was strongly associated with external lymphedema (Table 3). It is important to note that some patients who did not undergo surgery developed lymphedema. In our patients, high doses of radiation alone did not seem to predispose to lymphedema. That suggests that no group of head and neck cancer patients should be ignored, which is why we did routine screening of all patients before, during, and after treatment.
Our protocol falls short in the detection of internal lymphedema. For example, information on swallowing gathered by our speech pathologists (in a different department) has not, so far, been included in our assessment. This is one opportunity to improve on our approach, especially because speech difficulties may be associated with internal lymphedema. In addition, we are not equipped for the requisite internal examinations. Unfortunately, there are no practical and successful treatments for patients suffering from internal swelling. This represents a challenge for the medical community to better meet this need. Therefore, although we are missing some assessments of internal lymphedema, this is of little therapeutic consequence at this time.
The increase in the detected incidence of external lymphedema points to a practice gap that has been resolved by the appointment of a dedicated nurse navigator who attends oncology reviews to share knowledge and information. Another educational effort has been made with the patients themselves to increase compliance and improve continuous care at home.
There is always room for improvement, however, either by feedback acquired from other institutions and hospitals or through the future introduction of more objective assessment techniques.
Conclusions
The introduction of the prehabilitation program at our center has coincided with a significantly improved detection rate for external lymphedema in head and neck cancer patients. It may be because the program emphasizes education about lymphedema that awareness of the condition has increased throughout the center. It is now widely recognized that all patients are at risk of lymphedema regardless of whether they fall into an acknowledged high-risk group. Our experience shows that there is no significant difference between treatment modalities apart from neck dissection. In our population, the use of this procedure is decreasing. External lymphedema can develop even in patients who do not have surgery. Therefore, there is no sound way to predict which patients are most likely to suffer from the accumulation of fluid in their head and neck after treatment for head and neck cancer. Thus, an assessment as described here, during and after treatment for all patients, is warranted. Patients are now being seen earlier as a part of the prehabilitation program, which facilitates access to complete decongestive treatment at an earlier stage, improves patient outcomes, and increases patient satisfaction with their treatment. Our prehabilitation program could serve as a model for other community hospital centers in achieving outcomes that are as good as those in academic centers.
Acknowledgments
The authors thank Irene Kadota and Heather Peters, from the Department of Radiation Oncology, and Julianne Courtenay, from the Department of Physical Therapy at the Disney Family Cancer Center, Burbank, California, for providing the original clinical data for analysis.
1. The National Lymphedema Medical Advisory Committee. The diagnosis and treatment of lymphedema. National Lymphedema Network. http://www.lymphnet.org/pdfDocs/nlntreatment.pdf. Updated February 2011. Accessed April 26, 2017.
2. McGarvey AC, Osmotherly PG, Hoffman GR, Chiarelli PE. Lymphedema following treatment for head and neck cancer: impact on patients, and beliefs of health professionals. Eur J Cancer Care (Engl). 2014;23(3):317-327.
3. Bhattacharyya N, Abemayor E. Patterns of hospital utilization for head and neck cancer care: changing demographics. JAMA Otolaryngol Head Neck Surg. 2015;141(4):307-312.
4. Deng J, Ridner SH, Dietrich MS, et al. Prevalence of secondary lymphedema in patients with head and neck cancer. J Pain Symptom Manage. 2012;43(2):244-252.
5. Deng J, Ridner SH, Dietrich MS, et al. Factors associated with external and internal lymphedema in patients with head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2012;84(3):e319-328.
6. Flores AM, Spinelli BA, Eden MM, Galantino ML. EDGE task force on head and neck cancer outcomes: a systematic review of outcomes measures for quantifying external lymphedema. Rehabil Oncol. 2015;33(2):15-23.
7. Ridner SH, Doersam J, Galford E. An update on lymphedema of the head and neck. http://www.lymphnet.org/pdfDocs/Vol_28-N2_Update_HN.pdf. Published April-June 2015. Accessed April 26, 2017.
8. Smith BG, Hutcheson KA, Little LG, et al. Lymphedema outcomes in patients with head and neck cancer. Otolaryngol Head Neck Surg. 2015;152(2);284-291.
9. Naqvi SHS, Karni RJ, Tan IC, et al. Int J Rad Oncol Biol Phys. 2016;4:927-928.
10. Deng J, Ridner SH, Aulino JM, Murphy BA. Assessment and measurement of head and neck lymphedema: state-of-the-science and future directions. Oral Oncol. 2015; 51(5):431-437.
11. Purcell A. Head and neck lymphedema management practices. J Lymphedema. 2013;8(2):8-15.
12. Paskett ED, Dean JA, Oliveri JM, Harrop JP. Cancer-related lymphedema risk factors, diagnosis treatment and impact: a review. J Clinl Oncol. 2012;30(30):3726-3733.
13. Quick DASH questionnaire. http://www.dash.iwh.on.ca/about-quickdash. [Last update not stated.] Accessed May 18, 2017.
14. Neck Disability Index questionnaire. www.aaos.org/uploadedFiles/NDI.pdf Accessed May 18, 2017.
15. Care Connections questionnaire. http://www.careconnections.com/. Accessed May 18, 2017.
16. Galantino ML, Eden MM, Spinelli BA, Flores AM. EDGE task force on head and neck cancer outcomes a systematic review of outcome measures for temporomandibular-related dysfunction. Rehabil Oncol. 2015;33(1):6-14.
17. Deng J, Ridner SH, Murphy BA. Lymphedema in patients with head and neck cancer. Oncol Nurs Forum. 2011;38(1):e1-e10.
18. What are oral cavity and oropharyngeal cancers? American Cancer Society. http://www.cancer.org/cancer/oralcavityandoropharyngealcancer/detailedguide/oral-cavity-and-oropharyngeal-cancer-what-is-oral-cavity-cancer. Last revised August 8, 2016. Accessed April 26, 2017.
19. Hamoir M, Schmitz S, Gregoire V. The role of neck dissection in squamous cell carcinoma of the head and neck. Curr Treat Options Oncol. 2014;15:611-624.
1. The National Lymphedema Medical Advisory Committee. The diagnosis and treatment of lymphedema. National Lymphedema Network. http://www.lymphnet.org/pdfDocs/nlntreatment.pdf. Updated February 2011. Accessed April 26, 2017.
2. McGarvey AC, Osmotherly PG, Hoffman GR, Chiarelli PE. Lymphedema following treatment for head and neck cancer: impact on patients, and beliefs of health professionals. Eur J Cancer Care (Engl). 2014;23(3):317-327.
3. Bhattacharyya N, Abemayor E. Patterns of hospital utilization for head and neck cancer care: changing demographics. JAMA Otolaryngol Head Neck Surg. 2015;141(4):307-312.
4. Deng J, Ridner SH, Dietrich MS, et al. Prevalence of secondary lymphedema in patients with head and neck cancer. J Pain Symptom Manage. 2012;43(2):244-252.
5. Deng J, Ridner SH, Dietrich MS, et al. Factors associated with external and internal lymphedema in patients with head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2012;84(3):e319-328.
6. Flores AM, Spinelli BA, Eden MM, Galantino ML. EDGE task force on head and neck cancer outcomes: a systematic review of outcomes measures for quantifying external lymphedema. Rehabil Oncol. 2015;33(2):15-23.
7. Ridner SH, Doersam J, Galford E. An update on lymphedema of the head and neck. http://www.lymphnet.org/pdfDocs/Vol_28-N2_Update_HN.pdf. Published April-June 2015. Accessed April 26, 2017.
8. Smith BG, Hutcheson KA, Little LG, et al. Lymphedema outcomes in patients with head and neck cancer. Otolaryngol Head Neck Surg. 2015;152(2);284-291.
9. Naqvi SHS, Karni RJ, Tan IC, et al. Int J Rad Oncol Biol Phys. 2016;4:927-928.
10. Deng J, Ridner SH, Aulino JM, Murphy BA. Assessment and measurement of head and neck lymphedema: state-of-the-science and future directions. Oral Oncol. 2015; 51(5):431-437.
11. Purcell A. Head and neck lymphedema management practices. J Lymphedema. 2013;8(2):8-15.
12. Paskett ED, Dean JA, Oliveri JM, Harrop JP. Cancer-related lymphedema risk factors, diagnosis treatment and impact: a review. J Clinl Oncol. 2012;30(30):3726-3733.
13. Quick DASH questionnaire. http://www.dash.iwh.on.ca/about-quickdash. [Last update not stated.] Accessed May 18, 2017.
14. Neck Disability Index questionnaire. www.aaos.org/uploadedFiles/NDI.pdf Accessed May 18, 2017.
15. Care Connections questionnaire. http://www.careconnections.com/. Accessed May 18, 2017.
16. Galantino ML, Eden MM, Spinelli BA, Flores AM. EDGE task force on head and neck cancer outcomes a systematic review of outcome measures for temporomandibular-related dysfunction. Rehabil Oncol. 2015;33(1):6-14.
17. Deng J, Ridner SH, Murphy BA. Lymphedema in patients with head and neck cancer. Oncol Nurs Forum. 2011;38(1):e1-e10.
18. What are oral cavity and oropharyngeal cancers? American Cancer Society. http://www.cancer.org/cancer/oralcavityandoropharyngealcancer/detailedguide/oral-cavity-and-oropharyngeal-cancer-what-is-oral-cavity-cancer. Last revised August 8, 2016. Accessed April 26, 2017.
19. Hamoir M, Schmitz S, Gregoire V. The role of neck dissection in squamous cell carcinoma of the head and neck. Curr Treat Options Oncol. 2014;15:611-624.
VIDEO: Routine genomic testing identifies actionable alterations in 52% of tumors
CHICAGO –
Molecular profiling, including genetic sequencing and copy number variation analysis, was performed in 1944 tumors from patients with advanced tumors enrolled in the profiLER study. Of the tumors screened, mutations deemed actionable were identified in 1,004 (52%), with 394 patients having two or more actionable targets, and the remainder having one identified targeted treatment. A molecular targeted treatment was recommended for 676 patients (35% of those tested).
“We showed that the patients who did receive the molecular targeted agents were doing better in terms of overall survival,” said Olivier Tredan, MD, PhD, the study’s lead investigator. Noting that these are trends as the trial was not randomized, he reported that the overall survival (OS) for those receiving targeted treatments was 53.7% at 3 years, compared with 46.1% for those who did not receive targeted treatment. The trend continued out to 5 years, with the OS for the targeted treatment group at 34.8%, compared with 28.1% OS for those who did not receive targeted treatment, he said at the annual meeting of the American Society of Clinical Oncology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Many patients either were too sick to receive the recommended treatment or died before they could be treated, Dr. Tredan said in a video interview.
Of the patients who did receive targeted treatment, over 60% received mTOR inhibitors. The next most common therapies were multitarget tyrosine kinase receptor (TKR)–inhibiting/antiangiogenic therapies, received by about one-third of patients. Fewer than one in five patients received any other therapies. Tumor types were colorectal, gynecological, breast, head and neck carcinomas, sarcomas, and brain tumors.
A new randomized clinical study, profiLER 2, is planned. The new study will pit a 315-gene commercial test against the 69-gene test used in profiLER 1, to see whether casting a wider net yields more targets for therapy.
Still, knowing that a treatment might help is useful only if the patient can actually receive the drug, said Dr. Tredan. “What we want is more molecular targeted agent initiation, so we need to have larger screening programs, but we need also to have access to novel targeted agents.”
Dr. Tredan reported financial relationships with Bayer, GlaxoSmithKline, and Novartis.
[email protected]
On Twitter @karioakes
CHICAGO –
Molecular profiling, including genetic sequencing and copy number variation analysis, was performed in 1944 tumors from patients with advanced tumors enrolled in the profiLER study. Of the tumors screened, mutations deemed actionable were identified in 1,004 (52%), with 394 patients having two or more actionable targets, and the remainder having one identified targeted treatment. A molecular targeted treatment was recommended for 676 patients (35% of those tested).
“We showed that the patients who did receive the molecular targeted agents were doing better in terms of overall survival,” said Olivier Tredan, MD, PhD, the study’s lead investigator. Noting that these are trends as the trial was not randomized, he reported that the overall survival (OS) for those receiving targeted treatments was 53.7% at 3 years, compared with 46.1% for those who did not receive targeted treatment. The trend continued out to 5 years, with the OS for the targeted treatment group at 34.8%, compared with 28.1% OS for those who did not receive targeted treatment, he said at the annual meeting of the American Society of Clinical Oncology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Many patients either were too sick to receive the recommended treatment or died before they could be treated, Dr. Tredan said in a video interview.
Of the patients who did receive targeted treatment, over 60% received mTOR inhibitors. The next most common therapies were multitarget tyrosine kinase receptor (TKR)–inhibiting/antiangiogenic therapies, received by about one-third of patients. Fewer than one in five patients received any other therapies. Tumor types were colorectal, gynecological, breast, head and neck carcinomas, sarcomas, and brain tumors.
A new randomized clinical study, profiLER 2, is planned. The new study will pit a 315-gene commercial test against the 69-gene test used in profiLER 1, to see whether casting a wider net yields more targets for therapy.
Still, knowing that a treatment might help is useful only if the patient can actually receive the drug, said Dr. Tredan. “What we want is more molecular targeted agent initiation, so we need to have larger screening programs, but we need also to have access to novel targeted agents.”
Dr. Tredan reported financial relationships with Bayer, GlaxoSmithKline, and Novartis.
[email protected]
On Twitter @karioakes
CHICAGO –
Molecular profiling, including genetic sequencing and copy number variation analysis, was performed in 1944 tumors from patients with advanced tumors enrolled in the profiLER study. Of the tumors screened, mutations deemed actionable were identified in 1,004 (52%), with 394 patients having two or more actionable targets, and the remainder having one identified targeted treatment. A molecular targeted treatment was recommended for 676 patients (35% of those tested).
“We showed that the patients who did receive the molecular targeted agents were doing better in terms of overall survival,” said Olivier Tredan, MD, PhD, the study’s lead investigator. Noting that these are trends as the trial was not randomized, he reported that the overall survival (OS) for those receiving targeted treatments was 53.7% at 3 years, compared with 46.1% for those who did not receive targeted treatment. The trend continued out to 5 years, with the OS for the targeted treatment group at 34.8%, compared with 28.1% OS for those who did not receive targeted treatment, he said at the annual meeting of the American Society of Clinical Oncology.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Many patients either were too sick to receive the recommended treatment or died before they could be treated, Dr. Tredan said in a video interview.
Of the patients who did receive targeted treatment, over 60% received mTOR inhibitors. The next most common therapies were multitarget tyrosine kinase receptor (TKR)–inhibiting/antiangiogenic therapies, received by about one-third of patients. Fewer than one in five patients received any other therapies. Tumor types were colorectal, gynecological, breast, head and neck carcinomas, sarcomas, and brain tumors.
A new randomized clinical study, profiLER 2, is planned. The new study will pit a 315-gene commercial test against the 69-gene test used in profiLER 1, to see whether casting a wider net yields more targets for therapy.
Still, knowing that a treatment might help is useful only if the patient can actually receive the drug, said Dr. Tredan. “What we want is more molecular targeted agent initiation, so we need to have larger screening programs, but we need also to have access to novel targeted agents.”
Dr. Tredan reported financial relationships with Bayer, GlaxoSmithKline, and Novartis.
[email protected]
On Twitter @karioakes
AT ASCO 2017
Time-to-Treatment Predicts Oral Cancer Survival
Oral cancer has a low 5-year survival rate compared with other major types of cancer, and the rate hasn’t improved much in recent years, say researchers from China Medical University, Asia University, Taichung Veterans General Hospital, and National Yang-Ming University; all in Taichung, Taiwan. That may be due in part to delay in treatment, they say. Their analysis of data of 21,263 patients with oral cavity squamous cell carcinoma (OCSCC) bears out their theory.
Related: Predicting Tongue Cancer Recurrence
About 40% of the patients presented with stage IV disease. More than one-third had tongue cancer. The average time from diagnosis to treatment was 24 days. Most patients (86%) were treated within 30 days of diagnosis; 12% were treated between 31 and 120 days after diagnosis, and 2.7% received treatment 120 days after diagnosis. Surgery was usually the first treatment.
The time interval from diagnosis to treatment (the phrase the researchers prefer to “treatment delay” or “wait time”) was an independent prognostic factor in OCSCC patients. The average interval was 24 days. Those treated within 30 days tended to have a higher survival rate. Treatment after 120 days from diagnosis increased the risk of death by 1.32-fold.
The average follow-up period was 44 months. When the researchers stratified patients according to the time between diagnosis and treatment, they found patients aged ≥ 65 years or who had advanced cancer were more likely to be treated later. Patients treated initially with radiotherapy or chemotherapy were more likely to have a longer mean time interval when compared with those who were treated first with surgery.
Related: Shorter Length of Stay May Not Mean Higher Readmission Rates
The researchers also found that patients treated in private hospitals had a shorter time interval compared with those treated in public hospitals (although the latter were more likely to survive). Patients who received treatment in hospitals with a low- to medium-service volume had a longer interval compared with those treated in hospitals with a high-service volume. Other predictors of longer survival included being female, younger, primary tumor site at the tongue, and earlier stage disease.
The researchers cite a study that found the median duration of clinical upstaging from early to late stage was 11.3 months, whereas the average period from advanced tumor to untreatable tumor was 3.8 months. That might explain why they found that the longer delay to treatment increased the risk of death, they suggest. The researchers also point to reasons such as pending second opinions, shortages of therapeutic instruments and manpower, and lack of public awareness.
Related: IBD and the Risk of Oral Cancer
All told however, the researchers conclude, the reasons for the increased time interval from diagnosis to treatment of OCSCC patients remain “multifaceted, integrated, and poorly understood.”
Source:
Tsai WC, Kung PT, Wang YH, Huang KH, Liu SA. PLoS One. 2017;12(4):e0175148.
doi: 10.1371/journal.pone.0175148.
Oral cancer has a low 5-year survival rate compared with other major types of cancer, and the rate hasn’t improved much in recent years, say researchers from China Medical University, Asia University, Taichung Veterans General Hospital, and National Yang-Ming University; all in Taichung, Taiwan. That may be due in part to delay in treatment, they say. Their analysis of data of 21,263 patients with oral cavity squamous cell carcinoma (OCSCC) bears out their theory.
Related: Predicting Tongue Cancer Recurrence
About 40% of the patients presented with stage IV disease. More than one-third had tongue cancer. The average time from diagnosis to treatment was 24 days. Most patients (86%) were treated within 30 days of diagnosis; 12% were treated between 31 and 120 days after diagnosis, and 2.7% received treatment 120 days after diagnosis. Surgery was usually the first treatment.
The time interval from diagnosis to treatment (the phrase the researchers prefer to “treatment delay” or “wait time”) was an independent prognostic factor in OCSCC patients. The average interval was 24 days. Those treated within 30 days tended to have a higher survival rate. Treatment after 120 days from diagnosis increased the risk of death by 1.32-fold.
The average follow-up period was 44 months. When the researchers stratified patients according to the time between diagnosis and treatment, they found patients aged ≥ 65 years or who had advanced cancer were more likely to be treated later. Patients treated initially with radiotherapy or chemotherapy were more likely to have a longer mean time interval when compared with those who were treated first with surgery.
Related: Shorter Length of Stay May Not Mean Higher Readmission Rates
The researchers also found that patients treated in private hospitals had a shorter time interval compared with those treated in public hospitals (although the latter were more likely to survive). Patients who received treatment in hospitals with a low- to medium-service volume had a longer interval compared with those treated in hospitals with a high-service volume. Other predictors of longer survival included being female, younger, primary tumor site at the tongue, and earlier stage disease.
The researchers cite a study that found the median duration of clinical upstaging from early to late stage was 11.3 months, whereas the average period from advanced tumor to untreatable tumor was 3.8 months. That might explain why they found that the longer delay to treatment increased the risk of death, they suggest. The researchers also point to reasons such as pending second opinions, shortages of therapeutic instruments and manpower, and lack of public awareness.
Related: IBD and the Risk of Oral Cancer
All told however, the researchers conclude, the reasons for the increased time interval from diagnosis to treatment of OCSCC patients remain “multifaceted, integrated, and poorly understood.”
Source:
Tsai WC, Kung PT, Wang YH, Huang KH, Liu SA. PLoS One. 2017;12(4):e0175148.
doi: 10.1371/journal.pone.0175148.
Oral cancer has a low 5-year survival rate compared with other major types of cancer, and the rate hasn’t improved much in recent years, say researchers from China Medical University, Asia University, Taichung Veterans General Hospital, and National Yang-Ming University; all in Taichung, Taiwan. That may be due in part to delay in treatment, they say. Their analysis of data of 21,263 patients with oral cavity squamous cell carcinoma (OCSCC) bears out their theory.
Related: Predicting Tongue Cancer Recurrence
About 40% of the patients presented with stage IV disease. More than one-third had tongue cancer. The average time from diagnosis to treatment was 24 days. Most patients (86%) were treated within 30 days of diagnosis; 12% were treated between 31 and 120 days after diagnosis, and 2.7% received treatment 120 days after diagnosis. Surgery was usually the first treatment.
The time interval from diagnosis to treatment (the phrase the researchers prefer to “treatment delay” or “wait time”) was an independent prognostic factor in OCSCC patients. The average interval was 24 days. Those treated within 30 days tended to have a higher survival rate. Treatment after 120 days from diagnosis increased the risk of death by 1.32-fold.
The average follow-up period was 44 months. When the researchers stratified patients according to the time between diagnosis and treatment, they found patients aged ≥ 65 years or who had advanced cancer were more likely to be treated later. Patients treated initially with radiotherapy or chemotherapy were more likely to have a longer mean time interval when compared with those who were treated first with surgery.
Related: Shorter Length of Stay May Not Mean Higher Readmission Rates
The researchers also found that patients treated in private hospitals had a shorter time interval compared with those treated in public hospitals (although the latter were more likely to survive). Patients who received treatment in hospitals with a low- to medium-service volume had a longer interval compared with those treated in hospitals with a high-service volume. Other predictors of longer survival included being female, younger, primary tumor site at the tongue, and earlier stage disease.
The researchers cite a study that found the median duration of clinical upstaging from early to late stage was 11.3 months, whereas the average period from advanced tumor to untreatable tumor was 3.8 months. That might explain why they found that the longer delay to treatment increased the risk of death, they suggest. The researchers also point to reasons such as pending second opinions, shortages of therapeutic instruments and manpower, and lack of public awareness.
Related: IBD and the Risk of Oral Cancer
All told however, the researchers conclude, the reasons for the increased time interval from diagnosis to treatment of OCSCC patients remain “multifaceted, integrated, and poorly understood.”
Source:
Tsai WC, Kung PT, Wang YH, Huang KH, Liu SA. PLoS One. 2017;12(4):e0175148.
doi: 10.1371/journal.pone.0175148.