User login
POLST: An improvement over traditional advance directives
An 89-year-old woman with advanced dementia is living in a nursing home and is fully dependent in all aspects of personal care, including feeding. She has a health care proxy and a living will.
Her husband is her health care agent and has established that the primary goal of her care should be to keep her comfortable. He has repeatedly discussed this goal with her attending physician and the nursing-home staff and has reiterated that when his wife had capacity, she wanted “no heroics,” “no feeding tube,” and no life-sustaining treatment that would prolong her dying. He has requested that she not be transferred to the hospital and that she receive all further care at the nursing home. These preferences are consistent with her living will.
One evening, she becomes somnolent and febrile, with rapid breathing. The physician covering for the attending physician does not know the patient, cannot reach her husband, and sends her to the hospital, where she is admitted with aspiration pneumonia.
Her level of alertness improves with hydration. However, the hospital nurses have a difficult time feeding her. She does not seem to want to eat, “pockets” food in her cheeks, is slow to swallow, and sometimes coughs during feeding. This is nothing new—at the nursing home, her feeding pattern had been the same for nearly 6 months. During this time she always had a cough; fevers came and went. She has slowly lost weight; she now weighs 100 lb (45 kg), down 30 lb (14 kg) in 3 years.
With treatment, her respiratory distress and fever resolve. The physician orders a swallowing evaluation by a speech therapist, who determines that she needs a feeding tube. After that, a meeting is scheduled with her husband and physician to discuss the speech therapist’s assessment. The patient’s husband emphatically refuses the feeding tube and is upset that she was transferred to the hospital against his expressed wishes.
Why did this happen?
TRADITIONAL ADVANCE DIRECTIVES ARE OFTEN NOT ENOUGH
Even when patients fill out advance directives in accordance with state law, their preferences for care at the end of life are not consistently followed.
Problems with living wills
Living wills state patients’ wishes about medical care in the event that they develop an irreversible condition that prevents them from making their own medical decisions. The living will becomes effective if they become terminally ill, permanently unconscious, or minimally conscious due to brain damage and will never regain the ability to make decisions. People who want to indicate under what set of circumstances they favor or object to receiving any specific treatments use a living will.
The Patient Self-Determination Act of 1990 states that on admission to a hospital or nursing home, patients have to be informed of their rights, including the right to accept or refuse treatment.1 However, the current system of communicating wishes about end-of-life care using solely traditional advance directives such as the living will has proven insufficient. This is because traditional advance directives, being general statements of patients’ preferences, need to be carried out through specifications in medical orders when the need arises.2
Further, traditional advance directives require patients to recognize the importance of advance care planning, understand medical interventions, evaluate their own personal values and beliefs, and communicate their wishes to their agents, loved ones, physicians, and health care providers. Moreover, these documents apply to future circumstances, require further interpretation by the agent and health care professionals, and do not result in actionable medical orders. Decisions about care depend on interpreting earlier conversations, the physician’s estimates of prognosis, and, possibly, the personal convictions of the physician, agent, and loved ones, even though ethically, all involved need to focus on the patient’s stated wishes or best interest. A living will does not help clarify the patient’s wishes in the absence of antecedent conversation with the family, close friends, and the patient’s personal physician. And living wills cannot be read and interpreted in an emergency.
The situation is further complicated by difficulty in defining “terminal” or “irreversible” conditions and accounting for the different perspective that physicians, agents, and loved ones bring to the situation. For example, imagine a patient with dementia nearing the end of life who eats less, has difficulty managing secretions, aspirates, and develops pneumonia. While end-stage dementia is terminal, pneumonia may be reversible.
Increasingly, therefore, people are being counseled to appoint a health care agent (see below).3
The importance of a health care proxy (durable power of attorney for health care)
In a health care proxy document (also known as durable power of attorney for health care), the patient names a health care agent. This person has authority to make decisions about the patient’s medical care, including life-sustaining treatment. In other words, you the patient appoint someone to speak for you in the event you are unable to make your own medical decisions (not only at the end of life).
Since anyone may face a sudden and unexpected acute illness or injury with the risk of becoming incapacitated and unable to make medical decisions, everyone age 18 and older should be encouraged to complete a health care proxy document and to engage in advance care planning discussions with family and loved ones. Physicians can initiate this process as a wellness initiative and can help patients and families understand advance care planning. In all health care settings, trained and qualified health care professionals can provide education on advance care planning to patients, families, and loved ones.
A key issue when naming a health care agent is choosing the right one, someone who will make decisions in accordance with the person’s current values and beliefs and who can separate his or her personal values from the patient’s values. Another key issue: people need to have proactive discussions about their personal values, beliefs, and goals of care, which many are reluctant to do, and the health care agent must be willing to talk about sensitive issues ahead of time. Even when a health care agent is available in an emergency, emergency medical services personnel cannot follow directions from a health care agent. Most importantly, a health care agent must be able to handle potential conflicts between family and providers.
POLST ENSURES PATIENT PREFERENCES ARE HONORED AT THE END OF LIFE
Approximately 20 years ago, a team of health care professionals at the University of Oregon recognized these problems and realized that physicians needed to be more involved in discussions with patients about end-of-life care and in translating the patient’s preferences and values into concrete medical orders. The result was the Physician Orders for Life-Sustaining Treatment (POLST) Paradigm Program.4
What is POLST?
POLST is an end-of-life-care transitions program that focuses on patient-centered goals for care and shared informed medical decision-making.5,6 It offers a mechanism to communicate the wishes of seriously ill patients to have or to limit medical treatment as they move from one care setting to another. Table 1 lists the differences between traditional advance directives and POLST.
The aim is to improve the quality of care that seriously ill patients receive at the end of life. POLST is based on effective communication of the patient’s wishes, with actionable medical orders documented on a brightly colored form (www.ohsu.edu/polst/programs/sample-forms.htm; Figure 1) and a promise by health care professionals to honor these wishes.7 Key features of the program include education, training, and a quality-improvement process.
Who is POLST for?
POLST is for patients with serious life-limiting illness who have a life expectancy of less than 1 year, or anyone of advanced age interested in defining their end-of-life care wishes. Qualified and trained health care professionals (physicians, physician’s assistants, nurse practitioners, and social workers) participate in discussions leading to the completion of a POLST form in all settings, particularly along the long-term care continuum and for home hospice.
The key element of the POLST process: Shared, informed medical decision-making
Health care professionals working as an interdisciplinary team play a key role in educating patients and their families about advance care planning and shared, informed medical decision-making, as well as in resolving conflict. To be effective, shared medical decision-making must be well-informed. The decision-maker (patient, health care agent, or surrogate) must weigh the following questions (Table 2):
- Will treatment make a difference?
- Do the burdens of treatment outweigh its benefits?
- Is there hope of recovery? If so, what will life be like afterward?
- What does the patient value? What is the patient’s goal for his or her care?
In-depth discussions with patients, family members, and surrogates are needed, and these people are often reluctant to ask these questions and afraid to discuss the dying process. Even if they are informed of their diagnosis and prognosis, they may not know what they mean in terms of their everyday experience and future.
Health care professionals engaging in these conversations can use the eight-step POLST protocol (Table 3) to elicit their preferences at the end of life. Table 4 lists tools and resources to enhance the understanding of advance care planning and POLST.
What does the POLST form cover?
The POLST form (Figure 1) provides instructions about resuscitation if the patient has no pulse and is not breathing. Additionally, the medical orders indicate decisions about the level of medical intervention that the patient wants or does not want, eg, intubation, mechanical ventilation, transport to the hospital, intensive care, artificial nutrition and hydration, and antibiotics.
Thus, POLST is outcome-neutral and can be used either to limit medical interventions or to clarify a request for any or all medically indicated treatments.
Both the practitioner and the patient or patient’s surrogate sign the form. The original goes into the patient’s chart, and a copy should accompany the patient if he or she is transferred or discharged. Additionally, if the state has a POLST registry, the POLST information should be entered into the registry.
POLST is expanding across the country
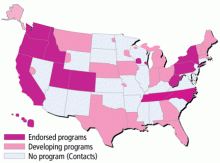
The use of POLST has been expanding across the United States, with POLST programs now implemented in all or part of at least 30 states. There are endorsed programs in 14 states, and programs are being developed in 26 more. Requirements for endorsement are found at www.polst.org. Figure 2 shows the status of POLST in the 50 states.
Oregon’s POLST form is the original model for other forms designed to meet specific legislative or regulatory requirements in other states. POLST-like programs are known by different names in different states: eg, New York’s Medical Orders for Life-Sustaining Treatment (MOLST) and West Virginia’s Physicians Orders for Scope of Treatment (POST), but all endorsed programs share common core elements.
POLST research
A number of studies in the past 10 years have shown that POLST improves the documentation and honoring of patient preferences, whatever they may be.4,8–16
Emergency medical technicians in Oregon reported that the POLST form provides clear instructions about patient preferences and is useful when deciding which treatments to provide. In contrast to the single-intervention focus of out-of-hospital do-not-resuscitate orders, the POLST form provides patients the opportunity to document treatment goals and preferences for interventions across a range of treatment options, thus permitting greater individualization.13
Comfort care is not sacrificed if a POLST document is in place. Most hospice patients choose at least one life-sustaining treatment on their POLST form.14
In a multistate study published in 2010, the medical records of residents in 90 randomly chosen Medicaid-eligible nursing homes were reviewed.15 POLST was compared with traditional advance care planning in terms of the effect on the presence of medical orders reflecting treatment preferences, symptom management, and use of life-sustaining treatments. The study found that residents with POLST forms had significantly more medical orders about life-sustaining treatments than residents with traditional advance directives. There were no differences between residents with or without POLST forms on symptom assessment or management measures. POLST was more effective than traditional advance planning at limiting unwanted life-sustaining treatments. The study suggests that POLST offers significant advantages over traditional advance directives in nursing facilities.15,16
In summary, more than a decade of research has shown that the POLST Paradigm Program serves as an emerging national model for implementing shared, informed medical decision-making. Furthermore, POLST more accurately conveys end-of-life care preferences for patients with advanced chronic illness and for dying patients than traditional advance directives and yields higher adherence by medical professionals.
CLINICAL CASE REVISITED
Let’s consider if the physician for our 89-year-old woman with dementia had completed a POLST form with orders indicating “do not attempt resuscitation (DNR/no CPR)” and “comfort measures only, do not transfer to hospital for life-sustaining treatment and transfer if comfort needs cannot be met in current location.”
The patient’s respiratory distress and fever would have been treated at her nursing home with medication and oxygen. She would have been transferred to the hospital only if her comfort needs would not have been met at the nursing home. Unwanted life-sustaining treatment would have been avoided. The wishes of the patient, based on her values and careful consideration of options, would have been respected.
- Dunn PM, Tolle SW, Moss AH, Black JS. The POLST paradigm: respecting the wishes of patients and families. Ann Long-Term Care 2007; 15:33–40.
- Patient Self-Determination Act of 1990. Pub. L. No. 101-508, ss 4206, 104 Stat. 1388.
- Bomba PA, Sabatino CP. POLST: an emerging model for end-of-life care planning. The ElderLaw Report 2009; 20:1–5.
- Karp Sabatino C. AARP Public Policy Institute, Improving advance illness care: the evolution of state POLST programs 2011. http://assets.aarp.org/rgcenter/ppi/cons-prot/POLST-Report-04-11.pdf. Accessed May 30, 2012.
- Bomba PA. Discussing patient p and end of life care, Journal of the Monroe County Medical Society, 7th District Branch, MSSNY 2011;12–15. www.compassionandsupport.org/index.php/research_/. Accessed May 30, 2012.
- Citko J, Moss AH, Carley M, Tolle SW. The National POLST Paradigm Initiative, 2ND ed. Fast Facts and Concepts 2010;178. www.eperc.mcw.edu/fastfact/ff_178.htm. Accessed May 30, 2012.
- Center for Ethics in Health Care, Oregon Health & Science University. www.ohsu.edu/polst/. Accessed May 30, 2012.
- Lee MA, Brummel-Smith K, Meyer J, Drew N, London MR. Physician orders for life-sustaining treatment (POLST): outcomes in a PACE program. Program of All-Inclusive Care for the Elderly. J Am Geriatr Soc 2000; 48:1219–1225.
- Meyers JL, Moore C, McGrory A, Sparr J, Ahern M. Physician orders for life-sustaining treatment form: honoring end-of-life directives for nursing home residents. J Gerontol Nurs 2004; 30:37–46.
- Dunn PM, Schmidt TA, Carley MM, Donius M, Weinstein MA, Dull VT. A method to communicate patient p about medically indicated life-sustaining treatment in the out-of-hospital setting. J Am Geriatr Soc 1996; 44:785–791.
- Cantor MD. Improving advance care planning: lessons from POLST. Physician Orders for Life-Sustaining Treatment (comment). J Am Geriatr Soc 2000; 48:1343–1344.
- Tolle SW, Tilden VP, Nelson CA, Dunn PM. A prospective study of the efficacy of the physician order form for life-sustaining treatment. J Am Geriatr Soc 1998; 46:1097–1102.
- Schmidt TA, Hickman SE, Tolle SW, Brooks HS. The Physician Orders for Life-Sustaining Treatment program: Oregon emergency medical technicians’ practical experiences and attitudes. J Am Geriatr Soc 2004; 52:1430–1434.
- Hickman SE, Nelson CA, Moss AH, et al. Use of the Physician Orders for Life-Sustaining Treatment (POLST) paradigm program in the hospice setting. J Palliat Med 2009; 12:133–141.
- Hickman SE, Nelson CA, Perrin NA, Moss AH, Hammes BJ, Tolle SW. A comparison of methods to communicate treatment p in nursing facilities: traditional practices versus the physician orders for life-sustaining treatment program. J Am Geriatr Soc 2010; 58:1241–1248.
- Hickman SE, Nelson CA, Moss AH, Tolle SW, Perrin NA, Hammes BJ. The consistency between treatments provided to nursing facility residents and orders on the physician orders for life-sustaining treatment form. J Am Geriatr Soc 2011; 59:2091–2099.
An 89-year-old woman with advanced dementia is living in a nursing home and is fully dependent in all aspects of personal care, including feeding. She has a health care proxy and a living will.
Her husband is her health care agent and has established that the primary goal of her care should be to keep her comfortable. He has repeatedly discussed this goal with her attending physician and the nursing-home staff and has reiterated that when his wife had capacity, she wanted “no heroics,” “no feeding tube,” and no life-sustaining treatment that would prolong her dying. He has requested that she not be transferred to the hospital and that she receive all further care at the nursing home. These preferences are consistent with her living will.
One evening, she becomes somnolent and febrile, with rapid breathing. The physician covering for the attending physician does not know the patient, cannot reach her husband, and sends her to the hospital, where she is admitted with aspiration pneumonia.
Her level of alertness improves with hydration. However, the hospital nurses have a difficult time feeding her. She does not seem to want to eat, “pockets” food in her cheeks, is slow to swallow, and sometimes coughs during feeding. This is nothing new—at the nursing home, her feeding pattern had been the same for nearly 6 months. During this time she always had a cough; fevers came and went. She has slowly lost weight; she now weighs 100 lb (45 kg), down 30 lb (14 kg) in 3 years.
With treatment, her respiratory distress and fever resolve. The physician orders a swallowing evaluation by a speech therapist, who determines that she needs a feeding tube. After that, a meeting is scheduled with her husband and physician to discuss the speech therapist’s assessment. The patient’s husband emphatically refuses the feeding tube and is upset that she was transferred to the hospital against his expressed wishes.
Why did this happen?
TRADITIONAL ADVANCE DIRECTIVES ARE OFTEN NOT ENOUGH
Even when patients fill out advance directives in accordance with state law, their preferences for care at the end of life are not consistently followed.
Problems with living wills
Living wills state patients’ wishes about medical care in the event that they develop an irreversible condition that prevents them from making their own medical decisions. The living will becomes effective if they become terminally ill, permanently unconscious, or minimally conscious due to brain damage and will never regain the ability to make decisions. People who want to indicate under what set of circumstances they favor or object to receiving any specific treatments use a living will.
The Patient Self-Determination Act of 1990 states that on admission to a hospital or nursing home, patients have to be informed of their rights, including the right to accept or refuse treatment.1 However, the current system of communicating wishes about end-of-life care using solely traditional advance directives such as the living will has proven insufficient. This is because traditional advance directives, being general statements of patients’ preferences, need to be carried out through specifications in medical orders when the need arises.2
Further, traditional advance directives require patients to recognize the importance of advance care planning, understand medical interventions, evaluate their own personal values and beliefs, and communicate their wishes to their agents, loved ones, physicians, and health care providers. Moreover, these documents apply to future circumstances, require further interpretation by the agent and health care professionals, and do not result in actionable medical orders. Decisions about care depend on interpreting earlier conversations, the physician’s estimates of prognosis, and, possibly, the personal convictions of the physician, agent, and loved ones, even though ethically, all involved need to focus on the patient’s stated wishes or best interest. A living will does not help clarify the patient’s wishes in the absence of antecedent conversation with the family, close friends, and the patient’s personal physician. And living wills cannot be read and interpreted in an emergency.
The situation is further complicated by difficulty in defining “terminal” or “irreversible” conditions and accounting for the different perspective that physicians, agents, and loved ones bring to the situation. For example, imagine a patient with dementia nearing the end of life who eats less, has difficulty managing secretions, aspirates, and develops pneumonia. While end-stage dementia is terminal, pneumonia may be reversible.
Increasingly, therefore, people are being counseled to appoint a health care agent (see below).3
The importance of a health care proxy (durable power of attorney for health care)
In a health care proxy document (also known as durable power of attorney for health care), the patient names a health care agent. This person has authority to make decisions about the patient’s medical care, including life-sustaining treatment. In other words, you the patient appoint someone to speak for you in the event you are unable to make your own medical decisions (not only at the end of life).
Since anyone may face a sudden and unexpected acute illness or injury with the risk of becoming incapacitated and unable to make medical decisions, everyone age 18 and older should be encouraged to complete a health care proxy document and to engage in advance care planning discussions with family and loved ones. Physicians can initiate this process as a wellness initiative and can help patients and families understand advance care planning. In all health care settings, trained and qualified health care professionals can provide education on advance care planning to patients, families, and loved ones.
A key issue when naming a health care agent is choosing the right one, someone who will make decisions in accordance with the person’s current values and beliefs and who can separate his or her personal values from the patient’s values. Another key issue: people need to have proactive discussions about their personal values, beliefs, and goals of care, which many are reluctant to do, and the health care agent must be willing to talk about sensitive issues ahead of time. Even when a health care agent is available in an emergency, emergency medical services personnel cannot follow directions from a health care agent. Most importantly, a health care agent must be able to handle potential conflicts between family and providers.
POLST ENSURES PATIENT PREFERENCES ARE HONORED AT THE END OF LIFE
Approximately 20 years ago, a team of health care professionals at the University of Oregon recognized these problems and realized that physicians needed to be more involved in discussions with patients about end-of-life care and in translating the patient’s preferences and values into concrete medical orders. The result was the Physician Orders for Life-Sustaining Treatment (POLST) Paradigm Program.4
What is POLST?
POLST is an end-of-life-care transitions program that focuses on patient-centered goals for care and shared informed medical decision-making.5,6 It offers a mechanism to communicate the wishes of seriously ill patients to have or to limit medical treatment as they move from one care setting to another. Table 1 lists the differences between traditional advance directives and POLST.
The aim is to improve the quality of care that seriously ill patients receive at the end of life. POLST is based on effective communication of the patient’s wishes, with actionable medical orders documented on a brightly colored form (www.ohsu.edu/polst/programs/sample-forms.htm; Figure 1) and a promise by health care professionals to honor these wishes.7 Key features of the program include education, training, and a quality-improvement process.
Who is POLST for?
POLST is for patients with serious life-limiting illness who have a life expectancy of less than 1 year, or anyone of advanced age interested in defining their end-of-life care wishes. Qualified and trained health care professionals (physicians, physician’s assistants, nurse practitioners, and social workers) participate in discussions leading to the completion of a POLST form in all settings, particularly along the long-term care continuum and for home hospice.
The key element of the POLST process: Shared, informed medical decision-making
Health care professionals working as an interdisciplinary team play a key role in educating patients and their families about advance care planning and shared, informed medical decision-making, as well as in resolving conflict. To be effective, shared medical decision-making must be well-informed. The decision-maker (patient, health care agent, or surrogate) must weigh the following questions (Table 2):
- Will treatment make a difference?
- Do the burdens of treatment outweigh its benefits?
- Is there hope of recovery? If so, what will life be like afterward?
- What does the patient value? What is the patient’s goal for his or her care?
In-depth discussions with patients, family members, and surrogates are needed, and these people are often reluctant to ask these questions and afraid to discuss the dying process. Even if they are informed of their diagnosis and prognosis, they may not know what they mean in terms of their everyday experience and future.
Health care professionals engaging in these conversations can use the eight-step POLST protocol (Table 3) to elicit their preferences at the end of life. Table 4 lists tools and resources to enhance the understanding of advance care planning and POLST.
What does the POLST form cover?
The POLST form (Figure 1) provides instructions about resuscitation if the patient has no pulse and is not breathing. Additionally, the medical orders indicate decisions about the level of medical intervention that the patient wants or does not want, eg, intubation, mechanical ventilation, transport to the hospital, intensive care, artificial nutrition and hydration, and antibiotics.
Thus, POLST is outcome-neutral and can be used either to limit medical interventions or to clarify a request for any or all medically indicated treatments.
Both the practitioner and the patient or patient’s surrogate sign the form. The original goes into the patient’s chart, and a copy should accompany the patient if he or she is transferred or discharged. Additionally, if the state has a POLST registry, the POLST information should be entered into the registry.
POLST is expanding across the country
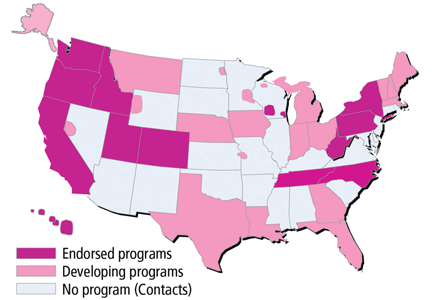
The use of POLST has been expanding across the United States, with POLST programs now implemented in all or part of at least 30 states. There are endorsed programs in 14 states, and programs are being developed in 26 more. Requirements for endorsement are found at www.polst.org. Figure 2 shows the status of POLST in the 50 states.
Oregon’s POLST form is the original model for other forms designed to meet specific legislative or regulatory requirements in other states. POLST-like programs are known by different names in different states: eg, New York’s Medical Orders for Life-Sustaining Treatment (MOLST) and West Virginia’s Physicians Orders for Scope of Treatment (POST), but all endorsed programs share common core elements.
POLST research
A number of studies in the past 10 years have shown that POLST improves the documentation and honoring of patient preferences, whatever they may be.4,8–16
Emergency medical technicians in Oregon reported that the POLST form provides clear instructions about patient preferences and is useful when deciding which treatments to provide. In contrast to the single-intervention focus of out-of-hospital do-not-resuscitate orders, the POLST form provides patients the opportunity to document treatment goals and preferences for interventions across a range of treatment options, thus permitting greater individualization.13
Comfort care is not sacrificed if a POLST document is in place. Most hospice patients choose at least one life-sustaining treatment on their POLST form.14
In a multistate study published in 2010, the medical records of residents in 90 randomly chosen Medicaid-eligible nursing homes were reviewed.15 POLST was compared with traditional advance care planning in terms of the effect on the presence of medical orders reflecting treatment preferences, symptom management, and use of life-sustaining treatments. The study found that residents with POLST forms had significantly more medical orders about life-sustaining treatments than residents with traditional advance directives. There were no differences between residents with or without POLST forms on symptom assessment or management measures. POLST was more effective than traditional advance planning at limiting unwanted life-sustaining treatments. The study suggests that POLST offers significant advantages over traditional advance directives in nursing facilities.15,16
In summary, more than a decade of research has shown that the POLST Paradigm Program serves as an emerging national model for implementing shared, informed medical decision-making. Furthermore, POLST more accurately conveys end-of-life care preferences for patients with advanced chronic illness and for dying patients than traditional advance directives and yields higher adherence by medical professionals.
CLINICAL CASE REVISITED
Let’s consider if the physician for our 89-year-old woman with dementia had completed a POLST form with orders indicating “do not attempt resuscitation (DNR/no CPR)” and “comfort measures only, do not transfer to hospital for life-sustaining treatment and transfer if comfort needs cannot be met in current location.”
The patient’s respiratory distress and fever would have been treated at her nursing home with medication and oxygen. She would have been transferred to the hospital only if her comfort needs would not have been met at the nursing home. Unwanted life-sustaining treatment would have been avoided. The wishes of the patient, based on her values and careful consideration of options, would have been respected.
An 89-year-old woman with advanced dementia is living in a nursing home and is fully dependent in all aspects of personal care, including feeding. She has a health care proxy and a living will.
Her husband is her health care agent and has established that the primary goal of her care should be to keep her comfortable. He has repeatedly discussed this goal with her attending physician and the nursing-home staff and has reiterated that when his wife had capacity, she wanted “no heroics,” “no feeding tube,” and no life-sustaining treatment that would prolong her dying. He has requested that she not be transferred to the hospital and that she receive all further care at the nursing home. These preferences are consistent with her living will.
One evening, she becomes somnolent and febrile, with rapid breathing. The physician covering for the attending physician does not know the patient, cannot reach her husband, and sends her to the hospital, where she is admitted with aspiration pneumonia.
Her level of alertness improves with hydration. However, the hospital nurses have a difficult time feeding her. She does not seem to want to eat, “pockets” food in her cheeks, is slow to swallow, and sometimes coughs during feeding. This is nothing new—at the nursing home, her feeding pattern had been the same for nearly 6 months. During this time she always had a cough; fevers came and went. She has slowly lost weight; she now weighs 100 lb (45 kg), down 30 lb (14 kg) in 3 years.
With treatment, her respiratory distress and fever resolve. The physician orders a swallowing evaluation by a speech therapist, who determines that she needs a feeding tube. After that, a meeting is scheduled with her husband and physician to discuss the speech therapist’s assessment. The patient’s husband emphatically refuses the feeding tube and is upset that she was transferred to the hospital against his expressed wishes.
Why did this happen?
TRADITIONAL ADVANCE DIRECTIVES ARE OFTEN NOT ENOUGH
Even when patients fill out advance directives in accordance with state law, their preferences for care at the end of life are not consistently followed.
Problems with living wills
Living wills state patients’ wishes about medical care in the event that they develop an irreversible condition that prevents them from making their own medical decisions. The living will becomes effective if they become terminally ill, permanently unconscious, or minimally conscious due to brain damage and will never regain the ability to make decisions. People who want to indicate under what set of circumstances they favor or object to receiving any specific treatments use a living will.
The Patient Self-Determination Act of 1990 states that on admission to a hospital or nursing home, patients have to be informed of their rights, including the right to accept or refuse treatment.1 However, the current system of communicating wishes about end-of-life care using solely traditional advance directives such as the living will has proven insufficient. This is because traditional advance directives, being general statements of patients’ preferences, need to be carried out through specifications in medical orders when the need arises.2
Further, traditional advance directives require patients to recognize the importance of advance care planning, understand medical interventions, evaluate their own personal values and beliefs, and communicate their wishes to their agents, loved ones, physicians, and health care providers. Moreover, these documents apply to future circumstances, require further interpretation by the agent and health care professionals, and do not result in actionable medical orders. Decisions about care depend on interpreting earlier conversations, the physician’s estimates of prognosis, and, possibly, the personal convictions of the physician, agent, and loved ones, even though ethically, all involved need to focus on the patient’s stated wishes or best interest. A living will does not help clarify the patient’s wishes in the absence of antecedent conversation with the family, close friends, and the patient’s personal physician. And living wills cannot be read and interpreted in an emergency.
The situation is further complicated by difficulty in defining “terminal” or “irreversible” conditions and accounting for the different perspective that physicians, agents, and loved ones bring to the situation. For example, imagine a patient with dementia nearing the end of life who eats less, has difficulty managing secretions, aspirates, and develops pneumonia. While end-stage dementia is terminal, pneumonia may be reversible.
Increasingly, therefore, people are being counseled to appoint a health care agent (see below).3
The importance of a health care proxy (durable power of attorney for health care)
In a health care proxy document (also known as durable power of attorney for health care), the patient names a health care agent. This person has authority to make decisions about the patient’s medical care, including life-sustaining treatment. In other words, you the patient appoint someone to speak for you in the event you are unable to make your own medical decisions (not only at the end of life).
Since anyone may face a sudden and unexpected acute illness or injury with the risk of becoming incapacitated and unable to make medical decisions, everyone age 18 and older should be encouraged to complete a health care proxy document and to engage in advance care planning discussions with family and loved ones. Physicians can initiate this process as a wellness initiative and can help patients and families understand advance care planning. In all health care settings, trained and qualified health care professionals can provide education on advance care planning to patients, families, and loved ones.
A key issue when naming a health care agent is choosing the right one, someone who will make decisions in accordance with the person’s current values and beliefs and who can separate his or her personal values from the patient’s values. Another key issue: people need to have proactive discussions about their personal values, beliefs, and goals of care, which many are reluctant to do, and the health care agent must be willing to talk about sensitive issues ahead of time. Even when a health care agent is available in an emergency, emergency medical services personnel cannot follow directions from a health care agent. Most importantly, a health care agent must be able to handle potential conflicts between family and providers.
POLST ENSURES PATIENT PREFERENCES ARE HONORED AT THE END OF LIFE
Approximately 20 years ago, a team of health care professionals at the University of Oregon recognized these problems and realized that physicians needed to be more involved in discussions with patients about end-of-life care and in translating the patient’s preferences and values into concrete medical orders. The result was the Physician Orders for Life-Sustaining Treatment (POLST) Paradigm Program.4
What is POLST?
POLST is an end-of-life-care transitions program that focuses on patient-centered goals for care and shared informed medical decision-making.5,6 It offers a mechanism to communicate the wishes of seriously ill patients to have or to limit medical treatment as they move from one care setting to another. Table 1 lists the differences between traditional advance directives and POLST.
The aim is to improve the quality of care that seriously ill patients receive at the end of life. POLST is based on effective communication of the patient’s wishes, with actionable medical orders documented on a brightly colored form (www.ohsu.edu/polst/programs/sample-forms.htm; Figure 1) and a promise by health care professionals to honor these wishes.7 Key features of the program include education, training, and a quality-improvement process.
Who is POLST for?
POLST is for patients with serious life-limiting illness who have a life expectancy of less than 1 year, or anyone of advanced age interested in defining their end-of-life care wishes. Qualified and trained health care professionals (physicians, physician’s assistants, nurse practitioners, and social workers) participate in discussions leading to the completion of a POLST form in all settings, particularly along the long-term care continuum and for home hospice.
The key element of the POLST process: Shared, informed medical decision-making
Health care professionals working as an interdisciplinary team play a key role in educating patients and their families about advance care planning and shared, informed medical decision-making, as well as in resolving conflict. To be effective, shared medical decision-making must be well-informed. The decision-maker (patient, health care agent, or surrogate) must weigh the following questions (Table 2):
- Will treatment make a difference?
- Do the burdens of treatment outweigh its benefits?
- Is there hope of recovery? If so, what will life be like afterward?
- What does the patient value? What is the patient’s goal for his or her care?
In-depth discussions with patients, family members, and surrogates are needed, and these people are often reluctant to ask these questions and afraid to discuss the dying process. Even if they are informed of their diagnosis and prognosis, they may not know what they mean in terms of their everyday experience and future.
Health care professionals engaging in these conversations can use the eight-step POLST protocol (Table 3) to elicit their preferences at the end of life. Table 4 lists tools and resources to enhance the understanding of advance care planning and POLST.
What does the POLST form cover?
The POLST form (Figure 1) provides instructions about resuscitation if the patient has no pulse and is not breathing. Additionally, the medical orders indicate decisions about the level of medical intervention that the patient wants or does not want, eg, intubation, mechanical ventilation, transport to the hospital, intensive care, artificial nutrition and hydration, and antibiotics.
Thus, POLST is outcome-neutral and can be used either to limit medical interventions or to clarify a request for any or all medically indicated treatments.
Both the practitioner and the patient or patient’s surrogate sign the form. The original goes into the patient’s chart, and a copy should accompany the patient if he or she is transferred or discharged. Additionally, if the state has a POLST registry, the POLST information should be entered into the registry.
POLST is expanding across the country
The use of POLST has been expanding across the United States, with POLST programs now implemented in all or part of at least 30 states. There are endorsed programs in 14 states, and programs are being developed in 26 more. Requirements for endorsement are found at www.polst.org. Figure 2 shows the status of POLST in the 50 states.
Oregon’s POLST form is the original model for other forms designed to meet specific legislative or regulatory requirements in other states. POLST-like programs are known by different names in different states: eg, New York’s Medical Orders for Life-Sustaining Treatment (MOLST) and West Virginia’s Physicians Orders for Scope of Treatment (POST), but all endorsed programs share common core elements.
POLST research
A number of studies in the past 10 years have shown that POLST improves the documentation and honoring of patient preferences, whatever they may be.4,8–16
Emergency medical technicians in Oregon reported that the POLST form provides clear instructions about patient preferences and is useful when deciding which treatments to provide. In contrast to the single-intervention focus of out-of-hospital do-not-resuscitate orders, the POLST form provides patients the opportunity to document treatment goals and preferences for interventions across a range of treatment options, thus permitting greater individualization.13
Comfort care is not sacrificed if a POLST document is in place. Most hospice patients choose at least one life-sustaining treatment on their POLST form.14
In a multistate study published in 2010, the medical records of residents in 90 randomly chosen Medicaid-eligible nursing homes were reviewed.15 POLST was compared with traditional advance care planning in terms of the effect on the presence of medical orders reflecting treatment preferences, symptom management, and use of life-sustaining treatments. The study found that residents with POLST forms had significantly more medical orders about life-sustaining treatments than residents with traditional advance directives. There were no differences between residents with or without POLST forms on symptom assessment or management measures. POLST was more effective than traditional advance planning at limiting unwanted life-sustaining treatments. The study suggests that POLST offers significant advantages over traditional advance directives in nursing facilities.15,16
In summary, more than a decade of research has shown that the POLST Paradigm Program serves as an emerging national model for implementing shared, informed medical decision-making. Furthermore, POLST more accurately conveys end-of-life care preferences for patients with advanced chronic illness and for dying patients than traditional advance directives and yields higher adherence by medical professionals.
CLINICAL CASE REVISITED
Let’s consider if the physician for our 89-year-old woman with dementia had completed a POLST form with orders indicating “do not attempt resuscitation (DNR/no CPR)” and “comfort measures only, do not transfer to hospital for life-sustaining treatment and transfer if comfort needs cannot be met in current location.”
The patient’s respiratory distress and fever would have been treated at her nursing home with medication and oxygen. She would have been transferred to the hospital only if her comfort needs would not have been met at the nursing home. Unwanted life-sustaining treatment would have been avoided. The wishes of the patient, based on her values and careful consideration of options, would have been respected.
- Dunn PM, Tolle SW, Moss AH, Black JS. The POLST paradigm: respecting the wishes of patients and families. Ann Long-Term Care 2007; 15:33–40.
- Patient Self-Determination Act of 1990. Pub. L. No. 101-508, ss 4206, 104 Stat. 1388.
- Bomba PA, Sabatino CP. POLST: an emerging model for end-of-life care planning. The ElderLaw Report 2009; 20:1–5.
- Karp Sabatino C. AARP Public Policy Institute, Improving advance illness care: the evolution of state POLST programs 2011. http://assets.aarp.org/rgcenter/ppi/cons-prot/POLST-Report-04-11.pdf. Accessed May 30, 2012.
- Bomba PA. Discussing patient p and end of life care, Journal of the Monroe County Medical Society, 7th District Branch, MSSNY 2011;12–15. www.compassionandsupport.org/index.php/research_/. Accessed May 30, 2012.
- Citko J, Moss AH, Carley M, Tolle SW. The National POLST Paradigm Initiative, 2ND ed. Fast Facts and Concepts 2010;178. www.eperc.mcw.edu/fastfact/ff_178.htm. Accessed May 30, 2012.
- Center for Ethics in Health Care, Oregon Health & Science University. www.ohsu.edu/polst/. Accessed May 30, 2012.
- Lee MA, Brummel-Smith K, Meyer J, Drew N, London MR. Physician orders for life-sustaining treatment (POLST): outcomes in a PACE program. Program of All-Inclusive Care for the Elderly. J Am Geriatr Soc 2000; 48:1219–1225.
- Meyers JL, Moore C, McGrory A, Sparr J, Ahern M. Physician orders for life-sustaining treatment form: honoring end-of-life directives for nursing home residents. J Gerontol Nurs 2004; 30:37–46.
- Dunn PM, Schmidt TA, Carley MM, Donius M, Weinstein MA, Dull VT. A method to communicate patient p about medically indicated life-sustaining treatment in the out-of-hospital setting. J Am Geriatr Soc 1996; 44:785–791.
- Cantor MD. Improving advance care planning: lessons from POLST. Physician Orders for Life-Sustaining Treatment (comment). J Am Geriatr Soc 2000; 48:1343–1344.
- Tolle SW, Tilden VP, Nelson CA, Dunn PM. A prospective study of the efficacy of the physician order form for life-sustaining treatment. J Am Geriatr Soc 1998; 46:1097–1102.
- Schmidt TA, Hickman SE, Tolle SW, Brooks HS. The Physician Orders for Life-Sustaining Treatment program: Oregon emergency medical technicians’ practical experiences and attitudes. J Am Geriatr Soc 2004; 52:1430–1434.
- Hickman SE, Nelson CA, Moss AH, et al. Use of the Physician Orders for Life-Sustaining Treatment (POLST) paradigm program in the hospice setting. J Palliat Med 2009; 12:133–141.
- Hickman SE, Nelson CA, Perrin NA, Moss AH, Hammes BJ, Tolle SW. A comparison of methods to communicate treatment p in nursing facilities: traditional practices versus the physician orders for life-sustaining treatment program. J Am Geriatr Soc 2010; 58:1241–1248.
- Hickman SE, Nelson CA, Moss AH, Tolle SW, Perrin NA, Hammes BJ. The consistency between treatments provided to nursing facility residents and orders on the physician orders for life-sustaining treatment form. J Am Geriatr Soc 2011; 59:2091–2099.
- Dunn PM, Tolle SW, Moss AH, Black JS. The POLST paradigm: respecting the wishes of patients and families. Ann Long-Term Care 2007; 15:33–40.
- Patient Self-Determination Act of 1990. Pub. L. No. 101-508, ss 4206, 104 Stat. 1388.
- Bomba PA, Sabatino CP. POLST: an emerging model for end-of-life care planning. The ElderLaw Report 2009; 20:1–5.
- Karp Sabatino C. AARP Public Policy Institute, Improving advance illness care: the evolution of state POLST programs 2011. http://assets.aarp.org/rgcenter/ppi/cons-prot/POLST-Report-04-11.pdf. Accessed May 30, 2012.
- Bomba PA. Discussing patient p and end of life care, Journal of the Monroe County Medical Society, 7th District Branch, MSSNY 2011;12–15. www.compassionandsupport.org/index.php/research_/. Accessed May 30, 2012.
- Citko J, Moss AH, Carley M, Tolle SW. The National POLST Paradigm Initiative, 2ND ed. Fast Facts and Concepts 2010;178. www.eperc.mcw.edu/fastfact/ff_178.htm. Accessed May 30, 2012.
- Center for Ethics in Health Care, Oregon Health & Science University. www.ohsu.edu/polst/. Accessed May 30, 2012.
- Lee MA, Brummel-Smith K, Meyer J, Drew N, London MR. Physician orders for life-sustaining treatment (POLST): outcomes in a PACE program. Program of All-Inclusive Care for the Elderly. J Am Geriatr Soc 2000; 48:1219–1225.
- Meyers JL, Moore C, McGrory A, Sparr J, Ahern M. Physician orders for life-sustaining treatment form: honoring end-of-life directives for nursing home residents. J Gerontol Nurs 2004; 30:37–46.
- Dunn PM, Schmidt TA, Carley MM, Donius M, Weinstein MA, Dull VT. A method to communicate patient p about medically indicated life-sustaining treatment in the out-of-hospital setting. J Am Geriatr Soc 1996; 44:785–791.
- Cantor MD. Improving advance care planning: lessons from POLST. Physician Orders for Life-Sustaining Treatment (comment). J Am Geriatr Soc 2000; 48:1343–1344.
- Tolle SW, Tilden VP, Nelson CA, Dunn PM. A prospective study of the efficacy of the physician order form for life-sustaining treatment. J Am Geriatr Soc 1998; 46:1097–1102.
- Schmidt TA, Hickman SE, Tolle SW, Brooks HS. The Physician Orders for Life-Sustaining Treatment program: Oregon emergency medical technicians’ practical experiences and attitudes. J Am Geriatr Soc 2004; 52:1430–1434.
- Hickman SE, Nelson CA, Moss AH, et al. Use of the Physician Orders for Life-Sustaining Treatment (POLST) paradigm program in the hospice setting. J Palliat Med 2009; 12:133–141.
- Hickman SE, Nelson CA, Perrin NA, Moss AH, Hammes BJ, Tolle SW. A comparison of methods to communicate treatment p in nursing facilities: traditional practices versus the physician orders for life-sustaining treatment program. J Am Geriatr Soc 2010; 58:1241–1248.
- Hickman SE, Nelson CA, Moss AH, Tolle SW, Perrin NA, Hammes BJ. The consistency between treatments provided to nursing facility residents and orders on the physician orders for life-sustaining treatment form. J Am Geriatr Soc 2011; 59:2091–2099.
KEY POINTS
- Failures and opportunities for improvement in current advance care planning processes highlight the need for change.
- Differences exist between traditional advance directives and actionable medical orders.
- Advance care planning discussions can be initiated by physicians as a wellness initiative for everyone 18 years of age and older and can help patients and families understand advance care planning.
- POLST is outcome-neutral and may be used either to limit medical interventions or to clarify a request for any or all medically indicated treatments.
- Shared, informed medical decision-making is an essential element of the POLST process.
Tool Boosts Power to Predict Delirium in Adult ICU
A recently developed tool could help doctors stay ahead of the game in preventing delirium in intensive care patients.
Dutch researchers say their delirium prediction model, known as PRE-DELIRIC, was significantly more successful than doctors and nurses at predicting delirium in hospitalized adults.
Preventive measures for delirium can limit its incidence, severity, and duration. While several assessment tools exist for other segments of hospitalized patients, "no evidence-based prediction model for general intensive care patients is available," Mark van den Boogaard, Ph.D., of Radboud University Nijmegen (Netherlands) Medical Centre and his colleagues said (BMJ 2012;344:e420 [doi: 10.1136/bmj.e420]).
General preventive measures in all ICU patients are time consuming, and may expose many patients to unnecessary risks such as adverse events related to drug prophylaxis, the researchers explained.
For PRE-DELIRIC (Prediction of Delirium in ICU Patients), Dr. van den Boogaard and his colleagues defined 10 risk factors that can be easily assessed within 24 hours of admission to the ICU: age, APACHE II (Acute Physiology and Chronic Health Evaluation II) score, admission category, coma, infection, metabolic acidosis, morphine use, sedative use, urea concentration, and urgent admission.
"The use of the PRE-DELERIC model to identify and consequently preventively treat high-risk patients could offer an important contribution to intensive care practice and ensure efficient use of research resources to study only high-risk patients," the researchers said.
Clinically, the model may improve the use of nondrug measures to prevent delirium in high-risk patients, the researchers added. Such measures include improvement of orientation, cognitive stimulation, early mobilization, and listening to music, they said.
In noncritical patients, nondrug preventive measures have been shown to reduce delirium incidence and duration, and haloperidol treatment has lessened severity, duration, and associated length of stay. But for ICU patients, data are hard to come by. PRE-DELIRIC could inform the choice to use prophylactic haloperidol in these patients, the authors said. Existing research (Lancet 2009;373:1874-82) does show that "early mobilisation of mechanically ventilated patients in intensive care, besides other significant effects, resulted in a reduced duration of delirium," Dr. van den Boogaard and his coauthors wrote.
After testing their model for temporal validation, the researchers conducted an external validation study of data from intensive care patients admitted to four Dutch hospitals between Jan. 1 and Sept. 1, 2009. The pooled data included information from 3,056 patients aged 18 years and older, yielding an area under the receiver operating characteristics curve (AUROC) of 0.85. The patients were divided into four risk groups: low, moderate, high, and very high. The sensitivity and specificity were, respectively, 81% and 75% for the group with low-risk group; 62% and 89% for the moderate-risk group; 46% and 95% for the high-risk group; and 30% and 98% for the group with very high risk.
The researchers compared the predictions of patient delirium made by their model to predictions made by doctors and nurses in the hospital, using a convenience sample of 124 patients.
The AUROC for the PRE-DELIRIC model (0.87) was significantly higher than that of the doctors and nurses (0.59).
No significant differences appeared in the predictions of intensive care nurses compared with student intensive care nurses or among intensivists, fellow-intensivists, and residents, the researchers said.
The PRE-DELIRIC model is being used in daily practice in the hospital where the model was developed, the researchers said. "Intensive care patients with a high risk of delirium (at least a 50% PRE-DELIRIC score), and patients with dementia or alcohol misuse, receive preventive measures. The optimal cut-off point of the PRE-DELIRIC model and the most effective delirium preventive interventions for intensive care patients need to be studied in the near future."
The findings were limited by the static nature of the model, which does not account for changes in health status that might affect the odds of developing delirium, the researchers noted.
The researchers reported having no financial conflicts of interest.
haloperidol treatment,
A recently developed tool could help doctors stay ahead of the game in preventing delirium in intensive care patients.
Dutch researchers say their delirium prediction model, known as PRE-DELIRIC, was significantly more successful than doctors and nurses at predicting delirium in hospitalized adults.
Preventive measures for delirium can limit its incidence, severity, and duration. While several assessment tools exist for other segments of hospitalized patients, "no evidence-based prediction model for general intensive care patients is available," Mark van den Boogaard, Ph.D., of Radboud University Nijmegen (Netherlands) Medical Centre and his colleagues said (BMJ 2012;344:e420 [doi: 10.1136/bmj.e420]).
General preventive measures in all ICU patients are time consuming, and may expose many patients to unnecessary risks such as adverse events related to drug prophylaxis, the researchers explained.
For PRE-DELIRIC (Prediction of Delirium in ICU Patients), Dr. van den Boogaard and his colleagues defined 10 risk factors that can be easily assessed within 24 hours of admission to the ICU: age, APACHE II (Acute Physiology and Chronic Health Evaluation II) score, admission category, coma, infection, metabolic acidosis, morphine use, sedative use, urea concentration, and urgent admission.
"The use of the PRE-DELERIC model to identify and consequently preventively treat high-risk patients could offer an important contribution to intensive care practice and ensure efficient use of research resources to study only high-risk patients," the researchers said.
Clinically, the model may improve the use of nondrug measures to prevent delirium in high-risk patients, the researchers added. Such measures include improvement of orientation, cognitive stimulation, early mobilization, and listening to music, they said.
In noncritical patients, nondrug preventive measures have been shown to reduce delirium incidence and duration, and haloperidol treatment has lessened severity, duration, and associated length of stay. But for ICU patients, data are hard to come by. PRE-DELIRIC could inform the choice to use prophylactic haloperidol in these patients, the authors said. Existing research (Lancet 2009;373:1874-82) does show that "early mobilisation of mechanically ventilated patients in intensive care, besides other significant effects, resulted in a reduced duration of delirium," Dr. van den Boogaard and his coauthors wrote.
After testing their model for temporal validation, the researchers conducted an external validation study of data from intensive care patients admitted to four Dutch hospitals between Jan. 1 and Sept. 1, 2009. The pooled data included information from 3,056 patients aged 18 years and older, yielding an area under the receiver operating characteristics curve (AUROC) of 0.85. The patients were divided into four risk groups: low, moderate, high, and very high. The sensitivity and specificity were, respectively, 81% and 75% for the group with low-risk group; 62% and 89% for the moderate-risk group; 46% and 95% for the high-risk group; and 30% and 98% for the group with very high risk.
The researchers compared the predictions of patient delirium made by their model to predictions made by doctors and nurses in the hospital, using a convenience sample of 124 patients.
The AUROC for the PRE-DELIRIC model (0.87) was significantly higher than that of the doctors and nurses (0.59).
No significant differences appeared in the predictions of intensive care nurses compared with student intensive care nurses or among intensivists, fellow-intensivists, and residents, the researchers said.
The PRE-DELIRIC model is being used in daily practice in the hospital where the model was developed, the researchers said. "Intensive care patients with a high risk of delirium (at least a 50% PRE-DELIRIC score), and patients with dementia or alcohol misuse, receive preventive measures. The optimal cut-off point of the PRE-DELIRIC model and the most effective delirium preventive interventions for intensive care patients need to be studied in the near future."
The findings were limited by the static nature of the model, which does not account for changes in health status that might affect the odds of developing delirium, the researchers noted.
The researchers reported having no financial conflicts of interest.
A recently developed tool could help doctors stay ahead of the game in preventing delirium in intensive care patients.
Dutch researchers say their delirium prediction model, known as PRE-DELIRIC, was significantly more successful than doctors and nurses at predicting delirium in hospitalized adults.
Preventive measures for delirium can limit its incidence, severity, and duration. While several assessment tools exist for other segments of hospitalized patients, "no evidence-based prediction model for general intensive care patients is available," Mark van den Boogaard, Ph.D., of Radboud University Nijmegen (Netherlands) Medical Centre and his colleagues said (BMJ 2012;344:e420 [doi: 10.1136/bmj.e420]).
General preventive measures in all ICU patients are time consuming, and may expose many patients to unnecessary risks such as adverse events related to drug prophylaxis, the researchers explained.
For PRE-DELIRIC (Prediction of Delirium in ICU Patients), Dr. van den Boogaard and his colleagues defined 10 risk factors that can be easily assessed within 24 hours of admission to the ICU: age, APACHE II (Acute Physiology and Chronic Health Evaluation II) score, admission category, coma, infection, metabolic acidosis, morphine use, sedative use, urea concentration, and urgent admission.
"The use of the PRE-DELERIC model to identify and consequently preventively treat high-risk patients could offer an important contribution to intensive care practice and ensure efficient use of research resources to study only high-risk patients," the researchers said.
Clinically, the model may improve the use of nondrug measures to prevent delirium in high-risk patients, the researchers added. Such measures include improvement of orientation, cognitive stimulation, early mobilization, and listening to music, they said.
In noncritical patients, nondrug preventive measures have been shown to reduce delirium incidence and duration, and haloperidol treatment has lessened severity, duration, and associated length of stay. But for ICU patients, data are hard to come by. PRE-DELIRIC could inform the choice to use prophylactic haloperidol in these patients, the authors said. Existing research (Lancet 2009;373:1874-82) does show that "early mobilisation of mechanically ventilated patients in intensive care, besides other significant effects, resulted in a reduced duration of delirium," Dr. van den Boogaard and his coauthors wrote.
After testing their model for temporal validation, the researchers conducted an external validation study of data from intensive care patients admitted to four Dutch hospitals between Jan. 1 and Sept. 1, 2009. The pooled data included information from 3,056 patients aged 18 years and older, yielding an area under the receiver operating characteristics curve (AUROC) of 0.85. The patients were divided into four risk groups: low, moderate, high, and very high. The sensitivity and specificity were, respectively, 81% and 75% for the group with low-risk group; 62% and 89% for the moderate-risk group; 46% and 95% for the high-risk group; and 30% and 98% for the group with very high risk.
The researchers compared the predictions of patient delirium made by their model to predictions made by doctors and nurses in the hospital, using a convenience sample of 124 patients.
The AUROC for the PRE-DELIRIC model (0.87) was significantly higher than that of the doctors and nurses (0.59).
No significant differences appeared in the predictions of intensive care nurses compared with student intensive care nurses or among intensivists, fellow-intensivists, and residents, the researchers said.
The PRE-DELIRIC model is being used in daily practice in the hospital where the model was developed, the researchers said. "Intensive care patients with a high risk of delirium (at least a 50% PRE-DELIRIC score), and patients with dementia or alcohol misuse, receive preventive measures. The optimal cut-off point of the PRE-DELIRIC model and the most effective delirium preventive interventions for intensive care patients need to be studied in the near future."
The findings were limited by the static nature of the model, which does not account for changes in health status that might affect the odds of developing delirium, the researchers noted.
The researchers reported having no financial conflicts of interest.
haloperidol treatment,
haloperidol treatment,
FROM THE BRITISH MEDICAL JOURNAL
FDA: Drug Shortages Decline but Persist
The Food and Drug Administration has prevented at least 50 drug shortages so far this year, mainly as a result of manufacturers giving the agency early warning of impending problems, Dr. Sandra Kweder announced June 4.
Shortages persist, however, especially for key oncology therapies, according to Dr. Kweder, deputy director of the FDA Office of New Drugs, and oncologists who participated in a press briefing at the annual meeting of the American Society of Clinical Oncologists (ASCO) in Chicago.
Since the White House issued an executive order last Oct. 31 encouraging manufacturers to give the FDA "early notification" of potential disruptions, the agency, working with physicians, manufacturers, pharmacists, and others, has been able to avoid more than 150 drug shortages, Dr. Kweder said.
"Early notification by manufacturers of when they are having production difficulties makes a huge difference in our ability to prevent the public from bearing the burden of drug shortages," she said.
"The good news is that the frequency of drug shortages has begun to decline," agreed Dr. Richard Schilsky, chair of the ASCO Government Relations Committee and chief of the hematology/oncology section at the University of Chicago.
However, unpredictability still exists, which means that oncologists are "never sure when a generic is going to go out of supply," said Dr. Schilsky. "That creates a tremendous amount of uncertainty and anxiety for our patients."
Dr. Michael P. Link, ASCO president, said that several commonly used chemotherapy agents remain in short supply, including fluorouracil, nitrogen mustard, and paclitaxel. In addition, although a methotrexate shortage has been addressed, there is now a shortage of sodium bicarbonate, which is needed to give high-dose injections of the drug, said Dr. Link, chief of pediatric hematology/oncology at the Lucile Packard Children’s Hospital at Stanford (Calif.) University.
Shortages often lead to delays in treatment or less-than-desirable substitutions, said Dr. W. Charles Penley, the incoming ASCO Government Relations Committee chair. His practice – Tennessee Oncology in Nashville – is large enough that it generally has been able to locate products in short supply through its purchasing groups. But a leucovorin shortage last summer led him to switch some patients to oral capecitabine (Xeloda).
That drug is more expensive and is not tolerated well by some patients, he said.
And even that product almost went into short supply, Dr. Kweder said. There were some quality control issues that led the agency to temporarily import capecitabine while the manufacturer, Roche, fixed the problem, she said.
Manufacturing and quality control – especially for generic sterile injectables – continue to be behind many shortages, Dr. Kweder said. Sterility has been a consistent problem, with many products found to contain glass particles or metal shavings. She described a zero tolerance policy for such issues with injectables. "No patient should ever be exposed to risks of those sorts," she said.
When asked why generic injectables seem to be having more frequent quality control issues, Dr. Kweder said that the requirements and rules for good manufacturing have not changed over the years, but that the explosion of generic products in the last decade has perhaps put more pressure on manufacturers.
Dr. Schilsky and Dr. Penley said they were hopeful that legislation creating user fees for generic drugmakers would help address the shortage issue. Bills to reauthorize all FDA user fee programs have passed both the House and the Senate; differences are being hashed out in conference right now. The generic user fee – expected to add $1.5 billion to the FDA budget over the next few years – is expected to be retained. Experts expect the president to sign the final bill, said Dr. Schilsky.
The additional funds would give the FDA the ability to cut generic drug review times from the current 24-30 months to about 6-10 months, Dr. Kweder said.
It also would give the agency more resources to eradicate a current backlog of reviews and move more quickly on manufacturing issues, she said.
The legislation includes several additional items on ASCO’s wish list, including a requirement that manufacturers give the FDA early warning of impending shortages. But it does not include any penalties for noncompliance, said Dr. Schilsky.
The Food and Drug Administration has prevented at least 50 drug shortages so far this year, mainly as a result of manufacturers giving the agency early warning of impending problems, Dr. Sandra Kweder announced June 4.
Shortages persist, however, especially for key oncology therapies, according to Dr. Kweder, deputy director of the FDA Office of New Drugs, and oncologists who participated in a press briefing at the annual meeting of the American Society of Clinical Oncologists (ASCO) in Chicago.
Since the White House issued an executive order last Oct. 31 encouraging manufacturers to give the FDA "early notification" of potential disruptions, the agency, working with physicians, manufacturers, pharmacists, and others, has been able to avoid more than 150 drug shortages, Dr. Kweder said.
"Early notification by manufacturers of when they are having production difficulties makes a huge difference in our ability to prevent the public from bearing the burden of drug shortages," she said.
"The good news is that the frequency of drug shortages has begun to decline," agreed Dr. Richard Schilsky, chair of the ASCO Government Relations Committee and chief of the hematology/oncology section at the University of Chicago.
However, unpredictability still exists, which means that oncologists are "never sure when a generic is going to go out of supply," said Dr. Schilsky. "That creates a tremendous amount of uncertainty and anxiety for our patients."
Dr. Michael P. Link, ASCO president, said that several commonly used chemotherapy agents remain in short supply, including fluorouracil, nitrogen mustard, and paclitaxel. In addition, although a methotrexate shortage has been addressed, there is now a shortage of sodium bicarbonate, which is needed to give high-dose injections of the drug, said Dr. Link, chief of pediatric hematology/oncology at the Lucile Packard Children’s Hospital at Stanford (Calif.) University.
Shortages often lead to delays in treatment or less-than-desirable substitutions, said Dr. W. Charles Penley, the incoming ASCO Government Relations Committee chair. His practice – Tennessee Oncology in Nashville – is large enough that it generally has been able to locate products in short supply through its purchasing groups. But a leucovorin shortage last summer led him to switch some patients to oral capecitabine (Xeloda).
That drug is more expensive and is not tolerated well by some patients, he said.
And even that product almost went into short supply, Dr. Kweder said. There were some quality control issues that led the agency to temporarily import capecitabine while the manufacturer, Roche, fixed the problem, she said.
Manufacturing and quality control – especially for generic sterile injectables – continue to be behind many shortages, Dr. Kweder said. Sterility has been a consistent problem, with many products found to contain glass particles or metal shavings. She described a zero tolerance policy for such issues with injectables. "No patient should ever be exposed to risks of those sorts," she said.
When asked why generic injectables seem to be having more frequent quality control issues, Dr. Kweder said that the requirements and rules for good manufacturing have not changed over the years, but that the explosion of generic products in the last decade has perhaps put more pressure on manufacturers.
Dr. Schilsky and Dr. Penley said they were hopeful that legislation creating user fees for generic drugmakers would help address the shortage issue. Bills to reauthorize all FDA user fee programs have passed both the House and the Senate; differences are being hashed out in conference right now. The generic user fee – expected to add $1.5 billion to the FDA budget over the next few years – is expected to be retained. Experts expect the president to sign the final bill, said Dr. Schilsky.
The additional funds would give the FDA the ability to cut generic drug review times from the current 24-30 months to about 6-10 months, Dr. Kweder said.
It also would give the agency more resources to eradicate a current backlog of reviews and move more quickly on manufacturing issues, she said.
The legislation includes several additional items on ASCO’s wish list, including a requirement that manufacturers give the FDA early warning of impending shortages. But it does not include any penalties for noncompliance, said Dr. Schilsky.
The Food and Drug Administration has prevented at least 50 drug shortages so far this year, mainly as a result of manufacturers giving the agency early warning of impending problems, Dr. Sandra Kweder announced June 4.
Shortages persist, however, especially for key oncology therapies, according to Dr. Kweder, deputy director of the FDA Office of New Drugs, and oncologists who participated in a press briefing at the annual meeting of the American Society of Clinical Oncologists (ASCO) in Chicago.
Since the White House issued an executive order last Oct. 31 encouraging manufacturers to give the FDA "early notification" of potential disruptions, the agency, working with physicians, manufacturers, pharmacists, and others, has been able to avoid more than 150 drug shortages, Dr. Kweder said.
"Early notification by manufacturers of when they are having production difficulties makes a huge difference in our ability to prevent the public from bearing the burden of drug shortages," she said.
"The good news is that the frequency of drug shortages has begun to decline," agreed Dr. Richard Schilsky, chair of the ASCO Government Relations Committee and chief of the hematology/oncology section at the University of Chicago.
However, unpredictability still exists, which means that oncologists are "never sure when a generic is going to go out of supply," said Dr. Schilsky. "That creates a tremendous amount of uncertainty and anxiety for our patients."
Dr. Michael P. Link, ASCO president, said that several commonly used chemotherapy agents remain in short supply, including fluorouracil, nitrogen mustard, and paclitaxel. In addition, although a methotrexate shortage has been addressed, there is now a shortage of sodium bicarbonate, which is needed to give high-dose injections of the drug, said Dr. Link, chief of pediatric hematology/oncology at the Lucile Packard Children’s Hospital at Stanford (Calif.) University.
Shortages often lead to delays in treatment or less-than-desirable substitutions, said Dr. W. Charles Penley, the incoming ASCO Government Relations Committee chair. His practice – Tennessee Oncology in Nashville – is large enough that it generally has been able to locate products in short supply through its purchasing groups. But a leucovorin shortage last summer led him to switch some patients to oral capecitabine (Xeloda).
That drug is more expensive and is not tolerated well by some patients, he said.
And even that product almost went into short supply, Dr. Kweder said. There were some quality control issues that led the agency to temporarily import capecitabine while the manufacturer, Roche, fixed the problem, she said.
Manufacturing and quality control – especially for generic sterile injectables – continue to be behind many shortages, Dr. Kweder said. Sterility has been a consistent problem, with many products found to contain glass particles or metal shavings. She described a zero tolerance policy for such issues with injectables. "No patient should ever be exposed to risks of those sorts," she said.
When asked why generic injectables seem to be having more frequent quality control issues, Dr. Kweder said that the requirements and rules for good manufacturing have not changed over the years, but that the explosion of generic products in the last decade has perhaps put more pressure on manufacturers.
Dr. Schilsky and Dr. Penley said they were hopeful that legislation creating user fees for generic drugmakers would help address the shortage issue. Bills to reauthorize all FDA user fee programs have passed both the House and the Senate; differences are being hashed out in conference right now. The generic user fee – expected to add $1.5 billion to the FDA budget over the next few years – is expected to be retained. Experts expect the president to sign the final bill, said Dr. Schilsky.
The additional funds would give the FDA the ability to cut generic drug review times from the current 24-30 months to about 6-10 months, Dr. Kweder said.
It also would give the agency more resources to eradicate a current backlog of reviews and move more quickly on manufacturing issues, she said.
The legislation includes several additional items on ASCO’s wish list, including a requirement that manufacturers give the FDA early warning of impending shortages. But it does not include any penalties for noncompliance, said Dr. Schilsky.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Cluster Headache: Hastening Diagnosis and Treatment
Headache is one of the health problems most commonly associated with significant morbidity, as well as considerable social and economic repercussions.1,2 Headaches are classified into three main types:
- Primary headaches, or headaches without known organic causes
- Secondary headaches, or headaches manifesting with symptoms due to organic causes; and
- Cranial neuralgias, facial pain, and other headaches.3
Cluster headache (CH) is a type of primary headache—one of the headache types encompassed by the term trigeminal autonomic cephalalgia.3 It is one of the most intense, excruciating headaches a patient can experience, and the diagnosis is often missed or delayed. Only 21% of patients receive a correct diagnosis of CH on first presentation, and the average patient visits three health care providers before the correct diagnosis is made.4,5 According to recently published results from the US Cluster Headache Survey,4 the diagnostic delay for CH averages five years or longer, limiting the patient’s access to correct treatment.
Patients with CH are prone to significant physical, social, and economic disability; most patients, for example, find it difficult to work during a CH period.1,2 Almost 20% of patients with CH report having lost a job because of their headaches, and about 8% are unemployed or on disability.4
Because of the pain severity and the associated impairment, the risk for suicide in the CH patient population is real.2,6,7 Jürgens et al7 report that 22% of patients with chronic CH and about 15% of those with episodic CH had suicidal tendencies; Rozen and Fishman4 report suicidal ideation in 55% of CH patients.
CHARACTERISTICS OF CLUSTER HEADACHE
As the name implies, attacks of this headache type tend to “cluster” together. In 85% to 90% of patients, CH is episodic, with cluster periods of headache attacks commonly lasting for one week to one year, and intervening remission periods that may last from one month to years.3,8 The remaining 10% to 15% of CH patients have the chronic CH type, in which cluster periods typically last for more than one year and are separated by remission periods lasting one month or less.3,8
Cluster headaches tend to occur in predictable patterns, often in the spring and fall.9-11 Most headaches begin between early evening and early morning, and patients are often awakened by CH during the night; according to responses in the US Cluster Headache Survey,4 onset times peak between midnight and 3 AM. Attacks can occur when the neck is rotated or flexed in specific ways; external pressure to the transverse processes of C4 or the nerve root of C2 can trigger a CH attack.12
Other CH triggers include alcohol (especially beer and red wine4,13), histamine, nitroglycerine, carbon dioxide, certain odors, and weather changes.3,4,11,14 Eighty percent or more of CH patients have a history of prolonged tobacco use, and at least 60% of CH patients who do not smoke were the children of smokers.15 No clear relationship has been found between CH and hormones.9,10
EPIDEMIOLOGY
Cluster headache is relatively rare, affecting about 0.1% of the population.8-10,16 Onset of the condition usually occurs between ages 20 and 40, and men are three to four times more likely to be affected than are women.3,16 A familial/genetic relationship may exist.10,17,18
DIAGNOSIS
Patient History
Diagnosis of headache relies heavily on the patient’s clinical history and physical exam.3,8,10 A detailed history should include the initial onset of CH, progression of the condition, and information about any precipitating event(s) and prodromal symptoms. Clinicians should document the pattern of pain by including specific information regarding its location, severity, quality, frequency, and duration. Of considerable value is the patient’s use of an accurate headache diary, which clinicians should encourage headache patients to maintain; in these, patients should be instructed to record the headache characteristics mentioned.3,8
Associated symptoms (assessed by conducting a complete review of systems), aggravating and alleviating factors, previous medical history, and psychosocial and family history are important in formulating the differential diagnosis, as misdiagnosis of CH is often related to inadequate history intake.13
Presentation
Cluster headaches share three main features: they are unilateral; they are associated with autonomic symptoms; and attacks tend to “cluster” in a circannual pattern (ie, clusters occurring at the same time of year) and/or circadian pattern (headache at the same time of day).8,19 The most common locations for cluster headaches are unilateral orbital, supraorbital, temporal, or a combination of these locations.3,8
About 30% of patients describe the pain as “stabbing,”3 and it is often compared to “a hot poker in the eye.” Pain peaks rapidly, usually within five to 10 minutes. It may radiate to the ipsilateral forehead, jaw, cheek, and/or teeth.3 Patients appear restless and agitated, unable to lie still.2,10 They often sit, holding their heads, and may pace the floor or bang their heads against the wall.
CH is associated with at least one of the following autonomic symptoms, occurring in the ipsilateral side of the head: conjunctival injection, nasal congestion, forehead and facial sweating, eyelid edema, lacrimation, rhinorrhea, ptosis, and miosis.3,13 Headaches may occur on one side of the head throughout one cluster episode, then shift to the contralateral side in subsequent periods.10 Aura occurs in 14% to 20% of patients,13,20-22 and nausea, as well as ipsilateral
visual, sensory, and speech/language disturbances have also been reported.3 Each CH attack lasts between 15 minutes and three hours, and attacks may range in frequency from one every other day to eight per day.3,13
Patients who have experienced at least five episodes of these headache symptoms, with severe pain in the specified areas and duration, accompanying autonomic symptoms, specified attack frequency, and symptoms not attributed to another disorder meet the diagnostic criteria for cluster headache given in the second edition of the International Classification of Headache Disorders (ICHD-II, 2004).3 The ICHD-II criteria, based on clinical and epidemiologic research, are recognized as a consensus guideline that is accepted worldwide to facilitate clinical practice.3 Patients who have experienced attacks fulfilling all but one of the ICHD-II criteria for CH are diagnosed with probable CH3 or cluster-like headache (CLH).23
Physical Examination
A thorough physical examination, including an investigation of the neurologic system, is essential to differentiate among primary, secondary, and other headache types. In the patient with CH, no neurologic deficits or deficits that suggest underlying disorders are usually found.3,10
Differential Diagnosis
In the evaluation of headache, it is important to differentiate CH from the other trigeminal autonomic cephalalgias: paroxysmal hemicrania (PH), short-lasting unilateral neuralgiform headache with conjunctival injection and tearing (SUNCT), and possibly hemicrania continua.3,24,25 As in CH, the pain of PH is severe, unilateral, and stabbing in quality; it, too, is associated with autonomic symptoms, often occurs at night, and can be episodic or chronic.3 However, PH headache lasts for only 2 to 30 minutes and can occur five times or more per day. Though difficult to distinguish from CH patients, those with PH usually respond to indomethacin, whereas those with CH ordinarily do not.3,8
As in patients with CH, those affected by SUNCT experience autonomic symptoms—most commonly, conjunctival injection and tearing.3,26 SUNCT differs from CH, however, in that the pain is moderate in severity, with a pulsating, burning, electric-like quality. Duration is much shorter, with episodes lasting between 5 seconds and 4 minutes.3,26
Hemicrania continua, though unilateral, is described as continuous and moderate in intensity. Like PH, it is also indomethacin-responsive.8,25,27
A broader differential diagnosis for CH, as detailed in the table,11,3,8,13,26,28 includes the other primary headaches: tension headache, migraine headache, and trigeminal neuralgia.3,26Tension headache, which affects 30% to 78% of the general population,3 is subdivided into infrequent episodic, frequent episodic, and chronic tension-type headache. Unlike CH, tension headache is mild to moderate in intensity and occurs bilaterally, with nonpulsating pressure or a tightening sensation. It is not aggravated by routine physical activity, nor is it associated with nausea, vomiting, or photophobia.
Migraine headache, also a more common primary headache type than CH,3 occurs unilaterally, is moderate to severe in intensity, and is often described as throbbing. More gradual than CH in onset, migraine is often associated with nausea, vomiting, photophobia, phonophobia, and/or visual aura. Migraine headache lacks the ipsilateral autonomic manifestations of CH, and migraineurs prefer to rest or sleep—in contrast to the extreme restlessness or agitation seen in CH patients.3
Also like CH, trigeminal neuralgia is unilateral with a trigeminal nerve distribution, and the pain can be severe and stabbing.3 However, trigeminal neuralgia lacks the autonomic symptoms associated with CH, and the pain lasts from only seconds to minutes. This headache type is often triggered by washing, shaving, or brushing teeth.3
It is also critical to exclude secondary headaches, especially those with serious causes, including meningitis, subarachnoid hemorrhage, epidural or subdural hematoma, glaucoma, tumors, temporal arteritis, or purulent sinusitis.27 Red flags associated with these conditions are:
- A complaint of the patient’s “worst headache ever” (thunderclap headache)
- First severe headache
- A subacute headache worsening over days or weeks
- An abnormal neurologic examination
- Fever or other unexplained systemic signs
- A headache preceded by vomiting
- Headache that is induced by bending, lifting, or coughing
- Headache that disturbs the patient’s sleep or presents immediately upon awakening
- History of known systemic illness
- Headache onset after age 55; and
- Pain associated with local tenderness, for example, near the temporal artery.27
DIAGNOSTIC TESTING
Since, by definition, primary headaches are those without underlying organic causes, diagnostic tests and neuroimaging studies are generally not recommended,10,29 especially when the patient history and presentation confirm the required ICHD-II diagnostic criteria. However, neuroimaging is often recommended for a patient with CH or CH-like presentations.8,23,30-32
In a literature review published in 2006, Detsky et al30 examined the correlation between clinical features of headache (as described in the ICHD-II criteria) and intracranial abnormality (as found on CT or MRI). They found an increased risk for serious intracranial abnormalities among study subjects with cluster-type headache. In any patient with chronic headache, they found, abnormal neurologic findings on physical exam represent the greatest predictor for intracranial pathology.30
In a similar study of 1,872 consecutive patients with nonacute headaches who underwent CT or MRI, one of 20 patients with CH was found to have a pituitary adenoma.33 When Favier et al34 reviewed 31 cases of trigeminal autonomic cephalalgia (TAC), including 10 with atypical symptoms, they found that even typical TAC can result from underlying pathologies with rare warning signs and symptoms. In some patients, neuroimaging study results were normal on initial diagnosis, but pathologies were discovered later after symptoms worsened or treatments ceased to be effective, prompting further imaging studies.
In a review of case studies of patients with CLH, Mainardi et al23 found that of 38 patients who fulfilled the ICHD-II criteria for CH, 12 patients (31.6%) had vascular pathologies, 12 (31.6%) had tumors, and five (13.2%) had inflammatory or infectious pathologies. The researchers recommended that all patients with symptoms of CH or CLH undergo cerebral MRI with contrast medium, even though the yield for abnormal findings would likely be low.23 Wilbrink et al32 also found a wide range of pathologies without typical warning signs or symptoms among 56 case studies of TAC and TAC-like syndromes—and recommended that all such patients be considered candidates for neuroimaging.
Recommendations from both the Scottish Intercollegiate Guidelines Network (SIGN)8 and the Taiwan Headache Society treatment guidelines31 include neuroimaging of patients with CH or CLH.
MANAGEMENT
Clinicians may wish to consider referring patients to a neurologist at the initial diagnosis of CH. Patients with atypical symptomatology or neurologic abnormalities, and those who respond insufficiently to treatment warrant a neurology referral for further investigation.
The two treatment strategies for CH are first, symptomatic treatment for acute attacks; and second, intervention to prevent or reduce further attacks and to shorten the cluster period.8,11,14,35,36
Acute Treatment
Acute symptomatic treatment is aimed at aborting the pain within 15 to 30 minutes from headache onset.14,35,36 Currently, it is generally accepted that 100% oxygen and parenteral triptans (5-HT1B/1D, not through the alimentary tract) are considered first-line treatment options.8,11,13,14,35,36
For the majority of patients (particularly those with episodic CH), inhaled normobaric oxygen effectively relieves CH pain within 15 minutes.14,37-39 Oxygen is administered at 6 to 12 L/min with a nonrebreather mask for at least 15 to 20 minutes.8,14,13 Although associated adverse events are rare, oxygen is inconvenient to transport, and it poses a fire hazard. Additionally, high-flow oxygen is contraindicated in patients with chronic obstructive pulmonary disease, as these patients depend on the hypoxic drive and run the risk of respiratory depression.14,40,41
Triptans, too, have been found effective in the acute treatment of CH; administration by subcutaneous injection or intranasal delivery is considered more effective than the oral route due to faster onset of action,11,14,35 and oxygen use may enhance triptans’ efficacy.38 In two 2010 reviews of the relevant literature, subcutaneous sumatriptan, dosed at either 6 mg or 12 mg, provided effective pain relief within 15 minutes for most patients, with no statistical between-dosage differences.11,35 The most common adverse effects were injection-site reactions, nausea, vomiting, dizziness, fatigue, and paresthesias.35
Intranasal zolmitriptan (5 mg and 10 mg) and intranasal sumatriptan 20 mg were also found effective, with significant pain relief within 30 minutes. Bad taste is a common complaint.35 Of note, both sumatriptan and zolmitriptan are contraindicated in patients with cardiovascular or cerebrovascular disease.14
In an older study of efficacy, safety, and tolerability of subcutaneous sumatriptan, almost 70% of patients averaging between one and six CH attacks per day were found to be using more than the 12-mg maximum recommended daily dosage—as much as 36 mg in a 24-hour period.42 Nevertheless, the researchers concluded that subcutaneous sumatriptan was effective and well tolerated without decreased efficacy over one year in patients with CH.
Ergotamine, once commonly used for the acute treatment of CH, has fallen out of favor in recent years due to its vasoconstrictive effects and serious adverse effects profile.14,35 Dihydroergotamine (DHE) is most effective when administered by IV (though not easily accessible for an acute attack); however, evidence regarding its efficacy and tolerability in other forms is insufficient to recommend DHE for acute CH therapy.14,35
Intranasal lidocaine, somatostatin by infusion, and subcutaneous octreotide are considered second-line treatment choices for patients who are resistant to first-line therapies or who cannot tolerate them.14,35,43,44
Cluster Headache Prophylaxis
A CH period can last for weeks to months. Prophylactic modalities, which are intended to shorten this period and to reduce the frequency and severity of headache attacks, are categorized into transitional and maintenance prophylaxis treatments.14,35
Transitional prophylaxis, a shorter course of treatment, is often started with maintenance prophylaxis (which is used throughout each cluster period) to hasten the response to the maintenance treatment. Corticosteroids are commonly used as a transitional treatment modality. In prednisone use, a starting dose of at least 40 mg/d by mouth is often required to provide benefit.14 The peak dose is usually given for three to 10 days, then gradually tapered over the succeeding 10 to 30 days. Headache recurrence is common during the prednisone taper. Ergotamine tartrate and DHE are also used as transitional prophylaxis treatment for CH.14
Verapamil is considered the maintenance prophylaxis drug of choice due to its efficacy and safety.14,35,45 The dosage required for adequate response ranges from 200 mg to 960 mg/d, in divided doses or in extended-release formulation. Most patients respond to daily doses between 200 mg and 480 mg.14,46,47 Constipation is the most common adverse effect. Slow titration and frequent ECG monitoring, particularly when dosing is increased, are necessary to avoid heart block, bradycardia, hypotension, and peripheral edema.13
Lithium is often used as second-line therapy for maintenance prophylaxis, possibly in combination with verapamil or topiramate, to improve pain control.35 Lithium carbonate, given at a dosage of 600 to 900 mg/d to maintain a serum level of 0.4 to 0.8 mEq/L, and topiramate, at dosages ranging from 50 to 200 mg/d, may be needed to achieve an adequate response.14
In at least one small study, melatonin (10 mg/d) has been associated with CH remission in 50% of treated patients.14,48 It may be used in combination with other prophylactic medications.14
Among numerous other agents that have been used for CH prophylaxis, neither sodium valproate, sumatriptan, cimetidine/chlorpheniramine, misoprostol, nor oxygen is recommended for prevention of CH.35
Narcotics
Because of its excruciating pain, CH has been referred to as “suicide headache.”2,6,7,10,13 Acute and prophylactic treatments for CH will likely reduce the number of headache attacks; however, with CH attacks as frequent as eight times per day, these treatments may not be adequate.24,49
The use of any narcotic is not ideal due to its potential for addiction, and it may cause medication-overuse headache. Furthermore, in oral form, a narcotic may not relieve CH pain quickly enough. Low-dose levomethadone is an opioid that has been used prophylactically with some success in patients with chronic CH.24,49 However, the primary care provider whose CH patient finds pain control inadequate should refer to a neurologist or a pain management specialist for evaluation—and possibly for treatment with an invasive procedure.
Invasive Procedures
Greater occipital nerve block has shown promise in the treatment of CH.50,51 In a small, double-blind study, patients with episodic or chronic CH were randomized to receive a suboccipital injection, of either combined long- and rapid-acting betamethasone or saline (placebo), in the area of the greater occipital nerve.50 Eighty-five percent of treated patients were free of headache attacks within 72 hours, compared with none in the control group. Use of lidocaine with triamcinolone was found somewhat less effective.51
Occipital nerve stimulation has also shown promise for patients with chronic CH who become resistant or are unresponsive to conventional treatments, or who cannot tolerate them.14,24 It appears to induce gradual neuromodulation, with gradual benefits (after six to 30 months). Deep brain stimulation (ie, of the posterior hypothalamus), delivered via implanted electrodes, and other procedures have produced results ranging from “excellent” to “transient remission,” reducing the use of ablative surgeries.14,52 Because invasive modalities carry a risk for serious adverse effects,14 their use should be reserved for a select patient population.
PATIENT/FAMILY EDUCATION
Patients with CH need to be educated regarding the nature, signs and symptoms, and triggers of CH. The indications for acute and prophylactic treatments and the adverse effects associated with each therapy must also be reviewed. Clear follow-up instructions are essential, including what conditions warrant further evaluation: worsening of the condition, changes in symptoms (impaired alertness, vision, movement, or sensation; onset of seizures), or treatment failure.
CONCLUSION
Cluster headache, a relatively uncommon primary headache that can cause excruciating and debilitating pain, is often misdiagnosed and inappropriately treated, with serious physical, social, and economic ramifications. This headache type is unilateral, associated with autonomic symptoms, and characterized by clustering of headache/remission periods in a circannual and/or circadian pattern. Diagnosis is made through the health history and physical exam, based on criteria from the ICHD-II. Neuroimaging may not be necessary, but given the evidence that CH and TAC are often associated with serious underlying pathology, MRI with contrast should be considered, and consultation with a neurologist at initial diagnosis is recommended.
Treatment is aimed at aborting the pain within 15 to 30 minutes of an acute headache attack and preventing further episodes through transitional and maintenance prophylaxis. Newer invasive options that are showing great promise may be considered for a select patient population. Clinicians should involve patients in treatment decisions that will address their individual needs, improving function and optimizing outcomes.
1. Stovner LJ, Hagen H, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia. 2007;27(3):193-210.
2. Vaughan R. My own private purgatory: how cluster headaches affect my life. Headache. 2008;48(10):1541-1543.
3. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders. 2nd ed. Cephalalgia. 2004;24 suppl 1:9-160.
4. Rozen TD, Fishman RS. Cluster headache in the United States of America: demographics, clinical characteristics, triggers, suicidality, and personal burden. Headache. 2012;52(1): 99-113.
5. Bahra A, Goadsby PJ. Diagnostic delays and mis-management in cluster headache. Acta Neurol Scand. 2004;109(3):175-179.
6. Dousset V, Laporte A, Legoff M, et al. Validation of a brief self-administered questionnaire for cluster headache screening in a tertiary center. Headache. 2009;49(1):64-70.
7. Jürgens TP, Gaul C, Lindwurm A, et al. Impairment in episodic and chronic cluster headache. Cephalalgia. 2011;31(6):671-682.
8. Scottish Intercollegiate Guidelines Network (SIGN). Diagnosis and management of headache in adults: a national clinical guideline. Edinburgh (Scotland): Scottish Intercollegiate Guidelines Network (SIGN). 2008;1-81. (SIGN publication; no. 107). www.sign.ac.uk/guide lines/fulltext/107/index.html. Accessed May 8, 2012.
9. Bahra A, May A, Goadsby PJ. Cluster headache: a prospective clinical study with diagnostic implications. Neurology. 2002;58(3): 354-361.
10. May A. Cluster headache: pathogenesis, diagnosis, and management. Lancet. 2005; 366(9488):843-855.
11. Law S, Derry S, Moore RA. Triptans for acute cluster headache. Cochrane Database Syst Rev. 2010 Apr 14;(4):CD008042.
12. Rozen TD. Trigeminal autonomic cephalalgias. Neurol Clin. 2009;27(2):537-556.
13. Nesbitt AD, Goadsby PJ. Cluster headache. BMJ. 2012;344:e2407. doi: 10.1136/bmj
.e2407.
14. Ashkenazi A, Schwedt T. Cluster headache: acute and prophylactic therapy. Headache. 2011;51(2):272-286.
15. Rozen TD. Cluster headache as the result of secondhand cigarette smoke exposure during childhood. Headache. 2010;50(1):130-132.
16. Fischera M, Marziniak M, Gralow I, Evers S. The incidence and prevalence of cluster headache: a meta-analysis of population-based studies. Cephalalgia. 2008;28(6):614-618.
17. De Simone R, Fiorillo C, Bonuso S, Castaldo G. A cluster headache family with possible autosomal recessive inheritance. Neurology. 2003;61(4):578-579.
18. Leone M, Russell MB, Rigamonti A, et al. Increased familial risk of cluster headache. Neurology. 2001;56(9):1233-1236.
19. Matharu MS, Boes CJ, Goadsby PJ. Management of trigeminal autonomic cephalgias and hemicrania continua. Drugs. 2003;63(16): 1637-1677.
20. Evans RW, Krymchantowski AV. Cluster and other nonmigraine primary headaches with aura. Headache. 2011;51(4):604-608.
21. Rozen TD. Cluster headache with aura. Curr Pain Headache Rep. 2011;15(2):98-100.
22. Silberstein SD, Niknam R, Rozen TD, Young WB. Cluster headache with aura. Neurology. 2000;54(1):219-221.
23. Mainardi F, Trucco M, Maggioni F, et al. Cluster-like headache: a comprehensive reappraisal. Cephalalgia. 2010;30(4):399-412.
24. Magis D, Bruno MA, Fumal A, et al. Central modulation in cluster headache patients treated with occipital nerve stimulation: an FDG-PET study. BMC Neurol. 2011;11:25.
25. Goadsby PJ, Lipton RB. A review of paroxysmal hemicranias, SUNCT syndrome and other short-lasting headaches with autonomic feature, including new cases. Brain. 1997; 120(pt 1):193-209.
26. Leone M, Bussone G. Pathophysiology of trigeminal autonomic cephalalgias. Lancet Neurol. 2009;8(8):755-764.
27. Goadsby PJ, Raskin NH. Chapter 14. Headache. In: Longo DL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York: McGraw-Hill; 2011:112-128.
28. Bendtsen L, Jensen R. Tension-type headache. Neurol Clin. 2009;27(2):525-535.
29. van Kleef M, Lataster A, Narouze S, et al. Evidenced-based interventional pain medicine according to clinical diagnoses. 2. Cluster headache. Pain Pract. 2009;9(6):435-442.
30. Detsky ME, McDonald DR, Baerlocher MO, et al. Does this patient with headache have a migraine or need neuroimaging? JAMA. 2006; 296(10):1274-1283.
31. Treatment Guideline Subcommittee of the Taiwan Headache Society. Neuroimaging guidelines in nonacute headaches [in Chinese]. Acta Neurol Taiwan. 2010;19(2):137-144.
32. Wilbrink LA, Ferrari MD, Kruit MC, Haan J. Neuroimaging in trigeminal autonomic cephalgias: when, how, and of what? Curr Opin Neurol. 2009;22(3):247-253.
33. Sempere AP, Porta-Etessam J, Medrano V, et al. Neuroimaging in the evaluation of patients with non-acute headache. Cephalalgia. 2005;25(1):30-35.
34. Favier I, van Vliet J, Roon K, et al. Trigeminal autonomic cephalgias due to structural lesions: a review of 31 cases. Arch Neurol. 2007;64(1):25-31.
35. Francis GJ, Becker WJ, Pringsheim TM. Acute and preventive pharmacologic treatment of cluster headache. Neurology. 2010; 75(5):463-473.
36. Bennett MH, French C, Schnabel A, et al. Normobaric and hyperbaric oxygen therapy for migraine and cluster headache. Cochrane Database Syst Rev. 2008 Jul 16;(3):CD005219.
37. Rozen TD. Inhaled oxygen for cluster headache: efficacy, mechanism of action, utilization, and economics. Curr Pain Headache Rep. 2012 Jan 29. [Epub ahead of print]
38. Rozen TD, Fishman RS. Inhaled oxygen and cluster headache sufferers in the United States: use, efficacy and economics: results from the United States Cluster Headache Survey. Headache. 2011;51(2):191-200.
39. Kudrow L. Response of cluster headache attacks to oxygen inhalation. Headache. 1981; 21:1-4.
40. Cohen AS, Burns B, Goadsby PJ. High-flow oxygen for treatment of cluster headache: a randomized trial. JAMA. 2009;302(22):2451-2457.
41. Mahadevan SV. Emergency airway management. In: Auerbach PS. Wilderness Medicine. 6th ed. Stanford, CA: Elsevier; 2011.
42. Göbel H, Lindner V, Heinze A, et al. Acute therapy for cluster headache with sumatriptan: findings of a one-year long-term study. Neurology. 1998;51(3):908-911.
43. Sicuteri F, Geppetti P, Marabini S, Lembeck F. Pain relief by somatostatin in attacks of cluster headache. Pain. 1984;184:359-365.
44. Matharu MS, Levy MJ, Meeran K, Goadsby PJ. Subcutaneous octreotide in cluster headache: randomized placebo-controlled double-blind crossover study. Ann Neurol. 2004;56(4): 488-494.
45. Leone M, D’Amico D, Frediani F, et al. Verapamil in the prophylaxis of episodic cluster headache: a double-blind study versus placebo. Neurology. 2000;54(6):1382-1385.
46. Tfelt-Hansen P, Tfelt-Hansen J. Verapamil for cluster headache: clinical pharmacology and possible mode of action. Headache. 2009;49(1): 117-125.
47. Blau JN, Engel HO. Individualizing treatment with verapamil for cluster headache patients. Headache. 2004;44(10):1013-1018.
48. Leone M, D’Amico D, Moschiano F, et al. Melatonin versus placebo in the prophylaxis of cluster headache: a double-blind pilot study with parallel groups. Cephalalgia. 1996;16(7): 494-496.
49. Sprenger T, Seifert CL, Miederer M, et al. Successful prophylactic treatment of chronic cluster headache with low-dose levomethadone. J Neurol. 2008;255(11):1832-1833.
50. Ambrosini A, Vandenheede M, Rossi P, et al. Suboccipital injection with a mixture of rapid- and long-acting steroids in cluster headache: a double-blind placebo-controlled study. Pain. 2005;118(1-2):92-96.
51. Peres MF, Stiles MA, Siow HC, et al. Greater occipital nerve blockade for cluster headache. Cephalalgia. 2002;22(7):520-522.
52. Leone M, Proietti Cecchini A, Franzini A, et al. Lessons from 8 years’ experience of hypothalamic stimulation in cluster headache. Cephalalgia. 2008;28(7):787-797.
Headache is one of the health problems most commonly associated with significant morbidity, as well as considerable social and economic repercussions.1,2 Headaches are classified into three main types:
- Primary headaches, or headaches without known organic causes
- Secondary headaches, or headaches manifesting with symptoms due to organic causes; and
- Cranial neuralgias, facial pain, and other headaches.3
Cluster headache (CH) is a type of primary headache—one of the headache types encompassed by the term trigeminal autonomic cephalalgia.3 It is one of the most intense, excruciating headaches a patient can experience, and the diagnosis is often missed or delayed. Only 21% of patients receive a correct diagnosis of CH on first presentation, and the average patient visits three health care providers before the correct diagnosis is made.4,5 According to recently published results from the US Cluster Headache Survey,4 the diagnostic delay for CH averages five years or longer, limiting the patient’s access to correct treatment.
Patients with CH are prone to significant physical, social, and economic disability; most patients, for example, find it difficult to work during a CH period.1,2 Almost 20% of patients with CH report having lost a job because of their headaches, and about 8% are unemployed or on disability.4
Because of the pain severity and the associated impairment, the risk for suicide in the CH patient population is real.2,6,7 Jürgens et al7 report that 22% of patients with chronic CH and about 15% of those with episodic CH had suicidal tendencies; Rozen and Fishman4 report suicidal ideation in 55% of CH patients.
CHARACTERISTICS OF CLUSTER HEADACHE
As the name implies, attacks of this headache type tend to “cluster” together. In 85% to 90% of patients, CH is episodic, with cluster periods of headache attacks commonly lasting for one week to one year, and intervening remission periods that may last from one month to years.3,8 The remaining 10% to 15% of CH patients have the chronic CH type, in which cluster periods typically last for more than one year and are separated by remission periods lasting one month or less.3,8
Cluster headaches tend to occur in predictable patterns, often in the spring and fall.9-11 Most headaches begin between early evening and early morning, and patients are often awakened by CH during the night; according to responses in the US Cluster Headache Survey,4 onset times peak between midnight and 3 AM. Attacks can occur when the neck is rotated or flexed in specific ways; external pressure to the transverse processes of C4 or the nerve root of C2 can trigger a CH attack.12
Other CH triggers include alcohol (especially beer and red wine4,13), histamine, nitroglycerine, carbon dioxide, certain odors, and weather changes.3,4,11,14 Eighty percent or more of CH patients have a history of prolonged tobacco use, and at least 60% of CH patients who do not smoke were the children of smokers.15 No clear relationship has been found between CH and hormones.9,10
EPIDEMIOLOGY
Cluster headache is relatively rare, affecting about 0.1% of the population.8-10,16 Onset of the condition usually occurs between ages 20 and 40, and men are three to four times more likely to be affected than are women.3,16 A familial/genetic relationship may exist.10,17,18
DIAGNOSIS
Patient History
Diagnosis of headache relies heavily on the patient’s clinical history and physical exam.3,8,10 A detailed history should include the initial onset of CH, progression of the condition, and information about any precipitating event(s) and prodromal symptoms. Clinicians should document the pattern of pain by including specific information regarding its location, severity, quality, frequency, and duration. Of considerable value is the patient’s use of an accurate headache diary, which clinicians should encourage headache patients to maintain; in these, patients should be instructed to record the headache characteristics mentioned.3,8
Associated symptoms (assessed by conducting a complete review of systems), aggravating and alleviating factors, previous medical history, and psychosocial and family history are important in formulating the differential diagnosis, as misdiagnosis of CH is often related to inadequate history intake.13
Presentation
Cluster headaches share three main features: they are unilateral; they are associated with autonomic symptoms; and attacks tend to “cluster” in a circannual pattern (ie, clusters occurring at the same time of year) and/or circadian pattern (headache at the same time of day).8,19 The most common locations for cluster headaches are unilateral orbital, supraorbital, temporal, or a combination of these locations.3,8
About 30% of patients describe the pain as “stabbing,”3 and it is often compared to “a hot poker in the eye.” Pain peaks rapidly, usually within five to 10 minutes. It may radiate to the ipsilateral forehead, jaw, cheek, and/or teeth.3 Patients appear restless and agitated, unable to lie still.2,10 They often sit, holding their heads, and may pace the floor or bang their heads against the wall.
CH is associated with at least one of the following autonomic symptoms, occurring in the ipsilateral side of the head: conjunctival injection, nasal congestion, forehead and facial sweating, eyelid edema, lacrimation, rhinorrhea, ptosis, and miosis.3,13 Headaches may occur on one side of the head throughout one cluster episode, then shift to the contralateral side in subsequent periods.10 Aura occurs in 14% to 20% of patients,13,20-22 and nausea, as well as ipsilateral
visual, sensory, and speech/language disturbances have also been reported.3 Each CH attack lasts between 15 minutes and three hours, and attacks may range in frequency from one every other day to eight per day.3,13
Patients who have experienced at least five episodes of these headache symptoms, with severe pain in the specified areas and duration, accompanying autonomic symptoms, specified attack frequency, and symptoms not attributed to another disorder meet the diagnostic criteria for cluster headache given in the second edition of the International Classification of Headache Disorders (ICHD-II, 2004).3 The ICHD-II criteria, based on clinical and epidemiologic research, are recognized as a consensus guideline that is accepted worldwide to facilitate clinical practice.3 Patients who have experienced attacks fulfilling all but one of the ICHD-II criteria for CH are diagnosed with probable CH3 or cluster-like headache (CLH).23
Physical Examination
A thorough physical examination, including an investigation of the neurologic system, is essential to differentiate among primary, secondary, and other headache types. In the patient with CH, no neurologic deficits or deficits that suggest underlying disorders are usually found.3,10
Differential Diagnosis
In the evaluation of headache, it is important to differentiate CH from the other trigeminal autonomic cephalalgias: paroxysmal hemicrania (PH), short-lasting unilateral neuralgiform headache with conjunctival injection and tearing (SUNCT), and possibly hemicrania continua.3,24,25 As in CH, the pain of PH is severe, unilateral, and stabbing in quality; it, too, is associated with autonomic symptoms, often occurs at night, and can be episodic or chronic.3 However, PH headache lasts for only 2 to 30 minutes and can occur five times or more per day. Though difficult to distinguish from CH patients, those with PH usually respond to indomethacin, whereas those with CH ordinarily do not.3,8
As in patients with CH, those affected by SUNCT experience autonomic symptoms—most commonly, conjunctival injection and tearing.3,26 SUNCT differs from CH, however, in that the pain is moderate in severity, with a pulsating, burning, electric-like quality. Duration is much shorter, with episodes lasting between 5 seconds and 4 minutes.3,26
Hemicrania continua, though unilateral, is described as continuous and moderate in intensity. Like PH, it is also indomethacin-responsive.8,25,27
A broader differential diagnosis for CH, as detailed in the table,11,3,8,13,26,28 includes the other primary headaches: tension headache, migraine headache, and trigeminal neuralgia.3,26Tension headache, which affects 30% to 78% of the general population,3 is subdivided into infrequent episodic, frequent episodic, and chronic tension-type headache. Unlike CH, tension headache is mild to moderate in intensity and occurs bilaterally, with nonpulsating pressure or a tightening sensation. It is not aggravated by routine physical activity, nor is it associated with nausea, vomiting, or photophobia.
Migraine headache, also a more common primary headache type than CH,3 occurs unilaterally, is moderate to severe in intensity, and is often described as throbbing. More gradual than CH in onset, migraine is often associated with nausea, vomiting, photophobia, phonophobia, and/or visual aura. Migraine headache lacks the ipsilateral autonomic manifestations of CH, and migraineurs prefer to rest or sleep—in contrast to the extreme restlessness or agitation seen in CH patients.3
Also like CH, trigeminal neuralgia is unilateral with a trigeminal nerve distribution, and the pain can be severe and stabbing.3 However, trigeminal neuralgia lacks the autonomic symptoms associated with CH, and the pain lasts from only seconds to minutes. This headache type is often triggered by washing, shaving, or brushing teeth.3
It is also critical to exclude secondary headaches, especially those with serious causes, including meningitis, subarachnoid hemorrhage, epidural or subdural hematoma, glaucoma, tumors, temporal arteritis, or purulent sinusitis.27 Red flags associated with these conditions are:
- A complaint of the patient’s “worst headache ever” (thunderclap headache)
- First severe headache
- A subacute headache worsening over days or weeks
- An abnormal neurologic examination
- Fever or other unexplained systemic signs
- A headache preceded by vomiting
- Headache that is induced by bending, lifting, or coughing
- Headache that disturbs the patient’s sleep or presents immediately upon awakening
- History of known systemic illness
- Headache onset after age 55; and
- Pain associated with local tenderness, for example, near the temporal artery.27
DIAGNOSTIC TESTING
Since, by definition, primary headaches are those without underlying organic causes, diagnostic tests and neuroimaging studies are generally not recommended,10,29 especially when the patient history and presentation confirm the required ICHD-II diagnostic criteria. However, neuroimaging is often recommended for a patient with CH or CH-like presentations.8,23,30-32
In a literature review published in 2006, Detsky et al30 examined the correlation between clinical features of headache (as described in the ICHD-II criteria) and intracranial abnormality (as found on CT or MRI). They found an increased risk for serious intracranial abnormalities among study subjects with cluster-type headache. In any patient with chronic headache, they found, abnormal neurologic findings on physical exam represent the greatest predictor for intracranial pathology.30
In a similar study of 1,872 consecutive patients with nonacute headaches who underwent CT or MRI, one of 20 patients with CH was found to have a pituitary adenoma.33 When Favier et al34 reviewed 31 cases of trigeminal autonomic cephalalgia (TAC), including 10 with atypical symptoms, they found that even typical TAC can result from underlying pathologies with rare warning signs and symptoms. In some patients, neuroimaging study results were normal on initial diagnosis, but pathologies were discovered later after symptoms worsened or treatments ceased to be effective, prompting further imaging studies.
In a review of case studies of patients with CLH, Mainardi et al23 found that of 38 patients who fulfilled the ICHD-II criteria for CH, 12 patients (31.6%) had vascular pathologies, 12 (31.6%) had tumors, and five (13.2%) had inflammatory or infectious pathologies. The researchers recommended that all patients with symptoms of CH or CLH undergo cerebral MRI with contrast medium, even though the yield for abnormal findings would likely be low.23 Wilbrink et al32 also found a wide range of pathologies without typical warning signs or symptoms among 56 case studies of TAC and TAC-like syndromes—and recommended that all such patients be considered candidates for neuroimaging.
Recommendations from both the Scottish Intercollegiate Guidelines Network (SIGN)8 and the Taiwan Headache Society treatment guidelines31 include neuroimaging of patients with CH or CLH.
MANAGEMENT
Clinicians may wish to consider referring patients to a neurologist at the initial diagnosis of CH. Patients with atypical symptomatology or neurologic abnormalities, and those who respond insufficiently to treatment warrant a neurology referral for further investigation.
The two treatment strategies for CH are first, symptomatic treatment for acute attacks; and second, intervention to prevent or reduce further attacks and to shorten the cluster period.8,11,14,35,36
Acute Treatment
Acute symptomatic treatment is aimed at aborting the pain within 15 to 30 minutes from headache onset.14,35,36 Currently, it is generally accepted that 100% oxygen and parenteral triptans (5-HT1B/1D, not through the alimentary tract) are considered first-line treatment options.8,11,13,14,35,36
For the majority of patients (particularly those with episodic CH), inhaled normobaric oxygen effectively relieves CH pain within 15 minutes.14,37-39 Oxygen is administered at 6 to 12 L/min with a nonrebreather mask for at least 15 to 20 minutes.8,14,13 Although associated adverse events are rare, oxygen is inconvenient to transport, and it poses a fire hazard. Additionally, high-flow oxygen is contraindicated in patients with chronic obstructive pulmonary disease, as these patients depend on the hypoxic drive and run the risk of respiratory depression.14,40,41
Triptans, too, have been found effective in the acute treatment of CH; administration by subcutaneous injection or intranasal delivery is considered more effective than the oral route due to faster onset of action,11,14,35 and oxygen use may enhance triptans’ efficacy.38 In two 2010 reviews of the relevant literature, subcutaneous sumatriptan, dosed at either 6 mg or 12 mg, provided effective pain relief within 15 minutes for most patients, with no statistical between-dosage differences.11,35 The most common adverse effects were injection-site reactions, nausea, vomiting, dizziness, fatigue, and paresthesias.35
Intranasal zolmitriptan (5 mg and 10 mg) and intranasal sumatriptan 20 mg were also found effective, with significant pain relief within 30 minutes. Bad taste is a common complaint.35 Of note, both sumatriptan and zolmitriptan are contraindicated in patients with cardiovascular or cerebrovascular disease.14
In an older study of efficacy, safety, and tolerability of subcutaneous sumatriptan, almost 70% of patients averaging between one and six CH attacks per day were found to be using more than the 12-mg maximum recommended daily dosage—as much as 36 mg in a 24-hour period.42 Nevertheless, the researchers concluded that subcutaneous sumatriptan was effective and well tolerated without decreased efficacy over one year in patients with CH.
Ergotamine, once commonly used for the acute treatment of CH, has fallen out of favor in recent years due to its vasoconstrictive effects and serious adverse effects profile.14,35 Dihydroergotamine (DHE) is most effective when administered by IV (though not easily accessible for an acute attack); however, evidence regarding its efficacy and tolerability in other forms is insufficient to recommend DHE for acute CH therapy.14,35
Intranasal lidocaine, somatostatin by infusion, and subcutaneous octreotide are considered second-line treatment choices for patients who are resistant to first-line therapies or who cannot tolerate them.14,35,43,44
Cluster Headache Prophylaxis
A CH period can last for weeks to months. Prophylactic modalities, which are intended to shorten this period and to reduce the frequency and severity of headache attacks, are categorized into transitional and maintenance prophylaxis treatments.14,35
Transitional prophylaxis, a shorter course of treatment, is often started with maintenance prophylaxis (which is used throughout each cluster period) to hasten the response to the maintenance treatment. Corticosteroids are commonly used as a transitional treatment modality. In prednisone use, a starting dose of at least 40 mg/d by mouth is often required to provide benefit.14 The peak dose is usually given for three to 10 days, then gradually tapered over the succeeding 10 to 30 days. Headache recurrence is common during the prednisone taper. Ergotamine tartrate and DHE are also used as transitional prophylaxis treatment for CH.14
Verapamil is considered the maintenance prophylaxis drug of choice due to its efficacy and safety.14,35,45 The dosage required for adequate response ranges from 200 mg to 960 mg/d, in divided doses or in extended-release formulation. Most patients respond to daily doses between 200 mg and 480 mg.14,46,47 Constipation is the most common adverse effect. Slow titration and frequent ECG monitoring, particularly when dosing is increased, are necessary to avoid heart block, bradycardia, hypotension, and peripheral edema.13
Lithium is often used as second-line therapy for maintenance prophylaxis, possibly in combination with verapamil or topiramate, to improve pain control.35 Lithium carbonate, given at a dosage of 600 to 900 mg/d to maintain a serum level of 0.4 to 0.8 mEq/L, and topiramate, at dosages ranging from 50 to 200 mg/d, may be needed to achieve an adequate response.14
In at least one small study, melatonin (10 mg/d) has been associated with CH remission in 50% of treated patients.14,48 It may be used in combination with other prophylactic medications.14
Among numerous other agents that have been used for CH prophylaxis, neither sodium valproate, sumatriptan, cimetidine/chlorpheniramine, misoprostol, nor oxygen is recommended for prevention of CH.35
Narcotics
Because of its excruciating pain, CH has been referred to as “suicide headache.”2,6,7,10,13 Acute and prophylactic treatments for CH will likely reduce the number of headache attacks; however, with CH attacks as frequent as eight times per day, these treatments may not be adequate.24,49
The use of any narcotic is not ideal due to its potential for addiction, and it may cause medication-overuse headache. Furthermore, in oral form, a narcotic may not relieve CH pain quickly enough. Low-dose levomethadone is an opioid that has been used prophylactically with some success in patients with chronic CH.24,49 However, the primary care provider whose CH patient finds pain control inadequate should refer to a neurologist or a pain management specialist for evaluation—and possibly for treatment with an invasive procedure.
Invasive Procedures
Greater occipital nerve block has shown promise in the treatment of CH.50,51 In a small, double-blind study, patients with episodic or chronic CH were randomized to receive a suboccipital injection, of either combined long- and rapid-acting betamethasone or saline (placebo), in the area of the greater occipital nerve.50 Eighty-five percent of treated patients were free of headache attacks within 72 hours, compared with none in the control group. Use of lidocaine with triamcinolone was found somewhat less effective.51
Occipital nerve stimulation has also shown promise for patients with chronic CH who become resistant or are unresponsive to conventional treatments, or who cannot tolerate them.14,24 It appears to induce gradual neuromodulation, with gradual benefits (after six to 30 months). Deep brain stimulation (ie, of the posterior hypothalamus), delivered via implanted electrodes, and other procedures have produced results ranging from “excellent” to “transient remission,” reducing the use of ablative surgeries.14,52 Because invasive modalities carry a risk for serious adverse effects,14 their use should be reserved for a select patient population.
PATIENT/FAMILY EDUCATION
Patients with CH need to be educated regarding the nature, signs and symptoms, and triggers of CH. The indications for acute and prophylactic treatments and the adverse effects associated with each therapy must also be reviewed. Clear follow-up instructions are essential, including what conditions warrant further evaluation: worsening of the condition, changes in symptoms (impaired alertness, vision, movement, or sensation; onset of seizures), or treatment failure.
CONCLUSION
Cluster headache, a relatively uncommon primary headache that can cause excruciating and debilitating pain, is often misdiagnosed and inappropriately treated, with serious physical, social, and economic ramifications. This headache type is unilateral, associated with autonomic symptoms, and characterized by clustering of headache/remission periods in a circannual and/or circadian pattern. Diagnosis is made through the health history and physical exam, based on criteria from the ICHD-II. Neuroimaging may not be necessary, but given the evidence that CH and TAC are often associated with serious underlying pathology, MRI with contrast should be considered, and consultation with a neurologist at initial diagnosis is recommended.
Treatment is aimed at aborting the pain within 15 to 30 minutes of an acute headache attack and preventing further episodes through transitional and maintenance prophylaxis. Newer invasive options that are showing great promise may be considered for a select patient population. Clinicians should involve patients in treatment decisions that will address their individual needs, improving function and optimizing outcomes.
Headache is one of the health problems most commonly associated with significant morbidity, as well as considerable social and economic repercussions.1,2 Headaches are classified into three main types:
- Primary headaches, or headaches without known organic causes
- Secondary headaches, or headaches manifesting with symptoms due to organic causes; and
- Cranial neuralgias, facial pain, and other headaches.3
Cluster headache (CH) is a type of primary headache—one of the headache types encompassed by the term trigeminal autonomic cephalalgia.3 It is one of the most intense, excruciating headaches a patient can experience, and the diagnosis is often missed or delayed. Only 21% of patients receive a correct diagnosis of CH on first presentation, and the average patient visits three health care providers before the correct diagnosis is made.4,5 According to recently published results from the US Cluster Headache Survey,4 the diagnostic delay for CH averages five years or longer, limiting the patient’s access to correct treatment.
Patients with CH are prone to significant physical, social, and economic disability; most patients, for example, find it difficult to work during a CH period.1,2 Almost 20% of patients with CH report having lost a job because of their headaches, and about 8% are unemployed or on disability.4
Because of the pain severity and the associated impairment, the risk for suicide in the CH patient population is real.2,6,7 Jürgens et al7 report that 22% of patients with chronic CH and about 15% of those with episodic CH had suicidal tendencies; Rozen and Fishman4 report suicidal ideation in 55% of CH patients.
CHARACTERISTICS OF CLUSTER HEADACHE
As the name implies, attacks of this headache type tend to “cluster” together. In 85% to 90% of patients, CH is episodic, with cluster periods of headache attacks commonly lasting for one week to one year, and intervening remission periods that may last from one month to years.3,8 The remaining 10% to 15% of CH patients have the chronic CH type, in which cluster periods typically last for more than one year and are separated by remission periods lasting one month or less.3,8
Cluster headaches tend to occur in predictable patterns, often in the spring and fall.9-11 Most headaches begin between early evening and early morning, and patients are often awakened by CH during the night; according to responses in the US Cluster Headache Survey,4 onset times peak between midnight and 3 AM. Attacks can occur when the neck is rotated or flexed in specific ways; external pressure to the transverse processes of C4 or the nerve root of C2 can trigger a CH attack.12
Other CH triggers include alcohol (especially beer and red wine4,13), histamine, nitroglycerine, carbon dioxide, certain odors, and weather changes.3,4,11,14 Eighty percent or more of CH patients have a history of prolonged tobacco use, and at least 60% of CH patients who do not smoke were the children of smokers.15 No clear relationship has been found between CH and hormones.9,10
EPIDEMIOLOGY
Cluster headache is relatively rare, affecting about 0.1% of the population.8-10,16 Onset of the condition usually occurs between ages 20 and 40, and men are three to four times more likely to be affected than are women.3,16 A familial/genetic relationship may exist.10,17,18
DIAGNOSIS
Patient History
Diagnosis of headache relies heavily on the patient’s clinical history and physical exam.3,8,10 A detailed history should include the initial onset of CH, progression of the condition, and information about any precipitating event(s) and prodromal symptoms. Clinicians should document the pattern of pain by including specific information regarding its location, severity, quality, frequency, and duration. Of considerable value is the patient’s use of an accurate headache diary, which clinicians should encourage headache patients to maintain; in these, patients should be instructed to record the headache characteristics mentioned.3,8
Associated symptoms (assessed by conducting a complete review of systems), aggravating and alleviating factors, previous medical history, and psychosocial and family history are important in formulating the differential diagnosis, as misdiagnosis of CH is often related to inadequate history intake.13
Presentation
Cluster headaches share three main features: they are unilateral; they are associated with autonomic symptoms; and attacks tend to “cluster” in a circannual pattern (ie, clusters occurring at the same time of year) and/or circadian pattern (headache at the same time of day).8,19 The most common locations for cluster headaches are unilateral orbital, supraorbital, temporal, or a combination of these locations.3,8
About 30% of patients describe the pain as “stabbing,”3 and it is often compared to “a hot poker in the eye.” Pain peaks rapidly, usually within five to 10 minutes. It may radiate to the ipsilateral forehead, jaw, cheek, and/or teeth.3 Patients appear restless and agitated, unable to lie still.2,10 They often sit, holding their heads, and may pace the floor or bang their heads against the wall.
CH is associated with at least one of the following autonomic symptoms, occurring in the ipsilateral side of the head: conjunctival injection, nasal congestion, forehead and facial sweating, eyelid edema, lacrimation, rhinorrhea, ptosis, and miosis.3,13 Headaches may occur on one side of the head throughout one cluster episode, then shift to the contralateral side in subsequent periods.10 Aura occurs in 14% to 20% of patients,13,20-22 and nausea, as well as ipsilateral
visual, sensory, and speech/language disturbances have also been reported.3 Each CH attack lasts between 15 minutes and three hours, and attacks may range in frequency from one every other day to eight per day.3,13
Patients who have experienced at least five episodes of these headache symptoms, with severe pain in the specified areas and duration, accompanying autonomic symptoms, specified attack frequency, and symptoms not attributed to another disorder meet the diagnostic criteria for cluster headache given in the second edition of the International Classification of Headache Disorders (ICHD-II, 2004).3 The ICHD-II criteria, based on clinical and epidemiologic research, are recognized as a consensus guideline that is accepted worldwide to facilitate clinical practice.3 Patients who have experienced attacks fulfilling all but one of the ICHD-II criteria for CH are diagnosed with probable CH3 or cluster-like headache (CLH).23
Physical Examination
A thorough physical examination, including an investigation of the neurologic system, is essential to differentiate among primary, secondary, and other headache types. In the patient with CH, no neurologic deficits or deficits that suggest underlying disorders are usually found.3,10
Differential Diagnosis
In the evaluation of headache, it is important to differentiate CH from the other trigeminal autonomic cephalalgias: paroxysmal hemicrania (PH), short-lasting unilateral neuralgiform headache with conjunctival injection and tearing (SUNCT), and possibly hemicrania continua.3,24,25 As in CH, the pain of PH is severe, unilateral, and stabbing in quality; it, too, is associated with autonomic symptoms, often occurs at night, and can be episodic or chronic.3 However, PH headache lasts for only 2 to 30 minutes and can occur five times or more per day. Though difficult to distinguish from CH patients, those with PH usually respond to indomethacin, whereas those with CH ordinarily do not.3,8
As in patients with CH, those affected by SUNCT experience autonomic symptoms—most commonly, conjunctival injection and tearing.3,26 SUNCT differs from CH, however, in that the pain is moderate in severity, with a pulsating, burning, electric-like quality. Duration is much shorter, with episodes lasting between 5 seconds and 4 minutes.3,26
Hemicrania continua, though unilateral, is described as continuous and moderate in intensity. Like PH, it is also indomethacin-responsive.8,25,27
A broader differential diagnosis for CH, as detailed in the table,11,3,8,13,26,28 includes the other primary headaches: tension headache, migraine headache, and trigeminal neuralgia.3,26Tension headache, which affects 30% to 78% of the general population,3 is subdivided into infrequent episodic, frequent episodic, and chronic tension-type headache. Unlike CH, tension headache is mild to moderate in intensity and occurs bilaterally, with nonpulsating pressure or a tightening sensation. It is not aggravated by routine physical activity, nor is it associated with nausea, vomiting, or photophobia.
Migraine headache, also a more common primary headache type than CH,3 occurs unilaterally, is moderate to severe in intensity, and is often described as throbbing. More gradual than CH in onset, migraine is often associated with nausea, vomiting, photophobia, phonophobia, and/or visual aura. Migraine headache lacks the ipsilateral autonomic manifestations of CH, and migraineurs prefer to rest or sleep—in contrast to the extreme restlessness or agitation seen in CH patients.3
Also like CH, trigeminal neuralgia is unilateral with a trigeminal nerve distribution, and the pain can be severe and stabbing.3 However, trigeminal neuralgia lacks the autonomic symptoms associated with CH, and the pain lasts from only seconds to minutes. This headache type is often triggered by washing, shaving, or brushing teeth.3
It is also critical to exclude secondary headaches, especially those with serious causes, including meningitis, subarachnoid hemorrhage, epidural or subdural hematoma, glaucoma, tumors, temporal arteritis, or purulent sinusitis.27 Red flags associated with these conditions are:
- A complaint of the patient’s “worst headache ever” (thunderclap headache)
- First severe headache
- A subacute headache worsening over days or weeks
- An abnormal neurologic examination
- Fever or other unexplained systemic signs
- A headache preceded by vomiting
- Headache that is induced by bending, lifting, or coughing
- Headache that disturbs the patient’s sleep or presents immediately upon awakening
- History of known systemic illness
- Headache onset after age 55; and
- Pain associated with local tenderness, for example, near the temporal artery.27
DIAGNOSTIC TESTING
Since, by definition, primary headaches are those without underlying organic causes, diagnostic tests and neuroimaging studies are generally not recommended,10,29 especially when the patient history and presentation confirm the required ICHD-II diagnostic criteria. However, neuroimaging is often recommended for a patient with CH or CH-like presentations.8,23,30-32
In a literature review published in 2006, Detsky et al30 examined the correlation between clinical features of headache (as described in the ICHD-II criteria) and intracranial abnormality (as found on CT or MRI). They found an increased risk for serious intracranial abnormalities among study subjects with cluster-type headache. In any patient with chronic headache, they found, abnormal neurologic findings on physical exam represent the greatest predictor for intracranial pathology.30
In a similar study of 1,872 consecutive patients with nonacute headaches who underwent CT or MRI, one of 20 patients with CH was found to have a pituitary adenoma.33 When Favier et al34 reviewed 31 cases of trigeminal autonomic cephalalgia (TAC), including 10 with atypical symptoms, they found that even typical TAC can result from underlying pathologies with rare warning signs and symptoms. In some patients, neuroimaging study results were normal on initial diagnosis, but pathologies were discovered later after symptoms worsened or treatments ceased to be effective, prompting further imaging studies.
In a review of case studies of patients with CLH, Mainardi et al23 found that of 38 patients who fulfilled the ICHD-II criteria for CH, 12 patients (31.6%) had vascular pathologies, 12 (31.6%) had tumors, and five (13.2%) had inflammatory or infectious pathologies. The researchers recommended that all patients with symptoms of CH or CLH undergo cerebral MRI with contrast medium, even though the yield for abnormal findings would likely be low.23 Wilbrink et al32 also found a wide range of pathologies without typical warning signs or symptoms among 56 case studies of TAC and TAC-like syndromes—and recommended that all such patients be considered candidates for neuroimaging.
Recommendations from both the Scottish Intercollegiate Guidelines Network (SIGN)8 and the Taiwan Headache Society treatment guidelines31 include neuroimaging of patients with CH or CLH.
MANAGEMENT
Clinicians may wish to consider referring patients to a neurologist at the initial diagnosis of CH. Patients with atypical symptomatology or neurologic abnormalities, and those who respond insufficiently to treatment warrant a neurology referral for further investigation.
The two treatment strategies for CH are first, symptomatic treatment for acute attacks; and second, intervention to prevent or reduce further attacks and to shorten the cluster period.8,11,14,35,36
Acute Treatment
Acute symptomatic treatment is aimed at aborting the pain within 15 to 30 minutes from headache onset.14,35,36 Currently, it is generally accepted that 100% oxygen and parenteral triptans (5-HT1B/1D, not through the alimentary tract) are considered first-line treatment options.8,11,13,14,35,36
For the majority of patients (particularly those with episodic CH), inhaled normobaric oxygen effectively relieves CH pain within 15 minutes.14,37-39 Oxygen is administered at 6 to 12 L/min with a nonrebreather mask for at least 15 to 20 minutes.8,14,13 Although associated adverse events are rare, oxygen is inconvenient to transport, and it poses a fire hazard. Additionally, high-flow oxygen is contraindicated in patients with chronic obstructive pulmonary disease, as these patients depend on the hypoxic drive and run the risk of respiratory depression.14,40,41
Triptans, too, have been found effective in the acute treatment of CH; administration by subcutaneous injection or intranasal delivery is considered more effective than the oral route due to faster onset of action,11,14,35 and oxygen use may enhance triptans’ efficacy.38 In two 2010 reviews of the relevant literature, subcutaneous sumatriptan, dosed at either 6 mg or 12 mg, provided effective pain relief within 15 minutes for most patients, with no statistical between-dosage differences.11,35 The most common adverse effects were injection-site reactions, nausea, vomiting, dizziness, fatigue, and paresthesias.35
Intranasal zolmitriptan (5 mg and 10 mg) and intranasal sumatriptan 20 mg were also found effective, with significant pain relief within 30 minutes. Bad taste is a common complaint.35 Of note, both sumatriptan and zolmitriptan are contraindicated in patients with cardiovascular or cerebrovascular disease.14
In an older study of efficacy, safety, and tolerability of subcutaneous sumatriptan, almost 70% of patients averaging between one and six CH attacks per day were found to be using more than the 12-mg maximum recommended daily dosage—as much as 36 mg in a 24-hour period.42 Nevertheless, the researchers concluded that subcutaneous sumatriptan was effective and well tolerated without decreased efficacy over one year in patients with CH.
Ergotamine, once commonly used for the acute treatment of CH, has fallen out of favor in recent years due to its vasoconstrictive effects and serious adverse effects profile.14,35 Dihydroergotamine (DHE) is most effective when administered by IV (though not easily accessible for an acute attack); however, evidence regarding its efficacy and tolerability in other forms is insufficient to recommend DHE for acute CH therapy.14,35
Intranasal lidocaine, somatostatin by infusion, and subcutaneous octreotide are considered second-line treatment choices for patients who are resistant to first-line therapies or who cannot tolerate them.14,35,43,44
Cluster Headache Prophylaxis
A CH period can last for weeks to months. Prophylactic modalities, which are intended to shorten this period and to reduce the frequency and severity of headache attacks, are categorized into transitional and maintenance prophylaxis treatments.14,35
Transitional prophylaxis, a shorter course of treatment, is often started with maintenance prophylaxis (which is used throughout each cluster period) to hasten the response to the maintenance treatment. Corticosteroids are commonly used as a transitional treatment modality. In prednisone use, a starting dose of at least 40 mg/d by mouth is often required to provide benefit.14 The peak dose is usually given for three to 10 days, then gradually tapered over the succeeding 10 to 30 days. Headache recurrence is common during the prednisone taper. Ergotamine tartrate and DHE are also used as transitional prophylaxis treatment for CH.14
Verapamil is considered the maintenance prophylaxis drug of choice due to its efficacy and safety.14,35,45 The dosage required for adequate response ranges from 200 mg to 960 mg/d, in divided doses or in extended-release formulation. Most patients respond to daily doses between 200 mg and 480 mg.14,46,47 Constipation is the most common adverse effect. Slow titration and frequent ECG monitoring, particularly when dosing is increased, are necessary to avoid heart block, bradycardia, hypotension, and peripheral edema.13
Lithium is often used as second-line therapy for maintenance prophylaxis, possibly in combination with verapamil or topiramate, to improve pain control.35 Lithium carbonate, given at a dosage of 600 to 900 mg/d to maintain a serum level of 0.4 to 0.8 mEq/L, and topiramate, at dosages ranging from 50 to 200 mg/d, may be needed to achieve an adequate response.14
In at least one small study, melatonin (10 mg/d) has been associated with CH remission in 50% of treated patients.14,48 It may be used in combination with other prophylactic medications.14
Among numerous other agents that have been used for CH prophylaxis, neither sodium valproate, sumatriptan, cimetidine/chlorpheniramine, misoprostol, nor oxygen is recommended for prevention of CH.35
Narcotics
Because of its excruciating pain, CH has been referred to as “suicide headache.”2,6,7,10,13 Acute and prophylactic treatments for CH will likely reduce the number of headache attacks; however, with CH attacks as frequent as eight times per day, these treatments may not be adequate.24,49
The use of any narcotic is not ideal due to its potential for addiction, and it may cause medication-overuse headache. Furthermore, in oral form, a narcotic may not relieve CH pain quickly enough. Low-dose levomethadone is an opioid that has been used prophylactically with some success in patients with chronic CH.24,49 However, the primary care provider whose CH patient finds pain control inadequate should refer to a neurologist or a pain management specialist for evaluation—and possibly for treatment with an invasive procedure.
Invasive Procedures
Greater occipital nerve block has shown promise in the treatment of CH.50,51 In a small, double-blind study, patients with episodic or chronic CH were randomized to receive a suboccipital injection, of either combined long- and rapid-acting betamethasone or saline (placebo), in the area of the greater occipital nerve.50 Eighty-five percent of treated patients were free of headache attacks within 72 hours, compared with none in the control group. Use of lidocaine with triamcinolone was found somewhat less effective.51
Occipital nerve stimulation has also shown promise for patients with chronic CH who become resistant or are unresponsive to conventional treatments, or who cannot tolerate them.14,24 It appears to induce gradual neuromodulation, with gradual benefits (after six to 30 months). Deep brain stimulation (ie, of the posterior hypothalamus), delivered via implanted electrodes, and other procedures have produced results ranging from “excellent” to “transient remission,” reducing the use of ablative surgeries.14,52 Because invasive modalities carry a risk for serious adverse effects,14 their use should be reserved for a select patient population.
PATIENT/FAMILY EDUCATION
Patients with CH need to be educated regarding the nature, signs and symptoms, and triggers of CH. The indications for acute and prophylactic treatments and the adverse effects associated with each therapy must also be reviewed. Clear follow-up instructions are essential, including what conditions warrant further evaluation: worsening of the condition, changes in symptoms (impaired alertness, vision, movement, or sensation; onset of seizures), or treatment failure.
CONCLUSION
Cluster headache, a relatively uncommon primary headache that can cause excruciating and debilitating pain, is often misdiagnosed and inappropriately treated, with serious physical, social, and economic ramifications. This headache type is unilateral, associated with autonomic symptoms, and characterized by clustering of headache/remission periods in a circannual and/or circadian pattern. Diagnosis is made through the health history and physical exam, based on criteria from the ICHD-II. Neuroimaging may not be necessary, but given the evidence that CH and TAC are often associated with serious underlying pathology, MRI with contrast should be considered, and consultation with a neurologist at initial diagnosis is recommended.
Treatment is aimed at aborting the pain within 15 to 30 minutes of an acute headache attack and preventing further episodes through transitional and maintenance prophylaxis. Newer invasive options that are showing great promise may be considered for a select patient population. Clinicians should involve patients in treatment decisions that will address their individual needs, improving function and optimizing outcomes.
1. Stovner LJ, Hagen H, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia. 2007;27(3):193-210.
2. Vaughan R. My own private purgatory: how cluster headaches affect my life. Headache. 2008;48(10):1541-1543.
3. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders. 2nd ed. Cephalalgia. 2004;24 suppl 1:9-160.
4. Rozen TD, Fishman RS. Cluster headache in the United States of America: demographics, clinical characteristics, triggers, suicidality, and personal burden. Headache. 2012;52(1): 99-113.
5. Bahra A, Goadsby PJ. Diagnostic delays and mis-management in cluster headache. Acta Neurol Scand. 2004;109(3):175-179.
6. Dousset V, Laporte A, Legoff M, et al. Validation of a brief self-administered questionnaire for cluster headache screening in a tertiary center. Headache. 2009;49(1):64-70.
7. Jürgens TP, Gaul C, Lindwurm A, et al. Impairment in episodic and chronic cluster headache. Cephalalgia. 2011;31(6):671-682.
8. Scottish Intercollegiate Guidelines Network (SIGN). Diagnosis and management of headache in adults: a national clinical guideline. Edinburgh (Scotland): Scottish Intercollegiate Guidelines Network (SIGN). 2008;1-81. (SIGN publication; no. 107). www.sign.ac.uk/guide lines/fulltext/107/index.html. Accessed May 8, 2012.
9. Bahra A, May A, Goadsby PJ. Cluster headache: a prospective clinical study with diagnostic implications. Neurology. 2002;58(3): 354-361.
10. May A. Cluster headache: pathogenesis, diagnosis, and management. Lancet. 2005; 366(9488):843-855.
11. Law S, Derry S, Moore RA. Triptans for acute cluster headache. Cochrane Database Syst Rev. 2010 Apr 14;(4):CD008042.
12. Rozen TD. Trigeminal autonomic cephalalgias. Neurol Clin. 2009;27(2):537-556.
13. Nesbitt AD, Goadsby PJ. Cluster headache. BMJ. 2012;344:e2407. doi: 10.1136/bmj
.e2407.
14. Ashkenazi A, Schwedt T. Cluster headache: acute and prophylactic therapy. Headache. 2011;51(2):272-286.
15. Rozen TD. Cluster headache as the result of secondhand cigarette smoke exposure during childhood. Headache. 2010;50(1):130-132.
16. Fischera M, Marziniak M, Gralow I, Evers S. The incidence and prevalence of cluster headache: a meta-analysis of population-based studies. Cephalalgia. 2008;28(6):614-618.
17. De Simone R, Fiorillo C, Bonuso S, Castaldo G. A cluster headache family with possible autosomal recessive inheritance. Neurology. 2003;61(4):578-579.
18. Leone M, Russell MB, Rigamonti A, et al. Increased familial risk of cluster headache. Neurology. 2001;56(9):1233-1236.
19. Matharu MS, Boes CJ, Goadsby PJ. Management of trigeminal autonomic cephalgias and hemicrania continua. Drugs. 2003;63(16): 1637-1677.
20. Evans RW, Krymchantowski AV. Cluster and other nonmigraine primary headaches with aura. Headache. 2011;51(4):604-608.
21. Rozen TD. Cluster headache with aura. Curr Pain Headache Rep. 2011;15(2):98-100.
22. Silberstein SD, Niknam R, Rozen TD, Young WB. Cluster headache with aura. Neurology. 2000;54(1):219-221.
23. Mainardi F, Trucco M, Maggioni F, et al. Cluster-like headache: a comprehensive reappraisal. Cephalalgia. 2010;30(4):399-412.
24. Magis D, Bruno MA, Fumal A, et al. Central modulation in cluster headache patients treated with occipital nerve stimulation: an FDG-PET study. BMC Neurol. 2011;11:25.
25. Goadsby PJ, Lipton RB. A review of paroxysmal hemicranias, SUNCT syndrome and other short-lasting headaches with autonomic feature, including new cases. Brain. 1997; 120(pt 1):193-209.
26. Leone M, Bussone G. Pathophysiology of trigeminal autonomic cephalalgias. Lancet Neurol. 2009;8(8):755-764.
27. Goadsby PJ, Raskin NH. Chapter 14. Headache. In: Longo DL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York: McGraw-Hill; 2011:112-128.
28. Bendtsen L, Jensen R. Tension-type headache. Neurol Clin. 2009;27(2):525-535.
29. van Kleef M, Lataster A, Narouze S, et al. Evidenced-based interventional pain medicine according to clinical diagnoses. 2. Cluster headache. Pain Pract. 2009;9(6):435-442.
30. Detsky ME, McDonald DR, Baerlocher MO, et al. Does this patient with headache have a migraine or need neuroimaging? JAMA. 2006; 296(10):1274-1283.
31. Treatment Guideline Subcommittee of the Taiwan Headache Society. Neuroimaging guidelines in nonacute headaches [in Chinese]. Acta Neurol Taiwan. 2010;19(2):137-144.
32. Wilbrink LA, Ferrari MD, Kruit MC, Haan J. Neuroimaging in trigeminal autonomic cephalgias: when, how, and of what? Curr Opin Neurol. 2009;22(3):247-253.
33. Sempere AP, Porta-Etessam J, Medrano V, et al. Neuroimaging in the evaluation of patients with non-acute headache. Cephalalgia. 2005;25(1):30-35.
34. Favier I, van Vliet J, Roon K, et al. Trigeminal autonomic cephalgias due to structural lesions: a review of 31 cases. Arch Neurol. 2007;64(1):25-31.
35. Francis GJ, Becker WJ, Pringsheim TM. Acute and preventive pharmacologic treatment of cluster headache. Neurology. 2010; 75(5):463-473.
36. Bennett MH, French C, Schnabel A, et al. Normobaric and hyperbaric oxygen therapy for migraine and cluster headache. Cochrane Database Syst Rev. 2008 Jul 16;(3):CD005219.
37. Rozen TD. Inhaled oxygen for cluster headache: efficacy, mechanism of action, utilization, and economics. Curr Pain Headache Rep. 2012 Jan 29. [Epub ahead of print]
38. Rozen TD, Fishman RS. Inhaled oxygen and cluster headache sufferers in the United States: use, efficacy and economics: results from the United States Cluster Headache Survey. Headache. 2011;51(2):191-200.
39. Kudrow L. Response of cluster headache attacks to oxygen inhalation. Headache. 1981; 21:1-4.
40. Cohen AS, Burns B, Goadsby PJ. High-flow oxygen for treatment of cluster headache: a randomized trial. JAMA. 2009;302(22):2451-2457.
41. Mahadevan SV. Emergency airway management. In: Auerbach PS. Wilderness Medicine. 6th ed. Stanford, CA: Elsevier; 2011.
42. Göbel H, Lindner V, Heinze A, et al. Acute therapy for cluster headache with sumatriptan: findings of a one-year long-term study. Neurology. 1998;51(3):908-911.
43. Sicuteri F, Geppetti P, Marabini S, Lembeck F. Pain relief by somatostatin in attacks of cluster headache. Pain. 1984;184:359-365.
44. Matharu MS, Levy MJ, Meeran K, Goadsby PJ. Subcutaneous octreotide in cluster headache: randomized placebo-controlled double-blind crossover study. Ann Neurol. 2004;56(4): 488-494.
45. Leone M, D’Amico D, Frediani F, et al. Verapamil in the prophylaxis of episodic cluster headache: a double-blind study versus placebo. Neurology. 2000;54(6):1382-1385.
46. Tfelt-Hansen P, Tfelt-Hansen J. Verapamil for cluster headache: clinical pharmacology and possible mode of action. Headache. 2009;49(1): 117-125.
47. Blau JN, Engel HO. Individualizing treatment with verapamil for cluster headache patients. Headache. 2004;44(10):1013-1018.
48. Leone M, D’Amico D, Moschiano F, et al. Melatonin versus placebo in the prophylaxis of cluster headache: a double-blind pilot study with parallel groups. Cephalalgia. 1996;16(7): 494-496.
49. Sprenger T, Seifert CL, Miederer M, et al. Successful prophylactic treatment of chronic cluster headache with low-dose levomethadone. J Neurol. 2008;255(11):1832-1833.
50. Ambrosini A, Vandenheede M, Rossi P, et al. Suboccipital injection with a mixture of rapid- and long-acting steroids in cluster headache: a double-blind placebo-controlled study. Pain. 2005;118(1-2):92-96.
51. Peres MF, Stiles MA, Siow HC, et al. Greater occipital nerve blockade for cluster headache. Cephalalgia. 2002;22(7):520-522.
52. Leone M, Proietti Cecchini A, Franzini A, et al. Lessons from 8 years’ experience of hypothalamic stimulation in cluster headache. Cephalalgia. 2008;28(7):787-797.
1. Stovner LJ, Hagen H, Jensen R, et al. The global burden of headache: a documentation of headache prevalence and disability worldwide. Cephalalgia. 2007;27(3):193-210.
2. Vaughan R. My own private purgatory: how cluster headaches affect my life. Headache. 2008;48(10):1541-1543.
3. Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders. 2nd ed. Cephalalgia. 2004;24 suppl 1:9-160.
4. Rozen TD, Fishman RS. Cluster headache in the United States of America: demographics, clinical characteristics, triggers, suicidality, and personal burden. Headache. 2012;52(1): 99-113.
5. Bahra A, Goadsby PJ. Diagnostic delays and mis-management in cluster headache. Acta Neurol Scand. 2004;109(3):175-179.
6. Dousset V, Laporte A, Legoff M, et al. Validation of a brief self-administered questionnaire for cluster headache screening in a tertiary center. Headache. 2009;49(1):64-70.
7. Jürgens TP, Gaul C, Lindwurm A, et al. Impairment in episodic and chronic cluster headache. Cephalalgia. 2011;31(6):671-682.
8. Scottish Intercollegiate Guidelines Network (SIGN). Diagnosis and management of headache in adults: a national clinical guideline. Edinburgh (Scotland): Scottish Intercollegiate Guidelines Network (SIGN). 2008;1-81. (SIGN publication; no. 107). www.sign.ac.uk/guide lines/fulltext/107/index.html. Accessed May 8, 2012.
9. Bahra A, May A, Goadsby PJ. Cluster headache: a prospective clinical study with diagnostic implications. Neurology. 2002;58(3): 354-361.
10. May A. Cluster headache: pathogenesis, diagnosis, and management. Lancet. 2005; 366(9488):843-855.
11. Law S, Derry S, Moore RA. Triptans for acute cluster headache. Cochrane Database Syst Rev. 2010 Apr 14;(4):CD008042.
12. Rozen TD. Trigeminal autonomic cephalalgias. Neurol Clin. 2009;27(2):537-556.
13. Nesbitt AD, Goadsby PJ. Cluster headache. BMJ. 2012;344:e2407. doi: 10.1136/bmj
.e2407.
14. Ashkenazi A, Schwedt T. Cluster headache: acute and prophylactic therapy. Headache. 2011;51(2):272-286.
15. Rozen TD. Cluster headache as the result of secondhand cigarette smoke exposure during childhood. Headache. 2010;50(1):130-132.
16. Fischera M, Marziniak M, Gralow I, Evers S. The incidence and prevalence of cluster headache: a meta-analysis of population-based studies. Cephalalgia. 2008;28(6):614-618.
17. De Simone R, Fiorillo C, Bonuso S, Castaldo G. A cluster headache family with possible autosomal recessive inheritance. Neurology. 2003;61(4):578-579.
18. Leone M, Russell MB, Rigamonti A, et al. Increased familial risk of cluster headache. Neurology. 2001;56(9):1233-1236.
19. Matharu MS, Boes CJ, Goadsby PJ. Management of trigeminal autonomic cephalgias and hemicrania continua. Drugs. 2003;63(16): 1637-1677.
20. Evans RW, Krymchantowski AV. Cluster and other nonmigraine primary headaches with aura. Headache. 2011;51(4):604-608.
21. Rozen TD. Cluster headache with aura. Curr Pain Headache Rep. 2011;15(2):98-100.
22. Silberstein SD, Niknam R, Rozen TD, Young WB. Cluster headache with aura. Neurology. 2000;54(1):219-221.
23. Mainardi F, Trucco M, Maggioni F, et al. Cluster-like headache: a comprehensive reappraisal. Cephalalgia. 2010;30(4):399-412.
24. Magis D, Bruno MA, Fumal A, et al. Central modulation in cluster headache patients treated with occipital nerve stimulation: an FDG-PET study. BMC Neurol. 2011;11:25.
25. Goadsby PJ, Lipton RB. A review of paroxysmal hemicranias, SUNCT syndrome and other short-lasting headaches with autonomic feature, including new cases. Brain. 1997; 120(pt 1):193-209.
26. Leone M, Bussone G. Pathophysiology of trigeminal autonomic cephalalgias. Lancet Neurol. 2009;8(8):755-764.
27. Goadsby PJ, Raskin NH. Chapter 14. Headache. In: Longo DL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York: McGraw-Hill; 2011:112-128.
28. Bendtsen L, Jensen R. Tension-type headache. Neurol Clin. 2009;27(2):525-535.
29. van Kleef M, Lataster A, Narouze S, et al. Evidenced-based interventional pain medicine according to clinical diagnoses. 2. Cluster headache. Pain Pract. 2009;9(6):435-442.
30. Detsky ME, McDonald DR, Baerlocher MO, et al. Does this patient with headache have a migraine or need neuroimaging? JAMA. 2006; 296(10):1274-1283.
31. Treatment Guideline Subcommittee of the Taiwan Headache Society. Neuroimaging guidelines in nonacute headaches [in Chinese]. Acta Neurol Taiwan. 2010;19(2):137-144.
32. Wilbrink LA, Ferrari MD, Kruit MC, Haan J. Neuroimaging in trigeminal autonomic cephalgias: when, how, and of what? Curr Opin Neurol. 2009;22(3):247-253.
33. Sempere AP, Porta-Etessam J, Medrano V, et al. Neuroimaging in the evaluation of patients with non-acute headache. Cephalalgia. 2005;25(1):30-35.
34. Favier I, van Vliet J, Roon K, et al. Trigeminal autonomic cephalgias due to structural lesions: a review of 31 cases. Arch Neurol. 2007;64(1):25-31.
35. Francis GJ, Becker WJ, Pringsheim TM. Acute and preventive pharmacologic treatment of cluster headache. Neurology. 2010; 75(5):463-473.
36. Bennett MH, French C, Schnabel A, et al. Normobaric and hyperbaric oxygen therapy for migraine and cluster headache. Cochrane Database Syst Rev. 2008 Jul 16;(3):CD005219.
37. Rozen TD. Inhaled oxygen for cluster headache: efficacy, mechanism of action, utilization, and economics. Curr Pain Headache Rep. 2012 Jan 29. [Epub ahead of print]
38. Rozen TD, Fishman RS. Inhaled oxygen and cluster headache sufferers in the United States: use, efficacy and economics: results from the United States Cluster Headache Survey. Headache. 2011;51(2):191-200.
39. Kudrow L. Response of cluster headache attacks to oxygen inhalation. Headache. 1981; 21:1-4.
40. Cohen AS, Burns B, Goadsby PJ. High-flow oxygen for treatment of cluster headache: a randomized trial. JAMA. 2009;302(22):2451-2457.
41. Mahadevan SV. Emergency airway management. In: Auerbach PS. Wilderness Medicine. 6th ed. Stanford, CA: Elsevier; 2011.
42. Göbel H, Lindner V, Heinze A, et al. Acute therapy for cluster headache with sumatriptan: findings of a one-year long-term study. Neurology. 1998;51(3):908-911.
43. Sicuteri F, Geppetti P, Marabini S, Lembeck F. Pain relief by somatostatin in attacks of cluster headache. Pain. 1984;184:359-365.
44. Matharu MS, Levy MJ, Meeran K, Goadsby PJ. Subcutaneous octreotide in cluster headache: randomized placebo-controlled double-blind crossover study. Ann Neurol. 2004;56(4): 488-494.
45. Leone M, D’Amico D, Frediani F, et al. Verapamil in the prophylaxis of episodic cluster headache: a double-blind study versus placebo. Neurology. 2000;54(6):1382-1385.
46. Tfelt-Hansen P, Tfelt-Hansen J. Verapamil for cluster headache: clinical pharmacology and possible mode of action. Headache. 2009;49(1): 117-125.
47. Blau JN, Engel HO. Individualizing treatment with verapamil for cluster headache patients. Headache. 2004;44(10):1013-1018.
48. Leone M, D’Amico D, Moschiano F, et al. Melatonin versus placebo in the prophylaxis of cluster headache: a double-blind pilot study with parallel groups. Cephalalgia. 1996;16(7): 494-496.
49. Sprenger T, Seifert CL, Miederer M, et al. Successful prophylactic treatment of chronic cluster headache with low-dose levomethadone. J Neurol. 2008;255(11):1832-1833.
50. Ambrosini A, Vandenheede M, Rossi P, et al. Suboccipital injection with a mixture of rapid- and long-acting steroids in cluster headache: a double-blind placebo-controlled study. Pain. 2005;118(1-2):92-96.
51. Peres MF, Stiles MA, Siow HC, et al. Greater occipital nerve blockade for cluster headache. Cephalalgia. 2002;22(7):520-522.
52. Leone M, Proietti Cecchini A, Franzini A, et al. Lessons from 8 years’ experience of hypothalamic stimulation in cluster headache. Cephalalgia. 2008;28(7):787-797.
End-of-Life Discussions Don’t Decrease Rate of Survival
Engaging in advance-care-planning discussions with their physicians or having advance directives filed in their medical records resulted in no significant difference in survival time for patients at three Colorado hospitals, according to a report in the Journal of Hospital Medicine.1
A total of 458 adult patients admitted to general IM services at the hospitals were asked whether they’d had discussions with their physicians about advance directives, which are legal documents allowing patients to spell out treatment preferences (including a desire for more aggressive treatment) in advance of situations in which they are no longer able to communicate them. Charts were reviewed for the presence of advance directives, and the patients were then stratified based on low, medium, or high risk of death within a year. The high-risk patients were excluded from the study, and those in the low- and medium-risk groups were followed from 2003 to 2009.
“In regard to the current national debate about the merits of advance-care planning, this study suggests that honoring patients’ wishes to engage in advance directive discussions and documentation does not lead to harm,” the study concludes.
Lead author Stacy Fischer, MD, of the University of Colorado Denver says that it is striking how few hospitalized patients have actually engaged in these conversations, even though the population is quite ill. “So often, the conversation happens too late,” she says, “and then not with the patient but with a surrogate.”
Dr. Fischer encourages hospitalists to view the hospital admission as an important opportunity to start conversations with patients about their future care preferences. When patients come into the hospital, they must be asked about advance directives, but that process tends to be cursory, she says. At a minimum, hospitalists should clarify who the surrogate decision maker is, who would speak for the patient at a time of incapacity.
What should the hospitalist’s role be in end-of-life discussions? “That’s a complicated question in the current environment, where nobody seems to think it’s their role,” Dr. Fischer says. “I believe we all need to help move the conversation along. If [advance directive] forms can be available on the floor and if patients express interest in them, then encouraging them would be important.”
Larry Beresford is a freelance writer in Oakland, Calif.
References
Engaging in advance-care-planning discussions with their physicians or having advance directives filed in their medical records resulted in no significant difference in survival time for patients at three Colorado hospitals, according to a report in the Journal of Hospital Medicine.1
A total of 458 adult patients admitted to general IM services at the hospitals were asked whether they’d had discussions with their physicians about advance directives, which are legal documents allowing patients to spell out treatment preferences (including a desire for more aggressive treatment) in advance of situations in which they are no longer able to communicate them. Charts were reviewed for the presence of advance directives, and the patients were then stratified based on low, medium, or high risk of death within a year. The high-risk patients were excluded from the study, and those in the low- and medium-risk groups were followed from 2003 to 2009.
“In regard to the current national debate about the merits of advance-care planning, this study suggests that honoring patients’ wishes to engage in advance directive discussions and documentation does not lead to harm,” the study concludes.
Lead author Stacy Fischer, MD, of the University of Colorado Denver says that it is striking how few hospitalized patients have actually engaged in these conversations, even though the population is quite ill. “So often, the conversation happens too late,” she says, “and then not with the patient but with a surrogate.”
Dr. Fischer encourages hospitalists to view the hospital admission as an important opportunity to start conversations with patients about their future care preferences. When patients come into the hospital, they must be asked about advance directives, but that process tends to be cursory, she says. At a minimum, hospitalists should clarify who the surrogate decision maker is, who would speak for the patient at a time of incapacity.
What should the hospitalist’s role be in end-of-life discussions? “That’s a complicated question in the current environment, where nobody seems to think it’s their role,” Dr. Fischer says. “I believe we all need to help move the conversation along. If [advance directive] forms can be available on the floor and if patients express interest in them, then encouraging them would be important.”
Larry Beresford is a freelance writer in Oakland, Calif.
References
Engaging in advance-care-planning discussions with their physicians or having advance directives filed in their medical records resulted in no significant difference in survival time for patients at three Colorado hospitals, according to a report in the Journal of Hospital Medicine.1
A total of 458 adult patients admitted to general IM services at the hospitals were asked whether they’d had discussions with their physicians about advance directives, which are legal documents allowing patients to spell out treatment preferences (including a desire for more aggressive treatment) in advance of situations in which they are no longer able to communicate them. Charts were reviewed for the presence of advance directives, and the patients were then stratified based on low, medium, or high risk of death within a year. The high-risk patients were excluded from the study, and those in the low- and medium-risk groups were followed from 2003 to 2009.
“In regard to the current national debate about the merits of advance-care planning, this study suggests that honoring patients’ wishes to engage in advance directive discussions and documentation does not lead to harm,” the study concludes.
Lead author Stacy Fischer, MD, of the University of Colorado Denver says that it is striking how few hospitalized patients have actually engaged in these conversations, even though the population is quite ill. “So often, the conversation happens too late,” she says, “and then not with the patient but with a surrogate.”
Dr. Fischer encourages hospitalists to view the hospital admission as an important opportunity to start conversations with patients about their future care preferences. When patients come into the hospital, they must be asked about advance directives, but that process tends to be cursory, she says. At a minimum, hospitalists should clarify who the surrogate decision maker is, who would speak for the patient at a time of incapacity.
What should the hospitalist’s role be in end-of-life discussions? “That’s a complicated question in the current environment, where nobody seems to think it’s their role,” Dr. Fischer says. “I believe we all need to help move the conversation along. If [advance directive] forms can be available on the floor and if patients express interest in them, then encouraging them would be important.”
Larry Beresford is a freelance writer in Oakland, Calif.
References
Communication Vital to End-of-Life Care
A year ago in March, I looked my father in the eyes for the last time as he mouthed the words "help me" from his ICU bed. But despite being surrounded by teams of medical personnel and the latest healthcare technology, I felt utterly powerless to make a clear decision—and unclear to whom to turn for sound advice.
After 30 days of care in a well-known teaching hospital in the Northeast, my father was about to succumb to Stage 4 lung cancer, a tumor invading his spine. Moments before his plea, the ICU team had conducted a breathing test that apparently went awry—beginning the trial while my mother and I were downstairs receiving the latest round of conflicting information from a pair of doctors debating his outlook for discharge, physical rehabilitation, and hospice care. They casually informed us that a breathing test was about to occur; we rushed back to my father's side to learn the unfortunate outcome.
Prior to the episode that led to his being moved to the ICU, my father had been residing in a room directly across from a small hospitalist oncology office. What ensued was dizzying to behold: an endless parade of consultations; a narrowly averted million-dollar-plus spinal surgery in the wee hours; a too-zealous resident's further injuring of my father's right leg, which had already been compromised by a tumor degrading the femur.
My mother, my wife, and I struggled to maintain Dad's always-indomitable spirit while parsing the barrage of input regarding his potential for quality of life outside the hospital. We sat in numerous meetings, often with a pair of doctors espousing diametrically opposed outlooks. We tried to keep track of whom we were speaking with and who was in charge at any given moment; the lists we kept looked like the roster of a sports team, amply covered in scribbled-out names, phone numbers—and question marks.
It was only after my father tried feebly to speak his last words to me that the doctor who'd appeared to be most in charge pulled me aside at the door of the ICU. My mother and I hemmed and hawed in trying to decide whether to accede to another round of heroic measures. I was surprised by the somewhat terse tone of voice this senior physician used in dissuading us from allowing further life-extending efforts. I would have welcomed such honesty wholeheartedly far earlier in the process.
One of the value propositions hospitalists tout to their employers and patients is their expertise in coordinating care and facilitating communication among caregivers. Of course, there are nearly as many methods for doing so as there are hospitalist teams.
As the medical process grows more complex and specialized, with more "stakeholders" weighing in on the conversation, the hospitalist's role in taking charge of and energetically managing the flow of information for the benefit of beleaguered kin is more vital than ever. I can't speak for all loved ones who must witness the passage of a parent, a child, or a spouse, but for me, a hospitalist's firm hand would have made a world of difference in how we navigated this inevitable event.
Geoff Giordano was editor of The Hospitalist from 2007 to 2008. His father, Thomas, a lifelong journalist, wrote several articles for the magazine during that period.
A year ago in March, I looked my father in the eyes for the last time as he mouthed the words "help me" from his ICU bed. But despite being surrounded by teams of medical personnel and the latest healthcare technology, I felt utterly powerless to make a clear decision—and unclear to whom to turn for sound advice.
After 30 days of care in a well-known teaching hospital in the Northeast, my father was about to succumb to Stage 4 lung cancer, a tumor invading his spine. Moments before his plea, the ICU team had conducted a breathing test that apparently went awry—beginning the trial while my mother and I were downstairs receiving the latest round of conflicting information from a pair of doctors debating his outlook for discharge, physical rehabilitation, and hospice care. They casually informed us that a breathing test was about to occur; we rushed back to my father's side to learn the unfortunate outcome.
Prior to the episode that led to his being moved to the ICU, my father had been residing in a room directly across from a small hospitalist oncology office. What ensued was dizzying to behold: an endless parade of consultations; a narrowly averted million-dollar-plus spinal surgery in the wee hours; a too-zealous resident's further injuring of my father's right leg, which had already been compromised by a tumor degrading the femur.
My mother, my wife, and I struggled to maintain Dad's always-indomitable spirit while parsing the barrage of input regarding his potential for quality of life outside the hospital. We sat in numerous meetings, often with a pair of doctors espousing diametrically opposed outlooks. We tried to keep track of whom we were speaking with and who was in charge at any given moment; the lists we kept looked like the roster of a sports team, amply covered in scribbled-out names, phone numbers—and question marks.
It was only after my father tried feebly to speak his last words to me that the doctor who'd appeared to be most in charge pulled me aside at the door of the ICU. My mother and I hemmed and hawed in trying to decide whether to accede to another round of heroic measures. I was surprised by the somewhat terse tone of voice this senior physician used in dissuading us from allowing further life-extending efforts. I would have welcomed such honesty wholeheartedly far earlier in the process.
One of the value propositions hospitalists tout to their employers and patients is their expertise in coordinating care and facilitating communication among caregivers. Of course, there are nearly as many methods for doing so as there are hospitalist teams.
As the medical process grows more complex and specialized, with more "stakeholders" weighing in on the conversation, the hospitalist's role in taking charge of and energetically managing the flow of information for the benefit of beleaguered kin is more vital than ever. I can't speak for all loved ones who must witness the passage of a parent, a child, or a spouse, but for me, a hospitalist's firm hand would have made a world of difference in how we navigated this inevitable event.
Geoff Giordano was editor of The Hospitalist from 2007 to 2008. His father, Thomas, a lifelong journalist, wrote several articles for the magazine during that period.
A year ago in March, I looked my father in the eyes for the last time as he mouthed the words "help me" from his ICU bed. But despite being surrounded by teams of medical personnel and the latest healthcare technology, I felt utterly powerless to make a clear decision—and unclear to whom to turn for sound advice.
After 30 days of care in a well-known teaching hospital in the Northeast, my father was about to succumb to Stage 4 lung cancer, a tumor invading his spine. Moments before his plea, the ICU team had conducted a breathing test that apparently went awry—beginning the trial while my mother and I were downstairs receiving the latest round of conflicting information from a pair of doctors debating his outlook for discharge, physical rehabilitation, and hospice care. They casually informed us that a breathing test was about to occur; we rushed back to my father's side to learn the unfortunate outcome.
Prior to the episode that led to his being moved to the ICU, my father had been residing in a room directly across from a small hospitalist oncology office. What ensued was dizzying to behold: an endless parade of consultations; a narrowly averted million-dollar-plus spinal surgery in the wee hours; a too-zealous resident's further injuring of my father's right leg, which had already been compromised by a tumor degrading the femur.
My mother, my wife, and I struggled to maintain Dad's always-indomitable spirit while parsing the barrage of input regarding his potential for quality of life outside the hospital. We sat in numerous meetings, often with a pair of doctors espousing diametrically opposed outlooks. We tried to keep track of whom we were speaking with and who was in charge at any given moment; the lists we kept looked like the roster of a sports team, amply covered in scribbled-out names, phone numbers—and question marks.
It was only after my father tried feebly to speak his last words to me that the doctor who'd appeared to be most in charge pulled me aside at the door of the ICU. My mother and I hemmed and hawed in trying to decide whether to accede to another round of heroic measures. I was surprised by the somewhat terse tone of voice this senior physician used in dissuading us from allowing further life-extending efforts. I would have welcomed such honesty wholeheartedly far earlier in the process.
One of the value propositions hospitalists tout to their employers and patients is their expertise in coordinating care and facilitating communication among caregivers. Of course, there are nearly as many methods for doing so as there are hospitalist teams.
As the medical process grows more complex and specialized, with more "stakeholders" weighing in on the conversation, the hospitalist's role in taking charge of and energetically managing the flow of information for the benefit of beleaguered kin is more vital than ever. I can't speak for all loved ones who must witness the passage of a parent, a child, or a spouse, but for me, a hospitalist's firm hand would have made a world of difference in how we navigated this inevitable event.
Geoff Giordano was editor of The Hospitalist from 2007 to 2008. His father, Thomas, a lifelong journalist, wrote several articles for the magazine during that period.
Role of DNRs in Elderly Patients' Outcomes Analyzed
SAN FRANCISCO – Elderly patients with preexisting Do Not Resuscitate directives appear to be less likely to pursue rescue from complications following emergency surgery than similar patients without such orders, according to an analysis of data from the National Surgical Quality Improvement Program.
When patients with preoperative DNRs were propensity-matched with non-DNR patients, major complication rates were similar – 42% for the DNR group and 41% for the non-DNR group. However, 37% of DNR patients died, compared with 22% of non-DNR patients, Dr. John E. Scarborough reported at the annual meeting of the American Surgical Association. The investigators adjusted for baseline differences in level of illness to create a propensity-matched cohort of 1,053 patients in each group.
"While we called this outcome failure-to-rescue, we believe that term to be misleading. The term implies that rescue from complications is attempted but is unsuccessful. ... We had no reason to believe that the DNR patients in this well-matched cohort were any less capable of being rescued than non-DNR patients," Dr. Scarborough said. "Instead, we believe that the DNR patients in the matched cohort were less likely than non-DNR patients to pursue rescue from complications."
This conclusion is supported by the finding that DNR patients were significantly less likely to undergo reoperation within 30 days of the index procedure (odds ratio, 0.67).
The authors used participant files from the National Surgical Quality Improvement Program (NSQIP) for 2005-2010, involving medical records for 25,558 patients. These patients were at least 65 years old and underwent an emergency operation for one of 10 common surgical diagnoses. The primary predictor variable was preoperative DNR status, which was defined as "an order signed or cosigned by an attending physician in the 30 days prior to surgery ... regardless of whether the DNR order was subsequently rescinded immediately before the index operation." Other predictor variables included patient demographics, chronic comorbid disease burden, acute physical condition at presentation, and complexity of the emergency operation.
Outcome variables included the 30-day postoperative mortality rate and the 30-day major complication rate – organ/space surgical site infection, wound dehiscence, deep vein thrombosis, pulmonary embolism, pneumonia, reintubation, ventilator use longer than 48 hours, cardiac arrest, myocardial infarction, sepsis, shock, coma longer than 24 hours, prosthetic/graft failure, and bleeding. The failure-to-rescue rate was defined as the mortality rate among patients who had one or more major complications.
A total of 1,061 patients had DNR orders, and 24,497 patients did not. The overall 30-day mortality rate for patients with a DNR order was 37% (395/1,061). The overall 30-day morbidity for patients with a DNR was 42% (446/1,061).
Patients with DNR orders were older: 22% were at least 90 years of age, compared with 5% of the non-DNR patients. They were also sicker, with significantly greater rates of non–independent functional status, cognitive dysfunction, known malignancy, congestive heart failure, chronic obstructive pulmonary disease, ASA physical status class 4, preoperative hypoalbuminemia, and septic shock.
"Although the DNR patients were sicker, we did not find any overt evidence that they were treated less aggressively than non-DNR patients in the preoperative period," said Dr. Scarborough, of the department of surgery at Duke University in Durham, N.C. There was no significant difference between DNR and non-DNR patients in terms of preoperative mechanical ventilation (6% vs. 5%, respectively); and DNR patients were significantly more likely to receive a preoperative transfusion.
There was also no indication that DNR patients were treated less aggressively in the operating room. Operative time was significantly longer for DNR patients, and DNR patients underwent procedures at least as complex as, if not more than, procedures for non-DNR patients.
Invited discussant Dr. Ronnie A. Rosenthal asked how these data could be used to improve the way families are counseled before operations involving elderly patients with DNRs. Dr. Rosenthal is surgeon-in-chief at the VA Connecticut Healthcare System in New Haven.
"What we hope this study provides is a more reliable and sturdy resource for surgeons to counsel such patients than merely explaining to them what the average outcomes are," said Dr. Scarborough. He noted that the oncology literature suggests that patients who better understand their prognosis are in a better position to evaluate whether they want to pursue more aggressive treatment or treatments that have a lot of side effects.
Dr. Norman Estes, chair of the surgery department at the University of Illinois in Peoria, questioned how much of a role surgeons should play in advance planning. "I think that sometimes the advance directive creates a self-fulfilling prophecy for the patient."
Dr. Scarborough noted that advance directives need to be signed by the attending physician. "As to whether the surgeon should be more engaged in the conversation, I guess I would say that it depends on the surgeon. This is a very delicate conversation and obviously one that requires a fair amount of time," he said. Other physicians – such as geriatricians and palliative care physicians – are often more skilled at handling these conversations.
However, it is important for surgeons to have a greater understanding of the patient’s intent with regard to DNR directives, he concluded.
The authors reported that they had no financial disclosures.
SAN FRANCISCO – Elderly patients with preexisting Do Not Resuscitate directives appear to be less likely to pursue rescue from complications following emergency surgery than similar patients without such orders, according to an analysis of data from the National Surgical Quality Improvement Program.
When patients with preoperative DNRs were propensity-matched with non-DNR patients, major complication rates were similar – 42% for the DNR group and 41% for the non-DNR group. However, 37% of DNR patients died, compared with 22% of non-DNR patients, Dr. John E. Scarborough reported at the annual meeting of the American Surgical Association. The investigators adjusted for baseline differences in level of illness to create a propensity-matched cohort of 1,053 patients in each group.
"While we called this outcome failure-to-rescue, we believe that term to be misleading. The term implies that rescue from complications is attempted but is unsuccessful. ... We had no reason to believe that the DNR patients in this well-matched cohort were any less capable of being rescued than non-DNR patients," Dr. Scarborough said. "Instead, we believe that the DNR patients in the matched cohort were less likely than non-DNR patients to pursue rescue from complications."
This conclusion is supported by the finding that DNR patients were significantly less likely to undergo reoperation within 30 days of the index procedure (odds ratio, 0.67).
The authors used participant files from the National Surgical Quality Improvement Program (NSQIP) for 2005-2010, involving medical records for 25,558 patients. These patients were at least 65 years old and underwent an emergency operation for one of 10 common surgical diagnoses. The primary predictor variable was preoperative DNR status, which was defined as "an order signed or cosigned by an attending physician in the 30 days prior to surgery ... regardless of whether the DNR order was subsequently rescinded immediately before the index operation." Other predictor variables included patient demographics, chronic comorbid disease burden, acute physical condition at presentation, and complexity of the emergency operation.
Outcome variables included the 30-day postoperative mortality rate and the 30-day major complication rate – organ/space surgical site infection, wound dehiscence, deep vein thrombosis, pulmonary embolism, pneumonia, reintubation, ventilator use longer than 48 hours, cardiac arrest, myocardial infarction, sepsis, shock, coma longer than 24 hours, prosthetic/graft failure, and bleeding. The failure-to-rescue rate was defined as the mortality rate among patients who had one or more major complications.
A total of 1,061 patients had DNR orders, and 24,497 patients did not. The overall 30-day mortality rate for patients with a DNR order was 37% (395/1,061). The overall 30-day morbidity for patients with a DNR was 42% (446/1,061).
Patients with DNR orders were older: 22% were at least 90 years of age, compared with 5% of the non-DNR patients. They were also sicker, with significantly greater rates of non–independent functional status, cognitive dysfunction, known malignancy, congestive heart failure, chronic obstructive pulmonary disease, ASA physical status class 4, preoperative hypoalbuminemia, and septic shock.
"Although the DNR patients were sicker, we did not find any overt evidence that they were treated less aggressively than non-DNR patients in the preoperative period," said Dr. Scarborough, of the department of surgery at Duke University in Durham, N.C. There was no significant difference between DNR and non-DNR patients in terms of preoperative mechanical ventilation (6% vs. 5%, respectively); and DNR patients were significantly more likely to receive a preoperative transfusion.
There was also no indication that DNR patients were treated less aggressively in the operating room. Operative time was significantly longer for DNR patients, and DNR patients underwent procedures at least as complex as, if not more than, procedures for non-DNR patients.
Invited discussant Dr. Ronnie A. Rosenthal asked how these data could be used to improve the way families are counseled before operations involving elderly patients with DNRs. Dr. Rosenthal is surgeon-in-chief at the VA Connecticut Healthcare System in New Haven.
"What we hope this study provides is a more reliable and sturdy resource for surgeons to counsel such patients than merely explaining to them what the average outcomes are," said Dr. Scarborough. He noted that the oncology literature suggests that patients who better understand their prognosis are in a better position to evaluate whether they want to pursue more aggressive treatment or treatments that have a lot of side effects.
Dr. Norman Estes, chair of the surgery department at the University of Illinois in Peoria, questioned how much of a role surgeons should play in advance planning. "I think that sometimes the advance directive creates a self-fulfilling prophecy for the patient."
Dr. Scarborough noted that advance directives need to be signed by the attending physician. "As to whether the surgeon should be more engaged in the conversation, I guess I would say that it depends on the surgeon. This is a very delicate conversation and obviously one that requires a fair amount of time," he said. Other physicians – such as geriatricians and palliative care physicians – are often more skilled at handling these conversations.
However, it is important for surgeons to have a greater understanding of the patient’s intent with regard to DNR directives, he concluded.
The authors reported that they had no financial disclosures.
SAN FRANCISCO – Elderly patients with preexisting Do Not Resuscitate directives appear to be less likely to pursue rescue from complications following emergency surgery than similar patients without such orders, according to an analysis of data from the National Surgical Quality Improvement Program.
When patients with preoperative DNRs were propensity-matched with non-DNR patients, major complication rates were similar – 42% for the DNR group and 41% for the non-DNR group. However, 37% of DNR patients died, compared with 22% of non-DNR patients, Dr. John E. Scarborough reported at the annual meeting of the American Surgical Association. The investigators adjusted for baseline differences in level of illness to create a propensity-matched cohort of 1,053 patients in each group.
"While we called this outcome failure-to-rescue, we believe that term to be misleading. The term implies that rescue from complications is attempted but is unsuccessful. ... We had no reason to believe that the DNR patients in this well-matched cohort were any less capable of being rescued than non-DNR patients," Dr. Scarborough said. "Instead, we believe that the DNR patients in the matched cohort were less likely than non-DNR patients to pursue rescue from complications."
This conclusion is supported by the finding that DNR patients were significantly less likely to undergo reoperation within 30 days of the index procedure (odds ratio, 0.67).
The authors used participant files from the National Surgical Quality Improvement Program (NSQIP) for 2005-2010, involving medical records for 25,558 patients. These patients were at least 65 years old and underwent an emergency operation for one of 10 common surgical diagnoses. The primary predictor variable was preoperative DNR status, which was defined as "an order signed or cosigned by an attending physician in the 30 days prior to surgery ... regardless of whether the DNR order was subsequently rescinded immediately before the index operation." Other predictor variables included patient demographics, chronic comorbid disease burden, acute physical condition at presentation, and complexity of the emergency operation.
Outcome variables included the 30-day postoperative mortality rate and the 30-day major complication rate – organ/space surgical site infection, wound dehiscence, deep vein thrombosis, pulmonary embolism, pneumonia, reintubation, ventilator use longer than 48 hours, cardiac arrest, myocardial infarction, sepsis, shock, coma longer than 24 hours, prosthetic/graft failure, and bleeding. The failure-to-rescue rate was defined as the mortality rate among patients who had one or more major complications.
A total of 1,061 patients had DNR orders, and 24,497 patients did not. The overall 30-day mortality rate for patients with a DNR order was 37% (395/1,061). The overall 30-day morbidity for patients with a DNR was 42% (446/1,061).
Patients with DNR orders were older: 22% were at least 90 years of age, compared with 5% of the non-DNR patients. They were also sicker, with significantly greater rates of non–independent functional status, cognitive dysfunction, known malignancy, congestive heart failure, chronic obstructive pulmonary disease, ASA physical status class 4, preoperative hypoalbuminemia, and septic shock.
"Although the DNR patients were sicker, we did not find any overt evidence that they were treated less aggressively than non-DNR patients in the preoperative period," said Dr. Scarborough, of the department of surgery at Duke University in Durham, N.C. There was no significant difference between DNR and non-DNR patients in terms of preoperative mechanical ventilation (6% vs. 5%, respectively); and DNR patients were significantly more likely to receive a preoperative transfusion.
There was also no indication that DNR patients were treated less aggressively in the operating room. Operative time was significantly longer for DNR patients, and DNR patients underwent procedures at least as complex as, if not more than, procedures for non-DNR patients.
Invited discussant Dr. Ronnie A. Rosenthal asked how these data could be used to improve the way families are counseled before operations involving elderly patients with DNRs. Dr. Rosenthal is surgeon-in-chief at the VA Connecticut Healthcare System in New Haven.
"What we hope this study provides is a more reliable and sturdy resource for surgeons to counsel such patients than merely explaining to them what the average outcomes are," said Dr. Scarborough. He noted that the oncology literature suggests that patients who better understand their prognosis are in a better position to evaluate whether they want to pursue more aggressive treatment or treatments that have a lot of side effects.
Dr. Norman Estes, chair of the surgery department at the University of Illinois in Peoria, questioned how much of a role surgeons should play in advance planning. "I think that sometimes the advance directive creates a self-fulfilling prophecy for the patient."
Dr. Scarborough noted that advance directives need to be signed by the attending physician. "As to whether the surgeon should be more engaged in the conversation, I guess I would say that it depends on the surgeon. This is a very delicate conversation and obviously one that requires a fair amount of time," he said. Other physicians – such as geriatricians and palliative care physicians – are often more skilled at handling these conversations.
However, it is important for surgeons to have a greater understanding of the patient’s intent with regard to DNR directives, he concluded.
The authors reported that they had no financial disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN SURGICAL ASSOCIATION
Psychiatric Comorbidity Should Not Forestall Cancer Treatment
MIAMI – Serious psychiatric comorbidities should not deprive cancer patients of life-salvaging or life-enriching therapies, according to two psychiatrists who have helped such patients to achieve successful outcomes with treatment for their physical and mental disorders.
Aggressive psychiatric treatment preserved patients’ eligibility for bone marrow transplantation (BMT) and allowed terminally ill patients to "participate in the important process of saying goodbye," the clinicians reported during separate presentations at the annual meeting of the American Psychosocial Oncology Society.
Bone Marrow Transplantation Feasible
Dr. Isabel Schuermeyer, director of psycho-oncology at the Cleveland Clinic, underscored her contention that even suicidal ideation should not, in most cases, be a contraindication for BMT. She described the case of a 55-year-old mantle cell lymphoma patient with a lengthy history of depression who developed myelodysplastic syndrome following an initial autologous BMT.
"I think there should be no psychiatric contraindication to BMT."
While under consideration for repeat BMT, he developed suicidal ideation, including formulating a plan to drive a car into a wall or poison himself with carbon monoxide. Intensive, collaborative management and a brief inpatient psychiatry admission combined to stabilize the patient on mirtazapine (Remeron) and clonazepam within 2 weeks, she reported. Although rapidly progressive medical complications precluded a second BMT, his psychiatric condition became stable.
A second psychiatric intervention involved a 57-year-old woman with multiple myeloma who drove 1,000 miles without stopping, just to make an appointment at the Cleveland Clinic. This followed her firing of four oncologists in 6 months in her city of origin. Relevant history included an early suicide attempt, alcohol abuse, and anxiety.
On examination, the patient exhibited racing thoughts, a diminished need for sleep, and excessive activity and distractibility. With close follow-up by the psychosocial oncology team and initiation of quetiapine (Seroquel) therapy, the patient’s hypomanic symptoms soon abated, and she was able to safely undergo a transplant.
"She actually did really well and had some insight," Dr. Schuermeyer said in an interview. "She was sleeping 8-9 hours a night."
The literature on psychiatric suitability for BMT is sparse, she noted. In 2006, Foster and colleagues reported (Bone Marrow Transplant. 2006:37:223-8) that nearly 90% of BMT professionals would not transplant a patient with suicidal ideation.
A small case series (Psychiatry Clin. Neurosci. 2003:57:311-5) reported that six of seven patients with psychiatric illnesses were able to undergo BMT with the exception of one patient with borderline personality disorder.
There is a widespread belief that patients with a psychiatric history may not be good candidates for BMT due to concerns about adherence, Dr. Schuermeyer noted. "Everyone wants great outcomes with BMT," she said in an interview.
However, her belief, grounded in experience, is that close collaboration and competent mental health care can overcome psychiatric obstacles in the vast majority of cases. "I think there should be no psychiatric contraindication to BMT. At the end of the day, this is a lifesaving treatment," she said.
Unlike in solid organ transplantation, years of compliance are not required. "Let’s just save peoples’ lives. Let’s be aggressive and work together as a team," she said.
Depression Eased by ECT
Dr. Carlos G. Fernandez-Robles of the psychiatry oncology service at the Massachusetts General Hospital Cancer Center in Boston advocated aggressive treatment of another sort. Using three clinical cases as examples, he outlined the effective use of electroconvulsive therapy in cancer patients with profound treatment-resistant depression that included catatonic symptoms.
Catatonia, he said, is an uncommon entity in general, and even more so in cancer patients; it is likely often unrecognized, however, and can be a manifestation of paraneoplastic encephalopathy, CNS chemotherapy toxicity, or psychiatric in origin.
In one such patient, a 40-year-woman with unresectable T4bN2cM0 maxillary adenoid cystic carcinoma treated with radiation and combination chemotherapy, was admitted to the hospital for failure to thrive, weight loss, and immobility. On examination, she demonstrated mutism, posturing, limb rigidity and mild waxy flexibility, gegenhalten hypertonia, and grasp reflex.
While lorazepam (Ativan) 2 mg IV temporarily resolved her symptoms, the team was unable to achieve any sustained response to psychopharmacologic treatment. The patient responded fully to six treatments with electroconvulsive therapy and was able to complete radiation and chemotherapy, Dr. Fernandez-Robles said.
A second patient with profound depression had complete response to six rounds of ECT after he failed to respond to a variety of medications following resection of right temporal lesions and an anterior temporal lobectomy for multicentric glioblastoma multiforme.
Very few studies have been published of ECT in cancer patients, in large measure because of a stigma not only among members of the general public, but in medical training as well, Dr. Fernandez-Robles said.
"Jack Nicholson did for shock therapy what 'Jaws' did for sharks," he wryly paraphrased British psychiatrist Susan Benbow as saying in an allusion to the depiction of ECT in the movie "One Flew Over the Cuckoo’s Nest."
In practice, ECT can reduce emotional suffering and physical pain and can "restore capacity for enjoyment of whatever life remains to be lived," said Dr. Fernandez-Robles.
For terminally ill patients with unremitting depression, it offers hope for engagement "in the important process of saying good-byes and wrapping things up."
Adverse effects can include nausea, headache, postictal delirium, cardiovascular complications, prolonged apnea, and well-known effects on anterograde and recent retrograde memory. Indeed, patients described by Dr. Fernandez-Robles did experience memory-related side effects.
Nonetheless, he said each of the patients had "clear-cut" indications for ECT and their quality of life improved dramatically as a result of the therapy.
"I am really interested in ... the impact of serious mental illness on the quality of care we give to our patients," he said following the meeting.
Neither presenter disclosed any relevant conflicts of interest.
MIAMI – Serious psychiatric comorbidities should not deprive cancer patients of life-salvaging or life-enriching therapies, according to two psychiatrists who have helped such patients to achieve successful outcomes with treatment for their physical and mental disorders.
Aggressive psychiatric treatment preserved patients’ eligibility for bone marrow transplantation (BMT) and allowed terminally ill patients to "participate in the important process of saying goodbye," the clinicians reported during separate presentations at the annual meeting of the American Psychosocial Oncology Society.
Bone Marrow Transplantation Feasible
Dr. Isabel Schuermeyer, director of psycho-oncology at the Cleveland Clinic, underscored her contention that even suicidal ideation should not, in most cases, be a contraindication for BMT. She described the case of a 55-year-old mantle cell lymphoma patient with a lengthy history of depression who developed myelodysplastic syndrome following an initial autologous BMT.
"I think there should be no psychiatric contraindication to BMT."
While under consideration for repeat BMT, he developed suicidal ideation, including formulating a plan to drive a car into a wall or poison himself with carbon monoxide. Intensive, collaborative management and a brief inpatient psychiatry admission combined to stabilize the patient on mirtazapine (Remeron) and clonazepam within 2 weeks, she reported. Although rapidly progressive medical complications precluded a second BMT, his psychiatric condition became stable.
A second psychiatric intervention involved a 57-year-old woman with multiple myeloma who drove 1,000 miles without stopping, just to make an appointment at the Cleveland Clinic. This followed her firing of four oncologists in 6 months in her city of origin. Relevant history included an early suicide attempt, alcohol abuse, and anxiety.
On examination, the patient exhibited racing thoughts, a diminished need for sleep, and excessive activity and distractibility. With close follow-up by the psychosocial oncology team and initiation of quetiapine (Seroquel) therapy, the patient’s hypomanic symptoms soon abated, and she was able to safely undergo a transplant.
"She actually did really well and had some insight," Dr. Schuermeyer said in an interview. "She was sleeping 8-9 hours a night."
The literature on psychiatric suitability for BMT is sparse, she noted. In 2006, Foster and colleagues reported (Bone Marrow Transplant. 2006:37:223-8) that nearly 90% of BMT professionals would not transplant a patient with suicidal ideation.
A small case series (Psychiatry Clin. Neurosci. 2003:57:311-5) reported that six of seven patients with psychiatric illnesses were able to undergo BMT with the exception of one patient with borderline personality disorder.
There is a widespread belief that patients with a psychiatric history may not be good candidates for BMT due to concerns about adherence, Dr. Schuermeyer noted. "Everyone wants great outcomes with BMT," she said in an interview.
However, her belief, grounded in experience, is that close collaboration and competent mental health care can overcome psychiatric obstacles in the vast majority of cases. "I think there should be no psychiatric contraindication to BMT. At the end of the day, this is a lifesaving treatment," she said.
Unlike in solid organ transplantation, years of compliance are not required. "Let’s just save peoples’ lives. Let’s be aggressive and work together as a team," she said.
Depression Eased by ECT
Dr. Carlos G. Fernandez-Robles of the psychiatry oncology service at the Massachusetts General Hospital Cancer Center in Boston advocated aggressive treatment of another sort. Using three clinical cases as examples, he outlined the effective use of electroconvulsive therapy in cancer patients with profound treatment-resistant depression that included catatonic symptoms.
Catatonia, he said, is an uncommon entity in general, and even more so in cancer patients; it is likely often unrecognized, however, and can be a manifestation of paraneoplastic encephalopathy, CNS chemotherapy toxicity, or psychiatric in origin.
In one such patient, a 40-year-woman with unresectable T4bN2cM0 maxillary adenoid cystic carcinoma treated with radiation and combination chemotherapy, was admitted to the hospital for failure to thrive, weight loss, and immobility. On examination, she demonstrated mutism, posturing, limb rigidity and mild waxy flexibility, gegenhalten hypertonia, and grasp reflex.
While lorazepam (Ativan) 2 mg IV temporarily resolved her symptoms, the team was unable to achieve any sustained response to psychopharmacologic treatment. The patient responded fully to six treatments with electroconvulsive therapy and was able to complete radiation and chemotherapy, Dr. Fernandez-Robles said.
A second patient with profound depression had complete response to six rounds of ECT after he failed to respond to a variety of medications following resection of right temporal lesions and an anterior temporal lobectomy for multicentric glioblastoma multiforme.
Very few studies have been published of ECT in cancer patients, in large measure because of a stigma not only among members of the general public, but in medical training as well, Dr. Fernandez-Robles said.
"Jack Nicholson did for shock therapy what 'Jaws' did for sharks," he wryly paraphrased British psychiatrist Susan Benbow as saying in an allusion to the depiction of ECT in the movie "One Flew Over the Cuckoo’s Nest."
In practice, ECT can reduce emotional suffering and physical pain and can "restore capacity for enjoyment of whatever life remains to be lived," said Dr. Fernandez-Robles.
For terminally ill patients with unremitting depression, it offers hope for engagement "in the important process of saying good-byes and wrapping things up."
Adverse effects can include nausea, headache, postictal delirium, cardiovascular complications, prolonged apnea, and well-known effects on anterograde and recent retrograde memory. Indeed, patients described by Dr. Fernandez-Robles did experience memory-related side effects.
Nonetheless, he said each of the patients had "clear-cut" indications for ECT and their quality of life improved dramatically as a result of the therapy.
"I am really interested in ... the impact of serious mental illness on the quality of care we give to our patients," he said following the meeting.
Neither presenter disclosed any relevant conflicts of interest.
MIAMI – Serious psychiatric comorbidities should not deprive cancer patients of life-salvaging or life-enriching therapies, according to two psychiatrists who have helped such patients to achieve successful outcomes with treatment for their physical and mental disorders.
Aggressive psychiatric treatment preserved patients’ eligibility for bone marrow transplantation (BMT) and allowed terminally ill patients to "participate in the important process of saying goodbye," the clinicians reported during separate presentations at the annual meeting of the American Psychosocial Oncology Society.
Bone Marrow Transplantation Feasible
Dr. Isabel Schuermeyer, director of psycho-oncology at the Cleveland Clinic, underscored her contention that even suicidal ideation should not, in most cases, be a contraindication for BMT. She described the case of a 55-year-old mantle cell lymphoma patient with a lengthy history of depression who developed myelodysplastic syndrome following an initial autologous BMT.
"I think there should be no psychiatric contraindication to BMT."
While under consideration for repeat BMT, he developed suicidal ideation, including formulating a plan to drive a car into a wall or poison himself with carbon monoxide. Intensive, collaborative management and a brief inpatient psychiatry admission combined to stabilize the patient on mirtazapine (Remeron) and clonazepam within 2 weeks, she reported. Although rapidly progressive medical complications precluded a second BMT, his psychiatric condition became stable.
A second psychiatric intervention involved a 57-year-old woman with multiple myeloma who drove 1,000 miles without stopping, just to make an appointment at the Cleveland Clinic. This followed her firing of four oncologists in 6 months in her city of origin. Relevant history included an early suicide attempt, alcohol abuse, and anxiety.
On examination, the patient exhibited racing thoughts, a diminished need for sleep, and excessive activity and distractibility. With close follow-up by the psychosocial oncology team and initiation of quetiapine (Seroquel) therapy, the patient’s hypomanic symptoms soon abated, and she was able to safely undergo a transplant.
"She actually did really well and had some insight," Dr. Schuermeyer said in an interview. "She was sleeping 8-9 hours a night."
The literature on psychiatric suitability for BMT is sparse, she noted. In 2006, Foster and colleagues reported (Bone Marrow Transplant. 2006:37:223-8) that nearly 90% of BMT professionals would not transplant a patient with suicidal ideation.
A small case series (Psychiatry Clin. Neurosci. 2003:57:311-5) reported that six of seven patients with psychiatric illnesses were able to undergo BMT with the exception of one patient with borderline personality disorder.
There is a widespread belief that patients with a psychiatric history may not be good candidates for BMT due to concerns about adherence, Dr. Schuermeyer noted. "Everyone wants great outcomes with BMT," she said in an interview.
However, her belief, grounded in experience, is that close collaboration and competent mental health care can overcome psychiatric obstacles in the vast majority of cases. "I think there should be no psychiatric contraindication to BMT. At the end of the day, this is a lifesaving treatment," she said.
Unlike in solid organ transplantation, years of compliance are not required. "Let’s just save peoples’ lives. Let’s be aggressive and work together as a team," she said.
Depression Eased by ECT
Dr. Carlos G. Fernandez-Robles of the psychiatry oncology service at the Massachusetts General Hospital Cancer Center in Boston advocated aggressive treatment of another sort. Using three clinical cases as examples, he outlined the effective use of electroconvulsive therapy in cancer patients with profound treatment-resistant depression that included catatonic symptoms.
Catatonia, he said, is an uncommon entity in general, and even more so in cancer patients; it is likely often unrecognized, however, and can be a manifestation of paraneoplastic encephalopathy, CNS chemotherapy toxicity, or psychiatric in origin.
In one such patient, a 40-year-woman with unresectable T4bN2cM0 maxillary adenoid cystic carcinoma treated with radiation and combination chemotherapy, was admitted to the hospital for failure to thrive, weight loss, and immobility. On examination, she demonstrated mutism, posturing, limb rigidity and mild waxy flexibility, gegenhalten hypertonia, and grasp reflex.
While lorazepam (Ativan) 2 mg IV temporarily resolved her symptoms, the team was unable to achieve any sustained response to psychopharmacologic treatment. The patient responded fully to six treatments with electroconvulsive therapy and was able to complete radiation and chemotherapy, Dr. Fernandez-Robles said.
A second patient with profound depression had complete response to six rounds of ECT after he failed to respond to a variety of medications following resection of right temporal lesions and an anterior temporal lobectomy for multicentric glioblastoma multiforme.
Very few studies have been published of ECT in cancer patients, in large measure because of a stigma not only among members of the general public, but in medical training as well, Dr. Fernandez-Robles said.
"Jack Nicholson did for shock therapy what 'Jaws' did for sharks," he wryly paraphrased British psychiatrist Susan Benbow as saying in an allusion to the depiction of ECT in the movie "One Flew Over the Cuckoo’s Nest."
In practice, ECT can reduce emotional suffering and physical pain and can "restore capacity for enjoyment of whatever life remains to be lived," said Dr. Fernandez-Robles.
For terminally ill patients with unremitting depression, it offers hope for engagement "in the important process of saying good-byes and wrapping things up."
Adverse effects can include nausea, headache, postictal delirium, cardiovascular complications, prolonged apnea, and well-known effects on anterograde and recent retrograde memory. Indeed, patients described by Dr. Fernandez-Robles did experience memory-related side effects.
Nonetheless, he said each of the patients had "clear-cut" indications for ECT and their quality of life improved dramatically as a result of the therapy.
"I am really interested in ... the impact of serious mental illness on the quality of care we give to our patients," he said following the meeting.
Neither presenter disclosed any relevant conflicts of interest.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN PSYCHOSOCIAL ONCOLOGY SOCIETY
Compounded Morphine Gel Relieves Wound Pain
DENVER – Morphine sulfate can be readily formulated into a gel for effective topical analgesia in patients with painful skin ulcers and other wounds.
All that’s necessary is for a compounding pharmacist to mix 10 mg of intravenous morphine sulfate in 8 g of IntraSite Gel, a hydrogel widely used in wound care. The morphine gel can be applied to the wound one to three times daily, according to Mary Lynn McPherson, professor and vice chair of the department of pharmacy practice and science at the University of Maryland, Baltimore.
There is very little systemic absorption of the morphine. External irritation that manifests as burning or itching is the only side effect. This therapy works despite the limited systemic absorption because there are both central and peripheral opioid receptors.
Dressing changes can be excruciating for hospice patients. Administering an oral dose of ketamine beforehand provides effective pain relief.
"We’ve been doing this frequently in our inpatient hospice unit for patients who have these horrific, horrific wounds. We’ll give them 10, 20, or even 30 mg of ketamine orally 15-20 minutes before wound care. It has made absolutely all the difference in the world. The results have been spectacular," she said at the meeting.
The oral dose consists of intravenous ketamine solution, the only form in which the drug is available. The taste is awful, so the medication should be mixed with cherry syrup, spearmint, orange juice, or another masking flavor.
Dr. McPherson reported having no financial conflicts.
DENVER – Morphine sulfate can be readily formulated into a gel for effective topical analgesia in patients with painful skin ulcers and other wounds.
All that’s necessary is for a compounding pharmacist to mix 10 mg of intravenous morphine sulfate in 8 g of IntraSite Gel, a hydrogel widely used in wound care. The morphine gel can be applied to the wound one to three times daily, according to Mary Lynn McPherson, professor and vice chair of the department of pharmacy practice and science at the University of Maryland, Baltimore.
There is very little systemic absorption of the morphine. External irritation that manifests as burning or itching is the only side effect. This therapy works despite the limited systemic absorption because there are both central and peripheral opioid receptors.
Dressing changes can be excruciating for hospice patients. Administering an oral dose of ketamine beforehand provides effective pain relief.
"We’ve been doing this frequently in our inpatient hospice unit for patients who have these horrific, horrific wounds. We’ll give them 10, 20, or even 30 mg of ketamine orally 15-20 minutes before wound care. It has made absolutely all the difference in the world. The results have been spectacular," she said at the meeting.
The oral dose consists of intravenous ketamine solution, the only form in which the drug is available. The taste is awful, so the medication should be mixed with cherry syrup, spearmint, orange juice, or another masking flavor.
Dr. McPherson reported having no financial conflicts.
DENVER – Morphine sulfate can be readily formulated into a gel for effective topical analgesia in patients with painful skin ulcers and other wounds.
All that’s necessary is for a compounding pharmacist to mix 10 mg of intravenous morphine sulfate in 8 g of IntraSite Gel, a hydrogel widely used in wound care. The morphine gel can be applied to the wound one to three times daily, according to Mary Lynn McPherson, professor and vice chair of the department of pharmacy practice and science at the University of Maryland, Baltimore.
There is very little systemic absorption of the morphine. External irritation that manifests as burning or itching is the only side effect. This therapy works despite the limited systemic absorption because there are both central and peripheral opioid receptors.
Dressing changes can be excruciating for hospice patients. Administering an oral dose of ketamine beforehand provides effective pain relief.
"We’ve been doing this frequently in our inpatient hospice unit for patients who have these horrific, horrific wounds. We’ll give them 10, 20, or even 30 mg of ketamine orally 15-20 minutes before wound care. It has made absolutely all the difference in the world. The results have been spectacular," she said at the meeting.
The oral dose consists of intravenous ketamine solution, the only form in which the drug is available. The taste is awful, so the medication should be mixed with cherry syrup, spearmint, orange juice, or another masking flavor.
Dr. McPherson reported having no financial conflicts.
EXPERT ANALYSIS FROM THE ANNUAL ASSEMBLY OF THE AMERICAN ACADEMY OF HOSPICE AND PALLIATIVE MEDICINE
Palliative Care Specialists Ponder Public Awareness Campaign
DENVER – More than three-quarters of the general public have no idea what palliative care is, according to a national survey. And that, as it turns out, is actually excellent for the field’s future growth prospects, according to one of the nation’s top palliative care specialists.
"This is good news for us. We can create the cognitive frame where there isn’t one already in place," said Dr. Diane E. Meier, director of the Center to Advance Palliative Care and professor of geriatrics and internal medicine at Mount Sinai School of Medicine, New York.
While the public is largely a blank slate with regard to palliative care, nonpalliative care physicians and other health care professionals tend to believe that palliative care is simply end of life care. Many don’t understand that palliative care is actually about relieving the pain, symptoms, and stress of serious illness in patients of any age and at any stage of disease, and that palliative care can be delivered alongside curative or life-prolonging therapies, Dr. Meier said at the annual meeting of the American Academy of Hospice and Palliative Medicine.
The consumer survey sponsored by the Center to Advance Palliative Care and the American Cancer Society involved 800 adults; 70% indicated they were "not at all knowledgeable" about palliative care, and another 8% had never heard of the term. Only 5% were categorized as "very knowledgeable" about palliative care.
Once they were informed about what palliative care truly is, however, survey participants of all political persuasions were very positive about it.
For example, once they were educated about palliative care, 95% of those surveyed said it’s important for patients with serious illnesses and their families to learn about palliative care. Most (92%) indicated they would likely consider it for themselves or a loved one, and an equal percentage said it’s important that palliative care services be available at all hospitals, Dr. Meier reported.
She and other leaders in the palliative care field are now seeking funding for an ambitious 5-year, multimillion-dollar social marketing campaign to increase public awareness regarding palliative care.
"We’ve recognized that we’re not going to see policy change without public support," Dr. Meier said.
Among the policy changes she and her colleagues seek is a big boost in the palliative care workforce, which at present is so small as to constitute a major barrier to access. While there is one oncologist for every 145 patients in the United States with a new cancer diagnosis, and one cardiologist for every 71 patients who have a myocardial infarction, there is just one palliative care specialist for every 1,300 people with a serious illness. And in nearly half of the states, no postgraduate training in palliative care is available.
Dr. Meier would like to see an increased number of physician and nurse practitioner fellowship programs established in palliative care. Another priority is to develop a midcareer board certification track in palliative care across all medical disciplines.
"We have a lot of people coming in from oncology, surgery, and other fields who are seeking work with meaning and purpose," she observed.
Starting in 2013, the specialty will require fellowship training for board certification in palliative care. "Grandfathering in" will no longer be possible.
Dr. Meier reported having no financial conflicts.
DENVER – More than three-quarters of the general public have no idea what palliative care is, according to a national survey. And that, as it turns out, is actually excellent for the field’s future growth prospects, according to one of the nation’s top palliative care specialists.
"This is good news for us. We can create the cognitive frame where there isn’t one already in place," said Dr. Diane E. Meier, director of the Center to Advance Palliative Care and professor of geriatrics and internal medicine at Mount Sinai School of Medicine, New York.
While the public is largely a blank slate with regard to palliative care, nonpalliative care physicians and other health care professionals tend to believe that palliative care is simply end of life care. Many don’t understand that palliative care is actually about relieving the pain, symptoms, and stress of serious illness in patients of any age and at any stage of disease, and that palliative care can be delivered alongside curative or life-prolonging therapies, Dr. Meier said at the annual meeting of the American Academy of Hospice and Palliative Medicine.
The consumer survey sponsored by the Center to Advance Palliative Care and the American Cancer Society involved 800 adults; 70% indicated they were "not at all knowledgeable" about palliative care, and another 8% had never heard of the term. Only 5% were categorized as "very knowledgeable" about palliative care.
Once they were informed about what palliative care truly is, however, survey participants of all political persuasions were very positive about it.
For example, once they were educated about palliative care, 95% of those surveyed said it’s important for patients with serious illnesses and their families to learn about palliative care. Most (92%) indicated they would likely consider it for themselves or a loved one, and an equal percentage said it’s important that palliative care services be available at all hospitals, Dr. Meier reported.
She and other leaders in the palliative care field are now seeking funding for an ambitious 5-year, multimillion-dollar social marketing campaign to increase public awareness regarding palliative care.
"We’ve recognized that we’re not going to see policy change without public support," Dr. Meier said.
Among the policy changes she and her colleagues seek is a big boost in the palliative care workforce, which at present is so small as to constitute a major barrier to access. While there is one oncologist for every 145 patients in the United States with a new cancer diagnosis, and one cardiologist for every 71 patients who have a myocardial infarction, there is just one palliative care specialist for every 1,300 people with a serious illness. And in nearly half of the states, no postgraduate training in palliative care is available.
Dr. Meier would like to see an increased number of physician and nurse practitioner fellowship programs established in palliative care. Another priority is to develop a midcareer board certification track in palliative care across all medical disciplines.
"We have a lot of people coming in from oncology, surgery, and other fields who are seeking work with meaning and purpose," she observed.
Starting in 2013, the specialty will require fellowship training for board certification in palliative care. "Grandfathering in" will no longer be possible.
Dr. Meier reported having no financial conflicts.
DENVER – More than three-quarters of the general public have no idea what palliative care is, according to a national survey. And that, as it turns out, is actually excellent for the field’s future growth prospects, according to one of the nation’s top palliative care specialists.
"This is good news for us. We can create the cognitive frame where there isn’t one already in place," said Dr. Diane E. Meier, director of the Center to Advance Palliative Care and professor of geriatrics and internal medicine at Mount Sinai School of Medicine, New York.
While the public is largely a blank slate with regard to palliative care, nonpalliative care physicians and other health care professionals tend to believe that palliative care is simply end of life care. Many don’t understand that palliative care is actually about relieving the pain, symptoms, and stress of serious illness in patients of any age and at any stage of disease, and that palliative care can be delivered alongside curative or life-prolonging therapies, Dr. Meier said at the annual meeting of the American Academy of Hospice and Palliative Medicine.
The consumer survey sponsored by the Center to Advance Palliative Care and the American Cancer Society involved 800 adults; 70% indicated they were "not at all knowledgeable" about palliative care, and another 8% had never heard of the term. Only 5% were categorized as "very knowledgeable" about palliative care.
Once they were informed about what palliative care truly is, however, survey participants of all political persuasions were very positive about it.
For example, once they were educated about palliative care, 95% of those surveyed said it’s important for patients with serious illnesses and their families to learn about palliative care. Most (92%) indicated they would likely consider it for themselves or a loved one, and an equal percentage said it’s important that palliative care services be available at all hospitals, Dr. Meier reported.
She and other leaders in the palliative care field are now seeking funding for an ambitious 5-year, multimillion-dollar social marketing campaign to increase public awareness regarding palliative care.
"We’ve recognized that we’re not going to see policy change without public support," Dr. Meier said.
Among the policy changes she and her colleagues seek is a big boost in the palliative care workforce, which at present is so small as to constitute a major barrier to access. While there is one oncologist for every 145 patients in the United States with a new cancer diagnosis, and one cardiologist for every 71 patients who have a myocardial infarction, there is just one palliative care specialist for every 1,300 people with a serious illness. And in nearly half of the states, no postgraduate training in palliative care is available.
Dr. Meier would like to see an increased number of physician and nurse practitioner fellowship programs established in palliative care. Another priority is to develop a midcareer board certification track in palliative care across all medical disciplines.
"We have a lot of people coming in from oncology, surgery, and other fields who are seeking work with meaning and purpose," she observed.
Starting in 2013, the specialty will require fellowship training for board certification in palliative care. "Grandfathering in" will no longer be possible.
Dr. Meier reported having no financial conflicts.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF HOSPICE AND PALLIATIVE MEDICINE