User login
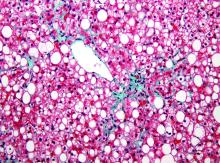
Long-term albumin shows survival benefit in decompensated cirrhosis
AMSTERDAM – Long-term treatment with human albumin improved the overall survival of patients with decompensated liver cirrhosis, compared with standard medical care, in a randomized, controlled trial presented at the International Liver Congress.
The final results of the ANSWER study showed that a 38% reduction in the risk of death could be achieved at 18 months’ follow-up by giving patients human albumin, with an overall survival of 78% vs. 66% in the two groups, respectively (hazard ratio, 0.62; 95% confidence interval, 0.40-0.95; P = .028).
“Long-term albumin administration to patients with decompensated cirrhosis may be seen as a disease-modifying treatment,” said the presenting study author, Mauro Bernardi, MD, professor in the department of medical and surgical sciences at the University of Bologna (Italy).
Not only was the overall survival improved, but there was improvement in the management of ascites, a reduction in the incidence of severe complications (such as spontaneous bacterial peritonitis, renal dysfunction, and hepatic encephalopathy), a reduction in the number of hospitalizations and duration of in-hospital treatment, and a signal for improved quality of life, he said.
“We know that it is good to give albumin as an infusion in many, many circumstances,” said Frank Tacke, MD, PhD, who was not involved in the study, during a press briefing at the meeting, sponsored by the European Association for the Study of the Liver (EASL).
“What we did not know before was if there was a role for giving albumin – which is unfortunately quite expensive – for a longer period of time in patients with liver cirrhosis,” said Dr. Tacke, who is the EASL vice-secretary and professor of medicine in the department of gastroenterology, metabolic diseases and intensive care medicine at University Hospital Aachen (Germany).
Although long-term treatment with human albumin is a relatively expensive treatment, particularly because it requires weekly infusion, Dr. Bernardi noted that a cost analysis was being performed, and potentially the cost of treatment could be offset by the reduction in paracentesis, duration of hospitalization, and reduced need for treating patients with complications, compared with standard medical care alone.
“The idea of supplying albumin to patients with advanced cirrhosis is quite an old one, and there is long debate. The point is that a reliable study that could resolve this was simply lacking up to now,” Dr. Bernard said at the briefing.
The results could mean that patients with decompensated cirrhosis now have a much-needed therapeutic option. These patients have “a very poor prognosis,” Dr. Bernardi said. The 1-year probability of survival is about 20%, and the only curative therapy at present is liver transplantation.
A total of 440 patients, mostly male with an average age of 61 years, with cirrhosis and uncomplicated ascites were randomized at 33 Italian centers to receive standard medical treatment alone (n = 213) or with human albumin (n = 218) given at an infused dose of 40 g twice a week for the first 2 weeks, then 40 g every week. The albumin used in the trial was provided by five different pharmaceutical companies and sent to a central location for generic relabeling and distribution out to the participating trial centers.
Significantly fewer patients who were given human albumin than those who were not (66% vs. 38%, P less than .001) needed at least one paracentesis. The incidence rate for the removal of peritoneal fluid in the standard medical treatment arm was 3.5/person per year. There was a 54% reduction in this rate by the addition of human albumin (HR = 0.46; 95% CI, 0.40-0.53; P less than .0001). There was also a significant 46% reduction in the incidence of refractory ascites (48% vs. 25%, P less than .0001).
Patients who received standard medical treatment plus albumin needed fewer hospitalizations and fewer days of in-hospital care per person per year than those in the standard care–only arm. The use of human albumin reduced the number of hospital stays by 35% (HR = 0.65; 95% CI, 0.55-0.77; P less than .0001) and the duration of days in hospital by 45% (HR = 0.55; 95% CI, 0.52-0.58; P less than .0001).
Although not statistically significant, a trend for greater improvements and fewer decreases in quality of life, as measured using the EQ-5D visual assessment scale, at 3, 6, and 12 months, was seen with the use of human albumin.
Four patients had adverse drug reactions: two were mild allergic reactions, and two were potentially life-threatening septic shock that needed intensive care treatment. One of the latter cases might have been caused by pneumonia, and the other required study interruption. But in all cases the patients recovered, Dr. Bernardi reported.
The study was funded by the Italian Drug Agency. Dr. Bernardi had acted as a speaker for and consultant to CLS Behring and Baxter Healthcare, and as a speaker to the Plasma Protein Therapeutics Association’s Europe division, Grifols, Gilead Sciences, and AbbVie Italia. Dr. Tacke had nothing to disclose.
AMSTERDAM – Long-term treatment with human albumin improved the overall survival of patients with decompensated liver cirrhosis, compared with standard medical care, in a randomized, controlled trial presented at the International Liver Congress.
The final results of the ANSWER study showed that a 38% reduction in the risk of death could be achieved at 18 months’ follow-up by giving patients human albumin, with an overall survival of 78% vs. 66% in the two groups, respectively (hazard ratio, 0.62; 95% confidence interval, 0.40-0.95; P = .028).
“Long-term albumin administration to patients with decompensated cirrhosis may be seen as a disease-modifying treatment,” said the presenting study author, Mauro Bernardi, MD, professor in the department of medical and surgical sciences at the University of Bologna (Italy).
Not only was the overall survival improved, but there was improvement in the management of ascites, a reduction in the incidence of severe complications (such as spontaneous bacterial peritonitis, renal dysfunction, and hepatic encephalopathy), a reduction in the number of hospitalizations and duration of in-hospital treatment, and a signal for improved quality of life, he said.
“We know that it is good to give albumin as an infusion in many, many circumstances,” said Frank Tacke, MD, PhD, who was not involved in the study, during a press briefing at the meeting, sponsored by the European Association for the Study of the Liver (EASL).
“What we did not know before was if there was a role for giving albumin – which is unfortunately quite expensive – for a longer period of time in patients with liver cirrhosis,” said Dr. Tacke, who is the EASL vice-secretary and professor of medicine in the department of gastroenterology, metabolic diseases and intensive care medicine at University Hospital Aachen (Germany).
Although long-term treatment with human albumin is a relatively expensive treatment, particularly because it requires weekly infusion, Dr. Bernardi noted that a cost analysis was being performed, and potentially the cost of treatment could be offset by the reduction in paracentesis, duration of hospitalization, and reduced need for treating patients with complications, compared with standard medical care alone.
“The idea of supplying albumin to patients with advanced cirrhosis is quite an old one, and there is long debate. The point is that a reliable study that could resolve this was simply lacking up to now,” Dr. Bernard said at the briefing.
The results could mean that patients with decompensated cirrhosis now have a much-needed therapeutic option. These patients have “a very poor prognosis,” Dr. Bernardi said. The 1-year probability of survival is about 20%, and the only curative therapy at present is liver transplantation.
A total of 440 patients, mostly male with an average age of 61 years, with cirrhosis and uncomplicated ascites were randomized at 33 Italian centers to receive standard medical treatment alone (n = 213) or with human albumin (n = 218) given at an infused dose of 40 g twice a week for the first 2 weeks, then 40 g every week. The albumin used in the trial was provided by five different pharmaceutical companies and sent to a central location for generic relabeling and distribution out to the participating trial centers.
Significantly fewer patients who were given human albumin than those who were not (66% vs. 38%, P less than .001) needed at least one paracentesis. The incidence rate for the removal of peritoneal fluid in the standard medical treatment arm was 3.5/person per year. There was a 54% reduction in this rate by the addition of human albumin (HR = 0.46; 95% CI, 0.40-0.53; P less than .0001). There was also a significant 46% reduction in the incidence of refractory ascites (48% vs. 25%, P less than .0001).
Patients who received standard medical treatment plus albumin needed fewer hospitalizations and fewer days of in-hospital care per person per year than those in the standard care–only arm. The use of human albumin reduced the number of hospital stays by 35% (HR = 0.65; 95% CI, 0.55-0.77; P less than .0001) and the duration of days in hospital by 45% (HR = 0.55; 95% CI, 0.52-0.58; P less than .0001).
Although not statistically significant, a trend for greater improvements and fewer decreases in quality of life, as measured using the EQ-5D visual assessment scale, at 3, 6, and 12 months, was seen with the use of human albumin.
Four patients had adverse drug reactions: two were mild allergic reactions, and two were potentially life-threatening septic shock that needed intensive care treatment. One of the latter cases might have been caused by pneumonia, and the other required study interruption. But in all cases the patients recovered, Dr. Bernardi reported.
The study was funded by the Italian Drug Agency. Dr. Bernardi had acted as a speaker for and consultant to CLS Behring and Baxter Healthcare, and as a speaker to the Plasma Protein Therapeutics Association’s Europe division, Grifols, Gilead Sciences, and AbbVie Italia. Dr. Tacke had nothing to disclose.
AMSTERDAM – Long-term treatment with human albumin improved the overall survival of patients with decompensated liver cirrhosis, compared with standard medical care, in a randomized, controlled trial presented at the International Liver Congress.
The final results of the ANSWER study showed that a 38% reduction in the risk of death could be achieved at 18 months’ follow-up by giving patients human albumin, with an overall survival of 78% vs. 66% in the two groups, respectively (hazard ratio, 0.62; 95% confidence interval, 0.40-0.95; P = .028).
“Long-term albumin administration to patients with decompensated cirrhosis may be seen as a disease-modifying treatment,” said the presenting study author, Mauro Bernardi, MD, professor in the department of medical and surgical sciences at the University of Bologna (Italy).
Not only was the overall survival improved, but there was improvement in the management of ascites, a reduction in the incidence of severe complications (such as spontaneous bacterial peritonitis, renal dysfunction, and hepatic encephalopathy), a reduction in the number of hospitalizations and duration of in-hospital treatment, and a signal for improved quality of life, he said.
“We know that it is good to give albumin as an infusion in many, many circumstances,” said Frank Tacke, MD, PhD, who was not involved in the study, during a press briefing at the meeting, sponsored by the European Association for the Study of the Liver (EASL).
“What we did not know before was if there was a role for giving albumin – which is unfortunately quite expensive – for a longer period of time in patients with liver cirrhosis,” said Dr. Tacke, who is the EASL vice-secretary and professor of medicine in the department of gastroenterology, metabolic diseases and intensive care medicine at University Hospital Aachen (Germany).
Although long-term treatment with human albumin is a relatively expensive treatment, particularly because it requires weekly infusion, Dr. Bernardi noted that a cost analysis was being performed, and potentially the cost of treatment could be offset by the reduction in paracentesis, duration of hospitalization, and reduced need for treating patients with complications, compared with standard medical care alone.
“The idea of supplying albumin to patients with advanced cirrhosis is quite an old one, and there is long debate. The point is that a reliable study that could resolve this was simply lacking up to now,” Dr. Bernard said at the briefing.
The results could mean that patients with decompensated cirrhosis now have a much-needed therapeutic option. These patients have “a very poor prognosis,” Dr. Bernardi said. The 1-year probability of survival is about 20%, and the only curative therapy at present is liver transplantation.
A total of 440 patients, mostly male with an average age of 61 years, with cirrhosis and uncomplicated ascites were randomized at 33 Italian centers to receive standard medical treatment alone (n = 213) or with human albumin (n = 218) given at an infused dose of 40 g twice a week for the first 2 weeks, then 40 g every week. The albumin used in the trial was provided by five different pharmaceutical companies and sent to a central location for generic relabeling and distribution out to the participating trial centers.
Significantly fewer patients who were given human albumin than those who were not (66% vs. 38%, P less than .001) needed at least one paracentesis. The incidence rate for the removal of peritoneal fluid in the standard medical treatment arm was 3.5/person per year. There was a 54% reduction in this rate by the addition of human albumin (HR = 0.46; 95% CI, 0.40-0.53; P less than .0001). There was also a significant 46% reduction in the incidence of refractory ascites (48% vs. 25%, P less than .0001).
Patients who received standard medical treatment plus albumin needed fewer hospitalizations and fewer days of in-hospital care per person per year than those in the standard care–only arm. The use of human albumin reduced the number of hospital stays by 35% (HR = 0.65; 95% CI, 0.55-0.77; P less than .0001) and the duration of days in hospital by 45% (HR = 0.55; 95% CI, 0.52-0.58; P less than .0001).
Although not statistically significant, a trend for greater improvements and fewer decreases in quality of life, as measured using the EQ-5D visual assessment scale, at 3, 6, and 12 months, was seen with the use of human albumin.
Four patients had adverse drug reactions: two were mild allergic reactions, and two were potentially life-threatening septic shock that needed intensive care treatment. One of the latter cases might have been caused by pneumonia, and the other required study interruption. But in all cases the patients recovered, Dr. Bernardi reported.
The study was funded by the Italian Drug Agency. Dr. Bernardi had acted as a speaker for and consultant to CLS Behring and Baxter Healthcare, and as a speaker to the Plasma Protein Therapeutics Association’s Europe division, Grifols, Gilead Sciences, and AbbVie Italia. Dr. Tacke had nothing to disclose.
AT ILC 2017
Key clinical point: A weekly infusion of human albumin has a beneficial effect in patients with decompensated cirrhosis.
Major finding: Overall survival was 78% vs. 66% for standard medical care with albumin vs. no albumin (HR, 0.62; 95% CI, 0.40-0.95; P = .028).
Data source: The ANSWER study, a multicenter, open-label, randomized clinical trial of 440 patients with decompensated cirrhosis.
Disclosures: The study was funded by the Italian Drug Agency. Dr. Bernardi had acted as a speaker for and consultant to CLS Behring and Baxter Healthcare, and as a speaker to the Plasma Protein Therapeutics Association’s Europe division, Grifols, Gilead Sciences, and AbbVie Italia. Dr. Tacke had nothing to disclose.
WHO report sets baseline for viral hepatitis elimination
AMSTERDAM – An estimated 328 million people worldwide were living with chronic hepatitis B or C virus infection in 2015 according to a new report issued by the World Health Organization and launched at the International Liver Congress sponsored by the European Association for the Study of the Liver (EASL).
The WHO Global Hepatitis Report gives the worldwide prevalence of chronic hepatitis B (HBV) infection as 257 million and that of chronic hepatitis C (HCV) infection as 71 million at this time point, reported Yvan Hutin, MD, medical officer at the WHO Department of HIV and Global Hepatitis Programme (HIV/GHP) in Geneva.
Dr. Hutin explained that the report was needed as it sets the baseline or “year zero” for tracking the success of WHO’s new global health sector strategy on viral hepatitis, which aims to eliminate viral hepatitis as a public health threat, reduce the number of new HBV and HCV infections by 90%, and reduce viral hepatitis mortality by 65% by 2030.
The report was “a very important statement for all of us who work in this field,” said EASL Vice-Secretary Tom Hemming Karslen, MD, during a press briefing. “This is a wonderful initiative helping all the activities that are now already ongoing and need to be strengthened to move in a coordinated manner.”
The launch of the report at the International Liver Congress was “win-win situation”, Gottfried Hirnschall, MD, director of the WHO Department of HIV/GHP, said at the press briefing.
“We are in the era of elimination. It is not only the commitment of the WHO, it is the commitment of the 194 member states who have signed up for elimination,” he said.
“An important message is that people are still dying of hepatitis, the numbers are still going up,” Dr. Hirnschall said. There were an estimated 1.34 million viral hepatitis deaths worldwide in 2015, most (95%) were due to the development of cirrhosis or hepatocellular carcinoma, according to the new report. “We have a public health issue that obviously still needs to be addressed.”
Three decades ago, little could be done to prevent or treat infection with HBV or HCV, Dr. Hutin said during the opening general scientific session. A lot has changed since then, prevention of hepatitis B started to become a reality with the availability of a vaccine and understanding of the importance of improved blood safety and injection practices. Since 2010, there have also been improvements in the drugs available to treat, and potentially eliminate, HCV, notably direct-acting antiviral agents.
“To reach elimination, we modeled that we needed to reach sufficient service coverage for five core interventions,” Dr. Hutin said. Specifically:
- At least 90% of the world’s eligible population receives the three-dose hepatitis B vaccine
- 100% of blood donations are screened appropriately
- Proper injection technique is employed in 90% of cases
- Clean needles made available where they are needed
- 90% of people infected are diagnosed and 80% are treated.
Vaccination against HBV has been one success in the past 20 years, Ana Maria Henao Restrepo, MD, medical officer at the WHO Department of Immunization Vaccination and Biologicals, said at the press briefing.
Vaccination against HBV started in 1982, she said, “when the first safe and effective vaccine became available, and now four out of five children receive this life-saving vaccine. We are very pleased with this achievement but we know that there is still more work to do.”
The WHO report estimates that the global incidence of chronic HBV infection in children under 5 years of age was reduced from 4.7% in the pre-vaccination era to 1.3% in 2015 because of immunization.
But while uptake of the three-dose hepatitis B vaccine has increased, with 85% coverage of the worlds population in 2015, the number of children receiving this vaccine at birth is just 39% overall, with lower rates in the African region.
“Unsafe health care injections and injection drug use continue to transmit HCV, particularly in the eastern Mediterranean region and the European region,” Dr. Hutin said.
The WHO has already set up a campaign to improve blood and injection safety called “Get the Point,” but there is still a long way to go. The target is to provide 300 needle and syringe sets per person per year to people who inject drugs; the current rate is around 27 sets.
Of the 257 people infected with hepatitis B in 2015, only 9% were diagnosed and 1.7 million received treatment. As for hepatitis C, 20% of 71 million were diagnosed and 1.1 million received treatment.
“We need a public health approach that delivers all the basic services to all, including to specific groups that may differ from the general population in terms of incidence, prevalence, vulnerability, or needs,” said Dr. Hutin. This includes health care workers, intravenous drug users, prisoners, migrants, blood donors, men who have sex with men, sex workers, and indigenous populations.
“We have all the tools we need to eliminate hepatitis,” he said, adding that improved point of care tests, a functional cure for HBV, and a vaccine against HCV would accelerate the process.
“A year ago, elimination by 2030 looked very ambitious, but not that we’ve carefully looked at the baseline, it seems that we have a start. It’s going to be a lot of work but the train has left the station and we should get there,” Dr. Hutin concluded.
The U.S. Centers for Disease Control and Prevention provided funding for the production of the report. All speakers had no conflicts of interest.
AMSTERDAM – An estimated 328 million people worldwide were living with chronic hepatitis B or C virus infection in 2015 according to a new report issued by the World Health Organization and launched at the International Liver Congress sponsored by the European Association for the Study of the Liver (EASL).
The WHO Global Hepatitis Report gives the worldwide prevalence of chronic hepatitis B (HBV) infection as 257 million and that of chronic hepatitis C (HCV) infection as 71 million at this time point, reported Yvan Hutin, MD, medical officer at the WHO Department of HIV and Global Hepatitis Programme (HIV/GHP) in Geneva.
Dr. Hutin explained that the report was needed as it sets the baseline or “year zero” for tracking the success of WHO’s new global health sector strategy on viral hepatitis, which aims to eliminate viral hepatitis as a public health threat, reduce the number of new HBV and HCV infections by 90%, and reduce viral hepatitis mortality by 65% by 2030.
The report was “a very important statement for all of us who work in this field,” said EASL Vice-Secretary Tom Hemming Karslen, MD, during a press briefing. “This is a wonderful initiative helping all the activities that are now already ongoing and need to be strengthened to move in a coordinated manner.”
The launch of the report at the International Liver Congress was “win-win situation”, Gottfried Hirnschall, MD, director of the WHO Department of HIV/GHP, said at the press briefing.
“We are in the era of elimination. It is not only the commitment of the WHO, it is the commitment of the 194 member states who have signed up for elimination,” he said.
“An important message is that people are still dying of hepatitis, the numbers are still going up,” Dr. Hirnschall said. There were an estimated 1.34 million viral hepatitis deaths worldwide in 2015, most (95%) were due to the development of cirrhosis or hepatocellular carcinoma, according to the new report. “We have a public health issue that obviously still needs to be addressed.”
Three decades ago, little could be done to prevent or treat infection with HBV or HCV, Dr. Hutin said during the opening general scientific session. A lot has changed since then, prevention of hepatitis B started to become a reality with the availability of a vaccine and understanding of the importance of improved blood safety and injection practices. Since 2010, there have also been improvements in the drugs available to treat, and potentially eliminate, HCV, notably direct-acting antiviral agents.
“To reach elimination, we modeled that we needed to reach sufficient service coverage for five core interventions,” Dr. Hutin said. Specifically:
- At least 90% of the world’s eligible population receives the three-dose hepatitis B vaccine
- 100% of blood donations are screened appropriately
- Proper injection technique is employed in 90% of cases
- Clean needles made available where they are needed
- 90% of people infected are diagnosed and 80% are treated.
Vaccination against HBV has been one success in the past 20 years, Ana Maria Henao Restrepo, MD, medical officer at the WHO Department of Immunization Vaccination and Biologicals, said at the press briefing.
Vaccination against HBV started in 1982, she said, “when the first safe and effective vaccine became available, and now four out of five children receive this life-saving vaccine. We are very pleased with this achievement but we know that there is still more work to do.”
The WHO report estimates that the global incidence of chronic HBV infection in children under 5 years of age was reduced from 4.7% in the pre-vaccination era to 1.3% in 2015 because of immunization.
But while uptake of the three-dose hepatitis B vaccine has increased, with 85% coverage of the worlds population in 2015, the number of children receiving this vaccine at birth is just 39% overall, with lower rates in the African region.
“Unsafe health care injections and injection drug use continue to transmit HCV, particularly in the eastern Mediterranean region and the European region,” Dr. Hutin said.
The WHO has already set up a campaign to improve blood and injection safety called “Get the Point,” but there is still a long way to go. The target is to provide 300 needle and syringe sets per person per year to people who inject drugs; the current rate is around 27 sets.
Of the 257 people infected with hepatitis B in 2015, only 9% were diagnosed and 1.7 million received treatment. As for hepatitis C, 20% of 71 million were diagnosed and 1.1 million received treatment.
“We need a public health approach that delivers all the basic services to all, including to specific groups that may differ from the general population in terms of incidence, prevalence, vulnerability, or needs,” said Dr. Hutin. This includes health care workers, intravenous drug users, prisoners, migrants, blood donors, men who have sex with men, sex workers, and indigenous populations.
“We have all the tools we need to eliminate hepatitis,” he said, adding that improved point of care tests, a functional cure for HBV, and a vaccine against HCV would accelerate the process.
“A year ago, elimination by 2030 looked very ambitious, but not that we’ve carefully looked at the baseline, it seems that we have a start. It’s going to be a lot of work but the train has left the station and we should get there,” Dr. Hutin concluded.
The U.S. Centers for Disease Control and Prevention provided funding for the production of the report. All speakers had no conflicts of interest.
AMSTERDAM – An estimated 328 million people worldwide were living with chronic hepatitis B or C virus infection in 2015 according to a new report issued by the World Health Organization and launched at the International Liver Congress sponsored by the European Association for the Study of the Liver (EASL).
The WHO Global Hepatitis Report gives the worldwide prevalence of chronic hepatitis B (HBV) infection as 257 million and that of chronic hepatitis C (HCV) infection as 71 million at this time point, reported Yvan Hutin, MD, medical officer at the WHO Department of HIV and Global Hepatitis Programme (HIV/GHP) in Geneva.
Dr. Hutin explained that the report was needed as it sets the baseline or “year zero” for tracking the success of WHO’s new global health sector strategy on viral hepatitis, which aims to eliminate viral hepatitis as a public health threat, reduce the number of new HBV and HCV infections by 90%, and reduce viral hepatitis mortality by 65% by 2030.
The report was “a very important statement for all of us who work in this field,” said EASL Vice-Secretary Tom Hemming Karslen, MD, during a press briefing. “This is a wonderful initiative helping all the activities that are now already ongoing and need to be strengthened to move in a coordinated manner.”
The launch of the report at the International Liver Congress was “win-win situation”, Gottfried Hirnschall, MD, director of the WHO Department of HIV/GHP, said at the press briefing.
“We are in the era of elimination. It is not only the commitment of the WHO, it is the commitment of the 194 member states who have signed up for elimination,” he said.
“An important message is that people are still dying of hepatitis, the numbers are still going up,” Dr. Hirnschall said. There were an estimated 1.34 million viral hepatitis deaths worldwide in 2015, most (95%) were due to the development of cirrhosis or hepatocellular carcinoma, according to the new report. “We have a public health issue that obviously still needs to be addressed.”
Three decades ago, little could be done to prevent or treat infection with HBV or HCV, Dr. Hutin said during the opening general scientific session. A lot has changed since then, prevention of hepatitis B started to become a reality with the availability of a vaccine and understanding of the importance of improved blood safety and injection practices. Since 2010, there have also been improvements in the drugs available to treat, and potentially eliminate, HCV, notably direct-acting antiviral agents.
“To reach elimination, we modeled that we needed to reach sufficient service coverage for five core interventions,” Dr. Hutin said. Specifically:
- At least 90% of the world’s eligible population receives the three-dose hepatitis B vaccine
- 100% of blood donations are screened appropriately
- Proper injection technique is employed in 90% of cases
- Clean needles made available where they are needed
- 90% of people infected are diagnosed and 80% are treated.
Vaccination against HBV has been one success in the past 20 years, Ana Maria Henao Restrepo, MD, medical officer at the WHO Department of Immunization Vaccination and Biologicals, said at the press briefing.
Vaccination against HBV started in 1982, she said, “when the first safe and effective vaccine became available, and now four out of five children receive this life-saving vaccine. We are very pleased with this achievement but we know that there is still more work to do.”
The WHO report estimates that the global incidence of chronic HBV infection in children under 5 years of age was reduced from 4.7% in the pre-vaccination era to 1.3% in 2015 because of immunization.
But while uptake of the three-dose hepatitis B vaccine has increased, with 85% coverage of the worlds population in 2015, the number of children receiving this vaccine at birth is just 39% overall, with lower rates in the African region.
“Unsafe health care injections and injection drug use continue to transmit HCV, particularly in the eastern Mediterranean region and the European region,” Dr. Hutin said.
The WHO has already set up a campaign to improve blood and injection safety called “Get the Point,” but there is still a long way to go. The target is to provide 300 needle and syringe sets per person per year to people who inject drugs; the current rate is around 27 sets.
Of the 257 people infected with hepatitis B in 2015, only 9% were diagnosed and 1.7 million received treatment. As for hepatitis C, 20% of 71 million were diagnosed and 1.1 million received treatment.
“We need a public health approach that delivers all the basic services to all, including to specific groups that may differ from the general population in terms of incidence, prevalence, vulnerability, or needs,” said Dr. Hutin. This includes health care workers, intravenous drug users, prisoners, migrants, blood donors, men who have sex with men, sex workers, and indigenous populations.
“We have all the tools we need to eliminate hepatitis,” he said, adding that improved point of care tests, a functional cure for HBV, and a vaccine against HCV would accelerate the process.
“A year ago, elimination by 2030 looked very ambitious, but not that we’ve carefully looked at the baseline, it seems that we have a start. It’s going to be a lot of work but the train has left the station and we should get there,” Dr. Hutin concluded.
The U.S. Centers for Disease Control and Prevention provided funding for the production of the report. All speakers had no conflicts of interest.
AT ILC 2017
Letter from the Editor: Spring brings flowers and liver stories
Happy spring (finally, for many of us)! This month’s issue of GI & Hepatology News is “weighted” towards liver. The decrease in hepatitis C–related liver disease means that steatohepatitis will emerge as the most frequent cause of cirrhosis and transplantation. Finding medical therapies to slow obesity-related liver damage has proven challenging. Bariatric surgery may be the best option for patients, as pointed out by one of our lead stories. Another page one story lays out a roadmap to eliminate viral hepatitis in the United States, a situation unheard of until direct-acting antiviral agents were developed.
The AGA’s contribution to this month’s issue is excellent. First, there is the continuing controversy regarding maintenance of certification. AGA has worked hard to eliminate the 10-year high-impact closed book examination (now an anachronism). We will have the option of a 2-year exam (open book) and you will need to become familiar with testing proposals so we all can add voices of reason to the ABIM process.
Additionally, the AGA highlights the POWER guideline (weight management) and its obesity resources, DDSEP® 8 and a new clinical guideline concerning transient elastography.
We close this month’s issue with a discussion from Ray Cross and Sunanda Kane about telemedicine and its impact on gastroenterology. There are multiple examples of how telemedicine is changing our practices and the piece provides hope for increased efficiencies and leveraged resources.
I hope you enjoy this issue. I have avoided my usual hints about our chaotic politics and its impact on our practices. We all need some relief and should take time to note the spring flowers.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Happy spring (finally, for many of us)! This month’s issue of GI & Hepatology News is “weighted” towards liver. The decrease in hepatitis C–related liver disease means that steatohepatitis will emerge as the most frequent cause of cirrhosis and transplantation. Finding medical therapies to slow obesity-related liver damage has proven challenging. Bariatric surgery may be the best option for patients, as pointed out by one of our lead stories. Another page one story lays out a roadmap to eliminate viral hepatitis in the United States, a situation unheard of until direct-acting antiviral agents were developed.
The AGA’s contribution to this month’s issue is excellent. First, there is the continuing controversy regarding maintenance of certification. AGA has worked hard to eliminate the 10-year high-impact closed book examination (now an anachronism). We will have the option of a 2-year exam (open book) and you will need to become familiar with testing proposals so we all can add voices of reason to the ABIM process.
Additionally, the AGA highlights the POWER guideline (weight management) and its obesity resources, DDSEP® 8 and a new clinical guideline concerning transient elastography.
We close this month’s issue with a discussion from Ray Cross and Sunanda Kane about telemedicine and its impact on gastroenterology. There are multiple examples of how telemedicine is changing our practices and the piece provides hope for increased efficiencies and leveraged resources.
I hope you enjoy this issue. I have avoided my usual hints about our chaotic politics and its impact on our practices. We all need some relief and should take time to note the spring flowers.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Happy spring (finally, for many of us)! This month’s issue of GI & Hepatology News is “weighted” towards liver. The decrease in hepatitis C–related liver disease means that steatohepatitis will emerge as the most frequent cause of cirrhosis and transplantation. Finding medical therapies to slow obesity-related liver damage has proven challenging. Bariatric surgery may be the best option for patients, as pointed out by one of our lead stories. Another page one story lays out a roadmap to eliminate viral hepatitis in the United States, a situation unheard of until direct-acting antiviral agents were developed.
The AGA’s contribution to this month’s issue is excellent. First, there is the continuing controversy regarding maintenance of certification. AGA has worked hard to eliminate the 10-year high-impact closed book examination (now an anachronism). We will have the option of a 2-year exam (open book) and you will need to become familiar with testing proposals so we all can add voices of reason to the ABIM process.
Additionally, the AGA highlights the POWER guideline (weight management) and its obesity resources, DDSEP® 8 and a new clinical guideline concerning transient elastography.
We close this month’s issue with a discussion from Ray Cross and Sunanda Kane about telemedicine and its impact on gastroenterology. There are multiple examples of how telemedicine is changing our practices and the piece provides hope for increased efficiencies and leveraged resources.
I hope you enjoy this issue. I have avoided my usual hints about our chaotic politics and its impact on our practices. We all need some relief and should take time to note the spring flowers.
John I. Allen, MD, MBA, AGAF
Editor in Chief
FDA approves Sovaldi, Harvoni for HCV in ages 12-plus
The Food and Drug Administration has approved the use of Sovaldi (sofosbuvir) and Harvoni (ledipasvir and sofosbuvir) for the treatment of hepatitis C virus (HCV) in children aged 12 years and older.
The drugs – the first direct-acting, potentially curative antiviral treatments approved for children and adolescents with HCV – previously were approved for adults. The supplemental applications submitted by Gilead Sciences, which markets the drugs, were approved by the FDA on April 7 and expand the use of these drugs to pediatric patients aged 12 and up who weigh at least 77 pounds, and who have either mild or no cirrhosis; Sovaldi is indicated for those with HCV genotypes 2 or 3, and Harvoni is indicated for those with HCV genotypes 1, 4, 5, or 6.
“These approvals will help change the landscape for HCV treatment by addressing an unmet need in children and adolescents,” Edward Cox, MD, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research, said in a press statement.
Of note, hepatitis B virus (HBV) reactivation has been reported in adults with HCV/HBV coinfection who were treated with these drugs, but who were not receiving HBV antiviral therapy; therefore, all patients should be screened for evidence of current or prior HBV infection before starting treatment with Harvoni or Sovaldi, according to the FDA statement.
The Food and Drug Administration has approved the use of Sovaldi (sofosbuvir) and Harvoni (ledipasvir and sofosbuvir) for the treatment of hepatitis C virus (HCV) in children aged 12 years and older.
The drugs – the first direct-acting, potentially curative antiviral treatments approved for children and adolescents with HCV – previously were approved for adults. The supplemental applications submitted by Gilead Sciences, which markets the drugs, were approved by the FDA on April 7 and expand the use of these drugs to pediatric patients aged 12 and up who weigh at least 77 pounds, and who have either mild or no cirrhosis; Sovaldi is indicated for those with HCV genotypes 2 or 3, and Harvoni is indicated for those with HCV genotypes 1, 4, 5, or 6.
“These approvals will help change the landscape for HCV treatment by addressing an unmet need in children and adolescents,” Edward Cox, MD, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research, said in a press statement.
Of note, hepatitis B virus (HBV) reactivation has been reported in adults with HCV/HBV coinfection who were treated with these drugs, but who were not receiving HBV antiviral therapy; therefore, all patients should be screened for evidence of current or prior HBV infection before starting treatment with Harvoni or Sovaldi, according to the FDA statement.
The Food and Drug Administration has approved the use of Sovaldi (sofosbuvir) and Harvoni (ledipasvir and sofosbuvir) for the treatment of hepatitis C virus (HCV) in children aged 12 years and older.
The drugs – the first direct-acting, potentially curative antiviral treatments approved for children and adolescents with HCV – previously were approved for adults. The supplemental applications submitted by Gilead Sciences, which markets the drugs, were approved by the FDA on April 7 and expand the use of these drugs to pediatric patients aged 12 and up who weigh at least 77 pounds, and who have either mild or no cirrhosis; Sovaldi is indicated for those with HCV genotypes 2 or 3, and Harvoni is indicated for those with HCV genotypes 1, 4, 5, or 6.
“These approvals will help change the landscape for HCV treatment by addressing an unmet need in children and adolescents,” Edward Cox, MD, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research, said in a press statement.
Of note, hepatitis B virus (HBV) reactivation has been reported in adults with HCV/HBV coinfection who were treated with these drugs, but who were not receiving HBV antiviral therapy; therefore, all patients should be screened for evidence of current or prior HBV infection before starting treatment with Harvoni or Sovaldi, according to the FDA statement.
Liver disease likely to become increasing indication for bariatric surgery
PHILADELPHIA – There is a long list of benefits from bariatric surgery in the morbidly obese, but prevention of end-stage liver disease and the need for a first or second liver transplant is likely to grow as an indication, according to an overview of weight loss surgery at Digestive Diseases: New Advances, held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education.
“Bariatric surgery is associated with significant improvement not just in diabetes, dyslipidemia, hypertension, and other complications of metabolic disorders but for me more interestingly, it is effective for treating fatty liver disease where you can see a 90% improvement in steatosis,” reported Subhashini Ayloo, MD, chief of minimally invasive robotic hepato-pancreato-biliary surgery and liver transplantation at New Jersey Medical School, Newark.
Trained in both bariatric surgery and liver transplant, Dr. Ayloo predicts that these fields will become increasingly connected because of the obesity epidemic and the related rise in nonalcoholic fatty liver disease (NAFLD). Dr. Ayloo reported that bariatric surgery is already being used in her center to avoid a second liver transplant in obese patients who are unable to lose sufficient weight to prevent progressive NAFLD after a first transplant.
The emphasis Dr. Ayloo placed on the role of bariatric surgery in preventing progression of NAFLD to nonalcoholic steatohepatitis and the inflammatory process that leads to fibrosis, cirrhosis, and liver decompensation, was drawn from her interest in these two fields. However, she did not ignore the potential of protection from obesity control for other diseases.
“Obesity adversely affects every organ in the body,” Dr. Ayloo pointed out. As a result of weight loss achieved with bariatric surgery, there is now a large body of evidence supporting broad benefits, not just those related to fat deposited in hepatocytes.
“We have a couple of decades of experience that has been published [with bariatric surgery], and this has shown that it maintains weight loss long term, it improves all the obesity-associated comorbidities, and it is cost effective,” Dr. Ayloo said. Now with long-term follow-up, “all of the studies are showing that bariatric surgery improves survival.”
Although most of the survival data have been generated by retrospective cohort studies, Dr. Ayloo cited nine sets of data showing odds ratios associating bariatric surgery with up to a 90% reduction in death over periods of up to 10 years of follow-up. In a summary slide presented by Dr. Ayloo, the estimated mortality benefit over 5 years was listed as 85%. The same summary slide listed large improvements in relevant measures of morbidity for more than 10 organ systems, such as improvement or resolution of dyslipidemia and hypertension in the circulatory system, improvement or resolution of asthma and other diseases affecting the respiratory system, and resolution or improvement of gastroesophageal reflux disease and other diseases affecting the gastrointestinal system.
Specific to the liver, these benefits included a nearly 40% reduction in liver inflammation and 20% reduction in fibrosis. According to Dr. Ayloo, who noted that NAFLD is expected to overtake hepatitis C virus as the No. 1 cause of liver transplant within the next 5 years, these data are important for drawing attention to bariatric surgery as a strategy to control liver disease. She suggested that there is a need to create a tighter link between efforts to treat morbid obesity and advanced liver disease.
“There is an established literature showing that if somebody is morbidly obese, the rate of liver transplant is lower than when compared to patients with normal weight,” Dr. Ayloo said. “There is a call out in the transplant community that we need to address this and we cannot just be throwing this under the table.”
Because of the strong relationship between obesity and NAFLD, a systematic approach is needed to consider liver disease in obese patients and obesity in patients with liver disease, she said. The close relationship is relevant when planning interventions for either. Liver disease should be assessed prior to bariatric surgery regardless of the indication and then monitored closely as part of postoperative care, she said.
Dr. Ayloo identified weight control as an essential part of posttransplant care to prevent hepatic fat deposition that threatens transplant-free survival.
Global Academy and this news organization are owned by the same company. Dr. Ayloo reports no relevant financial relationships.
AGA Resource
The AGA Obesity Practice Guide provides tools for gastroenterologists to lead a multidisciplinary team of health-care professionals for the management of patients with obesity. Learn more at www.gastro.org/obesity.
PHILADELPHIA – There is a long list of benefits from bariatric surgery in the morbidly obese, but prevention of end-stage liver disease and the need for a first or second liver transplant is likely to grow as an indication, according to an overview of weight loss surgery at Digestive Diseases: New Advances, held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education.
“Bariatric surgery is associated with significant improvement not just in diabetes, dyslipidemia, hypertension, and other complications of metabolic disorders but for me more interestingly, it is effective for treating fatty liver disease where you can see a 90% improvement in steatosis,” reported Subhashini Ayloo, MD, chief of minimally invasive robotic hepato-pancreato-biliary surgery and liver transplantation at New Jersey Medical School, Newark.
Trained in both bariatric surgery and liver transplant, Dr. Ayloo predicts that these fields will become increasingly connected because of the obesity epidemic and the related rise in nonalcoholic fatty liver disease (NAFLD). Dr. Ayloo reported that bariatric surgery is already being used in her center to avoid a second liver transplant in obese patients who are unable to lose sufficient weight to prevent progressive NAFLD after a first transplant.
The emphasis Dr. Ayloo placed on the role of bariatric surgery in preventing progression of NAFLD to nonalcoholic steatohepatitis and the inflammatory process that leads to fibrosis, cirrhosis, and liver decompensation, was drawn from her interest in these two fields. However, she did not ignore the potential of protection from obesity control for other diseases.
“Obesity adversely affects every organ in the body,” Dr. Ayloo pointed out. As a result of weight loss achieved with bariatric surgery, there is now a large body of evidence supporting broad benefits, not just those related to fat deposited in hepatocytes.
“We have a couple of decades of experience that has been published [with bariatric surgery], and this has shown that it maintains weight loss long term, it improves all the obesity-associated comorbidities, and it is cost effective,” Dr. Ayloo said. Now with long-term follow-up, “all of the studies are showing that bariatric surgery improves survival.”
Although most of the survival data have been generated by retrospective cohort studies, Dr. Ayloo cited nine sets of data showing odds ratios associating bariatric surgery with up to a 90% reduction in death over periods of up to 10 years of follow-up. In a summary slide presented by Dr. Ayloo, the estimated mortality benefit over 5 years was listed as 85%. The same summary slide listed large improvements in relevant measures of morbidity for more than 10 organ systems, such as improvement or resolution of dyslipidemia and hypertension in the circulatory system, improvement or resolution of asthma and other diseases affecting the respiratory system, and resolution or improvement of gastroesophageal reflux disease and other diseases affecting the gastrointestinal system.
Specific to the liver, these benefits included a nearly 40% reduction in liver inflammation and 20% reduction in fibrosis. According to Dr. Ayloo, who noted that NAFLD is expected to overtake hepatitis C virus as the No. 1 cause of liver transplant within the next 5 years, these data are important for drawing attention to bariatric surgery as a strategy to control liver disease. She suggested that there is a need to create a tighter link between efforts to treat morbid obesity and advanced liver disease.
“There is an established literature showing that if somebody is morbidly obese, the rate of liver transplant is lower than when compared to patients with normal weight,” Dr. Ayloo said. “There is a call out in the transplant community that we need to address this and we cannot just be throwing this under the table.”
Because of the strong relationship between obesity and NAFLD, a systematic approach is needed to consider liver disease in obese patients and obesity in patients with liver disease, she said. The close relationship is relevant when planning interventions for either. Liver disease should be assessed prior to bariatric surgery regardless of the indication and then monitored closely as part of postoperative care, she said.
Dr. Ayloo identified weight control as an essential part of posttransplant care to prevent hepatic fat deposition that threatens transplant-free survival.
Global Academy and this news organization are owned by the same company. Dr. Ayloo reports no relevant financial relationships.
AGA Resource
The AGA Obesity Practice Guide provides tools for gastroenterologists to lead a multidisciplinary team of health-care professionals for the management of patients with obesity. Learn more at www.gastro.org/obesity.
PHILADELPHIA – There is a long list of benefits from bariatric surgery in the morbidly obese, but prevention of end-stage liver disease and the need for a first or second liver transplant is likely to grow as an indication, according to an overview of weight loss surgery at Digestive Diseases: New Advances, held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education.
“Bariatric surgery is associated with significant improvement not just in diabetes, dyslipidemia, hypertension, and other complications of metabolic disorders but for me more interestingly, it is effective for treating fatty liver disease where you can see a 90% improvement in steatosis,” reported Subhashini Ayloo, MD, chief of minimally invasive robotic hepato-pancreato-biliary surgery and liver transplantation at New Jersey Medical School, Newark.
Trained in both bariatric surgery and liver transplant, Dr. Ayloo predicts that these fields will become increasingly connected because of the obesity epidemic and the related rise in nonalcoholic fatty liver disease (NAFLD). Dr. Ayloo reported that bariatric surgery is already being used in her center to avoid a second liver transplant in obese patients who are unable to lose sufficient weight to prevent progressive NAFLD after a first transplant.
The emphasis Dr. Ayloo placed on the role of bariatric surgery in preventing progression of NAFLD to nonalcoholic steatohepatitis and the inflammatory process that leads to fibrosis, cirrhosis, and liver decompensation, was drawn from her interest in these two fields. However, she did not ignore the potential of protection from obesity control for other diseases.
“Obesity adversely affects every organ in the body,” Dr. Ayloo pointed out. As a result of weight loss achieved with bariatric surgery, there is now a large body of evidence supporting broad benefits, not just those related to fat deposited in hepatocytes.
“We have a couple of decades of experience that has been published [with bariatric surgery], and this has shown that it maintains weight loss long term, it improves all the obesity-associated comorbidities, and it is cost effective,” Dr. Ayloo said. Now with long-term follow-up, “all of the studies are showing that bariatric surgery improves survival.”
Although most of the survival data have been generated by retrospective cohort studies, Dr. Ayloo cited nine sets of data showing odds ratios associating bariatric surgery with up to a 90% reduction in death over periods of up to 10 years of follow-up. In a summary slide presented by Dr. Ayloo, the estimated mortality benefit over 5 years was listed as 85%. The same summary slide listed large improvements in relevant measures of morbidity for more than 10 organ systems, such as improvement or resolution of dyslipidemia and hypertension in the circulatory system, improvement or resolution of asthma and other diseases affecting the respiratory system, and resolution or improvement of gastroesophageal reflux disease and other diseases affecting the gastrointestinal system.
Specific to the liver, these benefits included a nearly 40% reduction in liver inflammation and 20% reduction in fibrosis. According to Dr. Ayloo, who noted that NAFLD is expected to overtake hepatitis C virus as the No. 1 cause of liver transplant within the next 5 years, these data are important for drawing attention to bariatric surgery as a strategy to control liver disease. She suggested that there is a need to create a tighter link between efforts to treat morbid obesity and advanced liver disease.
“There is an established literature showing that if somebody is morbidly obese, the rate of liver transplant is lower than when compared to patients with normal weight,” Dr. Ayloo said. “There is a call out in the transplant community that we need to address this and we cannot just be throwing this under the table.”
Because of the strong relationship between obesity and NAFLD, a systematic approach is needed to consider liver disease in obese patients and obesity in patients with liver disease, she said. The close relationship is relevant when planning interventions for either. Liver disease should be assessed prior to bariatric surgery regardless of the indication and then monitored closely as part of postoperative care, she said.
Dr. Ayloo identified weight control as an essential part of posttransplant care to prevent hepatic fat deposition that threatens transplant-free survival.
Global Academy and this news organization are owned by the same company. Dr. Ayloo reports no relevant financial relationships.
AGA Resource
The AGA Obesity Practice Guide provides tools for gastroenterologists to lead a multidisciplinary team of health-care professionals for the management of patients with obesity. Learn more at www.gastro.org/obesity.
AT DIGESTIVE DISEASES: NEW ADVANCES
Eliminating hepatitis in the United States: A road map
An ambitious new report by the National Academies of Sciences, Engineering, and Medicine lays out a detailed path by which some 90,000 deaths from hepatitis B and C infection could be prevented by 2030.
The National Academies, a group of nongovernmental advisory bodies that includes the former Institute of Medicine, said that “the tools to prevent these deaths” exist – namely vaccination to prevent new hepatitis B infections and antiviral drugs, including new oral medications that can cure chronic hepatitis C infections within months.
The authors of the 200-plus-page report, led by Brian Strom, MD, MPH, of Rutgers University in Newark, NJ, calculate that deaths from hepatitis B infection could be halved by 2030 if 90% of patients are diagnosed, if 90% of those diagnosed are connected to care, and if 80% of those for whom treatment is indicated receive it. Treating everyone with chronic hepatitis C would reduce new infections by 90% by 2030, while reducing related deaths by 65%, Dr. Strom and his colleagues estimate.
But the authors also concede that drastic changes to current health policy would be required to reach these goals. These include the adoption of “aggressive testing, diagnosis, treatment, and prevention methods, such as needle exchange.”
They propose that the federal government seek a unique licensing arrangement with one or more manufacturers to bring down the notoriously high cost of direct-acting drugs used in hepatitis C, as a way of raising treatment rates. Currently, fewer than half the patients on Medicaid who are eligible for hepatitis C treatment receive it, and fewer than 1% of prisoners, who have high rates of infection.
Dr. Joseph Lim, director of the viral hepatitis program at Yale University in New Haven. Conn., who was not involved in the National Academies report, called it helpful in the sense that “it casts a spotlight on something that those of us involved in the care of people with viral hepatitis have long known – which is that this is a national and global public health burden that has been under the radar and in the shadow of other important health priorities.”
Both hepatitis B and C increase the risk of liver cancer and are associated with significant morbidity and mortality. Though approximately 4 million people in the United States are estimated to be infected with chronic hepatitis B (1.3 million) or C (2.7 million), these diseases account for less than 1% of the research budget at the National Institutes of Health, the report said. This compares unfavorably to funding for HIV, which affects about 1 million Americans.
As the report states, the tools to radically reduce hepatitis B and C deaths already exist. However, Dr. Lim cautioned in an interview, “the public health infrastructure to address viral hepatitis has been woefully inadequate.” In the United States, he noted, most states receive federal funding for at most a single person in charge of viral hepatitis epidemiology. “The resources currently available are in no way adequate to achieve the very aggressive goals described in the report,” he said.
Even among people with a known diagnosis of hepatitis B or C, only some receive confirmatory testing, Dr. Lim said. And of those with confirmed infections, “only a fraction are linked to care from the diagnosing clinician to a provider with the capacity to assess the state of liver disease and determine whether antiviral therapy is warranted.” Finally, he said, “many patients continue to face barriers to curative therapy due to cost and restrictions by public and private payers.”
Among the recommendations contained in the report is that unrestricted, mass treatment of hepatitis C infections be undertaken – regardless of disease stage. Currently, direct-acting antiviral agents remain costly and are poorly covered, notably by Medicaid. The National Academies advise that the government rectify this by purchasing “a license or assignment to the patent on a direct-acting antiviral drug, and use it only in those market segments where the government pays for treatment and access is now limited, such as Medicaid and prisons.”
Dr. Lim called the licensing proposal “very novel and bold,” but noted that there is no precedent in the United States for diseases such as hepatitis C. “If it could be done it would be an incredible model of government-pharma partnership for the public health good, and have a very significant impact.”
Steven Flamm, MD, chief of the liver transplantation program at Northwestern University in Chicago, who like Dr. Lim was not involved in the creation of the report, said in an interview that it contained innovative ideas and helped underscore the fact that “hepatitis has been given short shrift. The NIH and other agencies do not devote time and energy to this particular medical issue for reasons that are not completely clear.”
But “the problem with these kinds of analyses,” he said, “is that carrying them out is harder than making the recommendations.”
Dr. Flamm echoed Dr. Lim’s concerns about the practicability of implementing some of the recommendations in what he considers a resource-deprived health care environment for viral hepatitis.
“Is elimination possible or can you take a big bite out of it? The answer to that question is yes. We now have agents that can treat chronic viral hepatitis well, which we didn’t have a few years ago.”
Still, he emphasized, having the tools is only one part of the picture. Hepatitis C diagnostic tests have been available since the early 1990s. Yet, Dr. Flamm pointed out, fewer than half of patients have been diagnosed. “If the new CDC screening guidelines gain traction, we will do better than that.”
Dr. Flamm said that he considered the report’s call for a unique government licensing agreement for hepatitis C drugs a tall order. The drugs are already heavily discounted by manufacturers in many cases, he said, yet remain unavailable to those in need of them. In Illinois, Dr. Flamm said, few Medicaid patients with confirmed hepatitis C are given the short-acting antivirals that have revolutionized treatment. “The vast majority have no access to the therapy at all,” he said.
One of the report’s strengths, he said, is in detailing innovative prevention strategies such as delivering and promoting hepatitis B vaccinations to adults through local pharmacies, after the model of influenza vaccinations, and also conducting needle exchanges through pharmacies for intravenous drug users, who are at high risk of contracting both hepatitis B and C.
“Many of these strategies are not very costly,” he said. “The problem is you run into moral platitudes – to eliminate hepatitis, we will have to overcome that,” Dr. Flamm said, something that cannot be taken for granted in the current political environment.
But even if the goals outlined in the report seem ambitious, its authors have done an important service in underscoring the burden of viral hepatitis and laying out how some barriers to prevention, diagnosis, and treatment might be broken, he said.
Viral hepatitis “is a big deal, and it does cost a tremendous amount of money,” he added. “Everybody focuses on the therapeutic cost, but nobody focuses on the costs, direct and indirect, of all the sick people that are out there.”
An ambitious new report by the National Academies of Sciences, Engineering, and Medicine lays out a detailed path by which some 90,000 deaths from hepatitis B and C infection could be prevented by 2030.
The National Academies, a group of nongovernmental advisory bodies that includes the former Institute of Medicine, said that “the tools to prevent these deaths” exist – namely vaccination to prevent new hepatitis B infections and antiviral drugs, including new oral medications that can cure chronic hepatitis C infections within months.
The authors of the 200-plus-page report, led by Brian Strom, MD, MPH, of Rutgers University in Newark, NJ, calculate that deaths from hepatitis B infection could be halved by 2030 if 90% of patients are diagnosed, if 90% of those diagnosed are connected to care, and if 80% of those for whom treatment is indicated receive it. Treating everyone with chronic hepatitis C would reduce new infections by 90% by 2030, while reducing related deaths by 65%, Dr. Strom and his colleagues estimate.
But the authors also concede that drastic changes to current health policy would be required to reach these goals. These include the adoption of “aggressive testing, diagnosis, treatment, and prevention methods, such as needle exchange.”
They propose that the federal government seek a unique licensing arrangement with one or more manufacturers to bring down the notoriously high cost of direct-acting drugs used in hepatitis C, as a way of raising treatment rates. Currently, fewer than half the patients on Medicaid who are eligible for hepatitis C treatment receive it, and fewer than 1% of prisoners, who have high rates of infection.
Dr. Joseph Lim, director of the viral hepatitis program at Yale University in New Haven. Conn., who was not involved in the National Academies report, called it helpful in the sense that “it casts a spotlight on something that those of us involved in the care of people with viral hepatitis have long known – which is that this is a national and global public health burden that has been under the radar and in the shadow of other important health priorities.”
Both hepatitis B and C increase the risk of liver cancer and are associated with significant morbidity and mortality. Though approximately 4 million people in the United States are estimated to be infected with chronic hepatitis B (1.3 million) or C (2.7 million), these diseases account for less than 1% of the research budget at the National Institutes of Health, the report said. This compares unfavorably to funding for HIV, which affects about 1 million Americans.
As the report states, the tools to radically reduce hepatitis B and C deaths already exist. However, Dr. Lim cautioned in an interview, “the public health infrastructure to address viral hepatitis has been woefully inadequate.” In the United States, he noted, most states receive federal funding for at most a single person in charge of viral hepatitis epidemiology. “The resources currently available are in no way adequate to achieve the very aggressive goals described in the report,” he said.
Even among people with a known diagnosis of hepatitis B or C, only some receive confirmatory testing, Dr. Lim said. And of those with confirmed infections, “only a fraction are linked to care from the diagnosing clinician to a provider with the capacity to assess the state of liver disease and determine whether antiviral therapy is warranted.” Finally, he said, “many patients continue to face barriers to curative therapy due to cost and restrictions by public and private payers.”
Among the recommendations contained in the report is that unrestricted, mass treatment of hepatitis C infections be undertaken – regardless of disease stage. Currently, direct-acting antiviral agents remain costly and are poorly covered, notably by Medicaid. The National Academies advise that the government rectify this by purchasing “a license or assignment to the patent on a direct-acting antiviral drug, and use it only in those market segments where the government pays for treatment and access is now limited, such as Medicaid and prisons.”
Dr. Lim called the licensing proposal “very novel and bold,” but noted that there is no precedent in the United States for diseases such as hepatitis C. “If it could be done it would be an incredible model of government-pharma partnership for the public health good, and have a very significant impact.”
Steven Flamm, MD, chief of the liver transplantation program at Northwestern University in Chicago, who like Dr. Lim was not involved in the creation of the report, said in an interview that it contained innovative ideas and helped underscore the fact that “hepatitis has been given short shrift. The NIH and other agencies do not devote time and energy to this particular medical issue for reasons that are not completely clear.”
But “the problem with these kinds of analyses,” he said, “is that carrying them out is harder than making the recommendations.”
Dr. Flamm echoed Dr. Lim’s concerns about the practicability of implementing some of the recommendations in what he considers a resource-deprived health care environment for viral hepatitis.
“Is elimination possible or can you take a big bite out of it? The answer to that question is yes. We now have agents that can treat chronic viral hepatitis well, which we didn’t have a few years ago.”
Still, he emphasized, having the tools is only one part of the picture. Hepatitis C diagnostic tests have been available since the early 1990s. Yet, Dr. Flamm pointed out, fewer than half of patients have been diagnosed. “If the new CDC screening guidelines gain traction, we will do better than that.”
Dr. Flamm said that he considered the report’s call for a unique government licensing agreement for hepatitis C drugs a tall order. The drugs are already heavily discounted by manufacturers in many cases, he said, yet remain unavailable to those in need of them. In Illinois, Dr. Flamm said, few Medicaid patients with confirmed hepatitis C are given the short-acting antivirals that have revolutionized treatment. “The vast majority have no access to the therapy at all,” he said.
One of the report’s strengths, he said, is in detailing innovative prevention strategies such as delivering and promoting hepatitis B vaccinations to adults through local pharmacies, after the model of influenza vaccinations, and also conducting needle exchanges through pharmacies for intravenous drug users, who are at high risk of contracting both hepatitis B and C.
“Many of these strategies are not very costly,” he said. “The problem is you run into moral platitudes – to eliminate hepatitis, we will have to overcome that,” Dr. Flamm said, something that cannot be taken for granted in the current political environment.
But even if the goals outlined in the report seem ambitious, its authors have done an important service in underscoring the burden of viral hepatitis and laying out how some barriers to prevention, diagnosis, and treatment might be broken, he said.
Viral hepatitis “is a big deal, and it does cost a tremendous amount of money,” he added. “Everybody focuses on the therapeutic cost, but nobody focuses on the costs, direct and indirect, of all the sick people that are out there.”
An ambitious new report by the National Academies of Sciences, Engineering, and Medicine lays out a detailed path by which some 90,000 deaths from hepatitis B and C infection could be prevented by 2030.
The National Academies, a group of nongovernmental advisory bodies that includes the former Institute of Medicine, said that “the tools to prevent these deaths” exist – namely vaccination to prevent new hepatitis B infections and antiviral drugs, including new oral medications that can cure chronic hepatitis C infections within months.
The authors of the 200-plus-page report, led by Brian Strom, MD, MPH, of Rutgers University in Newark, NJ, calculate that deaths from hepatitis B infection could be halved by 2030 if 90% of patients are diagnosed, if 90% of those diagnosed are connected to care, and if 80% of those for whom treatment is indicated receive it. Treating everyone with chronic hepatitis C would reduce new infections by 90% by 2030, while reducing related deaths by 65%, Dr. Strom and his colleagues estimate.
But the authors also concede that drastic changes to current health policy would be required to reach these goals. These include the adoption of “aggressive testing, diagnosis, treatment, and prevention methods, such as needle exchange.”
They propose that the federal government seek a unique licensing arrangement with one or more manufacturers to bring down the notoriously high cost of direct-acting drugs used in hepatitis C, as a way of raising treatment rates. Currently, fewer than half the patients on Medicaid who are eligible for hepatitis C treatment receive it, and fewer than 1% of prisoners, who have high rates of infection.
Dr. Joseph Lim, director of the viral hepatitis program at Yale University in New Haven. Conn., who was not involved in the National Academies report, called it helpful in the sense that “it casts a spotlight on something that those of us involved in the care of people with viral hepatitis have long known – which is that this is a national and global public health burden that has been under the radar and in the shadow of other important health priorities.”
Both hepatitis B and C increase the risk of liver cancer and are associated with significant morbidity and mortality. Though approximately 4 million people in the United States are estimated to be infected with chronic hepatitis B (1.3 million) or C (2.7 million), these diseases account for less than 1% of the research budget at the National Institutes of Health, the report said. This compares unfavorably to funding for HIV, which affects about 1 million Americans.
As the report states, the tools to radically reduce hepatitis B and C deaths already exist. However, Dr. Lim cautioned in an interview, “the public health infrastructure to address viral hepatitis has been woefully inadequate.” In the United States, he noted, most states receive federal funding for at most a single person in charge of viral hepatitis epidemiology. “The resources currently available are in no way adequate to achieve the very aggressive goals described in the report,” he said.
Even among people with a known diagnosis of hepatitis B or C, only some receive confirmatory testing, Dr. Lim said. And of those with confirmed infections, “only a fraction are linked to care from the diagnosing clinician to a provider with the capacity to assess the state of liver disease and determine whether antiviral therapy is warranted.” Finally, he said, “many patients continue to face barriers to curative therapy due to cost and restrictions by public and private payers.”
Among the recommendations contained in the report is that unrestricted, mass treatment of hepatitis C infections be undertaken – regardless of disease stage. Currently, direct-acting antiviral agents remain costly and are poorly covered, notably by Medicaid. The National Academies advise that the government rectify this by purchasing “a license or assignment to the patent on a direct-acting antiviral drug, and use it only in those market segments where the government pays for treatment and access is now limited, such as Medicaid and prisons.”
Dr. Lim called the licensing proposal “very novel and bold,” but noted that there is no precedent in the United States for diseases such as hepatitis C. “If it could be done it would be an incredible model of government-pharma partnership for the public health good, and have a very significant impact.”
Steven Flamm, MD, chief of the liver transplantation program at Northwestern University in Chicago, who like Dr. Lim was not involved in the creation of the report, said in an interview that it contained innovative ideas and helped underscore the fact that “hepatitis has been given short shrift. The NIH and other agencies do not devote time and energy to this particular medical issue for reasons that are not completely clear.”
But “the problem with these kinds of analyses,” he said, “is that carrying them out is harder than making the recommendations.”
Dr. Flamm echoed Dr. Lim’s concerns about the practicability of implementing some of the recommendations in what he considers a resource-deprived health care environment for viral hepatitis.
“Is elimination possible or can you take a big bite out of it? The answer to that question is yes. We now have agents that can treat chronic viral hepatitis well, which we didn’t have a few years ago.”
Still, he emphasized, having the tools is only one part of the picture. Hepatitis C diagnostic tests have been available since the early 1990s. Yet, Dr. Flamm pointed out, fewer than half of patients have been diagnosed. “If the new CDC screening guidelines gain traction, we will do better than that.”
Dr. Flamm said that he considered the report’s call for a unique government licensing agreement for hepatitis C drugs a tall order. The drugs are already heavily discounted by manufacturers in many cases, he said, yet remain unavailable to those in need of them. In Illinois, Dr. Flamm said, few Medicaid patients with confirmed hepatitis C are given the short-acting antivirals that have revolutionized treatment. “The vast majority have no access to the therapy at all,” he said.
One of the report’s strengths, he said, is in detailing innovative prevention strategies such as delivering and promoting hepatitis B vaccinations to adults through local pharmacies, after the model of influenza vaccinations, and also conducting needle exchanges through pharmacies for intravenous drug users, who are at high risk of contracting both hepatitis B and C.
“Many of these strategies are not very costly,” he said. “The problem is you run into moral platitudes – to eliminate hepatitis, we will have to overcome that,” Dr. Flamm said, something that cannot be taken for granted in the current political environment.
But even if the goals outlined in the report seem ambitious, its authors have done an important service in underscoring the burden of viral hepatitis and laying out how some barriers to prevention, diagnosis, and treatment might be broken, he said.
Viral hepatitis “is a big deal, and it does cost a tremendous amount of money,” he added. “Everybody focuses on the therapeutic cost, but nobody focuses on the costs, direct and indirect, of all the sick people that are out there.”
FROM THE NATIONAL ACADEMIES OF SCIENCES, ENGINEERING, AND MEDICINE
Doubt expressed about potential of any single regimen to treat all hep C
PHILADELPHIA – Despite combination therapies for hepatitis C virus (HCV) that are now showing high rates of sustained viral remission (SVR) across all genotypes, a one-size-fits-all treatment will not be practical in the near future, according to a review of current and coming HCV therapies at Digestive Diseases: New Advances meeting held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
The focus for improving HCV care is “now shifting, I think, from this one-size-fits-all treatment to identifying, really, the one-size-fits all provider,” reported Kimberly A. Brown, MD, division head of gastroenterology/hepatology at Henry Ford Health System, Detroit.
The presence of cirrhosis in some patients demonstrates the need for this care, for example. Many of the most effective single-pill DAA combinations are demonstrating high SVR rates for HCV patients with cirrhosis, but Dr. Brown said that identification of cirrhosis prior to HCV treatment “remains imperative.” Some pangenotypic therapies require a longer duration of treatment when cirrhosis is present, and patients with cirrhosis require posttreatment monitoring for decompensation and hepatocellular carcinoma (HCC). Patients who have failed a prior anti-HCV regimen or who are in renal failure also require more individualized care.
“There is no current therapy in my opinion that allows for one combination, for one length of treatment, without consideration of any patient characteristics,” Dr. Brown said at the meeting. Although several newer combination drugs with pangenotypic properties are likely to be approved for HCV in 2017, Dr. Brown believes that the one-size-fits-all ideal is not going to be fulfilled “anytime soon.”
However, Dr. Brown does believe that HCV care can and should be shifted to trained primary care providers in order to increase the proportion of infected patients who are treated. She indicated that the pangenotypic drugs are making this easier to accomplish and cited a study from the most recent annual meeting of the American Society for the Study of Liver Diseases that showed comparable SVR rates for primary care physicians, nurse practitioners, and specialists when the primary care clinicians underwent a uniform 3-hour training program (Emmanuel B. et al. AASLD 2016;Abstract 22).
“There is an evidence basis for shifting care to primary care providers in order to expand treatment, but, certainly, these providers must have an interest,” Dr. Brown said. She also said that treatment from a primary care provider must be accompanied by follow-up care, which, for example, might include clinics specializing in alcohol or drug dependency.
In treatment-naive patients with uncomplicated HCV, nearly 100% of patients will achieve an SVR on 8-12 weeks of therapy, regardless of genotype, with the newest and most potent DAA regimens, according to data cited by Dr. Brown. However, she cautioned that, even in these patients, it would be inaccurate to conclude that one-size-fits-all therapy is sufficient.
One relatively recent concern is HBV activation. “The reactivation of HBV appears to be temporally related to use of DAAs, and this seems to be independent of HCV genotype, type of DAA received, or [the patient’s] HCV parameters,” Dr. Brown reported, citing data from the Food and Drug Administration. “The clinical implications for this are that HBV DNA must now be monitored, so this is another level of complexity for our care providers.”
Other considerations for care of HCV despite achieving SVR with current treatments include monitoring for HCC and preventing reinfection. Dr. Brown cautioned that the risk of HCC, although greatly reduced after SVR, is not eliminated, and specific monitoring strategies are particularly important for those with fibrosis or cirrhosis prior to SVR.
In addition, the same risks for primary HCV are relevant for reinfection, according to Dr. Brown. She pointed out that these reinfection rates can be substantial in populations that persist in behaviors that result in HCV exposure.
“We are getting very close to the ideal of a one-size-fits-all treatment regimen for HCV, which would include no need to check genotype, no contraindications, no need for close monitoring, and no need to document cirrhosis, but we are not there yet,” Dr. Brown said. Even if such a regimen does emerge, she indicated that clinicians are not likely to ever be absolved from important management decisions that ensure an optimal long-term outcome.
PHILADELPHIA – Despite combination therapies for hepatitis C virus (HCV) that are now showing high rates of sustained viral remission (SVR) across all genotypes, a one-size-fits-all treatment will not be practical in the near future, according to a review of current and coming HCV therapies at Digestive Diseases: New Advances meeting held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
The focus for improving HCV care is “now shifting, I think, from this one-size-fits-all treatment to identifying, really, the one-size-fits all provider,” reported Kimberly A. Brown, MD, division head of gastroenterology/hepatology at Henry Ford Health System, Detroit.
The presence of cirrhosis in some patients demonstrates the need for this care, for example. Many of the most effective single-pill DAA combinations are demonstrating high SVR rates for HCV patients with cirrhosis, but Dr. Brown said that identification of cirrhosis prior to HCV treatment “remains imperative.” Some pangenotypic therapies require a longer duration of treatment when cirrhosis is present, and patients with cirrhosis require posttreatment monitoring for decompensation and hepatocellular carcinoma (HCC). Patients who have failed a prior anti-HCV regimen or who are in renal failure also require more individualized care.
“There is no current therapy in my opinion that allows for one combination, for one length of treatment, without consideration of any patient characteristics,” Dr. Brown said at the meeting. Although several newer combination drugs with pangenotypic properties are likely to be approved for HCV in 2017, Dr. Brown believes that the one-size-fits-all ideal is not going to be fulfilled “anytime soon.”
However, Dr. Brown does believe that HCV care can and should be shifted to trained primary care providers in order to increase the proportion of infected patients who are treated. She indicated that the pangenotypic drugs are making this easier to accomplish and cited a study from the most recent annual meeting of the American Society for the Study of Liver Diseases that showed comparable SVR rates for primary care physicians, nurse practitioners, and specialists when the primary care clinicians underwent a uniform 3-hour training program (Emmanuel B. et al. AASLD 2016;Abstract 22).
“There is an evidence basis for shifting care to primary care providers in order to expand treatment, but, certainly, these providers must have an interest,” Dr. Brown said. She also said that treatment from a primary care provider must be accompanied by follow-up care, which, for example, might include clinics specializing in alcohol or drug dependency.
In treatment-naive patients with uncomplicated HCV, nearly 100% of patients will achieve an SVR on 8-12 weeks of therapy, regardless of genotype, with the newest and most potent DAA regimens, according to data cited by Dr. Brown. However, she cautioned that, even in these patients, it would be inaccurate to conclude that one-size-fits-all therapy is sufficient.
One relatively recent concern is HBV activation. “The reactivation of HBV appears to be temporally related to use of DAAs, and this seems to be independent of HCV genotype, type of DAA received, or [the patient’s] HCV parameters,” Dr. Brown reported, citing data from the Food and Drug Administration. “The clinical implications for this are that HBV DNA must now be monitored, so this is another level of complexity for our care providers.”
Other considerations for care of HCV despite achieving SVR with current treatments include monitoring for HCC and preventing reinfection. Dr. Brown cautioned that the risk of HCC, although greatly reduced after SVR, is not eliminated, and specific monitoring strategies are particularly important for those with fibrosis or cirrhosis prior to SVR.
In addition, the same risks for primary HCV are relevant for reinfection, according to Dr. Brown. She pointed out that these reinfection rates can be substantial in populations that persist in behaviors that result in HCV exposure.
“We are getting very close to the ideal of a one-size-fits-all treatment regimen for HCV, which would include no need to check genotype, no contraindications, no need for close monitoring, and no need to document cirrhosis, but we are not there yet,” Dr. Brown said. Even if such a regimen does emerge, she indicated that clinicians are not likely to ever be absolved from important management decisions that ensure an optimal long-term outcome.
PHILADELPHIA – Despite combination therapies for hepatitis C virus (HCV) that are now showing high rates of sustained viral remission (SVR) across all genotypes, a one-size-fits-all treatment will not be practical in the near future, according to a review of current and coming HCV therapies at Digestive Diseases: New Advances meeting held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education. Global Academy and this news organization are owned by the same company.
The focus for improving HCV care is “now shifting, I think, from this one-size-fits-all treatment to identifying, really, the one-size-fits all provider,” reported Kimberly A. Brown, MD, division head of gastroenterology/hepatology at Henry Ford Health System, Detroit.
The presence of cirrhosis in some patients demonstrates the need for this care, for example. Many of the most effective single-pill DAA combinations are demonstrating high SVR rates for HCV patients with cirrhosis, but Dr. Brown said that identification of cirrhosis prior to HCV treatment “remains imperative.” Some pangenotypic therapies require a longer duration of treatment when cirrhosis is present, and patients with cirrhosis require posttreatment monitoring for decompensation and hepatocellular carcinoma (HCC). Patients who have failed a prior anti-HCV regimen or who are in renal failure also require more individualized care.
“There is no current therapy in my opinion that allows for one combination, for one length of treatment, without consideration of any patient characteristics,” Dr. Brown said at the meeting. Although several newer combination drugs with pangenotypic properties are likely to be approved for HCV in 2017, Dr. Brown believes that the one-size-fits-all ideal is not going to be fulfilled “anytime soon.”
However, Dr. Brown does believe that HCV care can and should be shifted to trained primary care providers in order to increase the proportion of infected patients who are treated. She indicated that the pangenotypic drugs are making this easier to accomplish and cited a study from the most recent annual meeting of the American Society for the Study of Liver Diseases that showed comparable SVR rates for primary care physicians, nurse practitioners, and specialists when the primary care clinicians underwent a uniform 3-hour training program (Emmanuel B. et al. AASLD 2016;Abstract 22).
“There is an evidence basis for shifting care to primary care providers in order to expand treatment, but, certainly, these providers must have an interest,” Dr. Brown said. She also said that treatment from a primary care provider must be accompanied by follow-up care, which, for example, might include clinics specializing in alcohol or drug dependency.
In treatment-naive patients with uncomplicated HCV, nearly 100% of patients will achieve an SVR on 8-12 weeks of therapy, regardless of genotype, with the newest and most potent DAA regimens, according to data cited by Dr. Brown. However, she cautioned that, even in these patients, it would be inaccurate to conclude that one-size-fits-all therapy is sufficient.
One relatively recent concern is HBV activation. “The reactivation of HBV appears to be temporally related to use of DAAs, and this seems to be independent of HCV genotype, type of DAA received, or [the patient’s] HCV parameters,” Dr. Brown reported, citing data from the Food and Drug Administration. “The clinical implications for this are that HBV DNA must now be monitored, so this is another level of complexity for our care providers.”
Other considerations for care of HCV despite achieving SVR with current treatments include monitoring for HCC and preventing reinfection. Dr. Brown cautioned that the risk of HCC, although greatly reduced after SVR, is not eliminated, and specific monitoring strategies are particularly important for those with fibrosis or cirrhosis prior to SVR.
In addition, the same risks for primary HCV are relevant for reinfection, according to Dr. Brown. She pointed out that these reinfection rates can be substantial in populations that persist in behaviors that result in HCV exposure.
“We are getting very close to the ideal of a one-size-fits-all treatment regimen for HCV, which would include no need to check genotype, no contraindications, no need for close monitoring, and no need to document cirrhosis, but we are not there yet,” Dr. Brown said. Even if such a regimen does emerge, she indicated that clinicians are not likely to ever be absolved from important management decisions that ensure an optimal long-term outcome.
AT DIGESTIVE DISEASES: NEW ADVANCES MEETING
Key clinical point: Pangenotypic efficacy is not sufficient to create a one-size-fits-all regimen for HCV treatment, according to an assessment of evolving options.
Major finding: More-effective pills have moved the focus for progress in hepatitis C to more effective delivery of care.
Data source: A research review of current and coming HCV therapies.
Disclosures: Dr. Brown reported financial relationships with AbbVie, Bayer, Bristol-Myers Squibb, Gilead, Merck, and Novartis.
Of six milestones, only two left on route to hep B cure
PHILADELPHIA – The road to absolute cure of hepatitis B virus (HBV) is so well understood that the strategies now being actively pursued may conceivably eliminate this infection from the human population, according to a summary of progress presented at Digestive Diseases: New Advances.
“As in HIV and hepatitis C infections, combination therapies of drugs with different mechanisms of action will be the solution. Which combination will deliver the ultimate cure is yet to be determined, but within 5 years we should have a cure,” reported Vinod K. Rustgi, MD, chief of hepatology at Robert Wood Johnson Medical School, New Brunswick, N.J.
The biggest hurdle to cure may be clearing covalently closed circular DNA (cccDNA) from the hepatocytes of HBV-infected individuals. This structure is derived from cytoplasmic capsid–relaxed circular DNA (rcDNA) and creates a transcriptional template in the nucleus of hepatocytes. This HBV reservoir has been thus far resistant to therapy. According to Dr. Rustgi, eliminating cccDNA along with integrated HBV RNA sequences represents the final two milestones in the road to cure. There are at least theoretical approaches to eliminating both obstacles.
For many patients, available therapies already provide a functional cure of HBV, defined as viral loads below detection. Long-term data show that such patients can anticipate a lifespan equivalent to those without HBV infection, according to Dr. Rustgi, who listed this as one of the three milestones on the road to cure that have already been achieved. The first milestone was identified as a simple reduction in viral load. The second was anti-HBe seroconversion, which can be achieved in most with available treatments. Following functional cure, the fourth milestone, clearance of HBsAg antigen, is not yet reliably achieved, although this has been documented in a proportion of patients on the most effective long-term regimens.
Understanding these milestones is important, because each has prognostic relevance, according to Dr. Rustgi. Noting the direct correlation between increasing levels of serum HBV DNA and risk of hepatocellular carcinoma (HCC) over time, he indicated that reducing HBV DNA replication already reduces risk of complications. It is possible that virologic cure, which he defined as a stable off-drug suppression of HBV viremia and normalization of liver function tests, may be achieved in a larger proportion of patients with more potent versions of existing therapies. However, absolute cure, which includes complete clearance of cccDNA and other integrated HBV DNA, is the ultimate goal. It is expected that complete clearance will eliminate the threat of HCC or other liver complications.
Based on what is understood about HBV infection, increasing the potency of nucleoside analogues, which are the mainstay of current HBV therapy, will only go so far toward absolute cure. These drugs reduce viral replication to prevent liver damage. Although new drugs to prevent cccDNA formation and HBV life cycle events such as capsid assembly and HBeAg expression may further improve outcomes, they still may not be curative without better host defenses.
“One of the ways for trying to get rid of the virus is restoration of antiviral immunity, and there are several strategies to accomplish this,” reported Dr. Rustgi, citing active efforts to mobilize T cells that attack the virus and create toll-like receptor agonists, which play a key role in the innate immune response. He suggested that several strategies now demonstrating benefit in cancer, such as blocking proteins involved in the inhibition of the immune response, are being explored in the treatment of HBV. All of these strategies may be needed.
“No single approach will be sufficient to deliver a cure,” maintained Dr. Rustgi, outlining the many mechanisms HBV employs to defend against eradication. However, he provided a long list of HBV-targeted drugs and immunotherapies, including vaccines, which are now in clinical trials, enabling combinations to attack this infection from multiple fronts.
“A curative regimen might consist of an HBV antigen inhibitor, an immune activator, a cccDNA inhibitor, and a potent nucleoside analogue,” Dr. Rustgi said. Despite the multiple mechanisms that HBV now employs to evade eradication, there are now strategies being pursued to address most of the known vulnerabilities, he said. “I hope I have given you an idea where we are going.”
Digestive Diseases: New Advances was held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education. Global Academy and this news organization are owned by the same company. Dr. Rustgi reported financial relationships with AbbVie, Gilead, Innovimmune, Intercept, and Merck.
PHILADELPHIA – The road to absolute cure of hepatitis B virus (HBV) is so well understood that the strategies now being actively pursued may conceivably eliminate this infection from the human population, according to a summary of progress presented at Digestive Diseases: New Advances.
“As in HIV and hepatitis C infections, combination therapies of drugs with different mechanisms of action will be the solution. Which combination will deliver the ultimate cure is yet to be determined, but within 5 years we should have a cure,” reported Vinod K. Rustgi, MD, chief of hepatology at Robert Wood Johnson Medical School, New Brunswick, N.J.
The biggest hurdle to cure may be clearing covalently closed circular DNA (cccDNA) from the hepatocytes of HBV-infected individuals. This structure is derived from cytoplasmic capsid–relaxed circular DNA (rcDNA) and creates a transcriptional template in the nucleus of hepatocytes. This HBV reservoir has been thus far resistant to therapy. According to Dr. Rustgi, eliminating cccDNA along with integrated HBV RNA sequences represents the final two milestones in the road to cure. There are at least theoretical approaches to eliminating both obstacles.
For many patients, available therapies already provide a functional cure of HBV, defined as viral loads below detection. Long-term data show that such patients can anticipate a lifespan equivalent to those without HBV infection, according to Dr. Rustgi, who listed this as one of the three milestones on the road to cure that have already been achieved. The first milestone was identified as a simple reduction in viral load. The second was anti-HBe seroconversion, which can be achieved in most with available treatments. Following functional cure, the fourth milestone, clearance of HBsAg antigen, is not yet reliably achieved, although this has been documented in a proportion of patients on the most effective long-term regimens.
Understanding these milestones is important, because each has prognostic relevance, according to Dr. Rustgi. Noting the direct correlation between increasing levels of serum HBV DNA and risk of hepatocellular carcinoma (HCC) over time, he indicated that reducing HBV DNA replication already reduces risk of complications. It is possible that virologic cure, which he defined as a stable off-drug suppression of HBV viremia and normalization of liver function tests, may be achieved in a larger proportion of patients with more potent versions of existing therapies. However, absolute cure, which includes complete clearance of cccDNA and other integrated HBV DNA, is the ultimate goal. It is expected that complete clearance will eliminate the threat of HCC or other liver complications.
Based on what is understood about HBV infection, increasing the potency of nucleoside analogues, which are the mainstay of current HBV therapy, will only go so far toward absolute cure. These drugs reduce viral replication to prevent liver damage. Although new drugs to prevent cccDNA formation and HBV life cycle events such as capsid assembly and HBeAg expression may further improve outcomes, they still may not be curative without better host defenses.
“One of the ways for trying to get rid of the virus is restoration of antiviral immunity, and there are several strategies to accomplish this,” reported Dr. Rustgi, citing active efforts to mobilize T cells that attack the virus and create toll-like receptor agonists, which play a key role in the innate immune response. He suggested that several strategies now demonstrating benefit in cancer, such as blocking proteins involved in the inhibition of the immune response, are being explored in the treatment of HBV. All of these strategies may be needed.
“No single approach will be sufficient to deliver a cure,” maintained Dr. Rustgi, outlining the many mechanisms HBV employs to defend against eradication. However, he provided a long list of HBV-targeted drugs and immunotherapies, including vaccines, which are now in clinical trials, enabling combinations to attack this infection from multiple fronts.
“A curative regimen might consist of an HBV antigen inhibitor, an immune activator, a cccDNA inhibitor, and a potent nucleoside analogue,” Dr. Rustgi said. Despite the multiple mechanisms that HBV now employs to evade eradication, there are now strategies being pursued to address most of the known vulnerabilities, he said. “I hope I have given you an idea where we are going.”
Digestive Diseases: New Advances was held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education. Global Academy and this news organization are owned by the same company. Dr. Rustgi reported financial relationships with AbbVie, Gilead, Innovimmune, Intercept, and Merck.
PHILADELPHIA – The road to absolute cure of hepatitis B virus (HBV) is so well understood that the strategies now being actively pursued may conceivably eliminate this infection from the human population, according to a summary of progress presented at Digestive Diseases: New Advances.
“As in HIV and hepatitis C infections, combination therapies of drugs with different mechanisms of action will be the solution. Which combination will deliver the ultimate cure is yet to be determined, but within 5 years we should have a cure,” reported Vinod K. Rustgi, MD, chief of hepatology at Robert Wood Johnson Medical School, New Brunswick, N.J.
The biggest hurdle to cure may be clearing covalently closed circular DNA (cccDNA) from the hepatocytes of HBV-infected individuals. This structure is derived from cytoplasmic capsid–relaxed circular DNA (rcDNA) and creates a transcriptional template in the nucleus of hepatocytes. This HBV reservoir has been thus far resistant to therapy. According to Dr. Rustgi, eliminating cccDNA along with integrated HBV RNA sequences represents the final two milestones in the road to cure. There are at least theoretical approaches to eliminating both obstacles.
For many patients, available therapies already provide a functional cure of HBV, defined as viral loads below detection. Long-term data show that such patients can anticipate a lifespan equivalent to those without HBV infection, according to Dr. Rustgi, who listed this as one of the three milestones on the road to cure that have already been achieved. The first milestone was identified as a simple reduction in viral load. The second was anti-HBe seroconversion, which can be achieved in most with available treatments. Following functional cure, the fourth milestone, clearance of HBsAg antigen, is not yet reliably achieved, although this has been documented in a proportion of patients on the most effective long-term regimens.
Understanding these milestones is important, because each has prognostic relevance, according to Dr. Rustgi. Noting the direct correlation between increasing levels of serum HBV DNA and risk of hepatocellular carcinoma (HCC) over time, he indicated that reducing HBV DNA replication already reduces risk of complications. It is possible that virologic cure, which he defined as a stable off-drug suppression of HBV viremia and normalization of liver function tests, may be achieved in a larger proportion of patients with more potent versions of existing therapies. However, absolute cure, which includes complete clearance of cccDNA and other integrated HBV DNA, is the ultimate goal. It is expected that complete clearance will eliminate the threat of HCC or other liver complications.
Based on what is understood about HBV infection, increasing the potency of nucleoside analogues, which are the mainstay of current HBV therapy, will only go so far toward absolute cure. These drugs reduce viral replication to prevent liver damage. Although new drugs to prevent cccDNA formation and HBV life cycle events such as capsid assembly and HBeAg expression may further improve outcomes, they still may not be curative without better host defenses.
“One of the ways for trying to get rid of the virus is restoration of antiviral immunity, and there are several strategies to accomplish this,” reported Dr. Rustgi, citing active efforts to mobilize T cells that attack the virus and create toll-like receptor agonists, which play a key role in the innate immune response. He suggested that several strategies now demonstrating benefit in cancer, such as blocking proteins involved in the inhibition of the immune response, are being explored in the treatment of HBV. All of these strategies may be needed.
“No single approach will be sufficient to deliver a cure,” maintained Dr. Rustgi, outlining the many mechanisms HBV employs to defend against eradication. However, he provided a long list of HBV-targeted drugs and immunotherapies, including vaccines, which are now in clinical trials, enabling combinations to attack this infection from multiple fronts.
“A curative regimen might consist of an HBV antigen inhibitor, an immune activator, a cccDNA inhibitor, and a potent nucleoside analogue,” Dr. Rustgi said. Despite the multiple mechanisms that HBV now employs to evade eradication, there are now strategies being pursued to address most of the known vulnerabilities, he said. “I hope I have given you an idea where we are going.”
Digestive Diseases: New Advances was held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education. Global Academy and this news organization are owned by the same company. Dr. Rustgi reported financial relationships with AbbVie, Gilead, Innovimmune, Intercept, and Merck.
EXPERT ANALYSIS FROM DIGESTIVE DISEASES: NEW ADVANCES
Therapeutic window is narrow for steroids in alcoholic hepatitis
PHILADELPHIA – The main lesson for discordant data regarding the benefit of corticosteroids for alcoholic hepatitis is that mortality reductions accrue only to those patients who have advanced hepatitis but have not yet developed end-stage disease, according to a detailed look at published studies presented at Digestive Diseases: New Advances, held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education.
The relative benefit even in appropriate candidates is modest, according to Kevin Mullen, MD, chief of digestive diseases, West Virginia University, Morgantown. By his calculations, there is a survival benefit for one of every five to seven patients treated, and that survival benefit endures only 6-12 months.
These calculations were extrapolated from a long list of studies published over the last 45 years, some of the largest of which concluded that steroids are ineffective, according to Dr. Mullen. The likely source of the conflicting data is the timing of steroids over the course of the disease and the disparity in the scales used to define severity.
Of the scales employed to select candidates for steroid therapy, the Maddrey Discriminant Function (MDF) may be the best supported, according to Dr. Mullen. He suggested that other options, such as the MELD (Model for End-Stage Liver Disease) score and the presence or absence of hepatic encephalopathy are also likely to have discriminatory value in selecting patients for steroid therapy, but these have been largely evaluated in retrospective studies using disparate methodologies.
“The problem arises from so many trials using different criteria for patient selection,” Dr. Mullen explained.
Nevertheless, drawing on the preponderance of data, Dr. Mullen concluded that there is likely to be a therapeutic window within which steroids are beneficial. Using one prednisolone study that stratified patients by MDF score to illustrate this point, he noted that 6-month survival on active therapy was no better than placebo in patients with an MDF less than 25 and numerically but not necessarily clinically significantly better in those scoring 25-34. In the groups with an MDF score of 35-44 or 44-54, the survival at 6 months was several times higher (greater than 60% vs. less than 20%), but there was no advantage with scores greater than 54. In this latter group, the mortality rate at 6 months was 100% in those receiving steroids but only 80% among those given placebo.
“In my mind, there is no question that steroids can be of benefit, but it is a question of picking the right patient. If steroids are given too late in the disease process, it can exacerbate end-stage problems, leading to death,” Dr. Mullen said.
The potential mechanisms of benefit from steroids in alcoholic hepatitis include a reduction in collagen formation and an increase in albumin production, according to Dr. Mullen. In addition, steroids have the potential to suppress the cytokine-mediated inflammation that drives progressive liver dysfunction. However, steroids also have the potential of exacerbating existing infections by suppressing immune function. Moreover, he cautioned that steroids are contraindicated in patients with gastrointestinal bleeding or pancreatitis.
Importantly, patients with alcoholic hepatitis who are going to respond to steroids typically demonstrate a reduction in bilirubin within the first week, according to Dr. Mullen. He cautioned that continuing steroids in the absence of a change in bilirubin should be weighed again potential harms, including the exacerbation of liver disease or comorbidities. Even in responders, he recommended no more than 3 weeks to preserve a favorable benefit-to-risk ratio.
“Four weeks may be too long,” Dr. Mullen advised, but he also suggested that the management of advanced alcoholic hepatitis may be best left to specialists.
“Patients with severe alcoholic hepatitis should be referred and the referral should be to a hepatologist accustomed to managing these patients,” said Dr. Mullen, who cautioned that this is a challenging disease. “We have not been making a huge amount of progress” in the treatment of alcoholic hepatitis, which can be a frustrating disease because of alcoholic recidivism and poor prognosis in advanced stages, he said.
“I would argue that severe alcoholic liver disease has been one of the barriers for recruiting physicians into hepatology, because it is a very arduous group of people to look after, they get very sick, and the treatments are often not very successful,” he noted.
Global Academy and this news organization are owned by the same company. Dr. Mullen had no disclosures to report.
PHILADELPHIA – The main lesson for discordant data regarding the benefit of corticosteroids for alcoholic hepatitis is that mortality reductions accrue only to those patients who have advanced hepatitis but have not yet developed end-stage disease, according to a detailed look at published studies presented at Digestive Diseases: New Advances, held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education.
The relative benefit even in appropriate candidates is modest, according to Kevin Mullen, MD, chief of digestive diseases, West Virginia University, Morgantown. By his calculations, there is a survival benefit for one of every five to seven patients treated, and that survival benefit endures only 6-12 months.
These calculations were extrapolated from a long list of studies published over the last 45 years, some of the largest of which concluded that steroids are ineffective, according to Dr. Mullen. The likely source of the conflicting data is the timing of steroids over the course of the disease and the disparity in the scales used to define severity.
Of the scales employed to select candidates for steroid therapy, the Maddrey Discriminant Function (MDF) may be the best supported, according to Dr. Mullen. He suggested that other options, such as the MELD (Model for End-Stage Liver Disease) score and the presence or absence of hepatic encephalopathy are also likely to have discriminatory value in selecting patients for steroid therapy, but these have been largely evaluated in retrospective studies using disparate methodologies.
“The problem arises from so many trials using different criteria for patient selection,” Dr. Mullen explained.
Nevertheless, drawing on the preponderance of data, Dr. Mullen concluded that there is likely to be a therapeutic window within which steroids are beneficial. Using one prednisolone study that stratified patients by MDF score to illustrate this point, he noted that 6-month survival on active therapy was no better than placebo in patients with an MDF less than 25 and numerically but not necessarily clinically significantly better in those scoring 25-34. In the groups with an MDF score of 35-44 or 44-54, the survival at 6 months was several times higher (greater than 60% vs. less than 20%), but there was no advantage with scores greater than 54. In this latter group, the mortality rate at 6 months was 100% in those receiving steroids but only 80% among those given placebo.
“In my mind, there is no question that steroids can be of benefit, but it is a question of picking the right patient. If steroids are given too late in the disease process, it can exacerbate end-stage problems, leading to death,” Dr. Mullen said.
The potential mechanisms of benefit from steroids in alcoholic hepatitis include a reduction in collagen formation and an increase in albumin production, according to Dr. Mullen. In addition, steroids have the potential to suppress the cytokine-mediated inflammation that drives progressive liver dysfunction. However, steroids also have the potential of exacerbating existing infections by suppressing immune function. Moreover, he cautioned that steroids are contraindicated in patients with gastrointestinal bleeding or pancreatitis.
Importantly, patients with alcoholic hepatitis who are going to respond to steroids typically demonstrate a reduction in bilirubin within the first week, according to Dr. Mullen. He cautioned that continuing steroids in the absence of a change in bilirubin should be weighed again potential harms, including the exacerbation of liver disease or comorbidities. Even in responders, he recommended no more than 3 weeks to preserve a favorable benefit-to-risk ratio.
“Four weeks may be too long,” Dr. Mullen advised, but he also suggested that the management of advanced alcoholic hepatitis may be best left to specialists.
“Patients with severe alcoholic hepatitis should be referred and the referral should be to a hepatologist accustomed to managing these patients,” said Dr. Mullen, who cautioned that this is a challenging disease. “We have not been making a huge amount of progress” in the treatment of alcoholic hepatitis, which can be a frustrating disease because of alcoholic recidivism and poor prognosis in advanced stages, he said.
“I would argue that severe alcoholic liver disease has been one of the barriers for recruiting physicians into hepatology, because it is a very arduous group of people to look after, they get very sick, and the treatments are often not very successful,” he noted.
Global Academy and this news organization are owned by the same company. Dr. Mullen had no disclosures to report.
PHILADELPHIA – The main lesson for discordant data regarding the benefit of corticosteroids for alcoholic hepatitis is that mortality reductions accrue only to those patients who have advanced hepatitis but have not yet developed end-stage disease, according to a detailed look at published studies presented at Digestive Diseases: New Advances, held by Rutgers, the State University of New Jersey, and Global Academy for Medical Education.
The relative benefit even in appropriate candidates is modest, according to Kevin Mullen, MD, chief of digestive diseases, West Virginia University, Morgantown. By his calculations, there is a survival benefit for one of every five to seven patients treated, and that survival benefit endures only 6-12 months.
These calculations were extrapolated from a long list of studies published over the last 45 years, some of the largest of which concluded that steroids are ineffective, according to Dr. Mullen. The likely source of the conflicting data is the timing of steroids over the course of the disease and the disparity in the scales used to define severity.
Of the scales employed to select candidates for steroid therapy, the Maddrey Discriminant Function (MDF) may be the best supported, according to Dr. Mullen. He suggested that other options, such as the MELD (Model for End-Stage Liver Disease) score and the presence or absence of hepatic encephalopathy are also likely to have discriminatory value in selecting patients for steroid therapy, but these have been largely evaluated in retrospective studies using disparate methodologies.
“The problem arises from so many trials using different criteria for patient selection,” Dr. Mullen explained.
Nevertheless, drawing on the preponderance of data, Dr. Mullen concluded that there is likely to be a therapeutic window within which steroids are beneficial. Using one prednisolone study that stratified patients by MDF score to illustrate this point, he noted that 6-month survival on active therapy was no better than placebo in patients with an MDF less than 25 and numerically but not necessarily clinically significantly better in those scoring 25-34. In the groups with an MDF score of 35-44 or 44-54, the survival at 6 months was several times higher (greater than 60% vs. less than 20%), but there was no advantage with scores greater than 54. In this latter group, the mortality rate at 6 months was 100% in those receiving steroids but only 80% among those given placebo.
“In my mind, there is no question that steroids can be of benefit, but it is a question of picking the right patient. If steroids are given too late in the disease process, it can exacerbate end-stage problems, leading to death,” Dr. Mullen said.
The potential mechanisms of benefit from steroids in alcoholic hepatitis include a reduction in collagen formation and an increase in albumin production, according to Dr. Mullen. In addition, steroids have the potential to suppress the cytokine-mediated inflammation that drives progressive liver dysfunction. However, steroids also have the potential of exacerbating existing infections by suppressing immune function. Moreover, he cautioned that steroids are contraindicated in patients with gastrointestinal bleeding or pancreatitis.
Importantly, patients with alcoholic hepatitis who are going to respond to steroids typically demonstrate a reduction in bilirubin within the first week, according to Dr. Mullen. He cautioned that continuing steroids in the absence of a change in bilirubin should be weighed again potential harms, including the exacerbation of liver disease or comorbidities. Even in responders, he recommended no more than 3 weeks to preserve a favorable benefit-to-risk ratio.
“Four weeks may be too long,” Dr. Mullen advised, but he also suggested that the management of advanced alcoholic hepatitis may be best left to specialists.
“Patients with severe alcoholic hepatitis should be referred and the referral should be to a hepatologist accustomed to managing these patients,” said Dr. Mullen, who cautioned that this is a challenging disease. “We have not been making a huge amount of progress” in the treatment of alcoholic hepatitis, which can be a frustrating disease because of alcoholic recidivism and poor prognosis in advanced stages, he said.
“I would argue that severe alcoholic liver disease has been one of the barriers for recruiting physicians into hepatology, because it is a very arduous group of people to look after, they get very sick, and the treatments are often not very successful,” he noted.
Global Academy and this news organization are owned by the same company. Dr. Mullen had no disclosures to report.
HCV ‘cure’ within the VA appears likely
The number of Veterans Affairs patients with hepatitis C who have achieved a sustained virologic response to antiviral therapy has escalated so rapidly and reached such a height that the disease may well be eradicated in that health care system within a few years, according to a report in Alimentary Pharmacology and Therapeutics.
The potential public health benefits are substantial, “considering that HCV infection is the most common cause of cirrhosis and liver cancer in the VA and the United States, that the benefits of SVR are long-lasting, and that HCV clearance reduces the risk of liver cancer by 76% and all-cause mortality by 50%,” said Andrew M. Moon, MD, of the division of general internal medicine, University of Washington, Seattle, and his associates.
An estimated 124,662 VA patients currently are infected, and curing them “would substantially reduce the burden of HCV within the entire country and prevent tens of thousands of deaths,” they noted.
The VA dramatically increased the number of patients who were offered treatment in recent years, because it was able to allocate nearly $700 million to offset the high costs of highly effective direct antiviral agents, which in turn made these better-tolerated drugs more widely available at clinics across the country. The VA also removed all treatment prioritization criteria, allowing all patients, not just those with severe disease, to receive highly effective direct antiviral agents. This is “in stark contrast to most health care systems, state Medicaid programs, and insurance carriers in the U.S., which still restrict access ... based on severity of liver disease,” the investigators said (Aliment Pharmacol Ther. 2017 March 8. doi: 10.1111/apt.14021).
To examine the impact of these changes, Dr. Moon and his associates performed a retrospective cohort study, analyzing the electronic medical records of all 105,369 HCV treatment regimens given to 78,947 VA patients (mean age, 56 years) during a 17-year period. They found that annual treatment rates more than doubled from 2,726 to 6,679 patients when pegylated interferon was introduced, declined for a while and then rose modestly to 4,900 patients when boceprevir and telaprevir were introduced, declined again to an all-time low of 2,609 and then rebounded to 9,180 patients when sofosbuvir and simeprevir were introduced, and finally skyrocketed to 31,028 patients when ledipasvir/sofosbuvir and paritaprevir/ritonavir/ombitasvir/dasabuvir were introduced.
Correspondingly, SVR rates rose from less than 25% at the beginning of the study period to a “remarkable” 90.5% at the end. The improvement in SVR rates was even more pronounced among traditionally “hard to treat” cases, such as patients with concomitant cirrhosis (from 11.0% to 87.0%), decompensated cirrhosis (from 14.6% to 85.2%), highly refractory infection (from 16.4% to 89.3%), and genotype-1 infection (from 1.3% to 91.7%). “The number of patients achieving SVR increased 21-fold from 1,313 in 2010 to an estimated 28,084 in 2015,” Dr. Moon and his associates said.
“We believe that our findings based on the VA health care system might be relevant and informative for other comprehensive health care systems,” providing proof-of-concept that similar results can be achieved if aggressive screening; affordable, tolerable treatment; and open access to all patients are implemented.
The number of Veterans Affairs patients with hepatitis C who have achieved a sustained virologic response to antiviral therapy has escalated so rapidly and reached such a height that the disease may well be eradicated in that health care system within a few years, according to a report in Alimentary Pharmacology and Therapeutics.
The potential public health benefits are substantial, “considering that HCV infection is the most common cause of cirrhosis and liver cancer in the VA and the United States, that the benefits of SVR are long-lasting, and that HCV clearance reduces the risk of liver cancer by 76% and all-cause mortality by 50%,” said Andrew M. Moon, MD, of the division of general internal medicine, University of Washington, Seattle, and his associates.
An estimated 124,662 VA patients currently are infected, and curing them “would substantially reduce the burden of HCV within the entire country and prevent tens of thousands of deaths,” they noted.
The VA dramatically increased the number of patients who were offered treatment in recent years, because it was able to allocate nearly $700 million to offset the high costs of highly effective direct antiviral agents, which in turn made these better-tolerated drugs more widely available at clinics across the country. The VA also removed all treatment prioritization criteria, allowing all patients, not just those with severe disease, to receive highly effective direct antiviral agents. This is “in stark contrast to most health care systems, state Medicaid programs, and insurance carriers in the U.S., which still restrict access ... based on severity of liver disease,” the investigators said (Aliment Pharmacol Ther. 2017 March 8. doi: 10.1111/apt.14021).
To examine the impact of these changes, Dr. Moon and his associates performed a retrospective cohort study, analyzing the electronic medical records of all 105,369 HCV treatment regimens given to 78,947 VA patients (mean age, 56 years) during a 17-year period. They found that annual treatment rates more than doubled from 2,726 to 6,679 patients when pegylated interferon was introduced, declined for a while and then rose modestly to 4,900 patients when boceprevir and telaprevir were introduced, declined again to an all-time low of 2,609 and then rebounded to 9,180 patients when sofosbuvir and simeprevir were introduced, and finally skyrocketed to 31,028 patients when ledipasvir/sofosbuvir and paritaprevir/ritonavir/ombitasvir/dasabuvir were introduced.
Correspondingly, SVR rates rose from less than 25% at the beginning of the study period to a “remarkable” 90.5% at the end. The improvement in SVR rates was even more pronounced among traditionally “hard to treat” cases, such as patients with concomitant cirrhosis (from 11.0% to 87.0%), decompensated cirrhosis (from 14.6% to 85.2%), highly refractory infection (from 16.4% to 89.3%), and genotype-1 infection (from 1.3% to 91.7%). “The number of patients achieving SVR increased 21-fold from 1,313 in 2010 to an estimated 28,084 in 2015,” Dr. Moon and his associates said.
“We believe that our findings based on the VA health care system might be relevant and informative for other comprehensive health care systems,” providing proof-of-concept that similar results can be achieved if aggressive screening; affordable, tolerable treatment; and open access to all patients are implemented.
The number of Veterans Affairs patients with hepatitis C who have achieved a sustained virologic response to antiviral therapy has escalated so rapidly and reached such a height that the disease may well be eradicated in that health care system within a few years, according to a report in Alimentary Pharmacology and Therapeutics.
The potential public health benefits are substantial, “considering that HCV infection is the most common cause of cirrhosis and liver cancer in the VA and the United States, that the benefits of SVR are long-lasting, and that HCV clearance reduces the risk of liver cancer by 76% and all-cause mortality by 50%,” said Andrew M. Moon, MD, of the division of general internal medicine, University of Washington, Seattle, and his associates.
An estimated 124,662 VA patients currently are infected, and curing them “would substantially reduce the burden of HCV within the entire country and prevent tens of thousands of deaths,” they noted.
The VA dramatically increased the number of patients who were offered treatment in recent years, because it was able to allocate nearly $700 million to offset the high costs of highly effective direct antiviral agents, which in turn made these better-tolerated drugs more widely available at clinics across the country. The VA also removed all treatment prioritization criteria, allowing all patients, not just those with severe disease, to receive highly effective direct antiviral agents. This is “in stark contrast to most health care systems, state Medicaid programs, and insurance carriers in the U.S., which still restrict access ... based on severity of liver disease,” the investigators said (Aliment Pharmacol Ther. 2017 March 8. doi: 10.1111/apt.14021).
To examine the impact of these changes, Dr. Moon and his associates performed a retrospective cohort study, analyzing the electronic medical records of all 105,369 HCV treatment regimens given to 78,947 VA patients (mean age, 56 years) during a 17-year period. They found that annual treatment rates more than doubled from 2,726 to 6,679 patients when pegylated interferon was introduced, declined for a while and then rose modestly to 4,900 patients when boceprevir and telaprevir were introduced, declined again to an all-time low of 2,609 and then rebounded to 9,180 patients when sofosbuvir and simeprevir were introduced, and finally skyrocketed to 31,028 patients when ledipasvir/sofosbuvir and paritaprevir/ritonavir/ombitasvir/dasabuvir were introduced.
Correspondingly, SVR rates rose from less than 25% at the beginning of the study period to a “remarkable” 90.5% at the end. The improvement in SVR rates was even more pronounced among traditionally “hard to treat” cases, such as patients with concomitant cirrhosis (from 11.0% to 87.0%), decompensated cirrhosis (from 14.6% to 85.2%), highly refractory infection (from 16.4% to 89.3%), and genotype-1 infection (from 1.3% to 91.7%). “The number of patients achieving SVR increased 21-fold from 1,313 in 2010 to an estimated 28,084 in 2015,” Dr. Moon and his associates said.
“We believe that our findings based on the VA health care system might be relevant and informative for other comprehensive health care systems,” providing proof-of-concept that similar results can be achieved if aggressive screening; affordable, tolerable treatment; and open access to all patients are implemented.
FROM ALIMENTARY PHARMACOLOGY AND THERAPEUTICS
Key clinical point: The number of VA patients with hepatitis C virus who have achieved a sustained virologic response has escalated so rapidly and so high that the disease may be eradicated in that health care system within a few years.
Major finding: SVR rates rose from less than 25% at the beginning of the study period to a “remarkable” 90.5% at the end; the number of patients achieving SVR increased 21-fold from 1,313 in 2010 to an estimated 28,084 in 2015.
Data source: A retrospective cohort study examining all 105,369 antiviral regimens administered within the VA in 1999-2016.
Disclosures: The VA Office of Research and Development funded the study. Dr. Moon and his associates reported having no relevant financial disclosures.