User login
Fatigued absent of medical history
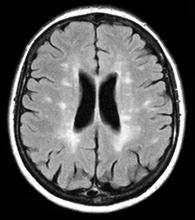
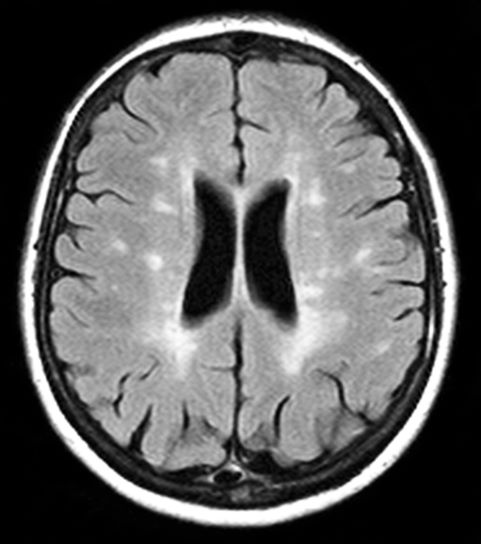
The patient is probably presenting with relapsing-remitting multiple sclerosis (RRMS). MS is characterized by symptomatic episodes that are heralded by symptoms of central nervous system involvement. These attacks last longer than 24 hours and may occur months or years apart and affect different anatomic locations. Consistent with other autoimmune conditions, MS is more common in women. Patients are usually diagnosed between the ages of 20 and 49 years. The condition presents differently from patient to patient; some experience cognitive changes or visual symptoms, while others may have numbness, ataxia, clumsiness, hemiparesis, paraparesis, depression, or seizures. Symptoms can also include fatigue, impaired mobility, mood diagnosed changes, elimination dysfunction, and pain.
Of the four disease courses identified in MS, the most common is RRMS, characterized by a cycle of relapse and remission. In the initial stages, RRMS is characterized largely by an inflammatory pathology which, over time, becomes largely neurodegenerative. Most cases of RRMS evolve to secondary progressive MS after about 15 years. Early in the spectrum of demyelinating disease is clinically isolated syndrome (CIS), defined by a single episode of neurologic symptoms and MRI showing more than two classic lesions seen in MS. CIS patients subsequently will present with a second episode or relapse, at which point the diagnosis of RRMS is usually confirmed.
MS is diagnosed on the basis of clinical findings, exclusion of mimickers, and supporting evidence from the workup, namely MRI of the brain and spinal cord as well as cerebrospinal fluid examination. From a clinical perspective, presentation must align with the constellation of neurologic deficits seen in MS. Typically, the duration of deficit is days to weeks, as seen in the present case. While MRI alone cannot be used to diagnose MS, imaging may confirm diagnosis and offer value in monitoring disease progression in the brain and spinal cord. New lesions on MRI usually occur with relapses in RRMS.
Treatment of MS encompasses immunomodulatory therapy to address the underlying immune disorder together with therapies to relieve symptoms. In general, disease-modifying therapy (DMT) should be considered for patients who have experienced a single demyelinating event and exhibit two or more brain lesions on MRI testing. This recommendation holds true even for patients with CIS or those who have experienced their first clinical event and have MRI features consistent with MS, so long as all other conditions in the differential are ruled out. Pivotal trials support the early initiation of DMT with CIS to delay disability.
Krupa Pandey, MD, Director, Multiple Sclerosis Center, Department of Neurology & Neuroscience Institute, Hackensack University Medical Center; Neurologist, Department of Neurology, Hackensack Meridian Health, Hackensack, NJ
Krupa Pandey, MD, has serve(d) as a speaker or a member of a speakers bureau for: Bristol-Myers Squibb; Biogen; Alexion; Genentech; Sanofi-Genzyme
The patient is probably presenting with relapsing-remitting multiple sclerosis (RRMS). MS is characterized by symptomatic episodes that are heralded by symptoms of central nervous system involvement. These attacks last longer than 24 hours and may occur months or years apart and affect different anatomic locations. Consistent with other autoimmune conditions, MS is more common in women. Patients are usually diagnosed between the ages of 20 and 49 years. The condition presents differently from patient to patient; some experience cognitive changes or visual symptoms, while others may have numbness, ataxia, clumsiness, hemiparesis, paraparesis, depression, or seizures. Symptoms can also include fatigue, impaired mobility, mood diagnosed changes, elimination dysfunction, and pain.
Of the four disease courses identified in MS, the most common is RRMS, characterized by a cycle of relapse and remission. In the initial stages, RRMS is characterized largely by an inflammatory pathology which, over time, becomes largely neurodegenerative. Most cases of RRMS evolve to secondary progressive MS after about 15 years. Early in the spectrum of demyelinating disease is clinically isolated syndrome (CIS), defined by a single episode of neurologic symptoms and MRI showing more than two classic lesions seen in MS. CIS patients subsequently will present with a second episode or relapse, at which point the diagnosis of RRMS is usually confirmed.
MS is diagnosed on the basis of clinical findings, exclusion of mimickers, and supporting evidence from the workup, namely MRI of the brain and spinal cord as well as cerebrospinal fluid examination. From a clinical perspective, presentation must align with the constellation of neurologic deficits seen in MS. Typically, the duration of deficit is days to weeks, as seen in the present case. While MRI alone cannot be used to diagnose MS, imaging may confirm diagnosis and offer value in monitoring disease progression in the brain and spinal cord. New lesions on MRI usually occur with relapses in RRMS.
Treatment of MS encompasses immunomodulatory therapy to address the underlying immune disorder together with therapies to relieve symptoms. In general, disease-modifying therapy (DMT) should be considered for patients who have experienced a single demyelinating event and exhibit two or more brain lesions on MRI testing. This recommendation holds true even for patients with CIS or those who have experienced their first clinical event and have MRI features consistent with MS, so long as all other conditions in the differential are ruled out. Pivotal trials support the early initiation of DMT with CIS to delay disability.
Krupa Pandey, MD, Director, Multiple Sclerosis Center, Department of Neurology & Neuroscience Institute, Hackensack University Medical Center; Neurologist, Department of Neurology, Hackensack Meridian Health, Hackensack, NJ
Krupa Pandey, MD, has serve(d) as a speaker or a member of a speakers bureau for: Bristol-Myers Squibb; Biogen; Alexion; Genentech; Sanofi-Genzyme
The patient is probably presenting with relapsing-remitting multiple sclerosis (RRMS). MS is characterized by symptomatic episodes that are heralded by symptoms of central nervous system involvement. These attacks last longer than 24 hours and may occur months or years apart and affect different anatomic locations. Consistent with other autoimmune conditions, MS is more common in women. Patients are usually diagnosed between the ages of 20 and 49 years. The condition presents differently from patient to patient; some experience cognitive changes or visual symptoms, while others may have numbness, ataxia, clumsiness, hemiparesis, paraparesis, depression, or seizures. Symptoms can also include fatigue, impaired mobility, mood diagnosed changes, elimination dysfunction, and pain.
Of the four disease courses identified in MS, the most common is RRMS, characterized by a cycle of relapse and remission. In the initial stages, RRMS is characterized largely by an inflammatory pathology which, over time, becomes largely neurodegenerative. Most cases of RRMS evolve to secondary progressive MS after about 15 years. Early in the spectrum of demyelinating disease is clinically isolated syndrome (CIS), defined by a single episode of neurologic symptoms and MRI showing more than two classic lesions seen in MS. CIS patients subsequently will present with a second episode or relapse, at which point the diagnosis of RRMS is usually confirmed.
MS is diagnosed on the basis of clinical findings, exclusion of mimickers, and supporting evidence from the workup, namely MRI of the brain and spinal cord as well as cerebrospinal fluid examination. From a clinical perspective, presentation must align with the constellation of neurologic deficits seen in MS. Typically, the duration of deficit is days to weeks, as seen in the present case. While MRI alone cannot be used to diagnose MS, imaging may confirm diagnosis and offer value in monitoring disease progression in the brain and spinal cord. New lesions on MRI usually occur with relapses in RRMS.
Treatment of MS encompasses immunomodulatory therapy to address the underlying immune disorder together with therapies to relieve symptoms. In general, disease-modifying therapy (DMT) should be considered for patients who have experienced a single demyelinating event and exhibit two or more brain lesions on MRI testing. This recommendation holds true even for patients with CIS or those who have experienced their first clinical event and have MRI features consistent with MS, so long as all other conditions in the differential are ruled out. Pivotal trials support the early initiation of DMT with CIS to delay disability.
Krupa Pandey, MD, Director, Multiple Sclerosis Center, Department of Neurology & Neuroscience Institute, Hackensack University Medical Center; Neurologist, Department of Neurology, Hackensack Meridian Health, Hackensack, NJ
Krupa Pandey, MD, has serve(d) as a speaker or a member of a speakers bureau for: Bristol-Myers Squibb; Biogen; Alexion; Genentech; Sanofi-Genzyme
A 51-year-old woman reports that she has been feeling fatigued despite the absence of any significant medical history. Although she usually walks to work, lately she has not had the energy to participate in her daily routine. She notes that over the past 2 weeks, colleagues have asked her if she is feeling well due to unusual ocular symptoms. She explains that several months ago she felt similarly unwell, with fatigue and generalized weakness, but her symptoms seemed to resolve. Upon presentation, she has diplopia on lateral gaze. MRI reveals lesions with high T2 signal intensity.
Multiple Sclerosis: Presentation and Diagnosis
Multiple Sclerosis: The Basics
Guideline gives weak support to trying oral medical cannabis for chronic pain
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
“Evidence alone is not sufficient for clinical decision-making, particularly in chronic pain,” said Jason Busse, DC, PhD, director of Michael G. DeGroote Centre for Medicinal Cannabis Research at McMaster University, Hamilton, Ont., and lead author of a newly released rapid guideline on medical cannabis or cannabinoids for chronic pain.
The recommendations, published online Sept. 9, 2021 in the British Medical Journal, suggest that providers offer patients with chronic pain a trial of noninhaled medical cannabis or cannabinoids if standard care or management is ineffective. However, the “weak” rating attached to the recommendation may compel some clinicians to automatically write off the panel’s recommendations.
“Because of the close balance between benefits and harms and wide variability in patient attitudes, the panel came to the conclusion that [some] patients presented with the current best evidence would likely choose to engage in a trial of medicinal cannabis, if their current care was felt to be suboptimal,” Dr. Busse explained in an interview.
But more importantly, “the recommendation allows for shared decision making to occur, and for different patients to make different decisions based on individual preferences and circumstances,” he said.
Evidence supports improved pain and sleep quality, physical functioning
Evidence supporting the use of medical cannabis in chronic pain is derived from a rigorous systematic review and meta-analysis of 32 studies enrolling 5,174 patients randomized to oral (capsule, spray, sublingual drops) or topical (transdermal cream) medical cannabis or placebo. Of note, three types of cannabinoids were represented: phytocannabinoids, synthetic, and endocannabinoids.
The studies included both patients with chronic noncancer pain (28 studies, n = 3,812) and chronic cancer pain not receiving palliative care (4 studies, n = 1,362). On average, baseline pain scores were a median 6.28 cm on a 10-cm visual analog scale (VAS), and median participant age was 53 years. 60% of trials reporting sex differences enrolled female participants. Overall, patients were followed for roughly 2 months (median, 50 days).
Findings (27 studies, n = 3,939) showed that, compared with placebo, medical cannabis resulted in a small, albeit important, improvement in the proportion of patients experiencing pain relief at or above the minimally important difference (MID) (moderate-certainty evidence, 10% modeled risk difference [RD; 95% confidence interval, 5%-15%] for achieving at least the MID of 1 cm).
Medical cannabis (15 studies, n = 2,425) also provided a small increase in the proportion of patients experiencing improvements in physical functioning at or above the MID (high certainty evidence, 4% modeled RD [95% CI, 0.1%-8%] for achieving at least a MID of 10 points).
Additionally, participants experienced significant improvements in sleep quality, compared with placebo (16 studies, 3,124 participants, high-quality evidence), demonstrating a weighted mean difference of –0.53 cm on a 10-cm VAS (95% CI, –0.75 to –0.30 cm). A total of nine larger trials (n = 2,652, high-certainty evidence) saw a small increase in the proportion of patients experiencing improved sleep quality at or above the MID: 6% modeled RD (95% CI, 2%-9%).
On the other hand, benefits did not extend to emotional, role, or social functioning (high-certainty evidence).
First do no harm: Start low, go slow
While these findings provide a rationale for medical cannabis in chronic pain, exploring options with patients can be challenging. Studies on medical cannabis consistently note that patients want information, but data also show that many providers express a lack of knowledge to provide adequate counseling.
There are also legal hurdles. Despite the authorization of medicinal cannabis across a majority of states and territories, cannabis is still a schedule I substance under the Federal Controlled Substances Act. In addition, the absence of standards around formulations, potency, and dosing has also been cited as a major barrier to recommending medical cannabis, as have concerns about adverse events (AEs), especially with inhaled and tetrahydrocannabinol (THC)-predominant formulations.
Like most medications, medical cannabis dosing should be individualized depending on product, patient, and ability to titrate the dose, but the guidelines provide a general rule of thumb. Providers considering therapeutic noninhaled medical cannabis trials are encouraged to start with a low-dose cannabidiol (CBD) oral tablet, spray, or sublingual oil drops 5 mg twice daily, increasing it by 10 mg every 2-3 days depending on the clinical response (to a maximum daily dose of 40 mg/day). If patient response is unsatisfactory, they should consider adding 1-2.5 mg THC/daily, titrated every 2-7 days to a maximum of 40 mg/day.
Still, an important caveat is whether or not adjunctive CBD alone is effective for chronic pain.
“While we know that one out of seven U.S. adults are using cannabidiol, we know very little about its therapeutic effects when given by itself for pain,” Ziva Cooper, PhD, director of the Cannabis Research Initiative at the University of California, Los Angeles, and an associate professor at-large of psychology and behavioral science, said in an interview. (Dr. Cooper was not involved in the guideline development.)
“But patients tend to self-report that CBD is helpful, and at low doses, we know that it is unlikely to have adverse effects of any significant concern,” Dr. Cooper noted.
Depending on its components, medical cannabis is associated with a wide range of AEs. Studies comprising the evidence base for the guideline reported transient cognitive impairment (relative risk, 2.39; 95% CI, 1.06-5.38), vomiting (RR, 1.46; 95% CI, 1.07-1.99), and drowsiness (RR, 2.14; 95% CI, 1.55-2.95), attention impairment (RR, 4.04; 95% CI, 1.67-9.74), and nausea (RR, 1.59; 95% CI, 1.28-1.99). Of note, findings of a subgroup analysis showed that the risk of dizziness increased with treatment duration, starting at 3 months (test of interaction P = .002).
However, Dr. Cooper explained that, because the included studies were inconsistent in terms of cannabis type (e.g., some looked at synthetic THC or THC-like substances where others looked at a THC/CBD combination) and formulation (capsules, oral mucosal sprays), it’s difficult to tease out component-specific AEs.
“These are really important things to note, especially when you think about different populations that might be using these types of medicines moving forward,” she said.
Toward that end, the guideline specifically states that there is “no reason why the expected benefits would be systematically different among adolescents and emerging adults.”
Among children with cancer, prior study findings reinforce the conclusion that benefits are similar to adults, but studies in this area are limited to end-of-life treatment, childhood cancer with primarily palliative intent, or progressive or relapsed cancer. Because THC’s safety profile is less certain in children, it’s also important to consider adverse neurocognitive effects before initiating a medical cannabis trial in this population.
Navigating the landscape
Although promising, the medical cannabis landscape is undoubtedly difficult to navigate, with land mines ranging from a limited inability to simply pick up a prescribing pad to quality control.
With the exception of three Food and Drug Administration–approved products – dronabinol, cannabidiol Rx, and nabilone – U.S. providers are only able to ‘certify,’ not prescribe, medical cannabis for chronic pain, and only if it is included within the state cannabis board’s list of eligible conditions. (A state-by-state guide is available.)
Quality control also varies by product but is critical. “You want to look for certificates of quality assurance,” Jenny Wilkerson, PhD, a research assistant professor of pharmacodynamics at the University of Florida, Gainesville, said in an interview. (Dr. Wilkerson was not involved in the guideline development.)
“A good dispensary should have that information or at least be willing to get that information, but generally speaking, that is something that patients need to ask for,” she emphasized, noting that “most available mass readouts are not divided by lots.”
Initial counseling and AE monitoring and regular follow-up is important, especially among patients who’ve never tried medical cannabis (or older patients whose prior experience may be limited to weaker recreational marijuana).
Notably, the reliance on medical dispensaries to deliver the right information at the right time may prove to be faulty. While recent data show that frontline dispensary workers regularly provide information to customers on their medical conditions and available products, they rarely, if ever, base recommendations on provider input, and never or rarely discuss potential AEs and other risks.
Per the new guideline, inexperienced patients should be seen monthly until a stable dose is achieved; longer times between visits can be considered in those who are more experienced. Still, patients should be advised to contact their provider when pain relief or other goals are insufficient, or when response or problematic AEs occur. This facilitates down-titration to a previously tolerated dose, up-titration in CBD and/or THC, or a different route of administration/formulation altogether.
Dr. Wilkerson pointed out that follow-up visits also provide an opportunity to do a blood draw and ask the lab to conduct pharmacokinetic analysis.
If possible, “ask patients to [ensure that they] take a standard dose before the visit so that the lab can assess the blood percentage of primary compounds and metabolites in the product that they are using,” she explained, noting that the information is helping to determine how “the different ratios may be affecting therapeutic response in individual patients.”
Granted, the guideline is only a start. But it is a good one.
“A lot of physicians want to be able to hang their hat on evidence of the safety and efficacy of these products, and the analysis that was leveraged for this guideline was very rigorous,” Dr. Cooper said.
Not only do they reinforce that “oral cannabinoids can produce small improvements in pain and provide a dosing structure that minimizes risk to the patient, [but they] should be able to help educate physicians who [are looking] for a sense of what the literature tells us at this time,” she added.
“With chronic pain, we often find that different treatments will show small potential benefits and they have a certain risk profile,” Dr. Busse said.
“It’s almost impossible to know what patients think about this option unless you present them with the evidence and ask them to make a decision based on their values and preferences,” he said.
The Michael G. DeGroote Centre for Medicinal Cannabis Research funded the MAGIC Evidence Ecosystem Foundation to support the creation of the guideline. The center receives no funding from industry Dr. Busse, Dr. Cooper, and Dr. Wilkerson reported having no relevant financial relationships.
FROM THE BMJ
MS plus depression can increase risk of death, vascular disease
, a new study has found. “The effects of depression and MS on all-cause mortality are synergistic,” wrote lead author Raffaele Palladino, MD, PhD, research associate, faculty of medicine, Imperial College London.
The study was published in Neurology.
To assess the association between depression, vascular disease, and death in patients with MS, the researchers launched a population-based retrospective cohort study that reviewed English medical records from January 1987 to December 2018 and matched people with and without MS. Ultimately, 12,251 people with MS were matched with 72,572 controls. At baseline, 21% of the MS group (n = 2,535) and 9% of the controls (n = 6,278) had depression. Women were the majority in both cohorts and were more likely than men to be depressed.
People with both MS and depression had an all-cause mortality rate of 10.3 cases per 100,000 person-years (95% confidence interval, 9.17-11.57), compared with 10.6 for people with MS without depression (95% CI, 9.99-11.21), 3.6 for people with depression but not MS (95% CI, 3.18-4.05), and 2.5 for people with neither condition (95% CI, 2.42-2.64). Compared with controls without depression, the 10-year hazard of all-cause mortality was increasingly greater in controls with depression (hazard ratio, 1.75; 95% CI, 1.59-1.91), people with MS but not depression (HR, 3.88; 95% CI, 3.66-4.10), and people with MS and depression (HR, 5.43; 95% CI, 4.88-5.96). Overall, 14% of the observed effect on mortality was attributable to the interaction between MS status and depression.
As for vascular diseases, people with MS had an increased risk regardless of their depression status. That said, people with MS and depression (HR, 3.30; 95% CI, 2.37-4.23) had a notably higher risk than people with MS and no depression (HR, 1.48; 95% CI, 1.23-1.74). Women with MS and depression also had a greater risk of vascular disease than women with MS and no depression, while men with MS did not have significantly different risks of acute coronary syndrome or composite macrovascular disease than those in the control group who did not suffer from depression.
Does treating depression decrease the likelihood of vascular disease?
“The take-home message for me is the importance of treating depression in this population, in which we see it with great regularity,” Joseph Berger, MD, professor of neurology and associate chief of the multiple sclerosis division at the University of Pennsylvania, Philadelphia, said in an interview. “The question that I have is: If you treat depression in an individual with MS or an individual who is simply depressed and thus at risk for the subsequent development of vascular disease, does it decrease the likelihood of their subsequent development of vascular disease in comparison to had you not?
“I presume it does,” he added, noting that “the theories underlying why depression would increase one’s risk of subsequent vascular disease are enumerated by the authors, including such things as increased inflammation. Now, the inflammation may be contributing to the depression, or the depression may be contributing to the inflammation; it may be one of those chicken-and-egg scenarios. But if you decrease the depression, do you thereby decrease the inflammation, which has a pernicious effect on endothelial cells and increases one’s vascular risk?
“Alternatively, lifestyle in depressed patients is also altered,” he said. “They’re far less likely to engage in exercise, healthy habits, and healthy diets, and more likely perhaps to smoke. These all need to be addressed, but this study certainly gives you a greater impetus as a MS neurologist to address the issue of depression, realizing that there is also this comorbidity of vascular disease.”
Evaluating the biological interaction between MS and depression
Based on this and other studies, the joint effect of MS and depression on all-cause mortality may qualify as a biological interaction, Amber Salter, PhD, of the University of Texas Southwestern Medical Center, Dallas, wrote in an accompanying editorial.
“Biological interactions consider whether the joint effect of two factors follow an additive pattern, or the joint effect of two factors is greater than the sum of the individual effects for each factor alone,” she wrote. And though the interaction was not found to be present for vascular disease and cardiovascular mortality, it was for all-cause mortality.
“When warranted, the evaluation of biological interactions in future studies should be considered to provide insight on target subpopulations for interventions or test for potential mechanistic forms of interaction,” she added.
Dr. Salter highlighted the study’s strengths, including a large sample size and six controls matched to each MS patient. She also stated that the researchers’ inability to control for risk factors like body mass index and physical activity means the 14% increase in mortality “may not be a large absolute increase in mortality when other covariates cannot be considered.” In addition, their lack of data on suicide – and its association with depression – offers up the possibility that increases in mortality could be tied to a “potentially modifiable risk” as opposed to a biologically increased one.
In acknowledging their study’s limitations, the authors stated that body mass index, though an important vascular risk factor, has a “modest” association with mortality, and that the average annual suicide rate in the MS population – though higher than in the non-MS population – is still “relatively low.”
Two of the authors disclosed receiving support, including grants and research funding, from various institutions and organizations in the United Kingdom, the United States, and Canada, as well as several pharmaceutical companies. Dr. Salter reported no relevant disclosures.
, a new study has found. “The effects of depression and MS on all-cause mortality are synergistic,” wrote lead author Raffaele Palladino, MD, PhD, research associate, faculty of medicine, Imperial College London.
The study was published in Neurology.
To assess the association between depression, vascular disease, and death in patients with MS, the researchers launched a population-based retrospective cohort study that reviewed English medical records from January 1987 to December 2018 and matched people with and without MS. Ultimately, 12,251 people with MS were matched with 72,572 controls. At baseline, 21% of the MS group (n = 2,535) and 9% of the controls (n = 6,278) had depression. Women were the majority in both cohorts and were more likely than men to be depressed.
People with both MS and depression had an all-cause mortality rate of 10.3 cases per 100,000 person-years (95% confidence interval, 9.17-11.57), compared with 10.6 for people with MS without depression (95% CI, 9.99-11.21), 3.6 for people with depression but not MS (95% CI, 3.18-4.05), and 2.5 for people with neither condition (95% CI, 2.42-2.64). Compared with controls without depression, the 10-year hazard of all-cause mortality was increasingly greater in controls with depression (hazard ratio, 1.75; 95% CI, 1.59-1.91), people with MS but not depression (HR, 3.88; 95% CI, 3.66-4.10), and people with MS and depression (HR, 5.43; 95% CI, 4.88-5.96). Overall, 14% of the observed effect on mortality was attributable to the interaction between MS status and depression.
As for vascular diseases, people with MS had an increased risk regardless of their depression status. That said, people with MS and depression (HR, 3.30; 95% CI, 2.37-4.23) had a notably higher risk than people with MS and no depression (HR, 1.48; 95% CI, 1.23-1.74). Women with MS and depression also had a greater risk of vascular disease than women with MS and no depression, while men with MS did not have significantly different risks of acute coronary syndrome or composite macrovascular disease than those in the control group who did not suffer from depression.
Does treating depression decrease the likelihood of vascular disease?
“The take-home message for me is the importance of treating depression in this population, in which we see it with great regularity,” Joseph Berger, MD, professor of neurology and associate chief of the multiple sclerosis division at the University of Pennsylvania, Philadelphia, said in an interview. “The question that I have is: If you treat depression in an individual with MS or an individual who is simply depressed and thus at risk for the subsequent development of vascular disease, does it decrease the likelihood of their subsequent development of vascular disease in comparison to had you not?
“I presume it does,” he added, noting that “the theories underlying why depression would increase one’s risk of subsequent vascular disease are enumerated by the authors, including such things as increased inflammation. Now, the inflammation may be contributing to the depression, or the depression may be contributing to the inflammation; it may be one of those chicken-and-egg scenarios. But if you decrease the depression, do you thereby decrease the inflammation, which has a pernicious effect on endothelial cells and increases one’s vascular risk?
“Alternatively, lifestyle in depressed patients is also altered,” he said. “They’re far less likely to engage in exercise, healthy habits, and healthy diets, and more likely perhaps to smoke. These all need to be addressed, but this study certainly gives you a greater impetus as a MS neurologist to address the issue of depression, realizing that there is also this comorbidity of vascular disease.”
Evaluating the biological interaction between MS and depression
Based on this and other studies, the joint effect of MS and depression on all-cause mortality may qualify as a biological interaction, Amber Salter, PhD, of the University of Texas Southwestern Medical Center, Dallas, wrote in an accompanying editorial.
“Biological interactions consider whether the joint effect of two factors follow an additive pattern, or the joint effect of two factors is greater than the sum of the individual effects for each factor alone,” she wrote. And though the interaction was not found to be present for vascular disease and cardiovascular mortality, it was for all-cause mortality.
“When warranted, the evaluation of biological interactions in future studies should be considered to provide insight on target subpopulations for interventions or test for potential mechanistic forms of interaction,” she added.
Dr. Salter highlighted the study’s strengths, including a large sample size and six controls matched to each MS patient. She also stated that the researchers’ inability to control for risk factors like body mass index and physical activity means the 14% increase in mortality “may not be a large absolute increase in mortality when other covariates cannot be considered.” In addition, their lack of data on suicide – and its association with depression – offers up the possibility that increases in mortality could be tied to a “potentially modifiable risk” as opposed to a biologically increased one.
In acknowledging their study’s limitations, the authors stated that body mass index, though an important vascular risk factor, has a “modest” association with mortality, and that the average annual suicide rate in the MS population – though higher than in the non-MS population – is still “relatively low.”
Two of the authors disclosed receiving support, including grants and research funding, from various institutions and organizations in the United Kingdom, the United States, and Canada, as well as several pharmaceutical companies. Dr. Salter reported no relevant disclosures.
, a new study has found. “The effects of depression and MS on all-cause mortality are synergistic,” wrote lead author Raffaele Palladino, MD, PhD, research associate, faculty of medicine, Imperial College London.
The study was published in Neurology.
To assess the association between depression, vascular disease, and death in patients with MS, the researchers launched a population-based retrospective cohort study that reviewed English medical records from January 1987 to December 2018 and matched people with and without MS. Ultimately, 12,251 people with MS were matched with 72,572 controls. At baseline, 21% of the MS group (n = 2,535) and 9% of the controls (n = 6,278) had depression. Women were the majority in both cohorts and were more likely than men to be depressed.
People with both MS and depression had an all-cause mortality rate of 10.3 cases per 100,000 person-years (95% confidence interval, 9.17-11.57), compared with 10.6 for people with MS without depression (95% CI, 9.99-11.21), 3.6 for people with depression but not MS (95% CI, 3.18-4.05), and 2.5 for people with neither condition (95% CI, 2.42-2.64). Compared with controls without depression, the 10-year hazard of all-cause mortality was increasingly greater in controls with depression (hazard ratio, 1.75; 95% CI, 1.59-1.91), people with MS but not depression (HR, 3.88; 95% CI, 3.66-4.10), and people with MS and depression (HR, 5.43; 95% CI, 4.88-5.96). Overall, 14% of the observed effect on mortality was attributable to the interaction between MS status and depression.
As for vascular diseases, people with MS had an increased risk regardless of their depression status. That said, people with MS and depression (HR, 3.30; 95% CI, 2.37-4.23) had a notably higher risk than people with MS and no depression (HR, 1.48; 95% CI, 1.23-1.74). Women with MS and depression also had a greater risk of vascular disease than women with MS and no depression, while men with MS did not have significantly different risks of acute coronary syndrome or composite macrovascular disease than those in the control group who did not suffer from depression.
Does treating depression decrease the likelihood of vascular disease?
“The take-home message for me is the importance of treating depression in this population, in which we see it with great regularity,” Joseph Berger, MD, professor of neurology and associate chief of the multiple sclerosis division at the University of Pennsylvania, Philadelphia, said in an interview. “The question that I have is: If you treat depression in an individual with MS or an individual who is simply depressed and thus at risk for the subsequent development of vascular disease, does it decrease the likelihood of their subsequent development of vascular disease in comparison to had you not?
“I presume it does,” he added, noting that “the theories underlying why depression would increase one’s risk of subsequent vascular disease are enumerated by the authors, including such things as increased inflammation. Now, the inflammation may be contributing to the depression, or the depression may be contributing to the inflammation; it may be one of those chicken-and-egg scenarios. But if you decrease the depression, do you thereby decrease the inflammation, which has a pernicious effect on endothelial cells and increases one’s vascular risk?
“Alternatively, lifestyle in depressed patients is also altered,” he said. “They’re far less likely to engage in exercise, healthy habits, and healthy diets, and more likely perhaps to smoke. These all need to be addressed, but this study certainly gives you a greater impetus as a MS neurologist to address the issue of depression, realizing that there is also this comorbidity of vascular disease.”
Evaluating the biological interaction between MS and depression
Based on this and other studies, the joint effect of MS and depression on all-cause mortality may qualify as a biological interaction, Amber Salter, PhD, of the University of Texas Southwestern Medical Center, Dallas, wrote in an accompanying editorial.
“Biological interactions consider whether the joint effect of two factors follow an additive pattern, or the joint effect of two factors is greater than the sum of the individual effects for each factor alone,” she wrote. And though the interaction was not found to be present for vascular disease and cardiovascular mortality, it was for all-cause mortality.
“When warranted, the evaluation of biological interactions in future studies should be considered to provide insight on target subpopulations for interventions or test for potential mechanistic forms of interaction,” she added.
Dr. Salter highlighted the study’s strengths, including a large sample size and six controls matched to each MS patient. She also stated that the researchers’ inability to control for risk factors like body mass index and physical activity means the 14% increase in mortality “may not be a large absolute increase in mortality when other covariates cannot be considered.” In addition, their lack of data on suicide – and its association with depression – offers up the possibility that increases in mortality could be tied to a “potentially modifiable risk” as opposed to a biologically increased one.
In acknowledging their study’s limitations, the authors stated that body mass index, though an important vascular risk factor, has a “modest” association with mortality, and that the average annual suicide rate in the MS population – though higher than in the non-MS population – is still “relatively low.”
Two of the authors disclosed receiving support, including grants and research funding, from various institutions and organizations in the United Kingdom, the United States, and Canada, as well as several pharmaceutical companies. Dr. Salter reported no relevant disclosures.
FROM NEUROLOGY
MRI is a poor disability predictor in secondary progressive MS
, new research suggests. Analysis from the phase 3 ASCEND trial of nearly 900 patients showed that MRI measures were not associated with worsening of scores on the Expanded Disability Status Scale (EDSS), the most widely used physical outcome measure.
The few associations that were shown between MRI measures and clinical outcomes “were with the newer and possibly more sensitive outcomes” – the Timed 25-Foot Walk (T25FW) and Nine-Hole Peg Test (NHPT), wrote the investigators, led by Marcus W. Koch, MD, PhD, associate professor of neurology in the MS program at the University of Calgary, Canada.
However, “it is unclear if these associations are clinically meaningful,” they added.
Worsening on the NHPT at 48 weeks was associated with a 0.86% loss in normalized brain volume; worsening at 96 weeks was associated with a 1.47% loss.
The findings were published online July 26 in the Multiple Sclerosis Journal.
ASCEND data analysis
Although brain volume loss occurs in all forms of MS, it is believed to be particularly relevant in SPMS. Clinical trials often use MRI measures of brain volume as endpoints, likely on the assumption that these measures indicate worsening disability.
However, brain volume loss proceeds slowly. Changes that occur during the typical 2-year study period may not be associated with significant physical or cognitive disability.
In the current study, investigators examined data from the ASCEND trial, which assessed the use of natalizumab for patients with SPMS, to examine these potential associations. Eligible participants in ASCEND were between ages 18 and 58 years, had had SPMS for 2 or more years, had had disability progression during the previous year, and had an EDSS score between 3.0 and 6.5 at baseline.
Participants underwent gadolinium-enhanced cranial MRI at screening and at 24, 48, 72, and 96 weeks. MRI outcomes included normalized brain volume, normalized cortical gray matter volume, and normalized whole gray matter volume. The ASCEND investigators also examined the number and volume of T2 and contrast-enhancing lesions.
The study’s clinical outcomes included scores on the EDSS, T25FW, and NHPT, which were administered at baseline and every 12 weeks thereafter. Participants also underwent the Symbol Digit Modalities Test (SDMT), which is a cognitive assessment, at baseline and every 4 weeks thereafter. In addition, 3-month confirmed disability progression was measured every 12 weeks.
Few significant associations
The investigators’ analysis included 889 patients (61.9% women; median age, 48 years). The median EDSS score at screening was 6.
Brain volume measures decreased consistently during follow-up. Mean volume loss at 96 weeks was about 1%. In contrast, T2 lesion volume changed little during follow-up. The cumulative number of contrast-enhancing lesions and the cumulative number of new or newly enlarging T2 lesions increased steadily during follow-up.
For an increasing number of participants, scores on the EDSS, NHPT, and T25FW worsened significantly during follow-up. Performance on SDMT, however, changed little. Of all the clinical measures, the NHPT was most consistently associated with MRI measures.
Among patients whose NHPT score worsened at 48 weeks, there was greater loss of normalized brain volume (0.86%, P = .02), normalized cortical gray matter volume (1.15%, P = .03), and normalized whole gray matter volume (1.08%, P = .03) than among those whose NHPT score did not worsen.
Among patients whose NHPT score worsened at 96 weeks, there was greater normalized brain volume loss (1.47%, P = .002), greater increase in T2 lesion volume (4.68%, P = .02), and a greater number of cumulative new or newly enlarging T2 lesions (7.81, P = .03) than those whose NHPT score did not worsen.
After adjusting the data for covariables, the investigators found few significant associations between MRI measures and clinical outcomes. Worsening on the EDSS and SDMT was not associated with any MRI outcome.
Important disability contributors missed
The odds ratio of 3-month confirmed worsening on the T25FW at 96 weeks was 2.25 for patients with more than 10 cumulative new or newly enlarging T2 lesions (P = .03). The OR of 3-month confirmed worsening on the NHPT at 96 weeks was 3.04 for patients with more than 10 such lesions (P = .03).
Greater normalized brain volume loss at 48 weeks was associated with a greater risk for worsening disability on the NHPT at 48 and 96 weeks. For patients with a volume loss greater than 1.5%, the OR of worsening NHPT at 96 weeks was 4.69 (P = .05).
Although previous cross-sectional studies have shown correlations between brain volume and cognitive dysfunction, the current investigators found no association between change in SDMT performance and MRI measures.
From the ASCEND dataset, they found that performance on the SDMT unexpectedly improved with time, perhaps because of a practice effect.
“The SDMT may therefore not adequately reflect the steady cognitive decline that people with SPMS experience,” the investigators wrote.
The lack of association between MRI measures and clinical outcomes may indicate that traditional MRI does not measure important contributors to disability, they noted.
“Although the investigated volume measures in this study are currently the most commonly used in clinical trials, newer MRI metrics such as thalamic or corpus callosum atrophy may have a closer relation to clinical outcome,” they added.
‘Interesting and provocative’
Commenting on the findings, E. Ann Yeh, MD, director of the Pediatric MS and Neuroinflammatory Disorders Program at the Hospital for Sick Children, Toronto, called the study “interesting and provocative.”
“Other studies previously have shown associations between disability and progression, but many have been cross-sectional,” said Dr. Yeh, who was not involved with the research.
The current study is longitudinal and analyzes carefully documented follow-up data from a clinical trial, she noted. However, the 2-year follow-up period was short, considering the pace at which whole brain volume change occurs, Dr. Yeh said.
Some patients with MS have greater brain volume loss than others. Because of this variability, researchers often examine a population’s average brain volume loss. “When you look at averages, it makes it more difficult to understand if the larger brain volume losses are actually associated with change,” said Dr. Yeh.
She noted that because the study population had high EDSS scores at baseline, it is not surprising that the NHPT and the T25FW were more strongly associated with change in brain volume than the EDSS was. Large changes in EDSS score probably did not occur during follow-up, she added.
“We’ll continue to use the EDSS, because it’s what we have,” said Dr. Yeh. However, newer measures, such as the NHPT and the T25FW, may provide better information, she said. Similarly, composite measures of cognition, such as the Brief International Cognitive Assessment for MS, may be superior to the SDMT but take longer to administer.
“We need to look more deeply at which MRI measures are the best for predicting outcome and that correlate well in a short period of time,” said Dr. Yeh.
These measures could include specific regional brain volumes “and more advanced measures that look at axonal injury or axonal loss.” Studies with longer follow-up are also necessary, she concluded.
The investigators and Dr. Yeh have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests. Analysis from the phase 3 ASCEND trial of nearly 900 patients showed that MRI measures were not associated with worsening of scores on the Expanded Disability Status Scale (EDSS), the most widely used physical outcome measure.
The few associations that were shown between MRI measures and clinical outcomes “were with the newer and possibly more sensitive outcomes” – the Timed 25-Foot Walk (T25FW) and Nine-Hole Peg Test (NHPT), wrote the investigators, led by Marcus W. Koch, MD, PhD, associate professor of neurology in the MS program at the University of Calgary, Canada.
However, “it is unclear if these associations are clinically meaningful,” they added.
Worsening on the NHPT at 48 weeks was associated with a 0.86% loss in normalized brain volume; worsening at 96 weeks was associated with a 1.47% loss.
The findings were published online July 26 in the Multiple Sclerosis Journal.
ASCEND data analysis
Although brain volume loss occurs in all forms of MS, it is believed to be particularly relevant in SPMS. Clinical trials often use MRI measures of brain volume as endpoints, likely on the assumption that these measures indicate worsening disability.
However, brain volume loss proceeds slowly. Changes that occur during the typical 2-year study period may not be associated with significant physical or cognitive disability.
In the current study, investigators examined data from the ASCEND trial, which assessed the use of natalizumab for patients with SPMS, to examine these potential associations. Eligible participants in ASCEND were between ages 18 and 58 years, had had SPMS for 2 or more years, had had disability progression during the previous year, and had an EDSS score between 3.0 and 6.5 at baseline.
Participants underwent gadolinium-enhanced cranial MRI at screening and at 24, 48, 72, and 96 weeks. MRI outcomes included normalized brain volume, normalized cortical gray matter volume, and normalized whole gray matter volume. The ASCEND investigators also examined the number and volume of T2 and contrast-enhancing lesions.
The study’s clinical outcomes included scores on the EDSS, T25FW, and NHPT, which were administered at baseline and every 12 weeks thereafter. Participants also underwent the Symbol Digit Modalities Test (SDMT), which is a cognitive assessment, at baseline and every 4 weeks thereafter. In addition, 3-month confirmed disability progression was measured every 12 weeks.
Few significant associations
The investigators’ analysis included 889 patients (61.9% women; median age, 48 years). The median EDSS score at screening was 6.
Brain volume measures decreased consistently during follow-up. Mean volume loss at 96 weeks was about 1%. In contrast, T2 lesion volume changed little during follow-up. The cumulative number of contrast-enhancing lesions and the cumulative number of new or newly enlarging T2 lesions increased steadily during follow-up.
For an increasing number of participants, scores on the EDSS, NHPT, and T25FW worsened significantly during follow-up. Performance on SDMT, however, changed little. Of all the clinical measures, the NHPT was most consistently associated with MRI measures.
Among patients whose NHPT score worsened at 48 weeks, there was greater loss of normalized brain volume (0.86%, P = .02), normalized cortical gray matter volume (1.15%, P = .03), and normalized whole gray matter volume (1.08%, P = .03) than among those whose NHPT score did not worsen.
Among patients whose NHPT score worsened at 96 weeks, there was greater normalized brain volume loss (1.47%, P = .002), greater increase in T2 lesion volume (4.68%, P = .02), and a greater number of cumulative new or newly enlarging T2 lesions (7.81, P = .03) than those whose NHPT score did not worsen.
After adjusting the data for covariables, the investigators found few significant associations between MRI measures and clinical outcomes. Worsening on the EDSS and SDMT was not associated with any MRI outcome.
Important disability contributors missed
The odds ratio of 3-month confirmed worsening on the T25FW at 96 weeks was 2.25 for patients with more than 10 cumulative new or newly enlarging T2 lesions (P = .03). The OR of 3-month confirmed worsening on the NHPT at 96 weeks was 3.04 for patients with more than 10 such lesions (P = .03).
Greater normalized brain volume loss at 48 weeks was associated with a greater risk for worsening disability on the NHPT at 48 and 96 weeks. For patients with a volume loss greater than 1.5%, the OR of worsening NHPT at 96 weeks was 4.69 (P = .05).
Although previous cross-sectional studies have shown correlations between brain volume and cognitive dysfunction, the current investigators found no association between change in SDMT performance and MRI measures.
From the ASCEND dataset, they found that performance on the SDMT unexpectedly improved with time, perhaps because of a practice effect.
“The SDMT may therefore not adequately reflect the steady cognitive decline that people with SPMS experience,” the investigators wrote.
The lack of association between MRI measures and clinical outcomes may indicate that traditional MRI does not measure important contributors to disability, they noted.
“Although the investigated volume measures in this study are currently the most commonly used in clinical trials, newer MRI metrics such as thalamic or corpus callosum atrophy may have a closer relation to clinical outcome,” they added.
‘Interesting and provocative’
Commenting on the findings, E. Ann Yeh, MD, director of the Pediatric MS and Neuroinflammatory Disorders Program at the Hospital for Sick Children, Toronto, called the study “interesting and provocative.”
“Other studies previously have shown associations between disability and progression, but many have been cross-sectional,” said Dr. Yeh, who was not involved with the research.
The current study is longitudinal and analyzes carefully documented follow-up data from a clinical trial, she noted. However, the 2-year follow-up period was short, considering the pace at which whole brain volume change occurs, Dr. Yeh said.
Some patients with MS have greater brain volume loss than others. Because of this variability, researchers often examine a population’s average brain volume loss. “When you look at averages, it makes it more difficult to understand if the larger brain volume losses are actually associated with change,” said Dr. Yeh.
She noted that because the study population had high EDSS scores at baseline, it is not surprising that the NHPT and the T25FW were more strongly associated with change in brain volume than the EDSS was. Large changes in EDSS score probably did not occur during follow-up, she added.
“We’ll continue to use the EDSS, because it’s what we have,” said Dr. Yeh. However, newer measures, such as the NHPT and the T25FW, may provide better information, she said. Similarly, composite measures of cognition, such as the Brief International Cognitive Assessment for MS, may be superior to the SDMT but take longer to administer.
“We need to look more deeply at which MRI measures are the best for predicting outcome and that correlate well in a short period of time,” said Dr. Yeh.
These measures could include specific regional brain volumes “and more advanced measures that look at axonal injury or axonal loss.” Studies with longer follow-up are also necessary, she concluded.
The investigators and Dr. Yeh have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests. Analysis from the phase 3 ASCEND trial of nearly 900 patients showed that MRI measures were not associated with worsening of scores on the Expanded Disability Status Scale (EDSS), the most widely used physical outcome measure.
The few associations that were shown between MRI measures and clinical outcomes “were with the newer and possibly more sensitive outcomes” – the Timed 25-Foot Walk (T25FW) and Nine-Hole Peg Test (NHPT), wrote the investigators, led by Marcus W. Koch, MD, PhD, associate professor of neurology in the MS program at the University of Calgary, Canada.
However, “it is unclear if these associations are clinically meaningful,” they added.
Worsening on the NHPT at 48 weeks was associated with a 0.86% loss in normalized brain volume; worsening at 96 weeks was associated with a 1.47% loss.
The findings were published online July 26 in the Multiple Sclerosis Journal.
ASCEND data analysis
Although brain volume loss occurs in all forms of MS, it is believed to be particularly relevant in SPMS. Clinical trials often use MRI measures of brain volume as endpoints, likely on the assumption that these measures indicate worsening disability.
However, brain volume loss proceeds slowly. Changes that occur during the typical 2-year study period may not be associated with significant physical or cognitive disability.
In the current study, investigators examined data from the ASCEND trial, which assessed the use of natalizumab for patients with SPMS, to examine these potential associations. Eligible participants in ASCEND were between ages 18 and 58 years, had had SPMS for 2 or more years, had had disability progression during the previous year, and had an EDSS score between 3.0 and 6.5 at baseline.
Participants underwent gadolinium-enhanced cranial MRI at screening and at 24, 48, 72, and 96 weeks. MRI outcomes included normalized brain volume, normalized cortical gray matter volume, and normalized whole gray matter volume. The ASCEND investigators also examined the number and volume of T2 and contrast-enhancing lesions.
The study’s clinical outcomes included scores on the EDSS, T25FW, and NHPT, which were administered at baseline and every 12 weeks thereafter. Participants also underwent the Symbol Digit Modalities Test (SDMT), which is a cognitive assessment, at baseline and every 4 weeks thereafter. In addition, 3-month confirmed disability progression was measured every 12 weeks.
Few significant associations
The investigators’ analysis included 889 patients (61.9% women; median age, 48 years). The median EDSS score at screening was 6.
Brain volume measures decreased consistently during follow-up. Mean volume loss at 96 weeks was about 1%. In contrast, T2 lesion volume changed little during follow-up. The cumulative number of contrast-enhancing lesions and the cumulative number of new or newly enlarging T2 lesions increased steadily during follow-up.
For an increasing number of participants, scores on the EDSS, NHPT, and T25FW worsened significantly during follow-up. Performance on SDMT, however, changed little. Of all the clinical measures, the NHPT was most consistently associated with MRI measures.
Among patients whose NHPT score worsened at 48 weeks, there was greater loss of normalized brain volume (0.86%, P = .02), normalized cortical gray matter volume (1.15%, P = .03), and normalized whole gray matter volume (1.08%, P = .03) than among those whose NHPT score did not worsen.
Among patients whose NHPT score worsened at 96 weeks, there was greater normalized brain volume loss (1.47%, P = .002), greater increase in T2 lesion volume (4.68%, P = .02), and a greater number of cumulative new or newly enlarging T2 lesions (7.81, P = .03) than those whose NHPT score did not worsen.
After adjusting the data for covariables, the investigators found few significant associations between MRI measures and clinical outcomes. Worsening on the EDSS and SDMT was not associated with any MRI outcome.
Important disability contributors missed
The odds ratio of 3-month confirmed worsening on the T25FW at 96 weeks was 2.25 for patients with more than 10 cumulative new or newly enlarging T2 lesions (P = .03). The OR of 3-month confirmed worsening on the NHPT at 96 weeks was 3.04 for patients with more than 10 such lesions (P = .03).
Greater normalized brain volume loss at 48 weeks was associated with a greater risk for worsening disability on the NHPT at 48 and 96 weeks. For patients with a volume loss greater than 1.5%, the OR of worsening NHPT at 96 weeks was 4.69 (P = .05).
Although previous cross-sectional studies have shown correlations between brain volume and cognitive dysfunction, the current investigators found no association between change in SDMT performance and MRI measures.
From the ASCEND dataset, they found that performance on the SDMT unexpectedly improved with time, perhaps because of a practice effect.
“The SDMT may therefore not adequately reflect the steady cognitive decline that people with SPMS experience,” the investigators wrote.
The lack of association between MRI measures and clinical outcomes may indicate that traditional MRI does not measure important contributors to disability, they noted.
“Although the investigated volume measures in this study are currently the most commonly used in clinical trials, newer MRI metrics such as thalamic or corpus callosum atrophy may have a closer relation to clinical outcome,” they added.
‘Interesting and provocative’
Commenting on the findings, E. Ann Yeh, MD, director of the Pediatric MS and Neuroinflammatory Disorders Program at the Hospital for Sick Children, Toronto, called the study “interesting and provocative.”
“Other studies previously have shown associations between disability and progression, but many have been cross-sectional,” said Dr. Yeh, who was not involved with the research.
The current study is longitudinal and analyzes carefully documented follow-up data from a clinical trial, she noted. However, the 2-year follow-up period was short, considering the pace at which whole brain volume change occurs, Dr. Yeh said.
Some patients with MS have greater brain volume loss than others. Because of this variability, researchers often examine a population’s average brain volume loss. “When you look at averages, it makes it more difficult to understand if the larger brain volume losses are actually associated with change,” said Dr. Yeh.
She noted that because the study population had high EDSS scores at baseline, it is not surprising that the NHPT and the T25FW were more strongly associated with change in brain volume than the EDSS was. Large changes in EDSS score probably did not occur during follow-up, she added.
“We’ll continue to use the EDSS, because it’s what we have,” said Dr. Yeh. However, newer measures, such as the NHPT and the T25FW, may provide better information, she said. Similarly, composite measures of cognition, such as the Brief International Cognitive Assessment for MS, may be superior to the SDMT but take longer to administer.
“We need to look more deeply at which MRI measures are the best for predicting outcome and that correlate well in a short period of time,” said Dr. Yeh.
These measures could include specific regional brain volumes “and more advanced measures that look at axonal injury or axonal loss.” Studies with longer follow-up are also necessary, she concluded.
The investigators and Dr. Yeh have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From Multiple Sclerosis Journal
NIH to study COVID vaccine booster in people with autoimmune disease
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
Progressive disability in MS explained?
Results from a retrospective study show that complete resolution of brain lesions on MRI was more common among patients with myelin-oligodendrocyte-glycoprotein-IgG-associated disorder (MOGAD). Complete resolution occurred in 72% of the group with MOGAD, versus 17% of those with MS and 14% of those with aquaporin-4-positive neuromyelitis optica spectrum disorder (AQP4+ NMOSD).
“What we found was, with MOGAD in particular, many of the lesions resolved completely,” said co-investigator Eoin Flanagan, MBBCh, neurologist, Mayo Clinic, Rochester, Minn. “That fits with MOGAD having a fairly good prognosis and patients not developing much long-term disability with that disease,” he said.
The researchers also studied whether scarring may account for the absence of slowly progressive disability among patients with AQP4+ NMOSD and MOGAD compared with patients with MS. “The differences in scarring that we found will help physicians distinguish these three diseases more easily to aid in diagnosis. More importantly, our findings improve our understanding of the mechanisms of nerve damage in these three diseases and may suggest an important role of such scars in the development of long-term disability in MS,” Dr. Flanagan said in a statement.
The findings were published online July 14 in Neurology.
Lesion evolution
MOGAD, AQP4+ NMOSD, and MS are inflammatory demyelinating disorders that share certain manifestations. However, these disorders differ in important ways, including the severity of attacks and their clinical course.
Although patients with MOGAD and AQP4+ NMOSD generally have severe attacks that bring major disability, the clinical course of these disorders is better than initial attacks would suggest. In contrast, patients with MS have comparatively mild attacks that are associated with a high risk for progressive disability.
Previous studies of these demyelinating disorders have examined the shape and location of lesions but not change over time. Observing these lesions’ development and resolution could provide information about disease course and influence treatment and the monitoring of disease activity, the current researchers noted.
They retrospectively identified consecutive patients with MOGAD, AQP4+ NMOSD, or MS who presented to the Mayo Clinic between January 2000 and August 2019. Data from a cohort of patients with MS in Olmsted County, Minn., were also included.
Eligible participants had experienced a first brain or myelitis attack, had undergone MRI of the brain or spinal cord within 6 weeks of the attack nadir, and had undergone a follow-up MRI 6 months after the attack.
Patients who experienced a relapse during follow-up in the same region as the initial attack were excluded. Concomitant brain and myelitis attacks were analyzed separately.
An index lesion was identified for each patient. The index lesion was defined as an acute lesion that provided an anatomic explanation for the clinical symptoms. If multiple lesions were present, the largest of them was chosen as the index lesion. MRIs were examined by neuroradiologists who were blinded to patients’ diagnoses and serology results.
Among the 156 participants, 67 had MS (76% women), 51 had AQP4+ NMOSD (80% women), and 38 had MOGAD (45% women). The median age at first attack for the groups was 37, 53, and 25 years, respectively.
In addition, 63 patients had relapsing-remitting MS, two had a single attack of progressive MS, and two had clinically isolated syndrome. No patients with NMOSD or MOGAD had developed progressive disease at final follow-up.
Participants experienced a total of 81 brain attacks and 91 myelitis attacks. Sixteen patients had experienced both a brain attack and a myelitis attack.
Symptoms corresponding to the index brain lesions were brainstem or cerebellar syndrome (56), encephalopathy or focal symptoms (12), or combinations of these (13). Among patients with an index myelitis attack, 31 had cervical involvement, 21 had thoracic involvement, and 39 had involvement of both regions.
Complete resolution
Results showed that 72% of patients with MOGAD experienced complete resolution of the brain index lesion, compared with 17% of patients with MS and 14% of patients with NMOSD (P < .001).
Similarly, 79% of the MOGAD group experienced complete resolution of the myelitis index lesion, compared with no members of the MS or NMOSD groups (P < .001 for both comparisons).
Complete resolution of all T2-abnormalities at MRI follow-up was more common in the MOGAD group than in the other two groups.
For brain attacks, complete resolution occurred in 39% of patients with MOGAD, 10% of patients with NMOSD, and 5% of patients with MS. For spinal cord attacks, complete resolution occurred in 79% of patients with MOGAD, versus none of the patients with NMOSD or MS.
Median reduction in T2 lesion area on follow-up axial brain MRI was larger in patients with MOGAD (213 mm2) than in those with NMOSD (104 mm2; P = .02) or MS (36 mm2; P < .001).
Reductions in lesion size on sagittal spine MRI follow-up were similar between the MOGAD (262 mm2) and NMOSD (309 mm2) groups; both experienced greater reductions than the MS group (23 mm2; P < .001).
Lesion prevention
Dr. Flanagan noted that the diagnosis of MOGAD is based on a test for MOG antibody, but sometimes false positive results occur. “A single follow-up MRI can be useful, showing that if all the lesions went away, you would be more confident that it would be MOGAD,” he said.
Study participants with MS experienced less lesion healing than the patients with MOGAD or NMOSD.
“We now have very effective medications in MS to prevent new lesions from occurring,” Dr. Flanagan said. The study highlights the importance of lesion prevention, “because when you do get a lesion, it does tend to stay and not recover completely,” he added.
He noted that the resolution of lesions in the study population may reflect remyelination. Future research examining whether remyelination is more efficient in MOGAD than in the other disorders could possibly lead to new approaches for MS treatment, said Dr. Flanagan.
“Maybe some of the MOGAD lesions are from edema. When we use steroids, that tends to resolve and not leave a scar. So, that’s another possibility. We’d like to better understand that,” he said.
Differences in pathology
Commenting on the findings, Bruce Cree, MD, PhD, professor of neurology, Weill Institute for Neurosciences, University of California, San Francisco, noted that the study is one of the first to systematically examine and compare MRI lesion evolution across three disease states.
“What they put their finger on are differences in the fundamental pathology of these three different diseases,” said Dr. Cree, who was not involved with the research.
The study’s cross-sectional comparison was its main strength, he noted.
“The main weakness, from my point of view, is that in these three disorders, optic nerve involvement is very common,” Dr. Cree said. “In this paper, no analysis of optic nerve lesions by MRI was performed.”
The researchers acknowledge this limitation and explain that they did not have consistent, dedicated orbital imaging for such an analysis.
Dr. Cree noted that the findings also provide a reminder that the pathogenesis of MOGAD is not yet clear.
“We know that these anti-MOG antibodies are associated with this demyelinating disorder, but whether these antibodies have a pathogenic role has yet to be clearly demonstrated,” said Dr. Cree. “What is actually going on within these lesions [is also] not fully understood.”
The finding that MOGAD lesions can resolve completely suggests that repair mechanisms are at work within the brain and spinal cord, he noted.
Being able to understand and comprehend what those mechanisms at work are and why they occur in MOGAD but not in NMOSD or MS “would be of enormous clinical advantage,” he said.
The current study also highlights the importance of incorporating imaging into clinical trials that study these rare disorders, especially serial imaging for MOGAD, Dr. Cree added.
This imaging is vital not only for developing new treatments but also for understanding the clinical impact of a given medication. “We really need rigorous imaging to be applied to these rare disorders, just as was done with MS,” Dr. Cree concluded.
The study was funded by the National Institute of Neurological Disorders and Stroke. Dr. Flanagan has received research support from MedImmune/Viela Bio. Dr. Cree is working with two of the researchers on the steering committee for the N-MOmentum trial of inebilizumab in patients with NMOSD. He has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results from a retrospective study show that complete resolution of brain lesions on MRI was more common among patients with myelin-oligodendrocyte-glycoprotein-IgG-associated disorder (MOGAD). Complete resolution occurred in 72% of the group with MOGAD, versus 17% of those with MS and 14% of those with aquaporin-4-positive neuromyelitis optica spectrum disorder (AQP4+ NMOSD).
“What we found was, with MOGAD in particular, many of the lesions resolved completely,” said co-investigator Eoin Flanagan, MBBCh, neurologist, Mayo Clinic, Rochester, Minn. “That fits with MOGAD having a fairly good prognosis and patients not developing much long-term disability with that disease,” he said.
The researchers also studied whether scarring may account for the absence of slowly progressive disability among patients with AQP4+ NMOSD and MOGAD compared with patients with MS. “The differences in scarring that we found will help physicians distinguish these three diseases more easily to aid in diagnosis. More importantly, our findings improve our understanding of the mechanisms of nerve damage in these three diseases and may suggest an important role of such scars in the development of long-term disability in MS,” Dr. Flanagan said in a statement.
The findings were published online July 14 in Neurology.
Lesion evolution
MOGAD, AQP4+ NMOSD, and MS are inflammatory demyelinating disorders that share certain manifestations. However, these disorders differ in important ways, including the severity of attacks and their clinical course.
Although patients with MOGAD and AQP4+ NMOSD generally have severe attacks that bring major disability, the clinical course of these disorders is better than initial attacks would suggest. In contrast, patients with MS have comparatively mild attacks that are associated with a high risk for progressive disability.
Previous studies of these demyelinating disorders have examined the shape and location of lesions but not change over time. Observing these lesions’ development and resolution could provide information about disease course and influence treatment and the monitoring of disease activity, the current researchers noted.
They retrospectively identified consecutive patients with MOGAD, AQP4+ NMOSD, or MS who presented to the Mayo Clinic between January 2000 and August 2019. Data from a cohort of patients with MS in Olmsted County, Minn., were also included.
Eligible participants had experienced a first brain or myelitis attack, had undergone MRI of the brain or spinal cord within 6 weeks of the attack nadir, and had undergone a follow-up MRI 6 months after the attack.
Patients who experienced a relapse during follow-up in the same region as the initial attack were excluded. Concomitant brain and myelitis attacks were analyzed separately.
An index lesion was identified for each patient. The index lesion was defined as an acute lesion that provided an anatomic explanation for the clinical symptoms. If multiple lesions were present, the largest of them was chosen as the index lesion. MRIs were examined by neuroradiologists who were blinded to patients’ diagnoses and serology results.
Among the 156 participants, 67 had MS (76% women), 51 had AQP4+ NMOSD (80% women), and 38 had MOGAD (45% women). The median age at first attack for the groups was 37, 53, and 25 years, respectively.
In addition, 63 patients had relapsing-remitting MS, two had a single attack of progressive MS, and two had clinically isolated syndrome. No patients with NMOSD or MOGAD had developed progressive disease at final follow-up.
Participants experienced a total of 81 brain attacks and 91 myelitis attacks. Sixteen patients had experienced both a brain attack and a myelitis attack.
Symptoms corresponding to the index brain lesions were brainstem or cerebellar syndrome (56), encephalopathy or focal symptoms (12), or combinations of these (13). Among patients with an index myelitis attack, 31 had cervical involvement, 21 had thoracic involvement, and 39 had involvement of both regions.
Complete resolution
Results showed that 72% of patients with MOGAD experienced complete resolution of the brain index lesion, compared with 17% of patients with MS and 14% of patients with NMOSD (P < .001).
Similarly, 79% of the MOGAD group experienced complete resolution of the myelitis index lesion, compared with no members of the MS or NMOSD groups (P < .001 for both comparisons).
Complete resolution of all T2-abnormalities at MRI follow-up was more common in the MOGAD group than in the other two groups.
For brain attacks, complete resolution occurred in 39% of patients with MOGAD, 10% of patients with NMOSD, and 5% of patients with MS. For spinal cord attacks, complete resolution occurred in 79% of patients with MOGAD, versus none of the patients with NMOSD or MS.
Median reduction in T2 lesion area on follow-up axial brain MRI was larger in patients with MOGAD (213 mm2) than in those with NMOSD (104 mm2; P = .02) or MS (36 mm2; P < .001).
Reductions in lesion size on sagittal spine MRI follow-up were similar between the MOGAD (262 mm2) and NMOSD (309 mm2) groups; both experienced greater reductions than the MS group (23 mm2; P < .001).
Lesion prevention
Dr. Flanagan noted that the diagnosis of MOGAD is based on a test for MOG antibody, but sometimes false positive results occur. “A single follow-up MRI can be useful, showing that if all the lesions went away, you would be more confident that it would be MOGAD,” he said.
Study participants with MS experienced less lesion healing than the patients with MOGAD or NMOSD.
“We now have very effective medications in MS to prevent new lesions from occurring,” Dr. Flanagan said. The study highlights the importance of lesion prevention, “because when you do get a lesion, it does tend to stay and not recover completely,” he added.
He noted that the resolution of lesions in the study population may reflect remyelination. Future research examining whether remyelination is more efficient in MOGAD than in the other disorders could possibly lead to new approaches for MS treatment, said Dr. Flanagan.
“Maybe some of the MOGAD lesions are from edema. When we use steroids, that tends to resolve and not leave a scar. So, that’s another possibility. We’d like to better understand that,” he said.
Differences in pathology
Commenting on the findings, Bruce Cree, MD, PhD, professor of neurology, Weill Institute for Neurosciences, University of California, San Francisco, noted that the study is one of the first to systematically examine and compare MRI lesion evolution across three disease states.
“What they put their finger on are differences in the fundamental pathology of these three different diseases,” said Dr. Cree, who was not involved with the research.
The study’s cross-sectional comparison was its main strength, he noted.
“The main weakness, from my point of view, is that in these three disorders, optic nerve involvement is very common,” Dr. Cree said. “In this paper, no analysis of optic nerve lesions by MRI was performed.”
The researchers acknowledge this limitation and explain that they did not have consistent, dedicated orbital imaging for such an analysis.
Dr. Cree noted that the findings also provide a reminder that the pathogenesis of MOGAD is not yet clear.
“We know that these anti-MOG antibodies are associated with this demyelinating disorder, but whether these antibodies have a pathogenic role has yet to be clearly demonstrated,” said Dr. Cree. “What is actually going on within these lesions [is also] not fully understood.”
The finding that MOGAD lesions can resolve completely suggests that repair mechanisms are at work within the brain and spinal cord, he noted.
Being able to understand and comprehend what those mechanisms at work are and why they occur in MOGAD but not in NMOSD or MS “would be of enormous clinical advantage,” he said.
The current study also highlights the importance of incorporating imaging into clinical trials that study these rare disorders, especially serial imaging for MOGAD, Dr. Cree added.
This imaging is vital not only for developing new treatments but also for understanding the clinical impact of a given medication. “We really need rigorous imaging to be applied to these rare disorders, just as was done with MS,” Dr. Cree concluded.
The study was funded by the National Institute of Neurological Disorders and Stroke. Dr. Flanagan has received research support from MedImmune/Viela Bio. Dr. Cree is working with two of the researchers on the steering committee for the N-MOmentum trial of inebilizumab in patients with NMOSD. He has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results from a retrospective study show that complete resolution of brain lesions on MRI was more common among patients with myelin-oligodendrocyte-glycoprotein-IgG-associated disorder (MOGAD). Complete resolution occurred in 72% of the group with MOGAD, versus 17% of those with MS and 14% of those with aquaporin-4-positive neuromyelitis optica spectrum disorder (AQP4+ NMOSD).
“What we found was, with MOGAD in particular, many of the lesions resolved completely,” said co-investigator Eoin Flanagan, MBBCh, neurologist, Mayo Clinic, Rochester, Minn. “That fits with MOGAD having a fairly good prognosis and patients not developing much long-term disability with that disease,” he said.
The researchers also studied whether scarring may account for the absence of slowly progressive disability among patients with AQP4+ NMOSD and MOGAD compared with patients with MS. “The differences in scarring that we found will help physicians distinguish these three diseases more easily to aid in diagnosis. More importantly, our findings improve our understanding of the mechanisms of nerve damage in these three diseases and may suggest an important role of such scars in the development of long-term disability in MS,” Dr. Flanagan said in a statement.
The findings were published online July 14 in Neurology.
Lesion evolution
MOGAD, AQP4+ NMOSD, and MS are inflammatory demyelinating disorders that share certain manifestations. However, these disorders differ in important ways, including the severity of attacks and their clinical course.
Although patients with MOGAD and AQP4+ NMOSD generally have severe attacks that bring major disability, the clinical course of these disorders is better than initial attacks would suggest. In contrast, patients with MS have comparatively mild attacks that are associated with a high risk for progressive disability.
Previous studies of these demyelinating disorders have examined the shape and location of lesions but not change over time. Observing these lesions’ development and resolution could provide information about disease course and influence treatment and the monitoring of disease activity, the current researchers noted.
They retrospectively identified consecutive patients with MOGAD, AQP4+ NMOSD, or MS who presented to the Mayo Clinic between January 2000 and August 2019. Data from a cohort of patients with MS in Olmsted County, Minn., were also included.
Eligible participants had experienced a first brain or myelitis attack, had undergone MRI of the brain or spinal cord within 6 weeks of the attack nadir, and had undergone a follow-up MRI 6 months after the attack.
Patients who experienced a relapse during follow-up in the same region as the initial attack were excluded. Concomitant brain and myelitis attacks were analyzed separately.
An index lesion was identified for each patient. The index lesion was defined as an acute lesion that provided an anatomic explanation for the clinical symptoms. If multiple lesions were present, the largest of them was chosen as the index lesion. MRIs were examined by neuroradiologists who were blinded to patients’ diagnoses and serology results.
Among the 156 participants, 67 had MS (76% women), 51 had AQP4+ NMOSD (80% women), and 38 had MOGAD (45% women). The median age at first attack for the groups was 37, 53, and 25 years, respectively.
In addition, 63 patients had relapsing-remitting MS, two had a single attack of progressive MS, and two had clinically isolated syndrome. No patients with NMOSD or MOGAD had developed progressive disease at final follow-up.
Participants experienced a total of 81 brain attacks and 91 myelitis attacks. Sixteen patients had experienced both a brain attack and a myelitis attack.
Symptoms corresponding to the index brain lesions were brainstem or cerebellar syndrome (56), encephalopathy or focal symptoms (12), or combinations of these (13). Among patients with an index myelitis attack, 31 had cervical involvement, 21 had thoracic involvement, and 39 had involvement of both regions.
Complete resolution
Results showed that 72% of patients with MOGAD experienced complete resolution of the brain index lesion, compared with 17% of patients with MS and 14% of patients with NMOSD (P < .001).
Similarly, 79% of the MOGAD group experienced complete resolution of the myelitis index lesion, compared with no members of the MS or NMOSD groups (P < .001 for both comparisons).
Complete resolution of all T2-abnormalities at MRI follow-up was more common in the MOGAD group than in the other two groups.
For brain attacks, complete resolution occurred in 39% of patients with MOGAD, 10% of patients with NMOSD, and 5% of patients with MS. For spinal cord attacks, complete resolution occurred in 79% of patients with MOGAD, versus none of the patients with NMOSD or MS.
Median reduction in T2 lesion area on follow-up axial brain MRI was larger in patients with MOGAD (213 mm2) than in those with NMOSD (104 mm2; P = .02) or MS (36 mm2; P < .001).
Reductions in lesion size on sagittal spine MRI follow-up were similar between the MOGAD (262 mm2) and NMOSD (309 mm2) groups; both experienced greater reductions than the MS group (23 mm2; P < .001).
Lesion prevention
Dr. Flanagan noted that the diagnosis of MOGAD is based on a test for MOG antibody, but sometimes false positive results occur. “A single follow-up MRI can be useful, showing that if all the lesions went away, you would be more confident that it would be MOGAD,” he said.
Study participants with MS experienced less lesion healing than the patients with MOGAD or NMOSD.
“We now have very effective medications in MS to prevent new lesions from occurring,” Dr. Flanagan said. The study highlights the importance of lesion prevention, “because when you do get a lesion, it does tend to stay and not recover completely,” he added.
He noted that the resolution of lesions in the study population may reflect remyelination. Future research examining whether remyelination is more efficient in MOGAD than in the other disorders could possibly lead to new approaches for MS treatment, said Dr. Flanagan.
“Maybe some of the MOGAD lesions are from edema. When we use steroids, that tends to resolve and not leave a scar. So, that’s another possibility. We’d like to better understand that,” he said.
Differences in pathology
Commenting on the findings, Bruce Cree, MD, PhD, professor of neurology, Weill Institute for Neurosciences, University of California, San Francisco, noted that the study is one of the first to systematically examine and compare MRI lesion evolution across three disease states.
“What they put their finger on are differences in the fundamental pathology of these three different diseases,” said Dr. Cree, who was not involved with the research.
The study’s cross-sectional comparison was its main strength, he noted.
“The main weakness, from my point of view, is that in these three disorders, optic nerve involvement is very common,” Dr. Cree said. “In this paper, no analysis of optic nerve lesions by MRI was performed.”
The researchers acknowledge this limitation and explain that they did not have consistent, dedicated orbital imaging for such an analysis.
Dr. Cree noted that the findings also provide a reminder that the pathogenesis of MOGAD is not yet clear.
“We know that these anti-MOG antibodies are associated with this demyelinating disorder, but whether these antibodies have a pathogenic role has yet to be clearly demonstrated,” said Dr. Cree. “What is actually going on within these lesions [is also] not fully understood.”
The finding that MOGAD lesions can resolve completely suggests that repair mechanisms are at work within the brain and spinal cord, he noted.
Being able to understand and comprehend what those mechanisms at work are and why they occur in MOGAD but not in NMOSD or MS “would be of enormous clinical advantage,” he said.
The current study also highlights the importance of incorporating imaging into clinical trials that study these rare disorders, especially serial imaging for MOGAD, Dr. Cree added.
This imaging is vital not only for developing new treatments but also for understanding the clinical impact of a given medication. “We really need rigorous imaging to be applied to these rare disorders, just as was done with MS,” Dr. Cree concluded.
The study was funded by the National Institute of Neurological Disorders and Stroke. Dr. Flanagan has received research support from MedImmune/Viela Bio. Dr. Cree is working with two of the researchers on the steering committee for the N-MOmentum trial of inebilizumab in patients with NMOSD. He has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From Neurology
Guidance on additional COVID-19 vaccine dose for MS patients
Patients aged 12 years and older with multiple sclerosis (MS) who are fully immunized against COVID-19 with either the Pfizer-BioNTech or Moderna mRNA vaccine may be eligible to receive an additional dose now, the National Multiple Sclerosis Society has announced.
New guidance, which is “based on available data from studies and expert consensus opinion” by a panel of MS neurologists and experts, was published Aug. 19 on the organization’s website.
The Food and Drug Administration has authorized an additional dose of the coronavirus vaccine for patients who are expected to not have a normal or adequate immune response to the first two doses. Patients with MS who use certain treatments have a reduced or absent antibody response to the vaccine, according to recent data.
“We want people living with MS to be aware of this additional dose and discuss when they need an additional dose or booster dose with their health care provider,” Julie Fiol, RN, MSW, associate vice president of health care access, National MS Society, said in an interview.
Those who may benefit from an additional dose include patients with MS who use sphingosine 1-phosphate receptor modulators, anti-CD20 monoclonal antibodies, or alemtuzumab (Lemtrada), the National MS Society noted. These particular disease modifying therapies (DMTs) have a stronger effect on the immune system than do other treatments.
Protecting ‘the most vulnerable’
Sphingosine 1-phosphate receptor modulators include fingolimod (Gilenya), siponimod (Mayzent), ozanimod (Zeposia), and ponesimod (Ponvory).
Anti-CD20 monoclonal antibodies include ocrelizumab (Ocrevus), ofatumumab (Kesimpta), rituximab (Rituxan), and corresponding biosimilars.
Current data do not support an additional dose for immunocompromised patients who received the Johnson & Johnson vaccine. The FDA and the Centers for Disease Control and Prevention are developing recommendations for these patients, and the National MS Society will update its guidance as needed, the organization noted in its statement.
“Like other medical decisions, the decision to get an additional dose is best made in partnership with your health care provider,” said Ms. Fiol. “Talk to your MS health care provider to determine what is best for you.”
MS itself does not compromise the immune system, but some MS therapies alter the immune system and reduce the body’s response to vaccination. Patients with MS who use B cell-depleting therapies have a better antibody response when they receive the vaccine 3 months or more after the last dose of MS therapy, according to the National MS Society.
Data suggest that patients with MS are not more susceptible to COVID-19 infection, severe illness, or death than are patients without MS. However, certain groups of patients with MS, such as those who receive B cell-depleting treatments, are more susceptible to having a severe case of COVID-19.
That said, “everyone will need a booster at some point. Those who take DMTs that have greater impact on the immune system are the most urgent need now,” the organization noted.
“Vaccination against COVID-19 is critical for public safety and, especially, the safety of the most vulnerable among us,” said Ms. Fiol. “We encourage everyone with MS get vaccinated.”
A version of this article first appeared on Medscape.com.
Patients aged 12 years and older with multiple sclerosis (MS) who are fully immunized against COVID-19 with either the Pfizer-BioNTech or Moderna mRNA vaccine may be eligible to receive an additional dose now, the National Multiple Sclerosis Society has announced.
New guidance, which is “based on available data from studies and expert consensus opinion” by a panel of MS neurologists and experts, was published Aug. 19 on the organization’s website.
The Food and Drug Administration has authorized an additional dose of the coronavirus vaccine for patients who are expected to not have a normal or adequate immune response to the first two doses. Patients with MS who use certain treatments have a reduced or absent antibody response to the vaccine, according to recent data.
“We want people living with MS to be aware of this additional dose and discuss when they need an additional dose or booster dose with their health care provider,” Julie Fiol, RN, MSW, associate vice president of health care access, National MS Society, said in an interview.
Those who may benefit from an additional dose include patients with MS who use sphingosine 1-phosphate receptor modulators, anti-CD20 monoclonal antibodies, or alemtuzumab (Lemtrada), the National MS Society noted. These particular disease modifying therapies (DMTs) have a stronger effect on the immune system than do other treatments.
Protecting ‘the most vulnerable’
Sphingosine 1-phosphate receptor modulators include fingolimod (Gilenya), siponimod (Mayzent), ozanimod (Zeposia), and ponesimod (Ponvory).
Anti-CD20 monoclonal antibodies include ocrelizumab (Ocrevus), ofatumumab (Kesimpta), rituximab (Rituxan), and corresponding biosimilars.
Current data do not support an additional dose for immunocompromised patients who received the Johnson & Johnson vaccine. The FDA and the Centers for Disease Control and Prevention are developing recommendations for these patients, and the National MS Society will update its guidance as needed, the organization noted in its statement.
“Like other medical decisions, the decision to get an additional dose is best made in partnership with your health care provider,” said Ms. Fiol. “Talk to your MS health care provider to determine what is best for you.”
MS itself does not compromise the immune system, but some MS therapies alter the immune system and reduce the body’s response to vaccination. Patients with MS who use B cell-depleting therapies have a better antibody response when they receive the vaccine 3 months or more after the last dose of MS therapy, according to the National MS Society.
Data suggest that patients with MS are not more susceptible to COVID-19 infection, severe illness, or death than are patients without MS. However, certain groups of patients with MS, such as those who receive B cell-depleting treatments, are more susceptible to having a severe case of COVID-19.
That said, “everyone will need a booster at some point. Those who take DMTs that have greater impact on the immune system are the most urgent need now,” the organization noted.
“Vaccination against COVID-19 is critical for public safety and, especially, the safety of the most vulnerable among us,” said Ms. Fiol. “We encourage everyone with MS get vaccinated.”
A version of this article first appeared on Medscape.com.
Patients aged 12 years and older with multiple sclerosis (MS) who are fully immunized against COVID-19 with either the Pfizer-BioNTech or Moderna mRNA vaccine may be eligible to receive an additional dose now, the National Multiple Sclerosis Society has announced.
New guidance, which is “based on available data from studies and expert consensus opinion” by a panel of MS neurologists and experts, was published Aug. 19 on the organization’s website.
The Food and Drug Administration has authorized an additional dose of the coronavirus vaccine for patients who are expected to not have a normal or adequate immune response to the first two doses. Patients with MS who use certain treatments have a reduced or absent antibody response to the vaccine, according to recent data.
“We want people living with MS to be aware of this additional dose and discuss when they need an additional dose or booster dose with their health care provider,” Julie Fiol, RN, MSW, associate vice president of health care access, National MS Society, said in an interview.
Those who may benefit from an additional dose include patients with MS who use sphingosine 1-phosphate receptor modulators, anti-CD20 monoclonal antibodies, or alemtuzumab (Lemtrada), the National MS Society noted. These particular disease modifying therapies (DMTs) have a stronger effect on the immune system than do other treatments.
Protecting ‘the most vulnerable’
Sphingosine 1-phosphate receptor modulators include fingolimod (Gilenya), siponimod (Mayzent), ozanimod (Zeposia), and ponesimod (Ponvory).
Anti-CD20 monoclonal antibodies include ocrelizumab (Ocrevus), ofatumumab (Kesimpta), rituximab (Rituxan), and corresponding biosimilars.
Current data do not support an additional dose for immunocompromised patients who received the Johnson & Johnson vaccine. The FDA and the Centers for Disease Control and Prevention are developing recommendations for these patients, and the National MS Society will update its guidance as needed, the organization noted in its statement.
“Like other medical decisions, the decision to get an additional dose is best made in partnership with your health care provider,” said Ms. Fiol. “Talk to your MS health care provider to determine what is best for you.”
MS itself does not compromise the immune system, but some MS therapies alter the immune system and reduce the body’s response to vaccination. Patients with MS who use B cell-depleting therapies have a better antibody response when they receive the vaccine 3 months or more after the last dose of MS therapy, according to the National MS Society.
Data suggest that patients with MS are not more susceptible to COVID-19 infection, severe illness, or death than are patients without MS. However, certain groups of patients with MS, such as those who receive B cell-depleting treatments, are more susceptible to having a severe case of COVID-19.
That said, “everyone will need a booster at some point. Those who take DMTs that have greater impact on the immune system are the most urgent need now,” the organization noted.
“Vaccination against COVID-19 is critical for public safety and, especially, the safety of the most vulnerable among us,” said Ms. Fiol. “We encourage everyone with MS get vaccinated.”
A version of this article first appeared on Medscape.com.
MS CONFERENCE NEWS
- Common MS treatment wears off more quickly in Black patients
- A less expensive, more convenient treatment option for MS?
- Investigational drug reduces brain lesions in highly active MS
- Evobrutinib may lower nerve damage biomarker levels
- Disease progression and therapy response vary in MS by ethnicity
- Some MS treatments may heighten COVID risk
- Natalizumab postinfusion reactions
- Common MS treatment wears off more quickly in Black patients
- A less expensive, more convenient treatment option for MS?
- Investigational drug reduces brain lesions in highly active MS
- Evobrutinib may lower nerve damage biomarker levels
- Disease progression and therapy response vary in MS by ethnicity
- Some MS treatments may heighten COVID risk
- Natalizumab postinfusion reactions
- Common MS treatment wears off more quickly in Black patients
- A less expensive, more convenient treatment option for MS?
- Investigational drug reduces brain lesions in highly active MS
- Evobrutinib may lower nerve damage biomarker levels
- Disease progression and therapy response vary in MS by ethnicity
- Some MS treatments may heighten COVID risk
- Natalizumab postinfusion reactions