User login
Restless legs syndrome occurs often in X-linked adrenoleukodystrophy
Patients with X-linked adrenoleukodystrophy (ALD), a neurodegenerative disease, often experience gait and balance problems, as well as leg discomfort, sleep disturbances, and pain, wrote John W. Winkelman, MD, of Massachusetts General Hospital, Boston, and colleagues. Restless legs syndrome (RLS) has been associated with neurological conditions including Parkinson’s disease, but the prevalence of RLS in ALD patients has not been examined, they said.
In a pilot study published in Sleep Medicine, the researchers identified 21 women and 11 men with ALD who were treated at a single center. The median age of the patients was 45.9 years. Twenty-seven patients had symptoms of myelopathy, with a median age of onset of 34 years.
The researchers assessed RLS severity using questionnaires and the Hopkins Telephone Diagnostic Interview (HTDI), a validated RLS assessment tool. They also reviewed patients’ charts for data on neurological examinations, functional gait measures, and laboratory assessments. Functional gait assessments included the 25-Foot Walk test (25-FW), the Timed Up and Go test (TUG), and Six Minute Walk test (6MW).
Thirteen patients (10 women and 3 men) met criteria for RLS based on the HTDI. The median age of RLS onset was 35 years. Six RLS patients (46.2%) reported using medication to relieve symptoms, and eight RLS patients had a history of antidepressant use.
In addition, six patients with RLS reported a history of anemia or iron deficiency. Ferritin levels were available for 14 patients: 8 women with RLS and 4 women and 2 men without RLS; the mean ferritin levels were 74.0 mcg/L in RLS patients and 99.5 mcg/L in those without RLS.
Of the seven ALD patients with brain lesions, all were men, only two were diagnosed with RLS, and all seven cases were mild, the researchers noted.
Overall, patients with RLS had more neurological signs and symptoms than those without RLS; the most significant were pain and gait difficulty. However, patients with RLS also were more likely than were those without RLS to report spasticity, muscle weakness, impaired coordination, hyperreflexia, impaired sensation, and paraesthesia, as well as bladder, bowel, and erectile dysfunction.
The 40.6% prevalence of RLS in patients with ALD is notably higher than that of the general population, in which the prevalence of RLS is 5%-10%, the researchers wrote in their discussion.
“Consistent with patterns observed in the general population, risk factors for RLS in this cohort of adults with ALD included female gender, increased age, lower iron indices, and use of serotonergic antidepressants,” they said.
The study findings were limited by several factors including the small size and the possible contribution of antidepressant use to the high rate of RLS, the researchers noted.
“Awareness of RLS in patients with ALD would allow for its effective treatment, which may improve the functional impairments as well as quality of life, mood, and anxiety issues in those with ALD,” they concluded.
The study received no outside funding.
Dr. Winkelman disclosed ties with Advance Medical, Avadel, Disc Medicine, Eisai, Emalex, Idorsia, Noctrix, UpToDate, and Merck Pharmaceuticals, as well as research support from the National Institute on Drug Abuse and the Baszucki Brain Research Foundation. The study also was supported by grants from the National Institute of Neurological Disorders and Stroke, the European Leukodystrophy Association, the Arrivederci Foundation, the Leblang Foundation, and the Hammer Family Fund Journal Preproof for ALD Research and Therapies for Women.
Patients with X-linked adrenoleukodystrophy (ALD), a neurodegenerative disease, often experience gait and balance problems, as well as leg discomfort, sleep disturbances, and pain, wrote John W. Winkelman, MD, of Massachusetts General Hospital, Boston, and colleagues. Restless legs syndrome (RLS) has been associated with neurological conditions including Parkinson’s disease, but the prevalence of RLS in ALD patients has not been examined, they said.
In a pilot study published in Sleep Medicine, the researchers identified 21 women and 11 men with ALD who were treated at a single center. The median age of the patients was 45.9 years. Twenty-seven patients had symptoms of myelopathy, with a median age of onset of 34 years.
The researchers assessed RLS severity using questionnaires and the Hopkins Telephone Diagnostic Interview (HTDI), a validated RLS assessment tool. They also reviewed patients’ charts for data on neurological examinations, functional gait measures, and laboratory assessments. Functional gait assessments included the 25-Foot Walk test (25-FW), the Timed Up and Go test (TUG), and Six Minute Walk test (6MW).
Thirteen patients (10 women and 3 men) met criteria for RLS based on the HTDI. The median age of RLS onset was 35 years. Six RLS patients (46.2%) reported using medication to relieve symptoms, and eight RLS patients had a history of antidepressant use.
In addition, six patients with RLS reported a history of anemia or iron deficiency. Ferritin levels were available for 14 patients: 8 women with RLS and 4 women and 2 men without RLS; the mean ferritin levels were 74.0 mcg/L in RLS patients and 99.5 mcg/L in those without RLS.
Of the seven ALD patients with brain lesions, all were men, only two were diagnosed with RLS, and all seven cases were mild, the researchers noted.
Overall, patients with RLS had more neurological signs and symptoms than those without RLS; the most significant were pain and gait difficulty. However, patients with RLS also were more likely than were those without RLS to report spasticity, muscle weakness, impaired coordination, hyperreflexia, impaired sensation, and paraesthesia, as well as bladder, bowel, and erectile dysfunction.
The 40.6% prevalence of RLS in patients with ALD is notably higher than that of the general population, in which the prevalence of RLS is 5%-10%, the researchers wrote in their discussion.
“Consistent with patterns observed in the general population, risk factors for RLS in this cohort of adults with ALD included female gender, increased age, lower iron indices, and use of serotonergic antidepressants,” they said.
The study findings were limited by several factors including the small size and the possible contribution of antidepressant use to the high rate of RLS, the researchers noted.
“Awareness of RLS in patients with ALD would allow for its effective treatment, which may improve the functional impairments as well as quality of life, mood, and anxiety issues in those with ALD,” they concluded.
The study received no outside funding.
Dr. Winkelman disclosed ties with Advance Medical, Avadel, Disc Medicine, Eisai, Emalex, Idorsia, Noctrix, UpToDate, and Merck Pharmaceuticals, as well as research support from the National Institute on Drug Abuse and the Baszucki Brain Research Foundation. The study also was supported by grants from the National Institute of Neurological Disorders and Stroke, the European Leukodystrophy Association, the Arrivederci Foundation, the Leblang Foundation, and the Hammer Family Fund Journal Preproof for ALD Research and Therapies for Women.
Patients with X-linked adrenoleukodystrophy (ALD), a neurodegenerative disease, often experience gait and balance problems, as well as leg discomfort, sleep disturbances, and pain, wrote John W. Winkelman, MD, of Massachusetts General Hospital, Boston, and colleagues. Restless legs syndrome (RLS) has been associated with neurological conditions including Parkinson’s disease, but the prevalence of RLS in ALD patients has not been examined, they said.
In a pilot study published in Sleep Medicine, the researchers identified 21 women and 11 men with ALD who were treated at a single center. The median age of the patients was 45.9 years. Twenty-seven patients had symptoms of myelopathy, with a median age of onset of 34 years.
The researchers assessed RLS severity using questionnaires and the Hopkins Telephone Diagnostic Interview (HTDI), a validated RLS assessment tool. They also reviewed patients’ charts for data on neurological examinations, functional gait measures, and laboratory assessments. Functional gait assessments included the 25-Foot Walk test (25-FW), the Timed Up and Go test (TUG), and Six Minute Walk test (6MW).
Thirteen patients (10 women and 3 men) met criteria for RLS based on the HTDI. The median age of RLS onset was 35 years. Six RLS patients (46.2%) reported using medication to relieve symptoms, and eight RLS patients had a history of antidepressant use.
In addition, six patients with RLS reported a history of anemia or iron deficiency. Ferritin levels were available for 14 patients: 8 women with RLS and 4 women and 2 men without RLS; the mean ferritin levels were 74.0 mcg/L in RLS patients and 99.5 mcg/L in those without RLS.
Of the seven ALD patients with brain lesions, all were men, only two were diagnosed with RLS, and all seven cases were mild, the researchers noted.
Overall, patients with RLS had more neurological signs and symptoms than those without RLS; the most significant were pain and gait difficulty. However, patients with RLS also were more likely than were those without RLS to report spasticity, muscle weakness, impaired coordination, hyperreflexia, impaired sensation, and paraesthesia, as well as bladder, bowel, and erectile dysfunction.
The 40.6% prevalence of RLS in patients with ALD is notably higher than that of the general population, in which the prevalence of RLS is 5%-10%, the researchers wrote in their discussion.
“Consistent with patterns observed in the general population, risk factors for RLS in this cohort of adults with ALD included female gender, increased age, lower iron indices, and use of serotonergic antidepressants,” they said.
The study findings were limited by several factors including the small size and the possible contribution of antidepressant use to the high rate of RLS, the researchers noted.
“Awareness of RLS in patients with ALD would allow for its effective treatment, which may improve the functional impairments as well as quality of life, mood, and anxiety issues in those with ALD,” they concluded.
The study received no outside funding.
Dr. Winkelman disclosed ties with Advance Medical, Avadel, Disc Medicine, Eisai, Emalex, Idorsia, Noctrix, UpToDate, and Merck Pharmaceuticals, as well as research support from the National Institute on Drug Abuse and the Baszucki Brain Research Foundation. The study also was supported by grants from the National Institute of Neurological Disorders and Stroke, the European Leukodystrophy Association, the Arrivederci Foundation, the Leblang Foundation, and the Hammer Family Fund Journal Preproof for ALD Research and Therapies for Women.
FROM SLEEP MEDICINE
Abnormal growth of the amygdala in infants tied to autism
A new study suggests that overgrowth of the amygdala in infants during the first 6-12 months of life is tied to a later diagnosis of autism spectrum disorder (ASD).
“The faster the amygdala grew in infancy, the more social difficulties the child showed when diagnosed with autism a year later,” first author Mark Shen, PhD, assistant professor of psychiatry and neuroscience, University of North Carolina, Chapel Hill, told this news organization.
The study was published online in the American Journal of Psychiatry.
Unique to autism
The amygdala plays a key role in processing memory, emotional responses, and decisionmaking.
It’s long been known that the amygdala is abnormally large in school-aged children with ASD, but until now, it was not known precisely when aberrant amygdala growth happens, what the clinical consequences may be, and whether amygdala overgrowth is unique to autism.
To investigate, Dr. Shen and colleagues evaluated 1,099 longitudinal MRI scans obtained during natural sleep at 6, 12, and 24 months of age in 408 infants in the Infant Brain Imaging Study (IBIS) Network.
The cohort included 58 infants at high likelihood of developing ASD who were later diagnosed with the disorder, 212 infants at high likelihood of ASD who did not develop ASD, 109 typically-developing control infants, and 29 infants with fragile X syndrome.
At 6 months, infants who developed ASD had typically sized amygdala volumes but showed significantly faster amygdala growth between 6 and 24 months, such that by 12 months the ASD group had significantly larger amygdala volume (Cohen’s d = 0.56), compared with all other groups.
Amygdala growth rate between 6 and 12 months was significantly associated with greater social deficits at 24 months when the children were diagnosed with ASD.
“We found that the amygdala grows too rapidly between 6 and 12 months of age, during a presymptomatic period in autism, prior to when the diagnostic symptoms of autism (social difficulties and repetitive behaviors) are evident and lead to the later diagnosis of autism,” Dr. Shen said in an interview.
This brain growth pattern appears to be unique to autism, as babies with the genetic disorder fragile X syndrome – another neurodevelopmental condition – showed a markedly different brain growth pattern: no differences in amygdala growth but enlargement of a different brain structure, the caudate, which was linked to increased repetitive behaviors, the investigators found.
Earlier intervention
Prior research has shown that children who are later diagnosed with ASD often display problems in infancy with how they attend to visual stimuli in their surroundings.
These early problems with processing visual and sensory information may put increased stress on the amygdala, potentially leading to amygdala hyperactivity, deficits in pruning dendritic connections, and overgrowth, Dr. Shen and colleagues hypothesize.
Amygdala overgrowth has also been linked to chronic stress in studies of other psychiatric conditions, such as depression and anxiety, and may provide a clue to understanding this observation in infants who later develop autism.
“This research suggests that an optimal time to begin supports for children who are at the highest likelihood of developing autism may be during the first year of life: to improve early precursors to social development, such as sensory processing, in babies even before social difficulties arise,” Dr. Shen said.
Cyrus A. Raji, MD, PhD, assistant professor of radiology and neurology, Washington University, St. Louis, said, “What makes this study important is the finding of abnormally increased amygdala growth rate in autism using a longitudinal design that focuses on earlier development.”
“While we are typically used to understanding brain structure as abnormally decreasing over time in certain disorders like Alzheimer’s disease, this study challenges us to understand that too much brain volume growth can also be abnormal in specific conditions,” Dr. Raji added.
This research was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institute of Environmental Health Sciences, and National Institute of Mental Health, along with Autism Speaks and the Simons Foundation. Dr. Shen and Dr. Raji have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study suggests that overgrowth of the amygdala in infants during the first 6-12 months of life is tied to a later diagnosis of autism spectrum disorder (ASD).
“The faster the amygdala grew in infancy, the more social difficulties the child showed when diagnosed with autism a year later,” first author Mark Shen, PhD, assistant professor of psychiatry and neuroscience, University of North Carolina, Chapel Hill, told this news organization.
The study was published online in the American Journal of Psychiatry.
Unique to autism
The amygdala plays a key role in processing memory, emotional responses, and decisionmaking.
It’s long been known that the amygdala is abnormally large in school-aged children with ASD, but until now, it was not known precisely when aberrant amygdala growth happens, what the clinical consequences may be, and whether amygdala overgrowth is unique to autism.
To investigate, Dr. Shen and colleagues evaluated 1,099 longitudinal MRI scans obtained during natural sleep at 6, 12, and 24 months of age in 408 infants in the Infant Brain Imaging Study (IBIS) Network.
The cohort included 58 infants at high likelihood of developing ASD who were later diagnosed with the disorder, 212 infants at high likelihood of ASD who did not develop ASD, 109 typically-developing control infants, and 29 infants with fragile X syndrome.
At 6 months, infants who developed ASD had typically sized amygdala volumes but showed significantly faster amygdala growth between 6 and 24 months, such that by 12 months the ASD group had significantly larger amygdala volume (Cohen’s d = 0.56), compared with all other groups.
Amygdala growth rate between 6 and 12 months was significantly associated with greater social deficits at 24 months when the children were diagnosed with ASD.
“We found that the amygdala grows too rapidly between 6 and 12 months of age, during a presymptomatic period in autism, prior to when the diagnostic symptoms of autism (social difficulties and repetitive behaviors) are evident and lead to the later diagnosis of autism,” Dr. Shen said in an interview.
This brain growth pattern appears to be unique to autism, as babies with the genetic disorder fragile X syndrome – another neurodevelopmental condition – showed a markedly different brain growth pattern: no differences in amygdala growth but enlargement of a different brain structure, the caudate, which was linked to increased repetitive behaviors, the investigators found.
Earlier intervention
Prior research has shown that children who are later diagnosed with ASD often display problems in infancy with how they attend to visual stimuli in their surroundings.
These early problems with processing visual and sensory information may put increased stress on the amygdala, potentially leading to amygdala hyperactivity, deficits in pruning dendritic connections, and overgrowth, Dr. Shen and colleagues hypothesize.
Amygdala overgrowth has also been linked to chronic stress in studies of other psychiatric conditions, such as depression and anxiety, and may provide a clue to understanding this observation in infants who later develop autism.
“This research suggests that an optimal time to begin supports for children who are at the highest likelihood of developing autism may be during the first year of life: to improve early precursors to social development, such as sensory processing, in babies even before social difficulties arise,” Dr. Shen said.
Cyrus A. Raji, MD, PhD, assistant professor of radiology and neurology, Washington University, St. Louis, said, “What makes this study important is the finding of abnormally increased amygdala growth rate in autism using a longitudinal design that focuses on earlier development.”
“While we are typically used to understanding brain structure as abnormally decreasing over time in certain disorders like Alzheimer’s disease, this study challenges us to understand that too much brain volume growth can also be abnormal in specific conditions,” Dr. Raji added.
This research was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institute of Environmental Health Sciences, and National Institute of Mental Health, along with Autism Speaks and the Simons Foundation. Dr. Shen and Dr. Raji have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study suggests that overgrowth of the amygdala in infants during the first 6-12 months of life is tied to a later diagnosis of autism spectrum disorder (ASD).
“The faster the amygdala grew in infancy, the more social difficulties the child showed when diagnosed with autism a year later,” first author Mark Shen, PhD, assistant professor of psychiatry and neuroscience, University of North Carolina, Chapel Hill, told this news organization.
The study was published online in the American Journal of Psychiatry.
Unique to autism
The amygdala plays a key role in processing memory, emotional responses, and decisionmaking.
It’s long been known that the amygdala is abnormally large in school-aged children with ASD, but until now, it was not known precisely when aberrant amygdala growth happens, what the clinical consequences may be, and whether amygdala overgrowth is unique to autism.
To investigate, Dr. Shen and colleagues evaluated 1,099 longitudinal MRI scans obtained during natural sleep at 6, 12, and 24 months of age in 408 infants in the Infant Brain Imaging Study (IBIS) Network.
The cohort included 58 infants at high likelihood of developing ASD who were later diagnosed with the disorder, 212 infants at high likelihood of ASD who did not develop ASD, 109 typically-developing control infants, and 29 infants with fragile X syndrome.
At 6 months, infants who developed ASD had typically sized amygdala volumes but showed significantly faster amygdala growth between 6 and 24 months, such that by 12 months the ASD group had significantly larger amygdala volume (Cohen’s d = 0.56), compared with all other groups.
Amygdala growth rate between 6 and 12 months was significantly associated with greater social deficits at 24 months when the children were diagnosed with ASD.
“We found that the amygdala grows too rapidly between 6 and 12 months of age, during a presymptomatic period in autism, prior to when the diagnostic symptoms of autism (social difficulties and repetitive behaviors) are evident and lead to the later diagnosis of autism,” Dr. Shen said in an interview.
This brain growth pattern appears to be unique to autism, as babies with the genetic disorder fragile X syndrome – another neurodevelopmental condition – showed a markedly different brain growth pattern: no differences in amygdala growth but enlargement of a different brain structure, the caudate, which was linked to increased repetitive behaviors, the investigators found.
Earlier intervention
Prior research has shown that children who are later diagnosed with ASD often display problems in infancy with how they attend to visual stimuli in their surroundings.
These early problems with processing visual and sensory information may put increased stress on the amygdala, potentially leading to amygdala hyperactivity, deficits in pruning dendritic connections, and overgrowth, Dr. Shen and colleagues hypothesize.
Amygdala overgrowth has also been linked to chronic stress in studies of other psychiatric conditions, such as depression and anxiety, and may provide a clue to understanding this observation in infants who later develop autism.
“This research suggests that an optimal time to begin supports for children who are at the highest likelihood of developing autism may be during the first year of life: to improve early precursors to social development, such as sensory processing, in babies even before social difficulties arise,” Dr. Shen said.
Cyrus A. Raji, MD, PhD, assistant professor of radiology and neurology, Washington University, St. Louis, said, “What makes this study important is the finding of abnormally increased amygdala growth rate in autism using a longitudinal design that focuses on earlier development.”
“While we are typically used to understanding brain structure as abnormally decreasing over time in certain disorders like Alzheimer’s disease, this study challenges us to understand that too much brain volume growth can also be abnormal in specific conditions,” Dr. Raji added.
This research was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institute of Environmental Health Sciences, and National Institute of Mental Health, along with Autism Speaks and the Simons Foundation. Dr. Shen and Dr. Raji have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Physical fitness tied to lower risk of Alzheimer’s disease
, new findings suggest. “One exciting finding of this study is that as people’s fitness improved, their risk of Alzheimer’s disease decreased – it was not an all-or-nothing proposition,” study investigator Edward Zamrini, MD, of the Washington DC VA Medical Center, said in a news release.
The findings suggest that people can work toward making incremental changes and improvements in their physical fitness, which may help decrease their risk of dementia, Dr. Zamrini added.
The findings were presented at the 2022 annual meeting of the American Academy of Neurology.
Effective prevention strategy
Using the Veterans Health Administration database, Dr. Zamrini and colleagues identified 649,605 veterans (mean age, 61 years) free of Alzheimer’s disease and related disorders (ADRD) when they completed standardized exercise treadmill tests between 2000 and 2017.
They divided participants into five age-specific fitness groups, from least fit to most fit, based on peak metabolic equivalents (METs) achieved during the treadmill test: lowest-fit (METs, ±3.8), low-fit (METs, ±5.8), moderate-fit (METs, ±7.5), fit (METs, ±9.2), and highest-fit (METs, ±11.7).
In unadjusted analysis, veterans with the lowest cardiorespiratory fitness developed ADRD at a rate of 9.5 cases per 1,000 person-years, compared with a rate of 6.4 cases per 1,000 person-years for the most fit group (P < .001).
After adjusting for factors that could affect risk of ADRD, compared with the lowest-fit group, the highest-fit and fit groups were 33% and 26% less likely to develop ADRD, respectively, while the moderate-fit and low-fit groups were 20% and 13% less likely to develop the disease, respectively.
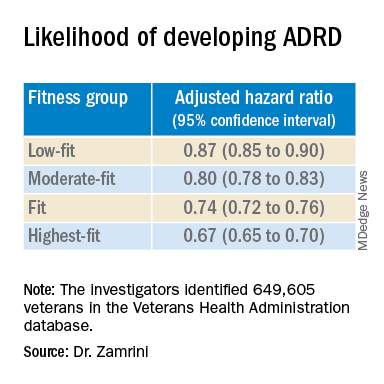
The findings suggest that the association between cardiorespiratory fitness and ADRD risk is “inverse, independent, and graded,” the researchers said in their conference abstract.
“The idea that you can reduce your risk for Alzheimer’s disease by simply increasing your activity is very promising, especially since there are no adequate treatments to prevent or stop the progression of the disease,” Dr. Zamrini added in the news release.
“We hope to develop a simple scale that can be individualized so people can see the benefits that even incremental improvements in fitness can deliver,” he said.
The next vital sign?
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Boston, noted that “for decades and with increasing body of support from studies like this, we have known that preventing dementia is based on healthy behaviors for the brain including a proper diet (NASH and/or Mediterranean), exercise regimen (aerobic/cardio more than anaerobic/weight-lifting), sleep hygiene, and social and intellectual engagements.”
“Frankly, what’s good for the body is good for the brain,” said Dr. Lakhan.
“It should be noted that the measure studied here is cardiorespiratory fitness, which has been associated with heart disease and resulting death, death from any cause, and now brain health,” Dr. Lakhan said.
“This powerful predictor may in fact be the next vital sign, after your heart rate and blood pressure, from which your primary care provider can make a personalized treatment plan,” he added.
“Accelerating this process, the ability to measure cardiorespiratory fitness traditionally from huge stationary machines down to wearables like a watch or ring, or even your iPhone or Android, is just on the horizon,” Dr. Lakhan said.
“Instead of tracking just your weight, shape, and BMI, personal fitness may be tailored to optimizing this indicator and further empowering individuals to take charge of their health,” he said.
The study was supported by the National Institute on Aging, the National Institutes of Health, the U.S. Department of Veterans Affairs, the Washington DC VA Medical Center, and George Washington University. Dr. Zamrini and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new findings suggest. “One exciting finding of this study is that as people’s fitness improved, their risk of Alzheimer’s disease decreased – it was not an all-or-nothing proposition,” study investigator Edward Zamrini, MD, of the Washington DC VA Medical Center, said in a news release.
The findings suggest that people can work toward making incremental changes and improvements in their physical fitness, which may help decrease their risk of dementia, Dr. Zamrini added.
The findings were presented at the 2022 annual meeting of the American Academy of Neurology.
Effective prevention strategy
Using the Veterans Health Administration database, Dr. Zamrini and colleagues identified 649,605 veterans (mean age, 61 years) free of Alzheimer’s disease and related disorders (ADRD) when they completed standardized exercise treadmill tests between 2000 and 2017.
They divided participants into five age-specific fitness groups, from least fit to most fit, based on peak metabolic equivalents (METs) achieved during the treadmill test: lowest-fit (METs, ±3.8), low-fit (METs, ±5.8), moderate-fit (METs, ±7.5), fit (METs, ±9.2), and highest-fit (METs, ±11.7).
In unadjusted analysis, veterans with the lowest cardiorespiratory fitness developed ADRD at a rate of 9.5 cases per 1,000 person-years, compared with a rate of 6.4 cases per 1,000 person-years for the most fit group (P < .001).
After adjusting for factors that could affect risk of ADRD, compared with the lowest-fit group, the highest-fit and fit groups were 33% and 26% less likely to develop ADRD, respectively, while the moderate-fit and low-fit groups were 20% and 13% less likely to develop the disease, respectively.

The findings suggest that the association between cardiorespiratory fitness and ADRD risk is “inverse, independent, and graded,” the researchers said in their conference abstract.
“The idea that you can reduce your risk for Alzheimer’s disease by simply increasing your activity is very promising, especially since there are no adequate treatments to prevent or stop the progression of the disease,” Dr. Zamrini added in the news release.
“We hope to develop a simple scale that can be individualized so people can see the benefits that even incremental improvements in fitness can deliver,” he said.
The next vital sign?
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Boston, noted that “for decades and with increasing body of support from studies like this, we have known that preventing dementia is based on healthy behaviors for the brain including a proper diet (NASH and/or Mediterranean), exercise regimen (aerobic/cardio more than anaerobic/weight-lifting), sleep hygiene, and social and intellectual engagements.”
“Frankly, what’s good for the body is good for the brain,” said Dr. Lakhan.
“It should be noted that the measure studied here is cardiorespiratory fitness, which has been associated with heart disease and resulting death, death from any cause, and now brain health,” Dr. Lakhan said.
“This powerful predictor may in fact be the next vital sign, after your heart rate and blood pressure, from which your primary care provider can make a personalized treatment plan,” he added.
“Accelerating this process, the ability to measure cardiorespiratory fitness traditionally from huge stationary machines down to wearables like a watch or ring, or even your iPhone or Android, is just on the horizon,” Dr. Lakhan said.
“Instead of tracking just your weight, shape, and BMI, personal fitness may be tailored to optimizing this indicator and further empowering individuals to take charge of their health,” he said.
The study was supported by the National Institute on Aging, the National Institutes of Health, the U.S. Department of Veterans Affairs, the Washington DC VA Medical Center, and George Washington University. Dr. Zamrini and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new findings suggest. “One exciting finding of this study is that as people’s fitness improved, their risk of Alzheimer’s disease decreased – it was not an all-or-nothing proposition,” study investigator Edward Zamrini, MD, of the Washington DC VA Medical Center, said in a news release.
The findings suggest that people can work toward making incremental changes and improvements in their physical fitness, which may help decrease their risk of dementia, Dr. Zamrini added.
The findings were presented at the 2022 annual meeting of the American Academy of Neurology.
Effective prevention strategy
Using the Veterans Health Administration database, Dr. Zamrini and colleagues identified 649,605 veterans (mean age, 61 years) free of Alzheimer’s disease and related disorders (ADRD) when they completed standardized exercise treadmill tests between 2000 and 2017.
They divided participants into five age-specific fitness groups, from least fit to most fit, based on peak metabolic equivalents (METs) achieved during the treadmill test: lowest-fit (METs, ±3.8), low-fit (METs, ±5.8), moderate-fit (METs, ±7.5), fit (METs, ±9.2), and highest-fit (METs, ±11.7).
In unadjusted analysis, veterans with the lowest cardiorespiratory fitness developed ADRD at a rate of 9.5 cases per 1,000 person-years, compared with a rate of 6.4 cases per 1,000 person-years for the most fit group (P < .001).
After adjusting for factors that could affect risk of ADRD, compared with the lowest-fit group, the highest-fit and fit groups were 33% and 26% less likely to develop ADRD, respectively, while the moderate-fit and low-fit groups were 20% and 13% less likely to develop the disease, respectively.

The findings suggest that the association between cardiorespiratory fitness and ADRD risk is “inverse, independent, and graded,” the researchers said in their conference abstract.
“The idea that you can reduce your risk for Alzheimer’s disease by simply increasing your activity is very promising, especially since there are no adequate treatments to prevent or stop the progression of the disease,” Dr. Zamrini added in the news release.
“We hope to develop a simple scale that can be individualized so people can see the benefits that even incremental improvements in fitness can deliver,” he said.
The next vital sign?
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Boston, noted that “for decades and with increasing body of support from studies like this, we have known that preventing dementia is based on healthy behaviors for the brain including a proper diet (NASH and/or Mediterranean), exercise regimen (aerobic/cardio more than anaerobic/weight-lifting), sleep hygiene, and social and intellectual engagements.”
“Frankly, what’s good for the body is good for the brain,” said Dr. Lakhan.
“It should be noted that the measure studied here is cardiorespiratory fitness, which has been associated with heart disease and resulting death, death from any cause, and now brain health,” Dr. Lakhan said.
“This powerful predictor may in fact be the next vital sign, after your heart rate and blood pressure, from which your primary care provider can make a personalized treatment plan,” he added.
“Accelerating this process, the ability to measure cardiorespiratory fitness traditionally from huge stationary machines down to wearables like a watch or ring, or even your iPhone or Android, is just on the horizon,” Dr. Lakhan said.
“Instead of tracking just your weight, shape, and BMI, personal fitness may be tailored to optimizing this indicator and further empowering individuals to take charge of their health,” he said.
The study was supported by the National Institute on Aging, the National Institutes of Health, the U.S. Department of Veterans Affairs, the Washington DC VA Medical Center, and George Washington University. Dr. Zamrini and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAN 2022
More years of ice hockey play tied to higher CTE risk
new research suggests. Early results from a study that examined donor brains showed that each additional year of ice hockey play increased the risk for CTE by 23%.
This information should be on the “radar” of all clinicians, said coinvestigator Jesse Mez, MD, associate professor of neurology at Boston University. “When they’re talking to kids and families and parents about playing contact sports, they should discuss the benefits as well as the risks so all that information can be taken into consideration.”
Dr. Mez noted that clinicians should also consider the amount of hockey played when assessing patients for thinking and memory trouble later in life. “CTE could be in the differential diagnosis,” he said.
The study findings were presented at the 2022 annual meeting of the American Academy of Neurology.
Football data
CTE is a neurodegenerative disease associated with repetitive hits to the head. In previous research, the investigators showed that the more that athletes play American football, the more likely they are to develop CTE.
“Hockey, like football, involves repetitive head impacts as part of the game,” said Dr. Mez. “So we hypothesized that we would see a similar type of dose-response relationship in hockey.”
From two brain banks – the Veterans Affairs–Boston University–Concussion Legacy Foundation and the Framingham Heart Study – the researchers accessed 74 consecutive brains from donors who had played ice hockey. They collected information about hockey play during “pretty comprehensive” interviews with next of kin, Dr. Mez reported.
The study participants ranged in age from 13 to 91 years. The cause of death varied; most died with end-stage dementia and neurodegenerative disease, but some died of cardiovascular disease, and others from accidents.
For 9% of the individuals, the highest level of play was a youth league; 34% had reached the high school level, 30% reached the juniors/college level, and 26% played professionally. In addition, 46% played another contact sport – including 43% who played American football.
Primary outcomes included evidence of CTE from stage 0 (no CTE) to stage IV and severity of CTE, which was defined by the amount of neurofibrillary tangle (NFT) burden in 11 brain regions. For this burden, the score ranged from 0 (absent) to 3 (severe) in each region for a total range of 0-33.
Dr. Mez noted that, in CTE, tau protein accumulates abnormally. “It typically begins in the cortex in the frontal lobe and then spreads to other parts of the brain, including to the medial temporal structures, and is widespread by stage IV.”
The researchers estimated the association of duration of ice hockey play in years with each neuropathologic outcome and adjusted for age at death and duration of football play.
Consistent findings
Results showed that, of the 74 donors, 40 (54%) had CTE. Each additional year of hockey play corresponded to increased chances for having CTE (odds ratio, 1.23; 95% confidence interval, 11%-36%; P < .01). This increase in risk is similar to that which was found with football players, Dr. Mez noted. This was somewhat surprising, as hockey involves fewer “hits” than football.
“Hits are not as quintessential to the game of hockey as they are in football, where contacts occur with nearly every play,” he said. “In football, you have several hundred impacts over the course of a season.”
Researchers also found a 15% increase in odds for increasing one CTE stage (95% CI, 8%-22%; P < .01), and a .03 standard deviation increase in cumulative NFT burden (95% CI, 0.01-0.05; P < .01).
Dr. Mez noted that the fact that the results were consistent across different outcomes “improves the validity” of the findings.
In a sensitivity analysis that excluded participants who also played football, estimates “were pretty similar” to those in the full analysis, said Dr. Mez.
The investigators have not yet examined the effect of level of hockey play, such as professionally or at the college level, on CTE risk. However, in football players, they found that level of play is another “valuable predictor of CTE pathology,” Dr. Mez said, adding that level of play, position played, and years of play “are all probably contributing” to CTE risk.
Asking about years of play is useful in a clinical setting. “It’s very easy for a clinician to ask patients how many years of hockey they played,” said Dr. Mez.
Overall, the new results are important, as “millions of individuals” play contact sports, whether that is hockey, football, or European soccer, he added. “And for all sports, there seems to be this relationship between more play and risk of this disease.”
‘Skewed’ population?
Commenting on the findings, Frank Conidi, MD, director, Florida Center for Health and Sports Neurology, Port St. Lucie, said he was surprised the investigators found a 23% per year increase in risk for CTE among hockey players.
Dr. Conidi has played hockey himself and works with the Florida Panthers of the National Hockey League. In his practice, he treats retired professional football players who have neurodegenerative disorders. From his experience, the number of repetitive direct head impacts in football is significantly higher than in hockey. “Most of the forces seen in hockey are from hits to the body, where the force is transferred to the head,” said Dr. Conidi, who was not involved with the research.
He noted differences in the way hockey is played around the world. In European countries, for example, the ice surface is relatively large and the emphasis tends to be more on skill than hitting.
“It would have been interesting to have the study group analyze the data based on where the athlete grew up,” he said. Dr. Conidi would also like to know when the participants played hockey. “The game is vastly different now than it was in the 1970s, ‘80s, and early ‘90s, when there was more fighting, less protective gear, and more hitting in general.”
As is the case for most studies of CTE in athletes, the study population is “skewed” because the participants likely had neurocognitive and other problems that led to their decision to donate their brain, said Dr. Conidi.
He also doesn’t believe the study should be the sole factor in a decision to continue or stop playing hockey. “We are still in the infancy stages of understanding the effects of high-impact sports on athletes’ brains.”
The study received funding from the National Institute of Neurological Diseases and Stroke and the National Institute on Aging. Dr. Mez and Dr. Conidi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Early results from a study that examined donor brains showed that each additional year of ice hockey play increased the risk for CTE by 23%.
This information should be on the “radar” of all clinicians, said coinvestigator Jesse Mez, MD, associate professor of neurology at Boston University. “When they’re talking to kids and families and parents about playing contact sports, they should discuss the benefits as well as the risks so all that information can be taken into consideration.”
Dr. Mez noted that clinicians should also consider the amount of hockey played when assessing patients for thinking and memory trouble later in life. “CTE could be in the differential diagnosis,” he said.
The study findings were presented at the 2022 annual meeting of the American Academy of Neurology.
Football data
CTE is a neurodegenerative disease associated with repetitive hits to the head. In previous research, the investigators showed that the more that athletes play American football, the more likely they are to develop CTE.
“Hockey, like football, involves repetitive head impacts as part of the game,” said Dr. Mez. “So we hypothesized that we would see a similar type of dose-response relationship in hockey.”
From two brain banks – the Veterans Affairs–Boston University–Concussion Legacy Foundation and the Framingham Heart Study – the researchers accessed 74 consecutive brains from donors who had played ice hockey. They collected information about hockey play during “pretty comprehensive” interviews with next of kin, Dr. Mez reported.
The study participants ranged in age from 13 to 91 years. The cause of death varied; most died with end-stage dementia and neurodegenerative disease, but some died of cardiovascular disease, and others from accidents.
For 9% of the individuals, the highest level of play was a youth league; 34% had reached the high school level, 30% reached the juniors/college level, and 26% played professionally. In addition, 46% played another contact sport – including 43% who played American football.
Primary outcomes included evidence of CTE from stage 0 (no CTE) to stage IV and severity of CTE, which was defined by the amount of neurofibrillary tangle (NFT) burden in 11 brain regions. For this burden, the score ranged from 0 (absent) to 3 (severe) in each region for a total range of 0-33.
Dr. Mez noted that, in CTE, tau protein accumulates abnormally. “It typically begins in the cortex in the frontal lobe and then spreads to other parts of the brain, including to the medial temporal structures, and is widespread by stage IV.”
The researchers estimated the association of duration of ice hockey play in years with each neuropathologic outcome and adjusted for age at death and duration of football play.
Consistent findings
Results showed that, of the 74 donors, 40 (54%) had CTE. Each additional year of hockey play corresponded to increased chances for having CTE (odds ratio, 1.23; 95% confidence interval, 11%-36%; P < .01). This increase in risk is similar to that which was found with football players, Dr. Mez noted. This was somewhat surprising, as hockey involves fewer “hits” than football.
“Hits are not as quintessential to the game of hockey as they are in football, where contacts occur with nearly every play,” he said. “In football, you have several hundred impacts over the course of a season.”
Researchers also found a 15% increase in odds for increasing one CTE stage (95% CI, 8%-22%; P < .01), and a .03 standard deviation increase in cumulative NFT burden (95% CI, 0.01-0.05; P < .01).
Dr. Mez noted that the fact that the results were consistent across different outcomes “improves the validity” of the findings.
In a sensitivity analysis that excluded participants who also played football, estimates “were pretty similar” to those in the full analysis, said Dr. Mez.
The investigators have not yet examined the effect of level of hockey play, such as professionally or at the college level, on CTE risk. However, in football players, they found that level of play is another “valuable predictor of CTE pathology,” Dr. Mez said, adding that level of play, position played, and years of play “are all probably contributing” to CTE risk.
Asking about years of play is useful in a clinical setting. “It’s very easy for a clinician to ask patients how many years of hockey they played,” said Dr. Mez.
Overall, the new results are important, as “millions of individuals” play contact sports, whether that is hockey, football, or European soccer, he added. “And for all sports, there seems to be this relationship between more play and risk of this disease.”
‘Skewed’ population?
Commenting on the findings, Frank Conidi, MD, director, Florida Center for Health and Sports Neurology, Port St. Lucie, said he was surprised the investigators found a 23% per year increase in risk for CTE among hockey players.
Dr. Conidi has played hockey himself and works with the Florida Panthers of the National Hockey League. In his practice, he treats retired professional football players who have neurodegenerative disorders. From his experience, the number of repetitive direct head impacts in football is significantly higher than in hockey. “Most of the forces seen in hockey are from hits to the body, where the force is transferred to the head,” said Dr. Conidi, who was not involved with the research.
He noted differences in the way hockey is played around the world. In European countries, for example, the ice surface is relatively large and the emphasis tends to be more on skill than hitting.
“It would have been interesting to have the study group analyze the data based on where the athlete grew up,” he said. Dr. Conidi would also like to know when the participants played hockey. “The game is vastly different now than it was in the 1970s, ‘80s, and early ‘90s, when there was more fighting, less protective gear, and more hitting in general.”
As is the case for most studies of CTE in athletes, the study population is “skewed” because the participants likely had neurocognitive and other problems that led to their decision to donate their brain, said Dr. Conidi.
He also doesn’t believe the study should be the sole factor in a decision to continue or stop playing hockey. “We are still in the infancy stages of understanding the effects of high-impact sports on athletes’ brains.”
The study received funding from the National Institute of Neurological Diseases and Stroke and the National Institute on Aging. Dr. Mez and Dr. Conidi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Early results from a study that examined donor brains showed that each additional year of ice hockey play increased the risk for CTE by 23%.
This information should be on the “radar” of all clinicians, said coinvestigator Jesse Mez, MD, associate professor of neurology at Boston University. “When they’re talking to kids and families and parents about playing contact sports, they should discuss the benefits as well as the risks so all that information can be taken into consideration.”
Dr. Mez noted that clinicians should also consider the amount of hockey played when assessing patients for thinking and memory trouble later in life. “CTE could be in the differential diagnosis,” he said.
The study findings were presented at the 2022 annual meeting of the American Academy of Neurology.
Football data
CTE is a neurodegenerative disease associated with repetitive hits to the head. In previous research, the investigators showed that the more that athletes play American football, the more likely they are to develop CTE.
“Hockey, like football, involves repetitive head impacts as part of the game,” said Dr. Mez. “So we hypothesized that we would see a similar type of dose-response relationship in hockey.”
From two brain banks – the Veterans Affairs–Boston University–Concussion Legacy Foundation and the Framingham Heart Study – the researchers accessed 74 consecutive brains from donors who had played ice hockey. They collected information about hockey play during “pretty comprehensive” interviews with next of kin, Dr. Mez reported.
The study participants ranged in age from 13 to 91 years. The cause of death varied; most died with end-stage dementia and neurodegenerative disease, but some died of cardiovascular disease, and others from accidents.
For 9% of the individuals, the highest level of play was a youth league; 34% had reached the high school level, 30% reached the juniors/college level, and 26% played professionally. In addition, 46% played another contact sport – including 43% who played American football.
Primary outcomes included evidence of CTE from stage 0 (no CTE) to stage IV and severity of CTE, which was defined by the amount of neurofibrillary tangle (NFT) burden in 11 brain regions. For this burden, the score ranged from 0 (absent) to 3 (severe) in each region for a total range of 0-33.
Dr. Mez noted that, in CTE, tau protein accumulates abnormally. “It typically begins in the cortex in the frontal lobe and then spreads to other parts of the brain, including to the medial temporal structures, and is widespread by stage IV.”
The researchers estimated the association of duration of ice hockey play in years with each neuropathologic outcome and adjusted for age at death and duration of football play.
Consistent findings
Results showed that, of the 74 donors, 40 (54%) had CTE. Each additional year of hockey play corresponded to increased chances for having CTE (odds ratio, 1.23; 95% confidence interval, 11%-36%; P < .01). This increase in risk is similar to that which was found with football players, Dr. Mez noted. This was somewhat surprising, as hockey involves fewer “hits” than football.
“Hits are not as quintessential to the game of hockey as they are in football, where contacts occur with nearly every play,” he said. “In football, you have several hundred impacts over the course of a season.”
Researchers also found a 15% increase in odds for increasing one CTE stage (95% CI, 8%-22%; P < .01), and a .03 standard deviation increase in cumulative NFT burden (95% CI, 0.01-0.05; P < .01).
Dr. Mez noted that the fact that the results were consistent across different outcomes “improves the validity” of the findings.
In a sensitivity analysis that excluded participants who also played football, estimates “were pretty similar” to those in the full analysis, said Dr. Mez.
The investigators have not yet examined the effect of level of hockey play, such as professionally or at the college level, on CTE risk. However, in football players, they found that level of play is another “valuable predictor of CTE pathology,” Dr. Mez said, adding that level of play, position played, and years of play “are all probably contributing” to CTE risk.
Asking about years of play is useful in a clinical setting. “It’s very easy for a clinician to ask patients how many years of hockey they played,” said Dr. Mez.
Overall, the new results are important, as “millions of individuals” play contact sports, whether that is hockey, football, or European soccer, he added. “And for all sports, there seems to be this relationship between more play and risk of this disease.”
‘Skewed’ population?
Commenting on the findings, Frank Conidi, MD, director, Florida Center for Health and Sports Neurology, Port St. Lucie, said he was surprised the investigators found a 23% per year increase in risk for CTE among hockey players.
Dr. Conidi has played hockey himself and works with the Florida Panthers of the National Hockey League. In his practice, he treats retired professional football players who have neurodegenerative disorders. From his experience, the number of repetitive direct head impacts in football is significantly higher than in hockey. “Most of the forces seen in hockey are from hits to the body, where the force is transferred to the head,” said Dr. Conidi, who was not involved with the research.
He noted differences in the way hockey is played around the world. In European countries, for example, the ice surface is relatively large and the emphasis tends to be more on skill than hitting.
“It would have been interesting to have the study group analyze the data based on where the athlete grew up,” he said. Dr. Conidi would also like to know when the participants played hockey. “The game is vastly different now than it was in the 1970s, ‘80s, and early ‘90s, when there was more fighting, less protective gear, and more hitting in general.”
As is the case for most studies of CTE in athletes, the study population is “skewed” because the participants likely had neurocognitive and other problems that led to their decision to donate their brain, said Dr. Conidi.
He also doesn’t believe the study should be the sole factor in a decision to continue or stop playing hockey. “We are still in the infancy stages of understanding the effects of high-impact sports on athletes’ brains.”
The study received funding from the National Institute of Neurological Diseases and Stroke and the National Institute on Aging. Dr. Mez and Dr. Conidi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAN 2022
Keto diet in MS tied to less disability, better quality of life
, new research suggests.
High-fat, low-carbohydrate ketogenic diets mimic a fasting state and promote a more efficient use of energy – and have previously been shown to affect immune regulation. The diet helps lower blood sugar in individuals with type 2 diabetes and has been used for years to improve seizure control in patients with epilepsy, researchers note.
However, “there is a paucity of literature on the ketogenic diet in MS currently,” said principal investigator J. Nicholas Brenton, MD, University of Virginia, Charlottesville.
“The current study demonstrates the safety, tolerability, and potential clinical benefits of a ketogenic diet over 6 months in patients with relapsing MS,” Dr. Brenton said.
The were presented at the 2022 annual meeting of the American Academy of Neurology.
Palatable, beneficial
The open-label, uncontrolled study included 65 patients with relapsing MS who followed a ketogenic diet for 6 months. Investigators monitored adherence by daily urine ketone testing.
Patient-reported fatigue, depression, and quality-of-life scores were obtained at baseline, in addition to fasting adipokines and pertinent MS-related clinical outcome metrics. Baseline study metrics were repeated at 3 and/or 6 months while on the ketogenic diet.
Of the patient group, 83% adhered to the ketogenic diet for the full 6-month study period.
The ketogenic diet was associated with reductions in fat mass from baseline to 6 months (41.3 vs. 32.0 kg; P < .001) and a significant decline in fatigue and depression scores, the investigators reported.
MS quality-of-life physical and mental composite scores also improved while on the ketogenic diet (P < .001 for both).
A significant decrease from baseline to 6 months in Expanded Disability Status Scale scores, signifying improvement, was observed (2.3 vs. 1.9; P < .001).
Improvements were also shown on the 6-minute walk (1,631 vs. 1,733 feet; P < .001) and the nine-hole peg test (21.5 vs. 20.3 seconds; P < .001).
At 6 months on the diet, fasting serum leptin was significantly lower (25.5 vs. 14 ng/mL; P <.001), and adiponectin was higher (11.4 vs. 13.5 μg/mL, P = .002).
Justifies further research
The current study builds on an earlier one that Dr. Brenton and colleagues conducted in 2019 that showed that the ketogenic diet was feasible in patients with MS. “Our data justify the need for future studies of ketogenic diets as a complementary therapeutic approach to the treatment of MS,” Dr. Brenton said.
He noted that there may be multiple mechanisms of benefit when considering the ketogenic diet. “One avenue is via reduction in total body fat. This is an important aspect as we continue to learn more about the role of obesity and fat-derived inflammation in MS,” Dr. Brenton said.
“Ketogenic diets also have immunomodulatory properties,” such as the capacity to reduce oxidative damage from metabolic stress, increase mitochondrial biogenesis, and reduce systemic inflammation, he added. “These intrinsic properties of the ketogenic diet make it appealing to study in immune-mediated diseases, such as MS.”
Dr. Brenton cautioned that the data demonstrate the diet’s safety over 6 months but that the study was not designed to assess its long-term implications in MS. “Thus, while our results support the rationale for a larger-scale study of ketogenic diets as a complementary treatment for MS, our data does not support its widespread adoption outside of a clinical trial,” he said.
Remarkable adherence
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Boston, noted that “variations of the ketogenic diet have been popularized in the general population for weight loss and further studied for other medical conditions [that are] largely immune-related, including MS.”
He noted that it was “remarkable” that the vast majority of study participants with MS adhered to the very regimented ketogenic diet for 6 months.
Seeing this translate into the real world “will be the next milestone, in addition to its impact on relapses and brain lesions as seen on MRI,” which are the classic markers of MS, said Dr. Lakhan, who was not involved with the research.
He cautioned that “even if one can follow the ketogenic diet, certain conditions can be made worse. This includes kidney stones, liver disease, reflux, constipation, and other metabolic disorders.”
The study was funded by the National Center for Advancing Translational Sciences of the National Institutes of Health and by the ZiMS Foundation. Dr. Brenton and Dr. Lakhan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
High-fat, low-carbohydrate ketogenic diets mimic a fasting state and promote a more efficient use of energy – and have previously been shown to affect immune regulation. The diet helps lower blood sugar in individuals with type 2 diabetes and has been used for years to improve seizure control in patients with epilepsy, researchers note.
However, “there is a paucity of literature on the ketogenic diet in MS currently,” said principal investigator J. Nicholas Brenton, MD, University of Virginia, Charlottesville.
“The current study demonstrates the safety, tolerability, and potential clinical benefits of a ketogenic diet over 6 months in patients with relapsing MS,” Dr. Brenton said.
The were presented at the 2022 annual meeting of the American Academy of Neurology.
Palatable, beneficial
The open-label, uncontrolled study included 65 patients with relapsing MS who followed a ketogenic diet for 6 months. Investigators monitored adherence by daily urine ketone testing.
Patient-reported fatigue, depression, and quality-of-life scores were obtained at baseline, in addition to fasting adipokines and pertinent MS-related clinical outcome metrics. Baseline study metrics were repeated at 3 and/or 6 months while on the ketogenic diet.
Of the patient group, 83% adhered to the ketogenic diet for the full 6-month study period.
The ketogenic diet was associated with reductions in fat mass from baseline to 6 months (41.3 vs. 32.0 kg; P < .001) and a significant decline in fatigue and depression scores, the investigators reported.
MS quality-of-life physical and mental composite scores also improved while on the ketogenic diet (P < .001 for both).
A significant decrease from baseline to 6 months in Expanded Disability Status Scale scores, signifying improvement, was observed (2.3 vs. 1.9; P < .001).
Improvements were also shown on the 6-minute walk (1,631 vs. 1,733 feet; P < .001) and the nine-hole peg test (21.5 vs. 20.3 seconds; P < .001).
At 6 months on the diet, fasting serum leptin was significantly lower (25.5 vs. 14 ng/mL; P <.001), and adiponectin was higher (11.4 vs. 13.5 μg/mL, P = .002).
Justifies further research
The current study builds on an earlier one that Dr. Brenton and colleagues conducted in 2019 that showed that the ketogenic diet was feasible in patients with MS. “Our data justify the need for future studies of ketogenic diets as a complementary therapeutic approach to the treatment of MS,” Dr. Brenton said.
He noted that there may be multiple mechanisms of benefit when considering the ketogenic diet. “One avenue is via reduction in total body fat. This is an important aspect as we continue to learn more about the role of obesity and fat-derived inflammation in MS,” Dr. Brenton said.
“Ketogenic diets also have immunomodulatory properties,” such as the capacity to reduce oxidative damage from metabolic stress, increase mitochondrial biogenesis, and reduce systemic inflammation, he added. “These intrinsic properties of the ketogenic diet make it appealing to study in immune-mediated diseases, such as MS.”
Dr. Brenton cautioned that the data demonstrate the diet’s safety over 6 months but that the study was not designed to assess its long-term implications in MS. “Thus, while our results support the rationale for a larger-scale study of ketogenic diets as a complementary treatment for MS, our data does not support its widespread adoption outside of a clinical trial,” he said.
Remarkable adherence
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Boston, noted that “variations of the ketogenic diet have been popularized in the general population for weight loss and further studied for other medical conditions [that are] largely immune-related, including MS.”
He noted that it was “remarkable” that the vast majority of study participants with MS adhered to the very regimented ketogenic diet for 6 months.
Seeing this translate into the real world “will be the next milestone, in addition to its impact on relapses and brain lesions as seen on MRI,” which are the classic markers of MS, said Dr. Lakhan, who was not involved with the research.
He cautioned that “even if one can follow the ketogenic diet, certain conditions can be made worse. This includes kidney stones, liver disease, reflux, constipation, and other metabolic disorders.”
The study was funded by the National Center for Advancing Translational Sciences of the National Institutes of Health and by the ZiMS Foundation. Dr. Brenton and Dr. Lakhan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
High-fat, low-carbohydrate ketogenic diets mimic a fasting state and promote a more efficient use of energy – and have previously been shown to affect immune regulation. The diet helps lower blood sugar in individuals with type 2 diabetes and has been used for years to improve seizure control in patients with epilepsy, researchers note.
However, “there is a paucity of literature on the ketogenic diet in MS currently,” said principal investigator J. Nicholas Brenton, MD, University of Virginia, Charlottesville.
“The current study demonstrates the safety, tolerability, and potential clinical benefits of a ketogenic diet over 6 months in patients with relapsing MS,” Dr. Brenton said.
The were presented at the 2022 annual meeting of the American Academy of Neurology.
Palatable, beneficial
The open-label, uncontrolled study included 65 patients with relapsing MS who followed a ketogenic diet for 6 months. Investigators monitored adherence by daily urine ketone testing.
Patient-reported fatigue, depression, and quality-of-life scores were obtained at baseline, in addition to fasting adipokines and pertinent MS-related clinical outcome metrics. Baseline study metrics were repeated at 3 and/or 6 months while on the ketogenic diet.
Of the patient group, 83% adhered to the ketogenic diet for the full 6-month study period.
The ketogenic diet was associated with reductions in fat mass from baseline to 6 months (41.3 vs. 32.0 kg; P < .001) and a significant decline in fatigue and depression scores, the investigators reported.
MS quality-of-life physical and mental composite scores also improved while on the ketogenic diet (P < .001 for both).
A significant decrease from baseline to 6 months in Expanded Disability Status Scale scores, signifying improvement, was observed (2.3 vs. 1.9; P < .001).
Improvements were also shown on the 6-minute walk (1,631 vs. 1,733 feet; P < .001) and the nine-hole peg test (21.5 vs. 20.3 seconds; P < .001).
At 6 months on the diet, fasting serum leptin was significantly lower (25.5 vs. 14 ng/mL; P <.001), and adiponectin was higher (11.4 vs. 13.5 μg/mL, P = .002).
Justifies further research
The current study builds on an earlier one that Dr. Brenton and colleagues conducted in 2019 that showed that the ketogenic diet was feasible in patients with MS. “Our data justify the need for future studies of ketogenic diets as a complementary therapeutic approach to the treatment of MS,” Dr. Brenton said.
He noted that there may be multiple mechanisms of benefit when considering the ketogenic diet. “One avenue is via reduction in total body fat. This is an important aspect as we continue to learn more about the role of obesity and fat-derived inflammation in MS,” Dr. Brenton said.
“Ketogenic diets also have immunomodulatory properties,” such as the capacity to reduce oxidative damage from metabolic stress, increase mitochondrial biogenesis, and reduce systemic inflammation, he added. “These intrinsic properties of the ketogenic diet make it appealing to study in immune-mediated diseases, such as MS.”
Dr. Brenton cautioned that the data demonstrate the diet’s safety over 6 months but that the study was not designed to assess its long-term implications in MS. “Thus, while our results support the rationale for a larger-scale study of ketogenic diets as a complementary treatment for MS, our data does not support its widespread adoption outside of a clinical trial,” he said.
Remarkable adherence
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Boston, noted that “variations of the ketogenic diet have been popularized in the general population for weight loss and further studied for other medical conditions [that are] largely immune-related, including MS.”
He noted that it was “remarkable” that the vast majority of study participants with MS adhered to the very regimented ketogenic diet for 6 months.
Seeing this translate into the real world “will be the next milestone, in addition to its impact on relapses and brain lesions as seen on MRI,” which are the classic markers of MS, said Dr. Lakhan, who was not involved with the research.
He cautioned that “even if one can follow the ketogenic diet, certain conditions can be made worse. This includes kidney stones, liver disease, reflux, constipation, and other metabolic disorders.”
The study was funded by the National Center for Advancing Translational Sciences of the National Institutes of Health and by the ZiMS Foundation. Dr. Brenton and Dr. Lakhan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAN 2022
Some reproductive factors linked with risk of dementia
Certain reproductive factors are associated with greater or lower risk of dementia, according to researchers who conducted a large population-based study with UK Biobank data.
Jessica Gong, a PhD candidate at the George Institute for Global Health at University of New South Wales in Australia, and coauthors found a greater dementia risk in women with early and late menarche, women who were younger when they first gave birth, and those who had had a hysterectomy, especially those who had a hysterectomy without concomitant oophorectomy or with a previous oophorectomy.
After controlling for key confounders, the researchers found lower risk of all-cause dementia if women had ever been pregnant, ever had an abortion, had a longer reproductive span, or had later menopause.
Use of oral contraceptive pills was associated with a lower dementia risk, they found.
In this study, there was no evidence that hormone therapy (HT) was associated with dementia risk (hazard ratio, 0.99, 95% confidence interval [0.90-1.09], P =.0828).
The analysis, published online April 5 in PLOS Medicine, comprised 273,240 women and 228,957 men without prevalent dementia.
The authors noted that dementia rates are increasing. Globally, 50 million people live with dementia, and the number is expected to triple by 2050, according to Alzheimer’s Disease International.
“Our study identified certain reproductive factors related to shorter exposure to endogenous estrogen were associated with increased risk of dementia, highlighting the susceptibility in dementia risk pertaining to women,” Ms. Gong told this publication.
Risk comparison of men and women
Men were included in this study to compare the association between number of children fathered and the risk of all-cause dementia, with the association in their female counterparts.
The U-shaped associations between the number of children and dementia risk were similar for both sexes, suggesting that the risk difference in women may not be associated with factors associated with childbearing
“It may be more related to social and behavioral factors in parenthood, rather than biological factors involved in childbearing,” Ms. Gong said.
Compared with those with two children, for those without children, the multiple adjusted HR (95% CI) was 1.18 (1.04, 1.33) (P = .027) for women and 1.10 (0.98-1.23) P = .164) for men.
For those with four or more children, the HR was 1.14 (0.98, 1.33) (P = .132) for women and 1.26 (1.10-1.45) (P = .003) for men.
Rachel Buckley, PhD, assistant professor of neurology with a dual appointment at Brigham and Women’s and Massachusetts General hospitals in Boston, told this publication she found the comparison of dementia risk with number of children in men and women “fascinating.”
She said the argument usually is that if women have had more births, then they have had more estrogen through their body because women get a huge injection of hormones in pregnancy.
“The idea is that the more pregnancies you have the more protected you are. But this study put that on its head, because if men and women are showing increased [dementia] risk in the number of children they have, it suggests there must be something about having the children – not necessarily the circulating hormones – that might be having an impact,” Dr. Buckley said.
“I had never thought to compare the number of children in men. I do find that very interesting,” she said.
As for the lack of a link between HT and dementia risk, in this study she said, she wouldn’t shut the door on that discussion just yet.
She noted the long history of controversy in the field about whether there is a protective factor against dementia for estrogen or whether exposure to estrogen leads to increased risk.
Before the landmark Women’s Health Initiative (WHI) study in the 1990s, she pointed out, there was evidence in many observational studies that women who had longer exposure to estrogen – whether that was earlier age at first period and later age at menopause combined or women had taken hormone therapy at some point, had less risk for dementia.
Dr. Buckley said that in a secondary outcome of WHI, however, “there was increased risk for progression to dementia in women who were taking hormone therapy which essentially flipped the field on its ahead because until that point everybody thought that estrogen was a protective factor.”
She said although this study found no association with dementia, she still thinks HT has a role to play and that it may just need to be better tailored to individuals.
“If you think about it, we have our tailored cocktail of hormones in our body and who’s to say that my hormones are going to be the same as yours? Why should you and I be put on the same hormone therapy and assume that will give us the same outcome? I think we could do a lot better with customization and calibration of hormones to aid in women’s health.”
Lifetime approach to dementia
Ms. Gong says future dementia risk-reduction strategies should consider sex-specific risk, and consider the reproductive events that took place in women’s lifespans as well as their entire hormone history when assessing dementia risk, to ensure that the strategies are sex sensitive.
Dr. Buckley agrees: “I don’t think we should ever think about dementia in terms of 65 onwards. We know this disease is insidious and it starts very, very early.”
Regarding limitations, the authors noted that it was a retrospective study that included self-reported measures of reproductive factors, which may be inherently subject to recall bias.
A coauthor does consultant work for Amgen, Freeline, and Kirin outside the submitted work. There were no other relevant financial disclosures.
Certain reproductive factors are associated with greater or lower risk of dementia, according to researchers who conducted a large population-based study with UK Biobank data.
Jessica Gong, a PhD candidate at the George Institute for Global Health at University of New South Wales in Australia, and coauthors found a greater dementia risk in women with early and late menarche, women who were younger when they first gave birth, and those who had had a hysterectomy, especially those who had a hysterectomy without concomitant oophorectomy or with a previous oophorectomy.
After controlling for key confounders, the researchers found lower risk of all-cause dementia if women had ever been pregnant, ever had an abortion, had a longer reproductive span, or had later menopause.
Use of oral contraceptive pills was associated with a lower dementia risk, they found.
In this study, there was no evidence that hormone therapy (HT) was associated with dementia risk (hazard ratio, 0.99, 95% confidence interval [0.90-1.09], P =.0828).
The analysis, published online April 5 in PLOS Medicine, comprised 273,240 women and 228,957 men without prevalent dementia.
The authors noted that dementia rates are increasing. Globally, 50 million people live with dementia, and the number is expected to triple by 2050, according to Alzheimer’s Disease International.
“Our study identified certain reproductive factors related to shorter exposure to endogenous estrogen were associated with increased risk of dementia, highlighting the susceptibility in dementia risk pertaining to women,” Ms. Gong told this publication.
Risk comparison of men and women
Men were included in this study to compare the association between number of children fathered and the risk of all-cause dementia, with the association in their female counterparts.
The U-shaped associations between the number of children and dementia risk were similar for both sexes, suggesting that the risk difference in women may not be associated with factors associated with childbearing
“It may be more related to social and behavioral factors in parenthood, rather than biological factors involved in childbearing,” Ms. Gong said.
Compared with those with two children, for those without children, the multiple adjusted HR (95% CI) was 1.18 (1.04, 1.33) (P = .027) for women and 1.10 (0.98-1.23) P = .164) for men.
For those with four or more children, the HR was 1.14 (0.98, 1.33) (P = .132) for women and 1.26 (1.10-1.45) (P = .003) for men.
Rachel Buckley, PhD, assistant professor of neurology with a dual appointment at Brigham and Women’s and Massachusetts General hospitals in Boston, told this publication she found the comparison of dementia risk with number of children in men and women “fascinating.”
She said the argument usually is that if women have had more births, then they have had more estrogen through their body because women get a huge injection of hormones in pregnancy.
“The idea is that the more pregnancies you have the more protected you are. But this study put that on its head, because if men and women are showing increased [dementia] risk in the number of children they have, it suggests there must be something about having the children – not necessarily the circulating hormones – that might be having an impact,” Dr. Buckley said.
“I had never thought to compare the number of children in men. I do find that very interesting,” she said.
As for the lack of a link between HT and dementia risk, in this study she said, she wouldn’t shut the door on that discussion just yet.
She noted the long history of controversy in the field about whether there is a protective factor against dementia for estrogen or whether exposure to estrogen leads to increased risk.
Before the landmark Women’s Health Initiative (WHI) study in the 1990s, she pointed out, there was evidence in many observational studies that women who had longer exposure to estrogen – whether that was earlier age at first period and later age at menopause combined or women had taken hormone therapy at some point, had less risk for dementia.
Dr. Buckley said that in a secondary outcome of WHI, however, “there was increased risk for progression to dementia in women who were taking hormone therapy which essentially flipped the field on its ahead because until that point everybody thought that estrogen was a protective factor.”
She said although this study found no association with dementia, she still thinks HT has a role to play and that it may just need to be better tailored to individuals.
“If you think about it, we have our tailored cocktail of hormones in our body and who’s to say that my hormones are going to be the same as yours? Why should you and I be put on the same hormone therapy and assume that will give us the same outcome? I think we could do a lot better with customization and calibration of hormones to aid in women’s health.”
Lifetime approach to dementia
Ms. Gong says future dementia risk-reduction strategies should consider sex-specific risk, and consider the reproductive events that took place in women’s lifespans as well as their entire hormone history when assessing dementia risk, to ensure that the strategies are sex sensitive.
Dr. Buckley agrees: “I don’t think we should ever think about dementia in terms of 65 onwards. We know this disease is insidious and it starts very, very early.”
Regarding limitations, the authors noted that it was a retrospective study that included self-reported measures of reproductive factors, which may be inherently subject to recall bias.
A coauthor does consultant work for Amgen, Freeline, and Kirin outside the submitted work. There were no other relevant financial disclosures.
Certain reproductive factors are associated with greater or lower risk of dementia, according to researchers who conducted a large population-based study with UK Biobank data.
Jessica Gong, a PhD candidate at the George Institute for Global Health at University of New South Wales in Australia, and coauthors found a greater dementia risk in women with early and late menarche, women who were younger when they first gave birth, and those who had had a hysterectomy, especially those who had a hysterectomy without concomitant oophorectomy or with a previous oophorectomy.
After controlling for key confounders, the researchers found lower risk of all-cause dementia if women had ever been pregnant, ever had an abortion, had a longer reproductive span, or had later menopause.
Use of oral contraceptive pills was associated with a lower dementia risk, they found.
In this study, there was no evidence that hormone therapy (HT) was associated with dementia risk (hazard ratio, 0.99, 95% confidence interval [0.90-1.09], P =.0828).
The analysis, published online April 5 in PLOS Medicine, comprised 273,240 women and 228,957 men without prevalent dementia.
The authors noted that dementia rates are increasing. Globally, 50 million people live with dementia, and the number is expected to triple by 2050, according to Alzheimer’s Disease International.
“Our study identified certain reproductive factors related to shorter exposure to endogenous estrogen were associated with increased risk of dementia, highlighting the susceptibility in dementia risk pertaining to women,” Ms. Gong told this publication.
Risk comparison of men and women
Men were included in this study to compare the association between number of children fathered and the risk of all-cause dementia, with the association in their female counterparts.
The U-shaped associations between the number of children and dementia risk were similar for both sexes, suggesting that the risk difference in women may not be associated with factors associated with childbearing
“It may be more related to social and behavioral factors in parenthood, rather than biological factors involved in childbearing,” Ms. Gong said.
Compared with those with two children, for those without children, the multiple adjusted HR (95% CI) was 1.18 (1.04, 1.33) (P = .027) for women and 1.10 (0.98-1.23) P = .164) for men.
For those with four or more children, the HR was 1.14 (0.98, 1.33) (P = .132) for women and 1.26 (1.10-1.45) (P = .003) for men.
Rachel Buckley, PhD, assistant professor of neurology with a dual appointment at Brigham and Women’s and Massachusetts General hospitals in Boston, told this publication she found the comparison of dementia risk with number of children in men and women “fascinating.”
She said the argument usually is that if women have had more births, then they have had more estrogen through their body because women get a huge injection of hormones in pregnancy.
“The idea is that the more pregnancies you have the more protected you are. But this study put that on its head, because if men and women are showing increased [dementia] risk in the number of children they have, it suggests there must be something about having the children – not necessarily the circulating hormones – that might be having an impact,” Dr. Buckley said.
“I had never thought to compare the number of children in men. I do find that very interesting,” she said.
As for the lack of a link between HT and dementia risk, in this study she said, she wouldn’t shut the door on that discussion just yet.
She noted the long history of controversy in the field about whether there is a protective factor against dementia for estrogen or whether exposure to estrogen leads to increased risk.
Before the landmark Women’s Health Initiative (WHI) study in the 1990s, she pointed out, there was evidence in many observational studies that women who had longer exposure to estrogen – whether that was earlier age at first period and later age at menopause combined or women had taken hormone therapy at some point, had less risk for dementia.
Dr. Buckley said that in a secondary outcome of WHI, however, “there was increased risk for progression to dementia in women who were taking hormone therapy which essentially flipped the field on its ahead because until that point everybody thought that estrogen was a protective factor.”
She said although this study found no association with dementia, she still thinks HT has a role to play and that it may just need to be better tailored to individuals.
“If you think about it, we have our tailored cocktail of hormones in our body and who’s to say that my hormones are going to be the same as yours? Why should you and I be put on the same hormone therapy and assume that will give us the same outcome? I think we could do a lot better with customization and calibration of hormones to aid in women’s health.”
Lifetime approach to dementia
Ms. Gong says future dementia risk-reduction strategies should consider sex-specific risk, and consider the reproductive events that took place in women’s lifespans as well as their entire hormone history when assessing dementia risk, to ensure that the strategies are sex sensitive.
Dr. Buckley agrees: “I don’t think we should ever think about dementia in terms of 65 onwards. We know this disease is insidious and it starts very, very early.”
Regarding limitations, the authors noted that it was a retrospective study that included self-reported measures of reproductive factors, which may be inherently subject to recall bias.
A coauthor does consultant work for Amgen, Freeline, and Kirin outside the submitted work. There were no other relevant financial disclosures.
FROM PLOS MEDICINE
Novel drug significantly reduces tics in Tourette syndrome – without side effects
, a new study shows.
Importantly, unlike current medications for the disorder, ecocipam does not lead to weight gain, anxiety, depression, or tardive dyskinesia, compared with placebo – a factor that may lead to better adherence.
For clinicians treating children with Tourette syndrome, the results suggest “help is on the way,” said study investigator Donald Gilbert, MD, professor of pediatrics and neurology, University of Cincinnati Children’s Hospital Medical Center.
“There may be a drug available with a new mechanism of action that is effective to suppress tics without causing weight gain or unwanted neuropsychiatric side effects,” Dr. Gilbert said.
The findings will be presented at the 2022 annual meeting of the American Academy of Neurology.
First-in-class agent
Tourette syndrome is a neurologic condition that causes sudden repetitive involuntary muscle movements and sounds known as tics. These movements typically develop in childhood and worsen during adolescence.
“There’s a risk of injury, such as to the neck, with tics in childhood, so it’s good to have something that makes tics less severe and less socially impairing in junior high,” said Dr. Gilbert.
While tics generally diminish by adulthood, “about 10% of the patients we see as kids persist into adulthood enough to need medical interventions,” said Dr. Gilbert.
Ecopipam is a first-in-class selective D1 receptor antagonist in clinical development for pediatric patients with Tourette syndrome. The compound was previously tested without success in schizophrenia and in obesity, the idea being that because dopamine is linked to pleasure or reward, blocking it might result in weight loss, said Dr. Gilbert.
However, earlier studies in Tourette syndrome suggested that ecopipam reduces tics in children and adults and had low metabolic and movement-related adverse effects.
Drugs currently used to treat tics include haloperidol, pimocide, and aripiprazole. All of these agents block the dopamine-2 (D2) receptor and can cause weight gain and tardive dyskinesia, said Dr. Gilbert.
Placebo-controlled trial
The new study included 149 patients with Tourette syndrome who had a score of at least 20 on the Yale Global Tic Severity Total Tic Score (YGTSS-TTS). The scale measures five aspects of motor and vocal tics: the number, frequency, intensity, complexity, and interference.
With that scale, intensity assesses whether tics cause injury, complexity evaluates the number of muscle group, and interference assesses whether tics interrupt functions, such as speaking or walking.
For each of the five areas, scores range from 0-5, with higher scores indicating greater severity. The motor and vocal parts have a maximum of 25 points each, for a maximum total of 50.
Participants were randomly assigned to receive once-daily oral ecopipam or placebo. A 4-week titration period was followed by an 8-week maintenance period and then a 1-week tapering period.
The primary endpoint was mean change from baseline to week 12 in scores on the YGTSS-TTS.
Results on the YGTSS-TTS showed a significant improvement in the ecopipam group, compared with placebo groups (least square [LS] mean difference: -3.44; 95% confidence interval: -6.09 to -0.79; P = .011).
The analysis indicated a 30% reduction, with an effect size of 0.48, which is “pretty good,” said Dr. Gilbert. “The amount of change is comparable to other drugs that are marketed” to treat tics.
The drug was effective for younger as well as older children. Among those aged 6-11 years, the LS mean difference was -4.95 (95% CI: -9.99 to 0.10; P = .054), and for those aged 12 to 17 years, the LS mean difference was -3.37 (95% CI: -6.51 to -0.24; P = .035).
A key secondary endpoint was the score on the Clinical Global Impression of Tourette Syndrome Severity, which Dr. Gilbert said is a more subjective measure of whether a patient’s life has improved. Here, the mean change at week 12 was significant (P = .001) for the treated group (improvement of 0.91 points), compared with the placebo group (improvement of 0.5 points).
Researchers also assessed safety and tolerability. Treatment-related adverse events (AEs) occurred in 34% of patients taking ecopipam and in 21% of those taking placebo. The most common AEs were headache (9.2%), fatigue (6.6%), somnolence (6.6%), and restlessness (5.3%).
There were no metabolic or movement-related AEs or treatment-related serious AEs.
“This drug doesn’t cause weight gain at all,” said Dr. Gilbert. He noted that there was also no difference in the groups in terms of rates of depression, anxiety, or tardive dyskinesia.
Significant tic reduction
Commenting on the findings, Jessica Frey, MD, a movement disorders fellow at the University of Florida, said the new double-blind, placebo-controlled study “is promising” in that it demonstrates significant tic reduction, compared with placebo without significant side effects.
“Ecopipam could potentially expand pharmacologic treatment options for children and adolescents with Tourette syndrome in the near future,” she said.
Dr. Frey will also be presenting results at the meeting of a study showing a significant correlation between tic severity and social media use among adolescents with Tourette syndrome during the COVID pandemic.
She noted that dopamine is an important neurotransmitter in the underlying pathophysiology of Tourette syndrome. In addition, although D2 receptor blockade can provide significant tic reduction, the “intolerable” side effects often linked to medications with this mechanism “can lead to discontinuation,” said Dr. Frey.
She also noted that ecopipam has previously been evaluated in an open-label study and a follow-up placebo-controlled study that demonstrated safety as well as significant tic reduction.
The study was supported by Emalex Biosciences. Dr. Gilbert and Dr. Frey report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a new study shows.
Importantly, unlike current medications for the disorder, ecocipam does not lead to weight gain, anxiety, depression, or tardive dyskinesia, compared with placebo – a factor that may lead to better adherence.
For clinicians treating children with Tourette syndrome, the results suggest “help is on the way,” said study investigator Donald Gilbert, MD, professor of pediatrics and neurology, University of Cincinnati Children’s Hospital Medical Center.
“There may be a drug available with a new mechanism of action that is effective to suppress tics without causing weight gain or unwanted neuropsychiatric side effects,” Dr. Gilbert said.
The findings will be presented at the 2022 annual meeting of the American Academy of Neurology.
First-in-class agent
Tourette syndrome is a neurologic condition that causes sudden repetitive involuntary muscle movements and sounds known as tics. These movements typically develop in childhood and worsen during adolescence.
“There’s a risk of injury, such as to the neck, with tics in childhood, so it’s good to have something that makes tics less severe and less socially impairing in junior high,” said Dr. Gilbert.
While tics generally diminish by adulthood, “about 10% of the patients we see as kids persist into adulthood enough to need medical interventions,” said Dr. Gilbert.
Ecopipam is a first-in-class selective D1 receptor antagonist in clinical development for pediatric patients with Tourette syndrome. The compound was previously tested without success in schizophrenia and in obesity, the idea being that because dopamine is linked to pleasure or reward, blocking it might result in weight loss, said Dr. Gilbert.
However, earlier studies in Tourette syndrome suggested that ecopipam reduces tics in children and adults and had low metabolic and movement-related adverse effects.
Drugs currently used to treat tics include haloperidol, pimocide, and aripiprazole. All of these agents block the dopamine-2 (D2) receptor and can cause weight gain and tardive dyskinesia, said Dr. Gilbert.
Placebo-controlled trial
The new study included 149 patients with Tourette syndrome who had a score of at least 20 on the Yale Global Tic Severity Total Tic Score (YGTSS-TTS). The scale measures five aspects of motor and vocal tics: the number, frequency, intensity, complexity, and interference.
With that scale, intensity assesses whether tics cause injury, complexity evaluates the number of muscle group, and interference assesses whether tics interrupt functions, such as speaking or walking.
For each of the five areas, scores range from 0-5, with higher scores indicating greater severity. The motor and vocal parts have a maximum of 25 points each, for a maximum total of 50.
Participants were randomly assigned to receive once-daily oral ecopipam or placebo. A 4-week titration period was followed by an 8-week maintenance period and then a 1-week tapering period.
The primary endpoint was mean change from baseline to week 12 in scores on the YGTSS-TTS.
Results on the YGTSS-TTS showed a significant improvement in the ecopipam group, compared with placebo groups (least square [LS] mean difference: -3.44; 95% confidence interval: -6.09 to -0.79; P = .011).
The analysis indicated a 30% reduction, with an effect size of 0.48, which is “pretty good,” said Dr. Gilbert. “The amount of change is comparable to other drugs that are marketed” to treat tics.
The drug was effective for younger as well as older children. Among those aged 6-11 years, the LS mean difference was -4.95 (95% CI: -9.99 to 0.10; P = .054), and for those aged 12 to 17 years, the LS mean difference was -3.37 (95% CI: -6.51 to -0.24; P = .035).
A key secondary endpoint was the score on the Clinical Global Impression of Tourette Syndrome Severity, which Dr. Gilbert said is a more subjective measure of whether a patient’s life has improved. Here, the mean change at week 12 was significant (P = .001) for the treated group (improvement of 0.91 points), compared with the placebo group (improvement of 0.5 points).
Researchers also assessed safety and tolerability. Treatment-related adverse events (AEs) occurred in 34% of patients taking ecopipam and in 21% of those taking placebo. The most common AEs were headache (9.2%), fatigue (6.6%), somnolence (6.6%), and restlessness (5.3%).
There were no metabolic or movement-related AEs or treatment-related serious AEs.
“This drug doesn’t cause weight gain at all,” said Dr. Gilbert. He noted that there was also no difference in the groups in terms of rates of depression, anxiety, or tardive dyskinesia.
Significant tic reduction
Commenting on the findings, Jessica Frey, MD, a movement disorders fellow at the University of Florida, said the new double-blind, placebo-controlled study “is promising” in that it demonstrates significant tic reduction, compared with placebo without significant side effects.
“Ecopipam could potentially expand pharmacologic treatment options for children and adolescents with Tourette syndrome in the near future,” she said.
Dr. Frey will also be presenting results at the meeting of a study showing a significant correlation between tic severity and social media use among adolescents with Tourette syndrome during the COVID pandemic.
She noted that dopamine is an important neurotransmitter in the underlying pathophysiology of Tourette syndrome. In addition, although D2 receptor blockade can provide significant tic reduction, the “intolerable” side effects often linked to medications with this mechanism “can lead to discontinuation,” said Dr. Frey.
She also noted that ecopipam has previously been evaluated in an open-label study and a follow-up placebo-controlled study that demonstrated safety as well as significant tic reduction.
The study was supported by Emalex Biosciences. Dr. Gilbert and Dr. Frey report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a new study shows.
Importantly, unlike current medications for the disorder, ecocipam does not lead to weight gain, anxiety, depression, or tardive dyskinesia, compared with placebo – a factor that may lead to better adherence.
For clinicians treating children with Tourette syndrome, the results suggest “help is on the way,” said study investigator Donald Gilbert, MD, professor of pediatrics and neurology, University of Cincinnati Children’s Hospital Medical Center.
“There may be a drug available with a new mechanism of action that is effective to suppress tics without causing weight gain or unwanted neuropsychiatric side effects,” Dr. Gilbert said.
The findings will be presented at the 2022 annual meeting of the American Academy of Neurology.
First-in-class agent
Tourette syndrome is a neurologic condition that causes sudden repetitive involuntary muscle movements and sounds known as tics. These movements typically develop in childhood and worsen during adolescence.
“There’s a risk of injury, such as to the neck, with tics in childhood, so it’s good to have something that makes tics less severe and less socially impairing in junior high,” said Dr. Gilbert.
While tics generally diminish by adulthood, “about 10% of the patients we see as kids persist into adulthood enough to need medical interventions,” said Dr. Gilbert.
Ecopipam is a first-in-class selective D1 receptor antagonist in clinical development for pediatric patients with Tourette syndrome. The compound was previously tested without success in schizophrenia and in obesity, the idea being that because dopamine is linked to pleasure or reward, blocking it might result in weight loss, said Dr. Gilbert.
However, earlier studies in Tourette syndrome suggested that ecopipam reduces tics in children and adults and had low metabolic and movement-related adverse effects.
Drugs currently used to treat tics include haloperidol, pimocide, and aripiprazole. All of these agents block the dopamine-2 (D2) receptor and can cause weight gain and tardive dyskinesia, said Dr. Gilbert.
Placebo-controlled trial
The new study included 149 patients with Tourette syndrome who had a score of at least 20 on the Yale Global Tic Severity Total Tic Score (YGTSS-TTS). The scale measures five aspects of motor and vocal tics: the number, frequency, intensity, complexity, and interference.
With that scale, intensity assesses whether tics cause injury, complexity evaluates the number of muscle group, and interference assesses whether tics interrupt functions, such as speaking or walking.
For each of the five areas, scores range from 0-5, with higher scores indicating greater severity. The motor and vocal parts have a maximum of 25 points each, for a maximum total of 50.
Participants were randomly assigned to receive once-daily oral ecopipam or placebo. A 4-week titration period was followed by an 8-week maintenance period and then a 1-week tapering period.
The primary endpoint was mean change from baseline to week 12 in scores on the YGTSS-TTS.
Results on the YGTSS-TTS showed a significant improvement in the ecopipam group, compared with placebo groups (least square [LS] mean difference: -3.44; 95% confidence interval: -6.09 to -0.79; P = .011).
The analysis indicated a 30% reduction, with an effect size of 0.48, which is “pretty good,” said Dr. Gilbert. “The amount of change is comparable to other drugs that are marketed” to treat tics.
The drug was effective for younger as well as older children. Among those aged 6-11 years, the LS mean difference was -4.95 (95% CI: -9.99 to 0.10; P = .054), and for those aged 12 to 17 years, the LS mean difference was -3.37 (95% CI: -6.51 to -0.24; P = .035).
A key secondary endpoint was the score on the Clinical Global Impression of Tourette Syndrome Severity, which Dr. Gilbert said is a more subjective measure of whether a patient’s life has improved. Here, the mean change at week 12 was significant (P = .001) for the treated group (improvement of 0.91 points), compared with the placebo group (improvement of 0.5 points).
Researchers also assessed safety and tolerability. Treatment-related adverse events (AEs) occurred in 34% of patients taking ecopipam and in 21% of those taking placebo. The most common AEs were headache (9.2%), fatigue (6.6%), somnolence (6.6%), and restlessness (5.3%).
There were no metabolic or movement-related AEs or treatment-related serious AEs.
“This drug doesn’t cause weight gain at all,” said Dr. Gilbert. He noted that there was also no difference in the groups in terms of rates of depression, anxiety, or tardive dyskinesia.
Significant tic reduction
Commenting on the findings, Jessica Frey, MD, a movement disorders fellow at the University of Florida, said the new double-blind, placebo-controlled study “is promising” in that it demonstrates significant tic reduction, compared with placebo without significant side effects.
“Ecopipam could potentially expand pharmacologic treatment options for children and adolescents with Tourette syndrome in the near future,” she said.
Dr. Frey will also be presenting results at the meeting of a study showing a significant correlation between tic severity and social media use among adolescents with Tourette syndrome during the COVID pandemic.
She noted that dopamine is an important neurotransmitter in the underlying pathophysiology of Tourette syndrome. In addition, although D2 receptor blockade can provide significant tic reduction, the “intolerable” side effects often linked to medications with this mechanism “can lead to discontinuation,” said Dr. Frey.
She also noted that ecopipam has previously been evaluated in an open-label study and a follow-up placebo-controlled study that demonstrated safety as well as significant tic reduction.
The study was supported by Emalex Biosciences. Dr. Gilbert and Dr. Frey report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAN 2022
More evidence that COVID ‘brain fog’ is biologically based
Researchers found elevated levels of CSF immune activation and immunovascular markers in individuals with cognitive postacute sequelae of SARS-CoV-2 infection (PASC). Patients whose cognitive symptoms developed during the acute phase of COVID-19 had the highest levels of brain inflammation.
The findings add to a growing body of evidence that suggests the condition often referred to as “brain fog” has a neurologic basis, said lead author Joanna Hellmuth, MD, MHS, assistant professor of neurology at the University of California, San Francisco Weill Institute of Neurosciences and the UCSF Memory and Aging Center.
The findings will be presented at the 2022 annual meeting of the American Academy of Neurology.
Inflammatory response
There are no effective diagnostic tests or treatments for cognitive PASC, which prompted the investigators to study inflammation in patients with the condition. Initial findings were reported earlier in 2022, which showed abnormalities in the CSF in 77% of patients with cognitive impairment. Patients without cognitive impairments had normal CSF.
Extending that work in this new study, researchers studied patients from the Long-term Impact of Infection With Novel Coronavirus (LIINC) study with confirmed SARS-CoV-2 infection who were not hospitalized. They conducted 2-hour neurocognitive interviews and identified 23 people with new, persistent cognitive symptoms (cognitive PASC) and 10 with no cognitive symptoms who served as controls.
All participants underwent additional neurologic examination and neuropsychological testing, and half agreed to a lumbar puncture to allow researchers to collect CSF samples. The CSF was collected a median of 10.2 months after initial COVID symptoms began.
Participants with cognitive PASC had higher median levels of CSF acute phase reactants C-reactive protein (0.007 mg/L vs. 0.000 mg/L; P =.004) and serum amyloid A (0.001 mg/L vs. 0.000 mg/L; P = .001), compared with COVID controls.
The PASC group also had elevated levels of CSF immune activation markers interferon gamma–inducible protein (IP-10), interleukin-8, and immunovascular markers vascular endothelial growth factor-C and VEGFR-1, although the differences with the control group were not statistically significant.
The timing of the onset of cognitive problems was also associated with higher levels of immune activation and immunovascular markers. Patients with brain fog that developed during the acute phase of COVID-19 had higher levels of CSF VEGF-C, compared with patients whose cognitive symptoms developed more than a month after initial COVID symptoms (173 pg/mL vs. 99 pg/mL; P = .048) and COVID controls (79 pg/mL; P = .048).
Acute onset cognitive PASC participants had higher CSF levels of IP-10 (P = .030), IL-8 (P = .048), placental growth factor (P = .030) and intercellular adhesion molecule-1 (P = .045), compared with COVID controls.
Researchers believe these new findings could mean that intrathecal immune activation and endothelial activation/dysfunction may contribute to cognitive PASC and that the mechanisms involved may be different in patients with acute cognitive PASC versus those with delayed onset.
“Our data suggests that perhaps in these people with more acute cognitive changes they don’t have the return to homeostasis,” Dr. Hellmuth said, while patients with delayed onset cognitive PASC had levels more in line with COVID patients who had no cognitive issues.
Moving the needle forward
Commenting on the findings, William Schaffner, MD, professor of infectious diseases, Vanderbilt University Medical Center, Nashville, Tenn., said that, while the study doesn’t rule out a possible psychological basis for cognitive PASC, it adds more weight to the biological argument.
“When you have nonspecific symptoms for which specific tests are unavailable,” Dr. Schaffner explained, “there is a natural question that always comes up: Is this principally a biologically induced phenomenon or psychological? This moves the needle substantially in the direction of a biological phenomenon.”
Another important element to the study, Dr. Schaffner said, is that the patients involved had mild COVID.
“Not every patient with long COVID symptoms had been hospitalized with severe disease,” he said. “There are inflammatory phenomenon in various organ systems such that even if the inflammatory response in the lung was not severe enough to get you into the hospital, there were inflammatory responses in other organ systems that could persist once the acute infection resolved.”
Although the small size of the study is a limitation, Dr. Schaffner said that shouldn’t minimize the importance of these findings.
“That it’s small doesn’t diminish its value,” he said. “The next step forward might be to try to associate the markers more specifically with COVID. The more precise we can be, the more convincing the story will become.”
The study was funded by the National Institutes of Health. Dr. Hellmuth received grant support from the National Institutes of Health/National Institute of Mental Health supporting this work and personal fees for medical-legal consultation outside of the submitted work. Dr. Schaffner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Researchers found elevated levels of CSF immune activation and immunovascular markers in individuals with cognitive postacute sequelae of SARS-CoV-2 infection (PASC). Patients whose cognitive symptoms developed during the acute phase of COVID-19 had the highest levels of brain inflammation.
The findings add to a growing body of evidence that suggests the condition often referred to as “brain fog” has a neurologic basis, said lead author Joanna Hellmuth, MD, MHS, assistant professor of neurology at the University of California, San Francisco Weill Institute of Neurosciences and the UCSF Memory and Aging Center.
The findings will be presented at the 2022 annual meeting of the American Academy of Neurology.
Inflammatory response
There are no effective diagnostic tests or treatments for cognitive PASC, which prompted the investigators to study inflammation in patients with the condition. Initial findings were reported earlier in 2022, which showed abnormalities in the CSF in 77% of patients with cognitive impairment. Patients without cognitive impairments had normal CSF.
Extending that work in this new study, researchers studied patients from the Long-term Impact of Infection With Novel Coronavirus (LIINC) study with confirmed SARS-CoV-2 infection who were not hospitalized. They conducted 2-hour neurocognitive interviews and identified 23 people with new, persistent cognitive symptoms (cognitive PASC) and 10 with no cognitive symptoms who served as controls.
All participants underwent additional neurologic examination and neuropsychological testing, and half agreed to a lumbar puncture to allow researchers to collect CSF samples. The CSF was collected a median of 10.2 months after initial COVID symptoms began.
Participants with cognitive PASC had higher median levels of CSF acute phase reactants C-reactive protein (0.007 mg/L vs. 0.000 mg/L; P =.004) and serum amyloid A (0.001 mg/L vs. 0.000 mg/L; P = .001), compared with COVID controls.
The PASC group also had elevated levels of CSF immune activation markers interferon gamma–inducible protein (IP-10), interleukin-8, and immunovascular markers vascular endothelial growth factor-C and VEGFR-1, although the differences with the control group were not statistically significant.
The timing of the onset of cognitive problems was also associated with higher levels of immune activation and immunovascular markers. Patients with brain fog that developed during the acute phase of COVID-19 had higher levels of CSF VEGF-C, compared with patients whose cognitive symptoms developed more than a month after initial COVID symptoms (173 pg/mL vs. 99 pg/mL; P = .048) and COVID controls (79 pg/mL; P = .048).
Acute onset cognitive PASC participants had higher CSF levels of IP-10 (P = .030), IL-8 (P = .048), placental growth factor (P = .030) and intercellular adhesion molecule-1 (P = .045), compared with COVID controls.
Researchers believe these new findings could mean that intrathecal immune activation and endothelial activation/dysfunction may contribute to cognitive PASC and that the mechanisms involved may be different in patients with acute cognitive PASC versus those with delayed onset.
“Our data suggests that perhaps in these people with more acute cognitive changes they don’t have the return to homeostasis,” Dr. Hellmuth said, while patients with delayed onset cognitive PASC had levels more in line with COVID patients who had no cognitive issues.
Moving the needle forward
Commenting on the findings, William Schaffner, MD, professor of infectious diseases, Vanderbilt University Medical Center, Nashville, Tenn., said that, while the study doesn’t rule out a possible psychological basis for cognitive PASC, it adds more weight to the biological argument.
“When you have nonspecific symptoms for which specific tests are unavailable,” Dr. Schaffner explained, “there is a natural question that always comes up: Is this principally a biologically induced phenomenon or psychological? This moves the needle substantially in the direction of a biological phenomenon.”
Another important element to the study, Dr. Schaffner said, is that the patients involved had mild COVID.
“Not every patient with long COVID symptoms had been hospitalized with severe disease,” he said. “There are inflammatory phenomenon in various organ systems such that even if the inflammatory response in the lung was not severe enough to get you into the hospital, there were inflammatory responses in other organ systems that could persist once the acute infection resolved.”
Although the small size of the study is a limitation, Dr. Schaffner said that shouldn’t minimize the importance of these findings.
“That it’s small doesn’t diminish its value,” he said. “The next step forward might be to try to associate the markers more specifically with COVID. The more precise we can be, the more convincing the story will become.”
The study was funded by the National Institutes of Health. Dr. Hellmuth received grant support from the National Institutes of Health/National Institute of Mental Health supporting this work and personal fees for medical-legal consultation outside of the submitted work. Dr. Schaffner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Researchers found elevated levels of CSF immune activation and immunovascular markers in individuals with cognitive postacute sequelae of SARS-CoV-2 infection (PASC). Patients whose cognitive symptoms developed during the acute phase of COVID-19 had the highest levels of brain inflammation.
The findings add to a growing body of evidence that suggests the condition often referred to as “brain fog” has a neurologic basis, said lead author Joanna Hellmuth, MD, MHS, assistant professor of neurology at the University of California, San Francisco Weill Institute of Neurosciences and the UCSF Memory and Aging Center.
The findings will be presented at the 2022 annual meeting of the American Academy of Neurology.
Inflammatory response
There are no effective diagnostic tests or treatments for cognitive PASC, which prompted the investigators to study inflammation in patients with the condition. Initial findings were reported earlier in 2022, which showed abnormalities in the CSF in 77% of patients with cognitive impairment. Patients without cognitive impairments had normal CSF.
Extending that work in this new study, researchers studied patients from the Long-term Impact of Infection With Novel Coronavirus (LIINC) study with confirmed SARS-CoV-2 infection who were not hospitalized. They conducted 2-hour neurocognitive interviews and identified 23 people with new, persistent cognitive symptoms (cognitive PASC) and 10 with no cognitive symptoms who served as controls.
All participants underwent additional neurologic examination and neuropsychological testing, and half agreed to a lumbar puncture to allow researchers to collect CSF samples. The CSF was collected a median of 10.2 months after initial COVID symptoms began.
Participants with cognitive PASC had higher median levels of CSF acute phase reactants C-reactive protein (0.007 mg/L vs. 0.000 mg/L; P =.004) and serum amyloid A (0.001 mg/L vs. 0.000 mg/L; P = .001), compared with COVID controls.
The PASC group also had elevated levels of CSF immune activation markers interferon gamma–inducible protein (IP-10), interleukin-8, and immunovascular markers vascular endothelial growth factor-C and VEGFR-1, although the differences with the control group were not statistically significant.
The timing of the onset of cognitive problems was also associated with higher levels of immune activation and immunovascular markers. Patients with brain fog that developed during the acute phase of COVID-19 had higher levels of CSF VEGF-C, compared with patients whose cognitive symptoms developed more than a month after initial COVID symptoms (173 pg/mL vs. 99 pg/mL; P = .048) and COVID controls (79 pg/mL; P = .048).
Acute onset cognitive PASC participants had higher CSF levels of IP-10 (P = .030), IL-8 (P = .048), placental growth factor (P = .030) and intercellular adhesion molecule-1 (P = .045), compared with COVID controls.
Researchers believe these new findings could mean that intrathecal immune activation and endothelial activation/dysfunction may contribute to cognitive PASC and that the mechanisms involved may be different in patients with acute cognitive PASC versus those with delayed onset.
“Our data suggests that perhaps in these people with more acute cognitive changes they don’t have the return to homeostasis,” Dr. Hellmuth said, while patients with delayed onset cognitive PASC had levels more in line with COVID patients who had no cognitive issues.
Moving the needle forward
Commenting on the findings, William Schaffner, MD, professor of infectious diseases, Vanderbilt University Medical Center, Nashville, Tenn., said that, while the study doesn’t rule out a possible psychological basis for cognitive PASC, it adds more weight to the biological argument.
“When you have nonspecific symptoms for which specific tests are unavailable,” Dr. Schaffner explained, “there is a natural question that always comes up: Is this principally a biologically induced phenomenon or psychological? This moves the needle substantially in the direction of a biological phenomenon.”
Another important element to the study, Dr. Schaffner said, is that the patients involved had mild COVID.
“Not every patient with long COVID symptoms had been hospitalized with severe disease,” he said. “There are inflammatory phenomenon in various organ systems such that even if the inflammatory response in the lung was not severe enough to get you into the hospital, there were inflammatory responses in other organ systems that could persist once the acute infection resolved.”
Although the small size of the study is a limitation, Dr. Schaffner said that shouldn’t minimize the importance of these findings.
“That it’s small doesn’t diminish its value,” he said. “The next step forward might be to try to associate the markers more specifically with COVID. The more precise we can be, the more convincing the story will become.”
The study was funded by the National Institutes of Health. Dr. Hellmuth received grant support from the National Institutes of Health/National Institute of Mental Health supporting this work and personal fees for medical-legal consultation outside of the submitted work. Dr. Schaffner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAN 2022
Atypical anxiety offers intervention target in Parkinson’s disease
Anxiety is common in Parkinson’s disease (PD) and has been shown to increase functional disability and decrease quality of life, but atypical presentations of anxiety are underrecognized and often undertreated in PD patients, wrote Nadeeka N. Dissanayaka, PhD, of the University of Queensland, Brisbane, Australia, and colleagues.
In a study published in the American Journal of Geriatric Psychiatry , the researchers conducted a systematic review of 60 studies to better characterize atypical PD-related anxiety. Fourteen studies involved Anxiety Not Otherwise Specified (NOS), 31 included fluctuating anxiety symptoms, and 22 included Fear of Falling (FOF).
Overall, the average prevalence rate for anxiety disorders in the PD population was 31%.
Anxiety NOS, fluctuating anxiety, and FOF accounted for a weighted mean prevalence of 14.9%, 34.19%, and 51.5%, respectively.
The symptomatology of anxiety NOS included psychological distress about the PD diagnosis, insecurity about the future, fear of losing control of motor and bodily functions, and social embarrassment. Clinically, anxiety NOS was associated with a range of factors including minor depression, on-off motor symptoms, muscle cramps, poor quality of life, and gait impairment.
The symptomatology of fluctuating anxiety was assessed in 9 studies of the “on” motor state and 16 studies of both “on” and “off.” Symptoms associated with the off state included panic attacks, feeling anxious or sad, and avoiding situations, as well as palpitations, dizziness, chills, and hot flashes.
Clinically, studies showed that anxiety was more severe in the off-medication state, and symptoms were reduced in the on state. Data from some studies showed that fluctuating anxiety was more common in PD patients who were female, and who had a younger age of PD onset and longer disease duration.
The symptomatology of FOF included associations between FOF and difficulty with walking and gait: Using a walker or other device, more frequent freezing in place, hesitation when turning, and slower speed while walking. Clinically, characteristics associated with FOF included older age, needing assistance for activities of daily living, a history of falls, and reduced quality of life.
The results of the review were limited by several factors including the varying assessment techniques, and the lack of data on treatment for atypical anxiety in PD, the researchers noted. “To our knowledge there are no treatment trials focused on Anxiety NOS,” and studies on the treatment of fluctuating anxiety and FOF are preliminary, they said.
However, the results support the need for early identification and classification of PD-related anxiety to improve treatment strategies and long-term outcomes, the researchers concluded. In the absence of evidence-based treatment strategies, “Given the heterogeneity of anxiety presentations in PD, the importance of tailoring interventions to meet the specific needs and unique symptom profiles of each individual cannot be overstated,” and routine screening of PD patients for anxiety every 6-12 months is recommended, they emphasized.
Dr. Dissanayaka disclosed support from the National Health and Medical Research Boosting Dementia Research Leadership Fellowship.
Anxiety is common in Parkinson’s disease (PD) and has been shown to increase functional disability and decrease quality of life, but atypical presentations of anxiety are underrecognized and often undertreated in PD patients, wrote Nadeeka N. Dissanayaka, PhD, of the University of Queensland, Brisbane, Australia, and colleagues.
In a study published in the American Journal of Geriatric Psychiatry , the researchers conducted a systematic review of 60 studies to better characterize atypical PD-related anxiety. Fourteen studies involved Anxiety Not Otherwise Specified (NOS), 31 included fluctuating anxiety symptoms, and 22 included Fear of Falling (FOF).
Overall, the average prevalence rate for anxiety disorders in the PD population was 31%.
Anxiety NOS, fluctuating anxiety, and FOF accounted for a weighted mean prevalence of 14.9%, 34.19%, and 51.5%, respectively.
The symptomatology of anxiety NOS included psychological distress about the PD diagnosis, insecurity about the future, fear of losing control of motor and bodily functions, and social embarrassment. Clinically, anxiety NOS was associated with a range of factors including minor depression, on-off motor symptoms, muscle cramps, poor quality of life, and gait impairment.
The symptomatology of fluctuating anxiety was assessed in 9 studies of the “on” motor state and 16 studies of both “on” and “off.” Symptoms associated with the off state included panic attacks, feeling anxious or sad, and avoiding situations, as well as palpitations, dizziness, chills, and hot flashes.
Clinically, studies showed that anxiety was more severe in the off-medication state, and symptoms were reduced in the on state. Data from some studies showed that fluctuating anxiety was more common in PD patients who were female, and who had a younger age of PD onset and longer disease duration.
The symptomatology of FOF included associations between FOF and difficulty with walking and gait: Using a walker or other device, more frequent freezing in place, hesitation when turning, and slower speed while walking. Clinically, characteristics associated with FOF included older age, needing assistance for activities of daily living, a history of falls, and reduced quality of life.
The results of the review were limited by several factors including the varying assessment techniques, and the lack of data on treatment for atypical anxiety in PD, the researchers noted. “To our knowledge there are no treatment trials focused on Anxiety NOS,” and studies on the treatment of fluctuating anxiety and FOF are preliminary, they said.
However, the results support the need for early identification and classification of PD-related anxiety to improve treatment strategies and long-term outcomes, the researchers concluded. In the absence of evidence-based treatment strategies, “Given the heterogeneity of anxiety presentations in PD, the importance of tailoring interventions to meet the specific needs and unique symptom profiles of each individual cannot be overstated,” and routine screening of PD patients for anxiety every 6-12 months is recommended, they emphasized.
Dr. Dissanayaka disclosed support from the National Health and Medical Research Boosting Dementia Research Leadership Fellowship.
Anxiety is common in Parkinson’s disease (PD) and has been shown to increase functional disability and decrease quality of life, but atypical presentations of anxiety are underrecognized and often undertreated in PD patients, wrote Nadeeka N. Dissanayaka, PhD, of the University of Queensland, Brisbane, Australia, and colleagues.
In a study published in the American Journal of Geriatric Psychiatry , the researchers conducted a systematic review of 60 studies to better characterize atypical PD-related anxiety. Fourteen studies involved Anxiety Not Otherwise Specified (NOS), 31 included fluctuating anxiety symptoms, and 22 included Fear of Falling (FOF).
Overall, the average prevalence rate for anxiety disorders in the PD population was 31%.
Anxiety NOS, fluctuating anxiety, and FOF accounted for a weighted mean prevalence of 14.9%, 34.19%, and 51.5%, respectively.
The symptomatology of anxiety NOS included psychological distress about the PD diagnosis, insecurity about the future, fear of losing control of motor and bodily functions, and social embarrassment. Clinically, anxiety NOS was associated with a range of factors including minor depression, on-off motor symptoms, muscle cramps, poor quality of life, and gait impairment.
The symptomatology of fluctuating anxiety was assessed in 9 studies of the “on” motor state and 16 studies of both “on” and “off.” Symptoms associated with the off state included panic attacks, feeling anxious or sad, and avoiding situations, as well as palpitations, dizziness, chills, and hot flashes.
Clinically, studies showed that anxiety was more severe in the off-medication state, and symptoms were reduced in the on state. Data from some studies showed that fluctuating anxiety was more common in PD patients who were female, and who had a younger age of PD onset and longer disease duration.
The symptomatology of FOF included associations between FOF and difficulty with walking and gait: Using a walker or other device, more frequent freezing in place, hesitation when turning, and slower speed while walking. Clinically, characteristics associated with FOF included older age, needing assistance for activities of daily living, a history of falls, and reduced quality of life.
The results of the review were limited by several factors including the varying assessment techniques, and the lack of data on treatment for atypical anxiety in PD, the researchers noted. “To our knowledge there are no treatment trials focused on Anxiety NOS,” and studies on the treatment of fluctuating anxiety and FOF are preliminary, they said.
However, the results support the need for early identification and classification of PD-related anxiety to improve treatment strategies and long-term outcomes, the researchers concluded. In the absence of evidence-based treatment strategies, “Given the heterogeneity of anxiety presentations in PD, the importance of tailoring interventions to meet the specific needs and unique symptom profiles of each individual cannot be overstated,” and routine screening of PD patients for anxiety every 6-12 months is recommended, they emphasized.
Dr. Dissanayaka disclosed support from the National Health and Medical Research Boosting Dementia Research Leadership Fellowship.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY
Psychotropic med use tied to ‘striking’ post-COVID dementia risk
, new research suggests.
Results from a large study of more than 1,700 patients who had been hospitalized with COVID showed a greater than twofold increased risk for post-COVID dementia in those taking antipsychotics and mood stabilizers/anticonvulsants – medications often used to treat schizophrenia, psychosis, bipolar disorder, and seizures.
“We know that pre-existing psychiatric illness is associated with poor COVID-19 outcomes, but our study is the first to show an association with certain psychiatric medications and dementia,” co-investigator Liron Sinvani, MD, the Feinstein Institutes for Medical Research, Manhasset, New York, said in an interview.
“Our study highlights the potential interaction between baseline neuropsychiatric disease, psychotropic medications, COVID-19, and dementia,” Dr. Sinvani added.
The findings were published online March 18 in Frontiers in Medicine.
‘Striking’ dementia rate
Using electronic health records, the researchers evaluated pre-COVID psychotropic medication use and post-COVID dementia onset in 1,755 adults aged 65 and older. All were hospitalized with COVID-19 at Northwell Health between March 1 and April 20, 2020.
A “striking” 13% of the participants (n = 223) developed dementia within 1-year of follow-up, the investigators report.
Among the 438 patients (25%) exposed to at least one psychotropic medication before COVID-19, 105 (24%) developed dementia in the year following COVID versus 118 of 1,317 (9%) patients with no pre-COVID exposure to psychotropic medication (odds ratio, 3.2; 95% confidence interval, 2.37-4.32).
Both pre-COVID psychotropic medication use (OR, 2.7; 95% CI, 1.8-4.0, P < .001) and delirium (OR, 3.0; 95% CI, 1.9-4.6, P < .001) were significantly associated with post-COVID dementia at 1 year.
In a sensitivity analysis in the subset of 423 patients with at least one documented neurologic or psychiatric diagnosis at the time of COVID admission, and after adjusting for confounding factors, pre-COVID psychotropic medication use remained significantly linked to post-COVID dementia onset (OR, 3.09; 95% CI, 1.5-6.6, P = .002).
Drug classes most strongly associated with 1-year post-COVID dementia onset were antipsychotics (OR, 2.8, 95% CI, 1.7-4.4, P < .001) and mood stabilizers/anticonvulsants (OR, 2.4, 95% CI, 1.39-4.02, P = .001).
In a further exploratory analysis, the psychotropics valproic acid (multiple brands) and haloperidol (Haldol) had the largest association with post-COVID dementia.
Antidepressants as a class were not associated with post-COVID dementia, but the potential effects of two commonly prescribed antidepressants in older adults, mirtazapine (Remeron) and escitalopram (Lexapro), “warrant further investigation,” the researchers note.
Predictive risk marker?
“This research shows that psychotropic medications can be considered a predictive risk marker for post-COVID dementia. In patients taking psychotropic medications, COVID-19 could have accelerated progression of dementia after hospitalization,” lead author Yun Freudenberg-Hua, MD, the Feinstein Institutes, said in a news release.
It is unclear why psychotropic medications may raise the risk for dementia onset after COVID, the investigators note.
“It is intuitive that psychotropic medications indicate pre-existing neuropsychiatric conditions in which COVID-19 occurs. It is possible that psychotropic medications may potentiate the neurostructural changes that have been found in the brain of those who have recovered from COVID-19,” they write.
The sensitivity analysis in patients with documented neurologic and psychiatric diagnoses supports this interpretation.
COVID-19 may also accelerate the underlying brain disorders for which psychotropic medications were prescribed, leading to the greater incidence of post-COVID dementia, the researchers write.
“It is important to note that this study is in no way recommending people should stop taking antipsychotics but simply that clinicians need to factor in a patient’s medication history while considering post-COVID aftereffects,” Dr. Freudenberg-Hua said.
“Given that the number of patients with dementia is projected to triple in the next 30 years, these findings have significant public health implications,” Dr. Sinvani added.
She noted that “care partners and health care professionals” should look for early signs of dementia, such as forgetfulness and depressive symptoms, in their patients.
“Future studies must continue to evaluate these associations, which are key for potential future interventions to prevent dementia,” Dr. Sinvani said.
The study was funded by the National Institutes of Health. Dr. Freudenberg-Hua co-owns stock and stock options from Regeneron Pharmaceuticals. Dr. Sinvani has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Results from a large study of more than 1,700 patients who had been hospitalized with COVID showed a greater than twofold increased risk for post-COVID dementia in those taking antipsychotics and mood stabilizers/anticonvulsants – medications often used to treat schizophrenia, psychosis, bipolar disorder, and seizures.
“We know that pre-existing psychiatric illness is associated with poor COVID-19 outcomes, but our study is the first to show an association with certain psychiatric medications and dementia,” co-investigator Liron Sinvani, MD, the Feinstein Institutes for Medical Research, Manhasset, New York, said in an interview.
“Our study highlights the potential interaction between baseline neuropsychiatric disease, psychotropic medications, COVID-19, and dementia,” Dr. Sinvani added.
The findings were published online March 18 in Frontiers in Medicine.
‘Striking’ dementia rate
Using electronic health records, the researchers evaluated pre-COVID psychotropic medication use and post-COVID dementia onset in 1,755 adults aged 65 and older. All were hospitalized with COVID-19 at Northwell Health between March 1 and April 20, 2020.
A “striking” 13% of the participants (n = 223) developed dementia within 1-year of follow-up, the investigators report.
Among the 438 patients (25%) exposed to at least one psychotropic medication before COVID-19, 105 (24%) developed dementia in the year following COVID versus 118 of 1,317 (9%) patients with no pre-COVID exposure to psychotropic medication (odds ratio, 3.2; 95% confidence interval, 2.37-4.32).
Both pre-COVID psychotropic medication use (OR, 2.7; 95% CI, 1.8-4.0, P < .001) and delirium (OR, 3.0; 95% CI, 1.9-4.6, P < .001) were significantly associated with post-COVID dementia at 1 year.
In a sensitivity analysis in the subset of 423 patients with at least one documented neurologic or psychiatric diagnosis at the time of COVID admission, and after adjusting for confounding factors, pre-COVID psychotropic medication use remained significantly linked to post-COVID dementia onset (OR, 3.09; 95% CI, 1.5-6.6, P = .002).
Drug classes most strongly associated with 1-year post-COVID dementia onset were antipsychotics (OR, 2.8, 95% CI, 1.7-4.4, P < .001) and mood stabilizers/anticonvulsants (OR, 2.4, 95% CI, 1.39-4.02, P = .001).
In a further exploratory analysis, the psychotropics valproic acid (multiple brands) and haloperidol (Haldol) had the largest association with post-COVID dementia.
Antidepressants as a class were not associated with post-COVID dementia, but the potential effects of two commonly prescribed antidepressants in older adults, mirtazapine (Remeron) and escitalopram (Lexapro), “warrant further investigation,” the researchers note.
Predictive risk marker?
“This research shows that psychotropic medications can be considered a predictive risk marker for post-COVID dementia. In patients taking psychotropic medications, COVID-19 could have accelerated progression of dementia after hospitalization,” lead author Yun Freudenberg-Hua, MD, the Feinstein Institutes, said in a news release.
It is unclear why psychotropic medications may raise the risk for dementia onset after COVID, the investigators note.
“It is intuitive that psychotropic medications indicate pre-existing neuropsychiatric conditions in which COVID-19 occurs. It is possible that psychotropic medications may potentiate the neurostructural changes that have been found in the brain of those who have recovered from COVID-19,” they write.
The sensitivity analysis in patients with documented neurologic and psychiatric diagnoses supports this interpretation.
COVID-19 may also accelerate the underlying brain disorders for which psychotropic medications were prescribed, leading to the greater incidence of post-COVID dementia, the researchers write.
“It is important to note that this study is in no way recommending people should stop taking antipsychotics but simply that clinicians need to factor in a patient’s medication history while considering post-COVID aftereffects,” Dr. Freudenberg-Hua said.
“Given that the number of patients with dementia is projected to triple in the next 30 years, these findings have significant public health implications,” Dr. Sinvani added.
She noted that “care partners and health care professionals” should look for early signs of dementia, such as forgetfulness and depressive symptoms, in their patients.
“Future studies must continue to evaluate these associations, which are key for potential future interventions to prevent dementia,” Dr. Sinvani said.
The study was funded by the National Institutes of Health. Dr. Freudenberg-Hua co-owns stock and stock options from Regeneron Pharmaceuticals. Dr. Sinvani has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
Results from a large study of more than 1,700 patients who had been hospitalized with COVID showed a greater than twofold increased risk for post-COVID dementia in those taking antipsychotics and mood stabilizers/anticonvulsants – medications often used to treat schizophrenia, psychosis, bipolar disorder, and seizures.
“We know that pre-existing psychiatric illness is associated with poor COVID-19 outcomes, but our study is the first to show an association with certain psychiatric medications and dementia,” co-investigator Liron Sinvani, MD, the Feinstein Institutes for Medical Research, Manhasset, New York, said in an interview.
“Our study highlights the potential interaction between baseline neuropsychiatric disease, psychotropic medications, COVID-19, and dementia,” Dr. Sinvani added.
The findings were published online March 18 in Frontiers in Medicine.
‘Striking’ dementia rate
Using electronic health records, the researchers evaluated pre-COVID psychotropic medication use and post-COVID dementia onset in 1,755 adults aged 65 and older. All were hospitalized with COVID-19 at Northwell Health between March 1 and April 20, 2020.
A “striking” 13% of the participants (n = 223) developed dementia within 1-year of follow-up, the investigators report.
Among the 438 patients (25%) exposed to at least one psychotropic medication before COVID-19, 105 (24%) developed dementia in the year following COVID versus 118 of 1,317 (9%) patients with no pre-COVID exposure to psychotropic medication (odds ratio, 3.2; 95% confidence interval, 2.37-4.32).
Both pre-COVID psychotropic medication use (OR, 2.7; 95% CI, 1.8-4.0, P < .001) and delirium (OR, 3.0; 95% CI, 1.9-4.6, P < .001) were significantly associated with post-COVID dementia at 1 year.
In a sensitivity analysis in the subset of 423 patients with at least one documented neurologic or psychiatric diagnosis at the time of COVID admission, and after adjusting for confounding factors, pre-COVID psychotropic medication use remained significantly linked to post-COVID dementia onset (OR, 3.09; 95% CI, 1.5-6.6, P = .002).
Drug classes most strongly associated with 1-year post-COVID dementia onset were antipsychotics (OR, 2.8, 95% CI, 1.7-4.4, P < .001) and mood stabilizers/anticonvulsants (OR, 2.4, 95% CI, 1.39-4.02, P = .001).
In a further exploratory analysis, the psychotropics valproic acid (multiple brands) and haloperidol (Haldol) had the largest association with post-COVID dementia.
Antidepressants as a class were not associated with post-COVID dementia, but the potential effects of two commonly prescribed antidepressants in older adults, mirtazapine (Remeron) and escitalopram (Lexapro), “warrant further investigation,” the researchers note.
Predictive risk marker?
“This research shows that psychotropic medications can be considered a predictive risk marker for post-COVID dementia. In patients taking psychotropic medications, COVID-19 could have accelerated progression of dementia after hospitalization,” lead author Yun Freudenberg-Hua, MD, the Feinstein Institutes, said in a news release.
It is unclear why psychotropic medications may raise the risk for dementia onset after COVID, the investigators note.
“It is intuitive that psychotropic medications indicate pre-existing neuropsychiatric conditions in which COVID-19 occurs. It is possible that psychotropic medications may potentiate the neurostructural changes that have been found in the brain of those who have recovered from COVID-19,” they write.
The sensitivity analysis in patients with documented neurologic and psychiatric diagnoses supports this interpretation.
COVID-19 may also accelerate the underlying brain disorders for which psychotropic medications were prescribed, leading to the greater incidence of post-COVID dementia, the researchers write.
“It is important to note that this study is in no way recommending people should stop taking antipsychotics but simply that clinicians need to factor in a patient’s medication history while considering post-COVID aftereffects,” Dr. Freudenberg-Hua said.
“Given that the number of patients with dementia is projected to triple in the next 30 years, these findings have significant public health implications,” Dr. Sinvani added.
She noted that “care partners and health care professionals” should look for early signs of dementia, such as forgetfulness and depressive symptoms, in their patients.
“Future studies must continue to evaluate these associations, which are key for potential future interventions to prevent dementia,” Dr. Sinvani said.
The study was funded by the National Institutes of Health. Dr. Freudenberg-Hua co-owns stock and stock options from Regeneron Pharmaceuticals. Dr. Sinvani has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM FRONTIERS IN MEDICINE







