User login
A gruesome murder changes two docs’ lives, and one was the killer
Driving from his home in Asheville, N.C., to his new job at the tiny Cane Creek clinic, Benjamin Gilmer, MD, was eager to start his new life and pay off his medical school debts.
The rural clinic had been forced to close after his predecessor, family physician Vince Gilmer, MD, (no relation) had been convicted of first-degree murder 4 years earlier. He was serving a life sentence in a West Virginia prison without the possibility of parole. He is still behind bars and could not comment on this story.
As the months flew by, Benjamin Gilmer’s patients shared stories about the other Dr. Gilmer that surprised him. They described Vince Gilmer as a caring, generous person who went out of his way to help them. He made house calls, and if a patient couldn’t afford to pay him, he would accept a bushel of corn instead.
Yet there was no doubt about the gruesome murder. Vince Gilmer was convicted of strangling his frail 60-year-old father with a rope in his Toyota truck. He then cut off all his father’s fingers and dumped his father’s body by the side of the road.
“Four years later, his patients were still shocked about what happened and couldn’t reconcile the person they knew with the event that happened,” says Benjamin Gilmer.
Yet, Vince Gilmer had admitted to the killing, and the prosecution had presented evidence at the trial that it was premeditated and that he tried to cover up the crime. The detectives found the “murder” weapons in Vince’s truck: the ropes he strangled his father with and the garden shears that he cut off his fingers with. They also had evidence that he drove to Virginia to dump the body, returned to see patients for several days as if nothing had happened, and then ran away when a detective came to arrest him.
But something kept gnawing away at Benjamin Gilmer.
Little did he know that he would embark on a journey to solve a medical mystery, and then even fight to get the convicted killer out of prison.
Solving a medical mystery
Benjamin Gilmer decided to investigate what might have happened to Vince in the months leading up to the murder. He talked to his friends and found several clues about Vince’s medical history. They recalled that he suffered a concussion in a car accident 6 months before the murder, which suggested he could have had a traumatic brain injury.
Benjamin Gilmer also discovered that Vince’s father was diagnosed with schizophrenia and had been in a residential psychiatric facility in Virginia until he was released that fateful night to Vince’s custody.
Vince had written to friends that “something is wrong with my brain and help me.” He mentioned SSRI discontinuation syndrome because he abruptly stopped taking his medication the week of the murder (which can cause electric shock sensations and mood swings among other symptoms).
Vince had mentioned the SSRI discontinuation syndrome at his trial and that his father had sexually molested him for years and that he tried to molest him again during the ride in his truck. However, the court dismissed that information because Vince represented himself, dismissed his court-appointed attorneys, and lacked expert testimony about his mental state.
The prosecutor portrayed Vince as a lying sociopath who had planned his father’s murder down to the last detail. The judge agreed. Two psychiatrists and a psychologist who later evaluated him in prison concluded that he was faking his symptoms and denied his requests for an SSRI.
Meanwhile, Benjamin Gilmer became increasingly preoccupied with what happened to Vince. “It was hard to erase a memory that had so tainted that community,” he said.
When Sarah Koenig, a journalist and former producer of the radio program This American Life, called Benjamin Gilmer to interview him about the coincidence of taking over Vince Gilmer’s practice and sharing the same last name, he refused. “I was scared and didn’t want to be on his radar, I was afraid of how he might react.”
In spring 2012, he called Koenig and agreed to collaborate on an episode about Vince’s case. Benjamin Gilmer wrote to Vince Gilmer in prison, asking for a meeting. To his surprise, Vince wanted to meet them.
When Vince shuffled into the waiting area at the Wallens Ridge State Prison in West Virginia, Benjamin Gilmer was shocked by his appearance. “He looked like a caged animal, it was very hard for him to string together ideas and express himself, and he was twitching and shaking dramatically. He looked 20 years older than his actual age of 50 and like someone you would imagine in the movie One Flew Over the Cuckoo’s Nest,” said Benjamin Gilmer.
He felt that “there was something clearly wrong with him.” They agreed to a second meeting, and this time Benjamin Gilmer invited a psychiatrist, Steve Buie, MD, to observe Vince. As the visit ended and Vince turned to leave, Dr. Buie watched his shuffling gait. They suspected he may have Huntington’s disease, “which explained why he had delusions and his mind was unraveling,” says Benjamin Gilmer. But they had no way of testing him in prison.
Unexpectedly, an event happened that turned the whole case on its head. Vince was moved to a psychiatric hospital in southern Virginia because he had threatened to commit suicide. The chief psychiatrist, Colin Angliker, MD, was willing to order a genetic test, and the results confirmed the diagnosis: Vince Gilmer had a terminal degenerative brain disease.
Benjamin Gilmer worried how Vince would take the news. To his surprise, Vince was grateful and relieved. He finally knew what was wrong with him.
Vince also improved with the SSRI that Dr. Angliker prescribed — he was less anxious and more mentally alert. “He expressed joy for the first time, despite the death sentence of a diagnosis.”
Still, he was going to spend the rest of his life in prison for the crime he committed.
After the This American Life episode aired in 2013, Benjamin Gilmer felt that he couldn’t just abandon Vince to the prison system, where thousands of inmates with mental illness languish without adequate treatment.
Benjamin Gilmer decided he had a new — although controversial — mission — to get Vince out.
Confronting the politics of a pardon
After nearly a decade of trying, Benjamin Gilmer now admits that he was naive to think he could get him released quickly.
After the episode aired, offers of legal help started to arrive, and a team was assembled who agreed to work on the case pro bono. They wanted justice for Vince but also to prevent anyone else with mental illness from experiencing a similar tragedy.
The goal was to get Vince transferred to a secure hospital, a psychiatric facility dedicated to Huntington’s patients, or a nursing home with a dementia unit.
However, after realizing that Vince may not survive a potentially lengthy court battle, the legal team decided to ask the governor of Virginia to grant a clemency pardon.
They gathered the evidence for Vince’s case and presented their petition to Gov. Terry McAuliffe (D). He rejected it at the end of his term in 2017.
The team tried again with his successor, Gov. Ralph Northam (D), a neurologist. He dashed their hopes when he rejected their petition in late 2021.
That was a huge setback. The team had spent $1 million and had exhausted every contact they could make with the governor’s office, says Gilmer. “We were totally demoralized.”
He dreaded having to tell Vince that yet another governor had rejected their clemency petition. “I went to prison and could see the hopelessness and despair in his reaction. I lost it emotionally,” says Benjamin Gilmer.
Vince surprised him by hugging and comforting him and thanking him for all his efforts. They had developed a strong bond over a decade of visits and calls. Benjamin Gilmer had even brought his wife and children along on special occasions.
“I thought of him as a friend, as a patient, and someone who was really suffering, all those things helped our relationship evolve and kept me engaged with him all these years and continued to inspire me to fight for him. I also liked him because I knew what he was like before the murder from the stories I was hearing from his friends and patients.”
But his continuous advocacy came at a personal cost. “This battle pushed me to my limits emotionally and intellectually. I was busy building my career, trying to be a good doctor, teacher, husband, and father to two young children. I became so distracted that my wife confronted me several times about not being more emotionally present,” says Benjamin Gilmer.
But he knows that without Vince in his life, he would not have written his first book (released earlier this year) about the case and their unlikely friendship.
A pardon is finally granted
He had also given Gov. Northam’s staff advance copies of the book. In a highly unusual move, the governor reversed his previous rejection and granted Vince Gilmer his long-awaited pardon on January 12.
Benjamin Gilmer isn’t ready to celebrate yet. “Despite being a free man, Vince is still living behind bars because we haven’t been able to find him an available bed in a secure treatment facility. There has been a shortage of beds due to COVID.”
He says Vince is looking forward to being safe and being surrounded by people who are committed to caring for him and not punishing him. He can’t wait to be around his family and to give and receive hugs.
“After a while, it was hard not to believe that I was supposed to be in his path and this was just part of my destiny,” says Benjamin Gilmer.
A version of this article first appeared on Medscape.com.
Driving from his home in Asheville, N.C., to his new job at the tiny Cane Creek clinic, Benjamin Gilmer, MD, was eager to start his new life and pay off his medical school debts.
The rural clinic had been forced to close after his predecessor, family physician Vince Gilmer, MD, (no relation) had been convicted of first-degree murder 4 years earlier. He was serving a life sentence in a West Virginia prison without the possibility of parole. He is still behind bars and could not comment on this story.
As the months flew by, Benjamin Gilmer’s patients shared stories about the other Dr. Gilmer that surprised him. They described Vince Gilmer as a caring, generous person who went out of his way to help them. He made house calls, and if a patient couldn’t afford to pay him, he would accept a bushel of corn instead.
Yet there was no doubt about the gruesome murder. Vince Gilmer was convicted of strangling his frail 60-year-old father with a rope in his Toyota truck. He then cut off all his father’s fingers and dumped his father’s body by the side of the road.
“Four years later, his patients were still shocked about what happened and couldn’t reconcile the person they knew with the event that happened,” says Benjamin Gilmer.
Yet, Vince Gilmer had admitted to the killing, and the prosecution had presented evidence at the trial that it was premeditated and that he tried to cover up the crime. The detectives found the “murder” weapons in Vince’s truck: the ropes he strangled his father with and the garden shears that he cut off his fingers with. They also had evidence that he drove to Virginia to dump the body, returned to see patients for several days as if nothing had happened, and then ran away when a detective came to arrest him.
But something kept gnawing away at Benjamin Gilmer.
Little did he know that he would embark on a journey to solve a medical mystery, and then even fight to get the convicted killer out of prison.
Solving a medical mystery
Benjamin Gilmer decided to investigate what might have happened to Vince in the months leading up to the murder. He talked to his friends and found several clues about Vince’s medical history. They recalled that he suffered a concussion in a car accident 6 months before the murder, which suggested he could have had a traumatic brain injury.
Benjamin Gilmer also discovered that Vince’s father was diagnosed with schizophrenia and had been in a residential psychiatric facility in Virginia until he was released that fateful night to Vince’s custody.
Vince had written to friends that “something is wrong with my brain and help me.” He mentioned SSRI discontinuation syndrome because he abruptly stopped taking his medication the week of the murder (which can cause electric shock sensations and mood swings among other symptoms).
Vince had mentioned the SSRI discontinuation syndrome at his trial and that his father had sexually molested him for years and that he tried to molest him again during the ride in his truck. However, the court dismissed that information because Vince represented himself, dismissed his court-appointed attorneys, and lacked expert testimony about his mental state.
The prosecutor portrayed Vince as a lying sociopath who had planned his father’s murder down to the last detail. The judge agreed. Two psychiatrists and a psychologist who later evaluated him in prison concluded that he was faking his symptoms and denied his requests for an SSRI.
Meanwhile, Benjamin Gilmer became increasingly preoccupied with what happened to Vince. “It was hard to erase a memory that had so tainted that community,” he said.
When Sarah Koenig, a journalist and former producer of the radio program This American Life, called Benjamin Gilmer to interview him about the coincidence of taking over Vince Gilmer’s practice and sharing the same last name, he refused. “I was scared and didn’t want to be on his radar, I was afraid of how he might react.”
In spring 2012, he called Koenig and agreed to collaborate on an episode about Vince’s case. Benjamin Gilmer wrote to Vince Gilmer in prison, asking for a meeting. To his surprise, Vince wanted to meet them.
When Vince shuffled into the waiting area at the Wallens Ridge State Prison in West Virginia, Benjamin Gilmer was shocked by his appearance. “He looked like a caged animal, it was very hard for him to string together ideas and express himself, and he was twitching and shaking dramatically. He looked 20 years older than his actual age of 50 and like someone you would imagine in the movie One Flew Over the Cuckoo’s Nest,” said Benjamin Gilmer.
He felt that “there was something clearly wrong with him.” They agreed to a second meeting, and this time Benjamin Gilmer invited a psychiatrist, Steve Buie, MD, to observe Vince. As the visit ended and Vince turned to leave, Dr. Buie watched his shuffling gait. They suspected he may have Huntington’s disease, “which explained why he had delusions and his mind was unraveling,” says Benjamin Gilmer. But they had no way of testing him in prison.
Unexpectedly, an event happened that turned the whole case on its head. Vince was moved to a psychiatric hospital in southern Virginia because he had threatened to commit suicide. The chief psychiatrist, Colin Angliker, MD, was willing to order a genetic test, and the results confirmed the diagnosis: Vince Gilmer had a terminal degenerative brain disease.
Benjamin Gilmer worried how Vince would take the news. To his surprise, Vince was grateful and relieved. He finally knew what was wrong with him.
Vince also improved with the SSRI that Dr. Angliker prescribed — he was less anxious and more mentally alert. “He expressed joy for the first time, despite the death sentence of a diagnosis.”
Still, he was going to spend the rest of his life in prison for the crime he committed.
After the This American Life episode aired in 2013, Benjamin Gilmer felt that he couldn’t just abandon Vince to the prison system, where thousands of inmates with mental illness languish without adequate treatment.
Benjamin Gilmer decided he had a new — although controversial — mission — to get Vince out.
Confronting the politics of a pardon
After nearly a decade of trying, Benjamin Gilmer now admits that he was naive to think he could get him released quickly.
After the episode aired, offers of legal help started to arrive, and a team was assembled who agreed to work on the case pro bono. They wanted justice for Vince but also to prevent anyone else with mental illness from experiencing a similar tragedy.
The goal was to get Vince transferred to a secure hospital, a psychiatric facility dedicated to Huntington’s patients, or a nursing home with a dementia unit.
However, after realizing that Vince may not survive a potentially lengthy court battle, the legal team decided to ask the governor of Virginia to grant a clemency pardon.
They gathered the evidence for Vince’s case and presented their petition to Gov. Terry McAuliffe (D). He rejected it at the end of his term in 2017.
The team tried again with his successor, Gov. Ralph Northam (D), a neurologist. He dashed their hopes when he rejected their petition in late 2021.
That was a huge setback. The team had spent $1 million and had exhausted every contact they could make with the governor’s office, says Gilmer. “We were totally demoralized.”
He dreaded having to tell Vince that yet another governor had rejected their clemency petition. “I went to prison and could see the hopelessness and despair in his reaction. I lost it emotionally,” says Benjamin Gilmer.
Vince surprised him by hugging and comforting him and thanking him for all his efforts. They had developed a strong bond over a decade of visits and calls. Benjamin Gilmer had even brought his wife and children along on special occasions.
“I thought of him as a friend, as a patient, and someone who was really suffering, all those things helped our relationship evolve and kept me engaged with him all these years and continued to inspire me to fight for him. I also liked him because I knew what he was like before the murder from the stories I was hearing from his friends and patients.”
But his continuous advocacy came at a personal cost. “This battle pushed me to my limits emotionally and intellectually. I was busy building my career, trying to be a good doctor, teacher, husband, and father to two young children. I became so distracted that my wife confronted me several times about not being more emotionally present,” says Benjamin Gilmer.
But he knows that without Vince in his life, he would not have written his first book (released earlier this year) about the case and their unlikely friendship.
A pardon is finally granted
He had also given Gov. Northam’s staff advance copies of the book. In a highly unusual move, the governor reversed his previous rejection and granted Vince Gilmer his long-awaited pardon on January 12.
Benjamin Gilmer isn’t ready to celebrate yet. “Despite being a free man, Vince is still living behind bars because we haven’t been able to find him an available bed in a secure treatment facility. There has been a shortage of beds due to COVID.”
He says Vince is looking forward to being safe and being surrounded by people who are committed to caring for him and not punishing him. He can’t wait to be around his family and to give and receive hugs.
“After a while, it was hard not to believe that I was supposed to be in his path and this was just part of my destiny,” says Benjamin Gilmer.
A version of this article first appeared on Medscape.com.
Driving from his home in Asheville, N.C., to his new job at the tiny Cane Creek clinic, Benjamin Gilmer, MD, was eager to start his new life and pay off his medical school debts.
The rural clinic had been forced to close after his predecessor, family physician Vince Gilmer, MD, (no relation) had been convicted of first-degree murder 4 years earlier. He was serving a life sentence in a West Virginia prison without the possibility of parole. He is still behind bars and could not comment on this story.
As the months flew by, Benjamin Gilmer’s patients shared stories about the other Dr. Gilmer that surprised him. They described Vince Gilmer as a caring, generous person who went out of his way to help them. He made house calls, and if a patient couldn’t afford to pay him, he would accept a bushel of corn instead.
Yet there was no doubt about the gruesome murder. Vince Gilmer was convicted of strangling his frail 60-year-old father with a rope in his Toyota truck. He then cut off all his father’s fingers and dumped his father’s body by the side of the road.
“Four years later, his patients were still shocked about what happened and couldn’t reconcile the person they knew with the event that happened,” says Benjamin Gilmer.
Yet, Vince Gilmer had admitted to the killing, and the prosecution had presented evidence at the trial that it was premeditated and that he tried to cover up the crime. The detectives found the “murder” weapons in Vince’s truck: the ropes he strangled his father with and the garden shears that he cut off his fingers with. They also had evidence that he drove to Virginia to dump the body, returned to see patients for several days as if nothing had happened, and then ran away when a detective came to arrest him.
But something kept gnawing away at Benjamin Gilmer.
Little did he know that he would embark on a journey to solve a medical mystery, and then even fight to get the convicted killer out of prison.
Solving a medical mystery
Benjamin Gilmer decided to investigate what might have happened to Vince in the months leading up to the murder. He talked to his friends and found several clues about Vince’s medical history. They recalled that he suffered a concussion in a car accident 6 months before the murder, which suggested he could have had a traumatic brain injury.
Benjamin Gilmer also discovered that Vince’s father was diagnosed with schizophrenia and had been in a residential psychiatric facility in Virginia until he was released that fateful night to Vince’s custody.
Vince had written to friends that “something is wrong with my brain and help me.” He mentioned SSRI discontinuation syndrome because he abruptly stopped taking his medication the week of the murder (which can cause electric shock sensations and mood swings among other symptoms).
Vince had mentioned the SSRI discontinuation syndrome at his trial and that his father had sexually molested him for years and that he tried to molest him again during the ride in his truck. However, the court dismissed that information because Vince represented himself, dismissed his court-appointed attorneys, and lacked expert testimony about his mental state.
The prosecutor portrayed Vince as a lying sociopath who had planned his father’s murder down to the last detail. The judge agreed. Two psychiatrists and a psychologist who later evaluated him in prison concluded that he was faking his symptoms and denied his requests for an SSRI.
Meanwhile, Benjamin Gilmer became increasingly preoccupied with what happened to Vince. “It was hard to erase a memory that had so tainted that community,” he said.
When Sarah Koenig, a journalist and former producer of the radio program This American Life, called Benjamin Gilmer to interview him about the coincidence of taking over Vince Gilmer’s practice and sharing the same last name, he refused. “I was scared and didn’t want to be on his radar, I was afraid of how he might react.”
In spring 2012, he called Koenig and agreed to collaborate on an episode about Vince’s case. Benjamin Gilmer wrote to Vince Gilmer in prison, asking for a meeting. To his surprise, Vince wanted to meet them.
When Vince shuffled into the waiting area at the Wallens Ridge State Prison in West Virginia, Benjamin Gilmer was shocked by his appearance. “He looked like a caged animal, it was very hard for him to string together ideas and express himself, and he was twitching and shaking dramatically. He looked 20 years older than his actual age of 50 and like someone you would imagine in the movie One Flew Over the Cuckoo’s Nest,” said Benjamin Gilmer.
He felt that “there was something clearly wrong with him.” They agreed to a second meeting, and this time Benjamin Gilmer invited a psychiatrist, Steve Buie, MD, to observe Vince. As the visit ended and Vince turned to leave, Dr. Buie watched his shuffling gait. They suspected he may have Huntington’s disease, “which explained why he had delusions and his mind was unraveling,” says Benjamin Gilmer. But they had no way of testing him in prison.
Unexpectedly, an event happened that turned the whole case on its head. Vince was moved to a psychiatric hospital in southern Virginia because he had threatened to commit suicide. The chief psychiatrist, Colin Angliker, MD, was willing to order a genetic test, and the results confirmed the diagnosis: Vince Gilmer had a terminal degenerative brain disease.
Benjamin Gilmer worried how Vince would take the news. To his surprise, Vince was grateful and relieved. He finally knew what was wrong with him.
Vince also improved with the SSRI that Dr. Angliker prescribed — he was less anxious and more mentally alert. “He expressed joy for the first time, despite the death sentence of a diagnosis.”
Still, he was going to spend the rest of his life in prison for the crime he committed.
After the This American Life episode aired in 2013, Benjamin Gilmer felt that he couldn’t just abandon Vince to the prison system, where thousands of inmates with mental illness languish without adequate treatment.
Benjamin Gilmer decided he had a new — although controversial — mission — to get Vince out.
Confronting the politics of a pardon
After nearly a decade of trying, Benjamin Gilmer now admits that he was naive to think he could get him released quickly.
After the episode aired, offers of legal help started to arrive, and a team was assembled who agreed to work on the case pro bono. They wanted justice for Vince but also to prevent anyone else with mental illness from experiencing a similar tragedy.
The goal was to get Vince transferred to a secure hospital, a psychiatric facility dedicated to Huntington’s patients, or a nursing home with a dementia unit.
However, after realizing that Vince may not survive a potentially lengthy court battle, the legal team decided to ask the governor of Virginia to grant a clemency pardon.
They gathered the evidence for Vince’s case and presented their petition to Gov. Terry McAuliffe (D). He rejected it at the end of his term in 2017.
The team tried again with his successor, Gov. Ralph Northam (D), a neurologist. He dashed their hopes when he rejected their petition in late 2021.
That was a huge setback. The team had spent $1 million and had exhausted every contact they could make with the governor’s office, says Gilmer. “We were totally demoralized.”
He dreaded having to tell Vince that yet another governor had rejected their clemency petition. “I went to prison and could see the hopelessness and despair in his reaction. I lost it emotionally,” says Benjamin Gilmer.
Vince surprised him by hugging and comforting him and thanking him for all his efforts. They had developed a strong bond over a decade of visits and calls. Benjamin Gilmer had even brought his wife and children along on special occasions.
“I thought of him as a friend, as a patient, and someone who was really suffering, all those things helped our relationship evolve and kept me engaged with him all these years and continued to inspire me to fight for him. I also liked him because I knew what he was like before the murder from the stories I was hearing from his friends and patients.”
But his continuous advocacy came at a personal cost. “This battle pushed me to my limits emotionally and intellectually. I was busy building my career, trying to be a good doctor, teacher, husband, and father to two young children. I became so distracted that my wife confronted me several times about not being more emotionally present,” says Benjamin Gilmer.
But he knows that without Vince in his life, he would not have written his first book (released earlier this year) about the case and their unlikely friendship.
A pardon is finally granted
He had also given Gov. Northam’s staff advance copies of the book. In a highly unusual move, the governor reversed his previous rejection and granted Vince Gilmer his long-awaited pardon on January 12.
Benjamin Gilmer isn’t ready to celebrate yet. “Despite being a free man, Vince is still living behind bars because we haven’t been able to find him an available bed in a secure treatment facility. There has been a shortage of beds due to COVID.”
He says Vince is looking forward to being safe and being surrounded by people who are committed to caring for him and not punishing him. He can’t wait to be around his family and to give and receive hugs.
“After a while, it was hard not to believe that I was supposed to be in his path and this was just part of my destiny,” says Benjamin Gilmer.
A version of this article first appeared on Medscape.com.
Statins tied to lower risk for parkinsonism
, new research suggests. An observational study showed older adults taking statins had a lower risk for parkinsonism than their counterparts not taking statins – an effect that may be partially mediated by less severe intracranial atherosclerosis in statin users.
“These findings further support the idea that cerebrovascular disease pathologies accumulating in older brains may be an unrecognized contributor to the common occurrence of parkinsonism in old age,” the investigators wrote. “More importantly, these findings suggest that statins may have a potential therapeutic role in decreasing the magnitude of parkinsonism in older adults,” they added.
The study was published online in Neurology.
No clinical recommendations ... yet
The findings are based on 2,841 older adults enrolled in one of three ongoing clinical pathological studies at Rush Alzheimer’s Disease Center, Chicago.
Participants’ average age at baseline was 76 years, and 75% were women. None had parkinsonism at the start of the study. One-third of participants (n = 936) were taking statins. During an average follow-up of 6 years, 1,432 (50%) participants developed parkinsonism.
After controlling for demographics, vascular risk factors, and diseases, use of a statin at baseline was associated with a 16% lower risk for parkinsonism (hazard ratio, 0.84; 95% confidence interval, 0.74-0.96; P = .008). Compared with low-intensity statin therapy, moderate- or high-intensity statin therapy was associated with a 7% lower risk for parkinsonism (HR, 0.93; 95% CI, 0.87-1.00; P = .043).
The researchers also examined the brains of 1,044 people who died during the study at a mean age of 89 years. They found statin use prior to death was associated with a 37% lower odds of cerebral atherosclerosis, compared with no statin use prior to death (odds ratio, 0.63; 95% CI, 0.50-0.79; P < .001).
In a mediation analysis, both a direct (OR, 0.73; 95% CI, 0.54-0.93; P = .008) and an indirect (OR, 0.92; 95% CI, 0.88-0.97; P = .002) pathway via less severe cerebrovascular disease linked statins to parkinsonism, indicating that cerebral atherosclerosis mediated 17% of the association between statins and parkinsonism.
In line with other studies, there was no association between statins and other neurodegenerative pathologies, including Parkinson’s disease pathology. However, even older adults with a clinical diagnosis of Parkinson’s disease often show mixed brain pathologies, including cerebrovascular disease pathologies.
“Therefore, we think that statins may be beneficial against parkinsonism in patients with Parkinson’s disease, dependent on how much cerebrovascular disease pathologies they have, including atherosclerosis,” said study investigator Shahram Oveisgharan, MD, with Rush Alzheimer’s Disease Center, Rush University Medical Center, Chicago.
However, since the results stem from an observational study, “we do not yet recommend using statins in large scale for older adults at risk for parkinsonism,” Dr. Oveisgharan said.
A mixed picture
Reached for comment, Shaheen Lakhan, MD, neurologist in Newton, Massachusetts, noted that since statins were first discovered in the fermented broth of a common soil fungus in the late 1970s, they have proven to reduce cholesterol, heart disease, and stroke.
“The jury is out, however, on [their] effects on diseases such as dementia, autoimmune/inflammatory conditions, bacterial/viral infections, cancer, and parkinsonism,” he said.
“Also, the question often remains whether any benefit gained from statins is from cholesterol-lowering or through another mechanism. When there is such a mixed picture, it generally means that the drug has an effect, but not for everyone,” Dr. Lakhan said. “Much work must now be done to stratify for which patients are statins effective, ineffective, or even harmful in these conditions,” he added.
The study was supported by the National Institutes of Health. Dr. Oveisgharan and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests. An observational study showed older adults taking statins had a lower risk for parkinsonism than their counterparts not taking statins – an effect that may be partially mediated by less severe intracranial atherosclerosis in statin users.
“These findings further support the idea that cerebrovascular disease pathologies accumulating in older brains may be an unrecognized contributor to the common occurrence of parkinsonism in old age,” the investigators wrote. “More importantly, these findings suggest that statins may have a potential therapeutic role in decreasing the magnitude of parkinsonism in older adults,” they added.
The study was published online in Neurology.
No clinical recommendations ... yet
The findings are based on 2,841 older adults enrolled in one of three ongoing clinical pathological studies at Rush Alzheimer’s Disease Center, Chicago.
Participants’ average age at baseline was 76 years, and 75% were women. None had parkinsonism at the start of the study. One-third of participants (n = 936) were taking statins. During an average follow-up of 6 years, 1,432 (50%) participants developed parkinsonism.
After controlling for demographics, vascular risk factors, and diseases, use of a statin at baseline was associated with a 16% lower risk for parkinsonism (hazard ratio, 0.84; 95% confidence interval, 0.74-0.96; P = .008). Compared with low-intensity statin therapy, moderate- or high-intensity statin therapy was associated with a 7% lower risk for parkinsonism (HR, 0.93; 95% CI, 0.87-1.00; P = .043).
The researchers also examined the brains of 1,044 people who died during the study at a mean age of 89 years. They found statin use prior to death was associated with a 37% lower odds of cerebral atherosclerosis, compared with no statin use prior to death (odds ratio, 0.63; 95% CI, 0.50-0.79; P < .001).
In a mediation analysis, both a direct (OR, 0.73; 95% CI, 0.54-0.93; P = .008) and an indirect (OR, 0.92; 95% CI, 0.88-0.97; P = .002) pathway via less severe cerebrovascular disease linked statins to parkinsonism, indicating that cerebral atherosclerosis mediated 17% of the association between statins and parkinsonism.
In line with other studies, there was no association between statins and other neurodegenerative pathologies, including Parkinson’s disease pathology. However, even older adults with a clinical diagnosis of Parkinson’s disease often show mixed brain pathologies, including cerebrovascular disease pathologies.
“Therefore, we think that statins may be beneficial against parkinsonism in patients with Parkinson’s disease, dependent on how much cerebrovascular disease pathologies they have, including atherosclerosis,” said study investigator Shahram Oveisgharan, MD, with Rush Alzheimer’s Disease Center, Rush University Medical Center, Chicago.
However, since the results stem from an observational study, “we do not yet recommend using statins in large scale for older adults at risk for parkinsonism,” Dr. Oveisgharan said.
A mixed picture
Reached for comment, Shaheen Lakhan, MD, neurologist in Newton, Massachusetts, noted that since statins were first discovered in the fermented broth of a common soil fungus in the late 1970s, they have proven to reduce cholesterol, heart disease, and stroke.
“The jury is out, however, on [their] effects on diseases such as dementia, autoimmune/inflammatory conditions, bacterial/viral infections, cancer, and parkinsonism,” he said.
“Also, the question often remains whether any benefit gained from statins is from cholesterol-lowering or through another mechanism. When there is such a mixed picture, it generally means that the drug has an effect, but not for everyone,” Dr. Lakhan said. “Much work must now be done to stratify for which patients are statins effective, ineffective, or even harmful in these conditions,” he added.
The study was supported by the National Institutes of Health. Dr. Oveisgharan and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests. An observational study showed older adults taking statins had a lower risk for parkinsonism than their counterparts not taking statins – an effect that may be partially mediated by less severe intracranial atherosclerosis in statin users.
“These findings further support the idea that cerebrovascular disease pathologies accumulating in older brains may be an unrecognized contributor to the common occurrence of parkinsonism in old age,” the investigators wrote. “More importantly, these findings suggest that statins may have a potential therapeutic role in decreasing the magnitude of parkinsonism in older adults,” they added.
The study was published online in Neurology.
No clinical recommendations ... yet
The findings are based on 2,841 older adults enrolled in one of three ongoing clinical pathological studies at Rush Alzheimer’s Disease Center, Chicago.
Participants’ average age at baseline was 76 years, and 75% were women. None had parkinsonism at the start of the study. One-third of participants (n = 936) were taking statins. During an average follow-up of 6 years, 1,432 (50%) participants developed parkinsonism.
After controlling for demographics, vascular risk factors, and diseases, use of a statin at baseline was associated with a 16% lower risk for parkinsonism (hazard ratio, 0.84; 95% confidence interval, 0.74-0.96; P = .008). Compared with low-intensity statin therapy, moderate- or high-intensity statin therapy was associated with a 7% lower risk for parkinsonism (HR, 0.93; 95% CI, 0.87-1.00; P = .043).
The researchers also examined the brains of 1,044 people who died during the study at a mean age of 89 years. They found statin use prior to death was associated with a 37% lower odds of cerebral atherosclerosis, compared with no statin use prior to death (odds ratio, 0.63; 95% CI, 0.50-0.79; P < .001).
In a mediation analysis, both a direct (OR, 0.73; 95% CI, 0.54-0.93; P = .008) and an indirect (OR, 0.92; 95% CI, 0.88-0.97; P = .002) pathway via less severe cerebrovascular disease linked statins to parkinsonism, indicating that cerebral atherosclerosis mediated 17% of the association between statins and parkinsonism.
In line with other studies, there was no association between statins and other neurodegenerative pathologies, including Parkinson’s disease pathology. However, even older adults with a clinical diagnosis of Parkinson’s disease often show mixed brain pathologies, including cerebrovascular disease pathologies.
“Therefore, we think that statins may be beneficial against parkinsonism in patients with Parkinson’s disease, dependent on how much cerebrovascular disease pathologies they have, including atherosclerosis,” said study investigator Shahram Oveisgharan, MD, with Rush Alzheimer’s Disease Center, Rush University Medical Center, Chicago.
However, since the results stem from an observational study, “we do not yet recommend using statins in large scale for older adults at risk for parkinsonism,” Dr. Oveisgharan said.
A mixed picture
Reached for comment, Shaheen Lakhan, MD, neurologist in Newton, Massachusetts, noted that since statins were first discovered in the fermented broth of a common soil fungus in the late 1970s, they have proven to reduce cholesterol, heart disease, and stroke.
“The jury is out, however, on [their] effects on diseases such as dementia, autoimmune/inflammatory conditions, bacterial/viral infections, cancer, and parkinsonism,” he said.
“Also, the question often remains whether any benefit gained from statins is from cholesterol-lowering or through another mechanism. When there is such a mixed picture, it generally means that the drug has an effect, but not for everyone,” Dr. Lakhan said. “Much work must now be done to stratify for which patients are statins effective, ineffective, or even harmful in these conditions,” he added.
The study was supported by the National Institutes of Health. Dr. Oveisgharan and Dr. Lakhan have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Long-term smell loss in COVID-19 tied to damage in the brain’s olfactory bulb
Patients with COVID-19, especially those with an altered sense of smell, have significantly more axon and microvasculopathy damage in the brain’s olfactory tissue versus non-COVID patients. These new findings from a postmortem study may explain long-term loss of smell in some patients with the virus.
“The striking axonal pathology in some cases indicates that olfactory dysfunction in COVID-19 may be severe and permanent,” the investigators led by Cheng-Ying Ho, MD, PhD, associate professor, department of pathology, Johns Hopkins University School of Medicine, Baltimore, write.
“The results show the damage caused by COVID can extend beyond the nasal cavity and involve the brain,” Dr. Ho told this news organization.
The study was published online April 11 in JAMA Neurology.
A more thorough investigation
Patients infected with SARS-CoV-2, which causes COVID-19, present with a wide range of symptoms. In addition to respiratory illnesses, they may exhibit various nonrespiratory manifestations of COVID-19.
One of the most prevalent of these is olfactory dysfunction. Research shows such dysfunction, including anosmia (loss of smell), hyposmia (reduced sense of smell), and parosmia (smells that are distorted or unpleasant), affects 30%-60% of patients with COVID-19, said Dr. Ho.
However, these statistics come from research before the advent of the Omicron variant, which evidence suggests causes less smell loss in patients with COVID, she said.
Previous studies in this area mainly focused on the lining of the nasal cavity. “We wanted to go a step beyond to see how the olfactory bulb was affected by COVID infection,” said Dr. Ho.
The study included 23 deceased patients with confirmed COVID-19 ranging in age from 28 to 93 years at death (median 62 years, 60.9% men). It also included 14 controls who tested negative for COVID-19, ranging in age from 20 to 77 years (median 53.5 years, 50% men).
Researchers collected postmortem tissue from the brain, lung, and other organs and reviewed pertinent clinical information.
Most patients with COVID died of COVID pneumonia or related complications, although some died from a different cause. Some had an active COVID infection and others were “post infection, meaning they were in the recovery stage,” said Dr. Ho.
Six patients with COVID-19 and eight controls had significant brain pathology.
Compared with controls, those with COVID-19 showed significantly worse olfactory axonal damage. The mean axon pathology score (range 1-3 with 3 the worst) was 1.921 in patients with COVID-19 and 1.198 in controls (95% confidence interval, 0.444-1.002; P < .001).
The mean axon density in the lateral olfactory tract was significantly less in patients with COVID-19 than in controls (P = .002), indicating a 23% loss of olfactory axons in the COVID group.
Comparing COVID patients with and without reported loss of smell, researchers found those with an altered sense of smell had significantly more severe olfactory axon pathology.
Vascular damage
Patients with COVID also had worse vascular damage. The mean microvasculopathy score (range, 1-3) was 1.907 in patients with COVID-19 and 1.405 in controls (95% CI, 0.259-0.745; P < .001).
There was no evidence of the virus in the olfactory tissue of most patients, suggesting the olfactory pathology was likely caused by vascular damage, said Dr. Ho.
What’s unique about SARS-CoV-2 is that, although it’s a respiratory virus, it’s capable of infecting endothelial cells lining vessels.
“Other respiratory viruses only attack the airways and won’t attack vessels, but vascular damage has been seen in the heart and lung in COVID patients, and our study showed the same findings in the olfactory bulb,” Dr. Ho explained.
The researchers divided patients with COVID by infection severity: mild, moderate, severe, and critical. Interestingly, those with the most severe olfactory pathology were the ones with milder infections, said Dr. Ho.
She noted other studies have reported patients with mild infection are more likely to lose the sense of smell than those with severe infection, but she’s skeptical about this finding.
“Patients with severe COVID are usually hospitalized and intubated, so it’s hard to get them to tell you whether they’ve lost smell or not; they have other more important issues to deal with like respiratory failure,” said Dr. Ho.
Advanced age is associated with neuropathologic changes, such as tau deposits, so the researchers conducted an analysis factoring in age-related brain changes. They found a COVID-19 diagnosis remained associated with increased axonal pathology, reduced axonal density, and increased vascular pathology.
“This means that the COVID patients had more severe olfactory pathology not just because they had more tau pathology,” Dr. Ho added.
New guidance for patients
Commenting for this news organization, Davangere P. Devanand, MD, professor of psychiatry and neurology and director of geriatric psychiatry, Columbia University Irving Medical Center, New York, said the findings indicate the damage from COVID in the olfactory pathway may not be reversible as was previously thought.
“This has been suggested before as a possibility, but the autopsy findings in this case series indicate clearly that there may be permanent damage,” he said.
The results highlight the need to monitor patients with COVID for a smell deficit, said Dr. Devanand.
“Assuring patients of a full recovery in smell and taste may not be sound advice, although recovery does occur in many patients,” he added.
He praised the study design, especially the blinding of raters, but noted a number of weaknesses, including the small sample size and the age and gender discrepancies between the groups.
Another possible limitation was inclusion of patients with Alzheimer’s and Lewy body pathology, said Dr. Devanand.
“These patients typically already have pathology in the olfactory pathways, which means we don’t know if it was COVID or the underlying brain pathology contributing to smell difficulties in these patients,” he said.
He noted that, unlike deceased COVID cases in the study, patients who survive COVID may not experience axonal and microvascular injury in olfactory neurons and pathways and their sense of smell may make a full return.
Dr. Devanand said he would have liked more detailed information on the clinical history and course of study participants and whether these factors affected the pathology findings.
The study was supported by grants from the National Institutes of Health.
Dr. Ho and Dr. Devanand have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Patients with COVID-19, especially those with an altered sense of smell, have significantly more axon and microvasculopathy damage in the brain’s olfactory tissue versus non-COVID patients. These new findings from a postmortem study may explain long-term loss of smell in some patients with the virus.
“The striking axonal pathology in some cases indicates that olfactory dysfunction in COVID-19 may be severe and permanent,” the investigators led by Cheng-Ying Ho, MD, PhD, associate professor, department of pathology, Johns Hopkins University School of Medicine, Baltimore, write.
“The results show the damage caused by COVID can extend beyond the nasal cavity and involve the brain,” Dr. Ho told this news organization.
The study was published online April 11 in JAMA Neurology.
A more thorough investigation
Patients infected with SARS-CoV-2, which causes COVID-19, present with a wide range of symptoms. In addition to respiratory illnesses, they may exhibit various nonrespiratory manifestations of COVID-19.
One of the most prevalent of these is olfactory dysfunction. Research shows such dysfunction, including anosmia (loss of smell), hyposmia (reduced sense of smell), and parosmia (smells that are distorted or unpleasant), affects 30%-60% of patients with COVID-19, said Dr. Ho.
However, these statistics come from research before the advent of the Omicron variant, which evidence suggests causes less smell loss in patients with COVID, she said.
Previous studies in this area mainly focused on the lining of the nasal cavity. “We wanted to go a step beyond to see how the olfactory bulb was affected by COVID infection,” said Dr. Ho.
The study included 23 deceased patients with confirmed COVID-19 ranging in age from 28 to 93 years at death (median 62 years, 60.9% men). It also included 14 controls who tested negative for COVID-19, ranging in age from 20 to 77 years (median 53.5 years, 50% men).
Researchers collected postmortem tissue from the brain, lung, and other organs and reviewed pertinent clinical information.
Most patients with COVID died of COVID pneumonia or related complications, although some died from a different cause. Some had an active COVID infection and others were “post infection, meaning they were in the recovery stage,” said Dr. Ho.
Six patients with COVID-19 and eight controls had significant brain pathology.
Compared with controls, those with COVID-19 showed significantly worse olfactory axonal damage. The mean axon pathology score (range 1-3 with 3 the worst) was 1.921 in patients with COVID-19 and 1.198 in controls (95% confidence interval, 0.444-1.002; P < .001).
The mean axon density in the lateral olfactory tract was significantly less in patients with COVID-19 than in controls (P = .002), indicating a 23% loss of olfactory axons in the COVID group.
Comparing COVID patients with and without reported loss of smell, researchers found those with an altered sense of smell had significantly more severe olfactory axon pathology.
Vascular damage
Patients with COVID also had worse vascular damage. The mean microvasculopathy score (range, 1-3) was 1.907 in patients with COVID-19 and 1.405 in controls (95% CI, 0.259-0.745; P < .001).
There was no evidence of the virus in the olfactory tissue of most patients, suggesting the olfactory pathology was likely caused by vascular damage, said Dr. Ho.
What’s unique about SARS-CoV-2 is that, although it’s a respiratory virus, it’s capable of infecting endothelial cells lining vessels.
“Other respiratory viruses only attack the airways and won’t attack vessels, but vascular damage has been seen in the heart and lung in COVID patients, and our study showed the same findings in the olfactory bulb,” Dr. Ho explained.
The researchers divided patients with COVID by infection severity: mild, moderate, severe, and critical. Interestingly, those with the most severe olfactory pathology were the ones with milder infections, said Dr. Ho.
She noted other studies have reported patients with mild infection are more likely to lose the sense of smell than those with severe infection, but she’s skeptical about this finding.
“Patients with severe COVID are usually hospitalized and intubated, so it’s hard to get them to tell you whether they’ve lost smell or not; they have other more important issues to deal with like respiratory failure,” said Dr. Ho.
Advanced age is associated with neuropathologic changes, such as tau deposits, so the researchers conducted an analysis factoring in age-related brain changes. They found a COVID-19 diagnosis remained associated with increased axonal pathology, reduced axonal density, and increased vascular pathology.
“This means that the COVID patients had more severe olfactory pathology not just because they had more tau pathology,” Dr. Ho added.
New guidance for patients
Commenting for this news organization, Davangere P. Devanand, MD, professor of psychiatry and neurology and director of geriatric psychiatry, Columbia University Irving Medical Center, New York, said the findings indicate the damage from COVID in the olfactory pathway may not be reversible as was previously thought.
“This has been suggested before as a possibility, but the autopsy findings in this case series indicate clearly that there may be permanent damage,” he said.
The results highlight the need to monitor patients with COVID for a smell deficit, said Dr. Devanand.
“Assuring patients of a full recovery in smell and taste may not be sound advice, although recovery does occur in many patients,” he added.
He praised the study design, especially the blinding of raters, but noted a number of weaknesses, including the small sample size and the age and gender discrepancies between the groups.
Another possible limitation was inclusion of patients with Alzheimer’s and Lewy body pathology, said Dr. Devanand.
“These patients typically already have pathology in the olfactory pathways, which means we don’t know if it was COVID or the underlying brain pathology contributing to smell difficulties in these patients,” he said.
He noted that, unlike deceased COVID cases in the study, patients who survive COVID may not experience axonal and microvascular injury in olfactory neurons and pathways and their sense of smell may make a full return.
Dr. Devanand said he would have liked more detailed information on the clinical history and course of study participants and whether these factors affected the pathology findings.
The study was supported by grants from the National Institutes of Health.
Dr. Ho and Dr. Devanand have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
Patients with COVID-19, especially those with an altered sense of smell, have significantly more axon and microvasculopathy damage in the brain’s olfactory tissue versus non-COVID patients. These new findings from a postmortem study may explain long-term loss of smell in some patients with the virus.
“The striking axonal pathology in some cases indicates that olfactory dysfunction in COVID-19 may be severe and permanent,” the investigators led by Cheng-Ying Ho, MD, PhD, associate professor, department of pathology, Johns Hopkins University School of Medicine, Baltimore, write.
“The results show the damage caused by COVID can extend beyond the nasal cavity and involve the brain,” Dr. Ho told this news organization.
The study was published online April 11 in JAMA Neurology.
A more thorough investigation
Patients infected with SARS-CoV-2, which causes COVID-19, present with a wide range of symptoms. In addition to respiratory illnesses, they may exhibit various nonrespiratory manifestations of COVID-19.
One of the most prevalent of these is olfactory dysfunction. Research shows such dysfunction, including anosmia (loss of smell), hyposmia (reduced sense of smell), and parosmia (smells that are distorted or unpleasant), affects 30%-60% of patients with COVID-19, said Dr. Ho.
However, these statistics come from research before the advent of the Omicron variant, which evidence suggests causes less smell loss in patients with COVID, she said.
Previous studies in this area mainly focused on the lining of the nasal cavity. “We wanted to go a step beyond to see how the olfactory bulb was affected by COVID infection,” said Dr. Ho.
The study included 23 deceased patients with confirmed COVID-19 ranging in age from 28 to 93 years at death (median 62 years, 60.9% men). It also included 14 controls who tested negative for COVID-19, ranging in age from 20 to 77 years (median 53.5 years, 50% men).
Researchers collected postmortem tissue from the brain, lung, and other organs and reviewed pertinent clinical information.
Most patients with COVID died of COVID pneumonia or related complications, although some died from a different cause. Some had an active COVID infection and others were “post infection, meaning they were in the recovery stage,” said Dr. Ho.
Six patients with COVID-19 and eight controls had significant brain pathology.
Compared with controls, those with COVID-19 showed significantly worse olfactory axonal damage. The mean axon pathology score (range 1-3 with 3 the worst) was 1.921 in patients with COVID-19 and 1.198 in controls (95% confidence interval, 0.444-1.002; P < .001).
The mean axon density in the lateral olfactory tract was significantly less in patients with COVID-19 than in controls (P = .002), indicating a 23% loss of olfactory axons in the COVID group.
Comparing COVID patients with and without reported loss of smell, researchers found those with an altered sense of smell had significantly more severe olfactory axon pathology.
Vascular damage
Patients with COVID also had worse vascular damage. The mean microvasculopathy score (range, 1-3) was 1.907 in patients with COVID-19 and 1.405 in controls (95% CI, 0.259-0.745; P < .001).
There was no evidence of the virus in the olfactory tissue of most patients, suggesting the olfactory pathology was likely caused by vascular damage, said Dr. Ho.
What’s unique about SARS-CoV-2 is that, although it’s a respiratory virus, it’s capable of infecting endothelial cells lining vessels.
“Other respiratory viruses only attack the airways and won’t attack vessels, but vascular damage has been seen in the heart and lung in COVID patients, and our study showed the same findings in the olfactory bulb,” Dr. Ho explained.
The researchers divided patients with COVID by infection severity: mild, moderate, severe, and critical. Interestingly, those with the most severe olfactory pathology were the ones with milder infections, said Dr. Ho.
She noted other studies have reported patients with mild infection are more likely to lose the sense of smell than those with severe infection, but she’s skeptical about this finding.
“Patients with severe COVID are usually hospitalized and intubated, so it’s hard to get them to tell you whether they’ve lost smell or not; they have other more important issues to deal with like respiratory failure,” said Dr. Ho.
Advanced age is associated with neuropathologic changes, such as tau deposits, so the researchers conducted an analysis factoring in age-related brain changes. They found a COVID-19 diagnosis remained associated with increased axonal pathology, reduced axonal density, and increased vascular pathology.
“This means that the COVID patients had more severe olfactory pathology not just because they had more tau pathology,” Dr. Ho added.
New guidance for patients
Commenting for this news organization, Davangere P. Devanand, MD, professor of psychiatry and neurology and director of geriatric psychiatry, Columbia University Irving Medical Center, New York, said the findings indicate the damage from COVID in the olfactory pathway may not be reversible as was previously thought.
“This has been suggested before as a possibility, but the autopsy findings in this case series indicate clearly that there may be permanent damage,” he said.
The results highlight the need to monitor patients with COVID for a smell deficit, said Dr. Devanand.
“Assuring patients of a full recovery in smell and taste may not be sound advice, although recovery does occur in many patients,” he added.
He praised the study design, especially the blinding of raters, but noted a number of weaknesses, including the small sample size and the age and gender discrepancies between the groups.
Another possible limitation was inclusion of patients with Alzheimer’s and Lewy body pathology, said Dr. Devanand.
“These patients typically already have pathology in the olfactory pathways, which means we don’t know if it was COVID or the underlying brain pathology contributing to smell difficulties in these patients,” he said.
He noted that, unlike deceased COVID cases in the study, patients who survive COVID may not experience axonal and microvascular injury in olfactory neurons and pathways and their sense of smell may make a full return.
Dr. Devanand said he would have liked more detailed information on the clinical history and course of study participants and whether these factors affected the pathology findings.
The study was supported by grants from the National Institutes of Health.
Dr. Ho and Dr. Devanand have reported no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM JAMA NEUROLOGY
Getting a jump on recovery from sports-related concussion
ILLUSTRATIVE CASE
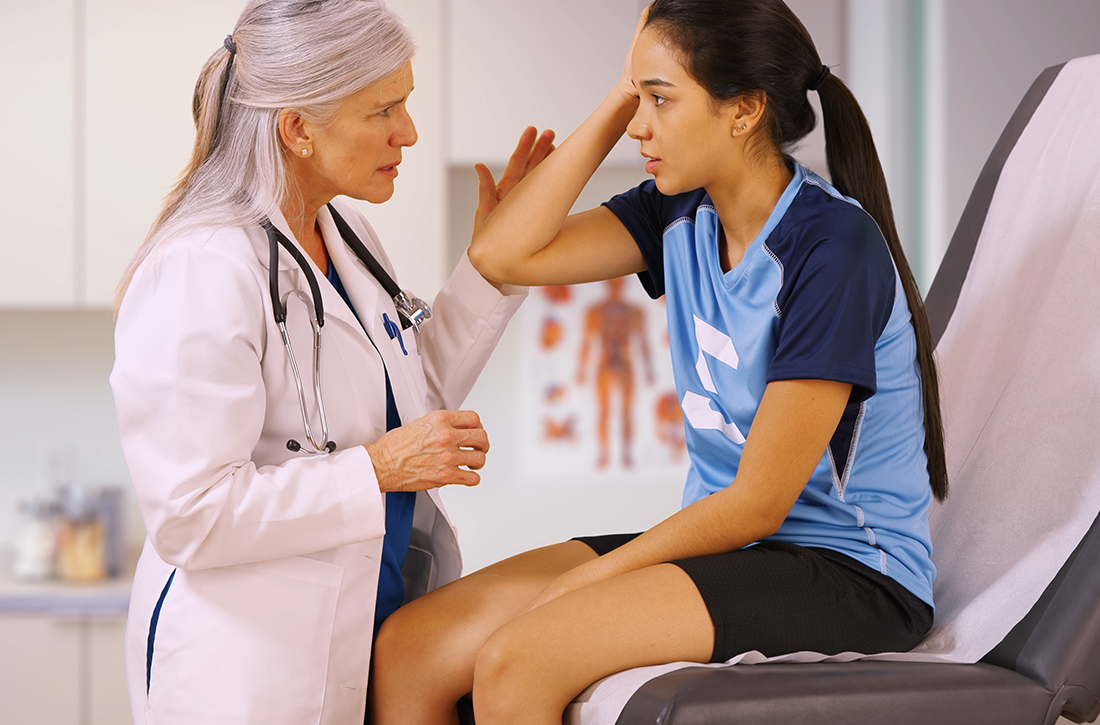
A 16-year-old girl with no chronic medical illness presents to your office with her parents after sustaining a head injury at a soccer game over the weekend. She collided with another player while attempting to head the ball. Immediately afterward, she was taken off the field and assessed. She was confused but had a normal level of consciousness and denied vision changes, weakness or tingling in her arms or legs, severe headache, or neck pain. Further testing revealed dizziness and abnormal balance. Her confusion and abnormal balance resolved after 1 day. She has had a mild headache and light sensitivity since the event. She otherwise feels well at rest in the office. She wants to recover quickly but safely and has heard conflicting statements about whether she should completely rest or start back to light activity now.
Sports-related concussions (SRCs) are highly prevalent in the United States, with as many as 3.8 million cases annually. Of those, 1.1 to 1.9 million cases are in children 18 years old or younger.2,3 SRCs are defined by the Concussion in Sport Group (CISG) 2017 consensus statement as involving the following criteria: (1) direct or indirect trauma anywhere on the body with force transmitted to the head; (2) rapid or delayed symptom presentation, typically with spontaneous resolution; (3) functional rather than structural injury; and (4) occurrence with or without loss of consciousness with stepwise symptom resolution.4
SRCs do not have a proven, effective treatment and can have short- or long-term consequences. Initial treatment includes removing athletes from play immediately after an event. The American Academy of Neurology recommends that athletes not return to play until the concussion is resolved, as judged by a health care provider, and the athlete is asymptomatic when off medication.2
The CISG recommends a 6-step approach, with each step taking at least 24 hours.4 The final step is a return to normal activity.4 This working group recommended extensive study of rehabilitation programs involving subsymptom threshold exercise (ie, exercise performed at a level that does not exacerbate symptoms) before implementation as routine practice. Evidence from a 2015 study suggests that following strict rest for 5 days until complete symptom resolution may prolong recovery compared with rest for only 1 to 2 days.5 Additionally, strict rest did not show a difference in neurocognitive or balance outcomes in that study, and the authors noted it may also negatively impact academic, sports, and social function in adolescents.5 This study looked at the potential benefit of subsymptom threshold exercise during recovery from SRC.1
STUDY SUMMARY
Light aerobic exercise may help speed recovery
This multicenter, prospective, parallel, randomized clinical trial compared subsymptom threshold aerobic exercise to placebo-like stretching. Patients were included if they were ages 13 to 18 years and presented within 10 days of an SRC, as diagnosed using the CISG criteria. Exclusion criteria included focal neurologic deficits; history of moderate or severe traumatic brain injury; inability to exercise due to orthopedic injury, cervical spine injury, diabetes, or heart disease; increased cardiac risk; or low postconcussion symptom severity. Patients with a diagnosis of and treatment with medication for attention-deficit/hyperactivity disorder (ADHD), depression, anxiety, or learning disorder were excluded, as were patients with a history of more than 3 previous concussions.
Patients in the aerobic exercise group were instructed to use a stationary bike or treadmill (or equivalent walking or jogging if they did not have access to this equipment) at a prescribed heart rate. The target heart rate was 80% of the heart rate achieved during initial assessment with the Buffalo Concussion Treadmill Test (BCTT).6 Patients in this group were instructed to exercise for 20 minutes or to the point at which their symptoms increased by 2 points (on a 10-point scale) from pre-exercise levels, whichever came first, with rest prescribed at all other times.
For the placebo-like group, a stretching instruction booklet was provided, with the goal of achieving a heart rate that was not significantly elevated. Participants in this group were told to perform the stretches for 20 minutes daily. Of note, researchers ensured the level of physician and research staff attention was similar for each patient, regardless of treatment group, to prevent intervention bias. Additionally, interventions were not initiated prior to 48 hours from the time of injury.
Continue to: The primary outcome...
The primary outcome was number of days to recovery since the date of injury. This was defined as symptom resolution to normal (as evaluated by a physician blinded to the study group) and by the patient’s ability to exercise to exhaustion without symptom exacerbation on the BCTT. Secondary outcomes measured the proportion of patients with delayed recovery (defined as recovery requiring > 30 days) and daily symptom scores.
Of 165 patients meeting the inclusion criteria, 52 patients were excluded prior to randomization (12 patients chose not to participate, 39 were excluded for lack of symptoms, and 1 withdrew due to severe symptoms on the BCTT). A total of 113 were randomized to either group, and 103 patients completed the study (10 patients did not complete the study or had another illness during the intervention). The study analysis included 52 patients in the aerobic exercise group and 51 in the placebo-like stretching group. The study was powered to detect a significant difference in recovery time.
Patients were about equally divided by sex, with a mean age of 15 years. Patients who had no previous concussion made up 50% of the aerobic group and 57% of the stretching group. The average time since injury was similar in the aerobic and stretching groups (4.9 days and 4.8 days, respectively). The aerobic exercise group recovered in a median of 13 days (interquartile range [IQR] = 10-18.5 days) compared with a median of 17 days (IQR = 13-23 days) for the stretching group (P = .009). The incidence of delayed recovery (> 30 days) was higher in the stretching group (n = 7) compared with the aerobic exercise group (n = 2) but was not statistically significant. Daily symptom reporting occurred at a high rate in both groups, with patients stating that they performed their prescribed exercise 89% of the time. No adverse events were reported.
WHAT’S NEW
First high-quality study to support evidence for early light activity
This is the first high-quality study of subsymptom threshold exercise for SRC. Its findings add to the growing body of evidence that early engagement in light aerobic activity that does not provoke symptoms (but not fully returning to sports activity) can aid in recovery from an SRC.
CAVEATS
Narrow study population limits application of findings
It is unclear if subsymptom threshold exercise is safe and effective in adolescents with a history of multiple concussions, as those with more than 3 concussions were excluded from this study. Additionally, patients with comorbidities such as ADHD, depression, anxiety, or learning disorders were not included in this study, which limits the application of these findings. The generalizability of this study is limited in younger children, adults, those with increased cardiovascular risk, and in patients with concussions that are not sports related.
CHALLENGES TO IMPLEMENTATION
More real-world studies needed to confirm benefit
The majority of adolescent athletes in this study completed the subsymptom threshold exercise in a monitored environment with trainers, heart rate monitors, and access to equipment, limiting the study’s generalizability. Additionally, physicians need to be familiar with the BCTT to assign heart rate goals and assess improvement. The study environment may be feasible for some but not others. Studies evaluating real-world settings with athletes self-monitoring for symptom threshold with stepwise evaluations are needed and may be more broadly applicable.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Leddy JJ, Haider MN, Ellis MJ, et al. Early subthreshold aerobic exercise for sport-related concussion: a randomized clinical trial. JAMA Pediatr. 2019;173:319-325. doi: 10.1001/jamapediatrics.2018.4397
2. Giza CC, Kutcher JS, Ashwal S, et al. Summary of evidence-based guideline update: evaluation and management of concussion in sports: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013;80:2250-2257. doi: 10.1212/WNL.0b013e31828d57dd
3. Bryan MA, Rowhani-Rahbar A, Comstock RD, et al; Seattle Sports Concussion Research Collaborative. Sports- and recreation-related concussions in US youth. Pediatrics. 2016;138:e20154635. doi: 10.1542/peds.2015-4635
4. McCrory P, Meeuwisse W, Dvořák J, et al. Consensus statement on concussion in sport—the 5th international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med. 2017;51:838-847. doi: 10.1136/bjsports-2017-097699
5. Thomas DG, Apps JN, Hoffmann RG, et al. Benefits of strict rest after acute concussion: a randomized controlled trial. Pediatrics. 2015;135:213-223.
6. Leddy JJ, Haider MN, Willer BS. Buffalo Concussion Treadmill Test (BCTT) – Instruction Manual. Accessed March 16, 2022. https://cdn-links.lww.com/permalink/jsm/a/jsm_2020_01_28_haider_19-313_sdc1.pdf
ILLUSTRATIVE CASE
A 16-year-old girl with no chronic medical illness presents to your office with her parents after sustaining a head injury at a soccer game over the weekend. She collided with another player while attempting to head the ball. Immediately afterward, she was taken off the field and assessed. She was confused but had a normal level of consciousness and denied vision changes, weakness or tingling in her arms or legs, severe headache, or neck pain. Further testing revealed dizziness and abnormal balance. Her confusion and abnormal balance resolved after 1 day. She has had a mild headache and light sensitivity since the event. She otherwise feels well at rest in the office. She wants to recover quickly but safely and has heard conflicting statements about whether she should completely rest or start back to light activity now.
Sports-related concussions (SRCs) are highly prevalent in the United States, with as many as 3.8 million cases annually. Of those, 1.1 to 1.9 million cases are in children 18 years old or younger.2,3 SRCs are defined by the Concussion in Sport Group (CISG) 2017 consensus statement as involving the following criteria: (1) direct or indirect trauma anywhere on the body with force transmitted to the head; (2) rapid or delayed symptom presentation, typically with spontaneous resolution; (3) functional rather than structural injury; and (4) occurrence with or without loss of consciousness with stepwise symptom resolution.4
SRCs do not have a proven, effective treatment and can have short- or long-term consequences. Initial treatment includes removing athletes from play immediately after an event. The American Academy of Neurology recommends that athletes not return to play until the concussion is resolved, as judged by a health care provider, and the athlete is asymptomatic when off medication.2
The CISG recommends a 6-step approach, with each step taking at least 24 hours.4 The final step is a return to normal activity.4 This working group recommended extensive study of rehabilitation programs involving subsymptom threshold exercise (ie, exercise performed at a level that does not exacerbate symptoms) before implementation as routine practice. Evidence from a 2015 study suggests that following strict rest for 5 days until complete symptom resolution may prolong recovery compared with rest for only 1 to 2 days.5 Additionally, strict rest did not show a difference in neurocognitive or balance outcomes in that study, and the authors noted it may also negatively impact academic, sports, and social function in adolescents.5 This study looked at the potential benefit of subsymptom threshold exercise during recovery from SRC.1
STUDY SUMMARY
Light aerobic exercise may help speed recovery
This multicenter, prospective, parallel, randomized clinical trial compared subsymptom threshold aerobic exercise to placebo-like stretching. Patients were included if they were ages 13 to 18 years and presented within 10 days of an SRC, as diagnosed using the CISG criteria. Exclusion criteria included focal neurologic deficits; history of moderate or severe traumatic brain injury; inability to exercise due to orthopedic injury, cervical spine injury, diabetes, or heart disease; increased cardiac risk; or low postconcussion symptom severity. Patients with a diagnosis of and treatment with medication for attention-deficit/hyperactivity disorder (ADHD), depression, anxiety, or learning disorder were excluded, as were patients with a history of more than 3 previous concussions.
Patients in the aerobic exercise group were instructed to use a stationary bike or treadmill (or equivalent walking or jogging if they did not have access to this equipment) at a prescribed heart rate. The target heart rate was 80% of the heart rate achieved during initial assessment with the Buffalo Concussion Treadmill Test (BCTT).6 Patients in this group were instructed to exercise for 20 minutes or to the point at which their symptoms increased by 2 points (on a 10-point scale) from pre-exercise levels, whichever came first, with rest prescribed at all other times.
For the placebo-like group, a stretching instruction booklet was provided, with the goal of achieving a heart rate that was not significantly elevated. Participants in this group were told to perform the stretches for 20 minutes daily. Of note, researchers ensured the level of physician and research staff attention was similar for each patient, regardless of treatment group, to prevent intervention bias. Additionally, interventions were not initiated prior to 48 hours from the time of injury.
Continue to: The primary outcome...
The primary outcome was number of days to recovery since the date of injury. This was defined as symptom resolution to normal (as evaluated by a physician blinded to the study group) and by the patient’s ability to exercise to exhaustion without symptom exacerbation on the BCTT. Secondary outcomes measured the proportion of patients with delayed recovery (defined as recovery requiring > 30 days) and daily symptom scores.
Of 165 patients meeting the inclusion criteria, 52 patients were excluded prior to randomization (12 patients chose not to participate, 39 were excluded for lack of symptoms, and 1 withdrew due to severe symptoms on the BCTT). A total of 113 were randomized to either group, and 103 patients completed the study (10 patients did not complete the study or had another illness during the intervention). The study analysis included 52 patients in the aerobic exercise group and 51 in the placebo-like stretching group. The study was powered to detect a significant difference in recovery time.
Patients were about equally divided by sex, with a mean age of 15 years. Patients who had no previous concussion made up 50% of the aerobic group and 57% of the stretching group. The average time since injury was similar in the aerobic and stretching groups (4.9 days and 4.8 days, respectively). The aerobic exercise group recovered in a median of 13 days (interquartile range [IQR] = 10-18.5 days) compared with a median of 17 days (IQR = 13-23 days) for the stretching group (P = .009). The incidence of delayed recovery (> 30 days) was higher in the stretching group (n = 7) compared with the aerobic exercise group (n = 2) but was not statistically significant. Daily symptom reporting occurred at a high rate in both groups, with patients stating that they performed their prescribed exercise 89% of the time. No adverse events were reported.
WHAT’S NEW
First high-quality study to support evidence for early light activity
This is the first high-quality study of subsymptom threshold exercise for SRC. Its findings add to the growing body of evidence that early engagement in light aerobic activity that does not provoke symptoms (but not fully returning to sports activity) can aid in recovery from an SRC.
CAVEATS
Narrow study population limits application of findings
It is unclear if subsymptom threshold exercise is safe and effective in adolescents with a history of multiple concussions, as those with more than 3 concussions were excluded from this study. Additionally, patients with comorbidities such as ADHD, depression, anxiety, or learning disorders were not included in this study, which limits the application of these findings. The generalizability of this study is limited in younger children, adults, those with increased cardiovascular risk, and in patients with concussions that are not sports related.
CHALLENGES TO IMPLEMENTATION
More real-world studies needed to confirm benefit
The majority of adolescent athletes in this study completed the subsymptom threshold exercise in a monitored environment with trainers, heart rate monitors, and access to equipment, limiting the study’s generalizability. Additionally, physicians need to be familiar with the BCTT to assign heart rate goals and assess improvement. The study environment may be feasible for some but not others. Studies evaluating real-world settings with athletes self-monitoring for symptom threshold with stepwise evaluations are needed and may be more broadly applicable.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 16-year-old girl with no chronic medical illness presents to your office with her parents after sustaining a head injury at a soccer game over the weekend. She collided with another player while attempting to head the ball. Immediately afterward, she was taken off the field and assessed. She was confused but had a normal level of consciousness and denied vision changes, weakness or tingling in her arms or legs, severe headache, or neck pain. Further testing revealed dizziness and abnormal balance. Her confusion and abnormal balance resolved after 1 day. She has had a mild headache and light sensitivity since the event. She otherwise feels well at rest in the office. She wants to recover quickly but safely and has heard conflicting statements about whether she should completely rest or start back to light activity now.
Sports-related concussions (SRCs) are highly prevalent in the United States, with as many as 3.8 million cases annually. Of those, 1.1 to 1.9 million cases are in children 18 years old or younger.2,3 SRCs are defined by the Concussion in Sport Group (CISG) 2017 consensus statement as involving the following criteria: (1) direct or indirect trauma anywhere on the body with force transmitted to the head; (2) rapid or delayed symptom presentation, typically with spontaneous resolution; (3) functional rather than structural injury; and (4) occurrence with or without loss of consciousness with stepwise symptom resolution.4
SRCs do not have a proven, effective treatment and can have short- or long-term consequences. Initial treatment includes removing athletes from play immediately after an event. The American Academy of Neurology recommends that athletes not return to play until the concussion is resolved, as judged by a health care provider, and the athlete is asymptomatic when off medication.2
The CISG recommends a 6-step approach, with each step taking at least 24 hours.4 The final step is a return to normal activity.4 This working group recommended extensive study of rehabilitation programs involving subsymptom threshold exercise (ie, exercise performed at a level that does not exacerbate symptoms) before implementation as routine practice. Evidence from a 2015 study suggests that following strict rest for 5 days until complete symptom resolution may prolong recovery compared with rest for only 1 to 2 days.5 Additionally, strict rest did not show a difference in neurocognitive or balance outcomes in that study, and the authors noted it may also negatively impact academic, sports, and social function in adolescents.5 This study looked at the potential benefit of subsymptom threshold exercise during recovery from SRC.1
STUDY SUMMARY
Light aerobic exercise may help speed recovery
This multicenter, prospective, parallel, randomized clinical trial compared subsymptom threshold aerobic exercise to placebo-like stretching. Patients were included if they were ages 13 to 18 years and presented within 10 days of an SRC, as diagnosed using the CISG criteria. Exclusion criteria included focal neurologic deficits; history of moderate or severe traumatic brain injury; inability to exercise due to orthopedic injury, cervical spine injury, diabetes, or heart disease; increased cardiac risk; or low postconcussion symptom severity. Patients with a diagnosis of and treatment with medication for attention-deficit/hyperactivity disorder (ADHD), depression, anxiety, or learning disorder were excluded, as were patients with a history of more than 3 previous concussions.
Patients in the aerobic exercise group were instructed to use a stationary bike or treadmill (or equivalent walking or jogging if they did not have access to this equipment) at a prescribed heart rate. The target heart rate was 80% of the heart rate achieved during initial assessment with the Buffalo Concussion Treadmill Test (BCTT).6 Patients in this group were instructed to exercise for 20 minutes or to the point at which their symptoms increased by 2 points (on a 10-point scale) from pre-exercise levels, whichever came first, with rest prescribed at all other times.
For the placebo-like group, a stretching instruction booklet was provided, with the goal of achieving a heart rate that was not significantly elevated. Participants in this group were told to perform the stretches for 20 minutes daily. Of note, researchers ensured the level of physician and research staff attention was similar for each patient, regardless of treatment group, to prevent intervention bias. Additionally, interventions were not initiated prior to 48 hours from the time of injury.
Continue to: The primary outcome...
The primary outcome was number of days to recovery since the date of injury. This was defined as symptom resolution to normal (as evaluated by a physician blinded to the study group) and by the patient’s ability to exercise to exhaustion without symptom exacerbation on the BCTT. Secondary outcomes measured the proportion of patients with delayed recovery (defined as recovery requiring > 30 days) and daily symptom scores.
Of 165 patients meeting the inclusion criteria, 52 patients were excluded prior to randomization (12 patients chose not to participate, 39 were excluded for lack of symptoms, and 1 withdrew due to severe symptoms on the BCTT). A total of 113 were randomized to either group, and 103 patients completed the study (10 patients did not complete the study or had another illness during the intervention). The study analysis included 52 patients in the aerobic exercise group and 51 in the placebo-like stretching group. The study was powered to detect a significant difference in recovery time.
Patients were about equally divided by sex, with a mean age of 15 years. Patients who had no previous concussion made up 50% of the aerobic group and 57% of the stretching group. The average time since injury was similar in the aerobic and stretching groups (4.9 days and 4.8 days, respectively). The aerobic exercise group recovered in a median of 13 days (interquartile range [IQR] = 10-18.5 days) compared with a median of 17 days (IQR = 13-23 days) for the stretching group (P = .009). The incidence of delayed recovery (> 30 days) was higher in the stretching group (n = 7) compared with the aerobic exercise group (n = 2) but was not statistically significant. Daily symptom reporting occurred at a high rate in both groups, with patients stating that they performed their prescribed exercise 89% of the time. No adverse events were reported.
WHAT’S NEW
First high-quality study to support evidence for early light activity
This is the first high-quality study of subsymptom threshold exercise for SRC. Its findings add to the growing body of evidence that early engagement in light aerobic activity that does not provoke symptoms (but not fully returning to sports activity) can aid in recovery from an SRC.
CAVEATS
Narrow study population limits application of findings
It is unclear if subsymptom threshold exercise is safe and effective in adolescents with a history of multiple concussions, as those with more than 3 concussions were excluded from this study. Additionally, patients with comorbidities such as ADHD, depression, anxiety, or learning disorders were not included in this study, which limits the application of these findings. The generalizability of this study is limited in younger children, adults, those with increased cardiovascular risk, and in patients with concussions that are not sports related.
CHALLENGES TO IMPLEMENTATION
More real-world studies needed to confirm benefit
The majority of adolescent athletes in this study completed the subsymptom threshold exercise in a monitored environment with trainers, heart rate monitors, and access to equipment, limiting the study’s generalizability. Additionally, physicians need to be familiar with the BCTT to assign heart rate goals and assess improvement. The study environment may be feasible for some but not others. Studies evaluating real-world settings with athletes self-monitoring for symptom threshold with stepwise evaluations are needed and may be more broadly applicable.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Leddy JJ, Haider MN, Ellis MJ, et al. Early subthreshold aerobic exercise for sport-related concussion: a randomized clinical trial. JAMA Pediatr. 2019;173:319-325. doi: 10.1001/jamapediatrics.2018.4397
2. Giza CC, Kutcher JS, Ashwal S, et al. Summary of evidence-based guideline update: evaluation and management of concussion in sports: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013;80:2250-2257. doi: 10.1212/WNL.0b013e31828d57dd
3. Bryan MA, Rowhani-Rahbar A, Comstock RD, et al; Seattle Sports Concussion Research Collaborative. Sports- and recreation-related concussions in US youth. Pediatrics. 2016;138:e20154635. doi: 10.1542/peds.2015-4635
4. McCrory P, Meeuwisse W, Dvořák J, et al. Consensus statement on concussion in sport—the 5th international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med. 2017;51:838-847. doi: 10.1136/bjsports-2017-097699
5. Thomas DG, Apps JN, Hoffmann RG, et al. Benefits of strict rest after acute concussion: a randomized controlled trial. Pediatrics. 2015;135:213-223.
6. Leddy JJ, Haider MN, Willer BS. Buffalo Concussion Treadmill Test (BCTT) – Instruction Manual. Accessed March 16, 2022. https://cdn-links.lww.com/permalink/jsm/a/jsm_2020_01_28_haider_19-313_sdc1.pdf
1. Leddy JJ, Haider MN, Ellis MJ, et al. Early subthreshold aerobic exercise for sport-related concussion: a randomized clinical trial. JAMA Pediatr. 2019;173:319-325. doi: 10.1001/jamapediatrics.2018.4397
2. Giza CC, Kutcher JS, Ashwal S, et al. Summary of evidence-based guideline update: evaluation and management of concussion in sports: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013;80:2250-2257. doi: 10.1212/WNL.0b013e31828d57dd
3. Bryan MA, Rowhani-Rahbar A, Comstock RD, et al; Seattle Sports Concussion Research Collaborative. Sports- and recreation-related concussions in US youth. Pediatrics. 2016;138:e20154635. doi: 10.1542/peds.2015-4635
4. McCrory P, Meeuwisse W, Dvořák J, et al. Consensus statement on concussion in sport—the 5th international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med. 2017;51:838-847. doi: 10.1136/bjsports-2017-097699
5. Thomas DG, Apps JN, Hoffmann RG, et al. Benefits of strict rest after acute concussion: a randomized controlled trial. Pediatrics. 2015;135:213-223.
6. Leddy JJ, Haider MN, Willer BS. Buffalo Concussion Treadmill Test (BCTT) – Instruction Manual. Accessed March 16, 2022. https://cdn-links.lww.com/permalink/jsm/a/jsm_2020_01_28_haider_19-313_sdc1.pdf
PRACTICE CHANGER
Recommend subsymptom threshold exercise in adolescents with a sports-related concussion. Early return to light aerobic activity not only seems safe but may help speed recovery compared with stretching alone in this patient population.
STRENGTH OF RECOMMENDATION
B: Based on a single multicenter, prospective, randomized clinical trial1
Leddy JJ, Haider MN, Ellis MJ, et al. Early subthreshold aerobic exercise for sport-related concussion: a randomized clinical trial. JAMA Pediatr. 2019;173:319-325. doi: 10.1001/jamapediatrics.2018.4397
Contraception for women taking enzyme-inducing antiepileptics
Topiramate, introduced as an antiepileptic drug (AED), is currently most widely used for prevention of migraine headaches.
Because reproductive-aged women represent a population in which migraines are prevalent, clinicians need guidance to help women taking topiramate make sound contraceptive choices.
Several issues are relevant here. First, women who have migraines with aura should avoid estrogen-containing contraceptive pills, patches, and rings. Instead, progestin-only methods, including the contraceptive implant, may be recommended to patients with migraines.
Second, because topiramate, as with a number of other AEDs, is a teratogen, women using this medication need highly effective contraception. This consideration may also lead clinicians to recommend use of the implant in women with migraines.
Finally, topiramate, along with other AEDs (phenytoin, carbamazepine, barbiturates, primidone, and oxcarbazepine) induces hepatic enzymes, which results in reduced serum contraceptive steroid levels.
Because there is uncertainty regarding the degree to which the use of topiramate reduces serum levels of etonogestrel (the progestin released by the implant), investigators performed a prospective study to assess the pharmacokinetic impact of topiramate in women with the implant.
Ongoing users of contraceptive implants who agreed to use additional nonhormonal contraception were recruited to a 6-week study, during which they took topiramate and periodically had blood drawn.
Overall, use of topiramate was found to lower serum etonogestrel levels from baseline on a dose-related basis. At study completion, almost one-third of study participants were found to have serum progestin levels lower than the threshold associated with predictable ovulation suppression.
The results of this carefully conducted study support guidance from the Centers for Disease Control and Prevention that women seeking contraception and using topiramate or other enzyme-inducing AEDs should be encouraged to use intrauterine devices or injectable contraception. The contraceptive efficacy of these latter methods is not diminished by concomitant use of enzyme inducers.
I am Andrew Kaunitz. Please take care of yourself and each other.
Any views expressed above are the author’s own and do not necessarily reflect the views of WebMD or Medscape.
Andrew M. Kaunitz is a professor and Associate Chairman, department of obstetrics and gynecology, University of Florida, Jacksonville.
A version of this article first appeared on Medscape.com.
Topiramate, introduced as an antiepileptic drug (AED), is currently most widely used for prevention of migraine headaches.
Because reproductive-aged women represent a population in which migraines are prevalent, clinicians need guidance to help women taking topiramate make sound contraceptive choices.
Several issues are relevant here. First, women who have migraines with aura should avoid estrogen-containing contraceptive pills, patches, and rings. Instead, progestin-only methods, including the contraceptive implant, may be recommended to patients with migraines.
Second, because topiramate, as with a number of other AEDs, is a teratogen, women using this medication need highly effective contraception. This consideration may also lead clinicians to recommend use of the implant in women with migraines.
Finally, topiramate, along with other AEDs (phenytoin, carbamazepine, barbiturates, primidone, and oxcarbazepine) induces hepatic enzymes, which results in reduced serum contraceptive steroid levels.
Because there is uncertainty regarding the degree to which the use of topiramate reduces serum levels of etonogestrel (the progestin released by the implant), investigators performed a prospective study to assess the pharmacokinetic impact of topiramate in women with the implant.
Ongoing users of contraceptive implants who agreed to use additional nonhormonal contraception were recruited to a 6-week study, during which they took topiramate and periodically had blood drawn.
Overall, use of topiramate was found to lower serum etonogestrel levels from baseline on a dose-related basis. At study completion, almost one-third of study participants were found to have serum progestin levels lower than the threshold associated with predictable ovulation suppression.
The results of this carefully conducted study support guidance from the Centers for Disease Control and Prevention that women seeking contraception and using topiramate or other enzyme-inducing AEDs should be encouraged to use intrauterine devices or injectable contraception. The contraceptive efficacy of these latter methods is not diminished by concomitant use of enzyme inducers.
I am Andrew Kaunitz. Please take care of yourself and each other.
Any views expressed above are the author’s own and do not necessarily reflect the views of WebMD or Medscape.
Andrew M. Kaunitz is a professor and Associate Chairman, department of obstetrics and gynecology, University of Florida, Jacksonville.
A version of this article first appeared on Medscape.com.
Topiramate, introduced as an antiepileptic drug (AED), is currently most widely used for prevention of migraine headaches.
Because reproductive-aged women represent a population in which migraines are prevalent, clinicians need guidance to help women taking topiramate make sound contraceptive choices.
Several issues are relevant here. First, women who have migraines with aura should avoid estrogen-containing contraceptive pills, patches, and rings. Instead, progestin-only methods, including the contraceptive implant, may be recommended to patients with migraines.
Second, because topiramate, as with a number of other AEDs, is a teratogen, women using this medication need highly effective contraception. This consideration may also lead clinicians to recommend use of the implant in women with migraines.
Finally, topiramate, along with other AEDs (phenytoin, carbamazepine, barbiturates, primidone, and oxcarbazepine) induces hepatic enzymes, which results in reduced serum contraceptive steroid levels.
Because there is uncertainty regarding the degree to which the use of topiramate reduces serum levels of etonogestrel (the progestin released by the implant), investigators performed a prospective study to assess the pharmacokinetic impact of topiramate in women with the implant.
Ongoing users of contraceptive implants who agreed to use additional nonhormonal contraception were recruited to a 6-week study, during which they took topiramate and periodically had blood drawn.
Overall, use of topiramate was found to lower serum etonogestrel levels from baseline on a dose-related basis. At study completion, almost one-third of study participants were found to have serum progestin levels lower than the threshold associated with predictable ovulation suppression.
The results of this carefully conducted study support guidance from the Centers for Disease Control and Prevention that women seeking contraception and using topiramate or other enzyme-inducing AEDs should be encouraged to use intrauterine devices or injectable contraception. The contraceptive efficacy of these latter methods is not diminished by concomitant use of enzyme inducers.
I am Andrew Kaunitz. Please take care of yourself and each other.
Any views expressed above are the author’s own and do not necessarily reflect the views of WebMD or Medscape.
Andrew M. Kaunitz is a professor and Associate Chairman, department of obstetrics and gynecology, University of Florida, Jacksonville.
A version of this article first appeared on Medscape.com.
What can be new about developmental milestones?
The American Academy of Pediatrics, with funding from the Centers for Disease Control and Prevention, studied the CDC’s “Learn the Signs. Act Early” developmental surveillance milestones for children 0-5 years to update the milestones based on published studies. The goal was to improve this tool for developmental surveillance and use by the public. Developmental surveillance is not just observing a child at a check-up but rather “is a longitudinal process that involves eliciting concerns, taking a developmental history based on milestone attainment, observing milestones and other behaviors, examining the child, and applying clinical judgment during health supervision visits (HSVs).”1
While the milestones we were trained on were a good start and highlighted the developmental progression central to pediatrics, they were not based on norms or cut scores indicating significant developmental risk unless taught from a validated tool. The CDC was concerned that their public handouts and apps were based on median ages (middle number of the entire range) of attainment not the mode (most common) or even average ages. That means that about half of all typically developing children would “not have attained” that skill at the age noted, potentially evoking unnecessary concern for parents and a “wait-and-see” message from a knowledgeable provider who realized the statistical meaning and the broad range of normal. Another potential problem with using milestones set at the median age is that parents, especially those with several children or experienced friends, may see the provider as an alarmist when they have seen great variation in children who later were normal. This reaction can dampen provider willingness to discuss development or even to screen with validated tools. We have learned the hard way from COVID-19 that it is difficult to convey concepts of risk effectively both balancing fear and stimulating action.
The AAP experts reviewed the English literature for data-based milestones, finding 34 articles, 10 of which had an opinion for at least one milestone. If this sounds like a very small number, you are correct. You may not realize that almost all screening and diagnostic tools have been based on data collected by Gesell in 1928!2 While most of health care has changed since then, which milestones are measured in infants has not.
The biggest change from this review was deciding to use as milestones skills reported for 75% of children at each age of typical HSVs, adding ones for 15 and 30 months. The implication is that children not attaining these milestones are all at risk and deserving of more careful history, examination, and administration of a validated screening tool; not true when based on median data. Of the 94 existing CDC milestones retained after the review, one-third were moved to a different age with 21 of 31 assigned to an older age. Domains of functioning for the milestones were consolidated into social emotional, cognitive, language/communication, and motor, to help parents learn to distinguish these areas, and, although many milestones reflect several domains, each was included only once to reduce confusion.
Psychosocial assessment is recommended by the AAP and Bright Futures at every HSV but the fewest milestones with normative data were identified for this domain, often self-help rather than social engagement or emotion regulation skills. The cross-cultural study cited for many of the new milestones was reassuring overall in that the median ages for 67%-88% of milestones in most domains were equivalent across the four countries sampled, but only 22% of self-help skills were equivalent.3 This should remind us that parenting has more influence over psychosocial skills than other domains. Psychosocial and behavioral functioning, especially emotional regulation, also deserve “surveillance” as they have enormous impact on life outcomes but need to be measured and supported differently. Routine use of validated tools such as the Early Childhood Screening Assessment or the Ages & Stages Questionnaires: Social-Emotional for these domains are also needed.
Normal variations in temperament and patterns of attachment can affect many milestones including courage for walking, exploration, social engagement, and prosocial behaviors or self-control for social situations, attention, range of affect, and cooperation. All of these skills are among the 42 total (14 new) social-emotional milestones for 0- to 5-year-olds. Variations in these functions are at the root of the most common “challenging behaviors” in our studies in primary care. They are also the most vulnerable to suboptimal parent-child relationships, adverse childhood experiences, and social determinants of health.
As primary care providers, we not only need to detect children at risk for developmental problems but also promote and celebrate developmental progress. I hope that changing the threshold for concern to 75% will allow for a more positive review with the family (as fewer will be flagged as at risk) and chance to congratulate parents on all that is going well. But I also hope the change will not make us overlook parenting challenges, often from the psychosocial milestones most amenable to our guidance and support.
Early identification is mainly important to obtain the early intervention shown to improve outcomes. However, less than 25% of children with delays or disabilities receive early intervention before age 3 and most with emotional, behavioral, and developmental conditions, other than autism spectrum disorder, not before age 5. Since early intervention services are freely available in all states, we also need to do better at getting children to this care.
Let’s reconsider the process of developmental surveillance in this light of delayed referral: “Eliciting concerns” is key as parents have been shown to be usually correct in their worries. Listening to how they express the concerns can help you connect their specific issues when discussing reasons for referral. While most parent “recall of past milestones” is not accurate, current milestones reported are; thus, the need to have the new more accurate norms for all ages for comparison. When we make observations of a child’s abilities and behaviors ourselves we may not only pick up on issues missed by the parent, but will be more convincing in conveying the need for referral when indicated. When we “examine” the child we can use our professional skills to determine the very important risk factor of the quality of how a skill is performed, not just that it is. The recommended “use of validated screening tools” when the new milestones are not met give us an objective tool to share with parents, more confidence in when referral is warranted, which we will convey to parents (and perhaps skeptical relatives), and baseline documentation from which we can “track” referrals, progress, and, hopefully, better outcomes.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. Email her at [email protected].
References
1. Zubler JM et al. Pediatrics. 2022;149(3):e2021052138.
2. Gessell A et al. Macmillan: New York, 1928.
3. Ertem IO et al. Lancet Glob Health. 2018 Mar;6(3):e279-91.
The American Academy of Pediatrics, with funding from the Centers for Disease Control and Prevention, studied the CDC’s “Learn the Signs. Act Early” developmental surveillance milestones for children 0-5 years to update the milestones based on published studies. The goal was to improve this tool for developmental surveillance and use by the public. Developmental surveillance is not just observing a child at a check-up but rather “is a longitudinal process that involves eliciting concerns, taking a developmental history based on milestone attainment, observing milestones and other behaviors, examining the child, and applying clinical judgment during health supervision visits (HSVs).”1
While the milestones we were trained on were a good start and highlighted the developmental progression central to pediatrics, they were not based on norms or cut scores indicating significant developmental risk unless taught from a validated tool. The CDC was concerned that their public handouts and apps were based on median ages (middle number of the entire range) of attainment not the mode (most common) or even average ages. That means that about half of all typically developing children would “not have attained” that skill at the age noted, potentially evoking unnecessary concern for parents and a “wait-and-see” message from a knowledgeable provider who realized the statistical meaning and the broad range of normal. Another potential problem with using milestones set at the median age is that parents, especially those with several children or experienced friends, may see the provider as an alarmist when they have seen great variation in children who later were normal. This reaction can dampen provider willingness to discuss development or even to screen with validated tools. We have learned the hard way from COVID-19 that it is difficult to convey concepts of risk effectively both balancing fear and stimulating action.
The AAP experts reviewed the English literature for data-based milestones, finding 34 articles, 10 of which had an opinion for at least one milestone. If this sounds like a very small number, you are correct. You may not realize that almost all screening and diagnostic tools have been based on data collected by Gesell in 1928!2 While most of health care has changed since then, which milestones are measured in infants has not.
The biggest change from this review was deciding to use as milestones skills reported for 75% of children at each age of typical HSVs, adding ones for 15 and 30 months. The implication is that children not attaining these milestones are all at risk and deserving of more careful history, examination, and administration of a validated screening tool; not true when based on median data. Of the 94 existing CDC milestones retained after the review, one-third were moved to a different age with 21 of 31 assigned to an older age. Domains of functioning for the milestones were consolidated into social emotional, cognitive, language/communication, and motor, to help parents learn to distinguish these areas, and, although many milestones reflect several domains, each was included only once to reduce confusion.
Psychosocial assessment is recommended by the AAP and Bright Futures at every HSV but the fewest milestones with normative data were identified for this domain, often self-help rather than social engagement or emotion regulation skills. The cross-cultural study cited for many of the new milestones was reassuring overall in that the median ages for 67%-88% of milestones in most domains were equivalent across the four countries sampled, but only 22% of self-help skills were equivalent.3 This should remind us that parenting has more influence over psychosocial skills than other domains. Psychosocial and behavioral functioning, especially emotional regulation, also deserve “surveillance” as they have enormous impact on life outcomes but need to be measured and supported differently. Routine use of validated tools such as the Early Childhood Screening Assessment or the Ages & Stages Questionnaires: Social-Emotional for these domains are also needed.
Normal variations in temperament and patterns of attachment can affect many milestones including courage for walking, exploration, social engagement, and prosocial behaviors or self-control for social situations, attention, range of affect, and cooperation. All of these skills are among the 42 total (14 new) social-emotional milestones for 0- to 5-year-olds. Variations in these functions are at the root of the most common “challenging behaviors” in our studies in primary care. They are also the most vulnerable to suboptimal parent-child relationships, adverse childhood experiences, and social determinants of health.
As primary care providers, we not only need to detect children at risk for developmental problems but also promote and celebrate developmental progress. I hope that changing the threshold for concern to 75% will allow for a more positive review with the family (as fewer will be flagged as at risk) and chance to congratulate parents on all that is going well. But I also hope the change will not make us overlook parenting challenges, often from the psychosocial milestones most amenable to our guidance and support.
Early identification is mainly important to obtain the early intervention shown to improve outcomes. However, less than 25% of children with delays or disabilities receive early intervention before age 3 and most with emotional, behavioral, and developmental conditions, other than autism spectrum disorder, not before age 5. Since early intervention services are freely available in all states, we also need to do better at getting children to this care.
Let’s reconsider the process of developmental surveillance in this light of delayed referral: “Eliciting concerns” is key as parents have been shown to be usually correct in their worries. Listening to how they express the concerns can help you connect their specific issues when discussing reasons for referral. While most parent “recall of past milestones” is not accurate, current milestones reported are; thus, the need to have the new more accurate norms for all ages for comparison. When we make observations of a child’s abilities and behaviors ourselves we may not only pick up on issues missed by the parent, but will be more convincing in conveying the need for referral when indicated. When we “examine” the child we can use our professional skills to determine the very important risk factor of the quality of how a skill is performed, not just that it is. The recommended “use of validated screening tools” when the new milestones are not met give us an objective tool to share with parents, more confidence in when referral is warranted, which we will convey to parents (and perhaps skeptical relatives), and baseline documentation from which we can “track” referrals, progress, and, hopefully, better outcomes.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. Email her at [email protected].
References
1. Zubler JM et al. Pediatrics. 2022;149(3):e2021052138.
2. Gessell A et al. Macmillan: New York, 1928.
3. Ertem IO et al. Lancet Glob Health. 2018 Mar;6(3):e279-91.
The American Academy of Pediatrics, with funding from the Centers for Disease Control and Prevention, studied the CDC’s “Learn the Signs. Act Early” developmental surveillance milestones for children 0-5 years to update the milestones based on published studies. The goal was to improve this tool for developmental surveillance and use by the public. Developmental surveillance is not just observing a child at a check-up but rather “is a longitudinal process that involves eliciting concerns, taking a developmental history based on milestone attainment, observing milestones and other behaviors, examining the child, and applying clinical judgment during health supervision visits (HSVs).”1
While the milestones we were trained on were a good start and highlighted the developmental progression central to pediatrics, they were not based on norms or cut scores indicating significant developmental risk unless taught from a validated tool. The CDC was concerned that their public handouts and apps were based on median ages (middle number of the entire range) of attainment not the mode (most common) or even average ages. That means that about half of all typically developing children would “not have attained” that skill at the age noted, potentially evoking unnecessary concern for parents and a “wait-and-see” message from a knowledgeable provider who realized the statistical meaning and the broad range of normal. Another potential problem with using milestones set at the median age is that parents, especially those with several children or experienced friends, may see the provider as an alarmist when they have seen great variation in children who later were normal. This reaction can dampen provider willingness to discuss development or even to screen with validated tools. We have learned the hard way from COVID-19 that it is difficult to convey concepts of risk effectively both balancing fear and stimulating action.
The AAP experts reviewed the English literature for data-based milestones, finding 34 articles, 10 of which had an opinion for at least one milestone. If this sounds like a very small number, you are correct. You may not realize that almost all screening and diagnostic tools have been based on data collected by Gesell in 1928!2 While most of health care has changed since then, which milestones are measured in infants has not.
The biggest change from this review was deciding to use as milestones skills reported for 75% of children at each age of typical HSVs, adding ones for 15 and 30 months. The implication is that children not attaining these milestones are all at risk and deserving of more careful history, examination, and administration of a validated screening tool; not true when based on median data. Of the 94 existing CDC milestones retained after the review, one-third were moved to a different age with 21 of 31 assigned to an older age. Domains of functioning for the milestones were consolidated into social emotional, cognitive, language/communication, and motor, to help parents learn to distinguish these areas, and, although many milestones reflect several domains, each was included only once to reduce confusion.
Psychosocial assessment is recommended by the AAP and Bright Futures at every HSV but the fewest milestones with normative data were identified for this domain, often self-help rather than social engagement or emotion regulation skills. The cross-cultural study cited for many of the new milestones was reassuring overall in that the median ages for 67%-88% of milestones in most domains were equivalent across the four countries sampled, but only 22% of self-help skills were equivalent.3 This should remind us that parenting has more influence over psychosocial skills than other domains. Psychosocial and behavioral functioning, especially emotional regulation, also deserve “surveillance” as they have enormous impact on life outcomes but need to be measured and supported differently. Routine use of validated tools such as the Early Childhood Screening Assessment or the Ages & Stages Questionnaires: Social-Emotional for these domains are also needed.
Normal variations in temperament and patterns of attachment can affect many milestones including courage for walking, exploration, social engagement, and prosocial behaviors or self-control for social situations, attention, range of affect, and cooperation. All of these skills are among the 42 total (14 new) social-emotional milestones for 0- to 5-year-olds. Variations in these functions are at the root of the most common “challenging behaviors” in our studies in primary care. They are also the most vulnerable to suboptimal parent-child relationships, adverse childhood experiences, and social determinants of health.
As primary care providers, we not only need to detect children at risk for developmental problems but also promote and celebrate developmental progress. I hope that changing the threshold for concern to 75% will allow for a more positive review with the family (as fewer will be flagged as at risk) and chance to congratulate parents on all that is going well. But I also hope the change will not make us overlook parenting challenges, often from the psychosocial milestones most amenable to our guidance and support.
Early identification is mainly important to obtain the early intervention shown to improve outcomes. However, less than 25% of children with delays or disabilities receive early intervention before age 3 and most with emotional, behavioral, and developmental conditions, other than autism spectrum disorder, not before age 5. Since early intervention services are freely available in all states, we also need to do better at getting children to this care.
Let’s reconsider the process of developmental surveillance in this light of delayed referral: “Eliciting concerns” is key as parents have been shown to be usually correct in their worries. Listening to how they express the concerns can help you connect their specific issues when discussing reasons for referral. While most parent “recall of past milestones” is not accurate, current milestones reported are; thus, the need to have the new more accurate norms for all ages for comparison. When we make observations of a child’s abilities and behaviors ourselves we may not only pick up on issues missed by the parent, but will be more convincing in conveying the need for referral when indicated. When we “examine” the child we can use our professional skills to determine the very important risk factor of the quality of how a skill is performed, not just that it is. The recommended “use of validated screening tools” when the new milestones are not met give us an objective tool to share with parents, more confidence in when referral is warranted, which we will convey to parents (and perhaps skeptical relatives), and baseline documentation from which we can “track” referrals, progress, and, hopefully, better outcomes.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. Email her at [email protected].
References
1. Zubler JM et al. Pediatrics. 2022;149(3):e2021052138.
2. Gessell A et al. Macmillan: New York, 1928.
3. Ertem IO et al. Lancet Glob Health. 2018 Mar;6(3):e279-91.
Neighborhood-level data sheds new light on racial and ethnic diversity in MS
SEATTLE – These populations often have more severe disease, likely driven by socioeconomic factors and health care access, according to a new study that examined neighborhood-level data and disease severity in the United States.
“It has previously been thought that MS is less common among non-European Caucasian White populations, driven partly by the well-known association of incidence with latitude. It is abundantly clear at this point that this idea is not true,” said Christopher Orlando, MD, during a presentation at the 2022 annual meeting of the American Academy of Neurology.
He noted that several U.S. studies with large sample sizes have shown greater disease severity and a higher disability burden among Hispanic and Black patients. “Black patients in particular appear to have a higher incidence of disease and a greater proportion of progressive disease phenotypes,” said Dr. Orlando.
Race and ethnicity are unlikely explanations for this disparity, according to Dr. Orlando. “While much remains to be discovered of the genetic underpinnings of MS, what we do know does not support the idea that minorities would have a predilection to more severe disease. For example, the well-known high-risk allele HLA DRB1*1501 appears to have a lower frequency in African populations, compared with European [populations].”
Instead, evidence suggests that interrelated social causes include access to resources, environmental exposures, and psychosocial stress. “These affect health via a number of pathways including direct physical injury, allostatic load, and access to health care,” said Dr. Orlando.
Probing racial and ethnic disparities
Previous studies that corrected for social determinants of health such as socioeconomic and insurance status reduce the association between MS disability and race, but they do not completely explain it.
To get a better understanding of the impacts of these factors, researchers have used neighborhood-level data combined with information on socioeconomic status and social deprivation to identify associations with MS severity.
At the conference, Dr. Orlando presented a new study that is the first to use this methodology in the United States, and it is the first to apply it to the study of racial and ethnic disparities in MS.
The study confirmed more severe disability in Hispanic and Black patients than in White patients. Clinical factors associated with more severe disease were similar across the three groups, with some small differences among individual traits. “More stark differences appeared when we compared social determinants of health. Hispanic patients were less likely to speak English as a primary language or to complete 12 years of education. Black patients were less likely to live in a rural county and more likely to be unemployed. One particularly stark difference was in the number of unemployed specifically due to their MS, with only 1 White patient [1.1%], 7 Hispanic patients [7.8%] and 27 Black patients [31.0%],” said Dr. Orlando.
The researchers found that Black and Hispanic patients tend to live in more vulnerable neighborhoods than White patients. The researchers found no significant association between social vulnerability index (SVI) values and MS severity, though there was an association in a separate analysis that only included White patients. The SVI uses 15 measures taken from the U.S. Census to identify communities that might require additional support during natural disasters.
“It would appear that the sheer complexity both in variety and magnitude of the social determinants of health are such that by far the stronger association is with race and ethnicity, which are surrogates for any number of social determinants and societal inequities,” said Dr. Orlando.
What drives the inequity?
Dr. Orlando acknowledged that some might wonder if these results indicate a true biologically intrinsic factor such as genetic predisposition. “I want to warn against that kind of thinking in the strongest possible terms. It is implausible on several levels. It’s not biologically plausible based on our understanding that race and ethnicity are not genetic constructs. And it’s also not numerically plausible based on these data,” said Dr. Orlando.
While some of the drivers of this inequity have been partially examined, many have not been studied. “As long as this is the case, our ability to fulfill our roles as physicians will be limited in several important ways. Our ability to assess our patients’ individual risk will be missing key information, which will limit the efficacy of shared decision-making, which of course is the cornerstone of MS treatment. In addition, we will continue to struggle to include minority patients in our research studies, and the very design and results of those studies may be misguided, as we will either fail to include these populations, or we will fail to adjust for important confounders,” he said.
New answers, new questions
The neighborhood-level data examined by Dr. Orlando’s group “brings extra information in terms of the negative impact of social determinants of health. The disparity seen in neighborhood living is quite striking,” said Lilyana Amezcua, MD, who served as a discussant for Dr. Orlando’s presentation. The study reinforces findings of her own group in Hispanic and Latinx individuals with MS. Some comorbidities are more common among these groups, which is exacerbated by poor health access.
“We have noted that almost 30% of them also have this comorbidity of hypertension, but what is also observed is that only 7% of them are aware [that they have hypertension],” said Dr. Amezcua, who is an associate professor of neurology at the University of Southern California, Los Angeles.
The findings should prompt further research to understand the impact of systemic racism and neighborhood factors, such as disinvestment in the public and private sectors, underresourced hospitals and clinics, and negative infrastructure. “We need to start discussing the (patient’s) environment so we can better understand the community resources they may have available, as well as create innovative transitional care services. We need to also recognize and accept that structural racism and imbalanced distribution of resources and neighborhoods does restrict educational and economic opportunities, as well as health care access and the safety of these marginalized communities,” said Dr. Amezcua.
Dr. Amezcua has consulted for, received speaking fees from, or served on steering committees or advisory boards for Biogen Idec, Novartis, Genentech, and EMD Serono. She has received research support from the Bristol-Myers Squibb Foundation and Biogen Idec. Dr. Orlando has no relevant financial disclosures.
SEATTLE – These populations often have more severe disease, likely driven by socioeconomic factors and health care access, according to a new study that examined neighborhood-level data and disease severity in the United States.
“It has previously been thought that MS is less common among non-European Caucasian White populations, driven partly by the well-known association of incidence with latitude. It is abundantly clear at this point that this idea is not true,” said Christopher Orlando, MD, during a presentation at the 2022 annual meeting of the American Academy of Neurology.
He noted that several U.S. studies with large sample sizes have shown greater disease severity and a higher disability burden among Hispanic and Black patients. “Black patients in particular appear to have a higher incidence of disease and a greater proportion of progressive disease phenotypes,” said Dr. Orlando.
Race and ethnicity are unlikely explanations for this disparity, according to Dr. Orlando. “While much remains to be discovered of the genetic underpinnings of MS, what we do know does not support the idea that minorities would have a predilection to more severe disease. For example, the well-known high-risk allele HLA DRB1*1501 appears to have a lower frequency in African populations, compared with European [populations].”
Instead, evidence suggests that interrelated social causes include access to resources, environmental exposures, and psychosocial stress. “These affect health via a number of pathways including direct physical injury, allostatic load, and access to health care,” said Dr. Orlando.
Probing racial and ethnic disparities
Previous studies that corrected for social determinants of health such as socioeconomic and insurance status reduce the association between MS disability and race, but they do not completely explain it.
To get a better understanding of the impacts of these factors, researchers have used neighborhood-level data combined with information on socioeconomic status and social deprivation to identify associations with MS severity.
At the conference, Dr. Orlando presented a new study that is the first to use this methodology in the United States, and it is the first to apply it to the study of racial and ethnic disparities in MS.
The study confirmed more severe disability in Hispanic and Black patients than in White patients. Clinical factors associated with more severe disease were similar across the three groups, with some small differences among individual traits. “More stark differences appeared when we compared social determinants of health. Hispanic patients were less likely to speak English as a primary language or to complete 12 years of education. Black patients were less likely to live in a rural county and more likely to be unemployed. One particularly stark difference was in the number of unemployed specifically due to their MS, with only 1 White patient [1.1%], 7 Hispanic patients [7.8%] and 27 Black patients [31.0%],” said Dr. Orlando.
The researchers found that Black and Hispanic patients tend to live in more vulnerable neighborhoods than White patients. The researchers found no significant association between social vulnerability index (SVI) values and MS severity, though there was an association in a separate analysis that only included White patients. The SVI uses 15 measures taken from the U.S. Census to identify communities that might require additional support during natural disasters.
“It would appear that the sheer complexity both in variety and magnitude of the social determinants of health are such that by far the stronger association is with race and ethnicity, which are surrogates for any number of social determinants and societal inequities,” said Dr. Orlando.
What drives the inequity?
Dr. Orlando acknowledged that some might wonder if these results indicate a true biologically intrinsic factor such as genetic predisposition. “I want to warn against that kind of thinking in the strongest possible terms. It is implausible on several levels. It’s not biologically plausible based on our understanding that race and ethnicity are not genetic constructs. And it’s also not numerically plausible based on these data,” said Dr. Orlando.
While some of the drivers of this inequity have been partially examined, many have not been studied. “As long as this is the case, our ability to fulfill our roles as physicians will be limited in several important ways. Our ability to assess our patients’ individual risk will be missing key information, which will limit the efficacy of shared decision-making, which of course is the cornerstone of MS treatment. In addition, we will continue to struggle to include minority patients in our research studies, and the very design and results of those studies may be misguided, as we will either fail to include these populations, or we will fail to adjust for important confounders,” he said.
New answers, new questions
The neighborhood-level data examined by Dr. Orlando’s group “brings extra information in terms of the negative impact of social determinants of health. The disparity seen in neighborhood living is quite striking,” said Lilyana Amezcua, MD, who served as a discussant for Dr. Orlando’s presentation. The study reinforces findings of her own group in Hispanic and Latinx individuals with MS. Some comorbidities are more common among these groups, which is exacerbated by poor health access.
“We have noted that almost 30% of them also have this comorbidity of hypertension, but what is also observed is that only 7% of them are aware [that they have hypertension],” said Dr. Amezcua, who is an associate professor of neurology at the University of Southern California, Los Angeles.
The findings should prompt further research to understand the impact of systemic racism and neighborhood factors, such as disinvestment in the public and private sectors, underresourced hospitals and clinics, and negative infrastructure. “We need to start discussing the (patient’s) environment so we can better understand the community resources they may have available, as well as create innovative transitional care services. We need to also recognize and accept that structural racism and imbalanced distribution of resources and neighborhoods does restrict educational and economic opportunities, as well as health care access and the safety of these marginalized communities,” said Dr. Amezcua.
Dr. Amezcua has consulted for, received speaking fees from, or served on steering committees or advisory boards for Biogen Idec, Novartis, Genentech, and EMD Serono. She has received research support from the Bristol-Myers Squibb Foundation and Biogen Idec. Dr. Orlando has no relevant financial disclosures.
SEATTLE – These populations often have more severe disease, likely driven by socioeconomic factors and health care access, according to a new study that examined neighborhood-level data and disease severity in the United States.
“It has previously been thought that MS is less common among non-European Caucasian White populations, driven partly by the well-known association of incidence with latitude. It is abundantly clear at this point that this idea is not true,” said Christopher Orlando, MD, during a presentation at the 2022 annual meeting of the American Academy of Neurology.
He noted that several U.S. studies with large sample sizes have shown greater disease severity and a higher disability burden among Hispanic and Black patients. “Black patients in particular appear to have a higher incidence of disease and a greater proportion of progressive disease phenotypes,” said Dr. Orlando.
Race and ethnicity are unlikely explanations for this disparity, according to Dr. Orlando. “While much remains to be discovered of the genetic underpinnings of MS, what we do know does not support the idea that minorities would have a predilection to more severe disease. For example, the well-known high-risk allele HLA DRB1*1501 appears to have a lower frequency in African populations, compared with European [populations].”
Instead, evidence suggests that interrelated social causes include access to resources, environmental exposures, and psychosocial stress. “These affect health via a number of pathways including direct physical injury, allostatic load, and access to health care,” said Dr. Orlando.
Probing racial and ethnic disparities
Previous studies that corrected for social determinants of health such as socioeconomic and insurance status reduce the association between MS disability and race, but they do not completely explain it.
To get a better understanding of the impacts of these factors, researchers have used neighborhood-level data combined with information on socioeconomic status and social deprivation to identify associations with MS severity.
At the conference, Dr. Orlando presented a new study that is the first to use this methodology in the United States, and it is the first to apply it to the study of racial and ethnic disparities in MS.
The study confirmed more severe disability in Hispanic and Black patients than in White patients. Clinical factors associated with more severe disease were similar across the three groups, with some small differences among individual traits. “More stark differences appeared when we compared social determinants of health. Hispanic patients were less likely to speak English as a primary language or to complete 12 years of education. Black patients were less likely to live in a rural county and more likely to be unemployed. One particularly stark difference was in the number of unemployed specifically due to their MS, with only 1 White patient [1.1%], 7 Hispanic patients [7.8%] and 27 Black patients [31.0%],” said Dr. Orlando.
The researchers found that Black and Hispanic patients tend to live in more vulnerable neighborhoods than White patients. The researchers found no significant association between social vulnerability index (SVI) values and MS severity, though there was an association in a separate analysis that only included White patients. The SVI uses 15 measures taken from the U.S. Census to identify communities that might require additional support during natural disasters.
“It would appear that the sheer complexity both in variety and magnitude of the social determinants of health are such that by far the stronger association is with race and ethnicity, which are surrogates for any number of social determinants and societal inequities,” said Dr. Orlando.
What drives the inequity?
Dr. Orlando acknowledged that some might wonder if these results indicate a true biologically intrinsic factor such as genetic predisposition. “I want to warn against that kind of thinking in the strongest possible terms. It is implausible on several levels. It’s not biologically plausible based on our understanding that race and ethnicity are not genetic constructs. And it’s also not numerically plausible based on these data,” said Dr. Orlando.
While some of the drivers of this inequity have been partially examined, many have not been studied. “As long as this is the case, our ability to fulfill our roles as physicians will be limited in several important ways. Our ability to assess our patients’ individual risk will be missing key information, which will limit the efficacy of shared decision-making, which of course is the cornerstone of MS treatment. In addition, we will continue to struggle to include minority patients in our research studies, and the very design and results of those studies may be misguided, as we will either fail to include these populations, or we will fail to adjust for important confounders,” he said.
New answers, new questions
The neighborhood-level data examined by Dr. Orlando’s group “brings extra information in terms of the negative impact of social determinants of health. The disparity seen in neighborhood living is quite striking,” said Lilyana Amezcua, MD, who served as a discussant for Dr. Orlando’s presentation. The study reinforces findings of her own group in Hispanic and Latinx individuals with MS. Some comorbidities are more common among these groups, which is exacerbated by poor health access.
“We have noted that almost 30% of them also have this comorbidity of hypertension, but what is also observed is that only 7% of them are aware [that they have hypertension],” said Dr. Amezcua, who is an associate professor of neurology at the University of Southern California, Los Angeles.
The findings should prompt further research to understand the impact of systemic racism and neighborhood factors, such as disinvestment in the public and private sectors, underresourced hospitals and clinics, and negative infrastructure. “We need to start discussing the (patient’s) environment so we can better understand the community resources they may have available, as well as create innovative transitional care services. We need to also recognize and accept that structural racism and imbalanced distribution of resources and neighborhoods does restrict educational and economic opportunities, as well as health care access and the safety of these marginalized communities,” said Dr. Amezcua.
Dr. Amezcua has consulted for, received speaking fees from, or served on steering committees or advisory boards for Biogen Idec, Novartis, Genentech, and EMD Serono. She has received research support from the Bristol-Myers Squibb Foundation and Biogen Idec. Dr. Orlando has no relevant financial disclosures.
AT AAN 2022
Study: Physical fitness in children linked with concentration, quality of life
The findings of the German study involving more than 6,500 kids emphasize the importance of cardiorespiratory health in childhood, and support physical fitness initiatives in schools, according to lead author Katharina Köble, MSc, of the Technical University of Munich (Germany), and colleagues.
“Recent studies show that only a few children meet the recommendations of physical activity,” the investigators wrote in Journal of Clinical Medicine.
While the health benefits of physical activity are clearly documented, Ms. Köble and colleagues noted that typical measures of activity, such as accelerometers or self-reported questionnaires, are suboptimal research tools.
“Physical fitness is a more objective parameter to quantify when evaluating health promotion,” the investigators wrote. “Furthermore, cardiorespiratory fitness as part of physical fitness is more strongly related to risk factors of cardiovascular disease than physical activity.”
According to the investigators, physical fitness has also been linked with better concentration and HRQOL, but never in the same population of children.
The new study aimed to address this knowledge gap by assessing 6,533 healthy children aged 6-10 years, approximately half boys and half girls. Associations between physical fitness, concentration, and HRQOL were evaluated using multiple linear regression analysis in participants aged 9-10 years.
Physical fitness was measured using a series of challenges, including curl-ups (pull-ups with palms facing body), push-ups, standing long jump, handgrip strength measurement, and Progressive Aerobic Cardiovascular Endurance Run (PACER). Performing the multistage shuttle run, PACER, “requires participants to maintain the pace set by an audio signal, which progressively increases the intensity every minute.” Results of the PACER test were used to estimate VO2max.
Concentration was measured using the d2-R test, “a paper-pencil cancellation test, where subjects have to cross out all ‘d’ letters with two dashes under a time limit.”
HRQOL was evaluated with the KINDL questionnaire, which covers emotional well-being, physical well-being, everyday functioning (school), friends, family, and self-esteem.
Analysis showed that physical fitness improved with age (P < .001), except for VO2max in girls (P = .129). Concentration also improved with age (P < .001), while HRQOL did not (P = .179).
Among children aged 9-10 years, VO2max scores were strongly associated with both HRQOL (P < .001) and concentration (P < .001).
“VO2max was found to be one of the main factors influencing concentration levels and HRQOL dimensions in primary school children,” the investigators wrote. “Physical fitness, especially cardiorespiratory performance, should therefore be promoted more specifically in school settings to support the promotion of an overall healthy lifestyle in children and adolescents.”
Findings are having a real-word impact, according to researcher
In an interview, Ms. Köble noted that the findings are already having a real-world impact.
“We continued data assessment in the long-term and specifically adapted prevention programs in school to the needs of the school children we identified in our study,” she said. “Schools are partially offering specific movement and nutrition classes now.”
In addition, Ms. Köble and colleagues plan on educating teachers about the “urgent need for sufficient physical activity.”
“Academic performance should be considered as an additional health factor in future studies, as well as screen time and eating patterns, as all those variables showed interactions with physical fitness and concentration. In a subanalysis, we showed that children with better physical fitness and concentration values were those who usually went to higher education secondary schools,” they wrote.
VO2max did not correlate with BMI
Gregory Weaver, MD, a pediatrician at Cleveland Clinic Children’s, voiced some concerns about the reliability of the findings. He noted that VO2max did not correlate with body mass index or other measures of physical fitness, and that using the PACER test to estimate VO2max may have skewed the association between physical fitness and concentration.
“It is quite conceivable that children who can maintain the focus to perform maximally on this test will also do well on other tests of attention/concentration,” Dr. Weaver said. “Most children I know would have a very difficult time performing a physical fitness test which requires them to match a recorded pace that slowly increases overtime. I’m not an expert in the area, but it is my understanding that usually VO2max tests involve a treadmill which allows investigators to have complete control over pace.”
Dr. Weaver concluded that more work is needed to determine if physical fitness interventions can have a positive impact on HRQOL and concentration.
“I think the authors of this study attempted to ask an important question about the possible association between physical fitness and concentration among school aged children,” Dr. Weaver said in an interview. “But what is even more vital are studies demonstrating that a change in modifiable health factors like nutrition, physical fitness, or the built environment can improve quality of life. I was hoping the authors would show that an improvement in VO2max over time resulted in an improvement in concentration. Frustratingly, that is not what this article demonstrates.”
The investigators and Dr. Weaver reported no conflicts of interest.
The findings of the German study involving more than 6,500 kids emphasize the importance of cardiorespiratory health in childhood, and support physical fitness initiatives in schools, according to lead author Katharina Köble, MSc, of the Technical University of Munich (Germany), and colleagues.
“Recent studies show that only a few children meet the recommendations of physical activity,” the investigators wrote in Journal of Clinical Medicine.
While the health benefits of physical activity are clearly documented, Ms. Köble and colleagues noted that typical measures of activity, such as accelerometers or self-reported questionnaires, are suboptimal research tools.
“Physical fitness is a more objective parameter to quantify when evaluating health promotion,” the investigators wrote. “Furthermore, cardiorespiratory fitness as part of physical fitness is more strongly related to risk factors of cardiovascular disease than physical activity.”
According to the investigators, physical fitness has also been linked with better concentration and HRQOL, but never in the same population of children.
The new study aimed to address this knowledge gap by assessing 6,533 healthy children aged 6-10 years, approximately half boys and half girls. Associations between physical fitness, concentration, and HRQOL were evaluated using multiple linear regression analysis in participants aged 9-10 years.
Physical fitness was measured using a series of challenges, including curl-ups (pull-ups with palms facing body), push-ups, standing long jump, handgrip strength measurement, and Progressive Aerobic Cardiovascular Endurance Run (PACER). Performing the multistage shuttle run, PACER, “requires participants to maintain the pace set by an audio signal, which progressively increases the intensity every minute.” Results of the PACER test were used to estimate VO2max.
Concentration was measured using the d2-R test, “a paper-pencil cancellation test, where subjects have to cross out all ‘d’ letters with two dashes under a time limit.”
HRQOL was evaluated with the KINDL questionnaire, which covers emotional well-being, physical well-being, everyday functioning (school), friends, family, and self-esteem.
Analysis showed that physical fitness improved with age (P < .001), except for VO2max in girls (P = .129). Concentration also improved with age (P < .001), while HRQOL did not (P = .179).
Among children aged 9-10 years, VO2max scores were strongly associated with both HRQOL (P < .001) and concentration (P < .001).
“VO2max was found to be one of the main factors influencing concentration levels and HRQOL dimensions in primary school children,” the investigators wrote. “Physical fitness, especially cardiorespiratory performance, should therefore be promoted more specifically in school settings to support the promotion of an overall healthy lifestyle in children and adolescents.”
Findings are having a real-word impact, according to researcher
In an interview, Ms. Köble noted that the findings are already having a real-world impact.
“We continued data assessment in the long-term and specifically adapted prevention programs in school to the needs of the school children we identified in our study,” she said. “Schools are partially offering specific movement and nutrition classes now.”
In addition, Ms. Köble and colleagues plan on educating teachers about the “urgent need for sufficient physical activity.”
“Academic performance should be considered as an additional health factor in future studies, as well as screen time and eating patterns, as all those variables showed interactions with physical fitness and concentration. In a subanalysis, we showed that children with better physical fitness and concentration values were those who usually went to higher education secondary schools,” they wrote.
VO2max did not correlate with BMI
Gregory Weaver, MD, a pediatrician at Cleveland Clinic Children’s, voiced some concerns about the reliability of the findings. He noted that VO2max did not correlate with body mass index or other measures of physical fitness, and that using the PACER test to estimate VO2max may have skewed the association between physical fitness and concentration.
“It is quite conceivable that children who can maintain the focus to perform maximally on this test will also do well on other tests of attention/concentration,” Dr. Weaver said. “Most children I know would have a very difficult time performing a physical fitness test which requires them to match a recorded pace that slowly increases overtime. I’m not an expert in the area, but it is my understanding that usually VO2max tests involve a treadmill which allows investigators to have complete control over pace.”
Dr. Weaver concluded that more work is needed to determine if physical fitness interventions can have a positive impact on HRQOL and concentration.
“I think the authors of this study attempted to ask an important question about the possible association between physical fitness and concentration among school aged children,” Dr. Weaver said in an interview. “But what is even more vital are studies demonstrating that a change in modifiable health factors like nutrition, physical fitness, or the built environment can improve quality of life. I was hoping the authors would show that an improvement in VO2max over time resulted in an improvement in concentration. Frustratingly, that is not what this article demonstrates.”
The investigators and Dr. Weaver reported no conflicts of interest.
The findings of the German study involving more than 6,500 kids emphasize the importance of cardiorespiratory health in childhood, and support physical fitness initiatives in schools, according to lead author Katharina Köble, MSc, of the Technical University of Munich (Germany), and colleagues.
“Recent studies show that only a few children meet the recommendations of physical activity,” the investigators wrote in Journal of Clinical Medicine.
While the health benefits of physical activity are clearly documented, Ms. Köble and colleagues noted that typical measures of activity, such as accelerometers or self-reported questionnaires, are suboptimal research tools.
“Physical fitness is a more objective parameter to quantify when evaluating health promotion,” the investigators wrote. “Furthermore, cardiorespiratory fitness as part of physical fitness is more strongly related to risk factors of cardiovascular disease than physical activity.”
According to the investigators, physical fitness has also been linked with better concentration and HRQOL, but never in the same population of children.
The new study aimed to address this knowledge gap by assessing 6,533 healthy children aged 6-10 years, approximately half boys and half girls. Associations between physical fitness, concentration, and HRQOL were evaluated using multiple linear regression analysis in participants aged 9-10 years.
Physical fitness was measured using a series of challenges, including curl-ups (pull-ups with palms facing body), push-ups, standing long jump, handgrip strength measurement, and Progressive Aerobic Cardiovascular Endurance Run (PACER). Performing the multistage shuttle run, PACER, “requires participants to maintain the pace set by an audio signal, which progressively increases the intensity every minute.” Results of the PACER test were used to estimate VO2max.
Concentration was measured using the d2-R test, “a paper-pencil cancellation test, where subjects have to cross out all ‘d’ letters with two dashes under a time limit.”
HRQOL was evaluated with the KINDL questionnaire, which covers emotional well-being, physical well-being, everyday functioning (school), friends, family, and self-esteem.
Analysis showed that physical fitness improved with age (P < .001), except for VO2max in girls (P = .129). Concentration also improved with age (P < .001), while HRQOL did not (P = .179).
Among children aged 9-10 years, VO2max scores were strongly associated with both HRQOL (P < .001) and concentration (P < .001).
“VO2max was found to be one of the main factors influencing concentration levels and HRQOL dimensions in primary school children,” the investigators wrote. “Physical fitness, especially cardiorespiratory performance, should therefore be promoted more specifically in school settings to support the promotion of an overall healthy lifestyle in children and adolescents.”
Findings are having a real-word impact, according to researcher
In an interview, Ms. Köble noted that the findings are already having a real-world impact.
“We continued data assessment in the long-term and specifically adapted prevention programs in school to the needs of the school children we identified in our study,” she said. “Schools are partially offering specific movement and nutrition classes now.”
In addition, Ms. Köble and colleagues plan on educating teachers about the “urgent need for sufficient physical activity.”
“Academic performance should be considered as an additional health factor in future studies, as well as screen time and eating patterns, as all those variables showed interactions with physical fitness and concentration. In a subanalysis, we showed that children with better physical fitness and concentration values were those who usually went to higher education secondary schools,” they wrote.
VO2max did not correlate with BMI
Gregory Weaver, MD, a pediatrician at Cleveland Clinic Children’s, voiced some concerns about the reliability of the findings. He noted that VO2max did not correlate with body mass index or other measures of physical fitness, and that using the PACER test to estimate VO2max may have skewed the association between physical fitness and concentration.
“It is quite conceivable that children who can maintain the focus to perform maximally on this test will also do well on other tests of attention/concentration,” Dr. Weaver said. “Most children I know would have a very difficult time performing a physical fitness test which requires them to match a recorded pace that slowly increases overtime. I’m not an expert in the area, but it is my understanding that usually VO2max tests involve a treadmill which allows investigators to have complete control over pace.”
Dr. Weaver concluded that more work is needed to determine if physical fitness interventions can have a positive impact on HRQOL and concentration.
“I think the authors of this study attempted to ask an important question about the possible association between physical fitness and concentration among school aged children,” Dr. Weaver said in an interview. “But what is even more vital are studies demonstrating that a change in modifiable health factors like nutrition, physical fitness, or the built environment can improve quality of life. I was hoping the authors would show that an improvement in VO2max over time resulted in an improvement in concentration. Frustratingly, that is not what this article demonstrates.”
The investigators and Dr. Weaver reported no conflicts of interest.
FROM THE JOURNAL OF CLINICAL MEDICINE
DIAMOND: Adding patiromer helps optimize HF meds, foils hyperkalemia
Several of the core medications for patients with heart failure with reduced ejection fraction (HFrEF) come with a well-known risk of causing hyperkalemia, to which many clinicians respond by pulling back on dosing or withdrawing the culprit drug.
But accompanying renin-angiotensin system–inhibiting agents with the potassium-sequestrant patiromer (Veltassa, Vifor Pharma) appears to shield patients against hyperkalemia enough that they can take more RASI medications at higher doses, suggests a randomized, a controlled study.
The DIAMOND trial’s HFrEF patients, who had current or a history of RASI-related hyperkalemia, added either patiromer or placebo to their guideline-directed medical therapy (GDMT), which includes, even emphasizes, the culprit medication. They include ACE inhibitors, angiotensin-receptor blockers (ARBs), angiotensin-receptor/neprilysin inhibitors (ARNIs), and mineralocorticoid receptor antagonists (MRAs).
Those taking patiromer tolerated more intense RASI therapy – including MRAs, which are especially prone to causing hyperkalemia – than the patients assigned to placebo. They also maintained lower potassium concentrations and experienced fewer clinically important hyperkalemia episodes, reported Javed Butler, MD, MPH, MBA, Baylor Scott and White Research Institute, Dallas, at the annual scientific sessions of the American College of Cardiology.
The apparent benefit from patiromer came in part from an advantage for a composite hyperkalemia-event endpoint that included mortality, Dr. Butler noted. That advantage seemed to hold regardless of age, sex, body mass index, HFrEF symptom severity, or initial natriuretic peptide levels.
Patients who took patiromer, compared with those who took placebo, showed a 37% reduction in risk for hyperkalemia (P = .006), defined as potassium levels exceeding 5.5 mEq/L, over a median follow-up of 27 weeks. They were 38% less likely to have their MRA dosage reduced to below target level (P = .006).
More patients in the patiromer group than in the control group attained at least 50% of target dosage for MRAs and ACE inhibitors, ARBs, or ARNIs (92% vs. 87%; P = .015).
Patients with HFrEF are unlikely to achieve best possible outcomes without GDMT optimization, but failure to optimize is often attributed to hyperkalemia concerns. DIAMOND, Dr. Butler said, suggests that, by adding the potassium sequestrant to GDMT, “you can simultaneously control potassium and optimize RASI therapy.” Many clinicians seem to believe they can achieve only one or the other.
DIAMOND was too underpowered to show whether preventing hyperkalemia with patiromer could improve clinical outcomes. But failure to optimize RASI medication in HFrEF can worsen risk for heart failure events and death. So “it stands to reason that optimization of RASI therapy without a concomitant risk of hyperkalemia may, in the long run, lead to better outcomes for these patients,” Dr. Butler said in an interview.
Given the drug’s ability to keep potassium levels in check during RASI therapy, Dr. Butler said, “hypokalemia should not be a reason for suboptimal therapy.”
Patiromer and other potassium sequestrants have been available in the United States and Europe for 4-6 years, but their value as adjuncts to RASI medication in HFrEF or other heart failure has been unclear.
“There’s a good opportunity to expand the use of the drug. The question is, in whom and when?” James L. Januzzi, MD, Massachusetts General Hospital, Boston, said in an interview.
Some HFrEF patients on GDMT “should be treated with patiromer. The bigger question is, should we give someone who has a history of hyperkalemia another chance at GDMT before we treat them with patiromer? Because they may not necessarily develop hyperkalemia a second time,” said Dr. Januzzi, who was on the DIAMOND endpoint-adjudication committee.
Among the most notable findings of the trial, he said, is that the number of people who developed hyperkalemia on RASI medication, although significantly elevated, “wasn’t as high as they expected it would be,” he said. “The data from DIAMOND argue that if a really significant majority does not become hyperkalemic on rechallenge, jumping straight to a potassium-binding drug may be premature.”
Physicians across specialties can differ in how they interpret potassium-level elevation and can use various cut points to flag when to stop RASI medication or at least hold back on up-titration, Dr. Butler observed. “Cardiologists have a different threshold of potassium that they tolerate than say, for instance, a nephrologist.”
Useful, then, might be a way to tell which patients are most likely to develop hyperkalemia with RASI up-titration and so might benefit from a potassium-binding agent right away. But DIAMOND, Dr. Butler said, “does not necessarily define any patient phenotype or any potassium level where we would say that you should use a potassium binder.”
The trial entered 1,642 patients with HFrEF and current or past RASI-related hyperkalemia to a 12-week run-in phase for optimization of GDMT with patiromer. The trial was conducted at nearly 400 centers in 21 countries.
RASI medication could be optimized in 85% of the cohort, from which 878 patients were randomly assigned either to continue optimized GDMT with patiromer or to have the potassium-sequestrant replaced with a placebo.
The patients on patiromer showed a 0.03-mEq/L mean rise in serum potassium levels from randomization to the end of the study, the primary endpoint, compared with a 0.13 mEq/L mean increase for those in the control group (P < .001), Dr. Butler reported.
The win ratio for a RASI-use score hierarchically featuring cardiovascular death and CV hospitalization for hyperkalemia at several levels of severity was 1.25 (95% confidence interval, 1.003-1.564; P = .048), favoring the patiromer group. The win ratio solely for hyperkalemia-related events also favored patients on patiromer, at 1.53 (95% CI, 1.23-1.91; P < .001).
Patiromer also seemed well tolerated, Dr. Butler said.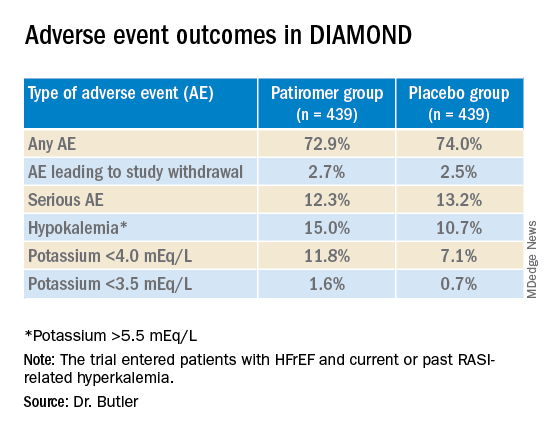
Hyperkalemia is “one of the most common excuses” from clinicians for failing to up-titrate RASI medicine in patients with heart failure, Dr. Januzzi said. DIAMOND was less about patiromer itself than about ways “to facilitate better GDMT, where we’re really falling short of the mark. During the run-in phase they were able to get the vast majority of individuals to target, which to me is a critically important point, and emblematic of the need for things that facilitate this kind of excellent care.”
DIAMOND was funded by Vifor Pharma. Dr. Butler disclosed receiving consulting fees from Abbott, Adrenomed, Amgen, Applied Therapeutics, Array, AstraZeneca, Bayer, Boehringer Ingelheim, CVRx, G3 Pharma, Impulse Dynamics, Innolife, Janssen, LivaNova, Luitpold, Medtronic, Merck, Novartis, Novo Nordisk, Relypsa, Sequana Medical, and Vifor Pharma. Dr. Januzzi disclosed receiving consultant fees or honoraria from Abbott Laboratories, Imbria, Jana Care, Novartis, Prevencio, and Roche Diagnostics; serving on a data safety monitoring board for AbbVie, Amgen, Bayer Healthcare Pharmaceuticals, Beyer, CVRx, and Takeda Pharmaceuticals North America; and receiving research grants from Abbott Laboratories, Janssen, and Vifor Pharma.
A version of this article first appeared on Medscape.com.
Several of the core medications for patients with heart failure with reduced ejection fraction (HFrEF) come with a well-known risk of causing hyperkalemia, to which many clinicians respond by pulling back on dosing or withdrawing the culprit drug.
But accompanying renin-angiotensin system–inhibiting agents with the potassium-sequestrant patiromer (Veltassa, Vifor Pharma) appears to shield patients against hyperkalemia enough that they can take more RASI medications at higher doses, suggests a randomized, a controlled study.
The DIAMOND trial’s HFrEF patients, who had current or a history of RASI-related hyperkalemia, added either patiromer or placebo to their guideline-directed medical therapy (GDMT), which includes, even emphasizes, the culprit medication. They include ACE inhibitors, angiotensin-receptor blockers (ARBs), angiotensin-receptor/neprilysin inhibitors (ARNIs), and mineralocorticoid receptor antagonists (MRAs).
Those taking patiromer tolerated more intense RASI therapy – including MRAs, which are especially prone to causing hyperkalemia – than the patients assigned to placebo. They also maintained lower potassium concentrations and experienced fewer clinically important hyperkalemia episodes, reported Javed Butler, MD, MPH, MBA, Baylor Scott and White Research Institute, Dallas, at the annual scientific sessions of the American College of Cardiology.
The apparent benefit from patiromer came in part from an advantage for a composite hyperkalemia-event endpoint that included mortality, Dr. Butler noted. That advantage seemed to hold regardless of age, sex, body mass index, HFrEF symptom severity, or initial natriuretic peptide levels.
Patients who took patiromer, compared with those who took placebo, showed a 37% reduction in risk for hyperkalemia (P = .006), defined as potassium levels exceeding 5.5 mEq/L, over a median follow-up of 27 weeks. They were 38% less likely to have their MRA dosage reduced to below target level (P = .006).
More patients in the patiromer group than in the control group attained at least 50% of target dosage for MRAs and ACE inhibitors, ARBs, or ARNIs (92% vs. 87%; P = .015).
Patients with HFrEF are unlikely to achieve best possible outcomes without GDMT optimization, but failure to optimize is often attributed to hyperkalemia concerns. DIAMOND, Dr. Butler said, suggests that, by adding the potassium sequestrant to GDMT, “you can simultaneously control potassium and optimize RASI therapy.” Many clinicians seem to believe they can achieve only one or the other.
DIAMOND was too underpowered to show whether preventing hyperkalemia with patiromer could improve clinical outcomes. But failure to optimize RASI medication in HFrEF can worsen risk for heart failure events and death. So “it stands to reason that optimization of RASI therapy without a concomitant risk of hyperkalemia may, in the long run, lead to better outcomes for these patients,” Dr. Butler said in an interview.
Given the drug’s ability to keep potassium levels in check during RASI therapy, Dr. Butler said, “hypokalemia should not be a reason for suboptimal therapy.”
Patiromer and other potassium sequestrants have been available in the United States and Europe for 4-6 years, but their value as adjuncts to RASI medication in HFrEF or other heart failure has been unclear.
“There’s a good opportunity to expand the use of the drug. The question is, in whom and when?” James L. Januzzi, MD, Massachusetts General Hospital, Boston, said in an interview.
Some HFrEF patients on GDMT “should be treated with patiromer. The bigger question is, should we give someone who has a history of hyperkalemia another chance at GDMT before we treat them with patiromer? Because they may not necessarily develop hyperkalemia a second time,” said Dr. Januzzi, who was on the DIAMOND endpoint-adjudication committee.
Among the most notable findings of the trial, he said, is that the number of people who developed hyperkalemia on RASI medication, although significantly elevated, “wasn’t as high as they expected it would be,” he said. “The data from DIAMOND argue that if a really significant majority does not become hyperkalemic on rechallenge, jumping straight to a potassium-binding drug may be premature.”
Physicians across specialties can differ in how they interpret potassium-level elevation and can use various cut points to flag when to stop RASI medication or at least hold back on up-titration, Dr. Butler observed. “Cardiologists have a different threshold of potassium that they tolerate than say, for instance, a nephrologist.”
Useful, then, might be a way to tell which patients are most likely to develop hyperkalemia with RASI up-titration and so might benefit from a potassium-binding agent right away. But DIAMOND, Dr. Butler said, “does not necessarily define any patient phenotype or any potassium level where we would say that you should use a potassium binder.”
The trial entered 1,642 patients with HFrEF and current or past RASI-related hyperkalemia to a 12-week run-in phase for optimization of GDMT with patiromer. The trial was conducted at nearly 400 centers in 21 countries.
RASI medication could be optimized in 85% of the cohort, from which 878 patients were randomly assigned either to continue optimized GDMT with patiromer or to have the potassium-sequestrant replaced with a placebo.
The patients on patiromer showed a 0.03-mEq/L mean rise in serum potassium levels from randomization to the end of the study, the primary endpoint, compared with a 0.13 mEq/L mean increase for those in the control group (P < .001), Dr. Butler reported.
The win ratio for a RASI-use score hierarchically featuring cardiovascular death and CV hospitalization for hyperkalemia at several levels of severity was 1.25 (95% confidence interval, 1.003-1.564; P = .048), favoring the patiromer group. The win ratio solely for hyperkalemia-related events also favored patients on patiromer, at 1.53 (95% CI, 1.23-1.91; P < .001).
Patiromer also seemed well tolerated, Dr. Butler said.
Hyperkalemia is “one of the most common excuses” from clinicians for failing to up-titrate RASI medicine in patients with heart failure, Dr. Januzzi said. DIAMOND was less about patiromer itself than about ways “to facilitate better GDMT, where we’re really falling short of the mark. During the run-in phase they were able to get the vast majority of individuals to target, which to me is a critically important point, and emblematic of the need for things that facilitate this kind of excellent care.”
DIAMOND was funded by Vifor Pharma. Dr. Butler disclosed receiving consulting fees from Abbott, Adrenomed, Amgen, Applied Therapeutics, Array, AstraZeneca, Bayer, Boehringer Ingelheim, CVRx, G3 Pharma, Impulse Dynamics, Innolife, Janssen, LivaNova, Luitpold, Medtronic, Merck, Novartis, Novo Nordisk, Relypsa, Sequana Medical, and Vifor Pharma. Dr. Januzzi disclosed receiving consultant fees or honoraria from Abbott Laboratories, Imbria, Jana Care, Novartis, Prevencio, and Roche Diagnostics; serving on a data safety monitoring board for AbbVie, Amgen, Bayer Healthcare Pharmaceuticals, Beyer, CVRx, and Takeda Pharmaceuticals North America; and receiving research grants from Abbott Laboratories, Janssen, and Vifor Pharma.
A version of this article first appeared on Medscape.com.
Several of the core medications for patients with heart failure with reduced ejection fraction (HFrEF) come with a well-known risk of causing hyperkalemia, to which many clinicians respond by pulling back on dosing or withdrawing the culprit drug.
But accompanying renin-angiotensin system–inhibiting agents with the potassium-sequestrant patiromer (Veltassa, Vifor Pharma) appears to shield patients against hyperkalemia enough that they can take more RASI medications at higher doses, suggests a randomized, a controlled study.
The DIAMOND trial’s HFrEF patients, who had current or a history of RASI-related hyperkalemia, added either patiromer or placebo to their guideline-directed medical therapy (GDMT), which includes, even emphasizes, the culprit medication. They include ACE inhibitors, angiotensin-receptor blockers (ARBs), angiotensin-receptor/neprilysin inhibitors (ARNIs), and mineralocorticoid receptor antagonists (MRAs).
Those taking patiromer tolerated more intense RASI therapy – including MRAs, which are especially prone to causing hyperkalemia – than the patients assigned to placebo. They also maintained lower potassium concentrations and experienced fewer clinically important hyperkalemia episodes, reported Javed Butler, MD, MPH, MBA, Baylor Scott and White Research Institute, Dallas, at the annual scientific sessions of the American College of Cardiology.
The apparent benefit from patiromer came in part from an advantage for a composite hyperkalemia-event endpoint that included mortality, Dr. Butler noted. That advantage seemed to hold regardless of age, sex, body mass index, HFrEF symptom severity, or initial natriuretic peptide levels.
Patients who took patiromer, compared with those who took placebo, showed a 37% reduction in risk for hyperkalemia (P = .006), defined as potassium levels exceeding 5.5 mEq/L, over a median follow-up of 27 weeks. They were 38% less likely to have their MRA dosage reduced to below target level (P = .006).
More patients in the patiromer group than in the control group attained at least 50% of target dosage for MRAs and ACE inhibitors, ARBs, or ARNIs (92% vs. 87%; P = .015).
Patients with HFrEF are unlikely to achieve best possible outcomes without GDMT optimization, but failure to optimize is often attributed to hyperkalemia concerns. DIAMOND, Dr. Butler said, suggests that, by adding the potassium sequestrant to GDMT, “you can simultaneously control potassium and optimize RASI therapy.” Many clinicians seem to believe they can achieve only one or the other.
DIAMOND was too underpowered to show whether preventing hyperkalemia with patiromer could improve clinical outcomes. But failure to optimize RASI medication in HFrEF can worsen risk for heart failure events and death. So “it stands to reason that optimization of RASI therapy without a concomitant risk of hyperkalemia may, in the long run, lead to better outcomes for these patients,” Dr. Butler said in an interview.
Given the drug’s ability to keep potassium levels in check during RASI therapy, Dr. Butler said, “hypokalemia should not be a reason for suboptimal therapy.”
Patiromer and other potassium sequestrants have been available in the United States and Europe for 4-6 years, but their value as adjuncts to RASI medication in HFrEF or other heart failure has been unclear.
“There’s a good opportunity to expand the use of the drug. The question is, in whom and when?” James L. Januzzi, MD, Massachusetts General Hospital, Boston, said in an interview.
Some HFrEF patients on GDMT “should be treated with patiromer. The bigger question is, should we give someone who has a history of hyperkalemia another chance at GDMT before we treat them with patiromer? Because they may not necessarily develop hyperkalemia a second time,” said Dr. Januzzi, who was on the DIAMOND endpoint-adjudication committee.
Among the most notable findings of the trial, he said, is that the number of people who developed hyperkalemia on RASI medication, although significantly elevated, “wasn’t as high as they expected it would be,” he said. “The data from DIAMOND argue that if a really significant majority does not become hyperkalemic on rechallenge, jumping straight to a potassium-binding drug may be premature.”
Physicians across specialties can differ in how they interpret potassium-level elevation and can use various cut points to flag when to stop RASI medication or at least hold back on up-titration, Dr. Butler observed. “Cardiologists have a different threshold of potassium that they tolerate than say, for instance, a nephrologist.”
Useful, then, might be a way to tell which patients are most likely to develop hyperkalemia with RASI up-titration and so might benefit from a potassium-binding agent right away. But DIAMOND, Dr. Butler said, “does not necessarily define any patient phenotype or any potassium level where we would say that you should use a potassium binder.”
The trial entered 1,642 patients with HFrEF and current or past RASI-related hyperkalemia to a 12-week run-in phase for optimization of GDMT with patiromer. The trial was conducted at nearly 400 centers in 21 countries.
RASI medication could be optimized in 85% of the cohort, from which 878 patients were randomly assigned either to continue optimized GDMT with patiromer or to have the potassium-sequestrant replaced with a placebo.
The patients on patiromer showed a 0.03-mEq/L mean rise in serum potassium levels from randomization to the end of the study, the primary endpoint, compared with a 0.13 mEq/L mean increase for those in the control group (P < .001), Dr. Butler reported.
The win ratio for a RASI-use score hierarchically featuring cardiovascular death and CV hospitalization for hyperkalemia at several levels of severity was 1.25 (95% confidence interval, 1.003-1.564; P = .048), favoring the patiromer group. The win ratio solely for hyperkalemia-related events also favored patients on patiromer, at 1.53 (95% CI, 1.23-1.91; P < .001).
Patiromer also seemed well tolerated, Dr. Butler said.
Hyperkalemia is “one of the most common excuses” from clinicians for failing to up-titrate RASI medicine in patients with heart failure, Dr. Januzzi said. DIAMOND was less about patiromer itself than about ways “to facilitate better GDMT, where we’re really falling short of the mark. During the run-in phase they were able to get the vast majority of individuals to target, which to me is a critically important point, and emblematic of the need for things that facilitate this kind of excellent care.”
DIAMOND was funded by Vifor Pharma. Dr. Butler disclosed receiving consulting fees from Abbott, Adrenomed, Amgen, Applied Therapeutics, Array, AstraZeneca, Bayer, Boehringer Ingelheim, CVRx, G3 Pharma, Impulse Dynamics, Innolife, Janssen, LivaNova, Luitpold, Medtronic, Merck, Novartis, Novo Nordisk, Relypsa, Sequana Medical, and Vifor Pharma. Dr. Januzzi disclosed receiving consultant fees or honoraria from Abbott Laboratories, Imbria, Jana Care, Novartis, Prevencio, and Roche Diagnostics; serving on a data safety monitoring board for AbbVie, Amgen, Bayer Healthcare Pharmaceuticals, Beyer, CVRx, and Takeda Pharmaceuticals North America; and receiving research grants from Abbott Laboratories, Janssen, and Vifor Pharma.
A version of this article first appeared on Medscape.com.
FROM ACC 2022
Steroids counter ataxia telangiectasia
SEATTLE –
The disease is an autosomal recessive disorder caused by mutations in the ATM gene, which is critical to the response to cellular insults such as DNA breaks, oxidative damage, and other forms of stress. The result is clinical manifestations that range from a suppressed immune system to organ damage and neurological symptoms that typically lead patients to be wheelchair bound by their teenage years.
“It’s really multisystem and a very, very difficult disease for people to live with,” Howard M. Lederman, MD, PhD, said in an interview. Dr. Lederman is a coauthor of the study, which was presented by Stefan Zielen, PhD, professor at the University of Goethe, at the 2022 annual meeting of the American Academy of Neurology.
Various therapies have been developed to improve immunodeficiency, lung disease, and some of the other clinical aspects of the condition, but there is no treatment for its neurological effects. “There’s not really been a good animal model, which has been a big problem in trying to test drugs and design treatment trials,” said Dr. Lederman, professor of pediatrics and medicine at Johns Hopkins University, Baltimore.
The new results may change that. “In the children under the age of 9, there was really a very clear slowdown in the neurodegeneration, and specifically the time that it took for them to lose the ability to ambulate. It’s very exciting, because it’s the first time that anybody has really shown in a double-blind, placebo-controlled, large phase 3 study that any drug has been able to do this. And there were really no steroid side effects, which is the other really remarkable thing about this study,” said Dr. Lederman.
The therapy grew out of a study by researchers in Italy who treated pediatric ataxia telangiectasia patients with corticosteroids and found some transitory improvements in gross motor function, but concerns about long-term exposure to steroids limited its application. EryDel, which specializes in encapsulating therapeutics in red blood cells, became interested and developed a formulation using the patient’s own red blood cells infused with DSP. Reinfused to the patients, the red blood cells slowly release the steroid.
It isn’t clear how dexamethasone works. There are data suggesting that it might lead to transcription of small pieces of the ATM protein, “but that has really not been nailed down in any way at this point. Corticosteroids act on all kinds of cells in all kinds of ways, and so there might be a little bit of this so-called mini-ATM that’s produced, but that may or may not be related to the way in which corticosteroids have a beneficial effect on the rate of neurodegeneration,” said Dr. Lederman.
The treatment process is not easy. Children must have 50-60 cc of blood removed. Red blood cells treated to become porous are exposed to DSP, and then resealed. Then the cells are reinfused. “The whole process takes from beginning to end probably about 3 hours, with a really experienced team of people doing it. And it’s limiting because it’s not easy to put in an IV and take 50 or 60 cc of blood out of children much younger than 5 or 6. The process is now being modified to see whether we could do it with 20 to 30 cc instead,” said Dr. Lederman.
A ‘promising and impressive’ study
The study is promising, according to Nicholas Johnson, MD, who comoderated the session where the study was presented. “They were able to show a slower rate of neurological degeneration or duration on both the lower and higher dose compared with the placebo. This is promising and impressive, in the sense that it’s a really large (trial) for a rare condition,” Dr. Johnson, vice chair of research at Virginia Commonwealth University, Richmond, said in an interview.
The study included 164 patients Europe, Australia, Israel, Tunisia, India, and the United States, who received 5-10 mg dexamethasone, 14-22 mg DSP, or placebo. Mean ages in each group ranged from 9.6 to 10.4 years.
In an intention-to-treat analysis, modified International Cooperative Ataxia Rating Scale (mICARS) scores trended toward improvement in the low-dose (–1.37; P = .0847) and high-dose groups (–1.40; P = .0765) when determined by central raters during the COVID-19 pandemic. There was also a trend toward improvement when determined by local raters in the low dose group (–1.73; P = .0720) and a statistically significant change in the high dose group (–2.11; P = .0277). The researchers noted some inconsistency between local and central raters, due to inconsistency of videography and language challenges for central raters.
An intention-to-treat analysis of a subgroup of 89 patients age 6-9, who were compared with natural history data from 245 patients, found a deterioration of mICARS of 3.7 per year, compared with 0.92 in the high-dose group, for a reduction of 75% (P = .020). In the high-dose group, 51.7% had a minimal or significant improvement compared with baseline according to the Clinical Global Impression of Change, as did 29.0% on low dose, and 27.6% in the placebo group.
SEATTLE –
The disease is an autosomal recessive disorder caused by mutations in the ATM gene, which is critical to the response to cellular insults such as DNA breaks, oxidative damage, and other forms of stress. The result is clinical manifestations that range from a suppressed immune system to organ damage and neurological symptoms that typically lead patients to be wheelchair bound by their teenage years.
“It’s really multisystem and a very, very difficult disease for people to live with,” Howard M. Lederman, MD, PhD, said in an interview. Dr. Lederman is a coauthor of the study, which was presented by Stefan Zielen, PhD, professor at the University of Goethe, at the 2022 annual meeting of the American Academy of Neurology.
Various therapies have been developed to improve immunodeficiency, lung disease, and some of the other clinical aspects of the condition, but there is no treatment for its neurological effects. “There’s not really been a good animal model, which has been a big problem in trying to test drugs and design treatment trials,” said Dr. Lederman, professor of pediatrics and medicine at Johns Hopkins University, Baltimore.
The new results may change that. “In the children under the age of 9, there was really a very clear slowdown in the neurodegeneration, and specifically the time that it took for them to lose the ability to ambulate. It’s very exciting, because it’s the first time that anybody has really shown in a double-blind, placebo-controlled, large phase 3 study that any drug has been able to do this. And there were really no steroid side effects, which is the other really remarkable thing about this study,” said Dr. Lederman.
The therapy grew out of a study by researchers in Italy who treated pediatric ataxia telangiectasia patients with corticosteroids and found some transitory improvements in gross motor function, but concerns about long-term exposure to steroids limited its application. EryDel, which specializes in encapsulating therapeutics in red blood cells, became interested and developed a formulation using the patient’s own red blood cells infused with DSP. Reinfused to the patients, the red blood cells slowly release the steroid.
It isn’t clear how dexamethasone works. There are data suggesting that it might lead to transcription of small pieces of the ATM protein, “but that has really not been nailed down in any way at this point. Corticosteroids act on all kinds of cells in all kinds of ways, and so there might be a little bit of this so-called mini-ATM that’s produced, but that may or may not be related to the way in which corticosteroids have a beneficial effect on the rate of neurodegeneration,” said Dr. Lederman.
The treatment process is not easy. Children must have 50-60 cc of blood removed. Red blood cells treated to become porous are exposed to DSP, and then resealed. Then the cells are reinfused. “The whole process takes from beginning to end probably about 3 hours, with a really experienced team of people doing it. And it’s limiting because it’s not easy to put in an IV and take 50 or 60 cc of blood out of children much younger than 5 or 6. The process is now being modified to see whether we could do it with 20 to 30 cc instead,” said Dr. Lederman.
A ‘promising and impressive’ study
The study is promising, according to Nicholas Johnson, MD, who comoderated the session where the study was presented. “They were able to show a slower rate of neurological degeneration or duration on both the lower and higher dose compared with the placebo. This is promising and impressive, in the sense that it’s a really large (trial) for a rare condition,” Dr. Johnson, vice chair of research at Virginia Commonwealth University, Richmond, said in an interview.
The study included 164 patients Europe, Australia, Israel, Tunisia, India, and the United States, who received 5-10 mg dexamethasone, 14-22 mg DSP, or placebo. Mean ages in each group ranged from 9.6 to 10.4 years.
In an intention-to-treat analysis, modified International Cooperative Ataxia Rating Scale (mICARS) scores trended toward improvement in the low-dose (–1.37; P = .0847) and high-dose groups (–1.40; P = .0765) when determined by central raters during the COVID-19 pandemic. There was also a trend toward improvement when determined by local raters in the low dose group (–1.73; P = .0720) and a statistically significant change in the high dose group (–2.11; P = .0277). The researchers noted some inconsistency between local and central raters, due to inconsistency of videography and language challenges for central raters.
An intention-to-treat analysis of a subgroup of 89 patients age 6-9, who were compared with natural history data from 245 patients, found a deterioration of mICARS of 3.7 per year, compared with 0.92 in the high-dose group, for a reduction of 75% (P = .020). In the high-dose group, 51.7% had a minimal or significant improvement compared with baseline according to the Clinical Global Impression of Change, as did 29.0% on low dose, and 27.6% in the placebo group.
SEATTLE –
The disease is an autosomal recessive disorder caused by mutations in the ATM gene, which is critical to the response to cellular insults such as DNA breaks, oxidative damage, and other forms of stress. The result is clinical manifestations that range from a suppressed immune system to organ damage and neurological symptoms that typically lead patients to be wheelchair bound by their teenage years.
“It’s really multisystem and a very, very difficult disease for people to live with,” Howard M. Lederman, MD, PhD, said in an interview. Dr. Lederman is a coauthor of the study, which was presented by Stefan Zielen, PhD, professor at the University of Goethe, at the 2022 annual meeting of the American Academy of Neurology.
Various therapies have been developed to improve immunodeficiency, lung disease, and some of the other clinical aspects of the condition, but there is no treatment for its neurological effects. “There’s not really been a good animal model, which has been a big problem in trying to test drugs and design treatment trials,” said Dr. Lederman, professor of pediatrics and medicine at Johns Hopkins University, Baltimore.
The new results may change that. “In the children under the age of 9, there was really a very clear slowdown in the neurodegeneration, and specifically the time that it took for them to lose the ability to ambulate. It’s very exciting, because it’s the first time that anybody has really shown in a double-blind, placebo-controlled, large phase 3 study that any drug has been able to do this. And there were really no steroid side effects, which is the other really remarkable thing about this study,” said Dr. Lederman.
The therapy grew out of a study by researchers in Italy who treated pediatric ataxia telangiectasia patients with corticosteroids and found some transitory improvements in gross motor function, but concerns about long-term exposure to steroids limited its application. EryDel, which specializes in encapsulating therapeutics in red blood cells, became interested and developed a formulation using the patient’s own red blood cells infused with DSP. Reinfused to the patients, the red blood cells slowly release the steroid.
It isn’t clear how dexamethasone works. There are data suggesting that it might lead to transcription of small pieces of the ATM protein, “but that has really not been nailed down in any way at this point. Corticosteroids act on all kinds of cells in all kinds of ways, and so there might be a little bit of this so-called mini-ATM that’s produced, but that may or may not be related to the way in which corticosteroids have a beneficial effect on the rate of neurodegeneration,” said Dr. Lederman.
The treatment process is not easy. Children must have 50-60 cc of blood removed. Red blood cells treated to become porous are exposed to DSP, and then resealed. Then the cells are reinfused. “The whole process takes from beginning to end probably about 3 hours, with a really experienced team of people doing it. And it’s limiting because it’s not easy to put in an IV and take 50 or 60 cc of blood out of children much younger than 5 or 6. The process is now being modified to see whether we could do it with 20 to 30 cc instead,” said Dr. Lederman.
A ‘promising and impressive’ study
The study is promising, according to Nicholas Johnson, MD, who comoderated the session where the study was presented. “They were able to show a slower rate of neurological degeneration or duration on both the lower and higher dose compared with the placebo. This is promising and impressive, in the sense that it’s a really large (trial) for a rare condition,” Dr. Johnson, vice chair of research at Virginia Commonwealth University, Richmond, said in an interview.
The study included 164 patients Europe, Australia, Israel, Tunisia, India, and the United States, who received 5-10 mg dexamethasone, 14-22 mg DSP, or placebo. Mean ages in each group ranged from 9.6 to 10.4 years.
In an intention-to-treat analysis, modified International Cooperative Ataxia Rating Scale (mICARS) scores trended toward improvement in the low-dose (–1.37; P = .0847) and high-dose groups (–1.40; P = .0765) when determined by central raters during the COVID-19 pandemic. There was also a trend toward improvement when determined by local raters in the low dose group (–1.73; P = .0720) and a statistically significant change in the high dose group (–2.11; P = .0277). The researchers noted some inconsistency between local and central raters, due to inconsistency of videography and language challenges for central raters.
An intention-to-treat analysis of a subgroup of 89 patients age 6-9, who were compared with natural history data from 245 patients, found a deterioration of mICARS of 3.7 per year, compared with 0.92 in the high-dose group, for a reduction of 75% (P = .020). In the high-dose group, 51.7% had a minimal or significant improvement compared with baseline according to the Clinical Global Impression of Change, as did 29.0% on low dose, and 27.6% in the placebo group.
AT AAN 2022