User login
Study backs broader use of aspirin to prevent preeclampsia
Prescribing low-dose aspirin to all women at high risk for preeclampsia and women with two or more moderate risk factors is the most cost-beneficial approach to lowering the risk of preeclampsia, according to a new study.
The study, which modeled four approaches to low-dose aspirin prophylaxis in pregnant women, shows that the approach recommended by the U.S. Preventive Services Task Force is superior to the one currently recommended by the American College of Obstetricians and Gynecologists when it comes to lowering the risk of preeclampsia and reducing costs (Obstet Gynecol 2015. doi: 10.1097/AOG.0000000000001115).
Dr. Erika F. Werner and her colleagues at Brown University, Providence, R.I., used a hypothetical cohort of four million U.S. women to analyze the effectiveness of four distinct distribution models for low-dose aspirin prophylaxis: no prophylaxis; following the ACOG recommendations; following the USPSTF recommendations; and universal prophylaxis.
Prior randomized studies have established that low-dose aspirin prophylaxis lowers the risk of preeclampsia and preterm birth, the authors wrote. “The remaining question over low-dose aspirin use in pregnancy is not a scientific one, but rather a question of health policy: Who should be treated and what are the expected risks, benefits, and costs of one policy, compared with another?”
ACOG recommends that low-dose aspirin be given to pregnant women with a history of preeclampsia necessitating delivery before 34 weeks of gestation and to women with preeclampsia in more than one prior pregnancy. The USPSTF, on the other hand, takes a broader approach, recommending that women with a history of preeclampsia and those with multiple gestation, chronic hypertension, diabetes mellitus, renal disease, or autoimmune disease should receive low-dose aspirin. Women with two or more moderate risk factors such as nulliparity, obesity, African American race, age 35 years or older, a family history of preeclampsia, or a personal history of pregnancy complications should also receive aspirin, according to the USPSTF.
Under the decision model created by the researchers, women were assumed to be 77% compliant with adhering to their aspirin regimen of 81 mg daily.
Without any aspirin prophylaxis, researchers estimated that the rate of preeclampsia in this population would be 4.18%, meaning 167,200 women would develop preeclampsia. If ACOG’s approach were followed, the rate would dip to 4.17% (166,720 women). But following the USPSTF’s recommendations would result in a 3.83% rate of preeclampsia (153,160), while universal administration of low-dose aspirin prophylaxis would result in a rate of 3.81% (152,240).
The more widespread use of aspirin would also reduce preterm births and maternal deaths, but increase significant maternal gastrointestinal bleeding events, placental abruption, and aspirin-exacerbated respiratory disease, according to the study.
In the baseline analysis, the USPSTF approach was the most cost beneficial, with a direct annual medical cost savings of more than $364 million when compared with the ACOG approach.
“Our analysis suggests that the U.S. Preventive Services Task Force approach is the most cost beneficial, achieving 94% of the possible preeclampsia rate reduction that can be obtained with aspirin while exposing only one-fourth of pregnant women to the theoretical risks of aspirin,” the researchers wrote.
When compared with the USPSTF approach, universal administration would require more than 3 million women to take aspirin to prevent 920 cases of preeclampsia, an incremental number needed to treat of 3,325. Even after taking this into account, universal coverage would still be “highly cost-effective” and would maximize reductions in preterm birth and preeclampsia, according to the researchers.
“From the standpoint of parsimony, and minimization of potential aspirin risks, the U.S. Preventive Services Task Force approach is a better approach than universal administration,” the researchers wrote. “However, universal administration, with its ease of implementation and potential to maximize the health benefits of aspirin, may in fact be the more rational and clinically pragmatic approach.”
The researchers reported having no financial disclosures.
Prescribing low-dose aspirin to all women at high risk for preeclampsia and women with two or more moderate risk factors is the most cost-beneficial approach to lowering the risk of preeclampsia, according to a new study.
The study, which modeled four approaches to low-dose aspirin prophylaxis in pregnant women, shows that the approach recommended by the U.S. Preventive Services Task Force is superior to the one currently recommended by the American College of Obstetricians and Gynecologists when it comes to lowering the risk of preeclampsia and reducing costs (Obstet Gynecol 2015. doi: 10.1097/AOG.0000000000001115).
Dr. Erika F. Werner and her colleagues at Brown University, Providence, R.I., used a hypothetical cohort of four million U.S. women to analyze the effectiveness of four distinct distribution models for low-dose aspirin prophylaxis: no prophylaxis; following the ACOG recommendations; following the USPSTF recommendations; and universal prophylaxis.
Prior randomized studies have established that low-dose aspirin prophylaxis lowers the risk of preeclampsia and preterm birth, the authors wrote. “The remaining question over low-dose aspirin use in pregnancy is not a scientific one, but rather a question of health policy: Who should be treated and what are the expected risks, benefits, and costs of one policy, compared with another?”
ACOG recommends that low-dose aspirin be given to pregnant women with a history of preeclampsia necessitating delivery before 34 weeks of gestation and to women with preeclampsia in more than one prior pregnancy. The USPSTF, on the other hand, takes a broader approach, recommending that women with a history of preeclampsia and those with multiple gestation, chronic hypertension, diabetes mellitus, renal disease, or autoimmune disease should receive low-dose aspirin. Women with two or more moderate risk factors such as nulliparity, obesity, African American race, age 35 years or older, a family history of preeclampsia, or a personal history of pregnancy complications should also receive aspirin, according to the USPSTF.
Under the decision model created by the researchers, women were assumed to be 77% compliant with adhering to their aspirin regimen of 81 mg daily.
Without any aspirin prophylaxis, researchers estimated that the rate of preeclampsia in this population would be 4.18%, meaning 167,200 women would develop preeclampsia. If ACOG’s approach were followed, the rate would dip to 4.17% (166,720 women). But following the USPSTF’s recommendations would result in a 3.83% rate of preeclampsia (153,160), while universal administration of low-dose aspirin prophylaxis would result in a rate of 3.81% (152,240).
The more widespread use of aspirin would also reduce preterm births and maternal deaths, but increase significant maternal gastrointestinal bleeding events, placental abruption, and aspirin-exacerbated respiratory disease, according to the study.
In the baseline analysis, the USPSTF approach was the most cost beneficial, with a direct annual medical cost savings of more than $364 million when compared with the ACOG approach.
“Our analysis suggests that the U.S. Preventive Services Task Force approach is the most cost beneficial, achieving 94% of the possible preeclampsia rate reduction that can be obtained with aspirin while exposing only one-fourth of pregnant women to the theoretical risks of aspirin,” the researchers wrote.
When compared with the USPSTF approach, universal administration would require more than 3 million women to take aspirin to prevent 920 cases of preeclampsia, an incremental number needed to treat of 3,325. Even after taking this into account, universal coverage would still be “highly cost-effective” and would maximize reductions in preterm birth and preeclampsia, according to the researchers.
“From the standpoint of parsimony, and minimization of potential aspirin risks, the U.S. Preventive Services Task Force approach is a better approach than universal administration,” the researchers wrote. “However, universal administration, with its ease of implementation and potential to maximize the health benefits of aspirin, may in fact be the more rational and clinically pragmatic approach.”
The researchers reported having no financial disclosures.
Prescribing low-dose aspirin to all women at high risk for preeclampsia and women with two or more moderate risk factors is the most cost-beneficial approach to lowering the risk of preeclampsia, according to a new study.
The study, which modeled four approaches to low-dose aspirin prophylaxis in pregnant women, shows that the approach recommended by the U.S. Preventive Services Task Force is superior to the one currently recommended by the American College of Obstetricians and Gynecologists when it comes to lowering the risk of preeclampsia and reducing costs (Obstet Gynecol 2015. doi: 10.1097/AOG.0000000000001115).
Dr. Erika F. Werner and her colleagues at Brown University, Providence, R.I., used a hypothetical cohort of four million U.S. women to analyze the effectiveness of four distinct distribution models for low-dose aspirin prophylaxis: no prophylaxis; following the ACOG recommendations; following the USPSTF recommendations; and universal prophylaxis.
Prior randomized studies have established that low-dose aspirin prophylaxis lowers the risk of preeclampsia and preterm birth, the authors wrote. “The remaining question over low-dose aspirin use in pregnancy is not a scientific one, but rather a question of health policy: Who should be treated and what are the expected risks, benefits, and costs of one policy, compared with another?”
ACOG recommends that low-dose aspirin be given to pregnant women with a history of preeclampsia necessitating delivery before 34 weeks of gestation and to women with preeclampsia in more than one prior pregnancy. The USPSTF, on the other hand, takes a broader approach, recommending that women with a history of preeclampsia and those with multiple gestation, chronic hypertension, diabetes mellitus, renal disease, or autoimmune disease should receive low-dose aspirin. Women with two or more moderate risk factors such as nulliparity, obesity, African American race, age 35 years or older, a family history of preeclampsia, or a personal history of pregnancy complications should also receive aspirin, according to the USPSTF.
Under the decision model created by the researchers, women were assumed to be 77% compliant with adhering to their aspirin regimen of 81 mg daily.
Without any aspirin prophylaxis, researchers estimated that the rate of preeclampsia in this population would be 4.18%, meaning 167,200 women would develop preeclampsia. If ACOG’s approach were followed, the rate would dip to 4.17% (166,720 women). But following the USPSTF’s recommendations would result in a 3.83% rate of preeclampsia (153,160), while universal administration of low-dose aspirin prophylaxis would result in a rate of 3.81% (152,240).
The more widespread use of aspirin would also reduce preterm births and maternal deaths, but increase significant maternal gastrointestinal bleeding events, placental abruption, and aspirin-exacerbated respiratory disease, according to the study.
In the baseline analysis, the USPSTF approach was the most cost beneficial, with a direct annual medical cost savings of more than $364 million when compared with the ACOG approach.
“Our analysis suggests that the U.S. Preventive Services Task Force approach is the most cost beneficial, achieving 94% of the possible preeclampsia rate reduction that can be obtained with aspirin while exposing only one-fourth of pregnant women to the theoretical risks of aspirin,” the researchers wrote.
When compared with the USPSTF approach, universal administration would require more than 3 million women to take aspirin to prevent 920 cases of preeclampsia, an incremental number needed to treat of 3,325. Even after taking this into account, universal coverage would still be “highly cost-effective” and would maximize reductions in preterm birth and preeclampsia, according to the researchers.
“From the standpoint of parsimony, and minimization of potential aspirin risks, the U.S. Preventive Services Task Force approach is a better approach than universal administration,” the researchers wrote. “However, universal administration, with its ease of implementation and potential to maximize the health benefits of aspirin, may in fact be the more rational and clinically pragmatic approach.”
The researchers reported having no financial disclosures.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point: Following the U.S. Preventive Services Task Force’s recommendations for the distribution of low-dose aspirin prophylaxis is the most cost-beneficial solution to significantly reduce morbidity and mortality associated with preeclampsia.
Major finding: The USPSTF approach would result in a preeclampsia rate of 3.83%, compared with a rate of 4.17% if the ACOG recommendations were followed.
Data source: Prospective cohort study of four million women and four decision models of distributing low-dose aspirin to prevent preeclampsia.
Disclosures: The researchers reported having no financial disclosures.
Cesarean rate is not doubled in nulliparous inductions
Nulliparous pregnant women with an unfavorable cervix who were induced after 39 weeks’ gestation were more likely to have a cesarean delivery than were women undergoing expectant management, but the ceserean rate was not doubled, according to a new study.
The randomized clinical trial of 162 pregnant women was powered only to detect a twofold difference in cesearean rates since that rate has been cited in older observational studies and some guidelines as evidence against elective induction, according to Dr. Nathaniel R. Miller of the Carl R. Darnall Army Medical Center in Fort Hood, Tex., and his associates.
“Clearly, less-modest increases in the cesarean delivery rate may be clinically important, and future studies should be powered to detect smaller differences in the cesarean delivery rate between induction and expectant management,” the researchers wrote.
Researchers randomized 162 nulliparous pregnant women to elective induction of labor or expectant management when they were between 38 weeks 0 days and 38 weeks 6 days of gestation. The women were at least 18 years old with a singleton pregnancy with cephalic presentation, and a Bishop score no higher than 5. Inductions occurred within a week of enrollment, which ran from March 2010 to February 2014, but no women were induced before 39 weeks 0 days (Obstet Gynecol. 2015;126:1258-64).
Cesarean delivery was indicated if at least one of the following was present: nonreassuring fetal status (persistent category II or III); active phase arrest of dilation; arrest of descent in the second stage of labor; or failed induction of labor.
Among the 82 women assigned to induction, 3 did not receive the intervention, 2% were admitted in spontaneous labor, and 30.5% had a cesarean delivery. Among 80 women randomized to expectant management, after excluding 1 lost to follow-up, 44% were admitted in spontaneous labor and 17.7% had a cesarean delivery.
The risk of cesarean delivery between the groups, however, was not statistically significant (relative risk, 1.72; 95% confidence interval 0.96-3.06). Neither postpartum length of stay nor indications for cesarean delivery significantly differed between the two groups, though maternal total hospital length of stay was 10 hours longer in the induction group and inductions for hypertensive disorders were greater in the expectant management group.
“The consistent message from the expanding literature on this topic is that there are and will continue to be important trade-offs to weigh in the balance, especially when it comes to discussing the risk of cesarean delivery with induction in this seemingly highest-risk population of nulliparous women with an unfavorable cervix,” the resarchers wrote.
Additionally, the study defined active phase arrest of dilation as “2 hours of no cervical change once 4 cm or more dilation had been reached after rupture of membranes,” rather than the 4 hours recommended by guidelines for prevention of the first cesarean delivery, published in November 2012 by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the Society for Maternal Fetal Medicine, and the American College of Obstetricians and Gynecologists. Since the most common indication for cesarean delivery in the induction group was arrest of dilation, use of the 4-hour criteria “would have resulted in a reduction in the cesarean delivery rate for the induction of labor arm to 20% and the expectant management arm to 15%,” the researchers noted.
The researchers reported having no financial disclosures.
Nulliparous pregnant women with an unfavorable cervix who were induced after 39 weeks’ gestation were more likely to have a cesarean delivery than were women undergoing expectant management, but the ceserean rate was not doubled, according to a new study.
The randomized clinical trial of 162 pregnant women was powered only to detect a twofold difference in cesearean rates since that rate has been cited in older observational studies and some guidelines as evidence against elective induction, according to Dr. Nathaniel R. Miller of the Carl R. Darnall Army Medical Center in Fort Hood, Tex., and his associates.
“Clearly, less-modest increases in the cesarean delivery rate may be clinically important, and future studies should be powered to detect smaller differences in the cesarean delivery rate between induction and expectant management,” the researchers wrote.
Researchers randomized 162 nulliparous pregnant women to elective induction of labor or expectant management when they were between 38 weeks 0 days and 38 weeks 6 days of gestation. The women were at least 18 years old with a singleton pregnancy with cephalic presentation, and a Bishop score no higher than 5. Inductions occurred within a week of enrollment, which ran from March 2010 to February 2014, but no women were induced before 39 weeks 0 days (Obstet Gynecol. 2015;126:1258-64).
Cesarean delivery was indicated if at least one of the following was present: nonreassuring fetal status (persistent category II or III); active phase arrest of dilation; arrest of descent in the second stage of labor; or failed induction of labor.
Among the 82 women assigned to induction, 3 did not receive the intervention, 2% were admitted in spontaneous labor, and 30.5% had a cesarean delivery. Among 80 women randomized to expectant management, after excluding 1 lost to follow-up, 44% were admitted in spontaneous labor and 17.7% had a cesarean delivery.
The risk of cesarean delivery between the groups, however, was not statistically significant (relative risk, 1.72; 95% confidence interval 0.96-3.06). Neither postpartum length of stay nor indications for cesarean delivery significantly differed between the two groups, though maternal total hospital length of stay was 10 hours longer in the induction group and inductions for hypertensive disorders were greater in the expectant management group.
“The consistent message from the expanding literature on this topic is that there are and will continue to be important trade-offs to weigh in the balance, especially when it comes to discussing the risk of cesarean delivery with induction in this seemingly highest-risk population of nulliparous women with an unfavorable cervix,” the resarchers wrote.
Additionally, the study defined active phase arrest of dilation as “2 hours of no cervical change once 4 cm or more dilation had been reached after rupture of membranes,” rather than the 4 hours recommended by guidelines for prevention of the first cesarean delivery, published in November 2012 by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the Society for Maternal Fetal Medicine, and the American College of Obstetricians and Gynecologists. Since the most common indication for cesarean delivery in the induction group was arrest of dilation, use of the 4-hour criteria “would have resulted in a reduction in the cesarean delivery rate for the induction of labor arm to 20% and the expectant management arm to 15%,” the researchers noted.
The researchers reported having no financial disclosures.
Nulliparous pregnant women with an unfavorable cervix who were induced after 39 weeks’ gestation were more likely to have a cesarean delivery than were women undergoing expectant management, but the ceserean rate was not doubled, according to a new study.
The randomized clinical trial of 162 pregnant women was powered only to detect a twofold difference in cesearean rates since that rate has been cited in older observational studies and some guidelines as evidence against elective induction, according to Dr. Nathaniel R. Miller of the Carl R. Darnall Army Medical Center in Fort Hood, Tex., and his associates.
“Clearly, less-modest increases in the cesarean delivery rate may be clinically important, and future studies should be powered to detect smaller differences in the cesarean delivery rate between induction and expectant management,” the researchers wrote.
Researchers randomized 162 nulliparous pregnant women to elective induction of labor or expectant management when they were between 38 weeks 0 days and 38 weeks 6 days of gestation. The women were at least 18 years old with a singleton pregnancy with cephalic presentation, and a Bishop score no higher than 5. Inductions occurred within a week of enrollment, which ran from March 2010 to February 2014, but no women were induced before 39 weeks 0 days (Obstet Gynecol. 2015;126:1258-64).
Cesarean delivery was indicated if at least one of the following was present: nonreassuring fetal status (persistent category II or III); active phase arrest of dilation; arrest of descent in the second stage of labor; or failed induction of labor.
Among the 82 women assigned to induction, 3 did not receive the intervention, 2% were admitted in spontaneous labor, and 30.5% had a cesarean delivery. Among 80 women randomized to expectant management, after excluding 1 lost to follow-up, 44% were admitted in spontaneous labor and 17.7% had a cesarean delivery.
The risk of cesarean delivery between the groups, however, was not statistically significant (relative risk, 1.72; 95% confidence interval 0.96-3.06). Neither postpartum length of stay nor indications for cesarean delivery significantly differed between the two groups, though maternal total hospital length of stay was 10 hours longer in the induction group and inductions for hypertensive disorders were greater in the expectant management group.
“The consistent message from the expanding literature on this topic is that there are and will continue to be important trade-offs to weigh in the balance, especially when it comes to discussing the risk of cesarean delivery with induction in this seemingly highest-risk population of nulliparous women with an unfavorable cervix,” the resarchers wrote.
Additionally, the study defined active phase arrest of dilation as “2 hours of no cervical change once 4 cm or more dilation had been reached after rupture of membranes,” rather than the 4 hours recommended by guidelines for prevention of the first cesarean delivery, published in November 2012 by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the Society for Maternal Fetal Medicine, and the American College of Obstetricians and Gynecologists. Since the most common indication for cesarean delivery in the induction group was arrest of dilation, use of the 4-hour criteria “would have resulted in a reduction in the cesarean delivery rate for the induction of labor arm to 20% and the expectant management arm to 15%,” the researchers noted.
The researchers reported having no financial disclosures.
FROM OBSTETRICS AND GYNECOLOGY
Key clinical point: Cesarean delivery rates were not doubled with elective induction after 39 weeks, compared with expectant management in nulliparous women with an unfavorable cervix.
Major finding: A total of 30.5% of induced women had a cesarean delivery, compared with 17.7% of women undergoing expectant management for a relative risk 1.72 (95% CI, 0.96-3.06).
Data source: A randomized controlled trial of 162 nulliparous pregnant women enrolled between March 2010 and February 2014.
Disclosures: The authors reported having no financial disclosures.
Stillbirth rates stable despite decline in induced births
Stillbirth rates in the United States plateaued between 2006 and 2012 at 6.05 stillbirths per 1,000 deliveries, reported Marian F. MacDorman, Ph.D., of the Maryland Population Research Center, University of Maryland, College Park, and colleagues.
The study was published online Nov. 9 in Obstetrics and Gynecology.
National Center for Health Statistics data (totaling 50,045 stillbirths and 8,268,441 live births) were employed to evaluate rates of fetal death and live birth in the 2006 and 2012 delivery cohorts. Age-specific stillbirth rates were calculated at 20 weeks’ of gestation or greater. Changes in rates of stillbirths and live births along with changes in percentage distribution among cohorts (segmented by gestational period duration) were assessed. Although only minimal changes were evident in the percent distribution of stillbirths by gestational age from 2006 to 2012, the percent distribution of live births by gestational age demonstrated considerable variation: births at 34-36 weeks’ gestation decreased by 12% from 2006 to 2012, by 10% at 37 weeks, and by 16% at 38 weeks. Conversely, births at 39 weeks’ gestation increased by 17% (Obstet Gynecol. 2015;126:1146-50).
As the aforementioned data reflect, there is broad support among clinicians for reducing nonmedically indicated deliveries before 39 weeks’ of gestation, but critics have suggested that longer pregnancies might lead to increased stillbirth incidence. However, “the lack of change in the prospective stillbirth rate from 2006 to 2012 suggests that preventing nonmedically indicated deliveries before 39 weeks’ of gestation did not increase the stillbirth rate at the national level,” Dr. MacDorman and associates said.
The research team characterized the lack of improvement in U.S. stillbirth outcomes from 2006 to 2012 as “disappointing.” To effectively decrease the stillbirth rate in a statistically significant manner, investigators suggested more concentrated research focused on better targeting of women in the early stages of pregnancy who are at the highest risk for stillbirth, to facilitate more careful monitoring and necessary interventions.
Dr. MacDorman and colleagues did not report any conflicts of interest.
Stillbirth rates in the United States plateaued between 2006 and 2012 at 6.05 stillbirths per 1,000 deliveries, reported Marian F. MacDorman, Ph.D., of the Maryland Population Research Center, University of Maryland, College Park, and colleagues.
The study was published online Nov. 9 in Obstetrics and Gynecology.
National Center for Health Statistics data (totaling 50,045 stillbirths and 8,268,441 live births) were employed to evaluate rates of fetal death and live birth in the 2006 and 2012 delivery cohorts. Age-specific stillbirth rates were calculated at 20 weeks’ of gestation or greater. Changes in rates of stillbirths and live births along with changes in percentage distribution among cohorts (segmented by gestational period duration) were assessed. Although only minimal changes were evident in the percent distribution of stillbirths by gestational age from 2006 to 2012, the percent distribution of live births by gestational age demonstrated considerable variation: births at 34-36 weeks’ gestation decreased by 12% from 2006 to 2012, by 10% at 37 weeks, and by 16% at 38 weeks. Conversely, births at 39 weeks’ gestation increased by 17% (Obstet Gynecol. 2015;126:1146-50).
As the aforementioned data reflect, there is broad support among clinicians for reducing nonmedically indicated deliveries before 39 weeks’ of gestation, but critics have suggested that longer pregnancies might lead to increased stillbirth incidence. However, “the lack of change in the prospective stillbirth rate from 2006 to 2012 suggests that preventing nonmedically indicated deliveries before 39 weeks’ of gestation did not increase the stillbirth rate at the national level,” Dr. MacDorman and associates said.
The research team characterized the lack of improvement in U.S. stillbirth outcomes from 2006 to 2012 as “disappointing.” To effectively decrease the stillbirth rate in a statistically significant manner, investigators suggested more concentrated research focused on better targeting of women in the early stages of pregnancy who are at the highest risk for stillbirth, to facilitate more careful monitoring and necessary interventions.
Dr. MacDorman and colleagues did not report any conflicts of interest.
Stillbirth rates in the United States plateaued between 2006 and 2012 at 6.05 stillbirths per 1,000 deliveries, reported Marian F. MacDorman, Ph.D., of the Maryland Population Research Center, University of Maryland, College Park, and colleagues.
The study was published online Nov. 9 in Obstetrics and Gynecology.
National Center for Health Statistics data (totaling 50,045 stillbirths and 8,268,441 live births) were employed to evaluate rates of fetal death and live birth in the 2006 and 2012 delivery cohorts. Age-specific stillbirth rates were calculated at 20 weeks’ of gestation or greater. Changes in rates of stillbirths and live births along with changes in percentage distribution among cohorts (segmented by gestational period duration) were assessed. Although only minimal changes were evident in the percent distribution of stillbirths by gestational age from 2006 to 2012, the percent distribution of live births by gestational age demonstrated considerable variation: births at 34-36 weeks’ gestation decreased by 12% from 2006 to 2012, by 10% at 37 weeks, and by 16% at 38 weeks. Conversely, births at 39 weeks’ gestation increased by 17% (Obstet Gynecol. 2015;126:1146-50).
As the aforementioned data reflect, there is broad support among clinicians for reducing nonmedically indicated deliveries before 39 weeks’ of gestation, but critics have suggested that longer pregnancies might lead to increased stillbirth incidence. However, “the lack of change in the prospective stillbirth rate from 2006 to 2012 suggests that preventing nonmedically indicated deliveries before 39 weeks’ of gestation did not increase the stillbirth rate at the national level,” Dr. MacDorman and associates said.
The research team characterized the lack of improvement in U.S. stillbirth outcomes from 2006 to 2012 as “disappointing.” To effectively decrease the stillbirth rate in a statistically significant manner, investigators suggested more concentrated research focused on better targeting of women in the early stages of pregnancy who are at the highest risk for stillbirth, to facilitate more careful monitoring and necessary interventions.
Dr. MacDorman and colleagues did not report any conflicts of interest.
FROM OBSTETRICS AND GYNECOLOGY
Key clinical point: Longer gestational periods due to recent declines in cesarean delivery and induced birth have not resulted in increased fetal mortality.
Major finding: In both 2006 and 2012, the national stillbirth rate was 6.05 stillbirths per 1,000 deliveries.
Data source: A retrospective analysis of National Center for Health Statistics data encompassing a 50,045 stillbirths and 8,268,441 live births in 2006 and 2012.
Disclosures: Dr. MacDorman and colleagues did not report any conflicts of interest.
Drinking while pregnant: Not worth the risk
Recent findings by the American Academy of Pediatrics (AAP) regarding alcohol consumption during pregnancy are quite clear: Don’t drink at all while pregnant. This warning from the AAP is consistent with advice from the American College of Obstetricians and Gynecologists (ACOG) and the Centers for Disease Control and Prevention: No safe level of alcohol consumption during pregnancy has been identified, and no period during pregnancy appears to be safe for alcohol consumption.
In fact, ACOG has recommended screening for alcohol use early in pregnancy since 1977. However, the CDC reports that 1 in 10 pregnant women aged 18-44 years has consumed alcohol in the past 30 days. Some recent studies claim that a small number of drinks during pregnancy could be safe, and I’ve even seen some reports of ob.gyns. allowing alcohol use during pregnancy.
With such disparities between recommendations and patient behavior, the role of the obstetrician is more crucial than ever. Education, prevention, and intervention are essential to good health for mother and baby.
It has long been recognized that heavy drinking during pregnancy is associated with Fetal Alcohol Syndrome, but there are more subtle effects that fall under the umbrella of Fetal Alcohol Spectrum Disorders. These effects may include physical, mental, behavioral, and learning disabilities with lifelong implications and may not be detected until a child starts school.
Screening for alcohol use is of the utmost importance. Three simple steps have been proven effective in identifying women who drink at risky levels and engage them in changing behavior to reduce their risk for an alcohol-exposed pregnancy. These steps include asking about alcohol use, providing brief motivation-enhancing interventions, and follow-up for women who engage in risky drinking. See ACOG’s Fetal Alcohol Syndrome Prevention Tool Kit for more detail.
It is worth noting that a positive screen does not always mean a woman needs to be referred to treatment. Brief intervention by an ob.gyn. is effective and has been shown to reduce risky behavior by 70%. Randomized studies reported significant reductions in alcohol use and improved newborn outcomes after intervention.
Prenatal exposure to alcohol is one of the leading preventable causes of birth defects. With so many risk factors out of a mother’s control during pregnancy, why risk it?
Dr. Conry is assistant physician in chief at the Permanente Medical Group in Roseville, Calif., and associate clinical professor of ob.gyn. at the University of California, Davis. She has been a practicing ob.gyn. with the Permanente Medical Group for more than 20 years. She is a past president of the American College of Obstetricians and Gynecologists.
Recent findings by the American Academy of Pediatrics (AAP) regarding alcohol consumption during pregnancy are quite clear: Don’t drink at all while pregnant. This warning from the AAP is consistent with advice from the American College of Obstetricians and Gynecologists (ACOG) and the Centers for Disease Control and Prevention: No safe level of alcohol consumption during pregnancy has been identified, and no period during pregnancy appears to be safe for alcohol consumption.
In fact, ACOG has recommended screening for alcohol use early in pregnancy since 1977. However, the CDC reports that 1 in 10 pregnant women aged 18-44 years has consumed alcohol in the past 30 days. Some recent studies claim that a small number of drinks during pregnancy could be safe, and I’ve even seen some reports of ob.gyns. allowing alcohol use during pregnancy.
With such disparities between recommendations and patient behavior, the role of the obstetrician is more crucial than ever. Education, prevention, and intervention are essential to good health for mother and baby.
It has long been recognized that heavy drinking during pregnancy is associated with Fetal Alcohol Syndrome, but there are more subtle effects that fall under the umbrella of Fetal Alcohol Spectrum Disorders. These effects may include physical, mental, behavioral, and learning disabilities with lifelong implications and may not be detected until a child starts school.
Screening for alcohol use is of the utmost importance. Three simple steps have been proven effective in identifying women who drink at risky levels and engage them in changing behavior to reduce their risk for an alcohol-exposed pregnancy. These steps include asking about alcohol use, providing brief motivation-enhancing interventions, and follow-up for women who engage in risky drinking. See ACOG’s Fetal Alcohol Syndrome Prevention Tool Kit for more detail.
It is worth noting that a positive screen does not always mean a woman needs to be referred to treatment. Brief intervention by an ob.gyn. is effective and has been shown to reduce risky behavior by 70%. Randomized studies reported significant reductions in alcohol use and improved newborn outcomes after intervention.
Prenatal exposure to alcohol is one of the leading preventable causes of birth defects. With so many risk factors out of a mother’s control during pregnancy, why risk it?
Dr. Conry is assistant physician in chief at the Permanente Medical Group in Roseville, Calif., and associate clinical professor of ob.gyn. at the University of California, Davis. She has been a practicing ob.gyn. with the Permanente Medical Group for more than 20 years. She is a past president of the American College of Obstetricians and Gynecologists.
Recent findings by the American Academy of Pediatrics (AAP) regarding alcohol consumption during pregnancy are quite clear: Don’t drink at all while pregnant. This warning from the AAP is consistent with advice from the American College of Obstetricians and Gynecologists (ACOG) and the Centers for Disease Control and Prevention: No safe level of alcohol consumption during pregnancy has been identified, and no period during pregnancy appears to be safe for alcohol consumption.
In fact, ACOG has recommended screening for alcohol use early in pregnancy since 1977. However, the CDC reports that 1 in 10 pregnant women aged 18-44 years has consumed alcohol in the past 30 days. Some recent studies claim that a small number of drinks during pregnancy could be safe, and I’ve even seen some reports of ob.gyns. allowing alcohol use during pregnancy.
With such disparities between recommendations and patient behavior, the role of the obstetrician is more crucial than ever. Education, prevention, and intervention are essential to good health for mother and baby.
It has long been recognized that heavy drinking during pregnancy is associated with Fetal Alcohol Syndrome, but there are more subtle effects that fall under the umbrella of Fetal Alcohol Spectrum Disorders. These effects may include physical, mental, behavioral, and learning disabilities with lifelong implications and may not be detected until a child starts school.
Screening for alcohol use is of the utmost importance. Three simple steps have been proven effective in identifying women who drink at risky levels and engage them in changing behavior to reduce their risk for an alcohol-exposed pregnancy. These steps include asking about alcohol use, providing brief motivation-enhancing interventions, and follow-up for women who engage in risky drinking. See ACOG’s Fetal Alcohol Syndrome Prevention Tool Kit for more detail.
It is worth noting that a positive screen does not always mean a woman needs to be referred to treatment. Brief intervention by an ob.gyn. is effective and has been shown to reduce risky behavior by 70%. Randomized studies reported significant reductions in alcohol use and improved newborn outcomes after intervention.
Prenatal exposure to alcohol is one of the leading preventable causes of birth defects. With so many risk factors out of a mother’s control during pregnancy, why risk it?
Dr. Conry is assistant physician in chief at the Permanente Medical Group in Roseville, Calif., and associate clinical professor of ob.gyn. at the University of California, Davis. She has been a practicing ob.gyn. with the Permanente Medical Group for more than 20 years. She is a past president of the American College of Obstetricians and Gynecologists.
AAP: Most Parents Develop Vaccine Preferences Before Pregnancy
WASHINGTON – Most parents know whether they want to vaccinate their child before the child is even conceived, a study showed.
“Is giving [vaccine] information out at … well-child visits in the pediatrician’s office too late? I think we’re still too early [in our research] to say if that’s the case,” said James N. Yarnall, a fourth-year medical student at the University of North Carolina at Chapel Hill.
The study grew out of talking to parents about their hesitancy toward the hepatitis B vaccine, recommended just after birth, said Mr. Yarnall, whose mentor, pediatrician Jacob Lohr of University of North Carolina Health Care, began asking parents why they were turning down the vaccine.
“For a lot of them, they said, ‘We’ve known for a long time,’ ” Mr. Yarnall said.
The two gave surveys to 171 parents (56% of the 304 they asked) who had given birth to a child between February and April 2015 at the University of North Carolina Women’s Hospital in Chapel Hill. The parents answered the question, “When did you develop your preferences for all/certain vaccines for your new baby?”
Among all the parents, 72% said they had developed their vaccination preferences for their newborn before the child had been conceived. The parents reported that friends and family, medical staff, and government agencies such as the Centers for Disease Control and Prevention, played a role in their decisions about vaccines.
Two thirds (66%) of first-time parents reported developing immunization preferences before conception, while 77% of parents with previous children said they had, although the difference was not significant (P = .12). Fathers trended toward being slightly more likely than mothers to have decided their vaccine preferences preconception, but that finding was not significant and weakened further after demographic differences were accounted for.
Parents who had discussed vaccines with one another and who had higher levels of education also were more likely to have decided before conception whether they would vaccinate their newborn. Those who made their vaccine decisions before conception were significantly more likely to plan to vaccinate their children than to decline some or all vaccines (P = .01). Again, this finding was no longer significant following adjustment for demographic covariates.
Although these findings suggest that discussions about vaccines in the pediatrician’s office are coming long past the time when most parents have made up their minds, it’s less clear where to go from here, Mr. Yarnall said.
He and Dr. Lohr are working on a larger study with multiple sites through the Better Outcomes Through Research in Newborns (BORN) network to confirm these findings and determine what the next steps might be.
“We want to make sure the results from this pilot study are reproducible with more national sites,” Mr. Yarnall said. “Once we get those results, we can take it from there.”
WASHINGTON – Most parents know whether they want to vaccinate their child before the child is even conceived, a study showed.
“Is giving [vaccine] information out at … well-child visits in the pediatrician’s office too late? I think we’re still too early [in our research] to say if that’s the case,” said James N. Yarnall, a fourth-year medical student at the University of North Carolina at Chapel Hill.
The study grew out of talking to parents about their hesitancy toward the hepatitis B vaccine, recommended just after birth, said Mr. Yarnall, whose mentor, pediatrician Jacob Lohr of University of North Carolina Health Care, began asking parents why they were turning down the vaccine.
“For a lot of them, they said, ‘We’ve known for a long time,’ ” Mr. Yarnall said.
The two gave surveys to 171 parents (56% of the 304 they asked) who had given birth to a child between February and April 2015 at the University of North Carolina Women’s Hospital in Chapel Hill. The parents answered the question, “When did you develop your preferences for all/certain vaccines for your new baby?”
Among all the parents, 72% said they had developed their vaccination preferences for their newborn before the child had been conceived. The parents reported that friends and family, medical staff, and government agencies such as the Centers for Disease Control and Prevention, played a role in their decisions about vaccines.
Two thirds (66%) of first-time parents reported developing immunization preferences before conception, while 77% of parents with previous children said they had, although the difference was not significant (P = .12). Fathers trended toward being slightly more likely than mothers to have decided their vaccine preferences preconception, but that finding was not significant and weakened further after demographic differences were accounted for.
Parents who had discussed vaccines with one another and who had higher levels of education also were more likely to have decided before conception whether they would vaccinate their newborn. Those who made their vaccine decisions before conception were significantly more likely to plan to vaccinate their children than to decline some or all vaccines (P = .01). Again, this finding was no longer significant following adjustment for demographic covariates.
Although these findings suggest that discussions about vaccines in the pediatrician’s office are coming long past the time when most parents have made up their minds, it’s less clear where to go from here, Mr. Yarnall said.
He and Dr. Lohr are working on a larger study with multiple sites through the Better Outcomes Through Research in Newborns (BORN) network to confirm these findings and determine what the next steps might be.
“We want to make sure the results from this pilot study are reproducible with more national sites,” Mr. Yarnall said. “Once we get those results, we can take it from there.”
WASHINGTON – Most parents know whether they want to vaccinate their child before the child is even conceived, a study showed.
“Is giving [vaccine] information out at … well-child visits in the pediatrician’s office too late? I think we’re still too early [in our research] to say if that’s the case,” said James N. Yarnall, a fourth-year medical student at the University of North Carolina at Chapel Hill.
The study grew out of talking to parents about their hesitancy toward the hepatitis B vaccine, recommended just after birth, said Mr. Yarnall, whose mentor, pediatrician Jacob Lohr of University of North Carolina Health Care, began asking parents why they were turning down the vaccine.
“For a lot of them, they said, ‘We’ve known for a long time,’ ” Mr. Yarnall said.
The two gave surveys to 171 parents (56% of the 304 they asked) who had given birth to a child between February and April 2015 at the University of North Carolina Women’s Hospital in Chapel Hill. The parents answered the question, “When did you develop your preferences for all/certain vaccines for your new baby?”
Among all the parents, 72% said they had developed their vaccination preferences for their newborn before the child had been conceived. The parents reported that friends and family, medical staff, and government agencies such as the Centers for Disease Control and Prevention, played a role in their decisions about vaccines.
Two thirds (66%) of first-time parents reported developing immunization preferences before conception, while 77% of parents with previous children said they had, although the difference was not significant (P = .12). Fathers trended toward being slightly more likely than mothers to have decided their vaccine preferences preconception, but that finding was not significant and weakened further after demographic differences were accounted for.
Parents who had discussed vaccines with one another and who had higher levels of education also were more likely to have decided before conception whether they would vaccinate their newborn. Those who made their vaccine decisions before conception were significantly more likely to plan to vaccinate their children than to decline some or all vaccines (P = .01). Again, this finding was no longer significant following adjustment for demographic covariates.
Although these findings suggest that discussions about vaccines in the pediatrician’s office are coming long past the time when most parents have made up their minds, it’s less clear where to go from here, Mr. Yarnall said.
He and Dr. Lohr are working on a larger study with multiple sites through the Better Outcomes Through Research in Newborns (BORN) network to confirm these findings and determine what the next steps might be.
“We want to make sure the results from this pilot study are reproducible with more national sites,” Mr. Yarnall said. “Once we get those results, we can take it from there.”
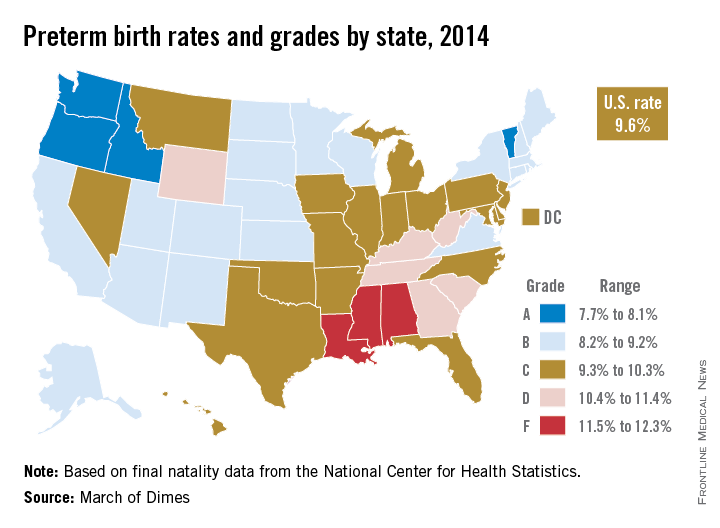
U.S. brings home a ‘C’ on preterm birth report card
The United States’ 9.6% preterm birth rate in 2014 earned a grade of C on the March of Dimes 2015 Premature Birth Report Card.
Although the United States ranks poorly among high-resource countries when it comes to preterm births, the organization said in a statement, the rate for 2014 was good enough to meet the March of Dimes 2020 goal, which was 9.6%. The organization has reset the goal to 8.1% of live births by 2020.
“Reaching our goal ahead of schedule is progress, but it is not victory – our work is far from done,” Dr. Jennifer L. Howse, president of the March of Dimes, said in the statement.
Among the states, Oregon (7.7%), Vermont (7.9%), Idaho (8.1%), and Washington (8.1%) were the only ones to receive a grade of A. Mississippi (12.9%), Louisiana (12.3%), and Alabama (11.7%) were at the bottom of the class with Fs. Nineteen states earned a B, 18 states and the District of Columbia received a C, and six states received a D, according to final natality data for 2014 from the National Center for Health Statistics.
Among the 100 cities with the highest numbers of births, Portland, Ore., had the lowest preterm birth rate at 7.2%, while Shreveport, La., had the highest rate at 18.8%. The only other cities that earned an A were Oxnard, Calif.; Seattle;and St. Paul, Minn. A total of 17 cities received a grade of F.
“As our new list of city preterm birth rates highlights, many areas of the country, and tens of thousands of families, are not sharing in [the country’s] success. No baby should have to battle the health consequences of an early birth. All babies, everywhere deserve a healthy start in life,” Dr. Howse said.
The United States’ 9.6% preterm birth rate in 2014 earned a grade of C on the March of Dimes 2015 Premature Birth Report Card.
Although the United States ranks poorly among high-resource countries when it comes to preterm births, the organization said in a statement, the rate for 2014 was good enough to meet the March of Dimes 2020 goal, which was 9.6%. The organization has reset the goal to 8.1% of live births by 2020.
“Reaching our goal ahead of schedule is progress, but it is not victory – our work is far from done,” Dr. Jennifer L. Howse, president of the March of Dimes, said in the statement.
Among the states, Oregon (7.7%), Vermont (7.9%), Idaho (8.1%), and Washington (8.1%) were the only ones to receive a grade of A. Mississippi (12.9%), Louisiana (12.3%), and Alabama (11.7%) were at the bottom of the class with Fs. Nineteen states earned a B, 18 states and the District of Columbia received a C, and six states received a D, according to final natality data for 2014 from the National Center for Health Statistics.
Among the 100 cities with the highest numbers of births, Portland, Ore., had the lowest preterm birth rate at 7.2%, while Shreveport, La., had the highest rate at 18.8%. The only other cities that earned an A were Oxnard, Calif.; Seattle;and St. Paul, Minn. A total of 17 cities received a grade of F.
“As our new list of city preterm birth rates highlights, many areas of the country, and tens of thousands of families, are not sharing in [the country’s] success. No baby should have to battle the health consequences of an early birth. All babies, everywhere deserve a healthy start in life,” Dr. Howse said.

The United States’ 9.6% preterm birth rate in 2014 earned a grade of C on the March of Dimes 2015 Premature Birth Report Card.
Although the United States ranks poorly among high-resource countries when it comes to preterm births, the organization said in a statement, the rate for 2014 was good enough to meet the March of Dimes 2020 goal, which was 9.6%. The organization has reset the goal to 8.1% of live births by 2020.
“Reaching our goal ahead of schedule is progress, but it is not victory – our work is far from done,” Dr. Jennifer L. Howse, president of the March of Dimes, said in the statement.
Among the states, Oregon (7.7%), Vermont (7.9%), Idaho (8.1%), and Washington (8.1%) were the only ones to receive a grade of A. Mississippi (12.9%), Louisiana (12.3%), and Alabama (11.7%) were at the bottom of the class with Fs. Nineteen states earned a B, 18 states and the District of Columbia received a C, and six states received a D, according to final natality data for 2014 from the National Center for Health Statistics.
Among the 100 cities with the highest numbers of births, Portland, Ore., had the lowest preterm birth rate at 7.2%, while Shreveport, La., had the highest rate at 18.8%. The only other cities that earned an A were Oxnard, Calif.; Seattle;and St. Paul, Minn. A total of 17 cities received a grade of F.
“As our new list of city preterm birth rates highlights, many areas of the country, and tens of thousands of families, are not sharing in [the country’s] success. No baby should have to battle the health consequences of an early birth. All babies, everywhere deserve a healthy start in life,” Dr. Howse said.

ITC: Levothyroxine may improve obstetric outcomes in women with low thyroid function
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
AT ITC 2015
Key clinical point: Levothyroxine may improve obstetric outcomes in women with low thyroid function.
Major finding: Stillbirths were five times more likely among women who were untreated for high levels of thyroid-stimulating hormone during pregnancy.
Data source: The analysis comprised about 14,000 mother-child pairs.
Disclosures: Dr. Taylor had no financial disclosures.
Endometriosis: Expert Answers to 7 Crucial Questions on Diagnosis
IN THIS ARTICLE
• The “why” of endometriosis
• Environmental factors, estrogen, and endometriosis
• Is imaging useful?
• When is diagnostic laparoscopy clearly indicated?
CASE M.L. is a 32-year-old nulliparous woman who is referred to your office by her primary care provider for chronic pelvic pain. She reports severe dysmenorrhea as her main symptom, but she also mentions dyspareunia. She says these symptoms have been present for several years but have increased in intensity gradually. She asks what you consider to be the most likely diagnosis.
What potential diagnoses do you mention to her? And how do you identify the cause of her pain?
Although endometriosis—the presence of endometrial tissue outside the uterus—affects at least 5 million women of reproductive age in the United States alone, it can be a challenging diagnosis for several reasons.
“Endometriosis is a great masquerader,” says Linda Giudice, MD, PhD. “It presents with a variety of pain patterns, intensities, and triggers. It can also involve symptoms that overlap those of other disorders, including disorders of the gastrointestinal and urinary tracts.”
Although endometriosis falls within the differential diagnosis of chronic pelvic pain, “it is usually not high on the list in the primary care setting (adult and adolescent),” adds Dr. Giudice.
John R. Lue, MD, MPH, an author of the most recent practice bulletin on endometriosis from the American College of Obstetricians and Gynecologists,1 sees the situation similarly.
“The main challenge in the diagnosis of endometriosis is that its presentation mimics other causes of chronic pelvic pain,” he says. “Pelvic pain due to endometriosis is usually chronic (lasting ≥ 6 months). It is associated with dysmenorrhea in 50% to 90% of cases, as well as with dyspareunia, deep pelvic pain, and lower abdominal pain with or without back and loin pain. The pain can occur unpredictably and intermittently throughout the menstrual cycle or it can be continuous. In addition, it can be dull, throbbing, or sharp and may be exacerbated by physical activity.2,3 Up to 20% of women with endometriosis have concurrent pain conditions.”4
Among other diseases of the female pelvis that have a relatively similar presentation, Dr. Lue adds, are pathologies of the
• Uterus (adenomyosis, fibroids)
• Fallopian tube (hydrosalpinx)
• Ovaries (ovarian cysts)
• Bladder (interstitial cystitis)
• Bowel (irritable bowel syndrome)
• Musculoskeletal system (piriformis syndrome).
Before pelvic pain is attributed to endometriosis, he says, the provider should rule out bowel, bladder, musculoskeletal, and psychiatric causes.
This article focuses on seven questions, the answers to which are critical to narrowing in on the diagnosis of endometriosis, including essential factors to consider in the patient history, imaging and other diagnostic tools, and considerations in surgical exploration.
1. WHY SUCH A LONG DELAY IN DIAGNOSIS?
Investigators exploring the length of time from a patient’s presentation with symptoms to diagnosis have found it to be particularly long for endometriosis, ranging from six to 11 years.
Because endometriosis is usually not high on the list of differential diagnoses for chronic pelvic pain in the primary care setting, a patient may not be referred to a gynecologist unless those symptoms include severe dysmenorrhea, dyspareunia, or similar findings. Once the referral is made, the gynecologist “will usually try contraceptive steroids, NSAIDs, or second-line progestins before a diagnosis is made,” says Dr. Giudice.5
The delay in diagnosis “is astounding,” she adds, “and has its roots in empiric medical therapies and a combination of patients fearing a diagnosis of cancer and reluctance of gynecologists to perform laparoscopy on adolescents.”6 Another possible cause of diagnostic delay: Some adolescent girls may not realize when their pain is severe. Because they may have always experienced a high degree of pain since menarche, they may assume it to be a normal aspect of womanhood and delay seeking help, says Pamela Stratton, MD.
Continue to learn any biomarkers proved to be useful diagnostic tools >>
2. HAVE ANY BIOMARKERS PROVED TO BE USEFUL DIAGNOSTIC TOOLS?
Any biomarker proven to reliably identify endometriosis would be a boon to medicine, as it would provide a noninvasive or minimally invasive alternative to diagnostic laparoscopy, the current gold standard. Regrettably, the search for such a biomarker has produced “disappointing results,” says Dr. Giudice.
“Recent systematic reviews of all proposed endometriosis-related biomarkers over the past 25 years in serum, plasma, urine, and endometrium could not identify an unequivocally clinically useful biomarker or panel of biomarkers,” she notes.7,8 “This is due mainly to low numbers of subjects, small populations for validations, cycle/hormonal- and disease stage–dependence, poorly defined controls, and low sensitivity and specificity.”
One hopeful development: “Whole genome transcriptomics of archived endometrial tissue and machine learning found several classifiers to diagnose and stage endometriosis with high accuracy that were validated on an independent sample set,” says Dr. Giudice.9 “However, these data now warrant a prospective, multisite study for further validation.”
Continue for key aspects of patient history >>
3. WHAT ASPECTS OF THE PATIENT HISTORY ARE KEY?
Dr. Stratton recommends that clinicians begin their evaluation of the patient with pain by asking her to describe that pain: how long she has had it, when it occurs, and which areas are affected.
“Most women with endometriosis-associated pain have chronic pelvic pain,” Dr. Stratton continues.5 “Up to 90% of those have dysmenorrhea or cyclic pain with menses.”10 In addition, women with endometriosis “commonly report having pain with any bleeding or spotting. About 30% of women diagnosed with endometriosis initially present to their gynecologist with dyspareunia.”11
“Episodic pain with menses may become more constant, lasting for many days of the month,” says Dr. Stratton. “Women with dyschezia or dysuria may have endometriosis lesions associated with the bowel or bladder, respectively.12 When women with these symptoms do not have lesions on the bowel or bladder, these pain symptoms may occur because of higher peritoneal hormone and inflammatory factor levels or because adjacent organs share the neural networks.”
Dr. Giudice views the history similarly: “I believe listening to the patient is essential in evaluating the possibility of her having endometriosis. This involves asking her to describe where her pain is, grading it on a scale of 1 to 10, identifying when in her cycle it occurs, and learning what makes it better or worse.”
“It also is important to assess the quality of the pain,” she adds. “Does it radiate, does it limit her daily activities, does it interfere with her relationships, intercourse, work, school? Is it associated with bowel movements, urination, other pain syndromes?”
“Having a pain questionnaire is a great help so that patients have a chance to reflect on these and other questions that help to frame the pain associated with endometriosis when they come for consultation,” she adds.
By determining if pain is associated with menstruation or spotting, the clinician is better informed about the value of menstrual suppression, says Dr. Stratton. “Determining what makes the pain better or worse can help define triggers which, if treated, can decrease the likelihood of episodes of pain.”
“A detailed history of any medical or surgical treatments and their outcome is helpful in guiding future treatment,” she adds. “While hormonal therapy has been a mainstay of treatment, in some women, some hormonal treatments may worsen pain or have unacceptable adverse effects, such as worsening depression or anxiety. In addition, some pain—especially that associated with deep lesions—may be relieved by surgical treatment;13,14 pain that worsened after surgery may suggest neural damage.”
“As there is an engagement of the central nervous system, endometriosis is considered a central sensitivity syndrome in which women may also have other sites of pain,” Dr. Stratton says. “Thus, obtaining a history about current symptoms or prior diagnosis of irritable bowel syndrome, interstitial cystitis/painful bladder, migraines, fibromyalgia, or chronic fatigue syndrome is beneficial.10,15-17 Facilitating treatment for these comorbidities is a key principle in helping women with endometriosis-associated pain, as any condition that triggers or perpetuates pain warrants treatment.”
Continue for what the physical exam entails >>
4. WHAT SHOULD THE PHYSICAL EXAM ENTAIL?
“An abdominal exam and a pelvic exam are essential in evaluating pain in a woman when endometriosis is suspected,” says Dr. Giudice. “Sometimes the latter is challenging in young teens and can be deferred.” Overall, however, “the pelvic exam can give insight into pain triggers, adnexal masses (possible endometriomas), and mobility of pelvic organs. A rectovaginal exam is important in evaluating deep infiltrating disease and to gauge the pelvic pain landscape overall. In addition, palpating the pelvic floor musculature is important to distinguish pelvic floor muscle spasm from endometriosis pain.”
“The challenge for clinicians is to think beyond the endometrial implants, taking into account multiple factors that influence pain perception,” says Dr. Stratton. During the examination, the clinician should begin by mapping the regions of pain in the abdomen and back, “distinguishing musculoskeletal pain from deep pain. Determining whether pains are focused or diffuse is also important.”
Dr. Stratton recommends that the routine pelvic exam be modified because a standard bimanual exam “confuses pain signals from the pelvic floor, abdominal wall, bladder, and other viscera. For this reason, a pain-oriented assessment is mandatory.”
Begin with a single digital examination to map tender areas, Dr. Stratton advises. Then consider the size, shape, and mobility of reproductive and pelvic organs. “A bimanual exam will help identify adnexal masses like endometriomas,” she says.
Endometriomas usually are not associated with pain, she adds, but “they are associated with deep infiltrating lesions. Nodularity along the uterosacral ligaments, limited reproductive organ mobility, and thickening of the rectovaginal septum also suggest deep infiltrating lesions. Importantly, deep infiltrating lesions are the lesion type most associated with pain.”18,19
Continue to learn if imaging is useful in the diagnosis of endometriosis >>
5. IS IMAGING USEFUL IN THE DIAGNOSIS OF ENDOMETRIOSIS?
Laparoscopy remains the gold standard for diagnosis of endometriosis, observes Steven R. Goldstein, MD. Visualization of endometriotic implants at the time of surgery—with histologic assessment—offers definitive confirmation of the diagnosis. The physical examination, too, can offer a strong suggestion of endometriosis, he says.
“In the past, the pelvic examination and history often were the sine qua non for patients with pain,” Dr. Goldstein says. “Extreme dysmenorrhea and pain between periods, especially with intercourse, defecation, and exercise, all increased the suspicion of endometriosis. People used to talk about feeling nodularity in the uterosacral ligaments and finding decreased mobility of pelvic structures—but I don’t have any question that the skill of today’s gynecologists in doing a bimanual pelvic exam is a fraction of what it was in years gone by, because they haven’t had the necessity of experience. The first thing they do if there’s any question is send the patient for an ultrasound.”
Of course, ultrasound can be especially helpful in identifying endometriomas—sometimes called chocolate cysts—in the ovary. Endometriomas can have a solid appearance on ultrasound, says Dr. Goldstein, because the fluid they contain (dried blood) is sonolucent or pure black on ultrasound, similar to amniotic fluid or the fluid seen in the bladder. “This ‘chocolate’ fluid contained in endometriomas is homogeneous, particulate, and very monotonous in its appearance, in contrast to the internal echoes observed in hemorrhagic corpus lutea, which are very cobweb-like and can sometimes mimic papillary projections,” he adds.
“What’s absolutely essential when imaging a suspected endometrioma by ultrasound is that there be no evidence of any blood flow contained within that structure. Because it’s dried blood, it shouldn’t have any vascularity. If you see blood flow inside what you would call an endometrioma, you need to rethink your diagnosis,” he says.
In some cases, a supposed endometrioma lacks a black, sonolucent appearance, but “the clinician often can tell that it’s a cystic structure by the very bright posterior wall—what we call posterior wall acoustic enhancement—even though the interior of the structure may appear sort of grayish or whitish rather than the pure black of a simple cyst. It’s still fluid-filled,” Dr. Goldstein says.
In some instances, even endometriotic nodules can be imaged by ultrasound, he adds. “There’s an increasing body of literature that suggests that, if you look carefully in people with deep infiltrating endometriosis, you can often see solid-appearing nodules in the rectovaginal septum or between the uterus and bladder. With the kind of resolution that we now have with the vaginal probe, some of these nodules can be seen. That’s somewhat new, and it’s a function of two things—people looking for endometriosis and the better resolution of more modern equipment.”
Dr. Goldstein believes that MRI is “almost never” indicated in the diagnosis of endometriosis. A more helpful approach would be a consultative ultrasound with someone with more experience. However, when that is not available, or “in areas where you have excellent backup in terms of pelvic MRI, that may be the way to go. I don’t think so,” he demurs, “and some of my colleagues would be very upset at the thought of needing to use MRI to diagnose endometriosis. But in the occasional confusing or difficult case, depending on the quality of the referral pattern you have, it might make sense."
Continue to learn when diagnostic laparoscopy is clearly indicated >>
6. WHEN IS DIAGNOSTIC LAPAROSCOPY CLEARLY INDICATED?
Dr. Giudice believes that laparoscopy—with the intention to treat endometriosis, if present—“is essential when firstline medical therapy fails or when pain is acute and severe.”5
Dr. Stratton concurs. “Any woman with chronic pain wants to know what is causing the pain,” she says. Therefore, “women report a benefit from knowing that their pain is associated with endometriosis.6 However, diagnostic laparoscopy alone, with the sole purpose of determining the presence of endometriosis but not treating the lesions, is no longer performed, as it poses little benefit to the patient other than peace of mind.”
“The general trend in the US has been to first use hormonal treatments when the diagnosis of endometriosis is suspected, prior to performing surgery,” Dr. Stratton says.1 In many cases, by using cyclic combined hormonal contraceptives to reduce menstrual flow or “suppressing menstruation with continuous combined hormonal contraceptives,” gonadotropin-releasing hormone analogues (combined with progestin to prevent bone loss) “or continuous progestin alone may be effective in decreasing pain. Not surprisingly, these hormonal approaches are effective for any chronic pelvic pain, even for women who do not have the surgical diagnosis of endometriosis.”20
“When the firstline approach to chronic pelvic pain is hormonal treatment, laparoscopy is considered when these medical treatments have failed to control the pain or are poorly tolerated, or when the diagnosis of endometriosis is in question,” Dr. Stratton says.
“Laparoscopy to treat endometriomas is indicated if an endometrioma is enlarging or measures more than 4 cm in diameter, or if the diagnosis of an ovarian mass is in question,” she explains. “While surgeons have previously been aggressive in removing endometriomas, this practice may have negative consequences on ovarian function. Because endometriomas are pseudocysts, removing them completely leads to the removal of viable ovarian tissue and may diminish ovarian reserve.”21,22
Continue for the surgical appearance of endometriosis >>
7. WHAT IS THE SURGICAL APPEARANCE OF ENDOMETRIOSIS?
Dr. Giudice returns to the enigmatic nature of endometriosis in addressing this question, mentioning its “many faces” at the time of surgery. “It is imperative that the surgeon recognize the disease in its many forms,” she says. “Also, it is especially helpful at the time of surgery if suspected lesions are biopsied and sent to pathology to have the diagnosis made unequivocally.”5
As for the surgical appearance of endometriosis, Dr. Stratton notes that there are three types of lesions—“superficial lesions, deep infiltrating lesions, and endometriomas. Endometriomas occur almost exclusively in the ovary and are pseudocysts without an identifiable cystic lining. They vary in dimension from a few millimeters to several centimeters.”
“Superficial peritoneal endometriosis lesions have a variable appearance, with some lesions being clear or red; some brown, blue or black; and some having a white appearance, like a scar,” says Dr. Stratton. “Endometriosis can be diagnosed on histologic examination of any of these lesion types."
“Overall, single-color lesions have similar frequencies of biopsy-confirmed endometriosis (59% to 62%),” she says.23 “These lesion appearances likely represent different stages of development of endometriosis, with red or clear lesions occurring first, soon after endometrial tissue implantation; black, blue, or brown lesions occurring later, in response to the hormones varying in the menstrual cycle; and white lesions occurring as the lesions age. Deep infiltrating lesions generally have blue/black or white features.”
“Wide, deep, multiple-color lesions in the cul-de-sac, ovarian fossa, or uterosacral ligaments are most likely endometriosis,” Dr. Stratton adds.23 Only lesions with multiple colors have a significantly higher percentage of positive biopsies (76%). Importantly, more than half of women with only subtle lesions (small red or white lesions) have endometriosis.
You tell the patient that endometriosis is one of the possible diagnoses for her chronic pelvic pain, and you take a focused history. During a pelvic examination, you observe that her right ovary lacks mobility, and you map a number of trigger points for her pain. Transvaginal ultrasound results suggest the presence of nodules in the rectovaginal septum. You begin empiric treatment with continuous combined hormonal contraceptives to suppress menstruation. On her next visit, M.L. reports reduced but still bothersome pain. Laparoscopy reveals a 2-cm endometrioma in the right ovary and deep infiltrating lesions in the cul-de-sac. The endometrioma is resected. Histology confirms the diagnosis of endometriosis.
REFERENCES
1. American College of Obstetricians and Gynecologists. Practice Bulletin #114: Management of endometriosis. Obstet Gynecol. 2010; 116(1):223-236.
2. Sanfilippo JS, Wakim NG, Schikler KN, Yussman MA. Endometriosis in association with uterine anomaly. Am J Obstet Gynecol. 1986; 154(1):39-43.
3. Taylor HS, Bagot C, Kardana A. HOX gene expression is altered in the endometrium of women with endometriosis. Hum Reprod. 1999; 14(5):1328-1331.
4. Berkley KJ, Stratton P. Mechanisms: lessons from translational studies of endometriosis. In: Giamberardino MA, ed. Visceral Pain: Clinical, Pathophysiological and Therapeutic Aspects. Oxford, UK: Oxford University Press; 2009:39-50.
5. Giudice LC. Clinical practice: endometriosis. N Engl J Med. 2010;362(25): 2389-2398.
6. Ballard K, Lowton K, Wright J. What’s the delay: a qualitative study of women’s experiences of reaching a diagnosis of endometriosis. Fertil Steril. 2006;86(5):1296-1301.
7. May KE, Conduit-Hulbert SA, Villar J, et al. Peripheral biomarkers of endometriosis: a systematic review. Hum Reprod Update. 2010; 16(6):651-674.
8. May KE, Villar J, Kirtley S, et al. Endometrial alterations in endometriosis: a systematic review of putative biomarkers. Hum Reprod Update. 2011; 17(5):637-653.
9. Tamaresis JS, Irwin JC, Goldfien GA, et al. Molecular classification of endometriosis and disease stage using high-dimensional genomic data. Endocrinology. 2014;155(12):4986-4999.
10. Sinaii N, Cleary SD, Ballweg ML, et al. High rates of autoimmune and endocrine disorders, fibromyalgia, chronic fatigue syndrome and atopic diseases among women with endometriosis: a survey analysis. Hum Reprod. 2002;17(10):2715-2724.
11. De Graaff AA, D’Hooghe TM, Dunselman GA, et al. The significant effect of endometriosis on physical, mental and social wellbeing: results from an international cross-sectional survey. Hum Reprod. 2013;28(10):2677-2685.
12. Lafay Pillet MC, Huchon C, Santulli P, et al. A clinical score can predict associated deep infiltrating endometriosis before surgery for an endometrioma. Hum Reprod. 2014;29(8):1666-1676.
13. Healey M, Cheng C, Kaur H. To excise or ablate endometriosis? A prospective randomized double-blinded trial after 5-year follow-up. J Minim Invasive Gynecol. 2014;21(6):999-1004.
14. Anaf V, El Nakadi I, De Moor V, et al. Increased nerve density in deep infiltrating endometriotic nodules. Gynecol Obstet Invest. 2011;71(2):112-117.
15. Stratton P, Berkley KJ. Chronic pelvic pain and endometriosis: translational evidence of the relationship and implications. Hum Reprod Update. 2011;17(3):327-346.
16. Karp BI, Sinaii N, Nieman LK, et al. Migraine in women with chronic pelvic pain with and without endometriosis. Fertil Steril. 2011;95(3):895-899.
17. Berkley KJ. A life of pelvic pain. Physiol Behav. 2005;86(3):272-280.
18. Fauconnier A, Chapron C. Endometriosis and pelvic pain: epidemiological evidence of the relationship and implications. Hum Reprod Update. 2005;11(6):595-606.
19. Vercellini P, Fedele L, Aimi G, et al. Association between endometriosis stage, lesion type, patient characteristics and severity of pelvic pain symptoms: a multivariate analysis of over 1000 patients. Hum Reprod. 2007;22(1):266-271.
20. Ling FW. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Pelvic Pain Study Group. Obstet Gynecol. 1999;93(1):51-58.
21. Muzii L, Di Tucci C, Di Feliciantonio M, et al. The effect of surgery for endometrioma on ovarian reserve evaluated by antral follicle count: a systematic review and meta-analysis. Hum Reprod. 2014;29(10):2190-2198.
22. Muzii L, Luciano AA, Zupi E, Panici PB. Effect of surgery for endometrioma on ovarian function: a different point of view. J Minim Invasive Gynecol. 2014;21(4):531-533.
23. Stegmann BJ, Sinaii N, Liu S, et al. Using location, color, size, and depth to characterize and identify endometriosis lesions in a cohort of 133 women. Fertil Steril. 2008;89(6):1632-1636.
IN THIS ARTICLE
• The “why” of endometriosis
• Environmental factors, estrogen, and endometriosis
• Is imaging useful?
• When is diagnostic laparoscopy clearly indicated?
CASE M.L. is a 32-year-old nulliparous woman who is referred to your office by her primary care provider for chronic pelvic pain. She reports severe dysmenorrhea as her main symptom, but she also mentions dyspareunia. She says these symptoms have been present for several years but have increased in intensity gradually. She asks what you consider to be the most likely diagnosis.
What potential diagnoses do you mention to her? And how do you identify the cause of her pain?
Although endometriosis—the presence of endometrial tissue outside the uterus—affects at least 5 million women of reproductive age in the United States alone, it can be a challenging diagnosis for several reasons.
“Endometriosis is a great masquerader,” says Linda Giudice, MD, PhD. “It presents with a variety of pain patterns, intensities, and triggers. It can also involve symptoms that overlap those of other disorders, including disorders of the gastrointestinal and urinary tracts.”
Although endometriosis falls within the differential diagnosis of chronic pelvic pain, “it is usually not high on the list in the primary care setting (adult and adolescent),” adds Dr. Giudice.
John R. Lue, MD, MPH, an author of the most recent practice bulletin on endometriosis from the American College of Obstetricians and Gynecologists,1 sees the situation similarly.
“The main challenge in the diagnosis of endometriosis is that its presentation mimics other causes of chronic pelvic pain,” he says. “Pelvic pain due to endometriosis is usually chronic (lasting ≥ 6 months). It is associated with dysmenorrhea in 50% to 90% of cases, as well as with dyspareunia, deep pelvic pain, and lower abdominal pain with or without back and loin pain. The pain can occur unpredictably and intermittently throughout the menstrual cycle or it can be continuous. In addition, it can be dull, throbbing, or sharp and may be exacerbated by physical activity.2,3 Up to 20% of women with endometriosis have concurrent pain conditions.”4
Among other diseases of the female pelvis that have a relatively similar presentation, Dr. Lue adds, are pathologies of the
• Uterus (adenomyosis, fibroids)
• Fallopian tube (hydrosalpinx)
• Ovaries (ovarian cysts)
• Bladder (interstitial cystitis)
• Bowel (irritable bowel syndrome)
• Musculoskeletal system (piriformis syndrome).
Before pelvic pain is attributed to endometriosis, he says, the provider should rule out bowel, bladder, musculoskeletal, and psychiatric causes.
This article focuses on seven questions, the answers to which are critical to narrowing in on the diagnosis of endometriosis, including essential factors to consider in the patient history, imaging and other diagnostic tools, and considerations in surgical exploration.
1. WHY SUCH A LONG DELAY IN DIAGNOSIS?
Investigators exploring the length of time from a patient’s presentation with symptoms to diagnosis have found it to be particularly long for endometriosis, ranging from six to 11 years.
Because endometriosis is usually not high on the list of differential diagnoses for chronic pelvic pain in the primary care setting, a patient may not be referred to a gynecologist unless those symptoms include severe dysmenorrhea, dyspareunia, or similar findings. Once the referral is made, the gynecologist “will usually try contraceptive steroids, NSAIDs, or second-line progestins before a diagnosis is made,” says Dr. Giudice.5
The delay in diagnosis “is astounding,” she adds, “and has its roots in empiric medical therapies and a combination of patients fearing a diagnosis of cancer and reluctance of gynecologists to perform laparoscopy on adolescents.”6 Another possible cause of diagnostic delay: Some adolescent girls may not realize when their pain is severe. Because they may have always experienced a high degree of pain since menarche, they may assume it to be a normal aspect of womanhood and delay seeking help, says Pamela Stratton, MD.
Continue to learn any biomarkers proved to be useful diagnostic tools >>
2. HAVE ANY BIOMARKERS PROVED TO BE USEFUL DIAGNOSTIC TOOLS?
Any biomarker proven to reliably identify endometriosis would be a boon to medicine, as it would provide a noninvasive or minimally invasive alternative to diagnostic laparoscopy, the current gold standard. Regrettably, the search for such a biomarker has produced “disappointing results,” says Dr. Giudice.
“Recent systematic reviews of all proposed endometriosis-related biomarkers over the past 25 years in serum, plasma, urine, and endometrium could not identify an unequivocally clinically useful biomarker or panel of biomarkers,” she notes.7,8 “This is due mainly to low numbers of subjects, small populations for validations, cycle/hormonal- and disease stage–dependence, poorly defined controls, and low sensitivity and specificity.”
One hopeful development: “Whole genome transcriptomics of archived endometrial tissue and machine learning found several classifiers to diagnose and stage endometriosis with high accuracy that were validated on an independent sample set,” says Dr. Giudice.9 “However, these data now warrant a prospective, multisite study for further validation.”
Continue for key aspects of patient history >>
3. WHAT ASPECTS OF THE PATIENT HISTORY ARE KEY?
Dr. Stratton recommends that clinicians begin their evaluation of the patient with pain by asking her to describe that pain: how long she has had it, when it occurs, and which areas are affected.
“Most women with endometriosis-associated pain have chronic pelvic pain,” Dr. Stratton continues.5 “Up to 90% of those have dysmenorrhea or cyclic pain with menses.”10 In addition, women with endometriosis “commonly report having pain with any bleeding or spotting. About 30% of women diagnosed with endometriosis initially present to their gynecologist with dyspareunia.”11
“Episodic pain with menses may become more constant, lasting for many days of the month,” says Dr. Stratton. “Women with dyschezia or dysuria may have endometriosis lesions associated with the bowel or bladder, respectively.12 When women with these symptoms do not have lesions on the bowel or bladder, these pain symptoms may occur because of higher peritoneal hormone and inflammatory factor levels or because adjacent organs share the neural networks.”
Dr. Giudice views the history similarly: “I believe listening to the patient is essential in evaluating the possibility of her having endometriosis. This involves asking her to describe where her pain is, grading it on a scale of 1 to 10, identifying when in her cycle it occurs, and learning what makes it better or worse.”
“It also is important to assess the quality of the pain,” she adds. “Does it radiate, does it limit her daily activities, does it interfere with her relationships, intercourse, work, school? Is it associated with bowel movements, urination, other pain syndromes?”
“Having a pain questionnaire is a great help so that patients have a chance to reflect on these and other questions that help to frame the pain associated with endometriosis when they come for consultation,” she adds.
By determining if pain is associated with menstruation or spotting, the clinician is better informed about the value of menstrual suppression, says Dr. Stratton. “Determining what makes the pain better or worse can help define triggers which, if treated, can decrease the likelihood of episodes of pain.”
“A detailed history of any medical or surgical treatments and their outcome is helpful in guiding future treatment,” she adds. “While hormonal therapy has been a mainstay of treatment, in some women, some hormonal treatments may worsen pain or have unacceptable adverse effects, such as worsening depression or anxiety. In addition, some pain—especially that associated with deep lesions—may be relieved by surgical treatment;13,14 pain that worsened after surgery may suggest neural damage.”
“As there is an engagement of the central nervous system, endometriosis is considered a central sensitivity syndrome in which women may also have other sites of pain,” Dr. Stratton says. “Thus, obtaining a history about current symptoms or prior diagnosis of irritable bowel syndrome, interstitial cystitis/painful bladder, migraines, fibromyalgia, or chronic fatigue syndrome is beneficial.10,15-17 Facilitating treatment for these comorbidities is a key principle in helping women with endometriosis-associated pain, as any condition that triggers or perpetuates pain warrants treatment.”
Continue for what the physical exam entails >>
4. WHAT SHOULD THE PHYSICAL EXAM ENTAIL?
“An abdominal exam and a pelvic exam are essential in evaluating pain in a woman when endometriosis is suspected,” says Dr. Giudice. “Sometimes the latter is challenging in young teens and can be deferred.” Overall, however, “the pelvic exam can give insight into pain triggers, adnexal masses (possible endometriomas), and mobility of pelvic organs. A rectovaginal exam is important in evaluating deep infiltrating disease and to gauge the pelvic pain landscape overall. In addition, palpating the pelvic floor musculature is important to distinguish pelvic floor muscle spasm from endometriosis pain.”
“The challenge for clinicians is to think beyond the endometrial implants, taking into account multiple factors that influence pain perception,” says Dr. Stratton. During the examination, the clinician should begin by mapping the regions of pain in the abdomen and back, “distinguishing musculoskeletal pain from deep pain. Determining whether pains are focused or diffuse is also important.”
Dr. Stratton recommends that the routine pelvic exam be modified because a standard bimanual exam “confuses pain signals from the pelvic floor, abdominal wall, bladder, and other viscera. For this reason, a pain-oriented assessment is mandatory.”
Begin with a single digital examination to map tender areas, Dr. Stratton advises. Then consider the size, shape, and mobility of reproductive and pelvic organs. “A bimanual exam will help identify adnexal masses like endometriomas,” she says.
Endometriomas usually are not associated with pain, she adds, but “they are associated with deep infiltrating lesions. Nodularity along the uterosacral ligaments, limited reproductive organ mobility, and thickening of the rectovaginal septum also suggest deep infiltrating lesions. Importantly, deep infiltrating lesions are the lesion type most associated with pain.”18,19
Continue to learn if imaging is useful in the diagnosis of endometriosis >>
5. IS IMAGING USEFUL IN THE DIAGNOSIS OF ENDOMETRIOSIS?
Laparoscopy remains the gold standard for diagnosis of endometriosis, observes Steven R. Goldstein, MD. Visualization of endometriotic implants at the time of surgery—with histologic assessment—offers definitive confirmation of the diagnosis. The physical examination, too, can offer a strong suggestion of endometriosis, he says.
“In the past, the pelvic examination and history often were the sine qua non for patients with pain,” Dr. Goldstein says. “Extreme dysmenorrhea and pain between periods, especially with intercourse, defecation, and exercise, all increased the suspicion of endometriosis. People used to talk about feeling nodularity in the uterosacral ligaments and finding decreased mobility of pelvic structures—but I don’t have any question that the skill of today’s gynecologists in doing a bimanual pelvic exam is a fraction of what it was in years gone by, because they haven’t had the necessity of experience. The first thing they do if there’s any question is send the patient for an ultrasound.”
Of course, ultrasound can be especially helpful in identifying endometriomas—sometimes called chocolate cysts—in the ovary. Endometriomas can have a solid appearance on ultrasound, says Dr. Goldstein, because the fluid they contain (dried blood) is sonolucent or pure black on ultrasound, similar to amniotic fluid or the fluid seen in the bladder. “This ‘chocolate’ fluid contained in endometriomas is homogeneous, particulate, and very monotonous in its appearance, in contrast to the internal echoes observed in hemorrhagic corpus lutea, which are very cobweb-like and can sometimes mimic papillary projections,” he adds.
“What’s absolutely essential when imaging a suspected endometrioma by ultrasound is that there be no evidence of any blood flow contained within that structure. Because it’s dried blood, it shouldn’t have any vascularity. If you see blood flow inside what you would call an endometrioma, you need to rethink your diagnosis,” he says.
In some cases, a supposed endometrioma lacks a black, sonolucent appearance, but “the clinician often can tell that it’s a cystic structure by the very bright posterior wall—what we call posterior wall acoustic enhancement—even though the interior of the structure may appear sort of grayish or whitish rather than the pure black of a simple cyst. It’s still fluid-filled,” Dr. Goldstein says.
In some instances, even endometriotic nodules can be imaged by ultrasound, he adds. “There’s an increasing body of literature that suggests that, if you look carefully in people with deep infiltrating endometriosis, you can often see solid-appearing nodules in the rectovaginal septum or between the uterus and bladder. With the kind of resolution that we now have with the vaginal probe, some of these nodules can be seen. That’s somewhat new, and it’s a function of two things—people looking for endometriosis and the better resolution of more modern equipment.”
Dr. Goldstein believes that MRI is “almost never” indicated in the diagnosis of endometriosis. A more helpful approach would be a consultative ultrasound with someone with more experience. However, when that is not available, or “in areas where you have excellent backup in terms of pelvic MRI, that may be the way to go. I don’t think so,” he demurs, “and some of my colleagues would be very upset at the thought of needing to use MRI to diagnose endometriosis. But in the occasional confusing or difficult case, depending on the quality of the referral pattern you have, it might make sense."
Continue to learn when diagnostic laparoscopy is clearly indicated >>
6. WHEN IS DIAGNOSTIC LAPAROSCOPY CLEARLY INDICATED?
Dr. Giudice believes that laparoscopy—with the intention to treat endometriosis, if present—“is essential when firstline medical therapy fails or when pain is acute and severe.”5
Dr. Stratton concurs. “Any woman with chronic pain wants to know what is causing the pain,” she says. Therefore, “women report a benefit from knowing that their pain is associated with endometriosis.6 However, diagnostic laparoscopy alone, with the sole purpose of determining the presence of endometriosis but not treating the lesions, is no longer performed, as it poses little benefit to the patient other than peace of mind.”
“The general trend in the US has been to first use hormonal treatments when the diagnosis of endometriosis is suspected, prior to performing surgery,” Dr. Stratton says.1 In many cases, by using cyclic combined hormonal contraceptives to reduce menstrual flow or “suppressing menstruation with continuous combined hormonal contraceptives,” gonadotropin-releasing hormone analogues (combined with progestin to prevent bone loss) “or continuous progestin alone may be effective in decreasing pain. Not surprisingly, these hormonal approaches are effective for any chronic pelvic pain, even for women who do not have the surgical diagnosis of endometriosis.”20
“When the firstline approach to chronic pelvic pain is hormonal treatment, laparoscopy is considered when these medical treatments have failed to control the pain or are poorly tolerated, or when the diagnosis of endometriosis is in question,” Dr. Stratton says.
“Laparoscopy to treat endometriomas is indicated if an endometrioma is enlarging or measures more than 4 cm in diameter, or if the diagnosis of an ovarian mass is in question,” she explains. “While surgeons have previously been aggressive in removing endometriomas, this practice may have negative consequences on ovarian function. Because endometriomas are pseudocysts, removing them completely leads to the removal of viable ovarian tissue and may diminish ovarian reserve.”21,22
Continue for the surgical appearance of endometriosis >>
7. WHAT IS THE SURGICAL APPEARANCE OF ENDOMETRIOSIS?
Dr. Giudice returns to the enigmatic nature of endometriosis in addressing this question, mentioning its “many faces” at the time of surgery. “It is imperative that the surgeon recognize the disease in its many forms,” she says. “Also, it is especially helpful at the time of surgery if suspected lesions are biopsied and sent to pathology to have the diagnosis made unequivocally.”5
As for the surgical appearance of endometriosis, Dr. Stratton notes that there are three types of lesions—“superficial lesions, deep infiltrating lesions, and endometriomas. Endometriomas occur almost exclusively in the ovary and are pseudocysts without an identifiable cystic lining. They vary in dimension from a few millimeters to several centimeters.”
“Superficial peritoneal endometriosis lesions have a variable appearance, with some lesions being clear or red; some brown, blue or black; and some having a white appearance, like a scar,” says Dr. Stratton. “Endometriosis can be diagnosed on histologic examination of any of these lesion types."
“Overall, single-color lesions have similar frequencies of biopsy-confirmed endometriosis (59% to 62%),” she says.23 “These lesion appearances likely represent different stages of development of endometriosis, with red or clear lesions occurring first, soon after endometrial tissue implantation; black, blue, or brown lesions occurring later, in response to the hormones varying in the menstrual cycle; and white lesions occurring as the lesions age. Deep infiltrating lesions generally have blue/black or white features.”
“Wide, deep, multiple-color lesions in the cul-de-sac, ovarian fossa, or uterosacral ligaments are most likely endometriosis,” Dr. Stratton adds.23 Only lesions with multiple colors have a significantly higher percentage of positive biopsies (76%). Importantly, more than half of women with only subtle lesions (small red or white lesions) have endometriosis.
You tell the patient that endometriosis is one of the possible diagnoses for her chronic pelvic pain, and you take a focused history. During a pelvic examination, you observe that her right ovary lacks mobility, and you map a number of trigger points for her pain. Transvaginal ultrasound results suggest the presence of nodules in the rectovaginal septum. You begin empiric treatment with continuous combined hormonal contraceptives to suppress menstruation. On her next visit, M.L. reports reduced but still bothersome pain. Laparoscopy reveals a 2-cm endometrioma in the right ovary and deep infiltrating lesions in the cul-de-sac. The endometrioma is resected. Histology confirms the diagnosis of endometriosis.
REFERENCES
1. American College of Obstetricians and Gynecologists. Practice Bulletin #114: Management of endometriosis. Obstet Gynecol. 2010; 116(1):223-236.
2. Sanfilippo JS, Wakim NG, Schikler KN, Yussman MA. Endometriosis in association with uterine anomaly. Am J Obstet Gynecol. 1986; 154(1):39-43.
3. Taylor HS, Bagot C, Kardana A. HOX gene expression is altered in the endometrium of women with endometriosis. Hum Reprod. 1999; 14(5):1328-1331.
4. Berkley KJ, Stratton P. Mechanisms: lessons from translational studies of endometriosis. In: Giamberardino MA, ed. Visceral Pain: Clinical, Pathophysiological and Therapeutic Aspects. Oxford, UK: Oxford University Press; 2009:39-50.
5. Giudice LC. Clinical practice: endometriosis. N Engl J Med. 2010;362(25): 2389-2398.
6. Ballard K, Lowton K, Wright J. What’s the delay: a qualitative study of women’s experiences of reaching a diagnosis of endometriosis. Fertil Steril. 2006;86(5):1296-1301.
7. May KE, Conduit-Hulbert SA, Villar J, et al. Peripheral biomarkers of endometriosis: a systematic review. Hum Reprod Update. 2010; 16(6):651-674.
8. May KE, Villar J, Kirtley S, et al. Endometrial alterations in endometriosis: a systematic review of putative biomarkers. Hum Reprod Update. 2011; 17(5):637-653.
9. Tamaresis JS, Irwin JC, Goldfien GA, et al. Molecular classification of endometriosis and disease stage using high-dimensional genomic data. Endocrinology. 2014;155(12):4986-4999.
10. Sinaii N, Cleary SD, Ballweg ML, et al. High rates of autoimmune and endocrine disorders, fibromyalgia, chronic fatigue syndrome and atopic diseases among women with endometriosis: a survey analysis. Hum Reprod. 2002;17(10):2715-2724.
11. De Graaff AA, D’Hooghe TM, Dunselman GA, et al. The significant effect of endometriosis on physical, mental and social wellbeing: results from an international cross-sectional survey. Hum Reprod. 2013;28(10):2677-2685.
12. Lafay Pillet MC, Huchon C, Santulli P, et al. A clinical score can predict associated deep infiltrating endometriosis before surgery for an endometrioma. Hum Reprod. 2014;29(8):1666-1676.
13. Healey M, Cheng C, Kaur H. To excise or ablate endometriosis? A prospective randomized double-blinded trial after 5-year follow-up. J Minim Invasive Gynecol. 2014;21(6):999-1004.
14. Anaf V, El Nakadi I, De Moor V, et al. Increased nerve density in deep infiltrating endometriotic nodules. Gynecol Obstet Invest. 2011;71(2):112-117.
15. Stratton P, Berkley KJ. Chronic pelvic pain and endometriosis: translational evidence of the relationship and implications. Hum Reprod Update. 2011;17(3):327-346.
16. Karp BI, Sinaii N, Nieman LK, et al. Migraine in women with chronic pelvic pain with and without endometriosis. Fertil Steril. 2011;95(3):895-899.
17. Berkley KJ. A life of pelvic pain. Physiol Behav. 2005;86(3):272-280.
18. Fauconnier A, Chapron C. Endometriosis and pelvic pain: epidemiological evidence of the relationship and implications. Hum Reprod Update. 2005;11(6):595-606.
19. Vercellini P, Fedele L, Aimi G, et al. Association between endometriosis stage, lesion type, patient characteristics and severity of pelvic pain symptoms: a multivariate analysis of over 1000 patients. Hum Reprod. 2007;22(1):266-271.
20. Ling FW. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Pelvic Pain Study Group. Obstet Gynecol. 1999;93(1):51-58.
21. Muzii L, Di Tucci C, Di Feliciantonio M, et al. The effect of surgery for endometrioma on ovarian reserve evaluated by antral follicle count: a systematic review and meta-analysis. Hum Reprod. 2014;29(10):2190-2198.
22. Muzii L, Luciano AA, Zupi E, Panici PB. Effect of surgery for endometrioma on ovarian function: a different point of view. J Minim Invasive Gynecol. 2014;21(4):531-533.
23. Stegmann BJ, Sinaii N, Liu S, et al. Using location, color, size, and depth to characterize and identify endometriosis lesions in a cohort of 133 women. Fertil Steril. 2008;89(6):1632-1636.
IN THIS ARTICLE
• The “why” of endometriosis
• Environmental factors, estrogen, and endometriosis
• Is imaging useful?
• When is diagnostic laparoscopy clearly indicated?
CASE M.L. is a 32-year-old nulliparous woman who is referred to your office by her primary care provider for chronic pelvic pain. She reports severe dysmenorrhea as her main symptom, but she also mentions dyspareunia. She says these symptoms have been present for several years but have increased in intensity gradually. She asks what you consider to be the most likely diagnosis.
What potential diagnoses do you mention to her? And how do you identify the cause of her pain?
Although endometriosis—the presence of endometrial tissue outside the uterus—affects at least 5 million women of reproductive age in the United States alone, it can be a challenging diagnosis for several reasons.
“Endometriosis is a great masquerader,” says Linda Giudice, MD, PhD. “It presents with a variety of pain patterns, intensities, and triggers. It can also involve symptoms that overlap those of other disorders, including disorders of the gastrointestinal and urinary tracts.”
Although endometriosis falls within the differential diagnosis of chronic pelvic pain, “it is usually not high on the list in the primary care setting (adult and adolescent),” adds Dr. Giudice.
John R. Lue, MD, MPH, an author of the most recent practice bulletin on endometriosis from the American College of Obstetricians and Gynecologists,1 sees the situation similarly.
“The main challenge in the diagnosis of endometriosis is that its presentation mimics other causes of chronic pelvic pain,” he says. “Pelvic pain due to endometriosis is usually chronic (lasting ≥ 6 months). It is associated with dysmenorrhea in 50% to 90% of cases, as well as with dyspareunia, deep pelvic pain, and lower abdominal pain with or without back and loin pain. The pain can occur unpredictably and intermittently throughout the menstrual cycle or it can be continuous. In addition, it can be dull, throbbing, or sharp and may be exacerbated by physical activity.2,3 Up to 20% of women with endometriosis have concurrent pain conditions.”4
Among other diseases of the female pelvis that have a relatively similar presentation, Dr. Lue adds, are pathologies of the
• Uterus (adenomyosis, fibroids)
• Fallopian tube (hydrosalpinx)
• Ovaries (ovarian cysts)
• Bladder (interstitial cystitis)
• Bowel (irritable bowel syndrome)
• Musculoskeletal system (piriformis syndrome).
Before pelvic pain is attributed to endometriosis, he says, the provider should rule out bowel, bladder, musculoskeletal, and psychiatric causes.
This article focuses on seven questions, the answers to which are critical to narrowing in on the diagnosis of endometriosis, including essential factors to consider in the patient history, imaging and other diagnostic tools, and considerations in surgical exploration.
1. WHY SUCH A LONG DELAY IN DIAGNOSIS?
Investigators exploring the length of time from a patient’s presentation with symptoms to diagnosis have found it to be particularly long for endometriosis, ranging from six to 11 years.
Because endometriosis is usually not high on the list of differential diagnoses for chronic pelvic pain in the primary care setting, a patient may not be referred to a gynecologist unless those symptoms include severe dysmenorrhea, dyspareunia, or similar findings. Once the referral is made, the gynecologist “will usually try contraceptive steroids, NSAIDs, or second-line progestins before a diagnosis is made,” says Dr. Giudice.5
The delay in diagnosis “is astounding,” she adds, “and has its roots in empiric medical therapies and a combination of patients fearing a diagnosis of cancer and reluctance of gynecologists to perform laparoscopy on adolescents.”6 Another possible cause of diagnostic delay: Some adolescent girls may not realize when their pain is severe. Because they may have always experienced a high degree of pain since menarche, they may assume it to be a normal aspect of womanhood and delay seeking help, says Pamela Stratton, MD.
Continue to learn any biomarkers proved to be useful diagnostic tools >>
2. HAVE ANY BIOMARKERS PROVED TO BE USEFUL DIAGNOSTIC TOOLS?
Any biomarker proven to reliably identify endometriosis would be a boon to medicine, as it would provide a noninvasive or minimally invasive alternative to diagnostic laparoscopy, the current gold standard. Regrettably, the search for such a biomarker has produced “disappointing results,” says Dr. Giudice.
“Recent systematic reviews of all proposed endometriosis-related biomarkers over the past 25 years in serum, plasma, urine, and endometrium could not identify an unequivocally clinically useful biomarker or panel of biomarkers,” she notes.7,8 “This is due mainly to low numbers of subjects, small populations for validations, cycle/hormonal- and disease stage–dependence, poorly defined controls, and low sensitivity and specificity.”
One hopeful development: “Whole genome transcriptomics of archived endometrial tissue and machine learning found several classifiers to diagnose and stage endometriosis with high accuracy that were validated on an independent sample set,” says Dr. Giudice.9 “However, these data now warrant a prospective, multisite study for further validation.”
Continue for key aspects of patient history >>
3. WHAT ASPECTS OF THE PATIENT HISTORY ARE KEY?
Dr. Stratton recommends that clinicians begin their evaluation of the patient with pain by asking her to describe that pain: how long she has had it, when it occurs, and which areas are affected.
“Most women with endometriosis-associated pain have chronic pelvic pain,” Dr. Stratton continues.5 “Up to 90% of those have dysmenorrhea or cyclic pain with menses.”10 In addition, women with endometriosis “commonly report having pain with any bleeding or spotting. About 30% of women diagnosed with endometriosis initially present to their gynecologist with dyspareunia.”11
“Episodic pain with menses may become more constant, lasting for many days of the month,” says Dr. Stratton. “Women with dyschezia or dysuria may have endometriosis lesions associated with the bowel or bladder, respectively.12 When women with these symptoms do not have lesions on the bowel or bladder, these pain symptoms may occur because of higher peritoneal hormone and inflammatory factor levels or because adjacent organs share the neural networks.”
Dr. Giudice views the history similarly: “I believe listening to the patient is essential in evaluating the possibility of her having endometriosis. This involves asking her to describe where her pain is, grading it on a scale of 1 to 10, identifying when in her cycle it occurs, and learning what makes it better or worse.”
“It also is important to assess the quality of the pain,” she adds. “Does it radiate, does it limit her daily activities, does it interfere with her relationships, intercourse, work, school? Is it associated with bowel movements, urination, other pain syndromes?”
“Having a pain questionnaire is a great help so that patients have a chance to reflect on these and other questions that help to frame the pain associated with endometriosis when they come for consultation,” she adds.
By determining if pain is associated with menstruation or spotting, the clinician is better informed about the value of menstrual suppression, says Dr. Stratton. “Determining what makes the pain better or worse can help define triggers which, if treated, can decrease the likelihood of episodes of pain.”
“A detailed history of any medical or surgical treatments and their outcome is helpful in guiding future treatment,” she adds. “While hormonal therapy has been a mainstay of treatment, in some women, some hormonal treatments may worsen pain or have unacceptable adverse effects, such as worsening depression or anxiety. In addition, some pain—especially that associated with deep lesions—may be relieved by surgical treatment;13,14 pain that worsened after surgery may suggest neural damage.”
“As there is an engagement of the central nervous system, endometriosis is considered a central sensitivity syndrome in which women may also have other sites of pain,” Dr. Stratton says. “Thus, obtaining a history about current symptoms or prior diagnosis of irritable bowel syndrome, interstitial cystitis/painful bladder, migraines, fibromyalgia, or chronic fatigue syndrome is beneficial.10,15-17 Facilitating treatment for these comorbidities is a key principle in helping women with endometriosis-associated pain, as any condition that triggers or perpetuates pain warrants treatment.”
Continue for what the physical exam entails >>
4. WHAT SHOULD THE PHYSICAL EXAM ENTAIL?
“An abdominal exam and a pelvic exam are essential in evaluating pain in a woman when endometriosis is suspected,” says Dr. Giudice. “Sometimes the latter is challenging in young teens and can be deferred.” Overall, however, “the pelvic exam can give insight into pain triggers, adnexal masses (possible endometriomas), and mobility of pelvic organs. A rectovaginal exam is important in evaluating deep infiltrating disease and to gauge the pelvic pain landscape overall. In addition, palpating the pelvic floor musculature is important to distinguish pelvic floor muscle spasm from endometriosis pain.”
“The challenge for clinicians is to think beyond the endometrial implants, taking into account multiple factors that influence pain perception,” says Dr. Stratton. During the examination, the clinician should begin by mapping the regions of pain in the abdomen and back, “distinguishing musculoskeletal pain from deep pain. Determining whether pains are focused or diffuse is also important.”
Dr. Stratton recommends that the routine pelvic exam be modified because a standard bimanual exam “confuses pain signals from the pelvic floor, abdominal wall, bladder, and other viscera. For this reason, a pain-oriented assessment is mandatory.”
Begin with a single digital examination to map tender areas, Dr. Stratton advises. Then consider the size, shape, and mobility of reproductive and pelvic organs. “A bimanual exam will help identify adnexal masses like endometriomas,” she says.
Endometriomas usually are not associated with pain, she adds, but “they are associated with deep infiltrating lesions. Nodularity along the uterosacral ligaments, limited reproductive organ mobility, and thickening of the rectovaginal septum also suggest deep infiltrating lesions. Importantly, deep infiltrating lesions are the lesion type most associated with pain.”18,19
Continue to learn if imaging is useful in the diagnosis of endometriosis >>
5. IS IMAGING USEFUL IN THE DIAGNOSIS OF ENDOMETRIOSIS?
Laparoscopy remains the gold standard for diagnosis of endometriosis, observes Steven R. Goldstein, MD. Visualization of endometriotic implants at the time of surgery—with histologic assessment—offers definitive confirmation of the diagnosis. The physical examination, too, can offer a strong suggestion of endometriosis, he says.
“In the past, the pelvic examination and history often were the sine qua non for patients with pain,” Dr. Goldstein says. “Extreme dysmenorrhea and pain between periods, especially with intercourse, defecation, and exercise, all increased the suspicion of endometriosis. People used to talk about feeling nodularity in the uterosacral ligaments and finding decreased mobility of pelvic structures—but I don’t have any question that the skill of today’s gynecologists in doing a bimanual pelvic exam is a fraction of what it was in years gone by, because they haven’t had the necessity of experience. The first thing they do if there’s any question is send the patient for an ultrasound.”
Of course, ultrasound can be especially helpful in identifying endometriomas—sometimes called chocolate cysts—in the ovary. Endometriomas can have a solid appearance on ultrasound, says Dr. Goldstein, because the fluid they contain (dried blood) is sonolucent or pure black on ultrasound, similar to amniotic fluid or the fluid seen in the bladder. “This ‘chocolate’ fluid contained in endometriomas is homogeneous, particulate, and very monotonous in its appearance, in contrast to the internal echoes observed in hemorrhagic corpus lutea, which are very cobweb-like and can sometimes mimic papillary projections,” he adds.
“What’s absolutely essential when imaging a suspected endometrioma by ultrasound is that there be no evidence of any blood flow contained within that structure. Because it’s dried blood, it shouldn’t have any vascularity. If you see blood flow inside what you would call an endometrioma, you need to rethink your diagnosis,” he says.
In some cases, a supposed endometrioma lacks a black, sonolucent appearance, but “the clinician often can tell that it’s a cystic structure by the very bright posterior wall—what we call posterior wall acoustic enhancement—even though the interior of the structure may appear sort of grayish or whitish rather than the pure black of a simple cyst. It’s still fluid-filled,” Dr. Goldstein says.
In some instances, even endometriotic nodules can be imaged by ultrasound, he adds. “There’s an increasing body of literature that suggests that, if you look carefully in people with deep infiltrating endometriosis, you can often see solid-appearing nodules in the rectovaginal septum or between the uterus and bladder. With the kind of resolution that we now have with the vaginal probe, some of these nodules can be seen. That’s somewhat new, and it’s a function of two things—people looking for endometriosis and the better resolution of more modern equipment.”
Dr. Goldstein believes that MRI is “almost never” indicated in the diagnosis of endometriosis. A more helpful approach would be a consultative ultrasound with someone with more experience. However, when that is not available, or “in areas where you have excellent backup in terms of pelvic MRI, that may be the way to go. I don’t think so,” he demurs, “and some of my colleagues would be very upset at the thought of needing to use MRI to diagnose endometriosis. But in the occasional confusing or difficult case, depending on the quality of the referral pattern you have, it might make sense."
Continue to learn when diagnostic laparoscopy is clearly indicated >>
6. WHEN IS DIAGNOSTIC LAPAROSCOPY CLEARLY INDICATED?
Dr. Giudice believes that laparoscopy—with the intention to treat endometriosis, if present—“is essential when firstline medical therapy fails or when pain is acute and severe.”5
Dr. Stratton concurs. “Any woman with chronic pain wants to know what is causing the pain,” she says. Therefore, “women report a benefit from knowing that their pain is associated with endometriosis.6 However, diagnostic laparoscopy alone, with the sole purpose of determining the presence of endometriosis but not treating the lesions, is no longer performed, as it poses little benefit to the patient other than peace of mind.”
“The general trend in the US has been to first use hormonal treatments when the diagnosis of endometriosis is suspected, prior to performing surgery,” Dr. Stratton says.1 In many cases, by using cyclic combined hormonal contraceptives to reduce menstrual flow or “suppressing menstruation with continuous combined hormonal contraceptives,” gonadotropin-releasing hormone analogues (combined with progestin to prevent bone loss) “or continuous progestin alone may be effective in decreasing pain. Not surprisingly, these hormonal approaches are effective for any chronic pelvic pain, even for women who do not have the surgical diagnosis of endometriosis.”20
“When the firstline approach to chronic pelvic pain is hormonal treatment, laparoscopy is considered when these medical treatments have failed to control the pain or are poorly tolerated, or when the diagnosis of endometriosis is in question,” Dr. Stratton says.
“Laparoscopy to treat endometriomas is indicated if an endometrioma is enlarging or measures more than 4 cm in diameter, or if the diagnosis of an ovarian mass is in question,” she explains. “While surgeons have previously been aggressive in removing endometriomas, this practice may have negative consequences on ovarian function. Because endometriomas are pseudocysts, removing them completely leads to the removal of viable ovarian tissue and may diminish ovarian reserve.”21,22
Continue for the surgical appearance of endometriosis >>
7. WHAT IS THE SURGICAL APPEARANCE OF ENDOMETRIOSIS?
Dr. Giudice returns to the enigmatic nature of endometriosis in addressing this question, mentioning its “many faces” at the time of surgery. “It is imperative that the surgeon recognize the disease in its many forms,” she says. “Also, it is especially helpful at the time of surgery if suspected lesions are biopsied and sent to pathology to have the diagnosis made unequivocally.”5
As for the surgical appearance of endometriosis, Dr. Stratton notes that there are three types of lesions—“superficial lesions, deep infiltrating lesions, and endometriomas. Endometriomas occur almost exclusively in the ovary and are pseudocysts without an identifiable cystic lining. They vary in dimension from a few millimeters to several centimeters.”
“Superficial peritoneal endometriosis lesions have a variable appearance, with some lesions being clear or red; some brown, blue or black; and some having a white appearance, like a scar,” says Dr. Stratton. “Endometriosis can be diagnosed on histologic examination of any of these lesion types."
“Overall, single-color lesions have similar frequencies of biopsy-confirmed endometriosis (59% to 62%),” she says.23 “These lesion appearances likely represent different stages of development of endometriosis, with red or clear lesions occurring first, soon after endometrial tissue implantation; black, blue, or brown lesions occurring later, in response to the hormones varying in the menstrual cycle; and white lesions occurring as the lesions age. Deep infiltrating lesions generally have blue/black or white features.”
“Wide, deep, multiple-color lesions in the cul-de-sac, ovarian fossa, or uterosacral ligaments are most likely endometriosis,” Dr. Stratton adds.23 Only lesions with multiple colors have a significantly higher percentage of positive biopsies (76%). Importantly, more than half of women with only subtle lesions (small red or white lesions) have endometriosis.
You tell the patient that endometriosis is one of the possible diagnoses for her chronic pelvic pain, and you take a focused history. During a pelvic examination, you observe that her right ovary lacks mobility, and you map a number of trigger points for her pain. Transvaginal ultrasound results suggest the presence of nodules in the rectovaginal septum. You begin empiric treatment with continuous combined hormonal contraceptives to suppress menstruation. On her next visit, M.L. reports reduced but still bothersome pain. Laparoscopy reveals a 2-cm endometrioma in the right ovary and deep infiltrating lesions in the cul-de-sac. The endometrioma is resected. Histology confirms the diagnosis of endometriosis.
REFERENCES
1. American College of Obstetricians and Gynecologists. Practice Bulletin #114: Management of endometriosis. Obstet Gynecol. 2010; 116(1):223-236.
2. Sanfilippo JS, Wakim NG, Schikler KN, Yussman MA. Endometriosis in association with uterine anomaly. Am J Obstet Gynecol. 1986; 154(1):39-43.
3. Taylor HS, Bagot C, Kardana A. HOX gene expression is altered in the endometrium of women with endometriosis. Hum Reprod. 1999; 14(5):1328-1331.
4. Berkley KJ, Stratton P. Mechanisms: lessons from translational studies of endometriosis. In: Giamberardino MA, ed. Visceral Pain: Clinical, Pathophysiological and Therapeutic Aspects. Oxford, UK: Oxford University Press; 2009:39-50.
5. Giudice LC. Clinical practice: endometriosis. N Engl J Med. 2010;362(25): 2389-2398.
6. Ballard K, Lowton K, Wright J. What’s the delay: a qualitative study of women’s experiences of reaching a diagnosis of endometriosis. Fertil Steril. 2006;86(5):1296-1301.
7. May KE, Conduit-Hulbert SA, Villar J, et al. Peripheral biomarkers of endometriosis: a systematic review. Hum Reprod Update. 2010; 16(6):651-674.
8. May KE, Villar J, Kirtley S, et al. Endometrial alterations in endometriosis: a systematic review of putative biomarkers. Hum Reprod Update. 2011; 17(5):637-653.
9. Tamaresis JS, Irwin JC, Goldfien GA, et al. Molecular classification of endometriosis and disease stage using high-dimensional genomic data. Endocrinology. 2014;155(12):4986-4999.
10. Sinaii N, Cleary SD, Ballweg ML, et al. High rates of autoimmune and endocrine disorders, fibromyalgia, chronic fatigue syndrome and atopic diseases among women with endometriosis: a survey analysis. Hum Reprod. 2002;17(10):2715-2724.
11. De Graaff AA, D’Hooghe TM, Dunselman GA, et al. The significant effect of endometriosis on physical, mental and social wellbeing: results from an international cross-sectional survey. Hum Reprod. 2013;28(10):2677-2685.
12. Lafay Pillet MC, Huchon C, Santulli P, et al. A clinical score can predict associated deep infiltrating endometriosis before surgery for an endometrioma. Hum Reprod. 2014;29(8):1666-1676.
13. Healey M, Cheng C, Kaur H. To excise or ablate endometriosis? A prospective randomized double-blinded trial after 5-year follow-up. J Minim Invasive Gynecol. 2014;21(6):999-1004.
14. Anaf V, El Nakadi I, De Moor V, et al. Increased nerve density in deep infiltrating endometriotic nodules. Gynecol Obstet Invest. 2011;71(2):112-117.
15. Stratton P, Berkley KJ. Chronic pelvic pain and endometriosis: translational evidence of the relationship and implications. Hum Reprod Update. 2011;17(3):327-346.
16. Karp BI, Sinaii N, Nieman LK, et al. Migraine in women with chronic pelvic pain with and without endometriosis. Fertil Steril. 2011;95(3):895-899.
17. Berkley KJ. A life of pelvic pain. Physiol Behav. 2005;86(3):272-280.
18. Fauconnier A, Chapron C. Endometriosis and pelvic pain: epidemiological evidence of the relationship and implications. Hum Reprod Update. 2005;11(6):595-606.
19. Vercellini P, Fedele L, Aimi G, et al. Association between endometriosis stage, lesion type, patient characteristics and severity of pelvic pain symptoms: a multivariate analysis of over 1000 patients. Hum Reprod. 2007;22(1):266-271.
20. Ling FW. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Pelvic Pain Study Group. Obstet Gynecol. 1999;93(1):51-58.
21. Muzii L, Di Tucci C, Di Feliciantonio M, et al. The effect of surgery for endometrioma on ovarian reserve evaluated by antral follicle count: a systematic review and meta-analysis. Hum Reprod. 2014;29(10):2190-2198.
22. Muzii L, Luciano AA, Zupi E, Panici PB. Effect of surgery for endometrioma on ovarian function: a different point of view. J Minim Invasive Gynecol. 2014;21(4):531-533.
23. Stegmann BJ, Sinaii N, Liu S, et al. Using location, color, size, and depth to characterize and identify endometriosis lesions in a cohort of 133 women. Fertil Steril. 2008;89(6):1632-1636.
Manual vacuum aspiration: A safe and effective treatment for early miscarriage
Case Miscarriage in a 29-year-old woman
A woman (G0P0) presents to her gynecologist with amenorrhea for 3 months and a positive home urine pregnancy test. She is 29 years of age. She denies any bleeding or pain and intends to continue the pregnancy, though it was unplanned. Results of office ultrasonography to assess fetal viability reveal an intrauterine gestation with an 8-mm fetal pole but no heartbeat. The diagnosis is miscarriage.
This case illustrates a typical miscarriage diagnosis; most women with miscarriage are asymptomatic and without serious bleeding requiring emergency intervention. The management options include surgical, medical, and expectant. Women should be offered all 3 of these, and clinicians should explain the risks and benefits of each approach. But while each strategy can be safe, effective, and acceptable, many women, as well as their health care providers, will benefit from office-based uterine aspiration. In this article, we present the data available on office-based manual vacuum aspiration (MVA) as well as procedure pointers and urge you to consider MVA in your practice for your patients.
Surgical management
Surgical management of miscarriage offers several clear advantages over medical and expectant management. Perhaps the most important advantage to patients is that surgery offers rapid resolution of miscarriage with the shortest duration of bleeding.1,2 When skilled providers perform electric vacuum aspiration (EVA) or MVA in outpatient or emergency department settings, successful uterine evacuation is completed in a single medical encounter 99% of the time.1 By comparison, several follow-up visits and additional ultrasounds may be required during medical or expectant management. Uterine aspiration rarely requires an operating room (OR). Such a setting should be limited to cases in which the clinical picture reflects:
- hemodynamic instability with active uterine bleeding
- serious uterine infection
- the presence of medical comorbidities in patients who may benefit from additional blood bank and anesthesia resources.
Office-based MVA
Office-based MVA is well tolerated when performed using a combination of verbal distraction and reassurance, oral nonsteroidal anti-inflammatory drugs (NSAIDs), and a paracervical block with or without intravenous sedation.
Evidence on managing pain at MVA. Multiple studies have assessed preprocedure and postprocedure pain using NSAIDs, oral anxiolytics, and local anesthesia at the time of EVA or MVA.3,4 Renner and colleagues found that women who received a paracervical block prior to MVA or EVA reported moderate levels of pain, according to a 100-point visual analogue scale (VAS), at the time of cervical dilation (mean, 42) and uterine aspiration (mean, 63).4 In this same study, patients’ willingness to treat a future pregnancy with EVA or MVA using local anesthesia and their overall satisfaction with the procedure was high (mean, 90 on 100-point VAS).
In-office advantages over the OR. Women and clinicians can avoid the extensive scheduling delays associated with ORs, as well as the complications associated with medical and expectant management, if office-based EVA and MVA services are readily available. Compared with surgical management of miscarriage in an OR, office-based EVA and MVA are faster to complete. For example, Dalton and colleagues compared patients undergoing first-trimester procedures in an office setting with those undergoing a procedure in an OR. The mean procedure time for women treated in an office was 10 minutes, compared with 19 minutes for women treated in the OR. In addition, women treated in an office setting spent a mean total of 97 minutes at the office; women treated in an OR spent a mean total of 290 minutes at the hospital.5
Patients’ satisfaction with care provided in the OR was comparable to patients’ satisfaction with care provided in a medical office. In fact, the median total satisfaction score was high among women who had a procedure in either setting (office score, 19 of 20; OR score, 20 of 20).
Cost and equipment for in-office MVA
Office-based surgical management of miscarriage is more cost-effective than OR-based management. In 2006, Dalton and colleagues conducted a cost analysis and found that average charges for office-based MVA were less than half the cost of charges for a dilation and curettage (D&C) in the OR ($968 vs $1,965, respectively).5
More recently, these researchers found that usual care (expectant or OR management) was more costly than a model that also included medical and office-based surgical options. They found that the expanded care model—with use of the OR only when needed—cost $1,033.29 per case. This was compared with $1,247.58 per case when management options did not include medical and office-based surgical treatments.6
The cost of supplies needed to initiate MVA services within an established outpatient gynecologist’s office is modest. Equipment includes manual vacuum aspirators; disposable cannulae of various sizes; reusable plastic or metal dilators; supplies for disinfection, allowing reuse of MVA aspirators; and supplies for examination of products of conception (POC; FIGURE 1).
According to WomanCare Global, manufacturer of the IPAS MVA Plus, equipment should be sterilized after each use with soap and water, medical cleaning solution (such as Cidex, SPOROX II, etc.), or autoclaving.7 If 2 reusable aspirators are purchased along with dilators, disposable cannulae, and tools for tissue assessment, the price of supplies is estimated at US $500.8 WomanCare Global also offers prepackaged, single-use aspirator kits, which may be ideal for the emergency department setting.9
The procedure
To view a video on the MVA device and procedure, including step-by-step technique (FIGURE 2), local anesthesia administration, choosing cannula size, and cervical dilation, visit the Managing Early Pregnancy Loss Web site (http://www.earlypregnancylossresources.org) and access “Videos.” The video “Uterine aspiration for EPL” is available under password protection and broken into chapters for viewing ease.
The risk of endometritis after surgical management of miscarriage is low. Antibiotic prophylaxis prior to MVA or EVA should be considered. Experts recommend giving a single dose of doxycycline 200 mg orally at least 1 hour prior to uterine aspiration.2,10
Use of EVA or MVA for outpatient management of miscarriage yields the opportunity to conduct immediate gross examination of the evacuated tissue and to verify the presence of complete POC. The process is simple: rinse the specimen through a sieve with water or saline, placed in a clear glass container under a small water bath and backlit on a light box. This allows clinicians to separate uterine decidua and pregnancy tissues. “Floating” tissue in this manner is especially useful in patients with pregnancy of unknown location, as immediate confirmation of a gestational sac rules out ectopic pregnancy.
Examine evacuated tissue for macroscopic evidence of pregnancy. Chorionic villi, which arise from syncytiotrophoblasts, can be seen with the naked eye. Immediate evaluation of POC is also useful for patients who desire diagnostic testing to ascertain a cause of their miscarriage because evacuated tissue stored in saline may be sent to a laboratory for cytogenetic analysis.
Medical management
Management of miscarriage with misoprostol is also safe and acceptable to women, though it has a lower success rate than surgical management.
Comparing efficacy: Medical vs surgical management. The Management of Early Pregnancy Failure Trial (MEPF) is the largest randomized controlled trial comparing medical management of miscarriage to surgical management. This multicenter study compared treatment with office-based EVA or MVA to vaginal misoprostol 800 µg. A repeat dose of vaginal misoprostol was offered 48 hours after the initial dose if a gestational sac was present on ultrasound.
Findings from the MEPF trial revealed a 71% complete uterine evacuation rate after 1 dose of misoprostol and an 84% rate after 2 doses.1 The average (SD) reported pain score documented within 48 hours of treatment with misoprostol or MVA/EVA was moderate (5.7 cm [2.4] on 10-cm VAS). The rate of infection or hospitalization was less than 1% in both treatment groups.
These data should provide patients who are clinically stable and who wish to avoid an invasive procedure reassurance that using medication for the management of miscarriage is a reasonable option.
Misoprostol. Use of misoprostol is associated with a longer median duration of bleeding compared with suction aspiration. After misoprostol, bleeding usually begins after several hours and may continue for weeks.11 Based on 2-week prospective bleeding diary entries from the MEPF trial, women who used misoprostol for management of miscarriage were more likely to have any bleeding during the 2 weeks after initiation of treatment, compared with women who had suction aspiration.12
Clinically significant changes in hemoglobin levels are more common in women treated with misoprostol than in those who choose EVA or MVA; however, these differences rarely require hospitalization or transfusion.1 Women who are considering use of misoprostol should be aware of common adverse effects, including nausea, vomiting, diarrhea, and low-grade temperature.
Medical management of miscarriage requires multiple office visits with repeat ultrasounds or serum beta–human chorionic gonadotropin (β-hCG) levels to confirm treatment success. In cases of medication failure (persistent gestational sac with or without bleeding) or suspected retained POC (endometrial stripe greater than 30 mm measured on ultrasound or persistent vaginal bleeding remote from treatment), women should be prepared for surgical resolution of pregnancy and clinicians should be able to perform an office-based procedure.
Expectant management
Women who choose the “watch and wait” approach should be advised that the process is unpredictable and occasionally requires urgent surgical intervention. Successful resolution of pregnancies that are expectantly managed depends on the type of miscarriage diagnosed at initial presentation. Luise and colleagues conducted a prospective study of 451 women with miscarriage who declined medical and surgical management. They found that the watch-and-wait approach was successful in 91% of women with an incomplete abortion, 76% of women with missed abortion, and 66% of women with anembryonic pregnancies.13 Success was defined by the absence of vaginal bleeding and an anterior-posterior endometrial stripe measuring less than 15 mm 4 weeks after initial diagnosis of miscarriage.
Like medical management for miscarriage, expectant management requires multiple office visits plus repeat ultrasounds or β-hCG measurement trends to confirm treatment success. Women who fail expectant management will require medical or surgical intervention to resolve the pregnancy. For those who are seeking pregnancy right away, the unpredictability and longer time to resolution of miscarriage may render expectant management anxiety provoking and unacceptable.
Etiology: Do true and perceived causes match?
Miscarriage during the first 13 weeks of gestation occurs in at least 10% of all clinically diagnosed pregnancies.10 A recent survey administered by Bardos and colleagues
assessed perceived prevalence and causes of miscarriage in more than 1,000 US men and women.14 The majority of respondents believed miscarriage is uncommon, occurring in less than 5% of pregnancies. Respondents also believed stressful events, lifting heavy objects, and prior use of intrauterine or hormonal contraception are often to blame for pregnancy loss.
Despite more than 3 decades of data confirming that more than 60% of early losses are associated with chromosomal abnormalities and that an additional 18% may be associated with fetal anomalies, women often blame themselves.15 Bardos and colleagues found that 47% of women felt guilty about the experience of miscarriage.
Diagnosis: Updated ultrasonography criteria issued
When miscarriage is suspected based on symptoms of pain and bleeding in preg-
nancy, obtain a thorough history and conduct a limited physical examination. If an intrauterine pregnancy (IUP) was previously identified, a repeat ultrasound can confirm the presence or absence of the gestational sac. If an IUP has not been documented, then additional studies, including serial serum β-hCG examinations and ultrasonography, are essential to rule out ectopic pregnancy. Rh status should be determined and a 50-µg dose of Rh(D)-immune globulin administered to Rh(D)-unsensitized women within 72 hours of documented bleeding.
Ultrasonography is often used to diagnose miscarriage. Many gynecologists use ultrasound criteria based on studies conducted in the early 1990s that define nonviability by an empty gestational sac with mean gestational sac diameter greater than 16 mm or a crown-rump length (CRL) without evidence of fetal cardiac activity greater than 5 mm.10 In 2012, members of the Society of Radiologists in Ultrasound Multispecialty Panel on Early First Trimester Diagnosis of Miscarriage and Exclusion of a Viable Intrauterine Pregnancy developed more conservative criteria for the diagnosis of miscarriage.16
Doubilet and colleagues suggested new cutoffs, based on their reanalysis of 2 large prospective studies conducted in the United Kingdom.17 Calculations for these new cut-offs are based on mathematical adjustments for interobserver variability. Strict adherence to these more conservative criteria is sensible when a pregnancy is desired. For women who do not want to continue the pregnancy there is no medical justification for using this diagnostic process. Indeed, delays can lead to stress and poor outcomes including emergent surgical management for spontaneous and heavy bleeding.
Culture change is needed
Patients’ beliefs and scientific evidence about miscarriage are incongruous. By making simple changes in practice and providing straightforward patient education, ObGyns
can demystify the causes of miscarriage and improve its management. In particular, providing office-based MVA when requested can streamline treatment for many women. For too long, patients have blamed themselves for miscarriage and physicians have relied on D&C in the OR. Changes in culture surrounding miscarriage are
long overdue.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
1. Zhang J, Gilles JM, Barnhart K, Creinin MD, Westhoff C, Frederick MM. A comparison of medical management with misoprostol and surgical management for early pregnancy failure. N Eng J Med. 2005;353(8):761−769.
2. Paul M, Lichtenberg ES, Borgatta L, Grimes DA, Stubblefield PG, Creinin MD, eds. Management of Unintended and Abnormal Pregnancy: Comprehensive Abortion Care. Oxford, United Kingdom: Wiley-Blackwell; 2009.
3. Edelman A, Nichols MD, Jensen J. Comparison of pain and time of procedures with two first-trimester abortion techniques performed by residents and faculty. Am J Obstet Gynecol. 2001;184(7):1564−1567.
4. Renner RM, Nichols MD, Jensen JT, Li H, Edelman AB. Paracervical block for pain control in first-trimester surgical abortion: a randomized controlled trial. Obstet Gynecol. 2012;119(5):1030−1037.
5. Dalton VK, Harris L, Weisman CS, Guire K, Castleman L, Lebovic D. Patient p, satisfaction, and resource use in office evacuation of early pregnancy failure. Obstet Gynecol. 2006;108(1):103−110.
6. Dalton VK, Liang A, Hutton DW, Zochowski MK, Fendrick AM. Beyond usual care: the economic consequences of expanding treatment options in early pregnancy loss. Am J Obstet Gynecol. 2015;212(2):177.e171−177.e176.
7. Ipas. Ipas start-up kit for integrating manual vacuum aspiration (MVA) for early pregnancy loss into women’s reproductive healthcare services. Chapel Hill, NC: Ipas; 2009.
8. MVA Products page. HPSRx Web site. http://www.hpsrx.com/mva-products.html. Accessed October 13, 2015.
9. Kinariwala M, Quinley KE, Datner EM, Schreiber CA. Manual vacuum aspiration in the emergency department for management of early pregnancy failure. Am J Emerg Med. 2013;31(1):244−247.
10. The American College of Obstetricians and Gynecologists. Practice Bulletin No. 150: early pregnancy loss. Obstet Gynecol. 2015;125(5):1258−1267.
11. Meckstroth KR, Whitaker AK, Bertisch S, Goldberg AB, Darney PD. Misoprostol administered by epithelial routes: drug absorption and uterine response. Obstet Gynecol. 2006;108(3 Part 1):582−590.
12. Davis AR, Hendlish SK, Westhoff C, et al. Bleeding patterns after misoprostol vs surgical treatment of early pregnancy failure: results from a randomized trial. Am J Obstet Gynecol. 2007;196(1):31.e31−31.e37.
13. Luise C, Jermy K, May C, Costello G, Collins WP, Bourne TH. Outcome of expectant management of spontaneous first trimester miscarriage: observational study. BMJ. 2002;324(7342):873−875.
14. Bardos J, Hercz D, Friedenthal J, Missmer SA, Williams Z. A national survey on public perceptions of miscarriage. Obstet Gynecol. 2015;125(6):1313−1320.
15. The Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012;98(5):1103−1111.
16. Doubilet PM, Benson CB, Bourne T, Blaivas M. Diagnostic criteria for nonviable pregnancy early in the first trimester. N Eng JMed. 2013;369(15):1443−1451.
17. Abdallah Y, Daemen A, Kirk E, et al. Limitations of current definitions of miscarriage using mean gestational sac diameter and crown–rump length measurements: a multicenter observational study. Ultrasound Obstet Gynecol. 2011;38(5):497−502.
Case Miscarriage in a 29-year-old woman
A woman (G0P0) presents to her gynecologist with amenorrhea for 3 months and a positive home urine pregnancy test. She is 29 years of age. She denies any bleeding or pain and intends to continue the pregnancy, though it was unplanned. Results of office ultrasonography to assess fetal viability reveal an intrauterine gestation with an 8-mm fetal pole but no heartbeat. The diagnosis is miscarriage.
This case illustrates a typical miscarriage diagnosis; most women with miscarriage are asymptomatic and without serious bleeding requiring emergency intervention. The management options include surgical, medical, and expectant. Women should be offered all 3 of these, and clinicians should explain the risks and benefits of each approach. But while each strategy can be safe, effective, and acceptable, many women, as well as their health care providers, will benefit from office-based uterine aspiration. In this article, we present the data available on office-based manual vacuum aspiration (MVA) as well as procedure pointers and urge you to consider MVA in your practice for your patients.
Surgical management
Surgical management of miscarriage offers several clear advantages over medical and expectant management. Perhaps the most important advantage to patients is that surgery offers rapid resolution of miscarriage with the shortest duration of bleeding.1,2 When skilled providers perform electric vacuum aspiration (EVA) or MVA in outpatient or emergency department settings, successful uterine evacuation is completed in a single medical encounter 99% of the time.1 By comparison, several follow-up visits and additional ultrasounds may be required during medical or expectant management. Uterine aspiration rarely requires an operating room (OR). Such a setting should be limited to cases in which the clinical picture reflects:
- hemodynamic instability with active uterine bleeding
- serious uterine infection
- the presence of medical comorbidities in patients who may benefit from additional blood bank and anesthesia resources.
Office-based MVA
Office-based MVA is well tolerated when performed using a combination of verbal distraction and reassurance, oral nonsteroidal anti-inflammatory drugs (NSAIDs), and a paracervical block with or without intravenous sedation.
Evidence on managing pain at MVA. Multiple studies have assessed preprocedure and postprocedure pain using NSAIDs, oral anxiolytics, and local anesthesia at the time of EVA or MVA.3,4 Renner and colleagues found that women who received a paracervical block prior to MVA or EVA reported moderate levels of pain, according to a 100-point visual analogue scale (VAS), at the time of cervical dilation (mean, 42) and uterine aspiration (mean, 63).4 In this same study, patients’ willingness to treat a future pregnancy with EVA or MVA using local anesthesia and their overall satisfaction with the procedure was high (mean, 90 on 100-point VAS).
In-office advantages over the OR. Women and clinicians can avoid the extensive scheduling delays associated with ORs, as well as the complications associated with medical and expectant management, if office-based EVA and MVA services are readily available. Compared with surgical management of miscarriage in an OR, office-based EVA and MVA are faster to complete. For example, Dalton and colleagues compared patients undergoing first-trimester procedures in an office setting with those undergoing a procedure in an OR. The mean procedure time for women treated in an office was 10 minutes, compared with 19 minutes for women treated in the OR. In addition, women treated in an office setting spent a mean total of 97 minutes at the office; women treated in an OR spent a mean total of 290 minutes at the hospital.5
Patients’ satisfaction with care provided in the OR was comparable to patients’ satisfaction with care provided in a medical office. In fact, the median total satisfaction score was high among women who had a procedure in either setting (office score, 19 of 20; OR score, 20 of 20).
Cost and equipment for in-office MVA
Office-based surgical management of miscarriage is more cost-effective than OR-based management. In 2006, Dalton and colleagues conducted a cost analysis and found that average charges for office-based MVA were less than half the cost of charges for a dilation and curettage (D&C) in the OR ($968 vs $1,965, respectively).5
More recently, these researchers found that usual care (expectant or OR management) was more costly than a model that also included medical and office-based surgical options. They found that the expanded care model—with use of the OR only when needed—cost $1,033.29 per case. This was compared with $1,247.58 per case when management options did not include medical and office-based surgical treatments.6
The cost of supplies needed to initiate MVA services within an established outpatient gynecologist’s office is modest. Equipment includes manual vacuum aspirators; disposable cannulae of various sizes; reusable plastic or metal dilators; supplies for disinfection, allowing reuse of MVA aspirators; and supplies for examination of products of conception (POC; FIGURE 1).
According to WomanCare Global, manufacturer of the IPAS MVA Plus, equipment should be sterilized after each use with soap and water, medical cleaning solution (such as Cidex, SPOROX II, etc.), or autoclaving.7 If 2 reusable aspirators are purchased along with dilators, disposable cannulae, and tools for tissue assessment, the price of supplies is estimated at US $500.8 WomanCare Global also offers prepackaged, single-use aspirator kits, which may be ideal for the emergency department setting.9
The procedure
To view a video on the MVA device and procedure, including step-by-step technique (FIGURE 2), local anesthesia administration, choosing cannula size, and cervical dilation, visit the Managing Early Pregnancy Loss Web site (http://www.earlypregnancylossresources.org) and access “Videos.” The video “Uterine aspiration for EPL” is available under password protection and broken into chapters for viewing ease.
The risk of endometritis after surgical management of miscarriage is low. Antibiotic prophylaxis prior to MVA or EVA should be considered. Experts recommend giving a single dose of doxycycline 200 mg orally at least 1 hour prior to uterine aspiration.2,10
Use of EVA or MVA for outpatient management of miscarriage yields the opportunity to conduct immediate gross examination of the evacuated tissue and to verify the presence of complete POC. The process is simple: rinse the specimen through a sieve with water or saline, placed in a clear glass container under a small water bath and backlit on a light box. This allows clinicians to separate uterine decidua and pregnancy tissues. “Floating” tissue in this manner is especially useful in patients with pregnancy of unknown location, as immediate confirmation of a gestational sac rules out ectopic pregnancy.
Examine evacuated tissue for macroscopic evidence of pregnancy. Chorionic villi, which arise from syncytiotrophoblasts, can be seen with the naked eye. Immediate evaluation of POC is also useful for patients who desire diagnostic testing to ascertain a cause of their miscarriage because evacuated tissue stored in saline may be sent to a laboratory for cytogenetic analysis.
Medical management
Management of miscarriage with misoprostol is also safe and acceptable to women, though it has a lower success rate than surgical management.
Comparing efficacy: Medical vs surgical management. The Management of Early Pregnancy Failure Trial (MEPF) is the largest randomized controlled trial comparing medical management of miscarriage to surgical management. This multicenter study compared treatment with office-based EVA or MVA to vaginal misoprostol 800 µg. A repeat dose of vaginal misoprostol was offered 48 hours after the initial dose if a gestational sac was present on ultrasound.
Findings from the MEPF trial revealed a 71% complete uterine evacuation rate after 1 dose of misoprostol and an 84% rate after 2 doses.1 The average (SD) reported pain score documented within 48 hours of treatment with misoprostol or MVA/EVA was moderate (5.7 cm [2.4] on 10-cm VAS). The rate of infection or hospitalization was less than 1% in both treatment groups.
These data should provide patients who are clinically stable and who wish to avoid an invasive procedure reassurance that using medication for the management of miscarriage is a reasonable option.
Misoprostol. Use of misoprostol is associated with a longer median duration of bleeding compared with suction aspiration. After misoprostol, bleeding usually begins after several hours and may continue for weeks.11 Based on 2-week prospective bleeding diary entries from the MEPF trial, women who used misoprostol for management of miscarriage were more likely to have any bleeding during the 2 weeks after initiation of treatment, compared with women who had suction aspiration.12
Clinically significant changes in hemoglobin levels are more common in women treated with misoprostol than in those who choose EVA or MVA; however, these differences rarely require hospitalization or transfusion.1 Women who are considering use of misoprostol should be aware of common adverse effects, including nausea, vomiting, diarrhea, and low-grade temperature.
Medical management of miscarriage requires multiple office visits with repeat ultrasounds or serum beta–human chorionic gonadotropin (β-hCG) levels to confirm treatment success. In cases of medication failure (persistent gestational sac with or without bleeding) or suspected retained POC (endometrial stripe greater than 30 mm measured on ultrasound or persistent vaginal bleeding remote from treatment), women should be prepared for surgical resolution of pregnancy and clinicians should be able to perform an office-based procedure.
Expectant management
Women who choose the “watch and wait” approach should be advised that the process is unpredictable and occasionally requires urgent surgical intervention. Successful resolution of pregnancies that are expectantly managed depends on the type of miscarriage diagnosed at initial presentation. Luise and colleagues conducted a prospective study of 451 women with miscarriage who declined medical and surgical management. They found that the watch-and-wait approach was successful in 91% of women with an incomplete abortion, 76% of women with missed abortion, and 66% of women with anembryonic pregnancies.13 Success was defined by the absence of vaginal bleeding and an anterior-posterior endometrial stripe measuring less than 15 mm 4 weeks after initial diagnosis of miscarriage.
Like medical management for miscarriage, expectant management requires multiple office visits plus repeat ultrasounds or β-hCG measurement trends to confirm treatment success. Women who fail expectant management will require medical or surgical intervention to resolve the pregnancy. For those who are seeking pregnancy right away, the unpredictability and longer time to resolution of miscarriage may render expectant management anxiety provoking and unacceptable.
Etiology: Do true and perceived causes match?
Miscarriage during the first 13 weeks of gestation occurs in at least 10% of all clinically diagnosed pregnancies.10 A recent survey administered by Bardos and colleagues
assessed perceived prevalence and causes of miscarriage in more than 1,000 US men and women.14 The majority of respondents believed miscarriage is uncommon, occurring in less than 5% of pregnancies. Respondents also believed stressful events, lifting heavy objects, and prior use of intrauterine or hormonal contraception are often to blame for pregnancy loss.
Despite more than 3 decades of data confirming that more than 60% of early losses are associated with chromosomal abnormalities and that an additional 18% may be associated with fetal anomalies, women often blame themselves.15 Bardos and colleagues found that 47% of women felt guilty about the experience of miscarriage.
Diagnosis: Updated ultrasonography criteria issued
When miscarriage is suspected based on symptoms of pain and bleeding in preg-
nancy, obtain a thorough history and conduct a limited physical examination. If an intrauterine pregnancy (IUP) was previously identified, a repeat ultrasound can confirm the presence or absence of the gestational sac. If an IUP has not been documented, then additional studies, including serial serum β-hCG examinations and ultrasonography, are essential to rule out ectopic pregnancy. Rh status should be determined and a 50-µg dose of Rh(D)-immune globulin administered to Rh(D)-unsensitized women within 72 hours of documented bleeding.
Ultrasonography is often used to diagnose miscarriage. Many gynecologists use ultrasound criteria based on studies conducted in the early 1990s that define nonviability by an empty gestational sac with mean gestational sac diameter greater than 16 mm or a crown-rump length (CRL) without evidence of fetal cardiac activity greater than 5 mm.10 In 2012, members of the Society of Radiologists in Ultrasound Multispecialty Panel on Early First Trimester Diagnosis of Miscarriage and Exclusion of a Viable Intrauterine Pregnancy developed more conservative criteria for the diagnosis of miscarriage.16
Doubilet and colleagues suggested new cutoffs, based on their reanalysis of 2 large prospective studies conducted in the United Kingdom.17 Calculations for these new cut-offs are based on mathematical adjustments for interobserver variability. Strict adherence to these more conservative criteria is sensible when a pregnancy is desired. For women who do not want to continue the pregnancy there is no medical justification for using this diagnostic process. Indeed, delays can lead to stress and poor outcomes including emergent surgical management for spontaneous and heavy bleeding.
Culture change is needed
Patients’ beliefs and scientific evidence about miscarriage are incongruous. By making simple changes in practice and providing straightforward patient education, ObGyns
can demystify the causes of miscarriage and improve its management. In particular, providing office-based MVA when requested can streamline treatment for many women. For too long, patients have blamed themselves for miscarriage and physicians have relied on D&C in the OR. Changes in culture surrounding miscarriage are
long overdue.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Case Miscarriage in a 29-year-old woman
A woman (G0P0) presents to her gynecologist with amenorrhea for 3 months and a positive home urine pregnancy test. She is 29 years of age. She denies any bleeding or pain and intends to continue the pregnancy, though it was unplanned. Results of office ultrasonography to assess fetal viability reveal an intrauterine gestation with an 8-mm fetal pole but no heartbeat. The diagnosis is miscarriage.
This case illustrates a typical miscarriage diagnosis; most women with miscarriage are asymptomatic and without serious bleeding requiring emergency intervention. The management options include surgical, medical, and expectant. Women should be offered all 3 of these, and clinicians should explain the risks and benefits of each approach. But while each strategy can be safe, effective, and acceptable, many women, as well as their health care providers, will benefit from office-based uterine aspiration. In this article, we present the data available on office-based manual vacuum aspiration (MVA) as well as procedure pointers and urge you to consider MVA in your practice for your patients.
Surgical management
Surgical management of miscarriage offers several clear advantages over medical and expectant management. Perhaps the most important advantage to patients is that surgery offers rapid resolution of miscarriage with the shortest duration of bleeding.1,2 When skilled providers perform electric vacuum aspiration (EVA) or MVA in outpatient or emergency department settings, successful uterine evacuation is completed in a single medical encounter 99% of the time.1 By comparison, several follow-up visits and additional ultrasounds may be required during medical or expectant management. Uterine aspiration rarely requires an operating room (OR). Such a setting should be limited to cases in which the clinical picture reflects:
- hemodynamic instability with active uterine bleeding
- serious uterine infection
- the presence of medical comorbidities in patients who may benefit from additional blood bank and anesthesia resources.
Office-based MVA
Office-based MVA is well tolerated when performed using a combination of verbal distraction and reassurance, oral nonsteroidal anti-inflammatory drugs (NSAIDs), and a paracervical block with or without intravenous sedation.
Evidence on managing pain at MVA. Multiple studies have assessed preprocedure and postprocedure pain using NSAIDs, oral anxiolytics, and local anesthesia at the time of EVA or MVA.3,4 Renner and colleagues found that women who received a paracervical block prior to MVA or EVA reported moderate levels of pain, according to a 100-point visual analogue scale (VAS), at the time of cervical dilation (mean, 42) and uterine aspiration (mean, 63).4 In this same study, patients’ willingness to treat a future pregnancy with EVA or MVA using local anesthesia and their overall satisfaction with the procedure was high (mean, 90 on 100-point VAS).
In-office advantages over the OR. Women and clinicians can avoid the extensive scheduling delays associated with ORs, as well as the complications associated with medical and expectant management, if office-based EVA and MVA services are readily available. Compared with surgical management of miscarriage in an OR, office-based EVA and MVA are faster to complete. For example, Dalton and colleagues compared patients undergoing first-trimester procedures in an office setting with those undergoing a procedure in an OR. The mean procedure time for women treated in an office was 10 minutes, compared with 19 minutes for women treated in the OR. In addition, women treated in an office setting spent a mean total of 97 minutes at the office; women treated in an OR spent a mean total of 290 minutes at the hospital.5
Patients’ satisfaction with care provided in the OR was comparable to patients’ satisfaction with care provided in a medical office. In fact, the median total satisfaction score was high among women who had a procedure in either setting (office score, 19 of 20; OR score, 20 of 20).
Cost and equipment for in-office MVA
Office-based surgical management of miscarriage is more cost-effective than OR-based management. In 2006, Dalton and colleagues conducted a cost analysis and found that average charges for office-based MVA were less than half the cost of charges for a dilation and curettage (D&C) in the OR ($968 vs $1,965, respectively).5
More recently, these researchers found that usual care (expectant or OR management) was more costly than a model that also included medical and office-based surgical options. They found that the expanded care model—with use of the OR only when needed—cost $1,033.29 per case. This was compared with $1,247.58 per case when management options did not include medical and office-based surgical treatments.6
The cost of supplies needed to initiate MVA services within an established outpatient gynecologist’s office is modest. Equipment includes manual vacuum aspirators; disposable cannulae of various sizes; reusable plastic or metal dilators; supplies for disinfection, allowing reuse of MVA aspirators; and supplies for examination of products of conception (POC; FIGURE 1).
According to WomanCare Global, manufacturer of the IPAS MVA Plus, equipment should be sterilized after each use with soap and water, medical cleaning solution (such as Cidex, SPOROX II, etc.), or autoclaving.7 If 2 reusable aspirators are purchased along with dilators, disposable cannulae, and tools for tissue assessment, the price of supplies is estimated at US $500.8 WomanCare Global also offers prepackaged, single-use aspirator kits, which may be ideal for the emergency department setting.9
The procedure
To view a video on the MVA device and procedure, including step-by-step technique (FIGURE 2), local anesthesia administration, choosing cannula size, and cervical dilation, visit the Managing Early Pregnancy Loss Web site (http://www.earlypregnancylossresources.org) and access “Videos.” The video “Uterine aspiration for EPL” is available under password protection and broken into chapters for viewing ease.
The risk of endometritis after surgical management of miscarriage is low. Antibiotic prophylaxis prior to MVA or EVA should be considered. Experts recommend giving a single dose of doxycycline 200 mg orally at least 1 hour prior to uterine aspiration.2,10
Use of EVA or MVA for outpatient management of miscarriage yields the opportunity to conduct immediate gross examination of the evacuated tissue and to verify the presence of complete POC. The process is simple: rinse the specimen through a sieve with water or saline, placed in a clear glass container under a small water bath and backlit on a light box. This allows clinicians to separate uterine decidua and pregnancy tissues. “Floating” tissue in this manner is especially useful in patients with pregnancy of unknown location, as immediate confirmation of a gestational sac rules out ectopic pregnancy.
Examine evacuated tissue for macroscopic evidence of pregnancy. Chorionic villi, which arise from syncytiotrophoblasts, can be seen with the naked eye. Immediate evaluation of POC is also useful for patients who desire diagnostic testing to ascertain a cause of their miscarriage because evacuated tissue stored in saline may be sent to a laboratory for cytogenetic analysis.
Medical management
Management of miscarriage with misoprostol is also safe and acceptable to women, though it has a lower success rate than surgical management.
Comparing efficacy: Medical vs surgical management. The Management of Early Pregnancy Failure Trial (MEPF) is the largest randomized controlled trial comparing medical management of miscarriage to surgical management. This multicenter study compared treatment with office-based EVA or MVA to vaginal misoprostol 800 µg. A repeat dose of vaginal misoprostol was offered 48 hours after the initial dose if a gestational sac was present on ultrasound.
Findings from the MEPF trial revealed a 71% complete uterine evacuation rate after 1 dose of misoprostol and an 84% rate after 2 doses.1 The average (SD) reported pain score documented within 48 hours of treatment with misoprostol or MVA/EVA was moderate (5.7 cm [2.4] on 10-cm VAS). The rate of infection or hospitalization was less than 1% in both treatment groups.
These data should provide patients who are clinically stable and who wish to avoid an invasive procedure reassurance that using medication for the management of miscarriage is a reasonable option.
Misoprostol. Use of misoprostol is associated with a longer median duration of bleeding compared with suction aspiration. After misoprostol, bleeding usually begins after several hours and may continue for weeks.11 Based on 2-week prospective bleeding diary entries from the MEPF trial, women who used misoprostol for management of miscarriage were more likely to have any bleeding during the 2 weeks after initiation of treatment, compared with women who had suction aspiration.12
Clinically significant changes in hemoglobin levels are more common in women treated with misoprostol than in those who choose EVA or MVA; however, these differences rarely require hospitalization or transfusion.1 Women who are considering use of misoprostol should be aware of common adverse effects, including nausea, vomiting, diarrhea, and low-grade temperature.
Medical management of miscarriage requires multiple office visits with repeat ultrasounds or serum beta–human chorionic gonadotropin (β-hCG) levels to confirm treatment success. In cases of medication failure (persistent gestational sac with or without bleeding) or suspected retained POC (endometrial stripe greater than 30 mm measured on ultrasound or persistent vaginal bleeding remote from treatment), women should be prepared for surgical resolution of pregnancy and clinicians should be able to perform an office-based procedure.
Expectant management
Women who choose the “watch and wait” approach should be advised that the process is unpredictable and occasionally requires urgent surgical intervention. Successful resolution of pregnancies that are expectantly managed depends on the type of miscarriage diagnosed at initial presentation. Luise and colleagues conducted a prospective study of 451 women with miscarriage who declined medical and surgical management. They found that the watch-and-wait approach was successful in 91% of women with an incomplete abortion, 76% of women with missed abortion, and 66% of women with anembryonic pregnancies.13 Success was defined by the absence of vaginal bleeding and an anterior-posterior endometrial stripe measuring less than 15 mm 4 weeks after initial diagnosis of miscarriage.
Like medical management for miscarriage, expectant management requires multiple office visits plus repeat ultrasounds or β-hCG measurement trends to confirm treatment success. Women who fail expectant management will require medical or surgical intervention to resolve the pregnancy. For those who are seeking pregnancy right away, the unpredictability and longer time to resolution of miscarriage may render expectant management anxiety provoking and unacceptable.
Etiology: Do true and perceived causes match?
Miscarriage during the first 13 weeks of gestation occurs in at least 10% of all clinically diagnosed pregnancies.10 A recent survey administered by Bardos and colleagues
assessed perceived prevalence and causes of miscarriage in more than 1,000 US men and women.14 The majority of respondents believed miscarriage is uncommon, occurring in less than 5% of pregnancies. Respondents also believed stressful events, lifting heavy objects, and prior use of intrauterine or hormonal contraception are often to blame for pregnancy loss.
Despite more than 3 decades of data confirming that more than 60% of early losses are associated with chromosomal abnormalities and that an additional 18% may be associated with fetal anomalies, women often blame themselves.15 Bardos and colleagues found that 47% of women felt guilty about the experience of miscarriage.
Diagnosis: Updated ultrasonography criteria issued
When miscarriage is suspected based on symptoms of pain and bleeding in preg-
nancy, obtain a thorough history and conduct a limited physical examination. If an intrauterine pregnancy (IUP) was previously identified, a repeat ultrasound can confirm the presence or absence of the gestational sac. If an IUP has not been documented, then additional studies, including serial serum β-hCG examinations and ultrasonography, are essential to rule out ectopic pregnancy. Rh status should be determined and a 50-µg dose of Rh(D)-immune globulin administered to Rh(D)-unsensitized women within 72 hours of documented bleeding.
Ultrasonography is often used to diagnose miscarriage. Many gynecologists use ultrasound criteria based on studies conducted in the early 1990s that define nonviability by an empty gestational sac with mean gestational sac diameter greater than 16 mm or a crown-rump length (CRL) without evidence of fetal cardiac activity greater than 5 mm.10 In 2012, members of the Society of Radiologists in Ultrasound Multispecialty Panel on Early First Trimester Diagnosis of Miscarriage and Exclusion of a Viable Intrauterine Pregnancy developed more conservative criteria for the diagnosis of miscarriage.16
Doubilet and colleagues suggested new cutoffs, based on their reanalysis of 2 large prospective studies conducted in the United Kingdom.17 Calculations for these new cut-offs are based on mathematical adjustments for interobserver variability. Strict adherence to these more conservative criteria is sensible when a pregnancy is desired. For women who do not want to continue the pregnancy there is no medical justification for using this diagnostic process. Indeed, delays can lead to stress and poor outcomes including emergent surgical management for spontaneous and heavy bleeding.
Culture change is needed
Patients’ beliefs and scientific evidence about miscarriage are incongruous. By making simple changes in practice and providing straightforward patient education, ObGyns
can demystify the causes of miscarriage and improve its management. In particular, providing office-based MVA when requested can streamline treatment for many women. For too long, patients have blamed themselves for miscarriage and physicians have relied on D&C in the OR. Changes in culture surrounding miscarriage are
long overdue.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
1. Zhang J, Gilles JM, Barnhart K, Creinin MD, Westhoff C, Frederick MM. A comparison of medical management with misoprostol and surgical management for early pregnancy failure. N Eng J Med. 2005;353(8):761−769.
2. Paul M, Lichtenberg ES, Borgatta L, Grimes DA, Stubblefield PG, Creinin MD, eds. Management of Unintended and Abnormal Pregnancy: Comprehensive Abortion Care. Oxford, United Kingdom: Wiley-Blackwell; 2009.
3. Edelman A, Nichols MD, Jensen J. Comparison of pain and time of procedures with two first-trimester abortion techniques performed by residents and faculty. Am J Obstet Gynecol. 2001;184(7):1564−1567.
4. Renner RM, Nichols MD, Jensen JT, Li H, Edelman AB. Paracervical block for pain control in first-trimester surgical abortion: a randomized controlled trial. Obstet Gynecol. 2012;119(5):1030−1037.
5. Dalton VK, Harris L, Weisman CS, Guire K, Castleman L, Lebovic D. Patient p, satisfaction, and resource use in office evacuation of early pregnancy failure. Obstet Gynecol. 2006;108(1):103−110.
6. Dalton VK, Liang A, Hutton DW, Zochowski MK, Fendrick AM. Beyond usual care: the economic consequences of expanding treatment options in early pregnancy loss. Am J Obstet Gynecol. 2015;212(2):177.e171−177.e176.
7. Ipas. Ipas start-up kit for integrating manual vacuum aspiration (MVA) for early pregnancy loss into women’s reproductive healthcare services. Chapel Hill, NC: Ipas; 2009.
8. MVA Products page. HPSRx Web site. http://www.hpsrx.com/mva-products.html. Accessed October 13, 2015.
9. Kinariwala M, Quinley KE, Datner EM, Schreiber CA. Manual vacuum aspiration in the emergency department for management of early pregnancy failure. Am J Emerg Med. 2013;31(1):244−247.
10. The American College of Obstetricians and Gynecologists. Practice Bulletin No. 150: early pregnancy loss. Obstet Gynecol. 2015;125(5):1258−1267.
11. Meckstroth KR, Whitaker AK, Bertisch S, Goldberg AB, Darney PD. Misoprostol administered by epithelial routes: drug absorption and uterine response. Obstet Gynecol. 2006;108(3 Part 1):582−590.
12. Davis AR, Hendlish SK, Westhoff C, et al. Bleeding patterns after misoprostol vs surgical treatment of early pregnancy failure: results from a randomized trial. Am J Obstet Gynecol. 2007;196(1):31.e31−31.e37.
13. Luise C, Jermy K, May C, Costello G, Collins WP, Bourne TH. Outcome of expectant management of spontaneous first trimester miscarriage: observational study. BMJ. 2002;324(7342):873−875.
14. Bardos J, Hercz D, Friedenthal J, Missmer SA, Williams Z. A national survey on public perceptions of miscarriage. Obstet Gynecol. 2015;125(6):1313−1320.
15. The Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012;98(5):1103−1111.
16. Doubilet PM, Benson CB, Bourne T, Blaivas M. Diagnostic criteria for nonviable pregnancy early in the first trimester. N Eng JMed. 2013;369(15):1443−1451.
17. Abdallah Y, Daemen A, Kirk E, et al. Limitations of current definitions of miscarriage using mean gestational sac diameter and crown–rump length measurements: a multicenter observational study. Ultrasound Obstet Gynecol. 2011;38(5):497−502.
1. Zhang J, Gilles JM, Barnhart K, Creinin MD, Westhoff C, Frederick MM. A comparison of medical management with misoprostol and surgical management for early pregnancy failure. N Eng J Med. 2005;353(8):761−769.
2. Paul M, Lichtenberg ES, Borgatta L, Grimes DA, Stubblefield PG, Creinin MD, eds. Management of Unintended and Abnormal Pregnancy: Comprehensive Abortion Care. Oxford, United Kingdom: Wiley-Blackwell; 2009.
3. Edelman A, Nichols MD, Jensen J. Comparison of pain and time of procedures with two first-trimester abortion techniques performed by residents and faculty. Am J Obstet Gynecol. 2001;184(7):1564−1567.
4. Renner RM, Nichols MD, Jensen JT, Li H, Edelman AB. Paracervical block for pain control in first-trimester surgical abortion: a randomized controlled trial. Obstet Gynecol. 2012;119(5):1030−1037.
5. Dalton VK, Harris L, Weisman CS, Guire K, Castleman L, Lebovic D. Patient p, satisfaction, and resource use in office evacuation of early pregnancy failure. Obstet Gynecol. 2006;108(1):103−110.
6. Dalton VK, Liang A, Hutton DW, Zochowski MK, Fendrick AM. Beyond usual care: the economic consequences of expanding treatment options in early pregnancy loss. Am J Obstet Gynecol. 2015;212(2):177.e171−177.e176.
7. Ipas. Ipas start-up kit for integrating manual vacuum aspiration (MVA) for early pregnancy loss into women’s reproductive healthcare services. Chapel Hill, NC: Ipas; 2009.
8. MVA Products page. HPSRx Web site. http://www.hpsrx.com/mva-products.html. Accessed October 13, 2015.
9. Kinariwala M, Quinley KE, Datner EM, Schreiber CA. Manual vacuum aspiration in the emergency department for management of early pregnancy failure. Am J Emerg Med. 2013;31(1):244−247.
10. The American College of Obstetricians and Gynecologists. Practice Bulletin No. 150: early pregnancy loss. Obstet Gynecol. 2015;125(5):1258−1267.
11. Meckstroth KR, Whitaker AK, Bertisch S, Goldberg AB, Darney PD. Misoprostol administered by epithelial routes: drug absorption and uterine response. Obstet Gynecol. 2006;108(3 Part 1):582−590.
12. Davis AR, Hendlish SK, Westhoff C, et al. Bleeding patterns after misoprostol vs surgical treatment of early pregnancy failure: results from a randomized trial. Am J Obstet Gynecol. 2007;196(1):31.e31−31.e37.
13. Luise C, Jermy K, May C, Costello G, Collins WP, Bourne TH. Outcome of expectant management of spontaneous first trimester miscarriage: observational study. BMJ. 2002;324(7342):873−875.
14. Bardos J, Hercz D, Friedenthal J, Missmer SA, Williams Z. A national survey on public perceptions of miscarriage. Obstet Gynecol. 2015;125(6):1313−1320.
15. The Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012;98(5):1103−1111.
16. Doubilet PM, Benson CB, Bourne T, Blaivas M. Diagnostic criteria for nonviable pregnancy early in the first trimester. N Eng JMed. 2013;369(15):1443−1451.
17. Abdallah Y, Daemen A, Kirk E, et al. Limitations of current definitions of miscarriage using mean gestational sac diameter and crown–rump length measurements: a multicenter observational study. Ultrasound Obstet Gynecol. 2011;38(5):497−502.
In this Article
- Advantages of office-based manual vacuum aspiration
- Medical management
- Current diagnostic criteria
When is cell-free DNA best used as a primary screen?
Cell-free DNA screening, or so-called noninvasive prenatal testing (NIPT), has had greatly increased utilization recently as advances in technology have elevated it almost to the level of a diagnostic test for detection of certain aneuploidies. Although it is still considered a screening test, recent interest has arisen regarding population screening and whether or not this test should be universally used as first-line or whether it should still be restricted to specific high-risk populations.
In the current study, Kaimal and colleagues attempted to determine the best strategy for utilization of NIPT using a decision-analytic model. For their study many assumptions had to be made in order to allow for calculation of detection rates and for determination of cost and quality-adjusted life years. (The model followed a theoretical cohort of women desiring prenatal testing [screening or diagnostic or both] from the time of their initial test through the end of their pregnancy, the birth of their neonate, and the remainder of their own life expectancy.)
The conclusion of the authors is that traditional multiple marker screening remains the optimal choice for most women (those aged 20 to 38 years) but that NIPT becomes the optimal strategy at age 38. The goal of this study was not just to determine cost-effectiveness but also to attempt to devise a strategy that would optimize detection of aneuploidy and minimize the need for the performance of diagnostic procedures.
Data not available in this study included such things as population differences with respect to the acceptance of pregnancy termination as an option, and the potential utility of first-trimester ultrasound screening for structural defects that might not be accounted for with NIPT (such as thickened nuchal translucency, altered cardiac axis, cranial defects, and abdominal wall defects).
What this evidence means for practice
For now, the best approach would be to adhere to current recommendations as outlined in the 2015 American College of Obstetricians and Gynecologists (ACOG) Committee Opinion.1 Summarized, these are:
- Do not utilize NIPT in low-risk populations (although this opinion also suggests that patients may opt to have this test performed regardless of risk status with the understanding that detection rates are lower in low-risk populations and insurance coverage may be different).
- Offer NIPT to high-risk women as a first-line screen, as is suggested in the current study with respect to maternal age criteria (ACOG uses an age cut-off of 35 years, not 38).
- Utilize NIPT as a follow-up test after conventional testing suggests increased risk status in those patients wishing to avoid invasive diagnostic testing.
- The ACOG position remains that all women, regardless of age, who desire the most comprehensive information available regarding fetal chromosomal abnormalities should be offered diagnostic testing (chorionic villus sampling and amniocentesis).
Also, as mentioned in the committee opinion, this technology is evolving rapidly and all practitioners should closely follow this evolution with respect to changing efficacy and changing cost.
—John T. Repke, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Reference
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640. Cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.
Cell-free DNA screening, or so-called noninvasive prenatal testing (NIPT), has had greatly increased utilization recently as advances in technology have elevated it almost to the level of a diagnostic test for detection of certain aneuploidies. Although it is still considered a screening test, recent interest has arisen regarding population screening and whether or not this test should be universally used as first-line or whether it should still be restricted to specific high-risk populations.
In the current study, Kaimal and colleagues attempted to determine the best strategy for utilization of NIPT using a decision-analytic model. For their study many assumptions had to be made in order to allow for calculation of detection rates and for determination of cost and quality-adjusted life years. (The model followed a theoretical cohort of women desiring prenatal testing [screening or diagnostic or both] from the time of their initial test through the end of their pregnancy, the birth of their neonate, and the remainder of their own life expectancy.)
The conclusion of the authors is that traditional multiple marker screening remains the optimal choice for most women (those aged 20 to 38 years) but that NIPT becomes the optimal strategy at age 38. The goal of this study was not just to determine cost-effectiveness but also to attempt to devise a strategy that would optimize detection of aneuploidy and minimize the need for the performance of diagnostic procedures.
Data not available in this study included such things as population differences with respect to the acceptance of pregnancy termination as an option, and the potential utility of first-trimester ultrasound screening for structural defects that might not be accounted for with NIPT (such as thickened nuchal translucency, altered cardiac axis, cranial defects, and abdominal wall defects).
What this evidence means for practice
For now, the best approach would be to adhere to current recommendations as outlined in the 2015 American College of Obstetricians and Gynecologists (ACOG) Committee Opinion.1 Summarized, these are:
- Do not utilize NIPT in low-risk populations (although this opinion also suggests that patients may opt to have this test performed regardless of risk status with the understanding that detection rates are lower in low-risk populations and insurance coverage may be different).
- Offer NIPT to high-risk women as a first-line screen, as is suggested in the current study with respect to maternal age criteria (ACOG uses an age cut-off of 35 years, not 38).
- Utilize NIPT as a follow-up test after conventional testing suggests increased risk status in those patients wishing to avoid invasive diagnostic testing.
- The ACOG position remains that all women, regardless of age, who desire the most comprehensive information available regarding fetal chromosomal abnormalities should be offered diagnostic testing (chorionic villus sampling and amniocentesis).
Also, as mentioned in the committee opinion, this technology is evolving rapidly and all practitioners should closely follow this evolution with respect to changing efficacy and changing cost.
—John T. Repke, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Cell-free DNA screening, or so-called noninvasive prenatal testing (NIPT), has had greatly increased utilization recently as advances in technology have elevated it almost to the level of a diagnostic test for detection of certain aneuploidies. Although it is still considered a screening test, recent interest has arisen regarding population screening and whether or not this test should be universally used as first-line or whether it should still be restricted to specific high-risk populations.
In the current study, Kaimal and colleagues attempted to determine the best strategy for utilization of NIPT using a decision-analytic model. For their study many assumptions had to be made in order to allow for calculation of detection rates and for determination of cost and quality-adjusted life years. (The model followed a theoretical cohort of women desiring prenatal testing [screening or diagnostic or both] from the time of their initial test through the end of their pregnancy, the birth of their neonate, and the remainder of their own life expectancy.)
The conclusion of the authors is that traditional multiple marker screening remains the optimal choice for most women (those aged 20 to 38 years) but that NIPT becomes the optimal strategy at age 38. The goal of this study was not just to determine cost-effectiveness but also to attempt to devise a strategy that would optimize detection of aneuploidy and minimize the need for the performance of diagnostic procedures.
Data not available in this study included such things as population differences with respect to the acceptance of pregnancy termination as an option, and the potential utility of first-trimester ultrasound screening for structural defects that might not be accounted for with NIPT (such as thickened nuchal translucency, altered cardiac axis, cranial defects, and abdominal wall defects).
What this evidence means for practice
For now, the best approach would be to adhere to current recommendations as outlined in the 2015 American College of Obstetricians and Gynecologists (ACOG) Committee Opinion.1 Summarized, these are:
- Do not utilize NIPT in low-risk populations (although this opinion also suggests that patients may opt to have this test performed regardless of risk status with the understanding that detection rates are lower in low-risk populations and insurance coverage may be different).
- Offer NIPT to high-risk women as a first-line screen, as is suggested in the current study with respect to maternal age criteria (ACOG uses an age cut-off of 35 years, not 38).
- Utilize NIPT as a follow-up test after conventional testing suggests increased risk status in those patients wishing to avoid invasive diagnostic testing.
- The ACOG position remains that all women, regardless of age, who desire the most comprehensive information available regarding fetal chromosomal abnormalities should be offered diagnostic testing (chorionic villus sampling and amniocentesis).
Also, as mentioned in the committee opinion, this technology is evolving rapidly and all practitioners should closely follow this evolution with respect to changing efficacy and changing cost.
—John T. Repke, MD
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Reference
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640. Cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.
Reference
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640. Cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.