User login
Signs of chorioamnionitis ignored? $3.5M settlement
Signs of chorioamnionitis ignored? $3.5M settlement
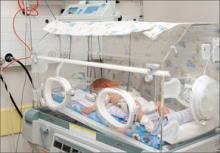
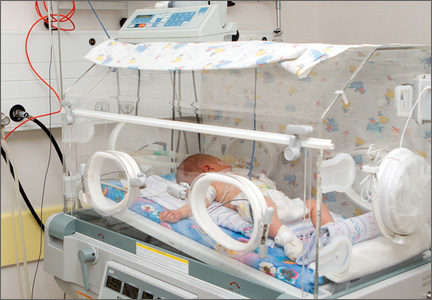
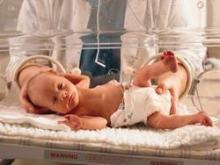
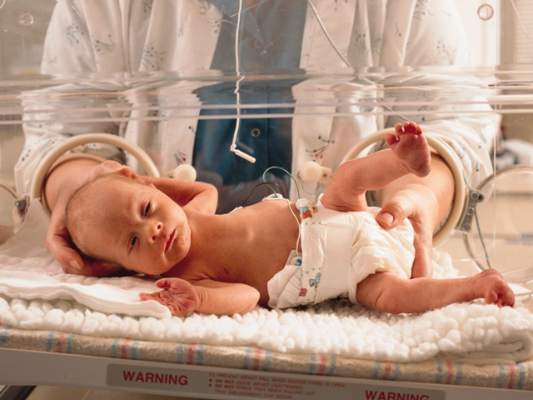
At 31 weeks’ gestation, a mother at risk for preterm labor was admitted to the hospital for 2 days. Examination and test results showed evidence of infection. She was given antenatal corticosteroids for fetal lung development in case of premature delivery. At discharge, bed rest was ordered and she complied. At 32 weeks’ gestation, she returned to the hospital with worsening symptoms, was prescribed antibiotics to treat a urinary tract infection, and was discharged. She went to the hospital a third time at almost 33 weeks’ gestation, experiencing contractions and leaking fluid. She was admitted with a plan to deliver the baby if any signs or symptoms of intra-amniotic infection (clinical chorioamnionitis) were present. Four days later, a cesarean delivery was ordered due to fetal tachycardia and decreased fetal heart rate. Imaging results performed in the neonatal intensive care unit showed that the baby received a brain injury. The child has physical and mental impairments including cerebral palsy, cortical blindness, and epilepsy.
Parents’ claim Hospital health care providers failed to communicate with each other or to obtain records from prior admissions, although the mother told them that she had been to the hospital twice within the past 2 weeks. Medical records from all 3 admissions showed clear signs and symptoms of a vaginal/cervical infection that had progressed to clinical chorioamnionitis 2 days before delivery. Examination of the placenta by a pathologist confirmed that the infection had spread to the umbilical cord, injuring the child.
Defendant’s defense The standard of care was met. There was no indication that an earlier delivery was needed.
Verdict A $3.5 million Michigan settlement was reached by the hospital during the trial.
Surgical approach questioned
A woman went to her ObGyn for tubal ligation and ventral hernia repair. The patient was concerned about infection and scarring. She agreed to a laparoscopic procedure, knowing that the procedure might have to be altered to laparotomy.
Patient’s claim The patient consented to laparoscopic surgery. However, surgery did not begin as laparoscopy but as an open procedure. The patient has a 6-inch scar on her abdomen. She accused both the ObGyn and the hospital of lack of informed consent for laparotomy.
Defendant’s defense The hospital claimed that their nurses’ role was to read the consent form signed by the patient in the ObGyn’s office. The ObGyn claimed that the patient signed a general consent form that permitted him to do what was reasonable. He had determined after surgery began that a laparoscopic procedure would have been more dangerous.
Verdict A $150,000 Louisiana verdict was returned against the ObGyn; the hospital was acquitted.
Macrosomic baby and mother both injured during delivery
Delivery of a mother’s fourth child was managed by a hospital-employed family physician (FP). Shoulder dystocia was encountered, and the FP made a 4th-degree extension of the episiotomy. The baby weighed 10 lb 14 oz at birth. The mother has fecal and urinary incontinence and pain as a result of the large episiotomy. The child has a right-sided brachial plexus injury.
Parents’ claim Failure to perform cesarean delivery caused injury to the mother and child. The FP should have recognized from the mother’s history of delivering 3 macrosomic babies and the progress of this pregnancy, that the baby was large.
Defendant’s defense The case was settled during trial.
Verdict A $1.5 million Minnesota settlement was reached that included $1.2 million for the child and $300,000 for the mother.
Surgical table folds during hysterectomy: $5.3M verdict
While a woman was undergoing a hysterectomy, the surgical table she was lying on folded up into a “U” position, causing the inserted speculum to tear the patient from vagina to rectum. The fall also caused a back injury usually attributed to falls from great distances. The patient has permanent pain, recurring diarrhea, and depression as a result of the injuries.
Patient’s claim The injuries occurred because of the defendants’ failure to read, understand, and follow the warning labels on the surgical table.
Defendant’s defense The case was settled before trial.
Verdict A $5.3 million settlement was reached with the hospital.
Signs of chorioamnionitis ignored? $3.5M settlement
At 31 weeks’ gestation, a mother at risk for preterm labor was admitted to the hospital for 2 days. Examination and test results showed evidence of infection. She was given antenatal corticosteroids for fetal lung development in case of premature delivery. At discharge, bed rest was ordered and she complied. At 32 weeks’ gestation, she returned to the hospital with worsening symptoms, was prescribed antibiotics to treat a urinary tract infection, and was discharged. She went to the hospital a third time at almost 33 weeks’ gestation, experiencing contractions and leaking fluid. She was admitted with a plan to deliver the baby if any signs or symptoms of intra-amniotic infection (clinical chorioamnionitis) were present. Four days later, a cesarean delivery was ordered due to fetal tachycardia and decreased fetal heart rate. Imaging results performed in the neonatal intensive care unit showed that the baby received a brain injury. The child has physical and mental impairments including cerebral palsy, cortical blindness, and epilepsy.
Parents’ claim Hospital health care providers failed to communicate with each other or to obtain records from prior admissions, although the mother told them that she had been to the hospital twice within the past 2 weeks. Medical records from all 3 admissions showed clear signs and symptoms of a vaginal/cervical infection that had progressed to clinical chorioamnionitis 2 days before delivery. Examination of the placenta by a pathologist confirmed that the infection had spread to the umbilical cord, injuring the child.
Defendant’s defense The standard of care was met. There was no indication that an earlier delivery was needed.
Verdict A $3.5 million Michigan settlement was reached by the hospital during the trial.
Surgical approach questioned
A woman went to her ObGyn for tubal ligation and ventral hernia repair. The patient was concerned about infection and scarring. She agreed to a laparoscopic procedure, knowing that the procedure might have to be altered to laparotomy.
Patient’s claim The patient consented to laparoscopic surgery. However, surgery did not begin as laparoscopy but as an open procedure. The patient has a 6-inch scar on her abdomen. She accused both the ObGyn and the hospital of lack of informed consent for laparotomy.
Defendant’s defense The hospital claimed that their nurses’ role was to read the consent form signed by the patient in the ObGyn’s office. The ObGyn claimed that the patient signed a general consent form that permitted him to do what was reasonable. He had determined after surgery began that a laparoscopic procedure would have been more dangerous.
Verdict A $150,000 Louisiana verdict was returned against the ObGyn; the hospital was acquitted.
Macrosomic baby and mother both injured during delivery
Delivery of a mother’s fourth child was managed by a hospital-employed family physician (FP). Shoulder dystocia was encountered, and the FP made a 4th-degree extension of the episiotomy. The baby weighed 10 lb 14 oz at birth. The mother has fecal and urinary incontinence and pain as a result of the large episiotomy. The child has a right-sided brachial plexus injury.
Parents’ claim Failure to perform cesarean delivery caused injury to the mother and child. The FP should have recognized from the mother’s history of delivering 3 macrosomic babies and the progress of this pregnancy, that the baby was large.
Defendant’s defense The case was settled during trial.
Verdict A $1.5 million Minnesota settlement was reached that included $1.2 million for the child and $300,000 for the mother.
Surgical table folds during hysterectomy: $5.3M verdict
While a woman was undergoing a hysterectomy, the surgical table she was lying on folded up into a “U” position, causing the inserted speculum to tear the patient from vagina to rectum. The fall also caused a back injury usually attributed to falls from great distances. The patient has permanent pain, recurring diarrhea, and depression as a result of the injuries.
Patient’s claim The injuries occurred because of the defendants’ failure to read, understand, and follow the warning labels on the surgical table.
Defendant’s defense The case was settled before trial.
Verdict A $5.3 million settlement was reached with the hospital.
Signs of chorioamnionitis ignored? $3.5M settlement
At 31 weeks’ gestation, a mother at risk for preterm labor was admitted to the hospital for 2 days. Examination and test results showed evidence of infection. She was given antenatal corticosteroids for fetal lung development in case of premature delivery. At discharge, bed rest was ordered and she complied. At 32 weeks’ gestation, she returned to the hospital with worsening symptoms, was prescribed antibiotics to treat a urinary tract infection, and was discharged. She went to the hospital a third time at almost 33 weeks’ gestation, experiencing contractions and leaking fluid. She was admitted with a plan to deliver the baby if any signs or symptoms of intra-amniotic infection (clinical chorioamnionitis) were present. Four days later, a cesarean delivery was ordered due to fetal tachycardia and decreased fetal heart rate. Imaging results performed in the neonatal intensive care unit showed that the baby received a brain injury. The child has physical and mental impairments including cerebral palsy, cortical blindness, and epilepsy.
Parents’ claim Hospital health care providers failed to communicate with each other or to obtain records from prior admissions, although the mother told them that she had been to the hospital twice within the past 2 weeks. Medical records from all 3 admissions showed clear signs and symptoms of a vaginal/cervical infection that had progressed to clinical chorioamnionitis 2 days before delivery. Examination of the placenta by a pathologist confirmed that the infection had spread to the umbilical cord, injuring the child.
Defendant’s defense The standard of care was met. There was no indication that an earlier delivery was needed.
Verdict A $3.5 million Michigan settlement was reached by the hospital during the trial.
Surgical approach questioned
A woman went to her ObGyn for tubal ligation and ventral hernia repair. The patient was concerned about infection and scarring. She agreed to a laparoscopic procedure, knowing that the procedure might have to be altered to laparotomy.
Patient’s claim The patient consented to laparoscopic surgery. However, surgery did not begin as laparoscopy but as an open procedure. The patient has a 6-inch scar on her abdomen. She accused both the ObGyn and the hospital of lack of informed consent for laparotomy.
Defendant’s defense The hospital claimed that their nurses’ role was to read the consent form signed by the patient in the ObGyn’s office. The ObGyn claimed that the patient signed a general consent form that permitted him to do what was reasonable. He had determined after surgery began that a laparoscopic procedure would have been more dangerous.
Verdict A $150,000 Louisiana verdict was returned against the ObGyn; the hospital was acquitted.
Macrosomic baby and mother both injured during delivery
Delivery of a mother’s fourth child was managed by a hospital-employed family physician (FP). Shoulder dystocia was encountered, and the FP made a 4th-degree extension of the episiotomy. The baby weighed 10 lb 14 oz at birth. The mother has fecal and urinary incontinence and pain as a result of the large episiotomy. The child has a right-sided brachial plexus injury.
Parents’ claim Failure to perform cesarean delivery caused injury to the mother and child. The FP should have recognized from the mother’s history of delivering 3 macrosomic babies and the progress of this pregnancy, that the baby was large.
Defendant’s defense The case was settled during trial.
Verdict A $1.5 million Minnesota settlement was reached that included $1.2 million for the child and $300,000 for the mother.
Surgical table folds during hysterectomy: $5.3M verdict
While a woman was undergoing a hysterectomy, the surgical table she was lying on folded up into a “U” position, causing the inserted speculum to tear the patient from vagina to rectum. The fall also caused a back injury usually attributed to falls from great distances. The patient has permanent pain, recurring diarrhea, and depression as a result of the injuries.
Patient’s claim The injuries occurred because of the defendants’ failure to read, understand, and follow the warning labels on the surgical table.
Defendant’s defense The case was settled before trial.
Verdict A $5.3 million settlement was reached with the hospital.
In this Article
- Surgical approach questioned
- Macrosomic baby and mother both injured during delivery
- Surgical table folds during hysterectomy: $5.3M verdict
What does Liletta cost to non-340B providers?
“YOUR TEENAGE PATIENT AND CONTRACEPTION: THINK
‘LONG-ACTING’ FIRST”
DAVID R. KATTAN, MD, MPH, AND
RONALD T. BURKMAN, MD (SEPTEMBER 2015)
What does Liletta cost
to non-340B providers?
Drs. Kattan and Burkman state in their article: “For providers who practice in settings eligible for 340B pricing, Liletta costs $50, a fraction of the cost of alternative intrauterine devices (IUDs). The cost is slightly higher for non-340B providers but is still significantly lower than the cost of other IUDs.”
Could you provide a cost range and the source for the non-340B cost?
Sharon J. Hawthorne, MBA
St. Louis, Missouri
Drs. Kattan and Burkman respond:
Thank you for your question and for allowing us to clarify. The manufacturer of Liletta, Actavis, offers a Patient Savings Program for private insurance patients to limit their out-of-pocket cost to $75. This program will end on December 31, 2015. Information is available at http://www.lilettacard.com.
For non-340B providers, the cost per IUD is higher, although this should be reimbursed by the patient’s insurance program. After volume discounts, the price per device is as low as $537. Without volume discounts, the price per device is $600. For more information, visit: https://www.lilettahcp.com/content/pdf/LILETTA-Quick-Reference-Guide.pdf.
Medicines360, the nonprofit partner of Actavis, states the following on its Web site (http://medicines360.org/our-mission): “Through our pharmaceutical partnerships, commercial product sales help support an affordable price to public sector clinics. This allows low income women or those without insurance the opportunity to access more healthcare choices.”
“DOES PREOPERATIVE URODYNAMICS IMPROVE OUTCOMES FOR WOMEN UNDERGOING SURGERY FOR STRESS URINARY INCONTINENCE?”
CHARLES W. NAGER, MD
(EXAMINING THE EVIDENCE; AUGUST 2015)
Priorities for determining the etiology of incontinence
While I believe Dr. Nager’s approach accurately interprets current clinical evidence, it also reflects an inadequate paradigm. Whether or not incontinence surgery should be preceded by formal invasive urodynamic evaluation is not the question. As director of urodynamics at UConn, I understand that even the most advanced clinical urodynamics evaluation is limited in what it can measure. Nowhere in that data set is “determine the etiology of incontinence.” Therefore, the more appropriate question is: When should one consider
urodynamic evaluation before making a diagnosis requiring therapy? The answer: By prioritizing aspects of lower urinary tract function.
As recommended by the International Continence Society, the diagnosing physician actually must conduct the urodynamic testing. This physician’s first priority is to determine if the bladder can maintain low storage pressures. History and physical examination must include an acknowledgment of potential causes, including chronic urethral obstruction or failure of autonomic/sympathetic regulation. Yes, in an otherwise healthy 45-year-old vaginally parous woman with stress urinary incontinence (SUI) symptoms, it is unlikely that storage pressures aren’t normally regulated. It takes little office visit time to reach that conclusion.
The diagnosing physician’s second priority is to determine the actual functional size of the urinary reservoir. Only the bladder can expel urine actively. Is there a bladder diverticulum or reflux into the upper tracts augmenting the reservoir? Bladder/urethral function is about volume management, yet the sphincteric mechanism is not tolerant of very high volumes, even in “normal” patients. Knowing reservoir volumes when leakage occurs and the relationship of these volumes to perceptions of “empty” and “full” is critical to determining how to respond to sphincteric insufficiency that produces SUI symptoms. I agree that an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms will have a problem here. However, if the diagnosing physician has any reason to doubt that the urinary reservoir has the same functionality as the bladder and that operational volumes are “normal,” then videourodynamic investigation is the most direct approach.
The third priority during evaluation is to determine how the reservoir empties. What is the source of the expulsive pressure of voiding? What is the interaction of the expulsive pressure and the urethral opening? How effectively does the bladder empty? In an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms, it is unlikely that there is a problem, but if the physician doesn’t consider how this patient’s bladder empties, determining how the sphincter is stressed during storage and how the patient might respond to intervention is impossible. If normal efficient voiding by detrusor pressurization cannot be assured by office evaluation, then urodynamic examination, including a pressure/flow study, is necessary.
The last priority is to determine how the urine storage/emptying system is controlled. This is most important to the patient but least important for diagnosis. Often this can be deduced from a simple office evaluation that includes urinalysis, a voiding diary, standing stress test, possibly simple “office cystometry” (with a large Toomey syringe, a straight catheter, and saline solution), and the patient’s history. No aspect of this last priority requires invasive computerized urodynamics—unless the physician just cannot figure it out even after considering results of the first 3 steps.
Once these evaluative priorities have been completed, a diagnosis can be considered and treatment options determined. But only then.
Phillip P. Smith, MD
Farmington, Connecticut
Dr. Nager responds:
Dr. Smith provides a very nice review of what the bladder and urethra need to do. As he points out, the most appropriate question is: When should one consider urodynamic evaluation before making a diagnosis requiring therapy? Well, when a reliable diagnosis cannot be made by history, physical examination, and simple office tests.
The literature suggests that a neurologically normal woman without prolapse and without previous incontinence surgeries can receive a reliable diagnosis without urodynamic testing. If she demonstrates SUI on office stress testing, she is not storing urine normally and urodynamics will confirm urodynamic stress incontinence 97% of the time.1 If she voluntarily voids with a normal postvoid residual, her emptying function has been assessed and is normal.
I think Dr. Smith and I both agree that, “In an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms, it is unlikely that there is a problem.” We also both agree that whenever the diagnosis is unclear, or the situation is complicated, urodynamic testing is a helpful tool to assess the bladder’s storage and emptying function. I perform urodynamics regularly in my practice; it just is not necessary before surgery in a woman without prolapse and without previous incontinence surgeries who demonstrates her SUI and has a normal urinalysis and normal postvoid residual. We seem to agree on that point also.
Reference
- Nager C, Brubaker L, Litman H, et al; Urinary Incontinence Treatment Network. A randomized trial of urodynamic testing before stress-incontinence surgery. N Engl J Med. 2012;366(21):1987–1997.
“UPDATE ON MENOPAUSE”
ANDREW M. KAUNITZ, MD (JUNE 2015)
Should Provera still be used?
Dr. Kaunitz provided an excellent review of the Women’s Health Initiative (WHI) study and a recent testosterone trial in women in his update on hormone therapy in menopause.
After the WHI revealed differences between the estrogen-alone and estrogen–progestin study arms, implicating medroxyprogesterone acetate for increased risk of breast cancer, why is Provera still being advocated by the American College of Obstetricians and Gynecologists as a progestin safe for use in menopause?
Kathleen Norman, MD
Phoenix, Arizona
Dr. Barbieri responds:
Many insurance formularies favor the use of Provera because it is inexpensive. I try to avoid using it in my practice. Many experts do not yet diligently avoid the use of Provera; some are worried about the cost impact for patients.
For additional information on reducing the use of Provera, see my July 2014 editorial, “Hormone therapy for menopausal vasomotor symptoms,” at obgmanagement.com.
Dr. Kaunitz responds:
My preference is to use micronized oral progesterone (formulated in peanut oil) for endometrial protection in menopausal women using estrogen. I use progesterone 100 mg nightly in women taking standard-dose estrogen (estradiol patch 0.05 mg, oral estradiol 1 mg, or conjugated equine estrogen 0.625 mg). However, some patients request generic medroxyprogesterone acetate because it is so inexpensive (often $4 each month).
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
“YOUR TEENAGE PATIENT AND CONTRACEPTION: THINK
‘LONG-ACTING’ FIRST”
DAVID R. KATTAN, MD, MPH, AND
RONALD T. BURKMAN, MD (SEPTEMBER 2015)
What does Liletta cost
to non-340B providers?
Drs. Kattan and Burkman state in their article: “For providers who practice in settings eligible for 340B pricing, Liletta costs $50, a fraction of the cost of alternative intrauterine devices (IUDs). The cost is slightly higher for non-340B providers but is still significantly lower than the cost of other IUDs.”
Could you provide a cost range and the source for the non-340B cost?
Sharon J. Hawthorne, MBA
St. Louis, Missouri
Drs. Kattan and Burkman respond:
Thank you for your question and for allowing us to clarify. The manufacturer of Liletta, Actavis, offers a Patient Savings Program for private insurance patients to limit their out-of-pocket cost to $75. This program will end on December 31, 2015. Information is available at http://www.lilettacard.com.
For non-340B providers, the cost per IUD is higher, although this should be reimbursed by the patient’s insurance program. After volume discounts, the price per device is as low as $537. Without volume discounts, the price per device is $600. For more information, visit: https://www.lilettahcp.com/content/pdf/LILETTA-Quick-Reference-Guide.pdf.
Medicines360, the nonprofit partner of Actavis, states the following on its Web site (http://medicines360.org/our-mission): “Through our pharmaceutical partnerships, commercial product sales help support an affordable price to public sector clinics. This allows low income women or those without insurance the opportunity to access more healthcare choices.”
“DOES PREOPERATIVE URODYNAMICS IMPROVE OUTCOMES FOR WOMEN UNDERGOING SURGERY FOR STRESS URINARY INCONTINENCE?”
CHARLES W. NAGER, MD
(EXAMINING THE EVIDENCE; AUGUST 2015)
Priorities for determining the etiology of incontinence
While I believe Dr. Nager’s approach accurately interprets current clinical evidence, it also reflects an inadequate paradigm. Whether or not incontinence surgery should be preceded by formal invasive urodynamic evaluation is not the question. As director of urodynamics at UConn, I understand that even the most advanced clinical urodynamics evaluation is limited in what it can measure. Nowhere in that data set is “determine the etiology of incontinence.” Therefore, the more appropriate question is: When should one consider
urodynamic evaluation before making a diagnosis requiring therapy? The answer: By prioritizing aspects of lower urinary tract function.
As recommended by the International Continence Society, the diagnosing physician actually must conduct the urodynamic testing. This physician’s first priority is to determine if the bladder can maintain low storage pressures. History and physical examination must include an acknowledgment of potential causes, including chronic urethral obstruction or failure of autonomic/sympathetic regulation. Yes, in an otherwise healthy 45-year-old vaginally parous woman with stress urinary incontinence (SUI) symptoms, it is unlikely that storage pressures aren’t normally regulated. It takes little office visit time to reach that conclusion.
The diagnosing physician’s second priority is to determine the actual functional size of the urinary reservoir. Only the bladder can expel urine actively. Is there a bladder diverticulum or reflux into the upper tracts augmenting the reservoir? Bladder/urethral function is about volume management, yet the sphincteric mechanism is not tolerant of very high volumes, even in “normal” patients. Knowing reservoir volumes when leakage occurs and the relationship of these volumes to perceptions of “empty” and “full” is critical to determining how to respond to sphincteric insufficiency that produces SUI symptoms. I agree that an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms will have a problem here. However, if the diagnosing physician has any reason to doubt that the urinary reservoir has the same functionality as the bladder and that operational volumes are “normal,” then videourodynamic investigation is the most direct approach.
The third priority during evaluation is to determine how the reservoir empties. What is the source of the expulsive pressure of voiding? What is the interaction of the expulsive pressure and the urethral opening? How effectively does the bladder empty? In an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms, it is unlikely that there is a problem, but if the physician doesn’t consider how this patient’s bladder empties, determining how the sphincter is stressed during storage and how the patient might respond to intervention is impossible. If normal efficient voiding by detrusor pressurization cannot be assured by office evaluation, then urodynamic examination, including a pressure/flow study, is necessary.
The last priority is to determine how the urine storage/emptying system is controlled. This is most important to the patient but least important for diagnosis. Often this can be deduced from a simple office evaluation that includes urinalysis, a voiding diary, standing stress test, possibly simple “office cystometry” (with a large Toomey syringe, a straight catheter, and saline solution), and the patient’s history. No aspect of this last priority requires invasive computerized urodynamics—unless the physician just cannot figure it out even after considering results of the first 3 steps.
Once these evaluative priorities have been completed, a diagnosis can be considered and treatment options determined. But only then.
Phillip P. Smith, MD
Farmington, Connecticut
Dr. Nager responds:
Dr. Smith provides a very nice review of what the bladder and urethra need to do. As he points out, the most appropriate question is: When should one consider urodynamic evaluation before making a diagnosis requiring therapy? Well, when a reliable diagnosis cannot be made by history, physical examination, and simple office tests.
The literature suggests that a neurologically normal woman without prolapse and without previous incontinence surgeries can receive a reliable diagnosis without urodynamic testing. If she demonstrates SUI on office stress testing, she is not storing urine normally and urodynamics will confirm urodynamic stress incontinence 97% of the time.1 If she voluntarily voids with a normal postvoid residual, her emptying function has been assessed and is normal.
I think Dr. Smith and I both agree that, “In an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms, it is unlikely that there is a problem.” We also both agree that whenever the diagnosis is unclear, or the situation is complicated, urodynamic testing is a helpful tool to assess the bladder’s storage and emptying function. I perform urodynamics regularly in my practice; it just is not necessary before surgery in a woman without prolapse and without previous incontinence surgeries who demonstrates her SUI and has a normal urinalysis and normal postvoid residual. We seem to agree on that point also.
Reference
- Nager C, Brubaker L, Litman H, et al; Urinary Incontinence Treatment Network. A randomized trial of urodynamic testing before stress-incontinence surgery. N Engl J Med. 2012;366(21):1987–1997.
“UPDATE ON MENOPAUSE”
ANDREW M. KAUNITZ, MD (JUNE 2015)
Should Provera still be used?
Dr. Kaunitz provided an excellent review of the Women’s Health Initiative (WHI) study and a recent testosterone trial in women in his update on hormone therapy in menopause.
After the WHI revealed differences between the estrogen-alone and estrogen–progestin study arms, implicating medroxyprogesterone acetate for increased risk of breast cancer, why is Provera still being advocated by the American College of Obstetricians and Gynecologists as a progestin safe for use in menopause?
Kathleen Norman, MD
Phoenix, Arizona
Dr. Barbieri responds:
Many insurance formularies favor the use of Provera because it is inexpensive. I try to avoid using it in my practice. Many experts do not yet diligently avoid the use of Provera; some are worried about the cost impact for patients.
For additional information on reducing the use of Provera, see my July 2014 editorial, “Hormone therapy for menopausal vasomotor symptoms,” at obgmanagement.com.
Dr. Kaunitz responds:
My preference is to use micronized oral progesterone (formulated in peanut oil) for endometrial protection in menopausal women using estrogen. I use progesterone 100 mg nightly in women taking standard-dose estrogen (estradiol patch 0.05 mg, oral estradiol 1 mg, or conjugated equine estrogen 0.625 mg). However, some patients request generic medroxyprogesterone acetate because it is so inexpensive (often $4 each month).
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
“YOUR TEENAGE PATIENT AND CONTRACEPTION: THINK
‘LONG-ACTING’ FIRST”
DAVID R. KATTAN, MD, MPH, AND
RONALD T. BURKMAN, MD (SEPTEMBER 2015)
What does Liletta cost
to non-340B providers?
Drs. Kattan and Burkman state in their article: “For providers who practice in settings eligible for 340B pricing, Liletta costs $50, a fraction of the cost of alternative intrauterine devices (IUDs). The cost is slightly higher for non-340B providers but is still significantly lower than the cost of other IUDs.”
Could you provide a cost range and the source for the non-340B cost?
Sharon J. Hawthorne, MBA
St. Louis, Missouri
Drs. Kattan and Burkman respond:
Thank you for your question and for allowing us to clarify. The manufacturer of Liletta, Actavis, offers a Patient Savings Program for private insurance patients to limit their out-of-pocket cost to $75. This program will end on December 31, 2015. Information is available at http://www.lilettacard.com.
For non-340B providers, the cost per IUD is higher, although this should be reimbursed by the patient’s insurance program. After volume discounts, the price per device is as low as $537. Without volume discounts, the price per device is $600. For more information, visit: https://www.lilettahcp.com/content/pdf/LILETTA-Quick-Reference-Guide.pdf.
Medicines360, the nonprofit partner of Actavis, states the following on its Web site (http://medicines360.org/our-mission): “Through our pharmaceutical partnerships, commercial product sales help support an affordable price to public sector clinics. This allows low income women or those without insurance the opportunity to access more healthcare choices.”
“DOES PREOPERATIVE URODYNAMICS IMPROVE OUTCOMES FOR WOMEN UNDERGOING SURGERY FOR STRESS URINARY INCONTINENCE?”
CHARLES W. NAGER, MD
(EXAMINING THE EVIDENCE; AUGUST 2015)
Priorities for determining the etiology of incontinence
While I believe Dr. Nager’s approach accurately interprets current clinical evidence, it also reflects an inadequate paradigm. Whether or not incontinence surgery should be preceded by formal invasive urodynamic evaluation is not the question. As director of urodynamics at UConn, I understand that even the most advanced clinical urodynamics evaluation is limited in what it can measure. Nowhere in that data set is “determine the etiology of incontinence.” Therefore, the more appropriate question is: When should one consider
urodynamic evaluation before making a diagnosis requiring therapy? The answer: By prioritizing aspects of lower urinary tract function.
As recommended by the International Continence Society, the diagnosing physician actually must conduct the urodynamic testing. This physician’s first priority is to determine if the bladder can maintain low storage pressures. History and physical examination must include an acknowledgment of potential causes, including chronic urethral obstruction or failure of autonomic/sympathetic regulation. Yes, in an otherwise healthy 45-year-old vaginally parous woman with stress urinary incontinence (SUI) symptoms, it is unlikely that storage pressures aren’t normally regulated. It takes little office visit time to reach that conclusion.
The diagnosing physician’s second priority is to determine the actual functional size of the urinary reservoir. Only the bladder can expel urine actively. Is there a bladder diverticulum or reflux into the upper tracts augmenting the reservoir? Bladder/urethral function is about volume management, yet the sphincteric mechanism is not tolerant of very high volumes, even in “normal” patients. Knowing reservoir volumes when leakage occurs and the relationship of these volumes to perceptions of “empty” and “full” is critical to determining how to respond to sphincteric insufficiency that produces SUI symptoms. I agree that an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms will have a problem here. However, if the diagnosing physician has any reason to doubt that the urinary reservoir has the same functionality as the bladder and that operational volumes are “normal,” then videourodynamic investigation is the most direct approach.
The third priority during evaluation is to determine how the reservoir empties. What is the source of the expulsive pressure of voiding? What is the interaction of the expulsive pressure and the urethral opening? How effectively does the bladder empty? In an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms, it is unlikely that there is a problem, but if the physician doesn’t consider how this patient’s bladder empties, determining how the sphincter is stressed during storage and how the patient might respond to intervention is impossible. If normal efficient voiding by detrusor pressurization cannot be assured by office evaluation, then urodynamic examination, including a pressure/flow study, is necessary.
The last priority is to determine how the urine storage/emptying system is controlled. This is most important to the patient but least important for diagnosis. Often this can be deduced from a simple office evaluation that includes urinalysis, a voiding diary, standing stress test, possibly simple “office cystometry” (with a large Toomey syringe, a straight catheter, and saline solution), and the patient’s history. No aspect of this last priority requires invasive computerized urodynamics—unless the physician just cannot figure it out even after considering results of the first 3 steps.
Once these evaluative priorities have been completed, a diagnosis can be considered and treatment options determined. But only then.
Phillip P. Smith, MD
Farmington, Connecticut
Dr. Nager responds:
Dr. Smith provides a very nice review of what the bladder and urethra need to do. As he points out, the most appropriate question is: When should one consider urodynamic evaluation before making a diagnosis requiring therapy? Well, when a reliable diagnosis cannot be made by history, physical examination, and simple office tests.
The literature suggests that a neurologically normal woman without prolapse and without previous incontinence surgeries can receive a reliable diagnosis without urodynamic testing. If she demonstrates SUI on office stress testing, she is not storing urine normally and urodynamics will confirm urodynamic stress incontinence 97% of the time.1 If she voluntarily voids with a normal postvoid residual, her emptying function has been assessed and is normal.
I think Dr. Smith and I both agree that, “In an otherwise healthy 45-year-old vaginally parous woman with SUI symptoms, it is unlikely that there is a problem.” We also both agree that whenever the diagnosis is unclear, or the situation is complicated, urodynamic testing is a helpful tool to assess the bladder’s storage and emptying function. I perform urodynamics regularly in my practice; it just is not necessary before surgery in a woman without prolapse and without previous incontinence surgeries who demonstrates her SUI and has a normal urinalysis and normal postvoid residual. We seem to agree on that point also.
Reference
- Nager C, Brubaker L, Litman H, et al; Urinary Incontinence Treatment Network. A randomized trial of urodynamic testing before stress-incontinence surgery. N Engl J Med. 2012;366(21):1987–1997.
“UPDATE ON MENOPAUSE”
ANDREW M. KAUNITZ, MD (JUNE 2015)
Should Provera still be used?
Dr. Kaunitz provided an excellent review of the Women’s Health Initiative (WHI) study and a recent testosterone trial in women in his update on hormone therapy in menopause.
After the WHI revealed differences between the estrogen-alone and estrogen–progestin study arms, implicating medroxyprogesterone acetate for increased risk of breast cancer, why is Provera still being advocated by the American College of Obstetricians and Gynecologists as a progestin safe for use in menopause?
Kathleen Norman, MD
Phoenix, Arizona
Dr. Barbieri responds:
Many insurance formularies favor the use of Provera because it is inexpensive. I try to avoid using it in my practice. Many experts do not yet diligently avoid the use of Provera; some are worried about the cost impact for patients.
For additional information on reducing the use of Provera, see my July 2014 editorial, “Hormone therapy for menopausal vasomotor symptoms,” at obgmanagement.com.
Dr. Kaunitz responds:
My preference is to use micronized oral progesterone (formulated in peanut oil) for endometrial protection in menopausal women using estrogen. I use progesterone 100 mg nightly in women taking standard-dose estrogen (estradiol patch 0.05 mg, oral estradiol 1 mg, or conjugated equine estrogen 0.625 mg). However, some patients request generic medroxyprogesterone acetate because it is so inexpensive (often $4 each month).
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Teaching patients how to eat for 1.2 in pregnancy
The setting is a medical office with a newly pregnant couple and their doctor. There is a lot of discussion and counseling planned at this visit. Some patients are anxious, some are not, but always they have questions. This scene plays itself out in my office multiple times a day.
The order will vary with the practitioner, but is likely to include a review of medical symptoms of the pregnancy, such as nausea and fatigue, or abnormal bleeding. Additionally, we will explore medical questions including family history, medical history, medications, and any hereditary genetic risk. The couples’ list of questions will sometimes be short, sometimes extensive, and inevitably includes several of the following: Can I color my hair? Can I use self-tanner or teeth whitener? Can I get a bikini wax? Can I get a massage? Can I travel? Some come in with a bag of varying herbal or vitamin supplements that they want to know are safe during pregnancy.
While these questions are important, they often supersede questions about nutrition, exercise, and pregnancy weight gain, and we do need to address those before the visit is over.
Recommendations
With that in mind, here are the key messages related to exercise and nutrition that I proactively weave into my patients’ early pregnancy visits.
Continuing exercise in pregnancy is important to maintain cardiovascular health, muscle tone, and well-being. Just as when we are not pregnant, a sedentary lifestyle affects our overall health in a negative manner, unless avoiding exercise is recommended for a medical indication. Neither overdoing nor under exercising are a good way to achieve the body’s goals. Exercising to a conversational pace is a good measurement to achieve. For those who do not have a regular exercise routine, a good-paced walk several times a week or a prenatal fitness class can be a reasonable option.
The old adage of “eating for two” is one that we need to dispense with early in the process. In actuality, eating for “1.2” should be adequate for most patients. When starting a singleton pregnancy with a normal body mass index, only about 300 more calories a day should meet the new nutritional demands. Patients who are overweight or underweight need those guidelines adjusted and sometimes, in those situations, a nutritionist’s input can be a helpful addition.
Although the nutritional demands during pregnancy increase only a little bit, what we choose to eat while pregnant is important. While cravings influence our appetite, it continues to be important to pay attention to the variety of foods on our plate.
There is no specific pregnancy diet. Simply following the normal recommendations for healthy eating is the correct idea. Making sure to get adequate folic acid – at least 800 mcg daily – beginning preconceptionally to prevent neural tube defects, and then enough calcium to encourage healthy bone development – 1,000 mg per day either through supplements or food sources – is a good place to start.
Focusing on nutrient-dense foods such as lean proteins, low-fat dairy products, fruits, vegetables, and whole grains and incorporating a variety of these foods into the diet is ideal. That looks like this: Two to three servings of vegetables of different colors, two servings of fruit, three servings of whole grains, and two to three servings of lean protein sources on a daily basis.
Protein-rich foods should be varied to include seafood, lean meats, eggs, beans, nuts, and seed sources. Ideally, all women – especially those who are pregnant or breastfeeding – should incorporate two to three servings of a variety of seafood a week into this rotation to optimize the natural benefits of omega-3 fatty acids. The data suggest that this has not been the case, and it is important to emphasize these benefits to fetal and maternal well-being both for the short and long term.
Quite frankly, avoiding seafood is likely to pose more harm than otherwise. And there are only four types of fish that should be avoided during pregnancy: shark, swordfish, tilefish, and king mackerel. That leaves us with a long list of choices to fit varying tastes and budgets; everything from salmon and canned tuna to tilapia or cod and more can be safely enjoyed during pregnancy.
Managing the visit
This new pregnancy visit does take a long time. Our office has put together a folder that includes information and handouts on recommended genetic testing; good health and nutrition in pregnancy; a schedule of visits; information on nausea and vomiting symptoms; and testing done in routine prenatal care. This serves to help the physician remember the points to discuss, streamlines the visit, and allows the patient to take material home to review without having to commit the entire visit to memory.
We also have information on our website about medications that can be used in pregnancy and other common questions that patients and their spouses can use as a reference later.
There are many topics to discuss and multiple questions to be addressed, both medical and not. Getting the couple off to a good start and with a healthy plan will impact their pregnancy and baby’s development and outcome over the next 9 months, and hopefully, encourage these healthy habits to continue. Ultimately, I remind my patients that our goals are the same, and that these 40 weeks are just practice for the parenting ahead.
Dr. Siegel is an ob.gyn. at Atlanta Women’s Obstetrics & Gynecology in Georgia. She also consults with the National Fisheries Institute.
The setting is a medical office with a newly pregnant couple and their doctor. There is a lot of discussion and counseling planned at this visit. Some patients are anxious, some are not, but always they have questions. This scene plays itself out in my office multiple times a day.
The order will vary with the practitioner, but is likely to include a review of medical symptoms of the pregnancy, such as nausea and fatigue, or abnormal bleeding. Additionally, we will explore medical questions including family history, medical history, medications, and any hereditary genetic risk. The couples’ list of questions will sometimes be short, sometimes extensive, and inevitably includes several of the following: Can I color my hair? Can I use self-tanner or teeth whitener? Can I get a bikini wax? Can I get a massage? Can I travel? Some come in with a bag of varying herbal or vitamin supplements that they want to know are safe during pregnancy.
While these questions are important, they often supersede questions about nutrition, exercise, and pregnancy weight gain, and we do need to address those before the visit is over.
Recommendations
With that in mind, here are the key messages related to exercise and nutrition that I proactively weave into my patients’ early pregnancy visits.
Continuing exercise in pregnancy is important to maintain cardiovascular health, muscle tone, and well-being. Just as when we are not pregnant, a sedentary lifestyle affects our overall health in a negative manner, unless avoiding exercise is recommended for a medical indication. Neither overdoing nor under exercising are a good way to achieve the body’s goals. Exercising to a conversational pace is a good measurement to achieve. For those who do not have a regular exercise routine, a good-paced walk several times a week or a prenatal fitness class can be a reasonable option.
The old adage of “eating for two” is one that we need to dispense with early in the process. In actuality, eating for “1.2” should be adequate for most patients. When starting a singleton pregnancy with a normal body mass index, only about 300 more calories a day should meet the new nutritional demands. Patients who are overweight or underweight need those guidelines adjusted and sometimes, in those situations, a nutritionist’s input can be a helpful addition.
Although the nutritional demands during pregnancy increase only a little bit, what we choose to eat while pregnant is important. While cravings influence our appetite, it continues to be important to pay attention to the variety of foods on our plate.
There is no specific pregnancy diet. Simply following the normal recommendations for healthy eating is the correct idea. Making sure to get adequate folic acid – at least 800 mcg daily – beginning preconceptionally to prevent neural tube defects, and then enough calcium to encourage healthy bone development – 1,000 mg per day either through supplements or food sources – is a good place to start.
Focusing on nutrient-dense foods such as lean proteins, low-fat dairy products, fruits, vegetables, and whole grains and incorporating a variety of these foods into the diet is ideal. That looks like this: Two to three servings of vegetables of different colors, two servings of fruit, three servings of whole grains, and two to three servings of lean protein sources on a daily basis.
Protein-rich foods should be varied to include seafood, lean meats, eggs, beans, nuts, and seed sources. Ideally, all women – especially those who are pregnant or breastfeeding – should incorporate two to three servings of a variety of seafood a week into this rotation to optimize the natural benefits of omega-3 fatty acids. The data suggest that this has not been the case, and it is important to emphasize these benefits to fetal and maternal well-being both for the short and long term.
Quite frankly, avoiding seafood is likely to pose more harm than otherwise. And there are only four types of fish that should be avoided during pregnancy: shark, swordfish, tilefish, and king mackerel. That leaves us with a long list of choices to fit varying tastes and budgets; everything from salmon and canned tuna to tilapia or cod and more can be safely enjoyed during pregnancy.
Managing the visit
This new pregnancy visit does take a long time. Our office has put together a folder that includes information and handouts on recommended genetic testing; good health and nutrition in pregnancy; a schedule of visits; information on nausea and vomiting symptoms; and testing done in routine prenatal care. This serves to help the physician remember the points to discuss, streamlines the visit, and allows the patient to take material home to review without having to commit the entire visit to memory.
We also have information on our website about medications that can be used in pregnancy and other common questions that patients and their spouses can use as a reference later.
There are many topics to discuss and multiple questions to be addressed, both medical and not. Getting the couple off to a good start and with a healthy plan will impact their pregnancy and baby’s development and outcome over the next 9 months, and hopefully, encourage these healthy habits to continue. Ultimately, I remind my patients that our goals are the same, and that these 40 weeks are just practice for the parenting ahead.
Dr. Siegel is an ob.gyn. at Atlanta Women’s Obstetrics & Gynecology in Georgia. She also consults with the National Fisheries Institute.
The setting is a medical office with a newly pregnant couple and their doctor. There is a lot of discussion and counseling planned at this visit. Some patients are anxious, some are not, but always they have questions. This scene plays itself out in my office multiple times a day.
The order will vary with the practitioner, but is likely to include a review of medical symptoms of the pregnancy, such as nausea and fatigue, or abnormal bleeding. Additionally, we will explore medical questions including family history, medical history, medications, and any hereditary genetic risk. The couples’ list of questions will sometimes be short, sometimes extensive, and inevitably includes several of the following: Can I color my hair? Can I use self-tanner or teeth whitener? Can I get a bikini wax? Can I get a massage? Can I travel? Some come in with a bag of varying herbal or vitamin supplements that they want to know are safe during pregnancy.
While these questions are important, they often supersede questions about nutrition, exercise, and pregnancy weight gain, and we do need to address those before the visit is over.
Recommendations
With that in mind, here are the key messages related to exercise and nutrition that I proactively weave into my patients’ early pregnancy visits.
Continuing exercise in pregnancy is important to maintain cardiovascular health, muscle tone, and well-being. Just as when we are not pregnant, a sedentary lifestyle affects our overall health in a negative manner, unless avoiding exercise is recommended for a medical indication. Neither overdoing nor under exercising are a good way to achieve the body’s goals. Exercising to a conversational pace is a good measurement to achieve. For those who do not have a regular exercise routine, a good-paced walk several times a week or a prenatal fitness class can be a reasonable option.
The old adage of “eating for two” is one that we need to dispense with early in the process. In actuality, eating for “1.2” should be adequate for most patients. When starting a singleton pregnancy with a normal body mass index, only about 300 more calories a day should meet the new nutritional demands. Patients who are overweight or underweight need those guidelines adjusted and sometimes, in those situations, a nutritionist’s input can be a helpful addition.
Although the nutritional demands during pregnancy increase only a little bit, what we choose to eat while pregnant is important. While cravings influence our appetite, it continues to be important to pay attention to the variety of foods on our plate.
There is no specific pregnancy diet. Simply following the normal recommendations for healthy eating is the correct idea. Making sure to get adequate folic acid – at least 800 mcg daily – beginning preconceptionally to prevent neural tube defects, and then enough calcium to encourage healthy bone development – 1,000 mg per day either through supplements or food sources – is a good place to start.
Focusing on nutrient-dense foods such as lean proteins, low-fat dairy products, fruits, vegetables, and whole grains and incorporating a variety of these foods into the diet is ideal. That looks like this: Two to three servings of vegetables of different colors, two servings of fruit, three servings of whole grains, and two to three servings of lean protein sources on a daily basis.
Protein-rich foods should be varied to include seafood, lean meats, eggs, beans, nuts, and seed sources. Ideally, all women – especially those who are pregnant or breastfeeding – should incorporate two to three servings of a variety of seafood a week into this rotation to optimize the natural benefits of omega-3 fatty acids. The data suggest that this has not been the case, and it is important to emphasize these benefits to fetal and maternal well-being both for the short and long term.
Quite frankly, avoiding seafood is likely to pose more harm than otherwise. And there are only four types of fish that should be avoided during pregnancy: shark, swordfish, tilefish, and king mackerel. That leaves us with a long list of choices to fit varying tastes and budgets; everything from salmon and canned tuna to tilapia or cod and more can be safely enjoyed during pregnancy.
Managing the visit
This new pregnancy visit does take a long time. Our office has put together a folder that includes information and handouts on recommended genetic testing; good health and nutrition in pregnancy; a schedule of visits; information on nausea and vomiting symptoms; and testing done in routine prenatal care. This serves to help the physician remember the points to discuss, streamlines the visit, and allows the patient to take material home to review without having to commit the entire visit to memory.
We also have information on our website about medications that can be used in pregnancy and other common questions that patients and their spouses can use as a reference later.
There are many topics to discuss and multiple questions to be addressed, both medical and not. Getting the couple off to a good start and with a healthy plan will impact their pregnancy and baby’s development and outcome over the next 9 months, and hopefully, encourage these healthy habits to continue. Ultimately, I remind my patients that our goals are the same, and that these 40 weeks are just practice for the parenting ahead.
Dr. Siegel is an ob.gyn. at Atlanta Women’s Obstetrics & Gynecology in Georgia. She also consults with the National Fisheries Institute.
Approaches to smoking cessation before and during pregnancy
Data consistently support the negative impact of tobacco use in pregnancy with respect to pregnancy outcome, and the benefits of discontinuation or reduction as early as possible.
Recent analyses by investigators at the National Center on Birth Defects and Developmental Disabilities at the Centers for Disease Control and Prevention suggest that more than 6% of oral clefts, or 430 affected infants per year in the United States, could be prevented annually if women can discontinue tobacco use prior to conception (Birth Defects Res A Clin Mol Teratol. 2014 Nov;100[11]:822-5). Similarly, preconception smoking cessation could prevent the 1.4% of nonsyndromic congenital heart defects that are attributable to maternal smoking in the first trimester (J Pediatr. 2015 Apr;166[4]:978-984.e2).
With respect to the risk for adverse outcomes beyond the first trimester, recent data also show clearly that the trimester of discontinuation is related to intrauterine growth restriction in a dose-response fashion (Obstet Gynecol. 2015 Jun;125[6]:1452-9).
In a recent population-based retrospective cohort study of 927,424 singleton births 2006-2012 in Ohio, the adjusted relative risk of infant birth weight less than the 5th percentile for those women who discontinued smoking after the first trimester compared with nonsmokers was 1.25 (95% confidence interval, 1.17-1.33); for those who discontinued after the second trimester the relative risk was 1.83 (95% CI, 1.68-1.99); and for those who smoked throughout pregnancy the relative risk was 2.44 (95% CI, 2.37-2.51).
Given the compelling reasons to encourage women to stop smoking or at least to reduce harm during pregnancy, options for assistance with smoking cessation are of high interest. Beyond simple screening and advice to quit, cognitive behavioral therapy has been shown to provide some benefit. The addition of pharmacologic treatment with nicotine replacement therapy (NRT) has been studied in six randomized clinical trials (RCTs) conducted in pregnant women, four of which compared NRT plus advice/behavioral support to placebo plus advice/behavioral support, and two of which compared NRT plus advice/behavioral support to advice/behavioral support alone.
In a recent Cochrane systematic review of these studies, no statistically significant evidence of effectiveness was demonstrated for NRT versus placebo/control in a pooled sample of 1,745 pregnant patients (risk ratio, 1.33, 95% CI, 0.93-1.91). However, there was high heterogeneity in the dose of NRT and the delivery method (e.g., gum, patch) across studies, and poor adherence to the NRT treatment in all trials (Cochrane Database Syst Rev. 2012 Sep 12;9:CD010078).
With respect to safety, in the same Cochrane review there were no statistically significant differences in rates of miscarriage, stillbirth, premature birth, birth weight, low birth weight, admissions to neonatal intensive care, or neonatal death between NRT and control groups. However, small sample sizes and adherence issues across these trials hampered the interpretability of these data.
One relatively large claims database study from the United Kingdom, which was published recently, examined major congenital anomalies following prescription of NRT. The investigators found no increased risks for most major defects following NRT prescription; the only significant association was with respiratory defects (Pediatrics. 2015 May;135[5]:859-67).
The general thinking has been that for a woman who is unable to quit smoking without pharmacological assistance, NRT that delivers nicotine alone to the developing fetus may be a better option than exposure to the multiple toxins that are contained in tobacco smoke. However, there is considerable controversy over the potential adverse neurotoxic effects of nicotine itself and long-term neurodevelopmental studies on children prenatally exposed to NRT are lacking.
Other options include bupropion and varenicline, neither of which have been studied in RCTs in pregnancy. Bupropion has been evaluated in a small controlled cohort study, a claims database study (n = 1,236 first-trimester exposed), and two case-control studies. None of these studies was focused on use of bupropion exclusively for smoking cessation, but rather for the more common indication of maternal depression. The first two studies suggested no increased risks for adverse pregnancy outcomes compared to women the same underlying conditions; the case control studies suggested small increased risks for heart defects but not the same ones in both studies (Expert Opin Drug Saf. 2014 Dec;13[12]:1721-31). The limited data on varenicline are too sparse to make any inferences.
Another possible alternative that has been gaining in popularity are e-cigarettes or related vapor products, which are touted to have advantages with respect to harm reduction, primarily because of possible improved adherence due to their similarity to conventional smoking. However, there is large variability in the amount of nicotine in the vapor of various e-cigarette brands, and some have suggested that e-cigarette users engage in longer puff duration than do those who smoke conventional cigarettes. To my knowledge, there are no controlled studies of e-cigarette use in pregnancy, but the concerns previously raised regarding nicotine exposure in any form likely apply to this delivery method (Birth Defects Res A Clin Mol Teratol. 2015 Mar;103[3]:186-95).
What is the role of the obstetrician in identifying nicotine and tobacco exposure in their patients and encouraging cessation or reduction prior to and during pregnancy?
The first responsibility is to screen pregnant women. A recent survey study suggests that 40% of the responding ob.gyns. never screened pregnant patients for use of noncombustible tobacco products such as e-cigarettes (Am J Obstet Gynecol. 2014 Dec;211[6]:695.e1-7). In the United States, an analysis of the Pregnancy Risk Assessment Monitoring System data collected across several states from 2009 to 2010 suggests that about a quarter of 3,559 pregnant women who reported smoking in the 3 months before pregnancy did not receive any interventions to stop smoking (Prev Med. 2015 Sep;78:92-100). In addition, four out of five ob.gyns. surveyed in 2012 were unaware of the Affordable Care Act provision that requires states to provide tobacco cessation coverage for pregnant Medicaid beneficiaries (Prev Med Rep. 2015;2:686-88).
Dr. Chambers is professor of pediatrics and director of clinical research at Rady Children’s Hospital, and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is director of MotherToBaby California, past president of the Organization of Teratology Information Specialists, and past president of the Teratology Society. She reported having no financial disclosures relevant to this column, but has received research funding from GlaxoSmithKline and Pfizer for unrelated products.
Data consistently support the negative impact of tobacco use in pregnancy with respect to pregnancy outcome, and the benefits of discontinuation or reduction as early as possible.
Recent analyses by investigators at the National Center on Birth Defects and Developmental Disabilities at the Centers for Disease Control and Prevention suggest that more than 6% of oral clefts, or 430 affected infants per year in the United States, could be prevented annually if women can discontinue tobacco use prior to conception (Birth Defects Res A Clin Mol Teratol. 2014 Nov;100[11]:822-5). Similarly, preconception smoking cessation could prevent the 1.4% of nonsyndromic congenital heart defects that are attributable to maternal smoking in the first trimester (J Pediatr. 2015 Apr;166[4]:978-984.e2).
With respect to the risk for adverse outcomes beyond the first trimester, recent data also show clearly that the trimester of discontinuation is related to intrauterine growth restriction in a dose-response fashion (Obstet Gynecol. 2015 Jun;125[6]:1452-9).
In a recent population-based retrospective cohort study of 927,424 singleton births 2006-2012 in Ohio, the adjusted relative risk of infant birth weight less than the 5th percentile for those women who discontinued smoking after the first trimester compared with nonsmokers was 1.25 (95% confidence interval, 1.17-1.33); for those who discontinued after the second trimester the relative risk was 1.83 (95% CI, 1.68-1.99); and for those who smoked throughout pregnancy the relative risk was 2.44 (95% CI, 2.37-2.51).
Given the compelling reasons to encourage women to stop smoking or at least to reduce harm during pregnancy, options for assistance with smoking cessation are of high interest. Beyond simple screening and advice to quit, cognitive behavioral therapy has been shown to provide some benefit. The addition of pharmacologic treatment with nicotine replacement therapy (NRT) has been studied in six randomized clinical trials (RCTs) conducted in pregnant women, four of which compared NRT plus advice/behavioral support to placebo plus advice/behavioral support, and two of which compared NRT plus advice/behavioral support to advice/behavioral support alone.
In a recent Cochrane systematic review of these studies, no statistically significant evidence of effectiveness was demonstrated for NRT versus placebo/control in a pooled sample of 1,745 pregnant patients (risk ratio, 1.33, 95% CI, 0.93-1.91). However, there was high heterogeneity in the dose of NRT and the delivery method (e.g., gum, patch) across studies, and poor adherence to the NRT treatment in all trials (Cochrane Database Syst Rev. 2012 Sep 12;9:CD010078).
With respect to safety, in the same Cochrane review there were no statistically significant differences in rates of miscarriage, stillbirth, premature birth, birth weight, low birth weight, admissions to neonatal intensive care, or neonatal death between NRT and control groups. However, small sample sizes and adherence issues across these trials hampered the interpretability of these data.
One relatively large claims database study from the United Kingdom, which was published recently, examined major congenital anomalies following prescription of NRT. The investigators found no increased risks for most major defects following NRT prescription; the only significant association was with respiratory defects (Pediatrics. 2015 May;135[5]:859-67).
The general thinking has been that for a woman who is unable to quit smoking without pharmacological assistance, NRT that delivers nicotine alone to the developing fetus may be a better option than exposure to the multiple toxins that are contained in tobacco smoke. However, there is considerable controversy over the potential adverse neurotoxic effects of nicotine itself and long-term neurodevelopmental studies on children prenatally exposed to NRT are lacking.
Other options include bupropion and varenicline, neither of which have been studied in RCTs in pregnancy. Bupropion has been evaluated in a small controlled cohort study, a claims database study (n = 1,236 first-trimester exposed), and two case-control studies. None of these studies was focused on use of bupropion exclusively for smoking cessation, but rather for the more common indication of maternal depression. The first two studies suggested no increased risks for adverse pregnancy outcomes compared to women the same underlying conditions; the case control studies suggested small increased risks for heart defects but not the same ones in both studies (Expert Opin Drug Saf. 2014 Dec;13[12]:1721-31). The limited data on varenicline are too sparse to make any inferences.
Another possible alternative that has been gaining in popularity are e-cigarettes or related vapor products, which are touted to have advantages with respect to harm reduction, primarily because of possible improved adherence due to their similarity to conventional smoking. However, there is large variability in the amount of nicotine in the vapor of various e-cigarette brands, and some have suggested that e-cigarette users engage in longer puff duration than do those who smoke conventional cigarettes. To my knowledge, there are no controlled studies of e-cigarette use in pregnancy, but the concerns previously raised regarding nicotine exposure in any form likely apply to this delivery method (Birth Defects Res A Clin Mol Teratol. 2015 Mar;103[3]:186-95).
What is the role of the obstetrician in identifying nicotine and tobacco exposure in their patients and encouraging cessation or reduction prior to and during pregnancy?
The first responsibility is to screen pregnant women. A recent survey study suggests that 40% of the responding ob.gyns. never screened pregnant patients for use of noncombustible tobacco products such as e-cigarettes (Am J Obstet Gynecol. 2014 Dec;211[6]:695.e1-7). In the United States, an analysis of the Pregnancy Risk Assessment Monitoring System data collected across several states from 2009 to 2010 suggests that about a quarter of 3,559 pregnant women who reported smoking in the 3 months before pregnancy did not receive any interventions to stop smoking (Prev Med. 2015 Sep;78:92-100). In addition, four out of five ob.gyns. surveyed in 2012 were unaware of the Affordable Care Act provision that requires states to provide tobacco cessation coverage for pregnant Medicaid beneficiaries (Prev Med Rep. 2015;2:686-88).
Dr. Chambers is professor of pediatrics and director of clinical research at Rady Children’s Hospital, and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is director of MotherToBaby California, past president of the Organization of Teratology Information Specialists, and past president of the Teratology Society. She reported having no financial disclosures relevant to this column, but has received research funding from GlaxoSmithKline and Pfizer for unrelated products.
Data consistently support the negative impact of tobacco use in pregnancy with respect to pregnancy outcome, and the benefits of discontinuation or reduction as early as possible.
Recent analyses by investigators at the National Center on Birth Defects and Developmental Disabilities at the Centers for Disease Control and Prevention suggest that more than 6% of oral clefts, or 430 affected infants per year in the United States, could be prevented annually if women can discontinue tobacco use prior to conception (Birth Defects Res A Clin Mol Teratol. 2014 Nov;100[11]:822-5). Similarly, preconception smoking cessation could prevent the 1.4% of nonsyndromic congenital heart defects that are attributable to maternal smoking in the first trimester (J Pediatr. 2015 Apr;166[4]:978-984.e2).
With respect to the risk for adverse outcomes beyond the first trimester, recent data also show clearly that the trimester of discontinuation is related to intrauterine growth restriction in a dose-response fashion (Obstet Gynecol. 2015 Jun;125[6]:1452-9).
In a recent population-based retrospective cohort study of 927,424 singleton births 2006-2012 in Ohio, the adjusted relative risk of infant birth weight less than the 5th percentile for those women who discontinued smoking after the first trimester compared with nonsmokers was 1.25 (95% confidence interval, 1.17-1.33); for those who discontinued after the second trimester the relative risk was 1.83 (95% CI, 1.68-1.99); and for those who smoked throughout pregnancy the relative risk was 2.44 (95% CI, 2.37-2.51).
Given the compelling reasons to encourage women to stop smoking or at least to reduce harm during pregnancy, options for assistance with smoking cessation are of high interest. Beyond simple screening and advice to quit, cognitive behavioral therapy has been shown to provide some benefit. The addition of pharmacologic treatment with nicotine replacement therapy (NRT) has been studied in six randomized clinical trials (RCTs) conducted in pregnant women, four of which compared NRT plus advice/behavioral support to placebo plus advice/behavioral support, and two of which compared NRT plus advice/behavioral support to advice/behavioral support alone.
In a recent Cochrane systematic review of these studies, no statistically significant evidence of effectiveness was demonstrated for NRT versus placebo/control in a pooled sample of 1,745 pregnant patients (risk ratio, 1.33, 95% CI, 0.93-1.91). However, there was high heterogeneity in the dose of NRT and the delivery method (e.g., gum, patch) across studies, and poor adherence to the NRT treatment in all trials (Cochrane Database Syst Rev. 2012 Sep 12;9:CD010078).
With respect to safety, in the same Cochrane review there were no statistically significant differences in rates of miscarriage, stillbirth, premature birth, birth weight, low birth weight, admissions to neonatal intensive care, or neonatal death between NRT and control groups. However, small sample sizes and adherence issues across these trials hampered the interpretability of these data.
One relatively large claims database study from the United Kingdom, which was published recently, examined major congenital anomalies following prescription of NRT. The investigators found no increased risks for most major defects following NRT prescription; the only significant association was with respiratory defects (Pediatrics. 2015 May;135[5]:859-67).
The general thinking has been that for a woman who is unable to quit smoking without pharmacological assistance, NRT that delivers nicotine alone to the developing fetus may be a better option than exposure to the multiple toxins that are contained in tobacco smoke. However, there is considerable controversy over the potential adverse neurotoxic effects of nicotine itself and long-term neurodevelopmental studies on children prenatally exposed to NRT are lacking.
Other options include bupropion and varenicline, neither of which have been studied in RCTs in pregnancy. Bupropion has been evaluated in a small controlled cohort study, a claims database study (n = 1,236 first-trimester exposed), and two case-control studies. None of these studies was focused on use of bupropion exclusively for smoking cessation, but rather for the more common indication of maternal depression. The first two studies suggested no increased risks for adverse pregnancy outcomes compared to women the same underlying conditions; the case control studies suggested small increased risks for heart defects but not the same ones in both studies (Expert Opin Drug Saf. 2014 Dec;13[12]:1721-31). The limited data on varenicline are too sparse to make any inferences.
Another possible alternative that has been gaining in popularity are e-cigarettes or related vapor products, which are touted to have advantages with respect to harm reduction, primarily because of possible improved adherence due to their similarity to conventional smoking. However, there is large variability in the amount of nicotine in the vapor of various e-cigarette brands, and some have suggested that e-cigarette users engage in longer puff duration than do those who smoke conventional cigarettes. To my knowledge, there are no controlled studies of e-cigarette use in pregnancy, but the concerns previously raised regarding nicotine exposure in any form likely apply to this delivery method (Birth Defects Res A Clin Mol Teratol. 2015 Mar;103[3]:186-95).
What is the role of the obstetrician in identifying nicotine and tobacco exposure in their patients and encouraging cessation or reduction prior to and during pregnancy?
The first responsibility is to screen pregnant women. A recent survey study suggests that 40% of the responding ob.gyns. never screened pregnant patients for use of noncombustible tobacco products such as e-cigarettes (Am J Obstet Gynecol. 2014 Dec;211[6]:695.e1-7). In the United States, an analysis of the Pregnancy Risk Assessment Monitoring System data collected across several states from 2009 to 2010 suggests that about a quarter of 3,559 pregnant women who reported smoking in the 3 months before pregnancy did not receive any interventions to stop smoking (Prev Med. 2015 Sep;78:92-100). In addition, four out of five ob.gyns. surveyed in 2012 were unaware of the Affordable Care Act provision that requires states to provide tobacco cessation coverage for pregnant Medicaid beneficiaries (Prev Med Rep. 2015;2:686-88).
Dr. Chambers is professor of pediatrics and director of clinical research at Rady Children’s Hospital, and associate director of the Clinical and Translational Research Institute at the University of California, San Diego. She is director of MotherToBaby California, past president of the Organization of Teratology Information Specialists, and past president of the Teratology Society. She reported having no financial disclosures relevant to this column, but has received research funding from GlaxoSmithKline and Pfizer for unrelated products.
ACOG, SMFM offer guidance on periviable births
The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine have released a new consensus document recommending best practices in the care of neonates born between 20 and 25 weeks’ gestation.
The periviable birth guidance provides recommendations on the proper level of care at the hospital, the need for counseling on short- and long-term neonatal outcomes, family counseling, and a predelivery plan. The guidance also makes specific recommendations for interventions based on the best estimate of gestational age (Obstet Gynecol. 2015;126:e82-e94).
“Care during the periviable period is incredibly complex, and requires providers to take into account a wide variety of considerations,” Dr. Brian M. Mercer, past president of SMFM and a lead author of the document, said in a statement. “Just as important as trying to predict outcomes is the role of counseling patients in a way that is both accurate and empathetic.”
ACOG and SMFM suggest that clinicians use a prediction tool, rather than relying on gestational age and birth weight estimates alone, when estimating outcomes before a periviable birth. The National Institute of Child Health and Human Development has a tool that can aid in predicting outcomes for extremely preterm births that is based on data from more than 4,400 live births between 22 and 25 weeks’ gestation.
Since both infant and mother will need immediate advanced care after delivery, the groups also recommend that deliveries during the periviable period occur at centers with level III-IV neonatal intensive care units and a level III-IV maternal care designation. These facilities can provide immediate resuscitation, as well as other resources such as respiratory technology and 24-hour newborn imaging. When possible, transfers should be made before delivery, according to the consensus document.
Counseling is another essential element in the management of periviable birth. Family counseling with a team of physicians, including the ob.gyn., maternal-fetal medicine specialist, and neonatologist, should address the likely maternal and neonatal outcomes and the family’s values, including the option for palliative care. ACOG and SMFM recommended making a predelivery plan with the family with the recognition that it would be modified based on the clinical situation.
The document also provides general guidance for performing specific obstetric interventions based on gestational age. For instance, ACOG and SMFM suggest that clinicians “consider” antenatal corticosteriod administration when the estimated gestational age is 23 weeks. They “recommend” antenatal corticosteriods starting at 24 weeks’ gestation through 25 weeks’ gestation.
The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine have released a new consensus document recommending best practices in the care of neonates born between 20 and 25 weeks’ gestation.
The periviable birth guidance provides recommendations on the proper level of care at the hospital, the need for counseling on short- and long-term neonatal outcomes, family counseling, and a predelivery plan. The guidance also makes specific recommendations for interventions based on the best estimate of gestational age (Obstet Gynecol. 2015;126:e82-e94).
“Care during the periviable period is incredibly complex, and requires providers to take into account a wide variety of considerations,” Dr. Brian M. Mercer, past president of SMFM and a lead author of the document, said in a statement. “Just as important as trying to predict outcomes is the role of counseling patients in a way that is both accurate and empathetic.”
ACOG and SMFM suggest that clinicians use a prediction tool, rather than relying on gestational age and birth weight estimates alone, when estimating outcomes before a periviable birth. The National Institute of Child Health and Human Development has a tool that can aid in predicting outcomes for extremely preterm births that is based on data from more than 4,400 live births between 22 and 25 weeks’ gestation.
Since both infant and mother will need immediate advanced care after delivery, the groups also recommend that deliveries during the periviable period occur at centers with level III-IV neonatal intensive care units and a level III-IV maternal care designation. These facilities can provide immediate resuscitation, as well as other resources such as respiratory technology and 24-hour newborn imaging. When possible, transfers should be made before delivery, according to the consensus document.
Counseling is another essential element in the management of periviable birth. Family counseling with a team of physicians, including the ob.gyn., maternal-fetal medicine specialist, and neonatologist, should address the likely maternal and neonatal outcomes and the family’s values, including the option for palliative care. ACOG and SMFM recommended making a predelivery plan with the family with the recognition that it would be modified based on the clinical situation.
The document also provides general guidance for performing specific obstetric interventions based on gestational age. For instance, ACOG and SMFM suggest that clinicians “consider” antenatal corticosteriod administration when the estimated gestational age is 23 weeks. They “recommend” antenatal corticosteriods starting at 24 weeks’ gestation through 25 weeks’ gestation.
The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine have released a new consensus document recommending best practices in the care of neonates born between 20 and 25 weeks’ gestation.
The periviable birth guidance provides recommendations on the proper level of care at the hospital, the need for counseling on short- and long-term neonatal outcomes, family counseling, and a predelivery plan. The guidance also makes specific recommendations for interventions based on the best estimate of gestational age (Obstet Gynecol. 2015;126:e82-e94).
“Care during the periviable period is incredibly complex, and requires providers to take into account a wide variety of considerations,” Dr. Brian M. Mercer, past president of SMFM and a lead author of the document, said in a statement. “Just as important as trying to predict outcomes is the role of counseling patients in a way that is both accurate and empathetic.”
ACOG and SMFM suggest that clinicians use a prediction tool, rather than relying on gestational age and birth weight estimates alone, when estimating outcomes before a periviable birth. The National Institute of Child Health and Human Development has a tool that can aid in predicting outcomes for extremely preterm births that is based on data from more than 4,400 live births between 22 and 25 weeks’ gestation.
Since both infant and mother will need immediate advanced care after delivery, the groups also recommend that deliveries during the periviable period occur at centers with level III-IV neonatal intensive care units and a level III-IV maternal care designation. These facilities can provide immediate resuscitation, as well as other resources such as respiratory technology and 24-hour newborn imaging. When possible, transfers should be made before delivery, according to the consensus document.
Counseling is another essential element in the management of periviable birth. Family counseling with a team of physicians, including the ob.gyn., maternal-fetal medicine specialist, and neonatologist, should address the likely maternal and neonatal outcomes and the family’s values, including the option for palliative care. ACOG and SMFM recommended making a predelivery plan with the family with the recognition that it would be modified based on the clinical situation.
The document also provides general guidance for performing specific obstetric interventions based on gestational age. For instance, ACOG and SMFM suggest that clinicians “consider” antenatal corticosteriod administration when the estimated gestational age is 23 weeks. They “recommend” antenatal corticosteriods starting at 24 weeks’ gestation through 25 weeks’ gestation.
FROM OBSTETRICS AND GYNECOLOGY
Lessons learned from the history of VBAC
In December 2014, The Wall Street Journal ran an article about a young mother who wanted a vaginal birth after C-section (VBAC) for her second child. After her hospital stopped offering VBACs, the woman had to find another place to deliver. She did have a successful VBAC, but her story is not unique – many women may not receive adequate consultations about or provider support for VBAC as a delivery option.
According to the article, a lack of clinical support was the reason the hospital discontinued VBACs. Although the hospital’s decision may have frustrated the mother, this ensured that she would not be promised a birthing option that the hospital could not deliver – in all senses of this word. Successful VBAC requires proper patient selection, appropriate consent and adequate provisions in case of emergencies.
Not every hospital has made such a choice. Based on studies of a trial of labor after cesarean, conducted after the 1960s, the rate of VBACs increased. As VBACs became more common, the approach to the procedure became more relaxed. VBACs went from only being performed in tertiary care hospitals with appropriate support for emergencies, to community hospitals with no backup. Patient selection became less rigorous, and the rate of complications went up, which, in turn, caused the number of associated legal claims to rise. Hospitals started discouraging VBACs, and ob.gyns. no longer counseled their patients about this option. The VBAC rate decreased, and the C-section rate increased.
Today, many women want to pursue a trial of labor after cesarean. Data from large clinical studies have demonstrated the safety and success of VBAC with proper care. Because of the storied history and a revival of interest in VBACs, we have invited Dr. Mark Landon, the Richard L. Meiling Professor and chairman of the department of obstetrics and gynecology at the Ohio State University, and the lead on one of the recent seminal VBAC studies, to address this topic.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece reported having no relevant financial disclosures. He is the medical editor of this column. Contact him at [email protected].
In December 2014, The Wall Street Journal ran an article about a young mother who wanted a vaginal birth after C-section (VBAC) for her second child. After her hospital stopped offering VBACs, the woman had to find another place to deliver. She did have a successful VBAC, but her story is not unique – many women may not receive adequate consultations about or provider support for VBAC as a delivery option.
According to the article, a lack of clinical support was the reason the hospital discontinued VBACs. Although the hospital’s decision may have frustrated the mother, this ensured that she would not be promised a birthing option that the hospital could not deliver – in all senses of this word. Successful VBAC requires proper patient selection, appropriate consent and adequate provisions in case of emergencies.
Not every hospital has made such a choice. Based on studies of a trial of labor after cesarean, conducted after the 1960s, the rate of VBACs increased. As VBACs became more common, the approach to the procedure became more relaxed. VBACs went from only being performed in tertiary care hospitals with appropriate support for emergencies, to community hospitals with no backup. Patient selection became less rigorous, and the rate of complications went up, which, in turn, caused the number of associated legal claims to rise. Hospitals started discouraging VBACs, and ob.gyns. no longer counseled their patients about this option. The VBAC rate decreased, and the C-section rate increased.
Today, many women want to pursue a trial of labor after cesarean. Data from large clinical studies have demonstrated the safety and success of VBAC with proper care. Because of the storied history and a revival of interest in VBACs, we have invited Dr. Mark Landon, the Richard L. Meiling Professor and chairman of the department of obstetrics and gynecology at the Ohio State University, and the lead on one of the recent seminal VBAC studies, to address this topic.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece reported having no relevant financial disclosures. He is the medical editor of this column. Contact him at [email protected].
In December 2014, The Wall Street Journal ran an article about a young mother who wanted a vaginal birth after C-section (VBAC) for her second child. After her hospital stopped offering VBACs, the woman had to find another place to deliver. She did have a successful VBAC, but her story is not unique – many women may not receive adequate consultations about or provider support for VBAC as a delivery option.
According to the article, a lack of clinical support was the reason the hospital discontinued VBACs. Although the hospital’s decision may have frustrated the mother, this ensured that she would not be promised a birthing option that the hospital could not deliver – in all senses of this word. Successful VBAC requires proper patient selection, appropriate consent and adequate provisions in case of emergencies.
Not every hospital has made such a choice. Based on studies of a trial of labor after cesarean, conducted after the 1960s, the rate of VBACs increased. As VBACs became more common, the approach to the procedure became more relaxed. VBACs went from only being performed in tertiary care hospitals with appropriate support for emergencies, to community hospitals with no backup. Patient selection became less rigorous, and the rate of complications went up, which, in turn, caused the number of associated legal claims to rise. Hospitals started discouraging VBACs, and ob.gyns. no longer counseled their patients about this option. The VBAC rate decreased, and the C-section rate increased.
Today, many women want to pursue a trial of labor after cesarean. Data from large clinical studies have demonstrated the safety and success of VBAC with proper care. Because of the storied history and a revival of interest in VBACs, we have invited Dr. Mark Landon, the Richard L. Meiling Professor and chairman of the department of obstetrics and gynecology at the Ohio State University, and the lead on one of the recent seminal VBAC studies, to address this topic.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece reported having no relevant financial disclosures. He is the medical editor of this column. Contact him at [email protected].
Barriers to VBAC remain in spite of evidence
The relative safety of vaginal birth after cesarean (VBAC) has been documented in several large-scale studies in the past 15 years, and was affirmed in 2010 through a National Institutes of Health consensus development conference and a practice bulletin from the American College of Obstetricians and Gynecologists. Yet, despite all this research and review, rates of a trial of labor after cesarean (TOLAC) have increased only modestly in the last several years.
Approximately 20% of all births in 2013 in women with a history of one cesarean section involved a trial of labor, according to a recent report from the Centers for Disease Control and Prevention. This represents only a small increase from 2006, when the TOLAC rate had plummeted to approximately 15%.
The limited change is concerning because up to two-thirds of women with a prior cesarean delivery are candidates for a trial of labor, and many of them are excellent candidates. In total, 70% of the women who attempted labor in 2013 after a previous cesarean had successful VBACs, the CDC data shows.
Several European countries have TOLAC rates between 50% and 70%, but in the United States, as evidenced by the recent CDC data, there continues to be an underutilization of attempted VBAC. We must ask ourselves, are women truly able to choose TOLAC, or are they being dissuaded by the health care system?
I believe that the barriers are still pervasive. Too often, women who are TOLAC candidates are not receiving appropriate counseling – and too often, women are not even being presented the option of a trial of labor, even when staff are immediately available to provide emergency care if needed.
Rupture concerns in perspective
When the NIH consensus development panel reviewed VBAC in 2010, it concluded that TOLAC is a reasonable option for many women with a prior cesarean. The panel found that restricted access to VBAC/TOLAC stemmed from existing practice guidelines and the medical liability climate, and it called upon providers and others to “mitigate or even eliminate” the barriers that women face in finding clinicians and facilities able and willing to offer TOLAC.
ACOG’s 2010 practice bulletin also acknowledged the problem of limited access. ACOG recommended, as it had in an earlier bulletin, that TOLAC-VBAC be undertaken in facilities where staff are immediately available for emergency care. It added, however, that when such resources are not available, the best alternative may be to refer patients to a facility with available resources. Health care providers and insurance carriers “should do all they can to facilitate transfer of care or comanagement in support of a desired TOLAC,” ACOG’s document states.
Why, given such recommendations, are we still falling so short of where we should be?
A number of nonclinical factors are involved, but clearly, as both the NIH and ACOG have stated, the fear of litigation in cases of uterine rupture is a contributing factor. A ruptured uterus is indeed the principal risk associated with TOLAC, and it can have serious sequelae including perinatal death, hypoxic ischemic encephalopathy (HIE), and hysterectomy.
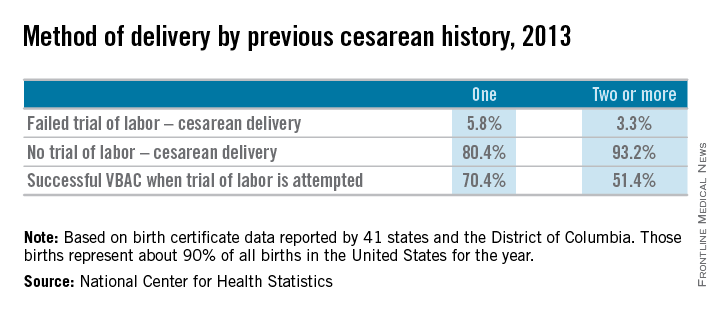
We must appreciate, however, that the absolute rates of uterine rupture and of serious adverse outcomes are quite low. The rupture rate in 2013 among women who underwent TOLAC but ultimately had a repeat cesarean section – the highest-risk group – was 495 per 100,000 live births, according to the CDC. This rate of approximately 0.5% is consistent with the level of risk reported in the literature for several decades.
In one of the two large observational studies done in the United States that have shed light on TOLAC outcomes, the rate of uterine rupture among women who underwent TOLAC was 0.7% for women with a prior low transverse incision, 2.0% for those with a prior low vertical incision, and 0.5% for those with an unknown type of prior incision. Overall, the rate of uterine rupture in this study’s cohort of 17,898 women who underwent TOLAC was 0.7% (N Engl J Med. 2004 Dec 16;351[25]:2581-9). The study was conducted at 19 medical centers belonging to the Eunice Kennedy Shriver National Institute of Child Health and Human Development’s Maternal-Fetal Medical Units (MFMU) Network.
The second large study conducted in the United States – a multicenter observational study in which records of approximately 25,000 women with a prior low-transverse cesarean section were reviewed – also showed rates of uterine rupture less than 1% (Am J Obstet Gynecol. 2005 Nov;193[5]:1656-62).
The attributable risk for perinatal death or HIE at term appears to be 1 per 2,000 TOLAC, according to the MFMU Network study.
Failed trials of labor resulting in repeat cesarean deliveries have consistently been associated with higher morbidity than scheduled repeat cesarean deliveries, with the greatest difference in rates for ruptured uterus. In the first MFMU Network study, there were no cases of uterine rupture among a cohort of 15,801 women who underwent elective repeat cesarean delivery, and in the second multicenter study of 25,000 women, this patient group had a rupture rate of 0.004%.
Yet, as ACOG points out, neither elective repeat cesarean deliveries nor TOLAC are without maternal or neonatal risk. Women who have successful VBAC delivery, on the other hand, have significantly lower morbidity and better outcomes than women who do not attempt labor. Women who undergo VBAC also avoid exposure to the significant risks of repeat cesarean deliveries in the long term.
Research unequivocally shows that the risk of placenta accreta, hysterectomy, hemorrhage, and other serious maternal morbidity increases progressively with each repeat cesarean delivery. Rates of placenta accreta have, in fact, been rising in the United States – a trend that should prompt us to think more about TOLAC.
Moreover, TOLAC is being shown to be a cost-effective strategy. In one analysis, TOLAC in a second pregnancy was cost-effective as long as the chance of VBAC exceeded approximately 74% (Obstet Gynecol. 2001 Jun;97[6]:932-41). More recently, TOLAC was found to be cost-effective across a wide variety of circumstances, including when a woman had a probability of VBAC as low as 43%. The model in this analysis, which used probability estimates from the MFMU Cesarean Registry, took a longer-term view by including probabilities of outcomes throughout a woman’s reproductive life that were contingent upon her initial choice regarding TOLAC (Am J Perinatol. 2013 Jan;30[1]:11-20).
Likelihood of success
Evaluating and discussing the likelihood of success with TOLAC is therefore key to the counseling process. The higher the likelihood of achieving VBAC, the more favorable the risk-benefit ratio will be and the more appealing it will be to consider.
According to one analysis, if a woman undergoing a TOLAC has at least a 60%-70% chance of VBAC, her chance of having major or minor morbidity is no greater than a woman undergoing a planned repeat cesarean delivery (Am J Obstet Gynecol 2009;200:56.e1-e6).
There are several prediction tools available that can be used at the first prenatal visit and in early labor to give a reasonably good estimate of success. One of these tools is available at the MFMU Network website (http://mfmu.bsc.gwu.edu). The tools take into account factors such as prior indication for cesarean delivery; history of vaginal delivery; demographic characteristics such as maternal age and body mass index; the occurrence of spontaneous labor; and cervical status at admission.
Prior vaginal delivery is one of the strongest predictors of a successful TOLAC. Research has consistently shown that women with a prior vaginal delivery – including a vaginal delivery predating an unsuccessful TOLAC – have significantly higher TOLAC success rates than women who did not have any prior vaginal delivery.
The indication for a prior cesarean delivery also clearly affects the likelihood of a successful TOLAC. Women whose first cesarean delivery was performed for a nonrecurring indication, such as breech presentation or low intolerance of labor, have TOLAC success rates that are similar to vaginal delivery rates for nulliparous women. Success rates for these women may exceed 85%. On the other hand, women who had a prior cesarean delivery for cephalopelvic disproportion or failure to progress have been shown to have lower TOLAC success rates ranging from 50%-67%.
Labor induction should be approached cautiously, as women who undergo induction of labor in TOLAC have an increased risk of repeat cesarean delivery. Still, success rates with induction are high. Data from the MFMU Cesarean Registry showed that about 66% of women undergoing induction after one prior cesarean delivery achieved VBAC versus 76% of women entering TOLAC spontaneously (Obstet Gynecol. 2007 Feb;109[2 Pt 1]:262-9). Another study of women undergoing induction after one prior cesarean reported an overall success rate of 78% (Obstet Gynecol. 2004 Mar;103[3]:534-8).
Whether induction specifically increases the risk for uterine rupture in TOLAC, compared with expectant management, is unclear. There also are conflicting data as to whether particular induction methods increase this risk.
Based on available data, ACOG considers induction of labor for either maternal or fetal indications to be an option for women undergoing TOLAC. Oxytocin may be used for induction as well as augmentation, but caution should be exercised at higher doses. While there is no clear dosing threshold for increased risk of rupture, research has suggested that higher doses of oxytocin are best avoided.
The use of prostaglandins is more controversial: Based on evidence from several small studies, ACOG concluded in its 2010 bulletin that misoprostol (prostaglandin E1) for cervical ripening is contraindicated in women undergoing TOLAC. It appears likely that rupture risk increases in patients who received both prostaglandins and oxytocin, so ACOG has advised avoiding their sequential use when prostaglandin E2 is used. This of course limits the options for the practitioner. Therefore, utilizing a Foley catheter followed by pitocin has been an approach advocated in some cases.
Uterine rupture is not predictable, and it is far more difficult to assess an individual’s risk of this complication than it is to assess the likelihood of VBAC. Still, there is value to discussing with the patient whether there are any other modifiers that could potentially influence the risk of rupture.
Since rates of uterine rupture are highest in women with previous classical or T-shaped incision, for example, it is important to try to ascertain what type of incision was previously used. It is widely appreciated that low-transverse uterine incisions are most favorable, but findings are mixed in regard to low-vertical incisions. Some research shows that women with a previous low-vertical incision do not have significantly lower VBAC success rates or higher risks of uterine rupture. TOLAC should therefore not be ruled out in these cases.
Additionally, TOLAC should not be ruled out for women who have had more than one cesarean delivery. Several studies have shown an increased risk of uterine rupture after two prior cesarean deliveries, compared with one, and one meta-analysis suggested a more than twofold increased risk (BJOG. 2010 Jan;117(1):5-19.).
In contrast, an analysis of the MFMU Cesarean Registry found no significant difference in rupture rates in women with one prior cesarean versus multiple prior cesareans (Obstet Gynecol. 2006 Jul;108[1]:12-20.).
It appears, therefore, that even if having more than one prior cesarean section is associated with an increased risk of rupture, the magnitude of this increase is small.
Just as women with a prior vaginal delivery have the highest chance of VBAC success, they also have the lowest rates of rupture among all women undergoing TOLAC.
Patient counseling
We must inform our patients who have had a cesarean section in the past of their options for childbirth in an unbiased manner.
The complications of both TOLAC and elective repeat cesarean section should be discussed, and every attempt should be made to individually assess both the likelihood of a successful VBAC and the comparative risk of maternal and perinatal morbidity. A shared decision-making process should be adopted, and whenever possible, the patient’s preference should be respected. In the end, a woman undergoing TOLAC should be truly motivated to pursue a trial of labor, because there are inherent risks.
One thing I’ve learned from my clinical practice and research on this issue is that the desire to undergo a vaginal delivery is powerful for some women. Many of my patients have self-referred for consultation about TOLAC after their ob.gyn. informed them that their hospital is not equipped, and they should therefore have a scheduled repeat operation. In many cases they discover that TOLAC is an option if they are willing to travel a half-hour or so.
We need to honor this desire and inform our patients of the option, and help facilitate delivery at another nearby hospital when our own facility is not equipped for TOLAC.
Dr. Landon is the Richard L. Meiling Professor and chairman of the department of obstetrics and gynecology at the Ohio State University, Columbus. He served for more than 25 years as Ohio State’s coinvestigator for the National Institutes of Child Health and Human Development Maternal Fetal Medicine Units Network. He reported having no relevant financial disclosures.
The relative safety of vaginal birth after cesarean (VBAC) has been documented in several large-scale studies in the past 15 years, and was affirmed in 2010 through a National Institutes of Health consensus development conference and a practice bulletin from the American College of Obstetricians and Gynecologists. Yet, despite all this research and review, rates of a trial of labor after cesarean (TOLAC) have increased only modestly in the last several years.
Approximately 20% of all births in 2013 in women with a history of one cesarean section involved a trial of labor, according to a recent report from the Centers for Disease Control and Prevention. This represents only a small increase from 2006, when the TOLAC rate had plummeted to approximately 15%.
The limited change is concerning because up to two-thirds of women with a prior cesarean delivery are candidates for a trial of labor, and many of them are excellent candidates. In total, 70% of the women who attempted labor in 2013 after a previous cesarean had successful VBACs, the CDC data shows.
Several European countries have TOLAC rates between 50% and 70%, but in the United States, as evidenced by the recent CDC data, there continues to be an underutilization of attempted VBAC. We must ask ourselves, are women truly able to choose TOLAC, or are they being dissuaded by the health care system?
I believe that the barriers are still pervasive. Too often, women who are TOLAC candidates are not receiving appropriate counseling – and too often, women are not even being presented the option of a trial of labor, even when staff are immediately available to provide emergency care if needed.
Rupture concerns in perspective
When the NIH consensus development panel reviewed VBAC in 2010, it concluded that TOLAC is a reasonable option for many women with a prior cesarean. The panel found that restricted access to VBAC/TOLAC stemmed from existing practice guidelines and the medical liability climate, and it called upon providers and others to “mitigate or even eliminate” the barriers that women face in finding clinicians and facilities able and willing to offer TOLAC.
ACOG’s 2010 practice bulletin also acknowledged the problem of limited access. ACOG recommended, as it had in an earlier bulletin, that TOLAC-VBAC be undertaken in facilities where staff are immediately available for emergency care. It added, however, that when such resources are not available, the best alternative may be to refer patients to a facility with available resources. Health care providers and insurance carriers “should do all they can to facilitate transfer of care or comanagement in support of a desired TOLAC,” ACOG’s document states.
Why, given such recommendations, are we still falling so short of where we should be?
A number of nonclinical factors are involved, but clearly, as both the NIH and ACOG have stated, the fear of litigation in cases of uterine rupture is a contributing factor. A ruptured uterus is indeed the principal risk associated with TOLAC, and it can have serious sequelae including perinatal death, hypoxic ischemic encephalopathy (HIE), and hysterectomy.

We must appreciate, however, that the absolute rates of uterine rupture and of serious adverse outcomes are quite low. The rupture rate in 2013 among women who underwent TOLAC but ultimately had a repeat cesarean section – the highest-risk group – was 495 per 100,000 live births, according to the CDC. This rate of approximately 0.5% is consistent with the level of risk reported in the literature for several decades.
In one of the two large observational studies done in the United States that have shed light on TOLAC outcomes, the rate of uterine rupture among women who underwent TOLAC was 0.7% for women with a prior low transverse incision, 2.0% for those with a prior low vertical incision, and 0.5% for those with an unknown type of prior incision. Overall, the rate of uterine rupture in this study’s cohort of 17,898 women who underwent TOLAC was 0.7% (N Engl J Med. 2004 Dec 16;351[25]:2581-9). The study was conducted at 19 medical centers belonging to the Eunice Kennedy Shriver National Institute of Child Health and Human Development’s Maternal-Fetal Medical Units (MFMU) Network.
The second large study conducted in the United States – a multicenter observational study in which records of approximately 25,000 women with a prior low-transverse cesarean section were reviewed – also showed rates of uterine rupture less than 1% (Am J Obstet Gynecol. 2005 Nov;193[5]:1656-62).
The attributable risk for perinatal death or HIE at term appears to be 1 per 2,000 TOLAC, according to the MFMU Network study.
Failed trials of labor resulting in repeat cesarean deliveries have consistently been associated with higher morbidity than scheduled repeat cesarean deliveries, with the greatest difference in rates for ruptured uterus. In the first MFMU Network study, there were no cases of uterine rupture among a cohort of 15,801 women who underwent elective repeat cesarean delivery, and in the second multicenter study of 25,000 women, this patient group had a rupture rate of 0.004%.
Yet, as ACOG points out, neither elective repeat cesarean deliveries nor TOLAC are without maternal or neonatal risk. Women who have successful VBAC delivery, on the other hand, have significantly lower morbidity and better outcomes than women who do not attempt labor. Women who undergo VBAC also avoid exposure to the significant risks of repeat cesarean deliveries in the long term.
Research unequivocally shows that the risk of placenta accreta, hysterectomy, hemorrhage, and other serious maternal morbidity increases progressively with each repeat cesarean delivery. Rates of placenta accreta have, in fact, been rising in the United States – a trend that should prompt us to think more about TOLAC.
Moreover, TOLAC is being shown to be a cost-effective strategy. In one analysis, TOLAC in a second pregnancy was cost-effective as long as the chance of VBAC exceeded approximately 74% (Obstet Gynecol. 2001 Jun;97[6]:932-41). More recently, TOLAC was found to be cost-effective across a wide variety of circumstances, including when a woman had a probability of VBAC as low as 43%. The model in this analysis, which used probability estimates from the MFMU Cesarean Registry, took a longer-term view by including probabilities of outcomes throughout a woman’s reproductive life that were contingent upon her initial choice regarding TOLAC (Am J Perinatol. 2013 Jan;30[1]:11-20).
Likelihood of success
Evaluating and discussing the likelihood of success with TOLAC is therefore key to the counseling process. The higher the likelihood of achieving VBAC, the more favorable the risk-benefit ratio will be and the more appealing it will be to consider.
According to one analysis, if a woman undergoing a TOLAC has at least a 60%-70% chance of VBAC, her chance of having major or minor morbidity is no greater than a woman undergoing a planned repeat cesarean delivery (Am J Obstet Gynecol 2009;200:56.e1-e6).
There are several prediction tools available that can be used at the first prenatal visit and in early labor to give a reasonably good estimate of success. One of these tools is available at the MFMU Network website (http://mfmu.bsc.gwu.edu). The tools take into account factors such as prior indication for cesarean delivery; history of vaginal delivery; demographic characteristics such as maternal age and body mass index; the occurrence of spontaneous labor; and cervical status at admission.
Prior vaginal delivery is one of the strongest predictors of a successful TOLAC. Research has consistently shown that women with a prior vaginal delivery – including a vaginal delivery predating an unsuccessful TOLAC – have significantly higher TOLAC success rates than women who did not have any prior vaginal delivery.
The indication for a prior cesarean delivery also clearly affects the likelihood of a successful TOLAC. Women whose first cesarean delivery was performed for a nonrecurring indication, such as breech presentation or low intolerance of labor, have TOLAC success rates that are similar to vaginal delivery rates for nulliparous women. Success rates for these women may exceed 85%. On the other hand, women who had a prior cesarean delivery for cephalopelvic disproportion or failure to progress have been shown to have lower TOLAC success rates ranging from 50%-67%.
Labor induction should be approached cautiously, as women who undergo induction of labor in TOLAC have an increased risk of repeat cesarean delivery. Still, success rates with induction are high. Data from the MFMU Cesarean Registry showed that about 66% of women undergoing induction after one prior cesarean delivery achieved VBAC versus 76% of women entering TOLAC spontaneously (Obstet Gynecol. 2007 Feb;109[2 Pt 1]:262-9). Another study of women undergoing induction after one prior cesarean reported an overall success rate of 78% (Obstet Gynecol. 2004 Mar;103[3]:534-8).
Whether induction specifically increases the risk for uterine rupture in TOLAC, compared with expectant management, is unclear. There also are conflicting data as to whether particular induction methods increase this risk.
Based on available data, ACOG considers induction of labor for either maternal or fetal indications to be an option for women undergoing TOLAC. Oxytocin may be used for induction as well as augmentation, but caution should be exercised at higher doses. While there is no clear dosing threshold for increased risk of rupture, research has suggested that higher doses of oxytocin are best avoided.
The use of prostaglandins is more controversial: Based on evidence from several small studies, ACOG concluded in its 2010 bulletin that misoprostol (prostaglandin E1) for cervical ripening is contraindicated in women undergoing TOLAC. It appears likely that rupture risk increases in patients who received both prostaglandins and oxytocin, so ACOG has advised avoiding their sequential use when prostaglandin E2 is used. This of course limits the options for the practitioner. Therefore, utilizing a Foley catheter followed by pitocin has been an approach advocated in some cases.
Uterine rupture is not predictable, and it is far more difficult to assess an individual’s risk of this complication than it is to assess the likelihood of VBAC. Still, there is value to discussing with the patient whether there are any other modifiers that could potentially influence the risk of rupture.
Since rates of uterine rupture are highest in women with previous classical or T-shaped incision, for example, it is important to try to ascertain what type of incision was previously used. It is widely appreciated that low-transverse uterine incisions are most favorable, but findings are mixed in regard to low-vertical incisions. Some research shows that women with a previous low-vertical incision do not have significantly lower VBAC success rates or higher risks of uterine rupture. TOLAC should therefore not be ruled out in these cases.
Additionally, TOLAC should not be ruled out for women who have had more than one cesarean delivery. Several studies have shown an increased risk of uterine rupture after two prior cesarean deliveries, compared with one, and one meta-analysis suggested a more than twofold increased risk (BJOG. 2010 Jan;117(1):5-19.).
In contrast, an analysis of the MFMU Cesarean Registry found no significant difference in rupture rates in women with one prior cesarean versus multiple prior cesareans (Obstet Gynecol. 2006 Jul;108[1]:12-20.).
It appears, therefore, that even if having more than one prior cesarean section is associated with an increased risk of rupture, the magnitude of this increase is small.
Just as women with a prior vaginal delivery have the highest chance of VBAC success, they also have the lowest rates of rupture among all women undergoing TOLAC.
Patient counseling
We must inform our patients who have had a cesarean section in the past of their options for childbirth in an unbiased manner.
The complications of both TOLAC and elective repeat cesarean section should be discussed, and every attempt should be made to individually assess both the likelihood of a successful VBAC and the comparative risk of maternal and perinatal morbidity. A shared decision-making process should be adopted, and whenever possible, the patient’s preference should be respected. In the end, a woman undergoing TOLAC should be truly motivated to pursue a trial of labor, because there are inherent risks.
One thing I’ve learned from my clinical practice and research on this issue is that the desire to undergo a vaginal delivery is powerful for some women. Many of my patients have self-referred for consultation about TOLAC after their ob.gyn. informed them that their hospital is not equipped, and they should therefore have a scheduled repeat operation. In many cases they discover that TOLAC is an option if they are willing to travel a half-hour or so.
We need to honor this desire and inform our patients of the option, and help facilitate delivery at another nearby hospital when our own facility is not equipped for TOLAC.
Dr. Landon is the Richard L. Meiling Professor and chairman of the department of obstetrics and gynecology at the Ohio State University, Columbus. He served for more than 25 years as Ohio State’s coinvestigator for the National Institutes of Child Health and Human Development Maternal Fetal Medicine Units Network. He reported having no relevant financial disclosures.
The relative safety of vaginal birth after cesarean (VBAC) has been documented in several large-scale studies in the past 15 years, and was affirmed in 2010 through a National Institutes of Health consensus development conference and a practice bulletin from the American College of Obstetricians and Gynecologists. Yet, despite all this research and review, rates of a trial of labor after cesarean (TOLAC) have increased only modestly in the last several years.
Approximately 20% of all births in 2013 in women with a history of one cesarean section involved a trial of labor, according to a recent report from the Centers for Disease Control and Prevention. This represents only a small increase from 2006, when the TOLAC rate had plummeted to approximately 15%.
The limited change is concerning because up to two-thirds of women with a prior cesarean delivery are candidates for a trial of labor, and many of them are excellent candidates. In total, 70% of the women who attempted labor in 2013 after a previous cesarean had successful VBACs, the CDC data shows.
Several European countries have TOLAC rates between 50% and 70%, but in the United States, as evidenced by the recent CDC data, there continues to be an underutilization of attempted VBAC. We must ask ourselves, are women truly able to choose TOLAC, or are they being dissuaded by the health care system?
I believe that the barriers are still pervasive. Too often, women who are TOLAC candidates are not receiving appropriate counseling – and too often, women are not even being presented the option of a trial of labor, even when staff are immediately available to provide emergency care if needed.
Rupture concerns in perspective
When the NIH consensus development panel reviewed VBAC in 2010, it concluded that TOLAC is a reasonable option for many women with a prior cesarean. The panel found that restricted access to VBAC/TOLAC stemmed from existing practice guidelines and the medical liability climate, and it called upon providers and others to “mitigate or even eliminate” the barriers that women face in finding clinicians and facilities able and willing to offer TOLAC.
ACOG’s 2010 practice bulletin also acknowledged the problem of limited access. ACOG recommended, as it had in an earlier bulletin, that TOLAC-VBAC be undertaken in facilities where staff are immediately available for emergency care. It added, however, that when such resources are not available, the best alternative may be to refer patients to a facility with available resources. Health care providers and insurance carriers “should do all they can to facilitate transfer of care or comanagement in support of a desired TOLAC,” ACOG’s document states.
Why, given such recommendations, are we still falling so short of where we should be?
A number of nonclinical factors are involved, but clearly, as both the NIH and ACOG have stated, the fear of litigation in cases of uterine rupture is a contributing factor. A ruptured uterus is indeed the principal risk associated with TOLAC, and it can have serious sequelae including perinatal death, hypoxic ischemic encephalopathy (HIE), and hysterectomy.

We must appreciate, however, that the absolute rates of uterine rupture and of serious adverse outcomes are quite low. The rupture rate in 2013 among women who underwent TOLAC but ultimately had a repeat cesarean section – the highest-risk group – was 495 per 100,000 live births, according to the CDC. This rate of approximately 0.5% is consistent with the level of risk reported in the literature for several decades.
In one of the two large observational studies done in the United States that have shed light on TOLAC outcomes, the rate of uterine rupture among women who underwent TOLAC was 0.7% for women with a prior low transverse incision, 2.0% for those with a prior low vertical incision, and 0.5% for those with an unknown type of prior incision. Overall, the rate of uterine rupture in this study’s cohort of 17,898 women who underwent TOLAC was 0.7% (N Engl J Med. 2004 Dec 16;351[25]:2581-9). The study was conducted at 19 medical centers belonging to the Eunice Kennedy Shriver National Institute of Child Health and Human Development’s Maternal-Fetal Medical Units (MFMU) Network.
The second large study conducted in the United States – a multicenter observational study in which records of approximately 25,000 women with a prior low-transverse cesarean section were reviewed – also showed rates of uterine rupture less than 1% (Am J Obstet Gynecol. 2005 Nov;193[5]:1656-62).
The attributable risk for perinatal death or HIE at term appears to be 1 per 2,000 TOLAC, according to the MFMU Network study.
Failed trials of labor resulting in repeat cesarean deliveries have consistently been associated with higher morbidity than scheduled repeat cesarean deliveries, with the greatest difference in rates for ruptured uterus. In the first MFMU Network study, there were no cases of uterine rupture among a cohort of 15,801 women who underwent elective repeat cesarean delivery, and in the second multicenter study of 25,000 women, this patient group had a rupture rate of 0.004%.
Yet, as ACOG points out, neither elective repeat cesarean deliveries nor TOLAC are without maternal or neonatal risk. Women who have successful VBAC delivery, on the other hand, have significantly lower morbidity and better outcomes than women who do not attempt labor. Women who undergo VBAC also avoid exposure to the significant risks of repeat cesarean deliveries in the long term.
Research unequivocally shows that the risk of placenta accreta, hysterectomy, hemorrhage, and other serious maternal morbidity increases progressively with each repeat cesarean delivery. Rates of placenta accreta have, in fact, been rising in the United States – a trend that should prompt us to think more about TOLAC.
Moreover, TOLAC is being shown to be a cost-effective strategy. In one analysis, TOLAC in a second pregnancy was cost-effective as long as the chance of VBAC exceeded approximately 74% (Obstet Gynecol. 2001 Jun;97[6]:932-41). More recently, TOLAC was found to be cost-effective across a wide variety of circumstances, including when a woman had a probability of VBAC as low as 43%. The model in this analysis, which used probability estimates from the MFMU Cesarean Registry, took a longer-term view by including probabilities of outcomes throughout a woman’s reproductive life that were contingent upon her initial choice regarding TOLAC (Am J Perinatol. 2013 Jan;30[1]:11-20).
Likelihood of success
Evaluating and discussing the likelihood of success with TOLAC is therefore key to the counseling process. The higher the likelihood of achieving VBAC, the more favorable the risk-benefit ratio will be and the more appealing it will be to consider.
According to one analysis, if a woman undergoing a TOLAC has at least a 60%-70% chance of VBAC, her chance of having major or minor morbidity is no greater than a woman undergoing a planned repeat cesarean delivery (Am J Obstet Gynecol 2009;200:56.e1-e6).
There are several prediction tools available that can be used at the first prenatal visit and in early labor to give a reasonably good estimate of success. One of these tools is available at the MFMU Network website (http://mfmu.bsc.gwu.edu). The tools take into account factors such as prior indication for cesarean delivery; history of vaginal delivery; demographic characteristics such as maternal age and body mass index; the occurrence of spontaneous labor; and cervical status at admission.
Prior vaginal delivery is one of the strongest predictors of a successful TOLAC. Research has consistently shown that women with a prior vaginal delivery – including a vaginal delivery predating an unsuccessful TOLAC – have significantly higher TOLAC success rates than women who did not have any prior vaginal delivery.
The indication for a prior cesarean delivery also clearly affects the likelihood of a successful TOLAC. Women whose first cesarean delivery was performed for a nonrecurring indication, such as breech presentation or low intolerance of labor, have TOLAC success rates that are similar to vaginal delivery rates for nulliparous women. Success rates for these women may exceed 85%. On the other hand, women who had a prior cesarean delivery for cephalopelvic disproportion or failure to progress have been shown to have lower TOLAC success rates ranging from 50%-67%.
Labor induction should be approached cautiously, as women who undergo induction of labor in TOLAC have an increased risk of repeat cesarean delivery. Still, success rates with induction are high. Data from the MFMU Cesarean Registry showed that about 66% of women undergoing induction after one prior cesarean delivery achieved VBAC versus 76% of women entering TOLAC spontaneously (Obstet Gynecol. 2007 Feb;109[2 Pt 1]:262-9). Another study of women undergoing induction after one prior cesarean reported an overall success rate of 78% (Obstet Gynecol. 2004 Mar;103[3]:534-8).
Whether induction specifically increases the risk for uterine rupture in TOLAC, compared with expectant management, is unclear. There also are conflicting data as to whether particular induction methods increase this risk.
Based on available data, ACOG considers induction of labor for either maternal or fetal indications to be an option for women undergoing TOLAC. Oxytocin may be used for induction as well as augmentation, but caution should be exercised at higher doses. While there is no clear dosing threshold for increased risk of rupture, research has suggested that higher doses of oxytocin are best avoided.
The use of prostaglandins is more controversial: Based on evidence from several small studies, ACOG concluded in its 2010 bulletin that misoprostol (prostaglandin E1) for cervical ripening is contraindicated in women undergoing TOLAC. It appears likely that rupture risk increases in patients who received both prostaglandins and oxytocin, so ACOG has advised avoiding their sequential use when prostaglandin E2 is used. This of course limits the options for the practitioner. Therefore, utilizing a Foley catheter followed by pitocin has been an approach advocated in some cases.
Uterine rupture is not predictable, and it is far more difficult to assess an individual’s risk of this complication than it is to assess the likelihood of VBAC. Still, there is value to discussing with the patient whether there are any other modifiers that could potentially influence the risk of rupture.
Since rates of uterine rupture are highest in women with previous classical or T-shaped incision, for example, it is important to try to ascertain what type of incision was previously used. It is widely appreciated that low-transverse uterine incisions are most favorable, but findings are mixed in regard to low-vertical incisions. Some research shows that women with a previous low-vertical incision do not have significantly lower VBAC success rates or higher risks of uterine rupture. TOLAC should therefore not be ruled out in these cases.
Additionally, TOLAC should not be ruled out for women who have had more than one cesarean delivery. Several studies have shown an increased risk of uterine rupture after two prior cesarean deliveries, compared with one, and one meta-analysis suggested a more than twofold increased risk (BJOG. 2010 Jan;117(1):5-19.).
In contrast, an analysis of the MFMU Cesarean Registry found no significant difference in rupture rates in women with one prior cesarean versus multiple prior cesareans (Obstet Gynecol. 2006 Jul;108[1]:12-20.).
It appears, therefore, that even if having more than one prior cesarean section is associated with an increased risk of rupture, the magnitude of this increase is small.
Just as women with a prior vaginal delivery have the highest chance of VBAC success, they also have the lowest rates of rupture among all women undergoing TOLAC.
Patient counseling
We must inform our patients who have had a cesarean section in the past of their options for childbirth in an unbiased manner.
The complications of both TOLAC and elective repeat cesarean section should be discussed, and every attempt should be made to individually assess both the likelihood of a successful VBAC and the comparative risk of maternal and perinatal morbidity. A shared decision-making process should be adopted, and whenever possible, the patient’s preference should be respected. In the end, a woman undergoing TOLAC should be truly motivated to pursue a trial of labor, because there are inherent risks.
One thing I’ve learned from my clinical practice and research on this issue is that the desire to undergo a vaginal delivery is powerful for some women. Many of my patients have self-referred for consultation about TOLAC after their ob.gyn. informed them that their hospital is not equipped, and they should therefore have a scheduled repeat operation. In many cases they discover that TOLAC is an option if they are willing to travel a half-hour or so.
We need to honor this desire and inform our patients of the option, and help facilitate delivery at another nearby hospital when our own facility is not equipped for TOLAC.
Dr. Landon is the Richard L. Meiling Professor and chairman of the department of obstetrics and gynecology at the Ohio State University, Columbus. He served for more than 25 years as Ohio State’s coinvestigator for the National Institutes of Child Health and Human Development Maternal Fetal Medicine Units Network. He reported having no relevant financial disclosures.
Repeat Tdap Vaccination Is Safe in Pregnancy
Receiving the tetanus, diphtheria, acellular pertussis vaccine during pregnancy does not increase the risk of adverse events for mother or baby, even if the mother has previously received the vaccine within the past 2 years, according to a report published Oct. 20 in JAMA.
Women should receive a Tdap vaccination during each pregnancy – preferably in the third trimester – so that the fetus receives some maternal antibodies against pertussis for limited protection until the first DTaP vaccine at age 2 months, according to 2012 recommendation from the Advisory Committee on Immunization Practices (ACIP). Yet previous research has not clarified the safety of repeated doses of maternal Tdap immunization.
“These findings suggest that relatively recent receipt of a prior tetanus-containing vaccination does not increase risk after Tdap vaccination in pregnancy,” wrote Dr. Lakshmi Sukumaran of the Centers for Disease Control and Prevention Immunization Safety Office, and colleagues. “Our findings should reassure patients and clinicians who might be hesitant to give Tdap vaccine to pregnant women who recently received a Tdap or other tetanus-containing vaccination.”
In the retrospective cohort study involving pregnancies between Jan. 1, 2007, through Nov. 15, 2013, the researchers examined data from 29,155 pregnant women, aged 14-49, who enrolled in seven Vaccine Safety Datalink sites in California, Colorado, Minnesota, Oregon, Washington, and Wisconsin.
The researchers compared the 49% of women who had received a tetanus-containing vaccine more than 5 years before their current pregnancy (controls) with the 17% of women who had received a tetanus-containing vaccine less than 2 years before, and the 34% of women who had been vaccinated 2-5 years earlier. More than two-thirds of the women (67.4%) received the Tdap during the third trimester, compared with 27.5% in the second trimester, and 5.1% in the first trimester (JAMA. 2015;314[15]:1581-7. doi: 10.1001/jama.2015.12790).
The researchers evaluated several adverse events including fever, allergy, and local reactions in mothers in the week following immunization, as well as preterm birth, low birth weight and small for gestational age in newborns. Local reactions included limb pain, limb swelling, cellulitis, lymphadenitis, and Arthus reaction. The researchers also looked for Guillain-Barré syndrome through day 42 after vaccination.
Few acute adverse events occurred after vaccination, and no statistically significant differences occurred across the three groups in terms of fever, allergic reactions, and local reactions.
Women who had received a tetanus-containing vaccine less than 2 years earlier had fevers at a rate of 2.1 per 10,000 women, compared with 3.5 per 10,000 women among controls (adjusted relative rate: 0.66; P = .70). Similarly, allergic reactions occurred at a rate of 2.1 per 10,000 women for those who had a vaccine less than 2 years prior, compared to 1 per 10,000 women who had the vaccine 2-5 years earlier, and 1.4 per 10,000 women among controls.
Local reactions occurred at a rate of 4.2, 7.0 and 11.2 per 10,000 women for those who had the vaccine less than 2 years prior, between 2-5 years prior, and more than 5 years prior, respectively. Preterm delivery rates, low birth weight, and small for gestation age were all similar across the three groups.
The Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases funded the study. Dr. Sukumaran reported receiving research support from the National Institutes of Health. The other researchers reported receiving research support from GlaxoSmithKline, Sanofi Pasteur, Merck, Pfizer, Nuron Biotech, MedImmune, Novartis, and Protein Science.
Receiving the tetanus, diphtheria, acellular pertussis vaccine during pregnancy does not increase the risk of adverse events for mother or baby, even if the mother has previously received the vaccine within the past 2 years, according to a report published Oct. 20 in JAMA.
Women should receive a Tdap vaccination during each pregnancy – preferably in the third trimester – so that the fetus receives some maternal antibodies against pertussis for limited protection until the first DTaP vaccine at age 2 months, according to 2012 recommendation from the Advisory Committee on Immunization Practices (ACIP). Yet previous research has not clarified the safety of repeated doses of maternal Tdap immunization.
“These findings suggest that relatively recent receipt of a prior tetanus-containing vaccination does not increase risk after Tdap vaccination in pregnancy,” wrote Dr. Lakshmi Sukumaran of the Centers for Disease Control and Prevention Immunization Safety Office, and colleagues. “Our findings should reassure patients and clinicians who might be hesitant to give Tdap vaccine to pregnant women who recently received a Tdap or other tetanus-containing vaccination.”
In the retrospective cohort study involving pregnancies between Jan. 1, 2007, through Nov. 15, 2013, the researchers examined data from 29,155 pregnant women, aged 14-49, who enrolled in seven Vaccine Safety Datalink sites in California, Colorado, Minnesota, Oregon, Washington, and Wisconsin.
The researchers compared the 49% of women who had received a tetanus-containing vaccine more than 5 years before their current pregnancy (controls) with the 17% of women who had received a tetanus-containing vaccine less than 2 years before, and the 34% of women who had been vaccinated 2-5 years earlier. More than two-thirds of the women (67.4%) received the Tdap during the third trimester, compared with 27.5% in the second trimester, and 5.1% in the first trimester (JAMA. 2015;314[15]:1581-7. doi: 10.1001/jama.2015.12790).
The researchers evaluated several adverse events including fever, allergy, and local reactions in mothers in the week following immunization, as well as preterm birth, low birth weight and small for gestational age in newborns. Local reactions included limb pain, limb swelling, cellulitis, lymphadenitis, and Arthus reaction. The researchers also looked for Guillain-Barré syndrome through day 42 after vaccination.
Few acute adverse events occurred after vaccination, and no statistically significant differences occurred across the three groups in terms of fever, allergic reactions, and local reactions.
Women who had received a tetanus-containing vaccine less than 2 years earlier had fevers at a rate of 2.1 per 10,000 women, compared with 3.5 per 10,000 women among controls (adjusted relative rate: 0.66; P = .70). Similarly, allergic reactions occurred at a rate of 2.1 per 10,000 women for those who had a vaccine less than 2 years prior, compared to 1 per 10,000 women who had the vaccine 2-5 years earlier, and 1.4 per 10,000 women among controls.
Local reactions occurred at a rate of 4.2, 7.0 and 11.2 per 10,000 women for those who had the vaccine less than 2 years prior, between 2-5 years prior, and more than 5 years prior, respectively. Preterm delivery rates, low birth weight, and small for gestation age were all similar across the three groups.
The Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases funded the study. Dr. Sukumaran reported receiving research support from the National Institutes of Health. The other researchers reported receiving research support from GlaxoSmithKline, Sanofi Pasteur, Merck, Pfizer, Nuron Biotech, MedImmune, Novartis, and Protein Science.
Receiving the tetanus, diphtheria, acellular pertussis vaccine during pregnancy does not increase the risk of adverse events for mother or baby, even if the mother has previously received the vaccine within the past 2 years, according to a report published Oct. 20 in JAMA.
Women should receive a Tdap vaccination during each pregnancy – preferably in the third trimester – so that the fetus receives some maternal antibodies against pertussis for limited protection until the first DTaP vaccine at age 2 months, according to 2012 recommendation from the Advisory Committee on Immunization Practices (ACIP). Yet previous research has not clarified the safety of repeated doses of maternal Tdap immunization.
“These findings suggest that relatively recent receipt of a prior tetanus-containing vaccination does not increase risk after Tdap vaccination in pregnancy,” wrote Dr. Lakshmi Sukumaran of the Centers for Disease Control and Prevention Immunization Safety Office, and colleagues. “Our findings should reassure patients and clinicians who might be hesitant to give Tdap vaccine to pregnant women who recently received a Tdap or other tetanus-containing vaccination.”
In the retrospective cohort study involving pregnancies between Jan. 1, 2007, through Nov. 15, 2013, the researchers examined data from 29,155 pregnant women, aged 14-49, who enrolled in seven Vaccine Safety Datalink sites in California, Colorado, Minnesota, Oregon, Washington, and Wisconsin.
The researchers compared the 49% of women who had received a tetanus-containing vaccine more than 5 years before their current pregnancy (controls) with the 17% of women who had received a tetanus-containing vaccine less than 2 years before, and the 34% of women who had been vaccinated 2-5 years earlier. More than two-thirds of the women (67.4%) received the Tdap during the third trimester, compared with 27.5% in the second trimester, and 5.1% in the first trimester (JAMA. 2015;314[15]:1581-7. doi: 10.1001/jama.2015.12790).
The researchers evaluated several adverse events including fever, allergy, and local reactions in mothers in the week following immunization, as well as preterm birth, low birth weight and small for gestational age in newborns. Local reactions included limb pain, limb swelling, cellulitis, lymphadenitis, and Arthus reaction. The researchers also looked for Guillain-Barré syndrome through day 42 after vaccination.
Few acute adverse events occurred after vaccination, and no statistically significant differences occurred across the three groups in terms of fever, allergic reactions, and local reactions.
Women who had received a tetanus-containing vaccine less than 2 years earlier had fevers at a rate of 2.1 per 10,000 women, compared with 3.5 per 10,000 women among controls (adjusted relative rate: 0.66; P = .70). Similarly, allergic reactions occurred at a rate of 2.1 per 10,000 women for those who had a vaccine less than 2 years prior, compared to 1 per 10,000 women who had the vaccine 2-5 years earlier, and 1.4 per 10,000 women among controls.
Local reactions occurred at a rate of 4.2, 7.0 and 11.2 per 10,000 women for those who had the vaccine less than 2 years prior, between 2-5 years prior, and more than 5 years prior, respectively. Preterm delivery rates, low birth weight, and small for gestation age were all similar across the three groups.
The Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases funded the study. Dr. Sukumaran reported receiving research support from the National Institutes of Health. The other researchers reported receiving research support from GlaxoSmithKline, Sanofi Pasteur, Merck, Pfizer, Nuron Biotech, MedImmune, Novartis, and Protein Science.
FROM JAMA
Repeat Tdap vaccination is safe in pregnancy
Receiving the tetanus, diphtheria, acellular pertussis vaccine during pregnancy does not increase the risk of adverse events for mother or baby, even if the mother has previously received the vaccine within the past 2 years, according to a report published Oct. 20 in JAMA.
Women should receive a Tdap vaccination during each pregnancy – preferably in the third trimester – so that the fetus receives some maternal antibodies against pertussis for limited protection until the first DTaP vaccine at age 2 months, according to 2012 recommendation from the Advisory Committee on Immunization Practices (ACIP). Yet previous research has not clarified the safety of repeated doses of maternal Tdap immunization.
“These findings suggest that relatively recent receipt of a prior tetanus-containing vaccination does not increase risk after Tdap vaccination in pregnancy,” wrote Dr. Lakshmi Sukumaran of the Centers for Disease Control and Prevention Immunization Safety Office, and colleagues. “Our findings should reassure patients and clinicians who might be hesitant to give Tdap vaccine to pregnant women who recently received a Tdap or other tetanus-containing vaccination.”
In the retrospective cohort study involving pregnancies between Jan. 1, 2007, through Nov. 15, 2013, the researchers examined data from 29,155 pregnant women, aged 14-49, who enrolled in seven Vaccine Safety Datalink sites in California, Colorado, Minnesota, Oregon, Washington, and Wisconsin.
The researchers compared the 49% of women who had received a tetanus-containing vaccine more than 5 years before their current pregnancy (controls) with the 17% of women who had received a tetanus-containing vaccine less than 2 years before, and the 34% of women who had been vaccinated 2-5 years earlier. More than two-thirds of the women (67.4%) received the Tdap during the third trimester, compared with 27.5% in the second trimester, and 5.1% in the first trimester (JAMA. 2015;314[15]:1581-7. doi: 10.1001/jama.2015.12790).
The researchers evaluated several adverse events including fever, allergy, and local reactions in mothers in the week following immunization, as well as preterm birth, low birth weight and small for gestational age in newborns. Local reactions included limb pain, limb swelling, cellulitis, lymphadenitis, and Arthus reaction. The researchers also looked for Guillain-Barré syndrome through day 42 after vaccination.
Few acute adverse events occurred after vaccination, and no statistically significant differences occurred across the three groups in terms of fever, allergic reactions, and local reactions.
Women who had received a tetanus-containing vaccine less than 2 years earlier had fevers at a rate of 2.1 per 10,000 women, compared with 3.5 per 10,000 women among controls (adjusted relative rate: 0.66; P = .70). Similarly, allergic reactions occurred at a rate of 2.1 per 10,000 women for those who had a vaccine less than 2 years prior, compared to 1 per 10,000 women who had the vaccine 2-5 years earlier, and 1.4 per 10,000 women among controls.
Local reactions occurred at a rate of 4.2, 7.0 and 11.2 per 10,000 women for those who had the vaccine less than 2 years prior, between 2-5 years prior, and more than 5 years prior, respectively. Preterm delivery rates, low birth weight, and small for gestation age were all similar across the three groups.
The Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases funded the study. Dr. Sukumaran reported receiving research support from the National Institutes of Health. The other researchers reported receiving research support from GlaxoSmithKline, Sanofi Pasteur, Merck, Pfizer, Nuron Biotech, MedImmune, Novartis, and Protein Science.
Receiving the tetanus, diphtheria, acellular pertussis vaccine during pregnancy does not increase the risk of adverse events for mother or baby, even if the mother has previously received the vaccine within the past 2 years, according to a report published Oct. 20 in JAMA.
Women should receive a Tdap vaccination during each pregnancy – preferably in the third trimester – so that the fetus receives some maternal antibodies against pertussis for limited protection until the first DTaP vaccine at age 2 months, according to 2012 recommendation from the Advisory Committee on Immunization Practices (ACIP). Yet previous research has not clarified the safety of repeated doses of maternal Tdap immunization.
“These findings suggest that relatively recent receipt of a prior tetanus-containing vaccination does not increase risk after Tdap vaccination in pregnancy,” wrote Dr. Lakshmi Sukumaran of the Centers for Disease Control and Prevention Immunization Safety Office, and colleagues. “Our findings should reassure patients and clinicians who might be hesitant to give Tdap vaccine to pregnant women who recently received a Tdap or other tetanus-containing vaccination.”
In the retrospective cohort study involving pregnancies between Jan. 1, 2007, through Nov. 15, 2013, the researchers examined data from 29,155 pregnant women, aged 14-49, who enrolled in seven Vaccine Safety Datalink sites in California, Colorado, Minnesota, Oregon, Washington, and Wisconsin.
The researchers compared the 49% of women who had received a tetanus-containing vaccine more than 5 years before their current pregnancy (controls) with the 17% of women who had received a tetanus-containing vaccine less than 2 years before, and the 34% of women who had been vaccinated 2-5 years earlier. More than two-thirds of the women (67.4%) received the Tdap during the third trimester, compared with 27.5% in the second trimester, and 5.1% in the first trimester (JAMA. 2015;314[15]:1581-7. doi: 10.1001/jama.2015.12790).
The researchers evaluated several adverse events including fever, allergy, and local reactions in mothers in the week following immunization, as well as preterm birth, low birth weight and small for gestational age in newborns. Local reactions included limb pain, limb swelling, cellulitis, lymphadenitis, and Arthus reaction. The researchers also looked for Guillain-Barré syndrome through day 42 after vaccination.
Few acute adverse events occurred after vaccination, and no statistically significant differences occurred across the three groups in terms of fever, allergic reactions, and local reactions.
Women who had received a tetanus-containing vaccine less than 2 years earlier had fevers at a rate of 2.1 per 10,000 women, compared with 3.5 per 10,000 women among controls (adjusted relative rate: 0.66; P = .70). Similarly, allergic reactions occurred at a rate of 2.1 per 10,000 women for those who had a vaccine less than 2 years prior, compared to 1 per 10,000 women who had the vaccine 2-5 years earlier, and 1.4 per 10,000 women among controls.
Local reactions occurred at a rate of 4.2, 7.0 and 11.2 per 10,000 women for those who had the vaccine less than 2 years prior, between 2-5 years prior, and more than 5 years prior, respectively. Preterm delivery rates, low birth weight, and small for gestation age were all similar across the three groups.
The Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases funded the study. Dr. Sukumaran reported receiving research support from the National Institutes of Health. The other researchers reported receiving research support from GlaxoSmithKline, Sanofi Pasteur, Merck, Pfizer, Nuron Biotech, MedImmune, Novartis, and Protein Science.
Receiving the tetanus, diphtheria, acellular pertussis vaccine during pregnancy does not increase the risk of adverse events for mother or baby, even if the mother has previously received the vaccine within the past 2 years, according to a report published Oct. 20 in JAMA.
Women should receive a Tdap vaccination during each pregnancy – preferably in the third trimester – so that the fetus receives some maternal antibodies against pertussis for limited protection until the first DTaP vaccine at age 2 months, according to 2012 recommendation from the Advisory Committee on Immunization Practices (ACIP). Yet previous research has not clarified the safety of repeated doses of maternal Tdap immunization.
“These findings suggest that relatively recent receipt of a prior tetanus-containing vaccination does not increase risk after Tdap vaccination in pregnancy,” wrote Dr. Lakshmi Sukumaran of the Centers for Disease Control and Prevention Immunization Safety Office, and colleagues. “Our findings should reassure patients and clinicians who might be hesitant to give Tdap vaccine to pregnant women who recently received a Tdap or other tetanus-containing vaccination.”
In the retrospective cohort study involving pregnancies between Jan. 1, 2007, through Nov. 15, 2013, the researchers examined data from 29,155 pregnant women, aged 14-49, who enrolled in seven Vaccine Safety Datalink sites in California, Colorado, Minnesota, Oregon, Washington, and Wisconsin.
The researchers compared the 49% of women who had received a tetanus-containing vaccine more than 5 years before their current pregnancy (controls) with the 17% of women who had received a tetanus-containing vaccine less than 2 years before, and the 34% of women who had been vaccinated 2-5 years earlier. More than two-thirds of the women (67.4%) received the Tdap during the third trimester, compared with 27.5% in the second trimester, and 5.1% in the first trimester (JAMA. 2015;314[15]:1581-7. doi: 10.1001/jama.2015.12790).
The researchers evaluated several adverse events including fever, allergy, and local reactions in mothers in the week following immunization, as well as preterm birth, low birth weight and small for gestational age in newborns. Local reactions included limb pain, limb swelling, cellulitis, lymphadenitis, and Arthus reaction. The researchers also looked for Guillain-Barré syndrome through day 42 after vaccination.
Few acute adverse events occurred after vaccination, and no statistically significant differences occurred across the three groups in terms of fever, allergic reactions, and local reactions.
Women who had received a tetanus-containing vaccine less than 2 years earlier had fevers at a rate of 2.1 per 10,000 women, compared with 3.5 per 10,000 women among controls (adjusted relative rate: 0.66; P = .70). Similarly, allergic reactions occurred at a rate of 2.1 per 10,000 women for those who had a vaccine less than 2 years prior, compared to 1 per 10,000 women who had the vaccine 2-5 years earlier, and 1.4 per 10,000 women among controls.
Local reactions occurred at a rate of 4.2, 7.0 and 11.2 per 10,000 women for those who had the vaccine less than 2 years prior, between 2-5 years prior, and more than 5 years prior, respectively. Preterm delivery rates, low birth weight, and small for gestation age were all similar across the three groups.
The Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases funded the study. Dr. Sukumaran reported receiving research support from the National Institutes of Health. The other researchers reported receiving research support from GlaxoSmithKline, Sanofi Pasteur, Merck, Pfizer, Nuron Biotech, MedImmune, Novartis, and Protein Science.
FROM JAMA
Key clinical point: Tdap vaccination in multiple pregnancies does not increase risk of adverse events.
Major finding: No significant differences in maternal or neonatal adverse outcomes were found between mothers receiving a tetanus-containing vaccine more than 5 years prior to pregnancy compared with pregnant women receiving one within the previous 2 years.
Data source: A retrospective cohort study of the period from Jan. 1, 2007, through Nov. 15, 2013, involving 29,155 pregnant women, aged 14-49, enrolled in seven Vaccine Safety Datalink sites.
Disclosures: The Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases funded the study. Dr. Sukumaran reported receiving research support from the National Institutes of Health. The other researchers reported receiving research support from GlaxoSmithKline, Sanofi Pasteur, Merck, Pfizer, Nuron Biotech, MedImmune, Novartis, and Protein Science.
More than half of hospitals exceed cesarean delivery benchmark
More than 60% of hospitals have cesarean delivery rates that are higher than national benchmarks, according to a new report from the Leapfrog Group.
The Leapfrog Group, a national quality organization, surveyed 1,122 hospitals around the country to determine how many institutions were hitting the target rate of no more than 23.9% C-sections per year. The fact that less than half of U.S. hospitals are meeting the benchmark rate is troubling, said Leapfrog Group president and CEO, Leah Binder.
“This means that far too many women are undergoing a major abdominal surgery without medical necessity – with all the risks that any surgery entails,” Ms. Binder said in a statement, adding that the number of hospitals that did not report their cesarean rates is another cause for concern among patients and providers.
Rural hospitals performed best in the survey, with nearly half of hospitals performing cesareans at the target rate or less frequently. Utah and Idaho, in particular, had statewide cesarean rates lower than 20%. However, only about 37% of urban hospitals met the benchmark rate.
The Leapfrog survey used the Nulliparous Term Singleton Vertex Cesarean Section Rate measure, which identifies pregnancies that are unlikely to need surgical intervention during labor. The measure has been endorsed by the Joint Commission, the National Quality Forum, and the Centers for Medicare & Medicaid Services. The Leapfrog Group’s target cesarean rate of 23.9% comes from the Healthy People 2020 initiative.
“This is really about how well we – as doctors, nurses, midwives, and hospitals – support labor,” Dr. Elliott Main, chair of Leapfrog’s Maternity Care Expert Panel, said in a statement. “Hospital staff that support labor appropriately and are sensitive to families’ birth plans are shown to have lower C-section rates overall. If we want to improve this rate across the board, then hospitals must hold themselves to this standard to ensure safe short- and long-term outcomes for both mom and baby.”
More than 60% of hospitals have cesarean delivery rates that are higher than national benchmarks, according to a new report from the Leapfrog Group.
The Leapfrog Group, a national quality organization, surveyed 1,122 hospitals around the country to determine how many institutions were hitting the target rate of no more than 23.9% C-sections per year. The fact that less than half of U.S. hospitals are meeting the benchmark rate is troubling, said Leapfrog Group president and CEO, Leah Binder.
“This means that far too many women are undergoing a major abdominal surgery without medical necessity – with all the risks that any surgery entails,” Ms. Binder said in a statement, adding that the number of hospitals that did not report their cesarean rates is another cause for concern among patients and providers.
Rural hospitals performed best in the survey, with nearly half of hospitals performing cesareans at the target rate or less frequently. Utah and Idaho, in particular, had statewide cesarean rates lower than 20%. However, only about 37% of urban hospitals met the benchmark rate.
The Leapfrog survey used the Nulliparous Term Singleton Vertex Cesarean Section Rate measure, which identifies pregnancies that are unlikely to need surgical intervention during labor. The measure has been endorsed by the Joint Commission, the National Quality Forum, and the Centers for Medicare & Medicaid Services. The Leapfrog Group’s target cesarean rate of 23.9% comes from the Healthy People 2020 initiative.
“This is really about how well we – as doctors, nurses, midwives, and hospitals – support labor,” Dr. Elliott Main, chair of Leapfrog’s Maternity Care Expert Panel, said in a statement. “Hospital staff that support labor appropriately and are sensitive to families’ birth plans are shown to have lower C-section rates overall. If we want to improve this rate across the board, then hospitals must hold themselves to this standard to ensure safe short- and long-term outcomes for both mom and baby.”
More than 60% of hospitals have cesarean delivery rates that are higher than national benchmarks, according to a new report from the Leapfrog Group.
The Leapfrog Group, a national quality organization, surveyed 1,122 hospitals around the country to determine how many institutions were hitting the target rate of no more than 23.9% C-sections per year. The fact that less than half of U.S. hospitals are meeting the benchmark rate is troubling, said Leapfrog Group president and CEO, Leah Binder.
“This means that far too many women are undergoing a major abdominal surgery without medical necessity – with all the risks that any surgery entails,” Ms. Binder said in a statement, adding that the number of hospitals that did not report their cesarean rates is another cause for concern among patients and providers.
Rural hospitals performed best in the survey, with nearly half of hospitals performing cesareans at the target rate or less frequently. Utah and Idaho, in particular, had statewide cesarean rates lower than 20%. However, only about 37% of urban hospitals met the benchmark rate.
The Leapfrog survey used the Nulliparous Term Singleton Vertex Cesarean Section Rate measure, which identifies pregnancies that are unlikely to need surgical intervention during labor. The measure has been endorsed by the Joint Commission, the National Quality Forum, and the Centers for Medicare & Medicaid Services. The Leapfrog Group’s target cesarean rate of 23.9% comes from the Healthy People 2020 initiative.
“This is really about how well we – as doctors, nurses, midwives, and hospitals – support labor,” Dr. Elliott Main, chair of Leapfrog’s Maternity Care Expert Panel, said in a statement. “Hospital staff that support labor appropriately and are sensitive to families’ birth plans are shown to have lower C-section rates overall. If we want to improve this rate across the board, then hospitals must hold themselves to this standard to ensure safe short- and long-term outcomes for both mom and baby.”