User login
Screening for anxiety in young children
On April 12, 2022, the U.S. Preventive Services Task Force released the draft of a recommendation statement titled Screening for Anxiety in Children and Adolescents. Based on their observation that 7.8% of children and adolescents have a current anxiety disorder and their analysis of the magnitude of the net benefit, the Task Force plans on recommending that children ages 8-18 years be screened for the condition. However, the group could not find evidence to support screening for children 7 years and younger.
Over more than 4 decades of general pediatric practice, it became obvious to me that anxiety was driving a high percentage of my office visits. Most often in young children it was parental anxiety that was prompting the phone call or office visit. In older childhood and adolescence it was patient anxiety that began to play a larger role.
Over the last 2 decades the level of anxiety in all age groups has seemed to increase. How large a role the events of Sept. 11, 2001, and other terrorist attacks were playing in this phenomenon is unclear to me. However, I suspect they were significant. More recently the pandemic and the failure of both political parties to forge a working arrangement have fueled even more anxiety in many demographic segments. It may be safe to say that everyone is anxious to one degree or another.
Broad-based anxiety in the general population and the incidence of anxiety disorders severe enough to disrupt a child’s life are certainly two different kettles of fish. However, the factors that have raised the level of anxiety across all age groups certainly hasn’t made things any easier for the child who has inherited or developed an anxiety disorder.
Glancing at the 600-page evidence synthesis that accompanies the task force’s report it is clear that they have taken their challenge seriously. However, I wonder whether looking at the 7-and-under age group with a different lens might have resulted in the inclusion of younger children in their recommendation.
I understand that to support their recommendations the U.S. Preventive Services Task Forces must rely on data from peer-reviewed studies that have looked at quantifiable outcomes. However, I suspect the task force would agree that its recommendations shouldn’t prevent the rest of us from using our own observations and intuition when deciding whether to selectively screen our younger patients for anxiety disorders.
Although it may not generate a measurable data point, providing the parents of a 5-year-old whose troubling behavior is in part the result of an anxiety disorder is invaluable. Do we need to screen all 5-year-olds? The task force says probably not given the current state of our knowledge and I agree. But, the fact that almost 8% of the pediatric population carries the diagnosis and my anecdotal observations suggest that as pediatricians we should be learning more about anxiety disorders and their wide variety of presentations. Then we should selectively screen more of our patients. In fact, I suspect we might help our patients and ourselves by questioning more parents about their own mental health histories even before we have any inkling that their child has a problem. While the degree to which anxiety disorders are inheritable and the exact mechanism is far from clear, I think this history might be a valuable piece of information to learn as early as the prenatal get-acquainted visit. A simple question to a new or expecting parent about what worries them most about becoming a parent would be a good opener. Your reassurance that you expect parents to be worried and welcome hearing about their concerns should be a step in building a strong foundation for a family-provider relationship.
Anxiety happens and unfortunately so do anxiety disorders. We need to be doing a better job of acknowledging and responding to these two realities.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
*This column was updated on 5/4/2022.
On April 12, 2022, the U.S. Preventive Services Task Force released the draft of a recommendation statement titled Screening for Anxiety in Children and Adolescents. Based on their observation that 7.8% of children and adolescents have a current anxiety disorder and their analysis of the magnitude of the net benefit, the Task Force plans on recommending that children ages 8-18 years be screened for the condition. However, the group could not find evidence to support screening for children 7 years and younger.
Over more than 4 decades of general pediatric practice, it became obvious to me that anxiety was driving a high percentage of my office visits. Most often in young children it was parental anxiety that was prompting the phone call or office visit. In older childhood and adolescence it was patient anxiety that began to play a larger role.
Over the last 2 decades the level of anxiety in all age groups has seemed to increase. How large a role the events of Sept. 11, 2001, and other terrorist attacks were playing in this phenomenon is unclear to me. However, I suspect they were significant. More recently the pandemic and the failure of both political parties to forge a working arrangement have fueled even more anxiety in many demographic segments. It may be safe to say that everyone is anxious to one degree or another.
Broad-based anxiety in the general population and the incidence of anxiety disorders severe enough to disrupt a child’s life are certainly two different kettles of fish. However, the factors that have raised the level of anxiety across all age groups certainly hasn’t made things any easier for the child who has inherited or developed an anxiety disorder.
Glancing at the 600-page evidence synthesis that accompanies the task force’s report it is clear that they have taken their challenge seriously. However, I wonder whether looking at the 7-and-under age group with a different lens might have resulted in the inclusion of younger children in their recommendation.
I understand that to support their recommendations the U.S. Preventive Services Task Forces must rely on data from peer-reviewed studies that have looked at quantifiable outcomes. However, I suspect the task force would agree that its recommendations shouldn’t prevent the rest of us from using our own observations and intuition when deciding whether to selectively screen our younger patients for anxiety disorders.
Although it may not generate a measurable data point, providing the parents of a 5-year-old whose troubling behavior is in part the result of an anxiety disorder is invaluable. Do we need to screen all 5-year-olds? The task force says probably not given the current state of our knowledge and I agree. But, the fact that almost 8% of the pediatric population carries the diagnosis and my anecdotal observations suggest that as pediatricians we should be learning more about anxiety disorders and their wide variety of presentations. Then we should selectively screen more of our patients. In fact, I suspect we might help our patients and ourselves by questioning more parents about their own mental health histories even before we have any inkling that their child has a problem. While the degree to which anxiety disorders are inheritable and the exact mechanism is far from clear, I think this history might be a valuable piece of information to learn as early as the prenatal get-acquainted visit. A simple question to a new or expecting parent about what worries them most about becoming a parent would be a good opener. Your reassurance that you expect parents to be worried and welcome hearing about their concerns should be a step in building a strong foundation for a family-provider relationship.
Anxiety happens and unfortunately so do anxiety disorders. We need to be doing a better job of acknowledging and responding to these two realities.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
*This column was updated on 5/4/2022.
On April 12, 2022, the U.S. Preventive Services Task Force released the draft of a recommendation statement titled Screening for Anxiety in Children and Adolescents. Based on their observation that 7.8% of children and adolescents have a current anxiety disorder and their analysis of the magnitude of the net benefit, the Task Force plans on recommending that children ages 8-18 years be screened for the condition. However, the group could not find evidence to support screening for children 7 years and younger.
Over more than 4 decades of general pediatric practice, it became obvious to me that anxiety was driving a high percentage of my office visits. Most often in young children it was parental anxiety that was prompting the phone call or office visit. In older childhood and adolescence it was patient anxiety that began to play a larger role.
Over the last 2 decades the level of anxiety in all age groups has seemed to increase. How large a role the events of Sept. 11, 2001, and other terrorist attacks were playing in this phenomenon is unclear to me. However, I suspect they were significant. More recently the pandemic and the failure of both political parties to forge a working arrangement have fueled even more anxiety in many demographic segments. It may be safe to say that everyone is anxious to one degree or another.
Broad-based anxiety in the general population and the incidence of anxiety disorders severe enough to disrupt a child’s life are certainly two different kettles of fish. However, the factors that have raised the level of anxiety across all age groups certainly hasn’t made things any easier for the child who has inherited or developed an anxiety disorder.
Glancing at the 600-page evidence synthesis that accompanies the task force’s report it is clear that they have taken their challenge seriously. However, I wonder whether looking at the 7-and-under age group with a different lens might have resulted in the inclusion of younger children in their recommendation.
I understand that to support their recommendations the U.S. Preventive Services Task Forces must rely on data from peer-reviewed studies that have looked at quantifiable outcomes. However, I suspect the task force would agree that its recommendations shouldn’t prevent the rest of us from using our own observations and intuition when deciding whether to selectively screen our younger patients for anxiety disorders.
Although it may not generate a measurable data point, providing the parents of a 5-year-old whose troubling behavior is in part the result of an anxiety disorder is invaluable. Do we need to screen all 5-year-olds? The task force says probably not given the current state of our knowledge and I agree. But, the fact that almost 8% of the pediatric population carries the diagnosis and my anecdotal observations suggest that as pediatricians we should be learning more about anxiety disorders and their wide variety of presentations. Then we should selectively screen more of our patients. In fact, I suspect we might help our patients and ourselves by questioning more parents about their own mental health histories even before we have any inkling that their child has a problem. While the degree to which anxiety disorders are inheritable and the exact mechanism is far from clear, I think this history might be a valuable piece of information to learn as early as the prenatal get-acquainted visit. A simple question to a new or expecting parent about what worries them most about becoming a parent would be a good opener. Your reassurance that you expect parents to be worried and welcome hearing about their concerns should be a step in building a strong foundation for a family-provider relationship.
Anxiety happens and unfortunately so do anxiety disorders. We need to be doing a better job of acknowledging and responding to these two realities.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
*This column was updated on 5/4/2022.
Implant may alleviate sleep apnea in teens with Down syndrome
Upper airway hypoglossal nerve stimulation is safe and effective in adolescents with Down syndrome and severe persistent obstructive sleep apnea (OSA) occurring after adenotonsillectomy and who couldn’t tolerate positive airway pressure, early research suggests.
In a phase I study, 42 adolescents received a surgically implanted device that moves the tongue forward during sleep. Results at 1-year follow-up showed 66% “responded well” to treatment and showed a drop in apnea-hypopnea index (AHI) of at least 50%.
“Parents came back to us and said not only is the sleep better but my child seems to be doing better during the day,” lead investigator Christopher Hartnick, MD, director of the Division of Pediatric Otolaryngology and the Pediatric Airway, Voice, and Swallowing Center at Massachusetts Eye and Ear, Boston, told this news organization.
The findings were published online in JAMA Otolaryngology – Head and Neck Surgery.
Limited options
Upper airway simulation has been shown previously to be effective for adults with OSA, but up until now, the process has not been evaluated in children.
The device used in the current study “stimulates the hypoglossal nerve to protrude the tongue and open the airway on inspiration during sleep,” the investigators note.
“Hypoglossal nerve stimulation may be a particularly suitable therapy for patients with Down syndrome because it can augment neuromuscular airway tone and reduce anatomical obstruction at the base of the tongue, a common site of residual obstruction in children with Down syndrome,” they add.
“This study was born out of the frustration of not having an effective treatment option for children with Down syndrome who struggle with sleep apnea,” Dr. Hartnick said in a news release.
A total of 42 adolescents (67% male; mean age, 15 years) with Down syndrome and persistent severe OSA after adenotonsillectomy were implanted with the hypoglossal nerve stimulator. All were followed for 12 months.
The surgery was safe, with the most common adverse event being temporary tongue discomfort in five patients (12%). This typically resolved in weeks, the researchers note.
High response, adherence rates
Results showed response rates and adherence to therapy was high. The mean duration of nightly therapy was 9 hours, with 40 children (95.2%) using the device at least 4 hours every night.
The implant was also effective, with a mean decrease in AHI of 12.9 events per hour (95% confidence interval, –17.0 to –8.7 events per hour).
Nearly two-thirds of the children had at least a 50% reduction in their AHI, while roughly three-fourths had a 12-month follow-up AHI of less than 10 events per hour.
There were also significant improvements in polysomnographic and parent-reported quality of life outcomes 12 months after the implant, including improvement in sleep and daily functioning, behavior, and language.
“Sleep apnea remains one of the most common conditions that I grapple with working with patients with Down syndrome and their families,” co-investigator Brian Skotko, MD, Emma Campbell endowed chair on Down syndrome at Massachusetts General Hospital, Boston, said in the release.
“Until now, so many of our patients had run out of treatment options, and their health and well-being were declining. Now, with the hypoglossal nerve stimulator treatment, we may have an effective and safe way to treat apnea and maximize brain health for people with Down syndrome,” Dr. Skotko added.
Dr. Hartnick and Dr. Skotko have received a $4 million, 5-year grant from the National Institutes of Health to assess whether upper airway stimulation might help cognition in children with Down syndrome.
Landmark investigation
Co-authors of an invited commentary said they “applaud” the researchers for their “landmark” investigation, which demonstrated a response to upper airway stimulation in children with Down syndrome and OSA that is on par with what has been achieved in adults with OSA.
“They have established the safety of the procedure; however, future research is necessary to optimize the results of implant,” write Norman Friedman, MD, and Katherine Green, MD, both from the department of otolaryngology – head and neck surgery, University of Colorado School of Medicine, Aurora.
“Further assessment regarding patient selection and the systematic preoperative identification of potential barriers that might affect successful use of therapy will be beneficial to improve longitudinal outcomes and success in this population that is uniquely different from the adult cohorts that have received implants to date,” they add.
The study was funded by Inspire Medical Systems, which provided eight devices for the study but otherwise did not have a role in its design and conduct. The LuMind IDSC Down Syndrome Foundation also provided funding for the study. Dr. Hartnick and the editorialists have disclosed no relevant financial relationships. A complete list of disclosures for the other investigators is available in the original article.
A version of this article first appeared on Medscape.com.
Upper airway hypoglossal nerve stimulation is safe and effective in adolescents with Down syndrome and severe persistent obstructive sleep apnea (OSA) occurring after adenotonsillectomy and who couldn’t tolerate positive airway pressure, early research suggests.
In a phase I study, 42 adolescents received a surgically implanted device that moves the tongue forward during sleep. Results at 1-year follow-up showed 66% “responded well” to treatment and showed a drop in apnea-hypopnea index (AHI) of at least 50%.
“Parents came back to us and said not only is the sleep better but my child seems to be doing better during the day,” lead investigator Christopher Hartnick, MD, director of the Division of Pediatric Otolaryngology and the Pediatric Airway, Voice, and Swallowing Center at Massachusetts Eye and Ear, Boston, told this news organization.
The findings were published online in JAMA Otolaryngology – Head and Neck Surgery.
Limited options
Upper airway simulation has been shown previously to be effective for adults with OSA, but up until now, the process has not been evaluated in children.
The device used in the current study “stimulates the hypoglossal nerve to protrude the tongue and open the airway on inspiration during sleep,” the investigators note.
“Hypoglossal nerve stimulation may be a particularly suitable therapy for patients with Down syndrome because it can augment neuromuscular airway tone and reduce anatomical obstruction at the base of the tongue, a common site of residual obstruction in children with Down syndrome,” they add.
“This study was born out of the frustration of not having an effective treatment option for children with Down syndrome who struggle with sleep apnea,” Dr. Hartnick said in a news release.
A total of 42 adolescents (67% male; mean age, 15 years) with Down syndrome and persistent severe OSA after adenotonsillectomy were implanted with the hypoglossal nerve stimulator. All were followed for 12 months.
The surgery was safe, with the most common adverse event being temporary tongue discomfort in five patients (12%). This typically resolved in weeks, the researchers note.
High response, adherence rates
Results showed response rates and adherence to therapy was high. The mean duration of nightly therapy was 9 hours, with 40 children (95.2%) using the device at least 4 hours every night.
The implant was also effective, with a mean decrease in AHI of 12.9 events per hour (95% confidence interval, –17.0 to –8.7 events per hour).
Nearly two-thirds of the children had at least a 50% reduction in their AHI, while roughly three-fourths had a 12-month follow-up AHI of less than 10 events per hour.
There were also significant improvements in polysomnographic and parent-reported quality of life outcomes 12 months after the implant, including improvement in sleep and daily functioning, behavior, and language.
“Sleep apnea remains one of the most common conditions that I grapple with working with patients with Down syndrome and their families,” co-investigator Brian Skotko, MD, Emma Campbell endowed chair on Down syndrome at Massachusetts General Hospital, Boston, said in the release.
“Until now, so many of our patients had run out of treatment options, and their health and well-being were declining. Now, with the hypoglossal nerve stimulator treatment, we may have an effective and safe way to treat apnea and maximize brain health for people with Down syndrome,” Dr. Skotko added.
Dr. Hartnick and Dr. Skotko have received a $4 million, 5-year grant from the National Institutes of Health to assess whether upper airway stimulation might help cognition in children with Down syndrome.
Landmark investigation
Co-authors of an invited commentary said they “applaud” the researchers for their “landmark” investigation, which demonstrated a response to upper airway stimulation in children with Down syndrome and OSA that is on par with what has been achieved in adults with OSA.
“They have established the safety of the procedure; however, future research is necessary to optimize the results of implant,” write Norman Friedman, MD, and Katherine Green, MD, both from the department of otolaryngology – head and neck surgery, University of Colorado School of Medicine, Aurora.
“Further assessment regarding patient selection and the systematic preoperative identification of potential barriers that might affect successful use of therapy will be beneficial to improve longitudinal outcomes and success in this population that is uniquely different from the adult cohorts that have received implants to date,” they add.
The study was funded by Inspire Medical Systems, which provided eight devices for the study but otherwise did not have a role in its design and conduct. The LuMind IDSC Down Syndrome Foundation also provided funding for the study. Dr. Hartnick and the editorialists have disclosed no relevant financial relationships. A complete list of disclosures for the other investigators is available in the original article.
A version of this article first appeared on Medscape.com.
Upper airway hypoglossal nerve stimulation is safe and effective in adolescents with Down syndrome and severe persistent obstructive sleep apnea (OSA) occurring after adenotonsillectomy and who couldn’t tolerate positive airway pressure, early research suggests.
In a phase I study, 42 adolescents received a surgically implanted device that moves the tongue forward during sleep. Results at 1-year follow-up showed 66% “responded well” to treatment and showed a drop in apnea-hypopnea index (AHI) of at least 50%.
“Parents came back to us and said not only is the sleep better but my child seems to be doing better during the day,” lead investigator Christopher Hartnick, MD, director of the Division of Pediatric Otolaryngology and the Pediatric Airway, Voice, and Swallowing Center at Massachusetts Eye and Ear, Boston, told this news organization.
The findings were published online in JAMA Otolaryngology – Head and Neck Surgery.
Limited options
Upper airway simulation has been shown previously to be effective for adults with OSA, but up until now, the process has not been evaluated in children.
The device used in the current study “stimulates the hypoglossal nerve to protrude the tongue and open the airway on inspiration during sleep,” the investigators note.
“Hypoglossal nerve stimulation may be a particularly suitable therapy for patients with Down syndrome because it can augment neuromuscular airway tone and reduce anatomical obstruction at the base of the tongue, a common site of residual obstruction in children with Down syndrome,” they add.
“This study was born out of the frustration of not having an effective treatment option for children with Down syndrome who struggle with sleep apnea,” Dr. Hartnick said in a news release.
A total of 42 adolescents (67% male; mean age, 15 years) with Down syndrome and persistent severe OSA after adenotonsillectomy were implanted with the hypoglossal nerve stimulator. All were followed for 12 months.
The surgery was safe, with the most common adverse event being temporary tongue discomfort in five patients (12%). This typically resolved in weeks, the researchers note.
High response, adherence rates
Results showed response rates and adherence to therapy was high. The mean duration of nightly therapy was 9 hours, with 40 children (95.2%) using the device at least 4 hours every night.
The implant was also effective, with a mean decrease in AHI of 12.9 events per hour (95% confidence interval, –17.0 to –8.7 events per hour).
Nearly two-thirds of the children had at least a 50% reduction in their AHI, while roughly three-fourths had a 12-month follow-up AHI of less than 10 events per hour.
There were also significant improvements in polysomnographic and parent-reported quality of life outcomes 12 months after the implant, including improvement in sleep and daily functioning, behavior, and language.
“Sleep apnea remains one of the most common conditions that I grapple with working with patients with Down syndrome and their families,” co-investigator Brian Skotko, MD, Emma Campbell endowed chair on Down syndrome at Massachusetts General Hospital, Boston, said in the release.
“Until now, so many of our patients had run out of treatment options, and their health and well-being were declining. Now, with the hypoglossal nerve stimulator treatment, we may have an effective and safe way to treat apnea and maximize brain health for people with Down syndrome,” Dr. Skotko added.
Dr. Hartnick and Dr. Skotko have received a $4 million, 5-year grant from the National Institutes of Health to assess whether upper airway stimulation might help cognition in children with Down syndrome.
Landmark investigation
Co-authors of an invited commentary said they “applaud” the researchers for their “landmark” investigation, which demonstrated a response to upper airway stimulation in children with Down syndrome and OSA that is on par with what has been achieved in adults with OSA.
“They have established the safety of the procedure; however, future research is necessary to optimize the results of implant,” write Norman Friedman, MD, and Katherine Green, MD, both from the department of otolaryngology – head and neck surgery, University of Colorado School of Medicine, Aurora.
“Further assessment regarding patient selection and the systematic preoperative identification of potential barriers that might affect successful use of therapy will be beneficial to improve longitudinal outcomes and success in this population that is uniquely different from the adult cohorts that have received implants to date,” they add.
The study was funded by Inspire Medical Systems, which provided eight devices for the study but otherwise did not have a role in its design and conduct. The LuMind IDSC Down Syndrome Foundation also provided funding for the study. Dr. Hartnick and the editorialists have disclosed no relevant financial relationships. A complete list of disclosures for the other investigators is available in the original article.
A version of this article first appeared on Medscape.com.
Hospital readmission remains common for teens with nonfatal drug overdose
Approximately 1 in 5 adolescents hospitalized for nonfatal drug overdoses were readmitted within 6 months, based on data from more than 12,000 individuals.
Previous studies suggest that many adolescents fail to receive timely treatment for addiction after a nonfatal overdose, but the rates of hospital readmission in this population have not been examined, according to Julie Gaither, PhD, of Yale University, New Haven, Conn.
In a study presented at the annual meeting of the Pediatric Academic Societies, Dr. Gaither and her colleague, John M. Leventhal, MD, also of Yale University, used data from the 2016 Nationwide Readmissions Database to examine incidence and recurrent hospitalizations for nonfatal drug overdoses in adolescents. The study population included 12,952 patients aged 11-21 years who were admitted to a hospital after a nonfatal drug overdose in 2016. Of these, 15% were younger than 15 years, and 52.1% were females.
Overall, 76.2% of the overdoses involved opioids; 77.9% involved a prescription opioid, 15.3% involved heroin, and 7.9% involved fentanyl.
Across all drug overdoses, the majority (86.5%) were attributed to accidental intent and 11.8% were attributed to self-harm. Notably, females were nearly four times more likely than males to attempt suicide (odds ratio, 3.57). After the initial hospitalization, 79.3% of the patients were discharged home, and 11.5% went to a short-term care facility.
The 6-month hospital readmission rate was 21.4%. Of the patients readmitted for any cause, 18.2% of readmissions were for recurrent overdoses, and 92.1% of these were attributed to opioids.
The median cost of the initial hospital admission was $23,705 (ranging from $11,902 to $54,682) and the median cost of the first readmission was $25,416 (ranging from $13,905 to $48,810). In 42.1% of all hospitalizations, Medicaid was the primary payer.
The study findings were limited by the relatively high number of Medicaid patients, which may limit generalizability, but is strengthened by the large sample size.
The findings highlight not only the need for prevention efforts to limit opioid use among adolescents, but also “speak to the need for timely evidenced-based addiction treatment and appropriate follow-up care for teens following hospitalization for a nonfatal drug overdose,” the researchers wrote in their abstract.
Potential for postpandemic surge in drug use
Interestingly, some recent research has shown a decline in teens’ substance use during the pandemic, Kelly Curran, MD, of the University of Oklahoma Health Sciences Center, Oklahoma City, said in an interview.
“However, as the world begins ‘opening up’ again, I suspect rates of drug use will rise – especially with the significant burden of mental health issues adolescents have struggled with during the last few years,” said Dr. Curran, who was not involved with the current study.
“Sadly, I am not surprised by this study’s findings. Too often, teens with substance abuse issues are not connected to effective, evidenced-based treatment, and for those who are, the wait list can be long,” she said.
“Teens who are misusing drugs – either to get high or to attempt suicide – who are admitted for nonfatal overdose have a high rate of readmission for recurrent drug overdose,” Dr. Curran said. “This high rate of readmission has serious social and financial implications,” she added. “This study is part of a growing body of literature that supports the importance of getting adolescents into effective, evidence-based substance abuse treatment, such as medication-assisted treatment in opioid abuse. However, we also should be advocating for improved funding for and access to these treatments for all individuals.”
The study received no outside funding. Dr. Gaither had no financial conflicts to disclose. Dr. Curran had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
Approximately 1 in 5 adolescents hospitalized for nonfatal drug overdoses were readmitted within 6 months, based on data from more than 12,000 individuals.
Previous studies suggest that many adolescents fail to receive timely treatment for addiction after a nonfatal overdose, but the rates of hospital readmission in this population have not been examined, according to Julie Gaither, PhD, of Yale University, New Haven, Conn.
In a study presented at the annual meeting of the Pediatric Academic Societies, Dr. Gaither and her colleague, John M. Leventhal, MD, also of Yale University, used data from the 2016 Nationwide Readmissions Database to examine incidence and recurrent hospitalizations for nonfatal drug overdoses in adolescents. The study population included 12,952 patients aged 11-21 years who were admitted to a hospital after a nonfatal drug overdose in 2016. Of these, 15% were younger than 15 years, and 52.1% were females.
Overall, 76.2% of the overdoses involved opioids; 77.9% involved a prescription opioid, 15.3% involved heroin, and 7.9% involved fentanyl.
Across all drug overdoses, the majority (86.5%) were attributed to accidental intent and 11.8% were attributed to self-harm. Notably, females were nearly four times more likely than males to attempt suicide (odds ratio, 3.57). After the initial hospitalization, 79.3% of the patients were discharged home, and 11.5% went to a short-term care facility.
The 6-month hospital readmission rate was 21.4%. Of the patients readmitted for any cause, 18.2% of readmissions were for recurrent overdoses, and 92.1% of these were attributed to opioids.
The median cost of the initial hospital admission was $23,705 (ranging from $11,902 to $54,682) and the median cost of the first readmission was $25,416 (ranging from $13,905 to $48,810). In 42.1% of all hospitalizations, Medicaid was the primary payer.
The study findings were limited by the relatively high number of Medicaid patients, which may limit generalizability, but is strengthened by the large sample size.
The findings highlight not only the need for prevention efforts to limit opioid use among adolescents, but also “speak to the need for timely evidenced-based addiction treatment and appropriate follow-up care for teens following hospitalization for a nonfatal drug overdose,” the researchers wrote in their abstract.
Potential for postpandemic surge in drug use
Interestingly, some recent research has shown a decline in teens’ substance use during the pandemic, Kelly Curran, MD, of the University of Oklahoma Health Sciences Center, Oklahoma City, said in an interview.
“However, as the world begins ‘opening up’ again, I suspect rates of drug use will rise – especially with the significant burden of mental health issues adolescents have struggled with during the last few years,” said Dr. Curran, who was not involved with the current study.
“Sadly, I am not surprised by this study’s findings. Too often, teens with substance abuse issues are not connected to effective, evidenced-based treatment, and for those who are, the wait list can be long,” she said.
“Teens who are misusing drugs – either to get high or to attempt suicide – who are admitted for nonfatal overdose have a high rate of readmission for recurrent drug overdose,” Dr. Curran said. “This high rate of readmission has serious social and financial implications,” she added. “This study is part of a growing body of literature that supports the importance of getting adolescents into effective, evidence-based substance abuse treatment, such as medication-assisted treatment in opioid abuse. However, we also should be advocating for improved funding for and access to these treatments for all individuals.”
The study received no outside funding. Dr. Gaither had no financial conflicts to disclose. Dr. Curran had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
Approximately 1 in 5 adolescents hospitalized for nonfatal drug overdoses were readmitted within 6 months, based on data from more than 12,000 individuals.
Previous studies suggest that many adolescents fail to receive timely treatment for addiction after a nonfatal overdose, but the rates of hospital readmission in this population have not been examined, according to Julie Gaither, PhD, of Yale University, New Haven, Conn.
In a study presented at the annual meeting of the Pediatric Academic Societies, Dr. Gaither and her colleague, John M. Leventhal, MD, also of Yale University, used data from the 2016 Nationwide Readmissions Database to examine incidence and recurrent hospitalizations for nonfatal drug overdoses in adolescents. The study population included 12,952 patients aged 11-21 years who were admitted to a hospital after a nonfatal drug overdose in 2016. Of these, 15% were younger than 15 years, and 52.1% were females.
Overall, 76.2% of the overdoses involved opioids; 77.9% involved a prescription opioid, 15.3% involved heroin, and 7.9% involved fentanyl.
Across all drug overdoses, the majority (86.5%) were attributed to accidental intent and 11.8% were attributed to self-harm. Notably, females were nearly four times more likely than males to attempt suicide (odds ratio, 3.57). After the initial hospitalization, 79.3% of the patients were discharged home, and 11.5% went to a short-term care facility.
The 6-month hospital readmission rate was 21.4%. Of the patients readmitted for any cause, 18.2% of readmissions were for recurrent overdoses, and 92.1% of these were attributed to opioids.
The median cost of the initial hospital admission was $23,705 (ranging from $11,902 to $54,682) and the median cost of the first readmission was $25,416 (ranging from $13,905 to $48,810). In 42.1% of all hospitalizations, Medicaid was the primary payer.
The study findings were limited by the relatively high number of Medicaid patients, which may limit generalizability, but is strengthened by the large sample size.
The findings highlight not only the need for prevention efforts to limit opioid use among adolescents, but also “speak to the need for timely evidenced-based addiction treatment and appropriate follow-up care for teens following hospitalization for a nonfatal drug overdose,” the researchers wrote in their abstract.
Potential for postpandemic surge in drug use
Interestingly, some recent research has shown a decline in teens’ substance use during the pandemic, Kelly Curran, MD, of the University of Oklahoma Health Sciences Center, Oklahoma City, said in an interview.
“However, as the world begins ‘opening up’ again, I suspect rates of drug use will rise – especially with the significant burden of mental health issues adolescents have struggled with during the last few years,” said Dr. Curran, who was not involved with the current study.
“Sadly, I am not surprised by this study’s findings. Too often, teens with substance abuse issues are not connected to effective, evidenced-based treatment, and for those who are, the wait list can be long,” she said.
“Teens who are misusing drugs – either to get high or to attempt suicide – who are admitted for nonfatal overdose have a high rate of readmission for recurrent drug overdose,” Dr. Curran said. “This high rate of readmission has serious social and financial implications,” she added. “This study is part of a growing body of literature that supports the importance of getting adolescents into effective, evidence-based substance abuse treatment, such as medication-assisted treatment in opioid abuse. However, we also should be advocating for improved funding for and access to these treatments for all individuals.”
The study received no outside funding. Dr. Gaither had no financial conflicts to disclose. Dr. Curran had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
FROM PAS 2022
What to expect when you’re expecting ... a preemie
The prospect of having a premature infant can be highly stressful. But a new study found that providing pregnant patients hospitalized for preterm labor with detailed information about what to expect with an early birth significantly reduced their anxiety about the process.
The study found that both printed handouts and a tablet app were associated with a 50% reduction in anxiety and appeared to be equally effective, although the handouts are likely easier to use in the high-stress environment of neonatal intensive care facilities, according to the researchers, who presented the findings April 25 at the annual meeting of the Pediatric Academic Societies.
“When patients get admitted for preterm labor a neonatologist comes to talk to parents about outcomes, short- and long-term, like bleeding in the baby’s brain and the possible need to have surgeries,” said Nicole Rau, MD, assistant professor of clinical pediatrics at University of Illinois at Peoria, who led the study. “Then parents are asked to make decisions during a high-stress time while they’re still processing everything. Everyone agrees that’s really not ideal.”
About 1 in 10 babies in the United States are born prematurely – or before 37 weeks of gestation – each year. That adds up to about 500,000 per year. Many spend days or weeks in neonatal intensive care units – watched from a distance by their anxious parents desperate for answers and reassurance. Potential complications for infants born prematurely include heart issues, trouble breathing, brain bleeds, and difficulty controlling their body temperature.
The American Academy of Pediatrics and the National Institute of Child Health and Human Development have warned that birth parents at risk for premature delivery may not be adequately prepared for what to expect. According to the groups, although clinicians may counsel these patients on admission to the hospital, factors such as stress, pain, and maternal medication can make the message difficult to comprehend.
For the study, Dr. Rau and her colleagues divided patients at the Medical College of Wisconsin and Children’s Hospital of Wisconsin who were hospitalized between 22 and 33 weeks of pregnancy into two groups: Some received a handout on preterm labor, and some were given a bedside tablet with an app called Preemie Prep for Parents.
Seventy-six women were randomized in gestational age blocks of 22-24 weeks and 25-33 weeks. After some opted not to complete the study, 59 participants remained – 32 of whom received handouts, and 27 who had access to tablets.
After distributing the materials, Dr. Rau’s group gave patients a questionnaire asking about delivery resuscitation, short-term problems, long-term problems, treatments, length of stay, and miscellaneous questions about their care. The two groups performed similarly – the tablet group’s median score was 20/30, and the handout group’s median score was 22/30.
Using the State-Trait Anxiety Inventory, researchers found both groups experienced a 50% reduction in anxiety after learning more from their respective materials.
Dr. Rau said she and her colleagues expected patients with access to the app would perform better based on cognition studies that have shown multimedia tools are more effective than tools that use visual or audio information but not both. However, both groups seemed to benefit comparably, which she said may reflect underuse of the app.
What was clear, though, is that patients absorbed more information and felt better prepared when they received it in ways beyond verbal communication.
“Well-written, parent-friendly information is a great tool to supplement counseling,” Dr. Rau told this news organization.
Because preterm labor is a relatively common occurrence, expectant parents should be well-prepared with proper information, said Erika Werner, MD, chair of obstetrics & gynecology at Tufts Medical Center, Boston, who was not involved in the study.
“Preterm labor is something that’s way more common than people think,” Dr. Werner told this news organization. “As long as it’s coming from a trusted source, additional information is a good thing. Knowing in advance some of the things that might be different from what you expect is always important. The more that we as providers have time to educate patients about potential risks, the better the outcomes will be.”
The authors reported no relevant financial conflicts of interest. The study was supported by grants from Children’s Research Institute and AMAG Pharmaceuticals.
A version of this article first appeared on Medscape.com.
The prospect of having a premature infant can be highly stressful. But a new study found that providing pregnant patients hospitalized for preterm labor with detailed information about what to expect with an early birth significantly reduced their anxiety about the process.
The study found that both printed handouts and a tablet app were associated with a 50% reduction in anxiety and appeared to be equally effective, although the handouts are likely easier to use in the high-stress environment of neonatal intensive care facilities, according to the researchers, who presented the findings April 25 at the annual meeting of the Pediatric Academic Societies.
“When patients get admitted for preterm labor a neonatologist comes to talk to parents about outcomes, short- and long-term, like bleeding in the baby’s brain and the possible need to have surgeries,” said Nicole Rau, MD, assistant professor of clinical pediatrics at University of Illinois at Peoria, who led the study. “Then parents are asked to make decisions during a high-stress time while they’re still processing everything. Everyone agrees that’s really not ideal.”
About 1 in 10 babies in the United States are born prematurely – or before 37 weeks of gestation – each year. That adds up to about 500,000 per year. Many spend days or weeks in neonatal intensive care units – watched from a distance by their anxious parents desperate for answers and reassurance. Potential complications for infants born prematurely include heart issues, trouble breathing, brain bleeds, and difficulty controlling their body temperature.
The American Academy of Pediatrics and the National Institute of Child Health and Human Development have warned that birth parents at risk for premature delivery may not be adequately prepared for what to expect. According to the groups, although clinicians may counsel these patients on admission to the hospital, factors such as stress, pain, and maternal medication can make the message difficult to comprehend.
For the study, Dr. Rau and her colleagues divided patients at the Medical College of Wisconsin and Children’s Hospital of Wisconsin who were hospitalized between 22 and 33 weeks of pregnancy into two groups: Some received a handout on preterm labor, and some were given a bedside tablet with an app called Preemie Prep for Parents.
Seventy-six women were randomized in gestational age blocks of 22-24 weeks and 25-33 weeks. After some opted not to complete the study, 59 participants remained – 32 of whom received handouts, and 27 who had access to tablets.
After distributing the materials, Dr. Rau’s group gave patients a questionnaire asking about delivery resuscitation, short-term problems, long-term problems, treatments, length of stay, and miscellaneous questions about their care. The two groups performed similarly – the tablet group’s median score was 20/30, and the handout group’s median score was 22/30.
Using the State-Trait Anxiety Inventory, researchers found both groups experienced a 50% reduction in anxiety after learning more from their respective materials.
Dr. Rau said she and her colleagues expected patients with access to the app would perform better based on cognition studies that have shown multimedia tools are more effective than tools that use visual or audio information but not both. However, both groups seemed to benefit comparably, which she said may reflect underuse of the app.
What was clear, though, is that patients absorbed more information and felt better prepared when they received it in ways beyond verbal communication.
“Well-written, parent-friendly information is a great tool to supplement counseling,” Dr. Rau told this news organization.
Because preterm labor is a relatively common occurrence, expectant parents should be well-prepared with proper information, said Erika Werner, MD, chair of obstetrics & gynecology at Tufts Medical Center, Boston, who was not involved in the study.
“Preterm labor is something that’s way more common than people think,” Dr. Werner told this news organization. “As long as it’s coming from a trusted source, additional information is a good thing. Knowing in advance some of the things that might be different from what you expect is always important. The more that we as providers have time to educate patients about potential risks, the better the outcomes will be.”
The authors reported no relevant financial conflicts of interest. The study was supported by grants from Children’s Research Institute and AMAG Pharmaceuticals.
A version of this article first appeared on Medscape.com.
The prospect of having a premature infant can be highly stressful. But a new study found that providing pregnant patients hospitalized for preterm labor with detailed information about what to expect with an early birth significantly reduced their anxiety about the process.
The study found that both printed handouts and a tablet app were associated with a 50% reduction in anxiety and appeared to be equally effective, although the handouts are likely easier to use in the high-stress environment of neonatal intensive care facilities, according to the researchers, who presented the findings April 25 at the annual meeting of the Pediatric Academic Societies.
“When patients get admitted for preterm labor a neonatologist comes to talk to parents about outcomes, short- and long-term, like bleeding in the baby’s brain and the possible need to have surgeries,” said Nicole Rau, MD, assistant professor of clinical pediatrics at University of Illinois at Peoria, who led the study. “Then parents are asked to make decisions during a high-stress time while they’re still processing everything. Everyone agrees that’s really not ideal.”
About 1 in 10 babies in the United States are born prematurely – or before 37 weeks of gestation – each year. That adds up to about 500,000 per year. Many spend days or weeks in neonatal intensive care units – watched from a distance by their anxious parents desperate for answers and reassurance. Potential complications for infants born prematurely include heart issues, trouble breathing, brain bleeds, and difficulty controlling their body temperature.
The American Academy of Pediatrics and the National Institute of Child Health and Human Development have warned that birth parents at risk for premature delivery may not be adequately prepared for what to expect. According to the groups, although clinicians may counsel these patients on admission to the hospital, factors such as stress, pain, and maternal medication can make the message difficult to comprehend.
For the study, Dr. Rau and her colleagues divided patients at the Medical College of Wisconsin and Children’s Hospital of Wisconsin who were hospitalized between 22 and 33 weeks of pregnancy into two groups: Some received a handout on preterm labor, and some were given a bedside tablet with an app called Preemie Prep for Parents.
Seventy-six women were randomized in gestational age blocks of 22-24 weeks and 25-33 weeks. After some opted not to complete the study, 59 participants remained – 32 of whom received handouts, and 27 who had access to tablets.
After distributing the materials, Dr. Rau’s group gave patients a questionnaire asking about delivery resuscitation, short-term problems, long-term problems, treatments, length of stay, and miscellaneous questions about their care. The two groups performed similarly – the tablet group’s median score was 20/30, and the handout group’s median score was 22/30.
Using the State-Trait Anxiety Inventory, researchers found both groups experienced a 50% reduction in anxiety after learning more from their respective materials.
Dr. Rau said she and her colleagues expected patients with access to the app would perform better based on cognition studies that have shown multimedia tools are more effective than tools that use visual or audio information but not both. However, both groups seemed to benefit comparably, which she said may reflect underuse of the app.
What was clear, though, is that patients absorbed more information and felt better prepared when they received it in ways beyond verbal communication.
“Well-written, parent-friendly information is a great tool to supplement counseling,” Dr. Rau told this news organization.
Because preterm labor is a relatively common occurrence, expectant parents should be well-prepared with proper information, said Erika Werner, MD, chair of obstetrics & gynecology at Tufts Medical Center, Boston, who was not involved in the study.
“Preterm labor is something that’s way more common than people think,” Dr. Werner told this news organization. “As long as it’s coming from a trusted source, additional information is a good thing. Knowing in advance some of the things that might be different from what you expect is always important. The more that we as providers have time to educate patients about potential risks, the better the outcomes will be.”
The authors reported no relevant financial conflicts of interest. The study was supported by grants from Children’s Research Institute and AMAG Pharmaceuticals.
A version of this article first appeared on Medscape.com.
FROM PAS 2022
Topical treatment for EB recommended for approval in the EU
A topical (EMA’s) Committee for Medicinal Products for Human Use.
“The benefit of Filsuvez is its ability to promote healing of EB partial thickness wounds,” the EMA said in an announcement on April 22. “It is thought to work by modulating inflammatory mediators and stimulating keratinocyte differentiation and migration, thereby promoting wound health and closure,” the statement adds.
The recommended indication for the product – developed by Amryt Pharmaceuticals DAC and currently designated as an orphan drug – is for the treatment of partial-thickness wounds associated with dystrophic and junctional EB in patients aged 6 months and older. The recommendation for approval came after the EMA sought and received external advice from independent physicians treating EB and from patients with the rare disease.
The most common side effects, according to the EMA announcement, are wound complications, application site reactions, wound infections, pruritus, and hypersensitivity reactions.
In February 2022, the Food and Drug Administration declined to approve the company’s new drug application as it was presented and asked the company to submit additional evidence of effectiveness for Oleogel-S10 in EB, the company announced at that time. The statement noted that the company was committed to working with the FDA to identify "the most expeditious pathway towards a potential approval.”
The company’s pivotal phase 3 trial enrolled 223 patients with EB, including 156 pediatric patients. The patients variously had three types of EB. The trial has two components: A 3-month, double-blind, randomized controlled phase, which has been completed, and an ongoing 24-month open-label, single-arm phase. The trial is being performed at 58 sites in 28 countries.
Results from the randomized controlled phase, reported in 2020, include a statistically significant increase in the proportion of patients achieving complete closure of an EB target wound within 45 days: 41.3% in the Oleogel-S10 group and 28.9% in the control group (P = .013). (Target wounds measured 10 cm² to 50 cm² and were present for at least 21 days but less than 9 months.) The safety profile of the treatment gel was acceptable and was well tolerated, compared with the control gel, according to Amryt’s press release. The results were presented at the European Academy of Dermatology and Venereology Congress in October 2020.
Data from a 12-month interim analysis of the follow-up phase were presented at the annual meeting of the American Academy of Dermatology in March 2022. Results showed further reductions in total body surface area percentage wounding to 5.4% among (from 7.4% at the end of the double-blind period and 12.1% at the beginning of the study) among the patients who continued treatment and who underwent assessment, according to a company press release. Treatment was well tolerated, and no new safety signals were identified, the release said.
A decision by the European Commission is expected within the next 2 months.
A version of this article first appeared on Medscape.com.
A topical (EMA’s) Committee for Medicinal Products for Human Use.
“The benefit of Filsuvez is its ability to promote healing of EB partial thickness wounds,” the EMA said in an announcement on April 22. “It is thought to work by modulating inflammatory mediators and stimulating keratinocyte differentiation and migration, thereby promoting wound health and closure,” the statement adds.
The recommended indication for the product – developed by Amryt Pharmaceuticals DAC and currently designated as an orphan drug – is for the treatment of partial-thickness wounds associated with dystrophic and junctional EB in patients aged 6 months and older. The recommendation for approval came after the EMA sought and received external advice from independent physicians treating EB and from patients with the rare disease.
The most common side effects, according to the EMA announcement, are wound complications, application site reactions, wound infections, pruritus, and hypersensitivity reactions.
In February 2022, the Food and Drug Administration declined to approve the company’s new drug application as it was presented and asked the company to submit additional evidence of effectiveness for Oleogel-S10 in EB, the company announced at that time. The statement noted that the company was committed to working with the FDA to identify "the most expeditious pathway towards a potential approval.”
The company’s pivotal phase 3 trial enrolled 223 patients with EB, including 156 pediatric patients. The patients variously had three types of EB. The trial has two components: A 3-month, double-blind, randomized controlled phase, which has been completed, and an ongoing 24-month open-label, single-arm phase. The trial is being performed at 58 sites in 28 countries.
Results from the randomized controlled phase, reported in 2020, include a statistically significant increase in the proportion of patients achieving complete closure of an EB target wound within 45 days: 41.3% in the Oleogel-S10 group and 28.9% in the control group (P = .013). (Target wounds measured 10 cm² to 50 cm² and were present for at least 21 days but less than 9 months.) The safety profile of the treatment gel was acceptable and was well tolerated, compared with the control gel, according to Amryt’s press release. The results were presented at the European Academy of Dermatology and Venereology Congress in October 2020.
Data from a 12-month interim analysis of the follow-up phase were presented at the annual meeting of the American Academy of Dermatology in March 2022. Results showed further reductions in total body surface area percentage wounding to 5.4% among (from 7.4% at the end of the double-blind period and 12.1% at the beginning of the study) among the patients who continued treatment and who underwent assessment, according to a company press release. Treatment was well tolerated, and no new safety signals were identified, the release said.
A decision by the European Commission is expected within the next 2 months.
A version of this article first appeared on Medscape.com.
A topical (EMA’s) Committee for Medicinal Products for Human Use.
“The benefit of Filsuvez is its ability to promote healing of EB partial thickness wounds,” the EMA said in an announcement on April 22. “It is thought to work by modulating inflammatory mediators and stimulating keratinocyte differentiation and migration, thereby promoting wound health and closure,” the statement adds.
The recommended indication for the product – developed by Amryt Pharmaceuticals DAC and currently designated as an orphan drug – is for the treatment of partial-thickness wounds associated with dystrophic and junctional EB in patients aged 6 months and older. The recommendation for approval came after the EMA sought and received external advice from independent physicians treating EB and from patients with the rare disease.
The most common side effects, according to the EMA announcement, are wound complications, application site reactions, wound infections, pruritus, and hypersensitivity reactions.
In February 2022, the Food and Drug Administration declined to approve the company’s new drug application as it was presented and asked the company to submit additional evidence of effectiveness for Oleogel-S10 in EB, the company announced at that time. The statement noted that the company was committed to working with the FDA to identify "the most expeditious pathway towards a potential approval.”
The company’s pivotal phase 3 trial enrolled 223 patients with EB, including 156 pediatric patients. The patients variously had three types of EB. The trial has two components: A 3-month, double-blind, randomized controlled phase, which has been completed, and an ongoing 24-month open-label, single-arm phase. The trial is being performed at 58 sites in 28 countries.
Results from the randomized controlled phase, reported in 2020, include a statistically significant increase in the proportion of patients achieving complete closure of an EB target wound within 45 days: 41.3% in the Oleogel-S10 group and 28.9% in the control group (P = .013). (Target wounds measured 10 cm² to 50 cm² and were present for at least 21 days but less than 9 months.) The safety profile of the treatment gel was acceptable and was well tolerated, compared with the control gel, according to Amryt’s press release. The results were presented at the European Academy of Dermatology and Venereology Congress in October 2020.
Data from a 12-month interim analysis of the follow-up phase were presented at the annual meeting of the American Academy of Dermatology in March 2022. Results showed further reductions in total body surface area percentage wounding to 5.4% among (from 7.4% at the end of the double-blind period and 12.1% at the beginning of the study) among the patients who continued treatment and who underwent assessment, according to a company press release. Treatment was well tolerated, and no new safety signals were identified, the release said.
A decision by the European Commission is expected within the next 2 months.
A version of this article first appeared on Medscape.com.
Children and COVID: Weekly cases rise again, but more slowly
New cases of COVID-19 in U.S. children went up for a second consecutive week, but the pace of increase slowed considerably, based on a report from the American Academy of Pediatrics and the Children’s Hospital Association.
The previous week’s count – about 33,000 new COVID cases for April 8-14 – was almost 30% higher than the week before and marked the first rise in incidence after 11 straight weeks of declines, the AAP and CHA said in their weekly COVID-19 report, which is based on data from state and territorial health departments.
The cumulative number of child COVID-19 cases since the start of the pandemic is now over 12.9 million, with children representing 19.0% of cases among all ages. The Centers for Disease Control and Prevention, which uses a different age range for children (0-17 years) than many states, reports corresponding figures of 12.4 million and 17.6%, along with 1,501 deaths.
ED visits show a similar rising trend over recent weeks, as the 7-day average of ED visits with confirmed COVID has crept up from 0.5% in late March/early April to 0.8% on April 22 for children aged 0-11 years, from 0.3% for 0.5% for those aged 12-15, and from 0.3% to 0.6% for 16- and 17-year-olds, based on CDC data.
The daily rate for new admissions for children with confirmed COVID has also moved up slightly, rising from 0.13 per 100,000 population as late as April 13 to 0.15 per 100,000 on April 23. For the number of actual admissions, the latest 7-day (April 17-23) average was 107 in children aged 0-17, compared with 102 for the week of April 10-16, the CDC reported.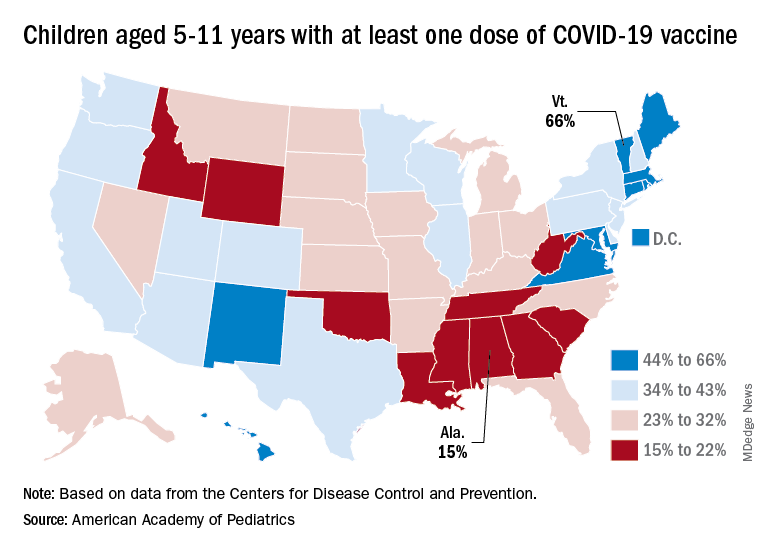
Uptake of the COVID vaccine, however, continued to slide since spiking in January. Initial vaccinations for the latest available week (April 14-20) were down to 48,000 from 59,000 the week before in children aged 5-11 years and 35,000 (vs. 47,000) for those aged 12-17. The weekly highs hit 500,000 and 331,000, respectively, during the Omicron surge, the AAP reported based on CDC data.
Among children aged 5-11, the CDC said that 35.0% had received at least one dose of COVID vaccine as of April 25 and that 28.3% are fully vaccinated, with corresponding figures of 68.8% and 58.8% for 12- to 17-year-olds on April 25.
Among the states, the highest vaccination rates generally are found in New England and the lowest in the Southeast. In Alabama, just 15% of children aged 5-11 have received an initial dose of the vaccine, compared with 66% in Vermont, while Wyoming is the lowest (41%) for children aged 12-17 and Massachusetts is the highest (96%), the AAP said in a separate report.
New cases of COVID-19 in U.S. children went up for a second consecutive week, but the pace of increase slowed considerably, based on a report from the American Academy of Pediatrics and the Children’s Hospital Association.
The previous week’s count – about 33,000 new COVID cases for April 8-14 – was almost 30% higher than the week before and marked the first rise in incidence after 11 straight weeks of declines, the AAP and CHA said in their weekly COVID-19 report, which is based on data from state and territorial health departments.
The cumulative number of child COVID-19 cases since the start of the pandemic is now over 12.9 million, with children representing 19.0% of cases among all ages. The Centers for Disease Control and Prevention, which uses a different age range for children (0-17 years) than many states, reports corresponding figures of 12.4 million and 17.6%, along with 1,501 deaths.
ED visits show a similar rising trend over recent weeks, as the 7-day average of ED visits with confirmed COVID has crept up from 0.5% in late March/early April to 0.8% on April 22 for children aged 0-11 years, from 0.3% for 0.5% for those aged 12-15, and from 0.3% to 0.6% for 16- and 17-year-olds, based on CDC data.
The daily rate for new admissions for children with confirmed COVID has also moved up slightly, rising from 0.13 per 100,000 population as late as April 13 to 0.15 per 100,000 on April 23. For the number of actual admissions, the latest 7-day (April 17-23) average was 107 in children aged 0-17, compared with 102 for the week of April 10-16, the CDC reported.
Uptake of the COVID vaccine, however, continued to slide since spiking in January. Initial vaccinations for the latest available week (April 14-20) were down to 48,000 from 59,000 the week before in children aged 5-11 years and 35,000 (vs. 47,000) for those aged 12-17. The weekly highs hit 500,000 and 331,000, respectively, during the Omicron surge, the AAP reported based on CDC data.
Among children aged 5-11, the CDC said that 35.0% had received at least one dose of COVID vaccine as of April 25 and that 28.3% are fully vaccinated, with corresponding figures of 68.8% and 58.8% for 12- to 17-year-olds on April 25.
Among the states, the highest vaccination rates generally are found in New England and the lowest in the Southeast. In Alabama, just 15% of children aged 5-11 have received an initial dose of the vaccine, compared with 66% in Vermont, while Wyoming is the lowest (41%) for children aged 12-17 and Massachusetts is the highest (96%), the AAP said in a separate report.
New cases of COVID-19 in U.S. children went up for a second consecutive week, but the pace of increase slowed considerably, based on a report from the American Academy of Pediatrics and the Children’s Hospital Association.
The previous week’s count – about 33,000 new COVID cases for April 8-14 – was almost 30% higher than the week before and marked the first rise in incidence after 11 straight weeks of declines, the AAP and CHA said in their weekly COVID-19 report, which is based on data from state and territorial health departments.
The cumulative number of child COVID-19 cases since the start of the pandemic is now over 12.9 million, with children representing 19.0% of cases among all ages. The Centers for Disease Control and Prevention, which uses a different age range for children (0-17 years) than many states, reports corresponding figures of 12.4 million and 17.6%, along with 1,501 deaths.
ED visits show a similar rising trend over recent weeks, as the 7-day average of ED visits with confirmed COVID has crept up from 0.5% in late March/early April to 0.8% on April 22 for children aged 0-11 years, from 0.3% for 0.5% for those aged 12-15, and from 0.3% to 0.6% for 16- and 17-year-olds, based on CDC data.
The daily rate for new admissions for children with confirmed COVID has also moved up slightly, rising from 0.13 per 100,000 population as late as April 13 to 0.15 per 100,000 on April 23. For the number of actual admissions, the latest 7-day (April 17-23) average was 107 in children aged 0-17, compared with 102 for the week of April 10-16, the CDC reported.
Uptake of the COVID vaccine, however, continued to slide since spiking in January. Initial vaccinations for the latest available week (April 14-20) were down to 48,000 from 59,000 the week before in children aged 5-11 years and 35,000 (vs. 47,000) for those aged 12-17. The weekly highs hit 500,000 and 331,000, respectively, during the Omicron surge, the AAP reported based on CDC data.
Among children aged 5-11, the CDC said that 35.0% had received at least one dose of COVID vaccine as of April 25 and that 28.3% are fully vaccinated, with corresponding figures of 68.8% and 58.8% for 12- to 17-year-olds on April 25.
Among the states, the highest vaccination rates generally are found in New England and the lowest in the Southeast. In Alabama, just 15% of children aged 5-11 have received an initial dose of the vaccine, compared with 66% in Vermont, while Wyoming is the lowest (41%) for children aged 12-17 and Massachusetts is the highest (96%), the AAP said in a separate report.
TV time related to poor eating in toddlers
Toddlers who watched more TV were significantly more likely than those who watched less TV to consume sugar-sweetened drinks and junk foods, based on data from 529 children.
Previous research had shown an association between screen time and poor diet, but most have involved school-aged children; the relationship in toddlers has not been well studied, Melissa R. Lutz, MD, of Johns Hopkins University, Baltimore, said in a presentation at the Pediatric Academic Societies annual meeting.
The American Academy of Pediatrics currently recommends no digital media for children younger than 18-24 months, and an hour or less daily for children aged 2-5 years.
To examine the association between TV time and dietary practices in 2-year-olds, the researchers conducted a secondary analysis of data from 529 children who presented for their 2-year-old well-child visit at a single center. The study population was 52% Latino/Hispanic and 30% non-Latino/Hispanic Black, and 69% had an annual household income less than $20,000. The median time spent watching TV daily was 42 minutes. The data were taken from participants in the Greenlight Intervention Study, a randomized trial of an obesity prevention program at four academic pediatric primary care clinics in the United States.
Daily screen time and dietary practices were based on parent reports, and included daily volume of juice, daily counts of fruits and vegetables, daily count of junk foods such as chips, ice cream, French fries, and fast food, and consumption of sugar-sweetened beverages. The cross-sectional analysis controlled for race/ethnicity, Women, Infants, and Children Program benefits, number of children at home, caregiver education level, and family income.
In adjusted analysis, more than an hour of TV time was significantly associated with junk food intake, with odds ratios of 1.12 for 90 minutes and 1.25 for 120 minutes (P < .05 for both). Similar associations were seen for TV times of 90 minutes and 120 minutes and intake of fast food and sugar-sweetened beverages.
Additionally, the researchers found that toddlers who watched TV during mealtimes were more than twice as likely to consume sugar-sweetened beverages (OR, 2.74), junk food (OR, 2.72), fast food (OR, 2.09), and only about half as likely to consume fruits and vegetables (OR, 0.62).
The study findings were limited by several factors including the cross-sectional design, the reliance on caregiver self-reports, potential for residual confounding, and the low average screen time, Dr. Lutz noted.
However, the results suggest that “increased screen TV time and mealtime TV were both associated with poor dietary practices in 2-year-old children,” she said.
Future research should include analysis of passive screen time, as well as the relationship between screen time and diet with other digital devices beyond TV, she added.
COVID drove screen time higher
The current study is especially important at this time because of the increased screen exposure for many young children in the wake of the ongoing pandemic, Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn., said in an interview. “Screen time use is up even more than before [the pandemic], and this study is a reminder to ask parents of young children about screen time and dietary history.”
Dr. Kinsella said she was not surprised by the study findings. In her practice, “I see families with more screen time use in general who also are more likely to have juice and junk food available. If kids had no access to screens, I believe they would still have access to unhealthy foods. I believe more research is needed into why screen time is so high in some families.”
The study received funding from NIH. The researchers had no financial conflicts to disclose. Dr. Kinsella had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
Toddlers who watched more TV were significantly more likely than those who watched less TV to consume sugar-sweetened drinks and junk foods, based on data from 529 children.
Previous research had shown an association between screen time and poor diet, but most have involved school-aged children; the relationship in toddlers has not been well studied, Melissa R. Lutz, MD, of Johns Hopkins University, Baltimore, said in a presentation at the Pediatric Academic Societies annual meeting.
The American Academy of Pediatrics currently recommends no digital media for children younger than 18-24 months, and an hour or less daily for children aged 2-5 years.
To examine the association between TV time and dietary practices in 2-year-olds, the researchers conducted a secondary analysis of data from 529 children who presented for their 2-year-old well-child visit at a single center. The study population was 52% Latino/Hispanic and 30% non-Latino/Hispanic Black, and 69% had an annual household income less than $20,000. The median time spent watching TV daily was 42 minutes. The data were taken from participants in the Greenlight Intervention Study, a randomized trial of an obesity prevention program at four academic pediatric primary care clinics in the United States.
Daily screen time and dietary practices were based on parent reports, and included daily volume of juice, daily counts of fruits and vegetables, daily count of junk foods such as chips, ice cream, French fries, and fast food, and consumption of sugar-sweetened beverages. The cross-sectional analysis controlled for race/ethnicity, Women, Infants, and Children Program benefits, number of children at home, caregiver education level, and family income.
In adjusted analysis, more than an hour of TV time was significantly associated with junk food intake, with odds ratios of 1.12 for 90 minutes and 1.25 for 120 minutes (P < .05 for both). Similar associations were seen for TV times of 90 minutes and 120 minutes and intake of fast food and sugar-sweetened beverages.
Additionally, the researchers found that toddlers who watched TV during mealtimes were more than twice as likely to consume sugar-sweetened beverages (OR, 2.74), junk food (OR, 2.72), fast food (OR, 2.09), and only about half as likely to consume fruits and vegetables (OR, 0.62).
The study findings were limited by several factors including the cross-sectional design, the reliance on caregiver self-reports, potential for residual confounding, and the low average screen time, Dr. Lutz noted.
However, the results suggest that “increased screen TV time and mealtime TV were both associated with poor dietary practices in 2-year-old children,” she said.
Future research should include analysis of passive screen time, as well as the relationship between screen time and diet with other digital devices beyond TV, she added.
COVID drove screen time higher
The current study is especially important at this time because of the increased screen exposure for many young children in the wake of the ongoing pandemic, Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn., said in an interview. “Screen time use is up even more than before [the pandemic], and this study is a reminder to ask parents of young children about screen time and dietary history.”
Dr. Kinsella said she was not surprised by the study findings. In her practice, “I see families with more screen time use in general who also are more likely to have juice and junk food available. If kids had no access to screens, I believe they would still have access to unhealthy foods. I believe more research is needed into why screen time is so high in some families.”
The study received funding from NIH. The researchers had no financial conflicts to disclose. Dr. Kinsella had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
Toddlers who watched more TV were significantly more likely than those who watched less TV to consume sugar-sweetened drinks and junk foods, based on data from 529 children.
Previous research had shown an association between screen time and poor diet, but most have involved school-aged children; the relationship in toddlers has not been well studied, Melissa R. Lutz, MD, of Johns Hopkins University, Baltimore, said in a presentation at the Pediatric Academic Societies annual meeting.
The American Academy of Pediatrics currently recommends no digital media for children younger than 18-24 months, and an hour or less daily for children aged 2-5 years.
To examine the association between TV time and dietary practices in 2-year-olds, the researchers conducted a secondary analysis of data from 529 children who presented for their 2-year-old well-child visit at a single center. The study population was 52% Latino/Hispanic and 30% non-Latino/Hispanic Black, and 69% had an annual household income less than $20,000. The median time spent watching TV daily was 42 minutes. The data were taken from participants in the Greenlight Intervention Study, a randomized trial of an obesity prevention program at four academic pediatric primary care clinics in the United States.
Daily screen time and dietary practices were based on parent reports, and included daily volume of juice, daily counts of fruits and vegetables, daily count of junk foods such as chips, ice cream, French fries, and fast food, and consumption of sugar-sweetened beverages. The cross-sectional analysis controlled for race/ethnicity, Women, Infants, and Children Program benefits, number of children at home, caregiver education level, and family income.
In adjusted analysis, more than an hour of TV time was significantly associated with junk food intake, with odds ratios of 1.12 for 90 minutes and 1.25 for 120 minutes (P < .05 for both). Similar associations were seen for TV times of 90 minutes and 120 minutes and intake of fast food and sugar-sweetened beverages.
Additionally, the researchers found that toddlers who watched TV during mealtimes were more than twice as likely to consume sugar-sweetened beverages (OR, 2.74), junk food (OR, 2.72), fast food (OR, 2.09), and only about half as likely to consume fruits and vegetables (OR, 0.62).
The study findings were limited by several factors including the cross-sectional design, the reliance on caregiver self-reports, potential for residual confounding, and the low average screen time, Dr. Lutz noted.
However, the results suggest that “increased screen TV time and mealtime TV were both associated with poor dietary practices in 2-year-old children,” she said.
Future research should include analysis of passive screen time, as well as the relationship between screen time and diet with other digital devices beyond TV, she added.
COVID drove screen time higher
The current study is especially important at this time because of the increased screen exposure for many young children in the wake of the ongoing pandemic, Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn., said in an interview. “Screen time use is up even more than before [the pandemic], and this study is a reminder to ask parents of young children about screen time and dietary history.”
Dr. Kinsella said she was not surprised by the study findings. In her practice, “I see families with more screen time use in general who also are more likely to have juice and junk food available. If kids had no access to screens, I believe they would still have access to unhealthy foods. I believe more research is needed into why screen time is so high in some families.”
The study received funding from NIH. The researchers had no financial conflicts to disclose. Dr. Kinsella had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
FROM PAS 2022
Consider climate change in pediatric clinical visits
Climate-informed clinical encounters have the potential to enhance pediatric care in a variety of ways, according to Aaron Bernstein, MD, of Boston Children’s Hospital.
“Each primary care visit offers opportunities to screen for and support children burdened with risks to health that are increasingly intense due to climate change,” Rebecca P. Philipsborn, MD, of Emory University, Atlanta, and colleagues wrote in “A pediatrician’s guide to climate change–informed primary care,” on which Dr. Bernstein served as corresponding author (Curr Probl Pediatr Adolesc Health Care. 2021 June. doi: 10.1016/j.cppeds.2021.101027).
In a presentation at the annual meeting of the Pediatric Academic Societies, Dr. Bernstein highlighted five components of climate-informed pediatric care mentioned in the article: climate-informed screening, health promotion that includes health and climate benefits, care management that anticipates climate risks for at-risk children, climate-informed anticipatory guidance, and engagement with community resources and advocacy.
Pediatricians can incorporate climate-related issues into screening protocols by asking patients about their home environment, Dr. Bernstein said. Potential questions to ask include whether the family has air conditioning in the home, and whether they are concerned about being able to pay the bill if they use air conditioning, he said.
Health promotion discussions during clinical encounters can emphasize that eating more fruits and vegetables not only is good for the health of the child and the whole family, but “also is good for the planet we live on,” he said.
Care management strategies should anticipate climate risks for at-risk children, such those with complex or chronic medical conditions, and outdoor athletes for whom air quality might be an issue, he said.
Medication management has a climate-informed aspect, Dr. Bernstein said. “How safe are the medications you prescribe?” he asked. During the summer months, the relative risk of hospitalization with heat exposure is increased for a range of drugs including ACE inhibitors (RR 1.42), loop diuretics (RR 1.52), stimulants (RR 1.53), anticholinergics (RR 1.26), antipsychotics (RR 1.51), and beta-blockers (RR 1.08), he noted.
For children who play outdoor sports, previous studies suggest they acclimatize for approximately 7 days if traveling prior to vigorous exercise outdoors. “Monitor the heat index and limit the intensity or length of exercise on extreme heat index days,” Dr. Bernstein said. He emphasized the need to remind children and parents to try to limit intense physical activity to the coolest parts of the day, before 10 a.m. and after 4 p.m., to wear sunscreen and light-colored, lightweight clothing, and to drink 5-8 ounces of fluid every 20 minutes during exercise.
Approximately 12% of all-cause attributable fractions of emergency department visits are associated with heat exposure, Dr. Bernstein added. He recommended that pediatricians and patients be aware of airnow.gov and iqair.com as resources to monitor air quality. Pay attention to the heat index, which factors in humidity and presents the real-feel temperature, not just the thermometer reading.
Last but not least, Dr. Bernstein explained that pediatricians can use a clinical visit to ask adolescent patients about civic engagement, and offer resources for those who want to learn more about climate change, such as climatechangeresources.org/organizations-kids/.
For more detailed guidance, Dr. Bernstein recommended “A pediatrician’s guide to climate change–informed primary care.”
Use websites and handouts
Including climate issues in pediatric visits is definitely important, Suzanne Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H., said in an interview.
“Some questions are more critical in warm weather environments where children are at higher risk for dehydration and heat exposure,” said Dr. Boulter, who was not involved with the guide preparation. “The list of suggestions for participation in outdoor sports in hot weather is comprehensive, and the data on ER visits in summer months is surprising,” she noted. However, some of the data could have included more explanation, such as what air quality actually measures, and the difference between ambient temperature and heat index, and how they are calculated, she noted.
Questions about diet, air conditioning, and backup power sources may be covered in other areas of the pediatric visit, or on questionnaires prior to the visit, Dr. Boulter added.
The main barrier to incorporating climate-related information during the pediatric visit is the limited time allotted for the visit and number of topics to address, said Dr. Boulter. “Pediatric practices that have websites could post seasonal reminders about sports participation health in hot weather, or have printed brief handouts for patients in the office,” she noted. Alternatively, guidance about sports and the impact of climate could easily be given as a short handout to families during the health visit, she said.
Future research might include a focus on assessing families’ knowledge and behavior before and after climate change counseling, Dr. Boulter added.
Dr. Bernstein and Dr. Boulter had no financial conflicts to disclose. Dr. Boulter serves on the editorial advisory board of Pediatric News.
Climate-informed clinical encounters have the potential to enhance pediatric care in a variety of ways, according to Aaron Bernstein, MD, of Boston Children’s Hospital.
“Each primary care visit offers opportunities to screen for and support children burdened with risks to health that are increasingly intense due to climate change,” Rebecca P. Philipsborn, MD, of Emory University, Atlanta, and colleagues wrote in “A pediatrician’s guide to climate change–informed primary care,” on which Dr. Bernstein served as corresponding author (Curr Probl Pediatr Adolesc Health Care. 2021 June. doi: 10.1016/j.cppeds.2021.101027).
In a presentation at the annual meeting of the Pediatric Academic Societies, Dr. Bernstein highlighted five components of climate-informed pediatric care mentioned in the article: climate-informed screening, health promotion that includes health and climate benefits, care management that anticipates climate risks for at-risk children, climate-informed anticipatory guidance, and engagement with community resources and advocacy.
Pediatricians can incorporate climate-related issues into screening protocols by asking patients about their home environment, Dr. Bernstein said. Potential questions to ask include whether the family has air conditioning in the home, and whether they are concerned about being able to pay the bill if they use air conditioning, he said.
Health promotion discussions during clinical encounters can emphasize that eating more fruits and vegetables not only is good for the health of the child and the whole family, but “also is good for the planet we live on,” he said.
Care management strategies should anticipate climate risks for at-risk children, such those with complex or chronic medical conditions, and outdoor athletes for whom air quality might be an issue, he said.
Medication management has a climate-informed aspect, Dr. Bernstein said. “How safe are the medications you prescribe?” he asked. During the summer months, the relative risk of hospitalization with heat exposure is increased for a range of drugs including ACE inhibitors (RR 1.42), loop diuretics (RR 1.52), stimulants (RR 1.53), anticholinergics (RR 1.26), antipsychotics (RR 1.51), and beta-blockers (RR 1.08), he noted.
For children who play outdoor sports, previous studies suggest they acclimatize for approximately 7 days if traveling prior to vigorous exercise outdoors. “Monitor the heat index and limit the intensity or length of exercise on extreme heat index days,” Dr. Bernstein said. He emphasized the need to remind children and parents to try to limit intense physical activity to the coolest parts of the day, before 10 a.m. and after 4 p.m., to wear sunscreen and light-colored, lightweight clothing, and to drink 5-8 ounces of fluid every 20 minutes during exercise.
Approximately 12% of all-cause attributable fractions of emergency department visits are associated with heat exposure, Dr. Bernstein added. He recommended that pediatricians and patients be aware of airnow.gov and iqair.com as resources to monitor air quality. Pay attention to the heat index, which factors in humidity and presents the real-feel temperature, not just the thermometer reading.
Last but not least, Dr. Bernstein explained that pediatricians can use a clinical visit to ask adolescent patients about civic engagement, and offer resources for those who want to learn more about climate change, such as climatechangeresources.org/organizations-kids/.
For more detailed guidance, Dr. Bernstein recommended “A pediatrician’s guide to climate change–informed primary care.”
Use websites and handouts
Including climate issues in pediatric visits is definitely important, Suzanne Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H., said in an interview.
“Some questions are more critical in warm weather environments where children are at higher risk for dehydration and heat exposure,” said Dr. Boulter, who was not involved with the guide preparation. “The list of suggestions for participation in outdoor sports in hot weather is comprehensive, and the data on ER visits in summer months is surprising,” she noted. However, some of the data could have included more explanation, such as what air quality actually measures, and the difference between ambient temperature and heat index, and how they are calculated, she noted.
Questions about diet, air conditioning, and backup power sources may be covered in other areas of the pediatric visit, or on questionnaires prior to the visit, Dr. Boulter added.
The main barrier to incorporating climate-related information during the pediatric visit is the limited time allotted for the visit and number of topics to address, said Dr. Boulter. “Pediatric practices that have websites could post seasonal reminders about sports participation health in hot weather, or have printed brief handouts for patients in the office,” she noted. Alternatively, guidance about sports and the impact of climate could easily be given as a short handout to families during the health visit, she said.
Future research might include a focus on assessing families’ knowledge and behavior before and after climate change counseling, Dr. Boulter added.
Dr. Bernstein and Dr. Boulter had no financial conflicts to disclose. Dr. Boulter serves on the editorial advisory board of Pediatric News.
Climate-informed clinical encounters have the potential to enhance pediatric care in a variety of ways, according to Aaron Bernstein, MD, of Boston Children’s Hospital.
“Each primary care visit offers opportunities to screen for and support children burdened with risks to health that are increasingly intense due to climate change,” Rebecca P. Philipsborn, MD, of Emory University, Atlanta, and colleagues wrote in “A pediatrician’s guide to climate change–informed primary care,” on which Dr. Bernstein served as corresponding author (Curr Probl Pediatr Adolesc Health Care. 2021 June. doi: 10.1016/j.cppeds.2021.101027).
In a presentation at the annual meeting of the Pediatric Academic Societies, Dr. Bernstein highlighted five components of climate-informed pediatric care mentioned in the article: climate-informed screening, health promotion that includes health and climate benefits, care management that anticipates climate risks for at-risk children, climate-informed anticipatory guidance, and engagement with community resources and advocacy.
Pediatricians can incorporate climate-related issues into screening protocols by asking patients about their home environment, Dr. Bernstein said. Potential questions to ask include whether the family has air conditioning in the home, and whether they are concerned about being able to pay the bill if they use air conditioning, he said.
Health promotion discussions during clinical encounters can emphasize that eating more fruits and vegetables not only is good for the health of the child and the whole family, but “also is good for the planet we live on,” he said.
Care management strategies should anticipate climate risks for at-risk children, such those with complex or chronic medical conditions, and outdoor athletes for whom air quality might be an issue, he said.
Medication management has a climate-informed aspect, Dr. Bernstein said. “How safe are the medications you prescribe?” he asked. During the summer months, the relative risk of hospitalization with heat exposure is increased for a range of drugs including ACE inhibitors (RR 1.42), loop diuretics (RR 1.52), stimulants (RR 1.53), anticholinergics (RR 1.26), antipsychotics (RR 1.51), and beta-blockers (RR 1.08), he noted.
For children who play outdoor sports, previous studies suggest they acclimatize for approximately 7 days if traveling prior to vigorous exercise outdoors. “Monitor the heat index and limit the intensity or length of exercise on extreme heat index days,” Dr. Bernstein said. He emphasized the need to remind children and parents to try to limit intense physical activity to the coolest parts of the day, before 10 a.m. and after 4 p.m., to wear sunscreen and light-colored, lightweight clothing, and to drink 5-8 ounces of fluid every 20 minutes during exercise.
Approximately 12% of all-cause attributable fractions of emergency department visits are associated with heat exposure, Dr. Bernstein added. He recommended that pediatricians and patients be aware of airnow.gov and iqair.com as resources to monitor air quality. Pay attention to the heat index, which factors in humidity and presents the real-feel temperature, not just the thermometer reading.
Last but not least, Dr. Bernstein explained that pediatricians can use a clinical visit to ask adolescent patients about civic engagement, and offer resources for those who want to learn more about climate change, such as climatechangeresources.org/organizations-kids/.
For more detailed guidance, Dr. Bernstein recommended “A pediatrician’s guide to climate change–informed primary care.”
Use websites and handouts
Including climate issues in pediatric visits is definitely important, Suzanne Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H., said in an interview.
“Some questions are more critical in warm weather environments where children are at higher risk for dehydration and heat exposure,” said Dr. Boulter, who was not involved with the guide preparation. “The list of suggestions for participation in outdoor sports in hot weather is comprehensive, and the data on ER visits in summer months is surprising,” she noted. However, some of the data could have included more explanation, such as what air quality actually measures, and the difference between ambient temperature and heat index, and how they are calculated, she noted.
Questions about diet, air conditioning, and backup power sources may be covered in other areas of the pediatric visit, or on questionnaires prior to the visit, Dr. Boulter added.
The main barrier to incorporating climate-related information during the pediatric visit is the limited time allotted for the visit and number of topics to address, said Dr. Boulter. “Pediatric practices that have websites could post seasonal reminders about sports participation health in hot weather, or have printed brief handouts for patients in the office,” she noted. Alternatively, guidance about sports and the impact of climate could easily be given as a short handout to families during the health visit, she said.
Future research might include a focus on assessing families’ knowledge and behavior before and after climate change counseling, Dr. Boulter added.
Dr. Bernstein and Dr. Boulter had no financial conflicts to disclose. Dr. Boulter serves on the editorial advisory board of Pediatric News.
FROM PAS 2022
Furosemide seen as safe for preventing newborn lung disease
A medication used to reduce fluid retention can also safely be used to prevent a dangerous lung condition that affects newborns, particularly those born premature, according to a new study.
Furosemide (Lasix) – which can reduce excess fluid in the body caused by heart failure, liver disease, and kidney trouble – is commonly used off-label to prevent bronchopulmonary dysplasia (BPD), a disorder that causes irritation and poor development of lungs in premature infants. But until now, researchers have not studied its safety in this setting.
BPD often affects babies born more than 2 months early and can sometimes result in breathing difficulties into adolescence and young adulthood.
“There are so few drugs that have been tested for newborns, and there are very little data to help neonatologists decide if certain medications are safe and effective,” said Rachel Greenberg, MD, MHS, a neonatologist and member of the Duke Clinical Research Institute, Durham, N.C. “We found there was no greater risk of safety events for newborns given furosemide.”
Dr. Greenberg presented the findings at the 2022 Pediatric Academic Societies meeting in Denver.
For the 28-day randomized controlled trial, Dr. Greenberg and colleagues enrolled 80 preterm newborns, born at less than 29 weeks’ gestation, at 17 centers within the Eunice Kennedy Shriver National Institute of Child Health and Human Development Pediatric Trials Network. Of those, 61 received furosemide and 19 received a placebo.
Although babies given furosemide had more problems with electrolytes – an expected outcome from the use of diuretic medications – the researchers observed no greater risk for more serious issues, namely hearing loss or kidney stones, Dr. Greenberg told this news organization.
“The mechanism here is we know that extra fluid can damage the lungs and can cause you to have to use more respiratory support and more oxygen,” she said. “The thought from a physiological standpoint is using a diuretic can decrease fluid in the lungs and lead to improvements in lung outcomes.”
The researchers did not observe a reduction in BDP or death in babies who received furosemide, but Dr. Greenberg said the study was underpowered to detect such an effect.
“We were not powered to detect a difference in that outcome; the overall objective of this study was always to evaluate safety,” she said. “Of course, we wanted to capture variables that would measure effectiveness as well.
“Because this was a pragmatic trial, we did not limit the amount of fluids that the clinicians could give the participating infants. This could have impacted the effectiveness of furosemide. We would need a different design and larger study to truly determine effectiveness.”
Dr. Greenberg said she hoped the new data will provide greater insight to neonatal providers and help bolster future, more large-scale trials using furosemide in premature infants.
The drug has previously been associated with both kidney stones and ototoxicity, which occurs when medication causes a person to develop hearing or balance problems, said Nicolas Bamat, MD, MSCE, assistant professor of pediatrics at the Perelman School of Medicine, University of Pennsylvania, Philadelphia.
Although the number of children in the latest study was too small to generate any firm conclusions, he said, the trial provides the best data to date on furosemide in premature infants.
The medication is used frequently both on babies at risk of developing BPD and babies who have already reached BPD status. Among newborns with highest risk of dying, furosemide is indeed the “most frequently used pharmacotherapy,” Dr. Bamat said.
“What’s worth noting is that furosemide is an old medication that has been used extensively in the neonatal populations for 40 years, and that is occurring in the absence of data,” Dr. Bamat added. “This is a very important step forward.”
Dr. Greenberg and Dr. Bamat have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A medication used to reduce fluid retention can also safely be used to prevent a dangerous lung condition that affects newborns, particularly those born premature, according to a new study.
Furosemide (Lasix) – which can reduce excess fluid in the body caused by heart failure, liver disease, and kidney trouble – is commonly used off-label to prevent bronchopulmonary dysplasia (BPD), a disorder that causes irritation and poor development of lungs in premature infants. But until now, researchers have not studied its safety in this setting.
BPD often affects babies born more than 2 months early and can sometimes result in breathing difficulties into adolescence and young adulthood.
“There are so few drugs that have been tested for newborns, and there are very little data to help neonatologists decide if certain medications are safe and effective,” said Rachel Greenberg, MD, MHS, a neonatologist and member of the Duke Clinical Research Institute, Durham, N.C. “We found there was no greater risk of safety events for newborns given furosemide.”
Dr. Greenberg presented the findings at the 2022 Pediatric Academic Societies meeting in Denver.
For the 28-day randomized controlled trial, Dr. Greenberg and colleagues enrolled 80 preterm newborns, born at less than 29 weeks’ gestation, at 17 centers within the Eunice Kennedy Shriver National Institute of Child Health and Human Development Pediatric Trials Network. Of those, 61 received furosemide and 19 received a placebo.
Although babies given furosemide had more problems with electrolytes – an expected outcome from the use of diuretic medications – the researchers observed no greater risk for more serious issues, namely hearing loss or kidney stones, Dr. Greenberg told this news organization.
“The mechanism here is we know that extra fluid can damage the lungs and can cause you to have to use more respiratory support and more oxygen,” she said. “The thought from a physiological standpoint is using a diuretic can decrease fluid in the lungs and lead to improvements in lung outcomes.”
The researchers did not observe a reduction in BDP or death in babies who received furosemide, but Dr. Greenberg said the study was underpowered to detect such an effect.
“We were not powered to detect a difference in that outcome; the overall objective of this study was always to evaluate safety,” she said. “Of course, we wanted to capture variables that would measure effectiveness as well.
“Because this was a pragmatic trial, we did not limit the amount of fluids that the clinicians could give the participating infants. This could have impacted the effectiveness of furosemide. We would need a different design and larger study to truly determine effectiveness.”
Dr. Greenberg said she hoped the new data will provide greater insight to neonatal providers and help bolster future, more large-scale trials using furosemide in premature infants.
The drug has previously been associated with both kidney stones and ototoxicity, which occurs when medication causes a person to develop hearing or balance problems, said Nicolas Bamat, MD, MSCE, assistant professor of pediatrics at the Perelman School of Medicine, University of Pennsylvania, Philadelphia.
Although the number of children in the latest study was too small to generate any firm conclusions, he said, the trial provides the best data to date on furosemide in premature infants.
The medication is used frequently both on babies at risk of developing BPD and babies who have already reached BPD status. Among newborns with highest risk of dying, furosemide is indeed the “most frequently used pharmacotherapy,” Dr. Bamat said.
“What’s worth noting is that furosemide is an old medication that has been used extensively in the neonatal populations for 40 years, and that is occurring in the absence of data,” Dr. Bamat added. “This is a very important step forward.”
Dr. Greenberg and Dr. Bamat have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A medication used to reduce fluid retention can also safely be used to prevent a dangerous lung condition that affects newborns, particularly those born premature, according to a new study.
Furosemide (Lasix) – which can reduce excess fluid in the body caused by heart failure, liver disease, and kidney trouble – is commonly used off-label to prevent bronchopulmonary dysplasia (BPD), a disorder that causes irritation and poor development of lungs in premature infants. But until now, researchers have not studied its safety in this setting.
BPD often affects babies born more than 2 months early and can sometimes result in breathing difficulties into adolescence and young adulthood.
“There are so few drugs that have been tested for newborns, and there are very little data to help neonatologists decide if certain medications are safe and effective,” said Rachel Greenberg, MD, MHS, a neonatologist and member of the Duke Clinical Research Institute, Durham, N.C. “We found there was no greater risk of safety events for newborns given furosemide.”
Dr. Greenberg presented the findings at the 2022 Pediatric Academic Societies meeting in Denver.
For the 28-day randomized controlled trial, Dr. Greenberg and colleagues enrolled 80 preterm newborns, born at less than 29 weeks’ gestation, at 17 centers within the Eunice Kennedy Shriver National Institute of Child Health and Human Development Pediatric Trials Network. Of those, 61 received furosemide and 19 received a placebo.
Although babies given furosemide had more problems with electrolytes – an expected outcome from the use of diuretic medications – the researchers observed no greater risk for more serious issues, namely hearing loss or kidney stones, Dr. Greenberg told this news organization.
“The mechanism here is we know that extra fluid can damage the lungs and can cause you to have to use more respiratory support and more oxygen,” she said. “The thought from a physiological standpoint is using a diuretic can decrease fluid in the lungs and lead to improvements in lung outcomes.”
The researchers did not observe a reduction in BDP or death in babies who received furosemide, but Dr. Greenberg said the study was underpowered to detect such an effect.
“We were not powered to detect a difference in that outcome; the overall objective of this study was always to evaluate safety,” she said. “Of course, we wanted to capture variables that would measure effectiveness as well.
“Because this was a pragmatic trial, we did not limit the amount of fluids that the clinicians could give the participating infants. This could have impacted the effectiveness of furosemide. We would need a different design and larger study to truly determine effectiveness.”
Dr. Greenberg said she hoped the new data will provide greater insight to neonatal providers and help bolster future, more large-scale trials using furosemide in premature infants.
The drug has previously been associated with both kidney stones and ototoxicity, which occurs when medication causes a person to develop hearing or balance problems, said Nicolas Bamat, MD, MSCE, assistant professor of pediatrics at the Perelman School of Medicine, University of Pennsylvania, Philadelphia.
Although the number of children in the latest study was too small to generate any firm conclusions, he said, the trial provides the best data to date on furosemide in premature infants.
The medication is used frequently both on babies at risk of developing BPD and babies who have already reached BPD status. Among newborns with highest risk of dying, furosemide is indeed the “most frequently used pharmacotherapy,” Dr. Bamat said.
“What’s worth noting is that furosemide is an old medication that has been used extensively in the neonatal populations for 40 years, and that is occurring in the absence of data,” Dr. Bamat added. “This is a very important step forward.”
Dr. Greenberg and Dr. Bamat have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PAS 2022
Firearm counseling in the ED could be lifesaving for teens
Caregivers who brought suicidal adolescents to the emergency department reported safer gun storage practices after firearm counseling – a crucial way to cut gun deaths among children, according to researchers from Cincinnati Children’s Hospital.
In the study, which took place between June 2021 and Feb 2022, gun safety counseling and handouts were provided to 99 families of children who had come to the ED with mental health problems. A separate set of 101 families in similar situations received counseling and handouts, along with two cable-style gun locks.
Four weeks later, parents in both groups reported an increase in safe storage practices in which they locked away all guns in the household. Those offered only counseling increased safe storage by 7.2% – from 89.9% to 97.1%.
The gains were greater for families that received locks in addition to counseling. The number of those who locked away all guns rose from 82.2% to 98.5% – a 16.3% increase. (Roughly one-third of families in both arms of the study were lost to follow-up, according to the researchers, which left 68 families in each group for analysis.)
Several caregivers in each group reported that guns had been removed entirely from the home, and more than 60% in each group said they had bought additional gun locks to secure their weapons.
“The main point of our study is that just-in-time counseling is very effective in helping these families of children with mental health concerns in securing all their guns, and an emergency department visit is a great time to do that,” said Bijan Ketabchi, MD, a clinical fellow in the division of emergency medicine at Cincinnati Children’s Hospital Medical Center, who presented the findings at the Pediatric Academic Societies annual meeting.
Dr. Ketabchi said his department sees 500-700 children each month with mental health concerns, most commonly depression. The mean age of adolescent patients in the study was 14 years.
Suicide is the second-leading cause of death among children in the United States. Both pediatric suicides and firearm suicides have increased in the past 2 decades, Dr. Ketabchi said. The number of youth suicides who use guns has risen 90% since 2008. One in three U.S. families own a firearm, and 4.6 million children live in a home with loaded, unlocked guns.
Among children aged 17 years and younger who die by firearm suicide, 82% used guns belonging to a family member.
The right time for the message
Interventions to encourage safe gun storage – at a time when caregivers are really listening – can be lifesaving, Dr. Ketabchi said.
“We know that counseling is really helpful for these families, because when they come to the emergency department with a concern, they can have a teachable moment,” he said in an interview. “It resonates with them a lot more than it normally would because they have experienced something traumatic.”
The importance of safe gun storage in households with adolescents can’t be overstated, even if the children are not at risk of suicide, said Naoka Carey, a doctoral candidate at Boston College.
Ms. Carey authored an article on the prevalence of handguns among adolescents that will be published in May in Pediatrics.
“Three kinds of harm for adolescents with access to guns are accidental injury, homicide, and suicide,” she said. “Families who own guns don’t always know their teens have access to the guns.”
The problem is getting worse. Ms. Carey and colleagues found that, between 2002 and 2019, the rate of children aged 12-17 who reported carrying handguns increased 41%. Most of them were White, and their families were in high-income brackets. New data show that firearm injuries have become the leading cause of death among youth in the United States, eclipsing auto accidents for the first time.
“Preventing tragedy in your family is more than reason enough to secure guns you have,” she said.
Dr. Ketabchi disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Caregivers who brought suicidal adolescents to the emergency department reported safer gun storage practices after firearm counseling – a crucial way to cut gun deaths among children, according to researchers from Cincinnati Children’s Hospital.
In the study, which took place between June 2021 and Feb 2022, gun safety counseling and handouts were provided to 99 families of children who had come to the ED with mental health problems. A separate set of 101 families in similar situations received counseling and handouts, along with two cable-style gun locks.
Four weeks later, parents in both groups reported an increase in safe storage practices in which they locked away all guns in the household. Those offered only counseling increased safe storage by 7.2% – from 89.9% to 97.1%.
The gains were greater for families that received locks in addition to counseling. The number of those who locked away all guns rose from 82.2% to 98.5% – a 16.3% increase. (Roughly one-third of families in both arms of the study were lost to follow-up, according to the researchers, which left 68 families in each group for analysis.)
Several caregivers in each group reported that guns had been removed entirely from the home, and more than 60% in each group said they had bought additional gun locks to secure their weapons.
“The main point of our study is that just-in-time counseling is very effective in helping these families of children with mental health concerns in securing all their guns, and an emergency department visit is a great time to do that,” said Bijan Ketabchi, MD, a clinical fellow in the division of emergency medicine at Cincinnati Children’s Hospital Medical Center, who presented the findings at the Pediatric Academic Societies annual meeting.
Dr. Ketabchi said his department sees 500-700 children each month with mental health concerns, most commonly depression. The mean age of adolescent patients in the study was 14 years.
Suicide is the second-leading cause of death among children in the United States. Both pediatric suicides and firearm suicides have increased in the past 2 decades, Dr. Ketabchi said. The number of youth suicides who use guns has risen 90% since 2008. One in three U.S. families own a firearm, and 4.6 million children live in a home with loaded, unlocked guns.
Among children aged 17 years and younger who die by firearm suicide, 82% used guns belonging to a family member.
The right time for the message
Interventions to encourage safe gun storage – at a time when caregivers are really listening – can be lifesaving, Dr. Ketabchi said.
“We know that counseling is really helpful for these families, because when they come to the emergency department with a concern, they can have a teachable moment,” he said in an interview. “It resonates with them a lot more than it normally would because they have experienced something traumatic.”
The importance of safe gun storage in households with adolescents can’t be overstated, even if the children are not at risk of suicide, said Naoka Carey, a doctoral candidate at Boston College.
Ms. Carey authored an article on the prevalence of handguns among adolescents that will be published in May in Pediatrics.
“Three kinds of harm for adolescents with access to guns are accidental injury, homicide, and suicide,” she said. “Families who own guns don’t always know their teens have access to the guns.”
The problem is getting worse. Ms. Carey and colleagues found that, between 2002 and 2019, the rate of children aged 12-17 who reported carrying handguns increased 41%. Most of them were White, and their families were in high-income brackets. New data show that firearm injuries have become the leading cause of death among youth in the United States, eclipsing auto accidents for the first time.
“Preventing tragedy in your family is more than reason enough to secure guns you have,” she said.
Dr. Ketabchi disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Caregivers who brought suicidal adolescents to the emergency department reported safer gun storage practices after firearm counseling – a crucial way to cut gun deaths among children, according to researchers from Cincinnati Children’s Hospital.
In the study, which took place between June 2021 and Feb 2022, gun safety counseling and handouts were provided to 99 families of children who had come to the ED with mental health problems. A separate set of 101 families in similar situations received counseling and handouts, along with two cable-style gun locks.
Four weeks later, parents in both groups reported an increase in safe storage practices in which they locked away all guns in the household. Those offered only counseling increased safe storage by 7.2% – from 89.9% to 97.1%.
The gains were greater for families that received locks in addition to counseling. The number of those who locked away all guns rose from 82.2% to 98.5% – a 16.3% increase. (Roughly one-third of families in both arms of the study were lost to follow-up, according to the researchers, which left 68 families in each group for analysis.)
Several caregivers in each group reported that guns had been removed entirely from the home, and more than 60% in each group said they had bought additional gun locks to secure their weapons.
“The main point of our study is that just-in-time counseling is very effective in helping these families of children with mental health concerns in securing all their guns, and an emergency department visit is a great time to do that,” said Bijan Ketabchi, MD, a clinical fellow in the division of emergency medicine at Cincinnati Children’s Hospital Medical Center, who presented the findings at the Pediatric Academic Societies annual meeting.
Dr. Ketabchi said his department sees 500-700 children each month with mental health concerns, most commonly depression. The mean age of adolescent patients in the study was 14 years.
Suicide is the second-leading cause of death among children in the United States. Both pediatric suicides and firearm suicides have increased in the past 2 decades, Dr. Ketabchi said. The number of youth suicides who use guns has risen 90% since 2008. One in three U.S. families own a firearm, and 4.6 million children live in a home with loaded, unlocked guns.
Among children aged 17 years and younger who die by firearm suicide, 82% used guns belonging to a family member.
The right time for the message
Interventions to encourage safe gun storage – at a time when caregivers are really listening – can be lifesaving, Dr. Ketabchi said.
“We know that counseling is really helpful for these families, because when they come to the emergency department with a concern, they can have a teachable moment,” he said in an interview. “It resonates with them a lot more than it normally would because they have experienced something traumatic.”
The importance of safe gun storage in households with adolescents can’t be overstated, even if the children are not at risk of suicide, said Naoka Carey, a doctoral candidate at Boston College.
Ms. Carey authored an article on the prevalence of handguns among adolescents that will be published in May in Pediatrics.
“Three kinds of harm for adolescents with access to guns are accidental injury, homicide, and suicide,” she said. “Families who own guns don’t always know their teens have access to the guns.”
The problem is getting worse. Ms. Carey and colleagues found that, between 2002 and 2019, the rate of children aged 12-17 who reported carrying handguns increased 41%. Most of them were White, and their families were in high-income brackets. New data show that firearm injuries have become the leading cause of death among youth in the United States, eclipsing auto accidents for the first time.
“Preventing tragedy in your family is more than reason enough to secure guns you have,” she said.
Dr. Ketabchi disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PAS 2022

