User login
Steroid and immunoglobulin standard of care for MIS-C
The combination of methylprednisolone and intravenous immunoglobulins works better than intravenous immunoglobulins alone for multisystem inflammatory syndrome in children (MIS-C), researchers say.
“I’m not sure it’s the best treatment because we have not studied every possible treatment,” François Angoulvant, MD, PhD, told this news organization, “but right now, it’s the standard of care.”
Dr. Angoulvant, a professor of pediatrics at University of Paris, and colleagues published a comparison of the two treatments in the Journal of the American Medical Association.
A small percentage of children infected with SARS-CoV-2 develop MIS-C about 2 to 4 weeks later. It is considered a separate disease entity from COVID-19 and is associated with persistent fever, digestive symptoms, rash, bilateral nonpurulent conjunctivitis, mucocutaneous inflammation signs, and frequent cardiovascular involvement. In more than 60% of cases, it leads to hemodynamic failure, with acute cardiac dysfunction.
Because MIS-C resembles Kawasaki disease, clinicians modeled their treatment on that condition and started with immunoglobulins alone, Dr. Angoulvant said.
Based on expert opinion, the National Health Service in the United Kingdom published a consensus statement in Sept. listing immunoglobulins alone as the first-line treatment.
But anecdotal reports have emerged that combining the immunoglobulins with a corticosteroid worked better. To investigate this possibility, Dr. Angoulvant and colleagues analyzed records of MIS-C cases in France, where physicians are required to report all suspected cases of MIS-C to the French National Public Health Agency.
Among the 181 cases they scrutinized, 111 fulfilled the World Health Organization criteria for MIS-C. Of these, the researchers were able to match 64 patients who had received immunoglobulins alone with 32 who had received the combined therapy and could be matched using propensity scores.
The researchers defined treatment failure as persistence of fever for 2 days after the start of therapy or recurrence of fever within a week. By this measure, the combination treatment failed in only 9% of cases while immunoglobulins alone failed in 38% of cases. The difference was statistically significant (P = .008). Most of those for whom these treatments failed received second-line treatments such as steroids or biological agents.
Patients treated with the combination therapy also had a lower risk of secondary acute left ventricular dysfunction (odds ratio, 0.20; 95% confidence interval, 0.06-0.66) and a lower risk of needing hemodynamic support (OR, 0.21; 95% CI, 0.06-0.76).
Those receiving the combination therapy spent a mean of 4 days in the pediatric intensive care unit compared with 6 days for those receiving immunoglobulins alone. (Difference in days, −2.4; 95% CI, −4.0 to −0.7; P = .005).
There are few drawbacks to the combination approach, Dr. Angoulvant said, as the side effects of corticosteroids are generally not severe and they can be anticipated because this class of medications has been used for many years.
The study raises the question of whether corticosteroids might work as well by themselves, but it could not be answered with this database as no one is using that approach in France, Dr. Angoulvant said. “I hope other teams around the world could bring us the answer.”
In the United States, most physicians appear to already be using the combination therapy, said David Teachey, MD, an associate professor of pediatrics at the Children’s Hospital of Philadelphia and the University of Pennsylvania, Philadelphia.
The reduction in time in pediatric intensive care and the reduced risk of cardiac dysfunction are important findings, he said.
This retrospective study falls short of the evidence provided by a randomized clinical trial, Dr. Teachey noted. But he acknowledged that few families would agree to participate in such a trial as they would have to take a chance that the sick children would receive a less effective therapy than what they would otherwise get. “It’s hard to [talk] about a therapy reduction,” he told this news organization.
Given that impediment, he agreed with Dr. Angoulvant that the current study and others like it may provide the best data available pointing to a treatment approach for MIS-C.
The study received an unrestricted grant from Pfizer. The French COVID-19 Paediatric Inflammation Consortium received an unrestricted grant from the Square Foundation (Grandir–Fonds de Solidarité pour L’Enfance). Dr. Angoulvant and Dr. Teachey have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The combination of methylprednisolone and intravenous immunoglobulins works better than intravenous immunoglobulins alone for multisystem inflammatory syndrome in children (MIS-C), researchers say.
“I’m not sure it’s the best treatment because we have not studied every possible treatment,” François Angoulvant, MD, PhD, told this news organization, “but right now, it’s the standard of care.”
Dr. Angoulvant, a professor of pediatrics at University of Paris, and colleagues published a comparison of the two treatments in the Journal of the American Medical Association.
A small percentage of children infected with SARS-CoV-2 develop MIS-C about 2 to 4 weeks later. It is considered a separate disease entity from COVID-19 and is associated with persistent fever, digestive symptoms, rash, bilateral nonpurulent conjunctivitis, mucocutaneous inflammation signs, and frequent cardiovascular involvement. In more than 60% of cases, it leads to hemodynamic failure, with acute cardiac dysfunction.
Because MIS-C resembles Kawasaki disease, clinicians modeled their treatment on that condition and started with immunoglobulins alone, Dr. Angoulvant said.
Based on expert opinion, the National Health Service in the United Kingdom published a consensus statement in Sept. listing immunoglobulins alone as the first-line treatment.
But anecdotal reports have emerged that combining the immunoglobulins with a corticosteroid worked better. To investigate this possibility, Dr. Angoulvant and colleagues analyzed records of MIS-C cases in France, where physicians are required to report all suspected cases of MIS-C to the French National Public Health Agency.
Among the 181 cases they scrutinized, 111 fulfilled the World Health Organization criteria for MIS-C. Of these, the researchers were able to match 64 patients who had received immunoglobulins alone with 32 who had received the combined therapy and could be matched using propensity scores.
The researchers defined treatment failure as persistence of fever for 2 days after the start of therapy or recurrence of fever within a week. By this measure, the combination treatment failed in only 9% of cases while immunoglobulins alone failed in 38% of cases. The difference was statistically significant (P = .008). Most of those for whom these treatments failed received second-line treatments such as steroids or biological agents.
Patients treated with the combination therapy also had a lower risk of secondary acute left ventricular dysfunction (odds ratio, 0.20; 95% confidence interval, 0.06-0.66) and a lower risk of needing hemodynamic support (OR, 0.21; 95% CI, 0.06-0.76).
Those receiving the combination therapy spent a mean of 4 days in the pediatric intensive care unit compared with 6 days for those receiving immunoglobulins alone. (Difference in days, −2.4; 95% CI, −4.0 to −0.7; P = .005).
There are few drawbacks to the combination approach, Dr. Angoulvant said, as the side effects of corticosteroids are generally not severe and they can be anticipated because this class of medications has been used for many years.
The study raises the question of whether corticosteroids might work as well by themselves, but it could not be answered with this database as no one is using that approach in France, Dr. Angoulvant said. “I hope other teams around the world could bring us the answer.”
In the United States, most physicians appear to already be using the combination therapy, said David Teachey, MD, an associate professor of pediatrics at the Children’s Hospital of Philadelphia and the University of Pennsylvania, Philadelphia.
The reduction in time in pediatric intensive care and the reduced risk of cardiac dysfunction are important findings, he said.
This retrospective study falls short of the evidence provided by a randomized clinical trial, Dr. Teachey noted. But he acknowledged that few families would agree to participate in such a trial as they would have to take a chance that the sick children would receive a less effective therapy than what they would otherwise get. “It’s hard to [talk] about a therapy reduction,” he told this news organization.
Given that impediment, he agreed with Dr. Angoulvant that the current study and others like it may provide the best data available pointing to a treatment approach for MIS-C.
The study received an unrestricted grant from Pfizer. The French COVID-19 Paediatric Inflammation Consortium received an unrestricted grant from the Square Foundation (Grandir–Fonds de Solidarité pour L’Enfance). Dr. Angoulvant and Dr. Teachey have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The combination of methylprednisolone and intravenous immunoglobulins works better than intravenous immunoglobulins alone for multisystem inflammatory syndrome in children (MIS-C), researchers say.
“I’m not sure it’s the best treatment because we have not studied every possible treatment,” François Angoulvant, MD, PhD, told this news organization, “but right now, it’s the standard of care.”
Dr. Angoulvant, a professor of pediatrics at University of Paris, and colleagues published a comparison of the two treatments in the Journal of the American Medical Association.
A small percentage of children infected with SARS-CoV-2 develop MIS-C about 2 to 4 weeks later. It is considered a separate disease entity from COVID-19 and is associated with persistent fever, digestive symptoms, rash, bilateral nonpurulent conjunctivitis, mucocutaneous inflammation signs, and frequent cardiovascular involvement. In more than 60% of cases, it leads to hemodynamic failure, with acute cardiac dysfunction.
Because MIS-C resembles Kawasaki disease, clinicians modeled their treatment on that condition and started with immunoglobulins alone, Dr. Angoulvant said.
Based on expert opinion, the National Health Service in the United Kingdom published a consensus statement in Sept. listing immunoglobulins alone as the first-line treatment.
But anecdotal reports have emerged that combining the immunoglobulins with a corticosteroid worked better. To investigate this possibility, Dr. Angoulvant and colleagues analyzed records of MIS-C cases in France, where physicians are required to report all suspected cases of MIS-C to the French National Public Health Agency.
Among the 181 cases they scrutinized, 111 fulfilled the World Health Organization criteria for MIS-C. Of these, the researchers were able to match 64 patients who had received immunoglobulins alone with 32 who had received the combined therapy and could be matched using propensity scores.
The researchers defined treatment failure as persistence of fever for 2 days after the start of therapy or recurrence of fever within a week. By this measure, the combination treatment failed in only 9% of cases while immunoglobulins alone failed in 38% of cases. The difference was statistically significant (P = .008). Most of those for whom these treatments failed received second-line treatments such as steroids or biological agents.
Patients treated with the combination therapy also had a lower risk of secondary acute left ventricular dysfunction (odds ratio, 0.20; 95% confidence interval, 0.06-0.66) and a lower risk of needing hemodynamic support (OR, 0.21; 95% CI, 0.06-0.76).
Those receiving the combination therapy spent a mean of 4 days in the pediatric intensive care unit compared with 6 days for those receiving immunoglobulins alone. (Difference in days, −2.4; 95% CI, −4.0 to −0.7; P = .005).
There are few drawbacks to the combination approach, Dr. Angoulvant said, as the side effects of corticosteroids are generally not severe and they can be anticipated because this class of medications has been used for many years.
The study raises the question of whether corticosteroids might work as well by themselves, but it could not be answered with this database as no one is using that approach in France, Dr. Angoulvant said. “I hope other teams around the world could bring us the answer.”
In the United States, most physicians appear to already be using the combination therapy, said David Teachey, MD, an associate professor of pediatrics at the Children’s Hospital of Philadelphia and the University of Pennsylvania, Philadelphia.
The reduction in time in pediatric intensive care and the reduced risk of cardiac dysfunction are important findings, he said.
This retrospective study falls short of the evidence provided by a randomized clinical trial, Dr. Teachey noted. But he acknowledged that few families would agree to participate in such a trial as they would have to take a chance that the sick children would receive a less effective therapy than what they would otherwise get. “It’s hard to [talk] about a therapy reduction,” he told this news organization.
Given that impediment, he agreed with Dr. Angoulvant that the current study and others like it may provide the best data available pointing to a treatment approach for MIS-C.
The study received an unrestricted grant from Pfizer. The French COVID-19 Paediatric Inflammation Consortium received an unrestricted grant from the Square Foundation (Grandir–Fonds de Solidarité pour L’Enfance). Dr. Angoulvant and Dr. Teachey have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Child with yellow nodule
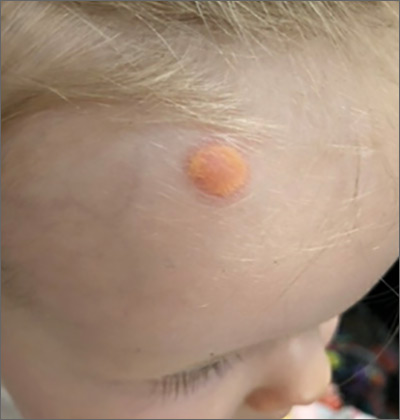
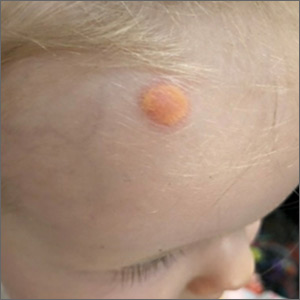
The characteristic orange-yellow color is the tip-off to the diagnosis of juvenile xanthogranuloma (JXG). It manifests as asymptomatic solitary or scattered papules or nodules, congenitally, or most commonly during the first year of life.
JXG is an unusual non-Langerhans cell histiocytosis that more commonly affects males. The etiology of JXG is unclear; it is presumed to be due to physical or infectious stimuli that produce a granulomatous histiocytic reaction. JXG typically manifests on the head, neck, upper extremities, and trunk. The appearance of JXG may be similar to that of Langerhans cell histiocytosis. If necessary, the diagnosis of JXG can be confirmed with a skin biopsy, which will reveal Touton-type giant cells and foamy histiocytes.
JXG is a benign and self-limiting disorder and spontaneously regresses within a few years. In rare cases, it can be systemic. If there are multiple lesions, relevant history, or physical exam features suggesting space-occupying lesions, imaging should be performed to rule out lesions in internal organs or structures. Treatment is indicated when there is systemic or symptomatic ocular involvement and may include surgical excision, radiotherapy, and/or systemic chemotherapy. In this case, the patient’s JXG management involved routine monitoring in anticipation of spontaneous resolution.
Image courtesy of John Durkin, MD, FAAD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kerry Song, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Collie JS, Harper CD, Fillman EP. Juvenile Xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2020 Jan. Accessed January 29, 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/#_NBK526103_pubdet

The characteristic orange-yellow color is the tip-off to the diagnosis of juvenile xanthogranuloma (JXG). It manifests as asymptomatic solitary or scattered papules or nodules, congenitally, or most commonly during the first year of life.
JXG is an unusual non-Langerhans cell histiocytosis that more commonly affects males. The etiology of JXG is unclear; it is presumed to be due to physical or infectious stimuli that produce a granulomatous histiocytic reaction. JXG typically manifests on the head, neck, upper extremities, and trunk. The appearance of JXG may be similar to that of Langerhans cell histiocytosis. If necessary, the diagnosis of JXG can be confirmed with a skin biopsy, which will reveal Touton-type giant cells and foamy histiocytes.
JXG is a benign and self-limiting disorder and spontaneously regresses within a few years. In rare cases, it can be systemic. If there are multiple lesions, relevant history, or physical exam features suggesting space-occupying lesions, imaging should be performed to rule out lesions in internal organs or structures. Treatment is indicated when there is systemic or symptomatic ocular involvement and may include surgical excision, radiotherapy, and/or systemic chemotherapy. In this case, the patient’s JXG management involved routine monitoring in anticipation of spontaneous resolution.
Image courtesy of John Durkin, MD, FAAD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kerry Song, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The characteristic orange-yellow color is the tip-off to the diagnosis of juvenile xanthogranuloma (JXG). It manifests as asymptomatic solitary or scattered papules or nodules, congenitally, or most commonly during the first year of life.
JXG is an unusual non-Langerhans cell histiocytosis that more commonly affects males. The etiology of JXG is unclear; it is presumed to be due to physical or infectious stimuli that produce a granulomatous histiocytic reaction. JXG typically manifests on the head, neck, upper extremities, and trunk. The appearance of JXG may be similar to that of Langerhans cell histiocytosis. If necessary, the diagnosis of JXG can be confirmed with a skin biopsy, which will reveal Touton-type giant cells and foamy histiocytes.
JXG is a benign and self-limiting disorder and spontaneously regresses within a few years. In rare cases, it can be systemic. If there are multiple lesions, relevant history, or physical exam features suggesting space-occupying lesions, imaging should be performed to rule out lesions in internal organs or structures. Treatment is indicated when there is systemic or symptomatic ocular involvement and may include surgical excision, radiotherapy, and/or systemic chemotherapy. In this case, the patient’s JXG management involved routine monitoring in anticipation of spontaneous resolution.
Image courtesy of John Durkin, MD, FAAD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kerry Song, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Collie JS, Harper CD, Fillman EP. Juvenile Xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2020 Jan. Accessed January 29, 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/#_NBK526103_pubdet
Collie JS, Harper CD, Fillman EP. Juvenile Xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2020 Jan. Accessed January 29, 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/#_NBK526103_pubdet
Updated WIC in pregnancy boosts infant outcomes
Developmental outcomes in the first 2 years of life improved in children whose mothers received the revised Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) while pregnant, based on data from approximately 1,200 women.
Maternal nutrition is essential to healthy fetal development, and the WIC was revised in 2009 to align with current dietary guidelines and to support the health of women and children in low-income households, wrote Alice Guan, MPH, of the University of California, San Francisco, and colleagues.
“However, no researchers, to our knowledge, have evaluated effects of this revision on downstream child health or development,” they said.
In a study published in Pediatrics, the researchers reviewed data from mothers and their children who participated in the Conditions Affecting Neurocognitive Development and Learning in Early Childhood (CANDLE) longitudinal cohort study conducted in Tennessee between 2006 and 2011. Their quasi-experimental analysis included 700 women who received WIC during pregnancy and 525 women who did not.
The researchers considered core developmental outcomes of child growth, cognitive development, and socioemotional development at age 12 months and 24 months, and age 4-6 years.
Overall, infants of women who received the WIC food package showed significant increases in length-for-age z scores at 12 months of age (.33, representing approximately one-fifth of a standard deviation), compared to infants of women who did not receive the revised WIC package.
In addition, the Bayley Scales of Infant Development cognitive composite score showed a 4.3-point increase at 24 months of age (approximately one-third of a standard deviation) compared to infants of women who did not receive the revised WIC package.
No effects on growth at age 24 months or on cognitive development at age 4-6 years were noted, which suggests that the impact of the WIC program during pregnancy may fade over time, the researchers said.
“The magnitude of the findings in this study represents clinically relevant effect sizes and provides evidence that one of the largest U.S. safety net policies improves developmental outcomes among low-income and marginalized children,” they noted.
The study findings were limited by several factors including the statistical, quasi-experimental design; the reliance on self-reports for information on income, receipt of WIC, and other variables; and a potential lack of generalizability to other states, the researchers noted. However, the results support findings from previous studies and were strengthened by the review of multiple outcomes and use of a longitudinal database, they said.
“These findings provide timely and critical evidence for the role that WIC plays in improving the health of the nation’s most vulnerable populations, suggesting meaningful impacts of the revised WIC food package on child development,” the researchers said. In addition, “considering the relatively modest scope of the 2009 revision, more substantial updates to the program based on up-to-date nutritional guidance may have substantial effects on improving the health of WIC recipients,” they concluded.
Findings support program’s value
“Pediatrics has always had a commitment to reducing disparities in health care, and we are the main clinicians to see many Medicaid patients on a regular basis,” Herschel Lessin, MD, of Children’s Medical Group, Poughkeepsie, N.Y., said in an interview.
“We all know that pregnant women eating nutritiously ought to help child outcomes, but the current study provides an evidence base for something that seems like common sense,” he noted.
Having such an evidence base is helpful to reinforce the value of the WIC program for its intended recipients, especially in the wake of the COVID-19 pandemic when many funding sources are stretched thin, Dr. Lessin said.
The WIC is intended to try to reduce racial and socioeconomic disparities in the most basic form possible, by helping people who are disadvantaged get enough high-quality food to eat, but results of the program’s impact have not been well studied, he said.
“Outcomes are fiendishly difficult to measure,” and the study is subject to the limitations of its statistical nature, he said. But the large sample size adds support to the findings, which are encouraging, Dr. Lessin noted.
Other potential areas for research include comparing the quality of WIC programs in different states, but such research is very difficult, Dr. Lessin noted. However, the findings might encourage states with less robust WIC programs to consider increasing support, he said.
The study was funded by the National Institutes of Health (National Heart, Lung, and Blood Institute); the National Institute on Aging; the University of California, San Francisco, National Center of Excellence in Women’s Health; and the Urban Child Institute. The researchers had no financial conflicts to disclose. Dr. Lessin serves on the editorial advisory board of Pediatric News and had no relevant financial conflicts to disclose.
Developmental outcomes in the first 2 years of life improved in children whose mothers received the revised Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) while pregnant, based on data from approximately 1,200 women.
Maternal nutrition is essential to healthy fetal development, and the WIC was revised in 2009 to align with current dietary guidelines and to support the health of women and children in low-income households, wrote Alice Guan, MPH, of the University of California, San Francisco, and colleagues.
“However, no researchers, to our knowledge, have evaluated effects of this revision on downstream child health or development,” they said.
In a study published in Pediatrics, the researchers reviewed data from mothers and their children who participated in the Conditions Affecting Neurocognitive Development and Learning in Early Childhood (CANDLE) longitudinal cohort study conducted in Tennessee between 2006 and 2011. Their quasi-experimental analysis included 700 women who received WIC during pregnancy and 525 women who did not.
The researchers considered core developmental outcomes of child growth, cognitive development, and socioemotional development at age 12 months and 24 months, and age 4-6 years.
Overall, infants of women who received the WIC food package showed significant increases in length-for-age z scores at 12 months of age (.33, representing approximately one-fifth of a standard deviation), compared to infants of women who did not receive the revised WIC package.
In addition, the Bayley Scales of Infant Development cognitive composite score showed a 4.3-point increase at 24 months of age (approximately one-third of a standard deviation) compared to infants of women who did not receive the revised WIC package.
No effects on growth at age 24 months or on cognitive development at age 4-6 years were noted, which suggests that the impact of the WIC program during pregnancy may fade over time, the researchers said.
“The magnitude of the findings in this study represents clinically relevant effect sizes and provides evidence that one of the largest U.S. safety net policies improves developmental outcomes among low-income and marginalized children,” they noted.
The study findings were limited by several factors including the statistical, quasi-experimental design; the reliance on self-reports for information on income, receipt of WIC, and other variables; and a potential lack of generalizability to other states, the researchers noted. However, the results support findings from previous studies and were strengthened by the review of multiple outcomes and use of a longitudinal database, they said.
“These findings provide timely and critical evidence for the role that WIC plays in improving the health of the nation’s most vulnerable populations, suggesting meaningful impacts of the revised WIC food package on child development,” the researchers said. In addition, “considering the relatively modest scope of the 2009 revision, more substantial updates to the program based on up-to-date nutritional guidance may have substantial effects on improving the health of WIC recipients,” they concluded.
Findings support program’s value
“Pediatrics has always had a commitment to reducing disparities in health care, and we are the main clinicians to see many Medicaid patients on a regular basis,” Herschel Lessin, MD, of Children’s Medical Group, Poughkeepsie, N.Y., said in an interview.
“We all know that pregnant women eating nutritiously ought to help child outcomes, but the current study provides an evidence base for something that seems like common sense,” he noted.
Having such an evidence base is helpful to reinforce the value of the WIC program for its intended recipients, especially in the wake of the COVID-19 pandemic when many funding sources are stretched thin, Dr. Lessin said.
The WIC is intended to try to reduce racial and socioeconomic disparities in the most basic form possible, by helping people who are disadvantaged get enough high-quality food to eat, but results of the program’s impact have not been well studied, he said.
“Outcomes are fiendishly difficult to measure,” and the study is subject to the limitations of its statistical nature, he said. But the large sample size adds support to the findings, which are encouraging, Dr. Lessin noted.
Other potential areas for research include comparing the quality of WIC programs in different states, but such research is very difficult, Dr. Lessin noted. However, the findings might encourage states with less robust WIC programs to consider increasing support, he said.
The study was funded by the National Institutes of Health (National Heart, Lung, and Blood Institute); the National Institute on Aging; the University of California, San Francisco, National Center of Excellence in Women’s Health; and the Urban Child Institute. The researchers had no financial conflicts to disclose. Dr. Lessin serves on the editorial advisory board of Pediatric News and had no relevant financial conflicts to disclose.
Developmental outcomes in the first 2 years of life improved in children whose mothers received the revised Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) while pregnant, based on data from approximately 1,200 women.
Maternal nutrition is essential to healthy fetal development, and the WIC was revised in 2009 to align with current dietary guidelines and to support the health of women and children in low-income households, wrote Alice Guan, MPH, of the University of California, San Francisco, and colleagues.
“However, no researchers, to our knowledge, have evaluated effects of this revision on downstream child health or development,” they said.
In a study published in Pediatrics, the researchers reviewed data from mothers and their children who participated in the Conditions Affecting Neurocognitive Development and Learning in Early Childhood (CANDLE) longitudinal cohort study conducted in Tennessee between 2006 and 2011. Their quasi-experimental analysis included 700 women who received WIC during pregnancy and 525 women who did not.
The researchers considered core developmental outcomes of child growth, cognitive development, and socioemotional development at age 12 months and 24 months, and age 4-6 years.
Overall, infants of women who received the WIC food package showed significant increases in length-for-age z scores at 12 months of age (.33, representing approximately one-fifth of a standard deviation), compared to infants of women who did not receive the revised WIC package.
In addition, the Bayley Scales of Infant Development cognitive composite score showed a 4.3-point increase at 24 months of age (approximately one-third of a standard deviation) compared to infants of women who did not receive the revised WIC package.
No effects on growth at age 24 months or on cognitive development at age 4-6 years were noted, which suggests that the impact of the WIC program during pregnancy may fade over time, the researchers said.
“The magnitude of the findings in this study represents clinically relevant effect sizes and provides evidence that one of the largest U.S. safety net policies improves developmental outcomes among low-income and marginalized children,” they noted.
The study findings were limited by several factors including the statistical, quasi-experimental design; the reliance on self-reports for information on income, receipt of WIC, and other variables; and a potential lack of generalizability to other states, the researchers noted. However, the results support findings from previous studies and were strengthened by the review of multiple outcomes and use of a longitudinal database, they said.
“These findings provide timely and critical evidence for the role that WIC plays in improving the health of the nation’s most vulnerable populations, suggesting meaningful impacts of the revised WIC food package on child development,” the researchers said. In addition, “considering the relatively modest scope of the 2009 revision, more substantial updates to the program based on up-to-date nutritional guidance may have substantial effects on improving the health of WIC recipients,” they concluded.
Findings support program’s value
“Pediatrics has always had a commitment to reducing disparities in health care, and we are the main clinicians to see many Medicaid patients on a regular basis,” Herschel Lessin, MD, of Children’s Medical Group, Poughkeepsie, N.Y., said in an interview.
“We all know that pregnant women eating nutritiously ought to help child outcomes, but the current study provides an evidence base for something that seems like common sense,” he noted.
Having such an evidence base is helpful to reinforce the value of the WIC program for its intended recipients, especially in the wake of the COVID-19 pandemic when many funding sources are stretched thin, Dr. Lessin said.
The WIC is intended to try to reduce racial and socioeconomic disparities in the most basic form possible, by helping people who are disadvantaged get enough high-quality food to eat, but results of the program’s impact have not been well studied, he said.
“Outcomes are fiendishly difficult to measure,” and the study is subject to the limitations of its statistical nature, he said. But the large sample size adds support to the findings, which are encouraging, Dr. Lessin noted.
Other potential areas for research include comparing the quality of WIC programs in different states, but such research is very difficult, Dr. Lessin noted. However, the findings might encourage states with less robust WIC programs to consider increasing support, he said.
The study was funded by the National Institutes of Health (National Heart, Lung, and Blood Institute); the National Institute on Aging; the University of California, San Francisco, National Center of Excellence in Women’s Health; and the Urban Child Institute. The researchers had no financial conflicts to disclose. Dr. Lessin serves on the editorial advisory board of Pediatric News and had no relevant financial conflicts to disclose.
FROM PEDIATRICS
I Spy, Near the Corner of My Eye
ANSWER
The correct answer—the false statement—is that SA has no pathologic implications (choice “a”).
DISCUSSION
SAs are usually benign and occur in 10% to 15% of the population (particularly in children). But they can sometimes be a sign of serious disease such as liver failure, with related esophageal varices, especially if > 3 lesions are present.
SAs are caused by a failure in the sphincter muscle surrounding a dilated cutaneous arteriole, which in turn is caused by increased estrogen levels in the blood. This increase can be related to the estrogen in birth control medications or to pregnancy.
A diseased liver, unable to metabolize estrogen properly, can contribute to increased blood levels of estrogen. For example, about one-third of patients with cirrhosis will develop multiple SAs.
SAs are seen only in the distribution of the superior vena cava. This means that—in addition to manifesting on the face—they can also appear on the arms, hands, trunk, and fingers.
Momentarily fading completely when central pressure is applied is a peculiar trait of SAs and is therefore diagnostic.
TREATMENT
While these lesions do, in fact, usually resolve on their own, treatment attempts are usually highly satisfactory. In my experience, destruction by laser ablation is superior to electrodessication, though recurrences are common. At the time of this presentation, the patient and her mother were still pondering the treatment options.
ANSWER
The correct answer—the false statement—is that SA has no pathologic implications (choice “a”).
DISCUSSION
SAs are usually benign and occur in 10% to 15% of the population (particularly in children). But they can sometimes be a sign of serious disease such as liver failure, with related esophageal varices, especially if > 3 lesions are present.
SAs are caused by a failure in the sphincter muscle surrounding a dilated cutaneous arteriole, which in turn is caused by increased estrogen levels in the blood. This increase can be related to the estrogen in birth control medications or to pregnancy.
A diseased liver, unable to metabolize estrogen properly, can contribute to increased blood levels of estrogen. For example, about one-third of patients with cirrhosis will develop multiple SAs.
SAs are seen only in the distribution of the superior vena cava. This means that—in addition to manifesting on the face—they can also appear on the arms, hands, trunk, and fingers.
Momentarily fading completely when central pressure is applied is a peculiar trait of SAs and is therefore diagnostic.
TREATMENT
While these lesions do, in fact, usually resolve on their own, treatment attempts are usually highly satisfactory. In my experience, destruction by laser ablation is superior to electrodessication, though recurrences are common. At the time of this presentation, the patient and her mother were still pondering the treatment options.
ANSWER
The correct answer—the false statement—is that SA has no pathologic implications (choice “a”).
DISCUSSION
SAs are usually benign and occur in 10% to 15% of the population (particularly in children). But they can sometimes be a sign of serious disease such as liver failure, with related esophageal varices, especially if > 3 lesions are present.
SAs are caused by a failure in the sphincter muscle surrounding a dilated cutaneous arteriole, which in turn is caused by increased estrogen levels in the blood. This increase can be related to the estrogen in birth control medications or to pregnancy.
A diseased liver, unable to metabolize estrogen properly, can contribute to increased blood levels of estrogen. For example, about one-third of patients with cirrhosis will develop multiple SAs.
SAs are seen only in the distribution of the superior vena cava. This means that—in addition to manifesting on the face—they can also appear on the arms, hands, trunk, and fingers.
Momentarily fading completely when central pressure is applied is a peculiar trait of SAs and is therefore diagnostic.
TREATMENT
While these lesions do, in fact, usually resolve on their own, treatment attempts are usually highly satisfactory. In my experience, destruction by laser ablation is superior to electrodessication, though recurrences are common. At the time of this presentation, the patient and her mother were still pondering the treatment options.
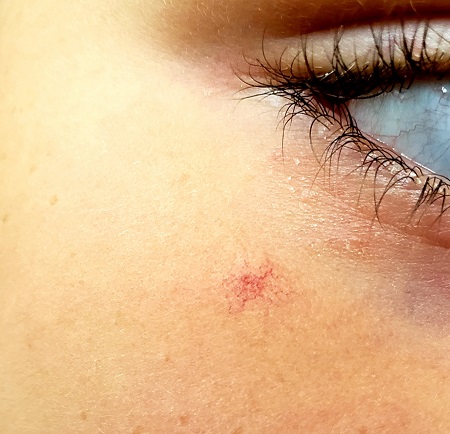
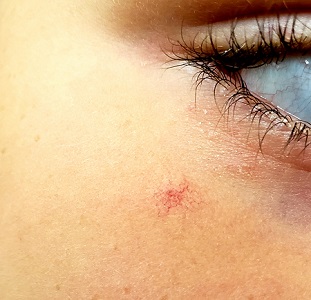
About 3 years ago, an asymptomatic lesion appeared on an 8-year-old girl’s right cheek. Because the spot made her feel self-conscious, the child’s mother had tried covering it up with makeup—but the makeup was even more obvious than the spot.
Their primary care provider (PCP) advised them to do nothing, noting that such lesions usually resolve on their own. The PCP did not believe the lesion was indicative of any related health problems. Dissatisfied with this instruction, the mother brings her daughter to dermatology for evaluation.
The girl is in otherwise good health. The lesion in question is a curious, bright red macule consisting of a tiny pinpoint red dot with very narrow “legs” (tiny, slender, red, vascular lines) emanating from the periphery. It is about 7 mm, and the center red dot is about 1 mm in diameter.
Using a dull pencil to create gentle pinpoint pressure causes the whole lesion to instantly and completely fade, only to return fully after pressure is released. The lesion is diagnosed as a typical spider angioma (SA; also known as spider nevi).
Teenagers get in the queue for COVID-19 vaccines
The vaccinations can’t come soon enough for parents like Stacy Hillenburg, a developmental therapist in Aurora, Ill., whose 9-year-old son takes immunosuppressants because he had a heart transplant when he was 7 weeks old. Although school-age children aren’t yet included in clinical trials, if her 12- and 13-year-old daughters could get vaccinated, along with both parents, then the family could relax some of the protocols they currently follow to prevent infection.
Whenever they are around other people, even masked and socially distanced, they come home and immediately shower and change their clothes. So far, no one in the family has been infected with COVID, but the anxiety is ever-present. “I can’t wait for it to come out,” Ms. Hillenburg said of a pediatric COVID vaccine. “It will ease my mind so much.”
She isn’t alone in that anticipation. In the fall, the American Academy of Pediatrics and other pediatric vaccine experts urged faster action on pediatric vaccine trials and worried that children would be left behind as adults gained protection from COVID. But recent developments have eased those concerns.
“Over the next couple of months, we will be doing trials in an age-deescalation manner,” with studies moving gradually to younger children, Anthony S. Fauci, MD, chief medical adviser on COVID-19 to the president, said in a coronavirus response team briefing on Jan. 29. “So that hopefully, as we get to the late spring and summer, we will have children being able to be vaccinated.”
Pfizer completed enrollment of 2,259 teens aged 12-15 years in late January and expects to move forward with a separate pediatric trial of children aged 5-11 years by this spring, Keanna Ghazvini, senior associate for global media relations at Pfizer, said in an interview.
Enrollment in Moderna’s TeenCove study of adolescents ages 12-17 years began slowly in late December, but the pace has since picked up, said company spokesperson Colleen Hussey. “We continue to bring clinical trial sites online, and we are on track to provide updated data around mid-year 2021.” A trial extension in children 11 years and younger is expected to begin later in 2021.
Johnson & Johnson and AstraZeneca said they expect to begin adolescent trials in early 2021, according to data shared by the Advisory Committee on Immunization Practices. An interim analysis of J&J’s Janssen COVID-19 vaccine trial data, released on Jan. 29, showed it was 72% effective in US participants aged 18 years or older. AstraZeneca’s U.S. trial in adults is ongoing.
Easing the burden
Vaccination could lessen children’s risk of severe disease as well as the social and emotional burdens of the pandemic, says James Campbell, MD, a pediatric infectious disease specialist at the University of Maryland’s Center for Vaccine Development in Baltimore, which was involved in the Moderna and early-phase Pfizer trials. He coauthored a September 2020 article in Clinical Infectious Diseases titled: “Warp Speed for COVID-19 vaccines: Why are children stuck in neutral?”
The adolescent trials are a vital step to ensure timely vaccine access for teens and younger children. “It is reasonable, when you have limited vaccine, that your rollout goes to the highest priority and then moves to lower and lower priorities. In adults, we’re just saying: ‘Wait your turn,’ ” he said of the current vaccination effort. “If we didn’t have the [vaccine trial] data in children, we’d be saying: ‘You don’t have a turn.’ ”
As the pandemic has worn on, the burden on children has grown. As of Tuesday, 269 children had died of COVID-19. That is well above the highest annual death toll recorded during a regular flu season – 188 flu deaths among children and adolescents under 18 in the 2019-2020 and 2017-2018 flu seasons.
Children are less likely to transmit COVID-19 in their household than adults, according to a meta-analysis of 54 studies published in JAMA Network Open. But that does not necessarily mean children are less infectious, the authors said, noting that unmeasured factors could have affected the spread of infection among adults.
Moreover, children and adolescents need protection from COVID infection – and from the potential for severe disease or lingering effects – and, given that there are 74 million children and teens in the United States, their vaccination is an important part of stopping the pandemic, said Grace Lee, MD, professor of pediatrics at Stanford (Calif.) University, and cochair of ACIP’s COVID-19 Vaccine Safety Technical Subgroup.
“In order to interrupt transmission, I don’t see how we’re going to do that without vaccinating children and adolescents,” she said.
Dr. Lee said her 16-year-old daughter misses the normal teenage social life and is excited about getting the vaccine when she is eligible. (Adolescents without high-risk conditions are in the lowest vaccination tier, according to ACIP recommendations.) “There is truly individual protection to be gained,” Dr. Lee said.
She noted that researchers continue to assess the immune responses to the adult vaccines – even looking at immune characteristics of the small percentage of people who aren’t protected from infection – and that information helps in the evaluation of the pediatric immune responses. As the trials expand to younger children and infants, dosing will be a major focus. “How many doses do they need they need to receive the same immunity? Safety considerations will be critically important,” she said.
Teen trials underway
Pfizer/BioNTech extended its adult trial to 16- and 17-year-olds in October, which enabled older teens to be included in its emergency-use authorization. They and younger teens, ages 12-15, receive the same dose as adults.
The ongoing trials with Pfizer and Moderna vaccines are immunobridging trials, designed to study safety and immunogenicity. Investigators will compare the teens’ immune response with the findings from the larger adult trials. When the trials expand to school-age children (6-12 years), protocols call for testing the safety and immunogenicity of a half-dose vaccine as well as the full dose.
Children ages 2-5 years and infants and toddlers will be enrolled in future trials, studying safety and immunogenicity of full, half, or even quarter dosages. The Pediatric Research Equity Act of 2003 requires licensed vaccines to be tested for safety and efficacy in children, unless they are not appropriate for a pediatric population.
Demand for the teen trials has been strong. At Cincinnati Children’s Hospital Medical Center, 259 teenagers joined the Pfizer/BioNTech trial, but some teenagers were turned away when the trial’s national enrollment closed in late January.
“Many of the children are having no side effects, and if they are, they’re having the same [effects] as the young adults – local redness or pain, fatigue, and headaches,” said Robert Frenck, MD, director of the Cincinnati Children’s Gamble Program for Clinical Studies.
Parents may share some of the vaccine hesitancy that has affected adult vaccination. But that is balanced by the hope that vaccines will end the pandemic and usher in a new normal. “If it looks like [vaccines] will increase the likelihood of children returning to school safely, that may be a motivating factor,” Dr. Frenck said.
Cody Meissner, MD, chief of the pediatric infectious disease service at Tufts Medical Center, Boston, was initially cautious about the extension of vaccination to adolescents. A member of the Vaccine and Related Biological Products Advisory Committee, which evaluates data and makes recommendations to the Food and Drug Administration, Dr. Meissner initially abstained in the vote on the Pfizer/BioNTech emergency-use authorization for people 16 and older.
He noted that, at the time the committee reviewed the Pfizer vaccine, the company had data available for just 134 teenagers, half of whom received a placebo. But the vaccination of 34 million adults has provided robust data about the vaccine’s safety, and the trial expansion into adolescents is important.
“I’m comfortable with the way these trials are going now,” he said. “This is the way I was hoping they would go.”
Ms. Hillenburg is on the parent advisory board of Voices for Vaccines, an organization of parents supporting vaccination that is affiliated with the Task Force for Global Health, an Atlanta-based independent public health organization. Dr. Campbell’s institution has received funds to conduct clinical trials from the National Institutes of Health and several companies, including Merck, GlaxoSmithKline, Sanofi, Pfizer, and Moderna. He has served pro bono on many safety and data monitoring committees. Dr. Frenck’s institution is receiving funds to conduct the Pfizer trial. In the past 5 years, he has also participated in clinical trials for GlaxoSmithKline, Merck, and Meissa vaccines. Dr. Lee and Dr. Meissner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The vaccinations can’t come soon enough for parents like Stacy Hillenburg, a developmental therapist in Aurora, Ill., whose 9-year-old son takes immunosuppressants because he had a heart transplant when he was 7 weeks old. Although school-age children aren’t yet included in clinical trials, if her 12- and 13-year-old daughters could get vaccinated, along with both parents, then the family could relax some of the protocols they currently follow to prevent infection.
Whenever they are around other people, even masked and socially distanced, they come home and immediately shower and change their clothes. So far, no one in the family has been infected with COVID, but the anxiety is ever-present. “I can’t wait for it to come out,” Ms. Hillenburg said of a pediatric COVID vaccine. “It will ease my mind so much.”
She isn’t alone in that anticipation. In the fall, the American Academy of Pediatrics and other pediatric vaccine experts urged faster action on pediatric vaccine trials and worried that children would be left behind as adults gained protection from COVID. But recent developments have eased those concerns.
“Over the next couple of months, we will be doing trials in an age-deescalation manner,” with studies moving gradually to younger children, Anthony S. Fauci, MD, chief medical adviser on COVID-19 to the president, said in a coronavirus response team briefing on Jan. 29. “So that hopefully, as we get to the late spring and summer, we will have children being able to be vaccinated.”
Pfizer completed enrollment of 2,259 teens aged 12-15 years in late January and expects to move forward with a separate pediatric trial of children aged 5-11 years by this spring, Keanna Ghazvini, senior associate for global media relations at Pfizer, said in an interview.
Enrollment in Moderna’s TeenCove study of adolescents ages 12-17 years began slowly in late December, but the pace has since picked up, said company spokesperson Colleen Hussey. “We continue to bring clinical trial sites online, and we are on track to provide updated data around mid-year 2021.” A trial extension in children 11 years and younger is expected to begin later in 2021.
Johnson & Johnson and AstraZeneca said they expect to begin adolescent trials in early 2021, according to data shared by the Advisory Committee on Immunization Practices. An interim analysis of J&J’s Janssen COVID-19 vaccine trial data, released on Jan. 29, showed it was 72% effective in US participants aged 18 years or older. AstraZeneca’s U.S. trial in adults is ongoing.
Easing the burden
Vaccination could lessen children’s risk of severe disease as well as the social and emotional burdens of the pandemic, says James Campbell, MD, a pediatric infectious disease specialist at the University of Maryland’s Center for Vaccine Development in Baltimore, which was involved in the Moderna and early-phase Pfizer trials. He coauthored a September 2020 article in Clinical Infectious Diseases titled: “Warp Speed for COVID-19 vaccines: Why are children stuck in neutral?”
The adolescent trials are a vital step to ensure timely vaccine access for teens and younger children. “It is reasonable, when you have limited vaccine, that your rollout goes to the highest priority and then moves to lower and lower priorities. In adults, we’re just saying: ‘Wait your turn,’ ” he said of the current vaccination effort. “If we didn’t have the [vaccine trial] data in children, we’d be saying: ‘You don’t have a turn.’ ”
As the pandemic has worn on, the burden on children has grown. As of Tuesday, 269 children had died of COVID-19. That is well above the highest annual death toll recorded during a regular flu season – 188 flu deaths among children and adolescents under 18 in the 2019-2020 and 2017-2018 flu seasons.
Children are less likely to transmit COVID-19 in their household than adults, according to a meta-analysis of 54 studies published in JAMA Network Open. But that does not necessarily mean children are less infectious, the authors said, noting that unmeasured factors could have affected the spread of infection among adults.
Moreover, children and adolescents need protection from COVID infection – and from the potential for severe disease or lingering effects – and, given that there are 74 million children and teens in the United States, their vaccination is an important part of stopping the pandemic, said Grace Lee, MD, professor of pediatrics at Stanford (Calif.) University, and cochair of ACIP’s COVID-19 Vaccine Safety Technical Subgroup.
“In order to interrupt transmission, I don’t see how we’re going to do that without vaccinating children and adolescents,” she said.
Dr. Lee said her 16-year-old daughter misses the normal teenage social life and is excited about getting the vaccine when she is eligible. (Adolescents without high-risk conditions are in the lowest vaccination tier, according to ACIP recommendations.) “There is truly individual protection to be gained,” Dr. Lee said.
She noted that researchers continue to assess the immune responses to the adult vaccines – even looking at immune characteristics of the small percentage of people who aren’t protected from infection – and that information helps in the evaluation of the pediatric immune responses. As the trials expand to younger children and infants, dosing will be a major focus. “How many doses do they need they need to receive the same immunity? Safety considerations will be critically important,” she said.
Teen trials underway
Pfizer/BioNTech extended its adult trial to 16- and 17-year-olds in October, which enabled older teens to be included in its emergency-use authorization. They and younger teens, ages 12-15, receive the same dose as adults.
The ongoing trials with Pfizer and Moderna vaccines are immunobridging trials, designed to study safety and immunogenicity. Investigators will compare the teens’ immune response with the findings from the larger adult trials. When the trials expand to school-age children (6-12 years), protocols call for testing the safety and immunogenicity of a half-dose vaccine as well as the full dose.
Children ages 2-5 years and infants and toddlers will be enrolled in future trials, studying safety and immunogenicity of full, half, or even quarter dosages. The Pediatric Research Equity Act of 2003 requires licensed vaccines to be tested for safety and efficacy in children, unless they are not appropriate for a pediatric population.
Demand for the teen trials has been strong. At Cincinnati Children’s Hospital Medical Center, 259 teenagers joined the Pfizer/BioNTech trial, but some teenagers were turned away when the trial’s national enrollment closed in late January.
“Many of the children are having no side effects, and if they are, they’re having the same [effects] as the young adults – local redness or pain, fatigue, and headaches,” said Robert Frenck, MD, director of the Cincinnati Children’s Gamble Program for Clinical Studies.
Parents may share some of the vaccine hesitancy that has affected adult vaccination. But that is balanced by the hope that vaccines will end the pandemic and usher in a new normal. “If it looks like [vaccines] will increase the likelihood of children returning to school safely, that may be a motivating factor,” Dr. Frenck said.
Cody Meissner, MD, chief of the pediatric infectious disease service at Tufts Medical Center, Boston, was initially cautious about the extension of vaccination to adolescents. A member of the Vaccine and Related Biological Products Advisory Committee, which evaluates data and makes recommendations to the Food and Drug Administration, Dr. Meissner initially abstained in the vote on the Pfizer/BioNTech emergency-use authorization for people 16 and older.
He noted that, at the time the committee reviewed the Pfizer vaccine, the company had data available for just 134 teenagers, half of whom received a placebo. But the vaccination of 34 million adults has provided robust data about the vaccine’s safety, and the trial expansion into adolescents is important.
“I’m comfortable with the way these trials are going now,” he said. “This is the way I was hoping they would go.”
Ms. Hillenburg is on the parent advisory board of Voices for Vaccines, an organization of parents supporting vaccination that is affiliated with the Task Force for Global Health, an Atlanta-based independent public health organization. Dr. Campbell’s institution has received funds to conduct clinical trials from the National Institutes of Health and several companies, including Merck, GlaxoSmithKline, Sanofi, Pfizer, and Moderna. He has served pro bono on many safety and data monitoring committees. Dr. Frenck’s institution is receiving funds to conduct the Pfizer trial. In the past 5 years, he has also participated in clinical trials for GlaxoSmithKline, Merck, and Meissa vaccines. Dr. Lee and Dr. Meissner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The vaccinations can’t come soon enough for parents like Stacy Hillenburg, a developmental therapist in Aurora, Ill., whose 9-year-old son takes immunosuppressants because he had a heart transplant when he was 7 weeks old. Although school-age children aren’t yet included in clinical trials, if her 12- and 13-year-old daughters could get vaccinated, along with both parents, then the family could relax some of the protocols they currently follow to prevent infection.
Whenever they are around other people, even masked and socially distanced, they come home and immediately shower and change their clothes. So far, no one in the family has been infected with COVID, but the anxiety is ever-present. “I can’t wait for it to come out,” Ms. Hillenburg said of a pediatric COVID vaccine. “It will ease my mind so much.”
She isn’t alone in that anticipation. In the fall, the American Academy of Pediatrics and other pediatric vaccine experts urged faster action on pediatric vaccine trials and worried that children would be left behind as adults gained protection from COVID. But recent developments have eased those concerns.
“Over the next couple of months, we will be doing trials in an age-deescalation manner,” with studies moving gradually to younger children, Anthony S. Fauci, MD, chief medical adviser on COVID-19 to the president, said in a coronavirus response team briefing on Jan. 29. “So that hopefully, as we get to the late spring and summer, we will have children being able to be vaccinated.”
Pfizer completed enrollment of 2,259 teens aged 12-15 years in late January and expects to move forward with a separate pediatric trial of children aged 5-11 years by this spring, Keanna Ghazvini, senior associate for global media relations at Pfizer, said in an interview.
Enrollment in Moderna’s TeenCove study of adolescents ages 12-17 years began slowly in late December, but the pace has since picked up, said company spokesperson Colleen Hussey. “We continue to bring clinical trial sites online, and we are on track to provide updated data around mid-year 2021.” A trial extension in children 11 years and younger is expected to begin later in 2021.
Johnson & Johnson and AstraZeneca said they expect to begin adolescent trials in early 2021, according to data shared by the Advisory Committee on Immunization Practices. An interim analysis of J&J’s Janssen COVID-19 vaccine trial data, released on Jan. 29, showed it was 72% effective in US participants aged 18 years or older. AstraZeneca’s U.S. trial in adults is ongoing.
Easing the burden
Vaccination could lessen children’s risk of severe disease as well as the social and emotional burdens of the pandemic, says James Campbell, MD, a pediatric infectious disease specialist at the University of Maryland’s Center for Vaccine Development in Baltimore, which was involved in the Moderna and early-phase Pfizer trials. He coauthored a September 2020 article in Clinical Infectious Diseases titled: “Warp Speed for COVID-19 vaccines: Why are children stuck in neutral?”
The adolescent trials are a vital step to ensure timely vaccine access for teens and younger children. “It is reasonable, when you have limited vaccine, that your rollout goes to the highest priority and then moves to lower and lower priorities. In adults, we’re just saying: ‘Wait your turn,’ ” he said of the current vaccination effort. “If we didn’t have the [vaccine trial] data in children, we’d be saying: ‘You don’t have a turn.’ ”
As the pandemic has worn on, the burden on children has grown. As of Tuesday, 269 children had died of COVID-19. That is well above the highest annual death toll recorded during a regular flu season – 188 flu deaths among children and adolescents under 18 in the 2019-2020 and 2017-2018 flu seasons.
Children are less likely to transmit COVID-19 in their household than adults, according to a meta-analysis of 54 studies published in JAMA Network Open. But that does not necessarily mean children are less infectious, the authors said, noting that unmeasured factors could have affected the spread of infection among adults.
Moreover, children and adolescents need protection from COVID infection – and from the potential for severe disease or lingering effects – and, given that there are 74 million children and teens in the United States, their vaccination is an important part of stopping the pandemic, said Grace Lee, MD, professor of pediatrics at Stanford (Calif.) University, and cochair of ACIP’s COVID-19 Vaccine Safety Technical Subgroup.
“In order to interrupt transmission, I don’t see how we’re going to do that without vaccinating children and adolescents,” she said.
Dr. Lee said her 16-year-old daughter misses the normal teenage social life and is excited about getting the vaccine when she is eligible. (Adolescents without high-risk conditions are in the lowest vaccination tier, according to ACIP recommendations.) “There is truly individual protection to be gained,” Dr. Lee said.
She noted that researchers continue to assess the immune responses to the adult vaccines – even looking at immune characteristics of the small percentage of people who aren’t protected from infection – and that information helps in the evaluation of the pediatric immune responses. As the trials expand to younger children and infants, dosing will be a major focus. “How many doses do they need they need to receive the same immunity? Safety considerations will be critically important,” she said.
Teen trials underway
Pfizer/BioNTech extended its adult trial to 16- and 17-year-olds in October, which enabled older teens to be included in its emergency-use authorization. They and younger teens, ages 12-15, receive the same dose as adults.
The ongoing trials with Pfizer and Moderna vaccines are immunobridging trials, designed to study safety and immunogenicity. Investigators will compare the teens’ immune response with the findings from the larger adult trials. When the trials expand to school-age children (6-12 years), protocols call for testing the safety and immunogenicity of a half-dose vaccine as well as the full dose.
Children ages 2-5 years and infants and toddlers will be enrolled in future trials, studying safety and immunogenicity of full, half, or even quarter dosages. The Pediatric Research Equity Act of 2003 requires licensed vaccines to be tested for safety and efficacy in children, unless they are not appropriate for a pediatric population.
Demand for the teen trials has been strong. At Cincinnati Children’s Hospital Medical Center, 259 teenagers joined the Pfizer/BioNTech trial, but some teenagers were turned away when the trial’s national enrollment closed in late January.
“Many of the children are having no side effects, and if they are, they’re having the same [effects] as the young adults – local redness or pain, fatigue, and headaches,” said Robert Frenck, MD, director of the Cincinnati Children’s Gamble Program for Clinical Studies.
Parents may share some of the vaccine hesitancy that has affected adult vaccination. But that is balanced by the hope that vaccines will end the pandemic and usher in a new normal. “If it looks like [vaccines] will increase the likelihood of children returning to school safely, that may be a motivating factor,” Dr. Frenck said.
Cody Meissner, MD, chief of the pediatric infectious disease service at Tufts Medical Center, Boston, was initially cautious about the extension of vaccination to adolescents. A member of the Vaccine and Related Biological Products Advisory Committee, which evaluates data and makes recommendations to the Food and Drug Administration, Dr. Meissner initially abstained in the vote on the Pfizer/BioNTech emergency-use authorization for people 16 and older.
He noted that, at the time the committee reviewed the Pfizer vaccine, the company had data available for just 134 teenagers, half of whom received a placebo. But the vaccination of 34 million adults has provided robust data about the vaccine’s safety, and the trial expansion into adolescents is important.
“I’m comfortable with the way these trials are going now,” he said. “This is the way I was hoping they would go.”
Ms. Hillenburg is on the parent advisory board of Voices for Vaccines, an organization of parents supporting vaccination that is affiliated with the Task Force for Global Health, an Atlanta-based independent public health organization. Dr. Campbell’s institution has received funds to conduct clinical trials from the National Institutes of Health and several companies, including Merck, GlaxoSmithKline, Sanofi, Pfizer, and Moderna. He has served pro bono on many safety and data monitoring committees. Dr. Frenck’s institution is receiving funds to conduct the Pfizer trial. In the past 5 years, he has also participated in clinical trials for GlaxoSmithKline, Merck, and Meissa vaccines. Dr. Lee and Dr. Meissner disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 in children: New cases down for third straight week
New COVID-19 cases in children dropped for the third consecutive week, even as children continue to make up a larger share of all cases, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
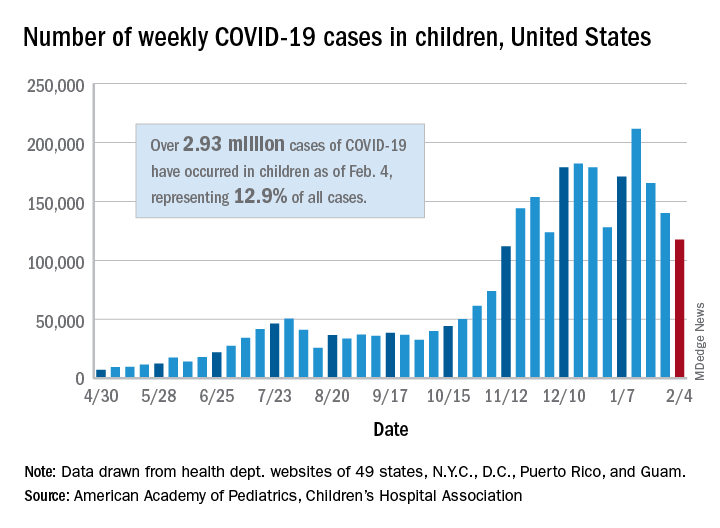
New child cases totaled almost 118,000 for the week of Jan. 29-Feb. 4, continuing the decline that began right after the United States topped 200,000 cases for the only time Jan. 8-14, the AAP and the CHA said in their weekly COVID-19 report.
For the latest week, however, children represented 16.0% of all new COVID-19 cases, continuing a 5-week increase that began in early December 2020, after the proportion had dropped to 12.6%, based on data collected from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. During the week of Sept. 11-17, children made up 16.9% of all cases, the highest level seen during the pandemic.
The 2.93 million cases that have been reported in children make up 12.9% of all cases since the pandemic began, and the overall rate of pediatric coronavirus infection is 3,899 cases per 100,000 children in the population. Taking a step down from the national level, 30 states are above that rate and 18 are below it, along with D.C., New York City, Puerto Rico, and Guam (New York and Texas are excluded), the AAP and CHA reported.
There were 12 new COVID-19–related child deaths in the 43 states, along with New York City and Guam, that are reporting such data, bringing the total to 227. Nationally, 0.06% of all deaths have occurred in children, with rates ranging from 0.00% (11 states) to 0.26% (Nebraska) in the 45 jurisdictions, the AAP/CHA report shows.
Child hospitalizations rose to 1.9% of all hospitalizations after holding at 1.8% since mid-November in 25 reporting jurisdictions (24 states and New York City), but the hospitalization rate among children with COVID held at 0.8%, where it has been for the last 4 weeks. Hospitalization rates as high as 3.8% were recorded early in the pandemic, the AAP and CHA noted.
New COVID-19 cases in children dropped for the third consecutive week, even as children continue to make up a larger share of all cases, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

New child cases totaled almost 118,000 for the week of Jan. 29-Feb. 4, continuing the decline that began right after the United States topped 200,000 cases for the only time Jan. 8-14, the AAP and the CHA said in their weekly COVID-19 report.
For the latest week, however, children represented 16.0% of all new COVID-19 cases, continuing a 5-week increase that began in early December 2020, after the proportion had dropped to 12.6%, based on data collected from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. During the week of Sept. 11-17, children made up 16.9% of all cases, the highest level seen during the pandemic.
The 2.93 million cases that have been reported in children make up 12.9% of all cases since the pandemic began, and the overall rate of pediatric coronavirus infection is 3,899 cases per 100,000 children in the population. Taking a step down from the national level, 30 states are above that rate and 18 are below it, along with D.C., New York City, Puerto Rico, and Guam (New York and Texas are excluded), the AAP and CHA reported.
There were 12 new COVID-19–related child deaths in the 43 states, along with New York City and Guam, that are reporting such data, bringing the total to 227. Nationally, 0.06% of all deaths have occurred in children, with rates ranging from 0.00% (11 states) to 0.26% (Nebraska) in the 45 jurisdictions, the AAP/CHA report shows.
Child hospitalizations rose to 1.9% of all hospitalizations after holding at 1.8% since mid-November in 25 reporting jurisdictions (24 states and New York City), but the hospitalization rate among children with COVID held at 0.8%, where it has been for the last 4 weeks. Hospitalization rates as high as 3.8% were recorded early in the pandemic, the AAP and CHA noted.
New COVID-19 cases in children dropped for the third consecutive week, even as children continue to make up a larger share of all cases, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

New child cases totaled almost 118,000 for the week of Jan. 29-Feb. 4, continuing the decline that began right after the United States topped 200,000 cases for the only time Jan. 8-14, the AAP and the CHA said in their weekly COVID-19 report.
For the latest week, however, children represented 16.0% of all new COVID-19 cases, continuing a 5-week increase that began in early December 2020, after the proportion had dropped to 12.6%, based on data collected from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. During the week of Sept. 11-17, children made up 16.9% of all cases, the highest level seen during the pandemic.
The 2.93 million cases that have been reported in children make up 12.9% of all cases since the pandemic began, and the overall rate of pediatric coronavirus infection is 3,899 cases per 100,000 children in the population. Taking a step down from the national level, 30 states are above that rate and 18 are below it, along with D.C., New York City, Puerto Rico, and Guam (New York and Texas are excluded), the AAP and CHA reported.
There were 12 new COVID-19–related child deaths in the 43 states, along with New York City and Guam, that are reporting such data, bringing the total to 227. Nationally, 0.06% of all deaths have occurred in children, with rates ranging from 0.00% (11 states) to 0.26% (Nebraska) in the 45 jurisdictions, the AAP/CHA report shows.
Child hospitalizations rose to 1.9% of all hospitalizations after holding at 1.8% since mid-November in 25 reporting jurisdictions (24 states and New York City), but the hospitalization rate among children with COVID held at 0.8%, where it has been for the last 4 weeks. Hospitalization rates as high as 3.8% were recorded early in the pandemic, the AAP and CHA noted.
Study tests ways to increase autism screening and referrals
To improve autism screening rates, researchers in Utah tried a range of interventions.
They added automatic reminders to the electronic health record (EHR). They started using a shorter, more sensitive screening instrument. And they trained clinicians to perform autism-specific evaluations in a primary care clinic.
The researchers found that these interventions were associated with increased rates of autism screening and referrals.
At the same time, they looked at screening and referral rates at other community clinics in their health care system. These clinics incorporated EHR reminders but not all of the other changes.
“The community clinics had an increase in screening frequency with only automatic reminders,” the researchers reported. At the two intervention clinics, however, screening rates increased more than they did at the community clinics. Referrals did not significantly increase at the community clinics.
Kathleen Campbell, MD, MHSc, a pediatric resident at the University of Utah in Salt Lake City, and colleagues described their research in a study published in Pediatrics.
Three phases
They examined more than 12,000 well-child visits for children aged 16-30 months between July 2017 and June 2019.
In all, 4,155 visits occurred at the 2 intervention clinics, and 8,078 visits occurred at the 27 community clinics in the University of Utah health care system.
From baseline through the interventions, the proportion of visits with screening increased by 51% in the intervention clinics (from 58.6% to 88.8%), and by 21% in the community clinics (from 43.4% to 52.4%). The proportion of referrals increased 1.5-fold in intervention clinics, from 1.3% to 3.3%, the authors said.
The American Academy of Pediatrics (AAP) supports screening for autism in all children starting at age 18 months, but “only 44% of children with autism have had a comprehensive autism evaluation before age 36 months,” Dr. Campbell and colleagues wrote.
In their system, about half of the children were being screened for autism, and 0.5% had autism diagnosed.
In an effort to increase the proportion of visits with screening for autism and the proportion of visits with referrals for autism evaluation, Dr. Campbell and colleagues designed a quality improvement study.
Following a baseline period, they implemented interventions in three phases.
Initially, all clinics used the Modified Checklist for Autism in Toddlers, Revised (M-CHAT-R) for autism screening. For the first phase starting in July 2018, the researchers changed the screening instrument at the two intervention clinics to the Parent’s Observation of Social Interaction (POSI). This instrument “is embedded in a broadband developmental screen, is shorter than the M-CHAT-R, and includes questions about the consistency of the child’s behavior,” the authors said. “The POSI has greater sensitivity than the M-CHAT-R ... and similar, although somewhat lower, specificity.”
In intervention phase 2 starting in November 2018, the researchers “added an automatic reminder for autism screening to the EHR health maintenance screen.” Both the intervention clinics and the community clinics received the automatic reminders.
In intervention phase 3 starting in February 2019, they “added a referral option that clinicians could use for rapid access to autism-specific evaluation ... for children who had a POSI result suggestive of autism and for whom the clinician had sufficient concerns about autism that would indicate the need for referral for autism evaluation,” the researchers said.
“Using an online tutorial, we trained three clinicians in the intervention clinics to administer an observational assessment known as the Screening Tool for Autism in Toddlers (STAT),” which requires a 30-minute visit, they said. “Children who had a STAT result suggestive of autism were referred for expedited autism diagnostic evaluation, which was performed by a multidisciplinary team in our university-based developmental assessment clinic. Children who had a STAT result that did not suggest autism did not receive further autism evaluations unless the clinician felt they still needed further evaluation at the developmental clinic.”
After the switch to POSI, the percentage of visits with a positive screen result increased from 4.7% to 13.5% in the intervention clinics.
Furthermore, referrals were 3.4 times more frequent for visits during phase 3 in the intervention clinics, relative to the baseline period.
Potential to overwhelm
“The change to a more sensitive screening instrument increased the frequency of screening results suggestive of autism and informed our improvement team of the need to implement autism evaluation in primary care to avoid overwhelming our referral system,” Dr. Campbell and coauthors reported.
Future studies may assess whether increased screening and referrals speed the time to diagnosis and treatment and improve long-term functional abilities of children with autism. Some children in the study have received an autism diagnosis, while others have not yet been evaluated.
The use of STAT in primary care may be limited by “the barriers of training providers and purchasing materials,” the authors noted. “However, the time-based billing for lengthier appointments and billing for developmental testing help to cover cost.”
The intervention clinics and community clinics were staffed by pediatric providers, including residents and attendings, said Dr. Campbell.
“The staffing is similar at the community and intervention clinics, with mostly pediatricians and some nurse practitioners,” Dr. Campbell said. “One difference is that there are a few family medicine physicians in the community clinics, but we did not study whether that made a difference in screening. At the beginning of the study the approach to screening was the same.”
From the start, the community clinics were screening for autism and referring for further autism evaluation less often than the intervention clinics. “I don’t know why they were screening less, but they did improve with the automatic reminders,” said Dr. Campbell. “We didn’t examine type of provider or type of practice in this study, but the literature suggests that family physicians do not screen for autism as often as pediatricians.”
Payment and referral challenges
In theory, the approach in the study is a great idea, but it may not be feasible to implement for many private practices, said Herschel Lessin, MD. Dr. Lessin is a senior partner of the Children’s Medical Group in New York.
“We desperately need autism screening in a primary care setting,” Dr. Lessin said. “These authors found that wasn’t being done as recommended by the AAP Bright Futures, which is a problem.”
However, the researchers incorporated the interventions in a health care system with “far more resources than most people in practice would ever have” and substituted a less familiar screening tool.
In addition, the ability to use confirmatory STAT for primary care evaluations may be limited. “Unless you can find pediatricians willing to commit 30 to 45 minutes on one of these evaluations ... few are going to do that,” he said.
“The whole problem is that there are no referrals available or very few referrals available, and that insurance payments so underpay for developmental screening and evaluation that it does not justify the time doing it, so a lot of doctors are unable to do it,” said Dr. Lessin. When a referral is warranted, developmental pediatricians may have 6- to 12-month waiting lists, he said.
“For people in clinical practice, this is not news,” Dr. Lessin said. “We know we should screen for autism. The problem is it’s time consuming. Nobody pays for it. We have no place to send them even when we are suspicious.”
From screening to diagnosis to treatment
“Autism screen approaches vary but with educational efforts on the part of the AAP, CDC, and family organizations the rates for autism screening have dramatically improved,” said Susan L. Hyman, MD, professor of pediatrics at the University of Rochester in New York. “I do not know if screening rates have been impacted by COVID.”
Dr. Hyman and coauthors wrote an AAP clinical report on the identification, evaluation, and management of children with autism spectrum disorder. The report was published in the January 2020 issue of Pediatrics.
After screening and diagnostic testing, patients most importantly need to be able to access “timely and equitable evidence-based intervention,” which should be available, said Dr. Hyman.
Although researchers have proposed training primary care providers in autism diagnostics, “older, more complex patients with co-occurring behavioral health or other developmental disorders may need more specialized diagnostic assessment than could be accomplished in a primary care setting,” Dr. Hyman added.
“However, it is very important to identify children with therapeutic needs as early as possible and move them through the continuum from screening to diagnosis to treatment in a timely fashion. It would be wonderful if symptoms could be addressed without the need for diagnosis in the very youngest children,” Dr. Hyman said. “Early symptoms, even if not autism, are likely to be appropriate for intervention – whether it is speech therapy, attention to food selectivity, sleep problems – things that impact quality of life and potential future symptoms.”
The research was supported by the Utah Stimulating Access to Research in Residency Transition Scholar award, which is funded by the National Institutes of Health.
Dr. Campbell is an inventor on a patent related to screening for autism. The study authors otherwise had no disclosures. Dr. Lessin is on the editorial advisory board for Pediatric News and is on an advisory board for Cognoa, which is developing a medical device to diagnose autism and he is also the co-editor of the AAP's current ADHD Toolkit. Dr. Hyman had no relevant financial disclosures.
*This story was updated on Feb. 11, 2021.
To improve autism screening rates, researchers in Utah tried a range of interventions.
They added automatic reminders to the electronic health record (EHR). They started using a shorter, more sensitive screening instrument. And they trained clinicians to perform autism-specific evaluations in a primary care clinic.
The researchers found that these interventions were associated with increased rates of autism screening and referrals.
At the same time, they looked at screening and referral rates at other community clinics in their health care system. These clinics incorporated EHR reminders but not all of the other changes.
“The community clinics had an increase in screening frequency with only automatic reminders,” the researchers reported. At the two intervention clinics, however, screening rates increased more than they did at the community clinics. Referrals did not significantly increase at the community clinics.
Kathleen Campbell, MD, MHSc, a pediatric resident at the University of Utah in Salt Lake City, and colleagues described their research in a study published in Pediatrics.
Three phases
They examined more than 12,000 well-child visits for children aged 16-30 months between July 2017 and June 2019.
In all, 4,155 visits occurred at the 2 intervention clinics, and 8,078 visits occurred at the 27 community clinics in the University of Utah health care system.
From baseline through the interventions, the proportion of visits with screening increased by 51% in the intervention clinics (from 58.6% to 88.8%), and by 21% in the community clinics (from 43.4% to 52.4%). The proportion of referrals increased 1.5-fold in intervention clinics, from 1.3% to 3.3%, the authors said.
The American Academy of Pediatrics (AAP) supports screening for autism in all children starting at age 18 months, but “only 44% of children with autism have had a comprehensive autism evaluation before age 36 months,” Dr. Campbell and colleagues wrote.
In their system, about half of the children were being screened for autism, and 0.5% had autism diagnosed.
In an effort to increase the proportion of visits with screening for autism and the proportion of visits with referrals for autism evaluation, Dr. Campbell and colleagues designed a quality improvement study.
Following a baseline period, they implemented interventions in three phases.
Initially, all clinics used the Modified Checklist for Autism in Toddlers, Revised (M-CHAT-R) for autism screening. For the first phase starting in July 2018, the researchers changed the screening instrument at the two intervention clinics to the Parent’s Observation of Social Interaction (POSI). This instrument “is embedded in a broadband developmental screen, is shorter than the M-CHAT-R, and includes questions about the consistency of the child’s behavior,” the authors said. “The POSI has greater sensitivity than the M-CHAT-R ... and similar, although somewhat lower, specificity.”
In intervention phase 2 starting in November 2018, the researchers “added an automatic reminder for autism screening to the EHR health maintenance screen.” Both the intervention clinics and the community clinics received the automatic reminders.
In intervention phase 3 starting in February 2019, they “added a referral option that clinicians could use for rapid access to autism-specific evaluation ... for children who had a POSI result suggestive of autism and for whom the clinician had sufficient concerns about autism that would indicate the need for referral for autism evaluation,” the researchers said.
“Using an online tutorial, we trained three clinicians in the intervention clinics to administer an observational assessment known as the Screening Tool for Autism in Toddlers (STAT),” which requires a 30-minute visit, they said. “Children who had a STAT result suggestive of autism were referred for expedited autism diagnostic evaluation, which was performed by a multidisciplinary team in our university-based developmental assessment clinic. Children who had a STAT result that did not suggest autism did not receive further autism evaluations unless the clinician felt they still needed further evaluation at the developmental clinic.”
After the switch to POSI, the percentage of visits with a positive screen result increased from 4.7% to 13.5% in the intervention clinics.
Furthermore, referrals were 3.4 times more frequent for visits during phase 3 in the intervention clinics, relative to the baseline period.
Potential to overwhelm
“The change to a more sensitive screening instrument increased the frequency of screening results suggestive of autism and informed our improvement team of the need to implement autism evaluation in primary care to avoid overwhelming our referral system,” Dr. Campbell and coauthors reported.
Future studies may assess whether increased screening and referrals speed the time to diagnosis and treatment and improve long-term functional abilities of children with autism. Some children in the study have received an autism diagnosis, while others have not yet been evaluated.
The use of STAT in primary care may be limited by “the barriers of training providers and purchasing materials,” the authors noted. “However, the time-based billing for lengthier appointments and billing for developmental testing help to cover cost.”
The intervention clinics and community clinics were staffed by pediatric providers, including residents and attendings, said Dr. Campbell.
“The staffing is similar at the community and intervention clinics, with mostly pediatricians and some nurse practitioners,” Dr. Campbell said. “One difference is that there are a few family medicine physicians in the community clinics, but we did not study whether that made a difference in screening. At the beginning of the study the approach to screening was the same.”
From the start, the community clinics were screening for autism and referring for further autism evaluation less often than the intervention clinics. “I don’t know why they were screening less, but they did improve with the automatic reminders,” said Dr. Campbell. “We didn’t examine type of provider or type of practice in this study, but the literature suggests that family physicians do not screen for autism as often as pediatricians.”
Payment and referral challenges
In theory, the approach in the study is a great idea, but it may not be feasible to implement for many private practices, said Herschel Lessin, MD. Dr. Lessin is a senior partner of the Children’s Medical Group in New York.
“We desperately need autism screening in a primary care setting,” Dr. Lessin said. “These authors found that wasn’t being done as recommended by the AAP Bright Futures, which is a problem.”
However, the researchers incorporated the interventions in a health care system with “far more resources than most people in practice would ever have” and substituted a less familiar screening tool.
In addition, the ability to use confirmatory STAT for primary care evaluations may be limited. “Unless you can find pediatricians willing to commit 30 to 45 minutes on one of these evaluations ... few are going to do that,” he said.
“The whole problem is that there are no referrals available or very few referrals available, and that insurance payments so underpay for developmental screening and evaluation that it does not justify the time doing it, so a lot of doctors are unable to do it,” said Dr. Lessin. When a referral is warranted, developmental pediatricians may have 6- to 12-month waiting lists, he said.
“For people in clinical practice, this is not news,” Dr. Lessin said. “We know we should screen for autism. The problem is it’s time consuming. Nobody pays for it. We have no place to send them even when we are suspicious.”
From screening to diagnosis to treatment
“Autism screen approaches vary but with educational efforts on the part of the AAP, CDC, and family organizations the rates for autism screening have dramatically improved,” said Susan L. Hyman, MD, professor of pediatrics at the University of Rochester in New York. “I do not know if screening rates have been impacted by COVID.”
Dr. Hyman and coauthors wrote an AAP clinical report on the identification, evaluation, and management of children with autism spectrum disorder. The report was published in the January 2020 issue of Pediatrics.
After screening and diagnostic testing, patients most importantly need to be able to access “timely and equitable evidence-based intervention,” which should be available, said Dr. Hyman.
Although researchers have proposed training primary care providers in autism diagnostics, “older, more complex patients with co-occurring behavioral health or other developmental disorders may need more specialized diagnostic assessment than could be accomplished in a primary care setting,” Dr. Hyman added.
“However, it is very important to identify children with therapeutic needs as early as possible and move them through the continuum from screening to diagnosis to treatment in a timely fashion. It would be wonderful if symptoms could be addressed without the need for diagnosis in the very youngest children,” Dr. Hyman said. “Early symptoms, even if not autism, are likely to be appropriate for intervention – whether it is speech therapy, attention to food selectivity, sleep problems – things that impact quality of life and potential future symptoms.”
The research was supported by the Utah Stimulating Access to Research in Residency Transition Scholar award, which is funded by the National Institutes of Health.
Dr. Campbell is an inventor on a patent related to screening for autism. The study authors otherwise had no disclosures. Dr. Lessin is on the editorial advisory board for Pediatric News and is on an advisory board for Cognoa, which is developing a medical device to diagnose autism and he is also the co-editor of the AAP's current ADHD Toolkit. Dr. Hyman had no relevant financial disclosures.
*This story was updated on Feb. 11, 2021.
To improve autism screening rates, researchers in Utah tried a range of interventions.
They added automatic reminders to the electronic health record (EHR). They started using a shorter, more sensitive screening instrument. And they trained clinicians to perform autism-specific evaluations in a primary care clinic.
The researchers found that these interventions were associated with increased rates of autism screening and referrals.
At the same time, they looked at screening and referral rates at other community clinics in their health care system. These clinics incorporated EHR reminders but not all of the other changes.
“The community clinics had an increase in screening frequency with only automatic reminders,” the researchers reported. At the two intervention clinics, however, screening rates increased more than they did at the community clinics. Referrals did not significantly increase at the community clinics.
Kathleen Campbell, MD, MHSc, a pediatric resident at the University of Utah in Salt Lake City, and colleagues described their research in a study published in Pediatrics.
Three phases
They examined more than 12,000 well-child visits for children aged 16-30 months between July 2017 and June 2019.
In all, 4,155 visits occurred at the 2 intervention clinics, and 8,078 visits occurred at the 27 community clinics in the University of Utah health care system.
From baseline through the interventions, the proportion of visits with screening increased by 51% in the intervention clinics (from 58.6% to 88.8%), and by 21% in the community clinics (from 43.4% to 52.4%). The proportion of referrals increased 1.5-fold in intervention clinics, from 1.3% to 3.3%, the authors said.
The American Academy of Pediatrics (AAP) supports screening for autism in all children starting at age 18 months, but “only 44% of children with autism have had a comprehensive autism evaluation before age 36 months,” Dr. Campbell and colleagues wrote.
In their system, about half of the children were being screened for autism, and 0.5% had autism diagnosed.
In an effort to increase the proportion of visits with screening for autism and the proportion of visits with referrals for autism evaluation, Dr. Campbell and colleagues designed a quality improvement study.
Following a baseline period, they implemented interventions in three phases.
Initially, all clinics used the Modified Checklist for Autism in Toddlers, Revised (M-CHAT-R) for autism screening. For the first phase starting in July 2018, the researchers changed the screening instrument at the two intervention clinics to the Parent’s Observation of Social Interaction (POSI). This instrument “is embedded in a broadband developmental screen, is shorter than the M-CHAT-R, and includes questions about the consistency of the child’s behavior,” the authors said. “The POSI has greater sensitivity than the M-CHAT-R ... and similar, although somewhat lower, specificity.”
In intervention phase 2 starting in November 2018, the researchers “added an automatic reminder for autism screening to the EHR health maintenance screen.” Both the intervention clinics and the community clinics received the automatic reminders.
In intervention phase 3 starting in February 2019, they “added a referral option that clinicians could use for rapid access to autism-specific evaluation ... for children who had a POSI result suggestive of autism and for whom the clinician had sufficient concerns about autism that would indicate the need for referral for autism evaluation,” the researchers said.
“Using an online tutorial, we trained three clinicians in the intervention clinics to administer an observational assessment known as the Screening Tool for Autism in Toddlers (STAT),” which requires a 30-minute visit, they said. “Children who had a STAT result suggestive of autism were referred for expedited autism diagnostic evaluation, which was performed by a multidisciplinary team in our university-based developmental assessment clinic. Children who had a STAT result that did not suggest autism did not receive further autism evaluations unless the clinician felt they still needed further evaluation at the developmental clinic.”
After the switch to POSI, the percentage of visits with a positive screen result increased from 4.7% to 13.5% in the intervention clinics.
Furthermore, referrals were 3.4 times more frequent for visits during phase 3 in the intervention clinics, relative to the baseline period.
Potential to overwhelm
“The change to a more sensitive screening instrument increased the frequency of screening results suggestive of autism and informed our improvement team of the need to implement autism evaluation in primary care to avoid overwhelming our referral system,” Dr. Campbell and coauthors reported.
Future studies may assess whether increased screening and referrals speed the time to diagnosis and treatment and improve long-term functional abilities of children with autism. Some children in the study have received an autism diagnosis, while others have not yet been evaluated.
The use of STAT in primary care may be limited by “the barriers of training providers and purchasing materials,” the authors noted. “However, the time-based billing for lengthier appointments and billing for developmental testing help to cover cost.”
The intervention clinics and community clinics were staffed by pediatric providers, including residents and attendings, said Dr. Campbell.
“The staffing is similar at the community and intervention clinics, with mostly pediatricians and some nurse practitioners,” Dr. Campbell said. “One difference is that there are a few family medicine physicians in the community clinics, but we did not study whether that made a difference in screening. At the beginning of the study the approach to screening was the same.”
From the start, the community clinics were screening for autism and referring for further autism evaluation less often than the intervention clinics. “I don’t know why they were screening less, but they did improve with the automatic reminders,” said Dr. Campbell. “We didn’t examine type of provider or type of practice in this study, but the literature suggests that family physicians do not screen for autism as often as pediatricians.”
Payment and referral challenges
In theory, the approach in the study is a great idea, but it may not be feasible to implement for many private practices, said Herschel Lessin, MD. Dr. Lessin is a senior partner of the Children’s Medical Group in New York.
“We desperately need autism screening in a primary care setting,” Dr. Lessin said. “These authors found that wasn’t being done as recommended by the AAP Bright Futures, which is a problem.”
However, the researchers incorporated the interventions in a health care system with “far more resources than most people in practice would ever have” and substituted a less familiar screening tool.
In addition, the ability to use confirmatory STAT for primary care evaluations may be limited. “Unless you can find pediatricians willing to commit 30 to 45 minutes on one of these evaluations ... few are going to do that,” he said.
“The whole problem is that there are no referrals available or very few referrals available, and that insurance payments so underpay for developmental screening and evaluation that it does not justify the time doing it, so a lot of doctors are unable to do it,” said Dr. Lessin. When a referral is warranted, developmental pediatricians may have 6- to 12-month waiting lists, he said.
“For people in clinical practice, this is not news,” Dr. Lessin said. “We know we should screen for autism. The problem is it’s time consuming. Nobody pays for it. We have no place to send them even when we are suspicious.”
From screening to diagnosis to treatment
“Autism screen approaches vary but with educational efforts on the part of the AAP, CDC, and family organizations the rates for autism screening have dramatically improved,” said Susan L. Hyman, MD, professor of pediatrics at the University of Rochester in New York. “I do not know if screening rates have been impacted by COVID.”
Dr. Hyman and coauthors wrote an AAP clinical report on the identification, evaluation, and management of children with autism spectrum disorder. The report was published in the January 2020 issue of Pediatrics.
After screening and diagnostic testing, patients most importantly need to be able to access “timely and equitable evidence-based intervention,” which should be available, said Dr. Hyman.
Although researchers have proposed training primary care providers in autism diagnostics, “older, more complex patients with co-occurring behavioral health or other developmental disorders may need more specialized diagnostic assessment than could be accomplished in a primary care setting,” Dr. Hyman added.
“However, it is very important to identify children with therapeutic needs as early as possible and move them through the continuum from screening to diagnosis to treatment in a timely fashion. It would be wonderful if symptoms could be addressed without the need for diagnosis in the very youngest children,” Dr. Hyman said. “Early symptoms, even if not autism, are likely to be appropriate for intervention – whether it is speech therapy, attention to food selectivity, sleep problems – things that impact quality of life and potential future symptoms.”
The research was supported by the Utah Stimulating Access to Research in Residency Transition Scholar award, which is funded by the National Institutes of Health.
Dr. Campbell is an inventor on a patent related to screening for autism. The study authors otherwise had no disclosures. Dr. Lessin is on the editorial advisory board for Pediatric News and is on an advisory board for Cognoa, which is developing a medical device to diagnose autism and he is also the co-editor of the AAP's current ADHD Toolkit. Dr. Hyman had no relevant financial disclosures.
*This story was updated on Feb. 11, 2021.
FROM PEDIATRICS
Are diagnosticians chasing COVID-linked zebras and missing horses?
The emergence of multiple inflammatory syndrome in children (MIS-C) in association with COVID-19 may be complicating the investigation and diagnosis of more common viral and bacterial infections, potentially delaying treatment and prolonging hospital stays.
Two recent articles published online in Hospital Pediatrics provide evidence of this phenomenon. The articles outlined case studies of children who underwent extensive investigation for MIS-C when in fact they had less severe and more common infections. MIS-C is a severe but rare syndrome that involves systemic hyperinflammation with fever and multisystem organ dysfunction similar to that of Kawasaki disease (KD).
In one of the articles, Matthew Molloy, MD, MPH, of the division of pediatric hospital medicine at Cincinnati Children’s Hospital Medical Center, and colleagues aptly asked: “What are we missing in our search for MIS-C?”
E. coli, not SARS-CoV-2
That question arose from a case involving a 3-year-old boy who had a 6-day history of fever and fatigue. Three days earlier, he had tested negative for strep antigen and COVID-19. He had a persistent, high fever, reduced appetite, and reduced urine output and was taken to the ED. On physical examination, there was no rash, skin peeling, redness of the eye or oral mucosa, congestion, rhinorrhea, cough, shortness of breath, chest pain, abdominal pain, nausea, vomiting, or diarrhea.
Urinalysis results and exam findings were suspicious for pyelonephritis. Other findings from an extensive laboratory workup raised the alarm that the boy was suffering from MIS-C as opposed to incomplete KD. After admission to hospital medicine, the cardiology, rheumatology, and infectious disease teams were called in to consult.
Repeat labs were planned for the following day before initiating therapy. On day 2, the child’s urine culture was positive for gram-negative rods, later identified as Escherichia coli. The boy was started on ceftriaxone. Left renal scarring was apparent on ultrasound. The patient’s condition resolved after 36 hours, and he was discharged home with antibiotics.
‘Diagnosis derailed’
Calling this a case of “diagnosis derailed,” the authors noted that, in the pre-COVID era, this child’s signs and symptoms would likely have triggered a more targeted and less costly evaluation for more common infectious and noninfectious causes, including pyelonephritis, absent any physical exam findings consistent with KD.
“However, the patient presented in the midst of the COVID-19 pandemic with growing awareness of a new clinical entity,” Dr. Molloy and colleagues wrote. “Anchored to the patient’s persistent fever, the medical team initiated an extensive, costly, and ultimately unnecessary workup to avoid missing the diagnosis of MIS-C; a not yet well-described diagnosis with potentially severe morbidity.”
Confirmation bias and diagnostic momentum likely contributed to the early focus on MIS-C rather than more common alternatives, the authors acknowledged. The addition of mildly abnormal laboratory data not typically obtained in the evaluation of fever led the team astray. “The diagnosis and definitive treatment may have been made earlier had the focus on concern for MIS-C not been present,” Dr. Molloy said in an interview.
Keeping value in care
The authors recognized that their initial approach to evaluating for MIS-C provided low-value care. “In our desire to not ‘miss’ MIS-C, we were performing costly evaluations that at times produced mildly abnormal, nonspecific results,” they wrote. That triggered a cascade of specialty consultations, follow-up testing, and an unwarranted diagnostic preoccupation with MIS-C.
Determining the extra price tag for the child’s workup would be complex and difficult because there is a difference in the cost to the hospital and the cost to the family, Dr. Molloy said. “However, there are potential cost savings that would be related to making a correct diagnosis in a timely manner in terms of preventing downstream effects from delayed diagnoses.”
Even as clinicians struggle with the challenging SARS-CoV-2 learning curve, Dr. Molloy and associates urged them to continue to strive for high-value care, with an unwavering focus on using only necessary resources, a stewardship the pandemic has shown to be critical.
“The COVID-19 pandemic has been an incredibly stressful time for physicians and for families,” Dr. Molloy said. “COVID-19 and related conditions like MIS-C are new, and we are learning more and more about them every week. These diagnoses are understandably on the minds of physicians and families when children present with fever.” Notwithstanding, the boy’s case underscores the need for clinicians to consider alternate diagnoses and the value of the care provided.
Impact of bias
Dr. Molloy’s group brings home the cognitive biases practitioners often suffer from, including anchoring and confirmation bias and diagnostic momentum, according to J. Howard Smart, MD, chief of pediatrics at Sharp Mary Birch Hospital for Women and Newborns, San Diego, and an assistant clinical professor of pediatrics at University of California, San Diego.
“But it is one thing to recognize these in retrospect and quite another to consider whether they may be happening to you yourself in real time,” he said in an interview. “It is almost as if we need to have a ‘time out,’ where we stop and ask ourselves whether there is something else that could be explaining our patient’s presentation, something that would be more common and more likely to be occurring.”
According to Dr. Smart, who was not involved in Dr. Molloy’s study, the team’s premature diagnostic focus on MIS-C was almost the inverse of what typically happens with KD. “It is usually the case that Kawasaki disease does not enter the differential diagnosis until late in the course of the fever, typically on day 5 or later, when it may have been better to think of it earlier,” he said.
In the second article, Andrea Dean, MD, of the department of pediatrics at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, and colleagues outlined the cases of five patients aged 8-17 years who were hospitalized in May 2020 for suspected MIS-C. They exhibited inflammatory and other concerning indicators but were eventually discharged with a diagnosis of murine typhus.
This flea-borne infection, most commonly reported in the United States in the southeastern Gulf Coast region, Hawaii, and California, is often associated with a triad of fever, rash, and headache.
Cases have been rising in southern Texas, and Dr. Dean and colleagues postulated that school closures and social distancing may have increased exposure as a result of children spending more time outdoors or with pets. “Alternatively, parental concern for SARS-CoV-2 infection could mean children with symptoms are presenting to care and being referred or admitted to the hospital more frequently due to provider concern for MIS-C,” they wrote.
Cardiac involvement
The most concerning of the five cases in terms of possible MIS-C, Dr. Dean said in an interview, was that of a 12-year-old boy who had fever for 6 days in association with headache, eczematous rash, dry lips, and conjunctivitis. Laboratory tests showed a mildly elevated C-reactive protein level, hyponatremia, and thrombocytopenia, as well as sterile pyuria and mildly elevated prothrombin time. He was treated empirically with doxycycline, and his fever resolved over the next 24 hours.
An echocardiogram at initial evaluation, however, revealed mild dilation of the left anterior descending and right coronary arteries, which led to the administration of intravenous immunoglobulin and aspirin for atypical KD, in contrast to MIS-C. The authors postulated that mild cardiac involvement in disorders other than MIS-C and KD may be underrecognized.
The lesson from these cases, Dr. Dean and associates concluded, is that hospitalists must maintain a wide differential diagnosis when assessing a child with prolonged fever and evidence of systemic inflammation. The CDC stipulates that a diagnosis of MIS-C requires the absence of a plausible alternative diagnosis.
In addition to common viral, bacterial, and noninfectious disorders, a range of regional endemic rickettsial and parasitic infections must be considered as alternative diagnoses to MIS-C. “Many of these diseases cannot be reliably differentiated from MIS-C on presentation, and as community exposure to SARS-CoV-2 grows, hospitalists should be prepared to admit febrile children with evidence of systemic inflammation for brief observation periods to evaluate for MIS-C,” Dr. Dean’s group wrote. In this context, however, empiric treatment for common or even uncommon infectious diseases may avoid overdiagnosis and overtreatment of MIS-C as well as improve patient outcomes.
“We do have specific MIS-C guidelines at our institution,” Dr. Dean said, “but like all institutions, we are dealing with the broad definition of MIS-C according to the World Health Organization and the CDC, which is really the takeaway from this paper.”
More difficult differentiation
Both groups of authors pointed out that, as SARS-CoV-2 spreads throughout a community, a higher percentage of the population will have positive results on antibody testing, and such results will become less useful for differentiating between MIS-C and other conditions.
Despite these series’ cautionary lessons, other experts point to the critical importance of including MIS-C early on in the interest of efficient diagnosis and therapy. “In the cases cited, other pathologies were evaluated for and treated accordingly,” said Kara Gross Margolis, MD, AGAF, an associate professor of pediatrics in the division of pediatric gastroenterology, hepatology, and nutrition at Morgan Stanley Children’s Hospital,New York. “These papers stress the need for a balance that is important, and all potential diagnoses need to be considered, but MIS-C, due to its potential severe consequences, also needs to be on our differential now.”
In her view, as this new high-morbidity entity becomes more widespread during the pandemic, it will be increasingly important to keep this condition on the diagnostic radar.
Interestingly, in a converse example of diagnostic clouding, Dr. Gross Margolis’s group reported (Gastroenterology. 2020 Oct;159[4]:1571-4.e2) last year on a pediatric case series in which the presence of gastrointestinal symptoms in children with COVID-19–related MIS-C muddied the diagnosis by confusing this potentially severe syndrome with more common and less toxic gastrointestinal infections.
According to Dr. Smart, although the two reports don’t offer evidence for a particular diagnostic practice, they can inform the decision-making process. “It may be that we will have enough evidence shortly to say what the best practice is regarding diagnostic evaluation of possible MIS-C cases,” he said. “Until then, we must remember that common things occur commonly, even during a global pandemic.”
Neither of the two reports received any specific funding. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The emergence of multiple inflammatory syndrome in children (MIS-C) in association with COVID-19 may be complicating the investigation and diagnosis of more common viral and bacterial infections, potentially delaying treatment and prolonging hospital stays.
Two recent articles published online in Hospital Pediatrics provide evidence of this phenomenon. The articles outlined case studies of children who underwent extensive investigation for MIS-C when in fact they had less severe and more common infections. MIS-C is a severe but rare syndrome that involves systemic hyperinflammation with fever and multisystem organ dysfunction similar to that of Kawasaki disease (KD).
In one of the articles, Matthew Molloy, MD, MPH, of the division of pediatric hospital medicine at Cincinnati Children’s Hospital Medical Center, and colleagues aptly asked: “What are we missing in our search for MIS-C?”
E. coli, not SARS-CoV-2
That question arose from a case involving a 3-year-old boy who had a 6-day history of fever and fatigue. Three days earlier, he had tested negative for strep antigen and COVID-19. He had a persistent, high fever, reduced appetite, and reduced urine output and was taken to the ED. On physical examination, there was no rash, skin peeling, redness of the eye or oral mucosa, congestion, rhinorrhea, cough, shortness of breath, chest pain, abdominal pain, nausea, vomiting, or diarrhea.
Urinalysis results and exam findings were suspicious for pyelonephritis. Other findings from an extensive laboratory workup raised the alarm that the boy was suffering from MIS-C as opposed to incomplete KD. After admission to hospital medicine, the cardiology, rheumatology, and infectious disease teams were called in to consult.
Repeat labs were planned for the following day before initiating therapy. On day 2, the child’s urine culture was positive for gram-negative rods, later identified as Escherichia coli. The boy was started on ceftriaxone. Left renal scarring was apparent on ultrasound. The patient’s condition resolved after 36 hours, and he was discharged home with antibiotics.
‘Diagnosis derailed’
Calling this a case of “diagnosis derailed,” the authors noted that, in the pre-COVID era, this child’s signs and symptoms would likely have triggered a more targeted and less costly evaluation for more common infectious and noninfectious causes, including pyelonephritis, absent any physical exam findings consistent with KD.
“However, the patient presented in the midst of the COVID-19 pandemic with growing awareness of a new clinical entity,” Dr. Molloy and colleagues wrote. “Anchored to the patient’s persistent fever, the medical team initiated an extensive, costly, and ultimately unnecessary workup to avoid missing the diagnosis of MIS-C; a not yet well-described diagnosis with potentially severe morbidity.”
Confirmation bias and diagnostic momentum likely contributed to the early focus on MIS-C rather than more common alternatives, the authors acknowledged. The addition of mildly abnormal laboratory data not typically obtained in the evaluation of fever led the team astray. “The diagnosis and definitive treatment may have been made earlier had the focus on concern for MIS-C not been present,” Dr. Molloy said in an interview.
Keeping value in care
The authors recognized that their initial approach to evaluating for MIS-C provided low-value care. “In our desire to not ‘miss’ MIS-C, we were performing costly evaluations that at times produced mildly abnormal, nonspecific results,” they wrote. That triggered a cascade of specialty consultations, follow-up testing, and an unwarranted diagnostic preoccupation with MIS-C.
Determining the extra price tag for the child’s workup would be complex and difficult because there is a difference in the cost to the hospital and the cost to the family, Dr. Molloy said. “However, there are potential cost savings that would be related to making a correct diagnosis in a timely manner in terms of preventing downstream effects from delayed diagnoses.”
Even as clinicians struggle with the challenging SARS-CoV-2 learning curve, Dr. Molloy and associates urged them to continue to strive for high-value care, with an unwavering focus on using only necessary resources, a stewardship the pandemic has shown to be critical.
“The COVID-19 pandemic has been an incredibly stressful time for physicians and for families,” Dr. Molloy said. “COVID-19 and related conditions like MIS-C are new, and we are learning more and more about them every week. These diagnoses are understandably on the minds of physicians and families when children present with fever.” Notwithstanding, the boy’s case underscores the need for clinicians to consider alternate diagnoses and the value of the care provided.
Impact of bias
Dr. Molloy’s group brings home the cognitive biases practitioners often suffer from, including anchoring and confirmation bias and diagnostic momentum, according to J. Howard Smart, MD, chief of pediatrics at Sharp Mary Birch Hospital for Women and Newborns, San Diego, and an assistant clinical professor of pediatrics at University of California, San Diego.
“But it is one thing to recognize these in retrospect and quite another to consider whether they may be happening to you yourself in real time,” he said in an interview. “It is almost as if we need to have a ‘time out,’ where we stop and ask ourselves whether there is something else that could be explaining our patient’s presentation, something that would be more common and more likely to be occurring.”
According to Dr. Smart, who was not involved in Dr. Molloy’s study, the team’s premature diagnostic focus on MIS-C was almost the inverse of what typically happens with KD. “It is usually the case that Kawasaki disease does not enter the differential diagnosis until late in the course of the fever, typically on day 5 or later, when it may have been better to think of it earlier,” he said.
In the second article, Andrea Dean, MD, of the department of pediatrics at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, and colleagues outlined the cases of five patients aged 8-17 years who were hospitalized in May 2020 for suspected MIS-C. They exhibited inflammatory and other concerning indicators but were eventually discharged with a diagnosis of murine typhus.
This flea-borne infection, most commonly reported in the United States in the southeastern Gulf Coast region, Hawaii, and California, is often associated with a triad of fever, rash, and headache.
Cases have been rising in southern Texas, and Dr. Dean and colleagues postulated that school closures and social distancing may have increased exposure as a result of children spending more time outdoors or with pets. “Alternatively, parental concern for SARS-CoV-2 infection could mean children with symptoms are presenting to care and being referred or admitted to the hospital more frequently due to provider concern for MIS-C,” they wrote.
Cardiac involvement
The most concerning of the five cases in terms of possible MIS-C, Dr. Dean said in an interview, was that of a 12-year-old boy who had fever for 6 days in association with headache, eczematous rash, dry lips, and conjunctivitis. Laboratory tests showed a mildly elevated C-reactive protein level, hyponatremia, and thrombocytopenia, as well as sterile pyuria and mildly elevated prothrombin time. He was treated empirically with doxycycline, and his fever resolved over the next 24 hours.
An echocardiogram at initial evaluation, however, revealed mild dilation of the left anterior descending and right coronary arteries, which led to the administration of intravenous immunoglobulin and aspirin for atypical KD, in contrast to MIS-C. The authors postulated that mild cardiac involvement in disorders other than MIS-C and KD may be underrecognized.
The lesson from these cases, Dr. Dean and associates concluded, is that hospitalists must maintain a wide differential diagnosis when assessing a child with prolonged fever and evidence of systemic inflammation. The CDC stipulates that a diagnosis of MIS-C requires the absence of a plausible alternative diagnosis.
In addition to common viral, bacterial, and noninfectious disorders, a range of regional endemic rickettsial and parasitic infections must be considered as alternative diagnoses to MIS-C. “Many of these diseases cannot be reliably differentiated from MIS-C on presentation, and as community exposure to SARS-CoV-2 grows, hospitalists should be prepared to admit febrile children with evidence of systemic inflammation for brief observation periods to evaluate for MIS-C,” Dr. Dean’s group wrote. In this context, however, empiric treatment for common or even uncommon infectious diseases may avoid overdiagnosis and overtreatment of MIS-C as well as improve patient outcomes.
“We do have specific MIS-C guidelines at our institution,” Dr. Dean said, “but like all institutions, we are dealing with the broad definition of MIS-C according to the World Health Organization and the CDC, which is really the takeaway from this paper.”
More difficult differentiation
Both groups of authors pointed out that, as SARS-CoV-2 spreads throughout a community, a higher percentage of the population will have positive results on antibody testing, and such results will become less useful for differentiating between MIS-C and other conditions.
Despite these series’ cautionary lessons, other experts point to the critical importance of including MIS-C early on in the interest of efficient diagnosis and therapy. “In the cases cited, other pathologies were evaluated for and treated accordingly,” said Kara Gross Margolis, MD, AGAF, an associate professor of pediatrics in the division of pediatric gastroenterology, hepatology, and nutrition at Morgan Stanley Children’s Hospital,New York. “These papers stress the need for a balance that is important, and all potential diagnoses need to be considered, but MIS-C, due to its potential severe consequences, also needs to be on our differential now.”
In her view, as this new high-morbidity entity becomes more widespread during the pandemic, it will be increasingly important to keep this condition on the diagnostic radar.
Interestingly, in a converse example of diagnostic clouding, Dr. Gross Margolis’s group reported (Gastroenterology. 2020 Oct;159[4]:1571-4.e2) last year on a pediatric case series in which the presence of gastrointestinal symptoms in children with COVID-19–related MIS-C muddied the diagnosis by confusing this potentially severe syndrome with more common and less toxic gastrointestinal infections.
According to Dr. Smart, although the two reports don’t offer evidence for a particular diagnostic practice, they can inform the decision-making process. “It may be that we will have enough evidence shortly to say what the best practice is regarding diagnostic evaluation of possible MIS-C cases,” he said. “Until then, we must remember that common things occur commonly, even during a global pandemic.”
Neither of the two reports received any specific funding. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The emergence of multiple inflammatory syndrome in children (MIS-C) in association with COVID-19 may be complicating the investigation and diagnosis of more common viral and bacterial infections, potentially delaying treatment and prolonging hospital stays.
Two recent articles published online in Hospital Pediatrics provide evidence of this phenomenon. The articles outlined case studies of children who underwent extensive investigation for MIS-C when in fact they had less severe and more common infections. MIS-C is a severe but rare syndrome that involves systemic hyperinflammation with fever and multisystem organ dysfunction similar to that of Kawasaki disease (KD).
In one of the articles, Matthew Molloy, MD, MPH, of the division of pediatric hospital medicine at Cincinnati Children’s Hospital Medical Center, and colleagues aptly asked: “What are we missing in our search for MIS-C?”
E. coli, not SARS-CoV-2
That question arose from a case involving a 3-year-old boy who had a 6-day history of fever and fatigue. Three days earlier, he had tested negative for strep antigen and COVID-19. He had a persistent, high fever, reduced appetite, and reduced urine output and was taken to the ED. On physical examination, there was no rash, skin peeling, redness of the eye or oral mucosa, congestion, rhinorrhea, cough, shortness of breath, chest pain, abdominal pain, nausea, vomiting, or diarrhea.
Urinalysis results and exam findings were suspicious for pyelonephritis. Other findings from an extensive laboratory workup raised the alarm that the boy was suffering from MIS-C as opposed to incomplete KD. After admission to hospital medicine, the cardiology, rheumatology, and infectious disease teams were called in to consult.
Repeat labs were planned for the following day before initiating therapy. On day 2, the child’s urine culture was positive for gram-negative rods, later identified as Escherichia coli. The boy was started on ceftriaxone. Left renal scarring was apparent on ultrasound. The patient’s condition resolved after 36 hours, and he was discharged home with antibiotics.
‘Diagnosis derailed’
Calling this a case of “diagnosis derailed,” the authors noted that, in the pre-COVID era, this child’s signs and symptoms would likely have triggered a more targeted and less costly evaluation for more common infectious and noninfectious causes, including pyelonephritis, absent any physical exam findings consistent with KD.
“However, the patient presented in the midst of the COVID-19 pandemic with growing awareness of a new clinical entity,” Dr. Molloy and colleagues wrote. “Anchored to the patient’s persistent fever, the medical team initiated an extensive, costly, and ultimately unnecessary workup to avoid missing the diagnosis of MIS-C; a not yet well-described diagnosis with potentially severe morbidity.”
Confirmation bias and diagnostic momentum likely contributed to the early focus on MIS-C rather than more common alternatives, the authors acknowledged. The addition of mildly abnormal laboratory data not typically obtained in the evaluation of fever led the team astray. “The diagnosis and definitive treatment may have been made earlier had the focus on concern for MIS-C not been present,” Dr. Molloy said in an interview.
Keeping value in care
The authors recognized that their initial approach to evaluating for MIS-C provided low-value care. “In our desire to not ‘miss’ MIS-C, we were performing costly evaluations that at times produced mildly abnormal, nonspecific results,” they wrote. That triggered a cascade of specialty consultations, follow-up testing, and an unwarranted diagnostic preoccupation with MIS-C.
Determining the extra price tag for the child’s workup would be complex and difficult because there is a difference in the cost to the hospital and the cost to the family, Dr. Molloy said. “However, there are potential cost savings that would be related to making a correct diagnosis in a timely manner in terms of preventing downstream effects from delayed diagnoses.”
Even as clinicians struggle with the challenging SARS-CoV-2 learning curve, Dr. Molloy and associates urged them to continue to strive for high-value care, with an unwavering focus on using only necessary resources, a stewardship the pandemic has shown to be critical.
“The COVID-19 pandemic has been an incredibly stressful time for physicians and for families,” Dr. Molloy said. “COVID-19 and related conditions like MIS-C are new, and we are learning more and more about them every week. These diagnoses are understandably on the minds of physicians and families when children present with fever.” Notwithstanding, the boy’s case underscores the need for clinicians to consider alternate diagnoses and the value of the care provided.
Impact of bias
Dr. Molloy’s group brings home the cognitive biases practitioners often suffer from, including anchoring and confirmation bias and diagnostic momentum, according to J. Howard Smart, MD, chief of pediatrics at Sharp Mary Birch Hospital for Women and Newborns, San Diego, and an assistant clinical professor of pediatrics at University of California, San Diego.
“But it is one thing to recognize these in retrospect and quite another to consider whether they may be happening to you yourself in real time,” he said in an interview. “It is almost as if we need to have a ‘time out,’ where we stop and ask ourselves whether there is something else that could be explaining our patient’s presentation, something that would be more common and more likely to be occurring.”
According to Dr. Smart, who was not involved in Dr. Molloy’s study, the team’s premature diagnostic focus on MIS-C was almost the inverse of what typically happens with KD. “It is usually the case that Kawasaki disease does not enter the differential diagnosis until late in the course of the fever, typically on day 5 or later, when it may have been better to think of it earlier,” he said.
In the second article, Andrea Dean, MD, of the department of pediatrics at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, and colleagues outlined the cases of five patients aged 8-17 years who were hospitalized in May 2020 for suspected MIS-C. They exhibited inflammatory and other concerning indicators but were eventually discharged with a diagnosis of murine typhus.
This flea-borne infection, most commonly reported in the United States in the southeastern Gulf Coast region, Hawaii, and California, is often associated with a triad of fever, rash, and headache.
Cases have been rising in southern Texas, and Dr. Dean and colleagues postulated that school closures and social distancing may have increased exposure as a result of children spending more time outdoors or with pets. “Alternatively, parental concern for SARS-CoV-2 infection could mean children with symptoms are presenting to care and being referred or admitted to the hospital more frequently due to provider concern for MIS-C,” they wrote.
Cardiac involvement
The most concerning of the five cases in terms of possible MIS-C, Dr. Dean said in an interview, was that of a 12-year-old boy who had fever for 6 days in association with headache, eczematous rash, dry lips, and conjunctivitis. Laboratory tests showed a mildly elevated C-reactive protein level, hyponatremia, and thrombocytopenia, as well as sterile pyuria and mildly elevated prothrombin time. He was treated empirically with doxycycline, and his fever resolved over the next 24 hours.
An echocardiogram at initial evaluation, however, revealed mild dilation of the left anterior descending and right coronary arteries, which led to the administration of intravenous immunoglobulin and aspirin for atypical KD, in contrast to MIS-C. The authors postulated that mild cardiac involvement in disorders other than MIS-C and KD may be underrecognized.
The lesson from these cases, Dr. Dean and associates concluded, is that hospitalists must maintain a wide differential diagnosis when assessing a child with prolonged fever and evidence of systemic inflammation. The CDC stipulates that a diagnosis of MIS-C requires the absence of a plausible alternative diagnosis.
In addition to common viral, bacterial, and noninfectious disorders, a range of regional endemic rickettsial and parasitic infections must be considered as alternative diagnoses to MIS-C. “Many of these diseases cannot be reliably differentiated from MIS-C on presentation, and as community exposure to SARS-CoV-2 grows, hospitalists should be prepared to admit febrile children with evidence of systemic inflammation for brief observation periods to evaluate for MIS-C,” Dr. Dean’s group wrote. In this context, however, empiric treatment for common or even uncommon infectious diseases may avoid overdiagnosis and overtreatment of MIS-C as well as improve patient outcomes.
“We do have specific MIS-C guidelines at our institution,” Dr. Dean said, “but like all institutions, we are dealing with the broad definition of MIS-C according to the World Health Organization and the CDC, which is really the takeaway from this paper.”
More difficult differentiation
Both groups of authors pointed out that, as SARS-CoV-2 spreads throughout a community, a higher percentage of the population will have positive results on antibody testing, and such results will become less useful for differentiating between MIS-C and other conditions.
Despite these series’ cautionary lessons, other experts point to the critical importance of including MIS-C early on in the interest of efficient diagnosis and therapy. “In the cases cited, other pathologies were evaluated for and treated accordingly,” said Kara Gross Margolis, MD, AGAF, an associate professor of pediatrics in the division of pediatric gastroenterology, hepatology, and nutrition at Morgan Stanley Children’s Hospital,New York. “These papers stress the need for a balance that is important, and all potential diagnoses need to be considered, but MIS-C, due to its potential severe consequences, also needs to be on our differential now.”
In her view, as this new high-morbidity entity becomes more widespread during the pandemic, it will be increasingly important to keep this condition on the diagnostic radar.
Interestingly, in a converse example of diagnostic clouding, Dr. Gross Margolis’s group reported (Gastroenterology. 2020 Oct;159[4]:1571-4.e2) last year on a pediatric case series in which the presence of gastrointestinal symptoms in children with COVID-19–related MIS-C muddied the diagnosis by confusing this potentially severe syndrome with more common and less toxic gastrointestinal infections.
According to Dr. Smart, although the two reports don’t offer evidence for a particular diagnostic practice, they can inform the decision-making process. “It may be that we will have enough evidence shortly to say what the best practice is regarding diagnostic evaluation of possible MIS-C cases,” he said. “Until then, we must remember that common things occur commonly, even during a global pandemic.”
Neither of the two reports received any specific funding. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SARS-CoV-2 in hospitalized children and youth
Clinical syndromes and predictors of disease severity
Clinical questions: What are the demographics and clinical features of pediatric severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) syndromes, and which admitting demographics and clinical features are predictive of disease severity?
Background: In children, SARS-CoV-2 causes respiratory disease and multisystem inflammatory syndrome in children (MIS-C) as well as other clinical manifestations. The authors of this study chose to address the gap of identifying characteristics for severe disease caused by SARS-CoV-2, including respiratory disease, MIS-C and other manifestations.
Study design: Retrospective and prospective cohort analysis of hospitalized children
Setting: Participating hospitals in Tri-State Pediatric COVID-19 Consortium, including hospitals in New York, New Jersey, and Connecticut.
Synopsis: The authors identified hospitalized patients 22 years old or younger who had a positive SARS-CoV-2 test or met the U.S. Centers for Disease Control and Preventions’ MIS-C case definition. For comparative analysis, patients were divided into a respiratory disease group (based on the World Health Organization’s criteria for COVID-19), MIS-C group or other group (based on the primary reason for hospitalization).
The authors included 281 patients in the study. 51% of the patients presented with respiratory disease, 25% with MIS-C and 25% with other symptoms, including gastrointestinal, or fever. 51% of all patients were Hispanic and 23% were non-Black Hispanic. The most common pre-existing comorbidities amongst all groups were obesity (34%) and asthma (14%).
Patients with respiratory disease had a median age of 14 years while those with MIS-C had a median age of 7 years. Patients more commonly identified as non-Hispanic Black in the MIS-C group vs the respiratory group (35% vs. 18%). Obesity and medical complexity were more prevalent in the respiratory group relative to the MIS-C group. 75% of patients with MIS-C had gastrointestinal symptoms. 44% of respiratory patients had a chest radiograph with bilateral infiltrates on admission, and 18% or respiratory patients required invasive mechanical ventilation. The most common complications in the respiratory group were acute respiratory distress syndrome (17%) and acute kidney injury (11%), whereas shock (35%) and cardiac dysfunction (25%) were the most common complications in the MIS-C group. The median length of stay for all patients was 4 days (IQR 2-8 days).
Patients with MIS-C were more likely to be admitted to the intensive care unit (ICU) but all deaths (7 patients) occurred in the respiratory group. 40% of patients with respiratory disease, 56% of patients with MIS-C, and 6% of other patients met the authors’ definition of severe disease (ICU admission > 48 hours). For the respiratory group, younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph were independent predictors of severe disease based on multivariate analyses. For the MIS-C group, lower absolute lymphocyte count and increasing CRP at admission were independent predictors of severity.
Bottom line: Mortality in pediatric patients is low. Ethnicity and race were not predictive of disease severity in this model, even though 51% of the patients studied were Hispanic and 23% were non-Hispanic Black. Severity of illness for patients with respiratory disease was found to be associated with younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph. Severity of illness in patients with MIS-C was associated with lower absolute lymphocyte count and increasing CRP.
Citation: Fernandes DM, et al. Severe acute respiratory syndrome coronavirus 2 clinical syndromes and predictors of disease severity in hospitalized children and youth. J Pediatr. 2020 Nov 14;S0022-3476(20):31393-7. DOI: 10.1016/j.jpeds.2020.11.016.
Dr. Kumar is an assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of The Hospitalist.
Clinical syndromes and predictors of disease severity
Clinical syndromes and predictors of disease severity
Clinical questions: What are the demographics and clinical features of pediatric severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) syndromes, and which admitting demographics and clinical features are predictive of disease severity?
Background: In children, SARS-CoV-2 causes respiratory disease and multisystem inflammatory syndrome in children (MIS-C) as well as other clinical manifestations. The authors of this study chose to address the gap of identifying characteristics for severe disease caused by SARS-CoV-2, including respiratory disease, MIS-C and other manifestations.
Study design: Retrospective and prospective cohort analysis of hospitalized children
Setting: Participating hospitals in Tri-State Pediatric COVID-19 Consortium, including hospitals in New York, New Jersey, and Connecticut.
Synopsis: The authors identified hospitalized patients 22 years old or younger who had a positive SARS-CoV-2 test or met the U.S. Centers for Disease Control and Preventions’ MIS-C case definition. For comparative analysis, patients were divided into a respiratory disease group (based on the World Health Organization’s criteria for COVID-19), MIS-C group or other group (based on the primary reason for hospitalization).
The authors included 281 patients in the study. 51% of the patients presented with respiratory disease, 25% with MIS-C and 25% with other symptoms, including gastrointestinal, or fever. 51% of all patients were Hispanic and 23% were non-Black Hispanic. The most common pre-existing comorbidities amongst all groups were obesity (34%) and asthma (14%).
Patients with respiratory disease had a median age of 14 years while those with MIS-C had a median age of 7 years. Patients more commonly identified as non-Hispanic Black in the MIS-C group vs the respiratory group (35% vs. 18%). Obesity and medical complexity were more prevalent in the respiratory group relative to the MIS-C group. 75% of patients with MIS-C had gastrointestinal symptoms. 44% of respiratory patients had a chest radiograph with bilateral infiltrates on admission, and 18% or respiratory patients required invasive mechanical ventilation. The most common complications in the respiratory group were acute respiratory distress syndrome (17%) and acute kidney injury (11%), whereas shock (35%) and cardiac dysfunction (25%) were the most common complications in the MIS-C group. The median length of stay for all patients was 4 days (IQR 2-8 days).
Patients with MIS-C were more likely to be admitted to the intensive care unit (ICU) but all deaths (7 patients) occurred in the respiratory group. 40% of patients with respiratory disease, 56% of patients with MIS-C, and 6% of other patients met the authors’ definition of severe disease (ICU admission > 48 hours). For the respiratory group, younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph were independent predictors of severe disease based on multivariate analyses. For the MIS-C group, lower absolute lymphocyte count and increasing CRP at admission were independent predictors of severity.
Bottom line: Mortality in pediatric patients is low. Ethnicity and race were not predictive of disease severity in this model, even though 51% of the patients studied were Hispanic and 23% were non-Hispanic Black. Severity of illness for patients with respiratory disease was found to be associated with younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph. Severity of illness in patients with MIS-C was associated with lower absolute lymphocyte count and increasing CRP.
Citation: Fernandes DM, et al. Severe acute respiratory syndrome coronavirus 2 clinical syndromes and predictors of disease severity in hospitalized children and youth. J Pediatr. 2020 Nov 14;S0022-3476(20):31393-7. DOI: 10.1016/j.jpeds.2020.11.016.
Dr. Kumar is an assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of The Hospitalist.
Clinical questions: What are the demographics and clinical features of pediatric severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) syndromes, and which admitting demographics and clinical features are predictive of disease severity?
Background: In children, SARS-CoV-2 causes respiratory disease and multisystem inflammatory syndrome in children (MIS-C) as well as other clinical manifestations. The authors of this study chose to address the gap of identifying characteristics for severe disease caused by SARS-CoV-2, including respiratory disease, MIS-C and other manifestations.
Study design: Retrospective and prospective cohort analysis of hospitalized children
Setting: Participating hospitals in Tri-State Pediatric COVID-19 Consortium, including hospitals in New York, New Jersey, and Connecticut.
Synopsis: The authors identified hospitalized patients 22 years old or younger who had a positive SARS-CoV-2 test or met the U.S. Centers for Disease Control and Preventions’ MIS-C case definition. For comparative analysis, patients were divided into a respiratory disease group (based on the World Health Organization’s criteria for COVID-19), MIS-C group or other group (based on the primary reason for hospitalization).
The authors included 281 patients in the study. 51% of the patients presented with respiratory disease, 25% with MIS-C and 25% with other symptoms, including gastrointestinal, or fever. 51% of all patients were Hispanic and 23% were non-Black Hispanic. The most common pre-existing comorbidities amongst all groups were obesity (34%) and asthma (14%).
Patients with respiratory disease had a median age of 14 years while those with MIS-C had a median age of 7 years. Patients more commonly identified as non-Hispanic Black in the MIS-C group vs the respiratory group (35% vs. 18%). Obesity and medical complexity were more prevalent in the respiratory group relative to the MIS-C group. 75% of patients with MIS-C had gastrointestinal symptoms. 44% of respiratory patients had a chest radiograph with bilateral infiltrates on admission, and 18% or respiratory patients required invasive mechanical ventilation. The most common complications in the respiratory group were acute respiratory distress syndrome (17%) and acute kidney injury (11%), whereas shock (35%) and cardiac dysfunction (25%) were the most common complications in the MIS-C group. The median length of stay for all patients was 4 days (IQR 2-8 days).
Patients with MIS-C were more likely to be admitted to the intensive care unit (ICU) but all deaths (7 patients) occurred in the respiratory group. 40% of patients with respiratory disease, 56% of patients with MIS-C, and 6% of other patients met the authors’ definition of severe disease (ICU admission > 48 hours). For the respiratory group, younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph were independent predictors of severe disease based on multivariate analyses. For the MIS-C group, lower absolute lymphocyte count and increasing CRP at admission were independent predictors of severity.
Bottom line: Mortality in pediatric patients is low. Ethnicity and race were not predictive of disease severity in this model, even though 51% of the patients studied were Hispanic and 23% were non-Hispanic Black. Severity of illness for patients with respiratory disease was found to be associated with younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph. Severity of illness in patients with MIS-C was associated with lower absolute lymphocyte count and increasing CRP.
Citation: Fernandes DM, et al. Severe acute respiratory syndrome coronavirus 2 clinical syndromes and predictors of disease severity in hospitalized children and youth. J Pediatr. 2020 Nov 14;S0022-3476(20):31393-7. DOI: 10.1016/j.jpeds.2020.11.016.
Dr. Kumar is an assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of The Hospitalist.
FROM THE JOURNAL OF PEDIATRICS
Pediatric minority patients less likely to undergo ED imaging
Significant racial and ethnic differences in diagnostic imaging rates exist among children receiving care in pediatric EDs across the United States, Jennifer R. Marin, MD, of the University of Pittsburgh Medical Center Children’s Hospital of Pittsburgh, and associates reported.
Specifically, visits with non-Hispanic Black and Hispanic patients less frequently included radiography, CT, ultrasonography, and MRI than those of non-Hispanic White patients. The findings persisted across most diagnostic groups, even when stratified according to insurance type, Dr. Marin and colleagues reported in a multicenter cross-sectional study in JAMA Network Open.
The authors collected administrative data from the Pediatric Health Information System on 44 tertiary care children’s hospitals in 17 major metropolitan areas across the United States. They evaluated a total of 13,087,522 ED visits by 6,230,911 patients that occurred between Jan. 1, 2016, and Dec. 31, 2019. Of these, 28.2% included at least one imaging study. Altogether, 33.5% were performed on non-Hispanic White children, compared with just 24.1% of non-Hispanic Black children (adjusted odds ratio, 0.82) and 26.1% of Hispanic children (aOR, 0.87). After adjusting for relevant confounding factors, non-Hispanic Black and Hispanic children were less likely to have any imaging at all during their visits.
“Our findings suggest that a child’s race and ethnicity may be independently associated with the decision to perform imaging during ED visits,” Dr. Marin and associates said, adding that “the differential use of diagnostic imaging by race/ethnicity may reflect underuse of imaging in non-Hispanic Black and Hispanic children, or alternatively, overuse in non-Hispanic White children.”
Overuse vs. underuse: Racial bias or parental anxiety?
Overuse of imaging carries its own risks, but underuse can lead to misdiagnosis, the need for additional care, and possibly worse outcomes in the long run, Dr. Marin and colleagues explained. “Although we were unable to discern underuse from overuse using an administrative database, it is likely that much of the imaging in White children is unnecessary.”
Higher parental anxiety was just one of the explanations the authors offered for excessive imaging in White children. Especially in cases of diagnostic imaging for head trauma, one survey of adult ED patients showed that the peace of mind CT offers with its more definitive diagnosis was worth the additional possible risk of radiation.
Language barriers in non–English-speaking patients may also affect likelihood of testing as part of an ED visit.
Implicit physician racial bias, which can be amplified under the stress of working in an ED, can affect patient interactions, treatment decisions and adherence, and ultimately overall health outcomes, the authors noted. The goal in ensuring parity is to routinely follow clinical guidelines and use objective scoring tools that minimize subjectivity. At the institutional level, internal quality assurance evaluations go a long way toward understanding and limiting bias.
Historically, White patients are more likely than minority patients to have a medical home, which can influence whether ED physicians order imaging studies and whether imaging of White patients may have been triggered by a primary care physician referral, Dr. Marin and associates said.
In an accompanying editorial, Anupam B. Kharbanda, MD, said these findings are “consistent with decades of previous research documenting inequalities in health care delivery ... [and] must be examined in the context of inequities within the social framework of a community.”
Going back to the drawing board
“Physicians, researchers, and health care leaders must partner with the communities they serve to develop and implement interventions to address these substantial inequities in care,” said Dr. Kharbanda, pediatric emergency medicine physician at Children’s Minnesota Hospital, Minneapolis. As previous studies have demonstrated, implicit bias and antiracism training are needed to help physicians develop empathy so they are better equipped to help patients and families in a multicultural environment. Partnering with community-based organizations to ensure that care is more community centered, as has been done successfully within the Kaiser Permanente health system, for example, and employing a more diverse workforce that mirrors the populations cared for will go a long way.
Citing a 1966 speech of the late Dr. Martin Luther King, Jr., in which he said: “Of all the forms of inequality, injustice in health care is the most shocking and inhumane,” Dr. Kharbanda urged clinicians to not only hear but believe these words and act on them by working in partnerships with the communities they serve.
In a separate interview, Walter Palmer, MD, pediatric emergency medicine fellow at Children’s National Medical Center, Washington, noted: “This study’s findings are disappointing and yet not at all unexpected, as the authors convincingly identify yet another step at which patients of color are treated unequally in the U.S. health care system. It highlights a frightening truth: That we are all at risk of letting invisible implicit biases impact our clinical decision-making process. This is especially true in the busy emergency department environment, where pressure to make swift decisions regarding diagnostic workup and management invites the influence of imperceptible biases. It is now incumbent upon us as health care providers to monitor our personal and departmental patterns of practice for areas in which we can improve racial health equity and become advocates for the children and families who entrust us with their care.”
The authors reported multiple financial disclosures. Dr. Kharbanda and Dr. Palmer had no conflicts of interest and reported no disclosures.
Significant racial and ethnic differences in diagnostic imaging rates exist among children receiving care in pediatric EDs across the United States, Jennifer R. Marin, MD, of the University of Pittsburgh Medical Center Children’s Hospital of Pittsburgh, and associates reported.
Specifically, visits with non-Hispanic Black and Hispanic patients less frequently included radiography, CT, ultrasonography, and MRI than those of non-Hispanic White patients. The findings persisted across most diagnostic groups, even when stratified according to insurance type, Dr. Marin and colleagues reported in a multicenter cross-sectional study in JAMA Network Open.
The authors collected administrative data from the Pediatric Health Information System on 44 tertiary care children’s hospitals in 17 major metropolitan areas across the United States. They evaluated a total of 13,087,522 ED visits by 6,230,911 patients that occurred between Jan. 1, 2016, and Dec. 31, 2019. Of these, 28.2% included at least one imaging study. Altogether, 33.5% were performed on non-Hispanic White children, compared with just 24.1% of non-Hispanic Black children (adjusted odds ratio, 0.82) and 26.1% of Hispanic children (aOR, 0.87). After adjusting for relevant confounding factors, non-Hispanic Black and Hispanic children were less likely to have any imaging at all during their visits.
“Our findings suggest that a child’s race and ethnicity may be independently associated with the decision to perform imaging during ED visits,” Dr. Marin and associates said, adding that “the differential use of diagnostic imaging by race/ethnicity may reflect underuse of imaging in non-Hispanic Black and Hispanic children, or alternatively, overuse in non-Hispanic White children.”
Overuse vs. underuse: Racial bias or parental anxiety?
Overuse of imaging carries its own risks, but underuse can lead to misdiagnosis, the need for additional care, and possibly worse outcomes in the long run, Dr. Marin and colleagues explained. “Although we were unable to discern underuse from overuse using an administrative database, it is likely that much of the imaging in White children is unnecessary.”
Higher parental anxiety was just one of the explanations the authors offered for excessive imaging in White children. Especially in cases of diagnostic imaging for head trauma, one survey of adult ED patients showed that the peace of mind CT offers with its more definitive diagnosis was worth the additional possible risk of radiation.
Language barriers in non–English-speaking patients may also affect likelihood of testing as part of an ED visit.
Implicit physician racial bias, which can be amplified under the stress of working in an ED, can affect patient interactions, treatment decisions and adherence, and ultimately overall health outcomes, the authors noted. The goal in ensuring parity is to routinely follow clinical guidelines and use objective scoring tools that minimize subjectivity. At the institutional level, internal quality assurance evaluations go a long way toward understanding and limiting bias.
Historically, White patients are more likely than minority patients to have a medical home, which can influence whether ED physicians order imaging studies and whether imaging of White patients may have been triggered by a primary care physician referral, Dr. Marin and associates said.
In an accompanying editorial, Anupam B. Kharbanda, MD, said these findings are “consistent with decades of previous research documenting inequalities in health care delivery ... [and] must be examined in the context of inequities within the social framework of a community.”
Going back to the drawing board
“Physicians, researchers, and health care leaders must partner with the communities they serve to develop and implement interventions to address these substantial inequities in care,” said Dr. Kharbanda, pediatric emergency medicine physician at Children’s Minnesota Hospital, Minneapolis. As previous studies have demonstrated, implicit bias and antiracism training are needed to help physicians develop empathy so they are better equipped to help patients and families in a multicultural environment. Partnering with community-based organizations to ensure that care is more community centered, as has been done successfully within the Kaiser Permanente health system, for example, and employing a more diverse workforce that mirrors the populations cared for will go a long way.
Citing a 1966 speech of the late Dr. Martin Luther King, Jr., in which he said: “Of all the forms of inequality, injustice in health care is the most shocking and inhumane,” Dr. Kharbanda urged clinicians to not only hear but believe these words and act on them by working in partnerships with the communities they serve.
In a separate interview, Walter Palmer, MD, pediatric emergency medicine fellow at Children’s National Medical Center, Washington, noted: “This study’s findings are disappointing and yet not at all unexpected, as the authors convincingly identify yet another step at which patients of color are treated unequally in the U.S. health care system. It highlights a frightening truth: That we are all at risk of letting invisible implicit biases impact our clinical decision-making process. This is especially true in the busy emergency department environment, where pressure to make swift decisions regarding diagnostic workup and management invites the influence of imperceptible biases. It is now incumbent upon us as health care providers to monitor our personal and departmental patterns of practice for areas in which we can improve racial health equity and become advocates for the children and families who entrust us with their care.”
The authors reported multiple financial disclosures. Dr. Kharbanda and Dr. Palmer had no conflicts of interest and reported no disclosures.
Significant racial and ethnic differences in diagnostic imaging rates exist among children receiving care in pediatric EDs across the United States, Jennifer R. Marin, MD, of the University of Pittsburgh Medical Center Children’s Hospital of Pittsburgh, and associates reported.
Specifically, visits with non-Hispanic Black and Hispanic patients less frequently included radiography, CT, ultrasonography, and MRI than those of non-Hispanic White patients. The findings persisted across most diagnostic groups, even when stratified according to insurance type, Dr. Marin and colleagues reported in a multicenter cross-sectional study in JAMA Network Open.
The authors collected administrative data from the Pediatric Health Information System on 44 tertiary care children’s hospitals in 17 major metropolitan areas across the United States. They evaluated a total of 13,087,522 ED visits by 6,230,911 patients that occurred between Jan. 1, 2016, and Dec. 31, 2019. Of these, 28.2% included at least one imaging study. Altogether, 33.5% were performed on non-Hispanic White children, compared with just 24.1% of non-Hispanic Black children (adjusted odds ratio, 0.82) and 26.1% of Hispanic children (aOR, 0.87). After adjusting for relevant confounding factors, non-Hispanic Black and Hispanic children were less likely to have any imaging at all during their visits.
“Our findings suggest that a child’s race and ethnicity may be independently associated with the decision to perform imaging during ED visits,” Dr. Marin and associates said, adding that “the differential use of diagnostic imaging by race/ethnicity may reflect underuse of imaging in non-Hispanic Black and Hispanic children, or alternatively, overuse in non-Hispanic White children.”
Overuse vs. underuse: Racial bias or parental anxiety?
Overuse of imaging carries its own risks, but underuse can lead to misdiagnosis, the need for additional care, and possibly worse outcomes in the long run, Dr. Marin and colleagues explained. “Although we were unable to discern underuse from overuse using an administrative database, it is likely that much of the imaging in White children is unnecessary.”
Higher parental anxiety was just one of the explanations the authors offered for excessive imaging in White children. Especially in cases of diagnostic imaging for head trauma, one survey of adult ED patients showed that the peace of mind CT offers with its more definitive diagnosis was worth the additional possible risk of radiation.
Language barriers in non–English-speaking patients may also affect likelihood of testing as part of an ED visit.
Implicit physician racial bias, which can be amplified under the stress of working in an ED, can affect patient interactions, treatment decisions and adherence, and ultimately overall health outcomes, the authors noted. The goal in ensuring parity is to routinely follow clinical guidelines and use objective scoring tools that minimize subjectivity. At the institutional level, internal quality assurance evaluations go a long way toward understanding and limiting bias.
Historically, White patients are more likely than minority patients to have a medical home, which can influence whether ED physicians order imaging studies and whether imaging of White patients may have been triggered by a primary care physician referral, Dr. Marin and associates said.
In an accompanying editorial, Anupam B. Kharbanda, MD, said these findings are “consistent with decades of previous research documenting inequalities in health care delivery ... [and] must be examined in the context of inequities within the social framework of a community.”
Going back to the drawing board
“Physicians, researchers, and health care leaders must partner with the communities they serve to develop and implement interventions to address these substantial inequities in care,” said Dr. Kharbanda, pediatric emergency medicine physician at Children’s Minnesota Hospital, Minneapolis. As previous studies have demonstrated, implicit bias and antiracism training are needed to help physicians develop empathy so they are better equipped to help patients and families in a multicultural environment. Partnering with community-based organizations to ensure that care is more community centered, as has been done successfully within the Kaiser Permanente health system, for example, and employing a more diverse workforce that mirrors the populations cared for will go a long way.
Citing a 1966 speech of the late Dr. Martin Luther King, Jr., in which he said: “Of all the forms of inequality, injustice in health care is the most shocking and inhumane,” Dr. Kharbanda urged clinicians to not only hear but believe these words and act on them by working in partnerships with the communities they serve.
In a separate interview, Walter Palmer, MD, pediatric emergency medicine fellow at Children’s National Medical Center, Washington, noted: “This study’s findings are disappointing and yet not at all unexpected, as the authors convincingly identify yet another step at which patients of color are treated unequally in the U.S. health care system. It highlights a frightening truth: That we are all at risk of letting invisible implicit biases impact our clinical decision-making process. This is especially true in the busy emergency department environment, where pressure to make swift decisions regarding diagnostic workup and management invites the influence of imperceptible biases. It is now incumbent upon us as health care providers to monitor our personal and departmental patterns of practice for areas in which we can improve racial health equity and become advocates for the children and families who entrust us with their care.”
The authors reported multiple financial disclosures. Dr. Kharbanda and Dr. Palmer had no conflicts of interest and reported no disclosures.
FROM JAMA NETWORK OPEN