User login
Researchers examine vitamin D, skin pigmentation, and outcomes of pediatric MS
PHILADELPHIA – according to research described at the annual meeting of the American Academy of Neurology. Future research will be required to understand the interactions between dietary vitamin D ingestion, sun exposure, pigmentation of sun-exposed skin, seasonal vitamin D concentrations, and the genetic influences of vitamin D pathways with MS risk.
Race, vitamin D status, HLA-DRB1*15 genotype, and place of residence during childhood all affect the risk of MS. The place of residence also can affect exposure to ultraviolet radiation and, thus, dermal pigmentation.
Candice Dunn, a clinical research coordinator at the Children’s Hospital of Philadelphia, and colleagues conducted a prospective study to determine whether HLA-DRB1*15 status, 25-hydroxyvitamin D (25[OH]D) levels measured at baseline, and skin tone are associated with MS outcome in children with ADS. They enrolled 259 children with incident ADS in a multisite study in Toronto and Philadelphia (latitudes 43° to 51°). The investigators measured non–sun-exposed upper inner arm melanin content using the DSM II ColorMeter device. They measured 25(OH)D concentrations in serum obtained within 60 days of symptom onset and compared them with laboratory-reported normative values. Vitamin D insufficiency was defined as 25(OH)D less than 75 nmol/L. Ms. Dunn and colleagues quantified HLA-DRB1*15 alleles using allele-specific polymerase chain reaction amplification. Statistical analysis was performed using Spearman correlation models and Wilcoxon or Kruskal-Wallis tests as appropriate.
In all, 68 children were diagnosed with MS, 191 remained monophasic (monoADS). Approximately 46% of children with MS were HLA-DRB1*15-positive, compared with 29.9% of monoADS children. In addition, children with MS had significantly lower 25(OH)D levels (mean, 45.4 nmol/L) than monoADS children (mean, 61.9 nmol/L) at baseline. Non–sun-exposed skin tone measured in the upper inner arm did not differ between children diagnosed with MS (mean melanin index, 46.4) and monoADS (mean melanin index, 43.5). Furthermore, 25(OH)D levels correlated with upper inner arm melanin index in the MS group, but not in children with monoADS.
Ms. Dunn had nothing to disclose, but various coauthors have received compensation from companies such as Novartis, Merck, Teva, Celgene, and Genentech.
SOURCE: Dunn C et al. AAN 2019, Abstract S19.007.
PHILADELPHIA – according to research described at the annual meeting of the American Academy of Neurology. Future research will be required to understand the interactions between dietary vitamin D ingestion, sun exposure, pigmentation of sun-exposed skin, seasonal vitamin D concentrations, and the genetic influences of vitamin D pathways with MS risk.
Race, vitamin D status, HLA-DRB1*15 genotype, and place of residence during childhood all affect the risk of MS. The place of residence also can affect exposure to ultraviolet radiation and, thus, dermal pigmentation.
Candice Dunn, a clinical research coordinator at the Children’s Hospital of Philadelphia, and colleagues conducted a prospective study to determine whether HLA-DRB1*15 status, 25-hydroxyvitamin D (25[OH]D) levels measured at baseline, and skin tone are associated with MS outcome in children with ADS. They enrolled 259 children with incident ADS in a multisite study in Toronto and Philadelphia (latitudes 43° to 51°). The investigators measured non–sun-exposed upper inner arm melanin content using the DSM II ColorMeter device. They measured 25(OH)D concentrations in serum obtained within 60 days of symptom onset and compared them with laboratory-reported normative values. Vitamin D insufficiency was defined as 25(OH)D less than 75 nmol/L. Ms. Dunn and colleagues quantified HLA-DRB1*15 alleles using allele-specific polymerase chain reaction amplification. Statistical analysis was performed using Spearman correlation models and Wilcoxon or Kruskal-Wallis tests as appropriate.
In all, 68 children were diagnosed with MS, 191 remained monophasic (monoADS). Approximately 46% of children with MS were HLA-DRB1*15-positive, compared with 29.9% of monoADS children. In addition, children with MS had significantly lower 25(OH)D levels (mean, 45.4 nmol/L) than monoADS children (mean, 61.9 nmol/L) at baseline. Non–sun-exposed skin tone measured in the upper inner arm did not differ between children diagnosed with MS (mean melanin index, 46.4) and monoADS (mean melanin index, 43.5). Furthermore, 25(OH)D levels correlated with upper inner arm melanin index in the MS group, but not in children with monoADS.
Ms. Dunn had nothing to disclose, but various coauthors have received compensation from companies such as Novartis, Merck, Teva, Celgene, and Genentech.
SOURCE: Dunn C et al. AAN 2019, Abstract S19.007.
PHILADELPHIA – according to research described at the annual meeting of the American Academy of Neurology. Future research will be required to understand the interactions between dietary vitamin D ingestion, sun exposure, pigmentation of sun-exposed skin, seasonal vitamin D concentrations, and the genetic influences of vitamin D pathways with MS risk.
Race, vitamin D status, HLA-DRB1*15 genotype, and place of residence during childhood all affect the risk of MS. The place of residence also can affect exposure to ultraviolet radiation and, thus, dermal pigmentation.
Candice Dunn, a clinical research coordinator at the Children’s Hospital of Philadelphia, and colleagues conducted a prospective study to determine whether HLA-DRB1*15 status, 25-hydroxyvitamin D (25[OH]D) levels measured at baseline, and skin tone are associated with MS outcome in children with ADS. They enrolled 259 children with incident ADS in a multisite study in Toronto and Philadelphia (latitudes 43° to 51°). The investigators measured non–sun-exposed upper inner arm melanin content using the DSM II ColorMeter device. They measured 25(OH)D concentrations in serum obtained within 60 days of symptom onset and compared them with laboratory-reported normative values. Vitamin D insufficiency was defined as 25(OH)D less than 75 nmol/L. Ms. Dunn and colleagues quantified HLA-DRB1*15 alleles using allele-specific polymerase chain reaction amplification. Statistical analysis was performed using Spearman correlation models and Wilcoxon or Kruskal-Wallis tests as appropriate.
In all, 68 children were diagnosed with MS, 191 remained monophasic (monoADS). Approximately 46% of children with MS were HLA-DRB1*15-positive, compared with 29.9% of monoADS children. In addition, children with MS had significantly lower 25(OH)D levels (mean, 45.4 nmol/L) than monoADS children (mean, 61.9 nmol/L) at baseline. Non–sun-exposed skin tone measured in the upper inner arm did not differ between children diagnosed with MS (mean melanin index, 46.4) and monoADS (mean melanin index, 43.5). Furthermore, 25(OH)D levels correlated with upper inner arm melanin index in the MS group, but not in children with monoADS.
Ms. Dunn had nothing to disclose, but various coauthors have received compensation from companies such as Novartis, Merck, Teva, Celgene, and Genentech.
SOURCE: Dunn C et al. AAN 2019, Abstract S19.007.
REPORTING FROM AAN 2019
Key clinical point: The relationship between vitamin D status and MS outcome in children relates to skin pigmentation.
Major finding: About 46% of children with MS were HLA-DRB1*15 positive.
Study details: A multisite, prospective study of 259 children with MS.
Disclosures: Ms. Dunn had no disclosures, but various coauthors have received compensation from companies such as Novartis, Merck, Teva, Celgene, and Genentech.
Source: Dunn C et al. AAN 2019, Abstract S19.007.
Gabapentin falls short in treating sickle cell pain
NEW ORLEANS – Adding gabapentin to standard therapy did not significantly reduce vaso-occlusive pain in most patients with sickle cell disease enrolled in a phase 2 trial.
In the entire cohort, there were no significant differences in pain response between patients who received gabapentin and those who received placebo. However, patients with the HbSS genotype had a significantly greater decrease in pain score from baseline to discharge if they received gabapentin rather than placebo.
Additional studies are needed to confirm these findings because this trial was limited by a small sample size, according to study investigator Latika Puri, MD, of St. Jude Children’s Research Hospital in Memphis. Dr. Puri presented the trial at the annual meeting of the American Society of Pediatric Hematology/Oncology.
The trial included 86 evaluable patients who had vaso-occlusive pain and a pain score of at least 4. All patients received standard therapy for vaso-occlusive pain and were randomized to receive placebo (n = 44) or a single oral dose of gabapentin at 15 mg/kg (n = 42).
Baseline characteristics were similar between the treatment arms. For the entire cohort, the mean age was 11.8 years (range, 1-21 years), and 51% of patients were male. Forty-four patients had the HbSS genotype, 25 had the HbSC genotype, 8 had HbS/beta0-thalassemia, and 9 had other genotypes.
The mean pain score at baseline was 7.8 for the entire cohort, 8.0 for the gabapentin arm, and 7.7 for the placebo arm.
For the entire cohort, there was no significant difference in pain response between the gabapentin and placebo arms.
The proportion of patients who experienced a greater than 33% decrease in pain from baseline to 3 hours posttreatment was 67% in the gabapentin arm and 59% in the placebo arm (P = .23). The proportion of patients who experienced a greater than 33% decrease from baseline to discharge from the acute care clinic was 75% and 61%, respectively (P = .18).
In the entire cohort, decreases in pain scores from baseline to 3 hours posttreatment were not significantly different between the gabapentin and placebo arms, at 1.3 and 0.7, respectively (P = .74). Likewise, decreases in pain scores from baseline to discharge were not significantly different, at 1.6 and 0.8 (P = .38).
Among patients who had the HbSS genotype, there was a significantly greater decrease in pain score from baseline to discharge in the gabapentin arm than in the placebo arm, 5.9 versus 3.6 (P = .03). However, there were no other significant differences in pain response for the HbSS subgroup.
There were no significant differences in opioid consumption or hospitalization for the HbSS subgroup or the entire cohort. For the entire cohort, the mean morphine equivalent dose from baseline to 3 hours posttreatment was 0.16 mg/kg in the gabapentin arm and 0.17 mg/kg in the placebo arm (P = .89). For the HbSS subgroup, the mean dose was 0.16 mg/kg and 0.15 mg/kg, respectively (P = .93).
In the entire cohort, 24% of patients in the gabapentin arm and 27% of those in the placebo arm were hospitalized (P = .71). In the HbSS subgroup, hospitalizations occurred in 11% and 35% (P = .15).
Dr. Puri pointed out several challenges that led to limitations in this study. Specifically, the investigators had to obtain patient consent while delivering standard treatment, while patients were in pain and distress, and from patients who had already received opioids and were sleepy. Additionally, gabapentin had to be delivered within 1 hour of opioid administration, and a lack of after-hours staff limited enrollment.
“These challenges led to one of our biggest limitations, which was a small sample size, leading to a limited power to observe real differences,” Dr. Puri said. “We also defined a very short time period of evaluation for the primary outcomes; that was 3 hours from the gabapentin dose or placebo dose. This limited our capability to see real differences if they existed.”
Dr. Puri said additional studies with larger sample sizes are needed to confirm these findings. She added that efforts to better characterize pain in sickle cell disease could reveal patients who may benefit from gabapentin because they have a neuropathic component to their pain.
The trial was sponsored by St. Jude Children’s Research Hospital in collaboration with Scan|Design Foundation. Dr. Puri did not provide disclosure information at the meeting.
SOURCE: Puri L et al. ASPHO 2019, Abstract 2011.
NEW ORLEANS – Adding gabapentin to standard therapy did not significantly reduce vaso-occlusive pain in most patients with sickle cell disease enrolled in a phase 2 trial.
In the entire cohort, there were no significant differences in pain response between patients who received gabapentin and those who received placebo. However, patients with the HbSS genotype had a significantly greater decrease in pain score from baseline to discharge if they received gabapentin rather than placebo.
Additional studies are needed to confirm these findings because this trial was limited by a small sample size, according to study investigator Latika Puri, MD, of St. Jude Children’s Research Hospital in Memphis. Dr. Puri presented the trial at the annual meeting of the American Society of Pediatric Hematology/Oncology.
The trial included 86 evaluable patients who had vaso-occlusive pain and a pain score of at least 4. All patients received standard therapy for vaso-occlusive pain and were randomized to receive placebo (n = 44) or a single oral dose of gabapentin at 15 mg/kg (n = 42).
Baseline characteristics were similar between the treatment arms. For the entire cohort, the mean age was 11.8 years (range, 1-21 years), and 51% of patients were male. Forty-four patients had the HbSS genotype, 25 had the HbSC genotype, 8 had HbS/beta0-thalassemia, and 9 had other genotypes.
The mean pain score at baseline was 7.8 for the entire cohort, 8.0 for the gabapentin arm, and 7.7 for the placebo arm.
For the entire cohort, there was no significant difference in pain response between the gabapentin and placebo arms.
The proportion of patients who experienced a greater than 33% decrease in pain from baseline to 3 hours posttreatment was 67% in the gabapentin arm and 59% in the placebo arm (P = .23). The proportion of patients who experienced a greater than 33% decrease from baseline to discharge from the acute care clinic was 75% and 61%, respectively (P = .18).
In the entire cohort, decreases in pain scores from baseline to 3 hours posttreatment were not significantly different between the gabapentin and placebo arms, at 1.3 and 0.7, respectively (P = .74). Likewise, decreases in pain scores from baseline to discharge were not significantly different, at 1.6 and 0.8 (P = .38).
Among patients who had the HbSS genotype, there was a significantly greater decrease in pain score from baseline to discharge in the gabapentin arm than in the placebo arm, 5.9 versus 3.6 (P = .03). However, there were no other significant differences in pain response for the HbSS subgroup.
There were no significant differences in opioid consumption or hospitalization for the HbSS subgroup or the entire cohort. For the entire cohort, the mean morphine equivalent dose from baseline to 3 hours posttreatment was 0.16 mg/kg in the gabapentin arm and 0.17 mg/kg in the placebo arm (P = .89). For the HbSS subgroup, the mean dose was 0.16 mg/kg and 0.15 mg/kg, respectively (P = .93).
In the entire cohort, 24% of patients in the gabapentin arm and 27% of those in the placebo arm were hospitalized (P = .71). In the HbSS subgroup, hospitalizations occurred in 11% and 35% (P = .15).
Dr. Puri pointed out several challenges that led to limitations in this study. Specifically, the investigators had to obtain patient consent while delivering standard treatment, while patients were in pain and distress, and from patients who had already received opioids and were sleepy. Additionally, gabapentin had to be delivered within 1 hour of opioid administration, and a lack of after-hours staff limited enrollment.
“These challenges led to one of our biggest limitations, which was a small sample size, leading to a limited power to observe real differences,” Dr. Puri said. “We also defined a very short time period of evaluation for the primary outcomes; that was 3 hours from the gabapentin dose or placebo dose. This limited our capability to see real differences if they existed.”
Dr. Puri said additional studies with larger sample sizes are needed to confirm these findings. She added that efforts to better characterize pain in sickle cell disease could reveal patients who may benefit from gabapentin because they have a neuropathic component to their pain.
The trial was sponsored by St. Jude Children’s Research Hospital in collaboration with Scan|Design Foundation. Dr. Puri did not provide disclosure information at the meeting.
SOURCE: Puri L et al. ASPHO 2019, Abstract 2011.
NEW ORLEANS – Adding gabapentin to standard therapy did not significantly reduce vaso-occlusive pain in most patients with sickle cell disease enrolled in a phase 2 trial.
In the entire cohort, there were no significant differences in pain response between patients who received gabapentin and those who received placebo. However, patients with the HbSS genotype had a significantly greater decrease in pain score from baseline to discharge if they received gabapentin rather than placebo.
Additional studies are needed to confirm these findings because this trial was limited by a small sample size, according to study investigator Latika Puri, MD, of St. Jude Children’s Research Hospital in Memphis. Dr. Puri presented the trial at the annual meeting of the American Society of Pediatric Hematology/Oncology.
The trial included 86 evaluable patients who had vaso-occlusive pain and a pain score of at least 4. All patients received standard therapy for vaso-occlusive pain and were randomized to receive placebo (n = 44) or a single oral dose of gabapentin at 15 mg/kg (n = 42).
Baseline characteristics were similar between the treatment arms. For the entire cohort, the mean age was 11.8 years (range, 1-21 years), and 51% of patients were male. Forty-four patients had the HbSS genotype, 25 had the HbSC genotype, 8 had HbS/beta0-thalassemia, and 9 had other genotypes.
The mean pain score at baseline was 7.8 for the entire cohort, 8.0 for the gabapentin arm, and 7.7 for the placebo arm.
For the entire cohort, there was no significant difference in pain response between the gabapentin and placebo arms.
The proportion of patients who experienced a greater than 33% decrease in pain from baseline to 3 hours posttreatment was 67% in the gabapentin arm and 59% in the placebo arm (P = .23). The proportion of patients who experienced a greater than 33% decrease from baseline to discharge from the acute care clinic was 75% and 61%, respectively (P = .18).
In the entire cohort, decreases in pain scores from baseline to 3 hours posttreatment were not significantly different between the gabapentin and placebo arms, at 1.3 and 0.7, respectively (P = .74). Likewise, decreases in pain scores from baseline to discharge were not significantly different, at 1.6 and 0.8 (P = .38).
Among patients who had the HbSS genotype, there was a significantly greater decrease in pain score from baseline to discharge in the gabapentin arm than in the placebo arm, 5.9 versus 3.6 (P = .03). However, there were no other significant differences in pain response for the HbSS subgroup.
There were no significant differences in opioid consumption or hospitalization for the HbSS subgroup or the entire cohort. For the entire cohort, the mean morphine equivalent dose from baseline to 3 hours posttreatment was 0.16 mg/kg in the gabapentin arm and 0.17 mg/kg in the placebo arm (P = .89). For the HbSS subgroup, the mean dose was 0.16 mg/kg and 0.15 mg/kg, respectively (P = .93).
In the entire cohort, 24% of patients in the gabapentin arm and 27% of those in the placebo arm were hospitalized (P = .71). In the HbSS subgroup, hospitalizations occurred in 11% and 35% (P = .15).
Dr. Puri pointed out several challenges that led to limitations in this study. Specifically, the investigators had to obtain patient consent while delivering standard treatment, while patients were in pain and distress, and from patients who had already received opioids and were sleepy. Additionally, gabapentin had to be delivered within 1 hour of opioid administration, and a lack of after-hours staff limited enrollment.
“These challenges led to one of our biggest limitations, which was a small sample size, leading to a limited power to observe real differences,” Dr. Puri said. “We also defined a very short time period of evaluation for the primary outcomes; that was 3 hours from the gabapentin dose or placebo dose. This limited our capability to see real differences if they existed.”
Dr. Puri said additional studies with larger sample sizes are needed to confirm these findings. She added that efforts to better characterize pain in sickle cell disease could reveal patients who may benefit from gabapentin because they have a neuropathic component to their pain.
The trial was sponsored by St. Jude Children’s Research Hospital in collaboration with Scan|Design Foundation. Dr. Puri did not provide disclosure information at the meeting.
SOURCE: Puri L et al. ASPHO 2019, Abstract 2011.
REPORTING FROM THE 2019 ASPHO CONFERENCE
Key clinical point:
Major finding: The proportion of patients who experienced a greater than 33% decrease in pain from baseline to 3 hours posttreatment was 67% in the gabapentin arm and 59% in the placebo arm (P = .23).
Study details: A phase 2 trial of 86 evaluable patients.
Disclosures: The trial was sponsored by St. Jude Children’s Research Hospital in collaboration with Scan|Design Foundation. The speaker did not provide disclosure information at the meeting.
Source: Puri L et al. 2019 ASPHO Conference, Abstract 2011.
United States up to 764 measles cases for the year
The number of U.S. measles cases in 2019 rose by 60 during the week ending May 3, which puts the new postelimination high at 764 cases for the year, according to the Centers for Disease Control and Prevention.
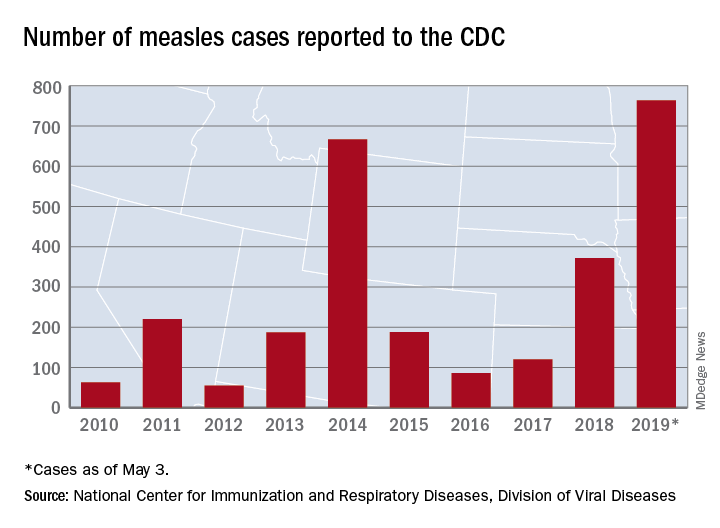
The CDC is currently tracking nine measles outbreaks in six states. The largest outbreak this year has been in New York City, mainly Brooklyn, which has almost half (367) of all cases in the country. An outbreak in New York’s Rockland County has resulted in another 109 cases so far this year. California is experiencing outbreaks in three counties – Butte, Los Angeles, and Sacramento – and the state was up to 40 confirmed cases as of May 1. The other states with outbreaks are Michigan, New Jersey, Georgia, and Maryland.
Twenty-three states now have reported measles cases in 2019, as officials in Pittsburgh reported Pennsylvania’s first case on April 30. Four additional cases in the area were reported on May 2 by the Allegheny County Health Department. A measles outbreak is “defined as three or more cases” by the CDC, but the situation in Pennsylvania is not yet being reported as such.
One Pennsylvania legislator in the state, Rep. Daryl Metcalfe (R) of Butler County, has authored a bill that would “bar health care practitioners and facilities and insurance companies from denying care if parents refuse or delay” recommended vaccinations, PennLive.com reported.
Pennsylvania Governor Tom Wolf (D) said that the “bill would put children, pregnant women, and vulnerable patients at risk of being exposed to horrific diseases – at the doctor’s office.”
The number of U.S. measles cases in 2019 rose by 60 during the week ending May 3, which puts the new postelimination high at 764 cases for the year, according to the Centers for Disease Control and Prevention.

The CDC is currently tracking nine measles outbreaks in six states. The largest outbreak this year has been in New York City, mainly Brooklyn, which has almost half (367) of all cases in the country. An outbreak in New York’s Rockland County has resulted in another 109 cases so far this year. California is experiencing outbreaks in three counties – Butte, Los Angeles, and Sacramento – and the state was up to 40 confirmed cases as of May 1. The other states with outbreaks are Michigan, New Jersey, Georgia, and Maryland.
Twenty-three states now have reported measles cases in 2019, as officials in Pittsburgh reported Pennsylvania’s first case on April 30. Four additional cases in the area were reported on May 2 by the Allegheny County Health Department. A measles outbreak is “defined as three or more cases” by the CDC, but the situation in Pennsylvania is not yet being reported as such.
One Pennsylvania legislator in the state, Rep. Daryl Metcalfe (R) of Butler County, has authored a bill that would “bar health care practitioners and facilities and insurance companies from denying care if parents refuse or delay” recommended vaccinations, PennLive.com reported.
Pennsylvania Governor Tom Wolf (D) said that the “bill would put children, pregnant women, and vulnerable patients at risk of being exposed to horrific diseases – at the doctor’s office.”
The number of U.S. measles cases in 2019 rose by 60 during the week ending May 3, which puts the new postelimination high at 764 cases for the year, according to the Centers for Disease Control and Prevention.

The CDC is currently tracking nine measles outbreaks in six states. The largest outbreak this year has been in New York City, mainly Brooklyn, which has almost half (367) of all cases in the country. An outbreak in New York’s Rockland County has resulted in another 109 cases so far this year. California is experiencing outbreaks in three counties – Butte, Los Angeles, and Sacramento – and the state was up to 40 confirmed cases as of May 1. The other states with outbreaks are Michigan, New Jersey, Georgia, and Maryland.
Twenty-three states now have reported measles cases in 2019, as officials in Pittsburgh reported Pennsylvania’s first case on April 30. Four additional cases in the area were reported on May 2 by the Allegheny County Health Department. A measles outbreak is “defined as three or more cases” by the CDC, but the situation in Pennsylvania is not yet being reported as such.
One Pennsylvania legislator in the state, Rep. Daryl Metcalfe (R) of Butler County, has authored a bill that would “bar health care practitioners and facilities and insurance companies from denying care if parents refuse or delay” recommended vaccinations, PennLive.com reported.
Pennsylvania Governor Tom Wolf (D) said that the “bill would put children, pregnant women, and vulnerable patients at risk of being exposed to horrific diseases – at the doctor’s office.”
Restroom, locker room restrictions foster abuse of transgender teens
Transgender youth with restricted restroom and locker room use at school were significantly more likely to experience sexual assault at school than those without such restrictions, based on surveys from more than 3,000 teens in the United States who identified as transgender or nonbinary.
“Little is known about risk factors for sexual assault in gender minority adolescents, but school policies and practices play an important role in other forms of victimization,” including restricting transgender students from using restrooms or locker rooms that match their gender identities, wrote Gabriel R. Murchison, MPH, of Harvard University, Boston, Mass., and colleagues in Pediatrics (2019 May 6. doi: https://doi.org/10.1542/peds.2018-2902).
To examine the relationship between school restroom/locker room policies and sexual assault on transgender teens, the researchers reviewed data from the Lesbian, Gay, Bisexual, Transgender, and Queer or Questioning (LGBTQ) Teen Study, an anonymous web-based survey of U.S. adolescents aged 13 to 17 years who could read and write in English. Participants were assigned to one of four gender groups: trans male, trans female, nonbinary who were assigned male at birth (AMAB), or nonbinary who were assigned female at birth (AFAB) based on the survey questions asking their sex assigned at birth and current gender identity. The final study population of 3,673 individuals included 1,359 boys and 1,947 nonbinary youth AFAB and 158 transgender girls and 209 nonbinary youth AMAB. The results were published in Pediatrics.
with risk ratios of 1.26 for transgender boys and 2.49 for transgender girls, and 1.42 for nonbinary AFAB youth. Restroom/locker room restrictions were not significantly associated with sexual abuse in nonbinary AMAB youth.
The 12-month prevalence of sexual assault was highest among nonbinary youth AFAB (27%), followed by 26.5% among transgender boys, 18.5% among transgender girls, and 17.6% among nonbinary youth AMAB.
Sexual assault was determined based on participants’ response to the question, “During the past 12 months, how many times did anyone force you to do sexual things that you did not want to do? (Count such things as kissing, touching, or being physically forced to have sexual intercourse.)” The researchers adjusted for multiple factors associated with adolescent sexual assault including alcohol use, family connectedness, and educational attainment of caregivers; as well as variables including exposure to antitransgender stigma and perception of teacher support at school.
The researchers also identified four mediating variables: sexual harassment, feeling safe in restrooms and locker rooms, feeling safe in other locations at school, and classmates’ knowledge of gender status.
“Significant indirect effects were present for all 4 mediating variables,” which included feel safe in restrooms and locker rooms, feel safe elsewhere in school, classmates know gender minority status, and sexual harassment. The fourth mediating variable mentioned fully explains “the association between restroom and locker room restrictions and sexual assault victimization,” the researchers wrote.
The findings were limited by several factors including the lack of racial diversity and the reliance on cross-sectional, nonprobability data, the researchers said.
However, the results are strengthened by the large sample size and suggest that avoiding restrictive policies at school can make a difference in reducing abuse of transgender teens, they wrote.
“From a prevention perspective, pediatricians are key advocates for transgender and nonbinary patients, and their role may include educating school officials and submitting letters confirming the patient’s need to express their gender identity,” that emphasize the importance of “safe, identity-congruent restrooms and locker rooms,” the researchers concluded.
The study was supported in part by the Office of Vice President for Research at the University of Connecticut, and the Human Rights Campaign Foundation provided in-kind support for the LGBTQ Teen Study. Mr. Murchison disclosed participation in survey development and data collection for the LGBTQ Teen Study as an employee of the Human Rights Campaign Foundation.
SOURCE: Murchison G et al. Pediatrics . 2019 May 6. doi: https://doi.org/10.1542/peds.2018-2902 .
The study findings “make a compelling case for what we as gender specialist providers witness every day in our work: failure to support transgender and gender-expansive youth in being able to fully live in their affirmed gender puts them at physical as well as psychological risk,” wrote Diane Ehrensaft, PhD, and Stephen M. Rosenthal, MD, in an accompanying editorial.
What can reduce the risk of these youth experiencing abuse and assault, according to the editorialists, is putting policies in place that support them. Dr. Ehrensaft and Dr. Rosenthal cited the state of California’s 2013 decision to allow all students in public schools, from kindergarten through 12th grade “the right to use the bathroom and locker room consistent with their affirmed gender identity” as an example of something pediatricians should be advocating for in other states.
Restrictions on bathroom use to their assigned birth identity may cause transgender youth to be at increased risk for verbal and physical harassment and abuse, they said. It may also lead some to avoid restroom use and increase their risk for urinary tract infections, impacted stool, and school avoidance, the editorialists noted. They added that “[such] policies are often fear based, with nontransgender students thought to be the ones at risk for sexual assault by transgender intruders, by anyone whose genitalia does not match the one associated with the sign on the door, or by predators posing as transgender students.” The editorialists noted that these attitudes can come from school personnel or parents and that pediatricians should be aware of “the high prevalence of sexual assault” on transgender and gender nonbinary youth” (Pediatrics. 2019 May 6. doi: 10.1542/peds.2019-0554).
Dr. Ehrensaft and Dr. Rosenthal are affiliated with the Benioff Children’s Hospital at the University of California, San Francisco. They had no financial conflicts to disclose.
The study findings “make a compelling case for what we as gender specialist providers witness every day in our work: failure to support transgender and gender-expansive youth in being able to fully live in their affirmed gender puts them at physical as well as psychological risk,” wrote Diane Ehrensaft, PhD, and Stephen M. Rosenthal, MD, in an accompanying editorial.
What can reduce the risk of these youth experiencing abuse and assault, according to the editorialists, is putting policies in place that support them. Dr. Ehrensaft and Dr. Rosenthal cited the state of California’s 2013 decision to allow all students in public schools, from kindergarten through 12th grade “the right to use the bathroom and locker room consistent with their affirmed gender identity” as an example of something pediatricians should be advocating for in other states.
Restrictions on bathroom use to their assigned birth identity may cause transgender youth to be at increased risk for verbal and physical harassment and abuse, they said. It may also lead some to avoid restroom use and increase their risk for urinary tract infections, impacted stool, and school avoidance, the editorialists noted. They added that “[such] policies are often fear based, with nontransgender students thought to be the ones at risk for sexual assault by transgender intruders, by anyone whose genitalia does not match the one associated with the sign on the door, or by predators posing as transgender students.” The editorialists noted that these attitudes can come from school personnel or parents and that pediatricians should be aware of “the high prevalence of sexual assault” on transgender and gender nonbinary youth” (Pediatrics. 2019 May 6. doi: 10.1542/peds.2019-0554).
Dr. Ehrensaft and Dr. Rosenthal are affiliated with the Benioff Children’s Hospital at the University of California, San Francisco. They had no financial conflicts to disclose.
The study findings “make a compelling case for what we as gender specialist providers witness every day in our work: failure to support transgender and gender-expansive youth in being able to fully live in their affirmed gender puts them at physical as well as psychological risk,” wrote Diane Ehrensaft, PhD, and Stephen M. Rosenthal, MD, in an accompanying editorial.
What can reduce the risk of these youth experiencing abuse and assault, according to the editorialists, is putting policies in place that support them. Dr. Ehrensaft and Dr. Rosenthal cited the state of California’s 2013 decision to allow all students in public schools, from kindergarten through 12th grade “the right to use the bathroom and locker room consistent with their affirmed gender identity” as an example of something pediatricians should be advocating for in other states.
Restrictions on bathroom use to their assigned birth identity may cause transgender youth to be at increased risk for verbal and physical harassment and abuse, they said. It may also lead some to avoid restroom use and increase their risk for urinary tract infections, impacted stool, and school avoidance, the editorialists noted. They added that “[such] policies are often fear based, with nontransgender students thought to be the ones at risk for sexual assault by transgender intruders, by anyone whose genitalia does not match the one associated with the sign on the door, or by predators posing as transgender students.” The editorialists noted that these attitudes can come from school personnel or parents and that pediatricians should be aware of “the high prevalence of sexual assault” on transgender and gender nonbinary youth” (Pediatrics. 2019 May 6. doi: 10.1542/peds.2019-0554).
Dr. Ehrensaft and Dr. Rosenthal are affiliated with the Benioff Children’s Hospital at the University of California, San Francisco. They had no financial conflicts to disclose.
Transgender youth with restricted restroom and locker room use at school were significantly more likely to experience sexual assault at school than those without such restrictions, based on surveys from more than 3,000 teens in the United States who identified as transgender or nonbinary.
“Little is known about risk factors for sexual assault in gender minority adolescents, but school policies and practices play an important role in other forms of victimization,” including restricting transgender students from using restrooms or locker rooms that match their gender identities, wrote Gabriel R. Murchison, MPH, of Harvard University, Boston, Mass., and colleagues in Pediatrics (2019 May 6. doi: https://doi.org/10.1542/peds.2018-2902).
To examine the relationship between school restroom/locker room policies and sexual assault on transgender teens, the researchers reviewed data from the Lesbian, Gay, Bisexual, Transgender, and Queer or Questioning (LGBTQ) Teen Study, an anonymous web-based survey of U.S. adolescents aged 13 to 17 years who could read and write in English. Participants were assigned to one of four gender groups: trans male, trans female, nonbinary who were assigned male at birth (AMAB), or nonbinary who were assigned female at birth (AFAB) based on the survey questions asking their sex assigned at birth and current gender identity. The final study population of 3,673 individuals included 1,359 boys and 1,947 nonbinary youth AFAB and 158 transgender girls and 209 nonbinary youth AMAB. The results were published in Pediatrics.
with risk ratios of 1.26 for transgender boys and 2.49 for transgender girls, and 1.42 for nonbinary AFAB youth. Restroom/locker room restrictions were not significantly associated with sexual abuse in nonbinary AMAB youth.
The 12-month prevalence of sexual assault was highest among nonbinary youth AFAB (27%), followed by 26.5% among transgender boys, 18.5% among transgender girls, and 17.6% among nonbinary youth AMAB.
Sexual assault was determined based on participants’ response to the question, “During the past 12 months, how many times did anyone force you to do sexual things that you did not want to do? (Count such things as kissing, touching, or being physically forced to have sexual intercourse.)” The researchers adjusted for multiple factors associated with adolescent sexual assault including alcohol use, family connectedness, and educational attainment of caregivers; as well as variables including exposure to antitransgender stigma and perception of teacher support at school.
The researchers also identified four mediating variables: sexual harassment, feeling safe in restrooms and locker rooms, feeling safe in other locations at school, and classmates’ knowledge of gender status.
“Significant indirect effects were present for all 4 mediating variables,” which included feel safe in restrooms and locker rooms, feel safe elsewhere in school, classmates know gender minority status, and sexual harassment. The fourth mediating variable mentioned fully explains “the association between restroom and locker room restrictions and sexual assault victimization,” the researchers wrote.
The findings were limited by several factors including the lack of racial diversity and the reliance on cross-sectional, nonprobability data, the researchers said.
However, the results are strengthened by the large sample size and suggest that avoiding restrictive policies at school can make a difference in reducing abuse of transgender teens, they wrote.
“From a prevention perspective, pediatricians are key advocates for transgender and nonbinary patients, and their role may include educating school officials and submitting letters confirming the patient’s need to express their gender identity,” that emphasize the importance of “safe, identity-congruent restrooms and locker rooms,” the researchers concluded.
The study was supported in part by the Office of Vice President for Research at the University of Connecticut, and the Human Rights Campaign Foundation provided in-kind support for the LGBTQ Teen Study. Mr. Murchison disclosed participation in survey development and data collection for the LGBTQ Teen Study as an employee of the Human Rights Campaign Foundation.
SOURCE: Murchison G et al. Pediatrics . 2019 May 6. doi: https://doi.org/10.1542/peds.2018-2902 .
Transgender youth with restricted restroom and locker room use at school were significantly more likely to experience sexual assault at school than those without such restrictions, based on surveys from more than 3,000 teens in the United States who identified as transgender or nonbinary.
“Little is known about risk factors for sexual assault in gender minority adolescents, but school policies and practices play an important role in other forms of victimization,” including restricting transgender students from using restrooms or locker rooms that match their gender identities, wrote Gabriel R. Murchison, MPH, of Harvard University, Boston, Mass., and colleagues in Pediatrics (2019 May 6. doi: https://doi.org/10.1542/peds.2018-2902).
To examine the relationship between school restroom/locker room policies and sexual assault on transgender teens, the researchers reviewed data from the Lesbian, Gay, Bisexual, Transgender, and Queer or Questioning (LGBTQ) Teen Study, an anonymous web-based survey of U.S. adolescents aged 13 to 17 years who could read and write in English. Participants were assigned to one of four gender groups: trans male, trans female, nonbinary who were assigned male at birth (AMAB), or nonbinary who were assigned female at birth (AFAB) based on the survey questions asking their sex assigned at birth and current gender identity. The final study population of 3,673 individuals included 1,359 boys and 1,947 nonbinary youth AFAB and 158 transgender girls and 209 nonbinary youth AMAB. The results were published in Pediatrics.
with risk ratios of 1.26 for transgender boys and 2.49 for transgender girls, and 1.42 for nonbinary AFAB youth. Restroom/locker room restrictions were not significantly associated with sexual abuse in nonbinary AMAB youth.
The 12-month prevalence of sexual assault was highest among nonbinary youth AFAB (27%), followed by 26.5% among transgender boys, 18.5% among transgender girls, and 17.6% among nonbinary youth AMAB.
Sexual assault was determined based on participants’ response to the question, “During the past 12 months, how many times did anyone force you to do sexual things that you did not want to do? (Count such things as kissing, touching, or being physically forced to have sexual intercourse.)” The researchers adjusted for multiple factors associated with adolescent sexual assault including alcohol use, family connectedness, and educational attainment of caregivers; as well as variables including exposure to antitransgender stigma and perception of teacher support at school.
The researchers also identified four mediating variables: sexual harassment, feeling safe in restrooms and locker rooms, feeling safe in other locations at school, and classmates’ knowledge of gender status.
“Significant indirect effects were present for all 4 mediating variables,” which included feel safe in restrooms and locker rooms, feel safe elsewhere in school, classmates know gender minority status, and sexual harassment. The fourth mediating variable mentioned fully explains “the association between restroom and locker room restrictions and sexual assault victimization,” the researchers wrote.
The findings were limited by several factors including the lack of racial diversity and the reliance on cross-sectional, nonprobability data, the researchers said.
However, the results are strengthened by the large sample size and suggest that avoiding restrictive policies at school can make a difference in reducing abuse of transgender teens, they wrote.
“From a prevention perspective, pediatricians are key advocates for transgender and nonbinary patients, and their role may include educating school officials and submitting letters confirming the patient’s need to express their gender identity,” that emphasize the importance of “safe, identity-congruent restrooms and locker rooms,” the researchers concluded.
The study was supported in part by the Office of Vice President for Research at the University of Connecticut, and the Human Rights Campaign Foundation provided in-kind support for the LGBTQ Teen Study. Mr. Murchison disclosed participation in survey development and data collection for the LGBTQ Teen Study as an employee of the Human Rights Campaign Foundation.
SOURCE: Murchison G et al. Pediatrics . 2019 May 6. doi: https://doi.org/10.1542/peds.2018-2902 .
FROM PEDIATRICS
Key clinical point: Restrictive restroom and locker room environments in schools promote abuse of transgender teens.
Major finding: Sexual assault was significantly more likely against transgender teens with restroom and locker room restrictions vs those without restrictions, with risk ratios of 1.26 for transgender boys and 2.49 for transgender girls.
Study details: The data came from web-based surveys of 3,673 teens aged 13 to 17 years who identified as transgender or nonbinary.
Disclosures: The study was supported in part by the Office of Vice President for Research at the University of Connecticut, and the Human Rights Campaign Foundation provided in-kind support for the LGBTQ Teen Study. Mr. Murchison disclosed participation in survey development and data collection for the LGBTQ Teen Study as an employee of the Human Rights Campaign Foundation.
Source: Murchison G et al. Pediatrics. 2019 May 6. doi: https://doi.org/10.1542/peds.2018-2902
A gentler approach to gastroschisis improves outcomes
BALTIMORE – a condition in which infants are born with their intestines and sometimes other organs protruding through a hole beside the umbilicus.
Neonatologists, maternal-fetal health experts, and pediatric surgeons standardized a literature-based approach that was gentler and less invasive than usual management, emphasizing sutureless closure, sometimes at bedside on the first day of life, and early feeding. Often, it turned out, that’s all that children require.
It’s made a big difference. “We reduced the number of trips to the operating room and exposure to general anesthesia. We reduced the number of babies intubated and days on the ventilator. We reduced opioid days and antibiotic days” without increasing bacteremia, and “there are probably long-term benefits beyond the NICU,” said Kara Calkins, MD, at the Pediatric Academic Societies annual meeting.
I think this is definitely ahead of the curve for NICUs. My hope is that the vast majority of universities adopt a similar approach,” said Dr. Calkins, who is an assistant professor of neonatology at the University of California, Los Angeles.
“When I was a fellow,” she explained, “we took all of these babies and intubated them right away and put them on a drip to paralyze and sedate them. We put their bowels into a silo,” essentially a plastic bag suspended by a string, in the hopes that gravity would pull the bowels back into the abdomen. More often than not, however, “the surgeon would come by every day and slowly push them” back in over a week or so. “The fear was if you did it too quickly, you’d invoke an abdominal compartment syndrome, or respiratory decompensation. You had a baby intubated for a week, sedated and paralyzed.”
Infants were kept on total parenteral nutrition for weeks, sometimes through a Broviac central catheter.
It was overkill, Dr. Calkins said, when only the intestines are out and the abdominal wall defect isn’t too large or too small, which is the case for many infants.
For those children, sutureless closure over 1-3 days is the new goal. The bowel is worked back into the abdomen and the umbilical cord is pulled to the side to approximate the edges of the wound, and tacked down; the defect then heals itself. Antibiotics are discontinued 48 hours after closure. Gastric and rectal decompression helps with reduction.
Also, “we give drops of breast milk in their cheek right away, every couple of hours starting on the first day of life. Once the output from the gastric tube is clear, we start feeds. We still give total parenteral nutrition, but through a [peripherally inserted central catheter] in the arm,” Dr. Calkins said. “Use of breast milk for this population is important” to help establish a healthy microbiome, among other reasons.
Another improvement that had been made, according to Dr. Calkins, is that if only the intestines are out, women carry their baby to term and deliver vaginally. The old practice was to deliver babies preterm by Cesarean section, she explained.
To see how it’s worked out, Dr. Calkins and her colleagues reviewed 70 gastroschisis cases managed under the new guidelines. They were uncomplicated cases, with no intestinal atresia, stricture, or ischemia.
Paralysis was avoided for silo placement in 53 infants (76%) and 32 (46%) avoided intubation. Antibiotics were discontinued in 56 (80%) within 48 hours of abdominal wall closure, and routine narcotics were discontinued in 53 infants (76%). Feeds were initiated in almost all children within 48 hours of non-bilious gastric tube output.
Compared with 168 infants treated before the changes were made, silo placement dropped from 71% to 58% of infants, and total ventilator days from a median of 5 to 2.
There was no difference in length of stay, perhaps because the “intestinal dysmotility intrinsic to gastroschisis remains a rate limiting factor for discharge,” the team concluded.
There was no industry funding, and Dr. Calkins didn’t have any disclosures.
SOURCE: Rottkamp CA et al., PAS 2019. Abstract 51.
BALTIMORE – a condition in which infants are born with their intestines and sometimes other organs protruding through a hole beside the umbilicus.
Neonatologists, maternal-fetal health experts, and pediatric surgeons standardized a literature-based approach that was gentler and less invasive than usual management, emphasizing sutureless closure, sometimes at bedside on the first day of life, and early feeding. Often, it turned out, that’s all that children require.
It’s made a big difference. “We reduced the number of trips to the operating room and exposure to general anesthesia. We reduced the number of babies intubated and days on the ventilator. We reduced opioid days and antibiotic days” without increasing bacteremia, and “there are probably long-term benefits beyond the NICU,” said Kara Calkins, MD, at the Pediatric Academic Societies annual meeting.
I think this is definitely ahead of the curve for NICUs. My hope is that the vast majority of universities adopt a similar approach,” said Dr. Calkins, who is an assistant professor of neonatology at the University of California, Los Angeles.
“When I was a fellow,” she explained, “we took all of these babies and intubated them right away and put them on a drip to paralyze and sedate them. We put their bowels into a silo,” essentially a plastic bag suspended by a string, in the hopes that gravity would pull the bowels back into the abdomen. More often than not, however, “the surgeon would come by every day and slowly push them” back in over a week or so. “The fear was if you did it too quickly, you’d invoke an abdominal compartment syndrome, or respiratory decompensation. You had a baby intubated for a week, sedated and paralyzed.”
Infants were kept on total parenteral nutrition for weeks, sometimes through a Broviac central catheter.
It was overkill, Dr. Calkins said, when only the intestines are out and the abdominal wall defect isn’t too large or too small, which is the case for many infants.
For those children, sutureless closure over 1-3 days is the new goal. The bowel is worked back into the abdomen and the umbilical cord is pulled to the side to approximate the edges of the wound, and tacked down; the defect then heals itself. Antibiotics are discontinued 48 hours after closure. Gastric and rectal decompression helps with reduction.
Also, “we give drops of breast milk in their cheek right away, every couple of hours starting on the first day of life. Once the output from the gastric tube is clear, we start feeds. We still give total parenteral nutrition, but through a [peripherally inserted central catheter] in the arm,” Dr. Calkins said. “Use of breast milk for this population is important” to help establish a healthy microbiome, among other reasons.
Another improvement that had been made, according to Dr. Calkins, is that if only the intestines are out, women carry their baby to term and deliver vaginally. The old practice was to deliver babies preterm by Cesarean section, she explained.
To see how it’s worked out, Dr. Calkins and her colleagues reviewed 70 gastroschisis cases managed under the new guidelines. They were uncomplicated cases, with no intestinal atresia, stricture, or ischemia.
Paralysis was avoided for silo placement in 53 infants (76%) and 32 (46%) avoided intubation. Antibiotics were discontinued in 56 (80%) within 48 hours of abdominal wall closure, and routine narcotics were discontinued in 53 infants (76%). Feeds were initiated in almost all children within 48 hours of non-bilious gastric tube output.
Compared with 168 infants treated before the changes were made, silo placement dropped from 71% to 58% of infants, and total ventilator days from a median of 5 to 2.
There was no difference in length of stay, perhaps because the “intestinal dysmotility intrinsic to gastroschisis remains a rate limiting factor for discharge,” the team concluded.
There was no industry funding, and Dr. Calkins didn’t have any disclosures.
SOURCE: Rottkamp CA et al., PAS 2019. Abstract 51.
BALTIMORE – a condition in which infants are born with their intestines and sometimes other organs protruding through a hole beside the umbilicus.
Neonatologists, maternal-fetal health experts, and pediatric surgeons standardized a literature-based approach that was gentler and less invasive than usual management, emphasizing sutureless closure, sometimes at bedside on the first day of life, and early feeding. Often, it turned out, that’s all that children require.
It’s made a big difference. “We reduced the number of trips to the operating room and exposure to general anesthesia. We reduced the number of babies intubated and days on the ventilator. We reduced opioid days and antibiotic days” without increasing bacteremia, and “there are probably long-term benefits beyond the NICU,” said Kara Calkins, MD, at the Pediatric Academic Societies annual meeting.
I think this is definitely ahead of the curve for NICUs. My hope is that the vast majority of universities adopt a similar approach,” said Dr. Calkins, who is an assistant professor of neonatology at the University of California, Los Angeles.
“When I was a fellow,” she explained, “we took all of these babies and intubated them right away and put them on a drip to paralyze and sedate them. We put their bowels into a silo,” essentially a plastic bag suspended by a string, in the hopes that gravity would pull the bowels back into the abdomen. More often than not, however, “the surgeon would come by every day and slowly push them” back in over a week or so. “The fear was if you did it too quickly, you’d invoke an abdominal compartment syndrome, or respiratory decompensation. You had a baby intubated for a week, sedated and paralyzed.”
Infants were kept on total parenteral nutrition for weeks, sometimes through a Broviac central catheter.
It was overkill, Dr. Calkins said, when only the intestines are out and the abdominal wall defect isn’t too large or too small, which is the case for many infants.
For those children, sutureless closure over 1-3 days is the new goal. The bowel is worked back into the abdomen and the umbilical cord is pulled to the side to approximate the edges of the wound, and tacked down; the defect then heals itself. Antibiotics are discontinued 48 hours after closure. Gastric and rectal decompression helps with reduction.
Also, “we give drops of breast milk in their cheek right away, every couple of hours starting on the first day of life. Once the output from the gastric tube is clear, we start feeds. We still give total parenteral nutrition, but through a [peripherally inserted central catheter] in the arm,” Dr. Calkins said. “Use of breast milk for this population is important” to help establish a healthy microbiome, among other reasons.
Another improvement that had been made, according to Dr. Calkins, is that if only the intestines are out, women carry their baby to term and deliver vaginally. The old practice was to deliver babies preterm by Cesarean section, she explained.
To see how it’s worked out, Dr. Calkins and her colleagues reviewed 70 gastroschisis cases managed under the new guidelines. They were uncomplicated cases, with no intestinal atresia, stricture, or ischemia.
Paralysis was avoided for silo placement in 53 infants (76%) and 32 (46%) avoided intubation. Antibiotics were discontinued in 56 (80%) within 48 hours of abdominal wall closure, and routine narcotics were discontinued in 53 infants (76%). Feeds were initiated in almost all children within 48 hours of non-bilious gastric tube output.
Compared with 168 infants treated before the changes were made, silo placement dropped from 71% to 58% of infants, and total ventilator days from a median of 5 to 2.
There was no difference in length of stay, perhaps because the “intestinal dysmotility intrinsic to gastroschisis remains a rate limiting factor for discharge,” the team concluded.
There was no industry funding, and Dr. Calkins didn’t have any disclosures.
SOURCE: Rottkamp CA et al., PAS 2019. Abstract 51.
REPORTING FROM PAS 2019
FDA approves first vaccine for prevention of dengue disease
The vaccine was approved for children aged 9-16 years who live in endemic areas and have previously had laboratory-confirmed dengue disease.
Dengue is endemic in the U.S. territories of American Samoa, Guam, Puerto Rico, and the U.S. Virgin Islands, according to an FDA statement announcing the approval.
While the first infection with dengue virus typically results in either no symptoms or a mild illness that can be mistaken for the flu, a second infection can lead to a more severe form of the disease, including dengue hemorrhagic fever, which can be fatal. About 95% of hospitalized patients with dengue disease have a second dengue virus infection.
FDA approval of Dengvaxia is based on results from three randomized, placebo-controlled studies of 35,000 individuals in dengue-endemic areas. The vaccine was about 76% effective in preventing symptomatic, laboratory-confirmed dengue disease in people aged 9-16 years with a previous dengue diagnosis. The most common adverse events were headache, muscle pain, joint pain, fatigue, injection site pain, and low-grade fever; the frequency of adverse events decreased after each subsequent dose.
“Infection by one type of dengue virus usually provides immunity against that specific serotype, but a subsequent infection by any of the other three serotypes of the virus increases the risk of developing severe dengue disease. ... The FDA’s approval of this vaccine will help protect people previously infected with dengue virus from subsequent development of dengue disease,” Peter Marks, MD, director of the FDA’s Center for Biologics Evaluation and Research, said in the FDA statement.
The vaccine was approved for children aged 9-16 years who live in endemic areas and have previously had laboratory-confirmed dengue disease.
Dengue is endemic in the U.S. territories of American Samoa, Guam, Puerto Rico, and the U.S. Virgin Islands, according to an FDA statement announcing the approval.
While the first infection with dengue virus typically results in either no symptoms or a mild illness that can be mistaken for the flu, a second infection can lead to a more severe form of the disease, including dengue hemorrhagic fever, which can be fatal. About 95% of hospitalized patients with dengue disease have a second dengue virus infection.
FDA approval of Dengvaxia is based on results from three randomized, placebo-controlled studies of 35,000 individuals in dengue-endemic areas. The vaccine was about 76% effective in preventing symptomatic, laboratory-confirmed dengue disease in people aged 9-16 years with a previous dengue diagnosis. The most common adverse events were headache, muscle pain, joint pain, fatigue, injection site pain, and low-grade fever; the frequency of adverse events decreased after each subsequent dose.
“Infection by one type of dengue virus usually provides immunity against that specific serotype, but a subsequent infection by any of the other three serotypes of the virus increases the risk of developing severe dengue disease. ... The FDA’s approval of this vaccine will help protect people previously infected with dengue virus from subsequent development of dengue disease,” Peter Marks, MD, director of the FDA’s Center for Biologics Evaluation and Research, said in the FDA statement.
The vaccine was approved for children aged 9-16 years who live in endemic areas and have previously had laboratory-confirmed dengue disease.
Dengue is endemic in the U.S. territories of American Samoa, Guam, Puerto Rico, and the U.S. Virgin Islands, according to an FDA statement announcing the approval.
While the first infection with dengue virus typically results in either no symptoms or a mild illness that can be mistaken for the flu, a second infection can lead to a more severe form of the disease, including dengue hemorrhagic fever, which can be fatal. About 95% of hospitalized patients with dengue disease have a second dengue virus infection.
FDA approval of Dengvaxia is based on results from three randomized, placebo-controlled studies of 35,000 individuals in dengue-endemic areas. The vaccine was about 76% effective in preventing symptomatic, laboratory-confirmed dengue disease in people aged 9-16 years with a previous dengue diagnosis. The most common adverse events were headache, muscle pain, joint pain, fatigue, injection site pain, and low-grade fever; the frequency of adverse events decreased after each subsequent dose.
“Infection by one type of dengue virus usually provides immunity against that specific serotype, but a subsequent infection by any of the other three serotypes of the virus increases the risk of developing severe dengue disease. ... The FDA’s approval of this vaccine will help protect people previously infected with dengue virus from subsequent development of dengue disease,” Peter Marks, MD, director of the FDA’s Center for Biologics Evaluation and Research, said in the FDA statement.
Liraglutide seems safe, effective in children already on metformin
The addition of liraglutide to metformin shows significantly improved glycemic control in children and adolescents with type 2 diabetes, compared with metformin alone, according to data presented at the Pediatric Academic Societies annual meeting in Baltimore.
The phase 3 study, which was simultaneously published in the New England Journal of Medicine, involved 134 patients aged 10-17 years with type 2 diabetes who were managing their diabetes with diet and exercise, metformin, or insulin.
Participants were randomized either to subcutaneous liraglutide – dose-escalated up to 1.8 mg/day, depending on efficacy and side effects – or placebo for 52 weeks. The first 26 weeks were double blind and the second 26 weeks were an open-label extension period.
At 26 weeks, mean glycated hemoglobin levels in the liraglutide group had decreased by 0.64 percentage points from baseline, but in the placebo group they had increased by 0.42 percentage points, representing a treatment difference of –1.06 percentage points (P less than .001). By week 52, the treatment difference between the two groups had increased to –1.30 percentage points.
William V. Tamborlane, MD, from the department of pediatrics at Yale University, New Haven, Conn., and his coauthors wrote that metformin is the approved drug of choice for pediatric patients with type 2 diabetes, and that insulin currently is the only approved option for those who do not have an adequate response to metformin monotherapy.
“This discrepancy in available treatments for youth as compared with adults persists because of a lack of successfully completed trials needed for approval of new drugs for the treatment of type 2 diabetes in children since a trial of metformin was completed in 1999,” they wrote.
The study showed that significantly more patients in the liraglutide group (63.7%) achieved glycated hemoglobin levels below 7%, compared with 36.5% of patients in the placebo group. Fasting plasma glucose levels were decreased in the liraglutide group at both 26 and 52 weeks, but had increased in the placebo group.
Although the number of reported adverse events were similar between the two groups, there were significantly more reports of gastrointestinal adverse events – particularly nausea – in patients taking liraglutide, compared with those on placebo.
However, the study did not show a difference between liraglutide and placebo in lowering body mass index, although mean body weight decreases – which were seen in both groups – were maintained at week 52 only in the liraglutide group. The authors suggested this might be owing to the relatively small number of patients enrolled in the study and that some of the children were still growing.
Novo Nordisk, which manufactures liraglutide, supported the study. Twelve authors reported grants or support from Novo Nordisk in relation to the trial. Three authors were employees of Novo Nordisk. Eight authors reported unrelated grants and fees from Novo Nordisk and other pharmaceutical companies.
SOURCE: Tamborlane WV et al. N Engl J Med. 2019 Apr 28. doi: 10.1056/NEJMoa1903822.
The addition of liraglutide to metformin shows significantly improved glycemic control in children and adolescents with type 2 diabetes, compared with metformin alone, according to data presented at the Pediatric Academic Societies annual meeting in Baltimore.
The phase 3 study, which was simultaneously published in the New England Journal of Medicine, involved 134 patients aged 10-17 years with type 2 diabetes who were managing their diabetes with diet and exercise, metformin, or insulin.
Participants were randomized either to subcutaneous liraglutide – dose-escalated up to 1.8 mg/day, depending on efficacy and side effects – or placebo for 52 weeks. The first 26 weeks were double blind and the second 26 weeks were an open-label extension period.
At 26 weeks, mean glycated hemoglobin levels in the liraglutide group had decreased by 0.64 percentage points from baseline, but in the placebo group they had increased by 0.42 percentage points, representing a treatment difference of –1.06 percentage points (P less than .001). By week 52, the treatment difference between the two groups had increased to –1.30 percentage points.
William V. Tamborlane, MD, from the department of pediatrics at Yale University, New Haven, Conn., and his coauthors wrote that metformin is the approved drug of choice for pediatric patients with type 2 diabetes, and that insulin currently is the only approved option for those who do not have an adequate response to metformin monotherapy.
“This discrepancy in available treatments for youth as compared with adults persists because of a lack of successfully completed trials needed for approval of new drugs for the treatment of type 2 diabetes in children since a trial of metformin was completed in 1999,” they wrote.
The study showed that significantly more patients in the liraglutide group (63.7%) achieved glycated hemoglobin levels below 7%, compared with 36.5% of patients in the placebo group. Fasting plasma glucose levels were decreased in the liraglutide group at both 26 and 52 weeks, but had increased in the placebo group.
Although the number of reported adverse events were similar between the two groups, there were significantly more reports of gastrointestinal adverse events – particularly nausea – in patients taking liraglutide, compared with those on placebo.
However, the study did not show a difference between liraglutide and placebo in lowering body mass index, although mean body weight decreases – which were seen in both groups – were maintained at week 52 only in the liraglutide group. The authors suggested this might be owing to the relatively small number of patients enrolled in the study and that some of the children were still growing.
Novo Nordisk, which manufactures liraglutide, supported the study. Twelve authors reported grants or support from Novo Nordisk in relation to the trial. Three authors were employees of Novo Nordisk. Eight authors reported unrelated grants and fees from Novo Nordisk and other pharmaceutical companies.
SOURCE: Tamborlane WV et al. N Engl J Med. 2019 Apr 28. doi: 10.1056/NEJMoa1903822.
The addition of liraglutide to metformin shows significantly improved glycemic control in children and adolescents with type 2 diabetes, compared with metformin alone, according to data presented at the Pediatric Academic Societies annual meeting in Baltimore.
The phase 3 study, which was simultaneously published in the New England Journal of Medicine, involved 134 patients aged 10-17 years with type 2 diabetes who were managing their diabetes with diet and exercise, metformin, or insulin.
Participants were randomized either to subcutaneous liraglutide – dose-escalated up to 1.8 mg/day, depending on efficacy and side effects – or placebo for 52 weeks. The first 26 weeks were double blind and the second 26 weeks were an open-label extension period.
At 26 weeks, mean glycated hemoglobin levels in the liraglutide group had decreased by 0.64 percentage points from baseline, but in the placebo group they had increased by 0.42 percentage points, representing a treatment difference of –1.06 percentage points (P less than .001). By week 52, the treatment difference between the two groups had increased to –1.30 percentage points.
William V. Tamborlane, MD, from the department of pediatrics at Yale University, New Haven, Conn., and his coauthors wrote that metformin is the approved drug of choice for pediatric patients with type 2 diabetes, and that insulin currently is the only approved option for those who do not have an adequate response to metformin monotherapy.
“This discrepancy in available treatments for youth as compared with adults persists because of a lack of successfully completed trials needed for approval of new drugs for the treatment of type 2 diabetes in children since a trial of metformin was completed in 1999,” they wrote.
The study showed that significantly more patients in the liraglutide group (63.7%) achieved glycated hemoglobin levels below 7%, compared with 36.5% of patients in the placebo group. Fasting plasma glucose levels were decreased in the liraglutide group at both 26 and 52 weeks, but had increased in the placebo group.
Although the number of reported adverse events were similar between the two groups, there were significantly more reports of gastrointestinal adverse events – particularly nausea – in patients taking liraglutide, compared with those on placebo.
However, the study did not show a difference between liraglutide and placebo in lowering body mass index, although mean body weight decreases – which were seen in both groups – were maintained at week 52 only in the liraglutide group. The authors suggested this might be owing to the relatively small number of patients enrolled in the study and that some of the children were still growing.
Novo Nordisk, which manufactures liraglutide, supported the study. Twelve authors reported grants or support from Novo Nordisk in relation to the trial. Three authors were employees of Novo Nordisk. Eight authors reported unrelated grants and fees from Novo Nordisk and other pharmaceutical companies.
SOURCE: Tamborlane WV et al. N Engl J Med. 2019 Apr 28. doi: 10.1056/NEJMoa1903822.
FROM PAS 2019
LentiGlobin reduces transfusion dependence in young thalassemia patients
NEW ORLEANS – The gene therapy LentiGlobin can reduce transfusion dependence in children and young adults with non-beta0/beta0 thalassemia, according to two trials.
In a phase 1/2 trial, 8 of 10 of patients achieved transfusion independence at a median follow-up of 36.0 months. In a phase 3 trial, transfusion independence was achieved by 2 of 3 patients with follow-up of at least 12 months.
Timothy S. Olson, MD, PhD, of Children’s Hospital of Philadelphia, presented results from the phase 1/2 HGB-204 trial and the phase 3 HGB-207 trial at the annual meeting of the American Society of Pediatric Hematology/Oncology.
Treatment
In both trials, patients received granulocyte colony-stimulating factor and plerixafor for hematopoietic stem cell mobilization. Their cells were collected via apheresis and transduced with the betibeglogene darolentivec (BB305) lentiviral vector. The patients received busulfan (for an average of 4 days) as conditioning and were infused with the transduced cells.
The manufacturing process for LentiGlobin was refined in the HGB-207 trial, which translated to a product with a higher vector copy number and higher proportion of CD34+ cells transduced, Dr. Olson said.
The median vector copy number was 3.1 in the HGB-207 trial and 0.7 in the HGB-204 trial. The median proportion of CD34+ cells transfused was 81% and 29%, respectively. The median cell dose was 7.7 x 106 CD34+ cells/kg and 7.1 x 106 CD34+ cells/kg, respectively.
HGB-204 patients and efficacy
The HGB-204 trial included 10 patients with non-beta0/beta0 genotypes – 6 with betaE/beta0, 1 with beta+/beta0, 2 with beta+/beta+, and 1 with an “other” genotype.
The patients’ median age at consent was 19.5 years (range, 16-34). The annualized median prestudy red blood cell (RBC) transfusion volume was 151 mL/kg per year.
At a median follow-up of 36 months, 8 of the 10 patients achieved transfusion independence. The median duration of transfusion independence was 38 months. The median weighted average hemoglobin during transfusion independence was 10.2 g/dL.
“Two patients did not achieve transfusion independence, and both patients were on the lower end of the spectrum both in terms of vector copy number per cell and the percentage of CD34+ cells that were successfully transduced,” Dr. Olson said. “Both patients actually experienced a reduction in the annualized transfusion volume requirements of between 43% and 77%.”
HGB-207 patients and efficacy
The HGB-207 trial included 16 patients with non-beta0/beta0 genotypes – 6 with betaE/beta0, 7 with beta+/beta0, and 3 with the beta+/beta+ genotype.
The patients’ median age at consent was 19 years . The annualized median prestudy RBC transfusion volume was 192 mL/kg per year.
The median follow-up in this trial is 9.3 months. Ten of 11 patients with at least 3 months of follow-up are transfusion-free with hemoglobin levels greater than 11 g/dL.
Two patients have achieved transfusion independence according to the protocol definition, which is weighted average hemoglobin of 9 g/dL or greater without any RBC transfusions for at least 12 months.
“In the one patient in this study who did not achieve transfusion independence, the vector-derived hemoglobin was quite low, and this correlated with a very low vector copy number seen in circulating peripheral blood mononuclear cells,” Dr. Olson said.
It isn’t clear why this occurred, however, as the vector copy number wasn’t especially low in the LentiGlobin product the patient received. Therefore, the researchers are still investigating why this patient failed to achieve transfusion independence.
Safety in both trials
“Very importantly, there were no deaths, there were no engraftment failures, there was no evidence of vector-mediated replication-competent lentivirus, and integration site analysis revealed no evidence of clonal dominance,” Dr. Olson said.
He added that most of the grade 3 or greater adverse events seen in both trials were directly attributable to busulfan-based myeloablative conditioning, including four episodes of veno-occlusive disease.
Nonhematologic grade 3 or higher adverse events in HGB-204 included stomatitis (n = 8), febrile neutropenia (n = 6), irregular menstruation (n = 3), pharyngeal inflammation (n = 2), and veno-occlusive liver disease (n = 1).
Nonhematologic grade 3 or higher adverse events in HGB-207 included stomatitis (n = 9), febrile neutropenia (n = 4), pharyngeal inflammation (n = 2), epistaxis (n = 3), pyrexia (n = 3), veno-occlusive liver disease (n = 3), ALT increase (n = 2), bilirubin increase (n = 2), and hypoxia (n = 2).
One patient in HGB-207 had grade 3 thrombocytopenia considered possibly related to LentiGlobin.
Dr. Olson reported advisory board engagement with bluebird bio, which sponsored both trials.
SOURCE: Olson TS et al. ASPHO 2019. Abstract 2002.
NEW ORLEANS – The gene therapy LentiGlobin can reduce transfusion dependence in children and young adults with non-beta0/beta0 thalassemia, according to two trials.
In a phase 1/2 trial, 8 of 10 of patients achieved transfusion independence at a median follow-up of 36.0 months. In a phase 3 trial, transfusion independence was achieved by 2 of 3 patients with follow-up of at least 12 months.
Timothy S. Olson, MD, PhD, of Children’s Hospital of Philadelphia, presented results from the phase 1/2 HGB-204 trial and the phase 3 HGB-207 trial at the annual meeting of the American Society of Pediatric Hematology/Oncology.
Treatment
In both trials, patients received granulocyte colony-stimulating factor and plerixafor for hematopoietic stem cell mobilization. Their cells were collected via apheresis and transduced with the betibeglogene darolentivec (BB305) lentiviral vector. The patients received busulfan (for an average of 4 days) as conditioning and were infused with the transduced cells.
The manufacturing process for LentiGlobin was refined in the HGB-207 trial, which translated to a product with a higher vector copy number and higher proportion of CD34+ cells transduced, Dr. Olson said.
The median vector copy number was 3.1 in the HGB-207 trial and 0.7 in the HGB-204 trial. The median proportion of CD34+ cells transfused was 81% and 29%, respectively. The median cell dose was 7.7 x 106 CD34+ cells/kg and 7.1 x 106 CD34+ cells/kg, respectively.
HGB-204 patients and efficacy
The HGB-204 trial included 10 patients with non-beta0/beta0 genotypes – 6 with betaE/beta0, 1 with beta+/beta0, 2 with beta+/beta+, and 1 with an “other” genotype.
The patients’ median age at consent was 19.5 years (range, 16-34). The annualized median prestudy red blood cell (RBC) transfusion volume was 151 mL/kg per year.
At a median follow-up of 36 months, 8 of the 10 patients achieved transfusion independence. The median duration of transfusion independence was 38 months. The median weighted average hemoglobin during transfusion independence was 10.2 g/dL.
“Two patients did not achieve transfusion independence, and both patients were on the lower end of the spectrum both in terms of vector copy number per cell and the percentage of CD34+ cells that were successfully transduced,” Dr. Olson said. “Both patients actually experienced a reduction in the annualized transfusion volume requirements of between 43% and 77%.”
HGB-207 patients and efficacy
The HGB-207 trial included 16 patients with non-beta0/beta0 genotypes – 6 with betaE/beta0, 7 with beta+/beta0, and 3 with the beta+/beta+ genotype.
The patients’ median age at consent was 19 years . The annualized median prestudy RBC transfusion volume was 192 mL/kg per year.
The median follow-up in this trial is 9.3 months. Ten of 11 patients with at least 3 months of follow-up are transfusion-free with hemoglobin levels greater than 11 g/dL.
Two patients have achieved transfusion independence according to the protocol definition, which is weighted average hemoglobin of 9 g/dL or greater without any RBC transfusions for at least 12 months.
“In the one patient in this study who did not achieve transfusion independence, the vector-derived hemoglobin was quite low, and this correlated with a very low vector copy number seen in circulating peripheral blood mononuclear cells,” Dr. Olson said.
It isn’t clear why this occurred, however, as the vector copy number wasn’t especially low in the LentiGlobin product the patient received. Therefore, the researchers are still investigating why this patient failed to achieve transfusion independence.
Safety in both trials
“Very importantly, there were no deaths, there were no engraftment failures, there was no evidence of vector-mediated replication-competent lentivirus, and integration site analysis revealed no evidence of clonal dominance,” Dr. Olson said.
He added that most of the grade 3 or greater adverse events seen in both trials were directly attributable to busulfan-based myeloablative conditioning, including four episodes of veno-occlusive disease.
Nonhematologic grade 3 or higher adverse events in HGB-204 included stomatitis (n = 8), febrile neutropenia (n = 6), irregular menstruation (n = 3), pharyngeal inflammation (n = 2), and veno-occlusive liver disease (n = 1).
Nonhematologic grade 3 or higher adverse events in HGB-207 included stomatitis (n = 9), febrile neutropenia (n = 4), pharyngeal inflammation (n = 2), epistaxis (n = 3), pyrexia (n = 3), veno-occlusive liver disease (n = 3), ALT increase (n = 2), bilirubin increase (n = 2), and hypoxia (n = 2).
One patient in HGB-207 had grade 3 thrombocytopenia considered possibly related to LentiGlobin.
Dr. Olson reported advisory board engagement with bluebird bio, which sponsored both trials.
SOURCE: Olson TS et al. ASPHO 2019. Abstract 2002.
NEW ORLEANS – The gene therapy LentiGlobin can reduce transfusion dependence in children and young adults with non-beta0/beta0 thalassemia, according to two trials.
In a phase 1/2 trial, 8 of 10 of patients achieved transfusion independence at a median follow-up of 36.0 months. In a phase 3 trial, transfusion independence was achieved by 2 of 3 patients with follow-up of at least 12 months.
Timothy S. Olson, MD, PhD, of Children’s Hospital of Philadelphia, presented results from the phase 1/2 HGB-204 trial and the phase 3 HGB-207 trial at the annual meeting of the American Society of Pediatric Hematology/Oncology.
Treatment
In both trials, patients received granulocyte colony-stimulating factor and plerixafor for hematopoietic stem cell mobilization. Their cells were collected via apheresis and transduced with the betibeglogene darolentivec (BB305) lentiviral vector. The patients received busulfan (for an average of 4 days) as conditioning and were infused with the transduced cells.
The manufacturing process for LentiGlobin was refined in the HGB-207 trial, which translated to a product with a higher vector copy number and higher proportion of CD34+ cells transduced, Dr. Olson said.
The median vector copy number was 3.1 in the HGB-207 trial and 0.7 in the HGB-204 trial. The median proportion of CD34+ cells transfused was 81% and 29%, respectively. The median cell dose was 7.7 x 106 CD34+ cells/kg and 7.1 x 106 CD34+ cells/kg, respectively.
HGB-204 patients and efficacy
The HGB-204 trial included 10 patients with non-beta0/beta0 genotypes – 6 with betaE/beta0, 1 with beta+/beta0, 2 with beta+/beta+, and 1 with an “other” genotype.
The patients’ median age at consent was 19.5 years (range, 16-34). The annualized median prestudy red blood cell (RBC) transfusion volume was 151 mL/kg per year.
At a median follow-up of 36 months, 8 of the 10 patients achieved transfusion independence. The median duration of transfusion independence was 38 months. The median weighted average hemoglobin during transfusion independence was 10.2 g/dL.
“Two patients did not achieve transfusion independence, and both patients were on the lower end of the spectrum both in terms of vector copy number per cell and the percentage of CD34+ cells that were successfully transduced,” Dr. Olson said. “Both patients actually experienced a reduction in the annualized transfusion volume requirements of between 43% and 77%.”
HGB-207 patients and efficacy
The HGB-207 trial included 16 patients with non-beta0/beta0 genotypes – 6 with betaE/beta0, 7 with beta+/beta0, and 3 with the beta+/beta+ genotype.
The patients’ median age at consent was 19 years . The annualized median prestudy RBC transfusion volume was 192 mL/kg per year.
The median follow-up in this trial is 9.3 months. Ten of 11 patients with at least 3 months of follow-up are transfusion-free with hemoglobin levels greater than 11 g/dL.
Two patients have achieved transfusion independence according to the protocol definition, which is weighted average hemoglobin of 9 g/dL or greater without any RBC transfusions for at least 12 months.
“In the one patient in this study who did not achieve transfusion independence, the vector-derived hemoglobin was quite low, and this correlated with a very low vector copy number seen in circulating peripheral blood mononuclear cells,” Dr. Olson said.
It isn’t clear why this occurred, however, as the vector copy number wasn’t especially low in the LentiGlobin product the patient received. Therefore, the researchers are still investigating why this patient failed to achieve transfusion independence.
Safety in both trials
“Very importantly, there were no deaths, there were no engraftment failures, there was no evidence of vector-mediated replication-competent lentivirus, and integration site analysis revealed no evidence of clonal dominance,” Dr. Olson said.
He added that most of the grade 3 or greater adverse events seen in both trials were directly attributable to busulfan-based myeloablative conditioning, including four episodes of veno-occlusive disease.
Nonhematologic grade 3 or higher adverse events in HGB-204 included stomatitis (n = 8), febrile neutropenia (n = 6), irregular menstruation (n = 3), pharyngeal inflammation (n = 2), and veno-occlusive liver disease (n = 1).
Nonhematologic grade 3 or higher adverse events in HGB-207 included stomatitis (n = 9), febrile neutropenia (n = 4), pharyngeal inflammation (n = 2), epistaxis (n = 3), pyrexia (n = 3), veno-occlusive liver disease (n = 3), ALT increase (n = 2), bilirubin increase (n = 2), and hypoxia (n = 2).
One patient in HGB-207 had grade 3 thrombocytopenia considered possibly related to LentiGlobin.
Dr. Olson reported advisory board engagement with bluebird bio, which sponsored both trials.
SOURCE: Olson TS et al. ASPHO 2019. Abstract 2002.
REPORTING FROM 2019 ASPHO CONFERENCE
A telemedicine compromise
It’s late on a Thursday afternoon. Even through the six walls that separate you from the waiting room you can feel the impatient throng of families as you struggle to see the tympanic membrane of a feverish and uncooperative 3-year-old. You already have scraped his auditory canal once with your curette. Your gut tells you that he must have an otitis but deeper in your soul there are other voices reminding you that to make the diagnosis you must visualize his ear drum. Your skill and the technology on hand has failed you.
It’s a Sunday morning, weekend hours, and you are seeing a 12-year-old with a sore throat and fever. Her physical exam suggests that she has strep pharyngitis but the team member in charge of restocking supplies has forgotten to reorder rapid strep kits and you used the last one yesterday afternoon.
Do you ignore your training and treat these sick children with antibiotics?
If you are someone who perceives the world in black and white, your response to these scenarios is simple because you NEVER prescribe antibiotics without seeing a tympanic membrane or confirming your suspicion with a rapid strep test. There are unrealistic solutions that could include requesting an immediate ear/nose/throat consult or sending the patient on an hour-long odyssey to the hospital lab. But for the rest of us who see in shades of gray, we may have to compromise our principles and temporarily become poor antibiotic stewards. The question is, how often do you compromise? Once a week, once a month, twice a year, or twice a day?
A study published in Pediatrics looks at the issue of antibiotic stewardship as it relates to telemedicine (“Antibiotic Prescribing During Pediatrics Direct-to-Consumer Telemedicine Visits,” Pediatrics. 2019 May. doi: 10.1542/peds.2018-2491).
The investigators found that children with acute respiratory infections were more likely to receive antibiotics and less likely to receive guideline concordant management at direct-to-consumer (DTC) telemedicine visits than when they were seen by their primary care physician or at an urgent care center.
In their discussion, the researchers note several possible explanations for the discrepancies they observed. DTC telemedicine visits are limited by the devices used by the families and physicians and generally lack availability of otoscopy and strep testing. The authors also wonder whether “there may be differential expectations for antibiotics among children and parents who use DTC telemedicine versus in person care.” Does this mean that families who utilize DTC telemedicine undervalue in-person care and/or are willing to compromise by accepting what they may suspect is substandard care for the convenience of DTC telemedicine?
Which brings me to my point. A physician who accepts the challenge of seeing pediatric patients with acute respiratory illnesses knowing that he or she will not be able to visualize tympanic membranes or perform strep testing also has accepted the fact that he or she will be compromising the principles of antibiotic stewardship he or she must have – or maybe should have – learned in medical school or residency.
We all occasionally compromise our principles when technology fails us or when the situations are extraordinary. But I am troubled that there some physicians who are willing to practice in an environment in which they are aware that they will be compromising their antibiotic stewardship on a daily or even hourly basis.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
It’s late on a Thursday afternoon. Even through the six walls that separate you from the waiting room you can feel the impatient throng of families as you struggle to see the tympanic membrane of a feverish and uncooperative 3-year-old. You already have scraped his auditory canal once with your curette. Your gut tells you that he must have an otitis but deeper in your soul there are other voices reminding you that to make the diagnosis you must visualize his ear drum. Your skill and the technology on hand has failed you.
It’s a Sunday morning, weekend hours, and you are seeing a 12-year-old with a sore throat and fever. Her physical exam suggests that she has strep pharyngitis but the team member in charge of restocking supplies has forgotten to reorder rapid strep kits and you used the last one yesterday afternoon.
Do you ignore your training and treat these sick children with antibiotics?
If you are someone who perceives the world in black and white, your response to these scenarios is simple because you NEVER prescribe antibiotics without seeing a tympanic membrane or confirming your suspicion with a rapid strep test. There are unrealistic solutions that could include requesting an immediate ear/nose/throat consult or sending the patient on an hour-long odyssey to the hospital lab. But for the rest of us who see in shades of gray, we may have to compromise our principles and temporarily become poor antibiotic stewards. The question is, how often do you compromise? Once a week, once a month, twice a year, or twice a day?
A study published in Pediatrics looks at the issue of antibiotic stewardship as it relates to telemedicine (“Antibiotic Prescribing During Pediatrics Direct-to-Consumer Telemedicine Visits,” Pediatrics. 2019 May. doi: 10.1542/peds.2018-2491).
The investigators found that children with acute respiratory infections were more likely to receive antibiotics and less likely to receive guideline concordant management at direct-to-consumer (DTC) telemedicine visits than when they were seen by their primary care physician or at an urgent care center.
In their discussion, the researchers note several possible explanations for the discrepancies they observed. DTC telemedicine visits are limited by the devices used by the families and physicians and generally lack availability of otoscopy and strep testing. The authors also wonder whether “there may be differential expectations for antibiotics among children and parents who use DTC telemedicine versus in person care.” Does this mean that families who utilize DTC telemedicine undervalue in-person care and/or are willing to compromise by accepting what they may suspect is substandard care for the convenience of DTC telemedicine?
Which brings me to my point. A physician who accepts the challenge of seeing pediatric patients with acute respiratory illnesses knowing that he or she will not be able to visualize tympanic membranes or perform strep testing also has accepted the fact that he or she will be compromising the principles of antibiotic stewardship he or she must have – or maybe should have – learned in medical school or residency.
We all occasionally compromise our principles when technology fails us or when the situations are extraordinary. But I am troubled that there some physicians who are willing to practice in an environment in which they are aware that they will be compromising their antibiotic stewardship on a daily or even hourly basis.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
It’s late on a Thursday afternoon. Even through the six walls that separate you from the waiting room you can feel the impatient throng of families as you struggle to see the tympanic membrane of a feverish and uncooperative 3-year-old. You already have scraped his auditory canal once with your curette. Your gut tells you that he must have an otitis but deeper in your soul there are other voices reminding you that to make the diagnosis you must visualize his ear drum. Your skill and the technology on hand has failed you.
It’s a Sunday morning, weekend hours, and you are seeing a 12-year-old with a sore throat and fever. Her physical exam suggests that she has strep pharyngitis but the team member in charge of restocking supplies has forgotten to reorder rapid strep kits and you used the last one yesterday afternoon.
Do you ignore your training and treat these sick children with antibiotics?
If you are someone who perceives the world in black and white, your response to these scenarios is simple because you NEVER prescribe antibiotics without seeing a tympanic membrane or confirming your suspicion with a rapid strep test. There are unrealistic solutions that could include requesting an immediate ear/nose/throat consult or sending the patient on an hour-long odyssey to the hospital lab. But for the rest of us who see in shades of gray, we may have to compromise our principles and temporarily become poor antibiotic stewards. The question is, how often do you compromise? Once a week, once a month, twice a year, or twice a day?
A study published in Pediatrics looks at the issue of antibiotic stewardship as it relates to telemedicine (“Antibiotic Prescribing During Pediatrics Direct-to-Consumer Telemedicine Visits,” Pediatrics. 2019 May. doi: 10.1542/peds.2018-2491).
The investigators found that children with acute respiratory infections were more likely to receive antibiotics and less likely to receive guideline concordant management at direct-to-consumer (DTC) telemedicine visits than when they were seen by their primary care physician or at an urgent care center.
In their discussion, the researchers note several possible explanations for the discrepancies they observed. DTC telemedicine visits are limited by the devices used by the families and physicians and generally lack availability of otoscopy and strep testing. The authors also wonder whether “there may be differential expectations for antibiotics among children and parents who use DTC telemedicine versus in person care.” Does this mean that families who utilize DTC telemedicine undervalue in-person care and/or are willing to compromise by accepting what they may suspect is substandard care for the convenience of DTC telemedicine?
Which brings me to my point. A physician who accepts the challenge of seeing pediatric patients with acute respiratory illnesses knowing that he or she will not be able to visualize tympanic membranes or perform strep testing also has accepted the fact that he or she will be compromising the principles of antibiotic stewardship he or she must have – or maybe should have – learned in medical school or residency.
We all occasionally compromise our principles when technology fails us or when the situations are extraordinary. But I am troubled that there some physicians who are willing to practice in an environment in which they are aware that they will be compromising their antibiotic stewardship on a daily or even hourly basis.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Increase in pediatric thyroid cancers: overdiagnosis or something more?
Thyroid cancer rates are on the rise in U.S. pediatric patients, a large epidemiologic study shows, and researchers say the trend can’t be explained away purely as overdiagnosis.
However, the extent of a real increase in clinically significant cancers and what might be causing that increase remains unclear, according to authors of the study and two related editorials appearing in the journal Cancer.
Cases of pediatric differentiated thyroid cancer (DTC) cases increased by 4.43% per year in the study, which was based on data from 39 U.S. cancer registries. Increases were seen in both smaller early-stage and larger late-stage tumors, leading study authors to assert that the trend was “unlikely to be explained solely by increased medical surveillance or improved detection.”
“Environmental and individual factors may also have affected rising trends,” said Meredith S. Shiels, PhD, of the National Cancer Institute and coauthors in a report on the study.
A true increase in pediatric thyroid cancer incidence is a possibility, authors of a related editorial said; however, they also expressed concern that inferences drawn from this U.S. cancer epidemiology data may be “artifactual.”
“We believe that first it is most important to closely examine the reasons for the increase that could be attributable to overdiagnosis, given this appears to be a likely explanation,” said authors of one editorial, including Amy Y. Chen, MD, MPH, of Emory University, Atlanta, and Louise Davies, MD, MS, of the Dartmouth Institute for Health Policy and Clinical Practice in Lebanon, N.H.
Overdiagnosis does occur, but the “real component” of the rise in thyroid cancer incidence cannot be ignored, according to a second editorial by David Goldenberg, MD, of Penn State University, Hershey.
“Although many authors are quick to explain the rise in thyroid cancer as an artifact of the overdiagnosis of clinically insignificant thyroid cancers, multiple groups all over the world have shown that this is not sufficient to explain the rise in thyroid cancer,” Dr. Goldenberg said in his editorial.
New data in pediatric patients
Pediatric DTCs are rare, representing just 2%-4% of pediatric malignancies, and they’re particularly rare in relation to adult cases, comprising about 2.3% of thyroid cancer diagnoses overall, according to Dr. Shiels and coauthors of the current study.
“The rarity of pediatric DTC, the lack of information on histologic features, or both have prevented prior studies from analyzing trends by tumor size or cancer stage at diagnosis,” they said.
To address this knowledge gap, Dr. Shiels and colleagues analyzed a total of 7,296 primary DTCs in children aged 0-19 years in data obtained from the North American Association of Central Cancer Registries. Ninety-one percent of these pediatric patients had papillary thyroid cancer, 83% were female, and 76% were between the ages of 15 to 19 years.
The rate of pediatric DTCs increased from 4.77 per million in 1998 to 8.82 per million in 2013, representing an increase of 4.43% per year, they found.
Both localized and more aggressive tumors increased in incidence over that time period, they also found. The annual increase was 4.06% for local stage at diagnosis, 5.68% for regional, and 8.55% for distant disease.
Similarly, increases were seen in small and large tumors alike. The annual percentage increase was 9.46% for tumors smaller than 1 cm, and 4.69% for tumors greater than 2 cm, according to the report.
Looking at age, investigators found that the increases in incidence were significant only for 10- to 19-year-olds, while significant increases were consistently observed for both sexes and for all races and ethnicities.
Overdiagnosis vs. environmental factors and lifestyle changes
If further investigations point to detection of subclinical disease as the cause of the increases in pediatric DTC incidence then initiatives may be needed to curtail use of CT scans, ultrasound, and needle biopsies, as has been done in adults, Dr. Chen and Dr. Davies said in their editorial.
“When the American Thyroid Association modified their guidelines for needle biopsy of nodules to discourage sampling of small lesions, a corresponding decrease in incidence rates was observed, suggesting that, indeed, overdiagnosis was the culprit,” they said.
Although it’s not clear what environmental factors or lifestyle changes are driving an increase, obesity has been consistently linked to increases in thyroid cancer, Dr. Goldenberg said. Conversely, smoking has been linked to reduced thyroid cancer risk, which means reduced prevalence of smoking in the community could potentially contribute to increased thyroid cancer incidence.
“It is our role as physicians to protect our patients from complacency and undertreatment,” he concluded in his editorial. “Explaining away thyroid cancers as being subclinical or clinically insignificant is reminiscent of days past when we told our patients: ‘Don’t worry, it’s good cancer.’ ”
The research by Dr. Shiels and colleagues was supported by the Intramural Research Program of the National Cancer Institute. Dr. Shiels and coauthors made no conflict of interest disclosures related to their report.
SOURCE: Shiels MO et al. Cancer. 2019 Apr 23. doi: 10.1002/cncr.32125.
Thyroid cancer rates are on the rise in U.S. pediatric patients, a large epidemiologic study shows, and researchers say the trend can’t be explained away purely as overdiagnosis.
However, the extent of a real increase in clinically significant cancers and what might be causing that increase remains unclear, according to authors of the study and two related editorials appearing in the journal Cancer.
Cases of pediatric differentiated thyroid cancer (DTC) cases increased by 4.43% per year in the study, which was based on data from 39 U.S. cancer registries. Increases were seen in both smaller early-stage and larger late-stage tumors, leading study authors to assert that the trend was “unlikely to be explained solely by increased medical surveillance or improved detection.”
“Environmental and individual factors may also have affected rising trends,” said Meredith S. Shiels, PhD, of the National Cancer Institute and coauthors in a report on the study.
A true increase in pediatric thyroid cancer incidence is a possibility, authors of a related editorial said; however, they also expressed concern that inferences drawn from this U.S. cancer epidemiology data may be “artifactual.”
“We believe that first it is most important to closely examine the reasons for the increase that could be attributable to overdiagnosis, given this appears to be a likely explanation,” said authors of one editorial, including Amy Y. Chen, MD, MPH, of Emory University, Atlanta, and Louise Davies, MD, MS, of the Dartmouth Institute for Health Policy and Clinical Practice in Lebanon, N.H.
Overdiagnosis does occur, but the “real component” of the rise in thyroid cancer incidence cannot be ignored, according to a second editorial by David Goldenberg, MD, of Penn State University, Hershey.
“Although many authors are quick to explain the rise in thyroid cancer as an artifact of the overdiagnosis of clinically insignificant thyroid cancers, multiple groups all over the world have shown that this is not sufficient to explain the rise in thyroid cancer,” Dr. Goldenberg said in his editorial.
New data in pediatric patients
Pediatric DTCs are rare, representing just 2%-4% of pediatric malignancies, and they’re particularly rare in relation to adult cases, comprising about 2.3% of thyroid cancer diagnoses overall, according to Dr. Shiels and coauthors of the current study.
“The rarity of pediatric DTC, the lack of information on histologic features, or both have prevented prior studies from analyzing trends by tumor size or cancer stage at diagnosis,” they said.
To address this knowledge gap, Dr. Shiels and colleagues analyzed a total of 7,296 primary DTCs in children aged 0-19 years in data obtained from the North American Association of Central Cancer Registries. Ninety-one percent of these pediatric patients had papillary thyroid cancer, 83% were female, and 76% were between the ages of 15 to 19 years.
The rate of pediatric DTCs increased from 4.77 per million in 1998 to 8.82 per million in 2013, representing an increase of 4.43% per year, they found.
Both localized and more aggressive tumors increased in incidence over that time period, they also found. The annual increase was 4.06% for local stage at diagnosis, 5.68% for regional, and 8.55% for distant disease.
Similarly, increases were seen in small and large tumors alike. The annual percentage increase was 9.46% for tumors smaller than 1 cm, and 4.69% for tumors greater than 2 cm, according to the report.
Looking at age, investigators found that the increases in incidence were significant only for 10- to 19-year-olds, while significant increases were consistently observed for both sexes and for all races and ethnicities.
Overdiagnosis vs. environmental factors and lifestyle changes
If further investigations point to detection of subclinical disease as the cause of the increases in pediatric DTC incidence then initiatives may be needed to curtail use of CT scans, ultrasound, and needle biopsies, as has been done in adults, Dr. Chen and Dr. Davies said in their editorial.
“When the American Thyroid Association modified their guidelines for needle biopsy of nodules to discourage sampling of small lesions, a corresponding decrease in incidence rates was observed, suggesting that, indeed, overdiagnosis was the culprit,” they said.
Although it’s not clear what environmental factors or lifestyle changes are driving an increase, obesity has been consistently linked to increases in thyroid cancer, Dr. Goldenberg said. Conversely, smoking has been linked to reduced thyroid cancer risk, which means reduced prevalence of smoking in the community could potentially contribute to increased thyroid cancer incidence.
“It is our role as physicians to protect our patients from complacency and undertreatment,” he concluded in his editorial. “Explaining away thyroid cancers as being subclinical or clinically insignificant is reminiscent of days past when we told our patients: ‘Don’t worry, it’s good cancer.’ ”
The research by Dr. Shiels and colleagues was supported by the Intramural Research Program of the National Cancer Institute. Dr. Shiels and coauthors made no conflict of interest disclosures related to their report.
SOURCE: Shiels MO et al. Cancer. 2019 Apr 23. doi: 10.1002/cncr.32125.
Thyroid cancer rates are on the rise in U.S. pediatric patients, a large epidemiologic study shows, and researchers say the trend can’t be explained away purely as overdiagnosis.
However, the extent of a real increase in clinically significant cancers and what might be causing that increase remains unclear, according to authors of the study and two related editorials appearing in the journal Cancer.
Cases of pediatric differentiated thyroid cancer (DTC) cases increased by 4.43% per year in the study, which was based on data from 39 U.S. cancer registries. Increases were seen in both smaller early-stage and larger late-stage tumors, leading study authors to assert that the trend was “unlikely to be explained solely by increased medical surveillance or improved detection.”
“Environmental and individual factors may also have affected rising trends,” said Meredith S. Shiels, PhD, of the National Cancer Institute and coauthors in a report on the study.
A true increase in pediatric thyroid cancer incidence is a possibility, authors of a related editorial said; however, they also expressed concern that inferences drawn from this U.S. cancer epidemiology data may be “artifactual.”
“We believe that first it is most important to closely examine the reasons for the increase that could be attributable to overdiagnosis, given this appears to be a likely explanation,” said authors of one editorial, including Amy Y. Chen, MD, MPH, of Emory University, Atlanta, and Louise Davies, MD, MS, of the Dartmouth Institute for Health Policy and Clinical Practice in Lebanon, N.H.
Overdiagnosis does occur, but the “real component” of the rise in thyroid cancer incidence cannot be ignored, according to a second editorial by David Goldenberg, MD, of Penn State University, Hershey.
“Although many authors are quick to explain the rise in thyroid cancer as an artifact of the overdiagnosis of clinically insignificant thyroid cancers, multiple groups all over the world have shown that this is not sufficient to explain the rise in thyroid cancer,” Dr. Goldenberg said in his editorial.
New data in pediatric patients
Pediatric DTCs are rare, representing just 2%-4% of pediatric malignancies, and they’re particularly rare in relation to adult cases, comprising about 2.3% of thyroid cancer diagnoses overall, according to Dr. Shiels and coauthors of the current study.
“The rarity of pediatric DTC, the lack of information on histologic features, or both have prevented prior studies from analyzing trends by tumor size or cancer stage at diagnosis,” they said.
To address this knowledge gap, Dr. Shiels and colleagues analyzed a total of 7,296 primary DTCs in children aged 0-19 years in data obtained from the North American Association of Central Cancer Registries. Ninety-one percent of these pediatric patients had papillary thyroid cancer, 83% were female, and 76% were between the ages of 15 to 19 years.
The rate of pediatric DTCs increased from 4.77 per million in 1998 to 8.82 per million in 2013, representing an increase of 4.43% per year, they found.
Both localized and more aggressive tumors increased in incidence over that time period, they also found. The annual increase was 4.06% for local stage at diagnosis, 5.68% for regional, and 8.55% for distant disease.
Similarly, increases were seen in small and large tumors alike. The annual percentage increase was 9.46% for tumors smaller than 1 cm, and 4.69% for tumors greater than 2 cm, according to the report.
Looking at age, investigators found that the increases in incidence were significant only for 10- to 19-year-olds, while significant increases were consistently observed for both sexes and for all races and ethnicities.
Overdiagnosis vs. environmental factors and lifestyle changes
If further investigations point to detection of subclinical disease as the cause of the increases in pediatric DTC incidence then initiatives may be needed to curtail use of CT scans, ultrasound, and needle biopsies, as has been done in adults, Dr. Chen and Dr. Davies said in their editorial.
“When the American Thyroid Association modified their guidelines for needle biopsy of nodules to discourage sampling of small lesions, a corresponding decrease in incidence rates was observed, suggesting that, indeed, overdiagnosis was the culprit,” they said.
Although it’s not clear what environmental factors or lifestyle changes are driving an increase, obesity has been consistently linked to increases in thyroid cancer, Dr. Goldenberg said. Conversely, smoking has been linked to reduced thyroid cancer risk, which means reduced prevalence of smoking in the community could potentially contribute to increased thyroid cancer incidence.
“It is our role as physicians to protect our patients from complacency and undertreatment,” he concluded in his editorial. “Explaining away thyroid cancers as being subclinical or clinically insignificant is reminiscent of days past when we told our patients: ‘Don’t worry, it’s good cancer.’ ”
The research by Dr. Shiels and colleagues was supported by the Intramural Research Program of the National Cancer Institute. Dr. Shiels and coauthors made no conflict of interest disclosures related to their report.
SOURCE: Shiels MO et al. Cancer. 2019 Apr 23. doi: 10.1002/cncr.32125.
FROM CANCER









