User login
No benefit to infants from e-books over board books
BALTIMORE – Despite the greater portability and seeming convenience of electronic tablets or smartphones, compared with giving them physical board books, according to preliminary findings.
James Guevara, MD, MPH, professor of pediatrics at the Children’s Hospital of Philadelphia, presented his findings at the annual meeting of Pediatric Academic Societies in a session focused on the changing nature of children’s digital media use as digital natives.
The session chair, Danielle C. Erkoboni, MD, of the University of Pennsylvania, Philadelphia, opened the session with a review of research to date regarding children’s frequency of digital use and relative benefits of independent and shared reading, both with traditional books and e-books.
Nearly all U.S. children (98%) live in homes with mobile devices, and about a third of U.S. children (35%) use those devices, according to a 2017 Common Sense Media Report. The same survey found that children aged under 2 years get an average 42 minutes a day of overall screen time, but children in low-income households get twice as much screen time as those in middle- and upper-income homes.
Research into e-books exists for preschoolers, showing that animated pictures and sounds directly matching an e-book’s story text can potentially promote language memory, but that too many interactive features or other bells and whistles can overwhelm children and contribute to poor vocabulary development and comprehension.
Researchers also have found that parent reading of e-books has greater benefits for children than children listening to an e-book’s audio narration. However, little to no research has looked into e-reading for infants and toddlers, a gap especially relevant for physicians who participate in the Reach Out and Read program.
Dr Guevara’s study enrolled 100 Medicaid-eligible children aged 5-7 months from three participating practices in a single geographic area. Only English- or Spanish-speaking parents who owned a smartphone or tablet participated.
At the children’s 6-month well visit, the clinicians gave parents information about the importance of early parent-child reading. Then parents received Dr. Seuss’s “Hop on Pop” as either an e-book download (n = 45) or a physical board book (n = 54). The parents similarly received “Barnyard Dance” at the 9-month visit and “Goodnight Moon” at the 12-month visit.
No significant differences between the two groups existed in terms of race/ethnicity, parent age, household income, education level, marital status or the total number of adults or children in the household. Maternal depression rates (based on the Edinburgh Postnatal Depression Scale), Adverse Childhood Experience scale scores, health literacy scores (Short Assessment of Health Literacy) and scores on the StimQ home cognitive environment assessment also were similar at baseline between the two groups.
The children were assessed with the StimQ Infant measure at 7-8 months and 10-11 months, and then with the StimQ Toddler and Bayley Scale of Infant and Toddler Development (BSID-III) at 13-15 months. At the final check-up, 37 parents remained in the e-book group and 43 parents remained in the board book group.
A similar proportion of parents in both groups reported reading to their children, and parents in both groups said they read an average of 5 days a week to their child.
However, parents who received the e-books had read an average 18 books at the final follow-up, compared with an average of 31 books among parents who received the board books (P = .039).
No significant differences in children’s StimQ scores or any of the BSID-III scores (cognitive, language, motor) existed between those who received e-books versus those who received board books.
The study results showed the feasibility of promoting literacy by providing e-books to parents of older infants, but doing so did not appear to confer any advantage over providing families with traditional, physical board books. Further, language development among children in both groups remained below average, albeit not statistically different from one another.
“Pediatric clinicians should exercise caution in recommending e-books to parents of young children.” Dr Guevara said. “Additional strategies beyond clinic-based literacy promotion are needed to enhance language development among poor children.”
The research was funded by the Vanguard Strong Start for Kids Program. Dr. Guevara reported no disclosures.
BALTIMORE – Despite the greater portability and seeming convenience of electronic tablets or smartphones, compared with giving them physical board books, according to preliminary findings.
James Guevara, MD, MPH, professor of pediatrics at the Children’s Hospital of Philadelphia, presented his findings at the annual meeting of Pediatric Academic Societies in a session focused on the changing nature of children’s digital media use as digital natives.
The session chair, Danielle C. Erkoboni, MD, of the University of Pennsylvania, Philadelphia, opened the session with a review of research to date regarding children’s frequency of digital use and relative benefits of independent and shared reading, both with traditional books and e-books.
Nearly all U.S. children (98%) live in homes with mobile devices, and about a third of U.S. children (35%) use those devices, according to a 2017 Common Sense Media Report. The same survey found that children aged under 2 years get an average 42 minutes a day of overall screen time, but children in low-income households get twice as much screen time as those in middle- and upper-income homes.
Research into e-books exists for preschoolers, showing that animated pictures and sounds directly matching an e-book’s story text can potentially promote language memory, but that too many interactive features or other bells and whistles can overwhelm children and contribute to poor vocabulary development and comprehension.
Researchers also have found that parent reading of e-books has greater benefits for children than children listening to an e-book’s audio narration. However, little to no research has looked into e-reading for infants and toddlers, a gap especially relevant for physicians who participate in the Reach Out and Read program.
Dr Guevara’s study enrolled 100 Medicaid-eligible children aged 5-7 months from three participating practices in a single geographic area. Only English- or Spanish-speaking parents who owned a smartphone or tablet participated.
At the children’s 6-month well visit, the clinicians gave parents information about the importance of early parent-child reading. Then parents received Dr. Seuss’s “Hop on Pop” as either an e-book download (n = 45) or a physical board book (n = 54). The parents similarly received “Barnyard Dance” at the 9-month visit and “Goodnight Moon” at the 12-month visit.
No significant differences between the two groups existed in terms of race/ethnicity, parent age, household income, education level, marital status or the total number of adults or children in the household. Maternal depression rates (based on the Edinburgh Postnatal Depression Scale), Adverse Childhood Experience scale scores, health literacy scores (Short Assessment of Health Literacy) and scores on the StimQ home cognitive environment assessment also were similar at baseline between the two groups.
The children were assessed with the StimQ Infant measure at 7-8 months and 10-11 months, and then with the StimQ Toddler and Bayley Scale of Infant and Toddler Development (BSID-III) at 13-15 months. At the final check-up, 37 parents remained in the e-book group and 43 parents remained in the board book group.
A similar proportion of parents in both groups reported reading to their children, and parents in both groups said they read an average of 5 days a week to their child.
However, parents who received the e-books had read an average 18 books at the final follow-up, compared with an average of 31 books among parents who received the board books (P = .039).
No significant differences in children’s StimQ scores or any of the BSID-III scores (cognitive, language, motor) existed between those who received e-books versus those who received board books.
The study results showed the feasibility of promoting literacy by providing e-books to parents of older infants, but doing so did not appear to confer any advantage over providing families with traditional, physical board books. Further, language development among children in both groups remained below average, albeit not statistically different from one another.
“Pediatric clinicians should exercise caution in recommending e-books to parents of young children.” Dr Guevara said. “Additional strategies beyond clinic-based literacy promotion are needed to enhance language development among poor children.”
The research was funded by the Vanguard Strong Start for Kids Program. Dr. Guevara reported no disclosures.
BALTIMORE – Despite the greater portability and seeming convenience of electronic tablets or smartphones, compared with giving them physical board books, according to preliminary findings.
James Guevara, MD, MPH, professor of pediatrics at the Children’s Hospital of Philadelphia, presented his findings at the annual meeting of Pediatric Academic Societies in a session focused on the changing nature of children’s digital media use as digital natives.
The session chair, Danielle C. Erkoboni, MD, of the University of Pennsylvania, Philadelphia, opened the session with a review of research to date regarding children’s frequency of digital use and relative benefits of independent and shared reading, both with traditional books and e-books.
Nearly all U.S. children (98%) live in homes with mobile devices, and about a third of U.S. children (35%) use those devices, according to a 2017 Common Sense Media Report. The same survey found that children aged under 2 years get an average 42 minutes a day of overall screen time, but children in low-income households get twice as much screen time as those in middle- and upper-income homes.
Research into e-books exists for preschoolers, showing that animated pictures and sounds directly matching an e-book’s story text can potentially promote language memory, but that too many interactive features or other bells and whistles can overwhelm children and contribute to poor vocabulary development and comprehension.
Researchers also have found that parent reading of e-books has greater benefits for children than children listening to an e-book’s audio narration. However, little to no research has looked into e-reading for infants and toddlers, a gap especially relevant for physicians who participate in the Reach Out and Read program.
Dr Guevara’s study enrolled 100 Medicaid-eligible children aged 5-7 months from three participating practices in a single geographic area. Only English- or Spanish-speaking parents who owned a smartphone or tablet participated.
At the children’s 6-month well visit, the clinicians gave parents information about the importance of early parent-child reading. Then parents received Dr. Seuss’s “Hop on Pop” as either an e-book download (n = 45) or a physical board book (n = 54). The parents similarly received “Barnyard Dance” at the 9-month visit and “Goodnight Moon” at the 12-month visit.
No significant differences between the two groups existed in terms of race/ethnicity, parent age, household income, education level, marital status or the total number of adults or children in the household. Maternal depression rates (based on the Edinburgh Postnatal Depression Scale), Adverse Childhood Experience scale scores, health literacy scores (Short Assessment of Health Literacy) and scores on the StimQ home cognitive environment assessment also were similar at baseline between the two groups.
The children were assessed with the StimQ Infant measure at 7-8 months and 10-11 months, and then with the StimQ Toddler and Bayley Scale of Infant and Toddler Development (BSID-III) at 13-15 months. At the final check-up, 37 parents remained in the e-book group and 43 parents remained in the board book group.
A similar proportion of parents in both groups reported reading to their children, and parents in both groups said they read an average of 5 days a week to their child.
However, parents who received the e-books had read an average 18 books at the final follow-up, compared with an average of 31 books among parents who received the board books (P = .039).
No significant differences in children’s StimQ scores or any of the BSID-III scores (cognitive, language, motor) existed between those who received e-books versus those who received board books.
The study results showed the feasibility of promoting literacy by providing e-books to parents of older infants, but doing so did not appear to confer any advantage over providing families with traditional, physical board books. Further, language development among children in both groups remained below average, albeit not statistically different from one another.
“Pediatric clinicians should exercise caution in recommending e-books to parents of young children.” Dr Guevara said. “Additional strategies beyond clinic-based literacy promotion are needed to enhance language development among poor children.”
The research was funded by the Vanguard Strong Start for Kids Program. Dr. Guevara reported no disclosures.
REPORTING FROM PAS 2019
HM19: Practice management tips for pediatric HMGs
Presenter
H. Barrett Fromme, MD, MHPE, FAAP
Session title
Sustainability Isn’t Just For The Forests: Practice management tips for long-term success in your Pediatric Hospital Medicine Group
Session summary
Dr. H. Barrett Fromme of the University of Chicago presented and facilitated a dialogue of sustainability. The audience was guided through a discussion of how efficiency and resources, workload and job demands, work-life integration and social support, and community at work can either lead to burnout or engagement within a Pediatric Hospital Medicine Group.
For each of the four topics, Dr. Fromme presented how individuals and leaders can leverage these areas to counteract burnout and promote engagement, ultimately leading to vitality within the practice group.
She closed her discussion stating that sustainability is a “process that maintains change in a balanced environment of resources, technology, and institutional change [that] are in harmony, and enhances current and future potential to meet human aspirations and needs.”
Key takeaways for HM
- Leaders can advocate with hospital leadership to optimize individual workload and job demands.
- Individuals and leaders can improve care process and clinical work flow to optimize efficiency and resources.
- Individuals and leaders can build high-functioning teams and cultivate communities of practice.
- Individuals and leaders can work together to develop goals to optimize work-life integration.
- Leaders can support values, autonomy, and growth to create an environment where individuals actively value and support their colleagues.
Dr. Kumar is a pediatric hospitalist at Cleveland Clinic Children’s and clinical assistant professor of pediatrics at Cleveland Clinic Lerner College of Medicine at Case Western Reserve University. She serves as the cochair of Pediatric Grand Rounds and is the research director for the Pediatric Hospital Medicine Fellowship at Cleveland Clinic Children’s.
Presenter
H. Barrett Fromme, MD, MHPE, FAAP
Session title
Sustainability Isn’t Just For The Forests: Practice management tips for long-term success in your Pediatric Hospital Medicine Group
Session summary
Dr. H. Barrett Fromme of the University of Chicago presented and facilitated a dialogue of sustainability. The audience was guided through a discussion of how efficiency and resources, workload and job demands, work-life integration and social support, and community at work can either lead to burnout or engagement within a Pediatric Hospital Medicine Group.
For each of the four topics, Dr. Fromme presented how individuals and leaders can leverage these areas to counteract burnout and promote engagement, ultimately leading to vitality within the practice group.
She closed her discussion stating that sustainability is a “process that maintains change in a balanced environment of resources, technology, and institutional change [that] are in harmony, and enhances current and future potential to meet human aspirations and needs.”
Key takeaways for HM
- Leaders can advocate with hospital leadership to optimize individual workload and job demands.
- Individuals and leaders can improve care process and clinical work flow to optimize efficiency and resources.
- Individuals and leaders can build high-functioning teams and cultivate communities of practice.
- Individuals and leaders can work together to develop goals to optimize work-life integration.
- Leaders can support values, autonomy, and growth to create an environment where individuals actively value and support their colleagues.
Dr. Kumar is a pediatric hospitalist at Cleveland Clinic Children’s and clinical assistant professor of pediatrics at Cleveland Clinic Lerner College of Medicine at Case Western Reserve University. She serves as the cochair of Pediatric Grand Rounds and is the research director for the Pediatric Hospital Medicine Fellowship at Cleveland Clinic Children’s.
Presenter
H. Barrett Fromme, MD, MHPE, FAAP
Session title
Sustainability Isn’t Just For The Forests: Practice management tips for long-term success in your Pediatric Hospital Medicine Group
Session summary
Dr. H. Barrett Fromme of the University of Chicago presented and facilitated a dialogue of sustainability. The audience was guided through a discussion of how efficiency and resources, workload and job demands, work-life integration and social support, and community at work can either lead to burnout or engagement within a Pediatric Hospital Medicine Group.
For each of the four topics, Dr. Fromme presented how individuals and leaders can leverage these areas to counteract burnout and promote engagement, ultimately leading to vitality within the practice group.
She closed her discussion stating that sustainability is a “process that maintains change in a balanced environment of resources, technology, and institutional change [that] are in harmony, and enhances current and future potential to meet human aspirations and needs.”
Key takeaways for HM
- Leaders can advocate with hospital leadership to optimize individual workload and job demands.
- Individuals and leaders can improve care process and clinical work flow to optimize efficiency and resources.
- Individuals and leaders can build high-functioning teams and cultivate communities of practice.
- Individuals and leaders can work together to develop goals to optimize work-life integration.
- Leaders can support values, autonomy, and growth to create an environment where individuals actively value and support their colleagues.
Dr. Kumar is a pediatric hospitalist at Cleveland Clinic Children’s and clinical assistant professor of pediatrics at Cleveland Clinic Lerner College of Medicine at Case Western Reserve University. She serves as the cochair of Pediatric Grand Rounds and is the research director for the Pediatric Hospital Medicine Fellowship at Cleveland Clinic Children’s.
Mavyret approved for children with any HCV genotype
The Food and Drug Administration has approved glecaprevir/pibrentasvir tablets (Mavyret) for treating any of six identified genotypes of hepatitis C virus in children ages 12-17 years.
The agency noted in its press announcement that, Dosing information now will be provided for patients aged 12 years and older or weighing at least 99 lbs, without cirrhosis or who have compensated cirrhosis. It is not recommended for patients with moderate cirrhosis, and it is contraindicated in patients with severe cirrhosis, as well as patients taking atazanavir and rifampin.
In clinical trials of 47 patients with genotype 1, 2, 3, or 4 HCV without cirrhosis or with only mild cirrhosis, results at 12 weeks after 8 or 16 weeks’ treatment suggested patients’ infections had been cured – 100% had no virus detected in their blood. Adverse reactions observed were consistent with those previously observed in adults during clinical trials.
The most common reactions were headache and fatigue. Hepatitis B virus reactivation has been reported in coinfected adults during or after treatment with direct-acting antivirals, and in those who were not receiving HBV antiviral treatment. Full prescribing information can be found on the FDA website, and more information about this approval can be found in the agency’s announcement.
The Food and Drug Administration has approved glecaprevir/pibrentasvir tablets (Mavyret) for treating any of six identified genotypes of hepatitis C virus in children ages 12-17 years.
The agency noted in its press announcement that, Dosing information now will be provided for patients aged 12 years and older or weighing at least 99 lbs, without cirrhosis or who have compensated cirrhosis. It is not recommended for patients with moderate cirrhosis, and it is contraindicated in patients with severe cirrhosis, as well as patients taking atazanavir and rifampin.
In clinical trials of 47 patients with genotype 1, 2, 3, or 4 HCV without cirrhosis or with only mild cirrhosis, results at 12 weeks after 8 or 16 weeks’ treatment suggested patients’ infections had been cured – 100% had no virus detected in their blood. Adverse reactions observed were consistent with those previously observed in adults during clinical trials.
The most common reactions were headache and fatigue. Hepatitis B virus reactivation has been reported in coinfected adults during or after treatment with direct-acting antivirals, and in those who were not receiving HBV antiviral treatment. Full prescribing information can be found on the FDA website, and more information about this approval can be found in the agency’s announcement.
The Food and Drug Administration has approved glecaprevir/pibrentasvir tablets (Mavyret) for treating any of six identified genotypes of hepatitis C virus in children ages 12-17 years.
The agency noted in its press announcement that, Dosing information now will be provided for patients aged 12 years and older or weighing at least 99 lbs, without cirrhosis or who have compensated cirrhosis. It is not recommended for patients with moderate cirrhosis, and it is contraindicated in patients with severe cirrhosis, as well as patients taking atazanavir and rifampin.
In clinical trials of 47 patients with genotype 1, 2, 3, or 4 HCV without cirrhosis or with only mild cirrhosis, results at 12 weeks after 8 or 16 weeks’ treatment suggested patients’ infections had been cured – 100% had no virus detected in their blood. Adverse reactions observed were consistent with those previously observed in adults during clinical trials.
The most common reactions were headache and fatigue. Hepatitis B virus reactivation has been reported in coinfected adults during or after treatment with direct-acting antivirals, and in those who were not receiving HBV antiviral treatment. Full prescribing information can be found on the FDA website, and more information about this approval can be found in the agency’s announcement.
Early childhood infections tied to psychosis risk
ORLANDO – Infections before the age of 4 are linked to the risk of nonaffective psychosis (NAP) in adulthood, according to a study presented at the annual congress of the Schizophrenia International Research Society. Researchers also found that a lower IQ seems to make the psychosis risk more likely.
It’s well-established in the literature that infections are tied to schizophrenia and that a premorbid IQ deficit is linked as well. Researchers looked to a huge data pool from the Swedish population to try to better define these risks.
“We know that there is an association between infection and schizophrenia,” said Golam Khandaker, MRCPsych, PhD, head of the inflammation and psychiatry research group at the University of Cambridge (England). “We know that there is premorbid IQ deficit in schizophrenia. So, we wanted to know, is there a sensitive period during childhood when exposure to infection is more harmful?”
Researchers analyzed data for 647,000 people in the Swedish population who were born between 1973 and 1997, and conscripted for military service through 2010. Exposure to infection was considered to be any hospitalization with any serious infection between birth and age 13. IQ measurements were taken during military conscription at the age of 18. And researchers looked for risk of nonaffective psychosis from the age of 18 on.
There was a significant increase in risk of nonaffective psychosis in adulthood among those who’d had an infection, with a hazard ratio of 1.16 (95% confidence interval, 1.08-1.24). But when researchers broke down this risk into smaller age spans, they found that only infection between birth and 1 year old (HR, 1.19; 95% CI, 1.06-1.33) and between age 2 and 4 (HR, 1.11; 95% CI, 1.02-1.22) was linked with a significantly elevated risk of NAP. Researchers also saw a link between infection and IQ.
Researchers assessed whether familial factors could be confounding this link. They looked at rates of NAP among those with an early infection and no early infection in the general population and found that it was no different statistically than among full siblings with an early infection, compared with those with no early infection. In other words, the infection-psychosis risk was the same – whether someone was a close family member or not.
Dr. Khandaker said the findings more definitively establish a link between infection and psychosis risk and suggest that the early years are when children are at their most vulnerable.
“The association between adult nonaffective psychosis with premorbid IQ and childhood infection are not explained by shared familial confounding,” he said. “So these associations could be causal.”
When they looked at the role of IQ and the link between infection and psychosis risk, researchers found an interaction: With every 1-point decrease in IQ score, there was a corresponding increased risk of NAP among those with childhood infections (odds ratio, 1.006; P = .02).
“Childhood infections,” Dr. Khandaker said, “increase psychosis risk partly by interfering with neurodevelopment, and partly by exaggerating the effects of cognitive vulnerability to psychosis.”
Dr. Khandaker disclosed no relevant financial relationships.
ORLANDO – Infections before the age of 4 are linked to the risk of nonaffective psychosis (NAP) in adulthood, according to a study presented at the annual congress of the Schizophrenia International Research Society. Researchers also found that a lower IQ seems to make the psychosis risk more likely.
It’s well-established in the literature that infections are tied to schizophrenia and that a premorbid IQ deficit is linked as well. Researchers looked to a huge data pool from the Swedish population to try to better define these risks.
“We know that there is an association between infection and schizophrenia,” said Golam Khandaker, MRCPsych, PhD, head of the inflammation and psychiatry research group at the University of Cambridge (England). “We know that there is premorbid IQ deficit in schizophrenia. So, we wanted to know, is there a sensitive period during childhood when exposure to infection is more harmful?”
Researchers analyzed data for 647,000 people in the Swedish population who were born between 1973 and 1997, and conscripted for military service through 2010. Exposure to infection was considered to be any hospitalization with any serious infection between birth and age 13. IQ measurements were taken during military conscription at the age of 18. And researchers looked for risk of nonaffective psychosis from the age of 18 on.
There was a significant increase in risk of nonaffective psychosis in adulthood among those who’d had an infection, with a hazard ratio of 1.16 (95% confidence interval, 1.08-1.24). But when researchers broke down this risk into smaller age spans, they found that only infection between birth and 1 year old (HR, 1.19; 95% CI, 1.06-1.33) and between age 2 and 4 (HR, 1.11; 95% CI, 1.02-1.22) was linked with a significantly elevated risk of NAP. Researchers also saw a link between infection and IQ.
Researchers assessed whether familial factors could be confounding this link. They looked at rates of NAP among those with an early infection and no early infection in the general population and found that it was no different statistically than among full siblings with an early infection, compared with those with no early infection. In other words, the infection-psychosis risk was the same – whether someone was a close family member or not.
Dr. Khandaker said the findings more definitively establish a link between infection and psychosis risk and suggest that the early years are when children are at their most vulnerable.
“The association between adult nonaffective psychosis with premorbid IQ and childhood infection are not explained by shared familial confounding,” he said. “So these associations could be causal.”
When they looked at the role of IQ and the link between infection and psychosis risk, researchers found an interaction: With every 1-point decrease in IQ score, there was a corresponding increased risk of NAP among those with childhood infections (odds ratio, 1.006; P = .02).
“Childhood infections,” Dr. Khandaker said, “increase psychosis risk partly by interfering with neurodevelopment, and partly by exaggerating the effects of cognitive vulnerability to psychosis.”
Dr. Khandaker disclosed no relevant financial relationships.
ORLANDO – Infections before the age of 4 are linked to the risk of nonaffective psychosis (NAP) in adulthood, according to a study presented at the annual congress of the Schizophrenia International Research Society. Researchers also found that a lower IQ seems to make the psychosis risk more likely.
It’s well-established in the literature that infections are tied to schizophrenia and that a premorbid IQ deficit is linked as well. Researchers looked to a huge data pool from the Swedish population to try to better define these risks.
“We know that there is an association between infection and schizophrenia,” said Golam Khandaker, MRCPsych, PhD, head of the inflammation and psychiatry research group at the University of Cambridge (England). “We know that there is premorbid IQ deficit in schizophrenia. So, we wanted to know, is there a sensitive period during childhood when exposure to infection is more harmful?”
Researchers analyzed data for 647,000 people in the Swedish population who were born between 1973 and 1997, and conscripted for military service through 2010. Exposure to infection was considered to be any hospitalization with any serious infection between birth and age 13. IQ measurements were taken during military conscription at the age of 18. And researchers looked for risk of nonaffective psychosis from the age of 18 on.
There was a significant increase in risk of nonaffective psychosis in adulthood among those who’d had an infection, with a hazard ratio of 1.16 (95% confidence interval, 1.08-1.24). But when researchers broke down this risk into smaller age spans, they found that only infection between birth and 1 year old (HR, 1.19; 95% CI, 1.06-1.33) and between age 2 and 4 (HR, 1.11; 95% CI, 1.02-1.22) was linked with a significantly elevated risk of NAP. Researchers also saw a link between infection and IQ.
Researchers assessed whether familial factors could be confounding this link. They looked at rates of NAP among those with an early infection and no early infection in the general population and found that it was no different statistically than among full siblings with an early infection, compared with those with no early infection. In other words, the infection-psychosis risk was the same – whether someone was a close family member or not.
Dr. Khandaker said the findings more definitively establish a link between infection and psychosis risk and suggest that the early years are when children are at their most vulnerable.
“The association between adult nonaffective psychosis with premorbid IQ and childhood infection are not explained by shared familial confounding,” he said. “So these associations could be causal.”
When they looked at the role of IQ and the link between infection and psychosis risk, researchers found an interaction: With every 1-point decrease in IQ score, there was a corresponding increased risk of NAP among those with childhood infections (odds ratio, 1.006; P = .02).
“Childhood infections,” Dr. Khandaker said, “increase psychosis risk partly by interfering with neurodevelopment, and partly by exaggerating the effects of cognitive vulnerability to psychosis.”
Dr. Khandaker disclosed no relevant financial relationships.
REPORTING FROM SIRS 2019
Benlysta approved for children with SLE
The B-lymphocyte stimulator–inhibitor called Benlysta already is approved for use in adults alongside standard therapy for SLE, and this approval marks the first such treatment available for children. Although there are regulatory submissions for use of this drug among children elsewhere, the United States is the first to approve its use among this age group, according to a press release from GSK. According to an FDA press announcement, the agency expedited the review and approval because belimumab could fulfill an unmet need.
The approval is based on a 1-year postapproval commitment study, which assessed efficacy, safety, and pharmacokinetics of 10 mg/kg belimumab plus standard therapy versus placebo plus standard therapy among children with SLE aged 5-11 years (n = 13) and those aged 12-17 years (n = 80). Although the study was not fully powered because of the rarity of the disease in this age group, it did find numerically higher SLE responder index response rates over 1 year among children treated with belimumab plus standard therapy (53%) than in those treated with placebo and standard therapy (44%).
Adverse reactions seen among this age group were consistent with those seen in adults, including nausea, diarrhea, pyrexia, nasopharyngitis, and bronchitis. The most common serious adverse reactions were serious infections. Belimumab has not been studied in combination with certain other drugs, such as other biologics or cyclophosphamide; therefore, combining it with such treatments is not recommended. Acute hypersensitivity reactions – including anaphylaxis and death – have been observed, even among patients who had previously tolerated belimumab.
Infusion reactions were common, so pretreat patients with an antihistamine. Also, do not administer the drug with live vaccines, the FDA noted.
More information can be found in the press announcement on the FDA website.
The B-lymphocyte stimulator–inhibitor called Benlysta already is approved for use in adults alongside standard therapy for SLE, and this approval marks the first such treatment available for children. Although there are regulatory submissions for use of this drug among children elsewhere, the United States is the first to approve its use among this age group, according to a press release from GSK. According to an FDA press announcement, the agency expedited the review and approval because belimumab could fulfill an unmet need.
The approval is based on a 1-year postapproval commitment study, which assessed efficacy, safety, and pharmacokinetics of 10 mg/kg belimumab plus standard therapy versus placebo plus standard therapy among children with SLE aged 5-11 years (n = 13) and those aged 12-17 years (n = 80). Although the study was not fully powered because of the rarity of the disease in this age group, it did find numerically higher SLE responder index response rates over 1 year among children treated with belimumab plus standard therapy (53%) than in those treated with placebo and standard therapy (44%).
Adverse reactions seen among this age group were consistent with those seen in adults, including nausea, diarrhea, pyrexia, nasopharyngitis, and bronchitis. The most common serious adverse reactions were serious infections. Belimumab has not been studied in combination with certain other drugs, such as other biologics or cyclophosphamide; therefore, combining it with such treatments is not recommended. Acute hypersensitivity reactions – including anaphylaxis and death – have been observed, even among patients who had previously tolerated belimumab.
Infusion reactions were common, so pretreat patients with an antihistamine. Also, do not administer the drug with live vaccines, the FDA noted.
More information can be found in the press announcement on the FDA website.
The B-lymphocyte stimulator–inhibitor called Benlysta already is approved for use in adults alongside standard therapy for SLE, and this approval marks the first such treatment available for children. Although there are regulatory submissions for use of this drug among children elsewhere, the United States is the first to approve its use among this age group, according to a press release from GSK. According to an FDA press announcement, the agency expedited the review and approval because belimumab could fulfill an unmet need.
The approval is based on a 1-year postapproval commitment study, which assessed efficacy, safety, and pharmacokinetics of 10 mg/kg belimumab plus standard therapy versus placebo plus standard therapy among children with SLE aged 5-11 years (n = 13) and those aged 12-17 years (n = 80). Although the study was not fully powered because of the rarity of the disease in this age group, it did find numerically higher SLE responder index response rates over 1 year among children treated with belimumab plus standard therapy (53%) than in those treated with placebo and standard therapy (44%).
Adverse reactions seen among this age group were consistent with those seen in adults, including nausea, diarrhea, pyrexia, nasopharyngitis, and bronchitis. The most common serious adverse reactions were serious infections. Belimumab has not been studied in combination with certain other drugs, such as other biologics or cyclophosphamide; therefore, combining it with such treatments is not recommended. Acute hypersensitivity reactions – including anaphylaxis and death – have been observed, even among patients who had previously tolerated belimumab.
Infusion reactions were common, so pretreat patients with an antihistamine. Also, do not administer the drug with live vaccines, the FDA noted.
More information can be found in the press announcement on the FDA website.
Most measles cases in 25 years prompts government pleas to vaccinate
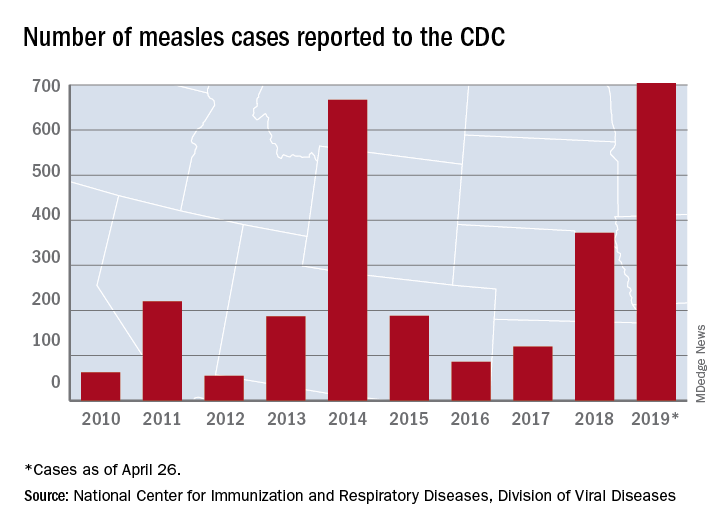
The updated figure adds 9 cases to the previous tally of 695 cases as of April 24, when the CDC announced that the number of cases in 2019 had surpassed the total for any year since the disease was considered effectively eliminated from the country in 2000.
Cases have been reported in 22 states, with the largest outbreaks in Washington and New York. The outbreak in Washington, which included 72 cases, was declared over last week. Two outbreaks in New York, however, are the largest and longest-lasting measles outbreaks since the disease was considered eliminated, said Nancy Messonnier, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases. The longer they continue, the “greater the chance that measles will again gain a foothold in the United States,” she said at CDC telebriefing on measles.
The outbreaks are linked to travelers who are exposed to measles abroad and bring it to the United States. The disease then may spread, especially in communities with high rates of unvaccinated people. “A significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine,” according to the CDC.
National Infant Immunization Week
Until last week, 2014 – with 667 measles cases – had been the year with the most cases since the disease was effectively eliminated. The last time the United States had more measles cases was in 1994, when there were 963 cases for the year.
Health and Human Services Secretary Alex Azar, also at the telebriefing, pointed out that 1994 also was the year that the United States first observed National Infant Immunization Week, which is April 27–May 4 this year. The CDC is marking the 25th anniversary of the annual observance, which highlights “the importance of protecting infants from vaccine-preventable diseases” and celebrates “the achievements of immunization programs in promoting healthy communities,” Secretary Azar said.
Message to health care providers
CDC director Robert Redfield Jr., MD, noted that measles has “no treatment, no cure, and no way to predict how bad a case will be.”
Some patients may have mild symptoms, whereas others may have serious complications such as pneumonia or encephalitis. In 2019, 3% of the patients with measles have developed pneumonia, he said. No patients have died.
Dr. Redfield, a virologist, noted that the CDC is recommending that children aged 6-12 months receive 1 dose of the measles vaccine if traveling abroad.
“As CDC director and as a physician, I have and continue to wholeheartedly advocate for infant immunization,” he said in a statement. “More importantly, as a father and grandfather I have ensured all of my children and grandchildren are vaccinated on the recommended schedule. Vaccines are safe. Vaccines do not cause autism. Vaccine-preventable diseases are dangerous.”
More than 94% of parents vaccinate their children, Dr. Redfield added. “CDC is working to reach the small percentage of vaccine-hesitant individuals so they too understand the importance of vaccines. It is imperative that we correct misinformation and reassure fearful parents so they protect their children from illnesses with long-lasting health impacts.”
About 1.3%, or 100,000 children, in the United States under 2 years old have not been vaccinated, he said.
“I call upon health care providers to encourage parents and expectant parents to vaccinate their children for their own protection and to avoid the spread of vaccine-preventable diseases within their families and communities,” he said. “We must join together as a nation to once again eliminate measles and prevent future disease outbreaks.”
The CDC has a complete list of clinical recommendations for health care providers on its website.
The president weighs in
President Donald Trump said that children should receive vaccinations – his first public comment about vaccines since his inauguration. Previously, he had questioned the safety of vaccines.
Asked by reporters about the measles outbreaks and his message for parents about having their kids vaccinated, he said: “They have to get the shot. The vaccinations are so important. This is really going around now. They have to get their shots.”

The updated figure adds 9 cases to the previous tally of 695 cases as of April 24, when the CDC announced that the number of cases in 2019 had surpassed the total for any year since the disease was considered effectively eliminated from the country in 2000.
Cases have been reported in 22 states, with the largest outbreaks in Washington and New York. The outbreak in Washington, which included 72 cases, was declared over last week. Two outbreaks in New York, however, are the largest and longest-lasting measles outbreaks since the disease was considered eliminated, said Nancy Messonnier, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases. The longer they continue, the “greater the chance that measles will again gain a foothold in the United States,” she said at CDC telebriefing on measles.
The outbreaks are linked to travelers who are exposed to measles abroad and bring it to the United States. The disease then may spread, especially in communities with high rates of unvaccinated people. “A significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine,” according to the CDC.
National Infant Immunization Week
Until last week, 2014 – with 667 measles cases – had been the year with the most cases since the disease was effectively eliminated. The last time the United States had more measles cases was in 1994, when there were 963 cases for the year.
Health and Human Services Secretary Alex Azar, also at the telebriefing, pointed out that 1994 also was the year that the United States first observed National Infant Immunization Week, which is April 27–May 4 this year. The CDC is marking the 25th anniversary of the annual observance, which highlights “the importance of protecting infants from vaccine-preventable diseases” and celebrates “the achievements of immunization programs in promoting healthy communities,” Secretary Azar said.
Message to health care providers
CDC director Robert Redfield Jr., MD, noted that measles has “no treatment, no cure, and no way to predict how bad a case will be.”
Some patients may have mild symptoms, whereas others may have serious complications such as pneumonia or encephalitis. In 2019, 3% of the patients with measles have developed pneumonia, he said. No patients have died.
Dr. Redfield, a virologist, noted that the CDC is recommending that children aged 6-12 months receive 1 dose of the measles vaccine if traveling abroad.
“As CDC director and as a physician, I have and continue to wholeheartedly advocate for infant immunization,” he said in a statement. “More importantly, as a father and grandfather I have ensured all of my children and grandchildren are vaccinated on the recommended schedule. Vaccines are safe. Vaccines do not cause autism. Vaccine-preventable diseases are dangerous.”
More than 94% of parents vaccinate their children, Dr. Redfield added. “CDC is working to reach the small percentage of vaccine-hesitant individuals so they too understand the importance of vaccines. It is imperative that we correct misinformation and reassure fearful parents so they protect their children from illnesses with long-lasting health impacts.”
About 1.3%, or 100,000 children, in the United States under 2 years old have not been vaccinated, he said.
“I call upon health care providers to encourage parents and expectant parents to vaccinate their children for their own protection and to avoid the spread of vaccine-preventable diseases within their families and communities,” he said. “We must join together as a nation to once again eliminate measles and prevent future disease outbreaks.”
The CDC has a complete list of clinical recommendations for health care providers on its website.
The president weighs in
President Donald Trump said that children should receive vaccinations – his first public comment about vaccines since his inauguration. Previously, he had questioned the safety of vaccines.
Asked by reporters about the measles outbreaks and his message for parents about having their kids vaccinated, he said: “They have to get the shot. The vaccinations are so important. This is really going around now. They have to get their shots.”

The updated figure adds 9 cases to the previous tally of 695 cases as of April 24, when the CDC announced that the number of cases in 2019 had surpassed the total for any year since the disease was considered effectively eliminated from the country in 2000.
Cases have been reported in 22 states, with the largest outbreaks in Washington and New York. The outbreak in Washington, which included 72 cases, was declared over last week. Two outbreaks in New York, however, are the largest and longest-lasting measles outbreaks since the disease was considered eliminated, said Nancy Messonnier, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases. The longer they continue, the “greater the chance that measles will again gain a foothold in the United States,” she said at CDC telebriefing on measles.
The outbreaks are linked to travelers who are exposed to measles abroad and bring it to the United States. The disease then may spread, especially in communities with high rates of unvaccinated people. “A significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine,” according to the CDC.
National Infant Immunization Week
Until last week, 2014 – with 667 measles cases – had been the year with the most cases since the disease was effectively eliminated. The last time the United States had more measles cases was in 1994, when there were 963 cases for the year.
Health and Human Services Secretary Alex Azar, also at the telebriefing, pointed out that 1994 also was the year that the United States first observed National Infant Immunization Week, which is April 27–May 4 this year. The CDC is marking the 25th anniversary of the annual observance, which highlights “the importance of protecting infants from vaccine-preventable diseases” and celebrates “the achievements of immunization programs in promoting healthy communities,” Secretary Azar said.
Message to health care providers
CDC director Robert Redfield Jr., MD, noted that measles has “no treatment, no cure, and no way to predict how bad a case will be.”
Some patients may have mild symptoms, whereas others may have serious complications such as pneumonia or encephalitis. In 2019, 3% of the patients with measles have developed pneumonia, he said. No patients have died.
Dr. Redfield, a virologist, noted that the CDC is recommending that children aged 6-12 months receive 1 dose of the measles vaccine if traveling abroad.
“As CDC director and as a physician, I have and continue to wholeheartedly advocate for infant immunization,” he said in a statement. “More importantly, as a father and grandfather I have ensured all of my children and grandchildren are vaccinated on the recommended schedule. Vaccines are safe. Vaccines do not cause autism. Vaccine-preventable diseases are dangerous.”
More than 94% of parents vaccinate their children, Dr. Redfield added. “CDC is working to reach the small percentage of vaccine-hesitant individuals so they too understand the importance of vaccines. It is imperative that we correct misinformation and reassure fearful parents so they protect their children from illnesses with long-lasting health impacts.”
About 1.3%, or 100,000 children, in the United States under 2 years old have not been vaccinated, he said.
“I call upon health care providers to encourage parents and expectant parents to vaccinate their children for their own protection and to avoid the spread of vaccine-preventable diseases within their families and communities,” he said. “We must join together as a nation to once again eliminate measles and prevent future disease outbreaks.”
The CDC has a complete list of clinical recommendations for health care providers on its website.
The president weighs in
President Donald Trump said that children should receive vaccinations – his first public comment about vaccines since his inauguration. Previously, he had questioned the safety of vaccines.
Asked by reporters about the measles outbreaks and his message for parents about having their kids vaccinated, he said: “They have to get the shot. The vaccinations are so important. This is really going around now. They have to get their shots.”
FROM A CDC TELEBRIEFING
Combo respiratory pathogen tests miss pertussis
BALTIMORE – Ann Arbor.
Respiratory pathogen panels are popular because they test for many things at once, but providers have to know their limits, said lead investigator Colleen Mayhew, MD, a pediatric emergency medicine fellow at the University of Michigan.
“Should RPAN be used to diagnosis pertussis? No,” she said at the Pediatric Academic Societies annual meeting. RPAN was negative for confirmed pertussis 44% of the time in the study.
“In our cohort, [it] was no better than a coin flip for detecting pertussis,” she said. Also, even when it missed pertussis, it still detected other pathogens, which raises the risk that symptoms might be attributed to a different infection. “This has serious public health implications.”
“The bottom line is, if you are concerned about pertussis, it’s important to use a dedicated pertussis PCR [polymerase chain reaction] assay, and to use comprehensive respiratory pathogen testing only if there are other, specific targets that will change your clinical management,” such as mycoplasma or the flu, Dr. Mayhew said.
In the study, 102 nasopharyngeal swabs positive for pertussis on standalone PCR testing – the university uses an assay from Focus Diagnostics – were thawed and tested with RPAN.
RPAN was negative for pertussis on 45 swabs (44%). “These are the potential missed pertussis cases if RPAN is used alone,” Dr. Mayhew said. RPAN detected other pathogens, such as coronavirus, about half the time, whether or not it tested positive for pertussis. “Those additional pathogens might represent coinfection, but might also represent asymptomatic carriage.” It’s impossible to differentiate between the two, she noted.
In short, “neither positive testing for other respiratory pathogens, nor negative testing for pertussis by RPAN, is reliable for excluding the diagnosis of pertussis. Dedicated pertussis PCR testing should be used for diagnosis,” she and her team concluded.
RPAN also is a PCR test, but with a different, perhaps less robust, genetic target.
The 102 positive swabs were from patients aged 1 month to 73 years, so “it’s important for all of us to keep pertussis on our differential diagnose” no matter how old patients are, Dr. Mayhew said.
Freezing and thawing the swabs shouldn’t have degraded the genetic material, but it might have; that was one of the limits of the study.
The team hopes to run a quality improvement project to encourage the use of standalone pertussis PCR in Ann Arbor.
There was no industry funding. Dr. Mayhew didn’t report any disclosures.
BALTIMORE – Ann Arbor.
Respiratory pathogen panels are popular because they test for many things at once, but providers have to know their limits, said lead investigator Colleen Mayhew, MD, a pediatric emergency medicine fellow at the University of Michigan.
“Should RPAN be used to diagnosis pertussis? No,” she said at the Pediatric Academic Societies annual meeting. RPAN was negative for confirmed pertussis 44% of the time in the study.
“In our cohort, [it] was no better than a coin flip for detecting pertussis,” she said. Also, even when it missed pertussis, it still detected other pathogens, which raises the risk that symptoms might be attributed to a different infection. “This has serious public health implications.”
“The bottom line is, if you are concerned about pertussis, it’s important to use a dedicated pertussis PCR [polymerase chain reaction] assay, and to use comprehensive respiratory pathogen testing only if there are other, specific targets that will change your clinical management,” such as mycoplasma or the flu, Dr. Mayhew said.
In the study, 102 nasopharyngeal swabs positive for pertussis on standalone PCR testing – the university uses an assay from Focus Diagnostics – were thawed and tested with RPAN.
RPAN was negative for pertussis on 45 swabs (44%). “These are the potential missed pertussis cases if RPAN is used alone,” Dr. Mayhew said. RPAN detected other pathogens, such as coronavirus, about half the time, whether or not it tested positive for pertussis. “Those additional pathogens might represent coinfection, but might also represent asymptomatic carriage.” It’s impossible to differentiate between the two, she noted.
In short, “neither positive testing for other respiratory pathogens, nor negative testing for pertussis by RPAN, is reliable for excluding the diagnosis of pertussis. Dedicated pertussis PCR testing should be used for diagnosis,” she and her team concluded.
RPAN also is a PCR test, but with a different, perhaps less robust, genetic target.
The 102 positive swabs were from patients aged 1 month to 73 years, so “it’s important for all of us to keep pertussis on our differential diagnose” no matter how old patients are, Dr. Mayhew said.
Freezing and thawing the swabs shouldn’t have degraded the genetic material, but it might have; that was one of the limits of the study.
The team hopes to run a quality improvement project to encourage the use of standalone pertussis PCR in Ann Arbor.
There was no industry funding. Dr. Mayhew didn’t report any disclosures.
BALTIMORE – Ann Arbor.
Respiratory pathogen panels are popular because they test for many things at once, but providers have to know their limits, said lead investigator Colleen Mayhew, MD, a pediatric emergency medicine fellow at the University of Michigan.
“Should RPAN be used to diagnosis pertussis? No,” she said at the Pediatric Academic Societies annual meeting. RPAN was negative for confirmed pertussis 44% of the time in the study.
“In our cohort, [it] was no better than a coin flip for detecting pertussis,” she said. Also, even when it missed pertussis, it still detected other pathogens, which raises the risk that symptoms might be attributed to a different infection. “This has serious public health implications.”
“The bottom line is, if you are concerned about pertussis, it’s important to use a dedicated pertussis PCR [polymerase chain reaction] assay, and to use comprehensive respiratory pathogen testing only if there are other, specific targets that will change your clinical management,” such as mycoplasma or the flu, Dr. Mayhew said.
In the study, 102 nasopharyngeal swabs positive for pertussis on standalone PCR testing – the university uses an assay from Focus Diagnostics – were thawed and tested with RPAN.
RPAN was negative for pertussis on 45 swabs (44%). “These are the potential missed pertussis cases if RPAN is used alone,” Dr. Mayhew said. RPAN detected other pathogens, such as coronavirus, about half the time, whether or not it tested positive for pertussis. “Those additional pathogens might represent coinfection, but might also represent asymptomatic carriage.” It’s impossible to differentiate between the two, she noted.
In short, “neither positive testing for other respiratory pathogens, nor negative testing for pertussis by RPAN, is reliable for excluding the diagnosis of pertussis. Dedicated pertussis PCR testing should be used for diagnosis,” she and her team concluded.
RPAN also is a PCR test, but with a different, perhaps less robust, genetic target.
The 102 positive swabs were from patients aged 1 month to 73 years, so “it’s important for all of us to keep pertussis on our differential diagnose” no matter how old patients are, Dr. Mayhew said.
Freezing and thawing the swabs shouldn’t have degraded the genetic material, but it might have; that was one of the limits of the study.
The team hopes to run a quality improvement project to encourage the use of standalone pertussis PCR in Ann Arbor.
There was no industry funding. Dr. Mayhew didn’t report any disclosures.
REPORTING FROM PAS 2019
Neurodevelopmental concerns may emerge later in Zika-exposed infants
BALTIMORE – Most infants prenatally exposed to Zika showed relatively normal neurodevelopment if their fetal MRI and birth head circumference were normal, but others with similarly initial normal measures appeared to struggle with social cognition and mobility as they got older, according to a new study.
“I think we need to be cautious with saying that these children are normal when these normal-appearing children may not be doing as well as we think,” lead author Sarah Mulkey, MD, of Children’s National Health System and George Washington University, Washington, said in an interview. “While most children are showing fairly normal development, there are some children who are … becoming more abnormal over time.”
Dr. Mulkey shared her findings at the Pediatric Academic Societies annual meeting. She and her colleagues had previously published a prospective study of 82 Zika-exposed infants’ fetal brain MRIs. In their new study, they followed up with the 78 Colombian infants from that study whose fetal neuroimaging and birth head circumstance had been normal.
The researchers used the Alberta Infant Motor Scale (AIMS) and the Warner Initial Developmental Evaluation of Adaptive and Functional Skills (WIDEA) to evaluate 72 of the children, 34 of whom underwent assessment twice. Forty of the children were an average 5.7 months old when evaluated, and 66 were an average 13.5 months old.
As the children got older, their overall WIDEA z-score and their subscores in the social cognition domain and especially in the mobility domain trended downward. Three of the children had AIMS scores two standard deviations below normal, but the rest fell within the normal range.
Their WIDEA communication z-score hovered relatively close to the norm, but self-care also showed a very slight slope downward, albeit not as substantially as in the social cognition and mobility domains.
The younger a child is, the fewer skills they generally show related to neurocognitive development, Dr. Mulkey explained. But as they grow older and are expected to show more skills, it becomes more apparent where gaps and delays might exist.
“We can see that there are a lot of kids doing well, but some of these kids certainly are not,” she said. “Until children have a long time to develop, you really can’t see these changes unless you follow them long-term.”
The researchers also looked separately at a subgroup of 19 children (26%) whose cranial ultrasounds showed mild nonspecific findings. These findings – such as lenticulostriate vasculopathy, choroid plexus cysts, subependymal cysts and calcifications – do not usually indicate any problems, but they appeared in a quarter of this population, considerably more than the approximately 5% typically seen in the general population, Dr. Mulkey said.
Though the findings did not reach significance, infants in this subgroup tended to have a lower WIDEA mobility z-scores (P = .054) and lower AIMS scores (P = .26) than the Zika-exposed infants with normal cranial ultrasounds.
“Mild nonspecific cranial ultrasound findings may represent a mild injury” related to exposure to their mother’s Zika infection during pregnancy, the researchers suggested. “It may be a risk factor for the lower mobility outcome,” Dr. Mulkey said.
The researchers hope to continue later follow-ups as the children age.
The research was funded by the Thrasher Research Fund. Dr. Mulkey had no conflicts of interest.
BALTIMORE – Most infants prenatally exposed to Zika showed relatively normal neurodevelopment if their fetal MRI and birth head circumference were normal, but others with similarly initial normal measures appeared to struggle with social cognition and mobility as they got older, according to a new study.
“I think we need to be cautious with saying that these children are normal when these normal-appearing children may not be doing as well as we think,” lead author Sarah Mulkey, MD, of Children’s National Health System and George Washington University, Washington, said in an interview. “While most children are showing fairly normal development, there are some children who are … becoming more abnormal over time.”
Dr. Mulkey shared her findings at the Pediatric Academic Societies annual meeting. She and her colleagues had previously published a prospective study of 82 Zika-exposed infants’ fetal brain MRIs. In their new study, they followed up with the 78 Colombian infants from that study whose fetal neuroimaging and birth head circumstance had been normal.
The researchers used the Alberta Infant Motor Scale (AIMS) and the Warner Initial Developmental Evaluation of Adaptive and Functional Skills (WIDEA) to evaluate 72 of the children, 34 of whom underwent assessment twice. Forty of the children were an average 5.7 months old when evaluated, and 66 were an average 13.5 months old.
As the children got older, their overall WIDEA z-score and their subscores in the social cognition domain and especially in the mobility domain trended downward. Three of the children had AIMS scores two standard deviations below normal, but the rest fell within the normal range.
Their WIDEA communication z-score hovered relatively close to the norm, but self-care also showed a very slight slope downward, albeit not as substantially as in the social cognition and mobility domains.
The younger a child is, the fewer skills they generally show related to neurocognitive development, Dr. Mulkey explained. But as they grow older and are expected to show more skills, it becomes more apparent where gaps and delays might exist.
“We can see that there are a lot of kids doing well, but some of these kids certainly are not,” she said. “Until children have a long time to develop, you really can’t see these changes unless you follow them long-term.”
The researchers also looked separately at a subgroup of 19 children (26%) whose cranial ultrasounds showed mild nonspecific findings. These findings – such as lenticulostriate vasculopathy, choroid plexus cysts, subependymal cysts and calcifications – do not usually indicate any problems, but they appeared in a quarter of this population, considerably more than the approximately 5% typically seen in the general population, Dr. Mulkey said.
Though the findings did not reach significance, infants in this subgroup tended to have a lower WIDEA mobility z-scores (P = .054) and lower AIMS scores (P = .26) than the Zika-exposed infants with normal cranial ultrasounds.
“Mild nonspecific cranial ultrasound findings may represent a mild injury” related to exposure to their mother’s Zika infection during pregnancy, the researchers suggested. “It may be a risk factor for the lower mobility outcome,” Dr. Mulkey said.
The researchers hope to continue later follow-ups as the children age.
The research was funded by the Thrasher Research Fund. Dr. Mulkey had no conflicts of interest.
BALTIMORE – Most infants prenatally exposed to Zika showed relatively normal neurodevelopment if their fetal MRI and birth head circumference were normal, but others with similarly initial normal measures appeared to struggle with social cognition and mobility as they got older, according to a new study.
“I think we need to be cautious with saying that these children are normal when these normal-appearing children may not be doing as well as we think,” lead author Sarah Mulkey, MD, of Children’s National Health System and George Washington University, Washington, said in an interview. “While most children are showing fairly normal development, there are some children who are … becoming more abnormal over time.”
Dr. Mulkey shared her findings at the Pediatric Academic Societies annual meeting. She and her colleagues had previously published a prospective study of 82 Zika-exposed infants’ fetal brain MRIs. In their new study, they followed up with the 78 Colombian infants from that study whose fetal neuroimaging and birth head circumstance had been normal.
The researchers used the Alberta Infant Motor Scale (AIMS) and the Warner Initial Developmental Evaluation of Adaptive and Functional Skills (WIDEA) to evaluate 72 of the children, 34 of whom underwent assessment twice. Forty of the children were an average 5.7 months old when evaluated, and 66 were an average 13.5 months old.
As the children got older, their overall WIDEA z-score and their subscores in the social cognition domain and especially in the mobility domain trended downward. Three of the children had AIMS scores two standard deviations below normal, but the rest fell within the normal range.
Their WIDEA communication z-score hovered relatively close to the norm, but self-care also showed a very slight slope downward, albeit not as substantially as in the social cognition and mobility domains.
The younger a child is, the fewer skills they generally show related to neurocognitive development, Dr. Mulkey explained. But as they grow older and are expected to show more skills, it becomes more apparent where gaps and delays might exist.
“We can see that there are a lot of kids doing well, but some of these kids certainly are not,” she said. “Until children have a long time to develop, you really can’t see these changes unless you follow them long-term.”
The researchers also looked separately at a subgroup of 19 children (26%) whose cranial ultrasounds showed mild nonspecific findings. These findings – such as lenticulostriate vasculopathy, choroid plexus cysts, subependymal cysts and calcifications – do not usually indicate any problems, but they appeared in a quarter of this population, considerably more than the approximately 5% typically seen in the general population, Dr. Mulkey said.
Though the findings did not reach significance, infants in this subgroup tended to have a lower WIDEA mobility z-scores (P = .054) and lower AIMS scores (P = .26) than the Zika-exposed infants with normal cranial ultrasounds.
“Mild nonspecific cranial ultrasound findings may represent a mild injury” related to exposure to their mother’s Zika infection during pregnancy, the researchers suggested. “It may be a risk factor for the lower mobility outcome,” Dr. Mulkey said.
The researchers hope to continue later follow-ups as the children age.
The research was funded by the Thrasher Research Fund. Dr. Mulkey had no conflicts of interest.
REPORTING FROM PAS 2019
Key clinical point: .
Major finding: Zika-exposed infants with normal fetal MRI neuroimaging showed increasingly lower mobility and social cognition skills as they approached their first birthday.
Study details: The findings are based on neurodevelopmental assessments of 72 Zika-exposed Colombian children at 4-18 months old.
Disclosures: The research was funded by the Thrasher Research Fund. Dr. Mulkey had no conflicts of interest.
FDA approves new etanercept biosimilar, Eticovo
The Food and Drug Administration has approved Eticovo (etanercept-ykro), a biosimilar of Enbrel (etanercept), for the treatment of several different rheumatologic and dermatologic conditions.
FDA approval was based in part on the results of a phase 3 trial in which 596 patients with moderate to severe rheumatoid arthritis uncontrolled by methotrexate received either Eticovo or Enbrel. The American College of Rheumatology 20% response rate after 24 weeks was 78.1% for Eticovo and 80.3% for Enbrel; the two drugs were statistically equivalent. Both groups had statistically equivalent rates of treatment-emergent adverse events (55.2% vs. 58.2%).
According to the label, Eticovo is a tumor necrosis factor blocker approved for the treatment of rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis in patients aged 4 years or older. The most common adverse events associated with the drug include infections and injection site reactions.
Eticovo is the second etanercept biosimilar approved by the FDA. The first FDA-approved etanercept biosimilar, etanercept-szzs (Erelzi), is currently facing a legal challenge from Amgen, the manufacturer of Enbrel.
The Food and Drug Administration has approved Eticovo (etanercept-ykro), a biosimilar of Enbrel (etanercept), for the treatment of several different rheumatologic and dermatologic conditions.
FDA approval was based in part on the results of a phase 3 trial in which 596 patients with moderate to severe rheumatoid arthritis uncontrolled by methotrexate received either Eticovo or Enbrel. The American College of Rheumatology 20% response rate after 24 weeks was 78.1% for Eticovo and 80.3% for Enbrel; the two drugs were statistically equivalent. Both groups had statistically equivalent rates of treatment-emergent adverse events (55.2% vs. 58.2%).
According to the label, Eticovo is a tumor necrosis factor blocker approved for the treatment of rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis in patients aged 4 years or older. The most common adverse events associated with the drug include infections and injection site reactions.
Eticovo is the second etanercept biosimilar approved by the FDA. The first FDA-approved etanercept biosimilar, etanercept-szzs (Erelzi), is currently facing a legal challenge from Amgen, the manufacturer of Enbrel.
The Food and Drug Administration has approved Eticovo (etanercept-ykro), a biosimilar of Enbrel (etanercept), for the treatment of several different rheumatologic and dermatologic conditions.
FDA approval was based in part on the results of a phase 3 trial in which 596 patients with moderate to severe rheumatoid arthritis uncontrolled by methotrexate received either Eticovo or Enbrel. The American College of Rheumatology 20% response rate after 24 weeks was 78.1% for Eticovo and 80.3% for Enbrel; the two drugs were statistically equivalent. Both groups had statistically equivalent rates of treatment-emergent adverse events (55.2% vs. 58.2%).
According to the label, Eticovo is a tumor necrosis factor blocker approved for the treatment of rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis in patients aged 4 years or older. The most common adverse events associated with the drug include infections and injection site reactions.
Eticovo is the second etanercept biosimilar approved by the FDA. The first FDA-approved etanercept biosimilar, etanercept-szzs (Erelzi), is currently facing a legal challenge from Amgen, the manufacturer of Enbrel.
Measles cases for 2019 now at postelimination high
according to the Centers for Disease Control and Prevention.
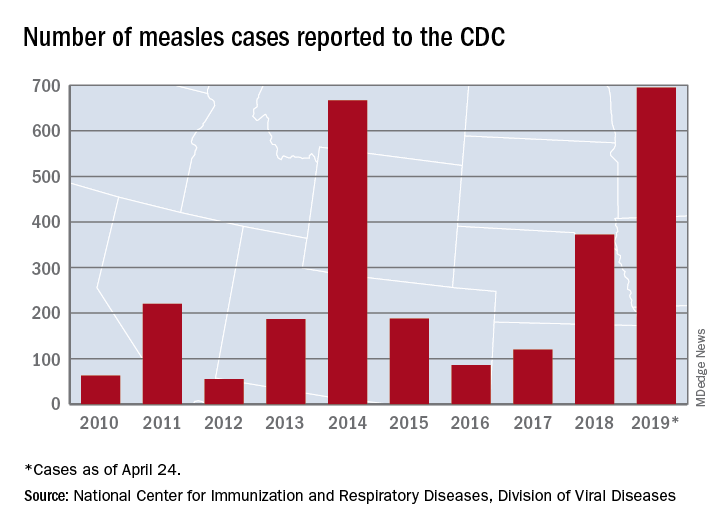
As of Wednesday, April 24, the case count for measles is 695, which eclipses the mark of 667 cases that had been the highest since the disease was declared to be eliminated from this country in 2000, the CDC reported.
“The high number of cases in 2019 is primarily the result of a few large outbreaks – one in Washington State and two large outbreaks in New York that started in late 2018. The outbreaks in New York City and New York State are among the largest and longest lasting since measles elimination in 2000. The longer these outbreaks continue, the greater the chance measles will again get a sustained foothold in the United States,” according to a written statement by the CDC.
Although these outbreaks began when the virus was brought into this country by unvaccinated travelers from other countries where there is widespread transmission, “a significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine. Some organizations are deliberately targeting these communities with inaccurate and misleading information about vaccines,” according to the statement.
“Measles is not a harmless childhood illness, but a highly contagious, potentially life-threatening disease,” Health and Human Services Secretary Alex Azar said in a separate statement. “We have the ability to safely protect our children and our communities. Vaccines are a safe, highly effective public health solution that can prevent this disease. The measles vaccines are among the most extensively studied medical products we have, and their safety has been firmly established over many years in some of the largest vaccine studies ever undertaken. With a safe and effective vaccine that protects against measles, the suffering we are seeing is avoidable.”
according to the Centers for Disease Control and Prevention.

As of Wednesday, April 24, the case count for measles is 695, which eclipses the mark of 667 cases that had been the highest since the disease was declared to be eliminated from this country in 2000, the CDC reported.
“The high number of cases in 2019 is primarily the result of a few large outbreaks – one in Washington State and two large outbreaks in New York that started in late 2018. The outbreaks in New York City and New York State are among the largest and longest lasting since measles elimination in 2000. The longer these outbreaks continue, the greater the chance measles will again get a sustained foothold in the United States,” according to a written statement by the CDC.
Although these outbreaks began when the virus was brought into this country by unvaccinated travelers from other countries where there is widespread transmission, “a significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine. Some organizations are deliberately targeting these communities with inaccurate and misleading information about vaccines,” according to the statement.
“Measles is not a harmless childhood illness, but a highly contagious, potentially life-threatening disease,” Health and Human Services Secretary Alex Azar said in a separate statement. “We have the ability to safely protect our children and our communities. Vaccines are a safe, highly effective public health solution that can prevent this disease. The measles vaccines are among the most extensively studied medical products we have, and their safety has been firmly established over many years in some of the largest vaccine studies ever undertaken. With a safe and effective vaccine that protects against measles, the suffering we are seeing is avoidable.”
according to the Centers for Disease Control and Prevention.

As of Wednesday, April 24, the case count for measles is 695, which eclipses the mark of 667 cases that had been the highest since the disease was declared to be eliminated from this country in 2000, the CDC reported.
“The high number of cases in 2019 is primarily the result of a few large outbreaks – one in Washington State and two large outbreaks in New York that started in late 2018. The outbreaks in New York City and New York State are among the largest and longest lasting since measles elimination in 2000. The longer these outbreaks continue, the greater the chance measles will again get a sustained foothold in the United States,” according to a written statement by the CDC.
Although these outbreaks began when the virus was brought into this country by unvaccinated travelers from other countries where there is widespread transmission, “a significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine. Some organizations are deliberately targeting these communities with inaccurate and misleading information about vaccines,” according to the statement.
“Measles is not a harmless childhood illness, but a highly contagious, potentially life-threatening disease,” Health and Human Services Secretary Alex Azar said in a separate statement. “We have the ability to safely protect our children and our communities. Vaccines are a safe, highly effective public health solution that can prevent this disease. The measles vaccines are among the most extensively studied medical products we have, and their safety has been firmly established over many years in some of the largest vaccine studies ever undertaken. With a safe and effective vaccine that protects against measles, the suffering we are seeing is avoidable.”









