User login
Hospital Dermatology: Review of Research in 2022-2023
Dermatologists improve the diagnostic accuracy and quality of care of patients in the hospital setting. They help shorten the length of stay, improve outpatient follow-up, and reduce the rate of hospital readmission.1 Medicare beneficiaries hospitalized with skin conditions at institutions with a dermatology hospitalist—a provider with a specialty interest in inpatient dermatology—have 24% lower odds of risk-adjusted 30-day mortality and 12% lower odds of risk-adjusted 30-day readmissions.2
In the last year, research among the dermatology hospitalist community has actively contributed to our understanding of challenging inpatient skin diseases and has identified new ways in which dermatologists can contribute to the care of hospitalized patients. In this review, we highlight 4 areas of focus from the published literature in 2022-2023—severe cutaneous adverse reactions, supportive oncodermatology, cost of inpatient services, and teledermatology.
Severe Cutaneous Adverse Reactions: Old and New
Severe cutaneous adverse reactions to medications frequently are encountered in the inpatient setting. Dermatology hospitalists are well positioned to phenotype these reactions, drawing insights that aid in identifying, characterizing, risk stratifying, and managing these conditions, which have considerable morbidity and mortality.
A recent 20-year retrospective review of cases of acute generalized exanthematous pustulosis (N=340) across 10 academic systems—the largest to date—improves our understanding of the features of this rare entity.3 The authors found that acute generalized exanthematous pustulosis most often is triggered by β-lactam and other antibiotics (75.5%) and is accompanied by fever (49.7%), neutrophilia (85.1%), and eosinophilia (52.1%). Kidney and liver involvement occur in less than 10% of cases, and mortality rates are low but not zero, with an all-cause 30-day mortality rate of 3.5%.3
In a multi-institutional retrospective study of 68 patients diagnosed with DRESS (drug reaction with eosinophilia and systemic symptoms) syndrome, Sharma et al4 developed a scoring system to identify those at greatest risk for DRESS recurrence. Variables associated with recurrence including younger age, female sex, and features considered atypical for DRESS syndrome—nonmorbilliform rash; absence of facial edema; antinuclear antibody positivity; medication class other than antibiotic, antigout, or antiseizure—were used to develop a “ReDRESS” score. This predictive model had a sensitivity of 73% and specificity of 83% for predicting DRESS recurrence.4
Another case series characterized SCoRCH (sudden conjunctivitis, lymphopenia, sunburnlike rash, and hemodynamic changes), a newly described hypersensitivity reaction to trimethoprim-sulfamethoxazole.5 The onset of this reaction typically occurs 4 to 11 days after initiation of trimethoprim-sulfamethoxazole but can occur as quickly as 1 day following re-exposure. Patients are systemically ill with fever, hypotension, tachycardia, acute renal insufficiency, and transaminitis, and they have a diffuse sunburnlike erythema without scale, facial edema, and conjunctivitis. It is thought this distinct hypersensitivity reaction may be mediated by IL-6, which has a role in triggering a sepsislike physiology, with vasodilation, hypotension, and edema.5
A systematic review and meta-analysis found that sulfonamides remain the most prominent cause of Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN).6 A case-control study described SJS/TEN presentations triggered by Mycoplasma, advocating for routine Mycoplasma screening, especially in patients without a clear medication culprit. Mycoplasma-induced cases carried statistically lower rates of mortality (0%) compared with medication-induced cases (22.5%).7 Another prospective open-label study evaluated SJS/TEN management by randomizing 25 patients to receive either combination therapy with methylprednisolone plus a tumor necrosis factor α inhibitor or methylprednisolone alone.8 Anti–tumor necrosis factor therapy was associated with a shorter length of initial steroid treatment and duration of the acute stage, hospitalization, and time to re-epithelialization8; however, as in a prior randomized unblinded trial,9 there was no difference in mortality between the 2 groups.
There is limited high-quality evidence to support the use of any systemic immunomodulator to decrease SJS/TEN–related mortality.10 A Cochrane systematic review highlighted the many limitations of the available data due to variations in presentation, assessment, and management.11 Because SJS/TEN is rare, powering studies based on mortality is infeasible; the authors calculated that 2872 participants were needed to detect a 50% mortality reduction among those with SCORTEN (severity-of-illness score for TEN) scores of 0 to 1.11 Therefore, collaborative efforts using appropriate outcomes measures (eg, time to re-epithelialization, length of hospital stay), standardized terminology and dosing regimens, and adaptive trial designs are needed. Consensus-derived assessment and treatment protocols could help account for variation, ensure consistency in treatment, and enable head-to-head comparisons. Members of the Society of Dermatology Hospitalists are working on efforts to standardize terminology and validate outcomes measures needed for future studies.12
Supportive Oncodermatology: A New Frontier
With the advent of immune checkpoint inhibitors (ICIs) for a growing number of cancers, dermatologists have become critical to identifying and managing cutaneous immune-related adverse events (cirAEs). Recent findings have demonstrated that dermatology input improves patient outcomes, not only regarding the treatment of dermatoses but also by augmenting cancer-related survival. One group found that patients with cirAEs who were evaluated by a dermatologist had improved progression-free (hazard ratio, 0.69; 95% CI, 0.54-0.87; P=.002) and overall survival rates (hazard ratio, 0.62; 95% CI, 0.45-0.84; P=.002), controlling for cirAE severity, age, sex, cancer type, and ICI subtype. Patients who were under the care of a dermatologist also were more likely to resume ICI therapy following an interruption (odds ratio, 10.52; 95% CI, 5.15-21.48; P<.001).13 Dermatologists help to optimize skin-directed and targeted therapies, such as dupilumab, minimizing exposure to systemic immunosuppression in these complex patients.14
Supportive oncodermatologists also have made important observations on how cirAEs relate to other adverse events and prognosis. A review of 628 patients found that almost half of those with cirAEs had co-occurring noncutaneous immune-related adverse events, most commonly pulmonary. Psoriasiform eruptions were most frequently associated with noncutaneous immune-related adverse events, and cutaneous reactions frequently preceded the development of systemic manifestations, serving as a clinical biomarker to provide prognostic information.15 A review of 95 patients found that spongiotic and lichenoid interface reactions were associated with decreased mortality rates, whereas vacuolar interface and perivascular dermatitis were associated with increased mortality.16
As with severe cutaneous adverse events, dermatology input has been critical for accurately phenotyping and risk stratifying these novel reactions. The dermatologist’s skill set is necessary for optimizing skin-directed and targeted therapies while minimizing systemic immunosuppression, thereby improving patient outcomes with respect to rash, cancer response, and survival.
The Cost of Inpatient Skin Disease
Hospitalizations account for approximately half of all health care expenditures, and hospital readmission, seen as a measure of the quality of health care delivery, can double this cost.17 Identifying and developing protocols for addressing patients with complex chronic inflammatory disorders is one strategy for improving outcomes and reducing financial burden. Inpatient dermatologists have identified hidradenitis suppurativa as one disease that can benefit from early intervention by dermatologists in the hospital, with its 30-day (17.8%) and 180-day (48.6%) readmission rates being comparable to those of heart failure.18
Following an index emergency department (ED) visit, 17.2% (3484/20,269) of patients with HS have at least 1 return ED visit within 30 days, while only 2.4% (483/20,269) have a dermatology visit within the same time frame.19 Understanding the risk factors for hospital readmission and ED utilization, including severity of illness, the presence of medical comorbidities, health coverage under Medicaid, and receipt of opioids, can allow dermatologists to anticipate those at greatest risk.19 Opportunities exist for cross-specialty interventions to anticipate and address modifiable risk factors. Shorter time to dermatology outpatient follow-up leads to improved clinic attendance and may help reduce ED utilization and hospital readmission.20
Teledermatology: Leveraging Inpatient Expertise
Although the benefit of inpatient dermatologic care is substantial, access to that care is finite. Following the COVID-19 pandemic, there is an increased acceptance of telemedicine and the long-term role it can play in leveraging dermatologic expertise, including meeting the increasing demand for inpatient dermatology care in rural and resource-poor communities.21
Recent studies conducted by dermatology hospitalists have illustrated the value of asynchronous store-and-forward technology in settings lacking access to consultative dermatology.22,23 Stephens et al22 found that expanding provider-to-provider electronic consultation (e-consultation) capacity to an inpatient rehabilitation facility resulted in completed consultations within 1.5 days compared with a 7- to 14-day wait time for patients attending an in-person urgent access dermatology clinic. In another study, the implementation of asynchronous dermatology e-consultations for immunobullous diseases, vasculitis, and herpes zoster resulted in a change in diagnosis 86% of the time, accompanied by at least 1 new systemic or topical therapy recommendation.23
Researchers also identified ways in which teledermatology can be inelegant and proposed specific supplemental data to aid in diagnosis. A review of 126 inpatient e-consultations demonstrated limitations related to the diagnosis of skin and soft-tissue infections. In two-thirds to three-quarters of cases, potentially useful descriptive information was missing, and in 70% (88/126), images were not appropriately focused. The authors developed a detailed checklist to help primary medical teams focus their differential diagnoses.24 A recent pilot study found that supplementation of clinical information with a standardized questionnaire and thermal images improved the accuracy of cellulitis diagnosis. Using this method, there was no difference in accuracy between dermatology hospitalists and other board-certified dermatologists, supporting the notion that any dermatologist can fulfill this need successfully, even without specific inpatient experience.25 Due to the high incidence and cost of cellulitis and related hospital admissions,26 such an intervention could have a considerable financial and patient safety impact.
Final Thoughts
This last year brought many changes to the health care landscape, the recession of a global pandemic, and an increasingly complex health care delivery system. Inpatient dermatologists met these challenges by providing high-quality dermatologic care and practice-modifying research in the areas of severe cutaneous adverse reactions, supportive oncodermatology, hospital readmission, telemedicine, and more, demonstrating the value of dermatologic expertise in the hospital setting.
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528.
- Puri P, Pollock BD, Yousif M, et al. Association of Society of Dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375.
- Creadore A, Desai S, Alloo A, et al. Clinical characteristics, disease course, and outcomes of patients with acute generalized exanthematous pustulosis in the US. JAMA Dermatol. 2022;158:176-183.
- Sharma AN, Murphy K, Shwe S, et al. Predicting DRESS syndrome recurrence—the ReDRESS score. JAMA Dermatol. 2022;158:1445-1447.
- Brian M, Rose EK, Mauskar MM, et al. Sudden conjunctivitis, lymphopenia, and rash combined with hemodynamic changes (SCoRCH) after trimethoprim-sulfamethoxazole use: a case series study of a hypersensitivity reaction. JAMA Dermatol. 2023;159:73-78.
- Lee EY, Knox C, Phillips EJ. Worldwide prevalence of antibiotic-associated Stevens-Johnson syndrome and toxic epidermal necrolysis: a systematic review and meta-analysis. JAMA Dermatol. 2023;159:384-392.
- Liew YCC, Choo KJL, Oh CC, et al. Mycoplasma-induced Stevens-Johnson syndrome/toxic epidermal necrolysis: case-control analysis of a cohort managed in a specialized center. J Am Acad Dermatol. 2022;86:811-817.
- Ao S, Gao X, Zhan J, et al. Inhibition of tumor necrosis factor improves conventional steroid therapy for Stevens-Johnson syndrome/toxic epidermal necrolysis in a cohort of patients. J Am Acad Dermatol. 2022;86:1236-1245.
- Wang CW, Yang LY, Chen CB, et al; the Taiwan Severe Cutaneous Adverse Reaction (TSCAR) Consortium. Randomized, controlled trial of TNF-α antagonist in CTL-mediated severe cutaneous adverse reactions. J Clin Invest. 2018;128:985-996.
- Han JJ, Creadore A, Seminario-Vidal L, et al. Medical management of Stevens-Johnson syndrome/toxic epidermal necrolysis among North American dermatologists. J Am Acad Dermatol. 2022;87:429-431.
- Noe MH, Micheletti RG. Systemic interventions for treatment of Stevens-Johnson syndrome/toxic epidermal necrolysis: summary of a Cochrane review. JAMA Dermatol. 2022;158:1436-1437.
- Waters M, Dobry A, Le ST, et al. Development of a skin-directed scoring system for Stevens-Johnson syndrome and epidermal necrolysis: a Delphi consensus exercise. JAMA Dermatol. 2023;159:772-777.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Said JT, Elman SA, Perez-Chada LM, et al. Treatment of immune checkpoint inhibitor-mediated psoriasis: a systematic review. J Am Acad Dermatol. 2022;87:399-400.
- Asdourian MS, Shah N, Jacoby TV, et al. Evaluating patterns of co-occurrence between cutaneous and noncutaneous immune-related adverse events after immune checkpoint inhibitor therapy. J Am Acad Dermatol. 2023;88:246-249.
- Hirotsu KE, Scott MKD, Marquez C, et al. Histologic subtype of cutaneous immune-related adverse events predicts overall survival in patients receiving immune checkpoint inhibitors. J Am Acad Dermatol. 2022;87:651-653.
- Benbassat J, Taragin M. Hospital readmissions as a measure of quality of health care: advantages and limitations. Arch Intern Med. 2000;160:1074-1081.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the nationwide readmissions database. J Am Acad Dermatol. 2022;87:188-192.
- Wang CX, Buss JL, Keller M, et al. Factors associated with dermatologic follow-up vs emergency department return in patients with hidradenitis suppurativa after an initial emergency department visit. JAMA Dermatol. 2022;158:1378-1386.
- Zakaria A, Chang AY, Kim-Lim P, et al. Predictors of postdischarge follow-up attendance among hospitalized dermatology patients: disparities and potential interventions. J Am Acad Dermatol. 2022;87:186-188.
- Arnold JD, Yoon S, Kirkorian AY. The national burden of inpatient dermatology in adults. J Am Acad Dermatol. 2019;80:425-432. doi:10.1016/j.jaad.2018.06.070
- Stephens MR, Das S, Smith GP. Utilization and outcomes of an asynchronous teledermatology pilot for an inpatient rehabilitation hospital. J Am Acad Dermatol. 2022;87:421-423.
- Ortiz C, Khosravi H, Kettering C, et al. Concordance data for inpatient asynchronous eDermatology consultation for immunobullous disease, zoster, and vasculitis. J Am Acad Dermatol. 2022;86:918-920.
- Salle R, Hua C, Mongereau M, et al. Challenges and limitations of teledermatology for skin and soft-tissue infections: a real-world study of an expert center. J Am Acad Dermatol. 2023;88:457-459.
- Creadore A, Manjaly P, Tkachenko E, et al. The utility of augmented teledermatology to improve dermatologist diagnosis of cellulitis: a cross-sectional study. Arch Dermatol Res. 2023;315:1347-1353.
- Weng QY, Raff AB, Cohen JM, et al. Costs and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol. 2017;153:141-146.
Dermatologists improve the diagnostic accuracy and quality of care of patients in the hospital setting. They help shorten the length of stay, improve outpatient follow-up, and reduce the rate of hospital readmission.1 Medicare beneficiaries hospitalized with skin conditions at institutions with a dermatology hospitalist—a provider with a specialty interest in inpatient dermatology—have 24% lower odds of risk-adjusted 30-day mortality and 12% lower odds of risk-adjusted 30-day readmissions.2
In the last year, research among the dermatology hospitalist community has actively contributed to our understanding of challenging inpatient skin diseases and has identified new ways in which dermatologists can contribute to the care of hospitalized patients. In this review, we highlight 4 areas of focus from the published literature in 2022-2023—severe cutaneous adverse reactions, supportive oncodermatology, cost of inpatient services, and teledermatology.
Severe Cutaneous Adverse Reactions: Old and New
Severe cutaneous adverse reactions to medications frequently are encountered in the inpatient setting. Dermatology hospitalists are well positioned to phenotype these reactions, drawing insights that aid in identifying, characterizing, risk stratifying, and managing these conditions, which have considerable morbidity and mortality.
A recent 20-year retrospective review of cases of acute generalized exanthematous pustulosis (N=340) across 10 academic systems—the largest to date—improves our understanding of the features of this rare entity.3 The authors found that acute generalized exanthematous pustulosis most often is triggered by β-lactam and other antibiotics (75.5%) and is accompanied by fever (49.7%), neutrophilia (85.1%), and eosinophilia (52.1%). Kidney and liver involvement occur in less than 10% of cases, and mortality rates are low but not zero, with an all-cause 30-day mortality rate of 3.5%.3
In a multi-institutional retrospective study of 68 patients diagnosed with DRESS (drug reaction with eosinophilia and systemic symptoms) syndrome, Sharma et al4 developed a scoring system to identify those at greatest risk for DRESS recurrence. Variables associated with recurrence including younger age, female sex, and features considered atypical for DRESS syndrome—nonmorbilliform rash; absence of facial edema; antinuclear antibody positivity; medication class other than antibiotic, antigout, or antiseizure—were used to develop a “ReDRESS” score. This predictive model had a sensitivity of 73% and specificity of 83% for predicting DRESS recurrence.4
Another case series characterized SCoRCH (sudden conjunctivitis, lymphopenia, sunburnlike rash, and hemodynamic changes), a newly described hypersensitivity reaction to trimethoprim-sulfamethoxazole.5 The onset of this reaction typically occurs 4 to 11 days after initiation of trimethoprim-sulfamethoxazole but can occur as quickly as 1 day following re-exposure. Patients are systemically ill with fever, hypotension, tachycardia, acute renal insufficiency, and transaminitis, and they have a diffuse sunburnlike erythema without scale, facial edema, and conjunctivitis. It is thought this distinct hypersensitivity reaction may be mediated by IL-6, which has a role in triggering a sepsislike physiology, with vasodilation, hypotension, and edema.5
A systematic review and meta-analysis found that sulfonamides remain the most prominent cause of Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN).6 A case-control study described SJS/TEN presentations triggered by Mycoplasma, advocating for routine Mycoplasma screening, especially in patients without a clear medication culprit. Mycoplasma-induced cases carried statistically lower rates of mortality (0%) compared with medication-induced cases (22.5%).7 Another prospective open-label study evaluated SJS/TEN management by randomizing 25 patients to receive either combination therapy with methylprednisolone plus a tumor necrosis factor α inhibitor or methylprednisolone alone.8 Anti–tumor necrosis factor therapy was associated with a shorter length of initial steroid treatment and duration of the acute stage, hospitalization, and time to re-epithelialization8; however, as in a prior randomized unblinded trial,9 there was no difference in mortality between the 2 groups.
There is limited high-quality evidence to support the use of any systemic immunomodulator to decrease SJS/TEN–related mortality.10 A Cochrane systematic review highlighted the many limitations of the available data due to variations in presentation, assessment, and management.11 Because SJS/TEN is rare, powering studies based on mortality is infeasible; the authors calculated that 2872 participants were needed to detect a 50% mortality reduction among those with SCORTEN (severity-of-illness score for TEN) scores of 0 to 1.11 Therefore, collaborative efforts using appropriate outcomes measures (eg, time to re-epithelialization, length of hospital stay), standardized terminology and dosing regimens, and adaptive trial designs are needed. Consensus-derived assessment and treatment protocols could help account for variation, ensure consistency in treatment, and enable head-to-head comparisons. Members of the Society of Dermatology Hospitalists are working on efforts to standardize terminology and validate outcomes measures needed for future studies.12
Supportive Oncodermatology: A New Frontier
With the advent of immune checkpoint inhibitors (ICIs) for a growing number of cancers, dermatologists have become critical to identifying and managing cutaneous immune-related adverse events (cirAEs). Recent findings have demonstrated that dermatology input improves patient outcomes, not only regarding the treatment of dermatoses but also by augmenting cancer-related survival. One group found that patients with cirAEs who were evaluated by a dermatologist had improved progression-free (hazard ratio, 0.69; 95% CI, 0.54-0.87; P=.002) and overall survival rates (hazard ratio, 0.62; 95% CI, 0.45-0.84; P=.002), controlling for cirAE severity, age, sex, cancer type, and ICI subtype. Patients who were under the care of a dermatologist also were more likely to resume ICI therapy following an interruption (odds ratio, 10.52; 95% CI, 5.15-21.48; P<.001).13 Dermatologists help to optimize skin-directed and targeted therapies, such as dupilumab, minimizing exposure to systemic immunosuppression in these complex patients.14
Supportive oncodermatologists also have made important observations on how cirAEs relate to other adverse events and prognosis. A review of 628 patients found that almost half of those with cirAEs had co-occurring noncutaneous immune-related adverse events, most commonly pulmonary. Psoriasiform eruptions were most frequently associated with noncutaneous immune-related adverse events, and cutaneous reactions frequently preceded the development of systemic manifestations, serving as a clinical biomarker to provide prognostic information.15 A review of 95 patients found that spongiotic and lichenoid interface reactions were associated with decreased mortality rates, whereas vacuolar interface and perivascular dermatitis were associated with increased mortality.16
As with severe cutaneous adverse events, dermatology input has been critical for accurately phenotyping and risk stratifying these novel reactions. The dermatologist’s skill set is necessary for optimizing skin-directed and targeted therapies while minimizing systemic immunosuppression, thereby improving patient outcomes with respect to rash, cancer response, and survival.
The Cost of Inpatient Skin Disease
Hospitalizations account for approximately half of all health care expenditures, and hospital readmission, seen as a measure of the quality of health care delivery, can double this cost.17 Identifying and developing protocols for addressing patients with complex chronic inflammatory disorders is one strategy for improving outcomes and reducing financial burden. Inpatient dermatologists have identified hidradenitis suppurativa as one disease that can benefit from early intervention by dermatologists in the hospital, with its 30-day (17.8%) and 180-day (48.6%) readmission rates being comparable to those of heart failure.18
Following an index emergency department (ED) visit, 17.2% (3484/20,269) of patients with HS have at least 1 return ED visit within 30 days, while only 2.4% (483/20,269) have a dermatology visit within the same time frame.19 Understanding the risk factors for hospital readmission and ED utilization, including severity of illness, the presence of medical comorbidities, health coverage under Medicaid, and receipt of opioids, can allow dermatologists to anticipate those at greatest risk.19 Opportunities exist for cross-specialty interventions to anticipate and address modifiable risk factors. Shorter time to dermatology outpatient follow-up leads to improved clinic attendance and may help reduce ED utilization and hospital readmission.20
Teledermatology: Leveraging Inpatient Expertise
Although the benefit of inpatient dermatologic care is substantial, access to that care is finite. Following the COVID-19 pandemic, there is an increased acceptance of telemedicine and the long-term role it can play in leveraging dermatologic expertise, including meeting the increasing demand for inpatient dermatology care in rural and resource-poor communities.21
Recent studies conducted by dermatology hospitalists have illustrated the value of asynchronous store-and-forward technology in settings lacking access to consultative dermatology.22,23 Stephens et al22 found that expanding provider-to-provider electronic consultation (e-consultation) capacity to an inpatient rehabilitation facility resulted in completed consultations within 1.5 days compared with a 7- to 14-day wait time for patients attending an in-person urgent access dermatology clinic. In another study, the implementation of asynchronous dermatology e-consultations for immunobullous diseases, vasculitis, and herpes zoster resulted in a change in diagnosis 86% of the time, accompanied by at least 1 new systemic or topical therapy recommendation.23
Researchers also identified ways in which teledermatology can be inelegant and proposed specific supplemental data to aid in diagnosis. A review of 126 inpatient e-consultations demonstrated limitations related to the diagnosis of skin and soft-tissue infections. In two-thirds to three-quarters of cases, potentially useful descriptive information was missing, and in 70% (88/126), images were not appropriately focused. The authors developed a detailed checklist to help primary medical teams focus their differential diagnoses.24 A recent pilot study found that supplementation of clinical information with a standardized questionnaire and thermal images improved the accuracy of cellulitis diagnosis. Using this method, there was no difference in accuracy between dermatology hospitalists and other board-certified dermatologists, supporting the notion that any dermatologist can fulfill this need successfully, even without specific inpatient experience.25 Due to the high incidence and cost of cellulitis and related hospital admissions,26 such an intervention could have a considerable financial and patient safety impact.
Final Thoughts
This last year brought many changes to the health care landscape, the recession of a global pandemic, and an increasingly complex health care delivery system. Inpatient dermatologists met these challenges by providing high-quality dermatologic care and practice-modifying research in the areas of severe cutaneous adverse reactions, supportive oncodermatology, hospital readmission, telemedicine, and more, demonstrating the value of dermatologic expertise in the hospital setting.
Dermatologists improve the diagnostic accuracy and quality of care of patients in the hospital setting. They help shorten the length of stay, improve outpatient follow-up, and reduce the rate of hospital readmission.1 Medicare beneficiaries hospitalized with skin conditions at institutions with a dermatology hospitalist—a provider with a specialty interest in inpatient dermatology—have 24% lower odds of risk-adjusted 30-day mortality and 12% lower odds of risk-adjusted 30-day readmissions.2
In the last year, research among the dermatology hospitalist community has actively contributed to our understanding of challenging inpatient skin diseases and has identified new ways in which dermatologists can contribute to the care of hospitalized patients. In this review, we highlight 4 areas of focus from the published literature in 2022-2023—severe cutaneous adverse reactions, supportive oncodermatology, cost of inpatient services, and teledermatology.
Severe Cutaneous Adverse Reactions: Old and New
Severe cutaneous adverse reactions to medications frequently are encountered in the inpatient setting. Dermatology hospitalists are well positioned to phenotype these reactions, drawing insights that aid in identifying, characterizing, risk stratifying, and managing these conditions, which have considerable morbidity and mortality.
A recent 20-year retrospective review of cases of acute generalized exanthematous pustulosis (N=340) across 10 academic systems—the largest to date—improves our understanding of the features of this rare entity.3 The authors found that acute generalized exanthematous pustulosis most often is triggered by β-lactam and other antibiotics (75.5%) and is accompanied by fever (49.7%), neutrophilia (85.1%), and eosinophilia (52.1%). Kidney and liver involvement occur in less than 10% of cases, and mortality rates are low but not zero, with an all-cause 30-day mortality rate of 3.5%.3
In a multi-institutional retrospective study of 68 patients diagnosed with DRESS (drug reaction with eosinophilia and systemic symptoms) syndrome, Sharma et al4 developed a scoring system to identify those at greatest risk for DRESS recurrence. Variables associated with recurrence including younger age, female sex, and features considered atypical for DRESS syndrome—nonmorbilliform rash; absence of facial edema; antinuclear antibody positivity; medication class other than antibiotic, antigout, or antiseizure—were used to develop a “ReDRESS” score. This predictive model had a sensitivity of 73% and specificity of 83% for predicting DRESS recurrence.4
Another case series characterized SCoRCH (sudden conjunctivitis, lymphopenia, sunburnlike rash, and hemodynamic changes), a newly described hypersensitivity reaction to trimethoprim-sulfamethoxazole.5 The onset of this reaction typically occurs 4 to 11 days after initiation of trimethoprim-sulfamethoxazole but can occur as quickly as 1 day following re-exposure. Patients are systemically ill with fever, hypotension, tachycardia, acute renal insufficiency, and transaminitis, and they have a diffuse sunburnlike erythema without scale, facial edema, and conjunctivitis. It is thought this distinct hypersensitivity reaction may be mediated by IL-6, which has a role in triggering a sepsislike physiology, with vasodilation, hypotension, and edema.5
A systematic review and meta-analysis found that sulfonamides remain the most prominent cause of Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN).6 A case-control study described SJS/TEN presentations triggered by Mycoplasma, advocating for routine Mycoplasma screening, especially in patients without a clear medication culprit. Mycoplasma-induced cases carried statistically lower rates of mortality (0%) compared with medication-induced cases (22.5%).7 Another prospective open-label study evaluated SJS/TEN management by randomizing 25 patients to receive either combination therapy with methylprednisolone plus a tumor necrosis factor α inhibitor or methylprednisolone alone.8 Anti–tumor necrosis factor therapy was associated with a shorter length of initial steroid treatment and duration of the acute stage, hospitalization, and time to re-epithelialization8; however, as in a prior randomized unblinded trial,9 there was no difference in mortality between the 2 groups.
There is limited high-quality evidence to support the use of any systemic immunomodulator to decrease SJS/TEN–related mortality.10 A Cochrane systematic review highlighted the many limitations of the available data due to variations in presentation, assessment, and management.11 Because SJS/TEN is rare, powering studies based on mortality is infeasible; the authors calculated that 2872 participants were needed to detect a 50% mortality reduction among those with SCORTEN (severity-of-illness score for TEN) scores of 0 to 1.11 Therefore, collaborative efforts using appropriate outcomes measures (eg, time to re-epithelialization, length of hospital stay), standardized terminology and dosing regimens, and adaptive trial designs are needed. Consensus-derived assessment and treatment protocols could help account for variation, ensure consistency in treatment, and enable head-to-head comparisons. Members of the Society of Dermatology Hospitalists are working on efforts to standardize terminology and validate outcomes measures needed for future studies.12
Supportive Oncodermatology: A New Frontier
With the advent of immune checkpoint inhibitors (ICIs) for a growing number of cancers, dermatologists have become critical to identifying and managing cutaneous immune-related adverse events (cirAEs). Recent findings have demonstrated that dermatology input improves patient outcomes, not only regarding the treatment of dermatoses but also by augmenting cancer-related survival. One group found that patients with cirAEs who were evaluated by a dermatologist had improved progression-free (hazard ratio, 0.69; 95% CI, 0.54-0.87; P=.002) and overall survival rates (hazard ratio, 0.62; 95% CI, 0.45-0.84; P=.002), controlling for cirAE severity, age, sex, cancer type, and ICI subtype. Patients who were under the care of a dermatologist also were more likely to resume ICI therapy following an interruption (odds ratio, 10.52; 95% CI, 5.15-21.48; P<.001).13 Dermatologists help to optimize skin-directed and targeted therapies, such as dupilumab, minimizing exposure to systemic immunosuppression in these complex patients.14
Supportive oncodermatologists also have made important observations on how cirAEs relate to other adverse events and prognosis. A review of 628 patients found that almost half of those with cirAEs had co-occurring noncutaneous immune-related adverse events, most commonly pulmonary. Psoriasiform eruptions were most frequently associated with noncutaneous immune-related adverse events, and cutaneous reactions frequently preceded the development of systemic manifestations, serving as a clinical biomarker to provide prognostic information.15 A review of 95 patients found that spongiotic and lichenoid interface reactions were associated with decreased mortality rates, whereas vacuolar interface and perivascular dermatitis were associated with increased mortality.16
As with severe cutaneous adverse events, dermatology input has been critical for accurately phenotyping and risk stratifying these novel reactions. The dermatologist’s skill set is necessary for optimizing skin-directed and targeted therapies while minimizing systemic immunosuppression, thereby improving patient outcomes with respect to rash, cancer response, and survival.
The Cost of Inpatient Skin Disease
Hospitalizations account for approximately half of all health care expenditures, and hospital readmission, seen as a measure of the quality of health care delivery, can double this cost.17 Identifying and developing protocols for addressing patients with complex chronic inflammatory disorders is one strategy for improving outcomes and reducing financial burden. Inpatient dermatologists have identified hidradenitis suppurativa as one disease that can benefit from early intervention by dermatologists in the hospital, with its 30-day (17.8%) and 180-day (48.6%) readmission rates being comparable to those of heart failure.18
Following an index emergency department (ED) visit, 17.2% (3484/20,269) of patients with HS have at least 1 return ED visit within 30 days, while only 2.4% (483/20,269) have a dermatology visit within the same time frame.19 Understanding the risk factors for hospital readmission and ED utilization, including severity of illness, the presence of medical comorbidities, health coverage under Medicaid, and receipt of opioids, can allow dermatologists to anticipate those at greatest risk.19 Opportunities exist for cross-specialty interventions to anticipate and address modifiable risk factors. Shorter time to dermatology outpatient follow-up leads to improved clinic attendance and may help reduce ED utilization and hospital readmission.20
Teledermatology: Leveraging Inpatient Expertise
Although the benefit of inpatient dermatologic care is substantial, access to that care is finite. Following the COVID-19 pandemic, there is an increased acceptance of telemedicine and the long-term role it can play in leveraging dermatologic expertise, including meeting the increasing demand for inpatient dermatology care in rural and resource-poor communities.21
Recent studies conducted by dermatology hospitalists have illustrated the value of asynchronous store-and-forward technology in settings lacking access to consultative dermatology.22,23 Stephens et al22 found that expanding provider-to-provider electronic consultation (e-consultation) capacity to an inpatient rehabilitation facility resulted in completed consultations within 1.5 days compared with a 7- to 14-day wait time for patients attending an in-person urgent access dermatology clinic. In another study, the implementation of asynchronous dermatology e-consultations for immunobullous diseases, vasculitis, and herpes zoster resulted in a change in diagnosis 86% of the time, accompanied by at least 1 new systemic or topical therapy recommendation.23
Researchers also identified ways in which teledermatology can be inelegant and proposed specific supplemental data to aid in diagnosis. A review of 126 inpatient e-consultations demonstrated limitations related to the diagnosis of skin and soft-tissue infections. In two-thirds to three-quarters of cases, potentially useful descriptive information was missing, and in 70% (88/126), images were not appropriately focused. The authors developed a detailed checklist to help primary medical teams focus their differential diagnoses.24 A recent pilot study found that supplementation of clinical information with a standardized questionnaire and thermal images improved the accuracy of cellulitis diagnosis. Using this method, there was no difference in accuracy between dermatology hospitalists and other board-certified dermatologists, supporting the notion that any dermatologist can fulfill this need successfully, even without specific inpatient experience.25 Due to the high incidence and cost of cellulitis and related hospital admissions,26 such an intervention could have a considerable financial and patient safety impact.
Final Thoughts
This last year brought many changes to the health care landscape, the recession of a global pandemic, and an increasingly complex health care delivery system. Inpatient dermatologists met these challenges by providing high-quality dermatologic care and practice-modifying research in the areas of severe cutaneous adverse reactions, supportive oncodermatology, hospital readmission, telemedicine, and more, demonstrating the value of dermatologic expertise in the hospital setting.
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528.
- Puri P, Pollock BD, Yousif M, et al. Association of Society of Dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375.
- Creadore A, Desai S, Alloo A, et al. Clinical characteristics, disease course, and outcomes of patients with acute generalized exanthematous pustulosis in the US. JAMA Dermatol. 2022;158:176-183.
- Sharma AN, Murphy K, Shwe S, et al. Predicting DRESS syndrome recurrence—the ReDRESS score. JAMA Dermatol. 2022;158:1445-1447.
- Brian M, Rose EK, Mauskar MM, et al. Sudden conjunctivitis, lymphopenia, and rash combined with hemodynamic changes (SCoRCH) after trimethoprim-sulfamethoxazole use: a case series study of a hypersensitivity reaction. JAMA Dermatol. 2023;159:73-78.
- Lee EY, Knox C, Phillips EJ. Worldwide prevalence of antibiotic-associated Stevens-Johnson syndrome and toxic epidermal necrolysis: a systematic review and meta-analysis. JAMA Dermatol. 2023;159:384-392.
- Liew YCC, Choo KJL, Oh CC, et al. Mycoplasma-induced Stevens-Johnson syndrome/toxic epidermal necrolysis: case-control analysis of a cohort managed in a specialized center. J Am Acad Dermatol. 2022;86:811-817.
- Ao S, Gao X, Zhan J, et al. Inhibition of tumor necrosis factor improves conventional steroid therapy for Stevens-Johnson syndrome/toxic epidermal necrolysis in a cohort of patients. J Am Acad Dermatol. 2022;86:1236-1245.
- Wang CW, Yang LY, Chen CB, et al; the Taiwan Severe Cutaneous Adverse Reaction (TSCAR) Consortium. Randomized, controlled trial of TNF-α antagonist in CTL-mediated severe cutaneous adverse reactions. J Clin Invest. 2018;128:985-996.
- Han JJ, Creadore A, Seminario-Vidal L, et al. Medical management of Stevens-Johnson syndrome/toxic epidermal necrolysis among North American dermatologists. J Am Acad Dermatol. 2022;87:429-431.
- Noe MH, Micheletti RG. Systemic interventions for treatment of Stevens-Johnson syndrome/toxic epidermal necrolysis: summary of a Cochrane review. JAMA Dermatol. 2022;158:1436-1437.
- Waters M, Dobry A, Le ST, et al. Development of a skin-directed scoring system for Stevens-Johnson syndrome and epidermal necrolysis: a Delphi consensus exercise. JAMA Dermatol. 2023;159:772-777.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Said JT, Elman SA, Perez-Chada LM, et al. Treatment of immune checkpoint inhibitor-mediated psoriasis: a systematic review. J Am Acad Dermatol. 2022;87:399-400.
- Asdourian MS, Shah N, Jacoby TV, et al. Evaluating patterns of co-occurrence between cutaneous and noncutaneous immune-related adverse events after immune checkpoint inhibitor therapy. J Am Acad Dermatol. 2023;88:246-249.
- Hirotsu KE, Scott MKD, Marquez C, et al. Histologic subtype of cutaneous immune-related adverse events predicts overall survival in patients receiving immune checkpoint inhibitors. J Am Acad Dermatol. 2022;87:651-653.
- Benbassat J, Taragin M. Hospital readmissions as a measure of quality of health care: advantages and limitations. Arch Intern Med. 2000;160:1074-1081.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the nationwide readmissions database. J Am Acad Dermatol. 2022;87:188-192.
- Wang CX, Buss JL, Keller M, et al. Factors associated with dermatologic follow-up vs emergency department return in patients with hidradenitis suppurativa after an initial emergency department visit. JAMA Dermatol. 2022;158:1378-1386.
- Zakaria A, Chang AY, Kim-Lim P, et al. Predictors of postdischarge follow-up attendance among hospitalized dermatology patients: disparities and potential interventions. J Am Acad Dermatol. 2022;87:186-188.
- Arnold JD, Yoon S, Kirkorian AY. The national burden of inpatient dermatology in adults. J Am Acad Dermatol. 2019;80:425-432. doi:10.1016/j.jaad.2018.06.070
- Stephens MR, Das S, Smith GP. Utilization and outcomes of an asynchronous teledermatology pilot for an inpatient rehabilitation hospital. J Am Acad Dermatol. 2022;87:421-423.
- Ortiz C, Khosravi H, Kettering C, et al. Concordance data for inpatient asynchronous eDermatology consultation for immunobullous disease, zoster, and vasculitis. J Am Acad Dermatol. 2022;86:918-920.
- Salle R, Hua C, Mongereau M, et al. Challenges and limitations of teledermatology for skin and soft-tissue infections: a real-world study of an expert center. J Am Acad Dermatol. 2023;88:457-459.
- Creadore A, Manjaly P, Tkachenko E, et al. The utility of augmented teledermatology to improve dermatologist diagnosis of cellulitis: a cross-sectional study. Arch Dermatol Res. 2023;315:1347-1353.
- Weng QY, Raff AB, Cohen JM, et al. Costs and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol. 2017;153:141-146.
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528.
- Puri P, Pollock BD, Yousif M, et al. Association of Society of Dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375.
- Creadore A, Desai S, Alloo A, et al. Clinical characteristics, disease course, and outcomes of patients with acute generalized exanthematous pustulosis in the US. JAMA Dermatol. 2022;158:176-183.
- Sharma AN, Murphy K, Shwe S, et al. Predicting DRESS syndrome recurrence—the ReDRESS score. JAMA Dermatol. 2022;158:1445-1447.
- Brian M, Rose EK, Mauskar MM, et al. Sudden conjunctivitis, lymphopenia, and rash combined with hemodynamic changes (SCoRCH) after trimethoprim-sulfamethoxazole use: a case series study of a hypersensitivity reaction. JAMA Dermatol. 2023;159:73-78.
- Lee EY, Knox C, Phillips EJ. Worldwide prevalence of antibiotic-associated Stevens-Johnson syndrome and toxic epidermal necrolysis: a systematic review and meta-analysis. JAMA Dermatol. 2023;159:384-392.
- Liew YCC, Choo KJL, Oh CC, et al. Mycoplasma-induced Stevens-Johnson syndrome/toxic epidermal necrolysis: case-control analysis of a cohort managed in a specialized center. J Am Acad Dermatol. 2022;86:811-817.
- Ao S, Gao X, Zhan J, et al. Inhibition of tumor necrosis factor improves conventional steroid therapy for Stevens-Johnson syndrome/toxic epidermal necrolysis in a cohort of patients. J Am Acad Dermatol. 2022;86:1236-1245.
- Wang CW, Yang LY, Chen CB, et al; the Taiwan Severe Cutaneous Adverse Reaction (TSCAR) Consortium. Randomized, controlled trial of TNF-α antagonist in CTL-mediated severe cutaneous adverse reactions. J Clin Invest. 2018;128:985-996.
- Han JJ, Creadore A, Seminario-Vidal L, et al. Medical management of Stevens-Johnson syndrome/toxic epidermal necrolysis among North American dermatologists. J Am Acad Dermatol. 2022;87:429-431.
- Noe MH, Micheletti RG. Systemic interventions for treatment of Stevens-Johnson syndrome/toxic epidermal necrolysis: summary of a Cochrane review. JAMA Dermatol. 2022;158:1436-1437.
- Waters M, Dobry A, Le ST, et al. Development of a skin-directed scoring system for Stevens-Johnson syndrome and epidermal necrolysis: a Delphi consensus exercise. JAMA Dermatol. 2023;159:772-777.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Said JT, Elman SA, Perez-Chada LM, et al. Treatment of immune checkpoint inhibitor-mediated psoriasis: a systematic review. J Am Acad Dermatol. 2022;87:399-400.
- Asdourian MS, Shah N, Jacoby TV, et al. Evaluating patterns of co-occurrence between cutaneous and noncutaneous immune-related adverse events after immune checkpoint inhibitor therapy. J Am Acad Dermatol. 2023;88:246-249.
- Hirotsu KE, Scott MKD, Marquez C, et al. Histologic subtype of cutaneous immune-related adverse events predicts overall survival in patients receiving immune checkpoint inhibitors. J Am Acad Dermatol. 2022;87:651-653.
- Benbassat J, Taragin M. Hospital readmissions as a measure of quality of health care: advantages and limitations. Arch Intern Med. 2000;160:1074-1081.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the nationwide readmissions database. J Am Acad Dermatol. 2022;87:188-192.
- Wang CX, Buss JL, Keller M, et al. Factors associated with dermatologic follow-up vs emergency department return in patients with hidradenitis suppurativa after an initial emergency department visit. JAMA Dermatol. 2022;158:1378-1386.
- Zakaria A, Chang AY, Kim-Lim P, et al. Predictors of postdischarge follow-up attendance among hospitalized dermatology patients: disparities and potential interventions. J Am Acad Dermatol. 2022;87:186-188.
- Arnold JD, Yoon S, Kirkorian AY. The national burden of inpatient dermatology in adults. J Am Acad Dermatol. 2019;80:425-432. doi:10.1016/j.jaad.2018.06.070
- Stephens MR, Das S, Smith GP. Utilization and outcomes of an asynchronous teledermatology pilot for an inpatient rehabilitation hospital. J Am Acad Dermatol. 2022;87:421-423.
- Ortiz C, Khosravi H, Kettering C, et al. Concordance data for inpatient asynchronous eDermatology consultation for immunobullous disease, zoster, and vasculitis. J Am Acad Dermatol. 2022;86:918-920.
- Salle R, Hua C, Mongereau M, et al. Challenges and limitations of teledermatology for skin and soft-tissue infections: a real-world study of an expert center. J Am Acad Dermatol. 2023;88:457-459.
- Creadore A, Manjaly P, Tkachenko E, et al. The utility of augmented teledermatology to improve dermatologist diagnosis of cellulitis: a cross-sectional study. Arch Dermatol Res. 2023;315:1347-1353.
- Weng QY, Raff AB, Cohen JM, et al. Costs and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol. 2017;153:141-146.
Practice Points
- A severe hypersensitivity reaction to trimethoprim-sulfamethoxazole—sudden conjunctivitis, lymphopenia, sunburnlike rash, and hemodynamic changes (SCoRCH)—has been described.
- Patients experiencing cutaneous reactions to immune checkpoint inhibitors have improved progression-free and overall survival rates if evaluated by a dermatologist who can optimize skin-directed and targeted therapies.
- Interventions, including shorter time to dermatology outpatient follow-up, are needed to reduce emergency department utilization by patients with hidradenitis suppurativa.
- Asynchronous store-and-forward dermatology e-consultation is effective for immunobullous diseases, vasculitis, herpes zoster, and cellulitis, demonstrating the utility of teledermatology in the inpatient setting, particularly when standardized data capture tools are used.
Potential Uses of Nonthermal Atmospheric Pressure Technology for Dermatologic Conditions in Children
Nonthermal atmospheric plasma (NTAP)(or cold atmospheric plasma [CAP]) is a rapidly developing treatment modality for a wide range of dermatologic conditions. Plasma (or ionized gas) refers to a state of matter composed of electrons, protons, and neutral atoms that generate reactive oxygen and nitrogen species.1 Plasma previously was created using thermal energy, but recent advances have allowed the creation of plasma using atmospheric pressure and room temperature; thus, NTAP can be used without causing damage to living tissue through heat.1 Plasma technology varies greatly, but it generally can be classified as either direct or indirect therapy; direct therapy uses the human body as an electrode, whereas indirect therapy creates plasma through the interaction between 2 electrode devices.1,2 When used on the skin, important dose-dependent relationships have been observed, with CAP application longer than 2 minutes being associated with increased keratinocyte and fibroblast apoptosis.2 Thus, CAP can cause diverse changes to the skin depending on application time and methodology. At adequate yet low concentrations, plasma can promote fibroblast proliferation and upregulate genes involved in collagen and transforming growth factor synthesis.1 Additionally, the reactive oxygen and nitrogen species created by NTAP have been shown to inactivate microorganisms through the destruction of biofilms, lead to diminished immune cell infiltration and cytokine release in autoimmune dermatologic conditions, and exert antitumor properties through cellular DNA damage.1-3 In dermatology, these properties can be harvested to promote wound healing at low doses and the treatment of proliferative skin conditions at high doses.1
Because of its novelty, the safety profile of NTAP is still under investigation, but preliminary studies are promising and show no damage to the skin barrier when excessive plasma exposure is avoided.4 However, dose- and time-dependent damage to cells has been shown. As a result, the exact dose of plasma considered safe is highly variable depending on the vessel, technique, and user, and future clinical research is needed to guide this methodology.4 Additionally, CAP has been shown to cause little pain at the skin surface and may lead to decreased levels of pain in healing wound sites.5 Given this promising safety profile and minimal discomfort to patients, NTAP technology remains promising for use in pediatric dermatology, but there are limited data to characterize its potential use in this population. In this systematic review, we aimed to elucidate reported applications of NTAP for skin conditions in children and discuss the trajectory of this technology in the future of pediatric dermatology.
Methodology
A comprehensive literature review was conducted to identify studies evaluating NTAP technology in pediatric populations using PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) guidelines. A search of PubMed, Embase, and Web of Science articles was conducted in April 2023 using the terms nonthermal atmospheric plasma or cold atmospheric plasma. All English-language articles that described the use of NTAP as a treatment in pediatric populations or articles that described NTAP use in the treatment of common conditions in this patient group were included based on a review of the article titles and abstracts by 2 independent reviewers, followed by full-text review of relevant articles (M.G., C.L.). Any discrepancies in eligible articles were settled by a third independent researcher (M.V.). One hundred twenty studies were identified, and 95 were screened for inclusion; 9 studies met inclusion criteria and were summarized in this review.
Results
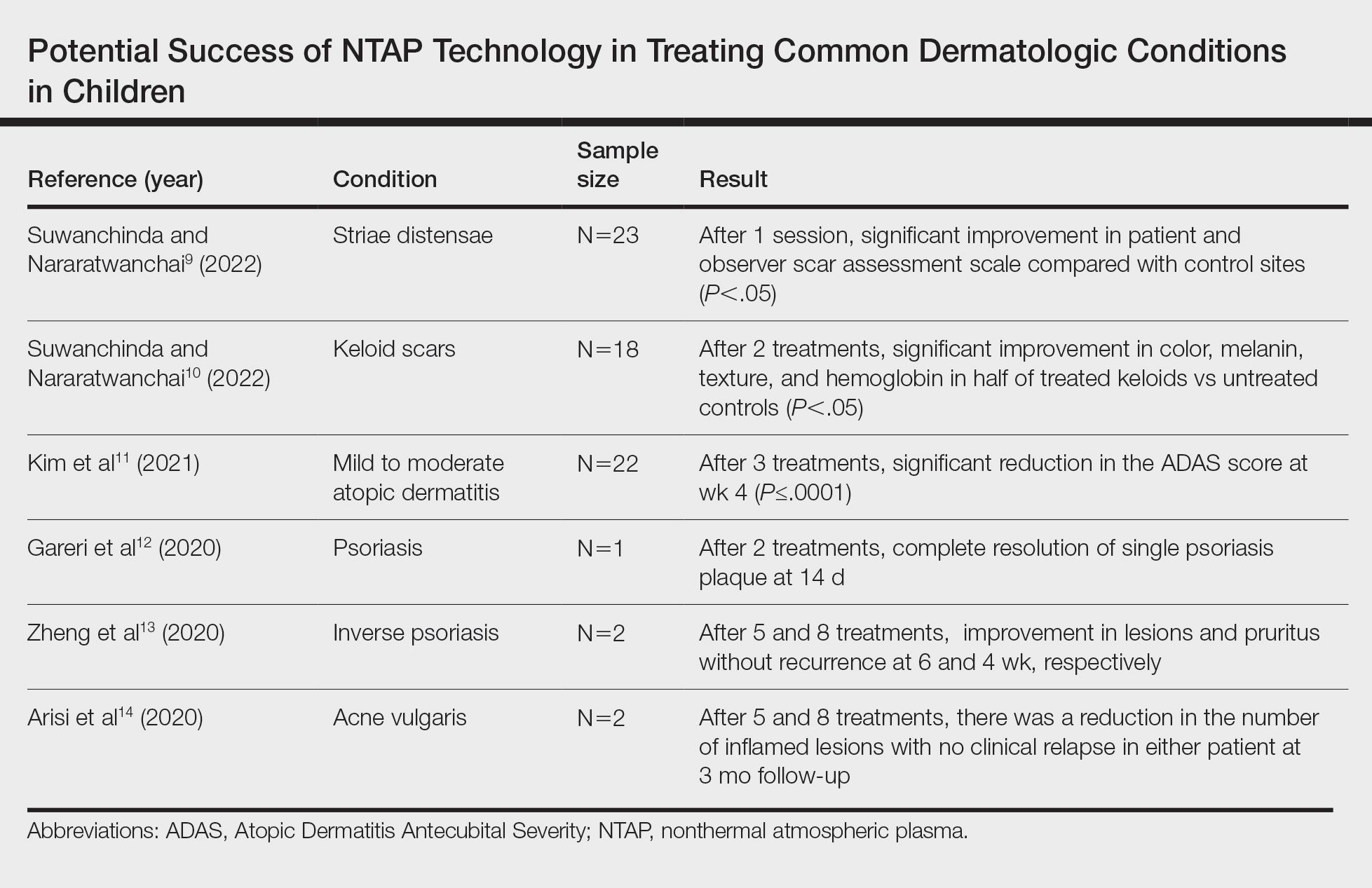
A total of 9 studies were included in this review: 3 describing the success of NTAP in pediatric populations6-8 and 6 describing the potential success of NTAP for dermatologic conditions commonly seen in children (Table).9-14
Studies Describing Success of NTAP—Three clinical reports described the efficacy of NTAP in pediatric dermatology. A case series from 2020 showed full clearance of warts in 100% of patients (n=5) with a 0% recurrence rate when NTAP treatment was applied for 2 minutes to each lesion during each treatment session with the electrode held 1 mm from the lesional surface.6 Each patient was followed up at 3 to 4 weeks, and treatment was repeated if lesions persisted. Patients reported no pain during the procedure, and no adverse effects were noted over the course of treatment.6 Second, a case report described full clearance of diaper dermatitis with no recurrence after 6 months following 6 treatments with NTAP in a 14-month-old girl.7 After treatment with econazole nitrate cream, oral antibiotics, and prednisone failed, CAP treatment was initiated. Each treatment lasted 15 minutes with 3-day time intervals between each of the 6 treatments. There were no adverse events or recurrence of rash at 6-month follow-up.7 A final case report described full clearance of molluscum contagiosum (MC), with no recurrence after 2 months following 4 treatments with NTAP in a 12-year-old boy.8 The patient had untreated MC on the face, neck, shoulder, and thighs. Lesions of the face were treated with CAP, while the other sites were treated with cantharidin using a 0.7% collodion-based solution. Four CAP treatments were performed at 1-month intervals, with CAP applied 1 mm from the lesional surfaces in a circular pattern for 2 minutes. At follow-up 2 months after the final treatment, the patient had no adverse effects and showed no pigmentary changes or scarring.8
Studies Describing the Potential Success of NTAP—Beyond these studies, limited research has been done on NTAP in pediatric populations. The Table summarizes 6 additional studies completed with promising treatment results for dermatologic conditions commonly seen in children: striae distensae, keloids, atopic dermatitis, psoriasis, inverse psoriasis, and acne vulgaris. Across all reports and studies, patients showed significant improvement in their dermatologic conditions following the use of NTAP technology with limited adverse effects reported (P<.05). Suwanchinda and Nararatwanchai9 studied the use of CAP for the treatment of striae distensae. They recruited 23 patients and treated half the body with CAP biweekly for 5 sessions; the other half was left untreated. At follow-up 30 days after the final treatment, striae distensae had improved for both patient and observer assessment scores.9 Another study performed by Suwanchinda and Nararatwanchai10 looked at the efficacy of CAP in treating keloids. They recruited 18 patients, and keloid scars were treated in halves—one half treated with CAP biweekly for 5 sessions and the other left untreated. At follow-up 30 days after the final treatment, keloids significantly improved in color, melanin, texture, and hemoglobin based on assessment by the Antera 3D imaging system (Miravex Limited)(P<.05).10
Kim et al11 studied the efficacy of CAP for the treatment of atopic dermatitis in 22 patients. Each patient had mild to moderate atopic dermatitis that had not been treated with topical agents or antibiotics for at least 2 weeks prior to beginning the study. Additionally, only patients with symmetric lesions—meaning only patients with lesions on both sides of the anatomical extremities—were included. Each patient then received CAP on 1 symmetric lesion and placebo on the other. Cold atmospheric plasma treatment was done 5 mm away from the lesion, and each treatment lasted for 5 minutes. Treatments were done at weeks 0, 1, and 2, with follow-up 4 weeks after the final treatment. The clinical severity of disease was assessed at weeks 0, 1, 2, and 4. Results showed that at week 4, the mean (SD) modified Atopic Dermatitis Antecubital Severity score decreased from 33.73 (21.21) at week 0 to 13.12 (15.92). Additionally, the pruritic visual analog scale showed significant improvement with treatment vs baseline (P≤.0001).11
Two studies examined how NTAP can be used in the treatment of psoriasis. First, Gareri et al12 used CAP to treat a psoriatic plaque in a 20-year-old woman. These plaques on the left hand previously had been unresponsive to topical psoriasis treatments. The patient received 2 treatments with CAP on days 0 and 3; at 14 days, the plaque completely resolved with an itch score of 0.12 Next, Zheng et al13 treated 2 patients with NTAP for inverse psoriasis. The first patient was a 26-year-old woman with plaques in the axilla and buttocks as well as inframammary lesions that failed to respond to treatment with topicals and vitamin D analogues. She received CAP treatments 2 to 3 times weekly for 5 total treatments with application to each region occurring 1 mm from the skin surface. The lesions completely resolved with no recurrence at 6 weeks. The second patient was a 38-year-old woman with inverse psoriasis in the axilla and groin; she received treatment every 3 days for 8 total treatments, which led to complete remission, with no recurrence noted at 1 month.13
Arisi et al14 used NTAP to treat acne vulgaris in 2 patients. The first patient was a 24-year-old man with moderate acne on the face that did not improve with topicals or oral antibiotics. The patient received 5 CAP treatments with no adverse events noted. The patient discontinued treatment on his own, but the number of lesions decreased after the fifth treatment. The second patient was a 21-year-old woman with moderate facial acne that failed to respond to treatment with topicals and oral tetracycline. The patient received 8 CAP treatments and experienced a reduction in the number of lesions during treatment. There were no adverse events, and improvement was maintained at 3-month follow-up.14
Comment
Although the use of NTAP in pediatric dermatology is scarcely described in the literature, the technology will certainly have applications in the future treatment of a wide variety of pediatric disorders. In addition to the clinical success shown in several studies,6-14 this technology has been shown to cause minimal damage to skin when application time is minimized. One study conducted on ex vivo skin showed that NTAP technology can safely be used for up to 2 minutes without major DNA damage.15 Through its diverse mechanisms of action, NTAP can induce modification of proteins and cell membranes in a noninvasive manner.2 In conditions with impaired barrier function, such as atopic and diaper dermatitis, studies in mouse models have shown improvement in lesions via upregulation of mesencephalic astrocyte-derived neurotrophic factor that contributes to decreased inflammation and cell apoptosis.16 Additionally, the generation of reactive oxygen and nitrogen species has been shown to decrease Staphylococcus aureus colonization to improve atopic dermatitis lesions in patients.11
Many other proposed benefits of NTAP in dermatologic disease also have been proposed. Nonthermal atmospheric plasma has been shown to increase messenger RNA expression of proinflammatory cytokines (IL-1, IL-6) and upregulate type III collagen production in early stages of wound healing.17 Furthermore, NTAP has been shown to stimulate nuclear factor erythroid 2–related pathways involved in antioxidant production in keratinocytes, further promoting wound healing.18 Additionally, CAP has been shown to increase expression of caspases and induce mitochondrial dysfunction that promotes cell death in different cancer cell lines.19 It is clear that the exact breadth of NTAP’s biochemical effects are unknown, but the current literature shows promise for its use in cutaneous healing and cancer treatment.
Beyond its diverse applications, treatment with NTAP yields a unique advantage to pharmacologic therapies in that there is no risk for medication interactions or risk for pharmacologic adverse effects. Cantharidin is not approved by the US Food and Drug Administration but commonly is used to treat MC. It is a blister beetle extract that causes a blister to form when applied to the skin. When orally ingested, the drug is toxic to the gastrointestinal tract and kidneys because of its phosphodiesterase inhibition, a feared complication in pediatric patients who may inadvertently ingest it during treatment.20 This utility extends beyond MC, such as the beneficial outcomes described by Suwanchinda and Nararatwanchai10 in using NTAP for keloid scars. Treatment with NTAP may replace triamcinolone injections, which are commonly associated with skin atrophy and ulceration. In addition, NTAP application to the skin has been reported to be relatively painless.5 Thus, NTAP maintains a distinct advantage over other commonly used nonpharmacologic treatment options, including curettage and cryosurgery. Curettage has widely been noted to be traumatic for the patient, may be more likely to leave a mark, and is prone to user error.20 Cryosurgery is a common form of treatment for MC because it is cost-effective and has good cosmetic results; however, it is more painful than cantharidin or anesthetized curettage.21 Treatment with NTAP is an emerging therapeutic tool with an expanding role in the treatment of dermatologic patients because it provides advantages over many standard therapies due to its minimal side-effect profile involving pain and nonpharmacologic nature.
Limitations of this report include exclusion of non–English-language articles and lack of control or comparison groups to standard therapies across studies. Additionally, reports of NTAP success occurred in many conditions that are self-limited and may have resolved on their own. Regardless, we aimed to summarize how NTAP currently is being used in pediatric populations and highlight its potential uses moving forward. Given its promising safety profile and painless nature, future clinical trials should prioritize the investigation of NTAP use in common pediatric dermatologic conditions to determine if they are equal or superior to current standards of care.
- Gan L, Zhang S, Poorun D, et al. Medical applications of nonthermal atmospheric pressure plasma in dermatology. J Dtsch Dermatol Ges. 2018;16:7-13. doi:https://doi.org/10.1111/ddg.13373
- Gay-Mimbrera J, García MC, Isla-Tejera B, et al. Clinical and biological principles of cold atmospheric plasma application in skin cancer. Adv Ther. 2016;33:894-909. doi:10.1007/s12325-016-0338-1. Published correction appears in Adv Ther. 2017;34:280. doi:10.1007/s12325-016-0437-z
- Zhai SY, Kong MG, Xia YM. Cold atmospheric plasma ameliorates skin diseases involving reactive oxygen/nitrogen species-mediated functions. Front Immunol. 2022;13:868386. doi:10.3389/fimmu.2022.868386
- Tan F, Wang Y, Zhang S, et al. Plasma dermatology: skin therapy using cold atmospheric plasma. Front Oncol. 2022;12:918484. doi:10.3389/fonc.2022.918484
- van Welzen A, Hoch M, Wahl P, et al. The response and tolerability of a novel cold atmospheric plasma wound dressing for the healing of split skin graft donor sites: a controlled pilot study. Skin Pharmacol Physiol. 2021;34:328-336. doi:10.1159/000517524
- Friedman PC, Fridman G, Fridman A. Using cold plasma to treat warts in children: a case series. Pediatr Dermatol. 2020;37:706-709. doi:10.1111/pde.14180
- Zhang C, Zhao J, Gao Y, et al. Cold atmospheric plasma treatment for diaper dermatitis: a case report [published online January 27, 2021]. Dermatol Ther. 2021;34:E14739. doi:10.1111/dth.14739
- Friedman PC, Fridman G, Fridman A. Cold atmospheric pressure plasma clears molluscum contagiosum. Exp Dermatol. 2023;32:562-563. doi:10.1111/exd.14695
- Suwanchinda A, Nararatwanchai T. The efficacy and safety of the innovative cold atmospheric-pressure plasma technology in the treatment of striae distensae: a randomized controlled trial. J Cosmet Dermatol. 2022;21:6805-6814. doi:10.1111/jocd.15458
- Suwanchinda A, Nararatwanchai T. Efficacy and safety of the innovative cold atmospheric-pressure plasma technology in the treatment of keloid: a randomized controlled trial. J Cosmet Dermatol. 2022;21:6788-6797. doi:10.1111/jocd.15397
- Kim YJ, Lim DJ, Lee MY, et al. Prospective, comparative clinical pilot study of cold atmospheric plasma device in the treatment of atopic dermatitis. Sci Rep. 2021;11:14461. doi:10.1038/s41598-021-93941-y
- Gareri C, Bennardo L, De Masi G. Use of a new cold plasma tool for psoriasis treatment: a case report. SAGE Open Med Case Rep. 2020;8:2050313X20922709. doi:10.1177/2050313X20922709
- Zheng L, Gao J, Cao Y, et al. Two case reports of inverse psoriasis treated with cold atmospheric plasma. Dermatol Ther. 2020;33:E14257. doi:10.1111/dth.14257
- Arisi M, Venturuzzo A, Gelmetti A, et al. Cold atmospheric plasma (CAP) as a promising therapeutic option for mild to moderate acne vulgaris: clinical and non-invasive evaluation of two cases. Clin Plasma Med. 2020;19-20:100110.
- Isbary G, Köritzer J, Mitra A, et al. Ex vivo human skin experiments for the evaluation of safety of new cold atmospheric plasma devices. Clin Plasma Med. 2013;1:36-44.
- Sun T, Zhang X, Hou C, et al. Cold plasma irradiation attenuates atopic dermatitis via enhancing HIF-1α-induced MANF transcription expression. Front Immunol. 2022;13:941219. doi:10.3389/fimmu.2022.941219
- Eggers B, Marciniak J, Memmert S, et al. The beneficial effect of cold atmospheric plasma on parameters of molecules and cell function involved in wound healing in human osteoblast-like cells in vitro. Odontology. 2020;108:607-616. doi:10.1007/s10266-020-00487-y
- Conway GE, He Z, Hutanu AL, et al. Cold atmospheric plasma induces accumulation of lysosomes and caspase-independent cell death in U373MG glioblastoma multiforme cells. Sci Rep. 2019;9:12891. doi:10.1038/s41598-019-49013-3
- Schmidt A, Dietrich S, Steuer A, et al. Non-thermal plasma activates human keratinocytes by stimulation of antioxidant and phase II pathways. J Biol Chem. 2015;290:6731-6750. doi:10.1074/jbc.M114.603555
- Silverberg NB. Pediatric molluscum contagiosum. Pediatr Drugs. 2003;5:505-511. doi:10.2165/00148581-200305080-00001
- Cotton DW, Cooper C, Barrett DF, et al. Severe atypical molluscum contagiosum infection in an immunocompromised host. Br J Dermatol. 1987;116:871-876. doi:10.1111/j.1365-2133.1987.tb04908.x
Nonthermal atmospheric plasma (NTAP)(or cold atmospheric plasma [CAP]) is a rapidly developing treatment modality for a wide range of dermatologic conditions. Plasma (or ionized gas) refers to a state of matter composed of electrons, protons, and neutral atoms that generate reactive oxygen and nitrogen species.1 Plasma previously was created using thermal energy, but recent advances have allowed the creation of plasma using atmospheric pressure and room temperature; thus, NTAP can be used without causing damage to living tissue through heat.1 Plasma technology varies greatly, but it generally can be classified as either direct or indirect therapy; direct therapy uses the human body as an electrode, whereas indirect therapy creates plasma through the interaction between 2 electrode devices.1,2 When used on the skin, important dose-dependent relationships have been observed, with CAP application longer than 2 minutes being associated with increased keratinocyte and fibroblast apoptosis.2 Thus, CAP can cause diverse changes to the skin depending on application time and methodology. At adequate yet low concentrations, plasma can promote fibroblast proliferation and upregulate genes involved in collagen and transforming growth factor synthesis.1 Additionally, the reactive oxygen and nitrogen species created by NTAP have been shown to inactivate microorganisms through the destruction of biofilms, lead to diminished immune cell infiltration and cytokine release in autoimmune dermatologic conditions, and exert antitumor properties through cellular DNA damage.1-3 In dermatology, these properties can be harvested to promote wound healing at low doses and the treatment of proliferative skin conditions at high doses.1
Because of its novelty, the safety profile of NTAP is still under investigation, but preliminary studies are promising and show no damage to the skin barrier when excessive plasma exposure is avoided.4 However, dose- and time-dependent damage to cells has been shown. As a result, the exact dose of plasma considered safe is highly variable depending on the vessel, technique, and user, and future clinical research is needed to guide this methodology.4 Additionally, CAP has been shown to cause little pain at the skin surface and may lead to decreased levels of pain in healing wound sites.5 Given this promising safety profile and minimal discomfort to patients, NTAP technology remains promising for use in pediatric dermatology, but there are limited data to characterize its potential use in this population. In this systematic review, we aimed to elucidate reported applications of NTAP for skin conditions in children and discuss the trajectory of this technology in the future of pediatric dermatology.
Methodology
A comprehensive literature review was conducted to identify studies evaluating NTAP technology in pediatric populations using PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) guidelines. A search of PubMed, Embase, and Web of Science articles was conducted in April 2023 using the terms nonthermal atmospheric plasma or cold atmospheric plasma. All English-language articles that described the use of NTAP as a treatment in pediatric populations or articles that described NTAP use in the treatment of common conditions in this patient group were included based on a review of the article titles and abstracts by 2 independent reviewers, followed by full-text review of relevant articles (M.G., C.L.). Any discrepancies in eligible articles were settled by a third independent researcher (M.V.). One hundred twenty studies were identified, and 95 were screened for inclusion; 9 studies met inclusion criteria and were summarized in this review.
Results
A total of 9 studies were included in this review: 3 describing the success of NTAP in pediatric populations6-8 and 6 describing the potential success of NTAP for dermatologic conditions commonly seen in children (Table).9-14

Studies Describing Success of NTAP—Three clinical reports described the efficacy of NTAP in pediatric dermatology. A case series from 2020 showed full clearance of warts in 100% of patients (n=5) with a 0% recurrence rate when NTAP treatment was applied for 2 minutes to each lesion during each treatment session with the electrode held 1 mm from the lesional surface.6 Each patient was followed up at 3 to 4 weeks, and treatment was repeated if lesions persisted. Patients reported no pain during the procedure, and no adverse effects were noted over the course of treatment.6 Second, a case report described full clearance of diaper dermatitis with no recurrence after 6 months following 6 treatments with NTAP in a 14-month-old girl.7 After treatment with econazole nitrate cream, oral antibiotics, and prednisone failed, CAP treatment was initiated. Each treatment lasted 15 minutes with 3-day time intervals between each of the 6 treatments. There were no adverse events or recurrence of rash at 6-month follow-up.7 A final case report described full clearance of molluscum contagiosum (MC), with no recurrence after 2 months following 4 treatments with NTAP in a 12-year-old boy.8 The patient had untreated MC on the face, neck, shoulder, and thighs. Lesions of the face were treated with CAP, while the other sites were treated with cantharidin using a 0.7% collodion-based solution. Four CAP treatments were performed at 1-month intervals, with CAP applied 1 mm from the lesional surfaces in a circular pattern for 2 minutes. At follow-up 2 months after the final treatment, the patient had no adverse effects and showed no pigmentary changes or scarring.8
Studies Describing the Potential Success of NTAP—Beyond these studies, limited research has been done on NTAP in pediatric populations. The Table summarizes 6 additional studies completed with promising treatment results for dermatologic conditions commonly seen in children: striae distensae, keloids, atopic dermatitis, psoriasis, inverse psoriasis, and acne vulgaris. Across all reports and studies, patients showed significant improvement in their dermatologic conditions following the use of NTAP technology with limited adverse effects reported (P<.05). Suwanchinda and Nararatwanchai9 studied the use of CAP for the treatment of striae distensae. They recruited 23 patients and treated half the body with CAP biweekly for 5 sessions; the other half was left untreated. At follow-up 30 days after the final treatment, striae distensae had improved for both patient and observer assessment scores.9 Another study performed by Suwanchinda and Nararatwanchai10 looked at the efficacy of CAP in treating keloids. They recruited 18 patients, and keloid scars were treated in halves—one half treated with CAP biweekly for 5 sessions and the other left untreated. At follow-up 30 days after the final treatment, keloids significantly improved in color, melanin, texture, and hemoglobin based on assessment by the Antera 3D imaging system (Miravex Limited)(P<.05).10
Kim et al11 studied the efficacy of CAP for the treatment of atopic dermatitis in 22 patients. Each patient had mild to moderate atopic dermatitis that had not been treated with topical agents or antibiotics for at least 2 weeks prior to beginning the study. Additionally, only patients with symmetric lesions—meaning only patients with lesions on both sides of the anatomical extremities—were included. Each patient then received CAP on 1 symmetric lesion and placebo on the other. Cold atmospheric plasma treatment was done 5 mm away from the lesion, and each treatment lasted for 5 minutes. Treatments were done at weeks 0, 1, and 2, with follow-up 4 weeks after the final treatment. The clinical severity of disease was assessed at weeks 0, 1, 2, and 4. Results showed that at week 4, the mean (SD) modified Atopic Dermatitis Antecubital Severity score decreased from 33.73 (21.21) at week 0 to 13.12 (15.92). Additionally, the pruritic visual analog scale showed significant improvement with treatment vs baseline (P≤.0001).11
Two studies examined how NTAP can be used in the treatment of psoriasis. First, Gareri et al12 used CAP to treat a psoriatic plaque in a 20-year-old woman. These plaques on the left hand previously had been unresponsive to topical psoriasis treatments. The patient received 2 treatments with CAP on days 0 and 3; at 14 days, the plaque completely resolved with an itch score of 0.12 Next, Zheng et al13 treated 2 patients with NTAP for inverse psoriasis. The first patient was a 26-year-old woman with plaques in the axilla and buttocks as well as inframammary lesions that failed to respond to treatment with topicals and vitamin D analogues. She received CAP treatments 2 to 3 times weekly for 5 total treatments with application to each region occurring 1 mm from the skin surface. The lesions completely resolved with no recurrence at 6 weeks. The second patient was a 38-year-old woman with inverse psoriasis in the axilla and groin; she received treatment every 3 days for 8 total treatments, which led to complete remission, with no recurrence noted at 1 month.13
Arisi et al14 used NTAP to treat acne vulgaris in 2 patients. The first patient was a 24-year-old man with moderate acne on the face that did not improve with topicals or oral antibiotics. The patient received 5 CAP treatments with no adverse events noted. The patient discontinued treatment on his own, but the number of lesions decreased after the fifth treatment. The second patient was a 21-year-old woman with moderate facial acne that failed to respond to treatment with topicals and oral tetracycline. The patient received 8 CAP treatments and experienced a reduction in the number of lesions during treatment. There were no adverse events, and improvement was maintained at 3-month follow-up.14
Comment
Although the use of NTAP in pediatric dermatology is scarcely described in the literature, the technology will certainly have applications in the future treatment of a wide variety of pediatric disorders. In addition to the clinical success shown in several studies,6-14 this technology has been shown to cause minimal damage to skin when application time is minimized. One study conducted on ex vivo skin showed that NTAP technology can safely be used for up to 2 minutes without major DNA damage.15 Through its diverse mechanisms of action, NTAP can induce modification of proteins and cell membranes in a noninvasive manner.2 In conditions with impaired barrier function, such as atopic and diaper dermatitis, studies in mouse models have shown improvement in lesions via upregulation of mesencephalic astrocyte-derived neurotrophic factor that contributes to decreased inflammation and cell apoptosis.16 Additionally, the generation of reactive oxygen and nitrogen species has been shown to decrease Staphylococcus aureus colonization to improve atopic dermatitis lesions in patients.11
Many other proposed benefits of NTAP in dermatologic disease also have been proposed. Nonthermal atmospheric plasma has been shown to increase messenger RNA expression of proinflammatory cytokines (IL-1, IL-6) and upregulate type III collagen production in early stages of wound healing.17 Furthermore, NTAP has been shown to stimulate nuclear factor erythroid 2–related pathways involved in antioxidant production in keratinocytes, further promoting wound healing.18 Additionally, CAP has been shown to increase expression of caspases and induce mitochondrial dysfunction that promotes cell death in different cancer cell lines.19 It is clear that the exact breadth of NTAP’s biochemical effects are unknown, but the current literature shows promise for its use in cutaneous healing and cancer treatment.
Beyond its diverse applications, treatment with NTAP yields a unique advantage to pharmacologic therapies in that there is no risk for medication interactions or risk for pharmacologic adverse effects. Cantharidin is not approved by the US Food and Drug Administration but commonly is used to treat MC. It is a blister beetle extract that causes a blister to form when applied to the skin. When orally ingested, the drug is toxic to the gastrointestinal tract and kidneys because of its phosphodiesterase inhibition, a feared complication in pediatric patients who may inadvertently ingest it during treatment.20 This utility extends beyond MC, such as the beneficial outcomes described by Suwanchinda and Nararatwanchai10 in using NTAP for keloid scars. Treatment with NTAP may replace triamcinolone injections, which are commonly associated with skin atrophy and ulceration. In addition, NTAP application to the skin has been reported to be relatively painless.5 Thus, NTAP maintains a distinct advantage over other commonly used nonpharmacologic treatment options, including curettage and cryosurgery. Curettage has widely been noted to be traumatic for the patient, may be more likely to leave a mark, and is prone to user error.20 Cryosurgery is a common form of treatment for MC because it is cost-effective and has good cosmetic results; however, it is more painful than cantharidin or anesthetized curettage.21 Treatment with NTAP is an emerging therapeutic tool with an expanding role in the treatment of dermatologic patients because it provides advantages over many standard therapies due to its minimal side-effect profile involving pain and nonpharmacologic nature.
Limitations of this report include exclusion of non–English-language articles and lack of control or comparison groups to standard therapies across studies. Additionally, reports of NTAP success occurred in many conditions that are self-limited and may have resolved on their own. Regardless, we aimed to summarize how NTAP currently is being used in pediatric populations and highlight its potential uses moving forward. Given its promising safety profile and painless nature, future clinical trials should prioritize the investigation of NTAP use in common pediatric dermatologic conditions to determine if they are equal or superior to current standards of care.
Nonthermal atmospheric plasma (NTAP)(or cold atmospheric plasma [CAP]) is a rapidly developing treatment modality for a wide range of dermatologic conditions. Plasma (or ionized gas) refers to a state of matter composed of electrons, protons, and neutral atoms that generate reactive oxygen and nitrogen species.1 Plasma previously was created using thermal energy, but recent advances have allowed the creation of plasma using atmospheric pressure and room temperature; thus, NTAP can be used without causing damage to living tissue through heat.1 Plasma technology varies greatly, but it generally can be classified as either direct or indirect therapy; direct therapy uses the human body as an electrode, whereas indirect therapy creates plasma through the interaction between 2 electrode devices.1,2 When used on the skin, important dose-dependent relationships have been observed, with CAP application longer than 2 minutes being associated with increased keratinocyte and fibroblast apoptosis.2 Thus, CAP can cause diverse changes to the skin depending on application time and methodology. At adequate yet low concentrations, plasma can promote fibroblast proliferation and upregulate genes involved in collagen and transforming growth factor synthesis.1 Additionally, the reactive oxygen and nitrogen species created by NTAP have been shown to inactivate microorganisms through the destruction of biofilms, lead to diminished immune cell infiltration and cytokine release in autoimmune dermatologic conditions, and exert antitumor properties through cellular DNA damage.1-3 In dermatology, these properties can be harvested to promote wound healing at low doses and the treatment of proliferative skin conditions at high doses.1
Because of its novelty, the safety profile of NTAP is still under investigation, but preliminary studies are promising and show no damage to the skin barrier when excessive plasma exposure is avoided.4 However, dose- and time-dependent damage to cells has been shown. As a result, the exact dose of plasma considered safe is highly variable depending on the vessel, technique, and user, and future clinical research is needed to guide this methodology.4 Additionally, CAP has been shown to cause little pain at the skin surface and may lead to decreased levels of pain in healing wound sites.5 Given this promising safety profile and minimal discomfort to patients, NTAP technology remains promising for use in pediatric dermatology, but there are limited data to characterize its potential use in this population. In this systematic review, we aimed to elucidate reported applications of NTAP for skin conditions in children and discuss the trajectory of this technology in the future of pediatric dermatology.
Methodology
A comprehensive literature review was conducted to identify studies evaluating NTAP technology in pediatric populations using PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) guidelines. A search of PubMed, Embase, and Web of Science articles was conducted in April 2023 using the terms nonthermal atmospheric plasma or cold atmospheric plasma. All English-language articles that described the use of NTAP as a treatment in pediatric populations or articles that described NTAP use in the treatment of common conditions in this patient group were included based on a review of the article titles and abstracts by 2 independent reviewers, followed by full-text review of relevant articles (M.G., C.L.). Any discrepancies in eligible articles were settled by a third independent researcher (M.V.). One hundred twenty studies were identified, and 95 were screened for inclusion; 9 studies met inclusion criteria and were summarized in this review.
Results
A total of 9 studies were included in this review: 3 describing the success of NTAP in pediatric populations6-8 and 6 describing the potential success of NTAP for dermatologic conditions commonly seen in children (Table).9-14

Studies Describing Success of NTAP—Three clinical reports described the efficacy of NTAP in pediatric dermatology. A case series from 2020 showed full clearance of warts in 100% of patients (n=5) with a 0% recurrence rate when NTAP treatment was applied for 2 minutes to each lesion during each treatment session with the electrode held 1 mm from the lesional surface.6 Each patient was followed up at 3 to 4 weeks, and treatment was repeated if lesions persisted. Patients reported no pain during the procedure, and no adverse effects were noted over the course of treatment.6 Second, a case report described full clearance of diaper dermatitis with no recurrence after 6 months following 6 treatments with NTAP in a 14-month-old girl.7 After treatment with econazole nitrate cream, oral antibiotics, and prednisone failed, CAP treatment was initiated. Each treatment lasted 15 minutes with 3-day time intervals between each of the 6 treatments. There were no adverse events or recurrence of rash at 6-month follow-up.7 A final case report described full clearance of molluscum contagiosum (MC), with no recurrence after 2 months following 4 treatments with NTAP in a 12-year-old boy.8 The patient had untreated MC on the face, neck, shoulder, and thighs. Lesions of the face were treated with CAP, while the other sites were treated with cantharidin using a 0.7% collodion-based solution. Four CAP treatments were performed at 1-month intervals, with CAP applied 1 mm from the lesional surfaces in a circular pattern for 2 minutes. At follow-up 2 months after the final treatment, the patient had no adverse effects and showed no pigmentary changes or scarring.8
Studies Describing the Potential Success of NTAP—Beyond these studies, limited research has been done on NTAP in pediatric populations. The Table summarizes 6 additional studies completed with promising treatment results for dermatologic conditions commonly seen in children: striae distensae, keloids, atopic dermatitis, psoriasis, inverse psoriasis, and acne vulgaris. Across all reports and studies, patients showed significant improvement in their dermatologic conditions following the use of NTAP technology with limited adverse effects reported (P<.05). Suwanchinda and Nararatwanchai9 studied the use of CAP for the treatment of striae distensae. They recruited 23 patients and treated half the body with CAP biweekly for 5 sessions; the other half was left untreated. At follow-up 30 days after the final treatment, striae distensae had improved for both patient and observer assessment scores.9 Another study performed by Suwanchinda and Nararatwanchai10 looked at the efficacy of CAP in treating keloids. They recruited 18 patients, and keloid scars were treated in halves—one half treated with CAP biweekly for 5 sessions and the other left untreated. At follow-up 30 days after the final treatment, keloids significantly improved in color, melanin, texture, and hemoglobin based on assessment by the Antera 3D imaging system (Miravex Limited)(P<.05).10
Kim et al11 studied the efficacy of CAP for the treatment of atopic dermatitis in 22 patients. Each patient had mild to moderate atopic dermatitis that had not been treated with topical agents or antibiotics for at least 2 weeks prior to beginning the study. Additionally, only patients with symmetric lesions—meaning only patients with lesions on both sides of the anatomical extremities—were included. Each patient then received CAP on 1 symmetric lesion and placebo on the other. Cold atmospheric plasma treatment was done 5 mm away from the lesion, and each treatment lasted for 5 minutes. Treatments were done at weeks 0, 1, and 2, with follow-up 4 weeks after the final treatment. The clinical severity of disease was assessed at weeks 0, 1, 2, and 4. Results showed that at week 4, the mean (SD) modified Atopic Dermatitis Antecubital Severity score decreased from 33.73 (21.21) at week 0 to 13.12 (15.92). Additionally, the pruritic visual analog scale showed significant improvement with treatment vs baseline (P≤.0001).11
Two studies examined how NTAP can be used in the treatment of psoriasis. First, Gareri et al12 used CAP to treat a psoriatic plaque in a 20-year-old woman. These plaques on the left hand previously had been unresponsive to topical psoriasis treatments. The patient received 2 treatments with CAP on days 0 and 3; at 14 days, the plaque completely resolved with an itch score of 0.12 Next, Zheng et al13 treated 2 patients with NTAP for inverse psoriasis. The first patient was a 26-year-old woman with plaques in the axilla and buttocks as well as inframammary lesions that failed to respond to treatment with topicals and vitamin D analogues. She received CAP treatments 2 to 3 times weekly for 5 total treatments with application to each region occurring 1 mm from the skin surface. The lesions completely resolved with no recurrence at 6 weeks. The second patient was a 38-year-old woman with inverse psoriasis in the axilla and groin; she received treatment every 3 days for 8 total treatments, which led to complete remission, with no recurrence noted at 1 month.13
Arisi et al14 used NTAP to treat acne vulgaris in 2 patients. The first patient was a 24-year-old man with moderate acne on the face that did not improve with topicals or oral antibiotics. The patient received 5 CAP treatments with no adverse events noted. The patient discontinued treatment on his own, but the number of lesions decreased after the fifth treatment. The second patient was a 21-year-old woman with moderate facial acne that failed to respond to treatment with topicals and oral tetracycline. The patient received 8 CAP treatments and experienced a reduction in the number of lesions during treatment. There were no adverse events, and improvement was maintained at 3-month follow-up.14
Comment
Although the use of NTAP in pediatric dermatology is scarcely described in the literature, the technology will certainly have applications in the future treatment of a wide variety of pediatric disorders. In addition to the clinical success shown in several studies,6-14 this technology has been shown to cause minimal damage to skin when application time is minimized. One study conducted on ex vivo skin showed that NTAP technology can safely be used for up to 2 minutes without major DNA damage.15 Through its diverse mechanisms of action, NTAP can induce modification of proteins and cell membranes in a noninvasive manner.2 In conditions with impaired barrier function, such as atopic and diaper dermatitis, studies in mouse models have shown improvement in lesions via upregulation of mesencephalic astrocyte-derived neurotrophic factor that contributes to decreased inflammation and cell apoptosis.16 Additionally, the generation of reactive oxygen and nitrogen species has been shown to decrease Staphylococcus aureus colonization to improve atopic dermatitis lesions in patients.11
Many other proposed benefits of NTAP in dermatologic disease also have been proposed. Nonthermal atmospheric plasma has been shown to increase messenger RNA expression of proinflammatory cytokines (IL-1, IL-6) and upregulate type III collagen production in early stages of wound healing.17 Furthermore, NTAP has been shown to stimulate nuclear factor erythroid 2–related pathways involved in antioxidant production in keratinocytes, further promoting wound healing.18 Additionally, CAP has been shown to increase expression of caspases and induce mitochondrial dysfunction that promotes cell death in different cancer cell lines.19 It is clear that the exact breadth of NTAP’s biochemical effects are unknown, but the current literature shows promise for its use in cutaneous healing and cancer treatment.
Beyond its diverse applications, treatment with NTAP yields a unique advantage to pharmacologic therapies in that there is no risk for medication interactions or risk for pharmacologic adverse effects. Cantharidin is not approved by the US Food and Drug Administration but commonly is used to treat MC. It is a blister beetle extract that causes a blister to form when applied to the skin. When orally ingested, the drug is toxic to the gastrointestinal tract and kidneys because of its phosphodiesterase inhibition, a feared complication in pediatric patients who may inadvertently ingest it during treatment.20 This utility extends beyond MC, such as the beneficial outcomes described by Suwanchinda and Nararatwanchai10 in using NTAP for keloid scars. Treatment with NTAP may replace triamcinolone injections, which are commonly associated with skin atrophy and ulceration. In addition, NTAP application to the skin has been reported to be relatively painless.5 Thus, NTAP maintains a distinct advantage over other commonly used nonpharmacologic treatment options, including curettage and cryosurgery. Curettage has widely been noted to be traumatic for the patient, may be more likely to leave a mark, and is prone to user error.20 Cryosurgery is a common form of treatment for MC because it is cost-effective and has good cosmetic results; however, it is more painful than cantharidin or anesthetized curettage.21 Treatment with NTAP is an emerging therapeutic tool with an expanding role in the treatment of dermatologic patients because it provides advantages over many standard therapies due to its minimal side-effect profile involving pain and nonpharmacologic nature.
Limitations of this report include exclusion of non–English-language articles and lack of control or comparison groups to standard therapies across studies. Additionally, reports of NTAP success occurred in many conditions that are self-limited and may have resolved on their own. Regardless, we aimed to summarize how NTAP currently is being used in pediatric populations and highlight its potential uses moving forward. Given its promising safety profile and painless nature, future clinical trials should prioritize the investigation of NTAP use in common pediatric dermatologic conditions to determine if they are equal or superior to current standards of care.
- Gan L, Zhang S, Poorun D, et al. Medical applications of nonthermal atmospheric pressure plasma in dermatology. J Dtsch Dermatol Ges. 2018;16:7-13. doi:https://doi.org/10.1111/ddg.13373
- Gay-Mimbrera J, García MC, Isla-Tejera B, et al. Clinical and biological principles of cold atmospheric plasma application in skin cancer. Adv Ther. 2016;33:894-909. doi:10.1007/s12325-016-0338-1. Published correction appears in Adv Ther. 2017;34:280. doi:10.1007/s12325-016-0437-z
- Zhai SY, Kong MG, Xia YM. Cold atmospheric plasma ameliorates skin diseases involving reactive oxygen/nitrogen species-mediated functions. Front Immunol. 2022;13:868386. doi:10.3389/fimmu.2022.868386
- Tan F, Wang Y, Zhang S, et al. Plasma dermatology: skin therapy using cold atmospheric plasma. Front Oncol. 2022;12:918484. doi:10.3389/fonc.2022.918484
- van Welzen A, Hoch M, Wahl P, et al. The response and tolerability of a novel cold atmospheric plasma wound dressing for the healing of split skin graft donor sites: a controlled pilot study. Skin Pharmacol Physiol. 2021;34:328-336. doi:10.1159/000517524
- Friedman PC, Fridman G, Fridman A. Using cold plasma to treat warts in children: a case series. Pediatr Dermatol. 2020;37:706-709. doi:10.1111/pde.14180
- Zhang C, Zhao J, Gao Y, et al. Cold atmospheric plasma treatment for diaper dermatitis: a case report [published online January 27, 2021]. Dermatol Ther. 2021;34:E14739. doi:10.1111/dth.14739
- Friedman PC, Fridman G, Fridman A. Cold atmospheric pressure plasma clears molluscum contagiosum. Exp Dermatol. 2023;32:562-563. doi:10.1111/exd.14695
- Suwanchinda A, Nararatwanchai T. The efficacy and safety of the innovative cold atmospheric-pressure plasma technology in the treatment of striae distensae: a randomized controlled trial. J Cosmet Dermatol. 2022;21:6805-6814. doi:10.1111/jocd.15458
- Suwanchinda A, Nararatwanchai T. Efficacy and safety of the innovative cold atmospheric-pressure plasma technology in the treatment of keloid: a randomized controlled trial. J Cosmet Dermatol. 2022;21:6788-6797. doi:10.1111/jocd.15397
- Kim YJ, Lim DJ, Lee MY, et al. Prospective, comparative clinical pilot study of cold atmospheric plasma device in the treatment of atopic dermatitis. Sci Rep. 2021;11:14461. doi:10.1038/s41598-021-93941-y
- Gareri C, Bennardo L, De Masi G. Use of a new cold plasma tool for psoriasis treatment: a case report. SAGE Open Med Case Rep. 2020;8:2050313X20922709. doi:10.1177/2050313X20922709
- Zheng L, Gao J, Cao Y, et al. Two case reports of inverse psoriasis treated with cold atmospheric plasma. Dermatol Ther. 2020;33:E14257. doi:10.1111/dth.14257
- Arisi M, Venturuzzo A, Gelmetti A, et al. Cold atmospheric plasma (CAP) as a promising therapeutic option for mild to moderate acne vulgaris: clinical and non-invasive evaluation of two cases. Clin Plasma Med. 2020;19-20:100110.
- Isbary G, Köritzer J, Mitra A, et al. Ex vivo human skin experiments for the evaluation of safety of new cold atmospheric plasma devices. Clin Plasma Med. 2013;1:36-44.
- Sun T, Zhang X, Hou C, et al. Cold plasma irradiation attenuates atopic dermatitis via enhancing HIF-1α-induced MANF transcription expression. Front Immunol. 2022;13:941219. doi:10.3389/fimmu.2022.941219
- Eggers B, Marciniak J, Memmert S, et al. The beneficial effect of cold atmospheric plasma on parameters of molecules and cell function involved in wound healing in human osteoblast-like cells in vitro. Odontology. 2020;108:607-616. doi:10.1007/s10266-020-00487-y
- Conway GE, He Z, Hutanu AL, et al. Cold atmospheric plasma induces accumulation of lysosomes and caspase-independent cell death in U373MG glioblastoma multiforme cells. Sci Rep. 2019;9:12891. doi:10.1038/s41598-019-49013-3
- Schmidt A, Dietrich S, Steuer A, et al. Non-thermal plasma activates human keratinocytes by stimulation of antioxidant and phase II pathways. J Biol Chem. 2015;290:6731-6750. doi:10.1074/jbc.M114.603555
- Silverberg NB. Pediatric molluscum contagiosum. Pediatr Drugs. 2003;5:505-511. doi:10.2165/00148581-200305080-00001
- Cotton DW, Cooper C, Barrett DF, et al. Severe atypical molluscum contagiosum infection in an immunocompromised host. Br J Dermatol. 1987;116:871-876. doi:10.1111/j.1365-2133.1987.tb04908.x
- Gan L, Zhang S, Poorun D, et al. Medical applications of nonthermal atmospheric pressure plasma in dermatology. J Dtsch Dermatol Ges. 2018;16:7-13. doi:https://doi.org/10.1111/ddg.13373
- Gay-Mimbrera J, García MC, Isla-Tejera B, et al. Clinical and biological principles of cold atmospheric plasma application in skin cancer. Adv Ther. 2016;33:894-909. doi:10.1007/s12325-016-0338-1. Published correction appears in Adv Ther. 2017;34:280. doi:10.1007/s12325-016-0437-z
- Zhai SY, Kong MG, Xia YM. Cold atmospheric plasma ameliorates skin diseases involving reactive oxygen/nitrogen species-mediated functions. Front Immunol. 2022;13:868386. doi:10.3389/fimmu.2022.868386
- Tan F, Wang Y, Zhang S, et al. Plasma dermatology: skin therapy using cold atmospheric plasma. Front Oncol. 2022;12:918484. doi:10.3389/fonc.2022.918484
- van Welzen A, Hoch M, Wahl P, et al. The response and tolerability of a novel cold atmospheric plasma wound dressing for the healing of split skin graft donor sites: a controlled pilot study. Skin Pharmacol Physiol. 2021;34:328-336. doi:10.1159/000517524
- Friedman PC, Fridman G, Fridman A. Using cold plasma to treat warts in children: a case series. Pediatr Dermatol. 2020;37:706-709. doi:10.1111/pde.14180
- Zhang C, Zhao J, Gao Y, et al. Cold atmospheric plasma treatment for diaper dermatitis: a case report [published online January 27, 2021]. Dermatol Ther. 2021;34:E14739. doi:10.1111/dth.14739
- Friedman PC, Fridman G, Fridman A. Cold atmospheric pressure plasma clears molluscum contagiosum. Exp Dermatol. 2023;32:562-563. doi:10.1111/exd.14695
- Suwanchinda A, Nararatwanchai T. The efficacy and safety of the innovative cold atmospheric-pressure plasma technology in the treatment of striae distensae: a randomized controlled trial. J Cosmet Dermatol. 2022;21:6805-6814. doi:10.1111/jocd.15458
- Suwanchinda A, Nararatwanchai T. Efficacy and safety of the innovative cold atmospheric-pressure plasma technology in the treatment of keloid: a randomized controlled trial. J Cosmet Dermatol. 2022;21:6788-6797. doi:10.1111/jocd.15397
- Kim YJ, Lim DJ, Lee MY, et al. Prospective, comparative clinical pilot study of cold atmospheric plasma device in the treatment of atopic dermatitis. Sci Rep. 2021;11:14461. doi:10.1038/s41598-021-93941-y
- Gareri C, Bennardo L, De Masi G. Use of a new cold plasma tool for psoriasis treatment: a case report. SAGE Open Med Case Rep. 2020;8:2050313X20922709. doi:10.1177/2050313X20922709
- Zheng L, Gao J, Cao Y, et al. Two case reports of inverse psoriasis treated with cold atmospheric plasma. Dermatol Ther. 2020;33:E14257. doi:10.1111/dth.14257
- Arisi M, Venturuzzo A, Gelmetti A, et al. Cold atmospheric plasma (CAP) as a promising therapeutic option for mild to moderate acne vulgaris: clinical and non-invasive evaluation of two cases. Clin Plasma Med. 2020;19-20:100110.
- Isbary G, Köritzer J, Mitra A, et al. Ex vivo human skin experiments for the evaluation of safety of new cold atmospheric plasma devices. Clin Plasma Med. 2013;1:36-44.
- Sun T, Zhang X, Hou C, et al. Cold plasma irradiation attenuates atopic dermatitis via enhancing HIF-1α-induced MANF transcription expression. Front Immunol. 2022;13:941219. doi:10.3389/fimmu.2022.941219
- Eggers B, Marciniak J, Memmert S, et al. The beneficial effect of cold atmospheric plasma on parameters of molecules and cell function involved in wound healing in human osteoblast-like cells in vitro. Odontology. 2020;108:607-616. doi:10.1007/s10266-020-00487-y
- Conway GE, He Z, Hutanu AL, et al. Cold atmospheric plasma induces accumulation of lysosomes and caspase-independent cell death in U373MG glioblastoma multiforme cells. Sci Rep. 2019;9:12891. doi:10.1038/s41598-019-49013-3
- Schmidt A, Dietrich S, Steuer A, et al. Non-thermal plasma activates human keratinocytes by stimulation of antioxidant and phase II pathways. J Biol Chem. 2015;290:6731-6750. doi:10.1074/jbc.M114.603555
- Silverberg NB. Pediatric molluscum contagiosum. Pediatr Drugs. 2003;5:505-511. doi:10.2165/00148581-200305080-00001
- Cotton DW, Cooper C, Barrett DF, et al. Severe atypical molluscum contagiosum infection in an immunocompromised host. Br J Dermatol. 1987;116:871-876. doi:10.1111/j.1365-2133.1987.tb04908.x
Practice Points
- Nonthermal atmospheric plasma (NTAP)(also known as cold atmospheric plasma) has been shown to cause minimal damage to skin when application time is minimized.
- Beyond its diverse applications, treatment with NTAP yields a unique advantage to pharmacologic therapies in that there is no risk for medication interactions or pharmacologic adverse effects.
- Although the use of NTAP in pediatric dermatology is scarcely described in the literature, the technology will certainly have applications in the future treatment of a wide variety of pediatric disorders.
Psoriatic disease: Researchers seek a PsA diagnostic test, phenotype-targeted treatment
WASHINGTON – In psoriatic disease, psoriatic arthritis (PsA) remains one of the greatest unmet needs, with the transition from cutaneous psoriasis poorly understood, diagnosis challenging, and therapeutic accomplishments trailing far behind advances for skin disease. However, leading researchers in rheumatology and dermatology believe that they’re turning the corner toward a day when therapies are phenotype-targeted and diagnosis can be made early and treatment begun well before inflammation worsens and pain and joint damage ensue.
“The challenge right now is that we don’t understand the discrete and overlapping endotypes that underlie the phenotypes or domains” of PsA, said Christopher Ritchlin, MD, MPH, professor of medicine in the division of allergy/immunology and rheumatology and the Center of Musculoskeletal Research at the University of Rochester (N.Y.), who spoke about PsA at the annual research symposium of the National Psoriasis Foundation.
“We know that mechanistically, there is dominance of the IL [interleukin]-23 and IL-17 pathways, as well as TNF [tumor necrosis factor], but we think there are tissue-specific cellular interactions [and] other pathways and mechanisms to be defined, and the goal now is to go into the tissues to find out,” he said at the meeting.
Dr. Ritchlin is co-leading a new research team dedicated to psoriatic spectrum diseases as part of the $64.5 million Autoimmune and Immune-Mediated Diseases Program (AIM) of the Accelerating Medicine Partnership (AMP), a huge public-private partnership involving the National Institutes of Health that is collecting and analyzing troves of biological data in order to better understand the cellular and molecular compositions and interactions that lead to disease.
As part of its work, this eight-center project – coined ELLIPSS, for Elucidating the Landscape of Immunoendotypes in Psoriatic Skin and Synovium – hopes to define at a molecular and single-cell level how the transition to PsA unfolds in the setting of psoriasis. Up to 30% of patients with cutaneous psoriasis also develop PsA.
The NPF, meanwhile, has invested over $3 million for research and development and validation of a diagnostic test for PsA – one that could potentially be used by dermatologists and primary care physicians to decrease the time to diagnosis. Researchers like Jose U. Scher, MD, director of the Psoriatic Arthritis Center at New York University and the NYU Colton Center for Autoimmunity, are in the thick of using multiple “-omics” tools and other sophisticated technologies to identify new targets and biomarkers.
As this work unfolds over the next several years, there is growing interest in combination therapy for PsA, Dr. Scher and Dr. Ritchlin said, and in addressing extra-articular traits, such as obesity and centralized pain, that are believed to have an impact on disease and on response to treatment.
A deep dive into the tissue
Dr. Ritchlin is among those rheumatology clinician-researchers who advocated early on for a “domain” approach to the diagnosis and management of PsA – that is, consideration of the key domains of peripheral arthritis, axial disease, enthesitis, dactylitis, and skin and nail psoriasis.
The approach is an especially important part of treatment recommendations from the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. But while interventions can be tailored to some extent to these domains, or phenotypes, there are limitations without an understanding of the different pathophysiology and mechanisms driving the heterogeneity in tissue involvement.
Dr. Ritchlin draws inspiration from pulmonology, which subtyped asthma into various phenotypes (for example, eosinophilic, allergic, intrinsic, exercise-induced) and “drilled down” on understanding underlying mechanisms to guide more specific treatment. Similar phenotype-endotype research has been done for chronic obstructive pulmonary disease, he said at the meeting, pointing to a phase 3 randomized controlled trial, published in the New England Journal of Medicine, that found dupilimab (Dupixent) was effective for patients with COPD who had type 2 inflammation as indicated by elevated eosinophil counts.
“It’s a beautiful example of how to define an endotype from a phenotypic biomarker and then use a specific intervention to improve outcomes,” Dr. Ritchlin said. “We need to do this for psoriasis and PsA.”
The ELLIPSS project will utilize the host of -omics tools and technologies (for proteomics, metabolomics, and genomics, for instance) that are making it increasingly possible to dissect the heterogeneity of single diseases and achieve more precision with treatments.
Researchers will collect blood samples and skin and/or synovial tissue biopsies from cohorts of patients with psoriasis and PsA who are treatment naïve as well as patients who are treated with a biologic or DMARD (looking at responders and non-responders). They’ll also study a cohort of psoriasis patients who may be “on a transition pathway” for PsA based on risk factors such as family history, nail psoriasis, scalp psoriasis, and body surface area greater than 5%.
Patients in all cohorts will represent distinct synovio-entheseal domains of PsA and the heterogeneity of psoriasis (for example, plaque, general, pustular, palmoplantar) and will be followed longitudinally.
With regards to PsA, one goal is to “find new pathways in the joint, then find surrogate markers in the blood that we can use to help mark particular subphenotypes [that will be identified through deep phenotyping],” Dr. Ritchlin said in an interview after the meeting. “This will lead us hopefully to a more precision-based approach.”
The ELLIPSS team joins other researchers who have been studying rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE) in an earlier iteration of the AIM program, and that will continue this work. Research on RA has thus far elucidated T-cell subsets in the rheumatoid synovium, as well as interactions of mesenchymal cells with the endothelium, for instance, and led to the identification of key molecules such as granzyme A that weren’t previously known to be involved in RA pathogenesis, Dr. Ritchin said in the interview. The current AIM work also includes Sjögren’s disease.
Finding biomarkers, diagnostic signatures
The psoriasis-PsA team has the advantage today of being able to utilize a new technology called spatial transcriptomics, which takes transcriptomics (RNA) from the single-cell level to the tissue level, enabling a look at how disease is affecting cellular organization/tissue architecture, gene activity, and cellular signaling within tissues. “It’s a huge advance in technology,” said Dr. Ritchlin. “We can actually see how the cells are interacting in the synovium [and other tissues].”
A paper published in Science Immunology and discussed at the NPF meeting demonstrates the power of special transcriptomics for learning about the skin. Dr. Scher, Dr. Ritchlin, first authors Rochelle L. Castillo, MD, and Ikjot Sidhu, MS, and other co-investigators reported a “dynamic re-organization of the immune milieu and fibroblasts in PsO lesional and non-lesional skin,” the presence of B cells in lesioned skin, and cellular organization/ecosystems that vary occurring according to clinical severity, among other findings.
Dr. Scher is using the tool for his NPF-funded diagnostic test research and as part of his work at NYU Langone for the ELLIPPS project. Among his goals: To “discover new cell populations in the microenvironment and study how they interact with each other, then compare those cells between psoriasis and PsA patients to first understand if they’re any different,” he explained after the meeting. Researchers can then investigate the synovial tissue, comparing cell populations and interactions in both compartments and looking for any shared markers/cytokines/proteins, he said.
Multiomics research, meanwhile, is showing that a test for early PsA detection could potentially combine clinical parameters with integrated multi-omic markers into a “diagnostic signature” of sorts.
At the meeting, Vinod Chandran, MD, PhD, a rheumatologist at the University of Toronto who also has an NPF PsA diagnostic test grant, said that his multi-omics analysis of blood samples from patients with psoriasis and PsA has identified signatures with a “high discriminatory value” and that certain metabolic pathways appear to play “a central role in the development and differentiation of PsA.” (Validation in other cohorts and economic analyses are ongoing, Dr. Chandran said. Low-cost alternatives that can be applied broadly in the clinic will need to be pursued, Dr. Scher said.)
Dr. Scher has also focused on skin microbiomics in looking for biomarkers for the transition to PsA. “There are potential biomarkers ... that need to be validated and expanded. We have clues,” he said at the meeting, noting that microbial signatures from nonlesional skin appear to differentiate psoriasis from PsA.
The microbiome of the skin and of the gut will also be investigated by the ELLIPPS team as they analyze biosamples and try to define psoriasis and PsA endotypes. The microbiome “is critical to psoriasis and PsA,” Dr. Ritchlin said at the meeting. “I think [our knowledge] will really expand dramatically in the next 5 years.”
Wilson Liao, MD, professor and associate vice-chair of research in the department of dermatology at the University of California, San Francisco, whose work has contributed to development of a diagnostic test, was among several experts who emphasized the importance of early diagnosis in the prevention of joint damage. Identifying the disease, he said, is “one of our true unmet needs” in psoriasis.
Dr. Liao’s research identified genes and proteins differentially expressed in PsA, psoriasis, and healthy subjects across 30 immune cells types and then identified potential biomarkers through machine learning classification of these genes and proteins along with previously published genetic risk factors for PsA.
Eyes on combination therapy
“The likelihood that all patients will respond to one biologic is very low in PsA, so we’ve been thinking about combination therapy for some time,” Dr. Ritchlin said at the meeting. “I think [dual inhibition] is coming.”
Safety has been the concern, but a phase 2 trial published this year compared a combination of IL-23 and TNF inhibition (guselkumab [Tremfya] plus golimumab [Simponi]) with monotherapy of both biologics in patients with ulcerative colitis and showed that the combination safely drove synergistic restoration of a normal epithelium and mucosal healing, he said.
A phase 2 trial in PsA, designed by Dr. Ritchlin and Dr. Scher and named AFFINITY, will study the safety and efficacy of the same combination of IL-23 and TNF blockade, compared with guselkumab (IL-23 inhibition) alone. The trial is currently completing enrollment of patients who have failed one or two anti-TNF agents.
In the meantime, combination therapy is being employed in clinics for “PsA patients who’ve been channeled through multiple biologics and are still not responding ... when [physicians] feel they’re forced to, not right away,” Dr. Ritchlin said in an interview after the meeting. “As we get a better understanding [through clinical trials], it might be something you’ll see earlier in the treatment process.”
It is wise, Dr. Ritchlin said, to devote more time to addressing “extra-articular traits” (for example, obesity, diabetes, uveitis, colitis, centralized pain) and treatable lifestyle/behavioral risk factors (for example, smoking, exercise, nutrition, adherence to therapy, social support) that can contribute to PsA and treatment nonresponse. He calls this the “treatable traits” strategy.
In practice, “there’s a big focus on inflammation and immune dysfunction, but the problem is, there are other factors involved in nonresponse, and I think ‘treatable traits’ gets to those,” Dr. Ritchlin said after the meeting. Rheumatologists and dermatologists lack time and alliances to address these issues, but “if we can find ways to do it, I think we’ll have improved outcomes.”
Dr. Ritchlin, Dr. Chandran, and Dr. Liao reported no relevant disclosures. Dr. Scher reported ties to Janssen, Pfizer, Sanofi, UCB, and Bristol-Myers Squibb.
WASHINGTON – In psoriatic disease, psoriatic arthritis (PsA) remains one of the greatest unmet needs, with the transition from cutaneous psoriasis poorly understood, diagnosis challenging, and therapeutic accomplishments trailing far behind advances for skin disease. However, leading researchers in rheumatology and dermatology believe that they’re turning the corner toward a day when therapies are phenotype-targeted and diagnosis can be made early and treatment begun well before inflammation worsens and pain and joint damage ensue.
“The challenge right now is that we don’t understand the discrete and overlapping endotypes that underlie the phenotypes or domains” of PsA, said Christopher Ritchlin, MD, MPH, professor of medicine in the division of allergy/immunology and rheumatology and the Center of Musculoskeletal Research at the University of Rochester (N.Y.), who spoke about PsA at the annual research symposium of the National Psoriasis Foundation.
“We know that mechanistically, there is dominance of the IL [interleukin]-23 and IL-17 pathways, as well as TNF [tumor necrosis factor], but we think there are tissue-specific cellular interactions [and] other pathways and mechanisms to be defined, and the goal now is to go into the tissues to find out,” he said at the meeting.
Dr. Ritchlin is co-leading a new research team dedicated to psoriatic spectrum diseases as part of the $64.5 million Autoimmune and Immune-Mediated Diseases Program (AIM) of the Accelerating Medicine Partnership (AMP), a huge public-private partnership involving the National Institutes of Health that is collecting and analyzing troves of biological data in order to better understand the cellular and molecular compositions and interactions that lead to disease.
As part of its work, this eight-center project – coined ELLIPSS, for Elucidating the Landscape of Immunoendotypes in Psoriatic Skin and Synovium – hopes to define at a molecular and single-cell level how the transition to PsA unfolds in the setting of psoriasis. Up to 30% of patients with cutaneous psoriasis also develop PsA.
The NPF, meanwhile, has invested over $3 million for research and development and validation of a diagnostic test for PsA – one that could potentially be used by dermatologists and primary care physicians to decrease the time to diagnosis. Researchers like Jose U. Scher, MD, director of the Psoriatic Arthritis Center at New York University and the NYU Colton Center for Autoimmunity, are in the thick of using multiple “-omics” tools and other sophisticated technologies to identify new targets and biomarkers.
As this work unfolds over the next several years, there is growing interest in combination therapy for PsA, Dr. Scher and Dr. Ritchlin said, and in addressing extra-articular traits, such as obesity and centralized pain, that are believed to have an impact on disease and on response to treatment.
A deep dive into the tissue
Dr. Ritchlin is among those rheumatology clinician-researchers who advocated early on for a “domain” approach to the diagnosis and management of PsA – that is, consideration of the key domains of peripheral arthritis, axial disease, enthesitis, dactylitis, and skin and nail psoriasis.
The approach is an especially important part of treatment recommendations from the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. But while interventions can be tailored to some extent to these domains, or phenotypes, there are limitations without an understanding of the different pathophysiology and mechanisms driving the heterogeneity in tissue involvement.
Dr. Ritchlin draws inspiration from pulmonology, which subtyped asthma into various phenotypes (for example, eosinophilic, allergic, intrinsic, exercise-induced) and “drilled down” on understanding underlying mechanisms to guide more specific treatment. Similar phenotype-endotype research has been done for chronic obstructive pulmonary disease, he said at the meeting, pointing to a phase 3 randomized controlled trial, published in the New England Journal of Medicine, that found dupilimab (Dupixent) was effective for patients with COPD who had type 2 inflammation as indicated by elevated eosinophil counts.
“It’s a beautiful example of how to define an endotype from a phenotypic biomarker and then use a specific intervention to improve outcomes,” Dr. Ritchlin said. “We need to do this for psoriasis and PsA.”
The ELLIPSS project will utilize the host of -omics tools and technologies (for proteomics, metabolomics, and genomics, for instance) that are making it increasingly possible to dissect the heterogeneity of single diseases and achieve more precision with treatments.
Researchers will collect blood samples and skin and/or synovial tissue biopsies from cohorts of patients with psoriasis and PsA who are treatment naïve as well as patients who are treated with a biologic or DMARD (looking at responders and non-responders). They’ll also study a cohort of psoriasis patients who may be “on a transition pathway” for PsA based on risk factors such as family history, nail psoriasis, scalp psoriasis, and body surface area greater than 5%.
Patients in all cohorts will represent distinct synovio-entheseal domains of PsA and the heterogeneity of psoriasis (for example, plaque, general, pustular, palmoplantar) and will be followed longitudinally.
With regards to PsA, one goal is to “find new pathways in the joint, then find surrogate markers in the blood that we can use to help mark particular subphenotypes [that will be identified through deep phenotyping],” Dr. Ritchlin said in an interview after the meeting. “This will lead us hopefully to a more precision-based approach.”
The ELLIPSS team joins other researchers who have been studying rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE) in an earlier iteration of the AIM program, and that will continue this work. Research on RA has thus far elucidated T-cell subsets in the rheumatoid synovium, as well as interactions of mesenchymal cells with the endothelium, for instance, and led to the identification of key molecules such as granzyme A that weren’t previously known to be involved in RA pathogenesis, Dr. Ritchin said in the interview. The current AIM work also includes Sjögren’s disease.
Finding biomarkers, diagnostic signatures
The psoriasis-PsA team has the advantage today of being able to utilize a new technology called spatial transcriptomics, which takes transcriptomics (RNA) from the single-cell level to the tissue level, enabling a look at how disease is affecting cellular organization/tissue architecture, gene activity, and cellular signaling within tissues. “It’s a huge advance in technology,” said Dr. Ritchlin. “We can actually see how the cells are interacting in the synovium [and other tissues].”
A paper published in Science Immunology and discussed at the NPF meeting demonstrates the power of special transcriptomics for learning about the skin. Dr. Scher, Dr. Ritchlin, first authors Rochelle L. Castillo, MD, and Ikjot Sidhu, MS, and other co-investigators reported a “dynamic re-organization of the immune milieu and fibroblasts in PsO lesional and non-lesional skin,” the presence of B cells in lesioned skin, and cellular organization/ecosystems that vary occurring according to clinical severity, among other findings.
Dr. Scher is using the tool for his NPF-funded diagnostic test research and as part of his work at NYU Langone for the ELLIPPS project. Among his goals: To “discover new cell populations in the microenvironment and study how they interact with each other, then compare those cells between psoriasis and PsA patients to first understand if they’re any different,” he explained after the meeting. Researchers can then investigate the synovial tissue, comparing cell populations and interactions in both compartments and looking for any shared markers/cytokines/proteins, he said.
Multiomics research, meanwhile, is showing that a test for early PsA detection could potentially combine clinical parameters with integrated multi-omic markers into a “diagnostic signature” of sorts.
At the meeting, Vinod Chandran, MD, PhD, a rheumatologist at the University of Toronto who also has an NPF PsA diagnostic test grant, said that his multi-omics analysis of blood samples from patients with psoriasis and PsA has identified signatures with a “high discriminatory value” and that certain metabolic pathways appear to play “a central role in the development and differentiation of PsA.” (Validation in other cohorts and economic analyses are ongoing, Dr. Chandran said. Low-cost alternatives that can be applied broadly in the clinic will need to be pursued, Dr. Scher said.)
Dr. Scher has also focused on skin microbiomics in looking for biomarkers for the transition to PsA. “There are potential biomarkers ... that need to be validated and expanded. We have clues,” he said at the meeting, noting that microbial signatures from nonlesional skin appear to differentiate psoriasis from PsA.
The microbiome of the skin and of the gut will also be investigated by the ELLIPPS team as they analyze biosamples and try to define psoriasis and PsA endotypes. The microbiome “is critical to psoriasis and PsA,” Dr. Ritchlin said at the meeting. “I think [our knowledge] will really expand dramatically in the next 5 years.”
Wilson Liao, MD, professor and associate vice-chair of research in the department of dermatology at the University of California, San Francisco, whose work has contributed to development of a diagnostic test, was among several experts who emphasized the importance of early diagnosis in the prevention of joint damage. Identifying the disease, he said, is “one of our true unmet needs” in psoriasis.
Dr. Liao’s research identified genes and proteins differentially expressed in PsA, psoriasis, and healthy subjects across 30 immune cells types and then identified potential biomarkers through machine learning classification of these genes and proteins along with previously published genetic risk factors for PsA.
Eyes on combination therapy
“The likelihood that all patients will respond to one biologic is very low in PsA, so we’ve been thinking about combination therapy for some time,” Dr. Ritchlin said at the meeting. “I think [dual inhibition] is coming.”
Safety has been the concern, but a phase 2 trial published this year compared a combination of IL-23 and TNF inhibition (guselkumab [Tremfya] plus golimumab [Simponi]) with monotherapy of both biologics in patients with ulcerative colitis and showed that the combination safely drove synergistic restoration of a normal epithelium and mucosal healing, he said.
A phase 2 trial in PsA, designed by Dr. Ritchlin and Dr. Scher and named AFFINITY, will study the safety and efficacy of the same combination of IL-23 and TNF blockade, compared with guselkumab (IL-23 inhibition) alone. The trial is currently completing enrollment of patients who have failed one or two anti-TNF agents.
In the meantime, combination therapy is being employed in clinics for “PsA patients who’ve been channeled through multiple biologics and are still not responding ... when [physicians] feel they’re forced to, not right away,” Dr. Ritchlin said in an interview after the meeting. “As we get a better understanding [through clinical trials], it might be something you’ll see earlier in the treatment process.”
It is wise, Dr. Ritchlin said, to devote more time to addressing “extra-articular traits” (for example, obesity, diabetes, uveitis, colitis, centralized pain) and treatable lifestyle/behavioral risk factors (for example, smoking, exercise, nutrition, adherence to therapy, social support) that can contribute to PsA and treatment nonresponse. He calls this the “treatable traits” strategy.
In practice, “there’s a big focus on inflammation and immune dysfunction, but the problem is, there are other factors involved in nonresponse, and I think ‘treatable traits’ gets to those,” Dr. Ritchlin said after the meeting. Rheumatologists and dermatologists lack time and alliances to address these issues, but “if we can find ways to do it, I think we’ll have improved outcomes.”
Dr. Ritchlin, Dr. Chandran, and Dr. Liao reported no relevant disclosures. Dr. Scher reported ties to Janssen, Pfizer, Sanofi, UCB, and Bristol-Myers Squibb.
WASHINGTON – In psoriatic disease, psoriatic arthritis (PsA) remains one of the greatest unmet needs, with the transition from cutaneous psoriasis poorly understood, diagnosis challenging, and therapeutic accomplishments trailing far behind advances for skin disease. However, leading researchers in rheumatology and dermatology believe that they’re turning the corner toward a day when therapies are phenotype-targeted and diagnosis can be made early and treatment begun well before inflammation worsens and pain and joint damage ensue.
“The challenge right now is that we don’t understand the discrete and overlapping endotypes that underlie the phenotypes or domains” of PsA, said Christopher Ritchlin, MD, MPH, professor of medicine in the division of allergy/immunology and rheumatology and the Center of Musculoskeletal Research at the University of Rochester (N.Y.), who spoke about PsA at the annual research symposium of the National Psoriasis Foundation.
“We know that mechanistically, there is dominance of the IL [interleukin]-23 and IL-17 pathways, as well as TNF [tumor necrosis factor], but we think there are tissue-specific cellular interactions [and] other pathways and mechanisms to be defined, and the goal now is to go into the tissues to find out,” he said at the meeting.
Dr. Ritchlin is co-leading a new research team dedicated to psoriatic spectrum diseases as part of the $64.5 million Autoimmune and Immune-Mediated Diseases Program (AIM) of the Accelerating Medicine Partnership (AMP), a huge public-private partnership involving the National Institutes of Health that is collecting and analyzing troves of biological data in order to better understand the cellular and molecular compositions and interactions that lead to disease.
As part of its work, this eight-center project – coined ELLIPSS, for Elucidating the Landscape of Immunoendotypes in Psoriatic Skin and Synovium – hopes to define at a molecular and single-cell level how the transition to PsA unfolds in the setting of psoriasis. Up to 30% of patients with cutaneous psoriasis also develop PsA.
The NPF, meanwhile, has invested over $3 million for research and development and validation of a diagnostic test for PsA – one that could potentially be used by dermatologists and primary care physicians to decrease the time to diagnosis. Researchers like Jose U. Scher, MD, director of the Psoriatic Arthritis Center at New York University and the NYU Colton Center for Autoimmunity, are in the thick of using multiple “-omics” tools and other sophisticated technologies to identify new targets and biomarkers.
As this work unfolds over the next several years, there is growing interest in combination therapy for PsA, Dr. Scher and Dr. Ritchlin said, and in addressing extra-articular traits, such as obesity and centralized pain, that are believed to have an impact on disease and on response to treatment.
A deep dive into the tissue
Dr. Ritchlin is among those rheumatology clinician-researchers who advocated early on for a “domain” approach to the diagnosis and management of PsA – that is, consideration of the key domains of peripheral arthritis, axial disease, enthesitis, dactylitis, and skin and nail psoriasis.
The approach is an especially important part of treatment recommendations from the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis. But while interventions can be tailored to some extent to these domains, or phenotypes, there are limitations without an understanding of the different pathophysiology and mechanisms driving the heterogeneity in tissue involvement.
Dr. Ritchlin draws inspiration from pulmonology, which subtyped asthma into various phenotypes (for example, eosinophilic, allergic, intrinsic, exercise-induced) and “drilled down” on understanding underlying mechanisms to guide more specific treatment. Similar phenotype-endotype research has been done for chronic obstructive pulmonary disease, he said at the meeting, pointing to a phase 3 randomized controlled trial, published in the New England Journal of Medicine, that found dupilimab (Dupixent) was effective for patients with COPD who had type 2 inflammation as indicated by elevated eosinophil counts.
“It’s a beautiful example of how to define an endotype from a phenotypic biomarker and then use a specific intervention to improve outcomes,” Dr. Ritchlin said. “We need to do this for psoriasis and PsA.”
The ELLIPSS project will utilize the host of -omics tools and technologies (for proteomics, metabolomics, and genomics, for instance) that are making it increasingly possible to dissect the heterogeneity of single diseases and achieve more precision with treatments.
Researchers will collect blood samples and skin and/or synovial tissue biopsies from cohorts of patients with psoriasis and PsA who are treatment naïve as well as patients who are treated with a biologic or DMARD (looking at responders and non-responders). They’ll also study a cohort of psoriasis patients who may be “on a transition pathway” for PsA based on risk factors such as family history, nail psoriasis, scalp psoriasis, and body surface area greater than 5%.
Patients in all cohorts will represent distinct synovio-entheseal domains of PsA and the heterogeneity of psoriasis (for example, plaque, general, pustular, palmoplantar) and will be followed longitudinally.
With regards to PsA, one goal is to “find new pathways in the joint, then find surrogate markers in the blood that we can use to help mark particular subphenotypes [that will be identified through deep phenotyping],” Dr. Ritchlin said in an interview after the meeting. “This will lead us hopefully to a more precision-based approach.”
The ELLIPSS team joins other researchers who have been studying rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE) in an earlier iteration of the AIM program, and that will continue this work. Research on RA has thus far elucidated T-cell subsets in the rheumatoid synovium, as well as interactions of mesenchymal cells with the endothelium, for instance, and led to the identification of key molecules such as granzyme A that weren’t previously known to be involved in RA pathogenesis, Dr. Ritchin said in the interview. The current AIM work also includes Sjögren’s disease.
Finding biomarkers, diagnostic signatures
The psoriasis-PsA team has the advantage today of being able to utilize a new technology called spatial transcriptomics, which takes transcriptomics (RNA) from the single-cell level to the tissue level, enabling a look at how disease is affecting cellular organization/tissue architecture, gene activity, and cellular signaling within tissues. “It’s a huge advance in technology,” said Dr. Ritchlin. “We can actually see how the cells are interacting in the synovium [and other tissues].”
A paper published in Science Immunology and discussed at the NPF meeting demonstrates the power of special transcriptomics for learning about the skin. Dr. Scher, Dr. Ritchlin, first authors Rochelle L. Castillo, MD, and Ikjot Sidhu, MS, and other co-investigators reported a “dynamic re-organization of the immune milieu and fibroblasts in PsO lesional and non-lesional skin,” the presence of B cells in lesioned skin, and cellular organization/ecosystems that vary occurring according to clinical severity, among other findings.
Dr. Scher is using the tool for his NPF-funded diagnostic test research and as part of his work at NYU Langone for the ELLIPPS project. Among his goals: To “discover new cell populations in the microenvironment and study how they interact with each other, then compare those cells between psoriasis and PsA patients to first understand if they’re any different,” he explained after the meeting. Researchers can then investigate the synovial tissue, comparing cell populations and interactions in both compartments and looking for any shared markers/cytokines/proteins, he said.
Multiomics research, meanwhile, is showing that a test for early PsA detection could potentially combine clinical parameters with integrated multi-omic markers into a “diagnostic signature” of sorts.
At the meeting, Vinod Chandran, MD, PhD, a rheumatologist at the University of Toronto who also has an NPF PsA diagnostic test grant, said that his multi-omics analysis of blood samples from patients with psoriasis and PsA has identified signatures with a “high discriminatory value” and that certain metabolic pathways appear to play “a central role in the development and differentiation of PsA.” (Validation in other cohorts and economic analyses are ongoing, Dr. Chandran said. Low-cost alternatives that can be applied broadly in the clinic will need to be pursued, Dr. Scher said.)
Dr. Scher has also focused on skin microbiomics in looking for biomarkers for the transition to PsA. “There are potential biomarkers ... that need to be validated and expanded. We have clues,” he said at the meeting, noting that microbial signatures from nonlesional skin appear to differentiate psoriasis from PsA.
The microbiome of the skin and of the gut will also be investigated by the ELLIPPS team as they analyze biosamples and try to define psoriasis and PsA endotypes. The microbiome “is critical to psoriasis and PsA,” Dr. Ritchlin said at the meeting. “I think [our knowledge] will really expand dramatically in the next 5 years.”
Wilson Liao, MD, professor and associate vice-chair of research in the department of dermatology at the University of California, San Francisco, whose work has contributed to development of a diagnostic test, was among several experts who emphasized the importance of early diagnosis in the prevention of joint damage. Identifying the disease, he said, is “one of our true unmet needs” in psoriasis.
Dr. Liao’s research identified genes and proteins differentially expressed in PsA, psoriasis, and healthy subjects across 30 immune cells types and then identified potential biomarkers through machine learning classification of these genes and proteins along with previously published genetic risk factors for PsA.
Eyes on combination therapy
“The likelihood that all patients will respond to one biologic is very low in PsA, so we’ve been thinking about combination therapy for some time,” Dr. Ritchlin said at the meeting. “I think [dual inhibition] is coming.”
Safety has been the concern, but a phase 2 trial published this year compared a combination of IL-23 and TNF inhibition (guselkumab [Tremfya] plus golimumab [Simponi]) with monotherapy of both biologics in patients with ulcerative colitis and showed that the combination safely drove synergistic restoration of a normal epithelium and mucosal healing, he said.
A phase 2 trial in PsA, designed by Dr. Ritchlin and Dr. Scher and named AFFINITY, will study the safety and efficacy of the same combination of IL-23 and TNF blockade, compared with guselkumab (IL-23 inhibition) alone. The trial is currently completing enrollment of patients who have failed one or two anti-TNF agents.
In the meantime, combination therapy is being employed in clinics for “PsA patients who’ve been channeled through multiple biologics and are still not responding ... when [physicians] feel they’re forced to, not right away,” Dr. Ritchlin said in an interview after the meeting. “As we get a better understanding [through clinical trials], it might be something you’ll see earlier in the treatment process.”
It is wise, Dr. Ritchlin said, to devote more time to addressing “extra-articular traits” (for example, obesity, diabetes, uveitis, colitis, centralized pain) and treatable lifestyle/behavioral risk factors (for example, smoking, exercise, nutrition, adherence to therapy, social support) that can contribute to PsA and treatment nonresponse. He calls this the “treatable traits” strategy.
In practice, “there’s a big focus on inflammation and immune dysfunction, but the problem is, there are other factors involved in nonresponse, and I think ‘treatable traits’ gets to those,” Dr. Ritchlin said after the meeting. Rheumatologists and dermatologists lack time and alliances to address these issues, but “if we can find ways to do it, I think we’ll have improved outcomes.”
Dr. Ritchlin, Dr. Chandran, and Dr. Liao reported no relevant disclosures. Dr. Scher reported ties to Janssen, Pfizer, Sanofi, UCB, and Bristol-Myers Squibb.
AT THE NPF RESEARCH SYMPOSIUM 2023
FDA approves bimekizumab for moderate to severe plaque psoriasis in adults
, the manufacturer announced in a press release.
The indication is for adults who are candidates for systemic therapy or phototherapy.
With this approval, bimekizumab becomes the only interleukin (IL)-17A and IL-17F inhibitor approved for the treatment of these patients. Psoriasis affects more than 7.5 million U.S. adults, according to the National Psoriasis Foundation.
“We have been eagerly awaiting bimekizumab,” Mark Lebwohl, MD, bimekizumab investigator and dean for clinical therapeutics at the Icahn School of Medicine at Mount Sinai, New York City, said in the press release.
Dr. Lebwohl states that bimekizumab “achieved superior levels of skin clearance at week 16 compared to placebo and three existing biologics for psoriasis, with responses being rapid and lasting up to a year. Long-term data have also shown that the majority of patients maintained high levels of clinical response through three years.”
The most common adverse reactions (occurring in at least 1% of patients) are upper respiratory infections, oral candidiasis, headache, tinea infections, gastroenteritis, herpes simplex infections, acne, folliculitis, other Candida infections, fatigue, and injection site reactions, according to the company, UCB.
Available in about 1 month in U.S.
Bimekizumab can be administered by a health care provider or it can be self-injected by a patient after training. It is available as a single-dose prefilled autoinjector and a single-dose prefilled syringe and will be available in the United States in about 1 month.
The recommended dosage of bimekizumab for patients with psoriasis is 320 mg (two subcutaneous injections of 160 mg each) at baseline, then on weeks 4, 8, 12, and 16, then every 8 weeks thereafter. For patients weighing at least 120 kg (about 265 lb), a dosage of 320 mg every 4 weeks after week 16 may be considered, the company states.
Three phase 3 trials
Approval was based on three phase 3 multicenter, randomized, placebo and/or active comparator-controlled trials: bimekizumab versus placebo and ustekinumab (BE VIVID); versus placebo (BE READY); and versus adalimumab (BE SURE).
“All studies met their co-primary endpoints and all ranked secondary endpoints,” the company reports. Secondary endpoints included the Psoriasis Area and Severity Index (PASI) 75 at week 4 and PASI 100 (complete skin clearance) at week 16.
Highlights from the trials include the following results, according to UCB:
- Clear or almost clear skin: More than 8 out of 10 patients achieved a 90% or greater reduction from baseline in the PASI 90 and an Investigator’s Global Assessment score of 0/1 at week 16.
- Complete skin clearance: About 60% of patients achieved PASI 100 at week 16.
- Time to response: More than 70% of patients achieved PASI 75 at week 4 following one 320-mg dose.
Safety information
The safety information includes the statement that bimekizumab may increase the risk for suicidal ideation and behavior, though a causal association has not been established. Prescribers should advise patients, caregivers, and families “to monitor for emergence or worsening of depression, suicidal ideation, or other mood changes,” according to the prescribing information.
Bimekizumab is being studied for other conditions, including hidradenitis suppurativa. In the European Union, it was approved for the treatment of psoriasis in 2021 and for the treatment of psoriatic arthritis and ankylosing spondylitis in June 2023.
Dr. Lebwohl is an investigator for UCB. He has not accepted any consulting payments from UCB.
A version of this article first appeared on Medscape.com.
, the manufacturer announced in a press release.
The indication is for adults who are candidates for systemic therapy or phototherapy.
With this approval, bimekizumab becomes the only interleukin (IL)-17A and IL-17F inhibitor approved for the treatment of these patients. Psoriasis affects more than 7.5 million U.S. adults, according to the National Psoriasis Foundation.
“We have been eagerly awaiting bimekizumab,” Mark Lebwohl, MD, bimekizumab investigator and dean for clinical therapeutics at the Icahn School of Medicine at Mount Sinai, New York City, said in the press release.
Dr. Lebwohl states that bimekizumab “achieved superior levels of skin clearance at week 16 compared to placebo and three existing biologics for psoriasis, with responses being rapid and lasting up to a year. Long-term data have also shown that the majority of patients maintained high levels of clinical response through three years.”
The most common adverse reactions (occurring in at least 1% of patients) are upper respiratory infections, oral candidiasis, headache, tinea infections, gastroenteritis, herpes simplex infections, acne, folliculitis, other Candida infections, fatigue, and injection site reactions, according to the company, UCB.
Available in about 1 month in U.S.
Bimekizumab can be administered by a health care provider or it can be self-injected by a patient after training. It is available as a single-dose prefilled autoinjector and a single-dose prefilled syringe and will be available in the United States in about 1 month.
The recommended dosage of bimekizumab for patients with psoriasis is 320 mg (two subcutaneous injections of 160 mg each) at baseline, then on weeks 4, 8, 12, and 16, then every 8 weeks thereafter. For patients weighing at least 120 kg (about 265 lb), a dosage of 320 mg every 4 weeks after week 16 may be considered, the company states.
Three phase 3 trials
Approval was based on three phase 3 multicenter, randomized, placebo and/or active comparator-controlled trials: bimekizumab versus placebo and ustekinumab (BE VIVID); versus placebo (BE READY); and versus adalimumab (BE SURE).
“All studies met their co-primary endpoints and all ranked secondary endpoints,” the company reports. Secondary endpoints included the Psoriasis Area and Severity Index (PASI) 75 at week 4 and PASI 100 (complete skin clearance) at week 16.
Highlights from the trials include the following results, according to UCB:
- Clear or almost clear skin: More than 8 out of 10 patients achieved a 90% or greater reduction from baseline in the PASI 90 and an Investigator’s Global Assessment score of 0/1 at week 16.
- Complete skin clearance: About 60% of patients achieved PASI 100 at week 16.
- Time to response: More than 70% of patients achieved PASI 75 at week 4 following one 320-mg dose.
Safety information
The safety information includes the statement that bimekizumab may increase the risk for suicidal ideation and behavior, though a causal association has not been established. Prescribers should advise patients, caregivers, and families “to monitor for emergence or worsening of depression, suicidal ideation, or other mood changes,” according to the prescribing information.
Bimekizumab is being studied for other conditions, including hidradenitis suppurativa. In the European Union, it was approved for the treatment of psoriasis in 2021 and for the treatment of psoriatic arthritis and ankylosing spondylitis in June 2023.
Dr. Lebwohl is an investigator for UCB. He has not accepted any consulting payments from UCB.
A version of this article first appeared on Medscape.com.
, the manufacturer announced in a press release.
The indication is for adults who are candidates for systemic therapy or phototherapy.
With this approval, bimekizumab becomes the only interleukin (IL)-17A and IL-17F inhibitor approved for the treatment of these patients. Psoriasis affects more than 7.5 million U.S. adults, according to the National Psoriasis Foundation.
“We have been eagerly awaiting bimekizumab,” Mark Lebwohl, MD, bimekizumab investigator and dean for clinical therapeutics at the Icahn School of Medicine at Mount Sinai, New York City, said in the press release.
Dr. Lebwohl states that bimekizumab “achieved superior levels of skin clearance at week 16 compared to placebo and three existing biologics for psoriasis, with responses being rapid and lasting up to a year. Long-term data have also shown that the majority of patients maintained high levels of clinical response through three years.”
The most common adverse reactions (occurring in at least 1% of patients) are upper respiratory infections, oral candidiasis, headache, tinea infections, gastroenteritis, herpes simplex infections, acne, folliculitis, other Candida infections, fatigue, and injection site reactions, according to the company, UCB.
Available in about 1 month in U.S.
Bimekizumab can be administered by a health care provider or it can be self-injected by a patient after training. It is available as a single-dose prefilled autoinjector and a single-dose prefilled syringe and will be available in the United States in about 1 month.
The recommended dosage of bimekizumab for patients with psoriasis is 320 mg (two subcutaneous injections of 160 mg each) at baseline, then on weeks 4, 8, 12, and 16, then every 8 weeks thereafter. For patients weighing at least 120 kg (about 265 lb), a dosage of 320 mg every 4 weeks after week 16 may be considered, the company states.
Three phase 3 trials
Approval was based on three phase 3 multicenter, randomized, placebo and/or active comparator-controlled trials: bimekizumab versus placebo and ustekinumab (BE VIVID); versus placebo (BE READY); and versus adalimumab (BE SURE).
“All studies met their co-primary endpoints and all ranked secondary endpoints,” the company reports. Secondary endpoints included the Psoriasis Area and Severity Index (PASI) 75 at week 4 and PASI 100 (complete skin clearance) at week 16.
Highlights from the trials include the following results, according to UCB:
- Clear or almost clear skin: More than 8 out of 10 patients achieved a 90% or greater reduction from baseline in the PASI 90 and an Investigator’s Global Assessment score of 0/1 at week 16.
- Complete skin clearance: About 60% of patients achieved PASI 100 at week 16.
- Time to response: More than 70% of patients achieved PASI 75 at week 4 following one 320-mg dose.
Safety information
The safety information includes the statement that bimekizumab may increase the risk for suicidal ideation and behavior, though a causal association has not been established. Prescribers should advise patients, caregivers, and families “to monitor for emergence or worsening of depression, suicidal ideation, or other mood changes,” according to the prescribing information.
Bimekizumab is being studied for other conditions, including hidradenitis suppurativa. In the European Union, it was approved for the treatment of psoriasis in 2021 and for the treatment of psoriatic arthritis and ankylosing spondylitis in June 2023.
Dr. Lebwohl is an investigator for UCB. He has not accepted any consulting payments from UCB.
A version of this article first appeared on Medscape.com.
Pilot study: High-dose IL-23 inhibition shows promise for psoriasis remission in some patients
WASHINGTON – A series of investigating whether high-dose IL-23 inhibition can target resident memory T cells and thereby induce long-term remission, Andrew Blauvelt, MD, MBA, reported at the annual research symposium of the National Psoriasis Foundation.
Understanding and trying to target resident memory T (Trm) cells has become a hot topic in psoriasis research, with possible importance for psoriatic arthritis as well. The cells, which normally develop within tissues in a pathogen-specific manner and persist after infections resolve, have been found in healed psoriatic skin and are believed to be responsible for recurrences of psoriasis at the same sites previously affected by the disease. Research suggests, moreover, that Trm cells are dependent on IL-23 for their survival, said Dr. Blauvelt, an investigator with the Oregon Medical Research Center, Portland.
Using an approach he has coined “knockout” therapy, 20 adult patients at the center were randomized 1:1 in double-blinded dosing to receive 300 mg or 600 mg of risankizumab (two and four times the standard initial doses, respectively) at 0, 4, and 16 weeks, and were seen every 4-6 weeks in a double-blinded follow-up period. Skin biopsies of lesional and nonlesional skin were collected at weeks 0 and 52 for RNA sequence analysis to evaluate changes in Trm cell number and effector function.
At week 28, almost all patients – 94% – achieved a Psoriasis Area and Severity Index 90 score, and 83% achieved PASI 100. At week 52, of the 18 still-enrolled patients, 69% had PASI 90 scores and 43% maintained PASI 100 scores. “We’re not curing psoriasis, but that is incredible to have 43% still clear 9 months after the last dose,” Dr. Blauvelt said.
The findings are interim results that pool the two doses of risankizumab (Skyrizi). An evaluation of maintenance of efficacy with the 300-mg versus 600-mg doses and results of the skin biopsies will be presented at the 2024 annual meeting of the American Academy of Dermatology, he said. Patients who have not had recurrences are being followed up to week 100 (unblinded).
Skin biopsy findings together with levels of clearance will “add insight as to whether high-dose IL-23 inhibition is associated with higher levels of complete clearance (i.e., PASI 100) over long periods of time, and whether remissions are associated with more profound knock-down of Trm cells,” Dr. Blauvelt wrote in an editorial on Trm cells in psoriasis published in the Journal of Psoriasis and Psoriatic Arthritis.
Risankizumab was approved by the Food and Drug Administration for moderate to severe plaque psoriasis in 2019, and for active psoriatic arthritis in 2022.
Impact of IL-23 inhibition
A 2021 study, Dr. Blauvelt noted in his presentation, showed that the IL-23 inhibitor guselkumab (Tremfya) reduced the number of Trm cells in healed psoriatic skin, while the IL-17A inhibitor secukinumab (Cosentyx) did not affect Trm cells.
Other researchers have concluded that local IL-23 is required for the proliferation and retention of Trm cells in the skin, he said, noting also that, as a class, IL-23 blockers “are associated with the longest disease-free intervals” in patients with psoriasis.
Research showing loss of Trm cells in biopsies of cleared skin is “incredibly important,” as is data showing that IL-23 may be “a critical survival factor” for Trm cells, said Christopher Ritchlin, MD, MPH, a rheumatologist and director of the clinical immunology research unit at the University of Rochester (N.Y.) Medical Center, who attended the NPF meeting and was asked to comment on the growing interest in Trm cells. He did not hear Dr. Blauvelt’s presentation but has followed Trm research in psoriasis.
Measuring Trm cells in synovial tissue of patients with psoriatic disease is, in fact, on the agenda of a new research team coled by Dr. Ritchlin that is part of the Autoimmune and Immune-Mediated Diseases Program of the Accelerating Medicine Partnership – a public-private partnership that aims to collect and analyze troves of biological data in order to illuminate the cellular and molecular interactions leading to autoimmune diseases.
“Resident memory T cells have been thought to be cells that are constrained to being resident in the tissue, and while that’s largely true, there’s more recent data showing that they actually can dedifferentiate and go back to the lymph node and then go back to the joint again,” Dr. Ritchlin noted in an interview. “So it’s a more complicated story,” but nonetheless targeting the cells is a concept worth exploring therapeutically.
Moving toward a cure?
The high induction doses of risankizumab in Dr. Blauvelt’s phase 2 “knockout” study were well tolerated through week 40, with safety profiles similar overall to those reported in previous studies of risankizumab and “no new safety signals,” Dr. Blauvelt said at the meeting.
(At baseline, patients had a mean disease duration of 21 years, a mean affected BSA of 21, and a mean PASI of 18.5. Seven had prior treatment with biologic medications, though not in the prior 4 months and not with risankizumab.)
In lieu of a cure, which is still possible in the next 10 years, he said, patients are eager for longer-term remission and the ability to break away from established regular dosing. After publication of a phase 1 study of risankizumab, in which a notable number of patients experienced complete skin clearance up to 1 year following a single high dose, “we started getting numerous calls in our [clinical trials] office from patients who said ‘I want to be on that drug that you give once a year,’ ” Dr. Blauvelt said. (The high dosing was not investigated any further in phase 2 and 3 research, he added.)
While the “knockout” study adopts a “hit hard” approach, it is also possible that a strategy to hit both hard and early after disease onset may induce long-term remission and/or cure of the disease. “I’m talking about using the best things first. There are hints that if we treat early, maybe we can keep [psoriasis] from becoming a chronic disease, keep it from ‘setting up shop’ if you will,” he said, noting that the “hit hard, hit early” concept is not unique to dermatology.
During a discussion period, Dr. Blauvelt said that in a future iteration of the “knockout” study, he would like to evaluate risankizumab in patients with disease duration under 12 months. It may also be valuable to look not only at Trm cells, but more broadly at other elements of the tissue architecture before and after treatment.
Among other strategies for achieving long-term remission and/or cure is the expansion of regulatory T cells, which work to “control inappropriate immune responses” and calm inflammation. Defects in the number and function of regulatory T cells are associated with psoriasis and other autoimmune diseases, and it appears in early research from other investigators that low-dose IL-2 can induce the expansion of regulatory T cells and improve psoriasis. “Keep tuned,” Dr. Blauvelt said.
Dr. Blauvelt disclosed ties with numerous pharmaceutical companies. Dr. Ritchlin had no disclosures.
WASHINGTON – A series of investigating whether high-dose IL-23 inhibition can target resident memory T cells and thereby induce long-term remission, Andrew Blauvelt, MD, MBA, reported at the annual research symposium of the National Psoriasis Foundation.
Understanding and trying to target resident memory T (Trm) cells has become a hot topic in psoriasis research, with possible importance for psoriatic arthritis as well. The cells, which normally develop within tissues in a pathogen-specific manner and persist after infections resolve, have been found in healed psoriatic skin and are believed to be responsible for recurrences of psoriasis at the same sites previously affected by the disease. Research suggests, moreover, that Trm cells are dependent on IL-23 for their survival, said Dr. Blauvelt, an investigator with the Oregon Medical Research Center, Portland.
Using an approach he has coined “knockout” therapy, 20 adult patients at the center were randomized 1:1 in double-blinded dosing to receive 300 mg or 600 mg of risankizumab (two and four times the standard initial doses, respectively) at 0, 4, and 16 weeks, and were seen every 4-6 weeks in a double-blinded follow-up period. Skin biopsies of lesional and nonlesional skin were collected at weeks 0 and 52 for RNA sequence analysis to evaluate changes in Trm cell number and effector function.
At week 28, almost all patients – 94% – achieved a Psoriasis Area and Severity Index 90 score, and 83% achieved PASI 100. At week 52, of the 18 still-enrolled patients, 69% had PASI 90 scores and 43% maintained PASI 100 scores. “We’re not curing psoriasis, but that is incredible to have 43% still clear 9 months after the last dose,” Dr. Blauvelt said.
The findings are interim results that pool the two doses of risankizumab (Skyrizi). An evaluation of maintenance of efficacy with the 300-mg versus 600-mg doses and results of the skin biopsies will be presented at the 2024 annual meeting of the American Academy of Dermatology, he said. Patients who have not had recurrences are being followed up to week 100 (unblinded).
Skin biopsy findings together with levels of clearance will “add insight as to whether high-dose IL-23 inhibition is associated with higher levels of complete clearance (i.e., PASI 100) over long periods of time, and whether remissions are associated with more profound knock-down of Trm cells,” Dr. Blauvelt wrote in an editorial on Trm cells in psoriasis published in the Journal of Psoriasis and Psoriatic Arthritis.
Risankizumab was approved by the Food and Drug Administration for moderate to severe plaque psoriasis in 2019, and for active psoriatic arthritis in 2022.
Impact of IL-23 inhibition
A 2021 study, Dr. Blauvelt noted in his presentation, showed that the IL-23 inhibitor guselkumab (Tremfya) reduced the number of Trm cells in healed psoriatic skin, while the IL-17A inhibitor secukinumab (Cosentyx) did not affect Trm cells.
Other researchers have concluded that local IL-23 is required for the proliferation and retention of Trm cells in the skin, he said, noting also that, as a class, IL-23 blockers “are associated with the longest disease-free intervals” in patients with psoriasis.
Research showing loss of Trm cells in biopsies of cleared skin is “incredibly important,” as is data showing that IL-23 may be “a critical survival factor” for Trm cells, said Christopher Ritchlin, MD, MPH, a rheumatologist and director of the clinical immunology research unit at the University of Rochester (N.Y.) Medical Center, who attended the NPF meeting and was asked to comment on the growing interest in Trm cells. He did not hear Dr. Blauvelt’s presentation but has followed Trm research in psoriasis.
Measuring Trm cells in synovial tissue of patients with psoriatic disease is, in fact, on the agenda of a new research team coled by Dr. Ritchlin that is part of the Autoimmune and Immune-Mediated Diseases Program of the Accelerating Medicine Partnership – a public-private partnership that aims to collect and analyze troves of biological data in order to illuminate the cellular and molecular interactions leading to autoimmune diseases.
“Resident memory T cells have been thought to be cells that are constrained to being resident in the tissue, and while that’s largely true, there’s more recent data showing that they actually can dedifferentiate and go back to the lymph node and then go back to the joint again,” Dr. Ritchlin noted in an interview. “So it’s a more complicated story,” but nonetheless targeting the cells is a concept worth exploring therapeutically.
Moving toward a cure?
The high induction doses of risankizumab in Dr. Blauvelt’s phase 2 “knockout” study were well tolerated through week 40, with safety profiles similar overall to those reported in previous studies of risankizumab and “no new safety signals,” Dr. Blauvelt said at the meeting.
(At baseline, patients had a mean disease duration of 21 years, a mean affected BSA of 21, and a mean PASI of 18.5. Seven had prior treatment with biologic medications, though not in the prior 4 months and not with risankizumab.)
In lieu of a cure, which is still possible in the next 10 years, he said, patients are eager for longer-term remission and the ability to break away from established regular dosing. After publication of a phase 1 study of risankizumab, in which a notable number of patients experienced complete skin clearance up to 1 year following a single high dose, “we started getting numerous calls in our [clinical trials] office from patients who said ‘I want to be on that drug that you give once a year,’ ” Dr. Blauvelt said. (The high dosing was not investigated any further in phase 2 and 3 research, he added.)
While the “knockout” study adopts a “hit hard” approach, it is also possible that a strategy to hit both hard and early after disease onset may induce long-term remission and/or cure of the disease. “I’m talking about using the best things first. There are hints that if we treat early, maybe we can keep [psoriasis] from becoming a chronic disease, keep it from ‘setting up shop’ if you will,” he said, noting that the “hit hard, hit early” concept is not unique to dermatology.
During a discussion period, Dr. Blauvelt said that in a future iteration of the “knockout” study, he would like to evaluate risankizumab in patients with disease duration under 12 months. It may also be valuable to look not only at Trm cells, but more broadly at other elements of the tissue architecture before and after treatment.
Among other strategies for achieving long-term remission and/or cure is the expansion of regulatory T cells, which work to “control inappropriate immune responses” and calm inflammation. Defects in the number and function of regulatory T cells are associated with psoriasis and other autoimmune diseases, and it appears in early research from other investigators that low-dose IL-2 can induce the expansion of regulatory T cells and improve psoriasis. “Keep tuned,” Dr. Blauvelt said.
Dr. Blauvelt disclosed ties with numerous pharmaceutical companies. Dr. Ritchlin had no disclosures.
WASHINGTON – A series of investigating whether high-dose IL-23 inhibition can target resident memory T cells and thereby induce long-term remission, Andrew Blauvelt, MD, MBA, reported at the annual research symposium of the National Psoriasis Foundation.
Understanding and trying to target resident memory T (Trm) cells has become a hot topic in psoriasis research, with possible importance for psoriatic arthritis as well. The cells, which normally develop within tissues in a pathogen-specific manner and persist after infections resolve, have been found in healed psoriatic skin and are believed to be responsible for recurrences of psoriasis at the same sites previously affected by the disease. Research suggests, moreover, that Trm cells are dependent on IL-23 for their survival, said Dr. Blauvelt, an investigator with the Oregon Medical Research Center, Portland.
Using an approach he has coined “knockout” therapy, 20 adult patients at the center were randomized 1:1 in double-blinded dosing to receive 300 mg or 600 mg of risankizumab (two and four times the standard initial doses, respectively) at 0, 4, and 16 weeks, and were seen every 4-6 weeks in a double-blinded follow-up period. Skin biopsies of lesional and nonlesional skin were collected at weeks 0 and 52 for RNA sequence analysis to evaluate changes in Trm cell number and effector function.
At week 28, almost all patients – 94% – achieved a Psoriasis Area and Severity Index 90 score, and 83% achieved PASI 100. At week 52, of the 18 still-enrolled patients, 69% had PASI 90 scores and 43% maintained PASI 100 scores. “We’re not curing psoriasis, but that is incredible to have 43% still clear 9 months after the last dose,” Dr. Blauvelt said.
The findings are interim results that pool the two doses of risankizumab (Skyrizi). An evaluation of maintenance of efficacy with the 300-mg versus 600-mg doses and results of the skin biopsies will be presented at the 2024 annual meeting of the American Academy of Dermatology, he said. Patients who have not had recurrences are being followed up to week 100 (unblinded).
Skin biopsy findings together with levels of clearance will “add insight as to whether high-dose IL-23 inhibition is associated with higher levels of complete clearance (i.e., PASI 100) over long periods of time, and whether remissions are associated with more profound knock-down of Trm cells,” Dr. Blauvelt wrote in an editorial on Trm cells in psoriasis published in the Journal of Psoriasis and Psoriatic Arthritis.
Risankizumab was approved by the Food and Drug Administration for moderate to severe plaque psoriasis in 2019, and for active psoriatic arthritis in 2022.
Impact of IL-23 inhibition
A 2021 study, Dr. Blauvelt noted in his presentation, showed that the IL-23 inhibitor guselkumab (Tremfya) reduced the number of Trm cells in healed psoriatic skin, while the IL-17A inhibitor secukinumab (Cosentyx) did not affect Trm cells.
Other researchers have concluded that local IL-23 is required for the proliferation and retention of Trm cells in the skin, he said, noting also that, as a class, IL-23 blockers “are associated with the longest disease-free intervals” in patients with psoriasis.
Research showing loss of Trm cells in biopsies of cleared skin is “incredibly important,” as is data showing that IL-23 may be “a critical survival factor” for Trm cells, said Christopher Ritchlin, MD, MPH, a rheumatologist and director of the clinical immunology research unit at the University of Rochester (N.Y.) Medical Center, who attended the NPF meeting and was asked to comment on the growing interest in Trm cells. He did not hear Dr. Blauvelt’s presentation but has followed Trm research in psoriasis.
Measuring Trm cells in synovial tissue of patients with psoriatic disease is, in fact, on the agenda of a new research team coled by Dr. Ritchlin that is part of the Autoimmune and Immune-Mediated Diseases Program of the Accelerating Medicine Partnership – a public-private partnership that aims to collect and analyze troves of biological data in order to illuminate the cellular and molecular interactions leading to autoimmune diseases.
“Resident memory T cells have been thought to be cells that are constrained to being resident in the tissue, and while that’s largely true, there’s more recent data showing that they actually can dedifferentiate and go back to the lymph node and then go back to the joint again,” Dr. Ritchlin noted in an interview. “So it’s a more complicated story,” but nonetheless targeting the cells is a concept worth exploring therapeutically.
Moving toward a cure?
The high induction doses of risankizumab in Dr. Blauvelt’s phase 2 “knockout” study were well tolerated through week 40, with safety profiles similar overall to those reported in previous studies of risankizumab and “no new safety signals,” Dr. Blauvelt said at the meeting.
(At baseline, patients had a mean disease duration of 21 years, a mean affected BSA of 21, and a mean PASI of 18.5. Seven had prior treatment with biologic medications, though not in the prior 4 months and not with risankizumab.)
In lieu of a cure, which is still possible in the next 10 years, he said, patients are eager for longer-term remission and the ability to break away from established regular dosing. After publication of a phase 1 study of risankizumab, in which a notable number of patients experienced complete skin clearance up to 1 year following a single high dose, “we started getting numerous calls in our [clinical trials] office from patients who said ‘I want to be on that drug that you give once a year,’ ” Dr. Blauvelt said. (The high dosing was not investigated any further in phase 2 and 3 research, he added.)
While the “knockout” study adopts a “hit hard” approach, it is also possible that a strategy to hit both hard and early after disease onset may induce long-term remission and/or cure of the disease. “I’m talking about using the best things first. There are hints that if we treat early, maybe we can keep [psoriasis] from becoming a chronic disease, keep it from ‘setting up shop’ if you will,” he said, noting that the “hit hard, hit early” concept is not unique to dermatology.
During a discussion period, Dr. Blauvelt said that in a future iteration of the “knockout” study, he would like to evaluate risankizumab in patients with disease duration under 12 months. It may also be valuable to look not only at Trm cells, but more broadly at other elements of the tissue architecture before and after treatment.
Among other strategies for achieving long-term remission and/or cure is the expansion of regulatory T cells, which work to “control inappropriate immune responses” and calm inflammation. Defects in the number and function of regulatory T cells are associated with psoriasis and other autoimmune diseases, and it appears in early research from other investigators that low-dose IL-2 can induce the expansion of regulatory T cells and improve psoriasis. “Keep tuned,” Dr. Blauvelt said.
Dr. Blauvelt disclosed ties with numerous pharmaceutical companies. Dr. Ritchlin had no disclosures.
AT THE NPF RESEARCH SYMPOSIUM 2023
Roflumilast side effect benefits patients with psoriasis and overweight/obesity
BERLIN – .
Reporting secondary outcomes from the investigator-led trial at the annual congress of the European Academy of Dermatology and Venereology, Alexander Egeberg, MD, PhD, DMSc, noted that “clinically significant weight loss” was seen among patients who were treated with oral roflumilast, 500 mcg once daily, versus those receiving placebo.
Indeed, after 12 weeks of therapy, one in three patients treated with oral roflumilast experienced at least a 5% drop in their baseline body weight vs no patients who received placebo (35% vs. 0%; P < .05).
Additionally, a respective 17% versus 0% of patients lost 10% or more of their body weight, and 4% versus 0% lost 15% or more of their baseline body weight at 12 weeks.
After 24 weeks’ treatment, a substantial percentage of patients still had greater than or equal to 5%, greater than or equal to 10%, or greater than or equal to 15% weight loss, at 30%, 17%, and 13% for oral roflumilast, compared with 9%, 0%, and 0% for placebo, respectively.
“We saw that the higher baseline weight correlated with the proportion of weight loss, so that the more heavy patients at baseline also were the ones who experienced the greatest weight loss,” said Dr. Egeberg, who is professor of dermatology at the University of Copenhagen and a senior consultant at the department of dermatology at Bispebjerg Hospital, Copenhagen.
A beneficial side effect in psoriasis?
“You may have heard in psoriasis about topical roflumilast, but oral roflumilast is actually also shown to be effective in treating psoriasis,” said Egeberg.
Topical roflumilast is approved in the United States and Canada for treating plaque psoriasis.
Efficacy results from the PSORRO study were published earlier this year and showed a significantly greater improvement in Psoriasis Area and Severity Index (PASI) 75 with oral roflumilast vs. placebo at 12 weeks (35% vs. 0%), with a sustained effect seen at 24 weeks (44% vs. 40%).
Weight loss was among the most common side effects seen, leading Dr. Egeberg and fellow PSORRO investigators to wonder whether this may actually be a beneficial effect in patients with psoriasis.
“Oral roflumilast is actually a drug that has been on the market for quite a number of years,” Dr. Egeberg said.
Although only currently licensed for chronic obstructive pulmonary disease (COPD) in the United States, oral roflumilast, a phosphodiesterase (PDE) 4 inhibitor, is available as a generic, “which also means that it is extremely affordable,” suggested Dr. Edeberg.
Weight loss may be a problem in patients with COPD, he acknowledged; these patients tend to be underweight as a result of their poor state of health caused by the lung condition. Weight loss could be an advantage in patients with psoriasis who are overweight or living with obesity and have poor cardiometabolic parameters.
The psoriasis treatment with oral roflumilast study
The PSORRO study was a phase 2, multicenter, placebo-controlled, randomized trial performed between 2021 and 2022. A total of 46 adults with plaque psoriasis participated; half were initially treated with oral roflumilast and half with placebo.
Treatment was double-blind for the first 12 weeks, with all patients then receiving open-label treatment with roflumilast for 12 weeks.
The primary endpoint was the proportion of patients achieving at least 75% reduction from baseline PASI (PASI75). A host of secondary endpoints were studied, including weight and cardiometabolic parameters, which Dr. Egeberg reported at the EADV meeting.
Looking at the baseline characteristics of the oral roflumilast and placebo groups, the mean age was a respective 38 and 39 years, 65% and 83% were men, and the mean starting body weight was 102 kg and 105.1 kg.
After 12 weeks of treatment, body weight fell by a mean of 5.4 kg in the oral roflumilast group, with a further decrease of 1.4 kg by 24 weeks, bringing the total average weight loss to 6.8 kg. By comparison, weight loss among those in the placebo group was 0 kg at 12 weeks and around 2 kg at 24 weeks.
The majority of participants in both groups had high baseline BMIs; 70% of those who received oral roflumilast and 61% of those who received placebo had a BMI of 30 or higher.
“We wanted to investigate the impact of body weight, [so] we didn’t allow patients to be underweight when they were included,” Dr. Egeberg explained. Thus, for inclusion, patients had to have a BMI of 20 or higher.
An “extraordinary” finding was how some patients’ weight status based on their BMI changed throughout the study.
“We could see people that went from obese class 3, all the way to obese class 1. And we could see people going from being overweight to normal weight, which is really extraordinary for patients with psoriasis,” Dr. Egeberg said.
“But most importantly,” he added, “we didn’t have any patients who became underweight, suggesting that it actually is safe to use also in normal-weight patients.”
Reduced appetite behind benefit?
Trying to see why the weight loss occurred, Dr. Egeberg noted that it looked like it could be a result of a reduced appetite.
In common with other PDE-4 inhibitors, oral roflumilast treatment was associated with gastrointestinal symptoms – nausea, diarrhea, and abdominal pain – but all of these “decrease to placebo levels again, quite quickly,” he said.
“This really suggests that it’s not because of diarrhea, it’s not because of nausea and abdominal pain; it is because of a reduced appetite that patients actually lose weight when treated with roflumilast,” Dr. Egeberg said. It’s a potential bonus for the drug’s effects on the skin and could afford clinicians an opportunity to help motivate patients to eat well when they do eat, he observed.
Other cardiometabolic parameters assessed included blood pressure, glycated hemoglobin, total cholesterol and other key lipids, creatinine, alanine aminotransferase, and high-sensitivity C-reactive protein, but there were no noteworthy differences between the groups.
Roflumilast is an inexpensive drug because it is generic, Dr. Egeberg observed, but that also means that its use is likely to be off-label.
“It will be up to the treating physician to decide if this is an optimal therapy for their patients,” he suggested.
Cardiometabolic comorbidities important to target
Obesity is a cardiometabolic comorbidity that is important to consider when treating your patients with psoriasis, Paolo Gisondi, MD, of the University of Verona (Italy), said at a separate presentation at the EADV meeting.
While not directly commenting on the roflumilast study, he noted that moderate to severe psoriasis was “frequently associated” with metabolic disorders that put people at additional risk for cardiovascular and fatty liver diseases.
The PSORRO study was an investigator-initiated and investigator-led study and received no commercial funding. Research funding came from the Danish Psoriasis Foundation, Herlev and Gentofte Hospital, and several charitable and humanitarian organizations. Dr. Egeberg acknowledged acting as the principal investigator, speaker, and/or consultant to multiple pharma companies, all of which were unrelated to the study he presented. Dr. Gisondi’s comments were from a separate presentation, and he was not involved in the study.
A version of this article first appeared on Medscape.com.
BERLIN – .
Reporting secondary outcomes from the investigator-led trial at the annual congress of the European Academy of Dermatology and Venereology, Alexander Egeberg, MD, PhD, DMSc, noted that “clinically significant weight loss” was seen among patients who were treated with oral roflumilast, 500 mcg once daily, versus those receiving placebo.
Indeed, after 12 weeks of therapy, one in three patients treated with oral roflumilast experienced at least a 5% drop in their baseline body weight vs no patients who received placebo (35% vs. 0%; P < .05).
Additionally, a respective 17% versus 0% of patients lost 10% or more of their body weight, and 4% versus 0% lost 15% or more of their baseline body weight at 12 weeks.
After 24 weeks’ treatment, a substantial percentage of patients still had greater than or equal to 5%, greater than or equal to 10%, or greater than or equal to 15% weight loss, at 30%, 17%, and 13% for oral roflumilast, compared with 9%, 0%, and 0% for placebo, respectively.
“We saw that the higher baseline weight correlated with the proportion of weight loss, so that the more heavy patients at baseline also were the ones who experienced the greatest weight loss,” said Dr. Egeberg, who is professor of dermatology at the University of Copenhagen and a senior consultant at the department of dermatology at Bispebjerg Hospital, Copenhagen.
A beneficial side effect in psoriasis?
“You may have heard in psoriasis about topical roflumilast, but oral roflumilast is actually also shown to be effective in treating psoriasis,” said Egeberg.
Topical roflumilast is approved in the United States and Canada for treating plaque psoriasis.
Efficacy results from the PSORRO study were published earlier this year and showed a significantly greater improvement in Psoriasis Area and Severity Index (PASI) 75 with oral roflumilast vs. placebo at 12 weeks (35% vs. 0%), with a sustained effect seen at 24 weeks (44% vs. 40%).
Weight loss was among the most common side effects seen, leading Dr. Egeberg and fellow PSORRO investigators to wonder whether this may actually be a beneficial effect in patients with psoriasis.
“Oral roflumilast is actually a drug that has been on the market for quite a number of years,” Dr. Egeberg said.
Although only currently licensed for chronic obstructive pulmonary disease (COPD) in the United States, oral roflumilast, a phosphodiesterase (PDE) 4 inhibitor, is available as a generic, “which also means that it is extremely affordable,” suggested Dr. Edeberg.
Weight loss may be a problem in patients with COPD, he acknowledged; these patients tend to be underweight as a result of their poor state of health caused by the lung condition. Weight loss could be an advantage in patients with psoriasis who are overweight or living with obesity and have poor cardiometabolic parameters.
The psoriasis treatment with oral roflumilast study
The PSORRO study was a phase 2, multicenter, placebo-controlled, randomized trial performed between 2021 and 2022. A total of 46 adults with plaque psoriasis participated; half were initially treated with oral roflumilast and half with placebo.
Treatment was double-blind for the first 12 weeks, with all patients then receiving open-label treatment with roflumilast for 12 weeks.
The primary endpoint was the proportion of patients achieving at least 75% reduction from baseline PASI (PASI75). A host of secondary endpoints were studied, including weight and cardiometabolic parameters, which Dr. Egeberg reported at the EADV meeting.
Looking at the baseline characteristics of the oral roflumilast and placebo groups, the mean age was a respective 38 and 39 years, 65% and 83% were men, and the mean starting body weight was 102 kg and 105.1 kg.
After 12 weeks of treatment, body weight fell by a mean of 5.4 kg in the oral roflumilast group, with a further decrease of 1.4 kg by 24 weeks, bringing the total average weight loss to 6.8 kg. By comparison, weight loss among those in the placebo group was 0 kg at 12 weeks and around 2 kg at 24 weeks.
The majority of participants in both groups had high baseline BMIs; 70% of those who received oral roflumilast and 61% of those who received placebo had a BMI of 30 or higher.
“We wanted to investigate the impact of body weight, [so] we didn’t allow patients to be underweight when they were included,” Dr. Egeberg explained. Thus, for inclusion, patients had to have a BMI of 20 or higher.
An “extraordinary” finding was how some patients’ weight status based on their BMI changed throughout the study.
“We could see people that went from obese class 3, all the way to obese class 1. And we could see people going from being overweight to normal weight, which is really extraordinary for patients with psoriasis,” Dr. Egeberg said.
“But most importantly,” he added, “we didn’t have any patients who became underweight, suggesting that it actually is safe to use also in normal-weight patients.”
Reduced appetite behind benefit?
Trying to see why the weight loss occurred, Dr. Egeberg noted that it looked like it could be a result of a reduced appetite.
In common with other PDE-4 inhibitors, oral roflumilast treatment was associated with gastrointestinal symptoms – nausea, diarrhea, and abdominal pain – but all of these “decrease to placebo levels again, quite quickly,” he said.
“This really suggests that it’s not because of diarrhea, it’s not because of nausea and abdominal pain; it is because of a reduced appetite that patients actually lose weight when treated with roflumilast,” Dr. Egeberg said. It’s a potential bonus for the drug’s effects on the skin and could afford clinicians an opportunity to help motivate patients to eat well when they do eat, he observed.
Other cardiometabolic parameters assessed included blood pressure, glycated hemoglobin, total cholesterol and other key lipids, creatinine, alanine aminotransferase, and high-sensitivity C-reactive protein, but there were no noteworthy differences between the groups.
Roflumilast is an inexpensive drug because it is generic, Dr. Egeberg observed, but that also means that its use is likely to be off-label.
“It will be up to the treating physician to decide if this is an optimal therapy for their patients,” he suggested.
Cardiometabolic comorbidities important to target
Obesity is a cardiometabolic comorbidity that is important to consider when treating your patients with psoriasis, Paolo Gisondi, MD, of the University of Verona (Italy), said at a separate presentation at the EADV meeting.
While not directly commenting on the roflumilast study, he noted that moderate to severe psoriasis was “frequently associated” with metabolic disorders that put people at additional risk for cardiovascular and fatty liver diseases.
The PSORRO study was an investigator-initiated and investigator-led study and received no commercial funding. Research funding came from the Danish Psoriasis Foundation, Herlev and Gentofte Hospital, and several charitable and humanitarian organizations. Dr. Egeberg acknowledged acting as the principal investigator, speaker, and/or consultant to multiple pharma companies, all of which were unrelated to the study he presented. Dr. Gisondi’s comments were from a separate presentation, and he was not involved in the study.
A version of this article first appeared on Medscape.com.
BERLIN – .
Reporting secondary outcomes from the investigator-led trial at the annual congress of the European Academy of Dermatology and Venereology, Alexander Egeberg, MD, PhD, DMSc, noted that “clinically significant weight loss” was seen among patients who were treated with oral roflumilast, 500 mcg once daily, versus those receiving placebo.
Indeed, after 12 weeks of therapy, one in three patients treated with oral roflumilast experienced at least a 5% drop in their baseline body weight vs no patients who received placebo (35% vs. 0%; P < .05).
Additionally, a respective 17% versus 0% of patients lost 10% or more of their body weight, and 4% versus 0% lost 15% or more of their baseline body weight at 12 weeks.
After 24 weeks’ treatment, a substantial percentage of patients still had greater than or equal to 5%, greater than or equal to 10%, or greater than or equal to 15% weight loss, at 30%, 17%, and 13% for oral roflumilast, compared with 9%, 0%, and 0% for placebo, respectively.
“We saw that the higher baseline weight correlated with the proportion of weight loss, so that the more heavy patients at baseline also were the ones who experienced the greatest weight loss,” said Dr. Egeberg, who is professor of dermatology at the University of Copenhagen and a senior consultant at the department of dermatology at Bispebjerg Hospital, Copenhagen.
A beneficial side effect in psoriasis?
“You may have heard in psoriasis about topical roflumilast, but oral roflumilast is actually also shown to be effective in treating psoriasis,” said Egeberg.
Topical roflumilast is approved in the United States and Canada for treating plaque psoriasis.
Efficacy results from the PSORRO study were published earlier this year and showed a significantly greater improvement in Psoriasis Area and Severity Index (PASI) 75 with oral roflumilast vs. placebo at 12 weeks (35% vs. 0%), with a sustained effect seen at 24 weeks (44% vs. 40%).
Weight loss was among the most common side effects seen, leading Dr. Egeberg and fellow PSORRO investigators to wonder whether this may actually be a beneficial effect in patients with psoriasis.
“Oral roflumilast is actually a drug that has been on the market for quite a number of years,” Dr. Egeberg said.
Although only currently licensed for chronic obstructive pulmonary disease (COPD) in the United States, oral roflumilast, a phosphodiesterase (PDE) 4 inhibitor, is available as a generic, “which also means that it is extremely affordable,” suggested Dr. Edeberg.
Weight loss may be a problem in patients with COPD, he acknowledged; these patients tend to be underweight as a result of their poor state of health caused by the lung condition. Weight loss could be an advantage in patients with psoriasis who are overweight or living with obesity and have poor cardiometabolic parameters.
The psoriasis treatment with oral roflumilast study
The PSORRO study was a phase 2, multicenter, placebo-controlled, randomized trial performed between 2021 and 2022. A total of 46 adults with plaque psoriasis participated; half were initially treated with oral roflumilast and half with placebo.
Treatment was double-blind for the first 12 weeks, with all patients then receiving open-label treatment with roflumilast for 12 weeks.
The primary endpoint was the proportion of patients achieving at least 75% reduction from baseline PASI (PASI75). A host of secondary endpoints were studied, including weight and cardiometabolic parameters, which Dr. Egeberg reported at the EADV meeting.
Looking at the baseline characteristics of the oral roflumilast and placebo groups, the mean age was a respective 38 and 39 years, 65% and 83% were men, and the mean starting body weight was 102 kg and 105.1 kg.
After 12 weeks of treatment, body weight fell by a mean of 5.4 kg in the oral roflumilast group, with a further decrease of 1.4 kg by 24 weeks, bringing the total average weight loss to 6.8 kg. By comparison, weight loss among those in the placebo group was 0 kg at 12 weeks and around 2 kg at 24 weeks.
The majority of participants in both groups had high baseline BMIs; 70% of those who received oral roflumilast and 61% of those who received placebo had a BMI of 30 or higher.
“We wanted to investigate the impact of body weight, [so] we didn’t allow patients to be underweight when they were included,” Dr. Egeberg explained. Thus, for inclusion, patients had to have a BMI of 20 or higher.
An “extraordinary” finding was how some patients’ weight status based on their BMI changed throughout the study.
“We could see people that went from obese class 3, all the way to obese class 1. And we could see people going from being overweight to normal weight, which is really extraordinary for patients with psoriasis,” Dr. Egeberg said.
“But most importantly,” he added, “we didn’t have any patients who became underweight, suggesting that it actually is safe to use also in normal-weight patients.”
Reduced appetite behind benefit?
Trying to see why the weight loss occurred, Dr. Egeberg noted that it looked like it could be a result of a reduced appetite.
In common with other PDE-4 inhibitors, oral roflumilast treatment was associated with gastrointestinal symptoms – nausea, diarrhea, and abdominal pain – but all of these “decrease to placebo levels again, quite quickly,” he said.
“This really suggests that it’s not because of diarrhea, it’s not because of nausea and abdominal pain; it is because of a reduced appetite that patients actually lose weight when treated with roflumilast,” Dr. Egeberg said. It’s a potential bonus for the drug’s effects on the skin and could afford clinicians an opportunity to help motivate patients to eat well when they do eat, he observed.
Other cardiometabolic parameters assessed included blood pressure, glycated hemoglobin, total cholesterol and other key lipids, creatinine, alanine aminotransferase, and high-sensitivity C-reactive protein, but there were no noteworthy differences between the groups.
Roflumilast is an inexpensive drug because it is generic, Dr. Egeberg observed, but that also means that its use is likely to be off-label.
“It will be up to the treating physician to decide if this is an optimal therapy for their patients,” he suggested.
Cardiometabolic comorbidities important to target
Obesity is a cardiometabolic comorbidity that is important to consider when treating your patients with psoriasis, Paolo Gisondi, MD, of the University of Verona (Italy), said at a separate presentation at the EADV meeting.
While not directly commenting on the roflumilast study, he noted that moderate to severe psoriasis was “frequently associated” with metabolic disorders that put people at additional risk for cardiovascular and fatty liver diseases.
The PSORRO study was an investigator-initiated and investigator-led study and received no commercial funding. Research funding came from the Danish Psoriasis Foundation, Herlev and Gentofte Hospital, and several charitable and humanitarian organizations. Dr. Egeberg acknowledged acting as the principal investigator, speaker, and/or consultant to multiple pharma companies, all of which were unrelated to the study he presented. Dr. Gisondi’s comments were from a separate presentation, and he was not involved in the study.
A version of this article first appeared on Medscape.com.
AT THE EADV CONGRESS
Topical botanical drug coacillium curbs childhood alopecia
Considerable hair regrowth can be achieved in children with alopecia areata with the use of a novel plant-based drug, according to research presented during the first late-breaking news session at the annual congress of the European Academy of Dermatology and Venereology.
(–8.0%), with a significant 31% overall difference (P < .0001).
“Coacillium cutaneous solution was used for the first time for treatment of alopecia areata and also for the first time used in a pediatric population,” the presenting investigator Ulrike Blume-Peytavi, MD, said at the meeting.
“It’s well tolerated, and in fact what is interesting is, it has a durable response, even after treatment discontinuation,” added Dr. Blume-Peytavi, who is the deputy head of the department of dermatology, venereology and allergology at Charité-Universitätsmedizin Berlin.
Backing the botanical?
Paola Pasquali, MD, a dermatologist at Pius Hospital de Valls in Spain, who cochaired the session where the findings were presented, commented, “Thank you for showing that chocolate is great! I knew it. It is fantastic to see how chocolate is used.”
Dr. Pasquali was referring to the coacillium ingredient Theobroma cacao extract. The seeds of T. cacao, or the cocoa tree, are used to make various types of chocolate products. Theobroma cacao is one of four plant extracts that make up coacillium, the others being Allium cepa (onion), Citrus limon (lemon), and Paullinia cupana (guaraná, a source of caffeine).
The four plant extracts are classified as “generally regarded as safe” (GRAS), Dr. Blume-Peytavi observed, noting that the development of coacillium fell under the category of a prescription botanical drug as set out by the U.S. Food and Drug Administration or a herbal medicinal product as set out by the European Medicines Agency.
But how does it work?
The botanical’s mode of action of acting positively on hair follicle cycling and endothelial cell activation was called into question, however, by Emma Guttman-Yassky, MD, PhD, who was in the audience.
She asked, “So how do you explain that, after three large studies with topical JAK inhibitors that did not work actually in alopecia areata because it’s very hard to penetrate the scalp for a topical [drug], this one works?”
Dr. Guttman-Yassky, professor of dermatology and immunology at the Icahn School of Medicine at Mount Sinai, New York, added: “Looking at the ingredients, to me, it seems that it’s more like a DPCP [diphenylcyclopropenone]-like reaction.”
DPCP, which has been used to treat alopecia, purportedly works by stimulating the immune response to target the skin surface – causing an allergic reaction – rather than the hair follicle.
It’s an interesting question as to how a molecule penetrates the hair follicle, and it depends on the size of the molecule, Dr. Blume-Peytavi responded.
“We have done a lot of studies on follicular penetration, and we are quite aware that you need a certain size of the molecule,” she said. Between 14 and 200 nanometers appears to produce “the best penetrators,” she observed.
Dr. Blume-Peytavi commented that even after topical JAK inhibitors are applied, the molecules that penetrate do not remain in the local area for very long, yet still produce an inhibitory signaling effect.
No scalp irritation was seen in the trial, which suggests that coacillium is not working in the same way as DPCP, Dr. Blume-Peytavi countered.
Evaluating efficacy and safety: The RAAINBOW study
Dr. Blume-Peytavi acknowledged that JAK inhibitors were “a tremendous advance in treating severe and very severe alopecia areata,” but because of their benefit-to-risk ratio, there was still an unmet need for new treatments, particularly in children, in whom drug safety is of critical importance.
Having a drug that could be given safely and also have an effect early on in the disease, while it is still at a mild to moderate stage, would be of considerable value, Dr. Blume-Peytavi maintained.
The RAAINBOW study was a randomized, double-blind, phase 2/3 trial conducted at 12 sites in Germany and three other countries between March 2018 and March 2022 to evaluate the efficacy and safety of coacillium in the treatment of children and adolescents with moderate to severe alopecia areata.
In all, 62 children aged 2-18 years (mean age, 11 years) participated; 42 were treated twice daily with coacillium cutaneous solution 22.5% and 20 received placebo for 24 weeks. Treatment was then stopped, and participants followed for another 24 weeks off treatment to check for disease relapse, bringing the total study duration up to 48 weeks.
Baseline characteristics were “relatively comparable for severity,” Dr. Blume-Peytavi said. Most of the children had severe alopecia areata (57% for coacillium and 65% for placebo); the remainder had moderate disease (43% vs. 35%, respectively).
The average SALT scores at the start of treatment were 56 in the coacillium group and 62 in the placebo group, and a respective 44 and 61 at the end of 24 weeks’ treatment.
Perhaps the most important results, Dr. Blume-Peytavi said, was that at 48 weeks of follow-up, which was 24 weeks after treatment had been discontinued, the mean SALT scores were 29 for coacillium and 56 for placebo (P < .0001).
“You can see the improvement in the treated group is continuing even without treatment. However, the placebo group stays relatively about the same range,” she said.
Overall, 82% of patients treated with coacillium and 37% of those who received placebo experienced hair growth after treatment had stopped, and by week 48, a respective 46.7% vs. 9.1% had a SALT score of 20 or less, and 30.0% vs. 0% had a SALT score of 10 or less.
No safety concerns were raised, with no serious treatment-related reactions, no immunosuppressant-like reactions, and no steroidlike side effects.
Beyond the RAAINBOW
Larger studies are needed, Dr. Blume-Peytavi said. According to developer Legacy Healthcare’s website, coacillium cutaneous solution is not being developed just for childhood alopecia areata. It is also under investigation as a treatment for persistent chemotherapy-induced alopecia, atopic dermatitis, and psoriasis. In addition, an oral solution is being tested for cancer-related fatigue.
The study was funded by Legacy Healthcare. Dr. Blume-Peytavi has received research funding and acts as an advisor to the company, among others; four of the study’s coauthors are employees of the company. Dr. Pasquali and Dr. Guttman-Yassky were not involved in the study and had no relevant financial ties to disclose.
A version of this article first appeared on Medscape.com.
Considerable hair regrowth can be achieved in children with alopecia areata with the use of a novel plant-based drug, according to research presented during the first late-breaking news session at the annual congress of the European Academy of Dermatology and Venereology.
(–8.0%), with a significant 31% overall difference (P < .0001).
“Coacillium cutaneous solution was used for the first time for treatment of alopecia areata and also for the first time used in a pediatric population,” the presenting investigator Ulrike Blume-Peytavi, MD, said at the meeting.
“It’s well tolerated, and in fact what is interesting is, it has a durable response, even after treatment discontinuation,” added Dr. Blume-Peytavi, who is the deputy head of the department of dermatology, venereology and allergology at Charité-Universitätsmedizin Berlin.
Backing the botanical?
Paola Pasquali, MD, a dermatologist at Pius Hospital de Valls in Spain, who cochaired the session where the findings were presented, commented, “Thank you for showing that chocolate is great! I knew it. It is fantastic to see how chocolate is used.”
Dr. Pasquali was referring to the coacillium ingredient Theobroma cacao extract. The seeds of T. cacao, or the cocoa tree, are used to make various types of chocolate products. Theobroma cacao is one of four plant extracts that make up coacillium, the others being Allium cepa (onion), Citrus limon (lemon), and Paullinia cupana (guaraná, a source of caffeine).
The four plant extracts are classified as “generally regarded as safe” (GRAS), Dr. Blume-Peytavi observed, noting that the development of coacillium fell under the category of a prescription botanical drug as set out by the U.S. Food and Drug Administration or a herbal medicinal product as set out by the European Medicines Agency.
But how does it work?
The botanical’s mode of action of acting positively on hair follicle cycling and endothelial cell activation was called into question, however, by Emma Guttman-Yassky, MD, PhD, who was in the audience.
She asked, “So how do you explain that, after three large studies with topical JAK inhibitors that did not work actually in alopecia areata because it’s very hard to penetrate the scalp for a topical [drug], this one works?”
Dr. Guttman-Yassky, professor of dermatology and immunology at the Icahn School of Medicine at Mount Sinai, New York, added: “Looking at the ingredients, to me, it seems that it’s more like a DPCP [diphenylcyclopropenone]-like reaction.”
DPCP, which has been used to treat alopecia, purportedly works by stimulating the immune response to target the skin surface – causing an allergic reaction – rather than the hair follicle.
It’s an interesting question as to how a molecule penetrates the hair follicle, and it depends on the size of the molecule, Dr. Blume-Peytavi responded.
“We have done a lot of studies on follicular penetration, and we are quite aware that you need a certain size of the molecule,” she said. Between 14 and 200 nanometers appears to produce “the best penetrators,” she observed.
Dr. Blume-Peytavi commented that even after topical JAK inhibitors are applied, the molecules that penetrate do not remain in the local area for very long, yet still produce an inhibitory signaling effect.
No scalp irritation was seen in the trial, which suggests that coacillium is not working in the same way as DPCP, Dr. Blume-Peytavi countered.
Evaluating efficacy and safety: The RAAINBOW study
Dr. Blume-Peytavi acknowledged that JAK inhibitors were “a tremendous advance in treating severe and very severe alopecia areata,” but because of their benefit-to-risk ratio, there was still an unmet need for new treatments, particularly in children, in whom drug safety is of critical importance.
Having a drug that could be given safely and also have an effect early on in the disease, while it is still at a mild to moderate stage, would be of considerable value, Dr. Blume-Peytavi maintained.
The RAAINBOW study was a randomized, double-blind, phase 2/3 trial conducted at 12 sites in Germany and three other countries between March 2018 and March 2022 to evaluate the efficacy and safety of coacillium in the treatment of children and adolescents with moderate to severe alopecia areata.
In all, 62 children aged 2-18 years (mean age, 11 years) participated; 42 were treated twice daily with coacillium cutaneous solution 22.5% and 20 received placebo for 24 weeks. Treatment was then stopped, and participants followed for another 24 weeks off treatment to check for disease relapse, bringing the total study duration up to 48 weeks.
Baseline characteristics were “relatively comparable for severity,” Dr. Blume-Peytavi said. Most of the children had severe alopecia areata (57% for coacillium and 65% for placebo); the remainder had moderate disease (43% vs. 35%, respectively).
The average SALT scores at the start of treatment were 56 in the coacillium group and 62 in the placebo group, and a respective 44 and 61 at the end of 24 weeks’ treatment.
Perhaps the most important results, Dr. Blume-Peytavi said, was that at 48 weeks of follow-up, which was 24 weeks after treatment had been discontinued, the mean SALT scores were 29 for coacillium and 56 for placebo (P < .0001).
“You can see the improvement in the treated group is continuing even without treatment. However, the placebo group stays relatively about the same range,” she said.
Overall, 82% of patients treated with coacillium and 37% of those who received placebo experienced hair growth after treatment had stopped, and by week 48, a respective 46.7% vs. 9.1% had a SALT score of 20 or less, and 30.0% vs. 0% had a SALT score of 10 or less.
No safety concerns were raised, with no serious treatment-related reactions, no immunosuppressant-like reactions, and no steroidlike side effects.
Beyond the RAAINBOW
Larger studies are needed, Dr. Blume-Peytavi said. According to developer Legacy Healthcare’s website, coacillium cutaneous solution is not being developed just for childhood alopecia areata. It is also under investigation as a treatment for persistent chemotherapy-induced alopecia, atopic dermatitis, and psoriasis. In addition, an oral solution is being tested for cancer-related fatigue.
The study was funded by Legacy Healthcare. Dr. Blume-Peytavi has received research funding and acts as an advisor to the company, among others; four of the study’s coauthors are employees of the company. Dr. Pasquali and Dr. Guttman-Yassky were not involved in the study and had no relevant financial ties to disclose.
A version of this article first appeared on Medscape.com.
Considerable hair regrowth can be achieved in children with alopecia areata with the use of a novel plant-based drug, according to research presented during the first late-breaking news session at the annual congress of the European Academy of Dermatology and Venereology.
(–8.0%), with a significant 31% overall difference (P < .0001).
“Coacillium cutaneous solution was used for the first time for treatment of alopecia areata and also for the first time used in a pediatric population,” the presenting investigator Ulrike Blume-Peytavi, MD, said at the meeting.
“It’s well tolerated, and in fact what is interesting is, it has a durable response, even after treatment discontinuation,” added Dr. Blume-Peytavi, who is the deputy head of the department of dermatology, venereology and allergology at Charité-Universitätsmedizin Berlin.
Backing the botanical?
Paola Pasquali, MD, a dermatologist at Pius Hospital de Valls in Spain, who cochaired the session where the findings were presented, commented, “Thank you for showing that chocolate is great! I knew it. It is fantastic to see how chocolate is used.”
Dr. Pasquali was referring to the coacillium ingredient Theobroma cacao extract. The seeds of T. cacao, or the cocoa tree, are used to make various types of chocolate products. Theobroma cacao is one of four plant extracts that make up coacillium, the others being Allium cepa (onion), Citrus limon (lemon), and Paullinia cupana (guaraná, a source of caffeine).
The four plant extracts are classified as “generally regarded as safe” (GRAS), Dr. Blume-Peytavi observed, noting that the development of coacillium fell under the category of a prescription botanical drug as set out by the U.S. Food and Drug Administration or a herbal medicinal product as set out by the European Medicines Agency.
But how does it work?
The botanical’s mode of action of acting positively on hair follicle cycling and endothelial cell activation was called into question, however, by Emma Guttman-Yassky, MD, PhD, who was in the audience.
She asked, “So how do you explain that, after three large studies with topical JAK inhibitors that did not work actually in alopecia areata because it’s very hard to penetrate the scalp for a topical [drug], this one works?”
Dr. Guttman-Yassky, professor of dermatology and immunology at the Icahn School of Medicine at Mount Sinai, New York, added: “Looking at the ingredients, to me, it seems that it’s more like a DPCP [diphenylcyclopropenone]-like reaction.”
DPCP, which has been used to treat alopecia, purportedly works by stimulating the immune response to target the skin surface – causing an allergic reaction – rather than the hair follicle.
It’s an interesting question as to how a molecule penetrates the hair follicle, and it depends on the size of the molecule, Dr. Blume-Peytavi responded.
“We have done a lot of studies on follicular penetration, and we are quite aware that you need a certain size of the molecule,” she said. Between 14 and 200 nanometers appears to produce “the best penetrators,” she observed.
Dr. Blume-Peytavi commented that even after topical JAK inhibitors are applied, the molecules that penetrate do not remain in the local area for very long, yet still produce an inhibitory signaling effect.
No scalp irritation was seen in the trial, which suggests that coacillium is not working in the same way as DPCP, Dr. Blume-Peytavi countered.
Evaluating efficacy and safety: The RAAINBOW study
Dr. Blume-Peytavi acknowledged that JAK inhibitors were “a tremendous advance in treating severe and very severe alopecia areata,” but because of their benefit-to-risk ratio, there was still an unmet need for new treatments, particularly in children, in whom drug safety is of critical importance.
Having a drug that could be given safely and also have an effect early on in the disease, while it is still at a mild to moderate stage, would be of considerable value, Dr. Blume-Peytavi maintained.
The RAAINBOW study was a randomized, double-blind, phase 2/3 trial conducted at 12 sites in Germany and three other countries between March 2018 and March 2022 to evaluate the efficacy and safety of coacillium in the treatment of children and adolescents with moderate to severe alopecia areata.
In all, 62 children aged 2-18 years (mean age, 11 years) participated; 42 were treated twice daily with coacillium cutaneous solution 22.5% and 20 received placebo for 24 weeks. Treatment was then stopped, and participants followed for another 24 weeks off treatment to check for disease relapse, bringing the total study duration up to 48 weeks.
Baseline characteristics were “relatively comparable for severity,” Dr. Blume-Peytavi said. Most of the children had severe alopecia areata (57% for coacillium and 65% for placebo); the remainder had moderate disease (43% vs. 35%, respectively).
The average SALT scores at the start of treatment were 56 in the coacillium group and 62 in the placebo group, and a respective 44 and 61 at the end of 24 weeks’ treatment.
Perhaps the most important results, Dr. Blume-Peytavi said, was that at 48 weeks of follow-up, which was 24 weeks after treatment had been discontinued, the mean SALT scores were 29 for coacillium and 56 for placebo (P < .0001).
“You can see the improvement in the treated group is continuing even without treatment. However, the placebo group stays relatively about the same range,” she said.
Overall, 82% of patients treated with coacillium and 37% of those who received placebo experienced hair growth after treatment had stopped, and by week 48, a respective 46.7% vs. 9.1% had a SALT score of 20 or less, and 30.0% vs. 0% had a SALT score of 10 or less.
No safety concerns were raised, with no serious treatment-related reactions, no immunosuppressant-like reactions, and no steroidlike side effects.
Beyond the RAAINBOW
Larger studies are needed, Dr. Blume-Peytavi said. According to developer Legacy Healthcare’s website, coacillium cutaneous solution is not being developed just for childhood alopecia areata. It is also under investigation as a treatment for persistent chemotherapy-induced alopecia, atopic dermatitis, and psoriasis. In addition, an oral solution is being tested for cancer-related fatigue.
The study was funded by Legacy Healthcare. Dr. Blume-Peytavi has received research funding and acts as an advisor to the company, among others; four of the study’s coauthors are employees of the company. Dr. Pasquali and Dr. Guttman-Yassky were not involved in the study and had no relevant financial ties to disclose.
A version of this article first appeared on Medscape.com.
AT THE EADV CONGRESS
Pediatric psoriasis: Black children, males more likely to have palmoplantar subtype, study finds
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Intravenous formulation of secukinumab gets FDA approval
The Food and Drug Administration has approved an intravenous (IV) formulation of secukinumab (Cosentyx) for the treatment of adults with psoriatic arthritis (PsA), ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis (nr-axSpA).
Secukinumab is the only treatment approved in an IV formulation that specifically targets and blocks interleukin-17A and the only non–tumor necrosis factor alpha IV option available to treat the three indications of PsA, AS, and nr-axSpA, according to a press release from the drug’s manufacturer, Novartis.
The approval marks the first new IV treatment in 6 years for these three conditions. The drug was first approved in 2015 and up to now has been available only as a subcutaneous injection.
The new formulation is also approved for secukinumab’s other indications of plaque psoriasis in people aged 6 years or older, children aged 2 years or older with PsA, and enthesitis-related arthritis in patients aged 4 years or older.
“A significant portion of the millions of PsA, AS, and nr-axSpA patients in the United States require treatment through IV infusions for a variety of reasons, including not being comfortable with self-injections or simply preferring to have treatments administered in their health care provider’s office,” Philip J. Mease, MD, clinical professor at the University of Washington, Seattle, and director of rheumatology research at the Swedish Medical Center, Seattle, said in the press release. “The approval of Cosentyx as an IV formulation is an important milestone for patients because it expands the treatment options available to them with a different mechanism of action than existing biologic IV therapies, along with the comfort and familiarity of an established treatment.”
This IV formulation is administered monthly in a 30-minute, weight-based dosing regimen. This new option will become available before the end of the year, Novartis said.
“With this approval of Cosentyx as an IV formulation, along with the subcutaneous formulation, we can broaden the use of Cosentyx to help more patients manage their condition with a medicine backed by more than a decade of clinical research and 8 years of real-world experience,” said Christy Siegel, vice president and head of immunology, Novartis U.S.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved an intravenous (IV) formulation of secukinumab (Cosentyx) for the treatment of adults with psoriatic arthritis (PsA), ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis (nr-axSpA).
Secukinumab is the only treatment approved in an IV formulation that specifically targets and blocks interleukin-17A and the only non–tumor necrosis factor alpha IV option available to treat the three indications of PsA, AS, and nr-axSpA, according to a press release from the drug’s manufacturer, Novartis.
The approval marks the first new IV treatment in 6 years for these three conditions. The drug was first approved in 2015 and up to now has been available only as a subcutaneous injection.
The new formulation is also approved for secukinumab’s other indications of plaque psoriasis in people aged 6 years or older, children aged 2 years or older with PsA, and enthesitis-related arthritis in patients aged 4 years or older.
“A significant portion of the millions of PsA, AS, and nr-axSpA patients in the United States require treatment through IV infusions for a variety of reasons, including not being comfortable with self-injections or simply preferring to have treatments administered in their health care provider’s office,” Philip J. Mease, MD, clinical professor at the University of Washington, Seattle, and director of rheumatology research at the Swedish Medical Center, Seattle, said in the press release. “The approval of Cosentyx as an IV formulation is an important milestone for patients because it expands the treatment options available to them with a different mechanism of action than existing biologic IV therapies, along with the comfort and familiarity of an established treatment.”
This IV formulation is administered monthly in a 30-minute, weight-based dosing regimen. This new option will become available before the end of the year, Novartis said.
“With this approval of Cosentyx as an IV formulation, along with the subcutaneous formulation, we can broaden the use of Cosentyx to help more patients manage their condition with a medicine backed by more than a decade of clinical research and 8 years of real-world experience,” said Christy Siegel, vice president and head of immunology, Novartis U.S.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved an intravenous (IV) formulation of secukinumab (Cosentyx) for the treatment of adults with psoriatic arthritis (PsA), ankylosing spondylitis (AS), and nonradiographic axial spondyloarthritis (nr-axSpA).
Secukinumab is the only treatment approved in an IV formulation that specifically targets and blocks interleukin-17A and the only non–tumor necrosis factor alpha IV option available to treat the three indications of PsA, AS, and nr-axSpA, according to a press release from the drug’s manufacturer, Novartis.
The approval marks the first new IV treatment in 6 years for these three conditions. The drug was first approved in 2015 and up to now has been available only as a subcutaneous injection.
The new formulation is also approved for secukinumab’s other indications of plaque psoriasis in people aged 6 years or older, children aged 2 years or older with PsA, and enthesitis-related arthritis in patients aged 4 years or older.
“A significant portion of the millions of PsA, AS, and nr-axSpA patients in the United States require treatment through IV infusions for a variety of reasons, including not being comfortable with self-injections or simply preferring to have treatments administered in their health care provider’s office,” Philip J. Mease, MD, clinical professor at the University of Washington, Seattle, and director of rheumatology research at the Swedish Medical Center, Seattle, said in the press release. “The approval of Cosentyx as an IV formulation is an important milestone for patients because it expands the treatment options available to them with a different mechanism of action than existing biologic IV therapies, along with the comfort and familiarity of an established treatment.”
This IV formulation is administered monthly in a 30-minute, weight-based dosing regimen. This new option will become available before the end of the year, Novartis said.
“With this approval of Cosentyx as an IV formulation, along with the subcutaneous formulation, we can broaden the use of Cosentyx to help more patients manage their condition with a medicine backed by more than a decade of clinical research and 8 years of real-world experience,” said Christy Siegel, vice president and head of immunology, Novartis U.S.
A version of this article first appeared on Medscape.com.
Choosing which biologic to prescribe for psoriasis
CARLSBAD, CALIF. –
“When you look at the list of options it can be confusing to many clinicians in deciding which one to choose,” April W. Armstrong, MD, MPH, professor and chief of dermatology at the University of California, Los Angeles, said at the annual symposium of the California Society of Dermatology & Dermatologic Surgery.
One approach is to consider how the biologics compare in short- and long-term efficacy. “Several different meta-analyses of biologics have been conducted,” which include some head-to head studies, Dr. Armstrong said. “In terms of efficacy, [biologics] are similar at the population level,” she said.
In a meta-analysis of 71 randomized, controlled trials through July 2020, Dr. Armstrong and colleagues found that in the short-term, Psoriasis Area and Severity Index (PASI) 90 response rates at 10-16 weeks from baseline were highest for ixekizumab (72.9%), risankizumab (72.5%), and brodalumab (72%). These PASI 90 responses were significantly higher than among patients on guselkumab (65%), secukinumab (65%), infliximab (56.8%), certolizumab (400 mg: 49.6%; 200 mg: 42.2%), ustekinumab (90 mg: 47.9%; weight-based: 45.7%; 45 mg: 44.6%), adalimumab (43%), tildrakizumab (200 mg: 39.7%; 100 mg: 37.2%), etanercept (18.0%), apremilast (12.4%), and dimethyl fumarate (12.2%).
In a more recent meta-analysis, Dr. Armstrong and coauthors used area under the curve (AUC) analyses to compare the cumulative clinical benefits of biologics over 1 year. They found that the placebo-adjusted normalized maximum AUC for a PASI 100 response was greatest for ixekizumab (0.436), risankizumab (0.423), and brodalumab (0.378), followed by guselkumab (0.358), secukinumab (0.324), ustekinumab (0.201), adalimumab (0.183), and etanercept (0.087).
In Dr. Armstrong’s opinion, the tumor necrosis factor (TNF) inhibitors etanercept, infliximab, adalimumab, and certolizumab “have served their purpose for plaque psoriasis over time, but these days I would probably choose either an IL [interleukin]-17 inhibitor or an IL-23 inhibitor first,” she said. Still, TNF inhibitors “are certainly good for psoriatic arthritis, and certolizumab is appropriate for patients who are pregnant or breastfeeding,” she said. “Avoid them in patients with demyelinating disease and in those with hepatitis B. They are not preferred in patients with latent TB or advanced CHF.”
Dr. Armstrong said that there are robust efficacy data for the IL-17 inhibitors ixekizumab, secukinumab, and brodalumab in psoriasis and in the peripheral and axial forms of psoriatic arthritis (PsA). “Avoid using them in patients with a personal history of inflammatory bowel disease,” she advised.
Low rates of oral candidiasis have been reported in the literature, “but this has not been issue with our approved IL-17 inhibitors so far,” she said.
The IL-23 inhibitors guselkumab, risankizumab, tildrakizumab, and ustekinumab have robust data for psoriasis efficacy, she said, and three – guselkumab, risankizumab, and ustekinumab – are also approved for PsA. “These agents have the advantage of fewer injections, and the evidence [of efficacy] for IL-23 inhibitors continues to evolve, such as in patients with psoriatic arthritis involving the spine,” Dr. Armstrong said.
She also shared how she deals with patients who fail to respond to biologics. “Do you switch drugs, or do you dose escalate?” she asked. “In most cases, the strategy for dose escalation is to shorten the interval between the injections so the dosing is delivered more frequently.” In a case of primary failure, which Dr. Armstrong defined as a patient who has never responded optimally to a biologic, consider revisiting the diagnosis. “Maybe it’s cutaneous T-cell lymphoma or some other condition, because our current IL-17 and IL-23 medications work extremely well,” she said. “So, if you have a patient who is not responding at all, I would question the diagnosis and consider a biopsy.”
She generally waits about 6 months before switching a patient to another biologic, “to see if they’re one of the late bloomers who may catch up in efficacy,” she explained. “Switching the class of biologic is another consideration.”
If a patient had responded to the biologic for a long time and then lost response – known as secondary failure – Dr. Armstrong considers dose escalation or a switch to another agent within the same class “if it helps to address comorbidities such as PsA,” she said. “You can also try across-class switching.”
Dr. Armstrong disclosed ties with AbbVie, Arcutis, ASLAN, Beiersdorf, Boehringer Ingelheim, Bristol Myers Squibb, Dermira, Dermavant, EPI, Galderma, InCyte, Janssen, Leo, Lilly, Meiji, Modmed, Nimbus, Novartis, Ortho Dermatologics, Parexel, Pfizer, Regeneron, Sanofi, Suna, UCB, and Ventyx.
CARLSBAD, CALIF. –
“When you look at the list of options it can be confusing to many clinicians in deciding which one to choose,” April W. Armstrong, MD, MPH, professor and chief of dermatology at the University of California, Los Angeles, said at the annual symposium of the California Society of Dermatology & Dermatologic Surgery.
One approach is to consider how the biologics compare in short- and long-term efficacy. “Several different meta-analyses of biologics have been conducted,” which include some head-to head studies, Dr. Armstrong said. “In terms of efficacy, [biologics] are similar at the population level,” she said.
In a meta-analysis of 71 randomized, controlled trials through July 2020, Dr. Armstrong and colleagues found that in the short-term, Psoriasis Area and Severity Index (PASI) 90 response rates at 10-16 weeks from baseline were highest for ixekizumab (72.9%), risankizumab (72.5%), and brodalumab (72%). These PASI 90 responses were significantly higher than among patients on guselkumab (65%), secukinumab (65%), infliximab (56.8%), certolizumab (400 mg: 49.6%; 200 mg: 42.2%), ustekinumab (90 mg: 47.9%; weight-based: 45.7%; 45 mg: 44.6%), adalimumab (43%), tildrakizumab (200 mg: 39.7%; 100 mg: 37.2%), etanercept (18.0%), apremilast (12.4%), and dimethyl fumarate (12.2%).
In a more recent meta-analysis, Dr. Armstrong and coauthors used area under the curve (AUC) analyses to compare the cumulative clinical benefits of biologics over 1 year. They found that the placebo-adjusted normalized maximum AUC for a PASI 100 response was greatest for ixekizumab (0.436), risankizumab (0.423), and brodalumab (0.378), followed by guselkumab (0.358), secukinumab (0.324), ustekinumab (0.201), adalimumab (0.183), and etanercept (0.087).
In Dr. Armstrong’s opinion, the tumor necrosis factor (TNF) inhibitors etanercept, infliximab, adalimumab, and certolizumab “have served their purpose for plaque psoriasis over time, but these days I would probably choose either an IL [interleukin]-17 inhibitor or an IL-23 inhibitor first,” she said. Still, TNF inhibitors “are certainly good for psoriatic arthritis, and certolizumab is appropriate for patients who are pregnant or breastfeeding,” she said. “Avoid them in patients with demyelinating disease and in those with hepatitis B. They are not preferred in patients with latent TB or advanced CHF.”
Dr. Armstrong said that there are robust efficacy data for the IL-17 inhibitors ixekizumab, secukinumab, and brodalumab in psoriasis and in the peripheral and axial forms of psoriatic arthritis (PsA). “Avoid using them in patients with a personal history of inflammatory bowel disease,” she advised.
Low rates of oral candidiasis have been reported in the literature, “but this has not been issue with our approved IL-17 inhibitors so far,” she said.
The IL-23 inhibitors guselkumab, risankizumab, tildrakizumab, and ustekinumab have robust data for psoriasis efficacy, she said, and three – guselkumab, risankizumab, and ustekinumab – are also approved for PsA. “These agents have the advantage of fewer injections, and the evidence [of efficacy] for IL-23 inhibitors continues to evolve, such as in patients with psoriatic arthritis involving the spine,” Dr. Armstrong said.
She also shared how she deals with patients who fail to respond to biologics. “Do you switch drugs, or do you dose escalate?” she asked. “In most cases, the strategy for dose escalation is to shorten the interval between the injections so the dosing is delivered more frequently.” In a case of primary failure, which Dr. Armstrong defined as a patient who has never responded optimally to a biologic, consider revisiting the diagnosis. “Maybe it’s cutaneous T-cell lymphoma or some other condition, because our current IL-17 and IL-23 medications work extremely well,” she said. “So, if you have a patient who is not responding at all, I would question the diagnosis and consider a biopsy.”
She generally waits about 6 months before switching a patient to another biologic, “to see if they’re one of the late bloomers who may catch up in efficacy,” she explained. “Switching the class of biologic is another consideration.”
If a patient had responded to the biologic for a long time and then lost response – known as secondary failure – Dr. Armstrong considers dose escalation or a switch to another agent within the same class “if it helps to address comorbidities such as PsA,” she said. “You can also try across-class switching.”
Dr. Armstrong disclosed ties with AbbVie, Arcutis, ASLAN, Beiersdorf, Boehringer Ingelheim, Bristol Myers Squibb, Dermira, Dermavant, EPI, Galderma, InCyte, Janssen, Leo, Lilly, Meiji, Modmed, Nimbus, Novartis, Ortho Dermatologics, Parexel, Pfizer, Regeneron, Sanofi, Suna, UCB, and Ventyx.
CARLSBAD, CALIF. –
“When you look at the list of options it can be confusing to many clinicians in deciding which one to choose,” April W. Armstrong, MD, MPH, professor and chief of dermatology at the University of California, Los Angeles, said at the annual symposium of the California Society of Dermatology & Dermatologic Surgery.
One approach is to consider how the biologics compare in short- and long-term efficacy. “Several different meta-analyses of biologics have been conducted,” which include some head-to head studies, Dr. Armstrong said. “In terms of efficacy, [biologics] are similar at the population level,” she said.
In a meta-analysis of 71 randomized, controlled trials through July 2020, Dr. Armstrong and colleagues found that in the short-term, Psoriasis Area and Severity Index (PASI) 90 response rates at 10-16 weeks from baseline were highest for ixekizumab (72.9%), risankizumab (72.5%), and brodalumab (72%). These PASI 90 responses were significantly higher than among patients on guselkumab (65%), secukinumab (65%), infliximab (56.8%), certolizumab (400 mg: 49.6%; 200 mg: 42.2%), ustekinumab (90 mg: 47.9%; weight-based: 45.7%; 45 mg: 44.6%), adalimumab (43%), tildrakizumab (200 mg: 39.7%; 100 mg: 37.2%), etanercept (18.0%), apremilast (12.4%), and dimethyl fumarate (12.2%).
In a more recent meta-analysis, Dr. Armstrong and coauthors used area under the curve (AUC) analyses to compare the cumulative clinical benefits of biologics over 1 year. They found that the placebo-adjusted normalized maximum AUC for a PASI 100 response was greatest for ixekizumab (0.436), risankizumab (0.423), and brodalumab (0.378), followed by guselkumab (0.358), secukinumab (0.324), ustekinumab (0.201), adalimumab (0.183), and etanercept (0.087).
In Dr. Armstrong’s opinion, the tumor necrosis factor (TNF) inhibitors etanercept, infliximab, adalimumab, and certolizumab “have served their purpose for plaque psoriasis over time, but these days I would probably choose either an IL [interleukin]-17 inhibitor or an IL-23 inhibitor first,” she said. Still, TNF inhibitors “are certainly good for psoriatic arthritis, and certolizumab is appropriate for patients who are pregnant or breastfeeding,” she said. “Avoid them in patients with demyelinating disease and in those with hepatitis B. They are not preferred in patients with latent TB or advanced CHF.”
Dr. Armstrong said that there are robust efficacy data for the IL-17 inhibitors ixekizumab, secukinumab, and brodalumab in psoriasis and in the peripheral and axial forms of psoriatic arthritis (PsA). “Avoid using them in patients with a personal history of inflammatory bowel disease,” she advised.
Low rates of oral candidiasis have been reported in the literature, “but this has not been issue with our approved IL-17 inhibitors so far,” she said.
The IL-23 inhibitors guselkumab, risankizumab, tildrakizumab, and ustekinumab have robust data for psoriasis efficacy, she said, and three – guselkumab, risankizumab, and ustekinumab – are also approved for PsA. “These agents have the advantage of fewer injections, and the evidence [of efficacy] for IL-23 inhibitors continues to evolve, such as in patients with psoriatic arthritis involving the spine,” Dr. Armstrong said.
She also shared how she deals with patients who fail to respond to biologics. “Do you switch drugs, or do you dose escalate?” she asked. “In most cases, the strategy for dose escalation is to shorten the interval between the injections so the dosing is delivered more frequently.” In a case of primary failure, which Dr. Armstrong defined as a patient who has never responded optimally to a biologic, consider revisiting the diagnosis. “Maybe it’s cutaneous T-cell lymphoma or some other condition, because our current IL-17 and IL-23 medications work extremely well,” she said. “So, if you have a patient who is not responding at all, I would question the diagnosis and consider a biopsy.”
She generally waits about 6 months before switching a patient to another biologic, “to see if they’re one of the late bloomers who may catch up in efficacy,” she explained. “Switching the class of biologic is another consideration.”
If a patient had responded to the biologic for a long time and then lost response – known as secondary failure – Dr. Armstrong considers dose escalation or a switch to another agent within the same class “if it helps to address comorbidities such as PsA,” she said. “You can also try across-class switching.”
Dr. Armstrong disclosed ties with AbbVie, Arcutis, ASLAN, Beiersdorf, Boehringer Ingelheim, Bristol Myers Squibb, Dermira, Dermavant, EPI, Galderma, InCyte, Janssen, Leo, Lilly, Meiji, Modmed, Nimbus, Novartis, Ortho Dermatologics, Parexel, Pfizer, Regeneron, Sanofi, Suna, UCB, and Ventyx.
AT CALDERM 2023







