User login
Scientific doubt tempers COVID-19 vaccine optimism
US government and industry projections that a COVID-19 vaccine will be ready by this fall or even January would take compressing what usually takes at least a decade into months, with little room for error or safety surprises.
“If all the cards fall into the right place and all the stars are aligned, you definitely could get a vaccine by December or January,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said last week.
But Fauci said a more realistic timeline is still 12 to 18 months, and experts interviewed by Medscape Medical News agree. They say that although recent developments are encouraging, history and scientific reason say the day when a COVID-19 vaccine is widely available will not come this year and may not come by the end of 2021.
The encouraging signals come primarily from two recent announcements: the $1.2 billion United States backing last week of one vaccine platform and the announcement on May 18 that the first human trials of another have produced some positive phase 1 results.
Recent developments
On May 21, the US Department of Health and Human Services (HHS) under “Operation Warp Speed” announced that the US will give AstraZeneca $1.2 billion “to make available at least 300 million doses of a coronavirus vaccine called AZD1222, with the first doses delivered as early as October 2020.”
On May 18, the Massachusetts-based biotechnology company Moderna announced that phase 1 clinical results showed that its vaccine candidate, which uses a new messenger RNA (mRNA) technology, appeared safe. Eight participants in the human trials were able to produce neutralizing antibodies that researchers believe are important in developing protection from the virus.
Moderna Chief Medical Officer Tal Zaks, MD, PhD told CNN that if the vaccine candidate does well in phase 2, “it could be ready by January 2021.”
The two candidates are among 10 in clinical trials for the SARS-CoV-2 virus, according to the World Health Organization (WHO). The AstraZeneca/ AZD1222 candidate (also called ChAdOx1 nCoV-19, in collaboration with the University of Oxford) has entered phase 2/3.
Moderna’s candidate and another being developed in Beijing, China, are in phase 2, WHO reports. As of yesterday, 115 other candidates are in preclinical evaluation.
Maria Elena Bottazzi, PhD, associate dean of the National School of Tropical Medicine at Baylor College of Medicine in Houston, Texas, told Medscape Medical News it’s important to realize that, in the case of the $1.2 billion US investment, “what they’re talking about is manufacturing.”
The idea, she said, is to pay AstraZeneca up front so that manufacturing can start before it is known whether the vaccine candidate is safe or effective, the reverse of how the clinical trial process usually works.
That way, if the candidate is deemed safe and effective, time is not lost by then deciding how to make it and distribute it.
By the end of this year, she said, “Maybe we will have many vaccines made and stored in a refrigerator somewhere. But between now and December, there’s absolutely no way you can show efficacy of the vaccine at the same time you confirm that it’s safe.”
“Take these things with a grain of salt”
Animal testing for the AstraZeneca candidate, made in partnership with the University of Oxford in the United Kingdom, has yielded lackluster results, according to results on the preprint server BioRxiv, which have not been peer-reviewed.
“The results were not bad, but they were not gangbusters,” Bottazzi said. The results show the vaccine offered only partial protection.
“Partial protection is better than no protection,” she noted. “You have to take these things with a grain of salt. We don’t know what’s going to happen in humans.”
As for the Moderna candidate, Bottazzi said, “the good news is they found an appropriate safety profile. But from an eight-person group to make the extrapolation that they have efficacy — it’s unrealistic.”
Nicole Lurie, MD, MSPH, is senior adviser to the CEO for the Coalition for Epidemic Preparedness Innovation (CEPI), a nongovernmental organization funded by the Wellcome Trust, the Bill and Melinda Gates Foundation, the European Commission, and eight countries (Australia, Belgium, Canada, Ethiopia, Germany, Japan, Norway, and the United Kingdom) charged with supporting development of vaccines for pathogens on WHO’s priority list.
She and her colleagues write in a paper published online in the New England Journal of Medicine on March 30 that “it typically takes multiple candidates and many years to produce a licensed vaccine.”
The fastest time for developing a vaccine to date is 4 years, for the mumps vaccine, licensed in 1967.
As to whether she would expect a rollout of any vaccine by the end of the year, Lurie told Medscape Medical News, “If everything goes according to plan in every way, shape or form, well then maybe you can get there. But I wouldn’t hold my breath.”
Lurie and her colleagues write that “it’s far from certain that these new platforms will be scalable or that existing capacity can provide sufficient quantities of vaccine fast enough.”
On a call with reporters today, leaders of some of the words largest pharmaceutical companies said that one of the key bottlenecks is the sheer number of vials needed in order to distribute billions of doses of a successful vaccine.
Pfizer CEO Albert Bourla, DVM, PhD, said, “Typically we are producing vaccines in single-dose vials. We are exploring with governments right now if it would be more convenient if there were 5-dose vials or 10-dose vials. I think we can resolve a significant part of the bottleneck.”
Despite the challenges, experts interviewed for this article agree that it will be possible to make a vaccine for COVID-19. They don’t expect attempts to meet the same complications that HIV researchers have seen over decades as the virus continues to confound with mutations.
Fred Ledley, MD, director of the Center for Integration of Science and Industry at Bentley University in Waltham, Massachusetts, told Medscape Medical News, “There doesn’t appear to be anything terribly diabolical about this virus. The mutation rate doesn’t appear to be anything like HIV. It appears to have some big, ugly proteins on the surface, which is good for vaccines — proteins with a lot of physical features look distinguishable from healthy cells. Signs all point to that it should be possible to make a vaccine.”
History raises safety concerns
However, Ledley said, “The idea of doing it in 6 months is largely unrealistic.”
He says 18 months is more realistic, primarily because of the sheer number of people that would have to be enrolled in a phase 3 study to truly test whether the endpoints are being met.
Vaccines are given to healthy volunteers. If safety signals arise, they may not be apparent until massive numbers of people are tested in phase 3.
“You’re never going to see the rates cut to 0%, but to see the difference between 10 people getting sick and seven people getting sick, takes very, very large numbers,” Ledley said. “There’s no way that can be done in 6 months. You’re talking about tens of thousands of people enrolled.”
He notes at this point it’s unclear what the endpoints will be and what the safety thresholds will be after consideration of risks and benefit.
Another big question for Ledley: “We don’t know what type of immunity we need to protect us against the virus. Do you just need the antibodies in your blood or do you need cells that are primed to attack the virus? Is it more of a chemical clearance or do the cells need to physically go in and digest the virus?”
History also points to the need for rigorous safety precautions that scientists fear could be compromised as trial phases overlap and processes are run in parallel instead of one step at a time.
An early batch of the Salk vaccine for polio in 1955, for example, turned out to be contaminated and caused paralysis in some children and 10 deaths, he points out.
CEPI’s Lurie adds that early candidates for another coronavirus, severe acute respiratory syndrome (SARS), “caused a reaction in the lungs that was very dangerous” before development was halted.
She also pointed to previous findings that a vaccine for dengue fever could worsen the disease in some people through a phenomenon called antibody-dependent enhancement.
Lurie and colleagues write in their paper that “it’s critical that vaccines also be developed using the tried-and-true methods, even if they may take longer to enter clinical trials or to result in large numbers of doses.”
Live attenuated vaccine
Raul Andino, PhD, a virologist at the University of California San Francisco, is among the scientists working with a tried-and-true method — a live attenuated vaccine — and he told Medscape Medical News he’s predicting it will take 2 years to develop.
He said it is cheaper to produce because scientists just have to learn how to grow the virus. Because the technology is already proven, a live attenuated vaccine could be rapidly produced on a worldwide scale.
The hope is also that a live attenuated vaccine would be given once in a lifetime and therefore be more affordable, especially in poorer countries.
“While a Moderna vaccine might be good for Europe and the United States,” he said, “It’s not going to be good for Africa, India, Brazil.”
Andino said, “I would bet money” that the front-runner vaccines so far will not be one-time vaccines.
He points out that most of the vaccine candidates are trying to protect people from disease. While there’s nothing wrong with that, he said, “In my opinion that is the lower-hanging fruit.”
“In my mind we need something that interrupts the chain of transmission and induces protection,” Andino said, important for developing herd immunity.
The reason this type of approach takes longer is because you are introducing a weakened form of the virus to the body and you have to make sure it doesn’t cause disease, not just in a small test population, but in populations who may be more susceptible to the disease, Andino said.
A call for unified strategies
Universities, countries, international consortiums, and public-private partnerships are all racing to find several safe and effective vaccines as no one entity will likely be able to provide the global solution.
Some of the efforts involve overlap of entities but with different focuses.
Along with “Operation Warp Speed” and CEPI, other collaborations include Gavi the Vaccine Alliance, whose core partners include WHO, UNICEF, the World Bank, and the Gates Foundation; and “Accelerating Therapeutic Interventions and Vaccines (ACTIV) partnership,” led by the National Institutes of Health.
Industry partners in ACTIV (18 biopharmaceutical companies), according to a May 18 article published online in the Journal of the American Medical Association, have said they will contribute their respective clinical trial capacities, regardless of which agent is studied.
Some, however, have called for more streamlining of efforts.
“Ideally we’d be working together,” Lurie told Medscape Medical News.
“I’m hopeful we will find ways to collaborate scientifically,” she said. “The US government’s responsibility is to make doses for the US. CEPI’s responsibility is to make doses for the world. A big focus of CEPI is to make sure we have manufacturing capacity outside of the US so those doses can be available to the world and they don’t get seized by wealthy countries.”
Bottazzi, Ledley, Lurie, and Andino report no relevant financial relationships.
This article first appeared on Medscape.com.
US government and industry projections that a COVID-19 vaccine will be ready by this fall or even January would take compressing what usually takes at least a decade into months, with little room for error or safety surprises.
“If all the cards fall into the right place and all the stars are aligned, you definitely could get a vaccine by December or January,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said last week.
But Fauci said a more realistic timeline is still 12 to 18 months, and experts interviewed by Medscape Medical News agree. They say that although recent developments are encouraging, history and scientific reason say the day when a COVID-19 vaccine is widely available will not come this year and may not come by the end of 2021.
The encouraging signals come primarily from two recent announcements: the $1.2 billion United States backing last week of one vaccine platform and the announcement on May 18 that the first human trials of another have produced some positive phase 1 results.
Recent developments
On May 21, the US Department of Health and Human Services (HHS) under “Operation Warp Speed” announced that the US will give AstraZeneca $1.2 billion “to make available at least 300 million doses of a coronavirus vaccine called AZD1222, with the first doses delivered as early as October 2020.”
On May 18, the Massachusetts-based biotechnology company Moderna announced that phase 1 clinical results showed that its vaccine candidate, which uses a new messenger RNA (mRNA) technology, appeared safe. Eight participants in the human trials were able to produce neutralizing antibodies that researchers believe are important in developing protection from the virus.
Moderna Chief Medical Officer Tal Zaks, MD, PhD told CNN that if the vaccine candidate does well in phase 2, “it could be ready by January 2021.”
The two candidates are among 10 in clinical trials for the SARS-CoV-2 virus, according to the World Health Organization (WHO). The AstraZeneca/ AZD1222 candidate (also called ChAdOx1 nCoV-19, in collaboration with the University of Oxford) has entered phase 2/3.
Moderna’s candidate and another being developed in Beijing, China, are in phase 2, WHO reports. As of yesterday, 115 other candidates are in preclinical evaluation.
Maria Elena Bottazzi, PhD, associate dean of the National School of Tropical Medicine at Baylor College of Medicine in Houston, Texas, told Medscape Medical News it’s important to realize that, in the case of the $1.2 billion US investment, “what they’re talking about is manufacturing.”
The idea, she said, is to pay AstraZeneca up front so that manufacturing can start before it is known whether the vaccine candidate is safe or effective, the reverse of how the clinical trial process usually works.
That way, if the candidate is deemed safe and effective, time is not lost by then deciding how to make it and distribute it.
By the end of this year, she said, “Maybe we will have many vaccines made and stored in a refrigerator somewhere. But between now and December, there’s absolutely no way you can show efficacy of the vaccine at the same time you confirm that it’s safe.”
“Take these things with a grain of salt”
Animal testing for the AstraZeneca candidate, made in partnership with the University of Oxford in the United Kingdom, has yielded lackluster results, according to results on the preprint server BioRxiv, which have not been peer-reviewed.
“The results were not bad, but they were not gangbusters,” Bottazzi said. The results show the vaccine offered only partial protection.
“Partial protection is better than no protection,” she noted. “You have to take these things with a grain of salt. We don’t know what’s going to happen in humans.”
As for the Moderna candidate, Bottazzi said, “the good news is they found an appropriate safety profile. But from an eight-person group to make the extrapolation that they have efficacy — it’s unrealistic.”
Nicole Lurie, MD, MSPH, is senior adviser to the CEO for the Coalition for Epidemic Preparedness Innovation (CEPI), a nongovernmental organization funded by the Wellcome Trust, the Bill and Melinda Gates Foundation, the European Commission, and eight countries (Australia, Belgium, Canada, Ethiopia, Germany, Japan, Norway, and the United Kingdom) charged with supporting development of vaccines for pathogens on WHO’s priority list.
She and her colleagues write in a paper published online in the New England Journal of Medicine on March 30 that “it typically takes multiple candidates and many years to produce a licensed vaccine.”
The fastest time for developing a vaccine to date is 4 years, for the mumps vaccine, licensed in 1967.
As to whether she would expect a rollout of any vaccine by the end of the year, Lurie told Medscape Medical News, “If everything goes according to plan in every way, shape or form, well then maybe you can get there. But I wouldn’t hold my breath.”
Lurie and her colleagues write that “it’s far from certain that these new platforms will be scalable or that existing capacity can provide sufficient quantities of vaccine fast enough.”
On a call with reporters today, leaders of some of the words largest pharmaceutical companies said that one of the key bottlenecks is the sheer number of vials needed in order to distribute billions of doses of a successful vaccine.
Pfizer CEO Albert Bourla, DVM, PhD, said, “Typically we are producing vaccines in single-dose vials. We are exploring with governments right now if it would be more convenient if there were 5-dose vials or 10-dose vials. I think we can resolve a significant part of the bottleneck.”
Despite the challenges, experts interviewed for this article agree that it will be possible to make a vaccine for COVID-19. They don’t expect attempts to meet the same complications that HIV researchers have seen over decades as the virus continues to confound with mutations.
Fred Ledley, MD, director of the Center for Integration of Science and Industry at Bentley University in Waltham, Massachusetts, told Medscape Medical News, “There doesn’t appear to be anything terribly diabolical about this virus. The mutation rate doesn’t appear to be anything like HIV. It appears to have some big, ugly proteins on the surface, which is good for vaccines — proteins with a lot of physical features look distinguishable from healthy cells. Signs all point to that it should be possible to make a vaccine.”
History raises safety concerns
However, Ledley said, “The idea of doing it in 6 months is largely unrealistic.”
He says 18 months is more realistic, primarily because of the sheer number of people that would have to be enrolled in a phase 3 study to truly test whether the endpoints are being met.
Vaccines are given to healthy volunteers. If safety signals arise, they may not be apparent until massive numbers of people are tested in phase 3.
“You’re never going to see the rates cut to 0%, but to see the difference between 10 people getting sick and seven people getting sick, takes very, very large numbers,” Ledley said. “There’s no way that can be done in 6 months. You’re talking about tens of thousands of people enrolled.”
He notes at this point it’s unclear what the endpoints will be and what the safety thresholds will be after consideration of risks and benefit.
Another big question for Ledley: “We don’t know what type of immunity we need to protect us against the virus. Do you just need the antibodies in your blood or do you need cells that are primed to attack the virus? Is it more of a chemical clearance or do the cells need to physically go in and digest the virus?”
History also points to the need for rigorous safety precautions that scientists fear could be compromised as trial phases overlap and processes are run in parallel instead of one step at a time.
An early batch of the Salk vaccine for polio in 1955, for example, turned out to be contaminated and caused paralysis in some children and 10 deaths, he points out.
CEPI’s Lurie adds that early candidates for another coronavirus, severe acute respiratory syndrome (SARS), “caused a reaction in the lungs that was very dangerous” before development was halted.
She also pointed to previous findings that a vaccine for dengue fever could worsen the disease in some people through a phenomenon called antibody-dependent enhancement.
Lurie and colleagues write in their paper that “it’s critical that vaccines also be developed using the tried-and-true methods, even if they may take longer to enter clinical trials or to result in large numbers of doses.”
Live attenuated vaccine
Raul Andino, PhD, a virologist at the University of California San Francisco, is among the scientists working with a tried-and-true method — a live attenuated vaccine — and he told Medscape Medical News he’s predicting it will take 2 years to develop.
He said it is cheaper to produce because scientists just have to learn how to grow the virus. Because the technology is already proven, a live attenuated vaccine could be rapidly produced on a worldwide scale.
The hope is also that a live attenuated vaccine would be given once in a lifetime and therefore be more affordable, especially in poorer countries.
“While a Moderna vaccine might be good for Europe and the United States,” he said, “It’s not going to be good for Africa, India, Brazil.”
Andino said, “I would bet money” that the front-runner vaccines so far will not be one-time vaccines.
He points out that most of the vaccine candidates are trying to protect people from disease. While there’s nothing wrong with that, he said, “In my opinion that is the lower-hanging fruit.”
“In my mind we need something that interrupts the chain of transmission and induces protection,” Andino said, important for developing herd immunity.
The reason this type of approach takes longer is because you are introducing a weakened form of the virus to the body and you have to make sure it doesn’t cause disease, not just in a small test population, but in populations who may be more susceptible to the disease, Andino said.
A call for unified strategies
Universities, countries, international consortiums, and public-private partnerships are all racing to find several safe and effective vaccines as no one entity will likely be able to provide the global solution.
Some of the efforts involve overlap of entities but with different focuses.
Along with “Operation Warp Speed” and CEPI, other collaborations include Gavi the Vaccine Alliance, whose core partners include WHO, UNICEF, the World Bank, and the Gates Foundation; and “Accelerating Therapeutic Interventions and Vaccines (ACTIV) partnership,” led by the National Institutes of Health.
Industry partners in ACTIV (18 biopharmaceutical companies), according to a May 18 article published online in the Journal of the American Medical Association, have said they will contribute their respective clinical trial capacities, regardless of which agent is studied.
Some, however, have called for more streamlining of efforts.
“Ideally we’d be working together,” Lurie told Medscape Medical News.
“I’m hopeful we will find ways to collaborate scientifically,” she said. “The US government’s responsibility is to make doses for the US. CEPI’s responsibility is to make doses for the world. A big focus of CEPI is to make sure we have manufacturing capacity outside of the US so those doses can be available to the world and they don’t get seized by wealthy countries.”
Bottazzi, Ledley, Lurie, and Andino report no relevant financial relationships.
This article first appeared on Medscape.com.
US government and industry projections that a COVID-19 vaccine will be ready by this fall or even January would take compressing what usually takes at least a decade into months, with little room for error or safety surprises.
“If all the cards fall into the right place and all the stars are aligned, you definitely could get a vaccine by December or January,” Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said last week.
But Fauci said a more realistic timeline is still 12 to 18 months, and experts interviewed by Medscape Medical News agree. They say that although recent developments are encouraging, history and scientific reason say the day when a COVID-19 vaccine is widely available will not come this year and may not come by the end of 2021.
The encouraging signals come primarily from two recent announcements: the $1.2 billion United States backing last week of one vaccine platform and the announcement on May 18 that the first human trials of another have produced some positive phase 1 results.
Recent developments
On May 21, the US Department of Health and Human Services (HHS) under “Operation Warp Speed” announced that the US will give AstraZeneca $1.2 billion “to make available at least 300 million doses of a coronavirus vaccine called AZD1222, with the first doses delivered as early as October 2020.”
On May 18, the Massachusetts-based biotechnology company Moderna announced that phase 1 clinical results showed that its vaccine candidate, which uses a new messenger RNA (mRNA) technology, appeared safe. Eight participants in the human trials were able to produce neutralizing antibodies that researchers believe are important in developing protection from the virus.
Moderna Chief Medical Officer Tal Zaks, MD, PhD told CNN that if the vaccine candidate does well in phase 2, “it could be ready by January 2021.”
The two candidates are among 10 in clinical trials for the SARS-CoV-2 virus, according to the World Health Organization (WHO). The AstraZeneca/ AZD1222 candidate (also called ChAdOx1 nCoV-19, in collaboration with the University of Oxford) has entered phase 2/3.
Moderna’s candidate and another being developed in Beijing, China, are in phase 2, WHO reports. As of yesterday, 115 other candidates are in preclinical evaluation.
Maria Elena Bottazzi, PhD, associate dean of the National School of Tropical Medicine at Baylor College of Medicine in Houston, Texas, told Medscape Medical News it’s important to realize that, in the case of the $1.2 billion US investment, “what they’re talking about is manufacturing.”
The idea, she said, is to pay AstraZeneca up front so that manufacturing can start before it is known whether the vaccine candidate is safe or effective, the reverse of how the clinical trial process usually works.
That way, if the candidate is deemed safe and effective, time is not lost by then deciding how to make it and distribute it.
By the end of this year, she said, “Maybe we will have many vaccines made and stored in a refrigerator somewhere. But between now and December, there’s absolutely no way you can show efficacy of the vaccine at the same time you confirm that it’s safe.”
“Take these things with a grain of salt”
Animal testing for the AstraZeneca candidate, made in partnership with the University of Oxford in the United Kingdom, has yielded lackluster results, according to results on the preprint server BioRxiv, which have not been peer-reviewed.
“The results were not bad, but they were not gangbusters,” Bottazzi said. The results show the vaccine offered only partial protection.
“Partial protection is better than no protection,” she noted. “You have to take these things with a grain of salt. We don’t know what’s going to happen in humans.”
As for the Moderna candidate, Bottazzi said, “the good news is they found an appropriate safety profile. But from an eight-person group to make the extrapolation that they have efficacy — it’s unrealistic.”
Nicole Lurie, MD, MSPH, is senior adviser to the CEO for the Coalition for Epidemic Preparedness Innovation (CEPI), a nongovernmental organization funded by the Wellcome Trust, the Bill and Melinda Gates Foundation, the European Commission, and eight countries (Australia, Belgium, Canada, Ethiopia, Germany, Japan, Norway, and the United Kingdom) charged with supporting development of vaccines for pathogens on WHO’s priority list.
She and her colleagues write in a paper published online in the New England Journal of Medicine on March 30 that “it typically takes multiple candidates and many years to produce a licensed vaccine.”
The fastest time for developing a vaccine to date is 4 years, for the mumps vaccine, licensed in 1967.
As to whether she would expect a rollout of any vaccine by the end of the year, Lurie told Medscape Medical News, “If everything goes according to plan in every way, shape or form, well then maybe you can get there. But I wouldn’t hold my breath.”
Lurie and her colleagues write that “it’s far from certain that these new platforms will be scalable or that existing capacity can provide sufficient quantities of vaccine fast enough.”
On a call with reporters today, leaders of some of the words largest pharmaceutical companies said that one of the key bottlenecks is the sheer number of vials needed in order to distribute billions of doses of a successful vaccine.
Pfizer CEO Albert Bourla, DVM, PhD, said, “Typically we are producing vaccines in single-dose vials. We are exploring with governments right now if it would be more convenient if there were 5-dose vials or 10-dose vials. I think we can resolve a significant part of the bottleneck.”
Despite the challenges, experts interviewed for this article agree that it will be possible to make a vaccine for COVID-19. They don’t expect attempts to meet the same complications that HIV researchers have seen over decades as the virus continues to confound with mutations.
Fred Ledley, MD, director of the Center for Integration of Science and Industry at Bentley University in Waltham, Massachusetts, told Medscape Medical News, “There doesn’t appear to be anything terribly diabolical about this virus. The mutation rate doesn’t appear to be anything like HIV. It appears to have some big, ugly proteins on the surface, which is good for vaccines — proteins with a lot of physical features look distinguishable from healthy cells. Signs all point to that it should be possible to make a vaccine.”
History raises safety concerns
However, Ledley said, “The idea of doing it in 6 months is largely unrealistic.”
He says 18 months is more realistic, primarily because of the sheer number of people that would have to be enrolled in a phase 3 study to truly test whether the endpoints are being met.
Vaccines are given to healthy volunteers. If safety signals arise, they may not be apparent until massive numbers of people are tested in phase 3.
“You’re never going to see the rates cut to 0%, but to see the difference between 10 people getting sick and seven people getting sick, takes very, very large numbers,” Ledley said. “There’s no way that can be done in 6 months. You’re talking about tens of thousands of people enrolled.”
He notes at this point it’s unclear what the endpoints will be and what the safety thresholds will be after consideration of risks and benefit.
Another big question for Ledley: “We don’t know what type of immunity we need to protect us against the virus. Do you just need the antibodies in your blood or do you need cells that are primed to attack the virus? Is it more of a chemical clearance or do the cells need to physically go in and digest the virus?”
History also points to the need for rigorous safety precautions that scientists fear could be compromised as trial phases overlap and processes are run in parallel instead of one step at a time.
An early batch of the Salk vaccine for polio in 1955, for example, turned out to be contaminated and caused paralysis in some children and 10 deaths, he points out.
CEPI’s Lurie adds that early candidates for another coronavirus, severe acute respiratory syndrome (SARS), “caused a reaction in the lungs that was very dangerous” before development was halted.
She also pointed to previous findings that a vaccine for dengue fever could worsen the disease in some people through a phenomenon called antibody-dependent enhancement.
Lurie and colleagues write in their paper that “it’s critical that vaccines also be developed using the tried-and-true methods, even if they may take longer to enter clinical trials or to result in large numbers of doses.”
Live attenuated vaccine
Raul Andino, PhD, a virologist at the University of California San Francisco, is among the scientists working with a tried-and-true method — a live attenuated vaccine — and he told Medscape Medical News he’s predicting it will take 2 years to develop.
He said it is cheaper to produce because scientists just have to learn how to grow the virus. Because the technology is already proven, a live attenuated vaccine could be rapidly produced on a worldwide scale.
The hope is also that a live attenuated vaccine would be given once in a lifetime and therefore be more affordable, especially in poorer countries.
“While a Moderna vaccine might be good for Europe and the United States,” he said, “It’s not going to be good for Africa, India, Brazil.”
Andino said, “I would bet money” that the front-runner vaccines so far will not be one-time vaccines.
He points out that most of the vaccine candidates are trying to protect people from disease. While there’s nothing wrong with that, he said, “In my opinion that is the lower-hanging fruit.”
“In my mind we need something that interrupts the chain of transmission and induces protection,” Andino said, important for developing herd immunity.
The reason this type of approach takes longer is because you are introducing a weakened form of the virus to the body and you have to make sure it doesn’t cause disease, not just in a small test population, but in populations who may be more susceptible to the disease, Andino said.
A call for unified strategies
Universities, countries, international consortiums, and public-private partnerships are all racing to find several safe and effective vaccines as no one entity will likely be able to provide the global solution.
Some of the efforts involve overlap of entities but with different focuses.
Along with “Operation Warp Speed” and CEPI, other collaborations include Gavi the Vaccine Alliance, whose core partners include WHO, UNICEF, the World Bank, and the Gates Foundation; and “Accelerating Therapeutic Interventions and Vaccines (ACTIV) partnership,” led by the National Institutes of Health.
Industry partners in ACTIV (18 biopharmaceutical companies), according to a May 18 article published online in the Journal of the American Medical Association, have said they will contribute their respective clinical trial capacities, regardless of which agent is studied.
Some, however, have called for more streamlining of efforts.
“Ideally we’d be working together,” Lurie told Medscape Medical News.
“I’m hopeful we will find ways to collaborate scientifically,” she said. “The US government’s responsibility is to make doses for the US. CEPI’s responsibility is to make doses for the world. A big focus of CEPI is to make sure we have manufacturing capacity outside of the US so those doses can be available to the world and they don’t get seized by wealthy countries.”
Bottazzi, Ledley, Lurie, and Andino report no relevant financial relationships.
This article first appeared on Medscape.com.
New York City inpatient detox unit keeps running: Here’s how
Substance use disorder and its daily consequences take no breaks even during a pandemic. The stressors created by COVID-19, including deaths of loved ones and the disruptions to normal life from policies aimed at flattening the curve, seem to have increased substance use.
I practice as a hospitalist with an internal medicine background and specialty in addiction medicine at BronxCare Health System’s inpatient detoxification unit, a 24/7, 20-bed medically-supervised unit in South Bronx in New York City. It is one of the comprehensive services provided by the BronxCare’s life recovery center and addiction services, which also includes an outpatient clinic, opioid treatment program, inpatient rehab, and a half-way house. Inpatient detoxification units like ours are designed to treat serious addictions and chemical dependency and prevent and treat life-threatening withdrawal symptoms and signs or complications. Our patients come from all over the city and its adjoining suburbs, including from emergency room referrals, referral clinics, courts and the justice system, walk-ins, and self-referrals.
At a time when many inpatient detoxification units within the city were temporarily closed due to fear of inpatient spread of the virus or to provide extra COVID beds in anticipation for the peak surge, we have been able to provide a needed service. In fact, several other inpatient detoxification programs within the city have been able to refer their patients to our facility.
Individuals with substance use disorder have historically been a vulnerable and underserved population and possess high risk for multiple health problems as well as preexisting conditions. Many have limited life options financially, educationally, and with housing, and encounter barriers to accessing primary health care services, including preventive services. The introduction of the COVID-19 pandemic into these patients’ precarious health situations only made things worse as many of the limited resources for patients with substance use disorder were diverted to battling the pandemic. Numerous inpatient and outpatient addiction services, for example, were temporarily shut down. This has led to an increase in domestic violence, and psychiatric decompensation, including psychosis, suicidal attempts, and worsening of medical comorbidities in these patients.
Our wake-up call came when the first case of COVID-19 was confirmed in New York in early March. Within a short period of time the state became the epicenter for COVID-19. With the projection of millions of cases being positive and the number of new cases doubling every third day at the onset in New York City, we knew we had a battle brewing and needed to radically transform our mode of operation fast.
Our first task was to ensure the safety of our patients and the dedicated health workers attending to them. We streamlined the patient point of entry through one screening site, while also brushing up on our history-taking to intently screen for COVID-19. This included not just focusing on travels from China, but from Europe and other parts of the world.
Yes, we did ask patients about cough, fever, shortness of breath or difficulty breathing, feeling fatigued, severe body ache, and possible contact with someone who is sick or has traveled overseas. But we were also attuned to the increased rate of community spread and the presentation of other symptoms, such as loss of taste and smell, early in the process. Hence we were able to triage patients with suspected cases to the appropriate sections of the hospital for further screening, testing, and evaluation, instead of having those patients admitted to the detox unit.
Early in the process a huddle team was instituted with daily briefing of staff lasting 30 minutes or less. This team consists of physicians, nurses, a physician assistant, a social worker, and a counselor. In addition to discussing treatment plans for the patient, they deliberate on the public health information from the hospital’s COVID-19 command center, New York State Department of Health, the Office of Mental Health, and the Centers for Disease Control and Prevention concerning the latest evidence-based information. These discussions have helped us modify our policies and practices.
We instituted a no visiting rule during a short hospital stay of 5-7 days, and this was initiated weeks in advance of many institutions, including nursing homes with vulnerable populations. Our admitting criteria was reviewed to allow for admission of only those patients who absolutely needed inpatient substance use disorder treatment, including patients with severe withdrawal symptoms and signs, comorbidities, or neuropsychiatric manifestations that made them unsafe for outpatient or home detoxification. Others were triaged to the outpatient services which was amply supported with telemedicine. Rooms and designated areas of the building were earmarked as places for isolation/quarantine if suspected COVID-19 cases were identified pending testing. To assess patients’ risk of COVID-19, we do point-of-care nasopharyngeal swab testing with polymerase chain reaction.
Regarding face masks, patients and staff were fitted with ones early in the process. Additionally, staff were trained on the importance of face mask use and how to ensure you have a tight seal around the mouth and nose and were provided with other appropriate personal protective equipment. Concerning social distancing, we reduced the patient population capacity for the unit down to 50% and offered only single room admissions. Social distancing was encouraged in the unit, including in the television and recreation room and dining room, and during small treatment groups of less than six individuals. Daily temperature checks with noncontact handheld thermometers were enforced for staff and anyone coming into the life recovery center.
Patients are continuously being educated on the presentations of COVID-19 and encouraged to report any symptoms. Any staff feeling sick or having symptoms are encouraged to stay home. Rigorous and continuous cleaning of surfaces, especially of areas subjected to common use, is done frequently by the hospital housekeeping and environmental crew and is the order of the day.
Dr. Fagbemi is a hospitalist at BronxCare Health System, a not-for-profit health and teaching hospital system serving South and Central Bronx in New York. He has no conflicts of interest to disclose.
Substance use disorder and its daily consequences take no breaks even during a pandemic. The stressors created by COVID-19, including deaths of loved ones and the disruptions to normal life from policies aimed at flattening the curve, seem to have increased substance use.
I practice as a hospitalist with an internal medicine background and specialty in addiction medicine at BronxCare Health System’s inpatient detoxification unit, a 24/7, 20-bed medically-supervised unit in South Bronx in New York City. It is one of the comprehensive services provided by the BronxCare’s life recovery center and addiction services, which also includes an outpatient clinic, opioid treatment program, inpatient rehab, and a half-way house. Inpatient detoxification units like ours are designed to treat serious addictions and chemical dependency and prevent and treat life-threatening withdrawal symptoms and signs or complications. Our patients come from all over the city and its adjoining suburbs, including from emergency room referrals, referral clinics, courts and the justice system, walk-ins, and self-referrals.
At a time when many inpatient detoxification units within the city were temporarily closed due to fear of inpatient spread of the virus or to provide extra COVID beds in anticipation for the peak surge, we have been able to provide a needed service. In fact, several other inpatient detoxification programs within the city have been able to refer their patients to our facility.
Individuals with substance use disorder have historically been a vulnerable and underserved population and possess high risk for multiple health problems as well as preexisting conditions. Many have limited life options financially, educationally, and with housing, and encounter barriers to accessing primary health care services, including preventive services. The introduction of the COVID-19 pandemic into these patients’ precarious health situations only made things worse as many of the limited resources for patients with substance use disorder were diverted to battling the pandemic. Numerous inpatient and outpatient addiction services, for example, were temporarily shut down. This has led to an increase in domestic violence, and psychiatric decompensation, including psychosis, suicidal attempts, and worsening of medical comorbidities in these patients.
Our wake-up call came when the first case of COVID-19 was confirmed in New York in early March. Within a short period of time the state became the epicenter for COVID-19. With the projection of millions of cases being positive and the number of new cases doubling every third day at the onset in New York City, we knew we had a battle brewing and needed to radically transform our mode of operation fast.
Our first task was to ensure the safety of our patients and the dedicated health workers attending to them. We streamlined the patient point of entry through one screening site, while also brushing up on our history-taking to intently screen for COVID-19. This included not just focusing on travels from China, but from Europe and other parts of the world.
Yes, we did ask patients about cough, fever, shortness of breath or difficulty breathing, feeling fatigued, severe body ache, and possible contact with someone who is sick or has traveled overseas. But we were also attuned to the increased rate of community spread and the presentation of other symptoms, such as loss of taste and smell, early in the process. Hence we were able to triage patients with suspected cases to the appropriate sections of the hospital for further screening, testing, and evaluation, instead of having those patients admitted to the detox unit.
Early in the process a huddle team was instituted with daily briefing of staff lasting 30 minutes or less. This team consists of physicians, nurses, a physician assistant, a social worker, and a counselor. In addition to discussing treatment plans for the patient, they deliberate on the public health information from the hospital’s COVID-19 command center, New York State Department of Health, the Office of Mental Health, and the Centers for Disease Control and Prevention concerning the latest evidence-based information. These discussions have helped us modify our policies and practices.
We instituted a no visiting rule during a short hospital stay of 5-7 days, and this was initiated weeks in advance of many institutions, including nursing homes with vulnerable populations. Our admitting criteria was reviewed to allow for admission of only those patients who absolutely needed inpatient substance use disorder treatment, including patients with severe withdrawal symptoms and signs, comorbidities, or neuropsychiatric manifestations that made them unsafe for outpatient or home detoxification. Others were triaged to the outpatient services which was amply supported with telemedicine. Rooms and designated areas of the building were earmarked as places for isolation/quarantine if suspected COVID-19 cases were identified pending testing. To assess patients’ risk of COVID-19, we do point-of-care nasopharyngeal swab testing with polymerase chain reaction.
Regarding face masks, patients and staff were fitted with ones early in the process. Additionally, staff were trained on the importance of face mask use and how to ensure you have a tight seal around the mouth and nose and were provided with other appropriate personal protective equipment. Concerning social distancing, we reduced the patient population capacity for the unit down to 50% and offered only single room admissions. Social distancing was encouraged in the unit, including in the television and recreation room and dining room, and during small treatment groups of less than six individuals. Daily temperature checks with noncontact handheld thermometers were enforced for staff and anyone coming into the life recovery center.
Patients are continuously being educated on the presentations of COVID-19 and encouraged to report any symptoms. Any staff feeling sick or having symptoms are encouraged to stay home. Rigorous and continuous cleaning of surfaces, especially of areas subjected to common use, is done frequently by the hospital housekeeping and environmental crew and is the order of the day.
Dr. Fagbemi is a hospitalist at BronxCare Health System, a not-for-profit health and teaching hospital system serving South and Central Bronx in New York. He has no conflicts of interest to disclose.
Substance use disorder and its daily consequences take no breaks even during a pandemic. The stressors created by COVID-19, including deaths of loved ones and the disruptions to normal life from policies aimed at flattening the curve, seem to have increased substance use.
I practice as a hospitalist with an internal medicine background and specialty in addiction medicine at BronxCare Health System’s inpatient detoxification unit, a 24/7, 20-bed medically-supervised unit in South Bronx in New York City. It is one of the comprehensive services provided by the BronxCare’s life recovery center and addiction services, which also includes an outpatient clinic, opioid treatment program, inpatient rehab, and a half-way house. Inpatient detoxification units like ours are designed to treat serious addictions and chemical dependency and prevent and treat life-threatening withdrawal symptoms and signs or complications. Our patients come from all over the city and its adjoining suburbs, including from emergency room referrals, referral clinics, courts and the justice system, walk-ins, and self-referrals.
At a time when many inpatient detoxification units within the city were temporarily closed due to fear of inpatient spread of the virus or to provide extra COVID beds in anticipation for the peak surge, we have been able to provide a needed service. In fact, several other inpatient detoxification programs within the city have been able to refer their patients to our facility.
Individuals with substance use disorder have historically been a vulnerable and underserved population and possess high risk for multiple health problems as well as preexisting conditions. Many have limited life options financially, educationally, and with housing, and encounter barriers to accessing primary health care services, including preventive services. The introduction of the COVID-19 pandemic into these patients’ precarious health situations only made things worse as many of the limited resources for patients with substance use disorder were diverted to battling the pandemic. Numerous inpatient and outpatient addiction services, for example, were temporarily shut down. This has led to an increase in domestic violence, and psychiatric decompensation, including psychosis, suicidal attempts, and worsening of medical comorbidities in these patients.
Our wake-up call came when the first case of COVID-19 was confirmed in New York in early March. Within a short period of time the state became the epicenter for COVID-19. With the projection of millions of cases being positive and the number of new cases doubling every third day at the onset in New York City, we knew we had a battle brewing and needed to radically transform our mode of operation fast.
Our first task was to ensure the safety of our patients and the dedicated health workers attending to them. We streamlined the patient point of entry through one screening site, while also brushing up on our history-taking to intently screen for COVID-19. This included not just focusing on travels from China, but from Europe and other parts of the world.
Yes, we did ask patients about cough, fever, shortness of breath or difficulty breathing, feeling fatigued, severe body ache, and possible contact with someone who is sick or has traveled overseas. But we were also attuned to the increased rate of community spread and the presentation of other symptoms, such as loss of taste and smell, early in the process. Hence we were able to triage patients with suspected cases to the appropriate sections of the hospital for further screening, testing, and evaluation, instead of having those patients admitted to the detox unit.
Early in the process a huddle team was instituted with daily briefing of staff lasting 30 minutes or less. This team consists of physicians, nurses, a physician assistant, a social worker, and a counselor. In addition to discussing treatment plans for the patient, they deliberate on the public health information from the hospital’s COVID-19 command center, New York State Department of Health, the Office of Mental Health, and the Centers for Disease Control and Prevention concerning the latest evidence-based information. These discussions have helped us modify our policies and practices.
We instituted a no visiting rule during a short hospital stay of 5-7 days, and this was initiated weeks in advance of many institutions, including nursing homes with vulnerable populations. Our admitting criteria was reviewed to allow for admission of only those patients who absolutely needed inpatient substance use disorder treatment, including patients with severe withdrawal symptoms and signs, comorbidities, or neuropsychiatric manifestations that made them unsafe for outpatient or home detoxification. Others were triaged to the outpatient services which was amply supported with telemedicine. Rooms and designated areas of the building were earmarked as places for isolation/quarantine if suspected COVID-19 cases were identified pending testing. To assess patients’ risk of COVID-19, we do point-of-care nasopharyngeal swab testing with polymerase chain reaction.
Regarding face masks, patients and staff were fitted with ones early in the process. Additionally, staff were trained on the importance of face mask use and how to ensure you have a tight seal around the mouth and nose and were provided with other appropriate personal protective equipment. Concerning social distancing, we reduced the patient population capacity for the unit down to 50% and offered only single room admissions. Social distancing was encouraged in the unit, including in the television and recreation room and dining room, and during small treatment groups of less than six individuals. Daily temperature checks with noncontact handheld thermometers were enforced for staff and anyone coming into the life recovery center.
Patients are continuously being educated on the presentations of COVID-19 and encouraged to report any symptoms. Any staff feeling sick or having symptoms are encouraged to stay home. Rigorous and continuous cleaning of surfaces, especially of areas subjected to common use, is done frequently by the hospital housekeeping and environmental crew and is the order of the day.
Dr. Fagbemi is a hospitalist at BronxCare Health System, a not-for-profit health and teaching hospital system serving South and Central Bronx in New York. He has no conflicts of interest to disclose.
A long road to recovery: Lung rehab needed after COVID-19
If one word describes Eladio (“Lad”) Braganza, age 77, it’s “tenacious.” For 28 days, he clung to life on a ventilator in a Seattle ICU. Now – after a 46-day hospitalization for SARS-CoV-2 infection – he’s making progress in inpatient rehab, determined to regain function.
“We were not sure if he was going to make it through his first night in the hospital, and for a while after that. We were really prepared that he would not survive his ventilator time,” his daughter, Maria Braganza, said in an interview just 5 days after her father had been transferred to inpatient rehab.
In many ways, Mr. Braganza’s experience is typical of seriously ill COVID-19 patients. Many go from walking and talking to being on a ventilator within 10 hours or less. Mr. Braganza was admitted to the hospital on March 21 and was intubated that day. To keep him on the ventilator, he was heavily sedated and unconscious at times. In the ICU, he experienced bouts of low blood pressure, a pattern of shock that occurs in COVID-19 patients and that does not always respond to fluids.
Doctors have quickly learned to treat these patients aggressively. Many patients in the ICU with COVID-19 develop an inflamed, atypical form of acute respiratory distress syndrome (ARDS), in which the lung’s compliance, or stiffness, does not match the severity of hypoxia. These patients require high levels of oxygen and high ventilator settings. Many develop pneumothorax, or collapsed lungs, because of the high pressures needed to deliver oxygen and the prolonged time on ventilation.
“The vast majority of COVID patients in the ICU have lung disease that is quite severe, much more severe than I have seen in my 20 years of doing this,” said critical care specialist Anna Nolan, MD, of the department of medicine at New York University.
After about 2 weeks, some of these patients can come off the ventilator, or they may undergo a tracheostomy, a hole in the neck through which a tube is placed to deliver oxygen. By this time, many have developed ICU-acquired weakness and muscle wasting. Some may be so debilitated that they cannot walk. Even the respiratory muscles that help them breathe may have weakened as a result of the ventilator doing the work for them.
These patients “get sick very fast, and it takes a long time for them to heal. What’s not really well appreciated is how much rehab and how much recovery time these patients are going to need,” said David Chong, MD. He is medical director of the ICU at New York–Presbyterian Hospital/Columbia University Medical Center, and he has been on the front lines during the COVID-19 surge in New York City.
The road to recovery
Regardless of the cause, many people who have a prolonged stint in the ICU face an even longer convalescence. Still-unanswered questions concern whether recovery time will be longer for those with COVID-19, compared with other illnesses, and whether some of the damage may be permanent. A number of small studies in Hong Kong and China, as well as studies of severe acute respiratory syndrome patients’ recoveries, have promoted speculation about possible long-lasting damage to lungs and other organs from COVID-19.
Yet some of these reports have left out important details about ARDS in COVID-19 patients who also may be most at risk for long-lasting damage. To clear up some of the confusion, the Pulmonary Fibrosis Foundation said on April 6 that some but not all of COVID-19 patients who develop ARDS may go on to develop lung fibrosis – scarring of the lungs – which may be permanent.
“Post-ARDS fibrosis typically is not progressive, but nonetheless can be severe and limiting. The recovery period for post-ARDS fibrosis is approximately 1 year and the residual deficits persist, but generally do not progress,” the foundation noted.
Emerging research on lung damage in COVID-19
Because the pandemic is only a few months in, it’s unclear as yet what the long-term consequences of severe COVID-19 may be. But emerging data are enabling researchers to venture an educated guess about what may happen in the months and years ahead.
The key to understanding the data is knowing that ARDS is a syndrome – the end product of a variety of diseases or insults to the lung. Under the microscope, lung damage from ARDS associated with COVID-19 is indistinguishable from lung damage resulting from other causes, such as vaping, sepsis, or shock caused by a motor vehicle accident, said Sanjay Mukhopadhyay, MD, director of pulmonary pathology at Cleveland Clinic.
Dr. Mukhopadhyay, who specializes in lung pathology, performed one of the first complete autopsies of a COVID-19 patient in the United States. In most autopsy series published to date, he said, the most common lung finding in patients who have died from COVID-19 is diffuse alveolar damage (DAD), a pattern of lung injury seen in ARDS from many other causes.
In DAD, the walls of the alveoli – thinly lined air sacs that facilitate gas exchange in the lung – develop a pink, hyaline membrane composed of damaged cells and plasma proteins that leak from capillaries in the wall of the alveolus. This hyaline membrane gets plastered against the wall of the alveolus and interferes with diffusion of oxygen into the body.
“We know what happens in ARDS from other causes. If you follow people who have been on a ventilator long term, some of their respiratory function goes back to normal,” Dr. Mukhopadhyay said. “But there are other people in whom some degree of respiratory impairment lingers. In these patients, we think the DAD progresses to an organizing stage.”
Organizing pneumonia refers to a family of diseases in which fibroblasts (cells involved in wound healing) arrive and form scar tissue that forms hyaline membranes and fibrin balls (tough proteins) that fill up the alveoli, making gas exchange very difficult.
Also called BOOP (bronchiolitis obliterans organizing pneumonia), this condition is sensitive to steroids. Early aggressive steroid treatment can prevent long-term lung damage. Without steroids, damage can become permanent. A variant of this condition is termed acute fibrinous and organizing pneumonia (AFOP), which is also sensitive to steroids. A report from France demonstrates AFOP in some patients who have died from COVID-19.
The trick is identifying who is developing BOOP and who is not, and beyond that, who might be most amenable to treatment. Use of steroids for patients with certain other problems, such as a bacterial infection on top of COVID-19, could be harmful. David H. Chong, MD, and colleagues at Columbia University Irving Medical Center, New York, are investigating this to determine which COVID-19 patients may benefit from early steroid therapy.
“It’s not clear if there is a predominant histologic type or if we are catching people at different phases of their disease, and therefore we’re seeing different lung pathology,” Dr. Chong said.
He thinks that many patients with severe COVID-19 probably will not develop this pattern of lung scarring. “We’re speculating that lung damage from severe COVID-19 is probably going to behave more like lung damage from regular ARDS, which is often reversible. We think the vast majority of these patients probably have DAD that is similar to most patients with ARDS from other etiologies,” Dr. Chong said.
That would be consistent with information from China. In an April interview with Chinese domestic media, Zhong Nanshan, MD, a pulmonologist at the head of China’s COVID-19 task force, stated that he expects that the lungs in most patients with COVID-19 will gradually recover. He was responding to a widely publicized small study that found evidence of residual lung abnormalities at hospital discharge in most patients (94%, 66/70) who suffered from COVID-19 pneumonia in Wuhan, China, from January to February 2020.
Tough research conditions
Experts say that follow-up in this Chinese study and others to date has not been nearly long enough to allow predictions about lasting lung damage in COVID-19.
They also highlight the tough conditions in which researchers are working. Few autopsies have been performed so far – autopsies take time, extra precautions must be taken to avoid spread of COVID-19, and many patients and families do not consent to an autopsy. Furthermore, autopsy data from patients who died of COVID-19 may not extrapolate to survivors.
“I would not hang my hat on any of the limited data I have seen on autopsies,” said Lina Miyakawa, MD, a critical care and pulmonary medicine specialist at Mount Sinai Hospital in New York City.
“Even though we have answers about how the lungs are damaged at the end stage, this does not elucidate any answers about the earlier lung damage from this disease,” she continued. “It would be informative to have pathological data from the early or transitional phase, to see if that may translate into a treatment modality for COVID-19 patients.”
The problem is that these patients often experience a large amount of sloughing of airway cells, along with mucous plugging (collections of mucous that can block airflow and collapse alveoli). Bronchoscopy, which is used to view the inside of the lungs and sometimes to retrieve biopsy specimens for microscopic evaluation, is too risky for many COVID-19 patients.
In addition, few CT data exist for severely ill COVID-19 patients, who can be so unstable that to transport them to undergo a CT scan can be dangerous, not to mention the concern regarding infection control.
Even if sufficient data did exist, findings from chest x-rays, CTs, pathology studies, and lung function tests do not always match up. A patient who has lung abnormalities on CT may not necessarily have clinically impaired lung function or abnormal pathologic findings, according to Ali Gholamrezanezhad, MD, an emergency radiologist who is with the department of clinical radiology at the University of Southern California, Los Angeles.
Together with colleagues at USC, Dr. Gholamrezanezhad has started a long-term study of patients who were hospitalized with COVID-19. The researchers will follow patients for at least 1 year and will use chest x-ray, chest CT, and exercise testing to evaluate lung recovery over time.
“In the acute phase, we have acute inflammation called ground glass opacities, which usually happen bilaterally in COVID-19. That is totally reversible damage that can return to normal with no scarring,” Dr. Gholamrezanezhad said.
On the basis of data from survivors of other severe pneumonias, such as Middle East respiratory syndrome, SARS-CoV-1 infection, and H1N1 influenza, Gholamrezanezhad thinks that most survivors of COVID-19 will be able to return to work and normal life, although some may show residual lung dysfunction. Age, underlying medical conditions, smoking, length of hospital stay, severity of illness, and quality of treatment may all play a role in how well these people recover.
The lung has a remarkable capacity to recover, he added. Critical illness can destroy type one pneumocytes — the cells that line the alveoli in the lung — but over time, these cells grow back and reline the lungs. When they do, they can also help repair the lungs.
On top of that, the lung has a large functional reserve, and when one section becomes damaged, the rest of the lung can compensate.
However, for some people, total maximum exercise capacity may be affected, he commented.
Mukhopadhyay said: “My feeling is you will get reversal to normal in some patients and you will get long-term fibrosis from ARDS in some survivors. The question is, how many will have complete resolution and how many will have fibrosis? To know the answer, we will need a lot more data than we have now.”
Convalescence of COVID-19 Patients
Like many who become seriously ill with COVID-19, Braganza had underlying medical problems. Before becoming ill, he had had a heart attack and stroke. He walked with a walker and had some age-related memory problems.
Five days after transfer to inpatient rehab, Braganza was walking up and down the hallway using a walker. He was still shaking off the effects of being heavily sedated for so long, and he experienced periods of confusion. When he first came off the ventilator, he mixed up days and nights. Sometimes he did not remember being so sick. A former software engineer, Braganza usually had no problem using technology, but he has had to relearn how to use his phone and connect his iPad to Wi-Fi.
“He is still struggling quite a bit with remembering how to do basic things,” Maria Braganza said. “He has times of being really depressed because he feels like he’s not making progress.”
Doctors are taking note and starting to think about what lies ahead for ICU survivors of COVID-19. They worry about the potential for disease recurrence as well as readmission for other problems, such as other infections and hip fractures.
“As COVID-19 survivors begin to recover, there will be a large burden of chronic critical illness. We expect a significant need for rehabilitation in most ICU survivors of COVID-19,” said Steve Lubinsky, MD, medical director of respiratory care at New York University Langone Tisch Hospital.
Thinking about her father, Maria Braganza brings an extra dimension to these concerns. She thinks about depression, loneliness, and social isolation among older survivors of COVID-19. These problems existed long before the pandemic, but COVID-19 has magnified them.
The rehab staff estimates that Mr. Braganza will spend 10-14 days in their program, but discharge home creates a conundrum. Before becoming ill, Mr. Braganza lived in an independent senior living facility. Now, because of social distancing, he will no longer be able to hang out and have meals with his friends.
“Dad’s already feeling really lonely in the hospital. If we stay on a semipermanent lockdown, will he be able to see the people he loves?” Maria Braganza said. “Even though somebody is older, they have a lot to give and a lot of experience. They just need a little extra to be able to have that life.”
Dr. Nolan, Dr. Chong, Dr. Mukhopadhyay, Dr. Miyakawa, Dr. Gholamrezanezhad, and Dr. Lubinsky report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
If one word describes Eladio (“Lad”) Braganza, age 77, it’s “tenacious.” For 28 days, he clung to life on a ventilator in a Seattle ICU. Now – after a 46-day hospitalization for SARS-CoV-2 infection – he’s making progress in inpatient rehab, determined to regain function.
“We were not sure if he was going to make it through his first night in the hospital, and for a while after that. We were really prepared that he would not survive his ventilator time,” his daughter, Maria Braganza, said in an interview just 5 days after her father had been transferred to inpatient rehab.
In many ways, Mr. Braganza’s experience is typical of seriously ill COVID-19 patients. Many go from walking and talking to being on a ventilator within 10 hours or less. Mr. Braganza was admitted to the hospital on March 21 and was intubated that day. To keep him on the ventilator, he was heavily sedated and unconscious at times. In the ICU, he experienced bouts of low blood pressure, a pattern of shock that occurs in COVID-19 patients and that does not always respond to fluids.
Doctors have quickly learned to treat these patients aggressively. Many patients in the ICU with COVID-19 develop an inflamed, atypical form of acute respiratory distress syndrome (ARDS), in which the lung’s compliance, or stiffness, does not match the severity of hypoxia. These patients require high levels of oxygen and high ventilator settings. Many develop pneumothorax, or collapsed lungs, because of the high pressures needed to deliver oxygen and the prolonged time on ventilation.
“The vast majority of COVID patients in the ICU have lung disease that is quite severe, much more severe than I have seen in my 20 years of doing this,” said critical care specialist Anna Nolan, MD, of the department of medicine at New York University.
After about 2 weeks, some of these patients can come off the ventilator, or they may undergo a tracheostomy, a hole in the neck through which a tube is placed to deliver oxygen. By this time, many have developed ICU-acquired weakness and muscle wasting. Some may be so debilitated that they cannot walk. Even the respiratory muscles that help them breathe may have weakened as a result of the ventilator doing the work for them.
These patients “get sick very fast, and it takes a long time for them to heal. What’s not really well appreciated is how much rehab and how much recovery time these patients are going to need,” said David Chong, MD. He is medical director of the ICU at New York–Presbyterian Hospital/Columbia University Medical Center, and he has been on the front lines during the COVID-19 surge in New York City.
The road to recovery
Regardless of the cause, many people who have a prolonged stint in the ICU face an even longer convalescence. Still-unanswered questions concern whether recovery time will be longer for those with COVID-19, compared with other illnesses, and whether some of the damage may be permanent. A number of small studies in Hong Kong and China, as well as studies of severe acute respiratory syndrome patients’ recoveries, have promoted speculation about possible long-lasting damage to lungs and other organs from COVID-19.
Yet some of these reports have left out important details about ARDS in COVID-19 patients who also may be most at risk for long-lasting damage. To clear up some of the confusion, the Pulmonary Fibrosis Foundation said on April 6 that some but not all of COVID-19 patients who develop ARDS may go on to develop lung fibrosis – scarring of the lungs – which may be permanent.
“Post-ARDS fibrosis typically is not progressive, but nonetheless can be severe and limiting. The recovery period for post-ARDS fibrosis is approximately 1 year and the residual deficits persist, but generally do not progress,” the foundation noted.
Emerging research on lung damage in COVID-19
Because the pandemic is only a few months in, it’s unclear as yet what the long-term consequences of severe COVID-19 may be. But emerging data are enabling researchers to venture an educated guess about what may happen in the months and years ahead.
The key to understanding the data is knowing that ARDS is a syndrome – the end product of a variety of diseases or insults to the lung. Under the microscope, lung damage from ARDS associated with COVID-19 is indistinguishable from lung damage resulting from other causes, such as vaping, sepsis, or shock caused by a motor vehicle accident, said Sanjay Mukhopadhyay, MD, director of pulmonary pathology at Cleveland Clinic.
Dr. Mukhopadhyay, who specializes in lung pathology, performed one of the first complete autopsies of a COVID-19 patient in the United States. In most autopsy series published to date, he said, the most common lung finding in patients who have died from COVID-19 is diffuse alveolar damage (DAD), a pattern of lung injury seen in ARDS from many other causes.
In DAD, the walls of the alveoli – thinly lined air sacs that facilitate gas exchange in the lung – develop a pink, hyaline membrane composed of damaged cells and plasma proteins that leak from capillaries in the wall of the alveolus. This hyaline membrane gets plastered against the wall of the alveolus and interferes with diffusion of oxygen into the body.
“We know what happens in ARDS from other causes. If you follow people who have been on a ventilator long term, some of their respiratory function goes back to normal,” Dr. Mukhopadhyay said. “But there are other people in whom some degree of respiratory impairment lingers. In these patients, we think the DAD progresses to an organizing stage.”
Organizing pneumonia refers to a family of diseases in which fibroblasts (cells involved in wound healing) arrive and form scar tissue that forms hyaline membranes and fibrin balls (tough proteins) that fill up the alveoli, making gas exchange very difficult.
Also called BOOP (bronchiolitis obliterans organizing pneumonia), this condition is sensitive to steroids. Early aggressive steroid treatment can prevent long-term lung damage. Without steroids, damage can become permanent. A variant of this condition is termed acute fibrinous and organizing pneumonia (AFOP), which is also sensitive to steroids. A report from France demonstrates AFOP in some patients who have died from COVID-19.
The trick is identifying who is developing BOOP and who is not, and beyond that, who might be most amenable to treatment. Use of steroids for patients with certain other problems, such as a bacterial infection on top of COVID-19, could be harmful. David H. Chong, MD, and colleagues at Columbia University Irving Medical Center, New York, are investigating this to determine which COVID-19 patients may benefit from early steroid therapy.
“It’s not clear if there is a predominant histologic type or if we are catching people at different phases of their disease, and therefore we’re seeing different lung pathology,” Dr. Chong said.
He thinks that many patients with severe COVID-19 probably will not develop this pattern of lung scarring. “We’re speculating that lung damage from severe COVID-19 is probably going to behave more like lung damage from regular ARDS, which is often reversible. We think the vast majority of these patients probably have DAD that is similar to most patients with ARDS from other etiologies,” Dr. Chong said.
That would be consistent with information from China. In an April interview with Chinese domestic media, Zhong Nanshan, MD, a pulmonologist at the head of China’s COVID-19 task force, stated that he expects that the lungs in most patients with COVID-19 will gradually recover. He was responding to a widely publicized small study that found evidence of residual lung abnormalities at hospital discharge in most patients (94%, 66/70) who suffered from COVID-19 pneumonia in Wuhan, China, from January to February 2020.
Tough research conditions
Experts say that follow-up in this Chinese study and others to date has not been nearly long enough to allow predictions about lasting lung damage in COVID-19.
They also highlight the tough conditions in which researchers are working. Few autopsies have been performed so far – autopsies take time, extra precautions must be taken to avoid spread of COVID-19, and many patients and families do not consent to an autopsy. Furthermore, autopsy data from patients who died of COVID-19 may not extrapolate to survivors.
“I would not hang my hat on any of the limited data I have seen on autopsies,” said Lina Miyakawa, MD, a critical care and pulmonary medicine specialist at Mount Sinai Hospital in New York City.
“Even though we have answers about how the lungs are damaged at the end stage, this does not elucidate any answers about the earlier lung damage from this disease,” she continued. “It would be informative to have pathological data from the early or transitional phase, to see if that may translate into a treatment modality for COVID-19 patients.”
The problem is that these patients often experience a large amount of sloughing of airway cells, along with mucous plugging (collections of mucous that can block airflow and collapse alveoli). Bronchoscopy, which is used to view the inside of the lungs and sometimes to retrieve biopsy specimens for microscopic evaluation, is too risky for many COVID-19 patients.
In addition, few CT data exist for severely ill COVID-19 patients, who can be so unstable that to transport them to undergo a CT scan can be dangerous, not to mention the concern regarding infection control.
Even if sufficient data did exist, findings from chest x-rays, CTs, pathology studies, and lung function tests do not always match up. A patient who has lung abnormalities on CT may not necessarily have clinically impaired lung function or abnormal pathologic findings, according to Ali Gholamrezanezhad, MD, an emergency radiologist who is with the department of clinical radiology at the University of Southern California, Los Angeles.
Together with colleagues at USC, Dr. Gholamrezanezhad has started a long-term study of patients who were hospitalized with COVID-19. The researchers will follow patients for at least 1 year and will use chest x-ray, chest CT, and exercise testing to evaluate lung recovery over time.
“In the acute phase, we have acute inflammation called ground glass opacities, which usually happen bilaterally in COVID-19. That is totally reversible damage that can return to normal with no scarring,” Dr. Gholamrezanezhad said.
On the basis of data from survivors of other severe pneumonias, such as Middle East respiratory syndrome, SARS-CoV-1 infection, and H1N1 influenza, Gholamrezanezhad thinks that most survivors of COVID-19 will be able to return to work and normal life, although some may show residual lung dysfunction. Age, underlying medical conditions, smoking, length of hospital stay, severity of illness, and quality of treatment may all play a role in how well these people recover.
The lung has a remarkable capacity to recover, he added. Critical illness can destroy type one pneumocytes — the cells that line the alveoli in the lung — but over time, these cells grow back and reline the lungs. When they do, they can also help repair the lungs.
On top of that, the lung has a large functional reserve, and when one section becomes damaged, the rest of the lung can compensate.
However, for some people, total maximum exercise capacity may be affected, he commented.
Mukhopadhyay said: “My feeling is you will get reversal to normal in some patients and you will get long-term fibrosis from ARDS in some survivors. The question is, how many will have complete resolution and how many will have fibrosis? To know the answer, we will need a lot more data than we have now.”
Convalescence of COVID-19 Patients
Like many who become seriously ill with COVID-19, Braganza had underlying medical problems. Before becoming ill, he had had a heart attack and stroke. He walked with a walker and had some age-related memory problems.
Five days after transfer to inpatient rehab, Braganza was walking up and down the hallway using a walker. He was still shaking off the effects of being heavily sedated for so long, and he experienced periods of confusion. When he first came off the ventilator, he mixed up days and nights. Sometimes he did not remember being so sick. A former software engineer, Braganza usually had no problem using technology, but he has had to relearn how to use his phone and connect his iPad to Wi-Fi.
“He is still struggling quite a bit with remembering how to do basic things,” Maria Braganza said. “He has times of being really depressed because he feels like he’s not making progress.”
Doctors are taking note and starting to think about what lies ahead for ICU survivors of COVID-19. They worry about the potential for disease recurrence as well as readmission for other problems, such as other infections and hip fractures.
“As COVID-19 survivors begin to recover, there will be a large burden of chronic critical illness. We expect a significant need for rehabilitation in most ICU survivors of COVID-19,” said Steve Lubinsky, MD, medical director of respiratory care at New York University Langone Tisch Hospital.
Thinking about her father, Maria Braganza brings an extra dimension to these concerns. She thinks about depression, loneliness, and social isolation among older survivors of COVID-19. These problems existed long before the pandemic, but COVID-19 has magnified them.
The rehab staff estimates that Mr. Braganza will spend 10-14 days in their program, but discharge home creates a conundrum. Before becoming ill, Mr. Braganza lived in an independent senior living facility. Now, because of social distancing, he will no longer be able to hang out and have meals with his friends.
“Dad’s already feeling really lonely in the hospital. If we stay on a semipermanent lockdown, will he be able to see the people he loves?” Maria Braganza said. “Even though somebody is older, they have a lot to give and a lot of experience. They just need a little extra to be able to have that life.”
Dr. Nolan, Dr. Chong, Dr. Mukhopadhyay, Dr. Miyakawa, Dr. Gholamrezanezhad, and Dr. Lubinsky report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
If one word describes Eladio (“Lad”) Braganza, age 77, it’s “tenacious.” For 28 days, he clung to life on a ventilator in a Seattle ICU. Now – after a 46-day hospitalization for SARS-CoV-2 infection – he’s making progress in inpatient rehab, determined to regain function.
“We were not sure if he was going to make it through his first night in the hospital, and for a while after that. We were really prepared that he would not survive his ventilator time,” his daughter, Maria Braganza, said in an interview just 5 days after her father had been transferred to inpatient rehab.
In many ways, Mr. Braganza’s experience is typical of seriously ill COVID-19 patients. Many go from walking and talking to being on a ventilator within 10 hours or less. Mr. Braganza was admitted to the hospital on March 21 and was intubated that day. To keep him on the ventilator, he was heavily sedated and unconscious at times. In the ICU, he experienced bouts of low blood pressure, a pattern of shock that occurs in COVID-19 patients and that does not always respond to fluids.
Doctors have quickly learned to treat these patients aggressively. Many patients in the ICU with COVID-19 develop an inflamed, atypical form of acute respiratory distress syndrome (ARDS), in which the lung’s compliance, or stiffness, does not match the severity of hypoxia. These patients require high levels of oxygen and high ventilator settings. Many develop pneumothorax, or collapsed lungs, because of the high pressures needed to deliver oxygen and the prolonged time on ventilation.
“The vast majority of COVID patients in the ICU have lung disease that is quite severe, much more severe than I have seen in my 20 years of doing this,” said critical care specialist Anna Nolan, MD, of the department of medicine at New York University.
After about 2 weeks, some of these patients can come off the ventilator, or they may undergo a tracheostomy, a hole in the neck through which a tube is placed to deliver oxygen. By this time, many have developed ICU-acquired weakness and muscle wasting. Some may be so debilitated that they cannot walk. Even the respiratory muscles that help them breathe may have weakened as a result of the ventilator doing the work for them.
These patients “get sick very fast, and it takes a long time for them to heal. What’s not really well appreciated is how much rehab and how much recovery time these patients are going to need,” said David Chong, MD. He is medical director of the ICU at New York–Presbyterian Hospital/Columbia University Medical Center, and he has been on the front lines during the COVID-19 surge in New York City.
The road to recovery
Regardless of the cause, many people who have a prolonged stint in the ICU face an even longer convalescence. Still-unanswered questions concern whether recovery time will be longer for those with COVID-19, compared with other illnesses, and whether some of the damage may be permanent. A number of small studies in Hong Kong and China, as well as studies of severe acute respiratory syndrome patients’ recoveries, have promoted speculation about possible long-lasting damage to lungs and other organs from COVID-19.
Yet some of these reports have left out important details about ARDS in COVID-19 patients who also may be most at risk for long-lasting damage. To clear up some of the confusion, the Pulmonary Fibrosis Foundation said on April 6 that some but not all of COVID-19 patients who develop ARDS may go on to develop lung fibrosis – scarring of the lungs – which may be permanent.
“Post-ARDS fibrosis typically is not progressive, but nonetheless can be severe and limiting. The recovery period for post-ARDS fibrosis is approximately 1 year and the residual deficits persist, but generally do not progress,” the foundation noted.
Emerging research on lung damage in COVID-19
Because the pandemic is only a few months in, it’s unclear as yet what the long-term consequences of severe COVID-19 may be. But emerging data are enabling researchers to venture an educated guess about what may happen in the months and years ahead.
The key to understanding the data is knowing that ARDS is a syndrome – the end product of a variety of diseases or insults to the lung. Under the microscope, lung damage from ARDS associated with COVID-19 is indistinguishable from lung damage resulting from other causes, such as vaping, sepsis, or shock caused by a motor vehicle accident, said Sanjay Mukhopadhyay, MD, director of pulmonary pathology at Cleveland Clinic.
Dr. Mukhopadhyay, who specializes in lung pathology, performed one of the first complete autopsies of a COVID-19 patient in the United States. In most autopsy series published to date, he said, the most common lung finding in patients who have died from COVID-19 is diffuse alveolar damage (DAD), a pattern of lung injury seen in ARDS from many other causes.
In DAD, the walls of the alveoli – thinly lined air sacs that facilitate gas exchange in the lung – develop a pink, hyaline membrane composed of damaged cells and plasma proteins that leak from capillaries in the wall of the alveolus. This hyaline membrane gets plastered against the wall of the alveolus and interferes with diffusion of oxygen into the body.
“We know what happens in ARDS from other causes. If you follow people who have been on a ventilator long term, some of their respiratory function goes back to normal,” Dr. Mukhopadhyay said. “But there are other people in whom some degree of respiratory impairment lingers. In these patients, we think the DAD progresses to an organizing stage.”
Organizing pneumonia refers to a family of diseases in which fibroblasts (cells involved in wound healing) arrive and form scar tissue that forms hyaline membranes and fibrin balls (tough proteins) that fill up the alveoli, making gas exchange very difficult.
Also called BOOP (bronchiolitis obliterans organizing pneumonia), this condition is sensitive to steroids. Early aggressive steroid treatment can prevent long-term lung damage. Without steroids, damage can become permanent. A variant of this condition is termed acute fibrinous and organizing pneumonia (AFOP), which is also sensitive to steroids. A report from France demonstrates AFOP in some patients who have died from COVID-19.
The trick is identifying who is developing BOOP and who is not, and beyond that, who might be most amenable to treatment. Use of steroids for patients with certain other problems, such as a bacterial infection on top of COVID-19, could be harmful. David H. Chong, MD, and colleagues at Columbia University Irving Medical Center, New York, are investigating this to determine which COVID-19 patients may benefit from early steroid therapy.
“It’s not clear if there is a predominant histologic type or if we are catching people at different phases of their disease, and therefore we’re seeing different lung pathology,” Dr. Chong said.
He thinks that many patients with severe COVID-19 probably will not develop this pattern of lung scarring. “We’re speculating that lung damage from severe COVID-19 is probably going to behave more like lung damage from regular ARDS, which is often reversible. We think the vast majority of these patients probably have DAD that is similar to most patients with ARDS from other etiologies,” Dr. Chong said.
That would be consistent with information from China. In an April interview with Chinese domestic media, Zhong Nanshan, MD, a pulmonologist at the head of China’s COVID-19 task force, stated that he expects that the lungs in most patients with COVID-19 will gradually recover. He was responding to a widely publicized small study that found evidence of residual lung abnormalities at hospital discharge in most patients (94%, 66/70) who suffered from COVID-19 pneumonia in Wuhan, China, from January to February 2020.
Tough research conditions
Experts say that follow-up in this Chinese study and others to date has not been nearly long enough to allow predictions about lasting lung damage in COVID-19.
They also highlight the tough conditions in which researchers are working. Few autopsies have been performed so far – autopsies take time, extra precautions must be taken to avoid spread of COVID-19, and many patients and families do not consent to an autopsy. Furthermore, autopsy data from patients who died of COVID-19 may not extrapolate to survivors.
“I would not hang my hat on any of the limited data I have seen on autopsies,” said Lina Miyakawa, MD, a critical care and pulmonary medicine specialist at Mount Sinai Hospital in New York City.
“Even though we have answers about how the lungs are damaged at the end stage, this does not elucidate any answers about the earlier lung damage from this disease,” she continued. “It would be informative to have pathological data from the early or transitional phase, to see if that may translate into a treatment modality for COVID-19 patients.”
The problem is that these patients often experience a large amount of sloughing of airway cells, along with mucous plugging (collections of mucous that can block airflow and collapse alveoli). Bronchoscopy, which is used to view the inside of the lungs and sometimes to retrieve biopsy specimens for microscopic evaluation, is too risky for many COVID-19 patients.
In addition, few CT data exist for severely ill COVID-19 patients, who can be so unstable that to transport them to undergo a CT scan can be dangerous, not to mention the concern regarding infection control.
Even if sufficient data did exist, findings from chest x-rays, CTs, pathology studies, and lung function tests do not always match up. A patient who has lung abnormalities on CT may not necessarily have clinically impaired lung function or abnormal pathologic findings, according to Ali Gholamrezanezhad, MD, an emergency radiologist who is with the department of clinical radiology at the University of Southern California, Los Angeles.
Together with colleagues at USC, Dr. Gholamrezanezhad has started a long-term study of patients who were hospitalized with COVID-19. The researchers will follow patients for at least 1 year and will use chest x-ray, chest CT, and exercise testing to evaluate lung recovery over time.
“In the acute phase, we have acute inflammation called ground glass opacities, which usually happen bilaterally in COVID-19. That is totally reversible damage that can return to normal with no scarring,” Dr. Gholamrezanezhad said.
On the basis of data from survivors of other severe pneumonias, such as Middle East respiratory syndrome, SARS-CoV-1 infection, and H1N1 influenza, Gholamrezanezhad thinks that most survivors of COVID-19 will be able to return to work and normal life, although some may show residual lung dysfunction. Age, underlying medical conditions, smoking, length of hospital stay, severity of illness, and quality of treatment may all play a role in how well these people recover.
The lung has a remarkable capacity to recover, he added. Critical illness can destroy type one pneumocytes — the cells that line the alveoli in the lung — but over time, these cells grow back and reline the lungs. When they do, they can also help repair the lungs.
On top of that, the lung has a large functional reserve, and when one section becomes damaged, the rest of the lung can compensate.
However, for some people, total maximum exercise capacity may be affected, he commented.
Mukhopadhyay said: “My feeling is you will get reversal to normal in some patients and you will get long-term fibrosis from ARDS in some survivors. The question is, how many will have complete resolution and how many will have fibrosis? To know the answer, we will need a lot more data than we have now.”
Convalescence of COVID-19 Patients
Like many who become seriously ill with COVID-19, Braganza had underlying medical problems. Before becoming ill, he had had a heart attack and stroke. He walked with a walker and had some age-related memory problems.
Five days after transfer to inpatient rehab, Braganza was walking up and down the hallway using a walker. He was still shaking off the effects of being heavily sedated for so long, and he experienced periods of confusion. When he first came off the ventilator, he mixed up days and nights. Sometimes he did not remember being so sick. A former software engineer, Braganza usually had no problem using technology, but he has had to relearn how to use his phone and connect his iPad to Wi-Fi.
“He is still struggling quite a bit with remembering how to do basic things,” Maria Braganza said. “He has times of being really depressed because he feels like he’s not making progress.”
Doctors are taking note and starting to think about what lies ahead for ICU survivors of COVID-19. They worry about the potential for disease recurrence as well as readmission for other problems, such as other infections and hip fractures.
“As COVID-19 survivors begin to recover, there will be a large burden of chronic critical illness. We expect a significant need for rehabilitation in most ICU survivors of COVID-19,” said Steve Lubinsky, MD, medical director of respiratory care at New York University Langone Tisch Hospital.
Thinking about her father, Maria Braganza brings an extra dimension to these concerns. She thinks about depression, loneliness, and social isolation among older survivors of COVID-19. These problems existed long before the pandemic, but COVID-19 has magnified them.
The rehab staff estimates that Mr. Braganza will spend 10-14 days in their program, but discharge home creates a conundrum. Before becoming ill, Mr. Braganza lived in an independent senior living facility. Now, because of social distancing, he will no longer be able to hang out and have meals with his friends.
“Dad’s already feeling really lonely in the hospital. If we stay on a semipermanent lockdown, will he be able to see the people he loves?” Maria Braganza said. “Even though somebody is older, they have a lot to give and a lot of experience. They just need a little extra to be able to have that life.”
Dr. Nolan, Dr. Chong, Dr. Mukhopadhyay, Dr. Miyakawa, Dr. Gholamrezanezhad, and Dr. Lubinsky report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘The story unfolding is worrisome’ for diabetes and COVID-19
The American Diabetes Association has dedicated a whole section of its journal, Diabetes Care, to the topic of “Diabetes and COVID-19,” publishing a range of articles with new data to help guide physicians in caring for patients.
“Certain groups are more vulnerable to COVID-19, notably older people and those with underlying medical conditions. Because diabetes is one of the conditions associated with high risk, the diabetes community urgently needs to know more about COVID-19 and its effects on people with diabetes,” an introductory commentary noted.
Entitled “COVID-19 in people with diabetes: Urgently needed lessons from early reports,” the commentary is penned by the journal’s editor-in-chief, Matthew Riddle, MD, of Oregon Health & Science University, Portland, and colleagues.
Also writing in the same issue, William T. Cefalu, MD, and colleagues from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) noted it is known that the SARS-CoV-2 virus enters cells via the angiotensin-converting enzyme 2 (ACE-2) receptor. The ACE-2 receptor is known to be in the lungs and upper respiratory tract, “but we also know that it is expressed in other tissues such as heart, small and large intestines, and pancreas,” they wrote, and also “in the kidney.”
Hence, there are emerging reports of acute kidney injury resulting from COVID-19, as well as the impact on many other endocrine/metabolic and gastrointestinal outcomes.
“Pilot clinical studies (observational and interventional) are needed that will support the understanding or treatment of COVID-19–related diseases within the mission of the NIDDK,” they stated.
Although rapidly collected, data “offer important clues”
Some of the new ground covered in the journal articles includes an analysis of COVID-19 outcomes by type of glucose-lowering medication; remote glucose monitoring in hospitalized patients with COVID-19; a suggested approach to cardiovascular risk management in the COVID-19 era, as already reported by Medscape Medical News; and the diagnosis and management of gestational diabetes during the pandemic.
Other articles provide new data for previously reported phenomena, including obesity as a risk factor for worse COVID-19 outcomes and the role of inpatient glycemic control on COVID-19 outcomes.
“The data reported in these articles were rapidly collected and analyzed, in most cases under urgent and stressful conditions,” Dr. Riddle and colleagues cautioned. “Thus, some of the analyses are understandably limited due to missing data, incomplete follow-up, and inability to identify infected but asymptomatic patients.”
Even so, they wrote, some points are clear. “The consistency of findings in these rapidly published reports is reassuring in terms of scientific validity, but the story unfolding is worrisome.”
Specifically, while diabetes does not appear to increase the likelihood of SARS-CoV-2 infection, progression to severe illness is more likely in people with diabetes and COVID-19: They are two to three times as likely to require intensive care, and to die, compared with those infected but without diabetes.
“Neither the mechanisms underlying the increased risk nor the best interventions to limit it have yet been defined, but the studies in this collection of articles offer important clues,” Dr. Riddle and colleagues wrote.
Existing insulin use linked to COVID-19 death risk
One of the articles is a retrospective study of 904 hospitalized COVID-19 patients by Yuchen Chen, MD, of the Huazhong University of Science and Technology, Wuhan, China, and colleagues.
Among the 136 patients with diabetes, risk factors for mortality included older age (adjusted odds ratio, 1.09 per year increase; P = .001) elevated C-reactive protein (aOR, 1.12; P = .043), and insulin use (aOR, 3.58; P = .009).
“Attention needs to be paid to patients with diabetes and COVID-19 who use insulin,” the Chinese authors wrote. “Whether this was due to effects of insulin itself or to characteristics of the patients for whom it was prescribed is not clear,” Dr. Riddle and colleagues noted.
Dr. Chen and colleagues also found no difference in clinical outcomes between those diabetes patients with COVID-19 who were taking an ACE inhibitor or angiotensin II type I receptor blocker, compared with those who did not, which supports existing recommendations to continue use of this type of medication.
Remote glucose monitoring a novel tool for COVID-19 isolation
Another publication, by Gilat Shehav-Zaltzman of Sheba Medical Center, Tel Hashomer, Israel, and colleagues, describes the use of remote continuous glucose monitoring (CGM) in two hospitalized COVID-19 patients who were in isolation – one with type 1 diabetes and the other with type 2 diabetes – treated with basal-bolus insulin.
Using Medtronic CGM systems, the hospital staff was able to view patients’ real-time data uploaded to the Web from computer terminals in virus-free areas outside the patients’ rooms. The hospital’s endocrinology team had trained the intensive care staff on how to replace the sensors weekly and calibrate them twice daily.
“Converting a personal CGM system originally designed for diabetes self-management to team-based, real-time remote glucose monitoring offers a novel tool for inpatient diabetes control in COVID-19 isolation facilities,” the authors wrote.
“Such a solution in addition to ongoing remotely monitored clinical parameters (such as pulse rate, electrocardiogram, and oxygen saturation) adds to quality of diabetes care while minimizing risk of staff exposure and burden,” they observed.
Dr. Riddle and colleagues concurred: “Newer methods of remotely monitoring glucose patterns could be uniquely helpful.”
Key question: Does glycemic management make a difference?
With regard to the important issue of in-hospital control of glucose, Celestino Sardu, MD, PhD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues reported on 59 patients hospitalized with confirmed COVID-19 and moderately severe pneumonia.
They were categorized as normoglycemic (n = 34) or hyperglycemic (n = 25), as well as with or without diabetes, on the basis of a diagnosis preceding the current illness. Of the 25 patients with hyperglycemia, 15 patients were treated with insulin infusion and 10 patients were not.
In a risk-adjusted analysis, both patients with hyperglycemia and patients with diabetes had a higher risk of severe disease than did those without diabetes and with normoglycemia. Patients with hyperglycemia treated with insulin infusion had a lower risk of severe disease than did patients who didn’t receive an insulin infusion.
And although they noted limitations, the authors wrote, “Our data evidenced that optimal glucose control in the immediate postadmission period for almost 18 days was associated with a significant reduction of inflammatory cytokines and procoagulative status.”
Dr. Riddle and colleagues wrote that the findings of this unrandomized comparison were interpreted “as suggesting that insulin infusion may improve outcomes.”
“If the benefits of seeking excellent glycemic control by this means are confirmed, close monitoring of glucose levels will be essential.”
More on obesity and COVID-19, this time from China
Because it has become increasingly clear that obesity is a risk factor for severe COVID-19, new data from China – where this was less apparent initially – support observations in Europe and the United States.
An article by Qingxian Cai, PhD, of Southern University of Science and Technology, Shenzhen, Guangdong, China, and colleagues looks at this. They found that, among 383 hospitalized patients with COVID-19, the 41 patients with obesity (defined as a body mass index ≥ 28 kg/m2) were significantly more likely to progress to severe disease compared with the 203 patients classified as having normal weight (BMI, 18.5-23.9), with an odds ratio of 3.4.
A similar finding comes from Feng Gao, MD, PhD, of the First Affiliated Hospital of Wenzhou (China) Medical University and colleagues, who studied 75 patients hospitalized with confirmed COVID-19 and obesity (defined as a BMI > 25 in this Asian population) to 75 patients without obesity matched by age and sex. After adjustment for clinical characteristics including the presence of diabetes, those with obesity had a threefold greater risk of progression to severe or critical COVID-19 status, with a nearly linear relationship.
Emerging from the crisis: Protect the vulnerable, increase knowledge base
As the research community emerges from the crisis, “there should be renewed efforts for multidisciplinary research ... aimed at greatly increasing the knowledge base to understand how ... the current COVID-19 threat” affects “both healthy people and people with chronic diseases and conditions,” Dr. Cefalu and colleagues concluded in their commentary.
Dr. Riddle and coauthors agreed: “We will enter a longer interval in which we must continue to support the most vulnerable populations – especially older people, those with diabetes or obesity, and those who lack the resources to limit day-to-day exposure to infection. We hope a growing sense of community will help in this task.”
Dr. Riddle has reported receiving research grant support through Oregon Health & Science University from AstraZeneca, Eli Lilly, and Novo Nordisk, and honoraria for consulting from Adocia, AstraZeneca, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Sanofi, and Theracos. Dr. Cefalu has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The American Diabetes Association has dedicated a whole section of its journal, Diabetes Care, to the topic of “Diabetes and COVID-19,” publishing a range of articles with new data to help guide physicians in caring for patients.
“Certain groups are more vulnerable to COVID-19, notably older people and those with underlying medical conditions. Because diabetes is one of the conditions associated with high risk, the diabetes community urgently needs to know more about COVID-19 and its effects on people with diabetes,” an introductory commentary noted.
Entitled “COVID-19 in people with diabetes: Urgently needed lessons from early reports,” the commentary is penned by the journal’s editor-in-chief, Matthew Riddle, MD, of Oregon Health & Science University, Portland, and colleagues.
Also writing in the same issue, William T. Cefalu, MD, and colleagues from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) noted it is known that the SARS-CoV-2 virus enters cells via the angiotensin-converting enzyme 2 (ACE-2) receptor. The ACE-2 receptor is known to be in the lungs and upper respiratory tract, “but we also know that it is expressed in other tissues such as heart, small and large intestines, and pancreas,” they wrote, and also “in the kidney.”
Hence, there are emerging reports of acute kidney injury resulting from COVID-19, as well as the impact on many other endocrine/metabolic and gastrointestinal outcomes.
“Pilot clinical studies (observational and interventional) are needed that will support the understanding or treatment of COVID-19–related diseases within the mission of the NIDDK,” they stated.
Although rapidly collected, data “offer important clues”
Some of the new ground covered in the journal articles includes an analysis of COVID-19 outcomes by type of glucose-lowering medication; remote glucose monitoring in hospitalized patients with COVID-19; a suggested approach to cardiovascular risk management in the COVID-19 era, as already reported by Medscape Medical News; and the diagnosis and management of gestational diabetes during the pandemic.
Other articles provide new data for previously reported phenomena, including obesity as a risk factor for worse COVID-19 outcomes and the role of inpatient glycemic control on COVID-19 outcomes.
“The data reported in these articles were rapidly collected and analyzed, in most cases under urgent and stressful conditions,” Dr. Riddle and colleagues cautioned. “Thus, some of the analyses are understandably limited due to missing data, incomplete follow-up, and inability to identify infected but asymptomatic patients.”
Even so, they wrote, some points are clear. “The consistency of findings in these rapidly published reports is reassuring in terms of scientific validity, but the story unfolding is worrisome.”
Specifically, while diabetes does not appear to increase the likelihood of SARS-CoV-2 infection, progression to severe illness is more likely in people with diabetes and COVID-19: They are two to three times as likely to require intensive care, and to die, compared with those infected but without diabetes.
“Neither the mechanisms underlying the increased risk nor the best interventions to limit it have yet been defined, but the studies in this collection of articles offer important clues,” Dr. Riddle and colleagues wrote.
Existing insulin use linked to COVID-19 death risk
One of the articles is a retrospective study of 904 hospitalized COVID-19 patients by Yuchen Chen, MD, of the Huazhong University of Science and Technology, Wuhan, China, and colleagues.
Among the 136 patients with diabetes, risk factors for mortality included older age (adjusted odds ratio, 1.09 per year increase; P = .001) elevated C-reactive protein (aOR, 1.12; P = .043), and insulin use (aOR, 3.58; P = .009).
“Attention needs to be paid to patients with diabetes and COVID-19 who use insulin,” the Chinese authors wrote. “Whether this was due to effects of insulin itself or to characteristics of the patients for whom it was prescribed is not clear,” Dr. Riddle and colleagues noted.
Dr. Chen and colleagues also found no difference in clinical outcomes between those diabetes patients with COVID-19 who were taking an ACE inhibitor or angiotensin II type I receptor blocker, compared with those who did not, which supports existing recommendations to continue use of this type of medication.
Remote glucose monitoring a novel tool for COVID-19 isolation
Another publication, by Gilat Shehav-Zaltzman of Sheba Medical Center, Tel Hashomer, Israel, and colleagues, describes the use of remote continuous glucose monitoring (CGM) in two hospitalized COVID-19 patients who were in isolation – one with type 1 diabetes and the other with type 2 diabetes – treated with basal-bolus insulin.
Using Medtronic CGM systems, the hospital staff was able to view patients’ real-time data uploaded to the Web from computer terminals in virus-free areas outside the patients’ rooms. The hospital’s endocrinology team had trained the intensive care staff on how to replace the sensors weekly and calibrate them twice daily.
“Converting a personal CGM system originally designed for diabetes self-management to team-based, real-time remote glucose monitoring offers a novel tool for inpatient diabetes control in COVID-19 isolation facilities,” the authors wrote.
“Such a solution in addition to ongoing remotely monitored clinical parameters (such as pulse rate, electrocardiogram, and oxygen saturation) adds to quality of diabetes care while minimizing risk of staff exposure and burden,” they observed.
Dr. Riddle and colleagues concurred: “Newer methods of remotely monitoring glucose patterns could be uniquely helpful.”
Key question: Does glycemic management make a difference?
With regard to the important issue of in-hospital control of glucose, Celestino Sardu, MD, PhD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues reported on 59 patients hospitalized with confirmed COVID-19 and moderately severe pneumonia.
They were categorized as normoglycemic (n = 34) or hyperglycemic (n = 25), as well as with or without diabetes, on the basis of a diagnosis preceding the current illness. Of the 25 patients with hyperglycemia, 15 patients were treated with insulin infusion and 10 patients were not.
In a risk-adjusted analysis, both patients with hyperglycemia and patients with diabetes had a higher risk of severe disease than did those without diabetes and with normoglycemia. Patients with hyperglycemia treated with insulin infusion had a lower risk of severe disease than did patients who didn’t receive an insulin infusion.
And although they noted limitations, the authors wrote, “Our data evidenced that optimal glucose control in the immediate postadmission period for almost 18 days was associated with a significant reduction of inflammatory cytokines and procoagulative status.”
Dr. Riddle and colleagues wrote that the findings of this unrandomized comparison were interpreted “as suggesting that insulin infusion may improve outcomes.”
“If the benefits of seeking excellent glycemic control by this means are confirmed, close monitoring of glucose levels will be essential.”
More on obesity and COVID-19, this time from China
Because it has become increasingly clear that obesity is a risk factor for severe COVID-19, new data from China – where this was less apparent initially – support observations in Europe and the United States.
An article by Qingxian Cai, PhD, of Southern University of Science and Technology, Shenzhen, Guangdong, China, and colleagues looks at this. They found that, among 383 hospitalized patients with COVID-19, the 41 patients with obesity (defined as a body mass index ≥ 28 kg/m2) were significantly more likely to progress to severe disease compared with the 203 patients classified as having normal weight (BMI, 18.5-23.9), with an odds ratio of 3.4.
A similar finding comes from Feng Gao, MD, PhD, of the First Affiliated Hospital of Wenzhou (China) Medical University and colleagues, who studied 75 patients hospitalized with confirmed COVID-19 and obesity (defined as a BMI > 25 in this Asian population) to 75 patients without obesity matched by age and sex. After adjustment for clinical characteristics including the presence of diabetes, those with obesity had a threefold greater risk of progression to severe or critical COVID-19 status, with a nearly linear relationship.
Emerging from the crisis: Protect the vulnerable, increase knowledge base
As the research community emerges from the crisis, “there should be renewed efforts for multidisciplinary research ... aimed at greatly increasing the knowledge base to understand how ... the current COVID-19 threat” affects “both healthy people and people with chronic diseases and conditions,” Dr. Cefalu and colleagues concluded in their commentary.
Dr. Riddle and coauthors agreed: “We will enter a longer interval in which we must continue to support the most vulnerable populations – especially older people, those with diabetes or obesity, and those who lack the resources to limit day-to-day exposure to infection. We hope a growing sense of community will help in this task.”
Dr. Riddle has reported receiving research grant support through Oregon Health & Science University from AstraZeneca, Eli Lilly, and Novo Nordisk, and honoraria for consulting from Adocia, AstraZeneca, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Sanofi, and Theracos. Dr. Cefalu has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The American Diabetes Association has dedicated a whole section of its journal, Diabetes Care, to the topic of “Diabetes and COVID-19,” publishing a range of articles with new data to help guide physicians in caring for patients.
“Certain groups are more vulnerable to COVID-19, notably older people and those with underlying medical conditions. Because diabetes is one of the conditions associated with high risk, the diabetes community urgently needs to know more about COVID-19 and its effects on people with diabetes,” an introductory commentary noted.
Entitled “COVID-19 in people with diabetes: Urgently needed lessons from early reports,” the commentary is penned by the journal’s editor-in-chief, Matthew Riddle, MD, of Oregon Health & Science University, Portland, and colleagues.
Also writing in the same issue, William T. Cefalu, MD, and colleagues from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) noted it is known that the SARS-CoV-2 virus enters cells via the angiotensin-converting enzyme 2 (ACE-2) receptor. The ACE-2 receptor is known to be in the lungs and upper respiratory tract, “but we also know that it is expressed in other tissues such as heart, small and large intestines, and pancreas,” they wrote, and also “in the kidney.”
Hence, there are emerging reports of acute kidney injury resulting from COVID-19, as well as the impact on many other endocrine/metabolic and gastrointestinal outcomes.
“Pilot clinical studies (observational and interventional) are needed that will support the understanding or treatment of COVID-19–related diseases within the mission of the NIDDK,” they stated.
Although rapidly collected, data “offer important clues”
Some of the new ground covered in the journal articles includes an analysis of COVID-19 outcomes by type of glucose-lowering medication; remote glucose monitoring in hospitalized patients with COVID-19; a suggested approach to cardiovascular risk management in the COVID-19 era, as already reported by Medscape Medical News; and the diagnosis and management of gestational diabetes during the pandemic.
Other articles provide new data for previously reported phenomena, including obesity as a risk factor for worse COVID-19 outcomes and the role of inpatient glycemic control on COVID-19 outcomes.
“The data reported in these articles were rapidly collected and analyzed, in most cases under urgent and stressful conditions,” Dr. Riddle and colleagues cautioned. “Thus, some of the analyses are understandably limited due to missing data, incomplete follow-up, and inability to identify infected but asymptomatic patients.”
Even so, they wrote, some points are clear. “The consistency of findings in these rapidly published reports is reassuring in terms of scientific validity, but the story unfolding is worrisome.”
Specifically, while diabetes does not appear to increase the likelihood of SARS-CoV-2 infection, progression to severe illness is more likely in people with diabetes and COVID-19: They are two to three times as likely to require intensive care, and to die, compared with those infected but without diabetes.
“Neither the mechanisms underlying the increased risk nor the best interventions to limit it have yet been defined, but the studies in this collection of articles offer important clues,” Dr. Riddle and colleagues wrote.
Existing insulin use linked to COVID-19 death risk
One of the articles is a retrospective study of 904 hospitalized COVID-19 patients by Yuchen Chen, MD, of the Huazhong University of Science and Technology, Wuhan, China, and colleagues.
Among the 136 patients with diabetes, risk factors for mortality included older age (adjusted odds ratio, 1.09 per year increase; P = .001) elevated C-reactive protein (aOR, 1.12; P = .043), and insulin use (aOR, 3.58; P = .009).
“Attention needs to be paid to patients with diabetes and COVID-19 who use insulin,” the Chinese authors wrote. “Whether this was due to effects of insulin itself or to characteristics of the patients for whom it was prescribed is not clear,” Dr. Riddle and colleagues noted.
Dr. Chen and colleagues also found no difference in clinical outcomes between those diabetes patients with COVID-19 who were taking an ACE inhibitor or angiotensin II type I receptor blocker, compared with those who did not, which supports existing recommendations to continue use of this type of medication.
Remote glucose monitoring a novel tool for COVID-19 isolation
Another publication, by Gilat Shehav-Zaltzman of Sheba Medical Center, Tel Hashomer, Israel, and colleagues, describes the use of remote continuous glucose monitoring (CGM) in two hospitalized COVID-19 patients who were in isolation – one with type 1 diabetes and the other with type 2 diabetes – treated with basal-bolus insulin.
Using Medtronic CGM systems, the hospital staff was able to view patients’ real-time data uploaded to the Web from computer terminals in virus-free areas outside the patients’ rooms. The hospital’s endocrinology team had trained the intensive care staff on how to replace the sensors weekly and calibrate them twice daily.
“Converting a personal CGM system originally designed for diabetes self-management to team-based, real-time remote glucose monitoring offers a novel tool for inpatient diabetes control in COVID-19 isolation facilities,” the authors wrote.
“Such a solution in addition to ongoing remotely monitored clinical parameters (such as pulse rate, electrocardiogram, and oxygen saturation) adds to quality of diabetes care while minimizing risk of staff exposure and burden,” they observed.
Dr. Riddle and colleagues concurred: “Newer methods of remotely monitoring glucose patterns could be uniquely helpful.”
Key question: Does glycemic management make a difference?
With regard to the important issue of in-hospital control of glucose, Celestino Sardu, MD, PhD, of the University of Campania Luigi Vanvitelli, Naples, Italy, and colleagues reported on 59 patients hospitalized with confirmed COVID-19 and moderately severe pneumonia.
They were categorized as normoglycemic (n = 34) or hyperglycemic (n = 25), as well as with or without diabetes, on the basis of a diagnosis preceding the current illness. Of the 25 patients with hyperglycemia, 15 patients were treated with insulin infusion and 10 patients were not.
In a risk-adjusted analysis, both patients with hyperglycemia and patients with diabetes had a higher risk of severe disease than did those without diabetes and with normoglycemia. Patients with hyperglycemia treated with insulin infusion had a lower risk of severe disease than did patients who didn’t receive an insulin infusion.
And although they noted limitations, the authors wrote, “Our data evidenced that optimal glucose control in the immediate postadmission period for almost 18 days was associated with a significant reduction of inflammatory cytokines and procoagulative status.”
Dr. Riddle and colleagues wrote that the findings of this unrandomized comparison were interpreted “as suggesting that insulin infusion may improve outcomes.”
“If the benefits of seeking excellent glycemic control by this means are confirmed, close monitoring of glucose levels will be essential.”
More on obesity and COVID-19, this time from China
Because it has become increasingly clear that obesity is a risk factor for severe COVID-19, new data from China – where this was less apparent initially – support observations in Europe and the United States.
An article by Qingxian Cai, PhD, of Southern University of Science and Technology, Shenzhen, Guangdong, China, and colleagues looks at this. They found that, among 383 hospitalized patients with COVID-19, the 41 patients with obesity (defined as a body mass index ≥ 28 kg/m2) were significantly more likely to progress to severe disease compared with the 203 patients classified as having normal weight (BMI, 18.5-23.9), with an odds ratio of 3.4.
A similar finding comes from Feng Gao, MD, PhD, of the First Affiliated Hospital of Wenzhou (China) Medical University and colleagues, who studied 75 patients hospitalized with confirmed COVID-19 and obesity (defined as a BMI > 25 in this Asian population) to 75 patients without obesity matched by age and sex. After adjustment for clinical characteristics including the presence of diabetes, those with obesity had a threefold greater risk of progression to severe or critical COVID-19 status, with a nearly linear relationship.
Emerging from the crisis: Protect the vulnerable, increase knowledge base
As the research community emerges from the crisis, “there should be renewed efforts for multidisciplinary research ... aimed at greatly increasing the knowledge base to understand how ... the current COVID-19 threat” affects “both healthy people and people with chronic diseases and conditions,” Dr. Cefalu and colleagues concluded in their commentary.
Dr. Riddle and coauthors agreed: “We will enter a longer interval in which we must continue to support the most vulnerable populations – especially older people, those with diabetes or obesity, and those who lack the resources to limit day-to-day exposure to infection. We hope a growing sense of community will help in this task.”
Dr. Riddle has reported receiving research grant support through Oregon Health & Science University from AstraZeneca, Eli Lilly, and Novo Nordisk, and honoraria for consulting from Adocia, AstraZeneca, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Sanofi, and Theracos. Dr. Cefalu has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Can you catch COVID-19 through your eyes?
You can catch COVID-19 if an infected person coughs or sneezes and contagious droplets enter your nose or mouth. But can you become ill if the virus lands in your eyes?
Virologist Joseph Fair, PhD, an NBC News contributor, raised that concern when he became critically ill with COVID-19, the disease caused by the coronavirus. From a hospital bed in his hometown of New Orleans, he told the network that he had flown on a crowded plane where flight attendants weren’t wearing masks. He wore a mask and gloves, but no eye protection.
“My best guess,” he told the interviewer, “was that it came through the eye route.”
Asked if people should start wearing eye protection, Dr. Fair replied, “In my opinion, yes.”
While Dr. Fair is convinced that eye protection helps, other experts aren’t sure. So much remains unknown about the new coronavirus, SARS-CoV-2, that researchers are still trying to establish whether infection can actually happen through the eyes.
“I don’t think we can answer that question with 100% confidence at this time,” said H. Nida Sen, MD, director of the uveitis clinic at the National Eye Institute in Bethesda, Md., and a clinical investigator who is studying the effects of COVID-19 on the eye. But, she says, “I think it is biologically plausible.”
Some research has begun pointing in that direction, according to Elia Duh, MD, a researcher and professor of ophthalmology at Johns Hopkins University in Baltimore.
The clear tissue that covers the white of the eye and lines the inside of the eyelid, known as the conjunctiva, “can be infected by other viruses, such as adenoviruses associated with the common cold and the herpes simplex virus,” he said.
There’s the same chance of infection with SARS-CoV-2, said Dr. Duh. “ just like the nasal passages are exposed. In addition, people rub and touch their eyes a lot. So there’s certainly already the vulnerability.”
To study whether SARS-CoV-2 could infect the eyes, Dr. Duh and fellow researchers at Johns Hopkins looked at whether the eye’s surface cells possess key factors that make the virus more likely to enter and infect them.
In their study (BioRxiv. 2020 May 9. doi: 10.1101/2020.05.09.086165), which is now being peer-reviewed, the team examined 10 postmortem eyes and five surgical samples of conjunctiva from patients who did not have the coronavirus. They wanted to see whether the eyes’ surface cells produced the key receptor for coronavirus, the ACE2 receptor.
For SARS-CoV-2 to enter a cell, “the cell has to have ACE2 on its surface so that the coronavirus can latch onto it and gain entry into the cell,” Dr. Duh said.
Not much research existed on ACE2 and the eye’s surface cells, he said. “We were really struck that ACE2 was clearly present in the surface cells of all of the specimens.” In addition, the researchers found that the eye’s surface cells also produce TMPRSS2, an enzyme that helps the virus enter the cell.
More research is needed for a definitive answer, Dr. Duh said. But “all of this evidence together seems to suggest that there’s a good likelihood that the ocular surface cells are susceptible to infection by coronavirus.”
If that’s the case, the virus then could be transmitted through the tear ducts that connect the eyes to the nasal cavity and subsequently infect the respiratory cells, he said.
Edward E. Manche, MD, professor of ophthalmology at Stanford (Calif.) University, said that while doctors don’t know for sure, many think eye infection can happen. “I think it’s widely believed now that you can acquire it through the eye. The way the virus works, it’s most commonly transmitted through the mouth and nasal passages. We have mucosal tissues where it can get in.”
Dr. Manche said the eyes would be “the least common mode of transmission.”
Besides looking at the eyes as an entryway, researchers are exploring whether people with SARS-CoV-2 in their eyes could infect others through their tears or eye secretions.
“The virus has been detected in tears and conjunctival swab specimens from individuals with COVID-19,” Dr. Duh said. “If someone rubs their eyes and then touches someone else or touches a surface, that kind of transmission mechanism could occur.
“It again highlights how contagious the coronavirus is and how stealthy it can be in its contagiousness,” he said.
If it turns out that the coronavirus can infect the eyes, the virus could persist there as a source of contagion, Dr. Duh said. “The eyes and tears could serve as a source of infection to others for longer.” He noted a case of a COVID-infected woman with conjunctivitis who still had detectable virus in her eyes 3 weeks after her symptoms started.
Conjunctivitis, commonly called pink eye, could be a symptom of COVID-19, said Dr. Sen, who is an ophthalmologist. She recommends that people get tested for COVID-19 if they have this condition, which is marked by redness, itchiness, tearing, discharge, and a gritty sensation in the eye.
Dr. Fair, the virologist, was released from the hospital to recover at home and continued to urge eye protection. “People like to call people like me fearmongers ... but the reality is, we’re just trying to keep them safe,” he told NBC News.
The CDC hasn’t issued such advice. In an email, the agency said it “does not have specific recommendations for the public regarding eye protection. However, in health care settings, the CDC does recommend eye protection for health care workers to prevent transmission via droplets.”
Dr. Sen agrees. “For the general public, I don’t think we have enough data to suggest that they should be covering the eyes in some form,” she said.
When she goes to the grocery store, she doesn’t wear eye protection. “I am only wearing goggles when I’m seeing ophthalmology patients up close, basically because I’m 4 or 5 inches away from them.”
But fuller protection – a mask, gloves, and even eye protection, such as goggles – might help those taking care of a COVID-19 patient at home, Dr. Manche said. “If you’re caring for somebody, that’s a much higher risk because they’re shedding viral load. You lessen the chance of transmission.”
For the public, Dr. Sen stresses the continued importance of hand hygiene. “In an abundance of caution, I would still encourage handwashing and not touching the eye for many reasons, not just COVID. You can transmit simple infections to your eye. We have other viruses and bacteria that are circulating in the environment and in our bodies elsewhere, so we can easily carry those to the eyes.”
Switching from contact lenses to eyeglasses could help cut down on touching the eyes, she says. Eyeglasses can also be a “mechanical barrier” to keep hands away.
Eyeglasses might block some droplets if someone nearby sneezes or coughs, Dr. Manche said, although they “aren’t sealed around the edges. They’re not like true medical goggles that are going to keep out the virus.”
Dr. Duh agrees that health care workers must don eye protection, but he said the public doesn’t need to start wearing goggles, face shields, or other eye protection. “I still think the major mode of transmission is through the nasal passages and the respiratory system,” he said.
It’s unclear whether eye protection is warranted for airplane passengers, Dr. Manche said. “It probably wouldn’t hurt, but I think the more important thing would be to take precautions: wearing a face mask, washing your hands, cleaning the seats and tray tables in front of you, and not touching things and touching your face and eyes.”
A version of this article originally appeared on WebMD.com.
You can catch COVID-19 if an infected person coughs or sneezes and contagious droplets enter your nose or mouth. But can you become ill if the virus lands in your eyes?
Virologist Joseph Fair, PhD, an NBC News contributor, raised that concern when he became critically ill with COVID-19, the disease caused by the coronavirus. From a hospital bed in his hometown of New Orleans, he told the network that he had flown on a crowded plane where flight attendants weren’t wearing masks. He wore a mask and gloves, but no eye protection.
“My best guess,” he told the interviewer, “was that it came through the eye route.”
Asked if people should start wearing eye protection, Dr. Fair replied, “In my opinion, yes.”
While Dr. Fair is convinced that eye protection helps, other experts aren’t sure. So much remains unknown about the new coronavirus, SARS-CoV-2, that researchers are still trying to establish whether infection can actually happen through the eyes.
“I don’t think we can answer that question with 100% confidence at this time,” said H. Nida Sen, MD, director of the uveitis clinic at the National Eye Institute in Bethesda, Md., and a clinical investigator who is studying the effects of COVID-19 on the eye. But, she says, “I think it is biologically plausible.”
Some research has begun pointing in that direction, according to Elia Duh, MD, a researcher and professor of ophthalmology at Johns Hopkins University in Baltimore.
The clear tissue that covers the white of the eye and lines the inside of the eyelid, known as the conjunctiva, “can be infected by other viruses, such as adenoviruses associated with the common cold and the herpes simplex virus,” he said.
There’s the same chance of infection with SARS-CoV-2, said Dr. Duh. “ just like the nasal passages are exposed. In addition, people rub and touch their eyes a lot. So there’s certainly already the vulnerability.”
To study whether SARS-CoV-2 could infect the eyes, Dr. Duh and fellow researchers at Johns Hopkins looked at whether the eye’s surface cells possess key factors that make the virus more likely to enter and infect them.
In their study (BioRxiv. 2020 May 9. doi: 10.1101/2020.05.09.086165), which is now being peer-reviewed, the team examined 10 postmortem eyes and five surgical samples of conjunctiva from patients who did not have the coronavirus. They wanted to see whether the eyes’ surface cells produced the key receptor for coronavirus, the ACE2 receptor.
For SARS-CoV-2 to enter a cell, “the cell has to have ACE2 on its surface so that the coronavirus can latch onto it and gain entry into the cell,” Dr. Duh said.
Not much research existed on ACE2 and the eye’s surface cells, he said. “We were really struck that ACE2 was clearly present in the surface cells of all of the specimens.” In addition, the researchers found that the eye’s surface cells also produce TMPRSS2, an enzyme that helps the virus enter the cell.
More research is needed for a definitive answer, Dr. Duh said. But “all of this evidence together seems to suggest that there’s a good likelihood that the ocular surface cells are susceptible to infection by coronavirus.”
If that’s the case, the virus then could be transmitted through the tear ducts that connect the eyes to the nasal cavity and subsequently infect the respiratory cells, he said.
Edward E. Manche, MD, professor of ophthalmology at Stanford (Calif.) University, said that while doctors don’t know for sure, many think eye infection can happen. “I think it’s widely believed now that you can acquire it through the eye. The way the virus works, it’s most commonly transmitted through the mouth and nasal passages. We have mucosal tissues where it can get in.”
Dr. Manche said the eyes would be “the least common mode of transmission.”
Besides looking at the eyes as an entryway, researchers are exploring whether people with SARS-CoV-2 in their eyes could infect others through their tears or eye secretions.
“The virus has been detected in tears and conjunctival swab specimens from individuals with COVID-19,” Dr. Duh said. “If someone rubs their eyes and then touches someone else or touches a surface, that kind of transmission mechanism could occur.
“It again highlights how contagious the coronavirus is and how stealthy it can be in its contagiousness,” he said.
If it turns out that the coronavirus can infect the eyes, the virus could persist there as a source of contagion, Dr. Duh said. “The eyes and tears could serve as a source of infection to others for longer.” He noted a case of a COVID-infected woman with conjunctivitis who still had detectable virus in her eyes 3 weeks after her symptoms started.
Conjunctivitis, commonly called pink eye, could be a symptom of COVID-19, said Dr. Sen, who is an ophthalmologist. She recommends that people get tested for COVID-19 if they have this condition, which is marked by redness, itchiness, tearing, discharge, and a gritty sensation in the eye.
Dr. Fair, the virologist, was released from the hospital to recover at home and continued to urge eye protection. “People like to call people like me fearmongers ... but the reality is, we’re just trying to keep them safe,” he told NBC News.
The CDC hasn’t issued such advice. In an email, the agency said it “does not have specific recommendations for the public regarding eye protection. However, in health care settings, the CDC does recommend eye protection for health care workers to prevent transmission via droplets.”
Dr. Sen agrees. “For the general public, I don’t think we have enough data to suggest that they should be covering the eyes in some form,” she said.
When she goes to the grocery store, she doesn’t wear eye protection. “I am only wearing goggles when I’m seeing ophthalmology patients up close, basically because I’m 4 or 5 inches away from them.”
But fuller protection – a mask, gloves, and even eye protection, such as goggles – might help those taking care of a COVID-19 patient at home, Dr. Manche said. “If you’re caring for somebody, that’s a much higher risk because they’re shedding viral load. You lessen the chance of transmission.”
For the public, Dr. Sen stresses the continued importance of hand hygiene. “In an abundance of caution, I would still encourage handwashing and not touching the eye for many reasons, not just COVID. You can transmit simple infections to your eye. We have other viruses and bacteria that are circulating in the environment and in our bodies elsewhere, so we can easily carry those to the eyes.”
Switching from contact lenses to eyeglasses could help cut down on touching the eyes, she says. Eyeglasses can also be a “mechanical barrier” to keep hands away.
Eyeglasses might block some droplets if someone nearby sneezes or coughs, Dr. Manche said, although they “aren’t sealed around the edges. They’re not like true medical goggles that are going to keep out the virus.”
Dr. Duh agrees that health care workers must don eye protection, but he said the public doesn’t need to start wearing goggles, face shields, or other eye protection. “I still think the major mode of transmission is through the nasal passages and the respiratory system,” he said.
It’s unclear whether eye protection is warranted for airplane passengers, Dr. Manche said. “It probably wouldn’t hurt, but I think the more important thing would be to take precautions: wearing a face mask, washing your hands, cleaning the seats and tray tables in front of you, and not touching things and touching your face and eyes.”
A version of this article originally appeared on WebMD.com.
You can catch COVID-19 if an infected person coughs or sneezes and contagious droplets enter your nose or mouth. But can you become ill if the virus lands in your eyes?
Virologist Joseph Fair, PhD, an NBC News contributor, raised that concern when he became critically ill with COVID-19, the disease caused by the coronavirus. From a hospital bed in his hometown of New Orleans, he told the network that he had flown on a crowded plane where flight attendants weren’t wearing masks. He wore a mask and gloves, but no eye protection.
“My best guess,” he told the interviewer, “was that it came through the eye route.”
Asked if people should start wearing eye protection, Dr. Fair replied, “In my opinion, yes.”
While Dr. Fair is convinced that eye protection helps, other experts aren’t sure. So much remains unknown about the new coronavirus, SARS-CoV-2, that researchers are still trying to establish whether infection can actually happen through the eyes.
“I don’t think we can answer that question with 100% confidence at this time,” said H. Nida Sen, MD, director of the uveitis clinic at the National Eye Institute in Bethesda, Md., and a clinical investigator who is studying the effects of COVID-19 on the eye. But, she says, “I think it is biologically plausible.”
Some research has begun pointing in that direction, according to Elia Duh, MD, a researcher and professor of ophthalmology at Johns Hopkins University in Baltimore.
The clear tissue that covers the white of the eye and lines the inside of the eyelid, known as the conjunctiva, “can be infected by other viruses, such as adenoviruses associated with the common cold and the herpes simplex virus,” he said.
There’s the same chance of infection with SARS-CoV-2, said Dr. Duh. “ just like the nasal passages are exposed. In addition, people rub and touch their eyes a lot. So there’s certainly already the vulnerability.”
To study whether SARS-CoV-2 could infect the eyes, Dr. Duh and fellow researchers at Johns Hopkins looked at whether the eye’s surface cells possess key factors that make the virus more likely to enter and infect them.
In their study (BioRxiv. 2020 May 9. doi: 10.1101/2020.05.09.086165), which is now being peer-reviewed, the team examined 10 postmortem eyes and five surgical samples of conjunctiva from patients who did not have the coronavirus. They wanted to see whether the eyes’ surface cells produced the key receptor for coronavirus, the ACE2 receptor.
For SARS-CoV-2 to enter a cell, “the cell has to have ACE2 on its surface so that the coronavirus can latch onto it and gain entry into the cell,” Dr. Duh said.
Not much research existed on ACE2 and the eye’s surface cells, he said. “We were really struck that ACE2 was clearly present in the surface cells of all of the specimens.” In addition, the researchers found that the eye’s surface cells also produce TMPRSS2, an enzyme that helps the virus enter the cell.
More research is needed for a definitive answer, Dr. Duh said. But “all of this evidence together seems to suggest that there’s a good likelihood that the ocular surface cells are susceptible to infection by coronavirus.”
If that’s the case, the virus then could be transmitted through the tear ducts that connect the eyes to the nasal cavity and subsequently infect the respiratory cells, he said.
Edward E. Manche, MD, professor of ophthalmology at Stanford (Calif.) University, said that while doctors don’t know for sure, many think eye infection can happen. “I think it’s widely believed now that you can acquire it through the eye. The way the virus works, it’s most commonly transmitted through the mouth and nasal passages. We have mucosal tissues where it can get in.”
Dr. Manche said the eyes would be “the least common mode of transmission.”
Besides looking at the eyes as an entryway, researchers are exploring whether people with SARS-CoV-2 in their eyes could infect others through their tears or eye secretions.
“The virus has been detected in tears and conjunctival swab specimens from individuals with COVID-19,” Dr. Duh said. “If someone rubs their eyes and then touches someone else or touches a surface, that kind of transmission mechanism could occur.
“It again highlights how contagious the coronavirus is and how stealthy it can be in its contagiousness,” he said.
If it turns out that the coronavirus can infect the eyes, the virus could persist there as a source of contagion, Dr. Duh said. “The eyes and tears could serve as a source of infection to others for longer.” He noted a case of a COVID-infected woman with conjunctivitis who still had detectable virus in her eyes 3 weeks after her symptoms started.
Conjunctivitis, commonly called pink eye, could be a symptom of COVID-19, said Dr. Sen, who is an ophthalmologist. She recommends that people get tested for COVID-19 if they have this condition, which is marked by redness, itchiness, tearing, discharge, and a gritty sensation in the eye.
Dr. Fair, the virologist, was released from the hospital to recover at home and continued to urge eye protection. “People like to call people like me fearmongers ... but the reality is, we’re just trying to keep them safe,” he told NBC News.
The CDC hasn’t issued such advice. In an email, the agency said it “does not have specific recommendations for the public regarding eye protection. However, in health care settings, the CDC does recommend eye protection for health care workers to prevent transmission via droplets.”
Dr. Sen agrees. “For the general public, I don’t think we have enough data to suggest that they should be covering the eyes in some form,” she said.
When she goes to the grocery store, she doesn’t wear eye protection. “I am only wearing goggles when I’m seeing ophthalmology patients up close, basically because I’m 4 or 5 inches away from them.”
But fuller protection – a mask, gloves, and even eye protection, such as goggles – might help those taking care of a COVID-19 patient at home, Dr. Manche said. “If you’re caring for somebody, that’s a much higher risk because they’re shedding viral load. You lessen the chance of transmission.”
For the public, Dr. Sen stresses the continued importance of hand hygiene. “In an abundance of caution, I would still encourage handwashing and not touching the eye for many reasons, not just COVID. You can transmit simple infections to your eye. We have other viruses and bacteria that are circulating in the environment and in our bodies elsewhere, so we can easily carry those to the eyes.”
Switching from contact lenses to eyeglasses could help cut down on touching the eyes, she says. Eyeglasses can also be a “mechanical barrier” to keep hands away.
Eyeglasses might block some droplets if someone nearby sneezes or coughs, Dr. Manche said, although they “aren’t sealed around the edges. They’re not like true medical goggles that are going to keep out the virus.”
Dr. Duh agrees that health care workers must don eye protection, but he said the public doesn’t need to start wearing goggles, face shields, or other eye protection. “I still think the major mode of transmission is through the nasal passages and the respiratory system,” he said.
It’s unclear whether eye protection is warranted for airplane passengers, Dr. Manche said. “It probably wouldn’t hurt, but I think the more important thing would be to take precautions: wearing a face mask, washing your hands, cleaning the seats and tray tables in front of you, and not touching things and touching your face and eyes.”
A version of this article originally appeared on WebMD.com.
More evidence hydroxychloroquine is ineffective, harmful in COVID-19
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hydroxychloroquine and chloroquine, with or without azithromycin or clarithromycin, offer no benefit in treating patients with COVID-19 and, instead, are associated with ventricular arrhythmias and higher rates of mortality, according to a major new international study.
In the largest observational study of its kind, including close to 100,000 people in 671 hospitals on six continents, investigators compared outcomes in 15,000 patients with COVID-19 treated with hydroxychloroquine and chloroquine alone or in combination with a macrolide with 80,000 control patients with COVID-19 not receiving these agents.
Treatment with any of these medications, either alone or in combination, was associated with increased death during hospitalization; compared with about 10% in control group patients, mortality rates ranged from more than 16% to almost 24% in the treated groups.
Patients treated with hydroxychloroquine plus a macrolide showed the highest rates of serious cardiac arrhythmias, and, even after accounting for demographic factors and comorbidities, this combination was found to be associated with a more than 5-fold increase in the risk of developing a serious arrhythmia while in the hospital.
“In this real-world study, the biggest yet, we looked at 100,000 patients [with COVID-19] across six continents and found not the slightest hint of benefits and only risks, and the data is pretty straightforward,” study coauthor Frank Ruschitzka, MD, director of the Heart Center at University Hospital, Zürich, said in an interview. The study was published online May 22 in The Lancet.
‘Inconclusive’ evidence
The absence of an effective treatment for COVID-19 has led to the “repurposing” of the antimalarial drug chloroquine and its analogue hydroxychloroquine, which is used for treating autoimmune disease, but this approach is based on anecdotal evidence or open-label randomized trials that have been “largely inconclusive,” the authors wrote.
Additional agents used to treat COVID-19 are second-generation macrolides (azithromycin or clarithromycin), in combination with chloroquine or hydroxychloroquine, “despite limited evidence” and the risk for ventricular arrhythmias, the authors noted.
“Our primary question was whether there was any associated benefits of the use of hydroxychloroquine, chloroquine, or a combined regimen with macrolides in treating COVID-19, and — if there was no benefit — would there be harm?” lead author Mandeep R. Mehra, MD, MSc, William Harvey Distinguished Chair in Advanced Cardiovascular Medicine, Brigham and Women’s Hospital, Boston, said in an interview.
The investigators used data from a multinational registry comprising 671 hospitals that included patients (n = 96,032; mean age 53.8 years; 46.3% female) who had been hospitalized between Dec. 20, 2019, and April 14, 2020, with confirmed COVID-19 infection.
They also collected data about demographics, underlying comorbidities, and medical history, and medications that patients were taking at baseline.
Patients receiving treatment (n = 14,888) were divided into four groups: those receiving chloroquine alone (n = 1,868), those receiving chloroquine with a macrolide (n = 3,783), those receiving hydroxychloroquine alone (n = 3,016) and those receiving hydroxychloroquine with a macrolide (n = 6,221).
The remaining patients not treated with these regimens (n = 81,144) were regarded as the control group.
Most patients (65.9%) came from North America, followed by Europe (17.39%), Asia (7.9%), Africa (4.6%), South America (3.7%), and Australia (0.6%). Most (66.9%) were white, followed by patients of Asian origin (14.1%), black patients (9.4%), and Hispanic patients (6.2%).
Comorbidities and underlying conditions included obesity, hyperlipidemia, and hypertension in about 30%.
Comorbidities and underlying conditions
The investigators conducted multiple analyses to control for confounding variables, including Cox proportional hazards regression and propensity score matching analyses.
“In an observational study, there is always a chance of residual confounding, which is why we did propensity score based matched analyses,” Dr. Ruschitzka explained.
No significant differences were found in distribution of demographics and comorbidities between the groups.
As good as it gets
“We found no benefit in any of the four treatment regimens for hospitalized patients with COVID-19, but we did notice higher rates of death and serious ventricular arrhythmias in these patients, compared to the controls,” Dr. Mehra reported.
Of the patients in the control group, roughly 9.3% died during their hospitalization, compared with 16.4% of patients treated with chloroquine alone, 18.0% of those treated with hydroxychloroquine alone, 22.2% of those treated with chloroquine and a macrolide, and 23.8% of those treated with hydroxychloroquine and a macrolide.
After accounting for confounding variables, the researchers estimated that the excess mortality risk attributable to use of the drug regimen ranged from 34% to 45%.
Patients treated with any of the four regimens sustained more serious arrhythmias, compared with those in the control group (0.35), with the biggest increase seen in the group treated with the combination of hydroxychloroquine plus a macrolide (8.1%), followed by chloroquine with a macrolide (6.5%), hydroxychloroquine alone (6.1%), and chloroquine alone (4.3%).
“We were fairly reassured that, although the study was observational, the signals were robust and consistent across all regions of the world in diverse populations, and we did not see any muting of that signal, depending on region,” Dr. Mehra said.
“Two months ago, we were all scratching our heads about how to treat patients with COVID-19, and then came a drug [hydroxychloroquine] with some anecdotal evidence, but now we have 2 months more experience, and we looked to science to provide some answer,” Dr. Ruschitzka said.
“Although this was not a randomized, controlled trial, so we do not have a definite answer, the data provided in this [large, multinational] real-world study is as good as it gets and the best data we have,” he concluded.
“Let the science speak for itself”
Commenting on the study in an interview, Christian Funck-Brentano, MD, from the Hospital Pitié-Salpêtrière and Sorbonne University, both in Paris, said that, although the study is observational and therefore not as reliable as a randomized controlled trial, it is “nevertheless well-documented, studied a huge amount of people, and utilized several sensitivity methods, all of which showed the same results.”
Dr. Funck-Brentano, who is the coauthor of an accompanying editorial in The Lancet and was not involved with the study, said that “we now have no evidence that hydroxychloroquine and chloroquine alone or in combination with a macrolide do any good and we have potential evidence that they do harm and kill people.”
Also commenting on the study in an interview, David Holtgrave, PhD, dean of the School of Public Health at the State University of New York at Albany, said that, “while no one observational study alone would lead to a firm clinical recommendation, I think it is helpful for physicians and public health officials to be aware of the findings of the peer-reviewed observational studies to date and the National Institutes of Health COVID-19 treatment guidelines and the Food and Drug Administration’s statement of drug safety concern about hydroxychloroquine to inform their decision-making as we await the results of randomized clinical trials of these drugs for the treatment of COVID-19,” said Dr. Holtgrave, who was not involved with the study.
He added that, to his knowledge, there are “still no published studies of prophylactic use of these drugs to prevent COVID-19.”
Dr. Mehra emphasized that a cardinal principle of practicing medicine is “first do no harm” and “even in situations where you believe a desperate disease calls for desperate measures, responsible physicians should take a step back and ask if we are doing harm, and until we can say we aren’t, I don’t think it’s wise to push something like this in the absence of good efficacy data.”
Dr. Ruschitzka added that those who are encouraging the use of these agents “should review their decision based on today’s data and let the science speak for itself.”
The study was supported by the William Harvey Distinguished Chair in Advanced Cardiovascular Medicine at Brigham and Women’s Hospital, Boston. Dr. Mehra reported personal fees from Abbott, Medtronic, Janssen, Mesoblast, Portola, Bayer, Baim Institute for Clinical Research, NuPulseCV, FineHeart, Leviticus, Roivant, and Triple Gene. Dr. Ruschitzka was paid for time spent as a committee member for clinical trials, advisory boards, other forms of consulting, and lectures or presentations; these payments were made directly to the University of Zürich and no personal payments were received in relation to these trials or other activities. Dr. Funck-Brentano, his coauthor, and Dr. Holtgrave declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Is HIPAA critical?
Ignorance may be bliss for some. But as I sit here in my scenic social isolation on the Maine coast I find that, like most people, what I don’t know unsettles me. How is the COVID-19 virus spread? Does my wife’s wipe down of the doorknobs after I return from the grocery store really make us any less likely to contract the virus? Is wearing my homemade bandana face mask doing anything to protect me? I suspect not, but I wear it as a statement of courtesy and solidarity to my fellow community members.
Does the 6-foot rule make any sense? I’ve read that it is based on a study dating back to the 1930s. I’ve seen images of the 25-foot droplet plume blasting out from a sneeze and understand that, as a bicyclist, I may be generating a shower of droplets in my wake. But, are those droplets a threat to anyone I pedal by if I am symptom free? What does being a carrier mean when we are talking about COVID-19?
What makes me more vulnerable to this particular virus as an apparently healthy septuagenarian? What collection of misfortunes have fallen on those younger victims of the pandemic? How often was it genetic?
Of course, none of us has the information yet that can provide us answers. This vacuum has attracted scores of “experts” bold enough or careless enough to venture an opinion. They may have also issued a caveat, but how often have the media failed to include it in the report or buried it in the fine print at the end of the story?
My discomfort with this information void has left me and you and everyone else to our imaginations to craft our own explanations. So, I try to piece together a construct based on what I can glean from what I read and see in the news because like most people I fortunately have no first-hand information about even a single case. The number of deaths is horrifying, but may not have hit close to home and given most of us a real personal sense of the illness and its character.
Maine is a small state with just over a million inhabitants, and most of us have some connection to one another. It may be that a person is the second cousin of someone who used to live 2 miles down the road. But, there is some feeling of familiarity. We have had deaths related to COVID-19, but very scanty information other than the county about where they occurred and whether the victim was a resident of an extended care facility. We are told very little if any details about exposure as officials invoke HIPAA regulations that leave us in the dark. Other than one vague reference to a “traveling salesman” who may have introduced the virus to several nursing homes, there has been very little information about how the virus may have been spread here in Maine. Even national reports of the deaths of high-profile entertainers and retired athletes are usually draped in the same haze of privacy.
Most of us don’t need to know the names and street addresses of the victims but a few anonymous narratives that include some general information on how epidemiologists believe clusters began and propagated would help us understand our risks with just a glimmer of clarity.
Of course the epidemiologists may not have the answers we are seeking because they too are struggling to untangle connections hampered by concerns of privacy. There is no question that privacy must remain an important part of the physician-patient relationship. But a pandemic has thrown us into a situation where common sense demands that HIPAA be interpreted with an emphasis on the greater good. Finding that balance between privacy and public knowledge will continue to be one of our greatest challenges.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Ignorance may be bliss for some. But as I sit here in my scenic social isolation on the Maine coast I find that, like most people, what I don’t know unsettles me. How is the COVID-19 virus spread? Does my wife’s wipe down of the doorknobs after I return from the grocery store really make us any less likely to contract the virus? Is wearing my homemade bandana face mask doing anything to protect me? I suspect not, but I wear it as a statement of courtesy and solidarity to my fellow community members.
Does the 6-foot rule make any sense? I’ve read that it is based on a study dating back to the 1930s. I’ve seen images of the 25-foot droplet plume blasting out from a sneeze and understand that, as a bicyclist, I may be generating a shower of droplets in my wake. But, are those droplets a threat to anyone I pedal by if I am symptom free? What does being a carrier mean when we are talking about COVID-19?
What makes me more vulnerable to this particular virus as an apparently healthy septuagenarian? What collection of misfortunes have fallen on those younger victims of the pandemic? How often was it genetic?
Of course, none of us has the information yet that can provide us answers. This vacuum has attracted scores of “experts” bold enough or careless enough to venture an opinion. They may have also issued a caveat, but how often have the media failed to include it in the report or buried it in the fine print at the end of the story?
My discomfort with this information void has left me and you and everyone else to our imaginations to craft our own explanations. So, I try to piece together a construct based on what I can glean from what I read and see in the news because like most people I fortunately have no first-hand information about even a single case. The number of deaths is horrifying, but may not have hit close to home and given most of us a real personal sense of the illness and its character.
Maine is a small state with just over a million inhabitants, and most of us have some connection to one another. It may be that a person is the second cousin of someone who used to live 2 miles down the road. But, there is some feeling of familiarity. We have had deaths related to COVID-19, but very scanty information other than the county about where they occurred and whether the victim was a resident of an extended care facility. We are told very little if any details about exposure as officials invoke HIPAA regulations that leave us in the dark. Other than one vague reference to a “traveling salesman” who may have introduced the virus to several nursing homes, there has been very little information about how the virus may have been spread here in Maine. Even national reports of the deaths of high-profile entertainers and retired athletes are usually draped in the same haze of privacy.
Most of us don’t need to know the names and street addresses of the victims but a few anonymous narratives that include some general information on how epidemiologists believe clusters began and propagated would help us understand our risks with just a glimmer of clarity.
Of course the epidemiologists may not have the answers we are seeking because they too are struggling to untangle connections hampered by concerns of privacy. There is no question that privacy must remain an important part of the physician-patient relationship. But a pandemic has thrown us into a situation where common sense demands that HIPAA be interpreted with an emphasis on the greater good. Finding that balance between privacy and public knowledge will continue to be one of our greatest challenges.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Ignorance may be bliss for some. But as I sit here in my scenic social isolation on the Maine coast I find that, like most people, what I don’t know unsettles me. How is the COVID-19 virus spread? Does my wife’s wipe down of the doorknobs after I return from the grocery store really make us any less likely to contract the virus? Is wearing my homemade bandana face mask doing anything to protect me? I suspect not, but I wear it as a statement of courtesy and solidarity to my fellow community members.
Does the 6-foot rule make any sense? I’ve read that it is based on a study dating back to the 1930s. I’ve seen images of the 25-foot droplet plume blasting out from a sneeze and understand that, as a bicyclist, I may be generating a shower of droplets in my wake. But, are those droplets a threat to anyone I pedal by if I am symptom free? What does being a carrier mean when we are talking about COVID-19?
What makes me more vulnerable to this particular virus as an apparently healthy septuagenarian? What collection of misfortunes have fallen on those younger victims of the pandemic? How often was it genetic?
Of course, none of us has the information yet that can provide us answers. This vacuum has attracted scores of “experts” bold enough or careless enough to venture an opinion. They may have also issued a caveat, but how often have the media failed to include it in the report or buried it in the fine print at the end of the story?
My discomfort with this information void has left me and you and everyone else to our imaginations to craft our own explanations. So, I try to piece together a construct based on what I can glean from what I read and see in the news because like most people I fortunately have no first-hand information about even a single case. The number of deaths is horrifying, but may not have hit close to home and given most of us a real personal sense of the illness and its character.
Maine is a small state with just over a million inhabitants, and most of us have some connection to one another. It may be that a person is the second cousin of someone who used to live 2 miles down the road. But, there is some feeling of familiarity. We have had deaths related to COVID-19, but very scanty information other than the county about where they occurred and whether the victim was a resident of an extended care facility. We are told very little if any details about exposure as officials invoke HIPAA regulations that leave us in the dark. Other than one vague reference to a “traveling salesman” who may have introduced the virus to several nursing homes, there has been very little information about how the virus may have been spread here in Maine. Even national reports of the deaths of high-profile entertainers and retired athletes are usually draped in the same haze of privacy.
Most of us don’t need to know the names and street addresses of the victims but a few anonymous narratives that include some general information on how epidemiologists believe clusters began and propagated would help us understand our risks with just a glimmer of clarity.
Of course the epidemiologists may not have the answers we are seeking because they too are struggling to untangle connections hampered by concerns of privacy. There is no question that privacy must remain an important part of the physician-patient relationship. But a pandemic has thrown us into a situation where common sense demands that HIPAA be interpreted with an emphasis on the greater good. Finding that balance between privacy and public knowledge will continue to be one of our greatest challenges.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at [email protected].
Armchair epidemiology
Real epidemiologists are out knocking on doors, chasing down contacts, or hunched over their computers trying to make sense out of screens full of data and maps. A few are trying valiantly to talk some sense into our elected officials.
This leaves the rest of us with time on our hands to fabricate our own less-than-scientific explanations for the behavior of the SARS-CoV-2 virus. So I have decided to put on hold my current mental challenge of choosing which pasta shape to pair with the sauce I’ve prepared from an online recipe. Here is my educated guess based on what I can glean from media sources that may have been filtered through a variety politically biased lenses. Remember, I did go to medical school; however, when I was in college the DNA helix was still just theoretical.
From those halcyon days of mid-February when our attention was focused on the Diamond Princess quarantined in Yokohama Harbor, it didn’t take a board-certified epidemiologist to suspect that the virus was spreading through the ventilating system in the ship’s tight quarters. Subsequent outbreaks on U.S. and French military ships suggests a similar explanation.
While still not proven, it sounds like SARS-CoV-2 jumped to humans from bats. It should not surprise us that having evolved in a dense population of mammals it would thrive in other high-density populations such as New York and nursing homes. Because we have lacked a robust testing capability, it has been less obvious until recently that, while it is easily transmitted, the virus has infected many who are asymptomatic (“Antibody surveys suggesting vast undercount of coronavirus infections may be unreliable,” Gretchen Vogel, Science, April 21, 2020). Subsequent surveys seem to confirm this higher level carrier state; it suggests that the virus is far less deadly than was previously suggested. However, it seems to be a crafty little bug attacking just about any organ system it lands on.
I don’t think any of us are surprised that the elderly population with weakened immune systems, particularly those in congregate housing, has been much more vulnerable. However, many of the deaths among younger apparently healthy people have defied explanation. The anecdotal observations that physicians, particularly those who practice in-your-face medicine (e.g., ophthalmologists and otolaryngologists) may be more vulnerable raises the issue of viral load. It may be that, although it can be extremely contagious, the virus is not terribly dangerous for most people until the inoculum dose of the virus reaches a certain level. To my knowledge this dose is unknown.
A published survey of more than 300 outbreaks from 120 Chinese cities also may support my suspicion that viral load is of critical importance. The researchers found that all the “identified outbreaks of three or more cases occurred in an indoor environment, which confirms that sharing indoor space is a major SARS-CoV-2 infection risk” (Huan Qian et al. “Indoor transmission of SARS-CoV-2,” MedRxiv. 2020 Apr 7. doi: 10.1101/2020.04.04.20053058). Again, this data shouldn’t surprise us when we look back at what little we know about the outbreaks in the confined spaces on cruise ships and in nursing homes.
I’m not sure that we have any data that helps us determine whether wearing a mask in an outdoor space has any more than symbolic value when we are talking about this particular virus. We may read that the virus in a droplet can survive on the surface it lands on for 8 minutes, and we can see those slow motion videos of the impressive plume of snot spray released by a sneeze. It would seem obvious that even outside someone within 10 feet of the sneeze has a good chance of being infected. However, how much of a threat is the asymptomatic carrier who passes within three feet of you while you are out on lovely summer day stroll? This armchair epidemiologist suspects that, when we are talking about an outside space, the 6-foot guideline for small groups of a dozen or less is overly restrictive. But until we know, I’m staying put in my armchair ... outside on the porch overlooking Casco Bay.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” He has no disclosures. Email him at [email protected].
Real epidemiologists are out knocking on doors, chasing down contacts, or hunched over their computers trying to make sense out of screens full of data and maps. A few are trying valiantly to talk some sense into our elected officials.
This leaves the rest of us with time on our hands to fabricate our own less-than-scientific explanations for the behavior of the SARS-CoV-2 virus. So I have decided to put on hold my current mental challenge of choosing which pasta shape to pair with the sauce I’ve prepared from an online recipe. Here is my educated guess based on what I can glean from media sources that may have been filtered through a variety politically biased lenses. Remember, I did go to medical school; however, when I was in college the DNA helix was still just theoretical.
From those halcyon days of mid-February when our attention was focused on the Diamond Princess quarantined in Yokohama Harbor, it didn’t take a board-certified epidemiologist to suspect that the virus was spreading through the ventilating system in the ship’s tight quarters. Subsequent outbreaks on U.S. and French military ships suggests a similar explanation.
While still not proven, it sounds like SARS-CoV-2 jumped to humans from bats. It should not surprise us that having evolved in a dense population of mammals it would thrive in other high-density populations such as New York and nursing homes. Because we have lacked a robust testing capability, it has been less obvious until recently that, while it is easily transmitted, the virus has infected many who are asymptomatic (“Antibody surveys suggesting vast undercount of coronavirus infections may be unreliable,” Gretchen Vogel, Science, April 21, 2020). Subsequent surveys seem to confirm this higher level carrier state; it suggests that the virus is far less deadly than was previously suggested. However, it seems to be a crafty little bug attacking just about any organ system it lands on.
I don’t think any of us are surprised that the elderly population with weakened immune systems, particularly those in congregate housing, has been much more vulnerable. However, many of the deaths among younger apparently healthy people have defied explanation. The anecdotal observations that physicians, particularly those who practice in-your-face medicine (e.g., ophthalmologists and otolaryngologists) may be more vulnerable raises the issue of viral load. It may be that, although it can be extremely contagious, the virus is not terribly dangerous for most people until the inoculum dose of the virus reaches a certain level. To my knowledge this dose is unknown.
A published survey of more than 300 outbreaks from 120 Chinese cities also may support my suspicion that viral load is of critical importance. The researchers found that all the “identified outbreaks of three or more cases occurred in an indoor environment, which confirms that sharing indoor space is a major SARS-CoV-2 infection risk” (Huan Qian et al. “Indoor transmission of SARS-CoV-2,” MedRxiv. 2020 Apr 7. doi: 10.1101/2020.04.04.20053058). Again, this data shouldn’t surprise us when we look back at what little we know about the outbreaks in the confined spaces on cruise ships and in nursing homes.
I’m not sure that we have any data that helps us determine whether wearing a mask in an outdoor space has any more than symbolic value when we are talking about this particular virus. We may read that the virus in a droplet can survive on the surface it lands on for 8 minutes, and we can see those slow motion videos of the impressive plume of snot spray released by a sneeze. It would seem obvious that even outside someone within 10 feet of the sneeze has a good chance of being infected. However, how much of a threat is the asymptomatic carrier who passes within three feet of you while you are out on lovely summer day stroll? This armchair epidemiologist suspects that, when we are talking about an outside space, the 6-foot guideline for small groups of a dozen or less is overly restrictive. But until we know, I’m staying put in my armchair ... outside on the porch overlooking Casco Bay.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” He has no disclosures. Email him at [email protected].
Real epidemiologists are out knocking on doors, chasing down contacts, or hunched over their computers trying to make sense out of screens full of data and maps. A few are trying valiantly to talk some sense into our elected officials.
This leaves the rest of us with time on our hands to fabricate our own less-than-scientific explanations for the behavior of the SARS-CoV-2 virus. So I have decided to put on hold my current mental challenge of choosing which pasta shape to pair with the sauce I’ve prepared from an online recipe. Here is my educated guess based on what I can glean from media sources that may have been filtered through a variety politically biased lenses. Remember, I did go to medical school; however, when I was in college the DNA helix was still just theoretical.
From those halcyon days of mid-February when our attention was focused on the Diamond Princess quarantined in Yokohama Harbor, it didn’t take a board-certified epidemiologist to suspect that the virus was spreading through the ventilating system in the ship’s tight quarters. Subsequent outbreaks on U.S. and French military ships suggests a similar explanation.
While still not proven, it sounds like SARS-CoV-2 jumped to humans from bats. It should not surprise us that having evolved in a dense population of mammals it would thrive in other high-density populations such as New York and nursing homes. Because we have lacked a robust testing capability, it has been less obvious until recently that, while it is easily transmitted, the virus has infected many who are asymptomatic (“Antibody surveys suggesting vast undercount of coronavirus infections may be unreliable,” Gretchen Vogel, Science, April 21, 2020). Subsequent surveys seem to confirm this higher level carrier state; it suggests that the virus is far less deadly than was previously suggested. However, it seems to be a crafty little bug attacking just about any organ system it lands on.
I don’t think any of us are surprised that the elderly population with weakened immune systems, particularly those in congregate housing, has been much more vulnerable. However, many of the deaths among younger apparently healthy people have defied explanation. The anecdotal observations that physicians, particularly those who practice in-your-face medicine (e.g., ophthalmologists and otolaryngologists) may be more vulnerable raises the issue of viral load. It may be that, although it can be extremely contagious, the virus is not terribly dangerous for most people until the inoculum dose of the virus reaches a certain level. To my knowledge this dose is unknown.
A published survey of more than 300 outbreaks from 120 Chinese cities also may support my suspicion that viral load is of critical importance. The researchers found that all the “identified outbreaks of three or more cases occurred in an indoor environment, which confirms that sharing indoor space is a major SARS-CoV-2 infection risk” (Huan Qian et al. “Indoor transmission of SARS-CoV-2,” MedRxiv. 2020 Apr 7. doi: 10.1101/2020.04.04.20053058). Again, this data shouldn’t surprise us when we look back at what little we know about the outbreaks in the confined spaces on cruise ships and in nursing homes.
I’m not sure that we have any data that helps us determine whether wearing a mask in an outdoor space has any more than symbolic value when we are talking about this particular virus. We may read that the virus in a droplet can survive on the surface it lands on for 8 minutes, and we can see those slow motion videos of the impressive plume of snot spray released by a sneeze. It would seem obvious that even outside someone within 10 feet of the sneeze has a good chance of being infected. However, how much of a threat is the asymptomatic carrier who passes within three feet of you while you are out on lovely summer day stroll? This armchair epidemiologist suspects that, when we are talking about an outside space, the 6-foot guideline for small groups of a dozen or less is overly restrictive. But until we know, I’m staying put in my armchair ... outside on the porch overlooking Casco Bay.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” He has no disclosures. Email him at [email protected].
Vaccination regimen effective in preventing pneumonia in MM patients
Patients with hematological malignancies are at high risk of invasive Staphylococcus pneumoniae. Multiple myeloma (MM) patients, in particular, have been found to have one of the highest incidences of invasive pneumococcal disease. However, researchers found that a full three-dose vaccination regimen by 13-valent pneumococcal conjugate (PCV13) vaccine was protective in MM patients when provided between treatment courses, according to a study reported in Vaccine.
The researchers performed a prospective study of 18 adult patients who were vaccinated with PCV13, compared with 18 control-matched patients from 2017 to 2020. The three-dose vaccination regimen was provided between treatment courses with novel target agents (bortezomib, lenalidomide, ixazomib) with a minimum of a 1-month interval. They used the incidence of pneumonias during the one-year observation period as the primary outcome.
Totally there were 12 cases (33.3%) of clinically and radiologically confirmed pneumonias in the entire study group (n = 36), with a distribution between the vaccinated and nonvaccinated groups of 3 (16.7%) and 9 (50%). respectively (P = .037).
The absolute risk reduction seen with vaccination was 33.3%, and the number needed to treat with PCV13 vaccination in MM patients receiving novel agents was 3.0; (95% confidence interval 1.61-22.1). In addition, there were no adverse effects seen from vaccination, according to the authors.
“Despite the expected decrease in immunological response to vaccination during the chemotherapy, we have shown the clinical effectiveness of a PCV13 vaccination schedule based on 3 doses given with a minimum 1 month interval between the courses of novel agents,” the investigators concluded.
The authors reported that they had no relevant disclosures.
SOURCE: Stoma I et al. Vaccine. 2020 May 14; doi.org/10.1016/j.vaccine.2020.05.024.
Patients with hematological malignancies are at high risk of invasive Staphylococcus pneumoniae. Multiple myeloma (MM) patients, in particular, have been found to have one of the highest incidences of invasive pneumococcal disease. However, researchers found that a full three-dose vaccination regimen by 13-valent pneumococcal conjugate (PCV13) vaccine was protective in MM patients when provided between treatment courses, according to a study reported in Vaccine.
The researchers performed a prospective study of 18 adult patients who were vaccinated with PCV13, compared with 18 control-matched patients from 2017 to 2020. The three-dose vaccination regimen was provided between treatment courses with novel target agents (bortezomib, lenalidomide, ixazomib) with a minimum of a 1-month interval. They used the incidence of pneumonias during the one-year observation period as the primary outcome.
Totally there were 12 cases (33.3%) of clinically and radiologically confirmed pneumonias in the entire study group (n = 36), with a distribution between the vaccinated and nonvaccinated groups of 3 (16.7%) and 9 (50%). respectively (P = .037).
The absolute risk reduction seen with vaccination was 33.3%, and the number needed to treat with PCV13 vaccination in MM patients receiving novel agents was 3.0; (95% confidence interval 1.61-22.1). In addition, there were no adverse effects seen from vaccination, according to the authors.
“Despite the expected decrease in immunological response to vaccination during the chemotherapy, we have shown the clinical effectiveness of a PCV13 vaccination schedule based on 3 doses given with a minimum 1 month interval between the courses of novel agents,” the investigators concluded.
The authors reported that they had no relevant disclosures.
SOURCE: Stoma I et al. Vaccine. 2020 May 14; doi.org/10.1016/j.vaccine.2020.05.024.
Patients with hematological malignancies are at high risk of invasive Staphylococcus pneumoniae. Multiple myeloma (MM) patients, in particular, have been found to have one of the highest incidences of invasive pneumococcal disease. However, researchers found that a full three-dose vaccination regimen by 13-valent pneumococcal conjugate (PCV13) vaccine was protective in MM patients when provided between treatment courses, according to a study reported in Vaccine.
The researchers performed a prospective study of 18 adult patients who were vaccinated with PCV13, compared with 18 control-matched patients from 2017 to 2020. The three-dose vaccination regimen was provided between treatment courses with novel target agents (bortezomib, lenalidomide, ixazomib) with a minimum of a 1-month interval. They used the incidence of pneumonias during the one-year observation period as the primary outcome.
Totally there were 12 cases (33.3%) of clinically and radiologically confirmed pneumonias in the entire study group (n = 36), with a distribution between the vaccinated and nonvaccinated groups of 3 (16.7%) and 9 (50%). respectively (P = .037).
The absolute risk reduction seen with vaccination was 33.3%, and the number needed to treat with PCV13 vaccination in MM patients receiving novel agents was 3.0; (95% confidence interval 1.61-22.1). In addition, there were no adverse effects seen from vaccination, according to the authors.
“Despite the expected decrease in immunological response to vaccination during the chemotherapy, we have shown the clinical effectiveness of a PCV13 vaccination schedule based on 3 doses given with a minimum 1 month interval between the courses of novel agents,” the investigators concluded.
The authors reported that they had no relevant disclosures.
SOURCE: Stoma I et al. Vaccine. 2020 May 14; doi.org/10.1016/j.vaccine.2020.05.024.
FROM VACCINE
Pulmonology, critical care earnings on the upswing before pandemic
As the COVID spring progresses, the days before the pandemic may seem like a dream: Practices were open, waiting rooms were full of unmasked people, and PPE was plentiful.
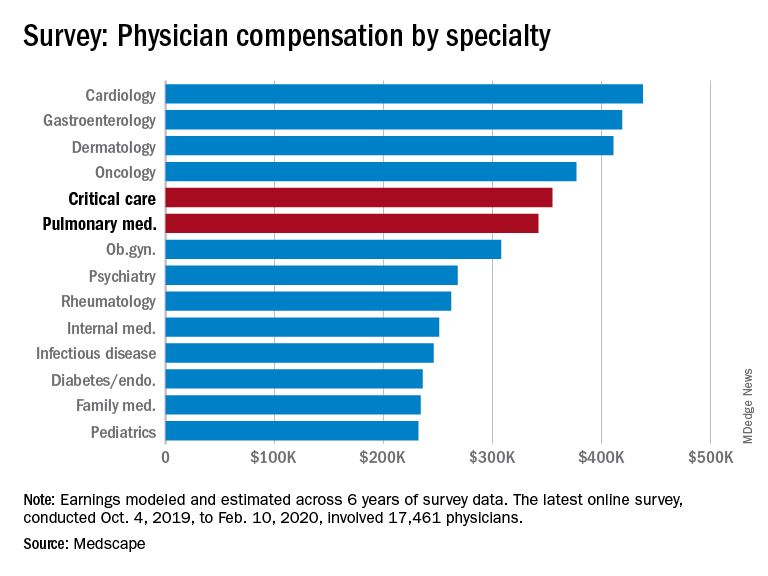
Medscape’s latest physician survey, conducted from Oct. 4, 2019, to Feb. 10, 2020, shows what pulmonology and critical care looked like just before the coronavirus arrived.
Back then, earnings were up. Average compensation reported by pulmonologists was up from $331,000 in 2019 to $342,000 this year, a 3.3% increase. For intensivists, earnings rose from $349,000 to $355,000, or 1.7%. Average income for all specialists was $346,000 in this year’s survey – 1.5% higher than the $341,000 earned in 2019, Medscape reported.
Prospects for the next year, however, are grim. “We found out that we have a 10% salary decrease effective May 2 to Dec. 25. Our bonus will be based on clinical productivity, and since our numbers are down, that is likely to go away,” a pediatric emergency physician told Medscape.
One problem area for intensivists, even before the pandemic, was paperwork and administration. Of the 26 specialties for which data are available, critical care was highest for amount of time spent on paperwork, at 19.1 hours per week. Those in pulmonary medicine spent 15.6 hours per week, which also happened to be the average for all specialists, the survey data show.
Both specialties also ranked high in denied/resubmitted claims: Intensivists were fourth among the 27 types of specialists with reliable data, with 20% of claims denied, and pulmonologists were tied for eighth at 18%, Medscape said.
Only 50% of pulmonologists surveyed said that they were being fairly compensated, putting them 26th among the 29 specialties on that list. Those in critical care medicine were 13th, with a 59% positive response, Medscape reported.
In the end, though, it looks like you can’t keep a good pulmonologist or intensivist down. When asked if they would choose medicine again, 83% of pulmonologists said yes, just one percentage point behind a three-way tie for first. Intensivists were just a little further down the list at 81%, according to the survey.
The respondents were Medscape members who had been invited to participate. The sample size was 17,461 physicians, and compensation was modeled and estimated based on a range of variables across 6 years of survey data. The sampling error was ±0.74%.
As the COVID spring progresses, the days before the pandemic may seem like a dream: Practices were open, waiting rooms were full of unmasked people, and PPE was plentiful.

Medscape’s latest physician survey, conducted from Oct. 4, 2019, to Feb. 10, 2020, shows what pulmonology and critical care looked like just before the coronavirus arrived.
Back then, earnings were up. Average compensation reported by pulmonologists was up from $331,000 in 2019 to $342,000 this year, a 3.3% increase. For intensivists, earnings rose from $349,000 to $355,000, or 1.7%. Average income for all specialists was $346,000 in this year’s survey – 1.5% higher than the $341,000 earned in 2019, Medscape reported.
Prospects for the next year, however, are grim. “We found out that we have a 10% salary decrease effective May 2 to Dec. 25. Our bonus will be based on clinical productivity, and since our numbers are down, that is likely to go away,” a pediatric emergency physician told Medscape.
One problem area for intensivists, even before the pandemic, was paperwork and administration. Of the 26 specialties for which data are available, critical care was highest for amount of time spent on paperwork, at 19.1 hours per week. Those in pulmonary medicine spent 15.6 hours per week, which also happened to be the average for all specialists, the survey data show.
Both specialties also ranked high in denied/resubmitted claims: Intensivists were fourth among the 27 types of specialists with reliable data, with 20% of claims denied, and pulmonologists were tied for eighth at 18%, Medscape said.
Only 50% of pulmonologists surveyed said that they were being fairly compensated, putting them 26th among the 29 specialties on that list. Those in critical care medicine were 13th, with a 59% positive response, Medscape reported.
In the end, though, it looks like you can’t keep a good pulmonologist or intensivist down. When asked if they would choose medicine again, 83% of pulmonologists said yes, just one percentage point behind a three-way tie for first. Intensivists were just a little further down the list at 81%, according to the survey.
The respondents were Medscape members who had been invited to participate. The sample size was 17,461 physicians, and compensation was modeled and estimated based on a range of variables across 6 years of survey data. The sampling error was ±0.74%.
As the COVID spring progresses, the days before the pandemic may seem like a dream: Practices were open, waiting rooms were full of unmasked people, and PPE was plentiful.

Medscape’s latest physician survey, conducted from Oct. 4, 2019, to Feb. 10, 2020, shows what pulmonology and critical care looked like just before the coronavirus arrived.
Back then, earnings were up. Average compensation reported by pulmonologists was up from $331,000 in 2019 to $342,000 this year, a 3.3% increase. For intensivists, earnings rose from $349,000 to $355,000, or 1.7%. Average income for all specialists was $346,000 in this year’s survey – 1.5% higher than the $341,000 earned in 2019, Medscape reported.
Prospects for the next year, however, are grim. “We found out that we have a 10% salary decrease effective May 2 to Dec. 25. Our bonus will be based on clinical productivity, and since our numbers are down, that is likely to go away,” a pediatric emergency physician told Medscape.
One problem area for intensivists, even before the pandemic, was paperwork and administration. Of the 26 specialties for which data are available, critical care was highest for amount of time spent on paperwork, at 19.1 hours per week. Those in pulmonary medicine spent 15.6 hours per week, which also happened to be the average for all specialists, the survey data show.
Both specialties also ranked high in denied/resubmitted claims: Intensivists were fourth among the 27 types of specialists with reliable data, with 20% of claims denied, and pulmonologists were tied for eighth at 18%, Medscape said.
Only 50% of pulmonologists surveyed said that they were being fairly compensated, putting them 26th among the 29 specialties on that list. Those in critical care medicine were 13th, with a 59% positive response, Medscape reported.
In the end, though, it looks like you can’t keep a good pulmonologist or intensivist down. When asked if they would choose medicine again, 83% of pulmonologists said yes, just one percentage point behind a three-way tie for first. Intensivists were just a little further down the list at 81%, according to the survey.
The respondents were Medscape members who had been invited to participate. The sample size was 17,461 physicians, and compensation was modeled and estimated based on a range of variables across 6 years of survey data. The sampling error was ±0.74%.