User login
Nintedanib cut lung function decline in interstitial lung disease with systemic sclerosis
DALLAS – Nintedanib, a tyrosine kinase inhibitor, decreased by 44% the annual rate of lung function decline among patients with interstitial lung disease associated with systemic sclerosis, a year-long study has found.
In a placebo-controlled 52-week trial, forced vital capacity (FVC) in patients who took nintedanib (Ofev) declined by a mean of 52 mL – significantly less than the mean 93 mL decline seen among those who were given placebo, Oliver Distler, MD, said at the annual meeting of the American Thoracic Society.
“These are people in their mid-40s and -50s,” said Dr. Distler of the University of Zürich. “They have a long time to go. If there is an annual preservation of lung function by 40%, if you have that every year, it becomes very surely clinically significant. A decline in FVC is also a good surrogate marker of mortality in interstitial lung disease associated with systemic sclerosis. Assuming the effects are ongoing above the 1 year we looked at, then indeed these results are clinically important.”
The study was simultaneously published in the New England Journal of Medicine. Nintedanib is already approved for idiopathic pulmonary fibrosis. But some data suggest that it also exerts antifibrotic and anti-inflammatory effects in animal models of systemic sclerosis and inflammatory lung disease (ILD). SENSCIS (the Safety and Efficacy of Nintedanib in Systemic Sclerosis trial) investigated the molecule’s use in patients with ILD associated with systemic sclerosis.
Conducted in 32 countries, SENSCIS comprised 576 patients with the disorder, whose sclerosis affected at least 10% of their lungs. They were assigned to 52 weeks of either placebo or 150 mg nintedanib twice weekly. However, patients stayed on their blinded treatment until the last patient enrolled had finished the year of treatment; some patients took the drug for 100 weeks, Dr. Distler said. The primary endpoint was annual rate of decline in the forced vital capacity (FEV). Secondary endpoints included changes of the modified Rodnan skin score and in the total score on the St. George’s Respiratory Questionnaire.
Patients were a mean of 54 years old, with a mean disease duration of about 3 years. About half had diffuse cutaneous systemic sclerosis; the sclerosis was limited in the remainder. The mean extent of lung fibrosis was about 36%. Half were taking mycophenolate at baseline, which was allowed as background treatment, along with up to 10 mg/day of prednisone. Any patient who experienced clinically significant lung function deterioration could receive additional therapy at the investigator’s discretion.
The mean baseline FEV for these patients was 72.5% of predicted value. The mean diffusing capacity of the lungs for carbon monoxide was 53% of expected capacity.
Most patients completed the study (80% of the active group and 89% of the placebo group). The mean drug exposure duration was 10 months in the active group and 11 in the placebo group.
Improvement began early in treatment, with the efficacy curves separating by week 12 and continuing to diverge. After 52 weeks of therapy, the annual rate of change was 41 mL less in the active group than in the placebo group (–54.4 mL vs. –93.3 mL). The mean adjusted absolute change from baseline was –54.6 mL in the active group and –101 mL in the placebo at week 52. Significantly fewer patients taking nintedanib also lost more than 10% of FVC by week 52 (16.7% vs. 18%).
The St. George’s Respiratory Questionnaire score improved about one point in the active group and declined about one point in the placebo group.
Nintedanib was equally effective across a number of subgroups, including those divided by sex, age, and race. Antitopoisomerase antibodies and so-called antitopoisomerase I antibody status did not affect nintedanib’s action. Nintedanib also significantly improved scores on the Health Assessment Questionnaire without Disability Index and dyspnea.
More patients in the active group than in on placebo discontinued treatment because of a serious adverse event (16% vs. 8.7%). The most common of these were diarrhea (75.7% vs. 31%), nausea (31.6% vs. 13.5%), and vomiting (24.7% vs.10.4%). Skin ulcers occurred in about 18% of each group. Patients in the active group were significantly more likely to develop elevated alanine and aspartate aminotransferase of up to three times normal levels (4.9% vs. 0.7%).
Treatment did not significantly affect mortality rates, however. Over the treatment period, 10 patients in the nintedanib group and 9 in the placebo group died (3.5% vs. 3.1%).
The study was sponsored by Boehringer Ingelheim. Dr. Distler was the primary investigator on the trial.
SOURCE: Distler O et al. ATS 2019, Abstract A7360.
DALLAS – Nintedanib, a tyrosine kinase inhibitor, decreased by 44% the annual rate of lung function decline among patients with interstitial lung disease associated with systemic sclerosis, a year-long study has found.
In a placebo-controlled 52-week trial, forced vital capacity (FVC) in patients who took nintedanib (Ofev) declined by a mean of 52 mL – significantly less than the mean 93 mL decline seen among those who were given placebo, Oliver Distler, MD, said at the annual meeting of the American Thoracic Society.
“These are people in their mid-40s and -50s,” said Dr. Distler of the University of Zürich. “They have a long time to go. If there is an annual preservation of lung function by 40%, if you have that every year, it becomes very surely clinically significant. A decline in FVC is also a good surrogate marker of mortality in interstitial lung disease associated with systemic sclerosis. Assuming the effects are ongoing above the 1 year we looked at, then indeed these results are clinically important.”
The study was simultaneously published in the New England Journal of Medicine. Nintedanib is already approved for idiopathic pulmonary fibrosis. But some data suggest that it also exerts antifibrotic and anti-inflammatory effects in animal models of systemic sclerosis and inflammatory lung disease (ILD). SENSCIS (the Safety and Efficacy of Nintedanib in Systemic Sclerosis trial) investigated the molecule’s use in patients with ILD associated with systemic sclerosis.
Conducted in 32 countries, SENSCIS comprised 576 patients with the disorder, whose sclerosis affected at least 10% of their lungs. They were assigned to 52 weeks of either placebo or 150 mg nintedanib twice weekly. However, patients stayed on their blinded treatment until the last patient enrolled had finished the year of treatment; some patients took the drug for 100 weeks, Dr. Distler said. The primary endpoint was annual rate of decline in the forced vital capacity (FEV). Secondary endpoints included changes of the modified Rodnan skin score and in the total score on the St. George’s Respiratory Questionnaire.
Patients were a mean of 54 years old, with a mean disease duration of about 3 years. About half had diffuse cutaneous systemic sclerosis; the sclerosis was limited in the remainder. The mean extent of lung fibrosis was about 36%. Half were taking mycophenolate at baseline, which was allowed as background treatment, along with up to 10 mg/day of prednisone. Any patient who experienced clinically significant lung function deterioration could receive additional therapy at the investigator’s discretion.
The mean baseline FEV for these patients was 72.5% of predicted value. The mean diffusing capacity of the lungs for carbon monoxide was 53% of expected capacity.
Most patients completed the study (80% of the active group and 89% of the placebo group). The mean drug exposure duration was 10 months in the active group and 11 in the placebo group.
Improvement began early in treatment, with the efficacy curves separating by week 12 and continuing to diverge. After 52 weeks of therapy, the annual rate of change was 41 mL less in the active group than in the placebo group (–54.4 mL vs. –93.3 mL). The mean adjusted absolute change from baseline was –54.6 mL in the active group and –101 mL in the placebo at week 52. Significantly fewer patients taking nintedanib also lost more than 10% of FVC by week 52 (16.7% vs. 18%).
The St. George’s Respiratory Questionnaire score improved about one point in the active group and declined about one point in the placebo group.
Nintedanib was equally effective across a number of subgroups, including those divided by sex, age, and race. Antitopoisomerase antibodies and so-called antitopoisomerase I antibody status did not affect nintedanib’s action. Nintedanib also significantly improved scores on the Health Assessment Questionnaire without Disability Index and dyspnea.
More patients in the active group than in on placebo discontinued treatment because of a serious adverse event (16% vs. 8.7%). The most common of these were diarrhea (75.7% vs. 31%), nausea (31.6% vs. 13.5%), and vomiting (24.7% vs.10.4%). Skin ulcers occurred in about 18% of each group. Patients in the active group were significantly more likely to develop elevated alanine and aspartate aminotransferase of up to three times normal levels (4.9% vs. 0.7%).
Treatment did not significantly affect mortality rates, however. Over the treatment period, 10 patients in the nintedanib group and 9 in the placebo group died (3.5% vs. 3.1%).
The study was sponsored by Boehringer Ingelheim. Dr. Distler was the primary investigator on the trial.
SOURCE: Distler O et al. ATS 2019, Abstract A7360.
DALLAS – Nintedanib, a tyrosine kinase inhibitor, decreased by 44% the annual rate of lung function decline among patients with interstitial lung disease associated with systemic sclerosis, a year-long study has found.
In a placebo-controlled 52-week trial, forced vital capacity (FVC) in patients who took nintedanib (Ofev) declined by a mean of 52 mL – significantly less than the mean 93 mL decline seen among those who were given placebo, Oliver Distler, MD, said at the annual meeting of the American Thoracic Society.
“These are people in their mid-40s and -50s,” said Dr. Distler of the University of Zürich. “They have a long time to go. If there is an annual preservation of lung function by 40%, if you have that every year, it becomes very surely clinically significant. A decline in FVC is also a good surrogate marker of mortality in interstitial lung disease associated with systemic sclerosis. Assuming the effects are ongoing above the 1 year we looked at, then indeed these results are clinically important.”
The study was simultaneously published in the New England Journal of Medicine. Nintedanib is already approved for idiopathic pulmonary fibrosis. But some data suggest that it also exerts antifibrotic and anti-inflammatory effects in animal models of systemic sclerosis and inflammatory lung disease (ILD). SENSCIS (the Safety and Efficacy of Nintedanib in Systemic Sclerosis trial) investigated the molecule’s use in patients with ILD associated with systemic sclerosis.
Conducted in 32 countries, SENSCIS comprised 576 patients with the disorder, whose sclerosis affected at least 10% of their lungs. They were assigned to 52 weeks of either placebo or 150 mg nintedanib twice weekly. However, patients stayed on their blinded treatment until the last patient enrolled had finished the year of treatment; some patients took the drug for 100 weeks, Dr. Distler said. The primary endpoint was annual rate of decline in the forced vital capacity (FEV). Secondary endpoints included changes of the modified Rodnan skin score and in the total score on the St. George’s Respiratory Questionnaire.
Patients were a mean of 54 years old, with a mean disease duration of about 3 years. About half had diffuse cutaneous systemic sclerosis; the sclerosis was limited in the remainder. The mean extent of lung fibrosis was about 36%. Half were taking mycophenolate at baseline, which was allowed as background treatment, along with up to 10 mg/day of prednisone. Any patient who experienced clinically significant lung function deterioration could receive additional therapy at the investigator’s discretion.
The mean baseline FEV for these patients was 72.5% of predicted value. The mean diffusing capacity of the lungs for carbon monoxide was 53% of expected capacity.
Most patients completed the study (80% of the active group and 89% of the placebo group). The mean drug exposure duration was 10 months in the active group and 11 in the placebo group.
Improvement began early in treatment, with the efficacy curves separating by week 12 and continuing to diverge. After 52 weeks of therapy, the annual rate of change was 41 mL less in the active group than in the placebo group (–54.4 mL vs. –93.3 mL). The mean adjusted absolute change from baseline was –54.6 mL in the active group and –101 mL in the placebo at week 52. Significantly fewer patients taking nintedanib also lost more than 10% of FVC by week 52 (16.7% vs. 18%).
The St. George’s Respiratory Questionnaire score improved about one point in the active group and declined about one point in the placebo group.
Nintedanib was equally effective across a number of subgroups, including those divided by sex, age, and race. Antitopoisomerase antibodies and so-called antitopoisomerase I antibody status did not affect nintedanib’s action. Nintedanib also significantly improved scores on the Health Assessment Questionnaire without Disability Index and dyspnea.
More patients in the active group than in on placebo discontinued treatment because of a serious adverse event (16% vs. 8.7%). The most common of these were diarrhea (75.7% vs. 31%), nausea (31.6% vs. 13.5%), and vomiting (24.7% vs.10.4%). Skin ulcers occurred in about 18% of each group. Patients in the active group were significantly more likely to develop elevated alanine and aspartate aminotransferase of up to three times normal levels (4.9% vs. 0.7%).
Treatment did not significantly affect mortality rates, however. Over the treatment period, 10 patients in the nintedanib group and 9 in the placebo group died (3.5% vs. 3.1%).
The study was sponsored by Boehringer Ingelheim. Dr. Distler was the primary investigator on the trial.
SOURCE: Distler O et al. ATS 2019, Abstract A7360.
AT ATS 2019
Key clinical point: The tyrosine kinase inhibitor nintedanib may be a useful treatment for interstitial lung disease associated with systemic sclerosis (SS-ILD).
Major finding: Nintedanib decreased the annual rate of lung function decline by 44% among patients with SS-ILD.
Study details: The randomized, placebo-controlled study comprised 576 patients.
Disclosures: The trial was sponsored by Boehringer Ingelheim. Dr. Distler is the primary investigator.
Source: Distler O et al. ATS 2019, Abstract A7360.
U.S. measles total sees smallest increase in 2 months
according to the Centers for Disease Control and Prevention.
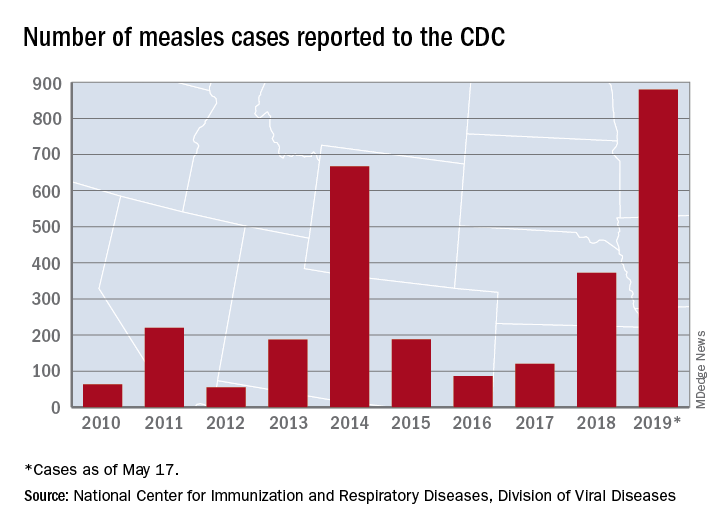
That weekly increase of 41 cases is the smallest since the week ending March 14, when the total rose by 40. The largest 1-week rise of the year came during the week ending April 11, when there were 90 new cases, CDC data show.
A case that has been reported by the media in the last week but not officially through the CDC would make New Mexico the 25th state with a measles case this year. The state’s health department has confirmed measles in a 1-year-old from Sierra County, which is New Mexico’s first case since 2014, the Las Cruces Sun News reported, adding that 4,441 school-aged children had an exemption for vaccination filed with the state in 2018.
Making a return appearance to the CDC’s list of outbreaks is Washington State, which reported six new cases last week in three Puget Sound counties (King, Pierce, and Snohomish). The most likely location and date of exposure was at Seattle-Tacoma International Airport on April 25, the Washington State Department of Health said. In February and March, there were 71 cases in Clark County on the state’s border with Oregon.
The ongoing outbreak in Michigan had been quiet since April, but the state’s Department of Health and Human Services confirmed a measles case in St. Clair County on May 17, bringing the total to 44 for the year. The new case, which is not related to an earlier outbreak that occurred mainly in Oakland County, involves an international traveler visiting Michigan.
according to the Centers for Disease Control and Prevention.

That weekly increase of 41 cases is the smallest since the week ending March 14, when the total rose by 40. The largest 1-week rise of the year came during the week ending April 11, when there were 90 new cases, CDC data show.
A case that has been reported by the media in the last week but not officially through the CDC would make New Mexico the 25th state with a measles case this year. The state’s health department has confirmed measles in a 1-year-old from Sierra County, which is New Mexico’s first case since 2014, the Las Cruces Sun News reported, adding that 4,441 school-aged children had an exemption for vaccination filed with the state in 2018.
Making a return appearance to the CDC’s list of outbreaks is Washington State, which reported six new cases last week in three Puget Sound counties (King, Pierce, and Snohomish). The most likely location and date of exposure was at Seattle-Tacoma International Airport on April 25, the Washington State Department of Health said. In February and March, there were 71 cases in Clark County on the state’s border with Oregon.
The ongoing outbreak in Michigan had been quiet since April, but the state’s Department of Health and Human Services confirmed a measles case in St. Clair County on May 17, bringing the total to 44 for the year. The new case, which is not related to an earlier outbreak that occurred mainly in Oakland County, involves an international traveler visiting Michigan.
according to the Centers for Disease Control and Prevention.

That weekly increase of 41 cases is the smallest since the week ending March 14, when the total rose by 40. The largest 1-week rise of the year came during the week ending April 11, when there were 90 new cases, CDC data show.
A case that has been reported by the media in the last week but not officially through the CDC would make New Mexico the 25th state with a measles case this year. The state’s health department has confirmed measles in a 1-year-old from Sierra County, which is New Mexico’s first case since 2014, the Las Cruces Sun News reported, adding that 4,441 school-aged children had an exemption for vaccination filed with the state in 2018.
Making a return appearance to the CDC’s list of outbreaks is Washington State, which reported six new cases last week in three Puget Sound counties (King, Pierce, and Snohomish). The most likely location and date of exposure was at Seattle-Tacoma International Airport on April 25, the Washington State Department of Health said. In February and March, there were 71 cases in Clark County on the state’s border with Oregon.
The ongoing outbreak in Michigan had been quiet since April, but the state’s Department of Health and Human Services confirmed a measles case in St. Clair County on May 17, bringing the total to 44 for the year. The new case, which is not related to an earlier outbreak that occurred mainly in Oakland County, involves an international traveler visiting Michigan.
AFib on the rise in end-stage COPD patients hospitalized for exacerbations
Atrial fibrillation is being seen with increasing frequency in patients admitted to U.S. hospitals for exacerbations of end-stage chronic obstructive pulmonary disease, based on a retrospective analysis of data from the Nationwide Inpatient Sample.
The prevalence of atrial fibrillation (AFib) among patients with end-stage chronic obstructive pulmonary disease (COPD) on home oxygen who were admitted with COPD exacerbations increased from 12.9% in 2003 to 21.3% in 2014, according to Xiaochun Xiao of the department of health statistics at Second Military Medical University in Shanghai and colleagues.
Additionally, “we found that comorbid [AFib] was associated with an increased risk of the need for mechanical ventilation, especially invasive mechanical ventilation. Moreover, comorbid [AFib] was associated with adverse clinical outcomes, including increased in-hospital death, acute respiratory failure, acute kidney injury, sepsis, and stroke,” the researchers wrote in the study published in the journal CHEST.
Patients included in the study were aged at least 18 years, were diagnosed with end-stage COPD and on home oxygen, and were hospitalized because of a COPD-related exacerbation. Based on 1,345,270 weighted hospital admissions of adults with end-stage COPD on home oxygen who met the inclusion criteria for the study, 18.2% (244,488 admissions) of patients had AFib, and the prevalence of AFib in COPD patients increased over time from 2003 (12.9%) to 2014 (21.3%; P less than .0001).
Patients with AFib, compared with patients without AFib, were older (75.5 years vs. 69.6 years; P less than .0001) and more likely to be male (50.7% vs. 59.1%; P less than .0001) and white (80.9% vs. 74.4%; P less than .0001). Patients with AFib also had higher stroke risk reflected in higher CHA2DS2-VASc scores (3.26 vs. 2.45; P less than .0001), and higher likelihood of in-hospital mortality and readmission reflected in Elixhauser scores greater than or equal to 4 (51.2% vs. 35.6%).
In addition, the prevalence of AFib increased with increasing income. Larger hospitals in terms of bed size, urban environment, and Medicare insurance status also were associated with a higher AFib prevalence.
AFib was associated with an increased cost of $1,415 and an increased length of stay of 0.6 days after adjustment for potential confounders. AFib also predicted risk for several adverse events, including stroke (odds ratio, 1.80; in-hospital death, [OR, 1.54]), invasive mechanical ventilation (OR, 1.37), sepsis (OR, 1.23), noninvasive mechanical ventilation (OR, 1.14), acute kidney injury (OR, 1.09), and acute respiratory failure (OR, 1.09).
The researchers noted the database could have potentially overinflated AFib prevalence, as they could not differentiate index admissions and readmissions. The database also does not contain information about secondary diagnoses codes present on admission, which could make it difficult to identify adverse events that occurred during hospitalization.
“Our findings should prompt further efforts to identify the reasons for increased [AFib] prevalence and provide better management strategies for end-stage COPD patients comorbid with [AFib],” the researchers concluded.
This study was funded by a grant from the Fourth Round of the Shanghai 3-year Action Plan on Public Health Discipline and Talent Program. The authors reported no relevant conflict of interest.
SOURCE: Xiao X et al. CHEST. 2019 Jan 23. doi: 10.1016/j.chest.2018.12.021.
Atrial fibrillation is being seen with increasing frequency in patients admitted to U.S. hospitals for exacerbations of end-stage chronic obstructive pulmonary disease, based on a retrospective analysis of data from the Nationwide Inpatient Sample.
The prevalence of atrial fibrillation (AFib) among patients with end-stage chronic obstructive pulmonary disease (COPD) on home oxygen who were admitted with COPD exacerbations increased from 12.9% in 2003 to 21.3% in 2014, according to Xiaochun Xiao of the department of health statistics at Second Military Medical University in Shanghai and colleagues.
Additionally, “we found that comorbid [AFib] was associated with an increased risk of the need for mechanical ventilation, especially invasive mechanical ventilation. Moreover, comorbid [AFib] was associated with adverse clinical outcomes, including increased in-hospital death, acute respiratory failure, acute kidney injury, sepsis, and stroke,” the researchers wrote in the study published in the journal CHEST.
Patients included in the study were aged at least 18 years, were diagnosed with end-stage COPD and on home oxygen, and were hospitalized because of a COPD-related exacerbation. Based on 1,345,270 weighted hospital admissions of adults with end-stage COPD on home oxygen who met the inclusion criteria for the study, 18.2% (244,488 admissions) of patients had AFib, and the prevalence of AFib in COPD patients increased over time from 2003 (12.9%) to 2014 (21.3%; P less than .0001).
Patients with AFib, compared with patients without AFib, were older (75.5 years vs. 69.6 years; P less than .0001) and more likely to be male (50.7% vs. 59.1%; P less than .0001) and white (80.9% vs. 74.4%; P less than .0001). Patients with AFib also had higher stroke risk reflected in higher CHA2DS2-VASc scores (3.26 vs. 2.45; P less than .0001), and higher likelihood of in-hospital mortality and readmission reflected in Elixhauser scores greater than or equal to 4 (51.2% vs. 35.6%).
In addition, the prevalence of AFib increased with increasing income. Larger hospitals in terms of bed size, urban environment, and Medicare insurance status also were associated with a higher AFib prevalence.
AFib was associated with an increased cost of $1,415 and an increased length of stay of 0.6 days after adjustment for potential confounders. AFib also predicted risk for several adverse events, including stroke (odds ratio, 1.80; in-hospital death, [OR, 1.54]), invasive mechanical ventilation (OR, 1.37), sepsis (OR, 1.23), noninvasive mechanical ventilation (OR, 1.14), acute kidney injury (OR, 1.09), and acute respiratory failure (OR, 1.09).
The researchers noted the database could have potentially overinflated AFib prevalence, as they could not differentiate index admissions and readmissions. The database also does not contain information about secondary diagnoses codes present on admission, which could make it difficult to identify adverse events that occurred during hospitalization.
“Our findings should prompt further efforts to identify the reasons for increased [AFib] prevalence and provide better management strategies for end-stage COPD patients comorbid with [AFib],” the researchers concluded.
This study was funded by a grant from the Fourth Round of the Shanghai 3-year Action Plan on Public Health Discipline and Talent Program. The authors reported no relevant conflict of interest.
SOURCE: Xiao X et al. CHEST. 2019 Jan 23. doi: 10.1016/j.chest.2018.12.021.
Atrial fibrillation is being seen with increasing frequency in patients admitted to U.S. hospitals for exacerbations of end-stage chronic obstructive pulmonary disease, based on a retrospective analysis of data from the Nationwide Inpatient Sample.
The prevalence of atrial fibrillation (AFib) among patients with end-stage chronic obstructive pulmonary disease (COPD) on home oxygen who were admitted with COPD exacerbations increased from 12.9% in 2003 to 21.3% in 2014, according to Xiaochun Xiao of the department of health statistics at Second Military Medical University in Shanghai and colleagues.
Additionally, “we found that comorbid [AFib] was associated with an increased risk of the need for mechanical ventilation, especially invasive mechanical ventilation. Moreover, comorbid [AFib] was associated with adverse clinical outcomes, including increased in-hospital death, acute respiratory failure, acute kidney injury, sepsis, and stroke,” the researchers wrote in the study published in the journal CHEST.
Patients included in the study were aged at least 18 years, were diagnosed with end-stage COPD and on home oxygen, and were hospitalized because of a COPD-related exacerbation. Based on 1,345,270 weighted hospital admissions of adults with end-stage COPD on home oxygen who met the inclusion criteria for the study, 18.2% (244,488 admissions) of patients had AFib, and the prevalence of AFib in COPD patients increased over time from 2003 (12.9%) to 2014 (21.3%; P less than .0001).
Patients with AFib, compared with patients without AFib, were older (75.5 years vs. 69.6 years; P less than .0001) and more likely to be male (50.7% vs. 59.1%; P less than .0001) and white (80.9% vs. 74.4%; P less than .0001). Patients with AFib also had higher stroke risk reflected in higher CHA2DS2-VASc scores (3.26 vs. 2.45; P less than .0001), and higher likelihood of in-hospital mortality and readmission reflected in Elixhauser scores greater than or equal to 4 (51.2% vs. 35.6%).
In addition, the prevalence of AFib increased with increasing income. Larger hospitals in terms of bed size, urban environment, and Medicare insurance status also were associated with a higher AFib prevalence.
AFib was associated with an increased cost of $1,415 and an increased length of stay of 0.6 days after adjustment for potential confounders. AFib also predicted risk for several adverse events, including stroke (odds ratio, 1.80; in-hospital death, [OR, 1.54]), invasive mechanical ventilation (OR, 1.37), sepsis (OR, 1.23), noninvasive mechanical ventilation (OR, 1.14), acute kidney injury (OR, 1.09), and acute respiratory failure (OR, 1.09).
The researchers noted the database could have potentially overinflated AFib prevalence, as they could not differentiate index admissions and readmissions. The database also does not contain information about secondary diagnoses codes present on admission, which could make it difficult to identify adverse events that occurred during hospitalization.
“Our findings should prompt further efforts to identify the reasons for increased [AFib] prevalence and provide better management strategies for end-stage COPD patients comorbid with [AFib],” the researchers concluded.
This study was funded by a grant from the Fourth Round of the Shanghai 3-year Action Plan on Public Health Discipline and Talent Program. The authors reported no relevant conflict of interest.
SOURCE: Xiao X et al. CHEST. 2019 Jan 23. doi: 10.1016/j.chest.2018.12.021.
FROM CHEST
Key clinical point: Comorbid atrial fibrillation was associated with an increased risk of the need for mechanical ventilation, especially invasive mechanical ventilation, and of adverse outcomes including in-hospital death, acute respiratory failure, acute kidney injury, sepsis, and stroke.
Major finding: The prevalence of atrial fibrillation with end-stage chronic obstructive pulmonary disease increased over time from 2003 (12.9%) to 2014 (21.3%). Study details: A retrospective analysis based on 1,345,270 weighted hospital admissions of adults with end-stage chronic obstructive pulmonary disease on home oxygen from the Nationwide Impatient Sample during 2003-2014.
Disclosures: The study was funded by a grant from the Fourth Round of the Shanghai 3-Year Action Plan on Public Health Discipline and Talent Program. The authors reported no conflicts of interest.
Source: Xiao X et al. CHEST. 2019 Jan 23. doi: 10.1016/j.chest.2018.12.021.
Smoking rates remain steady among the poor
While an increasing number of U.S. citizens are saying no to cigarettes, a recent study shows.
The odds of current smoking, versus never smoking, declined significantly during 2008-2017 for individuals with none of six disadvantages tied to cigarette use, including disability, unemployment, poverty, low education, psychological distress, and heavy alcohol intake, according to researchers.
Individuals with one or two of those disadvantages have also been cutting back, the data suggest. But, by contrast, odds of current versus never smoking did not significantly change for those with three or more disadvantages, according to Adam M. Leventhal, PhD, of the University of Southern California, Los Angeles, and coinvestigators.
“How this pattern can inform a cohesive policy agenda is unknown, but it is clear from these findings that the crux of the recently expanding tobacco-related health disparity problem in the United States is not tied to groups facing merely a single form of disadvantage,” Dr. Leventhal and coauthors wrote in a report on the study in JAMA Internal Medicine.
The cross-sectional analysis by Dr. Leventhal and colleagues was based on National Health Interview Survey (NHIS) data from 2008-2017 including more than 278,000 respondents aged 25 years or older.
A snapshot of that 10-year period showed that current smoking prevalence was successively higher depending on the number of socioeconomic or health-related disadvantages.
The mean prevalence of current smoking over that entire time period was just 13.8% for people with zero of the six disadvantages, 21.4% for those with one disadvantage, and so on, up to 58.2% for those with all six disadvantages, according to data in the published report.
Encouragingly, overall smoking prevalence fell from 20.8% in 2008-2009 to 15.8% in 2016-2017, the researchers found. However, the decreasing trend was not apparent for individuals with many disadvantages.
The odds ratio for change in odds of smoking per year was 0.951 (95% confidence interval, 0.944-0.958) for those with zero disadvantages, 0.96 (95% CI, 0.95-0.97) for one disadvantage, and 0.98 (95% CI, 0.97-0.99) for two, all representing significant annual reductions in current versus never smoking, investigators said. By contrast, no such significant changes were apparent for those with three, four, five, or six such disadvantages.
Tobacco control or regulatory policies that consider these disadvantages separately may be overlooking a “broader pattern” showing that the cumulative number of disadvantages correlates with the magnitude of disparity, wrote Dr. Leventhal and colleagues in their report.
“Successful prevention of smoking initiation and promotion of smoking cessation in multi-disadvantaged populations would substantially reduce the smoking-related public health burden in the United States,” they concluded.
Dr. Leventhal and colleagues reported no conflicts related to their research, which was supported in part by a Tobacco Centers of Regulatory Science award from the National Cancer Institute and the Food and Drug Administration, among other sources.
SOURCE: Leventhal AM et al. JAMA Intern Med. 2019 Apr 22. doi: 10.1001/jamainternmed.2019.0192.
While an increasing number of U.S. citizens are saying no to cigarettes, a recent study shows.
The odds of current smoking, versus never smoking, declined significantly during 2008-2017 for individuals with none of six disadvantages tied to cigarette use, including disability, unemployment, poverty, low education, psychological distress, and heavy alcohol intake, according to researchers.
Individuals with one or two of those disadvantages have also been cutting back, the data suggest. But, by contrast, odds of current versus never smoking did not significantly change for those with three or more disadvantages, according to Adam M. Leventhal, PhD, of the University of Southern California, Los Angeles, and coinvestigators.
“How this pattern can inform a cohesive policy agenda is unknown, but it is clear from these findings that the crux of the recently expanding tobacco-related health disparity problem in the United States is not tied to groups facing merely a single form of disadvantage,” Dr. Leventhal and coauthors wrote in a report on the study in JAMA Internal Medicine.
The cross-sectional analysis by Dr. Leventhal and colleagues was based on National Health Interview Survey (NHIS) data from 2008-2017 including more than 278,000 respondents aged 25 years or older.
A snapshot of that 10-year period showed that current smoking prevalence was successively higher depending on the number of socioeconomic or health-related disadvantages.
The mean prevalence of current smoking over that entire time period was just 13.8% for people with zero of the six disadvantages, 21.4% for those with one disadvantage, and so on, up to 58.2% for those with all six disadvantages, according to data in the published report.
Encouragingly, overall smoking prevalence fell from 20.8% in 2008-2009 to 15.8% in 2016-2017, the researchers found. However, the decreasing trend was not apparent for individuals with many disadvantages.
The odds ratio for change in odds of smoking per year was 0.951 (95% confidence interval, 0.944-0.958) for those with zero disadvantages, 0.96 (95% CI, 0.95-0.97) for one disadvantage, and 0.98 (95% CI, 0.97-0.99) for two, all representing significant annual reductions in current versus never smoking, investigators said. By contrast, no such significant changes were apparent for those with three, four, five, or six such disadvantages.
Tobacco control or regulatory policies that consider these disadvantages separately may be overlooking a “broader pattern” showing that the cumulative number of disadvantages correlates with the magnitude of disparity, wrote Dr. Leventhal and colleagues in their report.
“Successful prevention of smoking initiation and promotion of smoking cessation in multi-disadvantaged populations would substantially reduce the smoking-related public health burden in the United States,” they concluded.
Dr. Leventhal and colleagues reported no conflicts related to their research, which was supported in part by a Tobacco Centers of Regulatory Science award from the National Cancer Institute and the Food and Drug Administration, among other sources.
SOURCE: Leventhal AM et al. JAMA Intern Med. 2019 Apr 22. doi: 10.1001/jamainternmed.2019.0192.
While an increasing number of U.S. citizens are saying no to cigarettes, a recent study shows.
The odds of current smoking, versus never smoking, declined significantly during 2008-2017 for individuals with none of six disadvantages tied to cigarette use, including disability, unemployment, poverty, low education, psychological distress, and heavy alcohol intake, according to researchers.
Individuals with one or two of those disadvantages have also been cutting back, the data suggest. But, by contrast, odds of current versus never smoking did not significantly change for those with three or more disadvantages, according to Adam M. Leventhal, PhD, of the University of Southern California, Los Angeles, and coinvestigators.
“How this pattern can inform a cohesive policy agenda is unknown, but it is clear from these findings that the crux of the recently expanding tobacco-related health disparity problem in the United States is not tied to groups facing merely a single form of disadvantage,” Dr. Leventhal and coauthors wrote in a report on the study in JAMA Internal Medicine.
The cross-sectional analysis by Dr. Leventhal and colleagues was based on National Health Interview Survey (NHIS) data from 2008-2017 including more than 278,000 respondents aged 25 years or older.
A snapshot of that 10-year period showed that current smoking prevalence was successively higher depending on the number of socioeconomic or health-related disadvantages.
The mean prevalence of current smoking over that entire time period was just 13.8% for people with zero of the six disadvantages, 21.4% for those with one disadvantage, and so on, up to 58.2% for those with all six disadvantages, according to data in the published report.
Encouragingly, overall smoking prevalence fell from 20.8% in 2008-2009 to 15.8% in 2016-2017, the researchers found. However, the decreasing trend was not apparent for individuals with many disadvantages.
The odds ratio for change in odds of smoking per year was 0.951 (95% confidence interval, 0.944-0.958) for those with zero disadvantages, 0.96 (95% CI, 0.95-0.97) for one disadvantage, and 0.98 (95% CI, 0.97-0.99) for two, all representing significant annual reductions in current versus never smoking, investigators said. By contrast, no such significant changes were apparent for those with three, four, five, or six such disadvantages.
Tobacco control or regulatory policies that consider these disadvantages separately may be overlooking a “broader pattern” showing that the cumulative number of disadvantages correlates with the magnitude of disparity, wrote Dr. Leventhal and colleagues in their report.
“Successful prevention of smoking initiation and promotion of smoking cessation in multi-disadvantaged populations would substantially reduce the smoking-related public health burden in the United States,” they concluded.
Dr. Leventhal and colleagues reported no conflicts related to their research, which was supported in part by a Tobacco Centers of Regulatory Science award from the National Cancer Institute and the Food and Drug Administration, among other sources.
SOURCE: Leventhal AM et al. JAMA Intern Med. 2019 Apr 22. doi: 10.1001/jamainternmed.2019.0192.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Current U.S. smoking rates have not declined among individuals with multiple socioeconomic or health-related disadvantages.
Major finding: The odds ratio for change in odds of smoking per year was 0.951 for individuals with zero disadvantages, 0.96 for one disadvantage, and 0.97-0.99 for two, with no significant annual reductions in those with three or more disadvantages.
Study details: Cross-sectional analysis of 278,048 respondents aged 25 years or older in the National Health Interview Survey during 2008-2017.
Disclosures: Authors reported no conflicts of interest related to the study, which was supported in part by a Tobacco Centers of Regulatory Science award from the National Cancer Institute and the Food and Drug Administration, among other sources.
Source: Leventhal AM et al. JAMA Intern Med. 2019 Apr 22. doi: 10.1001/jamainternmed.2019.0192.
Methotrexate does not cause rheumatoid interstitial lung disease
BIRMINGHAM, ENGLAND – Data from two early RA inception cohorts provide reassurance that methotrexate does not cause interstitial lung disease and suggest that treatment with methotrexate might even be protective.
In the Early RA Study (ERAS) and Early RA Network (ERAN), which together include 2,701 patients with RA, 101 (3.7%) had interstitial lung disease (ILD). There were 92 patients with RA-ILD who had information available on exposure to any conventional synthetic disease-modifying antirheumatic drug (csDMARD); of these, 39 (2.5%) had been exposed to methotrexate (n = 1,578) and 53 (4.8%) to other csDMARDs (n = 1,114).
Multivariate analysis showed that methotrexate exposure was associated with a reduced risk of developing ILD, with an odds ratio of 0.48 (P = .004). In a separate analysis that excluded 25 patients who had ILD before they received any csDMARD therapy (n = 67), there was no association between methotrexate use and ILD (OR, 0.85; P = .578). In fact, there was a nonsignificant trend for a delayed onset of ILD in patients who had been treated with methotrexate (OR, 0.54; P = .072).
Methotrexate use is associated with an acute hypersensitivity pneumonitis in patients with RA, explained Patrick Kiely, MBBS, PhD, of St. George’s University Hospitals NHS Foundation Trust in London at the British Society for Rheumatology annual conference. “This is well recognized, it’s very rare [0.43%-1.00%], it’s easy to spot, and usually goes away if you stop methotrexate,” said Dr. Kiely, adding that “it’s not benign, and severe cases can be life threatening.”
Because of the association between methotrexate and pneumonitis, there has been concern that methotrexate may exacerbate or even cause ILD in RA but there are sparse data available to confirm this. The bottom line is that you should not start someone on methotrexate if you think their existing lung capacity is not up to treatment with methotrexate, Dr. Kiely said.
ILD is not always symptomatic in RA, but when it is, it is associated with very poor survival. The lung disease can be present before joint symptoms, Dr. Kiely said. Although less than 10% of cases may be symptomatic, this “is a big deal, because it has a high mortality, with death within 5 years. It’s the second-commonest cause of excess mortality in RA after cardiovascular disease.”
To look at the association between incident RA-ILD and the use of methotrexate, Dr. Kiely and associates analyzed data from ERAS (1986-2001) and ERAN (2002-2013), that together have more than 25 years of follow-up data on patients who were recruited at the first sign of RA symptoms. Patients within these cohorts have been treated according to best practice, and a range of outcomes – including RA-ILD – have been assessed at annual intervals.
In the patients who developed ILD after any csDMARD exposure, older age at RA onset (OR, 1.04; P less than .001) and having ever smoked (OR, 1.91; P = .016) were associated with the development of the lung disease. Incident ILD was also associated with being positive for rheumatoid factor (OR, 2.02; P = .029) at baseline. Being male was also associated with a higher risk for developing ILD, Dr. Kiely reported, as was a longer duration of time between the onset of first RA symptoms and the first secondary care visit. Conversely, the presence of nonrespiratory, major comorbidities at baseline appeared to be protective (OR, 0.62; P = .027).
“We found no association between methotrexate treatment and incident RA-ILD and a possibility that it may be protective,” Dr. Kiely concluded, noting that these data were now published in BMJ Open (2019;9:e028466. doi: 10.1136/bmjopen-2018-028466).
Following Dr. Kiely’s presentation, an audience member asked if the protective effect seen with methotrexate could have been caused by better disease control overall.
Dr. Kiely answered that, up until 2001, the time when ERAS was ongoing, standard practice in the United Kingdom was to use sulfasalazine, but then methotrexate started to be used in higher and higher doses, as seen in ERAN.
The interesting thing is that in ERAN more methotrexate was used in higher doses, but less RA-ILD was seen, Dr. Kiely observed. The overall prevalence of RA-ILD in the later early RA cohort was 3.2% and the median dose of methotrexate used was 20 mg. In ERAS, the prevalence was 4.2% and the median dose of methotrexate used was 10 mg.
There was a suggestion that disease control was slightly better in ERAN than ERAS, but that wasn’t statistically significant, Dr. Kiely said.
So, should a patient with RA and ILD be given methotrexate? There’s no reason not to, Dr. Kiely suggested, based on the evidence shown. Part of the challenge will now be convincing chest physician colleagues that methotrexate is not problematic in terms of causing ILD.
These findings are completely on board with the ILD group’s findings that methotrexate doesn’t cause pulmonary fibrosis in patients with RA, commented Julie Dawson, MD, of St. Helens and Knowsley Teaching Hospitals NHS Trust, St. Helens, England. Her own research, which includes a 10-year follow-up of patients with inflammatory arthritis, has shown that methotrexate does not appear to increase the risk of pulmonary fibrosis.
The study had no specific outside funding. Dr. Kiely reported having no conflicts of interest.
SOURCE: Kiely P et al. Rheumatology. 2019;58(suppl 3), Abstract 009.
BIRMINGHAM, ENGLAND – Data from two early RA inception cohorts provide reassurance that methotrexate does not cause interstitial lung disease and suggest that treatment with methotrexate might even be protective.
In the Early RA Study (ERAS) and Early RA Network (ERAN), which together include 2,701 patients with RA, 101 (3.7%) had interstitial lung disease (ILD). There were 92 patients with RA-ILD who had information available on exposure to any conventional synthetic disease-modifying antirheumatic drug (csDMARD); of these, 39 (2.5%) had been exposed to methotrexate (n = 1,578) and 53 (4.8%) to other csDMARDs (n = 1,114).
Multivariate analysis showed that methotrexate exposure was associated with a reduced risk of developing ILD, with an odds ratio of 0.48 (P = .004). In a separate analysis that excluded 25 patients who had ILD before they received any csDMARD therapy (n = 67), there was no association between methotrexate use and ILD (OR, 0.85; P = .578). In fact, there was a nonsignificant trend for a delayed onset of ILD in patients who had been treated with methotrexate (OR, 0.54; P = .072).
Methotrexate use is associated with an acute hypersensitivity pneumonitis in patients with RA, explained Patrick Kiely, MBBS, PhD, of St. George’s University Hospitals NHS Foundation Trust in London at the British Society for Rheumatology annual conference. “This is well recognized, it’s very rare [0.43%-1.00%], it’s easy to spot, and usually goes away if you stop methotrexate,” said Dr. Kiely, adding that “it’s not benign, and severe cases can be life threatening.”
Because of the association between methotrexate and pneumonitis, there has been concern that methotrexate may exacerbate or even cause ILD in RA but there are sparse data available to confirm this. The bottom line is that you should not start someone on methotrexate if you think their existing lung capacity is not up to treatment with methotrexate, Dr. Kiely said.
ILD is not always symptomatic in RA, but when it is, it is associated with very poor survival. The lung disease can be present before joint symptoms, Dr. Kiely said. Although less than 10% of cases may be symptomatic, this “is a big deal, because it has a high mortality, with death within 5 years. It’s the second-commonest cause of excess mortality in RA after cardiovascular disease.”
To look at the association between incident RA-ILD and the use of methotrexate, Dr. Kiely and associates analyzed data from ERAS (1986-2001) and ERAN (2002-2013), that together have more than 25 years of follow-up data on patients who were recruited at the first sign of RA symptoms. Patients within these cohorts have been treated according to best practice, and a range of outcomes – including RA-ILD – have been assessed at annual intervals.
In the patients who developed ILD after any csDMARD exposure, older age at RA onset (OR, 1.04; P less than .001) and having ever smoked (OR, 1.91; P = .016) were associated with the development of the lung disease. Incident ILD was also associated with being positive for rheumatoid factor (OR, 2.02; P = .029) at baseline. Being male was also associated with a higher risk for developing ILD, Dr. Kiely reported, as was a longer duration of time between the onset of first RA symptoms and the first secondary care visit. Conversely, the presence of nonrespiratory, major comorbidities at baseline appeared to be protective (OR, 0.62; P = .027).
“We found no association between methotrexate treatment and incident RA-ILD and a possibility that it may be protective,” Dr. Kiely concluded, noting that these data were now published in BMJ Open (2019;9:e028466. doi: 10.1136/bmjopen-2018-028466).
Following Dr. Kiely’s presentation, an audience member asked if the protective effect seen with methotrexate could have been caused by better disease control overall.
Dr. Kiely answered that, up until 2001, the time when ERAS was ongoing, standard practice in the United Kingdom was to use sulfasalazine, but then methotrexate started to be used in higher and higher doses, as seen in ERAN.
The interesting thing is that in ERAN more methotrexate was used in higher doses, but less RA-ILD was seen, Dr. Kiely observed. The overall prevalence of RA-ILD in the later early RA cohort was 3.2% and the median dose of methotrexate used was 20 mg. In ERAS, the prevalence was 4.2% and the median dose of methotrexate used was 10 mg.
There was a suggestion that disease control was slightly better in ERAN than ERAS, but that wasn’t statistically significant, Dr. Kiely said.
So, should a patient with RA and ILD be given methotrexate? There’s no reason not to, Dr. Kiely suggested, based on the evidence shown. Part of the challenge will now be convincing chest physician colleagues that methotrexate is not problematic in terms of causing ILD.
These findings are completely on board with the ILD group’s findings that methotrexate doesn’t cause pulmonary fibrosis in patients with RA, commented Julie Dawson, MD, of St. Helens and Knowsley Teaching Hospitals NHS Trust, St. Helens, England. Her own research, which includes a 10-year follow-up of patients with inflammatory arthritis, has shown that methotrexate does not appear to increase the risk of pulmonary fibrosis.
The study had no specific outside funding. Dr. Kiely reported having no conflicts of interest.
SOURCE: Kiely P et al. Rheumatology. 2019;58(suppl 3), Abstract 009.
BIRMINGHAM, ENGLAND – Data from two early RA inception cohorts provide reassurance that methotrexate does not cause interstitial lung disease and suggest that treatment with methotrexate might even be protective.
In the Early RA Study (ERAS) and Early RA Network (ERAN), which together include 2,701 patients with RA, 101 (3.7%) had interstitial lung disease (ILD). There were 92 patients with RA-ILD who had information available on exposure to any conventional synthetic disease-modifying antirheumatic drug (csDMARD); of these, 39 (2.5%) had been exposed to methotrexate (n = 1,578) and 53 (4.8%) to other csDMARDs (n = 1,114).
Multivariate analysis showed that methotrexate exposure was associated with a reduced risk of developing ILD, with an odds ratio of 0.48 (P = .004). In a separate analysis that excluded 25 patients who had ILD before they received any csDMARD therapy (n = 67), there was no association between methotrexate use and ILD (OR, 0.85; P = .578). In fact, there was a nonsignificant trend for a delayed onset of ILD in patients who had been treated with methotrexate (OR, 0.54; P = .072).
Methotrexate use is associated with an acute hypersensitivity pneumonitis in patients with RA, explained Patrick Kiely, MBBS, PhD, of St. George’s University Hospitals NHS Foundation Trust in London at the British Society for Rheumatology annual conference. “This is well recognized, it’s very rare [0.43%-1.00%], it’s easy to spot, and usually goes away if you stop methotrexate,” said Dr. Kiely, adding that “it’s not benign, and severe cases can be life threatening.”
Because of the association between methotrexate and pneumonitis, there has been concern that methotrexate may exacerbate or even cause ILD in RA but there are sparse data available to confirm this. The bottom line is that you should not start someone on methotrexate if you think their existing lung capacity is not up to treatment with methotrexate, Dr. Kiely said.
ILD is not always symptomatic in RA, but when it is, it is associated with very poor survival. The lung disease can be present before joint symptoms, Dr. Kiely said. Although less than 10% of cases may be symptomatic, this “is a big deal, because it has a high mortality, with death within 5 years. It’s the second-commonest cause of excess mortality in RA after cardiovascular disease.”
To look at the association between incident RA-ILD and the use of methotrexate, Dr. Kiely and associates analyzed data from ERAS (1986-2001) and ERAN (2002-2013), that together have more than 25 years of follow-up data on patients who were recruited at the first sign of RA symptoms. Patients within these cohorts have been treated according to best practice, and a range of outcomes – including RA-ILD – have been assessed at annual intervals.
In the patients who developed ILD after any csDMARD exposure, older age at RA onset (OR, 1.04; P less than .001) and having ever smoked (OR, 1.91; P = .016) were associated with the development of the lung disease. Incident ILD was also associated with being positive for rheumatoid factor (OR, 2.02; P = .029) at baseline. Being male was also associated with a higher risk for developing ILD, Dr. Kiely reported, as was a longer duration of time between the onset of first RA symptoms and the first secondary care visit. Conversely, the presence of nonrespiratory, major comorbidities at baseline appeared to be protective (OR, 0.62; P = .027).
“We found no association between methotrexate treatment and incident RA-ILD and a possibility that it may be protective,” Dr. Kiely concluded, noting that these data were now published in BMJ Open (2019;9:e028466. doi: 10.1136/bmjopen-2018-028466).
Following Dr. Kiely’s presentation, an audience member asked if the protective effect seen with methotrexate could have been caused by better disease control overall.
Dr. Kiely answered that, up until 2001, the time when ERAS was ongoing, standard practice in the United Kingdom was to use sulfasalazine, but then methotrexate started to be used in higher and higher doses, as seen in ERAN.
The interesting thing is that in ERAN more methotrexate was used in higher doses, but less RA-ILD was seen, Dr. Kiely observed. The overall prevalence of RA-ILD in the later early RA cohort was 3.2% and the median dose of methotrexate used was 20 mg. In ERAS, the prevalence was 4.2% and the median dose of methotrexate used was 10 mg.
There was a suggestion that disease control was slightly better in ERAN than ERAS, but that wasn’t statistically significant, Dr. Kiely said.
So, should a patient with RA and ILD be given methotrexate? There’s no reason not to, Dr. Kiely suggested, based on the evidence shown. Part of the challenge will now be convincing chest physician colleagues that methotrexate is not problematic in terms of causing ILD.
These findings are completely on board with the ILD group’s findings that methotrexate doesn’t cause pulmonary fibrosis in patients with RA, commented Julie Dawson, MD, of St. Helens and Knowsley Teaching Hospitals NHS Trust, St. Helens, England. Her own research, which includes a 10-year follow-up of patients with inflammatory arthritis, has shown that methotrexate does not appear to increase the risk of pulmonary fibrosis.
The study had no specific outside funding. Dr. Kiely reported having no conflicts of interest.
SOURCE: Kiely P et al. Rheumatology. 2019;58(suppl 3), Abstract 009.
REPORTING FROM BSR 2019
New recommendations on TB screening for health care workers
U.S. health care personnel no longer need to undergo routine tuberculosis testing in the absence of known exposure, according to new screening guidelines from the National Tuberculosis Controllers Association and CDC.
The revised guidelines on tuberculosis screening, testing, and treatment of U.S. health care personnel, published in Morbidity and Mortality Weekly Report, are the first update since 2005. The new recommendations reflect a reduction in concern about U.S. health care personnel’s risk of occupational exposure to latent and active tuberculosis infection.
Lynn E. Sosa, MD, from the Connecticut Department of Public Health and National Tuberculosis Controllers Association, and coauthors wrote that rates of tuberculosis infection in the United States have declined by 73% since 1991, from 10.4/100,000 population in 1991 to 2.8/100,000 in 2017. This has been matched by similar declines among health care workers, which the authors said raised questions about the cost-effectiveness of the previously recommended routine serial occupational testing.
“In addition, a recent retrospective cohort study of approximately 40,000 health care personnel at a tertiary U.S. medical center in a low TB-incidence state found an extremely low rate of TST conversion (0.3%) during 1998-2014, with a limited proportion attributable to occupational exposure,” they wrote.
The new guidelines recommend health care personnel undergo baseline or preplacement tuberculosis testing with an interferon-gamma release assay (IGRA) or a tuberculin skin test (TST), as well as individual risk assessment and symptom evaluation.
The individual risk assessment considers whether the person has lived in a country with a high tuberculosis rate, whether they are immunosuppressed, or whether they have had close contact with someone with infectious tuberculosis.
This risk assessment can help decide how to interpret an initial positive test result, the authors said.
“For example, health care personnel with a positive test who are asymptomatic, unlikely to be infected with M. [Mycobacterium] tuberculosis, and at low risk for progression on the basis of their risk assessment should have a second test (either an IGRA or a TST) as recommended in the 2017 TB diagnostic guidelines of the American Thoracic Society, Infectious Diseases Society of America, and CDC,” they wrote. “In this example, the health care personnel should be considered infected with M. tuberculosis only if both the first and second tests are positive.”
After that baseline testing, personnel do not need to undergo routine serial testing except in the case of known exposure or ongoing transmission. The guideline authors suggested serial screening might be considered for health care workers whose work puts them at greater risk – for example, pulmonologists or respiratory therapists – or for those working in settings in which transmission has happened in the past.
For personnel with latent tuberculosis infection, the guidelines recommend “encouragement of treatment” unless it is contraindicated, and annual symptom screening in those not undergoing treatment.
The guideline committee also advocated for annual tuberculosis education for all health care workers.
The new recommendations were based on a systematic review of 36 studies of tuberculosis screening and testing among health care personnel, 16 of which were performed in the United States, and all but two of which were conducted in a hospital setting.
The authors stressed that recommendations from the 2005 CDC guidelines – which do not pertain to health care personnel screening, testing, treatment and education – remain unchanged.
One author declared personal fees from the National Tuberculosis Controllers Association during the conduct of the study. Two others reported unrelated grants and personal fees from private industry. No other conflicts of interest were disclosed.
SOURCE: Sosa L et al. MMWR. 2019;68:439-43.
U.S. health care personnel no longer need to undergo routine tuberculosis testing in the absence of known exposure, according to new screening guidelines from the National Tuberculosis Controllers Association and CDC.
The revised guidelines on tuberculosis screening, testing, and treatment of U.S. health care personnel, published in Morbidity and Mortality Weekly Report, are the first update since 2005. The new recommendations reflect a reduction in concern about U.S. health care personnel’s risk of occupational exposure to latent and active tuberculosis infection.
Lynn E. Sosa, MD, from the Connecticut Department of Public Health and National Tuberculosis Controllers Association, and coauthors wrote that rates of tuberculosis infection in the United States have declined by 73% since 1991, from 10.4/100,000 population in 1991 to 2.8/100,000 in 2017. This has been matched by similar declines among health care workers, which the authors said raised questions about the cost-effectiveness of the previously recommended routine serial occupational testing.
“In addition, a recent retrospective cohort study of approximately 40,000 health care personnel at a tertiary U.S. medical center in a low TB-incidence state found an extremely low rate of TST conversion (0.3%) during 1998-2014, with a limited proportion attributable to occupational exposure,” they wrote.
The new guidelines recommend health care personnel undergo baseline or preplacement tuberculosis testing with an interferon-gamma release assay (IGRA) or a tuberculin skin test (TST), as well as individual risk assessment and symptom evaluation.
The individual risk assessment considers whether the person has lived in a country with a high tuberculosis rate, whether they are immunosuppressed, or whether they have had close contact with someone with infectious tuberculosis.
This risk assessment can help decide how to interpret an initial positive test result, the authors said.
“For example, health care personnel with a positive test who are asymptomatic, unlikely to be infected with M. [Mycobacterium] tuberculosis, and at low risk for progression on the basis of their risk assessment should have a second test (either an IGRA or a TST) as recommended in the 2017 TB diagnostic guidelines of the American Thoracic Society, Infectious Diseases Society of America, and CDC,” they wrote. “In this example, the health care personnel should be considered infected with M. tuberculosis only if both the first and second tests are positive.”
After that baseline testing, personnel do not need to undergo routine serial testing except in the case of known exposure or ongoing transmission. The guideline authors suggested serial screening might be considered for health care workers whose work puts them at greater risk – for example, pulmonologists or respiratory therapists – or for those working in settings in which transmission has happened in the past.
For personnel with latent tuberculosis infection, the guidelines recommend “encouragement of treatment” unless it is contraindicated, and annual symptom screening in those not undergoing treatment.
The guideline committee also advocated for annual tuberculosis education for all health care workers.
The new recommendations were based on a systematic review of 36 studies of tuberculosis screening and testing among health care personnel, 16 of which were performed in the United States, and all but two of which were conducted in a hospital setting.
The authors stressed that recommendations from the 2005 CDC guidelines – which do not pertain to health care personnel screening, testing, treatment and education – remain unchanged.
One author declared personal fees from the National Tuberculosis Controllers Association during the conduct of the study. Two others reported unrelated grants and personal fees from private industry. No other conflicts of interest were disclosed.
SOURCE: Sosa L et al. MMWR. 2019;68:439-43.
U.S. health care personnel no longer need to undergo routine tuberculosis testing in the absence of known exposure, according to new screening guidelines from the National Tuberculosis Controllers Association and CDC.
The revised guidelines on tuberculosis screening, testing, and treatment of U.S. health care personnel, published in Morbidity and Mortality Weekly Report, are the first update since 2005. The new recommendations reflect a reduction in concern about U.S. health care personnel’s risk of occupational exposure to latent and active tuberculosis infection.
Lynn E. Sosa, MD, from the Connecticut Department of Public Health and National Tuberculosis Controllers Association, and coauthors wrote that rates of tuberculosis infection in the United States have declined by 73% since 1991, from 10.4/100,000 population in 1991 to 2.8/100,000 in 2017. This has been matched by similar declines among health care workers, which the authors said raised questions about the cost-effectiveness of the previously recommended routine serial occupational testing.
“In addition, a recent retrospective cohort study of approximately 40,000 health care personnel at a tertiary U.S. medical center in a low TB-incidence state found an extremely low rate of TST conversion (0.3%) during 1998-2014, with a limited proportion attributable to occupational exposure,” they wrote.
The new guidelines recommend health care personnel undergo baseline or preplacement tuberculosis testing with an interferon-gamma release assay (IGRA) or a tuberculin skin test (TST), as well as individual risk assessment and symptom evaluation.
The individual risk assessment considers whether the person has lived in a country with a high tuberculosis rate, whether they are immunosuppressed, or whether they have had close contact with someone with infectious tuberculosis.
This risk assessment can help decide how to interpret an initial positive test result, the authors said.
“For example, health care personnel with a positive test who are asymptomatic, unlikely to be infected with M. [Mycobacterium] tuberculosis, and at low risk for progression on the basis of their risk assessment should have a second test (either an IGRA or a TST) as recommended in the 2017 TB diagnostic guidelines of the American Thoracic Society, Infectious Diseases Society of America, and CDC,” they wrote. “In this example, the health care personnel should be considered infected with M. tuberculosis only if both the first and second tests are positive.”
After that baseline testing, personnel do not need to undergo routine serial testing except in the case of known exposure or ongoing transmission. The guideline authors suggested serial screening might be considered for health care workers whose work puts them at greater risk – for example, pulmonologists or respiratory therapists – or for those working in settings in which transmission has happened in the past.
For personnel with latent tuberculosis infection, the guidelines recommend “encouragement of treatment” unless it is contraindicated, and annual symptom screening in those not undergoing treatment.
The guideline committee also advocated for annual tuberculosis education for all health care workers.
The new recommendations were based on a systematic review of 36 studies of tuberculosis screening and testing among health care personnel, 16 of which were performed in the United States, and all but two of which were conducted in a hospital setting.
The authors stressed that recommendations from the 2005 CDC guidelines – which do not pertain to health care personnel screening, testing, treatment and education – remain unchanged.
One author declared personal fees from the National Tuberculosis Controllers Association during the conduct of the study. Two others reported unrelated grants and personal fees from private industry. No other conflicts of interest were disclosed.
SOURCE: Sosa L et al. MMWR. 2019;68:439-43.
FROM MMWR
Survey: Physicians predict increase in measles deaths
by real-time market insights technology firm InCrowd.
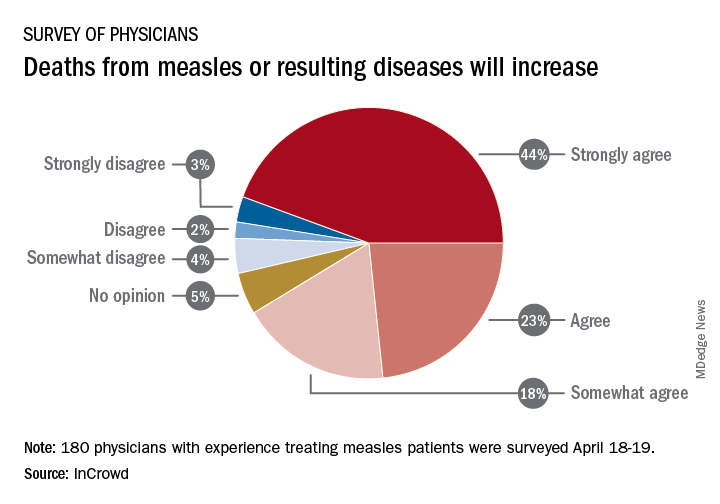
Among the 180 physicians with experience treating measles, 23% agreed and 44% said that they strongly agreed with the statement that measles deaths would increase, and another 18% said that they somewhat agreed. Only 9% expressed some level of disagreement, InCrowd said.
Most of those respondents also believe that summer travel will increase measles outbreaks (29% agreed and 30% strongly agreed) and that more communities will adopt requirements for measles vaccinations (26% and 36%). A majority also said that education about vaccinations will improve (26% agreed and 29% strongly agreed), but almost half of the physicians surveyed also expect vaccination misinformation to get worse (29% and 19%), InCrowd reported.
“With 44% of respondents predicting a high likelihood that deaths caused by measles will increase, the data show the imperative for physicians and patients to keep up the dialogue. … We have a long way to go before declaring victory,” said Diane Hayes, PhD, president and cofounder of InCrowd.
The InCrowd 5-minute microsurvey was conducted on April 18-19, 2019, and included 455 primary care physicians, of whom 40% said that they have treated or knew of colleagues in their facility or community who have treated patients with measles. Of those 180 respondents, 89 were pediatricians and 91 were in other primary care specialties.
by real-time market insights technology firm InCrowd.

Among the 180 physicians with experience treating measles, 23% agreed and 44% said that they strongly agreed with the statement that measles deaths would increase, and another 18% said that they somewhat agreed. Only 9% expressed some level of disagreement, InCrowd said.
Most of those respondents also believe that summer travel will increase measles outbreaks (29% agreed and 30% strongly agreed) and that more communities will adopt requirements for measles vaccinations (26% and 36%). A majority also said that education about vaccinations will improve (26% agreed and 29% strongly agreed), but almost half of the physicians surveyed also expect vaccination misinformation to get worse (29% and 19%), InCrowd reported.
“With 44% of respondents predicting a high likelihood that deaths caused by measles will increase, the data show the imperative for physicians and patients to keep up the dialogue. … We have a long way to go before declaring victory,” said Diane Hayes, PhD, president and cofounder of InCrowd.
The InCrowd 5-minute microsurvey was conducted on April 18-19, 2019, and included 455 primary care physicians, of whom 40% said that they have treated or knew of colleagues in their facility or community who have treated patients with measles. Of those 180 respondents, 89 were pediatricians and 91 were in other primary care specialties.
by real-time market insights technology firm InCrowd.

Among the 180 physicians with experience treating measles, 23% agreed and 44% said that they strongly agreed with the statement that measles deaths would increase, and another 18% said that they somewhat agreed. Only 9% expressed some level of disagreement, InCrowd said.
Most of those respondents also believe that summer travel will increase measles outbreaks (29% agreed and 30% strongly agreed) and that more communities will adopt requirements for measles vaccinations (26% and 36%). A majority also said that education about vaccinations will improve (26% agreed and 29% strongly agreed), but almost half of the physicians surveyed also expect vaccination misinformation to get worse (29% and 19%), InCrowd reported.
“With 44% of respondents predicting a high likelihood that deaths caused by measles will increase, the data show the imperative for physicians and patients to keep up the dialogue. … We have a long way to go before declaring victory,” said Diane Hayes, PhD, president and cofounder of InCrowd.
The InCrowd 5-minute microsurvey was conducted on April 18-19, 2019, and included 455 primary care physicians, of whom 40% said that they have treated or knew of colleagues in their facility or community who have treated patients with measles. Of those 180 respondents, 89 were pediatricians and 91 were in other primary care specialties.
Young children with neuromuscular disease are vulnerable to respiratory viruses
This highlights the need for new vaccines
Influenza gets a lot of attention each winter, but respiratory syncytial virus (RSV) and other respiratory viruses have as much or more impact on pediatric populations, particularly certain high-risk groups. But currently there are no vaccines for noninfluenza respiratory viruses. That said, several are under development, for RSV and parainfluenza.
Which groups are likely to get the most benefit from these newer vaccines?
We all are aware of the extra vulnerability to respiratory viruses (RSV being the most frequent) in premature infants, those with chronic lung disease, or those with congenital heart syndromes; such vulnerable patients are not infrequently seen in routine practice. A recent report shined a brighter light on such a group.
Real-world data from a nationwide Canadian surveillance system (CARESS) was used to analyze relative risks of categories of young children who are thought to be vulnerable to respiratory viruses, with a particular focus on those with neuromuscular disease. The CARESS investigators analyzed 12 years’ data on respiratory hospitalizations from among palivizumab-prophylaxed patients (including specific data on RSV when patients were tested for RSV per standard of care).1 Unfortunately, RSV testing was not universal despite hospitalization, so the true incidence of RSV-specific hospitalizations was likely underestimated.
Nevertheless, more than 25,000 children from 2005 through 2017 were grouped into three categories of palivizumab-prophylaxed high-risk children: standard indications (SI), n = 20,335; chronic medical conditions (CMD), n = 4,063; and neuromuscular disease (NMD), n = 605. This study is notable for having a relatively large number of neuromuscular disease subjects. Two-thirds of each group were fully palivizumab adherent.
The SI group included the standard American Academy of Pediatrics–recommended groups, such as premature infants, congenital heart disease, etc.
The CMD group included conditions that lead clinicians to use palivizumab off label, such as cystic fibrosis, congenital airway anomalies, immunodeficiency, and pulmonary disorders.
The NMD participants were subdivided into two groups. Group 1 comprised general hypotonic neuromuscular diseases such as hypoxic-ischemic encephalopathy, Prader-Willi syndrome, chromosomal disorders, and migration/demyelinating diseases. Group 2 included more severe infantile neuromuscular disorders, such as spinal muscular atrophy, myotonic dystrophy, centronuclear and nemaline myopathy, mitochondrial and glycogen storage myopathies, or arthrogryposis.
Overall, 6.9% of CARESS RSV-prophylaxed subjects were hospitalized. About one in five hospitalized patients from each group was hospitalized more than once. Specific respiratory hospitalization rates for each group were 6% (n = 1,228) for SI subjects and 9.4% (n = 380) for CMD, compared with 19.2% (n = 116) for NMD subjects.
It is unclear what proportion underwent RSV testing, but a total of 334 were confirmed RSV positive: 261 were SI, 54 were CMD and 19 were NMD. The RSV-test-positive rate was 1.5% for SI, 1.6% for CMD and 3.3% for NMD; so while a higher number of SI children were RSV positive, the rate of RSV positivity was actually highest with NMD.
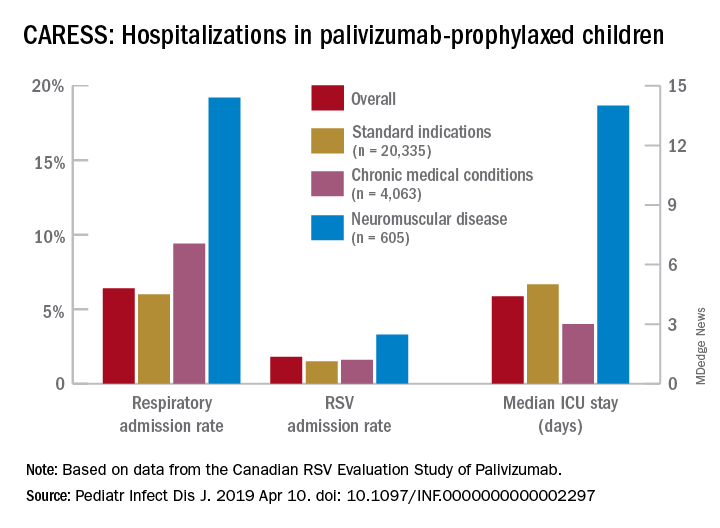
RSV-positive subjects needing ICU care among NMD patients also had longer ICU stays (median 14 days), compared with RSV-positive CMD or SI subjects (median 3 and 5 days, respectively). Further, hospitalized RSV-positive NMD subjects presented more frequently with pneumonia (42% vs. 30% for CMD and 20% for SI) while hospitalized RSV-positive SI subjects more often had apnea (17% vs. 10% for NMD and 5% for CMD, P less than .05).
These differences in the courses of NMD patients raise the question as to whether the NMD group was somehow different from the SI and CMD groups, other than muscular weakness that likely leads to less ability to clear secretions and a less efficient cough. It turns out that NMD children were older and had worse neonatal medical courses (longer hospital stays, more often ventilated, and used oxygen longer). It could be argued that these differences may have been in part due to the muscular weakness inherent in their underlying disease, but they appear to be predictors of worse respiratory infectious disease than other vulnerable populations as the NMD children get older.
Indeed, the overall risk of any respiratory admission among NMD subjects was nearly twice as high, compared with SI (hazard ratio, 1.90, P less than .0005); but the somewhat higher risk for NMD vs. CMD was not significant (HR, 1.33, P = .090). However, when looking specifically at RSV confirmed admissions, NMD had more than twice the hospitalization risk than either other group (HR, 2.26, P = .001 vs. SI; and HR, 2.74, P = .001 vs. CMD).
Further, an NMD subgroup analysis showed 1.69 times the overall respiratory hospitalization risk among the more severe vs. less severe NMD group, but a similar risk of RSV admission. The authors point out that one reason for this discrepancy may be a higher probability of aspiration causing hospitalization because of more dramatic acute events during respiratory infections in patients with more severe NMD. It also may be that palivizumab evened the playing field for RSV but not for other viruses such as parainfluenza, adenovirus, or even rhinovirus.
Nevertheless, these data tell us that risk of respiratory disease severe enough to need hospitalization continues to an older age in NMD than SI or CMD patients, well past 2 years of age. And the risk is not only from RSV. That said, RSV remains a player in some patients (particularly NMD patients) despite palivizumab prophylaxis, highlighting the need for RSV as well as parainfluenza vaccines. While these vaccines should help all young children, they seem likely to be even more beneficial for high-risk children including those with NMD, and particularly those with more severe NMD.
Eleven among 60 total candidate RSV vaccines (live attenuated, particle based, or vector based) are currently in clinical trials.2 Fewer parainfluenza vaccines are in the pipeline, but clinical trials also are underway.3-5 Approval of such vaccines is not expected until the mid-2020s, so at present we are left with providing palivizumab to our vulnerable patients while emphasizing nonmedical strategies that may help prevent respiratory viruses. These only partially successful preventive interventions include breastfeeding, avoiding secondhand smoke, and avoiding known high-risk exposures, such as large day care centers.
My hope is for quicker than projected progress on the vaccine front so that winter admissions for respiratory viruses might decrease in numbers similar to the decrease we have noted with another vaccine successful against a seasonally active pathogen – rotavirus.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatr Infect Dis J. 2019 Apr 10. doi: 10.1097/INF.0000000000002297.
2. “Advances in RSV Vaccine Research and Development – A Global Agenda.”
3. J Pediatric Infect Dis Soc. 2015 Dec;4(4): e143-6.
4. J Virol. 2015 Oct;89(20):10319-32.
5. Vaccine. 2017 Dec 18;35(51):7139-46.
This highlights the need for new vaccines
This highlights the need for new vaccines
Influenza gets a lot of attention each winter, but respiratory syncytial virus (RSV) and other respiratory viruses have as much or more impact on pediatric populations, particularly certain high-risk groups. But currently there are no vaccines for noninfluenza respiratory viruses. That said, several are under development, for RSV and parainfluenza.
Which groups are likely to get the most benefit from these newer vaccines?
We all are aware of the extra vulnerability to respiratory viruses (RSV being the most frequent) in premature infants, those with chronic lung disease, or those with congenital heart syndromes; such vulnerable patients are not infrequently seen in routine practice. A recent report shined a brighter light on such a group.
Real-world data from a nationwide Canadian surveillance system (CARESS) was used to analyze relative risks of categories of young children who are thought to be vulnerable to respiratory viruses, with a particular focus on those with neuromuscular disease. The CARESS investigators analyzed 12 years’ data on respiratory hospitalizations from among palivizumab-prophylaxed patients (including specific data on RSV when patients were tested for RSV per standard of care).1 Unfortunately, RSV testing was not universal despite hospitalization, so the true incidence of RSV-specific hospitalizations was likely underestimated.
Nevertheless, more than 25,000 children from 2005 through 2017 were grouped into three categories of palivizumab-prophylaxed high-risk children: standard indications (SI), n = 20,335; chronic medical conditions (CMD), n = 4,063; and neuromuscular disease (NMD), n = 605. This study is notable for having a relatively large number of neuromuscular disease subjects. Two-thirds of each group were fully palivizumab adherent.
The SI group included the standard American Academy of Pediatrics–recommended groups, such as premature infants, congenital heart disease, etc.
The CMD group included conditions that lead clinicians to use palivizumab off label, such as cystic fibrosis, congenital airway anomalies, immunodeficiency, and pulmonary disorders.
The NMD participants were subdivided into two groups. Group 1 comprised general hypotonic neuromuscular diseases such as hypoxic-ischemic encephalopathy, Prader-Willi syndrome, chromosomal disorders, and migration/demyelinating diseases. Group 2 included more severe infantile neuromuscular disorders, such as spinal muscular atrophy, myotonic dystrophy, centronuclear and nemaline myopathy, mitochondrial and glycogen storage myopathies, or arthrogryposis.
Overall, 6.9% of CARESS RSV-prophylaxed subjects were hospitalized. About one in five hospitalized patients from each group was hospitalized more than once. Specific respiratory hospitalization rates for each group were 6% (n = 1,228) for SI subjects and 9.4% (n = 380) for CMD, compared with 19.2% (n = 116) for NMD subjects.
It is unclear what proportion underwent RSV testing, but a total of 334 were confirmed RSV positive: 261 were SI, 54 were CMD and 19 were NMD. The RSV-test-positive rate was 1.5% for SI, 1.6% for CMD and 3.3% for NMD; so while a higher number of SI children were RSV positive, the rate of RSV positivity was actually highest with NMD.

RSV-positive subjects needing ICU care among NMD patients also had longer ICU stays (median 14 days), compared with RSV-positive CMD or SI subjects (median 3 and 5 days, respectively). Further, hospitalized RSV-positive NMD subjects presented more frequently with pneumonia (42% vs. 30% for CMD and 20% for SI) while hospitalized RSV-positive SI subjects more often had apnea (17% vs. 10% for NMD and 5% for CMD, P less than .05).
These differences in the courses of NMD patients raise the question as to whether the NMD group was somehow different from the SI and CMD groups, other than muscular weakness that likely leads to less ability to clear secretions and a less efficient cough. It turns out that NMD children were older and had worse neonatal medical courses (longer hospital stays, more often ventilated, and used oxygen longer). It could be argued that these differences may have been in part due to the muscular weakness inherent in their underlying disease, but they appear to be predictors of worse respiratory infectious disease than other vulnerable populations as the NMD children get older.
Indeed, the overall risk of any respiratory admission among NMD subjects was nearly twice as high, compared with SI (hazard ratio, 1.90, P less than .0005); but the somewhat higher risk for NMD vs. CMD was not significant (HR, 1.33, P = .090). However, when looking specifically at RSV confirmed admissions, NMD had more than twice the hospitalization risk than either other group (HR, 2.26, P = .001 vs. SI; and HR, 2.74, P = .001 vs. CMD).
Further, an NMD subgroup analysis showed 1.69 times the overall respiratory hospitalization risk among the more severe vs. less severe NMD group, but a similar risk of RSV admission. The authors point out that one reason for this discrepancy may be a higher probability of aspiration causing hospitalization because of more dramatic acute events during respiratory infections in patients with more severe NMD. It also may be that palivizumab evened the playing field for RSV but not for other viruses such as parainfluenza, adenovirus, or even rhinovirus.
Nevertheless, these data tell us that risk of respiratory disease severe enough to need hospitalization continues to an older age in NMD than SI or CMD patients, well past 2 years of age. And the risk is not only from RSV. That said, RSV remains a player in some patients (particularly NMD patients) despite palivizumab prophylaxis, highlighting the need for RSV as well as parainfluenza vaccines. While these vaccines should help all young children, they seem likely to be even more beneficial for high-risk children including those with NMD, and particularly those with more severe NMD.
Eleven among 60 total candidate RSV vaccines (live attenuated, particle based, or vector based) are currently in clinical trials.2 Fewer parainfluenza vaccines are in the pipeline, but clinical trials also are underway.3-5 Approval of such vaccines is not expected until the mid-2020s, so at present we are left with providing palivizumab to our vulnerable patients while emphasizing nonmedical strategies that may help prevent respiratory viruses. These only partially successful preventive interventions include breastfeeding, avoiding secondhand smoke, and avoiding known high-risk exposures, such as large day care centers.
My hope is for quicker than projected progress on the vaccine front so that winter admissions for respiratory viruses might decrease in numbers similar to the decrease we have noted with another vaccine successful against a seasonally active pathogen – rotavirus.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatr Infect Dis J. 2019 Apr 10. doi: 10.1097/INF.0000000000002297.
2. “Advances in RSV Vaccine Research and Development – A Global Agenda.”
3. J Pediatric Infect Dis Soc. 2015 Dec;4(4): e143-6.
4. J Virol. 2015 Oct;89(20):10319-32.
5. Vaccine. 2017 Dec 18;35(51):7139-46.
Influenza gets a lot of attention each winter, but respiratory syncytial virus (RSV) and other respiratory viruses have as much or more impact on pediatric populations, particularly certain high-risk groups. But currently there are no vaccines for noninfluenza respiratory viruses. That said, several are under development, for RSV and parainfluenza.
Which groups are likely to get the most benefit from these newer vaccines?
We all are aware of the extra vulnerability to respiratory viruses (RSV being the most frequent) in premature infants, those with chronic lung disease, or those with congenital heart syndromes; such vulnerable patients are not infrequently seen in routine practice. A recent report shined a brighter light on such a group.
Real-world data from a nationwide Canadian surveillance system (CARESS) was used to analyze relative risks of categories of young children who are thought to be vulnerable to respiratory viruses, with a particular focus on those with neuromuscular disease. The CARESS investigators analyzed 12 years’ data on respiratory hospitalizations from among palivizumab-prophylaxed patients (including specific data on RSV when patients were tested for RSV per standard of care).1 Unfortunately, RSV testing was not universal despite hospitalization, so the true incidence of RSV-specific hospitalizations was likely underestimated.
Nevertheless, more than 25,000 children from 2005 through 2017 were grouped into three categories of palivizumab-prophylaxed high-risk children: standard indications (SI), n = 20,335; chronic medical conditions (CMD), n = 4,063; and neuromuscular disease (NMD), n = 605. This study is notable for having a relatively large number of neuromuscular disease subjects. Two-thirds of each group were fully palivizumab adherent.
The SI group included the standard American Academy of Pediatrics–recommended groups, such as premature infants, congenital heart disease, etc.
The CMD group included conditions that lead clinicians to use palivizumab off label, such as cystic fibrosis, congenital airway anomalies, immunodeficiency, and pulmonary disorders.
The NMD participants were subdivided into two groups. Group 1 comprised general hypotonic neuromuscular diseases such as hypoxic-ischemic encephalopathy, Prader-Willi syndrome, chromosomal disorders, and migration/demyelinating diseases. Group 2 included more severe infantile neuromuscular disorders, such as spinal muscular atrophy, myotonic dystrophy, centronuclear and nemaline myopathy, mitochondrial and glycogen storage myopathies, or arthrogryposis.
Overall, 6.9% of CARESS RSV-prophylaxed subjects were hospitalized. About one in five hospitalized patients from each group was hospitalized more than once. Specific respiratory hospitalization rates for each group were 6% (n = 1,228) for SI subjects and 9.4% (n = 380) for CMD, compared with 19.2% (n = 116) for NMD subjects.
It is unclear what proportion underwent RSV testing, but a total of 334 were confirmed RSV positive: 261 were SI, 54 were CMD and 19 were NMD. The RSV-test-positive rate was 1.5% for SI, 1.6% for CMD and 3.3% for NMD; so while a higher number of SI children were RSV positive, the rate of RSV positivity was actually highest with NMD.

RSV-positive subjects needing ICU care among NMD patients also had longer ICU stays (median 14 days), compared with RSV-positive CMD or SI subjects (median 3 and 5 days, respectively). Further, hospitalized RSV-positive NMD subjects presented more frequently with pneumonia (42% vs. 30% for CMD and 20% for SI) while hospitalized RSV-positive SI subjects more often had apnea (17% vs. 10% for NMD and 5% for CMD, P less than .05).
These differences in the courses of NMD patients raise the question as to whether the NMD group was somehow different from the SI and CMD groups, other than muscular weakness that likely leads to less ability to clear secretions and a less efficient cough. It turns out that NMD children were older and had worse neonatal medical courses (longer hospital stays, more often ventilated, and used oxygen longer). It could be argued that these differences may have been in part due to the muscular weakness inherent in their underlying disease, but they appear to be predictors of worse respiratory infectious disease than other vulnerable populations as the NMD children get older.
Indeed, the overall risk of any respiratory admission among NMD subjects was nearly twice as high, compared with SI (hazard ratio, 1.90, P less than .0005); but the somewhat higher risk for NMD vs. CMD was not significant (HR, 1.33, P = .090). However, when looking specifically at RSV confirmed admissions, NMD had more than twice the hospitalization risk than either other group (HR, 2.26, P = .001 vs. SI; and HR, 2.74, P = .001 vs. CMD).
Further, an NMD subgroup analysis showed 1.69 times the overall respiratory hospitalization risk among the more severe vs. less severe NMD group, but a similar risk of RSV admission. The authors point out that one reason for this discrepancy may be a higher probability of aspiration causing hospitalization because of more dramatic acute events during respiratory infections in patients with more severe NMD. It also may be that palivizumab evened the playing field for RSV but not for other viruses such as parainfluenza, adenovirus, or even rhinovirus.
Nevertheless, these data tell us that risk of respiratory disease severe enough to need hospitalization continues to an older age in NMD than SI or CMD patients, well past 2 years of age. And the risk is not only from RSV. That said, RSV remains a player in some patients (particularly NMD patients) despite palivizumab prophylaxis, highlighting the need for RSV as well as parainfluenza vaccines. While these vaccines should help all young children, they seem likely to be even more beneficial for high-risk children including those with NMD, and particularly those with more severe NMD.
Eleven among 60 total candidate RSV vaccines (live attenuated, particle based, or vector based) are currently in clinical trials.2 Fewer parainfluenza vaccines are in the pipeline, but clinical trials also are underway.3-5 Approval of such vaccines is not expected until the mid-2020s, so at present we are left with providing palivizumab to our vulnerable patients while emphasizing nonmedical strategies that may help prevent respiratory viruses. These only partially successful preventive interventions include breastfeeding, avoiding secondhand smoke, and avoiding known high-risk exposures, such as large day care centers.
My hope is for quicker than projected progress on the vaccine front so that winter admissions for respiratory viruses might decrease in numbers similar to the decrease we have noted with another vaccine successful against a seasonally active pathogen – rotavirus.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Children’s Mercy Hospital receives grant funding to study two candidate RSV vaccines. The hospital also receives CDC funding under the New Vaccine Surveillance Network for multicenter surveillance of acute respiratory infections, including influenza, RSV, and parainfluenza virus. Email Dr. Harrison at [email protected].
References
1. Pediatr Infect Dis J. 2019 Apr 10. doi: 10.1097/INF.0000000000002297.
2. “Advances in RSV Vaccine Research and Development – A Global Agenda.”
3. J Pediatric Infect Dis Soc. 2015 Dec;4(4): e143-6.
4. J Virol. 2015 Oct;89(20):10319-32.
5. Vaccine. 2017 Dec 18;35(51):7139-46.
Is it measles? – Diagnosis and management for the pediatric provider
The mother of an 8-month-old calls your office and is hysterical. Her daughter has had cough for a few days with high fevers and now has developed a full body rash. She is worried about measles and is on her way to your office.
We are in the middle of a measles epidemic, there’s no denying it. Measles was declared eliminated in 2000, but reported cases in the United States have been on the rise, and are now at the highest number since 2014. Five months into 2019, there have been 839 reported cases as of May 13). Measles outbreaks (defined by the Centers for Disease Control and Prevention as three or more cases) have been reported in California, Georgia, Maryland, Michigan, New Jersey, New York, and Pennsylvania. When vaccination rates fall, it is easy for measles to spread. The virus is highly contagious in nonimmune people, because of its airborne spread and its persistence in the environment for hours.
First – is it really measles?
It can be difficult to distinguish the maculopapular rash of measles from similar rashes that occur with more benign viral illnesses. Adding to the challenge, the last major measles outbreak in the United States was over 2 decades ago, and many practicing pediatricians have never seen a single case. So, what clinical features can help distinguish measles from other febrile illnesses?
The prodromal phase of measles lasts approximately 2-4 days and children have high fevers (103°-105° F), anorexia, and malaise. Conjunctivitis, coryza, and cough develop during this phase, and precede any rash. Koplik spots appear during the prodromal phase, but are not seen in all cases. These spots are 1- to 3-mm blue-white lesions on an erythematous base on the buccal mucosa, classically opposite the first molar. The spots often slough once the rash appears. The rash appears 2-4 days after the onset of fever, and is initially maculopapular and blanching. The first lesions appear on the face and neck, and the rash spreads cranial to caudal, typically sparing palms and soles. After days 3-4, the rash will no longer blanch. High fevers persist for 2-4 more days with rash, ongoing respiratory symptoms, conjunctivitis, and pharyngitis. Note that the fever will persist even with development of the rash, unlike in roseola.
It is not only important to diagnosis measles from a public health standpoint, but also because measles can have severe complications, especially in infants and children under 5 years. During the 1989-1991 outbreak, the mortality rate was 2.2 deaths per 1,000 cases (J Infect Dis. 2004 May 1. doi: 10.1086/377694).
Six percent of patients develop pneumonia, which in infants and toddlers can lead to respiratory distress or failure requiring hospitalization. Pneumonia is responsible for 60% of measles deaths, according to the CDC “Pink Book,” Epidemiology and Prevention of Vaccine-Preventable Diseases, chapter 13 on measles, 13th Ed., 2015. Ocular complications include keratitis and corneal ulceration. Measles also can cause serious neurologic complications. Encephalitis, seen in 1 per 1,000 cases, usually arises several days after the rash and may present with seizure or encephalopathy. Acute disseminated encephalomyelitis (ADEM), an inflammatory demyelinating disease of the central nervous system, occurs in approximately 1 per 1,000 cases, typically presents during the recovery phase (1-2 weeks after rash), and can have long-term sequelae. Subacute sclerosing panencephalitis (SSPE) is a progressive and fatal neurodegenerative disorder, and presents 7-10 years after measles infection.
Should you transfer the patient to a hospital?
Unless there is a medical need for the child to be admitted, sending a patient with potential measles to the hospital is not necessary, and can cause exposure to a large group of medical personnel, and patients who cannot be vaccinated (such as infants, immunocompromised patients, and pregnant women). However, if there is concern for complications such as seizures, encephalitis, or pneumonia, then transfer is indicated. Call the accepting hospital in advance so the staff can prepare for the patient. During transfer, place a standard face mask on the patient and instruct the patient not to remove it.
For hospitals accepting a suspected measles case, meet the patient outside of the facility and ensure that the patient is wearing a standard face mask. All staff interacting with the patient should practice contact and airborne precautions (N95 respirator mask). Take the patient directly to an isolation room with negative airflow. Caution pregnant staff that they should not have contact with the patient.
Which diagnostic tests should you use?
Diagnosis can be made based on serum antibody tests (measles IgM and IgG), throat or urine viral cultures, and nasopharyngeal and throat specimen polymerase chain reaction (PCR) testing. The CDC recommends obtaining a serum sample for measles IgM testing and a throat swab for PCR in all suspected cases, but local health departments vary in their specific testing recommendations. Familiarize yourself with the tests recommended by your local department of health, and where they prefer testing on outpatients to be done. Confirmed measles should be reported to your department of health.
What are considerations for community pediatric offices?
Update families in emails to call ahead if they suspect measles. This way the office can prepare a room for the family, and have the family immediately brought back without exposing staff and other families in the waiting area. It may be more prudent to examine these children at the end of the clinic day as the virus can persist for up to 2 hours on fomites and in the air. Therefore, all waiting areas and shared air spaces (including those with shared air ducts) should be cleared for 2 hours after the patient leaves.
When should you provide prophylaxis after exposure?
A patient with suspected measles does not require immediate vaccination. If it is measles, it is already too late to vaccinate. If measles is ruled out, the child should follow the standard measles vaccination guidelines.
Individuals are contagious from 4 days before to 4 days after the rash appears.
If measles is confirmed, all people who are unvaccinated or undervaccinated and were exposed to the confirmed case during the contagious period should be vaccinated within 72 hours of exposure. Infants 6 months or older may safely receive the MMR vaccine. However, infants vaccinated with MMR before their first birthday must be vaccinated again at age 12-15 months (greater than 28 days after prior vaccine) and at 4-6 years. Immunoglobulin prophylaxis should be given intramuscularly in exposed infants ages birth to less than 6 months, and in those ages 6-12 months who present beyond the 72-hour window. Unvaccinated or undervaccinated, exposed individuals at high risk for complications from measles (immunocompromised, pregnant) also should receive immunoglobulin.
What should you tell traveling families?
Several countries have large, ongoing measles outbreaks, including Israel, Ukraine, and the Philippines. Before international travel, infants 6-11 months should receive one dose of MMR vaccine, and children 12 months and older need two doses separated by at least 28 days. For unvaccinated or undervaccinated children, consider advising families to hold off travel to high-risk countries, or understand the indications to vaccinate a child upon return.
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Medical Center in Washington. She said she has no relevant financial disclosures. Dr. Emily Willner is a pediatric emergency medicine attending at Children’s National Medical Center, and an assistant professor of pediatrics and emergency medicine at George Washington University, Washington. She has no relevant financial disclosures.
The mother of an 8-month-old calls your office and is hysterical. Her daughter has had cough for a few days with high fevers and now has developed a full body rash. She is worried about measles and is on her way to your office.
We are in the middle of a measles epidemic, there’s no denying it. Measles was declared eliminated in 2000, but reported cases in the United States have been on the rise, and are now at the highest number since 2014. Five months into 2019, there have been 839 reported cases as of May 13). Measles outbreaks (defined by the Centers for Disease Control and Prevention as three or more cases) have been reported in California, Georgia, Maryland, Michigan, New Jersey, New York, and Pennsylvania. When vaccination rates fall, it is easy for measles to spread. The virus is highly contagious in nonimmune people, because of its airborne spread and its persistence in the environment for hours.
First – is it really measles?
It can be difficult to distinguish the maculopapular rash of measles from similar rashes that occur with more benign viral illnesses. Adding to the challenge, the last major measles outbreak in the United States was over 2 decades ago, and many practicing pediatricians have never seen a single case. So, what clinical features can help distinguish measles from other febrile illnesses?
The prodromal phase of measles lasts approximately 2-4 days and children have high fevers (103°-105° F), anorexia, and malaise. Conjunctivitis, coryza, and cough develop during this phase, and precede any rash. Koplik spots appear during the prodromal phase, but are not seen in all cases. These spots are 1- to 3-mm blue-white lesions on an erythematous base on the buccal mucosa, classically opposite the first molar. The spots often slough once the rash appears. The rash appears 2-4 days after the onset of fever, and is initially maculopapular and blanching. The first lesions appear on the face and neck, and the rash spreads cranial to caudal, typically sparing palms and soles. After days 3-4, the rash will no longer blanch. High fevers persist for 2-4 more days with rash, ongoing respiratory symptoms, conjunctivitis, and pharyngitis. Note that the fever will persist even with development of the rash, unlike in roseola.
It is not only important to diagnosis measles from a public health standpoint, but also because measles can have severe complications, especially in infants and children under 5 years. During the 1989-1991 outbreak, the mortality rate was 2.2 deaths per 1,000 cases (J Infect Dis. 2004 May 1. doi: 10.1086/377694).
Six percent of patients develop pneumonia, which in infants and toddlers can lead to respiratory distress or failure requiring hospitalization. Pneumonia is responsible for 60% of measles deaths, according to the CDC “Pink Book,” Epidemiology and Prevention of Vaccine-Preventable Diseases, chapter 13 on measles, 13th Ed., 2015. Ocular complications include keratitis and corneal ulceration. Measles also can cause serious neurologic complications. Encephalitis, seen in 1 per 1,000 cases, usually arises several days after the rash and may present with seizure or encephalopathy. Acute disseminated encephalomyelitis (ADEM), an inflammatory demyelinating disease of the central nervous system, occurs in approximately 1 per 1,000 cases, typically presents during the recovery phase (1-2 weeks after rash), and can have long-term sequelae. Subacute sclerosing panencephalitis (SSPE) is a progressive and fatal neurodegenerative disorder, and presents 7-10 years after measles infection.
Should you transfer the patient to a hospital?
Unless there is a medical need for the child to be admitted, sending a patient with potential measles to the hospital is not necessary, and can cause exposure to a large group of medical personnel, and patients who cannot be vaccinated (such as infants, immunocompromised patients, and pregnant women). However, if there is concern for complications such as seizures, encephalitis, or pneumonia, then transfer is indicated. Call the accepting hospital in advance so the staff can prepare for the patient. During transfer, place a standard face mask on the patient and instruct the patient not to remove it.
For hospitals accepting a suspected measles case, meet the patient outside of the facility and ensure that the patient is wearing a standard face mask. All staff interacting with the patient should practice contact and airborne precautions (N95 respirator mask). Take the patient directly to an isolation room with negative airflow. Caution pregnant staff that they should not have contact with the patient.
Which diagnostic tests should you use?
Diagnosis can be made based on serum antibody tests (measles IgM and IgG), throat or urine viral cultures, and nasopharyngeal and throat specimen polymerase chain reaction (PCR) testing. The CDC recommends obtaining a serum sample for measles IgM testing and a throat swab for PCR in all suspected cases, but local health departments vary in their specific testing recommendations. Familiarize yourself with the tests recommended by your local department of health, and where they prefer testing on outpatients to be done. Confirmed measles should be reported to your department of health.
What are considerations for community pediatric offices?
Update families in emails to call ahead if they suspect measles. This way the office can prepare a room for the family, and have the family immediately brought back without exposing staff and other families in the waiting area. It may be more prudent to examine these children at the end of the clinic day as the virus can persist for up to 2 hours on fomites and in the air. Therefore, all waiting areas and shared air spaces (including those with shared air ducts) should be cleared for 2 hours after the patient leaves.
When should you provide prophylaxis after exposure?
A patient with suspected measles does not require immediate vaccination. If it is measles, it is already too late to vaccinate. If measles is ruled out, the child should follow the standard measles vaccination guidelines.
Individuals are contagious from 4 days before to 4 days after the rash appears.
If measles is confirmed, all people who are unvaccinated or undervaccinated and were exposed to the confirmed case during the contagious period should be vaccinated within 72 hours of exposure. Infants 6 months or older may safely receive the MMR vaccine. However, infants vaccinated with MMR before their first birthday must be vaccinated again at age 12-15 months (greater than 28 days after prior vaccine) and at 4-6 years. Immunoglobulin prophylaxis should be given intramuscularly in exposed infants ages birth to less than 6 months, and in those ages 6-12 months who present beyond the 72-hour window. Unvaccinated or undervaccinated, exposed individuals at high risk for complications from measles (immunocompromised, pregnant) also should receive immunoglobulin.
What should you tell traveling families?
Several countries have large, ongoing measles outbreaks, including Israel, Ukraine, and the Philippines. Before international travel, infants 6-11 months should receive one dose of MMR vaccine, and children 12 months and older need two doses separated by at least 28 days. For unvaccinated or undervaccinated children, consider advising families to hold off travel to high-risk countries, or understand the indications to vaccinate a child upon return.
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Medical Center in Washington. She said she has no relevant financial disclosures. Dr. Emily Willner is a pediatric emergency medicine attending at Children’s National Medical Center, and an assistant professor of pediatrics and emergency medicine at George Washington University, Washington. She has no relevant financial disclosures.
The mother of an 8-month-old calls your office and is hysterical. Her daughter has had cough for a few days with high fevers and now has developed a full body rash. She is worried about measles and is on her way to your office.
We are in the middle of a measles epidemic, there’s no denying it. Measles was declared eliminated in 2000, but reported cases in the United States have been on the rise, and are now at the highest number since 2014. Five months into 2019, there have been 839 reported cases as of May 13). Measles outbreaks (defined by the Centers for Disease Control and Prevention as three or more cases) have been reported in California, Georgia, Maryland, Michigan, New Jersey, New York, and Pennsylvania. When vaccination rates fall, it is easy for measles to spread. The virus is highly contagious in nonimmune people, because of its airborne spread and its persistence in the environment for hours.
First – is it really measles?
It can be difficult to distinguish the maculopapular rash of measles from similar rashes that occur with more benign viral illnesses. Adding to the challenge, the last major measles outbreak in the United States was over 2 decades ago, and many practicing pediatricians have never seen a single case. So, what clinical features can help distinguish measles from other febrile illnesses?
The prodromal phase of measles lasts approximately 2-4 days and children have high fevers (103°-105° F), anorexia, and malaise. Conjunctivitis, coryza, and cough develop during this phase, and precede any rash. Koplik spots appear during the prodromal phase, but are not seen in all cases. These spots are 1- to 3-mm blue-white lesions on an erythematous base on the buccal mucosa, classically opposite the first molar. The spots often slough once the rash appears. The rash appears 2-4 days after the onset of fever, and is initially maculopapular and blanching. The first lesions appear on the face and neck, and the rash spreads cranial to caudal, typically sparing palms and soles. After days 3-4, the rash will no longer blanch. High fevers persist for 2-4 more days with rash, ongoing respiratory symptoms, conjunctivitis, and pharyngitis. Note that the fever will persist even with development of the rash, unlike in roseola.
It is not only important to diagnosis measles from a public health standpoint, but also because measles can have severe complications, especially in infants and children under 5 years. During the 1989-1991 outbreak, the mortality rate was 2.2 deaths per 1,000 cases (J Infect Dis. 2004 May 1. doi: 10.1086/377694).
Six percent of patients develop pneumonia, which in infants and toddlers can lead to respiratory distress or failure requiring hospitalization. Pneumonia is responsible for 60% of measles deaths, according to the CDC “Pink Book,” Epidemiology and Prevention of Vaccine-Preventable Diseases, chapter 13 on measles, 13th Ed., 2015. Ocular complications include keratitis and corneal ulceration. Measles also can cause serious neurologic complications. Encephalitis, seen in 1 per 1,000 cases, usually arises several days after the rash and may present with seizure or encephalopathy. Acute disseminated encephalomyelitis (ADEM), an inflammatory demyelinating disease of the central nervous system, occurs in approximately 1 per 1,000 cases, typically presents during the recovery phase (1-2 weeks after rash), and can have long-term sequelae. Subacute sclerosing panencephalitis (SSPE) is a progressive and fatal neurodegenerative disorder, and presents 7-10 years after measles infection.
Should you transfer the patient to a hospital?
Unless there is a medical need for the child to be admitted, sending a patient with potential measles to the hospital is not necessary, and can cause exposure to a large group of medical personnel, and patients who cannot be vaccinated (such as infants, immunocompromised patients, and pregnant women). However, if there is concern for complications such as seizures, encephalitis, or pneumonia, then transfer is indicated. Call the accepting hospital in advance so the staff can prepare for the patient. During transfer, place a standard face mask on the patient and instruct the patient not to remove it.
For hospitals accepting a suspected measles case, meet the patient outside of the facility and ensure that the patient is wearing a standard face mask. All staff interacting with the patient should practice contact and airborne precautions (N95 respirator mask). Take the patient directly to an isolation room with negative airflow. Caution pregnant staff that they should not have contact with the patient.
Which diagnostic tests should you use?
Diagnosis can be made based on serum antibody tests (measles IgM and IgG), throat or urine viral cultures, and nasopharyngeal and throat specimen polymerase chain reaction (PCR) testing. The CDC recommends obtaining a serum sample for measles IgM testing and a throat swab for PCR in all suspected cases, but local health departments vary in their specific testing recommendations. Familiarize yourself with the tests recommended by your local department of health, and where they prefer testing on outpatients to be done. Confirmed measles should be reported to your department of health.
What are considerations for community pediatric offices?
Update families in emails to call ahead if they suspect measles. This way the office can prepare a room for the family, and have the family immediately brought back without exposing staff and other families in the waiting area. It may be more prudent to examine these children at the end of the clinic day as the virus can persist for up to 2 hours on fomites and in the air. Therefore, all waiting areas and shared air spaces (including those with shared air ducts) should be cleared for 2 hours after the patient leaves.
When should you provide prophylaxis after exposure?
A patient with suspected measles does not require immediate vaccination. If it is measles, it is already too late to vaccinate. If measles is ruled out, the child should follow the standard measles vaccination guidelines.
Individuals are contagious from 4 days before to 4 days after the rash appears.
If measles is confirmed, all people who are unvaccinated or undervaccinated and were exposed to the confirmed case during the contagious period should be vaccinated within 72 hours of exposure. Infants 6 months or older may safely receive the MMR vaccine. However, infants vaccinated with MMR before their first birthday must be vaccinated again at age 12-15 months (greater than 28 days after prior vaccine) and at 4-6 years. Immunoglobulin prophylaxis should be given intramuscularly in exposed infants ages birth to less than 6 months, and in those ages 6-12 months who present beyond the 72-hour window. Unvaccinated or undervaccinated, exposed individuals at high risk for complications from measles (immunocompromised, pregnant) also should receive immunoglobulin.
What should you tell traveling families?
Several countries have large, ongoing measles outbreaks, including Israel, Ukraine, and the Philippines. Before international travel, infants 6-11 months should receive one dose of MMR vaccine, and children 12 months and older need two doses separated by at least 28 days. For unvaccinated or undervaccinated children, consider advising families to hold off travel to high-risk countries, or understand the indications to vaccinate a child upon return.
Dr. Angelica DesPain is a pediatric emergency medicine fellow at Children’s National Medical Center in Washington. She said she has no relevant financial disclosures. Dr. Emily Willner is a pediatric emergency medicine attending at Children’s National Medical Center, and an assistant professor of pediatrics and emergency medicine at George Washington University, Washington. She has no relevant financial disclosures.
Younger patients with NSCLC tend to live longer
Younger patients with non–small cell lung cancer (NSCLC) may have better survival, despite higher rates of brain metastasis and driver mutations, according to results from a retrospective analysis.
“We carried out a comprehensive analysis of patient clinicopathologic features and clinical outcomes in both young (age ≤ 50 years) and older (age > 60 years) patients with NSCLC,” wrote Anna May Suidan of Tel Aviv University, and colleagues. The findings were published in the Journal of Global Oncology.
The researchers reviewed medical records of patients who were diagnosed with lung cancer at a large cancer treatment facility in Israel from 2010 to 2015. Patients were categorized into two groups according to age at cancer diagnosis, which was established based on tumor pathology.
Various clinical data were collected, including demographic information, history of malignancy, smoking history, histologic subtype, and survival data.
In all, 62 patients were included in the younger cohort (median age, 44.5 years) and 124 patients in the older cohort (median age, 68.0 years).
After analysis, the researchers found that younger patients had a higher incidence of brain metastasis (39% vs. 25%, respectively; P = .04), and increased rates of EGFR mutations (23% vs. 18%, respectively; P = .4) and ALK translocations (13% vs. 2%, respectively; P = .002) versus older patients.
“Our cohort, which was [composed] of white patients, demonstrated that younger patients harbored more targetable driver mutations compared with older patients (34% vs. 18%; P = .01),” the researchers wrote.
In addition, among those with a driver mutation, younger patients showed a trend toward better survival (median survival, 33 vs. 25 months, respectively; P = .4).
Two key limitations of the study were the small sample size and retrospective design.
“[These results] highlight the importance of genetic background assessments and considering lung cancer as a possible diagnosis in young symptomatic patients in clinical settings,” the researchers concluded.
No funding sources were reported. The authors reported financial affiliations with Astra Zeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Novartis, Roche, Teva Pharmaceuticals, and several others.
SOURCE: Suidan AM et al. J Glob Oncol. 2019 May 8. doi: 10.1200/JGO.18.00216.
Younger patients with non–small cell lung cancer (NSCLC) may have better survival, despite higher rates of brain metastasis and driver mutations, according to results from a retrospective analysis.
“We carried out a comprehensive analysis of patient clinicopathologic features and clinical outcomes in both young (age ≤ 50 years) and older (age > 60 years) patients with NSCLC,” wrote Anna May Suidan of Tel Aviv University, and colleagues. The findings were published in the Journal of Global Oncology.
The researchers reviewed medical records of patients who were diagnosed with lung cancer at a large cancer treatment facility in Israel from 2010 to 2015. Patients were categorized into two groups according to age at cancer diagnosis, which was established based on tumor pathology.
Various clinical data were collected, including demographic information, history of malignancy, smoking history, histologic subtype, and survival data.
In all, 62 patients were included in the younger cohort (median age, 44.5 years) and 124 patients in the older cohort (median age, 68.0 years).
After analysis, the researchers found that younger patients had a higher incidence of brain metastasis (39% vs. 25%, respectively; P = .04), and increased rates of EGFR mutations (23% vs. 18%, respectively; P = .4) and ALK translocations (13% vs. 2%, respectively; P = .002) versus older patients.
“Our cohort, which was [composed] of white patients, demonstrated that younger patients harbored more targetable driver mutations compared with older patients (34% vs. 18%; P = .01),” the researchers wrote.
In addition, among those with a driver mutation, younger patients showed a trend toward better survival (median survival, 33 vs. 25 months, respectively; P = .4).
Two key limitations of the study were the small sample size and retrospective design.
“[These results] highlight the importance of genetic background assessments and considering lung cancer as a possible diagnosis in young symptomatic patients in clinical settings,” the researchers concluded.
No funding sources were reported. The authors reported financial affiliations with Astra Zeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Novartis, Roche, Teva Pharmaceuticals, and several others.
SOURCE: Suidan AM et al. J Glob Oncol. 2019 May 8. doi: 10.1200/JGO.18.00216.
Younger patients with non–small cell lung cancer (NSCLC) may have better survival, despite higher rates of brain metastasis and driver mutations, according to results from a retrospective analysis.
“We carried out a comprehensive analysis of patient clinicopathologic features and clinical outcomes in both young (age ≤ 50 years) and older (age > 60 years) patients with NSCLC,” wrote Anna May Suidan of Tel Aviv University, and colleagues. The findings were published in the Journal of Global Oncology.
The researchers reviewed medical records of patients who were diagnosed with lung cancer at a large cancer treatment facility in Israel from 2010 to 2015. Patients were categorized into two groups according to age at cancer diagnosis, which was established based on tumor pathology.
Various clinical data were collected, including demographic information, history of malignancy, smoking history, histologic subtype, and survival data.
In all, 62 patients were included in the younger cohort (median age, 44.5 years) and 124 patients in the older cohort (median age, 68.0 years).
After analysis, the researchers found that younger patients had a higher incidence of brain metastasis (39% vs. 25%, respectively; P = .04), and increased rates of EGFR mutations (23% vs. 18%, respectively; P = .4) and ALK translocations (13% vs. 2%, respectively; P = .002) versus older patients.
“Our cohort, which was [composed] of white patients, demonstrated that younger patients harbored more targetable driver mutations compared with older patients (34% vs. 18%; P = .01),” the researchers wrote.
In addition, among those with a driver mutation, younger patients showed a trend toward better survival (median survival, 33 vs. 25 months, respectively; P = .4).
Two key limitations of the study were the small sample size and retrospective design.
“[These results] highlight the importance of genetic background assessments and considering lung cancer as a possible diagnosis in young symptomatic patients in clinical settings,” the researchers concluded.
No funding sources were reported. The authors reported financial affiliations with Astra Zeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Novartis, Roche, Teva Pharmaceuticals, and several others.
SOURCE: Suidan AM et al. J Glob Oncol. 2019 May 8. doi: 10.1200/JGO.18.00216.
FROM THE JOURNAL OF GLOBAL ONCOLOGY