User login
Encephalitis linked to psychosis, suicidal thoughts
Investigators assessed 120 patients hospitalized in a neurological center and diagnosed with ANMDARE. Most had psychosis and other severe mental health disturbances. Of these, 13% also had suicidal thoughts and behaviors.
However, after medical treatment that included immunotherapy, neurologic and psychiatric pharmacotherapy, and rehabilitation and psychotherapy, almost all patients with suicidal thoughts and behaviors had sustained remission of their suicidality.
“Most patients [with ANMDARE] suffer with severe mental health problems, and it is not infrequent that suicidal thoughts and behaviors emerge in this context – mainly in patients with clinical features of psychotic depression,” senior author Jesús Ramirez-Bermúdez, MD, PhD, from the neuropsychiatry unit, National Institute of Neurology and Neurosurgery of Mexico, told this news organization.
“The good news is that, in most cases, the suicidal thoughts and behaviors as well as the features of psychotic depression improve significantly with the specific immunological therapy. However, careful psychiatric and psychotherapeutic support are helpful to restore the long-term psychological well-being,” Dr. Ramirez-Bermúdez said.
The findings were published online in the Journal of Neuropsychiatry and Clinical Neurosciences.
Delayed recognition
ANMDARE is a “frequent form of autoimmune encephalitis,” the authors write. It often begins with an “abrupt onset of behavioral and cognitive symptoms, followed by seizures and movement disorders,” they add.
“The clinical care of persons with encephalitis is challenging because these patients suffer from acute and severe mental health disturbances [and] are often misdiagnosed as having a primary psychiatric disorder, for instance, schizophrenia or bipolar disorder; but, they do not improve with the use of psychiatric medication or psychotherapy,” Dr. Ramirez-Bermúdez said.
Rather, the disease requires specific treatments, such as the use of antiviral medication or immunotherapy, he added. Without these, “the mortality rate is high, and many patients have bad outcomes, including disability related to cognitive and affective disturbances,” he said.
Dr. Ramirez-Bermúdez noted that there are “many cultural problems in the conventional approach to mental health problems, including prejudices, fear, myths, stigma, and discrimination.” And these attitudes can contribute to delayed recognition of ANMDARE.
During recent years, Dr. Ramirez-Bermúdez and colleagues observed that some patients with autoimmune encephalitis and, more specifically, patients suffering from ANMDARE had suicidal behavior. A previous study conducted in China suggested that the problem of suicidal behavior is not infrequent in this population.
“We wanted to make a structured, systematic, and prospective approach to this problem to answer some questions related to ANMDARE,” Dr. Ramirez-Bermúdez said. These questions included: What is the frequency of suicidal thoughts and behaviors, what are the neurological and psychiatric features related to suicidal behavior in this population, and what is the outcome after receiving immunological treatment?
The researchers conducted an observational longitudinal study that included patients hospitalized between 2014 and 2021 who had definite ANMDARE (n = 120).
Patients were diagnosed as having encephalitis by means of clinical interviews, neuropsychological studies, brain imaging, EEG, and analysis of cerebrospinal fluid (CSF).
All participants had antibodies against the NMDA glutamate receptor in their CSF and were classified as having ANMDARE based on Graus criteria, “which are considered the best current standard for diagnosis,” Dr. Ramirez-Bermúdez noted.
Clinical measures were obtained both before and after treatment with immunotherapy, and all clinical data were registered prospectively and included a “broad scope of neurological and psychiatric variables seen in patients with ANMDARE.”
Information regarding suicidal thoughts and behaviors was gathered from patients as well as relatives, with assessments occurring at admission and at discharge.
Biological signaling
Results showed that 15 patients presented with suicidal thoughts and/or behaviors. Of this subgroup, the median age was 32 years (range, 19-48 years) and 53.3% were women.
All members of this subgroup had psychotic features, including persecutory, grandiose, nihilistic, or jealousy delusion (n = 14), delirium (n = 13), visual or auditory hallucinations (n = 11), psychotic depression (n = 10), and/or catatonia (n = 8).
Most (n = 12) had suicidal ideation with intent, three had preparatory behaviors, and seven actually engaged in suicidal self-directed violence.
Of these 15 patients, 7 had abnormal CSF findings, 8 had MRI abnormalities involving the medial temporal lobe, and all had abnormal EEG involving generalized slowing.
Fourteen suicidal patients were treated with an antipsychotic, 4 with dexmedetomidine, and 12 with lorazepam. In addition, 10 received plasmapheresis and 7 received immunoglobulin.
Of note, at discharge, self-directed violent thoughts and behaviors completely remitted in 14 of the 15 patients. Long-term follow-up showed that they remained free of suicidality.
Dr. Ramirez-Bermúdez noted that in some patients with neuropsychiatric disturbances, “there are autoantibodies against the NR1 subunit of the NMDA glutamate receptor: the main excitatory neurotransmitter in the human brain.”
The NMDA receptor is “particularly important as part of the biological signaling that is required in several cognitive and affective processes leading to complex behaviors,” he said. NMDA receptor dysfunction “may lead to states in which these cognitive and affective processes are disturbed,” frequently resulting in psychosis.
Study coauthor Ava Easton, MD, chief executive of the Encephalitis Society, told this news organization that mental health issues, self-injurious thoughts, and suicidal behaviors after encephalitis “may occur for a number of reasons and stigma around talking about mental health can be a real barrier to speaking up about symptoms; but it is an important barrier to overcome.”
Dr. Easton, an honorary fellow in the department of clinical infection, microbiology, and immunology, University of Liverpool, England, added that their study “provides a platform on which to break taboo, show tangible links which are based on data between suicide and encephalitis, and call for more awareness of the risk of mental health issues during and after encephalitis.”
‘Neglected symptom’
Commenting on the study, Carsten Finke, MD, Heisenberg Professor for Cognitive Neurology and consultant neurologist, department of neurology at Charité, Berlin, and professor at Berlin School of Mind and Brain, said that the research was on “a very important topic on a so far rather neglected symptom of encephalitis.”
Dr. Finke, a founding member of the scientific council of the German Network for Research on Autoimmune Encephalitis, was not involved in the current study.
He noted that 77% of people don’t know what encephalitis is. “This lack of awareness leads to delays in diagnoses and treatment – and poorer outcomes for patients,” Dr. Finke said.
Also commenting, Michael Eriksen Benros, MD, PhD, professor of immune-psychiatry, department of immunology and microbiology, Health and Medical Sciences, University of Copenhagen, said that the study “underlines the clinical importance of screening individuals with psychotic symptoms for suicidal ideations during acute phases,” as well as those with definite ANMDARE as a likely underlying cause of the psychotic symptoms.
This is important because patients with ANMDARE “might not necessarily be admitted at psychiatric departments where screenings for suicidal ideation are part of the clinical routine,” said Dr. Benros, who was not involved with the research.
Instead, “many patients with ANMDARE are at neurological departments during acute phases,” he added.
The study was supported by the National Council of Science and Technology of Mexico. Dr. Ramirez-Bermúdez, Dr. Easton, Dr. Benros, and Dr. Finke report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators assessed 120 patients hospitalized in a neurological center and diagnosed with ANMDARE. Most had psychosis and other severe mental health disturbances. Of these, 13% also had suicidal thoughts and behaviors.
However, after medical treatment that included immunotherapy, neurologic and psychiatric pharmacotherapy, and rehabilitation and psychotherapy, almost all patients with suicidal thoughts and behaviors had sustained remission of their suicidality.
“Most patients [with ANMDARE] suffer with severe mental health problems, and it is not infrequent that suicidal thoughts and behaviors emerge in this context – mainly in patients with clinical features of psychotic depression,” senior author Jesús Ramirez-Bermúdez, MD, PhD, from the neuropsychiatry unit, National Institute of Neurology and Neurosurgery of Mexico, told this news organization.
“The good news is that, in most cases, the suicidal thoughts and behaviors as well as the features of psychotic depression improve significantly with the specific immunological therapy. However, careful psychiatric and psychotherapeutic support are helpful to restore the long-term psychological well-being,” Dr. Ramirez-Bermúdez said.
The findings were published online in the Journal of Neuropsychiatry and Clinical Neurosciences.
Delayed recognition
ANMDARE is a “frequent form of autoimmune encephalitis,” the authors write. It often begins with an “abrupt onset of behavioral and cognitive symptoms, followed by seizures and movement disorders,” they add.
“The clinical care of persons with encephalitis is challenging because these patients suffer from acute and severe mental health disturbances [and] are often misdiagnosed as having a primary psychiatric disorder, for instance, schizophrenia or bipolar disorder; but, they do not improve with the use of psychiatric medication or psychotherapy,” Dr. Ramirez-Bermúdez said.
Rather, the disease requires specific treatments, such as the use of antiviral medication or immunotherapy, he added. Without these, “the mortality rate is high, and many patients have bad outcomes, including disability related to cognitive and affective disturbances,” he said.
Dr. Ramirez-Bermúdez noted that there are “many cultural problems in the conventional approach to mental health problems, including prejudices, fear, myths, stigma, and discrimination.” And these attitudes can contribute to delayed recognition of ANMDARE.
During recent years, Dr. Ramirez-Bermúdez and colleagues observed that some patients with autoimmune encephalitis and, more specifically, patients suffering from ANMDARE had suicidal behavior. A previous study conducted in China suggested that the problem of suicidal behavior is not infrequent in this population.
“We wanted to make a structured, systematic, and prospective approach to this problem to answer some questions related to ANMDARE,” Dr. Ramirez-Bermúdez said. These questions included: What is the frequency of suicidal thoughts and behaviors, what are the neurological and psychiatric features related to suicidal behavior in this population, and what is the outcome after receiving immunological treatment?
The researchers conducted an observational longitudinal study that included patients hospitalized between 2014 and 2021 who had definite ANMDARE (n = 120).
Patients were diagnosed as having encephalitis by means of clinical interviews, neuropsychological studies, brain imaging, EEG, and analysis of cerebrospinal fluid (CSF).
All participants had antibodies against the NMDA glutamate receptor in their CSF and were classified as having ANMDARE based on Graus criteria, “which are considered the best current standard for diagnosis,” Dr. Ramirez-Bermúdez noted.
Clinical measures were obtained both before and after treatment with immunotherapy, and all clinical data were registered prospectively and included a “broad scope of neurological and psychiatric variables seen in patients with ANMDARE.”
Information regarding suicidal thoughts and behaviors was gathered from patients as well as relatives, with assessments occurring at admission and at discharge.
Biological signaling
Results showed that 15 patients presented with suicidal thoughts and/or behaviors. Of this subgroup, the median age was 32 years (range, 19-48 years) and 53.3% were women.
All members of this subgroup had psychotic features, including persecutory, grandiose, nihilistic, or jealousy delusion (n = 14), delirium (n = 13), visual or auditory hallucinations (n = 11), psychotic depression (n = 10), and/or catatonia (n = 8).
Most (n = 12) had suicidal ideation with intent, three had preparatory behaviors, and seven actually engaged in suicidal self-directed violence.
Of these 15 patients, 7 had abnormal CSF findings, 8 had MRI abnormalities involving the medial temporal lobe, and all had abnormal EEG involving generalized slowing.
Fourteen suicidal patients were treated with an antipsychotic, 4 with dexmedetomidine, and 12 with lorazepam. In addition, 10 received plasmapheresis and 7 received immunoglobulin.
Of note, at discharge, self-directed violent thoughts and behaviors completely remitted in 14 of the 15 patients. Long-term follow-up showed that they remained free of suicidality.
Dr. Ramirez-Bermúdez noted that in some patients with neuropsychiatric disturbances, “there are autoantibodies against the NR1 subunit of the NMDA glutamate receptor: the main excitatory neurotransmitter in the human brain.”
The NMDA receptor is “particularly important as part of the biological signaling that is required in several cognitive and affective processes leading to complex behaviors,” he said. NMDA receptor dysfunction “may lead to states in which these cognitive and affective processes are disturbed,” frequently resulting in psychosis.
Study coauthor Ava Easton, MD, chief executive of the Encephalitis Society, told this news organization that mental health issues, self-injurious thoughts, and suicidal behaviors after encephalitis “may occur for a number of reasons and stigma around talking about mental health can be a real barrier to speaking up about symptoms; but it is an important barrier to overcome.”
Dr. Easton, an honorary fellow in the department of clinical infection, microbiology, and immunology, University of Liverpool, England, added that their study “provides a platform on which to break taboo, show tangible links which are based on data between suicide and encephalitis, and call for more awareness of the risk of mental health issues during and after encephalitis.”
‘Neglected symptom’
Commenting on the study, Carsten Finke, MD, Heisenberg Professor for Cognitive Neurology and consultant neurologist, department of neurology at Charité, Berlin, and professor at Berlin School of Mind and Brain, said that the research was on “a very important topic on a so far rather neglected symptom of encephalitis.”
Dr. Finke, a founding member of the scientific council of the German Network for Research on Autoimmune Encephalitis, was not involved in the current study.
He noted that 77% of people don’t know what encephalitis is. “This lack of awareness leads to delays in diagnoses and treatment – and poorer outcomes for patients,” Dr. Finke said.
Also commenting, Michael Eriksen Benros, MD, PhD, professor of immune-psychiatry, department of immunology and microbiology, Health and Medical Sciences, University of Copenhagen, said that the study “underlines the clinical importance of screening individuals with psychotic symptoms for suicidal ideations during acute phases,” as well as those with definite ANMDARE as a likely underlying cause of the psychotic symptoms.
This is important because patients with ANMDARE “might not necessarily be admitted at psychiatric departments where screenings for suicidal ideation are part of the clinical routine,” said Dr. Benros, who was not involved with the research.
Instead, “many patients with ANMDARE are at neurological departments during acute phases,” he added.
The study was supported by the National Council of Science and Technology of Mexico. Dr. Ramirez-Bermúdez, Dr. Easton, Dr. Benros, and Dr. Finke report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators assessed 120 patients hospitalized in a neurological center and diagnosed with ANMDARE. Most had psychosis and other severe mental health disturbances. Of these, 13% also had suicidal thoughts and behaviors.
However, after medical treatment that included immunotherapy, neurologic and psychiatric pharmacotherapy, and rehabilitation and psychotherapy, almost all patients with suicidal thoughts and behaviors had sustained remission of their suicidality.
“Most patients [with ANMDARE] suffer with severe mental health problems, and it is not infrequent that suicidal thoughts and behaviors emerge in this context – mainly in patients with clinical features of psychotic depression,” senior author Jesús Ramirez-Bermúdez, MD, PhD, from the neuropsychiatry unit, National Institute of Neurology and Neurosurgery of Mexico, told this news organization.
“The good news is that, in most cases, the suicidal thoughts and behaviors as well as the features of psychotic depression improve significantly with the specific immunological therapy. However, careful psychiatric and psychotherapeutic support are helpful to restore the long-term psychological well-being,” Dr. Ramirez-Bermúdez said.
The findings were published online in the Journal of Neuropsychiatry and Clinical Neurosciences.
Delayed recognition
ANMDARE is a “frequent form of autoimmune encephalitis,” the authors write. It often begins with an “abrupt onset of behavioral and cognitive symptoms, followed by seizures and movement disorders,” they add.
“The clinical care of persons with encephalitis is challenging because these patients suffer from acute and severe mental health disturbances [and] are often misdiagnosed as having a primary psychiatric disorder, for instance, schizophrenia or bipolar disorder; but, they do not improve with the use of psychiatric medication or psychotherapy,” Dr. Ramirez-Bermúdez said.
Rather, the disease requires specific treatments, such as the use of antiviral medication or immunotherapy, he added. Without these, “the mortality rate is high, and many patients have bad outcomes, including disability related to cognitive and affective disturbances,” he said.
Dr. Ramirez-Bermúdez noted that there are “many cultural problems in the conventional approach to mental health problems, including prejudices, fear, myths, stigma, and discrimination.” And these attitudes can contribute to delayed recognition of ANMDARE.
During recent years, Dr. Ramirez-Bermúdez and colleagues observed that some patients with autoimmune encephalitis and, more specifically, patients suffering from ANMDARE had suicidal behavior. A previous study conducted in China suggested that the problem of suicidal behavior is not infrequent in this population.
“We wanted to make a structured, systematic, and prospective approach to this problem to answer some questions related to ANMDARE,” Dr. Ramirez-Bermúdez said. These questions included: What is the frequency of suicidal thoughts and behaviors, what are the neurological and psychiatric features related to suicidal behavior in this population, and what is the outcome after receiving immunological treatment?
The researchers conducted an observational longitudinal study that included patients hospitalized between 2014 and 2021 who had definite ANMDARE (n = 120).
Patients were diagnosed as having encephalitis by means of clinical interviews, neuropsychological studies, brain imaging, EEG, and analysis of cerebrospinal fluid (CSF).
All participants had antibodies against the NMDA glutamate receptor in their CSF and were classified as having ANMDARE based on Graus criteria, “which are considered the best current standard for diagnosis,” Dr. Ramirez-Bermúdez noted.
Clinical measures were obtained both before and after treatment with immunotherapy, and all clinical data were registered prospectively and included a “broad scope of neurological and psychiatric variables seen in patients with ANMDARE.”
Information regarding suicidal thoughts and behaviors was gathered from patients as well as relatives, with assessments occurring at admission and at discharge.
Biological signaling
Results showed that 15 patients presented with suicidal thoughts and/or behaviors. Of this subgroup, the median age was 32 years (range, 19-48 years) and 53.3% were women.
All members of this subgroup had psychotic features, including persecutory, grandiose, nihilistic, or jealousy delusion (n = 14), delirium (n = 13), visual or auditory hallucinations (n = 11), psychotic depression (n = 10), and/or catatonia (n = 8).
Most (n = 12) had suicidal ideation with intent, three had preparatory behaviors, and seven actually engaged in suicidal self-directed violence.
Of these 15 patients, 7 had abnormal CSF findings, 8 had MRI abnormalities involving the medial temporal lobe, and all had abnormal EEG involving generalized slowing.
Fourteen suicidal patients were treated with an antipsychotic, 4 with dexmedetomidine, and 12 with lorazepam. In addition, 10 received plasmapheresis and 7 received immunoglobulin.
Of note, at discharge, self-directed violent thoughts and behaviors completely remitted in 14 of the 15 patients. Long-term follow-up showed that they remained free of suicidality.
Dr. Ramirez-Bermúdez noted that in some patients with neuropsychiatric disturbances, “there are autoantibodies against the NR1 subunit of the NMDA glutamate receptor: the main excitatory neurotransmitter in the human brain.”
The NMDA receptor is “particularly important as part of the biological signaling that is required in several cognitive and affective processes leading to complex behaviors,” he said. NMDA receptor dysfunction “may lead to states in which these cognitive and affective processes are disturbed,” frequently resulting in psychosis.
Study coauthor Ava Easton, MD, chief executive of the Encephalitis Society, told this news organization that mental health issues, self-injurious thoughts, and suicidal behaviors after encephalitis “may occur for a number of reasons and stigma around talking about mental health can be a real barrier to speaking up about symptoms; but it is an important barrier to overcome.”
Dr. Easton, an honorary fellow in the department of clinical infection, microbiology, and immunology, University of Liverpool, England, added that their study “provides a platform on which to break taboo, show tangible links which are based on data between suicide and encephalitis, and call for more awareness of the risk of mental health issues during and after encephalitis.”
‘Neglected symptom’
Commenting on the study, Carsten Finke, MD, Heisenberg Professor for Cognitive Neurology and consultant neurologist, department of neurology at Charité, Berlin, and professor at Berlin School of Mind and Brain, said that the research was on “a very important topic on a so far rather neglected symptom of encephalitis.”
Dr. Finke, a founding member of the scientific council of the German Network for Research on Autoimmune Encephalitis, was not involved in the current study.
He noted that 77% of people don’t know what encephalitis is. “This lack of awareness leads to delays in diagnoses and treatment – and poorer outcomes for patients,” Dr. Finke said.
Also commenting, Michael Eriksen Benros, MD, PhD, professor of immune-psychiatry, department of immunology and microbiology, Health and Medical Sciences, University of Copenhagen, said that the study “underlines the clinical importance of screening individuals with psychotic symptoms for suicidal ideations during acute phases,” as well as those with definite ANMDARE as a likely underlying cause of the psychotic symptoms.
This is important because patients with ANMDARE “might not necessarily be admitted at psychiatric departments where screenings for suicidal ideation are part of the clinical routine,” said Dr. Benros, who was not involved with the research.
Instead, “many patients with ANMDARE are at neurological departments during acute phases,” he added.
The study was supported by the National Council of Science and Technology of Mexico. Dr. Ramirez-Bermúdez, Dr. Easton, Dr. Benros, and Dr. Finke report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF NEUROPSYCHIATRY AND CLINICAL NEUROSCIENCES
Thyroid hormones predict psychotic depression in MDD patients
Thyroid dysfunction is common among major depressive disorder (MDD) patients, but its relationship with the psychotic depression (PD) subtype has not been well studied, wrote Pu Peng, of The Second Xiangya Hospital of Central South University, Changsha, Hunan, China, and colleagues.
Given the significant negative consequences of PD in MDD, including comorbid psychosis, suicidal attempts, and worse prognosis, more ways to identify PD risk factors in MDD are needed, they said. Previous research suggests a role for thyroid hormones in the pathophysiology of PD, but data on specific associations are limited, they noted.
In a study published in Psychiatry Research, the authors recruited 1,718 adults aged 18-60 years with MDD who were treated at a single center. The median age was 34 years, 66% were female, and 10% were identified with PD.
Clinical symptoms were identified using the positive subscale of the Positive and Negative Symptom Scale (PANSS-P), Hamilton Anxiety Rating Scale (HAMA), and Hamilton Depression Rating Scale (HAMD). The median PANSS-P score was 7. The researchers measured serum levels of thyroid stimulating hormone (TSH), anti-thyroglobulin (TgAb), and thyroid peroxidases antibody (TPOAb). Subclinical hyperthyroidism (SCH) was defined as TSH levels greater than 8.0 uIU/L and FT4 within normal values.
Overall, the prevalence of SCH, abnormal TgAb, TPOAb, FT3, and FT4 were 13%, 17%, 25%, <0.1%, and 0.3%, respectively. Serum TSH levels, TgAb levels, and TPOAb levels were significantly higher in PD patients than in non-PD patients. No differences appeared in FT3 and FT4 levels between the two groups.
In a multivariate analysis, subclinical hypothyroidism was associated with a ninefold increased risk of PD (odds ratio, 9.32) as were abnormal TPOAb (OR, 1.89) and abnormal TgAb (OR, 2.09).
The findings were limited by several factors including the cross-sectional design, and the inclusion of participants from only a single center in China, which may limit generalizability, the researchers noted.
In addition, “It should be noted that the association between thyroid hormones and PD was small to moderate and the underlying mechanism remained unexplored,” they said. Other limitations include the use of only 17 of the 20 HAMD items and the lack of data on the relationship between anxiety and depressive features and thyroid dysfunction, they wrote.
More research is needed to confirm the findings in other populations, however; the results suggest that regular thyroid function tests may help with early detection of PD in MDD patients, they concluded.
The study was funded by the CAS Pioneer Hundred Talents Program and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
Thyroid dysfunction is common among major depressive disorder (MDD) patients, but its relationship with the psychotic depression (PD) subtype has not been well studied, wrote Pu Peng, of The Second Xiangya Hospital of Central South University, Changsha, Hunan, China, and colleagues.
Given the significant negative consequences of PD in MDD, including comorbid psychosis, suicidal attempts, and worse prognosis, more ways to identify PD risk factors in MDD are needed, they said. Previous research suggests a role for thyroid hormones in the pathophysiology of PD, but data on specific associations are limited, they noted.
In a study published in Psychiatry Research, the authors recruited 1,718 adults aged 18-60 years with MDD who were treated at a single center. The median age was 34 years, 66% were female, and 10% were identified with PD.
Clinical symptoms were identified using the positive subscale of the Positive and Negative Symptom Scale (PANSS-P), Hamilton Anxiety Rating Scale (HAMA), and Hamilton Depression Rating Scale (HAMD). The median PANSS-P score was 7. The researchers measured serum levels of thyroid stimulating hormone (TSH), anti-thyroglobulin (TgAb), and thyroid peroxidases antibody (TPOAb). Subclinical hyperthyroidism (SCH) was defined as TSH levels greater than 8.0 uIU/L and FT4 within normal values.
Overall, the prevalence of SCH, abnormal TgAb, TPOAb, FT3, and FT4 were 13%, 17%, 25%, <0.1%, and 0.3%, respectively. Serum TSH levels, TgAb levels, and TPOAb levels were significantly higher in PD patients than in non-PD patients. No differences appeared in FT3 and FT4 levels between the two groups.
In a multivariate analysis, subclinical hypothyroidism was associated with a ninefold increased risk of PD (odds ratio, 9.32) as were abnormal TPOAb (OR, 1.89) and abnormal TgAb (OR, 2.09).
The findings were limited by several factors including the cross-sectional design, and the inclusion of participants from only a single center in China, which may limit generalizability, the researchers noted.
In addition, “It should be noted that the association between thyroid hormones and PD was small to moderate and the underlying mechanism remained unexplored,” they said. Other limitations include the use of only 17 of the 20 HAMD items and the lack of data on the relationship between anxiety and depressive features and thyroid dysfunction, they wrote.
More research is needed to confirm the findings in other populations, however; the results suggest that regular thyroid function tests may help with early detection of PD in MDD patients, they concluded.
The study was funded by the CAS Pioneer Hundred Talents Program and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
Thyroid dysfunction is common among major depressive disorder (MDD) patients, but its relationship with the psychotic depression (PD) subtype has not been well studied, wrote Pu Peng, of The Second Xiangya Hospital of Central South University, Changsha, Hunan, China, and colleagues.
Given the significant negative consequences of PD in MDD, including comorbid psychosis, suicidal attempts, and worse prognosis, more ways to identify PD risk factors in MDD are needed, they said. Previous research suggests a role for thyroid hormones in the pathophysiology of PD, but data on specific associations are limited, they noted.
In a study published in Psychiatry Research, the authors recruited 1,718 adults aged 18-60 years with MDD who were treated at a single center. The median age was 34 years, 66% were female, and 10% were identified with PD.
Clinical symptoms were identified using the positive subscale of the Positive and Negative Symptom Scale (PANSS-P), Hamilton Anxiety Rating Scale (HAMA), and Hamilton Depression Rating Scale (HAMD). The median PANSS-P score was 7. The researchers measured serum levels of thyroid stimulating hormone (TSH), anti-thyroglobulin (TgAb), and thyroid peroxidases antibody (TPOAb). Subclinical hyperthyroidism (SCH) was defined as TSH levels greater than 8.0 uIU/L and FT4 within normal values.
Overall, the prevalence of SCH, abnormal TgAb, TPOAb, FT3, and FT4 were 13%, 17%, 25%, <0.1%, and 0.3%, respectively. Serum TSH levels, TgAb levels, and TPOAb levels were significantly higher in PD patients than in non-PD patients. No differences appeared in FT3 and FT4 levels between the two groups.
In a multivariate analysis, subclinical hypothyroidism was associated with a ninefold increased risk of PD (odds ratio, 9.32) as were abnormal TPOAb (OR, 1.89) and abnormal TgAb (OR, 2.09).
The findings were limited by several factors including the cross-sectional design, and the inclusion of participants from only a single center in China, which may limit generalizability, the researchers noted.
In addition, “It should be noted that the association between thyroid hormones and PD was small to moderate and the underlying mechanism remained unexplored,” they said. Other limitations include the use of only 17 of the 20 HAMD items and the lack of data on the relationship between anxiety and depressive features and thyroid dysfunction, they wrote.
More research is needed to confirm the findings in other populations, however; the results suggest that regular thyroid function tests may help with early detection of PD in MDD patients, they concluded.
The study was funded by the CAS Pioneer Hundred Talents Program and the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose.
FROM PSYCHIATRY RESEARCH
Visual hallucinations: Differentiating psychiatric and neurologic causes
A visual hallucination is a visual percept experienced when awake that is not elicited by an external stimulus. Historically, hallucinations have been synonymous with psychiatric disease, most notably schizophrenia; however, over recent decades, hallucinations have been categorized based on their underlying etiology as psychodynamic (primary psychiatric), psychophysiologic (primary neurologic/structural), and psychobiochemical (neurotransmitter dysfunction).1 Presently, visual hallucinations are known to be caused by a wide variety of primary psychiatric, neurologic, ophthalmologic, and chemically-mediated conditions. Despite these causes, clinically differentiating the characteristics and qualities of visual hallucinations is often a lesser-known skillset among clinicians. The utility of this skillset is important for the clinician’s ability to differentiate the expected and unexpected characteristics of visual hallucinations in patients with both known and unknown neuropsychiatric conditions.
Though many primary psychiatric and neurologic conditions have been associated with and/or known to cause visual hallucinations, this review focuses on the following grouped causes:
- Primary psychiatric causes: psychiatric disorders with psychotic features and delirium; and
- Primary neurologic causes: neurodegenerative disease/dementias, seizure disorders, migraine disorders, vision loss, peduncular hallucinosis, and hypnagogic/hypnopompic phenomena.
Because the accepted definition of visual hallucinations excludes visual percepts elicited by external stimuli, drug-induced hallucinations would not qualify for either of these categories. Additionally, most studies reporting on the effects of drug-induced hallucinations did not control for underlying comorbid psychiatric conditions, dementia, or delirium, and thus the results cannot be attributed to the drug alone, nor is it possible to identify reliable trends in the properties of the hallucinations.2 The goals of this review are to characterize visual hallucinations experienced as a result of primary psychiatric and primary neurologic conditions and describe key grouping and differentiating features to help guide the diagnosis.
Visual hallucinations in the general population
A review of 6 studies (N = 42,519) reported that the prevalence of visual hallucinations in the general population is 7.3%.3 The prevalence decreases to 6% when visual hallucinations arising from physical illness or drug/chemical consumption are excluded. The prevalence of visual hallucinations in the general population has been associated with comorbid anxiety, stress, bereavement, and psychotic pathology.4,5 Regarding the age of occurrence of visual hallucinations in the general population, there appears to be a bimodal distribution.3 One peak appears in later adolescence and early adulthood, which corresponds with higher rates of psychosis, and another peak occurs late in life, which corresponds to a higher prevalence of neurodegenerative conditions and visual impairment.
Primary psychiatric causes
Most studies of visual hallucinations in primary psychiatric conditions have specifically evaluated patients with schizophrenia and mood disorders with psychotic features.6,7 In a review of 29 studies (N = 5,873) that specifically examined visual hallucinations in individuals diagnosed with schizophrenia, Waters et al3 found a wide range of reported prevalence (4% to 65%) and a weighted mean prevalence of 27%. In contrast, the prevalence of auditory hallucinations in these participants ranged from 25% to 86%, with a weighted mean of 59%.3
Hallucinations are a known but less common symptom of mood disorders that present with psychotic features.8 Waters et al3 also examined the prevalence of visual and auditory hallucinations in mood disorders (including mania, bipolar disorder, and depression) reported in 12 studies (N = 2,892).3 They found the prevalence of visual hallucinations in patients with mood disorders ranged from 6% to 27%, with a weighted mean of 15%, compared to the weighted mean of 28% who experienced auditory hallucinations. Visual hallucinations in primary psychiatric conditions are associated with more severe disease, longer hospitalizations, and poorer prognoses.9-11
Visual hallucinations of psychosis
In patients with psychotic symptoms, the characteristics of the visually hallucinated entity as well as the cognitive and emotional perception of the hallucinations are notably different than in patients with other, nonpsychiatric causes of visual hallucations.3
Continue to: Content and perceived physical properties
Content and perceived physical properties. Hallucinated entities are most often perceived as solid, 3-dimensional, well-detailed, life-sized people, animals, and objects (often fire) or events existing in the real world.3 The entity is almost always perceived as real, with accurate form and color, fine edges, and shadow; is often out of reach of the perceiver; and can be stationary or moving within the physical properties of the external environment.3
Timing and triggers. The temporal properties vary widely. Hallucinations can last from seconds to minutes and occur at any time of day, though by definition, they must occur while the individual is awake.3 Visual hallucinations in psychosis are more common during times of acute stress, strong emotions, and tiredness.3
Patient reaction and belief. Because of realistic qualities of the visual hallucination and the perception that it is real, patients commonly attempt to participate in some activity in relation to the hallucination, such as moving away from or attempting to interact with it.3 Additionally, patients usually perceive the hallucinated entity as uncontrollable, and are surprised when the entity appears or disappears. Though the content of the hallucination is usually impersonal, the meaning the patient attributes to the presence of the hallucinated entity is usually perceived as very personal and often requiring action. The hallucination may represent a harbinger, sign, or omen, and is often interpreted religiously or spiritually and accompanied by comorbid delusions.3
Visual hallucinations of delirium
Delirium is a syndrome of altered mentation—most notably consciousness, attention, and orientation—that occurs as a result of ≥1 metabolic, infectious, drug-induced, or other medical conditions and often manifests as an acute secondary psychotic illness.12 Multiple patient and environmental characteristics have been identified as risk factors for developing delirium, including multiple and/or severe medical illnesses, preexisting dementia, depression, advanced age, polypharmacy, having an indwelling urinary catheter, impaired sight or hearing, and low albumin levels.13-15 The development of delirium is significantly and positively associated with regular alcohol use, benzodiazepine withdrawal, and angiotensin receptor blocker and dopamine receptor agonist usage.15 Approximately 40% of patients with delirium have symptoms of psychosis, and in contrast to the hallucinations experienced by patients with schizophrenia, visual hallucinations are the most common type of hallucinations seen in delirium (27%).13 In a 2021 review that included 602 patients with delirium, Tachibana et al15 found that approximately 26% experienced hallucinations, 92% of which were visual hallucinations.
Content, perceived physical properties, and reaction. Because of the limited attention and cognitive function of patients with delirium, less is known about the content of their visual hallucinations. However, much like those with primary psychotic symptoms, patients with delirium often report seeing complex, normal-sized, concrete entities, most commonly people. Tachibana et al15 found that the hallucinated person is more often a stranger than a familiar person, but (rarely) may be an ethereal being such as a devil or ghost. The next most common visually hallucinated entities were creatures, most frequently insects and animals. Other common hallucinations were visions of events or objects, such as fires, falling ceilings, or water. Similar to those with primary psychotic illness such as schizophrenia, patients with delirium often experience emotional distress, anxiety, fear, and confusion in response to the hallucinated person, object, and/or event.15
Continue to: Primary neurologic causes
Primary neurologic causes
Visual hallucinations in neurodegenerative diseases
Patients with neurodegenerative diseases such as Parkinson disease (PD), dementia with Lewy bodies (DLB), or Creutzfeldt-Jakob disease (CJD) commonly experience hallucinations as a feature of their condition. However, the true cause of these hallucinations often cannot be directly attributed to any specific pathophysiology because these patients often have multiple coexisting risk factors, such as advanced age, major depressive disorder, use of neuroactive medications, and co-occurring somatic illness. Though the prevalence of visual hallucinations varies widely between studies, with 15% to 40% reported in patients with PD, the prevalence roughly doubles in patients with PD-associated dementia (30% to 60%), and is reported by 60% to 90% of those with DLB.16-18 Hallucinations are generally thought to be less common in Alzheimer disease; such patients most commonly experience visual hallucinations, although the reported prevalence ranges widely (4% to 59%).19,20 Notably, similarly to hallucinations experienced in patients with delirium, and in contrast to those with psychosis, visual hallucinations are more common than auditory hallucinations in neurodegenerative diseases.20 Hallucinations are not common in individuals with CJD but are a key defining feature of the He
Content, perceived physical properties, and reaction. Similar to the visual hallucinations experienced by patients with psychosis or delirium, those experienced in patients with PD, DLB, or CJD are often complex, most commonly of people, followed by animals and objects. The presence of “passage hallucinations”—in which a person or animal is seen in a patient’s peripheral vision, but passes out of their visual field before the entity can be directly visualized—is common.20 Those with PD also commonly have visual hallucinations in which the form of an object appears distorted (dysmorphopsia) or the color of an object appears distorted (metachromatopsia), though these would better be classified as illusions because a real object is being perceived with distortion.22
Hallucinations are more common in the evening and at night. “Presence hallucinations” are a common type of hallucination that cannot be directly related to a specific sensory modality such as vision, though they are commonly described by patients with PD as a seen or perceived image (usually a person) that is not directly in the individual’s visual field.17 These presence hallucinations are often described as being behind the patient or in a visualized scene of what was about to happen. Before developing the dementia and myoclonus also seen in sporadic CJD, patients with the Heidenhain variant of CJD describe illusions such as metachromatopsia, dysmorphia, and micropsia that eventually develop into frank visual hallucinations, which have been poorly reported in medical literature.22,23 There are no generalizable trends in the temporal nature of visual hallucinations in patients with neurodegenerative diseases. In most cases of visual hallucinations in patients with PD and dementia, insight relating to the perception varies widely based on the patient’s cognitive status. Subsequently, patients’ reactions to the hallucinations also vary widely.
Visual hallucinations in epileptic seizures
Occipital lobe epilepsies represent 1% to 4.6% of all epilepsies; however, these represent 20% to 30% of benign childhood partial epilepsies.24,25 These are commonly associated with various types of visual hallucinations depending upon the location of the seizure onset within the occipital lobe. These are referred to as visual auras.26 Visual auras are classified into simple visual hallucinations, complex visual hallucinations, visual illusions, and ictal amaurosis (hemifield blindness or complete blindness).
Content, perceived physical properties, and reaction. Simple visual hallucinations are often described as brief, stereotypical flashing lights of various shapes and colors. These images may flicker, change shape, or take on a geometric or irregular pattern. Appearances can be repetitive and stereotyped, are often reported as moving horizontally from the periphery to the center of the visual field, and can spread to the entire visual field. Most often, these hallucinations occur for 5 to 30 seconds, and have no discernible provoking factors. Complex visual hallucinations consist of formed images of animals, people, or elaborate scenes. These are believed to reflect activation of a larger area of cortex in the temporo-parieto-occipital region, which is the visual association cortex. Very rarely, occipital lobe seizures can manifest with ictal amaurosis.24
Continue to: Simple visual auras...
Simple visual auras have a very high localizing value to the occipital lobe. The primary visual cortex (Brodmann area 17) is situated in the banks of calcarine fissure and activation of this region produces these simple hallucinations. If the hallucinations are consistently lateralized, the seizures are very likely to be coming from the contralateral occipital lobe.
Visual hallucinations in brain tumors
In general, a tumor anywhere along the optic path can produce visual hallucinations; however, the exact causal mechanism of the hallucinations is unknown. Moreover, tumors in different locations—namely the occipital lobes, temporal lobes, and frontal lobes—appear to produce visual hallucinations with substantially different characteristics.27-29 Further complicating the search for the mechanism of these hallucinations is the fact that tumors are epileptogenic. In addition, 36% to 48% of patients with brain tumors have mood symptoms (depression/mania), and 22% to 24% have psychotic symptoms (delusions/hallucinations); these symptoms are considerably location-dependent.30-32
Content and associated signs/symptoms. There are some grouped symptoms and/or hallucination characteristics associated with cerebral tumors in different lobes of the brain, though these symptoms are not specific. The visual hallucinations associated with brain tumors are typically confined to the field of vision that corresponds to the location of the tumor. Additionally, many such patients have a baseline visual field defect to some extent due to the tumor location.
In patients with occipital lobe tumors, visual hallucinations closely resemble those experienced in occipital lobe seizures, specifically bright flashes of light in colorful simple and complex shapes. Interestingly, those with occipital lobe tumors report xanthopsia, a form of chromatopsia in which objects in their field of view appear abnormally colored a yellowish shade.26,27
In patients with temporal lobe tumors, more complex visual hallucinations of people, objects, and events occurring around them are often accompanied by auditory hallucinations, olfactory hallucinations, and/or anosmia.28In those with frontal lobe tumors, similar complex visual hallucinations of people, objects, and events are seen, and olfactory hallucinations and/or anosmia are often experienced. However, these patients often have a lower likelihood of experiencing auditory hallucinations, and a higher likelihood of developing personality changes and depression than other psychotic symptoms. The visual hallucinations experienced in those with frontal lobe tumors are more likely to have violent content.29
Continue to: Visual hallucinations in migraine with aura
Visual hallucinations in migraine with aura
The estimated prevalence of migraine in the general population is 15% to 29%; 31% of those with migraine experience auras.33-35 Approximately 99% of those with migraine auras experience some type of associated visual phenomena.33,36 The pathophysiology of migraine is believed to be related to spreading cortical depression, in which a slowly propagating wave of neuroelectric depolarization travels over the cortex, followed by a depression of normal brain activity. Visual aura is thought to occur due to the resulting changes in cortical activity in the visual cortex; however, the exact electrophysiology of visual migraine aura is not entirely known.37,38 Though most patients with visual migraine aura experience simple visual hallucinations, complex hallucinations have been reported in the (very rare) cases of migraine coma and familial hemiplegic migraine.39
Content and associated signs/symptoms. The most common hallucinated entities reported by patients with migraine with aura are zigzag, flashing/sparkling, black and white curved figure(s) in the center of the visual field, commonly called a scintillating phosphene or scintillating scotoma.36 The perceived entity is often singular and gradually moves from the center to the periphery of the visual field. These visual hallucinations appear in front of all other objects in the visual field and do not interact with the environment or observer, or resemble or morph into any real-world objects, though they may change in contour, size, and color. The scintillating nature of the hallucination often resolves within minutes, usually leaving a scotoma, or area of vision loss, in the area, with resolution back to baseline vision within 1 hour. The straight, zigzag, and usually black-and-white nature of the scintillating phosphenes of migraine are in notable contrast to the colorful, often circular visual hallucinations experienced in patients with occipital lobe seizures.25
Visual hallucinations in peduncular hallucinosis
Peduncular hallucinosis is a syndrome of predominantly dreamlike visual hallucinations that occurs in the setting of lesions in the midbrain and/or thalamus.40 A recent review of the lesion etiology found that approximately 63% are caused by focal infarction and approximately 15% are caused by mass lesions; subarachnoid hemorrhage, intracerebral hemorrhage, and demyelination cause approximately 5% of cases each.40 Additionally, a review of the affected brainstem anatomy showed almost all lesions were found in the paramedian reticular formations of the midbrain and pons, with the vast majority of lesions affecting or adjacent to the oculomotor and raphe nuclei of the midbrain.39 Due to the commonly involved visual pathway, some researchers have suggested these hallucinations may be the result of a release phenomenon.39
Content and associated signs/symptoms. The visual hallucinations of peduncular hallucinosis usually start 1 to 5 days after the causal lesion forms, last several minutes to hours, and most stop after 1 to 3 weeks; however, cases of hallucinations lasting for years have been reported. These hallucinations have a diurnal pattern of usually appearing while the patient is resting in the evening and/or preparing for sleep. The characteristics of visual hallucinations vary widely from simple distortions in how real objects appear to colorful and vivid hallucinated events and people who can interact with the observer. The content of the visual hallucinations often changes in nature during the hallucination, or from one hallucination to the next. The hallucinated entities can be worldly or extraterrestrial. Once these patients fall asleep, they often have equally vivid and unusual dreams, with content similar to their visual hallucinations. Due to the anatomical involvement of the nigrostriatal pathway and oculomotor nuclei, co-occurring parkinsonism, ataxia, and oculomotor nerve palsy are common and can be a key clinical feature in establishing the diagnosis. Though patients with peduncular hallucinations commonly fear their hallucinations, they often eventually gain insight, which eases their anxiety.39
Other causes
Visual hallucinations in visual impairment
Visual hallucinations are a diagnostic requirement for Charles Bonnet syndrome, in which individuals with vision loss experience visual hallucinations in the corresponding field of vision loss.41 A lesion at any point in the visual pathway that produces visual loss can lead to Charles Bonnet syndrome; however, age-related macular degeneration is the most common cause.42 The hallucinations of Charles Bonnet syndrome are believed to be a release phenomenon, given the defective visual pathway and resultant dysfunction in visual processing. The prevalence of Charles Bonnet syndrome ranges widely by study. Larger studies report a prevalence of 11% to 27% in patients with age-related macular degeneration, depending on the severity of vision loss.43,44 Because there are many causes of Charles Bonnet syndrome, and because a recent study found that only 15% of patients with this syndrome told their eye care clinician and that 21% had not reported their hallucinatory symptoms to anyone, the true prevalence is unknown.42 Though the onset of visual hallucinations correlates with the onset of vision loss, there appears to be no association between the nature or complexity of the hallucinations and the severity or progression of the patient’s vision loss.45 Some studies have reported either the onset of or a higher frequency of visual hallucinations at a time of visual recovery (for example, treatment or exudative age-related macular degeneration), which suggests that hallucinations may be triggered by fluctuations in visual acuity.46,47 Additional risk factors for experiencing visual hallucinations in the setting of visual pathway deficit include a history of stroke, social isolation, poor cognitive function, poor lighting, and age ≥65.
Continue to: Content and associated signs/symptoms
Content and associated signs/symptoms. The visual hallucinations of patients with Charles Bonnet syndrome appear almost exclusively in the defective visual field. Images tend to be complex, colored, with moving parts, and appear in front of the patient. The hallucinations are usually of familiar or normal-appearing people or mundane objects, and as such, the patient often does not realize the hallucinated entity is not real. In patients without comorbid psychiatric disease, visual hallucinations are not accompanied by any other types of hallucinations. The most commonly hallucinated entities are people, followed by simple visual hallucinations of geometric patterns, and then by faces (natural or cartoon-like) and inanimate objects. Hallucinations most commonly occur daily or weekly, and upon waking. These hallucinations most often last several minutes, though they can last just a few seconds or for hours. Hallucinations are usually emotionally neutral, but most patients report feeling confused by their appearance and having a fear of underlying psychiatric disease. They often gain insight to the unreal nature of the hallucinations after counseling.48
Visual hallucinations at the sleep/wake interface
Hypnagogic and hypnopompic hallucinations are fleeting perceptual experiences that occur while an individual is falling asleep or waking, respectively.49 Because by definition visual hallucinations occur while the individual is fully awake, categorizing hallucination-like experiences such as hypnagogia and hypnopompia is difficult, especially since these are similar to other states in which alterations in perception are expected (namely a dream state). They are commonly associated with sleep disorders such as narcolepsy, cataplexy, and sleep paralysis.50,51 In a study of 13,057 individuals in the general population, Ohayon et al4 found the overall prevalence of hypnagogic or hypnopompic hallucinations was 24.8% (5.3% visual) and 6.6% (1.5% visual), respectively. Approximately one-third of participants reported having experienced ≥1 hallucinatory experience in their lifetime, regardless of being asleep or awake.4 There was a higher prevalence of hypnagogic/hypnopompic experiences among those who also reported daytime hallucinations or other psychotic features.
Content and associated signs/symptoms. Unfortunately, because of the frequent co-occurrence of sleep disorders and psychiatric conditions, as well as the general paucity of research, it is difficult to characterize the visual phenomenology of hypnagogic/hypnopompic hallucinations. Some evidence suggests the nature of the perception of the objects hallucinated is substantially impacted by the presence of preexisting psychotic symptoms. Insight into the reality of these hallucinations also depends upon the presence of comorbid psychiatric disease. Hypnagogic/hypnopompic hallucinations are often described as complex, colorful, vivid, and dream-like, as if the patient was in a “half sleep” state.52 They are usually described as highly detailed events involving people and/or animals, though they may be grotesque in nature. Perceived entities are often described as undergoing a transformation or being mobile in their environment. Rarely do these perceptions invoke emotion or change the patient’s beliefs. Hypnagogia/hypnopompia also often have an auditory or haptic component to them. Visual phenomena can either appear to take place within an alternative background environment or appear superimposed on the patient’s actual physical environment.
How to determine the cause
In many of the studies cited in this review, the participants had a considerable amount of psychiatric comorbidity, which makes it difficult to discriminate between pure neurologic and pure psychiatric causes of hallucinations. Though the visual content of the hallucinations (people, objects, shapes, lights) can help clinicians broadly differentiate causes, many other characteristics of both the hallucinations and the patient can help determine the cause (Table3,4,12-39,41-52). The most useful characteristics for discerning the etiology of an individual’s visual hallucinations are the patient’s age, the visual field in which the hallucination occurs, and the complexity/simplicity of the hallucination.
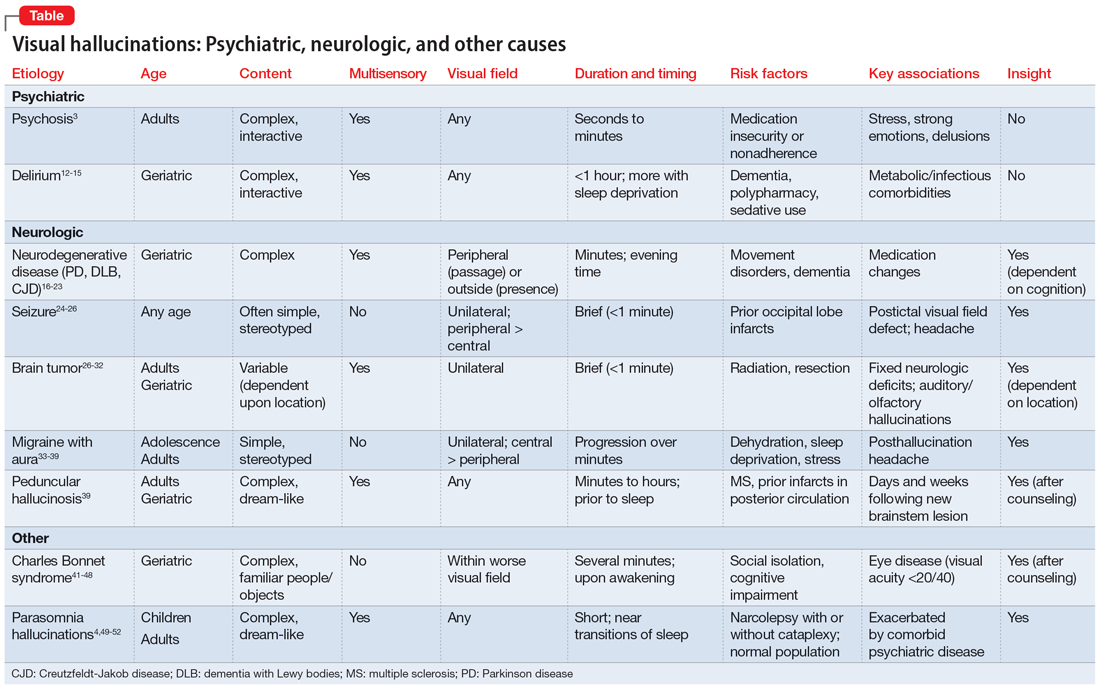
Patient age. Hallucinations associated with primary psychosis decrease with age. The average age of onset of migraine with aura is 21. Occipital lobe seizures occur in early childhood to age 40, but most commonly occur in the second decade.32,36 No trend in age can be reliably determined in individuals who experience hypnagogia/hypnopompia. In contrast, other potential causes of visual hallucinations, such as delirium, neurodegenerative disease, eye disease, and peduncular hallucinosis, are more commonly associated with advanced age.
Continue to: The visual field(s)
The visual field(s) in which the hallucination occurs can help differentiate possible causes in patients with seizure, brain tumor, migraine, or visual impairment. In patients with psychosis, delirium, peduncular hallucinosis, or hypnagogia/hypnopompia, hallucinations can occur in any visual field. Those with neurodegenerative disease, particularly PD, commonly describe seeing so-called passage hallucinations and presence hallucinations, which occur outside of the patient’s direct vision. Visual hallucinations associated with seizure are often unilateral (homonymous left or right hemifield), and contralateral to the affected neurologic structures in the visual neural pathway; they start in the left or right peripheral vision and gradually move to the central visual field. In hallucinations experienced by patients with brain tumors, the hallucinated entities typically appear on the visual field contralateral to the underlying tumor. Visual hallucinations seen in migraine often include a figure that moves from central vision to more lateral in the visual field. The visual hallucinations seen in eye disease (namely Charles Bonnet syndrome) are almost exclusively perceived in the visual fields affected by decreased visual acuity, though non-side-locked visual hallucinations are common in patients with age-related macular degeneration.
Content and complexity. The visual hallucinations perceived in those with psychosis, delirium, neurodegenerative disease, and sleep disorders are generally complex. These hallucinations tend to be of people, animals, scenes, or faces and include color and associated sound, with moving parts and interactivity with either the patient or the environment. These are in contrast to the simple visual hallucinations of visual cortex seizures, brain tumors, and migraine aura, which are often reported as brightly colored or black/white lights, flashes, and shapes, with or without associated auditory, olfactory, or somatic sensation. Furthermore, hallucinations due to seizure and brain tumor (also likely due to seizure) are often of brightly colored shapes and lights with curved edges, while patients with migraine more commonly report singular sparkling black/white objects with straight lines.
Bottom Line
Though there are no features known to be specific to only 1 cause of visual hallucinations, some characteristics of both the patient and the hallucinations can help direct the diagnostic differential. The most useful characteristics are the patient’s age, the visual field in which the hallucination occurs, and the complexity/ simplicity of the hallucination.
Related Resources
- Wang J, Patel D, Francois D. Elaborate hallucinations, but is it a psychotic disorder? Current Psychiatry. 2021;20(2):46-50. doi:10.12788/cp.0091
- O’Brien J, Taylor JP, Ballard C, et al. Visual hallucinations in neurological and ophthalmological disease: pathophysiology and management. J Neurol Neurosurg Psychiatry. 2020; 91(5):512-519. doi:10.1136/jnnp-2019-322702
1. Asaad G, Shapiro B. Hallucinations: theoretical and clinical overview. Am J Psychiatry. 1987;143(9):1088-1097.
2. Taam MA, Boissieu P, Taam RA, et al. Drug-induced hallucination: a case/non-case study in the French Pharmacovigilance Database. Article in French. Eur J Psychiatry. 2015;29(1):21-31.
3. Waters F, Collerton D, Ffytche DH, et al. Visual hallucinations in the psychosis spectrum and comparative information from neurodegenerative disorders and disease. Schizophr Bull. 2014;40(Suppl 4):S233-S245.
4. Ohayon MM. Prevalence of hallucinations and their pathological associations in the general population. Psychiatry Res. 2000;97(2-3):153-164.
5. Rees WD. The hallucinations of widowhood. Br Med J. 1971;4(5778):37-41.
6. Delespaul P, deVries M, van Os J. Determinants of occurrence and recovery from hallucinations in daily life. Soc Psychiatry Psychiatr Epidemiol. 2002;37(3):97-104.
7. Gauntlett-Gilbert J, Kuipers E. Phenomenology of visual hallucinations in psychiatric conditions. J Nerv Ment Dis. 2003;191(3):203-205.
8. Goodwin FK, Jamison KR. Manic Depressive Illness. Oxford University Press, Inc.; 1999.
9. Mueser KT, Bellack AS, Brady EU. Hallucinations in schizophrenia. Acta Psychiatr Scand. 1990;82(1):26-29.
10. McCabe MS, Fowler RC, Cadoret RJ, et al. Symptom differences in schizophrenia with good and bad prognosis. Am J Psychiatry. 1972;128(10):1239-1243.
11. Baethge C, Baldessarini RJ, Freudenthal K, et al. Hallucinations in bipolar disorder: characteristics and comparison to unipolar depression and schizophrenia. Bipolar Disord. 2005;7(2):136-145.
12. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Publishing; 2013.
13. Ahmed S, Leurent B, Sampson EL. Risk factors for incident delirium among older people in acute hospital medical units: a systematic review and meta-analysis. Age Ageing. 2014;43(3):326-333.
14. Webster R, Holroyd S. Prevalence of psychotic symptoms in delirium. Psychosomatics. 2000;41(6):519-522.
15. Tachibana M, Inada T, Ichida M, et al. Factors affecting hallucinations in patients with delirium. Sci Rep. 2021;11(1):13005. doi:10.1038/s41598-021-92578-1
16. Fenelon G, Mahieux F, Huon R, et al. Hallucinations in Parkinson’s disease: prevalence, phenomenology and risk factors. Brain. 2000;123(Pt 4):733-745.
17. Papapetropoulos S, Argyriou AA, Ellul J. Factors associated with drug-induced visual hallucinations in Parkinson’s disease. J Neurol. 2005;252(10):1223-1228.
18. Williams DR, Warren JD, Lees AJ. Using the presence of visual hallucinations to differentiate Parkinson’s disease from atypical parkinsonism. J Neurol Neurosurg Psychiatry. 2008;79(6):652-655.
19. Linszen MMJ, Lemstra AW, Dauwan M, et al. Understanding hallucinations in probable Alzheimer’s disease: very low prevalence rates in a tertiary memory clinic. Alzheimers Dement (Amst). 2018;10:358-362.
20. Burghaus L, Eggers C, Timmermann L, et al. Hallucinations in neurodegenerative diseases. CNS Neurosci Ther. 2012;18(2):149-159.
21. Brar HK, Vaddigiri V, Scicutella A. Of illusions, hallucinations, and Creutzfeldt-Jakob disease (Heidenhain’s variant). J Neuropsychiatry Clin Neurosci. 2005;17(1):124-126.
22. Sasaki C, Yokoi K, Takahashi H, et al. Visual illusions in Parkinson’s disease: an interview survey of symptomatology. Psychogeriatrics. 2022;22(1):28-48.
23. Kropp S, Schulz-Schaeffer WJ, Finkenstaedt M, et al. The Heidenhain variant of Creutzfeldt-Jakob disease. Arch Neurol. 1999;56(1):55-61.
24. Taylor I, Scheffer IE, Berkovic SF. Occipital epilepsies: identification of specific and newly recognized syndromes. Brain. 2003;126(Pt 4):753-769.
25. Caraballo R, Cersosimo R, Medina C, et al. Panayiotopoulos-type benign childhood occipital epilepsy: a prospective study. Neurology. 2000;5(8):1096-1100.
26. Chowdhury FA, Silva R, Whatley B, et al. Localisation in focal epilepsy: a practical guide. Practical Neurol. 2021;21(6):481-491.
27. Horrax G, Putnam TJ. Distortions of the visual fields in cases of brain tumour: the field defects and hallucinations produced by tumours of the occipital lobe. Brain. 1932;55(4):499-523.
28. Cushing H. Distortions of the visual fields in cases of brain tumor (6th paper): the field defects produced by temporal lobe lesions. Brain. 1922;44(4):341-396.
29. Fornazzari L, Farcnik K, Smith I, et al. Violent visual hallucinations and aggression in frontal lobe dysfunction: clinical manifestations of deep orbitofrontal foci. J Neuropsychiatry Clin Neurosci. 1992;4(1):42-44.
30. Madhusoodanan S, Opler MGA, Moise D, et al. Brain tumor location and psychiatric symptoms: is there an association? A meta-analysis of published cases studies. Expert Rev Neurother. 2010;10(10):1529-1536.
31. Madhusoodanan S, Sinha A, Moise D. Brain tumors and psychiatric manifestations: a review and analysis. Poster presented at: The American Association for Geriatric Psychiatry Annual Meeting; March 10-13; 2006; San Juan, Puerto Rico.
32. Madhusoodanan S, Danan D, Moise D. Psychiatric manifestations of brain tumors/gliomas. Rivistica Medica. 2007;13(4):209-215.
33. Kirchmann M. Migraine with aura: new understanding from clinical epidemiological studies. Curr Opin Neurol. 2006;19:286-293.
34. Goadsby PJ, Lipton RB, Ferrari MD. Migraine: current understanding and treatment. N Engl J Med. 2002;346(4):257-270.
35. Waters WE, O’Connor PJ. Prevalence of migraine. J Neurol Neurosurg Psychiatry. 1975;38(6):613-616.
36. Russell MB, Olesen J. A nosographic analysis of the migraine aura in a general population. Brain. 1996;119(Pt 2):355-361.
37. Cozzolino O, Marchese M, Trovato F, et al. Understanding spreading depression from headache to sudden unexpected death. Front Neurol. 2018;9:19.
38. Hadjikhani N, Sanchez del Rio M, Wu O, et al. Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci U S A. 2001;98(8):4687-4692.
39. Manford M, Andermann F. Complex visual hallucinations. Clinical and neurobiological insights. Brain. 1998;121(Pt 10):1819-1840.
40. Galetta KM, Prasad S. Historical trends in the diagnosis of peduncular hallucinosis. J Neuroophthalmol. 2018;38(4):438-441.
41. Schadlu AP, Schadlu R, Shepherd JB III. Charles Bonnet syndrome: a review. Curr Opin Ophthalmol. 2009;20(3):219-222.
42. Vukicevic M, Fitzmaurice K. Butterflies and black lace patterns: the prevalence and characteristics of Charles Bonnet hallucinations in an Australian population. Clin Exp Ophthalmol. 2008;36(7):659-665.
43. Teunisse RJ, Cruysberg JR, Verbeek A, et al. The Charles Bonnet syndrome: a large prospective study in the Netherlands. A study of the prevalence of the Charles Bonnet syndrome and associated factors in 500 patients attending the University Department of Ophthalmology at Nijmegen. Br J Psychiatry. 1995;166(2):254-257.
44. Holroyd S, Rabins PV, Finkelstein D, et al. Visual hallucination in patients with macular degeneration. Am J Psychiatry. 1992;149(12):1701-1706.
45. Khan JC, Shahid H, Thurlby DA, et al. Charles Bonnet syndrome in age-related macular degeneration: the nature and frequency of images in subjects with end-stage disease. Ophthalmic Epidemiol. 2008;15(3):202-208.
46. Cohen SY, Bulik A, Tadayoni R, et al. Visual hallucinations and Charles Bonnet syndrome after photodynamic therapy for age related macular degeneration. Br J Ophthalmol. 2003;87(8):977-979.
47. Meyer CH, Mennel S, Horle S, et al. Visual hallucinations after intravitreal injection of bevacizumab in vascular age-related macular degeneration. Am J Ophthalmol. 2007;143(1):169-170.
48. Jan T, Del Castillo J. Visual hallucinations: Charles Bonnet syndrome. West J Emerg Med. 2012;13(6):544-547. doi:10.5811/westjem.2012.7.12891
49. Foulkes D, Vogel G. Mental activity at sleep onset. J Abnorm Psychol. 1965;70:231-243.
50. Mitler MM, Hajdukovic R, Erman M, et al. Narcolepsy. J Clin Neurophysiol. 1990;7(1):93-118.
51. Nishino S. Clinical and neurobiological aspects of narcolepsy. Sleep Med. 2007;8(4):373-399.
52. Schultz SK, Miller DD, Oliver SE, et al. The life course of schizophrenia: age and symptom dimensions. Schizophr Res. 1997;23(1):15-23.
A visual hallucination is a visual percept experienced when awake that is not elicited by an external stimulus. Historically, hallucinations have been synonymous with psychiatric disease, most notably schizophrenia; however, over recent decades, hallucinations have been categorized based on their underlying etiology as psychodynamic (primary psychiatric), psychophysiologic (primary neurologic/structural), and psychobiochemical (neurotransmitter dysfunction).1 Presently, visual hallucinations are known to be caused by a wide variety of primary psychiatric, neurologic, ophthalmologic, and chemically-mediated conditions. Despite these causes, clinically differentiating the characteristics and qualities of visual hallucinations is often a lesser-known skillset among clinicians. The utility of this skillset is important for the clinician’s ability to differentiate the expected and unexpected characteristics of visual hallucinations in patients with both known and unknown neuropsychiatric conditions.
Though many primary psychiatric and neurologic conditions have been associated with and/or known to cause visual hallucinations, this review focuses on the following grouped causes:
- Primary psychiatric causes: psychiatric disorders with psychotic features and delirium; and
- Primary neurologic causes: neurodegenerative disease/dementias, seizure disorders, migraine disorders, vision loss, peduncular hallucinosis, and hypnagogic/hypnopompic phenomena.
Because the accepted definition of visual hallucinations excludes visual percepts elicited by external stimuli, drug-induced hallucinations would not qualify for either of these categories. Additionally, most studies reporting on the effects of drug-induced hallucinations did not control for underlying comorbid psychiatric conditions, dementia, or delirium, and thus the results cannot be attributed to the drug alone, nor is it possible to identify reliable trends in the properties of the hallucinations.2 The goals of this review are to characterize visual hallucinations experienced as a result of primary psychiatric and primary neurologic conditions and describe key grouping and differentiating features to help guide the diagnosis.
Visual hallucinations in the general population
A review of 6 studies (N = 42,519) reported that the prevalence of visual hallucinations in the general population is 7.3%.3 The prevalence decreases to 6% when visual hallucinations arising from physical illness or drug/chemical consumption are excluded. The prevalence of visual hallucinations in the general population has been associated with comorbid anxiety, stress, bereavement, and psychotic pathology.4,5 Regarding the age of occurrence of visual hallucinations in the general population, there appears to be a bimodal distribution.3 One peak appears in later adolescence and early adulthood, which corresponds with higher rates of psychosis, and another peak occurs late in life, which corresponds to a higher prevalence of neurodegenerative conditions and visual impairment.
Primary psychiatric causes
Most studies of visual hallucinations in primary psychiatric conditions have specifically evaluated patients with schizophrenia and mood disorders with psychotic features.6,7 In a review of 29 studies (N = 5,873) that specifically examined visual hallucinations in individuals diagnosed with schizophrenia, Waters et al3 found a wide range of reported prevalence (4% to 65%) and a weighted mean prevalence of 27%. In contrast, the prevalence of auditory hallucinations in these participants ranged from 25% to 86%, with a weighted mean of 59%.3
Hallucinations are a known but less common symptom of mood disorders that present with psychotic features.8 Waters et al3 also examined the prevalence of visual and auditory hallucinations in mood disorders (including mania, bipolar disorder, and depression) reported in 12 studies (N = 2,892).3 They found the prevalence of visual hallucinations in patients with mood disorders ranged from 6% to 27%, with a weighted mean of 15%, compared to the weighted mean of 28% who experienced auditory hallucinations. Visual hallucinations in primary psychiatric conditions are associated with more severe disease, longer hospitalizations, and poorer prognoses.9-11
Visual hallucinations of psychosis
In patients with psychotic symptoms, the characteristics of the visually hallucinated entity as well as the cognitive and emotional perception of the hallucinations are notably different than in patients with other, nonpsychiatric causes of visual hallucations.3
Continue to: Content and perceived physical properties
Content and perceived physical properties. Hallucinated entities are most often perceived as solid, 3-dimensional, well-detailed, life-sized people, animals, and objects (often fire) or events existing in the real world.3 The entity is almost always perceived as real, with accurate form and color, fine edges, and shadow; is often out of reach of the perceiver; and can be stationary or moving within the physical properties of the external environment.3
Timing and triggers. The temporal properties vary widely. Hallucinations can last from seconds to minutes and occur at any time of day, though by definition, they must occur while the individual is awake.3 Visual hallucinations in psychosis are more common during times of acute stress, strong emotions, and tiredness.3
Patient reaction and belief. Because of realistic qualities of the visual hallucination and the perception that it is real, patients commonly attempt to participate in some activity in relation to the hallucination, such as moving away from or attempting to interact with it.3 Additionally, patients usually perceive the hallucinated entity as uncontrollable, and are surprised when the entity appears or disappears. Though the content of the hallucination is usually impersonal, the meaning the patient attributes to the presence of the hallucinated entity is usually perceived as very personal and often requiring action. The hallucination may represent a harbinger, sign, or omen, and is often interpreted religiously or spiritually and accompanied by comorbid delusions.3
Visual hallucinations of delirium
Delirium is a syndrome of altered mentation—most notably consciousness, attention, and orientation—that occurs as a result of ≥1 metabolic, infectious, drug-induced, or other medical conditions and often manifests as an acute secondary psychotic illness.12 Multiple patient and environmental characteristics have been identified as risk factors for developing delirium, including multiple and/or severe medical illnesses, preexisting dementia, depression, advanced age, polypharmacy, having an indwelling urinary catheter, impaired sight or hearing, and low albumin levels.13-15 The development of delirium is significantly and positively associated with regular alcohol use, benzodiazepine withdrawal, and angiotensin receptor blocker and dopamine receptor agonist usage.15 Approximately 40% of patients with delirium have symptoms of psychosis, and in contrast to the hallucinations experienced by patients with schizophrenia, visual hallucinations are the most common type of hallucinations seen in delirium (27%).13 In a 2021 review that included 602 patients with delirium, Tachibana et al15 found that approximately 26% experienced hallucinations, 92% of which were visual hallucinations.
Content, perceived physical properties, and reaction. Because of the limited attention and cognitive function of patients with delirium, less is known about the content of their visual hallucinations. However, much like those with primary psychotic symptoms, patients with delirium often report seeing complex, normal-sized, concrete entities, most commonly people. Tachibana et al15 found that the hallucinated person is more often a stranger than a familiar person, but (rarely) may be an ethereal being such as a devil or ghost. The next most common visually hallucinated entities were creatures, most frequently insects and animals. Other common hallucinations were visions of events or objects, such as fires, falling ceilings, or water. Similar to those with primary psychotic illness such as schizophrenia, patients with delirium often experience emotional distress, anxiety, fear, and confusion in response to the hallucinated person, object, and/or event.15
Continue to: Primary neurologic causes
Primary neurologic causes
Visual hallucinations in neurodegenerative diseases
Patients with neurodegenerative diseases such as Parkinson disease (PD), dementia with Lewy bodies (DLB), or Creutzfeldt-Jakob disease (CJD) commonly experience hallucinations as a feature of their condition. However, the true cause of these hallucinations often cannot be directly attributed to any specific pathophysiology because these patients often have multiple coexisting risk factors, such as advanced age, major depressive disorder, use of neuroactive medications, and co-occurring somatic illness. Though the prevalence of visual hallucinations varies widely between studies, with 15% to 40% reported in patients with PD, the prevalence roughly doubles in patients with PD-associated dementia (30% to 60%), and is reported by 60% to 90% of those with DLB.16-18 Hallucinations are generally thought to be less common in Alzheimer disease; such patients most commonly experience visual hallucinations, although the reported prevalence ranges widely (4% to 59%).19,20 Notably, similarly to hallucinations experienced in patients with delirium, and in contrast to those with psychosis, visual hallucinations are more common than auditory hallucinations in neurodegenerative diseases.20 Hallucinations are not common in individuals with CJD but are a key defining feature of the He
Content, perceived physical properties, and reaction. Similar to the visual hallucinations experienced by patients with psychosis or delirium, those experienced in patients with PD, DLB, or CJD are often complex, most commonly of people, followed by animals and objects. The presence of “passage hallucinations”—in which a person or animal is seen in a patient’s peripheral vision, but passes out of their visual field before the entity can be directly visualized—is common.20 Those with PD also commonly have visual hallucinations in which the form of an object appears distorted (dysmorphopsia) or the color of an object appears distorted (metachromatopsia), though these would better be classified as illusions because a real object is being perceived with distortion.22
Hallucinations are more common in the evening and at night. “Presence hallucinations” are a common type of hallucination that cannot be directly related to a specific sensory modality such as vision, though they are commonly described by patients with PD as a seen or perceived image (usually a person) that is not directly in the individual’s visual field.17 These presence hallucinations are often described as being behind the patient or in a visualized scene of what was about to happen. Before developing the dementia and myoclonus also seen in sporadic CJD, patients with the Heidenhain variant of CJD describe illusions such as metachromatopsia, dysmorphia, and micropsia that eventually develop into frank visual hallucinations, which have been poorly reported in medical literature.22,23 There are no generalizable trends in the temporal nature of visual hallucinations in patients with neurodegenerative diseases. In most cases of visual hallucinations in patients with PD and dementia, insight relating to the perception varies widely based on the patient’s cognitive status. Subsequently, patients’ reactions to the hallucinations also vary widely.
Visual hallucinations in epileptic seizures
Occipital lobe epilepsies represent 1% to 4.6% of all epilepsies; however, these represent 20% to 30% of benign childhood partial epilepsies.24,25 These are commonly associated with various types of visual hallucinations depending upon the location of the seizure onset within the occipital lobe. These are referred to as visual auras.26 Visual auras are classified into simple visual hallucinations, complex visual hallucinations, visual illusions, and ictal amaurosis (hemifield blindness or complete blindness).
Content, perceived physical properties, and reaction. Simple visual hallucinations are often described as brief, stereotypical flashing lights of various shapes and colors. These images may flicker, change shape, or take on a geometric or irregular pattern. Appearances can be repetitive and stereotyped, are often reported as moving horizontally from the periphery to the center of the visual field, and can spread to the entire visual field. Most often, these hallucinations occur for 5 to 30 seconds, and have no discernible provoking factors. Complex visual hallucinations consist of formed images of animals, people, or elaborate scenes. These are believed to reflect activation of a larger area of cortex in the temporo-parieto-occipital region, which is the visual association cortex. Very rarely, occipital lobe seizures can manifest with ictal amaurosis.24
Continue to: Simple visual auras...
Simple visual auras have a very high localizing value to the occipital lobe. The primary visual cortex (Brodmann area 17) is situated in the banks of calcarine fissure and activation of this region produces these simple hallucinations. If the hallucinations are consistently lateralized, the seizures are very likely to be coming from the contralateral occipital lobe.
Visual hallucinations in brain tumors
In general, a tumor anywhere along the optic path can produce visual hallucinations; however, the exact causal mechanism of the hallucinations is unknown. Moreover, tumors in different locations—namely the occipital lobes, temporal lobes, and frontal lobes—appear to produce visual hallucinations with substantially different characteristics.27-29 Further complicating the search for the mechanism of these hallucinations is the fact that tumors are epileptogenic. In addition, 36% to 48% of patients with brain tumors have mood symptoms (depression/mania), and 22% to 24% have psychotic symptoms (delusions/hallucinations); these symptoms are considerably location-dependent.30-32
Content and associated signs/symptoms. There are some grouped symptoms and/or hallucination characteristics associated with cerebral tumors in different lobes of the brain, though these symptoms are not specific. The visual hallucinations associated with brain tumors are typically confined to the field of vision that corresponds to the location of the tumor. Additionally, many such patients have a baseline visual field defect to some extent due to the tumor location.
In patients with occipital lobe tumors, visual hallucinations closely resemble those experienced in occipital lobe seizures, specifically bright flashes of light in colorful simple and complex shapes. Interestingly, those with occipital lobe tumors report xanthopsia, a form of chromatopsia in which objects in their field of view appear abnormally colored a yellowish shade.26,27
In patients with temporal lobe tumors, more complex visual hallucinations of people, objects, and events occurring around them are often accompanied by auditory hallucinations, olfactory hallucinations, and/or anosmia.28In those with frontal lobe tumors, similar complex visual hallucinations of people, objects, and events are seen, and olfactory hallucinations and/or anosmia are often experienced. However, these patients often have a lower likelihood of experiencing auditory hallucinations, and a higher likelihood of developing personality changes and depression than other psychotic symptoms. The visual hallucinations experienced in those with frontal lobe tumors are more likely to have violent content.29
Continue to: Visual hallucinations in migraine with aura
Visual hallucinations in migraine with aura
The estimated prevalence of migraine in the general population is 15% to 29%; 31% of those with migraine experience auras.33-35 Approximately 99% of those with migraine auras experience some type of associated visual phenomena.33,36 The pathophysiology of migraine is believed to be related to spreading cortical depression, in which a slowly propagating wave of neuroelectric depolarization travels over the cortex, followed by a depression of normal brain activity. Visual aura is thought to occur due to the resulting changes in cortical activity in the visual cortex; however, the exact electrophysiology of visual migraine aura is not entirely known.37,38 Though most patients with visual migraine aura experience simple visual hallucinations, complex hallucinations have been reported in the (very rare) cases of migraine coma and familial hemiplegic migraine.39
Content and associated signs/symptoms. The most common hallucinated entities reported by patients with migraine with aura are zigzag, flashing/sparkling, black and white curved figure(s) in the center of the visual field, commonly called a scintillating phosphene or scintillating scotoma.36 The perceived entity is often singular and gradually moves from the center to the periphery of the visual field. These visual hallucinations appear in front of all other objects in the visual field and do not interact with the environment or observer, or resemble or morph into any real-world objects, though they may change in contour, size, and color. The scintillating nature of the hallucination often resolves within minutes, usually leaving a scotoma, or area of vision loss, in the area, with resolution back to baseline vision within 1 hour. The straight, zigzag, and usually black-and-white nature of the scintillating phosphenes of migraine are in notable contrast to the colorful, often circular visual hallucinations experienced in patients with occipital lobe seizures.25
Visual hallucinations in peduncular hallucinosis
Peduncular hallucinosis is a syndrome of predominantly dreamlike visual hallucinations that occurs in the setting of lesions in the midbrain and/or thalamus.40 A recent review of the lesion etiology found that approximately 63% are caused by focal infarction and approximately 15% are caused by mass lesions; subarachnoid hemorrhage, intracerebral hemorrhage, and demyelination cause approximately 5% of cases each.40 Additionally, a review of the affected brainstem anatomy showed almost all lesions were found in the paramedian reticular formations of the midbrain and pons, with the vast majority of lesions affecting or adjacent to the oculomotor and raphe nuclei of the midbrain.39 Due to the commonly involved visual pathway, some researchers have suggested these hallucinations may be the result of a release phenomenon.39
Content and associated signs/symptoms. The visual hallucinations of peduncular hallucinosis usually start 1 to 5 days after the causal lesion forms, last several minutes to hours, and most stop after 1 to 3 weeks; however, cases of hallucinations lasting for years have been reported. These hallucinations have a diurnal pattern of usually appearing while the patient is resting in the evening and/or preparing for sleep. The characteristics of visual hallucinations vary widely from simple distortions in how real objects appear to colorful and vivid hallucinated events and people who can interact with the observer. The content of the visual hallucinations often changes in nature during the hallucination, or from one hallucination to the next. The hallucinated entities can be worldly or extraterrestrial. Once these patients fall asleep, they often have equally vivid and unusual dreams, with content similar to their visual hallucinations. Due to the anatomical involvement of the nigrostriatal pathway and oculomotor nuclei, co-occurring parkinsonism, ataxia, and oculomotor nerve palsy are common and can be a key clinical feature in establishing the diagnosis. Though patients with peduncular hallucinations commonly fear their hallucinations, they often eventually gain insight, which eases their anxiety.39
Other causes
Visual hallucinations in visual impairment
Visual hallucinations are a diagnostic requirement for Charles Bonnet syndrome, in which individuals with vision loss experience visual hallucinations in the corresponding field of vision loss.41 A lesion at any point in the visual pathway that produces visual loss can lead to Charles Bonnet syndrome; however, age-related macular degeneration is the most common cause.42 The hallucinations of Charles Bonnet syndrome are believed to be a release phenomenon, given the defective visual pathway and resultant dysfunction in visual processing. The prevalence of Charles Bonnet syndrome ranges widely by study. Larger studies report a prevalence of 11% to 27% in patients with age-related macular degeneration, depending on the severity of vision loss.43,44 Because there are many causes of Charles Bonnet syndrome, and because a recent study found that only 15% of patients with this syndrome told their eye care clinician and that 21% had not reported their hallucinatory symptoms to anyone, the true prevalence is unknown.42 Though the onset of visual hallucinations correlates with the onset of vision loss, there appears to be no association between the nature or complexity of the hallucinations and the severity or progression of the patient’s vision loss.45 Some studies have reported either the onset of or a higher frequency of visual hallucinations at a time of visual recovery (for example, treatment or exudative age-related macular degeneration), which suggests that hallucinations may be triggered by fluctuations in visual acuity.46,47 Additional risk factors for experiencing visual hallucinations in the setting of visual pathway deficit include a history of stroke, social isolation, poor cognitive function, poor lighting, and age ≥65.
Continue to: Content and associated signs/symptoms
Content and associated signs/symptoms. The visual hallucinations of patients with Charles Bonnet syndrome appear almost exclusively in the defective visual field. Images tend to be complex, colored, with moving parts, and appear in front of the patient. The hallucinations are usually of familiar or normal-appearing people or mundane objects, and as such, the patient often does not realize the hallucinated entity is not real. In patients without comorbid psychiatric disease, visual hallucinations are not accompanied by any other types of hallucinations. The most commonly hallucinated entities are people, followed by simple visual hallucinations of geometric patterns, and then by faces (natural or cartoon-like) and inanimate objects. Hallucinations most commonly occur daily or weekly, and upon waking. These hallucinations most often last several minutes, though they can last just a few seconds or for hours. Hallucinations are usually emotionally neutral, but most patients report feeling confused by their appearance and having a fear of underlying psychiatric disease. They often gain insight to the unreal nature of the hallucinations after counseling.48
Visual hallucinations at the sleep/wake interface
Hypnagogic and hypnopompic hallucinations are fleeting perceptual experiences that occur while an individual is falling asleep or waking, respectively.49 Because by definition visual hallucinations occur while the individual is fully awake, categorizing hallucination-like experiences such as hypnagogia and hypnopompia is difficult, especially since these are similar to other states in which alterations in perception are expected (namely a dream state). They are commonly associated with sleep disorders such as narcolepsy, cataplexy, and sleep paralysis.50,51 In a study of 13,057 individuals in the general population, Ohayon et al4 found the overall prevalence of hypnagogic or hypnopompic hallucinations was 24.8% (5.3% visual) and 6.6% (1.5% visual), respectively. Approximately one-third of participants reported having experienced ≥1 hallucinatory experience in their lifetime, regardless of being asleep or awake.4 There was a higher prevalence of hypnagogic/hypnopompic experiences among those who also reported daytime hallucinations or other psychotic features.
Content and associated signs/symptoms. Unfortunately, because of the frequent co-occurrence of sleep disorders and psychiatric conditions, as well as the general paucity of research, it is difficult to characterize the visual phenomenology of hypnagogic/hypnopompic hallucinations. Some evidence suggests the nature of the perception of the objects hallucinated is substantially impacted by the presence of preexisting psychotic symptoms. Insight into the reality of these hallucinations also depends upon the presence of comorbid psychiatric disease. Hypnagogic/hypnopompic hallucinations are often described as complex, colorful, vivid, and dream-like, as if the patient was in a “half sleep” state.52 They are usually described as highly detailed events involving people and/or animals, though they may be grotesque in nature. Perceived entities are often described as undergoing a transformation or being mobile in their environment. Rarely do these perceptions invoke emotion or change the patient’s beliefs. Hypnagogia/hypnopompia also often have an auditory or haptic component to them. Visual phenomena can either appear to take place within an alternative background environment or appear superimposed on the patient’s actual physical environment.
How to determine the cause
In many of the studies cited in this review, the participants had a considerable amount of psychiatric comorbidity, which makes it difficult to discriminate between pure neurologic and pure psychiatric causes of hallucinations. Though the visual content of the hallucinations (people, objects, shapes, lights) can help clinicians broadly differentiate causes, many other characteristics of both the hallucinations and the patient can help determine the cause (Table3,4,12-39,41-52). The most useful characteristics for discerning the etiology of an individual’s visual hallucinations are the patient’s age, the visual field in which the hallucination occurs, and the complexity/simplicity of the hallucination.

Patient age. Hallucinations associated with primary psychosis decrease with age. The average age of onset of migraine with aura is 21. Occipital lobe seizures occur in early childhood to age 40, but most commonly occur in the second decade.32,36 No trend in age can be reliably determined in individuals who experience hypnagogia/hypnopompia. In contrast, other potential causes of visual hallucinations, such as delirium, neurodegenerative disease, eye disease, and peduncular hallucinosis, are more commonly associated with advanced age.
Continue to: The visual field(s)
The visual field(s) in which the hallucination occurs can help differentiate possible causes in patients with seizure, brain tumor, migraine, or visual impairment. In patients with psychosis, delirium, peduncular hallucinosis, or hypnagogia/hypnopompia, hallucinations can occur in any visual field. Those with neurodegenerative disease, particularly PD, commonly describe seeing so-called passage hallucinations and presence hallucinations, which occur outside of the patient’s direct vision. Visual hallucinations associated with seizure are often unilateral (homonymous left or right hemifield), and contralateral to the affected neurologic structures in the visual neural pathway; they start in the left or right peripheral vision and gradually move to the central visual field. In hallucinations experienced by patients with brain tumors, the hallucinated entities typically appear on the visual field contralateral to the underlying tumor. Visual hallucinations seen in migraine often include a figure that moves from central vision to more lateral in the visual field. The visual hallucinations seen in eye disease (namely Charles Bonnet syndrome) are almost exclusively perceived in the visual fields affected by decreased visual acuity, though non-side-locked visual hallucinations are common in patients with age-related macular degeneration.
Content and complexity. The visual hallucinations perceived in those with psychosis, delirium, neurodegenerative disease, and sleep disorders are generally complex. These hallucinations tend to be of people, animals, scenes, or faces and include color and associated sound, with moving parts and interactivity with either the patient or the environment. These are in contrast to the simple visual hallucinations of visual cortex seizures, brain tumors, and migraine aura, which are often reported as brightly colored or black/white lights, flashes, and shapes, with or without associated auditory, olfactory, or somatic sensation. Furthermore, hallucinations due to seizure and brain tumor (also likely due to seizure) are often of brightly colored shapes and lights with curved edges, while patients with migraine more commonly report singular sparkling black/white objects with straight lines.
Bottom Line
Though there are no features known to be specific to only 1 cause of visual hallucinations, some characteristics of both the patient and the hallucinations can help direct the diagnostic differential. The most useful characteristics are the patient’s age, the visual field in which the hallucination occurs, and the complexity/ simplicity of the hallucination.
Related Resources
- Wang J, Patel D, Francois D. Elaborate hallucinations, but is it a psychotic disorder? Current Psychiatry. 2021;20(2):46-50. doi:10.12788/cp.0091
- O’Brien J, Taylor JP, Ballard C, et al. Visual hallucinations in neurological and ophthalmological disease: pathophysiology and management. J Neurol Neurosurg Psychiatry. 2020; 91(5):512-519. doi:10.1136/jnnp-2019-322702
A visual hallucination is a visual percept experienced when awake that is not elicited by an external stimulus. Historically, hallucinations have been synonymous with psychiatric disease, most notably schizophrenia; however, over recent decades, hallucinations have been categorized based on their underlying etiology as psychodynamic (primary psychiatric), psychophysiologic (primary neurologic/structural), and psychobiochemical (neurotransmitter dysfunction).1 Presently, visual hallucinations are known to be caused by a wide variety of primary psychiatric, neurologic, ophthalmologic, and chemically-mediated conditions. Despite these causes, clinically differentiating the characteristics and qualities of visual hallucinations is often a lesser-known skillset among clinicians. The utility of this skillset is important for the clinician’s ability to differentiate the expected and unexpected characteristics of visual hallucinations in patients with both known and unknown neuropsychiatric conditions.
Though many primary psychiatric and neurologic conditions have been associated with and/or known to cause visual hallucinations, this review focuses on the following grouped causes:
- Primary psychiatric causes: psychiatric disorders with psychotic features and delirium; and
- Primary neurologic causes: neurodegenerative disease/dementias, seizure disorders, migraine disorders, vision loss, peduncular hallucinosis, and hypnagogic/hypnopompic phenomena.
Because the accepted definition of visual hallucinations excludes visual percepts elicited by external stimuli, drug-induced hallucinations would not qualify for either of these categories. Additionally, most studies reporting on the effects of drug-induced hallucinations did not control for underlying comorbid psychiatric conditions, dementia, or delirium, and thus the results cannot be attributed to the drug alone, nor is it possible to identify reliable trends in the properties of the hallucinations.2 The goals of this review are to characterize visual hallucinations experienced as a result of primary psychiatric and primary neurologic conditions and describe key grouping and differentiating features to help guide the diagnosis.
Visual hallucinations in the general population
A review of 6 studies (N = 42,519) reported that the prevalence of visual hallucinations in the general population is 7.3%.3 The prevalence decreases to 6% when visual hallucinations arising from physical illness or drug/chemical consumption are excluded. The prevalence of visual hallucinations in the general population has been associated with comorbid anxiety, stress, bereavement, and psychotic pathology.4,5 Regarding the age of occurrence of visual hallucinations in the general population, there appears to be a bimodal distribution.3 One peak appears in later adolescence and early adulthood, which corresponds with higher rates of psychosis, and another peak occurs late in life, which corresponds to a higher prevalence of neurodegenerative conditions and visual impairment.
Primary psychiatric causes
Most studies of visual hallucinations in primary psychiatric conditions have specifically evaluated patients with schizophrenia and mood disorders with psychotic features.6,7 In a review of 29 studies (N = 5,873) that specifically examined visual hallucinations in individuals diagnosed with schizophrenia, Waters et al3 found a wide range of reported prevalence (4% to 65%) and a weighted mean prevalence of 27%. In contrast, the prevalence of auditory hallucinations in these participants ranged from 25% to 86%, with a weighted mean of 59%.3
Hallucinations are a known but less common symptom of mood disorders that present with psychotic features.8 Waters et al3 also examined the prevalence of visual and auditory hallucinations in mood disorders (including mania, bipolar disorder, and depression) reported in 12 studies (N = 2,892).3 They found the prevalence of visual hallucinations in patients with mood disorders ranged from 6% to 27%, with a weighted mean of 15%, compared to the weighted mean of 28% who experienced auditory hallucinations. Visual hallucinations in primary psychiatric conditions are associated with more severe disease, longer hospitalizations, and poorer prognoses.9-11
Visual hallucinations of psychosis
In patients with psychotic symptoms, the characteristics of the visually hallucinated entity as well as the cognitive and emotional perception of the hallucinations are notably different than in patients with other, nonpsychiatric causes of visual hallucations.3
Continue to: Content and perceived physical properties
Content and perceived physical properties. Hallucinated entities are most often perceived as solid, 3-dimensional, well-detailed, life-sized people, animals, and objects (often fire) or events existing in the real world.3 The entity is almost always perceived as real, with accurate form and color, fine edges, and shadow; is often out of reach of the perceiver; and can be stationary or moving within the physical properties of the external environment.3
Timing and triggers. The temporal properties vary widely. Hallucinations can last from seconds to minutes and occur at any time of day, though by definition, they must occur while the individual is awake.3 Visual hallucinations in psychosis are more common during times of acute stress, strong emotions, and tiredness.3
Patient reaction and belief. Because of realistic qualities of the visual hallucination and the perception that it is real, patients commonly attempt to participate in some activity in relation to the hallucination, such as moving away from or attempting to interact with it.3 Additionally, patients usually perceive the hallucinated entity as uncontrollable, and are surprised when the entity appears or disappears. Though the content of the hallucination is usually impersonal, the meaning the patient attributes to the presence of the hallucinated entity is usually perceived as very personal and often requiring action. The hallucination may represent a harbinger, sign, or omen, and is often interpreted religiously or spiritually and accompanied by comorbid delusions.3
Visual hallucinations of delirium
Delirium is a syndrome of altered mentation—most notably consciousness, attention, and orientation—that occurs as a result of ≥1 metabolic, infectious, drug-induced, or other medical conditions and often manifests as an acute secondary psychotic illness.12 Multiple patient and environmental characteristics have been identified as risk factors for developing delirium, including multiple and/or severe medical illnesses, preexisting dementia, depression, advanced age, polypharmacy, having an indwelling urinary catheter, impaired sight or hearing, and low albumin levels.13-15 The development of delirium is significantly and positively associated with regular alcohol use, benzodiazepine withdrawal, and angiotensin receptor blocker and dopamine receptor agonist usage.15 Approximately 40% of patients with delirium have symptoms of psychosis, and in contrast to the hallucinations experienced by patients with schizophrenia, visual hallucinations are the most common type of hallucinations seen in delirium (27%).13 In a 2021 review that included 602 patients with delirium, Tachibana et al15 found that approximately 26% experienced hallucinations, 92% of which were visual hallucinations.
Content, perceived physical properties, and reaction. Because of the limited attention and cognitive function of patients with delirium, less is known about the content of their visual hallucinations. However, much like those with primary psychotic symptoms, patients with delirium often report seeing complex, normal-sized, concrete entities, most commonly people. Tachibana et al15 found that the hallucinated person is more often a stranger than a familiar person, but (rarely) may be an ethereal being such as a devil or ghost. The next most common visually hallucinated entities were creatures, most frequently insects and animals. Other common hallucinations were visions of events or objects, such as fires, falling ceilings, or water. Similar to those with primary psychotic illness such as schizophrenia, patients with delirium often experience emotional distress, anxiety, fear, and confusion in response to the hallucinated person, object, and/or event.15
Continue to: Primary neurologic causes
Primary neurologic causes
Visual hallucinations in neurodegenerative diseases
Patients with neurodegenerative diseases such as Parkinson disease (PD), dementia with Lewy bodies (DLB), or Creutzfeldt-Jakob disease (CJD) commonly experience hallucinations as a feature of their condition. However, the true cause of these hallucinations often cannot be directly attributed to any specific pathophysiology because these patients often have multiple coexisting risk factors, such as advanced age, major depressive disorder, use of neuroactive medications, and co-occurring somatic illness. Though the prevalence of visual hallucinations varies widely between studies, with 15% to 40% reported in patients with PD, the prevalence roughly doubles in patients with PD-associated dementia (30% to 60%), and is reported by 60% to 90% of those with DLB.16-18 Hallucinations are generally thought to be less common in Alzheimer disease; such patients most commonly experience visual hallucinations, although the reported prevalence ranges widely (4% to 59%).19,20 Notably, similarly to hallucinations experienced in patients with delirium, and in contrast to those with psychosis, visual hallucinations are more common than auditory hallucinations in neurodegenerative diseases.20 Hallucinations are not common in individuals with CJD but are a key defining feature of the He
Content, perceived physical properties, and reaction. Similar to the visual hallucinations experienced by patients with psychosis or delirium, those experienced in patients with PD, DLB, or CJD are often complex, most commonly of people, followed by animals and objects. The presence of “passage hallucinations”—in which a person or animal is seen in a patient’s peripheral vision, but passes out of their visual field before the entity can be directly visualized—is common.20 Those with PD also commonly have visual hallucinations in which the form of an object appears distorted (dysmorphopsia) or the color of an object appears distorted (metachromatopsia), though these would better be classified as illusions because a real object is being perceived with distortion.22
Hallucinations are more common in the evening and at night. “Presence hallucinations” are a common type of hallucination that cannot be directly related to a specific sensory modality such as vision, though they are commonly described by patients with PD as a seen or perceived image (usually a person) that is not directly in the individual’s visual field.17 These presence hallucinations are often described as being behind the patient or in a visualized scene of what was about to happen. Before developing the dementia and myoclonus also seen in sporadic CJD, patients with the Heidenhain variant of CJD describe illusions such as metachromatopsia, dysmorphia, and micropsia that eventually develop into frank visual hallucinations, which have been poorly reported in medical literature.22,23 There are no generalizable trends in the temporal nature of visual hallucinations in patients with neurodegenerative diseases. In most cases of visual hallucinations in patients with PD and dementia, insight relating to the perception varies widely based on the patient’s cognitive status. Subsequently, patients’ reactions to the hallucinations also vary widely.
Visual hallucinations in epileptic seizures
Occipital lobe epilepsies represent 1% to 4.6% of all epilepsies; however, these represent 20% to 30% of benign childhood partial epilepsies.24,25 These are commonly associated with various types of visual hallucinations depending upon the location of the seizure onset within the occipital lobe. These are referred to as visual auras.26 Visual auras are classified into simple visual hallucinations, complex visual hallucinations, visual illusions, and ictal amaurosis (hemifield blindness or complete blindness).
Content, perceived physical properties, and reaction. Simple visual hallucinations are often described as brief, stereotypical flashing lights of various shapes and colors. These images may flicker, change shape, or take on a geometric or irregular pattern. Appearances can be repetitive and stereotyped, are often reported as moving horizontally from the periphery to the center of the visual field, and can spread to the entire visual field. Most often, these hallucinations occur for 5 to 30 seconds, and have no discernible provoking factors. Complex visual hallucinations consist of formed images of animals, people, or elaborate scenes. These are believed to reflect activation of a larger area of cortex in the temporo-parieto-occipital region, which is the visual association cortex. Very rarely, occipital lobe seizures can manifest with ictal amaurosis.24
Continue to: Simple visual auras...
Simple visual auras have a very high localizing value to the occipital lobe. The primary visual cortex (Brodmann area 17) is situated in the banks of calcarine fissure and activation of this region produces these simple hallucinations. If the hallucinations are consistently lateralized, the seizures are very likely to be coming from the contralateral occipital lobe.
Visual hallucinations in brain tumors
In general, a tumor anywhere along the optic path can produce visual hallucinations; however, the exact causal mechanism of the hallucinations is unknown. Moreover, tumors in different locations—namely the occipital lobes, temporal lobes, and frontal lobes—appear to produce visual hallucinations with substantially different characteristics.27-29 Further complicating the search for the mechanism of these hallucinations is the fact that tumors are epileptogenic. In addition, 36% to 48% of patients with brain tumors have mood symptoms (depression/mania), and 22% to 24% have psychotic symptoms (delusions/hallucinations); these symptoms are considerably location-dependent.30-32
Content and associated signs/symptoms. There are some grouped symptoms and/or hallucination characteristics associated with cerebral tumors in different lobes of the brain, though these symptoms are not specific. The visual hallucinations associated with brain tumors are typically confined to the field of vision that corresponds to the location of the tumor. Additionally, many such patients have a baseline visual field defect to some extent due to the tumor location.
In patients with occipital lobe tumors, visual hallucinations closely resemble those experienced in occipital lobe seizures, specifically bright flashes of light in colorful simple and complex shapes. Interestingly, those with occipital lobe tumors report xanthopsia, a form of chromatopsia in which objects in their field of view appear abnormally colored a yellowish shade.26,27
In patients with temporal lobe tumors, more complex visual hallucinations of people, objects, and events occurring around them are often accompanied by auditory hallucinations, olfactory hallucinations, and/or anosmia.28In those with frontal lobe tumors, similar complex visual hallucinations of people, objects, and events are seen, and olfactory hallucinations and/or anosmia are often experienced. However, these patients often have a lower likelihood of experiencing auditory hallucinations, and a higher likelihood of developing personality changes and depression than other psychotic symptoms. The visual hallucinations experienced in those with frontal lobe tumors are more likely to have violent content.29
Continue to: Visual hallucinations in migraine with aura
Visual hallucinations in migraine with aura
The estimated prevalence of migraine in the general population is 15% to 29%; 31% of those with migraine experience auras.33-35 Approximately 99% of those with migraine auras experience some type of associated visual phenomena.33,36 The pathophysiology of migraine is believed to be related to spreading cortical depression, in which a slowly propagating wave of neuroelectric depolarization travels over the cortex, followed by a depression of normal brain activity. Visual aura is thought to occur due to the resulting changes in cortical activity in the visual cortex; however, the exact electrophysiology of visual migraine aura is not entirely known.37,38 Though most patients with visual migraine aura experience simple visual hallucinations, complex hallucinations have been reported in the (very rare) cases of migraine coma and familial hemiplegic migraine.39
Content and associated signs/symptoms. The most common hallucinated entities reported by patients with migraine with aura are zigzag, flashing/sparkling, black and white curved figure(s) in the center of the visual field, commonly called a scintillating phosphene or scintillating scotoma.36 The perceived entity is often singular and gradually moves from the center to the periphery of the visual field. These visual hallucinations appear in front of all other objects in the visual field and do not interact with the environment or observer, or resemble or morph into any real-world objects, though they may change in contour, size, and color. The scintillating nature of the hallucination often resolves within minutes, usually leaving a scotoma, or area of vision loss, in the area, with resolution back to baseline vision within 1 hour. The straight, zigzag, and usually black-and-white nature of the scintillating phosphenes of migraine are in notable contrast to the colorful, often circular visual hallucinations experienced in patients with occipital lobe seizures.25
Visual hallucinations in peduncular hallucinosis
Peduncular hallucinosis is a syndrome of predominantly dreamlike visual hallucinations that occurs in the setting of lesions in the midbrain and/or thalamus.40 A recent review of the lesion etiology found that approximately 63% are caused by focal infarction and approximately 15% are caused by mass lesions; subarachnoid hemorrhage, intracerebral hemorrhage, and demyelination cause approximately 5% of cases each.40 Additionally, a review of the affected brainstem anatomy showed almost all lesions were found in the paramedian reticular formations of the midbrain and pons, with the vast majority of lesions affecting or adjacent to the oculomotor and raphe nuclei of the midbrain.39 Due to the commonly involved visual pathway, some researchers have suggested these hallucinations may be the result of a release phenomenon.39
Content and associated signs/symptoms. The visual hallucinations of peduncular hallucinosis usually start 1 to 5 days after the causal lesion forms, last several minutes to hours, and most stop after 1 to 3 weeks; however, cases of hallucinations lasting for years have been reported. These hallucinations have a diurnal pattern of usually appearing while the patient is resting in the evening and/or preparing for sleep. The characteristics of visual hallucinations vary widely from simple distortions in how real objects appear to colorful and vivid hallucinated events and people who can interact with the observer. The content of the visual hallucinations often changes in nature during the hallucination, or from one hallucination to the next. The hallucinated entities can be worldly or extraterrestrial. Once these patients fall asleep, they often have equally vivid and unusual dreams, with content similar to their visual hallucinations. Due to the anatomical involvement of the nigrostriatal pathway and oculomotor nuclei, co-occurring parkinsonism, ataxia, and oculomotor nerve palsy are common and can be a key clinical feature in establishing the diagnosis. Though patients with peduncular hallucinations commonly fear their hallucinations, they often eventually gain insight, which eases their anxiety.39
Other causes
Visual hallucinations in visual impairment
Visual hallucinations are a diagnostic requirement for Charles Bonnet syndrome, in which individuals with vision loss experience visual hallucinations in the corresponding field of vision loss.41 A lesion at any point in the visual pathway that produces visual loss can lead to Charles Bonnet syndrome; however, age-related macular degeneration is the most common cause.42 The hallucinations of Charles Bonnet syndrome are believed to be a release phenomenon, given the defective visual pathway and resultant dysfunction in visual processing. The prevalence of Charles Bonnet syndrome ranges widely by study. Larger studies report a prevalence of 11% to 27% in patients with age-related macular degeneration, depending on the severity of vision loss.43,44 Because there are many causes of Charles Bonnet syndrome, and because a recent study found that only 15% of patients with this syndrome told their eye care clinician and that 21% had not reported their hallucinatory symptoms to anyone, the true prevalence is unknown.42 Though the onset of visual hallucinations correlates with the onset of vision loss, there appears to be no association between the nature or complexity of the hallucinations and the severity or progression of the patient’s vision loss.45 Some studies have reported either the onset of or a higher frequency of visual hallucinations at a time of visual recovery (for example, treatment or exudative age-related macular degeneration), which suggests that hallucinations may be triggered by fluctuations in visual acuity.46,47 Additional risk factors for experiencing visual hallucinations in the setting of visual pathway deficit include a history of stroke, social isolation, poor cognitive function, poor lighting, and age ≥65.
Continue to: Content and associated signs/symptoms
Content and associated signs/symptoms. The visual hallucinations of patients with Charles Bonnet syndrome appear almost exclusively in the defective visual field. Images tend to be complex, colored, with moving parts, and appear in front of the patient. The hallucinations are usually of familiar or normal-appearing people or mundane objects, and as such, the patient often does not realize the hallucinated entity is not real. In patients without comorbid psychiatric disease, visual hallucinations are not accompanied by any other types of hallucinations. The most commonly hallucinated entities are people, followed by simple visual hallucinations of geometric patterns, and then by faces (natural or cartoon-like) and inanimate objects. Hallucinations most commonly occur daily or weekly, and upon waking. These hallucinations most often last several minutes, though they can last just a few seconds or for hours. Hallucinations are usually emotionally neutral, but most patients report feeling confused by their appearance and having a fear of underlying psychiatric disease. They often gain insight to the unreal nature of the hallucinations after counseling.48
Visual hallucinations at the sleep/wake interface
Hypnagogic and hypnopompic hallucinations are fleeting perceptual experiences that occur while an individual is falling asleep or waking, respectively.49 Because by definition visual hallucinations occur while the individual is fully awake, categorizing hallucination-like experiences such as hypnagogia and hypnopompia is difficult, especially since these are similar to other states in which alterations in perception are expected (namely a dream state). They are commonly associated with sleep disorders such as narcolepsy, cataplexy, and sleep paralysis.50,51 In a study of 13,057 individuals in the general population, Ohayon et al4 found the overall prevalence of hypnagogic or hypnopompic hallucinations was 24.8% (5.3% visual) and 6.6% (1.5% visual), respectively. Approximately one-third of participants reported having experienced ≥1 hallucinatory experience in their lifetime, regardless of being asleep or awake.4 There was a higher prevalence of hypnagogic/hypnopompic experiences among those who also reported daytime hallucinations or other psychotic features.
Content and associated signs/symptoms. Unfortunately, because of the frequent co-occurrence of sleep disorders and psychiatric conditions, as well as the general paucity of research, it is difficult to characterize the visual phenomenology of hypnagogic/hypnopompic hallucinations. Some evidence suggests the nature of the perception of the objects hallucinated is substantially impacted by the presence of preexisting psychotic symptoms. Insight into the reality of these hallucinations also depends upon the presence of comorbid psychiatric disease. Hypnagogic/hypnopompic hallucinations are often described as complex, colorful, vivid, and dream-like, as if the patient was in a “half sleep” state.52 They are usually described as highly detailed events involving people and/or animals, though they may be grotesque in nature. Perceived entities are often described as undergoing a transformation or being mobile in their environment. Rarely do these perceptions invoke emotion or change the patient’s beliefs. Hypnagogia/hypnopompia also often have an auditory or haptic component to them. Visual phenomena can either appear to take place within an alternative background environment or appear superimposed on the patient’s actual physical environment.
How to determine the cause
In many of the studies cited in this review, the participants had a considerable amount of psychiatric comorbidity, which makes it difficult to discriminate between pure neurologic and pure psychiatric causes of hallucinations. Though the visual content of the hallucinations (people, objects, shapes, lights) can help clinicians broadly differentiate causes, many other characteristics of both the hallucinations and the patient can help determine the cause (Table3,4,12-39,41-52). The most useful characteristics for discerning the etiology of an individual’s visual hallucinations are the patient’s age, the visual field in which the hallucination occurs, and the complexity/simplicity of the hallucination.

Patient age. Hallucinations associated with primary psychosis decrease with age. The average age of onset of migraine with aura is 21. Occipital lobe seizures occur in early childhood to age 40, but most commonly occur in the second decade.32,36 No trend in age can be reliably determined in individuals who experience hypnagogia/hypnopompia. In contrast, other potential causes of visual hallucinations, such as delirium, neurodegenerative disease, eye disease, and peduncular hallucinosis, are more commonly associated with advanced age.
Continue to: The visual field(s)
The visual field(s) in which the hallucination occurs can help differentiate possible causes in patients with seizure, brain tumor, migraine, or visual impairment. In patients with psychosis, delirium, peduncular hallucinosis, or hypnagogia/hypnopompia, hallucinations can occur in any visual field. Those with neurodegenerative disease, particularly PD, commonly describe seeing so-called passage hallucinations and presence hallucinations, which occur outside of the patient’s direct vision. Visual hallucinations associated with seizure are often unilateral (homonymous left or right hemifield), and contralateral to the affected neurologic structures in the visual neural pathway; they start in the left or right peripheral vision and gradually move to the central visual field. In hallucinations experienced by patients with brain tumors, the hallucinated entities typically appear on the visual field contralateral to the underlying tumor. Visual hallucinations seen in migraine often include a figure that moves from central vision to more lateral in the visual field. The visual hallucinations seen in eye disease (namely Charles Bonnet syndrome) are almost exclusively perceived in the visual fields affected by decreased visual acuity, though non-side-locked visual hallucinations are common in patients with age-related macular degeneration.
Content and complexity. The visual hallucinations perceived in those with psychosis, delirium, neurodegenerative disease, and sleep disorders are generally complex. These hallucinations tend to be of people, animals, scenes, or faces and include color and associated sound, with moving parts and interactivity with either the patient or the environment. These are in contrast to the simple visual hallucinations of visual cortex seizures, brain tumors, and migraine aura, which are often reported as brightly colored or black/white lights, flashes, and shapes, with or without associated auditory, olfactory, or somatic sensation. Furthermore, hallucinations due to seizure and brain tumor (also likely due to seizure) are often of brightly colored shapes and lights with curved edges, while patients with migraine more commonly report singular sparkling black/white objects with straight lines.
Bottom Line
Though there are no features known to be specific to only 1 cause of visual hallucinations, some characteristics of both the patient and the hallucinations can help direct the diagnostic differential. The most useful characteristics are the patient’s age, the visual field in which the hallucination occurs, and the complexity/ simplicity of the hallucination.
Related Resources
- Wang J, Patel D, Francois D. Elaborate hallucinations, but is it a psychotic disorder? Current Psychiatry. 2021;20(2):46-50. doi:10.12788/cp.0091
- O’Brien J, Taylor JP, Ballard C, et al. Visual hallucinations in neurological and ophthalmological disease: pathophysiology and management. J Neurol Neurosurg Psychiatry. 2020; 91(5):512-519. doi:10.1136/jnnp-2019-322702
1. Asaad G, Shapiro B. Hallucinations: theoretical and clinical overview. Am J Psychiatry. 1987;143(9):1088-1097.
2. Taam MA, Boissieu P, Taam RA, et al. Drug-induced hallucination: a case/non-case study in the French Pharmacovigilance Database. Article in French. Eur J Psychiatry. 2015;29(1):21-31.
3. Waters F, Collerton D, Ffytche DH, et al. Visual hallucinations in the psychosis spectrum and comparative information from neurodegenerative disorders and disease. Schizophr Bull. 2014;40(Suppl 4):S233-S245.
4. Ohayon MM. Prevalence of hallucinations and their pathological associations in the general population. Psychiatry Res. 2000;97(2-3):153-164.
5. Rees WD. The hallucinations of widowhood. Br Med J. 1971;4(5778):37-41.
6. Delespaul P, deVries M, van Os J. Determinants of occurrence and recovery from hallucinations in daily life. Soc Psychiatry Psychiatr Epidemiol. 2002;37(3):97-104.
7. Gauntlett-Gilbert J, Kuipers E. Phenomenology of visual hallucinations in psychiatric conditions. J Nerv Ment Dis. 2003;191(3):203-205.
8. Goodwin FK, Jamison KR. Manic Depressive Illness. Oxford University Press, Inc.; 1999.
9. Mueser KT, Bellack AS, Brady EU. Hallucinations in schizophrenia. Acta Psychiatr Scand. 1990;82(1):26-29.
10. McCabe MS, Fowler RC, Cadoret RJ, et al. Symptom differences in schizophrenia with good and bad prognosis. Am J Psychiatry. 1972;128(10):1239-1243.
11. Baethge C, Baldessarini RJ, Freudenthal K, et al. Hallucinations in bipolar disorder: characteristics and comparison to unipolar depression and schizophrenia. Bipolar Disord. 2005;7(2):136-145.
12. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Publishing; 2013.
13. Ahmed S, Leurent B, Sampson EL. Risk factors for incident delirium among older people in acute hospital medical units: a systematic review and meta-analysis. Age Ageing. 2014;43(3):326-333.
14. Webster R, Holroyd S. Prevalence of psychotic symptoms in delirium. Psychosomatics. 2000;41(6):519-522.
15. Tachibana M, Inada T, Ichida M, et al. Factors affecting hallucinations in patients with delirium. Sci Rep. 2021;11(1):13005. doi:10.1038/s41598-021-92578-1
16. Fenelon G, Mahieux F, Huon R, et al. Hallucinations in Parkinson’s disease: prevalence, phenomenology and risk factors. Brain. 2000;123(Pt 4):733-745.
17. Papapetropoulos S, Argyriou AA, Ellul J. Factors associated with drug-induced visual hallucinations in Parkinson’s disease. J Neurol. 2005;252(10):1223-1228.
18. Williams DR, Warren JD, Lees AJ. Using the presence of visual hallucinations to differentiate Parkinson’s disease from atypical parkinsonism. J Neurol Neurosurg Psychiatry. 2008;79(6):652-655.
19. Linszen MMJ, Lemstra AW, Dauwan M, et al. Understanding hallucinations in probable Alzheimer’s disease: very low prevalence rates in a tertiary memory clinic. Alzheimers Dement (Amst). 2018;10:358-362.
20. Burghaus L, Eggers C, Timmermann L, et al. Hallucinations in neurodegenerative diseases. CNS Neurosci Ther. 2012;18(2):149-159.
21. Brar HK, Vaddigiri V, Scicutella A. Of illusions, hallucinations, and Creutzfeldt-Jakob disease (Heidenhain’s variant). J Neuropsychiatry Clin Neurosci. 2005;17(1):124-126.
22. Sasaki C, Yokoi K, Takahashi H, et al. Visual illusions in Parkinson’s disease: an interview survey of symptomatology. Psychogeriatrics. 2022;22(1):28-48.
23. Kropp S, Schulz-Schaeffer WJ, Finkenstaedt M, et al. The Heidenhain variant of Creutzfeldt-Jakob disease. Arch Neurol. 1999;56(1):55-61.
24. Taylor I, Scheffer IE, Berkovic SF. Occipital epilepsies: identification of specific and newly recognized syndromes. Brain. 2003;126(Pt 4):753-769.
25. Caraballo R, Cersosimo R, Medina C, et al. Panayiotopoulos-type benign childhood occipital epilepsy: a prospective study. Neurology. 2000;5(8):1096-1100.
26. Chowdhury FA, Silva R, Whatley B, et al. Localisation in focal epilepsy: a practical guide. Practical Neurol. 2021;21(6):481-491.
27. Horrax G, Putnam TJ. Distortions of the visual fields in cases of brain tumour: the field defects and hallucinations produced by tumours of the occipital lobe. Brain. 1932;55(4):499-523.
28. Cushing H. Distortions of the visual fields in cases of brain tumor (6th paper): the field defects produced by temporal lobe lesions. Brain. 1922;44(4):341-396.
29. Fornazzari L, Farcnik K, Smith I, et al. Violent visual hallucinations and aggression in frontal lobe dysfunction: clinical manifestations of deep orbitofrontal foci. J Neuropsychiatry Clin Neurosci. 1992;4(1):42-44.
30. Madhusoodanan S, Opler MGA, Moise D, et al. Brain tumor location and psychiatric symptoms: is there an association? A meta-analysis of published cases studies. Expert Rev Neurother. 2010;10(10):1529-1536.
31. Madhusoodanan S, Sinha A, Moise D. Brain tumors and psychiatric manifestations: a review and analysis. Poster presented at: The American Association for Geriatric Psychiatry Annual Meeting; March 10-13; 2006; San Juan, Puerto Rico.
32. Madhusoodanan S, Danan D, Moise D. Psychiatric manifestations of brain tumors/gliomas. Rivistica Medica. 2007;13(4):209-215.
33. Kirchmann M. Migraine with aura: new understanding from clinical epidemiological studies. Curr Opin Neurol. 2006;19:286-293.
34. Goadsby PJ, Lipton RB, Ferrari MD. Migraine: current understanding and treatment. N Engl J Med. 2002;346(4):257-270.
35. Waters WE, O’Connor PJ. Prevalence of migraine. J Neurol Neurosurg Psychiatry. 1975;38(6):613-616.
36. Russell MB, Olesen J. A nosographic analysis of the migraine aura in a general population. Brain. 1996;119(Pt 2):355-361.
37. Cozzolino O, Marchese M, Trovato F, et al. Understanding spreading depression from headache to sudden unexpected death. Front Neurol. 2018;9:19.
38. Hadjikhani N, Sanchez del Rio M, Wu O, et al. Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci U S A. 2001;98(8):4687-4692.
39. Manford M, Andermann F. Complex visual hallucinations. Clinical and neurobiological insights. Brain. 1998;121(Pt 10):1819-1840.
40. Galetta KM, Prasad S. Historical trends in the diagnosis of peduncular hallucinosis. J Neuroophthalmol. 2018;38(4):438-441.
41. Schadlu AP, Schadlu R, Shepherd JB III. Charles Bonnet syndrome: a review. Curr Opin Ophthalmol. 2009;20(3):219-222.
42. Vukicevic M, Fitzmaurice K. Butterflies and black lace patterns: the prevalence and characteristics of Charles Bonnet hallucinations in an Australian population. Clin Exp Ophthalmol. 2008;36(7):659-665.
43. Teunisse RJ, Cruysberg JR, Verbeek A, et al. The Charles Bonnet syndrome: a large prospective study in the Netherlands. A study of the prevalence of the Charles Bonnet syndrome and associated factors in 500 patients attending the University Department of Ophthalmology at Nijmegen. Br J Psychiatry. 1995;166(2):254-257.
44. Holroyd S, Rabins PV, Finkelstein D, et al. Visual hallucination in patients with macular degeneration. Am J Psychiatry. 1992;149(12):1701-1706.
45. Khan JC, Shahid H, Thurlby DA, et al. Charles Bonnet syndrome in age-related macular degeneration: the nature and frequency of images in subjects with end-stage disease. Ophthalmic Epidemiol. 2008;15(3):202-208.
46. Cohen SY, Bulik A, Tadayoni R, et al. Visual hallucinations and Charles Bonnet syndrome after photodynamic therapy for age related macular degeneration. Br J Ophthalmol. 2003;87(8):977-979.
47. Meyer CH, Mennel S, Horle S, et al. Visual hallucinations after intravitreal injection of bevacizumab in vascular age-related macular degeneration. Am J Ophthalmol. 2007;143(1):169-170.
48. Jan T, Del Castillo J. Visual hallucinations: Charles Bonnet syndrome. West J Emerg Med. 2012;13(6):544-547. doi:10.5811/westjem.2012.7.12891
49. Foulkes D, Vogel G. Mental activity at sleep onset. J Abnorm Psychol. 1965;70:231-243.
50. Mitler MM, Hajdukovic R, Erman M, et al. Narcolepsy. J Clin Neurophysiol. 1990;7(1):93-118.
51. Nishino S. Clinical and neurobiological aspects of narcolepsy. Sleep Med. 2007;8(4):373-399.
52. Schultz SK, Miller DD, Oliver SE, et al. The life course of schizophrenia: age and symptom dimensions. Schizophr Res. 1997;23(1):15-23.
1. Asaad G, Shapiro B. Hallucinations: theoretical and clinical overview. Am J Psychiatry. 1987;143(9):1088-1097.
2. Taam MA, Boissieu P, Taam RA, et al. Drug-induced hallucination: a case/non-case study in the French Pharmacovigilance Database. Article in French. Eur J Psychiatry. 2015;29(1):21-31.
3. Waters F, Collerton D, Ffytche DH, et al. Visual hallucinations in the psychosis spectrum and comparative information from neurodegenerative disorders and disease. Schizophr Bull. 2014;40(Suppl 4):S233-S245.
4. Ohayon MM. Prevalence of hallucinations and their pathological associations in the general population. Psychiatry Res. 2000;97(2-3):153-164.
5. Rees WD. The hallucinations of widowhood. Br Med J. 1971;4(5778):37-41.
6. Delespaul P, deVries M, van Os J. Determinants of occurrence and recovery from hallucinations in daily life. Soc Psychiatry Psychiatr Epidemiol. 2002;37(3):97-104.
7. Gauntlett-Gilbert J, Kuipers E. Phenomenology of visual hallucinations in psychiatric conditions. J Nerv Ment Dis. 2003;191(3):203-205.
8. Goodwin FK, Jamison KR. Manic Depressive Illness. Oxford University Press, Inc.; 1999.
9. Mueser KT, Bellack AS, Brady EU. Hallucinations in schizophrenia. Acta Psychiatr Scand. 1990;82(1):26-29.
10. McCabe MS, Fowler RC, Cadoret RJ, et al. Symptom differences in schizophrenia with good and bad prognosis. Am J Psychiatry. 1972;128(10):1239-1243.
11. Baethge C, Baldessarini RJ, Freudenthal K, et al. Hallucinations in bipolar disorder: characteristics and comparison to unipolar depression and schizophrenia. Bipolar Disord. 2005;7(2):136-145.
12. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Publishing; 2013.
13. Ahmed S, Leurent B, Sampson EL. Risk factors for incident delirium among older people in acute hospital medical units: a systematic review and meta-analysis. Age Ageing. 2014;43(3):326-333.
14. Webster R, Holroyd S. Prevalence of psychotic symptoms in delirium. Psychosomatics. 2000;41(6):519-522.
15. Tachibana M, Inada T, Ichida M, et al. Factors affecting hallucinations in patients with delirium. Sci Rep. 2021;11(1):13005. doi:10.1038/s41598-021-92578-1
16. Fenelon G, Mahieux F, Huon R, et al. Hallucinations in Parkinson’s disease: prevalence, phenomenology and risk factors. Brain. 2000;123(Pt 4):733-745.
17. Papapetropoulos S, Argyriou AA, Ellul J. Factors associated with drug-induced visual hallucinations in Parkinson’s disease. J Neurol. 2005;252(10):1223-1228.
18. Williams DR, Warren JD, Lees AJ. Using the presence of visual hallucinations to differentiate Parkinson’s disease from atypical parkinsonism. J Neurol Neurosurg Psychiatry. 2008;79(6):652-655.
19. Linszen MMJ, Lemstra AW, Dauwan M, et al. Understanding hallucinations in probable Alzheimer’s disease: very low prevalence rates in a tertiary memory clinic. Alzheimers Dement (Amst). 2018;10:358-362.
20. Burghaus L, Eggers C, Timmermann L, et al. Hallucinations in neurodegenerative diseases. CNS Neurosci Ther. 2012;18(2):149-159.
21. Brar HK, Vaddigiri V, Scicutella A. Of illusions, hallucinations, and Creutzfeldt-Jakob disease (Heidenhain’s variant). J Neuropsychiatry Clin Neurosci. 2005;17(1):124-126.
22. Sasaki C, Yokoi K, Takahashi H, et al. Visual illusions in Parkinson’s disease: an interview survey of symptomatology. Psychogeriatrics. 2022;22(1):28-48.
23. Kropp S, Schulz-Schaeffer WJ, Finkenstaedt M, et al. The Heidenhain variant of Creutzfeldt-Jakob disease. Arch Neurol. 1999;56(1):55-61.
24. Taylor I, Scheffer IE, Berkovic SF. Occipital epilepsies: identification of specific and newly recognized syndromes. Brain. 2003;126(Pt 4):753-769.
25. Caraballo R, Cersosimo R, Medina C, et al. Panayiotopoulos-type benign childhood occipital epilepsy: a prospective study. Neurology. 2000;5(8):1096-1100.
26. Chowdhury FA, Silva R, Whatley B, et al. Localisation in focal epilepsy: a practical guide. Practical Neurol. 2021;21(6):481-491.
27. Horrax G, Putnam TJ. Distortions of the visual fields in cases of brain tumour: the field defects and hallucinations produced by tumours of the occipital lobe. Brain. 1932;55(4):499-523.
28. Cushing H. Distortions of the visual fields in cases of brain tumor (6th paper): the field defects produced by temporal lobe lesions. Brain. 1922;44(4):341-396.
29. Fornazzari L, Farcnik K, Smith I, et al. Violent visual hallucinations and aggression in frontal lobe dysfunction: clinical manifestations of deep orbitofrontal foci. J Neuropsychiatry Clin Neurosci. 1992;4(1):42-44.
30. Madhusoodanan S, Opler MGA, Moise D, et al. Brain tumor location and psychiatric symptoms: is there an association? A meta-analysis of published cases studies. Expert Rev Neurother. 2010;10(10):1529-1536.
31. Madhusoodanan S, Sinha A, Moise D. Brain tumors and psychiatric manifestations: a review and analysis. Poster presented at: The American Association for Geriatric Psychiatry Annual Meeting; March 10-13; 2006; San Juan, Puerto Rico.
32. Madhusoodanan S, Danan D, Moise D. Psychiatric manifestations of brain tumors/gliomas. Rivistica Medica. 2007;13(4):209-215.
33. Kirchmann M. Migraine with aura: new understanding from clinical epidemiological studies. Curr Opin Neurol. 2006;19:286-293.
34. Goadsby PJ, Lipton RB, Ferrari MD. Migraine: current understanding and treatment. N Engl J Med. 2002;346(4):257-270.
35. Waters WE, O’Connor PJ. Prevalence of migraine. J Neurol Neurosurg Psychiatry. 1975;38(6):613-616.
36. Russell MB, Olesen J. A nosographic analysis of the migraine aura in a general population. Brain. 1996;119(Pt 2):355-361.
37. Cozzolino O, Marchese M, Trovato F, et al. Understanding spreading depression from headache to sudden unexpected death. Front Neurol. 2018;9:19.
38. Hadjikhani N, Sanchez del Rio M, Wu O, et al. Mechanisms of migraine aura revealed by functional MRI in human visual cortex. Proc Natl Acad Sci U S A. 2001;98(8):4687-4692.
39. Manford M, Andermann F. Complex visual hallucinations. Clinical and neurobiological insights. Brain. 1998;121(Pt 10):1819-1840.
40. Galetta KM, Prasad S. Historical trends in the diagnosis of peduncular hallucinosis. J Neuroophthalmol. 2018;38(4):438-441.
41. Schadlu AP, Schadlu R, Shepherd JB III. Charles Bonnet syndrome: a review. Curr Opin Ophthalmol. 2009;20(3):219-222.
42. Vukicevic M, Fitzmaurice K. Butterflies and black lace patterns: the prevalence and characteristics of Charles Bonnet hallucinations in an Australian population. Clin Exp Ophthalmol. 2008;36(7):659-665.
43. Teunisse RJ, Cruysberg JR, Verbeek A, et al. The Charles Bonnet syndrome: a large prospective study in the Netherlands. A study of the prevalence of the Charles Bonnet syndrome and associated factors in 500 patients attending the University Department of Ophthalmology at Nijmegen. Br J Psychiatry. 1995;166(2):254-257.
44. Holroyd S, Rabins PV, Finkelstein D, et al. Visual hallucination in patients with macular degeneration. Am J Psychiatry. 1992;149(12):1701-1706.
45. Khan JC, Shahid H, Thurlby DA, et al. Charles Bonnet syndrome in age-related macular degeneration: the nature and frequency of images in subjects with end-stage disease. Ophthalmic Epidemiol. 2008;15(3):202-208.
46. Cohen SY, Bulik A, Tadayoni R, et al. Visual hallucinations and Charles Bonnet syndrome after photodynamic therapy for age related macular degeneration. Br J Ophthalmol. 2003;87(8):977-979.
47. Meyer CH, Mennel S, Horle S, et al. Visual hallucinations after intravitreal injection of bevacizumab in vascular age-related macular degeneration. Am J Ophthalmol. 2007;143(1):169-170.
48. Jan T, Del Castillo J. Visual hallucinations: Charles Bonnet syndrome. West J Emerg Med. 2012;13(6):544-547. doi:10.5811/westjem.2012.7.12891
49. Foulkes D, Vogel G. Mental activity at sleep onset. J Abnorm Psychol. 1965;70:231-243.
50. Mitler MM, Hajdukovic R, Erman M, et al. Narcolepsy. J Clin Neurophysiol. 1990;7(1):93-118.
51. Nishino S. Clinical and neurobiological aspects of narcolepsy. Sleep Med. 2007;8(4):373-399.
52. Schultz SK, Miller DD, Oliver SE, et al. The life course of schizophrenia: age and symptom dimensions. Schizophr Res. 1997;23(1):15-23.
Prodromal symptoms of schizophrenia: What to look for
Schizophrenia is characterized by psychotic symptoms that typically follow a prodromal period of premonitory signs and symptoms that appear before the manifestation of the full-blown syndrome. Signs and symptoms during the prodromal phase are subsyndromal, which implies a lower degree of intensity, duration, or frequency than observed when the patient meets the full criteria for the syndrome. Early detection of prodromal symptoms can improve prognosis, but these subtle symptoms may go unrecognized.
In schizophrenia, a patient may exhibit prodromal signs and symptoms before the appearance of pathognomonic symptoms, such as delusions, hallucinations, and disorganization. The schizophrenia prodrome can be conceptualized as a period of prepsychotic disturbances depicting an alteration in the individual’s behavior and perception. Prodromal symptoms can last from weeks to years before the psychotic illness clinically manifests.1 The prodromal symptom cluster typically becomes evident during adolescence and young adulthood.2
In the mid-1990s, investigators tried to identify a “putative prodrome” for psychosis. The term “at-risk mental state” (ARMS) for psychosis is based on retrospective reports of prodromal symptoms in first-episode psychosis. Over the next 2 decades, scales such as the Comprehensive Assessment of ARMS (CAARMS)3 and the Structured Interview for Prodromal Syndrome4 were designed to enhance the objectivity and diagnostic accuracy of the ARMS. These scales have reasonable interrater reliability.5
Researchers also have attempted to stage the severity of ARMS.6 Key symptom group predictors were studied to determine which individual symptoms or cluster of symptoms are most associated with poor outcomes and progression to psychosis. Raballo et al7 found the severity of the CAARMS disorganization dimension was the strongest predictor of transition to frank psychosis. Other research suggests that approximately one-third of ARMS patients transition to psychosis within 3 years, another one-third have persistent attenuated psychotic symptoms, and the remaining one-third experience symptom remission.8,9
Despite multiple studies and meta-analyses, current scales and clinical predictors continue to be imperfect.8 Efforts to identify specific biological markers and predictors of transition to clinical psychosis have not been successful for ARMS.10,11 The Table8,9,12,13 summarizes diagnostic criteria that have been developed to more clearly identify which ARMS patients face the highest imminent risk for transition to psychosis; these have been referred to as ultra high-risk (UHR) criteria.14 These UHR criteria depict 3 categories of clinical presentation believed to confer risk of transition to psychosis: attenuated psychotic symptoms, transient psychotic symptoms, and genetic predisposition. Subsequent research found that certain additional symptom variables, as well as combinations of specific symptom clusters, conferred increased risk and improved the positive predictive sensitivity to as high as 83%.15 In addition to the UHR criteria, the Table8,9,12,13 also lists these additional variables shown to confer a high positive predictive value (PPV) of transition, alone or in combination with the UHR criterion. Thompson et al16 provide more detailed information on these later variables and their relative PPV.

What about treatment?
While discussion of the optimal treatment options for patients with prodromal symptoms of schizophrenia is beyond the scope of this article, early interventions can focus on preventing the biological, psychological, and social disruption that results from such symptoms. Establishing a therapeutic alliance with the patient while they retain insight and engaging supportive family members is a key starting point. Case management, cognitive-behavioral or supportive therapy, and treatment of comorbid mood, anxiety, or substance use disorders are helpful. There is no clear consensus on the utility of pharmacotherapy in the prodromal stage of psychosis. While scales and structured interviews can guide assessment, clinical judgment is the key driver of the appropriateness of initiating pharmacologic treatment to address symptoms. Because up to two-thirds of patients who satisfy UHR criteria do not go on to develop schizophrenia,16 clinicians should be thoughtful about the risks and benefits of antipsychotics.
1. George M, Maheshwari S, Chandran S, et al. Understanding the schizophrenia prodrome. Indian J Psychiatry. 2017;59(4):505-509.
2. Yung AR, McGorry PD. The prodromal phase of first-episode psychosis: past and current conceptualizations. Schizophr Bull. 1996;22(2):353-370.
3. Yung AR, Yuen HP, McGorry PD, et al. Mapping the onset of psychosis: the Comprehensive Assessment of At-Risk Mental States. Aust N Z J Psychiatry. 2005;39(11-12):964-971.
4. Miller TJ, McGlashan TH, Rosen JL, et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull. 2003;29(4):703-715.
5. Loewy RL, Pearson R, Vinogradov S, et al. Psychosis risk screening with the Prodromal Questionnaire--brief version (PQ-B). Schizophr Res. 2011;129(1):42-46.
6. Nieman DH, McGorry PD. Detection and treatment of at-risk mental state for developing a first psychosis: making up the balance. Lancet Psychiatry. 2015;2(9):825-834.
7. Raballo A, Nelson B, Thompson A, et al. The comprehensive assessment of at-risk mental states: from mapping the onset to mapping the structure. Schizophr Res. 2011;127(1-3):107-114.
8. Fusar-Poli P, Bonoldi I, Yung AR, et al. Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry. 2012;69(3):220-229.
9. Cannon TD. How schizophrenia develops: cognitive and brain mechanisms underlying onset of psychosis. Trends Cogn Sci. 2015;19(12):744-756.
10. Castle DJ. Is it appropriate to treat people at high-risk of psychosis before first onset? - no. Med J Aust. 2012;196(9):557.
11. Wood SJ, Reniers RL, Heinze K. Neuroimaging findings in the at-risk mental state: a review of recent literature. Can J Psychiatry. 2013;58(1):13-18.
12. Nelson B, Yung AR. Can clinicians predict psychosis in an ultra high risk group? Aust N Z J Psychiatry. 2010;44(7):625-630.
13. Schultze-Lutter F, Michel C, Schmidt SJ, et al. EPA guidance on the early detection of clinical high risk states of psychoses. Eur Psychiatry. 2015;30(3):405-416.
14. Yung AR, Phillips LJ, Yuen HP, et al. Risk factors for psychosis in an ultra high-risk group: psychopathology and clinical features. Schizophr Res. 2004;67(2-3):131-142.
15. Ruhrmann S, Schultze-Lutter F, Salokangas RK, et al. Prediction of psychosis in adolescents and young adults at high risk: results from the prospective European prediction of psychosis study. Arch Gen Psychiatry. 2010;67(3):241-251.
16. Thompson A, Marwaha S, Broome MR. At-risk mental state for psychosis: identification and current treatment approaches. BJPsych Advances. 2016;22(3):186-193.
Schizophrenia is characterized by psychotic symptoms that typically follow a prodromal period of premonitory signs and symptoms that appear before the manifestation of the full-blown syndrome. Signs and symptoms during the prodromal phase are subsyndromal, which implies a lower degree of intensity, duration, or frequency than observed when the patient meets the full criteria for the syndrome. Early detection of prodromal symptoms can improve prognosis, but these subtle symptoms may go unrecognized.
In schizophrenia, a patient may exhibit prodromal signs and symptoms before the appearance of pathognomonic symptoms, such as delusions, hallucinations, and disorganization. The schizophrenia prodrome can be conceptualized as a period of prepsychotic disturbances depicting an alteration in the individual’s behavior and perception. Prodromal symptoms can last from weeks to years before the psychotic illness clinically manifests.1 The prodromal symptom cluster typically becomes evident during adolescence and young adulthood.2
In the mid-1990s, investigators tried to identify a “putative prodrome” for psychosis. The term “at-risk mental state” (ARMS) for psychosis is based on retrospective reports of prodromal symptoms in first-episode psychosis. Over the next 2 decades, scales such as the Comprehensive Assessment of ARMS (CAARMS)3 and the Structured Interview for Prodromal Syndrome4 were designed to enhance the objectivity and diagnostic accuracy of the ARMS. These scales have reasonable interrater reliability.5
Researchers also have attempted to stage the severity of ARMS.6 Key symptom group predictors were studied to determine which individual symptoms or cluster of symptoms are most associated with poor outcomes and progression to psychosis. Raballo et al7 found the severity of the CAARMS disorganization dimension was the strongest predictor of transition to frank psychosis. Other research suggests that approximately one-third of ARMS patients transition to psychosis within 3 years, another one-third have persistent attenuated psychotic symptoms, and the remaining one-third experience symptom remission.8,9
Despite multiple studies and meta-analyses, current scales and clinical predictors continue to be imperfect.8 Efforts to identify specific biological markers and predictors of transition to clinical psychosis have not been successful for ARMS.10,11 The Table8,9,12,13 summarizes diagnostic criteria that have been developed to more clearly identify which ARMS patients face the highest imminent risk for transition to psychosis; these have been referred to as ultra high-risk (UHR) criteria.14 These UHR criteria depict 3 categories of clinical presentation believed to confer risk of transition to psychosis: attenuated psychotic symptoms, transient psychotic symptoms, and genetic predisposition. Subsequent research found that certain additional symptom variables, as well as combinations of specific symptom clusters, conferred increased risk and improved the positive predictive sensitivity to as high as 83%.15 In addition to the UHR criteria, the Table8,9,12,13 also lists these additional variables shown to confer a high positive predictive value (PPV) of transition, alone or in combination with the UHR criterion. Thompson et al16 provide more detailed information on these later variables and their relative PPV.

What about treatment?
While discussion of the optimal treatment options for patients with prodromal symptoms of schizophrenia is beyond the scope of this article, early interventions can focus on preventing the biological, psychological, and social disruption that results from such symptoms. Establishing a therapeutic alliance with the patient while they retain insight and engaging supportive family members is a key starting point. Case management, cognitive-behavioral or supportive therapy, and treatment of comorbid mood, anxiety, or substance use disorders are helpful. There is no clear consensus on the utility of pharmacotherapy in the prodromal stage of psychosis. While scales and structured interviews can guide assessment, clinical judgment is the key driver of the appropriateness of initiating pharmacologic treatment to address symptoms. Because up to two-thirds of patients who satisfy UHR criteria do not go on to develop schizophrenia,16 clinicians should be thoughtful about the risks and benefits of antipsychotics.
Schizophrenia is characterized by psychotic symptoms that typically follow a prodromal period of premonitory signs and symptoms that appear before the manifestation of the full-blown syndrome. Signs and symptoms during the prodromal phase are subsyndromal, which implies a lower degree of intensity, duration, or frequency than observed when the patient meets the full criteria for the syndrome. Early detection of prodromal symptoms can improve prognosis, but these subtle symptoms may go unrecognized.
In schizophrenia, a patient may exhibit prodromal signs and symptoms before the appearance of pathognomonic symptoms, such as delusions, hallucinations, and disorganization. The schizophrenia prodrome can be conceptualized as a period of prepsychotic disturbances depicting an alteration in the individual’s behavior and perception. Prodromal symptoms can last from weeks to years before the psychotic illness clinically manifests.1 The prodromal symptom cluster typically becomes evident during adolescence and young adulthood.2
In the mid-1990s, investigators tried to identify a “putative prodrome” for psychosis. The term “at-risk mental state” (ARMS) for psychosis is based on retrospective reports of prodromal symptoms in first-episode psychosis. Over the next 2 decades, scales such as the Comprehensive Assessment of ARMS (CAARMS)3 and the Structured Interview for Prodromal Syndrome4 were designed to enhance the objectivity and diagnostic accuracy of the ARMS. These scales have reasonable interrater reliability.5
Researchers also have attempted to stage the severity of ARMS.6 Key symptom group predictors were studied to determine which individual symptoms or cluster of symptoms are most associated with poor outcomes and progression to psychosis. Raballo et al7 found the severity of the CAARMS disorganization dimension was the strongest predictor of transition to frank psychosis. Other research suggests that approximately one-third of ARMS patients transition to psychosis within 3 years, another one-third have persistent attenuated psychotic symptoms, and the remaining one-third experience symptom remission.8,9
Despite multiple studies and meta-analyses, current scales and clinical predictors continue to be imperfect.8 Efforts to identify specific biological markers and predictors of transition to clinical psychosis have not been successful for ARMS.10,11 The Table8,9,12,13 summarizes diagnostic criteria that have been developed to more clearly identify which ARMS patients face the highest imminent risk for transition to psychosis; these have been referred to as ultra high-risk (UHR) criteria.14 These UHR criteria depict 3 categories of clinical presentation believed to confer risk of transition to psychosis: attenuated psychotic symptoms, transient psychotic symptoms, and genetic predisposition. Subsequent research found that certain additional symptom variables, as well as combinations of specific symptom clusters, conferred increased risk and improved the positive predictive sensitivity to as high as 83%.15 In addition to the UHR criteria, the Table8,9,12,13 also lists these additional variables shown to confer a high positive predictive value (PPV) of transition, alone or in combination with the UHR criterion. Thompson et al16 provide more detailed information on these later variables and their relative PPV.

What about treatment?
While discussion of the optimal treatment options for patients with prodromal symptoms of schizophrenia is beyond the scope of this article, early interventions can focus on preventing the biological, psychological, and social disruption that results from such symptoms. Establishing a therapeutic alliance with the patient while they retain insight and engaging supportive family members is a key starting point. Case management, cognitive-behavioral or supportive therapy, and treatment of comorbid mood, anxiety, or substance use disorders are helpful. There is no clear consensus on the utility of pharmacotherapy in the prodromal stage of psychosis. While scales and structured interviews can guide assessment, clinical judgment is the key driver of the appropriateness of initiating pharmacologic treatment to address symptoms. Because up to two-thirds of patients who satisfy UHR criteria do not go on to develop schizophrenia,16 clinicians should be thoughtful about the risks and benefits of antipsychotics.
1. George M, Maheshwari S, Chandran S, et al. Understanding the schizophrenia prodrome. Indian J Psychiatry. 2017;59(4):505-509.
2. Yung AR, McGorry PD. The prodromal phase of first-episode psychosis: past and current conceptualizations. Schizophr Bull. 1996;22(2):353-370.
3. Yung AR, Yuen HP, McGorry PD, et al. Mapping the onset of psychosis: the Comprehensive Assessment of At-Risk Mental States. Aust N Z J Psychiatry. 2005;39(11-12):964-971.
4. Miller TJ, McGlashan TH, Rosen JL, et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull. 2003;29(4):703-715.
5. Loewy RL, Pearson R, Vinogradov S, et al. Psychosis risk screening with the Prodromal Questionnaire--brief version (PQ-B). Schizophr Res. 2011;129(1):42-46.
6. Nieman DH, McGorry PD. Detection and treatment of at-risk mental state for developing a first psychosis: making up the balance. Lancet Psychiatry. 2015;2(9):825-834.
7. Raballo A, Nelson B, Thompson A, et al. The comprehensive assessment of at-risk mental states: from mapping the onset to mapping the structure. Schizophr Res. 2011;127(1-3):107-114.
8. Fusar-Poli P, Bonoldi I, Yung AR, et al. Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry. 2012;69(3):220-229.
9. Cannon TD. How schizophrenia develops: cognitive and brain mechanisms underlying onset of psychosis. Trends Cogn Sci. 2015;19(12):744-756.
10. Castle DJ. Is it appropriate to treat people at high-risk of psychosis before first onset? - no. Med J Aust. 2012;196(9):557.
11. Wood SJ, Reniers RL, Heinze K. Neuroimaging findings in the at-risk mental state: a review of recent literature. Can J Psychiatry. 2013;58(1):13-18.
12. Nelson B, Yung AR. Can clinicians predict psychosis in an ultra high risk group? Aust N Z J Psychiatry. 2010;44(7):625-630.
13. Schultze-Lutter F, Michel C, Schmidt SJ, et al. EPA guidance on the early detection of clinical high risk states of psychoses. Eur Psychiatry. 2015;30(3):405-416.
14. Yung AR, Phillips LJ, Yuen HP, et al. Risk factors for psychosis in an ultra high-risk group: psychopathology and clinical features. Schizophr Res. 2004;67(2-3):131-142.
15. Ruhrmann S, Schultze-Lutter F, Salokangas RK, et al. Prediction of psychosis in adolescents and young adults at high risk: results from the prospective European prediction of psychosis study. Arch Gen Psychiatry. 2010;67(3):241-251.
16. Thompson A, Marwaha S, Broome MR. At-risk mental state for psychosis: identification and current treatment approaches. BJPsych Advances. 2016;22(3):186-193.
1. George M, Maheshwari S, Chandran S, et al. Understanding the schizophrenia prodrome. Indian J Psychiatry. 2017;59(4):505-509.
2. Yung AR, McGorry PD. The prodromal phase of first-episode psychosis: past and current conceptualizations. Schizophr Bull. 1996;22(2):353-370.
3. Yung AR, Yuen HP, McGorry PD, et al. Mapping the onset of psychosis: the Comprehensive Assessment of At-Risk Mental States. Aust N Z J Psychiatry. 2005;39(11-12):964-971.
4. Miller TJ, McGlashan TH, Rosen JL, et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull. 2003;29(4):703-715.
5. Loewy RL, Pearson R, Vinogradov S, et al. Psychosis risk screening with the Prodromal Questionnaire--brief version (PQ-B). Schizophr Res. 2011;129(1):42-46.
6. Nieman DH, McGorry PD. Detection and treatment of at-risk mental state for developing a first psychosis: making up the balance. Lancet Psychiatry. 2015;2(9):825-834.
7. Raballo A, Nelson B, Thompson A, et al. The comprehensive assessment of at-risk mental states: from mapping the onset to mapping the structure. Schizophr Res. 2011;127(1-3):107-114.
8. Fusar-Poli P, Bonoldi I, Yung AR, et al. Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry. 2012;69(3):220-229.
9. Cannon TD. How schizophrenia develops: cognitive and brain mechanisms underlying onset of psychosis. Trends Cogn Sci. 2015;19(12):744-756.
10. Castle DJ. Is it appropriate to treat people at high-risk of psychosis before first onset? - no. Med J Aust. 2012;196(9):557.
11. Wood SJ, Reniers RL, Heinze K. Neuroimaging findings in the at-risk mental state: a review of recent literature. Can J Psychiatry. 2013;58(1):13-18.
12. Nelson B, Yung AR. Can clinicians predict psychosis in an ultra high risk group? Aust N Z J Psychiatry. 2010;44(7):625-630.
13. Schultze-Lutter F, Michel C, Schmidt SJ, et al. EPA guidance on the early detection of clinical high risk states of psychoses. Eur Psychiatry. 2015;30(3):405-416.
14. Yung AR, Phillips LJ, Yuen HP, et al. Risk factors for psychosis in an ultra high-risk group: psychopathology and clinical features. Schizophr Res. 2004;67(2-3):131-142.
15. Ruhrmann S, Schultze-Lutter F, Salokangas RK, et al. Prediction of psychosis in adolescents and young adults at high risk: results from the prospective European prediction of psychosis study. Arch Gen Psychiatry. 2010;67(3):241-251.
16. Thompson A, Marwaha S, Broome MR. At-risk mental state for psychosis: identification and current treatment approaches. BJPsych Advances. 2016;22(3):186-193.
Immunodeficiencies tied to psychiatric disorders in offspring
new research suggests.
Results from a cohort study of more than 4.2 million individuals showed that offspring of mothers with PIDs had a 17% increased risk for a psychiatric disorder and a 20% increased risk for suicidal behavior, compared with their peers with mothers who did not have PIDs.
The risk was more pronounced in offspring of mothers with both PIDs and autoimmune diseases. These risks remained after strictly controlling for different covariates, such as the parents’ psychiatric history, offspring PIDs, and offspring autoimmune diseases.
The investigators, led by Josef Isung, MD, PhD, Centre for Psychiatry Research, department of clinical neuroscience, Karolinska Institutet, Stockholm, noted that they could not “pinpoint a precise causal mechanism” underlying these findings.
Still, “the results add to the existing literature suggesting that the intrauterine immune environment may have implications for fetal neurodevelopment and that a compromised maternal immune system during pregnancy may be a risk factor for psychiatric disorders and suicidal behavior in their offspring in the long term,” they wrote.
The findings were published online in JAMA Psychiatry.
‘Natural experiment’
Maternal immune activation (MIA) is “an overarching term for aberrant and disrupted immune activity in the mother during gestation [and] has long been of interest in relation to adverse health outcomes in the offspring,” Dr. Isung noted.
“In relation to negative psychiatric outcomes, there is an abundance of preclinical evidence that has shown a negative impact on offspring secondary to MIA. And in humans, there are several observational studies supporting this link,” he said in an interview.
Dr. Isung added that PIDs are “rare conditions” known to be associated with repeated infections and high rates of autoimmune diseases, causing substantial disability.
“PIDs represent an interesting ‘natural experiment’ for researchers to understand more about the association between immune system dysfunctions and mental health,” he said.
Dr. Isung’s group previously showed that individuals with PIDs have increased odds of psychiatric disorders and suicidal behavior. The link was more pronounced in women with PIDs – and was even more pronounced in those with both PIDs and autoimmune diseases.
In the current study, “we wanted to see whether offspring of individuals were differentially at risk of psychiatric disorders and suicidal behavior, depending on being offspring of mothers or fathers with PIDs,” Dr. Isung said.
“Our hypothesis was that mothers with PIDs would have an increased risk of having offspring with neuropsychiatric outcomes, and that this risk could be due to MIA,” he added.
The researchers turned to Swedish nationwide health and administrative registers. They analyzed data on all individuals with diagnoses of PIDs identified between 1973 and 2013. Offspring born prior to 2003 were included, and parent-offspring pairs in which both parents had a history of PIDs were excluded.
The final study sample consisted of 4,294,169 offspring (51.4% boys). Of these participants, 7,270 (0.17%) had a parent with PIDs.
The researchers identified lifetime records of 10 psychiatric disorders: obsessive-compulsive disorder, ADHD, autism spectrum disorders, schizophrenia and other psychotic disorders, bipolar disorders, major depressive disorder and other mood disorders, anxiety and stress-related disorders, eating disorders, substance use disorders, and Tourette syndrome and chronic tic disorders.
The investigators included parental birth year, psychopathology, suicide attempts, suicide deaths, and autoimmune diseases as covariates, as well as offsprings’ birth year and gender.
Elucidation needed
Results showed that, of the 4,676 offspring of mothers with PID, 17.1% had a psychiatric disorder versus 12.7% of offspring of mothers without PIDs. This translated “into a 17% increased risk for offspring of mothers with PIDs in the fully adjusted model,” the investigators reported.
The risk was even higher for offspring of mothers who had not only PIDs but also one of six of the individual psychiatric disorders, with incident rate ratios ranging from 1.15 to 1.71.
“In fully adjusted models, offspring of mothers with PIDs had an increased risk of any psychiatric disorder, while no such risks were observed in offspring of fathers with PIDs” (IRR, 1.17 vs. 1.03; P < .001), the researchers reported.
A higher risk for suicidal behavior was also observed among offspring of mothers with PIDS, in contrast to those of fathers with PIDs (IRR, 1.2 vs. 1.1; P = .01).
The greatest risk for any psychiatric disorder, as well as suicidal behavior, was found in offspring of mothers who had both PIDs and autoimmune diseases (IRRs, 1.24 and 1.44, respectively).
“The results could be seen as substantiating the hypothesis that immune disruption may be important in the pathophysiology of psychiatric disorders and suicidal behavior,” Dr. Isung said.
“Furthermore, the fact that only offspring of mothers and not offspring of fathers with PIDs had this association would align with our hypothesis that MIA is of importance,” he added.
However, he noted that “the specific mechanisms are most likely multifactorial and remain to be elucidated.”
Important piece of the puzzle?
In a comment, Michael Eriksen Benros, MD, PhD, professor of immunopsychiatry, department of immunology and microbiology, health, and medical sciences, University of Copenhagen, said this was a “high-quality study” that used a “rich data source.”
Dr. Benros, who is also head of research (biological and precision psychiatry) at the Copenhagen Research Centre for Mental Health, Copenhagen University Hospital, was not involved with the current study.
He noted that prior studies, including some conducted by his own group, have shown that maternal infections overall did not seem to be “specifically linked to mental disorders in the offspring.”
However, “specific maternal infections or specific brain-reactive antibodies during the pregnancy period have been shown to be associated with neurodevelopmental outcomes among the children,” such as intellectual disability, he said.
Regarding direct clinical implications of the study, “it is important to note that the increased risk of psychiatric disorders and suicidality in the offspring of mothers with PID were small,” Dr. Benros said.
“However, it adds an important part to the scientific puzzle regarding the role of maternal immune activation during pregnancy and the risk of mental disorders,” he added.
The study was funded by the Söderström König Foundation and the Fredrik and Ingrid Thuring Foundation. Neither Dr. Isung nor Dr. Benros reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests.
Results from a cohort study of more than 4.2 million individuals showed that offspring of mothers with PIDs had a 17% increased risk for a psychiatric disorder and a 20% increased risk for suicidal behavior, compared with their peers with mothers who did not have PIDs.
The risk was more pronounced in offspring of mothers with both PIDs and autoimmune diseases. These risks remained after strictly controlling for different covariates, such as the parents’ psychiatric history, offspring PIDs, and offspring autoimmune diseases.
The investigators, led by Josef Isung, MD, PhD, Centre for Psychiatry Research, department of clinical neuroscience, Karolinska Institutet, Stockholm, noted that they could not “pinpoint a precise causal mechanism” underlying these findings.
Still, “the results add to the existing literature suggesting that the intrauterine immune environment may have implications for fetal neurodevelopment and that a compromised maternal immune system during pregnancy may be a risk factor for psychiatric disorders and suicidal behavior in their offspring in the long term,” they wrote.
The findings were published online in JAMA Psychiatry.
‘Natural experiment’
Maternal immune activation (MIA) is “an overarching term for aberrant and disrupted immune activity in the mother during gestation [and] has long been of interest in relation to adverse health outcomes in the offspring,” Dr. Isung noted.
“In relation to negative psychiatric outcomes, there is an abundance of preclinical evidence that has shown a negative impact on offspring secondary to MIA. And in humans, there are several observational studies supporting this link,” he said in an interview.
Dr. Isung added that PIDs are “rare conditions” known to be associated with repeated infections and high rates of autoimmune diseases, causing substantial disability.
“PIDs represent an interesting ‘natural experiment’ for researchers to understand more about the association between immune system dysfunctions and mental health,” he said.
Dr. Isung’s group previously showed that individuals with PIDs have increased odds of psychiatric disorders and suicidal behavior. The link was more pronounced in women with PIDs – and was even more pronounced in those with both PIDs and autoimmune diseases.
In the current study, “we wanted to see whether offspring of individuals were differentially at risk of psychiatric disorders and suicidal behavior, depending on being offspring of mothers or fathers with PIDs,” Dr. Isung said.
“Our hypothesis was that mothers with PIDs would have an increased risk of having offspring with neuropsychiatric outcomes, and that this risk could be due to MIA,” he added.
The researchers turned to Swedish nationwide health and administrative registers. They analyzed data on all individuals with diagnoses of PIDs identified between 1973 and 2013. Offspring born prior to 2003 were included, and parent-offspring pairs in which both parents had a history of PIDs were excluded.
The final study sample consisted of 4,294,169 offspring (51.4% boys). Of these participants, 7,270 (0.17%) had a parent with PIDs.
The researchers identified lifetime records of 10 psychiatric disorders: obsessive-compulsive disorder, ADHD, autism spectrum disorders, schizophrenia and other psychotic disorders, bipolar disorders, major depressive disorder and other mood disorders, anxiety and stress-related disorders, eating disorders, substance use disorders, and Tourette syndrome and chronic tic disorders.
The investigators included parental birth year, psychopathology, suicide attempts, suicide deaths, and autoimmune diseases as covariates, as well as offsprings’ birth year and gender.
Elucidation needed
Results showed that, of the 4,676 offspring of mothers with PID, 17.1% had a psychiatric disorder versus 12.7% of offspring of mothers without PIDs. This translated “into a 17% increased risk for offspring of mothers with PIDs in the fully adjusted model,” the investigators reported.
The risk was even higher for offspring of mothers who had not only PIDs but also one of six of the individual psychiatric disorders, with incident rate ratios ranging from 1.15 to 1.71.
“In fully adjusted models, offspring of mothers with PIDs had an increased risk of any psychiatric disorder, while no such risks were observed in offspring of fathers with PIDs” (IRR, 1.17 vs. 1.03; P < .001), the researchers reported.
A higher risk for suicidal behavior was also observed among offspring of mothers with PIDS, in contrast to those of fathers with PIDs (IRR, 1.2 vs. 1.1; P = .01).
The greatest risk for any psychiatric disorder, as well as suicidal behavior, was found in offspring of mothers who had both PIDs and autoimmune diseases (IRRs, 1.24 and 1.44, respectively).
“The results could be seen as substantiating the hypothesis that immune disruption may be important in the pathophysiology of psychiatric disorders and suicidal behavior,” Dr. Isung said.
“Furthermore, the fact that only offspring of mothers and not offspring of fathers with PIDs had this association would align with our hypothesis that MIA is of importance,” he added.
However, he noted that “the specific mechanisms are most likely multifactorial and remain to be elucidated.”
Important piece of the puzzle?
In a comment, Michael Eriksen Benros, MD, PhD, professor of immunopsychiatry, department of immunology and microbiology, health, and medical sciences, University of Copenhagen, said this was a “high-quality study” that used a “rich data source.”
Dr. Benros, who is also head of research (biological and precision psychiatry) at the Copenhagen Research Centre for Mental Health, Copenhagen University Hospital, was not involved with the current study.
He noted that prior studies, including some conducted by his own group, have shown that maternal infections overall did not seem to be “specifically linked to mental disorders in the offspring.”
However, “specific maternal infections or specific brain-reactive antibodies during the pregnancy period have been shown to be associated with neurodevelopmental outcomes among the children,” such as intellectual disability, he said.
Regarding direct clinical implications of the study, “it is important to note that the increased risk of psychiatric disorders and suicidality in the offspring of mothers with PID were small,” Dr. Benros said.
“However, it adds an important part to the scientific puzzle regarding the role of maternal immune activation during pregnancy and the risk of mental disorders,” he added.
The study was funded by the Söderström König Foundation and the Fredrik and Ingrid Thuring Foundation. Neither Dr. Isung nor Dr. Benros reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
new research suggests.
Results from a cohort study of more than 4.2 million individuals showed that offspring of mothers with PIDs had a 17% increased risk for a psychiatric disorder and a 20% increased risk for suicidal behavior, compared with their peers with mothers who did not have PIDs.
The risk was more pronounced in offspring of mothers with both PIDs and autoimmune diseases. These risks remained after strictly controlling for different covariates, such as the parents’ psychiatric history, offspring PIDs, and offspring autoimmune diseases.
The investigators, led by Josef Isung, MD, PhD, Centre for Psychiatry Research, department of clinical neuroscience, Karolinska Institutet, Stockholm, noted that they could not “pinpoint a precise causal mechanism” underlying these findings.
Still, “the results add to the existing literature suggesting that the intrauterine immune environment may have implications for fetal neurodevelopment and that a compromised maternal immune system during pregnancy may be a risk factor for psychiatric disorders and suicidal behavior in their offspring in the long term,” they wrote.
The findings were published online in JAMA Psychiatry.
‘Natural experiment’
Maternal immune activation (MIA) is “an overarching term for aberrant and disrupted immune activity in the mother during gestation [and] has long been of interest in relation to adverse health outcomes in the offspring,” Dr. Isung noted.
“In relation to negative psychiatric outcomes, there is an abundance of preclinical evidence that has shown a negative impact on offspring secondary to MIA. And in humans, there are several observational studies supporting this link,” he said in an interview.
Dr. Isung added that PIDs are “rare conditions” known to be associated with repeated infections and high rates of autoimmune diseases, causing substantial disability.
“PIDs represent an interesting ‘natural experiment’ for researchers to understand more about the association between immune system dysfunctions and mental health,” he said.
Dr. Isung’s group previously showed that individuals with PIDs have increased odds of psychiatric disorders and suicidal behavior. The link was more pronounced in women with PIDs – and was even more pronounced in those with both PIDs and autoimmune diseases.
In the current study, “we wanted to see whether offspring of individuals were differentially at risk of psychiatric disorders and suicidal behavior, depending on being offspring of mothers or fathers with PIDs,” Dr. Isung said.
“Our hypothesis was that mothers with PIDs would have an increased risk of having offspring with neuropsychiatric outcomes, and that this risk could be due to MIA,” he added.
The researchers turned to Swedish nationwide health and administrative registers. They analyzed data on all individuals with diagnoses of PIDs identified between 1973 and 2013. Offspring born prior to 2003 were included, and parent-offspring pairs in which both parents had a history of PIDs were excluded.
The final study sample consisted of 4,294,169 offspring (51.4% boys). Of these participants, 7,270 (0.17%) had a parent with PIDs.
The researchers identified lifetime records of 10 psychiatric disorders: obsessive-compulsive disorder, ADHD, autism spectrum disorders, schizophrenia and other psychotic disorders, bipolar disorders, major depressive disorder and other mood disorders, anxiety and stress-related disorders, eating disorders, substance use disorders, and Tourette syndrome and chronic tic disorders.
The investigators included parental birth year, psychopathology, suicide attempts, suicide deaths, and autoimmune diseases as covariates, as well as offsprings’ birth year and gender.
Elucidation needed
Results showed that, of the 4,676 offspring of mothers with PID, 17.1% had a psychiatric disorder versus 12.7% of offspring of mothers without PIDs. This translated “into a 17% increased risk for offspring of mothers with PIDs in the fully adjusted model,” the investigators reported.
The risk was even higher for offspring of mothers who had not only PIDs but also one of six of the individual psychiatric disorders, with incident rate ratios ranging from 1.15 to 1.71.
“In fully adjusted models, offspring of mothers with PIDs had an increased risk of any psychiatric disorder, while no such risks were observed in offspring of fathers with PIDs” (IRR, 1.17 vs. 1.03; P < .001), the researchers reported.
A higher risk for suicidal behavior was also observed among offspring of mothers with PIDS, in contrast to those of fathers with PIDs (IRR, 1.2 vs. 1.1; P = .01).
The greatest risk for any psychiatric disorder, as well as suicidal behavior, was found in offspring of mothers who had both PIDs and autoimmune diseases (IRRs, 1.24 and 1.44, respectively).
“The results could be seen as substantiating the hypothesis that immune disruption may be important in the pathophysiology of psychiatric disorders and suicidal behavior,” Dr. Isung said.
“Furthermore, the fact that only offspring of mothers and not offspring of fathers with PIDs had this association would align with our hypothesis that MIA is of importance,” he added.
However, he noted that “the specific mechanisms are most likely multifactorial and remain to be elucidated.”
Important piece of the puzzle?
In a comment, Michael Eriksen Benros, MD, PhD, professor of immunopsychiatry, department of immunology and microbiology, health, and medical sciences, University of Copenhagen, said this was a “high-quality study” that used a “rich data source.”
Dr. Benros, who is also head of research (biological and precision psychiatry) at the Copenhagen Research Centre for Mental Health, Copenhagen University Hospital, was not involved with the current study.
He noted that prior studies, including some conducted by his own group, have shown that maternal infections overall did not seem to be “specifically linked to mental disorders in the offspring.”
However, “specific maternal infections or specific brain-reactive antibodies during the pregnancy period have been shown to be associated with neurodevelopmental outcomes among the children,” such as intellectual disability, he said.
Regarding direct clinical implications of the study, “it is important to note that the increased risk of psychiatric disorders and suicidality in the offspring of mothers with PID were small,” Dr. Benros said.
“However, it adds an important part to the scientific puzzle regarding the role of maternal immune activation during pregnancy and the risk of mental disorders,” he added.
The study was funded by the Söderström König Foundation and the Fredrik and Ingrid Thuring Foundation. Neither Dr. Isung nor Dr. Benros reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM JAMA PSYCHIATRY
No benefit of long-acting antipsychotics in schizophrenia?
In a multicountry, randomized, open-label study of more than 500 adults with schizophrenia, participants received either LAI paliperidone, LAI aripiprazole, or the respective oral formulation of these antipsychotics.
Results showed no significant difference between the combined oral and combined LAI treatment groups in time to all-cause discontinuation.
“We found no substantial advantage for LAI antipsychotic treatment over oral treatment, regarding time to discontinuation in patients with early-phase schizophrenia,” write investigators, led by Inge Winter-van Rossum, PhD, assistant visiting professor at Mount Sinai, New York, and affiliated with King’s College London and UMC Utrecht (the Netherlands).
This indicates that “there is no reason to prescribe LAIs instead of oral antipsychotics if the goal is to prevent discontinuation of antipsychotic medication in daily clinical practice,” they add.
The findings were published online in The Lancet Psychiatry.
Previous conflicting results
Maintenance treatment with antipsychotic medication reduces risk for relapse considerably, with treatment discontinuation being “by far the most important reason for relapse,” the investigators write.
LAIs “seem theoretically to be a way to enhance medication continuation and thereby reduce the risk for relapse,” they add. This is because LAIs enable a rapid response to nonadherence and remove the need for patients to remember to take their medications on a daily basis.
However, previous research has “provided conflicting results,” regarding the effectiveness of LAIs in accomplishing this. Moreover, the subject has not been thoroughly investigated in early-stage schizophrenia, the researchers note.
Therefore, they decided to conduct the EULAST study to compare LAI and oral formulations in terms of all-cause discontinuation.
The trial was conducted at 50 general hospitals and psychiatric specialty clinics located in 15 European countries and Israel and included 511 participants in the intention-to-treat sample (67% men; mean age, 30.5 years).
All were randomly assigned 1:1:1 to receive either LAI paliperidone, LAI aripiprazole, or their respective oral formulations.
The combined OA treatment group consisted of 247 patients; the combined LAI group consisted of 264 patients.
Randomization was stratified by country and illness duration (5 months to 3 years vs. 4-7 years). Participants were followed up to 19 months, with all-cause discontinuation during that time serving as the primary endpoint.
All-cause discontinuation was defined as the allocated treatment was stopped or used at doses outside the allowed range, medication was switched or augmented with another antipsychotic after visit four, the patient missed a monthly visit and did not show up after being reminded, the patient withdrew consent for the study, or the clinician withdrew the patient from the study.
After the baseline visit, patients already taking antipsychotics were also randomly assigned. The next 4 weeks were then used to cross-taper between the prestudy antipsychotic and the agent they would be treated with during the study.
LAIs not superior
Results showed the LAI group did not have lower rates of hospitalization.
In addition, the discontinuation rates between the two combined groups were very similar at 71% for the oral antipsychotics group versus 64% in the LAIs group (hazard ratio, 1.6; 95% confidence interval, 0.94-1.43; P = .18).
Moreover, “no significant difference was found in the time to all-cause discontinuation between the combined oral and combined LAI treatment groups (P = .17),” the researchers report.
Reasons for discontinuation also did not differ significantly between the groups: 12% of patients in the OA group discontinued treatment because of efficacy vs. 17% of patients in the combined LAI group. The difference was not significant and the time to discontinuation also did not differ.
The main reason for discontinuation in both groups was safety concerns, affecting 10% and 13% of the combined OA and LAI groups, respectively, which was not a significant between-group difference.
Illness duration had a significant effect on time to all-cause discontinuation, with patients who had longer illness duration showing a poorer response, compared with those who had shorter duration (HR, 1.26; 95% CI, 1.01-1.56; P = .038).
However, stratifying participants by illness duration showed no significant difference between the subgroups (P = .25 and .34, respectively).
There was a significant between-group difference in discontinuation due to “other reasons,” with 49% vs. 34% of patients in the OA and LAI groups, respectively, discontinuing (HR, 1.51; 95% CI, 1.15-1.98; P = .0034). Moreover, the LAI group showed significantly longer continued use of medication vs the OA group (P = .0029).
“After separating the reasons for discontinuation into no efficacy, safety reasons, and other reasons, we only found a significant difference in favor of LAI for the ‘other reasons’ category; although the number of patients discontinuing medication for this reason over the follow-up period did not differ, patients on LAI continued treatment for a longer time,” the investigators write.
They acknowledge that this finding is “difficult to interpret, given the wide variety of reasons for discontinuation captured in this category,” which prevented an “informative subgroup analysis.”
Nevertheless, since there is “no consistent evidence supporting the use of LAI over oral antipsychotics” in patients with early-phase schizophrenia, their use should be “carefully considered on an individual risk-benefit basis,” they conclude.
No ‘real-world’ implications?
John M. Kane, MD, codirector and professor, Institute of Behavioral Science, Feinstein Institutes for Medical Research, Manhasset, N.Y., said that overall, this was a “large, potentially valuable study.” However, he raised several concerns.
“I think the investigators made a much too emphatic statement about the lack of value of LAIs in early-phase patients when discontinuation is the primary outcome,” he said, noting that other studies have come to the opposite conclusion.
Dr. Kane, who is also a professor of psychiatry at Hofstra/Northwell, New York, was not involved with the current research.
“RCTs [randomized controlled trials] in general are not necessarily the best way to evaluate the impact of LAIs [which] usually represent a small percentage of potentially eligible patients and are likely to include patients who are more adherent than those who would not agree to participate in an RCT,” he said. He added that the investigators “did not report on how many patients were screened and refused to be considered.”
Also, Dr. Kane noted that half of the participants were recruited from inpatient services, and so may have been “more unstable” at baseline. “Patients with residual positive symptoms are more likely to relapse on LAIs than patients who are in remission. This could potentially reduce the advantage of the LAI,” he said.
In addition, he took issue with the definition of all-cause discontinuation, which included the need for augmentation with another antipsychotic or use outside the normal range.
“This happens often in clinical practice. If someone’s symptoms aren’t sufficiently controlled by an LAI alone, for example, they often receive more of that antipsychotic or another drug. This perhaps makes the EULAST study somewhat less ‘real-world’,” Dr. Kane said.
More information needed
In an accompanying editorial, Martina Hahn, PharmD, PhD, department of psychiatry, psychosomatics, and psychotherapy, University Hospital-Goethe University, Frankfurt, Germany, and Sibylle Christine Roll, MD, PHD, department of mental health, Varisano Hospital in Frankfurt, note that comedications were neither documented nor analyzed by the researchers.
“Drug-drug interactions could be responsible for relapse or poor tolerability,” they write.
Moreover, pharmacogenetic information was not available nor were serum concentrations that could have been used for dose optimization after switching antipsychotic formulations, they note.
This information would have provided “a deeper understanding of why some patients do not respond or show side effects,” the editorialists write. “The use of therapeutic drug monitoring, drug interaction checks, and pharmacogenetic testing could improve treatment outcomes in both study settings and clinical practice.”
Financial support and study medication was provided by Lundbeck and Otsuka. Dr. Winter-van Rossum reports no relevant financial relationships. Disclosures for the other investigators are fully listed in the original paper. Dr. Kane is or has been a consultant to or received honoraria for lectures from Alkermes , Biogen, Boehringer Ingelheim, Cerevel, Dainippon Sumitomo, H. Lundbeck, HLS, Intracellular Therapies, Janssen, Karuna, Merck, Newron, Otsuka, Roche, Saladax, Sunovion, and TEVA. He is also a shareholder in The Vanguard Research Group, LB Pharma, Health Rhythms, North Shore Therapeutics, and Medincell. Dr. Hahn reports having received honoraria for lecture from Otsuka and advisory board participation for Rovi. Dr. Roll reports advisory board participation for Recordati, Otsuka, and Janssen.
A version of this article first appeared on Medscape.com.
In a multicountry, randomized, open-label study of more than 500 adults with schizophrenia, participants received either LAI paliperidone, LAI aripiprazole, or the respective oral formulation of these antipsychotics.
Results showed no significant difference between the combined oral and combined LAI treatment groups in time to all-cause discontinuation.
“We found no substantial advantage for LAI antipsychotic treatment over oral treatment, regarding time to discontinuation in patients with early-phase schizophrenia,” write investigators, led by Inge Winter-van Rossum, PhD, assistant visiting professor at Mount Sinai, New York, and affiliated with King’s College London and UMC Utrecht (the Netherlands).
This indicates that “there is no reason to prescribe LAIs instead of oral antipsychotics if the goal is to prevent discontinuation of antipsychotic medication in daily clinical practice,” they add.
The findings were published online in The Lancet Psychiatry.
Previous conflicting results
Maintenance treatment with antipsychotic medication reduces risk for relapse considerably, with treatment discontinuation being “by far the most important reason for relapse,” the investigators write.
LAIs “seem theoretically to be a way to enhance medication continuation and thereby reduce the risk for relapse,” they add. This is because LAIs enable a rapid response to nonadherence and remove the need for patients to remember to take their medications on a daily basis.
However, previous research has “provided conflicting results,” regarding the effectiveness of LAIs in accomplishing this. Moreover, the subject has not been thoroughly investigated in early-stage schizophrenia, the researchers note.
Therefore, they decided to conduct the EULAST study to compare LAI and oral formulations in terms of all-cause discontinuation.
The trial was conducted at 50 general hospitals and psychiatric specialty clinics located in 15 European countries and Israel and included 511 participants in the intention-to-treat sample (67% men; mean age, 30.5 years).
All were randomly assigned 1:1:1 to receive either LAI paliperidone, LAI aripiprazole, or their respective oral formulations.
The combined OA treatment group consisted of 247 patients; the combined LAI group consisted of 264 patients.
Randomization was stratified by country and illness duration (5 months to 3 years vs. 4-7 years). Participants were followed up to 19 months, with all-cause discontinuation during that time serving as the primary endpoint.
All-cause discontinuation was defined as the allocated treatment was stopped or used at doses outside the allowed range, medication was switched or augmented with another antipsychotic after visit four, the patient missed a monthly visit and did not show up after being reminded, the patient withdrew consent for the study, or the clinician withdrew the patient from the study.
After the baseline visit, patients already taking antipsychotics were also randomly assigned. The next 4 weeks were then used to cross-taper between the prestudy antipsychotic and the agent they would be treated with during the study.
LAIs not superior
Results showed the LAI group did not have lower rates of hospitalization.
In addition, the discontinuation rates between the two combined groups were very similar at 71% for the oral antipsychotics group versus 64% in the LAIs group (hazard ratio, 1.6; 95% confidence interval, 0.94-1.43; P = .18).
Moreover, “no significant difference was found in the time to all-cause discontinuation between the combined oral and combined LAI treatment groups (P = .17),” the researchers report.
Reasons for discontinuation also did not differ significantly between the groups: 12% of patients in the OA group discontinued treatment because of efficacy vs. 17% of patients in the combined LAI group. The difference was not significant and the time to discontinuation also did not differ.
The main reason for discontinuation in both groups was safety concerns, affecting 10% and 13% of the combined OA and LAI groups, respectively, which was not a significant between-group difference.
Illness duration had a significant effect on time to all-cause discontinuation, with patients who had longer illness duration showing a poorer response, compared with those who had shorter duration (HR, 1.26; 95% CI, 1.01-1.56; P = .038).
However, stratifying participants by illness duration showed no significant difference between the subgroups (P = .25 and .34, respectively).
There was a significant between-group difference in discontinuation due to “other reasons,” with 49% vs. 34% of patients in the OA and LAI groups, respectively, discontinuing (HR, 1.51; 95% CI, 1.15-1.98; P = .0034). Moreover, the LAI group showed significantly longer continued use of medication vs the OA group (P = .0029).
“After separating the reasons for discontinuation into no efficacy, safety reasons, and other reasons, we only found a significant difference in favor of LAI for the ‘other reasons’ category; although the number of patients discontinuing medication for this reason over the follow-up period did not differ, patients on LAI continued treatment for a longer time,” the investigators write.
They acknowledge that this finding is “difficult to interpret, given the wide variety of reasons for discontinuation captured in this category,” which prevented an “informative subgroup analysis.”
Nevertheless, since there is “no consistent evidence supporting the use of LAI over oral antipsychotics” in patients with early-phase schizophrenia, their use should be “carefully considered on an individual risk-benefit basis,” they conclude.
No ‘real-world’ implications?
John M. Kane, MD, codirector and professor, Institute of Behavioral Science, Feinstein Institutes for Medical Research, Manhasset, N.Y., said that overall, this was a “large, potentially valuable study.” However, he raised several concerns.
“I think the investigators made a much too emphatic statement about the lack of value of LAIs in early-phase patients when discontinuation is the primary outcome,” he said, noting that other studies have come to the opposite conclusion.
Dr. Kane, who is also a professor of psychiatry at Hofstra/Northwell, New York, was not involved with the current research.
“RCTs [randomized controlled trials] in general are not necessarily the best way to evaluate the impact of LAIs [which] usually represent a small percentage of potentially eligible patients and are likely to include patients who are more adherent than those who would not agree to participate in an RCT,” he said. He added that the investigators “did not report on how many patients were screened and refused to be considered.”
Also, Dr. Kane noted that half of the participants were recruited from inpatient services, and so may have been “more unstable” at baseline. “Patients with residual positive symptoms are more likely to relapse on LAIs than patients who are in remission. This could potentially reduce the advantage of the LAI,” he said.
In addition, he took issue with the definition of all-cause discontinuation, which included the need for augmentation with another antipsychotic or use outside the normal range.
“This happens often in clinical practice. If someone’s symptoms aren’t sufficiently controlled by an LAI alone, for example, they often receive more of that antipsychotic or another drug. This perhaps makes the EULAST study somewhat less ‘real-world’,” Dr. Kane said.
More information needed
In an accompanying editorial, Martina Hahn, PharmD, PhD, department of psychiatry, psychosomatics, and psychotherapy, University Hospital-Goethe University, Frankfurt, Germany, and Sibylle Christine Roll, MD, PHD, department of mental health, Varisano Hospital in Frankfurt, note that comedications were neither documented nor analyzed by the researchers.
“Drug-drug interactions could be responsible for relapse or poor tolerability,” they write.
Moreover, pharmacogenetic information was not available nor were serum concentrations that could have been used for dose optimization after switching antipsychotic formulations, they note.
This information would have provided “a deeper understanding of why some patients do not respond or show side effects,” the editorialists write. “The use of therapeutic drug monitoring, drug interaction checks, and pharmacogenetic testing could improve treatment outcomes in both study settings and clinical practice.”
Financial support and study medication was provided by Lundbeck and Otsuka. Dr. Winter-van Rossum reports no relevant financial relationships. Disclosures for the other investigators are fully listed in the original paper. Dr. Kane is or has been a consultant to or received honoraria for lectures from Alkermes , Biogen, Boehringer Ingelheim, Cerevel, Dainippon Sumitomo, H. Lundbeck, HLS, Intracellular Therapies, Janssen, Karuna, Merck, Newron, Otsuka, Roche, Saladax, Sunovion, and TEVA. He is also a shareholder in The Vanguard Research Group, LB Pharma, Health Rhythms, North Shore Therapeutics, and Medincell. Dr. Hahn reports having received honoraria for lecture from Otsuka and advisory board participation for Rovi. Dr. Roll reports advisory board participation for Recordati, Otsuka, and Janssen.
A version of this article first appeared on Medscape.com.
In a multicountry, randomized, open-label study of more than 500 adults with schizophrenia, participants received either LAI paliperidone, LAI aripiprazole, or the respective oral formulation of these antipsychotics.
Results showed no significant difference between the combined oral and combined LAI treatment groups in time to all-cause discontinuation.
“We found no substantial advantage for LAI antipsychotic treatment over oral treatment, regarding time to discontinuation in patients with early-phase schizophrenia,” write investigators, led by Inge Winter-van Rossum, PhD, assistant visiting professor at Mount Sinai, New York, and affiliated with King’s College London and UMC Utrecht (the Netherlands).
This indicates that “there is no reason to prescribe LAIs instead of oral antipsychotics if the goal is to prevent discontinuation of antipsychotic medication in daily clinical practice,” they add.
The findings were published online in The Lancet Psychiatry.
Previous conflicting results
Maintenance treatment with antipsychotic medication reduces risk for relapse considerably, with treatment discontinuation being “by far the most important reason for relapse,” the investigators write.
LAIs “seem theoretically to be a way to enhance medication continuation and thereby reduce the risk for relapse,” they add. This is because LAIs enable a rapid response to nonadherence and remove the need for patients to remember to take their medications on a daily basis.
However, previous research has “provided conflicting results,” regarding the effectiveness of LAIs in accomplishing this. Moreover, the subject has not been thoroughly investigated in early-stage schizophrenia, the researchers note.
Therefore, they decided to conduct the EULAST study to compare LAI and oral formulations in terms of all-cause discontinuation.
The trial was conducted at 50 general hospitals and psychiatric specialty clinics located in 15 European countries and Israel and included 511 participants in the intention-to-treat sample (67% men; mean age, 30.5 years).
All were randomly assigned 1:1:1 to receive either LAI paliperidone, LAI aripiprazole, or their respective oral formulations.
The combined OA treatment group consisted of 247 patients; the combined LAI group consisted of 264 patients.
Randomization was stratified by country and illness duration (5 months to 3 years vs. 4-7 years). Participants were followed up to 19 months, with all-cause discontinuation during that time serving as the primary endpoint.
All-cause discontinuation was defined as the allocated treatment was stopped or used at doses outside the allowed range, medication was switched or augmented with another antipsychotic after visit four, the patient missed a monthly visit and did not show up after being reminded, the patient withdrew consent for the study, or the clinician withdrew the patient from the study.
After the baseline visit, patients already taking antipsychotics were also randomly assigned. The next 4 weeks were then used to cross-taper between the prestudy antipsychotic and the agent they would be treated with during the study.
LAIs not superior
Results showed the LAI group did not have lower rates of hospitalization.
In addition, the discontinuation rates between the two combined groups were very similar at 71% for the oral antipsychotics group versus 64% in the LAIs group (hazard ratio, 1.6; 95% confidence interval, 0.94-1.43; P = .18).
Moreover, “no significant difference was found in the time to all-cause discontinuation between the combined oral and combined LAI treatment groups (P = .17),” the researchers report.
Reasons for discontinuation also did not differ significantly between the groups: 12% of patients in the OA group discontinued treatment because of efficacy vs. 17% of patients in the combined LAI group. The difference was not significant and the time to discontinuation also did not differ.
The main reason for discontinuation in both groups was safety concerns, affecting 10% and 13% of the combined OA and LAI groups, respectively, which was not a significant between-group difference.
Illness duration had a significant effect on time to all-cause discontinuation, with patients who had longer illness duration showing a poorer response, compared with those who had shorter duration (HR, 1.26; 95% CI, 1.01-1.56; P = .038).
However, stratifying participants by illness duration showed no significant difference between the subgroups (P = .25 and .34, respectively).
There was a significant between-group difference in discontinuation due to “other reasons,” with 49% vs. 34% of patients in the OA and LAI groups, respectively, discontinuing (HR, 1.51; 95% CI, 1.15-1.98; P = .0034). Moreover, the LAI group showed significantly longer continued use of medication vs the OA group (P = .0029).
“After separating the reasons for discontinuation into no efficacy, safety reasons, and other reasons, we only found a significant difference in favor of LAI for the ‘other reasons’ category; although the number of patients discontinuing medication for this reason over the follow-up period did not differ, patients on LAI continued treatment for a longer time,” the investigators write.
They acknowledge that this finding is “difficult to interpret, given the wide variety of reasons for discontinuation captured in this category,” which prevented an “informative subgroup analysis.”
Nevertheless, since there is “no consistent evidence supporting the use of LAI over oral antipsychotics” in patients with early-phase schizophrenia, their use should be “carefully considered on an individual risk-benefit basis,” they conclude.
No ‘real-world’ implications?
John M. Kane, MD, codirector and professor, Institute of Behavioral Science, Feinstein Institutes for Medical Research, Manhasset, N.Y., said that overall, this was a “large, potentially valuable study.” However, he raised several concerns.
“I think the investigators made a much too emphatic statement about the lack of value of LAIs in early-phase patients when discontinuation is the primary outcome,” he said, noting that other studies have come to the opposite conclusion.
Dr. Kane, who is also a professor of psychiatry at Hofstra/Northwell, New York, was not involved with the current research.
“RCTs [randomized controlled trials] in general are not necessarily the best way to evaluate the impact of LAIs [which] usually represent a small percentage of potentially eligible patients and are likely to include patients who are more adherent than those who would not agree to participate in an RCT,” he said. He added that the investigators “did not report on how many patients were screened and refused to be considered.”
Also, Dr. Kane noted that half of the participants were recruited from inpatient services, and so may have been “more unstable” at baseline. “Patients with residual positive symptoms are more likely to relapse on LAIs than patients who are in remission. This could potentially reduce the advantage of the LAI,” he said.
In addition, he took issue with the definition of all-cause discontinuation, which included the need for augmentation with another antipsychotic or use outside the normal range.
“This happens often in clinical practice. If someone’s symptoms aren’t sufficiently controlled by an LAI alone, for example, they often receive more of that antipsychotic or another drug. This perhaps makes the EULAST study somewhat less ‘real-world’,” Dr. Kane said.
More information needed
In an accompanying editorial, Martina Hahn, PharmD, PhD, department of psychiatry, psychosomatics, and psychotherapy, University Hospital-Goethe University, Frankfurt, Germany, and Sibylle Christine Roll, MD, PHD, department of mental health, Varisano Hospital in Frankfurt, note that comedications were neither documented nor analyzed by the researchers.
“Drug-drug interactions could be responsible for relapse or poor tolerability,” they write.
Moreover, pharmacogenetic information was not available nor were serum concentrations that could have been used for dose optimization after switching antipsychotic formulations, they note.
This information would have provided “a deeper understanding of why some patients do not respond or show side effects,” the editorialists write. “The use of therapeutic drug monitoring, drug interaction checks, and pharmacogenetic testing could improve treatment outcomes in both study settings and clinical practice.”
Financial support and study medication was provided by Lundbeck and Otsuka. Dr. Winter-van Rossum reports no relevant financial relationships. Disclosures for the other investigators are fully listed in the original paper. Dr. Kane is or has been a consultant to or received honoraria for lectures from Alkermes , Biogen, Boehringer Ingelheim, Cerevel, Dainippon Sumitomo, H. Lundbeck, HLS, Intracellular Therapies, Janssen, Karuna, Merck, Newron, Otsuka, Roche, Saladax, Sunovion, and TEVA. He is also a shareholder in The Vanguard Research Group, LB Pharma, Health Rhythms, North Shore Therapeutics, and Medincell. Dr. Hahn reports having received honoraria for lecture from Otsuka and advisory board participation for Rovi. Dr. Roll reports advisory board participation for Recordati, Otsuka, and Janssen.
A version of this article first appeared on Medscape.com.
FROM THE LANCET PSYCHIATRY
Forced hospitalization for mental illness not a permanent solution
I met Eleanor when I was writing a book on involuntary psychiatric treatment. She was very ill when she presented to an emergency department in Northern California. She was looking for help and would have signed herself in, but after waiting 8 hours with no food or medical attention, she walked out and went to another hospital.
At this point, she was agitated and distressed and began screaming uncontrollably. The physician in the second ED did not offer her the option of signing in, and she was placed on a 72-hour hold and subsequently held in the hospital for 3 weeks after a judge committed her.
Like so many issues, involuntary psychiatric care is highly polarized. Some groups favor legislation to make involuntary treatment easier, while patient advocacy and civil rights groups vehemently oppose such legislation.
We don’t hear from these combatants as much as we hear from those who trumpet their views on abortion or gun control, yet this battlefield exists. It is not surprising that when New York City Mayor Eric Adams announced a plan to hospitalize homeless people with mental illnesses – involuntarily if necessary, and at the discretion of the police – people were outraged.
New York City is not the only place using this strategy to address the problem of mental illness and homelessness; California has enacted similar legislation, and every major city has homeless citizens.
Eleanor was not homeless, and fortunately, she recovered and returned to her family. However, she remained distressed and traumatized by her hospitalization for years. “It sticks with you,” she told me. “I would rather die than go in again.”
I wish I could tell you that Eleanor is unique in saying that she would rather die than go to a hospital unit for treatment, but it is not an uncommon sentiment for patients. Some people who are charged with crimes and end up in the judicial system will opt to go to jail rather than to a psychiatric hospital. It is also not easy to access outpatient psychiatric treatment.
Barriers to care
Many psychiatrists don’t participate with insurance networks, and publicly funded clinics may have long waiting lists, so illnesses escalate until there is a crisis and hospitalization is necessary. For many, stigma and fear of potential professional repercussions are significant barriers to care.
What are the issues that legislation attempts to address? The first is the standard for hospitalizing individuals against their will. In some states, the patient must be dangerous, while in others there is a lower standard of “gravely disabled,” and finally there are those that promote a standard of a “need for treatment.”
The second is related to medicating people against their will, a process that can be rightly perceived as an assault if the patient refuses to take oral medications and must be held down for injections. Next, the use of outpatient civil commitment – legally requiring people to get treatment if they are not in the hospital – has been increasingly invoked as a way to prevent mass murders and random violence against strangers.
All but four states have some legislation for outpatient commitment, euphemistically called Assisted Outpatient Treatment (AOT), yet these laws are difficult to enforce and expensive to enact. They are also not fully effective.
In New York City, Kendra’s Law has not eliminated subway violence by people with psychiatric disturbances, and the shooter who killed 32 people and wounded 17 others at Virginia Tech in 2007 had previously been ordered by a judge to go to outpatient treatment, but he simply never showed up for his appointment.
Finally, the battle includes the right of patients to refuse to have their psychiatric information released to their caretakers under the Health Insurance Portability and Accountability Act of 1996 – a measure that many families believe would help them to get loved ones to take medications and go to appointments.
The concern about how to negotiate the needs of society and the civil rights of people with psychiatric disorders has been with us for centuries. There is a strong antipsychiatry movement that asserts that psychotropic medications are ineffective or harmful and refers to patients as “psychiatric survivors.” We value the right to medical autonomy, and when there is controversy over the validity of a treatment, there is even more controversy over forcing it upon people.
Psychiatric medications are very effective and benefit many people, but they don’t help everyone, and some people experience side effects. Also, we can’t deny that involuntary care can go wrong; the conservatorship of Britney Spears for 13 years is a very public example.
Multiple stakeholders
Many have a stake in how this plays out. There are the patients, who may be suffering and unable to recognize that they are ill, who may have valid reasons for not wanting the treatments, and who ideally should have the right to refuse care.
There are the families who watch their loved ones suffer, deteriorate, and miss the opportunities that life has to offer; who do not want their children to be homeless or incarcerated; and who may be at risk from violent behavior.
There are the mental health professionals who want to do what’s in the best interest of their patients while following legal and ethical mandates, who worry about being sued for tragic outcomes, and who can’t meet the current demand for services.
There is the taxpayer who foots the bill for disability payments, lost productivity, and institutionalization. There is our society that worries that people with psychiatric disorders will commit random acts of violence.
Finally, there are the insurers, who want to pay for as little care as possible and throw up constant hurdles in the treatment process. We must acknowledge that resources used for involuntary treatment are diverted away from those who want care.
Eleanor had many advantages that unhoused people don’t have: a supportive family, health insurance, and the financial means to pay a psychiatrist who respected her wishes to wean off her medications. She returned to a comfortable home and to personal and occupational success.
It is tragic that we have people living on the streets because of a psychiatric disorder, addiction, poverty, or some combination of these. No one should be unhoused. If the rationale of hospitalization is to decrease violence, I am not hopeful. The Epidemiologic Catchment Area study shows that people with psychiatric disorders are responsible for only 4% of all violence.
The logistics of determining which people living on the streets have psychiatric disorders, transporting them safely to medical facilities, and then finding the resources to provide for compassionate and thoughtful care in meaningful and sustained ways are very challenging.
If we don’t want people living on the streets, we need to create supports, including infrastructure to facilitate housing, access to mental health care, and addiction treatment before we resort to involuntary hospitalization.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
I met Eleanor when I was writing a book on involuntary psychiatric treatment. She was very ill when she presented to an emergency department in Northern California. She was looking for help and would have signed herself in, but after waiting 8 hours with no food or medical attention, she walked out and went to another hospital.
At this point, she was agitated and distressed and began screaming uncontrollably. The physician in the second ED did not offer her the option of signing in, and she was placed on a 72-hour hold and subsequently held in the hospital for 3 weeks after a judge committed her.
Like so many issues, involuntary psychiatric care is highly polarized. Some groups favor legislation to make involuntary treatment easier, while patient advocacy and civil rights groups vehemently oppose such legislation.
We don’t hear from these combatants as much as we hear from those who trumpet their views on abortion or gun control, yet this battlefield exists. It is not surprising that when New York City Mayor Eric Adams announced a plan to hospitalize homeless people with mental illnesses – involuntarily if necessary, and at the discretion of the police – people were outraged.
New York City is not the only place using this strategy to address the problem of mental illness and homelessness; California has enacted similar legislation, and every major city has homeless citizens.
Eleanor was not homeless, and fortunately, she recovered and returned to her family. However, she remained distressed and traumatized by her hospitalization for years. “It sticks with you,” she told me. “I would rather die than go in again.”
I wish I could tell you that Eleanor is unique in saying that she would rather die than go to a hospital unit for treatment, but it is not an uncommon sentiment for patients. Some people who are charged with crimes and end up in the judicial system will opt to go to jail rather than to a psychiatric hospital. It is also not easy to access outpatient psychiatric treatment.
Barriers to care
Many psychiatrists don’t participate with insurance networks, and publicly funded clinics may have long waiting lists, so illnesses escalate until there is a crisis and hospitalization is necessary. For many, stigma and fear of potential professional repercussions are significant barriers to care.
What are the issues that legislation attempts to address? The first is the standard for hospitalizing individuals against their will. In some states, the patient must be dangerous, while in others there is a lower standard of “gravely disabled,” and finally there are those that promote a standard of a “need for treatment.”
The second is related to medicating people against their will, a process that can be rightly perceived as an assault if the patient refuses to take oral medications and must be held down for injections. Next, the use of outpatient civil commitment – legally requiring people to get treatment if they are not in the hospital – has been increasingly invoked as a way to prevent mass murders and random violence against strangers.
All but four states have some legislation for outpatient commitment, euphemistically called Assisted Outpatient Treatment (AOT), yet these laws are difficult to enforce and expensive to enact. They are also not fully effective.
In New York City, Kendra’s Law has not eliminated subway violence by people with psychiatric disturbances, and the shooter who killed 32 people and wounded 17 others at Virginia Tech in 2007 had previously been ordered by a judge to go to outpatient treatment, but he simply never showed up for his appointment.
Finally, the battle includes the right of patients to refuse to have their psychiatric information released to their caretakers under the Health Insurance Portability and Accountability Act of 1996 – a measure that many families believe would help them to get loved ones to take medications and go to appointments.
The concern about how to negotiate the needs of society and the civil rights of people with psychiatric disorders has been with us for centuries. There is a strong antipsychiatry movement that asserts that psychotropic medications are ineffective or harmful and refers to patients as “psychiatric survivors.” We value the right to medical autonomy, and when there is controversy over the validity of a treatment, there is even more controversy over forcing it upon people.
Psychiatric medications are very effective and benefit many people, but they don’t help everyone, and some people experience side effects. Also, we can’t deny that involuntary care can go wrong; the conservatorship of Britney Spears for 13 years is a very public example.
Multiple stakeholders
Many have a stake in how this plays out. There are the patients, who may be suffering and unable to recognize that they are ill, who may have valid reasons for not wanting the treatments, and who ideally should have the right to refuse care.
There are the families who watch their loved ones suffer, deteriorate, and miss the opportunities that life has to offer; who do not want their children to be homeless or incarcerated; and who may be at risk from violent behavior.
There are the mental health professionals who want to do what’s in the best interest of their patients while following legal and ethical mandates, who worry about being sued for tragic outcomes, and who can’t meet the current demand for services.
There is the taxpayer who foots the bill for disability payments, lost productivity, and institutionalization. There is our society that worries that people with psychiatric disorders will commit random acts of violence.
Finally, there are the insurers, who want to pay for as little care as possible and throw up constant hurdles in the treatment process. We must acknowledge that resources used for involuntary treatment are diverted away from those who want care.
Eleanor had many advantages that unhoused people don’t have: a supportive family, health insurance, and the financial means to pay a psychiatrist who respected her wishes to wean off her medications. She returned to a comfortable home and to personal and occupational success.
It is tragic that we have people living on the streets because of a psychiatric disorder, addiction, poverty, or some combination of these. No one should be unhoused. If the rationale of hospitalization is to decrease violence, I am not hopeful. The Epidemiologic Catchment Area study shows that people with psychiatric disorders are responsible for only 4% of all violence.
The logistics of determining which people living on the streets have psychiatric disorders, transporting them safely to medical facilities, and then finding the resources to provide for compassionate and thoughtful care in meaningful and sustained ways are very challenging.
If we don’t want people living on the streets, we need to create supports, including infrastructure to facilitate housing, access to mental health care, and addiction treatment before we resort to involuntary hospitalization.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
I met Eleanor when I was writing a book on involuntary psychiatric treatment. She was very ill when she presented to an emergency department in Northern California. She was looking for help and would have signed herself in, but after waiting 8 hours with no food or medical attention, she walked out and went to another hospital.
At this point, she was agitated and distressed and began screaming uncontrollably. The physician in the second ED did not offer her the option of signing in, and she was placed on a 72-hour hold and subsequently held in the hospital for 3 weeks after a judge committed her.
Like so many issues, involuntary psychiatric care is highly polarized. Some groups favor legislation to make involuntary treatment easier, while patient advocacy and civil rights groups vehemently oppose such legislation.
We don’t hear from these combatants as much as we hear from those who trumpet their views on abortion or gun control, yet this battlefield exists. It is not surprising that when New York City Mayor Eric Adams announced a plan to hospitalize homeless people with mental illnesses – involuntarily if necessary, and at the discretion of the police – people were outraged.
New York City is not the only place using this strategy to address the problem of mental illness and homelessness; California has enacted similar legislation, and every major city has homeless citizens.
Eleanor was not homeless, and fortunately, she recovered and returned to her family. However, she remained distressed and traumatized by her hospitalization for years. “It sticks with you,” she told me. “I would rather die than go in again.”
I wish I could tell you that Eleanor is unique in saying that she would rather die than go to a hospital unit for treatment, but it is not an uncommon sentiment for patients. Some people who are charged with crimes and end up in the judicial system will opt to go to jail rather than to a psychiatric hospital. It is also not easy to access outpatient psychiatric treatment.
Barriers to care
Many psychiatrists don’t participate with insurance networks, and publicly funded clinics may have long waiting lists, so illnesses escalate until there is a crisis and hospitalization is necessary. For many, stigma and fear of potential professional repercussions are significant barriers to care.
What are the issues that legislation attempts to address? The first is the standard for hospitalizing individuals against their will. In some states, the patient must be dangerous, while in others there is a lower standard of “gravely disabled,” and finally there are those that promote a standard of a “need for treatment.”
The second is related to medicating people against their will, a process that can be rightly perceived as an assault if the patient refuses to take oral medications and must be held down for injections. Next, the use of outpatient civil commitment – legally requiring people to get treatment if they are not in the hospital – has been increasingly invoked as a way to prevent mass murders and random violence against strangers.
All but four states have some legislation for outpatient commitment, euphemistically called Assisted Outpatient Treatment (AOT), yet these laws are difficult to enforce and expensive to enact. They are also not fully effective.
In New York City, Kendra’s Law has not eliminated subway violence by people with psychiatric disturbances, and the shooter who killed 32 people and wounded 17 others at Virginia Tech in 2007 had previously been ordered by a judge to go to outpatient treatment, but he simply never showed up for his appointment.
Finally, the battle includes the right of patients to refuse to have their psychiatric information released to their caretakers under the Health Insurance Portability and Accountability Act of 1996 – a measure that many families believe would help them to get loved ones to take medications and go to appointments.
The concern about how to negotiate the needs of society and the civil rights of people with psychiatric disorders has been with us for centuries. There is a strong antipsychiatry movement that asserts that psychotropic medications are ineffective or harmful and refers to patients as “psychiatric survivors.” We value the right to medical autonomy, and when there is controversy over the validity of a treatment, there is even more controversy over forcing it upon people.
Psychiatric medications are very effective and benefit many people, but they don’t help everyone, and some people experience side effects. Also, we can’t deny that involuntary care can go wrong; the conservatorship of Britney Spears for 13 years is a very public example.
Multiple stakeholders
Many have a stake in how this plays out. There are the patients, who may be suffering and unable to recognize that they are ill, who may have valid reasons for not wanting the treatments, and who ideally should have the right to refuse care.
There are the families who watch their loved ones suffer, deteriorate, and miss the opportunities that life has to offer; who do not want their children to be homeless or incarcerated; and who may be at risk from violent behavior.
There are the mental health professionals who want to do what’s in the best interest of their patients while following legal and ethical mandates, who worry about being sued for tragic outcomes, and who can’t meet the current demand for services.
There is the taxpayer who foots the bill for disability payments, lost productivity, and institutionalization. There is our society that worries that people with psychiatric disorders will commit random acts of violence.
Finally, there are the insurers, who want to pay for as little care as possible and throw up constant hurdles in the treatment process. We must acknowledge that resources used for involuntary treatment are diverted away from those who want care.
Eleanor had many advantages that unhoused people don’t have: a supportive family, health insurance, and the financial means to pay a psychiatrist who respected her wishes to wean off her medications. She returned to a comfortable home and to personal and occupational success.
It is tragic that we have people living on the streets because of a psychiatric disorder, addiction, poverty, or some combination of these. No one should be unhoused. If the rationale of hospitalization is to decrease violence, I am not hopeful. The Epidemiologic Catchment Area study shows that people with psychiatric disorders are responsible for only 4% of all violence.
The logistics of determining which people living on the streets have psychiatric disorders, transporting them safely to medical facilities, and then finding the resources to provide for compassionate and thoughtful care in meaningful and sustained ways are very challenging.
If we don’t want people living on the streets, we need to create supports, including infrastructure to facilitate housing, access to mental health care, and addiction treatment before we resort to involuntary hospitalization.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore. She has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Finding catatonia requires knowing what to look for
Catatonia is a psychomotor syndrome identified by its clinical phenotype. Unlike common psychiatric syndromes such as major depression that are characterized by self-report of symptoms, catatonia is identified chiefly by empirically evaluated signs on clinical evaluation. Its signs are recognized through observation, physical examination, or elicitation by clinical maneuvers or the presentation of stimuli. However, catatonia is often overlooked even though its clinical signs are often visibly apparent, including to the casual observer.
Why is catatonia underdiagnosed? A key modifiable factor appears to be a prevalent misunderstanding over what catatonia looks like.1 We have sought to address this in a few ways.
First identified was the need for comprehensive educational resources on how to assess for and recognize catatonia. Using the Bush-Francis Catatonia Rating Scale – the most widely used scale for catatonia in both research and clinical settings and the most cited publication in the catatonia literature – our team developed the BFCRS Training Manual and Coding Guide.2,3 This manual expands on the definitions of each BFCRS item based on how it was originally operationalized by the scale’s authors. Subsequently, we created a comprehensive set of educational resources including videos illustrating how to assess for catatonia, a video for each of the 23 items on the BFCRS, and self-assessment tools. All resources are freely available online at https://bfcrs.urmc.edu.4
Through this project it became apparent that there are many discrepancies across the field regarding the phenotype of catatonia. Specifically, a recent review inspired by this project set about to characterize the scope of distinctions across diagnostic systems and rating scales.5 For instance, each diagnostic system and rating scale includes a unique set of signs, approaches diagnostic thresholds differently, and often operationalizes clinical features in ways that lead either to criterion overlap (for example, combativeness would be scored both as combativeness and agitation on ICD-11) or contradictions with other systems or scales (for example, varied definitions of waxy flexibility). In the face of so many inconsistencies, what is a clinician to do? What follows is a discussion of how to apply the insights from this recent review in clinical and research settings.
Starting with DSM-5-TR and ICD-11 – the current editions of the two leading diagnostic systems – one might ask: How do they compare?6,7 Overall, these two systems are broadly aligned in terms of the catatonic syndrome. Both systems identify individual clinical signs (as opposed to symptom complexes). Both require three features as a diagnostic threshold. Most of the same clinical signs are included in both systems, and the definitions of individual items are largely equivalent. Additionally, both systems allow for diagnosis of catatonia in association with psychiatric and medical conditions and include a category for unspecified catatonia.
Despite these core agreements, though, there are several important distinctions. First, whereas all 12 signs included in DSM-5-TR count toward an ICD-11 catatonia diagnosis, the opposite cannot be said. ICD-11 includes several features that are not in DSM-5-TR: rigidity, verbigeration, withdrawal, staring, ambitendency, impulsivity, and combativeness. Next, autonomic abnormality, which signifies the most severe type of catatonia called malignant catatonia, is included as a potential comorbidity in ICD-11 but not mentioned in DSM-5-TR. Third, ICD-11 includes a separate diagnosis for substance-induced catatonia, whereas this condition would be diagnosed as unspecified catatonia in DSM-5-TR.
There are also elements missing from both systems. The most notable of these is that neither system specifies the period over which findings must be present for diagnosis. By clinical convention, the practical definition of 24 hours is appropriate in most instances. The clinical features identified during direct evaluation are usually sufficient for diagnosis, but additional signs observed or documented over the prior 24 hours should be incorporated as part of the clinical evaluation. Another distinction is how to handle clinical features before and after lorazepam challenge. As noted in the BFCRS Training Manual, it would be appropriate to compare “state assessments” (that is, restricted to features identified only during direct, in-person assessment) from before and after lorazepam administration to document improvement.4
Whereas DSM-5-TR and ICD-11 are broadly in agreement, comparing these systems with catatonia rating scales reveals many sources of potential confusion, but also concrete guidance on operationalizing individual items.5 How exactly should each of catatonia’s clinical signs be defined? Descriptions differ, and thresholds of duration and frequency vary considerably across scales. As a result, clinicians who use different scales and then convert these results to diagnostic criteria are liable to come to different clinical conclusions. For instance, both echophenomena and negativism must be elicited more than five times to be scored per Northoff,8 but even a single convincing instance of either would be scored on the BFCRS as “occasional.”2
Such discrepancies are important because, whereas the psychometric properties of several catatonia scales have been documented, there are no analogous studies on the DSM-5-TR and ICD-11 criteria. Therefore, it is essential for clinicians and researchers to document how diagnostic criteria have been operationalized. The most practical and evidence-based way to do this is to use a clinically validated scale and convert these to diagnostic criteria, yet in doing so a few modifications will be necessary.
Of the available clinical scales, the BFCRS is best positioned for clinical use. The BFCRS has been validated clinically and has good reliability, detailed item definitions and audiovisual examples available. In addition, it is the only scale with a published semistructured evaluation (see initial paper and Training Manual), which takes about 5 minutes.2,4 In terms of utility, all 12 signs included by DSM-5-TR are among the first 14 items on the BFCRS, which constitutes a standalone tool known as the Bush-Francis Catatonia Screening Instrument (BFCSI, see Table).
Many fundamental questions remain about catatonia,but the importance of a shared understanding of its clinical features is clear.9 Catatonia should be on the differential whenever a patient exhibits a markedly altered level of activity or grossly abnormal behavior, especially when inappropriate to context. We encourage readers to familiarize themselves with the phenotype of catatonia through online educational resources4 because the optimal care of patients with catatonia requires – at a minimum – that we know what we’re looking for.
Dr. Oldham is assistant professor of psychiatry at the University of Rochester (N.Y.) Medical Center. Dr. Francis is professor of psychiatry at Penn State University, Hershey. The authors declare no relevant conflicts of interest. Funding for the educational project hosted at https://bfcrs.urmc.edu was provided by the department of psychiatry at the University of Rochester Medical Center. Dr. Oldham is currently supported by a K23 career development award from the National Institute on Aging (AG072383). The educational resources referenced in this piece could not have been created were it not for the intellectual and thespian collaboration of Joshua R. Wortzel, MD, who is currently a fellow in child and adolescent psychiatry at Brown University, Providence, R.I. The authors are also indebted to Hochang B. Lee, MD, for his gracious support of this project.
References
1. Wortzel JR et al. J Clin Psychiatry. 2021 Aug 17;82(5):21m14025. doi: 10.4088/JCP.21m14025.
2. Bush G et al. Acta Psychiatr Scand. 1996 Feb;93(2):129-36. doi: 10.1111/j.1600-0447.1996.tb09814.x.
3. Weleff J et al. J Acad Consult Liaison Psychiatry. 2023 Jan-Feb;64(1):13-27. doi:10.1016/j.jaclp.2022.07.002.
4. Oldham MA et al. Bush-Francis Catatonia Rating Scale Assessment Resources. University of Rochester Medical Center, Department of Psychiatry. https://bfcrs.urmc.edu.
5. Oldham MA. Schizophr Res. 2022 Aug 19;S0920-9964(22)00294-8. doi: 10.1016/j.schres.2022.08.002.
6. American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5-TR. Washington, D.C.: American Psychiatric Association Publishing, 2022.
7. World Health Organization. ICD-11 for Mortality and Morbidity Stastistics. 2022. https://icd.who.int/browse11/l-m/en#/http://id.who.int/icd/entity/486722075.
8. Northoff G et al. Mov Disord. May 1999;14(3):404-16. doi: 10.1002/1531-8257(199905)14:3<404::AID-MDS1004>3.0.CO;2-5.
9. Walther S et al. The Lancet Psychiatry. 2019 Jul;6(7):610-9. doi: 10.1016/S2215-0366(18)30474-7.
Catatonia is a psychomotor syndrome identified by its clinical phenotype. Unlike common psychiatric syndromes such as major depression that are characterized by self-report of symptoms, catatonia is identified chiefly by empirically evaluated signs on clinical evaluation. Its signs are recognized through observation, physical examination, or elicitation by clinical maneuvers or the presentation of stimuli. However, catatonia is often overlooked even though its clinical signs are often visibly apparent, including to the casual observer.
Why is catatonia underdiagnosed? A key modifiable factor appears to be a prevalent misunderstanding over what catatonia looks like.1 We have sought to address this in a few ways.
First identified was the need for comprehensive educational resources on how to assess for and recognize catatonia. Using the Bush-Francis Catatonia Rating Scale – the most widely used scale for catatonia in both research and clinical settings and the most cited publication in the catatonia literature – our team developed the BFCRS Training Manual and Coding Guide.2,3 This manual expands on the definitions of each BFCRS item based on how it was originally operationalized by the scale’s authors. Subsequently, we created a comprehensive set of educational resources including videos illustrating how to assess for catatonia, a video for each of the 23 items on the BFCRS, and self-assessment tools. All resources are freely available online at https://bfcrs.urmc.edu.4
Through this project it became apparent that there are many discrepancies across the field regarding the phenotype of catatonia. Specifically, a recent review inspired by this project set about to characterize the scope of distinctions across diagnostic systems and rating scales.5 For instance, each diagnostic system and rating scale includes a unique set of signs, approaches diagnostic thresholds differently, and often operationalizes clinical features in ways that lead either to criterion overlap (for example, combativeness would be scored both as combativeness and agitation on ICD-11) or contradictions with other systems or scales (for example, varied definitions of waxy flexibility). In the face of so many inconsistencies, what is a clinician to do? What follows is a discussion of how to apply the insights from this recent review in clinical and research settings.
Starting with DSM-5-TR and ICD-11 – the current editions of the two leading diagnostic systems – one might ask: How do they compare?6,7 Overall, these two systems are broadly aligned in terms of the catatonic syndrome. Both systems identify individual clinical signs (as opposed to symptom complexes). Both require three features as a diagnostic threshold. Most of the same clinical signs are included in both systems, and the definitions of individual items are largely equivalent. Additionally, both systems allow for diagnosis of catatonia in association with psychiatric and medical conditions and include a category for unspecified catatonia.
Despite these core agreements, though, there are several important distinctions. First, whereas all 12 signs included in DSM-5-TR count toward an ICD-11 catatonia diagnosis, the opposite cannot be said. ICD-11 includes several features that are not in DSM-5-TR: rigidity, verbigeration, withdrawal, staring, ambitendency, impulsivity, and combativeness. Next, autonomic abnormality, which signifies the most severe type of catatonia called malignant catatonia, is included as a potential comorbidity in ICD-11 but not mentioned in DSM-5-TR. Third, ICD-11 includes a separate diagnosis for substance-induced catatonia, whereas this condition would be diagnosed as unspecified catatonia in DSM-5-TR.
There are also elements missing from both systems. The most notable of these is that neither system specifies the period over which findings must be present for diagnosis. By clinical convention, the practical definition of 24 hours is appropriate in most instances. The clinical features identified during direct evaluation are usually sufficient for diagnosis, but additional signs observed or documented over the prior 24 hours should be incorporated as part of the clinical evaluation. Another distinction is how to handle clinical features before and after lorazepam challenge. As noted in the BFCRS Training Manual, it would be appropriate to compare “state assessments” (that is, restricted to features identified only during direct, in-person assessment) from before and after lorazepam administration to document improvement.4
Whereas DSM-5-TR and ICD-11 are broadly in agreement, comparing these systems with catatonia rating scales reveals many sources of potential confusion, but also concrete guidance on operationalizing individual items.5 How exactly should each of catatonia’s clinical signs be defined? Descriptions differ, and thresholds of duration and frequency vary considerably across scales. As a result, clinicians who use different scales and then convert these results to diagnostic criteria are liable to come to different clinical conclusions. For instance, both echophenomena and negativism must be elicited more than five times to be scored per Northoff,8 but even a single convincing instance of either would be scored on the BFCRS as “occasional.”2
Such discrepancies are important because, whereas the psychometric properties of several catatonia scales have been documented, there are no analogous studies on the DSM-5-TR and ICD-11 criteria. Therefore, it is essential for clinicians and researchers to document how diagnostic criteria have been operationalized. The most practical and evidence-based way to do this is to use a clinically validated scale and convert these to diagnostic criteria, yet in doing so a few modifications will be necessary.
Of the available clinical scales, the BFCRS is best positioned for clinical use. The BFCRS has been validated clinically and has good reliability, detailed item definitions and audiovisual examples available. In addition, it is the only scale with a published semistructured evaluation (see initial paper and Training Manual), which takes about 5 minutes.2,4 In terms of utility, all 12 signs included by DSM-5-TR are among the first 14 items on the BFCRS, which constitutes a standalone tool known as the Bush-Francis Catatonia Screening Instrument (BFCSI, see Table).
Many fundamental questions remain about catatonia,but the importance of a shared understanding of its clinical features is clear.9 Catatonia should be on the differential whenever a patient exhibits a markedly altered level of activity or grossly abnormal behavior, especially when inappropriate to context. We encourage readers to familiarize themselves with the phenotype of catatonia through online educational resources4 because the optimal care of patients with catatonia requires – at a minimum – that we know what we’re looking for.
Dr. Oldham is assistant professor of psychiatry at the University of Rochester (N.Y.) Medical Center. Dr. Francis is professor of psychiatry at Penn State University, Hershey. The authors declare no relevant conflicts of interest. Funding for the educational project hosted at https://bfcrs.urmc.edu was provided by the department of psychiatry at the University of Rochester Medical Center. Dr. Oldham is currently supported by a K23 career development award from the National Institute on Aging (AG072383). The educational resources referenced in this piece could not have been created were it not for the intellectual and thespian collaboration of Joshua R. Wortzel, MD, who is currently a fellow in child and adolescent psychiatry at Brown University, Providence, R.I. The authors are also indebted to Hochang B. Lee, MD, for his gracious support of this project.
References
1. Wortzel JR et al. J Clin Psychiatry. 2021 Aug 17;82(5):21m14025. doi: 10.4088/JCP.21m14025.
2. Bush G et al. Acta Psychiatr Scand. 1996 Feb;93(2):129-36. doi: 10.1111/j.1600-0447.1996.tb09814.x.
3. Weleff J et al. J Acad Consult Liaison Psychiatry. 2023 Jan-Feb;64(1):13-27. doi:10.1016/j.jaclp.2022.07.002.
4. Oldham MA et al. Bush-Francis Catatonia Rating Scale Assessment Resources. University of Rochester Medical Center, Department of Psychiatry. https://bfcrs.urmc.edu.
5. Oldham MA. Schizophr Res. 2022 Aug 19;S0920-9964(22)00294-8. doi: 10.1016/j.schres.2022.08.002.
6. American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5-TR. Washington, D.C.: American Psychiatric Association Publishing, 2022.
7. World Health Organization. ICD-11 for Mortality and Morbidity Stastistics. 2022. https://icd.who.int/browse11/l-m/en#/http://id.who.int/icd/entity/486722075.
8. Northoff G et al. Mov Disord. May 1999;14(3):404-16. doi: 10.1002/1531-8257(199905)14:3<404::AID-MDS1004>3.0.CO;2-5.
9. Walther S et al. The Lancet Psychiatry. 2019 Jul;6(7):610-9. doi: 10.1016/S2215-0366(18)30474-7.
Catatonia is a psychomotor syndrome identified by its clinical phenotype. Unlike common psychiatric syndromes such as major depression that are characterized by self-report of symptoms, catatonia is identified chiefly by empirically evaluated signs on clinical evaluation. Its signs are recognized through observation, physical examination, or elicitation by clinical maneuvers or the presentation of stimuli. However, catatonia is often overlooked even though its clinical signs are often visibly apparent, including to the casual observer.
Why is catatonia underdiagnosed? A key modifiable factor appears to be a prevalent misunderstanding over what catatonia looks like.1 We have sought to address this in a few ways.
First identified was the need for comprehensive educational resources on how to assess for and recognize catatonia. Using the Bush-Francis Catatonia Rating Scale – the most widely used scale for catatonia in both research and clinical settings and the most cited publication in the catatonia literature – our team developed the BFCRS Training Manual and Coding Guide.2,3 This manual expands on the definitions of each BFCRS item based on how it was originally operationalized by the scale’s authors. Subsequently, we created a comprehensive set of educational resources including videos illustrating how to assess for catatonia, a video for each of the 23 items on the BFCRS, and self-assessment tools. All resources are freely available online at https://bfcrs.urmc.edu.4
Through this project it became apparent that there are many discrepancies across the field regarding the phenotype of catatonia. Specifically, a recent review inspired by this project set about to characterize the scope of distinctions across diagnostic systems and rating scales.5 For instance, each diagnostic system and rating scale includes a unique set of signs, approaches diagnostic thresholds differently, and often operationalizes clinical features in ways that lead either to criterion overlap (for example, combativeness would be scored both as combativeness and agitation on ICD-11) or contradictions with other systems or scales (for example, varied definitions of waxy flexibility). In the face of so many inconsistencies, what is a clinician to do? What follows is a discussion of how to apply the insights from this recent review in clinical and research settings.
Starting with DSM-5-TR and ICD-11 – the current editions of the two leading diagnostic systems – one might ask: How do they compare?6,7 Overall, these two systems are broadly aligned in terms of the catatonic syndrome. Both systems identify individual clinical signs (as opposed to symptom complexes). Both require three features as a diagnostic threshold. Most of the same clinical signs are included in both systems, and the definitions of individual items are largely equivalent. Additionally, both systems allow for diagnosis of catatonia in association with psychiatric and medical conditions and include a category for unspecified catatonia.
Despite these core agreements, though, there are several important distinctions. First, whereas all 12 signs included in DSM-5-TR count toward an ICD-11 catatonia diagnosis, the opposite cannot be said. ICD-11 includes several features that are not in DSM-5-TR: rigidity, verbigeration, withdrawal, staring, ambitendency, impulsivity, and combativeness. Next, autonomic abnormality, which signifies the most severe type of catatonia called malignant catatonia, is included as a potential comorbidity in ICD-11 but not mentioned in DSM-5-TR. Third, ICD-11 includes a separate diagnosis for substance-induced catatonia, whereas this condition would be diagnosed as unspecified catatonia in DSM-5-TR.
There are also elements missing from both systems. The most notable of these is that neither system specifies the period over which findings must be present for diagnosis. By clinical convention, the practical definition of 24 hours is appropriate in most instances. The clinical features identified during direct evaluation are usually sufficient for diagnosis, but additional signs observed or documented over the prior 24 hours should be incorporated as part of the clinical evaluation. Another distinction is how to handle clinical features before and after lorazepam challenge. As noted in the BFCRS Training Manual, it would be appropriate to compare “state assessments” (that is, restricted to features identified only during direct, in-person assessment) from before and after lorazepam administration to document improvement.4
Whereas DSM-5-TR and ICD-11 are broadly in agreement, comparing these systems with catatonia rating scales reveals many sources of potential confusion, but also concrete guidance on operationalizing individual items.5 How exactly should each of catatonia’s clinical signs be defined? Descriptions differ, and thresholds of duration and frequency vary considerably across scales. As a result, clinicians who use different scales and then convert these results to diagnostic criteria are liable to come to different clinical conclusions. For instance, both echophenomena and negativism must be elicited more than five times to be scored per Northoff,8 but even a single convincing instance of either would be scored on the BFCRS as “occasional.”2
Such discrepancies are important because, whereas the psychometric properties of several catatonia scales have been documented, there are no analogous studies on the DSM-5-TR and ICD-11 criteria. Therefore, it is essential for clinicians and researchers to document how diagnostic criteria have been operationalized. The most practical and evidence-based way to do this is to use a clinically validated scale and convert these to diagnostic criteria, yet in doing so a few modifications will be necessary.
Of the available clinical scales, the BFCRS is best positioned for clinical use. The BFCRS has been validated clinically and has good reliability, detailed item definitions and audiovisual examples available. In addition, it is the only scale with a published semistructured evaluation (see initial paper and Training Manual), which takes about 5 minutes.2,4 In terms of utility, all 12 signs included by DSM-5-TR are among the first 14 items on the BFCRS, which constitutes a standalone tool known as the Bush-Francis Catatonia Screening Instrument (BFCSI, see Table).
Many fundamental questions remain about catatonia,but the importance of a shared understanding of its clinical features is clear.9 Catatonia should be on the differential whenever a patient exhibits a markedly altered level of activity or grossly abnormal behavior, especially when inappropriate to context. We encourage readers to familiarize themselves with the phenotype of catatonia through online educational resources4 because the optimal care of patients with catatonia requires – at a minimum – that we know what we’re looking for.
Dr. Oldham is assistant professor of psychiatry at the University of Rochester (N.Y.) Medical Center. Dr. Francis is professor of psychiatry at Penn State University, Hershey. The authors declare no relevant conflicts of interest. Funding for the educational project hosted at https://bfcrs.urmc.edu was provided by the department of psychiatry at the University of Rochester Medical Center. Dr. Oldham is currently supported by a K23 career development award from the National Institute on Aging (AG072383). The educational resources referenced in this piece could not have been created were it not for the intellectual and thespian collaboration of Joshua R. Wortzel, MD, who is currently a fellow in child and adolescent psychiatry at Brown University, Providence, R.I. The authors are also indebted to Hochang B. Lee, MD, for his gracious support of this project.
References
1. Wortzel JR et al. J Clin Psychiatry. 2021 Aug 17;82(5):21m14025. doi: 10.4088/JCP.21m14025.
2. Bush G et al. Acta Psychiatr Scand. 1996 Feb;93(2):129-36. doi: 10.1111/j.1600-0447.1996.tb09814.x.
3. Weleff J et al. J Acad Consult Liaison Psychiatry. 2023 Jan-Feb;64(1):13-27. doi:10.1016/j.jaclp.2022.07.002.
4. Oldham MA et al. Bush-Francis Catatonia Rating Scale Assessment Resources. University of Rochester Medical Center, Department of Psychiatry. https://bfcrs.urmc.edu.
5. Oldham MA. Schizophr Res. 2022 Aug 19;S0920-9964(22)00294-8. doi: 10.1016/j.schres.2022.08.002.
6. American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5-TR. Washington, D.C.: American Psychiatric Association Publishing, 2022.
7. World Health Organization. ICD-11 for Mortality and Morbidity Stastistics. 2022. https://icd.who.int/browse11/l-m/en#/http://id.who.int/icd/entity/486722075.
8. Northoff G et al. Mov Disord. May 1999;14(3):404-16. doi: 10.1002/1531-8257(199905)14:3<404::AID-MDS1004>3.0.CO;2-5.
9. Walther S et al. The Lancet Psychiatry. 2019 Jul;6(7):610-9. doi: 10.1016/S2215-0366(18)30474-7.
Sleep abnormalities common in all stages of psychosis
For example, compared with their healthy peers, participants in a chronic psychosis stage had reduced density, amplitude, and duration of spindles – or bursts of brainwave activity during sleep identified by electroencephalography.
“The results suggest sleep could be an important target [and] an area of research and clinical intervention that could make a difference” in the lives of patients at risk for psychosis, study investigator Fabio Ferrarelli, MD, PhD, associate professor of psychiatry and director of the Sleep and Schizophrenia Program, University of Pittsburgh School of Medicine, told this news organization.
The findings were published online in JAMA Psychiatry.
‘Window of opportunity’
Researchers separate psychosis into stages. During the “clinically high-risk for psychosis” (CHR-P) stage, patients have milder symptoms but do not have a diagnosable psychotic disorder. Those in the early psychosis (EP) stage have had a first episode of psychosis. When they reach a cut-off, often at 5 years, they are considered to have chronic psychosis (CP).
Previous studies have shown that altered sleep often precedes a psychotic episode in early psychosis, and disrupted sleep contributes to predicting transition to psychosis in youth at risk for the condition. Individuals with CP commonly report sleep disturbances, such as insomnia.
Following a literature search, the investigators for this current meta-analysis selected 21 studies assessing sleep disturbance prevalence in 5,135 patients. They also selected 39 studies measuring sleep alterations subjectively (for example, sleep quality) and/or objectively (for example, sleep architecture and sleep oscillation) in 1,575 patients and 977 healthy controls.
The included studies measured the prevalence of sleep disturbances and/or sleep characteristics at different psychosis stages using polysomnography, EEG, actigraphy, or self-reports.
The pooled prevalence of sleep disturbances was 50% across clinical stages (95% confidence interval, 40%-61%). The prevalence was 54% in CHR-P, 68% in EP, and 44% in CP.
The prevalence of insomnia as the primary sleep disturbance was 34% of pooled cases, 48% of the EP group, and 27% of the CP group.
“What’s interesting is the rate of sleep disturbances is relatively stable across stages,” said Dr. Ferrarelli. “This is important because you have a window of opportunity to do some early intervention in people who are at risk that can prevent things from getting worse.”
He suggests clinicians screen for insomnia in early-course patients and perhaps recommend cognitive behavioral therapy (CBT) for insomnia. As well, they should promote sleep hygiene measures for at-risk patients, including such things as avoiding caffeine, alcohol, and screen time before bedtime and adopting a regular sleep pattern.
“These are people at risk, which means they have a 20%-30% chance of eventually developing a psychotic disorder,” said Dr. Ferrarelli. “Maybe disrupted sleep is one of the factors that can make a difference.”
Altered sleep architecture
To compare sleep quality between clinical and control groups, studies used total scores on the Pittsburgh Sleep Quality Index (PSQI), where a score over 5 indicates a sleep problem.
There was a significant standardized mean difference in pooled cases versus controls (SMD, 1.0; 95% CI, 0.7-1.3; P < .001). Each clinical group showed poorer sleep quality, compared with controls.
When assessing sleep architecture abnormalities, stage-specific case-control comparisons showed these were driven by EP and CP stages.
Altered sleep characteristics in both these stages included increased sleep onset latency, increased wake after sleep onset, and reduced sleep efficiency.
Compared with controls, CP was the only clinical group with more arousals. Patients with CP also had more arousals than the CHR-P group, and the number of arousals was significantly affected by medication.
The findings indicate the effects of antipsychotic medications on sleep should be closely monitored, especially in CP, the investigators write.
They add that clinicians should consider medication adjustments, such as decreased doses or switches to another compound.
‘Robust’ spindle results
As for spindle parameters, pooled cases showed significantly decreased spindle density (SMD, –1.06), spindle amplitude (SMD, –1.08), and spindle duration (SMD, −1.21), compared with controls. Stage-specific comparisons revealed these deficits were present in both EP and CP relative to controls.
Dr. Ferrarelli noted the results for spindle abnormalities were among “the most robust” and show that these abnormalities “tend to get worse over the course of the illness.”
The spindle data are “a lot more informative” than that provided by other sleep parameters “in the sense they can yield what could be wrong, where it could be, and potentially what you can do about it,” said Dr. Ferrarelli.
“This might be an objective measure that could be used to identify individuals who have a psychosis disorder, monitor progression of illness, and for prognostic reasons,” he added.
He noted that spindles may also represent a promising target for treatment interventions and added that non-invasive transcranial magnetic stimulation has shown promise in restoring sleep oscillations, including spindles.
Another way to evoke target-brain activity may be through auditory tones – with a patient listening to a particular sound through headphones while asleep, Dr. Ferrarelli said.
Reaffirms previous data
Commenting on the study, Jeffrey A. Lieberman, MD, professor and chair in psychiatry at Columbia University, New York, and a past president of the American Psychiatric Association, noted that the review “just reaffirms what has been reported by individual studies for decades.”
That so many at-risk study subjects had a sleep abnormality is not surprising, said Dr. Lieberman, who was not involved with the current research.
“How many individuals in late adolescence or early adulthood have sleep problems?” he asked. “I would venture to say it’s probably a lot. So the question is: How distinctive is this from what occurs in people who don’t develop the illness?”
The aim of sleep research in the area of schizophrenia has long been to disentangle the effects of medication and environmental factors from the disease and to be able to treat patients to normalize their sleep, said Dr. Lieberman.
“But it’s not clear from these results how one would do that,” he added.
The authors “don’t fundamentally tell us anything about the underlying cause of the illness or the pathophysiology, and they don’t really offer any kind of clear direction for clinical intervention,” he said.
The study was supported by the National Institute of Mental Health. Dr. Ferrarelli reported grants from the National Institute of Mental Health during the conduct of the study. Dr. Lieberman has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For example, compared with their healthy peers, participants in a chronic psychosis stage had reduced density, amplitude, and duration of spindles – or bursts of brainwave activity during sleep identified by electroencephalography.
“The results suggest sleep could be an important target [and] an area of research and clinical intervention that could make a difference” in the lives of patients at risk for psychosis, study investigator Fabio Ferrarelli, MD, PhD, associate professor of psychiatry and director of the Sleep and Schizophrenia Program, University of Pittsburgh School of Medicine, told this news organization.
The findings were published online in JAMA Psychiatry.
‘Window of opportunity’
Researchers separate psychosis into stages. During the “clinically high-risk for psychosis” (CHR-P) stage, patients have milder symptoms but do not have a diagnosable psychotic disorder. Those in the early psychosis (EP) stage have had a first episode of psychosis. When they reach a cut-off, often at 5 years, they are considered to have chronic psychosis (CP).
Previous studies have shown that altered sleep often precedes a psychotic episode in early psychosis, and disrupted sleep contributes to predicting transition to psychosis in youth at risk for the condition. Individuals with CP commonly report sleep disturbances, such as insomnia.
Following a literature search, the investigators for this current meta-analysis selected 21 studies assessing sleep disturbance prevalence in 5,135 patients. They also selected 39 studies measuring sleep alterations subjectively (for example, sleep quality) and/or objectively (for example, sleep architecture and sleep oscillation) in 1,575 patients and 977 healthy controls.
The included studies measured the prevalence of sleep disturbances and/or sleep characteristics at different psychosis stages using polysomnography, EEG, actigraphy, or self-reports.
The pooled prevalence of sleep disturbances was 50% across clinical stages (95% confidence interval, 40%-61%). The prevalence was 54% in CHR-P, 68% in EP, and 44% in CP.
The prevalence of insomnia as the primary sleep disturbance was 34% of pooled cases, 48% of the EP group, and 27% of the CP group.
“What’s interesting is the rate of sleep disturbances is relatively stable across stages,” said Dr. Ferrarelli. “This is important because you have a window of opportunity to do some early intervention in people who are at risk that can prevent things from getting worse.”
He suggests clinicians screen for insomnia in early-course patients and perhaps recommend cognitive behavioral therapy (CBT) for insomnia. As well, they should promote sleep hygiene measures for at-risk patients, including such things as avoiding caffeine, alcohol, and screen time before bedtime and adopting a regular sleep pattern.
“These are people at risk, which means they have a 20%-30% chance of eventually developing a psychotic disorder,” said Dr. Ferrarelli. “Maybe disrupted sleep is one of the factors that can make a difference.”
Altered sleep architecture
To compare sleep quality between clinical and control groups, studies used total scores on the Pittsburgh Sleep Quality Index (PSQI), where a score over 5 indicates a sleep problem.
There was a significant standardized mean difference in pooled cases versus controls (SMD, 1.0; 95% CI, 0.7-1.3; P < .001). Each clinical group showed poorer sleep quality, compared with controls.
When assessing sleep architecture abnormalities, stage-specific case-control comparisons showed these were driven by EP and CP stages.
Altered sleep characteristics in both these stages included increased sleep onset latency, increased wake after sleep onset, and reduced sleep efficiency.
Compared with controls, CP was the only clinical group with more arousals. Patients with CP also had more arousals than the CHR-P group, and the number of arousals was significantly affected by medication.
The findings indicate the effects of antipsychotic medications on sleep should be closely monitored, especially in CP, the investigators write.
They add that clinicians should consider medication adjustments, such as decreased doses or switches to another compound.
‘Robust’ spindle results
As for spindle parameters, pooled cases showed significantly decreased spindle density (SMD, –1.06), spindle amplitude (SMD, –1.08), and spindle duration (SMD, −1.21), compared with controls. Stage-specific comparisons revealed these deficits were present in both EP and CP relative to controls.
Dr. Ferrarelli noted the results for spindle abnormalities were among “the most robust” and show that these abnormalities “tend to get worse over the course of the illness.”
The spindle data are “a lot more informative” than that provided by other sleep parameters “in the sense they can yield what could be wrong, where it could be, and potentially what you can do about it,” said Dr. Ferrarelli.
“This might be an objective measure that could be used to identify individuals who have a psychosis disorder, monitor progression of illness, and for prognostic reasons,” he added.
He noted that spindles may also represent a promising target for treatment interventions and added that non-invasive transcranial magnetic stimulation has shown promise in restoring sleep oscillations, including spindles.
Another way to evoke target-brain activity may be through auditory tones – with a patient listening to a particular sound through headphones while asleep, Dr. Ferrarelli said.
Reaffirms previous data
Commenting on the study, Jeffrey A. Lieberman, MD, professor and chair in psychiatry at Columbia University, New York, and a past president of the American Psychiatric Association, noted that the review “just reaffirms what has been reported by individual studies for decades.”
That so many at-risk study subjects had a sleep abnormality is not surprising, said Dr. Lieberman, who was not involved with the current research.
“How many individuals in late adolescence or early adulthood have sleep problems?” he asked. “I would venture to say it’s probably a lot. So the question is: How distinctive is this from what occurs in people who don’t develop the illness?”
The aim of sleep research in the area of schizophrenia has long been to disentangle the effects of medication and environmental factors from the disease and to be able to treat patients to normalize their sleep, said Dr. Lieberman.
“But it’s not clear from these results how one would do that,” he added.
The authors “don’t fundamentally tell us anything about the underlying cause of the illness or the pathophysiology, and they don’t really offer any kind of clear direction for clinical intervention,” he said.
The study was supported by the National Institute of Mental Health. Dr. Ferrarelli reported grants from the National Institute of Mental Health during the conduct of the study. Dr. Lieberman has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For example, compared with their healthy peers, participants in a chronic psychosis stage had reduced density, amplitude, and duration of spindles – or bursts of brainwave activity during sleep identified by electroencephalography.
“The results suggest sleep could be an important target [and] an area of research and clinical intervention that could make a difference” in the lives of patients at risk for psychosis, study investigator Fabio Ferrarelli, MD, PhD, associate professor of psychiatry and director of the Sleep and Schizophrenia Program, University of Pittsburgh School of Medicine, told this news organization.
The findings were published online in JAMA Psychiatry.
‘Window of opportunity’
Researchers separate psychosis into stages. During the “clinically high-risk for psychosis” (CHR-P) stage, patients have milder symptoms but do not have a diagnosable psychotic disorder. Those in the early psychosis (EP) stage have had a first episode of psychosis. When they reach a cut-off, often at 5 years, they are considered to have chronic psychosis (CP).
Previous studies have shown that altered sleep often precedes a psychotic episode in early psychosis, and disrupted sleep contributes to predicting transition to psychosis in youth at risk for the condition. Individuals with CP commonly report sleep disturbances, such as insomnia.
Following a literature search, the investigators for this current meta-analysis selected 21 studies assessing sleep disturbance prevalence in 5,135 patients. They also selected 39 studies measuring sleep alterations subjectively (for example, sleep quality) and/or objectively (for example, sleep architecture and sleep oscillation) in 1,575 patients and 977 healthy controls.
The included studies measured the prevalence of sleep disturbances and/or sleep characteristics at different psychosis stages using polysomnography, EEG, actigraphy, or self-reports.
The pooled prevalence of sleep disturbances was 50% across clinical stages (95% confidence interval, 40%-61%). The prevalence was 54% in CHR-P, 68% in EP, and 44% in CP.
The prevalence of insomnia as the primary sleep disturbance was 34% of pooled cases, 48% of the EP group, and 27% of the CP group.
“What’s interesting is the rate of sleep disturbances is relatively stable across stages,” said Dr. Ferrarelli. “This is important because you have a window of opportunity to do some early intervention in people who are at risk that can prevent things from getting worse.”
He suggests clinicians screen for insomnia in early-course patients and perhaps recommend cognitive behavioral therapy (CBT) for insomnia. As well, they should promote sleep hygiene measures for at-risk patients, including such things as avoiding caffeine, alcohol, and screen time before bedtime and adopting a regular sleep pattern.
“These are people at risk, which means they have a 20%-30% chance of eventually developing a psychotic disorder,” said Dr. Ferrarelli. “Maybe disrupted sleep is one of the factors that can make a difference.”
Altered sleep architecture
To compare sleep quality between clinical and control groups, studies used total scores on the Pittsburgh Sleep Quality Index (PSQI), where a score over 5 indicates a sleep problem.
There was a significant standardized mean difference in pooled cases versus controls (SMD, 1.0; 95% CI, 0.7-1.3; P < .001). Each clinical group showed poorer sleep quality, compared with controls.
When assessing sleep architecture abnormalities, stage-specific case-control comparisons showed these were driven by EP and CP stages.
Altered sleep characteristics in both these stages included increased sleep onset latency, increased wake after sleep onset, and reduced sleep efficiency.
Compared with controls, CP was the only clinical group with more arousals. Patients with CP also had more arousals than the CHR-P group, and the number of arousals was significantly affected by medication.
The findings indicate the effects of antipsychotic medications on sleep should be closely monitored, especially in CP, the investigators write.
They add that clinicians should consider medication adjustments, such as decreased doses or switches to another compound.
‘Robust’ spindle results
As for spindle parameters, pooled cases showed significantly decreased spindle density (SMD, –1.06), spindle amplitude (SMD, –1.08), and spindle duration (SMD, −1.21), compared with controls. Stage-specific comparisons revealed these deficits were present in both EP and CP relative to controls.
Dr. Ferrarelli noted the results for spindle abnormalities were among “the most robust” and show that these abnormalities “tend to get worse over the course of the illness.”
The spindle data are “a lot more informative” than that provided by other sleep parameters “in the sense they can yield what could be wrong, where it could be, and potentially what you can do about it,” said Dr. Ferrarelli.
“This might be an objective measure that could be used to identify individuals who have a psychosis disorder, monitor progression of illness, and for prognostic reasons,” he added.
He noted that spindles may also represent a promising target for treatment interventions and added that non-invasive transcranial magnetic stimulation has shown promise in restoring sleep oscillations, including spindles.
Another way to evoke target-brain activity may be through auditory tones – with a patient listening to a particular sound through headphones while asleep, Dr. Ferrarelli said.
Reaffirms previous data
Commenting on the study, Jeffrey A. Lieberman, MD, professor and chair in psychiatry at Columbia University, New York, and a past president of the American Psychiatric Association, noted that the review “just reaffirms what has been reported by individual studies for decades.”
That so many at-risk study subjects had a sleep abnormality is not surprising, said Dr. Lieberman, who was not involved with the current research.
“How many individuals in late adolescence or early adulthood have sleep problems?” he asked. “I would venture to say it’s probably a lot. So the question is: How distinctive is this from what occurs in people who don’t develop the illness?”
The aim of sleep research in the area of schizophrenia has long been to disentangle the effects of medication and environmental factors from the disease and to be able to treat patients to normalize their sleep, said Dr. Lieberman.
“But it’s not clear from these results how one would do that,” he added.
The authors “don’t fundamentally tell us anything about the underlying cause of the illness or the pathophysiology, and they don’t really offer any kind of clear direction for clinical intervention,” he said.
The study was supported by the National Institute of Mental Health. Dr. Ferrarelli reported grants from the National Institute of Mental Health during the conduct of the study. Dr. Lieberman has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Lipid signature may flag schizophrenia
Although such a test remains a long way off, investigators said, the identification of the unique lipid signature is a critical first step. However, one expert noted that the lipid signature not accurately differentiating patients with schizophrenia from those with bipolar disorder (BD) and major depressive disorder (MDD) limits the findings’ applicability.
The profile includes 77 lipids identified from a large analysis of many different classes of lipid species. Lipids such as cholesterol and triglycerides made up only a small fraction of the classes assessed.
The investigators noted that some of the lipids in the profile associated with schizophrenia are involved in determining cell membrane structure and fluidity or cell-to-cell messaging, which could be important to synaptic function.
“These 77 lipids jointly constitute a lipidomic profile that discriminated between individuals with schizophrenia and individuals without a mental health diagnosis with very high accuracy,” investigator Eva C. Schulte, MD, PhD, of the Institute of Psychiatric Phenomics and Genomics (IPPG) and the department of psychiatry and psychotherapy at University Hospital of Ludwig-Maximilians-University, Munich, told this news organization.
“Of note, we did not see large profile differences between patients with a first psychotic episode who had only been treated for a few days and individuals on long-term antipsychotic therapy,” Dr. Schulte said.
The findings were published online in JAMA Psychiatry.
Detailed analysis
Lipid profiles in patients with psychiatric diagnoses have been reported previously, but those studies were small and did not identify a reliable signature independent of demographic and environmental factors.
For the current study, researchers analyzed blood plasma lipid levels from 980 individuals with severe psychiatric illness and 572 people without mental illness from three cohorts in China, Germany, Austria, and Russia.
The study sample included patients with schizophrenia (n = 478), BD (n = 184), and MDD (n = 256), as well as 104 patients with a first psychotic episode who had no long-term psychopharmacology use.
Results showed 77 lipids in 14 classes were significantly altered between participants with schizophrenia and the healthy control in all three cohorts.
The most prominent alterations at the lipid class level included increases in ceramide, triacylglyceride, and phosphatidylcholine and decreases in acylcarnitine and phosphatidylcholine plasmalogen (P < .05 for each cohort).
Schizophrenia-associated lipid differences were similar between patients with high and low symptom severity (P < .001), suggesting that the lipid alterations might represent a trait of the psychiatric disorder.
No medication effect
Most patients in the study received long-term antipsychotic medication, which has been shown previously to affect some plasma lipid compounds.
So, to assess a possible effect of medication, the investigators evaluated 13 patients with schizophrenia who were not medicated for at least 6 months prior to blood sample collection and the cohort of patients with a first psychotic episode who had been medicated for less than 1 week.
Comparison of the lipid intensity differences between the healthy controls group and either participants receiving medication or those who were not medicated revealed highly correlated alterations in both patient groups (P < .001).
“Taken together, these results indicate that the identified schizophrenia-associated alterations cannot be attributed to medication effects,” the investigators wrote.
Lipidome alterations in BPD and MDD, assessed in 184 and 256 individuals, respectively, were similar to those of schizophrenia but not identical.
Researchers isolated 97 lipids altered in the MDD cohorts and 47 in the BPD cohorts – with 30 and 28, respectively, overlapping with the schizophrenia-associated features and seven of the lipids found among all three disorders.
Although this was significantly more than expected by chance (P < .001), it was not strong enough to demonstrate a clear association, the investigators wrote.
“The profiles were very successful at differentiating individuals with severe mental health conditions from individuals without a diagnosed mental health condition, but much less so at differentiating between the different diagnostic entities,” coinvestigator Thomas G. Schulze, MD, director of IPPG, said in an interview.
“An important caveat, however, is that the available sample sizes for bipolar disorder and major depressive disorder were smaller than those for schizophrenia, which makes a direct comparison between these difficult,” added Dr. Schulze, clinical professor in psychiatry and behavioral sciences at State University of New York, Syracuse.
More work remains
Although the study is thought to be the largest to date to examine lipid profiles associated with serious psychiatric illness, much work remains, Dr. Schulze noted.
“At this time, based on these first results, no clinical diagnostic test can be derived from these results,” he said.
He added that the development of reliable biomarkers based on lipidomic profiles would require large prospective randomized trials, complemented by observational studies assessing full lipidomic profiles across the lifespan.
Researchers also need to better understand the exact mechanism by which lipid alterations are associated with schizophrenia and other illnesses.
Physiologically, the investigated lipids have many additional functions, such as determining cell membrane structure and fluidity or cell-to-cell messaging.
Dr. Schulte noted that several lipid species may be involved in determining mechanisms important to synaptic function, such as cell membrane fluidity and vesicle release.
“As is commonly known, alterations in synaptic function underly many severe psychiatric disorders,” she said. “Changes in lipid species could theoretically be related to these synaptic alterations.”
A better marker needed
In a comment, Stephen Strakowski, MD, professor and vice chair of research in the department of psychiatry, Indiana University, Indianapolis and Evansville, noted that while the findings are interesting, they don’t really offer the kind of information clinicians who treat patients with serious mental illness need most.
“Do we need a marker to tell us if someone’s got a major mental illness compared to a healthy person?” asked Dr. Strakowski, who was not part of the study. “The answer to that is no. We already know how to do that.”
A truly useful marker would help clinicians differentiate between schizophrenia, bipolar disorder, major depression, or another serious mental illness, he said.
“That’s the marker that would be most helpful,” he added. “This can’t address that, but perhaps it could be a step to start designing a test for that.”
Dr. Strakowksi noted that the findings do not clarify whether the lipid profile found in patients with schizophrenia predates diagnosis or whether it is a result of the mental illness, an unrelated illness, or another factor that could be critical in treating patients.
However, he was quick to point out the limitations don’t diminish the importance of the study.
“It’s a large dataset that’s cross-national, cross-diagnostic that says there appears to be a signal here that there’s something about lipid profiles that may be independent of treatment that could be worth understanding,” Dr. Strakowksi said.
“It allows us to think about developing different models based on lipid profiles, and that’s important,” he added.
The study was funded by the National Key R&D Program of China, National One Thousand Foreign Experts Plan, Moscow Center for Innovative Technologies in Healthcare, European Union’s Horizon 2020 Research and Innovation Programme, NARSAD Young Investigator Grant, German Research Foundation, German Ministry for Education and Research, the Dr. Lisa Oehler Foundation, and the Munich Clinician Scientist Program. Dr. Schulze and Dr. Schulte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Although such a test remains a long way off, investigators said, the identification of the unique lipid signature is a critical first step. However, one expert noted that the lipid signature not accurately differentiating patients with schizophrenia from those with bipolar disorder (BD) and major depressive disorder (MDD) limits the findings’ applicability.
The profile includes 77 lipids identified from a large analysis of many different classes of lipid species. Lipids such as cholesterol and triglycerides made up only a small fraction of the classes assessed.
The investigators noted that some of the lipids in the profile associated with schizophrenia are involved in determining cell membrane structure and fluidity or cell-to-cell messaging, which could be important to synaptic function.
“These 77 lipids jointly constitute a lipidomic profile that discriminated between individuals with schizophrenia and individuals without a mental health diagnosis with very high accuracy,” investigator Eva C. Schulte, MD, PhD, of the Institute of Psychiatric Phenomics and Genomics (IPPG) and the department of psychiatry and psychotherapy at University Hospital of Ludwig-Maximilians-University, Munich, told this news organization.
“Of note, we did not see large profile differences between patients with a first psychotic episode who had only been treated for a few days and individuals on long-term antipsychotic therapy,” Dr. Schulte said.
The findings were published online in JAMA Psychiatry.
Detailed analysis
Lipid profiles in patients with psychiatric diagnoses have been reported previously, but those studies were small and did not identify a reliable signature independent of demographic and environmental factors.
For the current study, researchers analyzed blood plasma lipid levels from 980 individuals with severe psychiatric illness and 572 people without mental illness from three cohorts in China, Germany, Austria, and Russia.
The study sample included patients with schizophrenia (n = 478), BD (n = 184), and MDD (n = 256), as well as 104 patients with a first psychotic episode who had no long-term psychopharmacology use.
Results showed 77 lipids in 14 classes were significantly altered between participants with schizophrenia and the healthy control in all three cohorts.
The most prominent alterations at the lipid class level included increases in ceramide, triacylglyceride, and phosphatidylcholine and decreases in acylcarnitine and phosphatidylcholine plasmalogen (P < .05 for each cohort).
Schizophrenia-associated lipid differences were similar between patients with high and low symptom severity (P < .001), suggesting that the lipid alterations might represent a trait of the psychiatric disorder.
No medication effect
Most patients in the study received long-term antipsychotic medication, which has been shown previously to affect some plasma lipid compounds.
So, to assess a possible effect of medication, the investigators evaluated 13 patients with schizophrenia who were not medicated for at least 6 months prior to blood sample collection and the cohort of patients with a first psychotic episode who had been medicated for less than 1 week.
Comparison of the lipid intensity differences between the healthy controls group and either participants receiving medication or those who were not medicated revealed highly correlated alterations in both patient groups (P < .001).
“Taken together, these results indicate that the identified schizophrenia-associated alterations cannot be attributed to medication effects,” the investigators wrote.
Lipidome alterations in BPD and MDD, assessed in 184 and 256 individuals, respectively, were similar to those of schizophrenia but not identical.
Researchers isolated 97 lipids altered in the MDD cohorts and 47 in the BPD cohorts – with 30 and 28, respectively, overlapping with the schizophrenia-associated features and seven of the lipids found among all three disorders.
Although this was significantly more than expected by chance (P < .001), it was not strong enough to demonstrate a clear association, the investigators wrote.
“The profiles were very successful at differentiating individuals with severe mental health conditions from individuals without a diagnosed mental health condition, but much less so at differentiating between the different diagnostic entities,” coinvestigator Thomas G. Schulze, MD, director of IPPG, said in an interview.
“An important caveat, however, is that the available sample sizes for bipolar disorder and major depressive disorder were smaller than those for schizophrenia, which makes a direct comparison between these difficult,” added Dr. Schulze, clinical professor in psychiatry and behavioral sciences at State University of New York, Syracuse.
More work remains
Although the study is thought to be the largest to date to examine lipid profiles associated with serious psychiatric illness, much work remains, Dr. Schulze noted.
“At this time, based on these first results, no clinical diagnostic test can be derived from these results,” he said.
He added that the development of reliable biomarkers based on lipidomic profiles would require large prospective randomized trials, complemented by observational studies assessing full lipidomic profiles across the lifespan.
Researchers also need to better understand the exact mechanism by which lipid alterations are associated with schizophrenia and other illnesses.
Physiologically, the investigated lipids have many additional functions, such as determining cell membrane structure and fluidity or cell-to-cell messaging.
Dr. Schulte noted that several lipid species may be involved in determining mechanisms important to synaptic function, such as cell membrane fluidity and vesicle release.
“As is commonly known, alterations in synaptic function underly many severe psychiatric disorders,” she said. “Changes in lipid species could theoretically be related to these synaptic alterations.”
A better marker needed
In a comment, Stephen Strakowski, MD, professor and vice chair of research in the department of psychiatry, Indiana University, Indianapolis and Evansville, noted that while the findings are interesting, they don’t really offer the kind of information clinicians who treat patients with serious mental illness need most.
“Do we need a marker to tell us if someone’s got a major mental illness compared to a healthy person?” asked Dr. Strakowski, who was not part of the study. “The answer to that is no. We already know how to do that.”
A truly useful marker would help clinicians differentiate between schizophrenia, bipolar disorder, major depression, or another serious mental illness, he said.
“That’s the marker that would be most helpful,” he added. “This can’t address that, but perhaps it could be a step to start designing a test for that.”
Dr. Strakowksi noted that the findings do not clarify whether the lipid profile found in patients with schizophrenia predates diagnosis or whether it is a result of the mental illness, an unrelated illness, or another factor that could be critical in treating patients.
However, he was quick to point out the limitations don’t diminish the importance of the study.
“It’s a large dataset that’s cross-national, cross-diagnostic that says there appears to be a signal here that there’s something about lipid profiles that may be independent of treatment that could be worth understanding,” Dr. Strakowksi said.
“It allows us to think about developing different models based on lipid profiles, and that’s important,” he added.
The study was funded by the National Key R&D Program of China, National One Thousand Foreign Experts Plan, Moscow Center for Innovative Technologies in Healthcare, European Union’s Horizon 2020 Research and Innovation Programme, NARSAD Young Investigator Grant, German Research Foundation, German Ministry for Education and Research, the Dr. Lisa Oehler Foundation, and the Munich Clinician Scientist Program. Dr. Schulze and Dr. Schulte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Although such a test remains a long way off, investigators said, the identification of the unique lipid signature is a critical first step. However, one expert noted that the lipid signature not accurately differentiating patients with schizophrenia from those with bipolar disorder (BD) and major depressive disorder (MDD) limits the findings’ applicability.
The profile includes 77 lipids identified from a large analysis of many different classes of lipid species. Lipids such as cholesterol and triglycerides made up only a small fraction of the classes assessed.
The investigators noted that some of the lipids in the profile associated with schizophrenia are involved in determining cell membrane structure and fluidity or cell-to-cell messaging, which could be important to synaptic function.
“These 77 lipids jointly constitute a lipidomic profile that discriminated between individuals with schizophrenia and individuals without a mental health diagnosis with very high accuracy,” investigator Eva C. Schulte, MD, PhD, of the Institute of Psychiatric Phenomics and Genomics (IPPG) and the department of psychiatry and psychotherapy at University Hospital of Ludwig-Maximilians-University, Munich, told this news organization.
“Of note, we did not see large profile differences between patients with a first psychotic episode who had only been treated for a few days and individuals on long-term antipsychotic therapy,” Dr. Schulte said.
The findings were published online in JAMA Psychiatry.
Detailed analysis
Lipid profiles in patients with psychiatric diagnoses have been reported previously, but those studies were small and did not identify a reliable signature independent of demographic and environmental factors.
For the current study, researchers analyzed blood plasma lipid levels from 980 individuals with severe psychiatric illness and 572 people without mental illness from three cohorts in China, Germany, Austria, and Russia.
The study sample included patients with schizophrenia (n = 478), BD (n = 184), and MDD (n = 256), as well as 104 patients with a first psychotic episode who had no long-term psychopharmacology use.
Results showed 77 lipids in 14 classes were significantly altered between participants with schizophrenia and the healthy control in all three cohorts.
The most prominent alterations at the lipid class level included increases in ceramide, triacylglyceride, and phosphatidylcholine and decreases in acylcarnitine and phosphatidylcholine plasmalogen (P < .05 for each cohort).
Schizophrenia-associated lipid differences were similar between patients with high and low symptom severity (P < .001), suggesting that the lipid alterations might represent a trait of the psychiatric disorder.
No medication effect
Most patients in the study received long-term antipsychotic medication, which has been shown previously to affect some plasma lipid compounds.
So, to assess a possible effect of medication, the investigators evaluated 13 patients with schizophrenia who were not medicated for at least 6 months prior to blood sample collection and the cohort of patients with a first psychotic episode who had been medicated for less than 1 week.
Comparison of the lipid intensity differences between the healthy controls group and either participants receiving medication or those who were not medicated revealed highly correlated alterations in both patient groups (P < .001).
“Taken together, these results indicate that the identified schizophrenia-associated alterations cannot be attributed to medication effects,” the investigators wrote.
Lipidome alterations in BPD and MDD, assessed in 184 and 256 individuals, respectively, were similar to those of schizophrenia but not identical.
Researchers isolated 97 lipids altered in the MDD cohorts and 47 in the BPD cohorts – with 30 and 28, respectively, overlapping with the schizophrenia-associated features and seven of the lipids found among all three disorders.
Although this was significantly more than expected by chance (P < .001), it was not strong enough to demonstrate a clear association, the investigators wrote.
“The profiles were very successful at differentiating individuals with severe mental health conditions from individuals without a diagnosed mental health condition, but much less so at differentiating between the different diagnostic entities,” coinvestigator Thomas G. Schulze, MD, director of IPPG, said in an interview.
“An important caveat, however, is that the available sample sizes for bipolar disorder and major depressive disorder were smaller than those for schizophrenia, which makes a direct comparison between these difficult,” added Dr. Schulze, clinical professor in psychiatry and behavioral sciences at State University of New York, Syracuse.
More work remains
Although the study is thought to be the largest to date to examine lipid profiles associated with serious psychiatric illness, much work remains, Dr. Schulze noted.
“At this time, based on these first results, no clinical diagnostic test can be derived from these results,” he said.
He added that the development of reliable biomarkers based on lipidomic profiles would require large prospective randomized trials, complemented by observational studies assessing full lipidomic profiles across the lifespan.
Researchers also need to better understand the exact mechanism by which lipid alterations are associated with schizophrenia and other illnesses.
Physiologically, the investigated lipids have many additional functions, such as determining cell membrane structure and fluidity or cell-to-cell messaging.
Dr. Schulte noted that several lipid species may be involved in determining mechanisms important to synaptic function, such as cell membrane fluidity and vesicle release.
“As is commonly known, alterations in synaptic function underly many severe psychiatric disorders,” she said. “Changes in lipid species could theoretically be related to these synaptic alterations.”
A better marker needed
In a comment, Stephen Strakowski, MD, professor and vice chair of research in the department of psychiatry, Indiana University, Indianapolis and Evansville, noted that while the findings are interesting, they don’t really offer the kind of information clinicians who treat patients with serious mental illness need most.
“Do we need a marker to tell us if someone’s got a major mental illness compared to a healthy person?” asked Dr. Strakowski, who was not part of the study. “The answer to that is no. We already know how to do that.”
A truly useful marker would help clinicians differentiate between schizophrenia, bipolar disorder, major depression, or another serious mental illness, he said.
“That’s the marker that would be most helpful,” he added. “This can’t address that, but perhaps it could be a step to start designing a test for that.”
Dr. Strakowksi noted that the findings do not clarify whether the lipid profile found in patients with schizophrenia predates diagnosis or whether it is a result of the mental illness, an unrelated illness, or another factor that could be critical in treating patients.
However, he was quick to point out the limitations don’t diminish the importance of the study.
“It’s a large dataset that’s cross-national, cross-diagnostic that says there appears to be a signal here that there’s something about lipid profiles that may be independent of treatment that could be worth understanding,” Dr. Strakowksi said.
“It allows us to think about developing different models based on lipid profiles, and that’s important,” he added.
The study was funded by the National Key R&D Program of China, National One Thousand Foreign Experts Plan, Moscow Center for Innovative Technologies in Healthcare, European Union’s Horizon 2020 Research and Innovation Programme, NARSAD Young Investigator Grant, German Research Foundation, German Ministry for Education and Research, the Dr. Lisa Oehler Foundation, and the Munich Clinician Scientist Program. Dr. Schulze and Dr. Schulte reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY

















