User login
Metformin fails as early COVID-19 treatment but shows potential
Neither metformin, ivermectin, or fluvoxamine had any impact on reducing disease severity, hospitalization, or death from COVID-19, according to results from more than 1,000 overweight or obese adult patients in the COVID-OUT randomized trial.
However, metformin showed some potential in a secondary analysis.
Early treatment to prevent severe disease remains a goal in managing the ongoing COVID-19 pandemic, and biophysical modeling suggested that metformin, ivermectin, and fluvoxamine may serve as antivirals to help reduce severe disease in COVID-19 patients, Carolyn T. Bramante, MD, of the University of Minnesota, Minneapolis, and colleagues wrote.
“We started enrolling patients at the end of December 2020,” Dr. Bramante said in an interview. “At that time, even though vaccine data were coming out, we thought it was important to test early outpatient treatment with widely available safe medications with no interactions, because the virus would evolve and vaccine availability may be limited.”
In a study published in the New England Journal of Medicine, the researchers used a two-by-three factorial design to test the ability of metformin, ivermectin, and fluvoxamine to prevent severe COVID-19 infection in nonhospitalized adults aged 30-85 years. A total of 1,431 patients at six U.S. sites were enrolled within 3 days of a confirmed infection and less than 7 days after the start of symptoms, then randomized to one of six groups: metformin plus fluvoxamine; metformin plus ivermectin; metformin plus placebo; placebo plus fluvoxamine; placebo plus ivermectin; and placebo plus placebo.
A total of 1,323 patients were included in the primary analysis. The median age of the patients was 46 years, 56% were female (of whom 6% were pregnant), and all individuals met criteria for overweight or obesity. About half (52%) of the patients had been vaccinated against COVID-19.
The primary endpoint was a composite of hypoxemia, ED visit, hospitalization, or death. The analyses were adjusted for COVID-19 vaccination and other trial medications. Overall, the adjusted odds ratios of any primary event, compared with placebo, was 0.84 for metformin (P = .19), 1.05 for ivermectin (P = .78), and 0.94 for fluvoxamine (P = .75).
The researchers also conducted a prespecified secondary analysis of components of the primary endpoint. In this analysis, the aORs for an ED visit, hospitalization, or death was 0.58 for metformin, 1.39 for ivermectin, and 1.17 for fluvoxamine. The aORs for hospitalization or death were 0.47, 0.73, and 1.11 for metformin, ivermectin, and fluvoxamine, respectively. No medication-related serious adverse events were reported with any of the drugs during the study period.
The possible benefit for prevention of severe COVID-19 with metformin was a prespecified secondary endpoint, and therefore not definitive until more research has been completed, the researchers said. Metformin has demonstrated anti-inflammatory actions in previous studies, and has shown protective effects against COVID-19 lung injury in animal studies.
Previous observational studies also have shown an association between metformin use and less severe COVID-19 in patients already taking metformin. “The proposed mechanisms of action against COVID-19 for metformin include anti-inflammatory and antiviral activity and the prevention of hyperglycemia during acute illness,” they added.
The study findings were limited by several factors including the population age range and focus on overweight and obese patients, which may limit generalizability, the researchers noted. Other limitations include the disproportionately small percentage of Black and Latino patients and the potential lack of accuracy in identifying hypoxemia via home oxygen monitors.
However, the results demonstrate that none of the three repurposed drugs – metformin, ivermectin, and fluvoxamine – prevented primary events or reduced symptom severity in COVID-19, compared with placebos, the researchers concluded.
“Metformin had several streams of evidence supporting its use: in vitro, in silico [computer modeled], observational, and in tissue. We were not surprised to see that it reduced emergency department visits, hospitalization, and death,” Dr. Bramante said in an interview.
The take-home message for clinicians is to continue to look to guideline committees for direction on COVID-19 treatments, but to continue to consider metformin along with other treatments, she said.
“All research should be replicated, whether the primary outcome is positive or negative,” Dr. Bramante emphasized. “In this case, when our positive outcome was negative and secondary outcome was positive, a confirmatory trial for metformin is particularly important.”
Ineffective drugs are inefficient use of resources
“The results of the COVID-OUT trial provide persuasive additional data that increase the confidence and degree of certainty that fluvoxamine and ivermectin are not effective in preventing progression to severe disease,” wrote Salim S. Abdool Karim, MB, and Nikita Devnarain, PhD, of the Centre for the AIDS Programme of Research in South Africa, Durban, in an accompanying editorial.
At the start of the study, in 2020, data on the use of the three drugs to prevent severe COVID-19 were “either unavailable or equivocal,” they said. Since then, accumulating data support the current study findings of the nonefficacy of ivermectin and fluvoxamine, and the World Health Organization has advised against their use for COVID-19, although the WHO has not provided guidance for the use of metformin.
The authors called on clinicians to stop using ivermectin and fluvoxamine to treat COVID-19 patients.
“With respect to clinical decisions about COVID-19 treatment, some drug choices, especially those that have negative [World Health Organization] recommendations, are clearly wrong,” they wrote. “In keeping with evidence-based medical practice, patients with COVID-19 must be treated with efficacious medications; they deserve nothing less.”
The study was supported by the Parsemus Foundation, Rainwater Charitable Foundation, Fast Grants, and UnitedHealth Group Foundation. The fluvoxamine placebo tablets were donated by Apotex Pharmaceuticals. The ivermectin placebo and active tablets were donated by Edenbridge Pharmaceuticals. Lead author Dr. Bramante was supported the National Center for Advancing Translational Sciences and the National Institute of Diabetes and Digestive and Kidney Diseases. The researchers had no financial conflicts to disclose. Dr. Abdool Karim serves as a member of the World Health Organization Science Council. Dr. Devnarain had no financial conflicts to disclose.
Neither metformin, ivermectin, or fluvoxamine had any impact on reducing disease severity, hospitalization, or death from COVID-19, according to results from more than 1,000 overweight or obese adult patients in the COVID-OUT randomized trial.
However, metformin showed some potential in a secondary analysis.
Early treatment to prevent severe disease remains a goal in managing the ongoing COVID-19 pandemic, and biophysical modeling suggested that metformin, ivermectin, and fluvoxamine may serve as antivirals to help reduce severe disease in COVID-19 patients, Carolyn T. Bramante, MD, of the University of Minnesota, Minneapolis, and colleagues wrote.
“We started enrolling patients at the end of December 2020,” Dr. Bramante said in an interview. “At that time, even though vaccine data were coming out, we thought it was important to test early outpatient treatment with widely available safe medications with no interactions, because the virus would evolve and vaccine availability may be limited.”
In a study published in the New England Journal of Medicine, the researchers used a two-by-three factorial design to test the ability of metformin, ivermectin, and fluvoxamine to prevent severe COVID-19 infection in nonhospitalized adults aged 30-85 years. A total of 1,431 patients at six U.S. sites were enrolled within 3 days of a confirmed infection and less than 7 days after the start of symptoms, then randomized to one of six groups: metformin plus fluvoxamine; metformin plus ivermectin; metformin plus placebo; placebo plus fluvoxamine; placebo plus ivermectin; and placebo plus placebo.
A total of 1,323 patients were included in the primary analysis. The median age of the patients was 46 years, 56% were female (of whom 6% were pregnant), and all individuals met criteria for overweight or obesity. About half (52%) of the patients had been vaccinated against COVID-19.
The primary endpoint was a composite of hypoxemia, ED visit, hospitalization, or death. The analyses were adjusted for COVID-19 vaccination and other trial medications. Overall, the adjusted odds ratios of any primary event, compared with placebo, was 0.84 for metformin (P = .19), 1.05 for ivermectin (P = .78), and 0.94 for fluvoxamine (P = .75).
The researchers also conducted a prespecified secondary analysis of components of the primary endpoint. In this analysis, the aORs for an ED visit, hospitalization, or death was 0.58 for metformin, 1.39 for ivermectin, and 1.17 for fluvoxamine. The aORs for hospitalization or death were 0.47, 0.73, and 1.11 for metformin, ivermectin, and fluvoxamine, respectively. No medication-related serious adverse events were reported with any of the drugs during the study period.
The possible benefit for prevention of severe COVID-19 with metformin was a prespecified secondary endpoint, and therefore not definitive until more research has been completed, the researchers said. Metformin has demonstrated anti-inflammatory actions in previous studies, and has shown protective effects against COVID-19 lung injury in animal studies.
Previous observational studies also have shown an association between metformin use and less severe COVID-19 in patients already taking metformin. “The proposed mechanisms of action against COVID-19 for metformin include anti-inflammatory and antiviral activity and the prevention of hyperglycemia during acute illness,” they added.
The study findings were limited by several factors including the population age range and focus on overweight and obese patients, which may limit generalizability, the researchers noted. Other limitations include the disproportionately small percentage of Black and Latino patients and the potential lack of accuracy in identifying hypoxemia via home oxygen monitors.
However, the results demonstrate that none of the three repurposed drugs – metformin, ivermectin, and fluvoxamine – prevented primary events or reduced symptom severity in COVID-19, compared with placebos, the researchers concluded.
“Metformin had several streams of evidence supporting its use: in vitro, in silico [computer modeled], observational, and in tissue. We were not surprised to see that it reduced emergency department visits, hospitalization, and death,” Dr. Bramante said in an interview.
The take-home message for clinicians is to continue to look to guideline committees for direction on COVID-19 treatments, but to continue to consider metformin along with other treatments, she said.
“All research should be replicated, whether the primary outcome is positive or negative,” Dr. Bramante emphasized. “In this case, when our positive outcome was negative and secondary outcome was positive, a confirmatory trial for metformin is particularly important.”
Ineffective drugs are inefficient use of resources
“The results of the COVID-OUT trial provide persuasive additional data that increase the confidence and degree of certainty that fluvoxamine and ivermectin are not effective in preventing progression to severe disease,” wrote Salim S. Abdool Karim, MB, and Nikita Devnarain, PhD, of the Centre for the AIDS Programme of Research in South Africa, Durban, in an accompanying editorial.
At the start of the study, in 2020, data on the use of the three drugs to prevent severe COVID-19 were “either unavailable or equivocal,” they said. Since then, accumulating data support the current study findings of the nonefficacy of ivermectin and fluvoxamine, and the World Health Organization has advised against their use for COVID-19, although the WHO has not provided guidance for the use of metformin.
The authors called on clinicians to stop using ivermectin and fluvoxamine to treat COVID-19 patients.
“With respect to clinical decisions about COVID-19 treatment, some drug choices, especially those that have negative [World Health Organization] recommendations, are clearly wrong,” they wrote. “In keeping with evidence-based medical practice, patients with COVID-19 must be treated with efficacious medications; they deserve nothing less.”
The study was supported by the Parsemus Foundation, Rainwater Charitable Foundation, Fast Grants, and UnitedHealth Group Foundation. The fluvoxamine placebo tablets were donated by Apotex Pharmaceuticals. The ivermectin placebo and active tablets were donated by Edenbridge Pharmaceuticals. Lead author Dr. Bramante was supported the National Center for Advancing Translational Sciences and the National Institute of Diabetes and Digestive and Kidney Diseases. The researchers had no financial conflicts to disclose. Dr. Abdool Karim serves as a member of the World Health Organization Science Council. Dr. Devnarain had no financial conflicts to disclose.
Neither metformin, ivermectin, or fluvoxamine had any impact on reducing disease severity, hospitalization, or death from COVID-19, according to results from more than 1,000 overweight or obese adult patients in the COVID-OUT randomized trial.
However, metformin showed some potential in a secondary analysis.
Early treatment to prevent severe disease remains a goal in managing the ongoing COVID-19 pandemic, and biophysical modeling suggested that metformin, ivermectin, and fluvoxamine may serve as antivirals to help reduce severe disease in COVID-19 patients, Carolyn T. Bramante, MD, of the University of Minnesota, Minneapolis, and colleagues wrote.
“We started enrolling patients at the end of December 2020,” Dr. Bramante said in an interview. “At that time, even though vaccine data were coming out, we thought it was important to test early outpatient treatment with widely available safe medications with no interactions, because the virus would evolve and vaccine availability may be limited.”
In a study published in the New England Journal of Medicine, the researchers used a two-by-three factorial design to test the ability of metformin, ivermectin, and fluvoxamine to prevent severe COVID-19 infection in nonhospitalized adults aged 30-85 years. A total of 1,431 patients at six U.S. sites were enrolled within 3 days of a confirmed infection and less than 7 days after the start of symptoms, then randomized to one of six groups: metformin plus fluvoxamine; metformin plus ivermectin; metformin plus placebo; placebo plus fluvoxamine; placebo plus ivermectin; and placebo plus placebo.
A total of 1,323 patients were included in the primary analysis. The median age of the patients was 46 years, 56% were female (of whom 6% were pregnant), and all individuals met criteria for overweight or obesity. About half (52%) of the patients had been vaccinated against COVID-19.
The primary endpoint was a composite of hypoxemia, ED visit, hospitalization, or death. The analyses were adjusted for COVID-19 vaccination and other trial medications. Overall, the adjusted odds ratios of any primary event, compared with placebo, was 0.84 for metformin (P = .19), 1.05 for ivermectin (P = .78), and 0.94 for fluvoxamine (P = .75).
The researchers also conducted a prespecified secondary analysis of components of the primary endpoint. In this analysis, the aORs for an ED visit, hospitalization, or death was 0.58 for metformin, 1.39 for ivermectin, and 1.17 for fluvoxamine. The aORs for hospitalization or death were 0.47, 0.73, and 1.11 for metformin, ivermectin, and fluvoxamine, respectively. No medication-related serious adverse events were reported with any of the drugs during the study period.
The possible benefit for prevention of severe COVID-19 with metformin was a prespecified secondary endpoint, and therefore not definitive until more research has been completed, the researchers said. Metformin has demonstrated anti-inflammatory actions in previous studies, and has shown protective effects against COVID-19 lung injury in animal studies.
Previous observational studies also have shown an association between metformin use and less severe COVID-19 in patients already taking metformin. “The proposed mechanisms of action against COVID-19 for metformin include anti-inflammatory and antiviral activity and the prevention of hyperglycemia during acute illness,” they added.
The study findings were limited by several factors including the population age range and focus on overweight and obese patients, which may limit generalizability, the researchers noted. Other limitations include the disproportionately small percentage of Black and Latino patients and the potential lack of accuracy in identifying hypoxemia via home oxygen monitors.
However, the results demonstrate that none of the three repurposed drugs – metformin, ivermectin, and fluvoxamine – prevented primary events or reduced symptom severity in COVID-19, compared with placebos, the researchers concluded.
“Metformin had several streams of evidence supporting its use: in vitro, in silico [computer modeled], observational, and in tissue. We were not surprised to see that it reduced emergency department visits, hospitalization, and death,” Dr. Bramante said in an interview.
The take-home message for clinicians is to continue to look to guideline committees for direction on COVID-19 treatments, but to continue to consider metformin along with other treatments, she said.
“All research should be replicated, whether the primary outcome is positive or negative,” Dr. Bramante emphasized. “In this case, when our positive outcome was negative and secondary outcome was positive, a confirmatory trial for metformin is particularly important.”
Ineffective drugs are inefficient use of resources
“The results of the COVID-OUT trial provide persuasive additional data that increase the confidence and degree of certainty that fluvoxamine and ivermectin are not effective in preventing progression to severe disease,” wrote Salim S. Abdool Karim, MB, and Nikita Devnarain, PhD, of the Centre for the AIDS Programme of Research in South Africa, Durban, in an accompanying editorial.
At the start of the study, in 2020, data on the use of the three drugs to prevent severe COVID-19 were “either unavailable or equivocal,” they said. Since then, accumulating data support the current study findings of the nonefficacy of ivermectin and fluvoxamine, and the World Health Organization has advised against their use for COVID-19, although the WHO has not provided guidance for the use of metformin.
The authors called on clinicians to stop using ivermectin and fluvoxamine to treat COVID-19 patients.
“With respect to clinical decisions about COVID-19 treatment, some drug choices, especially those that have negative [World Health Organization] recommendations, are clearly wrong,” they wrote. “In keeping with evidence-based medical practice, patients with COVID-19 must be treated with efficacious medications; they deserve nothing less.”
The study was supported by the Parsemus Foundation, Rainwater Charitable Foundation, Fast Grants, and UnitedHealth Group Foundation. The fluvoxamine placebo tablets were donated by Apotex Pharmaceuticals. The ivermectin placebo and active tablets were donated by Edenbridge Pharmaceuticals. Lead author Dr. Bramante was supported the National Center for Advancing Translational Sciences and the National Institute of Diabetes and Digestive and Kidney Diseases. The researchers had no financial conflicts to disclose. Dr. Abdool Karim serves as a member of the World Health Organization Science Council. Dr. Devnarain had no financial conflicts to disclose.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
‘Conservative’ USPSTF primary prevention statin guidance finalized
Questions about how to prescribe statins for primary prevention abound more than 3 decades after the drugs swept into clinical practice to become a first-line medical approach to cutting cardiovascular (CV) risk. Statin usage recommendations from different bodies can vary in ways both limited and fundamental, spurring the kind of debate that accompanies such a document newly issued by the United States Preventive Services Task Force.
The document, little changed from the draft guidance released for public comment in February, was published online Aug. 23 in JAMA and the USPSTF website. It replaces a similar document issued by the task force in 2016.
The guidance has much in common with, but also sharp differences from, the influential 2018 guidelines on blood cholesterol management developed by the American College of Cardiology, American Heart Association, and 10 other medical societies.
And it is provocative enough to elicit at least four editorials issued the same day across the JAMA family of journals. They highlight key differences between the two documents, among them the USPSTF guidance’s consistent, narrow reliance on 7.5% and 10% cut points for 10-year risk levels as estimated from the ACC/AHA pooled cohort equations (PCE).
The guidance pairs the 10-year risk metric with at least one of only four prescribed CV risk factors to arrive at a limited choice of statin therapy recommendations. But its decision process isn’t bolstered by coronary artery calcium (CAC) scores or the prespecified “risk enhancers” that allowed the ACC/AHA-multisociety guidelines to be applied broadly and still be closely personalized. Those guidelines provide more PCE-based risk tiers for greater discrimination of risk and allow statins to be considered across a broader age group.
The USPSTF guidance’s evidence base consists of 23 clinical trials and three observational studies that directly compared a statin to either placebo or no statin, task force member John B. Wong, MD, Tufts University School of Medicine, Boston, told this news organization.
“In either kind of study, we found that the vast majority of patients had one or more of four risk factors – dyslipidemia, hypertension, diabetes, or smoking. So, when we categorized high risk or increased risk, we included the presence of one or more of those risk factors,” said Dr. Wong, who is director of comparative effectiveness research at Tufts Clinical Translational Science Institute.
‘Sensible and practical’
The USPSTF guidance applies only to adults aged 40-75 without CV signs or symptoms and recommends a statin prescription for persons at “high risk,” that is with an estimated 10-year PCE-based risk for death or CV events of 10% or higher plus at least one of the four risk factors, a level B recommendation.
It recommends that “clinicians selectively offer a statin” to such persons at “increased risk,” who have at least one of the risk factors and an estimated 10-year risk for death or CV events of 7.5% to less than 10%, a level C recommendation. “The likelihood of benefit is smaller in this group” than in persons at high risk, the document states.
“These recommendations from the USPSTF are sensible and practical,” states Salim S. Virani, MD, PhD, DeBakey Veterans Affairs Medical Center, Houston, in a related editorial published the same day in JAMA Network Open. He calls the former B-level recommendation “a conservative approach” and the latter C-level recommendation a “nuanced approach.”
Both are “understandable” given that some studies suggest that the PCE may overestimate the CV risk, Dr. Virani observes. “On the other hand, statin therapy has been shown to be efficacious” at 10-year CV-risk levels down to about 5%.
The USPSTF document “I think is going to perpetuate a problem that we have in this country, which is vast undertreatment of lipids,” Eric D. Peterson, MD, MPH, University of Texas Southwestern Medical Center, Dallas, said in an interview.
“We have a ton of good drugs that can lower cholesterol like crazy. If you lower cholesterol a lot, you improve outcomes,” he said. Dyslipidemia needs to be more widely and consistently treated, but “right now we have a pool of people in primary prevention who undertreat lipids and wait until disease happens – and then cardiologists get engaged. That’s an avoidable miss,” Dr. Peterson adds. He and JAMA Cardiology associate editor Ann Marie Navar, MD, PhD, provided JAMA with an editorial that accompanies the USPSTF guidance.
“My own personal bias would be that the [ACC/AHA-multisociety guidelines] are closer to being right,” Dr. Peterson said. They – unlike the USPSTF guidance – cover people with risk levels below 7.5%, down to at least 5%. They allow risk enhancers like metabolic syndrome, inflammatory diseases, or family history into the decision process. “And they’re more aggressive in diabetes and more aggressive in older people,” he said.
Higher threshold for therapy
The USPSTF guidance also explicitly omits some high-risk groups and makes little accommodation for others who might especially benefit from statins, several of the editorials contend. For example, states a related JAMA Cardiology editorial published the same day, “The USPSTF does not comment on familial hypercholesterolemia or an LDL-C level of 190 mg/dL or higher,” yet they are covered by the ACC/AHA-multispecialty guidelines.
In addition, write the editorialists, led by Neil J. Stone, MD, Northwestern University, Chicago, “the USPSTF uses a slightly higher threshold for initiation of statin therapy” than was used in the ACC/AHA-multisociety guidelines. USPSTF, for example, calls for 10-year risk to reach 10% before recommending a statin prescription.
“One concern about the USPSTF setting the bar higher for statin initiation is that it reduces the number of young patients (age 40-50 years) at risk for premature myocardial infarction considered for treatment,” write Dr. Stone and colleagues.
That may be related to a weakness of the PCE-based decision process. “Because the PCE estimates of 10-year CV disease risk rely so heavily on age, sex, and race, use of these estimates to identify candidates for statins results in significant skewing of the population recommended for statins,” write Dr. Navar and Dr. Peterson in their JAMA editorial.
The risk enhancers in the ACC/AHA-multispecialty guidelines, about a dozen of them, compensate for that limitation to some extent. But the PCE-dominated USPSTF risk estimates will likely miss some groups that could potentially benefit from statin therapy, Dr. Peterson agreed in an interview.
For example, younger adults facing years of high LDL-cholesterol levels could easily have PCE-based 10-year risk below 10%. “Having a high LDL over a lifetime puts you at really high risk,” he said. “Young people are missed even though their longitudinal risk is high.” So, by waiting for the lofty 10% level of risk over 10 years, “we limit the use of medicine that’s pretty cheap and highly effective.”
Dose intensity, adverse events
Also at variance from the ACC/AHA-multispecialty guidelines, the USPSTF states that, “Based on available evidence, use of moderate-intensity statin therapy seems reasonable for the primary prevention of CV disease in most persons.”
The task force specifically explored whether evidence supports some use of high-intensity vs. moderate-intensity statins, Tufts University’s Dr. Wong said. “We found only one study that looked at that particular question, and it didn’t give us a strong answer.” An elevated rosuvastatin-related diabetes risk was apparent in the JUPITER trial, “but for the other studies, we did not find that association.”
Most of the studies that explored statins for reducing risk for a first stroke or myocardial infarction used a moderate-dose statin, Dr. Wong said. “So that’s what we would usually recommend.”
But, Dr. Virani writes, consistent with the ACC/AHA-multispecialty guidelines, “clinicians should consider titrating the intensity of therapy to the risk of the individual.” Persons in certain high-risk primary prevention groups, such as those with end-organ injury from diabetes or LDL cholesterol at least 190 mg/dL, “may derive further benefit from the use of high-intensity statin therapy.”
Low-intensity statins are another potential option, but “in contrast with its 2016 recommendations, the USPSTF no longer recommends use of low-intensity statins in certain situations,” observes a fourth editorial published the same day in JAMA Internal Medicine, with lead author Anand R. Habib, MD, MPhil, and senior author Rita F. Redberg, MD, MSc, both of the University of California, San Francisco. Dr. Redberg is the journal’s editor and has long expressed cautions about statin safety.
“While it is understandable that the Task Force was limited by lack of data on dosing, this change is unfortunate for patients because the frequency of adverse effects increases as the statin dose increases,” the editorial states. Although USPSTF did not find statistically significant harm from the drugs, “in clinical practice, adverse events are commonly reported with use of statins.”
It continues: “At present, there are further reasons to curb our enthusiasm about the use of statins for primary prevention of CV disease.” To illustrate, the editorial questioned primary-prevention statins’ balance of risk vs. clinically meaningful benefit, not benefit that is merely statistically significant.
“The purported benefits of statins in terms of relative risk reduction are fairly constant across baseline lipid levels and cardiovascular risk score categories for primary prevention,” the editorial states.
“Therefore, the absolute benefit for those in lower-risk categories is likely small given that their baseline absolute risk is low, while the chance of adverse effects is constant across risk categories.”
However, USPSTF states, “In pooled analyses of trial data, statin therapy was not associated with increased risk of study withdrawal due to adverse events or serious adverse events.” Nor did it find significant associations with cancers, liver enzyme abnormalities, or diabetes, including new-onset diabetes.
And, the USPSTF adds, “Evidence on the association between statins and renal or cognitive harms is very limited but does not indicate increased risk.”
USPSTF is supported by the U.S. Agency for Healthcare Research and Quality. Dr. Virani discloses receiving grants from the Department of Veterans Affairs, National Institutes of Health, and the World Heart Federation; and personal fees from the American College of Cardiology. Dr. Peterson discloses serving on the JAMA editorial board and receiving research support to his institution from Amgen, Bristol-Myers Squibb, Esperion, and Janssen; and consulting fees from Novo Nordisk, Bayer, and Novartis. Dr. Navar discloses receiving research support to her institution from Amgen, Bristol-Myers Squibb, Esperion, and Janssen; and receiving honoraria and consulting fees from AstraZeneca, Boehringer Ingelheim, Bayer, Janssen, Lilly, Novo Nordisk, Novartis, New Amsterdam, and Pfizer. Dr. Stone discloses receiving an honorarium from Knowledge to Practice, an educational company not associated with the pharmaceutical industry; disclosures for the other authors are in the report. Dr. Redberg discloses receiving research funding from the Arnold Ventures Foundation and the Greenwall Foundation.
A version of this article first appeared on Medscape.com.
Questions about how to prescribe statins for primary prevention abound more than 3 decades after the drugs swept into clinical practice to become a first-line medical approach to cutting cardiovascular (CV) risk. Statin usage recommendations from different bodies can vary in ways both limited and fundamental, spurring the kind of debate that accompanies such a document newly issued by the United States Preventive Services Task Force.
The document, little changed from the draft guidance released for public comment in February, was published online Aug. 23 in JAMA and the USPSTF website. It replaces a similar document issued by the task force in 2016.
The guidance has much in common with, but also sharp differences from, the influential 2018 guidelines on blood cholesterol management developed by the American College of Cardiology, American Heart Association, and 10 other medical societies.
And it is provocative enough to elicit at least four editorials issued the same day across the JAMA family of journals. They highlight key differences between the two documents, among them the USPSTF guidance’s consistent, narrow reliance on 7.5% and 10% cut points for 10-year risk levels as estimated from the ACC/AHA pooled cohort equations (PCE).
The guidance pairs the 10-year risk metric with at least one of only four prescribed CV risk factors to arrive at a limited choice of statin therapy recommendations. But its decision process isn’t bolstered by coronary artery calcium (CAC) scores or the prespecified “risk enhancers” that allowed the ACC/AHA-multisociety guidelines to be applied broadly and still be closely personalized. Those guidelines provide more PCE-based risk tiers for greater discrimination of risk and allow statins to be considered across a broader age group.
The USPSTF guidance’s evidence base consists of 23 clinical trials and three observational studies that directly compared a statin to either placebo or no statin, task force member John B. Wong, MD, Tufts University School of Medicine, Boston, told this news organization.
“In either kind of study, we found that the vast majority of patients had one or more of four risk factors – dyslipidemia, hypertension, diabetes, or smoking. So, when we categorized high risk or increased risk, we included the presence of one or more of those risk factors,” said Dr. Wong, who is director of comparative effectiveness research at Tufts Clinical Translational Science Institute.
‘Sensible and practical’
The USPSTF guidance applies only to adults aged 40-75 without CV signs or symptoms and recommends a statin prescription for persons at “high risk,” that is with an estimated 10-year PCE-based risk for death or CV events of 10% or higher plus at least one of the four risk factors, a level B recommendation.
It recommends that “clinicians selectively offer a statin” to such persons at “increased risk,” who have at least one of the risk factors and an estimated 10-year risk for death or CV events of 7.5% to less than 10%, a level C recommendation. “The likelihood of benefit is smaller in this group” than in persons at high risk, the document states.
“These recommendations from the USPSTF are sensible and practical,” states Salim S. Virani, MD, PhD, DeBakey Veterans Affairs Medical Center, Houston, in a related editorial published the same day in JAMA Network Open. He calls the former B-level recommendation “a conservative approach” and the latter C-level recommendation a “nuanced approach.”
Both are “understandable” given that some studies suggest that the PCE may overestimate the CV risk, Dr. Virani observes. “On the other hand, statin therapy has been shown to be efficacious” at 10-year CV-risk levels down to about 5%.
The USPSTF document “I think is going to perpetuate a problem that we have in this country, which is vast undertreatment of lipids,” Eric D. Peterson, MD, MPH, University of Texas Southwestern Medical Center, Dallas, said in an interview.
“We have a ton of good drugs that can lower cholesterol like crazy. If you lower cholesterol a lot, you improve outcomes,” he said. Dyslipidemia needs to be more widely and consistently treated, but “right now we have a pool of people in primary prevention who undertreat lipids and wait until disease happens – and then cardiologists get engaged. That’s an avoidable miss,” Dr. Peterson adds. He and JAMA Cardiology associate editor Ann Marie Navar, MD, PhD, provided JAMA with an editorial that accompanies the USPSTF guidance.
“My own personal bias would be that the [ACC/AHA-multisociety guidelines] are closer to being right,” Dr. Peterson said. They – unlike the USPSTF guidance – cover people with risk levels below 7.5%, down to at least 5%. They allow risk enhancers like metabolic syndrome, inflammatory diseases, or family history into the decision process. “And they’re more aggressive in diabetes and more aggressive in older people,” he said.
Higher threshold for therapy
The USPSTF guidance also explicitly omits some high-risk groups and makes little accommodation for others who might especially benefit from statins, several of the editorials contend. For example, states a related JAMA Cardiology editorial published the same day, “The USPSTF does not comment on familial hypercholesterolemia or an LDL-C level of 190 mg/dL or higher,” yet they are covered by the ACC/AHA-multispecialty guidelines.
In addition, write the editorialists, led by Neil J. Stone, MD, Northwestern University, Chicago, “the USPSTF uses a slightly higher threshold for initiation of statin therapy” than was used in the ACC/AHA-multisociety guidelines. USPSTF, for example, calls for 10-year risk to reach 10% before recommending a statin prescription.
“One concern about the USPSTF setting the bar higher for statin initiation is that it reduces the number of young patients (age 40-50 years) at risk for premature myocardial infarction considered for treatment,” write Dr. Stone and colleagues.
That may be related to a weakness of the PCE-based decision process. “Because the PCE estimates of 10-year CV disease risk rely so heavily on age, sex, and race, use of these estimates to identify candidates for statins results in significant skewing of the population recommended for statins,” write Dr. Navar and Dr. Peterson in their JAMA editorial.
The risk enhancers in the ACC/AHA-multispecialty guidelines, about a dozen of them, compensate for that limitation to some extent. But the PCE-dominated USPSTF risk estimates will likely miss some groups that could potentially benefit from statin therapy, Dr. Peterson agreed in an interview.
For example, younger adults facing years of high LDL-cholesterol levels could easily have PCE-based 10-year risk below 10%. “Having a high LDL over a lifetime puts you at really high risk,” he said. “Young people are missed even though their longitudinal risk is high.” So, by waiting for the lofty 10% level of risk over 10 years, “we limit the use of medicine that’s pretty cheap and highly effective.”
Dose intensity, adverse events
Also at variance from the ACC/AHA-multispecialty guidelines, the USPSTF states that, “Based on available evidence, use of moderate-intensity statin therapy seems reasonable for the primary prevention of CV disease in most persons.”
The task force specifically explored whether evidence supports some use of high-intensity vs. moderate-intensity statins, Tufts University’s Dr. Wong said. “We found only one study that looked at that particular question, and it didn’t give us a strong answer.” An elevated rosuvastatin-related diabetes risk was apparent in the JUPITER trial, “but for the other studies, we did not find that association.”
Most of the studies that explored statins for reducing risk for a first stroke or myocardial infarction used a moderate-dose statin, Dr. Wong said. “So that’s what we would usually recommend.”
But, Dr. Virani writes, consistent with the ACC/AHA-multispecialty guidelines, “clinicians should consider titrating the intensity of therapy to the risk of the individual.” Persons in certain high-risk primary prevention groups, such as those with end-organ injury from diabetes or LDL cholesterol at least 190 mg/dL, “may derive further benefit from the use of high-intensity statin therapy.”
Low-intensity statins are another potential option, but “in contrast with its 2016 recommendations, the USPSTF no longer recommends use of low-intensity statins in certain situations,” observes a fourth editorial published the same day in JAMA Internal Medicine, with lead author Anand R. Habib, MD, MPhil, and senior author Rita F. Redberg, MD, MSc, both of the University of California, San Francisco. Dr. Redberg is the journal’s editor and has long expressed cautions about statin safety.
“While it is understandable that the Task Force was limited by lack of data on dosing, this change is unfortunate for patients because the frequency of adverse effects increases as the statin dose increases,” the editorial states. Although USPSTF did not find statistically significant harm from the drugs, “in clinical practice, adverse events are commonly reported with use of statins.”
It continues: “At present, there are further reasons to curb our enthusiasm about the use of statins for primary prevention of CV disease.” To illustrate, the editorial questioned primary-prevention statins’ balance of risk vs. clinically meaningful benefit, not benefit that is merely statistically significant.
“The purported benefits of statins in terms of relative risk reduction are fairly constant across baseline lipid levels and cardiovascular risk score categories for primary prevention,” the editorial states.
“Therefore, the absolute benefit for those in lower-risk categories is likely small given that their baseline absolute risk is low, while the chance of adverse effects is constant across risk categories.”
However, USPSTF states, “In pooled analyses of trial data, statin therapy was not associated with increased risk of study withdrawal due to adverse events or serious adverse events.” Nor did it find significant associations with cancers, liver enzyme abnormalities, or diabetes, including new-onset diabetes.
And, the USPSTF adds, “Evidence on the association between statins and renal or cognitive harms is very limited but does not indicate increased risk.”
USPSTF is supported by the U.S. Agency for Healthcare Research and Quality. Dr. Virani discloses receiving grants from the Department of Veterans Affairs, National Institutes of Health, and the World Heart Federation; and personal fees from the American College of Cardiology. Dr. Peterson discloses serving on the JAMA editorial board and receiving research support to his institution from Amgen, Bristol-Myers Squibb, Esperion, and Janssen; and consulting fees from Novo Nordisk, Bayer, and Novartis. Dr. Navar discloses receiving research support to her institution from Amgen, Bristol-Myers Squibb, Esperion, and Janssen; and receiving honoraria and consulting fees from AstraZeneca, Boehringer Ingelheim, Bayer, Janssen, Lilly, Novo Nordisk, Novartis, New Amsterdam, and Pfizer. Dr. Stone discloses receiving an honorarium from Knowledge to Practice, an educational company not associated with the pharmaceutical industry; disclosures for the other authors are in the report. Dr. Redberg discloses receiving research funding from the Arnold Ventures Foundation and the Greenwall Foundation.
A version of this article first appeared on Medscape.com.
Questions about how to prescribe statins for primary prevention abound more than 3 decades after the drugs swept into clinical practice to become a first-line medical approach to cutting cardiovascular (CV) risk. Statin usage recommendations from different bodies can vary in ways both limited and fundamental, spurring the kind of debate that accompanies such a document newly issued by the United States Preventive Services Task Force.
The document, little changed from the draft guidance released for public comment in February, was published online Aug. 23 in JAMA and the USPSTF website. It replaces a similar document issued by the task force in 2016.
The guidance has much in common with, but also sharp differences from, the influential 2018 guidelines on blood cholesterol management developed by the American College of Cardiology, American Heart Association, and 10 other medical societies.
And it is provocative enough to elicit at least four editorials issued the same day across the JAMA family of journals. They highlight key differences between the two documents, among them the USPSTF guidance’s consistent, narrow reliance on 7.5% and 10% cut points for 10-year risk levels as estimated from the ACC/AHA pooled cohort equations (PCE).
The guidance pairs the 10-year risk metric with at least one of only four prescribed CV risk factors to arrive at a limited choice of statin therapy recommendations. But its decision process isn’t bolstered by coronary artery calcium (CAC) scores or the prespecified “risk enhancers” that allowed the ACC/AHA-multisociety guidelines to be applied broadly and still be closely personalized. Those guidelines provide more PCE-based risk tiers for greater discrimination of risk and allow statins to be considered across a broader age group.
The USPSTF guidance’s evidence base consists of 23 clinical trials and three observational studies that directly compared a statin to either placebo or no statin, task force member John B. Wong, MD, Tufts University School of Medicine, Boston, told this news organization.
“In either kind of study, we found that the vast majority of patients had one or more of four risk factors – dyslipidemia, hypertension, diabetes, or smoking. So, when we categorized high risk or increased risk, we included the presence of one or more of those risk factors,” said Dr. Wong, who is director of comparative effectiveness research at Tufts Clinical Translational Science Institute.
‘Sensible and practical’
The USPSTF guidance applies only to adults aged 40-75 without CV signs or symptoms and recommends a statin prescription for persons at “high risk,” that is with an estimated 10-year PCE-based risk for death or CV events of 10% or higher plus at least one of the four risk factors, a level B recommendation.
It recommends that “clinicians selectively offer a statin” to such persons at “increased risk,” who have at least one of the risk factors and an estimated 10-year risk for death or CV events of 7.5% to less than 10%, a level C recommendation. “The likelihood of benefit is smaller in this group” than in persons at high risk, the document states.
“These recommendations from the USPSTF are sensible and practical,” states Salim S. Virani, MD, PhD, DeBakey Veterans Affairs Medical Center, Houston, in a related editorial published the same day in JAMA Network Open. He calls the former B-level recommendation “a conservative approach” and the latter C-level recommendation a “nuanced approach.”
Both are “understandable” given that some studies suggest that the PCE may overestimate the CV risk, Dr. Virani observes. “On the other hand, statin therapy has been shown to be efficacious” at 10-year CV-risk levels down to about 5%.
The USPSTF document “I think is going to perpetuate a problem that we have in this country, which is vast undertreatment of lipids,” Eric D. Peterson, MD, MPH, University of Texas Southwestern Medical Center, Dallas, said in an interview.
“We have a ton of good drugs that can lower cholesterol like crazy. If you lower cholesterol a lot, you improve outcomes,” he said. Dyslipidemia needs to be more widely and consistently treated, but “right now we have a pool of people in primary prevention who undertreat lipids and wait until disease happens – and then cardiologists get engaged. That’s an avoidable miss,” Dr. Peterson adds. He and JAMA Cardiology associate editor Ann Marie Navar, MD, PhD, provided JAMA with an editorial that accompanies the USPSTF guidance.
“My own personal bias would be that the [ACC/AHA-multisociety guidelines] are closer to being right,” Dr. Peterson said. They – unlike the USPSTF guidance – cover people with risk levels below 7.5%, down to at least 5%. They allow risk enhancers like metabolic syndrome, inflammatory diseases, or family history into the decision process. “And they’re more aggressive in diabetes and more aggressive in older people,” he said.
Higher threshold for therapy
The USPSTF guidance also explicitly omits some high-risk groups and makes little accommodation for others who might especially benefit from statins, several of the editorials contend. For example, states a related JAMA Cardiology editorial published the same day, “The USPSTF does not comment on familial hypercholesterolemia or an LDL-C level of 190 mg/dL or higher,” yet they are covered by the ACC/AHA-multispecialty guidelines.
In addition, write the editorialists, led by Neil J. Stone, MD, Northwestern University, Chicago, “the USPSTF uses a slightly higher threshold for initiation of statin therapy” than was used in the ACC/AHA-multisociety guidelines. USPSTF, for example, calls for 10-year risk to reach 10% before recommending a statin prescription.
“One concern about the USPSTF setting the bar higher for statin initiation is that it reduces the number of young patients (age 40-50 years) at risk for premature myocardial infarction considered for treatment,” write Dr. Stone and colleagues.
That may be related to a weakness of the PCE-based decision process. “Because the PCE estimates of 10-year CV disease risk rely so heavily on age, sex, and race, use of these estimates to identify candidates for statins results in significant skewing of the population recommended for statins,” write Dr. Navar and Dr. Peterson in their JAMA editorial.
The risk enhancers in the ACC/AHA-multispecialty guidelines, about a dozen of them, compensate for that limitation to some extent. But the PCE-dominated USPSTF risk estimates will likely miss some groups that could potentially benefit from statin therapy, Dr. Peterson agreed in an interview.
For example, younger adults facing years of high LDL-cholesterol levels could easily have PCE-based 10-year risk below 10%. “Having a high LDL over a lifetime puts you at really high risk,” he said. “Young people are missed even though their longitudinal risk is high.” So, by waiting for the lofty 10% level of risk over 10 years, “we limit the use of medicine that’s pretty cheap and highly effective.”
Dose intensity, adverse events
Also at variance from the ACC/AHA-multispecialty guidelines, the USPSTF states that, “Based on available evidence, use of moderate-intensity statin therapy seems reasonable for the primary prevention of CV disease in most persons.”
The task force specifically explored whether evidence supports some use of high-intensity vs. moderate-intensity statins, Tufts University’s Dr. Wong said. “We found only one study that looked at that particular question, and it didn’t give us a strong answer.” An elevated rosuvastatin-related diabetes risk was apparent in the JUPITER trial, “but for the other studies, we did not find that association.”
Most of the studies that explored statins for reducing risk for a first stroke or myocardial infarction used a moderate-dose statin, Dr. Wong said. “So that’s what we would usually recommend.”
But, Dr. Virani writes, consistent with the ACC/AHA-multispecialty guidelines, “clinicians should consider titrating the intensity of therapy to the risk of the individual.” Persons in certain high-risk primary prevention groups, such as those with end-organ injury from diabetes or LDL cholesterol at least 190 mg/dL, “may derive further benefit from the use of high-intensity statin therapy.”
Low-intensity statins are another potential option, but “in contrast with its 2016 recommendations, the USPSTF no longer recommends use of low-intensity statins in certain situations,” observes a fourth editorial published the same day in JAMA Internal Medicine, with lead author Anand R. Habib, MD, MPhil, and senior author Rita F. Redberg, MD, MSc, both of the University of California, San Francisco. Dr. Redberg is the journal’s editor and has long expressed cautions about statin safety.
“While it is understandable that the Task Force was limited by lack of data on dosing, this change is unfortunate for patients because the frequency of adverse effects increases as the statin dose increases,” the editorial states. Although USPSTF did not find statistically significant harm from the drugs, “in clinical practice, adverse events are commonly reported with use of statins.”
It continues: “At present, there are further reasons to curb our enthusiasm about the use of statins for primary prevention of CV disease.” To illustrate, the editorial questioned primary-prevention statins’ balance of risk vs. clinically meaningful benefit, not benefit that is merely statistically significant.
“The purported benefits of statins in terms of relative risk reduction are fairly constant across baseline lipid levels and cardiovascular risk score categories for primary prevention,” the editorial states.
“Therefore, the absolute benefit for those in lower-risk categories is likely small given that their baseline absolute risk is low, while the chance of adverse effects is constant across risk categories.”
However, USPSTF states, “In pooled analyses of trial data, statin therapy was not associated with increased risk of study withdrawal due to adverse events or serious adverse events.” Nor did it find significant associations with cancers, liver enzyme abnormalities, or diabetes, including new-onset diabetes.
And, the USPSTF adds, “Evidence on the association between statins and renal or cognitive harms is very limited but does not indicate increased risk.”
USPSTF is supported by the U.S. Agency for Healthcare Research and Quality. Dr. Virani discloses receiving grants from the Department of Veterans Affairs, National Institutes of Health, and the World Heart Federation; and personal fees from the American College of Cardiology. Dr. Peterson discloses serving on the JAMA editorial board and receiving research support to his institution from Amgen, Bristol-Myers Squibb, Esperion, and Janssen; and consulting fees from Novo Nordisk, Bayer, and Novartis. Dr. Navar discloses receiving research support to her institution from Amgen, Bristol-Myers Squibb, Esperion, and Janssen; and receiving honoraria and consulting fees from AstraZeneca, Boehringer Ingelheim, Bayer, Janssen, Lilly, Novo Nordisk, Novartis, New Amsterdam, and Pfizer. Dr. Stone discloses receiving an honorarium from Knowledge to Practice, an educational company not associated with the pharmaceutical industry; disclosures for the other authors are in the report. Dr. Redberg discloses receiving research funding from the Arnold Ventures Foundation and the Greenwall Foundation.
A version of this article first appeared on Medscape.com.
FROM JAMA
Are artificial sweeteners really harmless?
New research discounts the long-held notion that aspartame and other nonnutritive sweeteners (NNS) have no effect on the human body.
Researchers found that these sugar substitutes are not metabolically inert and can alter the gut microbiome in a way that can influence blood glucose levels.
The study was published online in the journal Cell.
Gut reaction?
Several years ago, a team led by Eran Elinav, MD, PhD, an immunologist and microbiome researcher at the Weizmann Institute of Science, Rehovot, Israel, observed that these sweeteners affect the microbiome of mice in ways that could affect glycemic responses.
They have now confirmed this observation in a randomized controlled trial with 120 healthy adults.
Each sweetener “significantly and distinctly” altered stool and oral microbiome, and two of them (saccharin and sucralose) significantly impaired glucose tolerance, the researchers reported.
“Importantly, by performing extensive fecal transplantation of human microbiomes into germ-free mice, we demonstrate a causal and individualized link between NNS-altered microbiomes and glucose intolerance developing in non–NNS-consuming recipient mice,” they said.
They noted that the effects of these sweeteners will likely vary from person to person because of the unique composition of an individual’s microbiome.
“We need to raise awareness of the fact that NNS are not inert to the human body as we originally believed. With that said, the clinical health implications of the changes they may elicit in humans remain unknown and merit future long-term studies,” Dr. Elinav said in a news release.
For now, Dr. Elinav said it’s his personal view that “drinking only water seems to be the best solution.”
Weighing the evidence
Several experts weighed in on the results in a statement from the U.K. nonprofit organization, Science Media Centre.
Duane Mellor, PhD, RD, RNutr, registered dietitian and senior teaching fellow, Aston University, Birmingham, England, notes that the study does not show a link between all NNS and higher blood glucose levels in the long term (only after a glucose tolerance test).
“It did suggest, though, that some individuals who do not normally consume sweeteners may not tolerate glucose as well after consuming six sachets of either saccharin or sucralose mixed with glucose per day,” Dr. Mellor says.
Kim Barrett, PhD, distinguished professor of physiology and membrane biology, University of California, Davis, concurs, saying “this well-designed study indicates the potential for NNS to have adverse effects in at least some individuals.”
The study also does not provide any information about how people who normally consume sweeteners or people with either type 1 or type 2 diabetes respond to NNS.
“Therefore, for some people, it is likely to be a better option and more sustainable approach to use sweeteners as a ‘stepping stone’ allowing them to reduce the amount of added sugar in foods and drinks, to reduce their sugar intake and still enjoy what they eat and drink, on the way to reducing both added sugar and sweeteners in their diet,” Dr. Mellor suggests.
Kevin McConway, PhD, with the Open University, Milton Keynes, England, said it’s “important to understand that the research is not saying that these sweeteners are worse for us, in heath terms, than sugar.
“But exactly what the health consequences of all this, if any, might be is a subject for future research,” Dr. McConway added.
Kathy Redfern, PhD, lecturer in human nutrition, University of Plymouth (England) agrees.
“We still have a lot to learn about the human microbiome, and although this study suggests two of the sweeteners tested in this study (sucralose and saccharin) significantly affected glucose tolerance, these deviations were small,” she says.
The International Sweeteners Association also weighs in, saying, “No conclusions about the effects of low/no calorie sweeteners on glucose control or overall health can be extrapolated from this study for the general population or for people who typically consume sweeteners, including people living with diabetes.”
They add “a recent review of the literature concluded that there is clear evidence that changes in the diet unrelated to low/no calorie sweeteners consumption are likely the major determinants of change in gut microbiota.”
Nevertheless, Dr. Redfern says the results “warrant further investigation to assess how small changes in glucose tolerance in response to NNS consumption may influence longer-term glucose tolerance and risk for metabolic complications, such as type 2 diabetes.”
The study had no specific funding. Dr. Elinav is a scientific founder of DayTwo and BiomX, a paid consultant to Hello Inside and Aposense, and a member of the scientific advisory board of Cell. Dr. Mellor has provided consultancy to the International Sweetener Agency and has worked on projects funded by the Food Standards Agency that investigated the health effects of aspartame. Dr. Barrett, Dr. McConway, and Dr. Redfern report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This article was updated 8/29/22.
New research discounts the long-held notion that aspartame and other nonnutritive sweeteners (NNS) have no effect on the human body.
Researchers found that these sugar substitutes are not metabolically inert and can alter the gut microbiome in a way that can influence blood glucose levels.
The study was published online in the journal Cell.
Gut reaction?
Several years ago, a team led by Eran Elinav, MD, PhD, an immunologist and microbiome researcher at the Weizmann Institute of Science, Rehovot, Israel, observed that these sweeteners affect the microbiome of mice in ways that could affect glycemic responses.
They have now confirmed this observation in a randomized controlled trial with 120 healthy adults.
Each sweetener “significantly and distinctly” altered stool and oral microbiome, and two of them (saccharin and sucralose) significantly impaired glucose tolerance, the researchers reported.
“Importantly, by performing extensive fecal transplantation of human microbiomes into germ-free mice, we demonstrate a causal and individualized link between NNS-altered microbiomes and glucose intolerance developing in non–NNS-consuming recipient mice,” they said.
They noted that the effects of these sweeteners will likely vary from person to person because of the unique composition of an individual’s microbiome.
“We need to raise awareness of the fact that NNS are not inert to the human body as we originally believed. With that said, the clinical health implications of the changes they may elicit in humans remain unknown and merit future long-term studies,” Dr. Elinav said in a news release.
For now, Dr. Elinav said it’s his personal view that “drinking only water seems to be the best solution.”
Weighing the evidence
Several experts weighed in on the results in a statement from the U.K. nonprofit organization, Science Media Centre.
Duane Mellor, PhD, RD, RNutr, registered dietitian and senior teaching fellow, Aston University, Birmingham, England, notes that the study does not show a link between all NNS and higher blood glucose levels in the long term (only after a glucose tolerance test).
“It did suggest, though, that some individuals who do not normally consume sweeteners may not tolerate glucose as well after consuming six sachets of either saccharin or sucralose mixed with glucose per day,” Dr. Mellor says.
Kim Barrett, PhD, distinguished professor of physiology and membrane biology, University of California, Davis, concurs, saying “this well-designed study indicates the potential for NNS to have adverse effects in at least some individuals.”
The study also does not provide any information about how people who normally consume sweeteners or people with either type 1 or type 2 diabetes respond to NNS.
“Therefore, for some people, it is likely to be a better option and more sustainable approach to use sweeteners as a ‘stepping stone’ allowing them to reduce the amount of added sugar in foods and drinks, to reduce their sugar intake and still enjoy what they eat and drink, on the way to reducing both added sugar and sweeteners in their diet,” Dr. Mellor suggests.
Kevin McConway, PhD, with the Open University, Milton Keynes, England, said it’s “important to understand that the research is not saying that these sweeteners are worse for us, in heath terms, than sugar.
“But exactly what the health consequences of all this, if any, might be is a subject for future research,” Dr. McConway added.
Kathy Redfern, PhD, lecturer in human nutrition, University of Plymouth (England) agrees.
“We still have a lot to learn about the human microbiome, and although this study suggests two of the sweeteners tested in this study (sucralose and saccharin) significantly affected glucose tolerance, these deviations were small,” she says.
The International Sweeteners Association also weighs in, saying, “No conclusions about the effects of low/no calorie sweeteners on glucose control or overall health can be extrapolated from this study for the general population or for people who typically consume sweeteners, including people living with diabetes.”
They add “a recent review of the literature concluded that there is clear evidence that changes in the diet unrelated to low/no calorie sweeteners consumption are likely the major determinants of change in gut microbiota.”
Nevertheless, Dr. Redfern says the results “warrant further investigation to assess how small changes in glucose tolerance in response to NNS consumption may influence longer-term glucose tolerance and risk for metabolic complications, such as type 2 diabetes.”
The study had no specific funding. Dr. Elinav is a scientific founder of DayTwo and BiomX, a paid consultant to Hello Inside and Aposense, and a member of the scientific advisory board of Cell. Dr. Mellor has provided consultancy to the International Sweetener Agency and has worked on projects funded by the Food Standards Agency that investigated the health effects of aspartame. Dr. Barrett, Dr. McConway, and Dr. Redfern report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This article was updated 8/29/22.
New research discounts the long-held notion that aspartame and other nonnutritive sweeteners (NNS) have no effect on the human body.
Researchers found that these sugar substitutes are not metabolically inert and can alter the gut microbiome in a way that can influence blood glucose levels.
The study was published online in the journal Cell.
Gut reaction?
Several years ago, a team led by Eran Elinav, MD, PhD, an immunologist and microbiome researcher at the Weizmann Institute of Science, Rehovot, Israel, observed that these sweeteners affect the microbiome of mice in ways that could affect glycemic responses.
They have now confirmed this observation in a randomized controlled trial with 120 healthy adults.
Each sweetener “significantly and distinctly” altered stool and oral microbiome, and two of them (saccharin and sucralose) significantly impaired glucose tolerance, the researchers reported.
“Importantly, by performing extensive fecal transplantation of human microbiomes into germ-free mice, we demonstrate a causal and individualized link between NNS-altered microbiomes and glucose intolerance developing in non–NNS-consuming recipient mice,” they said.
They noted that the effects of these sweeteners will likely vary from person to person because of the unique composition of an individual’s microbiome.
“We need to raise awareness of the fact that NNS are not inert to the human body as we originally believed. With that said, the clinical health implications of the changes they may elicit in humans remain unknown and merit future long-term studies,” Dr. Elinav said in a news release.
For now, Dr. Elinav said it’s his personal view that “drinking only water seems to be the best solution.”
Weighing the evidence
Several experts weighed in on the results in a statement from the U.K. nonprofit organization, Science Media Centre.
Duane Mellor, PhD, RD, RNutr, registered dietitian and senior teaching fellow, Aston University, Birmingham, England, notes that the study does not show a link between all NNS and higher blood glucose levels in the long term (only after a glucose tolerance test).
“It did suggest, though, that some individuals who do not normally consume sweeteners may not tolerate glucose as well after consuming six sachets of either saccharin or sucralose mixed with glucose per day,” Dr. Mellor says.
Kim Barrett, PhD, distinguished professor of physiology and membrane biology, University of California, Davis, concurs, saying “this well-designed study indicates the potential for NNS to have adverse effects in at least some individuals.”
The study also does not provide any information about how people who normally consume sweeteners or people with either type 1 or type 2 diabetes respond to NNS.
“Therefore, for some people, it is likely to be a better option and more sustainable approach to use sweeteners as a ‘stepping stone’ allowing them to reduce the amount of added sugar in foods and drinks, to reduce their sugar intake and still enjoy what they eat and drink, on the way to reducing both added sugar and sweeteners in their diet,” Dr. Mellor suggests.
Kevin McConway, PhD, with the Open University, Milton Keynes, England, said it’s “important to understand that the research is not saying that these sweeteners are worse for us, in heath terms, than sugar.
“But exactly what the health consequences of all this, if any, might be is a subject for future research,” Dr. McConway added.
Kathy Redfern, PhD, lecturer in human nutrition, University of Plymouth (England) agrees.
“We still have a lot to learn about the human microbiome, and although this study suggests two of the sweeteners tested in this study (sucralose and saccharin) significantly affected glucose tolerance, these deviations were small,” she says.
The International Sweeteners Association also weighs in, saying, “No conclusions about the effects of low/no calorie sweeteners on glucose control or overall health can be extrapolated from this study for the general population or for people who typically consume sweeteners, including people living with diabetes.”
They add “a recent review of the literature concluded that there is clear evidence that changes in the diet unrelated to low/no calorie sweeteners consumption are likely the major determinants of change in gut microbiota.”
Nevertheless, Dr. Redfern says the results “warrant further investigation to assess how small changes in glucose tolerance in response to NNS consumption may influence longer-term glucose tolerance and risk for metabolic complications, such as type 2 diabetes.”
The study had no specific funding. Dr. Elinav is a scientific founder of DayTwo and BiomX, a paid consultant to Hello Inside and Aposense, and a member of the scientific advisory board of Cell. Dr. Mellor has provided consultancy to the International Sweetener Agency and has worked on projects funded by the Food Standards Agency that investigated the health effects of aspartame. Dr. Barrett, Dr. McConway, and Dr. Redfern report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This article was updated 8/29/22.
Guidelines on GLP1RAs and continuous glucose monitors are among biggest news in diabetes
glucagonlike peptide-1 receptor agonists (GLP1RAs) and continuous glucose monitoring (CGM) technology. I am hoping my discussion about these major advances in this edition of Highlights will be helpful to those caring for patients with diabetes.
Tirzepatide
The first GLP1RA, exenatide, was released in April 2005. Since then, numerous daily and weekly drugs of this class have been developed. We’ve learned they are effective glucose lowering drugs, and the weekly agents dulaglutide and semaglutide have shown impressive weight reduction properties as well as cardiovascular benefits.
Secondary outcomes have also shown renal benefits to these agents, and studies for primary renal efficacy are pending. Due to all of these properties, the GLP1RAs are recommended as the first injectable for the treatment of type 2 diabetes, prior to insulin initiation.1
The next generation of these agents are a combination of a GLP1RA and a glucose-dependent insulinotropic polypeptide (GIP). Glucagonlike peptide-1 (GLP-1) stimulates insulin secretion, inhibits glucagon secretion, delays gastric emptying, and has central effects inducing satiety.
We now understand that GIP is the main incretin hormone in those without diabetes, causative of most of the incretin effects. But the insulin response after GIP secretion in type 2 diabetes is strongly reduced. It is now appreciated that this poor effect of GIP can be reduced when used in combination with a GLP1RA. This combination incretin, called by some a “twincretin,” is the basis for the drug tirzepatide which was approved by the Food and Drug Administration in May of 2022.
The data supporting this agent for both diabetes and obesity are impressive. For example, in a 40-week study with a baseline HbA1c of 8.0%, those randomized to tirzepatide at 5 mg, 10 mg, and 15 mg had HbA1c reductions of 1.87%, 1.89%, and 2.07% respectively.2 Over 81% at all doses had HbA1c levels less than 6.5% at 40 weeks.
For the 5-mg, 10-mg, and 15-mg doses, weight change from baseline was 7.9%, 9.3%, and 11.0% respectively. Like older GLP1RAs, gastrointestinal side effects were the main problem. For the three doses, 3%, 5%, and 7%, respectively, had to stop the drug, compared with the 3% who stopped taking the placebo. In another study, tirzepatide was noninferior or superior at all three doses compared with semaglutide 1 mg weekly.3
In a population without diabetes, with 40% of patients having prediabetes, weight loss percentages for the three doses were 15.0%, 19.5%, and 20.9% respectively.4 Discontinuation percentages due to side effects were 4%-7%. The exciting part is we now have a drug that approaches weight loss from bariatric surgery. The cardiovascular and renal outcome trials are now underway, but the enthusiasm for this drug is clear from the data.
Like other GLP1RAs, the key is to start low and go slowly. It is recommended to start tirzepatide at 2.5 mg four times a week, then increase to 5 mg. Due to gastrointestinal side effects, some patients will do better at the lower dose before increasing. For those switching from another GLP1RA, there are no data to guide us but, in my practice, I start those patients at 5 mg weekly.
Continuous glucose monitoring
Data continue to accumulate that this form of glycemic self-monitoring is effective to reduce HbA1c levels and minimize hypoglycemia in both type 1 and type 2 diabetes. The most important change to the 2022 American Diabetes Association (ADA) standards of care is recognizing CGM as level A evidence for those receiving basal insulin without mealtime insulin.5 There are four CGMs on the market, but most of the market uses the Dexcom G6 or the Libre 2. Both of these devices will be updated within the next few months to newer generation sensors.
While there are similarities and differences between the two devices, by late 2022 and early 2023 changes to both will reduce the dissimilarities.
The next generation Libre (Libre 3) will be continuous, and “scanning” will no longer be required. For those unable to get insurance to cover CGM, the Libre will continue to be more affordable than the Dexcom. Alerts will be present on both, but the Dexcom G7 will be approved for both the arm and the abdomen. The Dexcom also can communicate with several automated insulin delivery systems and data can be shared real-time with family members.
For clinicians just starting patients on this technology, my suggestion is to focus on one system so both the provider and staff can become familiar with it. It is key to review downloaded glucose metrics, in addition to the “ambulatory glucose profile,” a graphic overview of daily glycemia where patterns can be identified. It is also helpful to ask for assistance from endocrinologists who have experience with CGMs, in addition to the representatives of the companies.
COVID-19 and new-onset diabetes
From the beginning of the COVID 19 pandemic in 2020, it was clear that stress hyperglycemia and glucose dysregulation was an important observation for those infected. What was not known at the time is that for some, the hyperglycemia continued, and permanent diabetes ensued.
In one study of over 2.7 million U.S. veterans, men infected with COVID-19, but not women, were at a higher risk of new incident diabetes at 120 days after infection compared to no infection (odds ratio for men = 2.56).6
Another literature review using meta-analyses and cross-sectional studies concluded new-onset diabetes following COVID-19 infection can have a varied phenotype, with no risk factors, presenting from diabetic ketoacidosis to milder forms of diabetes.7
The current thought is that COVID-19 binds to the ACE2 and TMPRSS2 receptors which appear to be located on the beta-cells in the islet, resulting in insulin deficiency, in addition to the insulin resistance that seems to persist after the acute infection. Much more needs to be learned about this, but clinicians need to appreciate this appears to be a new form of diabetes and optimal treatments are not yet clear.
Dr. Hirsch is an endocrinologist, professor of medicine, and diabetes treatment and teaching chair at the University of Washington, Seattle. He has received research grant support from Dexcom and Insulet and has provided consulting to Abbott, Roche, Lifescan, and GWave. You can contact him at [email protected].
References
1. American Diabetes Association Professional Practice Committee. Pharmacologic approaches to glycemic treatment: Standards of Medical Care in Diabetes-2022. Diabetes Care. 2022;45(Suppl 1):S125-S143.
2. Rosenstock J et al. Efficacy and safety of a novel GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): A double-blind, randomised, phase 3 trial. Lancet. 2021;398:143-55.
3. Frias JP et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385:503-15.
4. Jastreboff AM et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205-16.
5. American Diabetes Association Professional Practice Committee. Diabetes technology: Standards of Medical Care in Diabetes–2022. Diabetes Care. 2022;45(Suppl 1):S97-S112.
6. Wander PL et al. The incidence of diabetes in 2,777,768 veterans with and without recent SARS-CoV-2 infection. Diabetes Care 2022;45:782-8.
7. Joshi SC and Pozzilli P. COVID-19 induced diabetes: A novel presentation. Diabetes Res Clin Pract. 2022 Aug 6;191:110034.
glucagonlike peptide-1 receptor agonists (GLP1RAs) and continuous glucose monitoring (CGM) technology. I am hoping my discussion about these major advances in this edition of Highlights will be helpful to those caring for patients with diabetes.
Tirzepatide
The first GLP1RA, exenatide, was released in April 2005. Since then, numerous daily and weekly drugs of this class have been developed. We’ve learned they are effective glucose lowering drugs, and the weekly agents dulaglutide and semaglutide have shown impressive weight reduction properties as well as cardiovascular benefits.
Secondary outcomes have also shown renal benefits to these agents, and studies for primary renal efficacy are pending. Due to all of these properties, the GLP1RAs are recommended as the first injectable for the treatment of type 2 diabetes, prior to insulin initiation.1
The next generation of these agents are a combination of a GLP1RA and a glucose-dependent insulinotropic polypeptide (GIP). Glucagonlike peptide-1 (GLP-1) stimulates insulin secretion, inhibits glucagon secretion, delays gastric emptying, and has central effects inducing satiety.
We now understand that GIP is the main incretin hormone in those without diabetes, causative of most of the incretin effects. But the insulin response after GIP secretion in type 2 diabetes is strongly reduced. It is now appreciated that this poor effect of GIP can be reduced when used in combination with a GLP1RA. This combination incretin, called by some a “twincretin,” is the basis for the drug tirzepatide which was approved by the Food and Drug Administration in May of 2022.
The data supporting this agent for both diabetes and obesity are impressive. For example, in a 40-week study with a baseline HbA1c of 8.0%, those randomized to tirzepatide at 5 mg, 10 mg, and 15 mg had HbA1c reductions of 1.87%, 1.89%, and 2.07% respectively.2 Over 81% at all doses had HbA1c levels less than 6.5% at 40 weeks.
For the 5-mg, 10-mg, and 15-mg doses, weight change from baseline was 7.9%, 9.3%, and 11.0% respectively. Like older GLP1RAs, gastrointestinal side effects were the main problem. For the three doses, 3%, 5%, and 7%, respectively, had to stop the drug, compared with the 3% who stopped taking the placebo. In another study, tirzepatide was noninferior or superior at all three doses compared with semaglutide 1 mg weekly.3
In a population without diabetes, with 40% of patients having prediabetes, weight loss percentages for the three doses were 15.0%, 19.5%, and 20.9% respectively.4 Discontinuation percentages due to side effects were 4%-7%. The exciting part is we now have a drug that approaches weight loss from bariatric surgery. The cardiovascular and renal outcome trials are now underway, but the enthusiasm for this drug is clear from the data.
Like other GLP1RAs, the key is to start low and go slowly. It is recommended to start tirzepatide at 2.5 mg four times a week, then increase to 5 mg. Due to gastrointestinal side effects, some patients will do better at the lower dose before increasing. For those switching from another GLP1RA, there are no data to guide us but, in my practice, I start those patients at 5 mg weekly.
Continuous glucose monitoring
Data continue to accumulate that this form of glycemic self-monitoring is effective to reduce HbA1c levels and minimize hypoglycemia in both type 1 and type 2 diabetes. The most important change to the 2022 American Diabetes Association (ADA) standards of care is recognizing CGM as level A evidence for those receiving basal insulin without mealtime insulin.5 There are four CGMs on the market, but most of the market uses the Dexcom G6 or the Libre 2. Both of these devices will be updated within the next few months to newer generation sensors.
While there are similarities and differences between the two devices, by late 2022 and early 2023 changes to both will reduce the dissimilarities.
The next generation Libre (Libre 3) will be continuous, and “scanning” will no longer be required. For those unable to get insurance to cover CGM, the Libre will continue to be more affordable than the Dexcom. Alerts will be present on both, but the Dexcom G7 will be approved for both the arm and the abdomen. The Dexcom also can communicate with several automated insulin delivery systems and data can be shared real-time with family members.
For clinicians just starting patients on this technology, my suggestion is to focus on one system so both the provider and staff can become familiar with it. It is key to review downloaded glucose metrics, in addition to the “ambulatory glucose profile,” a graphic overview of daily glycemia where patterns can be identified. It is also helpful to ask for assistance from endocrinologists who have experience with CGMs, in addition to the representatives of the companies.
COVID-19 and new-onset diabetes
From the beginning of the COVID 19 pandemic in 2020, it was clear that stress hyperglycemia and glucose dysregulation was an important observation for those infected. What was not known at the time is that for some, the hyperglycemia continued, and permanent diabetes ensued.
In one study of over 2.7 million U.S. veterans, men infected with COVID-19, but not women, were at a higher risk of new incident diabetes at 120 days after infection compared to no infection (odds ratio for men = 2.56).6
Another literature review using meta-analyses and cross-sectional studies concluded new-onset diabetes following COVID-19 infection can have a varied phenotype, with no risk factors, presenting from diabetic ketoacidosis to milder forms of diabetes.7
The current thought is that COVID-19 binds to the ACE2 and TMPRSS2 receptors which appear to be located on the beta-cells in the islet, resulting in insulin deficiency, in addition to the insulin resistance that seems to persist after the acute infection. Much more needs to be learned about this, but clinicians need to appreciate this appears to be a new form of diabetes and optimal treatments are not yet clear.
Dr. Hirsch is an endocrinologist, professor of medicine, and diabetes treatment and teaching chair at the University of Washington, Seattle. He has received research grant support from Dexcom and Insulet and has provided consulting to Abbott, Roche, Lifescan, and GWave. You can contact him at [email protected].
References
1. American Diabetes Association Professional Practice Committee. Pharmacologic approaches to glycemic treatment: Standards of Medical Care in Diabetes-2022. Diabetes Care. 2022;45(Suppl 1):S125-S143.
2. Rosenstock J et al. Efficacy and safety of a novel GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): A double-blind, randomised, phase 3 trial. Lancet. 2021;398:143-55.
3. Frias JP et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385:503-15.
4. Jastreboff AM et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205-16.
5. American Diabetes Association Professional Practice Committee. Diabetes technology: Standards of Medical Care in Diabetes–2022. Diabetes Care. 2022;45(Suppl 1):S97-S112.
6. Wander PL et al. The incidence of diabetes in 2,777,768 veterans with and without recent SARS-CoV-2 infection. Diabetes Care 2022;45:782-8.
7. Joshi SC and Pozzilli P. COVID-19 induced diabetes: A novel presentation. Diabetes Res Clin Pract. 2022 Aug 6;191:110034.
glucagonlike peptide-1 receptor agonists (GLP1RAs) and continuous glucose monitoring (CGM) technology. I am hoping my discussion about these major advances in this edition of Highlights will be helpful to those caring for patients with diabetes.
Tirzepatide
The first GLP1RA, exenatide, was released in April 2005. Since then, numerous daily and weekly drugs of this class have been developed. We’ve learned they are effective glucose lowering drugs, and the weekly agents dulaglutide and semaglutide have shown impressive weight reduction properties as well as cardiovascular benefits.
Secondary outcomes have also shown renal benefits to these agents, and studies for primary renal efficacy are pending. Due to all of these properties, the GLP1RAs are recommended as the first injectable for the treatment of type 2 diabetes, prior to insulin initiation.1
The next generation of these agents are a combination of a GLP1RA and a glucose-dependent insulinotropic polypeptide (GIP). Glucagonlike peptide-1 (GLP-1) stimulates insulin secretion, inhibits glucagon secretion, delays gastric emptying, and has central effects inducing satiety.
We now understand that GIP is the main incretin hormone in those without diabetes, causative of most of the incretin effects. But the insulin response after GIP secretion in type 2 diabetes is strongly reduced. It is now appreciated that this poor effect of GIP can be reduced when used in combination with a GLP1RA. This combination incretin, called by some a “twincretin,” is the basis for the drug tirzepatide which was approved by the Food and Drug Administration in May of 2022.
The data supporting this agent for both diabetes and obesity are impressive. For example, in a 40-week study with a baseline HbA1c of 8.0%, those randomized to tirzepatide at 5 mg, 10 mg, and 15 mg had HbA1c reductions of 1.87%, 1.89%, and 2.07% respectively.2 Over 81% at all doses had HbA1c levels less than 6.5% at 40 weeks.
For the 5-mg, 10-mg, and 15-mg doses, weight change from baseline was 7.9%, 9.3%, and 11.0% respectively. Like older GLP1RAs, gastrointestinal side effects were the main problem. For the three doses, 3%, 5%, and 7%, respectively, had to stop the drug, compared with the 3% who stopped taking the placebo. In another study, tirzepatide was noninferior or superior at all three doses compared with semaglutide 1 mg weekly.3
In a population without diabetes, with 40% of patients having prediabetes, weight loss percentages for the three doses were 15.0%, 19.5%, and 20.9% respectively.4 Discontinuation percentages due to side effects were 4%-7%. The exciting part is we now have a drug that approaches weight loss from bariatric surgery. The cardiovascular and renal outcome trials are now underway, but the enthusiasm for this drug is clear from the data.
Like other GLP1RAs, the key is to start low and go slowly. It is recommended to start tirzepatide at 2.5 mg four times a week, then increase to 5 mg. Due to gastrointestinal side effects, some patients will do better at the lower dose before increasing. For those switching from another GLP1RA, there are no data to guide us but, in my practice, I start those patients at 5 mg weekly.
Continuous glucose monitoring
Data continue to accumulate that this form of glycemic self-monitoring is effective to reduce HbA1c levels and minimize hypoglycemia in both type 1 and type 2 diabetes. The most important change to the 2022 American Diabetes Association (ADA) standards of care is recognizing CGM as level A evidence for those receiving basal insulin without mealtime insulin.5 There are four CGMs on the market, but most of the market uses the Dexcom G6 or the Libre 2. Both of these devices will be updated within the next few months to newer generation sensors.
While there are similarities and differences between the two devices, by late 2022 and early 2023 changes to both will reduce the dissimilarities.
The next generation Libre (Libre 3) will be continuous, and “scanning” will no longer be required. For those unable to get insurance to cover CGM, the Libre will continue to be more affordable than the Dexcom. Alerts will be present on both, but the Dexcom G7 will be approved for both the arm and the abdomen. The Dexcom also can communicate with several automated insulin delivery systems and data can be shared real-time with family members.
For clinicians just starting patients on this technology, my suggestion is to focus on one system so both the provider and staff can become familiar with it. It is key to review downloaded glucose metrics, in addition to the “ambulatory glucose profile,” a graphic overview of daily glycemia where patterns can be identified. It is also helpful to ask for assistance from endocrinologists who have experience with CGMs, in addition to the representatives of the companies.
COVID-19 and new-onset diabetes
From the beginning of the COVID 19 pandemic in 2020, it was clear that stress hyperglycemia and glucose dysregulation was an important observation for those infected. What was not known at the time is that for some, the hyperglycemia continued, and permanent diabetes ensued.
In one study of over 2.7 million U.S. veterans, men infected with COVID-19, but not women, were at a higher risk of new incident diabetes at 120 days after infection compared to no infection (odds ratio for men = 2.56).6
Another literature review using meta-analyses and cross-sectional studies concluded new-onset diabetes following COVID-19 infection can have a varied phenotype, with no risk factors, presenting from diabetic ketoacidosis to milder forms of diabetes.7
The current thought is that COVID-19 binds to the ACE2 and TMPRSS2 receptors which appear to be located on the beta-cells in the islet, resulting in insulin deficiency, in addition to the insulin resistance that seems to persist after the acute infection. Much more needs to be learned about this, but clinicians need to appreciate this appears to be a new form of diabetes and optimal treatments are not yet clear.
Dr. Hirsch is an endocrinologist, professor of medicine, and diabetes treatment and teaching chair at the University of Washington, Seattle. He has received research grant support from Dexcom and Insulet and has provided consulting to Abbott, Roche, Lifescan, and GWave. You can contact him at [email protected].
References
1. American Diabetes Association Professional Practice Committee. Pharmacologic approaches to glycemic treatment: Standards of Medical Care in Diabetes-2022. Diabetes Care. 2022;45(Suppl 1):S125-S143.
2. Rosenstock J et al. Efficacy and safety of a novel GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): A double-blind, randomised, phase 3 trial. Lancet. 2021;398:143-55.
3. Frias JP et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385:503-15.
4. Jastreboff AM et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205-16.
5. American Diabetes Association Professional Practice Committee. Diabetes technology: Standards of Medical Care in Diabetes–2022. Diabetes Care. 2022;45(Suppl 1):S97-S112.
6. Wander PL et al. The incidence of diabetes in 2,777,768 veterans with and without recent SARS-CoV-2 infection. Diabetes Care 2022;45:782-8.
7. Joshi SC and Pozzilli P. COVID-19 induced diabetes: A novel presentation. Diabetes Res Clin Pract. 2022 Aug 6;191:110034.
‘Obesity paradox’ in AFib challenged as mortality climbs with BMI
The relationship between body mass index (BMI) and all-cause mortality in patients with atrial fibrillation (AFib) is U-shaped, with the risk highest in those who are underweight or severely obese and lowest in patients defined simply as obese, a registry analysis suggests. It also showed a similar relationship between BMI and risk for new or worsening heart failure (HF).
Mortality bottomed out at a BMI of about 30-35 kg/m2, which suggests that mild obesity was protective, compared even with “normal-weight” or “overweight” BMI. Still, mortality went up sharply from there with rising BMI.
But higher BMI, a surrogate for obesity, apparently didn’t worsen outcomes by itself. The risk for death from any cause at higher obesity levels was found to depend a lot on related risk factors and comorbidities when the analysis controlled for conditions such as diabetes and hypertension.
The findings suggest an inverse relationship between BMI and all-cause mortality in AFib only for patients with BMI less than about 30. They therefore argue against any “obesity paradox” in AFib that posits consistently better survival with increasing levels of obesity, say researchers, based on their analysis of patients with new-onset AFib in the GARFIELD-AF registry.
“It’s common practice now for clinicians to discuss weight within a clinic setting when they’re talking to their AFib patients,” observed Christian Fielder Camm, BM, BCh, University of Oxford (England), and Royal Berkshire NHS Foundation Trust, Reading, England. So studies suggesting an inverse association between BMI and AFib-related risk can be a concern.
Such studies “seem to suggest that once you’ve got AFib, maintaining a high or very high BMI may in some way be protective – which is contrary to what would seem to make sense and certainly contrary to what our results have shown,” Dr. Camm told this news organization.
“I think that having further evidence now to suggest, actually, that greater BMI is associated with a greater risk of all-cause mortality and heart failure helps reframe that discussion at the physician-patient interaction level more clearly, and ensures that we’re able to talk to our patients appropriately about risks associated with BMI and atrial fibrillation,” said Dr. Camm, who is lead author on the analysis published in Open Heart.
“Obesity is a cause of most cardiovascular diseases, but [these] data would support that being overweight or having mild obesity does not increase the risk,” observed Carl J. Lavie, MD, of the John Ochsner Heart and Vascular Institute, New Orleans, La., and the Ochsner Clinical School at the University of Queensland, Brisbane, Australia.
“At a BMI of 40, it’s very important for them to lose weight for their long-term prognosis,” Dr. Lavie noted, but “at a BMI of 30, the important thing would be to prevent further weight gain. And if they could keep their BMI of 30, they should have a good prognosis. Their prognosis would be particularly good if they didn’t gain weight and put themselves in a more extreme obesity class that is associated with worse risk.”
The current analysis, Dr. Lavie said, “is way better than the AFFIRM study,” which yielded an obesity-paradox report on its patients with AFib about a dozen years ago. “It’s got more data, more numbers, more statistical power,” and breaks BMI into more categories.
That previous analysis based on the influential AFFIRM randomized trial separated its 4,060 patients with AFib into normal (BMI, 18.5-25), overweight (BMI, 25-30), and obese (BMI, > 30) categories, per the convention at the time. It concluded that “obese patients with atrial fibrillation appear to have better long-term outcomes than nonobese patients.”
Bleeding risk on oral anticoagulants
Also noteworthy in the current analysis, variation in BMI didn’t seem to affect mortality or risk for major bleeding or nonhemorrhagic stroke according to choice of oral anticoagulant – whether a new oral anticoagulant (NOAC) or a vitamin K antagonist (VKA).
“We saw that even in the obese and extremely obese group, all-cause mortality was lower in the group taking NOACs, compared with taking warfarin,” Dr. Camm observed, “which goes against the idea that we would need any kind of dose adjustments for increased BMI.”
Indeed, the report notes, use of NOACs, compared with VKA, was associated with a 23% drop in risk for death among patients who were either normal weight or overweight and also in those who were obese or extremely obese.
Those findings “are basically saying that the NOACs look better than warfarin regardless of weight,” agreed Dr. Lavie. “The problem is that the study is not very powered.”
Whereas the benefits of NOACs, compared to VKA, seem similar for patients with a BMI of 30 or 34, compared with a BMI of 23, for example, “none of the studies has many people with 50 BMI.” Many clinicians “feel uncomfortable giving the same dose of NOAC to somebody who has a 60 BMI,” he said. At least with warfarin, “you can check the INR [international normalized ratio].”
The current analysis included 40,482 patients with recently diagnosed AFib and at least one other stroke risk factor from among the registry’s more than 50,000 patients from 35 countries, enrolled from 2010 to 2016. They were followed for 2 years.
The 703 patients with BMI under 18.5 at AFib diagnosis were classified per World Health Organization definitions as underweight; the 13,095 with BMI 18.5-25 as normal weight; the 15,043 with BMI 25-30 as overweight; the 7,560 with BMI 30-35 as obese; and the 4,081 with BMI above 35 as extremely obese. Their ages averaged 71 years, and 55.6% were men.
BMI effects on different outcomes
Relationships between BMI and all-cause mortality and between BMI and new or worsening HF emerged as U-shaped, the risk climbing with both increasing and decreasing BMI. The nadir BMI for risk was about 30 in the case of mortality and about 25 for new or worsening HF.
The all-cause mortality risk rose by 32% for every 5 BMI points lower than a BMI of 30, and by 16% for every 5 BMI points higher than 30, in a partially adjusted analysis. The risk for new or worsening HF rose significantly with increasing but not decreasing BMI, and the reverse was observed for the endpoint of major bleeding.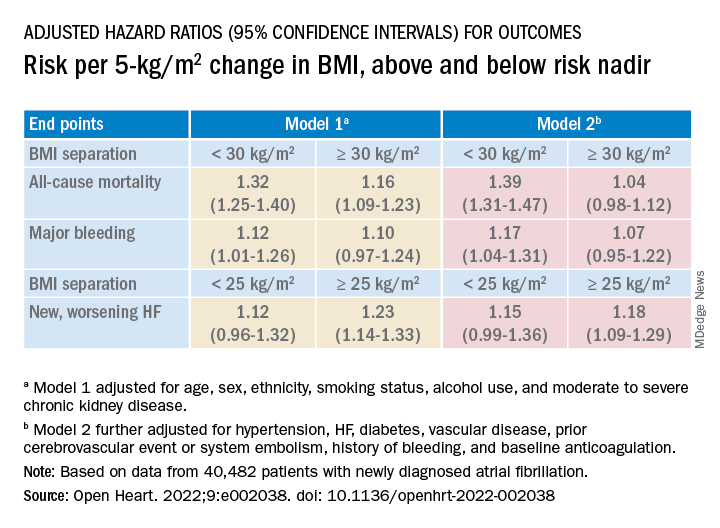
The effect of BMI on all-cause mortality was “substantially attenuated” when the analysis was further adjusted with “likely mediators of any association between BMI and outcomes,” including hypertension, diabetes, HF, cerebrovascular events, and history of bleeding, Dr. Camm said.
That blunted BMI-mortality relationship, he said, “suggests that a lot of the effect is mediated through relatively traditional risk factors like hypertension and diabetes.”
The 2010 AFFIRM analysis by BMI, Dr. Lavie noted, “didn’t even look at the underweight; they actually threw them out.” Yet, such patients with AFib, who tend to be extremely frail or have chronic diseases or conditions other than the arrhythmia, are common. A take-home of the current study is that “the underweight with atrial fibrillation have a really bad prognosis.”
That message isn’t heard as much, he observed, “but is as important as saying that BMI 30 has the best prognosis. The worst prognosis is with the underweight or the really extreme obese.”
Dr. Camm discloses research funding from the British Heart Foundation. Disclosures for the other authors are in the report. Dr. Lavie has previously disclosed serving as a speaker and consultant for PAI Health and DSM Nutritional Products and is the author of “The Obesity Paradox: When Thinner Means Sicker and Heavier Means Healthier” (Avery, 2014).
A version of this article first appeared on Medscape.com.
The relationship between body mass index (BMI) and all-cause mortality in patients with atrial fibrillation (AFib) is U-shaped, with the risk highest in those who are underweight or severely obese and lowest in patients defined simply as obese, a registry analysis suggests. It also showed a similar relationship between BMI and risk for new or worsening heart failure (HF).
Mortality bottomed out at a BMI of about 30-35 kg/m2, which suggests that mild obesity was protective, compared even with “normal-weight” or “overweight” BMI. Still, mortality went up sharply from there with rising BMI.
But higher BMI, a surrogate for obesity, apparently didn’t worsen outcomes by itself. The risk for death from any cause at higher obesity levels was found to depend a lot on related risk factors and comorbidities when the analysis controlled for conditions such as diabetes and hypertension.
The findings suggest an inverse relationship between BMI and all-cause mortality in AFib only for patients with BMI less than about 30. They therefore argue against any “obesity paradox” in AFib that posits consistently better survival with increasing levels of obesity, say researchers, based on their analysis of patients with new-onset AFib in the GARFIELD-AF registry.
“It’s common practice now for clinicians to discuss weight within a clinic setting when they’re talking to their AFib patients,” observed Christian Fielder Camm, BM, BCh, University of Oxford (England), and Royal Berkshire NHS Foundation Trust, Reading, England. So studies suggesting an inverse association between BMI and AFib-related risk can be a concern.
Such studies “seem to suggest that once you’ve got AFib, maintaining a high or very high BMI may in some way be protective – which is contrary to what would seem to make sense and certainly contrary to what our results have shown,” Dr. Camm told this news organization.
“I think that having further evidence now to suggest, actually, that greater BMI is associated with a greater risk of all-cause mortality and heart failure helps reframe that discussion at the physician-patient interaction level more clearly, and ensures that we’re able to talk to our patients appropriately about risks associated with BMI and atrial fibrillation,” said Dr. Camm, who is lead author on the analysis published in Open Heart.
“Obesity is a cause of most cardiovascular diseases, but [these] data would support that being overweight or having mild obesity does not increase the risk,” observed Carl J. Lavie, MD, of the John Ochsner Heart and Vascular Institute, New Orleans, La., and the Ochsner Clinical School at the University of Queensland, Brisbane, Australia.
“At a BMI of 40, it’s very important for them to lose weight for their long-term prognosis,” Dr. Lavie noted, but “at a BMI of 30, the important thing would be to prevent further weight gain. And if they could keep their BMI of 30, they should have a good prognosis. Their prognosis would be particularly good if they didn’t gain weight and put themselves in a more extreme obesity class that is associated with worse risk.”
The current analysis, Dr. Lavie said, “is way better than the AFFIRM study,” which yielded an obesity-paradox report on its patients with AFib about a dozen years ago. “It’s got more data, more numbers, more statistical power,” and breaks BMI into more categories.
That previous analysis based on the influential AFFIRM randomized trial separated its 4,060 patients with AFib into normal (BMI, 18.5-25), overweight (BMI, 25-30), and obese (BMI, > 30) categories, per the convention at the time. It concluded that “obese patients with atrial fibrillation appear to have better long-term outcomes than nonobese patients.”
Bleeding risk on oral anticoagulants
Also noteworthy in the current analysis, variation in BMI didn’t seem to affect mortality or risk for major bleeding or nonhemorrhagic stroke according to choice of oral anticoagulant – whether a new oral anticoagulant (NOAC) or a vitamin K antagonist (VKA).
“We saw that even in the obese and extremely obese group, all-cause mortality was lower in the group taking NOACs, compared with taking warfarin,” Dr. Camm observed, “which goes against the idea that we would need any kind of dose adjustments for increased BMI.”
Indeed, the report notes, use of NOACs, compared with VKA, was associated with a 23% drop in risk for death among patients who were either normal weight or overweight and also in those who were obese or extremely obese.
Those findings “are basically saying that the NOACs look better than warfarin regardless of weight,” agreed Dr. Lavie. “The problem is that the study is not very powered.”
Whereas the benefits of NOACs, compared to VKA, seem similar for patients with a BMI of 30 or 34, compared with a BMI of 23, for example, “none of the studies has many people with 50 BMI.” Many clinicians “feel uncomfortable giving the same dose of NOAC to somebody who has a 60 BMI,” he said. At least with warfarin, “you can check the INR [international normalized ratio].”
The current analysis included 40,482 patients with recently diagnosed AFib and at least one other stroke risk factor from among the registry’s more than 50,000 patients from 35 countries, enrolled from 2010 to 2016. They were followed for 2 years.
The 703 patients with BMI under 18.5 at AFib diagnosis were classified per World Health Organization definitions as underweight; the 13,095 with BMI 18.5-25 as normal weight; the 15,043 with BMI 25-30 as overweight; the 7,560 with BMI 30-35 as obese; and the 4,081 with BMI above 35 as extremely obese. Their ages averaged 71 years, and 55.6% were men.
BMI effects on different outcomes
Relationships between BMI and all-cause mortality and between BMI and new or worsening HF emerged as U-shaped, the risk climbing with both increasing and decreasing BMI. The nadir BMI for risk was about 30 in the case of mortality and about 25 for new or worsening HF.
The all-cause mortality risk rose by 32% for every 5 BMI points lower than a BMI of 30, and by 16% for every 5 BMI points higher than 30, in a partially adjusted analysis. The risk for new or worsening HF rose significantly with increasing but not decreasing BMI, and the reverse was observed for the endpoint of major bleeding.
The effect of BMI on all-cause mortality was “substantially attenuated” when the analysis was further adjusted with “likely mediators of any association between BMI and outcomes,” including hypertension, diabetes, HF, cerebrovascular events, and history of bleeding, Dr. Camm said.
That blunted BMI-mortality relationship, he said, “suggests that a lot of the effect is mediated through relatively traditional risk factors like hypertension and diabetes.”
The 2010 AFFIRM analysis by BMI, Dr. Lavie noted, “didn’t even look at the underweight; they actually threw them out.” Yet, such patients with AFib, who tend to be extremely frail or have chronic diseases or conditions other than the arrhythmia, are common. A take-home of the current study is that “the underweight with atrial fibrillation have a really bad prognosis.”
That message isn’t heard as much, he observed, “but is as important as saying that BMI 30 has the best prognosis. The worst prognosis is with the underweight or the really extreme obese.”
Dr. Camm discloses research funding from the British Heart Foundation. Disclosures for the other authors are in the report. Dr. Lavie has previously disclosed serving as a speaker and consultant for PAI Health and DSM Nutritional Products and is the author of “The Obesity Paradox: When Thinner Means Sicker and Heavier Means Healthier” (Avery, 2014).
A version of this article first appeared on Medscape.com.
The relationship between body mass index (BMI) and all-cause mortality in patients with atrial fibrillation (AFib) is U-shaped, with the risk highest in those who are underweight or severely obese and lowest in patients defined simply as obese, a registry analysis suggests. It also showed a similar relationship between BMI and risk for new or worsening heart failure (HF).
Mortality bottomed out at a BMI of about 30-35 kg/m2, which suggests that mild obesity was protective, compared even with “normal-weight” or “overweight” BMI. Still, mortality went up sharply from there with rising BMI.
But higher BMI, a surrogate for obesity, apparently didn’t worsen outcomes by itself. The risk for death from any cause at higher obesity levels was found to depend a lot on related risk factors and comorbidities when the analysis controlled for conditions such as diabetes and hypertension.
The findings suggest an inverse relationship between BMI and all-cause mortality in AFib only for patients with BMI less than about 30. They therefore argue against any “obesity paradox” in AFib that posits consistently better survival with increasing levels of obesity, say researchers, based on their analysis of patients with new-onset AFib in the GARFIELD-AF registry.
“It’s common practice now for clinicians to discuss weight within a clinic setting when they’re talking to their AFib patients,” observed Christian Fielder Camm, BM, BCh, University of Oxford (England), and Royal Berkshire NHS Foundation Trust, Reading, England. So studies suggesting an inverse association between BMI and AFib-related risk can be a concern.
Such studies “seem to suggest that once you’ve got AFib, maintaining a high or very high BMI may in some way be protective – which is contrary to what would seem to make sense and certainly contrary to what our results have shown,” Dr. Camm told this news organization.
“I think that having further evidence now to suggest, actually, that greater BMI is associated with a greater risk of all-cause mortality and heart failure helps reframe that discussion at the physician-patient interaction level more clearly, and ensures that we’re able to talk to our patients appropriately about risks associated with BMI and atrial fibrillation,” said Dr. Camm, who is lead author on the analysis published in Open Heart.
“Obesity is a cause of most cardiovascular diseases, but [these] data would support that being overweight or having mild obesity does not increase the risk,” observed Carl J. Lavie, MD, of the John Ochsner Heart and Vascular Institute, New Orleans, La., and the Ochsner Clinical School at the University of Queensland, Brisbane, Australia.
“At a BMI of 40, it’s very important for them to lose weight for their long-term prognosis,” Dr. Lavie noted, but “at a BMI of 30, the important thing would be to prevent further weight gain. And if they could keep their BMI of 30, they should have a good prognosis. Their prognosis would be particularly good if they didn’t gain weight and put themselves in a more extreme obesity class that is associated with worse risk.”
The current analysis, Dr. Lavie said, “is way better than the AFFIRM study,” which yielded an obesity-paradox report on its patients with AFib about a dozen years ago. “It’s got more data, more numbers, more statistical power,” and breaks BMI into more categories.
That previous analysis based on the influential AFFIRM randomized trial separated its 4,060 patients with AFib into normal (BMI, 18.5-25), overweight (BMI, 25-30), and obese (BMI, > 30) categories, per the convention at the time. It concluded that “obese patients with atrial fibrillation appear to have better long-term outcomes than nonobese patients.”
Bleeding risk on oral anticoagulants
Also noteworthy in the current analysis, variation in BMI didn’t seem to affect mortality or risk for major bleeding or nonhemorrhagic stroke according to choice of oral anticoagulant – whether a new oral anticoagulant (NOAC) or a vitamin K antagonist (VKA).
“We saw that even in the obese and extremely obese group, all-cause mortality was lower in the group taking NOACs, compared with taking warfarin,” Dr. Camm observed, “which goes against the idea that we would need any kind of dose adjustments for increased BMI.”
Indeed, the report notes, use of NOACs, compared with VKA, was associated with a 23% drop in risk for death among patients who were either normal weight or overweight and also in those who were obese or extremely obese.
Those findings “are basically saying that the NOACs look better than warfarin regardless of weight,” agreed Dr. Lavie. “The problem is that the study is not very powered.”
Whereas the benefits of NOACs, compared to VKA, seem similar for patients with a BMI of 30 or 34, compared with a BMI of 23, for example, “none of the studies has many people with 50 BMI.” Many clinicians “feel uncomfortable giving the same dose of NOAC to somebody who has a 60 BMI,” he said. At least with warfarin, “you can check the INR [international normalized ratio].”
The current analysis included 40,482 patients with recently diagnosed AFib and at least one other stroke risk factor from among the registry’s more than 50,000 patients from 35 countries, enrolled from 2010 to 2016. They were followed for 2 years.
The 703 patients with BMI under 18.5 at AFib diagnosis were classified per World Health Organization definitions as underweight; the 13,095 with BMI 18.5-25 as normal weight; the 15,043 with BMI 25-30 as overweight; the 7,560 with BMI 30-35 as obese; and the 4,081 with BMI above 35 as extremely obese. Their ages averaged 71 years, and 55.6% were men.
BMI effects on different outcomes
Relationships between BMI and all-cause mortality and between BMI and new or worsening HF emerged as U-shaped, the risk climbing with both increasing and decreasing BMI. The nadir BMI for risk was about 30 in the case of mortality and about 25 for new or worsening HF.
The all-cause mortality risk rose by 32% for every 5 BMI points lower than a BMI of 30, and by 16% for every 5 BMI points higher than 30, in a partially adjusted analysis. The risk for new or worsening HF rose significantly with increasing but not decreasing BMI, and the reverse was observed for the endpoint of major bleeding.
The effect of BMI on all-cause mortality was “substantially attenuated” when the analysis was further adjusted with “likely mediators of any association between BMI and outcomes,” including hypertension, diabetes, HF, cerebrovascular events, and history of bleeding, Dr. Camm said.
That blunted BMI-mortality relationship, he said, “suggests that a lot of the effect is mediated through relatively traditional risk factors like hypertension and diabetes.”
The 2010 AFFIRM analysis by BMI, Dr. Lavie noted, “didn’t even look at the underweight; they actually threw them out.” Yet, such patients with AFib, who tend to be extremely frail or have chronic diseases or conditions other than the arrhythmia, are common. A take-home of the current study is that “the underweight with atrial fibrillation have a really bad prognosis.”
That message isn’t heard as much, he observed, “but is as important as saying that BMI 30 has the best prognosis. The worst prognosis is with the underweight or the really extreme obese.”
Dr. Camm discloses research funding from the British Heart Foundation. Disclosures for the other authors are in the report. Dr. Lavie has previously disclosed serving as a speaker and consultant for PAI Health and DSM Nutritional Products and is the author of “The Obesity Paradox: When Thinner Means Sicker and Heavier Means Healthier” (Avery, 2014).
A version of this article first appeared on Medscape.com.
FROM OPEN HEART
Obesity drug shortage triggers frustrations, workarounds
The glucagon-like peptide-1 (GLP-1) agonist semaglutide formulated for treating obesity (Wegovy) had a roaring takeoff a little more than a year ago, with surging patient demand after the U.S. Food and Drug Administration approved it in June 2021. But starting doses of the Wegovy form of semaglutide went missing in action starting late 2021 and continue to date, frustrating patients and their health care providers.
The arrival of Wegovy last year was hailed by obesity medicine specialists and others as a “game changer” for treating people with obesity because of semaglutide’s proven safety and efficacy at the subcutaneous dose of 2.4 mg delivered once a week to produce at least 15% weight loss in half the people who received it, as documented last year in results from one of the drug’s pivotal clinical trials.
But during the months following semaglutide’s approval for treating obesity (it also received an FDA marketing nod in late 2017 as Ozempic for treating type 2 diabetes), a worldwide shortage of Wegovy, including in the United States, emerged.
A manufacturing glitch shut down the primary location for production of U.S.-bound Wegovy injector pens for several months starting in late 2021, according to a December report from Novo Nordisk, the company that makes and markets the agent. (The Wegovy production issue appears to have had a very modest impact, especially in U.S. pharmacies, on the supply of semaglutide formulated as Ozempic, also marketed by Novo Nordisk, although Wegovy supply and demand have dramatically limited Ozempic availability in Australia.)
‘Unprecedented demand’ for Wegovy derailed when plant went offline
The supply side for Wegovy became so hopelessly broken that just months after U.S. sales began and immediately skyrocketed, Novo Nordisk made the remarkable decision to pull starting doses of Wegovy from the market to make it much harder to initiate patients (semaglutide and other GLP-1 agonists require gradual dose ramp-up to avoid gastrointestinal side effects), and the company publicly implored clinicians to not start new patients on the agent, which is where the status remains as of early August 2022.
Novo Nordisk’s financial report for the second quarter of 2022, released on Aug. 3, said the company “expects to make all Wegovy dose strengths available in the United States towards the end of 2022.”
A Dear Health Care Provider letter that Novo Nordisk posted on its U.S. Wegovy website last spring cited “unprecedented demand” that exceeded every prior product launch in the company’s history. It forced Novo Nordisk to pull the plug on all U.S. promotion of Wegovy and compelled the company to ask U.S. clinicians to halt new patient starts.
“I stopped offering Wegovy to new patients” since about the beginning of 2022, says Lauren D. Oshman, MD, a family and obesity medicine specialist at the University of Michigan, Ann Arbor. “It’s very frustrating to not have patients [with obesity] receive the optimal treatment available.” Although she adds that she tries to match obesity treatments to each patient’s clinical needs, and a GLP-1 agonist is not the first choice for every person with obesity.
“It was a disastrous rollout,” says Catherine W. Varney, DO, a family and obesity medicine specialist at the University of Virginia, Charlottesville. “It’s frustrating to know that the treatment is there but not being able to use it,” she said in an interview.
“I had about 800 patients on Wegovy” when the supply dropped earlier this year, and “I couldn’t handle the volume of messages that I got from patients,” recalls Angela Fitch, MD, associate director of the Massachusetts General Hospital Weight Center, Boston. “It was painful,” she said in an interview.
“Frustrating and chaotic,” is the description from Ivania M. Rizo, MD, director of obesity medicine at Boston Medical Center.
The liraglutide/Saxenda workaround
The upshot is that people with obesity and their health care providers have been busy devising workarounds to try to meet the intense demand for this drug-assisted approach to appetite control and weight loss. Their tactics run a wide gamut based on the crazy-quilt diversity of health insurance coverage across America.
Because the bottleneck for starting Wegovy resulted from unavailable starting doses (dosing starts at 0.25 mg delivered subcutaneously once a week, eventually ramping up to a maximum of 2.4 mg weekly), one option was to start patients on a different GLP-1 agonist, such as liraglutide (Saxenda, approved for obesity).
Starting a patient on liraglutide involves the same sort of up-titration and acclimation to a GLP-1 agonist that semaglutide requires, and transition between these agents seems feasible for at least some. It also means daily injections of liraglutide rather than the weekly schedule for semaglutide, although some patients prefer maintaining a daily dosing schedule. Another limitation of liraglutide is that evidence shows it is not nearly as effective for weight loss as semaglutide.
Results from the head-to-head STEP 8 trial, published in JAMA, showed an average weight loss from baseline of about 16% with semaglutide and about 6% with liraglutide (and about 2% with placebo).
A ‘reasonable’ evidence base, but more work
Changing from Saxenda to Wegovy, or from Wegovy to Saxenda, “would be reasonably evidence-based medicine,” said Dr. Oshman in an interview. She has managed a Wegovy-to-Saxenda switch for a “handful” of patients to deal with Wegovy shortages, but she has not yet moved anyone to Wegovy after a Saxenda initiation.
“No prospective study has looked at this transition,” but dose equivalence tables exist based on expert opinion, noted Dr. Oshman, as in this 2020 report.
Dr. Varney has several patients on the Saxenda-to-Wegovy track. She up-titrates patients on Saxenda to the maximum daily dose of 3.0 mg and then switches them to the 1.7 mg weekly dose of Wegovy, one of the “destination” Wegovy doses that has remained generally available during the shortage. But Dr. Varney’s experience is that only half of her patients made the changeover smoothly, with the others having “severe gastrointestinal distress,” including vomiting, she notes.
Dr. Fitch has also successfully used this Saxenda-to-Wegovy approach for some of her patients, but it hasn’t been easy.
“It’s more work and more prior authorizations. It’s harder and adds a layer of stress,” but, Dr. Fitch adds, “people are willing to work on it because the weight loss is worth it.”
The liraglutide to semaglutide shuffle is “doable,” says Dr. Rizo, “but I’m looking forward to not having to do it and being able to just start Wegovy.”
The tirzepatide coupon program works ‘off label’ for obesity
Another workaround depends on the FDA approval in May for tirzepatide (Mounjaro) for type 2 diabetes. Tirzepatide is a related GLP-1 agonist that also adds a second incretin-like agonist activity that mimics the glucose-dependent insulinotropic polypeptide.
Soon after approval, Lilly, the company that markets tirzepatide, started a U.S. coupon program geared exclusively to people with commercial insurance. Within certain refill and dollar limits, the program lets patients buy tirzepatide at pharmacies at an out-of-pocket cost of $25 for a 4-week supply (tirzepatide is also dosed by weekly subcutaneous injections). The program will extend into 2023.
Novo Nordisk offered U.S. patients with commercial insurance a similar discount when Wegovy first hit the U.S. market in 2021, but the program closed down once the supply shortage began.
Despite tirzepatide’s current approval only for type 2 diabetes, Dr. Varney has been successfully prescribing it to patients without diabetes off-label for weight loss.
“The coupons still work even when tirzepatide is used off-label,” she notes. And while the drug’s rollout is still only a couple of months old, so far, it’s gone “beautifully” with no hints of supply issues, she says.
But a major drawback to relying on an introductory coupon program that makes these agents affordable to patients is their ability to maintain treatment once the discounts inevitably end.
“We try to only prescribe agents that patients can continue to access,” says Dr. Fitch, who has had some patients with commercial insurance start on Wegovy with coupon discounts only to later lose access.
Many commercial U.S. insurers do not cover obesity treatments, a decision often driven by the employers who sponsor the coverage, she notes.
Study results have documented that when people with obesity stop taking a GLP-1 agonist their lost weight rebounds, as in a study that tracked people who stopped taking semaglutide.
Dr. Fitch has had success prescribing tirzepatide to patients with obesity but without diabetes who have certain types of Medicare drug coverage policies, which often do not deny off-label drug coverage. That approach works until patients reach the “donut hole” in their drug coverage and are faced with a certain level of out-of-pocket costs that can balloon to several thousand dollars.
Even more workarounds
Other approaches patients have used to acquire Wegovy include purchasing it in other countries, such as Canada or Brazil, says Dr. Fitch. But prices outside the United States, while substantially lower, can still be a barrier for many patients, notes Dr. Oshman.
Semaglutide in Canada goes for about $300 for a 4-week supply, roughly a quarter the U.S. price, she says, but is “still too high for many of my patients.”
Intense patient demand sometimes bordering on desperation has prompted some to seek semaglutide from private compounding pharmacies, a step clinicians regard as downright dangerous.
“Semaglutide from compounding pharmacies is not known to be safe. We feel strongly that it’s not something that people should do,” says Dr. Fitch.
“Compounding pharmacies have no FDA regulation. People don’t know what they’re getting. It’s dangerous,” agrees Dr. Varney. Physicians who refer people for privately compounded semaglutide “are taking advantage of desperate people,” she adds.
Although it seems likely that Novo Nordisk will soon sort out the supply problems and Wegovy will once again become more widely available, some of the issues patients have had with access to the weight loss medication stem from more systemic issues in the United States health insurance landscape: an unwillingness by payers to cover the costs of weight loss medications, a shortcoming that also exists for Medicare and Medicaid.
“We need to make obesity treatment a standard benefit, and not something that can be carved out,” says Dr. Fitch. People with obesity “deserve access to effective treatments for their disease,” she declares.
Dr. Oshman, Dr. Varney, and Dr. Rizo have reported no relevant financial relationships. Dr. Fitch has reported being an advisor to Jenny Craig.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) agonist semaglutide formulated for treating obesity (Wegovy) had a roaring takeoff a little more than a year ago, with surging patient demand after the U.S. Food and Drug Administration approved it in June 2021. But starting doses of the Wegovy form of semaglutide went missing in action starting late 2021 and continue to date, frustrating patients and their health care providers.
The arrival of Wegovy last year was hailed by obesity medicine specialists and others as a “game changer” for treating people with obesity because of semaglutide’s proven safety and efficacy at the subcutaneous dose of 2.4 mg delivered once a week to produce at least 15% weight loss in half the people who received it, as documented last year in results from one of the drug’s pivotal clinical trials.
But during the months following semaglutide’s approval for treating obesity (it also received an FDA marketing nod in late 2017 as Ozempic for treating type 2 diabetes), a worldwide shortage of Wegovy, including in the United States, emerged.
A manufacturing glitch shut down the primary location for production of U.S.-bound Wegovy injector pens for several months starting in late 2021, according to a December report from Novo Nordisk, the company that makes and markets the agent. (The Wegovy production issue appears to have had a very modest impact, especially in U.S. pharmacies, on the supply of semaglutide formulated as Ozempic, also marketed by Novo Nordisk, although Wegovy supply and demand have dramatically limited Ozempic availability in Australia.)
‘Unprecedented demand’ for Wegovy derailed when plant went offline
The supply side for Wegovy became so hopelessly broken that just months after U.S. sales began and immediately skyrocketed, Novo Nordisk made the remarkable decision to pull starting doses of Wegovy from the market to make it much harder to initiate patients (semaglutide and other GLP-1 agonists require gradual dose ramp-up to avoid gastrointestinal side effects), and the company publicly implored clinicians to not start new patients on the agent, which is where the status remains as of early August 2022.
Novo Nordisk’s financial report for the second quarter of 2022, released on Aug. 3, said the company “expects to make all Wegovy dose strengths available in the United States towards the end of 2022.”
A Dear Health Care Provider letter that Novo Nordisk posted on its U.S. Wegovy website last spring cited “unprecedented demand” that exceeded every prior product launch in the company’s history. It forced Novo Nordisk to pull the plug on all U.S. promotion of Wegovy and compelled the company to ask U.S. clinicians to halt new patient starts.
“I stopped offering Wegovy to new patients” since about the beginning of 2022, says Lauren D. Oshman, MD, a family and obesity medicine specialist at the University of Michigan, Ann Arbor. “It’s very frustrating to not have patients [with obesity] receive the optimal treatment available.” Although she adds that she tries to match obesity treatments to each patient’s clinical needs, and a GLP-1 agonist is not the first choice for every person with obesity.
“It was a disastrous rollout,” says Catherine W. Varney, DO, a family and obesity medicine specialist at the University of Virginia, Charlottesville. “It’s frustrating to know that the treatment is there but not being able to use it,” she said in an interview.
“I had about 800 patients on Wegovy” when the supply dropped earlier this year, and “I couldn’t handle the volume of messages that I got from patients,” recalls Angela Fitch, MD, associate director of the Massachusetts General Hospital Weight Center, Boston. “It was painful,” she said in an interview.
“Frustrating and chaotic,” is the description from Ivania M. Rizo, MD, director of obesity medicine at Boston Medical Center.
The liraglutide/Saxenda workaround
The upshot is that people with obesity and their health care providers have been busy devising workarounds to try to meet the intense demand for this drug-assisted approach to appetite control and weight loss. Their tactics run a wide gamut based on the crazy-quilt diversity of health insurance coverage across America.
Because the bottleneck for starting Wegovy resulted from unavailable starting doses (dosing starts at 0.25 mg delivered subcutaneously once a week, eventually ramping up to a maximum of 2.4 mg weekly), one option was to start patients on a different GLP-1 agonist, such as liraglutide (Saxenda, approved for obesity).
Starting a patient on liraglutide involves the same sort of up-titration and acclimation to a GLP-1 agonist that semaglutide requires, and transition between these agents seems feasible for at least some. It also means daily injections of liraglutide rather than the weekly schedule for semaglutide, although some patients prefer maintaining a daily dosing schedule. Another limitation of liraglutide is that evidence shows it is not nearly as effective for weight loss as semaglutide.
Results from the head-to-head STEP 8 trial, published in JAMA, showed an average weight loss from baseline of about 16% with semaglutide and about 6% with liraglutide (and about 2% with placebo).
A ‘reasonable’ evidence base, but more work
Changing from Saxenda to Wegovy, or from Wegovy to Saxenda, “would be reasonably evidence-based medicine,” said Dr. Oshman in an interview. She has managed a Wegovy-to-Saxenda switch for a “handful” of patients to deal with Wegovy shortages, but she has not yet moved anyone to Wegovy after a Saxenda initiation.
“No prospective study has looked at this transition,” but dose equivalence tables exist based on expert opinion, noted Dr. Oshman, as in this 2020 report.
Dr. Varney has several patients on the Saxenda-to-Wegovy track. She up-titrates patients on Saxenda to the maximum daily dose of 3.0 mg and then switches them to the 1.7 mg weekly dose of Wegovy, one of the “destination” Wegovy doses that has remained generally available during the shortage. But Dr. Varney’s experience is that only half of her patients made the changeover smoothly, with the others having “severe gastrointestinal distress,” including vomiting, she notes.
Dr. Fitch has also successfully used this Saxenda-to-Wegovy approach for some of her patients, but it hasn’t been easy.
“It’s more work and more prior authorizations. It’s harder and adds a layer of stress,” but, Dr. Fitch adds, “people are willing to work on it because the weight loss is worth it.”
The liraglutide to semaglutide shuffle is “doable,” says Dr. Rizo, “but I’m looking forward to not having to do it and being able to just start Wegovy.”
The tirzepatide coupon program works ‘off label’ for obesity
Another workaround depends on the FDA approval in May for tirzepatide (Mounjaro) for type 2 diabetes. Tirzepatide is a related GLP-1 agonist that also adds a second incretin-like agonist activity that mimics the glucose-dependent insulinotropic polypeptide.
Soon after approval, Lilly, the company that markets tirzepatide, started a U.S. coupon program geared exclusively to people with commercial insurance. Within certain refill and dollar limits, the program lets patients buy tirzepatide at pharmacies at an out-of-pocket cost of $25 for a 4-week supply (tirzepatide is also dosed by weekly subcutaneous injections). The program will extend into 2023.
Novo Nordisk offered U.S. patients with commercial insurance a similar discount when Wegovy first hit the U.S. market in 2021, but the program closed down once the supply shortage began.
Despite tirzepatide’s current approval only for type 2 diabetes, Dr. Varney has been successfully prescribing it to patients without diabetes off-label for weight loss.
“The coupons still work even when tirzepatide is used off-label,” she notes. And while the drug’s rollout is still only a couple of months old, so far, it’s gone “beautifully” with no hints of supply issues, she says.
But a major drawback to relying on an introductory coupon program that makes these agents affordable to patients is their ability to maintain treatment once the discounts inevitably end.
“We try to only prescribe agents that patients can continue to access,” says Dr. Fitch, who has had some patients with commercial insurance start on Wegovy with coupon discounts only to later lose access.
Many commercial U.S. insurers do not cover obesity treatments, a decision often driven by the employers who sponsor the coverage, she notes.
Study results have documented that when people with obesity stop taking a GLP-1 agonist their lost weight rebounds, as in a study that tracked people who stopped taking semaglutide.
Dr. Fitch has had success prescribing tirzepatide to patients with obesity but without diabetes who have certain types of Medicare drug coverage policies, which often do not deny off-label drug coverage. That approach works until patients reach the “donut hole” in their drug coverage and are faced with a certain level of out-of-pocket costs that can balloon to several thousand dollars.
Even more workarounds
Other approaches patients have used to acquire Wegovy include purchasing it in other countries, such as Canada or Brazil, says Dr. Fitch. But prices outside the United States, while substantially lower, can still be a barrier for many patients, notes Dr. Oshman.
Semaglutide in Canada goes for about $300 for a 4-week supply, roughly a quarter the U.S. price, she says, but is “still too high for many of my patients.”
Intense patient demand sometimes bordering on desperation has prompted some to seek semaglutide from private compounding pharmacies, a step clinicians regard as downright dangerous.
“Semaglutide from compounding pharmacies is not known to be safe. We feel strongly that it’s not something that people should do,” says Dr. Fitch.
“Compounding pharmacies have no FDA regulation. People don’t know what they’re getting. It’s dangerous,” agrees Dr. Varney. Physicians who refer people for privately compounded semaglutide “are taking advantage of desperate people,” she adds.
Although it seems likely that Novo Nordisk will soon sort out the supply problems and Wegovy will once again become more widely available, some of the issues patients have had with access to the weight loss medication stem from more systemic issues in the United States health insurance landscape: an unwillingness by payers to cover the costs of weight loss medications, a shortcoming that also exists for Medicare and Medicaid.
“We need to make obesity treatment a standard benefit, and not something that can be carved out,” says Dr. Fitch. People with obesity “deserve access to effective treatments for their disease,” she declares.
Dr. Oshman, Dr. Varney, and Dr. Rizo have reported no relevant financial relationships. Dr. Fitch has reported being an advisor to Jenny Craig.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) agonist semaglutide formulated for treating obesity (Wegovy) had a roaring takeoff a little more than a year ago, with surging patient demand after the U.S. Food and Drug Administration approved it in June 2021. But starting doses of the Wegovy form of semaglutide went missing in action starting late 2021 and continue to date, frustrating patients and their health care providers.
The arrival of Wegovy last year was hailed by obesity medicine specialists and others as a “game changer” for treating people with obesity because of semaglutide’s proven safety and efficacy at the subcutaneous dose of 2.4 mg delivered once a week to produce at least 15% weight loss in half the people who received it, as documented last year in results from one of the drug’s pivotal clinical trials.
But during the months following semaglutide’s approval for treating obesity (it also received an FDA marketing nod in late 2017 as Ozempic for treating type 2 diabetes), a worldwide shortage of Wegovy, including in the United States, emerged.
A manufacturing glitch shut down the primary location for production of U.S.-bound Wegovy injector pens for several months starting in late 2021, according to a December report from Novo Nordisk, the company that makes and markets the agent. (The Wegovy production issue appears to have had a very modest impact, especially in U.S. pharmacies, on the supply of semaglutide formulated as Ozempic, also marketed by Novo Nordisk, although Wegovy supply and demand have dramatically limited Ozempic availability in Australia.)
‘Unprecedented demand’ for Wegovy derailed when plant went offline
The supply side for Wegovy became so hopelessly broken that just months after U.S. sales began and immediately skyrocketed, Novo Nordisk made the remarkable decision to pull starting doses of Wegovy from the market to make it much harder to initiate patients (semaglutide and other GLP-1 agonists require gradual dose ramp-up to avoid gastrointestinal side effects), and the company publicly implored clinicians to not start new patients on the agent, which is where the status remains as of early August 2022.
Novo Nordisk’s financial report for the second quarter of 2022, released on Aug. 3, said the company “expects to make all Wegovy dose strengths available in the United States towards the end of 2022.”
A Dear Health Care Provider letter that Novo Nordisk posted on its U.S. Wegovy website last spring cited “unprecedented demand” that exceeded every prior product launch in the company’s history. It forced Novo Nordisk to pull the plug on all U.S. promotion of Wegovy and compelled the company to ask U.S. clinicians to halt new patient starts.
“I stopped offering Wegovy to new patients” since about the beginning of 2022, says Lauren D. Oshman, MD, a family and obesity medicine specialist at the University of Michigan, Ann Arbor. “It’s very frustrating to not have patients [with obesity] receive the optimal treatment available.” Although she adds that she tries to match obesity treatments to each patient’s clinical needs, and a GLP-1 agonist is not the first choice for every person with obesity.
“It was a disastrous rollout,” says Catherine W. Varney, DO, a family and obesity medicine specialist at the University of Virginia, Charlottesville. “It’s frustrating to know that the treatment is there but not being able to use it,” she said in an interview.
“I had about 800 patients on Wegovy” when the supply dropped earlier this year, and “I couldn’t handle the volume of messages that I got from patients,” recalls Angela Fitch, MD, associate director of the Massachusetts General Hospital Weight Center, Boston. “It was painful,” she said in an interview.
“Frustrating and chaotic,” is the description from Ivania M. Rizo, MD, director of obesity medicine at Boston Medical Center.
The liraglutide/Saxenda workaround
The upshot is that people with obesity and their health care providers have been busy devising workarounds to try to meet the intense demand for this drug-assisted approach to appetite control and weight loss. Their tactics run a wide gamut based on the crazy-quilt diversity of health insurance coverage across America.
Because the bottleneck for starting Wegovy resulted from unavailable starting doses (dosing starts at 0.25 mg delivered subcutaneously once a week, eventually ramping up to a maximum of 2.4 mg weekly), one option was to start patients on a different GLP-1 agonist, such as liraglutide (Saxenda, approved for obesity).
Starting a patient on liraglutide involves the same sort of up-titration and acclimation to a GLP-1 agonist that semaglutide requires, and transition between these agents seems feasible for at least some. It also means daily injections of liraglutide rather than the weekly schedule for semaglutide, although some patients prefer maintaining a daily dosing schedule. Another limitation of liraglutide is that evidence shows it is not nearly as effective for weight loss as semaglutide.
Results from the head-to-head STEP 8 trial, published in JAMA, showed an average weight loss from baseline of about 16% with semaglutide and about 6% with liraglutide (and about 2% with placebo).
A ‘reasonable’ evidence base, but more work
Changing from Saxenda to Wegovy, or from Wegovy to Saxenda, “would be reasonably evidence-based medicine,” said Dr. Oshman in an interview. She has managed a Wegovy-to-Saxenda switch for a “handful” of patients to deal with Wegovy shortages, but she has not yet moved anyone to Wegovy after a Saxenda initiation.
“No prospective study has looked at this transition,” but dose equivalence tables exist based on expert opinion, noted Dr. Oshman, as in this 2020 report.
Dr. Varney has several patients on the Saxenda-to-Wegovy track. She up-titrates patients on Saxenda to the maximum daily dose of 3.0 mg and then switches them to the 1.7 mg weekly dose of Wegovy, one of the “destination” Wegovy doses that has remained generally available during the shortage. But Dr. Varney’s experience is that only half of her patients made the changeover smoothly, with the others having “severe gastrointestinal distress,” including vomiting, she notes.
Dr. Fitch has also successfully used this Saxenda-to-Wegovy approach for some of her patients, but it hasn’t been easy.
“It’s more work and more prior authorizations. It’s harder and adds a layer of stress,” but, Dr. Fitch adds, “people are willing to work on it because the weight loss is worth it.”
The liraglutide to semaglutide shuffle is “doable,” says Dr. Rizo, “but I’m looking forward to not having to do it and being able to just start Wegovy.”
The tirzepatide coupon program works ‘off label’ for obesity
Another workaround depends on the FDA approval in May for tirzepatide (Mounjaro) for type 2 diabetes. Tirzepatide is a related GLP-1 agonist that also adds a second incretin-like agonist activity that mimics the glucose-dependent insulinotropic polypeptide.
Soon after approval, Lilly, the company that markets tirzepatide, started a U.S. coupon program geared exclusively to people with commercial insurance. Within certain refill and dollar limits, the program lets patients buy tirzepatide at pharmacies at an out-of-pocket cost of $25 for a 4-week supply (tirzepatide is also dosed by weekly subcutaneous injections). The program will extend into 2023.
Novo Nordisk offered U.S. patients with commercial insurance a similar discount when Wegovy first hit the U.S. market in 2021, but the program closed down once the supply shortage began.
Despite tirzepatide’s current approval only for type 2 diabetes, Dr. Varney has been successfully prescribing it to patients without diabetes off-label for weight loss.
“The coupons still work even when tirzepatide is used off-label,” she notes. And while the drug’s rollout is still only a couple of months old, so far, it’s gone “beautifully” with no hints of supply issues, she says.
But a major drawback to relying on an introductory coupon program that makes these agents affordable to patients is their ability to maintain treatment once the discounts inevitably end.
“We try to only prescribe agents that patients can continue to access,” says Dr. Fitch, who has had some patients with commercial insurance start on Wegovy with coupon discounts only to later lose access.
Many commercial U.S. insurers do not cover obesity treatments, a decision often driven by the employers who sponsor the coverage, she notes.
Study results have documented that when people with obesity stop taking a GLP-1 agonist their lost weight rebounds, as in a study that tracked people who stopped taking semaglutide.
Dr. Fitch has had success prescribing tirzepatide to patients with obesity but without diabetes who have certain types of Medicare drug coverage policies, which often do not deny off-label drug coverage. That approach works until patients reach the “donut hole” in their drug coverage and are faced with a certain level of out-of-pocket costs that can balloon to several thousand dollars.
Even more workarounds
Other approaches patients have used to acquire Wegovy include purchasing it in other countries, such as Canada or Brazil, says Dr. Fitch. But prices outside the United States, while substantially lower, can still be a barrier for many patients, notes Dr. Oshman.
Semaglutide in Canada goes for about $300 for a 4-week supply, roughly a quarter the U.S. price, she says, but is “still too high for many of my patients.”
Intense patient demand sometimes bordering on desperation has prompted some to seek semaglutide from private compounding pharmacies, a step clinicians regard as downright dangerous.
“Semaglutide from compounding pharmacies is not known to be safe. We feel strongly that it’s not something that people should do,” says Dr. Fitch.
“Compounding pharmacies have no FDA regulation. People don’t know what they’re getting. It’s dangerous,” agrees Dr. Varney. Physicians who refer people for privately compounded semaglutide “are taking advantage of desperate people,” she adds.
Although it seems likely that Novo Nordisk will soon sort out the supply problems and Wegovy will once again become more widely available, some of the issues patients have had with access to the weight loss medication stem from more systemic issues in the United States health insurance landscape: an unwillingness by payers to cover the costs of weight loss medications, a shortcoming that also exists for Medicare and Medicaid.
“We need to make obesity treatment a standard benefit, and not something that can be carved out,” says Dr. Fitch. People with obesity “deserve access to effective treatments for their disease,” she declares.
Dr. Oshman, Dr. Varney, and Dr. Rizo have reported no relevant financial relationships. Dr. Fitch has reported being an advisor to Jenny Craig.
A version of this article first appeared on Medscape.com.
Short walks after meals can cut diabetes risk
Taking a brief walk after eating can help lower the risk of type 2 diabetes, according to a recent study published in Sports Medicine (2022 Aug;52:1765-87).
Light walking after a meal – even for 2-5 minutes – can reduce blood sugar and insulin levels, the researchers found.
Blood sugar levels spike after eating, and the insulin produced to control them can lead to diabetes and cardiovascular issues, the researchers explained.
“With standing and walking, there are contractions of your muscles” that use glucose and lower blood sugar levels, Aidan Buffey, the lead study author and a PhD student in physical education and sport sciences at the University of Limerick (Ireland), told The Times.
“If you can do physical activity before the glucose peak, typically 60-90 minutes [after eating], that is when you’re going to have the benefit of not having the glucose spike,” he said.
Mr. Buffey and colleagues looked at seven studies to understand what would happen if you used standing or easy walking to interrupt prolonged sitting.
In five of the studies, none of the participants had prediabetes or type 2 diabetes. The other two studies included people with and without diabetes. The people in the studies were asked to either stand or walk for 2-5 minutes every 20-30 minutes over the course of a full day.
All seven studies showed that standing after a meal is better than sitting, and taking a short walk offered even better health benefits. Those who stood up for a short period of time after a meal had improved blood sugar levels but not insulin, while those who took a brief walk after a meal had lower blood sugar and insulin levels. Those who walked also had blood sugar levels that rose and fell more gradually, which is critical for managing diabetes.
Going for a walk, doing housework, or finding other ways to move your body within 60-90 minutes after eating could offer the best results, the study authors concluded.
These “mini-walks” could also be useful during the workday to break up prolonged periods of sitting at a desk.
“People are not going to get up and run on a treadmill or run around the office,” Mr. Buffey told The New York Times.
But making mini-walks a normal thing during the workday could be easy and acceptable at the office, he said. Even if people can’t take walks, standing up will help somewhat.
“Each small thing you do will have benefits, even if it is a small step,” Kershaw Patel, MD, a preventive cardiologist at Houston Methodist Hospital, told the newspaper. Dr. Patel wasn’t involved with the study.
“It’s a gradual effect of more activity, better health,” he said. “Each incremental step, each incremental stand or brisk walk appears to have a benefit.”
A version of this article first appeared on WebMD.com.
Taking a brief walk after eating can help lower the risk of type 2 diabetes, according to a recent study published in Sports Medicine (2022 Aug;52:1765-87).
Light walking after a meal – even for 2-5 minutes – can reduce blood sugar and insulin levels, the researchers found.
Blood sugar levels spike after eating, and the insulin produced to control them can lead to diabetes and cardiovascular issues, the researchers explained.
“With standing and walking, there are contractions of your muscles” that use glucose and lower blood sugar levels, Aidan Buffey, the lead study author and a PhD student in physical education and sport sciences at the University of Limerick (Ireland), told The Times.
“If you can do physical activity before the glucose peak, typically 60-90 minutes [after eating], that is when you’re going to have the benefit of not having the glucose spike,” he said.
Mr. Buffey and colleagues looked at seven studies to understand what would happen if you used standing or easy walking to interrupt prolonged sitting.
In five of the studies, none of the participants had prediabetes or type 2 diabetes. The other two studies included people with and without diabetes. The people in the studies were asked to either stand or walk for 2-5 minutes every 20-30 minutes over the course of a full day.
All seven studies showed that standing after a meal is better than sitting, and taking a short walk offered even better health benefits. Those who stood up for a short period of time after a meal had improved blood sugar levels but not insulin, while those who took a brief walk after a meal had lower blood sugar and insulin levels. Those who walked also had blood sugar levels that rose and fell more gradually, which is critical for managing diabetes.
Going for a walk, doing housework, or finding other ways to move your body within 60-90 minutes after eating could offer the best results, the study authors concluded.
These “mini-walks” could also be useful during the workday to break up prolonged periods of sitting at a desk.
“People are not going to get up and run on a treadmill or run around the office,” Mr. Buffey told The New York Times.
But making mini-walks a normal thing during the workday could be easy and acceptable at the office, he said. Even if people can’t take walks, standing up will help somewhat.
“Each small thing you do will have benefits, even if it is a small step,” Kershaw Patel, MD, a preventive cardiologist at Houston Methodist Hospital, told the newspaper. Dr. Patel wasn’t involved with the study.
“It’s a gradual effect of more activity, better health,” he said. “Each incremental step, each incremental stand or brisk walk appears to have a benefit.”
A version of this article first appeared on WebMD.com.
Taking a brief walk after eating can help lower the risk of type 2 diabetes, according to a recent study published in Sports Medicine (2022 Aug;52:1765-87).
Light walking after a meal – even for 2-5 minutes – can reduce blood sugar and insulin levels, the researchers found.
Blood sugar levels spike after eating, and the insulin produced to control them can lead to diabetes and cardiovascular issues, the researchers explained.
“With standing and walking, there are contractions of your muscles” that use glucose and lower blood sugar levels, Aidan Buffey, the lead study author and a PhD student in physical education and sport sciences at the University of Limerick (Ireland), told The Times.
“If you can do physical activity before the glucose peak, typically 60-90 minutes [after eating], that is when you’re going to have the benefit of not having the glucose spike,” he said.
Mr. Buffey and colleagues looked at seven studies to understand what would happen if you used standing or easy walking to interrupt prolonged sitting.
In five of the studies, none of the participants had prediabetes or type 2 diabetes. The other two studies included people with and without diabetes. The people in the studies were asked to either stand or walk for 2-5 minutes every 20-30 minutes over the course of a full day.
All seven studies showed that standing after a meal is better than sitting, and taking a short walk offered even better health benefits. Those who stood up for a short period of time after a meal had improved blood sugar levels but not insulin, while those who took a brief walk after a meal had lower blood sugar and insulin levels. Those who walked also had blood sugar levels that rose and fell more gradually, which is critical for managing diabetes.
Going for a walk, doing housework, or finding other ways to move your body within 60-90 minutes after eating could offer the best results, the study authors concluded.
These “mini-walks” could also be useful during the workday to break up prolonged periods of sitting at a desk.
“People are not going to get up and run on a treadmill or run around the office,” Mr. Buffey told The New York Times.
But making mini-walks a normal thing during the workday could be easy and acceptable at the office, he said. Even if people can’t take walks, standing up will help somewhat.
“Each small thing you do will have benefits, even if it is a small step,” Kershaw Patel, MD, a preventive cardiologist at Houston Methodist Hospital, told the newspaper. Dr. Patel wasn’t involved with the study.
“It’s a gradual effect of more activity, better health,” he said. “Each incremental step, each incremental stand or brisk walk appears to have a benefit.”
A version of this article first appeared on WebMD.com.
‘Staggering’ CVD rise projected in U.S., especially in minorities
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.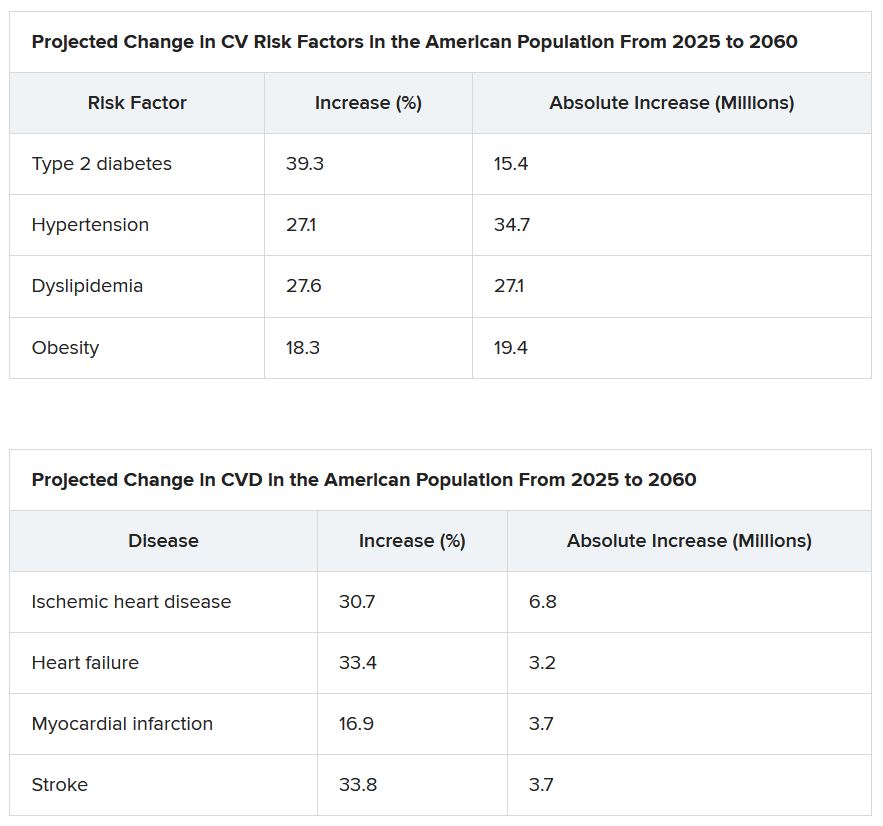
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF AMERICAN COLLEGE OF CARDIOLOGY
Commentary: Diabetes Drug Comparisons, August 2022
Meta-analyses of sodium-glucose cotransporter 2 inhibitor (SGLT2i) outcome trials have shown reductions in all-cause and cardiovascular mortality, but dipeptidyl peptidase 4 inhibitors (DPP4i) have been neutral for these outcomes. In a Taiwanese retrospective cohort study, Chung and colleagues compared 53,264 pairs of propensity-matched patients with type 2 diabetes who were treated with either an SGLT2i or DPP4i. They not only reported relative risk reductions of 34% and 32% for all-cause death and cardiovascular death, respectively, but also a reduction in cancer death of 27% and a reduction in noncancer, noncardiovascular death of 38%. Although limited by its retrospective, observational design, the finding of a benefit of SGLT2i treatment on both cancer death and noncancer, noncardiovascular death would benefit from further research.
In another large Taiwanese retrospective cohort study with propensity matching, Chan and colleagues compared patients treated with SGLT2i, DPP4i, and glucagon-like peptide 1 receptor agonists (GLP-1RA), with a main study outcome of new-onset atrial fibrillation (AF). They noted that SGLT2i treatment was associated with a 10% and 36% lower risk for AF compared with DPP4i and GLP-1RA treatment, respectively. These results are consistent with meta-analyses of SGLT2i outcome trials that have demonstrated reductions in AF with SGLT2i vs placebo. Perhaps it is time to recognize that another clinical benefit of the SGLT2i class is the reduction in risk for AF.
Although GLP-1RA have been linked to an increased risk for gallbladder-related events in many studies, there has been little data suggesting an increased risk with DPP4i. He and colleagues have published a pairwise meta-analysis of 82 randomized clinical trials and found that DPP4i compared with placebo or nonincretin drugs increased the risk for gallbladder or biliary diseases by 1.22-fold, with 11 more events per 10,000 person years. In a network meta-analysis of 184 randomized trials, they also found that DPP4i treatment increased the risk for gallbladder or biliary diseases compared with SGLT2i but not compared with GLP-1RA. This was the first meta-analysis to systematically study the association between DPP4i and gallbladder-related diseases. Although the absolute risk is small, clinicians need to be aware of this link and consider this adverse effect when deciding about the risks vs benefits of DPP4i treatment.
Individuals with obesity and prediabetes are at greater risk for type 2 diabetes. Trials of lifestyle modification and antiobesity agents have shown that restoration to normoglycemia can occur with weight loss. Phase 3A studies of the antiobesity agent semaglutide (2.4 mg/week) included 3375 individuals with prediabetes across three trials. In a post hoc analysis of these patients with prediabetes, Perreault and colleagues found that after 68 weeks of treatment, there was a much higher likelihood of normoglycemia with 2.4 mg/week of semaglutide compared with placebo. Though definite conclusions are limited owing to the post hoc nature of this analysis, these results make it very likely that the ongoing STEP 10 trial of 2.4 mg semaglutide vs placebo (201 participants with obesity and prediabetes) will probably show a significant benefit on the primary outcome of change to normoglycemia.
Meta-analyses of sodium-glucose cotransporter 2 inhibitor (SGLT2i) outcome trials have shown reductions in all-cause and cardiovascular mortality, but dipeptidyl peptidase 4 inhibitors (DPP4i) have been neutral for these outcomes. In a Taiwanese retrospective cohort study, Chung and colleagues compared 53,264 pairs of propensity-matched patients with type 2 diabetes who were treated with either an SGLT2i or DPP4i. They not only reported relative risk reductions of 34% and 32% for all-cause death and cardiovascular death, respectively, but also a reduction in cancer death of 27% and a reduction in noncancer, noncardiovascular death of 38%. Although limited by its retrospective, observational design, the finding of a benefit of SGLT2i treatment on both cancer death and noncancer, noncardiovascular death would benefit from further research.
In another large Taiwanese retrospective cohort study with propensity matching, Chan and colleagues compared patients treated with SGLT2i, DPP4i, and glucagon-like peptide 1 receptor agonists (GLP-1RA), with a main study outcome of new-onset atrial fibrillation (AF). They noted that SGLT2i treatment was associated with a 10% and 36% lower risk for AF compared with DPP4i and GLP-1RA treatment, respectively. These results are consistent with meta-analyses of SGLT2i outcome trials that have demonstrated reductions in AF with SGLT2i vs placebo. Perhaps it is time to recognize that another clinical benefit of the SGLT2i class is the reduction in risk for AF.
Although GLP-1RA have been linked to an increased risk for gallbladder-related events in many studies, there has been little data suggesting an increased risk with DPP4i. He and colleagues have published a pairwise meta-analysis of 82 randomized clinical trials and found that DPP4i compared with placebo or nonincretin drugs increased the risk for gallbladder or biliary diseases by 1.22-fold, with 11 more events per 10,000 person years. In a network meta-analysis of 184 randomized trials, they also found that DPP4i treatment increased the risk for gallbladder or biliary diseases compared with SGLT2i but not compared with GLP-1RA. This was the first meta-analysis to systematically study the association between DPP4i and gallbladder-related diseases. Although the absolute risk is small, clinicians need to be aware of this link and consider this adverse effect when deciding about the risks vs benefits of DPP4i treatment.
Individuals with obesity and prediabetes are at greater risk for type 2 diabetes. Trials of lifestyle modification and antiobesity agents have shown that restoration to normoglycemia can occur with weight loss. Phase 3A studies of the antiobesity agent semaglutide (2.4 mg/week) included 3375 individuals with prediabetes across three trials. In a post hoc analysis of these patients with prediabetes, Perreault and colleagues found that after 68 weeks of treatment, there was a much higher likelihood of normoglycemia with 2.4 mg/week of semaglutide compared with placebo. Though definite conclusions are limited owing to the post hoc nature of this analysis, these results make it very likely that the ongoing STEP 10 trial of 2.4 mg semaglutide vs placebo (201 participants with obesity and prediabetes) will probably show a significant benefit on the primary outcome of change to normoglycemia.
Meta-analyses of sodium-glucose cotransporter 2 inhibitor (SGLT2i) outcome trials have shown reductions in all-cause and cardiovascular mortality, but dipeptidyl peptidase 4 inhibitors (DPP4i) have been neutral for these outcomes. In a Taiwanese retrospective cohort study, Chung and colleagues compared 53,264 pairs of propensity-matched patients with type 2 diabetes who were treated with either an SGLT2i or DPP4i. They not only reported relative risk reductions of 34% and 32% for all-cause death and cardiovascular death, respectively, but also a reduction in cancer death of 27% and a reduction in noncancer, noncardiovascular death of 38%. Although limited by its retrospective, observational design, the finding of a benefit of SGLT2i treatment on both cancer death and noncancer, noncardiovascular death would benefit from further research.
In another large Taiwanese retrospective cohort study with propensity matching, Chan and colleagues compared patients treated with SGLT2i, DPP4i, and glucagon-like peptide 1 receptor agonists (GLP-1RA), with a main study outcome of new-onset atrial fibrillation (AF). They noted that SGLT2i treatment was associated with a 10% and 36% lower risk for AF compared with DPP4i and GLP-1RA treatment, respectively. These results are consistent with meta-analyses of SGLT2i outcome trials that have demonstrated reductions in AF with SGLT2i vs placebo. Perhaps it is time to recognize that another clinical benefit of the SGLT2i class is the reduction in risk for AF.
Although GLP-1RA have been linked to an increased risk for gallbladder-related events in many studies, there has been little data suggesting an increased risk with DPP4i. He and colleagues have published a pairwise meta-analysis of 82 randomized clinical trials and found that DPP4i compared with placebo or nonincretin drugs increased the risk for gallbladder or biliary diseases by 1.22-fold, with 11 more events per 10,000 person years. In a network meta-analysis of 184 randomized trials, they also found that DPP4i treatment increased the risk for gallbladder or biliary diseases compared with SGLT2i but not compared with GLP-1RA. This was the first meta-analysis to systematically study the association between DPP4i and gallbladder-related diseases. Although the absolute risk is small, clinicians need to be aware of this link and consider this adverse effect when deciding about the risks vs benefits of DPP4i treatment.
Individuals with obesity and prediabetes are at greater risk for type 2 diabetes. Trials of lifestyle modification and antiobesity agents have shown that restoration to normoglycemia can occur with weight loss. Phase 3A studies of the antiobesity agent semaglutide (2.4 mg/week) included 3375 individuals with prediabetes across three trials. In a post hoc analysis of these patients with prediabetes, Perreault and colleagues found that after 68 weeks of treatment, there was a much higher likelihood of normoglycemia with 2.4 mg/week of semaglutide compared with placebo. Though definite conclusions are limited owing to the post hoc nature of this analysis, these results make it very likely that the ongoing STEP 10 trial of 2.4 mg semaglutide vs placebo (201 participants with obesity and prediabetes) will probably show a significant benefit on the primary outcome of change to normoglycemia.
Serum 25-hydroxyvitamin D levels inversely correlated with albuminuria in patients with diabetes
Key clinical point: Serum level of 25-hydroxyvitamin D (25[OH]D) was inversely associated with albuminuria as an indicator of diabetic nephropathy in patients with type 2 diabetes mellitus (T2D), with the prevalence of macroalbuminuria increasing when the serum 25(OH)D level was <20 ng/mL.
Major finding: A strong negative association was observed between the serum 25(OH)D level and severity of albuminuria (r, −0.257; P < .001), with the prevalence of vitamin D deficiency being 61.8%, 33.3%, and 24.0% in patients with macroalbuminuria, microalbuminuria, and normoalbuminuria, respectively.
Study details: This was a cross-sectional study including 200 patients with T2D, of which 100, 66, and 34 had normoalbuminuria, microalbuminuria, and macroalbuminuria, respectively.
Disclosures: This study was supported by Shiraz University of Medical Sciences, Iran. The authors declared no competing interests.
Source: Zomorodian SA et al. Assessment of the relationship between 25-hydroxyvitamin D and albuminuria in type 2 diabetes mellitus. BMC Endocr Disord. 2022;22:171 (Jul 4). Doi: 10.1186/s12902-022-01088-2
Key clinical point: Serum level of 25-hydroxyvitamin D (25[OH]D) was inversely associated with albuminuria as an indicator of diabetic nephropathy in patients with type 2 diabetes mellitus (T2D), with the prevalence of macroalbuminuria increasing when the serum 25(OH)D level was <20 ng/mL.
Major finding: A strong negative association was observed between the serum 25(OH)D level and severity of albuminuria (r, −0.257; P < .001), with the prevalence of vitamin D deficiency being 61.8%, 33.3%, and 24.0% in patients with macroalbuminuria, microalbuminuria, and normoalbuminuria, respectively.
Study details: This was a cross-sectional study including 200 patients with T2D, of which 100, 66, and 34 had normoalbuminuria, microalbuminuria, and macroalbuminuria, respectively.
Disclosures: This study was supported by Shiraz University of Medical Sciences, Iran. The authors declared no competing interests.
Source: Zomorodian SA et al. Assessment of the relationship between 25-hydroxyvitamin D and albuminuria in type 2 diabetes mellitus. BMC Endocr Disord. 2022;22:171 (Jul 4). Doi: 10.1186/s12902-022-01088-2
Key clinical point: Serum level of 25-hydroxyvitamin D (25[OH]D) was inversely associated with albuminuria as an indicator of diabetic nephropathy in patients with type 2 diabetes mellitus (T2D), with the prevalence of macroalbuminuria increasing when the serum 25(OH)D level was <20 ng/mL.
Major finding: A strong negative association was observed between the serum 25(OH)D level and severity of albuminuria (r, −0.257; P < .001), with the prevalence of vitamin D deficiency being 61.8%, 33.3%, and 24.0% in patients with macroalbuminuria, microalbuminuria, and normoalbuminuria, respectively.
Study details: This was a cross-sectional study including 200 patients with T2D, of which 100, 66, and 34 had normoalbuminuria, microalbuminuria, and macroalbuminuria, respectively.
Disclosures: This study was supported by Shiraz University of Medical Sciences, Iran. The authors declared no competing interests.
Source: Zomorodian SA et al. Assessment of the relationship between 25-hydroxyvitamin D and albuminuria in type 2 diabetes mellitus. BMC Endocr Disord. 2022;22:171 (Jul 4). Doi: 10.1186/s12902-022-01088-2