User login
AAP adds specifics to policy on abusive head trauma
the American Academy of Pediatrics said in an updated policy statement.
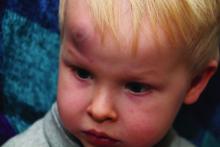
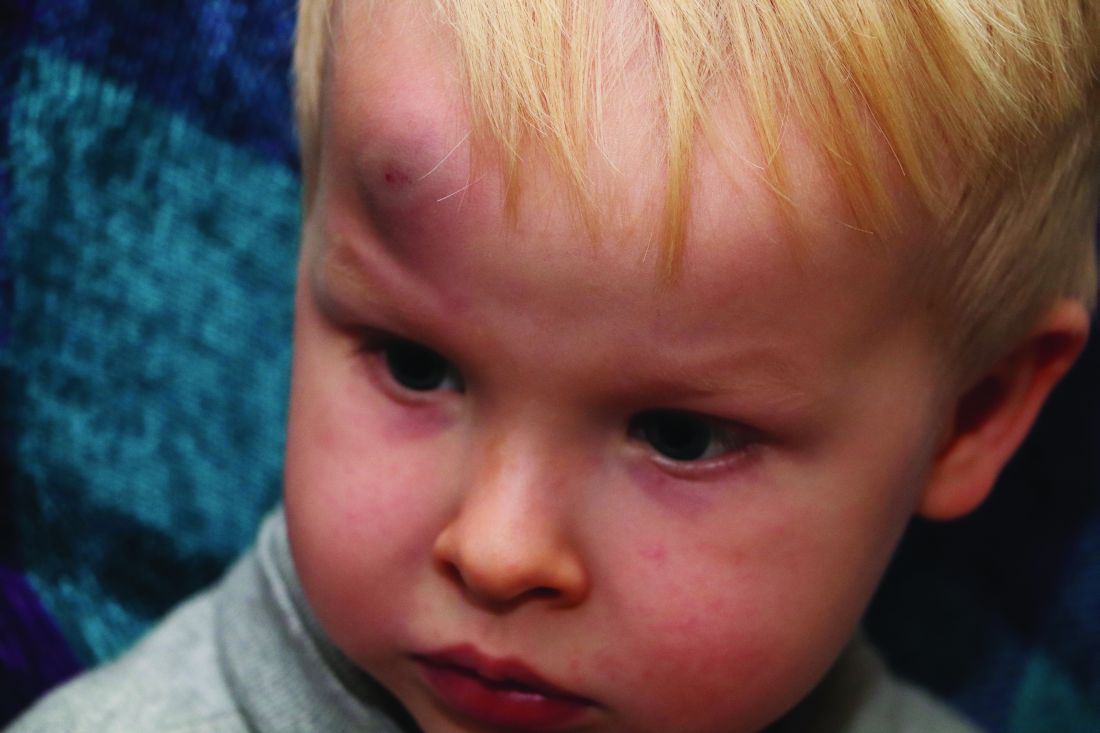
Abusive head trauma (AHT) is fatal in approximately one-quarter of cases in infants during the first year of life, and less-obvious clinical signs such as vomiting and fussiness often are missed, wrote Sandeep K. Narang, MD, JD, of Northwestern University, Chicago, and colleagues on the AAP Council on Child Abuse and Neglect.
In a policy statement published in Pediatrics, the AAP cautioned physicians to remain vigilant for signs that are common in AHT cases. In particular, bruising on the torso, ears, and neck in children aged younger than 4 years, or any bruising in infants younger than 4 months should be a red flag. In addition, the most recent data indicate that apnea and retinal hemorrhages are more common in cases of abuse than in accidental injuries. The AAP also recommends a skeletal survey in suspected AHT for children younger than 2 years to identify occult fractures.
“Oral injuries in infants, such as frenulum tears, may also accompany or precede AHT,” Dr. Narang and associates said.
In addition, secondary brain injury as a result of AHT can lead to poor outcomes that may be observed. “Almost 70% of survivors of AHT have some degree of lasting neurologic impairment, including static encephalopathy, intellectual disability, cerebral palsy, cortical blindness, seizure disorders, behavior problems, and learning disabilities,” according to the statement.
Endocrine dysfunction also is common in children with a history of AHT, but might not present until years later, the authors noted.
When AHT is suspected in a patient, the policy statement recommends that a subspecialist in child abuse pediatrics or in related areas including radiology, ophthalmology, neurosurgery, neurology, and general pediatric surgery “should also be consulted when necessary to ensure a complete and accurate evaluation.”
Although falls from a height of 1.5 m or 5 feet often are used as an explanation for AHT injuries, “numerous lines of clinical research have clarified the extreme rarity of short falls as a cause of severe neurologic injury or death in young infants,” Dr. Narang and associates wrote.
Other recommendations in the updated policy encourage use of the term “abusive head trauma” in medical communications, as well as encourage caregivers to serve as a medical home for survivors of AHT or refer them to medical homes for rehabilitation and monitoring. Parents and caregivers may need to be educated about the dangers of shaking or striking an infant, shown safe ways to manage a crying baby, and given tools to manage their own stress and frustration.
Physicians are legally required to report suspected cases of child abuse or neglect, and should be prepared to educate stakeholders if you are called on to work with legal and child protective services about the science behind AHT.
“The role of the pediatric practitioner is not to apportion blame or investigate potential criminal activity but to identify the medical problem, evaluate and treat the child’s injuries, and offer honest medical information to parents, families, investigators, and attorneys and/or judges,” Dr. Narang and associates wrote.
This policy statement updates the previous policy statement issued in 2009 and affirmed in 2013. The policy had no external funding, and the authors had no financial conflicts to disclose. Dr. Narang, Amanda Fingarson, DO, and James Lukefahr, MD, have served as paid expert witnesses/consultants in cases of abusive head trauma in infants and children.
SOURCE: Narang SK et al. Pediatrics. 2020 Mar 23. doi: 10.1542/peds.2020-0203.
the American Academy of Pediatrics said in an updated policy statement.
Abusive head trauma (AHT) is fatal in approximately one-quarter of cases in infants during the first year of life, and less-obvious clinical signs such as vomiting and fussiness often are missed, wrote Sandeep K. Narang, MD, JD, of Northwestern University, Chicago, and colleagues on the AAP Council on Child Abuse and Neglect.
In a policy statement published in Pediatrics, the AAP cautioned physicians to remain vigilant for signs that are common in AHT cases. In particular, bruising on the torso, ears, and neck in children aged younger than 4 years, or any bruising in infants younger than 4 months should be a red flag. In addition, the most recent data indicate that apnea and retinal hemorrhages are more common in cases of abuse than in accidental injuries. The AAP also recommends a skeletal survey in suspected AHT for children younger than 2 years to identify occult fractures.
“Oral injuries in infants, such as frenulum tears, may also accompany or precede AHT,” Dr. Narang and associates said.
In addition, secondary brain injury as a result of AHT can lead to poor outcomes that may be observed. “Almost 70% of survivors of AHT have some degree of lasting neurologic impairment, including static encephalopathy, intellectual disability, cerebral palsy, cortical blindness, seizure disorders, behavior problems, and learning disabilities,” according to the statement.
Endocrine dysfunction also is common in children with a history of AHT, but might not present until years later, the authors noted.
When AHT is suspected in a patient, the policy statement recommends that a subspecialist in child abuse pediatrics or in related areas including radiology, ophthalmology, neurosurgery, neurology, and general pediatric surgery “should also be consulted when necessary to ensure a complete and accurate evaluation.”
Although falls from a height of 1.5 m or 5 feet often are used as an explanation for AHT injuries, “numerous lines of clinical research have clarified the extreme rarity of short falls as a cause of severe neurologic injury or death in young infants,” Dr. Narang and associates wrote.
Other recommendations in the updated policy encourage use of the term “abusive head trauma” in medical communications, as well as encourage caregivers to serve as a medical home for survivors of AHT or refer them to medical homes for rehabilitation and monitoring. Parents and caregivers may need to be educated about the dangers of shaking or striking an infant, shown safe ways to manage a crying baby, and given tools to manage their own stress and frustration.
Physicians are legally required to report suspected cases of child abuse or neglect, and should be prepared to educate stakeholders if you are called on to work with legal and child protective services about the science behind AHT.
“The role of the pediatric practitioner is not to apportion blame or investigate potential criminal activity but to identify the medical problem, evaluate and treat the child’s injuries, and offer honest medical information to parents, families, investigators, and attorneys and/or judges,” Dr. Narang and associates wrote.
This policy statement updates the previous policy statement issued in 2009 and affirmed in 2013. The policy had no external funding, and the authors had no financial conflicts to disclose. Dr. Narang, Amanda Fingarson, DO, and James Lukefahr, MD, have served as paid expert witnesses/consultants in cases of abusive head trauma in infants and children.
SOURCE: Narang SK et al. Pediatrics. 2020 Mar 23. doi: 10.1542/peds.2020-0203.
the American Academy of Pediatrics said in an updated policy statement.
Abusive head trauma (AHT) is fatal in approximately one-quarter of cases in infants during the first year of life, and less-obvious clinical signs such as vomiting and fussiness often are missed, wrote Sandeep K. Narang, MD, JD, of Northwestern University, Chicago, and colleagues on the AAP Council on Child Abuse and Neglect.
In a policy statement published in Pediatrics, the AAP cautioned physicians to remain vigilant for signs that are common in AHT cases. In particular, bruising on the torso, ears, and neck in children aged younger than 4 years, or any bruising in infants younger than 4 months should be a red flag. In addition, the most recent data indicate that apnea and retinal hemorrhages are more common in cases of abuse than in accidental injuries. The AAP also recommends a skeletal survey in suspected AHT for children younger than 2 years to identify occult fractures.
“Oral injuries in infants, such as frenulum tears, may also accompany or precede AHT,” Dr. Narang and associates said.
In addition, secondary brain injury as a result of AHT can lead to poor outcomes that may be observed. “Almost 70% of survivors of AHT have some degree of lasting neurologic impairment, including static encephalopathy, intellectual disability, cerebral palsy, cortical blindness, seizure disorders, behavior problems, and learning disabilities,” according to the statement.
Endocrine dysfunction also is common in children with a history of AHT, but might not present until years later, the authors noted.
When AHT is suspected in a patient, the policy statement recommends that a subspecialist in child abuse pediatrics or in related areas including radiology, ophthalmology, neurosurgery, neurology, and general pediatric surgery “should also be consulted when necessary to ensure a complete and accurate evaluation.”
Although falls from a height of 1.5 m or 5 feet often are used as an explanation for AHT injuries, “numerous lines of clinical research have clarified the extreme rarity of short falls as a cause of severe neurologic injury or death in young infants,” Dr. Narang and associates wrote.
Other recommendations in the updated policy encourage use of the term “abusive head trauma” in medical communications, as well as encourage caregivers to serve as a medical home for survivors of AHT or refer them to medical homes for rehabilitation and monitoring. Parents and caregivers may need to be educated about the dangers of shaking or striking an infant, shown safe ways to manage a crying baby, and given tools to manage their own stress and frustration.
Physicians are legally required to report suspected cases of child abuse or neglect, and should be prepared to educate stakeholders if you are called on to work with legal and child protective services about the science behind AHT.
“The role of the pediatric practitioner is not to apportion blame or investigate potential criminal activity but to identify the medical problem, evaluate and treat the child’s injuries, and offer honest medical information to parents, families, investigators, and attorneys and/or judges,” Dr. Narang and associates wrote.
This policy statement updates the previous policy statement issued in 2009 and affirmed in 2013. The policy had no external funding, and the authors had no financial conflicts to disclose. Dr. Narang, Amanda Fingarson, DO, and James Lukefahr, MD, have served as paid expert witnesses/consultants in cases of abusive head trauma in infants and children.
SOURCE: Narang SK et al. Pediatrics. 2020 Mar 23. doi: 10.1542/peds.2020-0203.
FROM PEDIATRICS
Inactivated flu vaccine succeeds among autoimmune rheumatic disease patients
Use of the inactivated influenza vaccine by adults with autoimmune rheumatic diseases significantly reduced their risk of influenza-like illness, hospitalization for pneumonia and chronic obstructive pulmonary disease, and death from pneumonia, according to findings from an observational study of more than 30,000 patients in the U.K. Clinical Practice Research Datalink.
Although the inactivated vaccine has been recommended for patients with autoimmune rheumatic diseases (AIRDs), including rheumatoid arthritis and spondyloarthritis, the vaccine’s impact on patient outcomes including pneumonia, hospitalization, and death has not been well studied, wrote Georgina Nakafero, PhD, of the University of Nottingham, England, and colleagues.
In a study published in Rheumatology, the researchers identified 30,788 adults with AIRDs from the longitudinal Clinical Practice Research Datalink database in the United Kingdom. Of these, 66% were women, 76% had rheumatoid arthritis, and 61% had been prescribed methotrexate. The study included a total of 125,034 flu cycles between 2006 and 2009 and between 2010 and 2015.
Overall, vaccination with the inactivated influenza vaccine (IIV) reduced the risk of primary care consultation for influenza-like illness (adjusted odds ratio, 0.70), hospitalization for pneumonia (aOR, 0.61), exacerbation of chronic obstructive pulmonary disease (aOR, 0.67), and death caused by pneumonia (aOR, 0.48) in the study population. In a propensity score–adjusted analysis, only protection from influenza-like illness lost statistical significance.
In addition, vaccination was associated with a reduction in all-cause mortality among AIRDs patients, but restricting the outcomes to the active influenza periods may have confounded this result, the researchers said.
The study findings were limited by several factors including observational design, the use of a single vaccine efficacy estimate for each outcome, potential missed vaccination cycles, and potential confounding by indication and healthy user bias that could inflate the vaccine effectiveness, the researchers noted. However, the results were strengthened by the large sample size, including a range of AIRDs, and the use of both diagnostic and prescription codes, they said.
“The findings of this study, together with the results of our previous study demonstrating the safety of IIV in people with AIRDs, provides evidence to promote seasonal flu vaccination in this population,” they concluded. They still emphasized that randomized, controlled trials are needed for an assessment of vaccine efficacy.
The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
SOURCE: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
Use of the inactivated influenza vaccine by adults with autoimmune rheumatic diseases significantly reduced their risk of influenza-like illness, hospitalization for pneumonia and chronic obstructive pulmonary disease, and death from pneumonia, according to findings from an observational study of more than 30,000 patients in the U.K. Clinical Practice Research Datalink.
Although the inactivated vaccine has been recommended for patients with autoimmune rheumatic diseases (AIRDs), including rheumatoid arthritis and spondyloarthritis, the vaccine’s impact on patient outcomes including pneumonia, hospitalization, and death has not been well studied, wrote Georgina Nakafero, PhD, of the University of Nottingham, England, and colleagues.
In a study published in Rheumatology, the researchers identified 30,788 adults with AIRDs from the longitudinal Clinical Practice Research Datalink database in the United Kingdom. Of these, 66% were women, 76% had rheumatoid arthritis, and 61% had been prescribed methotrexate. The study included a total of 125,034 flu cycles between 2006 and 2009 and between 2010 and 2015.
Overall, vaccination with the inactivated influenza vaccine (IIV) reduced the risk of primary care consultation for influenza-like illness (adjusted odds ratio, 0.70), hospitalization for pneumonia (aOR, 0.61), exacerbation of chronic obstructive pulmonary disease (aOR, 0.67), and death caused by pneumonia (aOR, 0.48) in the study population. In a propensity score–adjusted analysis, only protection from influenza-like illness lost statistical significance.
In addition, vaccination was associated with a reduction in all-cause mortality among AIRDs patients, but restricting the outcomes to the active influenza periods may have confounded this result, the researchers said.
The study findings were limited by several factors including observational design, the use of a single vaccine efficacy estimate for each outcome, potential missed vaccination cycles, and potential confounding by indication and healthy user bias that could inflate the vaccine effectiveness, the researchers noted. However, the results were strengthened by the large sample size, including a range of AIRDs, and the use of both diagnostic and prescription codes, they said.
“The findings of this study, together with the results of our previous study demonstrating the safety of IIV in people with AIRDs, provides evidence to promote seasonal flu vaccination in this population,” they concluded. They still emphasized that randomized, controlled trials are needed for an assessment of vaccine efficacy.
The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
SOURCE: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
Use of the inactivated influenza vaccine by adults with autoimmune rheumatic diseases significantly reduced their risk of influenza-like illness, hospitalization for pneumonia and chronic obstructive pulmonary disease, and death from pneumonia, according to findings from an observational study of more than 30,000 patients in the U.K. Clinical Practice Research Datalink.
Although the inactivated vaccine has been recommended for patients with autoimmune rheumatic diseases (AIRDs), including rheumatoid arthritis and spondyloarthritis, the vaccine’s impact on patient outcomes including pneumonia, hospitalization, and death has not been well studied, wrote Georgina Nakafero, PhD, of the University of Nottingham, England, and colleagues.
In a study published in Rheumatology, the researchers identified 30,788 adults with AIRDs from the longitudinal Clinical Practice Research Datalink database in the United Kingdom. Of these, 66% were women, 76% had rheumatoid arthritis, and 61% had been prescribed methotrexate. The study included a total of 125,034 flu cycles between 2006 and 2009 and between 2010 and 2015.
Overall, vaccination with the inactivated influenza vaccine (IIV) reduced the risk of primary care consultation for influenza-like illness (adjusted odds ratio, 0.70), hospitalization for pneumonia (aOR, 0.61), exacerbation of chronic obstructive pulmonary disease (aOR, 0.67), and death caused by pneumonia (aOR, 0.48) in the study population. In a propensity score–adjusted analysis, only protection from influenza-like illness lost statistical significance.
In addition, vaccination was associated with a reduction in all-cause mortality among AIRDs patients, but restricting the outcomes to the active influenza periods may have confounded this result, the researchers said.
The study findings were limited by several factors including observational design, the use of a single vaccine efficacy estimate for each outcome, potential missed vaccination cycles, and potential confounding by indication and healthy user bias that could inflate the vaccine effectiveness, the researchers noted. However, the results were strengthened by the large sample size, including a range of AIRDs, and the use of both diagnostic and prescription codes, they said.
“The findings of this study, together with the results of our previous study demonstrating the safety of IIV in people with AIRDs, provides evidence to promote seasonal flu vaccination in this population,” they concluded. They still emphasized that randomized, controlled trials are needed for an assessment of vaccine efficacy.
The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
SOURCE: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
FROM RHEUMATOLOGY
Key clinical point: Adults with autoimmune rheumatic diseases who received the inactivated flu vaccine had lower rates of flu-like illness, hospitalization, and death than did those not vaccinated.
Major finding: Vaccination significantly reduced the risk of flu-like illness, hospitalization for pneumonia or COPD exacerbation, and death from pneumonia by 30%, 39%, 33%, and 52%, respectively.
Study details: The data come from 30,788 adults with AIRD and included 125,034 influenza cycles.
Disclosures: The study was supported by Versus Arthritis and the National Institute of Health Research. Lead author Dr. Nakafero had no financial conflicts to disclose. Several coauthors disclosed relationships with companies, including AstraZeneca, Roche, and Pfizer.
Source: Nakafero G et al. Rheumatology. 2020 Mar 11. doi: 10.1093/rheumatology/keaa078.
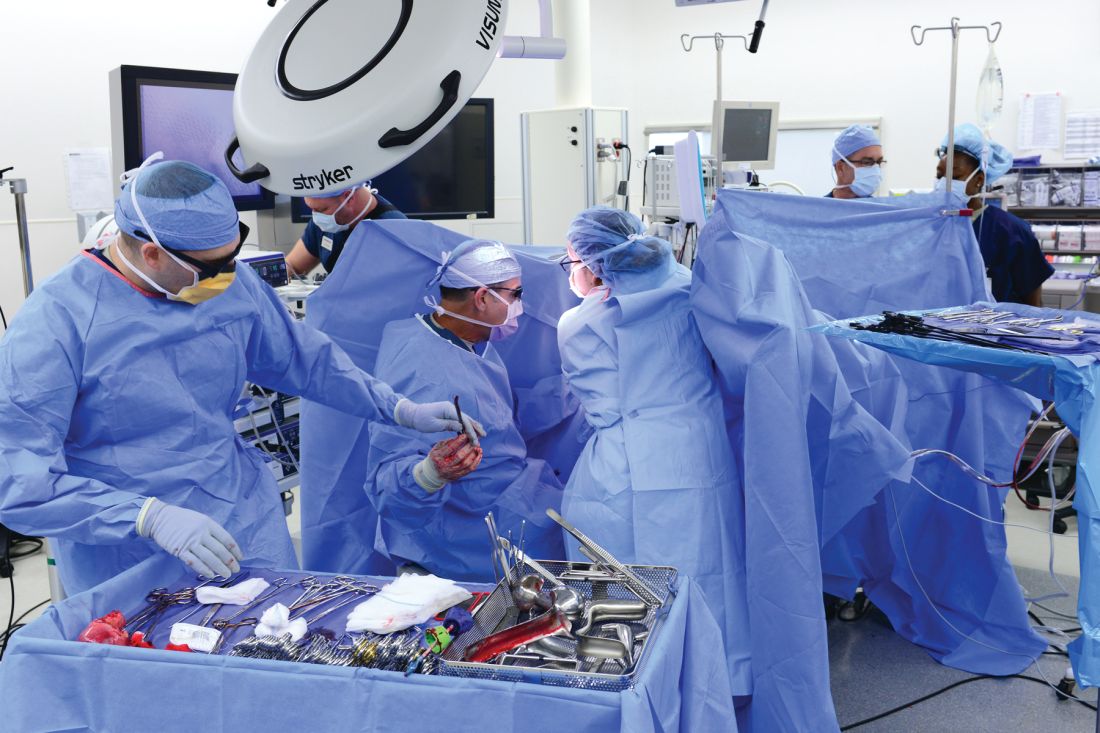
Prospective algorithm favors vaginal hysterectomy
based on data from a prospective study of 365 patients.
“Total vaginal hysterectomy is the most cost-effective route, with a low complication rate, and, therefore, should be performed when feasible,” wrote Jennifer J. Schmitt, DO, of the Mayo Clinic, Rochester, Minn., and colleagues.
However, algorithms to support the decision to choose vaginal hysterectomy are not widely used, they said.
To assess the optimal surgical route for hysterectomy, the researchers devised a prospective algorithm and decision tree based on history of laparotomy, uterine size, and vaginal access. The results of their study were published in Obstetrics & Gynecology.
The study population included 365 women aged 18 years and older who underwent hysterectomies between Nov. 24, 2015, and Dec. 31, 2017, at a single center. A total of 202 patients (55%) met criteria for a total vaginal hysterectomy using the algorithm, and 57 (15.6%) were assigned to have an examination under anesthesia followed by total vaginal hysterectomy, for a total of 259 expected vaginal hysterectomies. Ultimately, 211 (81.5%) of the patients identified as being the best candidates for having a vaginal hysterectomy underwent the procedure. Almost all of the procedures – 99.1% – were completed successfully.
The algorithm predicted that 52 patients were expected to have an examination under anesthesia followed by a robot-assisted total laparoscopic hysterectomy and 54 were expected to have an abdominal, robotic, or laparoscopic hysterectomy. A total of 46 procedures (44 robotic, when vaginal was expected and 2 abdominal, when vaginal was expected) deviated to a more invasive route than prescribed by the algorithm, and 7 procedures deviated from the algorithm-predicted robotic or abdominal procedure to total vaginal hysterectomy.
Approximately 95% of the patients were discharged within 24 hours of surgery. These patients included 7 who had vaginal surgery when a more invasive method was predicted and did not experience intraoperative complications or Accordion grade 3 complications.
“Prospective algorithm use predicts that 55.3% of all hysterectomies were expected to have an a priori total vaginal hysterectomy, which is higher than the actual total vaginal hysterectomy rate of 11.5% reported previously,” the researchers noted, and they added that vaginal hysterectomy would be associated with cost savings of $657,524 if the total hysterectomy rate was 55% instead of 11%.
The study findings were limited by several factors including an expertise bias at the center where the study was conducted, as well as the small number of patients with algorithm deviations or poor outcomes, and the lack of a control group, the researchers noted. However, the results support the use of the algorithm “in combination with educating gynecologic surgeons about the feasibility of vaginal surgery,” they said.
“Prospective use of this algorithm nationally may increase the rate of total vaginal hysterectomy and decrease health care delivery costs,” they concluded.
“The American College of Obstetricians and Gynecologists continues to recommend vaginal hysterectomy as the approach of choice whenever feasible, and although clinical evidence and societal endorsements support vaginal hysterectomy as a superior high-value modality, the rate of vaginal hysterectomy in the United States has continued to decline,” Arnold P. Advincula, MD, of Columbia University Medical Center, New York, wrote in an accompanying editorial.
Many variables beyond clinical will determine the optimal hysterectomy route, Dr. Advincula said.
“Although historical evidence demonstrates that vaginal hysterectomy is associated with better outcomes when compared with other approaches, a multitude of studies now exist that challenge this notion. Given the financial implications and overall costs of care with surgical complications and 30-day readmissions, experienced high-volume surgeons using all available routes have shown robotics to be the best surgical approach in terms of fewer postoperative complications and lowest 30-day readmission rates,” he noted. However, “one should not split hairs and subtly pit one minimally invasive option against another, but instead should work toward the goal of minimizing laparotomy, which is still performed at a high rate,” Dr. Advincula emphasized.
The study was supported in part by the National Center for Advancing Translational Science. Dr. Schmitt had no financial conflicts to disclose. Dr. Advincula disclosed serving as a consultant for AbbVie, Baxter, ConMed, Eximis Surgical, Intuitive Surgical, and Titan Medical, and performing consultancy work and receiving royalties from Cooper Surgical.
SOURCES: Schmitt JJ et al. Obstet Gynecol. 2020;135:761-9. doi: 10.1097/AOG.0000000000003725; Advincula A. Obstet Gynecol. 2020;135:759-60. doi: doi: 10.1097/AOG.0000000000003814.
based on data from a prospective study of 365 patients.
“Total vaginal hysterectomy is the most cost-effective route, with a low complication rate, and, therefore, should be performed when feasible,” wrote Jennifer J. Schmitt, DO, of the Mayo Clinic, Rochester, Minn., and colleagues.
However, algorithms to support the decision to choose vaginal hysterectomy are not widely used, they said.
To assess the optimal surgical route for hysterectomy, the researchers devised a prospective algorithm and decision tree based on history of laparotomy, uterine size, and vaginal access. The results of their study were published in Obstetrics & Gynecology.
The study population included 365 women aged 18 years and older who underwent hysterectomies between Nov. 24, 2015, and Dec. 31, 2017, at a single center. A total of 202 patients (55%) met criteria for a total vaginal hysterectomy using the algorithm, and 57 (15.6%) were assigned to have an examination under anesthesia followed by total vaginal hysterectomy, for a total of 259 expected vaginal hysterectomies. Ultimately, 211 (81.5%) of the patients identified as being the best candidates for having a vaginal hysterectomy underwent the procedure. Almost all of the procedures – 99.1% – were completed successfully.
The algorithm predicted that 52 patients were expected to have an examination under anesthesia followed by a robot-assisted total laparoscopic hysterectomy and 54 were expected to have an abdominal, robotic, or laparoscopic hysterectomy. A total of 46 procedures (44 robotic, when vaginal was expected and 2 abdominal, when vaginal was expected) deviated to a more invasive route than prescribed by the algorithm, and 7 procedures deviated from the algorithm-predicted robotic or abdominal procedure to total vaginal hysterectomy.
Approximately 95% of the patients were discharged within 24 hours of surgery. These patients included 7 who had vaginal surgery when a more invasive method was predicted and did not experience intraoperative complications or Accordion grade 3 complications.
“Prospective algorithm use predicts that 55.3% of all hysterectomies were expected to have an a priori total vaginal hysterectomy, which is higher than the actual total vaginal hysterectomy rate of 11.5% reported previously,” the researchers noted, and they added that vaginal hysterectomy would be associated with cost savings of $657,524 if the total hysterectomy rate was 55% instead of 11%.
The study findings were limited by several factors including an expertise bias at the center where the study was conducted, as well as the small number of patients with algorithm deviations or poor outcomes, and the lack of a control group, the researchers noted. However, the results support the use of the algorithm “in combination with educating gynecologic surgeons about the feasibility of vaginal surgery,” they said.
“Prospective use of this algorithm nationally may increase the rate of total vaginal hysterectomy and decrease health care delivery costs,” they concluded.
“The American College of Obstetricians and Gynecologists continues to recommend vaginal hysterectomy as the approach of choice whenever feasible, and although clinical evidence and societal endorsements support vaginal hysterectomy as a superior high-value modality, the rate of vaginal hysterectomy in the United States has continued to decline,” Arnold P. Advincula, MD, of Columbia University Medical Center, New York, wrote in an accompanying editorial.
Many variables beyond clinical will determine the optimal hysterectomy route, Dr. Advincula said.
“Although historical evidence demonstrates that vaginal hysterectomy is associated with better outcomes when compared with other approaches, a multitude of studies now exist that challenge this notion. Given the financial implications and overall costs of care with surgical complications and 30-day readmissions, experienced high-volume surgeons using all available routes have shown robotics to be the best surgical approach in terms of fewer postoperative complications and lowest 30-day readmission rates,” he noted. However, “one should not split hairs and subtly pit one minimally invasive option against another, but instead should work toward the goal of minimizing laparotomy, which is still performed at a high rate,” Dr. Advincula emphasized.
The study was supported in part by the National Center for Advancing Translational Science. Dr. Schmitt had no financial conflicts to disclose. Dr. Advincula disclosed serving as a consultant for AbbVie, Baxter, ConMed, Eximis Surgical, Intuitive Surgical, and Titan Medical, and performing consultancy work and receiving royalties from Cooper Surgical.
SOURCES: Schmitt JJ et al. Obstet Gynecol. 2020;135:761-9. doi: 10.1097/AOG.0000000000003725; Advincula A. Obstet Gynecol. 2020;135:759-60. doi: doi: 10.1097/AOG.0000000000003814.
based on data from a prospective study of 365 patients.
“Total vaginal hysterectomy is the most cost-effective route, with a low complication rate, and, therefore, should be performed when feasible,” wrote Jennifer J. Schmitt, DO, of the Mayo Clinic, Rochester, Minn., and colleagues.
However, algorithms to support the decision to choose vaginal hysterectomy are not widely used, they said.
To assess the optimal surgical route for hysterectomy, the researchers devised a prospective algorithm and decision tree based on history of laparotomy, uterine size, and vaginal access. The results of their study were published in Obstetrics & Gynecology.
The study population included 365 women aged 18 years and older who underwent hysterectomies between Nov. 24, 2015, and Dec. 31, 2017, at a single center. A total of 202 patients (55%) met criteria for a total vaginal hysterectomy using the algorithm, and 57 (15.6%) were assigned to have an examination under anesthesia followed by total vaginal hysterectomy, for a total of 259 expected vaginal hysterectomies. Ultimately, 211 (81.5%) of the patients identified as being the best candidates for having a vaginal hysterectomy underwent the procedure. Almost all of the procedures – 99.1% – were completed successfully.
The algorithm predicted that 52 patients were expected to have an examination under anesthesia followed by a robot-assisted total laparoscopic hysterectomy and 54 were expected to have an abdominal, robotic, or laparoscopic hysterectomy. A total of 46 procedures (44 robotic, when vaginal was expected and 2 abdominal, when vaginal was expected) deviated to a more invasive route than prescribed by the algorithm, and 7 procedures deviated from the algorithm-predicted robotic or abdominal procedure to total vaginal hysterectomy.
Approximately 95% of the patients were discharged within 24 hours of surgery. These patients included 7 who had vaginal surgery when a more invasive method was predicted and did not experience intraoperative complications or Accordion grade 3 complications.
“Prospective algorithm use predicts that 55.3% of all hysterectomies were expected to have an a priori total vaginal hysterectomy, which is higher than the actual total vaginal hysterectomy rate of 11.5% reported previously,” the researchers noted, and they added that vaginal hysterectomy would be associated with cost savings of $657,524 if the total hysterectomy rate was 55% instead of 11%.
The study findings were limited by several factors including an expertise bias at the center where the study was conducted, as well as the small number of patients with algorithm deviations or poor outcomes, and the lack of a control group, the researchers noted. However, the results support the use of the algorithm “in combination with educating gynecologic surgeons about the feasibility of vaginal surgery,” they said.
“Prospective use of this algorithm nationally may increase the rate of total vaginal hysterectomy and decrease health care delivery costs,” they concluded.
“The American College of Obstetricians and Gynecologists continues to recommend vaginal hysterectomy as the approach of choice whenever feasible, and although clinical evidence and societal endorsements support vaginal hysterectomy as a superior high-value modality, the rate of vaginal hysterectomy in the United States has continued to decline,” Arnold P. Advincula, MD, of Columbia University Medical Center, New York, wrote in an accompanying editorial.
Many variables beyond clinical will determine the optimal hysterectomy route, Dr. Advincula said.
“Although historical evidence demonstrates that vaginal hysterectomy is associated with better outcomes when compared with other approaches, a multitude of studies now exist that challenge this notion. Given the financial implications and overall costs of care with surgical complications and 30-day readmissions, experienced high-volume surgeons using all available routes have shown robotics to be the best surgical approach in terms of fewer postoperative complications and lowest 30-day readmission rates,” he noted. However, “one should not split hairs and subtly pit one minimally invasive option against another, but instead should work toward the goal of minimizing laparotomy, which is still performed at a high rate,” Dr. Advincula emphasized.
The study was supported in part by the National Center for Advancing Translational Science. Dr. Schmitt had no financial conflicts to disclose. Dr. Advincula disclosed serving as a consultant for AbbVie, Baxter, ConMed, Eximis Surgical, Intuitive Surgical, and Titan Medical, and performing consultancy work and receiving royalties from Cooper Surgical.
SOURCES: Schmitt JJ et al. Obstet Gynecol. 2020;135:761-9. doi: 10.1097/AOG.0000000000003725; Advincula A. Obstet Gynecol. 2020;135:759-60. doi: doi: 10.1097/AOG.0000000000003814.
FROM OBSTETRICS & GYNECOLOGY
Reduced TNFi dose does not maintain axial spondyloarthritis improvement
Patients with axial spondyloarthritis have a significantly lower likelihood of achieving improvement in disease activity or remission when their dose of tumor necrosis factor inhibitor therapy is reduced, based on a systematic review and meta-analysis of six trials that included 747 adults.
Tumor necrosis factor inhibitors (TNFi) “have shown significant sustained clinical improvement in axSpA and are introduced in patients with axial disease or as the next line of treatment after inadequate response to nonsteroidal anti-inflammatory drugs,” but this improvement comes with a degree of immunosuppression that can increase infection risk, wrote Daeria O. Lawson of Toronto Western Hospital and colleagues. However, the impact of reducing or discontinuing TNFi therapy, compared with standard dosing, has not been well examined, they said.
In a study published in Arthritis Care & Research, the investigators identified six randomized, controlled trials with a total of 747 adults. Overall, patients on a reduced dose had a lower likelihood of achieving 40% improvement in Assessment of SpondyloArthritis international Society response criteria (ASAS40) or ASAS partial remission, compared with those on a standard TNFi dose (risk ratios, 0.62 and 0.17, respectively).
In addition, the mean increase in the Bath Ankylosing Spondylitis Disease Activity Index score was 0.35 for patients on reduced TNFi therapy, and no differences were seen in C-reactive protein levels, infection rates, or injection/infusion reactions in patients on a reduced dose, compared with those on the standard dose.
Patients on the reduced TNFi dose also had more disease flares and/or relapses, compared with the standard group (risk ratio, 1.73).
The study findings were limited by several factors including the inability to compare subgroups based on dosing regimens, potential blinding and selection bias, and inadequate data to assess certain patient outcomes, including maintenance of disease remission and quality of life, the researchers noted. The results confirm findings from previous studies and support the benefit of standard dosing for maintaining stable disease, they said.
However, more research is needed to identify patients who may be more responsive to TNFi reduction, they wrote. “Although treatment recommendations for the best dose reduction strategies cannot be made at this time given the heterogeneity in tapering strategies reported in the literature, this decision should be an individualized one between the patient and their physician,” the researchers emphasized.
The study received no outside funding. Dr. Lawson is supported in part by the Ontario Drug Policy Research Network Student Training Program.
SOURCE: Lawson DO et al. Arthritis Care Res. 2020 Mar 12. doi: 10.1002/ACR.24184.
Patients with axial spondyloarthritis have a significantly lower likelihood of achieving improvement in disease activity or remission when their dose of tumor necrosis factor inhibitor therapy is reduced, based on a systematic review and meta-analysis of six trials that included 747 adults.
Tumor necrosis factor inhibitors (TNFi) “have shown significant sustained clinical improvement in axSpA and are introduced in patients with axial disease or as the next line of treatment after inadequate response to nonsteroidal anti-inflammatory drugs,” but this improvement comes with a degree of immunosuppression that can increase infection risk, wrote Daeria O. Lawson of Toronto Western Hospital and colleagues. However, the impact of reducing or discontinuing TNFi therapy, compared with standard dosing, has not been well examined, they said.
In a study published in Arthritis Care & Research, the investigators identified six randomized, controlled trials with a total of 747 adults. Overall, patients on a reduced dose had a lower likelihood of achieving 40% improvement in Assessment of SpondyloArthritis international Society response criteria (ASAS40) or ASAS partial remission, compared with those on a standard TNFi dose (risk ratios, 0.62 and 0.17, respectively).
In addition, the mean increase in the Bath Ankylosing Spondylitis Disease Activity Index score was 0.35 for patients on reduced TNFi therapy, and no differences were seen in C-reactive protein levels, infection rates, or injection/infusion reactions in patients on a reduced dose, compared with those on the standard dose.
Patients on the reduced TNFi dose also had more disease flares and/or relapses, compared with the standard group (risk ratio, 1.73).
The study findings were limited by several factors including the inability to compare subgroups based on dosing regimens, potential blinding and selection bias, and inadequate data to assess certain patient outcomes, including maintenance of disease remission and quality of life, the researchers noted. The results confirm findings from previous studies and support the benefit of standard dosing for maintaining stable disease, they said.
However, more research is needed to identify patients who may be more responsive to TNFi reduction, they wrote. “Although treatment recommendations for the best dose reduction strategies cannot be made at this time given the heterogeneity in tapering strategies reported in the literature, this decision should be an individualized one between the patient and their physician,” the researchers emphasized.
The study received no outside funding. Dr. Lawson is supported in part by the Ontario Drug Policy Research Network Student Training Program.
SOURCE: Lawson DO et al. Arthritis Care Res. 2020 Mar 12. doi: 10.1002/ACR.24184.
Patients with axial spondyloarthritis have a significantly lower likelihood of achieving improvement in disease activity or remission when their dose of tumor necrosis factor inhibitor therapy is reduced, based on a systematic review and meta-analysis of six trials that included 747 adults.
Tumor necrosis factor inhibitors (TNFi) “have shown significant sustained clinical improvement in axSpA and are introduced in patients with axial disease or as the next line of treatment after inadequate response to nonsteroidal anti-inflammatory drugs,” but this improvement comes with a degree of immunosuppression that can increase infection risk, wrote Daeria O. Lawson of Toronto Western Hospital and colleagues. However, the impact of reducing or discontinuing TNFi therapy, compared with standard dosing, has not been well examined, they said.
In a study published in Arthritis Care & Research, the investigators identified six randomized, controlled trials with a total of 747 adults. Overall, patients on a reduced dose had a lower likelihood of achieving 40% improvement in Assessment of SpondyloArthritis international Society response criteria (ASAS40) or ASAS partial remission, compared with those on a standard TNFi dose (risk ratios, 0.62 and 0.17, respectively).
In addition, the mean increase in the Bath Ankylosing Spondylitis Disease Activity Index score was 0.35 for patients on reduced TNFi therapy, and no differences were seen in C-reactive protein levels, infection rates, or injection/infusion reactions in patients on a reduced dose, compared with those on the standard dose.
Patients on the reduced TNFi dose also had more disease flares and/or relapses, compared with the standard group (risk ratio, 1.73).
The study findings were limited by several factors including the inability to compare subgroups based on dosing regimens, potential blinding and selection bias, and inadequate data to assess certain patient outcomes, including maintenance of disease remission and quality of life, the researchers noted. The results confirm findings from previous studies and support the benefit of standard dosing for maintaining stable disease, they said.
However, more research is needed to identify patients who may be more responsive to TNFi reduction, they wrote. “Although treatment recommendations for the best dose reduction strategies cannot be made at this time given the heterogeneity in tapering strategies reported in the literature, this decision should be an individualized one between the patient and their physician,” the researchers emphasized.
The study received no outside funding. Dr. Lawson is supported in part by the Ontario Drug Policy Research Network Student Training Program.
SOURCE: Lawson DO et al. Arthritis Care Res. 2020 Mar 12. doi: 10.1002/ACR.24184.
FROM ARTHRITIS CARE & RESEARCH
Patients accept artificial intelligence in skin cancer screening
In a small survey, 75% of dermatology patients said they would recommend the use of artificial intelligence (AI) for skin cancer screening to friends and family members, but 94% emphasized the need for symbiosis between doctors, patients, and AI.
AI under investigation in dermatology includes both direct-to-patient and clinician decision-support AI tools for skin cancer screening, but patients’ perceptions of AI in health care remains unclear, Caroline A. Nelson, MD, of Yale University in New Haven, Conn., and colleagues wrote in JAMA Dermatology.
“We sought to elucidate perceived benefits and risks, strengths and weaknesses, implementation, response to conflict between human and AI clinical decision making, and recommendation for or against AI,” the researchers wrote.
They identified 48 patients seen from May 6, 2019, to July 8, 2019, at general dermatology clinics and melanoma clinics. This included 16 patients with a history of melanoma, 16 with a history of nonmelanoma skin cancer, and 16 with no history of skin cancer. The average age of the patients was 53.3 years, 54% were women, and 94% were white.
The researchers interviewed 24 patients about a direct-to-patient AI tool and 24 patients about a clinician decision-support AI tool.
Overall, 36 patients (75%) said they would recommend the AI tool to family and friends, with 17 patients (71%) saying they would recommend the direct-to-patient tool and 19 (79%) saying they would recommend the clinician decision-support tool. Another nine patients (19%) were ambivalent about the AI tools, and three patients (6%) said they would not recommend the tools.
Diagnostic speed and health care access were the most common perceived benefits of AI (by 60% of patients for each), and increased patient anxiety was the most common perceived risk (by 40% of patients). In addition, 69% of patients perceived more accurate diagnosis to be the greatest strength of an AI tool, and 85% perceived less accurate diagnosis to be the greatest weakness.
The study findings were limited by several factors, including the small sample size, qualitative design, use of a hypothetical rather than real-world situation, and a homogeneous study population, the researchers noted. However, the results merit more studies to obtain perspectives from diverse populations, they said.
“This expansion is particularly important in light of concerns raised that AI tools may exacerbate health care disparities in dermatology,” the researchers wrote.
From the patient perspective, the use of AI “may improve health care quality but should be implemented in a manner that preserves the integrity of the human physician-patient relationship,” the authors concluded.
“Although AI technology has not been widely implemented in dermatology yet, it is the pivotal time to assess patients’ views on the subject to understand their knowledge base, as well as values, preferences, and concerns regarding AI,” wrote Carrie L. Kovarik, MD, of the University of Pennsylvania in Philadelphia, in an accompanying editorial.
“Vulnerable patients, including racial and ethnic minorities, the underinsured or uninsured, economically disadvantaged, and those with chronic health conditions, may be at risk for improper consent for or use of AI,” she wrote.
Dr. Kovarik cited the position statement on augmented intelligence from the American Academy of Dermatology, which states that, for both patients and clinicians, “there should be transparency and choice on how their medical information is gathered, utilized, and stored and when, what, and how augmented intelligence technologies are utilized in their care process. There should be clarity in the symbiotic and synergistic roles of augmented intelligence and human judgment so that it is clear to the patient and provider when and how this technology is utilized to augment human judgment and interpretation.”
Clinicians will need to understand the perspectives on AI from patients of a range of backgrounds to achieve this goal, Dr. Kovarik said.
Dr. Nelson had no financial conflicts to disclose, but her colleagues disclosed relationships with pharmaceutical companies, government agencies, and nonprofit organizations. Dr. Kovarik disclosed serving on the artificial intelligence task force for the American Academy of Dermatology.
SOURCES: Nelson CA et al. JAMA Dermatol. 2020 Mar 11. doi: 10.1001/jamadermatol.2019.5014; Kovarik CL. JAMA Dermatol. 2020 Mar 11. doi: 10.1001/jamadermatol.2019.5013.
In a small survey, 75% of dermatology patients said they would recommend the use of artificial intelligence (AI) for skin cancer screening to friends and family members, but 94% emphasized the need for symbiosis between doctors, patients, and AI.
AI under investigation in dermatology includes both direct-to-patient and clinician decision-support AI tools for skin cancer screening, but patients’ perceptions of AI in health care remains unclear, Caroline A. Nelson, MD, of Yale University in New Haven, Conn., and colleagues wrote in JAMA Dermatology.
“We sought to elucidate perceived benefits and risks, strengths and weaknesses, implementation, response to conflict between human and AI clinical decision making, and recommendation for or against AI,” the researchers wrote.
They identified 48 patients seen from May 6, 2019, to July 8, 2019, at general dermatology clinics and melanoma clinics. This included 16 patients with a history of melanoma, 16 with a history of nonmelanoma skin cancer, and 16 with no history of skin cancer. The average age of the patients was 53.3 years, 54% were women, and 94% were white.
The researchers interviewed 24 patients about a direct-to-patient AI tool and 24 patients about a clinician decision-support AI tool.
Overall, 36 patients (75%) said they would recommend the AI tool to family and friends, with 17 patients (71%) saying they would recommend the direct-to-patient tool and 19 (79%) saying they would recommend the clinician decision-support tool. Another nine patients (19%) were ambivalent about the AI tools, and three patients (6%) said they would not recommend the tools.
Diagnostic speed and health care access were the most common perceived benefits of AI (by 60% of patients for each), and increased patient anxiety was the most common perceived risk (by 40% of patients). In addition, 69% of patients perceived more accurate diagnosis to be the greatest strength of an AI tool, and 85% perceived less accurate diagnosis to be the greatest weakness.
The study findings were limited by several factors, including the small sample size, qualitative design, use of a hypothetical rather than real-world situation, and a homogeneous study population, the researchers noted. However, the results merit more studies to obtain perspectives from diverse populations, they said.
“This expansion is particularly important in light of concerns raised that AI tools may exacerbate health care disparities in dermatology,” the researchers wrote.
From the patient perspective, the use of AI “may improve health care quality but should be implemented in a manner that preserves the integrity of the human physician-patient relationship,” the authors concluded.
“Although AI technology has not been widely implemented in dermatology yet, it is the pivotal time to assess patients’ views on the subject to understand their knowledge base, as well as values, preferences, and concerns regarding AI,” wrote Carrie L. Kovarik, MD, of the University of Pennsylvania in Philadelphia, in an accompanying editorial.
“Vulnerable patients, including racial and ethnic minorities, the underinsured or uninsured, economically disadvantaged, and those with chronic health conditions, may be at risk for improper consent for or use of AI,” she wrote.
Dr. Kovarik cited the position statement on augmented intelligence from the American Academy of Dermatology, which states that, for both patients and clinicians, “there should be transparency and choice on how their medical information is gathered, utilized, and stored and when, what, and how augmented intelligence technologies are utilized in their care process. There should be clarity in the symbiotic and synergistic roles of augmented intelligence and human judgment so that it is clear to the patient and provider when and how this technology is utilized to augment human judgment and interpretation.”
Clinicians will need to understand the perspectives on AI from patients of a range of backgrounds to achieve this goal, Dr. Kovarik said.
Dr. Nelson had no financial conflicts to disclose, but her colleagues disclosed relationships with pharmaceutical companies, government agencies, and nonprofit organizations. Dr. Kovarik disclosed serving on the artificial intelligence task force for the American Academy of Dermatology.
SOURCES: Nelson CA et al. JAMA Dermatol. 2020 Mar 11. doi: 10.1001/jamadermatol.2019.5014; Kovarik CL. JAMA Dermatol. 2020 Mar 11. doi: 10.1001/jamadermatol.2019.5013.
In a small survey, 75% of dermatology patients said they would recommend the use of artificial intelligence (AI) for skin cancer screening to friends and family members, but 94% emphasized the need for symbiosis between doctors, patients, and AI.
AI under investigation in dermatology includes both direct-to-patient and clinician decision-support AI tools for skin cancer screening, but patients’ perceptions of AI in health care remains unclear, Caroline A. Nelson, MD, of Yale University in New Haven, Conn., and colleagues wrote in JAMA Dermatology.
“We sought to elucidate perceived benefits and risks, strengths and weaknesses, implementation, response to conflict between human and AI clinical decision making, and recommendation for or against AI,” the researchers wrote.
They identified 48 patients seen from May 6, 2019, to July 8, 2019, at general dermatology clinics and melanoma clinics. This included 16 patients with a history of melanoma, 16 with a history of nonmelanoma skin cancer, and 16 with no history of skin cancer. The average age of the patients was 53.3 years, 54% were women, and 94% were white.
The researchers interviewed 24 patients about a direct-to-patient AI tool and 24 patients about a clinician decision-support AI tool.
Overall, 36 patients (75%) said they would recommend the AI tool to family and friends, with 17 patients (71%) saying they would recommend the direct-to-patient tool and 19 (79%) saying they would recommend the clinician decision-support tool. Another nine patients (19%) were ambivalent about the AI tools, and three patients (6%) said they would not recommend the tools.
Diagnostic speed and health care access were the most common perceived benefits of AI (by 60% of patients for each), and increased patient anxiety was the most common perceived risk (by 40% of patients). In addition, 69% of patients perceived more accurate diagnosis to be the greatest strength of an AI tool, and 85% perceived less accurate diagnosis to be the greatest weakness.
The study findings were limited by several factors, including the small sample size, qualitative design, use of a hypothetical rather than real-world situation, and a homogeneous study population, the researchers noted. However, the results merit more studies to obtain perspectives from diverse populations, they said.
“This expansion is particularly important in light of concerns raised that AI tools may exacerbate health care disparities in dermatology,” the researchers wrote.
From the patient perspective, the use of AI “may improve health care quality but should be implemented in a manner that preserves the integrity of the human physician-patient relationship,” the authors concluded.
“Although AI technology has not been widely implemented in dermatology yet, it is the pivotal time to assess patients’ views on the subject to understand their knowledge base, as well as values, preferences, and concerns regarding AI,” wrote Carrie L. Kovarik, MD, of the University of Pennsylvania in Philadelphia, in an accompanying editorial.
“Vulnerable patients, including racial and ethnic minorities, the underinsured or uninsured, economically disadvantaged, and those with chronic health conditions, may be at risk for improper consent for or use of AI,” she wrote.
Dr. Kovarik cited the position statement on augmented intelligence from the American Academy of Dermatology, which states that, for both patients and clinicians, “there should be transparency and choice on how their medical information is gathered, utilized, and stored and when, what, and how augmented intelligence technologies are utilized in their care process. There should be clarity in the symbiotic and synergistic roles of augmented intelligence and human judgment so that it is clear to the patient and provider when and how this technology is utilized to augment human judgment and interpretation.”
Clinicians will need to understand the perspectives on AI from patients of a range of backgrounds to achieve this goal, Dr. Kovarik said.
Dr. Nelson had no financial conflicts to disclose, but her colleagues disclosed relationships with pharmaceutical companies, government agencies, and nonprofit organizations. Dr. Kovarik disclosed serving on the artificial intelligence task force for the American Academy of Dermatology.
SOURCES: Nelson CA et al. JAMA Dermatol. 2020 Mar 11. doi: 10.1001/jamadermatol.2019.5014; Kovarik CL. JAMA Dermatol. 2020 Mar 11. doi: 10.1001/jamadermatol.2019.5013.
FROM JAMA DERMATOLOGY
Bariatric surgery may curtail colorectal cancer risk
Bariatric surgery was associated with a significant reduction in the risk of colorectal cancer among obese adults in a retrospective study of more than 1 million individuals.
Although some studies have suggested that bariatric surgery may reduce the risk of obesity-associated cancers, such as colorectal cancer, other studies have shown an increased colorectal cancer risk after surgery, according to Laurent Bailly, MD, of Université Côte d’Azur in Nice, France, and colleagues.
In a study published in JAMA Surgery, Dr. Bailly and colleagues compared the incidence of colorectal cancer in obese patients who underwent bariatric surgery with the incidence in obese patients who did not have surgery and the incidence in the general population.
Using the French National Health Insurance Information System database, the researchers identified 1,045,348 obese adults aged 50-75 years who had no colorectal cancer at baseline. Of these patients, 74,131 underwent bariatric surgery and 971,217 did not. The mean age was 57.3 years in the surgery group and 63.4 years in the nonsurgery group.
The mean follow-up period was 6.2 years for patients who underwent adjustable gastric banding, 5.5 years for those with sleeve gastrectomy, 5.7 years for those who underwent gastric bypass, and 5.3 years for the nonsurgery group.
Results
Overall, the colorectal cancer rate was 0.6% in the surgery group and 1.3% in the nonsurgery group (P < .001).
The researchers calculated standardized incidence ratios (SIRs) to compare the risk of colorectal cancer in the study population with the risk among the French general population; in other words, the number of observed colorectal cancer cases divided by the number of expected cases.
In the surgery group, 423 cases of colorectal cancer were observed and 428 cases were expected, which leads to an SIR of 1.0. In the nonsurgery group, 12,629 cases were observed and 9,417 cases were expected, leading to an SIR of 1.34.
These results suggest patients in the nonsurgery group had a 34% higher risk of colorectal cancer compared with the general population, whereas the risk in the surgery group was similar to that in the general population.
Patients who underwent either gastric bypass or sleeve gastrectomy had fewer new colorectal cancer diagnoses (0.5% for both) compared with patients who had adjustable gastric banding (0.7%).
The researchers noted that this study was limited by several factors, including the retrospective, observational design and potential selection bias among surgery patients. However, the results were strengthened by the large study population and long-term follow-up.
Putting results into context
The authors of an invited commentary noted that this study is supported by results from a retrospective, U.S.-based study, which indicated that bariatric surgery has a “protective effect” against colorectal cancer (Ann Surg. 2019 Jan;269[1]:95-101).
However, these results conflict with other retrospective studies. A study of Nordic patients suggested that bariatric surgery is associated with an increased risk of colon cancer but perhaps not rectal cancer (Int J Cancer. 2019. doi: 10.1002/ijc.32770).
And a study of English patients showed an increased risk of colorectal cancer in patients who underwent gastric bypass but not in those who underwent gastric banding or sleeve gastrectomy (Br J Surg. 2018;105(12):1650-7).
These conflicting results “imply that the jury is still out on whether bariatric surgery increases or decreases” the risk of colorectal cancer, the commentators wrote. They added that future studies “must account for differences in study population (i.e., race/ethnicity and national origin), mechanistic variation in bariatric surgical type, and length of follow-up, while also distinguishing between rectal and colon cancer before the case is settled.”
This study had no outside sponsorship, and the researchers and commentators had no financial conflicts to disclose.
SOURCE: Bailly L et al. JAMA Surg. 2020 Mar 11. doi:10.1001/jamasurg.2020.0089; Davidson LE et al. JAMA Surg. 2020 Mar. 11. doi:10.1001/jamasurg.2020.0090.
Bariatric surgery was associated with a significant reduction in the risk of colorectal cancer among obese adults in a retrospective study of more than 1 million individuals.
Although some studies have suggested that bariatric surgery may reduce the risk of obesity-associated cancers, such as colorectal cancer, other studies have shown an increased colorectal cancer risk after surgery, according to Laurent Bailly, MD, of Université Côte d’Azur in Nice, France, and colleagues.
In a study published in JAMA Surgery, Dr. Bailly and colleagues compared the incidence of colorectal cancer in obese patients who underwent bariatric surgery with the incidence in obese patients who did not have surgery and the incidence in the general population.
Using the French National Health Insurance Information System database, the researchers identified 1,045,348 obese adults aged 50-75 years who had no colorectal cancer at baseline. Of these patients, 74,131 underwent bariatric surgery and 971,217 did not. The mean age was 57.3 years in the surgery group and 63.4 years in the nonsurgery group.
The mean follow-up period was 6.2 years for patients who underwent adjustable gastric banding, 5.5 years for those with sleeve gastrectomy, 5.7 years for those who underwent gastric bypass, and 5.3 years for the nonsurgery group.
Results
Overall, the colorectal cancer rate was 0.6% in the surgery group and 1.3% in the nonsurgery group (P < .001).
The researchers calculated standardized incidence ratios (SIRs) to compare the risk of colorectal cancer in the study population with the risk among the French general population; in other words, the number of observed colorectal cancer cases divided by the number of expected cases.
In the surgery group, 423 cases of colorectal cancer were observed and 428 cases were expected, which leads to an SIR of 1.0. In the nonsurgery group, 12,629 cases were observed and 9,417 cases were expected, leading to an SIR of 1.34.
These results suggest patients in the nonsurgery group had a 34% higher risk of colorectal cancer compared with the general population, whereas the risk in the surgery group was similar to that in the general population.
Patients who underwent either gastric bypass or sleeve gastrectomy had fewer new colorectal cancer diagnoses (0.5% for both) compared with patients who had adjustable gastric banding (0.7%).
The researchers noted that this study was limited by several factors, including the retrospective, observational design and potential selection bias among surgery patients. However, the results were strengthened by the large study population and long-term follow-up.
Putting results into context
The authors of an invited commentary noted that this study is supported by results from a retrospective, U.S.-based study, which indicated that bariatric surgery has a “protective effect” against colorectal cancer (Ann Surg. 2019 Jan;269[1]:95-101).
However, these results conflict with other retrospective studies. A study of Nordic patients suggested that bariatric surgery is associated with an increased risk of colon cancer but perhaps not rectal cancer (Int J Cancer. 2019. doi: 10.1002/ijc.32770).
And a study of English patients showed an increased risk of colorectal cancer in patients who underwent gastric bypass but not in those who underwent gastric banding or sleeve gastrectomy (Br J Surg. 2018;105(12):1650-7).
These conflicting results “imply that the jury is still out on whether bariatric surgery increases or decreases” the risk of colorectal cancer, the commentators wrote. They added that future studies “must account for differences in study population (i.e., race/ethnicity and national origin), mechanistic variation in bariatric surgical type, and length of follow-up, while also distinguishing between rectal and colon cancer before the case is settled.”
This study had no outside sponsorship, and the researchers and commentators had no financial conflicts to disclose.
SOURCE: Bailly L et al. JAMA Surg. 2020 Mar 11. doi:10.1001/jamasurg.2020.0089; Davidson LE et al. JAMA Surg. 2020 Mar. 11. doi:10.1001/jamasurg.2020.0090.
Bariatric surgery was associated with a significant reduction in the risk of colorectal cancer among obese adults in a retrospective study of more than 1 million individuals.
Although some studies have suggested that bariatric surgery may reduce the risk of obesity-associated cancers, such as colorectal cancer, other studies have shown an increased colorectal cancer risk after surgery, according to Laurent Bailly, MD, of Université Côte d’Azur in Nice, France, and colleagues.
In a study published in JAMA Surgery, Dr. Bailly and colleagues compared the incidence of colorectal cancer in obese patients who underwent bariatric surgery with the incidence in obese patients who did not have surgery and the incidence in the general population.
Using the French National Health Insurance Information System database, the researchers identified 1,045,348 obese adults aged 50-75 years who had no colorectal cancer at baseline. Of these patients, 74,131 underwent bariatric surgery and 971,217 did not. The mean age was 57.3 years in the surgery group and 63.4 years in the nonsurgery group.
The mean follow-up period was 6.2 years for patients who underwent adjustable gastric banding, 5.5 years for those with sleeve gastrectomy, 5.7 years for those who underwent gastric bypass, and 5.3 years for the nonsurgery group.
Results
Overall, the colorectal cancer rate was 0.6% in the surgery group and 1.3% in the nonsurgery group (P < .001).
The researchers calculated standardized incidence ratios (SIRs) to compare the risk of colorectal cancer in the study population with the risk among the French general population; in other words, the number of observed colorectal cancer cases divided by the number of expected cases.
In the surgery group, 423 cases of colorectal cancer were observed and 428 cases were expected, which leads to an SIR of 1.0. In the nonsurgery group, 12,629 cases were observed and 9,417 cases were expected, leading to an SIR of 1.34.
These results suggest patients in the nonsurgery group had a 34% higher risk of colorectal cancer compared with the general population, whereas the risk in the surgery group was similar to that in the general population.
Patients who underwent either gastric bypass or sleeve gastrectomy had fewer new colorectal cancer diagnoses (0.5% for both) compared with patients who had adjustable gastric banding (0.7%).
The researchers noted that this study was limited by several factors, including the retrospective, observational design and potential selection bias among surgery patients. However, the results were strengthened by the large study population and long-term follow-up.
Putting results into context
The authors of an invited commentary noted that this study is supported by results from a retrospective, U.S.-based study, which indicated that bariatric surgery has a “protective effect” against colorectal cancer (Ann Surg. 2019 Jan;269[1]:95-101).
However, these results conflict with other retrospective studies. A study of Nordic patients suggested that bariatric surgery is associated with an increased risk of colon cancer but perhaps not rectal cancer (Int J Cancer. 2019. doi: 10.1002/ijc.32770).
And a study of English patients showed an increased risk of colorectal cancer in patients who underwent gastric bypass but not in those who underwent gastric banding or sleeve gastrectomy (Br J Surg. 2018;105(12):1650-7).
These conflicting results “imply that the jury is still out on whether bariatric surgery increases or decreases” the risk of colorectal cancer, the commentators wrote. They added that future studies “must account for differences in study population (i.e., race/ethnicity and national origin), mechanistic variation in bariatric surgical type, and length of follow-up, while also distinguishing between rectal and colon cancer before the case is settled.”
This study had no outside sponsorship, and the researchers and commentators had no financial conflicts to disclose.
SOURCE: Bailly L et al. JAMA Surg. 2020 Mar 11. doi:10.1001/jamasurg.2020.0089; Davidson LE et al. JAMA Surg. 2020 Mar. 11. doi:10.1001/jamasurg.2020.0090.
FROM JAMA SURGERY
New RA diagnosis prompts patients to quit smoking
Health care factors, such as type of health system and being newly diagnosed, rather than patient socioeconomic factors, were significant predictors of smoking cessation in patients with RA, according to findings published in Arthritis Care & Research.
Smoking is a known risk factor for poor outcomes in RA, but data on factors that predict smoking cessation in these patients are limited, wrote Maria Schletzbaum, a student in the MD-PhD program at the University of Wisconsin–Madison, and colleagues. “Further, most patients with RA are not aware of the associations between smoking and RA development and complications, although such knowledge could influence cessation attempts.”
To identify predictors of smoking cessation, the investigators reviewed the 2005-2016 electronic medical records for 3,577 adults aged 18 years or older and with at least two RA diagnosis codes. The records were from two health systems, one urban and one rural.
Overall, patients who were baseline smokers and who were new to rheumatology care were 60% more likely to quit smoking (adjusted odds ratio, 1.60). However, patients who were seropositive and baseline smokers were 43% less likely to quit (aOR, 0.57). Demographic factors, including age, race, and sex, were not significantly associated with smoking cessation.
“The observed increased likelihood of quitting in patients new to rheumatology care may partially be due to cessation following a new RA diagnosis, a phenomenon previously reported in RA and other chronic diseases,” the researchers noted. Notably, the significance of being new to rheumatology disappeared in an analysis controlling for health system, “potentially suggesting greater importance of system-level factors versus individual patient factors.”
In addition, patients in the rural community health system were 66% more likely to quit smoking, possibly because about half of the patients in that system were covered by the systems’ insurance, and therefore qualified for various smoking cessation interventions, they wrote.
The study population included 915 former smokers and 507 current smokers. Seropositivity was most common in current smokers (71%), followed by former smokers (64%) and never smokers (59%). The disinclination of seropositive patients to quit smoking may reflect greater smoking intensity, and these patients may need greater support, the researchers wrote.
The study findings were limited by several factors; for example, some differences in patient populations, such as education and income levels, were not measured, and there was a lack of complete information on cumulative smoking exposure. However, the results were strengthened by the large sample size and use of data from two centers and support national guidelines for health system interventions in smoking cessation for RA patients, they noted.
The study was funded in part by the Rheumatology Research Foundation, the University of Wisconsin Medical Scientist Training Program, and the University of Wisconsin Clinical and Translational Science Award. The authors did not report any financial conflicts of interest.
SOURCE: Schletzbaum M et al. Arthritis Car Res. 2020 Mar 3. doi: 10.1002/ACR.24154.
Health care factors, such as type of health system and being newly diagnosed, rather than patient socioeconomic factors, were significant predictors of smoking cessation in patients with RA, according to findings published in Arthritis Care & Research.
Smoking is a known risk factor for poor outcomes in RA, but data on factors that predict smoking cessation in these patients are limited, wrote Maria Schletzbaum, a student in the MD-PhD program at the University of Wisconsin–Madison, and colleagues. “Further, most patients with RA are not aware of the associations between smoking and RA development and complications, although such knowledge could influence cessation attempts.”
To identify predictors of smoking cessation, the investigators reviewed the 2005-2016 electronic medical records for 3,577 adults aged 18 years or older and with at least two RA diagnosis codes. The records were from two health systems, one urban and one rural.
Overall, patients who were baseline smokers and who were new to rheumatology care were 60% more likely to quit smoking (adjusted odds ratio, 1.60). However, patients who were seropositive and baseline smokers were 43% less likely to quit (aOR, 0.57). Demographic factors, including age, race, and sex, were not significantly associated with smoking cessation.
“The observed increased likelihood of quitting in patients new to rheumatology care may partially be due to cessation following a new RA diagnosis, a phenomenon previously reported in RA and other chronic diseases,” the researchers noted. Notably, the significance of being new to rheumatology disappeared in an analysis controlling for health system, “potentially suggesting greater importance of system-level factors versus individual patient factors.”
In addition, patients in the rural community health system were 66% more likely to quit smoking, possibly because about half of the patients in that system were covered by the systems’ insurance, and therefore qualified for various smoking cessation interventions, they wrote.
The study population included 915 former smokers and 507 current smokers. Seropositivity was most common in current smokers (71%), followed by former smokers (64%) and never smokers (59%). The disinclination of seropositive patients to quit smoking may reflect greater smoking intensity, and these patients may need greater support, the researchers wrote.
The study findings were limited by several factors; for example, some differences in patient populations, such as education and income levels, were not measured, and there was a lack of complete information on cumulative smoking exposure. However, the results were strengthened by the large sample size and use of data from two centers and support national guidelines for health system interventions in smoking cessation for RA patients, they noted.
The study was funded in part by the Rheumatology Research Foundation, the University of Wisconsin Medical Scientist Training Program, and the University of Wisconsin Clinical and Translational Science Award. The authors did not report any financial conflicts of interest.
SOURCE: Schletzbaum M et al. Arthritis Car Res. 2020 Mar 3. doi: 10.1002/ACR.24154.
Health care factors, such as type of health system and being newly diagnosed, rather than patient socioeconomic factors, were significant predictors of smoking cessation in patients with RA, according to findings published in Arthritis Care & Research.
Smoking is a known risk factor for poor outcomes in RA, but data on factors that predict smoking cessation in these patients are limited, wrote Maria Schletzbaum, a student in the MD-PhD program at the University of Wisconsin–Madison, and colleagues. “Further, most patients with RA are not aware of the associations between smoking and RA development and complications, although such knowledge could influence cessation attempts.”
To identify predictors of smoking cessation, the investigators reviewed the 2005-2016 electronic medical records for 3,577 adults aged 18 years or older and with at least two RA diagnosis codes. The records were from two health systems, one urban and one rural.
Overall, patients who were baseline smokers and who were new to rheumatology care were 60% more likely to quit smoking (adjusted odds ratio, 1.60). However, patients who were seropositive and baseline smokers were 43% less likely to quit (aOR, 0.57). Demographic factors, including age, race, and sex, were not significantly associated with smoking cessation.
“The observed increased likelihood of quitting in patients new to rheumatology care may partially be due to cessation following a new RA diagnosis, a phenomenon previously reported in RA and other chronic diseases,” the researchers noted. Notably, the significance of being new to rheumatology disappeared in an analysis controlling for health system, “potentially suggesting greater importance of system-level factors versus individual patient factors.”
In addition, patients in the rural community health system were 66% more likely to quit smoking, possibly because about half of the patients in that system were covered by the systems’ insurance, and therefore qualified for various smoking cessation interventions, they wrote.
The study population included 915 former smokers and 507 current smokers. Seropositivity was most common in current smokers (71%), followed by former smokers (64%) and never smokers (59%). The disinclination of seropositive patients to quit smoking may reflect greater smoking intensity, and these patients may need greater support, the researchers wrote.
The study findings were limited by several factors; for example, some differences in patient populations, such as education and income levels, were not measured, and there was a lack of complete information on cumulative smoking exposure. However, the results were strengthened by the large sample size and use of data from two centers and support national guidelines for health system interventions in smoking cessation for RA patients, they noted.
The study was funded in part by the Rheumatology Research Foundation, the University of Wisconsin Medical Scientist Training Program, and the University of Wisconsin Clinical and Translational Science Award. The authors did not report any financial conflicts of interest.
SOURCE: Schletzbaum M et al. Arthritis Car Res. 2020 Mar 3. doi: 10.1002/ACR.24154.
FROM ARTHRITIS CARE & RESEARCH
Biomarker pattern flags risk for microalbuminuria in diabetes patients
A high-risk profile from the urinary biomarker CKD273 was significantly associated with an increased risk of microalbuminuria in patients with diabetes, according to findings from a multicenter European trial.
“Although microalbuminuria is the earliest clinical index of renal damage, histological changes might already be advanced by the time it is detectable. Thus, earlier identification of at-risk individuals is essential to guide targeted preventive therapy,” wrote Nete Tofte, MD, of the Steno Diabetes Center in Copenhagen, and colleagues.
“Increases in urinary albumin to microalbuminuria levels, or higher, are not only strongly associated with progression to more serious clinical endpoints, such as clinically significant loss of renal function and eventually, end-stage kidney disease, but also with an increased risk of cardiovascular complications,” the researchers noted in the Lancet Diabetes & Endocrinology.
They identified 1,775 adults with type 2 diabetes who had normal albumin levels and preserved renal function at baseline. The average age of the patients was 62 years, and 62% were men. The participants underwent urine proteomics testing via capillary electrophoresis–mass spectrometry analysis to generate a renal risk profile based on 273 peptides (CKD273). On the basis of their CKD273 scores, 216 patients (12%) were designated to the high-risk group, and 1,556 (88%) to the low-risk group.
Over a median follow-up of 2.5 years, 61 patients (28%) in the high-risk group progressed to microalbuminuria (the primary endpoint), compared with 139 patients (9%) in the low-risk group.
Of the original 216 high-risk patients, 209 were randomized to treatment with 25 mg of the mineralocorticoid receptor antagonist spironolactone (102 patients) or placebo (107) to examine whether spironolactone would stall progression to microalbuminuria.
The researchers found, however, that spironolactone did not prevent progression to microalbuminuria. In all, 26 of the 102 patients (25%) patients in the spironolactone group developed microalbuminuria, and 35 of the 107 patients (33%) in the placebo group developed it (hazard ratio, 0.81; 95% confidence interval, 0.49-1.34; P = .41).
The total number of adverse events was not significantly different between the spironolactone and placebo groups (312 vs. 321, respectively), although more patients in the spironolactone group experienced adverse events that led to drug discontinuation (25 vs. 9, respectively).
The study findings were limited by several factors, including the use of a single urine sample for risk stratification; the lower-than-expected number of high-risk patients; not testing spironolactone in the low-risk group; and the fact that microalbuminuria, although an accepted surrogate for diabetic kidney disease, is not approved as such by regulatory agencies, the researchers noted. However, the results were strengthened by the large study population and prospective design, as well as the additional register-based follow-up that is planned when possible.
In an accompanying editorial, Susanne B. Nicholas, MD, of the University of California, Los Angeles, reiterated that microalbuminuria cannot be used to track responses to therapy even if it is an acceptable indicator of potential renal damage.
“In fact, regression from microalbuminuria to normoalbuminuria is more likely than progression toward overt proteinuria, [which exposes] a need for a more dependable biomarker for diabetic kidney disease,” she wrote.
However, Dr. Nicholas supported the potential of proteomics as a tool “that could bridge the gap between discovery of diabetic kidney disease – possibly providing a panel, rather than a single or few urinary indicators of structural changes that predate microalbuminuria – and response to therapy, given the promise of targeted therapies for this complex disease.” Additional research into patient selection, comparators to verify findings, and cost containment is needed before proteomics can become part of routine care, she added.
The study was supported by the European Union Seventh Framework Programme. Dr. Tofte had no financial conflicts to disclose. Several other authors disclosed relationships with multiple companies, including Amgen, AstraZeneca, Boehringer Ingelheim, Novo Nordisk, and Sanofi. Dr. Nicholas had no financial conflicts to disclose.
SOURCES: Tofte N et al. Lancet Diabetes Endocrinol. 2020 Mar 2. doi: 10.1016/S2213-8587(20)30026-7; Nicholas SB. Lancet Diabetes Endocrinol. 2020 Mar 2. doi: 10.1016/S2213-8587(20)30067-X.
A high-risk profile from the urinary biomarker CKD273 was significantly associated with an increased risk of microalbuminuria in patients with diabetes, according to findings from a multicenter European trial.
“Although microalbuminuria is the earliest clinical index of renal damage, histological changes might already be advanced by the time it is detectable. Thus, earlier identification of at-risk individuals is essential to guide targeted preventive therapy,” wrote Nete Tofte, MD, of the Steno Diabetes Center in Copenhagen, and colleagues.
“Increases in urinary albumin to microalbuminuria levels, or higher, are not only strongly associated with progression to more serious clinical endpoints, such as clinically significant loss of renal function and eventually, end-stage kidney disease, but also with an increased risk of cardiovascular complications,” the researchers noted in the Lancet Diabetes & Endocrinology.
They identified 1,775 adults with type 2 diabetes who had normal albumin levels and preserved renal function at baseline. The average age of the patients was 62 years, and 62% were men. The participants underwent urine proteomics testing via capillary electrophoresis–mass spectrometry analysis to generate a renal risk profile based on 273 peptides (CKD273). On the basis of their CKD273 scores, 216 patients (12%) were designated to the high-risk group, and 1,556 (88%) to the low-risk group.
Over a median follow-up of 2.5 years, 61 patients (28%) in the high-risk group progressed to microalbuminuria (the primary endpoint), compared with 139 patients (9%) in the low-risk group.
Of the original 216 high-risk patients, 209 were randomized to treatment with 25 mg of the mineralocorticoid receptor antagonist spironolactone (102 patients) or placebo (107) to examine whether spironolactone would stall progression to microalbuminuria.
The researchers found, however, that spironolactone did not prevent progression to microalbuminuria. In all, 26 of the 102 patients (25%) patients in the spironolactone group developed microalbuminuria, and 35 of the 107 patients (33%) in the placebo group developed it (hazard ratio, 0.81; 95% confidence interval, 0.49-1.34; P = .41).
The total number of adverse events was not significantly different between the spironolactone and placebo groups (312 vs. 321, respectively), although more patients in the spironolactone group experienced adverse events that led to drug discontinuation (25 vs. 9, respectively).
The study findings were limited by several factors, including the use of a single urine sample for risk stratification; the lower-than-expected number of high-risk patients; not testing spironolactone in the low-risk group; and the fact that microalbuminuria, although an accepted surrogate for diabetic kidney disease, is not approved as such by regulatory agencies, the researchers noted. However, the results were strengthened by the large study population and prospective design, as well as the additional register-based follow-up that is planned when possible.
In an accompanying editorial, Susanne B. Nicholas, MD, of the University of California, Los Angeles, reiterated that microalbuminuria cannot be used to track responses to therapy even if it is an acceptable indicator of potential renal damage.
“In fact, regression from microalbuminuria to normoalbuminuria is more likely than progression toward overt proteinuria, [which exposes] a need for a more dependable biomarker for diabetic kidney disease,” she wrote.
However, Dr. Nicholas supported the potential of proteomics as a tool “that could bridge the gap between discovery of diabetic kidney disease – possibly providing a panel, rather than a single or few urinary indicators of structural changes that predate microalbuminuria – and response to therapy, given the promise of targeted therapies for this complex disease.” Additional research into patient selection, comparators to verify findings, and cost containment is needed before proteomics can become part of routine care, she added.
The study was supported by the European Union Seventh Framework Programme. Dr. Tofte had no financial conflicts to disclose. Several other authors disclosed relationships with multiple companies, including Amgen, AstraZeneca, Boehringer Ingelheim, Novo Nordisk, and Sanofi. Dr. Nicholas had no financial conflicts to disclose.
SOURCES: Tofte N et al. Lancet Diabetes Endocrinol. 2020 Mar 2. doi: 10.1016/S2213-8587(20)30026-7; Nicholas SB. Lancet Diabetes Endocrinol. 2020 Mar 2. doi: 10.1016/S2213-8587(20)30067-X.
A high-risk profile from the urinary biomarker CKD273 was significantly associated with an increased risk of microalbuminuria in patients with diabetes, according to findings from a multicenter European trial.
“Although microalbuminuria is the earliest clinical index of renal damage, histological changes might already be advanced by the time it is detectable. Thus, earlier identification of at-risk individuals is essential to guide targeted preventive therapy,” wrote Nete Tofte, MD, of the Steno Diabetes Center in Copenhagen, and colleagues.
“Increases in urinary albumin to microalbuminuria levels, or higher, are not only strongly associated with progression to more serious clinical endpoints, such as clinically significant loss of renal function and eventually, end-stage kidney disease, but also with an increased risk of cardiovascular complications,” the researchers noted in the Lancet Diabetes & Endocrinology.
They identified 1,775 adults with type 2 diabetes who had normal albumin levels and preserved renal function at baseline. The average age of the patients was 62 years, and 62% were men. The participants underwent urine proteomics testing via capillary electrophoresis–mass spectrometry analysis to generate a renal risk profile based on 273 peptides (CKD273). On the basis of their CKD273 scores, 216 patients (12%) were designated to the high-risk group, and 1,556 (88%) to the low-risk group.
Over a median follow-up of 2.5 years, 61 patients (28%) in the high-risk group progressed to microalbuminuria (the primary endpoint), compared with 139 patients (9%) in the low-risk group.
Of the original 216 high-risk patients, 209 were randomized to treatment with 25 mg of the mineralocorticoid receptor antagonist spironolactone (102 patients) or placebo (107) to examine whether spironolactone would stall progression to microalbuminuria.
The researchers found, however, that spironolactone did not prevent progression to microalbuminuria. In all, 26 of the 102 patients (25%) patients in the spironolactone group developed microalbuminuria, and 35 of the 107 patients (33%) in the placebo group developed it (hazard ratio, 0.81; 95% confidence interval, 0.49-1.34; P = .41).
The total number of adverse events was not significantly different between the spironolactone and placebo groups (312 vs. 321, respectively), although more patients in the spironolactone group experienced adverse events that led to drug discontinuation (25 vs. 9, respectively).
The study findings were limited by several factors, including the use of a single urine sample for risk stratification; the lower-than-expected number of high-risk patients; not testing spironolactone in the low-risk group; and the fact that microalbuminuria, although an accepted surrogate for diabetic kidney disease, is not approved as such by regulatory agencies, the researchers noted. However, the results were strengthened by the large study population and prospective design, as well as the additional register-based follow-up that is planned when possible.
In an accompanying editorial, Susanne B. Nicholas, MD, of the University of California, Los Angeles, reiterated that microalbuminuria cannot be used to track responses to therapy even if it is an acceptable indicator of potential renal damage.
“In fact, regression from microalbuminuria to normoalbuminuria is more likely than progression toward overt proteinuria, [which exposes] a need for a more dependable biomarker for diabetic kidney disease,” she wrote.
However, Dr. Nicholas supported the potential of proteomics as a tool “that could bridge the gap between discovery of diabetic kidney disease – possibly providing a panel, rather than a single or few urinary indicators of structural changes that predate microalbuminuria – and response to therapy, given the promise of targeted therapies for this complex disease.” Additional research into patient selection, comparators to verify findings, and cost containment is needed before proteomics can become part of routine care, she added.
The study was supported by the European Union Seventh Framework Programme. Dr. Tofte had no financial conflicts to disclose. Several other authors disclosed relationships with multiple companies, including Amgen, AstraZeneca, Boehringer Ingelheim, Novo Nordisk, and Sanofi. Dr. Nicholas had no financial conflicts to disclose.
SOURCES: Tofte N et al. Lancet Diabetes Endocrinol. 2020 Mar 2. doi: 10.1016/S2213-8587(20)30026-7; Nicholas SB. Lancet Diabetes Endocrinol. 2020 Mar 2. doi: 10.1016/S2213-8587(20)30067-X.
FROM THE LANCET DIABETES & ENDOCRINOLOGY
RYGB tops sleeve gastrectomy in long-term outcomes for diabetes
Patients with type 2 diabetes who underwent Roux-en-Y gastric bypass (RYGB) surgery experienced higher rates of diabetes remission, improved glycemic control, greater weight loss, and fewer diabetes relapse events, compared with those who had sleeve gastrectomy, according to findings from nearly 10,000 patients.
“Remission of type 2 diabetes is common after bariatric surgery and may reduce risk for subsequent microvascular and macrovascular disease,” but it is not clear which of the two most common procedures, RYGB or sleeve gastrectomy, has better long-term diabetes and weight outcomes, wrote Kathleen M. McTigue, MD, of the University of Pittsburgh, and colleagues in JAMA Surgery.
To examine the effectiveness of the two procedures, the researchers identified 9,710 adults with type 2 diabetes who were part of the National Patient-Centered Clinical Research Network Bariatric Study. They compared diabetes outcomes for up to 5 years after surgery for 6,233 patients who underwent RYGB and 3,477 who underwent sleeve gastrectomy. The average age of the patients was 50 years, and 73% were women. The average preoperative body mass index was 49 kg/m2.
Overall, 6,141 patients experienced diabetes remission. The estimated adjusted cumulative remission rates for the RYGB and sleeve gastrectomy groups after 1 year were 59% and 56%, respectively, and after 5 years were 86% and 84%.
Weight loss was significantly greater in RYGB patients, compared with those who had the sleeve gastrectomy, with average differences in percentage points of 6.3 at 1 year and 8.1 at year 5. RYGB patients also showed significantly better long-term glycemic control, compared with sleeve gastrectomy patients. At 5 years, hemoglobin A1c levels were 0.80 percentage points below baseline in the RYGB group, and 0.35 percentage points below baseline in the sleeve gastrectomy group.
In addition, after 1 year, diabetes relapse rates in the RYGB and sleeve gastrectomy groups were 8% and 11%, respectively, and 33% and 42% after 5 years.
The findings were limited by several factors, including the observational design of the study and the potential for confounding and coding inaccuracies, the researchers noted, adding that future studies should address the impact of weight loss on diabetes remission and relapse in bariatric surgery patients.
They also noted that their results were in contrast to findings in previous studies that established no significant differences in outcomes between the procedures, but emphasized that most previous studies were smaller and controlled and that outcome differences may be greater in clinical practice.
“For patients, clinicians, and policy makers to make informed decisions about which procedure is best suited to patients’ personal situations, additional data are needed to understand the adverse event profile of the procedures, as well as patient values regarding procedure choice and the role of surgery relative to other aspects of lifelong weight management,” they concluded.
In an accompanying commentary, Natalie Liu, MD, and Luke M. Funk, MD, of the department of surgery, University of Wisconsin–Madison, said the analysis made an important contribution to the existing literature, despite its limitations.
“It included long-term electronic health record data from a large cohort of U.S. patients who had bariatric surgery in a real-world setting,” they wrote, adding that, although the remission rates were high, the relapse rate in both treatment groups deserved further study.
Dr. Liu and Dr. Funk emphasized that the overall high remission rates for either surgery, compared with lifestyle interventions, suggest the need for continued advocacy for better insurance coverage of, and access to, bariatric surgery procedures for patients with type 2 diabetes, notably those with class 1 obesity.
The study was conducted using the National Patient-Centered Clinical Research Network, which was funded by the Patient-Centered Outcomes Research Institute. Dr. McTigue and Dr. Liu had reported no conflicts of interest. Dr. Funk disclosed a Veterans Affairs Health Services Research & Development Career Development Award, and grants from the VA, National Institutes of Health, and American College of Surgeons.
SOURCES: McTigue KM et al. JAMA Surg. 2020 Mar 4. doi: 10.1001/jamasurg.2020.0087; Lui N, Funk LM. JAMA Surg. 2020 Mar 4. doi: 10.1001/jamasurg.2020.0088.
Patients with type 2 diabetes who underwent Roux-en-Y gastric bypass (RYGB) surgery experienced higher rates of diabetes remission, improved glycemic control, greater weight loss, and fewer diabetes relapse events, compared with those who had sleeve gastrectomy, according to findings from nearly 10,000 patients.
“Remission of type 2 diabetes is common after bariatric surgery and may reduce risk for subsequent microvascular and macrovascular disease,” but it is not clear which of the two most common procedures, RYGB or sleeve gastrectomy, has better long-term diabetes and weight outcomes, wrote Kathleen M. McTigue, MD, of the University of Pittsburgh, and colleagues in JAMA Surgery.
To examine the effectiveness of the two procedures, the researchers identified 9,710 adults with type 2 diabetes who were part of the National Patient-Centered Clinical Research Network Bariatric Study. They compared diabetes outcomes for up to 5 years after surgery for 6,233 patients who underwent RYGB and 3,477 who underwent sleeve gastrectomy. The average age of the patients was 50 years, and 73% were women. The average preoperative body mass index was 49 kg/m2.
Overall, 6,141 patients experienced diabetes remission. The estimated adjusted cumulative remission rates for the RYGB and sleeve gastrectomy groups after 1 year were 59% and 56%, respectively, and after 5 years were 86% and 84%.
Weight loss was significantly greater in RYGB patients, compared with those who had the sleeve gastrectomy, with average differences in percentage points of 6.3 at 1 year and 8.1 at year 5. RYGB patients also showed significantly better long-term glycemic control, compared with sleeve gastrectomy patients. At 5 years, hemoglobin A1c levels were 0.80 percentage points below baseline in the RYGB group, and 0.35 percentage points below baseline in the sleeve gastrectomy group.
In addition, after 1 year, diabetes relapse rates in the RYGB and sleeve gastrectomy groups were 8% and 11%, respectively, and 33% and 42% after 5 years.
The findings were limited by several factors, including the observational design of the study and the potential for confounding and coding inaccuracies, the researchers noted, adding that future studies should address the impact of weight loss on diabetes remission and relapse in bariatric surgery patients.
They also noted that their results were in contrast to findings in previous studies that established no significant differences in outcomes between the procedures, but emphasized that most previous studies were smaller and controlled and that outcome differences may be greater in clinical practice.
“For patients, clinicians, and policy makers to make informed decisions about which procedure is best suited to patients’ personal situations, additional data are needed to understand the adverse event profile of the procedures, as well as patient values regarding procedure choice and the role of surgery relative to other aspects of lifelong weight management,” they concluded.
In an accompanying commentary, Natalie Liu, MD, and Luke M. Funk, MD, of the department of surgery, University of Wisconsin–Madison, said the analysis made an important contribution to the existing literature, despite its limitations.
“It included long-term electronic health record data from a large cohort of U.S. patients who had bariatric surgery in a real-world setting,” they wrote, adding that, although the remission rates were high, the relapse rate in both treatment groups deserved further study.
Dr. Liu and Dr. Funk emphasized that the overall high remission rates for either surgery, compared with lifestyle interventions, suggest the need for continued advocacy for better insurance coverage of, and access to, bariatric surgery procedures for patients with type 2 diabetes, notably those with class 1 obesity.
The study was conducted using the National Patient-Centered Clinical Research Network, which was funded by the Patient-Centered Outcomes Research Institute. Dr. McTigue and Dr. Liu had reported no conflicts of interest. Dr. Funk disclosed a Veterans Affairs Health Services Research & Development Career Development Award, and grants from the VA, National Institutes of Health, and American College of Surgeons.
SOURCES: McTigue KM et al. JAMA Surg. 2020 Mar 4. doi: 10.1001/jamasurg.2020.0087; Lui N, Funk LM. JAMA Surg. 2020 Mar 4. doi: 10.1001/jamasurg.2020.0088.
Patients with type 2 diabetes who underwent Roux-en-Y gastric bypass (RYGB) surgery experienced higher rates of diabetes remission, improved glycemic control, greater weight loss, and fewer diabetes relapse events, compared with those who had sleeve gastrectomy, according to findings from nearly 10,000 patients.
“Remission of type 2 diabetes is common after bariatric surgery and may reduce risk for subsequent microvascular and macrovascular disease,” but it is not clear which of the two most common procedures, RYGB or sleeve gastrectomy, has better long-term diabetes and weight outcomes, wrote Kathleen M. McTigue, MD, of the University of Pittsburgh, and colleagues in JAMA Surgery.
To examine the effectiveness of the two procedures, the researchers identified 9,710 adults with type 2 diabetes who were part of the National Patient-Centered Clinical Research Network Bariatric Study. They compared diabetes outcomes for up to 5 years after surgery for 6,233 patients who underwent RYGB and 3,477 who underwent sleeve gastrectomy. The average age of the patients was 50 years, and 73% were women. The average preoperative body mass index was 49 kg/m2.
Overall, 6,141 patients experienced diabetes remission. The estimated adjusted cumulative remission rates for the RYGB and sleeve gastrectomy groups after 1 year were 59% and 56%, respectively, and after 5 years were 86% and 84%.
Weight loss was significantly greater in RYGB patients, compared with those who had the sleeve gastrectomy, with average differences in percentage points of 6.3 at 1 year and 8.1 at year 5. RYGB patients also showed significantly better long-term glycemic control, compared with sleeve gastrectomy patients. At 5 years, hemoglobin A1c levels were 0.80 percentage points below baseline in the RYGB group, and 0.35 percentage points below baseline in the sleeve gastrectomy group.
In addition, after 1 year, diabetes relapse rates in the RYGB and sleeve gastrectomy groups were 8% and 11%, respectively, and 33% and 42% after 5 years.
The findings were limited by several factors, including the observational design of the study and the potential for confounding and coding inaccuracies, the researchers noted, adding that future studies should address the impact of weight loss on diabetes remission and relapse in bariatric surgery patients.
They also noted that their results were in contrast to findings in previous studies that established no significant differences in outcomes between the procedures, but emphasized that most previous studies were smaller and controlled and that outcome differences may be greater in clinical practice.
“For patients, clinicians, and policy makers to make informed decisions about which procedure is best suited to patients’ personal situations, additional data are needed to understand the adverse event profile of the procedures, as well as patient values regarding procedure choice and the role of surgery relative to other aspects of lifelong weight management,” they concluded.
In an accompanying commentary, Natalie Liu, MD, and Luke M. Funk, MD, of the department of surgery, University of Wisconsin–Madison, said the analysis made an important contribution to the existing literature, despite its limitations.
“It included long-term electronic health record data from a large cohort of U.S. patients who had bariatric surgery in a real-world setting,” they wrote, adding that, although the remission rates were high, the relapse rate in both treatment groups deserved further study.
Dr. Liu and Dr. Funk emphasized that the overall high remission rates for either surgery, compared with lifestyle interventions, suggest the need for continued advocacy for better insurance coverage of, and access to, bariatric surgery procedures for patients with type 2 diabetes, notably those with class 1 obesity.
The study was conducted using the National Patient-Centered Clinical Research Network, which was funded by the Patient-Centered Outcomes Research Institute. Dr. McTigue and Dr. Liu had reported no conflicts of interest. Dr. Funk disclosed a Veterans Affairs Health Services Research & Development Career Development Award, and grants from the VA, National Institutes of Health, and American College of Surgeons.
SOURCES: McTigue KM et al. JAMA Surg. 2020 Mar 4. doi: 10.1001/jamasurg.2020.0087; Lui N, Funk LM. JAMA Surg. 2020 Mar 4. doi: 10.1001/jamasurg.2020.0088.
FROM JAMA SURGERY
More pediatricians employ developmental screening tools
Pediatricians’ reported use of developmental screening tools increased significantly to 63% from 2002 to 2016, based on survey data from more than 1,000 pediatricians at three time points.
“In 2001, an AAP [American Academy of Pediatrics] policy statement called for pediatricians to screen all children for developmental disorders during routine well-child visits,” Paul H. Lipkin, MD, of the Kennedy Krieger Institute, Baltimore, and colleagues wrote in Pediatrics. “However, only 23% of pediatricians in 2002 reported using a standardized developmental screening tool, citing lack of time, staff, and reimbursement as barriers.”
To determine trends in pediatricians’ use of recommended screening tools, the researchers reviewed data from the American Academy of Pediatrics Periodic Surveys in 2002, 2009, and 2016 that included 562, 532, and 469 respondents, respectively.
The percentage of pediatricians who reported using screening tools increased from 21% in 2002 to 63% in 2016 (P less than .001). In addition, The screening tool with the greatest increase in use was the Ages and Stages Questionnaire (ASQ), reportedly used by 48% of pediatricians in 2016, up from 9% in 2002 (P less than .001).
Most reported barriers to screening, including time constraints, inadequate reimbursement, lack of staff to perform screenings, belief that screening is not an appropriate role for pediatricians, and lack of confidence in the screening effectiveness, declined over the study period. However, the percentage of pediatricians who reported lack of available treatment options as a barrier to screening increased from 9% in 2002 to 21% in 2016, the researchers noted.
The average ages of the survey respondents at the 2002, 2009, and 2016 time points were 44, 47, and 49 years, respectively, and the majority (44%, 45%, and 49%) worked in suburban practice areas.
The study findings were limited by several factors, including the use of self-reports, the potential bias of pediatricians to overestimate some of their developmental practices, and potential over- or underreporting if autism spectrum disorder screening was mistakenly included, Dr. Lipkin and associates noted. The results suggest that use of standardized screening has increased, but more attention is needed to improve screening and referrals.
Potential improvements include adding screening tests and referrals to EHRs, and improving communication between medical practices and community-based intervention, therapy, and education programs, they concluded.
The study findings can be seen as encouraging, but one-third of pediatricians still reported not using formal screening instruments, commonly citing lack of time and suboptimal reimbursement, Mei Elansary, MD, and Michael Silverstein, MD, both of Boston University, said in an accompanying editorial.
However, “Although time and financial barriers are real, it is also likely that some of the residual gaps in guideline-concordant practice reflect variability among pediatricians in their perception of the clinical relevance of certain developmental problems that require formal instruments to identify and in the availability and effectiveness of services targeted to children with these less severe developmental issues,” they said. The path for screening children with developmental risk factors but not obviously severe delays may not be straightforward, and many pediatricians rely on their clinical judgment, they emphasized.
“As important as developing strategies to achieve more widespread developmental screening, therefore, is developing a greater understanding of the root causes of practice variation and determining the range of viable clinical practices that lead to better developmental outcomes,” Dr. Elansary and Dr. Silverstein concluded.
The study was supported by the American Academy of Pediatrics, the Department of Health & Human Services, the Health Resources and Services Administration, and the Department of Education Office of Special Education Programs. The researchers had no financial conflicts to disclose.
Editorialist Dr. Silverstein disclosed an award from the Eunice Kennedy Shriver National Institute of Child Health and Human Development; the editorial was funded by the National Institutes of Health. Dr. Silverstein is a member of the U.S. Preventive Services Task Force. Dr. Elansary had no relevant financial disclosures.
SOURCES: Lipkin PH et al. Pediatrics. 2020 Mar 2. doi: 10.1542/peds.2019-0851; Elansary M, Silverstein M. Pediatrics. 2020 Mar 2. doi: 10.1542/peds.2020-0164.
Pediatricians’ reported use of developmental screening tools increased significantly to 63% from 2002 to 2016, based on survey data from more than 1,000 pediatricians at three time points.
“In 2001, an AAP [American Academy of Pediatrics] policy statement called for pediatricians to screen all children for developmental disorders during routine well-child visits,” Paul H. Lipkin, MD, of the Kennedy Krieger Institute, Baltimore, and colleagues wrote in Pediatrics. “However, only 23% of pediatricians in 2002 reported using a standardized developmental screening tool, citing lack of time, staff, and reimbursement as barriers.”
To determine trends in pediatricians’ use of recommended screening tools, the researchers reviewed data from the American Academy of Pediatrics Periodic Surveys in 2002, 2009, and 2016 that included 562, 532, and 469 respondents, respectively.
The percentage of pediatricians who reported using screening tools increased from 21% in 2002 to 63% in 2016 (P less than .001). In addition, The screening tool with the greatest increase in use was the Ages and Stages Questionnaire (ASQ), reportedly used by 48% of pediatricians in 2016, up from 9% in 2002 (P less than .001).
Most reported barriers to screening, including time constraints, inadequate reimbursement, lack of staff to perform screenings, belief that screening is not an appropriate role for pediatricians, and lack of confidence in the screening effectiveness, declined over the study period. However, the percentage of pediatricians who reported lack of available treatment options as a barrier to screening increased from 9% in 2002 to 21% in 2016, the researchers noted.
The average ages of the survey respondents at the 2002, 2009, and 2016 time points were 44, 47, and 49 years, respectively, and the majority (44%, 45%, and 49%) worked in suburban practice areas.
The study findings were limited by several factors, including the use of self-reports, the potential bias of pediatricians to overestimate some of their developmental practices, and potential over- or underreporting if autism spectrum disorder screening was mistakenly included, Dr. Lipkin and associates noted. The results suggest that use of standardized screening has increased, but more attention is needed to improve screening and referrals.
Potential improvements include adding screening tests and referrals to EHRs, and improving communication between medical practices and community-based intervention, therapy, and education programs, they concluded.
The study findings can be seen as encouraging, but one-third of pediatricians still reported not using formal screening instruments, commonly citing lack of time and suboptimal reimbursement, Mei Elansary, MD, and Michael Silverstein, MD, both of Boston University, said in an accompanying editorial.
However, “Although time and financial barriers are real, it is also likely that some of the residual gaps in guideline-concordant practice reflect variability among pediatricians in their perception of the clinical relevance of certain developmental problems that require formal instruments to identify and in the availability and effectiveness of services targeted to children with these less severe developmental issues,” they said. The path for screening children with developmental risk factors but not obviously severe delays may not be straightforward, and many pediatricians rely on their clinical judgment, they emphasized.
“As important as developing strategies to achieve more widespread developmental screening, therefore, is developing a greater understanding of the root causes of practice variation and determining the range of viable clinical practices that lead to better developmental outcomes,” Dr. Elansary and Dr. Silverstein concluded.
The study was supported by the American Academy of Pediatrics, the Department of Health & Human Services, the Health Resources and Services Administration, and the Department of Education Office of Special Education Programs. The researchers had no financial conflicts to disclose.
Editorialist Dr. Silverstein disclosed an award from the Eunice Kennedy Shriver National Institute of Child Health and Human Development; the editorial was funded by the National Institutes of Health. Dr. Silverstein is a member of the U.S. Preventive Services Task Force. Dr. Elansary had no relevant financial disclosures.
SOURCES: Lipkin PH et al. Pediatrics. 2020 Mar 2. doi: 10.1542/peds.2019-0851; Elansary M, Silverstein M. Pediatrics. 2020 Mar 2. doi: 10.1542/peds.2020-0164.
Pediatricians’ reported use of developmental screening tools increased significantly to 63% from 2002 to 2016, based on survey data from more than 1,000 pediatricians at three time points.
“In 2001, an AAP [American Academy of Pediatrics] policy statement called for pediatricians to screen all children for developmental disorders during routine well-child visits,” Paul H. Lipkin, MD, of the Kennedy Krieger Institute, Baltimore, and colleagues wrote in Pediatrics. “However, only 23% of pediatricians in 2002 reported using a standardized developmental screening tool, citing lack of time, staff, and reimbursement as barriers.”
To determine trends in pediatricians’ use of recommended screening tools, the researchers reviewed data from the American Academy of Pediatrics Periodic Surveys in 2002, 2009, and 2016 that included 562, 532, and 469 respondents, respectively.
The percentage of pediatricians who reported using screening tools increased from 21% in 2002 to 63% in 2016 (P less than .001). In addition, The screening tool with the greatest increase in use was the Ages and Stages Questionnaire (ASQ), reportedly used by 48% of pediatricians in 2016, up from 9% in 2002 (P less than .001).
Most reported barriers to screening, including time constraints, inadequate reimbursement, lack of staff to perform screenings, belief that screening is not an appropriate role for pediatricians, and lack of confidence in the screening effectiveness, declined over the study period. However, the percentage of pediatricians who reported lack of available treatment options as a barrier to screening increased from 9% in 2002 to 21% in 2016, the researchers noted.
The average ages of the survey respondents at the 2002, 2009, and 2016 time points were 44, 47, and 49 years, respectively, and the majority (44%, 45%, and 49%) worked in suburban practice areas.
The study findings were limited by several factors, including the use of self-reports, the potential bias of pediatricians to overestimate some of their developmental practices, and potential over- or underreporting if autism spectrum disorder screening was mistakenly included, Dr. Lipkin and associates noted. The results suggest that use of standardized screening has increased, but more attention is needed to improve screening and referrals.
Potential improvements include adding screening tests and referrals to EHRs, and improving communication between medical practices and community-based intervention, therapy, and education programs, they concluded.
The study findings can be seen as encouraging, but one-third of pediatricians still reported not using formal screening instruments, commonly citing lack of time and suboptimal reimbursement, Mei Elansary, MD, and Michael Silverstein, MD, both of Boston University, said in an accompanying editorial.
However, “Although time and financial barriers are real, it is also likely that some of the residual gaps in guideline-concordant practice reflect variability among pediatricians in their perception of the clinical relevance of certain developmental problems that require formal instruments to identify and in the availability and effectiveness of services targeted to children with these less severe developmental issues,” they said. The path for screening children with developmental risk factors but not obviously severe delays may not be straightforward, and many pediatricians rely on their clinical judgment, they emphasized.
“As important as developing strategies to achieve more widespread developmental screening, therefore, is developing a greater understanding of the root causes of practice variation and determining the range of viable clinical practices that lead to better developmental outcomes,” Dr. Elansary and Dr. Silverstein concluded.
The study was supported by the American Academy of Pediatrics, the Department of Health & Human Services, the Health Resources and Services Administration, and the Department of Education Office of Special Education Programs. The researchers had no financial conflicts to disclose.
Editorialist Dr. Silverstein disclosed an award from the Eunice Kennedy Shriver National Institute of Child Health and Human Development; the editorial was funded by the National Institutes of Health. Dr. Silverstein is a member of the U.S. Preventive Services Task Force. Dr. Elansary had no relevant financial disclosures.
SOURCES: Lipkin PH et al. Pediatrics. 2020 Mar 2. doi: 10.1542/peds.2019-0851; Elansary M, Silverstein M. Pediatrics. 2020 Mar 2. doi: 10.1542/peds.2020-0164.
FROM PEDIATRICS