User login
Lucas Franki is an associate editor for MDedge News, and has been with the company since 2014. He has a BA in English from Penn State University and is an Eagle Scout.
FDA gives orphan drug designation to BIV201 for ascites treatment
The Food and Drug Administration has given an orphan drug designation to the compound BIV201 for the treatment of ascites, according to a press release from the drug’s manufacturer, BioVie.
The FDA designation for BIV201 is for the treatment of ascites due to all etiologies except cancer. Clinical trials could commence by 2017, if accepted by the FDA. If trials are successful, other applications for BIV201 could be tested. BIV201 is a vasoconstricter, and could also be used to treat diseases like type 1 hepatorenal syndrome, esophageal variceal bleeds, and sepsis.
Ascites, a common complication of liver cirrhosis, has no specific approved treatment, and 40% of patients diagnosed with ascites die within 2 years. Other treatments may work initially, but become ineffective as the patient worsens. Treatment costs in the United States for liver cirrhosis and related complications, including ascites, totals over $4 billion.
“Orphan drug designation represents a major milestone supporting the clinical development and eventual commercialization of BIV201 therapy. It recognizes the importance of pioneering a new therapeutic approach for this relatively small group of desperately ill patients,” Jonathan Adams, BioVie CEO, said in the press release.
Find the full press release on the BioVie website.
The Food and Drug Administration has given an orphan drug designation to the compound BIV201 for the treatment of ascites, according to a press release from the drug’s manufacturer, BioVie.
The FDA designation for BIV201 is for the treatment of ascites due to all etiologies except cancer. Clinical trials could commence by 2017, if accepted by the FDA. If trials are successful, other applications for BIV201 could be tested. BIV201 is a vasoconstricter, and could also be used to treat diseases like type 1 hepatorenal syndrome, esophageal variceal bleeds, and sepsis.
Ascites, a common complication of liver cirrhosis, has no specific approved treatment, and 40% of patients diagnosed with ascites die within 2 years. Other treatments may work initially, but become ineffective as the patient worsens. Treatment costs in the United States for liver cirrhosis and related complications, including ascites, totals over $4 billion.
“Orphan drug designation represents a major milestone supporting the clinical development and eventual commercialization of BIV201 therapy. It recognizes the importance of pioneering a new therapeutic approach for this relatively small group of desperately ill patients,” Jonathan Adams, BioVie CEO, said in the press release.
Find the full press release on the BioVie website.
The Food and Drug Administration has given an orphan drug designation to the compound BIV201 for the treatment of ascites, according to a press release from the drug’s manufacturer, BioVie.
The FDA designation for BIV201 is for the treatment of ascites due to all etiologies except cancer. Clinical trials could commence by 2017, if accepted by the FDA. If trials are successful, other applications for BIV201 could be tested. BIV201 is a vasoconstricter, and could also be used to treat diseases like type 1 hepatorenal syndrome, esophageal variceal bleeds, and sepsis.
Ascites, a common complication of liver cirrhosis, has no specific approved treatment, and 40% of patients diagnosed with ascites die within 2 years. Other treatments may work initially, but become ineffective as the patient worsens. Treatment costs in the United States for liver cirrhosis and related complications, including ascites, totals over $4 billion.
“Orphan drug designation represents a major milestone supporting the clinical development and eventual commercialization of BIV201 therapy. It recognizes the importance of pioneering a new therapeutic approach for this relatively small group of desperately ill patients,” Jonathan Adams, BioVie CEO, said in the press release.
Find the full press release on the BioVie website.
No difference in serious infection rates between SLE treatments
There was no significant difference in serious infection and mortality rates between mycophenolate mofetil (MMF), azathioprine (AZA), and cyclophosphamide (CYC) for high-risk systemic lupus erythematosus patients who were newly initiating immunotherapy, according to Candace H. Feldman, MD, and her associates.
In a 6-month analysis of 1,350 systemic lupus erythematosus patients initiating MMF and 1,350 patients initiating AZA, the incidence rate of first hospitalized infection per 100 person-years for MMF users was 14.6, and the incidence rate for AZA was 15.2. In a separate 6-month analysis of 674 patients initiating MMF and 674 patients receiving CYC, the incidence rate of first hospitalized infection per 100 person-years was 24.1 for MMF and 24.6 for CYC. The hazard ratio for AZA was 0.99, and for CYC it was 0.95.
Mortality was also similar in both cohorts. In the MMF vs. AZA cohort, 13 deaths were reported in the MMF group, and 14 were reported in the AZA group. In the MMF vs. CYC cohort, 15 deaths were reported in each group. The hazard ratio for mortality in the AZA group was 0.90, and the HR for mortality in the CYC group was 0.95.
“Based on these findings, concerns about differential infection risks may not need to influence physician and patient choice between MMF vs. AZA and MMF vs. CYC, even in a population highly susceptible to adverse outcomes,” the investigators concluded.
Find the full study in Arthritis & Rheumatology (doi: 10.1002/art.39849).
There was no significant difference in serious infection and mortality rates between mycophenolate mofetil (MMF), azathioprine (AZA), and cyclophosphamide (CYC) for high-risk systemic lupus erythematosus patients who were newly initiating immunotherapy, according to Candace H. Feldman, MD, and her associates.
In a 6-month analysis of 1,350 systemic lupus erythematosus patients initiating MMF and 1,350 patients initiating AZA, the incidence rate of first hospitalized infection per 100 person-years for MMF users was 14.6, and the incidence rate for AZA was 15.2. In a separate 6-month analysis of 674 patients initiating MMF and 674 patients receiving CYC, the incidence rate of first hospitalized infection per 100 person-years was 24.1 for MMF and 24.6 for CYC. The hazard ratio for AZA was 0.99, and for CYC it was 0.95.
Mortality was also similar in both cohorts. In the MMF vs. AZA cohort, 13 deaths were reported in the MMF group, and 14 were reported in the AZA group. In the MMF vs. CYC cohort, 15 deaths were reported in each group. The hazard ratio for mortality in the AZA group was 0.90, and the HR for mortality in the CYC group was 0.95.
“Based on these findings, concerns about differential infection risks may not need to influence physician and patient choice between MMF vs. AZA and MMF vs. CYC, even in a population highly susceptible to adverse outcomes,” the investigators concluded.
Find the full study in Arthritis & Rheumatology (doi: 10.1002/art.39849).
There was no significant difference in serious infection and mortality rates between mycophenolate mofetil (MMF), azathioprine (AZA), and cyclophosphamide (CYC) for high-risk systemic lupus erythematosus patients who were newly initiating immunotherapy, according to Candace H. Feldman, MD, and her associates.
In a 6-month analysis of 1,350 systemic lupus erythematosus patients initiating MMF and 1,350 patients initiating AZA, the incidence rate of first hospitalized infection per 100 person-years for MMF users was 14.6, and the incidence rate for AZA was 15.2. In a separate 6-month analysis of 674 patients initiating MMF and 674 patients receiving CYC, the incidence rate of first hospitalized infection per 100 person-years was 24.1 for MMF and 24.6 for CYC. The hazard ratio for AZA was 0.99, and for CYC it was 0.95.
Mortality was also similar in both cohorts. In the MMF vs. AZA cohort, 13 deaths were reported in the MMF group, and 14 were reported in the AZA group. In the MMF vs. CYC cohort, 15 deaths were reported in each group. The hazard ratio for mortality in the AZA group was 0.90, and the HR for mortality in the CYC group was 0.95.
“Based on these findings, concerns about differential infection risks may not need to influence physician and patient choice between MMF vs. AZA and MMF vs. CYC, even in a population highly susceptible to adverse outcomes,” the investigators concluded.
Find the full study in Arthritis & Rheumatology (doi: 10.1002/art.39849).
FROM ARTHRITIS & RHEUMATOLOGY
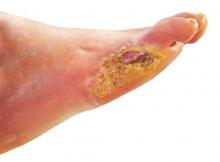
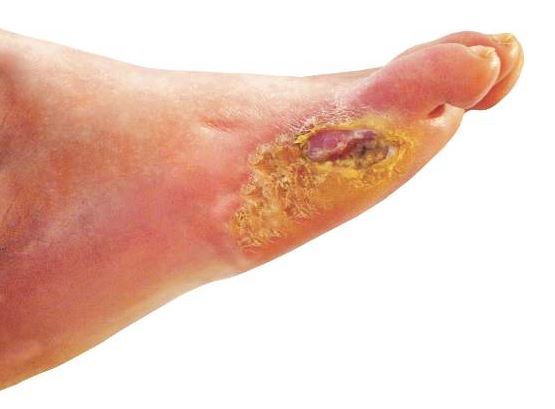
Many patients with diabetic foot infections get unnecessary MRSA treatment
Many patients with diabetic foot infections receive methicillin-resistant Staphylococcus aureus antibiotics unnecessarily, according to Kelly Reveles, PharmD, and her associates.
Among the 318 patients with diabetic foot infections (DFIs) in the study, S. aureus was the most common pathogen, accounting for 146 cases. MRSA accounted for 47 of S. aureus cases, and 15% of overall cases. Although MRSA accounted for a relatively small number of cases, MRSA antibiotics were administered to 86% of all patients, resulting in 71% of all patients receiving the treatment unnecessarily.
Independent risk factors for MRSA DFI were male sex and bone involvement. Other risk factors included previous MRSA infection, more severe infection, and a higher white cell count. The most common comorbidities of DFI were hypertension, dyslipidemia, and obesity.
“The improper use of antibiotics unnecessarily exposes the patient to potential complications of the therapy. Furthermore, the overuse of antibiotics drives antimicrobial resistance and is likely to increase the health care burden,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0161658).
Many patients with diabetic foot infections receive methicillin-resistant Staphylococcus aureus antibiotics unnecessarily, according to Kelly Reveles, PharmD, and her associates.
Among the 318 patients with diabetic foot infections (DFIs) in the study, S. aureus was the most common pathogen, accounting for 146 cases. MRSA accounted for 47 of S. aureus cases, and 15% of overall cases. Although MRSA accounted for a relatively small number of cases, MRSA antibiotics were administered to 86% of all patients, resulting in 71% of all patients receiving the treatment unnecessarily.
Independent risk factors for MRSA DFI were male sex and bone involvement. Other risk factors included previous MRSA infection, more severe infection, and a higher white cell count. The most common comorbidities of DFI were hypertension, dyslipidemia, and obesity.
“The improper use of antibiotics unnecessarily exposes the patient to potential complications of the therapy. Furthermore, the overuse of antibiotics drives antimicrobial resistance and is likely to increase the health care burden,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0161658).
Many patients with diabetic foot infections receive methicillin-resistant Staphylococcus aureus antibiotics unnecessarily, according to Kelly Reveles, PharmD, and her associates.
Among the 318 patients with diabetic foot infections (DFIs) in the study, S. aureus was the most common pathogen, accounting for 146 cases. MRSA accounted for 47 of S. aureus cases, and 15% of overall cases. Although MRSA accounted for a relatively small number of cases, MRSA antibiotics were administered to 86% of all patients, resulting in 71% of all patients receiving the treatment unnecessarily.
Independent risk factors for MRSA DFI were male sex and bone involvement. Other risk factors included previous MRSA infection, more severe infection, and a higher white cell count. The most common comorbidities of DFI were hypertension, dyslipidemia, and obesity.
“The improper use of antibiotics unnecessarily exposes the patient to potential complications of the therapy. Furthermore, the overuse of antibiotics drives antimicrobial resistance and is likely to increase the health care burden,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0161658).
FROM PLOS ONE
New biomarkers, standardization of AMA testing will improve PBC diagnosis
Standardization of liver diagnostic serology and clinical governance will improve understanding and diagnosis of primary biliary cholangitis (PBC), as will new biomarkers, according to a literature review from Nikolaos Gatselis, MD, PhD, and George Dalekos, MD, PhD, of the University of Thessaly, Greece.
Antimitochondrial antibodies are the most important diagnostic tool for the diagnosis of PBC, as AMA is seen in 90%-95% of patients with PBC. Indirect immunofluorescence assays using HEp-2 cells or cryostat sections of rat liver, kidney, and stomach remain the best way to detect AMA, but since the indirect immunofluorescence process cannot be fully automated, further analysis can be performed by immunoblotting. PBC-specific antinuclear antibodies are seen in 50%-70% of PBC cases, and are also important for the diagnosis of PBC.
Several new biomarkers have been proposed for improved diagnosis of PBC in recent years. The biomarkers include the autoantibodies KLHL12 and HK1, genetic markers in HLA regions, metabolomic profiling, miRNAs, and epigenetics.
“Development of clinical governance is of great importance in order to ensure that clinical standards are met, and that processes are in place to ensure continuing improvement and harmonization between laboratories. The guidance should include the whole testing process, beginning from the formulation of a reasonable clinical suspicion and the request of the most appropriate autoantibody test and continuing with handling of biological samples,” the investigators said.
Find the full review in Expert Review of Molecular Diagnostics (doi: 10.1080/14737159.2016.1217159).
Standardization of liver diagnostic serology and clinical governance will improve understanding and diagnosis of primary biliary cholangitis (PBC), as will new biomarkers, according to a literature review from Nikolaos Gatselis, MD, PhD, and George Dalekos, MD, PhD, of the University of Thessaly, Greece.
Antimitochondrial antibodies are the most important diagnostic tool for the diagnosis of PBC, as AMA is seen in 90%-95% of patients with PBC. Indirect immunofluorescence assays using HEp-2 cells or cryostat sections of rat liver, kidney, and stomach remain the best way to detect AMA, but since the indirect immunofluorescence process cannot be fully automated, further analysis can be performed by immunoblotting. PBC-specific antinuclear antibodies are seen in 50%-70% of PBC cases, and are also important for the diagnosis of PBC.
Several new biomarkers have been proposed for improved diagnosis of PBC in recent years. The biomarkers include the autoantibodies KLHL12 and HK1, genetic markers in HLA regions, metabolomic profiling, miRNAs, and epigenetics.
“Development of clinical governance is of great importance in order to ensure that clinical standards are met, and that processes are in place to ensure continuing improvement and harmonization between laboratories. The guidance should include the whole testing process, beginning from the formulation of a reasonable clinical suspicion and the request of the most appropriate autoantibody test and continuing with handling of biological samples,” the investigators said.
Find the full review in Expert Review of Molecular Diagnostics (doi: 10.1080/14737159.2016.1217159).
Standardization of liver diagnostic serology and clinical governance will improve understanding and diagnosis of primary biliary cholangitis (PBC), as will new biomarkers, according to a literature review from Nikolaos Gatselis, MD, PhD, and George Dalekos, MD, PhD, of the University of Thessaly, Greece.
Antimitochondrial antibodies are the most important diagnostic tool for the diagnosis of PBC, as AMA is seen in 90%-95% of patients with PBC. Indirect immunofluorescence assays using HEp-2 cells or cryostat sections of rat liver, kidney, and stomach remain the best way to detect AMA, but since the indirect immunofluorescence process cannot be fully automated, further analysis can be performed by immunoblotting. PBC-specific antinuclear antibodies are seen in 50%-70% of PBC cases, and are also important for the diagnosis of PBC.
Several new biomarkers have been proposed for improved diagnosis of PBC in recent years. The biomarkers include the autoantibodies KLHL12 and HK1, genetic markers in HLA regions, metabolomic profiling, miRNAs, and epigenetics.
“Development of clinical governance is of great importance in order to ensure that clinical standards are met, and that processes are in place to ensure continuing improvement and harmonization between laboratories. The guidance should include the whole testing process, beginning from the formulation of a reasonable clinical suspicion and the request of the most appropriate autoantibody test and continuing with handling of biological samples,” the investigators said.
Find the full review in Expert Review of Molecular Diagnostics (doi: 10.1080/14737159.2016.1217159).
FROM EXPERT REVIEW OF MOLECULAR DIAGNOSTICS
Clot retrieval devices approved for initial ischemic stroke treatment
Two Trevo clot retrieval devices can now be marketed as an initial therapy to reduce paralysis from strokes that are caused by blood clots, according to a press release from the Food and Drug Administration.
Previously, the only first-line treatment approved for acute ischemic stroke was tissue plasminogen activator (TPA) delivered intravenously. The FDA approved Trevo devices based on a clinical trial in which 29% of patients treated with the Trevo device combined with TPA and medical management of blood pressure and disability symptoms were shown to be functionally independent 3 months after their stroke, compared with only 19% of patients treated with TPA plus medical management alone.
The Trevo devices are approved for usage within 6 hours of symptom onset and only following treatment with TPA, which should be administered within 3 hours of stroke onset. Associated risks with Trevo device usage include failure to retrieve the blood clot, device malfunctions including breakage and navigation difficulties, potential damage of blood vessels, and the chance of perforation or hemorrhage.
The Trevo device was first approved by the FDA in 2012 to remove blood clots in order to restore blood flow in stroke patients who could not receive TPA or for those patients who did not respond to TPA therapy. The current approval expands the devices’ indication to a broader group of patients, according to the release.
“This is the first time FDA has allowed the use of these devices alongside TPA, which has the potential to help further reduce the devastating disabilities associated with strokes compared to the use of TPA alone. Now health care providers and their patients have another tool for treating stroke and potentially preventing long-term disability,” Carlos Peña, PhD, director of the division of neurological and physical medicine devices at the FDA’s Center for Devices and Radiological Health, said in the press release.
Find the full press release on the FDA website.
Two Trevo clot retrieval devices can now be marketed as an initial therapy to reduce paralysis from strokes that are caused by blood clots, according to a press release from the Food and Drug Administration.
Previously, the only first-line treatment approved for acute ischemic stroke was tissue plasminogen activator (TPA) delivered intravenously. The FDA approved Trevo devices based on a clinical trial in which 29% of patients treated with the Trevo device combined with TPA and medical management of blood pressure and disability symptoms were shown to be functionally independent 3 months after their stroke, compared with only 19% of patients treated with TPA plus medical management alone.
The Trevo devices are approved for usage within 6 hours of symptom onset and only following treatment with TPA, which should be administered within 3 hours of stroke onset. Associated risks with Trevo device usage include failure to retrieve the blood clot, device malfunctions including breakage and navigation difficulties, potential damage of blood vessels, and the chance of perforation or hemorrhage.
The Trevo device was first approved by the FDA in 2012 to remove blood clots in order to restore blood flow in stroke patients who could not receive TPA or for those patients who did not respond to TPA therapy. The current approval expands the devices’ indication to a broader group of patients, according to the release.
“This is the first time FDA has allowed the use of these devices alongside TPA, which has the potential to help further reduce the devastating disabilities associated with strokes compared to the use of TPA alone. Now health care providers and their patients have another tool for treating stroke and potentially preventing long-term disability,” Carlos Peña, PhD, director of the division of neurological and physical medicine devices at the FDA’s Center for Devices and Radiological Health, said in the press release.
Find the full press release on the FDA website.
Two Trevo clot retrieval devices can now be marketed as an initial therapy to reduce paralysis from strokes that are caused by blood clots, according to a press release from the Food and Drug Administration.
Previously, the only first-line treatment approved for acute ischemic stroke was tissue plasminogen activator (TPA) delivered intravenously. The FDA approved Trevo devices based on a clinical trial in which 29% of patients treated with the Trevo device combined with TPA and medical management of blood pressure and disability symptoms were shown to be functionally independent 3 months after their stroke, compared with only 19% of patients treated with TPA plus medical management alone.
The Trevo devices are approved for usage within 6 hours of symptom onset and only following treatment with TPA, which should be administered within 3 hours of stroke onset. Associated risks with Trevo device usage include failure to retrieve the blood clot, device malfunctions including breakage and navigation difficulties, potential damage of blood vessels, and the chance of perforation or hemorrhage.
The Trevo device was first approved by the FDA in 2012 to remove blood clots in order to restore blood flow in stroke patients who could not receive TPA or for those patients who did not respond to TPA therapy. The current approval expands the devices’ indication to a broader group of patients, according to the release.
“This is the first time FDA has allowed the use of these devices alongside TPA, which has the potential to help further reduce the devastating disabilities associated with strokes compared to the use of TPA alone. Now health care providers and their patients have another tool for treating stroke and potentially preventing long-term disability,” Carlos Peña, PhD, director of the division of neurological and physical medicine devices at the FDA’s Center for Devices and Radiological Health, said in the press release.
Find the full press release on the FDA website.
AAP: MenB vaccines are safe for healthy adolescents, young adults
The serotype B meningococcal vaccines MenB-FHbp and MenB-4C are safe and can be administered to healthy people aged 10-25 years, according to a policy statement from the American Academy of Pediatrics Committee on Infectious Diseases.
The AAP recommends that people older than 10 years at increased risk for serogroup B meningococcal disease (category A) should receive MenB vaccines regularly. Category A includes people with persistent complement component deficiencies, people with anatomic or functional asplenia, and healthy people at increased risk because of a disease outbreak.
Young adults aged 16-23 years old may receive a vaccination, but it is not routinely recommended (category B), with a preferred vaccination age between 16 and 18 years.
Annual incidence of serogroup B meningococcal disease in people aged 11-24 years in the United States is about 50-60 cases per year, and a routine vaccination program would prevent 15-29 cases and 2-5 deaths per year, the researchers noted. The cost of routine vaccination in the general population would range from $3.7 million per quality-adjusted life year (QALY) to $9.4 million per QALY.
Both MenB-FHbp and MenB-4C have been safely administered in clinical trials, with no deaths related to either vaccine. Data on duration of immunogenicity and proportion of MenB strains covered by vaccines in different geographic regions remain incomplete, and both vaccine manufacturers must complete postmarketing studies to determine overall vaccine effectiveness.
“Pediatricians are encouraged to discuss the availability of the MenB vaccines with families. Discussion should include the low incidence of MenB disease and the unknown efficacy of the vaccines... The treating clinician should discuss the benefits, risks, and costs with patients and their families and then work with them to determine what is in their best interest,” the AAP committee noted.
Find the full study in Pediatrics (doi: 10.1542/peds.2016-1890).
The serotype B meningococcal vaccines MenB-FHbp and MenB-4C are safe and can be administered to healthy people aged 10-25 years, according to a policy statement from the American Academy of Pediatrics Committee on Infectious Diseases.
The AAP recommends that people older than 10 years at increased risk for serogroup B meningococcal disease (category A) should receive MenB vaccines regularly. Category A includes people with persistent complement component deficiencies, people with anatomic or functional asplenia, and healthy people at increased risk because of a disease outbreak.
Young adults aged 16-23 years old may receive a vaccination, but it is not routinely recommended (category B), with a preferred vaccination age between 16 and 18 years.
Annual incidence of serogroup B meningococcal disease in people aged 11-24 years in the United States is about 50-60 cases per year, and a routine vaccination program would prevent 15-29 cases and 2-5 deaths per year, the researchers noted. The cost of routine vaccination in the general population would range from $3.7 million per quality-adjusted life year (QALY) to $9.4 million per QALY.
Both MenB-FHbp and MenB-4C have been safely administered in clinical trials, with no deaths related to either vaccine. Data on duration of immunogenicity and proportion of MenB strains covered by vaccines in different geographic regions remain incomplete, and both vaccine manufacturers must complete postmarketing studies to determine overall vaccine effectiveness.
“Pediatricians are encouraged to discuss the availability of the MenB vaccines with families. Discussion should include the low incidence of MenB disease and the unknown efficacy of the vaccines... The treating clinician should discuss the benefits, risks, and costs with patients and their families and then work with them to determine what is in their best interest,” the AAP committee noted.
Find the full study in Pediatrics (doi: 10.1542/peds.2016-1890).
The serotype B meningococcal vaccines MenB-FHbp and MenB-4C are safe and can be administered to healthy people aged 10-25 years, according to a policy statement from the American Academy of Pediatrics Committee on Infectious Diseases.
The AAP recommends that people older than 10 years at increased risk for serogroup B meningococcal disease (category A) should receive MenB vaccines regularly. Category A includes people with persistent complement component deficiencies, people with anatomic or functional asplenia, and healthy people at increased risk because of a disease outbreak.
Young adults aged 16-23 years old may receive a vaccination, but it is not routinely recommended (category B), with a preferred vaccination age between 16 and 18 years.
Annual incidence of serogroup B meningococcal disease in people aged 11-24 years in the United States is about 50-60 cases per year, and a routine vaccination program would prevent 15-29 cases and 2-5 deaths per year, the researchers noted. The cost of routine vaccination in the general population would range from $3.7 million per quality-adjusted life year (QALY) to $9.4 million per QALY.
Both MenB-FHbp and MenB-4C have been safely administered in clinical trials, with no deaths related to either vaccine. Data on duration of immunogenicity and proportion of MenB strains covered by vaccines in different geographic regions remain incomplete, and both vaccine manufacturers must complete postmarketing studies to determine overall vaccine effectiveness.
“Pediatricians are encouraged to discuss the availability of the MenB vaccines with families. Discussion should include the low incidence of MenB disease and the unknown efficacy of the vaccines... The treating clinician should discuss the benefits, risks, and costs with patients and their families and then work with them to determine what is in their best interest,” the AAP committee noted.
Find the full study in Pediatrics (doi: 10.1542/peds.2016-1890).
FROM PEDIATRICS
PAI-1 modifications, early-life LRIs increase asthma risk
A genetic modification of the plasminogen activator inhibitor-1 gene in conjunction with lower respiratory infections during early life was associated with increased risk of asthma, morbidities, and reduced lung function, according to Seong H. Cho, MD, and his associates.
A history of respiratory syncytial virus (RSV) and a history of other lower respiratory infections (LRIs) before the age of 2 were independently associated with asthma in Latino people aged 8-21, with odd ratios of 9.9 and 9.1, respectively, while PAI-1 was not independently associated. In combination, the OR for PAI-1/RSV increased to 17.7, and the OR for PAI-1/other LRIs increased to 11.7.
Lung function was also adversely affected by the joint effect of PAI-1 and early life infection. In patients with PAI-1/LRI, forced expiratory volume in 1 second (FEV1) percent predicted and FEV1/forced vital capacity (FVC) percent predicted were significantly less than in the control group. Similar but less significant results were seen in the PAI-1/RSV group. Recurring hospitalizations were also significantly more likely in the PAI-1/RSV group, with an OR of 3.1.
“Further prospective studies are needed to replicate our RSV-genotype findings in other non-Latino populations, and determine if PAI-1 variants may serve as a biomarker of risk, which may provide impetus for clinical trials of primary prevention of asthma. In the interim, PAI-1 genotype in combination with significant LRI identifies individuals at increased risk of developing asthma,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0157848).
A genetic modification of the plasminogen activator inhibitor-1 gene in conjunction with lower respiratory infections during early life was associated with increased risk of asthma, morbidities, and reduced lung function, according to Seong H. Cho, MD, and his associates.
A history of respiratory syncytial virus (RSV) and a history of other lower respiratory infections (LRIs) before the age of 2 were independently associated with asthma in Latino people aged 8-21, with odd ratios of 9.9 and 9.1, respectively, while PAI-1 was not independently associated. In combination, the OR for PAI-1/RSV increased to 17.7, and the OR for PAI-1/other LRIs increased to 11.7.
Lung function was also adversely affected by the joint effect of PAI-1 and early life infection. In patients with PAI-1/LRI, forced expiratory volume in 1 second (FEV1) percent predicted and FEV1/forced vital capacity (FVC) percent predicted were significantly less than in the control group. Similar but less significant results were seen in the PAI-1/RSV group. Recurring hospitalizations were also significantly more likely in the PAI-1/RSV group, with an OR of 3.1.
“Further prospective studies are needed to replicate our RSV-genotype findings in other non-Latino populations, and determine if PAI-1 variants may serve as a biomarker of risk, which may provide impetus for clinical trials of primary prevention of asthma. In the interim, PAI-1 genotype in combination with significant LRI identifies individuals at increased risk of developing asthma,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0157848).
A genetic modification of the plasminogen activator inhibitor-1 gene in conjunction with lower respiratory infections during early life was associated with increased risk of asthma, morbidities, and reduced lung function, according to Seong H. Cho, MD, and his associates.
A history of respiratory syncytial virus (RSV) and a history of other lower respiratory infections (LRIs) before the age of 2 were independently associated with asthma in Latino people aged 8-21, with odd ratios of 9.9 and 9.1, respectively, while PAI-1 was not independently associated. In combination, the OR for PAI-1/RSV increased to 17.7, and the OR for PAI-1/other LRIs increased to 11.7.
Lung function was also adversely affected by the joint effect of PAI-1 and early life infection. In patients with PAI-1/LRI, forced expiratory volume in 1 second (FEV1) percent predicted and FEV1/forced vital capacity (FVC) percent predicted were significantly less than in the control group. Similar but less significant results were seen in the PAI-1/RSV group. Recurring hospitalizations were also significantly more likely in the PAI-1/RSV group, with an OR of 3.1.
“Further prospective studies are needed to replicate our RSV-genotype findings in other non-Latino populations, and determine if PAI-1 variants may serve as a biomarker of risk, which may provide impetus for clinical trials of primary prevention of asthma. In the interim, PAI-1 genotype in combination with significant LRI identifies individuals at increased risk of developing asthma,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0157848).
FROM PLOS ONE
Thymidine phosphorylase increases myeloma-induced bone lesions
Myeloma-induced osteolytic bone lesions were reduced by suppressing expression of thymidine phosphorylase (TP), according to Huan Liu of the University of Texas MD Anderson Cancer Center in Houston and associates.
In osteoblast progenitors, methylation of alpha-1/runt-related transcription factor 2 (RUNX2) and osterix was upregulated by TP, resulting in decreased bone formation. TP also upregulated methylation of interferon regulatory factor 8 (IRF8) and enhanced nuclear factor of activated T cells, cytoplasmic 1 protein (NFATc1), which increased bone resorption. Thymidine was catalyzed into thymine and 2-deoxy-d-ribose, which bound to integrins alphavbeta3 and alpha5beta1, activated PI3K/Akt signaling, and increased DNA methyltransferase 3A (DNMT3A) expression, the investigators found.
In an experiment, myeloma was established in severe combined immunodeficient mice, and the mice were injected with ARP-1 cells, which produce high levels of TP. The mice were then treated with 7-deazaxanthine or tipiracil hydrochloride, which reduced ARP-1-induced bone lesions, DNMT3A expression, and 2DDR levels in the serum of tumor-bearing mice.
“This model could facilitate the translation of these inhibitors into human studies of myeloma bone disease. Because TP is often expressed by other malignancies including breast, prostate, and lung cancer, these findings may also have broader implications for the genesis of bone metastasis caused by these and other tumors,” the investigators concluded.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aad8949)
Myeloma-induced osteolytic bone lesions were reduced by suppressing expression of thymidine phosphorylase (TP), according to Huan Liu of the University of Texas MD Anderson Cancer Center in Houston and associates.
In osteoblast progenitors, methylation of alpha-1/runt-related transcription factor 2 (RUNX2) and osterix was upregulated by TP, resulting in decreased bone formation. TP also upregulated methylation of interferon regulatory factor 8 (IRF8) and enhanced nuclear factor of activated T cells, cytoplasmic 1 protein (NFATc1), which increased bone resorption. Thymidine was catalyzed into thymine and 2-deoxy-d-ribose, which bound to integrins alphavbeta3 and alpha5beta1, activated PI3K/Akt signaling, and increased DNA methyltransferase 3A (DNMT3A) expression, the investigators found.
In an experiment, myeloma was established in severe combined immunodeficient mice, and the mice were injected with ARP-1 cells, which produce high levels of TP. The mice were then treated with 7-deazaxanthine or tipiracil hydrochloride, which reduced ARP-1-induced bone lesions, DNMT3A expression, and 2DDR levels in the serum of tumor-bearing mice.
“This model could facilitate the translation of these inhibitors into human studies of myeloma bone disease. Because TP is often expressed by other malignancies including breast, prostate, and lung cancer, these findings may also have broader implications for the genesis of bone metastasis caused by these and other tumors,” the investigators concluded.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aad8949)
Myeloma-induced osteolytic bone lesions were reduced by suppressing expression of thymidine phosphorylase (TP), according to Huan Liu of the University of Texas MD Anderson Cancer Center in Houston and associates.
In osteoblast progenitors, methylation of alpha-1/runt-related transcription factor 2 (RUNX2) and osterix was upregulated by TP, resulting in decreased bone formation. TP also upregulated methylation of interferon regulatory factor 8 (IRF8) and enhanced nuclear factor of activated T cells, cytoplasmic 1 protein (NFATc1), which increased bone resorption. Thymidine was catalyzed into thymine and 2-deoxy-d-ribose, which bound to integrins alphavbeta3 and alpha5beta1, activated PI3K/Akt signaling, and increased DNA methyltransferase 3A (DNMT3A) expression, the investigators found.
In an experiment, myeloma was established in severe combined immunodeficient mice, and the mice were injected with ARP-1 cells, which produce high levels of TP. The mice were then treated with 7-deazaxanthine or tipiracil hydrochloride, which reduced ARP-1-induced bone lesions, DNMT3A expression, and 2DDR levels in the serum of tumor-bearing mice.
“This model could facilitate the translation of these inhibitors into human studies of myeloma bone disease. Because TP is often expressed by other malignancies including breast, prostate, and lung cancer, these findings may also have broader implications for the genesis of bone metastasis caused by these and other tumors,” the investigators concluded.
Find the full study in Science Translational Medicine (doi:10.1126/scitranslmed.aad8949)
FROM SCIENCE TRANSLATIONAL MEDICINE
Predisposition to overlapping AIH and PBC can exist within families
A genetic predisposition to autoimmune liver disease can cause overlapping cases of autoimmune hepatitis (AIH) and primary biliary cholangitis (PBC) within the same family, according to a case report from Japanese researchers.
A 68-year-old woman and her 49-year-old daughter were admitted to the hospital for liver dysfunction, 4 years apart. The mother had been diagnosed with PBC at a previous hospital, but was moved after treatment with 600 mg ursodeoxycholic acid (UDCA) was ineffective. The daughter had no previous diagnosis and had a variety of symptoms at admission, including jaundice, general fatigue, and darkness of the urine.
Both the mother and the daughter were negative for hepatitis A, B, and C. A simplified International Autoimmune Hepatitis Group score revealed probable AIH for both patients, and both patients were diagnosed with autoimmune liver disease with overlapping features of PBC and AIH. Treatment with UDCA and prednisolone was effective for both patients.
In a familial study, the mother and her two daughters were tested for liver function, autoantibodies, and human leukocyte antigen (HLA) haplotype. The second daughter, who was healthy with no history of liver dysfunction, had normal liver function, negative ANA, and positive AMA-M2 antibody, but had an HLA haplotype different from that of the mother and the first daughter. No difference in medication use, smoking status, alcohol consumption, or obstetric history was seen.
“The exact mechanism underlying the onset of PBC and AIH overlap within the same family remains unclear. Therefore, it is very important to monitor the healthy second daughter closely as she was positive for the AMA-M2 antibody, as this might yield further knowledge with regard to what factors influence the onset of PBC and AIH overlap within the same family,” the investigators noted.
Find the full study in the Journal of Clinical Gastroenterology (doi: 10.1007/s12328-016-0676-1)
A genetic predisposition to autoimmune liver disease can cause overlapping cases of autoimmune hepatitis (AIH) and primary biliary cholangitis (PBC) within the same family, according to a case report from Japanese researchers.
A 68-year-old woman and her 49-year-old daughter were admitted to the hospital for liver dysfunction, 4 years apart. The mother had been diagnosed with PBC at a previous hospital, but was moved after treatment with 600 mg ursodeoxycholic acid (UDCA) was ineffective. The daughter had no previous diagnosis and had a variety of symptoms at admission, including jaundice, general fatigue, and darkness of the urine.
Both the mother and the daughter were negative for hepatitis A, B, and C. A simplified International Autoimmune Hepatitis Group score revealed probable AIH for both patients, and both patients were diagnosed with autoimmune liver disease with overlapping features of PBC and AIH. Treatment with UDCA and prednisolone was effective for both patients.
In a familial study, the mother and her two daughters were tested for liver function, autoantibodies, and human leukocyte antigen (HLA) haplotype. The second daughter, who was healthy with no history of liver dysfunction, had normal liver function, negative ANA, and positive AMA-M2 antibody, but had an HLA haplotype different from that of the mother and the first daughter. No difference in medication use, smoking status, alcohol consumption, or obstetric history was seen.
“The exact mechanism underlying the onset of PBC and AIH overlap within the same family remains unclear. Therefore, it is very important to monitor the healthy second daughter closely as she was positive for the AMA-M2 antibody, as this might yield further knowledge with regard to what factors influence the onset of PBC and AIH overlap within the same family,” the investigators noted.
Find the full study in the Journal of Clinical Gastroenterology (doi: 10.1007/s12328-016-0676-1)
A genetic predisposition to autoimmune liver disease can cause overlapping cases of autoimmune hepatitis (AIH) and primary biliary cholangitis (PBC) within the same family, according to a case report from Japanese researchers.
A 68-year-old woman and her 49-year-old daughter were admitted to the hospital for liver dysfunction, 4 years apart. The mother had been diagnosed with PBC at a previous hospital, but was moved after treatment with 600 mg ursodeoxycholic acid (UDCA) was ineffective. The daughter had no previous diagnosis and had a variety of symptoms at admission, including jaundice, general fatigue, and darkness of the urine.
Both the mother and the daughter were negative for hepatitis A, B, and C. A simplified International Autoimmune Hepatitis Group score revealed probable AIH for both patients, and both patients were diagnosed with autoimmune liver disease with overlapping features of PBC and AIH. Treatment with UDCA and prednisolone was effective for both patients.
In a familial study, the mother and her two daughters were tested for liver function, autoantibodies, and human leukocyte antigen (HLA) haplotype. The second daughter, who was healthy with no history of liver dysfunction, had normal liver function, negative ANA, and positive AMA-M2 antibody, but had an HLA haplotype different from that of the mother and the first daughter. No difference in medication use, smoking status, alcohol consumption, or obstetric history was seen.
“The exact mechanism underlying the onset of PBC and AIH overlap within the same family remains unclear. Therefore, it is very important to monitor the healthy second daughter closely as she was positive for the AMA-M2 antibody, as this might yield further knowledge with regard to what factors influence the onset of PBC and AIH overlap within the same family,” the investigators noted.
Find the full study in the Journal of Clinical Gastroenterology (doi: 10.1007/s12328-016-0676-1)
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
PCR assay quicker but less sensitive at penicilliosis diagnosis
A real-time PCR assay was effective at rapidly diagnosing penicilliosis caused by Talaromyces marneffei, according to Thuy Le, MD, and her associates.
Sensitivity of the assay was better when samples were collected from plasma prior to antifungal therapy. In a group of 27 HIV-infected patients from whom samples were collected prior to antifungal therapy, the assay detected the T. marneffei MP1 gene in 19 samples, while in a group of 23 HIV-infected patients from whom samples were collected within 48 hours of antifungal therapy, the assay successfully detected the MP1 gene in 12 samples.
In an additional sample of 20 HIV-infected patients without penicilliosis, the assay found no signals of the T. marneffei MP1 gene in any of the tested plasma samples, giving a specificity of 100%. All testing was completed within 5-6 hours, significantly less than the 5 days needed for Bactec system testing.
“This real-time PCR assay should not replace the need for conventional microbiology methods in diagnosing penicilliosis. However, in conjunction with culturing, it can be used as a rapid rule-in test that can make a significant difference in patient management by allowing antifungal therapy to begin sooner, particularly in patients without skin lesions, and has the potential to improve the outcomes of T. marneffei–infected patients,” the investigators concluded.
Find the full study in Mycoses (doi: 10.1111/myc.12530).
A real-time PCR assay was effective at rapidly diagnosing penicilliosis caused by Talaromyces marneffei, according to Thuy Le, MD, and her associates.
Sensitivity of the assay was better when samples were collected from plasma prior to antifungal therapy. In a group of 27 HIV-infected patients from whom samples were collected prior to antifungal therapy, the assay detected the T. marneffei MP1 gene in 19 samples, while in a group of 23 HIV-infected patients from whom samples were collected within 48 hours of antifungal therapy, the assay successfully detected the MP1 gene in 12 samples.
In an additional sample of 20 HIV-infected patients without penicilliosis, the assay found no signals of the T. marneffei MP1 gene in any of the tested plasma samples, giving a specificity of 100%. All testing was completed within 5-6 hours, significantly less than the 5 days needed for Bactec system testing.
“This real-time PCR assay should not replace the need for conventional microbiology methods in diagnosing penicilliosis. However, in conjunction with culturing, it can be used as a rapid rule-in test that can make a significant difference in patient management by allowing antifungal therapy to begin sooner, particularly in patients without skin lesions, and has the potential to improve the outcomes of T. marneffei–infected patients,” the investigators concluded.
Find the full study in Mycoses (doi: 10.1111/myc.12530).
A real-time PCR assay was effective at rapidly diagnosing penicilliosis caused by Talaromyces marneffei, according to Thuy Le, MD, and her associates.
Sensitivity of the assay was better when samples were collected from plasma prior to antifungal therapy. In a group of 27 HIV-infected patients from whom samples were collected prior to antifungal therapy, the assay detected the T. marneffei MP1 gene in 19 samples, while in a group of 23 HIV-infected patients from whom samples were collected within 48 hours of antifungal therapy, the assay successfully detected the MP1 gene in 12 samples.
In an additional sample of 20 HIV-infected patients without penicilliosis, the assay found no signals of the T. marneffei MP1 gene in any of the tested plasma samples, giving a specificity of 100%. All testing was completed within 5-6 hours, significantly less than the 5 days needed for Bactec system testing.
“This real-time PCR assay should not replace the need for conventional microbiology methods in diagnosing penicilliosis. However, in conjunction with culturing, it can be used as a rapid rule-in test that can make a significant difference in patient management by allowing antifungal therapy to begin sooner, particularly in patients without skin lesions, and has the potential to improve the outcomes of T. marneffei–infected patients,” the investigators concluded.
Find the full study in Mycoses (doi: 10.1111/myc.12530).
FROM MYCOSES