User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Children and COVID: Weekly cases fall to lowest level in over a year
With the third autumn of the COVID era now upon us, the discussion has turned again to a possible influenza/COVID twindemic, as well as the new-for-2022 influenza/COVID/respiratory syncytial virus tripledemic. It appears, however, that COVID may have missed the memo.
For the sixth time in the last 7 weeks, the number of new COVID cases in children fell, with just under 23,000 reported during the week of Oct. 14-20, according to the American Academy of Pediatrics and the Children’s Hospital Association. That is the lowest weekly count so far this year, and the lowest since early July of 2021, just as the Delta surge was starting. New pediatric cases had dipped to 8,500, the lowest for any week during the pandemic, a couple of weeks before that, the AAP/CHA data show.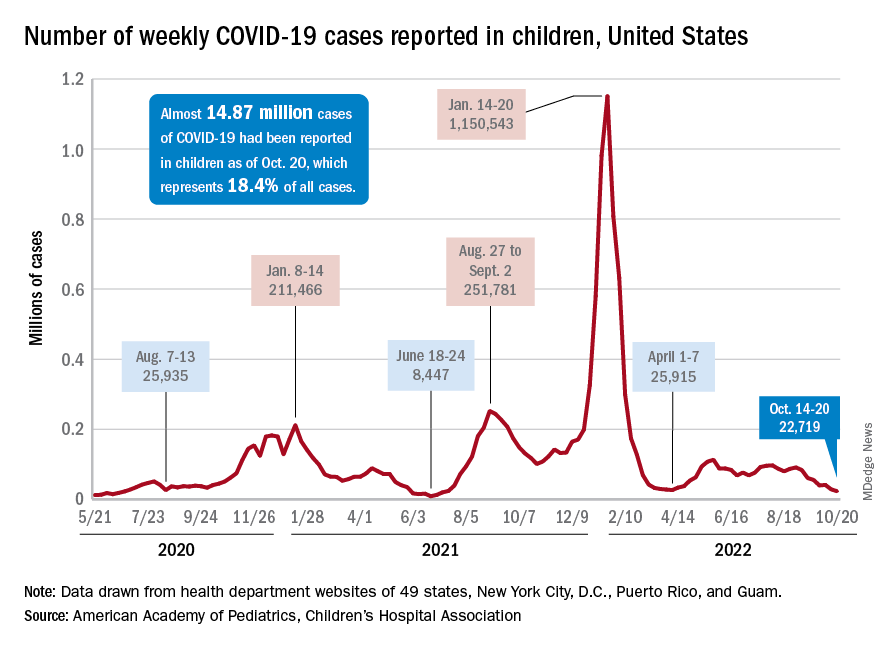
Weekly cases have fallen by almost 75% since over 90,000 were reported for the week of Aug. 26 to Sept. 1, even as children have returned to school and vaccine uptake remains slow in the youngest age groups. Rates of emergency department visits with diagnosed COVID also have continued to drop, as have new admissions, and both are nearing their 2021 lows, according to the Centers for Disease Control and Prevention.
New vaccinations in children under age 5 years were up slightly for the most recent week (Oct. 13-19), but total uptake for that age group is only 7.1% for an initial dose and 2.9% for full vaccination. Among children aged 5-11 years, 38.7% have received at least one dose and 31.6% have completed the primary series, with corresponding figures of 71.2% and 60.9% for those aged 12-17, the CDC said on its COVID Data Tracker.
Despite the low overall numbers, though, the youngest children are, in one respect, punching above their weight when it comes to vaccinations. In the 2 weeks from Oct. 6 to Oct. 19, children under 5 years of age, who represent 5.9% of the U.S. population, received 9.2% of the initial vaccine doses administered. Children aged 5-11 years, who represent 8.7% of the total population, got just 4.2% of all first doses over those same 2 weeks, while 12- to 17-year-olds, who make up 7.6% of the population, got 3.4% of the vaccine doses, the CDC reported.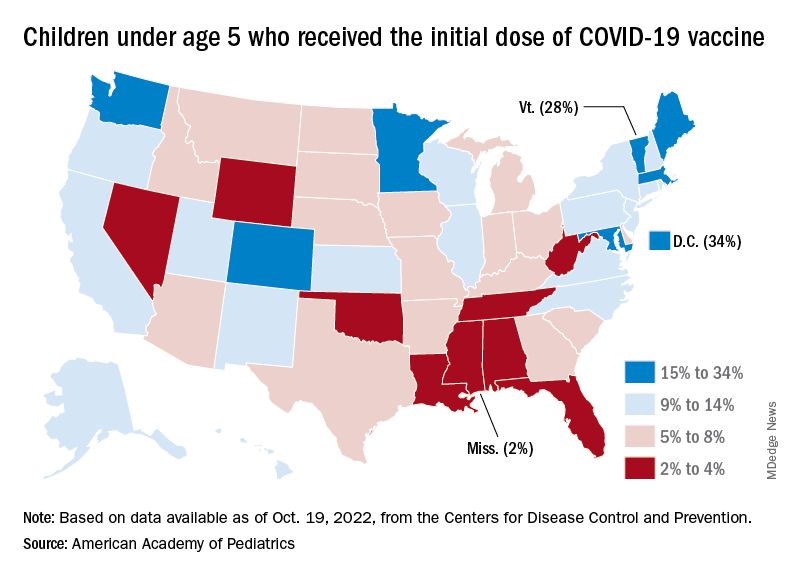
On the vaccine-approval front, the Food and Drug Administration recently announced that the new bivalent COVID-19 vaccines are now included in the emergency use authorizations for children who have completed primary or booster vaccination. The Moderna vaccine is authorized as a single-dose booster for children as young as 6 years and the Pfizer-BioNTech vaccine can be given as a single booster dose in children as young as 5 years, the FDA said.
“These bivalent COVID-19 vaccines include an mRNA component of the original strain to provide an immune response that is broadly protective against COVID-19 and an mRNA component in common between the omicron variant BA.4 and BA.5 lineages,” the FDA said.
With the third autumn of the COVID era now upon us, the discussion has turned again to a possible influenza/COVID twindemic, as well as the new-for-2022 influenza/COVID/respiratory syncytial virus tripledemic. It appears, however, that COVID may have missed the memo.
For the sixth time in the last 7 weeks, the number of new COVID cases in children fell, with just under 23,000 reported during the week of Oct. 14-20, according to the American Academy of Pediatrics and the Children’s Hospital Association. That is the lowest weekly count so far this year, and the lowest since early July of 2021, just as the Delta surge was starting. New pediatric cases had dipped to 8,500, the lowest for any week during the pandemic, a couple of weeks before that, the AAP/CHA data show.
Weekly cases have fallen by almost 75% since over 90,000 were reported for the week of Aug. 26 to Sept. 1, even as children have returned to school and vaccine uptake remains slow in the youngest age groups. Rates of emergency department visits with diagnosed COVID also have continued to drop, as have new admissions, and both are nearing their 2021 lows, according to the Centers for Disease Control and Prevention.
New vaccinations in children under age 5 years were up slightly for the most recent week (Oct. 13-19), but total uptake for that age group is only 7.1% for an initial dose and 2.9% for full vaccination. Among children aged 5-11 years, 38.7% have received at least one dose and 31.6% have completed the primary series, with corresponding figures of 71.2% and 60.9% for those aged 12-17, the CDC said on its COVID Data Tracker.
Despite the low overall numbers, though, the youngest children are, in one respect, punching above their weight when it comes to vaccinations. In the 2 weeks from Oct. 6 to Oct. 19, children under 5 years of age, who represent 5.9% of the U.S. population, received 9.2% of the initial vaccine doses administered. Children aged 5-11 years, who represent 8.7% of the total population, got just 4.2% of all first doses over those same 2 weeks, while 12- to 17-year-olds, who make up 7.6% of the population, got 3.4% of the vaccine doses, the CDC reported.
On the vaccine-approval front, the Food and Drug Administration recently announced that the new bivalent COVID-19 vaccines are now included in the emergency use authorizations for children who have completed primary or booster vaccination. The Moderna vaccine is authorized as a single-dose booster for children as young as 6 years and the Pfizer-BioNTech vaccine can be given as a single booster dose in children as young as 5 years, the FDA said.
“These bivalent COVID-19 vaccines include an mRNA component of the original strain to provide an immune response that is broadly protective against COVID-19 and an mRNA component in common between the omicron variant BA.4 and BA.5 lineages,” the FDA said.
With the third autumn of the COVID era now upon us, the discussion has turned again to a possible influenza/COVID twindemic, as well as the new-for-2022 influenza/COVID/respiratory syncytial virus tripledemic. It appears, however, that COVID may have missed the memo.
For the sixth time in the last 7 weeks, the number of new COVID cases in children fell, with just under 23,000 reported during the week of Oct. 14-20, according to the American Academy of Pediatrics and the Children’s Hospital Association. That is the lowest weekly count so far this year, and the lowest since early July of 2021, just as the Delta surge was starting. New pediatric cases had dipped to 8,500, the lowest for any week during the pandemic, a couple of weeks before that, the AAP/CHA data show.
Weekly cases have fallen by almost 75% since over 90,000 were reported for the week of Aug. 26 to Sept. 1, even as children have returned to school and vaccine uptake remains slow in the youngest age groups. Rates of emergency department visits with diagnosed COVID also have continued to drop, as have new admissions, and both are nearing their 2021 lows, according to the Centers for Disease Control and Prevention.
New vaccinations in children under age 5 years were up slightly for the most recent week (Oct. 13-19), but total uptake for that age group is only 7.1% for an initial dose and 2.9% for full vaccination. Among children aged 5-11 years, 38.7% have received at least one dose and 31.6% have completed the primary series, with corresponding figures of 71.2% and 60.9% for those aged 12-17, the CDC said on its COVID Data Tracker.
Despite the low overall numbers, though, the youngest children are, in one respect, punching above their weight when it comes to vaccinations. In the 2 weeks from Oct. 6 to Oct. 19, children under 5 years of age, who represent 5.9% of the U.S. population, received 9.2% of the initial vaccine doses administered. Children aged 5-11 years, who represent 8.7% of the total population, got just 4.2% of all first doses over those same 2 weeks, while 12- to 17-year-olds, who make up 7.6% of the population, got 3.4% of the vaccine doses, the CDC reported.
On the vaccine-approval front, the Food and Drug Administration recently announced that the new bivalent COVID-19 vaccines are now included in the emergency use authorizations for children who have completed primary or booster vaccination. The Moderna vaccine is authorized as a single-dose booster for children as young as 6 years and the Pfizer-BioNTech vaccine can be given as a single booster dose in children as young as 5 years, the FDA said.
“These bivalent COVID-19 vaccines include an mRNA component of the original strain to provide an immune response that is broadly protective against COVID-19 and an mRNA component in common between the omicron variant BA.4 and BA.5 lineages,” the FDA said.
Time to ditch clarithromycin for H. pylori?
Rates of resistance to clarithromycin among Helicobacter pylori isolates in the United States and Europe are high enough to warrant discontinuation of empiric use of proton pump inhibitor (PPI)–based triple therapy that includes the antibiotic in these regions, a new study has found.
Overall, 22.2% of participants were resistant to clarithromycin – a rate that is above the currently recommended threshold of 15% or higher for avoidance of PPI-based triple therapy that includes clarithromycin.
, study investigator William Chey, MD, professor and chief, Division of Gastroenterology and Hepatology, Michigan Medicine, Ann Arbor, said in an interview.
Judith Kim, MD, a gastroenterologist at NYU Langone Health and clinical instructor of medicine at NYU Grossman School of Medicine, who wasn’t involved in the study, agrees.
“The use of PPI-based triple therapy is still common practice despite recent recommendations to avoid clarithromycin in areas with high resistance rates,” Dr. Kim told this news organization.
“This study shows that multiple parts of the United States and Europe have high resistance rates,” rendering clarithromycin-based regimens “more likely to ineffectively eradicate H pylori,” Dr. Kim said.
The study was published online in The American Journal of Gastroenterology.
Better options now available
Guidelines advise against the use of PPI-based triple regimens with clarithromycin for H. pylori infection in areas where resistance is 15% or higher or for patients who have previously received macrolides. However, up-to-date information on H. pylori antimicrobial resistance patterns is limited, especially in the United States.
Dr. Chey and colleagues assessed resistance rates to antibiotics commonly used to treat H. pylori in isolates from 907 adults with the infection in the United States and Europe. They included four U.S. subregions and five participating European countries.
In all U.S. subregions and European countries, clarithromycin resistance rates were above 15% except possibly in the United Kingdom, where the sample size was too small to provide a reliable estimate.
Three-quarters of the clarithromycin-resistant isolates were also resistant to metronidazole.
The study also found that, overall, 1.2% of patients had isolates that were resistant to amoxicillin, and 69.2% had isolates resistant to metronidazole. Resistance patterns were similar in the United States and Europe; metronidazole resistance was the most common (50%-79% of isolates), and amoxicillin was the least common (≤ 5%).
“Overall, these data provide robust evidence to support a shift away from the default empiric prescription of triple combinations containing a PPI and clarithromycin for H. pylori infection in the United States and Europe,” the study team writes.
The high prevalence of resistance, including dual resistance, highlights the need for antibiotic stewardship and resistance surveillance, as well as novel treatment strategies for H. pylori infection, they add.
Last spring, as previously reported, the United States Food and Drug Administration approved two vonoprazan-based treatments for H. pylori: Voquezna Triple Pak (vonoprazan, amoxicillin, clarithromycin) and Voquezna Dual Pak (vonoprazan, amoxicillin), both from Phathom Pharmaceuticals.
“Vonoprazan-based treatment may be superior to standard PPI triple therapy for clarithromycin-resistant infections based on prior studies and is a potential good option,” Dr. Kim said.
Still, she added, she “would most likely first recommend regimens that do not have clarithromycin, such as bismuth quadruple therapy.”
Study’s importance
Because the study drew upon the largest dataset to date on U.S. resistance rates, it should be used to more precisely guide first-line therapy decisions, said Richard Peek, Jr., MD, professor of medicine and director of gastroenterology at Vanderbilt University Medical Center, Nashville, Tenn.
“To date, there has been a dearth of information in the United States regarding H. pylori resistance rates, which has often led to the use of ineffective empiric therapies and inappropriate exposure to antibiotics,” Dr. Peek, who wasn’t involved in the study, told this news organization.
“These data are particularly exciting when viewed within the context of new genomic sequencing tests that can determine H. pylori resistance patterns using DNA isolated from the stomach or the stool,” he said.
Dr. Peek agreed that the recent approval of vonoprazan-based therapies “adds another regimen to the therapeutic armamentarium available for eradicating H. pylori, and its value seems to be particularly beneficial for eradication failures.”
The research was funded by Phathom Pharmaceuticals. Dr. Chey is a board member of the American College of Gastroenterology, GI on Demand, the International Foundation of Functional GI Disorders, and the Rome Foundation. He has received compensation as a consultant from AbbVie, Alfasigma, Allakos, Alnylam, Bayer, BioAmerica, Cosmo, Intrinsic Medicine, Ironwood Pharmaceuticals, QOL Medical, Nestle, Phathom Pharmaceuticals, RedHill Biopharma, Salix/Valeant, Takeda, Urovant, and Vibrant; grant/research support from BioAmerica, Commonwealth Diagnostics International, QOL Medical, Salix, and Vibrant; owns stock/stock options in GI on Demand and Modify Health; and owns patents relating to methods and kits for identifying food sensitivities and intolerances, digital manometry, and a rectal expulsion device. Dr. Peek and Dr. Kim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Rates of resistance to clarithromycin among Helicobacter pylori isolates in the United States and Europe are high enough to warrant discontinuation of empiric use of proton pump inhibitor (PPI)–based triple therapy that includes the antibiotic in these regions, a new study has found.
Overall, 22.2% of participants were resistant to clarithromycin – a rate that is above the currently recommended threshold of 15% or higher for avoidance of PPI-based triple therapy that includes clarithromycin.
, study investigator William Chey, MD, professor and chief, Division of Gastroenterology and Hepatology, Michigan Medicine, Ann Arbor, said in an interview.
Judith Kim, MD, a gastroenterologist at NYU Langone Health and clinical instructor of medicine at NYU Grossman School of Medicine, who wasn’t involved in the study, agrees.
“The use of PPI-based triple therapy is still common practice despite recent recommendations to avoid clarithromycin in areas with high resistance rates,” Dr. Kim told this news organization.
“This study shows that multiple parts of the United States and Europe have high resistance rates,” rendering clarithromycin-based regimens “more likely to ineffectively eradicate H pylori,” Dr. Kim said.
The study was published online in The American Journal of Gastroenterology.
Better options now available
Guidelines advise against the use of PPI-based triple regimens with clarithromycin for H. pylori infection in areas where resistance is 15% or higher or for patients who have previously received macrolides. However, up-to-date information on H. pylori antimicrobial resistance patterns is limited, especially in the United States.
Dr. Chey and colleagues assessed resistance rates to antibiotics commonly used to treat H. pylori in isolates from 907 adults with the infection in the United States and Europe. They included four U.S. subregions and five participating European countries.
In all U.S. subregions and European countries, clarithromycin resistance rates were above 15% except possibly in the United Kingdom, where the sample size was too small to provide a reliable estimate.
Three-quarters of the clarithromycin-resistant isolates were also resistant to metronidazole.
The study also found that, overall, 1.2% of patients had isolates that were resistant to amoxicillin, and 69.2% had isolates resistant to metronidazole. Resistance patterns were similar in the United States and Europe; metronidazole resistance was the most common (50%-79% of isolates), and amoxicillin was the least common (≤ 5%).
“Overall, these data provide robust evidence to support a shift away from the default empiric prescription of triple combinations containing a PPI and clarithromycin for H. pylori infection in the United States and Europe,” the study team writes.
The high prevalence of resistance, including dual resistance, highlights the need for antibiotic stewardship and resistance surveillance, as well as novel treatment strategies for H. pylori infection, they add.
Last spring, as previously reported, the United States Food and Drug Administration approved two vonoprazan-based treatments for H. pylori: Voquezna Triple Pak (vonoprazan, amoxicillin, clarithromycin) and Voquezna Dual Pak (vonoprazan, amoxicillin), both from Phathom Pharmaceuticals.
“Vonoprazan-based treatment may be superior to standard PPI triple therapy for clarithromycin-resistant infections based on prior studies and is a potential good option,” Dr. Kim said.
Still, she added, she “would most likely first recommend regimens that do not have clarithromycin, such as bismuth quadruple therapy.”
Study’s importance
Because the study drew upon the largest dataset to date on U.S. resistance rates, it should be used to more precisely guide first-line therapy decisions, said Richard Peek, Jr., MD, professor of medicine and director of gastroenterology at Vanderbilt University Medical Center, Nashville, Tenn.
“To date, there has been a dearth of information in the United States regarding H. pylori resistance rates, which has often led to the use of ineffective empiric therapies and inappropriate exposure to antibiotics,” Dr. Peek, who wasn’t involved in the study, told this news organization.
“These data are particularly exciting when viewed within the context of new genomic sequencing tests that can determine H. pylori resistance patterns using DNA isolated from the stomach or the stool,” he said.
Dr. Peek agreed that the recent approval of vonoprazan-based therapies “adds another regimen to the therapeutic armamentarium available for eradicating H. pylori, and its value seems to be particularly beneficial for eradication failures.”
The research was funded by Phathom Pharmaceuticals. Dr. Chey is a board member of the American College of Gastroenterology, GI on Demand, the International Foundation of Functional GI Disorders, and the Rome Foundation. He has received compensation as a consultant from AbbVie, Alfasigma, Allakos, Alnylam, Bayer, BioAmerica, Cosmo, Intrinsic Medicine, Ironwood Pharmaceuticals, QOL Medical, Nestle, Phathom Pharmaceuticals, RedHill Biopharma, Salix/Valeant, Takeda, Urovant, and Vibrant; grant/research support from BioAmerica, Commonwealth Diagnostics International, QOL Medical, Salix, and Vibrant; owns stock/stock options in GI on Demand and Modify Health; and owns patents relating to methods and kits for identifying food sensitivities and intolerances, digital manometry, and a rectal expulsion device. Dr. Peek and Dr. Kim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Rates of resistance to clarithromycin among Helicobacter pylori isolates in the United States and Europe are high enough to warrant discontinuation of empiric use of proton pump inhibitor (PPI)–based triple therapy that includes the antibiotic in these regions, a new study has found.
Overall, 22.2% of participants were resistant to clarithromycin – a rate that is above the currently recommended threshold of 15% or higher for avoidance of PPI-based triple therapy that includes clarithromycin.
, study investigator William Chey, MD, professor and chief, Division of Gastroenterology and Hepatology, Michigan Medicine, Ann Arbor, said in an interview.
Judith Kim, MD, a gastroenterologist at NYU Langone Health and clinical instructor of medicine at NYU Grossman School of Medicine, who wasn’t involved in the study, agrees.
“The use of PPI-based triple therapy is still common practice despite recent recommendations to avoid clarithromycin in areas with high resistance rates,” Dr. Kim told this news organization.
“This study shows that multiple parts of the United States and Europe have high resistance rates,” rendering clarithromycin-based regimens “more likely to ineffectively eradicate H pylori,” Dr. Kim said.
The study was published online in The American Journal of Gastroenterology.
Better options now available
Guidelines advise against the use of PPI-based triple regimens with clarithromycin for H. pylori infection in areas where resistance is 15% or higher or for patients who have previously received macrolides. However, up-to-date information on H. pylori antimicrobial resistance patterns is limited, especially in the United States.
Dr. Chey and colleagues assessed resistance rates to antibiotics commonly used to treat H. pylori in isolates from 907 adults with the infection in the United States and Europe. They included four U.S. subregions and five participating European countries.
In all U.S. subregions and European countries, clarithromycin resistance rates were above 15% except possibly in the United Kingdom, where the sample size was too small to provide a reliable estimate.
Three-quarters of the clarithromycin-resistant isolates were also resistant to metronidazole.
The study also found that, overall, 1.2% of patients had isolates that were resistant to amoxicillin, and 69.2% had isolates resistant to metronidazole. Resistance patterns were similar in the United States and Europe; metronidazole resistance was the most common (50%-79% of isolates), and amoxicillin was the least common (≤ 5%).
“Overall, these data provide robust evidence to support a shift away from the default empiric prescription of triple combinations containing a PPI and clarithromycin for H. pylori infection in the United States and Europe,” the study team writes.
The high prevalence of resistance, including dual resistance, highlights the need for antibiotic stewardship and resistance surveillance, as well as novel treatment strategies for H. pylori infection, they add.
Last spring, as previously reported, the United States Food and Drug Administration approved two vonoprazan-based treatments for H. pylori: Voquezna Triple Pak (vonoprazan, amoxicillin, clarithromycin) and Voquezna Dual Pak (vonoprazan, amoxicillin), both from Phathom Pharmaceuticals.
“Vonoprazan-based treatment may be superior to standard PPI triple therapy for clarithromycin-resistant infections based on prior studies and is a potential good option,” Dr. Kim said.
Still, she added, she “would most likely first recommend regimens that do not have clarithromycin, such as bismuth quadruple therapy.”
Study’s importance
Because the study drew upon the largest dataset to date on U.S. resistance rates, it should be used to more precisely guide first-line therapy decisions, said Richard Peek, Jr., MD, professor of medicine and director of gastroenterology at Vanderbilt University Medical Center, Nashville, Tenn.
“To date, there has been a dearth of information in the United States regarding H. pylori resistance rates, which has often led to the use of ineffective empiric therapies and inappropriate exposure to antibiotics,” Dr. Peek, who wasn’t involved in the study, told this news organization.
“These data are particularly exciting when viewed within the context of new genomic sequencing tests that can determine H. pylori resistance patterns using DNA isolated from the stomach or the stool,” he said.
Dr. Peek agreed that the recent approval of vonoprazan-based therapies “adds another regimen to the therapeutic armamentarium available for eradicating H. pylori, and its value seems to be particularly beneficial for eradication failures.”
The research was funded by Phathom Pharmaceuticals. Dr. Chey is a board member of the American College of Gastroenterology, GI on Demand, the International Foundation of Functional GI Disorders, and the Rome Foundation. He has received compensation as a consultant from AbbVie, Alfasigma, Allakos, Alnylam, Bayer, BioAmerica, Cosmo, Intrinsic Medicine, Ironwood Pharmaceuticals, QOL Medical, Nestle, Phathom Pharmaceuticals, RedHill Biopharma, Salix/Valeant, Takeda, Urovant, and Vibrant; grant/research support from BioAmerica, Commonwealth Diagnostics International, QOL Medical, Salix, and Vibrant; owns stock/stock options in GI on Demand and Modify Health; and owns patents relating to methods and kits for identifying food sensitivities and intolerances, digital manometry, and a rectal expulsion device. Dr. Peek and Dr. Kim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Milk bad, cheese not? Dairy products tied to different CVD risks
The study, which analyzed a cohort from the Western Norway B-vitamin Intervention Trial (WENBIT), showed that higher dairy and milk consumption were associated with increased risk of mortality and stroke and butter was associated with an increased risk of acute myocardial infarction (AMI), but that cheese was associated with a decreased risk of AMI.
The findings are published in the European Journal of Preventive Cardiology.
“Dairy is a diverse food group, and different dairy products should be considered individually and not only in combination,” senior author Vegard Lysne, MSc, of the Centre for Nutrition, University of Bergen and the department of heart disease, Haukeland University Hospital, Bergen, Norway, said in an interview.
“Today’s dietary recommendations regarding dairy products are mainly based on the nutrient contents, with a focus on calcium, iodine, and saturated fat,” Dr. Lysne said.
Previous studies have indicated that different dairy products may influence cardiovascular health differently, even in opposite directions, but this has primarily been investigated in healthy populations, he noted.
“Data on CVD patients are scarce, and therefore, we wanted to investigate this in a population of patients with established CVD. Our primary aim in this study was to explore how the intake of different dairy products might be linked to cardiovascular outcomes and mortality in such a population,” he said.
The researchers analyzed 1,929 patients who had stable angina pectoris and were participants in WENBIT, a randomized, double-blind, placebo-controlled prospective secondary prevention study investigating the effect of vitamin B treatment on mortality and cardiovascular outcomes.
The majority, 80%, of the cohort were men, and the mean age of the patients was 61.8 years. In addition to stable angina pectoris, 47% of the cohort had hypertension, 31% had diabetes, and 29% were smokers. Most (90%) of the patients were taking acetylsalicylic acid, 90% were taking statins, and 77% were on beta-blockers.
Dietary data were obtained by a food frequency questionnaire that was given to patients at their first visit and returned either by mail or at a follow-up visit 1 month after the initial visit.
Frequency of consumption was given as times per day, week, month, or never consumed. Quantity was estimated using units such as slices, pieces, etc., or household measures.
The milk variable included high-fat, low-fat, skimmed, or unspecified milk. Cheese included brown cheese, which is a Norwegian caramel-like cheese made from whey, milk, and cream; white cheese; cream cheeses; cooked or processed cheeses; and boxed cheeses.
Total dairy was calculated as the sum, in grams, of milk, cheese, yogurt, cream, sour cream, ice cream, and butter.
Median follow-up times were 5.2 years for stroke, 7.8 years for AMI, and 14.1 years for mortality.
Patients who reported a higher intake of total dairy and milk had a higher risk of stroke and mortality.
Among those who reported a higher intake of total dairy, the hazard ratio for stroke was 1.4 (95% confidence interval [CI], 1.02-1.27).
Among those who reported a higher intake of milk, the HR for stroke was 1.13 (95% CI, 1.02-1.27).
Cardiovascular mortality appeared heightened in those who reported a higher intake of total dairy (HR, 1.06; 95% CI, 1.00-1.12) and in those who reported a higher intake of milk (HR, 1.07; 95% CI, 1.01-1.13).
Similarly, all-cause mortality was greater in those who reported higher total dairy consumption (HR, 1.07; 95% CI, 1.03-1.11) and in those who reported higher milk consumption (HR, 1.06; 95% CI, 1.03-1.10).
Higher cheese intake was inversely associated with AMI risk (HR, 0.92; 95% CI, 0.83-1.02).
Butter was associated with increased AMI risk (HR, 1.10; 95% CI, 0.97-1.24), as well as all-cause mortality (HR, 1.10; 95% CI, 1.00-1.20).
Dr. Lysne stressed that the results are from an observational study, and that doctors should not change what they tell their patients based on the results alone.
“There is a growing literature indicating that cheese might be linked to reduced cardiovascular risk, but if this is a causal effect, or if cheese is a marker of higher socioeconomic status and a healthier overall lifestyle remains unknown,” he said.
“I would like for future studies to evaluate dairy products on an individual basis, rather than a collective one. If the data suggest that different dairy products have distinct health effects, this should be implemented in dietary recommendations,” Dr. Lysne added.
Dairy a heterogeneous food group
“These results are not really surprising, because we have been hearing advice to consume low-fat milk, avoid whole milk, and so on, for a long time, so this study confirms what we already know,” Qi Sun, MD, ScD, associate professor in the departments of nutrition and epidemiology, Harvard T.H. Chan School of Public Health, Boston, told this news organization.
“However, I would be more specific about milk, and I don’t see any data regarding the fat content of the different types of milk. Their data only show the association for total milk. I would like to see data for low-fat milk versus high-fat milk in relation to heart disease,” Dr. Sun said.
“They also say in their conclusion that cheese was associated with a decreased risk of acute myocardial infarction, but as the hazard ratio shows, this is a nonsignificant association,” he said.
Dr. Sun agrees that dairy is a heterogeneous group of foods and that it is best to consider each type separately with regard to cardiovascular health.
“For example, heavy cream contains tons of saturated fat, butter contains a lot of saturated fat. Then there is yogurt, which also comes in regular, reduced-fat and low-fat varieties, which is a fantastic food. I would say it’s very healthy and is associated with a lower risk of heart disease and diabetes, so a good type of dairy. Yogurt and fermented dairy products should be beneficial, at least more so than full-fat milk or butter. I think butter and full-fat milk are still the primary dairy foods for people to avoid to reduce risk for cardiovascular disease,” he said.
Dr. Lysne and Dr. Sun have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The study, which analyzed a cohort from the Western Norway B-vitamin Intervention Trial (WENBIT), showed that higher dairy and milk consumption were associated with increased risk of mortality and stroke and butter was associated with an increased risk of acute myocardial infarction (AMI), but that cheese was associated with a decreased risk of AMI.
The findings are published in the European Journal of Preventive Cardiology.
“Dairy is a diverse food group, and different dairy products should be considered individually and not only in combination,” senior author Vegard Lysne, MSc, of the Centre for Nutrition, University of Bergen and the department of heart disease, Haukeland University Hospital, Bergen, Norway, said in an interview.
“Today’s dietary recommendations regarding dairy products are mainly based on the nutrient contents, with a focus on calcium, iodine, and saturated fat,” Dr. Lysne said.
Previous studies have indicated that different dairy products may influence cardiovascular health differently, even in opposite directions, but this has primarily been investigated in healthy populations, he noted.
“Data on CVD patients are scarce, and therefore, we wanted to investigate this in a population of patients with established CVD. Our primary aim in this study was to explore how the intake of different dairy products might be linked to cardiovascular outcomes and mortality in such a population,” he said.
The researchers analyzed 1,929 patients who had stable angina pectoris and were participants in WENBIT, a randomized, double-blind, placebo-controlled prospective secondary prevention study investigating the effect of vitamin B treatment on mortality and cardiovascular outcomes.
The majority, 80%, of the cohort were men, and the mean age of the patients was 61.8 years. In addition to stable angina pectoris, 47% of the cohort had hypertension, 31% had diabetes, and 29% were smokers. Most (90%) of the patients were taking acetylsalicylic acid, 90% were taking statins, and 77% were on beta-blockers.
Dietary data were obtained by a food frequency questionnaire that was given to patients at their first visit and returned either by mail or at a follow-up visit 1 month after the initial visit.
Frequency of consumption was given as times per day, week, month, or never consumed. Quantity was estimated using units such as slices, pieces, etc., or household measures.
The milk variable included high-fat, low-fat, skimmed, or unspecified milk. Cheese included brown cheese, which is a Norwegian caramel-like cheese made from whey, milk, and cream; white cheese; cream cheeses; cooked or processed cheeses; and boxed cheeses.
Total dairy was calculated as the sum, in grams, of milk, cheese, yogurt, cream, sour cream, ice cream, and butter.
Median follow-up times were 5.2 years for stroke, 7.8 years for AMI, and 14.1 years for mortality.
Patients who reported a higher intake of total dairy and milk had a higher risk of stroke and mortality.
Among those who reported a higher intake of total dairy, the hazard ratio for stroke was 1.4 (95% confidence interval [CI], 1.02-1.27).
Among those who reported a higher intake of milk, the HR for stroke was 1.13 (95% CI, 1.02-1.27).
Cardiovascular mortality appeared heightened in those who reported a higher intake of total dairy (HR, 1.06; 95% CI, 1.00-1.12) and in those who reported a higher intake of milk (HR, 1.07; 95% CI, 1.01-1.13).
Similarly, all-cause mortality was greater in those who reported higher total dairy consumption (HR, 1.07; 95% CI, 1.03-1.11) and in those who reported higher milk consumption (HR, 1.06; 95% CI, 1.03-1.10).
Higher cheese intake was inversely associated with AMI risk (HR, 0.92; 95% CI, 0.83-1.02).
Butter was associated with increased AMI risk (HR, 1.10; 95% CI, 0.97-1.24), as well as all-cause mortality (HR, 1.10; 95% CI, 1.00-1.20).
Dr. Lysne stressed that the results are from an observational study, and that doctors should not change what they tell their patients based on the results alone.
“There is a growing literature indicating that cheese might be linked to reduced cardiovascular risk, but if this is a causal effect, or if cheese is a marker of higher socioeconomic status and a healthier overall lifestyle remains unknown,” he said.
“I would like for future studies to evaluate dairy products on an individual basis, rather than a collective one. If the data suggest that different dairy products have distinct health effects, this should be implemented in dietary recommendations,” Dr. Lysne added.
Dairy a heterogeneous food group
“These results are not really surprising, because we have been hearing advice to consume low-fat milk, avoid whole milk, and so on, for a long time, so this study confirms what we already know,” Qi Sun, MD, ScD, associate professor in the departments of nutrition and epidemiology, Harvard T.H. Chan School of Public Health, Boston, told this news organization.
“However, I would be more specific about milk, and I don’t see any data regarding the fat content of the different types of milk. Their data only show the association for total milk. I would like to see data for low-fat milk versus high-fat milk in relation to heart disease,” Dr. Sun said.
“They also say in their conclusion that cheese was associated with a decreased risk of acute myocardial infarction, but as the hazard ratio shows, this is a nonsignificant association,” he said.
Dr. Sun agrees that dairy is a heterogeneous group of foods and that it is best to consider each type separately with regard to cardiovascular health.
“For example, heavy cream contains tons of saturated fat, butter contains a lot of saturated fat. Then there is yogurt, which also comes in regular, reduced-fat and low-fat varieties, which is a fantastic food. I would say it’s very healthy and is associated with a lower risk of heart disease and diabetes, so a good type of dairy. Yogurt and fermented dairy products should be beneficial, at least more so than full-fat milk or butter. I think butter and full-fat milk are still the primary dairy foods for people to avoid to reduce risk for cardiovascular disease,” he said.
Dr. Lysne and Dr. Sun have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The study, which analyzed a cohort from the Western Norway B-vitamin Intervention Trial (WENBIT), showed that higher dairy and milk consumption were associated with increased risk of mortality and stroke and butter was associated with an increased risk of acute myocardial infarction (AMI), but that cheese was associated with a decreased risk of AMI.
The findings are published in the European Journal of Preventive Cardiology.
“Dairy is a diverse food group, and different dairy products should be considered individually and not only in combination,” senior author Vegard Lysne, MSc, of the Centre for Nutrition, University of Bergen and the department of heart disease, Haukeland University Hospital, Bergen, Norway, said in an interview.
“Today’s dietary recommendations regarding dairy products are mainly based on the nutrient contents, with a focus on calcium, iodine, and saturated fat,” Dr. Lysne said.
Previous studies have indicated that different dairy products may influence cardiovascular health differently, even in opposite directions, but this has primarily been investigated in healthy populations, he noted.
“Data on CVD patients are scarce, and therefore, we wanted to investigate this in a population of patients with established CVD. Our primary aim in this study was to explore how the intake of different dairy products might be linked to cardiovascular outcomes and mortality in such a population,” he said.
The researchers analyzed 1,929 patients who had stable angina pectoris and were participants in WENBIT, a randomized, double-blind, placebo-controlled prospective secondary prevention study investigating the effect of vitamin B treatment on mortality and cardiovascular outcomes.
The majority, 80%, of the cohort were men, and the mean age of the patients was 61.8 years. In addition to stable angina pectoris, 47% of the cohort had hypertension, 31% had diabetes, and 29% were smokers. Most (90%) of the patients were taking acetylsalicylic acid, 90% were taking statins, and 77% were on beta-blockers.
Dietary data were obtained by a food frequency questionnaire that was given to patients at their first visit and returned either by mail or at a follow-up visit 1 month after the initial visit.
Frequency of consumption was given as times per day, week, month, or never consumed. Quantity was estimated using units such as slices, pieces, etc., or household measures.
The milk variable included high-fat, low-fat, skimmed, or unspecified milk. Cheese included brown cheese, which is a Norwegian caramel-like cheese made from whey, milk, and cream; white cheese; cream cheeses; cooked or processed cheeses; and boxed cheeses.
Total dairy was calculated as the sum, in grams, of milk, cheese, yogurt, cream, sour cream, ice cream, and butter.
Median follow-up times were 5.2 years for stroke, 7.8 years for AMI, and 14.1 years for mortality.
Patients who reported a higher intake of total dairy and milk had a higher risk of stroke and mortality.
Among those who reported a higher intake of total dairy, the hazard ratio for stroke was 1.4 (95% confidence interval [CI], 1.02-1.27).
Among those who reported a higher intake of milk, the HR for stroke was 1.13 (95% CI, 1.02-1.27).
Cardiovascular mortality appeared heightened in those who reported a higher intake of total dairy (HR, 1.06; 95% CI, 1.00-1.12) and in those who reported a higher intake of milk (HR, 1.07; 95% CI, 1.01-1.13).
Similarly, all-cause mortality was greater in those who reported higher total dairy consumption (HR, 1.07; 95% CI, 1.03-1.11) and in those who reported higher milk consumption (HR, 1.06; 95% CI, 1.03-1.10).
Higher cheese intake was inversely associated with AMI risk (HR, 0.92; 95% CI, 0.83-1.02).
Butter was associated with increased AMI risk (HR, 1.10; 95% CI, 0.97-1.24), as well as all-cause mortality (HR, 1.10; 95% CI, 1.00-1.20).
Dr. Lysne stressed that the results are from an observational study, and that doctors should not change what they tell their patients based on the results alone.
“There is a growing literature indicating that cheese might be linked to reduced cardiovascular risk, but if this is a causal effect, or if cheese is a marker of higher socioeconomic status and a healthier overall lifestyle remains unknown,” he said.
“I would like for future studies to evaluate dairy products on an individual basis, rather than a collective one. If the data suggest that different dairy products have distinct health effects, this should be implemented in dietary recommendations,” Dr. Lysne added.
Dairy a heterogeneous food group
“These results are not really surprising, because we have been hearing advice to consume low-fat milk, avoid whole milk, and so on, for a long time, so this study confirms what we already know,” Qi Sun, MD, ScD, associate professor in the departments of nutrition and epidemiology, Harvard T.H. Chan School of Public Health, Boston, told this news organization.
“However, I would be more specific about milk, and I don’t see any data regarding the fat content of the different types of milk. Their data only show the association for total milk. I would like to see data for low-fat milk versus high-fat milk in relation to heart disease,” Dr. Sun said.
“They also say in their conclusion that cheese was associated with a decreased risk of acute myocardial infarction, but as the hazard ratio shows, this is a nonsignificant association,” he said.
Dr. Sun agrees that dairy is a heterogeneous group of foods and that it is best to consider each type separately with regard to cardiovascular health.
“For example, heavy cream contains tons of saturated fat, butter contains a lot of saturated fat. Then there is yogurt, which also comes in regular, reduced-fat and low-fat varieties, which is a fantastic food. I would say it’s very healthy and is associated with a lower risk of heart disease and diabetes, so a good type of dairy. Yogurt and fermented dairy products should be beneficial, at least more so than full-fat milk or butter. I think butter and full-fat milk are still the primary dairy foods for people to avoid to reduce risk for cardiovascular disease,” he said.
Dr. Lysne and Dr. Sun have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM EUROPEAN JOURNAL OF PREVENTIVE CARDIOLOGY
Pancreatic cancer screening appears safe, effective for high-risk patients
Pancreatic cancer screening appears to be safe and effective for certain patients with high-risk indications due to genetic susceptibility, according to a prospective multicenter study presented at the annual meeting of the American College of Gastroenterology.
Screening in high-risk patients detected high-risk lesions in 0.8% of patients, which was lower than the typical range found in the literature, at 3%, said Andy Silva-Santisteban, MD, a research fellow at Beth Israel Deaconess Medical Center at Harvard Medical School in Boston.
Pancreatic cancer is the third leading cause of cancer death in the U.S., which is estimated to become the second leading cause by 2030. About 15%-20% of patients are candidates for surgical resection at the time of diagnosis, with survival rates below 10%.
“These statistics have led pancreatic cancer screening to be studied with the goal of detecting earlier stages of the disease to improve survival,” Dr. Silva-Santisteban said. “However, pancreatic cancer screening is not recommended for the general population.”
Pancreatic cancer screening is recommended for patients with increased risk due to genetic susceptibility, yet recent studies have found that screening studies face limitations from factors like small sample sizes, single-center focus, retrospective nature, nonconsecutive accrual of patients, varied inclusion criteria, and use of nonstandardized screening protocols.
To overcome these limitations, Dr. Silva-Santisteban and colleagues conducted a prospective multicenter study of pancreatic cancer screening in consecutive high-risk patients at five centers in the United States between 2020 and 2022, also called the Pancreas Scan Study. Dr. Silva-Santisteban presented results from the first round of enrollment, which was awarded the Outstanding Research Award in the Biliary/Pancreas Category for Trainee.
The research team evaluated the yield (low-, moderate-, and high-risk pancreatic pathology), safety, and outcomes of screening. Low-risk pancreas pathology was categorized as fatty pancreas and chronic pancreatitis-like changes. Intermediate-risk was categorized as branch duct–intraductal papillary mucinous neoplasm or neuroendocrine tumor under 2 cm. High-risk was categorized as main duct–intraductal papillary mucinous neoplasm (MD-IPMN), pancreatic intraepithelial neoplasia grade III (PanIN-III)/dysplasia, neuroendocrine tumor over 2 cm, or pancreatic cancer.
Patients were included if they were 18 years or older and had at least one of the following: BRCA1, BRCA2, or PALB2 plus a family history of pancreatic cancer; Lynch syndrome plus a family history of pancreatic cancer; Peutz-Jeghers syndrome; familial atypical multiple mole melanoma (FAMMM); ataxia telangiectasia mutated plus family history of pancreatic cancer; hereditary pancreatitis; or familial pancreatic cancer (FPC) syndrome.
Screening was performed annually with either endoscopic ultrasound (EUS) or magnetic resonance cholangiopancreatography (MRCP). Fasting blood sugar was recorded annually to screen for new-onset diabetes.
Among 252 patients, 208 underwent EUS and 44 underwent MRCP. At the time of enrollment, 38.5% underwent their first screening, and 61.5% had a prior screening. The average age was 60, 69% were women, and 79% were White.
The most common indication was a BRCA1 or BRCA2 pathogenic variant in 93 patients (or 36.5%), followed by FPC syndrome in 80 patients (or 31.7%).
Low-risk pancreas pathology was noted in 23.4% of patients, with 17.5% having chronic pancreatitis-like changes. Intermediate risk was found in 31.7%, with nearly all detected as branch-duct IPMNs without worrisome features, Dr. Silva-Santisteban said.
Two patients (.8%) fell into the high-risk category with pancreatic adenocarcinoma. Both were positive for BRCA2 mutation and family history of pancreatic cancer.
In the first patient, who was compliant with screening, EUS showed a 3-cm adenocarcinoma (T2N1M0 stage IIB). The patient underwent neoadjuvant chemotherapy, followed by total pancreatectomy, and is currently in cancer remission. No complications from surgery were noted.
In the second patient, who was not compliant with screening and was lost to follow-up for 6 years, EUS showed a 2.5-cm adenocarcinoma and four metastatic lesions in the liver (T2N1M1 stage IV). The patient underwent palliative chemotherapy.
EUS was more likely to identify chronic pancreatitis-like changes, but MRCP was more likely to identify BD-IPMN. The two patients with pancreatic adenocarcinoma were identified with EUS. However, there wasn’t a significant difference between EUS and MRCP in identifying high-risk lesions.
In patients undergoing screening, new-onset prediabetes was noted in 18.2%, and new-onset diabetes was noted in 1.7%. However, there was no association between abnormal blood sugar and pancreas pathology.
Twelve patients (4.8%) underwent further pancreatic evaluation because of screening findings. None of the patients underwent low-yield pancreatic surgery, which was lower than reported in the literature, at 2.8%. Overall, there were no complications as a direct result of screening with EUS or MRI.
“Patients should be carefully counseled regarding benefits and harms from pancreatic cancer screening,” Dr. Silva-Santisteban said. “When feasible, such screening should be performed within the confines of a research study so more precise estimates of screening outcomes can be determined.”
The study funding was not disclosed. One author reported a consultant relationship with Pentax Medical, and the other authors indicated no relevant financial relationships.
Pancreatic cancer screening appears to be safe and effective for certain patients with high-risk indications due to genetic susceptibility, according to a prospective multicenter study presented at the annual meeting of the American College of Gastroenterology.
Screening in high-risk patients detected high-risk lesions in 0.8% of patients, which was lower than the typical range found in the literature, at 3%, said Andy Silva-Santisteban, MD, a research fellow at Beth Israel Deaconess Medical Center at Harvard Medical School in Boston.
Pancreatic cancer is the third leading cause of cancer death in the U.S., which is estimated to become the second leading cause by 2030. About 15%-20% of patients are candidates for surgical resection at the time of diagnosis, with survival rates below 10%.
“These statistics have led pancreatic cancer screening to be studied with the goal of detecting earlier stages of the disease to improve survival,” Dr. Silva-Santisteban said. “However, pancreatic cancer screening is not recommended for the general population.”
Pancreatic cancer screening is recommended for patients with increased risk due to genetic susceptibility, yet recent studies have found that screening studies face limitations from factors like small sample sizes, single-center focus, retrospective nature, nonconsecutive accrual of patients, varied inclusion criteria, and use of nonstandardized screening protocols.
To overcome these limitations, Dr. Silva-Santisteban and colleagues conducted a prospective multicenter study of pancreatic cancer screening in consecutive high-risk patients at five centers in the United States between 2020 and 2022, also called the Pancreas Scan Study. Dr. Silva-Santisteban presented results from the first round of enrollment, which was awarded the Outstanding Research Award in the Biliary/Pancreas Category for Trainee.
The research team evaluated the yield (low-, moderate-, and high-risk pancreatic pathology), safety, and outcomes of screening. Low-risk pancreas pathology was categorized as fatty pancreas and chronic pancreatitis-like changes. Intermediate-risk was categorized as branch duct–intraductal papillary mucinous neoplasm or neuroendocrine tumor under 2 cm. High-risk was categorized as main duct–intraductal papillary mucinous neoplasm (MD-IPMN), pancreatic intraepithelial neoplasia grade III (PanIN-III)/dysplasia, neuroendocrine tumor over 2 cm, or pancreatic cancer.
Patients were included if they were 18 years or older and had at least one of the following: BRCA1, BRCA2, or PALB2 plus a family history of pancreatic cancer; Lynch syndrome plus a family history of pancreatic cancer; Peutz-Jeghers syndrome; familial atypical multiple mole melanoma (FAMMM); ataxia telangiectasia mutated plus family history of pancreatic cancer; hereditary pancreatitis; or familial pancreatic cancer (FPC) syndrome.
Screening was performed annually with either endoscopic ultrasound (EUS) or magnetic resonance cholangiopancreatography (MRCP). Fasting blood sugar was recorded annually to screen for new-onset diabetes.
Among 252 patients, 208 underwent EUS and 44 underwent MRCP. At the time of enrollment, 38.5% underwent their first screening, and 61.5% had a prior screening. The average age was 60, 69% were women, and 79% were White.
The most common indication was a BRCA1 or BRCA2 pathogenic variant in 93 patients (or 36.5%), followed by FPC syndrome in 80 patients (or 31.7%).
Low-risk pancreas pathology was noted in 23.4% of patients, with 17.5% having chronic pancreatitis-like changes. Intermediate risk was found in 31.7%, with nearly all detected as branch-duct IPMNs without worrisome features, Dr. Silva-Santisteban said.
Two patients (.8%) fell into the high-risk category with pancreatic adenocarcinoma. Both were positive for BRCA2 mutation and family history of pancreatic cancer.
In the first patient, who was compliant with screening, EUS showed a 3-cm adenocarcinoma (T2N1M0 stage IIB). The patient underwent neoadjuvant chemotherapy, followed by total pancreatectomy, and is currently in cancer remission. No complications from surgery were noted.
In the second patient, who was not compliant with screening and was lost to follow-up for 6 years, EUS showed a 2.5-cm adenocarcinoma and four metastatic lesions in the liver (T2N1M1 stage IV). The patient underwent palliative chemotherapy.
EUS was more likely to identify chronic pancreatitis-like changes, but MRCP was more likely to identify BD-IPMN. The two patients with pancreatic adenocarcinoma were identified with EUS. However, there wasn’t a significant difference between EUS and MRCP in identifying high-risk lesions.
In patients undergoing screening, new-onset prediabetes was noted in 18.2%, and new-onset diabetes was noted in 1.7%. However, there was no association between abnormal blood sugar and pancreas pathology.
Twelve patients (4.8%) underwent further pancreatic evaluation because of screening findings. None of the patients underwent low-yield pancreatic surgery, which was lower than reported in the literature, at 2.8%. Overall, there were no complications as a direct result of screening with EUS or MRI.
“Patients should be carefully counseled regarding benefits and harms from pancreatic cancer screening,” Dr. Silva-Santisteban said. “When feasible, such screening should be performed within the confines of a research study so more precise estimates of screening outcomes can be determined.”
The study funding was not disclosed. One author reported a consultant relationship with Pentax Medical, and the other authors indicated no relevant financial relationships.
Pancreatic cancer screening appears to be safe and effective for certain patients with high-risk indications due to genetic susceptibility, according to a prospective multicenter study presented at the annual meeting of the American College of Gastroenterology.
Screening in high-risk patients detected high-risk lesions in 0.8% of patients, which was lower than the typical range found in the literature, at 3%, said Andy Silva-Santisteban, MD, a research fellow at Beth Israel Deaconess Medical Center at Harvard Medical School in Boston.
Pancreatic cancer is the third leading cause of cancer death in the U.S., which is estimated to become the second leading cause by 2030. About 15%-20% of patients are candidates for surgical resection at the time of diagnosis, with survival rates below 10%.
“These statistics have led pancreatic cancer screening to be studied with the goal of detecting earlier stages of the disease to improve survival,” Dr. Silva-Santisteban said. “However, pancreatic cancer screening is not recommended for the general population.”
Pancreatic cancer screening is recommended for patients with increased risk due to genetic susceptibility, yet recent studies have found that screening studies face limitations from factors like small sample sizes, single-center focus, retrospective nature, nonconsecutive accrual of patients, varied inclusion criteria, and use of nonstandardized screening protocols.
To overcome these limitations, Dr. Silva-Santisteban and colleagues conducted a prospective multicenter study of pancreatic cancer screening in consecutive high-risk patients at five centers in the United States between 2020 and 2022, also called the Pancreas Scan Study. Dr. Silva-Santisteban presented results from the first round of enrollment, which was awarded the Outstanding Research Award in the Biliary/Pancreas Category for Trainee.
The research team evaluated the yield (low-, moderate-, and high-risk pancreatic pathology), safety, and outcomes of screening. Low-risk pancreas pathology was categorized as fatty pancreas and chronic pancreatitis-like changes. Intermediate-risk was categorized as branch duct–intraductal papillary mucinous neoplasm or neuroendocrine tumor under 2 cm. High-risk was categorized as main duct–intraductal papillary mucinous neoplasm (MD-IPMN), pancreatic intraepithelial neoplasia grade III (PanIN-III)/dysplasia, neuroendocrine tumor over 2 cm, or pancreatic cancer.
Patients were included if they were 18 years or older and had at least one of the following: BRCA1, BRCA2, or PALB2 plus a family history of pancreatic cancer; Lynch syndrome plus a family history of pancreatic cancer; Peutz-Jeghers syndrome; familial atypical multiple mole melanoma (FAMMM); ataxia telangiectasia mutated plus family history of pancreatic cancer; hereditary pancreatitis; or familial pancreatic cancer (FPC) syndrome.
Screening was performed annually with either endoscopic ultrasound (EUS) or magnetic resonance cholangiopancreatography (MRCP). Fasting blood sugar was recorded annually to screen for new-onset diabetes.
Among 252 patients, 208 underwent EUS and 44 underwent MRCP. At the time of enrollment, 38.5% underwent their first screening, and 61.5% had a prior screening. The average age was 60, 69% were women, and 79% were White.
The most common indication was a BRCA1 or BRCA2 pathogenic variant in 93 patients (or 36.5%), followed by FPC syndrome in 80 patients (or 31.7%).
Low-risk pancreas pathology was noted in 23.4% of patients, with 17.5% having chronic pancreatitis-like changes. Intermediate risk was found in 31.7%, with nearly all detected as branch-duct IPMNs without worrisome features, Dr. Silva-Santisteban said.
Two patients (.8%) fell into the high-risk category with pancreatic adenocarcinoma. Both were positive for BRCA2 mutation and family history of pancreatic cancer.
In the first patient, who was compliant with screening, EUS showed a 3-cm adenocarcinoma (T2N1M0 stage IIB). The patient underwent neoadjuvant chemotherapy, followed by total pancreatectomy, and is currently in cancer remission. No complications from surgery were noted.
In the second patient, who was not compliant with screening and was lost to follow-up for 6 years, EUS showed a 2.5-cm adenocarcinoma and four metastatic lesions in the liver (T2N1M1 stage IV). The patient underwent palliative chemotherapy.
EUS was more likely to identify chronic pancreatitis-like changes, but MRCP was more likely to identify BD-IPMN. The two patients with pancreatic adenocarcinoma were identified with EUS. However, there wasn’t a significant difference between EUS and MRCP in identifying high-risk lesions.
In patients undergoing screening, new-onset prediabetes was noted in 18.2%, and new-onset diabetes was noted in 1.7%. However, there was no association between abnormal blood sugar and pancreas pathology.
Twelve patients (4.8%) underwent further pancreatic evaluation because of screening findings. None of the patients underwent low-yield pancreatic surgery, which was lower than reported in the literature, at 2.8%. Overall, there were no complications as a direct result of screening with EUS or MRI.
“Patients should be carefully counseled regarding benefits and harms from pancreatic cancer screening,” Dr. Silva-Santisteban said. “When feasible, such screening should be performed within the confines of a research study so more precise estimates of screening outcomes can be determined.”
The study funding was not disclosed. One author reported a consultant relationship with Pentax Medical, and the other authors indicated no relevant financial relationships.
FROM ACG 2022
Stopping levothyroxine in subclinical hypothyroidism safe, feasible
MONTREAL – Patients who discontinue levothyroxine for subclinical hypothyroidism may gravitate towards becoming mildly hypothyroid again, but they importantly show no differences in terms of symptoms and quality of life – and sometimes show even improvement – compared with those who continue treatment, new research shows.
“Our results show feasibility of patient enrollment and safety of discontinuing levothyroxine in patients with subclinical hypothyroidism,” said first author Spyridoula Maraka, MD, when presenting the findings at the American Thyroid Association annual meeting.
With evidence showing widespread overtreatment with levothyroxine for a variety of reasons, “a discontinuation study like this is important to understand the true need for life-long thyroxine therapy,” commented James V. Hennessey, MD, director of clinical endocrinology at Beth Israel Deaconess Medical Center, Boston.
Recommendations against levothyroxine for subclinical hypothyroidism
Subclinical hypothyroidism is commonly over-diagnosed, and treatment with thyroid hormone replacement, levothyroxine, has been shown to provide little, if any, benefit in terms of quality of life or relief of thyroid-related symptoms for these patients.
The treatment is meanwhile associated with burdens including cost and lifestyle adjustments, and one guideline panel recently issued a strong recommendation against routine levothyroxine use in most adults with subclinical hypothyroidism.
Nevertheless, levothyroxine treatment has soared in popularity and become one of the most commonly prescribed drugs in the United States.
With research lacking on one key solution of discontinuation of the therapy, Dr. Maraka, who is part of the Division of Endocrinology and Metabolism at the University of Arkansas for Medical Sciences, Little Rock, and colleagues conducted a double-blind, placebo-controlled trial at the Central Arkansas Veterans Healthcare System. In total, 50 patients treated for subclinical hypothyroidism were randomized 1:1 to continue receiving levothyroxine (25-75 mcg daily) or to discontinue treatment and receive a placebo instead, with a planned 6-month follow-up.
In the current interim analysis, Dr. Maraka reported results for the first 40 patients, including 20 randomized to levothyroxine and 20 to discontinuation.
There were no significant differences between the discontinuation and levothyroxine groups at baseline, which were of a similar age (66.2 vs. 70.8 years) and gender (75% women vs. 85% men).
The groups had similar baseline thyroid-stimulating hormone (TSH) levels (3.0 vs. 2.6 mIU/L), free T4 (both 0.9 ng/dL), thyroid peroxidase antibody positivity (17% vs. 11%), and similar clinical symptoms. All patients had at least one elevated TSH reading prior to starting levothyroxine.
With a follow-up of 6-8 weeks, 36.8% of patients in the discontinuation group had subclinical hypothyroidism, compared with 10% of patients who remained on levothyroxine (P = .0648), TSH levels were 5.5 versus 2.7 mIU/L (P = .001) and free T4 levels were 0.8 versus 0.9 ng/dL (P = .011).
No differences in symptoms, quality of life between groups
Importantly, there were no significant differences between the discontinuation versus levothyroxine groups in terms of symptoms, and even some improvements with discontinuation, including Thyroid-Specific Quality of Life Patient-Reported Outcome (ThyPRO)-Hypothyroid Symptoms score (4.6 reduction vs. 2.2 increase), tiredness (2.6 reduction vs. 1.1 increase), and EuroQoL 5-Dimension Self-Report Questionnaire (EQ-5D) quality of life score, for which there were no differences between groups.
There were no reports of overt hypothyroidism; hyperthyroidism; cardiovascular events including atrial fibrillation, stroke, or heart failure; osteoporotic fractures; or deaths.
One patient in the discontinuation group had a TSH level of 11 mIU/L at 6-8 weeks and switched to open-label levothyroxine 75 mcg daily. Another patient in the discontinuation group switched to open-label levothyroxine 75 mcg daily at 10 weeks due to fatigue; however, the patient was diagnosed with metastatic colon cancer 1 month later.
The finding that only about a third of patients who discontinued levothyroxine developed subclinical hypothyroidism was lower than expected, Dr. Maraka noted.
“This was ... unexpected ... for us,” she said. “We were expecting a larger number of patients to develop hypothyroidism, but to our surprise, that was not the case.”
“But what is more important is that there was no difference in the quality of life measures,” she added. “If anything, the placebo group was a little better, though the [differences] were not statistically significant.”
Dr. Maraka also noted that in further research and a final 6-month analysis, the authors will look at factors associated with developing subclinical hypothyroidism after treatment discontinuation, among other issues.
Discontinuation of levothyroxine is manageable
The results are encouraging, as they provide assurance that discontinuation of levothyroxine is manageable.
“This research will pave the way for initiatives to promote levothyroxine deprescription and implementation of evidence-based care for patients with subclinical hypothyroidism,” she said.
In further comments, Dr. Hennessey noted that the dilemma of having patients on levothyroxine who may not be benefitting from treatment is “significant,” with patients sometimes reluctant to discontinue treatment due to concerns of developing hypothyroidism-associated symptoms such as brain fog and weight gain.
He noted, however, that “many with mildly elevated TSH actually go on to normalize with time, so they are not really hypothyroid, [and] if we remove thyroxine from people with normal thyroid function, they will remain normal.”
Dr. Maraka has reported no relevant financial relationships. Dr. Hennessey has reported consulting for pharmaceutical companies to design clinical studies for thyroid medications.
A version of this article first appeared on Medscape.com.
MONTREAL – Patients who discontinue levothyroxine for subclinical hypothyroidism may gravitate towards becoming mildly hypothyroid again, but they importantly show no differences in terms of symptoms and quality of life – and sometimes show even improvement – compared with those who continue treatment, new research shows.
“Our results show feasibility of patient enrollment and safety of discontinuing levothyroxine in patients with subclinical hypothyroidism,” said first author Spyridoula Maraka, MD, when presenting the findings at the American Thyroid Association annual meeting.
With evidence showing widespread overtreatment with levothyroxine for a variety of reasons, “a discontinuation study like this is important to understand the true need for life-long thyroxine therapy,” commented James V. Hennessey, MD, director of clinical endocrinology at Beth Israel Deaconess Medical Center, Boston.
Recommendations against levothyroxine for subclinical hypothyroidism
Subclinical hypothyroidism is commonly over-diagnosed, and treatment with thyroid hormone replacement, levothyroxine, has been shown to provide little, if any, benefit in terms of quality of life or relief of thyroid-related symptoms for these patients.
The treatment is meanwhile associated with burdens including cost and lifestyle adjustments, and one guideline panel recently issued a strong recommendation against routine levothyroxine use in most adults with subclinical hypothyroidism.
Nevertheless, levothyroxine treatment has soared in popularity and become one of the most commonly prescribed drugs in the United States.
With research lacking on one key solution of discontinuation of the therapy, Dr. Maraka, who is part of the Division of Endocrinology and Metabolism at the University of Arkansas for Medical Sciences, Little Rock, and colleagues conducted a double-blind, placebo-controlled trial at the Central Arkansas Veterans Healthcare System. In total, 50 patients treated for subclinical hypothyroidism were randomized 1:1 to continue receiving levothyroxine (25-75 mcg daily) or to discontinue treatment and receive a placebo instead, with a planned 6-month follow-up.
In the current interim analysis, Dr. Maraka reported results for the first 40 patients, including 20 randomized to levothyroxine and 20 to discontinuation.
There were no significant differences between the discontinuation and levothyroxine groups at baseline, which were of a similar age (66.2 vs. 70.8 years) and gender (75% women vs. 85% men).
The groups had similar baseline thyroid-stimulating hormone (TSH) levels (3.0 vs. 2.6 mIU/L), free T4 (both 0.9 ng/dL), thyroid peroxidase antibody positivity (17% vs. 11%), and similar clinical symptoms. All patients had at least one elevated TSH reading prior to starting levothyroxine.
With a follow-up of 6-8 weeks, 36.8% of patients in the discontinuation group had subclinical hypothyroidism, compared with 10% of patients who remained on levothyroxine (P = .0648), TSH levels were 5.5 versus 2.7 mIU/L (P = .001) and free T4 levels were 0.8 versus 0.9 ng/dL (P = .011).
No differences in symptoms, quality of life between groups
Importantly, there were no significant differences between the discontinuation versus levothyroxine groups in terms of symptoms, and even some improvements with discontinuation, including Thyroid-Specific Quality of Life Patient-Reported Outcome (ThyPRO)-Hypothyroid Symptoms score (4.6 reduction vs. 2.2 increase), tiredness (2.6 reduction vs. 1.1 increase), and EuroQoL 5-Dimension Self-Report Questionnaire (EQ-5D) quality of life score, for which there were no differences between groups.
There were no reports of overt hypothyroidism; hyperthyroidism; cardiovascular events including atrial fibrillation, stroke, or heart failure; osteoporotic fractures; or deaths.
One patient in the discontinuation group had a TSH level of 11 mIU/L at 6-8 weeks and switched to open-label levothyroxine 75 mcg daily. Another patient in the discontinuation group switched to open-label levothyroxine 75 mcg daily at 10 weeks due to fatigue; however, the patient was diagnosed with metastatic colon cancer 1 month later.
The finding that only about a third of patients who discontinued levothyroxine developed subclinical hypothyroidism was lower than expected, Dr. Maraka noted.
“This was ... unexpected ... for us,” she said. “We were expecting a larger number of patients to develop hypothyroidism, but to our surprise, that was not the case.”
“But what is more important is that there was no difference in the quality of life measures,” she added. “If anything, the placebo group was a little better, though the [differences] were not statistically significant.”
Dr. Maraka also noted that in further research and a final 6-month analysis, the authors will look at factors associated with developing subclinical hypothyroidism after treatment discontinuation, among other issues.
Discontinuation of levothyroxine is manageable
The results are encouraging, as they provide assurance that discontinuation of levothyroxine is manageable.
“This research will pave the way for initiatives to promote levothyroxine deprescription and implementation of evidence-based care for patients with subclinical hypothyroidism,” she said.
In further comments, Dr. Hennessey noted that the dilemma of having patients on levothyroxine who may not be benefitting from treatment is “significant,” with patients sometimes reluctant to discontinue treatment due to concerns of developing hypothyroidism-associated symptoms such as brain fog and weight gain.
He noted, however, that “many with mildly elevated TSH actually go on to normalize with time, so they are not really hypothyroid, [and] if we remove thyroxine from people with normal thyroid function, they will remain normal.”
Dr. Maraka has reported no relevant financial relationships. Dr. Hennessey has reported consulting for pharmaceutical companies to design clinical studies for thyroid medications.
A version of this article first appeared on Medscape.com.
MONTREAL – Patients who discontinue levothyroxine for subclinical hypothyroidism may gravitate towards becoming mildly hypothyroid again, but they importantly show no differences in terms of symptoms and quality of life – and sometimes show even improvement – compared with those who continue treatment, new research shows.
“Our results show feasibility of patient enrollment and safety of discontinuing levothyroxine in patients with subclinical hypothyroidism,” said first author Spyridoula Maraka, MD, when presenting the findings at the American Thyroid Association annual meeting.
With evidence showing widespread overtreatment with levothyroxine for a variety of reasons, “a discontinuation study like this is important to understand the true need for life-long thyroxine therapy,” commented James V. Hennessey, MD, director of clinical endocrinology at Beth Israel Deaconess Medical Center, Boston.
Recommendations against levothyroxine for subclinical hypothyroidism
Subclinical hypothyroidism is commonly over-diagnosed, and treatment with thyroid hormone replacement, levothyroxine, has been shown to provide little, if any, benefit in terms of quality of life or relief of thyroid-related symptoms for these patients.
The treatment is meanwhile associated with burdens including cost and lifestyle adjustments, and one guideline panel recently issued a strong recommendation against routine levothyroxine use in most adults with subclinical hypothyroidism.
Nevertheless, levothyroxine treatment has soared in popularity and become one of the most commonly prescribed drugs in the United States.
With research lacking on one key solution of discontinuation of the therapy, Dr. Maraka, who is part of the Division of Endocrinology and Metabolism at the University of Arkansas for Medical Sciences, Little Rock, and colleagues conducted a double-blind, placebo-controlled trial at the Central Arkansas Veterans Healthcare System. In total, 50 patients treated for subclinical hypothyroidism were randomized 1:1 to continue receiving levothyroxine (25-75 mcg daily) or to discontinue treatment and receive a placebo instead, with a planned 6-month follow-up.
In the current interim analysis, Dr. Maraka reported results for the first 40 patients, including 20 randomized to levothyroxine and 20 to discontinuation.
There were no significant differences between the discontinuation and levothyroxine groups at baseline, which were of a similar age (66.2 vs. 70.8 years) and gender (75% women vs. 85% men).
The groups had similar baseline thyroid-stimulating hormone (TSH) levels (3.0 vs. 2.6 mIU/L), free T4 (both 0.9 ng/dL), thyroid peroxidase antibody positivity (17% vs. 11%), and similar clinical symptoms. All patients had at least one elevated TSH reading prior to starting levothyroxine.
With a follow-up of 6-8 weeks, 36.8% of patients in the discontinuation group had subclinical hypothyroidism, compared with 10% of patients who remained on levothyroxine (P = .0648), TSH levels were 5.5 versus 2.7 mIU/L (P = .001) and free T4 levels were 0.8 versus 0.9 ng/dL (P = .011).
No differences in symptoms, quality of life between groups
Importantly, there were no significant differences between the discontinuation versus levothyroxine groups in terms of symptoms, and even some improvements with discontinuation, including Thyroid-Specific Quality of Life Patient-Reported Outcome (ThyPRO)-Hypothyroid Symptoms score (4.6 reduction vs. 2.2 increase), tiredness (2.6 reduction vs. 1.1 increase), and EuroQoL 5-Dimension Self-Report Questionnaire (EQ-5D) quality of life score, for which there were no differences between groups.
There were no reports of overt hypothyroidism; hyperthyroidism; cardiovascular events including atrial fibrillation, stroke, or heart failure; osteoporotic fractures; or deaths.
One patient in the discontinuation group had a TSH level of 11 mIU/L at 6-8 weeks and switched to open-label levothyroxine 75 mcg daily. Another patient in the discontinuation group switched to open-label levothyroxine 75 mcg daily at 10 weeks due to fatigue; however, the patient was diagnosed with metastatic colon cancer 1 month later.
The finding that only about a third of patients who discontinued levothyroxine developed subclinical hypothyroidism was lower than expected, Dr. Maraka noted.
“This was ... unexpected ... for us,” she said. “We were expecting a larger number of patients to develop hypothyroidism, but to our surprise, that was not the case.”
“But what is more important is that there was no difference in the quality of life measures,” she added. “If anything, the placebo group was a little better, though the [differences] were not statistically significant.”
Dr. Maraka also noted that in further research and a final 6-month analysis, the authors will look at factors associated with developing subclinical hypothyroidism after treatment discontinuation, among other issues.
Discontinuation of levothyroxine is manageable
The results are encouraging, as they provide assurance that discontinuation of levothyroxine is manageable.
“This research will pave the way for initiatives to promote levothyroxine deprescription and implementation of evidence-based care for patients with subclinical hypothyroidism,” she said.
In further comments, Dr. Hennessey noted that the dilemma of having patients on levothyroxine who may not be benefitting from treatment is “significant,” with patients sometimes reluctant to discontinue treatment due to concerns of developing hypothyroidism-associated symptoms such as brain fog and weight gain.
He noted, however, that “many with mildly elevated TSH actually go on to normalize with time, so they are not really hypothyroid, [and] if we remove thyroxine from people with normal thyroid function, they will remain normal.”
Dr. Maraka has reported no relevant financial relationships. Dr. Hennessey has reported consulting for pharmaceutical companies to design clinical studies for thyroid medications.
A version of this article first appeared on Medscape.com.
AT ATA 2022
Criminal profiles of medical murderers
Today’s health care professionals run the very real risk of being sued. This is especially true when a patient dies unexpectedly. But many times, doctors find that they’re defending themselves against very serious charges, such as murder and attempted murder.
In Mexico, a physician can be wrongfully accused of such crimes, as was Azucena Calvillo, MD, last year in Durango. The case drew much media attention, and the accusations were so implausible and ridiculous that the charges were dropped and the case was dismissed.
There are instances in which the authorities create a circuslike atmosphere by making farcical and false accusations against healthcare professionals. Still, there are medical murderers – and these killers are among the most difficult to identify. As John E. Douglas put it, “Medical murderers (physicians, nurses, elder care workers) can have a long list of victims, longer than other kinds of serial killers.” Ted Bundy, one of the most written-about serial killers, confessed to 30 murders. The cases discussed below involve from 60 to 200.
Mr. Douglas was a special agent with the United States Federal Bureau of Investigation. He is the author of Mindhunter, a nonfiction crime book in which he recounts the early days of the FBI’s Behavioral Science Unit and how he and his colleagues began to study the criminal profiles of serial killers. The book has been adapted into a Netflix TV series of the same name.
He is also one of the authors of Crime Classification Manual: A Standard System for Investigating and Classifying Violent Crime. In this book, there are descriptions of criminal profiles of medical murderers.
According to the authors, Each type is associated with a different motive. In the former, the murderers believe that they’re alleviating the patient’s suffering; in the latter, the murderers create a medical emergency so that they can play the hero in what they know will be an unsuccessful attempt to save the patient’s life.
Pseudo-mercy homicide
An example of pseudo-mercy homicide is the infamous case of Harold Shipman, MD, who was convicted of killing 15 people, although an investigation found that more than 200 persons, and possibly as many as 250, died at his hands. In Prescription for Murder: The True Story of Dr. Harold Frederick Shipman, biographer Brian Whittle writes that the general practitioner is England’s (if not the world’s) most prolific serial killer. Dr. Shipman is the only physician in that country’s history to have been convicted of killing his patients.
His modus operandi? Injecting morphine. Most of his victims were elderly women. And though unconfirmed, his youngest victim may have been only 4 years old. It was the death of 81-year-old Kathleen Grundy that led to the physician’s arrest. Her family became extremely suspicious when they learned that her will named Dr. Shipman as the beneficiary of her entire estate.
He always denied being involved in the murders, for which authorities have yet to determine a motive. The speculation is that he enjoyed watching people die. Almost none of the cases attributed to Dr. Shipman involved a critically ill individual with a life-threatening condition. Therefore, his acts were not real acts of mercy. He would make a house call to carry out a routine visit. Once in the patient’s home, he would inject a lethal dose of morphine. Sometimes, relatives and physicians alike would be struck by the strange turn of events.
In 2004, Dr. Shipman committed suicide in prison. His case led to numerous changes to British law with respect to the use of controlled substances, the issuance of death certificates, and the procedure for reporting healthcare staff suspected of engaging in illegal activities. Biographer Whittle concluded, “It is very unlikely that the world will ever see another physician as unrelentingly wicked as Dr. Shipman.”
Pseudo-hero homicide
The pseudo-hero creates serious situations, generally by administering drugs, and then tries to save the patient. Mr. Douglas presents a terrifying case study: Genene Jones, a nurse known as the “Angel of Death.”
Many of Ms. Jones’ colleagues considered her an excellent nurse, an expert at handling unexpected emergencies. If a child died while she was on duty, she would sometimes accompany their body to the morgue. She would even sing children’s songs to their lifeless body. When people started to question the number of deaths that were occurring during her shifts, the staff stood up for Ms. Jones, saying that it was because she took on the most serious cases.
Ms. Jones was found out when a vial of succinylcholine went missing. After it was located, a physician, who had been suspicious of the nurse, noticed that there were two puncture holes in the stopper. None of the staff could offer any explanation. A few days before this event, that same physician had left a healthy 15-month-old girl in Jones’ care. Within a few minutes, the child was showing signs of paralysis and started to have seizures. It appears that Ms. Jones had used succinylcholine to make it appear that the children were sick or were experiencing some sort of emergency so that she could then attempt to save them, and they could die in her arms.
This case highlights the need for mortality review committees and for proper statistical analysis to discern trends in deaths and complications among patients. Genene Jones was convicted of killing the 15-month-old girl and was sentenced to 99 years in prison. Authorities suspect that the nurse was responsible for the deaths of up to 60 children.
A new criminal profile?
Through the podcast and subsequent TV series Dr. Death, many people have come to know of a more recent medical murderer: Christopher Duntsch, MD, PhD. The Texas neurosurgeon killed at least two patients, and his actions left several others with adverse outcomes and serious injuries.
These acts occurred during surgical procedures. Witnesses said that the deaths and injuries were the result of unprecedented, egregious negligence, as though the operations had been performed by someone who had never been trained in the specialty. This is something that resonates very strongly for those who are aware of what’s going on in Mexico, where it’s well known that many physicians who lack specialty training perform operations (mainly cosmetic surgery). No doubt cases such as Dr. Duntsch’s are more frequent in Mexico.
What makes the situation in the United States involving Christopher Duntsch so astonishing is that it resulted from a perfect storm of a physician whom some colleagues described as a “sociopath” and legal loopholes in the country’s healthcare system. Apparently, during his residency, Dr. Duntsch never developed the skills necessary to perform operations. He spent more time carrying out research and engaging in other activities than in participating in the operating room. This is a case that calls into question the way specialists are trained, as it seems that what matters is not how much time they’re spending inside the hospital but what they’re doing and learning there.
Dr. Duntsch’s license was suspended and then permanently revoked. He is currently serving a life sentence. Through the podcast or the TV series, one comes to realize that it’s not easy to catch medical murderers. They are among the most difficult to identify – serial killers who commit numerous homicides before they are captured. Reading about the case of Christopher Duntsch, one might ask, What’s his criminal profile: pseudo-hero? Pseudo-mercy? It is hard to say. Maybe his is a different kind of profile – one that will open a new chapter in the books on medical murderers.
Dr. Sarmiento studied medicine and did his residency in anatomic pathology, internal medicine, and clinical hematology. He went on to study at Central University City Campus Law School, National Autonomous University of Mexico. He now runs a law firm that, among other things, advises physicians on matters of civil liability, administrative processes, and the legal implications of practicing medicine.
This article was translated from the Medscape Spanish edition.
Today’s health care professionals run the very real risk of being sued. This is especially true when a patient dies unexpectedly. But many times, doctors find that they’re defending themselves against very serious charges, such as murder and attempted murder.
In Mexico, a physician can be wrongfully accused of such crimes, as was Azucena Calvillo, MD, last year in Durango. The case drew much media attention, and the accusations were so implausible and ridiculous that the charges were dropped and the case was dismissed.
There are instances in which the authorities create a circuslike atmosphere by making farcical and false accusations against healthcare professionals. Still, there are medical murderers – and these killers are among the most difficult to identify. As John E. Douglas put it, “Medical murderers (physicians, nurses, elder care workers) can have a long list of victims, longer than other kinds of serial killers.” Ted Bundy, one of the most written-about serial killers, confessed to 30 murders. The cases discussed below involve from 60 to 200.
Mr. Douglas was a special agent with the United States Federal Bureau of Investigation. He is the author of Mindhunter, a nonfiction crime book in which he recounts the early days of the FBI’s Behavioral Science Unit and how he and his colleagues began to study the criminal profiles of serial killers. The book has been adapted into a Netflix TV series of the same name.
He is also one of the authors of Crime Classification Manual: A Standard System for Investigating and Classifying Violent Crime. In this book, there are descriptions of criminal profiles of medical murderers.
According to the authors, Each type is associated with a different motive. In the former, the murderers believe that they’re alleviating the patient’s suffering; in the latter, the murderers create a medical emergency so that they can play the hero in what they know will be an unsuccessful attempt to save the patient’s life.
Pseudo-mercy homicide
An example of pseudo-mercy homicide is the infamous case of Harold Shipman, MD, who was convicted of killing 15 people, although an investigation found that more than 200 persons, and possibly as many as 250, died at his hands. In Prescription for Murder: The True Story of Dr. Harold Frederick Shipman, biographer Brian Whittle writes that the general practitioner is England’s (if not the world’s) most prolific serial killer. Dr. Shipman is the only physician in that country’s history to have been convicted of killing his patients.
His modus operandi? Injecting morphine. Most of his victims were elderly women. And though unconfirmed, his youngest victim may have been only 4 years old. It was the death of 81-year-old Kathleen Grundy that led to the physician’s arrest. Her family became extremely suspicious when they learned that her will named Dr. Shipman as the beneficiary of her entire estate.
He always denied being involved in the murders, for which authorities have yet to determine a motive. The speculation is that he enjoyed watching people die. Almost none of the cases attributed to Dr. Shipman involved a critically ill individual with a life-threatening condition. Therefore, his acts were not real acts of mercy. He would make a house call to carry out a routine visit. Once in the patient’s home, he would inject a lethal dose of morphine. Sometimes, relatives and physicians alike would be struck by the strange turn of events.
In 2004, Dr. Shipman committed suicide in prison. His case led to numerous changes to British law with respect to the use of controlled substances, the issuance of death certificates, and the procedure for reporting healthcare staff suspected of engaging in illegal activities. Biographer Whittle concluded, “It is very unlikely that the world will ever see another physician as unrelentingly wicked as Dr. Shipman.”
Pseudo-hero homicide
The pseudo-hero creates serious situations, generally by administering drugs, and then tries to save the patient. Mr. Douglas presents a terrifying case study: Genene Jones, a nurse known as the “Angel of Death.”
Many of Ms. Jones’ colleagues considered her an excellent nurse, an expert at handling unexpected emergencies. If a child died while she was on duty, she would sometimes accompany their body to the morgue. She would even sing children’s songs to their lifeless body. When people started to question the number of deaths that were occurring during her shifts, the staff stood up for Ms. Jones, saying that it was because she took on the most serious cases.
Ms. Jones was found out when a vial of succinylcholine went missing. After it was located, a physician, who had been suspicious of the nurse, noticed that there were two puncture holes in the stopper. None of the staff could offer any explanation. A few days before this event, that same physician had left a healthy 15-month-old girl in Jones’ care. Within a few minutes, the child was showing signs of paralysis and started to have seizures. It appears that Ms. Jones had used succinylcholine to make it appear that the children were sick or were experiencing some sort of emergency so that she could then attempt to save them, and they could die in her arms.
This case highlights the need for mortality review committees and for proper statistical analysis to discern trends in deaths and complications among patients. Genene Jones was convicted of killing the 15-month-old girl and was sentenced to 99 years in prison. Authorities suspect that the nurse was responsible for the deaths of up to 60 children.
A new criminal profile?
Through the podcast and subsequent TV series Dr. Death, many people have come to know of a more recent medical murderer: Christopher Duntsch, MD, PhD. The Texas neurosurgeon killed at least two patients, and his actions left several others with adverse outcomes and serious injuries.
These acts occurred during surgical procedures. Witnesses said that the deaths and injuries were the result of unprecedented, egregious negligence, as though the operations had been performed by someone who had never been trained in the specialty. This is something that resonates very strongly for those who are aware of what’s going on in Mexico, where it’s well known that many physicians who lack specialty training perform operations (mainly cosmetic surgery). No doubt cases such as Dr. Duntsch’s are more frequent in Mexico.
What makes the situation in the United States involving Christopher Duntsch so astonishing is that it resulted from a perfect storm of a physician whom some colleagues described as a “sociopath” and legal loopholes in the country’s healthcare system. Apparently, during his residency, Dr. Duntsch never developed the skills necessary to perform operations. He spent more time carrying out research and engaging in other activities than in participating in the operating room. This is a case that calls into question the way specialists are trained, as it seems that what matters is not how much time they’re spending inside the hospital but what they’re doing and learning there.
Dr. Duntsch’s license was suspended and then permanently revoked. He is currently serving a life sentence. Through the podcast or the TV series, one comes to realize that it’s not easy to catch medical murderers. They are among the most difficult to identify – serial killers who commit numerous homicides before they are captured. Reading about the case of Christopher Duntsch, one might ask, What’s his criminal profile: pseudo-hero? Pseudo-mercy? It is hard to say. Maybe his is a different kind of profile – one that will open a new chapter in the books on medical murderers.
Dr. Sarmiento studied medicine and did his residency in anatomic pathology, internal medicine, and clinical hematology. He went on to study at Central University City Campus Law School, National Autonomous University of Mexico. He now runs a law firm that, among other things, advises physicians on matters of civil liability, administrative processes, and the legal implications of practicing medicine.
This article was translated from the Medscape Spanish edition.
Today’s health care professionals run the very real risk of being sued. This is especially true when a patient dies unexpectedly. But many times, doctors find that they’re defending themselves against very serious charges, such as murder and attempted murder.
In Mexico, a physician can be wrongfully accused of such crimes, as was Azucena Calvillo, MD, last year in Durango. The case drew much media attention, and the accusations were so implausible and ridiculous that the charges were dropped and the case was dismissed.
There are instances in which the authorities create a circuslike atmosphere by making farcical and false accusations against healthcare professionals. Still, there are medical murderers – and these killers are among the most difficult to identify. As John E. Douglas put it, “Medical murderers (physicians, nurses, elder care workers) can have a long list of victims, longer than other kinds of serial killers.” Ted Bundy, one of the most written-about serial killers, confessed to 30 murders. The cases discussed below involve from 60 to 200.
Mr. Douglas was a special agent with the United States Federal Bureau of Investigation. He is the author of Mindhunter, a nonfiction crime book in which he recounts the early days of the FBI’s Behavioral Science Unit and how he and his colleagues began to study the criminal profiles of serial killers. The book has been adapted into a Netflix TV series of the same name.
He is also one of the authors of Crime Classification Manual: A Standard System for Investigating and Classifying Violent Crime. In this book, there are descriptions of criminal profiles of medical murderers.
According to the authors, Each type is associated with a different motive. In the former, the murderers believe that they’re alleviating the patient’s suffering; in the latter, the murderers create a medical emergency so that they can play the hero in what they know will be an unsuccessful attempt to save the patient’s life.
Pseudo-mercy homicide
An example of pseudo-mercy homicide is the infamous case of Harold Shipman, MD, who was convicted of killing 15 people, although an investigation found that more than 200 persons, and possibly as many as 250, died at his hands. In Prescription for Murder: The True Story of Dr. Harold Frederick Shipman, biographer Brian Whittle writes that the general practitioner is England’s (if not the world’s) most prolific serial killer. Dr. Shipman is the only physician in that country’s history to have been convicted of killing his patients.
His modus operandi? Injecting morphine. Most of his victims were elderly women. And though unconfirmed, his youngest victim may have been only 4 years old. It was the death of 81-year-old Kathleen Grundy that led to the physician’s arrest. Her family became extremely suspicious when they learned that her will named Dr. Shipman as the beneficiary of her entire estate.
He always denied being involved in the murders, for which authorities have yet to determine a motive. The speculation is that he enjoyed watching people die. Almost none of the cases attributed to Dr. Shipman involved a critically ill individual with a life-threatening condition. Therefore, his acts were not real acts of mercy. He would make a house call to carry out a routine visit. Once in the patient’s home, he would inject a lethal dose of morphine. Sometimes, relatives and physicians alike would be struck by the strange turn of events.
In 2004, Dr. Shipman committed suicide in prison. His case led to numerous changes to British law with respect to the use of controlled substances, the issuance of death certificates, and the procedure for reporting healthcare staff suspected of engaging in illegal activities. Biographer Whittle concluded, “It is very unlikely that the world will ever see another physician as unrelentingly wicked as Dr. Shipman.”
Pseudo-hero homicide
The pseudo-hero creates serious situations, generally by administering drugs, and then tries to save the patient. Mr. Douglas presents a terrifying case study: Genene Jones, a nurse known as the “Angel of Death.”
Many of Ms. Jones’ colleagues considered her an excellent nurse, an expert at handling unexpected emergencies. If a child died while she was on duty, she would sometimes accompany their body to the morgue. She would even sing children’s songs to their lifeless body. When people started to question the number of deaths that were occurring during her shifts, the staff stood up for Ms. Jones, saying that it was because she took on the most serious cases.
Ms. Jones was found out when a vial of succinylcholine went missing. After it was located, a physician, who had been suspicious of the nurse, noticed that there were two puncture holes in the stopper. None of the staff could offer any explanation. A few days before this event, that same physician had left a healthy 15-month-old girl in Jones’ care. Within a few minutes, the child was showing signs of paralysis and started to have seizures. It appears that Ms. Jones had used succinylcholine to make it appear that the children were sick or were experiencing some sort of emergency so that she could then attempt to save them, and they could die in her arms.
This case highlights the need for mortality review committees and for proper statistical analysis to discern trends in deaths and complications among patients. Genene Jones was convicted of killing the 15-month-old girl and was sentenced to 99 years in prison. Authorities suspect that the nurse was responsible for the deaths of up to 60 children.
A new criminal profile?
Through the podcast and subsequent TV series Dr. Death, many people have come to know of a more recent medical murderer: Christopher Duntsch, MD, PhD. The Texas neurosurgeon killed at least two patients, and his actions left several others with adverse outcomes and serious injuries.
These acts occurred during surgical procedures. Witnesses said that the deaths and injuries were the result of unprecedented, egregious negligence, as though the operations had been performed by someone who had never been trained in the specialty. This is something that resonates very strongly for those who are aware of what’s going on in Mexico, where it’s well known that many physicians who lack specialty training perform operations (mainly cosmetic surgery). No doubt cases such as Dr. Duntsch’s are more frequent in Mexico.
What makes the situation in the United States involving Christopher Duntsch so astonishing is that it resulted from a perfect storm of a physician whom some colleagues described as a “sociopath” and legal loopholes in the country’s healthcare system. Apparently, during his residency, Dr. Duntsch never developed the skills necessary to perform operations. He spent more time carrying out research and engaging in other activities than in participating in the operating room. This is a case that calls into question the way specialists are trained, as it seems that what matters is not how much time they’re spending inside the hospital but what they’re doing and learning there.
Dr. Duntsch’s license was suspended and then permanently revoked. He is currently serving a life sentence. Through the podcast or the TV series, one comes to realize that it’s not easy to catch medical murderers. They are among the most difficult to identify – serial killers who commit numerous homicides before they are captured. Reading about the case of Christopher Duntsch, one might ask, What’s his criminal profile: pseudo-hero? Pseudo-mercy? It is hard to say. Maybe his is a different kind of profile – one that will open a new chapter in the books on medical murderers.
Dr. Sarmiento studied medicine and did his residency in anatomic pathology, internal medicine, and clinical hematology. He went on to study at Central University City Campus Law School, National Autonomous University of Mexico. He now runs a law firm that, among other things, advises physicians on matters of civil liability, administrative processes, and the legal implications of practicing medicine.
This article was translated from the Medscape Spanish edition.
Myocarditis after COVID vax rare and mild in teens
New data from Israel provide further evidence that myocarditis is a rare adverse event of vaccination with the Pfizer/BioNTech mRNA COVID-19 vaccine in adolescents – one that predominantly occurs in males and typically after the second dose.
The new data also indicate a “mild and benign” clinical course of myocarditis after vaccination, with “favorable” long-term prognosis based on cardiac imaging findings.
Guy Witberg, MD, MPH, Rabin Medical Center, Petah Tikva, Israel, and colleagues report their latest observations in correspondence in The New England Journal of Medicine, online.
The group previously reported in December 2021 that the incidence of myocarditis in Israel after receipt of the Pfizer/BioNTech BNT162b2 mRNA COVID-19 vaccine was highest among males between the ages of 16 and 29 (10.7 cases per 100,000).
The vaccine has since been approved for adolescents aged 12-15. Initial evidence for this age group, reported by Dr. Witberg and colleagues in March 2022, suggests a similar low incidence and mild course of myocarditis, although follow-up was limited to 30 days.
In their latest report, with follow-up out to 6 months, Dr. Witberg and colleagues identified nine probable or definite cases of myocarditis among 182,605 Israeli adolescents aged 12-15 who received the Pfizer/BioNTech mRNA vaccine – an incidence of 4.8 cases per 100,000.
Eight cases occurred after the second vaccine dose. All nine cases were mild.
Cardiac and inflammatory markers were elevated in all adolescent patients and electrocardiographic results were abnormal in two-thirds.
Eight patients had a normal ejection fraction, and four had a pericardial effusion. The patients spent 2-4 days hospitalized, and the in-hospital course was uneventful.
Echocardiographic findings were available a median of 10 days after discharge for eight patients. All echocardiograms showed a normal ejection fraction and resolution of pericardial effusion.
Five patients underwent cardiac MRI, including three scans performed at a median of 104 days after discharge. The scans showed “minimal evidence” of myocardial scarring or fibrosis, with evidence of late gadolinium enhancement ranging from 0% to 2%.
At a median of 206 days following discharge, all of the patients were alive, and none had been readmitted to the hospital, Dr. Witberg and colleagues report.
This research had no specific funding. Five authors have received research grants from Pfizer.
A version of this article first appeared on Medscape.com.
New data from Israel provide further evidence that myocarditis is a rare adverse event of vaccination with the Pfizer/BioNTech mRNA COVID-19 vaccine in adolescents – one that predominantly occurs in males and typically after the second dose.
The new data also indicate a “mild and benign” clinical course of myocarditis after vaccination, with “favorable” long-term prognosis based on cardiac imaging findings.
Guy Witberg, MD, MPH, Rabin Medical Center, Petah Tikva, Israel, and colleagues report their latest observations in correspondence in The New England Journal of Medicine, online.
The group previously reported in December 2021 that the incidence of myocarditis in Israel after receipt of the Pfizer/BioNTech BNT162b2 mRNA COVID-19 vaccine was highest among males between the ages of 16 and 29 (10.7 cases per 100,000).
The vaccine has since been approved for adolescents aged 12-15. Initial evidence for this age group, reported by Dr. Witberg and colleagues in March 2022, suggests a similar low incidence and mild course of myocarditis, although follow-up was limited to 30 days.
In their latest report, with follow-up out to 6 months, Dr. Witberg and colleagues identified nine probable or definite cases of myocarditis among 182,605 Israeli adolescents aged 12-15 who received the Pfizer/BioNTech mRNA vaccine – an incidence of 4.8 cases per 100,000.
Eight cases occurred after the second vaccine dose. All nine cases were mild.
Cardiac and inflammatory markers were elevated in all adolescent patients and electrocardiographic results were abnormal in two-thirds.
Eight patients had a normal ejection fraction, and four had a pericardial effusion. The patients spent 2-4 days hospitalized, and the in-hospital course was uneventful.
Echocardiographic findings were available a median of 10 days after discharge for eight patients. All echocardiograms showed a normal ejection fraction and resolution of pericardial effusion.
Five patients underwent cardiac MRI, including three scans performed at a median of 104 days after discharge. The scans showed “minimal evidence” of myocardial scarring or fibrosis, with evidence of late gadolinium enhancement ranging from 0% to 2%.
At a median of 206 days following discharge, all of the patients were alive, and none had been readmitted to the hospital, Dr. Witberg and colleagues report.
This research had no specific funding. Five authors have received research grants from Pfizer.
A version of this article first appeared on Medscape.com.
New data from Israel provide further evidence that myocarditis is a rare adverse event of vaccination with the Pfizer/BioNTech mRNA COVID-19 vaccine in adolescents – one that predominantly occurs in males and typically after the second dose.
The new data also indicate a “mild and benign” clinical course of myocarditis after vaccination, with “favorable” long-term prognosis based on cardiac imaging findings.
Guy Witberg, MD, MPH, Rabin Medical Center, Petah Tikva, Israel, and colleagues report their latest observations in correspondence in The New England Journal of Medicine, online.
The group previously reported in December 2021 that the incidence of myocarditis in Israel after receipt of the Pfizer/BioNTech BNT162b2 mRNA COVID-19 vaccine was highest among males between the ages of 16 and 29 (10.7 cases per 100,000).
The vaccine has since been approved for adolescents aged 12-15. Initial evidence for this age group, reported by Dr. Witberg and colleagues in March 2022, suggests a similar low incidence and mild course of myocarditis, although follow-up was limited to 30 days.
In their latest report, with follow-up out to 6 months, Dr. Witberg and colleagues identified nine probable or definite cases of myocarditis among 182,605 Israeli adolescents aged 12-15 who received the Pfizer/BioNTech mRNA vaccine – an incidence of 4.8 cases per 100,000.
Eight cases occurred after the second vaccine dose. All nine cases were mild.
Cardiac and inflammatory markers were elevated in all adolescent patients and electrocardiographic results were abnormal in two-thirds.
Eight patients had a normal ejection fraction, and four had a pericardial effusion. The patients spent 2-4 days hospitalized, and the in-hospital course was uneventful.
Echocardiographic findings were available a median of 10 days after discharge for eight patients. All echocardiograms showed a normal ejection fraction and resolution of pericardial effusion.
Five patients underwent cardiac MRI, including three scans performed at a median of 104 days after discharge. The scans showed “minimal evidence” of myocardial scarring or fibrosis, with evidence of late gadolinium enhancement ranging from 0% to 2%.
At a median of 206 days following discharge, all of the patients were alive, and none had been readmitted to the hospital, Dr. Witberg and colleagues report.
This research had no specific funding. Five authors have received research grants from Pfizer.
A version of this article first appeared on Medscape.com.
Goodbye ‘diabetes insipidus’, hello ‘AVP-D’ and ‘AVP-R’
An international group representing leading endocrinology associations has recommended that the name “diabetes insipidus” – which in some cases has led to harm – be changed to eliminate confusion with “diabetes mellitus” and to reflect the former condition’s pathophysiology.
The new proposed names are arginine vasopressin deficiency (AVP-D) for central (also called “cranial”) etiologies and arginine vasopressin resistance (AVP-R) for nephrogenic (kidney) etiologies.
“What we’re proposing is to rename the disease according to the pathophysiology that defines it,” statement co-author Joseph G. Verbalis, MD, professor of medicine and chief of endocrinology and metabolism at Georgetown University Medical Center, Washington, told this news organization.
The statement advises that henceforth the new names be used in manuscripts and the medical literature while keeping the old names in parentheses during a transition period, as in “AVP-deficiency (cranial diabetes insipidus)” and “AVP-resistance (nephrogenic diabetes insipidus).”
The condition formerly known as diabetes insipidus is relatively rare, occurring in about 1 person per 10-15,000 population. It is caused by either deficient production or resistance in the kidney to the hormone AVP, normally produced by the hypothalamus and stored in the pituitary gland. AVP, also called antidiuretic hormone, regulates the body’s water level and urine production by the kidney.
Both etiologies lead to extreme thirst and excessive production of urine. Common causes of the deficiency include head trauma or brain tumor, while resistance in the kidney is often congenital. It is currently treated with a synthetic form of AVP called desmopressin and fluid replacement.
What’s in a name?
The proposal to change the name by the Working Group for Renaming Diabetes Insipidus is endorsed by The Endocrine Society, European Society of Endocrinology, Pituitary Society, Society for Endocrinology, European Society for Paediatric Endocrinology, Endocrine Society of Australia, Brazilian Endocrine Society, and Japanese Endocrine Society and is under review by several other societies. It was published as a position statement in several of those society’s journals, with more to follow.
Historically, the word “diabetes,” a Greek word meaning “siphon,” was used in the 1st and 2nd century BC to describe excess flow of urine. The Latin word “mellitus” or “honey” was added in the late 17th century to describe the sweetness of the urine in the dysglycemic condition.
A century later, the Latin word “insipidus,” meaning insipid or tasteless, was coined to distinguish between the two types of polyuria, the position statement details.
In the late 19th to early 20th century, the vasopressor and antidiuretic actions of posterior pituitary extracts were discovered and used to treat people with both the central and nephrogenic etiologies, which were also recognized around that time, yet the name “diabetes insipidus” has persisted.
“From a historical perspective, the name is perfectly appropriate. At the time it was identified, and it was realized that it was different from diabetes mellitus, that was a perfectly appropriate terminology based on what was known in the late 19th century – but not now. It has persisted through the years simply because in medicine there’s a lot of inertia for change ... It’s just always been called that. If there’s not a compelling reason to change a name, generally there’s no move to change it,” Dr. Verbalis observed.
‘Dramatic cases of patient mismanagement’ due to name confusion
Unfortunately, the urgency for the change arose from tragedy. In 2009, a 22-year-old man was admitted to the orthopedics department of a London teaching hospital for a hip replacement. Despite his known panhypopituitarism and diabetes insipidus, the nurses continually checked his blood glucose but didn’t give him desmopressin or sufficient fluids. Laboratory testing showed normal glucose, but his serum sodium was 149 mmol/L. The morning after his operation, he had a fatal cardiac arrest with a serum sodium of 169 mmol/L.
“The nurses thought he had diabetes mellitus ... So that was death due to failure to recognize that diabetes insipidus is not diabetes mellitus,” Dr. Verbalis said. “If he had been admitted to endocrinology, this wouldn’t have happened. But he was admitted to orthopedics. Non-endocrinologists are not so aware of diabetes insipidus, because it is a rare disease.”
In 2016, National Health Service England issued a patient safety alert about the “risk of severe harm or death when desmopressin is omitted or delayed in patients with cranial diabetes insipidus,” citing at least four incidents within the prior 7 years where omission of desmopressin had resulted in severe dehydration and death, with another 76 cases of omission or delay that were acted on before the patients became critically ill.
Further impetus for the name change came from the results of an anonymous web-based survey of 1,034 adult and pediatric patients with central diabetes insipidus conducted between August 2021 and February 2022. Overall, 80% reported encountering situations in which their condition had been confused with diabetes mellitus by health care professionals, and 85% supported renaming the disease.
There was some divergence in opinion as to what the new name(s) should be, but clear agreement that the term “diabetes” should not be part of it.
“We’ve only become recently aware that there are dramatic cases of patient mismanagement due to the confusion caused by the word ‘diabetes.’ We think patients should have a voice. If a legitimate patient survey says over 80% think this name should be changed, then I think we as endocrinologists need to pay attention to that,” Dr. Verbalis said.
But while endocrinologists are the ones who see these patients the most often, Dr. Verbalis said a main aim of the position statement “is really to change the mindset of non-endocrinologist doctors and nurses and other health care professionals that this is not diabetes mellitus. It’s a totally different disease. And if we give it a totally different name, then I think they will better recognize that.”
As to how long Dr. Verbalis thinks it will take for the new names to catch on, he pointed out that it’s taken about a decade for the rheumatology field to fully adopt the name “granulomatosis with polyangiitis” as a replacement for “Wegener’s granulomatosis” after the eponymous physician’s Nazi ties were revealed.
“So we’re not anticipating that this is going to change terminology tomorrow. It’s a long process. We just wanted to get the process started,” he said.
Dr. Verbalis has reported consulting for Otsuka.
A version of this article first appeared on Medscape.com.
An international group representing leading endocrinology associations has recommended that the name “diabetes insipidus” – which in some cases has led to harm – be changed to eliminate confusion with “diabetes mellitus” and to reflect the former condition’s pathophysiology.
The new proposed names are arginine vasopressin deficiency (AVP-D) for central (also called “cranial”) etiologies and arginine vasopressin resistance (AVP-R) for nephrogenic (kidney) etiologies.
“What we’re proposing is to rename the disease according to the pathophysiology that defines it,” statement co-author Joseph G. Verbalis, MD, professor of medicine and chief of endocrinology and metabolism at Georgetown University Medical Center, Washington, told this news organization.
The statement advises that henceforth the new names be used in manuscripts and the medical literature while keeping the old names in parentheses during a transition period, as in “AVP-deficiency (cranial diabetes insipidus)” and “AVP-resistance (nephrogenic diabetes insipidus).”
The condition formerly known as diabetes insipidus is relatively rare, occurring in about 1 person per 10-15,000 population. It is caused by either deficient production or resistance in the kidney to the hormone AVP, normally produced by the hypothalamus and stored in the pituitary gland. AVP, also called antidiuretic hormone, regulates the body’s water level and urine production by the kidney.
Both etiologies lead to extreme thirst and excessive production of urine. Common causes of the deficiency include head trauma or brain tumor, while resistance in the kidney is often congenital. It is currently treated with a synthetic form of AVP called desmopressin and fluid replacement.
What’s in a name?
The proposal to change the name by the Working Group for Renaming Diabetes Insipidus is endorsed by The Endocrine Society, European Society of Endocrinology, Pituitary Society, Society for Endocrinology, European Society for Paediatric Endocrinology, Endocrine Society of Australia, Brazilian Endocrine Society, and Japanese Endocrine Society and is under review by several other societies. It was published as a position statement in several of those society’s journals, with more to follow.
Historically, the word “diabetes,” a Greek word meaning “siphon,” was used in the 1st and 2nd century BC to describe excess flow of urine. The Latin word “mellitus” or “honey” was added in the late 17th century to describe the sweetness of the urine in the dysglycemic condition.
A century later, the Latin word “insipidus,” meaning insipid or tasteless, was coined to distinguish between the two types of polyuria, the position statement details.
In the late 19th to early 20th century, the vasopressor and antidiuretic actions of posterior pituitary extracts were discovered and used to treat people with both the central and nephrogenic etiologies, which were also recognized around that time, yet the name “diabetes insipidus” has persisted.
“From a historical perspective, the name is perfectly appropriate. At the time it was identified, and it was realized that it was different from diabetes mellitus, that was a perfectly appropriate terminology based on what was known in the late 19th century – but not now. It has persisted through the years simply because in medicine there’s a lot of inertia for change ... It’s just always been called that. If there’s not a compelling reason to change a name, generally there’s no move to change it,” Dr. Verbalis observed.
‘Dramatic cases of patient mismanagement’ due to name confusion
Unfortunately, the urgency for the change arose from tragedy. In 2009, a 22-year-old man was admitted to the orthopedics department of a London teaching hospital for a hip replacement. Despite his known panhypopituitarism and diabetes insipidus, the nurses continually checked his blood glucose but didn’t give him desmopressin or sufficient fluids. Laboratory testing showed normal glucose, but his serum sodium was 149 mmol/L. The morning after his operation, he had a fatal cardiac arrest with a serum sodium of 169 mmol/L.
“The nurses thought he had diabetes mellitus ... So that was death due to failure to recognize that diabetes insipidus is not diabetes mellitus,” Dr. Verbalis said. “If he had been admitted to endocrinology, this wouldn’t have happened. But he was admitted to orthopedics. Non-endocrinologists are not so aware of diabetes insipidus, because it is a rare disease.”
In 2016, National Health Service England issued a patient safety alert about the “risk of severe harm or death when desmopressin is omitted or delayed in patients with cranial diabetes insipidus,” citing at least four incidents within the prior 7 years where omission of desmopressin had resulted in severe dehydration and death, with another 76 cases of omission or delay that were acted on before the patients became critically ill.
Further impetus for the name change came from the results of an anonymous web-based survey of 1,034 adult and pediatric patients with central diabetes insipidus conducted between August 2021 and February 2022. Overall, 80% reported encountering situations in which their condition had been confused with diabetes mellitus by health care professionals, and 85% supported renaming the disease.
There was some divergence in opinion as to what the new name(s) should be, but clear agreement that the term “diabetes” should not be part of it.
“We’ve only become recently aware that there are dramatic cases of patient mismanagement due to the confusion caused by the word ‘diabetes.’ We think patients should have a voice. If a legitimate patient survey says over 80% think this name should be changed, then I think we as endocrinologists need to pay attention to that,” Dr. Verbalis said.
But while endocrinologists are the ones who see these patients the most often, Dr. Verbalis said a main aim of the position statement “is really to change the mindset of non-endocrinologist doctors and nurses and other health care professionals that this is not diabetes mellitus. It’s a totally different disease. And if we give it a totally different name, then I think they will better recognize that.”
As to how long Dr. Verbalis thinks it will take for the new names to catch on, he pointed out that it’s taken about a decade for the rheumatology field to fully adopt the name “granulomatosis with polyangiitis” as a replacement for “Wegener’s granulomatosis” after the eponymous physician’s Nazi ties were revealed.
“So we’re not anticipating that this is going to change terminology tomorrow. It’s a long process. We just wanted to get the process started,” he said.
Dr. Verbalis has reported consulting for Otsuka.
A version of this article first appeared on Medscape.com.
An international group representing leading endocrinology associations has recommended that the name “diabetes insipidus” – which in some cases has led to harm – be changed to eliminate confusion with “diabetes mellitus” and to reflect the former condition’s pathophysiology.
The new proposed names are arginine vasopressin deficiency (AVP-D) for central (also called “cranial”) etiologies and arginine vasopressin resistance (AVP-R) for nephrogenic (kidney) etiologies.
“What we’re proposing is to rename the disease according to the pathophysiology that defines it,” statement co-author Joseph G. Verbalis, MD, professor of medicine and chief of endocrinology and metabolism at Georgetown University Medical Center, Washington, told this news organization.
The statement advises that henceforth the new names be used in manuscripts and the medical literature while keeping the old names in parentheses during a transition period, as in “AVP-deficiency (cranial diabetes insipidus)” and “AVP-resistance (nephrogenic diabetes insipidus).”
The condition formerly known as diabetes insipidus is relatively rare, occurring in about 1 person per 10-15,000 population. It is caused by either deficient production or resistance in the kidney to the hormone AVP, normally produced by the hypothalamus and stored in the pituitary gland. AVP, also called antidiuretic hormone, regulates the body’s water level and urine production by the kidney.
Both etiologies lead to extreme thirst and excessive production of urine. Common causes of the deficiency include head trauma or brain tumor, while resistance in the kidney is often congenital. It is currently treated with a synthetic form of AVP called desmopressin and fluid replacement.
What’s in a name?
The proposal to change the name by the Working Group for Renaming Diabetes Insipidus is endorsed by The Endocrine Society, European Society of Endocrinology, Pituitary Society, Society for Endocrinology, European Society for Paediatric Endocrinology, Endocrine Society of Australia, Brazilian Endocrine Society, and Japanese Endocrine Society and is under review by several other societies. It was published as a position statement in several of those society’s journals, with more to follow.
Historically, the word “diabetes,” a Greek word meaning “siphon,” was used in the 1st and 2nd century BC to describe excess flow of urine. The Latin word “mellitus” or “honey” was added in the late 17th century to describe the sweetness of the urine in the dysglycemic condition.
A century later, the Latin word “insipidus,” meaning insipid or tasteless, was coined to distinguish between the two types of polyuria, the position statement details.
In the late 19th to early 20th century, the vasopressor and antidiuretic actions of posterior pituitary extracts were discovered and used to treat people with both the central and nephrogenic etiologies, which were also recognized around that time, yet the name “diabetes insipidus” has persisted.
“From a historical perspective, the name is perfectly appropriate. At the time it was identified, and it was realized that it was different from diabetes mellitus, that was a perfectly appropriate terminology based on what was known in the late 19th century – but not now. It has persisted through the years simply because in medicine there’s a lot of inertia for change ... It’s just always been called that. If there’s not a compelling reason to change a name, generally there’s no move to change it,” Dr. Verbalis observed.
‘Dramatic cases of patient mismanagement’ due to name confusion
Unfortunately, the urgency for the change arose from tragedy. In 2009, a 22-year-old man was admitted to the orthopedics department of a London teaching hospital for a hip replacement. Despite his known panhypopituitarism and diabetes insipidus, the nurses continually checked his blood glucose but didn’t give him desmopressin or sufficient fluids. Laboratory testing showed normal glucose, but his serum sodium was 149 mmol/L. The morning after his operation, he had a fatal cardiac arrest with a serum sodium of 169 mmol/L.
“The nurses thought he had diabetes mellitus ... So that was death due to failure to recognize that diabetes insipidus is not diabetes mellitus,” Dr. Verbalis said. “If he had been admitted to endocrinology, this wouldn’t have happened. But he was admitted to orthopedics. Non-endocrinologists are not so aware of diabetes insipidus, because it is a rare disease.”
In 2016, National Health Service England issued a patient safety alert about the “risk of severe harm or death when desmopressin is omitted or delayed in patients with cranial diabetes insipidus,” citing at least four incidents within the prior 7 years where omission of desmopressin had resulted in severe dehydration and death, with another 76 cases of omission or delay that were acted on before the patients became critically ill.
Further impetus for the name change came from the results of an anonymous web-based survey of 1,034 adult and pediatric patients with central diabetes insipidus conducted between August 2021 and February 2022. Overall, 80% reported encountering situations in which their condition had been confused with diabetes mellitus by health care professionals, and 85% supported renaming the disease.
There was some divergence in opinion as to what the new name(s) should be, but clear agreement that the term “diabetes” should not be part of it.
“We’ve only become recently aware that there are dramatic cases of patient mismanagement due to the confusion caused by the word ‘diabetes.’ We think patients should have a voice. If a legitimate patient survey says over 80% think this name should be changed, then I think we as endocrinologists need to pay attention to that,” Dr. Verbalis said.
But while endocrinologists are the ones who see these patients the most often, Dr. Verbalis said a main aim of the position statement “is really to change the mindset of non-endocrinologist doctors and nurses and other health care professionals that this is not diabetes mellitus. It’s a totally different disease. And if we give it a totally different name, then I think they will better recognize that.”
As to how long Dr. Verbalis thinks it will take for the new names to catch on, he pointed out that it’s taken about a decade for the rheumatology field to fully adopt the name “granulomatosis with polyangiitis” as a replacement for “Wegener’s granulomatosis” after the eponymous physician’s Nazi ties were revealed.
“So we’re not anticipating that this is going to change terminology tomorrow. It’s a long process. We just wanted to get the process started,” he said.
Dr. Verbalis has reported consulting for Otsuka.
A version of this article first appeared on Medscape.com.
Life expectancy 6.3 years shorter for Black MSM with HIV
WASHINGTON –
Lead author Katherine Rich, MPH, a student at Harvard Medical School, Boston, presented the data during an annual scientific meeting on infectious diseases.
“Substantial disparities in care exist between Black and White MSM here in the United States,” she said. “The 2030 goals of the EHE [Ending the HIV Epidemic in the U.S. initiative] won’t be met until HIV-related disparities are reduced.”
Using modeling, the team was able to measure both the gaps and the potential of interventions to address those gaps.
- The team found that improving engagement in care had the largest benefit in narrowing the gap. Improving engagement and retention in care, they write, would result in a gain of 1.4 life-years for Black MSM and 1 year for White MSM.
- Annual testing would add 0.6 life-years for Black MSM and 0.3 life-years for White MSM, compared with standard care.
- In simulating viral suppression, the model-predicted gain would be 1.1 years for Black MSM and 0.3 for White.
Furthermore, a combination of annual testing, 95% engagement in care, and 95% virologic suppression would add 3.4 years for Black MSM (more than double the increase in life-years for any one intervention) and 1.6 years for their White counterparts, the research suggests.
The researchers projected life expectancy from age 15 to be 52.2 years for Black MSM (or 67.2 years old) and 58.5 years from age 15 for White MSM (or 73.5 years old), a difference of 6.3 years.
Kathleen McManus, MD, assistant professor of medicine in infectious diseases and international health at the University of Virginia, Charlottesville, said in an interview that the projected gap in years of life should be a call to action. Dr. McManus was not involved with the study.
Life expectancy gap ‘alarming’
“It is alarming that with current usual HIV care Black MSM with HIV have 6.3 fewer years of life expectancy than White MSM,” she said. “Black MSM having lower retention in care and a lower rate of viral suppression than White MSM demonstrates that there is a problem with our current health care delivery to Black MSM.”
With qualitative, community-engaged research, she said, “we need to ask the Black MSM community what care innovations they need – and then we need clinics and organizations to make the identified changes.”
Researchers used the validated CEPAC (Cost-Effectiveness of Preventing AIDS Complications) microsimulation HIV model to project life expectancy. Using data from the United States Centers for Disease Control and Prevention, they estimated the average age at HIV infection to be 26.8 years for Black MSM and 35 years for White MSM.
They estimated the proportion of time that MSM with diagnosed HIV are retained in care to be 75.2% for Black MSM and 80.6% for White MSM. They calculated the proportion who achieve virologic suppression to be 82% for Black MSM and 91.2% for White MSM.
Senior author Emily P. Hyle, MD, associate professor of medicine at Harvard and infectious diseases physician at Massachusetts General Hospital, both in Boston, said in a press conference before the presentation that strategies to narrow the gap will look different by region.
“Our study highlights that if you can find effective interventions, the effect can be incredibly large. These are very large differences in life-years and life expectancies,” she said.
Ms. Rich gave an example of promising interventions by pointing to work by study coauthor Aima Ahonkhai, MD, MPH, assistant professor of medicine at Vanderbilt University, Nashville, Tenn., who has received federal funding to pursue research on whether preventive care outreach in barbershops can improve prevention for Black men with HIV.
Ms. Rich noted that the modeling has limitations in that it focused on health outcomes and did not simulate transmissions. Results also reflect national data and not local HIV care continuums, which, she acknowledged, differ substantially.
Dr. McManus and the study authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON –
Lead author Katherine Rich, MPH, a student at Harvard Medical School, Boston, presented the data during an annual scientific meeting on infectious diseases.
“Substantial disparities in care exist between Black and White MSM here in the United States,” she said. “The 2030 goals of the EHE [Ending the HIV Epidemic in the U.S. initiative] won’t be met until HIV-related disparities are reduced.”
Using modeling, the team was able to measure both the gaps and the potential of interventions to address those gaps.
- The team found that improving engagement in care had the largest benefit in narrowing the gap. Improving engagement and retention in care, they write, would result in a gain of 1.4 life-years for Black MSM and 1 year for White MSM.
- Annual testing would add 0.6 life-years for Black MSM and 0.3 life-years for White MSM, compared with standard care.
- In simulating viral suppression, the model-predicted gain would be 1.1 years for Black MSM and 0.3 for White.
Furthermore, a combination of annual testing, 95% engagement in care, and 95% virologic suppression would add 3.4 years for Black MSM (more than double the increase in life-years for any one intervention) and 1.6 years for their White counterparts, the research suggests.
The researchers projected life expectancy from age 15 to be 52.2 years for Black MSM (or 67.2 years old) and 58.5 years from age 15 for White MSM (or 73.5 years old), a difference of 6.3 years.
Kathleen McManus, MD, assistant professor of medicine in infectious diseases and international health at the University of Virginia, Charlottesville, said in an interview that the projected gap in years of life should be a call to action. Dr. McManus was not involved with the study.
Life expectancy gap ‘alarming’
“It is alarming that with current usual HIV care Black MSM with HIV have 6.3 fewer years of life expectancy than White MSM,” she said. “Black MSM having lower retention in care and a lower rate of viral suppression than White MSM demonstrates that there is a problem with our current health care delivery to Black MSM.”
With qualitative, community-engaged research, she said, “we need to ask the Black MSM community what care innovations they need – and then we need clinics and organizations to make the identified changes.”
Researchers used the validated CEPAC (Cost-Effectiveness of Preventing AIDS Complications) microsimulation HIV model to project life expectancy. Using data from the United States Centers for Disease Control and Prevention, they estimated the average age at HIV infection to be 26.8 years for Black MSM and 35 years for White MSM.
They estimated the proportion of time that MSM with diagnosed HIV are retained in care to be 75.2% for Black MSM and 80.6% for White MSM. They calculated the proportion who achieve virologic suppression to be 82% for Black MSM and 91.2% for White MSM.
Senior author Emily P. Hyle, MD, associate professor of medicine at Harvard and infectious diseases physician at Massachusetts General Hospital, both in Boston, said in a press conference before the presentation that strategies to narrow the gap will look different by region.
“Our study highlights that if you can find effective interventions, the effect can be incredibly large. These are very large differences in life-years and life expectancies,” she said.
Ms. Rich gave an example of promising interventions by pointing to work by study coauthor Aima Ahonkhai, MD, MPH, assistant professor of medicine at Vanderbilt University, Nashville, Tenn., who has received federal funding to pursue research on whether preventive care outreach in barbershops can improve prevention for Black men with HIV.
Ms. Rich noted that the modeling has limitations in that it focused on health outcomes and did not simulate transmissions. Results also reflect national data and not local HIV care continuums, which, she acknowledged, differ substantially.
Dr. McManus and the study authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON –
Lead author Katherine Rich, MPH, a student at Harvard Medical School, Boston, presented the data during an annual scientific meeting on infectious diseases.
“Substantial disparities in care exist between Black and White MSM here in the United States,” she said. “The 2030 goals of the EHE [Ending the HIV Epidemic in the U.S. initiative] won’t be met until HIV-related disparities are reduced.”
Using modeling, the team was able to measure both the gaps and the potential of interventions to address those gaps.
- The team found that improving engagement in care had the largest benefit in narrowing the gap. Improving engagement and retention in care, they write, would result in a gain of 1.4 life-years for Black MSM and 1 year for White MSM.
- Annual testing would add 0.6 life-years for Black MSM and 0.3 life-years for White MSM, compared with standard care.
- In simulating viral suppression, the model-predicted gain would be 1.1 years for Black MSM and 0.3 for White.
Furthermore, a combination of annual testing, 95% engagement in care, and 95% virologic suppression would add 3.4 years for Black MSM (more than double the increase in life-years for any one intervention) and 1.6 years for their White counterparts, the research suggests.
The researchers projected life expectancy from age 15 to be 52.2 years for Black MSM (or 67.2 years old) and 58.5 years from age 15 for White MSM (or 73.5 years old), a difference of 6.3 years.
Kathleen McManus, MD, assistant professor of medicine in infectious diseases and international health at the University of Virginia, Charlottesville, said in an interview that the projected gap in years of life should be a call to action. Dr. McManus was not involved with the study.
Life expectancy gap ‘alarming’
“It is alarming that with current usual HIV care Black MSM with HIV have 6.3 fewer years of life expectancy than White MSM,” she said. “Black MSM having lower retention in care and a lower rate of viral suppression than White MSM demonstrates that there is a problem with our current health care delivery to Black MSM.”
With qualitative, community-engaged research, she said, “we need to ask the Black MSM community what care innovations they need – and then we need clinics and organizations to make the identified changes.”
Researchers used the validated CEPAC (Cost-Effectiveness of Preventing AIDS Complications) microsimulation HIV model to project life expectancy. Using data from the United States Centers for Disease Control and Prevention, they estimated the average age at HIV infection to be 26.8 years for Black MSM and 35 years for White MSM.
They estimated the proportion of time that MSM with diagnosed HIV are retained in care to be 75.2% for Black MSM and 80.6% for White MSM. They calculated the proportion who achieve virologic suppression to be 82% for Black MSM and 91.2% for White MSM.
Senior author Emily P. Hyle, MD, associate professor of medicine at Harvard and infectious diseases physician at Massachusetts General Hospital, both in Boston, said in a press conference before the presentation that strategies to narrow the gap will look different by region.
“Our study highlights that if you can find effective interventions, the effect can be incredibly large. These are very large differences in life-years and life expectancies,” she said.
Ms. Rich gave an example of promising interventions by pointing to work by study coauthor Aima Ahonkhai, MD, MPH, assistant professor of medicine at Vanderbilt University, Nashville, Tenn., who has received federal funding to pursue research on whether preventive care outreach in barbershops can improve prevention for Black men with HIV.
Ms. Rich noted that the modeling has limitations in that it focused on health outcomes and did not simulate transmissions. Results also reflect national data and not local HIV care continuums, which, she acknowledged, differ substantially.
Dr. McManus and the study authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT IDWEEK 2022
Vaginal estrogen not recommended with aromatase inhibitors
Women with breast cancer who are taking adjuvant endocrine therapy to reduce the risk for recurrence often report that the side effects of dampening down estrogen, such as hot flashes and vaginal dryness, spoil their quality of life, and these side effects can lead to discontinuation of therapy.
But medical measures to address these side effects carry risks, as shown in the results of a new study from Denmark.
The use of vaginal estrogen therapy (VET) increased the risk for breast cancer recurrence by 39% in women with early estrogen receptor–positive breast cancer who were taking aromatase inhibitors (AIs).
There was no increase in the risk for recurrence in women who were using VET and taking tamoxifen or in women who were using VET and not taking any adjuvant endocrine therapy.
The finding was published in the Journal of the National Cancer Institute.
“Patients who are taking aromatase inhibitors should try alternative strategies for management of genitourinary symptoms because (VET) will likely increase their risk for breast cancer recurrence,” warn the authors of an accompanying editorial, Elizabeth J. Cathcart-Rake, MD, and Kathryn J. Ruddy, MD, oncologists at the Mayo Clinic, Rochester, Minn.
The use of oral estrogen treatment, known as menopausal hormone therapy (MHT), is also not recommended in breast cancer survivors being treated with AIs, the editorialists added.
The study did not find an increase in the risk for recurrence with MHT added onto AIs, but that finding comes from a very small subgroup of only 37 women.
“The absence of an obvious detrimental impact of MHT on breast cancer recurrence or mortality” in this study “is not particularly reassuring,” especially given higher systemic estrogen levels seen with MHT, Dr. Cathcart-Rake and Dr. Ruddy commented.
Differences between endocrine therapies
“Our study is, to our knowledge, the first to report a potential increased risk of recurrence in patients receiving AIs treated with VET,” say the investigators, led by Søren Cold, MD, an oncology researcher at Odense University Hospital, Denmark.
They suggest that women who are taking VET and AIs should be switched to tamoxifen after 2-3 years.
Speculating as to the apparent safety differences between the two endocrine therapies, Dr. Cold and colleagues explained that “AIs lower or nearly eliminate estrogen. As such, even a modest increase in circulating estrogens may” increase recurrence risk.
Tamoxifen, on the other hand, competes for estrogen receptor binding, so “a modest elevation of the very low serum estrogen levels” with hormone therapy “is not assumed to counteract the receptor blockade,” they said.
Study details
Study participants, obtained from a nationwide registry in Denmark, were diagnosed with early-stage, invasive, estrogen receptor–positive breast cancer from 1997 to 2004. Upfront treatment included surgery plus, in the majority of women, radiation.
The review identified 8,461 such women. After initial treatment for breast cancer, 2,410 went on to adjuvant endocrine therapy, including 2,007 with tamoxifen and 403 with an AI.
Across the entire study population, nearly 2,000 women took VET and 133 women took MHT, as assessed by having redeemed at least two prescriptions. The hormone therapies were used in women who were both on and those who were not on endocrine therapy.
Overall, breast cancer recurred in 1,333 women (16%) over a median follow-up of 9.8 years.
The investigators then analyzed the risk for recurrence in various subgroups.
The 39% higher risk for recurrence was found among the 822 women who used VET while taking an AI, compared with 2,520 women who received AIs alone.
Findings in the study were adjusted for numerous potential confounders, including age, tumor biology, and comorbidities.
Women were a median of 61 years of age (range, 35-91 years). Seventy-seven percent had invasive ductal carcinoma, and 43% were node-positive. Women on hormone therapy tended to be younger, have smaller tumors, and be less likely to have lymph node metastases.
The investigators excluded women who had taken hormone replacement before their breast cancer diagnosis.
The work was funded by the Danish Cancer Society. Dr. Cold reports no disclosures, but some co-authors reported relationships with Samsung, Novartis, Pfizer, and other companies. Dr. Cathcart-Rake and Dr. Ruddy report no disclosures.
A version of this article first appeared on Medscape.com.
Women with breast cancer who are taking adjuvant endocrine therapy to reduce the risk for recurrence often report that the side effects of dampening down estrogen, such as hot flashes and vaginal dryness, spoil their quality of life, and these side effects can lead to discontinuation of therapy.
But medical measures to address these side effects carry risks, as shown in the results of a new study from Denmark.
The use of vaginal estrogen therapy (VET) increased the risk for breast cancer recurrence by 39% in women with early estrogen receptor–positive breast cancer who were taking aromatase inhibitors (AIs).
There was no increase in the risk for recurrence in women who were using VET and taking tamoxifen or in women who were using VET and not taking any adjuvant endocrine therapy.
The finding was published in the Journal of the National Cancer Institute.
“Patients who are taking aromatase inhibitors should try alternative strategies for management of genitourinary symptoms because (VET) will likely increase their risk for breast cancer recurrence,” warn the authors of an accompanying editorial, Elizabeth J. Cathcart-Rake, MD, and Kathryn J. Ruddy, MD, oncologists at the Mayo Clinic, Rochester, Minn.
The use of oral estrogen treatment, known as menopausal hormone therapy (MHT), is also not recommended in breast cancer survivors being treated with AIs, the editorialists added.
The study did not find an increase in the risk for recurrence with MHT added onto AIs, but that finding comes from a very small subgroup of only 37 women.
“The absence of an obvious detrimental impact of MHT on breast cancer recurrence or mortality” in this study “is not particularly reassuring,” especially given higher systemic estrogen levels seen with MHT, Dr. Cathcart-Rake and Dr. Ruddy commented.
Differences between endocrine therapies
“Our study is, to our knowledge, the first to report a potential increased risk of recurrence in patients receiving AIs treated with VET,” say the investigators, led by Søren Cold, MD, an oncology researcher at Odense University Hospital, Denmark.
They suggest that women who are taking VET and AIs should be switched to tamoxifen after 2-3 years.
Speculating as to the apparent safety differences between the two endocrine therapies, Dr. Cold and colleagues explained that “AIs lower or nearly eliminate estrogen. As such, even a modest increase in circulating estrogens may” increase recurrence risk.
Tamoxifen, on the other hand, competes for estrogen receptor binding, so “a modest elevation of the very low serum estrogen levels” with hormone therapy “is not assumed to counteract the receptor blockade,” they said.
Study details
Study participants, obtained from a nationwide registry in Denmark, were diagnosed with early-stage, invasive, estrogen receptor–positive breast cancer from 1997 to 2004. Upfront treatment included surgery plus, in the majority of women, radiation.
The review identified 8,461 such women. After initial treatment for breast cancer, 2,410 went on to adjuvant endocrine therapy, including 2,007 with tamoxifen and 403 with an AI.
Across the entire study population, nearly 2,000 women took VET and 133 women took MHT, as assessed by having redeemed at least two prescriptions. The hormone therapies were used in women who were both on and those who were not on endocrine therapy.
Overall, breast cancer recurred in 1,333 women (16%) over a median follow-up of 9.8 years.
The investigators then analyzed the risk for recurrence in various subgroups.
The 39% higher risk for recurrence was found among the 822 women who used VET while taking an AI, compared with 2,520 women who received AIs alone.
Findings in the study were adjusted for numerous potential confounders, including age, tumor biology, and comorbidities.
Women were a median of 61 years of age (range, 35-91 years). Seventy-seven percent had invasive ductal carcinoma, and 43% were node-positive. Women on hormone therapy tended to be younger, have smaller tumors, and be less likely to have lymph node metastases.
The investigators excluded women who had taken hormone replacement before their breast cancer diagnosis.
The work was funded by the Danish Cancer Society. Dr. Cold reports no disclosures, but some co-authors reported relationships with Samsung, Novartis, Pfizer, and other companies. Dr. Cathcart-Rake and Dr. Ruddy report no disclosures.
A version of this article first appeared on Medscape.com.
Women with breast cancer who are taking adjuvant endocrine therapy to reduce the risk for recurrence often report that the side effects of dampening down estrogen, such as hot flashes and vaginal dryness, spoil their quality of life, and these side effects can lead to discontinuation of therapy.
But medical measures to address these side effects carry risks, as shown in the results of a new study from Denmark.
The use of vaginal estrogen therapy (VET) increased the risk for breast cancer recurrence by 39% in women with early estrogen receptor–positive breast cancer who were taking aromatase inhibitors (AIs).
There was no increase in the risk for recurrence in women who were using VET and taking tamoxifen or in women who were using VET and not taking any adjuvant endocrine therapy.
The finding was published in the Journal of the National Cancer Institute.
“Patients who are taking aromatase inhibitors should try alternative strategies for management of genitourinary symptoms because (VET) will likely increase their risk for breast cancer recurrence,” warn the authors of an accompanying editorial, Elizabeth J. Cathcart-Rake, MD, and Kathryn J. Ruddy, MD, oncologists at the Mayo Clinic, Rochester, Minn.
The use of oral estrogen treatment, known as menopausal hormone therapy (MHT), is also not recommended in breast cancer survivors being treated with AIs, the editorialists added.
The study did not find an increase in the risk for recurrence with MHT added onto AIs, but that finding comes from a very small subgroup of only 37 women.
“The absence of an obvious detrimental impact of MHT on breast cancer recurrence or mortality” in this study “is not particularly reassuring,” especially given higher systemic estrogen levels seen with MHT, Dr. Cathcart-Rake and Dr. Ruddy commented.
Differences between endocrine therapies
“Our study is, to our knowledge, the first to report a potential increased risk of recurrence in patients receiving AIs treated with VET,” say the investigators, led by Søren Cold, MD, an oncology researcher at Odense University Hospital, Denmark.
They suggest that women who are taking VET and AIs should be switched to tamoxifen after 2-3 years.
Speculating as to the apparent safety differences between the two endocrine therapies, Dr. Cold and colleagues explained that “AIs lower or nearly eliminate estrogen. As such, even a modest increase in circulating estrogens may” increase recurrence risk.
Tamoxifen, on the other hand, competes for estrogen receptor binding, so “a modest elevation of the very low serum estrogen levels” with hormone therapy “is not assumed to counteract the receptor blockade,” they said.
Study details
Study participants, obtained from a nationwide registry in Denmark, were diagnosed with early-stage, invasive, estrogen receptor–positive breast cancer from 1997 to 2004. Upfront treatment included surgery plus, in the majority of women, radiation.
The review identified 8,461 such women. After initial treatment for breast cancer, 2,410 went on to adjuvant endocrine therapy, including 2,007 with tamoxifen and 403 with an AI.
Across the entire study population, nearly 2,000 women took VET and 133 women took MHT, as assessed by having redeemed at least two prescriptions. The hormone therapies were used in women who were both on and those who were not on endocrine therapy.
Overall, breast cancer recurred in 1,333 women (16%) over a median follow-up of 9.8 years.
The investigators then analyzed the risk for recurrence in various subgroups.
The 39% higher risk for recurrence was found among the 822 women who used VET while taking an AI, compared with 2,520 women who received AIs alone.
Findings in the study were adjusted for numerous potential confounders, including age, tumor biology, and comorbidities.
Women were a median of 61 years of age (range, 35-91 years). Seventy-seven percent had invasive ductal carcinoma, and 43% were node-positive. Women on hormone therapy tended to be younger, have smaller tumors, and be less likely to have lymph node metastases.
The investigators excluded women who had taken hormone replacement before their breast cancer diagnosis.
The work was funded by the Danish Cancer Society. Dr. Cold reports no disclosures, but some co-authors reported relationships with Samsung, Novartis, Pfizer, and other companies. Dr. Cathcart-Rake and Dr. Ruddy report no disclosures.
A version of this article first appeared on Medscape.com.

