User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
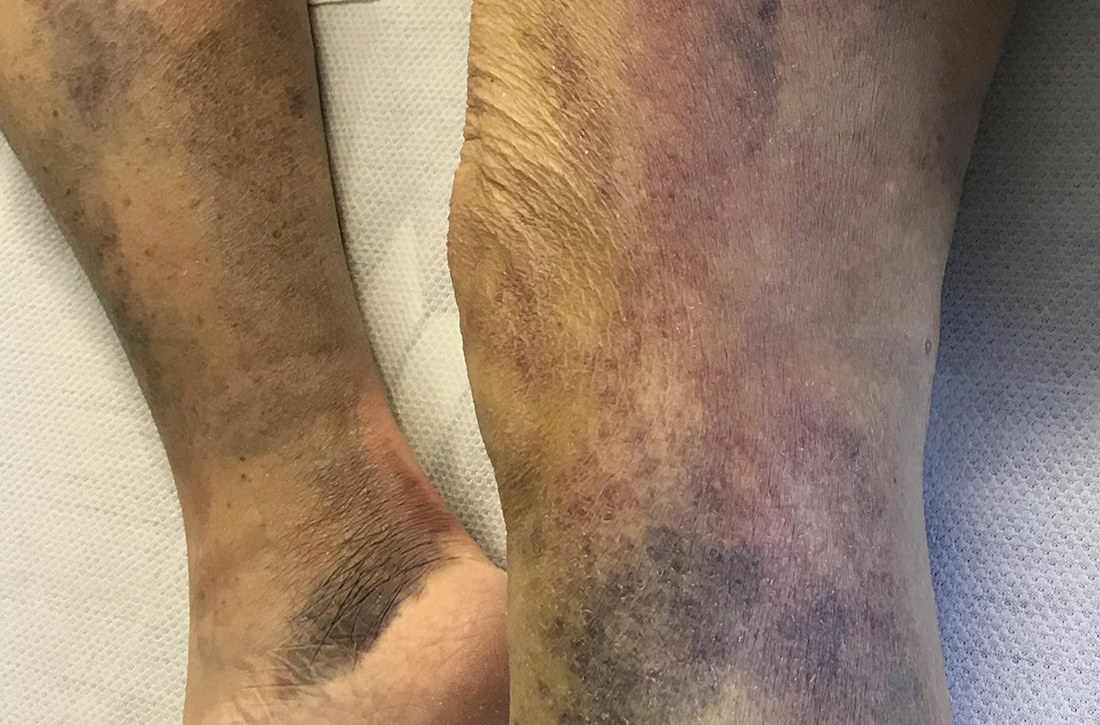
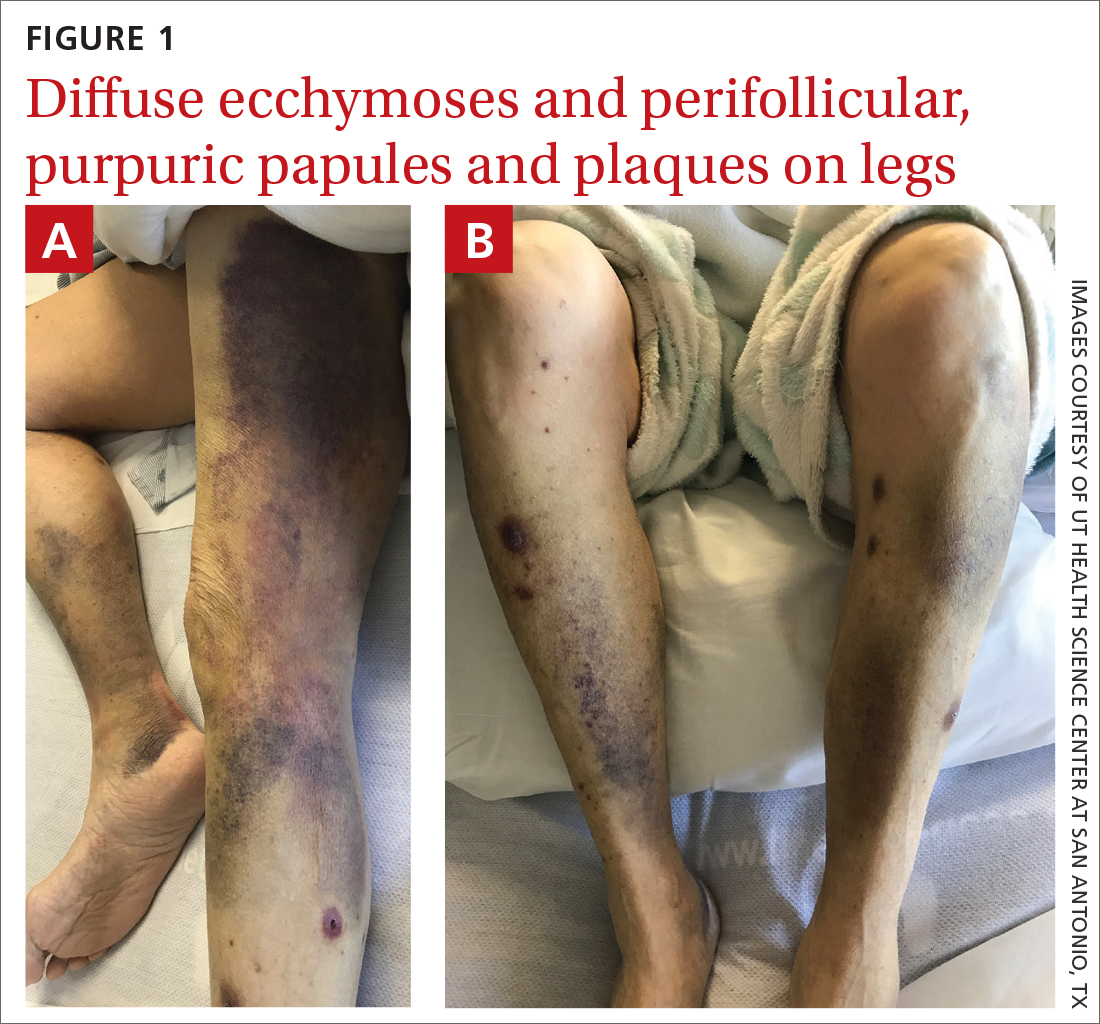
Spontaneous ecchymoses
A 65-YEAR-OLD WOMAN was brought into the emergency department by her daughter for spontaneous bruising, fatigue, and weakness of several weeks’ duration. She denied taking any medications or illicit drugs and had not experienced any falls or trauma. On a daily basis, she smoked 5 to 7 cigarettes and drank 6 or 7 beers, as had been her custom for several years. The patient lived alone and was grieving the death of her beloved dog, who had died a month earlier. She reported that since the death of her dog, her diet, which hadn’t been especially good to begin with, had deteriorated; it now consisted of beer and crackers.
On admission, she was mildly tachycardic (105 beats/min) with a blood pressure of 125/66 mm Hg. Physical examination revealed a frail-appearing woman who was in no acute distress but was unable to stand without assistance. She had diffuse ecchymoses and perifollicular, purpuric, hyperkeratotic papules and plaques on both of her legs (FIGURES 1A and 1B). In addition, she had faint perifollicular purpuric macules on her upper back. An oral examination revealed poor dentition.
A punch biopsy was performed on her leg, and it revealed noninflammatory dermal hemorrhage without evidence of vasculitis or vasculopathy.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Scurvy
Based on the patient’s appearance and her dietary history, we suspected scurvy, so a serum vitamin C level was ordered. The results took several days to return. In the meantime, additional lab work revealed hyponatremia (sodium, 129 mmol/L; normal range, 135-145 mmol/L), hypokalemia (potassium, 3 mmol/L; normal range, 3.5-5.2 mmol/L), hypophosphatemia (phosphorus, 2.3 mg/dL; normal range, 2.8-4.5 mg/dL); low serum vitamin D (6 ng/mL; normal range, 20-40 ng/mL); and macrocytic anemia (hemoglobin, 7.4 g/dL; normal range, 11-18 g/dL) with a mean corpuscular volume of 101.1 fL (normal range, 80-100 fL). Her iron panel showed normal serum iron and total iron binding capacity with a normal ferritin level (294 ng/mL; normal range, 30-300 ng/mL). A peripheral blood smear test uncovered mild anisocytosis and polychromasia, with no schistocytes. A fecal immunochemical test was negative.
Several days after admission, the results of the patient’s vitamin C test came back. Her levels were undetectable (< 5 µmol/L; normal range, 11-23 µmol/L), confirming that the patient had scurvy.
A health hazard to marinersthat is still around today
Scurvy is a condition that arises from a deficiency of vitamin C, or ascorbic acid. The first known case of scurvy was in 1550 BC.1 Hippocrates termed the condition “ileos ematitis” and stated that “the mouth feels bad; the gums are detached from the teeth; blood runs from the nostrils … ulcerations on the legs … skin is thin.”1 Scurvy was a major health hazard of mariners between the 15thand 18th centuries.2 Today, the deficiency is uncommon in industrialized countries because there are many sources of vitamin C available through diet and vitamin supplements.3 In the United States, the prevalence of vitamin C deficiency is approximately 7%.4
An essential nutrient in humans, vitamin C is required as a cofactor in the synthesis of mature collagen.3 Collagen is found in skin, bone, and endothelium. Inadequate collagen levels can result in poor dermal support of vessels and tissue fragility, leading to hemorrhage, which can occur in nearly any organ system.
Vitamin C deficiency occurs when serum concentration falls below 11.4
Continue to: Scurvy manifests after 8 to 12 weeks
Scurvy manifests after 8 to 12 weeks of inadequate vitamin C intake.1 Patients may initially experience malaise and irritability. Anemia is common. Dermatologic findings include hyperkeratotic lesions, ecchymoses, poor wound healing, gingival swelling with loss of teeth, petechiae, and corkscrew hairs. Perifollicular hemorrhage is a characteristic finding of scurvy, generally seen on the lower extremities, where the capillaries are under higher hydrostatic pressure.3 Patients may also have musculoskeletal involvement with osteopenia or hemarthroses, which may be seen on imaging.3,5 Cardiorespiratory, gastrointestinal, ophthalmologic, and neurologic findings have also been reported.3
Differential is broad; zero in on patient’s history
The differential diagnosis for hemorrhagic skin lesions is extensive and includes scurvy, coagulopathies, trauma, vasculitis, and vasculopathies.
The presence of perifollicular hemorrhage with corkscrew hairs and a dietary history of inadequate vitamin C intake can differentiate scurvy from other conditions. Serum testing revealing low plasma vitamin C will support the diagnosis, but this is an insensitive test, as values increase with recent intake. Leukocyte ascorbic acid concentrations are more representative of total body stores, but impractical for routine use.6 Skin biopsy is not necessary but may help to rule out other conditions.
Ascorbic acid will facilitate a speedy recovery
Treatment of scurvy includes vitamin C replacement. Response is rapid, with improvement to lethargy within several days and disappearance of other manifestations within several weeks.3 Recommendations on supplementation doses and forms vary, but adults require 300 to 1000 mg/d of ascorbic acid for at least 1 week or until clinical symptoms resolve and stores are repleted.3,5,7
During our patient’s hospital stay, she remained stable and improved clinically with vitamin supplementation (ascorbic acid 1 g/d for 3 days, 500 mg/d after that) and physical therapy. She was counseled on a healthy diet, which would include citrus fruits, tomatoes, and leafy vegetables. The patient was also advised to refrain from drinking alcohol and was given information on an alcohol abstinence program.
At her 1-month follow-up, her condition had improved with near resolution of the skin lesions. She reported that she had given up cigarettes and alcohol. She said she’d also begun eating more citrus fruits and leafy vegetables.
1. Maxfield L, Crane JS. Vitamin C deficiency (scurvy). In: StatPearls. StatPearls Publishing; 2020. Accessed on September 13, 2022. www.ncbi.nlm.nih.gov/books/NBK493187/
2. Worral S. A nightmare disease haunted ships during age of discovery. National Geographic. January 15, 2017. Accessed September 21, 2022. www.nationalgeographic.com/science/article/scurvy-disease-discovery-jonathan-lamb
3. Hirschmann JV, Raugi GJ. Adult Scurvy. J Am Acad Dermatol. 1999;41:895-906. doi: 10.1016/s0190-9622(99)70244-6
4. Schleicher RL, Carroll MD, Ford ES, et al. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003-2004 National Health and Nutrition Examination Survey (NHANES). Am J Clin Nutr. 2009;90:1252-1263. doi: 10.3945/ajcn.2008.27016
5. Agarwal A, Shaharyar A, Kumar A, et al. Scurvy in pediatric age group – A disease often forgotten? J Clin Orthop Trauma. 2015;6:101-107. doi: 10.1016/j.jcot.2014.12.003
6. Scurvy and its prevention and control in major emergencies. World Health Organization. February 23, 1999. Accessed September 13, 2022. www.who.int/publications/i/item/WHO-NHD-99.11
7. Weinstein M, Babyn P, Zlotkin S. An orange a day keeps the doctor away: scurvy in the year 2000. Pediatrics. 2001;108:E55. doi: 10.1542/peds.108.3.e55
A 65-YEAR-OLD WOMAN was brought into the emergency department by her daughter for spontaneous bruising, fatigue, and weakness of several weeks’ duration. She denied taking any medications or illicit drugs and had not experienced any falls or trauma. On a daily basis, she smoked 5 to 7 cigarettes and drank 6 or 7 beers, as had been her custom for several years. The patient lived alone and was grieving the death of her beloved dog, who had died a month earlier. She reported that since the death of her dog, her diet, which hadn’t been especially good to begin with, had deteriorated; it now consisted of beer and crackers.
On admission, she was mildly tachycardic (105 beats/min) with a blood pressure of 125/66 mm Hg. Physical examination revealed a frail-appearing woman who was in no acute distress but was unable to stand without assistance. She had diffuse ecchymoses and perifollicular, purpuric, hyperkeratotic papules and plaques on both of her legs (FIGURES 1A and 1B). In addition, she had faint perifollicular purpuric macules on her upper back. An oral examination revealed poor dentition.

A punch biopsy was performed on her leg, and it revealed noninflammatory dermal hemorrhage without evidence of vasculitis or vasculopathy.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Scurvy
Based on the patient’s appearance and her dietary history, we suspected scurvy, so a serum vitamin C level was ordered. The results took several days to return. In the meantime, additional lab work revealed hyponatremia (sodium, 129 mmol/L; normal range, 135-145 mmol/L), hypokalemia (potassium, 3 mmol/L; normal range, 3.5-5.2 mmol/L), hypophosphatemia (phosphorus, 2.3 mg/dL; normal range, 2.8-4.5 mg/dL); low serum vitamin D (6 ng/mL; normal range, 20-40 ng/mL); and macrocytic anemia (hemoglobin, 7.4 g/dL; normal range, 11-18 g/dL) with a mean corpuscular volume of 101.1 fL (normal range, 80-100 fL). Her iron panel showed normal serum iron and total iron binding capacity with a normal ferritin level (294 ng/mL; normal range, 30-300 ng/mL). A peripheral blood smear test uncovered mild anisocytosis and polychromasia, with no schistocytes. A fecal immunochemical test was negative.
Several days after admission, the results of the patient’s vitamin C test came back. Her levels were undetectable (< 5 µmol/L; normal range, 11-23 µmol/L), confirming that the patient had scurvy.
A health hazard to marinersthat is still around today
Scurvy is a condition that arises from a deficiency of vitamin C, or ascorbic acid. The first known case of scurvy was in 1550 BC.1 Hippocrates termed the condition “ileos ematitis” and stated that “the mouth feels bad; the gums are detached from the teeth; blood runs from the nostrils … ulcerations on the legs … skin is thin.”1 Scurvy was a major health hazard of mariners between the 15thand 18th centuries.2 Today, the deficiency is uncommon in industrialized countries because there are many sources of vitamin C available through diet and vitamin supplements.3 In the United States, the prevalence of vitamin C deficiency is approximately 7%.4
An essential nutrient in humans, vitamin C is required as a cofactor in the synthesis of mature collagen.3 Collagen is found in skin, bone, and endothelium. Inadequate collagen levels can result in poor dermal support of vessels and tissue fragility, leading to hemorrhage, which can occur in nearly any organ system.
Vitamin C deficiency occurs when serum concentration falls below 11.4
Continue to: Scurvy manifests after 8 to 12 weeks
Scurvy manifests after 8 to 12 weeks of inadequate vitamin C intake.1 Patients may initially experience malaise and irritability. Anemia is common. Dermatologic findings include hyperkeratotic lesions, ecchymoses, poor wound healing, gingival swelling with loss of teeth, petechiae, and corkscrew hairs. Perifollicular hemorrhage is a characteristic finding of scurvy, generally seen on the lower extremities, where the capillaries are under higher hydrostatic pressure.3 Patients may also have musculoskeletal involvement with osteopenia or hemarthroses, which may be seen on imaging.3,5 Cardiorespiratory, gastrointestinal, ophthalmologic, and neurologic findings have also been reported.3
Differential is broad; zero in on patient’s history
The differential diagnosis for hemorrhagic skin lesions is extensive and includes scurvy, coagulopathies, trauma, vasculitis, and vasculopathies.
The presence of perifollicular hemorrhage with corkscrew hairs and a dietary history of inadequate vitamin C intake can differentiate scurvy from other conditions. Serum testing revealing low plasma vitamin C will support the diagnosis, but this is an insensitive test, as values increase with recent intake. Leukocyte ascorbic acid concentrations are more representative of total body stores, but impractical for routine use.6 Skin biopsy is not necessary but may help to rule out other conditions.
Ascorbic acid will facilitate a speedy recovery
Treatment of scurvy includes vitamin C replacement. Response is rapid, with improvement to lethargy within several days and disappearance of other manifestations within several weeks.3 Recommendations on supplementation doses and forms vary, but adults require 300 to 1000 mg/d of ascorbic acid for at least 1 week or until clinical symptoms resolve and stores are repleted.3,5,7
During our patient’s hospital stay, she remained stable and improved clinically with vitamin supplementation (ascorbic acid 1 g/d for 3 days, 500 mg/d after that) and physical therapy. She was counseled on a healthy diet, which would include citrus fruits, tomatoes, and leafy vegetables. The patient was also advised to refrain from drinking alcohol and was given information on an alcohol abstinence program.
At her 1-month follow-up, her condition had improved with near resolution of the skin lesions. She reported that she had given up cigarettes and alcohol. She said she’d also begun eating more citrus fruits and leafy vegetables.
A 65-YEAR-OLD WOMAN was brought into the emergency department by her daughter for spontaneous bruising, fatigue, and weakness of several weeks’ duration. She denied taking any medications or illicit drugs and had not experienced any falls or trauma. On a daily basis, she smoked 5 to 7 cigarettes and drank 6 or 7 beers, as had been her custom for several years. The patient lived alone and was grieving the death of her beloved dog, who had died a month earlier. She reported that since the death of her dog, her diet, which hadn’t been especially good to begin with, had deteriorated; it now consisted of beer and crackers.
On admission, she was mildly tachycardic (105 beats/min) with a blood pressure of 125/66 mm Hg. Physical examination revealed a frail-appearing woman who was in no acute distress but was unable to stand without assistance. She had diffuse ecchymoses and perifollicular, purpuric, hyperkeratotic papules and plaques on both of her legs (FIGURES 1A and 1B). In addition, she had faint perifollicular purpuric macules on her upper back. An oral examination revealed poor dentition.

A punch biopsy was performed on her leg, and it revealed noninflammatory dermal hemorrhage without evidence of vasculitis or vasculopathy.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Scurvy
Based on the patient’s appearance and her dietary history, we suspected scurvy, so a serum vitamin C level was ordered. The results took several days to return. In the meantime, additional lab work revealed hyponatremia (sodium, 129 mmol/L; normal range, 135-145 mmol/L), hypokalemia (potassium, 3 mmol/L; normal range, 3.5-5.2 mmol/L), hypophosphatemia (phosphorus, 2.3 mg/dL; normal range, 2.8-4.5 mg/dL); low serum vitamin D (6 ng/mL; normal range, 20-40 ng/mL); and macrocytic anemia (hemoglobin, 7.4 g/dL; normal range, 11-18 g/dL) with a mean corpuscular volume of 101.1 fL (normal range, 80-100 fL). Her iron panel showed normal serum iron and total iron binding capacity with a normal ferritin level (294 ng/mL; normal range, 30-300 ng/mL). A peripheral blood smear test uncovered mild anisocytosis and polychromasia, with no schistocytes. A fecal immunochemical test was negative.
Several days after admission, the results of the patient’s vitamin C test came back. Her levels were undetectable (< 5 µmol/L; normal range, 11-23 µmol/L), confirming that the patient had scurvy.
A health hazard to marinersthat is still around today
Scurvy is a condition that arises from a deficiency of vitamin C, or ascorbic acid. The first known case of scurvy was in 1550 BC.1 Hippocrates termed the condition “ileos ematitis” and stated that “the mouth feels bad; the gums are detached from the teeth; blood runs from the nostrils … ulcerations on the legs … skin is thin.”1 Scurvy was a major health hazard of mariners between the 15thand 18th centuries.2 Today, the deficiency is uncommon in industrialized countries because there are many sources of vitamin C available through diet and vitamin supplements.3 In the United States, the prevalence of vitamin C deficiency is approximately 7%.4
An essential nutrient in humans, vitamin C is required as a cofactor in the synthesis of mature collagen.3 Collagen is found in skin, bone, and endothelium. Inadequate collagen levels can result in poor dermal support of vessels and tissue fragility, leading to hemorrhage, which can occur in nearly any organ system.
Vitamin C deficiency occurs when serum concentration falls below 11.4
Continue to: Scurvy manifests after 8 to 12 weeks
Scurvy manifests after 8 to 12 weeks of inadequate vitamin C intake.1 Patients may initially experience malaise and irritability. Anemia is common. Dermatologic findings include hyperkeratotic lesions, ecchymoses, poor wound healing, gingival swelling with loss of teeth, petechiae, and corkscrew hairs. Perifollicular hemorrhage is a characteristic finding of scurvy, generally seen on the lower extremities, where the capillaries are under higher hydrostatic pressure.3 Patients may also have musculoskeletal involvement with osteopenia or hemarthroses, which may be seen on imaging.3,5 Cardiorespiratory, gastrointestinal, ophthalmologic, and neurologic findings have also been reported.3
Differential is broad; zero in on patient’s history
The differential diagnosis for hemorrhagic skin lesions is extensive and includes scurvy, coagulopathies, trauma, vasculitis, and vasculopathies.
The presence of perifollicular hemorrhage with corkscrew hairs and a dietary history of inadequate vitamin C intake can differentiate scurvy from other conditions. Serum testing revealing low plasma vitamin C will support the diagnosis, but this is an insensitive test, as values increase with recent intake. Leukocyte ascorbic acid concentrations are more representative of total body stores, but impractical for routine use.6 Skin biopsy is not necessary but may help to rule out other conditions.
Ascorbic acid will facilitate a speedy recovery
Treatment of scurvy includes vitamin C replacement. Response is rapid, with improvement to lethargy within several days and disappearance of other manifestations within several weeks.3 Recommendations on supplementation doses and forms vary, but adults require 300 to 1000 mg/d of ascorbic acid for at least 1 week or until clinical symptoms resolve and stores are repleted.3,5,7
During our patient’s hospital stay, she remained stable and improved clinically with vitamin supplementation (ascorbic acid 1 g/d for 3 days, 500 mg/d after that) and physical therapy. She was counseled on a healthy diet, which would include citrus fruits, tomatoes, and leafy vegetables. The patient was also advised to refrain from drinking alcohol and was given information on an alcohol abstinence program.
At her 1-month follow-up, her condition had improved with near resolution of the skin lesions. She reported that she had given up cigarettes and alcohol. She said she’d also begun eating more citrus fruits and leafy vegetables.
1. Maxfield L, Crane JS. Vitamin C deficiency (scurvy). In: StatPearls. StatPearls Publishing; 2020. Accessed on September 13, 2022. www.ncbi.nlm.nih.gov/books/NBK493187/
2. Worral S. A nightmare disease haunted ships during age of discovery. National Geographic. January 15, 2017. Accessed September 21, 2022. www.nationalgeographic.com/science/article/scurvy-disease-discovery-jonathan-lamb
3. Hirschmann JV, Raugi GJ. Adult Scurvy. J Am Acad Dermatol. 1999;41:895-906. doi: 10.1016/s0190-9622(99)70244-6
4. Schleicher RL, Carroll MD, Ford ES, et al. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003-2004 National Health and Nutrition Examination Survey (NHANES). Am J Clin Nutr. 2009;90:1252-1263. doi: 10.3945/ajcn.2008.27016
5. Agarwal A, Shaharyar A, Kumar A, et al. Scurvy in pediatric age group – A disease often forgotten? J Clin Orthop Trauma. 2015;6:101-107. doi: 10.1016/j.jcot.2014.12.003
6. Scurvy and its prevention and control in major emergencies. World Health Organization. February 23, 1999. Accessed September 13, 2022. www.who.int/publications/i/item/WHO-NHD-99.11
7. Weinstein M, Babyn P, Zlotkin S. An orange a day keeps the doctor away: scurvy in the year 2000. Pediatrics. 2001;108:E55. doi: 10.1542/peds.108.3.e55
1. Maxfield L, Crane JS. Vitamin C deficiency (scurvy). In: StatPearls. StatPearls Publishing; 2020. Accessed on September 13, 2022. www.ncbi.nlm.nih.gov/books/NBK493187/
2. Worral S. A nightmare disease haunted ships during age of discovery. National Geographic. January 15, 2017. Accessed September 21, 2022. www.nationalgeographic.com/science/article/scurvy-disease-discovery-jonathan-lamb
3. Hirschmann JV, Raugi GJ. Adult Scurvy. J Am Acad Dermatol. 1999;41:895-906. doi: 10.1016/s0190-9622(99)70244-6
4. Schleicher RL, Carroll MD, Ford ES, et al. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003-2004 National Health and Nutrition Examination Survey (NHANES). Am J Clin Nutr. 2009;90:1252-1263. doi: 10.3945/ajcn.2008.27016
5. Agarwal A, Shaharyar A, Kumar A, et al. Scurvy in pediatric age group – A disease often forgotten? J Clin Orthop Trauma. 2015;6:101-107. doi: 10.1016/j.jcot.2014.12.003
6. Scurvy and its prevention and control in major emergencies. World Health Organization. February 23, 1999. Accessed September 13, 2022. www.who.int/publications/i/item/WHO-NHD-99.11
7. Weinstein M, Babyn P, Zlotkin S. An orange a day keeps the doctor away: scurvy in the year 2000. Pediatrics. 2001;108:E55. doi: 10.1542/peds.108.3.e55
Check biases when caring for children with obesity
Counting calories should not be the focus of weight-loss strategies for children with obesity, according to an expert who said pediatricians need to change the way they discuss weight with their patients.
During a plenary session of the American Academy of Pediatrics National Conference, Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, N.C., said pediatricians should recognize the behavioral, physical, environmental, and genetic factors that contribute to obesity. For instance, food deserts are on the rise, and they undermine the ability of parents to feed their children healthy meals. In addition, more children are less physically active.
“Obesity is a lot more complex than calories in, calories out,” Dr. Skelton said. “We choose to treat issues of obesity as personal responsibility – ‘you did this to yourself’ – but when you look at how we move around and live our lives, our food systems, our policies, the social and environmental changes have caused shifts in our behavior.”
According to Dr. Skelton, bias against children with obesity can harm their self-image and weaken their motivations for losing weight. In addition, doctors may change how they deliver care on the basis of stereotypes regarding obese children. These stereotypes are often reinforced in media portrayals, Dr. Skelton said.
“When children or when adults who have excess weight or obesity are portrayed, they are portrayed typically in a negative fashion,” Dr. Skelton said. “There’s increasing evidence that weight bias and weight discrimination are increasing the morbidity we see in patients who develop obesity.”
For many children with obesity, visits to the pediatrician often center on weight, regardless of the reason for the appointment. Weight stigma and bias on the part of health care providers can increase stress, as well as adverse health outcomes in children, according to a 2019 study (Curr Opin Endocrinol Diabetes Obes. 2019 Feb 1. doi: 10.1097/MED.0000000000000453). Dr. Skelton recommended that pediatricians listen to their patients’ concerns and make a personalized care plan.
Dr. Skelton said doctors can pull from projects such as Health at Every Size, which offers templates for personalized health plans for children with obesity. It has a heavy focus on a weight-neutral approach to pediatric health.
“There are various ways to manage weight in a healthy and safe way,” Dr. Skelton said.
Evidence-based methods of treating obesity include focusing on health and healthy behaviors rather than weight and using the body mass index as a screening tool for further conversations about overall health, rather than as an indicator of health based on weight.
Dr. Skelton also encouraged pediatricians to be on the alert for indicators of disordered eating, which can include dieting, teasing, or talking excessively about weight at home and can involve reading misinformation about dieting online.
“Your job is to educate people on the dangers of following unscientific information online,” Dr. Skelton said. “We can address issues of weight health in a way that is patient centered and is very safe, without unintended consequences.” Brooke Sweeney, MD, professor of internal medicine and pediatrics at University of Missouri–Kansas City, said problems with weight bias in society and in clinical practice can lead to false assumptions about people who have obesity.
“It’s normal to gain adipose, or fat tissue, at different times in life, during puberty or pregnancy, and some people normally gain more weight than others,” Dr. Sweeney said.
The body will try to maintain a weight set point. That set point is influenced by many factors, such as genetics, environment, and lifestyle.
“When you lose weight, your body tries to get you back to the set point, decreasing energy expenditure and increasing hunger and reward pathways,” she said. “We have gained so much knowledge through research to better understand the pathophysiology of obesity, and we are making good progress on improving advanced treatments for increased weight in children.”
Dr. Skelton reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Counting calories should not be the focus of weight-loss strategies for children with obesity, according to an expert who said pediatricians need to change the way they discuss weight with their patients.
During a plenary session of the American Academy of Pediatrics National Conference, Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, N.C., said pediatricians should recognize the behavioral, physical, environmental, and genetic factors that contribute to obesity. For instance, food deserts are on the rise, and they undermine the ability of parents to feed their children healthy meals. In addition, more children are less physically active.
“Obesity is a lot more complex than calories in, calories out,” Dr. Skelton said. “We choose to treat issues of obesity as personal responsibility – ‘you did this to yourself’ – but when you look at how we move around and live our lives, our food systems, our policies, the social and environmental changes have caused shifts in our behavior.”
According to Dr. Skelton, bias against children with obesity can harm their self-image and weaken their motivations for losing weight. In addition, doctors may change how they deliver care on the basis of stereotypes regarding obese children. These stereotypes are often reinforced in media portrayals, Dr. Skelton said.
“When children or when adults who have excess weight or obesity are portrayed, they are portrayed typically in a negative fashion,” Dr. Skelton said. “There’s increasing evidence that weight bias and weight discrimination are increasing the morbidity we see in patients who develop obesity.”
For many children with obesity, visits to the pediatrician often center on weight, regardless of the reason for the appointment. Weight stigma and bias on the part of health care providers can increase stress, as well as adverse health outcomes in children, according to a 2019 study (Curr Opin Endocrinol Diabetes Obes. 2019 Feb 1. doi: 10.1097/MED.0000000000000453). Dr. Skelton recommended that pediatricians listen to their patients’ concerns and make a personalized care plan.
Dr. Skelton said doctors can pull from projects such as Health at Every Size, which offers templates for personalized health plans for children with obesity. It has a heavy focus on a weight-neutral approach to pediatric health.
“There are various ways to manage weight in a healthy and safe way,” Dr. Skelton said.
Evidence-based methods of treating obesity include focusing on health and healthy behaviors rather than weight and using the body mass index as a screening tool for further conversations about overall health, rather than as an indicator of health based on weight.
Dr. Skelton also encouraged pediatricians to be on the alert for indicators of disordered eating, which can include dieting, teasing, or talking excessively about weight at home and can involve reading misinformation about dieting online.
“Your job is to educate people on the dangers of following unscientific information online,” Dr. Skelton said. “We can address issues of weight health in a way that is patient centered and is very safe, without unintended consequences.” Brooke Sweeney, MD, professor of internal medicine and pediatrics at University of Missouri–Kansas City, said problems with weight bias in society and in clinical practice can lead to false assumptions about people who have obesity.
“It’s normal to gain adipose, or fat tissue, at different times in life, during puberty or pregnancy, and some people normally gain more weight than others,” Dr. Sweeney said.
The body will try to maintain a weight set point. That set point is influenced by many factors, such as genetics, environment, and lifestyle.
“When you lose weight, your body tries to get you back to the set point, decreasing energy expenditure and increasing hunger and reward pathways,” she said. “We have gained so much knowledge through research to better understand the pathophysiology of obesity, and we are making good progress on improving advanced treatments for increased weight in children.”
Dr. Skelton reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Counting calories should not be the focus of weight-loss strategies for children with obesity, according to an expert who said pediatricians need to change the way they discuss weight with their patients.
During a plenary session of the American Academy of Pediatrics National Conference, Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, N.C., said pediatricians should recognize the behavioral, physical, environmental, and genetic factors that contribute to obesity. For instance, food deserts are on the rise, and they undermine the ability of parents to feed their children healthy meals. In addition, more children are less physically active.
“Obesity is a lot more complex than calories in, calories out,” Dr. Skelton said. “We choose to treat issues of obesity as personal responsibility – ‘you did this to yourself’ – but when you look at how we move around and live our lives, our food systems, our policies, the social and environmental changes have caused shifts in our behavior.”
According to Dr. Skelton, bias against children with obesity can harm their self-image and weaken their motivations for losing weight. In addition, doctors may change how they deliver care on the basis of stereotypes regarding obese children. These stereotypes are often reinforced in media portrayals, Dr. Skelton said.
“When children or when adults who have excess weight or obesity are portrayed, they are portrayed typically in a negative fashion,” Dr. Skelton said. “There’s increasing evidence that weight bias and weight discrimination are increasing the morbidity we see in patients who develop obesity.”
For many children with obesity, visits to the pediatrician often center on weight, regardless of the reason for the appointment. Weight stigma and bias on the part of health care providers can increase stress, as well as adverse health outcomes in children, according to a 2019 study (Curr Opin Endocrinol Diabetes Obes. 2019 Feb 1. doi: 10.1097/MED.0000000000000453). Dr. Skelton recommended that pediatricians listen to their patients’ concerns and make a personalized care plan.
Dr. Skelton said doctors can pull from projects such as Health at Every Size, which offers templates for personalized health plans for children with obesity. It has a heavy focus on a weight-neutral approach to pediatric health.
“There are various ways to manage weight in a healthy and safe way,” Dr. Skelton said.
Evidence-based methods of treating obesity include focusing on health and healthy behaviors rather than weight and using the body mass index as a screening tool for further conversations about overall health, rather than as an indicator of health based on weight.
Dr. Skelton also encouraged pediatricians to be on the alert for indicators of disordered eating, which can include dieting, teasing, or talking excessively about weight at home and can involve reading misinformation about dieting online.
“Your job is to educate people on the dangers of following unscientific information online,” Dr. Skelton said. “We can address issues of weight health in a way that is patient centered and is very safe, without unintended consequences.” Brooke Sweeney, MD, professor of internal medicine and pediatrics at University of Missouri–Kansas City, said problems with weight bias in society and in clinical practice can lead to false assumptions about people who have obesity.
“It’s normal to gain adipose, or fat tissue, at different times in life, during puberty or pregnancy, and some people normally gain more weight than others,” Dr. Sweeney said.
The body will try to maintain a weight set point. That set point is influenced by many factors, such as genetics, environment, and lifestyle.
“When you lose weight, your body tries to get you back to the set point, decreasing energy expenditure and increasing hunger and reward pathways,” she said. “We have gained so much knowledge through research to better understand the pathophysiology of obesity, and we are making good progress on improving advanced treatments for increased weight in children.”
Dr. Skelton reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AAP 2022
Congenital syphilis: It’s still a significant public health problem
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
You’re rounding in the nursery and informed of the following about one of your new patients: He’s a 38-week-old infant delivered to a mother diagnosed with syphilis at 12 weeks’ gestation at her initial prenatal visit. Her rapid plasma reagin (RPR) was 1:64 and the fluorescent treponemal antibody–absorption (FTA-ABS) test was positive. By report she was appropriately treated. Maternal RPRs obtained at 18 and 28 weeks’ gestation were 1:16 and 1:4, respectively. Maternal RPR at delivery and the infant’s RPR obtained shortly after birth were both 1:4. The mother wants to know if her baby is infected.
One result of syphilis during pregnancy is intrauterine infection and resultant congenital disease in the infant. Before you answer this mother, let’s discuss syphilis.
Congenital syphilis is a significant public health problem. In 2021, there were a total of 2,677 cases reported for a rate of 74.1 per 100,000 live births. Between 2020 and 2021, the number of cases of congenital syphilis increased 24.1% (2,158-2,677 cases), concurrent with a 45.8% increase (10.7-15.6 per 100,000) in the rate of primary and secondary syphilis in women aged 15-44 years. Between 2012 and 2021, the number of cases of congenital syphilis increased 701.5% (334-2,677 cases) and the increase in rates of primary and secondary syphilis in women aged 15-44 was 642.9% over the same period.
Why are the rates of congenital syphilis increasing? Most cases result from a lack of prenatal care and thus no testing for syphilis. The next most common cause is inadequate maternal treatment.
Congenital syphilis usually is acquired through transplacental transmission of spirochetes in the maternal bloodstream. Occasionally, it occurs at delivery via direct contact with maternal lesions. It is not transmitted in breast milk. Transmission of syphilis:
- Can occur any time during pregnancy.
- Is more likely to occur in women with untreated primary or secondary disease (60%-100%).
- Is approximately 40% in those with early latent syphilis and less than 8% in mothers with late latent syphilis.
- Is higher in women coinfected with HIV since they more frequently receive no prenatal care and their disease is inadequately treated.
Coinfection with syphilis may also increase the rate of mother-to-child transmission of HIV.
Untreated early syphilis during pregnancy results in spontaneous abortion, stillbirth, or perinatal death in up to 40% of cases. Infected newborns with early congenital syphilis can be asymptomatic or have evidence of hepatosplenomegaly, generalized lymphadenopathy, nasal discharge that is occasionally bloody, rash, and skeletal abnormalities (osteochondritis and periostitis). Other manifestations include edema, hemolytic anemia, jaundice, pneumonia, pseudoparalysis, and thrombocytopenia. Asymptomatic infants may have abnormal cerebrospinal fluid findings including elevated CSF white cell count, elevated protein, and a reactive venereal disease research laboratory test.
Late congenital syphilis, defined as the onset of symptoms after 2 years of age is secondary to scarring or persistent inflammation and gumma formation in a variety of tissues. It occurs in up to 40% of cases of untreated maternal disease. Most cases can be prevented by maternal treatment and treatment of the infant within the first 3 months of life. Common clinical manifestations include interstitial keratitis, sensorineural hearing loss, frontal bossing, saddle nose, Hutchinson teeth, mulberry molars, perforation of the hard palate, anterior bowing of the tibia (saber shins), and other skeletal abnormalities.
Diagnostic tests. Maternal diagnosis is dependent upon knowing the results of both a nontreponemal (RPR, VDRL) and a confirmatory treponemal test (TP-PA, TP-EIA, TP-CIA, FTA-ABS,) before or at delivery. TP-PA is the preferred test. When maternal disease is confirmed, the newborn should have the same quantitative nontreponemal test as the mother. A confirmatory treponemal test is not required
Evaluation and treatment. It’s imperative that children born to mothers with a reactive test, regardless of their treatment status, have a thorough exam performed before hospital discharge. The provider must determine what additional interventions should be performed.
The American Academy of Pediatrics and the Centers for Disease Control and Prevention (www.cdc.gov/std/treatment-guidelines/congenital-syphilis.htm) have developed standard algorithms for the diagnostic approach and treatment of infants born to mothers with reactive serologic tests for syphilis. It is available in the Red Book for AAP members (https://publications.aap.org/redbook). Recommendations based on various scenarios for neonates up to 1 month of age include proven or highly probable congenital syphilis, possible congenital syphilis, congenital syphilis less likely, and congenital syphilis unlikely. It is beyond the scope of this article to list the criteria and evaluation for each scenario. The reader is referred to the algorithm.
If syphilis is suspected in infants or children older than 1 month, the challenge is to determine if it is untreated congenital syphilis or acquired syphilis. Maternal syphilis status should be determined. Evaluation for congenital syphilis in this age group includes CSF analysis for VDRL, cell count and protein, CBC with differential and platelets, hepatic panel, abdominal ultrasound, long-bone radiographs, chest radiograph, neuroimaging, auditory brain stem response, and HIV testing.
Let’s go back to your patient. The mother was diagnosed with syphilis during pregnancy. You confirm that she was treated with benzathine penicillin G, and the course was completed at least 4 weeks before delivery. Treatment with any other drug during pregnancy is not appropriate. The RPR has declined, and the infant’s titer is equal to or less than four times the maternal titer. The exam is significant for generalized adenopathy and slightly bloody nasal discharge. This infant has two findings consistent with congenital syphilis regardless of RPR titer or treatment status. This places him in the proven or highly probable congenital syphilis group. Management includes CSF analysis (VDRL, cell count, and protein), CBC with differential and platelet count, and treatment with penicillin G for 10 days. Additional tests as clinically indicated include: long-bone radiograph, chest radiography, aspartate aminotranferase and alanine aminotransferase levels, neuroimaging, ophthalmologic exam, and auditory brain stem response. Despite maternal treatment, this newborn has congenital syphilis. The same nontreponemal test should be obtained every 2-3 months until it is nonreactive. It should be nonreactive by 6 months. If the infection persists to 6-12 months post treatment, reevaluation including CSF analysis and retreatment may be indicated.
Congenital syphilis can be prevented by maternal screening, diagnosis, and treatment. When that fails it is up to us to diagnosis and adequately treat our patients.
Dr. Word is a pediatric infectious disease specialist and director of the Houston Travel Medicine Clinic. She said she had no relevant financial disclosures. Email her at [email protected].
63% of long COVID patients are women, study says
according to a new study published in JAMA.
The global study also found that about 6% of people with symptomatic infections had long COVID in 2020 and 2021. The risk for long COVID seemed to be greater among those who needed hospitalization, especially those who needed intensive care.
“Quantifying the number of individuals with long COVID may help policy makers ensure adequate access to services to guide people toward recovery, return to the workplace or school, and restore their mental health and social life,” the researchers wrote.
The study team, which included dozens of researchers across nearly every continent, analyzed data from 54 studies and two databases for more than 1 million patients in 22 countries who had symptomatic COVID infections in 2020 and 2021. They looked at three long COVID symptom types: persistent fatigue with bodily pain or mood swings, ongoing respiratory problems, and cognitive issues. The study included people aged 4-66.
Overall, 6.2% of people reported one of the long COVID symptom types, including 3.7% with ongoing respiratory problems, 3.2% with persistent fatigue and bodily pain or mood swings, and 2.2% with cognitive problems. Among those with long COVID, 38% of people reported more than one symptom cluster.
At 3 months after infection, long COVID symptoms were nearly twice as common in women who were at least 20 years old at 10.6%, compared with men who were at least 20 years old at 5.4%.
Children and teens appeared to have lower risks of long COVID. About 2.8% of patients under age 20 with symptomatic infection developed long-term issues.
The estimated average duration of long COVID symptoms was 9 months among hospitalized patients and 4 months among those who weren’t hospitalized. About 15% of people with long COVID symptoms 3 months after the initial infection continued to have symptoms at 12 months.
The study was largely based on detailed data from ongoing COVID-19 studies in the United States, Austria, the Faroe Islands, Germany, Iran, Italy, the Netherlands, Russia, Sweden, and Switzerland, according to UPI. It was supplemented by published data and research conducted as part of the Global Burden of Diseases, Injuries and Risk Factors Study. The dozens of researchers are referred to as “Global Burden of Disease Long COVID Collaborators.”
The study had limitations, the researchers said, including the assumption that long COVID follows a similar course in all countries. Additional studies may show how long COVID symptoms and severity may vary in different countries and continents.
Ultimately, ongoing studies of large numbers of people with long COVID could help scientists and public health officials understand risk factors and ways to treat the debilitating condition, the study authors wrote, noting that “postinfection fatigue syndrome” has been reported before, namely during the 1918 flu pandemic, after the SARS outbreak in 2003, and after the Ebola epidemic in West Africa in 2014.
“Similar symptoms have been reported after other viral infections, including the Epstein-Barr virus, mononucleosis, and dengue, as well as after nonviral infections such as Q fever, Lyme disease and giardiasis,” they wrote.
Several study investigators reported receiving grants and personal fees from a variety of sources.
A version of this article first appeared on Medscape.com.
according to a new study published in JAMA.
The global study also found that about 6% of people with symptomatic infections had long COVID in 2020 and 2021. The risk for long COVID seemed to be greater among those who needed hospitalization, especially those who needed intensive care.
“Quantifying the number of individuals with long COVID may help policy makers ensure adequate access to services to guide people toward recovery, return to the workplace or school, and restore their mental health and social life,” the researchers wrote.
The study team, which included dozens of researchers across nearly every continent, analyzed data from 54 studies and two databases for more than 1 million patients in 22 countries who had symptomatic COVID infections in 2020 and 2021. They looked at three long COVID symptom types: persistent fatigue with bodily pain or mood swings, ongoing respiratory problems, and cognitive issues. The study included people aged 4-66.
Overall, 6.2% of people reported one of the long COVID symptom types, including 3.7% with ongoing respiratory problems, 3.2% with persistent fatigue and bodily pain or mood swings, and 2.2% with cognitive problems. Among those with long COVID, 38% of people reported more than one symptom cluster.
At 3 months after infection, long COVID symptoms were nearly twice as common in women who were at least 20 years old at 10.6%, compared with men who were at least 20 years old at 5.4%.
Children and teens appeared to have lower risks of long COVID. About 2.8% of patients under age 20 with symptomatic infection developed long-term issues.
The estimated average duration of long COVID symptoms was 9 months among hospitalized patients and 4 months among those who weren’t hospitalized. About 15% of people with long COVID symptoms 3 months after the initial infection continued to have symptoms at 12 months.
The study was largely based on detailed data from ongoing COVID-19 studies in the United States, Austria, the Faroe Islands, Germany, Iran, Italy, the Netherlands, Russia, Sweden, and Switzerland, according to UPI. It was supplemented by published data and research conducted as part of the Global Burden of Diseases, Injuries and Risk Factors Study. The dozens of researchers are referred to as “Global Burden of Disease Long COVID Collaborators.”
The study had limitations, the researchers said, including the assumption that long COVID follows a similar course in all countries. Additional studies may show how long COVID symptoms and severity may vary in different countries and continents.
Ultimately, ongoing studies of large numbers of people with long COVID could help scientists and public health officials understand risk factors and ways to treat the debilitating condition, the study authors wrote, noting that “postinfection fatigue syndrome” has been reported before, namely during the 1918 flu pandemic, after the SARS outbreak in 2003, and after the Ebola epidemic in West Africa in 2014.
“Similar symptoms have been reported after other viral infections, including the Epstein-Barr virus, mononucleosis, and dengue, as well as after nonviral infections such as Q fever, Lyme disease and giardiasis,” they wrote.
Several study investigators reported receiving grants and personal fees from a variety of sources.
A version of this article first appeared on Medscape.com.
according to a new study published in JAMA.
The global study also found that about 6% of people with symptomatic infections had long COVID in 2020 and 2021. The risk for long COVID seemed to be greater among those who needed hospitalization, especially those who needed intensive care.
“Quantifying the number of individuals with long COVID may help policy makers ensure adequate access to services to guide people toward recovery, return to the workplace or school, and restore their mental health and social life,” the researchers wrote.
The study team, which included dozens of researchers across nearly every continent, analyzed data from 54 studies and two databases for more than 1 million patients in 22 countries who had symptomatic COVID infections in 2020 and 2021. They looked at three long COVID symptom types: persistent fatigue with bodily pain or mood swings, ongoing respiratory problems, and cognitive issues. The study included people aged 4-66.
Overall, 6.2% of people reported one of the long COVID symptom types, including 3.7% with ongoing respiratory problems, 3.2% with persistent fatigue and bodily pain or mood swings, and 2.2% with cognitive problems. Among those with long COVID, 38% of people reported more than one symptom cluster.
At 3 months after infection, long COVID symptoms were nearly twice as common in women who were at least 20 years old at 10.6%, compared with men who were at least 20 years old at 5.4%.
Children and teens appeared to have lower risks of long COVID. About 2.8% of patients under age 20 with symptomatic infection developed long-term issues.
The estimated average duration of long COVID symptoms was 9 months among hospitalized patients and 4 months among those who weren’t hospitalized. About 15% of people with long COVID symptoms 3 months after the initial infection continued to have symptoms at 12 months.
The study was largely based on detailed data from ongoing COVID-19 studies in the United States, Austria, the Faroe Islands, Germany, Iran, Italy, the Netherlands, Russia, Sweden, and Switzerland, according to UPI. It was supplemented by published data and research conducted as part of the Global Burden of Diseases, Injuries and Risk Factors Study. The dozens of researchers are referred to as “Global Burden of Disease Long COVID Collaborators.”
The study had limitations, the researchers said, including the assumption that long COVID follows a similar course in all countries. Additional studies may show how long COVID symptoms and severity may vary in different countries and continents.
Ultimately, ongoing studies of large numbers of people with long COVID could help scientists and public health officials understand risk factors and ways to treat the debilitating condition, the study authors wrote, noting that “postinfection fatigue syndrome” has been reported before, namely during the 1918 flu pandemic, after the SARS outbreak in 2003, and after the Ebola epidemic in West Africa in 2014.
“Similar symptoms have been reported after other viral infections, including the Epstein-Barr virus, mononucleosis, and dengue, as well as after nonviral infections such as Q fever, Lyme disease and giardiasis,” they wrote.
Several study investigators reported receiving grants and personal fees from a variety of sources.
A version of this article first appeared on Medscape.com.
FROM JAMA
Why people lie about COVID
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
Have you ever lied about COVID-19?
Before you get upset, before the “how dare you,” I want you to think carefully.
Did you have COVID-19 (or think you did) and not mention it to someone you were going to be with? Did you tell someone you were taking more COVID precautions than you really were? Did you tell someone you were vaccinated when you weren’t? Have you avoided getting a COVID test even though you knew you should have?
Researchers appreciated the fact that public health interventions in COVID are important but are only as good as the percentage of people who actually abide by them. So, they designed a survey to ask the questions that many people don’t want to hear the answer to.
A total of 1,733 participants – 80% of those invited – responded to the survey. By design, approximately one-third of respondents (477) had already had COVID, one-third (499) were vaccinated and not yet infected, and one-third (509) were unvaccinated and not yet infected.
Of those surveyed, 41.6% admitted that they lied about COVID or didn’t adhere to COVID guidelines - a conservative estimate, if you ask me.
Breaking down some of the results, about 20% of people who previously were infected with COVID said they didn’t mention it when meeting with someone. A similar number said they didn’t tell anyone when they were entering a public place. A bit more concerning to me, roughly 20% reported not disclosing their COVID-positive status when going to a health care provider’s office.
About 10% of those who had not been vaccinated reported lying about their vaccination status. That’s actually less than the 15% of vaccinated people who lied and told someone they weren’t vaccinated.
About 17% of people lied about the need to quarantine, and many more broke quarantine rules.
The authors tried to see if certain personal characteristics predicted people who were more likely to lie about COVID-19–related issues. Turns out there was only one thing that predicted honesty: age.
Older people were more honest about their COVID status and COVID habits. Other factors – gender, education, race, political affiliation, COVID-19 conspiracy beliefs, and where you got your COVID information – did not seem to make much of a difference. Why are older people more honest? Because older people take COVID more seriously. And they should; COVID is more severe in older people.
The problem arises, of course, because people who are at lower risk for COVID complications interact with people at higher risk – and in those situations, honesty matters more.
On the other hand, isn’t lying about COVID stuff inevitable? If you know that a positive test means you can’t go to work, and not going to work means you won’t get paid, might you not be more likely to lie about the test? Or not get the test at all?
The authors explored the reasons for dishonesty and they are fairly broad, ranging from the desire for life to feel normal (more than half of people who lied) to not believing that COVID was real (a whopping 30%). Some of the reasons for lying included:
- Wanted life to feel normal (50%).
- Freedom (45%).
- It’s no one’s business (40%).
- COVID isn’t real (30%).
In the end, though, we need to realize that public health recommendations are not going to be universally followed, and people may tell us they are following them when, in fact, they are not.
What this adds is another data point to a trend we’ve seen across the course of the pandemic, a shift from collective to individual responsibility. If you can’t be sure what others are doing in regard to COVID, you need to focus on protecting yourself. Perhaps that shift was inevitable. Doesn’t mean we have to like it.
A version of this article first appeared on Medscape.com.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here on Medscape. He tweets @fperrywilson and hosts a repository of his communication work at www.methodsman.com.
Older diabetes drugs linked to dementia risk -- one lower, one higher
a new observational study in patients with type 2 diabetes suggests.
The data, obtained from nationwide electronic medical records from the Department of Veterans Affairs, yielded a 22% lower risk of dementia with TZD monotherapy and a 12% elevated risk with sulfonylurea monotherapy, compared with metformin monotherapy. The apparent protective effects of TZDs were greater among individuals with overweight or obesity.
“Our findings provide additional information to aid clinicians’ selection of [glucose-lowering medications] for patients with mild or moderate type 2 diabetes and [who] are at high risk of dementia,” Xin Tang and colleagues wrote in their article, published online in BMJ Open Diabetes Research & Care.
The results “add substantially to the literature concerning the effects of [glucose-lowering medications] on dementia where previous findings have been inconsistent. Studies with a follow-up time of less than 3 years have mainly reported null associations, while studies with longer a follow-up time typically yielded protective findings. With a mean follow-up time of 6.8 years, we had a sufficient duration to detect treatment differences,” the investigators wrote.
“Supplementing [a] sulfonylurea with either metformin or [a] TZD may partially offset its prodementia effects. These findings may help inform medication selection for elderly patients with T2D at high risk of dementia,” they added.
Randomized trials needed to determine cause and effect
Ivan Koychev, PhD, a senior clinical researcher in the department of psychiatry at the University of Oxford (England), told the UK Science Media Centre: “This is a large, well-conducted real-world data study that highlights the importance of checking whether already prescribed medications may be useful for preventing dementia.”
The findings regarding TZDs, also known as glitazones, are in line with existing literature suggesting dementia protection with other drugs prescribed for type 2 diabetes that weren’t examined in the current study, such as newer agents like glucagonlike peptide–1 (GLP-1) agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors, Dr. Koychev said.
“The main limitations of this study is that following the initial 2-year period the authors were interested in, the participants may have been prescribed one of the other type 2 diabetes drugs [GLP-1 agonists or SGLT2 inhibitors] that have been found to reduce dementia risk, thus potentially making the direct glitazone [TZD] effect more difficult to discern,” Dr. Koychev noted.
And, he pointed out that the study design limits attribution of causality. “It is also important to note that people with type 2 diabetes do run a higher risk of both dementia and cognitive deficits and that these medications are only prescribed in these patients, so all this data is from this patient group rather than the general population.”
James Connell, PhD, head of translational science at Alzheimer’s Research UK, agreed. “While this observational study found that those with type 2 diabetes taking thiazolidinedione had a lower dementia risk than those on the most common medication for type 2 diabetes, it only shows an association between taking the drug and dementia risk and not a causal relationship.
“Double-blind and placebo-controlled clinical trials are needed to see whether the drug [TDZ] could help lower dementia risk in people with and without diabetes. Anyone with any questions about what treatments they are receiving should speak to their doctor,” he told the UK Science Media Centre.
Opposite effects of sulfonylureas, TZDs versus metformin
The study authors analyzed 559,106 VA patients with type 2 diabetes who initiated glucose-lowering medication during 2001-2017 and took it for at least a year. They were aged 60 years or older and did not have dementia at baseline. Most were White (76.8%) and male (96.9%), two-thirds (63.1%) had obesity, and mean hemoglobin A1c was 6.8%.
Overall, 31,125 developed all-cause dementia. The incidence rate was 8.2 cases per 1,000 person-years, ranging from 6.2 cases per 1,000 person-years among those taking metformin monotherapy to 13.4 cases per 1,000 person-years in those taking both sulfonylurea and a TZD.
Compared with metformin monotherapy, the hazard ratio for all-cause dementia for sulfonylurea monotherapy was a significant 1.12. The increased risk was also seen for vascular dementia, with an HR of 1.14.
In contrast, TZD monotherapy was associated with a significantly lower risk for all-cause dementia (HR, 0.78), as well as for Alzheimer’s disease (HR, 0.89) and vascular dementia (HR, 0.43), compared with metformin monotherapy.
The combination of metformin and TZD also lowered the risk of all-cause dementia, while regimens including sulfonylureas raised the risks for all-cause and vascular dementia.
Most of the results didn’t change significantly when the drug exposure window was extended to 2 years.
Effects more pronounced in those with obesity
The protective 1-year effects of TZD monotherapy and of metformin plus TZD, compared with metformin alone, were more significant among participants aged 75 or younger and with a body mass index above 25 kg/m2, compared with those who were older than 75 years and with normal BMIs, respectively.
On the other hand, the greater risk for dementia incurred with sulfonylureas was further increased among those with higher BMI.
This research was partially funded by grants from the National Human Genome Research Institute, the National Science Foundation, the National Institute of Diabetes and Digestive and Kidney Disease, and the National Heart, Lung, and Blood Institute. Dr. Koychev is chief investigator for a trial, sponsored by Oxford University and funded by Novo Nordisk, testing whether the GLP-1 agonist semaglutide reduces the risk for dementia in aging adults.
A version of this article first appeared on Medscape.com.
a new observational study in patients with type 2 diabetes suggests.
The data, obtained from nationwide electronic medical records from the Department of Veterans Affairs, yielded a 22% lower risk of dementia with TZD monotherapy and a 12% elevated risk with sulfonylurea monotherapy, compared with metformin monotherapy. The apparent protective effects of TZDs were greater among individuals with overweight or obesity.
“Our findings provide additional information to aid clinicians’ selection of [glucose-lowering medications] for patients with mild or moderate type 2 diabetes and [who] are at high risk of dementia,” Xin Tang and colleagues wrote in their article, published online in BMJ Open Diabetes Research & Care.
The results “add substantially to the literature concerning the effects of [glucose-lowering medications] on dementia where previous findings have been inconsistent. Studies with a follow-up time of less than 3 years have mainly reported null associations, while studies with longer a follow-up time typically yielded protective findings. With a mean follow-up time of 6.8 years, we had a sufficient duration to detect treatment differences,” the investigators wrote.
“Supplementing [a] sulfonylurea with either metformin or [a] TZD may partially offset its prodementia effects. These findings may help inform medication selection for elderly patients with T2D at high risk of dementia,” they added.
Randomized trials needed to determine cause and effect
Ivan Koychev, PhD, a senior clinical researcher in the department of psychiatry at the University of Oxford (England), told the UK Science Media Centre: “This is a large, well-conducted real-world data study that highlights the importance of checking whether already prescribed medications may be useful for preventing dementia.”
The findings regarding TZDs, also known as glitazones, are in line with existing literature suggesting dementia protection with other drugs prescribed for type 2 diabetes that weren’t examined in the current study, such as newer agents like glucagonlike peptide–1 (GLP-1) agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors, Dr. Koychev said.
“The main limitations of this study is that following the initial 2-year period the authors were interested in, the participants may have been prescribed one of the other type 2 diabetes drugs [GLP-1 agonists or SGLT2 inhibitors] that have been found to reduce dementia risk, thus potentially making the direct glitazone [TZD] effect more difficult to discern,” Dr. Koychev noted.
And, he pointed out that the study design limits attribution of causality. “It is also important to note that people with type 2 diabetes do run a higher risk of both dementia and cognitive deficits and that these medications are only prescribed in these patients, so all this data is from this patient group rather than the general population.”
James Connell, PhD, head of translational science at Alzheimer’s Research UK, agreed. “While this observational study found that those with type 2 diabetes taking thiazolidinedione had a lower dementia risk than those on the most common medication for type 2 diabetes, it only shows an association between taking the drug and dementia risk and not a causal relationship.
“Double-blind and placebo-controlled clinical trials are needed to see whether the drug [TDZ] could help lower dementia risk in people with and without diabetes. Anyone with any questions about what treatments they are receiving should speak to their doctor,” he told the UK Science Media Centre.
Opposite effects of sulfonylureas, TZDs versus metformin
The study authors analyzed 559,106 VA patients with type 2 diabetes who initiated glucose-lowering medication during 2001-2017 and took it for at least a year. They were aged 60 years or older and did not have dementia at baseline. Most were White (76.8%) and male (96.9%), two-thirds (63.1%) had obesity, and mean hemoglobin A1c was 6.8%.
Overall, 31,125 developed all-cause dementia. The incidence rate was 8.2 cases per 1,000 person-years, ranging from 6.2 cases per 1,000 person-years among those taking metformin monotherapy to 13.4 cases per 1,000 person-years in those taking both sulfonylurea and a TZD.
Compared with metformin monotherapy, the hazard ratio for all-cause dementia for sulfonylurea monotherapy was a significant 1.12. The increased risk was also seen for vascular dementia, with an HR of 1.14.
In contrast, TZD monotherapy was associated with a significantly lower risk for all-cause dementia (HR, 0.78), as well as for Alzheimer’s disease (HR, 0.89) and vascular dementia (HR, 0.43), compared with metformin monotherapy.
The combination of metformin and TZD also lowered the risk of all-cause dementia, while regimens including sulfonylureas raised the risks for all-cause and vascular dementia.
Most of the results didn’t change significantly when the drug exposure window was extended to 2 years.
Effects more pronounced in those with obesity
The protective 1-year effects of TZD monotherapy and of metformin plus TZD, compared with metformin alone, were more significant among participants aged 75 or younger and with a body mass index above 25 kg/m2, compared with those who were older than 75 years and with normal BMIs, respectively.
On the other hand, the greater risk for dementia incurred with sulfonylureas was further increased among those with higher BMI.
This research was partially funded by grants from the National Human Genome Research Institute, the National Science Foundation, the National Institute of Diabetes and Digestive and Kidney Disease, and the National Heart, Lung, and Blood Institute. Dr. Koychev is chief investigator for a trial, sponsored by Oxford University and funded by Novo Nordisk, testing whether the GLP-1 agonist semaglutide reduces the risk for dementia in aging adults.
A version of this article first appeared on Medscape.com.
a new observational study in patients with type 2 diabetes suggests.
The data, obtained from nationwide electronic medical records from the Department of Veterans Affairs, yielded a 22% lower risk of dementia with TZD monotherapy and a 12% elevated risk with sulfonylurea monotherapy, compared with metformin monotherapy. The apparent protective effects of TZDs were greater among individuals with overweight or obesity.
“Our findings provide additional information to aid clinicians’ selection of [glucose-lowering medications] for patients with mild or moderate type 2 diabetes and [who] are at high risk of dementia,” Xin Tang and colleagues wrote in their article, published online in BMJ Open Diabetes Research & Care.
The results “add substantially to the literature concerning the effects of [glucose-lowering medications] on dementia where previous findings have been inconsistent. Studies with a follow-up time of less than 3 years have mainly reported null associations, while studies with longer a follow-up time typically yielded protective findings. With a mean follow-up time of 6.8 years, we had a sufficient duration to detect treatment differences,” the investigators wrote.
“Supplementing [a] sulfonylurea with either metformin or [a] TZD may partially offset its prodementia effects. These findings may help inform medication selection for elderly patients with T2D at high risk of dementia,” they added.
Randomized trials needed to determine cause and effect
Ivan Koychev, PhD, a senior clinical researcher in the department of psychiatry at the University of Oxford (England), told the UK Science Media Centre: “This is a large, well-conducted real-world data study that highlights the importance of checking whether already prescribed medications may be useful for preventing dementia.”
The findings regarding TZDs, also known as glitazones, are in line with existing literature suggesting dementia protection with other drugs prescribed for type 2 diabetes that weren’t examined in the current study, such as newer agents like glucagonlike peptide–1 (GLP-1) agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors, Dr. Koychev said.
“The main limitations of this study is that following the initial 2-year period the authors were interested in, the participants may have been prescribed one of the other type 2 diabetes drugs [GLP-1 agonists or SGLT2 inhibitors] that have been found to reduce dementia risk, thus potentially making the direct glitazone [TZD] effect more difficult to discern,” Dr. Koychev noted.
And, he pointed out that the study design limits attribution of causality. “It is also important to note that people with type 2 diabetes do run a higher risk of both dementia and cognitive deficits and that these medications are only prescribed in these patients, so all this data is from this patient group rather than the general population.”
James Connell, PhD, head of translational science at Alzheimer’s Research UK, agreed. “While this observational study found that those with type 2 diabetes taking thiazolidinedione had a lower dementia risk than those on the most common medication for type 2 diabetes, it only shows an association between taking the drug and dementia risk and not a causal relationship.
“Double-blind and placebo-controlled clinical trials are needed to see whether the drug [TDZ] could help lower dementia risk in people with and without diabetes. Anyone with any questions about what treatments they are receiving should speak to their doctor,” he told the UK Science Media Centre.
Opposite effects of sulfonylureas, TZDs versus metformin
The study authors analyzed 559,106 VA patients with type 2 diabetes who initiated glucose-lowering medication during 2001-2017 and took it for at least a year. They were aged 60 years or older and did not have dementia at baseline. Most were White (76.8%) and male (96.9%), two-thirds (63.1%) had obesity, and mean hemoglobin A1c was 6.8%.
Overall, 31,125 developed all-cause dementia. The incidence rate was 8.2 cases per 1,000 person-years, ranging from 6.2 cases per 1,000 person-years among those taking metformin monotherapy to 13.4 cases per 1,000 person-years in those taking both sulfonylurea and a TZD.
Compared with metformin monotherapy, the hazard ratio for all-cause dementia for sulfonylurea monotherapy was a significant 1.12. The increased risk was also seen for vascular dementia, with an HR of 1.14.
In contrast, TZD monotherapy was associated with a significantly lower risk for all-cause dementia (HR, 0.78), as well as for Alzheimer’s disease (HR, 0.89) and vascular dementia (HR, 0.43), compared with metformin monotherapy.
The combination of metformin and TZD also lowered the risk of all-cause dementia, while regimens including sulfonylureas raised the risks for all-cause and vascular dementia.
Most of the results didn’t change significantly when the drug exposure window was extended to 2 years.
Effects more pronounced in those with obesity
The protective 1-year effects of TZD monotherapy and of metformin plus TZD, compared with metformin alone, were more significant among participants aged 75 or younger and with a body mass index above 25 kg/m2, compared with those who were older than 75 years and with normal BMIs, respectively.
On the other hand, the greater risk for dementia incurred with sulfonylureas was further increased among those with higher BMI.
This research was partially funded by grants from the National Human Genome Research Institute, the National Science Foundation, the National Institute of Diabetes and Digestive and Kidney Disease, and the National Heart, Lung, and Blood Institute. Dr. Koychev is chief investigator for a trial, sponsored by Oxford University and funded by Novo Nordisk, testing whether the GLP-1 agonist semaglutide reduces the risk for dementia in aging adults.
A version of this article first appeared on Medscape.com.
FROM BMJ OPEN DIABETES RESEARCH & CARE
Epidemic of brain fog? Long COVID’s effects worry experts
Weeks after Jeannie Volpe caught COVID-19 in November 2020, she could no longer do her job running sexual assault support groups in Anniston, Ala., because she kept forgetting the details that survivors had shared with her. “People were telling me they were having to revisit their traumatic memories, which isn’t fair to anybody,” the 47-year-old says.
Ms. Volpe has been diagnosed with long-COVID autonomic dysfunction, which includes severe muscle pain, depression, anxiety, and a loss of thinking skills. Some of her symptoms are more commonly known as brain fog, and they’re among the most frequent problems reported by people who have long-term issues after a bout of COVID-19.
Many experts and medical professionals say they haven’t even begun to scratch the surface of what impact this will have in years to come.
“I’m very worried that we have an epidemic of neurologic dysfunction coming down the pike,” says Pamela Davis, MD, PhD, a research professor at Case Western Reserve University, Cleveland.
In the 2 years Ms. Volpe has been living with long COVID, her executive function – the mental processes that enable people to focus attention, retain information, and multitask – has been so diminished that she had to relearn to drive. One of the various doctors assessing her has suggested speech therapy to help Ms. Volpe relearn how to form words. “I can see the words I want to say in my mind, but I can’t make them come out of my mouth,” she says in a sluggish voice that gives away her condition.
All of those symptoms make it difficult for her to care for herself. Without a job and health insurance, Ms. Volpe says she’s researched assisted suicide in the states that allow it but has ultimately decided she wants to live.
“People tell you things like you should be grateful you survived it, and you should; but you shouldn’t expect somebody to not grieve after losing their autonomy, their career, their finances.”
The findings of researchers studying the brain effects of COVID-19 reinforce what people with long COVID have been dealing with from the start. Their experiences aren’t imaginary; they’re consistent with neurological disorders – including myalgic encephalomyelitis, also known as chronic fatigue syndrome, or ME/CFS – which carry much more weight in the public imagination than the term brain fog, which can often be used dismissively.
Studies have found that COVID-19 is linked to conditions such as strokes; seizures; and mood, memory, and movement disorders.
While there are still a lot of unanswered questions about exactly how COVID-19 affects the brain and what the long-term effects are, there’s enough reason to suggest people should be trying to avoid both infection and reinfection until researchers get more answers.
Worldwide, it’s estimated that COVID-19 has contributed to more than 40 million new cases of neurological disorders, says Ziyad Al-Aly, MD, a clinical epidemiologist and long COVID researcher at Washington University in St. Louis. In his latest study of 14 million medical records of the U.S. Department of Veterans Affairs, the country’s largest integrated health care system, researchers found that regardless of age, gender, race, and lifestyle,
He noted that some of the conditions, such as headaches and mild decline in memory and sharpness, may improve and go away over time. But others that showed up, such as stroke, encephalitis (inflammation of the brain), and Guillain-Barré syndrome (a rare disorder in which the body’s immune system attacks the nerves), often lead to lasting damage. Dr. Al-Aly’s team found that neurological conditions were 7% more likely in those who had COVID-19 than in those who had never been infected.
What’s more, researchers noticed that compared with control groups, the risk of post-COVID thinking problems was more pronounced in people in their 30s, 40s, and 50s – a group that usually would be very unlikely to have these problems. For those over the age of 60, the risks stood out less because at that stage of life, such thinking problems aren’t as rare.
Another study of the veterans system last year showed that COVID-19 survivors were at a 46% higher risk of considering suicide after 1 year.
“We need to be paying attention to this,” says Dr. Al-Aly. “What we’ve seen is really the tip of the iceberg.” He worries that millions of people, including youths, will lose out on employment and education while dealing with long-term disabilities – and the economic and societal implications of such a fallout. “What we will all be left with is the aftermath of sheer devastation in some people’s lives,” he says.
Igor Koralnik, MD, chief of neuro-infectious disease and global neurology at Northwestern University, Chicago, has been running a specialized long COVID clinic. His team published a paper in March 2021 detailing what they saw in their first 100 patients. “About half the population in the study missed at least 10 days of work. This is going to have persistent impact on the workforce,” Dr. Koralnik said in a podcast posted on the Northwestern website. “We have seen that not only [do] patients have symptoms, but they have decreased quality of life.”
For older people and their caregivers, the risk of potential neurodegenerative diseases that the virus has shown to accelerate, such as dementia, is also a big concern. Alzheimer’s is already the fifth leading cause of death for people 65 and older.
In a recent study of more than 6 million people over the age of 65, Dr. Davis and her team at Case Western found the risk of Alzheimer’s in the year after COVID-19 increased by 50%-80%. The chances were especially high for women older than 85.
To date, there are no good treatments for Alzheimer’s, yet total health care costs for long-term care and hospice services for people with dementia topped $300 billion in 2020. That doesn’t even include the related costs to families.
“The downstream effect of having someone with Alzheimer’s being taken care of by a family member can be devastating on everyone,” she says. “Sometimes the caregivers don’t weather that very well.”
When Dr. Davis’s own father got Alzheimer’s at age 86, her mother took care of him until she had a stroke one morning while making breakfast. Dr. Davis attributes the stroke to the stress of caregiving. That left Dr. Davis no choice but to seek housing where both her parents could get care.
Looking at the broader picture, Dr. Davis believes widespread isolation, loneliness, and grief during the pandemic, and the disease of COVID-19 itself, will continue to have a profound impact on psychiatric diagnoses. This in turn could trigger a wave of new substance abuse as a result of unchecked mental health problems.
Still, not all brain experts are jumping to worst-case scenarios, with a lot yet to be understood before sounding the alarm. Joanna Hellmuth, MD, a neurologist and researcher at the University of California, San Francisco, cautions against reading too much into early data, including any assumptions that COVID-19 causes neurodegeneration or irreversible damage in the brain.
Even with before-and-after brain scans by University of Oxford, England, researchers that show structural changes to the brain after infection, she points out that they didn’t actually study the clinical symptoms of the people in the study, so it’s too soon to reach conclusions about associated cognitive problems.
“It’s an important piece of the puzzle, but we don’t know how that fits together with everything else,” says Dr. Hellmuth. “Some of my patients get better. … I haven’t seen a single person get worse since the pandemic started, and so I’m hopeful.”
A version of this article first appeared on WebMD.com.
Weeks after Jeannie Volpe caught COVID-19 in November 2020, she could no longer do her job running sexual assault support groups in Anniston, Ala., because she kept forgetting the details that survivors had shared with her. “People were telling me they were having to revisit their traumatic memories, which isn’t fair to anybody,” the 47-year-old says.
Ms. Volpe has been diagnosed with long-COVID autonomic dysfunction, which includes severe muscle pain, depression, anxiety, and a loss of thinking skills. Some of her symptoms are more commonly known as brain fog, and they’re among the most frequent problems reported by people who have long-term issues after a bout of COVID-19.
Many experts and medical professionals say they haven’t even begun to scratch the surface of what impact this will have in years to come.
“I’m very worried that we have an epidemic of neurologic dysfunction coming down the pike,” says Pamela Davis, MD, PhD, a research professor at Case Western Reserve University, Cleveland.
In the 2 years Ms. Volpe has been living with long COVID, her executive function – the mental processes that enable people to focus attention, retain information, and multitask – has been so diminished that she had to relearn to drive. One of the various doctors assessing her has suggested speech therapy to help Ms. Volpe relearn how to form words. “I can see the words I want to say in my mind, but I can’t make them come out of my mouth,” she says in a sluggish voice that gives away her condition.
All of those symptoms make it difficult for her to care for herself. Without a job and health insurance, Ms. Volpe says she’s researched assisted suicide in the states that allow it but has ultimately decided she wants to live.
“People tell you things like you should be grateful you survived it, and you should; but you shouldn’t expect somebody to not grieve after losing their autonomy, their career, their finances.”
The findings of researchers studying the brain effects of COVID-19 reinforce what people with long COVID have been dealing with from the start. Their experiences aren’t imaginary; they’re consistent with neurological disorders – including myalgic encephalomyelitis, also known as chronic fatigue syndrome, or ME/CFS – which carry much more weight in the public imagination than the term brain fog, which can often be used dismissively.
Studies have found that COVID-19 is linked to conditions such as strokes; seizures; and mood, memory, and movement disorders.
While there are still a lot of unanswered questions about exactly how COVID-19 affects the brain and what the long-term effects are, there’s enough reason to suggest people should be trying to avoid both infection and reinfection until researchers get more answers.
Worldwide, it’s estimated that COVID-19 has contributed to more than 40 million new cases of neurological disorders, says Ziyad Al-Aly, MD, a clinical epidemiologist and long COVID researcher at Washington University in St. Louis. In his latest study of 14 million medical records of the U.S. Department of Veterans Affairs, the country’s largest integrated health care system, researchers found that regardless of age, gender, race, and lifestyle,
He noted that some of the conditions, such as headaches and mild decline in memory and sharpness, may improve and go away over time. But others that showed up, such as stroke, encephalitis (inflammation of the brain), and Guillain-Barré syndrome (a rare disorder in which the body’s immune system attacks the nerves), often lead to lasting damage. Dr. Al-Aly’s team found that neurological conditions were 7% more likely in those who had COVID-19 than in those who had never been infected.
What’s more, researchers noticed that compared with control groups, the risk of post-COVID thinking problems was more pronounced in people in their 30s, 40s, and 50s – a group that usually would be very unlikely to have these problems. For those over the age of 60, the risks stood out less because at that stage of life, such thinking problems aren’t as rare.
Another study of the veterans system last year showed that COVID-19 survivors were at a 46% higher risk of considering suicide after 1 year.
“We need to be paying attention to this,” says Dr. Al-Aly. “What we’ve seen is really the tip of the iceberg.” He worries that millions of people, including youths, will lose out on employment and education while dealing with long-term disabilities – and the economic and societal implications of such a fallout. “What we will all be left with is the aftermath of sheer devastation in some people’s lives,” he says.
Igor Koralnik, MD, chief of neuro-infectious disease and global neurology at Northwestern University, Chicago, has been running a specialized long COVID clinic. His team published a paper in March 2021 detailing what they saw in their first 100 patients. “About half the population in the study missed at least 10 days of work. This is going to have persistent impact on the workforce,” Dr. Koralnik said in a podcast posted on the Northwestern website. “We have seen that not only [do] patients have symptoms, but they have decreased quality of life.”
For older people and their caregivers, the risk of potential neurodegenerative diseases that the virus has shown to accelerate, such as dementia, is also a big concern. Alzheimer’s is already the fifth leading cause of death for people 65 and older.
In a recent study of more than 6 million people over the age of 65, Dr. Davis and her team at Case Western found the risk of Alzheimer’s in the year after COVID-19 increased by 50%-80%. The chances were especially high for women older than 85.
To date, there are no good treatments for Alzheimer’s, yet total health care costs for long-term care and hospice services for people with dementia topped $300 billion in 2020. That doesn’t even include the related costs to families.
“The downstream effect of having someone with Alzheimer’s being taken care of by a family member can be devastating on everyone,” she says. “Sometimes the caregivers don’t weather that very well.”
When Dr. Davis’s own father got Alzheimer’s at age 86, her mother took care of him until she had a stroke one morning while making breakfast. Dr. Davis attributes the stroke to the stress of caregiving. That left Dr. Davis no choice but to seek housing where both her parents could get care.
Looking at the broader picture, Dr. Davis believes widespread isolation, loneliness, and grief during the pandemic, and the disease of COVID-19 itself, will continue to have a profound impact on psychiatric diagnoses. This in turn could trigger a wave of new substance abuse as a result of unchecked mental health problems.
Still, not all brain experts are jumping to worst-case scenarios, with a lot yet to be understood before sounding the alarm. Joanna Hellmuth, MD, a neurologist and researcher at the University of California, San Francisco, cautions against reading too much into early data, including any assumptions that COVID-19 causes neurodegeneration or irreversible damage in the brain.
Even with before-and-after brain scans by University of Oxford, England, researchers that show structural changes to the brain after infection, she points out that they didn’t actually study the clinical symptoms of the people in the study, so it’s too soon to reach conclusions about associated cognitive problems.
“It’s an important piece of the puzzle, but we don’t know how that fits together with everything else,” says Dr. Hellmuth. “Some of my patients get better. … I haven’t seen a single person get worse since the pandemic started, and so I’m hopeful.”
A version of this article first appeared on WebMD.com.
Weeks after Jeannie Volpe caught COVID-19 in November 2020, she could no longer do her job running sexual assault support groups in Anniston, Ala., because she kept forgetting the details that survivors had shared with her. “People were telling me they were having to revisit their traumatic memories, which isn’t fair to anybody,” the 47-year-old says.
Ms. Volpe has been diagnosed with long-COVID autonomic dysfunction, which includes severe muscle pain, depression, anxiety, and a loss of thinking skills. Some of her symptoms are more commonly known as brain fog, and they’re among the most frequent problems reported by people who have long-term issues after a bout of COVID-19.
Many experts and medical professionals say they haven’t even begun to scratch the surface of what impact this will have in years to come.
“I’m very worried that we have an epidemic of neurologic dysfunction coming down the pike,” says Pamela Davis, MD, PhD, a research professor at Case Western Reserve University, Cleveland.
In the 2 years Ms. Volpe has been living with long COVID, her executive function – the mental processes that enable people to focus attention, retain information, and multitask – has been so diminished that she had to relearn to drive. One of the various doctors assessing her has suggested speech therapy to help Ms. Volpe relearn how to form words. “I can see the words I want to say in my mind, but I can’t make them come out of my mouth,” she says in a sluggish voice that gives away her condition.
All of those symptoms make it difficult for her to care for herself. Without a job and health insurance, Ms. Volpe says she’s researched assisted suicide in the states that allow it but has ultimately decided she wants to live.
“People tell you things like you should be grateful you survived it, and you should; but you shouldn’t expect somebody to not grieve after losing their autonomy, their career, their finances.”
The findings of researchers studying the brain effects of COVID-19 reinforce what people with long COVID have been dealing with from the start. Their experiences aren’t imaginary; they’re consistent with neurological disorders – including myalgic encephalomyelitis, also known as chronic fatigue syndrome, or ME/CFS – which carry much more weight in the public imagination than the term brain fog, which can often be used dismissively.
Studies have found that COVID-19 is linked to conditions such as strokes; seizures; and mood, memory, and movement disorders.
While there are still a lot of unanswered questions about exactly how COVID-19 affects the brain and what the long-term effects are, there’s enough reason to suggest people should be trying to avoid both infection and reinfection until researchers get more answers.
Worldwide, it’s estimated that COVID-19 has contributed to more than 40 million new cases of neurological disorders, says Ziyad Al-Aly, MD, a clinical epidemiologist and long COVID researcher at Washington University in St. Louis. In his latest study of 14 million medical records of the U.S. Department of Veterans Affairs, the country’s largest integrated health care system, researchers found that regardless of age, gender, race, and lifestyle,
He noted that some of the conditions, such as headaches and mild decline in memory and sharpness, may improve and go away over time. But others that showed up, such as stroke, encephalitis (inflammation of the brain), and Guillain-Barré syndrome (a rare disorder in which the body’s immune system attacks the nerves), often lead to lasting damage. Dr. Al-Aly’s team found that neurological conditions were 7% more likely in those who had COVID-19 than in those who had never been infected.
What’s more, researchers noticed that compared with control groups, the risk of post-COVID thinking problems was more pronounced in people in their 30s, 40s, and 50s – a group that usually would be very unlikely to have these problems. For those over the age of 60, the risks stood out less because at that stage of life, such thinking problems aren’t as rare.
Another study of the veterans system last year showed that COVID-19 survivors were at a 46% higher risk of considering suicide after 1 year.
“We need to be paying attention to this,” says Dr. Al-Aly. “What we’ve seen is really the tip of the iceberg.” He worries that millions of people, including youths, will lose out on employment and education while dealing with long-term disabilities – and the economic and societal implications of such a fallout. “What we will all be left with is the aftermath of sheer devastation in some people’s lives,” he says.
Igor Koralnik, MD, chief of neuro-infectious disease and global neurology at Northwestern University, Chicago, has been running a specialized long COVID clinic. His team published a paper in March 2021 detailing what they saw in their first 100 patients. “About half the population in the study missed at least 10 days of work. This is going to have persistent impact on the workforce,” Dr. Koralnik said in a podcast posted on the Northwestern website. “We have seen that not only [do] patients have symptoms, but they have decreased quality of life.”
For older people and their caregivers, the risk of potential neurodegenerative diseases that the virus has shown to accelerate, such as dementia, is also a big concern. Alzheimer’s is already the fifth leading cause of death for people 65 and older.
In a recent study of more than 6 million people over the age of 65, Dr. Davis and her team at Case Western found the risk of Alzheimer’s in the year after COVID-19 increased by 50%-80%. The chances were especially high for women older than 85.
To date, there are no good treatments for Alzheimer’s, yet total health care costs for long-term care and hospice services for people with dementia topped $300 billion in 2020. That doesn’t even include the related costs to families.
“The downstream effect of having someone with Alzheimer’s being taken care of by a family member can be devastating on everyone,” she says. “Sometimes the caregivers don’t weather that very well.”
When Dr. Davis’s own father got Alzheimer’s at age 86, her mother took care of him until she had a stroke one morning while making breakfast. Dr. Davis attributes the stroke to the stress of caregiving. That left Dr. Davis no choice but to seek housing where both her parents could get care.
Looking at the broader picture, Dr. Davis believes widespread isolation, loneliness, and grief during the pandemic, and the disease of COVID-19 itself, will continue to have a profound impact on psychiatric diagnoses. This in turn could trigger a wave of new substance abuse as a result of unchecked mental health problems.
Still, not all brain experts are jumping to worst-case scenarios, with a lot yet to be understood before sounding the alarm. Joanna Hellmuth, MD, a neurologist and researcher at the University of California, San Francisco, cautions against reading too much into early data, including any assumptions that COVID-19 causes neurodegeneration or irreversible damage in the brain.
Even with before-and-after brain scans by University of Oxford, England, researchers that show structural changes to the brain after infection, she points out that they didn’t actually study the clinical symptoms of the people in the study, so it’s too soon to reach conclusions about associated cognitive problems.
“It’s an important piece of the puzzle, but we don’t know how that fits together with everything else,” says Dr. Hellmuth. “Some of my patients get better. … I haven’t seen a single person get worse since the pandemic started, and so I’m hopeful.”
A version of this article first appeared on WebMD.com.
ACC issues guidance on ED evaluation of acute chest pain
Chest pain accounts for more than 7 million ED visits annually. A major challenge is to quickly identify the small number of patients with acute coronary syndrome (ACS) among the large number of patients who have noncardiac conditions.
The new document is intended to provide guidance on how to “practically apply” recommendations from the 2021 American Heart Association/American College of Cardiology Guideline for the Evaluation and Diagnosis of Chest Pain, focusing specifically on patients who present to the ED, the writing group explains.
“A systematic approach – both at the level of the institution and the individual patient – is essential to achieve optimal outcomes for patients presenting with chest pain to the ED,” say writing group chair Michael Kontos, MD, Virginia Commonwealth University, Richmond, and colleagues.
At the institution level, this decision pathway recommends high-sensitivity cardiac troponin (hs-cTn) assays coupled with a clinical decision pathway (CDP) to reduce ED “dwell” times and increase the number of patients with chest pain who can safely be discharged without additional testing. This will decrease ED crowding and limit unnecessary testing, they point out.
At the individual patient level, this document aims to provide structure for the ED evaluation of chest pain, accelerating the evaluation process and matching the intensity of testing and treatment to patient risk.
The 36-page document was published online in the Journal of the American College of Cardiology.
Key summary points in the document include the following:
- Electrocardiogram remains the best initial test for evaluation of chest pain in the ED and should be performed and interpreted within 10 minutes of ED arrival.
- In patients who arrive via ambulance, the prehospital ECG should be reviewed, because ischemic changes may have resolved before ED arrival.
- When the ECG shows evidence of acute infarction or ischemia, subsequent care should follow current guidelines for management of acute ST-segment elevation myocardial infarction (STEMI) and non–ST-segment elevation ACS (NSTE-ACS).
- Patients with a nonischemic ECG can enter an accelerated CDP designed to provide rapid risk assessment and exclusion of ACS.
- Patients who are hemodynamically unstable, have significant arrhythmias, or evidence of significant heart failure should be evaluated and treated appropriately and are not candidates for an accelerated CDP.
- High-sensitivity cardiac troponin T (hs-cTnT) and high-sensitivity cardiac troponin I (hs-cTnI) are the preferred biomarkers for evaluation of possible ACS.
- Patients classified as low risk (rule out) using the current hs-cTn-based CDPs can generally be discharged directly from the ED without additional testing, although outpatient testing may be considered in selected cases.
- Patients with substantially elevated initial hs-cTn values or those with significant dynamic changes over 1-3 hours are assigned to the abnormal/high-risk category and should be further classified according to the universal definition of myocardial infarction type 1 or 2 or acute or chronic nonischemic cardiac injury.
- High-risk patients should usually be admitted to an inpatient setting for further evaluation and treatment.
- Patients determined to be intermediate risk with the CDP should undergo additional observation with repeat hs-cTn measurements at 3-6 hours and risk assessment using either the modified HEART (history, ECG, age, risk factors, and troponin) score or the ED assessment of chest pain score (EDACS).
- Noninvasive testing should be considered for the intermediate-risk group unless low-risk features are identified using risk scores or noninvasive testing has been performed recently with normal or low-risk findings.
The writing group notes that “safe and efficient” management of chest pain in the ED requires appropriate follow-up after discharge. Timing of follow-up and referral for outpatient noninvasive testing should be influenced by patient risk and results of cardiac testing.
Disclosures for members of the writing group are available with the original article.
A version of this article first appeared on Medscape.com.
Chest pain accounts for more than 7 million ED visits annually. A major challenge is to quickly identify the small number of patients with acute coronary syndrome (ACS) among the large number of patients who have noncardiac conditions.
The new document is intended to provide guidance on how to “practically apply” recommendations from the 2021 American Heart Association/American College of Cardiology Guideline for the Evaluation and Diagnosis of Chest Pain, focusing specifically on patients who present to the ED, the writing group explains.
“A systematic approach – both at the level of the institution and the individual patient – is essential to achieve optimal outcomes for patients presenting with chest pain to the ED,” say writing group chair Michael Kontos, MD, Virginia Commonwealth University, Richmond, and colleagues.
At the institution level, this decision pathway recommends high-sensitivity cardiac troponin (hs-cTn) assays coupled with a clinical decision pathway (CDP) to reduce ED “dwell” times and increase the number of patients with chest pain who can safely be discharged without additional testing. This will decrease ED crowding and limit unnecessary testing, they point out.
At the individual patient level, this document aims to provide structure for the ED evaluation of chest pain, accelerating the evaluation process and matching the intensity of testing and treatment to patient risk.
The 36-page document was published online in the Journal of the American College of Cardiology.
Key summary points in the document include the following:
- Electrocardiogram remains the best initial test for evaluation of chest pain in the ED and should be performed and interpreted within 10 minutes of ED arrival.
- In patients who arrive via ambulance, the prehospital ECG should be reviewed, because ischemic changes may have resolved before ED arrival.
- When the ECG shows evidence of acute infarction or ischemia, subsequent care should follow current guidelines for management of acute ST-segment elevation myocardial infarction (STEMI) and non–ST-segment elevation ACS (NSTE-ACS).
- Patients with a nonischemic ECG can enter an accelerated CDP designed to provide rapid risk assessment and exclusion of ACS.
- Patients who are hemodynamically unstable, have significant arrhythmias, or evidence of significant heart failure should be evaluated and treated appropriately and are not candidates for an accelerated CDP.
- High-sensitivity cardiac troponin T (hs-cTnT) and high-sensitivity cardiac troponin I (hs-cTnI) are the preferred biomarkers for evaluation of possible ACS.
- Patients classified as low risk (rule out) using the current hs-cTn-based CDPs can generally be discharged directly from the ED without additional testing, although outpatient testing may be considered in selected cases.
- Patients with substantially elevated initial hs-cTn values or those with significant dynamic changes over 1-3 hours are assigned to the abnormal/high-risk category and should be further classified according to the universal definition of myocardial infarction type 1 or 2 or acute or chronic nonischemic cardiac injury.
- High-risk patients should usually be admitted to an inpatient setting for further evaluation and treatment.
- Patients determined to be intermediate risk with the CDP should undergo additional observation with repeat hs-cTn measurements at 3-6 hours and risk assessment using either the modified HEART (history, ECG, age, risk factors, and troponin) score or the ED assessment of chest pain score (EDACS).
- Noninvasive testing should be considered for the intermediate-risk group unless low-risk features are identified using risk scores or noninvasive testing has been performed recently with normal or low-risk findings.
The writing group notes that “safe and efficient” management of chest pain in the ED requires appropriate follow-up after discharge. Timing of follow-up and referral for outpatient noninvasive testing should be influenced by patient risk and results of cardiac testing.
Disclosures for members of the writing group are available with the original article.
A version of this article first appeared on Medscape.com.
Chest pain accounts for more than 7 million ED visits annually. A major challenge is to quickly identify the small number of patients with acute coronary syndrome (ACS) among the large number of patients who have noncardiac conditions.
The new document is intended to provide guidance on how to “practically apply” recommendations from the 2021 American Heart Association/American College of Cardiology Guideline for the Evaluation and Diagnosis of Chest Pain, focusing specifically on patients who present to the ED, the writing group explains.
“A systematic approach – both at the level of the institution and the individual patient – is essential to achieve optimal outcomes for patients presenting with chest pain to the ED,” say writing group chair Michael Kontos, MD, Virginia Commonwealth University, Richmond, and colleagues.
At the institution level, this decision pathway recommends high-sensitivity cardiac troponin (hs-cTn) assays coupled with a clinical decision pathway (CDP) to reduce ED “dwell” times and increase the number of patients with chest pain who can safely be discharged without additional testing. This will decrease ED crowding and limit unnecessary testing, they point out.
At the individual patient level, this document aims to provide structure for the ED evaluation of chest pain, accelerating the evaluation process and matching the intensity of testing and treatment to patient risk.
The 36-page document was published online in the Journal of the American College of Cardiology.
Key summary points in the document include the following:
- Electrocardiogram remains the best initial test for evaluation of chest pain in the ED and should be performed and interpreted within 10 minutes of ED arrival.
- In patients who arrive via ambulance, the prehospital ECG should be reviewed, because ischemic changes may have resolved before ED arrival.
- When the ECG shows evidence of acute infarction or ischemia, subsequent care should follow current guidelines for management of acute ST-segment elevation myocardial infarction (STEMI) and non–ST-segment elevation ACS (NSTE-ACS).
- Patients with a nonischemic ECG can enter an accelerated CDP designed to provide rapid risk assessment and exclusion of ACS.
- Patients who are hemodynamically unstable, have significant arrhythmias, or evidence of significant heart failure should be evaluated and treated appropriately and are not candidates for an accelerated CDP.
- High-sensitivity cardiac troponin T (hs-cTnT) and high-sensitivity cardiac troponin I (hs-cTnI) are the preferred biomarkers for evaluation of possible ACS.
- Patients classified as low risk (rule out) using the current hs-cTn-based CDPs can generally be discharged directly from the ED without additional testing, although outpatient testing may be considered in selected cases.
- Patients with substantially elevated initial hs-cTn values or those with significant dynamic changes over 1-3 hours are assigned to the abnormal/high-risk category and should be further classified according to the universal definition of myocardial infarction type 1 or 2 or acute or chronic nonischemic cardiac injury.
- High-risk patients should usually be admitted to an inpatient setting for further evaluation and treatment.
- Patients determined to be intermediate risk with the CDP should undergo additional observation with repeat hs-cTn measurements at 3-6 hours and risk assessment using either the modified HEART (history, ECG, age, risk factors, and troponin) score or the ED assessment of chest pain score (EDACS).
- Noninvasive testing should be considered for the intermediate-risk group unless low-risk features are identified using risk scores or noninvasive testing has been performed recently with normal or low-risk findings.
The writing group notes that “safe and efficient” management of chest pain in the ED requires appropriate follow-up after discharge. Timing of follow-up and referral for outpatient noninvasive testing should be influenced by patient risk and results of cardiac testing.
Disclosures for members of the writing group are available with the original article.
A version of this article first appeared on Medscape.com.
FROM JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
New advice on artificial pancreas insulin delivery systems
A new consensus statement summarizes the benefits, limitations, and challenges of using automated insulin delivery (AID) systems and provides recommendations for use by people with diabetes.
“Automated insulin delivery systems” is becoming the standard terminology – including by the U.S. Food and Drug Administration – to refer to systems that integrate data from a continuous glucose monitoring (CGM) system via a control algorithm into an insulin pump in order to automate subcutaneous insulin delivery. “Hybrid AID” or “hybrid closed-loop” refers to the current status of these systems, which still require some degree of user input to control glucose levels.
The term “artificial pancreas” was used interchangeably with AID in the past, but it doesn’t take into account exocrine pancreatic function. The term “bionic pancreas” refers to a specific system in development that would ultimately include glucagon along with insulin.
The new consensus report, titled “Automated insulin delivery: Benefits, challenges, and recommendations,” was published online in Diabetes Care and Diabetologia.
The document is geared toward not only diabetologists and other specialists, but also diabetes nurses and specialist dietitians. Colleagues working at regulatory agencies, health care organizations, and related media might also benefit from reading it.
It is endorsed by two professional societies – the European Association for the Study of Diabetes and the American Diabetes Association – and contrasts with other statements about AID systems that are sponsored by their manufacturers, noted document co-author Mark Evans, PhD, professor of diabetic medicine, University of Cambridge, England, in a statement.
“Many clinically relevant aspects, including safety, are addressed in this report. The aim ... is to encourage ongoing improvement of this technology, its safe and effective use, and its accessibility to all who can benefit from it,” Dr. Evans said.
Lead author Jennifer Sherr, MD, PhD, pediatric endocrinology, Yale University, New Haven, Conn., commented that the report “addresses the clinical usage of AID systems from a practical point of view rather than as ... a meta-analysis or a review of all relevant clinical studies. ... As such, the benefits and limitations of systems are discussed while also considering safety, regulatory pathways, and access to this technology.”
AID systems do not mean diabetes is “cured”
Separate recommendations provided at the end of the document are aimed at specific stakeholders, including health care providers, patients and their caregivers, manufacturers, regulatory agencies, and the research community.
The authors make clear in the introduction that, while representing “a significant movement toward optimizing glucose management for individuals with diabetes,” the use of AID systems doesn’t mean that diabetes is “cured.” Rather, “expectations need to be set adequately so that individuals with diabetes and providers understand what such systems can and cannot do.”
In particular, current commercially available AID systems require user input for mealtime insulin dosing and sometimes for correction doses of high blood glucose levels, although the systems at least partially automate that.
“When integrated into care, AID systems hold promise to relieve some of the daily burdens of diabetes care,” the authors write.
The statement also details problems that may arise with the physical devices, including skin irritation from adhesives, occlusion of insulin infusion sets, early CGM sensor failure, and inadequate dosing algorithms.
“Individuals with diabetes who are considering this type of advanced diabetes therapy should not only have appropriate technical understanding of the system but also be able to revert to standard diabetes treatment (that is, nonautomated subcutaneous insulin delivery by pump or injections) in case the AID system fails. They should be able to independently troubleshoot and have access to their health care provider if needed.”
To monitor the impact of the technology, the authors emphasize the importance of the time-in-range metric derived from CGM, with the goal of achieving 70% or greater time in target blood glucose range.
Separate sections of the document address the benefits and limitations of AID systems, education and expectations for both patients and providers, and patient and provider perspectives, including how to handle urgent questions.
Other sections cover special populations such as pregnant women and people with type 2 diabetes, considerations for patient selection for current AID systems, safety, improving access to the technology, liability, and do-it-yourself systems.
Recommendations for health care professionals
A table near the end of the document provides specific recommendations for health care professionals, including the following:
- Be knowledgeable about AID systems and nuances of different systems, including their distinguishing features as well as strengths and weaknesses.
- Inform patients with diabetes about AID systems, including review of currently available systems, and create realistic expectations for device use.
- Involve patients with diabetes in shared decision-making when considering use of AID systems.
- Share information with patients with diabetes, as well as their peers, about general standards set by national and international guidelines on AID systems.
- Provide an on-call number or method by which a person with diabetes can always access support from a health care provider at the practice, including weekends and nights.
- Implement, potentially, protocols on times when AID systems should not be used.
- Use an individual’s health data to improve quality of care and health outcomes.
Most members of the ADA/EASD Diabetes Technology Working Group work with industry, but industry had no input on the project. Dr. Sherr has reported conducting clinical trials for Eli Lilly, Insulet, and Medtronic, and has received in-kind support for research studies from Dexcom and Medtronic. She has also reported consulting for Eli Lilly, Lexicon, Medtronic, and Sanofi, and being an advisory board member for Bigfoot Biomedical, Cecelia Health, Eli Lilly, Insulet, T1D Fund, and Vertex Pharmaceuticals. Dr. Evans has reported conducting clinical trials or research collaborations for, serving on advisory boards for, or receiving speakers fees or travel support from Medtronic, Roche, Abbott Diabetes Care, Dexcom, Novo Nordisk, Eli Lilly, Sanofi, Zucara Therapeutics, Pila Pharma, and AstraZeneca. The University of Cambridge has received salary support for Dr. Evans from the National Health Service.
A version of this article first appeared on Medscape.com.
A new consensus statement summarizes the benefits, limitations, and challenges of using automated insulin delivery (AID) systems and provides recommendations for use by people with diabetes.
“Automated insulin delivery systems” is becoming the standard terminology – including by the U.S. Food and Drug Administration – to refer to systems that integrate data from a continuous glucose monitoring (CGM) system via a control algorithm into an insulin pump in order to automate subcutaneous insulin delivery. “Hybrid AID” or “hybrid closed-loop” refers to the current status of these systems, which still require some degree of user input to control glucose levels.
The term “artificial pancreas” was used interchangeably with AID in the past, but it doesn’t take into account exocrine pancreatic function. The term “bionic pancreas” refers to a specific system in development that would ultimately include glucagon along with insulin.
The new consensus report, titled “Automated insulin delivery: Benefits, challenges, and recommendations,” was published online in Diabetes Care and Diabetologia.
The document is geared toward not only diabetologists and other specialists, but also diabetes nurses and specialist dietitians. Colleagues working at regulatory agencies, health care organizations, and related media might also benefit from reading it.
It is endorsed by two professional societies – the European Association for the Study of Diabetes and the American Diabetes Association – and contrasts with other statements about AID systems that are sponsored by their manufacturers, noted document co-author Mark Evans, PhD, professor of diabetic medicine, University of Cambridge, England, in a statement.
“Many clinically relevant aspects, including safety, are addressed in this report. The aim ... is to encourage ongoing improvement of this technology, its safe and effective use, and its accessibility to all who can benefit from it,” Dr. Evans said.
Lead author Jennifer Sherr, MD, PhD, pediatric endocrinology, Yale University, New Haven, Conn., commented that the report “addresses the clinical usage of AID systems from a practical point of view rather than as ... a meta-analysis or a review of all relevant clinical studies. ... As such, the benefits and limitations of systems are discussed while also considering safety, regulatory pathways, and access to this technology.”
AID systems do not mean diabetes is “cured”
Separate recommendations provided at the end of the document are aimed at specific stakeholders, including health care providers, patients and their caregivers, manufacturers, regulatory agencies, and the research community.
The authors make clear in the introduction that, while representing “a significant movement toward optimizing glucose management for individuals with diabetes,” the use of AID systems doesn’t mean that diabetes is “cured.” Rather, “expectations need to be set adequately so that individuals with diabetes and providers understand what such systems can and cannot do.”
In particular, current commercially available AID systems require user input for mealtime insulin dosing and sometimes for correction doses of high blood glucose levels, although the systems at least partially automate that.
“When integrated into care, AID systems hold promise to relieve some of the daily burdens of diabetes care,” the authors write.
The statement also details problems that may arise with the physical devices, including skin irritation from adhesives, occlusion of insulin infusion sets, early CGM sensor failure, and inadequate dosing algorithms.
“Individuals with diabetes who are considering this type of advanced diabetes therapy should not only have appropriate technical understanding of the system but also be able to revert to standard diabetes treatment (that is, nonautomated subcutaneous insulin delivery by pump or injections) in case the AID system fails. They should be able to independently troubleshoot and have access to their health care provider if needed.”
To monitor the impact of the technology, the authors emphasize the importance of the time-in-range metric derived from CGM, with the goal of achieving 70% or greater time in target blood glucose range.
Separate sections of the document address the benefits and limitations of AID systems, education and expectations for both patients and providers, and patient and provider perspectives, including how to handle urgent questions.
Other sections cover special populations such as pregnant women and people with type 2 diabetes, considerations for patient selection for current AID systems, safety, improving access to the technology, liability, and do-it-yourself systems.
Recommendations for health care professionals
A table near the end of the document provides specific recommendations for health care professionals, including the following:
- Be knowledgeable about AID systems and nuances of different systems, including their distinguishing features as well as strengths and weaknesses.
- Inform patients with diabetes about AID systems, including review of currently available systems, and create realistic expectations for device use.
- Involve patients with diabetes in shared decision-making when considering use of AID systems.
- Share information with patients with diabetes, as well as their peers, about general standards set by national and international guidelines on AID systems.
- Provide an on-call number or method by which a person with diabetes can always access support from a health care provider at the practice, including weekends and nights.
- Implement, potentially, protocols on times when AID systems should not be used.
- Use an individual’s health data to improve quality of care and health outcomes.
Most members of the ADA/EASD Diabetes Technology Working Group work with industry, but industry had no input on the project. Dr. Sherr has reported conducting clinical trials for Eli Lilly, Insulet, and Medtronic, and has received in-kind support for research studies from Dexcom and Medtronic. She has also reported consulting for Eli Lilly, Lexicon, Medtronic, and Sanofi, and being an advisory board member for Bigfoot Biomedical, Cecelia Health, Eli Lilly, Insulet, T1D Fund, and Vertex Pharmaceuticals. Dr. Evans has reported conducting clinical trials or research collaborations for, serving on advisory boards for, or receiving speakers fees or travel support from Medtronic, Roche, Abbott Diabetes Care, Dexcom, Novo Nordisk, Eli Lilly, Sanofi, Zucara Therapeutics, Pila Pharma, and AstraZeneca. The University of Cambridge has received salary support for Dr. Evans from the National Health Service.
A version of this article first appeared on Medscape.com.
A new consensus statement summarizes the benefits, limitations, and challenges of using automated insulin delivery (AID) systems and provides recommendations for use by people with diabetes.
“Automated insulin delivery systems” is becoming the standard terminology – including by the U.S. Food and Drug Administration – to refer to systems that integrate data from a continuous glucose monitoring (CGM) system via a control algorithm into an insulin pump in order to automate subcutaneous insulin delivery. “Hybrid AID” or “hybrid closed-loop” refers to the current status of these systems, which still require some degree of user input to control glucose levels.
The term “artificial pancreas” was used interchangeably with AID in the past, but it doesn’t take into account exocrine pancreatic function. The term “bionic pancreas” refers to a specific system in development that would ultimately include glucagon along with insulin.
The new consensus report, titled “Automated insulin delivery: Benefits, challenges, and recommendations,” was published online in Diabetes Care and Diabetologia.
The document is geared toward not only diabetologists and other specialists, but also diabetes nurses and specialist dietitians. Colleagues working at regulatory agencies, health care organizations, and related media might also benefit from reading it.
It is endorsed by two professional societies – the European Association for the Study of Diabetes and the American Diabetes Association – and contrasts with other statements about AID systems that are sponsored by their manufacturers, noted document co-author Mark Evans, PhD, professor of diabetic medicine, University of Cambridge, England, in a statement.
“Many clinically relevant aspects, including safety, are addressed in this report. The aim ... is to encourage ongoing improvement of this technology, its safe and effective use, and its accessibility to all who can benefit from it,” Dr. Evans said.
Lead author Jennifer Sherr, MD, PhD, pediatric endocrinology, Yale University, New Haven, Conn., commented that the report “addresses the clinical usage of AID systems from a practical point of view rather than as ... a meta-analysis or a review of all relevant clinical studies. ... As such, the benefits and limitations of systems are discussed while also considering safety, regulatory pathways, and access to this technology.”
AID systems do not mean diabetes is “cured”
Separate recommendations provided at the end of the document are aimed at specific stakeholders, including health care providers, patients and their caregivers, manufacturers, regulatory agencies, and the research community.
The authors make clear in the introduction that, while representing “a significant movement toward optimizing glucose management for individuals with diabetes,” the use of AID systems doesn’t mean that diabetes is “cured.” Rather, “expectations need to be set adequately so that individuals with diabetes and providers understand what such systems can and cannot do.”
In particular, current commercially available AID systems require user input for mealtime insulin dosing and sometimes for correction doses of high blood glucose levels, although the systems at least partially automate that.
“When integrated into care, AID systems hold promise to relieve some of the daily burdens of diabetes care,” the authors write.
The statement also details problems that may arise with the physical devices, including skin irritation from adhesives, occlusion of insulin infusion sets, early CGM sensor failure, and inadequate dosing algorithms.
“Individuals with diabetes who are considering this type of advanced diabetes therapy should not only have appropriate technical understanding of the system but also be able to revert to standard diabetes treatment (that is, nonautomated subcutaneous insulin delivery by pump or injections) in case the AID system fails. They should be able to independently troubleshoot and have access to their health care provider if needed.”
To monitor the impact of the technology, the authors emphasize the importance of the time-in-range metric derived from CGM, with the goal of achieving 70% or greater time in target blood glucose range.
Separate sections of the document address the benefits and limitations of AID systems, education and expectations for both patients and providers, and patient and provider perspectives, including how to handle urgent questions.
Other sections cover special populations such as pregnant women and people with type 2 diabetes, considerations for patient selection for current AID systems, safety, improving access to the technology, liability, and do-it-yourself systems.
Recommendations for health care professionals
A table near the end of the document provides specific recommendations for health care professionals, including the following:
- Be knowledgeable about AID systems and nuances of different systems, including their distinguishing features as well as strengths and weaknesses.
- Inform patients with diabetes about AID systems, including review of currently available systems, and create realistic expectations for device use.
- Involve patients with diabetes in shared decision-making when considering use of AID systems.
- Share information with patients with diabetes, as well as their peers, about general standards set by national and international guidelines on AID systems.
- Provide an on-call number or method by which a person with diabetes can always access support from a health care provider at the practice, including weekends and nights.
- Implement, potentially, protocols on times when AID systems should not be used.
- Use an individual’s health data to improve quality of care and health outcomes.
Most members of the ADA/EASD Diabetes Technology Working Group work with industry, but industry had no input on the project. Dr. Sherr has reported conducting clinical trials for Eli Lilly, Insulet, and Medtronic, and has received in-kind support for research studies from Dexcom and Medtronic. She has also reported consulting for Eli Lilly, Lexicon, Medtronic, and Sanofi, and being an advisory board member for Bigfoot Biomedical, Cecelia Health, Eli Lilly, Insulet, T1D Fund, and Vertex Pharmaceuticals. Dr. Evans has reported conducting clinical trials or research collaborations for, serving on advisory boards for, or receiving speakers fees or travel support from Medtronic, Roche, Abbott Diabetes Care, Dexcom, Novo Nordisk, Eli Lilly, Sanofi, Zucara Therapeutics, Pila Pharma, and AstraZeneca. The University of Cambridge has received salary support for Dr. Evans from the National Health Service.
A version of this article first appeared on Medscape.com.
FROM DIABETES CARE AND DIABETOLOGIA
At the front lines of long COVID, local clinics prove vital
Big-name hospital chains across the United States are opening dedicated centers to help patients dealing with long COVID. But so are the lower-profile clinics and hospitals run by cities, counties and states – including Harborview Medical Center in Seattle.
They serve areas ranging from Campbell County, Wyo., with 47,000 residents, to New York City, with its 8.4 million people. Many providers working there are searching for innovative ways to approach this lingering illness with its variety of symptoms, from brain fog to shortness of breath to depression and more.
Their efforts often fall below the radar, with still-scant serious media attention to long COVID or the public health employees working to treat ailing patients.
Why are state and local health agencies taking on these duties?
They’re leading the way in part because the federal government has made only limited efforts, said Lisa McCorkell, a cofounder of the Patient-Led Research Collaborative. The international group was founded in spring 2020 by researchers who are also long COVID patients.
“It’s a big reason why long COVID isn’t talked about as much,” Ms. McCorkell said. “It’s definitely a national issue. But it trickles down to state and local health departments, and there’s not enough resources.”
The government clinics may be accessible to people without insurance and often are cheaper than clinics at private hospitals.
Harborview has treated more than 1,000 patients with long COVID, and another 200 patients are awaiting treatment, said Jessica Bender, MD, a codirector of the University of Washington Post-COVID Rehabilitation and Recovery Clinic in Seattle’s First Hill neighborhood.
The group Survivor Corps offers lists by states of clinics. While the publicly run clinics may be less expensive or even free for some patients, methods of payment vary from clinic to clinic. Federally qualified health clinics offer treatment on a sliding scale. For instance, the Riverside University Health System in California has federally qualified centers. And other providers who are not federally qualified also offer care paid for on a sliding scale. They include Campbell County Health, where some residents are eligible for discounts of 25%-100%, said spokesperson Norberto Orellana.
At Harborview, Dr. Bender said the public hospital’s post-COVID clinic initially began with a staff of rehabilitation doctors but expanded in 2021 to include family and internal medicine doctors. And it offers mental health programs with rehabilitation psychologists who instruct on how to deal with doctors or loved ones who don’t believe that long COVID exists.
“I have patients who really have been devastated by the lack of support from coworkers [and] family,” Dr. Bender said.
In Campbell County, Wyo., the pandemic surge did not arrive in earnest until late 2021. Physical therapists at Campbell County’s Health Rehabilitation Services organized a rehabilitation program for residents with long COVID after recognizing the need, said Shannon Sorensen, rehabilitation director at Campbell County Health.
“We had patients coming in showing chest pain, or heart palpitations. There were people trying to get back to work. They were frustrated,” Ms. Sorensen said.
Myalgic encephalomyelitis and chronic fatigue syndrome activists have embraced the fight to recognize and help long COVID patients, noting the similarities between the conditions, and hope to help kickstart more organized research, treatment and benefits for long COVID sufferers and myalgic encephalomyelitis/chronic fatigue syndrome patients alike.
In Ft. Collins, Colo., disability activist Alison Sbrana has long had myalgic encephalomyelitis. She and other members of the local chapter of ME Action have met with state officials for several years and are finally seeing the results of those efforts.
Colorado Gov. Jared Polis has created the full-time position of policy adviser for long COVID and post–viral infection planning.
“This is one way forward of how state governments are (finally) paying attention to infection-triggered chronic illnesses and starting to think ahead on them,” Ms. Sbrana said.
New York City’s Health + Hospitals launched what may be the most expansive long COVID treatment program in the nation in April 2021. Called AfterCare, it provides physical and mental health services as well as community support systems and financial assistance.
A persistent issue for patients is that there isn’t yet a test for long COVID, like there is for COVID-19, said Amanda Johnson, MD, assistant vice president for ambulatory care and population health at New York Health + Hospitals. “It’s in many ways a diagnosis of exclusion. You have to make sure their shortness of breath isn’t caused by something else. The same with anemia,” she said.
California’s Department of Public Health has a detailed website devoted to the topic, including videos of “long haulers” describing their experiences.
Vermont is one of several states studying long COVID, said Mark Levine, MD, the state health commissioner. The state, in collaboration with the University of Vermont, has established a surveillance project to determine how many people have long COVID, as well as how severe it is, how long it lasts, and potential predispositions.
The University of Utah, Salt Lake City, established a comprehensive COVID-19 clinic more than a year ago that also handles long COVID patients, said Jeannette Brown, MD, PhD, an associate professor at the school and director of the COVID-19 clinic.
Jennifer Chevinsky, MD, MPH, already had a deep understanding of long COVID when she landed in Riverside County, Calif., in the summer of 2021. She came from Atlanta, where as part of her job as an epidemic intelligence service officer at the CDC, she heard stories of COVID-19 patients who were not getting better.
Now she is a deputy public health officer for Riverside County, in a region known for its deserts, sizzling summer temperatures and diverse populations. She said her department has helped launch programs such as post–COVID-19 follow-up phone calls and long COVID training programs that reach out to the many Latino residents in this county of 2.4 million people. It also includes Black and Native American residents.
“We’re making sure information is circulated with community and faith-based organizations, and community health workers,” she said.
Ms. McCorkell said there is still much work to do to raise public awareness of the risks of long COVID and how to obtain care for patients. She would like to see a national public health campaign about long COVID, possibly spearheaded by the Centers for Disease Control and Prevention in partnership with local health workers and community-based organizations.
“That,” she said, “could make a big difference.”
A version of this article first appeared on WebMD.com.
Big-name hospital chains across the United States are opening dedicated centers to help patients dealing with long COVID. But so are the lower-profile clinics and hospitals run by cities, counties and states – including Harborview Medical Center in Seattle.
They serve areas ranging from Campbell County, Wyo., with 47,000 residents, to New York City, with its 8.4 million people. Many providers working there are searching for innovative ways to approach this lingering illness with its variety of symptoms, from brain fog to shortness of breath to depression and more.
Their efforts often fall below the radar, with still-scant serious media attention to long COVID or the public health employees working to treat ailing patients.
Why are state and local health agencies taking on these duties?
They’re leading the way in part because the federal government has made only limited efforts, said Lisa McCorkell, a cofounder of the Patient-Led Research Collaborative. The international group was founded in spring 2020 by researchers who are also long COVID patients.
“It’s a big reason why long COVID isn’t talked about as much,” Ms. McCorkell said. “It’s definitely a national issue. But it trickles down to state and local health departments, and there’s not enough resources.”
The government clinics may be accessible to people without insurance and often are cheaper than clinics at private hospitals.
Harborview has treated more than 1,000 patients with long COVID, and another 200 patients are awaiting treatment, said Jessica Bender, MD, a codirector of the University of Washington Post-COVID Rehabilitation and Recovery Clinic in Seattle’s First Hill neighborhood.
The group Survivor Corps offers lists by states of clinics. While the publicly run clinics may be less expensive or even free for some patients, methods of payment vary from clinic to clinic. Federally qualified health clinics offer treatment on a sliding scale. For instance, the Riverside University Health System in California has federally qualified centers. And other providers who are not federally qualified also offer care paid for on a sliding scale. They include Campbell County Health, where some residents are eligible for discounts of 25%-100%, said spokesperson Norberto Orellana.
At Harborview, Dr. Bender said the public hospital’s post-COVID clinic initially began with a staff of rehabilitation doctors but expanded in 2021 to include family and internal medicine doctors. And it offers mental health programs with rehabilitation psychologists who instruct on how to deal with doctors or loved ones who don’t believe that long COVID exists.
“I have patients who really have been devastated by the lack of support from coworkers [and] family,” Dr. Bender said.
In Campbell County, Wyo., the pandemic surge did not arrive in earnest until late 2021. Physical therapists at Campbell County’s Health Rehabilitation Services organized a rehabilitation program for residents with long COVID after recognizing the need, said Shannon Sorensen, rehabilitation director at Campbell County Health.
“We had patients coming in showing chest pain, or heart palpitations. There were people trying to get back to work. They were frustrated,” Ms. Sorensen said.
Myalgic encephalomyelitis and chronic fatigue syndrome activists have embraced the fight to recognize and help long COVID patients, noting the similarities between the conditions, and hope to help kickstart more organized research, treatment and benefits for long COVID sufferers and myalgic encephalomyelitis/chronic fatigue syndrome patients alike.
In Ft. Collins, Colo., disability activist Alison Sbrana has long had myalgic encephalomyelitis. She and other members of the local chapter of ME Action have met with state officials for several years and are finally seeing the results of those efforts.
Colorado Gov. Jared Polis has created the full-time position of policy adviser for long COVID and post–viral infection planning.
“This is one way forward of how state governments are (finally) paying attention to infection-triggered chronic illnesses and starting to think ahead on them,” Ms. Sbrana said.
New York City’s Health + Hospitals launched what may be the most expansive long COVID treatment program in the nation in April 2021. Called AfterCare, it provides physical and mental health services as well as community support systems and financial assistance.
A persistent issue for patients is that there isn’t yet a test for long COVID, like there is for COVID-19, said Amanda Johnson, MD, assistant vice president for ambulatory care and population health at New York Health + Hospitals. “It’s in many ways a diagnosis of exclusion. You have to make sure their shortness of breath isn’t caused by something else. The same with anemia,” she said.
California’s Department of Public Health has a detailed website devoted to the topic, including videos of “long haulers” describing their experiences.
Vermont is one of several states studying long COVID, said Mark Levine, MD, the state health commissioner. The state, in collaboration with the University of Vermont, has established a surveillance project to determine how many people have long COVID, as well as how severe it is, how long it lasts, and potential predispositions.
The University of Utah, Salt Lake City, established a comprehensive COVID-19 clinic more than a year ago that also handles long COVID patients, said Jeannette Brown, MD, PhD, an associate professor at the school and director of the COVID-19 clinic.
Jennifer Chevinsky, MD, MPH, already had a deep understanding of long COVID when she landed in Riverside County, Calif., in the summer of 2021. She came from Atlanta, where as part of her job as an epidemic intelligence service officer at the CDC, she heard stories of COVID-19 patients who were not getting better.
Now she is a deputy public health officer for Riverside County, in a region known for its deserts, sizzling summer temperatures and diverse populations. She said her department has helped launch programs such as post–COVID-19 follow-up phone calls and long COVID training programs that reach out to the many Latino residents in this county of 2.4 million people. It also includes Black and Native American residents.
“We’re making sure information is circulated with community and faith-based organizations, and community health workers,” she said.
Ms. McCorkell said there is still much work to do to raise public awareness of the risks of long COVID and how to obtain care for patients. She would like to see a national public health campaign about long COVID, possibly spearheaded by the Centers for Disease Control and Prevention in partnership with local health workers and community-based organizations.
“That,” she said, “could make a big difference.”
A version of this article first appeared on WebMD.com.
Big-name hospital chains across the United States are opening dedicated centers to help patients dealing with long COVID. But so are the lower-profile clinics and hospitals run by cities, counties and states – including Harborview Medical Center in Seattle.
They serve areas ranging from Campbell County, Wyo., with 47,000 residents, to New York City, with its 8.4 million people. Many providers working there are searching for innovative ways to approach this lingering illness with its variety of symptoms, from brain fog to shortness of breath to depression and more.
Their efforts often fall below the radar, with still-scant serious media attention to long COVID or the public health employees working to treat ailing patients.
Why are state and local health agencies taking on these duties?
They’re leading the way in part because the federal government has made only limited efforts, said Lisa McCorkell, a cofounder of the Patient-Led Research Collaborative. The international group was founded in spring 2020 by researchers who are also long COVID patients.
“It’s a big reason why long COVID isn’t talked about as much,” Ms. McCorkell said. “It’s definitely a national issue. But it trickles down to state and local health departments, and there’s not enough resources.”
The government clinics may be accessible to people without insurance and often are cheaper than clinics at private hospitals.
Harborview has treated more than 1,000 patients with long COVID, and another 200 patients are awaiting treatment, said Jessica Bender, MD, a codirector of the University of Washington Post-COVID Rehabilitation and Recovery Clinic in Seattle’s First Hill neighborhood.
The group Survivor Corps offers lists by states of clinics. While the publicly run clinics may be less expensive or even free for some patients, methods of payment vary from clinic to clinic. Federally qualified health clinics offer treatment on a sliding scale. For instance, the Riverside University Health System in California has federally qualified centers. And other providers who are not federally qualified also offer care paid for on a sliding scale. They include Campbell County Health, where some residents are eligible for discounts of 25%-100%, said spokesperson Norberto Orellana.
At Harborview, Dr. Bender said the public hospital’s post-COVID clinic initially began with a staff of rehabilitation doctors but expanded in 2021 to include family and internal medicine doctors. And it offers mental health programs with rehabilitation psychologists who instruct on how to deal with doctors or loved ones who don’t believe that long COVID exists.
“I have patients who really have been devastated by the lack of support from coworkers [and] family,” Dr. Bender said.
In Campbell County, Wyo., the pandemic surge did not arrive in earnest until late 2021. Physical therapists at Campbell County’s Health Rehabilitation Services organized a rehabilitation program for residents with long COVID after recognizing the need, said Shannon Sorensen, rehabilitation director at Campbell County Health.
“We had patients coming in showing chest pain, or heart palpitations. There were people trying to get back to work. They were frustrated,” Ms. Sorensen said.
Myalgic encephalomyelitis and chronic fatigue syndrome activists have embraced the fight to recognize and help long COVID patients, noting the similarities between the conditions, and hope to help kickstart more organized research, treatment and benefits for long COVID sufferers and myalgic encephalomyelitis/chronic fatigue syndrome patients alike.
In Ft. Collins, Colo., disability activist Alison Sbrana has long had myalgic encephalomyelitis. She and other members of the local chapter of ME Action have met with state officials for several years and are finally seeing the results of those efforts.
Colorado Gov. Jared Polis has created the full-time position of policy adviser for long COVID and post–viral infection planning.
“This is one way forward of how state governments are (finally) paying attention to infection-triggered chronic illnesses and starting to think ahead on them,” Ms. Sbrana said.
New York City’s Health + Hospitals launched what may be the most expansive long COVID treatment program in the nation in April 2021. Called AfterCare, it provides physical and mental health services as well as community support systems and financial assistance.
A persistent issue for patients is that there isn’t yet a test for long COVID, like there is for COVID-19, said Amanda Johnson, MD, assistant vice president for ambulatory care and population health at New York Health + Hospitals. “It’s in many ways a diagnosis of exclusion. You have to make sure their shortness of breath isn’t caused by something else. The same with anemia,” she said.
California’s Department of Public Health has a detailed website devoted to the topic, including videos of “long haulers” describing their experiences.
Vermont is one of several states studying long COVID, said Mark Levine, MD, the state health commissioner. The state, in collaboration with the University of Vermont, has established a surveillance project to determine how many people have long COVID, as well as how severe it is, how long it lasts, and potential predispositions.
The University of Utah, Salt Lake City, established a comprehensive COVID-19 clinic more than a year ago that also handles long COVID patients, said Jeannette Brown, MD, PhD, an associate professor at the school and director of the COVID-19 clinic.
Jennifer Chevinsky, MD, MPH, already had a deep understanding of long COVID when she landed in Riverside County, Calif., in the summer of 2021. She came from Atlanta, where as part of her job as an epidemic intelligence service officer at the CDC, she heard stories of COVID-19 patients who were not getting better.
Now she is a deputy public health officer for Riverside County, in a region known for its deserts, sizzling summer temperatures and diverse populations. She said her department has helped launch programs such as post–COVID-19 follow-up phone calls and long COVID training programs that reach out to the many Latino residents in this county of 2.4 million people. It also includes Black and Native American residents.
“We’re making sure information is circulated with community and faith-based organizations, and community health workers,” she said.
Ms. McCorkell said there is still much work to do to raise public awareness of the risks of long COVID and how to obtain care for patients. She would like to see a national public health campaign about long COVID, possibly spearheaded by the Centers for Disease Control and Prevention in partnership with local health workers and community-based organizations.
“That,” she said, “could make a big difference.”
A version of this article first appeared on WebMD.com.