User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Cutting dietary simple sugars may relieve GERD symptoms
Minimizing dietary consumption of simple sugars may help alleviate symptoms of gastroesophageal reflux disease (GERD), new data suggest.
People who consumed lower quantities of simple sugars experienced significant improvement in total acid exposure time, number of reflux episodes, and subjective reports of heartburn and reflux symptoms, compared with those consuming higher amounts of simple sugars, the authors report.
The authors call their study the first randomized controlled diet intervention trial to investigate both the amount and type of carbohydrate on symptomatic GERD and one of only a few to investigate any type of dietary intervention for GERD.
“There’s really almost no rigorous scientific evidence on any of the foods or ingredients or nutrients that are often recommended to avoid if you have GERD,” corresponding author Heidi J. Silver, RD, PhD, told this news organization. Dr. Silver is research professor of medicine at Vanderbilt University School of Medicine and director of the Vanderbilt Diet, Body Composition, and Human Metabolism Core in Nashville, Tenn.
Even the avoidance of fatty foods, which has been long promoted as part of GERD management, has little evidence to support it.
“With fat, there’s some belief that it may slow down gastric emptying. Therefore, if you had slower gastric emptying, you would have a longer time for the food to put pressure on the lower esophageal sphincter and create an environment for reflux. So, it’s kind of conceptually what is thought but not really tested,” she notes.
The findings were published online in the American Journal of Gastroenterology.
Greatest symptom reduction with low simple carb intake
To test the role of dietary carbohydrates, Dr. Silver and her colleagues randomly assigned 98 U.S. veterans with symptomatic GERD to intake of one of four diets with varying carbohydrate types and amounts for 9 weeks: high total/high simple (the control group), high total/low simple, low total/high simple, or low total/low simple. The total caloric intake was approximately the same for all groups.
At baseline, the average total carbohydrate consumption was 43.7% of calories, and the average simple sugar intake was 116.5 g/d. The two “low-total” groups averaged about a 10% reduction in carbohydrate calories. The “low-simple” groups reduced simple sugars by about half.
There were no changes in body weight in the control group, whereas all three of the other groups lost some weight, ranging from 1.5-2.0 kg (3.3-4.4 lb) despite calorie totals designed for weight maintenance.
There was a significant effect of diet on the two primary outcomes, total esophageal acid exposure time, and total number of reflux episodes, as measured by 24-hour ambulatory pH monitoring. The change in total acid exposure time was significantly greater for the high total/low simple group, compared with the high total/high simple group.
The participants’ ratings of symptoms assessed by the Gastroesophageal Reflux Disease Questionnaire and the GERD Symptom Assessment Scale, including heartburn frequency and severity, pain in throat/chest, and sleep disturbance, improved in all modified diet groups, compared with the control group. The mean degree of improvement in heartburn and regurgitation was twice as great for the modified diets, compared with the controls, and was greatest for the two “low-simple” carb groups.
Dr. Silver and colleagues hypothesize that the differential effects of the diets may relate to the way that dietary carbohydrates are sensed in the gastrointestinal tract after being enzymatically degraded into monosaccharides, possibly affecting lower esophageal tone via the effects of gut-derived hormones including ghrelin and glucagon-like peptide 1 that are secreted in response to macronutrient intake.
Although more data are needed about the effects of carbohydrates in GERD, Dr. Silver advised, “I do think it would be smart for clinicians, when they’re discussing diet, that they bring up the simple sugars. There’s no potential harm in reducing simple sugars. You’re only benefiting yourself in multiple ways. We know that the consumption of simple sugars is extremely excessive, not just in America but worldwide.”
Asked to comment, Philip O. Katz, MD, professor of medicine and director of the GI Function Laboratories at Weill Cornell Medicine, New York, told this news organization that “this is one of the better-designed studies with a lot of care looking at a lot of endpoints that are intriguing and useful.”
“What it says to me is there is potential for nonpharmacologic interventions for GERD that include diet change for helping patients,” he said. “This shows promise for a diet that doesn’t just concentrate on fat or acidic products and is a possible way of augmenting reflux treatment.”
However, Dr. Katz cautioned, “I don’t think anybody should do more with a 9-week study than look at it as good potential.”
“I would tell patients that this is something that they might try, but I wouldn’t make it a rigid requirement based on these data,” he added. “If I were involved in this study, the next thing I would do is transition it to real life and look at compliance to see if results were sustained at 18 weeks or 6 months.”
Diet part of an ‘overall reflux program’
Overall, Dr. Katz, who was the first author of the American College of Gastroenterology’s Clinical Guideline for the Diagnosis and Management of Gastroesophageal Reflux Disease, pointed out that the main nonpharmacologic advice for GERD management includes “Eat smaller meals, don’t eat before bed, don’t lie down after you eat, and reduce any individually known trigger foods.”
Essentially, he views dietary interventions as complementary to medication and other interventions.
“When people really have GERD – not just heartburn – dietary change is an adjunct,” Dr. Katz said. “GERD is a real disease, like diabetes. For some with GERD, maybe this is the only thing they need to do. But, in general, this will be an adjunct to augment an overall reflux program.”
And that program, both Dr. Silver and Dr. Katz said, should include referral to a dietitian or nutritionist.
“If you’re going to invest time in getting your patients to change their diet, it should be done with professional help, a qualified nutritionist who can work with the patient. This should not be a fad,” Dr. Katz said.
Dr. Silver noted, “In contrast to a physician, the dietitian looks at everything the person is eating. If they’re consuming a lot of simple sugars, I certainly would make that recommendation to reduce, along with other recommendations for GERD. It could be easily incorporated. We included examples in the paper of what we did to achieve that reduction and some things clinicians could focus on.”
One obvious approach, she said, is to advise patients to cut the amount of sugared soda they’re drinking, if not eliminate it entirely.
The study was funded by a VA Merit Award. Dr. Silver has no further disclosures. Dr. Katz is a consultant for Phathom Pharmaceuticals and Sebella Pharmaceuticals and serves on an advisory board for AstraZeneca.
A version of this article first appeared on Medscape.com.
Minimizing dietary consumption of simple sugars may help alleviate symptoms of gastroesophageal reflux disease (GERD), new data suggest.
People who consumed lower quantities of simple sugars experienced significant improvement in total acid exposure time, number of reflux episodes, and subjective reports of heartburn and reflux symptoms, compared with those consuming higher amounts of simple sugars, the authors report.
The authors call their study the first randomized controlled diet intervention trial to investigate both the amount and type of carbohydrate on symptomatic GERD and one of only a few to investigate any type of dietary intervention for GERD.
“There’s really almost no rigorous scientific evidence on any of the foods or ingredients or nutrients that are often recommended to avoid if you have GERD,” corresponding author Heidi J. Silver, RD, PhD, told this news organization. Dr. Silver is research professor of medicine at Vanderbilt University School of Medicine and director of the Vanderbilt Diet, Body Composition, and Human Metabolism Core in Nashville, Tenn.
Even the avoidance of fatty foods, which has been long promoted as part of GERD management, has little evidence to support it.
“With fat, there’s some belief that it may slow down gastric emptying. Therefore, if you had slower gastric emptying, you would have a longer time for the food to put pressure on the lower esophageal sphincter and create an environment for reflux. So, it’s kind of conceptually what is thought but not really tested,” she notes.
The findings were published online in the American Journal of Gastroenterology.
Greatest symptom reduction with low simple carb intake
To test the role of dietary carbohydrates, Dr. Silver and her colleagues randomly assigned 98 U.S. veterans with symptomatic GERD to intake of one of four diets with varying carbohydrate types and amounts for 9 weeks: high total/high simple (the control group), high total/low simple, low total/high simple, or low total/low simple. The total caloric intake was approximately the same for all groups.
At baseline, the average total carbohydrate consumption was 43.7% of calories, and the average simple sugar intake was 116.5 g/d. The two “low-total” groups averaged about a 10% reduction in carbohydrate calories. The “low-simple” groups reduced simple sugars by about half.
There were no changes in body weight in the control group, whereas all three of the other groups lost some weight, ranging from 1.5-2.0 kg (3.3-4.4 lb) despite calorie totals designed for weight maintenance.
There was a significant effect of diet on the two primary outcomes, total esophageal acid exposure time, and total number of reflux episodes, as measured by 24-hour ambulatory pH monitoring. The change in total acid exposure time was significantly greater for the high total/low simple group, compared with the high total/high simple group.
The participants’ ratings of symptoms assessed by the Gastroesophageal Reflux Disease Questionnaire and the GERD Symptom Assessment Scale, including heartburn frequency and severity, pain in throat/chest, and sleep disturbance, improved in all modified diet groups, compared with the control group. The mean degree of improvement in heartburn and regurgitation was twice as great for the modified diets, compared with the controls, and was greatest for the two “low-simple” carb groups.
Dr. Silver and colleagues hypothesize that the differential effects of the diets may relate to the way that dietary carbohydrates are sensed in the gastrointestinal tract after being enzymatically degraded into monosaccharides, possibly affecting lower esophageal tone via the effects of gut-derived hormones including ghrelin and glucagon-like peptide 1 that are secreted in response to macronutrient intake.
Although more data are needed about the effects of carbohydrates in GERD, Dr. Silver advised, “I do think it would be smart for clinicians, when they’re discussing diet, that they bring up the simple sugars. There’s no potential harm in reducing simple sugars. You’re only benefiting yourself in multiple ways. We know that the consumption of simple sugars is extremely excessive, not just in America but worldwide.”
Asked to comment, Philip O. Katz, MD, professor of medicine and director of the GI Function Laboratories at Weill Cornell Medicine, New York, told this news organization that “this is one of the better-designed studies with a lot of care looking at a lot of endpoints that are intriguing and useful.”
“What it says to me is there is potential for nonpharmacologic interventions for GERD that include diet change for helping patients,” he said. “This shows promise for a diet that doesn’t just concentrate on fat or acidic products and is a possible way of augmenting reflux treatment.”
However, Dr. Katz cautioned, “I don’t think anybody should do more with a 9-week study than look at it as good potential.”
“I would tell patients that this is something that they might try, but I wouldn’t make it a rigid requirement based on these data,” he added. “If I were involved in this study, the next thing I would do is transition it to real life and look at compliance to see if results were sustained at 18 weeks or 6 months.”
Diet part of an ‘overall reflux program’
Overall, Dr. Katz, who was the first author of the American College of Gastroenterology’s Clinical Guideline for the Diagnosis and Management of Gastroesophageal Reflux Disease, pointed out that the main nonpharmacologic advice for GERD management includes “Eat smaller meals, don’t eat before bed, don’t lie down after you eat, and reduce any individually known trigger foods.”
Essentially, he views dietary interventions as complementary to medication and other interventions.
“When people really have GERD – not just heartburn – dietary change is an adjunct,” Dr. Katz said. “GERD is a real disease, like diabetes. For some with GERD, maybe this is the only thing they need to do. But, in general, this will be an adjunct to augment an overall reflux program.”
And that program, both Dr. Silver and Dr. Katz said, should include referral to a dietitian or nutritionist.
“If you’re going to invest time in getting your patients to change their diet, it should be done with professional help, a qualified nutritionist who can work with the patient. This should not be a fad,” Dr. Katz said.
Dr. Silver noted, “In contrast to a physician, the dietitian looks at everything the person is eating. If they’re consuming a lot of simple sugars, I certainly would make that recommendation to reduce, along with other recommendations for GERD. It could be easily incorporated. We included examples in the paper of what we did to achieve that reduction and some things clinicians could focus on.”
One obvious approach, she said, is to advise patients to cut the amount of sugared soda they’re drinking, if not eliminate it entirely.
The study was funded by a VA Merit Award. Dr. Silver has no further disclosures. Dr. Katz is a consultant for Phathom Pharmaceuticals and Sebella Pharmaceuticals and serves on an advisory board for AstraZeneca.
A version of this article first appeared on Medscape.com.
Minimizing dietary consumption of simple sugars may help alleviate symptoms of gastroesophageal reflux disease (GERD), new data suggest.
People who consumed lower quantities of simple sugars experienced significant improvement in total acid exposure time, number of reflux episodes, and subjective reports of heartburn and reflux symptoms, compared with those consuming higher amounts of simple sugars, the authors report.
The authors call their study the first randomized controlled diet intervention trial to investigate both the amount and type of carbohydrate on symptomatic GERD and one of only a few to investigate any type of dietary intervention for GERD.
“There’s really almost no rigorous scientific evidence on any of the foods or ingredients or nutrients that are often recommended to avoid if you have GERD,” corresponding author Heidi J. Silver, RD, PhD, told this news organization. Dr. Silver is research professor of medicine at Vanderbilt University School of Medicine and director of the Vanderbilt Diet, Body Composition, and Human Metabolism Core in Nashville, Tenn.
Even the avoidance of fatty foods, which has been long promoted as part of GERD management, has little evidence to support it.
“With fat, there’s some belief that it may slow down gastric emptying. Therefore, if you had slower gastric emptying, you would have a longer time for the food to put pressure on the lower esophageal sphincter and create an environment for reflux. So, it’s kind of conceptually what is thought but not really tested,” she notes.
The findings were published online in the American Journal of Gastroenterology.
Greatest symptom reduction with low simple carb intake
To test the role of dietary carbohydrates, Dr. Silver and her colleagues randomly assigned 98 U.S. veterans with symptomatic GERD to intake of one of four diets with varying carbohydrate types and amounts for 9 weeks: high total/high simple (the control group), high total/low simple, low total/high simple, or low total/low simple. The total caloric intake was approximately the same for all groups.
At baseline, the average total carbohydrate consumption was 43.7% of calories, and the average simple sugar intake was 116.5 g/d. The two “low-total” groups averaged about a 10% reduction in carbohydrate calories. The “low-simple” groups reduced simple sugars by about half.
There were no changes in body weight in the control group, whereas all three of the other groups lost some weight, ranging from 1.5-2.0 kg (3.3-4.4 lb) despite calorie totals designed for weight maintenance.
There was a significant effect of diet on the two primary outcomes, total esophageal acid exposure time, and total number of reflux episodes, as measured by 24-hour ambulatory pH monitoring. The change in total acid exposure time was significantly greater for the high total/low simple group, compared with the high total/high simple group.
The participants’ ratings of symptoms assessed by the Gastroesophageal Reflux Disease Questionnaire and the GERD Symptom Assessment Scale, including heartburn frequency and severity, pain in throat/chest, and sleep disturbance, improved in all modified diet groups, compared with the control group. The mean degree of improvement in heartburn and regurgitation was twice as great for the modified diets, compared with the controls, and was greatest for the two “low-simple” carb groups.
Dr. Silver and colleagues hypothesize that the differential effects of the diets may relate to the way that dietary carbohydrates are sensed in the gastrointestinal tract after being enzymatically degraded into monosaccharides, possibly affecting lower esophageal tone via the effects of gut-derived hormones including ghrelin and glucagon-like peptide 1 that are secreted in response to macronutrient intake.
Although more data are needed about the effects of carbohydrates in GERD, Dr. Silver advised, “I do think it would be smart for clinicians, when they’re discussing diet, that they bring up the simple sugars. There’s no potential harm in reducing simple sugars. You’re only benefiting yourself in multiple ways. We know that the consumption of simple sugars is extremely excessive, not just in America but worldwide.”
Asked to comment, Philip O. Katz, MD, professor of medicine and director of the GI Function Laboratories at Weill Cornell Medicine, New York, told this news organization that “this is one of the better-designed studies with a lot of care looking at a lot of endpoints that are intriguing and useful.”
“What it says to me is there is potential for nonpharmacologic interventions for GERD that include diet change for helping patients,” he said. “This shows promise for a diet that doesn’t just concentrate on fat or acidic products and is a possible way of augmenting reflux treatment.”
However, Dr. Katz cautioned, “I don’t think anybody should do more with a 9-week study than look at it as good potential.”
“I would tell patients that this is something that they might try, but I wouldn’t make it a rigid requirement based on these data,” he added. “If I were involved in this study, the next thing I would do is transition it to real life and look at compliance to see if results were sustained at 18 weeks or 6 months.”
Diet part of an ‘overall reflux program’
Overall, Dr. Katz, who was the first author of the American College of Gastroenterology’s Clinical Guideline for the Diagnosis and Management of Gastroesophageal Reflux Disease, pointed out that the main nonpharmacologic advice for GERD management includes “Eat smaller meals, don’t eat before bed, don’t lie down after you eat, and reduce any individually known trigger foods.”
Essentially, he views dietary interventions as complementary to medication and other interventions.
“When people really have GERD – not just heartburn – dietary change is an adjunct,” Dr. Katz said. “GERD is a real disease, like diabetes. For some with GERD, maybe this is the only thing they need to do. But, in general, this will be an adjunct to augment an overall reflux program.”
And that program, both Dr. Silver and Dr. Katz said, should include referral to a dietitian or nutritionist.
“If you’re going to invest time in getting your patients to change their diet, it should be done with professional help, a qualified nutritionist who can work with the patient. This should not be a fad,” Dr. Katz said.
Dr. Silver noted, “In contrast to a physician, the dietitian looks at everything the person is eating. If they’re consuming a lot of simple sugars, I certainly would make that recommendation to reduce, along with other recommendations for GERD. It could be easily incorporated. We included examples in the paper of what we did to achieve that reduction and some things clinicians could focus on.”
One obvious approach, she said, is to advise patients to cut the amount of sugared soda they’re drinking, if not eliminate it entirely.
The study was funded by a VA Merit Award. Dr. Silver has no further disclosures. Dr. Katz is a consultant for Phathom Pharmaceuticals and Sebella Pharmaceuticals and serves on an advisory board for AstraZeneca.
A version of this article first appeared on Medscape.com.
Benzbromarone tops febuxostat for gout?
Benzbromarone is not approved in the United States because of concerns of acute liver injury but is approved in several other countries, including China, Brazil, and New Zealand.
“The results suggest that low dosing of benzbromarone may warrant stronger consideration as a safe and effective therapy to achieve serum urate target in gout without moderate chronic kidney disease,” the study team writes.
“Benzbromarone is severely hepatotoxic in some individuals and unlikely to ever gain approval in the United States,” one of the study’s investigators, Robert Terkeltaub, MD, professor of medicine, University of California, San Diego, told this news organization.
However, this study “illustrates the value and impact of uricosuric therapy in general in gout, including potentially as an initial urate-lowering monotherapy strategy, and the sheer number of subjects reaching urate target with low-dose uricosuric monotherapy was impressive,” Dr. Terkeltaub said.
The study was published online in Arthritis & Rheumatology.
“Renal uric acid underexcretion is the chief mechanism driving hyperuricemia in gout, yet the standard urate-lowering therapy recommendation is first-line xanthine oxidase inhibition irrespective of the cause of hyperuricemia,” the study team explains in their article.
Their prospective, randomized, single-center, open-labeled trial was conducted at the Gout Clinic of the Affiliated Hospital of Qingdao University, China.
A total of 196 relatively young healthy men with gout and uric acid underexcretion were randomly assigned to receive low-dose benzbromarone (25 mg/d) or low-dose febuxostat (20 mg/d) for 12 weeks.
Renal uric acid underexcretion was defined as fractional excretion of urate less than 5.5% and uric acid excretion less than or equal to 600 mg/d/1.73 m2.
A “major aspect” of this comparative effectiveness trial was its specific focus on gout-associated renal uric acid underexcretion, where the uricosuric targeted the dominant abnormality promoting the hyperuricemia, Dr. Terkeltaub told this news organization.
In addition, all participants received daily urine alkalinization with oral sodium bicarbonate. “This is not always done in clinical practice, nor in clinical trials of uricosuric agents,” Dr. Terkeltaub said.
The results showed that more participants in the benzbromarone group achieved the serum urate target of less than 6 mg/dL, compared with those in the febuxostat group (primary endpoint, 61% vs. 32%, P < .001).
Adverse events, including gout flares and urolithiasis, did not differ significantly between the two groups, with the exception of more transaminase elevation in the febuxostat group (15% vs. 4%; P = .008).
“We did not find severe hepatotoxicity with low-dose benzbromarone, but ethnic background may affect drug responses, and severe hepatotoxicity of benzbromarone has rarely been reported in Asia,” the authors write.
The incidence of urolithiasis was numerically, but not significantly, higher in the benzbromarone group (5% vs. 2%).
This study found no significant changes in participants’ triglyceride levels, though a previous study suggested febuxostat could increase serum triglycerides.
The investigators caution that the study only included patients who had baseline serum urate levels ranging from 8.0 to 10 mg/dL, who were relatively young and with few comorbidities.
The authors further noted that the “... results may not be generalizable to patients with higher serum urate levels or impaired kidney function, as well [as] patients from other geographical regions, age, and ethnicity groups. The study only included men, and the findings may not be generalizable to women with gout.”
‘Very useful’ in select cases
Weighing in on the results, Valderilio Feijó Azevedo, MD, PhD, adjunct professor of rheumatology, Federal University of Paraná, Brazil, noted that in some specific clinical circumstances, benzbromarone has been “a very useful medication, alone or combined, to treat gout patients.”
“We have great experience with the drug in Brazil. However, it is not used to treat all patients. Patients must be very well-selected in our clinical practice,” Dr. Azevedo said in an interview.
“For most patients, benzbromarone is effective for those who have failed to achieve serum uric acid goals with allopurinol treatment. We do not use it to treat patients with asymptomatic hyperuricemia. In general, we avoid patients with hepatic dysfunction due to previous hepatotoxicity reports. In every patient, we do active monitoring of enzymes,” Dr. Azevedo explained.
“We also avoid using it in patients with severe kidney disease. However, we have used it in some patients with estimated glomerular filtration rate less than 30. We also avoid dosage over 200 mg per day. On average, we use 100 mg per day combined with allopurinol or alone,” said Dr. Azevedo, who was not involved with the study.
Also weighing in, Michael Pillinger, MD, rheumatologist at NYU Langone Health, noted that while benzbromarone is not used in the United States, “in many parts of the world, it is used and is felt to be effective.” Dr. Pillinger was not associated with this current research.
This study, Dr. Pillinger said, “does underline the fact that an alternative drug that lowers urate by promoting urate excretion, if it could gain [U.S. Food and Drug Association] approval and if it were safe, could present a viable new option for therapy.”
He added, “If one conclusion to the study is that determining the basis of hyperuricemia is helpful in guiding benzbromarone use, that implies an additional layer of effort for physicians and patients in a disease that is already notoriously known for patient noncompliance – and in a case where febuxostat and allopurinol will work for both overproducers and underexcreters and would not need this additional assessment.”
The study was sponsored by Shandong Provincial Key Research and Development Plan, the National Natural Science Foundation of China, and Shandong Provincial Science Foundation for Outstanding Youth Scholarship. Dr. Terkeltaub was supported by the National Institutes of Health and the VA Research Service. Dr. Terkeltaub has received research funding from AstraZeneca, and has consulted with Horizon, Selecta, SOBI, Dyve BioSciences, Fortress, AstraZeneca, Allena, Fortress Biotech, and LG Life Sciences. Dr. Azevedo and Dr. Pillinger have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Benzbromarone is not approved in the United States because of concerns of acute liver injury but is approved in several other countries, including China, Brazil, and New Zealand.
“The results suggest that low dosing of benzbromarone may warrant stronger consideration as a safe and effective therapy to achieve serum urate target in gout without moderate chronic kidney disease,” the study team writes.
“Benzbromarone is severely hepatotoxic in some individuals and unlikely to ever gain approval in the United States,” one of the study’s investigators, Robert Terkeltaub, MD, professor of medicine, University of California, San Diego, told this news organization.
However, this study “illustrates the value and impact of uricosuric therapy in general in gout, including potentially as an initial urate-lowering monotherapy strategy, and the sheer number of subjects reaching urate target with low-dose uricosuric monotherapy was impressive,” Dr. Terkeltaub said.
The study was published online in Arthritis & Rheumatology.
“Renal uric acid underexcretion is the chief mechanism driving hyperuricemia in gout, yet the standard urate-lowering therapy recommendation is first-line xanthine oxidase inhibition irrespective of the cause of hyperuricemia,” the study team explains in their article.
Their prospective, randomized, single-center, open-labeled trial was conducted at the Gout Clinic of the Affiliated Hospital of Qingdao University, China.
A total of 196 relatively young healthy men with gout and uric acid underexcretion were randomly assigned to receive low-dose benzbromarone (25 mg/d) or low-dose febuxostat (20 mg/d) for 12 weeks.
Renal uric acid underexcretion was defined as fractional excretion of urate less than 5.5% and uric acid excretion less than or equal to 600 mg/d/1.73 m2.
A “major aspect” of this comparative effectiveness trial was its specific focus on gout-associated renal uric acid underexcretion, where the uricosuric targeted the dominant abnormality promoting the hyperuricemia, Dr. Terkeltaub told this news organization.
In addition, all participants received daily urine alkalinization with oral sodium bicarbonate. “This is not always done in clinical practice, nor in clinical trials of uricosuric agents,” Dr. Terkeltaub said.
The results showed that more participants in the benzbromarone group achieved the serum urate target of less than 6 mg/dL, compared with those in the febuxostat group (primary endpoint, 61% vs. 32%, P < .001).
Adverse events, including gout flares and urolithiasis, did not differ significantly between the two groups, with the exception of more transaminase elevation in the febuxostat group (15% vs. 4%; P = .008).
“We did not find severe hepatotoxicity with low-dose benzbromarone, but ethnic background may affect drug responses, and severe hepatotoxicity of benzbromarone has rarely been reported in Asia,” the authors write.
The incidence of urolithiasis was numerically, but not significantly, higher in the benzbromarone group (5% vs. 2%).
This study found no significant changes in participants’ triglyceride levels, though a previous study suggested febuxostat could increase serum triglycerides.
The investigators caution that the study only included patients who had baseline serum urate levels ranging from 8.0 to 10 mg/dL, who were relatively young and with few comorbidities.
The authors further noted that the “... results may not be generalizable to patients with higher serum urate levels or impaired kidney function, as well [as] patients from other geographical regions, age, and ethnicity groups. The study only included men, and the findings may not be generalizable to women with gout.”
‘Very useful’ in select cases
Weighing in on the results, Valderilio Feijó Azevedo, MD, PhD, adjunct professor of rheumatology, Federal University of Paraná, Brazil, noted that in some specific clinical circumstances, benzbromarone has been “a very useful medication, alone or combined, to treat gout patients.”
“We have great experience with the drug in Brazil. However, it is not used to treat all patients. Patients must be very well-selected in our clinical practice,” Dr. Azevedo said in an interview.
“For most patients, benzbromarone is effective for those who have failed to achieve serum uric acid goals with allopurinol treatment. We do not use it to treat patients with asymptomatic hyperuricemia. In general, we avoid patients with hepatic dysfunction due to previous hepatotoxicity reports. In every patient, we do active monitoring of enzymes,” Dr. Azevedo explained.
“We also avoid using it in patients with severe kidney disease. However, we have used it in some patients with estimated glomerular filtration rate less than 30. We also avoid dosage over 200 mg per day. On average, we use 100 mg per day combined with allopurinol or alone,” said Dr. Azevedo, who was not involved with the study.
Also weighing in, Michael Pillinger, MD, rheumatologist at NYU Langone Health, noted that while benzbromarone is not used in the United States, “in many parts of the world, it is used and is felt to be effective.” Dr. Pillinger was not associated with this current research.
This study, Dr. Pillinger said, “does underline the fact that an alternative drug that lowers urate by promoting urate excretion, if it could gain [U.S. Food and Drug Association] approval and if it were safe, could present a viable new option for therapy.”
He added, “If one conclusion to the study is that determining the basis of hyperuricemia is helpful in guiding benzbromarone use, that implies an additional layer of effort for physicians and patients in a disease that is already notoriously known for patient noncompliance – and in a case where febuxostat and allopurinol will work for both overproducers and underexcreters and would not need this additional assessment.”
The study was sponsored by Shandong Provincial Key Research and Development Plan, the National Natural Science Foundation of China, and Shandong Provincial Science Foundation for Outstanding Youth Scholarship. Dr. Terkeltaub was supported by the National Institutes of Health and the VA Research Service. Dr. Terkeltaub has received research funding from AstraZeneca, and has consulted with Horizon, Selecta, SOBI, Dyve BioSciences, Fortress, AstraZeneca, Allena, Fortress Biotech, and LG Life Sciences. Dr. Azevedo and Dr. Pillinger have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Benzbromarone is not approved in the United States because of concerns of acute liver injury but is approved in several other countries, including China, Brazil, and New Zealand.
“The results suggest that low dosing of benzbromarone may warrant stronger consideration as a safe and effective therapy to achieve serum urate target in gout without moderate chronic kidney disease,” the study team writes.
“Benzbromarone is severely hepatotoxic in some individuals and unlikely to ever gain approval in the United States,” one of the study’s investigators, Robert Terkeltaub, MD, professor of medicine, University of California, San Diego, told this news organization.
However, this study “illustrates the value and impact of uricosuric therapy in general in gout, including potentially as an initial urate-lowering monotherapy strategy, and the sheer number of subjects reaching urate target with low-dose uricosuric monotherapy was impressive,” Dr. Terkeltaub said.
The study was published online in Arthritis & Rheumatology.
“Renal uric acid underexcretion is the chief mechanism driving hyperuricemia in gout, yet the standard urate-lowering therapy recommendation is first-line xanthine oxidase inhibition irrespective of the cause of hyperuricemia,” the study team explains in their article.
Their prospective, randomized, single-center, open-labeled trial was conducted at the Gout Clinic of the Affiliated Hospital of Qingdao University, China.
A total of 196 relatively young healthy men with gout and uric acid underexcretion were randomly assigned to receive low-dose benzbromarone (25 mg/d) or low-dose febuxostat (20 mg/d) for 12 weeks.
Renal uric acid underexcretion was defined as fractional excretion of urate less than 5.5% and uric acid excretion less than or equal to 600 mg/d/1.73 m2.
A “major aspect” of this comparative effectiveness trial was its specific focus on gout-associated renal uric acid underexcretion, where the uricosuric targeted the dominant abnormality promoting the hyperuricemia, Dr. Terkeltaub told this news organization.
In addition, all participants received daily urine alkalinization with oral sodium bicarbonate. “This is not always done in clinical practice, nor in clinical trials of uricosuric agents,” Dr. Terkeltaub said.
The results showed that more participants in the benzbromarone group achieved the serum urate target of less than 6 mg/dL, compared with those in the febuxostat group (primary endpoint, 61% vs. 32%, P < .001).
Adverse events, including gout flares and urolithiasis, did not differ significantly between the two groups, with the exception of more transaminase elevation in the febuxostat group (15% vs. 4%; P = .008).
“We did not find severe hepatotoxicity with low-dose benzbromarone, but ethnic background may affect drug responses, and severe hepatotoxicity of benzbromarone has rarely been reported in Asia,” the authors write.
The incidence of urolithiasis was numerically, but not significantly, higher in the benzbromarone group (5% vs. 2%).
This study found no significant changes in participants’ triglyceride levels, though a previous study suggested febuxostat could increase serum triglycerides.
The investigators caution that the study only included patients who had baseline serum urate levels ranging from 8.0 to 10 mg/dL, who were relatively young and with few comorbidities.
The authors further noted that the “... results may not be generalizable to patients with higher serum urate levels or impaired kidney function, as well [as] patients from other geographical regions, age, and ethnicity groups. The study only included men, and the findings may not be generalizable to women with gout.”
‘Very useful’ in select cases
Weighing in on the results, Valderilio Feijó Azevedo, MD, PhD, adjunct professor of rheumatology, Federal University of Paraná, Brazil, noted that in some specific clinical circumstances, benzbromarone has been “a very useful medication, alone or combined, to treat gout patients.”
“We have great experience with the drug in Brazil. However, it is not used to treat all patients. Patients must be very well-selected in our clinical practice,” Dr. Azevedo said in an interview.
“For most patients, benzbromarone is effective for those who have failed to achieve serum uric acid goals with allopurinol treatment. We do not use it to treat patients with asymptomatic hyperuricemia. In general, we avoid patients with hepatic dysfunction due to previous hepatotoxicity reports. In every patient, we do active monitoring of enzymes,” Dr. Azevedo explained.
“We also avoid using it in patients with severe kidney disease. However, we have used it in some patients with estimated glomerular filtration rate less than 30. We also avoid dosage over 200 mg per day. On average, we use 100 mg per day combined with allopurinol or alone,” said Dr. Azevedo, who was not involved with the study.
Also weighing in, Michael Pillinger, MD, rheumatologist at NYU Langone Health, noted that while benzbromarone is not used in the United States, “in many parts of the world, it is used and is felt to be effective.” Dr. Pillinger was not associated with this current research.
This study, Dr. Pillinger said, “does underline the fact that an alternative drug that lowers urate by promoting urate excretion, if it could gain [U.S. Food and Drug Association] approval and if it were safe, could present a viable new option for therapy.”
He added, “If one conclusion to the study is that determining the basis of hyperuricemia is helpful in guiding benzbromarone use, that implies an additional layer of effort for physicians and patients in a disease that is already notoriously known for patient noncompliance – and in a case where febuxostat and allopurinol will work for both overproducers and underexcreters and would not need this additional assessment.”
The study was sponsored by Shandong Provincial Key Research and Development Plan, the National Natural Science Foundation of China, and Shandong Provincial Science Foundation for Outstanding Youth Scholarship. Dr. Terkeltaub was supported by the National Institutes of Health and the VA Research Service. Dr. Terkeltaub has received research funding from AstraZeneca, and has consulted with Horizon, Selecta, SOBI, Dyve BioSciences, Fortress, AstraZeneca, Allena, Fortress Biotech, and LG Life Sciences. Dr. Azevedo and Dr. Pillinger have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Coming soon: More breathable, more comfortable face masks
Sitting at his desk in Sea Girt, N.J., John Schwind is eager to demonstrate his ReadiMask 365. He holds up what looks like a white sheet of memo paper, peels off a protective liner, and sticks the mask first to his nose. He glides his fingers down his face, over his cheeks, and to his chin, sealing the mask and then demonstrating how easy it is to talk with it in place.
The mask’s medical adhesive sticks directly to the face, without causing breakouts, he said. It doesn’t let air leak and won’t fog his glasses. It’s strapless, so it won’t hurt his ears or make them stick out.
This fall, Mr. Schwind, the CEO of Global Safety First, is hoping to take home $150,000 as one of the two top winners of the federal Mask Innovation Challenge. He has made it to the top 10 but realizes he still has a ton of competition.
After the challenge launched in late 2021, nearly 1,500 submissions were received, says Kumiko Lippold, PhD, a health scientist and manager of the Mask Innovation Challenge. The challenge is run by Dr. Lippold and others at the Division of Research, Innovation, and Ventures (DRIVe), which is part of the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services.
Like the rest of us, Dr. Lippold knows that masks desperately need a makeover. The aim is not only to get us through this pandemic but also future pandemics and other public health emergencies. “We are focused on building masks for the next pandemic, the next wildfires,” she says.
The project is a partnership among BARDA’s DRIVe, the National Institute for Occupational Safety and Health (NIOSH), and the National Institute of Standards and Technology (NIST).
While NIOSH is a partner in the challenge, giving feedback to mask developers, “the mask challenge is entirely separate from the NIOSH approval process,” Dr. Lippold says. Companies can then pursue NIOSH approval on their own, later, if they wish. The agency certifies only masks and respirators.
Preview of masks to come
“We’ve seen some really amazing things,” Dr. Lippold said of the new designs. She didn’t want to play favorites, so she gave an overview of innovations. Some designs have transparent materials, or partially see-through materials, so facial expressions can be read. “We’ve also seen really unique bio-based materials that are derived from natural products. We’ve seen sensors in some.”
One mask model has origami folds, which increase overall surface and breathing area. Some 3D-printed masks promise a custom fit and take into account whether a person’s nose bridge is low or high.
And the finalists are ...
ReadiMask 365: “I can wear this all day long,” Mr. Schwind said of his new design. It has a nano fiber filter and is flexible. Besides the one in the BARDA challenge, the company has other ReadiMasks on the market. “The most important thing is comfort,” he says. “Second is protection. If they don’t feel they have a good seal, users don’t have confidence in the mask.”
He offers various sizes of ReadiMasks, from small sizes designed for women with smaller faces to extra-large, “for NFL linemen.”
ClearMask: “We are the original clear mask,” says Aaron Hsu, CEO and co-founder of ClearMask in Baltimore. The company began in 2017, and the clear design was inspired by a company co-founder who is deaf. She was scheduled to have surgery, and her sign language interpreter did not show up, leaving her to try to communicate in the operating room with masked health care providers. There were no transparent masks available then, Mr. Hsu says.
“Being able to work with BARDA and getting their wisdom is invaluable,” he says.
The makers of ClearMask think masks are here to stay, at least for some. “I think a certain percentage of the population will continue to wear them, regardless,” said Mr. Hsu. He predicts health care settings will become stricter about wearing masks.
“Even now, when you even walk in to a hospital, you might be required to wear a mask,” he says, even as a visitor. His company’s masks are easy to adjust and are secured around the head, so your ears don’t get sore, he says.
4C Air: The BreSafe transparent mask is semi-transparent and is made of a nanomaterial that provides high levels of filtration and breathability with some transparency.
Air99: Based on origami principles, the Airgami mask is meant to improve fit, breathability, and aesthetics over existing masks. “Airgami fits better, works better and looks better,” says Min Xiao, a company spokesperson. “It won’t fall off the nose or collapse onto the mouth, and eyeglasses fog less, she says. Voices are less muffled.” It’s also reusable, rinseable and can be heat disinfected, she says. It went on the market in November 2020.
Air Flo Labs: Flo Mask Pro, like the company’s other designs, conducted over 100 3D facial scans across many ethnicities to produce a better fit, says Kevin Ngo, its creator. For the adult masks, two nose bridge sizes are offered. And users can choose a Pro Filter, with 99% filtration, or an Everyday, which is meant to be much more breathable than other masks. “Our silicone gasket is incredibly soft and gentle on the skin,” Mr. Ngo says. “In addition,we offer indents for glasses, which prevent any fogging.” The company began shipping in May; several thousand masks are in use now, Mr. Ngo said.
Georgetown University: This team’s smart mask is made of metallic foams that can be cleaned and reused.
Levi Strauss: The form of the mask can be made by any basic garment factory. It aims to activate the apparel supply chain as another source of low-cost, high-performance masks.
Matregenix: This mask, made of a transparent nanofiber, allows for easier communication while having high filtration.
SEAL Lab: The SINEW mask stands for Smart, Individualized, Near-Face, Extended Wear. The mask used technology to overcome flaws of traditional respirators, with the same degree of protection. It doesn’t make contact with the skin of the wearer’s face.
StaySafeNow: A team from Harvard University developed Crystal Guard, a reusable, cost-effective clear mask. Its developers say it’s meant to be especially useful for essential workers, teachers, and others who have to communicate to do their work.
Bye-bye N95?
“From our perspective, our goal with the mask challenge was not to replace the N95 respirator,” Dr. Lippold says. N95 masks, which NIOSH certifies, are valuable and protect people in high-risk settings. “With the mask challenge, our goal was really to provide the public with a comparable alternative that really meets their specific level of risk.” Working in a health care setting carries a different risk, she says, than going to the grocery store.
“A common complaint with the N95 is that they are very uncomfortable.” It’s a major barrier to compliance, “and we wanted to address that gap. We didn’t directly compare [the entries] to an N95,” she says, although their testing was similar to NIOSH’s. A number of finalists say they will pursue NIOSH approval, she says.
Meanwhile, some of the finalists’ masks are for sale. Air Flo Labs, for instance, has its Flo Mask Pro for sale online, noting that BARDA allowed it to release the test results from NIOSH and NIST.
Getting from 1,500 to 10
In the first phase of the challenge, Dr. Lippold says, “the goal was to engage as wide an audience as possible.” With the second phase, the bar was set a bit higher. Instead of just submitting ideas on paper, companies had to submit prototypes for lab testing. “We got about 80 submissions,” she says.
Those 80 were whittled down to 10 finalists. Teams had sent prototypes, and experts, including those from NIOSH and NIST, rated them, sometimes looking at multiple copies of the masks. Experts looked at how well the masks filtered the air, how breathable they were, and other data. Once the feedback was given to the mask companies, they entered a redesign period. “Scientists can take this data and basically make these prototypes better,” Dr. Lippold says.
The final round of testing will be in September, and the winners will be announced in the fall. The opportunity allowed companies to have their products go through testing they might not otherwise have been able to get, she says.
A version of this article first appeared on WebMD.com.
Sitting at his desk in Sea Girt, N.J., John Schwind is eager to demonstrate his ReadiMask 365. He holds up what looks like a white sheet of memo paper, peels off a protective liner, and sticks the mask first to his nose. He glides his fingers down his face, over his cheeks, and to his chin, sealing the mask and then demonstrating how easy it is to talk with it in place.
The mask’s medical adhesive sticks directly to the face, without causing breakouts, he said. It doesn’t let air leak and won’t fog his glasses. It’s strapless, so it won’t hurt his ears or make them stick out.
This fall, Mr. Schwind, the CEO of Global Safety First, is hoping to take home $150,000 as one of the two top winners of the federal Mask Innovation Challenge. He has made it to the top 10 but realizes he still has a ton of competition.
After the challenge launched in late 2021, nearly 1,500 submissions were received, says Kumiko Lippold, PhD, a health scientist and manager of the Mask Innovation Challenge. The challenge is run by Dr. Lippold and others at the Division of Research, Innovation, and Ventures (DRIVe), which is part of the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services.
Like the rest of us, Dr. Lippold knows that masks desperately need a makeover. The aim is not only to get us through this pandemic but also future pandemics and other public health emergencies. “We are focused on building masks for the next pandemic, the next wildfires,” she says.
The project is a partnership among BARDA’s DRIVe, the National Institute for Occupational Safety and Health (NIOSH), and the National Institute of Standards and Technology (NIST).
While NIOSH is a partner in the challenge, giving feedback to mask developers, “the mask challenge is entirely separate from the NIOSH approval process,” Dr. Lippold says. Companies can then pursue NIOSH approval on their own, later, if they wish. The agency certifies only masks and respirators.
Preview of masks to come
“We’ve seen some really amazing things,” Dr. Lippold said of the new designs. She didn’t want to play favorites, so she gave an overview of innovations. Some designs have transparent materials, or partially see-through materials, so facial expressions can be read. “We’ve also seen really unique bio-based materials that are derived from natural products. We’ve seen sensors in some.”
One mask model has origami folds, which increase overall surface and breathing area. Some 3D-printed masks promise a custom fit and take into account whether a person’s nose bridge is low or high.
And the finalists are ...
ReadiMask 365: “I can wear this all day long,” Mr. Schwind said of his new design. It has a nano fiber filter and is flexible. Besides the one in the BARDA challenge, the company has other ReadiMasks on the market. “The most important thing is comfort,” he says. “Second is protection. If they don’t feel they have a good seal, users don’t have confidence in the mask.”
He offers various sizes of ReadiMasks, from small sizes designed for women with smaller faces to extra-large, “for NFL linemen.”
ClearMask: “We are the original clear mask,” says Aaron Hsu, CEO and co-founder of ClearMask in Baltimore. The company began in 2017, and the clear design was inspired by a company co-founder who is deaf. She was scheduled to have surgery, and her sign language interpreter did not show up, leaving her to try to communicate in the operating room with masked health care providers. There were no transparent masks available then, Mr. Hsu says.
“Being able to work with BARDA and getting their wisdom is invaluable,” he says.
The makers of ClearMask think masks are here to stay, at least for some. “I think a certain percentage of the population will continue to wear them, regardless,” said Mr. Hsu. He predicts health care settings will become stricter about wearing masks.
“Even now, when you even walk in to a hospital, you might be required to wear a mask,” he says, even as a visitor. His company’s masks are easy to adjust and are secured around the head, so your ears don’t get sore, he says.
4C Air: The BreSafe transparent mask is semi-transparent and is made of a nanomaterial that provides high levels of filtration and breathability with some transparency.
Air99: Based on origami principles, the Airgami mask is meant to improve fit, breathability, and aesthetics over existing masks. “Airgami fits better, works better and looks better,” says Min Xiao, a company spokesperson. “It won’t fall off the nose or collapse onto the mouth, and eyeglasses fog less, she says. Voices are less muffled.” It’s also reusable, rinseable and can be heat disinfected, she says. It went on the market in November 2020.
Air Flo Labs: Flo Mask Pro, like the company’s other designs, conducted over 100 3D facial scans across many ethnicities to produce a better fit, says Kevin Ngo, its creator. For the adult masks, two nose bridge sizes are offered. And users can choose a Pro Filter, with 99% filtration, or an Everyday, which is meant to be much more breathable than other masks. “Our silicone gasket is incredibly soft and gentle on the skin,” Mr. Ngo says. “In addition,we offer indents for glasses, which prevent any fogging.” The company began shipping in May; several thousand masks are in use now, Mr. Ngo said.
Georgetown University: This team’s smart mask is made of metallic foams that can be cleaned and reused.
Levi Strauss: The form of the mask can be made by any basic garment factory. It aims to activate the apparel supply chain as another source of low-cost, high-performance masks.
Matregenix: This mask, made of a transparent nanofiber, allows for easier communication while having high filtration.
SEAL Lab: The SINEW mask stands for Smart, Individualized, Near-Face, Extended Wear. The mask used technology to overcome flaws of traditional respirators, with the same degree of protection. It doesn’t make contact with the skin of the wearer’s face.
StaySafeNow: A team from Harvard University developed Crystal Guard, a reusable, cost-effective clear mask. Its developers say it’s meant to be especially useful for essential workers, teachers, and others who have to communicate to do their work.
Bye-bye N95?
“From our perspective, our goal with the mask challenge was not to replace the N95 respirator,” Dr. Lippold says. N95 masks, which NIOSH certifies, are valuable and protect people in high-risk settings. “With the mask challenge, our goal was really to provide the public with a comparable alternative that really meets their specific level of risk.” Working in a health care setting carries a different risk, she says, than going to the grocery store.
“A common complaint with the N95 is that they are very uncomfortable.” It’s a major barrier to compliance, “and we wanted to address that gap. We didn’t directly compare [the entries] to an N95,” she says, although their testing was similar to NIOSH’s. A number of finalists say they will pursue NIOSH approval, she says.
Meanwhile, some of the finalists’ masks are for sale. Air Flo Labs, for instance, has its Flo Mask Pro for sale online, noting that BARDA allowed it to release the test results from NIOSH and NIST.
Getting from 1,500 to 10
In the first phase of the challenge, Dr. Lippold says, “the goal was to engage as wide an audience as possible.” With the second phase, the bar was set a bit higher. Instead of just submitting ideas on paper, companies had to submit prototypes for lab testing. “We got about 80 submissions,” she says.
Those 80 were whittled down to 10 finalists. Teams had sent prototypes, and experts, including those from NIOSH and NIST, rated them, sometimes looking at multiple copies of the masks. Experts looked at how well the masks filtered the air, how breathable they were, and other data. Once the feedback was given to the mask companies, they entered a redesign period. “Scientists can take this data and basically make these prototypes better,” Dr. Lippold says.
The final round of testing will be in September, and the winners will be announced in the fall. The opportunity allowed companies to have their products go through testing they might not otherwise have been able to get, she says.
A version of this article first appeared on WebMD.com.
Sitting at his desk in Sea Girt, N.J., John Schwind is eager to demonstrate his ReadiMask 365. He holds up what looks like a white sheet of memo paper, peels off a protective liner, and sticks the mask first to his nose. He glides his fingers down his face, over his cheeks, and to his chin, sealing the mask and then demonstrating how easy it is to talk with it in place.
The mask’s medical adhesive sticks directly to the face, without causing breakouts, he said. It doesn’t let air leak and won’t fog his glasses. It’s strapless, so it won’t hurt his ears or make them stick out.
This fall, Mr. Schwind, the CEO of Global Safety First, is hoping to take home $150,000 as one of the two top winners of the federal Mask Innovation Challenge. He has made it to the top 10 but realizes he still has a ton of competition.
After the challenge launched in late 2021, nearly 1,500 submissions were received, says Kumiko Lippold, PhD, a health scientist and manager of the Mask Innovation Challenge. The challenge is run by Dr. Lippold and others at the Division of Research, Innovation, and Ventures (DRIVe), which is part of the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Department of Health & Human Services.
Like the rest of us, Dr. Lippold knows that masks desperately need a makeover. The aim is not only to get us through this pandemic but also future pandemics and other public health emergencies. “We are focused on building masks for the next pandemic, the next wildfires,” she says.
The project is a partnership among BARDA’s DRIVe, the National Institute for Occupational Safety and Health (NIOSH), and the National Institute of Standards and Technology (NIST).
While NIOSH is a partner in the challenge, giving feedback to mask developers, “the mask challenge is entirely separate from the NIOSH approval process,” Dr. Lippold says. Companies can then pursue NIOSH approval on their own, later, if they wish. The agency certifies only masks and respirators.
Preview of masks to come
“We’ve seen some really amazing things,” Dr. Lippold said of the new designs. She didn’t want to play favorites, so she gave an overview of innovations. Some designs have transparent materials, or partially see-through materials, so facial expressions can be read. “We’ve also seen really unique bio-based materials that are derived from natural products. We’ve seen sensors in some.”
One mask model has origami folds, which increase overall surface and breathing area. Some 3D-printed masks promise a custom fit and take into account whether a person’s nose bridge is low or high.
And the finalists are ...
ReadiMask 365: “I can wear this all day long,” Mr. Schwind said of his new design. It has a nano fiber filter and is flexible. Besides the one in the BARDA challenge, the company has other ReadiMasks on the market. “The most important thing is comfort,” he says. “Second is protection. If they don’t feel they have a good seal, users don’t have confidence in the mask.”
He offers various sizes of ReadiMasks, from small sizes designed for women with smaller faces to extra-large, “for NFL linemen.”
ClearMask: “We are the original clear mask,” says Aaron Hsu, CEO and co-founder of ClearMask in Baltimore. The company began in 2017, and the clear design was inspired by a company co-founder who is deaf. She was scheduled to have surgery, and her sign language interpreter did not show up, leaving her to try to communicate in the operating room with masked health care providers. There were no transparent masks available then, Mr. Hsu says.
“Being able to work with BARDA and getting their wisdom is invaluable,” he says.
The makers of ClearMask think masks are here to stay, at least for some. “I think a certain percentage of the population will continue to wear them, regardless,” said Mr. Hsu. He predicts health care settings will become stricter about wearing masks.
“Even now, when you even walk in to a hospital, you might be required to wear a mask,” he says, even as a visitor. His company’s masks are easy to adjust and are secured around the head, so your ears don’t get sore, he says.
4C Air: The BreSafe transparent mask is semi-transparent and is made of a nanomaterial that provides high levels of filtration and breathability with some transparency.
Air99: Based on origami principles, the Airgami mask is meant to improve fit, breathability, and aesthetics over existing masks. “Airgami fits better, works better and looks better,” says Min Xiao, a company spokesperson. “It won’t fall off the nose or collapse onto the mouth, and eyeglasses fog less, she says. Voices are less muffled.” It’s also reusable, rinseable and can be heat disinfected, she says. It went on the market in November 2020.
Air Flo Labs: Flo Mask Pro, like the company’s other designs, conducted over 100 3D facial scans across many ethnicities to produce a better fit, says Kevin Ngo, its creator. For the adult masks, two nose bridge sizes are offered. And users can choose a Pro Filter, with 99% filtration, or an Everyday, which is meant to be much more breathable than other masks. “Our silicone gasket is incredibly soft and gentle on the skin,” Mr. Ngo says. “In addition,we offer indents for glasses, which prevent any fogging.” The company began shipping in May; several thousand masks are in use now, Mr. Ngo said.
Georgetown University: This team’s smart mask is made of metallic foams that can be cleaned and reused.
Levi Strauss: The form of the mask can be made by any basic garment factory. It aims to activate the apparel supply chain as another source of low-cost, high-performance masks.
Matregenix: This mask, made of a transparent nanofiber, allows for easier communication while having high filtration.
SEAL Lab: The SINEW mask stands for Smart, Individualized, Near-Face, Extended Wear. The mask used technology to overcome flaws of traditional respirators, with the same degree of protection. It doesn’t make contact with the skin of the wearer’s face.
StaySafeNow: A team from Harvard University developed Crystal Guard, a reusable, cost-effective clear mask. Its developers say it’s meant to be especially useful for essential workers, teachers, and others who have to communicate to do their work.
Bye-bye N95?
“From our perspective, our goal with the mask challenge was not to replace the N95 respirator,” Dr. Lippold says. N95 masks, which NIOSH certifies, are valuable and protect people in high-risk settings. “With the mask challenge, our goal was really to provide the public with a comparable alternative that really meets their specific level of risk.” Working in a health care setting carries a different risk, she says, than going to the grocery store.
“A common complaint with the N95 is that they are very uncomfortable.” It’s a major barrier to compliance, “and we wanted to address that gap. We didn’t directly compare [the entries] to an N95,” she says, although their testing was similar to NIOSH’s. A number of finalists say they will pursue NIOSH approval, she says.
Meanwhile, some of the finalists’ masks are for sale. Air Flo Labs, for instance, has its Flo Mask Pro for sale online, noting that BARDA allowed it to release the test results from NIOSH and NIST.
Getting from 1,500 to 10
In the first phase of the challenge, Dr. Lippold says, “the goal was to engage as wide an audience as possible.” With the second phase, the bar was set a bit higher. Instead of just submitting ideas on paper, companies had to submit prototypes for lab testing. “We got about 80 submissions,” she says.
Those 80 were whittled down to 10 finalists. Teams had sent prototypes, and experts, including those from NIOSH and NIST, rated them, sometimes looking at multiple copies of the masks. Experts looked at how well the masks filtered the air, how breathable they were, and other data. Once the feedback was given to the mask companies, they entered a redesign period. “Scientists can take this data and basically make these prototypes better,” Dr. Lippold says.
The final round of testing will be in September, and the winners will be announced in the fall. The opportunity allowed companies to have their products go through testing they might not otherwise have been able to get, she says.
A version of this article first appeared on WebMD.com.
The heartache of bereavement can be fatal in heart failure
that points to the need for greater integration of psychosocial risk factors in the treatment of HF.
The adjusted relative risk of dying was nearly 30% higher among bereaved patients with HF (1.29; 95% confidence interval, 1.27-1.30) and slightly higher for those grieving the loss of more than one family member (RR, 1.35).
The highest risk was in the first week after the loss (RR, 1.78) but persisted after 5 years of follow-up (RR, 1.30).
“Heart failure is a very difficult condition and has a very poor prognosis comparable to many, many cancers,” senior author Krisztina László, PhD, Karolinska Institutet, Stockholm, said in an interview. “So it’s important for us to be aware of these increased risks and to understand them better.”
The early risk for death could be related to stress-induced cardiomyopathy, or Takotsubo syndrome, as well as activation of the hypothalamic-pituitary-adrenal axis, the renin-angiotensin-aldosterone system, and sympathetic nervous system, she explained. Higher long-term risks may reflect chronic stress, leading to poorly managed disease and an unhealthy lifestyle.
“If we understand better the underlying mechanisms maybe we can give more specific advice,” Dr. László said. “At this stage, I think having an awareness of the risk and trying to follow patients or at least not let them fall out of usual care, asking questions, trying to understand what their needs are, maybe that is what we can do well.”
A recent position paper by the European Association of Preventive Cardiology pointed out that psychosocial risk factors, like depression and social isolation, can exacerbate heart failure and calls for better integration of psychosocial factors in the treatment of patients with chronic HF.
“We don’t do a very good job of it, but I think they are very important,” observed Stuart D. Russell, MD, a professor of medicine who specializes in advanced HF at Duke University, Durham, N.C., and was not involved in the study.
“When we hear about a spouse dying, we might call and give condolences, but it’s probably a group of patients that for the next 6 months or so we need to watch more closely and see if there are things we can impact both medically as well as socially to perhaps prevent some of this increase in mortality,” he told this news organization.
Although several studies have linked bereavement with adverse health outcomes, this is just one of two studies to look specifically at its role in HF prognosis, Dr. László noted. A 2013 study of 66,000 male veterans reported that widowers had nearly a 38% higher all-cause mortality risk than did married veterans.
The present study extends those findings to 490,527 patients in the Swedish Heart Failure Registry between 2000 and 2018 and/or in the Swedish Patient Register with a primary diagnosis of HF between 1987 and 2018. During a mean follow-up of 3.7 years, 12% of participants had a family member die, and 383,674 participants died.
Results showed the HF mortality risk increased 10% after the death of a child, 20% with the death of a spouse/partner, 13% with a sibling’s death, and 5% with the death of a grandchild.
No increased risk was seen after the death of a parent, which is likely owed to a median patient age of about 75 years and “is in line with our expectations of the life cycle,” Dr. László said.
An association between bereavement and mortality risk was observed in cases of loss caused by cardiovascular disease (RR, 1.34) and other natural causes (RR, 1.27) but also in cases of unnatural deaths, such as suicide (RR, 1.13).
The overall findings were similar regardless of left ventricular ejection fraction and New York Heart Association functional class and were not affected by sex or country of birth.
Dr. Russell agreed that the death of a parent would be expected among these older patients with HF but said that “if the mechanism of this truly is kind of this increased stress hormones and Takotsubo-type mechanism, you’d think it would be worse if it was your kid that died. That shocked me a bit.”
The strong association between mortality and the loss of a spouse or partner was not surprising, given that they’re an important source of mutual social support, he added.
“If it’s a 75-year-old whose spouse dies, we need to make sure that we have the children’s phone number or other people that we can reach out to and say: ‘Can you check on them?’ ” he said. “And we need to make sure that somebody else is coming in with them because I would guess that probably at least half of what patients hear in a clinic visit goes in one ear and out the other and it’s going to make that much better. So we need to find who that new support person is for the patient.”
Asked whether there are efforts underway to incorporate psychosocial factors into current U.S. guidelines, Dr. Russell replied, “certainly within heart failure, I don’t think we’re really discussing it and, that may be the best part of this paper. It really makes us think about a different way of approaching these older patients.”
Dr. László said that future studies are needed to investigate whether less severe sources of stress may also contribute to poor HF prognosis.
“In our population, 12% of patients were affected, which is quite high, but there are patients with heart failure who experience on a daily basis other sources of stress, which are less severe but chronic and affect large numbers,” she said. “This may also have important public health implications and will be an important next step.”
The authors noted that they were unable to eliminate residual confounding by genetic factors or unmeasured socioeconomic-, lifestyle-, or health-related factors shared by family members. Other limitations are limited power to detect a modest effect in some of the subanalyses and that the findings may be generalizable only to countries with social and cultural contexts and health-related factors similar to those of Sweden.
The study was supported by grants from the Swedish Council for Working Life and Social Research, the Karolinska Institutet’s Research Foundation, and the China Scholarship Council. Dr. László is also supported by a grant from the Heart and Lung Foundation. All other authors and Dr. Russell reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
that points to the need for greater integration of psychosocial risk factors in the treatment of HF.
The adjusted relative risk of dying was nearly 30% higher among bereaved patients with HF (1.29; 95% confidence interval, 1.27-1.30) and slightly higher for those grieving the loss of more than one family member (RR, 1.35).
The highest risk was in the first week after the loss (RR, 1.78) but persisted after 5 years of follow-up (RR, 1.30).
“Heart failure is a very difficult condition and has a very poor prognosis comparable to many, many cancers,” senior author Krisztina László, PhD, Karolinska Institutet, Stockholm, said in an interview. “So it’s important for us to be aware of these increased risks and to understand them better.”
The early risk for death could be related to stress-induced cardiomyopathy, or Takotsubo syndrome, as well as activation of the hypothalamic-pituitary-adrenal axis, the renin-angiotensin-aldosterone system, and sympathetic nervous system, she explained. Higher long-term risks may reflect chronic stress, leading to poorly managed disease and an unhealthy lifestyle.
“If we understand better the underlying mechanisms maybe we can give more specific advice,” Dr. László said. “At this stage, I think having an awareness of the risk and trying to follow patients or at least not let them fall out of usual care, asking questions, trying to understand what their needs are, maybe that is what we can do well.”
A recent position paper by the European Association of Preventive Cardiology pointed out that psychosocial risk factors, like depression and social isolation, can exacerbate heart failure and calls for better integration of psychosocial factors in the treatment of patients with chronic HF.
“We don’t do a very good job of it, but I think they are very important,” observed Stuart D. Russell, MD, a professor of medicine who specializes in advanced HF at Duke University, Durham, N.C., and was not involved in the study.
“When we hear about a spouse dying, we might call and give condolences, but it’s probably a group of patients that for the next 6 months or so we need to watch more closely and see if there are things we can impact both medically as well as socially to perhaps prevent some of this increase in mortality,” he told this news organization.
Although several studies have linked bereavement with adverse health outcomes, this is just one of two studies to look specifically at its role in HF prognosis, Dr. László noted. A 2013 study of 66,000 male veterans reported that widowers had nearly a 38% higher all-cause mortality risk than did married veterans.
The present study extends those findings to 490,527 patients in the Swedish Heart Failure Registry between 2000 and 2018 and/or in the Swedish Patient Register with a primary diagnosis of HF between 1987 and 2018. During a mean follow-up of 3.7 years, 12% of participants had a family member die, and 383,674 participants died.
Results showed the HF mortality risk increased 10% after the death of a child, 20% with the death of a spouse/partner, 13% with a sibling’s death, and 5% with the death of a grandchild.
No increased risk was seen after the death of a parent, which is likely owed to a median patient age of about 75 years and “is in line with our expectations of the life cycle,” Dr. László said.
An association between bereavement and mortality risk was observed in cases of loss caused by cardiovascular disease (RR, 1.34) and other natural causes (RR, 1.27) but also in cases of unnatural deaths, such as suicide (RR, 1.13).
The overall findings were similar regardless of left ventricular ejection fraction and New York Heart Association functional class and were not affected by sex or country of birth.
Dr. Russell agreed that the death of a parent would be expected among these older patients with HF but said that “if the mechanism of this truly is kind of this increased stress hormones and Takotsubo-type mechanism, you’d think it would be worse if it was your kid that died. That shocked me a bit.”
The strong association between mortality and the loss of a spouse or partner was not surprising, given that they’re an important source of mutual social support, he added.
“If it’s a 75-year-old whose spouse dies, we need to make sure that we have the children’s phone number or other people that we can reach out to and say: ‘Can you check on them?’ ” he said. “And we need to make sure that somebody else is coming in with them because I would guess that probably at least half of what patients hear in a clinic visit goes in one ear and out the other and it’s going to make that much better. So we need to find who that new support person is for the patient.”
Asked whether there are efforts underway to incorporate psychosocial factors into current U.S. guidelines, Dr. Russell replied, “certainly within heart failure, I don’t think we’re really discussing it and, that may be the best part of this paper. It really makes us think about a different way of approaching these older patients.”
Dr. László said that future studies are needed to investigate whether less severe sources of stress may also contribute to poor HF prognosis.
“In our population, 12% of patients were affected, which is quite high, but there are patients with heart failure who experience on a daily basis other sources of stress, which are less severe but chronic and affect large numbers,” she said. “This may also have important public health implications and will be an important next step.”
The authors noted that they were unable to eliminate residual confounding by genetic factors or unmeasured socioeconomic-, lifestyle-, or health-related factors shared by family members. Other limitations are limited power to detect a modest effect in some of the subanalyses and that the findings may be generalizable only to countries with social and cultural contexts and health-related factors similar to those of Sweden.
The study was supported by grants from the Swedish Council for Working Life and Social Research, the Karolinska Institutet’s Research Foundation, and the China Scholarship Council. Dr. László is also supported by a grant from the Heart and Lung Foundation. All other authors and Dr. Russell reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
that points to the need for greater integration of psychosocial risk factors in the treatment of HF.
The adjusted relative risk of dying was nearly 30% higher among bereaved patients with HF (1.29; 95% confidence interval, 1.27-1.30) and slightly higher for those grieving the loss of more than one family member (RR, 1.35).
The highest risk was in the first week after the loss (RR, 1.78) but persisted after 5 years of follow-up (RR, 1.30).
“Heart failure is a very difficult condition and has a very poor prognosis comparable to many, many cancers,” senior author Krisztina László, PhD, Karolinska Institutet, Stockholm, said in an interview. “So it’s important for us to be aware of these increased risks and to understand them better.”
The early risk for death could be related to stress-induced cardiomyopathy, or Takotsubo syndrome, as well as activation of the hypothalamic-pituitary-adrenal axis, the renin-angiotensin-aldosterone system, and sympathetic nervous system, she explained. Higher long-term risks may reflect chronic stress, leading to poorly managed disease and an unhealthy lifestyle.
“If we understand better the underlying mechanisms maybe we can give more specific advice,” Dr. László said. “At this stage, I think having an awareness of the risk and trying to follow patients or at least not let them fall out of usual care, asking questions, trying to understand what their needs are, maybe that is what we can do well.”
A recent position paper by the European Association of Preventive Cardiology pointed out that psychosocial risk factors, like depression and social isolation, can exacerbate heart failure and calls for better integration of psychosocial factors in the treatment of patients with chronic HF.
“We don’t do a very good job of it, but I think they are very important,” observed Stuart D. Russell, MD, a professor of medicine who specializes in advanced HF at Duke University, Durham, N.C., and was not involved in the study.
“When we hear about a spouse dying, we might call and give condolences, but it’s probably a group of patients that for the next 6 months or so we need to watch more closely and see if there are things we can impact both medically as well as socially to perhaps prevent some of this increase in mortality,” he told this news organization.
Although several studies have linked bereavement with adverse health outcomes, this is just one of two studies to look specifically at its role in HF prognosis, Dr. László noted. A 2013 study of 66,000 male veterans reported that widowers had nearly a 38% higher all-cause mortality risk than did married veterans.
The present study extends those findings to 490,527 patients in the Swedish Heart Failure Registry between 2000 and 2018 and/or in the Swedish Patient Register with a primary diagnosis of HF between 1987 and 2018. During a mean follow-up of 3.7 years, 12% of participants had a family member die, and 383,674 participants died.
Results showed the HF mortality risk increased 10% after the death of a child, 20% with the death of a spouse/partner, 13% with a sibling’s death, and 5% with the death of a grandchild.
No increased risk was seen after the death of a parent, which is likely owed to a median patient age of about 75 years and “is in line with our expectations of the life cycle,” Dr. László said.
An association between bereavement and mortality risk was observed in cases of loss caused by cardiovascular disease (RR, 1.34) and other natural causes (RR, 1.27) but also in cases of unnatural deaths, such as suicide (RR, 1.13).
The overall findings were similar regardless of left ventricular ejection fraction and New York Heart Association functional class and were not affected by sex or country of birth.
Dr. Russell agreed that the death of a parent would be expected among these older patients with HF but said that “if the mechanism of this truly is kind of this increased stress hormones and Takotsubo-type mechanism, you’d think it would be worse if it was your kid that died. That shocked me a bit.”
The strong association between mortality and the loss of a spouse or partner was not surprising, given that they’re an important source of mutual social support, he added.
“If it’s a 75-year-old whose spouse dies, we need to make sure that we have the children’s phone number or other people that we can reach out to and say: ‘Can you check on them?’ ” he said. “And we need to make sure that somebody else is coming in with them because I would guess that probably at least half of what patients hear in a clinic visit goes in one ear and out the other and it’s going to make that much better. So we need to find who that new support person is for the patient.”
Asked whether there are efforts underway to incorporate psychosocial factors into current U.S. guidelines, Dr. Russell replied, “certainly within heart failure, I don’t think we’re really discussing it and, that may be the best part of this paper. It really makes us think about a different way of approaching these older patients.”
Dr. László said that future studies are needed to investigate whether less severe sources of stress may also contribute to poor HF prognosis.
“In our population, 12% of patients were affected, which is quite high, but there are patients with heart failure who experience on a daily basis other sources of stress, which are less severe but chronic and affect large numbers,” she said. “This may also have important public health implications and will be an important next step.”
The authors noted that they were unable to eliminate residual confounding by genetic factors or unmeasured socioeconomic-, lifestyle-, or health-related factors shared by family members. Other limitations are limited power to detect a modest effect in some of the subanalyses and that the findings may be generalizable only to countries with social and cultural contexts and health-related factors similar to those of Sweden.
The study was supported by grants from the Swedish Council for Working Life and Social Research, the Karolinska Institutet’s Research Foundation, and the China Scholarship Council. Dr. László is also supported by a grant from the Heart and Lung Foundation. All other authors and Dr. Russell reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JACC: HEART FAILURE
FDA approves combination pegloticase and methotrexate for refractory gout
Pegloticase, which has been available for 12 years, is a pegylated uric acid specific enzyme that lowers sUA by converting it to allantoin.
Though pegloticase is effective in treating chronic gout in patients refractory to conventional treatment, approximately 92% of patients develop antibodies against the drug, resulting in reduced efficacy.
Based on the immunomodulatory effects of methotrexate, researchers of the randomized, placebo-controlled MIRROR trial sought to determine whether combination treatment of pegloticase with methotrexate (multiple brands) would prevent the development of anti-drug antibodies.
Findings from the phase 4 trial found that co-administration of pegloticase and methotrexate reduced the formation of new anti-PEG antibodies. In the group receiving methotrexate and pegloticase, 23.2% (22 out of 95) of patients had an increase in anti-PEG antibodies, compared with 50% (24 of 48) in the pegloticase plus placebo group, according to a recent company press release.
Nearly three-quarters (71%) of participants in the group pretreated with methotrexate, followed by combination pegloticase-methotrexate, had sUA levels that dopped to below 6 mg/dL during the 52-week study. By comparison, 38.5% of participants in the pegloticase and placebo group reached the endpoint. Though gout flare occurred in both groups, methotrexate did not appear to increase the risk for adverse events or gout flare.
The study, led by John Botson, MD, RPh, CCD, a rheumatologist in Anchorage, Alaska, concluded that these measurements demonstrated a significant improvement from traditional pegloticase-only treatment of gout. “This trial confirms not only improved efficacy but improved safety in patients treated with pegloticase in combination with methotrexate 15 mg orally once weekly,” Dr. Botson said last month in an interview with this news organization.
The study was funded by Horizon. Dr. Botson reports receiving research support from Horizon and Radius Health and speaker fees from AbbVie, Amgen, Aurinia, ChemoCentryx, Horizon, Eli Lilly, and Novartis.
A version of this article first appeared on Medscape.com.
Pegloticase, which has been available for 12 years, is a pegylated uric acid specific enzyme that lowers sUA by converting it to allantoin.
Though pegloticase is effective in treating chronic gout in patients refractory to conventional treatment, approximately 92% of patients develop antibodies against the drug, resulting in reduced efficacy.
Based on the immunomodulatory effects of methotrexate, researchers of the randomized, placebo-controlled MIRROR trial sought to determine whether combination treatment of pegloticase with methotrexate (multiple brands) would prevent the development of anti-drug antibodies.
Findings from the phase 4 trial found that co-administration of pegloticase and methotrexate reduced the formation of new anti-PEG antibodies. In the group receiving methotrexate and pegloticase, 23.2% (22 out of 95) of patients had an increase in anti-PEG antibodies, compared with 50% (24 of 48) in the pegloticase plus placebo group, according to a recent company press release.
Nearly three-quarters (71%) of participants in the group pretreated with methotrexate, followed by combination pegloticase-methotrexate, had sUA levels that dopped to below 6 mg/dL during the 52-week study. By comparison, 38.5% of participants in the pegloticase and placebo group reached the endpoint. Though gout flare occurred in both groups, methotrexate did not appear to increase the risk for adverse events or gout flare.
The study, led by John Botson, MD, RPh, CCD, a rheumatologist in Anchorage, Alaska, concluded that these measurements demonstrated a significant improvement from traditional pegloticase-only treatment of gout. “This trial confirms not only improved efficacy but improved safety in patients treated with pegloticase in combination with methotrexate 15 mg orally once weekly,” Dr. Botson said last month in an interview with this news organization.
The study was funded by Horizon. Dr. Botson reports receiving research support from Horizon and Radius Health and speaker fees from AbbVie, Amgen, Aurinia, ChemoCentryx, Horizon, Eli Lilly, and Novartis.
A version of this article first appeared on Medscape.com.
Pegloticase, which has been available for 12 years, is a pegylated uric acid specific enzyme that lowers sUA by converting it to allantoin.
Though pegloticase is effective in treating chronic gout in patients refractory to conventional treatment, approximately 92% of patients develop antibodies against the drug, resulting in reduced efficacy.
Based on the immunomodulatory effects of methotrexate, researchers of the randomized, placebo-controlled MIRROR trial sought to determine whether combination treatment of pegloticase with methotrexate (multiple brands) would prevent the development of anti-drug antibodies.
Findings from the phase 4 trial found that co-administration of pegloticase and methotrexate reduced the formation of new anti-PEG antibodies. In the group receiving methotrexate and pegloticase, 23.2% (22 out of 95) of patients had an increase in anti-PEG antibodies, compared with 50% (24 of 48) in the pegloticase plus placebo group, according to a recent company press release.
Nearly three-quarters (71%) of participants in the group pretreated with methotrexate, followed by combination pegloticase-methotrexate, had sUA levels that dopped to below 6 mg/dL during the 52-week study. By comparison, 38.5% of participants in the pegloticase and placebo group reached the endpoint. Though gout flare occurred in both groups, methotrexate did not appear to increase the risk for adverse events or gout flare.
The study, led by John Botson, MD, RPh, CCD, a rheumatologist in Anchorage, Alaska, concluded that these measurements demonstrated a significant improvement from traditional pegloticase-only treatment of gout. “This trial confirms not only improved efficacy but improved safety in patients treated with pegloticase in combination with methotrexate 15 mg orally once weekly,” Dr. Botson said last month in an interview with this news organization.
The study was funded by Horizon. Dr. Botson reports receiving research support from Horizon and Radius Health and speaker fees from AbbVie, Amgen, Aurinia, ChemoCentryx, Horizon, Eli Lilly, and Novartis.
A version of this article first appeared on Medscape.com.
Children and COVID: Vaccination a harder sell in the summer
The COVID-19 vaccination effort in the youngest children has begun much more slowly than the most recent rollout for older children, according to the Centers for Disease Control and Prevention.
in early November of 2021, based on CDC data last updated on July 7.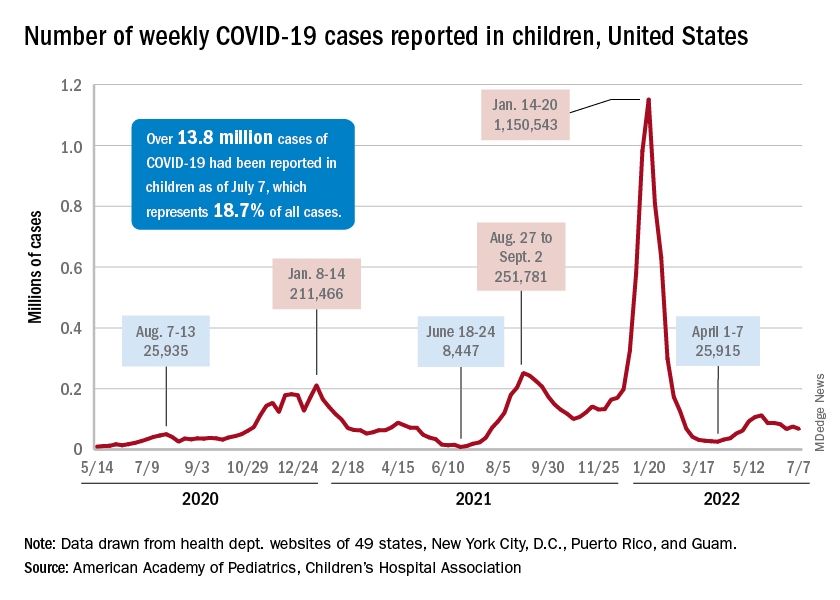
That approval, of course, came between the Delta and Omicron surges, when awareness was higher. The low initial uptake among those under age 5, however, was not unexpected by the Biden administration. “That number in and of itself is very much in line with our expectation, and we’re eager to continue working closely with partners to build on this start,” a senior administration official told ABC News.
With approval of the vaccine occurring after the school year was over, parents’ thoughts have been focused more on vacations and less on vaccinations. “Even before these vaccines officially became available, this was going to be a different rollout; it was going to take more time,” the official explained.
Incidence measures continue on different paths
New COVID-19 cases dropped during the latest reporting week (July 1-7), returning to the downward trend that began in late May and then stopped for 1 week (June 24-30), when cases were up by 12.4%, according to the American Academy of Pediatrics and the Children’s Hospital Association.
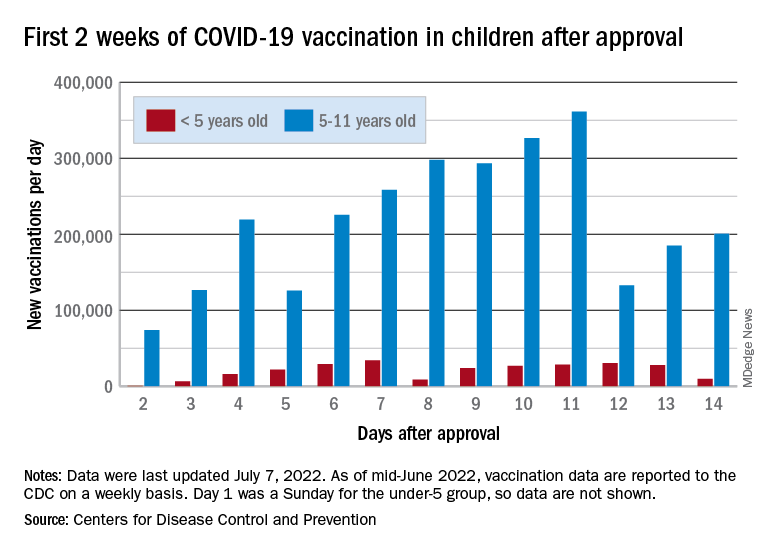
Children also represent a smaller share of cases, probably because of underreporting. “There has been a notable decline in the portion of reported weekly COVID-19 cases that are children,” the two groups said in their weekly COVID report. Although “cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases.”
Other measures, however, have been rising slowly but steadily since the spring. New admissions of patients aged 0-17 years with confirmed COVID, which were down to 0.13 per 100,000 population in early April, had climbed to 0.39 per 100,000 by July 7, the CDC said on its COVID Data Tracker.
Emergency department visits continue to show the same upward trend, despite a small decline in early June. A COVID diagnosis was involved in just 0.5% of ED visits in children aged 0-11 years on March 26, but by July 6 the rate was 4.7%. Increases were not as high among older children: From 0.3% on March 26 to 2.5% on July 6 for those aged 12-15 and from 0.3% to 2.4% for 16- and 17-year-olds, according to the CDC.
The COVID-19 vaccination effort in the youngest children has begun much more slowly than the most recent rollout for older children, according to the Centers for Disease Control and Prevention.
in early November of 2021, based on CDC data last updated on July 7.
That approval, of course, came between the Delta and Omicron surges, when awareness was higher. The low initial uptake among those under age 5, however, was not unexpected by the Biden administration. “That number in and of itself is very much in line with our expectation, and we’re eager to continue working closely with partners to build on this start,” a senior administration official told ABC News.
With approval of the vaccine occurring after the school year was over, parents’ thoughts have been focused more on vacations and less on vaccinations. “Even before these vaccines officially became available, this was going to be a different rollout; it was going to take more time,” the official explained.
Incidence measures continue on different paths
New COVID-19 cases dropped during the latest reporting week (July 1-7), returning to the downward trend that began in late May and then stopped for 1 week (June 24-30), when cases were up by 12.4%, according to the American Academy of Pediatrics and the Children’s Hospital Association.

Children also represent a smaller share of cases, probably because of underreporting. “There has been a notable decline in the portion of reported weekly COVID-19 cases that are children,” the two groups said in their weekly COVID report. Although “cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases.”
Other measures, however, have been rising slowly but steadily since the spring. New admissions of patients aged 0-17 years with confirmed COVID, which were down to 0.13 per 100,000 population in early April, had climbed to 0.39 per 100,000 by July 7, the CDC said on its COVID Data Tracker.
Emergency department visits continue to show the same upward trend, despite a small decline in early June. A COVID diagnosis was involved in just 0.5% of ED visits in children aged 0-11 years on March 26, but by July 6 the rate was 4.7%. Increases were not as high among older children: From 0.3% on March 26 to 2.5% on July 6 for those aged 12-15 and from 0.3% to 2.4% for 16- and 17-year-olds, according to the CDC.
The COVID-19 vaccination effort in the youngest children has begun much more slowly than the most recent rollout for older children, according to the Centers for Disease Control and Prevention.
in early November of 2021, based on CDC data last updated on July 7.
That approval, of course, came between the Delta and Omicron surges, when awareness was higher. The low initial uptake among those under age 5, however, was not unexpected by the Biden administration. “That number in and of itself is very much in line with our expectation, and we’re eager to continue working closely with partners to build on this start,” a senior administration official told ABC News.
With approval of the vaccine occurring after the school year was over, parents’ thoughts have been focused more on vacations and less on vaccinations. “Even before these vaccines officially became available, this was going to be a different rollout; it was going to take more time,” the official explained.
Incidence measures continue on different paths
New COVID-19 cases dropped during the latest reporting week (July 1-7), returning to the downward trend that began in late May and then stopped for 1 week (June 24-30), when cases were up by 12.4%, according to the American Academy of Pediatrics and the Children’s Hospital Association.

Children also represent a smaller share of cases, probably because of underreporting. “There has been a notable decline in the portion of reported weekly COVID-19 cases that are children,” the two groups said in their weekly COVID report. Although “cases are likely increasingly underreported for all age groups, this decline indicates that children are disproportionately undercounted in reported COVID-19 cases.”
Other measures, however, have been rising slowly but steadily since the spring. New admissions of patients aged 0-17 years with confirmed COVID, which were down to 0.13 per 100,000 population in early April, had climbed to 0.39 per 100,000 by July 7, the CDC said on its COVID Data Tracker.
Emergency department visits continue to show the same upward trend, despite a small decline in early June. A COVID diagnosis was involved in just 0.5% of ED visits in children aged 0-11 years on March 26, but by July 6 the rate was 4.7%. Increases were not as high among older children: From 0.3% on March 26 to 2.5% on July 6 for those aged 12-15 and from 0.3% to 2.4% for 16- and 17-year-olds, according to the CDC.
Childhood type 1 diabetes tests suggested at ages 2 and 6
, new data suggest.
Both genetic screening and islet-cell autoantibody screening for type 1 diabetes risk have become less expensive in recent years. Nonetheless, as of now, most children who receive such screening do so through programs that screen relatives of people who already have the condition, such as the global TrialNet program.
Some in the type 1 diabetes field have urged wider screening, with the rationale that knowledge of increased risk can prepare families to recognize the early signs of hyperglycemia and seek medical help to prevent the onset of diabetic ketoacidosis.
Moreover, potential therapies to prevent or delay type 1 diabetes are currently in development, including the anti-CD3 monoclonal antibody teplizumab (Tzield, Provention Bio).
However, given that the incidence of type 1 diabetes is about 1 in 300 children, any population-wide screening program would need to be implemented in the most efficient and cost-effective way possible with limited numbers of tests, say Mohamed Ghalwash, PhD, of the Center for Computational Health, IBM Research, Yorktown Heights, N.Y., and colleagues.
Results from their analysis of nearly 25,000 children from five prospective cohorts in Europe and the United States were published online in Lancet Diabetes & Endocrinology.
Screening in kids feasible, but may need geographic tweaking
“Our results show that initial screening for islet autoantibodies at two ages (2 years and 6 years) is sensitive and efficient for public health translation but might require adjustment by country on the basis of population-specific disease characteristics,” Dr. Ghalwash and colleagues write.
In an accompanying editorial, pediatric endocrinologist Maria J. Redondo, MD, PhD, writes: “This study is timely because recent successes in preventing type 1 diabetes highlight the need to identify the best candidates for intervention ... This paper constitutes an important contribution to the literature.”
However, Dr. Redondo, of Baylor College of Medicine and Texas Children’s Hospital, Houston, also cautioned: “It remains to be seen whether Dr. Ghalwash and colleagues’ strategy could work in the general population, because all the participants in the combined dataset had genetic risk factors for the disease or a relative with type 1 diabetes, in whom performance is expected to be higher.”
She also noted that most participants were of northern European ancestry and that it is unknown whether the same or a similar screening strategy could be applied to individuals older than 15 years, in whom preclinical type 1 diabetes progresses more slowly.
Two-time childhood screening yielded high sensitivity, specificity
The data from a total of 24,662 participants were pooled from five prospective cohorts from Finland (DIPP), Germany (BABYDIAB), Sweden (DiPiS), and the United States (DAISY and DEW-IT).
All were at elevated risk for type 1 diabetes based on human leukocyte antigen (HLA) genotyping, and some had first-degree relatives with the condition. Participants were screened annually for three type 1 diabetes–associated autoantibodies up to age 15 years or the onset of type 1 diabetes.
During follow-up, 672 children developed type 1 diabetes by age 15 years and 6,050 did not. (The rest hadn’t yet reached age 15 years or type 1 diabetes onset.) The median age at first appearance of islet autoantibodies was 4.5 years.
A two-age screening strategy at 2 years and 6 years was more sensitive than screening at just one age, with a sensitivity of 82% and a positive predictive value of 79% for the development of type 1 diabetes by age 15 years.
The predictive value increased with the number of autoantibodies tested. For example, a single islet autoantibody at age 2 years indicated a 4-year risk of developing type 1 diabetes by age 5.99 years of 31%, while multiple antibody positivity at age 2 years carried a 4-year risk of 55%.
By age 6 years, the risk over the next 9 years was 39% if the test had been negative at age 2 years and 70% if the test had been positive at 2 years. But overall, a 6-year-old with multiple autoantibodies had an overall 83% risk of type 1 diabetes regardless of the test result at 2 years.
The predictive performance of sensitivity by age differed by country, suggesting that the optimal ages for autoantibody testing might differ by geographic region, Dr. Ghalwash and colleagues say.
Dr. Redondo commented, “The model might require adaptation to local factors that affect the progression and prevalence of type 1 diabetes.” And, she added, “important aspects, such as screening cost, global access, acceptability, and follow-up support will need to be addressed for this strategy to be a viable public health option.”
The study was funded by JDRF. Dr. Ghalwash and another author are employees of IBM. A third author was a JDRF employee when the research was done and is now an employee of Janssen Research and Development. Dr. Redondo has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new data suggest.
Both genetic screening and islet-cell autoantibody screening for type 1 diabetes risk have become less expensive in recent years. Nonetheless, as of now, most children who receive such screening do so through programs that screen relatives of people who already have the condition, such as the global TrialNet program.
Some in the type 1 diabetes field have urged wider screening, with the rationale that knowledge of increased risk can prepare families to recognize the early signs of hyperglycemia and seek medical help to prevent the onset of diabetic ketoacidosis.
Moreover, potential therapies to prevent or delay type 1 diabetes are currently in development, including the anti-CD3 monoclonal antibody teplizumab (Tzield, Provention Bio).
However, given that the incidence of type 1 diabetes is about 1 in 300 children, any population-wide screening program would need to be implemented in the most efficient and cost-effective way possible with limited numbers of tests, say Mohamed Ghalwash, PhD, of the Center for Computational Health, IBM Research, Yorktown Heights, N.Y., and colleagues.
Results from their analysis of nearly 25,000 children from five prospective cohorts in Europe and the United States were published online in Lancet Diabetes & Endocrinology.
Screening in kids feasible, but may need geographic tweaking
“Our results show that initial screening for islet autoantibodies at two ages (2 years and 6 years) is sensitive and efficient for public health translation but might require adjustment by country on the basis of population-specific disease characteristics,” Dr. Ghalwash and colleagues write.
In an accompanying editorial, pediatric endocrinologist Maria J. Redondo, MD, PhD, writes: “This study is timely because recent successes in preventing type 1 diabetes highlight the need to identify the best candidates for intervention ... This paper constitutes an important contribution to the literature.”
However, Dr. Redondo, of Baylor College of Medicine and Texas Children’s Hospital, Houston, also cautioned: “It remains to be seen whether Dr. Ghalwash and colleagues’ strategy could work in the general population, because all the participants in the combined dataset had genetic risk factors for the disease or a relative with type 1 diabetes, in whom performance is expected to be higher.”
She also noted that most participants were of northern European ancestry and that it is unknown whether the same or a similar screening strategy could be applied to individuals older than 15 years, in whom preclinical type 1 diabetes progresses more slowly.
Two-time childhood screening yielded high sensitivity, specificity
The data from a total of 24,662 participants were pooled from five prospective cohorts from Finland (DIPP), Germany (BABYDIAB), Sweden (DiPiS), and the United States (DAISY and DEW-IT).
All were at elevated risk for type 1 diabetes based on human leukocyte antigen (HLA) genotyping, and some had first-degree relatives with the condition. Participants were screened annually for three type 1 diabetes–associated autoantibodies up to age 15 years or the onset of type 1 diabetes.
During follow-up, 672 children developed type 1 diabetes by age 15 years and 6,050 did not. (The rest hadn’t yet reached age 15 years or type 1 diabetes onset.) The median age at first appearance of islet autoantibodies was 4.5 years.
A two-age screening strategy at 2 years and 6 years was more sensitive than screening at just one age, with a sensitivity of 82% and a positive predictive value of 79% for the development of type 1 diabetes by age 15 years.
The predictive value increased with the number of autoantibodies tested. For example, a single islet autoantibody at age 2 years indicated a 4-year risk of developing type 1 diabetes by age 5.99 years of 31%, while multiple antibody positivity at age 2 years carried a 4-year risk of 55%.
By age 6 years, the risk over the next 9 years was 39% if the test had been negative at age 2 years and 70% if the test had been positive at 2 years. But overall, a 6-year-old with multiple autoantibodies had an overall 83% risk of type 1 diabetes regardless of the test result at 2 years.
The predictive performance of sensitivity by age differed by country, suggesting that the optimal ages for autoantibody testing might differ by geographic region, Dr. Ghalwash and colleagues say.
Dr. Redondo commented, “The model might require adaptation to local factors that affect the progression and prevalence of type 1 diabetes.” And, she added, “important aspects, such as screening cost, global access, acceptability, and follow-up support will need to be addressed for this strategy to be a viable public health option.”
The study was funded by JDRF. Dr. Ghalwash and another author are employees of IBM. A third author was a JDRF employee when the research was done and is now an employee of Janssen Research and Development. Dr. Redondo has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new data suggest.
Both genetic screening and islet-cell autoantibody screening for type 1 diabetes risk have become less expensive in recent years. Nonetheless, as of now, most children who receive such screening do so through programs that screen relatives of people who already have the condition, such as the global TrialNet program.
Some in the type 1 diabetes field have urged wider screening, with the rationale that knowledge of increased risk can prepare families to recognize the early signs of hyperglycemia and seek medical help to prevent the onset of diabetic ketoacidosis.
Moreover, potential therapies to prevent or delay type 1 diabetes are currently in development, including the anti-CD3 monoclonal antibody teplizumab (Tzield, Provention Bio).
However, given that the incidence of type 1 diabetes is about 1 in 300 children, any population-wide screening program would need to be implemented in the most efficient and cost-effective way possible with limited numbers of tests, say Mohamed Ghalwash, PhD, of the Center for Computational Health, IBM Research, Yorktown Heights, N.Y., and colleagues.
Results from their analysis of nearly 25,000 children from five prospective cohorts in Europe and the United States were published online in Lancet Diabetes & Endocrinology.
Screening in kids feasible, but may need geographic tweaking
“Our results show that initial screening for islet autoantibodies at two ages (2 years and 6 years) is sensitive and efficient for public health translation but might require adjustment by country on the basis of population-specific disease characteristics,” Dr. Ghalwash and colleagues write.
In an accompanying editorial, pediatric endocrinologist Maria J. Redondo, MD, PhD, writes: “This study is timely because recent successes in preventing type 1 diabetes highlight the need to identify the best candidates for intervention ... This paper constitutes an important contribution to the literature.”
However, Dr. Redondo, of Baylor College of Medicine and Texas Children’s Hospital, Houston, also cautioned: “It remains to be seen whether Dr. Ghalwash and colleagues’ strategy could work in the general population, because all the participants in the combined dataset had genetic risk factors for the disease or a relative with type 1 diabetes, in whom performance is expected to be higher.”
She also noted that most participants were of northern European ancestry and that it is unknown whether the same or a similar screening strategy could be applied to individuals older than 15 years, in whom preclinical type 1 diabetes progresses more slowly.
Two-time childhood screening yielded high sensitivity, specificity
The data from a total of 24,662 participants were pooled from five prospective cohorts from Finland (DIPP), Germany (BABYDIAB), Sweden (DiPiS), and the United States (DAISY and DEW-IT).
All were at elevated risk for type 1 diabetes based on human leukocyte antigen (HLA) genotyping, and some had first-degree relatives with the condition. Participants were screened annually for three type 1 diabetes–associated autoantibodies up to age 15 years or the onset of type 1 diabetes.
During follow-up, 672 children developed type 1 diabetes by age 15 years and 6,050 did not. (The rest hadn’t yet reached age 15 years or type 1 diabetes onset.) The median age at first appearance of islet autoantibodies was 4.5 years.
A two-age screening strategy at 2 years and 6 years was more sensitive than screening at just one age, with a sensitivity of 82% and a positive predictive value of 79% for the development of type 1 diabetes by age 15 years.
The predictive value increased with the number of autoantibodies tested. For example, a single islet autoantibody at age 2 years indicated a 4-year risk of developing type 1 diabetes by age 5.99 years of 31%, while multiple antibody positivity at age 2 years carried a 4-year risk of 55%.
By age 6 years, the risk over the next 9 years was 39% if the test had been negative at age 2 years and 70% if the test had been positive at 2 years. But overall, a 6-year-old with multiple autoantibodies had an overall 83% risk of type 1 diabetes regardless of the test result at 2 years.
The predictive performance of sensitivity by age differed by country, suggesting that the optimal ages for autoantibody testing might differ by geographic region, Dr. Ghalwash and colleagues say.
Dr. Redondo commented, “The model might require adaptation to local factors that affect the progression and prevalence of type 1 diabetes.” And, she added, “important aspects, such as screening cost, global access, acceptability, and follow-up support will need to be addressed for this strategy to be a viable public health option.”
The study was funded by JDRF. Dr. Ghalwash and another author are employees of IBM. A third author was a JDRF employee when the research was done and is now an employee of Janssen Research and Development. Dr. Redondo has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM LANCET DIABETES & ENDOCRINOLOGY
BA.4 and BA.5 subvariants are more evasive of antibodies, but not of cellular immunity
The picture around the BA.4 and BA.5 subvariants of Omicron has been really confusing in that the pair is driving up cases but global COVID-19 deaths remain at their lowest level since the beginning of the pandemic. Explaining the two components of the immune response – antibodies versus cellular immune responses – can help us understand where we are in the pandemic and future booster options.
These two subvariants of Omicron, as of July 5, make up more than half of the COVID-19 strains in the United States and are expected to keep increasing. One of two reasons can lead to a variant or subvariant becoming dominant strain: increased transmissibility or evasion of antibodies.
Although BA.4 and BA.5 could be more transmissible than other subvariants of Omicron (which is already very transmissible), this has not yet been established in experiments showing increased affinity for the human receptor or in animal models. What we do know is that BA.4 and BA.5 seem to evade neutralizing antibodies conferred by the vaccines or even prior BA.1 infection (an earlier subvariant of Omicron), which could be the reason we are seeing so many reinfections now. Of note, BA.1 infection conferred antibodies that protected against subsequent BA.2 infection, so we did not see the same spike in cases in the United States with BA.2 (after a large BA.1 spike over the winter) earlier this spring.
Okay, so isn’t evasion of antibodies a bad thing? Of course it is but, luckily, our immune system is “redundant” and doesn›t just rely on antibodies to protect us from infection. In fact, antibodies (such as IgA, which is the mucosal antibody most prevalent in the nose and mouth, and IgG, which is the most prevalent antibody in the bloodstream) are our first line of COVID-19 defense in the nasal mucosa. Therefore, mild upper respiratory infections will be common as BA.4/BA.5 evade our nasal antibodies. Luckily, the rate of severe disease is remaining low throughout the world, probably because of the high amounts of cellular immunity to the virus. B and T cells are our protectors from severe disease.
For instance, two-dose vaccines are still conferring high rates of protection from severe disease with the BA.4 and BA.5 variants, with 87% protection against hospitalization per South Africa data. This is probably attributable to the fact that T-cell immunity from the vaccines remains protective across variants “from Alpha to Omicron,” as described by a recent and elegant paper.
Data from Qatar show that natural infection (even occurring up to 14 months ago) remains very protective (97.3%) against severe disease with the current circulating subvariants, including BA.4 and BA.5. Again, this is probably attributable to T cells which specifically amplify in response to a piece of the virus and help recruit cells to attack the pathogen directly.
The original BA.1 subvariant of Omicron has 26-32 mutations along its spike protein that differ from the “ancestral strain,” and BA.4 and BA.5 variants have a few more. Our T-cell response, even across a mutated spike protein, is so robust that we have not seen Omicron yet able to evade the many T cells (which we produce from the vaccines or infection) that descend upon the mutated virus to fight severe disease. Antibody-producing memory B cells, generated by the vaccines (or prior infection), have been shown to actually adapt their immune response to the variant to which they are exposed.
Therefore, the story of the BA.4 and BA.5 subvariants seems to remain about antibodies vs. cellular immunity. Our immunity in the United States is growing and is from both vaccination and natural infection, with 78.3% of the population having had at least one dose of the vaccine and at least 60% of adults (and 75% of children 0-18) having been exposed to the virus by February 2022, per the Centers for Disease Control and Prevention (with exposure probably much higher now in July 2022 after subsequent Omicron subvariants waves).
So, what about Omicron-specific boosters? A booster shot will just raise antibodies temporarily, but their effectiveness wanes several months later. Moreover, a booster shot against the ancestral strain is not very effective in neutralizing BA.4 and BA.5 (with a prior BA.1 Omicron infection being more effective than a booster). Luckily, Pfizer has promised a BA.4/BA.5-specific mRNA vaccine by October, and Moderna has promised a bivalent vaccine containing BA.4/BA.5 mRNA sequences around the same time. A vaccine that specifically increases antibodies against the most prevalent circulating strain should be important as a booster for those who are predisposed to severe breakthrough infections (for example, those with immunocompromise or older individuals with multiple comorbidities). Moreover, BA.4/BA.5–specific booster vaccines may help prevent mild infections for many individuals. Finally, any booster (or exposure) should diversify and broaden T-cell responses to the virus, and a booster shot will also expand the potency of B cells, making them better able to respond to the newest subvariants as we continue to live with COVID-19.
Monica Gandhi, MD, MPH, is an infectious diseases doctor, professor of medicine, and associate chief in the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco.
A version of this article first appeared on Medscape.com.
The picture around the BA.4 and BA.5 subvariants of Omicron has been really confusing in that the pair is driving up cases but global COVID-19 deaths remain at their lowest level since the beginning of the pandemic. Explaining the two components of the immune response – antibodies versus cellular immune responses – can help us understand where we are in the pandemic and future booster options.
These two subvariants of Omicron, as of July 5, make up more than half of the COVID-19 strains in the United States and are expected to keep increasing. One of two reasons can lead to a variant or subvariant becoming dominant strain: increased transmissibility or evasion of antibodies.
Although BA.4 and BA.5 could be more transmissible than other subvariants of Omicron (which is already very transmissible), this has not yet been established in experiments showing increased affinity for the human receptor or in animal models. What we do know is that BA.4 and BA.5 seem to evade neutralizing antibodies conferred by the vaccines or even prior BA.1 infection (an earlier subvariant of Omicron), which could be the reason we are seeing so many reinfections now. Of note, BA.1 infection conferred antibodies that protected against subsequent BA.2 infection, so we did not see the same spike in cases in the United States with BA.2 (after a large BA.1 spike over the winter) earlier this spring.
Okay, so isn’t evasion of antibodies a bad thing? Of course it is but, luckily, our immune system is “redundant” and doesn›t just rely on antibodies to protect us from infection. In fact, antibodies (such as IgA, which is the mucosal antibody most prevalent in the nose and mouth, and IgG, which is the most prevalent antibody in the bloodstream) are our first line of COVID-19 defense in the nasal mucosa. Therefore, mild upper respiratory infections will be common as BA.4/BA.5 evade our nasal antibodies. Luckily, the rate of severe disease is remaining low throughout the world, probably because of the high amounts of cellular immunity to the virus. B and T cells are our protectors from severe disease.
For instance, two-dose vaccines are still conferring high rates of protection from severe disease with the BA.4 and BA.5 variants, with 87% protection against hospitalization per South Africa data. This is probably attributable to the fact that T-cell immunity from the vaccines remains protective across variants “from Alpha to Omicron,” as described by a recent and elegant paper.
Data from Qatar show that natural infection (even occurring up to 14 months ago) remains very protective (97.3%) against severe disease with the current circulating subvariants, including BA.4 and BA.5. Again, this is probably attributable to T cells which specifically amplify in response to a piece of the virus and help recruit cells to attack the pathogen directly.
The original BA.1 subvariant of Omicron has 26-32 mutations along its spike protein that differ from the “ancestral strain,” and BA.4 and BA.5 variants have a few more. Our T-cell response, even across a mutated spike protein, is so robust that we have not seen Omicron yet able to evade the many T cells (which we produce from the vaccines or infection) that descend upon the mutated virus to fight severe disease. Antibody-producing memory B cells, generated by the vaccines (or prior infection), have been shown to actually adapt their immune response to the variant to which they are exposed.
Therefore, the story of the BA.4 and BA.5 subvariants seems to remain about antibodies vs. cellular immunity. Our immunity in the United States is growing and is from both vaccination and natural infection, with 78.3% of the population having had at least one dose of the vaccine and at least 60% of adults (and 75% of children 0-18) having been exposed to the virus by February 2022, per the Centers for Disease Control and Prevention (with exposure probably much higher now in July 2022 after subsequent Omicron subvariants waves).
So, what about Omicron-specific boosters? A booster shot will just raise antibodies temporarily, but their effectiveness wanes several months later. Moreover, a booster shot against the ancestral strain is not very effective in neutralizing BA.4 and BA.5 (with a prior BA.1 Omicron infection being more effective than a booster). Luckily, Pfizer has promised a BA.4/BA.5-specific mRNA vaccine by October, and Moderna has promised a bivalent vaccine containing BA.4/BA.5 mRNA sequences around the same time. A vaccine that specifically increases antibodies against the most prevalent circulating strain should be important as a booster for those who are predisposed to severe breakthrough infections (for example, those with immunocompromise or older individuals with multiple comorbidities). Moreover, BA.4/BA.5–specific booster vaccines may help prevent mild infections for many individuals. Finally, any booster (or exposure) should diversify and broaden T-cell responses to the virus, and a booster shot will also expand the potency of B cells, making them better able to respond to the newest subvariants as we continue to live with COVID-19.
Monica Gandhi, MD, MPH, is an infectious diseases doctor, professor of medicine, and associate chief in the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco.
A version of this article first appeared on Medscape.com.
The picture around the BA.4 and BA.5 subvariants of Omicron has been really confusing in that the pair is driving up cases but global COVID-19 deaths remain at their lowest level since the beginning of the pandemic. Explaining the two components of the immune response – antibodies versus cellular immune responses – can help us understand where we are in the pandemic and future booster options.
These two subvariants of Omicron, as of July 5, make up more than half of the COVID-19 strains in the United States and are expected to keep increasing. One of two reasons can lead to a variant or subvariant becoming dominant strain: increased transmissibility or evasion of antibodies.
Although BA.4 and BA.5 could be more transmissible than other subvariants of Omicron (which is already very transmissible), this has not yet been established in experiments showing increased affinity for the human receptor or in animal models. What we do know is that BA.4 and BA.5 seem to evade neutralizing antibodies conferred by the vaccines or even prior BA.1 infection (an earlier subvariant of Omicron), which could be the reason we are seeing so many reinfections now. Of note, BA.1 infection conferred antibodies that protected against subsequent BA.2 infection, so we did not see the same spike in cases in the United States with BA.2 (after a large BA.1 spike over the winter) earlier this spring.
Okay, so isn’t evasion of antibodies a bad thing? Of course it is but, luckily, our immune system is “redundant” and doesn›t just rely on antibodies to protect us from infection. In fact, antibodies (such as IgA, which is the mucosal antibody most prevalent in the nose and mouth, and IgG, which is the most prevalent antibody in the bloodstream) are our first line of COVID-19 defense in the nasal mucosa. Therefore, mild upper respiratory infections will be common as BA.4/BA.5 evade our nasal antibodies. Luckily, the rate of severe disease is remaining low throughout the world, probably because of the high amounts of cellular immunity to the virus. B and T cells are our protectors from severe disease.
For instance, two-dose vaccines are still conferring high rates of protection from severe disease with the BA.4 and BA.5 variants, with 87% protection against hospitalization per South Africa data. This is probably attributable to the fact that T-cell immunity from the vaccines remains protective across variants “from Alpha to Omicron,” as described by a recent and elegant paper.
Data from Qatar show that natural infection (even occurring up to 14 months ago) remains very protective (97.3%) against severe disease with the current circulating subvariants, including BA.4 and BA.5. Again, this is probably attributable to T cells which specifically amplify in response to a piece of the virus and help recruit cells to attack the pathogen directly.
The original BA.1 subvariant of Omicron has 26-32 mutations along its spike protein that differ from the “ancestral strain,” and BA.4 and BA.5 variants have a few more. Our T-cell response, even across a mutated spike protein, is so robust that we have not seen Omicron yet able to evade the many T cells (which we produce from the vaccines or infection) that descend upon the mutated virus to fight severe disease. Antibody-producing memory B cells, generated by the vaccines (or prior infection), have been shown to actually adapt their immune response to the variant to which they are exposed.
Therefore, the story of the BA.4 and BA.5 subvariants seems to remain about antibodies vs. cellular immunity. Our immunity in the United States is growing and is from both vaccination and natural infection, with 78.3% of the population having had at least one dose of the vaccine and at least 60% of adults (and 75% of children 0-18) having been exposed to the virus by February 2022, per the Centers for Disease Control and Prevention (with exposure probably much higher now in July 2022 after subsequent Omicron subvariants waves).
So, what about Omicron-specific boosters? A booster shot will just raise antibodies temporarily, but their effectiveness wanes several months later. Moreover, a booster shot against the ancestral strain is not very effective in neutralizing BA.4 and BA.5 (with a prior BA.1 Omicron infection being more effective than a booster). Luckily, Pfizer has promised a BA.4/BA.5-specific mRNA vaccine by October, and Moderna has promised a bivalent vaccine containing BA.4/BA.5 mRNA sequences around the same time. A vaccine that specifically increases antibodies against the most prevalent circulating strain should be important as a booster for those who are predisposed to severe breakthrough infections (for example, those with immunocompromise or older individuals with multiple comorbidities). Moreover, BA.4/BA.5–specific booster vaccines may help prevent mild infections for many individuals. Finally, any booster (or exposure) should diversify and broaden T-cell responses to the virus, and a booster shot will also expand the potency of B cells, making them better able to respond to the newest subvariants as we continue to live with COVID-19.
Monica Gandhi, MD, MPH, is an infectious diseases doctor, professor of medicine, and associate chief in the division of HIV, infectious diseases, and global medicine at the University of California, San Francisco.
A version of this article first appeared on Medscape.com.
High residual liver cancer risk in HCV-cured cirrhosis
A new study confirms the very high risk of hepatocellular carcinoma faced by patients with cirrhosis who have been cured of hepatitis C, a finding the researchers hope will encourage clinicians to communicate risk information to patients and encourage regular HCC screening.
On average, the predicted probability of HCC in cirrhosis patients was 410 times greater than the equivalent probability in the general population, the study team found.
Hamish Innes, PhD, with Public Health Scotland, Glasgow, and colleagues wrote.
“Central to this is ensuring that cured cirrhosis patients understand the risk of HCC and are provided with appropriate surveillance,” they added.
“Most patients with cirrhosis do not adhere to HCC screening guidelines,” Nina Beri, MD, medical oncologist with New York University Perlmutter Cancer Center, who wasn’t involved in the study, said in an interview.
The “important” finding in this study “should be conveyed to patients, as this may help improve screening adherence rates,” Dr. Beri said.
The study was published online in the American Journal of Gastroenterology.
Findings may help promote screening uptake
Dr. Innes and colleagues compared the predicted probability of HCC in 1,803 Scottish adults (mean age, 50 years; 74% male) with cirrhosis and cured hepatitis C to the background risk in the general population of Scotland.
The mean predicted 3-year probability of HCC at the time of sustained viral response (SVR), determined using the aMAP prognostic model, was 3.64% (range, 0.012%-36.12%).
This contrasts with a 3-year HCC probability in the general population ranging from less than 0.0001% to 0.25% depending on demographics.
All patients with cirrhosis – even those at lowest risk – had a higher probability of HCC than the general population, but there was considerable heterogeneity from one patient to the next.
For example, the mean 3-year predicted probability was 18 times higher in the top quintile (9.8%) versus the lowest quintile (0.5%) of risk, the researchers found.
They could not identify a patient subgroup who exhibited a similar HCC risk profile to the general population, as was their hope going into the study.
Dr. Innes and colleagues have developed an online tool that allows clinicians to frame a patient›s 3-year HCC probability against the equivalent probability in the general population.
In the future, they said the scope of the tool could be extended by incorporating general population data from countries beyond Scotland.
“Our hope is that this tool will springboard patient-clinician discussions about HCC risk, and could mitigate low screening uptake,” Dr. Innes and colleagues wrote.
Curing HCV doesn’t eliminate risk
Commenting on the study, Nancy Reau, MD, section chief of hepatology at Rush University Medical Center, Chicago, said curing HCV is “very important and significantly reduces risk for complications, but it doesn’t return you to the normal population.”
Dr. Reau’s advice to cirrhosis patients: “Get screened twice a year.”
Dr. Beri said, in addition to conveying this risk to patients, “it is also important to disseminate this information to the community and to primary care practices, particularly as some patients may not currently follow in a specialized liver disease clinic.”
Also weighing in, Amit Singal, MD, chief of hepatology at the University of Texas Southwestern Medical Center, Dallas, said this study highlights that underlying cirrhosis is “the strongest risk factor for the development of HCC.”
In contrast to other cancers, such as breast and colorectal cancer, in which high risk populations can be identified by readily available information such as age and sex, implementation of HCC screening programs requires identification of patients with cirrhosis, Dr. Singal noted.
“Underuse of HCC screening in clinical practice is often related to providers having difficulty at this step in the process and contributes to the high proportion of HCC detected at late stages,” he told this news organization.
“Availability of accurate noninvasive markers of fibrosis will hopefully help with better identification of patients with cirrhosis moving forward,” Dr. Singal said, “although we as hepatologists need to work closely with our primary care colleagues to ensure these tools are used routinely in at-risk patients, such as those with nonalcoholic fatty liver disease, alcohol-associated liver disease, or history of cured (post-SVR) hepatitis C infection.”
The study was supported by the Medical Research Foundation and Public Health Scotland. Dr. Innes, Dr. Beri, Dr. Reau, and Dr. Singal reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study confirms the very high risk of hepatocellular carcinoma faced by patients with cirrhosis who have been cured of hepatitis C, a finding the researchers hope will encourage clinicians to communicate risk information to patients and encourage regular HCC screening.
On average, the predicted probability of HCC in cirrhosis patients was 410 times greater than the equivalent probability in the general population, the study team found.
Hamish Innes, PhD, with Public Health Scotland, Glasgow, and colleagues wrote.
“Central to this is ensuring that cured cirrhosis patients understand the risk of HCC and are provided with appropriate surveillance,” they added.
“Most patients with cirrhosis do not adhere to HCC screening guidelines,” Nina Beri, MD, medical oncologist with New York University Perlmutter Cancer Center, who wasn’t involved in the study, said in an interview.
The “important” finding in this study “should be conveyed to patients, as this may help improve screening adherence rates,” Dr. Beri said.
The study was published online in the American Journal of Gastroenterology.
Findings may help promote screening uptake
Dr. Innes and colleagues compared the predicted probability of HCC in 1,803 Scottish adults (mean age, 50 years; 74% male) with cirrhosis and cured hepatitis C to the background risk in the general population of Scotland.
The mean predicted 3-year probability of HCC at the time of sustained viral response (SVR), determined using the aMAP prognostic model, was 3.64% (range, 0.012%-36.12%).
This contrasts with a 3-year HCC probability in the general population ranging from less than 0.0001% to 0.25% depending on demographics.
All patients with cirrhosis – even those at lowest risk – had a higher probability of HCC than the general population, but there was considerable heterogeneity from one patient to the next.
For example, the mean 3-year predicted probability was 18 times higher in the top quintile (9.8%) versus the lowest quintile (0.5%) of risk, the researchers found.
They could not identify a patient subgroup who exhibited a similar HCC risk profile to the general population, as was their hope going into the study.
Dr. Innes and colleagues have developed an online tool that allows clinicians to frame a patient›s 3-year HCC probability against the equivalent probability in the general population.
In the future, they said the scope of the tool could be extended by incorporating general population data from countries beyond Scotland.
“Our hope is that this tool will springboard patient-clinician discussions about HCC risk, and could mitigate low screening uptake,” Dr. Innes and colleagues wrote.
Curing HCV doesn’t eliminate risk
Commenting on the study, Nancy Reau, MD, section chief of hepatology at Rush University Medical Center, Chicago, said curing HCV is “very important and significantly reduces risk for complications, but it doesn’t return you to the normal population.”
Dr. Reau’s advice to cirrhosis patients: “Get screened twice a year.”
Dr. Beri said, in addition to conveying this risk to patients, “it is also important to disseminate this information to the community and to primary care practices, particularly as some patients may not currently follow in a specialized liver disease clinic.”
Also weighing in, Amit Singal, MD, chief of hepatology at the University of Texas Southwestern Medical Center, Dallas, said this study highlights that underlying cirrhosis is “the strongest risk factor for the development of HCC.”
In contrast to other cancers, such as breast and colorectal cancer, in which high risk populations can be identified by readily available information such as age and sex, implementation of HCC screening programs requires identification of patients with cirrhosis, Dr. Singal noted.
“Underuse of HCC screening in clinical practice is often related to providers having difficulty at this step in the process and contributes to the high proportion of HCC detected at late stages,” he told this news organization.
“Availability of accurate noninvasive markers of fibrosis will hopefully help with better identification of patients with cirrhosis moving forward,” Dr. Singal said, “although we as hepatologists need to work closely with our primary care colleagues to ensure these tools are used routinely in at-risk patients, such as those with nonalcoholic fatty liver disease, alcohol-associated liver disease, or history of cured (post-SVR) hepatitis C infection.”
The study was supported by the Medical Research Foundation and Public Health Scotland. Dr. Innes, Dr. Beri, Dr. Reau, and Dr. Singal reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study confirms the very high risk of hepatocellular carcinoma faced by patients with cirrhosis who have been cured of hepatitis C, a finding the researchers hope will encourage clinicians to communicate risk information to patients and encourage regular HCC screening.
On average, the predicted probability of HCC in cirrhosis patients was 410 times greater than the equivalent probability in the general population, the study team found.
Hamish Innes, PhD, with Public Health Scotland, Glasgow, and colleagues wrote.
“Central to this is ensuring that cured cirrhosis patients understand the risk of HCC and are provided with appropriate surveillance,” they added.
“Most patients with cirrhosis do not adhere to HCC screening guidelines,” Nina Beri, MD, medical oncologist with New York University Perlmutter Cancer Center, who wasn’t involved in the study, said in an interview.
The “important” finding in this study “should be conveyed to patients, as this may help improve screening adherence rates,” Dr. Beri said.
The study was published online in the American Journal of Gastroenterology.
Findings may help promote screening uptake
Dr. Innes and colleagues compared the predicted probability of HCC in 1,803 Scottish adults (mean age, 50 years; 74% male) with cirrhosis and cured hepatitis C to the background risk in the general population of Scotland.
The mean predicted 3-year probability of HCC at the time of sustained viral response (SVR), determined using the aMAP prognostic model, was 3.64% (range, 0.012%-36.12%).
This contrasts with a 3-year HCC probability in the general population ranging from less than 0.0001% to 0.25% depending on demographics.
All patients with cirrhosis – even those at lowest risk – had a higher probability of HCC than the general population, but there was considerable heterogeneity from one patient to the next.
For example, the mean 3-year predicted probability was 18 times higher in the top quintile (9.8%) versus the lowest quintile (0.5%) of risk, the researchers found.
They could not identify a patient subgroup who exhibited a similar HCC risk profile to the general population, as was their hope going into the study.
Dr. Innes and colleagues have developed an online tool that allows clinicians to frame a patient›s 3-year HCC probability against the equivalent probability in the general population.
In the future, they said the scope of the tool could be extended by incorporating general population data from countries beyond Scotland.
“Our hope is that this tool will springboard patient-clinician discussions about HCC risk, and could mitigate low screening uptake,” Dr. Innes and colleagues wrote.
Curing HCV doesn’t eliminate risk
Commenting on the study, Nancy Reau, MD, section chief of hepatology at Rush University Medical Center, Chicago, said curing HCV is “very important and significantly reduces risk for complications, but it doesn’t return you to the normal population.”
Dr. Reau’s advice to cirrhosis patients: “Get screened twice a year.”
Dr. Beri said, in addition to conveying this risk to patients, “it is also important to disseminate this information to the community and to primary care practices, particularly as some patients may not currently follow in a specialized liver disease clinic.”
Also weighing in, Amit Singal, MD, chief of hepatology at the University of Texas Southwestern Medical Center, Dallas, said this study highlights that underlying cirrhosis is “the strongest risk factor for the development of HCC.”
In contrast to other cancers, such as breast and colorectal cancer, in which high risk populations can be identified by readily available information such as age and sex, implementation of HCC screening programs requires identification of patients with cirrhosis, Dr. Singal noted.
“Underuse of HCC screening in clinical practice is often related to providers having difficulty at this step in the process and contributes to the high proportion of HCC detected at late stages,” he told this news organization.
“Availability of accurate noninvasive markers of fibrosis will hopefully help with better identification of patients with cirrhosis moving forward,” Dr. Singal said, “although we as hepatologists need to work closely with our primary care colleagues to ensure these tools are used routinely in at-risk patients, such as those with nonalcoholic fatty liver disease, alcohol-associated liver disease, or history of cured (post-SVR) hepatitis C infection.”
The study was supported by the Medical Research Foundation and Public Health Scotland. Dr. Innes, Dr. Beri, Dr. Reau, and Dr. Singal reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Obesity links to faster fading of COVID vaccine protection
Researchers published the study covered in this summary on medRxiv.org as a preprint that has not yet been peer reviewed.
Key takeaways
- The study results suggest that
- The findings documented evidence of reduced neutralizing antibody capacity 6 months after primary vaccination in people with severe obesity.
- This was a large study involving about more than 3.5 million people who had received at least two doses of COVID-19 vaccine, including more than 650,000 with obesity.
Why this matters
- Obesity is associated with comorbidities that independently increase the risk for severe COVID-19, including type 2 diabetes, chronic kidney disease, and heart failure.
- The authors concluded that additional or more frequent booster doses are likely to be required to maintain protection among people with obesity against COVID-19.
Study design
- Prospective longitudinal study of the incidence and severity of COVID-19 infections and immune responses in a cohort of more than 3.5 million adults from a Scottish healthcare database who received two or three doses of COVID-19 vaccine. The data came from the study, centered at the University of Edinburgh.
- About 16% had obesity with a body mass index of 30-39.9 kg/m2, and an additional 3% had severe obesity with a BMI of 40 or greater.
- Although not specified in this preprint, another said that the vaccines administered in Scotland have been the Pfizer-BioNTech and Oxford-AstraZeneca formulations.
Key results
- Between Sept. 14, 2020, and March 19, 2022, 10,983 people (0.3% of the total cohort; 6.0 events per 1,000 person-years) had severe COVID-19, consisting of 9,733 who were hospitalized and 2,207 who died (957 of those hospitalized also died).
- People with obesity or severe obesity were at higher risk of hospitalization or death from COVID-19 after both a second and third (booster) dose of vaccine.
- Compared with those with normal weight, those with severe obesity (BMI higher than 40) were at significantly increased risk for severe COVID-19 after a second vaccine dose, with an adjusted rate ratio 1.76, whereas those with standard obesity (BMI, 30-40) were at a modestly but significantly increased risk with an adjusted rate ratio of 1.11.
- Breakthrough infections after the second dose for those with severe obesity, obesity, and normal weight occurred on average at 10 weeks, 15 weeks, and 20 weeks, respectively.
- Interaction testing showed that vaccine effectiveness significantly diminished over time across BMI groups, and protection waned more rapidly as BMI increased.
- Results from immunophenotyping studies run in a subgroup of several dozen subjects with severe obesity or normal weight showed significant decrements in the robustness of antibody responses in those with severe obesity 6 months after a second or third vaccine dose.
Limitations
- The authors did not specify any limitations.
Disclosures
- The study received no commercial funding.
- One author received funding from Wellcome.
This is a summary of a preprint research study , “Accelerated waning of the humoral response to SARS-CoV-2 vaccines in obesity,” published by researchers primarily at the University of Cambridge (England), on medRxiv. This study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Researchers published the study covered in this summary on medRxiv.org as a preprint that has not yet been peer reviewed.
Key takeaways
- The study results suggest that
- The findings documented evidence of reduced neutralizing antibody capacity 6 months after primary vaccination in people with severe obesity.
- This was a large study involving about more than 3.5 million people who had received at least two doses of COVID-19 vaccine, including more than 650,000 with obesity.
Why this matters
- Obesity is associated with comorbidities that independently increase the risk for severe COVID-19, including type 2 diabetes, chronic kidney disease, and heart failure.
- The authors concluded that additional or more frequent booster doses are likely to be required to maintain protection among people with obesity against COVID-19.
Study design
- Prospective longitudinal study of the incidence and severity of COVID-19 infections and immune responses in a cohort of more than 3.5 million adults from a Scottish healthcare database who received two or three doses of COVID-19 vaccine. The data came from the study, centered at the University of Edinburgh.
- About 16% had obesity with a body mass index of 30-39.9 kg/m2, and an additional 3% had severe obesity with a BMI of 40 or greater.
- Although not specified in this preprint, another said that the vaccines administered in Scotland have been the Pfizer-BioNTech and Oxford-AstraZeneca formulations.
Key results
- Between Sept. 14, 2020, and March 19, 2022, 10,983 people (0.3% of the total cohort; 6.0 events per 1,000 person-years) had severe COVID-19, consisting of 9,733 who were hospitalized and 2,207 who died (957 of those hospitalized also died).
- People with obesity or severe obesity were at higher risk of hospitalization or death from COVID-19 after both a second and third (booster) dose of vaccine.
- Compared with those with normal weight, those with severe obesity (BMI higher than 40) were at significantly increased risk for severe COVID-19 after a second vaccine dose, with an adjusted rate ratio 1.76, whereas those with standard obesity (BMI, 30-40) were at a modestly but significantly increased risk with an adjusted rate ratio of 1.11.
- Breakthrough infections after the second dose for those with severe obesity, obesity, and normal weight occurred on average at 10 weeks, 15 weeks, and 20 weeks, respectively.
- Interaction testing showed that vaccine effectiveness significantly diminished over time across BMI groups, and protection waned more rapidly as BMI increased.
- Results from immunophenotyping studies run in a subgroup of several dozen subjects with severe obesity or normal weight showed significant decrements in the robustness of antibody responses in those with severe obesity 6 months after a second or third vaccine dose.
Limitations
- The authors did not specify any limitations.
Disclosures
- The study received no commercial funding.
- One author received funding from Wellcome.
This is a summary of a preprint research study , “Accelerated waning of the humoral response to SARS-CoV-2 vaccines in obesity,” published by researchers primarily at the University of Cambridge (England), on medRxiv. This study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.
Researchers published the study covered in this summary on medRxiv.org as a preprint that has not yet been peer reviewed.
Key takeaways
- The study results suggest that
- The findings documented evidence of reduced neutralizing antibody capacity 6 months after primary vaccination in people with severe obesity.
- This was a large study involving about more than 3.5 million people who had received at least two doses of COVID-19 vaccine, including more than 650,000 with obesity.
Why this matters
- Obesity is associated with comorbidities that independently increase the risk for severe COVID-19, including type 2 diabetes, chronic kidney disease, and heart failure.
- The authors concluded that additional or more frequent booster doses are likely to be required to maintain protection among people with obesity against COVID-19.
Study design
- Prospective longitudinal study of the incidence and severity of COVID-19 infections and immune responses in a cohort of more than 3.5 million adults from a Scottish healthcare database who received two or three doses of COVID-19 vaccine. The data came from the study, centered at the University of Edinburgh.
- About 16% had obesity with a body mass index of 30-39.9 kg/m2, and an additional 3% had severe obesity with a BMI of 40 or greater.
- Although not specified in this preprint, another said that the vaccines administered in Scotland have been the Pfizer-BioNTech and Oxford-AstraZeneca formulations.
Key results
- Between Sept. 14, 2020, and March 19, 2022, 10,983 people (0.3% of the total cohort; 6.0 events per 1,000 person-years) had severe COVID-19, consisting of 9,733 who were hospitalized and 2,207 who died (957 of those hospitalized also died).
- People with obesity or severe obesity were at higher risk of hospitalization or death from COVID-19 after both a second and third (booster) dose of vaccine.
- Compared with those with normal weight, those with severe obesity (BMI higher than 40) were at significantly increased risk for severe COVID-19 after a second vaccine dose, with an adjusted rate ratio 1.76, whereas those with standard obesity (BMI, 30-40) were at a modestly but significantly increased risk with an adjusted rate ratio of 1.11.
- Breakthrough infections after the second dose for those with severe obesity, obesity, and normal weight occurred on average at 10 weeks, 15 weeks, and 20 weeks, respectively.
- Interaction testing showed that vaccine effectiveness significantly diminished over time across BMI groups, and protection waned more rapidly as BMI increased.
- Results from immunophenotyping studies run in a subgroup of several dozen subjects with severe obesity or normal weight showed significant decrements in the robustness of antibody responses in those with severe obesity 6 months after a second or third vaccine dose.
Limitations
- The authors did not specify any limitations.
Disclosures
- The study received no commercial funding.
- One author received funding from Wellcome.
This is a summary of a preprint research study , “Accelerated waning of the humoral response to SARS-CoV-2 vaccines in obesity,” published by researchers primarily at the University of Cambridge (England), on medRxiv. This study has not yet been peer reviewed. The full text of the study can be found on medRxiv.org.
A version of this article first appeared on Medscape.com.

