User login
-
‘Financial toxicity’: Harsh side effect of cancer care
When 32-year-old Brittany Dicks was diagnosed with stage II triple negative breast cancer in January 2022, she wasn’t worried about the cost of treatment. A medical assistant in Charleston, S.C., Ms. Dicks had full-time employment with health benefits.
But when she wasn’t able to work for several months because of chemotherapy and its side effects, Ms. Dicks lost her job. Her health insurance coverage ended in May. And although she filed for Medicaid at the beginning of June, it wasn’t approved until September.
Meanwhile, Ms. Dicks still needed treatment. She estimates that she ran up close to $20,000 in medical debt while finishing chemotherapy during the 4 months she was uninsured.
The surgeon she had seen since her diagnosis terminated her care when she could no longer pay her bills. That left her delaying a much-needed mastectomy.
“I don’t sleep at night,” said Ms. Dicks, a single mother of two young kids, ages 3 and 11. “Mentally, I’m drained. Just because I have cancer, doesn’t mean the bills aren’t due every month.”
As soon as she felt well enough over the summer, she started working as a part-time delivery driver for DoorDash to help pay for food and gas.
But that was just a Band-Aid. Even when her new insurance kicked in, covering the costs of daily life remained a struggle.
Ms. Dicks is still in deep medical debt. Her Medicaid has covered new medical expenses, and she hopes Medicaid will reimburse her for the debt she incurred over the summer while she waited for her coverage to kick in. So far, though, Medicaid has not touched her $20,000 debt.
“I fear that I’m not going to be able to dig out of this hole,” Ms. Dicks said.
Researchers who study the financial impacts of cancer have a term for Ms. Dicks’ experience: financial toxicity.
Financial toxicity is a catchall term for the burden many Americans with cancer experience.
“Financial toxicity is a multidimensional concept. There’s both a material burden and a psychosocial one,” said Grace Li Smith, MD, PhD, MPH, a radiation oncologist at the University of Texas MD Anderson Cancer Center, Houston.
Researchers are also now beginning to understand the psychological effects these financial burdens can have on patients and their family.
“Financial toxicity is not unique to the patient,” said Dr. Li Smith. It “very directly impacts the whole family or household.”
Stifling financial pressures
Early in her career, Dr. Li Smith was already seeing how her patients’ worries extended beyond their physical disease.
One of Dr. Li Smith’s first patients told her their greatest worry wasn’t whether the treatment would work or what physical toxicity to expect, it was how they would pay for their care.
“There was much more anxiety and true distress about the financial burden than about the treatment itself,” Dr. Li Smith recalled.
This fear about the costs of cancer care is well founded. In the United States, cancer treatment costs reached an estimated $150 billion in 2020 and continue to rise. Patients shoulder a significant portion of that burden – with one study estimating that patients paid $21 billion for their cancer care in 2019.
The burden is often compounded by decreased income. Between 40% and 85% of patients with cancer needed to take time off work or quit their jobs during treatment. And for those, like Dicks, who find themselves with no insurance, out-of-pocket costs can quickly skyrocket.
In fact, one study of newly diagnosed cancer patients over age 50 reported that more than 42% of patients fully depleted their financial assets and around 30% incurred debt by the second year of their diagnosis.
Younger adults may be even more financially vulnerable. A study of patients in Washington found that those under 65 – which represent about half of cancer cases – were two to five times more likely to declare bankruptcy than patients over 65.
Dr. Li Smith and colleagues have found that younger patients aged 18-64 experienced greater monetary hardships, which meant less money for food, worse adherence to medications, as well as greater distress and anxiety overall. In fact, younger adults were over 4.5 times more likely to encounter severe financial toxicity, compared with older adults, and about 4 times more likely to experience severe psychological effects from this burden.
The distress, if left unchecked, can spiral out of control.
Molly MacDonald had just gone through a financially devastating divorce in 2005 when she was diagnosed with breast cancer. Recently out of work and dealing with a $1,300 monthly COBRA premium, the mother of five had no financial safety net. She risked having her car repossessed and her utilities shut off.
“I gave tentative thought to how I could take my life and make it look like an accident,” said Ms. MacDonald. “I thought the kids would be better off without me.”
For some, the loss of income can be even more worrisome than the medical bills. Some patients may go back to work during treatment, often against medical advice.
When Stephanie Caputo, 43, of Monroe, N.J., began treatment for stage III breast cancer in 2021, her physician recommended she stop working. Treatment would make her immunocompromised, and her job in a medical clinic could expose her to harmful pathogens, including the coronavirus.
Ms. Caputo went on disability and received $900 every 2 weeks. But that wasn’t enough to pay her mortgage, let alone cover her other monthly expenses as a single mother of 4 teenagers.
After finishing chemotherapy, and during radiation, Ms. Caputo went back to work, part time, against her doctor’s advice.
“My doctor is telling me I can’t work, but I also can’t have my house go into default,” said Ms. Caputo.
But being on her feet through 12-hour shifts made treatment side effects, especially back and joint pain, kick into overdrive. “The physicality of my job was really difficult to tolerate,” she said.
The physical burden was too great to take on more work, but the extra money also wasn’t enough to keep her afloat. Fortunately, her brother stepped in and covered 6 months of her mortgage payments.
Financial toxicity impacts families
Although financial toxicity research to date has largely focused on the patient, researchers are also starting to understand that family members and caregivers often share in the burden.
“We are just at the beginning of realizing that this is a real problem,” said Fumiko Chino, MD, a radiation oncologist at Memorial Sloan Kettering Cancer Center, New York.
Dr. Chino and colleagues recently showed that family members of patients with cancer were more likely to delay or forgo medical care than family members of people without cancer. The study found the effect was greatest among family members of younger adults with cancer.
“The caregiver and family burden related to cancer diagnosis and treatment is really underappreciated,” said Dr. Chino. “Family members and caregivers are neglecting their own health concerns, passing up career opportunities, struggling with financial concerns.”
Dr. Chino speaks from personal experience. When her fiancé, later husband, was diagnosed with neuroendocrine carcinoma in 2005, Dr. Chino quit her job as art director at a television production company to take care of him.
The couple, both in their 20s, struggled to afford his care. Dr. Chino put her own dental, medical, and mental health care on hold. She never, for instance, went to physical therapy to address injuries sustained sleeping in hospital chairs and moving around her husband who was over 6 feet tall. At one point, she walked with a limp.
Dr. Chino’s husband passed away in 2007, and even 15 years later, her injury from sleeping in hospital chairs remains “a significant physical burden,” she said. But like many caregivers “I wasn’t really thinking about my own health.”
Danielle Hadfield, 35, an ED nurse in Rochester, N.Y., also delayed her own care when her mom got sick.
Ms. Hadfield quit her job shortly after her mom was diagnosed with cholangiocarcinoma in August 2020. Ms. Hadfield knew her mom, who lived 3.5 hours away in Albany, N.Y., would need a lot of care in the upcoming months.
“I knew this was going to be the last year or so of her life, and I wanted to be there for her,” said Ms. Hadfield.
When Ms. Hadfield quit her job, she and her husband – who was self-employed – purchased health insurance coverage through the New York state marketplace. The monthly insurance payments for Ms. Hadfield, who was pregnant with her second child, her husband, and their toddler cost as much as the family’s monthly mortgage payments.
In addition to providing childcare for her young daughter and making frequent trips to Albany, Ms. Hadfield began a side business as a legal nurse consultant, working mostly at night, to replace a portion of her lost income. During this time, she began to experience pain attacks that would migrate through her body along with intermittent tongue and facial numbness. She ignored these health issues for nearly a year, until after her mother died in November 2021.
Only after her mother passed away did Ms. Hadfield begin seeking answers to her own pain. In September 2022, she finally got them. She had a nerve condition called small-fiber sensory neuropathy.
But even with a diagnosis, she is still facing more tests to root out the cause and understand the best treatment.
Is help out there?
What can physicians do to help patients and families at risk for financial toxicity?
Specific guidelines for dealing with financial toxicity do not exist in most professional guidelines, nor are there standard screening tools to identify it, said Dr. Li Smith.
These gaps put pressure on physicians to ask about financial barriers and concerns, but most do not know how to broach the topic or how to help. “Physicians may not know how to fix the problem or what resources exist,” Dr. Li Smith said.
Patients and family members, on the other hand, are often reluctant to bring up cost with physicians. Some may be ashamed to talk about their financial problems while others may fear doing so will prevent them from being offered the best possible treatments, said Ms. MacDonald.
But, experts say, financial toxicity needs to be dealt with head on. That means involving financial navigators or counselors and social workers who can, for instance, help patients and families find financial support for their basic living expenses.
From a research perspective, more clinical trials should include financial toxicity outcomes, said Joshua Palmer, MD, a radiation oncologist at the University of Michigan, Ann Arbor.
Dr. Palmer and colleagues recently showed that the number of radiation therapy clinical trials including financial toxicity endpoints increased significantly from 2001 to 2020, though the absolute rate of inclusion remains low, at roughly 1.5% of radiation therapy-based clinical trials including financial toxicity endpoints from 2016 to 2020.
“Financial burden is part of the broader discussion about shared decision-making,” said Dr. Palmer.
In shared decision-making, physicians discuss the risks and benefits of different treatment options, empowering the patient to make an informed choice with the physician.
What we want to avoid is patients feeling like they will get inferior care, if they have financial barriers, said Dr. Palmer.
And every little bit can help. In 2006, Ms. MacDonald started the Pink Fund – a nonprofit to help patients with cancer cover nonmedical cost-of-living expenses. Both Ms. Caputo and Ms. Dicks received grants from the Pink Fund. For Ms. Caputo, the funds covered 2 months of car payments and for Ms. Dicks, it covered 2 months of rent.
While the one-time grant was a big help, said Ms. Dicks, “cancer is an everyday thing.” And “we all deserve peace of mind” when trying to heal.
A version of this article first appeared on WebMD.com.
When 32-year-old Brittany Dicks was diagnosed with stage II triple negative breast cancer in January 2022, she wasn’t worried about the cost of treatment. A medical assistant in Charleston, S.C., Ms. Dicks had full-time employment with health benefits.
But when she wasn’t able to work for several months because of chemotherapy and its side effects, Ms. Dicks lost her job. Her health insurance coverage ended in May. And although she filed for Medicaid at the beginning of June, it wasn’t approved until September.
Meanwhile, Ms. Dicks still needed treatment. She estimates that she ran up close to $20,000 in medical debt while finishing chemotherapy during the 4 months she was uninsured.
The surgeon she had seen since her diagnosis terminated her care when she could no longer pay her bills. That left her delaying a much-needed mastectomy.
“I don’t sleep at night,” said Ms. Dicks, a single mother of two young kids, ages 3 and 11. “Mentally, I’m drained. Just because I have cancer, doesn’t mean the bills aren’t due every month.”
As soon as she felt well enough over the summer, she started working as a part-time delivery driver for DoorDash to help pay for food and gas.
But that was just a Band-Aid. Even when her new insurance kicked in, covering the costs of daily life remained a struggle.
Ms. Dicks is still in deep medical debt. Her Medicaid has covered new medical expenses, and she hopes Medicaid will reimburse her for the debt she incurred over the summer while she waited for her coverage to kick in. So far, though, Medicaid has not touched her $20,000 debt.
“I fear that I’m not going to be able to dig out of this hole,” Ms. Dicks said.
Researchers who study the financial impacts of cancer have a term for Ms. Dicks’ experience: financial toxicity.
Financial toxicity is a catchall term for the burden many Americans with cancer experience.
“Financial toxicity is a multidimensional concept. There’s both a material burden and a psychosocial one,” said Grace Li Smith, MD, PhD, MPH, a radiation oncologist at the University of Texas MD Anderson Cancer Center, Houston.
Researchers are also now beginning to understand the psychological effects these financial burdens can have on patients and their family.
“Financial toxicity is not unique to the patient,” said Dr. Li Smith. It “very directly impacts the whole family or household.”
Stifling financial pressures
Early in her career, Dr. Li Smith was already seeing how her patients’ worries extended beyond their physical disease.
One of Dr. Li Smith’s first patients told her their greatest worry wasn’t whether the treatment would work or what physical toxicity to expect, it was how they would pay for their care.
“There was much more anxiety and true distress about the financial burden than about the treatment itself,” Dr. Li Smith recalled.
This fear about the costs of cancer care is well founded. In the United States, cancer treatment costs reached an estimated $150 billion in 2020 and continue to rise. Patients shoulder a significant portion of that burden – with one study estimating that patients paid $21 billion for their cancer care in 2019.
The burden is often compounded by decreased income. Between 40% and 85% of patients with cancer needed to take time off work or quit their jobs during treatment. And for those, like Dicks, who find themselves with no insurance, out-of-pocket costs can quickly skyrocket.
In fact, one study of newly diagnosed cancer patients over age 50 reported that more than 42% of patients fully depleted their financial assets and around 30% incurred debt by the second year of their diagnosis.
Younger adults may be even more financially vulnerable. A study of patients in Washington found that those under 65 – which represent about half of cancer cases – were two to five times more likely to declare bankruptcy than patients over 65.
Dr. Li Smith and colleagues have found that younger patients aged 18-64 experienced greater monetary hardships, which meant less money for food, worse adherence to medications, as well as greater distress and anxiety overall. In fact, younger adults were over 4.5 times more likely to encounter severe financial toxicity, compared with older adults, and about 4 times more likely to experience severe psychological effects from this burden.
The distress, if left unchecked, can spiral out of control.
Molly MacDonald had just gone through a financially devastating divorce in 2005 when she was diagnosed with breast cancer. Recently out of work and dealing with a $1,300 monthly COBRA premium, the mother of five had no financial safety net. She risked having her car repossessed and her utilities shut off.
“I gave tentative thought to how I could take my life and make it look like an accident,” said Ms. MacDonald. “I thought the kids would be better off without me.”
For some, the loss of income can be even more worrisome than the medical bills. Some patients may go back to work during treatment, often against medical advice.
When Stephanie Caputo, 43, of Monroe, N.J., began treatment for stage III breast cancer in 2021, her physician recommended she stop working. Treatment would make her immunocompromised, and her job in a medical clinic could expose her to harmful pathogens, including the coronavirus.
Ms. Caputo went on disability and received $900 every 2 weeks. But that wasn’t enough to pay her mortgage, let alone cover her other monthly expenses as a single mother of 4 teenagers.
After finishing chemotherapy, and during radiation, Ms. Caputo went back to work, part time, against her doctor’s advice.
“My doctor is telling me I can’t work, but I also can’t have my house go into default,” said Ms. Caputo.
But being on her feet through 12-hour shifts made treatment side effects, especially back and joint pain, kick into overdrive. “The physicality of my job was really difficult to tolerate,” she said.
The physical burden was too great to take on more work, but the extra money also wasn’t enough to keep her afloat. Fortunately, her brother stepped in and covered 6 months of her mortgage payments.
Financial toxicity impacts families
Although financial toxicity research to date has largely focused on the patient, researchers are also starting to understand that family members and caregivers often share in the burden.
“We are just at the beginning of realizing that this is a real problem,” said Fumiko Chino, MD, a radiation oncologist at Memorial Sloan Kettering Cancer Center, New York.
Dr. Chino and colleagues recently showed that family members of patients with cancer were more likely to delay or forgo medical care than family members of people without cancer. The study found the effect was greatest among family members of younger adults with cancer.
“The caregiver and family burden related to cancer diagnosis and treatment is really underappreciated,” said Dr. Chino. “Family members and caregivers are neglecting their own health concerns, passing up career opportunities, struggling with financial concerns.”
Dr. Chino speaks from personal experience. When her fiancé, later husband, was diagnosed with neuroendocrine carcinoma in 2005, Dr. Chino quit her job as art director at a television production company to take care of him.
The couple, both in their 20s, struggled to afford his care. Dr. Chino put her own dental, medical, and mental health care on hold. She never, for instance, went to physical therapy to address injuries sustained sleeping in hospital chairs and moving around her husband who was over 6 feet tall. At one point, she walked with a limp.
Dr. Chino’s husband passed away in 2007, and even 15 years later, her injury from sleeping in hospital chairs remains “a significant physical burden,” she said. But like many caregivers “I wasn’t really thinking about my own health.”
Danielle Hadfield, 35, an ED nurse in Rochester, N.Y., also delayed her own care when her mom got sick.
Ms. Hadfield quit her job shortly after her mom was diagnosed with cholangiocarcinoma in August 2020. Ms. Hadfield knew her mom, who lived 3.5 hours away in Albany, N.Y., would need a lot of care in the upcoming months.
“I knew this was going to be the last year or so of her life, and I wanted to be there for her,” said Ms. Hadfield.
When Ms. Hadfield quit her job, she and her husband – who was self-employed – purchased health insurance coverage through the New York state marketplace. The monthly insurance payments for Ms. Hadfield, who was pregnant with her second child, her husband, and their toddler cost as much as the family’s monthly mortgage payments.
In addition to providing childcare for her young daughter and making frequent trips to Albany, Ms. Hadfield began a side business as a legal nurse consultant, working mostly at night, to replace a portion of her lost income. During this time, she began to experience pain attacks that would migrate through her body along with intermittent tongue and facial numbness. She ignored these health issues for nearly a year, until after her mother died in November 2021.
Only after her mother passed away did Ms. Hadfield begin seeking answers to her own pain. In September 2022, she finally got them. She had a nerve condition called small-fiber sensory neuropathy.
But even with a diagnosis, she is still facing more tests to root out the cause and understand the best treatment.
Is help out there?
What can physicians do to help patients and families at risk for financial toxicity?
Specific guidelines for dealing with financial toxicity do not exist in most professional guidelines, nor are there standard screening tools to identify it, said Dr. Li Smith.
These gaps put pressure on physicians to ask about financial barriers and concerns, but most do not know how to broach the topic or how to help. “Physicians may not know how to fix the problem or what resources exist,” Dr. Li Smith said.
Patients and family members, on the other hand, are often reluctant to bring up cost with physicians. Some may be ashamed to talk about their financial problems while others may fear doing so will prevent them from being offered the best possible treatments, said Ms. MacDonald.
But, experts say, financial toxicity needs to be dealt with head on. That means involving financial navigators or counselors and social workers who can, for instance, help patients and families find financial support for their basic living expenses.
From a research perspective, more clinical trials should include financial toxicity outcomes, said Joshua Palmer, MD, a radiation oncologist at the University of Michigan, Ann Arbor.
Dr. Palmer and colleagues recently showed that the number of radiation therapy clinical trials including financial toxicity endpoints increased significantly from 2001 to 2020, though the absolute rate of inclusion remains low, at roughly 1.5% of radiation therapy-based clinical trials including financial toxicity endpoints from 2016 to 2020.
“Financial burden is part of the broader discussion about shared decision-making,” said Dr. Palmer.
In shared decision-making, physicians discuss the risks and benefits of different treatment options, empowering the patient to make an informed choice with the physician.
What we want to avoid is patients feeling like they will get inferior care, if they have financial barriers, said Dr. Palmer.
And every little bit can help. In 2006, Ms. MacDonald started the Pink Fund – a nonprofit to help patients with cancer cover nonmedical cost-of-living expenses. Both Ms. Caputo and Ms. Dicks received grants from the Pink Fund. For Ms. Caputo, the funds covered 2 months of car payments and for Ms. Dicks, it covered 2 months of rent.
While the one-time grant was a big help, said Ms. Dicks, “cancer is an everyday thing.” And “we all deserve peace of mind” when trying to heal.
A version of this article first appeared on WebMD.com.
When 32-year-old Brittany Dicks was diagnosed with stage II triple negative breast cancer in January 2022, she wasn’t worried about the cost of treatment. A medical assistant in Charleston, S.C., Ms. Dicks had full-time employment with health benefits.
But when she wasn’t able to work for several months because of chemotherapy and its side effects, Ms. Dicks lost her job. Her health insurance coverage ended in May. And although she filed for Medicaid at the beginning of June, it wasn’t approved until September.
Meanwhile, Ms. Dicks still needed treatment. She estimates that she ran up close to $20,000 in medical debt while finishing chemotherapy during the 4 months she was uninsured.
The surgeon she had seen since her diagnosis terminated her care when she could no longer pay her bills. That left her delaying a much-needed mastectomy.
“I don’t sleep at night,” said Ms. Dicks, a single mother of two young kids, ages 3 and 11. “Mentally, I’m drained. Just because I have cancer, doesn’t mean the bills aren’t due every month.”
As soon as she felt well enough over the summer, she started working as a part-time delivery driver for DoorDash to help pay for food and gas.
But that was just a Band-Aid. Even when her new insurance kicked in, covering the costs of daily life remained a struggle.
Ms. Dicks is still in deep medical debt. Her Medicaid has covered new medical expenses, and she hopes Medicaid will reimburse her for the debt she incurred over the summer while she waited for her coverage to kick in. So far, though, Medicaid has not touched her $20,000 debt.
“I fear that I’m not going to be able to dig out of this hole,” Ms. Dicks said.
Researchers who study the financial impacts of cancer have a term for Ms. Dicks’ experience: financial toxicity.
Financial toxicity is a catchall term for the burden many Americans with cancer experience.
“Financial toxicity is a multidimensional concept. There’s both a material burden and a psychosocial one,” said Grace Li Smith, MD, PhD, MPH, a radiation oncologist at the University of Texas MD Anderson Cancer Center, Houston.
Researchers are also now beginning to understand the psychological effects these financial burdens can have on patients and their family.
“Financial toxicity is not unique to the patient,” said Dr. Li Smith. It “very directly impacts the whole family or household.”
Stifling financial pressures
Early in her career, Dr. Li Smith was already seeing how her patients’ worries extended beyond their physical disease.
One of Dr. Li Smith’s first patients told her their greatest worry wasn’t whether the treatment would work or what physical toxicity to expect, it was how they would pay for their care.
“There was much more anxiety and true distress about the financial burden than about the treatment itself,” Dr. Li Smith recalled.
This fear about the costs of cancer care is well founded. In the United States, cancer treatment costs reached an estimated $150 billion in 2020 and continue to rise. Patients shoulder a significant portion of that burden – with one study estimating that patients paid $21 billion for their cancer care in 2019.
The burden is often compounded by decreased income. Between 40% and 85% of patients with cancer needed to take time off work or quit their jobs during treatment. And for those, like Dicks, who find themselves with no insurance, out-of-pocket costs can quickly skyrocket.
In fact, one study of newly diagnosed cancer patients over age 50 reported that more than 42% of patients fully depleted their financial assets and around 30% incurred debt by the second year of their diagnosis.
Younger adults may be even more financially vulnerable. A study of patients in Washington found that those under 65 – which represent about half of cancer cases – were two to five times more likely to declare bankruptcy than patients over 65.
Dr. Li Smith and colleagues have found that younger patients aged 18-64 experienced greater monetary hardships, which meant less money for food, worse adherence to medications, as well as greater distress and anxiety overall. In fact, younger adults were over 4.5 times more likely to encounter severe financial toxicity, compared with older adults, and about 4 times more likely to experience severe psychological effects from this burden.
The distress, if left unchecked, can spiral out of control.
Molly MacDonald had just gone through a financially devastating divorce in 2005 when she was diagnosed with breast cancer. Recently out of work and dealing with a $1,300 monthly COBRA premium, the mother of five had no financial safety net. She risked having her car repossessed and her utilities shut off.
“I gave tentative thought to how I could take my life and make it look like an accident,” said Ms. MacDonald. “I thought the kids would be better off without me.”
For some, the loss of income can be even more worrisome than the medical bills. Some patients may go back to work during treatment, often against medical advice.
When Stephanie Caputo, 43, of Monroe, N.J., began treatment for stage III breast cancer in 2021, her physician recommended she stop working. Treatment would make her immunocompromised, and her job in a medical clinic could expose her to harmful pathogens, including the coronavirus.
Ms. Caputo went on disability and received $900 every 2 weeks. But that wasn’t enough to pay her mortgage, let alone cover her other monthly expenses as a single mother of 4 teenagers.
After finishing chemotherapy, and during radiation, Ms. Caputo went back to work, part time, against her doctor’s advice.
“My doctor is telling me I can’t work, but I also can’t have my house go into default,” said Ms. Caputo.
But being on her feet through 12-hour shifts made treatment side effects, especially back and joint pain, kick into overdrive. “The physicality of my job was really difficult to tolerate,” she said.
The physical burden was too great to take on more work, but the extra money also wasn’t enough to keep her afloat. Fortunately, her brother stepped in and covered 6 months of her mortgage payments.
Financial toxicity impacts families
Although financial toxicity research to date has largely focused on the patient, researchers are also starting to understand that family members and caregivers often share in the burden.
“We are just at the beginning of realizing that this is a real problem,” said Fumiko Chino, MD, a radiation oncologist at Memorial Sloan Kettering Cancer Center, New York.
Dr. Chino and colleagues recently showed that family members of patients with cancer were more likely to delay or forgo medical care than family members of people without cancer. The study found the effect was greatest among family members of younger adults with cancer.
“The caregiver and family burden related to cancer diagnosis and treatment is really underappreciated,” said Dr. Chino. “Family members and caregivers are neglecting their own health concerns, passing up career opportunities, struggling with financial concerns.”
Dr. Chino speaks from personal experience. When her fiancé, later husband, was diagnosed with neuroendocrine carcinoma in 2005, Dr. Chino quit her job as art director at a television production company to take care of him.
The couple, both in their 20s, struggled to afford his care. Dr. Chino put her own dental, medical, and mental health care on hold. She never, for instance, went to physical therapy to address injuries sustained sleeping in hospital chairs and moving around her husband who was over 6 feet tall. At one point, she walked with a limp.
Dr. Chino’s husband passed away in 2007, and even 15 years later, her injury from sleeping in hospital chairs remains “a significant physical burden,” she said. But like many caregivers “I wasn’t really thinking about my own health.”
Danielle Hadfield, 35, an ED nurse in Rochester, N.Y., also delayed her own care when her mom got sick.
Ms. Hadfield quit her job shortly after her mom was diagnosed with cholangiocarcinoma in August 2020. Ms. Hadfield knew her mom, who lived 3.5 hours away in Albany, N.Y., would need a lot of care in the upcoming months.
“I knew this was going to be the last year or so of her life, and I wanted to be there for her,” said Ms. Hadfield.
When Ms. Hadfield quit her job, she and her husband – who was self-employed – purchased health insurance coverage through the New York state marketplace. The monthly insurance payments for Ms. Hadfield, who was pregnant with her second child, her husband, and their toddler cost as much as the family’s monthly mortgage payments.
In addition to providing childcare for her young daughter and making frequent trips to Albany, Ms. Hadfield began a side business as a legal nurse consultant, working mostly at night, to replace a portion of her lost income. During this time, she began to experience pain attacks that would migrate through her body along with intermittent tongue and facial numbness. She ignored these health issues for nearly a year, until after her mother died in November 2021.
Only after her mother passed away did Ms. Hadfield begin seeking answers to her own pain. In September 2022, she finally got them. She had a nerve condition called small-fiber sensory neuropathy.
But even with a diagnosis, she is still facing more tests to root out the cause and understand the best treatment.
Is help out there?
What can physicians do to help patients and families at risk for financial toxicity?
Specific guidelines for dealing with financial toxicity do not exist in most professional guidelines, nor are there standard screening tools to identify it, said Dr. Li Smith.
These gaps put pressure on physicians to ask about financial barriers and concerns, but most do not know how to broach the topic or how to help. “Physicians may not know how to fix the problem or what resources exist,” Dr. Li Smith said.
Patients and family members, on the other hand, are often reluctant to bring up cost with physicians. Some may be ashamed to talk about their financial problems while others may fear doing so will prevent them from being offered the best possible treatments, said Ms. MacDonald.
But, experts say, financial toxicity needs to be dealt with head on. That means involving financial navigators or counselors and social workers who can, for instance, help patients and families find financial support for their basic living expenses.
From a research perspective, more clinical trials should include financial toxicity outcomes, said Joshua Palmer, MD, a radiation oncologist at the University of Michigan, Ann Arbor.
Dr. Palmer and colleagues recently showed that the number of radiation therapy clinical trials including financial toxicity endpoints increased significantly from 2001 to 2020, though the absolute rate of inclusion remains low, at roughly 1.5% of radiation therapy-based clinical trials including financial toxicity endpoints from 2016 to 2020.
“Financial burden is part of the broader discussion about shared decision-making,” said Dr. Palmer.
In shared decision-making, physicians discuss the risks and benefits of different treatment options, empowering the patient to make an informed choice with the physician.
What we want to avoid is patients feeling like they will get inferior care, if they have financial barriers, said Dr. Palmer.
And every little bit can help. In 2006, Ms. MacDonald started the Pink Fund – a nonprofit to help patients with cancer cover nonmedical cost-of-living expenses. Both Ms. Caputo and Ms. Dicks received grants from the Pink Fund. For Ms. Caputo, the funds covered 2 months of car payments and for Ms. Dicks, it covered 2 months of rent.
While the one-time grant was a big help, said Ms. Dicks, “cancer is an everyday thing.” And “we all deserve peace of mind” when trying to heal.
A version of this article first appeared on WebMD.com.
Vitamin D deficiency linked to death, new study finds
Vitamin D deficiency increases mortality risk and raising levels even slightly could decrease the risk, researchers examining data from the UK Biobank have found.
They used a Mendelian randomization approach, which uses genetic variants as “proxy indicators” for external factors that affect vitamin D levels, such as sun exposure or dietary intake. It allows for analysis of the relationship between deficiency and outcomes including mortality, which can’t be done in randomized clinical trials for ethical reasons.
Using this method, nutritionist Joshua P. Sutherland, PhD, of the Australian Centre for Precision Health, Adelaide, and colleagues found an association between genetically predicted vitamin D levels [25-(OH)D] and mortality from several major causes, with evidence of causality among people with measured concentrations below, but not above, 50 nmol/L. The findings were published online in Annals of Internal Medicine.
“Unlike other types of observational studies, we have overcome some of the methodological obstacles. What is special about this new study is we were able to look at people with very low vitamin D concentrations and what would happen if their concentrations were a little bit higher. Most randomized controlled trials don’t show much of an effect. That’s because most people have sufficient concentrations. Ethically you can’t do a trial of people with very low levels without treating them,” senior author Elina Hypp
The data support the 50 nmol/L cut-off endorsed by the United States National Academy of Medicine and align with previous data suggesting the benefit of vitamin D supplementation is largely seen in people with deficiency.
“Everybody with vitamin D levels less than 50 nmol/L is recommended to increase their levels. Our results suggest there’s no need to go very high. The positive message is that if we are able to raise levels to just the current U.S. recommendations, that’s fine. There’s no need to use large supplement doses,” Dr. Hyppönen explained.
Thus, she advised, “Supplementation will clearly help, especially during wintertime or if a person isn’t getting enough vitamin D from the sun or in places where food isn’t fortified with vitamin D.”
But the data don’t support the approach of using large intermittent doses, she added.
“Sometimes doctors want to fix the deficiency quickly with a large ‘bolus’ dose, then continue with a maintenance dose. Increasing evidence suggests that’s not beneficial and might disturb the body’s metabolism so that it can’t get the amount it needs. It’s safe overall but might not work the way we want it to work.”
Rather, Dr. Hyppönen said, “My sense is that daily modest vitamin D dose supplementation when it’s needed is the best way forward.”
Genetic approach reveals causal relationship
The investigators analyzed data from 307,601 individuals in the UK Biobank, a prospective cohort of people recruited from England, Scotland, and Wales during March 2006 and July 2010. Most were of White European ancestry and were aged 37-73 years at baseline.
Genetically predicted vitamin D levels were estimated using 35 confirmed 25-(OH)D variants. Participants were followed for outcomes up to June 2020.
The average baseline measured 25-(OH)D concentration was 45.2 nmol/L, and 11.7% (n = 36,009) of participants had levels between 10.0 and 24.9 nmol/L. Higher levels were seen in people living in southern areas and nonsmokers as well as those with a higher level of physical activity, less socioeconomic deprivation, and lower body mass index.
During follow-up, 6.1% of participants died (n = 18,700). After adjustment for variables, odds ratios for all causes of mortality were highest among people with 25-(OH)D levels below 25 nmol/L and appeared to plateau between 50 and 75 nmol/L, with no further reduction in mortality at values of 75-125 nmol/L.
Mortality 36% higher in those deficient in vitamin D
The risk for mortality was a significant 36% higher for participants with 25-(OH)D 25 nmol/L compared with 50 nmol/L.
With the Mendelian randomization, there was an L-shaped association between genetically predicted 25-(OH)D level and all-cause mortality (P for nonlinearity < .001) and for mortality because of cancer and cardiovascular disease (P for nonlinearity ≤ .033).
Again, the strongest association with those outcomes and genetically predicted 25-(OH)D was found at levels below 25 nmol/L and a plateau was seen by 50 nmol/L.
Compared with a measured 25-(OH)D concentration of 50 nmol/L, investigators estimated that the genetically predicted odds of all-cause mortality would increase sixfold (odds ratio, 6.00) for participants at 10 nmol/L and by 25% (OR, 1.25) for those at 25 nmol/L.
And, compared with a measured 25-(OH)D concentration of 50 nmol/L, those with 10 nmol/L had genetically predicted odds ratios of 5.98 for cardiovascular mortality, 3.37 for cancer mortality, and 12.44 for respiratory mortality.
Comparing measured 25-(OH)D concentrations of 25 nmol/L versus 50 nmol/L, odds ratios for those outcomes were 1.25, 1.16, and 1.96 (95% confidence interval, 1.88-4.67), respectively. All were statistically significant.
Consistent results supportive of a causal effect of genetically predicted 25-(OH)D on all-cause mortality in those with low measured vitamin D concentrations were also found in a sensitivity analysis of 20,837 people of non-White ethnic origin.
The study was funded by the Australian National Health and Medical Research Council. Dr. Sutherland’s studentship is funded by an Australian Research Training Program Scholarship.
A version of this article first appeared on Medscape.com.
Vitamin D deficiency increases mortality risk and raising levels even slightly could decrease the risk, researchers examining data from the UK Biobank have found.
They used a Mendelian randomization approach, which uses genetic variants as “proxy indicators” for external factors that affect vitamin D levels, such as sun exposure or dietary intake. It allows for analysis of the relationship between deficiency and outcomes including mortality, which can’t be done in randomized clinical trials for ethical reasons.
Using this method, nutritionist Joshua P. Sutherland, PhD, of the Australian Centre for Precision Health, Adelaide, and colleagues found an association between genetically predicted vitamin D levels [25-(OH)D] and mortality from several major causes, with evidence of causality among people with measured concentrations below, but not above, 50 nmol/L. The findings were published online in Annals of Internal Medicine.
“Unlike other types of observational studies, we have overcome some of the methodological obstacles. What is special about this new study is we were able to look at people with very low vitamin D concentrations and what would happen if their concentrations were a little bit higher. Most randomized controlled trials don’t show much of an effect. That’s because most people have sufficient concentrations. Ethically you can’t do a trial of people with very low levels without treating them,” senior author Elina Hypp
The data support the 50 nmol/L cut-off endorsed by the United States National Academy of Medicine and align with previous data suggesting the benefit of vitamin D supplementation is largely seen in people with deficiency.
“Everybody with vitamin D levels less than 50 nmol/L is recommended to increase their levels. Our results suggest there’s no need to go very high. The positive message is that if we are able to raise levels to just the current U.S. recommendations, that’s fine. There’s no need to use large supplement doses,” Dr. Hyppönen explained.
Thus, she advised, “Supplementation will clearly help, especially during wintertime or if a person isn’t getting enough vitamin D from the sun or in places where food isn’t fortified with vitamin D.”
But the data don’t support the approach of using large intermittent doses, she added.
“Sometimes doctors want to fix the deficiency quickly with a large ‘bolus’ dose, then continue with a maintenance dose. Increasing evidence suggests that’s not beneficial and might disturb the body’s metabolism so that it can’t get the amount it needs. It’s safe overall but might not work the way we want it to work.”
Rather, Dr. Hyppönen said, “My sense is that daily modest vitamin D dose supplementation when it’s needed is the best way forward.”
Genetic approach reveals causal relationship
The investigators analyzed data from 307,601 individuals in the UK Biobank, a prospective cohort of people recruited from England, Scotland, and Wales during March 2006 and July 2010. Most were of White European ancestry and were aged 37-73 years at baseline.
Genetically predicted vitamin D levels were estimated using 35 confirmed 25-(OH)D variants. Participants were followed for outcomes up to June 2020.
The average baseline measured 25-(OH)D concentration was 45.2 nmol/L, and 11.7% (n = 36,009) of participants had levels between 10.0 and 24.9 nmol/L. Higher levels were seen in people living in southern areas and nonsmokers as well as those with a higher level of physical activity, less socioeconomic deprivation, and lower body mass index.
During follow-up, 6.1% of participants died (n = 18,700). After adjustment for variables, odds ratios for all causes of mortality were highest among people with 25-(OH)D levels below 25 nmol/L and appeared to plateau between 50 and 75 nmol/L, with no further reduction in mortality at values of 75-125 nmol/L.
Mortality 36% higher in those deficient in vitamin D
The risk for mortality was a significant 36% higher for participants with 25-(OH)D 25 nmol/L compared with 50 nmol/L.
With the Mendelian randomization, there was an L-shaped association between genetically predicted 25-(OH)D level and all-cause mortality (P for nonlinearity < .001) and for mortality because of cancer and cardiovascular disease (P for nonlinearity ≤ .033).
Again, the strongest association with those outcomes and genetically predicted 25-(OH)D was found at levels below 25 nmol/L and a plateau was seen by 50 nmol/L.
Compared with a measured 25-(OH)D concentration of 50 nmol/L, investigators estimated that the genetically predicted odds of all-cause mortality would increase sixfold (odds ratio, 6.00) for participants at 10 nmol/L and by 25% (OR, 1.25) for those at 25 nmol/L.
And, compared with a measured 25-(OH)D concentration of 50 nmol/L, those with 10 nmol/L had genetically predicted odds ratios of 5.98 for cardiovascular mortality, 3.37 for cancer mortality, and 12.44 for respiratory mortality.
Comparing measured 25-(OH)D concentrations of 25 nmol/L versus 50 nmol/L, odds ratios for those outcomes were 1.25, 1.16, and 1.96 (95% confidence interval, 1.88-4.67), respectively. All were statistically significant.
Consistent results supportive of a causal effect of genetically predicted 25-(OH)D on all-cause mortality in those with low measured vitamin D concentrations were also found in a sensitivity analysis of 20,837 people of non-White ethnic origin.
The study was funded by the Australian National Health and Medical Research Council. Dr. Sutherland’s studentship is funded by an Australian Research Training Program Scholarship.
A version of this article first appeared on Medscape.com.
Vitamin D deficiency increases mortality risk and raising levels even slightly could decrease the risk, researchers examining data from the UK Biobank have found.
They used a Mendelian randomization approach, which uses genetic variants as “proxy indicators” for external factors that affect vitamin D levels, such as sun exposure or dietary intake. It allows for analysis of the relationship between deficiency and outcomes including mortality, which can’t be done in randomized clinical trials for ethical reasons.
Using this method, nutritionist Joshua P. Sutherland, PhD, of the Australian Centre for Precision Health, Adelaide, and colleagues found an association between genetically predicted vitamin D levels [25-(OH)D] and mortality from several major causes, with evidence of causality among people with measured concentrations below, but not above, 50 nmol/L. The findings were published online in Annals of Internal Medicine.
“Unlike other types of observational studies, we have overcome some of the methodological obstacles. What is special about this new study is we were able to look at people with very low vitamin D concentrations and what would happen if their concentrations were a little bit higher. Most randomized controlled trials don’t show much of an effect. That’s because most people have sufficient concentrations. Ethically you can’t do a trial of people with very low levels without treating them,” senior author Elina Hypp
The data support the 50 nmol/L cut-off endorsed by the United States National Academy of Medicine and align with previous data suggesting the benefit of vitamin D supplementation is largely seen in people with deficiency.
“Everybody with vitamin D levels less than 50 nmol/L is recommended to increase their levels. Our results suggest there’s no need to go very high. The positive message is that if we are able to raise levels to just the current U.S. recommendations, that’s fine. There’s no need to use large supplement doses,” Dr. Hyppönen explained.
Thus, she advised, “Supplementation will clearly help, especially during wintertime or if a person isn’t getting enough vitamin D from the sun or in places where food isn’t fortified with vitamin D.”
But the data don’t support the approach of using large intermittent doses, she added.
“Sometimes doctors want to fix the deficiency quickly with a large ‘bolus’ dose, then continue with a maintenance dose. Increasing evidence suggests that’s not beneficial and might disturb the body’s metabolism so that it can’t get the amount it needs. It’s safe overall but might not work the way we want it to work.”
Rather, Dr. Hyppönen said, “My sense is that daily modest vitamin D dose supplementation when it’s needed is the best way forward.”
Genetic approach reveals causal relationship
The investigators analyzed data from 307,601 individuals in the UK Biobank, a prospective cohort of people recruited from England, Scotland, and Wales during March 2006 and July 2010. Most were of White European ancestry and were aged 37-73 years at baseline.
Genetically predicted vitamin D levels were estimated using 35 confirmed 25-(OH)D variants. Participants were followed for outcomes up to June 2020.
The average baseline measured 25-(OH)D concentration was 45.2 nmol/L, and 11.7% (n = 36,009) of participants had levels between 10.0 and 24.9 nmol/L. Higher levels were seen in people living in southern areas and nonsmokers as well as those with a higher level of physical activity, less socioeconomic deprivation, and lower body mass index.
During follow-up, 6.1% of participants died (n = 18,700). After adjustment for variables, odds ratios for all causes of mortality were highest among people with 25-(OH)D levels below 25 nmol/L and appeared to plateau between 50 and 75 nmol/L, with no further reduction in mortality at values of 75-125 nmol/L.
Mortality 36% higher in those deficient in vitamin D
The risk for mortality was a significant 36% higher for participants with 25-(OH)D 25 nmol/L compared with 50 nmol/L.
With the Mendelian randomization, there was an L-shaped association between genetically predicted 25-(OH)D level and all-cause mortality (P for nonlinearity < .001) and for mortality because of cancer and cardiovascular disease (P for nonlinearity ≤ .033).
Again, the strongest association with those outcomes and genetically predicted 25-(OH)D was found at levels below 25 nmol/L and a plateau was seen by 50 nmol/L.
Compared with a measured 25-(OH)D concentration of 50 nmol/L, investigators estimated that the genetically predicted odds of all-cause mortality would increase sixfold (odds ratio, 6.00) for participants at 10 nmol/L and by 25% (OR, 1.25) for those at 25 nmol/L.
And, compared with a measured 25-(OH)D concentration of 50 nmol/L, those with 10 nmol/L had genetically predicted odds ratios of 5.98 for cardiovascular mortality, 3.37 for cancer mortality, and 12.44 for respiratory mortality.
Comparing measured 25-(OH)D concentrations of 25 nmol/L versus 50 nmol/L, odds ratios for those outcomes were 1.25, 1.16, and 1.96 (95% confidence interval, 1.88-4.67), respectively. All were statistically significant.
Consistent results supportive of a causal effect of genetically predicted 25-(OH)D on all-cause mortality in those with low measured vitamin D concentrations were also found in a sensitivity analysis of 20,837 people of non-White ethnic origin.
The study was funded by the Australian National Health and Medical Research Council. Dr. Sutherland’s studentship is funded by an Australian Research Training Program Scholarship.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
FDA OKs teclistamab for relapsed/refractory multiple myeloma
The results made the case for “teclistamab as a monotherapy for eligible patients with heavily pretreated multiple myeloma, in need of new treatment options,” investigator Maria-Victoria Mateos, MD, PhD, a hematologist at the University Hospital of Salamanca (Spain) said in a June press release from drug maker Janssen/Johnson & Johnson.
The approval was based on the phase 1-2 single-arm MajesTEC-1 trial. The MajesTEC-1 findings, published in August in the New England Journal of Medicine, included 165 patients with relapsed or refractory multiple myeloma after at least three therapy lines – including an immunomodulatory drug, a proteasome inhibitor, and an anti-CD38 antibody. These patients received a weekly subcutaneous 1.5 mg/kg injection of teclistamab after stepping up from 0.06 mg and 0.3 mg/kg doses.
According to the FDA announcement, the overall response rate was nearly 62%. And the estimated duration of response rate among responders was 90.6% at 6 months and 66.5% at 9 months.
The NEJM results also indicated that almost 40% of patients had a complete response to the therapy, over a median follow-up of 14.1 months. More than a quarter of patients (26.7%) had no minimal residual disease.
The study investigators concluded that “teclistamab resulted in a high rate of deep and durable response in patients with triple-class exposed relapsed or refractory multiple myeloma.”
In a press release, Michael Andreini, president and CEO of the Multiple Myeloma Research Foundation, commented that “teclistamab is an important new treatment option for patients who have faced multiple relapses.”
The recommended dose for teclistamab is 0.06 mg/kg via subcutaneous injection on day 1, 0.3 mg/kg on day 4, and 1.5 mg/kg on day 7, followed by 1.5 mg/kg once weekly until disease progression or unacceptable toxicity.
The FDA noted, however, that the prescribing information for teclistamab comes with a Boxed warning for “life-threatening or fatal cytokine-release syndrome (CRS) and neurologic toxicity, including immune effector cell–associated neurotoxicity (ICANS).”
CRS was reported in 72.1% of patients (grade 3 in one patient but no grade 4 cases), neurologic toxicity in 57%, and ICANS in 6%. Other common adverse events in the NEJM report included neutropenia in 71% of subjects (grade 3 or 4 in 64%); anemia in 52% (grade 3 or 4 in 37%), and thrombocytopenia in 40% (grade 3 or 4 in 21%). Overall, 45% of patients developed grade 3 or 4 infections.
“Because of the risks of CRS and neurologic toxicity, including ICANS, teclistamab-cqyv is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), called the Tecvayli REMS,” according to the FDA’s press release.
Teclistamab is a T-cell–bispecific antibody that targets both CD3 expressed on the surface of T cells and B-cell maturation antigen (BCMA) expressed on the surface of myeloma cells, activating T-cells to kill cancer cells that express the antigen.
Three BCMA-directed therapies are already on the market in the United States that carry teclistamab’s indication: the antibody-drug conjugate belantamab mafodotin (Blenrep) and two chimeric antigen receptor (CAR) T-cell therapies, idecabtagene vicleucel (Abecma) and ciltacabtagene autoleucel (Carvykti).
The overall response rate is approximately 31% with belantamab mafodotin, 67% for idecabtagene vicleucel, and 83% for ciltacabtagene autoleucel.
Pfizer also has a bispecific BCMA-CD3–targeted antibody in development, elranatamab, for triple-class refractory multiple myeloma that is expected to compete with teclistamab.
MajesTEC-1 was funded by Janssen. Dr. Mateos is a paid speaker and consultant for the company.
A version of this article first appeared on Medscape.com.
The results made the case for “teclistamab as a monotherapy for eligible patients with heavily pretreated multiple myeloma, in need of new treatment options,” investigator Maria-Victoria Mateos, MD, PhD, a hematologist at the University Hospital of Salamanca (Spain) said in a June press release from drug maker Janssen/Johnson & Johnson.
The approval was based on the phase 1-2 single-arm MajesTEC-1 trial. The MajesTEC-1 findings, published in August in the New England Journal of Medicine, included 165 patients with relapsed or refractory multiple myeloma after at least three therapy lines – including an immunomodulatory drug, a proteasome inhibitor, and an anti-CD38 antibody. These patients received a weekly subcutaneous 1.5 mg/kg injection of teclistamab after stepping up from 0.06 mg and 0.3 mg/kg doses.
According to the FDA announcement, the overall response rate was nearly 62%. And the estimated duration of response rate among responders was 90.6% at 6 months and 66.5% at 9 months.
The NEJM results also indicated that almost 40% of patients had a complete response to the therapy, over a median follow-up of 14.1 months. More than a quarter of patients (26.7%) had no minimal residual disease.
The study investigators concluded that “teclistamab resulted in a high rate of deep and durable response in patients with triple-class exposed relapsed or refractory multiple myeloma.”
In a press release, Michael Andreini, president and CEO of the Multiple Myeloma Research Foundation, commented that “teclistamab is an important new treatment option for patients who have faced multiple relapses.”
The recommended dose for teclistamab is 0.06 mg/kg via subcutaneous injection on day 1, 0.3 mg/kg on day 4, and 1.5 mg/kg on day 7, followed by 1.5 mg/kg once weekly until disease progression or unacceptable toxicity.
The FDA noted, however, that the prescribing information for teclistamab comes with a Boxed warning for “life-threatening or fatal cytokine-release syndrome (CRS) and neurologic toxicity, including immune effector cell–associated neurotoxicity (ICANS).”
CRS was reported in 72.1% of patients (grade 3 in one patient but no grade 4 cases), neurologic toxicity in 57%, and ICANS in 6%. Other common adverse events in the NEJM report included neutropenia in 71% of subjects (grade 3 or 4 in 64%); anemia in 52% (grade 3 or 4 in 37%), and thrombocytopenia in 40% (grade 3 or 4 in 21%). Overall, 45% of patients developed grade 3 or 4 infections.
“Because of the risks of CRS and neurologic toxicity, including ICANS, teclistamab-cqyv is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), called the Tecvayli REMS,” according to the FDA’s press release.
Teclistamab is a T-cell–bispecific antibody that targets both CD3 expressed on the surface of T cells and B-cell maturation antigen (BCMA) expressed on the surface of myeloma cells, activating T-cells to kill cancer cells that express the antigen.
Three BCMA-directed therapies are already on the market in the United States that carry teclistamab’s indication: the antibody-drug conjugate belantamab mafodotin (Blenrep) and two chimeric antigen receptor (CAR) T-cell therapies, idecabtagene vicleucel (Abecma) and ciltacabtagene autoleucel (Carvykti).
The overall response rate is approximately 31% with belantamab mafodotin, 67% for idecabtagene vicleucel, and 83% for ciltacabtagene autoleucel.
Pfizer also has a bispecific BCMA-CD3–targeted antibody in development, elranatamab, for triple-class refractory multiple myeloma that is expected to compete with teclistamab.
MajesTEC-1 was funded by Janssen. Dr. Mateos is a paid speaker and consultant for the company.
A version of this article first appeared on Medscape.com.
The results made the case for “teclistamab as a monotherapy for eligible patients with heavily pretreated multiple myeloma, in need of new treatment options,” investigator Maria-Victoria Mateos, MD, PhD, a hematologist at the University Hospital of Salamanca (Spain) said in a June press release from drug maker Janssen/Johnson & Johnson.
The approval was based on the phase 1-2 single-arm MajesTEC-1 trial. The MajesTEC-1 findings, published in August in the New England Journal of Medicine, included 165 patients with relapsed or refractory multiple myeloma after at least three therapy lines – including an immunomodulatory drug, a proteasome inhibitor, and an anti-CD38 antibody. These patients received a weekly subcutaneous 1.5 mg/kg injection of teclistamab after stepping up from 0.06 mg and 0.3 mg/kg doses.
According to the FDA announcement, the overall response rate was nearly 62%. And the estimated duration of response rate among responders was 90.6% at 6 months and 66.5% at 9 months.
The NEJM results also indicated that almost 40% of patients had a complete response to the therapy, over a median follow-up of 14.1 months. More than a quarter of patients (26.7%) had no minimal residual disease.
The study investigators concluded that “teclistamab resulted in a high rate of deep and durable response in patients with triple-class exposed relapsed or refractory multiple myeloma.”
In a press release, Michael Andreini, president and CEO of the Multiple Myeloma Research Foundation, commented that “teclistamab is an important new treatment option for patients who have faced multiple relapses.”
The recommended dose for teclistamab is 0.06 mg/kg via subcutaneous injection on day 1, 0.3 mg/kg on day 4, and 1.5 mg/kg on day 7, followed by 1.5 mg/kg once weekly until disease progression or unacceptable toxicity.
The FDA noted, however, that the prescribing information for teclistamab comes with a Boxed warning for “life-threatening or fatal cytokine-release syndrome (CRS) and neurologic toxicity, including immune effector cell–associated neurotoxicity (ICANS).”
CRS was reported in 72.1% of patients (grade 3 in one patient but no grade 4 cases), neurologic toxicity in 57%, and ICANS in 6%. Other common adverse events in the NEJM report included neutropenia in 71% of subjects (grade 3 or 4 in 64%); anemia in 52% (grade 3 or 4 in 37%), and thrombocytopenia in 40% (grade 3 or 4 in 21%). Overall, 45% of patients developed grade 3 or 4 infections.
“Because of the risks of CRS and neurologic toxicity, including ICANS, teclistamab-cqyv is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS), called the Tecvayli REMS,” according to the FDA’s press release.
Teclistamab is a T-cell–bispecific antibody that targets both CD3 expressed on the surface of T cells and B-cell maturation antigen (BCMA) expressed on the surface of myeloma cells, activating T-cells to kill cancer cells that express the antigen.
Three BCMA-directed therapies are already on the market in the United States that carry teclistamab’s indication: the antibody-drug conjugate belantamab mafodotin (Blenrep) and two chimeric antigen receptor (CAR) T-cell therapies, idecabtagene vicleucel (Abecma) and ciltacabtagene autoleucel (Carvykti).
The overall response rate is approximately 31% with belantamab mafodotin, 67% for idecabtagene vicleucel, and 83% for ciltacabtagene autoleucel.
Pfizer also has a bispecific BCMA-CD3–targeted antibody in development, elranatamab, for triple-class refractory multiple myeloma that is expected to compete with teclistamab.
MajesTEC-1 was funded by Janssen. Dr. Mateos is a paid speaker and consultant for the company.
A version of this article first appeared on Medscape.com.
Achieving diversity, equity and inclusion: Invite everyone and build a team
What you really don’t want to do, if you want to improve diversity, equity, and inclusion (DEI) at your academic institution, is to recruit diverse people to your program and then have them come and feel not included, said Vivian Asare, MD. “That can work against your efforts,” she stated in an oral presentation at the annual meeting of the American College of Chest Physicians (CHEST). Dr. Asare is assistant professor and vice chief of DEI for Yale Pulmonary, Critical Care, and Sleep Medicine, and associate medical director of Yale Centers for Sleep Medicine, New Haven, Conn.
In offering a path to successful DEI, Dr. Asare said: “The first step is to build a team and discuss your mission. Invite everyone to participate and include your leadership because they’re the ones who set the stage, ensure sustainability, and can be a liaison with faculty.” Then a DEI leader should be elected, she added.
The next and very important step is to survey the current institutional climate. That entails speaking directly with the stakeholders (faculty, staff, trainees) and identifying their specific concerns and what they think is lacking. Retreats, serious group discussions, and self-reflecting (asking “what initiatives would be good for us?”), and meeting one-on-one with individuals for a truly personalized approach are among potentially productive strategies for identifying the priorities and DEI-related topics specific to a particular academic sleep program.
Dr. Asare offered up a sample DEI survey (Am J Obstet Gynecol. 2020 Nov;223[5]:715.e1-715.e7), that made direct statements inviting the respondent to check off one of the following responses: Yes, No, Somewhat, Do not know, and Not applicable. Among sample statements:
- Our department is actively committed to issues of diversity, equity, and inclusion.
- Faculty searches in the department regularly attract a diverse pool of highly qualified candidates and/or attract a pool that represents the availability of MDs in this field.
- Our outreach and recruitment processes employ targeted practices for attracting diverse populations.
Dr. Asare said that a survey can be a simple approach for garnering information that can be useful for prioritizing DEI topics of concern and igniting interest in them. Engagement requires regular DEI committee meetings with minutes or a newsletter and with updates and topics brought to faculty meetings.
Key DEI areas of focus
Dr. Asare listed several key DEI areas: Recruitment/retention, mentorship, scholarship, and inclusion and community engagement. Under scholarship, for example, she cited topics for potential inclusion in a DEI curriculum: Unconscious bias and anti-racism training, racism, discrimination and microaggression education (bystander/deescalation training), cultural competency and awareness, workplace civility, and health disparities. “We all know that implicit bias in providers is a reality, unfortunately,” Dr. Asare said. Being aware of these implicit biases is a start, but instruction on how to actively overcome them has to be provided. Tools may include perspective-taking, exploring common identity, and self-reflection.
To create an inclusive environment for all faculty, trainees, and staff may involve establishing a “welcome committee” for new faculty, perhaps with designating a “peer buddy,” creating social events and other opportunities for all opinions and ideas to be heard and valued. Particularly for underserved and disadvantaged patient populations, patient advocacy and community service need to be fostered through support groups and provision of resources.
Summarizing, Dr. Asare reiterated several key elements for a successful DEI program: Build a team and discuss the mission, survey the current climate allowing open communication and dialogue, plan and engage, organize, and form areas of DEI focus. Find out where you are and where you want to be with respect to DEI, she concluded.
Dr. Asare declared that she had no conflicts of interest.
What you really don’t want to do, if you want to improve diversity, equity, and inclusion (DEI) at your academic institution, is to recruit diverse people to your program and then have them come and feel not included, said Vivian Asare, MD. “That can work against your efforts,” she stated in an oral presentation at the annual meeting of the American College of Chest Physicians (CHEST). Dr. Asare is assistant professor and vice chief of DEI for Yale Pulmonary, Critical Care, and Sleep Medicine, and associate medical director of Yale Centers for Sleep Medicine, New Haven, Conn.
In offering a path to successful DEI, Dr. Asare said: “The first step is to build a team and discuss your mission. Invite everyone to participate and include your leadership because they’re the ones who set the stage, ensure sustainability, and can be a liaison with faculty.” Then a DEI leader should be elected, she added.
The next and very important step is to survey the current institutional climate. That entails speaking directly with the stakeholders (faculty, staff, trainees) and identifying their specific concerns and what they think is lacking. Retreats, serious group discussions, and self-reflecting (asking “what initiatives would be good for us?”), and meeting one-on-one with individuals for a truly personalized approach are among potentially productive strategies for identifying the priorities and DEI-related topics specific to a particular academic sleep program.
Dr. Asare offered up a sample DEI survey (Am J Obstet Gynecol. 2020 Nov;223[5]:715.e1-715.e7), that made direct statements inviting the respondent to check off one of the following responses: Yes, No, Somewhat, Do not know, and Not applicable. Among sample statements:
- Our department is actively committed to issues of diversity, equity, and inclusion.
- Faculty searches in the department regularly attract a diverse pool of highly qualified candidates and/or attract a pool that represents the availability of MDs in this field.
- Our outreach and recruitment processes employ targeted practices for attracting diverse populations.
Dr. Asare said that a survey can be a simple approach for garnering information that can be useful for prioritizing DEI topics of concern and igniting interest in them. Engagement requires regular DEI committee meetings with minutes or a newsletter and with updates and topics brought to faculty meetings.
Key DEI areas of focus
Dr. Asare listed several key DEI areas: Recruitment/retention, mentorship, scholarship, and inclusion and community engagement. Under scholarship, for example, she cited topics for potential inclusion in a DEI curriculum: Unconscious bias and anti-racism training, racism, discrimination and microaggression education (bystander/deescalation training), cultural competency and awareness, workplace civility, and health disparities. “We all know that implicit bias in providers is a reality, unfortunately,” Dr. Asare said. Being aware of these implicit biases is a start, but instruction on how to actively overcome them has to be provided. Tools may include perspective-taking, exploring common identity, and self-reflection.
To create an inclusive environment for all faculty, trainees, and staff may involve establishing a “welcome committee” for new faculty, perhaps with designating a “peer buddy,” creating social events and other opportunities for all opinions and ideas to be heard and valued. Particularly for underserved and disadvantaged patient populations, patient advocacy and community service need to be fostered through support groups and provision of resources.
Summarizing, Dr. Asare reiterated several key elements for a successful DEI program: Build a team and discuss the mission, survey the current climate allowing open communication and dialogue, plan and engage, organize, and form areas of DEI focus. Find out where you are and where you want to be with respect to DEI, she concluded.
Dr. Asare declared that she had no conflicts of interest.
What you really don’t want to do, if you want to improve diversity, equity, and inclusion (DEI) at your academic institution, is to recruit diverse people to your program and then have them come and feel not included, said Vivian Asare, MD. “That can work against your efforts,” she stated in an oral presentation at the annual meeting of the American College of Chest Physicians (CHEST). Dr. Asare is assistant professor and vice chief of DEI for Yale Pulmonary, Critical Care, and Sleep Medicine, and associate medical director of Yale Centers for Sleep Medicine, New Haven, Conn.
In offering a path to successful DEI, Dr. Asare said: “The first step is to build a team and discuss your mission. Invite everyone to participate and include your leadership because they’re the ones who set the stage, ensure sustainability, and can be a liaison with faculty.” Then a DEI leader should be elected, she added.
The next and very important step is to survey the current institutional climate. That entails speaking directly with the stakeholders (faculty, staff, trainees) and identifying their specific concerns and what they think is lacking. Retreats, serious group discussions, and self-reflecting (asking “what initiatives would be good for us?”), and meeting one-on-one with individuals for a truly personalized approach are among potentially productive strategies for identifying the priorities and DEI-related topics specific to a particular academic sleep program.
Dr. Asare offered up a sample DEI survey (Am J Obstet Gynecol. 2020 Nov;223[5]:715.e1-715.e7), that made direct statements inviting the respondent to check off one of the following responses: Yes, No, Somewhat, Do not know, and Not applicable. Among sample statements:
- Our department is actively committed to issues of diversity, equity, and inclusion.
- Faculty searches in the department regularly attract a diverse pool of highly qualified candidates and/or attract a pool that represents the availability of MDs in this field.
- Our outreach and recruitment processes employ targeted practices for attracting diverse populations.
Dr. Asare said that a survey can be a simple approach for garnering information that can be useful for prioritizing DEI topics of concern and igniting interest in them. Engagement requires regular DEI committee meetings with minutes or a newsletter and with updates and topics brought to faculty meetings.
Key DEI areas of focus
Dr. Asare listed several key DEI areas: Recruitment/retention, mentorship, scholarship, and inclusion and community engagement. Under scholarship, for example, she cited topics for potential inclusion in a DEI curriculum: Unconscious bias and anti-racism training, racism, discrimination and microaggression education (bystander/deescalation training), cultural competency and awareness, workplace civility, and health disparities. “We all know that implicit bias in providers is a reality, unfortunately,” Dr. Asare said. Being aware of these implicit biases is a start, but instruction on how to actively overcome them has to be provided. Tools may include perspective-taking, exploring common identity, and self-reflection.
To create an inclusive environment for all faculty, trainees, and staff may involve establishing a “welcome committee” for new faculty, perhaps with designating a “peer buddy,” creating social events and other opportunities for all opinions and ideas to be heard and valued. Particularly for underserved and disadvantaged patient populations, patient advocacy and community service need to be fostered through support groups and provision of resources.
Summarizing, Dr. Asare reiterated several key elements for a successful DEI program: Build a team and discuss the mission, survey the current climate allowing open communication and dialogue, plan and engage, organize, and form areas of DEI focus. Find out where you are and where you want to be with respect to DEI, she concluded.
Dr. Asare declared that she had no conflicts of interest.
FROM CHEST 2022
Gloom lifting as MCL treatments evolve
Traditionally, MCL has had a notoriously poor prognosis and is still impossible to cure. But survival rates are rising thanks to better treatments, the review authors wrote, and even relapsed/refractory patients have a growing number of options that can potentially give them extra years of life.
“Prognosis has certainly changed in past 10 years. We have been able to have an excellent control of disease, and patients are living longer, even past the 8- or 10-year mark,” Moffit Cancer Center/Memorial Healthcare System hematologist-oncologist Jose Sandoval‐Sus, MD, said in an interview. He is corresponding author of the review, which appeared in the October issue of Current Oncology Reports.
MCL – which affects cells in the mantle zone of lymph nodes – is rare. It usually strikes older men, often presents at an advanced stage, and accounts for 6%-8% of non-Hodgkin lymphomas in the United States.
Prognoses are improving. The review highlights a study released earlier this year that found that median 5-year overall survival has increased from 68.8% (2002-2009) to 81.6% (2010-2015).
Now, the review notes, there are several first-line chemotherapy options that combine agents with rituximab such as rituximab/bendamustine, which “has generally been established as an effective treatment for MCL at first relapse in patients who are bendamustine naive when compared to other chemotherapy agents.”
Other treatments include rituximab, bortezomib, cyclophosphamide, doxorubicin, vincristine, and prednisolone; rituximab, bendamustine and cytarabine; and rituximab, gemcitabine, and oxaliplatin.
“I think of rituximab as a medication of maintenance, either after autologous stem cell transplant or even in patients who have not been through transplant,” Dr. Sandoval‐Sus said. “As maintenance, it really has improved outcomes for these patients.”
But the first step before treatment, he said, is to explore prognostic factors such as alterations on the TP53 gene that “really dictate a lot in terms of the prognosis of patients.” As the review notes, these alterations – either bi-allelic del17p or TP53 mutations – “are associated with poor outcomes after frontline and salvage regimens, including targeted agents such as Burton’s tyrosine kinase inhibitors (BTKis).”
These patients, who make up about 20% of those with MCL, also are most unlikely to benefit from autologous stem cell transplantation, Dr. Sandoval‐Sus said.
What about refracted/relapsed (R/R) cases? BTKis have been a major advance for these patients, he said. However, choosing the best drug can be a challenge. As the review notes, “all approved BTKis for R/R MCL seem to have similar clinical outcomes based on identical mechanism of action, and there are no prospective trials comparing these agents in a head-to-head fashion.”
The authors added that “we wonder if AEs [adverse events] could be decreased by using combinations based on new generation BTKi, but it is still a question that needs to be resolved in the clinical trial arena.”
Stem cell transplants may be an option, the review said, but “in practice the clinical benefit ... is limited to single-center series or small multi-institutional registries with few prospective studies.”
Then there’s CAR-T cell therapy, the game-changer. A type called brexucabtagene autoleucel (Brexu-cel) is now approved in MCL, the review authors wrote, and real-world data “serve as a platform to expand CAR-T therapy to more R/R MCL patients that do not fit the strict inclusion criteria of the studies (e.g., controlled comorbidities and worse performance status)... We strongly recommend early referral of these patients to accredited institutions with ample cellular therapy experience, including high-risk MCL patients (e.g., blastoid/pleomorphic morphology, biallelic del17p, TP53 mutations) so an appropriate bridging strategy and a CAR-T cell roadmap is planned with the patient and caretakers.”
Some researchers are exploring combination treatment with both BTKis and CAR T-cell therapy, “which may be considered for patients with R/R MCL who are naive to both CAR T-cell and BTKi therapy, because combination therapy may increase treatment efficacy,” wrote the authors of another review that appeared in the October issue of Current Oncology Reports. “Based on limited data in patients with CLL, BTKi therapy may be initiated as bridging therapy and continued during lymphodepletion prior to CAR T-cell infusion”
What’s next? Multiple treatments are in the research stage, Dr. Sandoval‐Sus said. “There are a lot of things in development that are really incredible.”
Reversible BTKis, for example, appear to be effective at controlling disease and are well-tolerated, he said. “And we are awaiting the results of clinical trials of targeted therapies.”
For now, he said, the best advice for hematologists is to gain a full understanding of a patient’s MCL, in order to provide the most appropriate treatment. Community oncologists should get at least one second opinion from an academic center or other clinic that treats these kinds of lymphomas, he said, and molecular tests are crucial. A discussion about stem cell transplantation after remission is a good idea, he said, and so is an exploration of clinical trials “from the get-go.”
“In patients who relapse and have high-risk features, they should be started on a BTKi inhibitor for the most part,” he said, “although we need to weigh risks and benefits between the side effects of different BTKi inhibitors. And they should be referred earlier to a CAR T cell therapy center, so they can discuss the benefits and see if they’re an appropriate patient. I think patients are being referred a little bit too late in the second- or third-line setting.”
What about CAR T therapy as a first-line therapy? It’s not FDA-approved, Dr. Sandoval‐Sus said, and “definitely not a standard of care.” But clinical trials are exploring the idea, he said. As for messages to patients, Dr. Sandoval-Sus said he would tell them that MCL is not yet curable, “but the future is very bright.”
Dr. Sandoval-Sus declared advisory board relationships with Seagen, Incyte, Janssen, ADC Therapeutics, TG therapeutics, and Genmab. The other review authors had no disclosures.
Traditionally, MCL has had a notoriously poor prognosis and is still impossible to cure. But survival rates are rising thanks to better treatments, the review authors wrote, and even relapsed/refractory patients have a growing number of options that can potentially give them extra years of life.
“Prognosis has certainly changed in past 10 years. We have been able to have an excellent control of disease, and patients are living longer, even past the 8- or 10-year mark,” Moffit Cancer Center/Memorial Healthcare System hematologist-oncologist Jose Sandoval‐Sus, MD, said in an interview. He is corresponding author of the review, which appeared in the October issue of Current Oncology Reports.
MCL – which affects cells in the mantle zone of lymph nodes – is rare. It usually strikes older men, often presents at an advanced stage, and accounts for 6%-8% of non-Hodgkin lymphomas in the United States.
Prognoses are improving. The review highlights a study released earlier this year that found that median 5-year overall survival has increased from 68.8% (2002-2009) to 81.6% (2010-2015).
Now, the review notes, there are several first-line chemotherapy options that combine agents with rituximab such as rituximab/bendamustine, which “has generally been established as an effective treatment for MCL at first relapse in patients who are bendamustine naive when compared to other chemotherapy agents.”
Other treatments include rituximab, bortezomib, cyclophosphamide, doxorubicin, vincristine, and prednisolone; rituximab, bendamustine and cytarabine; and rituximab, gemcitabine, and oxaliplatin.
“I think of rituximab as a medication of maintenance, either after autologous stem cell transplant or even in patients who have not been through transplant,” Dr. Sandoval‐Sus said. “As maintenance, it really has improved outcomes for these patients.”
But the first step before treatment, he said, is to explore prognostic factors such as alterations on the TP53 gene that “really dictate a lot in terms of the prognosis of patients.” As the review notes, these alterations – either bi-allelic del17p or TP53 mutations – “are associated with poor outcomes after frontline and salvage regimens, including targeted agents such as Burton’s tyrosine kinase inhibitors (BTKis).”
These patients, who make up about 20% of those with MCL, also are most unlikely to benefit from autologous stem cell transplantation, Dr. Sandoval‐Sus said.
What about refracted/relapsed (R/R) cases? BTKis have been a major advance for these patients, he said. However, choosing the best drug can be a challenge. As the review notes, “all approved BTKis for R/R MCL seem to have similar clinical outcomes based on identical mechanism of action, and there are no prospective trials comparing these agents in a head-to-head fashion.”
The authors added that “we wonder if AEs [adverse events] could be decreased by using combinations based on new generation BTKi, but it is still a question that needs to be resolved in the clinical trial arena.”
Stem cell transplants may be an option, the review said, but “in practice the clinical benefit ... is limited to single-center series or small multi-institutional registries with few prospective studies.”
Then there’s CAR-T cell therapy, the game-changer. A type called brexucabtagene autoleucel (Brexu-cel) is now approved in MCL, the review authors wrote, and real-world data “serve as a platform to expand CAR-T therapy to more R/R MCL patients that do not fit the strict inclusion criteria of the studies (e.g., controlled comorbidities and worse performance status)... We strongly recommend early referral of these patients to accredited institutions with ample cellular therapy experience, including high-risk MCL patients (e.g., blastoid/pleomorphic morphology, biallelic del17p, TP53 mutations) so an appropriate bridging strategy and a CAR-T cell roadmap is planned with the patient and caretakers.”
Some researchers are exploring combination treatment with both BTKis and CAR T-cell therapy, “which may be considered for patients with R/R MCL who are naive to both CAR T-cell and BTKi therapy, because combination therapy may increase treatment efficacy,” wrote the authors of another review that appeared in the October issue of Current Oncology Reports. “Based on limited data in patients with CLL, BTKi therapy may be initiated as bridging therapy and continued during lymphodepletion prior to CAR T-cell infusion”
What’s next? Multiple treatments are in the research stage, Dr. Sandoval‐Sus said. “There are a lot of things in development that are really incredible.”
Reversible BTKis, for example, appear to be effective at controlling disease and are well-tolerated, he said. “And we are awaiting the results of clinical trials of targeted therapies.”
For now, he said, the best advice for hematologists is to gain a full understanding of a patient’s MCL, in order to provide the most appropriate treatment. Community oncologists should get at least one second opinion from an academic center or other clinic that treats these kinds of lymphomas, he said, and molecular tests are crucial. A discussion about stem cell transplantation after remission is a good idea, he said, and so is an exploration of clinical trials “from the get-go.”
“In patients who relapse and have high-risk features, they should be started on a BTKi inhibitor for the most part,” he said, “although we need to weigh risks and benefits between the side effects of different BTKi inhibitors. And they should be referred earlier to a CAR T cell therapy center, so they can discuss the benefits and see if they’re an appropriate patient. I think patients are being referred a little bit too late in the second- or third-line setting.”
What about CAR T therapy as a first-line therapy? It’s not FDA-approved, Dr. Sandoval‐Sus said, and “definitely not a standard of care.” But clinical trials are exploring the idea, he said. As for messages to patients, Dr. Sandoval-Sus said he would tell them that MCL is not yet curable, “but the future is very bright.”
Dr. Sandoval-Sus declared advisory board relationships with Seagen, Incyte, Janssen, ADC Therapeutics, TG therapeutics, and Genmab. The other review authors had no disclosures.
Traditionally, MCL has had a notoriously poor prognosis and is still impossible to cure. But survival rates are rising thanks to better treatments, the review authors wrote, and even relapsed/refractory patients have a growing number of options that can potentially give them extra years of life.
“Prognosis has certainly changed in past 10 years. We have been able to have an excellent control of disease, and patients are living longer, even past the 8- or 10-year mark,” Moffit Cancer Center/Memorial Healthcare System hematologist-oncologist Jose Sandoval‐Sus, MD, said in an interview. He is corresponding author of the review, which appeared in the October issue of Current Oncology Reports.
MCL – which affects cells in the mantle zone of lymph nodes – is rare. It usually strikes older men, often presents at an advanced stage, and accounts for 6%-8% of non-Hodgkin lymphomas in the United States.
Prognoses are improving. The review highlights a study released earlier this year that found that median 5-year overall survival has increased from 68.8% (2002-2009) to 81.6% (2010-2015).
Now, the review notes, there are several first-line chemotherapy options that combine agents with rituximab such as rituximab/bendamustine, which “has generally been established as an effective treatment for MCL at first relapse in patients who are bendamustine naive when compared to other chemotherapy agents.”
Other treatments include rituximab, bortezomib, cyclophosphamide, doxorubicin, vincristine, and prednisolone; rituximab, bendamustine and cytarabine; and rituximab, gemcitabine, and oxaliplatin.
“I think of rituximab as a medication of maintenance, either after autologous stem cell transplant or even in patients who have not been through transplant,” Dr. Sandoval‐Sus said. “As maintenance, it really has improved outcomes for these patients.”
But the first step before treatment, he said, is to explore prognostic factors such as alterations on the TP53 gene that “really dictate a lot in terms of the prognosis of patients.” As the review notes, these alterations – either bi-allelic del17p or TP53 mutations – “are associated with poor outcomes after frontline and salvage regimens, including targeted agents such as Burton’s tyrosine kinase inhibitors (BTKis).”
These patients, who make up about 20% of those with MCL, also are most unlikely to benefit from autologous stem cell transplantation, Dr. Sandoval‐Sus said.
What about refracted/relapsed (R/R) cases? BTKis have been a major advance for these patients, he said. However, choosing the best drug can be a challenge. As the review notes, “all approved BTKis for R/R MCL seem to have similar clinical outcomes based on identical mechanism of action, and there are no prospective trials comparing these agents in a head-to-head fashion.”
The authors added that “we wonder if AEs [adverse events] could be decreased by using combinations based on new generation BTKi, but it is still a question that needs to be resolved in the clinical trial arena.”
Stem cell transplants may be an option, the review said, but “in practice the clinical benefit ... is limited to single-center series or small multi-institutional registries with few prospective studies.”
Then there’s CAR-T cell therapy, the game-changer. A type called brexucabtagene autoleucel (Brexu-cel) is now approved in MCL, the review authors wrote, and real-world data “serve as a platform to expand CAR-T therapy to more R/R MCL patients that do not fit the strict inclusion criteria of the studies (e.g., controlled comorbidities and worse performance status)... We strongly recommend early referral of these patients to accredited institutions with ample cellular therapy experience, including high-risk MCL patients (e.g., blastoid/pleomorphic morphology, biallelic del17p, TP53 mutations) so an appropriate bridging strategy and a CAR-T cell roadmap is planned with the patient and caretakers.”
Some researchers are exploring combination treatment with both BTKis and CAR T-cell therapy, “which may be considered for patients with R/R MCL who are naive to both CAR T-cell and BTKi therapy, because combination therapy may increase treatment efficacy,” wrote the authors of another review that appeared in the October issue of Current Oncology Reports. “Based on limited data in patients with CLL, BTKi therapy may be initiated as bridging therapy and continued during lymphodepletion prior to CAR T-cell infusion”
What’s next? Multiple treatments are in the research stage, Dr. Sandoval‐Sus said. “There are a lot of things in development that are really incredible.”
Reversible BTKis, for example, appear to be effective at controlling disease and are well-tolerated, he said. “And we are awaiting the results of clinical trials of targeted therapies.”
For now, he said, the best advice for hematologists is to gain a full understanding of a patient’s MCL, in order to provide the most appropriate treatment. Community oncologists should get at least one second opinion from an academic center or other clinic that treats these kinds of lymphomas, he said, and molecular tests are crucial. A discussion about stem cell transplantation after remission is a good idea, he said, and so is an exploration of clinical trials “from the get-go.”
“In patients who relapse and have high-risk features, they should be started on a BTKi inhibitor for the most part,” he said, “although we need to weigh risks and benefits between the side effects of different BTKi inhibitors. And they should be referred earlier to a CAR T cell therapy center, so they can discuss the benefits and see if they’re an appropriate patient. I think patients are being referred a little bit too late in the second- or third-line setting.”
What about CAR T therapy as a first-line therapy? It’s not FDA-approved, Dr. Sandoval‐Sus said, and “definitely not a standard of care.” But clinical trials are exploring the idea, he said. As for messages to patients, Dr. Sandoval-Sus said he would tell them that MCL is not yet curable, “but the future is very bright.”
Dr. Sandoval-Sus declared advisory board relationships with Seagen, Incyte, Janssen, ADC Therapeutics, TG therapeutics, and Genmab. The other review authors had no disclosures.
Reminder that COVID-19 and cancer can be a deadly combo
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study underscores the importance of COVID-19 and regular COVID-19 testing among adults with a recent cancer diagnosis.
The Indiana statewide study, conducted at the beginning of the pandemic, found that
“This analysis provides additional empirical evidence on the magnitude of risk to patients with cancer whose immune systems are often weakened either by the disease or treatment,” the study team wrote.
The study was published online in JMIR Cancer.
Although evidence has consistently revealed similar findings, the risk of death among unvaccinated people with cancer and COVID-19 has not been nearly as high in previous studies, lead author Brian E. Dixon, PhD, MBA, with Indiana University Richard M. Fairbanks School of Public Health, Indianapolis, said in a statement. Previous studies from China, for instance, reported a two- to threefold greater risk of all-cause mortality among unvaccinated adults with cancer and COVID-19.
A potential reason for this discrepancy, Dr. Dixon noted, is that earlier studies were “generally smaller and made calculations based on data from a single cancer center or health system.”
Another reason is testing for COVID-19 early in the pandemic was limited to symptomatic individuals who may have had more severe infections, possibly leading to an overestimate of the association between SARS-CoV-2 infection, cancer, and all-cause mortality.
In the current analysis, researchers used electronic health records linked to Indiana’s statewide SARS-CoV-2 testing database and state vital records to evaluate the association between SARS-CoV-2 infection and all-cause mortality among 41,924 adults newly diagnosed with cancer between Jan. 1, 2019, and Dec. 31, 2020.
Most people with cancer were White (78.4%) and about half were male. At the time of diagnosis, 17% had one comorbid condition and about 10% had two or more. Most patients had breast cancer (14%), prostate cancer (13%), or melanoma (13%).
During the study period, 2,894 patients (7%) tested positive for SARS-CoV-2.
In multivariate adjusted analysis, the risk of death among those newly diagnosed with cancer increased by 91% (adjusted hazard ratio, 1.91) during the first year of the pandemic before vaccines were available, compared with the year before (January 2019 to Jan. 14, 2020).
During the pandemic period, the risk of death was roughly threefold higher among adults 65 years old and older, compared with adults 18-44 years old (aHR, 3.35).
When looking at the time from a cancer diagnosis to SARS-CoV-2 infection, infection was associated with an almost sevenfold increase in all-cause mortality (aHR, 6.91). Adults 65 years old and older had an almost threefold increased risk of dying, compared with their younger peers (aHR, 2.74).
Dr. Dixon and colleagues also observed an increased risk of death in men with cancer and COVID, compared with women (aHR, 1.23) and those with at least two comorbid conditions versus none (aHR, 2.12). In addition, the risk of dying was 9% higher among Indiana’s rural population than urban dwellers.
Compared with other cancer types, individuals with lung cancer and other digestive cancers had the highest risk of death after SARS-CoV-2 infection (aHR, 1.45 and 1.80, respectively).
“Our findings highlight the increased risk of death for adult cancer patients who test positive for COVID and underscore the importance to cancer patients – including those in remission – of vaccinations, boosters, and regular COVID testing,” Dr. Dixon commented.
“Our results should encourage individuals diagnosed with cancer not only to take preventive action, but also to expeditiously seek out treatments available in the marketplace should they test positive for COVID,” he added.
Support for the study was provided by Indiana University Simon Cancer Center and the Centers for Disease Control and Prevention. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JMIR CANCER
This brain surgery was BYOS: Bring your own saxophone
Tumor vs. saxophone: The surgical grudge match
Brain surgery is a notoriously difficult task. There’s a reason we say, “Well, at least it’s not brain surgery” when we’re trying to convince someone that a task isn’t that tough. Make one wrong incision, cut the wrong neuron, and it’s goodbye higher cognitive function. And most people appreciate thinking. Crazy, right?
One would imagine that the act of brain surgery would become even more difficult when the patient brings his saxophone and plays it randomly throughout the operation. It’s a hospital, after all, not a jazz club. Patients don’t get to play musical instruments during other surgeries. Why should brain surgery patients get special treatment?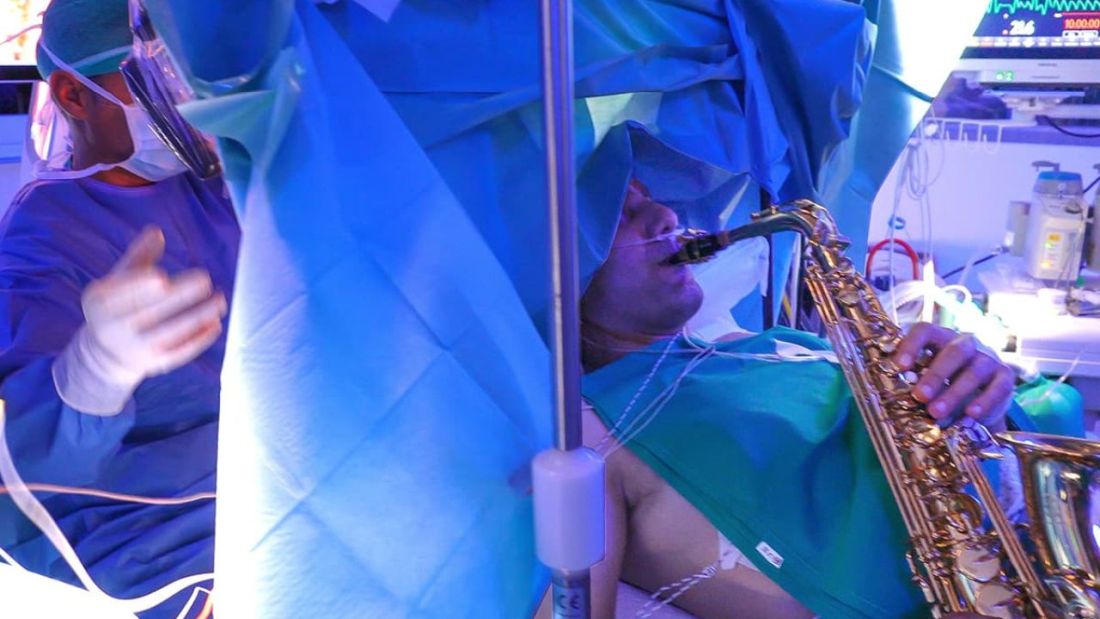
As it turns out, the musical performance was actually quite helpful. A man in Italy had a brain tumor in a particularly complex area, and he’s left-handed, which apparently makes the brain’s neural pathways much more complicated. Plus, he insisted that he retain his musical ability after the surgery. So he and his medical team had a crazy thought: Why not play the saxophone throughout the surgery? After all, according to head surgeon Christian Brogna, MD, playing an instrument means you understand music, which tests many higher cognitive functions such as coordination, mathematics, and memory.
And so, at various points throughout the 9-hour surgery, the patient played his saxophone for his doctors. Doing so allowed the surgeons to map the patient’s brain in a more complete and personalized fashion. With that extra knowledge, they were able to successfully remove the tumor while maintaining the patient’s musical ability, and the patient was discharged on Oct. 13, just 3 days after his operation.
While we’re happy the patient recovered, we do have to question his choice of music. During the surgery, he played the theme to the 1970 movie “Love Story” and the Italian national anthem. Perfectly fine pieces, no doubt, but the saxophone solo in “Jungleland” exists. And we could listen to that for 9 hours straight. In fact, we do that every Friday in the LOTME office.
Basketball has the Big Dance. Mosquitoes get the Big Sniff
In this week’s installment of our seemingly never-ending series, “Mosquitoes and the scientists who love them,” we visit The Rockefeller University in New York, where the olfactory capabilities of Aedes Aegypti – the primary vector species for Zika, dengue, yellow fever, and chikungunya – became the subject of a round robin–style tournament.
First things first, though. If you’re going to test mosquito noses, you have to give them something to smell. The researchers enrolled eight humans who were willing to wear nylon stockings on their forearms for 6 hours a day for multiple days. “Over the next few years, the researchers tested the nylons against each other in all possible pairings,” Leslie B. Vosshall, PhD, and associates said in a statement from the university. In other words, mosquito March Madness.
Nylons from different participants were hooked up in pairs to an olfactometer assay consisting of a plexiglass chamber divided into two tubes, each ending in a box that held a stocking. The mosquitoes were placed in the main chamber and observed as they flew down the tubes toward one stocking or the other.
Eventually, the “winner” of the “tournament” was Subject 33. And no, we don’t know why there was a Subject 33 since the study involved only eight participants. We do know that the nylons worn by Subject 33 were “four times more attractive to the mosquitoes than the next most-attractive study participant, and an astonishing 100 times more appealing than the least attractive, Subject 19,” according to the written statement.
Chemical analysis identified 50 molecular compounds that were elevated in the sebum of the high-attracting participants, and eventually the investigators discovered that mosquito magnets produced carboxylic acids at much higher levels than the less-attractive volunteers.
We could go on about the research team genetically engineering mosquitoes without odor receptors, but we have to save something for later. Tune in again next week for another exciting episode of “Mosquitoes and the scientists who love them.”
Are women better with words?
Men vs. Women is probably the oldest argument in the book, but there may now be movement. Researchers have been able not only to shift the advantage toward women, but also to use that knowledge to medical advantage.
When it comes to the matter of words and remembering them, women apparently have men beat. The margin is small, said lead author Marco Hirnstein, PhD, of the University of Bergen, Norway, but, after performing a meta-analysis of 168 published studies and PhD theses involving more than 350,000 participants, it’s pretty clear. The research supports women’s advantage over men in recall, verbal fluency (categorical and phonemic), and recognition.
So how is this information useful from a medical standpoint?
Dr. Hirnstein and colleagues suggested that this information can help in interpreting diagnostic assessment results. The example given was dementia diagnosis. Since women are underdiagnosed because their baseline exceeds average while men are overdiagnosed, taking gender and performance into account could clear up or catch cases that might otherwise slip through the cracks.
Now, let’s just put this part of the debate to rest and take this not only as a win for women but for science as well.
Tumor vs. saxophone: The surgical grudge match
Brain surgery is a notoriously difficult task. There’s a reason we say, “Well, at least it’s not brain surgery” when we’re trying to convince someone that a task isn’t that tough. Make one wrong incision, cut the wrong neuron, and it’s goodbye higher cognitive function. And most people appreciate thinking. Crazy, right?
One would imagine that the act of brain surgery would become even more difficult when the patient brings his saxophone and plays it randomly throughout the operation. It’s a hospital, after all, not a jazz club. Patients don’t get to play musical instruments during other surgeries. Why should brain surgery patients get special treatment?
As it turns out, the musical performance was actually quite helpful. A man in Italy had a brain tumor in a particularly complex area, and he’s left-handed, which apparently makes the brain’s neural pathways much more complicated. Plus, he insisted that he retain his musical ability after the surgery. So he and his medical team had a crazy thought: Why not play the saxophone throughout the surgery? After all, according to head surgeon Christian Brogna, MD, playing an instrument means you understand music, which tests many higher cognitive functions such as coordination, mathematics, and memory.
And so, at various points throughout the 9-hour surgery, the patient played his saxophone for his doctors. Doing so allowed the surgeons to map the patient’s brain in a more complete and personalized fashion. With that extra knowledge, they were able to successfully remove the tumor while maintaining the patient’s musical ability, and the patient was discharged on Oct. 13, just 3 days after his operation.
While we’re happy the patient recovered, we do have to question his choice of music. During the surgery, he played the theme to the 1970 movie “Love Story” and the Italian national anthem. Perfectly fine pieces, no doubt, but the saxophone solo in “Jungleland” exists. And we could listen to that for 9 hours straight. In fact, we do that every Friday in the LOTME office.
Basketball has the Big Dance. Mosquitoes get the Big Sniff
In this week’s installment of our seemingly never-ending series, “Mosquitoes and the scientists who love them,” we visit The Rockefeller University in New York, where the olfactory capabilities of Aedes Aegypti – the primary vector species for Zika, dengue, yellow fever, and chikungunya – became the subject of a round robin–style tournament.
First things first, though. If you’re going to test mosquito noses, you have to give them something to smell. The researchers enrolled eight humans who were willing to wear nylon stockings on their forearms for 6 hours a day for multiple days. “Over the next few years, the researchers tested the nylons against each other in all possible pairings,” Leslie B. Vosshall, PhD, and associates said in a statement from the university. In other words, mosquito March Madness.
Nylons from different participants were hooked up in pairs to an olfactometer assay consisting of a plexiglass chamber divided into two tubes, each ending in a box that held a stocking. The mosquitoes were placed in the main chamber and observed as they flew down the tubes toward one stocking or the other.
Eventually, the “winner” of the “tournament” was Subject 33. And no, we don’t know why there was a Subject 33 since the study involved only eight participants. We do know that the nylons worn by Subject 33 were “four times more attractive to the mosquitoes than the next most-attractive study participant, and an astonishing 100 times more appealing than the least attractive, Subject 19,” according to the written statement.
Chemical analysis identified 50 molecular compounds that were elevated in the sebum of the high-attracting participants, and eventually the investigators discovered that mosquito magnets produced carboxylic acids at much higher levels than the less-attractive volunteers.
We could go on about the research team genetically engineering mosquitoes without odor receptors, but we have to save something for later. Tune in again next week for another exciting episode of “Mosquitoes and the scientists who love them.”
Are women better with words?
Men vs. Women is probably the oldest argument in the book, but there may now be movement. Researchers have been able not only to shift the advantage toward women, but also to use that knowledge to medical advantage.
When it comes to the matter of words and remembering them, women apparently have men beat. The margin is small, said lead author Marco Hirnstein, PhD, of the University of Bergen, Norway, but, after performing a meta-analysis of 168 published studies and PhD theses involving more than 350,000 participants, it’s pretty clear. The research supports women’s advantage over men in recall, verbal fluency (categorical and phonemic), and recognition.
So how is this information useful from a medical standpoint?
Dr. Hirnstein and colleagues suggested that this information can help in interpreting diagnostic assessment results. The example given was dementia diagnosis. Since women are underdiagnosed because their baseline exceeds average while men are overdiagnosed, taking gender and performance into account could clear up or catch cases that might otherwise slip through the cracks.
Now, let’s just put this part of the debate to rest and take this not only as a win for women but for science as well.
Tumor vs. saxophone: The surgical grudge match
Brain surgery is a notoriously difficult task. There’s a reason we say, “Well, at least it’s not brain surgery” when we’re trying to convince someone that a task isn’t that tough. Make one wrong incision, cut the wrong neuron, and it’s goodbye higher cognitive function. And most people appreciate thinking. Crazy, right?
One would imagine that the act of brain surgery would become even more difficult when the patient brings his saxophone and plays it randomly throughout the operation. It’s a hospital, after all, not a jazz club. Patients don’t get to play musical instruments during other surgeries. Why should brain surgery patients get special treatment?
As it turns out, the musical performance was actually quite helpful. A man in Italy had a brain tumor in a particularly complex area, and he’s left-handed, which apparently makes the brain’s neural pathways much more complicated. Plus, he insisted that he retain his musical ability after the surgery. So he and his medical team had a crazy thought: Why not play the saxophone throughout the surgery? After all, according to head surgeon Christian Brogna, MD, playing an instrument means you understand music, which tests many higher cognitive functions such as coordination, mathematics, and memory.
And so, at various points throughout the 9-hour surgery, the patient played his saxophone for his doctors. Doing so allowed the surgeons to map the patient’s brain in a more complete and personalized fashion. With that extra knowledge, they were able to successfully remove the tumor while maintaining the patient’s musical ability, and the patient was discharged on Oct. 13, just 3 days after his operation.
While we’re happy the patient recovered, we do have to question his choice of music. During the surgery, he played the theme to the 1970 movie “Love Story” and the Italian national anthem. Perfectly fine pieces, no doubt, but the saxophone solo in “Jungleland” exists. And we could listen to that for 9 hours straight. In fact, we do that every Friday in the LOTME office.
Basketball has the Big Dance. Mosquitoes get the Big Sniff
In this week’s installment of our seemingly never-ending series, “Mosquitoes and the scientists who love them,” we visit The Rockefeller University in New York, where the olfactory capabilities of Aedes Aegypti – the primary vector species for Zika, dengue, yellow fever, and chikungunya – became the subject of a round robin–style tournament.
First things first, though. If you’re going to test mosquito noses, you have to give them something to smell. The researchers enrolled eight humans who were willing to wear nylon stockings on their forearms for 6 hours a day for multiple days. “Over the next few years, the researchers tested the nylons against each other in all possible pairings,” Leslie B. Vosshall, PhD, and associates said in a statement from the university. In other words, mosquito March Madness.
Nylons from different participants were hooked up in pairs to an olfactometer assay consisting of a plexiglass chamber divided into two tubes, each ending in a box that held a stocking. The mosquitoes were placed in the main chamber and observed as they flew down the tubes toward one stocking or the other.
Eventually, the “winner” of the “tournament” was Subject 33. And no, we don’t know why there was a Subject 33 since the study involved only eight participants. We do know that the nylons worn by Subject 33 were “four times more attractive to the mosquitoes than the next most-attractive study participant, and an astonishing 100 times more appealing than the least attractive, Subject 19,” according to the written statement.
Chemical analysis identified 50 molecular compounds that were elevated in the sebum of the high-attracting participants, and eventually the investigators discovered that mosquito magnets produced carboxylic acids at much higher levels than the less-attractive volunteers.
We could go on about the research team genetically engineering mosquitoes without odor receptors, but we have to save something for later. Tune in again next week for another exciting episode of “Mosquitoes and the scientists who love them.”
Are women better with words?
Men vs. Women is probably the oldest argument in the book, but there may now be movement. Researchers have been able not only to shift the advantage toward women, but also to use that knowledge to medical advantage.
When it comes to the matter of words and remembering them, women apparently have men beat. The margin is small, said lead author Marco Hirnstein, PhD, of the University of Bergen, Norway, but, after performing a meta-analysis of 168 published studies and PhD theses involving more than 350,000 participants, it’s pretty clear. The research supports women’s advantage over men in recall, verbal fluency (categorical and phonemic), and recognition.
So how is this information useful from a medical standpoint?
Dr. Hirnstein and colleagues suggested that this information can help in interpreting diagnostic assessment results. The example given was dementia diagnosis. Since women are underdiagnosed because their baseline exceeds average while men are overdiagnosed, taking gender and performance into account could clear up or catch cases that might otherwise slip through the cracks.
Now, let’s just put this part of the debate to rest and take this not only as a win for women but for science as well.
FDA approved acalabrutinib tablet for MCL, CLL, SLL
These include adult patients with chronic lymphocytic leukemia, small lymphocytic lymphoma, and relapsed or refractory mantle cell lymphoma.
Approval of the tablet formulation of the selective Bruton tyrosine kinase inhibitor was based on the ELEVATE-PLUS trials, which showed bioequivalence with the capsule. The tablet had the same efficacy and safety profile with the same dosing strength and schedule, AstraZeneca said in a press release.
The benefit of the tablet formulation is that patients with acid reflux and other problems can take it with proton pump inhibitors, antacids, and H2-receptor antagonists, the company noted.
A version of this article first appeared on Medscape.com.
These include adult patients with chronic lymphocytic leukemia, small lymphocytic lymphoma, and relapsed or refractory mantle cell lymphoma.
Approval of the tablet formulation of the selective Bruton tyrosine kinase inhibitor was based on the ELEVATE-PLUS trials, which showed bioequivalence with the capsule. The tablet had the same efficacy and safety profile with the same dosing strength and schedule, AstraZeneca said in a press release.
The benefit of the tablet formulation is that patients with acid reflux and other problems can take it with proton pump inhibitors, antacids, and H2-receptor antagonists, the company noted.
A version of this article first appeared on Medscape.com.
These include adult patients with chronic lymphocytic leukemia, small lymphocytic lymphoma, and relapsed or refractory mantle cell lymphoma.
Approval of the tablet formulation of the selective Bruton tyrosine kinase inhibitor was based on the ELEVATE-PLUS trials, which showed bioequivalence with the capsule. The tablet had the same efficacy and safety profile with the same dosing strength and schedule, AstraZeneca said in a press release.
The benefit of the tablet formulation is that patients with acid reflux and other problems can take it with proton pump inhibitors, antacids, and H2-receptor antagonists, the company noted.
A version of this article first appeared on Medscape.com.
Apixaban outmatches rivaroxaban in patients with AFib and valvular heart disease
Compared with rivaroxaban, apixaban cut risks nearly in half, suggesting that clinicians should consider these new data when choosing an anticoagulant, reported lead author Ghadeer K. Dawwas, PhD, of the University of Pennsylvania, Philadelphia, and colleagues.
In the new retrospective study involving almost 20,000 patients, Dr. Dawwas and her colleagues “emulated a target trial” using private insurance claims from Optum’s deidentified Clinformatics Data Mart Database. The cohort was narrowed from a screened population of 58,210 patients with concurrent AFib and VHD to 9,947 new apixaban users who could be closely matched with 9,947 new rivaroxaban users. Covariates included provider specialty, type of VHD, demographic characteristics, measures of health care use, baseline use of medications, and baseline comorbidities.
The primary effectiveness outcome was a composite of systemic embolism and ischemic stroke, while the primary safety outcome was a composite of intracranial or gastrointestinal bleeding.
“Although several ongoing trials aim to compare apixaban with warfarin in patients with AFib and VHD, none of these trials will directly compare apixaban and rivaroxaban,” the investigators wrote. Their report is in Annals of Internal Medicine.
Dr. Dawwas and colleagues previously showed that direct oral anticoagulants (DOACs) were safer and more effective than warfarin in the same patient population. Comparing apixaban and rivaroxaban – the two most common DOACs – was the next logical step, Dr. Dawwas said in an interview.
Study results
Compared with rivaroxaban, patients who received apixaban had a 43% reduced risk of stroke or embolism (hazard ratio [HR], 0.57; 95% confidence interval [CI], 0.40-0.80). Apixaban’s ability to protect against bleeding appeared even more pronounced, with a 49% reduced risk over rivaroxaban (HR, 0.51; 95% CI, 0.41-0.62).
Comparing the two agents on an absolute basis, apixaban reduced risk of embolism or stroke by 0.2% within the first 6 months of treatment initiation, and 1.1% within the first year of initiation. At the same time points, absolute risk reductions for bleeding were 1.2% and 1.9%, respectively.
The investigators noted that their results held consistent in an alternative analysis that considered separate types of VHD.
“Based on the results from our analysis, we showed that apixaban is effective and safe in patients with atrial fibrillation and valvular heart diseases,” Dr. Dawwas said.
Head-to-head trial needed to change practice
Christopher M. Bianco, DO, associate professor of medicine at West Virginia University Heart and Vascular Institute, Morgantown, said the findings “add to the growing body of literature,” but “a head-to-head trial would be necessary to make a definitive change to clinical practice.”
Dr. Bianco, who recently conducted a retrospective analysis of apixaban and rivaroxaban that found no difference in safety and efficacy among a different patient population, said these kinds of studies are helpful in generating hypotheses, but they can’t account for all relevant clinical factors.
“There are just so many things that go into the decision-making process of [prescribing] apixaban and rivaroxaban,” he said. “Even though [Dr. Dawwas and colleagues] used propensity matching, you’re never going to be able to sort that out with a retrospective analysis.”
Specifically, Dr. Bianco noted that the findings did not include dose data. This is a key gap, he said, considering how often real-world datasets have shown that providers underdose DOACs for a number of unaccountable reasons, and how frequently patients exhibit poor adherence.
The study also lacked detail concerning the degree of renal dysfunction, which can determine drug eligibility, Dr. Bianco said. Furthermore, attempts to stratify patients based on thrombosis and bleeding risk were likely “insufficient,” he added.
Dr. Bianco also cautioned that the investigators defined valvular heart disease as any valve-related disease of any severity. In contrast, previous studies have generally restricted valvular heart disease to patients with mitral stenosis or prosthetic valves.
“This is definitely not the traditional definition of valvular heart disease, so the title is a little bit misleading in that sense, although they certainly do disclose that in the methods,” Dr. Bianco said.
On a more positive note, he highlighted the size of the patient population, and the real-world data, which included many patients who would be excluded from clinical trials.
More broadly, the study helps drive research forward, Dr. Bianco concluded; namely, by attracting financial support for a more powerful head-to-head trial that drug makers are unlikely to fund due to inherent market risk.
This study was supported by the National Institutes of Health. The investigators disclosed additional relationships with Takeda, Spark, Sanofi, and others. Dr. Bianco disclosed no conflicts of interest.
Compared with rivaroxaban, apixaban cut risks nearly in half, suggesting that clinicians should consider these new data when choosing an anticoagulant, reported lead author Ghadeer K. Dawwas, PhD, of the University of Pennsylvania, Philadelphia, and colleagues.
In the new retrospective study involving almost 20,000 patients, Dr. Dawwas and her colleagues “emulated a target trial” using private insurance claims from Optum’s deidentified Clinformatics Data Mart Database. The cohort was narrowed from a screened population of 58,210 patients with concurrent AFib and VHD to 9,947 new apixaban users who could be closely matched with 9,947 new rivaroxaban users. Covariates included provider specialty, type of VHD, demographic characteristics, measures of health care use, baseline use of medications, and baseline comorbidities.
The primary effectiveness outcome was a composite of systemic embolism and ischemic stroke, while the primary safety outcome was a composite of intracranial or gastrointestinal bleeding.
“Although several ongoing trials aim to compare apixaban with warfarin in patients with AFib and VHD, none of these trials will directly compare apixaban and rivaroxaban,” the investigators wrote. Their report is in Annals of Internal Medicine.
Dr. Dawwas and colleagues previously showed that direct oral anticoagulants (DOACs) were safer and more effective than warfarin in the same patient population. Comparing apixaban and rivaroxaban – the two most common DOACs – was the next logical step, Dr. Dawwas said in an interview.
Study results
Compared with rivaroxaban, patients who received apixaban had a 43% reduced risk of stroke or embolism (hazard ratio [HR], 0.57; 95% confidence interval [CI], 0.40-0.80). Apixaban’s ability to protect against bleeding appeared even more pronounced, with a 49% reduced risk over rivaroxaban (HR, 0.51; 95% CI, 0.41-0.62).
Comparing the two agents on an absolute basis, apixaban reduced risk of embolism or stroke by 0.2% within the first 6 months of treatment initiation, and 1.1% within the first year of initiation. At the same time points, absolute risk reductions for bleeding were 1.2% and 1.9%, respectively.
The investigators noted that their results held consistent in an alternative analysis that considered separate types of VHD.
“Based on the results from our analysis, we showed that apixaban is effective and safe in patients with atrial fibrillation and valvular heart diseases,” Dr. Dawwas said.
Head-to-head trial needed to change practice
Christopher M. Bianco, DO, associate professor of medicine at West Virginia University Heart and Vascular Institute, Morgantown, said the findings “add to the growing body of literature,” but “a head-to-head trial would be necessary to make a definitive change to clinical practice.”
Dr. Bianco, who recently conducted a retrospective analysis of apixaban and rivaroxaban that found no difference in safety and efficacy among a different patient population, said these kinds of studies are helpful in generating hypotheses, but they can’t account for all relevant clinical factors.
“There are just so many things that go into the decision-making process of [prescribing] apixaban and rivaroxaban,” he said. “Even though [Dr. Dawwas and colleagues] used propensity matching, you’re never going to be able to sort that out with a retrospective analysis.”
Specifically, Dr. Bianco noted that the findings did not include dose data. This is a key gap, he said, considering how often real-world datasets have shown that providers underdose DOACs for a number of unaccountable reasons, and how frequently patients exhibit poor adherence.
The study also lacked detail concerning the degree of renal dysfunction, which can determine drug eligibility, Dr. Bianco said. Furthermore, attempts to stratify patients based on thrombosis and bleeding risk were likely “insufficient,” he added.
Dr. Bianco also cautioned that the investigators defined valvular heart disease as any valve-related disease of any severity. In contrast, previous studies have generally restricted valvular heart disease to patients with mitral stenosis or prosthetic valves.
“This is definitely not the traditional definition of valvular heart disease, so the title is a little bit misleading in that sense, although they certainly do disclose that in the methods,” Dr. Bianco said.
On a more positive note, he highlighted the size of the patient population, and the real-world data, which included many patients who would be excluded from clinical trials.
More broadly, the study helps drive research forward, Dr. Bianco concluded; namely, by attracting financial support for a more powerful head-to-head trial that drug makers are unlikely to fund due to inherent market risk.
This study was supported by the National Institutes of Health. The investigators disclosed additional relationships with Takeda, Spark, Sanofi, and others. Dr. Bianco disclosed no conflicts of interest.
Compared with rivaroxaban, apixaban cut risks nearly in half, suggesting that clinicians should consider these new data when choosing an anticoagulant, reported lead author Ghadeer K. Dawwas, PhD, of the University of Pennsylvania, Philadelphia, and colleagues.
In the new retrospective study involving almost 20,000 patients, Dr. Dawwas and her colleagues “emulated a target trial” using private insurance claims from Optum’s deidentified Clinformatics Data Mart Database. The cohort was narrowed from a screened population of 58,210 patients with concurrent AFib and VHD to 9,947 new apixaban users who could be closely matched with 9,947 new rivaroxaban users. Covariates included provider specialty, type of VHD, demographic characteristics, measures of health care use, baseline use of medications, and baseline comorbidities.
The primary effectiveness outcome was a composite of systemic embolism and ischemic stroke, while the primary safety outcome was a composite of intracranial or gastrointestinal bleeding.
“Although several ongoing trials aim to compare apixaban with warfarin in patients with AFib and VHD, none of these trials will directly compare apixaban and rivaroxaban,” the investigators wrote. Their report is in Annals of Internal Medicine.
Dr. Dawwas and colleagues previously showed that direct oral anticoagulants (DOACs) were safer and more effective than warfarin in the same patient population. Comparing apixaban and rivaroxaban – the two most common DOACs – was the next logical step, Dr. Dawwas said in an interview.
Study results
Compared with rivaroxaban, patients who received apixaban had a 43% reduced risk of stroke or embolism (hazard ratio [HR], 0.57; 95% confidence interval [CI], 0.40-0.80). Apixaban’s ability to protect against bleeding appeared even more pronounced, with a 49% reduced risk over rivaroxaban (HR, 0.51; 95% CI, 0.41-0.62).
Comparing the two agents on an absolute basis, apixaban reduced risk of embolism or stroke by 0.2% within the first 6 months of treatment initiation, and 1.1% within the first year of initiation. At the same time points, absolute risk reductions for bleeding were 1.2% and 1.9%, respectively.
The investigators noted that their results held consistent in an alternative analysis that considered separate types of VHD.
“Based on the results from our analysis, we showed that apixaban is effective and safe in patients with atrial fibrillation and valvular heart diseases,” Dr. Dawwas said.
Head-to-head trial needed to change practice
Christopher M. Bianco, DO, associate professor of medicine at West Virginia University Heart and Vascular Institute, Morgantown, said the findings “add to the growing body of literature,” but “a head-to-head trial would be necessary to make a definitive change to clinical practice.”
Dr. Bianco, who recently conducted a retrospective analysis of apixaban and rivaroxaban that found no difference in safety and efficacy among a different patient population, said these kinds of studies are helpful in generating hypotheses, but they can’t account for all relevant clinical factors.
“There are just so many things that go into the decision-making process of [prescribing] apixaban and rivaroxaban,” he said. “Even though [Dr. Dawwas and colleagues] used propensity matching, you’re never going to be able to sort that out with a retrospective analysis.”
Specifically, Dr. Bianco noted that the findings did not include dose data. This is a key gap, he said, considering how often real-world datasets have shown that providers underdose DOACs for a number of unaccountable reasons, and how frequently patients exhibit poor adherence.
The study also lacked detail concerning the degree of renal dysfunction, which can determine drug eligibility, Dr. Bianco said. Furthermore, attempts to stratify patients based on thrombosis and bleeding risk were likely “insufficient,” he added.
Dr. Bianco also cautioned that the investigators defined valvular heart disease as any valve-related disease of any severity. In contrast, previous studies have generally restricted valvular heart disease to patients with mitral stenosis or prosthetic valves.
“This is definitely not the traditional definition of valvular heart disease, so the title is a little bit misleading in that sense, although they certainly do disclose that in the methods,” Dr. Bianco said.
On a more positive note, he highlighted the size of the patient population, and the real-world data, which included many patients who would be excluded from clinical trials.
More broadly, the study helps drive research forward, Dr. Bianco concluded; namely, by attracting financial support for a more powerful head-to-head trial that drug makers are unlikely to fund due to inherent market risk.
This study was supported by the National Institutes of Health. The investigators disclosed additional relationships with Takeda, Spark, Sanofi, and others. Dr. Bianco disclosed no conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
Are doctors savers or spenders?
In a poll that ran from August 30 to Sept. 21, conducted by Medscape, physicians were asked if they lived within their means. They were asked whether they pay their bills on time, save at least 20% of their monthly income toward retirement, pay down student loan debt, and contribute to their kids’ college savings or a rainy-day emergency fund.
Medscape polled 468 U.S. physicians and 159 living outside of the United States. Eighty-nine percent of U.S. respondents report living within their means, while only 11% said they don’t.
Medscape’s Physician Wealth & Debt Report 2022 similarly reported that of 13,000 physicians in more than 29 specialties, 94% said they live at or below their means.
For example, over half of physicians have a net worth above $1 million. In contrast, according to Credit Suisse’s Global Wealth Report, less than 7% of the general population has a seven-figure net worth.
So just how do physicians stack up financially?
Habits of physician super savers
Physicians who consider themselves savers likely have money habits that correlate. They buy things on sale, are DIYers for home projects and maintenance, and wait to buy luxury or large expenses when the timing is right, an item is on sale, or they’ve saved for it.
For example, when it comes to life’s luxuries like buying a new car or dining out, overall, physicians seem to be more frugal, as 43% of those who buy cars said they only buy a new car every 10 years; 30% said they buy a new vehicle every 6-7 years, and 22% said every 4-5 years.
When asked about weekly dine-out or delivery habits, 82% of those polled who said they dine out, or order takeout, do so a nominal 1-2 times per week. That’s on par with the Centers for Disease Control, which reports that 3 in 5 Americans eat out once weekly. Another 14% of polled physicians said they dine out 3-5 nights per week. Only 4% revealed they eat out or grab to-go food more than 5 nights a week.
When hiring for essential home maintenance, like house cleaning and pool or lawn service, almost a third of physicians we polled who require such maintenance employ a service for these tasks, and 23% hire out often while 21% hire out only sometimes. However, 14% say they rarely hire out for home maintenance, and 11% never do.
Since physicians are typically tight on time, they tend to favor outsourcing things like housecleaning, lawn service, landscaping, maintenance, and even cooking. So, the fact that a quarter of physicians polled rarely or never hire out for household help is somewhat surprising.
Most physicians also prioritize saving. When asked how important it is to save money consistently, 93% think it’s either extremely or very important, while only 6% think it’s somewhat important.
Barriers to wealth
When asked what barriers prevent them from saving at least 20% of their monthly income, physician respondents who said they live within their means and encountered barriers reported that family necessities (35%), student loan debt (19%), and mortgage sizes (18%) were the top reasons. The average doctor earns five times as much as the average American, according to the Global Wealth Report.
“What prevents me from saving is holding too much debt, responsibilities at home, bills, being unprepared for what is coming, and making excuses to spend even when it’s not necessary,” says Sean Ormond, MD, a dual board-certified physician in Anesthesiology and Pain Management in Phoenix.
When physician respondents who said they didn’t live within their means were asked about the barriers preventing them from saving at least 20% of their monthly income, they cited the cost of family necessities (49%), the size of their mortgage (47%), credit card debt (30%), student loan debt (21%), other loans (15%), and car lease/loan (13%).
“My most significant financial splurge is vacation, since I always choose the best, and the best comes at an extra cost,” says Dr. Ormond.
What’s your financial grade?
Finally, physicians were asked who they considered better at saving money, themselves or their spouse/domestic partner. Forty-four percent think they are the better saver, whereas 41% said that both they and their partner were equally good at saving. Thirteen percent credited their partner with better saving habits, and 2% said neither themselves nor their partner were good at saving money.
More than half (63%) of physicians polled pay off their credit card balance monthly, but 18% carry a $1,000-$5,000 balance, 10% have $5,000-$10,000 in credit card debt, and 6% hold more than $10,000 of credit card debt.
“I would grade myself with a B, because however much I love having the best, I still have a budget, and I always ensure that I follow it to the dot,” says Dr. Ormond.
A version of this article first appeared on Medscape.com.
In a poll that ran from August 30 to Sept. 21, conducted by Medscape, physicians were asked if they lived within their means. They were asked whether they pay their bills on time, save at least 20% of their monthly income toward retirement, pay down student loan debt, and contribute to their kids’ college savings or a rainy-day emergency fund.
Medscape polled 468 U.S. physicians and 159 living outside of the United States. Eighty-nine percent of U.S. respondents report living within their means, while only 11% said they don’t.
Medscape’s Physician Wealth & Debt Report 2022 similarly reported that of 13,000 physicians in more than 29 specialties, 94% said they live at or below their means.
For example, over half of physicians have a net worth above $1 million. In contrast, according to Credit Suisse’s Global Wealth Report, less than 7% of the general population has a seven-figure net worth.
So just how do physicians stack up financially?
Habits of physician super savers
Physicians who consider themselves savers likely have money habits that correlate. They buy things on sale, are DIYers for home projects and maintenance, and wait to buy luxury or large expenses when the timing is right, an item is on sale, or they’ve saved for it.
For example, when it comes to life’s luxuries like buying a new car or dining out, overall, physicians seem to be more frugal, as 43% of those who buy cars said they only buy a new car every 10 years; 30% said they buy a new vehicle every 6-7 years, and 22% said every 4-5 years.
When asked about weekly dine-out or delivery habits, 82% of those polled who said they dine out, or order takeout, do so a nominal 1-2 times per week. That’s on par with the Centers for Disease Control, which reports that 3 in 5 Americans eat out once weekly. Another 14% of polled physicians said they dine out 3-5 nights per week. Only 4% revealed they eat out or grab to-go food more than 5 nights a week.
When hiring for essential home maintenance, like house cleaning and pool or lawn service, almost a third of physicians we polled who require such maintenance employ a service for these tasks, and 23% hire out often while 21% hire out only sometimes. However, 14% say they rarely hire out for home maintenance, and 11% never do.
Since physicians are typically tight on time, they tend to favor outsourcing things like housecleaning, lawn service, landscaping, maintenance, and even cooking. So, the fact that a quarter of physicians polled rarely or never hire out for household help is somewhat surprising.
Most physicians also prioritize saving. When asked how important it is to save money consistently, 93% think it’s either extremely or very important, while only 6% think it’s somewhat important.
Barriers to wealth
When asked what barriers prevent them from saving at least 20% of their monthly income, physician respondents who said they live within their means and encountered barriers reported that family necessities (35%), student loan debt (19%), and mortgage sizes (18%) were the top reasons. The average doctor earns five times as much as the average American, according to the Global Wealth Report.
“What prevents me from saving is holding too much debt, responsibilities at home, bills, being unprepared for what is coming, and making excuses to spend even when it’s not necessary,” says Sean Ormond, MD, a dual board-certified physician in Anesthesiology and Pain Management in Phoenix.
When physician respondents who said they didn’t live within their means were asked about the barriers preventing them from saving at least 20% of their monthly income, they cited the cost of family necessities (49%), the size of their mortgage (47%), credit card debt (30%), student loan debt (21%), other loans (15%), and car lease/loan (13%).
“My most significant financial splurge is vacation, since I always choose the best, and the best comes at an extra cost,” says Dr. Ormond.
What’s your financial grade?
Finally, physicians were asked who they considered better at saving money, themselves or their spouse/domestic partner. Forty-four percent think they are the better saver, whereas 41% said that both they and their partner were equally good at saving. Thirteen percent credited their partner with better saving habits, and 2% said neither themselves nor their partner were good at saving money.
More than half (63%) of physicians polled pay off their credit card balance monthly, but 18% carry a $1,000-$5,000 balance, 10% have $5,000-$10,000 in credit card debt, and 6% hold more than $10,000 of credit card debt.
“I would grade myself with a B, because however much I love having the best, I still have a budget, and I always ensure that I follow it to the dot,” says Dr. Ormond.
A version of this article first appeared on Medscape.com.
In a poll that ran from August 30 to Sept. 21, conducted by Medscape, physicians were asked if they lived within their means. They were asked whether they pay their bills on time, save at least 20% of their monthly income toward retirement, pay down student loan debt, and contribute to their kids’ college savings or a rainy-day emergency fund.
Medscape polled 468 U.S. physicians and 159 living outside of the United States. Eighty-nine percent of U.S. respondents report living within their means, while only 11% said they don’t.
Medscape’s Physician Wealth & Debt Report 2022 similarly reported that of 13,000 physicians in more than 29 specialties, 94% said they live at or below their means.
For example, over half of physicians have a net worth above $1 million. In contrast, according to Credit Suisse’s Global Wealth Report, less than 7% of the general population has a seven-figure net worth.
So just how do physicians stack up financially?
Habits of physician super savers
Physicians who consider themselves savers likely have money habits that correlate. They buy things on sale, are DIYers for home projects and maintenance, and wait to buy luxury or large expenses when the timing is right, an item is on sale, or they’ve saved for it.
For example, when it comes to life’s luxuries like buying a new car or dining out, overall, physicians seem to be more frugal, as 43% of those who buy cars said they only buy a new car every 10 years; 30% said they buy a new vehicle every 6-7 years, and 22% said every 4-5 years.
When asked about weekly dine-out or delivery habits, 82% of those polled who said they dine out, or order takeout, do so a nominal 1-2 times per week. That’s on par with the Centers for Disease Control, which reports that 3 in 5 Americans eat out once weekly. Another 14% of polled physicians said they dine out 3-5 nights per week. Only 4% revealed they eat out or grab to-go food more than 5 nights a week.
When hiring for essential home maintenance, like house cleaning and pool or lawn service, almost a third of physicians we polled who require such maintenance employ a service for these tasks, and 23% hire out often while 21% hire out only sometimes. However, 14% say they rarely hire out for home maintenance, and 11% never do.
Since physicians are typically tight on time, they tend to favor outsourcing things like housecleaning, lawn service, landscaping, maintenance, and even cooking. So, the fact that a quarter of physicians polled rarely or never hire out for household help is somewhat surprising.
Most physicians also prioritize saving. When asked how important it is to save money consistently, 93% think it’s either extremely or very important, while only 6% think it’s somewhat important.
Barriers to wealth
When asked what barriers prevent them from saving at least 20% of their monthly income, physician respondents who said they live within their means and encountered barriers reported that family necessities (35%), student loan debt (19%), and mortgage sizes (18%) were the top reasons. The average doctor earns five times as much as the average American, according to the Global Wealth Report.
“What prevents me from saving is holding too much debt, responsibilities at home, bills, being unprepared for what is coming, and making excuses to spend even when it’s not necessary,” says Sean Ormond, MD, a dual board-certified physician in Anesthesiology and Pain Management in Phoenix.
When physician respondents who said they didn’t live within their means were asked about the barriers preventing them from saving at least 20% of their monthly income, they cited the cost of family necessities (49%), the size of their mortgage (47%), credit card debt (30%), student loan debt (21%), other loans (15%), and car lease/loan (13%).
“My most significant financial splurge is vacation, since I always choose the best, and the best comes at an extra cost,” says Dr. Ormond.
What’s your financial grade?
Finally, physicians were asked who they considered better at saving money, themselves or their spouse/domestic partner. Forty-four percent think they are the better saver, whereas 41% said that both they and their partner were equally good at saving. Thirteen percent credited their partner with better saving habits, and 2% said neither themselves nor their partner were good at saving money.
More than half (63%) of physicians polled pay off their credit card balance monthly, but 18% carry a $1,000-$5,000 balance, 10% have $5,000-$10,000 in credit card debt, and 6% hold more than $10,000 of credit card debt.
“I would grade myself with a B, because however much I love having the best, I still have a budget, and I always ensure that I follow it to the dot,” says Dr. Ormond.
A version of this article first appeared on Medscape.com.





