User login
Official news magazine of the Society of Hospital Medicine
Copyright by Society of Hospital Medicine or related companies. All rights reserved. ISSN 1553-085X
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'pane-pub-article-hospitalist')]


Hospitalist movers and shakers – September 2019
Mark Williams, MD, MHM, FACP, recently was appointed chief quality and transformation officer for the University of Kentucky’s UK HealthCare (Lexington). Dr. Williams, a tenured professor in the division of hospital medicine at the UK College of Medicine, will serve as chair of UK HealthCare’s Executive Quality Committee. Dr. Williams will lead integration of quality improvement, safety, and quality reporting with data analytics.
Dr. Williams established the first hospitalist program at a public hospital (Grady Memorial Hospital) and academic hospitalist programs at Emory University, Northwestern University, and UK HealthCare. An inaugural member of SHM, he is a past president, was the founding editor-in-chief of the Journal of Hospital Medicine and led SHM’s Project BOOST.
Also at UK HealthCare, Romil Chadha, MD, MPH, SFHM, FACP, has been named interim chief of the division of hospital medicine and medical director of Physician Information Technology Services. Previously, he was associate chief of the division of hospital medicine, and he also serves as medical director of telemetry.
Dr. Chadha is the founder of the Kentucky chapter of SHM, where he is the immediate past president. He is also the codirector of the Heartland Hospital Medicine Conference.
Amit Vashist, MD, MBA, CPE, FHM, FACP, FAPA, has been named chief clinical officer at Ballad Health, a 21-hospital health system in Northeast Tennessee, Southwest Virginia, Northwest North Carolina, and Southeast Kentucky.
In his new role, he will focus on clinical quality, value-based initiatives to improve quality while reducing cost of care, performance improvement, oversight of the clinical delivery of care and will be the liaison to the Ballad Health Clinical Council. Dr. Vashist is a member of The Hospitalist’s editorial advisory board.
Nagendra Gupta, MD, FACP, CPE, has been appointed to the American Board of Internal Medicine’s Internal Medicine Specialty Board. ABIM Specialty Boards are responsible for the broad definition of the discipline across Certification and Maintenance of Certification (MOC). Specialty Board members work with physicians and medical societies to develop Certification and MOC credentials to recognize physicians for their specialized knowledge and commitment to staying current in their field.
Dr. Gupta is a full-time practicing hospitalist with Apogee Physicians and currently serves as the director of the hospitalist program at Texas Health Arlington (Tex.) Memorial Hospital. He also serves as vice president for SHM’s North Central Texas Chapter.
T. Steen Trawick Jr., MD, was named the CEO of Christus Shreveport-Bossier Health System in Shreveport, La., in August 2019.
Dr. Trawick has worked for Christus as a pediatric hospitalist since 2005 and most recently has served concurrently as associate chief medical officer for Sound Physicians. Through Sound Physicians, Dr. Trawick oversees the hospitalist and emergency medical programs for Christus and other hospitals – 14 in total – in Texas and Louisiana. He has worked in that role for the past 6 years.
Scott Shepherd, DO, FACP, has been selected chief medical officer of the health data enrichment and integration technology company Verinovum in Tulsa, Okla. Dr. Shepherd is the medical director for hospitalist medicine and a practicing hospitalist with St. John Health System in Tulsa, and also medical director of the Center for Health Systems Innovation at his alma mater, Oklahoma State University in Stillwater.
Amanda Logue, MD, has been elevated to chief medical officer at Lafayette (La.) General Hospital. Dr. Logue assumed her role in May 2019, which includes the title of senior vice president.
Dr. Logue has worked at Lafayette General since 2009. A hospitalist/internist, her duties at the facility have included department chair of medicine, physician champion for electronic medical record implementation, medical director of the hospitalist program, and most recently chief medical information officer.
Rina Bansal, MD, MBA, recently was appointed full-time president of Inova Alexandria (Va.) Hospital, taking the reins officially after serving as acting president since November 2018. Dr. Bansal has been at Inova since 2008, when she started as a hospitalist at Inova Fairfax (Va.).
Dr. Bansal created and led Inova’s Clinical Nurse Services Hospitalist program through its department of neurosciences and has done stints as Inova Fairfax’s associate chief medical officer, medical director of Inova Telemedicine, and chief medical officer at Inova Alexandria.
James Napoli, MD, has been named chief medical officer for Blue Cross and Blue Shield of Arizona (BCBSAZ). He has manned the CMO position in an interim role since March, taking those duties on top of his role as BCBSAZ’s enterprise medical director for health care ventures and innovation.
Dr. Napoli came to BCBSAZ in 2013 after more than a decade at Abrazo Arrowhead Campus (Glendale, Ariz.) At Abrazo, he was director of hospitalist services and vice-chief of staff, on top of his efforts as a practicing hospital medicine clinician.
Dr. Napoli was previously medical director at OptumHealth, working specifically in the medical management and quality improvement areas for the health management solutions organization’s Medicare Advantage clients.
Mercy Hospital Fort Smith (Ark.) has partnered with the Ob Hospitalist Group (Greenville, S.C.) to launch an obstetric hospitalist program. OB hospitalists deliver babies when a patient’s physician cannot be present, provide emergency care, and provide support to high-risk pregnancy patients, among other duties within the hospital.
The partnership has allowed Mercy Fort Smith to create a dedicated, four-room obstetric emergency department in its Mercy Childbirth Center. Eight OB hospitalists have been hired and will provide care 24 hours a day, 7 days a week.
Mark Williams, MD, MHM, FACP, recently was appointed chief quality and transformation officer for the University of Kentucky’s UK HealthCare (Lexington). Dr. Williams, a tenured professor in the division of hospital medicine at the UK College of Medicine, will serve as chair of UK HealthCare’s Executive Quality Committee. Dr. Williams will lead integration of quality improvement, safety, and quality reporting with data analytics.
Dr. Williams established the first hospitalist program at a public hospital (Grady Memorial Hospital) and academic hospitalist programs at Emory University, Northwestern University, and UK HealthCare. An inaugural member of SHM, he is a past president, was the founding editor-in-chief of the Journal of Hospital Medicine and led SHM’s Project BOOST.
Also at UK HealthCare, Romil Chadha, MD, MPH, SFHM, FACP, has been named interim chief of the division of hospital medicine and medical director of Physician Information Technology Services. Previously, he was associate chief of the division of hospital medicine, and he also serves as medical director of telemetry.
Dr. Chadha is the founder of the Kentucky chapter of SHM, where he is the immediate past president. He is also the codirector of the Heartland Hospital Medicine Conference.
Amit Vashist, MD, MBA, CPE, FHM, FACP, FAPA, has been named chief clinical officer at Ballad Health, a 21-hospital health system in Northeast Tennessee, Southwest Virginia, Northwest North Carolina, and Southeast Kentucky.
In his new role, he will focus on clinical quality, value-based initiatives to improve quality while reducing cost of care, performance improvement, oversight of the clinical delivery of care and will be the liaison to the Ballad Health Clinical Council. Dr. Vashist is a member of The Hospitalist’s editorial advisory board.
Nagendra Gupta, MD, FACP, CPE, has been appointed to the American Board of Internal Medicine’s Internal Medicine Specialty Board. ABIM Specialty Boards are responsible for the broad definition of the discipline across Certification and Maintenance of Certification (MOC). Specialty Board members work with physicians and medical societies to develop Certification and MOC credentials to recognize physicians for their specialized knowledge and commitment to staying current in their field.
Dr. Gupta is a full-time practicing hospitalist with Apogee Physicians and currently serves as the director of the hospitalist program at Texas Health Arlington (Tex.) Memorial Hospital. He also serves as vice president for SHM’s North Central Texas Chapter.
T. Steen Trawick Jr., MD, was named the CEO of Christus Shreveport-Bossier Health System in Shreveport, La., in August 2019.
Dr. Trawick has worked for Christus as a pediatric hospitalist since 2005 and most recently has served concurrently as associate chief medical officer for Sound Physicians. Through Sound Physicians, Dr. Trawick oversees the hospitalist and emergency medical programs for Christus and other hospitals – 14 in total – in Texas and Louisiana. He has worked in that role for the past 6 years.
Scott Shepherd, DO, FACP, has been selected chief medical officer of the health data enrichment and integration technology company Verinovum in Tulsa, Okla. Dr. Shepherd is the medical director for hospitalist medicine and a practicing hospitalist with St. John Health System in Tulsa, and also medical director of the Center for Health Systems Innovation at his alma mater, Oklahoma State University in Stillwater.
Amanda Logue, MD, has been elevated to chief medical officer at Lafayette (La.) General Hospital. Dr. Logue assumed her role in May 2019, which includes the title of senior vice president.
Dr. Logue has worked at Lafayette General since 2009. A hospitalist/internist, her duties at the facility have included department chair of medicine, physician champion for electronic medical record implementation, medical director of the hospitalist program, and most recently chief medical information officer.
Rina Bansal, MD, MBA, recently was appointed full-time president of Inova Alexandria (Va.) Hospital, taking the reins officially after serving as acting president since November 2018. Dr. Bansal has been at Inova since 2008, when she started as a hospitalist at Inova Fairfax (Va.).
Dr. Bansal created and led Inova’s Clinical Nurse Services Hospitalist program through its department of neurosciences and has done stints as Inova Fairfax’s associate chief medical officer, medical director of Inova Telemedicine, and chief medical officer at Inova Alexandria.
James Napoli, MD, has been named chief medical officer for Blue Cross and Blue Shield of Arizona (BCBSAZ). He has manned the CMO position in an interim role since March, taking those duties on top of his role as BCBSAZ’s enterprise medical director for health care ventures and innovation.
Dr. Napoli came to BCBSAZ in 2013 after more than a decade at Abrazo Arrowhead Campus (Glendale, Ariz.) At Abrazo, he was director of hospitalist services and vice-chief of staff, on top of his efforts as a practicing hospital medicine clinician.
Dr. Napoli was previously medical director at OptumHealth, working specifically in the medical management and quality improvement areas for the health management solutions organization’s Medicare Advantage clients.
Mercy Hospital Fort Smith (Ark.) has partnered with the Ob Hospitalist Group (Greenville, S.C.) to launch an obstetric hospitalist program. OB hospitalists deliver babies when a patient’s physician cannot be present, provide emergency care, and provide support to high-risk pregnancy patients, among other duties within the hospital.
The partnership has allowed Mercy Fort Smith to create a dedicated, four-room obstetric emergency department in its Mercy Childbirth Center. Eight OB hospitalists have been hired and will provide care 24 hours a day, 7 days a week.
Mark Williams, MD, MHM, FACP, recently was appointed chief quality and transformation officer for the University of Kentucky’s UK HealthCare (Lexington). Dr. Williams, a tenured professor in the division of hospital medicine at the UK College of Medicine, will serve as chair of UK HealthCare’s Executive Quality Committee. Dr. Williams will lead integration of quality improvement, safety, and quality reporting with data analytics.
Dr. Williams established the first hospitalist program at a public hospital (Grady Memorial Hospital) and academic hospitalist programs at Emory University, Northwestern University, and UK HealthCare. An inaugural member of SHM, he is a past president, was the founding editor-in-chief of the Journal of Hospital Medicine and led SHM’s Project BOOST.
Also at UK HealthCare, Romil Chadha, MD, MPH, SFHM, FACP, has been named interim chief of the division of hospital medicine and medical director of Physician Information Technology Services. Previously, he was associate chief of the division of hospital medicine, and he also serves as medical director of telemetry.
Dr. Chadha is the founder of the Kentucky chapter of SHM, where he is the immediate past president. He is also the codirector of the Heartland Hospital Medicine Conference.
Amit Vashist, MD, MBA, CPE, FHM, FACP, FAPA, has been named chief clinical officer at Ballad Health, a 21-hospital health system in Northeast Tennessee, Southwest Virginia, Northwest North Carolina, and Southeast Kentucky.
In his new role, he will focus on clinical quality, value-based initiatives to improve quality while reducing cost of care, performance improvement, oversight of the clinical delivery of care and will be the liaison to the Ballad Health Clinical Council. Dr. Vashist is a member of The Hospitalist’s editorial advisory board.
Nagendra Gupta, MD, FACP, CPE, has been appointed to the American Board of Internal Medicine’s Internal Medicine Specialty Board. ABIM Specialty Boards are responsible for the broad definition of the discipline across Certification and Maintenance of Certification (MOC). Specialty Board members work with physicians and medical societies to develop Certification and MOC credentials to recognize physicians for their specialized knowledge and commitment to staying current in their field.
Dr. Gupta is a full-time practicing hospitalist with Apogee Physicians and currently serves as the director of the hospitalist program at Texas Health Arlington (Tex.) Memorial Hospital. He also serves as vice president for SHM’s North Central Texas Chapter.
T. Steen Trawick Jr., MD, was named the CEO of Christus Shreveport-Bossier Health System in Shreveport, La., in August 2019.
Dr. Trawick has worked for Christus as a pediatric hospitalist since 2005 and most recently has served concurrently as associate chief medical officer for Sound Physicians. Through Sound Physicians, Dr. Trawick oversees the hospitalist and emergency medical programs for Christus and other hospitals – 14 in total – in Texas and Louisiana. He has worked in that role for the past 6 years.
Scott Shepherd, DO, FACP, has been selected chief medical officer of the health data enrichment and integration technology company Verinovum in Tulsa, Okla. Dr. Shepherd is the medical director for hospitalist medicine and a practicing hospitalist with St. John Health System in Tulsa, and also medical director of the Center for Health Systems Innovation at his alma mater, Oklahoma State University in Stillwater.
Amanda Logue, MD, has been elevated to chief medical officer at Lafayette (La.) General Hospital. Dr. Logue assumed her role in May 2019, which includes the title of senior vice president.
Dr. Logue has worked at Lafayette General since 2009. A hospitalist/internist, her duties at the facility have included department chair of medicine, physician champion for electronic medical record implementation, medical director of the hospitalist program, and most recently chief medical information officer.
Rina Bansal, MD, MBA, recently was appointed full-time president of Inova Alexandria (Va.) Hospital, taking the reins officially after serving as acting president since November 2018. Dr. Bansal has been at Inova since 2008, when she started as a hospitalist at Inova Fairfax (Va.).
Dr. Bansal created and led Inova’s Clinical Nurse Services Hospitalist program through its department of neurosciences and has done stints as Inova Fairfax’s associate chief medical officer, medical director of Inova Telemedicine, and chief medical officer at Inova Alexandria.
James Napoli, MD, has been named chief medical officer for Blue Cross and Blue Shield of Arizona (BCBSAZ). He has manned the CMO position in an interim role since March, taking those duties on top of his role as BCBSAZ’s enterprise medical director for health care ventures and innovation.
Dr. Napoli came to BCBSAZ in 2013 after more than a decade at Abrazo Arrowhead Campus (Glendale, Ariz.) At Abrazo, he was director of hospitalist services and vice-chief of staff, on top of his efforts as a practicing hospital medicine clinician.
Dr. Napoli was previously medical director at OptumHealth, working specifically in the medical management and quality improvement areas for the health management solutions organization’s Medicare Advantage clients.
Mercy Hospital Fort Smith (Ark.) has partnered with the Ob Hospitalist Group (Greenville, S.C.) to launch an obstetric hospitalist program. OB hospitalists deliver babies when a patient’s physician cannot be present, provide emergency care, and provide support to high-risk pregnancy patients, among other duties within the hospital.
The partnership has allowed Mercy Fort Smith to create a dedicated, four-room obstetric emergency department in its Mercy Childbirth Center. Eight OB hospitalists have been hired and will provide care 24 hours a day, 7 days a week.
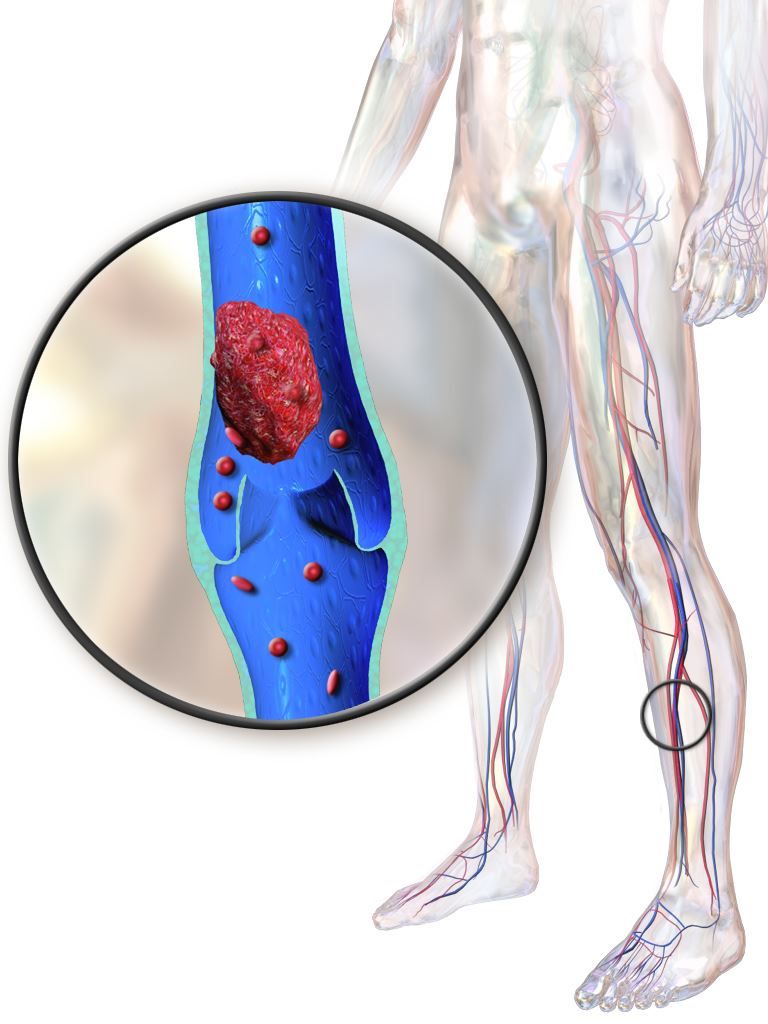
Drug abuse–linked infective endocarditis spiking in U.S.
Hospitalizations for infective endocarditis associated with drug abuse doubled in the United States from 2002 to 2016, in a trend investigators call “alarming,” and link to a concurrent rise in opioid abuse.
Patients tend to be younger, poorer white males, according to findings published online in the Journal of the American Heart Association.
For their research, Amer N. Kadri, MD, of the Cleveland Clinic and colleagues looked at records for nearly a million hospitalizations for infective endocarditis (IE) in the National Inpatient Sample registry. All U.S. regions saw increases in drug abuse–linked cases of IE as a share of IE hospitalizations. Incidence of drug abuse–associated IC rose from 48 cases/100,000 population in 2002 to 79/100,000 in 2016. The Midwest saw the highest rate of change, with an annual percent increase of 4.9%.
While most IE hospitalizations in the study cohort were of white men (including 68% for drug-linked cases), the drug abuse–related cases were younger (median age, 38 vs. 70 years for nondrug-related IE), and more likely male (55.5% vs. 50%). About 45% of the drug-related cases were in people receiving Medicaid, and 42% were in the lowest quartile of median household income.
The drug abuse cases had fewer renal and cardiovascular comorbidities, compared with the nondrug cases, but were significantly more likely to present with HIV, hepatitis C, alcohol abuse, and liver disease. Inpatient mortality was lower among the drug-linked cases – 6% vs. 9% – but the drug cases saw significantly more cardiac or valve surgeries, longer hospital stays, and higher costs.
“Hospitalizations for IE have been increasing side by side with the opioid epidemic,” the investigators wrote in their analysis. “The opioid crisis has reached epidemic levels, and now drug overdoses have been the leading cause of injury-related death in the U.S. Heroin deaths had remained relatively low from 1999 until 2010 whereas it then increased threefold from 2010-2015.” The analysis showed a rise in drug abuse–associated IE “that corresponds to this general period.” The findings argue, the investigators said, for better treatment for opioid addiction after hospitalization and greater efforts to make drug rehabilitation available after discharge. The researchers described as a limitation of their study the use of billing codes that changed late in the study period, increasing detection of drug abuse cases after 2015. They reported no outside funding or conflicts of interest.
SOURCE: Kadri AN et al. J Am Heart Assoc. 2019 Sep 18.
Hospitalizations for infective endocarditis associated with drug abuse doubled in the United States from 2002 to 2016, in a trend investigators call “alarming,” and link to a concurrent rise in opioid abuse.
Patients tend to be younger, poorer white males, according to findings published online in the Journal of the American Heart Association.
For their research, Amer N. Kadri, MD, of the Cleveland Clinic and colleagues looked at records for nearly a million hospitalizations for infective endocarditis (IE) in the National Inpatient Sample registry. All U.S. regions saw increases in drug abuse–linked cases of IE as a share of IE hospitalizations. Incidence of drug abuse–associated IC rose from 48 cases/100,000 population in 2002 to 79/100,000 in 2016. The Midwest saw the highest rate of change, with an annual percent increase of 4.9%.
While most IE hospitalizations in the study cohort were of white men (including 68% for drug-linked cases), the drug abuse–related cases were younger (median age, 38 vs. 70 years for nondrug-related IE), and more likely male (55.5% vs. 50%). About 45% of the drug-related cases were in people receiving Medicaid, and 42% were in the lowest quartile of median household income.
The drug abuse cases had fewer renal and cardiovascular comorbidities, compared with the nondrug cases, but were significantly more likely to present with HIV, hepatitis C, alcohol abuse, and liver disease. Inpatient mortality was lower among the drug-linked cases – 6% vs. 9% – but the drug cases saw significantly more cardiac or valve surgeries, longer hospital stays, and higher costs.
“Hospitalizations for IE have been increasing side by side with the opioid epidemic,” the investigators wrote in their analysis. “The opioid crisis has reached epidemic levels, and now drug overdoses have been the leading cause of injury-related death in the U.S. Heroin deaths had remained relatively low from 1999 until 2010 whereas it then increased threefold from 2010-2015.” The analysis showed a rise in drug abuse–associated IE “that corresponds to this general period.” The findings argue, the investigators said, for better treatment for opioid addiction after hospitalization and greater efforts to make drug rehabilitation available after discharge. The researchers described as a limitation of their study the use of billing codes that changed late in the study period, increasing detection of drug abuse cases after 2015. They reported no outside funding or conflicts of interest.
SOURCE: Kadri AN et al. J Am Heart Assoc. 2019 Sep 18.
Hospitalizations for infective endocarditis associated with drug abuse doubled in the United States from 2002 to 2016, in a trend investigators call “alarming,” and link to a concurrent rise in opioid abuse.
Patients tend to be younger, poorer white males, according to findings published online in the Journal of the American Heart Association.
For their research, Amer N. Kadri, MD, of the Cleveland Clinic and colleagues looked at records for nearly a million hospitalizations for infective endocarditis (IE) in the National Inpatient Sample registry. All U.S. regions saw increases in drug abuse–linked cases of IE as a share of IE hospitalizations. Incidence of drug abuse–associated IC rose from 48 cases/100,000 population in 2002 to 79/100,000 in 2016. The Midwest saw the highest rate of change, with an annual percent increase of 4.9%.
While most IE hospitalizations in the study cohort were of white men (including 68% for drug-linked cases), the drug abuse–related cases were younger (median age, 38 vs. 70 years for nondrug-related IE), and more likely male (55.5% vs. 50%). About 45% of the drug-related cases were in people receiving Medicaid, and 42% were in the lowest quartile of median household income.
The drug abuse cases had fewer renal and cardiovascular comorbidities, compared with the nondrug cases, but were significantly more likely to present with HIV, hepatitis C, alcohol abuse, and liver disease. Inpatient mortality was lower among the drug-linked cases – 6% vs. 9% – but the drug cases saw significantly more cardiac or valve surgeries, longer hospital stays, and higher costs.
“Hospitalizations for IE have been increasing side by side with the opioid epidemic,” the investigators wrote in their analysis. “The opioid crisis has reached epidemic levels, and now drug overdoses have been the leading cause of injury-related death in the U.S. Heroin deaths had remained relatively low from 1999 until 2010 whereas it then increased threefold from 2010-2015.” The analysis showed a rise in drug abuse–associated IE “that corresponds to this general period.” The findings argue, the investigators said, for better treatment for opioid addiction after hospitalization and greater efforts to make drug rehabilitation available after discharge. The researchers described as a limitation of their study the use of billing codes that changed late in the study period, increasing detection of drug abuse cases after 2015. They reported no outside funding or conflicts of interest.
SOURCE: Kadri AN et al. J Am Heart Assoc. 2019 Sep 18.
FROM THE JOURNAL OF THE AMERICAN HEART ASSOCIATION
Key clinical point:
Major finding: Incidence of drug abuse–associated IC increased from 48 cases/100,000 in 2002 to 79/100,000 in 2016.
Study details: A retrospective cohort study identifying about a more than 950,000 cases of IC from the National Inpatient Sample registry.
Disclosures: None.
Source: Kadri AN et al. J Am Heart Assoc. 2019 Sep 18.
Observation versus inpatient status
A dilemma for hospitalists and patients
A federal effort to reduce health care expenditures has left many older Medicare recipients experiencing the sticker shock of “observation status.” Patients who are not sick enough to meet inpatient admission criteria, however, still require hospitalization, and may be placed under Medicare observation care.
Seniors can get frustrated, confused, and anxious as their status can be changed while they are in the hospital, and they may receive large medical bills after they are discharged. The Centers for Medicare & Medicaid Services’ “3-day rule” mandates that Medicare will not pay for skilled nursing facility care unless the patient is admitted as an “inpatient” for at least 3 days. Observation days do not count towards this 3-day hospital stay.
There has been an increase in outpatient services over the years since 2006. The 2018 State of Hospital Medicine Report (SoHM) highlights the percentage of discharges based on hospitalists’ billed Current Procedural Terminology codes. Codes 99217 (observation discharge) and 99238-99239 (inpatient discharge) were used to calculate the percentages. 80.7% of adult medicine hospitalist discharges were coded using inpatient discharge codes, while 19.3% of patients were discharged with observation discharge codes.
In the 2016 SoHM report, the ratio was 76.0% inpatient and 21.1% observation codes and in the 2014 report we saw 80.3% inpatient and 16.1% observation discharges (see table 1). But in both of those surveys, same-day admission/discharge codes were also separately reported, which did not occur in 2018. That makes year-over-year comparison of the data challenging.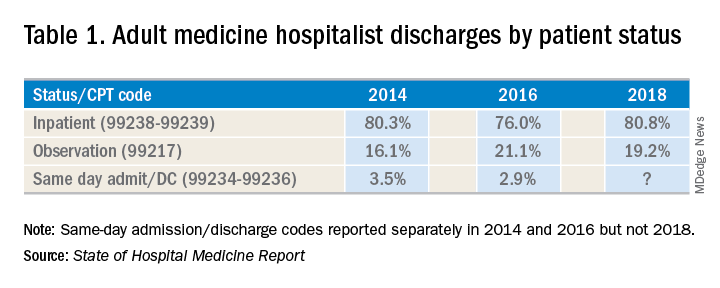
Interestingly, the 2017 CMS data on Evaluation and Management Codes by Specialty for the first time included separate data for hospitalists, based on hospitalists who credentialed with Medicare using the new C6 specialty code. Based on that data, when looking only at inpatient (99238-99239) and observation (99217) codes, 83% of the discharges were inpatient and 17% were observation.
Physicians feel the pressure of strained patient-physician relationships as a consequence of patients feeling the brunt of the financing gap related to observation status. Patients often feel they were not warned adequately about the financial ramifications of observation status. Even if Medicare beneficiaries have received the Medicare Outpatient Observation Notice, outlined by the Notice of Observation Treatment and Implication for Care Eligibility Act, they have no rights to appeal.
Currently Medicare beneficiaries admitted as inpatients only incur a Part A deductible; they are not liable for tests, procedures, and nursing care. On the other hand, in observation status all services are billed separately. For Medicare Part B services (which covers observation care) patients must pay 20% of services after the Part B deductible, which could result in a huge financial burden. Costs for skilled nursing facilities, when they are not covered by Medicare Part A, because of the 3-day rule, can easily go up to $20,000 or more. Medicare beneficiaries have no cap on costs for an observation stay. In some cases, hospitals have to apply a condition code 44 and retroactively change the stay to observation status.
I attended the 2019 Society of Hospital Medicine Annual Conference in Washington. Hospitalists from all parts of the country advocated on Capitol Hill against the “observation bill,” and “meet and greets” with congressional representatives increased their opposition to the bill. These efforts may work in favor of protecting patients from surprise medical bills. Hospital medicine physicians are on the front lines for providing health care in the hospital setting; they have demanded a fix to this legislative loophole which brings high out of pocket costs to our nation’s most vulnerable seniors. The observation status “2-midnight rule” utilized by CMS has increased financial barriers and decreased access to postacute care, affecting the provision of high-quality care for patients.
My hospital has a utilization review committee which reviews all cases to determine the appropriateness of an inpatient versus an observation designation. (An interesting question is whether the financial resources used to support this additional staff could be better assigned to provide high-quality care.) Distribution of these patients is determined on very specific criteria as outlined by Medicare. Observation is basically considered a billing method implemented by payers to decrease dollars paid to acute care hospitals for inpatient care. It pertains to admission status, not to the level of care provided in the hospital. Unfortunately, it is felt that no two payers define observation the same way. A few examples of common observation diagnoses are chest pain, abdominal pain, syncope, and migraine headache; in other words, patients with diagnoses where it is suspected that a less than 24-hour stay in the hospital could be sufficient.
Observation care is increasing and can sometimes contribute to work flow impediments and frustrations in hospitalists; thus, hospitalists are demanding reform. It has been proposed that observation could be eliminated altogether by creating a payment blend of inpatient/outpatient rates. Another option could be to assign lower Diagnosis Related Group coding to lower acuity disease processes, instead of separate observation reimbursement.
Patients and doctors lament that “Once you are in the hospital, you are admitted!” I don’t know the right answer that would solve the observation versus inpatient dilemma, but it is intriguing to consider changes in policy that might focus on the complete elimination of observation status.
Dr. Puri is a hospitalist at Lahey Hospital and Medical Center in Burlington, Mass.
A dilemma for hospitalists and patients
A dilemma for hospitalists and patients
A federal effort to reduce health care expenditures has left many older Medicare recipients experiencing the sticker shock of “observation status.” Patients who are not sick enough to meet inpatient admission criteria, however, still require hospitalization, and may be placed under Medicare observation care.
Seniors can get frustrated, confused, and anxious as their status can be changed while they are in the hospital, and they may receive large medical bills after they are discharged. The Centers for Medicare & Medicaid Services’ “3-day rule” mandates that Medicare will not pay for skilled nursing facility care unless the patient is admitted as an “inpatient” for at least 3 days. Observation days do not count towards this 3-day hospital stay.
There has been an increase in outpatient services over the years since 2006. The 2018 State of Hospital Medicine Report (SoHM) highlights the percentage of discharges based on hospitalists’ billed Current Procedural Terminology codes. Codes 99217 (observation discharge) and 99238-99239 (inpatient discharge) were used to calculate the percentages. 80.7% of adult medicine hospitalist discharges were coded using inpatient discharge codes, while 19.3% of patients were discharged with observation discharge codes.
In the 2016 SoHM report, the ratio was 76.0% inpatient and 21.1% observation codes and in the 2014 report we saw 80.3% inpatient and 16.1% observation discharges (see table 1). But in both of those surveys, same-day admission/discharge codes were also separately reported, which did not occur in 2018. That makes year-over-year comparison of the data challenging.
Interestingly, the 2017 CMS data on Evaluation and Management Codes by Specialty for the first time included separate data for hospitalists, based on hospitalists who credentialed with Medicare using the new C6 specialty code. Based on that data, when looking only at inpatient (99238-99239) and observation (99217) codes, 83% of the discharges were inpatient and 17% were observation.
Physicians feel the pressure of strained patient-physician relationships as a consequence of patients feeling the brunt of the financing gap related to observation status. Patients often feel they were not warned adequately about the financial ramifications of observation status. Even if Medicare beneficiaries have received the Medicare Outpatient Observation Notice, outlined by the Notice of Observation Treatment and Implication for Care Eligibility Act, they have no rights to appeal.
Currently Medicare beneficiaries admitted as inpatients only incur a Part A deductible; they are not liable for tests, procedures, and nursing care. On the other hand, in observation status all services are billed separately. For Medicare Part B services (which covers observation care) patients must pay 20% of services after the Part B deductible, which could result in a huge financial burden. Costs for skilled nursing facilities, when they are not covered by Medicare Part A, because of the 3-day rule, can easily go up to $20,000 or more. Medicare beneficiaries have no cap on costs for an observation stay. In some cases, hospitals have to apply a condition code 44 and retroactively change the stay to observation status.
I attended the 2019 Society of Hospital Medicine Annual Conference in Washington. Hospitalists from all parts of the country advocated on Capitol Hill against the “observation bill,” and “meet and greets” with congressional representatives increased their opposition to the bill. These efforts may work in favor of protecting patients from surprise medical bills. Hospital medicine physicians are on the front lines for providing health care in the hospital setting; they have demanded a fix to this legislative loophole which brings high out of pocket costs to our nation’s most vulnerable seniors. The observation status “2-midnight rule” utilized by CMS has increased financial barriers and decreased access to postacute care, affecting the provision of high-quality care for patients.
My hospital has a utilization review committee which reviews all cases to determine the appropriateness of an inpatient versus an observation designation. (An interesting question is whether the financial resources used to support this additional staff could be better assigned to provide high-quality care.) Distribution of these patients is determined on very specific criteria as outlined by Medicare. Observation is basically considered a billing method implemented by payers to decrease dollars paid to acute care hospitals for inpatient care. It pertains to admission status, not to the level of care provided in the hospital. Unfortunately, it is felt that no two payers define observation the same way. A few examples of common observation diagnoses are chest pain, abdominal pain, syncope, and migraine headache; in other words, patients with diagnoses where it is suspected that a less than 24-hour stay in the hospital could be sufficient.
Observation care is increasing and can sometimes contribute to work flow impediments and frustrations in hospitalists; thus, hospitalists are demanding reform. It has been proposed that observation could be eliminated altogether by creating a payment blend of inpatient/outpatient rates. Another option could be to assign lower Diagnosis Related Group coding to lower acuity disease processes, instead of separate observation reimbursement.
Patients and doctors lament that “Once you are in the hospital, you are admitted!” I don’t know the right answer that would solve the observation versus inpatient dilemma, but it is intriguing to consider changes in policy that might focus on the complete elimination of observation status.
Dr. Puri is a hospitalist at Lahey Hospital and Medical Center in Burlington, Mass.
A federal effort to reduce health care expenditures has left many older Medicare recipients experiencing the sticker shock of “observation status.” Patients who are not sick enough to meet inpatient admission criteria, however, still require hospitalization, and may be placed under Medicare observation care.
Seniors can get frustrated, confused, and anxious as their status can be changed while they are in the hospital, and they may receive large medical bills after they are discharged. The Centers for Medicare & Medicaid Services’ “3-day rule” mandates that Medicare will not pay for skilled nursing facility care unless the patient is admitted as an “inpatient” for at least 3 days. Observation days do not count towards this 3-day hospital stay.
There has been an increase in outpatient services over the years since 2006. The 2018 State of Hospital Medicine Report (SoHM) highlights the percentage of discharges based on hospitalists’ billed Current Procedural Terminology codes. Codes 99217 (observation discharge) and 99238-99239 (inpatient discharge) were used to calculate the percentages. 80.7% of adult medicine hospitalist discharges were coded using inpatient discharge codes, while 19.3% of patients were discharged with observation discharge codes.
In the 2016 SoHM report, the ratio was 76.0% inpatient and 21.1% observation codes and in the 2014 report we saw 80.3% inpatient and 16.1% observation discharges (see table 1). But in both of those surveys, same-day admission/discharge codes were also separately reported, which did not occur in 2018. That makes year-over-year comparison of the data challenging.
Interestingly, the 2017 CMS data on Evaluation and Management Codes by Specialty for the first time included separate data for hospitalists, based on hospitalists who credentialed with Medicare using the new C6 specialty code. Based on that data, when looking only at inpatient (99238-99239) and observation (99217) codes, 83% of the discharges were inpatient and 17% were observation.
Physicians feel the pressure of strained patient-physician relationships as a consequence of patients feeling the brunt of the financing gap related to observation status. Patients often feel they were not warned adequately about the financial ramifications of observation status. Even if Medicare beneficiaries have received the Medicare Outpatient Observation Notice, outlined by the Notice of Observation Treatment and Implication for Care Eligibility Act, they have no rights to appeal.
Currently Medicare beneficiaries admitted as inpatients only incur a Part A deductible; they are not liable for tests, procedures, and nursing care. On the other hand, in observation status all services are billed separately. For Medicare Part B services (which covers observation care) patients must pay 20% of services after the Part B deductible, which could result in a huge financial burden. Costs for skilled nursing facilities, when they are not covered by Medicare Part A, because of the 3-day rule, can easily go up to $20,000 or more. Medicare beneficiaries have no cap on costs for an observation stay. In some cases, hospitals have to apply a condition code 44 and retroactively change the stay to observation status.
I attended the 2019 Society of Hospital Medicine Annual Conference in Washington. Hospitalists from all parts of the country advocated on Capitol Hill against the “observation bill,” and “meet and greets” with congressional representatives increased their opposition to the bill. These efforts may work in favor of protecting patients from surprise medical bills. Hospital medicine physicians are on the front lines for providing health care in the hospital setting; they have demanded a fix to this legislative loophole which brings high out of pocket costs to our nation’s most vulnerable seniors. The observation status “2-midnight rule” utilized by CMS has increased financial barriers and decreased access to postacute care, affecting the provision of high-quality care for patients.
My hospital has a utilization review committee which reviews all cases to determine the appropriateness of an inpatient versus an observation designation. (An interesting question is whether the financial resources used to support this additional staff could be better assigned to provide high-quality care.) Distribution of these patients is determined on very specific criteria as outlined by Medicare. Observation is basically considered a billing method implemented by payers to decrease dollars paid to acute care hospitals for inpatient care. It pertains to admission status, not to the level of care provided in the hospital. Unfortunately, it is felt that no two payers define observation the same way. A few examples of common observation diagnoses are chest pain, abdominal pain, syncope, and migraine headache; in other words, patients with diagnoses where it is suspected that a less than 24-hour stay in the hospital could be sufficient.
Observation care is increasing and can sometimes contribute to work flow impediments and frustrations in hospitalists; thus, hospitalists are demanding reform. It has been proposed that observation could be eliminated altogether by creating a payment blend of inpatient/outpatient rates. Another option could be to assign lower Diagnosis Related Group coding to lower acuity disease processes, instead of separate observation reimbursement.
Patients and doctors lament that “Once you are in the hospital, you are admitted!” I don’t know the right answer that would solve the observation versus inpatient dilemma, but it is intriguing to consider changes in policy that might focus on the complete elimination of observation status.
Dr. Puri is a hospitalist at Lahey Hospital and Medical Center in Burlington, Mass.
Managing alcohol withdrawal in the hospitalized patient
Symptom-triggered therapy has multiple benefits
Case
A 57-year-old man with a history of alcohol abuse (no history of seizures) presents to the ED “feeling awful.” He claims his last drink was 1 day prior. Initial vital signs are: T = 99.1°F, HR 102 bpm, BP 162/85 mm Hg, respirations 18/minute, and 99% oxygen saturation. He is tremulous, diaphoretic, and has an unsteady gait. What is the best way to manage his symptoms while hospitalized?
Brief overview of the issue
With over 15 million people with alcohol use disorder (AUD) in the United States alone, alcohol dependence and misuse remain significant issues among hospitalized patients.1 It is estimated that over 20% of admitted patients meet DSM-5 criteria for AUD and that over 2 million will withdraw each year.2,3 Acute withdrawal includes a spectrum of symptoms ranging from mild anxiety and diaphoresis to hallucinations, seizures, and delirium tremens. Onset of these symptoms ranges from 24 hours up to 5 days.
Severe alcohol withdrawal syndrome (SAWS) attributable to abrupt discontinuation of alcohol leads to increased morbidity and mortality; therefore, early detection and prevention in the acute care setting is critical. Several factors can help predict who may withdraw, and once detected, pharmacological treatment is necessary.4 Thorough evaluation and treatment can help reduce mortality from the most severe forms of alcohol withdrawal including delirium tremens, which has up to 40% mortality if left untreated.5
Overview of the data
How do we use benzodiazepines to treat alcohol withdrawal?
Benzodiazepines are the mainstay of alcohol withdrawal treatment. Benzodiazepines work by stimulating the gamma-aminobutyric acid (GABA) receptor resulting in a reduction of neuronal activity. This leads to a sedative effect and thus slows the progression of withdrawal symptoms.
Long-acting benzodiazepines, such as chlordiazepoxide and diazepam, are the preferred choices for most patients. Their active metabolites have a rapid onset of action and their long half-lives allow for a lower incidence of breakthrough symptoms and rebound phenomena such as seizures.6 Benzodiazepines with shorter half-lives, such as lorazepam and oxazepam, are preferred in patients with liver dysfunction and those prone to respiratory depression.
Intravenous administration has a rapid onset of action and is the standard administration route of choice in patients with acute severe withdrawal, delirium tremens, and seizure activity. In patients with mild withdrawal symptoms or those in the outpatient setting, oral administration is generally effective.6
The Clinical Institute Withdrawal Assessment (CIWA) is one commonly used titration model that requires calculation of a symptom-based withdrawal score. Data have consistently demonstrated that a symptom-triggered method results in administration of less total benzodiazepines over a significantly shorter duration, thereby reducing cost and duration of treatment and minimizing side effects. This regimen may also reduce the risk of undermedicating or overmedicating a patient since the dosing is based upon an individual’s symptoms.7,8
The efficacy of symptom-triggered regimens however, depends on the reliability and accuracy of the patient assessment. A fixed-interval benzodiazepine-dosing approach where benzodiazepines are administered regardless of symptoms is useful when frequent monitoring and reassessment are not feasible or are unreliable.
What about phenobarbital?
Phenobarbital has similar pharmacokinetics to the benzodiazepines frequently used for alcohol withdrawal, including simultaneous effects on gamma-aminobutyric acid (GABA) and N-methyl-D-aspartate (NMDA) receptors, and has been proposed as a treatment option for delirium tremens.
In 2019, as reported in the American Journal of Emergency Medicine, Nelson et al. found that incorporating phenobarbital into a benzodiazepine-based protocol or as sole agent led to similar rates of ICU admission, length of stay, and need for mechanical ventilation in patients treated for alcohol withdrawal in the emergency department.9 The authors concluded that “phenobarbital (was) a safe and effective treatment alternative for alcohol withdrawal.” The systematic review by Hammond et al. in 2017 found that phenobarbital, either as monotherapy or in conjunction with benzodiazepines, could have comparable or superior results in comparison to other treatments, including benzodiazepines monotherapy.10 Further studies are needed to determine dosing and the most effective way to incorporate the use of phenobarbital in treatment of Alcohol Withdrawal Syndrome (AWS).
Should gabapentin or any other medications be added to his treatment regimen?
Chronic alcohol use induces a reduction in GABA activity (the major inhibitory neurotransmitter in the brain) and alcohol cessation results in decreased inhibitory tone. This physiologic imbalance contributes to the syndrome of alcohol withdrawal. As such, gabapentin has emerged as a promising treatment option in AWS and may help reduce the need for benzodiazepines.
Gabapentin has few drug-drug interactions and is safe for use in patients with impaired liver function; however, dosage adjustment is required for renal dysfunction (CrCl less than 60 mL/min). Gabapentin’s neuroprotective effects may also help decrease the neurotoxic effects associated with AWS. Common side effects of gabapentin include dizziness, drowsiness, ataxia, diarrhea, nausea, and vomiting. The potential for misuse has been reported.
In several small studies, gabapentin monotherapy was found to be comparable to benzodiazepines in the treatment of mild to moderate AWS. Gabapentin is efficacious in reducing cravings as well as improving mood, anxiety, and sleep, and showed an advantage over benzodiazepines in preventing relapse with no difference in length of hospital stay.6,11 Given the small sample sizes of these studies and the differing methods, settings, and inclusion/ exclusion criteria used, the generalizability of these findings to patients with significant medical and/or psychiatric comorbidities remains limited. Additional studies are needed to standardize dosing protocols and treatment strategies for both inpatients and outpatients.
Alternative agents such as antipsychotics (e.g., haloperidol), centrally acting alpha-2 agonists (e.g., clonidine), beta-blockers, and an agonist of the GABA-B receptor (e.g., baclofen) may also attenuate the symptoms of withdrawal. Since these all have limited evidence of their efficacy and have potential for harm, such as masking symptoms of progressive withdrawal and lowering seizure threshold, these agents are not routinely recommended for use. Valproic acid/divalproex, levetiracetam, topiramate, and zonisamide have also showed some efficacy in reducing symptoms of alcohol withdrawal in limited studies. The data on prevention of withdrawal seizures or delirium tremens when used as monotherapy is less robust.12
A daily multivitamin and folate are ordered. What about thiamine? Does the route matter?
Alarmingly, 80% of people who chronically abuse alcohol are thiamine deficient.13 This deficiency is attributable to several factors including inadequate oral intake, malabsorption, and decreased cellular utilization. Thiamine is a crucial factor in multiple enzymatic and metabolic pathways. Its deficiency can lead to free radical production, neurotoxicity, impaired glucose metabolism, and ultimately, cell death.14 A clinical concern stemming from thiamine deficiency is the development of Wernicke’s encephalopathy (WE), which is potentially reversible with prompt recognition and treatment, in comparison to its irreversible amnestic sequela, Korsakoff’s syndrome.
Wernicke’s encephalopathy had been defined as a triad of ataxia, ophthalmoplegia, and global confusion. However, Harper et al. discovered that only 16% of patients presented with the classic triad and 19% had none of these signs.15 Diagnosis is clinical since thiamine serology results do not accurately represent brain storage.
Currently, there are no consistent guidelines regarding repletion of thiamine administration in the treatment or prevention of WE attributable to alcohol overuse. Thiamine has a safe toxicity profile as excess thiamine is excreted in the urine. Outside of rare reports of anaphylactoid reactions involving large parenteral doses, there is no concern for overtreatment. As Wernicke-Korsakoff syndrome is associated with significant morbidity and mortality, high doses such as 200 to 500 mg are recommended to ensure blood-brain barrier passage. The intravenous route is optimal over oral administration to bypass concerns of gastrointestinal malabsorption. Thiamine 100 mg by mouth daily for ongoing supplementation can be considered for patients who are at risk for WE. It is also important to recognize that magnesium and thiamine are intertwined in several key enzymatic pathways. To optimize the responsiveness of thiamine repletion, magnesium levels should be tested and repleted if low.
Application of the data to our patient
Nurses are able to frequently monitor the patient so he is started on symptom-triggered treatment with chlordiazepoxide using the CIWA protocol. This strategy will help limit the amount of benzodiazepines he receives and shorten his treatment duration. Given the ataxia, the patient is also started on high-dose IV thiamine three times a day to treat possible Wernicke’s encephalopathy. Gabapentin is added to his regimen to help manage his moderate alcohol withdrawal syndrome.
Bottom line
Long-acting benzodiazepines using symptom-triggered administration when feasible are the mainstay of treating alcohol withdrawal. Other medications such as gabapentin, carbamazepine, and phenobarbital can be considered as adjunctive agents. Given the high rate of thiamine deficiency and the low risk of overreplacement, intravenous thiamine can be considered for inpatients with AWS.
Dr. Agrawal, Dr. Chernyavsky, Dr. Dharapak, Dr. Grabscheid, Dr. Merrill, Dr. Pillay, and Dr. Rizk are hospitalists at Mount Sinai Beth Israel in New York.
References
1. CDC - Fact Sheets: “Alcohol Use And Health – Alcohol.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 3 Jan. 2018.
2. Rawlani V et al. Treatment of the hospitalized alcohol-dependent patient with alcohol withdrawal syndrome. Internet J Intern Med. 2008;8(1).
3. Grant BF et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757.
4. Wood E et al. Will this hospitalized patient develop severe alcohol withdrawal syndrome?: The Rational Clinical Examination Systematic Review. JAMA. 2018;320:825.
5. Sarkar S et al. Risk factors for the development of delirium in alcohol dependence syndrome: Clinical and neurobiological implications. Indian J Psychiatry. 2017 Jul-Sep;59(3):300-5.
6. Sachdeva A et al. Alcohol withdrawal syndrome: Benzodiazepines and beyond. J Clinical Diagn Res. 2015 Sep 9(9).
7. Sullivan JT et al. Benzodiazepine requirements during alcohol withdrawal syndrome: Clinical implications of using a standardized withdrawal scale. J Clin Psychopharmacol. 1991;11:291-5.
8. Saitz R et al. Individualized treatment for alcohol withdrawal. A randomized double-blind controlled trial. JAMA. 1994;272(7):519.
9. Nelson AC et al. Benzodiazepines vs. barbiturates for alcohol withdrawal: Analysis of 3 different treatment protocols. Am J Emerg Med. 2019 Jan 3.
10. Hammond DA et al. Patient outcomes associated with phenobarbital use with or without benzodiazepines for alcohol withdrawal syndrome: A systematic review. Hosp Pharm. 2017 Oct;52(9):607-16.
11. Mo Y et al. Current practice patterns in the management of alcohol withdrawal syndrome. P T. 2018 Mar;43(3):158-62.
12. Leung JG et al. The role of gabapentin in the management of alcohol withdrawal and dependence. Ann Pharmacother. 2015 Aug;49(8):897-906.
13. Martin P et al. The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health. 2003:27(2):134-42.
14. Flannery A et al. Unpeeling the evidence for the banana bag: Evidence-based recommendations for the management of alcohol-associated vitamin and electrolyte deficiencies in the ICU. Crit Care Med. 2016 Aug:44(8):1545-52.
15. Harper CG et al. Clinical signs in Wernicke Korsakoff complex: A retrospective analysis of 131 cases diagnosed at autopsy. J Neurol Neurosurg Psychiatry. 1986;49(4):341-5.
Key points
- Alcohol use disorder and alcohol withdrawal are significant problems in hospitalized patients; early detection and treatment are crucial in preventing high morbidity and mortality.
- Long acting benzodiazepines with active metabolites such as chlordiazepoxide and diazepam are the preferred treatment for alcohol withdrawal except for patients with advanced liver disease or those prone to respiratory depression.
- Symptom-triggered therapy decreases the amount of medication, shortens treatment duration, and decreases inpatient length of stay, compared with fixed schedule dosing.
- Gabapentin may be effective in the treatment of mild to moderate AWS but cannot yet be routinely recommended as monotherapy in severe withdrawal, in patients with seizure history, or in patients who are at high risk for progression to delirium tremens.
- Thiamine deficiency is common in chronic alcohol use disorders; thiamine repletion should be considered for patients at risk or when Wernicke’s encephalopathy and Korsakoff’s syndrome are suspected.
Additional reading
1. Perry EC. Inpatient management of acute alcohol withdrawal syndrome. CNS Drugs. 2014;28(5):401-10.
2. Mayo-Smith MF. Pharmacological management of alcohol withdrawal: A meta-analysis and evidence-based practice guideline. JAMA. 1997;278(2):144-51.
3. Michael F. Mayo-Smith, MD, MPH et al. for the Working Group on the Management of Alcohol Withdrawal Delirium, Practice Guidelines Committee, American Society of Addiction Medicine. Management of alcohol withdrawal delirium: An evidence-based practice guideline. Arch Intern Med. 2004;164(13):1405-12.
Quiz
A 51-year-old female with a history of hypertension and continuous alcohol abuse presents to the hospital with fever and cough. She is found to have community-acquired pneumonia and is admitted for treatment. How else would you manage this patient?
A. Start scheduled benzodiazepines and oral thiamine.
B. Start CIWA protocol using a long-acting benzodiazepine and oral thiamine.
C. Start scheduled benzodiazepines and IV thiamine.
D. Start CIWA protocol using a long-acting benzodiazepine and consider IV or oral thiamine.
Answer: D. Symptom-triggered benzodiazepine therapy is favored as is consideration for thiamine repletion in the treatment of AWS.
Symptom-triggered therapy has multiple benefits
Symptom-triggered therapy has multiple benefits
Case
A 57-year-old man with a history of alcohol abuse (no history of seizures) presents to the ED “feeling awful.” He claims his last drink was 1 day prior. Initial vital signs are: T = 99.1°F, HR 102 bpm, BP 162/85 mm Hg, respirations 18/minute, and 99% oxygen saturation. He is tremulous, diaphoretic, and has an unsteady gait. What is the best way to manage his symptoms while hospitalized?
Brief overview of the issue
With over 15 million people with alcohol use disorder (AUD) in the United States alone, alcohol dependence and misuse remain significant issues among hospitalized patients.1 It is estimated that over 20% of admitted patients meet DSM-5 criteria for AUD and that over 2 million will withdraw each year.2,3 Acute withdrawal includes a spectrum of symptoms ranging from mild anxiety and diaphoresis to hallucinations, seizures, and delirium tremens. Onset of these symptoms ranges from 24 hours up to 5 days.
Severe alcohol withdrawal syndrome (SAWS) attributable to abrupt discontinuation of alcohol leads to increased morbidity and mortality; therefore, early detection and prevention in the acute care setting is critical. Several factors can help predict who may withdraw, and once detected, pharmacological treatment is necessary.4 Thorough evaluation and treatment can help reduce mortality from the most severe forms of alcohol withdrawal including delirium tremens, which has up to 40% mortality if left untreated.5
Overview of the data
How do we use benzodiazepines to treat alcohol withdrawal?
Benzodiazepines are the mainstay of alcohol withdrawal treatment. Benzodiazepines work by stimulating the gamma-aminobutyric acid (GABA) receptor resulting in a reduction of neuronal activity. This leads to a sedative effect and thus slows the progression of withdrawal symptoms.
Long-acting benzodiazepines, such as chlordiazepoxide and diazepam, are the preferred choices for most patients. Their active metabolites have a rapid onset of action and their long half-lives allow for a lower incidence of breakthrough symptoms and rebound phenomena such as seizures.6 Benzodiazepines with shorter half-lives, such as lorazepam and oxazepam, are preferred in patients with liver dysfunction and those prone to respiratory depression.
Intravenous administration has a rapid onset of action and is the standard administration route of choice in patients with acute severe withdrawal, delirium tremens, and seizure activity. In patients with mild withdrawal symptoms or those in the outpatient setting, oral administration is generally effective.6
The Clinical Institute Withdrawal Assessment (CIWA) is one commonly used titration model that requires calculation of a symptom-based withdrawal score. Data have consistently demonstrated that a symptom-triggered method results in administration of less total benzodiazepines over a significantly shorter duration, thereby reducing cost and duration of treatment and minimizing side effects. This regimen may also reduce the risk of undermedicating or overmedicating a patient since the dosing is based upon an individual’s symptoms.7,8
The efficacy of symptom-triggered regimens however, depends on the reliability and accuracy of the patient assessment. A fixed-interval benzodiazepine-dosing approach where benzodiazepines are administered regardless of symptoms is useful when frequent monitoring and reassessment are not feasible or are unreliable.
What about phenobarbital?
Phenobarbital has similar pharmacokinetics to the benzodiazepines frequently used for alcohol withdrawal, including simultaneous effects on gamma-aminobutyric acid (GABA) and N-methyl-D-aspartate (NMDA) receptors, and has been proposed as a treatment option for delirium tremens.
In 2019, as reported in the American Journal of Emergency Medicine, Nelson et al. found that incorporating phenobarbital into a benzodiazepine-based protocol or as sole agent led to similar rates of ICU admission, length of stay, and need for mechanical ventilation in patients treated for alcohol withdrawal in the emergency department.9 The authors concluded that “phenobarbital (was) a safe and effective treatment alternative for alcohol withdrawal.” The systematic review by Hammond et al. in 2017 found that phenobarbital, either as monotherapy or in conjunction with benzodiazepines, could have comparable or superior results in comparison to other treatments, including benzodiazepines monotherapy.10 Further studies are needed to determine dosing and the most effective way to incorporate the use of phenobarbital in treatment of Alcohol Withdrawal Syndrome (AWS).
Should gabapentin or any other medications be added to his treatment regimen?
Chronic alcohol use induces a reduction in GABA activity (the major inhibitory neurotransmitter in the brain) and alcohol cessation results in decreased inhibitory tone. This physiologic imbalance contributes to the syndrome of alcohol withdrawal. As such, gabapentin has emerged as a promising treatment option in AWS and may help reduce the need for benzodiazepines.
Gabapentin has few drug-drug interactions and is safe for use in patients with impaired liver function; however, dosage adjustment is required for renal dysfunction (CrCl less than 60 mL/min). Gabapentin’s neuroprotective effects may also help decrease the neurotoxic effects associated with AWS. Common side effects of gabapentin include dizziness, drowsiness, ataxia, diarrhea, nausea, and vomiting. The potential for misuse has been reported.
In several small studies, gabapentin monotherapy was found to be comparable to benzodiazepines in the treatment of mild to moderate AWS. Gabapentin is efficacious in reducing cravings as well as improving mood, anxiety, and sleep, and showed an advantage over benzodiazepines in preventing relapse with no difference in length of hospital stay.6,11 Given the small sample sizes of these studies and the differing methods, settings, and inclusion/ exclusion criteria used, the generalizability of these findings to patients with significant medical and/or psychiatric comorbidities remains limited. Additional studies are needed to standardize dosing protocols and treatment strategies for both inpatients and outpatients.
Alternative agents such as antipsychotics (e.g., haloperidol), centrally acting alpha-2 agonists (e.g., clonidine), beta-blockers, and an agonist of the GABA-B receptor (e.g., baclofen) may also attenuate the symptoms of withdrawal. Since these all have limited evidence of their efficacy and have potential for harm, such as masking symptoms of progressive withdrawal and lowering seizure threshold, these agents are not routinely recommended for use. Valproic acid/divalproex, levetiracetam, topiramate, and zonisamide have also showed some efficacy in reducing symptoms of alcohol withdrawal in limited studies. The data on prevention of withdrawal seizures or delirium tremens when used as monotherapy is less robust.12
A daily multivitamin and folate are ordered. What about thiamine? Does the route matter?
Alarmingly, 80% of people who chronically abuse alcohol are thiamine deficient.13 This deficiency is attributable to several factors including inadequate oral intake, malabsorption, and decreased cellular utilization. Thiamine is a crucial factor in multiple enzymatic and metabolic pathways. Its deficiency can lead to free radical production, neurotoxicity, impaired glucose metabolism, and ultimately, cell death.14 A clinical concern stemming from thiamine deficiency is the development of Wernicke’s encephalopathy (WE), which is potentially reversible with prompt recognition and treatment, in comparison to its irreversible amnestic sequela, Korsakoff’s syndrome.
Wernicke’s encephalopathy had been defined as a triad of ataxia, ophthalmoplegia, and global confusion. However, Harper et al. discovered that only 16% of patients presented with the classic triad and 19% had none of these signs.15 Diagnosis is clinical since thiamine serology results do not accurately represent brain storage.
Currently, there are no consistent guidelines regarding repletion of thiamine administration in the treatment or prevention of WE attributable to alcohol overuse. Thiamine has a safe toxicity profile as excess thiamine is excreted in the urine. Outside of rare reports of anaphylactoid reactions involving large parenteral doses, there is no concern for overtreatment. As Wernicke-Korsakoff syndrome is associated with significant morbidity and mortality, high doses such as 200 to 500 mg are recommended to ensure blood-brain barrier passage. The intravenous route is optimal over oral administration to bypass concerns of gastrointestinal malabsorption. Thiamine 100 mg by mouth daily for ongoing supplementation can be considered for patients who are at risk for WE. It is also important to recognize that magnesium and thiamine are intertwined in several key enzymatic pathways. To optimize the responsiveness of thiamine repletion, magnesium levels should be tested and repleted if low.
Application of the data to our patient
Nurses are able to frequently monitor the patient so he is started on symptom-triggered treatment with chlordiazepoxide using the CIWA protocol. This strategy will help limit the amount of benzodiazepines he receives and shorten his treatment duration. Given the ataxia, the patient is also started on high-dose IV thiamine three times a day to treat possible Wernicke’s encephalopathy. Gabapentin is added to his regimen to help manage his moderate alcohol withdrawal syndrome.
Bottom line
Long-acting benzodiazepines using symptom-triggered administration when feasible are the mainstay of treating alcohol withdrawal. Other medications such as gabapentin, carbamazepine, and phenobarbital can be considered as adjunctive agents. Given the high rate of thiamine deficiency and the low risk of overreplacement, intravenous thiamine can be considered for inpatients with AWS.
Dr. Agrawal, Dr. Chernyavsky, Dr. Dharapak, Dr. Grabscheid, Dr. Merrill, Dr. Pillay, and Dr. Rizk are hospitalists at Mount Sinai Beth Israel in New York.
References
1. CDC - Fact Sheets: “Alcohol Use And Health – Alcohol.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 3 Jan. 2018.
2. Rawlani V et al. Treatment of the hospitalized alcohol-dependent patient with alcohol withdrawal syndrome. Internet J Intern Med. 2008;8(1).
3. Grant BF et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757.
4. Wood E et al. Will this hospitalized patient develop severe alcohol withdrawal syndrome?: The Rational Clinical Examination Systematic Review. JAMA. 2018;320:825.
5. Sarkar S et al. Risk factors for the development of delirium in alcohol dependence syndrome: Clinical and neurobiological implications. Indian J Psychiatry. 2017 Jul-Sep;59(3):300-5.
6. Sachdeva A et al. Alcohol withdrawal syndrome: Benzodiazepines and beyond. J Clinical Diagn Res. 2015 Sep 9(9).
7. Sullivan JT et al. Benzodiazepine requirements during alcohol withdrawal syndrome: Clinical implications of using a standardized withdrawal scale. J Clin Psychopharmacol. 1991;11:291-5.
8. Saitz R et al. Individualized treatment for alcohol withdrawal. A randomized double-blind controlled trial. JAMA. 1994;272(7):519.
9. Nelson AC et al. Benzodiazepines vs. barbiturates for alcohol withdrawal: Analysis of 3 different treatment protocols. Am J Emerg Med. 2019 Jan 3.
10. Hammond DA et al. Patient outcomes associated with phenobarbital use with or without benzodiazepines for alcohol withdrawal syndrome: A systematic review. Hosp Pharm. 2017 Oct;52(9):607-16.
11. Mo Y et al. Current practice patterns in the management of alcohol withdrawal syndrome. P T. 2018 Mar;43(3):158-62.
12. Leung JG et al. The role of gabapentin in the management of alcohol withdrawal and dependence. Ann Pharmacother. 2015 Aug;49(8):897-906.
13. Martin P et al. The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health. 2003:27(2):134-42.
14. Flannery A et al. Unpeeling the evidence for the banana bag: Evidence-based recommendations for the management of alcohol-associated vitamin and electrolyte deficiencies in the ICU. Crit Care Med. 2016 Aug:44(8):1545-52.
15. Harper CG et al. Clinical signs in Wernicke Korsakoff complex: A retrospective analysis of 131 cases diagnosed at autopsy. J Neurol Neurosurg Psychiatry. 1986;49(4):341-5.
Key points
- Alcohol use disorder and alcohol withdrawal are significant problems in hospitalized patients; early detection and treatment are crucial in preventing high morbidity and mortality.
- Long acting benzodiazepines with active metabolites such as chlordiazepoxide and diazepam are the preferred treatment for alcohol withdrawal except for patients with advanced liver disease or those prone to respiratory depression.
- Symptom-triggered therapy decreases the amount of medication, shortens treatment duration, and decreases inpatient length of stay, compared with fixed schedule dosing.
- Gabapentin may be effective in the treatment of mild to moderate AWS but cannot yet be routinely recommended as monotherapy in severe withdrawal, in patients with seizure history, or in patients who are at high risk for progression to delirium tremens.
- Thiamine deficiency is common in chronic alcohol use disorders; thiamine repletion should be considered for patients at risk or when Wernicke’s encephalopathy and Korsakoff’s syndrome are suspected.
Additional reading
1. Perry EC. Inpatient management of acute alcohol withdrawal syndrome. CNS Drugs. 2014;28(5):401-10.
2. Mayo-Smith MF. Pharmacological management of alcohol withdrawal: A meta-analysis and evidence-based practice guideline. JAMA. 1997;278(2):144-51.
3. Michael F. Mayo-Smith, MD, MPH et al. for the Working Group on the Management of Alcohol Withdrawal Delirium, Practice Guidelines Committee, American Society of Addiction Medicine. Management of alcohol withdrawal delirium: An evidence-based practice guideline. Arch Intern Med. 2004;164(13):1405-12.
Quiz
A 51-year-old female with a history of hypertension and continuous alcohol abuse presents to the hospital with fever and cough. She is found to have community-acquired pneumonia and is admitted for treatment. How else would you manage this patient?
A. Start scheduled benzodiazepines and oral thiamine.
B. Start CIWA protocol using a long-acting benzodiazepine and oral thiamine.
C. Start scheduled benzodiazepines and IV thiamine.
D. Start CIWA protocol using a long-acting benzodiazepine and consider IV or oral thiamine.
Answer: D. Symptom-triggered benzodiazepine therapy is favored as is consideration for thiamine repletion in the treatment of AWS.
Case
A 57-year-old man with a history of alcohol abuse (no history of seizures) presents to the ED “feeling awful.” He claims his last drink was 1 day prior. Initial vital signs are: T = 99.1°F, HR 102 bpm, BP 162/85 mm Hg, respirations 18/minute, and 99% oxygen saturation. He is tremulous, diaphoretic, and has an unsteady gait. What is the best way to manage his symptoms while hospitalized?
Brief overview of the issue
With over 15 million people with alcohol use disorder (AUD) in the United States alone, alcohol dependence and misuse remain significant issues among hospitalized patients.1 It is estimated that over 20% of admitted patients meet DSM-5 criteria for AUD and that over 2 million will withdraw each year.2,3 Acute withdrawal includes a spectrum of symptoms ranging from mild anxiety and diaphoresis to hallucinations, seizures, and delirium tremens. Onset of these symptoms ranges from 24 hours up to 5 days.
Severe alcohol withdrawal syndrome (SAWS) attributable to abrupt discontinuation of alcohol leads to increased morbidity and mortality; therefore, early detection and prevention in the acute care setting is critical. Several factors can help predict who may withdraw, and once detected, pharmacological treatment is necessary.4 Thorough evaluation and treatment can help reduce mortality from the most severe forms of alcohol withdrawal including delirium tremens, which has up to 40% mortality if left untreated.5
Overview of the data
How do we use benzodiazepines to treat alcohol withdrawal?
Benzodiazepines are the mainstay of alcohol withdrawal treatment. Benzodiazepines work by stimulating the gamma-aminobutyric acid (GABA) receptor resulting in a reduction of neuronal activity. This leads to a sedative effect and thus slows the progression of withdrawal symptoms.
Long-acting benzodiazepines, such as chlordiazepoxide and diazepam, are the preferred choices for most patients. Their active metabolites have a rapid onset of action and their long half-lives allow for a lower incidence of breakthrough symptoms and rebound phenomena such as seizures.6 Benzodiazepines with shorter half-lives, such as lorazepam and oxazepam, are preferred in patients with liver dysfunction and those prone to respiratory depression.
Intravenous administration has a rapid onset of action and is the standard administration route of choice in patients with acute severe withdrawal, delirium tremens, and seizure activity. In patients with mild withdrawal symptoms or those in the outpatient setting, oral administration is generally effective.6
The Clinical Institute Withdrawal Assessment (CIWA) is one commonly used titration model that requires calculation of a symptom-based withdrawal score. Data have consistently demonstrated that a symptom-triggered method results in administration of less total benzodiazepines over a significantly shorter duration, thereby reducing cost and duration of treatment and minimizing side effects. This regimen may also reduce the risk of undermedicating or overmedicating a patient since the dosing is based upon an individual’s symptoms.7,8
The efficacy of symptom-triggered regimens however, depends on the reliability and accuracy of the patient assessment. A fixed-interval benzodiazepine-dosing approach where benzodiazepines are administered regardless of symptoms is useful when frequent monitoring and reassessment are not feasible or are unreliable.
What about phenobarbital?
Phenobarbital has similar pharmacokinetics to the benzodiazepines frequently used for alcohol withdrawal, including simultaneous effects on gamma-aminobutyric acid (GABA) and N-methyl-D-aspartate (NMDA) receptors, and has been proposed as a treatment option for delirium tremens.
In 2019, as reported in the American Journal of Emergency Medicine, Nelson et al. found that incorporating phenobarbital into a benzodiazepine-based protocol or as sole agent led to similar rates of ICU admission, length of stay, and need for mechanical ventilation in patients treated for alcohol withdrawal in the emergency department.9 The authors concluded that “phenobarbital (was) a safe and effective treatment alternative for alcohol withdrawal.” The systematic review by Hammond et al. in 2017 found that phenobarbital, either as monotherapy or in conjunction with benzodiazepines, could have comparable or superior results in comparison to other treatments, including benzodiazepines monotherapy.10 Further studies are needed to determine dosing and the most effective way to incorporate the use of phenobarbital in treatment of Alcohol Withdrawal Syndrome (AWS).
Should gabapentin or any other medications be added to his treatment regimen?
Chronic alcohol use induces a reduction in GABA activity (the major inhibitory neurotransmitter in the brain) and alcohol cessation results in decreased inhibitory tone. This physiologic imbalance contributes to the syndrome of alcohol withdrawal. As such, gabapentin has emerged as a promising treatment option in AWS and may help reduce the need for benzodiazepines.
Gabapentin has few drug-drug interactions and is safe for use in patients with impaired liver function; however, dosage adjustment is required for renal dysfunction (CrCl less than 60 mL/min). Gabapentin’s neuroprotective effects may also help decrease the neurotoxic effects associated with AWS. Common side effects of gabapentin include dizziness, drowsiness, ataxia, diarrhea, nausea, and vomiting. The potential for misuse has been reported.
In several small studies, gabapentin monotherapy was found to be comparable to benzodiazepines in the treatment of mild to moderate AWS. Gabapentin is efficacious in reducing cravings as well as improving mood, anxiety, and sleep, and showed an advantage over benzodiazepines in preventing relapse with no difference in length of hospital stay.6,11 Given the small sample sizes of these studies and the differing methods, settings, and inclusion/ exclusion criteria used, the generalizability of these findings to patients with significant medical and/or psychiatric comorbidities remains limited. Additional studies are needed to standardize dosing protocols and treatment strategies for both inpatients and outpatients.
Alternative agents such as antipsychotics (e.g., haloperidol), centrally acting alpha-2 agonists (e.g., clonidine), beta-blockers, and an agonist of the GABA-B receptor (e.g., baclofen) may also attenuate the symptoms of withdrawal. Since these all have limited evidence of their efficacy and have potential for harm, such as masking symptoms of progressive withdrawal and lowering seizure threshold, these agents are not routinely recommended for use. Valproic acid/divalproex, levetiracetam, topiramate, and zonisamide have also showed some efficacy in reducing symptoms of alcohol withdrawal in limited studies. The data on prevention of withdrawal seizures or delirium tremens when used as monotherapy is less robust.12
A daily multivitamin and folate are ordered. What about thiamine? Does the route matter?
Alarmingly, 80% of people who chronically abuse alcohol are thiamine deficient.13 This deficiency is attributable to several factors including inadequate oral intake, malabsorption, and decreased cellular utilization. Thiamine is a crucial factor in multiple enzymatic and metabolic pathways. Its deficiency can lead to free radical production, neurotoxicity, impaired glucose metabolism, and ultimately, cell death.14 A clinical concern stemming from thiamine deficiency is the development of Wernicke’s encephalopathy (WE), which is potentially reversible with prompt recognition and treatment, in comparison to its irreversible amnestic sequela, Korsakoff’s syndrome.
Wernicke’s encephalopathy had been defined as a triad of ataxia, ophthalmoplegia, and global confusion. However, Harper et al. discovered that only 16% of patients presented with the classic triad and 19% had none of these signs.15 Diagnosis is clinical since thiamine serology results do not accurately represent brain storage.
Currently, there are no consistent guidelines regarding repletion of thiamine administration in the treatment or prevention of WE attributable to alcohol overuse. Thiamine has a safe toxicity profile as excess thiamine is excreted in the urine. Outside of rare reports of anaphylactoid reactions involving large parenteral doses, there is no concern for overtreatment. As Wernicke-Korsakoff syndrome is associated with significant morbidity and mortality, high doses such as 200 to 500 mg are recommended to ensure blood-brain barrier passage. The intravenous route is optimal over oral administration to bypass concerns of gastrointestinal malabsorption. Thiamine 100 mg by mouth daily for ongoing supplementation can be considered for patients who are at risk for WE. It is also important to recognize that magnesium and thiamine are intertwined in several key enzymatic pathways. To optimize the responsiveness of thiamine repletion, magnesium levels should be tested and repleted if low.
Application of the data to our patient
Nurses are able to frequently monitor the patient so he is started on symptom-triggered treatment with chlordiazepoxide using the CIWA protocol. This strategy will help limit the amount of benzodiazepines he receives and shorten his treatment duration. Given the ataxia, the patient is also started on high-dose IV thiamine three times a day to treat possible Wernicke’s encephalopathy. Gabapentin is added to his regimen to help manage his moderate alcohol withdrawal syndrome.
Bottom line
Long-acting benzodiazepines using symptom-triggered administration when feasible are the mainstay of treating alcohol withdrawal. Other medications such as gabapentin, carbamazepine, and phenobarbital can be considered as adjunctive agents. Given the high rate of thiamine deficiency and the low risk of overreplacement, intravenous thiamine can be considered for inpatients with AWS.
Dr. Agrawal, Dr. Chernyavsky, Dr. Dharapak, Dr. Grabscheid, Dr. Merrill, Dr. Pillay, and Dr. Rizk are hospitalists at Mount Sinai Beth Israel in New York.
References
1. CDC - Fact Sheets: “Alcohol Use And Health – Alcohol.” Centers for Disease Control and Prevention, Centers for Disease Control and Prevention, 3 Jan. 2018.
2. Rawlani V et al. Treatment of the hospitalized alcohol-dependent patient with alcohol withdrawal syndrome. Internet J Intern Med. 2008;8(1).
3. Grant BF et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72:757.
4. Wood E et al. Will this hospitalized patient develop severe alcohol withdrawal syndrome?: The Rational Clinical Examination Systematic Review. JAMA. 2018;320:825.
5. Sarkar S et al. Risk factors for the development of delirium in alcohol dependence syndrome: Clinical and neurobiological implications. Indian J Psychiatry. 2017 Jul-Sep;59(3):300-5.
6. Sachdeva A et al. Alcohol withdrawal syndrome: Benzodiazepines and beyond. J Clinical Diagn Res. 2015 Sep 9(9).
7. Sullivan JT et al. Benzodiazepine requirements during alcohol withdrawal syndrome: Clinical implications of using a standardized withdrawal scale. J Clin Psychopharmacol. 1991;11:291-5.
8. Saitz R et al. Individualized treatment for alcohol withdrawal. A randomized double-blind controlled trial. JAMA. 1994;272(7):519.
9. Nelson AC et al. Benzodiazepines vs. barbiturates for alcohol withdrawal: Analysis of 3 different treatment protocols. Am J Emerg Med. 2019 Jan 3.
10. Hammond DA et al. Patient outcomes associated with phenobarbital use with or without benzodiazepines for alcohol withdrawal syndrome: A systematic review. Hosp Pharm. 2017 Oct;52(9):607-16.
11. Mo Y et al. Current practice patterns in the management of alcohol withdrawal syndrome. P T. 2018 Mar;43(3):158-62.
12. Leung JG et al. The role of gabapentin in the management of alcohol withdrawal and dependence. Ann Pharmacother. 2015 Aug;49(8):897-906.
13. Martin P et al. The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health. 2003:27(2):134-42.
14. Flannery A et al. Unpeeling the evidence for the banana bag: Evidence-based recommendations for the management of alcohol-associated vitamin and electrolyte deficiencies in the ICU. Crit Care Med. 2016 Aug:44(8):1545-52.
15. Harper CG et al. Clinical signs in Wernicke Korsakoff complex: A retrospective analysis of 131 cases diagnosed at autopsy. J Neurol Neurosurg Psychiatry. 1986;49(4):341-5.
Key points
- Alcohol use disorder and alcohol withdrawal are significant problems in hospitalized patients; early detection and treatment are crucial in preventing high morbidity and mortality.
- Long acting benzodiazepines with active metabolites such as chlordiazepoxide and diazepam are the preferred treatment for alcohol withdrawal except for patients with advanced liver disease or those prone to respiratory depression.
- Symptom-triggered therapy decreases the amount of medication, shortens treatment duration, and decreases inpatient length of stay, compared with fixed schedule dosing.
- Gabapentin may be effective in the treatment of mild to moderate AWS but cannot yet be routinely recommended as monotherapy in severe withdrawal, in patients with seizure history, or in patients who are at high risk for progression to delirium tremens.
- Thiamine deficiency is common in chronic alcohol use disorders; thiamine repletion should be considered for patients at risk or when Wernicke’s encephalopathy and Korsakoff’s syndrome are suspected.
Additional reading
1. Perry EC. Inpatient management of acute alcohol withdrawal syndrome. CNS Drugs. 2014;28(5):401-10.
2. Mayo-Smith MF. Pharmacological management of alcohol withdrawal: A meta-analysis and evidence-based practice guideline. JAMA. 1997;278(2):144-51.
3. Michael F. Mayo-Smith, MD, MPH et al. for the Working Group on the Management of Alcohol Withdrawal Delirium, Practice Guidelines Committee, American Society of Addiction Medicine. Management of alcohol withdrawal delirium: An evidence-based practice guideline. Arch Intern Med. 2004;164(13):1405-12.
Quiz
A 51-year-old female with a history of hypertension and continuous alcohol abuse presents to the hospital with fever and cough. She is found to have community-acquired pneumonia and is admitted for treatment. How else would you manage this patient?
A. Start scheduled benzodiazepines and oral thiamine.
B. Start CIWA protocol using a long-acting benzodiazepine and oral thiamine.
C. Start scheduled benzodiazepines and IV thiamine.
D. Start CIWA protocol using a long-acting benzodiazepine and consider IV or oral thiamine.
Answer: D. Symptom-triggered benzodiazepine therapy is favored as is consideration for thiamine repletion in the treatment of AWS.
Hospital-acquired C. diff. tied to four ‘high-risk’ antibiotic classes
The use of four antibiotic classes designated “high risk” was found to be an independent predictor of hospital-acquired Clostridioides difficile (CDI), based upon an analysis of microbiologic and pharmacy data from 171 hospitals in the United States.
The high-risk antibiotic classes were second-, third-, and fourth-generation cephalosporins, fluoroquinolones, carbapenems, and lincosamides, according to a report by Ying P. Tabak, PhD, of Becton Dickinson in Franklin Lakes, N.J., and colleagues published in Infection Control & Hospital Epidemiology.
Of the 171 study sites studied, 66 (39%) were teaching hospitals and 105 (61%) were nonteaching hospitals. The high-risk antibiotics most frequently used were cephalosporins (47.9%), fluoroquinolones (31.6%), carbapenems (13.0%), and lincosamides (7.6%). The sites were distributed across various regions of the United States. The hospital-level antibiotic use was measured as days of therapy (DOT) per 1,000 days present (DP).
The study was not able to determine specific links to individual antibiotic classes but to the use of high-risk antibiotics as a whole, except for cephalosporins, which were significantly correlated with hospital-acquired CDI (r = 0.23; P less than .01).
The overall correlation of high-risk antibiotic use and hospital-acquired CDI was 0.22 (P = .003). Higher correlation was observed in teaching hospitals (r = 0.38; P = .002) versus nonteaching hospitals (r = 0.19; P = .055), according to the researchers. The authors attributed this to the possibility of teaching hospitals dealing with more elderly and sicker patients.
After adjusting for significant confounders, the use of high-risk antibiotics was still independently associated with significant risk for hospital-acquired CDI. “For every 100-day increase of DOT per 1,000 DP in high-risk antibiotic use, there was a 12% increase in [hospital-acquired] CDI (RR, 1.12; 95% [confidence interval], 1.04-1.21; P = .002),” according to the authors. This translated to four additional hospital-acquired CDI cases with every 100 DOT increase per 1,000 DP.
“Using a large and current dataset, we found an independent impact of hospital-level high-risk antibiotic use on [hospital-acquired] CDI even after adjusting for confounding factors such as community CDI pressure, proportion of patients aged 65 years or older, average length of stay, and hospital teaching status,” the researchers concluded.
Funding was provided by Nabriva Therapeutics, an antibiotic development company. Four of the authors are full-time employees of Becton Dickinson, which sells diagnostics for infectious diseases, including CDI, and one author was an employee of Nabriva Therapeutics.
SOURCE: Tabak YP et al. Infect Control Hosp Epidemiol. 2019 Sep 16. doi: 10.1017/ice.2019.236.
The use of four antibiotic classes designated “high risk” was found to be an independent predictor of hospital-acquired Clostridioides difficile (CDI), based upon an analysis of microbiologic and pharmacy data from 171 hospitals in the United States.
The high-risk antibiotic classes were second-, third-, and fourth-generation cephalosporins, fluoroquinolones, carbapenems, and lincosamides, according to a report by Ying P. Tabak, PhD, of Becton Dickinson in Franklin Lakes, N.J., and colleagues published in Infection Control & Hospital Epidemiology.
Of the 171 study sites studied, 66 (39%) were teaching hospitals and 105 (61%) were nonteaching hospitals. The high-risk antibiotics most frequently used were cephalosporins (47.9%), fluoroquinolones (31.6%), carbapenems (13.0%), and lincosamides (7.6%). The sites were distributed across various regions of the United States. The hospital-level antibiotic use was measured as days of therapy (DOT) per 1,000 days present (DP).
The study was not able to determine specific links to individual antibiotic classes but to the use of high-risk antibiotics as a whole, except for cephalosporins, which were significantly correlated with hospital-acquired CDI (r = 0.23; P less than .01).
The overall correlation of high-risk antibiotic use and hospital-acquired CDI was 0.22 (P = .003). Higher correlation was observed in teaching hospitals (r = 0.38; P = .002) versus nonteaching hospitals (r = 0.19; P = .055), according to the researchers. The authors attributed this to the possibility of teaching hospitals dealing with more elderly and sicker patients.
After adjusting for significant confounders, the use of high-risk antibiotics was still independently associated with significant risk for hospital-acquired CDI. “For every 100-day increase of DOT per 1,000 DP in high-risk antibiotic use, there was a 12% increase in [hospital-acquired] CDI (RR, 1.12; 95% [confidence interval], 1.04-1.21; P = .002),” according to the authors. This translated to four additional hospital-acquired CDI cases with every 100 DOT increase per 1,000 DP.
“Using a large and current dataset, we found an independent impact of hospital-level high-risk antibiotic use on [hospital-acquired] CDI even after adjusting for confounding factors such as community CDI pressure, proportion of patients aged 65 years or older, average length of stay, and hospital teaching status,” the researchers concluded.
Funding was provided by Nabriva Therapeutics, an antibiotic development company. Four of the authors are full-time employees of Becton Dickinson, which sells diagnostics for infectious diseases, including CDI, and one author was an employee of Nabriva Therapeutics.
SOURCE: Tabak YP et al. Infect Control Hosp Epidemiol. 2019 Sep 16. doi: 10.1017/ice.2019.236.
The use of four antibiotic classes designated “high risk” was found to be an independent predictor of hospital-acquired Clostridioides difficile (CDI), based upon an analysis of microbiologic and pharmacy data from 171 hospitals in the United States.
The high-risk antibiotic classes were second-, third-, and fourth-generation cephalosporins, fluoroquinolones, carbapenems, and lincosamides, according to a report by Ying P. Tabak, PhD, of Becton Dickinson in Franklin Lakes, N.J., and colleagues published in Infection Control & Hospital Epidemiology.
Of the 171 study sites studied, 66 (39%) were teaching hospitals and 105 (61%) were nonteaching hospitals. The high-risk antibiotics most frequently used were cephalosporins (47.9%), fluoroquinolones (31.6%), carbapenems (13.0%), and lincosamides (7.6%). The sites were distributed across various regions of the United States. The hospital-level antibiotic use was measured as days of therapy (DOT) per 1,000 days present (DP).
The study was not able to determine specific links to individual antibiotic classes but to the use of high-risk antibiotics as a whole, except for cephalosporins, which were significantly correlated with hospital-acquired CDI (r = 0.23; P less than .01).
The overall correlation of high-risk antibiotic use and hospital-acquired CDI was 0.22 (P = .003). Higher correlation was observed in teaching hospitals (r = 0.38; P = .002) versus nonteaching hospitals (r = 0.19; P = .055), according to the researchers. The authors attributed this to the possibility of teaching hospitals dealing with more elderly and sicker patients.
After adjusting for significant confounders, the use of high-risk antibiotics was still independently associated with significant risk for hospital-acquired CDI. “For every 100-day increase of DOT per 1,000 DP in high-risk antibiotic use, there was a 12% increase in [hospital-acquired] CDI (RR, 1.12; 95% [confidence interval], 1.04-1.21; P = .002),” according to the authors. This translated to four additional hospital-acquired CDI cases with every 100 DOT increase per 1,000 DP.
“Using a large and current dataset, we found an independent impact of hospital-level high-risk antibiotic use on [hospital-acquired] CDI even after adjusting for confounding factors such as community CDI pressure, proportion of patients aged 65 years or older, average length of stay, and hospital teaching status,” the researchers concluded.
Funding was provided by Nabriva Therapeutics, an antibiotic development company. Four of the authors are full-time employees of Becton Dickinson, which sells diagnostics for infectious diseases, including CDI, and one author was an employee of Nabriva Therapeutics.
SOURCE: Tabak YP et al. Infect Control Hosp Epidemiol. 2019 Sep 16. doi: 10.1017/ice.2019.236.
FROM INFECTION CONTROL & HOSPITAL EPIDEMIOLOGY
Key clinical point:
Major finding: For every 100-day increase in high-risk antibiotic therapy, there was a 12% increase in hospital-acquired C. difficile.
Study details: Microbiological and pharmacy data from 171 hospitals comparing hospitalwide use of four antibiotics classes on hospital-acquired C. difficile.
Disclosures: Funding was provided Nabriva Therapeutics, an antibiotic development company. Four of the authors are full-time employees of Becton Dickinson, which sells diagnostics for infectious diseases, including C. difficile, and one author was an employee of Nabriva Therapeutics.
Source: Tabak YP et al. Infect Control Hosp Epidemiol. 2019 Sep 16. doi: 10.1017/ice.2019.236.
Introducing SHM’s president-elect
Hoping to expand membership beyond the traditional ‘core’
It is with great pleasure that I enter my president-elect year for the Society of Hospital Medicine! I am hopeful that this year will allow me time to get to know the organization even better than I already do, and truly understand the needs of our members so I can focus on meeting and exceeding your expectations!
I have been a hospitalist now for 17 years and have practiced in both academic tertiary care and community hospital settings. As a chief quality officer, I also work with improving quality and safety in all health care settings, including ambulatory, nursing homes, home health, and surgical centers. As such, I hope I can bring a broad lens of the medical industry to this position, improving the lives and careers of hospitalists and the patients and families they serve.
As we all know, the demands placed on hospitalists are greater than ever. With shortening length of stay, rising acuity and complexity, increasing administrative burdens, and high emphasis on care transitions, our skills (and our patience) need to rise to these increasing demands. As a member-based society, SHM (and the board of directors) seeks to ensure we are helping hospitalists be the very best they can be, regardless of hospitalist type or practice setting.
The good news is that we are still in high demand. Within the medical industry, there has been an explosive growth in the need for hospitalists, as we now occupy almost every hospital setting in the United States. But as a current commodity, it is imperative that we continue to prove the value we are adding to our patients and their families, the systems in which we work, and the industry as a whole. That is where our board and SHM come into play – to provide the resources you need to improve health care.
These resources come in the form of education and training (live or on demand); leadership and professional development; practice management assistance; advocacy work; mentored quality improvement; networking and project work (through special interest groups, local chapter meetings, and committee work); stimulation of research, new knowledge, and innovation; and promotion of evidence-based practice through our educational resources, publications, and other communications. The purpose of our existence is to provide you what you need to improve your work lives and your patients’ health.
SHM has always fostered a “big-tent” philosophy, so we will continue to explore ways to expand membership beyond “the core” of internal medicine, family medicine, and pediatrics, and reach a better understanding of what our constituents need and how we can add value to their work lives and careers. In addition to expanding membership within our borders, other expansions already include working with international chapters and members, with an “all teach, all learn” attitude to better understand mutually beneficial partnerships with international members. Through all these expansions, we will come closer to truly realizing our mission at SHM, which is to “promote exceptional care for hospitalized patients.”
My humble hope, as it is with any of my leadership positions, is to leave SHM better than I found it. As such, please contact me at any time if you have ideas or suggestions on how we can better help you be successful in improving the care for your patients, your systems, and health care as a whole. I look forward to serving you in this incredible journey and mission.
Dr. Scheurer is chief quality officer and professor of medicine at the Medical University of South Carolina, Charleston. She is the medical editor of the Hospitalist, and president-elect of SHM.
Hoping to expand membership beyond the traditional ‘core’
Hoping to expand membership beyond the traditional ‘core’
It is with great pleasure that I enter my president-elect year for the Society of Hospital Medicine! I am hopeful that this year will allow me time to get to know the organization even better than I already do, and truly understand the needs of our members so I can focus on meeting and exceeding your expectations!
I have been a hospitalist now for 17 years and have practiced in both academic tertiary care and community hospital settings. As a chief quality officer, I also work with improving quality and safety in all health care settings, including ambulatory, nursing homes, home health, and surgical centers. As such, I hope I can bring a broad lens of the medical industry to this position, improving the lives and careers of hospitalists and the patients and families they serve.
As we all know, the demands placed on hospitalists are greater than ever. With shortening length of stay, rising acuity and complexity, increasing administrative burdens, and high emphasis on care transitions, our skills (and our patience) need to rise to these increasing demands. As a member-based society, SHM (and the board of directors) seeks to ensure we are helping hospitalists be the very best they can be, regardless of hospitalist type or practice setting.
The good news is that we are still in high demand. Within the medical industry, there has been an explosive growth in the need for hospitalists, as we now occupy almost every hospital setting in the United States. But as a current commodity, it is imperative that we continue to prove the value we are adding to our patients and their families, the systems in which we work, and the industry as a whole. That is where our board and SHM come into play – to provide the resources you need to improve health care.
These resources come in the form of education and training (live or on demand); leadership and professional development; practice management assistance; advocacy work; mentored quality improvement; networking and project work (through special interest groups, local chapter meetings, and committee work); stimulation of research, new knowledge, and innovation; and promotion of evidence-based practice through our educational resources, publications, and other communications. The purpose of our existence is to provide you what you need to improve your work lives and your patients’ health.
SHM has always fostered a “big-tent” philosophy, so we will continue to explore ways to expand membership beyond “the core” of internal medicine, family medicine, and pediatrics, and reach a better understanding of what our constituents need and how we can add value to their work lives and careers. In addition to expanding membership within our borders, other expansions already include working with international chapters and members, with an “all teach, all learn” attitude to better understand mutually beneficial partnerships with international members. Through all these expansions, we will come closer to truly realizing our mission at SHM, which is to “promote exceptional care for hospitalized patients.”
My humble hope, as it is with any of my leadership positions, is to leave SHM better than I found it. As such, please contact me at any time if you have ideas or suggestions on how we can better help you be successful in improving the care for your patients, your systems, and health care as a whole. I look forward to serving you in this incredible journey and mission.
Dr. Scheurer is chief quality officer and professor of medicine at the Medical University of South Carolina, Charleston. She is the medical editor of the Hospitalist, and president-elect of SHM.
It is with great pleasure that I enter my president-elect year for the Society of Hospital Medicine! I am hopeful that this year will allow me time to get to know the organization even better than I already do, and truly understand the needs of our members so I can focus on meeting and exceeding your expectations!
I have been a hospitalist now for 17 years and have practiced in both academic tertiary care and community hospital settings. As a chief quality officer, I also work with improving quality and safety in all health care settings, including ambulatory, nursing homes, home health, and surgical centers. As such, I hope I can bring a broad lens of the medical industry to this position, improving the lives and careers of hospitalists and the patients and families they serve.
As we all know, the demands placed on hospitalists are greater than ever. With shortening length of stay, rising acuity and complexity, increasing administrative burdens, and high emphasis on care transitions, our skills (and our patience) need to rise to these increasing demands. As a member-based society, SHM (and the board of directors) seeks to ensure we are helping hospitalists be the very best they can be, regardless of hospitalist type or practice setting.
The good news is that we are still in high demand. Within the medical industry, there has been an explosive growth in the need for hospitalists, as we now occupy almost every hospital setting in the United States. But as a current commodity, it is imperative that we continue to prove the value we are adding to our patients and their families, the systems in which we work, and the industry as a whole. That is where our board and SHM come into play – to provide the resources you need to improve health care.
These resources come in the form of education and training (live or on demand); leadership and professional development; practice management assistance; advocacy work; mentored quality improvement; networking and project work (through special interest groups, local chapter meetings, and committee work); stimulation of research, new knowledge, and innovation; and promotion of evidence-based practice through our educational resources, publications, and other communications. The purpose of our existence is to provide you what you need to improve your work lives and your patients’ health.
SHM has always fostered a “big-tent” philosophy, so we will continue to explore ways to expand membership beyond “the core” of internal medicine, family medicine, and pediatrics, and reach a better understanding of what our constituents need and how we can add value to their work lives and careers. In addition to expanding membership within our borders, other expansions already include working with international chapters and members, with an “all teach, all learn” attitude to better understand mutually beneficial partnerships with international members. Through all these expansions, we will come closer to truly realizing our mission at SHM, which is to “promote exceptional care for hospitalized patients.”
My humble hope, as it is with any of my leadership positions, is to leave SHM better than I found it. As such, please contact me at any time if you have ideas or suggestions on how we can better help you be successful in improving the care for your patients, your systems, and health care as a whole. I look forward to serving you in this incredible journey and mission.
Dr. Scheurer is chief quality officer and professor of medicine at the Medical University of South Carolina, Charleston. She is the medical editor of the Hospitalist, and president-elect of SHM.
Short Takes
Pharmacist-led intervention reduced inappropriate medication prescriptions
An outpatient pharmacy-led intervention of notifying prescribing physicians to discontinue inappropriate Beers Criteria medications resulted in a greater discontinuation of inappropriate medications for older adults at 6 months, compared with the control group (43% vs. 12% discontinuation).
Citation: Martin P et al. Effect of a pharmacist-led educational intervention on inappropriate medication prescriptions in older adults: The D-PRESCRIBE randomized clinical trial. JAMA. 2018;320(18):1889-98.
Omadacycline noninferior for community-acquired pneumonia and acute bacterial soft tissue skin infections
Randomized, double-blind, double-dummy trials showed omadacycline is a noninferior alternative to moxifloxacin for the treatment of community-acquired pneumonia and to linezolid for acute bacterial soft-tissue skin infections.
Citation: Nuzyra (omadacycline) [package insert]. Boston, MA: Paratek Pharmaceuticals. 2018.
Lack of evidence to support low-salt diet in adult heart failure patients
Systematic review of multiple databases demonstrated there is limited high-quality evidence to support current guidelines that recommend a low-salt diet to heart failure patients.
Citation: Mahtani KR et al. Reduced salt intake for heart failure: A systematic review. JAMA Int Med. 2018;178(12):1693-700.
Magnesium for rate control in rapid atrial fibrillation
Randomized, controlled trial demonstrated that intravenous magnesium sulfate in combination with atrioventricular (AV) nodal blocking agents resulted in better rate control for atrial fibrillation with rapid ventricular response than did placebo given in combination with AV nodal blocking agents.
Citation: Bouida W et al. Low-dose magnesium sulfate versus high-dose in the early management of rapid atrial fibrillation: Randomized controlled double-blind study (LOMAGHI Study). Acad Emerg Med. 2019 Feb;26(2):183-91.
Low versus intermediate tidal volume strategy on ventilator-free days in ICU patients without ARDS
Randomized, clinical trial of low tidal volume versus intermediate tidal volume strategies in invasively ventilated patients without accute respiratory distress syndrome (ARDS) demonstrated no difference in number of ventilator-free days, ICU length of stay, hospital length of stay, incidence of ventilator-associated adverse events (ARDS, pneumonia, severe atelectasis, pneumothorax), or 28-day mortality.
Citation: Writing Group for the PReVENT Investigators, Simonis FD, Serpa Neto A. Effect of a low vs intermediate tidal volume strategy on ventilator-free days in intensive care unit patients without ARDS: A randomized clinical trial. JAMA. 2018;320(18):1872-80.
Pharmacist-led intervention reduced inappropriate medication prescriptions
An outpatient pharmacy-led intervention of notifying prescribing physicians to discontinue inappropriate Beers Criteria medications resulted in a greater discontinuation of inappropriate medications for older adults at 6 months, compared with the control group (43% vs. 12% discontinuation).
Citation: Martin P et al. Effect of a pharmacist-led educational intervention on inappropriate medication prescriptions in older adults: The D-PRESCRIBE randomized clinical trial. JAMA. 2018;320(18):1889-98.
Omadacycline noninferior for community-acquired pneumonia and acute bacterial soft tissue skin infections
Randomized, double-blind, double-dummy trials showed omadacycline is a noninferior alternative to moxifloxacin for the treatment of community-acquired pneumonia and to linezolid for acute bacterial soft-tissue skin infections.
Citation: Nuzyra (omadacycline) [package insert]. Boston, MA: Paratek Pharmaceuticals. 2018.
Lack of evidence to support low-salt diet in adult heart failure patients
Systematic review of multiple databases demonstrated there is limited high-quality evidence to support current guidelines that recommend a low-salt diet to heart failure patients.
Citation: Mahtani KR et al. Reduced salt intake for heart failure: A systematic review. JAMA Int Med. 2018;178(12):1693-700.
Magnesium for rate control in rapid atrial fibrillation
Randomized, controlled trial demonstrated that intravenous magnesium sulfate in combination with atrioventricular (AV) nodal blocking agents resulted in better rate control for atrial fibrillation with rapid ventricular response than did placebo given in combination with AV nodal blocking agents.
Citation: Bouida W et al. Low-dose magnesium sulfate versus high-dose in the early management of rapid atrial fibrillation: Randomized controlled double-blind study (LOMAGHI Study). Acad Emerg Med. 2019 Feb;26(2):183-91.
Low versus intermediate tidal volume strategy on ventilator-free days in ICU patients without ARDS
Randomized, clinical trial of low tidal volume versus intermediate tidal volume strategies in invasively ventilated patients without accute respiratory distress syndrome (ARDS) demonstrated no difference in number of ventilator-free days, ICU length of stay, hospital length of stay, incidence of ventilator-associated adverse events (ARDS, pneumonia, severe atelectasis, pneumothorax), or 28-day mortality.
Citation: Writing Group for the PReVENT Investigators, Simonis FD, Serpa Neto A. Effect of a low vs intermediate tidal volume strategy on ventilator-free days in intensive care unit patients without ARDS: A randomized clinical trial. JAMA. 2018;320(18):1872-80.
Pharmacist-led intervention reduced inappropriate medication prescriptions
An outpatient pharmacy-led intervention of notifying prescribing physicians to discontinue inappropriate Beers Criteria medications resulted in a greater discontinuation of inappropriate medications for older adults at 6 months, compared with the control group (43% vs. 12% discontinuation).
Citation: Martin P et al. Effect of a pharmacist-led educational intervention on inappropriate medication prescriptions in older adults: The D-PRESCRIBE randomized clinical trial. JAMA. 2018;320(18):1889-98.
Omadacycline noninferior for community-acquired pneumonia and acute bacterial soft tissue skin infections
Randomized, double-blind, double-dummy trials showed omadacycline is a noninferior alternative to moxifloxacin for the treatment of community-acquired pneumonia and to linezolid for acute bacterial soft-tissue skin infections.
Citation: Nuzyra (omadacycline) [package insert]. Boston, MA: Paratek Pharmaceuticals. 2018.
Lack of evidence to support low-salt diet in adult heart failure patients
Systematic review of multiple databases demonstrated there is limited high-quality evidence to support current guidelines that recommend a low-salt diet to heart failure patients.
Citation: Mahtani KR et al. Reduced salt intake for heart failure: A systematic review. JAMA Int Med. 2018;178(12):1693-700.
Magnesium for rate control in rapid atrial fibrillation
Randomized, controlled trial demonstrated that intravenous magnesium sulfate in combination with atrioventricular (AV) nodal blocking agents resulted in better rate control for atrial fibrillation with rapid ventricular response than did placebo given in combination with AV nodal blocking agents.
Citation: Bouida W et al. Low-dose magnesium sulfate versus high-dose in the early management of rapid atrial fibrillation: Randomized controlled double-blind study (LOMAGHI Study). Acad Emerg Med. 2019 Feb;26(2):183-91.
Low versus intermediate tidal volume strategy on ventilator-free days in ICU patients without ARDS
Randomized, clinical trial of low tidal volume versus intermediate tidal volume strategies in invasively ventilated patients without accute respiratory distress syndrome (ARDS) demonstrated no difference in number of ventilator-free days, ICU length of stay, hospital length of stay, incidence of ventilator-associated adverse events (ARDS, pneumonia, severe atelectasis, pneumothorax), or 28-day mortality.
Citation: Writing Group for the PReVENT Investigators, Simonis FD, Serpa Neto A. Effect of a low vs intermediate tidal volume strategy on ventilator-free days in intensive care unit patients without ARDS: A randomized clinical trial. JAMA. 2018;320(18):1872-80.
Drug doses for heart failure could possibly be halved for women
Men and women react differently to common drugs used to treat heart failure with reduced ejection fraction (HFrEF), according to findings from a new European study, and women may be able to safely cut their doses in half and get the same level of relief as that provided by larger doses.
“This study ... brings into question what the true optimal medical therapy is for women versus men,” the study authors, led by Bernadet T. Santema, MD, of the University Medical Center Groningen (the Netherlands), wrote in an article published in the Lancet.
Dr. Santema and colleagues noted that current guidelines for the use of ACE inhibitors or angiotensin-receptor blockers (ARBs) and beta-blockers for men and women with heart failure do not differentiate between the genders, despite findings showing that, “with the same dose, the maximum plasma concentrations of ACE inhibitors, ARBs, and beta-blockers were up to 2.5 times higher in women than in men.”
In addition, the researchers wrote, women are much more likely than men to suffer side effects from medications, and the effects tend to be more severe.
HFrEF accounts for an estimated 50% of the 5.7 million patients with heart failure in the United States (Nat Rev Dis Primers. 2017 Aug 24. doi: 10.1038/nrdp.2017.58; Card Fail Rev. 2017;3[1]:7-11.)
For the new study, researchers launched an ad hoc analysis of the findings of a prospective study of HFrEF patients in 11 European countries (1,308 men and 402 women) who took drugs in the three classes. Patients were receiving suboptimal medication doses at the start of the study, and physicians were encouraged to increase their medication. The median follow-up for the primary endpoint was 21 months.
“In men, the lowest hazards of death or hospitalization for heart failure occurred at 100% of the recommended dose of ACE inhibitors or ARBs and beta-blockers, but women showed about 30% lower risk at only 50% of the recommended doses, with no further decrease in risk at higher dose levels,” the researchers wrote. “These sex differences were still present after adjusting for clinical covariates, including age and body surface area.”
The researchers analyzed an Asian registry (3,539 men, 961 women) as a comparison and found the identical numbers.
“Our study provides evidence supporting the hypothesis that women with HFrEF might have the best outcomes with lower doses of ACE inhibitors or ARBs and beta-blockers than do men, and lower doses than recommended in international guidelines for heart failure,” they wrote. However, they added that it was not likely that sex-specific studies analyzing doses would be performed.
In an accompanying editorial, Heather P. Whitley, PharmD, and Warren D. Smith, PharmD, noted that clinical research has often failed to take gender differences into account. They wrote that the study – the first of its kind – was well executed and raises important questions, but the analysis did not take into account the prevalence of adverse effects or the serum concentrations of the various medications. Although those limitations weaken the findings, the study still offers evidence that gender-based, drug-dose guidelines deserve consideration, wrote Dr. Whitley, of Auburn (Ala.) University, and Dr. Smith, of Baptist Health System, Montgomery, Ala (Lancet. 2019 Aug 22. doi: 10.1016/S0140-6736[19]31812-4).
The study was funded by the European Commission. Several study authors reported various disclosures. Dr. Whitley and Dr. Smith reported no conflicts of interest.
SOURCE: Santema BT et al. Lancet. 2019 Aug 22. doi: 10.1016/S0140-6736(19)31792-1.
Men and women react differently to common drugs used to treat heart failure with reduced ejection fraction (HFrEF), according to findings from a new European study, and women may be able to safely cut their doses in half and get the same level of relief as that provided by larger doses.
“This study ... brings into question what the true optimal medical therapy is for women versus men,” the study authors, led by Bernadet T. Santema, MD, of the University Medical Center Groningen (the Netherlands), wrote in an article published in the Lancet.
Dr. Santema and colleagues noted that current guidelines for the use of ACE inhibitors or angiotensin-receptor blockers (ARBs) and beta-blockers for men and women with heart failure do not differentiate between the genders, despite findings showing that, “with the same dose, the maximum plasma concentrations of ACE inhibitors, ARBs, and beta-blockers were up to 2.5 times higher in women than in men.”
In addition, the researchers wrote, women are much more likely than men to suffer side effects from medications, and the effects tend to be more severe.
HFrEF accounts for an estimated 50% of the 5.7 million patients with heart failure in the United States (Nat Rev Dis Primers. 2017 Aug 24. doi: 10.1038/nrdp.2017.58; Card Fail Rev. 2017;3[1]:7-11.)
For the new study, researchers launched an ad hoc analysis of the findings of a prospective study of HFrEF patients in 11 European countries (1,308 men and 402 women) who took drugs in the three classes. Patients were receiving suboptimal medication doses at the start of the study, and physicians were encouraged to increase their medication. The median follow-up for the primary endpoint was 21 months.
“In men, the lowest hazards of death or hospitalization for heart failure occurred at 100% of the recommended dose of ACE inhibitors or ARBs and beta-blockers, but women showed about 30% lower risk at only 50% of the recommended doses, with no further decrease in risk at higher dose levels,” the researchers wrote. “These sex differences were still present after adjusting for clinical covariates, including age and body surface area.”
The researchers analyzed an Asian registry (3,539 men, 961 women) as a comparison and found the identical numbers.
“Our study provides evidence supporting the hypothesis that women with HFrEF might have the best outcomes with lower doses of ACE inhibitors or ARBs and beta-blockers than do men, and lower doses than recommended in international guidelines for heart failure,” they wrote. However, they added that it was not likely that sex-specific studies analyzing doses would be performed.
In an accompanying editorial, Heather P. Whitley, PharmD, and Warren D. Smith, PharmD, noted that clinical research has often failed to take gender differences into account. They wrote that the study – the first of its kind – was well executed and raises important questions, but the analysis did not take into account the prevalence of adverse effects or the serum concentrations of the various medications. Although those limitations weaken the findings, the study still offers evidence that gender-based, drug-dose guidelines deserve consideration, wrote Dr. Whitley, of Auburn (Ala.) University, and Dr. Smith, of Baptist Health System, Montgomery, Ala (Lancet. 2019 Aug 22. doi: 10.1016/S0140-6736[19]31812-4).
The study was funded by the European Commission. Several study authors reported various disclosures. Dr. Whitley and Dr. Smith reported no conflicts of interest.
SOURCE: Santema BT et al. Lancet. 2019 Aug 22. doi: 10.1016/S0140-6736(19)31792-1.
Men and women react differently to common drugs used to treat heart failure with reduced ejection fraction (HFrEF), according to findings from a new European study, and women may be able to safely cut their doses in half and get the same level of relief as that provided by larger doses.
“This study ... brings into question what the true optimal medical therapy is for women versus men,” the study authors, led by Bernadet T. Santema, MD, of the University Medical Center Groningen (the Netherlands), wrote in an article published in the Lancet.
Dr. Santema and colleagues noted that current guidelines for the use of ACE inhibitors or angiotensin-receptor blockers (ARBs) and beta-blockers for men and women with heart failure do not differentiate between the genders, despite findings showing that, “with the same dose, the maximum plasma concentrations of ACE inhibitors, ARBs, and beta-blockers were up to 2.5 times higher in women than in men.”
In addition, the researchers wrote, women are much more likely than men to suffer side effects from medications, and the effects tend to be more severe.
HFrEF accounts for an estimated 50% of the 5.7 million patients with heart failure in the United States (Nat Rev Dis Primers. 2017 Aug 24. doi: 10.1038/nrdp.2017.58; Card Fail Rev. 2017;3[1]:7-11.)
For the new study, researchers launched an ad hoc analysis of the findings of a prospective study of HFrEF patients in 11 European countries (1,308 men and 402 women) who took drugs in the three classes. Patients were receiving suboptimal medication doses at the start of the study, and physicians were encouraged to increase their medication. The median follow-up for the primary endpoint was 21 months.
“In men, the lowest hazards of death or hospitalization for heart failure occurred at 100% of the recommended dose of ACE inhibitors or ARBs and beta-blockers, but women showed about 30% lower risk at only 50% of the recommended doses, with no further decrease in risk at higher dose levels,” the researchers wrote. “These sex differences were still present after adjusting for clinical covariates, including age and body surface area.”
The researchers analyzed an Asian registry (3,539 men, 961 women) as a comparison and found the identical numbers.
“Our study provides evidence supporting the hypothesis that women with HFrEF might have the best outcomes with lower doses of ACE inhibitors or ARBs and beta-blockers than do men, and lower doses than recommended in international guidelines for heart failure,” they wrote. However, they added that it was not likely that sex-specific studies analyzing doses would be performed.
In an accompanying editorial, Heather P. Whitley, PharmD, and Warren D. Smith, PharmD, noted that clinical research has often failed to take gender differences into account. They wrote that the study – the first of its kind – was well executed and raises important questions, but the analysis did not take into account the prevalence of adverse effects or the serum concentrations of the various medications. Although those limitations weaken the findings, the study still offers evidence that gender-based, drug-dose guidelines deserve consideration, wrote Dr. Whitley, of Auburn (Ala.) University, and Dr. Smith, of Baptist Health System, Montgomery, Ala (Lancet. 2019 Aug 22. doi: 10.1016/S0140-6736[19]31812-4).
The study was funded by the European Commission. Several study authors reported various disclosures. Dr. Whitley and Dr. Smith reported no conflicts of interest.
SOURCE: Santema BT et al. Lancet. 2019 Aug 22. doi: 10.1016/S0140-6736(19)31792-1.
FROM THE LANCET
Consider triple therapy for the management of COPD
Background: The Global Initiative for Obstructive Lung Disease (GOLD) recommends triple therapy with inhaled corticosteroids, long-acting beta2-adrenoceptor agonists (LABA), and long-acting muscarinic receptor antagonists (LAMA) for patients with severe COPD who have frequent exacerbations despite treatment with a LABA and LAMA. Triple therapy has been shown to improve forced expiratory volume in 1 second (FEV1), but its effect on preventing exacerbations has not been well documented in previous meta-analyses.
Study design: Meta-analysis.
Setting: Studies published on PubMed, Embase, Cochrane Library website, Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov databases.
Synopsis: 21 randomized, controlled trials of triple therapy in stable cases of moderate to very severe COPD were included in this meta-analysis. Triple therapy was associated with a significantly greater reduction in the rate of COPD exacerbations, compared with dual therapy of LAMA and LABA (rate ratio, 0.78; 95% confidence interval, 0.70-0.88), inhaled corticosteroid and LABA (rate ratio, 0.77; 95% CI, 0.66-0.91), or LAMA monotherapy (rate ratio, 0.71; 95% CI, 0.60-0.85). Triple therapy was also associated with greater improvement in FEV1.
There was a significantly higher incidence of pneumonia in patients using triple therapy, compared with those using dual therapy (LAMA and LABA), and there also was a trend toward increased pneumonia incidence with triple therapy, compared with LAMA monotherapy. Triple therapy was not shown to improve survival; however, most trials lasted less than 6 months, which limits their analysis of survival outcomes.
Bottom line: In patients with advanced COPD, triple therapy is associated with lower rates of COPD exacerbations and improved lung function, compared with dual therapy or monotherapy.
Citation: Zheng Y et al. Triple therapy in the management of chronic obstructive pulmonary disease: Systemic review and meta-analysis. BMJ. 2018;363:k4388.
Dr. Chace is an associate physician in the division of hospital medicine at the University of California, San Diego.
Background: The Global Initiative for Obstructive Lung Disease (GOLD) recommends triple therapy with inhaled corticosteroids, long-acting beta2-adrenoceptor agonists (LABA), and long-acting muscarinic receptor antagonists (LAMA) for patients with severe COPD who have frequent exacerbations despite treatment with a LABA and LAMA. Triple therapy has been shown to improve forced expiratory volume in 1 second (FEV1), but its effect on preventing exacerbations has not been well documented in previous meta-analyses.
Study design: Meta-analysis.
Setting: Studies published on PubMed, Embase, Cochrane Library website, Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov databases.
Synopsis: 21 randomized, controlled trials of triple therapy in stable cases of moderate to very severe COPD were included in this meta-analysis. Triple therapy was associated with a significantly greater reduction in the rate of COPD exacerbations, compared with dual therapy of LAMA and LABA (rate ratio, 0.78; 95% confidence interval, 0.70-0.88), inhaled corticosteroid and LABA (rate ratio, 0.77; 95% CI, 0.66-0.91), or LAMA monotherapy (rate ratio, 0.71; 95% CI, 0.60-0.85). Triple therapy was also associated with greater improvement in FEV1.
There was a significantly higher incidence of pneumonia in patients using triple therapy, compared with those using dual therapy (LAMA and LABA), and there also was a trend toward increased pneumonia incidence with triple therapy, compared with LAMA monotherapy. Triple therapy was not shown to improve survival; however, most trials lasted less than 6 months, which limits their analysis of survival outcomes.
Bottom line: In patients with advanced COPD, triple therapy is associated with lower rates of COPD exacerbations and improved lung function, compared with dual therapy or monotherapy.
Citation: Zheng Y et al. Triple therapy in the management of chronic obstructive pulmonary disease: Systemic review and meta-analysis. BMJ. 2018;363:k4388.
Dr. Chace is an associate physician in the division of hospital medicine at the University of California, San Diego.
Background: The Global Initiative for Obstructive Lung Disease (GOLD) recommends triple therapy with inhaled corticosteroids, long-acting beta2-adrenoceptor agonists (LABA), and long-acting muscarinic receptor antagonists (LAMA) for patients with severe COPD who have frequent exacerbations despite treatment with a LABA and LAMA. Triple therapy has been shown to improve forced expiratory volume in 1 second (FEV1), but its effect on preventing exacerbations has not been well documented in previous meta-analyses.
Study design: Meta-analysis.
Setting: Studies published on PubMed, Embase, Cochrane Library website, Cochrane Central Register of Controlled Trials (CENTRAL), and ClinicalTrials.gov databases.
Synopsis: 21 randomized, controlled trials of triple therapy in stable cases of moderate to very severe COPD were included in this meta-analysis. Triple therapy was associated with a significantly greater reduction in the rate of COPD exacerbations, compared with dual therapy of LAMA and LABA (rate ratio, 0.78; 95% confidence interval, 0.70-0.88), inhaled corticosteroid and LABA (rate ratio, 0.77; 95% CI, 0.66-0.91), or LAMA monotherapy (rate ratio, 0.71; 95% CI, 0.60-0.85). Triple therapy was also associated with greater improvement in FEV1.
There was a significantly higher incidence of pneumonia in patients using triple therapy, compared with those using dual therapy (LAMA and LABA), and there also was a trend toward increased pneumonia incidence with triple therapy, compared with LAMA monotherapy. Triple therapy was not shown to improve survival; however, most trials lasted less than 6 months, which limits their analysis of survival outcomes.
Bottom line: In patients with advanced COPD, triple therapy is associated with lower rates of COPD exacerbations and improved lung function, compared with dual therapy or monotherapy.
Citation: Zheng Y et al. Triple therapy in the management of chronic obstructive pulmonary disease: Systemic review and meta-analysis. BMJ. 2018;363:k4388.
Dr. Chace is an associate physician in the division of hospital medicine at the University of California, San Diego.
Older IBD patients are most at risk of postdischarge VTE
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Readmission for VTE in patients with inflammatory bowel diseases most often occurs within 60 days of discharge.
Major finding: The highest readmission risk was in patients between the ages of 66 and 80 (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01).
Study details: A retrospective cohort study of 1,160 IBD patients who had VTE readmissions via 2010-2014 data from the Nationwide Readmissions Database.
Disclosures: The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
Source: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.