User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
ID experts dole out practical advice to help with mask confusion
The Centers for Disease Control and Prevention’s latest guidance on what fully vaccinated people can do safely – including not socially distancing and not wearing a mask indoors or outdoors unless other regulations require it – has been widely misinterpreted and caused confusion, two infectious disease experts said at a briefing on May 20 hosted by the Infectious Diseases Society of America (IDSA).
The CDC did not ‘’lift” the mask mandate, but rather supplied guidance for those who are fully vaccinated. However, many questions and gray areas remain, and the experts addressed those. ‘’The CDC guidance is really directed at people who are fully vaccinated and who we know are likely to have a really solid response to the vaccine,” said Jeanne Marrazzo, MD, MPH, director of infectious diseases at the University of Alabama at Birmingham and an IDSA board member.
That message was largely lost, said Dr. Marrazzo and Jeffrey Duchin, MD, health officer of public health for Seattle and King County, Washington, and also an IDSA board member. Dr. Duchin said many people mistakenly regarded the new guidance as a message that the pandemic is over.
Among their practical tips on how to interpret the guidance:
To mask or not?
To make the decision, people need to think about not only the numbers of vaccinated versus unvaccinated individuals in their community but the local rates of disease, the experts said. And they need to know that the CDC guidance doesn’t apply if regulations by federal or state authorities or businesses and workplace are in conflict.
Deciding on mask use sometimes depends on where you are going. What about going into grocery stores or large bin stores without a mask? “If you are fully vaccinated and have no other conditions that compromise your immune system, and the rates of COVID are relatively low where you live, and the vaccination rates are high, I would be 100% fine” without a mask, Dr. Marrazzo said. But it’s important to think of all these factors in calculating your risk.
“I’m still wearing a mask when I go anywhere in public,” she said, citing vaccination rates that have not yet reached 50% in her area.
If that rate reached 80%, the typical percentage talked about for herd immunity, and new cases were low, Dr. Marrazzo said she might shed the mask.
The CDC also continues to recommend masks on mass transit for all.
One population that also must be considered, and who must evaluate their risk, even if vaccinated, are the immunocompromised, Dr. Marrazzo said. While people think of the immunocompromised as those with HIV or organ transplants, the numbers are actually much larger.
“A study a couple of years ago indicated up to 3% of Americans may actually have been told by their physician they have some of level of being immunocompromised,” she said. Among the examples are those who are on dialysis, on chemotherapy, or those taking any of the medications that modify the immune system.
“Millions of people fit this bill, and we have [very] little data on whether the vaccine works in them. We think it does,” Dr. Marrazzo said.
Still, she said, it’s a reason for these people to be cautious. For some other vaccines, the dose is modified for those who are immunocompromised. What’s not known yet is whether additional doses of the COVID vaccines might boost protection for those who are immunocompromised.
Many people, even after vaccination, may choose to keep wearing a mask especially in indoor, crowded settings, Dr. Duchin said. “We need to expect, accept, and respect continued mask wearing by anyone at any time.”
In most outdoor settings, he said, “I think masks are probably not necessary, vaccinated or not, regardless of age.” One exception: close face-to-face contact, such as in certain sports.
How to protect toddlers and infants
With masks not practical or recommended for infants and toddlers under 2 years old, Dr. Marrazzo said adults should remember that ‘’those very little kids don’t do poorly at all [even if infected], although there is not a ton of data.”
Adults should still treat young children as vulnerable, especially newborns. Adults not yet vaccinated should wear a mask when around them, she said.
J & J vaccine recipients
With less ‘’real world” data on the Johnson & Johnson vaccine, should those who got it think of themselves in a different risk group than those who got Moderna or Pfizer and adjust their behavior accordingly?
“The J&J vaccine, based on everything we know, does provide a great deal of protection,” Dr. Marrazzo said. ‘’We don’t know as much about prevention of transmission in the asymptomatic cases in the J&J.”
Most of that data, she said, is from the mRNA vaccines Pfizer and Moderna. “I think it’s an important area to study and learn about.” But all three vaccines, overall, provide a high level of protection, she said.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention’s latest guidance on what fully vaccinated people can do safely – including not socially distancing and not wearing a mask indoors or outdoors unless other regulations require it – has been widely misinterpreted and caused confusion, two infectious disease experts said at a briefing on May 20 hosted by the Infectious Diseases Society of America (IDSA).
The CDC did not ‘’lift” the mask mandate, but rather supplied guidance for those who are fully vaccinated. However, many questions and gray areas remain, and the experts addressed those. ‘’The CDC guidance is really directed at people who are fully vaccinated and who we know are likely to have a really solid response to the vaccine,” said Jeanne Marrazzo, MD, MPH, director of infectious diseases at the University of Alabama at Birmingham and an IDSA board member.
That message was largely lost, said Dr. Marrazzo and Jeffrey Duchin, MD, health officer of public health for Seattle and King County, Washington, and also an IDSA board member. Dr. Duchin said many people mistakenly regarded the new guidance as a message that the pandemic is over.
Among their practical tips on how to interpret the guidance:
To mask or not?
To make the decision, people need to think about not only the numbers of vaccinated versus unvaccinated individuals in their community but the local rates of disease, the experts said. And they need to know that the CDC guidance doesn’t apply if regulations by federal or state authorities or businesses and workplace are in conflict.
Deciding on mask use sometimes depends on where you are going. What about going into grocery stores or large bin stores without a mask? “If you are fully vaccinated and have no other conditions that compromise your immune system, and the rates of COVID are relatively low where you live, and the vaccination rates are high, I would be 100% fine” without a mask, Dr. Marrazzo said. But it’s important to think of all these factors in calculating your risk.
“I’m still wearing a mask when I go anywhere in public,” she said, citing vaccination rates that have not yet reached 50% in her area.
If that rate reached 80%, the typical percentage talked about for herd immunity, and new cases were low, Dr. Marrazzo said she might shed the mask.
The CDC also continues to recommend masks on mass transit for all.
One population that also must be considered, and who must evaluate their risk, even if vaccinated, are the immunocompromised, Dr. Marrazzo said. While people think of the immunocompromised as those with HIV or organ transplants, the numbers are actually much larger.
“A study a couple of years ago indicated up to 3% of Americans may actually have been told by their physician they have some of level of being immunocompromised,” she said. Among the examples are those who are on dialysis, on chemotherapy, or those taking any of the medications that modify the immune system.
“Millions of people fit this bill, and we have [very] little data on whether the vaccine works in them. We think it does,” Dr. Marrazzo said.
Still, she said, it’s a reason for these people to be cautious. For some other vaccines, the dose is modified for those who are immunocompromised. What’s not known yet is whether additional doses of the COVID vaccines might boost protection for those who are immunocompromised.
Many people, even after vaccination, may choose to keep wearing a mask especially in indoor, crowded settings, Dr. Duchin said. “We need to expect, accept, and respect continued mask wearing by anyone at any time.”
In most outdoor settings, he said, “I think masks are probably not necessary, vaccinated or not, regardless of age.” One exception: close face-to-face contact, such as in certain sports.
How to protect toddlers and infants
With masks not practical or recommended for infants and toddlers under 2 years old, Dr. Marrazzo said adults should remember that ‘’those very little kids don’t do poorly at all [even if infected], although there is not a ton of data.”
Adults should still treat young children as vulnerable, especially newborns. Adults not yet vaccinated should wear a mask when around them, she said.
J & J vaccine recipients
With less ‘’real world” data on the Johnson & Johnson vaccine, should those who got it think of themselves in a different risk group than those who got Moderna or Pfizer and adjust their behavior accordingly?
“The J&J vaccine, based on everything we know, does provide a great deal of protection,” Dr. Marrazzo said. ‘’We don’t know as much about prevention of transmission in the asymptomatic cases in the J&J.”
Most of that data, she said, is from the mRNA vaccines Pfizer and Moderna. “I think it’s an important area to study and learn about.” But all three vaccines, overall, provide a high level of protection, she said.
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention’s latest guidance on what fully vaccinated people can do safely – including not socially distancing and not wearing a mask indoors or outdoors unless other regulations require it – has been widely misinterpreted and caused confusion, two infectious disease experts said at a briefing on May 20 hosted by the Infectious Diseases Society of America (IDSA).
The CDC did not ‘’lift” the mask mandate, but rather supplied guidance for those who are fully vaccinated. However, many questions and gray areas remain, and the experts addressed those. ‘’The CDC guidance is really directed at people who are fully vaccinated and who we know are likely to have a really solid response to the vaccine,” said Jeanne Marrazzo, MD, MPH, director of infectious diseases at the University of Alabama at Birmingham and an IDSA board member.
That message was largely lost, said Dr. Marrazzo and Jeffrey Duchin, MD, health officer of public health for Seattle and King County, Washington, and also an IDSA board member. Dr. Duchin said many people mistakenly regarded the new guidance as a message that the pandemic is over.
Among their practical tips on how to interpret the guidance:
To mask or not?
To make the decision, people need to think about not only the numbers of vaccinated versus unvaccinated individuals in their community but the local rates of disease, the experts said. And they need to know that the CDC guidance doesn’t apply if regulations by federal or state authorities or businesses and workplace are in conflict.
Deciding on mask use sometimes depends on where you are going. What about going into grocery stores or large bin stores without a mask? “If you are fully vaccinated and have no other conditions that compromise your immune system, and the rates of COVID are relatively low where you live, and the vaccination rates are high, I would be 100% fine” without a mask, Dr. Marrazzo said. But it’s important to think of all these factors in calculating your risk.
“I’m still wearing a mask when I go anywhere in public,” she said, citing vaccination rates that have not yet reached 50% in her area.
If that rate reached 80%, the typical percentage talked about for herd immunity, and new cases were low, Dr. Marrazzo said she might shed the mask.
The CDC also continues to recommend masks on mass transit for all.
One population that also must be considered, and who must evaluate their risk, even if vaccinated, are the immunocompromised, Dr. Marrazzo said. While people think of the immunocompromised as those with HIV or organ transplants, the numbers are actually much larger.
“A study a couple of years ago indicated up to 3% of Americans may actually have been told by their physician they have some of level of being immunocompromised,” she said. Among the examples are those who are on dialysis, on chemotherapy, or those taking any of the medications that modify the immune system.
“Millions of people fit this bill, and we have [very] little data on whether the vaccine works in them. We think it does,” Dr. Marrazzo said.
Still, she said, it’s a reason for these people to be cautious. For some other vaccines, the dose is modified for those who are immunocompromised. What’s not known yet is whether additional doses of the COVID vaccines might boost protection for those who are immunocompromised.
Many people, even after vaccination, may choose to keep wearing a mask especially in indoor, crowded settings, Dr. Duchin said. “We need to expect, accept, and respect continued mask wearing by anyone at any time.”
In most outdoor settings, he said, “I think masks are probably not necessary, vaccinated or not, regardless of age.” One exception: close face-to-face contact, such as in certain sports.
How to protect toddlers and infants
With masks not practical or recommended for infants and toddlers under 2 years old, Dr. Marrazzo said adults should remember that ‘’those very little kids don’t do poorly at all [even if infected], although there is not a ton of data.”
Adults should still treat young children as vulnerable, especially newborns. Adults not yet vaccinated should wear a mask when around them, she said.
J & J vaccine recipients
With less ‘’real world” data on the Johnson & Johnson vaccine, should those who got it think of themselves in a different risk group than those who got Moderna or Pfizer and adjust their behavior accordingly?
“The J&J vaccine, based on everything we know, does provide a great deal of protection,” Dr. Marrazzo said. ‘’We don’t know as much about prevention of transmission in the asymptomatic cases in the J&J.”
Most of that data, she said, is from the mRNA vaccines Pfizer and Moderna. “I think it’s an important area to study and learn about.” But all three vaccines, overall, provide a high level of protection, she said.
A version of this article first appeared on Medscape.com.
Study identifies strong association between use of rotavirus vaccines, 60% reduction in infection
Two widely used rotavirus vaccines performed comparably in a meta-analysis, reducing risk of rotavirus gastroenteritis (RVGE) by more than 60% in young children. While the findings evidence a high protection level and low-risk safety profile, investigators of the study called for additional head-to-head comparisons to assess risks and benefits.
RVGE, which accounts for 28.8% of all deaths from diarrhea worldwide, is the leading cause of diarrhea in children under age 5. More than 100 countries include rotavirus vaccines in their immunization programs. Among six types of vaccines currently in use, two live-attenuated oral vaccines: the two-dose monovalent Rotarix (RV1) and three-dose pentavalent RotaTeq (RV5]) are in use worldwide.
Not much is known about their interchangeability, although a previous meta-analysis reported similarities in effectiveness of Rotarix (83%), RotaTeq (85%), and Rotarix and RotaTeq mixed series (86%) in low-mortality countries. RVGE morbidity and mortality have declined since the introduction of these vaccines, but concerns persist about their safety, Zi-Wei Sun, MSc, of Nanjing (China) Medical University and colleagues wrote in JAMA Pediatrics.
Their systematic review and meta-analysis of randomized clinical trials, case-control, and cohort studies compared benefit, risk, and immunogenicity of these vaccines and their effectiveness in reducing RVGE. Combing through databases Embase, PubMed, the Cochrane Library, and Web of Science using search terms “rotavirus” and “vaccine,” they chose 121 randomized clinical trials and cohort and case-control studies that included more than 100 children younger than 5 years. Thirty-eight of the randomized clinical trials had related data that examined the vaccines’ protection against RVGE hospitalization, study coauthor Hemant Goyal, MD, FACP, explained in an interview.
All of the studies reported on the safety and effectiveness or immunogenicity of rotavirus vaccines. The investigators used a random-effects model to calculate relative risks, odds ratios, risk differences, and 95% confidence intervals. They also stratified studies by economic development of countries, given that vaccine efficacy is often higher in middle- and high-income countries, compared with low-income countries. An adjusted indirect treatment comparison evaluated differences in vaccine protection among different subgroups, adopting P < .05 as the level of statistical significance.
Primary outcomes included RVGE, severe RVGE, and RVGE hospitalization and safety-associated outcomes such as serious adverse events, intussusception, and mortality.
Rotarix and RotaTeq reduced RVGE in children younger than 5 years by 68.4% and 63.6%, respectively. Dr. Goyal and colleagues confirmed these results in case-control studies (65.3% and 72.8%, respectively). Both vaccines significantly reduced RVGE and RVGE hospitalization risk and demonstrated higher protection against severe RVGE. In adjusted indirect comparisons, the two vaccines showed no significant differences in protection. They also found a positive correlation between immunogenicity and vaccine protection.
“RotaTeq seems to show lower protection in low-income countries, compared with Rotarix, but these estimates should be interpreted with caution as there was only one study for low-income countries and indirect comparison," said Dr. Goyal, a second-year gastroenterology fellow at the Wright Center for Graduate Medical Education, Scranton, Penn.
None of the vaccines demonstrated risk of serious adverse events. However, an Australian study in 2013 did report a small increased risk of intussusception after RV1 and RV5 vaccination. “Therefore, continuous surveillance of the benefits and adverse effects of rotavirus vaccines is required after vaccination,” the investigators noted.
Analyzing newer, less widely distributed vaccines, Rotavac, Rotasiil, and Lanzhou lamb rotavirus vaccine also showed moderate effectiveness in reducing RVGE risk.
Immunity wanes over time
Protection against rotavirus diseases seems to wane over time after vaccination. “Although our results indicated that rotavirus vaccines can provide substantial protection against RVGE during the first 2 years of life, more studies following up the vaccine efficacy for more than 2 years are required,” the investigators recommended.
Declining vaccine-induced antibodies, RVGE-acquired protection from the vaccine’s indirect effects, or exposure to unvaccinated populations may explain gradual loss of immunity.
Monitoring of rotavirus strains following vaccination should take place “to avoid population-based selection of so-called escape strains, especially fully heterotypic strains and new strains, because of the long-term pressure of vaccine immunity,” they recommended.
The findings emphasize the importance of introducing vaccines worldwide to reduce infection, summarized Dr. Goyal and colleagues. Given how challenging it is to treat the wide varieties of rotavirus, “It encouraging that RV1 and RV5 work well against heterotypic strains,” they added. Similar performance between Rotarix and RotaTeq also makes it easier for clinicians to choose a vaccine.
Increasing the availability and efficacy of these vaccines in low-income countries with high mortality rates is a high priority,
David I. Bernstein, MD, MA, wrote in a related editorial: “A clear gradient in vaccine protections was noted by country income level in the analysis presented, and much effort has been spent to understand this discrepancy.”
Overall, the study confirmed the efficacy of these two vaccines and their equivalence, noted Dr. Bernstein.
The study’s literature search process had some limitations. “Especially in stratified analyses, sparse data in some subgroups limit generalizability. ... The most accurate method, head-to-head comparisons, to evaluate the comparative efficacy of different vaccines is required in further studies,” the study investigators wrote.
Such studies would directly compare Rotarix and RotaTeq from multiple perspectives: efficacy, cost-effectiveness, strain-specific protection, the duration of protection, safety, and immunogenicity, said Dr. Goyal.
*This story was updated on May 24, 2021.
Two widely used rotavirus vaccines performed comparably in a meta-analysis, reducing risk of rotavirus gastroenteritis (RVGE) by more than 60% in young children. While the findings evidence a high protection level and low-risk safety profile, investigators of the study called for additional head-to-head comparisons to assess risks and benefits.
RVGE, which accounts for 28.8% of all deaths from diarrhea worldwide, is the leading cause of diarrhea in children under age 5. More than 100 countries include rotavirus vaccines in their immunization programs. Among six types of vaccines currently in use, two live-attenuated oral vaccines: the two-dose monovalent Rotarix (RV1) and three-dose pentavalent RotaTeq (RV5]) are in use worldwide.
Not much is known about their interchangeability, although a previous meta-analysis reported similarities in effectiveness of Rotarix (83%), RotaTeq (85%), and Rotarix and RotaTeq mixed series (86%) in low-mortality countries. RVGE morbidity and mortality have declined since the introduction of these vaccines, but concerns persist about their safety, Zi-Wei Sun, MSc, of Nanjing (China) Medical University and colleagues wrote in JAMA Pediatrics.
Their systematic review and meta-analysis of randomized clinical trials, case-control, and cohort studies compared benefit, risk, and immunogenicity of these vaccines and their effectiveness in reducing RVGE. Combing through databases Embase, PubMed, the Cochrane Library, and Web of Science using search terms “rotavirus” and “vaccine,” they chose 121 randomized clinical trials and cohort and case-control studies that included more than 100 children younger than 5 years. Thirty-eight of the randomized clinical trials had related data that examined the vaccines’ protection against RVGE hospitalization, study coauthor Hemant Goyal, MD, FACP, explained in an interview.
All of the studies reported on the safety and effectiveness or immunogenicity of rotavirus vaccines. The investigators used a random-effects model to calculate relative risks, odds ratios, risk differences, and 95% confidence intervals. They also stratified studies by economic development of countries, given that vaccine efficacy is often higher in middle- and high-income countries, compared with low-income countries. An adjusted indirect treatment comparison evaluated differences in vaccine protection among different subgroups, adopting P < .05 as the level of statistical significance.
Primary outcomes included RVGE, severe RVGE, and RVGE hospitalization and safety-associated outcomes such as serious adverse events, intussusception, and mortality.
Rotarix and RotaTeq reduced RVGE in children younger than 5 years by 68.4% and 63.6%, respectively. Dr. Goyal and colleagues confirmed these results in case-control studies (65.3% and 72.8%, respectively). Both vaccines significantly reduced RVGE and RVGE hospitalization risk and demonstrated higher protection against severe RVGE. In adjusted indirect comparisons, the two vaccines showed no significant differences in protection. They also found a positive correlation between immunogenicity and vaccine protection.
“RotaTeq seems to show lower protection in low-income countries, compared with Rotarix, but these estimates should be interpreted with caution as there was only one study for low-income countries and indirect comparison," said Dr. Goyal, a second-year gastroenterology fellow at the Wright Center for Graduate Medical Education, Scranton, Penn.
None of the vaccines demonstrated risk of serious adverse events. However, an Australian study in 2013 did report a small increased risk of intussusception after RV1 and RV5 vaccination. “Therefore, continuous surveillance of the benefits and adverse effects of rotavirus vaccines is required after vaccination,” the investigators noted.
Analyzing newer, less widely distributed vaccines, Rotavac, Rotasiil, and Lanzhou lamb rotavirus vaccine also showed moderate effectiveness in reducing RVGE risk.
Immunity wanes over time
Protection against rotavirus diseases seems to wane over time after vaccination. “Although our results indicated that rotavirus vaccines can provide substantial protection against RVGE during the first 2 years of life, more studies following up the vaccine efficacy for more than 2 years are required,” the investigators recommended.
Declining vaccine-induced antibodies, RVGE-acquired protection from the vaccine’s indirect effects, or exposure to unvaccinated populations may explain gradual loss of immunity.
Monitoring of rotavirus strains following vaccination should take place “to avoid population-based selection of so-called escape strains, especially fully heterotypic strains and new strains, because of the long-term pressure of vaccine immunity,” they recommended.
The findings emphasize the importance of introducing vaccines worldwide to reduce infection, summarized Dr. Goyal and colleagues. Given how challenging it is to treat the wide varieties of rotavirus, “It encouraging that RV1 and RV5 work well against heterotypic strains,” they added. Similar performance between Rotarix and RotaTeq also makes it easier for clinicians to choose a vaccine.
Increasing the availability and efficacy of these vaccines in low-income countries with high mortality rates is a high priority,
David I. Bernstein, MD, MA, wrote in a related editorial: “A clear gradient in vaccine protections was noted by country income level in the analysis presented, and much effort has been spent to understand this discrepancy.”
Overall, the study confirmed the efficacy of these two vaccines and their equivalence, noted Dr. Bernstein.
The study’s literature search process had some limitations. “Especially in stratified analyses, sparse data in some subgroups limit generalizability. ... The most accurate method, head-to-head comparisons, to evaluate the comparative efficacy of different vaccines is required in further studies,” the study investigators wrote.
Such studies would directly compare Rotarix and RotaTeq from multiple perspectives: efficacy, cost-effectiveness, strain-specific protection, the duration of protection, safety, and immunogenicity, said Dr. Goyal.
*This story was updated on May 24, 2021.
Two widely used rotavirus vaccines performed comparably in a meta-analysis, reducing risk of rotavirus gastroenteritis (RVGE) by more than 60% in young children. While the findings evidence a high protection level and low-risk safety profile, investigators of the study called for additional head-to-head comparisons to assess risks and benefits.
RVGE, which accounts for 28.8% of all deaths from diarrhea worldwide, is the leading cause of diarrhea in children under age 5. More than 100 countries include rotavirus vaccines in their immunization programs. Among six types of vaccines currently in use, two live-attenuated oral vaccines: the two-dose monovalent Rotarix (RV1) and three-dose pentavalent RotaTeq (RV5]) are in use worldwide.
Not much is known about their interchangeability, although a previous meta-analysis reported similarities in effectiveness of Rotarix (83%), RotaTeq (85%), and Rotarix and RotaTeq mixed series (86%) in low-mortality countries. RVGE morbidity and mortality have declined since the introduction of these vaccines, but concerns persist about their safety, Zi-Wei Sun, MSc, of Nanjing (China) Medical University and colleagues wrote in JAMA Pediatrics.
Their systematic review and meta-analysis of randomized clinical trials, case-control, and cohort studies compared benefit, risk, and immunogenicity of these vaccines and their effectiveness in reducing RVGE. Combing through databases Embase, PubMed, the Cochrane Library, and Web of Science using search terms “rotavirus” and “vaccine,” they chose 121 randomized clinical trials and cohort and case-control studies that included more than 100 children younger than 5 years. Thirty-eight of the randomized clinical trials had related data that examined the vaccines’ protection against RVGE hospitalization, study coauthor Hemant Goyal, MD, FACP, explained in an interview.
All of the studies reported on the safety and effectiveness or immunogenicity of rotavirus vaccines. The investigators used a random-effects model to calculate relative risks, odds ratios, risk differences, and 95% confidence intervals. They also stratified studies by economic development of countries, given that vaccine efficacy is often higher in middle- and high-income countries, compared with low-income countries. An adjusted indirect treatment comparison evaluated differences in vaccine protection among different subgroups, adopting P < .05 as the level of statistical significance.
Primary outcomes included RVGE, severe RVGE, and RVGE hospitalization and safety-associated outcomes such as serious adverse events, intussusception, and mortality.
Rotarix and RotaTeq reduced RVGE in children younger than 5 years by 68.4% and 63.6%, respectively. Dr. Goyal and colleagues confirmed these results in case-control studies (65.3% and 72.8%, respectively). Both vaccines significantly reduced RVGE and RVGE hospitalization risk and demonstrated higher protection against severe RVGE. In adjusted indirect comparisons, the two vaccines showed no significant differences in protection. They also found a positive correlation between immunogenicity and vaccine protection.
“RotaTeq seems to show lower protection in low-income countries, compared with Rotarix, but these estimates should be interpreted with caution as there was only one study for low-income countries and indirect comparison," said Dr. Goyal, a second-year gastroenterology fellow at the Wright Center for Graduate Medical Education, Scranton, Penn.
None of the vaccines demonstrated risk of serious adverse events. However, an Australian study in 2013 did report a small increased risk of intussusception after RV1 and RV5 vaccination. “Therefore, continuous surveillance of the benefits and adverse effects of rotavirus vaccines is required after vaccination,” the investigators noted.
Analyzing newer, less widely distributed vaccines, Rotavac, Rotasiil, and Lanzhou lamb rotavirus vaccine also showed moderate effectiveness in reducing RVGE risk.
Immunity wanes over time
Protection against rotavirus diseases seems to wane over time after vaccination. “Although our results indicated that rotavirus vaccines can provide substantial protection against RVGE during the first 2 years of life, more studies following up the vaccine efficacy for more than 2 years are required,” the investigators recommended.
Declining vaccine-induced antibodies, RVGE-acquired protection from the vaccine’s indirect effects, or exposure to unvaccinated populations may explain gradual loss of immunity.
Monitoring of rotavirus strains following vaccination should take place “to avoid population-based selection of so-called escape strains, especially fully heterotypic strains and new strains, because of the long-term pressure of vaccine immunity,” they recommended.
The findings emphasize the importance of introducing vaccines worldwide to reduce infection, summarized Dr. Goyal and colleagues. Given how challenging it is to treat the wide varieties of rotavirus, “It encouraging that RV1 and RV5 work well against heterotypic strains,” they added. Similar performance between Rotarix and RotaTeq also makes it easier for clinicians to choose a vaccine.
Increasing the availability and efficacy of these vaccines in low-income countries with high mortality rates is a high priority,
David I. Bernstein, MD, MA, wrote in a related editorial: “A clear gradient in vaccine protections was noted by country income level in the analysis presented, and much effort has been spent to understand this discrepancy.”
Overall, the study confirmed the efficacy of these two vaccines and their equivalence, noted Dr. Bernstein.
The study’s literature search process had some limitations. “Especially in stratified analyses, sparse data in some subgroups limit generalizability. ... The most accurate method, head-to-head comparisons, to evaluate the comparative efficacy of different vaccines is required in further studies,” the study investigators wrote.
Such studies would directly compare Rotarix and RotaTeq from multiple perspectives: efficacy, cost-effectiveness, strain-specific protection, the duration of protection, safety, and immunogenicity, said Dr. Goyal.
*This story was updated on May 24, 2021.
FROM JAMA PEDIATRICS
Worse outcomes for patients with COPD and COVID-19
A study of COVID-19 outcomes across the United States bolsters reports from China and Europe that indicate that patients with chronic obstructive pulmonary disease (COPD) and SARS-CoV-2 infection have worse outcomes than those of patients with COVID-19 who do not have COPD.
Investigators at the University of Texas Medical Branch at Galveston, Texas, combed through electronic health records from four geographic regions of the United States and identified a cohort of 6,056 patients with COPD among 150,775 patients whose records indicate either a diagnostic code or a positive laboratory test result for COVID-19.
Their findings indicate that patients with both COPD and COVID-19 “have worse outcomes compared to non-COPD COVID-19 patients, including 14-day hospitalization, length of stay, ICU admission, 30-day mortality, and use of mechanical ventilation,” Daniel Puebla Neira, MD, and colleagues from the University of Texas Medical Branch reported in a thematic poster presented during the American Thoracic Society (ATS) 2021 virtual international conference.
A critical care specialist who was not involved in the study said that the results are concerning but not surprising.
“If you already have a lung disease and you develop an additional lung disease on top of that, you don’t have as much reserve and you’re not going to tolerate the acute COVID infection,” said ATS expert Marc Moss, MD, Roger S. Mitchell Professor of Medicine in the division of pulmonary sciences and critical care medicine at the University of Colorado, Aurora.
The evidence shows that “patients with COPD should be even more cautious, because if they get sick and develop, they could do worse,” he said in an interview.
Retrospective analysis
Dr. Neira and colleagues assessed the characteristics and outcomes of patients with COPD who were treated for COVID-19 in the United States from March through August 2020.
Baseline demographics of the patients with and those without COPD were similar except that the mean age was higher among patients with COPD (68.62 vs. 47.08 years).
In addition, a significantly higher proportion of patients with COPD had comorbidities compared with those without COPD. Comorbidities included diabetes, hypertension, asthma, chronic kidney disease, end-stage renal disease, stroke, heart failure, cancer, coronary artery disease, and liver disease (P < .0001 for all comparisons).
Among patients with COPD, percentages were higher with respect to the following parameters: 14-day hospitalization for any cause (28.7% vs. 10.4%), COVID-19-related 14-day hospitalization (28.1% vs. 9.9%), ICU use (26.3% vs. 17.9%), mechanical ventilation use (26.3% vs. 16.1%), and 30-day mortality (13.6% vs. 7.2%; P < .0001 for all comparisons).
‘Mechanisms unclear’
“It is unclear what mechanisms drive the association between COPD and mortality in hospitalized patients with COVID-19,” the investigators wrote. “Several biological factors have been proposed, including chronic lung inflammation, oxidative stress, protease-antiprotease imbalance, and increased airway mediators.”
They recommend use of multivariable logistic regression to tease out the effects of covariates among patients with COPD and COVID-19 and call for research into long-term outcomes for these patients, “as survivors of critical illness are increasingly recognized to have cognitive, psychological, and physical consequences.”
Dr. Moss said that in general, the management of patients with COPD and COVID-19 is similar to that for patients with COVID-19 who do not have COPD, although there may be “subtle” differences, such as ventilator settings for patients with COPD.
No source of funding for the study has been disclosed. The investigators and Dr. Moss have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A study of COVID-19 outcomes across the United States bolsters reports from China and Europe that indicate that patients with chronic obstructive pulmonary disease (COPD) and SARS-CoV-2 infection have worse outcomes than those of patients with COVID-19 who do not have COPD.
Investigators at the University of Texas Medical Branch at Galveston, Texas, combed through electronic health records from four geographic regions of the United States and identified a cohort of 6,056 patients with COPD among 150,775 patients whose records indicate either a diagnostic code or a positive laboratory test result for COVID-19.
Their findings indicate that patients with both COPD and COVID-19 “have worse outcomes compared to non-COPD COVID-19 patients, including 14-day hospitalization, length of stay, ICU admission, 30-day mortality, and use of mechanical ventilation,” Daniel Puebla Neira, MD, and colleagues from the University of Texas Medical Branch reported in a thematic poster presented during the American Thoracic Society (ATS) 2021 virtual international conference.
A critical care specialist who was not involved in the study said that the results are concerning but not surprising.
“If you already have a lung disease and you develop an additional lung disease on top of that, you don’t have as much reserve and you’re not going to tolerate the acute COVID infection,” said ATS expert Marc Moss, MD, Roger S. Mitchell Professor of Medicine in the division of pulmonary sciences and critical care medicine at the University of Colorado, Aurora.
The evidence shows that “patients with COPD should be even more cautious, because if they get sick and develop, they could do worse,” he said in an interview.
Retrospective analysis
Dr. Neira and colleagues assessed the characteristics and outcomes of patients with COPD who were treated for COVID-19 in the United States from March through August 2020.
Baseline demographics of the patients with and those without COPD were similar except that the mean age was higher among patients with COPD (68.62 vs. 47.08 years).
In addition, a significantly higher proportion of patients with COPD had comorbidities compared with those without COPD. Comorbidities included diabetes, hypertension, asthma, chronic kidney disease, end-stage renal disease, stroke, heart failure, cancer, coronary artery disease, and liver disease (P < .0001 for all comparisons).
Among patients with COPD, percentages were higher with respect to the following parameters: 14-day hospitalization for any cause (28.7% vs. 10.4%), COVID-19-related 14-day hospitalization (28.1% vs. 9.9%), ICU use (26.3% vs. 17.9%), mechanical ventilation use (26.3% vs. 16.1%), and 30-day mortality (13.6% vs. 7.2%; P < .0001 for all comparisons).
‘Mechanisms unclear’
“It is unclear what mechanisms drive the association between COPD and mortality in hospitalized patients with COVID-19,” the investigators wrote. “Several biological factors have been proposed, including chronic lung inflammation, oxidative stress, protease-antiprotease imbalance, and increased airway mediators.”
They recommend use of multivariable logistic regression to tease out the effects of covariates among patients with COPD and COVID-19 and call for research into long-term outcomes for these patients, “as survivors of critical illness are increasingly recognized to have cognitive, psychological, and physical consequences.”
Dr. Moss said that in general, the management of patients with COPD and COVID-19 is similar to that for patients with COVID-19 who do not have COPD, although there may be “subtle” differences, such as ventilator settings for patients with COPD.
No source of funding for the study has been disclosed. The investigators and Dr. Moss have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A study of COVID-19 outcomes across the United States bolsters reports from China and Europe that indicate that patients with chronic obstructive pulmonary disease (COPD) and SARS-CoV-2 infection have worse outcomes than those of patients with COVID-19 who do not have COPD.
Investigators at the University of Texas Medical Branch at Galveston, Texas, combed through electronic health records from four geographic regions of the United States and identified a cohort of 6,056 patients with COPD among 150,775 patients whose records indicate either a diagnostic code or a positive laboratory test result for COVID-19.
Their findings indicate that patients with both COPD and COVID-19 “have worse outcomes compared to non-COPD COVID-19 patients, including 14-day hospitalization, length of stay, ICU admission, 30-day mortality, and use of mechanical ventilation,” Daniel Puebla Neira, MD, and colleagues from the University of Texas Medical Branch reported in a thematic poster presented during the American Thoracic Society (ATS) 2021 virtual international conference.
A critical care specialist who was not involved in the study said that the results are concerning but not surprising.
“If you already have a lung disease and you develop an additional lung disease on top of that, you don’t have as much reserve and you’re not going to tolerate the acute COVID infection,” said ATS expert Marc Moss, MD, Roger S. Mitchell Professor of Medicine in the division of pulmonary sciences and critical care medicine at the University of Colorado, Aurora.
The evidence shows that “patients with COPD should be even more cautious, because if they get sick and develop, they could do worse,” he said in an interview.
Retrospective analysis
Dr. Neira and colleagues assessed the characteristics and outcomes of patients with COPD who were treated for COVID-19 in the United States from March through August 2020.
Baseline demographics of the patients with and those without COPD were similar except that the mean age was higher among patients with COPD (68.62 vs. 47.08 years).
In addition, a significantly higher proportion of patients with COPD had comorbidities compared with those without COPD. Comorbidities included diabetes, hypertension, asthma, chronic kidney disease, end-stage renal disease, stroke, heart failure, cancer, coronary artery disease, and liver disease (P < .0001 for all comparisons).
Among patients with COPD, percentages were higher with respect to the following parameters: 14-day hospitalization for any cause (28.7% vs. 10.4%), COVID-19-related 14-day hospitalization (28.1% vs. 9.9%), ICU use (26.3% vs. 17.9%), mechanical ventilation use (26.3% vs. 16.1%), and 30-day mortality (13.6% vs. 7.2%; P < .0001 for all comparisons).
‘Mechanisms unclear’
“It is unclear what mechanisms drive the association between COPD and mortality in hospitalized patients with COVID-19,” the investigators wrote. “Several biological factors have been proposed, including chronic lung inflammation, oxidative stress, protease-antiprotease imbalance, and increased airway mediators.”
They recommend use of multivariable logistic regression to tease out the effects of covariates among patients with COPD and COVID-19 and call for research into long-term outcomes for these patients, “as survivors of critical illness are increasingly recognized to have cognitive, psychological, and physical consequences.”
Dr. Moss said that in general, the management of patients with COPD and COVID-19 is similar to that for patients with COVID-19 who do not have COPD, although there may be “subtle” differences, such as ventilator settings for patients with COPD.
No source of funding for the study has been disclosed. The investigators and Dr. Moss have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Atorvastatin: A potential treatment in COVID-19?
For patients with COVID-19 admitted to intensive care, giving atorvastatin 20 mg/d did not result in a significant reduction in risk for venous or arterial thrombosis, for treatment with extracorporeal membrane oxygenation (ECMO), or for all-cause mortality, compared with placebo in the INSPIRATION-S study.
However, there was a suggestion of benefit in the subgroup of patients who were treated within 7 days of COVID-19 symptom onset.
The study was presented by Behnood Bikdeli, MD, Brigham and Women’s Hospital, Boston, on May 16 at the annual scientific sessions of the American College of Cardiology.
He explained that COVID-19 is characterized by an exuberant immune response and that there is a potential for thrombotic events because of enhanced endothelial activation and a prothrombotic state.
“In this context, it is interesting to think about statins as potential agents to be studied in COVID-19, because as well as having lipid-lowering actions, they are also thought to have anti-inflammatory and antithrombotic effects,” he said.
In the HARP-2 trial of simvastatin in acute respiratory distress syndrome (ARDS), published a few years ago, the main results were neutral, but in the subgroup of patients with hyperinflammatory ARDS, there was a reduction in mortality with simvastatin in comparison with placebo, Dr. Bikdeli noted.
Moreover, in a series of observational studies of patients with COVID-19, use of statins was associated with a reduction in mortality among hospitalized patients. However, there are limited high-quality data to guide clinical practice, he said.
The INSPIRATION study, conducted in 11 hospitals in Iran, had a two-by-two factorial design to investigate different anticoagulant strategies and the use of atorvastatin for COVID-19 patients in the ICU.
In the anticoagulation part of the trial, which was published in JAMA in March 2020, there was no difference in the primary endpoint of an intermediate dose and standard dose of enoxaparin.
For the statin part of the trial (INSPIRATION-S), 605 patients were randomly assigned to receive atorvastatin 20 mg daily or placebo. Patients who had been taking statins beforehand were excluded. Baseline characteristics were similar for the two groups, with around a quarter of patients taking aspirin and more than 90% taking steroids.
Results showed that atorvastatin was not associated with a significant reduction in the primary outcome – a composite of adjudicated venous or arterial thrombosis, treatment with ECMO, or mortality within 30 days – which occurred in 32.7% of the statin group versus 36.3% of the placebo group (odds ratio, 0.84; P = .35).
Atorvastatin was not associated with any significant differences in any of the individual components of the primary composite endpoint. There was also no significant difference in any of the safety endpoints, which included major bleeding and elevations in liver enzyme levels.
Subgroup analyses were mostly consistent with the main findings, with one exception.
In the subgroup of patients who presented within the first 7 days of COVID-19 symptom onset, there was a hint of a potential protective effect with atorvastatin.
In this group of 171 patients, the primary endpoint occurred in 30.9% of those taking atorvastatin versus 40.3% of those taking placebo (OR, 0.60; P = .055).
“This is an interesting observation, and it is plausible, as these patients may be in a different phase of COVID-19 disease. But we need to be cognizant of the multiplicity of comparisons, and this needs to be further investigated in subsequent studies,” Dr. Bikdeli said.
Higher dose in less sick patients a better strategy?
Discussing the study at the ACC presentation, Binita Shah, MD, said the importance of enrolling COVID-19 patients into clinical trials was paramount but that these patients in the ICU may not have been the right population in which to test a statin.
“Maybe for these very sick patients, it is just too late. Trying to rein in the inflammatory cytokine storm and the interaction with thrombosis at this point is very difficult,” Dr. Shah commented.
She suggested that it might be appropriate to try statins in an earlier phase of the disease in order to prevent the inflammatory process, rather than trying to stop it after it had already started.
Dr. Shah also questioned the use of such a low dose of atorvastatin for these patients. “In the cardiovascular literature – at least in ACS [acute coronary syndrome] – high statin doses are used to see short-term benefits. In this very inflammatory milieu, I wonder whether a high-intensity regimen would be more beneficial,” she speculated.
Dr. Bikdeli replied that a low dose of atorvastatin was chosen because early on, several antiviral agents, such as ritonavir, were being used for COVID-19 patients, and these drugs were associated with increases in liver enzyme levels.
“We didn’t want to exacerbate that with high doses of statins,” he said. “But we have now established the safety profile of atorvastatin in these patients, and in retrospect, yes, a higher dose might have been better.”
The INSPIRATION study was funded by the Rajaie Cardiovascular Medical and Research Center, Tehran, Iran. Dr. Bikdeli has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For patients with COVID-19 admitted to intensive care, giving atorvastatin 20 mg/d did not result in a significant reduction in risk for venous or arterial thrombosis, for treatment with extracorporeal membrane oxygenation (ECMO), or for all-cause mortality, compared with placebo in the INSPIRATION-S study.
However, there was a suggestion of benefit in the subgroup of patients who were treated within 7 days of COVID-19 symptom onset.
The study was presented by Behnood Bikdeli, MD, Brigham and Women’s Hospital, Boston, on May 16 at the annual scientific sessions of the American College of Cardiology.
He explained that COVID-19 is characterized by an exuberant immune response and that there is a potential for thrombotic events because of enhanced endothelial activation and a prothrombotic state.
“In this context, it is interesting to think about statins as potential agents to be studied in COVID-19, because as well as having lipid-lowering actions, they are also thought to have anti-inflammatory and antithrombotic effects,” he said.
In the HARP-2 trial of simvastatin in acute respiratory distress syndrome (ARDS), published a few years ago, the main results were neutral, but in the subgroup of patients with hyperinflammatory ARDS, there was a reduction in mortality with simvastatin in comparison with placebo, Dr. Bikdeli noted.
Moreover, in a series of observational studies of patients with COVID-19, use of statins was associated with a reduction in mortality among hospitalized patients. However, there are limited high-quality data to guide clinical practice, he said.
The INSPIRATION study, conducted in 11 hospitals in Iran, had a two-by-two factorial design to investigate different anticoagulant strategies and the use of atorvastatin for COVID-19 patients in the ICU.
In the anticoagulation part of the trial, which was published in JAMA in March 2020, there was no difference in the primary endpoint of an intermediate dose and standard dose of enoxaparin.
For the statin part of the trial (INSPIRATION-S), 605 patients were randomly assigned to receive atorvastatin 20 mg daily or placebo. Patients who had been taking statins beforehand were excluded. Baseline characteristics were similar for the two groups, with around a quarter of patients taking aspirin and more than 90% taking steroids.
Results showed that atorvastatin was not associated with a significant reduction in the primary outcome – a composite of adjudicated venous or arterial thrombosis, treatment with ECMO, or mortality within 30 days – which occurred in 32.7% of the statin group versus 36.3% of the placebo group (odds ratio, 0.84; P = .35).
Atorvastatin was not associated with any significant differences in any of the individual components of the primary composite endpoint. There was also no significant difference in any of the safety endpoints, which included major bleeding and elevations in liver enzyme levels.
Subgroup analyses were mostly consistent with the main findings, with one exception.
In the subgroup of patients who presented within the first 7 days of COVID-19 symptom onset, there was a hint of a potential protective effect with atorvastatin.
In this group of 171 patients, the primary endpoint occurred in 30.9% of those taking atorvastatin versus 40.3% of those taking placebo (OR, 0.60; P = .055).
“This is an interesting observation, and it is plausible, as these patients may be in a different phase of COVID-19 disease. But we need to be cognizant of the multiplicity of comparisons, and this needs to be further investigated in subsequent studies,” Dr. Bikdeli said.
Higher dose in less sick patients a better strategy?
Discussing the study at the ACC presentation, Binita Shah, MD, said the importance of enrolling COVID-19 patients into clinical trials was paramount but that these patients in the ICU may not have been the right population in which to test a statin.
“Maybe for these very sick patients, it is just too late. Trying to rein in the inflammatory cytokine storm and the interaction with thrombosis at this point is very difficult,” Dr. Shah commented.
She suggested that it might be appropriate to try statins in an earlier phase of the disease in order to prevent the inflammatory process, rather than trying to stop it after it had already started.
Dr. Shah also questioned the use of such a low dose of atorvastatin for these patients. “In the cardiovascular literature – at least in ACS [acute coronary syndrome] – high statin doses are used to see short-term benefits. In this very inflammatory milieu, I wonder whether a high-intensity regimen would be more beneficial,” she speculated.
Dr. Bikdeli replied that a low dose of atorvastatin was chosen because early on, several antiviral agents, such as ritonavir, were being used for COVID-19 patients, and these drugs were associated with increases in liver enzyme levels.
“We didn’t want to exacerbate that with high doses of statins,” he said. “But we have now established the safety profile of atorvastatin in these patients, and in retrospect, yes, a higher dose might have been better.”
The INSPIRATION study was funded by the Rajaie Cardiovascular Medical and Research Center, Tehran, Iran. Dr. Bikdeli has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For patients with COVID-19 admitted to intensive care, giving atorvastatin 20 mg/d did not result in a significant reduction in risk for venous or arterial thrombosis, for treatment with extracorporeal membrane oxygenation (ECMO), or for all-cause mortality, compared with placebo in the INSPIRATION-S study.
However, there was a suggestion of benefit in the subgroup of patients who were treated within 7 days of COVID-19 symptom onset.
The study was presented by Behnood Bikdeli, MD, Brigham and Women’s Hospital, Boston, on May 16 at the annual scientific sessions of the American College of Cardiology.
He explained that COVID-19 is characterized by an exuberant immune response and that there is a potential for thrombotic events because of enhanced endothelial activation and a prothrombotic state.
“In this context, it is interesting to think about statins as potential agents to be studied in COVID-19, because as well as having lipid-lowering actions, they are also thought to have anti-inflammatory and antithrombotic effects,” he said.
In the HARP-2 trial of simvastatin in acute respiratory distress syndrome (ARDS), published a few years ago, the main results were neutral, but in the subgroup of patients with hyperinflammatory ARDS, there was a reduction in mortality with simvastatin in comparison with placebo, Dr. Bikdeli noted.
Moreover, in a series of observational studies of patients with COVID-19, use of statins was associated with a reduction in mortality among hospitalized patients. However, there are limited high-quality data to guide clinical practice, he said.
The INSPIRATION study, conducted in 11 hospitals in Iran, had a two-by-two factorial design to investigate different anticoagulant strategies and the use of atorvastatin for COVID-19 patients in the ICU.
In the anticoagulation part of the trial, which was published in JAMA in March 2020, there was no difference in the primary endpoint of an intermediate dose and standard dose of enoxaparin.
For the statin part of the trial (INSPIRATION-S), 605 patients were randomly assigned to receive atorvastatin 20 mg daily or placebo. Patients who had been taking statins beforehand were excluded. Baseline characteristics were similar for the two groups, with around a quarter of patients taking aspirin and more than 90% taking steroids.
Results showed that atorvastatin was not associated with a significant reduction in the primary outcome – a composite of adjudicated venous or arterial thrombosis, treatment with ECMO, or mortality within 30 days – which occurred in 32.7% of the statin group versus 36.3% of the placebo group (odds ratio, 0.84; P = .35).
Atorvastatin was not associated with any significant differences in any of the individual components of the primary composite endpoint. There was also no significant difference in any of the safety endpoints, which included major bleeding and elevations in liver enzyme levels.
Subgroup analyses were mostly consistent with the main findings, with one exception.
In the subgroup of patients who presented within the first 7 days of COVID-19 symptom onset, there was a hint of a potential protective effect with atorvastatin.
In this group of 171 patients, the primary endpoint occurred in 30.9% of those taking atorvastatin versus 40.3% of those taking placebo (OR, 0.60; P = .055).
“This is an interesting observation, and it is plausible, as these patients may be in a different phase of COVID-19 disease. But we need to be cognizant of the multiplicity of comparisons, and this needs to be further investigated in subsequent studies,” Dr. Bikdeli said.
Higher dose in less sick patients a better strategy?
Discussing the study at the ACC presentation, Binita Shah, MD, said the importance of enrolling COVID-19 patients into clinical trials was paramount but that these patients in the ICU may not have been the right population in which to test a statin.
“Maybe for these very sick patients, it is just too late. Trying to rein in the inflammatory cytokine storm and the interaction with thrombosis at this point is very difficult,” Dr. Shah commented.
She suggested that it might be appropriate to try statins in an earlier phase of the disease in order to prevent the inflammatory process, rather than trying to stop it after it had already started.
Dr. Shah also questioned the use of such a low dose of atorvastatin for these patients. “In the cardiovascular literature – at least in ACS [acute coronary syndrome] – high statin doses are used to see short-term benefits. In this very inflammatory milieu, I wonder whether a high-intensity regimen would be more beneficial,” she speculated.
Dr. Bikdeli replied that a low dose of atorvastatin was chosen because early on, several antiviral agents, such as ritonavir, were being used for COVID-19 patients, and these drugs were associated with increases in liver enzyme levels.
“We didn’t want to exacerbate that with high doses of statins,” he said. “But we have now established the safety profile of atorvastatin in these patients, and in retrospect, yes, a higher dose might have been better.”
The INSPIRATION study was funded by the Rajaie Cardiovascular Medical and Research Center, Tehran, Iran. Dr. Bikdeli has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ID doctors have the most paperwork, administrative demands
Infectious disease physicians are among the doctors carrying the largest burdens in the COVID-19 pandemic.
Perhaps not surprisingly, they were the specialists least likely to feel they were fairly compensated in the Medscape Infectious Diseases Physician Compensation Report 2021.
Only 44% said the pay was fair (down from 51% the prior year) compared with those at the high end – 79% in oncology, 69% in psychiatry, and 68% in plastic surgery who answered that way.
Income, which averaged $245,000, varied little from the previous year overall, according to the survey, but nearly one-third of ID physicians saw a decline in pay.
Again this year, ID physicians ranked near the bottom on the compensation spectrum. Pediatricians were lowest paid at $221,000. Plastic surgeons topped the chart at $526,000, followed by orthopedists at $511,000.
At the same time, the ID specialty is facing increasing shortages, a gap made even more visible in the pandemic. Medscape reported last year that nearly 80% of U.S. counties have no infectious disease specialists.
Thomas File Jr., MD, last year’s president of the Infectious Diseases Society of America, emphasized that COVID-19 is not the only threat that ID specialists have had to deal with or will have. He cited the threats that Zika and SARS posed in past years.
“COVID-19 illustrates the need for more trained ID specialists, because we know we’re going to be seeing more outbreaks in the future,” he said in an interview at the onset of the pandemic in March 2020.
Longer hours in pandemic
ID physicians’ hours generally increased during the pandemic, and they remain inflated by 8 hours per week (60 compared with 52 prepandemic) as the nation struggles to manage continuing COVID-19 infections. Physicians in critical care and public health and preventive medicine are seeing heavier workloads as well, by an average of 6-7 hours per week.
At the same time, ID physicians spent the most time of physicians in all specialties on paperwork and administrative tasks. Those tasks, which include electronic health record entry and clinical reading, took ID doctors 24.2 hours a week, more the twice the hours spent by those in anesthesiology (10.1), ophthalmology (10.3), and radiology (11.6).
The 24.2 hours was a substantial increase from the last report, when ID physicians said they spent 18.5 hours on the tasks.
The survey asked about the most challenging part of the job. ID physicians reported “long hours” as number one followed by “having so many rules and regulations.”
Only 4% said the danger or risk associated with treating COVID-19 patients was the most challenging part.
The top two aspects of their work they deemed most rewarding were “being very good at what I do” (chosen by 33%) and “knowing that I’m making the world a better place” (31%).
Patient volume up 17%
ID physicians reported seeing 78 patients per week in this report compared with 66 prepandemic, a 17% increase. Conversely, pediatricians saw an 18% drop in patient visits, followed by dermatologists, orthopedists and otolaryngologists (all down about 15%).
Despite the challenges and dissatisfaction with pay, the great majority of ID physicians said they would choose both medicine (83%) and their specialty (89%, up from 85% last year) again.
A version of this article first appeared on Medscape.com.
Infectious disease physicians are among the doctors carrying the largest burdens in the COVID-19 pandemic.
Perhaps not surprisingly, they were the specialists least likely to feel they were fairly compensated in the Medscape Infectious Diseases Physician Compensation Report 2021.
Only 44% said the pay was fair (down from 51% the prior year) compared with those at the high end – 79% in oncology, 69% in psychiatry, and 68% in plastic surgery who answered that way.
Income, which averaged $245,000, varied little from the previous year overall, according to the survey, but nearly one-third of ID physicians saw a decline in pay.
Again this year, ID physicians ranked near the bottom on the compensation spectrum. Pediatricians were lowest paid at $221,000. Plastic surgeons topped the chart at $526,000, followed by orthopedists at $511,000.
At the same time, the ID specialty is facing increasing shortages, a gap made even more visible in the pandemic. Medscape reported last year that nearly 80% of U.S. counties have no infectious disease specialists.
Thomas File Jr., MD, last year’s president of the Infectious Diseases Society of America, emphasized that COVID-19 is not the only threat that ID specialists have had to deal with or will have. He cited the threats that Zika and SARS posed in past years.
“COVID-19 illustrates the need for more trained ID specialists, because we know we’re going to be seeing more outbreaks in the future,” he said in an interview at the onset of the pandemic in March 2020.
Longer hours in pandemic
ID physicians’ hours generally increased during the pandemic, and they remain inflated by 8 hours per week (60 compared with 52 prepandemic) as the nation struggles to manage continuing COVID-19 infections. Physicians in critical care and public health and preventive medicine are seeing heavier workloads as well, by an average of 6-7 hours per week.
At the same time, ID physicians spent the most time of physicians in all specialties on paperwork and administrative tasks. Those tasks, which include electronic health record entry and clinical reading, took ID doctors 24.2 hours a week, more the twice the hours spent by those in anesthesiology (10.1), ophthalmology (10.3), and radiology (11.6).
The 24.2 hours was a substantial increase from the last report, when ID physicians said they spent 18.5 hours on the tasks.
The survey asked about the most challenging part of the job. ID physicians reported “long hours” as number one followed by “having so many rules and regulations.”
Only 4% said the danger or risk associated with treating COVID-19 patients was the most challenging part.
The top two aspects of their work they deemed most rewarding were “being very good at what I do” (chosen by 33%) and “knowing that I’m making the world a better place” (31%).
Patient volume up 17%
ID physicians reported seeing 78 patients per week in this report compared with 66 prepandemic, a 17% increase. Conversely, pediatricians saw an 18% drop in patient visits, followed by dermatologists, orthopedists and otolaryngologists (all down about 15%).
Despite the challenges and dissatisfaction with pay, the great majority of ID physicians said they would choose both medicine (83%) and their specialty (89%, up from 85% last year) again.
A version of this article first appeared on Medscape.com.
Infectious disease physicians are among the doctors carrying the largest burdens in the COVID-19 pandemic.
Perhaps not surprisingly, they were the specialists least likely to feel they were fairly compensated in the Medscape Infectious Diseases Physician Compensation Report 2021.
Only 44% said the pay was fair (down from 51% the prior year) compared with those at the high end – 79% in oncology, 69% in psychiatry, and 68% in plastic surgery who answered that way.
Income, which averaged $245,000, varied little from the previous year overall, according to the survey, but nearly one-third of ID physicians saw a decline in pay.
Again this year, ID physicians ranked near the bottom on the compensation spectrum. Pediatricians were lowest paid at $221,000. Plastic surgeons topped the chart at $526,000, followed by orthopedists at $511,000.
At the same time, the ID specialty is facing increasing shortages, a gap made even more visible in the pandemic. Medscape reported last year that nearly 80% of U.S. counties have no infectious disease specialists.
Thomas File Jr., MD, last year’s president of the Infectious Diseases Society of America, emphasized that COVID-19 is not the only threat that ID specialists have had to deal with or will have. He cited the threats that Zika and SARS posed in past years.
“COVID-19 illustrates the need for more trained ID specialists, because we know we’re going to be seeing more outbreaks in the future,” he said in an interview at the onset of the pandemic in March 2020.
Longer hours in pandemic
ID physicians’ hours generally increased during the pandemic, and they remain inflated by 8 hours per week (60 compared with 52 prepandemic) as the nation struggles to manage continuing COVID-19 infections. Physicians in critical care and public health and preventive medicine are seeing heavier workloads as well, by an average of 6-7 hours per week.
At the same time, ID physicians spent the most time of physicians in all specialties on paperwork and administrative tasks. Those tasks, which include electronic health record entry and clinical reading, took ID doctors 24.2 hours a week, more the twice the hours spent by those in anesthesiology (10.1), ophthalmology (10.3), and radiology (11.6).
The 24.2 hours was a substantial increase from the last report, when ID physicians said they spent 18.5 hours on the tasks.
The survey asked about the most challenging part of the job. ID physicians reported “long hours” as number one followed by “having so many rules and regulations.”
Only 4% said the danger or risk associated with treating COVID-19 patients was the most challenging part.
The top two aspects of their work they deemed most rewarding were “being very good at what I do” (chosen by 33%) and “knowing that I’m making the world a better place” (31%).
Patient volume up 17%
ID physicians reported seeing 78 patients per week in this report compared with 66 prepandemic, a 17% increase. Conversely, pediatricians saw an 18% drop in patient visits, followed by dermatologists, orthopedists and otolaryngologists (all down about 15%).
Despite the challenges and dissatisfaction with pay, the great majority of ID physicians said they would choose both medicine (83%) and their specialty (89%, up from 85% last year) again.
A version of this article first appeared on Medscape.com.
New guidance for those fully vaccinated against COVID-19
As has been dominating the headlines, the Centers for Disease Control and Prevention recently released updated public health guidance for those who are fully vaccinated against COVID-19.
This new guidance applies to those who are fully vaccinated as indicated by 2 weeks after the second dose in a 2-dose series or 2 weeks after a single-dose vaccine. Those who meet these criteria no longer need to wear a mask or physically distance themselves from others in both indoor and outdoor settings. For those not fully vaccinated, masking and social distancing should continue to be practiced.
The new guidance indicates that quarantine after a known exposure is no longer necessary.
Unless required by local, state, or territorial health authorities, testing is no longer required following domestic travel for fully vaccinated individuals. A negative test is still required prior to boarding an international flight to the United States and testing 3-5 days after arrival is still recommended. Self-quarantine is no longer required after international travel for fully vaccinated individuals.
The new guidance recommends that individuals who are fully vaccinated not participate in routine screening programs when feasible. Finally, if an individual has tested positive for COVID-19, regardless of vaccination status, that person should isolate and not visit public or private settings for a minimum of ten days.1
Updated guidance for health care facilities
In addition to changes for the general public in all settings, the CDC updated guidance for health care facilities on April 27, 2021. These updated guidelines allow for communal dining and visitation for fully vaccinated patients and their visitors. The guidelines indicate that fully vaccinated health care personnel (HCP) do not require quarantine after exposure to patients who have tested positive for COVID-19 as long as the HCP remains asymptomatic. They should, however, continue to utilize personal protective equipment as previously recommended. HCPs are able to be in break and meeting rooms unmasked if all HCPs are vaccinated.2
There are some important caveats to these updated guidelines. They do not apply to those who have immunocompromising conditions, including those using immunosuppressant agents. They also do not apply to locations subject to federal, state, local, tribal, or territorial laws, rules, and regulations, including local business and workplace guidance.
Those who work or reside in correction or detention facilities and homeless shelters are also still required to test after known exposures. Masking is still required by all travelers on all forms of public transportation into and within the United States.
Most importantly, the guidelines apply only to those who are fully vaccinated. Finally, no vaccine is perfect. As such, anyone who experiences symptoms indicative of COVID-19, regardless of vaccination status, should obtain viral testing and isolate themselves from others.1,2
Pros and cons to new guidance
Both sets of updated guidelines are a great example of public health guidance that is changing as the evidence is gathered and changes. This guidance is also a welcome encouragement that the vaccines are effective at decreasing transmission of this virus that has upended our world.
These guidelines leave room for change as evidence is gathered on emerging novel variants. There are, however, a few remaining concerns.
My first concern is for those who are not yet able to be vaccinated, including children under the age of 12. For families with members who are not fully vaccinated, they may have first heard the headlines of “you do not have to mask” to then read the fine print that remains. When truly following these guidelines, many social situations in both the public and private setting should still include both masking and social distancing.
There is no clarity on how these guidelines are enforced. Within the guidance, it is clear that individuals’ privacy is of utmost importance. In the absence of knowledge, that means that the assumption should be that all are not yet vaccinated. Unless there is a way to reliably demonstrate vaccination status, it would likely still be safer to assume that there are individuals who are not fully vaccinated within the setting.
Finally, although this is great news surrounding the efficacy of the vaccine, some are concerned that local mask mandates that have already started to be lifted will be completely removed. As there is still a large portion of the population not yet fully vaccinated, it seems premature for local, state, and territorial authorities to lift these mandates.
How to continue exercising caution
With the outstanding concerns, I will continue to mask in settings, particularly indoors, where I do not definitely know that everyone is vaccinated. I will continue to do this to protect my children and my patients who are not yet vaccinated, and my patients who are immunosuppressed for whom we do not yet have enough information.
I will continue to advise my patients to be thoughtful about the risk for themselves and their families as well.
There has been more benefit to these public health measures then just decreased transmission of COVID-19. I hope that this year has reinforced within us the benefits of masking and self-isolation in the cases of any contagious illnesses.
Although I am looking forward to the opportunities to interact in person with more colleagues and friends, I think we should continue to do this with caution and thoughtfulness. We must be prepared for the possibility of vaccines having decreased efficacy against novel variants as well as eventually the possibility of waning immunity. If these should occur, we need to be prepared for additional recommendation changes and tightening of restrictions.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program at Humboldt Park, Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
1. Centers for Disease Control and Prevention. Interim Public Health Recommendations for Fully Vaccinated People. U.S. Department of Health & Human Services, May 13, 2021.
2. Centers for Disease Control and Prevention. Updated Healthcare Infection Prevention and Control Recommendations in Response to COVID-19 Vaccination. U.S. Department of Health and Human Services, April 27, 2021.
As has been dominating the headlines, the Centers for Disease Control and Prevention recently released updated public health guidance for those who are fully vaccinated against COVID-19.
This new guidance applies to those who are fully vaccinated as indicated by 2 weeks after the second dose in a 2-dose series or 2 weeks after a single-dose vaccine. Those who meet these criteria no longer need to wear a mask or physically distance themselves from others in both indoor and outdoor settings. For those not fully vaccinated, masking and social distancing should continue to be practiced.
The new guidance indicates that quarantine after a known exposure is no longer necessary.
Unless required by local, state, or territorial health authorities, testing is no longer required following domestic travel for fully vaccinated individuals. A negative test is still required prior to boarding an international flight to the United States and testing 3-5 days after arrival is still recommended. Self-quarantine is no longer required after international travel for fully vaccinated individuals.
The new guidance recommends that individuals who are fully vaccinated not participate in routine screening programs when feasible. Finally, if an individual has tested positive for COVID-19, regardless of vaccination status, that person should isolate and not visit public or private settings for a minimum of ten days.1
Updated guidance for health care facilities
In addition to changes for the general public in all settings, the CDC updated guidance for health care facilities on April 27, 2021. These updated guidelines allow for communal dining and visitation for fully vaccinated patients and their visitors. The guidelines indicate that fully vaccinated health care personnel (HCP) do not require quarantine after exposure to patients who have tested positive for COVID-19 as long as the HCP remains asymptomatic. They should, however, continue to utilize personal protective equipment as previously recommended. HCPs are able to be in break and meeting rooms unmasked if all HCPs are vaccinated.2
There are some important caveats to these updated guidelines. They do not apply to those who have immunocompromising conditions, including those using immunosuppressant agents. They also do not apply to locations subject to federal, state, local, tribal, or territorial laws, rules, and regulations, including local business and workplace guidance.
Those who work or reside in correction or detention facilities and homeless shelters are also still required to test after known exposures. Masking is still required by all travelers on all forms of public transportation into and within the United States.
Most importantly, the guidelines apply only to those who are fully vaccinated. Finally, no vaccine is perfect. As such, anyone who experiences symptoms indicative of COVID-19, regardless of vaccination status, should obtain viral testing and isolate themselves from others.1,2
Pros and cons to new guidance
Both sets of updated guidelines are a great example of public health guidance that is changing as the evidence is gathered and changes. This guidance is also a welcome encouragement that the vaccines are effective at decreasing transmission of this virus that has upended our world.
These guidelines leave room for change as evidence is gathered on emerging novel variants. There are, however, a few remaining concerns.
My first concern is for those who are not yet able to be vaccinated, including children under the age of 12. For families with members who are not fully vaccinated, they may have first heard the headlines of “you do not have to mask” to then read the fine print that remains. When truly following these guidelines, many social situations in both the public and private setting should still include both masking and social distancing.
There is no clarity on how these guidelines are enforced. Within the guidance, it is clear that individuals’ privacy is of utmost importance. In the absence of knowledge, that means that the assumption should be that all are not yet vaccinated. Unless there is a way to reliably demonstrate vaccination status, it would likely still be safer to assume that there are individuals who are not fully vaccinated within the setting.
Finally, although this is great news surrounding the efficacy of the vaccine, some are concerned that local mask mandates that have already started to be lifted will be completely removed. As there is still a large portion of the population not yet fully vaccinated, it seems premature for local, state, and territorial authorities to lift these mandates.
How to continue exercising caution
With the outstanding concerns, I will continue to mask in settings, particularly indoors, where I do not definitely know that everyone is vaccinated. I will continue to do this to protect my children and my patients who are not yet vaccinated, and my patients who are immunosuppressed for whom we do not yet have enough information.
I will continue to advise my patients to be thoughtful about the risk for themselves and their families as well.
There has been more benefit to these public health measures then just decreased transmission of COVID-19. I hope that this year has reinforced within us the benefits of masking and self-isolation in the cases of any contagious illnesses.
Although I am looking forward to the opportunities to interact in person with more colleagues and friends, I think we should continue to do this with caution and thoughtfulness. We must be prepared for the possibility of vaccines having decreased efficacy against novel variants as well as eventually the possibility of waning immunity. If these should occur, we need to be prepared for additional recommendation changes and tightening of restrictions.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program at Humboldt Park, Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
1. Centers for Disease Control and Prevention. Interim Public Health Recommendations for Fully Vaccinated People. U.S. Department of Health & Human Services, May 13, 2021.
2. Centers for Disease Control and Prevention. Updated Healthcare Infection Prevention and Control Recommendations in Response to COVID-19 Vaccination. U.S. Department of Health and Human Services, April 27, 2021.
As has been dominating the headlines, the Centers for Disease Control and Prevention recently released updated public health guidance for those who are fully vaccinated against COVID-19.
This new guidance applies to those who are fully vaccinated as indicated by 2 weeks after the second dose in a 2-dose series or 2 weeks after a single-dose vaccine. Those who meet these criteria no longer need to wear a mask or physically distance themselves from others in both indoor and outdoor settings. For those not fully vaccinated, masking and social distancing should continue to be practiced.
The new guidance indicates that quarantine after a known exposure is no longer necessary.
Unless required by local, state, or territorial health authorities, testing is no longer required following domestic travel for fully vaccinated individuals. A negative test is still required prior to boarding an international flight to the United States and testing 3-5 days after arrival is still recommended. Self-quarantine is no longer required after international travel for fully vaccinated individuals.
The new guidance recommends that individuals who are fully vaccinated not participate in routine screening programs when feasible. Finally, if an individual has tested positive for COVID-19, regardless of vaccination status, that person should isolate and not visit public or private settings for a minimum of ten days.1
Updated guidance for health care facilities
In addition to changes for the general public in all settings, the CDC updated guidance for health care facilities on April 27, 2021. These updated guidelines allow for communal dining and visitation for fully vaccinated patients and their visitors. The guidelines indicate that fully vaccinated health care personnel (HCP) do not require quarantine after exposure to patients who have tested positive for COVID-19 as long as the HCP remains asymptomatic. They should, however, continue to utilize personal protective equipment as previously recommended. HCPs are able to be in break and meeting rooms unmasked if all HCPs are vaccinated.2
There are some important caveats to these updated guidelines. They do not apply to those who have immunocompromising conditions, including those using immunosuppressant agents. They also do not apply to locations subject to federal, state, local, tribal, or territorial laws, rules, and regulations, including local business and workplace guidance.
Those who work or reside in correction or detention facilities and homeless shelters are also still required to test after known exposures. Masking is still required by all travelers on all forms of public transportation into and within the United States.
Most importantly, the guidelines apply only to those who are fully vaccinated. Finally, no vaccine is perfect. As such, anyone who experiences symptoms indicative of COVID-19, regardless of vaccination status, should obtain viral testing and isolate themselves from others.1,2
Pros and cons to new guidance
Both sets of updated guidelines are a great example of public health guidance that is changing as the evidence is gathered and changes. This guidance is also a welcome encouragement that the vaccines are effective at decreasing transmission of this virus that has upended our world.
These guidelines leave room for change as evidence is gathered on emerging novel variants. There are, however, a few remaining concerns.
My first concern is for those who are not yet able to be vaccinated, including children under the age of 12. For families with members who are not fully vaccinated, they may have first heard the headlines of “you do not have to mask” to then read the fine print that remains. When truly following these guidelines, many social situations in both the public and private setting should still include both masking and social distancing.
There is no clarity on how these guidelines are enforced. Within the guidance, it is clear that individuals’ privacy is of utmost importance. In the absence of knowledge, that means that the assumption should be that all are not yet vaccinated. Unless there is a way to reliably demonstrate vaccination status, it would likely still be safer to assume that there are individuals who are not fully vaccinated within the setting.
Finally, although this is great news surrounding the efficacy of the vaccine, some are concerned that local mask mandates that have already started to be lifted will be completely removed. As there is still a large portion of the population not yet fully vaccinated, it seems premature for local, state, and territorial authorities to lift these mandates.
How to continue exercising caution
With the outstanding concerns, I will continue to mask in settings, particularly indoors, where I do not definitely know that everyone is vaccinated. I will continue to do this to protect my children and my patients who are not yet vaccinated, and my patients who are immunosuppressed for whom we do not yet have enough information.
I will continue to advise my patients to be thoughtful about the risk for themselves and their families as well.
There has been more benefit to these public health measures then just decreased transmission of COVID-19. I hope that this year has reinforced within us the benefits of masking and self-isolation in the cases of any contagious illnesses.
Although I am looking forward to the opportunities to interact in person with more colleagues and friends, I think we should continue to do this with caution and thoughtfulness. We must be prepared for the possibility of vaccines having decreased efficacy against novel variants as well as eventually the possibility of waning immunity. If these should occur, we need to be prepared for additional recommendation changes and tightening of restrictions.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program at Humboldt Park, Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
1. Centers for Disease Control and Prevention. Interim Public Health Recommendations for Fully Vaccinated People. U.S. Department of Health & Human Services, May 13, 2021.
2. Centers for Disease Control and Prevention. Updated Healthcare Infection Prevention and Control Recommendations in Response to COVID-19 Vaccination. U.S. Department of Health and Human Services, April 27, 2021.
A woman with scaling, and painful, crusted, erythematous papules and pustules on her face
Biopsy for this patient revealed folliculitis with Demodex mites visualized on histology. Direct immunofluorescence was negative. A KOH preparation was performed and was positive for large numbers of Demodex. Bacterial cultures were negative. The patient was started on a course of submicrobial doxycycline and ivermectin and showed marked improvement 1 month following treatment.
Demodex folliculorum and Demodex brevis (collectively referred to as Demodex) are microscopic parasitic mites that commonly live on human skin.1 Typically, the mite remains asymptomatic. However, in higher numbers, the infestation may cause dermatoses, called demodicosis. Lesions often present as itchy papules, pustules, and erythematous scaling on the face, ears, and scalp. Blepharitis may be present. Demodex folliculitis is more common in immunocompromised patients.2
Demodex may have a causative role in rosacea and present similarly, with a key difference being that Demodex-type rosacea is more scaly/dry and pustular than common rosacea.1 In Demodex folliculitis, bacterial cultures are often negative. A skin scraping for KOH will reveal increased mite colonization. The Demodex mite may also be seen in histologic slides.
Treatment of Demodex folliculitis includes crotamiton cream, permethrin cream, oral tetracyclines, topical or systemic metronidazole, and topical or oral ivermectin.
This case and photos were submitted by Susannah McClain, MD, Three Rivers Dermatology, Pittsburgh.
References
1. Rather PA and Hassan I. Indian J Dermatol. 2014 Jan;59(1):60-6.
2. Bachmeyer C and Moreno-Sabater A. CMAJ. 2017 Jun 26;189(25):E865.
Biopsy for this patient revealed folliculitis with Demodex mites visualized on histology. Direct immunofluorescence was negative. A KOH preparation was performed and was positive for large numbers of Demodex. Bacterial cultures were negative. The patient was started on a course of submicrobial doxycycline and ivermectin and showed marked improvement 1 month following treatment.
Demodex folliculorum and Demodex brevis (collectively referred to as Demodex) are microscopic parasitic mites that commonly live on human skin.1 Typically, the mite remains asymptomatic. However, in higher numbers, the infestation may cause dermatoses, called demodicosis. Lesions often present as itchy papules, pustules, and erythematous scaling on the face, ears, and scalp. Blepharitis may be present. Demodex folliculitis is more common in immunocompromised patients.2
Demodex may have a causative role in rosacea and present similarly, with a key difference being that Demodex-type rosacea is more scaly/dry and pustular than common rosacea.1 In Demodex folliculitis, bacterial cultures are often negative. A skin scraping for KOH will reveal increased mite colonization. The Demodex mite may also be seen in histologic slides.
Treatment of Demodex folliculitis includes crotamiton cream, permethrin cream, oral tetracyclines, topical or systemic metronidazole, and topical or oral ivermectin.
This case and photos were submitted by Susannah McClain, MD, Three Rivers Dermatology, Pittsburgh.
References
1. Rather PA and Hassan I. Indian J Dermatol. 2014 Jan;59(1):60-6.
2. Bachmeyer C and Moreno-Sabater A. CMAJ. 2017 Jun 26;189(25):E865.
Biopsy for this patient revealed folliculitis with Demodex mites visualized on histology. Direct immunofluorescence was negative. A KOH preparation was performed and was positive for large numbers of Demodex. Bacterial cultures were negative. The patient was started on a course of submicrobial doxycycline and ivermectin and showed marked improvement 1 month following treatment.
Demodex folliculorum and Demodex brevis (collectively referred to as Demodex) are microscopic parasitic mites that commonly live on human skin.1 Typically, the mite remains asymptomatic. However, in higher numbers, the infestation may cause dermatoses, called demodicosis. Lesions often present as itchy papules, pustules, and erythematous scaling on the face, ears, and scalp. Blepharitis may be present. Demodex folliculitis is more common in immunocompromised patients.2
Demodex may have a causative role in rosacea and present similarly, with a key difference being that Demodex-type rosacea is more scaly/dry and pustular than common rosacea.1 In Demodex folliculitis, bacterial cultures are often negative. A skin scraping for KOH will reveal increased mite colonization. The Demodex mite may also be seen in histologic slides.
Treatment of Demodex folliculitis includes crotamiton cream, permethrin cream, oral tetracyclines, topical or systemic metronidazole, and topical or oral ivermectin.
This case and photos were submitted by Susannah McClain, MD, Three Rivers Dermatology, Pittsburgh.
References
1. Rather PA and Hassan I. Indian J Dermatol. 2014 Jan;59(1):60-6.
2. Bachmeyer C and Moreno-Sabater A. CMAJ. 2017 Jun 26;189(25):E865.
COVID-19 in children: Weekly cases drop to 6-month low
Just 1 week after it looked like the COVID-19 situation in children might be taking another turn for the worse, the number of new pediatric cases dropped to its lowest level since October, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
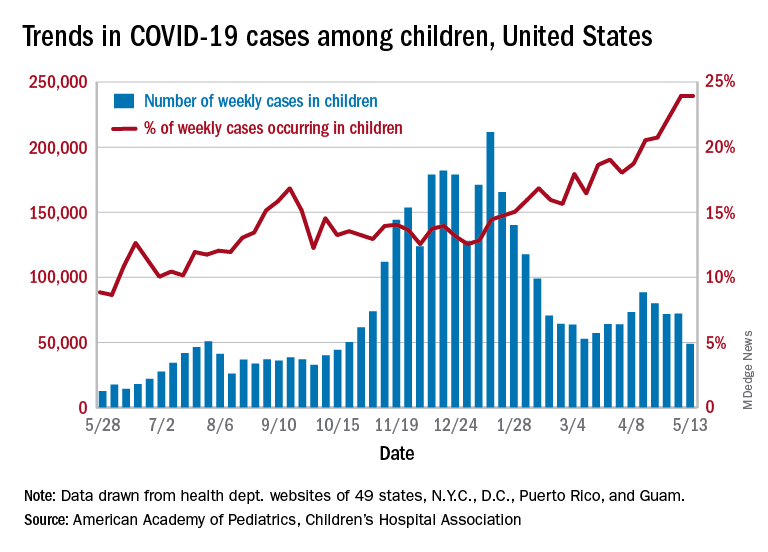
the AAP and CHA said in their weekly COVID-19 report. During the week of April 30 to May 6 – the same week Rhode Island reported a large backlog of cases and increased its total by 30% – the number of new cases went up slightly after 2 weeks of declines.
Other positive indicators come in the form of the proportion of cases occurring in children. The cumulative percentage of cases in children since the start of the pandemic remained at 14.0% for a second consecutive week, and the proportion of new cases in children held at 24.0% and did not increase for the first time in 6 weeks, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The total number of child COVID-19 cases reported in these jurisdictions is now up to 3.9 million, for a cumulative rate of 5,187 cases per 100,000 children in the United States. Among the states, total counts range from a low of 4,070 in Hawaii to 475,619 in California. Hawaii also has the lowest rate at 1,357 per 100,000 children, while the highest, 9,778 per 100,000, can be found in Rhode Island, the AAP and CHA said.
Deaths in children continue to accumulate at a relatively slow pace, with two more added during the week of May 7-13, bringing the total to 308 for the entire pandemic in 43 states, New York City, Puerto Rico, and Guam. Children’s share of the mortality burden is currently 0.06%, a figure that has not changed since mid-December, and the death rate for children with COVID-19 is 0.01%, according to the report.
Almost two-thirds (65%) of all deaths have occurred in just nine states – Arizona (31), California (21), Colorado (13), Georgia (10), Illinois (18), Maryland (10), Pennsylvania (10), Tennessee (10), and Texas (52) – and New York City (24), while eight states have not reported any deaths yet, the two groups said.
Just 1 week after it looked like the COVID-19 situation in children might be taking another turn for the worse, the number of new pediatric cases dropped to its lowest level since October, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

the AAP and CHA said in their weekly COVID-19 report. During the week of April 30 to May 6 – the same week Rhode Island reported a large backlog of cases and increased its total by 30% – the number of new cases went up slightly after 2 weeks of declines.
Other positive indicators come in the form of the proportion of cases occurring in children. The cumulative percentage of cases in children since the start of the pandemic remained at 14.0% for a second consecutive week, and the proportion of new cases in children held at 24.0% and did not increase for the first time in 6 weeks, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The total number of child COVID-19 cases reported in these jurisdictions is now up to 3.9 million, for a cumulative rate of 5,187 cases per 100,000 children in the United States. Among the states, total counts range from a low of 4,070 in Hawaii to 475,619 in California. Hawaii also has the lowest rate at 1,357 per 100,000 children, while the highest, 9,778 per 100,000, can be found in Rhode Island, the AAP and CHA said.
Deaths in children continue to accumulate at a relatively slow pace, with two more added during the week of May 7-13, bringing the total to 308 for the entire pandemic in 43 states, New York City, Puerto Rico, and Guam. Children’s share of the mortality burden is currently 0.06%, a figure that has not changed since mid-December, and the death rate for children with COVID-19 is 0.01%, according to the report.
Almost two-thirds (65%) of all deaths have occurred in just nine states – Arizona (31), California (21), Colorado (13), Georgia (10), Illinois (18), Maryland (10), Pennsylvania (10), Tennessee (10), and Texas (52) – and New York City (24), while eight states have not reported any deaths yet, the two groups said.
Just 1 week after it looked like the COVID-19 situation in children might be taking another turn for the worse, the number of new pediatric cases dropped to its lowest level since October, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

the AAP and CHA said in their weekly COVID-19 report. During the week of April 30 to May 6 – the same week Rhode Island reported a large backlog of cases and increased its total by 30% – the number of new cases went up slightly after 2 weeks of declines.
Other positive indicators come in the form of the proportion of cases occurring in children. The cumulative percentage of cases in children since the start of the pandemic remained at 14.0% for a second consecutive week, and the proportion of new cases in children held at 24.0% and did not increase for the first time in 6 weeks, based on data from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The total number of child COVID-19 cases reported in these jurisdictions is now up to 3.9 million, for a cumulative rate of 5,187 cases per 100,000 children in the United States. Among the states, total counts range from a low of 4,070 in Hawaii to 475,619 in California. Hawaii also has the lowest rate at 1,357 per 100,000 children, while the highest, 9,778 per 100,000, can be found in Rhode Island, the AAP and CHA said.
Deaths in children continue to accumulate at a relatively slow pace, with two more added during the week of May 7-13, bringing the total to 308 for the entire pandemic in 43 states, New York City, Puerto Rico, and Guam. Children’s share of the mortality burden is currently 0.06%, a figure that has not changed since mid-December, and the death rate for children with COVID-19 is 0.01%, according to the report.
Almost two-thirds (65%) of all deaths have occurred in just nine states – Arizona (31), California (21), Colorado (13), Georgia (10), Illinois (18), Maryland (10), Pennsylvania (10), Tennessee (10), and Texas (52) – and New York City (24), while eight states have not reported any deaths yet, the two groups said.
Dr. Fauci: Extraordinary challenges, scientific triumphs with COVID-19
“Vaccines have been the bright light of this extraordinary challenge that we’ve gone through,” said Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases.
In an address for the opening ceremony of the American Thoracic Society’s virtual international conference, Dr. Fauci emphasized the role of basic and clinical research and government support for science in helping turn the tide of the COVID-19 pandemic.
“A few weeks ago, I wrote an editorial in Science, because there was some misunderstanding about how and why we were able to go from a realization of a new pathogen in January of 2020, to getting doses of vaccines in the arms of individuals – a highly efficacious vaccine – 11 months later. Truly, an unprecedented accomplishment,” he said.
“But as I said in the editorial, the speed and efficiency with which these highly efficacious vaccines were developed, and their potential for saving millions of lives, are due to an extraordinary multidisciplinary effort, involving basic, preclinical, and clinical science that had been underway – out of the spotlight – for decades and decades before the unfolding of the COVID-19 pandemic, a fact that very few people really appreciate: namely, the importance of investment in biomedical research.”
The general addresses the troops
Perhaps no other audience is so well suited to receive Dr. Fauci’s speech as those who are currently attending (virtually) the ATS conference, including researchers who scrutinize the virus from every angle to describe its workings and identify its vulnerabilities, epidemiologists who study viral transmission and look for ways to thwart it, public health workers who fan out to communities across the country to push vaccine acceptance, and clinicians who specialize in critical care and pulmonary medicine, many of whom staff the respiratory floors and intensive care units where the most severely ill patients are treated.
Speaking about the lessons learned and challenges remaining from the COVID-19 pandemic, Dr. Fauci briefly reviewed the epidemiology, virology and transmission, diagnostics, and clinical course of SARS-CoV-2 infections and the therapeutics and vaccines for COVID-19.
Epidemiology
The pandemic began in December 2019 with recognition of a novel type of pneumonia in the Wuhan District of Central China, Dr. Fauci noted.
“Very quickly thereafter, in the first week of January 2020, the Chinese identified a new strain of coronavirus as [the] source of the outbreak. Fast forward to where we are right now: We have experienced and are experiencing the most devastating pandemic of a respiratory illness in the last 102 years, with already approximately 160 million individuals having been infected – and this is clearly a gross undercounting – and also 3.3 million deaths, again, very likely an undercounting,” he said.
According to the Centers for Disease Control and Prevention, as of May 9, 2021, there were approximately 32.5 million cases of COVID-19 and 578,520 deaths in the United States. Those cases and deaths occurred largely in three surges in the United States, in early spring, early summer, and late fall of 2020.
Virology and transmission
SARS-CoV-2 is a beta-coronavirus in the same subgenus as SARS-CoV-1 and some bat coronaviruses, Dr. Fauci explained. The viral genome is large, about 30,000 kilobases, and it has four structural proteins, most importantly the S or “spike” protein that allows the virus to attach to and fuse with cell membranes by binding to the ACE2 receptor on tissues in the upper and lower respiratory tract, gastrointestinal tract, cardiovascular system, and other organ systems.
The virus is transmitted mainly through exposure to respiratory droplets within 6 feet of an infected person, or sometimes through droplets or particles that remain in the air over time and various distances.
Contact with contaminated surfaces, once feared as a means of transmission, is now understood to be less common.
The virus has been detected in stool, blood, semen, and ocular secretions, although the role of transmission through these sources is still unknown.
“Some very interesting characteristics of this virus, really quite unique compared to other viruses, certainly other respiratory viruses, is [that] about a third to 40% of people who are infected never develop any symptoms,” Dr. Fauci said. “Importantly, and very problematic to what we do to contain it – particularly with regard to identification, isolation, and contract tracing – between 50% and 60% of the transmissions occur either from someone who will never develop symptoms, or someone in the presymptomatic phase of disease.”
The fundamentals of preventing acquisition and transmission are as familiar to most Americans now as the Pledge of Allegiance: universal mask wearing, physical distancing, avoiding crowds and congregate settings, preference for outdoor over indoor settings, and frequent hand washing, he noted.
Diagnostics
Tests for SARS-CoV-2 infection fall into three basic categories: molecular tests such as polymerase chain reaction (PCR) that are highly specific and highly sensitive for actual infections, antigen tests that detect the viral protein rather than the nucleic acids, and antibody tests to detect serum proteins made in response to viral infection.
Antigen testing is used largely for broader surveillance of groups of individuals to detect viral penetrance within that group, Dr. Fauci noted.
Clinical course
The clinical course of COVID-19 has some interesting characteristics but is not substantially different from a flu-like syndrome, Dr. Fauci said.
Symptoms and signs common to both types of infections include fever, cough, fatigue, anorexia, dyspnea, and myalgias, but the loss of smell and/or taste preceding the onset of respiratory symptoms is a unique feature of COVID-19.
Dr. Fauci cited data on more than 44,000 individuals with confirmed COVID-19 in China that showed that a large majority (81%) of cases were mild or moderate in nature, but 14% of patients experienced severe disease, and 5% were critically ill. The case-fatality rate in this study was 2.3%.
People at increased risk for severe disease include older adults and those of any age with certain comorbidities.
Manifestations of severe COVID-19 infections in adults can include neurological disorders, hyperinflammation, acute respiratory distress syndrome, cardiac dysfunction, hypercoagulability, and acute kidney injury.
In children, COVID-19 has been associated with a multisystem inflammatory syndrome (MIS-C) similar to Kawasaki disease.
In a substantial number of cases, the effects of COVID-19 can linger for 6 months or longer, Dr. Fauci said, pointing to a study from the University of Washington in Seattle.
Investigators there found that approximately 30% of patients enrolled at their center reported persistent symptoms for as long as 9 months after the initial illness, with fatigue as the most commonly reported symptom. One-third of outpatients with mild disease also reported persistent symptoms.
Therapeutics
Therapeutics that are either approved by the Food and Drug Administration, have emergency use authorization, or are in clinical trials for early or moderate disease include remdesivir (Veklury, Gilead Sciences), monoclonal antibodies, convalescent plasma, antiviral agents, hyperimmune globulin, anticoagulants, and immunomodulators.
Options for moderate to severe to advanced disease include dexamethasone, baricitinib (Olumiant, Eli Lilly and Company) plus remdesivir, and immunomodulators such as infliximab (Remicade, Janssen Biotech), and biosimilars.
Vaccines
Finally, Dr. Fauci reviewed the current state of vaccines, including the three with emergency use authorization from the FDA as of this writing: two nucleic acid, messenger RNA-based (mRNA) vaccines from Moderna and Pfizer/BioNTech, and an adenoviral vector-based vaccine from Johnson & Johnson.
Other vaccines in development or in use elsewhere in the world include recombinant protein and adjuvant approaches by GlaxoSmithKline and Sanofi (in a phase 2 clinical trial launched in February 2021) and by Novavax.
The three vaccines in use in the United States were highly efficacious in both clinical trials, with efficacy of about 95% for the mRNA vaccines and 67% for the Johnson & Johnson vaccine.
The real-world performance of these vaccines has been even more impressive, however.
For example, the Johnson & Johnson vaccine had 72% efficacy at preventing moderate to severe COVID 19 in the United States, 68% in Brazil, and 64% in South Africa, and 85% efficacy against severe disease across all regions studied, Dr. Fauci said.
He cited a study of 22,234 employees of the University of Texas Southwestern Medical Center in Dallas who were vaccinated under a program started on Dec. 15, 2020. The COVID-19 infection rate among these vaccinated employees was 0.05%.
Dr. Fauci recounted the experience in Israel, where the highly transmissible B.1.1.7 strain of SARS-CoV-2 is predominant. A chart of the progress shows clearly that as the vaccine doses delivered steadily increased, the number of COVID-19 cases began a precipitous decline.
Horse race
Fittingly for a speech presented on the day that the Preakness Stakes – the second leg in thoroughbred racing’s Triple Crown – was run, Dr. Fauci closed with a cartoon showing two racehorses, labeled “SARS-CoV-2” and “Vaccines,” nearly neck-and-neck, but with vaccines having a slight lead.
“We are in a race against the virus. The vaccines, and the virus: If we vaccinate the overwhelming proportion of our population, we will without a doubt be able to crush the outbreak in the same way as we have done with other viral-borne diseases like measles, smallpox, and polio.
“So, the message is: Get vaccinated,” he concluded.
“Vaccines have been the bright light of this extraordinary challenge that we’ve gone through,” said Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases.
In an address for the opening ceremony of the American Thoracic Society’s virtual international conference, Dr. Fauci emphasized the role of basic and clinical research and government support for science in helping turn the tide of the COVID-19 pandemic.
“A few weeks ago, I wrote an editorial in Science, because there was some misunderstanding about how and why we were able to go from a realization of a new pathogen in January of 2020, to getting doses of vaccines in the arms of individuals – a highly efficacious vaccine – 11 months later. Truly, an unprecedented accomplishment,” he said.
“But as I said in the editorial, the speed and efficiency with which these highly efficacious vaccines were developed, and their potential for saving millions of lives, are due to an extraordinary multidisciplinary effort, involving basic, preclinical, and clinical science that had been underway – out of the spotlight – for decades and decades before the unfolding of the COVID-19 pandemic, a fact that very few people really appreciate: namely, the importance of investment in biomedical research.”
The general addresses the troops
Perhaps no other audience is so well suited to receive Dr. Fauci’s speech as those who are currently attending (virtually) the ATS conference, including researchers who scrutinize the virus from every angle to describe its workings and identify its vulnerabilities, epidemiologists who study viral transmission and look for ways to thwart it, public health workers who fan out to communities across the country to push vaccine acceptance, and clinicians who specialize in critical care and pulmonary medicine, many of whom staff the respiratory floors and intensive care units where the most severely ill patients are treated.
Speaking about the lessons learned and challenges remaining from the COVID-19 pandemic, Dr. Fauci briefly reviewed the epidemiology, virology and transmission, diagnostics, and clinical course of SARS-CoV-2 infections and the therapeutics and vaccines for COVID-19.
Epidemiology
The pandemic began in December 2019 with recognition of a novel type of pneumonia in the Wuhan District of Central China, Dr. Fauci noted.
“Very quickly thereafter, in the first week of January 2020, the Chinese identified a new strain of coronavirus as [the] source of the outbreak. Fast forward to where we are right now: We have experienced and are experiencing the most devastating pandemic of a respiratory illness in the last 102 years, with already approximately 160 million individuals having been infected – and this is clearly a gross undercounting – and also 3.3 million deaths, again, very likely an undercounting,” he said.
According to the Centers for Disease Control and Prevention, as of May 9, 2021, there were approximately 32.5 million cases of COVID-19 and 578,520 deaths in the United States. Those cases and deaths occurred largely in three surges in the United States, in early spring, early summer, and late fall of 2020.
Virology and transmission
SARS-CoV-2 is a beta-coronavirus in the same subgenus as SARS-CoV-1 and some bat coronaviruses, Dr. Fauci explained. The viral genome is large, about 30,000 kilobases, and it has four structural proteins, most importantly the S or “spike” protein that allows the virus to attach to and fuse with cell membranes by binding to the ACE2 receptor on tissues in the upper and lower respiratory tract, gastrointestinal tract, cardiovascular system, and other organ systems.
The virus is transmitted mainly through exposure to respiratory droplets within 6 feet of an infected person, or sometimes through droplets or particles that remain in the air over time and various distances.
Contact with contaminated surfaces, once feared as a means of transmission, is now understood to be less common.
The virus has been detected in stool, blood, semen, and ocular secretions, although the role of transmission through these sources is still unknown.
“Some very interesting characteristics of this virus, really quite unique compared to other viruses, certainly other respiratory viruses, is [that] about a third to 40% of people who are infected never develop any symptoms,” Dr. Fauci said. “Importantly, and very problematic to what we do to contain it – particularly with regard to identification, isolation, and contract tracing – between 50% and 60% of the transmissions occur either from someone who will never develop symptoms, or someone in the presymptomatic phase of disease.”
The fundamentals of preventing acquisition and transmission are as familiar to most Americans now as the Pledge of Allegiance: universal mask wearing, physical distancing, avoiding crowds and congregate settings, preference for outdoor over indoor settings, and frequent hand washing, he noted.
Diagnostics
Tests for SARS-CoV-2 infection fall into three basic categories: molecular tests such as polymerase chain reaction (PCR) that are highly specific and highly sensitive for actual infections, antigen tests that detect the viral protein rather than the nucleic acids, and antibody tests to detect serum proteins made in response to viral infection.
Antigen testing is used largely for broader surveillance of groups of individuals to detect viral penetrance within that group, Dr. Fauci noted.
Clinical course
The clinical course of COVID-19 has some interesting characteristics but is not substantially different from a flu-like syndrome, Dr. Fauci said.
Symptoms and signs common to both types of infections include fever, cough, fatigue, anorexia, dyspnea, and myalgias, but the loss of smell and/or taste preceding the onset of respiratory symptoms is a unique feature of COVID-19.
Dr. Fauci cited data on more than 44,000 individuals with confirmed COVID-19 in China that showed that a large majority (81%) of cases were mild or moderate in nature, but 14% of patients experienced severe disease, and 5% were critically ill. The case-fatality rate in this study was 2.3%.
People at increased risk for severe disease include older adults and those of any age with certain comorbidities.
Manifestations of severe COVID-19 infections in adults can include neurological disorders, hyperinflammation, acute respiratory distress syndrome, cardiac dysfunction, hypercoagulability, and acute kidney injury.
In children, COVID-19 has been associated with a multisystem inflammatory syndrome (MIS-C) similar to Kawasaki disease.
In a substantial number of cases, the effects of COVID-19 can linger for 6 months or longer, Dr. Fauci said, pointing to a study from the University of Washington in Seattle.
Investigators there found that approximately 30% of patients enrolled at their center reported persistent symptoms for as long as 9 months after the initial illness, with fatigue as the most commonly reported symptom. One-third of outpatients with mild disease also reported persistent symptoms.
Therapeutics
Therapeutics that are either approved by the Food and Drug Administration, have emergency use authorization, or are in clinical trials for early or moderate disease include remdesivir (Veklury, Gilead Sciences), monoclonal antibodies, convalescent plasma, antiviral agents, hyperimmune globulin, anticoagulants, and immunomodulators.
Options for moderate to severe to advanced disease include dexamethasone, baricitinib (Olumiant, Eli Lilly and Company) plus remdesivir, and immunomodulators such as infliximab (Remicade, Janssen Biotech), and biosimilars.
Vaccines
Finally, Dr. Fauci reviewed the current state of vaccines, including the three with emergency use authorization from the FDA as of this writing: two nucleic acid, messenger RNA-based (mRNA) vaccines from Moderna and Pfizer/BioNTech, and an adenoviral vector-based vaccine from Johnson & Johnson.
Other vaccines in development or in use elsewhere in the world include recombinant protein and adjuvant approaches by GlaxoSmithKline and Sanofi (in a phase 2 clinical trial launched in February 2021) and by Novavax.
The three vaccines in use in the United States were highly efficacious in both clinical trials, with efficacy of about 95% for the mRNA vaccines and 67% for the Johnson & Johnson vaccine.
The real-world performance of these vaccines has been even more impressive, however.
For example, the Johnson & Johnson vaccine had 72% efficacy at preventing moderate to severe COVID 19 in the United States, 68% in Brazil, and 64% in South Africa, and 85% efficacy against severe disease across all regions studied, Dr. Fauci said.
He cited a study of 22,234 employees of the University of Texas Southwestern Medical Center in Dallas who were vaccinated under a program started on Dec. 15, 2020. The COVID-19 infection rate among these vaccinated employees was 0.05%.
Dr. Fauci recounted the experience in Israel, where the highly transmissible B.1.1.7 strain of SARS-CoV-2 is predominant. A chart of the progress shows clearly that as the vaccine doses delivered steadily increased, the number of COVID-19 cases began a precipitous decline.
Horse race
Fittingly for a speech presented on the day that the Preakness Stakes – the second leg in thoroughbred racing’s Triple Crown – was run, Dr. Fauci closed with a cartoon showing two racehorses, labeled “SARS-CoV-2” and “Vaccines,” nearly neck-and-neck, but with vaccines having a slight lead.
“We are in a race against the virus. The vaccines, and the virus: If we vaccinate the overwhelming proportion of our population, we will without a doubt be able to crush the outbreak in the same way as we have done with other viral-borne diseases like measles, smallpox, and polio.
“So, the message is: Get vaccinated,” he concluded.
“Vaccines have been the bright light of this extraordinary challenge that we’ve gone through,” said Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases.
In an address for the opening ceremony of the American Thoracic Society’s virtual international conference, Dr. Fauci emphasized the role of basic and clinical research and government support for science in helping turn the tide of the COVID-19 pandemic.
“A few weeks ago, I wrote an editorial in Science, because there was some misunderstanding about how and why we were able to go from a realization of a new pathogen in January of 2020, to getting doses of vaccines in the arms of individuals – a highly efficacious vaccine – 11 months later. Truly, an unprecedented accomplishment,” he said.
“But as I said in the editorial, the speed and efficiency with which these highly efficacious vaccines were developed, and their potential for saving millions of lives, are due to an extraordinary multidisciplinary effort, involving basic, preclinical, and clinical science that had been underway – out of the spotlight – for decades and decades before the unfolding of the COVID-19 pandemic, a fact that very few people really appreciate: namely, the importance of investment in biomedical research.”
The general addresses the troops
Perhaps no other audience is so well suited to receive Dr. Fauci’s speech as those who are currently attending (virtually) the ATS conference, including researchers who scrutinize the virus from every angle to describe its workings and identify its vulnerabilities, epidemiologists who study viral transmission and look for ways to thwart it, public health workers who fan out to communities across the country to push vaccine acceptance, and clinicians who specialize in critical care and pulmonary medicine, many of whom staff the respiratory floors and intensive care units where the most severely ill patients are treated.
Speaking about the lessons learned and challenges remaining from the COVID-19 pandemic, Dr. Fauci briefly reviewed the epidemiology, virology and transmission, diagnostics, and clinical course of SARS-CoV-2 infections and the therapeutics and vaccines for COVID-19.
Epidemiology
The pandemic began in December 2019 with recognition of a novel type of pneumonia in the Wuhan District of Central China, Dr. Fauci noted.
“Very quickly thereafter, in the first week of January 2020, the Chinese identified a new strain of coronavirus as [the] source of the outbreak. Fast forward to where we are right now: We have experienced and are experiencing the most devastating pandemic of a respiratory illness in the last 102 years, with already approximately 160 million individuals having been infected – and this is clearly a gross undercounting – and also 3.3 million deaths, again, very likely an undercounting,” he said.
According to the Centers for Disease Control and Prevention, as of May 9, 2021, there were approximately 32.5 million cases of COVID-19 and 578,520 deaths in the United States. Those cases and deaths occurred largely in three surges in the United States, in early spring, early summer, and late fall of 2020.
Virology and transmission
SARS-CoV-2 is a beta-coronavirus in the same subgenus as SARS-CoV-1 and some bat coronaviruses, Dr. Fauci explained. The viral genome is large, about 30,000 kilobases, and it has four structural proteins, most importantly the S or “spike” protein that allows the virus to attach to and fuse with cell membranes by binding to the ACE2 receptor on tissues in the upper and lower respiratory tract, gastrointestinal tract, cardiovascular system, and other organ systems.
The virus is transmitted mainly through exposure to respiratory droplets within 6 feet of an infected person, or sometimes through droplets or particles that remain in the air over time and various distances.
Contact with contaminated surfaces, once feared as a means of transmission, is now understood to be less common.
The virus has been detected in stool, blood, semen, and ocular secretions, although the role of transmission through these sources is still unknown.
“Some very interesting characteristics of this virus, really quite unique compared to other viruses, certainly other respiratory viruses, is [that] about a third to 40% of people who are infected never develop any symptoms,” Dr. Fauci said. “Importantly, and very problematic to what we do to contain it – particularly with regard to identification, isolation, and contract tracing – between 50% and 60% of the transmissions occur either from someone who will never develop symptoms, or someone in the presymptomatic phase of disease.”
The fundamentals of preventing acquisition and transmission are as familiar to most Americans now as the Pledge of Allegiance: universal mask wearing, physical distancing, avoiding crowds and congregate settings, preference for outdoor over indoor settings, and frequent hand washing, he noted.
Diagnostics
Tests for SARS-CoV-2 infection fall into three basic categories: molecular tests such as polymerase chain reaction (PCR) that are highly specific and highly sensitive for actual infections, antigen tests that detect the viral protein rather than the nucleic acids, and antibody tests to detect serum proteins made in response to viral infection.
Antigen testing is used largely for broader surveillance of groups of individuals to detect viral penetrance within that group, Dr. Fauci noted.
Clinical course
The clinical course of COVID-19 has some interesting characteristics but is not substantially different from a flu-like syndrome, Dr. Fauci said.
Symptoms and signs common to both types of infections include fever, cough, fatigue, anorexia, dyspnea, and myalgias, but the loss of smell and/or taste preceding the onset of respiratory symptoms is a unique feature of COVID-19.
Dr. Fauci cited data on more than 44,000 individuals with confirmed COVID-19 in China that showed that a large majority (81%) of cases were mild or moderate in nature, but 14% of patients experienced severe disease, and 5% were critically ill. The case-fatality rate in this study was 2.3%.
People at increased risk for severe disease include older adults and those of any age with certain comorbidities.
Manifestations of severe COVID-19 infections in adults can include neurological disorders, hyperinflammation, acute respiratory distress syndrome, cardiac dysfunction, hypercoagulability, and acute kidney injury.
In children, COVID-19 has been associated with a multisystem inflammatory syndrome (MIS-C) similar to Kawasaki disease.
In a substantial number of cases, the effects of COVID-19 can linger for 6 months or longer, Dr. Fauci said, pointing to a study from the University of Washington in Seattle.
Investigators there found that approximately 30% of patients enrolled at their center reported persistent symptoms for as long as 9 months after the initial illness, with fatigue as the most commonly reported symptom. One-third of outpatients with mild disease also reported persistent symptoms.
Therapeutics
Therapeutics that are either approved by the Food and Drug Administration, have emergency use authorization, or are in clinical trials for early or moderate disease include remdesivir (Veklury, Gilead Sciences), monoclonal antibodies, convalescent plasma, antiviral agents, hyperimmune globulin, anticoagulants, and immunomodulators.
Options for moderate to severe to advanced disease include dexamethasone, baricitinib (Olumiant, Eli Lilly and Company) plus remdesivir, and immunomodulators such as infliximab (Remicade, Janssen Biotech), and biosimilars.
Vaccines
Finally, Dr. Fauci reviewed the current state of vaccines, including the three with emergency use authorization from the FDA as of this writing: two nucleic acid, messenger RNA-based (mRNA) vaccines from Moderna and Pfizer/BioNTech, and an adenoviral vector-based vaccine from Johnson & Johnson.
Other vaccines in development or in use elsewhere in the world include recombinant protein and adjuvant approaches by GlaxoSmithKline and Sanofi (in a phase 2 clinical trial launched in February 2021) and by Novavax.
The three vaccines in use in the United States were highly efficacious in both clinical trials, with efficacy of about 95% for the mRNA vaccines and 67% for the Johnson & Johnson vaccine.
The real-world performance of these vaccines has been even more impressive, however.
For example, the Johnson & Johnson vaccine had 72% efficacy at preventing moderate to severe COVID 19 in the United States, 68% in Brazil, and 64% in South Africa, and 85% efficacy against severe disease across all regions studied, Dr. Fauci said.
He cited a study of 22,234 employees of the University of Texas Southwestern Medical Center in Dallas who were vaccinated under a program started on Dec. 15, 2020. The COVID-19 infection rate among these vaccinated employees was 0.05%.
Dr. Fauci recounted the experience in Israel, where the highly transmissible B.1.1.7 strain of SARS-CoV-2 is predominant. A chart of the progress shows clearly that as the vaccine doses delivered steadily increased, the number of COVID-19 cases began a precipitous decline.
Horse race
Fittingly for a speech presented on the day that the Preakness Stakes – the second leg in thoroughbred racing’s Triple Crown – was run, Dr. Fauci closed with a cartoon showing two racehorses, labeled “SARS-CoV-2” and “Vaccines,” nearly neck-and-neck, but with vaccines having a slight lead.
“We are in a race against the virus. The vaccines, and the virus: If we vaccinate the overwhelming proportion of our population, we will without a doubt be able to crush the outbreak in the same way as we have done with other viral-borne diseases like measles, smallpox, and polio.
“So, the message is: Get vaccinated,” he concluded.
Infants with UTI do not have an increased risk of bacterial meningitis
The decision to perform a spinal tap procedure in infants to determine whether they have bacterial meningitis should not be guided by abnormal urinalysis results alone, according to new research published in JAMA Network Open.
The findings suggest febrile infants with positive urinalysis results do not have a higher risk of bacterial meningitis than those with negative urinalysis results.
Nearly 1 in 100,000 people are diagnosed with bacterial meningitis in the United States each year, according to Boston Children’s Hospital. Infants have an increased risk for bacterial meningitis, compared with those in other age groups, according to the Centers for Disease Control and Prevention. However, rates of the infectious disease have been declining in the United States since the late 1990s.
Researchers of the current study said published guidelines and quality initiatives recommend performing a lumbar puncture on febrile infants with positive urinalysis results to exclude bacterial meningitis as a cause.
“It really raises the question of should we be doing everything we’re doing?” study author Brett Burstein, MD, PhD, MPH, said in an interview. “What we conclude here is that, contrary to all the published guidelines, this invasive strategy for testing in well-appearing infants should not be guided by the urinalysis results. That’s a major departure.”
The study adds to growing research that questions whether a lumbar puncture in infants with fever and a positive urinalysis results should be routinely required.
“[Our findings] certainly goes against 30 years of clinical decisions, rules, and guidelines,” Dr. Burstein said. “We think they’re very important and they stand to change practice because approximately 500 infants will undergo these invasive procedures to not miss that needle in the haystack.”
Dr. Burstein, a clinician-scientist in pediatric emergency medicine at Montreal Children’s Hospital, led a team of researchers to perform a meta-analysis of 48 studies, including data from more than 25,000 infants.
Researchers found that the prevalence of bacterial meningitis in well-appearing febrile infants aged 29-60 days with a positive urinalysis results was 0.44%, compared with 0.50% of infants with negative urinalysis results.
Instead of relying on urinalysis results alone, Dr. Burstein suggests doctors use other stratifying biomarkers to decide whether they should perform a lumbar puncture.
“If you’ve done blood testing, for example, and your blood test results suggest serious infection, that should guide the decision to go on to invasive testing,” Dr. Burstein said. “You can use your urinary tract infection information in combination now with blood results.”
This means that, if infants have reassuring blood results, despite having a urinary tract infection, they do not need invasive testing, according to Dr. Burstein.
Some of the risks involved with invasive lumbar puncture testing include infection, bleeding, respiratory problems, as well as pain for the infant and parental anxiety.
Paul Aronson, MD, MHS, of Yale University, New Haven, Conn., who was not involved in the study, said in an interview that he has personally moved away from routine lumbar puncture in infants with a positive urinalysis, but added that many doctors have not.
Dr. Aronson said that, although there have been previous studies on this topic, what sets Dr. Burstein’s study apart is the fact that it has a “tightly defined” group of patients, which are infants aged between 29 and 60 days. He also said it is helpful that the study compared the prevalence of meningitis between infants who had positive urinalysis results with those who had negative results.
“The study compared positive urinalyses to negative analyses, which in the meta-analysis form had not been done previously,” Dr. Aronson said. “And so I think this [current study] probably provides some of the strongest evidence.”
No relevant financial relationships were reported.
The decision to perform a spinal tap procedure in infants to determine whether they have bacterial meningitis should not be guided by abnormal urinalysis results alone, according to new research published in JAMA Network Open.
The findings suggest febrile infants with positive urinalysis results do not have a higher risk of bacterial meningitis than those with negative urinalysis results.
Nearly 1 in 100,000 people are diagnosed with bacterial meningitis in the United States each year, according to Boston Children’s Hospital. Infants have an increased risk for bacterial meningitis, compared with those in other age groups, according to the Centers for Disease Control and Prevention. However, rates of the infectious disease have been declining in the United States since the late 1990s.
Researchers of the current study said published guidelines and quality initiatives recommend performing a lumbar puncture on febrile infants with positive urinalysis results to exclude bacterial meningitis as a cause.
“It really raises the question of should we be doing everything we’re doing?” study author Brett Burstein, MD, PhD, MPH, said in an interview. “What we conclude here is that, contrary to all the published guidelines, this invasive strategy for testing in well-appearing infants should not be guided by the urinalysis results. That’s a major departure.”
The study adds to growing research that questions whether a lumbar puncture in infants with fever and a positive urinalysis results should be routinely required.
“[Our findings] certainly goes against 30 years of clinical decisions, rules, and guidelines,” Dr. Burstein said. “We think they’re very important and they stand to change practice because approximately 500 infants will undergo these invasive procedures to not miss that needle in the haystack.”
Dr. Burstein, a clinician-scientist in pediatric emergency medicine at Montreal Children’s Hospital, led a team of researchers to perform a meta-analysis of 48 studies, including data from more than 25,000 infants.
Researchers found that the prevalence of bacterial meningitis in well-appearing febrile infants aged 29-60 days with a positive urinalysis results was 0.44%, compared with 0.50% of infants with negative urinalysis results.
Instead of relying on urinalysis results alone, Dr. Burstein suggests doctors use other stratifying biomarkers to decide whether they should perform a lumbar puncture.
“If you’ve done blood testing, for example, and your blood test results suggest serious infection, that should guide the decision to go on to invasive testing,” Dr. Burstein said. “You can use your urinary tract infection information in combination now with blood results.”
This means that, if infants have reassuring blood results, despite having a urinary tract infection, they do not need invasive testing, according to Dr. Burstein.
Some of the risks involved with invasive lumbar puncture testing include infection, bleeding, respiratory problems, as well as pain for the infant and parental anxiety.
Paul Aronson, MD, MHS, of Yale University, New Haven, Conn., who was not involved in the study, said in an interview that he has personally moved away from routine lumbar puncture in infants with a positive urinalysis, but added that many doctors have not.
Dr. Aronson said that, although there have been previous studies on this topic, what sets Dr. Burstein’s study apart is the fact that it has a “tightly defined” group of patients, which are infants aged between 29 and 60 days. He also said it is helpful that the study compared the prevalence of meningitis between infants who had positive urinalysis results with those who had negative results.
“The study compared positive urinalyses to negative analyses, which in the meta-analysis form had not been done previously,” Dr. Aronson said. “And so I think this [current study] probably provides some of the strongest evidence.”
No relevant financial relationships were reported.
The decision to perform a spinal tap procedure in infants to determine whether they have bacterial meningitis should not be guided by abnormal urinalysis results alone, according to new research published in JAMA Network Open.
The findings suggest febrile infants with positive urinalysis results do not have a higher risk of bacterial meningitis than those with negative urinalysis results.
Nearly 1 in 100,000 people are diagnosed with bacterial meningitis in the United States each year, according to Boston Children’s Hospital. Infants have an increased risk for bacterial meningitis, compared with those in other age groups, according to the Centers for Disease Control and Prevention. However, rates of the infectious disease have been declining in the United States since the late 1990s.
Researchers of the current study said published guidelines and quality initiatives recommend performing a lumbar puncture on febrile infants with positive urinalysis results to exclude bacterial meningitis as a cause.
“It really raises the question of should we be doing everything we’re doing?” study author Brett Burstein, MD, PhD, MPH, said in an interview. “What we conclude here is that, contrary to all the published guidelines, this invasive strategy for testing in well-appearing infants should not be guided by the urinalysis results. That’s a major departure.”
The study adds to growing research that questions whether a lumbar puncture in infants with fever and a positive urinalysis results should be routinely required.
“[Our findings] certainly goes against 30 years of clinical decisions, rules, and guidelines,” Dr. Burstein said. “We think they’re very important and they stand to change practice because approximately 500 infants will undergo these invasive procedures to not miss that needle in the haystack.”
Dr. Burstein, a clinician-scientist in pediatric emergency medicine at Montreal Children’s Hospital, led a team of researchers to perform a meta-analysis of 48 studies, including data from more than 25,000 infants.
Researchers found that the prevalence of bacterial meningitis in well-appearing febrile infants aged 29-60 days with a positive urinalysis results was 0.44%, compared with 0.50% of infants with negative urinalysis results.
Instead of relying on urinalysis results alone, Dr. Burstein suggests doctors use other stratifying biomarkers to decide whether they should perform a lumbar puncture.
“If you’ve done blood testing, for example, and your blood test results suggest serious infection, that should guide the decision to go on to invasive testing,” Dr. Burstein said. “You can use your urinary tract infection information in combination now with blood results.”
This means that, if infants have reassuring blood results, despite having a urinary tract infection, they do not need invasive testing, according to Dr. Burstein.
Some of the risks involved with invasive lumbar puncture testing include infection, bleeding, respiratory problems, as well as pain for the infant and parental anxiety.
Paul Aronson, MD, MHS, of Yale University, New Haven, Conn., who was not involved in the study, said in an interview that he has personally moved away from routine lumbar puncture in infants with a positive urinalysis, but added that many doctors have not.
Dr. Aronson said that, although there have been previous studies on this topic, what sets Dr. Burstein’s study apart is the fact that it has a “tightly defined” group of patients, which are infants aged between 29 and 60 days. He also said it is helpful that the study compared the prevalence of meningitis between infants who had positive urinalysis results with those who had negative results.
“The study compared positive urinalyses to negative analyses, which in the meta-analysis form had not been done previously,” Dr. Aronson said. “And so I think this [current study] probably provides some of the strongest evidence.”
No relevant financial relationships were reported.
FROM JAMA NETWORK OPEN