User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Alcoholic drinks stand out in novel trial exploring AFib triggers
People with paroxysmal atrial fibrillation who explored potential triggers of their arrhythmia, and used them to make lifestyle changes, went on to show a 40% decline in subjectively experienced bouts of AFib in a randomized trial with an unusual design.
But the study didn’t provide evidence that the drop in self-reported AFib necessarily improved their quality of life, its primary endpoint. Nor was there any apparent relationship between potential triggers and AFib episodes detected less subjectively using a handheld electrocardiography monitor.
Although the study – called I-STOP-AFib – has limitations, its results jibe with alcohol intake’s increasingly appreciated status as a potential AFib trigger. It was alone among many possible triggers tested in showing a consistent association with self-reported AFib.
As a result, the study offers no support for such a link between the arrhythmia and caffeine intake, sleep deprivation, dehydration, exercise, or other conditions sometimes perceived as triggers, observed principal investigator Gregory M. Marcus, MD, MAS, University of California, San Francisco, when presenting results at the American Heart Association scientific sessions. He is also lead author on the study’s simultaneous publication in JAMA Cardiology.
The I-STOP-AFib trial was unusual in part for its virtual design, in which participants followed instructions and tracked AFib episodes – both perceived and detected by the handheld ECG device – through a smartphone application. It also featured an N-of-1 randomized comparisons of different weeks in which individuals were or were not exposed to their self-selected trigger.
Such patients following their own weekly personalized randomization were compared to an entirely separate randomized control arm of the trial, in which patients simply tracked any ECG-monitored and self-perceived AFib episodes.
Current use in patients
Although wearable and smartphone-based ECG recorders are increasingly popular for AFib screening, Dr. Marcus said the devices may be especially helpful for validating whether a person’s symptoms are actually caused by AFib.
“I have actually suggested to some of my patients that they run some of these experiments,” he said at a media briefing on I-STOP-AFib before his main presentation of the trial. The demonstration might help patients recognize that some perceived triggers actually do not induce AFib.
Allowing patients to determine on their own whether a substance indeed triggers their AFib “is an efficient use of these devices,” Dr. Marcus said. Such N-of-1 exploration of possible triggers “might help free patients up to enjoy substances – caffeine or coffee is one example – that they otherwise might not, and may help actually reassure them that certain exposures –like certain exercises, which can also be beneficial – might actually not be harmful.”
Dr. Marcus and the other authors on the report noted – as he did at the AHA sessions – that the study has several limitations, such as the subjectivity of self-reported AFib, dropouts from the trial that shrank the randomization arms, and a population that may not be very representative.
There is also the potential for detection bias in the group assigned to track their selected triggers, as Dr. Marcus and some observers have noted.
It follows that conscious avoidance of a potential AFib trigger might well lead to a reduction in AFib subjectively identified by symptoms, proposed David Conen, MD, MPH, Population Health Research Institute, McMaster University, Hamilton, Ont. But perhaps there would have been no reduction in AFib had it been objectively documented with the handheld ECG device, he said in an interview.
“If I were to redesign the study,” he said, “I think the primary endpoint should be confirmed atrial fibrillation, because we would have to show first that the specific trigger actually reduced objective AFib events before we then try to address the question whether reducing that trigger improves quality of life.”
Unrepresentative sample
The trial entered 446 overwhelmingly White and college-educated adults known to have symptomatic paroxysmal AFib who were “interested in testing a presumed AFib trigger they could readily introduce or withhold” and who owned a smartphone; the average age was 58 years, and 58% were men. The cohort was randomly assigned to the trigger-testing group or the control group, charged only with tracking their AFib.
Of the total, 320, or about 72%, completed the study; those who did not were mostly from the trigger-testing arm, leaving 136 in that group versus 184 patients in the control group.
Potential triggers that participants selected for tracking included, foremost, caffeine, alcohol, reduced sleep, and exercise, followed by lying on the left side, dehydration, large meals, and cold food or drink, the report noted.
Patients in the control group used the smartphone app and handheld ECG monitor (KardiaMobile, AliveCor) to document the duration and severity of AFib episodes daily and received data summary reports through the app weekly for 10 weeks. They then had the option to follow the trigger-testing protocol at least once.
Those in the trigger-testing group conducted their N-of-1 trials by exposing themselves to their chosen potential trigger during 3 separate weeks and avoiding the trigger during 3 other weeks, alternating each of the 6 weeks of trigger exposure or avoidance. They were instructed through the app to start the 6-week sequence with one or the other strategy randomly and to regularly track their AFib.
At the end of 6 weeks, each participant in the trigger-testing group had the opportunity to review their data for any potential trigger-AFib associations. They were then to use the next 4 weeks to enact lifestyle changes based on what they learned – as described in the report and on clinicaltrials.gov. They had the option of repeating the entire N-of-1 sequence at least one more time.
Participants in both the trigger-tracking and control arms were tested at baseline and at 10 weeks using the validated Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire.
AFEQT scores didn’t change significantly over the 10 weeks in either arm, nor were they significantly different in one arm, compared with the other.
On the other hand, patients in the trigger-tracking arm reported significantly fewer daily AFib episodes during the final 4-week period of lifestyle changes based on their N-of-1 trial results, compared to the monitoring-only control group’s final 4 weeks.
The adjusted relative risk in the trigger-tracking arm was 0.60 (95% confidence interval, 0.43-0.83; P < .001), the difference driven by patients who selected alcohol, dehydration, or exercise for their trigger, Dr. Marcus reported.
Only alcohol intake emerged consistently as a significant predictor of risk for self-reported AFib episodes in a series of meta-analyses conducted using all of the individual N-of-1 trials that provided per-protocol data. The odds ratio was 1.77 (95% CI, 1.20-2.69).
I-STOP-AFib explored an important subject “that has been understudied,” Dr. Conen said. “The trial has some limitations that the authors address themselves, but hopefully it opens the path to future studies that can build upon this experience.”
Dr. Marcus reported receiving personal fees and equity interest from InCarda Therapeutics; personal fees from Johnson & Johnson; and grants from Baylis Medical, Medtronic, the National Institutes of Health, the Patient-Centered Outcomes Research Institute, and the California Tobacco-Related Disease Research Program.
A version of this article first appeared on Medscape.com.
People with paroxysmal atrial fibrillation who explored potential triggers of their arrhythmia, and used them to make lifestyle changes, went on to show a 40% decline in subjectively experienced bouts of AFib in a randomized trial with an unusual design.
But the study didn’t provide evidence that the drop in self-reported AFib necessarily improved their quality of life, its primary endpoint. Nor was there any apparent relationship between potential triggers and AFib episodes detected less subjectively using a handheld electrocardiography monitor.
Although the study – called I-STOP-AFib – has limitations, its results jibe with alcohol intake’s increasingly appreciated status as a potential AFib trigger. It was alone among many possible triggers tested in showing a consistent association with self-reported AFib.
As a result, the study offers no support for such a link between the arrhythmia and caffeine intake, sleep deprivation, dehydration, exercise, or other conditions sometimes perceived as triggers, observed principal investigator Gregory M. Marcus, MD, MAS, University of California, San Francisco, when presenting results at the American Heart Association scientific sessions. He is also lead author on the study’s simultaneous publication in JAMA Cardiology.
The I-STOP-AFib trial was unusual in part for its virtual design, in which participants followed instructions and tracked AFib episodes – both perceived and detected by the handheld ECG device – through a smartphone application. It also featured an N-of-1 randomized comparisons of different weeks in which individuals were or were not exposed to their self-selected trigger.
Such patients following their own weekly personalized randomization were compared to an entirely separate randomized control arm of the trial, in which patients simply tracked any ECG-monitored and self-perceived AFib episodes.
Current use in patients
Although wearable and smartphone-based ECG recorders are increasingly popular for AFib screening, Dr. Marcus said the devices may be especially helpful for validating whether a person’s symptoms are actually caused by AFib.
“I have actually suggested to some of my patients that they run some of these experiments,” he said at a media briefing on I-STOP-AFib before his main presentation of the trial. The demonstration might help patients recognize that some perceived triggers actually do not induce AFib.
Allowing patients to determine on their own whether a substance indeed triggers their AFib “is an efficient use of these devices,” Dr. Marcus said. Such N-of-1 exploration of possible triggers “might help free patients up to enjoy substances – caffeine or coffee is one example – that they otherwise might not, and may help actually reassure them that certain exposures –like certain exercises, which can also be beneficial – might actually not be harmful.”
Dr. Marcus and the other authors on the report noted – as he did at the AHA sessions – that the study has several limitations, such as the subjectivity of self-reported AFib, dropouts from the trial that shrank the randomization arms, and a population that may not be very representative.
There is also the potential for detection bias in the group assigned to track their selected triggers, as Dr. Marcus and some observers have noted.
It follows that conscious avoidance of a potential AFib trigger might well lead to a reduction in AFib subjectively identified by symptoms, proposed David Conen, MD, MPH, Population Health Research Institute, McMaster University, Hamilton, Ont. But perhaps there would have been no reduction in AFib had it been objectively documented with the handheld ECG device, he said in an interview.
“If I were to redesign the study,” he said, “I think the primary endpoint should be confirmed atrial fibrillation, because we would have to show first that the specific trigger actually reduced objective AFib events before we then try to address the question whether reducing that trigger improves quality of life.”
Unrepresentative sample
The trial entered 446 overwhelmingly White and college-educated adults known to have symptomatic paroxysmal AFib who were “interested in testing a presumed AFib trigger they could readily introduce or withhold” and who owned a smartphone; the average age was 58 years, and 58% were men. The cohort was randomly assigned to the trigger-testing group or the control group, charged only with tracking their AFib.
Of the total, 320, or about 72%, completed the study; those who did not were mostly from the trigger-testing arm, leaving 136 in that group versus 184 patients in the control group.
Potential triggers that participants selected for tracking included, foremost, caffeine, alcohol, reduced sleep, and exercise, followed by lying on the left side, dehydration, large meals, and cold food or drink, the report noted.
Patients in the control group used the smartphone app and handheld ECG monitor (KardiaMobile, AliveCor) to document the duration and severity of AFib episodes daily and received data summary reports through the app weekly for 10 weeks. They then had the option to follow the trigger-testing protocol at least once.
Those in the trigger-testing group conducted their N-of-1 trials by exposing themselves to their chosen potential trigger during 3 separate weeks and avoiding the trigger during 3 other weeks, alternating each of the 6 weeks of trigger exposure or avoidance. They were instructed through the app to start the 6-week sequence with one or the other strategy randomly and to regularly track their AFib.
At the end of 6 weeks, each participant in the trigger-testing group had the opportunity to review their data for any potential trigger-AFib associations. They were then to use the next 4 weeks to enact lifestyle changes based on what they learned – as described in the report and on clinicaltrials.gov. They had the option of repeating the entire N-of-1 sequence at least one more time.
Participants in both the trigger-tracking and control arms were tested at baseline and at 10 weeks using the validated Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire.
AFEQT scores didn’t change significantly over the 10 weeks in either arm, nor were they significantly different in one arm, compared with the other.
On the other hand, patients in the trigger-tracking arm reported significantly fewer daily AFib episodes during the final 4-week period of lifestyle changes based on their N-of-1 trial results, compared to the monitoring-only control group’s final 4 weeks.
The adjusted relative risk in the trigger-tracking arm was 0.60 (95% confidence interval, 0.43-0.83; P < .001), the difference driven by patients who selected alcohol, dehydration, or exercise for their trigger, Dr. Marcus reported.
Only alcohol intake emerged consistently as a significant predictor of risk for self-reported AFib episodes in a series of meta-analyses conducted using all of the individual N-of-1 trials that provided per-protocol data. The odds ratio was 1.77 (95% CI, 1.20-2.69).
I-STOP-AFib explored an important subject “that has been understudied,” Dr. Conen said. “The trial has some limitations that the authors address themselves, but hopefully it opens the path to future studies that can build upon this experience.”
Dr. Marcus reported receiving personal fees and equity interest from InCarda Therapeutics; personal fees from Johnson & Johnson; and grants from Baylis Medical, Medtronic, the National Institutes of Health, the Patient-Centered Outcomes Research Institute, and the California Tobacco-Related Disease Research Program.
A version of this article first appeared on Medscape.com.
People with paroxysmal atrial fibrillation who explored potential triggers of their arrhythmia, and used them to make lifestyle changes, went on to show a 40% decline in subjectively experienced bouts of AFib in a randomized trial with an unusual design.
But the study didn’t provide evidence that the drop in self-reported AFib necessarily improved their quality of life, its primary endpoint. Nor was there any apparent relationship between potential triggers and AFib episodes detected less subjectively using a handheld electrocardiography monitor.
Although the study – called I-STOP-AFib – has limitations, its results jibe with alcohol intake’s increasingly appreciated status as a potential AFib trigger. It was alone among many possible triggers tested in showing a consistent association with self-reported AFib.
As a result, the study offers no support for such a link between the arrhythmia and caffeine intake, sleep deprivation, dehydration, exercise, or other conditions sometimes perceived as triggers, observed principal investigator Gregory M. Marcus, MD, MAS, University of California, San Francisco, when presenting results at the American Heart Association scientific sessions. He is also lead author on the study’s simultaneous publication in JAMA Cardiology.
The I-STOP-AFib trial was unusual in part for its virtual design, in which participants followed instructions and tracked AFib episodes – both perceived and detected by the handheld ECG device – through a smartphone application. It also featured an N-of-1 randomized comparisons of different weeks in which individuals were or were not exposed to their self-selected trigger.
Such patients following their own weekly personalized randomization were compared to an entirely separate randomized control arm of the trial, in which patients simply tracked any ECG-monitored and self-perceived AFib episodes.
Current use in patients
Although wearable and smartphone-based ECG recorders are increasingly popular for AFib screening, Dr. Marcus said the devices may be especially helpful for validating whether a person’s symptoms are actually caused by AFib.
“I have actually suggested to some of my patients that they run some of these experiments,” he said at a media briefing on I-STOP-AFib before his main presentation of the trial. The demonstration might help patients recognize that some perceived triggers actually do not induce AFib.
Allowing patients to determine on their own whether a substance indeed triggers their AFib “is an efficient use of these devices,” Dr. Marcus said. Such N-of-1 exploration of possible triggers “might help free patients up to enjoy substances – caffeine or coffee is one example – that they otherwise might not, and may help actually reassure them that certain exposures –like certain exercises, which can also be beneficial – might actually not be harmful.”
Dr. Marcus and the other authors on the report noted – as he did at the AHA sessions – that the study has several limitations, such as the subjectivity of self-reported AFib, dropouts from the trial that shrank the randomization arms, and a population that may not be very representative.
There is also the potential for detection bias in the group assigned to track their selected triggers, as Dr. Marcus and some observers have noted.
It follows that conscious avoidance of a potential AFib trigger might well lead to a reduction in AFib subjectively identified by symptoms, proposed David Conen, MD, MPH, Population Health Research Institute, McMaster University, Hamilton, Ont. But perhaps there would have been no reduction in AFib had it been objectively documented with the handheld ECG device, he said in an interview.
“If I were to redesign the study,” he said, “I think the primary endpoint should be confirmed atrial fibrillation, because we would have to show first that the specific trigger actually reduced objective AFib events before we then try to address the question whether reducing that trigger improves quality of life.”
Unrepresentative sample
The trial entered 446 overwhelmingly White and college-educated adults known to have symptomatic paroxysmal AFib who were “interested in testing a presumed AFib trigger they could readily introduce or withhold” and who owned a smartphone; the average age was 58 years, and 58% were men. The cohort was randomly assigned to the trigger-testing group or the control group, charged only with tracking their AFib.
Of the total, 320, or about 72%, completed the study; those who did not were mostly from the trigger-testing arm, leaving 136 in that group versus 184 patients in the control group.
Potential triggers that participants selected for tracking included, foremost, caffeine, alcohol, reduced sleep, and exercise, followed by lying on the left side, dehydration, large meals, and cold food or drink, the report noted.
Patients in the control group used the smartphone app and handheld ECG monitor (KardiaMobile, AliveCor) to document the duration and severity of AFib episodes daily and received data summary reports through the app weekly for 10 weeks. They then had the option to follow the trigger-testing protocol at least once.
Those in the trigger-testing group conducted their N-of-1 trials by exposing themselves to their chosen potential trigger during 3 separate weeks and avoiding the trigger during 3 other weeks, alternating each of the 6 weeks of trigger exposure or avoidance. They were instructed through the app to start the 6-week sequence with one or the other strategy randomly and to regularly track their AFib.
At the end of 6 weeks, each participant in the trigger-testing group had the opportunity to review their data for any potential trigger-AFib associations. They were then to use the next 4 weeks to enact lifestyle changes based on what they learned – as described in the report and on clinicaltrials.gov. They had the option of repeating the entire N-of-1 sequence at least one more time.
Participants in both the trigger-tracking and control arms were tested at baseline and at 10 weeks using the validated Atrial Fibrillation Effect on Quality-of-Life (AFEQT) questionnaire.
AFEQT scores didn’t change significantly over the 10 weeks in either arm, nor were they significantly different in one arm, compared with the other.
On the other hand, patients in the trigger-tracking arm reported significantly fewer daily AFib episodes during the final 4-week period of lifestyle changes based on their N-of-1 trial results, compared to the monitoring-only control group’s final 4 weeks.
The adjusted relative risk in the trigger-tracking arm was 0.60 (95% confidence interval, 0.43-0.83; P < .001), the difference driven by patients who selected alcohol, dehydration, or exercise for their trigger, Dr. Marcus reported.
Only alcohol intake emerged consistently as a significant predictor of risk for self-reported AFib episodes in a series of meta-analyses conducted using all of the individual N-of-1 trials that provided per-protocol data. The odds ratio was 1.77 (95% CI, 1.20-2.69).
I-STOP-AFib explored an important subject “that has been understudied,” Dr. Conen said. “The trial has some limitations that the authors address themselves, but hopefully it opens the path to future studies that can build upon this experience.”
Dr. Marcus reported receiving personal fees and equity interest from InCarda Therapeutics; personal fees from Johnson & Johnson; and grants from Baylis Medical, Medtronic, the National Institutes of Health, the Patient-Centered Outcomes Research Institute, and the California Tobacco-Related Disease Research Program.
A version of this article first appeared on Medscape.com.
Pandemic stresses harder on physician moms than physician dads: Study
COVID-19 has been difficult for parents trying to balance careers, home life, and keeping their loved ones safe. A new study indicates that, not only are physicians not immune to these stressors, but the long-term effects could be devastating for health care overall.
In a study published Nov. 11, 2021, in JAMA Network Open , researchers found that stresses to work/life balance and family life caused by the pandemic have differed among men and women physicians.
Physicians and other health care workers have been at the front lines of the COVID-19 pandemic, and their work lives have been the focus of a lot of attention in the media and by researchers. Their family lives, not so much. But physicians have families, and the pandemic has upended almost everything about their lives, particularly where work life and home life intersect. School and day care closures, working from home, working extra hours, or working less – all of these changes have consequences on family life and the mental health of parents who are also physicians.
Findings from a Medscape survey published in early 2021 indicate that more female physicians than male physicians were either “conflicted” or “very conflicted” as parents because of work demands (42% vs. 23%) nearly 6 months into the pandemic.
In the current study, researchers from the University of Michigan, Harvard University, and the Medical University of South Carolina teamed up to investigate gender differences in how work/family factors affected the mental health of early-career physician parents in the United States during the first year of the COVID-19 pandemic. The results suggest that the pandemic has increased gender disparity and added disproportionately to the burden of female physicians.
Managing the household falls mostly on moms
Participants were physicians enrolled in the Intern Health Study, a longitudinal study that regularly surveys medical interns in the United States to assess stress and mood. When researchers compared survey results from before the onset of the pandemic (2018) with later results (2020), they found a striking gender difference in how the pandemic has changed family and work duties for physicians.
The authors of the study pointed out that previous research had found that female physicians take on a greater share of household and childcare duties than male physicians. The current study found that their share had increased with the pandemic. Physician moms are now 30 times more likely to be in charge of these tasks than physician dads.
In families in which both parents were physicians, none of the men said they took the primary role in managing the extra demands caused by the pandemic. In addition, women were twice as likely as men to work primarily from home and to work reduced hours.
The extra stress seems to be taking a toll on women physicians. In the 2020 survey, physician mothers had higher scores for anxiety and depression symptoms, compared with men. Notably, the 2018 survey did not show a significant difference in depression scores between men and women. Nor were there significant differences in depression and anxiety scores between women and men who were not parents or in reports of work/family conflict before and after the pandemic.
In general, the results indicate that the pandemic has only widened the gender gap between women and men physicians when it comes to managing family life and dealing with the stresses of maintaining a suitable work-life balance.
‘Long-term repercussions’ for gender equity in medicine
Although these are serious problems for women physicians and their families, the effects go beyond the home and beyond individuals. Even before the pandemic, women in medicine struggled for parity in career advancement and opportunities as well as in pay, and this new setback could make those challenges even greater.
“Even short-term adjustments can have serious long-term repercussions as they may lead to lower earnings and negatively impact opportunities for promotion, further exacerbating gender inequalities in compensation and advancement,” the study’s authors wrote.
The potential damage extends to the entire profession and the health care system itself. The profession is already struggling to retain young female physicians, and this situation is likely to make that problem worse and have long-term consequences. Citing data showing that female physicians spend more time with patients and that their patients may have better outcomes, the authors wrote that the consequences of losing more early-career female physicians “could be devastating to the U.S. health care system, particularly in the context of a global pandemic and an impending physician shortage.”
The sample size was small (276 U.S. physicians), and the study relied on self-reported data. The findings suggest that more research on this topic is needed, especially research that includes other demographic factors, such as sexual orientation and ethnicity. The authors recommend that institutional and public policymakers take into account the effects of the pandemic on physician mothers to ensure that recent gains in gender equity for women physicians do not fall victim to COVID-19.
A version of this article first appeared on Medscape.com.
COVID-19 has been difficult for parents trying to balance careers, home life, and keeping their loved ones safe. A new study indicates that, not only are physicians not immune to these stressors, but the long-term effects could be devastating for health care overall.
In a study published Nov. 11, 2021, in JAMA Network Open , researchers found that stresses to work/life balance and family life caused by the pandemic have differed among men and women physicians.
Physicians and other health care workers have been at the front lines of the COVID-19 pandemic, and their work lives have been the focus of a lot of attention in the media and by researchers. Their family lives, not so much. But physicians have families, and the pandemic has upended almost everything about their lives, particularly where work life and home life intersect. School and day care closures, working from home, working extra hours, or working less – all of these changes have consequences on family life and the mental health of parents who are also physicians.
Findings from a Medscape survey published in early 2021 indicate that more female physicians than male physicians were either “conflicted” or “very conflicted” as parents because of work demands (42% vs. 23%) nearly 6 months into the pandemic.
In the current study, researchers from the University of Michigan, Harvard University, and the Medical University of South Carolina teamed up to investigate gender differences in how work/family factors affected the mental health of early-career physician parents in the United States during the first year of the COVID-19 pandemic. The results suggest that the pandemic has increased gender disparity and added disproportionately to the burden of female physicians.
Managing the household falls mostly on moms
Participants were physicians enrolled in the Intern Health Study, a longitudinal study that regularly surveys medical interns in the United States to assess stress and mood. When researchers compared survey results from before the onset of the pandemic (2018) with later results (2020), they found a striking gender difference in how the pandemic has changed family and work duties for physicians.
The authors of the study pointed out that previous research had found that female physicians take on a greater share of household and childcare duties than male physicians. The current study found that their share had increased with the pandemic. Physician moms are now 30 times more likely to be in charge of these tasks than physician dads.
In families in which both parents were physicians, none of the men said they took the primary role in managing the extra demands caused by the pandemic. In addition, women were twice as likely as men to work primarily from home and to work reduced hours.
The extra stress seems to be taking a toll on women physicians. In the 2020 survey, physician mothers had higher scores for anxiety and depression symptoms, compared with men. Notably, the 2018 survey did not show a significant difference in depression scores between men and women. Nor were there significant differences in depression and anxiety scores between women and men who were not parents or in reports of work/family conflict before and after the pandemic.
In general, the results indicate that the pandemic has only widened the gender gap between women and men physicians when it comes to managing family life and dealing with the stresses of maintaining a suitable work-life balance.
‘Long-term repercussions’ for gender equity in medicine
Although these are serious problems for women physicians and their families, the effects go beyond the home and beyond individuals. Even before the pandemic, women in medicine struggled for parity in career advancement and opportunities as well as in pay, and this new setback could make those challenges even greater.
“Even short-term adjustments can have serious long-term repercussions as they may lead to lower earnings and negatively impact opportunities for promotion, further exacerbating gender inequalities in compensation and advancement,” the study’s authors wrote.
The potential damage extends to the entire profession and the health care system itself. The profession is already struggling to retain young female physicians, and this situation is likely to make that problem worse and have long-term consequences. Citing data showing that female physicians spend more time with patients and that their patients may have better outcomes, the authors wrote that the consequences of losing more early-career female physicians “could be devastating to the U.S. health care system, particularly in the context of a global pandemic and an impending physician shortage.”
The sample size was small (276 U.S. physicians), and the study relied on self-reported data. The findings suggest that more research on this topic is needed, especially research that includes other demographic factors, such as sexual orientation and ethnicity. The authors recommend that institutional and public policymakers take into account the effects of the pandemic on physician mothers to ensure that recent gains in gender equity for women physicians do not fall victim to COVID-19.
A version of this article first appeared on Medscape.com.
COVID-19 has been difficult for parents trying to balance careers, home life, and keeping their loved ones safe. A new study indicates that, not only are physicians not immune to these stressors, but the long-term effects could be devastating for health care overall.
In a study published Nov. 11, 2021, in JAMA Network Open , researchers found that stresses to work/life balance and family life caused by the pandemic have differed among men and women physicians.
Physicians and other health care workers have been at the front lines of the COVID-19 pandemic, and their work lives have been the focus of a lot of attention in the media and by researchers. Their family lives, not so much. But physicians have families, and the pandemic has upended almost everything about their lives, particularly where work life and home life intersect. School and day care closures, working from home, working extra hours, or working less – all of these changes have consequences on family life and the mental health of parents who are also physicians.
Findings from a Medscape survey published in early 2021 indicate that more female physicians than male physicians were either “conflicted” or “very conflicted” as parents because of work demands (42% vs. 23%) nearly 6 months into the pandemic.
In the current study, researchers from the University of Michigan, Harvard University, and the Medical University of South Carolina teamed up to investigate gender differences in how work/family factors affected the mental health of early-career physician parents in the United States during the first year of the COVID-19 pandemic. The results suggest that the pandemic has increased gender disparity and added disproportionately to the burden of female physicians.
Managing the household falls mostly on moms
Participants were physicians enrolled in the Intern Health Study, a longitudinal study that regularly surveys medical interns in the United States to assess stress and mood. When researchers compared survey results from before the onset of the pandemic (2018) with later results (2020), they found a striking gender difference in how the pandemic has changed family and work duties for physicians.
The authors of the study pointed out that previous research had found that female physicians take on a greater share of household and childcare duties than male physicians. The current study found that their share had increased with the pandemic. Physician moms are now 30 times more likely to be in charge of these tasks than physician dads.
In families in which both parents were physicians, none of the men said they took the primary role in managing the extra demands caused by the pandemic. In addition, women were twice as likely as men to work primarily from home and to work reduced hours.
The extra stress seems to be taking a toll on women physicians. In the 2020 survey, physician mothers had higher scores for anxiety and depression symptoms, compared with men. Notably, the 2018 survey did not show a significant difference in depression scores between men and women. Nor were there significant differences in depression and anxiety scores between women and men who were not parents or in reports of work/family conflict before and after the pandemic.
In general, the results indicate that the pandemic has only widened the gender gap between women and men physicians when it comes to managing family life and dealing with the stresses of maintaining a suitable work-life balance.
‘Long-term repercussions’ for gender equity in medicine
Although these are serious problems for women physicians and their families, the effects go beyond the home and beyond individuals. Even before the pandemic, women in medicine struggled for parity in career advancement and opportunities as well as in pay, and this new setback could make those challenges even greater.
“Even short-term adjustments can have serious long-term repercussions as they may lead to lower earnings and negatively impact opportunities for promotion, further exacerbating gender inequalities in compensation and advancement,” the study’s authors wrote.
The potential damage extends to the entire profession and the health care system itself. The profession is already struggling to retain young female physicians, and this situation is likely to make that problem worse and have long-term consequences. Citing data showing that female physicians spend more time with patients and that their patients may have better outcomes, the authors wrote that the consequences of losing more early-career female physicians “could be devastating to the U.S. health care system, particularly in the context of a global pandemic and an impending physician shortage.”
The sample size was small (276 U.S. physicians), and the study relied on self-reported data. The findings suggest that more research on this topic is needed, especially research that includes other demographic factors, such as sexual orientation and ethnicity. The authors recommend that institutional and public policymakers take into account the effects of the pandemic on physician mothers to ensure that recent gains in gender equity for women physicians do not fall victim to COVID-19.
A version of this article first appeared on Medscape.com.
COVID-19 vaccine mandates are working, public health experts say
Some organizations have reported vaccination rates that jumped from less than 50% to more than 90%, according to ABC News. Workplace mandates have especially encouraged employees who were on the fence to get a shot.
“In general, vaccine mandates work,” James Colgrove, a public health professor at Columbia University’s Mailman School of Public Health, told ABC News.
For decades, the United States has monitored the effectiveness of vaccine mandates in schools, he noted, which have successfully required shots against measles, mumps, and other illnesses that used to be widespread. Certain employees, such as hospital workers, must take vaccines for their jobs, he said, and those requirements have also been effective over the years.
“The more normalized it becomes, the more people [know] someone else who is vaccinated, the more people will comply,” he said. “With any vaccine, the longer it’s been around, the more people get with it.”
With the widespread and contagious nature of COVID-19, workplaces have been forced to consider vaccine mandates to protect their employees and prevent worker shortages, Dr. Colgrove said.
Some companies began to issue vaccine rules this summer as the Delta variant caused a jump in cases, hospitalizations, and deaths. Major companies, including Google, Tyson Foods, United Airlines, and the Walt Disney Company, required in-person employees to get a shot. So far, the results from those mandates have been strong, ABC News reported.
For instance, Tyson announced a mandate in August, when less than half of its 140,000 employees were vaccinated. When the deadline came at the end of October, more than 60,000 additional employees had been vaccinated, and the vaccination rate was 96%.
“Has this made a difference in the health and safety of our team members? Absolutely. We’ve seen a significant decline in the number of active cases companywide,” Donnie King, CEO and president of Tyson Foods, said in a statement.
United Airlines has also shared that 99.7% of its 67,000 employees are vaccinated. Within 48 hours of announcing its mandate, the number of unvaccinated staffers fell from 593 to 320 people, ABC News reported.
Vaccine mandates appear to be working in the public sector as well. State health department officials in Washington told ABC News that the percentage of public employees who were vaccinated jumped from 49% in September to 96% by the vaccine mandate deadline in October.
Vaccination rates have also increased in New York City, where some employees in the fire, police, and sanitation departments protested the mandate. By the deadline, vaccination rates shifted from less than 75% to 82% in the fire department, 86% in the police department, and 91% of EMS personnel, ABC News reported.
Overall, vaccine mandates tend to reach groups who aren’t completely against the vaccine, medical experts told the news outlet. A small percentage of the population truly opposes the shot, and in most cases, unvaccinated people are on the fence or haven’t seen good enough messaging for it.
“When you look at vaccine resistance, the people who are the most opposed often make a very large amount of noise that is at odds with the actual numbers who are against vaccination,” Dr. Colgrove said.
A version of this article first appeared on WebMD.com.
Some organizations have reported vaccination rates that jumped from less than 50% to more than 90%, according to ABC News. Workplace mandates have especially encouraged employees who were on the fence to get a shot.
“In general, vaccine mandates work,” James Colgrove, a public health professor at Columbia University’s Mailman School of Public Health, told ABC News.
For decades, the United States has monitored the effectiveness of vaccine mandates in schools, he noted, which have successfully required shots against measles, mumps, and other illnesses that used to be widespread. Certain employees, such as hospital workers, must take vaccines for their jobs, he said, and those requirements have also been effective over the years.
“The more normalized it becomes, the more people [know] someone else who is vaccinated, the more people will comply,” he said. “With any vaccine, the longer it’s been around, the more people get with it.”
With the widespread and contagious nature of COVID-19, workplaces have been forced to consider vaccine mandates to protect their employees and prevent worker shortages, Dr. Colgrove said.
Some companies began to issue vaccine rules this summer as the Delta variant caused a jump in cases, hospitalizations, and deaths. Major companies, including Google, Tyson Foods, United Airlines, and the Walt Disney Company, required in-person employees to get a shot. So far, the results from those mandates have been strong, ABC News reported.
For instance, Tyson announced a mandate in August, when less than half of its 140,000 employees were vaccinated. When the deadline came at the end of October, more than 60,000 additional employees had been vaccinated, and the vaccination rate was 96%.
“Has this made a difference in the health and safety of our team members? Absolutely. We’ve seen a significant decline in the number of active cases companywide,” Donnie King, CEO and president of Tyson Foods, said in a statement.
United Airlines has also shared that 99.7% of its 67,000 employees are vaccinated. Within 48 hours of announcing its mandate, the number of unvaccinated staffers fell from 593 to 320 people, ABC News reported.
Vaccine mandates appear to be working in the public sector as well. State health department officials in Washington told ABC News that the percentage of public employees who were vaccinated jumped from 49% in September to 96% by the vaccine mandate deadline in October.
Vaccination rates have also increased in New York City, where some employees in the fire, police, and sanitation departments protested the mandate. By the deadline, vaccination rates shifted from less than 75% to 82% in the fire department, 86% in the police department, and 91% of EMS personnel, ABC News reported.
Overall, vaccine mandates tend to reach groups who aren’t completely against the vaccine, medical experts told the news outlet. A small percentage of the population truly opposes the shot, and in most cases, unvaccinated people are on the fence or haven’t seen good enough messaging for it.
“When you look at vaccine resistance, the people who are the most opposed often make a very large amount of noise that is at odds with the actual numbers who are against vaccination,” Dr. Colgrove said.
A version of this article first appeared on WebMD.com.
Some organizations have reported vaccination rates that jumped from less than 50% to more than 90%, according to ABC News. Workplace mandates have especially encouraged employees who were on the fence to get a shot.
“In general, vaccine mandates work,” James Colgrove, a public health professor at Columbia University’s Mailman School of Public Health, told ABC News.
For decades, the United States has monitored the effectiveness of vaccine mandates in schools, he noted, which have successfully required shots against measles, mumps, and other illnesses that used to be widespread. Certain employees, such as hospital workers, must take vaccines for their jobs, he said, and those requirements have also been effective over the years.
“The more normalized it becomes, the more people [know] someone else who is vaccinated, the more people will comply,” he said. “With any vaccine, the longer it’s been around, the more people get with it.”
With the widespread and contagious nature of COVID-19, workplaces have been forced to consider vaccine mandates to protect their employees and prevent worker shortages, Dr. Colgrove said.
Some companies began to issue vaccine rules this summer as the Delta variant caused a jump in cases, hospitalizations, and deaths. Major companies, including Google, Tyson Foods, United Airlines, and the Walt Disney Company, required in-person employees to get a shot. So far, the results from those mandates have been strong, ABC News reported.
For instance, Tyson announced a mandate in August, when less than half of its 140,000 employees were vaccinated. When the deadline came at the end of October, more than 60,000 additional employees had been vaccinated, and the vaccination rate was 96%.
“Has this made a difference in the health and safety of our team members? Absolutely. We’ve seen a significant decline in the number of active cases companywide,” Donnie King, CEO and president of Tyson Foods, said in a statement.
United Airlines has also shared that 99.7% of its 67,000 employees are vaccinated. Within 48 hours of announcing its mandate, the number of unvaccinated staffers fell from 593 to 320 people, ABC News reported.
Vaccine mandates appear to be working in the public sector as well. State health department officials in Washington told ABC News that the percentage of public employees who were vaccinated jumped from 49% in September to 96% by the vaccine mandate deadline in October.
Vaccination rates have also increased in New York City, where some employees in the fire, police, and sanitation departments protested the mandate. By the deadline, vaccination rates shifted from less than 75% to 82% in the fire department, 86% in the police department, and 91% of EMS personnel, ABC News reported.
Overall, vaccine mandates tend to reach groups who aren’t completely against the vaccine, medical experts told the news outlet. A small percentage of the population truly opposes the shot, and in most cases, unvaccinated people are on the fence or haven’t seen good enough messaging for it.
“When you look at vaccine resistance, the people who are the most opposed often make a very large amount of noise that is at odds with the actual numbers who are against vaccination,” Dr. Colgrove said.
A version of this article first appeared on WebMD.com.
Whistleblowers will play key role in enforcing workplace vaccine mandate
goes into effect in January.
The Occupational Safety and Health Administration (OSHA) doesn’t have enough workplace safety inspectors to cover the nation, the Associated Press reported, so the agency will count on people within organizations to identify violations.
“There is no army of OSHA inspectors that is going to be knocking on employers’ doors or even calling them,” Debbie Berkowitz, a former OSHA chief of staff who is a fellow at Georgetown University, told the news service.
“They’re going to rely on workers and their union representatives to file complaints where the company is totally flouting the law,” she said.
Last week, OSHA published the details of the Biden administration’s vaccine mandate. Companies with more than 100 employees must require their workers to get vaccinated or undergo weekly testing. Companies that don’t comply could face fines of $14,000 for each “serious” violation. Repeat violators could face 10 times that amount.
Employees who are concerned about workplace safety, unvaccinated co-workers, or people not being tested as required may report their employers, according to Reuters.
Jim Frederick, the acting chief for OSHA, told reporters that the agency will focus on job sites “where workers need assistance to have a safe and healthy workplace.”
“That typically comes through in the form of a complaint,” he said.
OSHA has jurisdiction in 29 states, the AP reported. OSHA is tasked with addressing violations of the Occupational Safety and Health Act of 1970, which is meant to create safe workplaces, and the agency has updated its guidance about COVID-19 safety in the workplace throughout this year.
Other states, such as California and Michigan, have their own workplace safety agencies, which will have until February to adopt their own version of a vaccine mandate, according to the AP.
OSHA and state counterparts will be tasked with enforcing the mandate, and their agencies are already short-staffed. About 1,850 inspectors will oversee 130 million workers at 8 million job sites.
OSHA has encouraged workers to first report complaints to employers “if possible.” Otherwise, employees can file a confidential safety complaint with OSHA or file a case through a representative, such as a lawyer or union leader, the AP reported.
But workplace experts have voiced caution about the potential risks of reporting. Whistleblowers tend to face retaliation and OSHA can’t always offer protection in these cases.
“Technically, the law says that companies can’t retaliate against a worker for raising a health and safety issue or filing an OSHA complaint or even reporting an injury,” Ms. Berkowitz said. “But retaliation is rampant.”
OSHA has some jurisdiction to pursue employers who punish workers for reporting unsafe working conditions, the AP reported. Last month, the agency sued a luxury car dealer in Texas for firing an employee who warned co-workers about potential coronavirus hazards.
But at the same time, Ms. Berkowitz and the National Employment Law Project found that OSHA dismissed more than half of the COVID-related complaints of retaliation that it received from whistleblowers. About 2% of complaints were resolved during a 5-month period last year, according to their report.
As the vaccine mandate deadline approaches, most companies are expected to comply, experts told the AP. Some employers wanted to require the shot but didn’t want to create their own rule, and others have said they’ll follow OSHA regulations as they always do.
“Most employers, they’re law abiding,” David Michaels, a former OSHA chief who is a public health professor at George Washington University, told the AP.
“They’re trying to make sure that they meet the requirements of every law and regulation,” he said. “Now OSHA will follow up. They’ll respond to complaints. They’ll do spot checks. They’ll issue citations and fines, and they’ll make a big deal of those.”
A version of this article first appeared on WebMD.com.
goes into effect in January.
The Occupational Safety and Health Administration (OSHA) doesn’t have enough workplace safety inspectors to cover the nation, the Associated Press reported, so the agency will count on people within organizations to identify violations.
“There is no army of OSHA inspectors that is going to be knocking on employers’ doors or even calling them,” Debbie Berkowitz, a former OSHA chief of staff who is a fellow at Georgetown University, told the news service.
“They’re going to rely on workers and their union representatives to file complaints where the company is totally flouting the law,” she said.
Last week, OSHA published the details of the Biden administration’s vaccine mandate. Companies with more than 100 employees must require their workers to get vaccinated or undergo weekly testing. Companies that don’t comply could face fines of $14,000 for each “serious” violation. Repeat violators could face 10 times that amount.
Employees who are concerned about workplace safety, unvaccinated co-workers, or people not being tested as required may report their employers, according to Reuters.
Jim Frederick, the acting chief for OSHA, told reporters that the agency will focus on job sites “where workers need assistance to have a safe and healthy workplace.”
“That typically comes through in the form of a complaint,” he said.
OSHA has jurisdiction in 29 states, the AP reported. OSHA is tasked with addressing violations of the Occupational Safety and Health Act of 1970, which is meant to create safe workplaces, and the agency has updated its guidance about COVID-19 safety in the workplace throughout this year.
Other states, such as California and Michigan, have their own workplace safety agencies, which will have until February to adopt their own version of a vaccine mandate, according to the AP.
OSHA and state counterparts will be tasked with enforcing the mandate, and their agencies are already short-staffed. About 1,850 inspectors will oversee 130 million workers at 8 million job sites.
OSHA has encouraged workers to first report complaints to employers “if possible.” Otherwise, employees can file a confidential safety complaint with OSHA or file a case through a representative, such as a lawyer or union leader, the AP reported.
But workplace experts have voiced caution about the potential risks of reporting. Whistleblowers tend to face retaliation and OSHA can’t always offer protection in these cases.
“Technically, the law says that companies can’t retaliate against a worker for raising a health and safety issue or filing an OSHA complaint or even reporting an injury,” Ms. Berkowitz said. “But retaliation is rampant.”
OSHA has some jurisdiction to pursue employers who punish workers for reporting unsafe working conditions, the AP reported. Last month, the agency sued a luxury car dealer in Texas for firing an employee who warned co-workers about potential coronavirus hazards.
But at the same time, Ms. Berkowitz and the National Employment Law Project found that OSHA dismissed more than half of the COVID-related complaints of retaliation that it received from whistleblowers. About 2% of complaints were resolved during a 5-month period last year, according to their report.
As the vaccine mandate deadline approaches, most companies are expected to comply, experts told the AP. Some employers wanted to require the shot but didn’t want to create their own rule, and others have said they’ll follow OSHA regulations as they always do.
“Most employers, they’re law abiding,” David Michaels, a former OSHA chief who is a public health professor at George Washington University, told the AP.
“They’re trying to make sure that they meet the requirements of every law and regulation,” he said. “Now OSHA will follow up. They’ll respond to complaints. They’ll do spot checks. They’ll issue citations and fines, and they’ll make a big deal of those.”
A version of this article first appeared on WebMD.com.
goes into effect in January.
The Occupational Safety and Health Administration (OSHA) doesn’t have enough workplace safety inspectors to cover the nation, the Associated Press reported, so the agency will count on people within organizations to identify violations.
“There is no army of OSHA inspectors that is going to be knocking on employers’ doors or even calling them,” Debbie Berkowitz, a former OSHA chief of staff who is a fellow at Georgetown University, told the news service.
“They’re going to rely on workers and their union representatives to file complaints where the company is totally flouting the law,” she said.
Last week, OSHA published the details of the Biden administration’s vaccine mandate. Companies with more than 100 employees must require their workers to get vaccinated or undergo weekly testing. Companies that don’t comply could face fines of $14,000 for each “serious” violation. Repeat violators could face 10 times that amount.
Employees who are concerned about workplace safety, unvaccinated co-workers, or people not being tested as required may report their employers, according to Reuters.
Jim Frederick, the acting chief for OSHA, told reporters that the agency will focus on job sites “where workers need assistance to have a safe and healthy workplace.”
“That typically comes through in the form of a complaint,” he said.
OSHA has jurisdiction in 29 states, the AP reported. OSHA is tasked with addressing violations of the Occupational Safety and Health Act of 1970, which is meant to create safe workplaces, and the agency has updated its guidance about COVID-19 safety in the workplace throughout this year.
Other states, such as California and Michigan, have their own workplace safety agencies, which will have until February to adopt their own version of a vaccine mandate, according to the AP.
OSHA and state counterparts will be tasked with enforcing the mandate, and their agencies are already short-staffed. About 1,850 inspectors will oversee 130 million workers at 8 million job sites.
OSHA has encouraged workers to first report complaints to employers “if possible.” Otherwise, employees can file a confidential safety complaint with OSHA or file a case through a representative, such as a lawyer or union leader, the AP reported.
But workplace experts have voiced caution about the potential risks of reporting. Whistleblowers tend to face retaliation and OSHA can’t always offer protection in these cases.
“Technically, the law says that companies can’t retaliate against a worker for raising a health and safety issue or filing an OSHA complaint or even reporting an injury,” Ms. Berkowitz said. “But retaliation is rampant.”
OSHA has some jurisdiction to pursue employers who punish workers for reporting unsafe working conditions, the AP reported. Last month, the agency sued a luxury car dealer in Texas for firing an employee who warned co-workers about potential coronavirus hazards.
But at the same time, Ms. Berkowitz and the National Employment Law Project found that OSHA dismissed more than half of the COVID-related complaints of retaliation that it received from whistleblowers. About 2% of complaints were resolved during a 5-month period last year, according to their report.
As the vaccine mandate deadline approaches, most companies are expected to comply, experts told the AP. Some employers wanted to require the shot but didn’t want to create their own rule, and others have said they’ll follow OSHA regulations as they always do.
“Most employers, they’re law abiding,” David Michaels, a former OSHA chief who is a public health professor at George Washington University, told the AP.
“They’re trying to make sure that they meet the requirements of every law and regulation,” he said. “Now OSHA will follow up. They’ll respond to complaints. They’ll do spot checks. They’ll issue citations and fines, and they’ll make a big deal of those.”
A version of this article first appeared on WebMD.com.
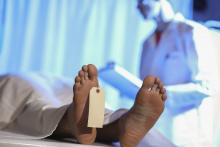
Step right up, folks, for a public dissection
The greatest autopsy on Earth?
The LOTME staff would like to apologize in advance. The following item contains historical facts.
P.T. Barnum is a rather controversial figure in American history. The greatest show on Earth was certainly popular in its day. However, Barnum got his start in 1835 by leasing a slave named Joyce Heth, an elderly Black woman who told vivid stories of caring for a young George Washington. He toured her around the country, advertising her as a 160-year-old woman who served as George Washington’s nanny. When Ms. Heth died the next year, Barnum sold tickets to the autopsy, charging the equivalent of $30 in today’s money.
When a doctor announced that Ms. Heth was actually 75-80 when she died, it caused great controversy in the press and ruined Barnum’s career. Wait, no, that’s not right. The opposite, actually. He weathered the storm, built his famous circus, and never again committed a hoax.
It’s difficult to quantify how wrong publicly dissecting a person and charging people to see said dissection is, but that was almost 200 years ago. At the very least, we can say that such terrible behavior is firmly in the distant past.
Oh wait.
David Saunders, a 98-year-old veteran of World War II and the Korean War, donated his body to science. His body, however, was purchased by DeathScience.org from a medical lab – with the buyer supposedly misleading the medical lab about its intentions, which was for use at the traveling Oddities and Curiosities Expo. Tickets went for up to $500 each to witness the public autopsy of Mr. Saunders’ body, which took place at a Marriott in Portland, Ore. It promised to be an exciting, all-day event from 9 a.m. to 4 p.m., with a break for lunch, of course. You can’t have an autopsy without a catered lunch.
Another public autopsy event was scheduled in Seattle but canceled after news of the first event broke. Oh, and for that extra little kick, Mr. Saunders died from COVID-19, meaning that all those paying customers were exposed.
P.T. Barnum is probably rolling over in his grave right now. His autopsy tickets were a bargain.
Go ahead, have that soda before math
We should all know by now that sugary drinks are bad, even artificially sweetened ones. It might not always stop us from drinking them, but we know the deal. But what if sugary drinks like soda could be helpful for girls in school?
You read that right. We said girls. A soda before class might have boys bouncing off the walls, but not girls. A recent study showed that not only was girls’ behavior unaffected by having a sugary drink, their math skills even improved.
Researchers analyzed the behavior of 4- to 6-year-old children before and after having a sugary drink. The sugar rush was actually calming for girls and helped them perform better with numerical skills, but the opposite was true for boys. “Our study is the first to provide large-scale experimental evidence on the impact of sugary drinks on preschool children. The results clearly indicate a causal impact of sugary drinks on children’s behavior and test scores,” Fritz Schiltz, PhD, said in a written statement.
This probably isn’t the green light to have as many sugary drinks as you want, but it might be interesting to see how your work is affected after a soda.
Chicken nuggets and the meat paradox
Two young children are fighting over the last chicken nugget when an adult comes in to see what’s going on.
Liam: Vegetable!
Olivia: Meat!
Liam: Chicken nuggets are vegetables!
Olivia: No, dorkface! They’re meat.
Caregiver: Good news, kids. You’re both right.
Olivia: How can we both be right?
At this point, a woman enters the room. She’s wearing a white lab coat, so she must be a scientist.
Dr. Scientist: You can’t both be right, Olivia. You are being fed a serving of the meat paradox. That’s why Liam here doesn’t know that chicken nuggets are made of chicken, which is a form of meat. Sadly, he’s not the only one.
In a recent study, scientists from Furman University in Greenville, S.C., found that 38% of 176 children aged 4-7 years thought that chicken nuggets were vegetables and more than 46% identified French fries as animal based.
Olivia: Did our caregiver lie to us, Dr. Scientist?
Dr. Scientist: Yes, Olivia. The researchers I mentioned explained that “many people experience unease while eating meat. Omnivores eat foods that entail animal suffering and death while at the same time endorsing the compassionate treatment of animals.” That’s the meat paradox.
Liam: What else did they say, Dr. Scientist?
Dr. Scientist: Over 70% of those children said that cows and pigs were not edible and 5% thought that cats and horses were. The investigators wrote “that children and youth should be viewed as agents of environmental change” in the future, but suggested that parents need to bring honesty to the table.
Caregiver: How did you get in here anyway? And how do you know their names?
Dr. Scientist: I’ve been rooting through your garbage for years. All in the name of science, of course.
Bedtimes aren’t just for children
There are multiple ways to prevent heart disease, but what if it could be as easy as switching your bedtime? A recent study in European Heart Journal–Digital Health suggests that there’s a sweet spot when it comes to sleep timing.
Through smartwatch-like devices, researchers measured the sleep-onset and wake-up times for 7 days in 88,026 participants aged 43-79 years. After 5.7 years of follow-up to see if anyone had a heart attack, stroke, or any other cardiovascular event, 3.6% developed some kind of cardiovascular disease.
Those who went to bed between 10 p.m. and 11 p.m. had a lower risk of developing heart disease. The risk was 25% higher for subjects who went to bed at midnight or later, 24% higher for bedtimes before 10 p.m., and 12% higher for bedtimes between 11 p.m. and midnight.
So, why can you go to bed before “The Tonight Show” and lower your cardiovascular risk but not before the nightly news? Well, it has something to do with your body’s natural clock.
“The optimum time to go to sleep is at a specific point in the body’s 24-hour cycle and deviations may be detrimental to health. The riskiest time was after midnight, potentially because it may reduce the likelihood of seeing morning light, which resets the body clock,” said study author Dr. David Plans of the University of Exeter, England.
Although a sleep schedule is preferred, it isn’t realistic all the time for those in certain occupations who might have to resort to other methods to keep their circadian clocks ticking optimally for their health. But if all it takes is prescribing a sleep time to reduce heart disease on a massive scale it would make a great “low-cost public health target.”
So bedtimes aren’t just for children.
The greatest autopsy on Earth?
The LOTME staff would like to apologize in advance. The following item contains historical facts.
P.T. Barnum is a rather controversial figure in American history. The greatest show on Earth was certainly popular in its day. However, Barnum got his start in 1835 by leasing a slave named Joyce Heth, an elderly Black woman who told vivid stories of caring for a young George Washington. He toured her around the country, advertising her as a 160-year-old woman who served as George Washington’s nanny. When Ms. Heth died the next year, Barnum sold tickets to the autopsy, charging the equivalent of $30 in today’s money.
When a doctor announced that Ms. Heth was actually 75-80 when she died, it caused great controversy in the press and ruined Barnum’s career. Wait, no, that’s not right. The opposite, actually. He weathered the storm, built his famous circus, and never again committed a hoax.
It’s difficult to quantify how wrong publicly dissecting a person and charging people to see said dissection is, but that was almost 200 years ago. At the very least, we can say that such terrible behavior is firmly in the distant past.
Oh wait.
David Saunders, a 98-year-old veteran of World War II and the Korean War, donated his body to science. His body, however, was purchased by DeathScience.org from a medical lab – with the buyer supposedly misleading the medical lab about its intentions, which was for use at the traveling Oddities and Curiosities Expo. Tickets went for up to $500 each to witness the public autopsy of Mr. Saunders’ body, which took place at a Marriott in Portland, Ore. It promised to be an exciting, all-day event from 9 a.m. to 4 p.m., with a break for lunch, of course. You can’t have an autopsy without a catered lunch.
Another public autopsy event was scheduled in Seattle but canceled after news of the first event broke. Oh, and for that extra little kick, Mr. Saunders died from COVID-19, meaning that all those paying customers were exposed.
P.T. Barnum is probably rolling over in his grave right now. His autopsy tickets were a bargain.
Go ahead, have that soda before math
We should all know by now that sugary drinks are bad, even artificially sweetened ones. It might not always stop us from drinking them, but we know the deal. But what if sugary drinks like soda could be helpful for girls in school?
You read that right. We said girls. A soda before class might have boys bouncing off the walls, but not girls. A recent study showed that not only was girls’ behavior unaffected by having a sugary drink, their math skills even improved.
Researchers analyzed the behavior of 4- to 6-year-old children before and after having a sugary drink. The sugar rush was actually calming for girls and helped them perform better with numerical skills, but the opposite was true for boys. “Our study is the first to provide large-scale experimental evidence on the impact of sugary drinks on preschool children. The results clearly indicate a causal impact of sugary drinks on children’s behavior and test scores,” Fritz Schiltz, PhD, said in a written statement.
This probably isn’t the green light to have as many sugary drinks as you want, but it might be interesting to see how your work is affected after a soda.
Chicken nuggets and the meat paradox
Two young children are fighting over the last chicken nugget when an adult comes in to see what’s going on.
Liam: Vegetable!
Olivia: Meat!
Liam: Chicken nuggets are vegetables!
Olivia: No, dorkface! They’re meat.
Caregiver: Good news, kids. You’re both right.
Olivia: How can we both be right?
At this point, a woman enters the room. She’s wearing a white lab coat, so she must be a scientist.
Dr. Scientist: You can’t both be right, Olivia. You are being fed a serving of the meat paradox. That’s why Liam here doesn’t know that chicken nuggets are made of chicken, which is a form of meat. Sadly, he’s not the only one.
In a recent study, scientists from Furman University in Greenville, S.C., found that 38% of 176 children aged 4-7 years thought that chicken nuggets were vegetables and more than 46% identified French fries as animal based.
Olivia: Did our caregiver lie to us, Dr. Scientist?
Dr. Scientist: Yes, Olivia. The researchers I mentioned explained that “many people experience unease while eating meat. Omnivores eat foods that entail animal suffering and death while at the same time endorsing the compassionate treatment of animals.” That’s the meat paradox.
Liam: What else did they say, Dr. Scientist?
Dr. Scientist: Over 70% of those children said that cows and pigs were not edible and 5% thought that cats and horses were. The investigators wrote “that children and youth should be viewed as agents of environmental change” in the future, but suggested that parents need to bring honesty to the table.
Caregiver: How did you get in here anyway? And how do you know their names?
Dr. Scientist: I’ve been rooting through your garbage for years. All in the name of science, of course.
Bedtimes aren’t just for children
There are multiple ways to prevent heart disease, but what if it could be as easy as switching your bedtime? A recent study in European Heart Journal–Digital Health suggests that there’s a sweet spot when it comes to sleep timing.
Through smartwatch-like devices, researchers measured the sleep-onset and wake-up times for 7 days in 88,026 participants aged 43-79 years. After 5.7 years of follow-up to see if anyone had a heart attack, stroke, or any other cardiovascular event, 3.6% developed some kind of cardiovascular disease.
Those who went to bed between 10 p.m. and 11 p.m. had a lower risk of developing heart disease. The risk was 25% higher for subjects who went to bed at midnight or later, 24% higher for bedtimes before 10 p.m., and 12% higher for bedtimes between 11 p.m. and midnight.
So, why can you go to bed before “The Tonight Show” and lower your cardiovascular risk but not before the nightly news? Well, it has something to do with your body’s natural clock.
“The optimum time to go to sleep is at a specific point in the body’s 24-hour cycle and deviations may be detrimental to health. The riskiest time was after midnight, potentially because it may reduce the likelihood of seeing morning light, which resets the body clock,” said study author Dr. David Plans of the University of Exeter, England.
Although a sleep schedule is preferred, it isn’t realistic all the time for those in certain occupations who might have to resort to other methods to keep their circadian clocks ticking optimally for their health. But if all it takes is prescribing a sleep time to reduce heart disease on a massive scale it would make a great “low-cost public health target.”
So bedtimes aren’t just for children.
The greatest autopsy on Earth?
The LOTME staff would like to apologize in advance. The following item contains historical facts.
P.T. Barnum is a rather controversial figure in American history. The greatest show on Earth was certainly popular in its day. However, Barnum got his start in 1835 by leasing a slave named Joyce Heth, an elderly Black woman who told vivid stories of caring for a young George Washington. He toured her around the country, advertising her as a 160-year-old woman who served as George Washington’s nanny. When Ms. Heth died the next year, Barnum sold tickets to the autopsy, charging the equivalent of $30 in today’s money.
When a doctor announced that Ms. Heth was actually 75-80 when she died, it caused great controversy in the press and ruined Barnum’s career. Wait, no, that’s not right. The opposite, actually. He weathered the storm, built his famous circus, and never again committed a hoax.
It’s difficult to quantify how wrong publicly dissecting a person and charging people to see said dissection is, but that was almost 200 years ago. At the very least, we can say that such terrible behavior is firmly in the distant past.
Oh wait.
David Saunders, a 98-year-old veteran of World War II and the Korean War, donated his body to science. His body, however, was purchased by DeathScience.org from a medical lab – with the buyer supposedly misleading the medical lab about its intentions, which was for use at the traveling Oddities and Curiosities Expo. Tickets went for up to $500 each to witness the public autopsy of Mr. Saunders’ body, which took place at a Marriott in Portland, Ore. It promised to be an exciting, all-day event from 9 a.m. to 4 p.m., with a break for lunch, of course. You can’t have an autopsy without a catered lunch.
Another public autopsy event was scheduled in Seattle but canceled after news of the first event broke. Oh, and for that extra little kick, Mr. Saunders died from COVID-19, meaning that all those paying customers were exposed.
P.T. Barnum is probably rolling over in his grave right now. His autopsy tickets were a bargain.
Go ahead, have that soda before math
We should all know by now that sugary drinks are bad, even artificially sweetened ones. It might not always stop us from drinking them, but we know the deal. But what if sugary drinks like soda could be helpful for girls in school?
You read that right. We said girls. A soda before class might have boys bouncing off the walls, but not girls. A recent study showed that not only was girls’ behavior unaffected by having a sugary drink, their math skills even improved.
Researchers analyzed the behavior of 4- to 6-year-old children before and after having a sugary drink. The sugar rush was actually calming for girls and helped them perform better with numerical skills, but the opposite was true for boys. “Our study is the first to provide large-scale experimental evidence on the impact of sugary drinks on preschool children. The results clearly indicate a causal impact of sugary drinks on children’s behavior and test scores,” Fritz Schiltz, PhD, said in a written statement.
This probably isn’t the green light to have as many sugary drinks as you want, but it might be interesting to see how your work is affected after a soda.
Chicken nuggets and the meat paradox
Two young children are fighting over the last chicken nugget when an adult comes in to see what’s going on.
Liam: Vegetable!
Olivia: Meat!
Liam: Chicken nuggets are vegetables!
Olivia: No, dorkface! They’re meat.
Caregiver: Good news, kids. You’re both right.
Olivia: How can we both be right?
At this point, a woman enters the room. She’s wearing a white lab coat, so she must be a scientist.
Dr. Scientist: You can’t both be right, Olivia. You are being fed a serving of the meat paradox. That’s why Liam here doesn’t know that chicken nuggets are made of chicken, which is a form of meat. Sadly, he’s not the only one.
In a recent study, scientists from Furman University in Greenville, S.C., found that 38% of 176 children aged 4-7 years thought that chicken nuggets were vegetables and more than 46% identified French fries as animal based.
Olivia: Did our caregiver lie to us, Dr. Scientist?
Dr. Scientist: Yes, Olivia. The researchers I mentioned explained that “many people experience unease while eating meat. Omnivores eat foods that entail animal suffering and death while at the same time endorsing the compassionate treatment of animals.” That’s the meat paradox.
Liam: What else did they say, Dr. Scientist?
Dr. Scientist: Over 70% of those children said that cows and pigs were not edible and 5% thought that cats and horses were. The investigators wrote “that children and youth should be viewed as agents of environmental change” in the future, but suggested that parents need to bring honesty to the table.
Caregiver: How did you get in here anyway? And how do you know their names?
Dr. Scientist: I’ve been rooting through your garbage for years. All in the name of science, of course.
Bedtimes aren’t just for children
There are multiple ways to prevent heart disease, but what if it could be as easy as switching your bedtime? A recent study in European Heart Journal–Digital Health suggests that there’s a sweet spot when it comes to sleep timing.
Through smartwatch-like devices, researchers measured the sleep-onset and wake-up times for 7 days in 88,026 participants aged 43-79 years. After 5.7 years of follow-up to see if anyone had a heart attack, stroke, or any other cardiovascular event, 3.6% developed some kind of cardiovascular disease.
Those who went to bed between 10 p.m. and 11 p.m. had a lower risk of developing heart disease. The risk was 25% higher for subjects who went to bed at midnight or later, 24% higher for bedtimes before 10 p.m., and 12% higher for bedtimes between 11 p.m. and midnight.
So, why can you go to bed before “The Tonight Show” and lower your cardiovascular risk but not before the nightly news? Well, it has something to do with your body’s natural clock.
“The optimum time to go to sleep is at a specific point in the body’s 24-hour cycle and deviations may be detrimental to health. The riskiest time was after midnight, potentially because it may reduce the likelihood of seeing morning light, which resets the body clock,” said study author Dr. David Plans of the University of Exeter, England.
Although a sleep schedule is preferred, it isn’t realistic all the time for those in certain occupations who might have to resort to other methods to keep their circadian clocks ticking optimally for their health. But if all it takes is prescribing a sleep time to reduce heart disease on a massive scale it would make a great “low-cost public health target.”
So bedtimes aren’t just for children.
mRNA COVID vaccine response found mostly robust in RA, SLE patients
Immunosuppressed patients with autoimmune diseases who received the Moderna mRNA-1273 SARS-CoV-2 two-dose vaccine series had a frequency of adverse events similar to the general population albeit with a somewhat reduced, but still significant, antibody response with no severe vaccine-related disease flares, results of a prospective, nonrandomized open-label comparative trial in Canada demonstrated.
At the same time, patients with RA who were taking rituximab and patients with systemic lupus erythematosus (SLE) who were taking mycophenolate mofetil seemed to have reduced humoral responses after receiving the vaccine, said Ines Colmegna, MD, reporting results of the COVID-19 Vaccine in Immunosuppressed Adults with Autoimmune Disease (COVIAAD) study as a late-breaking poster abstract at the virtual annual meeting of the American College of Rheumatology. Dr. Colmegna is an associate professor of rheumatology in the division of experimental medicine at McGill University, Montreal.
“The frequency of adverse events, specifically the reactogenicity in people with comorbid conditions regardless of their diagnosis, was similar to healthy controls in this study, and their frequency was similar also the initial studies in the general population,” Dr. Colmegna said.
COVIAAD prospectively enrolled 220 fully vaccinated patients, 162 with rheumatic disease (131 with RA, 23 with SLE, and 8 with other diseases) and 58 controls. Adverse events a week and a month after each dose was the primary outcome. The postvaccine presence of the IgG antibody against the SARS-CoV-2 spike protein and the receptor binding domain (IgG-RBD) was the secondary outcome. Dr. Colmegna said that the study will continue evaluating participants after they get a third dose.
The Canadian trial appears to validate the ACR’s COVID-19 vaccine guidance, the fourth version of which was issued in October, said Jeffrey Curtis, MD, MS, MPH, professor of immunology and rheumatology at the University of Alabama at Birmingham and lead of the ACR COVID-19 Vaccine Guidance Task Force. Specifically, the guidance recommends that patients on rituximab or other anti-CD20 B-cell–depleting agents discuss vaccine timing with their rheumatologist.
“A few things changed over time when there was a paucity of evidence for any vaccine, but as time has gone on, mostly we were more correct than we weren’t,” Dr. Curtis said of the task force’s work. “The evidence that now is in this poster with regard to systemic lupus erythematosus and mycophenolate mofetil is [that] you have impaired vaccine response. If you’re on a B-cell drug like rituximab, you really have impaired vaccine response.”
In the study, 100% of controls had immunogenicity in terms of anti-spike and anti-RBD levels after the first and second dose. The rate of immunogenicity after the first and second dose were 67% and 88% in all patients with RA, and 35% and 78% in patients with SLE who were taking mycophenolate mofetil. The subset of patients with RA on rituximab (n = 17) had rates of immunogenicity of 5.9% and 17.6%, respectively.
“Measured antibody response is not the only way in which people develop a response to a vaccine, and there are also similar responses that occur even in people who are on rituximab and have not developed antibodies,” Dr. Colmegna said. “That’s a very important message also that we need to convey to patients: The immune response really extends beyond antibody protection.”
Overall, disease activity in both patients with RA and SLE did not appreciably change from baseline within 7 days and 28 days of each vaccine dose.
The study raises important questions about the timing of the vaccine, particularly in patients on rituximab, Dr. Colmegna said in an interview. “In theory, there is no element to suggest that, if you would schedule the vaccine a month prior to the next dose of rituximab, the effect of the drug would have decreased the number of B cells, and that the possibility of developing antibodies in response to the vaccine might be better if you give rituximab a month later when the amount of the drug and the effect of the drug is maximal,” she said. The average interval between patients receiving rituximab and vaccines was 4.5 months, Dr. Colmegna said in answering a question after her presentation.
Dr. Curtis said that the effect of holding rituximab or the vaccine to boost antibodies “is somewhat yet unknown. We think it will help, but that’s not a guarantee,” he said. “We don’t have direct evidence that just because the drug impairs vaccine response, that holding that drug for a week or 2 is going to take care of the problem.”
The study does arm rheumatologists with more information for discussing COVID vaccines with vaccine-hesitant patients with autoimmune diseases, Dr. Curtis said.
“It gives them evidence that for most of our immunomodulatory drugs the vaccine works pretty well,” he said. “The poster provides evidence that, compared to healthy controls, the vaccine doesn’t work quite as well in some patients, but for most people it actually did work pretty well. That reinforces the message: Go get vaccinated because [you] will mount [an immune] response, even, if that response isn’t quite as brisk as it is in healthy people.”
Dr. Colmegna and Dr. Curtis have no relevant relationships to disclose. The study received funding from Health and Social Services Quebec.
Immunosuppressed patients with autoimmune diseases who received the Moderna mRNA-1273 SARS-CoV-2 two-dose vaccine series had a frequency of adverse events similar to the general population albeit with a somewhat reduced, but still significant, antibody response with no severe vaccine-related disease flares, results of a prospective, nonrandomized open-label comparative trial in Canada demonstrated.
At the same time, patients with RA who were taking rituximab and patients with systemic lupus erythematosus (SLE) who were taking mycophenolate mofetil seemed to have reduced humoral responses after receiving the vaccine, said Ines Colmegna, MD, reporting results of the COVID-19 Vaccine in Immunosuppressed Adults with Autoimmune Disease (COVIAAD) study as a late-breaking poster abstract at the virtual annual meeting of the American College of Rheumatology. Dr. Colmegna is an associate professor of rheumatology in the division of experimental medicine at McGill University, Montreal.
“The frequency of adverse events, specifically the reactogenicity in people with comorbid conditions regardless of their diagnosis, was similar to healthy controls in this study, and their frequency was similar also the initial studies in the general population,” Dr. Colmegna said.
COVIAAD prospectively enrolled 220 fully vaccinated patients, 162 with rheumatic disease (131 with RA, 23 with SLE, and 8 with other diseases) and 58 controls. Adverse events a week and a month after each dose was the primary outcome. The postvaccine presence of the IgG antibody against the SARS-CoV-2 spike protein and the receptor binding domain (IgG-RBD) was the secondary outcome. Dr. Colmegna said that the study will continue evaluating participants after they get a third dose.
The Canadian trial appears to validate the ACR’s COVID-19 vaccine guidance, the fourth version of which was issued in October, said Jeffrey Curtis, MD, MS, MPH, professor of immunology and rheumatology at the University of Alabama at Birmingham and lead of the ACR COVID-19 Vaccine Guidance Task Force. Specifically, the guidance recommends that patients on rituximab or other anti-CD20 B-cell–depleting agents discuss vaccine timing with their rheumatologist.
“A few things changed over time when there was a paucity of evidence for any vaccine, but as time has gone on, mostly we were more correct than we weren’t,” Dr. Curtis said of the task force’s work. “The evidence that now is in this poster with regard to systemic lupus erythematosus and mycophenolate mofetil is [that] you have impaired vaccine response. If you’re on a B-cell drug like rituximab, you really have impaired vaccine response.”
In the study, 100% of controls had immunogenicity in terms of anti-spike and anti-RBD levels after the first and second dose. The rate of immunogenicity after the first and second dose were 67% and 88% in all patients with RA, and 35% and 78% in patients with SLE who were taking mycophenolate mofetil. The subset of patients with RA on rituximab (n = 17) had rates of immunogenicity of 5.9% and 17.6%, respectively.
“Measured antibody response is not the only way in which people develop a response to a vaccine, and there are also similar responses that occur even in people who are on rituximab and have not developed antibodies,” Dr. Colmegna said. “That’s a very important message also that we need to convey to patients: The immune response really extends beyond antibody protection.”
Overall, disease activity in both patients with RA and SLE did not appreciably change from baseline within 7 days and 28 days of each vaccine dose.
The study raises important questions about the timing of the vaccine, particularly in patients on rituximab, Dr. Colmegna said in an interview. “In theory, there is no element to suggest that, if you would schedule the vaccine a month prior to the next dose of rituximab, the effect of the drug would have decreased the number of B cells, and that the possibility of developing antibodies in response to the vaccine might be better if you give rituximab a month later when the amount of the drug and the effect of the drug is maximal,” she said. The average interval between patients receiving rituximab and vaccines was 4.5 months, Dr. Colmegna said in answering a question after her presentation.
Dr. Curtis said that the effect of holding rituximab or the vaccine to boost antibodies “is somewhat yet unknown. We think it will help, but that’s not a guarantee,” he said. “We don’t have direct evidence that just because the drug impairs vaccine response, that holding that drug for a week or 2 is going to take care of the problem.”
The study does arm rheumatologists with more information for discussing COVID vaccines with vaccine-hesitant patients with autoimmune diseases, Dr. Curtis said.
“It gives them evidence that for most of our immunomodulatory drugs the vaccine works pretty well,” he said. “The poster provides evidence that, compared to healthy controls, the vaccine doesn’t work quite as well in some patients, but for most people it actually did work pretty well. That reinforces the message: Go get vaccinated because [you] will mount [an immune] response, even, if that response isn’t quite as brisk as it is in healthy people.”
Dr. Colmegna and Dr. Curtis have no relevant relationships to disclose. The study received funding from Health and Social Services Quebec.
Immunosuppressed patients with autoimmune diseases who received the Moderna mRNA-1273 SARS-CoV-2 two-dose vaccine series had a frequency of adverse events similar to the general population albeit with a somewhat reduced, but still significant, antibody response with no severe vaccine-related disease flares, results of a prospective, nonrandomized open-label comparative trial in Canada demonstrated.
At the same time, patients with RA who were taking rituximab and patients with systemic lupus erythematosus (SLE) who were taking mycophenolate mofetil seemed to have reduced humoral responses after receiving the vaccine, said Ines Colmegna, MD, reporting results of the COVID-19 Vaccine in Immunosuppressed Adults with Autoimmune Disease (COVIAAD) study as a late-breaking poster abstract at the virtual annual meeting of the American College of Rheumatology. Dr. Colmegna is an associate professor of rheumatology in the division of experimental medicine at McGill University, Montreal.
“The frequency of adverse events, specifically the reactogenicity in people with comorbid conditions regardless of their diagnosis, was similar to healthy controls in this study, and their frequency was similar also the initial studies in the general population,” Dr. Colmegna said.
COVIAAD prospectively enrolled 220 fully vaccinated patients, 162 with rheumatic disease (131 with RA, 23 with SLE, and 8 with other diseases) and 58 controls. Adverse events a week and a month after each dose was the primary outcome. The postvaccine presence of the IgG antibody against the SARS-CoV-2 spike protein and the receptor binding domain (IgG-RBD) was the secondary outcome. Dr. Colmegna said that the study will continue evaluating participants after they get a third dose.
The Canadian trial appears to validate the ACR’s COVID-19 vaccine guidance, the fourth version of which was issued in October, said Jeffrey Curtis, MD, MS, MPH, professor of immunology and rheumatology at the University of Alabama at Birmingham and lead of the ACR COVID-19 Vaccine Guidance Task Force. Specifically, the guidance recommends that patients on rituximab or other anti-CD20 B-cell–depleting agents discuss vaccine timing with their rheumatologist.
“A few things changed over time when there was a paucity of evidence for any vaccine, but as time has gone on, mostly we were more correct than we weren’t,” Dr. Curtis said of the task force’s work. “The evidence that now is in this poster with regard to systemic lupus erythematosus and mycophenolate mofetil is [that] you have impaired vaccine response. If you’re on a B-cell drug like rituximab, you really have impaired vaccine response.”
In the study, 100% of controls had immunogenicity in terms of anti-spike and anti-RBD levels after the first and second dose. The rate of immunogenicity after the first and second dose were 67% and 88% in all patients with RA, and 35% and 78% in patients with SLE who were taking mycophenolate mofetil. The subset of patients with RA on rituximab (n = 17) had rates of immunogenicity of 5.9% and 17.6%, respectively.
“Measured antibody response is not the only way in which people develop a response to a vaccine, and there are also similar responses that occur even in people who are on rituximab and have not developed antibodies,” Dr. Colmegna said. “That’s a very important message also that we need to convey to patients: The immune response really extends beyond antibody protection.”
Overall, disease activity in both patients with RA and SLE did not appreciably change from baseline within 7 days and 28 days of each vaccine dose.
The study raises important questions about the timing of the vaccine, particularly in patients on rituximab, Dr. Colmegna said in an interview. “In theory, there is no element to suggest that, if you would schedule the vaccine a month prior to the next dose of rituximab, the effect of the drug would have decreased the number of B cells, and that the possibility of developing antibodies in response to the vaccine might be better if you give rituximab a month later when the amount of the drug and the effect of the drug is maximal,” she said. The average interval between patients receiving rituximab and vaccines was 4.5 months, Dr. Colmegna said in answering a question after her presentation.
Dr. Curtis said that the effect of holding rituximab or the vaccine to boost antibodies “is somewhat yet unknown. We think it will help, but that’s not a guarantee,” he said. “We don’t have direct evidence that just because the drug impairs vaccine response, that holding that drug for a week or 2 is going to take care of the problem.”
The study does arm rheumatologists with more information for discussing COVID vaccines with vaccine-hesitant patients with autoimmune diseases, Dr. Curtis said.
“It gives them evidence that for most of our immunomodulatory drugs the vaccine works pretty well,” he said. “The poster provides evidence that, compared to healthy controls, the vaccine doesn’t work quite as well in some patients, but for most people it actually did work pretty well. That reinforces the message: Go get vaccinated because [you] will mount [an immune] response, even, if that response isn’t quite as brisk as it is in healthy people.”
Dr. Colmegna and Dr. Curtis have no relevant relationships to disclose. The study received funding from Health and Social Services Quebec.
FROM ACR 2021
Pfizer seeks EUA expansion for COVID-19 booster
Pfizer and its European partner BioNTech on Nov. 9 asked the U.S. government to expand emergency use authorization (EUA) to allow everybody over 18 to receive their COVID-19 booster shots.
If the request is approved, He announced the goal last August but backed off after some scientists said younger people may not need boosters, especially with large parts of the world unvaccinated.
Pfizer is submitting a study of booster effects on 10,000 people to make its case, according to The Associated Press.
This would be Pfizer’s second attempt. In September, a Food and Drug Administration advisory panel turned down Pfizer’s idea of booster shots for everybody over 18.
However, the committee recommended Pfizer booster shots for people 65 and over, essential workers, and people with underlying health conditions.
The FDA and the Centers for Disease Control and Prevention authorized the Pfizer booster for those other groups and later authorization was granted for the same groups with Moderna and Johnson & Johnson boosters. People who got the two-shot Pfizer or Moderna vaccines should get a booster 6 months after the second dose and people who got the one-dose J&J vaccine should get a booster 2 months later.
The pro-booster argument has strengthened because new data have come in from Israel that confirm boosters provide protection as vaccine effectiveness wanes over time, The Washington Post reported. Also, health officials are worried about a post-holiday surge and because COVID-19 case counts and deaths are not dropping in every part of the country, though they are declining overall, according to the The Post report.
The regulatory path for a booster-for-all application is unclear. The Post, citing two unnamed officials, said the FDA probably won’t send the Pfizer application to the FDA advisory committee this time because the committee has already had extensive discussions about boosters. If the FDA gives the green light, CDC Director Rochelle Walensky, MD, would have to make updated recommendations on boosters, The Post article noted.
A version of this article first appeared on WebMD.com.
Pfizer and its European partner BioNTech on Nov. 9 asked the U.S. government to expand emergency use authorization (EUA) to allow everybody over 18 to receive their COVID-19 booster shots.
If the request is approved, He announced the goal last August but backed off after some scientists said younger people may not need boosters, especially with large parts of the world unvaccinated.
Pfizer is submitting a study of booster effects on 10,000 people to make its case, according to The Associated Press.
This would be Pfizer’s second attempt. In September, a Food and Drug Administration advisory panel turned down Pfizer’s idea of booster shots for everybody over 18.
However, the committee recommended Pfizer booster shots for people 65 and over, essential workers, and people with underlying health conditions.
The FDA and the Centers for Disease Control and Prevention authorized the Pfizer booster for those other groups and later authorization was granted for the same groups with Moderna and Johnson & Johnson boosters. People who got the two-shot Pfizer or Moderna vaccines should get a booster 6 months after the second dose and people who got the one-dose J&J vaccine should get a booster 2 months later.
The pro-booster argument has strengthened because new data have come in from Israel that confirm boosters provide protection as vaccine effectiveness wanes over time, The Washington Post reported. Also, health officials are worried about a post-holiday surge and because COVID-19 case counts and deaths are not dropping in every part of the country, though they are declining overall, according to the The Post report.
The regulatory path for a booster-for-all application is unclear. The Post, citing two unnamed officials, said the FDA probably won’t send the Pfizer application to the FDA advisory committee this time because the committee has already had extensive discussions about boosters. If the FDA gives the green light, CDC Director Rochelle Walensky, MD, would have to make updated recommendations on boosters, The Post article noted.
A version of this article first appeared on WebMD.com.
Pfizer and its European partner BioNTech on Nov. 9 asked the U.S. government to expand emergency use authorization (EUA) to allow everybody over 18 to receive their COVID-19 booster shots.
If the request is approved, He announced the goal last August but backed off after some scientists said younger people may not need boosters, especially with large parts of the world unvaccinated.
Pfizer is submitting a study of booster effects on 10,000 people to make its case, according to The Associated Press.
This would be Pfizer’s second attempt. In September, a Food and Drug Administration advisory panel turned down Pfizer’s idea of booster shots for everybody over 18.
However, the committee recommended Pfizer booster shots for people 65 and over, essential workers, and people with underlying health conditions.
The FDA and the Centers for Disease Control and Prevention authorized the Pfizer booster for those other groups and later authorization was granted for the same groups with Moderna and Johnson & Johnson boosters. People who got the two-shot Pfizer or Moderna vaccines should get a booster 6 months after the second dose and people who got the one-dose J&J vaccine should get a booster 2 months later.
The pro-booster argument has strengthened because new data have come in from Israel that confirm boosters provide protection as vaccine effectiveness wanes over time, The Washington Post reported. Also, health officials are worried about a post-holiday surge and because COVID-19 case counts and deaths are not dropping in every part of the country, though they are declining overall, according to the The Post report.
The regulatory path for a booster-for-all application is unclear. The Post, citing two unnamed officials, said the FDA probably won’t send the Pfizer application to the FDA advisory committee this time because the committee has already had extensive discussions about boosters. If the FDA gives the green light, CDC Director Rochelle Walensky, MD, would have to make updated recommendations on boosters, The Post article noted.
A version of this article first appeared on WebMD.com.
Should you tell your doctor that you’re a doctor?
The question drew spirited debate when urologist Ashley Winter, MD, made a simple, straightforward request on Twitter: “If you are a doctor & you come to an appointment please tell me you are a doctor, not because I will treat you differently but because it’s easier to speak in jargon.”
She later added, “This doesn’t’ mean I would be less patient-focused or emotional with a physician or other [healthcare worker]. Just means that, instead of saying ‘you will have a catheter draining your urine to a bag,’ I can say, ‘you will have a Foley.’ ”
The Tweet followed an encounter with a patient who told Dr. Winter that he was a doctor only after she had gone to some length explaining a surgical procedure in lay terms.
“I explained the surgery, obviously assuming he was an intelligent adult, but using fully layman’s terms,” she said in an interview. The patient then told her that he was a doctor. “I guess I felt this embarrassment — I wouldn’t have treated him differently, but I just could have discussed the procedure with him in more professional terms.”
“To some extent, it was my own fault,” she commented in an interview. “I didn’t take the time to ask [about his work] at the beginning of the consultation, but that’s a fine line, also,” added Dr. Winter, a urologist and sexual medicine physician in Portland, Ore.
“You know that patient is there because they want care from you and it’s not necessarily always at the forefront of importance to be asking them what they do for their work, but alternatively, if you don’t ask then you put them in this position where they have to find a way to go ahead and tell you.”
Several people chimed in on the thread to voice their thoughts on the matter. Some commiserated with Dr. Winter’s experience:
“I took care of a retired cardiologist in the hospital as a second-year resident and honest to god he let me ramble on ‘explaining’ his echo result and never told me. I found out a couple days later and wanted to die,” posted @MaddyAndrewsMD.
Another recalled a similarly embarrassing experience when she “went on and on” discussing headaches with a patient whose husband “was in the corner smirking.”
“They told my attending later [that the] husband was a retired FM doc who practiced medicine longer than I’ve been alive. I wanted to die,” posted @JSinghDO.
Many on the thread, though, were doctors and other healthcare professionals speaking as patients. Some said they didn’t want to disclose their status as a healthcare provider because they felt it affected the care they received.
For example, @drhelenrainford commented: “In my experience my care is less ‘caring’ when they know I am a [doctor]. I get spoken to like they are discussing a patient with me — no empathy just facts and difficult results just blurted out without consideration. Awful awful time as an inpatient …but that’s another story!”
@Dr_B_Ring said: “Nope – You and I speak different jargon – I would want you to speak to me like a human that doesn’t know your jargon. My ego would get in the way of asking about the acronyms I don’t know if you knew I was a fellow physician.”
Conversely, @lozzlemcfozzle said: “Honestly I prefer not to tell my Doctors — I’ve found people skip explanations assuming I ‘know,’ or seem a little nervous when I tell them!”
Others said they felt uncomfortable — pretentious, even — in announcing their status, or worried that they might come across as expecting special care.
“It’s such a tough needle to thread. Want to tell people early but not come off as demanding special treatment, but don’t want to wait too long and it seems like a trap,” said @MDaware.
Twitter user @MsBabyCatcher wrote: “I have a hard time doing this because I don’t want people to think I’m being pretentious or going to micromanage/dictate care.”
Replying to @MsBabyCatcher, @RedStethoscope wrote: “I used to think this too until I got [very poor] care a few times, and was advised by other doctor moms to ‘play the doctor card.’ I have gotten better/more compassionate care by making sure it’s clear that I’m a physician (which is junk, but here we are).”
Several of those responding used the words “tricky” and “awkward,” suggesting a common theme for doctors presenting as patients.
“I struggle with this. My 5-year-old broke her arm this weekend, we spent hours in the ED, of my own hospital, I never mentioned it because I didn’t want to get preferential care. But as they were explaining her type of fracture, it felt awkward and inefficient,” said @lindsay_petty.
To avoid the awkwardness, a number of respondents said they purposefully use medical jargon to open up a conversation rather than just offering up the information that they are a doctor.
Still others offered suggestions on how to broach the subject more directly when presenting as a patient:
‘”Just FYI I’m a X doc but I’m here because I really want your help and advice!” That’s what I usually do,” wrote @drcakefm.
@BeeSting14618 Tweeted: “I usually say ‘I know some of this but I’m here because I want YOUR guidance. Also I may ask dumb questions, and I’ll tell you if a question is asking your opinion or making a request.’”
A few others injected a bit of humor: “I just do the 14-part handshake that only doctors know. Is that not customary?” quipped @Branmiz25.
“Ah yes, that transmits the entire [history of present illness],” replied Dr. Winter.
Jokes aside, the topic is obviously one that touched on a shared experience among healthcare providers, Dr. Winter commented. The Twitter thread she started just “blew up.”
That’s typically a sign that the Tweet is relatable for a lot of people, she said.
“It’s definitely something that all of us as care providers and as patients understand. It’s a funny, awkward thing that can really change an interaction, so we probably all feel pretty strongly about our experiences related to that,” she added.
The debate begs the question: Is there a duty or ethical reason to disclose?
“I definitely think it is very reasonable to disclose that one is a medical professional to another doctor,” medical ethicist Charlotte Blease, PhD, said in an interview. “There are good reasons to believe doing so might make a difference to the quality of communication and transparency.”
If the ability to use medical terminology or jargon more freely improves patient understanding, autonomy, and shared decision-making, then it may be of benefit, said Dr. Blease, a Keane OpenNotes Scholar at Beth Israel Deaconess Medical Center in Boston.
“Since doctors should strive to communicate effectively with every patient and to respect their unique needs and level of understanding, then I see no reason to deny that one is a medic,” she added.”
Knowing how to share the information is another story.
“This is something that affects all of us as physicians — we’re going to be patients at some point, right?” Dr. Winter commented. “But I don’t think how to disclose that is something that was ever brought up in my medical training.”
“Maybe there should just be a discussion of this one day when people are in medical school — maybe in a professionalism course — to broach this topic or look at if there’s any literature on outcomes related to disclosure of status or what are best practices,” she suggested.
A version of this article first appeared on Medscape.com.
The question drew spirited debate when urologist Ashley Winter, MD, made a simple, straightforward request on Twitter: “If you are a doctor & you come to an appointment please tell me you are a doctor, not because I will treat you differently but because it’s easier to speak in jargon.”
She later added, “This doesn’t’ mean I would be less patient-focused or emotional with a physician or other [healthcare worker]. Just means that, instead of saying ‘you will have a catheter draining your urine to a bag,’ I can say, ‘you will have a Foley.’ ”
The Tweet followed an encounter with a patient who told Dr. Winter that he was a doctor only after she had gone to some length explaining a surgical procedure in lay terms.
“I explained the surgery, obviously assuming he was an intelligent adult, but using fully layman’s terms,” she said in an interview. The patient then told her that he was a doctor. “I guess I felt this embarrassment — I wouldn’t have treated him differently, but I just could have discussed the procedure with him in more professional terms.”
“To some extent, it was my own fault,” she commented in an interview. “I didn’t take the time to ask [about his work] at the beginning of the consultation, but that’s a fine line, also,” added Dr. Winter, a urologist and sexual medicine physician in Portland, Ore.
“You know that patient is there because they want care from you and it’s not necessarily always at the forefront of importance to be asking them what they do for their work, but alternatively, if you don’t ask then you put them in this position where they have to find a way to go ahead and tell you.”
Several people chimed in on the thread to voice their thoughts on the matter. Some commiserated with Dr. Winter’s experience:
“I took care of a retired cardiologist in the hospital as a second-year resident and honest to god he let me ramble on ‘explaining’ his echo result and never told me. I found out a couple days later and wanted to die,” posted @MaddyAndrewsMD.
Another recalled a similarly embarrassing experience when she “went on and on” discussing headaches with a patient whose husband “was in the corner smirking.”
“They told my attending later [that the] husband was a retired FM doc who practiced medicine longer than I’ve been alive. I wanted to die,” posted @JSinghDO.
Many on the thread, though, were doctors and other healthcare professionals speaking as patients. Some said they didn’t want to disclose their status as a healthcare provider because they felt it affected the care they received.
For example, @drhelenrainford commented: “In my experience my care is less ‘caring’ when they know I am a [doctor]. I get spoken to like they are discussing a patient with me — no empathy just facts and difficult results just blurted out without consideration. Awful awful time as an inpatient …but that’s another story!”
@Dr_B_Ring said: “Nope – You and I speak different jargon – I would want you to speak to me like a human that doesn’t know your jargon. My ego would get in the way of asking about the acronyms I don’t know if you knew I was a fellow physician.”
Conversely, @lozzlemcfozzle said: “Honestly I prefer not to tell my Doctors — I’ve found people skip explanations assuming I ‘know,’ or seem a little nervous when I tell them!”
Others said they felt uncomfortable — pretentious, even — in announcing their status, or worried that they might come across as expecting special care.
“It’s such a tough needle to thread. Want to tell people early but not come off as demanding special treatment, but don’t want to wait too long and it seems like a trap,” said @MDaware.
Twitter user @MsBabyCatcher wrote: “I have a hard time doing this because I don’t want people to think I’m being pretentious or going to micromanage/dictate care.”
Replying to @MsBabyCatcher, @RedStethoscope wrote: “I used to think this too until I got [very poor] care a few times, and was advised by other doctor moms to ‘play the doctor card.’ I have gotten better/more compassionate care by making sure it’s clear that I’m a physician (which is junk, but here we are).”
Several of those responding used the words “tricky” and “awkward,” suggesting a common theme for doctors presenting as patients.
“I struggle with this. My 5-year-old broke her arm this weekend, we spent hours in the ED, of my own hospital, I never mentioned it because I didn’t want to get preferential care. But as they were explaining her type of fracture, it felt awkward and inefficient,” said @lindsay_petty.
To avoid the awkwardness, a number of respondents said they purposefully use medical jargon to open up a conversation rather than just offering up the information that they are a doctor.
Still others offered suggestions on how to broach the subject more directly when presenting as a patient:
‘”Just FYI I’m a X doc but I’m here because I really want your help and advice!” That’s what I usually do,” wrote @drcakefm.
@BeeSting14618 Tweeted: “I usually say ‘I know some of this but I’m here because I want YOUR guidance. Also I may ask dumb questions, and I’ll tell you if a question is asking your opinion or making a request.’”
A few others injected a bit of humor: “I just do the 14-part handshake that only doctors know. Is that not customary?” quipped @Branmiz25.
“Ah yes, that transmits the entire [history of present illness],” replied Dr. Winter.
Jokes aside, the topic is obviously one that touched on a shared experience among healthcare providers, Dr. Winter commented. The Twitter thread she started just “blew up.”
That’s typically a sign that the Tweet is relatable for a lot of people, she said.
“It’s definitely something that all of us as care providers and as patients understand. It’s a funny, awkward thing that can really change an interaction, so we probably all feel pretty strongly about our experiences related to that,” she added.
The debate begs the question: Is there a duty or ethical reason to disclose?
“I definitely think it is very reasonable to disclose that one is a medical professional to another doctor,” medical ethicist Charlotte Blease, PhD, said in an interview. “There are good reasons to believe doing so might make a difference to the quality of communication and transparency.”
If the ability to use medical terminology or jargon more freely improves patient understanding, autonomy, and shared decision-making, then it may be of benefit, said Dr. Blease, a Keane OpenNotes Scholar at Beth Israel Deaconess Medical Center in Boston.
“Since doctors should strive to communicate effectively with every patient and to respect their unique needs and level of understanding, then I see no reason to deny that one is a medic,” she added.”
Knowing how to share the information is another story.
“This is something that affects all of us as physicians — we’re going to be patients at some point, right?” Dr. Winter commented. “But I don’t think how to disclose that is something that was ever brought up in my medical training.”
“Maybe there should just be a discussion of this one day when people are in medical school — maybe in a professionalism course — to broach this topic or look at if there’s any literature on outcomes related to disclosure of status or what are best practices,” she suggested.
A version of this article first appeared on Medscape.com.
The question drew spirited debate when urologist Ashley Winter, MD, made a simple, straightforward request on Twitter: “If you are a doctor & you come to an appointment please tell me you are a doctor, not because I will treat you differently but because it’s easier to speak in jargon.”
She later added, “This doesn’t’ mean I would be less patient-focused or emotional with a physician or other [healthcare worker]. Just means that, instead of saying ‘you will have a catheter draining your urine to a bag,’ I can say, ‘you will have a Foley.’ ”
The Tweet followed an encounter with a patient who told Dr. Winter that he was a doctor only after she had gone to some length explaining a surgical procedure in lay terms.
“I explained the surgery, obviously assuming he was an intelligent adult, but using fully layman’s terms,” she said in an interview. The patient then told her that he was a doctor. “I guess I felt this embarrassment — I wouldn’t have treated him differently, but I just could have discussed the procedure with him in more professional terms.”
“To some extent, it was my own fault,” she commented in an interview. “I didn’t take the time to ask [about his work] at the beginning of the consultation, but that’s a fine line, also,” added Dr. Winter, a urologist and sexual medicine physician in Portland, Ore.
“You know that patient is there because they want care from you and it’s not necessarily always at the forefront of importance to be asking them what they do for their work, but alternatively, if you don’t ask then you put them in this position where they have to find a way to go ahead and tell you.”
Several people chimed in on the thread to voice their thoughts on the matter. Some commiserated with Dr. Winter’s experience:
“I took care of a retired cardiologist in the hospital as a second-year resident and honest to god he let me ramble on ‘explaining’ his echo result and never told me. I found out a couple days later and wanted to die,” posted @MaddyAndrewsMD.
Another recalled a similarly embarrassing experience when she “went on and on” discussing headaches with a patient whose husband “was in the corner smirking.”
“They told my attending later [that the] husband was a retired FM doc who practiced medicine longer than I’ve been alive. I wanted to die,” posted @JSinghDO.
Many on the thread, though, were doctors and other healthcare professionals speaking as patients. Some said they didn’t want to disclose their status as a healthcare provider because they felt it affected the care they received.
For example, @drhelenrainford commented: “In my experience my care is less ‘caring’ when they know I am a [doctor]. I get spoken to like they are discussing a patient with me — no empathy just facts and difficult results just blurted out without consideration. Awful awful time as an inpatient …but that’s another story!”
@Dr_B_Ring said: “Nope – You and I speak different jargon – I would want you to speak to me like a human that doesn’t know your jargon. My ego would get in the way of asking about the acronyms I don’t know if you knew I was a fellow physician.”
Conversely, @lozzlemcfozzle said: “Honestly I prefer not to tell my Doctors — I’ve found people skip explanations assuming I ‘know,’ or seem a little nervous when I tell them!”
Others said they felt uncomfortable — pretentious, even — in announcing their status, or worried that they might come across as expecting special care.
“It’s such a tough needle to thread. Want to tell people early but not come off as demanding special treatment, but don’t want to wait too long and it seems like a trap,” said @MDaware.
Twitter user @MsBabyCatcher wrote: “I have a hard time doing this because I don’t want people to think I’m being pretentious or going to micromanage/dictate care.”
Replying to @MsBabyCatcher, @RedStethoscope wrote: “I used to think this too until I got [very poor] care a few times, and was advised by other doctor moms to ‘play the doctor card.’ I have gotten better/more compassionate care by making sure it’s clear that I’m a physician (which is junk, but here we are).”
Several of those responding used the words “tricky” and “awkward,” suggesting a common theme for doctors presenting as patients.
“I struggle with this. My 5-year-old broke her arm this weekend, we spent hours in the ED, of my own hospital, I never mentioned it because I didn’t want to get preferential care. But as they were explaining her type of fracture, it felt awkward and inefficient,” said @lindsay_petty.
To avoid the awkwardness, a number of respondents said they purposefully use medical jargon to open up a conversation rather than just offering up the information that they are a doctor.
Still others offered suggestions on how to broach the subject more directly when presenting as a patient:
‘”Just FYI I’m a X doc but I’m here because I really want your help and advice!” That’s what I usually do,” wrote @drcakefm.
@BeeSting14618 Tweeted: “I usually say ‘I know some of this but I’m here because I want YOUR guidance. Also I may ask dumb questions, and I’ll tell you if a question is asking your opinion or making a request.’”
A few others injected a bit of humor: “I just do the 14-part handshake that only doctors know. Is that not customary?” quipped @Branmiz25.
“Ah yes, that transmits the entire [history of present illness],” replied Dr. Winter.
Jokes aside, the topic is obviously one that touched on a shared experience among healthcare providers, Dr. Winter commented. The Twitter thread she started just “blew up.”
That’s typically a sign that the Tweet is relatable for a lot of people, she said.
“It’s definitely something that all of us as care providers and as patients understand. It’s a funny, awkward thing that can really change an interaction, so we probably all feel pretty strongly about our experiences related to that,” she added.
The debate begs the question: Is there a duty or ethical reason to disclose?
“I definitely think it is very reasonable to disclose that one is a medical professional to another doctor,” medical ethicist Charlotte Blease, PhD, said in an interview. “There are good reasons to believe doing so might make a difference to the quality of communication and transparency.”
If the ability to use medical terminology or jargon more freely improves patient understanding, autonomy, and shared decision-making, then it may be of benefit, said Dr. Blease, a Keane OpenNotes Scholar at Beth Israel Deaconess Medical Center in Boston.
“Since doctors should strive to communicate effectively with every patient and to respect their unique needs and level of understanding, then I see no reason to deny that one is a medic,” she added.”
Knowing how to share the information is another story.
“This is something that affects all of us as physicians — we’re going to be patients at some point, right?” Dr. Winter commented. “But I don’t think how to disclose that is something that was ever brought up in my medical training.”
“Maybe there should just be a discussion of this one day when people are in medical school — maybe in a professionalism course — to broach this topic or look at if there’s any literature on outcomes related to disclosure of status or what are best practices,” she suggested.
A version of this article first appeared on Medscape.com.
Unvaccinated people 20 times more likely to die from COVID: Texas study
During the month of September, , according to a new study from the Texas Department of State Health Services.
The data also showed that unvaccinated people were 13 times more likely to test positive for COVID-19 than people who were fully vaccinated.
“This analysis quantifies what we’ve known for months,” Jennifer Shuford, MD, the state’s chief epidemiologist, told The Dallas Morning News.
“The COVID-19 vaccines are doing an excellent job of protecting people from getting sick and from dying from COVID-19,” she said. “Vaccination remains the best way to keep yourself and the people close to you safe from this deadly disease.”
As part of the study, researchers analyzed electronic lab reports, death certificates, and state immunization records, with a particular focus on September when the contagious Delta variant surged across Texas. The research marks the state’s first statistical analysis of COVID-19 vaccinations in Texas and the effects, the newspaper reported.
The protective effect of vaccination was most noticeable among younger groups. During September, the risk of COVID-19 death was 23 times higher in unvaccinated people in their 30s and 55 times higher for unvaccinated people in their 40s.
In addition, there were fewer than 10 COVID-19 deaths in September among fully vaccinated people between ages 18-29, as compared with 339 deaths among unvaccinated people in the same age group.
Then, looking at a longer time period -- from Jan. 15 to Oct. 1 -- the researchers found that unvaccinated people were 45 times more likely to contract COVID-19 than fully vaccinated people. The protective effect of vaccination against infection was strong across all adult age groups but greatest among ages 12-17.
“All authorized COVID-19 vaccines in the United States are highly effective at protecting people from getting sick or severely ill with COVID-19, including those infected with Delta and other known variants,” the study authors wrote. “Real world data from Texas clearly shows these benefits.”
About 15.6 million people in Texas have been fully vaccinated against COVID-19 in a state of about 29 million residents, according to state data. About 66% of the population has received at least one dose, while 58% is fully vaccinated.
A version of this article first appeared on WebMD.com.
During the month of September, , according to a new study from the Texas Department of State Health Services.
The data also showed that unvaccinated people were 13 times more likely to test positive for COVID-19 than people who were fully vaccinated.
“This analysis quantifies what we’ve known for months,” Jennifer Shuford, MD, the state’s chief epidemiologist, told The Dallas Morning News.
“The COVID-19 vaccines are doing an excellent job of protecting people from getting sick and from dying from COVID-19,” she said. “Vaccination remains the best way to keep yourself and the people close to you safe from this deadly disease.”
As part of the study, researchers analyzed electronic lab reports, death certificates, and state immunization records, with a particular focus on September when the contagious Delta variant surged across Texas. The research marks the state’s first statistical analysis of COVID-19 vaccinations in Texas and the effects, the newspaper reported.
The protective effect of vaccination was most noticeable among younger groups. During September, the risk of COVID-19 death was 23 times higher in unvaccinated people in their 30s and 55 times higher for unvaccinated people in their 40s.
In addition, there were fewer than 10 COVID-19 deaths in September among fully vaccinated people between ages 18-29, as compared with 339 deaths among unvaccinated people in the same age group.
Then, looking at a longer time period -- from Jan. 15 to Oct. 1 -- the researchers found that unvaccinated people were 45 times more likely to contract COVID-19 than fully vaccinated people. The protective effect of vaccination against infection was strong across all adult age groups but greatest among ages 12-17.
“All authorized COVID-19 vaccines in the United States are highly effective at protecting people from getting sick or severely ill with COVID-19, including those infected with Delta and other known variants,” the study authors wrote. “Real world data from Texas clearly shows these benefits.”
About 15.6 million people in Texas have been fully vaccinated against COVID-19 in a state of about 29 million residents, according to state data. About 66% of the population has received at least one dose, while 58% is fully vaccinated.
A version of this article first appeared on WebMD.com.
During the month of September, , according to a new study from the Texas Department of State Health Services.
The data also showed that unvaccinated people were 13 times more likely to test positive for COVID-19 than people who were fully vaccinated.
“This analysis quantifies what we’ve known for months,” Jennifer Shuford, MD, the state’s chief epidemiologist, told The Dallas Morning News.
“The COVID-19 vaccines are doing an excellent job of protecting people from getting sick and from dying from COVID-19,” she said. “Vaccination remains the best way to keep yourself and the people close to you safe from this deadly disease.”
As part of the study, researchers analyzed electronic lab reports, death certificates, and state immunization records, with a particular focus on September when the contagious Delta variant surged across Texas. The research marks the state’s first statistical analysis of COVID-19 vaccinations in Texas and the effects, the newspaper reported.
The protective effect of vaccination was most noticeable among younger groups. During September, the risk of COVID-19 death was 23 times higher in unvaccinated people in their 30s and 55 times higher for unvaccinated people in their 40s.
In addition, there were fewer than 10 COVID-19 deaths in September among fully vaccinated people between ages 18-29, as compared with 339 deaths among unvaccinated people in the same age group.
Then, looking at a longer time period -- from Jan. 15 to Oct. 1 -- the researchers found that unvaccinated people were 45 times more likely to contract COVID-19 than fully vaccinated people. The protective effect of vaccination against infection was strong across all adult age groups but greatest among ages 12-17.
“All authorized COVID-19 vaccines in the United States are highly effective at protecting people from getting sick or severely ill with COVID-19, including those infected with Delta and other known variants,” the study authors wrote. “Real world data from Texas clearly shows these benefits.”
About 15.6 million people in Texas have been fully vaccinated against COVID-19 in a state of about 29 million residents, according to state data. About 66% of the population has received at least one dose, while 58% is fully vaccinated.
A version of this article first appeared on WebMD.com.
Children and COVID: New cases up again after dropping for 8 weeks
As children aged 5-11 years began to receive the first officially approved doses of COVID-19 vaccine, new pediatric cases increased after 8 consecutive weeks of declines, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Weekly cases peaked at almost 252,000 in early September and then dropped for 8 straight weeks before this latest rise, the AAP and the CHA said in their weekly COVID report, which is based on data reported by 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.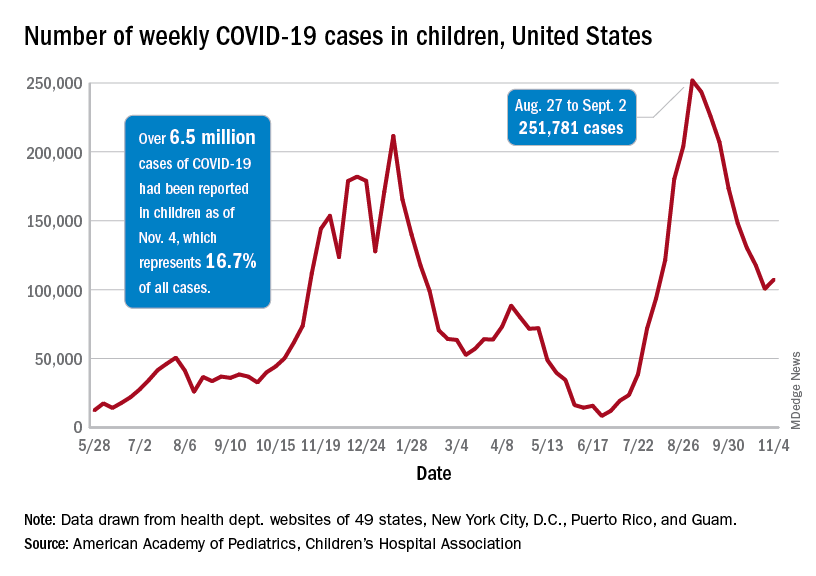
The end of that 8-week drop, unfortunately, allowed another streak to continue: New cases have been above 100,000 for 13 consecutive weeks, the AAP and CHA noted.
The cumulative COVID count in children as of Nov. 4 was 6.5 million, the AAP/CHA said, although that figure does not fully cover Alabama, Nebraska, and Texas, which stopped public reporting over the summer. The Centers for Disease Control and Prevention, with input from all states and territories, puts the total through Nov. 8 at almost 5.7 million cases in children under 18 years of age, while most states define a child as someone aged 0-19 years.
As for the newest group of vaccinees, the CDC said that “updated vaccination data for 5-11 year-olds will be added to COVID Data Tracker later this week,” meaning the week of Nov. 7-13. Currently available data, however, show that almost 157,000 children under age 12 initiated vaccination in the 14 days ending Nov. 8, which was more than those aged 12-15 and 16-17 years combined (127,000).
Among those older groups, the CDC reports that 57.1% of 12- to 15-year-olds have received at least one dose and 47.9% are fully vaccinated, while 64.0% of those aged 16-17 have gotten at least one dose and 55.2% are fully vaccinated. Altogether, about 13.9 million children under age 18 have gotten at least one dose and almost 11.6 million are fully vaccinated, according to the CDC.
As children aged 5-11 years began to receive the first officially approved doses of COVID-19 vaccine, new pediatric cases increased after 8 consecutive weeks of declines, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Weekly cases peaked at almost 252,000 in early September and then dropped for 8 straight weeks before this latest rise, the AAP and the CHA said in their weekly COVID report, which is based on data reported by 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The end of that 8-week drop, unfortunately, allowed another streak to continue: New cases have been above 100,000 for 13 consecutive weeks, the AAP and CHA noted.
The cumulative COVID count in children as of Nov. 4 was 6.5 million, the AAP/CHA said, although that figure does not fully cover Alabama, Nebraska, and Texas, which stopped public reporting over the summer. The Centers for Disease Control and Prevention, with input from all states and territories, puts the total through Nov. 8 at almost 5.7 million cases in children under 18 years of age, while most states define a child as someone aged 0-19 years.
As for the newest group of vaccinees, the CDC said that “updated vaccination data for 5-11 year-olds will be added to COVID Data Tracker later this week,” meaning the week of Nov. 7-13. Currently available data, however, show that almost 157,000 children under age 12 initiated vaccination in the 14 days ending Nov. 8, which was more than those aged 12-15 and 16-17 years combined (127,000).
Among those older groups, the CDC reports that 57.1% of 12- to 15-year-olds have received at least one dose and 47.9% are fully vaccinated, while 64.0% of those aged 16-17 have gotten at least one dose and 55.2% are fully vaccinated. Altogether, about 13.9 million children under age 18 have gotten at least one dose and almost 11.6 million are fully vaccinated, according to the CDC.
As children aged 5-11 years began to receive the first officially approved doses of COVID-19 vaccine, new pediatric cases increased after 8 consecutive weeks of declines, according to the American Academy of Pediatrics and the Children’s Hospital Association.
Weekly cases peaked at almost 252,000 in early September and then dropped for 8 straight weeks before this latest rise, the AAP and the CHA said in their weekly COVID report, which is based on data reported by 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The end of that 8-week drop, unfortunately, allowed another streak to continue: New cases have been above 100,000 for 13 consecutive weeks, the AAP and CHA noted.
The cumulative COVID count in children as of Nov. 4 was 6.5 million, the AAP/CHA said, although that figure does not fully cover Alabama, Nebraska, and Texas, which stopped public reporting over the summer. The Centers for Disease Control and Prevention, with input from all states and territories, puts the total through Nov. 8 at almost 5.7 million cases in children under 18 years of age, while most states define a child as someone aged 0-19 years.
As for the newest group of vaccinees, the CDC said that “updated vaccination data for 5-11 year-olds will be added to COVID Data Tracker later this week,” meaning the week of Nov. 7-13. Currently available data, however, show that almost 157,000 children under age 12 initiated vaccination in the 14 days ending Nov. 8, which was more than those aged 12-15 and 16-17 years combined (127,000).
Among those older groups, the CDC reports that 57.1% of 12- to 15-year-olds have received at least one dose and 47.9% are fully vaccinated, while 64.0% of those aged 16-17 have gotten at least one dose and 55.2% are fully vaccinated. Altogether, about 13.9 million children under age 18 have gotten at least one dose and almost 11.6 million are fully vaccinated, according to the CDC.