User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Early trials underway to test mushrooms as COVID treatment
The U.S. Food and Drug Administration (FDA) approved the MACH-19 trials (the acronym for Mushrooms and Chinese Herbs for COVID-19) after researchers applied for approval in April.
The first two phase 1 randomized, double-blind, placebo-controlled trials have begun at UCLA and the University of California San Diego to treat COVID-19 patients quarantining at home with mild to moderate symptoms. A third trial is investigating the use of medicinal mushrooms as an adjuvant to COVID-19 vaccines.
The researchers have also launched a fourth trial testing the mushrooms against placebo as an adjunct to a COVID booster shot. It looks at the effect in people who have comorbidities that would reduce their vaccine response. An article in JAMA described the trials.
The two mushroom varieties being tested — turkey tail and agarikon — are available as over-the-counter supplements, according to the report. They are a separate class from hallucinogenic or “magic” mushrooms being tested for other uses in medicine.
“They are not even as psychoactive as a cup of tea,” Gordon Saxe, MD, PhD, MPH, principal investigator for the MACH-19 trials, told this news organization.
For each of the MACH-19 treatment trials, researchers plan to recruit 66 people who are quarantined at home with mild to moderate COVID-19 symptoms. Participants will be randomly assigned either to receive the mushroom combination, the Chinese herbs, or a placebo for 2 weeks, according to the JAMA paper.
D. Craig Hopp, PhD, deputy director of the division of extramural research at the National Center for Complementary and Integrative Health (NCCIH), told JAMA in an interview that he was “mildly concerned” about using mushrooms to treat people with active SARS-CoV-2 infection.
“We know that a cytokine storm poses the greatest risk of COVID mortality, not the virus itself,” Dr. Hopp said. “The danger is that an immune-stimulating agent like mushrooms might supercharge an individual’s immune response, leading to a cytokine storm.”
Stephen Wilson, PhD, an immunologist who consulted on the trials when he was chief operating officer of the La Jolla Institute for Immunology, says in the JAMA article that a cytokine storm is unlikely for these patients because the mushroom components “don’t mimic inflammatory cytokines.” Dr. Wilson is now chief innovations officer at Statera Biopharma.
“We think the mushrooms increase the number of immunologic opportunities to better see and respond to a specific threat. In the doses used, the mushrooms perturb the immune system in a good way but fall far short of driving hyper or sustained inflammation,” Dr. Wilson said.
Dr. Saxe said the FDA process was extensive and rigorous and FDA investigators also asked about potential cytokine storms before approving the trials. Cytokine storm is not an issue with a healthy response, Dr. Saxe pointed out. It’s a response that’s not balanced or modulated.
“Mushrooms are immunomodulatory,” he said. “In some ways they very specifically enhance immunity. In other ways they calm down overimmunity.” Dr. Saxe noted that they did a sentinel study for the storm potential “and we didn’t see any evidence for it.”
“Not a crazy concept”
Dr. Saxe pointed out that one of the mushrooms in the combo they use — agarikon — was used to treat pulmonary infections 2,300 years ago.
“Hippocrates, the father of western medicine, used mushrooms,” he said. “Penicillin comes from fungi. It’s not a crazy concept. Most people who oppose this or are skeptics — to some extent, it’s a lack of information.”
Dr. Saxe explained that there are receptors on human cells that bind specific mushroom polysaccharides.
“There’s a hand-in-glove fit there,” Dr. Saxe said, and that’s one way mushrooms can modulate immune cell behavior, which could have an effect against SARS-CoV-2.
Daniel Kuritzkes, MD, chief of the division of infectious diseases at Brigham and Women’s Hospital in Boston, who was not part of the study, told this news organization that he wasn’t surprised the FDA approved moving forward with the trials.
“As long as you can demonstrate that there is a rationale for doing the trial and that you have some safety data or a plan to collect safety data, they are fairly liberal about doing early-phase studies. It would be a much different issue, I think, if they were proposing to do a study for actual licensing or approval of a drug,” Dr. Kuritzkes said.
As yet unanswered, he noted, is which component of the mushrooms or herbs is having the effect. It will be a challenge, he said, to know from one batch of the compound to the next that you have the same amount of material and that it’s going to have the same potency among lots.
Another challenge is how the mushrooms and herbs might interact with other therapies, Dr. Kuritzkes said.
He gave the example of St. John’s Wort, which has been problematic in HIV treatment.
“If someone is on certain HIV medicines and they also are taking St. John’s Wort, they basically are causing the liver to eat up the HIV drug and they don’t get adequate levels of the drug,” he said.
Though there are many challenges ahead, Dr. Kuritzkes acknowledged, but added that “this is a great starting point.”
He, too, pointed out that many traditional medicines were discovered from plants.
“The most famous of these is quinine, which came from cinchona bark that was used to treat malaria.” Dr. Kuritzkes said. Digitalis, often used to treat heart failure, comes from the fox glove plant, he added.
He said it’s important to remember that “people shouldn’t be seeking experimental therapies in place of proven therapies, they should be thinking of them in addition to proven therapies.»
A co-author reports an investment in the dietary supplement company Mycomedica Life Sciences, for which he also serves as an unpaid scientific adviser. Another co-author is a medical consultant for Evergreen Herbs and Medical Supplies. Dr. Hopp, Dr. Saxe, and Dr. Wilson have disclosed no relevant financial relationships. Dr. Kuritzkes consults for Merck, Gilead, and GlaxoSmithKline.
The U.S. Food and Drug Administration (FDA) approved the MACH-19 trials (the acronym for Mushrooms and Chinese Herbs for COVID-19) after researchers applied for approval in April.
The first two phase 1 randomized, double-blind, placebo-controlled trials have begun at UCLA and the University of California San Diego to treat COVID-19 patients quarantining at home with mild to moderate symptoms. A third trial is investigating the use of medicinal mushrooms as an adjuvant to COVID-19 vaccines.
The researchers have also launched a fourth trial testing the mushrooms against placebo as an adjunct to a COVID booster shot. It looks at the effect in people who have comorbidities that would reduce their vaccine response. An article in JAMA described the trials.
The two mushroom varieties being tested — turkey tail and agarikon — are available as over-the-counter supplements, according to the report. They are a separate class from hallucinogenic or “magic” mushrooms being tested for other uses in medicine.
“They are not even as psychoactive as a cup of tea,” Gordon Saxe, MD, PhD, MPH, principal investigator for the MACH-19 trials, told this news organization.
For each of the MACH-19 treatment trials, researchers plan to recruit 66 people who are quarantined at home with mild to moderate COVID-19 symptoms. Participants will be randomly assigned either to receive the mushroom combination, the Chinese herbs, or a placebo for 2 weeks, according to the JAMA paper.
D. Craig Hopp, PhD, deputy director of the division of extramural research at the National Center for Complementary and Integrative Health (NCCIH), told JAMA in an interview that he was “mildly concerned” about using mushrooms to treat people with active SARS-CoV-2 infection.
“We know that a cytokine storm poses the greatest risk of COVID mortality, not the virus itself,” Dr. Hopp said. “The danger is that an immune-stimulating agent like mushrooms might supercharge an individual’s immune response, leading to a cytokine storm.”
Stephen Wilson, PhD, an immunologist who consulted on the trials when he was chief operating officer of the La Jolla Institute for Immunology, says in the JAMA article that a cytokine storm is unlikely for these patients because the mushroom components “don’t mimic inflammatory cytokines.” Dr. Wilson is now chief innovations officer at Statera Biopharma.
“We think the mushrooms increase the number of immunologic opportunities to better see and respond to a specific threat. In the doses used, the mushrooms perturb the immune system in a good way but fall far short of driving hyper or sustained inflammation,” Dr. Wilson said.
Dr. Saxe said the FDA process was extensive and rigorous and FDA investigators also asked about potential cytokine storms before approving the trials. Cytokine storm is not an issue with a healthy response, Dr. Saxe pointed out. It’s a response that’s not balanced or modulated.
“Mushrooms are immunomodulatory,” he said. “In some ways they very specifically enhance immunity. In other ways they calm down overimmunity.” Dr. Saxe noted that they did a sentinel study for the storm potential “and we didn’t see any evidence for it.”
“Not a crazy concept”
Dr. Saxe pointed out that one of the mushrooms in the combo they use — agarikon — was used to treat pulmonary infections 2,300 years ago.
“Hippocrates, the father of western medicine, used mushrooms,” he said. “Penicillin comes from fungi. It’s not a crazy concept. Most people who oppose this or are skeptics — to some extent, it’s a lack of information.”
Dr. Saxe explained that there are receptors on human cells that bind specific mushroom polysaccharides.
“There’s a hand-in-glove fit there,” Dr. Saxe said, and that’s one way mushrooms can modulate immune cell behavior, which could have an effect against SARS-CoV-2.
Daniel Kuritzkes, MD, chief of the division of infectious diseases at Brigham and Women’s Hospital in Boston, who was not part of the study, told this news organization that he wasn’t surprised the FDA approved moving forward with the trials.
“As long as you can demonstrate that there is a rationale for doing the trial and that you have some safety data or a plan to collect safety data, they are fairly liberal about doing early-phase studies. It would be a much different issue, I think, if they were proposing to do a study for actual licensing or approval of a drug,” Dr. Kuritzkes said.
As yet unanswered, he noted, is which component of the mushrooms or herbs is having the effect. It will be a challenge, he said, to know from one batch of the compound to the next that you have the same amount of material and that it’s going to have the same potency among lots.
Another challenge is how the mushrooms and herbs might interact with other therapies, Dr. Kuritzkes said.
He gave the example of St. John’s Wort, which has been problematic in HIV treatment.
“If someone is on certain HIV medicines and they also are taking St. John’s Wort, they basically are causing the liver to eat up the HIV drug and they don’t get adequate levels of the drug,” he said.
Though there are many challenges ahead, Dr. Kuritzkes acknowledged, but added that “this is a great starting point.”
He, too, pointed out that many traditional medicines were discovered from plants.
“The most famous of these is quinine, which came from cinchona bark that was used to treat malaria.” Dr. Kuritzkes said. Digitalis, often used to treat heart failure, comes from the fox glove plant, he added.
He said it’s important to remember that “people shouldn’t be seeking experimental therapies in place of proven therapies, they should be thinking of them in addition to proven therapies.»
A co-author reports an investment in the dietary supplement company Mycomedica Life Sciences, for which he also serves as an unpaid scientific adviser. Another co-author is a medical consultant for Evergreen Herbs and Medical Supplies. Dr. Hopp, Dr. Saxe, and Dr. Wilson have disclosed no relevant financial relationships. Dr. Kuritzkes consults for Merck, Gilead, and GlaxoSmithKline.
The U.S. Food and Drug Administration (FDA) approved the MACH-19 trials (the acronym for Mushrooms and Chinese Herbs for COVID-19) after researchers applied for approval in April.
The first two phase 1 randomized, double-blind, placebo-controlled trials have begun at UCLA and the University of California San Diego to treat COVID-19 patients quarantining at home with mild to moderate symptoms. A third trial is investigating the use of medicinal mushrooms as an adjuvant to COVID-19 vaccines.
The researchers have also launched a fourth trial testing the mushrooms against placebo as an adjunct to a COVID booster shot. It looks at the effect in people who have comorbidities that would reduce their vaccine response. An article in JAMA described the trials.
The two mushroom varieties being tested — turkey tail and agarikon — are available as over-the-counter supplements, according to the report. They are a separate class from hallucinogenic or “magic” mushrooms being tested for other uses in medicine.
“They are not even as psychoactive as a cup of tea,” Gordon Saxe, MD, PhD, MPH, principal investigator for the MACH-19 trials, told this news organization.
For each of the MACH-19 treatment trials, researchers plan to recruit 66 people who are quarantined at home with mild to moderate COVID-19 symptoms. Participants will be randomly assigned either to receive the mushroom combination, the Chinese herbs, or a placebo for 2 weeks, according to the JAMA paper.
D. Craig Hopp, PhD, deputy director of the division of extramural research at the National Center for Complementary and Integrative Health (NCCIH), told JAMA in an interview that he was “mildly concerned” about using mushrooms to treat people with active SARS-CoV-2 infection.
“We know that a cytokine storm poses the greatest risk of COVID mortality, not the virus itself,” Dr. Hopp said. “The danger is that an immune-stimulating agent like mushrooms might supercharge an individual’s immune response, leading to a cytokine storm.”
Stephen Wilson, PhD, an immunologist who consulted on the trials when he was chief operating officer of the La Jolla Institute for Immunology, says in the JAMA article that a cytokine storm is unlikely for these patients because the mushroom components “don’t mimic inflammatory cytokines.” Dr. Wilson is now chief innovations officer at Statera Biopharma.
“We think the mushrooms increase the number of immunologic opportunities to better see and respond to a specific threat. In the doses used, the mushrooms perturb the immune system in a good way but fall far short of driving hyper or sustained inflammation,” Dr. Wilson said.
Dr. Saxe said the FDA process was extensive and rigorous and FDA investigators also asked about potential cytokine storms before approving the trials. Cytokine storm is not an issue with a healthy response, Dr. Saxe pointed out. It’s a response that’s not balanced or modulated.
“Mushrooms are immunomodulatory,” he said. “In some ways they very specifically enhance immunity. In other ways they calm down overimmunity.” Dr. Saxe noted that they did a sentinel study for the storm potential “and we didn’t see any evidence for it.”
“Not a crazy concept”
Dr. Saxe pointed out that one of the mushrooms in the combo they use — agarikon — was used to treat pulmonary infections 2,300 years ago.
“Hippocrates, the father of western medicine, used mushrooms,” he said. “Penicillin comes from fungi. It’s not a crazy concept. Most people who oppose this or are skeptics — to some extent, it’s a lack of information.”
Dr. Saxe explained that there are receptors on human cells that bind specific mushroom polysaccharides.
“There’s a hand-in-glove fit there,” Dr. Saxe said, and that’s one way mushrooms can modulate immune cell behavior, which could have an effect against SARS-CoV-2.
Daniel Kuritzkes, MD, chief of the division of infectious diseases at Brigham and Women’s Hospital in Boston, who was not part of the study, told this news organization that he wasn’t surprised the FDA approved moving forward with the trials.
“As long as you can demonstrate that there is a rationale for doing the trial and that you have some safety data or a plan to collect safety data, they are fairly liberal about doing early-phase studies. It would be a much different issue, I think, if they were proposing to do a study for actual licensing or approval of a drug,” Dr. Kuritzkes said.
As yet unanswered, he noted, is which component of the mushrooms or herbs is having the effect. It will be a challenge, he said, to know from one batch of the compound to the next that you have the same amount of material and that it’s going to have the same potency among lots.
Another challenge is how the mushrooms and herbs might interact with other therapies, Dr. Kuritzkes said.
He gave the example of St. John’s Wort, which has been problematic in HIV treatment.
“If someone is on certain HIV medicines and they also are taking St. John’s Wort, they basically are causing the liver to eat up the HIV drug and they don’t get adequate levels of the drug,” he said.
Though there are many challenges ahead, Dr. Kuritzkes acknowledged, but added that “this is a great starting point.”
He, too, pointed out that many traditional medicines were discovered from plants.
“The most famous of these is quinine, which came from cinchona bark that was used to treat malaria.” Dr. Kuritzkes said. Digitalis, often used to treat heart failure, comes from the fox glove plant, he added.
He said it’s important to remember that “people shouldn’t be seeking experimental therapies in place of proven therapies, they should be thinking of them in addition to proven therapies.»
A co-author reports an investment in the dietary supplement company Mycomedica Life Sciences, for which he also serves as an unpaid scientific adviser. Another co-author is a medical consultant for Evergreen Herbs and Medical Supplies. Dr. Hopp, Dr. Saxe, and Dr. Wilson have disclosed no relevant financial relationships. Dr. Kuritzkes consults for Merck, Gilead, and GlaxoSmithKline.
FROM JAMA
As constituents clamor for ivermectin, Republican politicians embrace the cause
When state senators in South Carolina held two hearings in September about COVID-19 treatments, they got an earful on the benefits of ivermectin — which many of the lawmakers echoed, sharing experiences of their own loved ones.
The demands for access to the drug were loud and insistent, despite federal regulators’ recent warning against using the drug to treat COVID.
Ivermectin is a generic drug that has been used for decades to treat river blindness, scabies, and even head lice. Veterinarians also use it, in different formulations and dosages, to treat animals for parasites like worms.
At one of the South Carolina hearings, Pressley Stutts III reminded the panel that his father, a prominent GOP leader in the state, had died of COVID a month earlier. He believed ivermectin could have helped him. But doctors at the hospital wouldn’t discuss it.
“I went every bit as far as I could without getting myself thrown in jail trying to save my father’s life,” he told the panel, as lawmakers offered condolences.
“What is going on here?” he asked, with the passion in his voice growing. “My dad’s dead!”
After the pandemic began, scientists launched clinical trials to see if ivermectin could help as a treatment for COVID. Some are still ongoing. But providers in mainstream medicine have rejected it as a COVID treatment, citing the poor quality of the studies to date, and two notorious “preprint” studies that were circulated before they were peer-reviewed, and later taken off the internet because of inaccurate and flawed data.
On Aug. 26, the Centers for Disease Control and Prevention advised clinicians not to use ivermectin, citing insufficient evidence of benefit and pointing out that unauthorized use had led to accidental poisonings. Vaccination, the CDC reiterated, is still the best way to avoid serious illness and death from the coronavirus.
But many Americans remain convinced ivermectin could be beneficial, and some politicians appear to be listening to them.
“If we have medications out here that are working — or seem to be working — I think it’s absolutely horrible that we’re not trying them,” said Republican state Sen. Tom Corbin in South Carolina. He questioned doctors who had come to the Statehouse to counter efforts to move ivermectin into mainstream use.
The doctors challenged the implied insult that they weren’t following best practices: “Any implication that any of us would do anything to withhold effective treatments from our patients is really insulting to our profession,” said Dr. Annie Andrews, a professor at the Medical University of South Carolina who has cared for COVID patients throughout the pandemic.
Instead of listening to the medical consensus, some politicians in states like South Carolina seem to be taking cues from doctors on the fringe. During one September hearing, state senators patched in a call from Dr. Pierre Kory.
Last year, Dr. Kory started a nonprofit called the Front Line COVID-19 Critical Care Alliance, which promotes ivermectin. He said he’s not making money by prescribing the drug, though the nonprofit does solicit donations and has not yet filed required financial documents with the IRS.
Dr. Kory acknowledged his medical opinions have landed him on “an island.”
He first testified about ivermectin to a U.S. Senate committee in December. That video went viral. Although it was taken down by YouTube, his Senate testimony prompted patients across the country to ask for ivermectin when they fell ill.
By late August, outpatient prescriptions had jumped 24-fold. Calls to poison control hotlines had tripled, mostly related to people taking ivermectin formulations meant for livestock.
Dr. Kory said he has effectively lost two jobs over his views on ivermectin. At his current hospital in Wisconsin, where he runs the intensive care unit two weeks a month, managers called him to a meeting in September, where he was informed he could no longer prescribe ivermectin. He’d been giving it to “every patient with COVID,” he said.
“After the pharma-geddon that was unleashed, yeah, they shut it down,” he told the South Carolina lawmakers. “And I will tell you that many hospitals across the country had already shut it down months ago.”
Framing the ivermectin fight as a battle against faceless federal agencies and big pharmaceutical corporations appealed to Americans already suspicious of the science behind the pandemic and the approved COVID vaccines.
Dr. Kory suggests success stories with COVID treatments in other parts of the world have been suppressed to instead promote the vaccines.
In an interview with NPR, Dr. Kory said he regrets the flashpoint he helped ignite.
“I feel really bad for the patients, and I feel really bad for the doctors,” he told NPR. “Both of them — both the patients and doctors — are trapped.”
Patients are still demanding the treatment, but doctors sympathetic to their wishes are being told by their health systems not to try it.
Now conservatives in elected office are sensing political payoff if they step in to help patients get the drug. State legislatures, including those in Tennessee and Alaska, are debating various ways to increase access to ivermectin — with proposals such as shielding doctors from repercussions for prescribing it, or forcing pharmacists to fill questionable prescriptions.
The Montana State News Bureau reported that the state’s Republican attorney general dispatched a state trooper to a hospital in Helena where a politically connected patient was dying of COVID. Her family was asking for ivermectin.
In a statement, St. Peter’s Hospital said doctors and nurses were “harassed and threatened by three public officials.”
“These officials have no medical training or experience, yet they were insisting our providers give treatments for COVID-19 that are not authorized, clinically approved, or within the guidelines established by the FDA and the CDC,” the statement added.
On Oct. 14, the Republican attorney general in Nebraska addressed the controversy, issuing a nearly 50-page legal opinion arguing that doctors who consider the “off-label” use of ivermectin and hydroxychloroquine for COVID are acting within the parameters of their state medical licenses, as long as the physician obtains appropriate informed consent from a patient.
Some patients have filed lawsuits to obtain ivermectin, with mixed success. A patient in Illinois was denied. But other hospitals, including one in Ohio, have been forced to administer the drug against the objections of their physicians.
Even as they gain powerful political supporters, some ivermectin fans say they’re now avoiding the health care system — because they’ve lost faith in it.
Lesa Berry, of Richmond, Va., had a friend who died earlier this year of COVID. The doctors refused to use ivermectin, despite requests from Ms. Berry and the patient’s daughter.
They know better now, she said.
“My first attempt would have been to keep her out of the hospital,” Ms. Berry said. “Because right now when you go to the hospital, they only give you what’s on the CDC protocol.”
Ms. Berry and her husband have purchased their own supply of ivermectin, which they keep at home.
This story is from a partnership that includes NPR, Nashville Public Radio and KHN. KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
When state senators in South Carolina held two hearings in September about COVID-19 treatments, they got an earful on the benefits of ivermectin — which many of the lawmakers echoed, sharing experiences of their own loved ones.
The demands for access to the drug were loud and insistent, despite federal regulators’ recent warning against using the drug to treat COVID.
Ivermectin is a generic drug that has been used for decades to treat river blindness, scabies, and even head lice. Veterinarians also use it, in different formulations and dosages, to treat animals for parasites like worms.
At one of the South Carolina hearings, Pressley Stutts III reminded the panel that his father, a prominent GOP leader in the state, had died of COVID a month earlier. He believed ivermectin could have helped him. But doctors at the hospital wouldn’t discuss it.
“I went every bit as far as I could without getting myself thrown in jail trying to save my father’s life,” he told the panel, as lawmakers offered condolences.
“What is going on here?” he asked, with the passion in his voice growing. “My dad’s dead!”
After the pandemic began, scientists launched clinical trials to see if ivermectin could help as a treatment for COVID. Some are still ongoing. But providers in mainstream medicine have rejected it as a COVID treatment, citing the poor quality of the studies to date, and two notorious “preprint” studies that were circulated before they were peer-reviewed, and later taken off the internet because of inaccurate and flawed data.
On Aug. 26, the Centers for Disease Control and Prevention advised clinicians not to use ivermectin, citing insufficient evidence of benefit and pointing out that unauthorized use had led to accidental poisonings. Vaccination, the CDC reiterated, is still the best way to avoid serious illness and death from the coronavirus.
But many Americans remain convinced ivermectin could be beneficial, and some politicians appear to be listening to them.
“If we have medications out here that are working — or seem to be working — I think it’s absolutely horrible that we’re not trying them,” said Republican state Sen. Tom Corbin in South Carolina. He questioned doctors who had come to the Statehouse to counter efforts to move ivermectin into mainstream use.
The doctors challenged the implied insult that they weren’t following best practices: “Any implication that any of us would do anything to withhold effective treatments from our patients is really insulting to our profession,” said Dr. Annie Andrews, a professor at the Medical University of South Carolina who has cared for COVID patients throughout the pandemic.
Instead of listening to the medical consensus, some politicians in states like South Carolina seem to be taking cues from doctors on the fringe. During one September hearing, state senators patched in a call from Dr. Pierre Kory.
Last year, Dr. Kory started a nonprofit called the Front Line COVID-19 Critical Care Alliance, which promotes ivermectin. He said he’s not making money by prescribing the drug, though the nonprofit does solicit donations and has not yet filed required financial documents with the IRS.
Dr. Kory acknowledged his medical opinions have landed him on “an island.”
He first testified about ivermectin to a U.S. Senate committee in December. That video went viral. Although it was taken down by YouTube, his Senate testimony prompted patients across the country to ask for ivermectin when they fell ill.
By late August, outpatient prescriptions had jumped 24-fold. Calls to poison control hotlines had tripled, mostly related to people taking ivermectin formulations meant for livestock.
Dr. Kory said he has effectively lost two jobs over his views on ivermectin. At his current hospital in Wisconsin, where he runs the intensive care unit two weeks a month, managers called him to a meeting in September, where he was informed he could no longer prescribe ivermectin. He’d been giving it to “every patient with COVID,” he said.
“After the pharma-geddon that was unleashed, yeah, they shut it down,” he told the South Carolina lawmakers. “And I will tell you that many hospitals across the country had already shut it down months ago.”
Framing the ivermectin fight as a battle against faceless federal agencies and big pharmaceutical corporations appealed to Americans already suspicious of the science behind the pandemic and the approved COVID vaccines.
Dr. Kory suggests success stories with COVID treatments in other parts of the world have been suppressed to instead promote the vaccines.
In an interview with NPR, Dr. Kory said he regrets the flashpoint he helped ignite.
“I feel really bad for the patients, and I feel really bad for the doctors,” he told NPR. “Both of them — both the patients and doctors — are trapped.”
Patients are still demanding the treatment, but doctors sympathetic to their wishes are being told by their health systems not to try it.
Now conservatives in elected office are sensing political payoff if they step in to help patients get the drug. State legislatures, including those in Tennessee and Alaska, are debating various ways to increase access to ivermectin — with proposals such as shielding doctors from repercussions for prescribing it, or forcing pharmacists to fill questionable prescriptions.
The Montana State News Bureau reported that the state’s Republican attorney general dispatched a state trooper to a hospital in Helena where a politically connected patient was dying of COVID. Her family was asking for ivermectin.
In a statement, St. Peter’s Hospital said doctors and nurses were “harassed and threatened by three public officials.”
“These officials have no medical training or experience, yet they were insisting our providers give treatments for COVID-19 that are not authorized, clinically approved, or within the guidelines established by the FDA and the CDC,” the statement added.
On Oct. 14, the Republican attorney general in Nebraska addressed the controversy, issuing a nearly 50-page legal opinion arguing that doctors who consider the “off-label” use of ivermectin and hydroxychloroquine for COVID are acting within the parameters of their state medical licenses, as long as the physician obtains appropriate informed consent from a patient.
Some patients have filed lawsuits to obtain ivermectin, with mixed success. A patient in Illinois was denied. But other hospitals, including one in Ohio, have been forced to administer the drug against the objections of their physicians.
Even as they gain powerful political supporters, some ivermectin fans say they’re now avoiding the health care system — because they’ve lost faith in it.
Lesa Berry, of Richmond, Va., had a friend who died earlier this year of COVID. The doctors refused to use ivermectin, despite requests from Ms. Berry and the patient’s daughter.
They know better now, she said.
“My first attempt would have been to keep her out of the hospital,” Ms. Berry said. “Because right now when you go to the hospital, they only give you what’s on the CDC protocol.”
Ms. Berry and her husband have purchased their own supply of ivermectin, which they keep at home.
This story is from a partnership that includes NPR, Nashville Public Radio and KHN. KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
When state senators in South Carolina held two hearings in September about COVID-19 treatments, they got an earful on the benefits of ivermectin — which many of the lawmakers echoed, sharing experiences of their own loved ones.
The demands for access to the drug were loud and insistent, despite federal regulators’ recent warning against using the drug to treat COVID.
Ivermectin is a generic drug that has been used for decades to treat river blindness, scabies, and even head lice. Veterinarians also use it, in different formulations and dosages, to treat animals for parasites like worms.
At one of the South Carolina hearings, Pressley Stutts III reminded the panel that his father, a prominent GOP leader in the state, had died of COVID a month earlier. He believed ivermectin could have helped him. But doctors at the hospital wouldn’t discuss it.
“I went every bit as far as I could without getting myself thrown in jail trying to save my father’s life,” he told the panel, as lawmakers offered condolences.
“What is going on here?” he asked, with the passion in his voice growing. “My dad’s dead!”
After the pandemic began, scientists launched clinical trials to see if ivermectin could help as a treatment for COVID. Some are still ongoing. But providers in mainstream medicine have rejected it as a COVID treatment, citing the poor quality of the studies to date, and two notorious “preprint” studies that were circulated before they were peer-reviewed, and later taken off the internet because of inaccurate and flawed data.
On Aug. 26, the Centers for Disease Control and Prevention advised clinicians not to use ivermectin, citing insufficient evidence of benefit and pointing out that unauthorized use had led to accidental poisonings. Vaccination, the CDC reiterated, is still the best way to avoid serious illness and death from the coronavirus.
But many Americans remain convinced ivermectin could be beneficial, and some politicians appear to be listening to them.
“If we have medications out here that are working — or seem to be working — I think it’s absolutely horrible that we’re not trying them,” said Republican state Sen. Tom Corbin in South Carolina. He questioned doctors who had come to the Statehouse to counter efforts to move ivermectin into mainstream use.
The doctors challenged the implied insult that they weren’t following best practices: “Any implication that any of us would do anything to withhold effective treatments from our patients is really insulting to our profession,” said Dr. Annie Andrews, a professor at the Medical University of South Carolina who has cared for COVID patients throughout the pandemic.
Instead of listening to the medical consensus, some politicians in states like South Carolina seem to be taking cues from doctors on the fringe. During one September hearing, state senators patched in a call from Dr. Pierre Kory.
Last year, Dr. Kory started a nonprofit called the Front Line COVID-19 Critical Care Alliance, which promotes ivermectin. He said he’s not making money by prescribing the drug, though the nonprofit does solicit donations and has not yet filed required financial documents with the IRS.
Dr. Kory acknowledged his medical opinions have landed him on “an island.”
He first testified about ivermectin to a U.S. Senate committee in December. That video went viral. Although it was taken down by YouTube, his Senate testimony prompted patients across the country to ask for ivermectin when they fell ill.
By late August, outpatient prescriptions had jumped 24-fold. Calls to poison control hotlines had tripled, mostly related to people taking ivermectin formulations meant for livestock.
Dr. Kory said he has effectively lost two jobs over his views on ivermectin. At his current hospital in Wisconsin, where he runs the intensive care unit two weeks a month, managers called him to a meeting in September, where he was informed he could no longer prescribe ivermectin. He’d been giving it to “every patient with COVID,” he said.
“After the pharma-geddon that was unleashed, yeah, they shut it down,” he told the South Carolina lawmakers. “And I will tell you that many hospitals across the country had already shut it down months ago.”
Framing the ivermectin fight as a battle against faceless federal agencies and big pharmaceutical corporations appealed to Americans already suspicious of the science behind the pandemic and the approved COVID vaccines.
Dr. Kory suggests success stories with COVID treatments in other parts of the world have been suppressed to instead promote the vaccines.
In an interview with NPR, Dr. Kory said he regrets the flashpoint he helped ignite.
“I feel really bad for the patients, and I feel really bad for the doctors,” he told NPR. “Both of them — both the patients and doctors — are trapped.”
Patients are still demanding the treatment, but doctors sympathetic to their wishes are being told by their health systems not to try it.
Now conservatives in elected office are sensing political payoff if they step in to help patients get the drug. State legislatures, including those in Tennessee and Alaska, are debating various ways to increase access to ivermectin — with proposals such as shielding doctors from repercussions for prescribing it, or forcing pharmacists to fill questionable prescriptions.
The Montana State News Bureau reported that the state’s Republican attorney general dispatched a state trooper to a hospital in Helena where a politically connected patient was dying of COVID. Her family was asking for ivermectin.
In a statement, St. Peter’s Hospital said doctors and nurses were “harassed and threatened by three public officials.”
“These officials have no medical training or experience, yet they were insisting our providers give treatments for COVID-19 that are not authorized, clinically approved, or within the guidelines established by the FDA and the CDC,” the statement added.
On Oct. 14, the Republican attorney general in Nebraska addressed the controversy, issuing a nearly 50-page legal opinion arguing that doctors who consider the “off-label” use of ivermectin and hydroxychloroquine for COVID are acting within the parameters of their state medical licenses, as long as the physician obtains appropriate informed consent from a patient.
Some patients have filed lawsuits to obtain ivermectin, with mixed success. A patient in Illinois was denied. But other hospitals, including one in Ohio, have been forced to administer the drug against the objections of their physicians.
Even as they gain powerful political supporters, some ivermectin fans say they’re now avoiding the health care system — because they’ve lost faith in it.
Lesa Berry, of Richmond, Va., had a friend who died earlier this year of COVID. The doctors refused to use ivermectin, despite requests from Ms. Berry and the patient’s daughter.
They know better now, she said.
“My first attempt would have been to keep her out of the hospital,” Ms. Berry said. “Because right now when you go to the hospital, they only give you what’s on the CDC protocol.”
Ms. Berry and her husband have purchased their own supply of ivermectin, which they keep at home.
This story is from a partnership that includes NPR, Nashville Public Radio and KHN. KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Breast milk of COVID-19–infected mothers helps build infant’s immune defenses
It’s rare for mothers with COVID-19 to transfer the infection to their newborns, according to a new small study.
The research, published in JAMA Network Open, found that newborns of mothers infected with the COVID-19 virus were able to develop their own immune defenses via their mother’s breast milk. Researchers detected antibodies in the infants’ saliva.
“It is the first time that this mechanism has been demonstrated,” said study author Rita Carsetti, MD, head of immunology diagnostics for Bambino Gesù Children’s Hospital in Rome. “We now know how breast milk can help babies develop their immune defenses. The system could work the same way for many other pathogens, which are present in the mother during breastfeeding.”
Dr. Carsetti and colleagues examined data from 28 pregnant women who tested positive for COVID-19 and who gave birth at Policlinico Umberto I in Rome between November 2020 and May 2021, and their newborns. They investigated the immune responses of the mothers and their newborns by detecting spike-specific antibodies in serum, and the mucosal immune response was assessed by measuring specific antibodies in maternal breast milk and infant saliva 48 hours after delivery and 2 months later.
Twenty-one mothers and their newborns completed the 2 months of follow-up. Researchers found that the majority of the mothers had mild symptoms of COVID-19, while only three of them were admitted for worsening condition. There was only one reported case of a possible vertical transmission – transmitted in utero – and one case of a horizontal infection through droplets or respiratory secretions, which occurred when the newborn was taken home.
The results of the study showed that antibodies specific to the virus were present in the mothers’ blood at 2 months after delivery, but not at 48 hours. However, in milk, specific antibodies were already present 48 hours after delivery.
Therefore, after 48 hours, the breastfed babies had specific mucosal antibodies against COVID-19 in their saliva that the other newborns did not have. Two months later, these antibodies continued to be present even though the mothers had stopped producing them.
The findings suggest that breast milk offers protection by transferring the antibodies produced by the mother to the baby, but also by helping them to produce their own immune defenses.
“I am not surprised that infants of mothers who had COVID-19 infection in the peripartum period pass anti-spike protein IgA to their infants,” J. Howard Smart, MD, FAAP, who was not involved with the study, said in an interview. “This confirmation is good news for breastfeeding mothers.
“I wonder whether we really know these infants did not become infected, and produce their own antibodies,” said Dr. Smart, chairman of the department of pediatrics at Sharp Rees-Stealy Medical Group in San Diego.
The American College of Obstetricians and Gynecologists said having COVID-19 should not stop mothers from giving their children breast milk. The organization also said that the chance of COVID-19 passing through the breast milk and causing infection in the newborn infant is slim.
“Breast milk also helps protect babies from infections, including infections of the ears, lungs, and digestive system. For these reasons, having COVID-19 should not stop you from giving your baby breast milk,” according to ACOG’s website.
Similar studies on mothers who received the COVID-19 vaccination rather than being infected would be interesting, Dr. Smart added.
The authors of the current study plan to broaden their research by evaluating the response of pregnant mothers vaccinated against SARS-CoV-2 for the presence of antibodies in the milk and the immunity of their newborns. Dr. Carsetti said her team plans to expand the study to other infections, such as cytomegalovirus and respiratory syncytial virus.
None of the researchers or commentators had financial disclosures.
It’s rare for mothers with COVID-19 to transfer the infection to their newborns, according to a new small study.
The research, published in JAMA Network Open, found that newborns of mothers infected with the COVID-19 virus were able to develop their own immune defenses via their mother’s breast milk. Researchers detected antibodies in the infants’ saliva.
“It is the first time that this mechanism has been demonstrated,” said study author Rita Carsetti, MD, head of immunology diagnostics for Bambino Gesù Children’s Hospital in Rome. “We now know how breast milk can help babies develop their immune defenses. The system could work the same way for many other pathogens, which are present in the mother during breastfeeding.”
Dr. Carsetti and colleagues examined data from 28 pregnant women who tested positive for COVID-19 and who gave birth at Policlinico Umberto I in Rome between November 2020 and May 2021, and their newborns. They investigated the immune responses of the mothers and their newborns by detecting spike-specific antibodies in serum, and the mucosal immune response was assessed by measuring specific antibodies in maternal breast milk and infant saliva 48 hours after delivery and 2 months later.
Twenty-one mothers and their newborns completed the 2 months of follow-up. Researchers found that the majority of the mothers had mild symptoms of COVID-19, while only three of them were admitted for worsening condition. There was only one reported case of a possible vertical transmission – transmitted in utero – and one case of a horizontal infection through droplets or respiratory secretions, which occurred when the newborn was taken home.
The results of the study showed that antibodies specific to the virus were present in the mothers’ blood at 2 months after delivery, but not at 48 hours. However, in milk, specific antibodies were already present 48 hours after delivery.
Therefore, after 48 hours, the breastfed babies had specific mucosal antibodies against COVID-19 in their saliva that the other newborns did not have. Two months later, these antibodies continued to be present even though the mothers had stopped producing them.
The findings suggest that breast milk offers protection by transferring the antibodies produced by the mother to the baby, but also by helping them to produce their own immune defenses.
“I am not surprised that infants of mothers who had COVID-19 infection in the peripartum period pass anti-spike protein IgA to their infants,” J. Howard Smart, MD, FAAP, who was not involved with the study, said in an interview. “This confirmation is good news for breastfeeding mothers.
“I wonder whether we really know these infants did not become infected, and produce their own antibodies,” said Dr. Smart, chairman of the department of pediatrics at Sharp Rees-Stealy Medical Group in San Diego.
The American College of Obstetricians and Gynecologists said having COVID-19 should not stop mothers from giving their children breast milk. The organization also said that the chance of COVID-19 passing through the breast milk and causing infection in the newborn infant is slim.
“Breast milk also helps protect babies from infections, including infections of the ears, lungs, and digestive system. For these reasons, having COVID-19 should not stop you from giving your baby breast milk,” according to ACOG’s website.
Similar studies on mothers who received the COVID-19 vaccination rather than being infected would be interesting, Dr. Smart added.
The authors of the current study plan to broaden their research by evaluating the response of pregnant mothers vaccinated against SARS-CoV-2 for the presence of antibodies in the milk and the immunity of their newborns. Dr. Carsetti said her team plans to expand the study to other infections, such as cytomegalovirus and respiratory syncytial virus.
None of the researchers or commentators had financial disclosures.
It’s rare for mothers with COVID-19 to transfer the infection to their newborns, according to a new small study.
The research, published in JAMA Network Open, found that newborns of mothers infected with the COVID-19 virus were able to develop their own immune defenses via their mother’s breast milk. Researchers detected antibodies in the infants’ saliva.
“It is the first time that this mechanism has been demonstrated,” said study author Rita Carsetti, MD, head of immunology diagnostics for Bambino Gesù Children’s Hospital in Rome. “We now know how breast milk can help babies develop their immune defenses. The system could work the same way for many other pathogens, which are present in the mother during breastfeeding.”
Dr. Carsetti and colleagues examined data from 28 pregnant women who tested positive for COVID-19 and who gave birth at Policlinico Umberto I in Rome between November 2020 and May 2021, and their newborns. They investigated the immune responses of the mothers and their newborns by detecting spike-specific antibodies in serum, and the mucosal immune response was assessed by measuring specific antibodies in maternal breast milk and infant saliva 48 hours after delivery and 2 months later.
Twenty-one mothers and their newborns completed the 2 months of follow-up. Researchers found that the majority of the mothers had mild symptoms of COVID-19, while only three of them were admitted for worsening condition. There was only one reported case of a possible vertical transmission – transmitted in utero – and one case of a horizontal infection through droplets or respiratory secretions, which occurred when the newborn was taken home.
The results of the study showed that antibodies specific to the virus were present in the mothers’ blood at 2 months after delivery, but not at 48 hours. However, in milk, specific antibodies were already present 48 hours after delivery.
Therefore, after 48 hours, the breastfed babies had specific mucosal antibodies against COVID-19 in their saliva that the other newborns did not have. Two months later, these antibodies continued to be present even though the mothers had stopped producing them.
The findings suggest that breast milk offers protection by transferring the antibodies produced by the mother to the baby, but also by helping them to produce their own immune defenses.
“I am not surprised that infants of mothers who had COVID-19 infection in the peripartum period pass anti-spike protein IgA to their infants,” J. Howard Smart, MD, FAAP, who was not involved with the study, said in an interview. “This confirmation is good news for breastfeeding mothers.
“I wonder whether we really know these infants did not become infected, and produce their own antibodies,” said Dr. Smart, chairman of the department of pediatrics at Sharp Rees-Stealy Medical Group in San Diego.
The American College of Obstetricians and Gynecologists said having COVID-19 should not stop mothers from giving their children breast milk. The organization also said that the chance of COVID-19 passing through the breast milk and causing infection in the newborn infant is slim.
“Breast milk also helps protect babies from infections, including infections of the ears, lungs, and digestive system. For these reasons, having COVID-19 should not stop you from giving your baby breast milk,” according to ACOG’s website.
Similar studies on mothers who received the COVID-19 vaccination rather than being infected would be interesting, Dr. Smart added.
The authors of the current study plan to broaden their research by evaluating the response of pregnant mothers vaccinated against SARS-CoV-2 for the presence of antibodies in the milk and the immunity of their newborns. Dr. Carsetti said her team plans to expand the study to other infections, such as cytomegalovirus and respiratory syncytial virus.
None of the researchers or commentators had financial disclosures.
FROM JAMA NETWORK OPEN
More than half of people living with HIV have coronary plaque
More than half of people living with HIV and suppressed viral loads nonetheless had imaging-confirmed coronary artery disease – and despite longtime use of HIV drugs that have been associated with cardiovascular trouble, none of those drugs were implicated in disease risk in this study.
“Traditional risk factors and duration of HIV infection were associated with severe coronary artery disease,” said Andreas Knudsen, MD, PhD, an infectious disease provider at Copenhagen University Hospital, Hvidovre, Denmark, during his presentation at the 18th European AIDS Conference. “When we adjusted for time since diagnosis of HIV, none of the drugs remained associated with the severity of coronary artery disease.”
Notably, that included abacavir, which was found in another EACS presentation and in past research to be associated with increased rates of heart attacks. Abacavir is sold individually as a generic as well as a component of Epzicom (abacavir/lamivudine) and the single-drug regimen Triumeq (dolutegravir/abacavir/lamivudine).
The Copenhagen Comorbidity in HIV Infection (COCOMO) study enrolled 1,099 people living with HIV in the Danish capital beginning in 2015, and 705 of them had angiographies via CT available to include in the results. The participants were almost all male (89%), at a healthy weight (BMI of 25), and 96% had undetectable viral loads.
Large minorities of participants also had traditional risk factors for coronary artery disease. More than one in four smoked, one in five had high cholesterol, and 42% had high blood pressure. In addition, many had used drugs that have been associated with cardiovascular trouble, including abacavir, which 26% of participants had used; indinavir, used by 17% of participants; zidovudine/AZT, used by 47%; and didanosine, which 14% used. (While abacavir is still in use, the other three drugs are considered legacy drugs and are not in current use.)
In addition, nearly one in three (29%) were currently using a protease inhibitor, which has been associated with heart failure.
When the investigators looked at participants’ CTs, they found that, by the Coronary Artery Disease-Reporting and Data Systems (CAD-RAMS) scoring system, close to half (46%) had clear arteries with no signs of coronary artery disease. But that also meant that 54% had some blockage or stiffening of the arteries. The good news is that 27% of those people had minimal or mild coronary artery disease.
But a full 17% had confirmed obstructive coronary artery disease, and another 1 in 10 participants had the highest level of blockages. When they broke the data down by traditional and HIV medication–related risk factors for coronary artery disease, they found something interesting. Although obesity was associated with the presence of atherosclerosis, it wasn’t associated with severe disease. But diabetes was the reverse of that: It wasn’t associated with the presence of the disease, but it was associated with more severe disease.
And when they looked at abacavir, they found no relationship between the drug and atherosclerosis. “Abacavir was not associated with the presence of atherosclerosis and was also not associated with severity of disease,” said Dr. Knudsen.
Although past use of AZT, indinavir, and didanosine were associated with severity of atherosclerosis, that association went away when Dr. Knudsen and team adjusted the findings for time since diagnosis. What was associated atherosclerosis was length of time living with HIV itself. For every 5 years a person lived with HIV, the study found the risk of having any atherosclerosis increased 20% and severity increased 23%. In addition, being a man was associated with a nearly 2.5-times increased risk of having any atherosclerosis and a 96% increased chance of having more severe atherosclerosis. Having diabetes was associated with a nearly threefold increased risk of atherosclerosis, as was every additional decade of life for a person who was living with HIV.
The findings confirm the baseline data of the REPRIEVE trial, which recently released data showing similarly high rates of atherosclerotic plaque in people living with HIV who didn’t register as “at risk” for cardiovascular disease using traditional scoring methods.
“It’s important in that it’s a huge study that’s confirmatory [of] what we know, which is that there are high levels of subclinical coronary artery disease in people living with HIV,” said Steven Grinspoon, MD, professor at Harvard Medical School in Boston, Massachusetts, and principal investigator of REPRIEVE.
As for the lack of association between abacavir and cardiovascular risk, he said he’s taking the findings with a grain of salt.
“It’s hard to make a lot out of that,” he said. “It’s hard to know in a cross-sectional study. People put people on different things.”
In Spain, where Jose Ignacio Bernardino, MD, treats people living with HIV at La Paz University Hospital in Madrid, abacavir is mostly a moot point, as clinicians have long since moved away from maintaining people living with HIV on any abacavir-containing regimens. What’s more important in the study, he told this news organization, is that “worrisome” high level of risk. REPRIEVE will test whether statins can reduce heart disease events in people living with HIV. But in the meantime, he said the take-away for clinicians from the study is the primary importance of traditional cardiovascular risk factors.
“We have to acknowledge that the major cardiovascular risk factor is age,” he said. “When patients are approaching their 50s, I usually try to stress a lot about cardiovascular risk factors in general. I stress healthy lifestyle – get physical exercise, hypertension, glucose, lipids – in every single patient.”
Dr. Knudsen and Dr. Bernardino have disclosed no relevant financial relationships. Dr. Grinspoon reports receiving personal and consulting fees from Theratechnologies and ViiV Healthcare.
A version of this article first appeared on Medscape.com.
More than half of people living with HIV and suppressed viral loads nonetheless had imaging-confirmed coronary artery disease – and despite longtime use of HIV drugs that have been associated with cardiovascular trouble, none of those drugs were implicated in disease risk in this study.
“Traditional risk factors and duration of HIV infection were associated with severe coronary artery disease,” said Andreas Knudsen, MD, PhD, an infectious disease provider at Copenhagen University Hospital, Hvidovre, Denmark, during his presentation at the 18th European AIDS Conference. “When we adjusted for time since diagnosis of HIV, none of the drugs remained associated with the severity of coronary artery disease.”
Notably, that included abacavir, which was found in another EACS presentation and in past research to be associated with increased rates of heart attacks. Abacavir is sold individually as a generic as well as a component of Epzicom (abacavir/lamivudine) and the single-drug regimen Triumeq (dolutegravir/abacavir/lamivudine).
The Copenhagen Comorbidity in HIV Infection (COCOMO) study enrolled 1,099 people living with HIV in the Danish capital beginning in 2015, and 705 of them had angiographies via CT available to include in the results. The participants were almost all male (89%), at a healthy weight (BMI of 25), and 96% had undetectable viral loads.
Large minorities of participants also had traditional risk factors for coronary artery disease. More than one in four smoked, one in five had high cholesterol, and 42% had high blood pressure. In addition, many had used drugs that have been associated with cardiovascular trouble, including abacavir, which 26% of participants had used; indinavir, used by 17% of participants; zidovudine/AZT, used by 47%; and didanosine, which 14% used. (While abacavir is still in use, the other three drugs are considered legacy drugs and are not in current use.)
In addition, nearly one in three (29%) were currently using a protease inhibitor, which has been associated with heart failure.
When the investigators looked at participants’ CTs, they found that, by the Coronary Artery Disease-Reporting and Data Systems (CAD-RAMS) scoring system, close to half (46%) had clear arteries with no signs of coronary artery disease. But that also meant that 54% had some blockage or stiffening of the arteries. The good news is that 27% of those people had minimal or mild coronary artery disease.
But a full 17% had confirmed obstructive coronary artery disease, and another 1 in 10 participants had the highest level of blockages. When they broke the data down by traditional and HIV medication–related risk factors for coronary artery disease, they found something interesting. Although obesity was associated with the presence of atherosclerosis, it wasn’t associated with severe disease. But diabetes was the reverse of that: It wasn’t associated with the presence of the disease, but it was associated with more severe disease.
And when they looked at abacavir, they found no relationship between the drug and atherosclerosis. “Abacavir was not associated with the presence of atherosclerosis and was also not associated with severity of disease,” said Dr. Knudsen.
Although past use of AZT, indinavir, and didanosine were associated with severity of atherosclerosis, that association went away when Dr. Knudsen and team adjusted the findings for time since diagnosis. What was associated atherosclerosis was length of time living with HIV itself. For every 5 years a person lived with HIV, the study found the risk of having any atherosclerosis increased 20% and severity increased 23%. In addition, being a man was associated with a nearly 2.5-times increased risk of having any atherosclerosis and a 96% increased chance of having more severe atherosclerosis. Having diabetes was associated with a nearly threefold increased risk of atherosclerosis, as was every additional decade of life for a person who was living with HIV.
The findings confirm the baseline data of the REPRIEVE trial, which recently released data showing similarly high rates of atherosclerotic plaque in people living with HIV who didn’t register as “at risk” for cardiovascular disease using traditional scoring methods.
“It’s important in that it’s a huge study that’s confirmatory [of] what we know, which is that there are high levels of subclinical coronary artery disease in people living with HIV,” said Steven Grinspoon, MD, professor at Harvard Medical School in Boston, Massachusetts, and principal investigator of REPRIEVE.
As for the lack of association between abacavir and cardiovascular risk, he said he’s taking the findings with a grain of salt.
“It’s hard to make a lot out of that,” he said. “It’s hard to know in a cross-sectional study. People put people on different things.”
In Spain, where Jose Ignacio Bernardino, MD, treats people living with HIV at La Paz University Hospital in Madrid, abacavir is mostly a moot point, as clinicians have long since moved away from maintaining people living with HIV on any abacavir-containing regimens. What’s more important in the study, he told this news organization, is that “worrisome” high level of risk. REPRIEVE will test whether statins can reduce heart disease events in people living with HIV. But in the meantime, he said the take-away for clinicians from the study is the primary importance of traditional cardiovascular risk factors.
“We have to acknowledge that the major cardiovascular risk factor is age,” he said. “When patients are approaching their 50s, I usually try to stress a lot about cardiovascular risk factors in general. I stress healthy lifestyle – get physical exercise, hypertension, glucose, lipids – in every single patient.”
Dr. Knudsen and Dr. Bernardino have disclosed no relevant financial relationships. Dr. Grinspoon reports receiving personal and consulting fees from Theratechnologies and ViiV Healthcare.
A version of this article first appeared on Medscape.com.
More than half of people living with HIV and suppressed viral loads nonetheless had imaging-confirmed coronary artery disease – and despite longtime use of HIV drugs that have been associated with cardiovascular trouble, none of those drugs were implicated in disease risk in this study.
“Traditional risk factors and duration of HIV infection were associated with severe coronary artery disease,” said Andreas Knudsen, MD, PhD, an infectious disease provider at Copenhagen University Hospital, Hvidovre, Denmark, during his presentation at the 18th European AIDS Conference. “When we adjusted for time since diagnosis of HIV, none of the drugs remained associated with the severity of coronary artery disease.”
Notably, that included abacavir, which was found in another EACS presentation and in past research to be associated with increased rates of heart attacks. Abacavir is sold individually as a generic as well as a component of Epzicom (abacavir/lamivudine) and the single-drug regimen Triumeq (dolutegravir/abacavir/lamivudine).
The Copenhagen Comorbidity in HIV Infection (COCOMO) study enrolled 1,099 people living with HIV in the Danish capital beginning in 2015, and 705 of them had angiographies via CT available to include in the results. The participants were almost all male (89%), at a healthy weight (BMI of 25), and 96% had undetectable viral loads.
Large minorities of participants also had traditional risk factors for coronary artery disease. More than one in four smoked, one in five had high cholesterol, and 42% had high blood pressure. In addition, many had used drugs that have been associated with cardiovascular trouble, including abacavir, which 26% of participants had used; indinavir, used by 17% of participants; zidovudine/AZT, used by 47%; and didanosine, which 14% used. (While abacavir is still in use, the other three drugs are considered legacy drugs and are not in current use.)
In addition, nearly one in three (29%) were currently using a protease inhibitor, which has been associated with heart failure.
When the investigators looked at participants’ CTs, they found that, by the Coronary Artery Disease-Reporting and Data Systems (CAD-RAMS) scoring system, close to half (46%) had clear arteries with no signs of coronary artery disease. But that also meant that 54% had some blockage or stiffening of the arteries. The good news is that 27% of those people had minimal or mild coronary artery disease.
But a full 17% had confirmed obstructive coronary artery disease, and another 1 in 10 participants had the highest level of blockages. When they broke the data down by traditional and HIV medication–related risk factors for coronary artery disease, they found something interesting. Although obesity was associated with the presence of atherosclerosis, it wasn’t associated with severe disease. But diabetes was the reverse of that: It wasn’t associated with the presence of the disease, but it was associated with more severe disease.
And when they looked at abacavir, they found no relationship between the drug and atherosclerosis. “Abacavir was not associated with the presence of atherosclerosis and was also not associated with severity of disease,” said Dr. Knudsen.
Although past use of AZT, indinavir, and didanosine were associated with severity of atherosclerosis, that association went away when Dr. Knudsen and team adjusted the findings for time since diagnosis. What was associated atherosclerosis was length of time living with HIV itself. For every 5 years a person lived with HIV, the study found the risk of having any atherosclerosis increased 20% and severity increased 23%. In addition, being a man was associated with a nearly 2.5-times increased risk of having any atherosclerosis and a 96% increased chance of having more severe atherosclerosis. Having diabetes was associated with a nearly threefold increased risk of atherosclerosis, as was every additional decade of life for a person who was living with HIV.
The findings confirm the baseline data of the REPRIEVE trial, which recently released data showing similarly high rates of atherosclerotic plaque in people living with HIV who didn’t register as “at risk” for cardiovascular disease using traditional scoring methods.
“It’s important in that it’s a huge study that’s confirmatory [of] what we know, which is that there are high levels of subclinical coronary artery disease in people living with HIV,” said Steven Grinspoon, MD, professor at Harvard Medical School in Boston, Massachusetts, and principal investigator of REPRIEVE.
As for the lack of association between abacavir and cardiovascular risk, he said he’s taking the findings with a grain of salt.
“It’s hard to make a lot out of that,” he said. “It’s hard to know in a cross-sectional study. People put people on different things.”
In Spain, where Jose Ignacio Bernardino, MD, treats people living with HIV at La Paz University Hospital in Madrid, abacavir is mostly a moot point, as clinicians have long since moved away from maintaining people living with HIV on any abacavir-containing regimens. What’s more important in the study, he told this news organization, is that “worrisome” high level of risk. REPRIEVE will test whether statins can reduce heart disease events in people living with HIV. But in the meantime, he said the take-away for clinicians from the study is the primary importance of traditional cardiovascular risk factors.
“We have to acknowledge that the major cardiovascular risk factor is age,” he said. “When patients are approaching their 50s, I usually try to stress a lot about cardiovascular risk factors in general. I stress healthy lifestyle – get physical exercise, hypertension, glucose, lipids – in every single patient.”
Dr. Knudsen and Dr. Bernardino have disclosed no relevant financial relationships. Dr. Grinspoon reports receiving personal and consulting fees from Theratechnologies and ViiV Healthcare.
A version of this article first appeared on Medscape.com.
Expected spike in acute flaccid myelitis did not occur in 2020
suggested researchers at the Centers for Disease Control and Prevention.
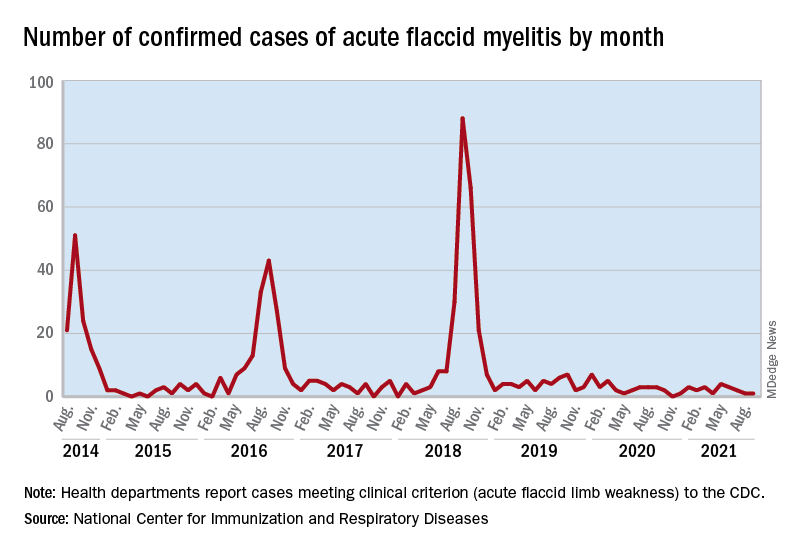
Acute flaccid myelitis (AFM) is an uncommon but serious complication of some viral infections, including West Nile virus and nonpolio enteroviruses. It is “characterized by sudden onset of limb weakness and lesions in the gray matter of the spinal cord,” they said, and more than 90% of cases occur in young children.
Cases of AFM, which can lead to respiratory insufficiency and permanent paralysis, spiked during the late summer and early fall in 2014, 2016, and 2018 and were expected to do so again in 2020, Sarah Kidd, MD, and associates at the division of viral diseases at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said in the Morbidity and Mortality Weekly Report.
Monthly peaks in those previous years – each occurring in September – reached 51 cases in 2014, 43 cases in 2016, and 88 cases in 2018, but in 2020 there was only 1 case reported in September, with a high of 4 coming in May, CDC data show. The total number of cases for 2020 (32) was, in fact, lower than in 2019, when 47 were reported.
The investigators’ main objective was to see if there were any differences between the 2018 and 2019-2020 cases. Reports from state health departments to the CDC showed that, in 2019-2020, “patients were older; more likely to have lower limb involvement; and less likely to have upper limb involvement, prodromal illness, [cerebrospinal fluid] pleocytosis, or specimens that tested positive for EV [enterovirus]-D68” than patients from 2018, Dr. Kidd and associates said.
Mask wearing and reduced in-school attendance may have decreased circulation of EV-D68 – the enterovirus type most often detected in the stool and respiratory specimens of AFM patients – as was seen with other respiratory viruses, such as influenza and respiratory syncytial virus, in 2020. Previous studies have suggested that EV-D68 drives the increases in cases during peak years, the researchers noted.
The absence of such an increase “in 2020 reflects a deviation from the previously observed biennial pattern, and it is unclear when the next increase in AFM should be expected. Clinicians should continue to maintain vigilance and suspect AFM in any child with acute flaccid limb weakness, particularly in the setting of recent febrile or respiratory illness,” they wrote.
suggested researchers at the Centers for Disease Control and Prevention.

Acute flaccid myelitis (AFM) is an uncommon but serious complication of some viral infections, including West Nile virus and nonpolio enteroviruses. It is “characterized by sudden onset of limb weakness and lesions in the gray matter of the spinal cord,” they said, and more than 90% of cases occur in young children.
Cases of AFM, which can lead to respiratory insufficiency and permanent paralysis, spiked during the late summer and early fall in 2014, 2016, and 2018 and were expected to do so again in 2020, Sarah Kidd, MD, and associates at the division of viral diseases at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said in the Morbidity and Mortality Weekly Report.
Monthly peaks in those previous years – each occurring in September – reached 51 cases in 2014, 43 cases in 2016, and 88 cases in 2018, but in 2020 there was only 1 case reported in September, with a high of 4 coming in May, CDC data show. The total number of cases for 2020 (32) was, in fact, lower than in 2019, when 47 were reported.
The investigators’ main objective was to see if there were any differences between the 2018 and 2019-2020 cases. Reports from state health departments to the CDC showed that, in 2019-2020, “patients were older; more likely to have lower limb involvement; and less likely to have upper limb involvement, prodromal illness, [cerebrospinal fluid] pleocytosis, or specimens that tested positive for EV [enterovirus]-D68” than patients from 2018, Dr. Kidd and associates said.
Mask wearing and reduced in-school attendance may have decreased circulation of EV-D68 – the enterovirus type most often detected in the stool and respiratory specimens of AFM patients – as was seen with other respiratory viruses, such as influenza and respiratory syncytial virus, in 2020. Previous studies have suggested that EV-D68 drives the increases in cases during peak years, the researchers noted.
The absence of such an increase “in 2020 reflects a deviation from the previously observed biennial pattern, and it is unclear when the next increase in AFM should be expected. Clinicians should continue to maintain vigilance and suspect AFM in any child with acute flaccid limb weakness, particularly in the setting of recent febrile or respiratory illness,” they wrote.
suggested researchers at the Centers for Disease Control and Prevention.

Acute flaccid myelitis (AFM) is an uncommon but serious complication of some viral infections, including West Nile virus and nonpolio enteroviruses. It is “characterized by sudden onset of limb weakness and lesions in the gray matter of the spinal cord,” they said, and more than 90% of cases occur in young children.
Cases of AFM, which can lead to respiratory insufficiency and permanent paralysis, spiked during the late summer and early fall in 2014, 2016, and 2018 and were expected to do so again in 2020, Sarah Kidd, MD, and associates at the division of viral diseases at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said in the Morbidity and Mortality Weekly Report.
Monthly peaks in those previous years – each occurring in September – reached 51 cases in 2014, 43 cases in 2016, and 88 cases in 2018, but in 2020 there was only 1 case reported in September, with a high of 4 coming in May, CDC data show. The total number of cases for 2020 (32) was, in fact, lower than in 2019, when 47 were reported.
The investigators’ main objective was to see if there were any differences between the 2018 and 2019-2020 cases. Reports from state health departments to the CDC showed that, in 2019-2020, “patients were older; more likely to have lower limb involvement; and less likely to have upper limb involvement, prodromal illness, [cerebrospinal fluid] pleocytosis, or specimens that tested positive for EV [enterovirus]-D68” than patients from 2018, Dr. Kidd and associates said.
Mask wearing and reduced in-school attendance may have decreased circulation of EV-D68 – the enterovirus type most often detected in the stool and respiratory specimens of AFM patients – as was seen with other respiratory viruses, such as influenza and respiratory syncytial virus, in 2020. Previous studies have suggested that EV-D68 drives the increases in cases during peak years, the researchers noted.
The absence of such an increase “in 2020 reflects a deviation from the previously observed biennial pattern, and it is unclear when the next increase in AFM should be expected. Clinicians should continue to maintain vigilance and suspect AFM in any child with acute flaccid limb weakness, particularly in the setting of recent febrile or respiratory illness,” they wrote.
FROM MMWR
Does zinc really help treat colds?
A new study published in BMJ Open adds to the evidence that zinc is effective against viral respiratory infections, such as colds.
Jennifer Hunter, PhD, BMed, of Western Sydney University’s NICM Health Research Institute, New South Wales, Australia, and colleagues conducted a meta-analysis of 28 randomized controlled trials (RCTs). They searched 17 English and Chinese databases to identify the trials and then used the Cochrane rapid review technique for the analysis.
The trials included 5,446 adults who had received zinc in a variety of formulations and routes — oral, sublingual, and nasal spray. The researchers separately analyzed whether zinc prevented or treated respiratory tract infections (RTIs)
Oral or intranasal zinc prevented five RTIs per 100 person-months (95% CI, 1 – 8; numbers needed to treat, 20). There was a 32% lower relative risk (RR) of developing mild to moderate symptoms consistent with a viral RTI.
Use of zinc was also associated with an 87% lower risk of developing moderately severe symptoms (incidence rate ratio, 0.13; 95% CI, 0.04 – 0.38) and a 28% lower risk of developing milder symptoms. The largest reductions in RR were for moderately severe symptoms consistent with an influenza-like illness.
Symptoms resolved 2 days earlier with sublingual or intranasal zinc compared with placebo (95% CI, 0.61 – 3.50; very low-certainty quality of evidence). There were clinically significant reductions in day 3 symptom severity scores (mean difference, -1.20 points; 95% CI, -0.66 to -1.74; low-certainty quality of evidence) but not in overall symptom severity. Participants who used sublingual or topical nasal zinc early in the course of illness were 1.8 times more likely to recover before those who used a placebo.
However, the investigators found no benefit of zinc when patients were inoculated with rhinovirus; there was no reduction in the risk of developing a cold. Asked about this disparity, Dr. Hunter said, “It might well be that when inoculating people to make sure they get infected, you give them a really high dose of the virus. [This] doesn’t really mimic what happens in the real world.”
On the downside of supplemental zinc, there were more side effects among those who used zinc, including nausea or gastrointestinal discomfort, mouth irritation, or soreness from sublingual lozenges (RR, 1.41; 95% CI, 1.17 – 1.69; number needed to harm, 7; moderate-certainty quality of evidence). The risk for a serious adverse event, such as loss of smell or copper deficiency, was low. Although not found in these studies, postmarketing studies have found that there is a risk for severe and in some cases permanent loss of smell associated with the use of nasal gels or sprays containing zinc. Three such products were recalled from the market.
The trial could not provide answers about the comparative efficacy of different types of zinc formulations, nor could the investigators recommend specific doses. The trial was not designed to assess zinc for the prevention or treatment of COVID-19.
Asked for independent comment, pediatrician Aamer Imdad, MBBS, assistant professor at the State University of New York Upstate Medical University, Syracuse, told this news organization, “It’s a very comprehensive review for zinc-related studies in adults” but was challenging because of the “significant clinical heterogeneity in the population.”
Dr. Imdad explained that zinc has “absolutely” been shown to be effective for children with diarrhea. The World Health Organization has recommended it since 2004. “The way it works in diarrhea is that it helps with the regeneration of the epithelium.... It also improves the immunity itself, especially the cell-mediated immunity.” He raised the question of whether it might work similarly in the respiratory tract. Dr. Imdad has a long-standing interest in the use of zinc for pediatric infections. Regarding this study, he concluded, “I think we still need to know the nuts and bolts of this intervention before we can recommend it more specifically.”
Dr. Hunter said, “We don’t have any high-quality studies that have evaluated zinc orally as treatment once you’re actually infected and have symptoms of the cold or influenza, or COVID.”
Asked about zinc’s possible role, Dr. Hunter said, “So I do think it gives us a viable alternative. More people are going, ‘What can I do?’ And you know as well as I do people come to you, and [they say], ‘Well, just give me something. Even if it’s a day or a little bit of symptom relief, anything to make me feel better that isn’t going to hurt me and doesn’t have any major risks.’ So I think in the short term, clinicians and consumers can consider trying it.”
Dr. Hunter was not keen on giving zinc to family members after they develop an RTI: “Consider it. But I don’t think we have enough evidence to say definitely yes.” But she does see a potential role for “people who are at risk of suboptimal zinc absorption, like people who are taking a variety of pharmaceuticals [notably proton pump inhibitors] that block or reduce the absorption of zinc, people with a whole lot of the chronic diseases that we know are associated with an increased risk of worse outcomes from respiratory viral infections, and older adults. Yes, I think [for] those high-risk groups, you could consider using zinc, either in a moderate dose longer term or in a higher dose for very short bursts of, like, 1 to 2 weeks.”
Dr. Hunter concluded, “Up until now, we all commonly thought that zinc’s role was only for people who were zinc deficient, and now we’ve got some signals pointing towards its potential role as an anti-infective and anti-inflammatory agent in people who don’t have zinc deficiency.”
But both Dr. Hunter and Dr. Imdad emphasized that zinc is not a game changer. There is a hint that it produces a small benefit in prevention and may slightly shorten the duration of RTIs. More research is needed.
Dr. Hunter has received payment for providing expert advice about traditional, complementary, and integrative medicine, including nutraceuticals, to industry, government bodies, and nongovernmental organizations and has spoken at workshops, seminars, and conferences for which registration, travel, and/or accommodation has been paid for by the organizers. Dr. Imdad has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study published in BMJ Open adds to the evidence that zinc is effective against viral respiratory infections, such as colds.
Jennifer Hunter, PhD, BMed, of Western Sydney University’s NICM Health Research Institute, New South Wales, Australia, and colleagues conducted a meta-analysis of 28 randomized controlled trials (RCTs). They searched 17 English and Chinese databases to identify the trials and then used the Cochrane rapid review technique for the analysis.
The trials included 5,446 adults who had received zinc in a variety of formulations and routes — oral, sublingual, and nasal spray. The researchers separately analyzed whether zinc prevented or treated respiratory tract infections (RTIs)
Oral or intranasal zinc prevented five RTIs per 100 person-months (95% CI, 1 – 8; numbers needed to treat, 20). There was a 32% lower relative risk (RR) of developing mild to moderate symptoms consistent with a viral RTI.
Use of zinc was also associated with an 87% lower risk of developing moderately severe symptoms (incidence rate ratio, 0.13; 95% CI, 0.04 – 0.38) and a 28% lower risk of developing milder symptoms. The largest reductions in RR were for moderately severe symptoms consistent with an influenza-like illness.
Symptoms resolved 2 days earlier with sublingual or intranasal zinc compared with placebo (95% CI, 0.61 – 3.50; very low-certainty quality of evidence). There were clinically significant reductions in day 3 symptom severity scores (mean difference, -1.20 points; 95% CI, -0.66 to -1.74; low-certainty quality of evidence) but not in overall symptom severity. Participants who used sublingual or topical nasal zinc early in the course of illness were 1.8 times more likely to recover before those who used a placebo.
However, the investigators found no benefit of zinc when patients were inoculated with rhinovirus; there was no reduction in the risk of developing a cold. Asked about this disparity, Dr. Hunter said, “It might well be that when inoculating people to make sure they get infected, you give them a really high dose of the virus. [This] doesn’t really mimic what happens in the real world.”
On the downside of supplemental zinc, there were more side effects among those who used zinc, including nausea or gastrointestinal discomfort, mouth irritation, or soreness from sublingual lozenges (RR, 1.41; 95% CI, 1.17 – 1.69; number needed to harm, 7; moderate-certainty quality of evidence). The risk for a serious adverse event, such as loss of smell or copper deficiency, was low. Although not found in these studies, postmarketing studies have found that there is a risk for severe and in some cases permanent loss of smell associated with the use of nasal gels or sprays containing zinc. Three such products were recalled from the market.
The trial could not provide answers about the comparative efficacy of different types of zinc formulations, nor could the investigators recommend specific doses. The trial was not designed to assess zinc for the prevention or treatment of COVID-19.
Asked for independent comment, pediatrician Aamer Imdad, MBBS, assistant professor at the State University of New York Upstate Medical University, Syracuse, told this news organization, “It’s a very comprehensive review for zinc-related studies in adults” but was challenging because of the “significant clinical heterogeneity in the population.”
Dr. Imdad explained that zinc has “absolutely” been shown to be effective for children with diarrhea. The World Health Organization has recommended it since 2004. “The way it works in diarrhea is that it helps with the regeneration of the epithelium.... It also improves the immunity itself, especially the cell-mediated immunity.” He raised the question of whether it might work similarly in the respiratory tract. Dr. Imdad has a long-standing interest in the use of zinc for pediatric infections. Regarding this study, he concluded, “I think we still need to know the nuts and bolts of this intervention before we can recommend it more specifically.”
Dr. Hunter said, “We don’t have any high-quality studies that have evaluated zinc orally as treatment once you’re actually infected and have symptoms of the cold or influenza, or COVID.”
Asked about zinc’s possible role, Dr. Hunter said, “So I do think it gives us a viable alternative. More people are going, ‘What can I do?’ And you know as well as I do people come to you, and [they say], ‘Well, just give me something. Even if it’s a day or a little bit of symptom relief, anything to make me feel better that isn’t going to hurt me and doesn’t have any major risks.’ So I think in the short term, clinicians and consumers can consider trying it.”
Dr. Hunter was not keen on giving zinc to family members after they develop an RTI: “Consider it. But I don’t think we have enough evidence to say definitely yes.” But she does see a potential role for “people who are at risk of suboptimal zinc absorption, like people who are taking a variety of pharmaceuticals [notably proton pump inhibitors] that block or reduce the absorption of zinc, people with a whole lot of the chronic diseases that we know are associated with an increased risk of worse outcomes from respiratory viral infections, and older adults. Yes, I think [for] those high-risk groups, you could consider using zinc, either in a moderate dose longer term or in a higher dose for very short bursts of, like, 1 to 2 weeks.”
Dr. Hunter concluded, “Up until now, we all commonly thought that zinc’s role was only for people who were zinc deficient, and now we’ve got some signals pointing towards its potential role as an anti-infective and anti-inflammatory agent in people who don’t have zinc deficiency.”
But both Dr. Hunter and Dr. Imdad emphasized that zinc is not a game changer. There is a hint that it produces a small benefit in prevention and may slightly shorten the duration of RTIs. More research is needed.
Dr. Hunter has received payment for providing expert advice about traditional, complementary, and integrative medicine, including nutraceuticals, to industry, government bodies, and nongovernmental organizations and has spoken at workshops, seminars, and conferences for which registration, travel, and/or accommodation has been paid for by the organizers. Dr. Imdad has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study published in BMJ Open adds to the evidence that zinc is effective against viral respiratory infections, such as colds.
Jennifer Hunter, PhD, BMed, of Western Sydney University’s NICM Health Research Institute, New South Wales, Australia, and colleagues conducted a meta-analysis of 28 randomized controlled trials (RCTs). They searched 17 English and Chinese databases to identify the trials and then used the Cochrane rapid review technique for the analysis.
The trials included 5,446 adults who had received zinc in a variety of formulations and routes — oral, sublingual, and nasal spray. The researchers separately analyzed whether zinc prevented or treated respiratory tract infections (RTIs)
Oral or intranasal zinc prevented five RTIs per 100 person-months (95% CI, 1 – 8; numbers needed to treat, 20). There was a 32% lower relative risk (RR) of developing mild to moderate symptoms consistent with a viral RTI.
Use of zinc was also associated with an 87% lower risk of developing moderately severe symptoms (incidence rate ratio, 0.13; 95% CI, 0.04 – 0.38) and a 28% lower risk of developing milder symptoms. The largest reductions in RR were for moderately severe symptoms consistent with an influenza-like illness.
Symptoms resolved 2 days earlier with sublingual or intranasal zinc compared with placebo (95% CI, 0.61 – 3.50; very low-certainty quality of evidence). There were clinically significant reductions in day 3 symptom severity scores (mean difference, -1.20 points; 95% CI, -0.66 to -1.74; low-certainty quality of evidence) but not in overall symptom severity. Participants who used sublingual or topical nasal zinc early in the course of illness were 1.8 times more likely to recover before those who used a placebo.
However, the investigators found no benefit of zinc when patients were inoculated with rhinovirus; there was no reduction in the risk of developing a cold. Asked about this disparity, Dr. Hunter said, “It might well be that when inoculating people to make sure they get infected, you give them a really high dose of the virus. [This] doesn’t really mimic what happens in the real world.”
On the downside of supplemental zinc, there were more side effects among those who used zinc, including nausea or gastrointestinal discomfort, mouth irritation, or soreness from sublingual lozenges (RR, 1.41; 95% CI, 1.17 – 1.69; number needed to harm, 7; moderate-certainty quality of evidence). The risk for a serious adverse event, such as loss of smell or copper deficiency, was low. Although not found in these studies, postmarketing studies have found that there is a risk for severe and in some cases permanent loss of smell associated with the use of nasal gels or sprays containing zinc. Three such products were recalled from the market.
The trial could not provide answers about the comparative efficacy of different types of zinc formulations, nor could the investigators recommend specific doses. The trial was not designed to assess zinc for the prevention or treatment of COVID-19.
Asked for independent comment, pediatrician Aamer Imdad, MBBS, assistant professor at the State University of New York Upstate Medical University, Syracuse, told this news organization, “It’s a very comprehensive review for zinc-related studies in adults” but was challenging because of the “significant clinical heterogeneity in the population.”
Dr. Imdad explained that zinc has “absolutely” been shown to be effective for children with diarrhea. The World Health Organization has recommended it since 2004. “The way it works in diarrhea is that it helps with the regeneration of the epithelium.... It also improves the immunity itself, especially the cell-mediated immunity.” He raised the question of whether it might work similarly in the respiratory tract. Dr. Imdad has a long-standing interest in the use of zinc for pediatric infections. Regarding this study, he concluded, “I think we still need to know the nuts and bolts of this intervention before we can recommend it more specifically.”
Dr. Hunter said, “We don’t have any high-quality studies that have evaluated zinc orally as treatment once you’re actually infected and have symptoms of the cold or influenza, or COVID.”
Asked about zinc’s possible role, Dr. Hunter said, “So I do think it gives us a viable alternative. More people are going, ‘What can I do?’ And you know as well as I do people come to you, and [they say], ‘Well, just give me something. Even if it’s a day or a little bit of symptom relief, anything to make me feel better that isn’t going to hurt me and doesn’t have any major risks.’ So I think in the short term, clinicians and consumers can consider trying it.”
Dr. Hunter was not keen on giving zinc to family members after they develop an RTI: “Consider it. But I don’t think we have enough evidence to say definitely yes.” But she does see a potential role for “people who are at risk of suboptimal zinc absorption, like people who are taking a variety of pharmaceuticals [notably proton pump inhibitors] that block or reduce the absorption of zinc, people with a whole lot of the chronic diseases that we know are associated with an increased risk of worse outcomes from respiratory viral infections, and older adults. Yes, I think [for] those high-risk groups, you could consider using zinc, either in a moderate dose longer term or in a higher dose for very short bursts of, like, 1 to 2 weeks.”
Dr. Hunter concluded, “Up until now, we all commonly thought that zinc’s role was only for people who were zinc deficient, and now we’ve got some signals pointing towards its potential role as an anti-infective and anti-inflammatory agent in people who don’t have zinc deficiency.”
But both Dr. Hunter and Dr. Imdad emphasized that zinc is not a game changer. There is a hint that it produces a small benefit in prevention and may slightly shorten the duration of RTIs. More research is needed.
Dr. Hunter has received payment for providing expert advice about traditional, complementary, and integrative medicine, including nutraceuticals, to industry, government bodies, and nongovernmental organizations and has spoken at workshops, seminars, and conferences for which registration, travel, and/or accommodation has been paid for by the organizers. Dr. Imdad has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM BMJ OPEN
COVID vaccines’ protection dropped sharply over 6 months: Study
, a study of almost 800,000 veterans found.
The study, published in the journal Science ., says the three vaccines offered about the same protection against the virus in March, when the Delta variant was first detected in the United States, but that changed 6 months later.
The Moderna two-dose vaccine went from being 89% effective in March to 58% effective in September, according to a story about the study in theLos Angeles Times.
Meanwhile, the Pfizer/BioNTech vaccine went from being 87% effective to 45% effective over the same time period.
The Johnson & Johnson vaccine showed the biggest drop -- from 86% effectiveness to 13% over those 6 months.
“In summary, although vaccination remains protective against SARS-CoV-2 infection, protection waned as the Delta variant emerged in the U.S., and this decline did not differ by age,” the study said.
The three vaccines also lost effectiveness in the ability to protect against death in veterans 65 and over after only 3 months, the Los Angeles Times reported.
Compared to unvaccinated veterans in that age group, veterans who got the Moderna vaccine and had a breakthrough case were 76% less likely to die of COVID-19 by July.
The protection was 70% for Pfizer/BioNTech vaccine recipients and 52% for J&J vaccine recipients for the same age group, compared to unvaccinated veterans, according to the newspaper.
For veterans under 65, the protectiveness against a fatal case of COVID was 84% for Pfizer/BioNTech recipients, 82% for Moderna recipients, and 73% for J&J recipients, compared to unvaccinated veterans in that age group.
The study confirms the need for booster vaccines and protective measures such as vaccine passports, vaccine mandates, masking, hand-washing, and social distancing, the researchers said.
Of the veterans studied, about 500,000 were vaccinated and 300,000 were not. Researchers noted that the study population had 6 times as many men as women. About 48% of the study group was 65 or older, 29% was 50-64, while 24% was under 50.
Researchers from the Public Health Institute in Oakland, the Veterans Affairs Medical Center in San Francisco, and the University of Texas Health Science Center conducted the study.
A version of this article first appeared on WebMD.com.
, a study of almost 800,000 veterans found.
The study, published in the journal Science ., says the three vaccines offered about the same protection against the virus in March, when the Delta variant was first detected in the United States, but that changed 6 months later.
The Moderna two-dose vaccine went from being 89% effective in March to 58% effective in September, according to a story about the study in theLos Angeles Times.
Meanwhile, the Pfizer/BioNTech vaccine went from being 87% effective to 45% effective over the same time period.
The Johnson & Johnson vaccine showed the biggest drop -- from 86% effectiveness to 13% over those 6 months.
“In summary, although vaccination remains protective against SARS-CoV-2 infection, protection waned as the Delta variant emerged in the U.S., and this decline did not differ by age,” the study said.
The three vaccines also lost effectiveness in the ability to protect against death in veterans 65 and over after only 3 months, the Los Angeles Times reported.
Compared to unvaccinated veterans in that age group, veterans who got the Moderna vaccine and had a breakthrough case were 76% less likely to die of COVID-19 by July.
The protection was 70% for Pfizer/BioNTech vaccine recipients and 52% for J&J vaccine recipients for the same age group, compared to unvaccinated veterans, according to the newspaper.
For veterans under 65, the protectiveness against a fatal case of COVID was 84% for Pfizer/BioNTech recipients, 82% for Moderna recipients, and 73% for J&J recipients, compared to unvaccinated veterans in that age group.
The study confirms the need for booster vaccines and protective measures such as vaccine passports, vaccine mandates, masking, hand-washing, and social distancing, the researchers said.
Of the veterans studied, about 500,000 were vaccinated and 300,000 were not. Researchers noted that the study population had 6 times as many men as women. About 48% of the study group was 65 or older, 29% was 50-64, while 24% was under 50.
Researchers from the Public Health Institute in Oakland, the Veterans Affairs Medical Center in San Francisco, and the University of Texas Health Science Center conducted the study.
A version of this article first appeared on WebMD.com.
, a study of almost 800,000 veterans found.
The study, published in the journal Science ., says the three vaccines offered about the same protection against the virus in March, when the Delta variant was first detected in the United States, but that changed 6 months later.
The Moderna two-dose vaccine went from being 89% effective in March to 58% effective in September, according to a story about the study in theLos Angeles Times.
Meanwhile, the Pfizer/BioNTech vaccine went from being 87% effective to 45% effective over the same time period.
The Johnson & Johnson vaccine showed the biggest drop -- from 86% effectiveness to 13% over those 6 months.
“In summary, although vaccination remains protective against SARS-CoV-2 infection, protection waned as the Delta variant emerged in the U.S., and this decline did not differ by age,” the study said.
The three vaccines also lost effectiveness in the ability to protect against death in veterans 65 and over after only 3 months, the Los Angeles Times reported.
Compared to unvaccinated veterans in that age group, veterans who got the Moderna vaccine and had a breakthrough case were 76% less likely to die of COVID-19 by July.
The protection was 70% for Pfizer/BioNTech vaccine recipients and 52% for J&J vaccine recipients for the same age group, compared to unvaccinated veterans, according to the newspaper.
For veterans under 65, the protectiveness against a fatal case of COVID was 84% for Pfizer/BioNTech recipients, 82% for Moderna recipients, and 73% for J&J recipients, compared to unvaccinated veterans in that age group.
The study confirms the need for booster vaccines and protective measures such as vaccine passports, vaccine mandates, masking, hand-washing, and social distancing, the researchers said.
Of the veterans studied, about 500,000 were vaccinated and 300,000 were not. Researchers noted that the study population had 6 times as many men as women. About 48% of the study group was 65 or older, 29% was 50-64, while 24% was under 50.
Researchers from the Public Health Institute in Oakland, the Veterans Affairs Medical Center in San Francisco, and the University of Texas Health Science Center conducted the study.
A version of this article first appeared on WebMD.com.
FROM SCIENCE
New transmission information should motivate hospitals to reexamine aerosol procedures, researchers say
Two studies published in Thorax have found that the use of continuous positive airways pressure (CPAP) or high-flow nasal oxygen (HFNO) to treat moderate to severe COVID-19 is not linked to a heightened risk of infection, as currently thought. Researchers say hospitals should use this information to re-examine aerosol procedures in regard to risk of transmission of SARS-CoV-2.
CPAP and HFNO have been thought to generate virus particles capable of contaminating the air and surfaces, necessitating additional infection control precautions such as segregating patients. However, this research demonstrates that both methods produced little measurable air or surface viral contamination. The amount of contamination was no more than with the use of supplemental oxygen and less than that produced when breathing, speaking, or coughing.
In one study, led by a team from the North Bristol NHS Trust, 25 healthy volunteers and eight hospitalized patients with COVID-19 were recruited and asked to breathe, speak, and cough in ultra-clean, laminar flow theaters followed by use of CPAP and HFNO. Aerosol emission was measured via two methodologies, simultaneously. Hospitalized patients with COVID-19 had cough recorded via the same methodology on the infectious diseases ward.
CPAP (with exhalation port filter) was found to produce less aerosol than breathing, speaking, and coughing, even with large > 50 L/min face mask leaks. Coughing was associated with the highest aerosol emissions of any recorded activity.
HFNO was associated with aerosol emission from the machine. Generated particles were small (< 1 mcm), passing from the machine through the patient and to the detector without coalescence with respiratory aerosol, and, consequently, would be unlikely to carry viral particles.
More aerosol was generated in cough from patients with COVID-19 (n = 8) than from volunteers.
In the second study, 30 hospitalized patients with COVID-19 requiring supplemental oxygen were prospectively enrolled. In this observational environmental sampling study, participants received either supplemental oxygen, CPAP, or HFNO (n = 10 in each group). A nasopharyngeal swab, three air, and three surface samples were collected from each participant and the clinical environment.
Overall, 21 of the 30 participants tested positive for SARS-CoV-2 RNA in the nasopharynx. In contrast, 4 out of 90 air samples and 6 of 90 surface samples tested positive for viral RNA, although there were an additional 10 suspected-positive samples in both air and surfaces samples.
Neither the use of CPAP nor HFNO nor coughing were associated with significantly more environmental contamination than supplemental oxygen use. Of the total positive or suspected-positive samples by viral PCR detection, only one nasopharynx sample from an HFNO patient was biologically viable in cell culture assay.
“Our findings show that the noninvasive breathing support methods do not pose a higher risk of transmitting infection, which has significant implications for the management of the patients,” said coauthor Danny McAuley, MD.
“If there isn’t a higher risk of infection transmission, current practices may be overcautious measures for certain settings, for example preventing relatives visiting the sickest patients, whilst underestimating the risk in other settings, such as coughing patients with early infection on general wards.”
Although both studies are small, the results do suggest that there is a need for an evidence-based reassessment of infection prevention and control measures for noninvasive respiratory support treatments that are currently considered aerosol generating procedures.
A version of this article first appeared on Univadis.com.
Two studies published in Thorax have found that the use of continuous positive airways pressure (CPAP) or high-flow nasal oxygen (HFNO) to treat moderate to severe COVID-19 is not linked to a heightened risk of infection, as currently thought. Researchers say hospitals should use this information to re-examine aerosol procedures in regard to risk of transmission of SARS-CoV-2.
CPAP and HFNO have been thought to generate virus particles capable of contaminating the air and surfaces, necessitating additional infection control precautions such as segregating patients. However, this research demonstrates that both methods produced little measurable air or surface viral contamination. The amount of contamination was no more than with the use of supplemental oxygen and less than that produced when breathing, speaking, or coughing.
In one study, led by a team from the North Bristol NHS Trust, 25 healthy volunteers and eight hospitalized patients with COVID-19 were recruited and asked to breathe, speak, and cough in ultra-clean, laminar flow theaters followed by use of CPAP and HFNO. Aerosol emission was measured via two methodologies, simultaneously. Hospitalized patients with COVID-19 had cough recorded via the same methodology on the infectious diseases ward.
CPAP (with exhalation port filter) was found to produce less aerosol than breathing, speaking, and coughing, even with large > 50 L/min face mask leaks. Coughing was associated with the highest aerosol emissions of any recorded activity.
HFNO was associated with aerosol emission from the machine. Generated particles were small (< 1 mcm), passing from the machine through the patient and to the detector without coalescence with respiratory aerosol, and, consequently, would be unlikely to carry viral particles.
More aerosol was generated in cough from patients with COVID-19 (n = 8) than from volunteers.
In the second study, 30 hospitalized patients with COVID-19 requiring supplemental oxygen were prospectively enrolled. In this observational environmental sampling study, participants received either supplemental oxygen, CPAP, or HFNO (n = 10 in each group). A nasopharyngeal swab, three air, and three surface samples were collected from each participant and the clinical environment.
Overall, 21 of the 30 participants tested positive for SARS-CoV-2 RNA in the nasopharynx. In contrast, 4 out of 90 air samples and 6 of 90 surface samples tested positive for viral RNA, although there were an additional 10 suspected-positive samples in both air and surfaces samples.
Neither the use of CPAP nor HFNO nor coughing were associated with significantly more environmental contamination than supplemental oxygen use. Of the total positive or suspected-positive samples by viral PCR detection, only one nasopharynx sample from an HFNO patient was biologically viable in cell culture assay.
“Our findings show that the noninvasive breathing support methods do not pose a higher risk of transmitting infection, which has significant implications for the management of the patients,” said coauthor Danny McAuley, MD.
“If there isn’t a higher risk of infection transmission, current practices may be overcautious measures for certain settings, for example preventing relatives visiting the sickest patients, whilst underestimating the risk in other settings, such as coughing patients with early infection on general wards.”
Although both studies are small, the results do suggest that there is a need for an evidence-based reassessment of infection prevention and control measures for noninvasive respiratory support treatments that are currently considered aerosol generating procedures.
A version of this article first appeared on Univadis.com.
Two studies published in Thorax have found that the use of continuous positive airways pressure (CPAP) or high-flow nasal oxygen (HFNO) to treat moderate to severe COVID-19 is not linked to a heightened risk of infection, as currently thought. Researchers say hospitals should use this information to re-examine aerosol procedures in regard to risk of transmission of SARS-CoV-2.
CPAP and HFNO have been thought to generate virus particles capable of contaminating the air and surfaces, necessitating additional infection control precautions such as segregating patients. However, this research demonstrates that both methods produced little measurable air or surface viral contamination. The amount of contamination was no more than with the use of supplemental oxygen and less than that produced when breathing, speaking, or coughing.
In one study, led by a team from the North Bristol NHS Trust, 25 healthy volunteers and eight hospitalized patients with COVID-19 were recruited and asked to breathe, speak, and cough in ultra-clean, laminar flow theaters followed by use of CPAP and HFNO. Aerosol emission was measured via two methodologies, simultaneously. Hospitalized patients with COVID-19 had cough recorded via the same methodology on the infectious diseases ward.
CPAP (with exhalation port filter) was found to produce less aerosol than breathing, speaking, and coughing, even with large > 50 L/min face mask leaks. Coughing was associated with the highest aerosol emissions of any recorded activity.
HFNO was associated with aerosol emission from the machine. Generated particles were small (< 1 mcm), passing from the machine through the patient and to the detector without coalescence with respiratory aerosol, and, consequently, would be unlikely to carry viral particles.
More aerosol was generated in cough from patients with COVID-19 (n = 8) than from volunteers.
In the second study, 30 hospitalized patients with COVID-19 requiring supplemental oxygen were prospectively enrolled. In this observational environmental sampling study, participants received either supplemental oxygen, CPAP, or HFNO (n = 10 in each group). A nasopharyngeal swab, three air, and three surface samples were collected from each participant and the clinical environment.
Overall, 21 of the 30 participants tested positive for SARS-CoV-2 RNA in the nasopharynx. In contrast, 4 out of 90 air samples and 6 of 90 surface samples tested positive for viral RNA, although there were an additional 10 suspected-positive samples in both air and surfaces samples.
Neither the use of CPAP nor HFNO nor coughing were associated with significantly more environmental contamination than supplemental oxygen use. Of the total positive or suspected-positive samples by viral PCR detection, only one nasopharynx sample from an HFNO patient was biologically viable in cell culture assay.
“Our findings show that the noninvasive breathing support methods do not pose a higher risk of transmitting infection, which has significant implications for the management of the patients,” said coauthor Danny McAuley, MD.
“If there isn’t a higher risk of infection transmission, current practices may be overcautious measures for certain settings, for example preventing relatives visiting the sickest patients, whilst underestimating the risk in other settings, such as coughing patients with early infection on general wards.”
Although both studies are small, the results do suggest that there is a need for an evidence-based reassessment of infection prevention and control measures for noninvasive respiratory support treatments that are currently considered aerosol generating procedures.
A version of this article first appeared on Univadis.com.
FROM THORAX
Maraviroc, metformin fail to control NAFLD in people with HIV
The MAVMET study, the first randomized controlled trial of – and in some cases, prolonged use actually increased liver fat.
And that means clinicians like Yvonne Gilleece, MB BCh, who was not involved in the study but does run a liver clinic in England for people living with HIV, are returning to the one intervention proven to work. “As yet, the only thing that is proven to have a very positive effect that is published is weight loss,” said Dr. Gilleece, who runs the clinic at Brighton and Sussex University Hospital. “You don’t put someone on these particular drugs, particularly this combination, easily. MAVMET has really demonstrated that, actually, it’s not effective, and it’s not particularly beneficial to patients.”
The MAVMET trial data was presented at the 18th European AIDS Conference,
There was good reason to think maraviroc might work. A 2018 study in the journal Hepatology found that one of maraviroc’s molecular cousins, cenicriviroc, significantly reduced fibrosis in people with NAFLD. Dr. Gilleece is co-investigator of another study of maraviroc in NAFLD, the HEPMARC trial, which is wrapping up now. In addition to those studies, there are other potential treatments in ongoing trials, including semaglutide, which is being studied in the United States under the study name SLIM LIVER.
MAVMET enrolled 90 people living with HIV from six clinical sites in London who were 35 or older and who had at least one marker for NAFLD, such as abnormal liver lab results. But 70% qualified via imaging- and/or biopsy-confirmed NAFLD. Almost all participants (93%) were men and 81% were White. The trial excluded people who were pregnant or breastfeeding. The median age was 52, and the participants met the criteria for overweight but not obesity, with a median BMI of 28.
In other words, participants generally had fatty livers without the inflammation that characterizes the more aggressive nonalcoholic steatohepatitis (NASH). Clinicians can’t yet differentiate between those who will continue to have asymptomatic fatty liver and those who will progress to NASH and potentially need a liver transplant.
All people living with HIV in the trial had undetectable viral loads and were on HIV treatment. Nearly 1 in 5 (19%) were using a treatment regimen containing tenofovir alafenamide (TAF), which has been associated with weight gain. Nearly half were on integrase strand inhibitors.
Investigators divided the participants up into four groups: 24 people stayed on their HIV treatment and added nothing else; 23 people took maraviroc only; 21 took metformin only; and the final group took both maraviroc and metformin. Across groups, liver fat at baseline was 8.9%, and 78% had mild hepatic steatosis.
After taking the medications for 48 weeks, participants returned to clinic to be scanned via MRI proton density fat fraction (MRI-PDFF), which has been found to successfully measure liver fat. However, because of the COVID-19 pandemic, 20 of the 83 people who returned to the clinic came later than 48 weeks after the trial began.
When investigators looked at the results, they didn’t see what they hypothesized, said Sarah Pett, professor of infectious diseases at University College, London: The scatter plot graph of change in weight looked, well, scattershot: People who didn’t take any additional treatment sometimes lost more liver fat than those on treatment. In fact, the mean liver fat percentage rose by 2.2% in the maraviroc group, 1.3% in the metformin group, and 0.8% in the combination group. The control group saw an increase of 1.4% – meaning that there was no difference between the change in fat between those on treatment and those not.
What’s more, those who had delayed scans – and stayed on their treatment for a median of an additional 16 weeks – saw their liver fat increase even more.
In an interview, Dr. Pett called the results “disappointing.” “The numbers are quite small, but we still didn’t expect this,” she said. “It’s not explained by lockdown weight gain, although we still have to look in detail at how alcohol consumption could have contributed.”
There were also some limits to what the design of this particular trial could tell the researchers. For instance, nearly half of the participants in the maraviroc group, a third of the people in the metformin group, and 36% of those in the combination group had hepatic steatosis grades of 0, meaning that their livers were healthy. And MRI-PDFF becomes less reliable at that level.
“One of the regrets is that perhaps we should have done FibroScan [ultrasound], as well,” Dr. Pett said. The consequence is that the study may have undercounted the fat level by using MRI-PFDD.
“This suggests that the surrogate markers of NAFLD used in MAVMET were not very sensitive to those with a higher percentage of fat,” Dr. Pett said during her presentation. “We were really trying to be pragmatic and not require an MRI at screening.”
Whatever the case, she said that the failure of this particular treatment just highlights the growing need to look more seriously, and more collaboratively, at fat and liver health in people living with HIV.
“We need to really focus on setting up large cohorts of people living with HIV to look in a rigorous way at weight gain, changes in waist circumference, ectopic fat, capture fatty liver disease index scores, and cardiovascular risk, to acquire some longitudinal data,” she said. “And [we need to] join with our fellow researchers in overweight and obesity medicine and hepatology to make sure that people living with HIV are included in new treatments for NASH, as several large RCTs have excluded [people living with HIV].”
From Dr. Gilleece’s perspective, it also just speaks to how far the field has to go in identifying those with asymptomatic fatty livers from those who will progress to fibrosis and potentially need liver transplants.
“MAVMET shows the difficulty in managing NAFLD,” she said. “It seems quite an innocuous disease, because for the majority of people it’s not going to cause a problem in their lifetime. But the reality is, for some it will, and we don’t really know how to treat it.”
Dr. Gilleece has disclosed no relevant financial relationships. Dr. Pett reported receiving funding for trials from Gilead Sciences and Janssen-Cilag. ViiV Healthcare funded the MAVMET trial.
A version of this article first appeared on Medscape.com.
The MAVMET study, the first randomized controlled trial of – and in some cases, prolonged use actually increased liver fat.
And that means clinicians like Yvonne Gilleece, MB BCh, who was not involved in the study but does run a liver clinic in England for people living with HIV, are returning to the one intervention proven to work. “As yet, the only thing that is proven to have a very positive effect that is published is weight loss,” said Dr. Gilleece, who runs the clinic at Brighton and Sussex University Hospital. “You don’t put someone on these particular drugs, particularly this combination, easily. MAVMET has really demonstrated that, actually, it’s not effective, and it’s not particularly beneficial to patients.”
The MAVMET trial data was presented at the 18th European AIDS Conference,
There was good reason to think maraviroc might work. A 2018 study in the journal Hepatology found that one of maraviroc’s molecular cousins, cenicriviroc, significantly reduced fibrosis in people with NAFLD. Dr. Gilleece is co-investigator of another study of maraviroc in NAFLD, the HEPMARC trial, which is wrapping up now. In addition to those studies, there are other potential treatments in ongoing trials, including semaglutide, which is being studied in the United States under the study name SLIM LIVER.
MAVMET enrolled 90 people living with HIV from six clinical sites in London who were 35 or older and who had at least one marker for NAFLD, such as abnormal liver lab results. But 70% qualified via imaging- and/or biopsy-confirmed NAFLD. Almost all participants (93%) were men and 81% were White. The trial excluded people who were pregnant or breastfeeding. The median age was 52, and the participants met the criteria for overweight but not obesity, with a median BMI of 28.
In other words, participants generally had fatty livers without the inflammation that characterizes the more aggressive nonalcoholic steatohepatitis (NASH). Clinicians can’t yet differentiate between those who will continue to have asymptomatic fatty liver and those who will progress to NASH and potentially need a liver transplant.
All people living with HIV in the trial had undetectable viral loads and were on HIV treatment. Nearly 1 in 5 (19%) were using a treatment regimen containing tenofovir alafenamide (TAF), which has been associated with weight gain. Nearly half were on integrase strand inhibitors.
Investigators divided the participants up into four groups: 24 people stayed on their HIV treatment and added nothing else; 23 people took maraviroc only; 21 took metformin only; and the final group took both maraviroc and metformin. Across groups, liver fat at baseline was 8.9%, and 78% had mild hepatic steatosis.
After taking the medications for 48 weeks, participants returned to clinic to be scanned via MRI proton density fat fraction (MRI-PDFF), which has been found to successfully measure liver fat. However, because of the COVID-19 pandemic, 20 of the 83 people who returned to the clinic came later than 48 weeks after the trial began.
When investigators looked at the results, they didn’t see what they hypothesized, said Sarah Pett, professor of infectious diseases at University College, London: The scatter plot graph of change in weight looked, well, scattershot: People who didn’t take any additional treatment sometimes lost more liver fat than those on treatment. In fact, the mean liver fat percentage rose by 2.2% in the maraviroc group, 1.3% in the metformin group, and 0.8% in the combination group. The control group saw an increase of 1.4% – meaning that there was no difference between the change in fat between those on treatment and those not.
What’s more, those who had delayed scans – and stayed on their treatment for a median of an additional 16 weeks – saw their liver fat increase even more.
In an interview, Dr. Pett called the results “disappointing.” “The numbers are quite small, but we still didn’t expect this,” she said. “It’s not explained by lockdown weight gain, although we still have to look in detail at how alcohol consumption could have contributed.”
There were also some limits to what the design of this particular trial could tell the researchers. For instance, nearly half of the participants in the maraviroc group, a third of the people in the metformin group, and 36% of those in the combination group had hepatic steatosis grades of 0, meaning that their livers were healthy. And MRI-PDFF becomes less reliable at that level.
“One of the regrets is that perhaps we should have done FibroScan [ultrasound], as well,” Dr. Pett said. The consequence is that the study may have undercounted the fat level by using MRI-PFDD.
“This suggests that the surrogate markers of NAFLD used in MAVMET were not very sensitive to those with a higher percentage of fat,” Dr. Pett said during her presentation. “We were really trying to be pragmatic and not require an MRI at screening.”
Whatever the case, she said that the failure of this particular treatment just highlights the growing need to look more seriously, and more collaboratively, at fat and liver health in people living with HIV.
“We need to really focus on setting up large cohorts of people living with HIV to look in a rigorous way at weight gain, changes in waist circumference, ectopic fat, capture fatty liver disease index scores, and cardiovascular risk, to acquire some longitudinal data,” she said. “And [we need to] join with our fellow researchers in overweight and obesity medicine and hepatology to make sure that people living with HIV are included in new treatments for NASH, as several large RCTs have excluded [people living with HIV].”
From Dr. Gilleece’s perspective, it also just speaks to how far the field has to go in identifying those with asymptomatic fatty livers from those who will progress to fibrosis and potentially need liver transplants.
“MAVMET shows the difficulty in managing NAFLD,” she said. “It seems quite an innocuous disease, because for the majority of people it’s not going to cause a problem in their lifetime. But the reality is, for some it will, and we don’t really know how to treat it.”
Dr. Gilleece has disclosed no relevant financial relationships. Dr. Pett reported receiving funding for trials from Gilead Sciences and Janssen-Cilag. ViiV Healthcare funded the MAVMET trial.
A version of this article first appeared on Medscape.com.
The MAVMET study, the first randomized controlled trial of – and in some cases, prolonged use actually increased liver fat.
And that means clinicians like Yvonne Gilleece, MB BCh, who was not involved in the study but does run a liver clinic in England for people living with HIV, are returning to the one intervention proven to work. “As yet, the only thing that is proven to have a very positive effect that is published is weight loss,” said Dr. Gilleece, who runs the clinic at Brighton and Sussex University Hospital. “You don’t put someone on these particular drugs, particularly this combination, easily. MAVMET has really demonstrated that, actually, it’s not effective, and it’s not particularly beneficial to patients.”
The MAVMET trial data was presented at the 18th European AIDS Conference,
There was good reason to think maraviroc might work. A 2018 study in the journal Hepatology found that one of maraviroc’s molecular cousins, cenicriviroc, significantly reduced fibrosis in people with NAFLD. Dr. Gilleece is co-investigator of another study of maraviroc in NAFLD, the HEPMARC trial, which is wrapping up now. In addition to those studies, there are other potential treatments in ongoing trials, including semaglutide, which is being studied in the United States under the study name SLIM LIVER.
MAVMET enrolled 90 people living with HIV from six clinical sites in London who were 35 or older and who had at least one marker for NAFLD, such as abnormal liver lab results. But 70% qualified via imaging- and/or biopsy-confirmed NAFLD. Almost all participants (93%) were men and 81% were White. The trial excluded people who were pregnant or breastfeeding. The median age was 52, and the participants met the criteria for overweight but not obesity, with a median BMI of 28.
In other words, participants generally had fatty livers without the inflammation that characterizes the more aggressive nonalcoholic steatohepatitis (NASH). Clinicians can’t yet differentiate between those who will continue to have asymptomatic fatty liver and those who will progress to NASH and potentially need a liver transplant.
All people living with HIV in the trial had undetectable viral loads and were on HIV treatment. Nearly 1 in 5 (19%) were using a treatment regimen containing tenofovir alafenamide (TAF), which has been associated with weight gain. Nearly half were on integrase strand inhibitors.
Investigators divided the participants up into four groups: 24 people stayed on their HIV treatment and added nothing else; 23 people took maraviroc only; 21 took metformin only; and the final group took both maraviroc and metformin. Across groups, liver fat at baseline was 8.9%, and 78% had mild hepatic steatosis.
After taking the medications for 48 weeks, participants returned to clinic to be scanned via MRI proton density fat fraction (MRI-PDFF), which has been found to successfully measure liver fat. However, because of the COVID-19 pandemic, 20 of the 83 people who returned to the clinic came later than 48 weeks after the trial began.
When investigators looked at the results, they didn’t see what they hypothesized, said Sarah Pett, professor of infectious diseases at University College, London: The scatter plot graph of change in weight looked, well, scattershot: People who didn’t take any additional treatment sometimes lost more liver fat than those on treatment. In fact, the mean liver fat percentage rose by 2.2% in the maraviroc group, 1.3% in the metformin group, and 0.8% in the combination group. The control group saw an increase of 1.4% – meaning that there was no difference between the change in fat between those on treatment and those not.
What’s more, those who had delayed scans – and stayed on their treatment for a median of an additional 16 weeks – saw their liver fat increase even more.
In an interview, Dr. Pett called the results “disappointing.” “The numbers are quite small, but we still didn’t expect this,” she said. “It’s not explained by lockdown weight gain, although we still have to look in detail at how alcohol consumption could have contributed.”
There were also some limits to what the design of this particular trial could tell the researchers. For instance, nearly half of the participants in the maraviroc group, a third of the people in the metformin group, and 36% of those in the combination group had hepatic steatosis grades of 0, meaning that their livers were healthy. And MRI-PDFF becomes less reliable at that level.
“One of the regrets is that perhaps we should have done FibroScan [ultrasound], as well,” Dr. Pett said. The consequence is that the study may have undercounted the fat level by using MRI-PFDD.
“This suggests that the surrogate markers of NAFLD used in MAVMET were not very sensitive to those with a higher percentage of fat,” Dr. Pett said during her presentation. “We were really trying to be pragmatic and not require an MRI at screening.”
Whatever the case, she said that the failure of this particular treatment just highlights the growing need to look more seriously, and more collaboratively, at fat and liver health in people living with HIV.
“We need to really focus on setting up large cohorts of people living with HIV to look in a rigorous way at weight gain, changes in waist circumference, ectopic fat, capture fatty liver disease index scores, and cardiovascular risk, to acquire some longitudinal data,” she said. “And [we need to] join with our fellow researchers in overweight and obesity medicine and hepatology to make sure that people living with HIV are included in new treatments for NASH, as several large RCTs have excluded [people living with HIV].”
From Dr. Gilleece’s perspective, it also just speaks to how far the field has to go in identifying those with asymptomatic fatty livers from those who will progress to fibrosis and potentially need liver transplants.
“MAVMET shows the difficulty in managing NAFLD,” she said. “It seems quite an innocuous disease, because for the majority of people it’s not going to cause a problem in their lifetime. But the reality is, for some it will, and we don’t really know how to treat it.”
Dr. Gilleece has disclosed no relevant financial relationships. Dr. Pett reported receiving funding for trials from Gilead Sciences and Janssen-Cilag. ViiV Healthcare funded the MAVMET trial.
A version of this article first appeared on Medscape.com.
ACIP recommends universal HBV vaccination for adults under 60, expands recommendations for vaccines against orthopoxviruses and Ebola
The group also voted to expand recommendations for vaccinating people at risk for occupational exposure to Ebola and to recommend Jynneos, a smallpox and monkeypox vaccine, for at-risk populations.
The recommendations were approved Nov. 3.
Previously, ACIP recommended HBV vaccination for unvaccinated adults at increased risk for infection because of sexual exposure, percutaneous or mucosal exposure to blood, hepatitis C infection, chronic liver disease, end-stage renal disease, HIV infection, and travel to areas with high to intermediate levels of HBV infection. But experts agreed a new strategy was needed, as previously falling rates of HBV have plateaued. “The past decade has illustrated that risk-based screening has got us as far as it can take us,” Mark Weng, MD, a lieutenant commander in the U.S. Public Health Service and lead of the ACIP Hepatitis Vaccine Working Group, said during the meeting.
There are 1.9 million people living with chronic HBV in the United States, with over 20,000 new acute infections every year. Rates are highest among those in their 40s and 50s, Dr. Weng noted.
The group debated whether to apply the universal recommendation to all ages, but in a close vote (eight yes, seven no), ACIP included an age cutoff of 59. The majority argued that adults 60 and older are at lower risk for infection and vaccination efforts targeting younger adults would be more effective. Those 60 and older would continue to follow the risk-based guidelines, but anyone, regardless of age, can receive the vaccine if they wish to be protected, the group added.
The CDC director as well as several professional societies need to approve the recommendation before it becomes public policy.
ACIP also voted to recommend the following:
- Adding updated recommendations to the 2022 immunization schedules for children, adolescents, and adults, including dengue vaccination for children aged 9-16 years in endemic areas and in adults over 65 and those aged 19-64 with certain chronic conditions.
- The use of Jynneos, a smallpox and monkeypox vaccine, as an alternative to ACAM2000 for those at risk for occupational exposure.
- Pre-exposure vaccination of health care personnel involved in the transport and treatment of suspected Ebola patients at special treatment centers, or lab and support staff working with or handling specimens that may contain the Ebola virus.
A version of this article first appeared on Medscape.com.
The group also voted to expand recommendations for vaccinating people at risk for occupational exposure to Ebola and to recommend Jynneos, a smallpox and monkeypox vaccine, for at-risk populations.
The recommendations were approved Nov. 3.
Previously, ACIP recommended HBV vaccination for unvaccinated adults at increased risk for infection because of sexual exposure, percutaneous or mucosal exposure to blood, hepatitis C infection, chronic liver disease, end-stage renal disease, HIV infection, and travel to areas with high to intermediate levels of HBV infection. But experts agreed a new strategy was needed, as previously falling rates of HBV have plateaued. “The past decade has illustrated that risk-based screening has got us as far as it can take us,” Mark Weng, MD, a lieutenant commander in the U.S. Public Health Service and lead of the ACIP Hepatitis Vaccine Working Group, said during the meeting.
There are 1.9 million people living with chronic HBV in the United States, with over 20,000 new acute infections every year. Rates are highest among those in their 40s and 50s, Dr. Weng noted.
The group debated whether to apply the universal recommendation to all ages, but in a close vote (eight yes, seven no), ACIP included an age cutoff of 59. The majority argued that adults 60 and older are at lower risk for infection and vaccination efforts targeting younger adults would be more effective. Those 60 and older would continue to follow the risk-based guidelines, but anyone, regardless of age, can receive the vaccine if they wish to be protected, the group added.
The CDC director as well as several professional societies need to approve the recommendation before it becomes public policy.
ACIP also voted to recommend the following:
- Adding updated recommendations to the 2022 immunization schedules for children, adolescents, and adults, including dengue vaccination for children aged 9-16 years in endemic areas and in adults over 65 and those aged 19-64 with certain chronic conditions.
- The use of Jynneos, a smallpox and monkeypox vaccine, as an alternative to ACAM2000 for those at risk for occupational exposure.
- Pre-exposure vaccination of health care personnel involved in the transport and treatment of suspected Ebola patients at special treatment centers, or lab and support staff working with or handling specimens that may contain the Ebola virus.
A version of this article first appeared on Medscape.com.
The group also voted to expand recommendations for vaccinating people at risk for occupational exposure to Ebola and to recommend Jynneos, a smallpox and monkeypox vaccine, for at-risk populations.
The recommendations were approved Nov. 3.
Previously, ACIP recommended HBV vaccination for unvaccinated adults at increased risk for infection because of sexual exposure, percutaneous or mucosal exposure to blood, hepatitis C infection, chronic liver disease, end-stage renal disease, HIV infection, and travel to areas with high to intermediate levels of HBV infection. But experts agreed a new strategy was needed, as previously falling rates of HBV have plateaued. “The past decade has illustrated that risk-based screening has got us as far as it can take us,” Mark Weng, MD, a lieutenant commander in the U.S. Public Health Service and lead of the ACIP Hepatitis Vaccine Working Group, said during the meeting.
There are 1.9 million people living with chronic HBV in the United States, with over 20,000 new acute infections every year. Rates are highest among those in their 40s and 50s, Dr. Weng noted.
The group debated whether to apply the universal recommendation to all ages, but in a close vote (eight yes, seven no), ACIP included an age cutoff of 59. The majority argued that adults 60 and older are at lower risk for infection and vaccination efforts targeting younger adults would be more effective. Those 60 and older would continue to follow the risk-based guidelines, but anyone, regardless of age, can receive the vaccine if they wish to be protected, the group added.
The CDC director as well as several professional societies need to approve the recommendation before it becomes public policy.
ACIP also voted to recommend the following:
- Adding updated recommendations to the 2022 immunization schedules for children, adolescents, and adults, including dengue vaccination for children aged 9-16 years in endemic areas and in adults over 65 and those aged 19-64 with certain chronic conditions.
- The use of Jynneos, a smallpox and monkeypox vaccine, as an alternative to ACAM2000 for those at risk for occupational exposure.
- Pre-exposure vaccination of health care personnel involved in the transport and treatment of suspected Ebola patients at special treatment centers, or lab and support staff working with or handling specimens that may contain the Ebola virus.
A version of this article first appeared on Medscape.com.