User login
Quick tips: How to get your study published
SAN DIEGO – Looking to get your study published in a top medical journal? Bob Löwenberg, MD, PhD, editor-in-chief of Blood, says to start thinking about what appeals to readers.
“What do readers want? They want important information with impact in a clinical or biological sense,” Dr. Löwenberg of Erasmus University Rotterdam (the Netherlands) said at the annual meeting of the American Society of Hematology. “Usually they want to get novel information – new and cutting-edge insights, if possible. And readers want to receive access to information that is right. This is about quality.”
Dr. Löwenberg offered several tips for getting published:
- Make sure your paper has a “clear message” that comes across in both its title and a concisely written abstract. “When your colleagues are going to scan the journal, they should say ‘Hey, this is an interesting title’ or ‘This is an interesting abstract,’ ” Dr. Löwenberg said.
- Avoid jargon and slang. And don’t fill your paper with abbreviations because that will make it unreadable.
- Don’t just cut and paste the abstract from your meeting submission. Update the information and rewrite it before submitting it. “The abstract is so important because it is the part of your manuscript that’s copied by reference systems,” Dr. Löwenberg said. “It’s more broadly published than your manuscript. Write it in such a way that it tells your entire story in a minimal number of words, without changing the overall message of your paper, and in clear language.”
- Focus on providing important background in the introduction, which usually summarizes existing research.
- “Distill the essentials” in the discussion section. “Don’t repeat the results. Discuss the importance of your findings in relation to the state-of-the-art information that you have presented in the introduction,” he said.
- Beware of plagiarism, which includes “self-plagiarism” – duplicating your own previous research without acknowledgment.
- Understand new rules regarding data-sharing requirements developed by the International Committee of Medical Journal Editors. In order to be considered for publication by the committee’s member journals, clinical trials that begin enrolling participants as of Jan. 1, 2019, must include a data-sharing plan in the trial’s registration.
- Don’t be surprised if your paper is turned down. “We all have experience with rejected papers,” he said. “This is part of the game.”
If you are rejected, you may wish to send a rebuttal – a form of appeal – to the journal. Consider this option if the journal “clearly misunderstood or misrepresented the paper,” he said. “Be polite, try to be unemotional and clear, and never [write] it the same day as when you are still angry about this decision.” Once you send a rebuttal, wait for at least a week for a response. If one doesn’t come, he said, feel free to submit the paper elsewhere.
Dr. Löwenberg reported having no relevant financial disclosures.
SAN DIEGO – Looking to get your study published in a top medical journal? Bob Löwenberg, MD, PhD, editor-in-chief of Blood, says to start thinking about what appeals to readers.
“What do readers want? They want important information with impact in a clinical or biological sense,” Dr. Löwenberg of Erasmus University Rotterdam (the Netherlands) said at the annual meeting of the American Society of Hematology. “Usually they want to get novel information – new and cutting-edge insights, if possible. And readers want to receive access to information that is right. This is about quality.”
Dr. Löwenberg offered several tips for getting published:
- Make sure your paper has a “clear message” that comes across in both its title and a concisely written abstract. “When your colleagues are going to scan the journal, they should say ‘Hey, this is an interesting title’ or ‘This is an interesting abstract,’ ” Dr. Löwenberg said.
- Avoid jargon and slang. And don’t fill your paper with abbreviations because that will make it unreadable.
- Don’t just cut and paste the abstract from your meeting submission. Update the information and rewrite it before submitting it. “The abstract is so important because it is the part of your manuscript that’s copied by reference systems,” Dr. Löwenberg said. “It’s more broadly published than your manuscript. Write it in such a way that it tells your entire story in a minimal number of words, without changing the overall message of your paper, and in clear language.”
- Focus on providing important background in the introduction, which usually summarizes existing research.
- “Distill the essentials” in the discussion section. “Don’t repeat the results. Discuss the importance of your findings in relation to the state-of-the-art information that you have presented in the introduction,” he said.
- Beware of plagiarism, which includes “self-plagiarism” – duplicating your own previous research without acknowledgment.
- Understand new rules regarding data-sharing requirements developed by the International Committee of Medical Journal Editors. In order to be considered for publication by the committee’s member journals, clinical trials that begin enrolling participants as of Jan. 1, 2019, must include a data-sharing plan in the trial’s registration.
- Don’t be surprised if your paper is turned down. “We all have experience with rejected papers,” he said. “This is part of the game.”
If you are rejected, you may wish to send a rebuttal – a form of appeal – to the journal. Consider this option if the journal “clearly misunderstood or misrepresented the paper,” he said. “Be polite, try to be unemotional and clear, and never [write] it the same day as when you are still angry about this decision.” Once you send a rebuttal, wait for at least a week for a response. If one doesn’t come, he said, feel free to submit the paper elsewhere.
Dr. Löwenberg reported having no relevant financial disclosures.
SAN DIEGO – Looking to get your study published in a top medical journal? Bob Löwenberg, MD, PhD, editor-in-chief of Blood, says to start thinking about what appeals to readers.
“What do readers want? They want important information with impact in a clinical or biological sense,” Dr. Löwenberg of Erasmus University Rotterdam (the Netherlands) said at the annual meeting of the American Society of Hematology. “Usually they want to get novel information – new and cutting-edge insights, if possible. And readers want to receive access to information that is right. This is about quality.”
Dr. Löwenberg offered several tips for getting published:
- Make sure your paper has a “clear message” that comes across in both its title and a concisely written abstract. “When your colleagues are going to scan the journal, they should say ‘Hey, this is an interesting title’ or ‘This is an interesting abstract,’ ” Dr. Löwenberg said.
- Avoid jargon and slang. And don’t fill your paper with abbreviations because that will make it unreadable.
- Don’t just cut and paste the abstract from your meeting submission. Update the information and rewrite it before submitting it. “The abstract is so important because it is the part of your manuscript that’s copied by reference systems,” Dr. Löwenberg said. “It’s more broadly published than your manuscript. Write it in such a way that it tells your entire story in a minimal number of words, without changing the overall message of your paper, and in clear language.”
- Focus on providing important background in the introduction, which usually summarizes existing research.
- “Distill the essentials” in the discussion section. “Don’t repeat the results. Discuss the importance of your findings in relation to the state-of-the-art information that you have presented in the introduction,” he said.
- Beware of plagiarism, which includes “self-plagiarism” – duplicating your own previous research without acknowledgment.
- Understand new rules regarding data-sharing requirements developed by the International Committee of Medical Journal Editors. In order to be considered for publication by the committee’s member journals, clinical trials that begin enrolling participants as of Jan. 1, 2019, must include a data-sharing plan in the trial’s registration.
- Don’t be surprised if your paper is turned down. “We all have experience with rejected papers,” he said. “This is part of the game.”
If you are rejected, you may wish to send a rebuttal – a form of appeal – to the journal. Consider this option if the journal “clearly misunderstood or misrepresented the paper,” he said. “Be polite, try to be unemotional and clear, and never [write] it the same day as when you are still angry about this decision.” Once you send a rebuttal, wait for at least a week for a response. If one doesn’t come, he said, feel free to submit the paper elsewhere.
Dr. Löwenberg reported having no relevant financial disclosures.
EXPERT ANALYSIS FROM ASH 2018
FDA approves new ALL treatment for children, young adults
The in pediatric and young adult patients aged 1 month to 21 years.
Calaspargase pegol-mknl is an asparagine-specific enzyme intended to provide a longer interval between doses, compared with other available pegaspargase products. The recommended dosage of calaspargase pegol-mknl is 2,500 units/m2 given no more frequently than every 21 days.
The FDA said it approved calaspargase pegol-mknl because the drug maintained nadir serum asparaginase activity above the level of 0.1 U/mL when given at 2,500 U/m2 every 3 weeks.
Calaspargase pegol-mknl was evaluated in Study DFCI 11-001, a trial of 237 children and adolescents with newly diagnosed ALL or lymphoblastic lymphoma. The patients’ median age was 5 years.
Study participants received calaspargase pegol-mknl at 2,500 U/m2 (n = 118) or pegaspargase at 2,500 U/m2 (n = 119) as part of a Dana-Farber Cancer Institute ALL Consortium backbone therapy. The median duration of exposure was 8 months for both calaspargase pegol-mknl and pegaspargase. Among the patients with B-cell lineage ALL, the complete remission rate was 98% in the calaspargase pegol-mknl arm and 99% in the pegaspargase arm. Estimated overall survival rates were comparable between the arms.
Common grade 3 or higher adverse events in the calaspargase pegol-mknl and pegaspargase arms included elevated transaminase (52% and 66%, respectively), bilirubin increase (20% and 25%), pancreatitis (18% and 24%), and abnormal clotting studies (14% and 21%). There was one fatal adverse event among patients on calaspargase pegol-mknl – multiorgan failure in the setting of chronic pancreatitis associated with a pancreatic pseudocyst.
The safety of calaspargase pegol-mknl was also evaluated in Study AALL07P4, a trial of patients with newly diagnosed, high-risk B-precursor ALL. The patients received calaspargase pegol-mknl at 2,500 U/m2 (n = 43) or 2,100 U/m2 (n = 68) or pegaspargase at 2,500 U/m2 (n = 52) as a component of an augmented Berlin-Frankfurt-Münster regimen. The patients’ median age was 11 years. The median duration of exposure was 7 months for both calaspargase pegol-mknl and pegaspargase. There were 3 induction deaths among the 111 patients who received calaspargase pegol-mknl (2.8%) but no induction deaths among the 52 patients treated with pegaspargase.
Additional details on these studies and calaspargase pegol-mknl can be found in the drug’s prescribing information. Calaspargase pegol-mknl is a product of Servier.
The in pediatric and young adult patients aged 1 month to 21 years.
Calaspargase pegol-mknl is an asparagine-specific enzyme intended to provide a longer interval between doses, compared with other available pegaspargase products. The recommended dosage of calaspargase pegol-mknl is 2,500 units/m2 given no more frequently than every 21 days.
The FDA said it approved calaspargase pegol-mknl because the drug maintained nadir serum asparaginase activity above the level of 0.1 U/mL when given at 2,500 U/m2 every 3 weeks.
Calaspargase pegol-mknl was evaluated in Study DFCI 11-001, a trial of 237 children and adolescents with newly diagnosed ALL or lymphoblastic lymphoma. The patients’ median age was 5 years.
Study participants received calaspargase pegol-mknl at 2,500 U/m2 (n = 118) or pegaspargase at 2,500 U/m2 (n = 119) as part of a Dana-Farber Cancer Institute ALL Consortium backbone therapy. The median duration of exposure was 8 months for both calaspargase pegol-mknl and pegaspargase. Among the patients with B-cell lineage ALL, the complete remission rate was 98% in the calaspargase pegol-mknl arm and 99% in the pegaspargase arm. Estimated overall survival rates were comparable between the arms.
Common grade 3 or higher adverse events in the calaspargase pegol-mknl and pegaspargase arms included elevated transaminase (52% and 66%, respectively), bilirubin increase (20% and 25%), pancreatitis (18% and 24%), and abnormal clotting studies (14% and 21%). There was one fatal adverse event among patients on calaspargase pegol-mknl – multiorgan failure in the setting of chronic pancreatitis associated with a pancreatic pseudocyst.
The safety of calaspargase pegol-mknl was also evaluated in Study AALL07P4, a trial of patients with newly diagnosed, high-risk B-precursor ALL. The patients received calaspargase pegol-mknl at 2,500 U/m2 (n = 43) or 2,100 U/m2 (n = 68) or pegaspargase at 2,500 U/m2 (n = 52) as a component of an augmented Berlin-Frankfurt-Münster regimen. The patients’ median age was 11 years. The median duration of exposure was 7 months for both calaspargase pegol-mknl and pegaspargase. There were 3 induction deaths among the 111 patients who received calaspargase pegol-mknl (2.8%) but no induction deaths among the 52 patients treated with pegaspargase.
Additional details on these studies and calaspargase pegol-mknl can be found in the drug’s prescribing information. Calaspargase pegol-mknl is a product of Servier.
The in pediatric and young adult patients aged 1 month to 21 years.
Calaspargase pegol-mknl is an asparagine-specific enzyme intended to provide a longer interval between doses, compared with other available pegaspargase products. The recommended dosage of calaspargase pegol-mknl is 2,500 units/m2 given no more frequently than every 21 days.
The FDA said it approved calaspargase pegol-mknl because the drug maintained nadir serum asparaginase activity above the level of 0.1 U/mL when given at 2,500 U/m2 every 3 weeks.
Calaspargase pegol-mknl was evaluated in Study DFCI 11-001, a trial of 237 children and adolescents with newly diagnosed ALL or lymphoblastic lymphoma. The patients’ median age was 5 years.
Study participants received calaspargase pegol-mknl at 2,500 U/m2 (n = 118) or pegaspargase at 2,500 U/m2 (n = 119) as part of a Dana-Farber Cancer Institute ALL Consortium backbone therapy. The median duration of exposure was 8 months for both calaspargase pegol-mknl and pegaspargase. Among the patients with B-cell lineage ALL, the complete remission rate was 98% in the calaspargase pegol-mknl arm and 99% in the pegaspargase arm. Estimated overall survival rates were comparable between the arms.
Common grade 3 or higher adverse events in the calaspargase pegol-mknl and pegaspargase arms included elevated transaminase (52% and 66%, respectively), bilirubin increase (20% and 25%), pancreatitis (18% and 24%), and abnormal clotting studies (14% and 21%). There was one fatal adverse event among patients on calaspargase pegol-mknl – multiorgan failure in the setting of chronic pancreatitis associated with a pancreatic pseudocyst.
The safety of calaspargase pegol-mknl was also evaluated in Study AALL07P4, a trial of patients with newly diagnosed, high-risk B-precursor ALL. The patients received calaspargase pegol-mknl at 2,500 U/m2 (n = 43) or 2,100 U/m2 (n = 68) or pegaspargase at 2,500 U/m2 (n = 52) as a component of an augmented Berlin-Frankfurt-Münster regimen. The patients’ median age was 11 years. The median duration of exposure was 7 months for both calaspargase pegol-mknl and pegaspargase. There were 3 induction deaths among the 111 patients who received calaspargase pegol-mknl (2.8%) but no induction deaths among the 52 patients treated with pegaspargase.
Additional details on these studies and calaspargase pegol-mknl can be found in the drug’s prescribing information. Calaspargase pegol-mknl is a product of Servier.
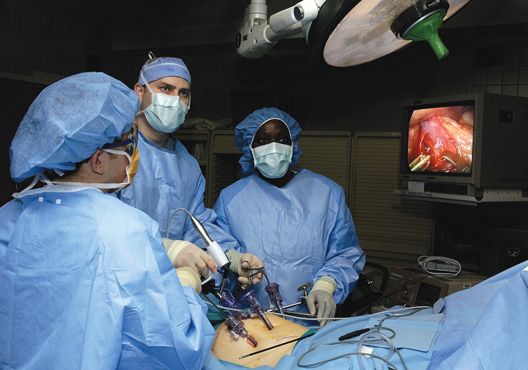
Don’t leave vaginal hysterectomies behind, surgeon urges
LAS VEGAS –
While “younger trainees are seeing fewer vaginal procedures being done and have less confidence to do the procedure,” research suggests that the vaginal approach can offer major benefits, compared with the alternatives, Rosanne M. Kho, MD, of the Cleveland Clinic, said at the Pelvic Anatomy and Gynecologic Surgery Symposium.
Dr. Kho pointed to several studies suggesting a decline in vaginal hysterectomies as laparoscopic and robot procedures become more common. One study compared hysterectomy surgery approaches during 2007-2010 and found a sharp rise in robotic procedures (0.5% to 10%) and a big decrease in abdominal procedures (from 54% to 40%). The rate of laparoscopic procedures grew (from 24% to 30%), while vaginal procedures dipped slightly (22% to 20%) (JAMA. 2013 Feb 20;309[7]:689-98). Another study tracked hysterectomy strategies at Pittsburgh’s Magee-Womens Hospital in almost 14,000 women during 2000-2010. It found that vaginal hysterectomy rates fell from 22% to 17% while laparoscopic rates grew remarkably from 3% to 43%. Open procedures fell dramatically from 75% to 36% (Am J Obstet Gynecol. 2013 Apr. doi: 10.1016/j.ajog.2013.01.022).
These findings are “telling me that surgeons are steering away from the vaginal approach because the laparoscopic and robotic approach are much more appealing,” Dr. Koh said at the meeting, which was jointly provided by Global Academy for Medical Education and the University of Cincinnati. Global Academy and this news organization are owned by the same company.
Specifically, it appears that surgeons think the vaginal hysterectomy is more “challenging” and “cumbersome,” Dr. Kho said, and they lack inadequate training.
Why should vaginal hysterectomy still be considered? Dr. Kho pointed to two pieces of evidence:
- Expert opinion. A 2017 committee opinion from the American College of Obstetricians and Gynecologists examined routes of hysterectomy in benign disease and declared that, despite the decrease in its use, “evidence supports the opinion that [when feasible] vaginal hysterectomy is associated with better outcomes” than are laparoscopic or abdominal hysterectomy. Also, the decision to perform a salpingo-oophorectomy is not necessarily a contraindication to performing a vaginal hysterectomy, according to the committee opinion (Obstet Gynecol. 2017 Jun;129[6]:e155-e9).The opinion also says, “the vaginal approach is preferred among the minimally invasive approaches. Laparoscopic hysterectomy is a preferable alternative to open abdominal hysterectomy for those patients in whom a vaginal hysterectomy is not indicated or feasible. Although minimally invasive approaches to hysterectomy are the preferred route, open abdominal hysterectomy remains an important surgical option for some patients.”
- Randomized, controlled studies. A 2015 Cochrane Library systematic review examined 47 randomized, controlled trials and found that “vaginal hysterectomy should be performed whenever possible. Where vaginal hysterectomy is not possible, both a laparoscopic approach and abdominal hysterectomy have their pros and cons, and these should be incorporated in the decision-making process” (Cochrane Database Syst Rev. 2015 Aug 12. doi: 10.1002/14651858.CD003677.pub5).
What if a patient has an enlarged uterus? Dr. Kho coauthored a 2017 review that suggested that vaginal hysterectomy may be appropriate in this case. Her report found that in women with large uteri, “vaginal hysterectomy is preferred over laparoscopic and laparoscopic assistance with less operative time and hospital cost. In morbidly obese patients with large uteri, total laparoscopic hysterectomy is superior to vaginal hysterectomy with lesser odds of blood transfusion and lower length of hospital stay” (Clin Obstet Gynecol. 2017 Jun;60[2]:286-95).
What about the removal of fallopian tubes – salpingectomy – during vaginal hysterectomy? Dr. Kho highlighted a 2017 decision analysis that said these procedures are frequently performed for cancer prevention during laparoscopic and open hysterectomies “but [fallopian tubes] are not routinely removed during vaginal hysterectomy because of perceptions of increased morbidity, difficulty, or inadequate surgical training.”
The analysis, however, determined that “salpingectomy should routinely be performed with vaginal hysterectomy because it was the dominant and therefore cost-effective strategy. Complications are minimally increased, but the trade-off with cancer prevention is highly favorable.” (Am J Obstet Gynecol. 2017 Nov;217[5]:603.e1-603.e6).
Dr. Kho reported consulting for AbbVie, Olympus, and Applied Medical.
LAS VEGAS –
While “younger trainees are seeing fewer vaginal procedures being done and have less confidence to do the procedure,” research suggests that the vaginal approach can offer major benefits, compared with the alternatives, Rosanne M. Kho, MD, of the Cleveland Clinic, said at the Pelvic Anatomy and Gynecologic Surgery Symposium.
Dr. Kho pointed to several studies suggesting a decline in vaginal hysterectomies as laparoscopic and robot procedures become more common. One study compared hysterectomy surgery approaches during 2007-2010 and found a sharp rise in robotic procedures (0.5% to 10%) and a big decrease in abdominal procedures (from 54% to 40%). The rate of laparoscopic procedures grew (from 24% to 30%), while vaginal procedures dipped slightly (22% to 20%) (JAMA. 2013 Feb 20;309[7]:689-98). Another study tracked hysterectomy strategies at Pittsburgh’s Magee-Womens Hospital in almost 14,000 women during 2000-2010. It found that vaginal hysterectomy rates fell from 22% to 17% while laparoscopic rates grew remarkably from 3% to 43%. Open procedures fell dramatically from 75% to 36% (Am J Obstet Gynecol. 2013 Apr. doi: 10.1016/j.ajog.2013.01.022).
These findings are “telling me that surgeons are steering away from the vaginal approach because the laparoscopic and robotic approach are much more appealing,” Dr. Koh said at the meeting, which was jointly provided by Global Academy for Medical Education and the University of Cincinnati. Global Academy and this news organization are owned by the same company.
Specifically, it appears that surgeons think the vaginal hysterectomy is more “challenging” and “cumbersome,” Dr. Kho said, and they lack inadequate training.
Why should vaginal hysterectomy still be considered? Dr. Kho pointed to two pieces of evidence:
- Expert opinion. A 2017 committee opinion from the American College of Obstetricians and Gynecologists examined routes of hysterectomy in benign disease and declared that, despite the decrease in its use, “evidence supports the opinion that [when feasible] vaginal hysterectomy is associated with better outcomes” than are laparoscopic or abdominal hysterectomy. Also, the decision to perform a salpingo-oophorectomy is not necessarily a contraindication to performing a vaginal hysterectomy, according to the committee opinion (Obstet Gynecol. 2017 Jun;129[6]:e155-e9).The opinion also says, “the vaginal approach is preferred among the minimally invasive approaches. Laparoscopic hysterectomy is a preferable alternative to open abdominal hysterectomy for those patients in whom a vaginal hysterectomy is not indicated or feasible. Although minimally invasive approaches to hysterectomy are the preferred route, open abdominal hysterectomy remains an important surgical option for some patients.”
- Randomized, controlled studies. A 2015 Cochrane Library systematic review examined 47 randomized, controlled trials and found that “vaginal hysterectomy should be performed whenever possible. Where vaginal hysterectomy is not possible, both a laparoscopic approach and abdominal hysterectomy have their pros and cons, and these should be incorporated in the decision-making process” (Cochrane Database Syst Rev. 2015 Aug 12. doi: 10.1002/14651858.CD003677.pub5).
What if a patient has an enlarged uterus? Dr. Kho coauthored a 2017 review that suggested that vaginal hysterectomy may be appropriate in this case. Her report found that in women with large uteri, “vaginal hysterectomy is preferred over laparoscopic and laparoscopic assistance with less operative time and hospital cost. In morbidly obese patients with large uteri, total laparoscopic hysterectomy is superior to vaginal hysterectomy with lesser odds of blood transfusion and lower length of hospital stay” (Clin Obstet Gynecol. 2017 Jun;60[2]:286-95).
What about the removal of fallopian tubes – salpingectomy – during vaginal hysterectomy? Dr. Kho highlighted a 2017 decision analysis that said these procedures are frequently performed for cancer prevention during laparoscopic and open hysterectomies “but [fallopian tubes] are not routinely removed during vaginal hysterectomy because of perceptions of increased morbidity, difficulty, or inadequate surgical training.”
The analysis, however, determined that “salpingectomy should routinely be performed with vaginal hysterectomy because it was the dominant and therefore cost-effective strategy. Complications are minimally increased, but the trade-off with cancer prevention is highly favorable.” (Am J Obstet Gynecol. 2017 Nov;217[5]:603.e1-603.e6).
Dr. Kho reported consulting for AbbVie, Olympus, and Applied Medical.
LAS VEGAS –
While “younger trainees are seeing fewer vaginal procedures being done and have less confidence to do the procedure,” research suggests that the vaginal approach can offer major benefits, compared with the alternatives, Rosanne M. Kho, MD, of the Cleveland Clinic, said at the Pelvic Anatomy and Gynecologic Surgery Symposium.
Dr. Kho pointed to several studies suggesting a decline in vaginal hysterectomies as laparoscopic and robot procedures become more common. One study compared hysterectomy surgery approaches during 2007-2010 and found a sharp rise in robotic procedures (0.5% to 10%) and a big decrease in abdominal procedures (from 54% to 40%). The rate of laparoscopic procedures grew (from 24% to 30%), while vaginal procedures dipped slightly (22% to 20%) (JAMA. 2013 Feb 20;309[7]:689-98). Another study tracked hysterectomy strategies at Pittsburgh’s Magee-Womens Hospital in almost 14,000 women during 2000-2010. It found that vaginal hysterectomy rates fell from 22% to 17% while laparoscopic rates grew remarkably from 3% to 43%. Open procedures fell dramatically from 75% to 36% (Am J Obstet Gynecol. 2013 Apr. doi: 10.1016/j.ajog.2013.01.022).
These findings are “telling me that surgeons are steering away from the vaginal approach because the laparoscopic and robotic approach are much more appealing,” Dr. Koh said at the meeting, which was jointly provided by Global Academy for Medical Education and the University of Cincinnati. Global Academy and this news organization are owned by the same company.
Specifically, it appears that surgeons think the vaginal hysterectomy is more “challenging” and “cumbersome,” Dr. Kho said, and they lack inadequate training.
Why should vaginal hysterectomy still be considered? Dr. Kho pointed to two pieces of evidence:
- Expert opinion. A 2017 committee opinion from the American College of Obstetricians and Gynecologists examined routes of hysterectomy in benign disease and declared that, despite the decrease in its use, “evidence supports the opinion that [when feasible] vaginal hysterectomy is associated with better outcomes” than are laparoscopic or abdominal hysterectomy. Also, the decision to perform a salpingo-oophorectomy is not necessarily a contraindication to performing a vaginal hysterectomy, according to the committee opinion (Obstet Gynecol. 2017 Jun;129[6]:e155-e9).The opinion also says, “the vaginal approach is preferred among the minimally invasive approaches. Laparoscopic hysterectomy is a preferable alternative to open abdominal hysterectomy for those patients in whom a vaginal hysterectomy is not indicated or feasible. Although minimally invasive approaches to hysterectomy are the preferred route, open abdominal hysterectomy remains an important surgical option for some patients.”
- Randomized, controlled studies. A 2015 Cochrane Library systematic review examined 47 randomized, controlled trials and found that “vaginal hysterectomy should be performed whenever possible. Where vaginal hysterectomy is not possible, both a laparoscopic approach and abdominal hysterectomy have their pros and cons, and these should be incorporated in the decision-making process” (Cochrane Database Syst Rev. 2015 Aug 12. doi: 10.1002/14651858.CD003677.pub5).
What if a patient has an enlarged uterus? Dr. Kho coauthored a 2017 review that suggested that vaginal hysterectomy may be appropriate in this case. Her report found that in women with large uteri, “vaginal hysterectomy is preferred over laparoscopic and laparoscopic assistance with less operative time and hospital cost. In morbidly obese patients with large uteri, total laparoscopic hysterectomy is superior to vaginal hysterectomy with lesser odds of blood transfusion and lower length of hospital stay” (Clin Obstet Gynecol. 2017 Jun;60[2]:286-95).
What about the removal of fallopian tubes – salpingectomy – during vaginal hysterectomy? Dr. Kho highlighted a 2017 decision analysis that said these procedures are frequently performed for cancer prevention during laparoscopic and open hysterectomies “but [fallopian tubes] are not routinely removed during vaginal hysterectomy because of perceptions of increased morbidity, difficulty, or inadequate surgical training.”
The analysis, however, determined that “salpingectomy should routinely be performed with vaginal hysterectomy because it was the dominant and therefore cost-effective strategy. Complications are minimally increased, but the trade-off with cancer prevention is highly favorable.” (Am J Obstet Gynecol. 2017 Nov;217[5]:603.e1-603.e6).
Dr. Kho reported consulting for AbbVie, Olympus, and Applied Medical.
EXPERT ANALYSIS FROM PAGS
Clinical trial: Surgical glue for hernia repair
A observational, prospective trial is underway to study the use of Cyanoacrylate Fixation for Laparoscopic Repair of Inguinal Hernias, a multicenter registry, is currently enrolling patients for laparoscopic inguinal hernia repair using surgical tissue glue for mesh fixation.
The trial expects to enroll 1,000 patients and to be completed by December 2019. The primary outcome is postoperative pain evaluated by patient self-assessment using a visual analog scale. Secondary outcomes include intraoperative and postoperative complications, analgesic intake, postoperative quality of life, recurrences, and longer-term complications.
Included in the participant group are adult patients of both sexes with primary inguinal hernia. Exclusions include patients with recurrent inguinal hernia, patients previously treated with Lichtenstein technique, those allergic to the components of the tissue glue, and those whose life expectancy is under 1 year. The patients will be followed up to 1 year.
For further information about to the study, go to clinicaltrials.gov (NCT01669837).
A observational, prospective trial is underway to study the use of Cyanoacrylate Fixation for Laparoscopic Repair of Inguinal Hernias, a multicenter registry, is currently enrolling patients for laparoscopic inguinal hernia repair using surgical tissue glue for mesh fixation.
The trial expects to enroll 1,000 patients and to be completed by December 2019. The primary outcome is postoperative pain evaluated by patient self-assessment using a visual analog scale. Secondary outcomes include intraoperative and postoperative complications, analgesic intake, postoperative quality of life, recurrences, and longer-term complications.
Included in the participant group are adult patients of both sexes with primary inguinal hernia. Exclusions include patients with recurrent inguinal hernia, patients previously treated with Lichtenstein technique, those allergic to the components of the tissue glue, and those whose life expectancy is under 1 year. The patients will be followed up to 1 year.
For further information about to the study, go to clinicaltrials.gov (NCT01669837).
A observational, prospective trial is underway to study the use of Cyanoacrylate Fixation for Laparoscopic Repair of Inguinal Hernias, a multicenter registry, is currently enrolling patients for laparoscopic inguinal hernia repair using surgical tissue glue for mesh fixation.
The trial expects to enroll 1,000 patients and to be completed by December 2019. The primary outcome is postoperative pain evaluated by patient self-assessment using a visual analog scale. Secondary outcomes include intraoperative and postoperative complications, analgesic intake, postoperative quality of life, recurrences, and longer-term complications.
Included in the participant group are adult patients of both sexes with primary inguinal hernia. Exclusions include patients with recurrent inguinal hernia, patients previously treated with Lichtenstein technique, those allergic to the components of the tissue glue, and those whose life expectancy is under 1 year. The patients will be followed up to 1 year.
For further information about to the study, go to clinicaltrials.gov (NCT01669837).
LAIV4 was less effective for children than IIV against influenza A/H1N1pdm09
The live attenuated influenza vaccine was less effective against the influenza A/H1N1pdm09 virus in children and adolescents across multiple influenza seasons between 2013 and 2016, compared with the inactivated influenza vaccine, according to research published in the journal Pediatrics.
Jessie R. Chung, MPH, from the influenza division at the Centers for Disease Control and Prevention in Atlanta, and her colleagues performed an analysis of five different studies where vaccine effectiveness (VE) was examined for quadrivalent live attenuated vaccine (LAIV4) and inactivated influenza vaccine (IIV) in children and adolescents aged 2-17 years from 42 states.
The analysis included data from the U.S. Influenza Vaccine Effectiveness Network (6,793 patients), a study from the Louisiana State University Health Sciences Center (3,822 patients), the Influenza Clinical Investigation for Children (3,521 patients), Department of Defense Global, Laboratory-based, Influenza Surveillance Program (1,935 patients), and the Influenza Incidence Surveillance Project (1,102 patients) between the periods of 2013-2014 and 2015-2016. The researchers sourced current and previous season vaccination history from electronic medical records and immunization registries.
Of patients who were vaccinated across all seasons, there was 67% effectiveness against influenza A/H1N1pdm09 (95% confidence interval, 62%-72%) for those who received the IIV and 20% (95% CI, −6%-39%) for LAIV4. Among patients who received the LAIV4 vaccination, there was a significantly higher likelihood of developing influenza A/H1N1pdm09 (odds ratio, 2.66; 95% CI, 2.06-3.44) compared with patients who received the IIV vaccination.
With regard to other strains, there was similar effectiveness against influenza A/H3N2 and influenza B with LAIV4 and IIV vaccinations.
“In contrast to findings of reduced LAIV4 effectiveness against influenza A/H1N1pdm09 viruses, our results suggest a possible but nonsignificant benefit of LAIV4 over IIV against influenza B viruses, which has been described previously,” the investigators wrote.
Limitations of the study included having data only one season prior to enrollment and little available demographic information beyond age, gender, and geographic location.
The Influenza Clinical Investigation for Children was funded by MedImmune, a member of the AstraZeneca Group. Two of the researchers are employees of AstraZeneca. The other authors reported having no conflicts of interest. The U.S. Influenza Vaccine Effectiveness Network was supported by the CDC through cooperative agreements with the University of Michigan, Kaiser Permanente Washington Health Research Institute, Marshfield Clinic Research Institute, University of Pittsburgh, and Baylor Scott & White Health. At the University of Pittsburgh, the project also was supported by the National Institutes of Health.
SOURCE: Chung JR et al. Pediatrics. 2018. doi: 10.1542/peds.2018-2094.
There are many explanations for the decline in effectiveness of the live attenuated influenza vaccine (LAIV4), but the data are complicated by conflicting information from studies outside the United States indicating “reasonable protection” against influenza A/H1N1pdm09, A/H3N2, and influenza B, compared with the inactivated influenza virus (IIV), Pedro A. Piedra, MD, wrote in an accompanying editorial.
In 2016, the World Health Organization met to discuss LAIV effectiveness and highlighted factors such as methodological study differences, inadequate vaccine handling at distribution centers, intrinsic virological differences of the A/H1N1pdm09 virus, and increased preexisting population immunity in the United States since 2010 as potential explanations. During the transition from LAIV3 to LAIV4 for the 2013-2014 influenza season, viral interference may have also occurred when the influenza B strain was introduced into the vaccine, he added.
According to the CDC’s Advisory Committee on Immunization Practices (ACIP), viral growth properties of A/H1N1pdm09 has improved in LAIV4, and viral shedding also has improved for children between 2 years and 4 years of age. Although effectiveness numbers were not available for the ACIP recommendation, an interim analysis from Public Health England for the 2017-2018 influenza season found a vaccine effectiveness of 90.3% (95% confidence interval, 16.4%-98.9%).
“This early result is encouraging and supports the reintroduction of LAIV4 in the United States as an option for the control of seasonal influenza,” he said. “It also highlights the need for annual influenza vaccine effectiveness estimates and the importance of the U.S. Influenza Vaccine Effectiveness Network in providing updated information for ACIP recommendations.”
Dr. Piedra is from the departments of molecular virology and microbiology and pediatrics, Baylor College of Medicine, Houston. He reports being a consultant for AstraZeneca, Sanofi Pasteur, GlaxoSmithKline, and Merck Sharp and Dohme, and he has received travel support to present at an influenza seminar supported by Seqirus. His comments are from an editorial accompanying the article by Chung and colleagues ( Pediatrics. 2019. doi: 10.1542/peds.2018- 3290 ).
There are many explanations for the decline in effectiveness of the live attenuated influenza vaccine (LAIV4), but the data are complicated by conflicting information from studies outside the United States indicating “reasonable protection” against influenza A/H1N1pdm09, A/H3N2, and influenza B, compared with the inactivated influenza virus (IIV), Pedro A. Piedra, MD, wrote in an accompanying editorial.
In 2016, the World Health Organization met to discuss LAIV effectiveness and highlighted factors such as methodological study differences, inadequate vaccine handling at distribution centers, intrinsic virological differences of the A/H1N1pdm09 virus, and increased preexisting population immunity in the United States since 2010 as potential explanations. During the transition from LAIV3 to LAIV4 for the 2013-2014 influenza season, viral interference may have also occurred when the influenza B strain was introduced into the vaccine, he added.
According to the CDC’s Advisory Committee on Immunization Practices (ACIP), viral growth properties of A/H1N1pdm09 has improved in LAIV4, and viral shedding also has improved for children between 2 years and 4 years of age. Although effectiveness numbers were not available for the ACIP recommendation, an interim analysis from Public Health England for the 2017-2018 influenza season found a vaccine effectiveness of 90.3% (95% confidence interval, 16.4%-98.9%).
“This early result is encouraging and supports the reintroduction of LAIV4 in the United States as an option for the control of seasonal influenza,” he said. “It also highlights the need for annual influenza vaccine effectiveness estimates and the importance of the U.S. Influenza Vaccine Effectiveness Network in providing updated information for ACIP recommendations.”
Dr. Piedra is from the departments of molecular virology and microbiology and pediatrics, Baylor College of Medicine, Houston. He reports being a consultant for AstraZeneca, Sanofi Pasteur, GlaxoSmithKline, and Merck Sharp and Dohme, and he has received travel support to present at an influenza seminar supported by Seqirus. His comments are from an editorial accompanying the article by Chung and colleagues ( Pediatrics. 2019. doi: 10.1542/peds.2018- 3290 ).
There are many explanations for the decline in effectiveness of the live attenuated influenza vaccine (LAIV4), but the data are complicated by conflicting information from studies outside the United States indicating “reasonable protection” against influenza A/H1N1pdm09, A/H3N2, and influenza B, compared with the inactivated influenza virus (IIV), Pedro A. Piedra, MD, wrote in an accompanying editorial.
In 2016, the World Health Organization met to discuss LAIV effectiveness and highlighted factors such as methodological study differences, inadequate vaccine handling at distribution centers, intrinsic virological differences of the A/H1N1pdm09 virus, and increased preexisting population immunity in the United States since 2010 as potential explanations. During the transition from LAIV3 to LAIV4 for the 2013-2014 influenza season, viral interference may have also occurred when the influenza B strain was introduced into the vaccine, he added.
According to the CDC’s Advisory Committee on Immunization Practices (ACIP), viral growth properties of A/H1N1pdm09 has improved in LAIV4, and viral shedding also has improved for children between 2 years and 4 years of age. Although effectiveness numbers were not available for the ACIP recommendation, an interim analysis from Public Health England for the 2017-2018 influenza season found a vaccine effectiveness of 90.3% (95% confidence interval, 16.4%-98.9%).
“This early result is encouraging and supports the reintroduction of LAIV4 in the United States as an option for the control of seasonal influenza,” he said. “It also highlights the need for annual influenza vaccine effectiveness estimates and the importance of the U.S. Influenza Vaccine Effectiveness Network in providing updated information for ACIP recommendations.”
Dr. Piedra is from the departments of molecular virology and microbiology and pediatrics, Baylor College of Medicine, Houston. He reports being a consultant for AstraZeneca, Sanofi Pasteur, GlaxoSmithKline, and Merck Sharp and Dohme, and he has received travel support to present at an influenza seminar supported by Seqirus. His comments are from an editorial accompanying the article by Chung and colleagues ( Pediatrics. 2019. doi: 10.1542/peds.2018- 3290 ).
The live attenuated influenza vaccine was less effective against the influenza A/H1N1pdm09 virus in children and adolescents across multiple influenza seasons between 2013 and 2016, compared with the inactivated influenza vaccine, according to research published in the journal Pediatrics.
Jessie R. Chung, MPH, from the influenza division at the Centers for Disease Control and Prevention in Atlanta, and her colleagues performed an analysis of five different studies where vaccine effectiveness (VE) was examined for quadrivalent live attenuated vaccine (LAIV4) and inactivated influenza vaccine (IIV) in children and adolescents aged 2-17 years from 42 states.
The analysis included data from the U.S. Influenza Vaccine Effectiveness Network (6,793 patients), a study from the Louisiana State University Health Sciences Center (3,822 patients), the Influenza Clinical Investigation for Children (3,521 patients), Department of Defense Global, Laboratory-based, Influenza Surveillance Program (1,935 patients), and the Influenza Incidence Surveillance Project (1,102 patients) between the periods of 2013-2014 and 2015-2016. The researchers sourced current and previous season vaccination history from electronic medical records and immunization registries.
Of patients who were vaccinated across all seasons, there was 67% effectiveness against influenza A/H1N1pdm09 (95% confidence interval, 62%-72%) for those who received the IIV and 20% (95% CI, −6%-39%) for LAIV4. Among patients who received the LAIV4 vaccination, there was a significantly higher likelihood of developing influenza A/H1N1pdm09 (odds ratio, 2.66; 95% CI, 2.06-3.44) compared with patients who received the IIV vaccination.
With regard to other strains, there was similar effectiveness against influenza A/H3N2 and influenza B with LAIV4 and IIV vaccinations.
“In contrast to findings of reduced LAIV4 effectiveness against influenza A/H1N1pdm09 viruses, our results suggest a possible but nonsignificant benefit of LAIV4 over IIV against influenza B viruses, which has been described previously,” the investigators wrote.
Limitations of the study included having data only one season prior to enrollment and little available demographic information beyond age, gender, and geographic location.
The Influenza Clinical Investigation for Children was funded by MedImmune, a member of the AstraZeneca Group. Two of the researchers are employees of AstraZeneca. The other authors reported having no conflicts of interest. The U.S. Influenza Vaccine Effectiveness Network was supported by the CDC through cooperative agreements with the University of Michigan, Kaiser Permanente Washington Health Research Institute, Marshfield Clinic Research Institute, University of Pittsburgh, and Baylor Scott & White Health. At the University of Pittsburgh, the project also was supported by the National Institutes of Health.
SOURCE: Chung JR et al. Pediatrics. 2018. doi: 10.1542/peds.2018-2094.
The live attenuated influenza vaccine was less effective against the influenza A/H1N1pdm09 virus in children and adolescents across multiple influenza seasons between 2013 and 2016, compared with the inactivated influenza vaccine, according to research published in the journal Pediatrics.
Jessie R. Chung, MPH, from the influenza division at the Centers for Disease Control and Prevention in Atlanta, and her colleagues performed an analysis of five different studies where vaccine effectiveness (VE) was examined for quadrivalent live attenuated vaccine (LAIV4) and inactivated influenza vaccine (IIV) in children and adolescents aged 2-17 years from 42 states.
The analysis included data from the U.S. Influenza Vaccine Effectiveness Network (6,793 patients), a study from the Louisiana State University Health Sciences Center (3,822 patients), the Influenza Clinical Investigation for Children (3,521 patients), Department of Defense Global, Laboratory-based, Influenza Surveillance Program (1,935 patients), and the Influenza Incidence Surveillance Project (1,102 patients) between the periods of 2013-2014 and 2015-2016. The researchers sourced current and previous season vaccination history from electronic medical records and immunization registries.
Of patients who were vaccinated across all seasons, there was 67% effectiveness against influenza A/H1N1pdm09 (95% confidence interval, 62%-72%) for those who received the IIV and 20% (95% CI, −6%-39%) for LAIV4. Among patients who received the LAIV4 vaccination, there was a significantly higher likelihood of developing influenza A/H1N1pdm09 (odds ratio, 2.66; 95% CI, 2.06-3.44) compared with patients who received the IIV vaccination.
With regard to other strains, there was similar effectiveness against influenza A/H3N2 and influenza B with LAIV4 and IIV vaccinations.
“In contrast to findings of reduced LAIV4 effectiveness against influenza A/H1N1pdm09 viruses, our results suggest a possible but nonsignificant benefit of LAIV4 over IIV against influenza B viruses, which has been described previously,” the investigators wrote.
Limitations of the study included having data only one season prior to enrollment and little available demographic information beyond age, gender, and geographic location.
The Influenza Clinical Investigation for Children was funded by MedImmune, a member of the AstraZeneca Group. Two of the researchers are employees of AstraZeneca. The other authors reported having no conflicts of interest. The U.S. Influenza Vaccine Effectiveness Network was supported by the CDC through cooperative agreements with the University of Michigan, Kaiser Permanente Washington Health Research Institute, Marshfield Clinic Research Institute, University of Pittsburgh, and Baylor Scott & White Health. At the University of Pittsburgh, the project also was supported by the National Institutes of Health.
SOURCE: Chung JR et al. Pediatrics. 2018. doi: 10.1542/peds.2018-2094.
FROM PEDIATRICS
Key clinical point: The live attenuated influenza vaccine (LAIV4) was significantly less effective than was the inactivated influenza vaccine (IIV) for children against the influenza A/H1N1pdm09 virus across multiple flu seasons.
Major finding:
Study details: A combined analysis of five studies in the United States between the periods of 2013-2014 and 2015-2016 from the U.S. Influenza Vaccine Effectiveness Network.
Disclosures: The Influenza Clinical Investigation for Children was funded by MedImmune, a member of the AstraZeneca Group. Two of the researchers are employees of AstraZeneca. The other authors reported having no conflicts of interest. The U.S. Influenza Vaccine Effectiveness Network was supported by the CDC through cooperative agreements with the University of Michigan, Kaiser Permanente Washington Health Research Institute, Marshfield Clinic Research Institute, University of Pittsburgh, and Baylor Scott & White Health. At the University of Pittsburgh, the project also was supported by the National Institutes of Health.
Source: Chung JR et al. Pediatrics. 2018. doi: 10.1542/peds.2018-2094.
Actionable mutations more likely in women with both breast and uterine cancer
Women with both breast and uterine cancer are more likely to carry genetic mutations than women with either breast or uterine cancer alone, according to results of a retrospective analysis of test results.
The majority of the mutations identified were actionable, suggesting that women with breast and uterine cancer should be offered expanded genetic testing, authors of the analysis wrote in Gynecologic Oncology.
“Expanded testing for patients with breast and uterine cancer can help guide management by identifying more patients who may benefit from additional surveillance, along with at-risk family members,” said Kelly Fulk, an oncology genetic specialist at Ambry Genetics, and her coauthors.
The analysis by Ms. Fulk and her colleagues at St. Thomas Health, Nashville, and the University of California, Irvine, was based on a cohort of 52,000 women who had undergone multigene panel testing.
That cohort included 1,650 women with both breast and uterine cancer, of whom about 70% were white, and more than 94% were older than age 50 years at the time of testing. Their median age at first diagnosis was 56 years for breast cancer and 58 years for uterine cancer.
A total of 231 women with breast and uterine cancer, or 14.0%, carried at least one pathogenic mutation or likely pathogenic variant, Ms. Fulk and her colleagues reported. By comparison, mutations were seen in 9.3% of women with breast cancer only (P less than .001), 11.5% of women with uterine cancer only (P = 4.63 x 10–3), and 6.8% of women with no personal cancer history (P less than .001).
Women with both breast and uterine cancer more often had mutations in ATM, BARD1, BRCA2, MSH2, MSH6, PALB2, PMS2, and PTEN, when compared with those women with no personal cancer history, according to the report by Ms. Fulk and her coauthors.
When compared with women with just breast cancer, the women with both breast and uterine cancer more often had mutations in BRCA1, MLH1, MSH2, MSH6, PMS2 and PTEN, they added, noting that women with both cancers were twice as likely to have a BRCA1 mutation (odds ratio, 2.01; 95% confidence interval, 1.08-3.39, P = .016).
While this study had limitations, including its retrospective nature and use of genetic tests with varying numbers of genes analyzed, authors said the results nonetheless support expanded testing in women with both breast and uterine cancers to help guide therapy and cancer surveillance.
“Mutations associated with hereditary breast and ovarian cancer, Lynch syndrome, Cowden syndrome, and Li-Fraumeni syndrome have clear guidelines regarding management and surveillance for other cancers, which can benefit patients and their at-risk family members,” the investigators said.
Ms. Fulk and five coauthors reported that they are paid employees of Ambry Genetics. No other disclosures were provided.
SOURCE: Fulk K et al. Gynecol Oncol. 2019 Jan 3. doi: 10.1016/j.ygyno.2018.12.021.
Women with both breast and uterine cancer are more likely to carry genetic mutations than women with either breast or uterine cancer alone, according to results of a retrospective analysis of test results.
The majority of the mutations identified were actionable, suggesting that women with breast and uterine cancer should be offered expanded genetic testing, authors of the analysis wrote in Gynecologic Oncology.
“Expanded testing for patients with breast and uterine cancer can help guide management by identifying more patients who may benefit from additional surveillance, along with at-risk family members,” said Kelly Fulk, an oncology genetic specialist at Ambry Genetics, and her coauthors.
The analysis by Ms. Fulk and her colleagues at St. Thomas Health, Nashville, and the University of California, Irvine, was based on a cohort of 52,000 women who had undergone multigene panel testing.
That cohort included 1,650 women with both breast and uterine cancer, of whom about 70% were white, and more than 94% were older than age 50 years at the time of testing. Their median age at first diagnosis was 56 years for breast cancer and 58 years for uterine cancer.
A total of 231 women with breast and uterine cancer, or 14.0%, carried at least one pathogenic mutation or likely pathogenic variant, Ms. Fulk and her colleagues reported. By comparison, mutations were seen in 9.3% of women with breast cancer only (P less than .001), 11.5% of women with uterine cancer only (P = 4.63 x 10–3), and 6.8% of women with no personal cancer history (P less than .001).
Women with both breast and uterine cancer more often had mutations in ATM, BARD1, BRCA2, MSH2, MSH6, PALB2, PMS2, and PTEN, when compared with those women with no personal cancer history, according to the report by Ms. Fulk and her coauthors.
When compared with women with just breast cancer, the women with both breast and uterine cancer more often had mutations in BRCA1, MLH1, MSH2, MSH6, PMS2 and PTEN, they added, noting that women with both cancers were twice as likely to have a BRCA1 mutation (odds ratio, 2.01; 95% confidence interval, 1.08-3.39, P = .016).
While this study had limitations, including its retrospective nature and use of genetic tests with varying numbers of genes analyzed, authors said the results nonetheless support expanded testing in women with both breast and uterine cancers to help guide therapy and cancer surveillance.
“Mutations associated with hereditary breast and ovarian cancer, Lynch syndrome, Cowden syndrome, and Li-Fraumeni syndrome have clear guidelines regarding management and surveillance for other cancers, which can benefit patients and their at-risk family members,” the investigators said.
Ms. Fulk and five coauthors reported that they are paid employees of Ambry Genetics. No other disclosures were provided.
SOURCE: Fulk K et al. Gynecol Oncol. 2019 Jan 3. doi: 10.1016/j.ygyno.2018.12.021.
Women with both breast and uterine cancer are more likely to carry genetic mutations than women with either breast or uterine cancer alone, according to results of a retrospective analysis of test results.
The majority of the mutations identified were actionable, suggesting that women with breast and uterine cancer should be offered expanded genetic testing, authors of the analysis wrote in Gynecologic Oncology.
“Expanded testing for patients with breast and uterine cancer can help guide management by identifying more patients who may benefit from additional surveillance, along with at-risk family members,” said Kelly Fulk, an oncology genetic specialist at Ambry Genetics, and her coauthors.
The analysis by Ms. Fulk and her colleagues at St. Thomas Health, Nashville, and the University of California, Irvine, was based on a cohort of 52,000 women who had undergone multigene panel testing.
That cohort included 1,650 women with both breast and uterine cancer, of whom about 70% were white, and more than 94% were older than age 50 years at the time of testing. Their median age at first diagnosis was 56 years for breast cancer and 58 years for uterine cancer.
A total of 231 women with breast and uterine cancer, or 14.0%, carried at least one pathogenic mutation or likely pathogenic variant, Ms. Fulk and her colleagues reported. By comparison, mutations were seen in 9.3% of women with breast cancer only (P less than .001), 11.5% of women with uterine cancer only (P = 4.63 x 10–3), and 6.8% of women with no personal cancer history (P less than .001).
Women with both breast and uterine cancer more often had mutations in ATM, BARD1, BRCA2, MSH2, MSH6, PALB2, PMS2, and PTEN, when compared with those women with no personal cancer history, according to the report by Ms. Fulk and her coauthors.
When compared with women with just breast cancer, the women with both breast and uterine cancer more often had mutations in BRCA1, MLH1, MSH2, MSH6, PMS2 and PTEN, they added, noting that women with both cancers were twice as likely to have a BRCA1 mutation (odds ratio, 2.01; 95% confidence interval, 1.08-3.39, P = .016).
While this study had limitations, including its retrospective nature and use of genetic tests with varying numbers of genes analyzed, authors said the results nonetheless support expanded testing in women with both breast and uterine cancers to help guide therapy and cancer surveillance.
“Mutations associated with hereditary breast and ovarian cancer, Lynch syndrome, Cowden syndrome, and Li-Fraumeni syndrome have clear guidelines regarding management and surveillance for other cancers, which can benefit patients and their at-risk family members,” the investigators said.
Ms. Fulk and five coauthors reported that they are paid employees of Ambry Genetics. No other disclosures were provided.
SOURCE: Fulk K et al. Gynecol Oncol. 2019 Jan 3. doi: 10.1016/j.ygyno.2018.12.021.
FROM GYNECOLOGIC ONCOLOGY
Key clinical point: Women with both breast and uterine cancer are more likely to carry actionable mutations than do women with breast or uterine cancer alone.
Major finding: At least one actionable mutation was seen in 14% of women with breast and uterine cancer, compared with 9.3% of women with breast cancer only, 11.5% of women with uterine cancer only, and 6.8% of women with no personal cancer history.
Study details: A retrospective analysis of a cohort of nearly 52,000 patients who underwent multigene panel testing.
Disclosures: Ms. Fulk and five coauthors reported that they are paid employees of Ambry Genetics.
Source: Fulk K et al. Gynecol Oncol. 2019 Jan 3. doi: 10.1016/j.ygyno.2018.12.021.
Appropriate use criteria for imaging in nonvalvular heart disease released
The American College of Cardiology, the American Heart Association, and other groups have jointly released an appropriate use criteria (AUC) document regarding the use of imaging modalities in diagnosing nonvalvular (that is, structural) heart disease.
Imaging plays an important role in diagnosing both valvular and nonvalvular heart diseases, so the goal of the document was to help clinicians provide high-quality care by standardizing the decision-making process. To do so, a committee was formed to devise scenarios that reflected situations in real-world practice; these scenarios were considered within categories to prevent the list from being too exhaustive. The scenarios were then reviewed by a rating panel in terms of how appropriate certain modalities were in each situation. The panel members first evaluated the scenarios independently then face to face as a panel before giving their final scores (from 1 to 9) independently.
For example, for the indication of nonsustained ventricular tachycardia, the panelists rated transthoracic echocardiography with or without 3-D and with contrast as needed as a 8, which means it’s an “appropriate test,” whereas they gave CT for the same indication a 3, which means “rarely appropriate.” For sustained ventricular tachycardia or ventricular fibrillation, they gave a 9 and a 6, respectively; this latter score indicates the test “may be appropriate.” These scenarios and the respective scores for any given test are organized into tables, such as initial evaluation or follow-up.
This AUC document “signals a shift from documents evaluating a single modality in various disease states to documents evaluating multiple imaging modalities and focusing on evidence and clinical experience within a given disease category,” the authors wrote. “We believe this approach better reflects clinical decision making in real-world scenarios and offers the diagnostic choices available to the clinician.”
The full document can be viewed in JACC.
The American College of Cardiology, the American Heart Association, and other groups have jointly released an appropriate use criteria (AUC) document regarding the use of imaging modalities in diagnosing nonvalvular (that is, structural) heart disease.
Imaging plays an important role in diagnosing both valvular and nonvalvular heart diseases, so the goal of the document was to help clinicians provide high-quality care by standardizing the decision-making process. To do so, a committee was formed to devise scenarios that reflected situations in real-world practice; these scenarios were considered within categories to prevent the list from being too exhaustive. The scenarios were then reviewed by a rating panel in terms of how appropriate certain modalities were in each situation. The panel members first evaluated the scenarios independently then face to face as a panel before giving their final scores (from 1 to 9) independently.
For example, for the indication of nonsustained ventricular tachycardia, the panelists rated transthoracic echocardiography with or without 3-D and with contrast as needed as a 8, which means it’s an “appropriate test,” whereas they gave CT for the same indication a 3, which means “rarely appropriate.” For sustained ventricular tachycardia or ventricular fibrillation, they gave a 9 and a 6, respectively; this latter score indicates the test “may be appropriate.” These scenarios and the respective scores for any given test are organized into tables, such as initial evaluation or follow-up.
This AUC document “signals a shift from documents evaluating a single modality in various disease states to documents evaluating multiple imaging modalities and focusing on evidence and clinical experience within a given disease category,” the authors wrote. “We believe this approach better reflects clinical decision making in real-world scenarios and offers the diagnostic choices available to the clinician.”
The full document can be viewed in JACC.
The American College of Cardiology, the American Heart Association, and other groups have jointly released an appropriate use criteria (AUC) document regarding the use of imaging modalities in diagnosing nonvalvular (that is, structural) heart disease.
Imaging plays an important role in diagnosing both valvular and nonvalvular heart diseases, so the goal of the document was to help clinicians provide high-quality care by standardizing the decision-making process. To do so, a committee was formed to devise scenarios that reflected situations in real-world practice; these scenarios were considered within categories to prevent the list from being too exhaustive. The scenarios were then reviewed by a rating panel in terms of how appropriate certain modalities were in each situation. The panel members first evaluated the scenarios independently then face to face as a panel before giving their final scores (from 1 to 9) independently.
For example, for the indication of nonsustained ventricular tachycardia, the panelists rated transthoracic echocardiography with or without 3-D and with contrast as needed as a 8, which means it’s an “appropriate test,” whereas they gave CT for the same indication a 3, which means “rarely appropriate.” For sustained ventricular tachycardia or ventricular fibrillation, they gave a 9 and a 6, respectively; this latter score indicates the test “may be appropriate.” These scenarios and the respective scores for any given test are organized into tables, such as initial evaluation or follow-up.
This AUC document “signals a shift from documents evaluating a single modality in various disease states to documents evaluating multiple imaging modalities and focusing on evidence and clinical experience within a given disease category,” the authors wrote. “We believe this approach better reflects clinical decision making in real-world scenarios and offers the diagnostic choices available to the clinician.”
The full document can be viewed in JACC.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Inguinal hernia recurrence rates in women lower after laparoscopic repair
according to findings published in JAMA Surgery.
In a systematic review of 43,870 female patients in 55 studies, the recurrence rate was 1.2% after laparoscopic repair, compared with 2.4% after open repair. The recurrent hernia was a femoral hernia in 40.9% of patients after open repair, compared with no recurrences after laparoscopic repair, reported Line Schmidt of the department of surgery at Herlev (Denmark) Hospital, and coauthors.
Patients were women aged 18 years and older who had repair of a primary unilateral or bilateral inguinal hernia. The review included all retrospective cohort studies, prospective cohort studies, prospective clinical trials, and retrospective cohort studies with 20 or more women with inguinal hernias. PubMed, Embase, and the Cochrane Library databases were searched. The primary outcome was recurrence rate after primary laparoscopic and open repairs, with or without mesh.
The overall recurrence rate among women was 2.6%. The overall crude recurrence rate for studies with low risk of bias was 3.9%, the authors wrote. A femoral hernia was found in 43% of reoperations, though in one study including both open and laparoscopic repairs in women, the rate of detection of incidental femoral hernia was only 2%.
The results “support the recommendation that women with inguinal hernias should undergo laparoscopic repair, unless there are concerns where an open repair might be more clinically appropriate,” the authors wrote. “A substantial number of recurrences after open repair were femoral hernias that may have been overlooked at the primary operation.”
Authors reported receiving personal fees from Bard and Merck. No other disclosures were reported.
SOURCE: Schmidt L et al. JAMA Surg. 2018;153(12):1135-42.
according to findings published in JAMA Surgery.
In a systematic review of 43,870 female patients in 55 studies, the recurrence rate was 1.2% after laparoscopic repair, compared with 2.4% after open repair. The recurrent hernia was a femoral hernia in 40.9% of patients after open repair, compared with no recurrences after laparoscopic repair, reported Line Schmidt of the department of surgery at Herlev (Denmark) Hospital, and coauthors.
Patients were women aged 18 years and older who had repair of a primary unilateral or bilateral inguinal hernia. The review included all retrospective cohort studies, prospective cohort studies, prospective clinical trials, and retrospective cohort studies with 20 or more women with inguinal hernias. PubMed, Embase, and the Cochrane Library databases were searched. The primary outcome was recurrence rate after primary laparoscopic and open repairs, with or without mesh.
The overall recurrence rate among women was 2.6%. The overall crude recurrence rate for studies with low risk of bias was 3.9%, the authors wrote. A femoral hernia was found in 43% of reoperations, though in one study including both open and laparoscopic repairs in women, the rate of detection of incidental femoral hernia was only 2%.
The results “support the recommendation that women with inguinal hernias should undergo laparoscopic repair, unless there are concerns where an open repair might be more clinically appropriate,” the authors wrote. “A substantial number of recurrences after open repair were femoral hernias that may have been overlooked at the primary operation.”
Authors reported receiving personal fees from Bard and Merck. No other disclosures were reported.
SOURCE: Schmidt L et al. JAMA Surg. 2018;153(12):1135-42.
according to findings published in JAMA Surgery.
In a systematic review of 43,870 female patients in 55 studies, the recurrence rate was 1.2% after laparoscopic repair, compared with 2.4% after open repair. The recurrent hernia was a femoral hernia in 40.9% of patients after open repair, compared with no recurrences after laparoscopic repair, reported Line Schmidt of the department of surgery at Herlev (Denmark) Hospital, and coauthors.
Patients were women aged 18 years and older who had repair of a primary unilateral or bilateral inguinal hernia. The review included all retrospective cohort studies, prospective cohort studies, prospective clinical trials, and retrospective cohort studies with 20 or more women with inguinal hernias. PubMed, Embase, and the Cochrane Library databases were searched. The primary outcome was recurrence rate after primary laparoscopic and open repairs, with or without mesh.
The overall recurrence rate among women was 2.6%. The overall crude recurrence rate for studies with low risk of bias was 3.9%, the authors wrote. A femoral hernia was found in 43% of reoperations, though in one study including both open and laparoscopic repairs in women, the rate of detection of incidental femoral hernia was only 2%.
The results “support the recommendation that women with inguinal hernias should undergo laparoscopic repair, unless there are concerns where an open repair might be more clinically appropriate,” the authors wrote. “A substantial number of recurrences after open repair were femoral hernias that may have been overlooked at the primary operation.”
Authors reported receiving personal fees from Bard and Merck. No other disclosures were reported.
SOURCE: Schmidt L et al. JAMA Surg. 2018;153(12):1135-42.
FROM JAMA SURGERY
SHM announces National Hospitalist Day
Inaugural day of recognition to honor hospital medicine care team
The Society of Hospital Medicine is proud to announce the inaugural National Hospitalist Day, to be held on Thursday, March 7, 2019. Occurring the first Thursday in March annually, National Hospitalist Day will serve to celebrate the fastest-growing specialty in modern medicine and hospitalists’ enduring contributions to the evolving health care landscape.
National Hospitalist Day was recently approved by the National Day Calendar and was one of approximately 30 national days to be approved for the year out of an applicant pool of more than 18,000.
“As the only society dedicated to the specialty of hospital medicine, it is appropriate that SHM spearhead a national day to recognize the countless contributions of hospitalists to health care, from clinical, academic, and leadership perspectives and more,” said Larry Wellikson, MD, MHM, chief executive officer of SHM. “We look forward to hospitalists across the nation contributing to the festivities and making this a tradition for years to come.”
In addition to celebrating hospitalists’ contributions to patient care, SHM will also be highlighting the diverse career paths of hospital medicine professionals, from frontline hospitalist physicians, nurse practitioners, and physician assistants to practice administrators, C-suite executives, and academic hospitalists.
Highlights of SHM’s campaign include the following:
- Downloadable customizable posters and assets for hospitals and individuals’ offices to celebrate their hospital medicine team, available on SHM’s website, hospitalmedicine.org.
- A series of spotlights of hospitalists at all stages of their careers in The Hospitalist, SHM’s monthly newsmagazine.
- A social media campaign inviting hospitalists and their employers to share their success stories using the hashtag #HowWeHospitalist, including banner graphics, profile photo overlays, and more.
- A social media contest to determine the most creative ways of celebrating use of the hashtag.
- A Twitter chat for hospitalists to celebrate virtually with their colleagues and peers from around the world.
“Hospitalists innovate, lead, and push the boundaries of clinical care and deserve to be recognized for their transformative contributions to health care,” said Eric E. Howell, MD, MHM, chief operating officer of SHM. “We hope this is the beginning of a long-standing tradition in honoring hospitalists and the noteworthy work they do.”
For more information, visit www.hospitalmedicine.org/hospitalistday.
Mr. Radler is marketing communications manager at the Society of Hospital Medicine.
Inaugural day of recognition to honor hospital medicine care team
Inaugural day of recognition to honor hospital medicine care team
The Society of Hospital Medicine is proud to announce the inaugural National Hospitalist Day, to be held on Thursday, March 7, 2019. Occurring the first Thursday in March annually, National Hospitalist Day will serve to celebrate the fastest-growing specialty in modern medicine and hospitalists’ enduring contributions to the evolving health care landscape.
National Hospitalist Day was recently approved by the National Day Calendar and was one of approximately 30 national days to be approved for the year out of an applicant pool of more than 18,000.
“As the only society dedicated to the specialty of hospital medicine, it is appropriate that SHM spearhead a national day to recognize the countless contributions of hospitalists to health care, from clinical, academic, and leadership perspectives and more,” said Larry Wellikson, MD, MHM, chief executive officer of SHM. “We look forward to hospitalists across the nation contributing to the festivities and making this a tradition for years to come.”
In addition to celebrating hospitalists’ contributions to patient care, SHM will also be highlighting the diverse career paths of hospital medicine professionals, from frontline hospitalist physicians, nurse practitioners, and physician assistants to practice administrators, C-suite executives, and academic hospitalists.
Highlights of SHM’s campaign include the following:
- Downloadable customizable posters and assets for hospitals and individuals’ offices to celebrate their hospital medicine team, available on SHM’s website, hospitalmedicine.org.
- A series of spotlights of hospitalists at all stages of their careers in The Hospitalist, SHM’s monthly newsmagazine.
- A social media campaign inviting hospitalists and their employers to share their success stories using the hashtag #HowWeHospitalist, including banner graphics, profile photo overlays, and more.
- A social media contest to determine the most creative ways of celebrating use of the hashtag.
- A Twitter chat for hospitalists to celebrate virtually with their colleagues and peers from around the world.
“Hospitalists innovate, lead, and push the boundaries of clinical care and deserve to be recognized for their transformative contributions to health care,” said Eric E. Howell, MD, MHM, chief operating officer of SHM. “We hope this is the beginning of a long-standing tradition in honoring hospitalists and the noteworthy work they do.”
For more information, visit www.hospitalmedicine.org/hospitalistday.
Mr. Radler is marketing communications manager at the Society of Hospital Medicine.
The Society of Hospital Medicine is proud to announce the inaugural National Hospitalist Day, to be held on Thursday, March 7, 2019. Occurring the first Thursday in March annually, National Hospitalist Day will serve to celebrate the fastest-growing specialty in modern medicine and hospitalists’ enduring contributions to the evolving health care landscape.
National Hospitalist Day was recently approved by the National Day Calendar and was one of approximately 30 national days to be approved for the year out of an applicant pool of more than 18,000.
“As the only society dedicated to the specialty of hospital medicine, it is appropriate that SHM spearhead a national day to recognize the countless contributions of hospitalists to health care, from clinical, academic, and leadership perspectives and more,” said Larry Wellikson, MD, MHM, chief executive officer of SHM. “We look forward to hospitalists across the nation contributing to the festivities and making this a tradition for years to come.”
In addition to celebrating hospitalists’ contributions to patient care, SHM will also be highlighting the diverse career paths of hospital medicine professionals, from frontline hospitalist physicians, nurse practitioners, and physician assistants to practice administrators, C-suite executives, and academic hospitalists.
Highlights of SHM’s campaign include the following:
- Downloadable customizable posters and assets for hospitals and individuals’ offices to celebrate their hospital medicine team, available on SHM’s website, hospitalmedicine.org.
- A series of spotlights of hospitalists at all stages of their careers in The Hospitalist, SHM’s monthly newsmagazine.
- A social media campaign inviting hospitalists and their employers to share their success stories using the hashtag #HowWeHospitalist, including banner graphics, profile photo overlays, and more.
- A social media contest to determine the most creative ways of celebrating use of the hashtag.
- A Twitter chat for hospitalists to celebrate virtually with their colleagues and peers from around the world.
“Hospitalists innovate, lead, and push the boundaries of clinical care and deserve to be recognized for their transformative contributions to health care,” said Eric E. Howell, MD, MHM, chief operating officer of SHM. “We hope this is the beginning of a long-standing tradition in honoring hospitalists and the noteworthy work they do.”
For more information, visit www.hospitalmedicine.org/hospitalistday.
Mr. Radler is marketing communications manager at the Society of Hospital Medicine.
Knee pathologies, including multiple meniscal tears, predict accelerated OA
Accelerated knee osteoarthritis is characterized by distinct features that include destabilizing meniscal tears in two or more areas as well as other pathologies, based on data from the Osteoarthritis Initiative.
The possibility of accelerated knee osteoarthritis (AKOA) as a unique subset of knee osteoarthritis has not been well studied, wrote Jeffrey B. Driban, PhD, of Tufts University, Boston, and his colleagues.
“If specific pathologies differentiate people at risk for AKOA it may help identify adults with early-stage or high-risk for AKOA and inspire novel prevention strategies,” they wrote in their report, published in Arthritis & Rheumatology.
The researchers reviewed data from three groups of adults selected from participants in the Osteoarthritis Initiative, a cohort of 4,796 adults with KOA or at risk for symptomatic KOA who were recruited at four clinical sites in the United States. These groups included 125 with AKOA, 125 with typical knee osteoarthritis (KOA), and 125 without knee OA.
Overall, patients with AKOA were approximately seven times more likely than were patients with KOA to have destabilizing meniscal tears in two or more areas at the time of the index visit (42% vs. 14%); less than 5% of adults with no KOA experienced destabilizing meniscal tears. In addition, patients with AKOA were more than four times as likely to have miscellaneous pathology starting the year before the index visit, compared with those without AKOA.
Approximately 63% of the participants in each group were women, and the majority were overweight. The average age, weight, and global impact of arthritis were greater in the AKOA group when compared against the typical KOA and no-KOA groups.
Participants were assessed via MRI reviewed by radiologists who were blinded to the groups.
At the index visit, 49% of adults with AKOA had either a destabilizing meniscal tear or miscellaneous pathology, compared with 15% of adults with KOA and 6% of adults without KOA.
Adults with AKOA also showed significantly greater cartilage loss prior to the index visit in comparison with typical KOA patients, and AKOA patients had less cartilage in the medial and lateral tibia and medial femur, compared with adults who had typical KOA or no KOA after the index visit.
Adults who developed AKOA showed a significantly higher bone marrow lesion volume when compared against the typical KOA and no-KOA groups at 1 year prior to the index visit, and their bone marrow lesion volume increased on average 13 times more compared with typical KOA patients over the 2 years before the index visit, the researchers noted (2.00 mL vs. 0.15 mL, respectively).
“These findings add to the evidence that AKOA is different [from] the typically perceived archetype of slow-progressing osteoarthritis” with a unique risk profile, the researchers said.
The study findings were limited by several factors, including the relatively small sample size, uncertain timing of disease onset, a potentially limited definition of a destabilizing meniscal tear (defined as a root tear, radial tear, or complex tear, which almost always featured a radial component), a lack of a universal AKOA pathology, and some missing MRI data, the researchers noted. However, the results support previous studies suggesting a link between meniscal pathology and increased risk for AKOA, they said.
“It is important to acknowledge that it remains unclear if AKOA has any relation to type 2 rapidly progressive osteoarthritis, which was characterized by a more dramatic joint space narrowing (2 mm or more within 1 year) and greater abnormal bone loss/destruction,” they noted.
“Future research with a larger sample size of adults at risk for AKOA may help further refine our understanding of AKOA and help develop a clinically useful predictive model,” they added.
The study was supported in part by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and private funding included Merck, Novartis, GlaxoSmithKline, and Pfizer. The researchers had no financial conflicts to disclose.
SOURCE: Driban JB et al. Arthritis Rheumatol. 2018 Dec 28. doi: 10.1002/art.40826.
Accelerated knee osteoarthritis is characterized by distinct features that include destabilizing meniscal tears in two or more areas as well as other pathologies, based on data from the Osteoarthritis Initiative.
The possibility of accelerated knee osteoarthritis (AKOA) as a unique subset of knee osteoarthritis has not been well studied, wrote Jeffrey B. Driban, PhD, of Tufts University, Boston, and his colleagues.
“If specific pathologies differentiate people at risk for AKOA it may help identify adults with early-stage or high-risk for AKOA and inspire novel prevention strategies,” they wrote in their report, published in Arthritis & Rheumatology.
The researchers reviewed data from three groups of adults selected from participants in the Osteoarthritis Initiative, a cohort of 4,796 adults with KOA or at risk for symptomatic KOA who were recruited at four clinical sites in the United States. These groups included 125 with AKOA, 125 with typical knee osteoarthritis (KOA), and 125 without knee OA.
Overall, patients with AKOA were approximately seven times more likely than were patients with KOA to have destabilizing meniscal tears in two or more areas at the time of the index visit (42% vs. 14%); less than 5% of adults with no KOA experienced destabilizing meniscal tears. In addition, patients with AKOA were more than four times as likely to have miscellaneous pathology starting the year before the index visit, compared with those without AKOA.
Approximately 63% of the participants in each group were women, and the majority were overweight. The average age, weight, and global impact of arthritis were greater in the AKOA group when compared against the typical KOA and no-KOA groups.
Participants were assessed via MRI reviewed by radiologists who were blinded to the groups.
At the index visit, 49% of adults with AKOA had either a destabilizing meniscal tear or miscellaneous pathology, compared with 15% of adults with KOA and 6% of adults without KOA.
Adults with AKOA also showed significantly greater cartilage loss prior to the index visit in comparison with typical KOA patients, and AKOA patients had less cartilage in the medial and lateral tibia and medial femur, compared with adults who had typical KOA or no KOA after the index visit.
Adults who developed AKOA showed a significantly higher bone marrow lesion volume when compared against the typical KOA and no-KOA groups at 1 year prior to the index visit, and their bone marrow lesion volume increased on average 13 times more compared with typical KOA patients over the 2 years before the index visit, the researchers noted (2.00 mL vs. 0.15 mL, respectively).
“These findings add to the evidence that AKOA is different [from] the typically perceived archetype of slow-progressing osteoarthritis” with a unique risk profile, the researchers said.
The study findings were limited by several factors, including the relatively small sample size, uncertain timing of disease onset, a potentially limited definition of a destabilizing meniscal tear (defined as a root tear, radial tear, or complex tear, which almost always featured a radial component), a lack of a universal AKOA pathology, and some missing MRI data, the researchers noted. However, the results support previous studies suggesting a link between meniscal pathology and increased risk for AKOA, they said.
“It is important to acknowledge that it remains unclear if AKOA has any relation to type 2 rapidly progressive osteoarthritis, which was characterized by a more dramatic joint space narrowing (2 mm or more within 1 year) and greater abnormal bone loss/destruction,” they noted.
“Future research with a larger sample size of adults at risk for AKOA may help further refine our understanding of AKOA and help develop a clinically useful predictive model,” they added.
The study was supported in part by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and private funding included Merck, Novartis, GlaxoSmithKline, and Pfizer. The researchers had no financial conflicts to disclose.
SOURCE: Driban JB et al. Arthritis Rheumatol. 2018 Dec 28. doi: 10.1002/art.40826.
Accelerated knee osteoarthritis is characterized by distinct features that include destabilizing meniscal tears in two or more areas as well as other pathologies, based on data from the Osteoarthritis Initiative.
The possibility of accelerated knee osteoarthritis (AKOA) as a unique subset of knee osteoarthritis has not been well studied, wrote Jeffrey B. Driban, PhD, of Tufts University, Boston, and his colleagues.
“If specific pathologies differentiate people at risk for AKOA it may help identify adults with early-stage or high-risk for AKOA and inspire novel prevention strategies,” they wrote in their report, published in Arthritis & Rheumatology.
The researchers reviewed data from three groups of adults selected from participants in the Osteoarthritis Initiative, a cohort of 4,796 adults with KOA or at risk for symptomatic KOA who were recruited at four clinical sites in the United States. These groups included 125 with AKOA, 125 with typical knee osteoarthritis (KOA), and 125 without knee OA.
Overall, patients with AKOA were approximately seven times more likely than were patients with KOA to have destabilizing meniscal tears in two or more areas at the time of the index visit (42% vs. 14%); less than 5% of adults with no KOA experienced destabilizing meniscal tears. In addition, patients with AKOA were more than four times as likely to have miscellaneous pathology starting the year before the index visit, compared with those without AKOA.
Approximately 63% of the participants in each group were women, and the majority were overweight. The average age, weight, and global impact of arthritis were greater in the AKOA group when compared against the typical KOA and no-KOA groups.
Participants were assessed via MRI reviewed by radiologists who were blinded to the groups.
At the index visit, 49% of adults with AKOA had either a destabilizing meniscal tear or miscellaneous pathology, compared with 15% of adults with KOA and 6% of adults without KOA.
Adults with AKOA also showed significantly greater cartilage loss prior to the index visit in comparison with typical KOA patients, and AKOA patients had less cartilage in the medial and lateral tibia and medial femur, compared with adults who had typical KOA or no KOA after the index visit.
Adults who developed AKOA showed a significantly higher bone marrow lesion volume when compared against the typical KOA and no-KOA groups at 1 year prior to the index visit, and their bone marrow lesion volume increased on average 13 times more compared with typical KOA patients over the 2 years before the index visit, the researchers noted (2.00 mL vs. 0.15 mL, respectively).
“These findings add to the evidence that AKOA is different [from] the typically perceived archetype of slow-progressing osteoarthritis” with a unique risk profile, the researchers said.
The study findings were limited by several factors, including the relatively small sample size, uncertain timing of disease onset, a potentially limited definition of a destabilizing meniscal tear (defined as a root tear, radial tear, or complex tear, which almost always featured a radial component), a lack of a universal AKOA pathology, and some missing MRI data, the researchers noted. However, the results support previous studies suggesting a link between meniscal pathology and increased risk for AKOA, they said.
“It is important to acknowledge that it remains unclear if AKOA has any relation to type 2 rapidly progressive osteoarthritis, which was characterized by a more dramatic joint space narrowing (2 mm or more within 1 year) and greater abnormal bone loss/destruction,” they noted.
“Future research with a larger sample size of adults at risk for AKOA may help further refine our understanding of AKOA and help develop a clinically useful predictive model,” they added.
The study was supported in part by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and private funding included Merck, Novartis, GlaxoSmithKline, and Pfizer. The researchers had no financial conflicts to disclose.
SOURCE: Driban JB et al. Arthritis Rheumatol. 2018 Dec 28. doi: 10.1002/art.40826.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point:
Major finding: One year before the knee OA index visit, more than 75% of patients with accelerated knee OA had meniscal damage in at least two regions.
Study details: The data come from 375 adults with typical knee OA, accelerated knee OA, or no knee OA in the longitudinal Osteoarthritis Initiative cohort study.
Disclosures: The study was supported in part by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and private funding included Merck, Novartis, GlaxoSmithKline, and Pfizer. The researchers had no financial conflicts to disclose.
Source: Driban JB et al. Arthritis Rheumatol. 2018 Dec 28. doi: 10.1002/art.40826.