User login
Ketamine infusions may be helpful in central sensitization pain syndromes
LAS VEGAS – Ketamine infusions are a reasonable option for patients with central sensitization pain syndromes, Jay Joshi, MD, said at the annual PAINWeek.
This disparate group of disorders includes pain experienced by both the body and mind: anxiety and depression, complex regional pain syndrome, opioid-induced hyperalgesia, phantom limb pain, fibromyalgia, and PTSD. In trained hands, ketamine infusions can benefit all of them, often providing the first relief for patients frustrated by years of seeking help for a medical disorder that has no obvious physical cause, said Dr. Joshi, CEO and medical director of the National Pain Centers in Vernon Hills, Ill.
Central sensitization is a CNS response to pain, often chronic, that results in increased neural activity or an increased response to stimuli that wouldn’t normally be interpreted as pain. The root causes can be peripheral injury, persistent inflammation, or neural injury.
“Central sensitization is produced by increases in excitability and reduction in inhibitory transmission, which may produce a persistent enhancement of pain sensitivity,” Dr. Joshi said. These changes include increased glutaminergic signaling – the target of ketamine’s action as an N-methyl-D-aspartate (NMDA) receptor blocker.
By blocking glutamate reuptake and increasing it in the synapse, ketamine “resets the hyperalgesia hyperexcitatory pathway that’s been stuck in this ‘on’ position,” Dr. Joshi said in an interview. “As a selective NMDA receptor antagonist, ketamine seems to be binding to a subreceptor that’s responsible for the symptoms that patients with these syndromes experience. Other NMDA receptor antagonists don’t give the same results. By turning off the signal, we’re giving the nervous system a chance to reset” and return to a more normally functioning state.
“Even though these people have had an injury and aren’t functioning normally, they still have normal neural pathways that can perceive sensation correctly,” he added.
Ketamine is only approved as an injectable anesthetic, but has been gaining popularity as a treatment for depression and other psychiatric disorders, as well as pain. Reports have been so positive that the Food and Drug Administration is considering approval of a ketamine-based nasal spray – esketamine – that’s being developed by Johnson & Johnson. The company reported positive phase 3 data during the May meeting of the American Society of Clinical Psychopharmacology.
When paired with an oral antidepressant, and compared with a placebo spray, esketamine significantly increased the number of responders and remitters and decreased relapses. Based on these results, and three other positive phase 3 studies, Johnson & Johnson submitted for FDA approval in September 2018.
Anecdotal reports of significant relief of chronic pain associated with ketamine have made pain another attractive off-label use, despite a paucity of high-quality data. In July 2018, a consortium of the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists published evidence-based guidelines for the drug’s use in chronic pain (Reg Anesth Pain Med. 2018 Jul;43[5]:521-46). Overall, the panel found weak evidence supporting its use for most conditions, except for moderate evidence for complex regional pain syndrome.
- Spinal cord injury pain: Weak evidence for short-term benefit at doses of 0.42-0.4 mg/kg per hour ranging from 17 minutes to 5 hours for 7 consecutive days.
- Complex regional pain syndrome: Moderate evidence for pain improvement up to 12 weeks at doses of 22 mg/hour for 4 days or 0.35 mg/kg per hour over 4 hours daily for 10 days.
- Mixed neuropathic pain, phantom limb pain, postherpetic neuralgia, fibromyalgia, cancer pain, ischemic pain, migraine headache, and low-back pain: weak to no evidence.
Nevertheless, Dr. Joshi is a firm believer in ketamine’s benefit for pain patients, when it’s administered at appropriate doses by clinicians trained in anesthesia. “Our main clinic is in a surgical center and we administer ketamine under a surgical protocol. This is a powerful anesthetic and should be treated as such,” he said. Patients are risk-stratified with the Anesthesiology Society of America physical status classification system and constantly monitored during the infusions.
These kinds of precautions are not generally taken in the dozens of unregulated “ketamine clinics” continue to open across the country, Dr. Joshi said. “They’re typically not staffed by anesthesiologists or nurse anesthetists, but by other providers without adequate training who may have only taken a weekend or online course in how to administer the drug.”
Dr. Joshi reported no disclosures relevant to his presentation.
LAS VEGAS – Ketamine infusions are a reasonable option for patients with central sensitization pain syndromes, Jay Joshi, MD, said at the annual PAINWeek.
This disparate group of disorders includes pain experienced by both the body and mind: anxiety and depression, complex regional pain syndrome, opioid-induced hyperalgesia, phantom limb pain, fibromyalgia, and PTSD. In trained hands, ketamine infusions can benefit all of them, often providing the first relief for patients frustrated by years of seeking help for a medical disorder that has no obvious physical cause, said Dr. Joshi, CEO and medical director of the National Pain Centers in Vernon Hills, Ill.
Central sensitization is a CNS response to pain, often chronic, that results in increased neural activity or an increased response to stimuli that wouldn’t normally be interpreted as pain. The root causes can be peripheral injury, persistent inflammation, or neural injury.
“Central sensitization is produced by increases in excitability and reduction in inhibitory transmission, which may produce a persistent enhancement of pain sensitivity,” Dr. Joshi said. These changes include increased glutaminergic signaling – the target of ketamine’s action as an N-methyl-D-aspartate (NMDA) receptor blocker.
By blocking glutamate reuptake and increasing it in the synapse, ketamine “resets the hyperalgesia hyperexcitatory pathway that’s been stuck in this ‘on’ position,” Dr. Joshi said in an interview. “As a selective NMDA receptor antagonist, ketamine seems to be binding to a subreceptor that’s responsible for the symptoms that patients with these syndromes experience. Other NMDA receptor antagonists don’t give the same results. By turning off the signal, we’re giving the nervous system a chance to reset” and return to a more normally functioning state.
“Even though these people have had an injury and aren’t functioning normally, they still have normal neural pathways that can perceive sensation correctly,” he added.
Ketamine is only approved as an injectable anesthetic, but has been gaining popularity as a treatment for depression and other psychiatric disorders, as well as pain. Reports have been so positive that the Food and Drug Administration is considering approval of a ketamine-based nasal spray – esketamine – that’s being developed by Johnson & Johnson. The company reported positive phase 3 data during the May meeting of the American Society of Clinical Psychopharmacology.
When paired with an oral antidepressant, and compared with a placebo spray, esketamine significantly increased the number of responders and remitters and decreased relapses. Based on these results, and three other positive phase 3 studies, Johnson & Johnson submitted for FDA approval in September 2018.
Anecdotal reports of significant relief of chronic pain associated with ketamine have made pain another attractive off-label use, despite a paucity of high-quality data. In July 2018, a consortium of the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists published evidence-based guidelines for the drug’s use in chronic pain (Reg Anesth Pain Med. 2018 Jul;43[5]:521-46). Overall, the panel found weak evidence supporting its use for most conditions, except for moderate evidence for complex regional pain syndrome.
- Spinal cord injury pain: Weak evidence for short-term benefit at doses of 0.42-0.4 mg/kg per hour ranging from 17 minutes to 5 hours for 7 consecutive days.
- Complex regional pain syndrome: Moderate evidence for pain improvement up to 12 weeks at doses of 22 mg/hour for 4 days or 0.35 mg/kg per hour over 4 hours daily for 10 days.
- Mixed neuropathic pain, phantom limb pain, postherpetic neuralgia, fibromyalgia, cancer pain, ischemic pain, migraine headache, and low-back pain: weak to no evidence.
Nevertheless, Dr. Joshi is a firm believer in ketamine’s benefit for pain patients, when it’s administered at appropriate doses by clinicians trained in anesthesia. “Our main clinic is in a surgical center and we administer ketamine under a surgical protocol. This is a powerful anesthetic and should be treated as such,” he said. Patients are risk-stratified with the Anesthesiology Society of America physical status classification system and constantly monitored during the infusions.
These kinds of precautions are not generally taken in the dozens of unregulated “ketamine clinics” continue to open across the country, Dr. Joshi said. “They’re typically not staffed by anesthesiologists or nurse anesthetists, but by other providers without adequate training who may have only taken a weekend or online course in how to administer the drug.”
Dr. Joshi reported no disclosures relevant to his presentation.
LAS VEGAS – Ketamine infusions are a reasonable option for patients with central sensitization pain syndromes, Jay Joshi, MD, said at the annual PAINWeek.
This disparate group of disorders includes pain experienced by both the body and mind: anxiety and depression, complex regional pain syndrome, opioid-induced hyperalgesia, phantom limb pain, fibromyalgia, and PTSD. In trained hands, ketamine infusions can benefit all of them, often providing the first relief for patients frustrated by years of seeking help for a medical disorder that has no obvious physical cause, said Dr. Joshi, CEO and medical director of the National Pain Centers in Vernon Hills, Ill.
Central sensitization is a CNS response to pain, often chronic, that results in increased neural activity or an increased response to stimuli that wouldn’t normally be interpreted as pain. The root causes can be peripheral injury, persistent inflammation, or neural injury.
“Central sensitization is produced by increases in excitability and reduction in inhibitory transmission, which may produce a persistent enhancement of pain sensitivity,” Dr. Joshi said. These changes include increased glutaminergic signaling – the target of ketamine’s action as an N-methyl-D-aspartate (NMDA) receptor blocker.
By blocking glutamate reuptake and increasing it in the synapse, ketamine “resets the hyperalgesia hyperexcitatory pathway that’s been stuck in this ‘on’ position,” Dr. Joshi said in an interview. “As a selective NMDA receptor antagonist, ketamine seems to be binding to a subreceptor that’s responsible for the symptoms that patients with these syndromes experience. Other NMDA receptor antagonists don’t give the same results. By turning off the signal, we’re giving the nervous system a chance to reset” and return to a more normally functioning state.
“Even though these people have had an injury and aren’t functioning normally, they still have normal neural pathways that can perceive sensation correctly,” he added.
Ketamine is only approved as an injectable anesthetic, but has been gaining popularity as a treatment for depression and other psychiatric disorders, as well as pain. Reports have been so positive that the Food and Drug Administration is considering approval of a ketamine-based nasal spray – esketamine – that’s being developed by Johnson & Johnson. The company reported positive phase 3 data during the May meeting of the American Society of Clinical Psychopharmacology.
When paired with an oral antidepressant, and compared with a placebo spray, esketamine significantly increased the number of responders and remitters and decreased relapses. Based on these results, and three other positive phase 3 studies, Johnson & Johnson submitted for FDA approval in September 2018.
Anecdotal reports of significant relief of chronic pain associated with ketamine have made pain another attractive off-label use, despite a paucity of high-quality data. In July 2018, a consortium of the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists published evidence-based guidelines for the drug’s use in chronic pain (Reg Anesth Pain Med. 2018 Jul;43[5]:521-46). Overall, the panel found weak evidence supporting its use for most conditions, except for moderate evidence for complex regional pain syndrome.
- Spinal cord injury pain: Weak evidence for short-term benefit at doses of 0.42-0.4 mg/kg per hour ranging from 17 minutes to 5 hours for 7 consecutive days.
- Complex regional pain syndrome: Moderate evidence for pain improvement up to 12 weeks at doses of 22 mg/hour for 4 days or 0.35 mg/kg per hour over 4 hours daily for 10 days.
- Mixed neuropathic pain, phantom limb pain, postherpetic neuralgia, fibromyalgia, cancer pain, ischemic pain, migraine headache, and low-back pain: weak to no evidence.
Nevertheless, Dr. Joshi is a firm believer in ketamine’s benefit for pain patients, when it’s administered at appropriate doses by clinicians trained in anesthesia. “Our main clinic is in a surgical center and we administer ketamine under a surgical protocol. This is a powerful anesthetic and should be treated as such,” he said. Patients are risk-stratified with the Anesthesiology Society of America physical status classification system and constantly monitored during the infusions.
These kinds of precautions are not generally taken in the dozens of unregulated “ketamine clinics” continue to open across the country, Dr. Joshi said. “They’re typically not staffed by anesthesiologists or nurse anesthetists, but by other providers without adequate training who may have only taken a weekend or online course in how to administer the drug.”
Dr. Joshi reported no disclosures relevant to his presentation.
EXPERT ANALYSIS FROM PAINWEEK 2018
Register for October Coding Course
Learn to use the correct codes, use all the codes necessary and submit everything properly the first time at the SVS Coding and Reimbursement Workshop, Oct. 19 and 20 at the Renaissance Chicago Downtown Hotel in Chicago. These skills learned can help vascular surgeons avoid an audit, as correct coding lessens the risk for one. And an audit costs staff time and money no matter what the outcome. Book hotel rooms by Sept. 27 to receive the special room rate for participants.
Learn to use the correct codes, use all the codes necessary and submit everything properly the first time at the SVS Coding and Reimbursement Workshop, Oct. 19 and 20 at the Renaissance Chicago Downtown Hotel in Chicago. These skills learned can help vascular surgeons avoid an audit, as correct coding lessens the risk for one. And an audit costs staff time and money no matter what the outcome. Book hotel rooms by Sept. 27 to receive the special room rate for participants.
Learn to use the correct codes, use all the codes necessary and submit everything properly the first time at the SVS Coding and Reimbursement Workshop, Oct. 19 and 20 at the Renaissance Chicago Downtown Hotel in Chicago. These skills learned can help vascular surgeons avoid an audit, as correct coding lessens the risk for one. And an audit costs staff time and money no matter what the outcome. Book hotel rooms by Sept. 27 to receive the special room rate for participants.
Bridge Grant Applications Due Oct. 1
Applications are due Oct. 1 for the SVS Foundation's new Bridge Grant, which provides funding from one grant to another. The grant targets those investigators with previous national funding for basic research, such as an NIH R01 grant, and who applied for another R01 grant but did not receive a high enough priority score to be funded again. Apply today.
Applications are due Oct. 1 for the SVS Foundation's new Bridge Grant, which provides funding from one grant to another. The grant targets those investigators with previous national funding for basic research, such as an NIH R01 grant, and who applied for another R01 grant but did not receive a high enough priority score to be funded again. Apply today.
Applications are due Oct. 1 for the SVS Foundation's new Bridge Grant, which provides funding from one grant to another. The grant targets those investigators with previous national funding for basic research, such as an NIH R01 grant, and who applied for another R01 grant but did not receive a high enough priority score to be funded again. Apply today.
Outcomes of Palliative Care Consults With Hospitalized Veterans
Families and patients receive emotional support and better care planning after palliative care consultations.
Inpatient palliative care (IPC) consultation services have been widely adopted in US hospitals. Outcomes research has demonstrated improved quality of life (QOL) for palliative inpatients for symptom control and satisfaction with care.1-5 Families benefit from emotional support, care planning, and transitions of care.4,6-8 Outcomes, including hospital length of stay, hospital costs, and discharge dispositionalso seem to improve.9-17 The Department of Veterans Affairs (VA) provides palliative care (PC) consultation teams at its hospitals nationwide; however, few studies exist to show how a PC service is used at a VA hospital. The following study of a PC consult team at an urban VA facility provides a unique picture of how a PC team is used.
Methods
The John Cochran Division of the VA St. Louis Health Care System (VASLHCS) in Missouri is a 509-bed adult acute care hospital with medical and surgical specialties and subspecialties available for veterans, including an intensive care unit (ICU). The PC team is one of the subspecialty teams following patients after consultation and consists of a PC physician, nurse practitioner, chaplain, social worker, and psychologist.
Data Collection
This study was exempt from internal review board approval. The attending physician kept track of each IPC encounter between September 2014 and April 2016. Data were retrieved from the Computerized Patient Record System by identifying charts that included family meeting notes during the specified time. All 130 patients included in this study were followed by the PC team. Patient charts were reviewed, and information was uploaded to spreadsheets, which became the database for this study. The data included age, patient location, diagnosis, number of days between admission and PC consultation, and number of days between admission and family meeting. Other data included code status changes and discharge dispositions. Only consultations that resulted in direct patient contact were included.
The VASLHCS requires therapeutic support level (TSL), or code status, documentation by the attending physician regarding the discussion with a competent patient or valid representative if the patient is incapacitated. Levels of support are TSL I ‘‘no limitation on care,’’ TSL II ‘‘partial code,’’ that is, usually no cardiopulmonary resuscitation or do not intubate with selected medical measures to continue, and TSL III ‘‘comfort measures only.’’ If a patient’s code level changed after IPC consultation, the change is recorded.
Data Analysis
The files were purged of all unique personal health history. Because there was no control group, multivariable analyses of association were not warranted. Analysis was confined to descriptive measures.
Results
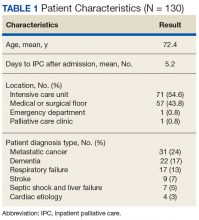
A total of 130 patients with IPC consultations were included in this retrospective study conducted from September 2014 to April 2016 (Table 1).
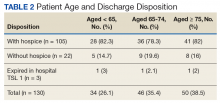
The scope of IPC consultations usually include medical recommendations about symptom management, discharge planning, discussion about goals of care (GOC), code status and prognosis, managing expected in-hospital expirations (deaths), and determination of hospice eligibility. Of the IPC cohort, 74% were aged > 65 years; 26.1% were aged < 65 years (Table 2).
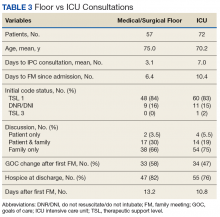
The mean days for an initial IPC consultation following admission was 3 on the medical/surgical floors and 7 days for ICU (P = .003; 95% CI, -6.37 to 1.36).
Discussion
Although small, the proportion of patients with serious illness or multiple chronic conditions account for a disproportionately large portion of health care spending.18 Despite the high cost, evidence demonstrates that these patients receive health care of inadequate quality characterized by fragmentation, overuse, medical errors, and poor QOL. Multiple studies show that IPC consultation provides improved patient outcomes and decreased hospital costs.9-17
From a purely outcomes-based interpretation, IPC consultation was associated with 83% of patients receiving a change in code status from full code/TSL 1. The study team drew 2 main conclusions from the data: (1) The IPC consultation is an effective way to broach GOC discussion and adjust code status; and (2) These data suggest room for earlier PC involvement. Remarkably, only 3 patients (2%) expired while inpatient with full code status.
The data also provide a unique comparison of timing of PC referrals. Pantilat and colleagues published characteristics of PC consultation services in California hospitals, and on average, patients were in the hospital 5.9 days (median 5.5; SD 3.3) prior to referral.19 This study’s average number of days for initial IPC consultation following admission was 3 days on the medical/surgical floors and 7 days in the ICU. Both time frames seem reasonable but again indicate some potential improvement for earlier IPC utilization.
Although the time frame of the intervention limited the number of patients in this study, early PC consultations in the acute care setting are a helpful intervention for veterans and families to better understand the complexity of their medical condition and prognosis and allow for a frank and open discussion about realistic goals. The importance of these discussions also were reflected in the high percentage of patients transitioning to hospice level of care (80%) and the low number of patients who remained full code (3 of 130). Other studies have shown conflicting results when interventions have been exclusively for cancer patients. In this study, 45% of patients were admitted with diagnoses other than cancer compared with 24% of patients with related diagnoses in a study by Gonsalves and colleagues.20
In this study, the majority (71.6%) of family meetings were held only with family (no patient involvement), resulting in missed opportunities for earlier patient and PC involvement especially for those patients with serious medical illnesses.
A systematic review published by Wendler and colleagues found that surrogate decision makers often find that role troubling and traumatizing even with advance directive documents.21 Earlier identification and PC consultations could initiate discussions between patients and their loved ones to decide “when enough is enough,” and about whether or not to prolong the dying process, when compatible with the patient’s wishes.
Early PC consultations also could highlight a potential highly vulnerable population of medically unbefriended patients (elder orphans). These patients may have no one in their lives to act as surrogate decision makers. This situation calls for further interventions regarding early identification of these patients and better processes to assist in their decision making. Many physicians believe it is not appropriate to begin advance directive planning on an outpatient basis. However, multiple studies have shown that patients want their doctors to discuss advance care planning with them before they become ill.22 Many other doctors have shown a positive response from patients when advance directive discussions are held during outpatient visits.23
The goals of this study were to evaluate the effectiveness of IPC consultation on goals of care and to address code status with patients and their families. Along with these conversations, the study team provided comprehensive PC evaluation. The PC team focused on providing excellent symptom management. The team of PC physicians, pain specialists, pain pharmacists, a chaplain, psychologists, and social workers addressed all the bio-psycho-social needs of patients/families and provided comprehensive recommendations. This multidimensional approach has gained significant acceptance.24
At VASLHCS, the program has grown to about 600 new consults per year, with a dedicated inpatient hospice unit, daily outpatient clinic, and myriad learning opportunities for trainees; the center has become a main site of rotation for hospice and palliative care fellows from training programs in St. Louis.
Utilization of PC consultation to help meeting the veterans’ needs at the bio-psycho-social level will also provide a benefit for the facility as it will decrease observed/expected standardized mortality ratio (SMR) data. This reduction of SMR data will be a result of successful patient transitions to hospice level of care at least 12 months prior to their passing or if their level of care is changed to inpatient hospice after they are admitted, the patients won’t be included as acute care mortality. However, with this initial small group of patients it was not possible to retrospectively calculate the impact on SMR or SAIL (Strategic Analytics for Improvement and Learning) indicators. The long-term expectation is to have a positive impact on those indicators represented by decreased inpatient mortality and improved SAIL.
Limitations
This study was a single-institution study, but every institution has its own internal culture. The team did not have a concurrent or historic control for comparison or use a questionnaire for patients and families rating their satisfaction.
Conclusion
This study provides multiple future directions of research as the authors now have baseline data about how the service is used. Future areas of interest would be to study the effectiveness of early palliative care interventions, such as a provider education series, implementation of consultation criteria, and prospective measurement of the impact of palliative care consultations on the SMR and SAIL indicators. This research could help identify which early interventions show the best efficacy, an area where research is notably lacking.25
1. El-Jawahri A, Greer JA, Temel JS. Does palliative care improve outcomes for patients with incurable illness? A review of the evidence. J Support Oncol. 2011;9(3):87-94.
2. Higginson IJ, Finlay I, Goodwin DM, et al. Do hospital-based palliative teams improve care for patients or families at the end of life? J Pain Symptom Manage. 2002;23(2):96-106.
3. Gade G, Venohr I, Conner D, et al. Impact of an inpatient palliative care team: a randomized control trial. J Palliat Med. 2008;11(2):180-190.
4. Benzar E, Hansen L, Kneitel AW, Fromme EK. Discharge planning for palliative care patients: a qualitative analysis. J Palliat Med. 2011;14(1):65-69.
5. Enguidanos S, Housen P, Penido M, Mejia B, Miller JA. Family members’ perceptions of inpatient palliative care consult services: a qualitative study. Palliat Med. 2014;28(1):42-48.
6. Granda-Cameron C, Viola SR, Lynch MP, Polomano RC. Measuring patient-oriented outcomes in palliative care: functionality and quality of life. Clin J Oncol Nurs. 2008;12(1):65-77.
7. Chand P, Gabriel T, Wallace CL, Nelson CM. Inpatient palliative care consultation: describing patient satisfaction. Perm J. 2013;17(1):53-55.
8. Tangeman JC, Rudra CB, Kerr CW, Grant PC. A hospice-hospital partnership: reducing hospitalization costs and 30-day readmissions among seriously ill adults. J Palliat Med. 2014;17(9):1005-1010.
9. Fromme EK, Bascom PB, Smith MD, et al. Survival, mortality, and location of death for patients seen by a hospital-based palliative care team. J Palliat Med. 2006;9(4):903-911.
10. Penrod JD, Deb P, Dellenbaugh C, et al. Hospital-based palliative care consultation: effects on hospital cost. J Palliat Med. 2010;13(8):973-979.
11. Ranganathan A, Dougherty M, Waite D, Casarett D. Can palliative home care reduce 30-day readmissions? Results of a propensity score matched cohort study. J Palliat Med. 2013;16(10):1290-1293.
12. Starks H, Wang S, Farber S, Owens DA, Curtis JR. Cost savings vary by length of stay for inpatients receiving palliative care consultation services. J Palliat Med. 2013;16(10):1215-1220.
13. Goldenheim A, Oates D, Parker V, Russell M, Winter M, Silliman RA. Rehospitalization of older adults discharged to home hospice care. J Palliat Med. 2014;17(7):841-844.
14. May P, Normand C, Morrison RS. Economic impact of hospital inpatient palliative care consultation: review of current evidence and directions for future research. J Palliat Med. 2014;17(9):1054-1063.
15. Granda-Cameron C, Behta M, Hovinga M, Rundio A, Mintzer D. Risk factors associated with unplanned hospital readmissions in adults with cancer. Oncol Nurs Forum. 2015;42(3):e257-e268.
16. Brody AA, Ciemins E, Newman J, Harrington C. The effects of an inpatient palliative care team on discharge disposition. J Palliat Med. 2010;13(5):541-548.
17. Penrod JD, Deb P, Luhrs C, et al. Cost and utilization outcomes of patients receiving hospital-based palliative care consultation. J Palliat Med. 2006;9(4):855-860.
18. Aldridge MD, Kelley AS. Appendix E, Epidemiology of serious illness and high utilization of health care. In: Institute of Medicine, Committee on Approaching Death: Addressing Key End of Life Issues. Dying in America: Improving Quality and Honoring Individual Preferences Near the End of Life. Washington, DC: National Academies Press; 2015.
19. Pantilat SZ, Kerr KM, Billings JA, Bruno KA, O’Riordan DL. Characteristics of palliative care consultation services in California hospitals. J Palliat Med. 2012;15(5):555-560.
20. Gonsalves WI, Tashi T, Krishnamurthy J, et al. Effect of palliative care services on the aggressiveness of end-of-life care in the Veterans Affairs cancer population. J Palliat Med. 2011;14(11):1231-1235.
21. Wendler D, Rid A. Systematic review: the effect on surrogates of making treatment decisions for others. Ann Intern Med. 2011;154(5):336-346.
22. American Bar Association Commission on Law and Aging. Myths and facts about health care advance directives. https://www.americanbar.org/content/dam/aba/publications/bifocal/BIFOCALSept-Oct2015.authcheckdam.pdf. Accessed July 10, 2018.
23. Tierney WM, Dexter PR, Gramelspacher GP, Perkins AJ, Zhou X-H, Wolinsky FD. The effect of discussions about advance directives on patients’ satisfaction with primary care. J Gen Intern Med. 2001;16(1):32-40.
24. Bailey FA, Williams BR, Woodby LL, et al. Intervention to improve care at life’s end in inpatient settings: the BEACON trial. J Gen Intern Med. 2014;29(6):836-843.
25. Dalgaard K, Bergenholtz H, Nielsen M, Timm H. Early integration of palliative care in hospitals: a systematic review on methods, barriers, and outcome. Palliat Support Care. 2014;12(6):495-513.
Families and patients receive emotional support and better care planning after palliative care consultations.
Families and patients receive emotional support and better care planning after palliative care consultations.
Inpatient palliative care (IPC) consultation services have been widely adopted in US hospitals. Outcomes research has demonstrated improved quality of life (QOL) for palliative inpatients for symptom control and satisfaction with care.1-5 Families benefit from emotional support, care planning, and transitions of care.4,6-8 Outcomes, including hospital length of stay, hospital costs, and discharge dispositionalso seem to improve.9-17 The Department of Veterans Affairs (VA) provides palliative care (PC) consultation teams at its hospitals nationwide; however, few studies exist to show how a PC service is used at a VA hospital. The following study of a PC consult team at an urban VA facility provides a unique picture of how a PC team is used.
Methods
The John Cochran Division of the VA St. Louis Health Care System (VASLHCS) in Missouri is a 509-bed adult acute care hospital with medical and surgical specialties and subspecialties available for veterans, including an intensive care unit (ICU). The PC team is one of the subspecialty teams following patients after consultation and consists of a PC physician, nurse practitioner, chaplain, social worker, and psychologist.
Data Collection
This study was exempt from internal review board approval. The attending physician kept track of each IPC encounter between September 2014 and April 2016. Data were retrieved from the Computerized Patient Record System by identifying charts that included family meeting notes during the specified time. All 130 patients included in this study were followed by the PC team. Patient charts were reviewed, and information was uploaded to spreadsheets, which became the database for this study. The data included age, patient location, diagnosis, number of days between admission and PC consultation, and number of days between admission and family meeting. Other data included code status changes and discharge dispositions. Only consultations that resulted in direct patient contact were included.
The VASLHCS requires therapeutic support level (TSL), or code status, documentation by the attending physician regarding the discussion with a competent patient or valid representative if the patient is incapacitated. Levels of support are TSL I ‘‘no limitation on care,’’ TSL II ‘‘partial code,’’ that is, usually no cardiopulmonary resuscitation or do not intubate with selected medical measures to continue, and TSL III ‘‘comfort measures only.’’ If a patient’s code level changed after IPC consultation, the change is recorded.
Data Analysis
The files were purged of all unique personal health history. Because there was no control group, multivariable analyses of association were not warranted. Analysis was confined to descriptive measures.
Results
A total of 130 patients with IPC consultations were included in this retrospective study conducted from September 2014 to April 2016 (Table 1).
The scope of IPC consultations usually include medical recommendations about symptom management, discharge planning, discussion about goals of care (GOC), code status and prognosis, managing expected in-hospital expirations (deaths), and determination of hospice eligibility. Of the IPC cohort, 74% were aged > 65 years; 26.1% were aged < 65 years (Table 2).
The mean days for an initial IPC consultation following admission was 3 on the medical/surgical floors and 7 days for ICU (P = .003; 95% CI, -6.37 to 1.36).
Discussion
Although small, the proportion of patients with serious illness or multiple chronic conditions account for a disproportionately large portion of health care spending.18 Despite the high cost, evidence demonstrates that these patients receive health care of inadequate quality characterized by fragmentation, overuse, medical errors, and poor QOL. Multiple studies show that IPC consultation provides improved patient outcomes and decreased hospital costs.9-17
From a purely outcomes-based interpretation, IPC consultation was associated with 83% of patients receiving a change in code status from full code/TSL 1. The study team drew 2 main conclusions from the data: (1) The IPC consultation is an effective way to broach GOC discussion and adjust code status; and (2) These data suggest room for earlier PC involvement. Remarkably, only 3 patients (2%) expired while inpatient with full code status.
The data also provide a unique comparison of timing of PC referrals. Pantilat and colleagues published characteristics of PC consultation services in California hospitals, and on average, patients were in the hospital 5.9 days (median 5.5; SD 3.3) prior to referral.19 This study’s average number of days for initial IPC consultation following admission was 3 days on the medical/surgical floors and 7 days in the ICU. Both time frames seem reasonable but again indicate some potential improvement for earlier IPC utilization.
Although the time frame of the intervention limited the number of patients in this study, early PC consultations in the acute care setting are a helpful intervention for veterans and families to better understand the complexity of their medical condition and prognosis and allow for a frank and open discussion about realistic goals. The importance of these discussions also were reflected in the high percentage of patients transitioning to hospice level of care (80%) and the low number of patients who remained full code (3 of 130). Other studies have shown conflicting results when interventions have been exclusively for cancer patients. In this study, 45% of patients were admitted with diagnoses other than cancer compared with 24% of patients with related diagnoses in a study by Gonsalves and colleagues.20
In this study, the majority (71.6%) of family meetings were held only with family (no patient involvement), resulting in missed opportunities for earlier patient and PC involvement especially for those patients with serious medical illnesses.
A systematic review published by Wendler and colleagues found that surrogate decision makers often find that role troubling and traumatizing even with advance directive documents.21 Earlier identification and PC consultations could initiate discussions between patients and their loved ones to decide “when enough is enough,” and about whether or not to prolong the dying process, when compatible with the patient’s wishes.
Early PC consultations also could highlight a potential highly vulnerable population of medically unbefriended patients (elder orphans). These patients may have no one in their lives to act as surrogate decision makers. This situation calls for further interventions regarding early identification of these patients and better processes to assist in their decision making. Many physicians believe it is not appropriate to begin advance directive planning on an outpatient basis. However, multiple studies have shown that patients want their doctors to discuss advance care planning with them before they become ill.22 Many other doctors have shown a positive response from patients when advance directive discussions are held during outpatient visits.23
The goals of this study were to evaluate the effectiveness of IPC consultation on goals of care and to address code status with patients and their families. Along with these conversations, the study team provided comprehensive PC evaluation. The PC team focused on providing excellent symptom management. The team of PC physicians, pain specialists, pain pharmacists, a chaplain, psychologists, and social workers addressed all the bio-psycho-social needs of patients/families and provided comprehensive recommendations. This multidimensional approach has gained significant acceptance.24
At VASLHCS, the program has grown to about 600 new consults per year, with a dedicated inpatient hospice unit, daily outpatient clinic, and myriad learning opportunities for trainees; the center has become a main site of rotation for hospice and palliative care fellows from training programs in St. Louis.
Utilization of PC consultation to help meeting the veterans’ needs at the bio-psycho-social level will also provide a benefit for the facility as it will decrease observed/expected standardized mortality ratio (SMR) data. This reduction of SMR data will be a result of successful patient transitions to hospice level of care at least 12 months prior to their passing or if their level of care is changed to inpatient hospice after they are admitted, the patients won’t be included as acute care mortality. However, with this initial small group of patients it was not possible to retrospectively calculate the impact on SMR or SAIL (Strategic Analytics for Improvement and Learning) indicators. The long-term expectation is to have a positive impact on those indicators represented by decreased inpatient mortality and improved SAIL.
Limitations
This study was a single-institution study, but every institution has its own internal culture. The team did not have a concurrent or historic control for comparison or use a questionnaire for patients and families rating their satisfaction.
Conclusion
This study provides multiple future directions of research as the authors now have baseline data about how the service is used. Future areas of interest would be to study the effectiveness of early palliative care interventions, such as a provider education series, implementation of consultation criteria, and prospective measurement of the impact of palliative care consultations on the SMR and SAIL indicators. This research could help identify which early interventions show the best efficacy, an area where research is notably lacking.25
Inpatient palliative care (IPC) consultation services have been widely adopted in US hospitals. Outcomes research has demonstrated improved quality of life (QOL) for palliative inpatients for symptom control and satisfaction with care.1-5 Families benefit from emotional support, care planning, and transitions of care.4,6-8 Outcomes, including hospital length of stay, hospital costs, and discharge dispositionalso seem to improve.9-17 The Department of Veterans Affairs (VA) provides palliative care (PC) consultation teams at its hospitals nationwide; however, few studies exist to show how a PC service is used at a VA hospital. The following study of a PC consult team at an urban VA facility provides a unique picture of how a PC team is used.
Methods
The John Cochran Division of the VA St. Louis Health Care System (VASLHCS) in Missouri is a 509-bed adult acute care hospital with medical and surgical specialties and subspecialties available for veterans, including an intensive care unit (ICU). The PC team is one of the subspecialty teams following patients after consultation and consists of a PC physician, nurse practitioner, chaplain, social worker, and psychologist.
Data Collection
This study was exempt from internal review board approval. The attending physician kept track of each IPC encounter between September 2014 and April 2016. Data were retrieved from the Computerized Patient Record System by identifying charts that included family meeting notes during the specified time. All 130 patients included in this study were followed by the PC team. Patient charts were reviewed, and information was uploaded to spreadsheets, which became the database for this study. The data included age, patient location, diagnosis, number of days between admission and PC consultation, and number of days between admission and family meeting. Other data included code status changes and discharge dispositions. Only consultations that resulted in direct patient contact were included.
The VASLHCS requires therapeutic support level (TSL), or code status, documentation by the attending physician regarding the discussion with a competent patient or valid representative if the patient is incapacitated. Levels of support are TSL I ‘‘no limitation on care,’’ TSL II ‘‘partial code,’’ that is, usually no cardiopulmonary resuscitation or do not intubate with selected medical measures to continue, and TSL III ‘‘comfort measures only.’’ If a patient’s code level changed after IPC consultation, the change is recorded.
Data Analysis
The files were purged of all unique personal health history. Because there was no control group, multivariable analyses of association were not warranted. Analysis was confined to descriptive measures.
Results
A total of 130 patients with IPC consultations were included in this retrospective study conducted from September 2014 to April 2016 (Table 1).
The scope of IPC consultations usually include medical recommendations about symptom management, discharge planning, discussion about goals of care (GOC), code status and prognosis, managing expected in-hospital expirations (deaths), and determination of hospice eligibility. Of the IPC cohort, 74% were aged > 65 years; 26.1% were aged < 65 years (Table 2).
The mean days for an initial IPC consultation following admission was 3 on the medical/surgical floors and 7 days for ICU (P = .003; 95% CI, -6.37 to 1.36).
Discussion
Although small, the proportion of patients with serious illness or multiple chronic conditions account for a disproportionately large portion of health care spending.18 Despite the high cost, evidence demonstrates that these patients receive health care of inadequate quality characterized by fragmentation, overuse, medical errors, and poor QOL. Multiple studies show that IPC consultation provides improved patient outcomes and decreased hospital costs.9-17
From a purely outcomes-based interpretation, IPC consultation was associated with 83% of patients receiving a change in code status from full code/TSL 1. The study team drew 2 main conclusions from the data: (1) The IPC consultation is an effective way to broach GOC discussion and adjust code status; and (2) These data suggest room for earlier PC involvement. Remarkably, only 3 patients (2%) expired while inpatient with full code status.
The data also provide a unique comparison of timing of PC referrals. Pantilat and colleagues published characteristics of PC consultation services in California hospitals, and on average, patients were in the hospital 5.9 days (median 5.5; SD 3.3) prior to referral.19 This study’s average number of days for initial IPC consultation following admission was 3 days on the medical/surgical floors and 7 days in the ICU. Both time frames seem reasonable but again indicate some potential improvement for earlier IPC utilization.
Although the time frame of the intervention limited the number of patients in this study, early PC consultations in the acute care setting are a helpful intervention for veterans and families to better understand the complexity of their medical condition and prognosis and allow for a frank and open discussion about realistic goals. The importance of these discussions also were reflected in the high percentage of patients transitioning to hospice level of care (80%) and the low number of patients who remained full code (3 of 130). Other studies have shown conflicting results when interventions have been exclusively for cancer patients. In this study, 45% of patients were admitted with diagnoses other than cancer compared with 24% of patients with related diagnoses in a study by Gonsalves and colleagues.20
In this study, the majority (71.6%) of family meetings were held only with family (no patient involvement), resulting in missed opportunities for earlier patient and PC involvement especially for those patients with serious medical illnesses.
A systematic review published by Wendler and colleagues found that surrogate decision makers often find that role troubling and traumatizing even with advance directive documents.21 Earlier identification and PC consultations could initiate discussions between patients and their loved ones to decide “when enough is enough,” and about whether or not to prolong the dying process, when compatible with the patient’s wishes.
Early PC consultations also could highlight a potential highly vulnerable population of medically unbefriended patients (elder orphans). These patients may have no one in their lives to act as surrogate decision makers. This situation calls for further interventions regarding early identification of these patients and better processes to assist in their decision making. Many physicians believe it is not appropriate to begin advance directive planning on an outpatient basis. However, multiple studies have shown that patients want their doctors to discuss advance care planning with them before they become ill.22 Many other doctors have shown a positive response from patients when advance directive discussions are held during outpatient visits.23
The goals of this study were to evaluate the effectiveness of IPC consultation on goals of care and to address code status with patients and their families. Along with these conversations, the study team provided comprehensive PC evaluation. The PC team focused on providing excellent symptom management. The team of PC physicians, pain specialists, pain pharmacists, a chaplain, psychologists, and social workers addressed all the bio-psycho-social needs of patients/families and provided comprehensive recommendations. This multidimensional approach has gained significant acceptance.24
At VASLHCS, the program has grown to about 600 new consults per year, with a dedicated inpatient hospice unit, daily outpatient clinic, and myriad learning opportunities for trainees; the center has become a main site of rotation for hospice and palliative care fellows from training programs in St. Louis.
Utilization of PC consultation to help meeting the veterans’ needs at the bio-psycho-social level will also provide a benefit for the facility as it will decrease observed/expected standardized mortality ratio (SMR) data. This reduction of SMR data will be a result of successful patient transitions to hospice level of care at least 12 months prior to their passing or if their level of care is changed to inpatient hospice after they are admitted, the patients won’t be included as acute care mortality. However, with this initial small group of patients it was not possible to retrospectively calculate the impact on SMR or SAIL (Strategic Analytics for Improvement and Learning) indicators. The long-term expectation is to have a positive impact on those indicators represented by decreased inpatient mortality and improved SAIL.
Limitations
This study was a single-institution study, but every institution has its own internal culture. The team did not have a concurrent or historic control for comparison or use a questionnaire for patients and families rating their satisfaction.
Conclusion
This study provides multiple future directions of research as the authors now have baseline data about how the service is used. Future areas of interest would be to study the effectiveness of early palliative care interventions, such as a provider education series, implementation of consultation criteria, and prospective measurement of the impact of palliative care consultations on the SMR and SAIL indicators. This research could help identify which early interventions show the best efficacy, an area where research is notably lacking.25
1. El-Jawahri A, Greer JA, Temel JS. Does palliative care improve outcomes for patients with incurable illness? A review of the evidence. J Support Oncol. 2011;9(3):87-94.
2. Higginson IJ, Finlay I, Goodwin DM, et al. Do hospital-based palliative teams improve care for patients or families at the end of life? J Pain Symptom Manage. 2002;23(2):96-106.
3. Gade G, Venohr I, Conner D, et al. Impact of an inpatient palliative care team: a randomized control trial. J Palliat Med. 2008;11(2):180-190.
4. Benzar E, Hansen L, Kneitel AW, Fromme EK. Discharge planning for palliative care patients: a qualitative analysis. J Palliat Med. 2011;14(1):65-69.
5. Enguidanos S, Housen P, Penido M, Mejia B, Miller JA. Family members’ perceptions of inpatient palliative care consult services: a qualitative study. Palliat Med. 2014;28(1):42-48.
6. Granda-Cameron C, Viola SR, Lynch MP, Polomano RC. Measuring patient-oriented outcomes in palliative care: functionality and quality of life. Clin J Oncol Nurs. 2008;12(1):65-77.
7. Chand P, Gabriel T, Wallace CL, Nelson CM. Inpatient palliative care consultation: describing patient satisfaction. Perm J. 2013;17(1):53-55.
8. Tangeman JC, Rudra CB, Kerr CW, Grant PC. A hospice-hospital partnership: reducing hospitalization costs and 30-day readmissions among seriously ill adults. J Palliat Med. 2014;17(9):1005-1010.
9. Fromme EK, Bascom PB, Smith MD, et al. Survival, mortality, and location of death for patients seen by a hospital-based palliative care team. J Palliat Med. 2006;9(4):903-911.
10. Penrod JD, Deb P, Dellenbaugh C, et al. Hospital-based palliative care consultation: effects on hospital cost. J Palliat Med. 2010;13(8):973-979.
11. Ranganathan A, Dougherty M, Waite D, Casarett D. Can palliative home care reduce 30-day readmissions? Results of a propensity score matched cohort study. J Palliat Med. 2013;16(10):1290-1293.
12. Starks H, Wang S, Farber S, Owens DA, Curtis JR. Cost savings vary by length of stay for inpatients receiving palliative care consultation services. J Palliat Med. 2013;16(10):1215-1220.
13. Goldenheim A, Oates D, Parker V, Russell M, Winter M, Silliman RA. Rehospitalization of older adults discharged to home hospice care. J Palliat Med. 2014;17(7):841-844.
14. May P, Normand C, Morrison RS. Economic impact of hospital inpatient palliative care consultation: review of current evidence and directions for future research. J Palliat Med. 2014;17(9):1054-1063.
15. Granda-Cameron C, Behta M, Hovinga M, Rundio A, Mintzer D. Risk factors associated with unplanned hospital readmissions in adults with cancer. Oncol Nurs Forum. 2015;42(3):e257-e268.
16. Brody AA, Ciemins E, Newman J, Harrington C. The effects of an inpatient palliative care team on discharge disposition. J Palliat Med. 2010;13(5):541-548.
17. Penrod JD, Deb P, Luhrs C, et al. Cost and utilization outcomes of patients receiving hospital-based palliative care consultation. J Palliat Med. 2006;9(4):855-860.
18. Aldridge MD, Kelley AS. Appendix E, Epidemiology of serious illness and high utilization of health care. In: Institute of Medicine, Committee on Approaching Death: Addressing Key End of Life Issues. Dying in America: Improving Quality and Honoring Individual Preferences Near the End of Life. Washington, DC: National Academies Press; 2015.
19. Pantilat SZ, Kerr KM, Billings JA, Bruno KA, O’Riordan DL. Characteristics of palliative care consultation services in California hospitals. J Palliat Med. 2012;15(5):555-560.
20. Gonsalves WI, Tashi T, Krishnamurthy J, et al. Effect of palliative care services on the aggressiveness of end-of-life care in the Veterans Affairs cancer population. J Palliat Med. 2011;14(11):1231-1235.
21. Wendler D, Rid A. Systematic review: the effect on surrogates of making treatment decisions for others. Ann Intern Med. 2011;154(5):336-346.
22. American Bar Association Commission on Law and Aging. Myths and facts about health care advance directives. https://www.americanbar.org/content/dam/aba/publications/bifocal/BIFOCALSept-Oct2015.authcheckdam.pdf. Accessed July 10, 2018.
23. Tierney WM, Dexter PR, Gramelspacher GP, Perkins AJ, Zhou X-H, Wolinsky FD. The effect of discussions about advance directives on patients’ satisfaction with primary care. J Gen Intern Med. 2001;16(1):32-40.
24. Bailey FA, Williams BR, Woodby LL, et al. Intervention to improve care at life’s end in inpatient settings: the BEACON trial. J Gen Intern Med. 2014;29(6):836-843.
25. Dalgaard K, Bergenholtz H, Nielsen M, Timm H. Early integration of palliative care in hospitals: a systematic review on methods, barriers, and outcome. Palliat Support Care. 2014;12(6):495-513.
1. El-Jawahri A, Greer JA, Temel JS. Does palliative care improve outcomes for patients with incurable illness? A review of the evidence. J Support Oncol. 2011;9(3):87-94.
2. Higginson IJ, Finlay I, Goodwin DM, et al. Do hospital-based palliative teams improve care for patients or families at the end of life? J Pain Symptom Manage. 2002;23(2):96-106.
3. Gade G, Venohr I, Conner D, et al. Impact of an inpatient palliative care team: a randomized control trial. J Palliat Med. 2008;11(2):180-190.
4. Benzar E, Hansen L, Kneitel AW, Fromme EK. Discharge planning for palliative care patients: a qualitative analysis. J Palliat Med. 2011;14(1):65-69.
5. Enguidanos S, Housen P, Penido M, Mejia B, Miller JA. Family members’ perceptions of inpatient palliative care consult services: a qualitative study. Palliat Med. 2014;28(1):42-48.
6. Granda-Cameron C, Viola SR, Lynch MP, Polomano RC. Measuring patient-oriented outcomes in palliative care: functionality and quality of life. Clin J Oncol Nurs. 2008;12(1):65-77.
7. Chand P, Gabriel T, Wallace CL, Nelson CM. Inpatient palliative care consultation: describing patient satisfaction. Perm J. 2013;17(1):53-55.
8. Tangeman JC, Rudra CB, Kerr CW, Grant PC. A hospice-hospital partnership: reducing hospitalization costs and 30-day readmissions among seriously ill adults. J Palliat Med. 2014;17(9):1005-1010.
9. Fromme EK, Bascom PB, Smith MD, et al. Survival, mortality, and location of death for patients seen by a hospital-based palliative care team. J Palliat Med. 2006;9(4):903-911.
10. Penrod JD, Deb P, Dellenbaugh C, et al. Hospital-based palliative care consultation: effects on hospital cost. J Palliat Med. 2010;13(8):973-979.
11. Ranganathan A, Dougherty M, Waite D, Casarett D. Can palliative home care reduce 30-day readmissions? Results of a propensity score matched cohort study. J Palliat Med. 2013;16(10):1290-1293.
12. Starks H, Wang S, Farber S, Owens DA, Curtis JR. Cost savings vary by length of stay for inpatients receiving palliative care consultation services. J Palliat Med. 2013;16(10):1215-1220.
13. Goldenheim A, Oates D, Parker V, Russell M, Winter M, Silliman RA. Rehospitalization of older adults discharged to home hospice care. J Palliat Med. 2014;17(7):841-844.
14. May P, Normand C, Morrison RS. Economic impact of hospital inpatient palliative care consultation: review of current evidence and directions for future research. J Palliat Med. 2014;17(9):1054-1063.
15. Granda-Cameron C, Behta M, Hovinga M, Rundio A, Mintzer D. Risk factors associated with unplanned hospital readmissions in adults with cancer. Oncol Nurs Forum. 2015;42(3):e257-e268.
16. Brody AA, Ciemins E, Newman J, Harrington C. The effects of an inpatient palliative care team on discharge disposition. J Palliat Med. 2010;13(5):541-548.
17. Penrod JD, Deb P, Luhrs C, et al. Cost and utilization outcomes of patients receiving hospital-based palliative care consultation. J Palliat Med. 2006;9(4):855-860.
18. Aldridge MD, Kelley AS. Appendix E, Epidemiology of serious illness and high utilization of health care. In: Institute of Medicine, Committee on Approaching Death: Addressing Key End of Life Issues. Dying in America: Improving Quality and Honoring Individual Preferences Near the End of Life. Washington, DC: National Academies Press; 2015.
19. Pantilat SZ, Kerr KM, Billings JA, Bruno KA, O’Riordan DL. Characteristics of palliative care consultation services in California hospitals. J Palliat Med. 2012;15(5):555-560.
20. Gonsalves WI, Tashi T, Krishnamurthy J, et al. Effect of palliative care services on the aggressiveness of end-of-life care in the Veterans Affairs cancer population. J Palliat Med. 2011;14(11):1231-1235.
21. Wendler D, Rid A. Systematic review: the effect on surrogates of making treatment decisions for others. Ann Intern Med. 2011;154(5):336-346.
22. American Bar Association Commission on Law and Aging. Myths and facts about health care advance directives. https://www.americanbar.org/content/dam/aba/publications/bifocal/BIFOCALSept-Oct2015.authcheckdam.pdf. Accessed July 10, 2018.
23. Tierney WM, Dexter PR, Gramelspacher GP, Perkins AJ, Zhou X-H, Wolinsky FD. The effect of discussions about advance directives on patients’ satisfaction with primary care. J Gen Intern Med. 2001;16(1):32-40.
24. Bailey FA, Williams BR, Woodby LL, et al. Intervention to improve care at life’s end in inpatient settings: the BEACON trial. J Gen Intern Med. 2014;29(6):836-843.
25. Dalgaard K, Bergenholtz H, Nielsen M, Timm H. Early integration of palliative care in hospitals: a systematic review on methods, barriers, and outcome. Palliat Support Care. 2014;12(6):495-513.
CHMP reconsiders new indication for blinatumomab
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) said it will re-examine a recent opinion on blinatumomab (Blincyto).
In July, the CHMP recommended against approving blinatumomab to treat patients with B-cell precursor acute lymphoblastic leukemia (BCP-ALL) who have minimal residual disease (MRD).
However, the CHMP has agreed to re-examine its position and issue a final recommendation.
Blinatumomab is currently approved by the European Commission (EC) as monotherapy for adults with Philadelphia chromosome-negative, CD19-positive, relapsed or refractory BCP-ALL.
Blinatumomab is also approved as monotherapy for pediatric patients age 1 year or older who have relapsed/refractory, Philadelphia chromosome-negative, CD19-positive BCP-ALL and have received at least two prior therapies or relapsed after allogeneic hematopoietic stem cell transplant.
Amgen is seeking an extension of the marketing authorization for blinatumomab to include BCP-ALL patients with MRD.
The CHMP previously recommended against approving blinatumomab for these patients based on data from the BLAST study. Results from this phase 2 trial were published in Blood in April.
The CHMP noted that, although blinatumomab helped clear away residual cells in many patients in the BLAST trial, there is no strong evidence that this leads to improved survival.
Given the uncertainty, the CHMP was of the opinion that the benefits of blinatumomab do not outweigh its risks in MRD-positive BCP-ALL patients.
However, Amgen request a re-examination of the CHMP’s opinion, and the CHMP has complied.
The CHMP’s recommendations are reviewed by the EC, which has the authority to approve medicines for use in the European Union, Norway, Iceland, and Liechtenstein. The EC usually makes a decision within 67 days of CHMP recommendations.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) said it will re-examine a recent opinion on blinatumomab (Blincyto).
In July, the CHMP recommended against approving blinatumomab to treat patients with B-cell precursor acute lymphoblastic leukemia (BCP-ALL) who have minimal residual disease (MRD).
However, the CHMP has agreed to re-examine its position and issue a final recommendation.
Blinatumomab is currently approved by the European Commission (EC) as monotherapy for adults with Philadelphia chromosome-negative, CD19-positive, relapsed or refractory BCP-ALL.
Blinatumomab is also approved as monotherapy for pediatric patients age 1 year or older who have relapsed/refractory, Philadelphia chromosome-negative, CD19-positive BCP-ALL and have received at least two prior therapies or relapsed after allogeneic hematopoietic stem cell transplant.
Amgen is seeking an extension of the marketing authorization for blinatumomab to include BCP-ALL patients with MRD.
The CHMP previously recommended against approving blinatumomab for these patients based on data from the BLAST study. Results from this phase 2 trial were published in Blood in April.
The CHMP noted that, although blinatumomab helped clear away residual cells in many patients in the BLAST trial, there is no strong evidence that this leads to improved survival.
Given the uncertainty, the CHMP was of the opinion that the benefits of blinatumomab do not outweigh its risks in MRD-positive BCP-ALL patients.
However, Amgen request a re-examination of the CHMP’s opinion, and the CHMP has complied.
The CHMP’s recommendations are reviewed by the EC, which has the authority to approve medicines for use in the European Union, Norway, Iceland, and Liechtenstein. The EC usually makes a decision within 67 days of CHMP recommendations.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) said it will re-examine a recent opinion on blinatumomab (Blincyto).
In July, the CHMP recommended against approving blinatumomab to treat patients with B-cell precursor acute lymphoblastic leukemia (BCP-ALL) who have minimal residual disease (MRD).
However, the CHMP has agreed to re-examine its position and issue a final recommendation.
Blinatumomab is currently approved by the European Commission (EC) as monotherapy for adults with Philadelphia chromosome-negative, CD19-positive, relapsed or refractory BCP-ALL.
Blinatumomab is also approved as monotherapy for pediatric patients age 1 year or older who have relapsed/refractory, Philadelphia chromosome-negative, CD19-positive BCP-ALL and have received at least two prior therapies or relapsed after allogeneic hematopoietic stem cell transplant.
Amgen is seeking an extension of the marketing authorization for blinatumomab to include BCP-ALL patients with MRD.
The CHMP previously recommended against approving blinatumomab for these patients based on data from the BLAST study. Results from this phase 2 trial were published in Blood in April.
The CHMP noted that, although blinatumomab helped clear away residual cells in many patients in the BLAST trial, there is no strong evidence that this leads to improved survival.
Given the uncertainty, the CHMP was of the opinion that the benefits of blinatumomab do not outweigh its risks in MRD-positive BCP-ALL patients.
However, Amgen request a re-examination of the CHMP’s opinion, and the CHMP has complied.
The CHMP’s recommendations are reviewed by the EC, which has the authority to approve medicines for use in the European Union, Norway, Iceland, and Liechtenstein. The EC usually makes a decision within 67 days of CHMP recommendations.
CHMP backs proposed biosimilars of pegfilgrastim
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for three proposed biosimilars of pegfilgrastim—Ziextenzo, Pelmeg, and Fulphila.
If approved by the European Commission (EC), these products would be used for the same indication as the reference medicine, Neulasta (pegfilgrastim).
Neulasta has been EC-approved since 2002 to reduce the duration of neutropenia and the incidence of febrile neutropenia in adults who receive cytotoxic chemotherapy to treat malignancies except chronic myeloid leukemia and myelodysplastic syndromes.
According to the CHMP, data suggest that Ziextenzo, Pelmeg, and Fulphila all have quality, efficacy, and safety profiles comparable to Neulasta.
The EC is expected to make a decision on the approval of Ziextenzo, Pelmeg, and Fulphila within 67 days of the CHMP’s opinion.
The EC’s decision will apply to the European Union. Norway, Iceland, and Liechtenstein will make corresponding decisions based on the EC’s judgement.
Ziextenzo is being developed by Sandoz GmbH, Pelmeg is being developed by Cinfa Biotech S.L., and Fulphila is being developed by MYLAN S.A.S.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for three proposed biosimilars of pegfilgrastim—Ziextenzo, Pelmeg, and Fulphila.
If approved by the European Commission (EC), these products would be used for the same indication as the reference medicine, Neulasta (pegfilgrastim).
Neulasta has been EC-approved since 2002 to reduce the duration of neutropenia and the incidence of febrile neutropenia in adults who receive cytotoxic chemotherapy to treat malignancies except chronic myeloid leukemia and myelodysplastic syndromes.
According to the CHMP, data suggest that Ziextenzo, Pelmeg, and Fulphila all have quality, efficacy, and safety profiles comparable to Neulasta.
The EC is expected to make a decision on the approval of Ziextenzo, Pelmeg, and Fulphila within 67 days of the CHMP’s opinion.
The EC’s decision will apply to the European Union. Norway, Iceland, and Liechtenstein will make corresponding decisions based on the EC’s judgement.
Ziextenzo is being developed by Sandoz GmbH, Pelmeg is being developed by Cinfa Biotech S.L., and Fulphila is being developed by MYLAN S.A.S.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for three proposed biosimilars of pegfilgrastim—Ziextenzo, Pelmeg, and Fulphila.
If approved by the European Commission (EC), these products would be used for the same indication as the reference medicine, Neulasta (pegfilgrastim).
Neulasta has been EC-approved since 2002 to reduce the duration of neutropenia and the incidence of febrile neutropenia in adults who receive cytotoxic chemotherapy to treat malignancies except chronic myeloid leukemia and myelodysplastic syndromes.
According to the CHMP, data suggest that Ziextenzo, Pelmeg, and Fulphila all have quality, efficacy, and safety profiles comparable to Neulasta.
The EC is expected to make a decision on the approval of Ziextenzo, Pelmeg, and Fulphila within 67 days of the CHMP’s opinion.
The EC’s decision will apply to the European Union. Norway, Iceland, and Liechtenstein will make corresponding decisions based on the EC’s judgement.
Ziextenzo is being developed by Sandoz GmbH, Pelmeg is being developed by Cinfa Biotech S.L., and Fulphila is being developed by MYLAN S.A.S.
CHMP recommends factor VIII therapy for hemophilia A
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for damoctocog alfa pegol, a recombinant human factor VIII therapy.
Bayer is seeking European marketing authorization for damoctocog alfa pegol (formerly BAY94-9027) for the treatment and prophylaxis of bleeding in previously treated patients age 12 and older with hemophilia A.
The CHMP’s recommendation for damoctocog alfa pegol, which is approved in the U.S. under the name Jivi, will be reviewed by the European Commission (EC).
The EC typically makes a decision about marketing authorization within 67 days of the CHMP’s opinion. The EC’s decision will apply to the European Union, Norway, Iceland, and Liechtenstein.
The CHMP’s recommendation for damoctocog alfa pegol is supported by the phase 2/3 PROTECT VIII trial. Some results from this trial were published in the Journal of Thrombosis and Haemostasis in 2016. Additional results are available in the U.S. prescribing information for Jivi.
PROTECT VIII enrolled previously treated adults and adolescents (ages 12 to 65) with severe hemophilia A.
In part A, researchers evaluated different dosing regimens for damoctocog alfa pegol used as prophylaxis and on-demand treatment. An optional extension study was available to patients who completed part A.
In part B, researchers evaluated damoctocog alfa pegol for perioperative management.
Efficacy
In part A, there were 132 patients in the intent‐to‐treat population—112 in the prophylaxis group and 20 in the on-demand group.
Patients received damoctocog alfa pegol for 36 weeks. For the first 10 weeks, patients in the prophylaxis group received twice-weekly dosing at 25 IU/kg.
Patients with more than one bleed during this time went on to receive 30–40 IU/kg twice weekly. Patients with one or fewer bleeds were eligible for randomization to dosing every 5 days (45–60 IU/kg) or every 7 days (60 IU/kg).
The median annualized bleeding rate (ABR) was 4.1 for the patients who were treated twice weekly and were not eligible for randomization (n=13) and 1.9 for patients who were eligible for randomization but continued on twice-weekly treatment (n=11).
The median ABR was 1.9 for patients who were randomized to treatment every 5 days (n=43) and 0.96 for patients who completed prophylaxis with dosing every 7 days (32/43).
The median ABR for patients treated on demand was 24.1.
There were 388 treated bleeds in the on-demand group and 317 treated bleeds in the prophylaxis group. Overall, 73.3% of responses to treatment were considered “excellent” or “good,” 23.3% were considered “moderate,” and 3.3% were considered “poor.”
There were 17 patients who underwent 20 major surgeries in part B or the extension study and 10 patients who underwent minor surgeries in part A. Damoctocog alfa pegol provided “good” or “excellent” hemostatic control during all surgeries.
Safety
Safety data are available for 148 patients age 12 and older.
Adverse events in these patients included abdominal pain (3%), nausea (5%), vomiting (3%), injection site reactions (1%), pyrexia (5%), hypersensitivity (2%), dizziness (2%), headache (14%), insomnia (3%), cough (7%), erythema (1%), pruritus (1%), rash (2%), and flushing (1%).
A factor VIII inhibitor was reported in one adult patient, but repeat testing did not confirm the report.
One adult with asthma had a clinical hypersensitivity reaction and a transient increase of IgM anti-PEG antibody titer, which was negative upon retesting.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for damoctocog alfa pegol, a recombinant human factor VIII therapy.
Bayer is seeking European marketing authorization for damoctocog alfa pegol (formerly BAY94-9027) for the treatment and prophylaxis of bleeding in previously treated patients age 12 and older with hemophilia A.
The CHMP’s recommendation for damoctocog alfa pegol, which is approved in the U.S. under the name Jivi, will be reviewed by the European Commission (EC).
The EC typically makes a decision about marketing authorization within 67 days of the CHMP’s opinion. The EC’s decision will apply to the European Union, Norway, Iceland, and Liechtenstein.
The CHMP’s recommendation for damoctocog alfa pegol is supported by the phase 2/3 PROTECT VIII trial. Some results from this trial were published in the Journal of Thrombosis and Haemostasis in 2016. Additional results are available in the U.S. prescribing information for Jivi.
PROTECT VIII enrolled previously treated adults and adolescents (ages 12 to 65) with severe hemophilia A.
In part A, researchers evaluated different dosing regimens for damoctocog alfa pegol used as prophylaxis and on-demand treatment. An optional extension study was available to patients who completed part A.
In part B, researchers evaluated damoctocog alfa pegol for perioperative management.
Efficacy
In part A, there were 132 patients in the intent‐to‐treat population—112 in the prophylaxis group and 20 in the on-demand group.
Patients received damoctocog alfa pegol for 36 weeks. For the first 10 weeks, patients in the prophylaxis group received twice-weekly dosing at 25 IU/kg.
Patients with more than one bleed during this time went on to receive 30–40 IU/kg twice weekly. Patients with one or fewer bleeds were eligible for randomization to dosing every 5 days (45–60 IU/kg) or every 7 days (60 IU/kg).
The median annualized bleeding rate (ABR) was 4.1 for the patients who were treated twice weekly and were not eligible for randomization (n=13) and 1.9 for patients who were eligible for randomization but continued on twice-weekly treatment (n=11).
The median ABR was 1.9 for patients who were randomized to treatment every 5 days (n=43) and 0.96 for patients who completed prophylaxis with dosing every 7 days (32/43).
The median ABR for patients treated on demand was 24.1.
There were 388 treated bleeds in the on-demand group and 317 treated bleeds in the prophylaxis group. Overall, 73.3% of responses to treatment were considered “excellent” or “good,” 23.3% were considered “moderate,” and 3.3% were considered “poor.”
There were 17 patients who underwent 20 major surgeries in part B or the extension study and 10 patients who underwent minor surgeries in part A. Damoctocog alfa pegol provided “good” or “excellent” hemostatic control during all surgeries.
Safety
Safety data are available for 148 patients age 12 and older.
Adverse events in these patients included abdominal pain (3%), nausea (5%), vomiting (3%), injection site reactions (1%), pyrexia (5%), hypersensitivity (2%), dizziness (2%), headache (14%), insomnia (3%), cough (7%), erythema (1%), pruritus (1%), rash (2%), and flushing (1%).
A factor VIII inhibitor was reported in one adult patient, but repeat testing did not confirm the report.
One adult with asthma had a clinical hypersensitivity reaction and a transient increase of IgM anti-PEG antibody titer, which was negative upon retesting.
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for damoctocog alfa pegol, a recombinant human factor VIII therapy.
Bayer is seeking European marketing authorization for damoctocog alfa pegol (formerly BAY94-9027) for the treatment and prophylaxis of bleeding in previously treated patients age 12 and older with hemophilia A.
The CHMP’s recommendation for damoctocog alfa pegol, which is approved in the U.S. under the name Jivi, will be reviewed by the European Commission (EC).
The EC typically makes a decision about marketing authorization within 67 days of the CHMP’s opinion. The EC’s decision will apply to the European Union, Norway, Iceland, and Liechtenstein.
The CHMP’s recommendation for damoctocog alfa pegol is supported by the phase 2/3 PROTECT VIII trial. Some results from this trial were published in the Journal of Thrombosis and Haemostasis in 2016. Additional results are available in the U.S. prescribing information for Jivi.
PROTECT VIII enrolled previously treated adults and adolescents (ages 12 to 65) with severe hemophilia A.
In part A, researchers evaluated different dosing regimens for damoctocog alfa pegol used as prophylaxis and on-demand treatment. An optional extension study was available to patients who completed part A.
In part B, researchers evaluated damoctocog alfa pegol for perioperative management.
Efficacy
In part A, there were 132 patients in the intent‐to‐treat population—112 in the prophylaxis group and 20 in the on-demand group.
Patients received damoctocog alfa pegol for 36 weeks. For the first 10 weeks, patients in the prophylaxis group received twice-weekly dosing at 25 IU/kg.
Patients with more than one bleed during this time went on to receive 30–40 IU/kg twice weekly. Patients with one or fewer bleeds were eligible for randomization to dosing every 5 days (45–60 IU/kg) or every 7 days (60 IU/kg).
The median annualized bleeding rate (ABR) was 4.1 for the patients who were treated twice weekly and were not eligible for randomization (n=13) and 1.9 for patients who were eligible for randomization but continued on twice-weekly treatment (n=11).
The median ABR was 1.9 for patients who were randomized to treatment every 5 days (n=43) and 0.96 for patients who completed prophylaxis with dosing every 7 days (32/43).
The median ABR for patients treated on demand was 24.1.
There were 388 treated bleeds in the on-demand group and 317 treated bleeds in the prophylaxis group. Overall, 73.3% of responses to treatment were considered “excellent” or “good,” 23.3% were considered “moderate,” and 3.3% were considered “poor.”
There were 17 patients who underwent 20 major surgeries in part B or the extension study and 10 patients who underwent minor surgeries in part A. Damoctocog alfa pegol provided “good” or “excellent” hemostatic control during all surgeries.
Safety
Safety data are available for 148 patients age 12 and older.
Adverse events in these patients included abdominal pain (3%), nausea (5%), vomiting (3%), injection site reactions (1%), pyrexia (5%), hypersensitivity (2%), dizziness (2%), headache (14%), insomnia (3%), cough (7%), erythema (1%), pruritus (1%), rash (2%), and flushing (1%).
A factor VIII inhibitor was reported in one adult patient, but repeat testing did not confirm the report.
One adult with asthma had a clinical hypersensitivity reaction and a transient increase of IgM anti-PEG antibody titer, which was negative upon retesting.
New AAP policy statement addresses teen driver risks
The American Academy of Pediatric has issued a policy statement regarding teen drivers because, although becoming a driver is a rite of passage for many, there are unique risks associated with teen drivers – and important ways to address them.
“In 2015, among 15- to 20-year-old individuals, 1,886 young drivers died in MVCs [motor vehicle crashes], which is an increase of 9% from 2014,” the statement explains. “Another 195,000 young drivers were in MVCs, which is up 14% from 2014.”
Risk factors associated with crashes in teenage drivers include their inexperience, the presence of other teens in the car, high-speed and risky driving, distraction, lack of sleep, nighttime driving, and alcohol, marijuana, and medication use. Mobile phones and other electronic devices pose a major threat to driver safety because they contribute opportunities for three different kinds of distraction – visual, manual, and cognitive. Drug and alcohol use poses another threat; for example, tetrahydrocannabinol, found in marijuana, is associated with a 1.25 higher risk of crash, according Elizabeth M. Alderman, MD, and her associates on the Committee on Adolescence and the Council on Injury, Violence, and Poison Prevention who crafted the policy statement.
The policy statement also outlined several interventions to help mitigate these risks. Graduated driver licensing, which has been adopted in all 50 states, has helped reduce teen crashes by promoting skills development and reducing exposure to risky-driving situations. Parents can help model safer-driving practices and set expectations, as well as monitor and affect their teens’ driving behaviors during the supervised-driving phase. Driver education programs, on the other hand, counterintuitively have little effect on teen driving safety, which the authors suggested is because “the knowledge required to pass licensing exams is seldom related to an evidence-based understanding of the behaviors and skills associated with novice driver crash risk.” There also have been technological advances that can help improve driving safety among teenage drivers, such as automatic braking and lane-maintenance alerts.
“Policies, programs, and technologies exist to mitigate these risks but, in most cases, depend on active participation by the teenager and parents,” the authors concluded. “Pediatricians, communities, and governments need to take action to better educate teen drivers and their parents around these risks and strategies to reduce them.”
The full policy statement includes specific recommendations for anticipatory guidance, professional practice, community advocacy, and legislative advocacy. It also includes many resources and links to further information.
SOURCE: Alderman EM et al. Pediatrics. 2018 Oct;142(4):e20182163.
The American Academy of Pediatric has issued a policy statement regarding teen drivers because, although becoming a driver is a rite of passage for many, there are unique risks associated with teen drivers – and important ways to address them.
“In 2015, among 15- to 20-year-old individuals, 1,886 young drivers died in MVCs [motor vehicle crashes], which is an increase of 9% from 2014,” the statement explains. “Another 195,000 young drivers were in MVCs, which is up 14% from 2014.”
Risk factors associated with crashes in teenage drivers include their inexperience, the presence of other teens in the car, high-speed and risky driving, distraction, lack of sleep, nighttime driving, and alcohol, marijuana, and medication use. Mobile phones and other electronic devices pose a major threat to driver safety because they contribute opportunities for three different kinds of distraction – visual, manual, and cognitive. Drug and alcohol use poses another threat; for example, tetrahydrocannabinol, found in marijuana, is associated with a 1.25 higher risk of crash, according Elizabeth M. Alderman, MD, and her associates on the Committee on Adolescence and the Council on Injury, Violence, and Poison Prevention who crafted the policy statement.
The policy statement also outlined several interventions to help mitigate these risks. Graduated driver licensing, which has been adopted in all 50 states, has helped reduce teen crashes by promoting skills development and reducing exposure to risky-driving situations. Parents can help model safer-driving practices and set expectations, as well as monitor and affect their teens’ driving behaviors during the supervised-driving phase. Driver education programs, on the other hand, counterintuitively have little effect on teen driving safety, which the authors suggested is because “the knowledge required to pass licensing exams is seldom related to an evidence-based understanding of the behaviors and skills associated with novice driver crash risk.” There also have been technological advances that can help improve driving safety among teenage drivers, such as automatic braking and lane-maintenance alerts.
“Policies, programs, and technologies exist to mitigate these risks but, in most cases, depend on active participation by the teenager and parents,” the authors concluded. “Pediatricians, communities, and governments need to take action to better educate teen drivers and their parents around these risks and strategies to reduce them.”
The full policy statement includes specific recommendations for anticipatory guidance, professional practice, community advocacy, and legislative advocacy. It also includes many resources and links to further information.
SOURCE: Alderman EM et al. Pediatrics. 2018 Oct;142(4):e20182163.
The American Academy of Pediatric has issued a policy statement regarding teen drivers because, although becoming a driver is a rite of passage for many, there are unique risks associated with teen drivers – and important ways to address them.
“In 2015, among 15- to 20-year-old individuals, 1,886 young drivers died in MVCs [motor vehicle crashes], which is an increase of 9% from 2014,” the statement explains. “Another 195,000 young drivers were in MVCs, which is up 14% from 2014.”
Risk factors associated with crashes in teenage drivers include their inexperience, the presence of other teens in the car, high-speed and risky driving, distraction, lack of sleep, nighttime driving, and alcohol, marijuana, and medication use. Mobile phones and other electronic devices pose a major threat to driver safety because they contribute opportunities for three different kinds of distraction – visual, manual, and cognitive. Drug and alcohol use poses another threat; for example, tetrahydrocannabinol, found in marijuana, is associated with a 1.25 higher risk of crash, according Elizabeth M. Alderman, MD, and her associates on the Committee on Adolescence and the Council on Injury, Violence, and Poison Prevention who crafted the policy statement.
The policy statement also outlined several interventions to help mitigate these risks. Graduated driver licensing, which has been adopted in all 50 states, has helped reduce teen crashes by promoting skills development and reducing exposure to risky-driving situations. Parents can help model safer-driving practices and set expectations, as well as monitor and affect their teens’ driving behaviors during the supervised-driving phase. Driver education programs, on the other hand, counterintuitively have little effect on teen driving safety, which the authors suggested is because “the knowledge required to pass licensing exams is seldom related to an evidence-based understanding of the behaviors and skills associated with novice driver crash risk.” There also have been technological advances that can help improve driving safety among teenage drivers, such as automatic braking and lane-maintenance alerts.
“Policies, programs, and technologies exist to mitigate these risks but, in most cases, depend on active participation by the teenager and parents,” the authors concluded. “Pediatricians, communities, and governments need to take action to better educate teen drivers and their parents around these risks and strategies to reduce them.”
The full policy statement includes specific recommendations for anticipatory guidance, professional practice, community advocacy, and legislative advocacy. It also includes many resources and links to further information.
SOURCE: Alderman EM et al. Pediatrics. 2018 Oct;142(4):e20182163.
FROM PEDIATRICS
Adjuvant Pembrolizumab Improves Progression-Free Survival in Stage III Melanoma
Study Overview
Objective. To evaluate pembrolizumab as adjuvant therapy for patients with resected, high-risk stage III melanoma.
Design. International randomized phase 3 trial.
Setting and participants. This multicenter international trial enrolled patients who had histologically confirmed cutaneous melanoma with regional lymph node metastasis (stage IIIA, IIIB or IIIC with no in-transit metastases). Patients had to have undergone a complete regional lymphadenectomy within 13 weeks before the start of treatment. Exclusion criteria were: ECOG performance status score > 1, autoimmune disease, current steroid use, and prior systemic therapy for melanoma. All tumor samples from melanoma-positive lymph nodes were required to be sent to the central lab for evaluation of programmed death ligand 1 (PD-L1) expression; PD-L1 positivity was defined as a tumor proportion score (TPS) ≥ 1%.
Intervention. Patients were randomized in a 1:1 fashion and stratified according to stage and geographic region. Local pharmacies were aware of trial-group assignments. Patients received either an intravenous infusion of pembrolizumab 200 mg or placebo every 3 weeks for a total of 18 doses or until disease recurrence or unacceptable toxicity occurred. If recurrence was detected, patients were able to cross over.
Main outcome measures. The primary outcome was recurrence-free survival (RFS) in the intention-to-treat population and in the subgroup of PD-L1–positive patients. Secondary endpoints included distant metastasis–free survival, overall survival (OS), safety, and quality of life.
Results. A total of 1019 patients were recruited from 123 centers in 23 countries: 514 were assigned to the pembrolizumab group and 505 were assigned to the placebo group. In the pembrolizumab group, 70 patients (13.8%) discontinued treatment because of an adverse event; in 66 patients of these patients the event was deemed drug-related. In the placebo group, 11 (2.2%) patients discontinued treatment due to an adverse event. Discontinuation due to disease recurrence was seen in 109 (21%) patients in the pembrolizumab group and 179 (35.7%) patients in the placebo group. The median duration of follow up was 15 months. In the overall intention-to-treat population, the 12-month RFS rate was 75.4% in the pembrolizumab group versus 61% in the placebo group (P < 0.001). At 18 months the RFS rates were 71.4% and 53.2%, respectively. The 18-month incidence of distant metastasis at recurrence was lower in the pembrolizumab group (16.7% vs. 29.7%, hazard ratio [HR] 0.53; 95% confidence interval 0.37 to 0.76). In those who were PD-L1–positive (n = 853), the 12-month RFS rate was 77.1% in the pembrolizumab group versus 62.6% in the placebo group. PD-L1 status had no impact on pembrolizumab efficacy. The benefit of pembrolizumab was noted across all subgroups, and no difference was seen in patients with stage IIIA, IIIB or IIIC disease. The benefit of pembrolizumab was similar in those with macroscopic or microscopic nodal metastasis. BRAF status did not influence RFS between the pembrolizumab and placebo groups.
Adverse events of grade 3 or higher were seen in 14.7% and 3.4% of the pembrolizumab and placebo groups, respectively. Immune-related adverse events of any grade were noted in 37% of patients in the pembrolizumab group. There was 1 pembrolizumab-related death secondary to myositis. Grades 3 or 4 immune-related events in the pembrolizumab group occurred at a low rate, including colitis (2% and 0.2%), hypophysitis (0.6% and 0%), and type 1 diabetes mellitus (1% and 0%).
Conclusion. Adjuvant pembrolizumab for patients with high-risk stage III melanoma significantly improved RFS compared with placebo and should be considered as an option for adjuvant therapy in this patient population.
Commentary
Prior to the development of immune checkpoint inhibitors, high-dose interferon alfa was the sole option for adjuvant therapy in high-risk melanoma. Although adjuvant interferon alfa is associated with improvements in disease-free survival [1], it is also associated with significant toxicity, including myelosuppression, neurologic adverse effects, and hepatotoxicity. The development of checkpoint inhibition represents an important advancement in the management of patients with melanoma. In the previously reported EORTC 18071 trial, Eggermont and colleagues demonstrated that adjuvant therapy with the CTLA-4 antibody ipilimumab improved both RFS (41% vs. 30%) and OS (65% vs. 54%) at 5 years in patients with stage III melanoma [2]. In 2017, Weber and colleagues demonstrated superior RFS (70% vs. 60%) and a lower rate of grade 3 or 4 adverse events with adjuvant nivolumab compared to ipilimumab in the CheckMate-238 trial [3].
In the current article, Eggermont and colleagues present the results of the EORTC 1325/KEYNOTE-054 study comparing the use of the PD-1 antibody pembrolizumab to placebo in the adjuvant setting for stage III melanoma. This study demonstrated a 43% reduced risk of recurrence or death favoring the pembrolizumab group (HR 0.57; P < 0.001). The 12-month RFS was 75.4% in the pembrolizumab arm versus 61% in the placebo arm. Treatment-related adverse events of grade 3 or higher occurred more commonly in the pembrolizumab arm (14.7% vs. 3.4%), with approximately 7% of these patients experiencing a grade 3 or higher immune-related adverse event. The results of this study corroborate prior data on the efficacy of PD-1 inhibitors in melanoma. Also, the investigators assessed RFS based on patient’s PD-L1 status (positivity defined as TPS ≥ 1% ) as a co-primary endpoint, and found consistent efficacy regardless of PD-L1 expression, with a hazard ratio of 0.47 in the 116 patients who had no PD-L1 expression.
Although the results of this study demonstrate a significant increase in RFS associated with adjuvant pembrolizumab therapy, an OS benefit has not yet been demonstrated. As noted, the only adjuvant checkpoint inhibitor trial to demonstrate an OS advantage thus far is the EORTC 18071 study of ipilimumab. However, the toxicity profile of adjuvant ipilimumab makes it an unattractive option compared to the PD-1 inhibitors. Which of the PD-1 inhibitors should be the treatment of choice for adjuvant therapy remains unclear, although it is worth noting that only nivolumab was compared to the best alternate therapy, ipilimumab [3]. It is also important to note that EORTC 1325/KEYNOTE-054 included patients with stage IIIA disease (N1a disease with at least 1 micrometastasis > 1 mm) or stage IIIB or IIIC without in-transit metastases, while CheckMate-238 did not include stage IIIA patients. Thus, for stage IIIA patients pembrolizumab remains the only PD-1 inhibitor with randomized data demonstrating a benefit.
Applications for Clinical Practice
The results from the EORTC 1325/KEYNOTE-054 study demonstrate a 43% reduction in the risk of progression or death with the use of adjuvant pembrolizumab in patients with stage III melanoma. As of now, the only checkpoint inhibitor to demonstrate an improvement in OS is ipilimumab, and whether the RFS benefit of both pembrolizumab and nivolumab will translate into an OS benefit is yet to be demonstrated.
—Daniel Isaac, DO, MS
1. Kirkwood JM, Strawderman MH, Ernstoff MS, et al. Interferon alfa-2b adjuvant therapy of high-risk cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J Clin Oncol 1996;14:7–17.
2. Eggermont AM, Chiarion-Sileni V, Grob JJ, et al. Prolonged survival in stage III melanoma with ipilimumab adjuvant therapy. N Engl J Med 2016;375:1845–55.
3. Weber J, Mandala M, Del Vecchio M, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med 2017;377:1824–35.
Study Overview
Objective. To evaluate pembrolizumab as adjuvant therapy for patients with resected, high-risk stage III melanoma.
Design. International randomized phase 3 trial.
Setting and participants. This multicenter international trial enrolled patients who had histologically confirmed cutaneous melanoma with regional lymph node metastasis (stage IIIA, IIIB or IIIC with no in-transit metastases). Patients had to have undergone a complete regional lymphadenectomy within 13 weeks before the start of treatment. Exclusion criteria were: ECOG performance status score > 1, autoimmune disease, current steroid use, and prior systemic therapy for melanoma. All tumor samples from melanoma-positive lymph nodes were required to be sent to the central lab for evaluation of programmed death ligand 1 (PD-L1) expression; PD-L1 positivity was defined as a tumor proportion score (TPS) ≥ 1%.
Intervention. Patients were randomized in a 1:1 fashion and stratified according to stage and geographic region. Local pharmacies were aware of trial-group assignments. Patients received either an intravenous infusion of pembrolizumab 200 mg or placebo every 3 weeks for a total of 18 doses or until disease recurrence or unacceptable toxicity occurred. If recurrence was detected, patients were able to cross over.
Main outcome measures. The primary outcome was recurrence-free survival (RFS) in the intention-to-treat population and in the subgroup of PD-L1–positive patients. Secondary endpoints included distant metastasis–free survival, overall survival (OS), safety, and quality of life.
Results. A total of 1019 patients were recruited from 123 centers in 23 countries: 514 were assigned to the pembrolizumab group and 505 were assigned to the placebo group. In the pembrolizumab group, 70 patients (13.8%) discontinued treatment because of an adverse event; in 66 patients of these patients the event was deemed drug-related. In the placebo group, 11 (2.2%) patients discontinued treatment due to an adverse event. Discontinuation due to disease recurrence was seen in 109 (21%) patients in the pembrolizumab group and 179 (35.7%) patients in the placebo group. The median duration of follow up was 15 months. In the overall intention-to-treat population, the 12-month RFS rate was 75.4% in the pembrolizumab group versus 61% in the placebo group (P < 0.001). At 18 months the RFS rates were 71.4% and 53.2%, respectively. The 18-month incidence of distant metastasis at recurrence was lower in the pembrolizumab group (16.7% vs. 29.7%, hazard ratio [HR] 0.53; 95% confidence interval 0.37 to 0.76). In those who were PD-L1–positive (n = 853), the 12-month RFS rate was 77.1% in the pembrolizumab group versus 62.6% in the placebo group. PD-L1 status had no impact on pembrolizumab efficacy. The benefit of pembrolizumab was noted across all subgroups, and no difference was seen in patients with stage IIIA, IIIB or IIIC disease. The benefit of pembrolizumab was similar in those with macroscopic or microscopic nodal metastasis. BRAF status did not influence RFS between the pembrolizumab and placebo groups.
Adverse events of grade 3 or higher were seen in 14.7% and 3.4% of the pembrolizumab and placebo groups, respectively. Immune-related adverse events of any grade were noted in 37% of patients in the pembrolizumab group. There was 1 pembrolizumab-related death secondary to myositis. Grades 3 or 4 immune-related events in the pembrolizumab group occurred at a low rate, including colitis (2% and 0.2%), hypophysitis (0.6% and 0%), and type 1 diabetes mellitus (1% and 0%).
Conclusion. Adjuvant pembrolizumab for patients with high-risk stage III melanoma significantly improved RFS compared with placebo and should be considered as an option for adjuvant therapy in this patient population.
Commentary
Prior to the development of immune checkpoint inhibitors, high-dose interferon alfa was the sole option for adjuvant therapy in high-risk melanoma. Although adjuvant interferon alfa is associated with improvements in disease-free survival [1], it is also associated with significant toxicity, including myelosuppression, neurologic adverse effects, and hepatotoxicity. The development of checkpoint inhibition represents an important advancement in the management of patients with melanoma. In the previously reported EORTC 18071 trial, Eggermont and colleagues demonstrated that adjuvant therapy with the CTLA-4 antibody ipilimumab improved both RFS (41% vs. 30%) and OS (65% vs. 54%) at 5 years in patients with stage III melanoma [2]. In 2017, Weber and colleagues demonstrated superior RFS (70% vs. 60%) and a lower rate of grade 3 or 4 adverse events with adjuvant nivolumab compared to ipilimumab in the CheckMate-238 trial [3].
In the current article, Eggermont and colleagues present the results of the EORTC 1325/KEYNOTE-054 study comparing the use of the PD-1 antibody pembrolizumab to placebo in the adjuvant setting for stage III melanoma. This study demonstrated a 43% reduced risk of recurrence or death favoring the pembrolizumab group (HR 0.57; P < 0.001). The 12-month RFS was 75.4% in the pembrolizumab arm versus 61% in the placebo arm. Treatment-related adverse events of grade 3 or higher occurred more commonly in the pembrolizumab arm (14.7% vs. 3.4%), with approximately 7% of these patients experiencing a grade 3 or higher immune-related adverse event. The results of this study corroborate prior data on the efficacy of PD-1 inhibitors in melanoma. Also, the investigators assessed RFS based on patient’s PD-L1 status (positivity defined as TPS ≥ 1% ) as a co-primary endpoint, and found consistent efficacy regardless of PD-L1 expression, with a hazard ratio of 0.47 in the 116 patients who had no PD-L1 expression.
Although the results of this study demonstrate a significant increase in RFS associated with adjuvant pembrolizumab therapy, an OS benefit has not yet been demonstrated. As noted, the only adjuvant checkpoint inhibitor trial to demonstrate an OS advantage thus far is the EORTC 18071 study of ipilimumab. However, the toxicity profile of adjuvant ipilimumab makes it an unattractive option compared to the PD-1 inhibitors. Which of the PD-1 inhibitors should be the treatment of choice for adjuvant therapy remains unclear, although it is worth noting that only nivolumab was compared to the best alternate therapy, ipilimumab [3]. It is also important to note that EORTC 1325/KEYNOTE-054 included patients with stage IIIA disease (N1a disease with at least 1 micrometastasis > 1 mm) or stage IIIB or IIIC without in-transit metastases, while CheckMate-238 did not include stage IIIA patients. Thus, for stage IIIA patients pembrolizumab remains the only PD-1 inhibitor with randomized data demonstrating a benefit.
Applications for Clinical Practice
The results from the EORTC 1325/KEYNOTE-054 study demonstrate a 43% reduction in the risk of progression or death with the use of adjuvant pembrolizumab in patients with stage III melanoma. As of now, the only checkpoint inhibitor to demonstrate an improvement in OS is ipilimumab, and whether the RFS benefit of both pembrolizumab and nivolumab will translate into an OS benefit is yet to be demonstrated.
—Daniel Isaac, DO, MS
Study Overview
Objective. To evaluate pembrolizumab as adjuvant therapy for patients with resected, high-risk stage III melanoma.
Design. International randomized phase 3 trial.
Setting and participants. This multicenter international trial enrolled patients who had histologically confirmed cutaneous melanoma with regional lymph node metastasis (stage IIIA, IIIB or IIIC with no in-transit metastases). Patients had to have undergone a complete regional lymphadenectomy within 13 weeks before the start of treatment. Exclusion criteria were: ECOG performance status score > 1, autoimmune disease, current steroid use, and prior systemic therapy for melanoma. All tumor samples from melanoma-positive lymph nodes were required to be sent to the central lab for evaluation of programmed death ligand 1 (PD-L1) expression; PD-L1 positivity was defined as a tumor proportion score (TPS) ≥ 1%.
Intervention. Patients were randomized in a 1:1 fashion and stratified according to stage and geographic region. Local pharmacies were aware of trial-group assignments. Patients received either an intravenous infusion of pembrolizumab 200 mg or placebo every 3 weeks for a total of 18 doses or until disease recurrence or unacceptable toxicity occurred. If recurrence was detected, patients were able to cross over.
Main outcome measures. The primary outcome was recurrence-free survival (RFS) in the intention-to-treat population and in the subgroup of PD-L1–positive patients. Secondary endpoints included distant metastasis–free survival, overall survival (OS), safety, and quality of life.
Results. A total of 1019 patients were recruited from 123 centers in 23 countries: 514 were assigned to the pembrolizumab group and 505 were assigned to the placebo group. In the pembrolizumab group, 70 patients (13.8%) discontinued treatment because of an adverse event; in 66 patients of these patients the event was deemed drug-related. In the placebo group, 11 (2.2%) patients discontinued treatment due to an adverse event. Discontinuation due to disease recurrence was seen in 109 (21%) patients in the pembrolizumab group and 179 (35.7%) patients in the placebo group. The median duration of follow up was 15 months. In the overall intention-to-treat population, the 12-month RFS rate was 75.4% in the pembrolizumab group versus 61% in the placebo group (P < 0.001). At 18 months the RFS rates were 71.4% and 53.2%, respectively. The 18-month incidence of distant metastasis at recurrence was lower in the pembrolizumab group (16.7% vs. 29.7%, hazard ratio [HR] 0.53; 95% confidence interval 0.37 to 0.76). In those who were PD-L1–positive (n = 853), the 12-month RFS rate was 77.1% in the pembrolizumab group versus 62.6% in the placebo group. PD-L1 status had no impact on pembrolizumab efficacy. The benefit of pembrolizumab was noted across all subgroups, and no difference was seen in patients with stage IIIA, IIIB or IIIC disease. The benefit of pembrolizumab was similar in those with macroscopic or microscopic nodal metastasis. BRAF status did not influence RFS between the pembrolizumab and placebo groups.
Adverse events of grade 3 or higher were seen in 14.7% and 3.4% of the pembrolizumab and placebo groups, respectively. Immune-related adverse events of any grade were noted in 37% of patients in the pembrolizumab group. There was 1 pembrolizumab-related death secondary to myositis. Grades 3 or 4 immune-related events in the pembrolizumab group occurred at a low rate, including colitis (2% and 0.2%), hypophysitis (0.6% and 0%), and type 1 diabetes mellitus (1% and 0%).
Conclusion. Adjuvant pembrolizumab for patients with high-risk stage III melanoma significantly improved RFS compared with placebo and should be considered as an option for adjuvant therapy in this patient population.
Commentary
Prior to the development of immune checkpoint inhibitors, high-dose interferon alfa was the sole option for adjuvant therapy in high-risk melanoma. Although adjuvant interferon alfa is associated with improvements in disease-free survival [1], it is also associated with significant toxicity, including myelosuppression, neurologic adverse effects, and hepatotoxicity. The development of checkpoint inhibition represents an important advancement in the management of patients with melanoma. In the previously reported EORTC 18071 trial, Eggermont and colleagues demonstrated that adjuvant therapy with the CTLA-4 antibody ipilimumab improved both RFS (41% vs. 30%) and OS (65% vs. 54%) at 5 years in patients with stage III melanoma [2]. In 2017, Weber and colleagues demonstrated superior RFS (70% vs. 60%) and a lower rate of grade 3 or 4 adverse events with adjuvant nivolumab compared to ipilimumab in the CheckMate-238 trial [3].
In the current article, Eggermont and colleagues present the results of the EORTC 1325/KEYNOTE-054 study comparing the use of the PD-1 antibody pembrolizumab to placebo in the adjuvant setting for stage III melanoma. This study demonstrated a 43% reduced risk of recurrence or death favoring the pembrolizumab group (HR 0.57; P < 0.001). The 12-month RFS was 75.4% in the pembrolizumab arm versus 61% in the placebo arm. Treatment-related adverse events of grade 3 or higher occurred more commonly in the pembrolizumab arm (14.7% vs. 3.4%), with approximately 7% of these patients experiencing a grade 3 or higher immune-related adverse event. The results of this study corroborate prior data on the efficacy of PD-1 inhibitors in melanoma. Also, the investigators assessed RFS based on patient’s PD-L1 status (positivity defined as TPS ≥ 1% ) as a co-primary endpoint, and found consistent efficacy regardless of PD-L1 expression, with a hazard ratio of 0.47 in the 116 patients who had no PD-L1 expression.
Although the results of this study demonstrate a significant increase in RFS associated with adjuvant pembrolizumab therapy, an OS benefit has not yet been demonstrated. As noted, the only adjuvant checkpoint inhibitor trial to demonstrate an OS advantage thus far is the EORTC 18071 study of ipilimumab. However, the toxicity profile of adjuvant ipilimumab makes it an unattractive option compared to the PD-1 inhibitors. Which of the PD-1 inhibitors should be the treatment of choice for adjuvant therapy remains unclear, although it is worth noting that only nivolumab was compared to the best alternate therapy, ipilimumab [3]. It is also important to note that EORTC 1325/KEYNOTE-054 included patients with stage IIIA disease (N1a disease with at least 1 micrometastasis > 1 mm) or stage IIIB or IIIC without in-transit metastases, while CheckMate-238 did not include stage IIIA patients. Thus, for stage IIIA patients pembrolizumab remains the only PD-1 inhibitor with randomized data demonstrating a benefit.
Applications for Clinical Practice
The results from the EORTC 1325/KEYNOTE-054 study demonstrate a 43% reduction in the risk of progression or death with the use of adjuvant pembrolizumab in patients with stage III melanoma. As of now, the only checkpoint inhibitor to demonstrate an improvement in OS is ipilimumab, and whether the RFS benefit of both pembrolizumab and nivolumab will translate into an OS benefit is yet to be demonstrated.
—Daniel Isaac, DO, MS
1. Kirkwood JM, Strawderman MH, Ernstoff MS, et al. Interferon alfa-2b adjuvant therapy of high-risk cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J Clin Oncol 1996;14:7–17.
2. Eggermont AM, Chiarion-Sileni V, Grob JJ, et al. Prolonged survival in stage III melanoma with ipilimumab adjuvant therapy. N Engl J Med 2016;375:1845–55.
3. Weber J, Mandala M, Del Vecchio M, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med 2017;377:1824–35.
1. Kirkwood JM, Strawderman MH, Ernstoff MS, et al. Interferon alfa-2b adjuvant therapy of high-risk cutaneous melanoma: the Eastern Cooperative Oncology Group Trial EST 1684. J Clin Oncol 1996;14:7–17.
2. Eggermont AM, Chiarion-Sileni V, Grob JJ, et al. Prolonged survival in stage III melanoma with ipilimumab adjuvant therapy. N Engl J Med 2016;375:1845–55.
3. Weber J, Mandala M, Del Vecchio M, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med 2017;377:1824–35.
Physician impairment: A need for prevention
Psychiatry is a field of passion. The reward of experiencing growth and change alongside our patients is what bolsters us through years of difficult training, overnight shifts, endless paperwork, regulatory mandates, and frequent worry about our patients. As physicians, we don’t live for weekends as many other professionals do. To the contrary, we spend them on call, moonlighting, laboring over journal articles, and perfecting lectures.
That passion is what makes us trusted clinicians and experts in our field. It can also make it difficult for us to disconnect from our work, frequently leading to burnout. Physician self-care, support, and professional development are critical topics that modern-day medicine minimizes at the peril of physicians and public health.1
Psychiatry lends itself to a deep and intimate understanding of another human being. The therapist delves into the lives of his or her patients, hears their stories, and holds their secrets. In some cases, we might be the only ones who truly see patients for who they uniquely are, and come to understand them on a deeper level than their closest family and friends. This can be both thrilling and intense. As we delve into the psyche of another individual, contemplate which interpretation we should share, and resonate with our patients, it is easy to become bogged down with our own countertransference, sentiment, and worry, and to become consumed by our work. A professional hazard, some might quip.
Therefore, personal restoration—a tool that keeps our clinical skills sharp—is vitally important to caring for oneself and one’s patient. Surprisingly, this can be neglected until we begin to experience burnout, which over time could transform into impairment, thus endangering ourselves, our patients, and our profession.
Over the past decade, physician impairment has been exhaustively described, researched, and addressed. However, most analyses have focused on identifying impairment, and offering guidance on how to properly report it. How do we shift from managing the crisis to preventing it? To answer this question, this article:
- reviews the dilemma of physician impairment
- explores the duty we have to patients, ourselves, and the profession
- discusses shifting the focus on impairment to prevention through well-being.
Continue to: Dilemma
Dilemma: Vulnerability to impairment
The cornerstone for well-being is a balanced life. No matter how much one loves his or her work, there must be balance between work, relationships, and hobbies. Without that equilibrium, everyone is put at risk.2
Just as our patients, we are not immune to mental illness, cognitive decline, or substance abuse.3 We might even be more susceptible. For many physicians, their identity is intimately tied to their work.4 Dr. Robin Weiss captured that intimate relationship5:
“… [A] therapist may spend hundreds of hours, perhaps more than a thousand, hearing about a patient’s most exalted aspirations and most murderous, hateful fantasies. During this time, the patient may endure excruciating losses, unbearable shame, bitter sadness and great triumphs. You may accompany patients through torturous adolescence into adulthood. Or you may meet them in middle age and be with them as they age and eventually die. You collaborate in a deep process of discovery. Few encounters are this deeply honest, and therefore intimate.”
Given the stories we hear and the resulting intimacy and countertransference that inevitably arise, psychiatrists are even more prone to burnout than other physicians.6 Physician impairment is a public health issue that affects not just physicians but also their families, colleagues, and patients.
“Impairment” for the purpose of this article means a physical, mental, or substance-related disorder that interferes with a physician’s ability to undertake professional activities competently and safely.7 Predisposing factors for physician impairment include an obsessive-compulsive personality type, a family history of mental illness, sensation-seeking behavior, denial of personal problems, perfectionism, and idealism.8,9 Also, work stress becomes a significant factor in already vulnerable physicians, leading to a greater risk for mental illness.10
Continue to: Some warning signs of impairment include...
Some warning signs of impairment include a lack of personal hygiene, emotional lability, sleep deprivation, inattention to our pages or phone calls, and increased professional errors.11 When it comes to addressing such impairment, previous research and literature has focused on how to monitor ourselves and our colleagues; anything less would put the reputation and integrity of the medical profession at risk.3 This has led to a culture of doing nothing but work until things go too far, and then reporting the problems. But what about intervening before things get too far?
Duty: To ourselves, our colleagues, and our patients
There has been much discussion on how to report impaired colleagues, but little on how to help and support ourselves and our colleagues before things escalate into serious problems. And this lack of discussion is at the detriment of individual practitioners, their families, and patients. Physicians-in-training, including psychiatric residents, are at particularly high risk for developing stress-related problems, depression, and substance misuse.12 Occupational demands, self-criticism, and denial of one’s distress are common among physicians, as is self-treatment with drugs and alcohol.13
We all know by now that doctors and physician health programs (PHPs) have a duty to report impaired colleagues who continue to practice despite reasonable offers of assistance. There are an abundance of PHPs that are in place to assist with such situations. The American Medical Association’s official position on reporting impairment is outlined in Policy H-275.952.7 There also is the Federation of State Medical Boards. Its policy states that PHPs have “a primary commitment to [help] state medical boards … protect the public … [These] programs [should] demonstrate an ongoing track of record of ensuring safety to the public and reveal deficiencies if they occur.”14
Legal and ethical issues, however, complicate interventions for colleagues who need assistance.15 Despite the existence of PHPs, it would be much easier—not to mention helpful—to help a colleague by carrying out early interventions.
Discussion: Prevention as a solution
More emphasis should be placed on prevention. That’s where self-care and well-being come into play. Awareness of and sensitivity to physician vulnerability, early detection, and prevention of impairment are important.
Continue to: There has been a paradigm shift in focus...
There has been a paradigm shift in focus across medical boards, professional societies, and medical colleges. They are recognizing that personal well-being can help prevent burnout and, in turn, change the landscape of medicine from endless work to balanced lives that yield more satisfying and joyful work. It is becoming an accepted fact in medicine that well-being is just as important as integrity, professionalism, and patient safety. For example, the American Academy of Medical Colleges (AAMC) issued a statement emphasizing the importance of clinician well-being and dedicated its June 2016 Leadership Forum to a range of topics addressing depression, resilience, burnout, and suicide in academic medicine.16
Anita Everett, MD, put the spotlight on physician well-being during her term as American Psychiatric Association President (2017 to 2018). She formed a specific workgroup on Physician Wellness and Burnout where there is a community focus on prevention and self-care.17 A strong sense of community and purpose is almost always part of the prescription for promoting greater well-being.2
The importance of this issue is also trickling down from policymakers into hospitals and community health centers. Consider an initiative at Minneapolis’s Hennepin County Medical Center. Leaders there created a “reset room” for physicians to quietly decompress. The room is complete with LED lights, flameless candles, a sound machine, comfortable chairs, several plants, and an “in use” sign on the door.18 Other personal strategies to help prevent burnout include making environmental changes, encouraging hobbies, and streamlining burdensome tasks such as paperwork and electronic medical record systems.
As physician health and well-being are finally emerging as a “hot topic,”2 educational and treatment resources are increasingly available for any of us to explore. Consider a simple Google search to look into your State’s PHPs, and get involved in your professional societies to make change.
The culture is starting to shift, and leading by example will be a key to propelling further progress in this area. Model our own self-care for colleagues and patients alike. As Mark Twain said, we might love our work, but we must remember that being solely defined by work comes to the detriment of our health. Maintaining balance is what will allow us to sustain long careers ahead doing what we love.
1. Mahoney, D, Freedy J, Brock C. Improving physician well-being. JAMA Intern Med. 2015;175(4):648-649.
2. Yellowlees P. Addressing physician health and well-being is patient safety issue. Psychiatric News. 2018;53(12):20-21.
3. Mossman D, Farrell HM. Physician impairment: when should you report? Current Psychiatry. 2011;10(9):67-71.
4. Lindeman S, Henriksson M, Isometsä E, et al. Treatment of mental disorders in seven physicians committing suicide. Crisis. 1999;20(2):86-89.
5. Weiss R. How therapists mourn. New York Times. July 4, 2015:SR2.
6. Kumar S. Burnout in psychiatrists. World Psychiatry. 2007;6(3):186-189.
7. American Medical Association. Report 2 of the Council on Science and Public Health (A-11). Physician health programs (Reference Committee D). https://www.ama-assn.org/sites/default/files/media-browser/public/about-ama/councils/Council%20Reports/council-on-science-public-health/a11-csaph-physician-health-programs.pdf. Accessed August 6, 2018.
8. Boisaubin EV, Levine RE. Identifying and assisting the impaired physician. Am J Med Sci. 2001;322(1):31-36.
9. Bissel L, Jones RW. The alcoholic physician: a survey. Am J Psychiatry. 1976;133(10):1142-1146.
10. Vaillant GE, Sobowale NC, McArthur C. Some psychologic vulnerabilities of physicians. N Engl J Med. 1972;287(8):372-375.
11. McGovern MP, Agnes DH, Leon S. Characteristics of physicians presenting for assessment at a behavioral health center. J Addict Dis. 2000;19(2):59-73.
12. Broquet KE, Rockey PH. Teaching residents and program directors about physician impairment. Acad Psychiatry. 2004;28(3):221-225.
13. Meier DE, Back AL, Morrison RS. The inner life of physicians and care of the seriously ill. JAMA. 2001;286(23):3007-3014.
14. Federation of State Medical Boards of the United States. Policy on physician impairment. http://www.csam-asam.org/pdf/misc/FSMB2011.pdf. Published 2011. Accessed July 15, 2018.
15. Bright RP, Krahn L. Impaired physicians: how to recognize, when to report, and where to refer. Current Psychiatry. 2010;9(6):11-20.
16. Academy of American Colleges. Well-being in academic medicine. https://www.aamc.org/initiatives/462280/well-being-academic-medicine.html. Updated July 9, 2018. Accessed July 17, 2018.
17. American Psychiatric Association. Well-being and burnout. https://www.psychiatry.org/psychiatrists/practice/well-being-and-burnout. Updated February 22, 2018. Accessed July 17, 2018.
18. Parks T. Physicians take to “reset room” to battle burnout. AMA Wire. https://wire.ama-assn.org/practice-management/physicians-take-reset-room-battle-burnout. Published June 8, 2016. Accessed July 18, 2018.
Psychiatry is a field of passion. The reward of experiencing growth and change alongside our patients is what bolsters us through years of difficult training, overnight shifts, endless paperwork, regulatory mandates, and frequent worry about our patients. As physicians, we don’t live for weekends as many other professionals do. To the contrary, we spend them on call, moonlighting, laboring over journal articles, and perfecting lectures.
That passion is what makes us trusted clinicians and experts in our field. It can also make it difficult for us to disconnect from our work, frequently leading to burnout. Physician self-care, support, and professional development are critical topics that modern-day medicine minimizes at the peril of physicians and public health.1
Psychiatry lends itself to a deep and intimate understanding of another human being. The therapist delves into the lives of his or her patients, hears their stories, and holds their secrets. In some cases, we might be the only ones who truly see patients for who they uniquely are, and come to understand them on a deeper level than their closest family and friends. This can be both thrilling and intense. As we delve into the psyche of another individual, contemplate which interpretation we should share, and resonate with our patients, it is easy to become bogged down with our own countertransference, sentiment, and worry, and to become consumed by our work. A professional hazard, some might quip.
Therefore, personal restoration—a tool that keeps our clinical skills sharp—is vitally important to caring for oneself and one’s patient. Surprisingly, this can be neglected until we begin to experience burnout, which over time could transform into impairment, thus endangering ourselves, our patients, and our profession.
Over the past decade, physician impairment has been exhaustively described, researched, and addressed. However, most analyses have focused on identifying impairment, and offering guidance on how to properly report it. How do we shift from managing the crisis to preventing it? To answer this question, this article:
- reviews the dilemma of physician impairment
- explores the duty we have to patients, ourselves, and the profession
- discusses shifting the focus on impairment to prevention through well-being.
Continue to: Dilemma
Dilemma: Vulnerability to impairment
The cornerstone for well-being is a balanced life. No matter how much one loves his or her work, there must be balance between work, relationships, and hobbies. Without that equilibrium, everyone is put at risk.2
Just as our patients, we are not immune to mental illness, cognitive decline, or substance abuse.3 We might even be more susceptible. For many physicians, their identity is intimately tied to their work.4 Dr. Robin Weiss captured that intimate relationship5:
“… [A] therapist may spend hundreds of hours, perhaps more than a thousand, hearing about a patient’s most exalted aspirations and most murderous, hateful fantasies. During this time, the patient may endure excruciating losses, unbearable shame, bitter sadness and great triumphs. You may accompany patients through torturous adolescence into adulthood. Or you may meet them in middle age and be with them as they age and eventually die. You collaborate in a deep process of discovery. Few encounters are this deeply honest, and therefore intimate.”
Given the stories we hear and the resulting intimacy and countertransference that inevitably arise, psychiatrists are even more prone to burnout than other physicians.6 Physician impairment is a public health issue that affects not just physicians but also their families, colleagues, and patients.
“Impairment” for the purpose of this article means a physical, mental, or substance-related disorder that interferes with a physician’s ability to undertake professional activities competently and safely.7 Predisposing factors for physician impairment include an obsessive-compulsive personality type, a family history of mental illness, sensation-seeking behavior, denial of personal problems, perfectionism, and idealism.8,9 Also, work stress becomes a significant factor in already vulnerable physicians, leading to a greater risk for mental illness.10
Continue to: Some warning signs of impairment include...
Some warning signs of impairment include a lack of personal hygiene, emotional lability, sleep deprivation, inattention to our pages or phone calls, and increased professional errors.11 When it comes to addressing such impairment, previous research and literature has focused on how to monitor ourselves and our colleagues; anything less would put the reputation and integrity of the medical profession at risk.3 This has led to a culture of doing nothing but work until things go too far, and then reporting the problems. But what about intervening before things get too far?
Duty: To ourselves, our colleagues, and our patients
There has been much discussion on how to report impaired colleagues, but little on how to help and support ourselves and our colleagues before things escalate into serious problems. And this lack of discussion is at the detriment of individual practitioners, their families, and patients. Physicians-in-training, including psychiatric residents, are at particularly high risk for developing stress-related problems, depression, and substance misuse.12 Occupational demands, self-criticism, and denial of one’s distress are common among physicians, as is self-treatment with drugs and alcohol.13
We all know by now that doctors and physician health programs (PHPs) have a duty to report impaired colleagues who continue to practice despite reasonable offers of assistance. There are an abundance of PHPs that are in place to assist with such situations. The American Medical Association’s official position on reporting impairment is outlined in Policy H-275.952.7 There also is the Federation of State Medical Boards. Its policy states that PHPs have “a primary commitment to [help] state medical boards … protect the public … [These] programs [should] demonstrate an ongoing track of record of ensuring safety to the public and reveal deficiencies if they occur.”14
Legal and ethical issues, however, complicate interventions for colleagues who need assistance.15 Despite the existence of PHPs, it would be much easier—not to mention helpful—to help a colleague by carrying out early interventions.
Discussion: Prevention as a solution
More emphasis should be placed on prevention. That’s where self-care and well-being come into play. Awareness of and sensitivity to physician vulnerability, early detection, and prevention of impairment are important.
Continue to: There has been a paradigm shift in focus...
There has been a paradigm shift in focus across medical boards, professional societies, and medical colleges. They are recognizing that personal well-being can help prevent burnout and, in turn, change the landscape of medicine from endless work to balanced lives that yield more satisfying and joyful work. It is becoming an accepted fact in medicine that well-being is just as important as integrity, professionalism, and patient safety. For example, the American Academy of Medical Colleges (AAMC) issued a statement emphasizing the importance of clinician well-being and dedicated its June 2016 Leadership Forum to a range of topics addressing depression, resilience, burnout, and suicide in academic medicine.16
Anita Everett, MD, put the spotlight on physician well-being during her term as American Psychiatric Association President (2017 to 2018). She formed a specific workgroup on Physician Wellness and Burnout where there is a community focus on prevention and self-care.17 A strong sense of community and purpose is almost always part of the prescription for promoting greater well-being.2
The importance of this issue is also trickling down from policymakers into hospitals and community health centers. Consider an initiative at Minneapolis’s Hennepin County Medical Center. Leaders there created a “reset room” for physicians to quietly decompress. The room is complete with LED lights, flameless candles, a sound machine, comfortable chairs, several plants, and an “in use” sign on the door.18 Other personal strategies to help prevent burnout include making environmental changes, encouraging hobbies, and streamlining burdensome tasks such as paperwork and electronic medical record systems.
As physician health and well-being are finally emerging as a “hot topic,”2 educational and treatment resources are increasingly available for any of us to explore. Consider a simple Google search to look into your State’s PHPs, and get involved in your professional societies to make change.
The culture is starting to shift, and leading by example will be a key to propelling further progress in this area. Model our own self-care for colleagues and patients alike. As Mark Twain said, we might love our work, but we must remember that being solely defined by work comes to the detriment of our health. Maintaining balance is what will allow us to sustain long careers ahead doing what we love.
Psychiatry is a field of passion. The reward of experiencing growth and change alongside our patients is what bolsters us through years of difficult training, overnight shifts, endless paperwork, regulatory mandates, and frequent worry about our patients. As physicians, we don’t live for weekends as many other professionals do. To the contrary, we spend them on call, moonlighting, laboring over journal articles, and perfecting lectures.
That passion is what makes us trusted clinicians and experts in our field. It can also make it difficult for us to disconnect from our work, frequently leading to burnout. Physician self-care, support, and professional development are critical topics that modern-day medicine minimizes at the peril of physicians and public health.1
Psychiatry lends itself to a deep and intimate understanding of another human being. The therapist delves into the lives of his or her patients, hears their stories, and holds their secrets. In some cases, we might be the only ones who truly see patients for who they uniquely are, and come to understand them on a deeper level than their closest family and friends. This can be both thrilling and intense. As we delve into the psyche of another individual, contemplate which interpretation we should share, and resonate with our patients, it is easy to become bogged down with our own countertransference, sentiment, and worry, and to become consumed by our work. A professional hazard, some might quip.
Therefore, personal restoration—a tool that keeps our clinical skills sharp—is vitally important to caring for oneself and one’s patient. Surprisingly, this can be neglected until we begin to experience burnout, which over time could transform into impairment, thus endangering ourselves, our patients, and our profession.
Over the past decade, physician impairment has been exhaustively described, researched, and addressed. However, most analyses have focused on identifying impairment, and offering guidance on how to properly report it. How do we shift from managing the crisis to preventing it? To answer this question, this article:
- reviews the dilemma of physician impairment
- explores the duty we have to patients, ourselves, and the profession
- discusses shifting the focus on impairment to prevention through well-being.
Continue to: Dilemma
Dilemma: Vulnerability to impairment
The cornerstone for well-being is a balanced life. No matter how much one loves his or her work, there must be balance between work, relationships, and hobbies. Without that equilibrium, everyone is put at risk.2
Just as our patients, we are not immune to mental illness, cognitive decline, or substance abuse.3 We might even be more susceptible. For many physicians, their identity is intimately tied to their work.4 Dr. Robin Weiss captured that intimate relationship5:
“… [A] therapist may spend hundreds of hours, perhaps more than a thousand, hearing about a patient’s most exalted aspirations and most murderous, hateful fantasies. During this time, the patient may endure excruciating losses, unbearable shame, bitter sadness and great triumphs. You may accompany patients through torturous adolescence into adulthood. Or you may meet them in middle age and be with them as they age and eventually die. You collaborate in a deep process of discovery. Few encounters are this deeply honest, and therefore intimate.”
Given the stories we hear and the resulting intimacy and countertransference that inevitably arise, psychiatrists are even more prone to burnout than other physicians.6 Physician impairment is a public health issue that affects not just physicians but also their families, colleagues, and patients.
“Impairment” for the purpose of this article means a physical, mental, or substance-related disorder that interferes with a physician’s ability to undertake professional activities competently and safely.7 Predisposing factors for physician impairment include an obsessive-compulsive personality type, a family history of mental illness, sensation-seeking behavior, denial of personal problems, perfectionism, and idealism.8,9 Also, work stress becomes a significant factor in already vulnerable physicians, leading to a greater risk for mental illness.10
Continue to: Some warning signs of impairment include...
Some warning signs of impairment include a lack of personal hygiene, emotional lability, sleep deprivation, inattention to our pages or phone calls, and increased professional errors.11 When it comes to addressing such impairment, previous research and literature has focused on how to monitor ourselves and our colleagues; anything less would put the reputation and integrity of the medical profession at risk.3 This has led to a culture of doing nothing but work until things go too far, and then reporting the problems. But what about intervening before things get too far?
Duty: To ourselves, our colleagues, and our patients
There has been much discussion on how to report impaired colleagues, but little on how to help and support ourselves and our colleagues before things escalate into serious problems. And this lack of discussion is at the detriment of individual practitioners, their families, and patients. Physicians-in-training, including psychiatric residents, are at particularly high risk for developing stress-related problems, depression, and substance misuse.12 Occupational demands, self-criticism, and denial of one’s distress are common among physicians, as is self-treatment with drugs and alcohol.13
We all know by now that doctors and physician health programs (PHPs) have a duty to report impaired colleagues who continue to practice despite reasonable offers of assistance. There are an abundance of PHPs that are in place to assist with such situations. The American Medical Association’s official position on reporting impairment is outlined in Policy H-275.952.7 There also is the Federation of State Medical Boards. Its policy states that PHPs have “a primary commitment to [help] state medical boards … protect the public … [These] programs [should] demonstrate an ongoing track of record of ensuring safety to the public and reveal deficiencies if they occur.”14
Legal and ethical issues, however, complicate interventions for colleagues who need assistance.15 Despite the existence of PHPs, it would be much easier—not to mention helpful—to help a colleague by carrying out early interventions.
Discussion: Prevention as a solution
More emphasis should be placed on prevention. That’s where self-care and well-being come into play. Awareness of and sensitivity to physician vulnerability, early detection, and prevention of impairment are important.
Continue to: There has been a paradigm shift in focus...
There has been a paradigm shift in focus across medical boards, professional societies, and medical colleges. They are recognizing that personal well-being can help prevent burnout and, in turn, change the landscape of medicine from endless work to balanced lives that yield more satisfying and joyful work. It is becoming an accepted fact in medicine that well-being is just as important as integrity, professionalism, and patient safety. For example, the American Academy of Medical Colleges (AAMC) issued a statement emphasizing the importance of clinician well-being and dedicated its June 2016 Leadership Forum to a range of topics addressing depression, resilience, burnout, and suicide in academic medicine.16
Anita Everett, MD, put the spotlight on physician well-being during her term as American Psychiatric Association President (2017 to 2018). She formed a specific workgroup on Physician Wellness and Burnout where there is a community focus on prevention and self-care.17 A strong sense of community and purpose is almost always part of the prescription for promoting greater well-being.2
The importance of this issue is also trickling down from policymakers into hospitals and community health centers. Consider an initiative at Minneapolis’s Hennepin County Medical Center. Leaders there created a “reset room” for physicians to quietly decompress. The room is complete with LED lights, flameless candles, a sound machine, comfortable chairs, several plants, and an “in use” sign on the door.18 Other personal strategies to help prevent burnout include making environmental changes, encouraging hobbies, and streamlining burdensome tasks such as paperwork and electronic medical record systems.
As physician health and well-being are finally emerging as a “hot topic,”2 educational and treatment resources are increasingly available for any of us to explore. Consider a simple Google search to look into your State’s PHPs, and get involved in your professional societies to make change.
The culture is starting to shift, and leading by example will be a key to propelling further progress in this area. Model our own self-care for colleagues and patients alike. As Mark Twain said, we might love our work, but we must remember that being solely defined by work comes to the detriment of our health. Maintaining balance is what will allow us to sustain long careers ahead doing what we love.
1. Mahoney, D, Freedy J, Brock C. Improving physician well-being. JAMA Intern Med. 2015;175(4):648-649.
2. Yellowlees P. Addressing physician health and well-being is patient safety issue. Psychiatric News. 2018;53(12):20-21.
3. Mossman D, Farrell HM. Physician impairment: when should you report? Current Psychiatry. 2011;10(9):67-71.
4. Lindeman S, Henriksson M, Isometsä E, et al. Treatment of mental disorders in seven physicians committing suicide. Crisis. 1999;20(2):86-89.
5. Weiss R. How therapists mourn. New York Times. July 4, 2015:SR2.
6. Kumar S. Burnout in psychiatrists. World Psychiatry. 2007;6(3):186-189.
7. American Medical Association. Report 2 of the Council on Science and Public Health (A-11). Physician health programs (Reference Committee D). https://www.ama-assn.org/sites/default/files/media-browser/public/about-ama/councils/Council%20Reports/council-on-science-public-health/a11-csaph-physician-health-programs.pdf. Accessed August 6, 2018.
8. Boisaubin EV, Levine RE. Identifying and assisting the impaired physician. Am J Med Sci. 2001;322(1):31-36.
9. Bissel L, Jones RW. The alcoholic physician: a survey. Am J Psychiatry. 1976;133(10):1142-1146.
10. Vaillant GE, Sobowale NC, McArthur C. Some psychologic vulnerabilities of physicians. N Engl J Med. 1972;287(8):372-375.
11. McGovern MP, Agnes DH, Leon S. Characteristics of physicians presenting for assessment at a behavioral health center. J Addict Dis. 2000;19(2):59-73.
12. Broquet KE, Rockey PH. Teaching residents and program directors about physician impairment. Acad Psychiatry. 2004;28(3):221-225.
13. Meier DE, Back AL, Morrison RS. The inner life of physicians and care of the seriously ill. JAMA. 2001;286(23):3007-3014.
14. Federation of State Medical Boards of the United States. Policy on physician impairment. http://www.csam-asam.org/pdf/misc/FSMB2011.pdf. Published 2011. Accessed July 15, 2018.
15. Bright RP, Krahn L. Impaired physicians: how to recognize, when to report, and where to refer. Current Psychiatry. 2010;9(6):11-20.
16. Academy of American Colleges. Well-being in academic medicine. https://www.aamc.org/initiatives/462280/well-being-academic-medicine.html. Updated July 9, 2018. Accessed July 17, 2018.
17. American Psychiatric Association. Well-being and burnout. https://www.psychiatry.org/psychiatrists/practice/well-being-and-burnout. Updated February 22, 2018. Accessed July 17, 2018.
18. Parks T. Physicians take to “reset room” to battle burnout. AMA Wire. https://wire.ama-assn.org/practice-management/physicians-take-reset-room-battle-burnout. Published June 8, 2016. Accessed July 18, 2018.
1. Mahoney, D, Freedy J, Brock C. Improving physician well-being. JAMA Intern Med. 2015;175(4):648-649.
2. Yellowlees P. Addressing physician health and well-being is patient safety issue. Psychiatric News. 2018;53(12):20-21.
3. Mossman D, Farrell HM. Physician impairment: when should you report? Current Psychiatry. 2011;10(9):67-71.
4. Lindeman S, Henriksson M, Isometsä E, et al. Treatment of mental disorders in seven physicians committing suicide. Crisis. 1999;20(2):86-89.
5. Weiss R. How therapists mourn. New York Times. July 4, 2015:SR2.
6. Kumar S. Burnout in psychiatrists. World Psychiatry. 2007;6(3):186-189.
7. American Medical Association. Report 2 of the Council on Science and Public Health (A-11). Physician health programs (Reference Committee D). https://www.ama-assn.org/sites/default/files/media-browser/public/about-ama/councils/Council%20Reports/council-on-science-public-health/a11-csaph-physician-health-programs.pdf. Accessed August 6, 2018.
8. Boisaubin EV, Levine RE. Identifying and assisting the impaired physician. Am J Med Sci. 2001;322(1):31-36.
9. Bissel L, Jones RW. The alcoholic physician: a survey. Am J Psychiatry. 1976;133(10):1142-1146.
10. Vaillant GE, Sobowale NC, McArthur C. Some psychologic vulnerabilities of physicians. N Engl J Med. 1972;287(8):372-375.
11. McGovern MP, Agnes DH, Leon S. Characteristics of physicians presenting for assessment at a behavioral health center. J Addict Dis. 2000;19(2):59-73.
12. Broquet KE, Rockey PH. Teaching residents and program directors about physician impairment. Acad Psychiatry. 2004;28(3):221-225.
13. Meier DE, Back AL, Morrison RS. The inner life of physicians and care of the seriously ill. JAMA. 2001;286(23):3007-3014.
14. Federation of State Medical Boards of the United States. Policy on physician impairment. http://www.csam-asam.org/pdf/misc/FSMB2011.pdf. Published 2011. Accessed July 15, 2018.
15. Bright RP, Krahn L. Impaired physicians: how to recognize, when to report, and where to refer. Current Psychiatry. 2010;9(6):11-20.
16. Academy of American Colleges. Well-being in academic medicine. https://www.aamc.org/initiatives/462280/well-being-academic-medicine.html. Updated July 9, 2018. Accessed July 17, 2018.
17. American Psychiatric Association. Well-being and burnout. https://www.psychiatry.org/psychiatrists/practice/well-being-and-burnout. Updated February 22, 2018. Accessed July 17, 2018.
18. Parks T. Physicians take to “reset room” to battle burnout. AMA Wire. https://wire.ama-assn.org/practice-management/physicians-take-reset-room-battle-burnout. Published June 8, 2016. Accessed July 18, 2018.