User login
FDA OKs first drug for Rett syndrome
Rett syndrome is a rare, genetic neurodevelopmental disorder that affects about 6,000-9,000 people in the United States, mostly females.
Symptoms typically present between 6 and 18 months of age, with patients experiencing a rapid decline with loss of fine motor and communication skills.
Trofinetide is a synthetic analogue of the amino-terminal tripeptide of insulinlike growth factor-1 (IGF-1), which occurs naturally in the brain. The drug is designed to treat the core symptoms of Rett syndrome by potentially reducing neuroinflammation and supporting synaptic function.
The approval of trofinetide was supported by results from the pivotal phase 3 LAVENDER study that tested the efficacy and safety of trofinetide vs. placebo in 187 female patients with Rett syndrome, aged 5-20 years.
A total of 93 participants were randomly assigned to twice-daily oral trofinetide, and 94 received placebo for 12 weeks.
After 12 weeks, trofinetide showed a statistically significant improvement from baseline, compared with placebo, on both the caregiver-assessed Rett Syndrome Behavior Questionnaire (RSBQ) and 7-point Clinical Global Impression-Improvement (CGI-I) scale.
The drug also outperformed placebo at 12 weeks in a key secondary endpoint: the composite score on the Communication and Symbolic Behavior Scales Developmental Profile Infant-Toddler Checklist-Social (CSBS-DP-IT Social), a scale on which caregivers assess nonverbal communication.
The most common adverse events with trofinetide treatment were diarrhea and vomiting. Almost all these events were considered mild or moderate.
‘Historic day’
“This is a historic day for the Rett syndrome community and a meaningful moment for the patients and caregivers who have eagerly awaited the arrival of an approved treatment for this condition,” Melissa Kennedy, MHA, chief executive officer of the International Rett Syndrome Foundation, said in a news release issued by Acadia.
“Rett syndrome is a complicated, devastating disease that affects not only the individual patient, but whole families. With today’s FDA decision, those impacted by Rett have a promising new treatment option that has demonstrated benefit across a variety of Rett symptoms, including those that impact the daily lives of those living with Rett and their loved ones,” Ms. Kennedy said.
Trofinetide is expected to be available in the United States by the end of April.
A version of this article first appeared on Medscape.com.
Rett syndrome is a rare, genetic neurodevelopmental disorder that affects about 6,000-9,000 people in the United States, mostly females.
Symptoms typically present between 6 and 18 months of age, with patients experiencing a rapid decline with loss of fine motor and communication skills.
Trofinetide is a synthetic analogue of the amino-terminal tripeptide of insulinlike growth factor-1 (IGF-1), which occurs naturally in the brain. The drug is designed to treat the core symptoms of Rett syndrome by potentially reducing neuroinflammation and supporting synaptic function.
The approval of trofinetide was supported by results from the pivotal phase 3 LAVENDER study that tested the efficacy and safety of trofinetide vs. placebo in 187 female patients with Rett syndrome, aged 5-20 years.
A total of 93 participants were randomly assigned to twice-daily oral trofinetide, and 94 received placebo for 12 weeks.
After 12 weeks, trofinetide showed a statistically significant improvement from baseline, compared with placebo, on both the caregiver-assessed Rett Syndrome Behavior Questionnaire (RSBQ) and 7-point Clinical Global Impression-Improvement (CGI-I) scale.
The drug also outperformed placebo at 12 weeks in a key secondary endpoint: the composite score on the Communication and Symbolic Behavior Scales Developmental Profile Infant-Toddler Checklist-Social (CSBS-DP-IT Social), a scale on which caregivers assess nonverbal communication.
The most common adverse events with trofinetide treatment were diarrhea and vomiting. Almost all these events were considered mild or moderate.
‘Historic day’
“This is a historic day for the Rett syndrome community and a meaningful moment for the patients and caregivers who have eagerly awaited the arrival of an approved treatment for this condition,” Melissa Kennedy, MHA, chief executive officer of the International Rett Syndrome Foundation, said in a news release issued by Acadia.
“Rett syndrome is a complicated, devastating disease that affects not only the individual patient, but whole families. With today’s FDA decision, those impacted by Rett have a promising new treatment option that has demonstrated benefit across a variety of Rett symptoms, including those that impact the daily lives of those living with Rett and their loved ones,” Ms. Kennedy said.
Trofinetide is expected to be available in the United States by the end of April.
A version of this article first appeared on Medscape.com.
Rett syndrome is a rare, genetic neurodevelopmental disorder that affects about 6,000-9,000 people in the United States, mostly females.
Symptoms typically present between 6 and 18 months of age, with patients experiencing a rapid decline with loss of fine motor and communication skills.
Trofinetide is a synthetic analogue of the amino-terminal tripeptide of insulinlike growth factor-1 (IGF-1), which occurs naturally in the brain. The drug is designed to treat the core symptoms of Rett syndrome by potentially reducing neuroinflammation and supporting synaptic function.
The approval of trofinetide was supported by results from the pivotal phase 3 LAVENDER study that tested the efficacy and safety of trofinetide vs. placebo in 187 female patients with Rett syndrome, aged 5-20 years.
A total of 93 participants were randomly assigned to twice-daily oral trofinetide, and 94 received placebo for 12 weeks.
After 12 weeks, trofinetide showed a statistically significant improvement from baseline, compared with placebo, on both the caregiver-assessed Rett Syndrome Behavior Questionnaire (RSBQ) and 7-point Clinical Global Impression-Improvement (CGI-I) scale.
The drug also outperformed placebo at 12 weeks in a key secondary endpoint: the composite score on the Communication and Symbolic Behavior Scales Developmental Profile Infant-Toddler Checklist-Social (CSBS-DP-IT Social), a scale on which caregivers assess nonverbal communication.
The most common adverse events with trofinetide treatment were diarrhea and vomiting. Almost all these events were considered mild or moderate.
‘Historic day’
“This is a historic day for the Rett syndrome community and a meaningful moment for the patients and caregivers who have eagerly awaited the arrival of an approved treatment for this condition,” Melissa Kennedy, MHA, chief executive officer of the International Rett Syndrome Foundation, said in a news release issued by Acadia.
“Rett syndrome is a complicated, devastating disease that affects not only the individual patient, but whole families. With today’s FDA decision, those impacted by Rett have a promising new treatment option that has demonstrated benefit across a variety of Rett symptoms, including those that impact the daily lives of those living with Rett and their loved ones,” Ms. Kennedy said.
Trofinetide is expected to be available in the United States by the end of April.
A version of this article first appeared on Medscape.com.
Children and COVID: A look back as the fourth year begins
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?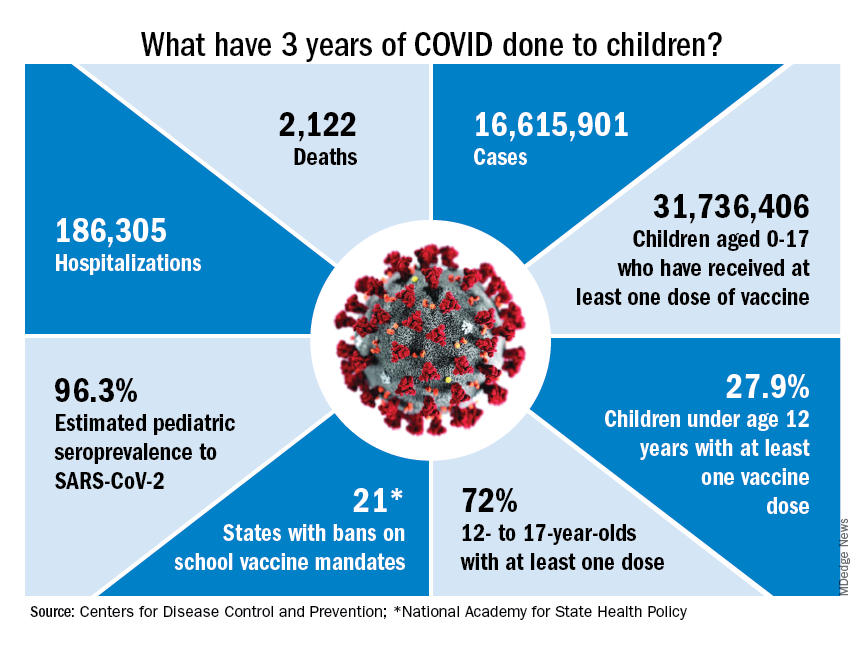
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
With 3 years of the COVID-19 experience now past, it’s safe to say that SARS-CoV-2 changed American society in ways that could not have been predicted when the first U.S. cases were reported in January of 2020.
Who would have guessed back then that not one but two vaccines would be developed, approved, and widely distributed before the end of the year? Or that those vaccines would be rejected by large segments of the population on ideological grounds? Could anyone have predicted in early 2020 that schools in 21 states would be forbidden by law to require COVID-19 vaccination in students?
Vaccination is generally considered to be an activity of childhood, but that practice has been turned upside down with COVID-19. Among Americans aged 65 years and older, 95% have received at least one dose of vaccine, versus 27.9% of children younger than 12 years old, according to the Centers for Disease Control and Prevention.
The vaccine situation for children mirrors that of the population as a whole. The oldest children have the highest vaccination rates, and the rates decline along with age: 72.0% of those aged 12-17 years have received at least one dose, compared with 39.8% of 5- to 11-year-olds, 10.5% of 2- to 4-year-olds, and 8.0% of children under age 2, the CDC said on its COVID Data Tracker.
The youngest children were, of course, the last ones to be eligible for the vaccine, but their uptake has been much slower since emergency use was authorized in June of 2022. In the nearly 9 months since then, 9.5% of children aged 4 and under have received at least one dose, versus 66% of children aged 12-15 years in the first 9 months (May 2021 to March 2022).
Altogether, a total of 31.7 million, or 43%, of all children under age 18 had received at least one dose of COVID-19 vaccine as of March 8, 2023, according to the most recent CDC data.
Incidence: Counting COVID
Vaccination and other prevention efforts have tried to stem the tide, but what has COVID actually done to children since the Trump administration declared a nationwide emergency on March 13, 2020?
- 16.6 million cases.
- 186,035 new hospital admissions.
- 2,122 deaths.
Even the proportion of total COVID cases in children, 17.2%, is less than might be expected, given their relatively undervaccinated status.
Seroprevalence estimates seem to support the undercounting of pediatric cases. A survey of commercial laboratories working with the CDC put the seroprevalance of SARS-CoV-2 antibodies in children at 96.3% as of late 2022, based on tests of almost 27,000 specimens performed over an 8-week period from mid-October to mid-December. That would put the number of infected children at 65.7 million children.
Since Omicron
There has not been another major COVID-19 surge since the winter of 2021-2022, when the weekly rate of new cases reached 1,900 per 100,000 population in children aged 16-17 years in early January 2022 – the highest seen among children of any of the CDC’s age groups (0-4, 5-11, 12-15, 16-17) during the entire pandemic. Since the Omicron surge, the highest weekly rate was 221 per 100,000 during the week of May 15-21, again in 16- to 17-year-olds, the CDC reports.
The widely anticipated surge of COVID in the fall and winter of 2022 and 2023 – the so-called “tripledemic” involving influenza and respiratory syncytial virus – did not occur, possibly because so many Americans were vaccinated or previously infected, experts suggested. New-case rates, emergency room visits, and hospitalizations in children have continued to drop as winter comes to a close, CDC data show.
Strong support for CBT as first-line treatment for insomnia in seniors
NEW ORLEANS –
“The lack of awareness among clinicians who take care of older adults that CBT for insomnia (CBT-I) is an effective treatment for insomnia is an issue,” Rajesh R. Tampi, MD, professor and chairman of the department of psychiatry, Creighton University, Omaha, Neb., told this news organization.
Dr. Tampi was among the speakers during a session as part of the American Association for Geriatric Psychiatry annual meeting addressing the complex challenges of treating insomnia in older patients, who tend to have higher rates of insomnia than their younger counterparts.
The prevalence of insomnia in older adults is estimated to be 20%-40%, and medication is frequently the first treatment choice, a less than ideal approach, said Dr. Tampi.
“Prescribing sedatives and hypnotics, which can cause severe adverse effects, without a thorough assessment that includes comorbidities that may be causing the insomnia” is among the biggest mistakes clinicians make in the treatment of insomnia in older patients, Dr. Tampi said in an interview.
“It’s our duty as providers to first take a good assessment, talk about polymorbidity, and try to address those conditions, and judiciously use medications in conjunction with at least components of CBT-I,” he said.
Long-term safety, efficacy unclear
About one-third of older adults take at least one form of pharmacological treatment for insomnia symptoms, said Ebony Dix, MD, assistant professor of psychiatry at Yale University, New Haven, Conn., in a separate talk during the session. This, despite the low-risk profile of CBT and recommendations from various medical societies that CBT should be tried first.
Dr. Dix noted that medications approved for insomnia by the U.S. Food and Drug Administration, including melatonin receptor agonists, heterocyclics, and dual orexin receptor antagonists (DORAs), can play an important role in the short-term management of insomnia, but their long-term effects are unknown.
“Pharmacotherapeutic agents may be effective in the short term, but there is a lack of sufficient, statistically significant data to support the long-term safety and efficacy of any [sleep] medication, especially in aging adults, due to the impact of hypnotic drugs on sleep architecture, the impact of aging on pharmacokinetics, as well as polypharmacy and drug-to-drug interactions,” Dr. Dix said. She noted that clinical trials of insomnia drugs rarely include geriatric patients.
The American Academy of Sleep Medicine recommends CBT-I as first-line treatment for insomnia, with the key benefit being its exemplary safety profile, said Shilpa Srinivasan, MD, a professor of clinical psychiatry at the University of South Carolina, Columbia, who also presented during the session.
“The biggest [attribute] of CBT-I management strategies is the low risk of side effects,” she said. “How many medications can we say that about?”
The CBT-I intervention includes a focus on key components of lifestyle and mental health issues to improve sleep. These include the following:
- Strictly restricting sleep hours for bedtime and arising (with napping discouraged).
- Control of stimulus to disrupt falling asleep.
- Cognitive therapy to identify and replace maladaptive beliefs.
- Control of sleep hygiene for optimal sleep.
- Relaxation training.
Keys to success
Dr. Srinivasan noted one recent study of CBT-I among patients aged 60 and older with insomnia and depression. The 156 participants randomized to receive weekly 120-minute CBT-I sessions over 2 months were significantly less likely to develop new or recurrent major depression versus their counterparts randomized to receive sleep education (hazard ratio, 0.51; P = .02).
However, CBT-I is more labor intensive than medication and requires provider training and motivation, and commitment on the part of the patient, to be successful.
“We really need to ensure that even when patients are receiving pharmacologic interventions for insomnia that we provide psychoeducation. At the end of the day, some of these nonpharmacologic components can make or break the success of pharmacotherapy,” said Dr. Srinivasan.
Whether using CBT-I alone or in combination with pharmacotherapy, the intervention does not necessarily have to include all components to be beneficial, she said.
“I think one of the challenges in incorporating CBT-I is the misconception that it is an all-or-nothing approach wherein every modality must be utilized,” she said. “While multicomponent CBT-I has been shown to be effective, the individual components can be incorporated into patient encounters in a stepped approach.”
Informing patients that they have options other than medications and involving them in decision-making is key, she added.
“In the case of insomnia, this is particularly relevant because of the physical and emotional distress that it causes,” Dr. Srinivasan said. “Patients often seek over-the-counter medications or other nonprescribed agents to try to obtain relief even before seeking treatment in a health care setting. There is less awareness about evidence-based and effective nonpharmacologic treatments such as CBT-I.”
Dr. Tampi, Dr. Dix, and Dr. Srinivasan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW ORLEANS –
“The lack of awareness among clinicians who take care of older adults that CBT for insomnia (CBT-I) is an effective treatment for insomnia is an issue,” Rajesh R. Tampi, MD, professor and chairman of the department of psychiatry, Creighton University, Omaha, Neb., told this news organization.
Dr. Tampi was among the speakers during a session as part of the American Association for Geriatric Psychiatry annual meeting addressing the complex challenges of treating insomnia in older patients, who tend to have higher rates of insomnia than their younger counterparts.
The prevalence of insomnia in older adults is estimated to be 20%-40%, and medication is frequently the first treatment choice, a less than ideal approach, said Dr. Tampi.
“Prescribing sedatives and hypnotics, which can cause severe adverse effects, without a thorough assessment that includes comorbidities that may be causing the insomnia” is among the biggest mistakes clinicians make in the treatment of insomnia in older patients, Dr. Tampi said in an interview.
“It’s our duty as providers to first take a good assessment, talk about polymorbidity, and try to address those conditions, and judiciously use medications in conjunction with at least components of CBT-I,” he said.
Long-term safety, efficacy unclear
About one-third of older adults take at least one form of pharmacological treatment for insomnia symptoms, said Ebony Dix, MD, assistant professor of psychiatry at Yale University, New Haven, Conn., in a separate talk during the session. This, despite the low-risk profile of CBT and recommendations from various medical societies that CBT should be tried first.
Dr. Dix noted that medications approved for insomnia by the U.S. Food and Drug Administration, including melatonin receptor agonists, heterocyclics, and dual orexin receptor antagonists (DORAs), can play an important role in the short-term management of insomnia, but their long-term effects are unknown.
“Pharmacotherapeutic agents may be effective in the short term, but there is a lack of sufficient, statistically significant data to support the long-term safety and efficacy of any [sleep] medication, especially in aging adults, due to the impact of hypnotic drugs on sleep architecture, the impact of aging on pharmacokinetics, as well as polypharmacy and drug-to-drug interactions,” Dr. Dix said. She noted that clinical trials of insomnia drugs rarely include geriatric patients.
The American Academy of Sleep Medicine recommends CBT-I as first-line treatment for insomnia, with the key benefit being its exemplary safety profile, said Shilpa Srinivasan, MD, a professor of clinical psychiatry at the University of South Carolina, Columbia, who also presented during the session.
“The biggest [attribute] of CBT-I management strategies is the low risk of side effects,” she said. “How many medications can we say that about?”
The CBT-I intervention includes a focus on key components of lifestyle and mental health issues to improve sleep. These include the following:
- Strictly restricting sleep hours for bedtime and arising (with napping discouraged).
- Control of stimulus to disrupt falling asleep.
- Cognitive therapy to identify and replace maladaptive beliefs.
- Control of sleep hygiene for optimal sleep.
- Relaxation training.
Keys to success
Dr. Srinivasan noted one recent study of CBT-I among patients aged 60 and older with insomnia and depression. The 156 participants randomized to receive weekly 120-minute CBT-I sessions over 2 months were significantly less likely to develop new or recurrent major depression versus their counterparts randomized to receive sleep education (hazard ratio, 0.51; P = .02).
However, CBT-I is more labor intensive than medication and requires provider training and motivation, and commitment on the part of the patient, to be successful.
“We really need to ensure that even when patients are receiving pharmacologic interventions for insomnia that we provide psychoeducation. At the end of the day, some of these nonpharmacologic components can make or break the success of pharmacotherapy,” said Dr. Srinivasan.
Whether using CBT-I alone or in combination with pharmacotherapy, the intervention does not necessarily have to include all components to be beneficial, she said.
“I think one of the challenges in incorporating CBT-I is the misconception that it is an all-or-nothing approach wherein every modality must be utilized,” she said. “While multicomponent CBT-I has been shown to be effective, the individual components can be incorporated into patient encounters in a stepped approach.”
Informing patients that they have options other than medications and involving them in decision-making is key, she added.
“In the case of insomnia, this is particularly relevant because of the physical and emotional distress that it causes,” Dr. Srinivasan said. “Patients often seek over-the-counter medications or other nonprescribed agents to try to obtain relief even before seeking treatment in a health care setting. There is less awareness about evidence-based and effective nonpharmacologic treatments such as CBT-I.”
Dr. Tampi, Dr. Dix, and Dr. Srinivasan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW ORLEANS –
“The lack of awareness among clinicians who take care of older adults that CBT for insomnia (CBT-I) is an effective treatment for insomnia is an issue,” Rajesh R. Tampi, MD, professor and chairman of the department of psychiatry, Creighton University, Omaha, Neb., told this news organization.
Dr. Tampi was among the speakers during a session as part of the American Association for Geriatric Psychiatry annual meeting addressing the complex challenges of treating insomnia in older patients, who tend to have higher rates of insomnia than their younger counterparts.
The prevalence of insomnia in older adults is estimated to be 20%-40%, and medication is frequently the first treatment choice, a less than ideal approach, said Dr. Tampi.
“Prescribing sedatives and hypnotics, which can cause severe adverse effects, without a thorough assessment that includes comorbidities that may be causing the insomnia” is among the biggest mistakes clinicians make in the treatment of insomnia in older patients, Dr. Tampi said in an interview.
“It’s our duty as providers to first take a good assessment, talk about polymorbidity, and try to address those conditions, and judiciously use medications in conjunction with at least components of CBT-I,” he said.
Long-term safety, efficacy unclear
About one-third of older adults take at least one form of pharmacological treatment for insomnia symptoms, said Ebony Dix, MD, assistant professor of psychiatry at Yale University, New Haven, Conn., in a separate talk during the session. This, despite the low-risk profile of CBT and recommendations from various medical societies that CBT should be tried first.
Dr. Dix noted that medications approved for insomnia by the U.S. Food and Drug Administration, including melatonin receptor agonists, heterocyclics, and dual orexin receptor antagonists (DORAs), can play an important role in the short-term management of insomnia, but their long-term effects are unknown.
“Pharmacotherapeutic agents may be effective in the short term, but there is a lack of sufficient, statistically significant data to support the long-term safety and efficacy of any [sleep] medication, especially in aging adults, due to the impact of hypnotic drugs on sleep architecture, the impact of aging on pharmacokinetics, as well as polypharmacy and drug-to-drug interactions,” Dr. Dix said. She noted that clinical trials of insomnia drugs rarely include geriatric patients.
The American Academy of Sleep Medicine recommends CBT-I as first-line treatment for insomnia, with the key benefit being its exemplary safety profile, said Shilpa Srinivasan, MD, a professor of clinical psychiatry at the University of South Carolina, Columbia, who also presented during the session.
“The biggest [attribute] of CBT-I management strategies is the low risk of side effects,” she said. “How many medications can we say that about?”
The CBT-I intervention includes a focus on key components of lifestyle and mental health issues to improve sleep. These include the following:
- Strictly restricting sleep hours for bedtime and arising (with napping discouraged).
- Control of stimulus to disrupt falling asleep.
- Cognitive therapy to identify and replace maladaptive beliefs.
- Control of sleep hygiene for optimal sleep.
- Relaxation training.
Keys to success
Dr. Srinivasan noted one recent study of CBT-I among patients aged 60 and older with insomnia and depression. The 156 participants randomized to receive weekly 120-minute CBT-I sessions over 2 months were significantly less likely to develop new or recurrent major depression versus their counterparts randomized to receive sleep education (hazard ratio, 0.51; P = .02).
However, CBT-I is more labor intensive than medication and requires provider training and motivation, and commitment on the part of the patient, to be successful.
“We really need to ensure that even when patients are receiving pharmacologic interventions for insomnia that we provide psychoeducation. At the end of the day, some of these nonpharmacologic components can make or break the success of pharmacotherapy,” said Dr. Srinivasan.
Whether using CBT-I alone or in combination with pharmacotherapy, the intervention does not necessarily have to include all components to be beneficial, she said.
“I think one of the challenges in incorporating CBT-I is the misconception that it is an all-or-nothing approach wherein every modality must be utilized,” she said. “While multicomponent CBT-I has been shown to be effective, the individual components can be incorporated into patient encounters in a stepped approach.”
Informing patients that they have options other than medications and involving them in decision-making is key, she added.
“In the case of insomnia, this is particularly relevant because of the physical and emotional distress that it causes,” Dr. Srinivasan said. “Patients often seek over-the-counter medications or other nonprescribed agents to try to obtain relief even before seeking treatment in a health care setting. There is less awareness about evidence-based and effective nonpharmacologic treatments such as CBT-I.”
Dr. Tampi, Dr. Dix, and Dr. Srinivasan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT AAGP 2023
Will new guidelines widen the gap in treating childhood obesity?
In the United States, the Centers for Disease Control and Prevention estimates that nearly one in five children have obesity. Since the 1980s, the number of children with obesity has been increasing, with each generation reaching higher rates and greater weights at earlier ages. Even with extensive efforts from parents, clinicians, educators, and policymakers to limit the excessive weight gain among children, the number of obesity and severe obesity diagnoses keeps rising.
In response to this critical public health challenge, the American Academy of Pediatrics (AAP) introduced new clinical practice guidelines for the evaluation and management of obesity in children and adolescents. Developed by an expert panel, the new AAP guidelines present a departure in the conceptualization of obesity, recognizing the role that social determinants of health play in contributing to excessive weight gain.
As a community health researcher who investigates disparities in childhood obesity, I applaud the paradigm shift from the AAP. I specifically endorse the recognition that obesity is a very serious metabolic disease that won’t go away unless we introduce systemic changes and effective treatments.
However, I, like so many of my colleagues and anyone aware of the access barriers to the recommended treatments, worry about the consequences that the new guidelines will have in the context of current and future health disparities.
A recent study, published in Pediatrics, showed that childhood obesity disparities are widening. Younger generations of children are reaching higher weights at younger ages. These alarming trends are greater among Black children and children growing up with the greatest socioeconomic disadvantages. The new AAP guidelines – even while driven by good intentions – can exacerbate these differences and set children who are able to live healthy lives further apart from those with disproportionate obesity risks, who lack access to the treatments recommended by the AAP.
Rather than “watchful waiting,” to see if children outgrow obesity, the new guidelines call for “aggressive treatment,” as reported by this news organization. At least 26 hours of in-person intensive health behavior and lifestyle counseling and treatment are recommended for children aged 2 years old or older who meet the obesity criteria. For children aged 12 years or older, the AAP recommends complementing lifestyle counseling with pharmacotherapy. This breakthrough welcomes the use of promising antiobesity medications (for example, orlistat, Wegovy [semaglutide], Saxenda [liraglutide], Qsymia (phentermine and topiramate]) approved by the Food and Drug Administration for long-term use in children aged 12 and up. For children 13 years or older with severe obesity, bariatric surgery should be considered.
Will cost barriers continue to increase disparity?
The very promising semaglutide (Wegovy) is a GLP-1–based medication currently offered for about $1,000 per month. As with other chronic diseases, children should be prepared to take obesity medications for prolonged periods of time. A study conducted in adults found that when the medication is suspended, any weight loss can be regained. The costs of bariatric surgery total over $20,000.
In the U.S. health care system, at current prices, very few of the children in need of the medications or surgical treatments have access to them. Most private health insurance companies and Medicaid reject coverage for childhood obesity treatments. Barriers to treatment access are greater for Black and Hispanic children, children growing up in poverty, and children living in the U.S. South region, all of whom are more likely to develop obesity earlier in life than their White and wealthier counterparts.
The AAP recognized that a substantial time and financial commitment is required to follow the new treatment recommendations. Members of the AAP Expert Committee that developed the guidelines stated that they are “aware of the multitude of barriers to treatment that patients and their families face.”
Nevertheless, the recognition of the role of social determinants of health in the development of childhood obesity didn’t motivate the introduction of treatment options that aren’t unattainable for most U.S. families.
It’s important to step away from the conclusion that because of the price tag, at the population level, the new AAP guidelines will be inconsequential. This conclusion fails to recognize the potential harm that the guidelines may introduce. In the context of childhood obesity disparities, the new treatment recommendations probably will widen the childhood obesity prevalence gap between the haves – who will benefit from the options available to reduce childhood obesity – and the have-nots, whose obesity rates will continue with their growth.
We live in a world of the haves and have-nots. This applies to financial resources as well as obesity rates. In the long term, the optimists hope that the GLP-1 medications will become ubiquitous, generics will be developed, and insurance companies will expand coverage and grant access to most children in need of effective obesity treatment options. Until this happens, unless intentional policies are promptly introduced, childhood obesity disparities will continue to widen.
To avoid the increasing disparities, brave and intentional actions are required. A lack of attention dealt to this known problem will result in a lost opportunity for the AAP, legislators, and others in a position to help U.S. children.
Liliana Aguayo, PhD, MPH, is assistant professor, Clinical Research Track, Hubert Department of Global Health, Emory University, Atlanta. A version of this article first appeared on Medscape.com.
In the United States, the Centers for Disease Control and Prevention estimates that nearly one in five children have obesity. Since the 1980s, the number of children with obesity has been increasing, with each generation reaching higher rates and greater weights at earlier ages. Even with extensive efforts from parents, clinicians, educators, and policymakers to limit the excessive weight gain among children, the number of obesity and severe obesity diagnoses keeps rising.
In response to this critical public health challenge, the American Academy of Pediatrics (AAP) introduced new clinical practice guidelines for the evaluation and management of obesity in children and adolescents. Developed by an expert panel, the new AAP guidelines present a departure in the conceptualization of obesity, recognizing the role that social determinants of health play in contributing to excessive weight gain.
As a community health researcher who investigates disparities in childhood obesity, I applaud the paradigm shift from the AAP. I specifically endorse the recognition that obesity is a very serious metabolic disease that won’t go away unless we introduce systemic changes and effective treatments.
However, I, like so many of my colleagues and anyone aware of the access barriers to the recommended treatments, worry about the consequences that the new guidelines will have in the context of current and future health disparities.
A recent study, published in Pediatrics, showed that childhood obesity disparities are widening. Younger generations of children are reaching higher weights at younger ages. These alarming trends are greater among Black children and children growing up with the greatest socioeconomic disadvantages. The new AAP guidelines – even while driven by good intentions – can exacerbate these differences and set children who are able to live healthy lives further apart from those with disproportionate obesity risks, who lack access to the treatments recommended by the AAP.
Rather than “watchful waiting,” to see if children outgrow obesity, the new guidelines call for “aggressive treatment,” as reported by this news organization. At least 26 hours of in-person intensive health behavior and lifestyle counseling and treatment are recommended for children aged 2 years old or older who meet the obesity criteria. For children aged 12 years or older, the AAP recommends complementing lifestyle counseling with pharmacotherapy. This breakthrough welcomes the use of promising antiobesity medications (for example, orlistat, Wegovy [semaglutide], Saxenda [liraglutide], Qsymia (phentermine and topiramate]) approved by the Food and Drug Administration for long-term use in children aged 12 and up. For children 13 years or older with severe obesity, bariatric surgery should be considered.
Will cost barriers continue to increase disparity?
The very promising semaglutide (Wegovy) is a GLP-1–based medication currently offered for about $1,000 per month. As with other chronic diseases, children should be prepared to take obesity medications for prolonged periods of time. A study conducted in adults found that when the medication is suspended, any weight loss can be regained. The costs of bariatric surgery total over $20,000.
In the U.S. health care system, at current prices, very few of the children in need of the medications or surgical treatments have access to them. Most private health insurance companies and Medicaid reject coverage for childhood obesity treatments. Barriers to treatment access are greater for Black and Hispanic children, children growing up in poverty, and children living in the U.S. South region, all of whom are more likely to develop obesity earlier in life than their White and wealthier counterparts.
The AAP recognized that a substantial time and financial commitment is required to follow the new treatment recommendations. Members of the AAP Expert Committee that developed the guidelines stated that they are “aware of the multitude of barriers to treatment that patients and their families face.”
Nevertheless, the recognition of the role of social determinants of health in the development of childhood obesity didn’t motivate the introduction of treatment options that aren’t unattainable for most U.S. families.
It’s important to step away from the conclusion that because of the price tag, at the population level, the new AAP guidelines will be inconsequential. This conclusion fails to recognize the potential harm that the guidelines may introduce. In the context of childhood obesity disparities, the new treatment recommendations probably will widen the childhood obesity prevalence gap between the haves – who will benefit from the options available to reduce childhood obesity – and the have-nots, whose obesity rates will continue with their growth.
We live in a world of the haves and have-nots. This applies to financial resources as well as obesity rates. In the long term, the optimists hope that the GLP-1 medications will become ubiquitous, generics will be developed, and insurance companies will expand coverage and grant access to most children in need of effective obesity treatment options. Until this happens, unless intentional policies are promptly introduced, childhood obesity disparities will continue to widen.
To avoid the increasing disparities, brave and intentional actions are required. A lack of attention dealt to this known problem will result in a lost opportunity for the AAP, legislators, and others in a position to help U.S. children.
Liliana Aguayo, PhD, MPH, is assistant professor, Clinical Research Track, Hubert Department of Global Health, Emory University, Atlanta. A version of this article first appeared on Medscape.com.
In the United States, the Centers for Disease Control and Prevention estimates that nearly one in five children have obesity. Since the 1980s, the number of children with obesity has been increasing, with each generation reaching higher rates and greater weights at earlier ages. Even with extensive efforts from parents, clinicians, educators, and policymakers to limit the excessive weight gain among children, the number of obesity and severe obesity diagnoses keeps rising.
In response to this critical public health challenge, the American Academy of Pediatrics (AAP) introduced new clinical practice guidelines for the evaluation and management of obesity in children and adolescents. Developed by an expert panel, the new AAP guidelines present a departure in the conceptualization of obesity, recognizing the role that social determinants of health play in contributing to excessive weight gain.
As a community health researcher who investigates disparities in childhood obesity, I applaud the paradigm shift from the AAP. I specifically endorse the recognition that obesity is a very serious metabolic disease that won’t go away unless we introduce systemic changes and effective treatments.
However, I, like so many of my colleagues and anyone aware of the access barriers to the recommended treatments, worry about the consequences that the new guidelines will have in the context of current and future health disparities.
A recent study, published in Pediatrics, showed that childhood obesity disparities are widening. Younger generations of children are reaching higher weights at younger ages. These alarming trends are greater among Black children and children growing up with the greatest socioeconomic disadvantages. The new AAP guidelines – even while driven by good intentions – can exacerbate these differences and set children who are able to live healthy lives further apart from those with disproportionate obesity risks, who lack access to the treatments recommended by the AAP.
Rather than “watchful waiting,” to see if children outgrow obesity, the new guidelines call for “aggressive treatment,” as reported by this news organization. At least 26 hours of in-person intensive health behavior and lifestyle counseling and treatment are recommended for children aged 2 years old or older who meet the obesity criteria. For children aged 12 years or older, the AAP recommends complementing lifestyle counseling with pharmacotherapy. This breakthrough welcomes the use of promising antiobesity medications (for example, orlistat, Wegovy [semaglutide], Saxenda [liraglutide], Qsymia (phentermine and topiramate]) approved by the Food and Drug Administration for long-term use in children aged 12 and up. For children 13 years or older with severe obesity, bariatric surgery should be considered.
Will cost barriers continue to increase disparity?
The very promising semaglutide (Wegovy) is a GLP-1–based medication currently offered for about $1,000 per month. As with other chronic diseases, children should be prepared to take obesity medications for prolonged periods of time. A study conducted in adults found that when the medication is suspended, any weight loss can be regained. The costs of bariatric surgery total over $20,000.
In the U.S. health care system, at current prices, very few of the children in need of the medications or surgical treatments have access to them. Most private health insurance companies and Medicaid reject coverage for childhood obesity treatments. Barriers to treatment access are greater for Black and Hispanic children, children growing up in poverty, and children living in the U.S. South region, all of whom are more likely to develop obesity earlier in life than their White and wealthier counterparts.
The AAP recognized that a substantial time and financial commitment is required to follow the new treatment recommendations. Members of the AAP Expert Committee that developed the guidelines stated that they are “aware of the multitude of barriers to treatment that patients and their families face.”
Nevertheless, the recognition of the role of social determinants of health in the development of childhood obesity didn’t motivate the introduction of treatment options that aren’t unattainable for most U.S. families.
It’s important to step away from the conclusion that because of the price tag, at the population level, the new AAP guidelines will be inconsequential. This conclusion fails to recognize the potential harm that the guidelines may introduce. In the context of childhood obesity disparities, the new treatment recommendations probably will widen the childhood obesity prevalence gap between the haves – who will benefit from the options available to reduce childhood obesity – and the have-nots, whose obesity rates will continue with their growth.
We live in a world of the haves and have-nots. This applies to financial resources as well as obesity rates. In the long term, the optimists hope that the GLP-1 medications will become ubiquitous, generics will be developed, and insurance companies will expand coverage and grant access to most children in need of effective obesity treatment options. Until this happens, unless intentional policies are promptly introduced, childhood obesity disparities will continue to widen.
To avoid the increasing disparities, brave and intentional actions are required. A lack of attention dealt to this known problem will result in a lost opportunity for the AAP, legislators, and others in a position to help U.S. children.
Liliana Aguayo, PhD, MPH, is assistant professor, Clinical Research Track, Hubert Department of Global Health, Emory University, Atlanta. A version of this article first appeared on Medscape.com.
Firing patients
One might assume that, just as patients are free to accept or reject their doctors, physicians have an equal right to reject their patients; to a certain extent, that is true. There are no specific laws prohibiting a provider from terminating a patient relationship for any reason, other than a discriminatory one – race, nationality, religion, age, gender, sexual orientation, and so on. However, the evolution of ever-larger practice environments has raised new questions.
While verbal abuse, inappropriate treatment demands (particularly for controlled substances), refusal to adhere to mutually agreed treatment plans, and failure to keep appointments or pay bills remain the most common reasons for dismissal, evolving practice environments may require us to modify our responses.
What happens, for example, when a patient is banned from a large clinic that employs most of that community’s physicians, or is the only practice in town with the specialists required by that patient? The medical profession does have an obligation to not exclude such patients from care.
In a large cross-specialty system or consolidated specialist practice, firing a patient has a very different level of consequences than in a small office. There must be a balance between separating patients and doctors who don’t get along and seeing that the patient in question receives competent treatment. The physician, as the professional, has a higher standard to live up to with respect to handling this kind of situation.
If the problem is a personality conflict, the solution may be as simple as transferring the patient to another caregiver within the practice. While it does not make sense for a patient to continue seeing a doctor who does not want to see them, it also does not make sense to ban a patient from a large system where there could well be one or more other doctors who would be a good match. If a patient is unable to pay outstanding bills, a large clinic might prohibit them from making new appointments until they have worked out a payment plan rather than firing them outright.
If you are part of a large practice, take the time to research your group’s official policies for dealing with such situations. If there is no written policy, you might want to start that discussion with your colleagues.
The point is that in any practice, large or small, firing a patient should be a last resort. Try to make every effort to resolve the problem amicably. Communicate with the patient in question, explain your concerns, and discuss options for resolution. Take time to listen to the patient, as they may have an explanation (rational or not) for their objectionable behavior.
You can also send a letter, repeating your concerns and proposed solutions, as further documentation of your efforts to achieve an amicable resolution. All verbal and written warnings must, of course, be documented. If the patient has a managed care policy, we review the managed care contract, which sometimes includes specific requirements for dismissal of its patients.
When such efforts fail, we send the patient two letters – one certified with return receipt, the other by conventional first class, in case the patient refuses the certified copy – explaining the reason for dismissal, and that care will be discontinued in 30 days from the letter’s date. (Most attorneys and medical associations agree that 30 days is sufficient reasonable notice.) We offer to provide care during the interim period, include a list of names and contact information for potential alternate providers, and offer to transfer records after receiving written permission.
Following these precautions will usually protect you from charges of “patient abandonment,” which is generally defined as the unilateral severance by the physician of the physician-patient relationship without giving the patient sufficient advance notice to obtain the services of another practitioner, and at a time when the patient still requires medical attention.
Some states have their own unique definitions of patient abandonment. You should check with your state’s health department, and your attorney, for any unusual requirements in your state, because violating them could lead to intervention by your state licensing board. There is also the risk of civil litigation, which is typically not covered by malpractice policies, and may not be covered by your general liability policy either.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
One might assume that, just as patients are free to accept or reject their doctors, physicians have an equal right to reject their patients; to a certain extent, that is true. There are no specific laws prohibiting a provider from terminating a patient relationship for any reason, other than a discriminatory one – race, nationality, religion, age, gender, sexual orientation, and so on. However, the evolution of ever-larger practice environments has raised new questions.
While verbal abuse, inappropriate treatment demands (particularly for controlled substances), refusal to adhere to mutually agreed treatment plans, and failure to keep appointments or pay bills remain the most common reasons for dismissal, evolving practice environments may require us to modify our responses.
What happens, for example, when a patient is banned from a large clinic that employs most of that community’s physicians, or is the only practice in town with the specialists required by that patient? The medical profession does have an obligation to not exclude such patients from care.
In a large cross-specialty system or consolidated specialist practice, firing a patient has a very different level of consequences than in a small office. There must be a balance between separating patients and doctors who don’t get along and seeing that the patient in question receives competent treatment. The physician, as the professional, has a higher standard to live up to with respect to handling this kind of situation.
If the problem is a personality conflict, the solution may be as simple as transferring the patient to another caregiver within the practice. While it does not make sense for a patient to continue seeing a doctor who does not want to see them, it also does not make sense to ban a patient from a large system where there could well be one or more other doctors who would be a good match. If a patient is unable to pay outstanding bills, a large clinic might prohibit them from making new appointments until they have worked out a payment plan rather than firing them outright.
If you are part of a large practice, take the time to research your group’s official policies for dealing with such situations. If there is no written policy, you might want to start that discussion with your colleagues.
The point is that in any practice, large or small, firing a patient should be a last resort. Try to make every effort to resolve the problem amicably. Communicate with the patient in question, explain your concerns, and discuss options for resolution. Take time to listen to the patient, as they may have an explanation (rational or not) for their objectionable behavior.
You can also send a letter, repeating your concerns and proposed solutions, as further documentation of your efforts to achieve an amicable resolution. All verbal and written warnings must, of course, be documented. If the patient has a managed care policy, we review the managed care contract, which sometimes includes specific requirements for dismissal of its patients.
When such efforts fail, we send the patient two letters – one certified with return receipt, the other by conventional first class, in case the patient refuses the certified copy – explaining the reason for dismissal, and that care will be discontinued in 30 days from the letter’s date. (Most attorneys and medical associations agree that 30 days is sufficient reasonable notice.) We offer to provide care during the interim period, include a list of names and contact information for potential alternate providers, and offer to transfer records after receiving written permission.
Following these precautions will usually protect you from charges of “patient abandonment,” which is generally defined as the unilateral severance by the physician of the physician-patient relationship without giving the patient sufficient advance notice to obtain the services of another practitioner, and at a time when the patient still requires medical attention.
Some states have their own unique definitions of patient abandonment. You should check with your state’s health department, and your attorney, for any unusual requirements in your state, because violating them could lead to intervention by your state licensing board. There is also the risk of civil litigation, which is typically not covered by malpractice policies, and may not be covered by your general liability policy either.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
One might assume that, just as patients are free to accept or reject their doctors, physicians have an equal right to reject their patients; to a certain extent, that is true. There are no specific laws prohibiting a provider from terminating a patient relationship for any reason, other than a discriminatory one – race, nationality, religion, age, gender, sexual orientation, and so on. However, the evolution of ever-larger practice environments has raised new questions.
While verbal abuse, inappropriate treatment demands (particularly for controlled substances), refusal to adhere to mutually agreed treatment plans, and failure to keep appointments or pay bills remain the most common reasons for dismissal, evolving practice environments may require us to modify our responses.
What happens, for example, when a patient is banned from a large clinic that employs most of that community’s physicians, or is the only practice in town with the specialists required by that patient? The medical profession does have an obligation to not exclude such patients from care.
In a large cross-specialty system or consolidated specialist practice, firing a patient has a very different level of consequences than in a small office. There must be a balance between separating patients and doctors who don’t get along and seeing that the patient in question receives competent treatment. The physician, as the professional, has a higher standard to live up to with respect to handling this kind of situation.
If the problem is a personality conflict, the solution may be as simple as transferring the patient to another caregiver within the practice. While it does not make sense for a patient to continue seeing a doctor who does not want to see them, it also does not make sense to ban a patient from a large system where there could well be one or more other doctors who would be a good match. If a patient is unable to pay outstanding bills, a large clinic might prohibit them from making new appointments until they have worked out a payment plan rather than firing them outright.
If you are part of a large practice, take the time to research your group’s official policies for dealing with such situations. If there is no written policy, you might want to start that discussion with your colleagues.
The point is that in any practice, large or small, firing a patient should be a last resort. Try to make every effort to resolve the problem amicably. Communicate with the patient in question, explain your concerns, and discuss options for resolution. Take time to listen to the patient, as they may have an explanation (rational or not) for their objectionable behavior.
You can also send a letter, repeating your concerns and proposed solutions, as further documentation of your efforts to achieve an amicable resolution. All verbal and written warnings must, of course, be documented. If the patient has a managed care policy, we review the managed care contract, which sometimes includes specific requirements for dismissal of its patients.
When such efforts fail, we send the patient two letters – one certified with return receipt, the other by conventional first class, in case the patient refuses the certified copy – explaining the reason for dismissal, and that care will be discontinued in 30 days from the letter’s date. (Most attorneys and medical associations agree that 30 days is sufficient reasonable notice.) We offer to provide care during the interim period, include a list of names and contact information for potential alternate providers, and offer to transfer records after receiving written permission.
Following these precautions will usually protect you from charges of “patient abandonment,” which is generally defined as the unilateral severance by the physician of the physician-patient relationship without giving the patient sufficient advance notice to obtain the services of another practitioner, and at a time when the patient still requires medical attention.
Some states have their own unique definitions of patient abandonment. You should check with your state’s health department, and your attorney, for any unusual requirements in your state, because violating them could lead to intervention by your state licensing board. There is also the risk of civil litigation, which is typically not covered by malpractice policies, and may not be covered by your general liability policy either.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
New clinical guideline for biliary strictures issued
The recommendations provide guidance on the care of patients with extrahepatic and perihilar strictures, with a focus on diagnosis and drainage. Although some of the principles may apply to intrahepatic strictures, the guideline doesn’t specifically address them. The new guideline is considered separate from the 2015 ACG guideline related to primary sclerosing cholangitis.
“The appropriate diagnosis and management of biliary strictures is still a big clinical challenge and has important implications in endoscopic, surgical, and oncological decision-making,” co-author Jennifer Maranki, MD, a professor of medicine and director of endoscopy at Penn State Hershey Medical Center, said in an interview.
“We wanted to provide the best possible guidance to gastroenterologists based on the available body of literature, with key shifts in diagnosis and management based on currently available modalities and tools,” she said.
The guideline was published in the March issue of the American Journal of Gastroenterology.
The recommendations were developed by a diverse group of authors from across the United States in recognition of the potential influence of commercial and intellectual conflicts of interest. The panel used a systematic process that involved structured literature searches by librarians and independent appraisal of the quality of evidence by dedicated methodologists, the authors write.
Overall, the team outlined 11 recommendations and 12 key concepts. A strong recommendation was made when the benefits of the test or intervention clearly outweighed the potential disadvantages. A conditional recommendation was made when some uncertainty remained about the balance of benefits and harms. Key concepts address important clinical questions that lack adequate evidence to inform recommendations. They are based on indirect evidence and expert opinion.
Epidemiology and diagnosis
The burden of biliary strictures is difficult to estimate, owing to the lack of a specific administrative code. The estimated cost of caring for biliary disease in the United States is about $16.9 billion annually, although this figure includes costs associated with gallbladder disease, choledocholithiasis, and other (nonobstructive) biliary disorders, the authors write.
Among the 57,000 new cases of pancreatic cancer each year, at least 60% will cause obstructive jaundice, resulting in about 34,000 annual cases of malignant extrahepatic biliary stricture, the team notes. In addition, about 3,000 cases of malignant perihilar stricture are expected in the United States each year. Patients may also seek care for benign strictures associated with chronic pancreatitis, primary sclerosing cholangitis, autoimmune disease, and post-cholecystectomy injury.
Under the first key concept, the authors note that biliary strictures in adults are more likely to be malignant than benign, except in certain well-defined scenarios. This underscores the importance of having a high index of clinical suspicion during evaluation, they add.
In general, a definitive tissue diagnosis is necessary to guide oncologic and endoscopic care for most strictures that aren’t surgically resectable at the time of presentation. For patients with extrahepatic biliary stricture due to an apparent or suspected pancreatic mass, endoscopic ultrasound (EUS) with fine-needle sampling (aspiration or biopsy) is recommended over endoscopic retrograde cholangiopancreatography (ERCP) as the preferred method of evaluation for malignancy.
For patients with suspected malignant perihilar stricture, multimodality sampling is recommended over brush cytology alone at the time of the index ERCP.
Guidance on drainage
For management, the principal objective is to restore the physiologic flow of bile into the duodenum. Although there is wide variability in the difficulty and risk of drainage, depending on location and complexity, perihilar strictures are generally more challenging and are riskier to drain than extrahepatic strictures. The goals should be to alleviate symptoms, reduce serum bilirubin to a level such that chemotherapy can be safely administered, and optimize surgical outcomes.
For benign extrahepatic biliary strictures, ERCP is the preferred modality for durable treatment. Fully covered self-expanding metallic stent (SEMS) placement is recommended over multiple plastic stents to reduce the number of procedures required for long-term treatment.
For extrahepatic strictures due to resectable pancreatic cancer or cholangiocarcinoma, the authors recommend against routine preoperative biliary drainage. However, drainage is warranted for some patients, including those with acute cholangitis, severe pruritus, very high serum bilirubin levels, those undergoing neoadjuvant therapy, and those for whom surgery is delayed.
For malignant extrahepatic strictures that are unresectable or borderline resectable, SEMS placement is recommended over plastic stents. The evidence is insufficient to recommend for or against uncovered SEMS versus fully covered SEMS.
For perihilar strictures due to suspected malignancy, the evidence is insufficient to recommend for or against ERCP versus percutaneous transhepatic biliary drainage. In addition, for malignant perihilar strictures, the evidence is insufficient to recommend for or against plastic stents versus uncovered SEMS.
For perihilar strictures due to cholangiocarcinoma in cases in which resection or transplantation is not possible, adjuvant endobiliary ablation plus plastic stent placement is recommended over plastic stent placement alone.
Overall, for patients with a biliary stricture for which ERCP is indicated but is unsuccessful or impossible, EUS-guided biliary access and drainage is recommended over PTBD, because it is associated with fewer adverse events. However, these interventional EUS procedures should be performed by an endoscopist with substantial experience.
“The workup of biliary strictures is challenging, invasive, and costly, requiring multiple diagnostic tools with highly variable yields,” co-author Victoria Gomez, MD, associate professor of medicine and director of bariatric endoscopy at Mayo Clinic, Jacksonville, Fla., said in an interview.
“Providers caring for these patients must be up to date with the most current evidence so that they can make the safest and most well-informed decisions for their patients,” she said. “These include considerations such as limiting the use of anesthesia, using tests that will result in the highest diagnostic yield, and providing effective therapies to decompress biliary obstruction.”
Future questions
Additional research is needed in several areas to strengthen recommendations and advance the field, the study authors write.
“Biliary strictures without an associated mass are a diagnostic challenge, and there are exciting opportunities to understand how new technologies, such as artificial intelligence, can be used to improve our assessment,” co-author Anna Tavakkoli, MD, assistant professor of internal medicine in digestive and liver diseases at the University of Texas Southwestern Medical Center, Dallas, said in an interview.
“Also, we highlighted several controversies in the drainage of perihilar strictures, including whether to use ERCP versus percutaneous drainage, whether metallic or plastic stents are better, and what the optimal stent placement should be,” she said. “Future multicenter studies are needed to address these key controversies.”
Although fully covered SEMS placement remains effective for benign biliary strictures, multiple plastic stents may be a better alternative in some cases. Such cases include those in which the stricture is close to the hilum, those in which the gallbladder is intact and in which crossing the cystic duct orifice cannot be avoided, those in which a fully covered SEMS has previously migrated or was not well tolerated, and those in which stricture has recurred after removal of a fully covered SEMS.
‘Comprehensive list’
“Overall, the authors have done a commendable job putting together a comprehensive list of recommendations that will invariably alter the practice of many therapeutic endoscopists for the diagnosis and management of biliary strictures,” Matthew Fasullo, DO, an advanced endoscopy and gastroenterology fellow at New York University Medical Center, told this news organization.
Dr. Fasullo, who wasn’t involved with the guideline, has published on advances in pathophysiology, diagnosis, and treatment for post-transplant biliary complications.
“The fact that ... cholangioscopy-directed biopsies after an initial negative evaluation via ERCP reveal malignancy in 54% of cases underscores the need for best practice guidelines and supports advancements in diagnostics to confidently rule in or out cancer,” he said.
“The movement toward multimodality sampling at the time of initial evaluation with a combination of brushing, fluoroscopy-directed biopsies, cholangioscopy-directed biopsies, and fluorescence in situ hybridization should become universally adopted in those with an ambiguous diagnosis,” he added. “As technology continues to improve, next-generation sequencing will prove to be an invaluable adjunct to the current pathological evaluation.”
The authors received no financial support for the guideline. One author has a consultant role for Takeda Pharmaceuticals and is an advisory board member role for Advarra. The other authors and Dr. Fasullo have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The recommendations provide guidance on the care of patients with extrahepatic and perihilar strictures, with a focus on diagnosis and drainage. Although some of the principles may apply to intrahepatic strictures, the guideline doesn’t specifically address them. The new guideline is considered separate from the 2015 ACG guideline related to primary sclerosing cholangitis.
“The appropriate diagnosis and management of biliary strictures is still a big clinical challenge and has important implications in endoscopic, surgical, and oncological decision-making,” co-author Jennifer Maranki, MD, a professor of medicine and director of endoscopy at Penn State Hershey Medical Center, said in an interview.
“We wanted to provide the best possible guidance to gastroenterologists based on the available body of literature, with key shifts in diagnosis and management based on currently available modalities and tools,” she said.
The guideline was published in the March issue of the American Journal of Gastroenterology.
The recommendations were developed by a diverse group of authors from across the United States in recognition of the potential influence of commercial and intellectual conflicts of interest. The panel used a systematic process that involved structured literature searches by librarians and independent appraisal of the quality of evidence by dedicated methodologists, the authors write.
Overall, the team outlined 11 recommendations and 12 key concepts. A strong recommendation was made when the benefits of the test or intervention clearly outweighed the potential disadvantages. A conditional recommendation was made when some uncertainty remained about the balance of benefits and harms. Key concepts address important clinical questions that lack adequate evidence to inform recommendations. They are based on indirect evidence and expert opinion.
Epidemiology and diagnosis
The burden of biliary strictures is difficult to estimate, owing to the lack of a specific administrative code. The estimated cost of caring for biliary disease in the United States is about $16.9 billion annually, although this figure includes costs associated with gallbladder disease, choledocholithiasis, and other (nonobstructive) biliary disorders, the authors write.
Among the 57,000 new cases of pancreatic cancer each year, at least 60% will cause obstructive jaundice, resulting in about 34,000 annual cases of malignant extrahepatic biliary stricture, the team notes. In addition, about 3,000 cases of malignant perihilar stricture are expected in the United States each year. Patients may also seek care for benign strictures associated with chronic pancreatitis, primary sclerosing cholangitis, autoimmune disease, and post-cholecystectomy injury.
Under the first key concept, the authors note that biliary strictures in adults are more likely to be malignant than benign, except in certain well-defined scenarios. This underscores the importance of having a high index of clinical suspicion during evaluation, they add.
In general, a definitive tissue diagnosis is necessary to guide oncologic and endoscopic care for most strictures that aren’t surgically resectable at the time of presentation. For patients with extrahepatic biliary stricture due to an apparent or suspected pancreatic mass, endoscopic ultrasound (EUS) with fine-needle sampling (aspiration or biopsy) is recommended over endoscopic retrograde cholangiopancreatography (ERCP) as the preferred method of evaluation for malignancy.
For patients with suspected malignant perihilar stricture, multimodality sampling is recommended over brush cytology alone at the time of the index ERCP.
Guidance on drainage
For management, the principal objective is to restore the physiologic flow of bile into the duodenum. Although there is wide variability in the difficulty and risk of drainage, depending on location and complexity, perihilar strictures are generally more challenging and are riskier to drain than extrahepatic strictures. The goals should be to alleviate symptoms, reduce serum bilirubin to a level such that chemotherapy can be safely administered, and optimize surgical outcomes.
For benign extrahepatic biliary strictures, ERCP is the preferred modality for durable treatment. Fully covered self-expanding metallic stent (SEMS) placement is recommended over multiple plastic stents to reduce the number of procedures required for long-term treatment.
For extrahepatic strictures due to resectable pancreatic cancer or cholangiocarcinoma, the authors recommend against routine preoperative biliary drainage. However, drainage is warranted for some patients, including those with acute cholangitis, severe pruritus, very high serum bilirubin levels, those undergoing neoadjuvant therapy, and those for whom surgery is delayed.
For malignant extrahepatic strictures that are unresectable or borderline resectable, SEMS placement is recommended over plastic stents. The evidence is insufficient to recommend for or against uncovered SEMS versus fully covered SEMS.
For perihilar strictures due to suspected malignancy, the evidence is insufficient to recommend for or against ERCP versus percutaneous transhepatic biliary drainage. In addition, for malignant perihilar strictures, the evidence is insufficient to recommend for or against plastic stents versus uncovered SEMS.
For perihilar strictures due to cholangiocarcinoma in cases in which resection or transplantation is not possible, adjuvant endobiliary ablation plus plastic stent placement is recommended over plastic stent placement alone.
Overall, for patients with a biliary stricture for which ERCP is indicated but is unsuccessful or impossible, EUS-guided biliary access and drainage is recommended over PTBD, because it is associated with fewer adverse events. However, these interventional EUS procedures should be performed by an endoscopist with substantial experience.
“The workup of biliary strictures is challenging, invasive, and costly, requiring multiple diagnostic tools with highly variable yields,” co-author Victoria Gomez, MD, associate professor of medicine and director of bariatric endoscopy at Mayo Clinic, Jacksonville, Fla., said in an interview.
“Providers caring for these patients must be up to date with the most current evidence so that they can make the safest and most well-informed decisions for their patients,” she said. “These include considerations such as limiting the use of anesthesia, using tests that will result in the highest diagnostic yield, and providing effective therapies to decompress biliary obstruction.”
Future questions
Additional research is needed in several areas to strengthen recommendations and advance the field, the study authors write.
“Biliary strictures without an associated mass are a diagnostic challenge, and there are exciting opportunities to understand how new technologies, such as artificial intelligence, can be used to improve our assessment,” co-author Anna Tavakkoli, MD, assistant professor of internal medicine in digestive and liver diseases at the University of Texas Southwestern Medical Center, Dallas, said in an interview.
“Also, we highlighted several controversies in the drainage of perihilar strictures, including whether to use ERCP versus percutaneous drainage, whether metallic or plastic stents are better, and what the optimal stent placement should be,” she said. “Future multicenter studies are needed to address these key controversies.”
Although fully covered SEMS placement remains effective for benign biliary strictures, multiple plastic stents may be a better alternative in some cases. Such cases include those in which the stricture is close to the hilum, those in which the gallbladder is intact and in which crossing the cystic duct orifice cannot be avoided, those in which a fully covered SEMS has previously migrated or was not well tolerated, and those in which stricture has recurred after removal of a fully covered SEMS.
‘Comprehensive list’
“Overall, the authors have done a commendable job putting together a comprehensive list of recommendations that will invariably alter the practice of many therapeutic endoscopists for the diagnosis and management of biliary strictures,” Matthew Fasullo, DO, an advanced endoscopy and gastroenterology fellow at New York University Medical Center, told this news organization.
Dr. Fasullo, who wasn’t involved with the guideline, has published on advances in pathophysiology, diagnosis, and treatment for post-transplant biliary complications.
“The fact that ... cholangioscopy-directed biopsies after an initial negative evaluation via ERCP reveal malignancy in 54% of cases underscores the need for best practice guidelines and supports advancements in diagnostics to confidently rule in or out cancer,” he said.
“The movement toward multimodality sampling at the time of initial evaluation with a combination of brushing, fluoroscopy-directed biopsies, cholangioscopy-directed biopsies, and fluorescence in situ hybridization should become universally adopted in those with an ambiguous diagnosis,” he added. “As technology continues to improve, next-generation sequencing will prove to be an invaluable adjunct to the current pathological evaluation.”
The authors received no financial support for the guideline. One author has a consultant role for Takeda Pharmaceuticals and is an advisory board member role for Advarra. The other authors and Dr. Fasullo have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The recommendations provide guidance on the care of patients with extrahepatic and perihilar strictures, with a focus on diagnosis and drainage. Although some of the principles may apply to intrahepatic strictures, the guideline doesn’t specifically address them. The new guideline is considered separate from the 2015 ACG guideline related to primary sclerosing cholangitis.
“The appropriate diagnosis and management of biliary strictures is still a big clinical challenge and has important implications in endoscopic, surgical, and oncological decision-making,” co-author Jennifer Maranki, MD, a professor of medicine and director of endoscopy at Penn State Hershey Medical Center, said in an interview.
“We wanted to provide the best possible guidance to gastroenterologists based on the available body of literature, with key shifts in diagnosis and management based on currently available modalities and tools,” she said.
The guideline was published in the March issue of the American Journal of Gastroenterology.
The recommendations were developed by a diverse group of authors from across the United States in recognition of the potential influence of commercial and intellectual conflicts of interest. The panel used a systematic process that involved structured literature searches by librarians and independent appraisal of the quality of evidence by dedicated methodologists, the authors write.
Overall, the team outlined 11 recommendations and 12 key concepts. A strong recommendation was made when the benefits of the test or intervention clearly outweighed the potential disadvantages. A conditional recommendation was made when some uncertainty remained about the balance of benefits and harms. Key concepts address important clinical questions that lack adequate evidence to inform recommendations. They are based on indirect evidence and expert opinion.
Epidemiology and diagnosis
The burden of biliary strictures is difficult to estimate, owing to the lack of a specific administrative code. The estimated cost of caring for biliary disease in the United States is about $16.9 billion annually, although this figure includes costs associated with gallbladder disease, choledocholithiasis, and other (nonobstructive) biliary disorders, the authors write.
Among the 57,000 new cases of pancreatic cancer each year, at least 60% will cause obstructive jaundice, resulting in about 34,000 annual cases of malignant extrahepatic biliary stricture, the team notes. In addition, about 3,000 cases of malignant perihilar stricture are expected in the United States each year. Patients may also seek care for benign strictures associated with chronic pancreatitis, primary sclerosing cholangitis, autoimmune disease, and post-cholecystectomy injury.
Under the first key concept, the authors note that biliary strictures in adults are more likely to be malignant than benign, except in certain well-defined scenarios. This underscores the importance of having a high index of clinical suspicion during evaluation, they add.
In general, a definitive tissue diagnosis is necessary to guide oncologic and endoscopic care for most strictures that aren’t surgically resectable at the time of presentation. For patients with extrahepatic biliary stricture due to an apparent or suspected pancreatic mass, endoscopic ultrasound (EUS) with fine-needle sampling (aspiration or biopsy) is recommended over endoscopic retrograde cholangiopancreatography (ERCP) as the preferred method of evaluation for malignancy.
For patients with suspected malignant perihilar stricture, multimodality sampling is recommended over brush cytology alone at the time of the index ERCP.
Guidance on drainage
For management, the principal objective is to restore the physiologic flow of bile into the duodenum. Although there is wide variability in the difficulty and risk of drainage, depending on location and complexity, perihilar strictures are generally more challenging and are riskier to drain than extrahepatic strictures. The goals should be to alleviate symptoms, reduce serum bilirubin to a level such that chemotherapy can be safely administered, and optimize surgical outcomes.
For benign extrahepatic biliary strictures, ERCP is the preferred modality for durable treatment. Fully covered self-expanding metallic stent (SEMS) placement is recommended over multiple plastic stents to reduce the number of procedures required for long-term treatment.
For extrahepatic strictures due to resectable pancreatic cancer or cholangiocarcinoma, the authors recommend against routine preoperative biliary drainage. However, drainage is warranted for some patients, including those with acute cholangitis, severe pruritus, very high serum bilirubin levels, those undergoing neoadjuvant therapy, and those for whom surgery is delayed.
For malignant extrahepatic strictures that are unresectable or borderline resectable, SEMS placement is recommended over plastic stents. The evidence is insufficient to recommend for or against uncovered SEMS versus fully covered SEMS.
For perihilar strictures due to suspected malignancy, the evidence is insufficient to recommend for or against ERCP versus percutaneous transhepatic biliary drainage. In addition, for malignant perihilar strictures, the evidence is insufficient to recommend for or against plastic stents versus uncovered SEMS.
For perihilar strictures due to cholangiocarcinoma in cases in which resection or transplantation is not possible, adjuvant endobiliary ablation plus plastic stent placement is recommended over plastic stent placement alone.
Overall, for patients with a biliary stricture for which ERCP is indicated but is unsuccessful or impossible, EUS-guided biliary access and drainage is recommended over PTBD, because it is associated with fewer adverse events. However, these interventional EUS procedures should be performed by an endoscopist with substantial experience.
“The workup of biliary strictures is challenging, invasive, and costly, requiring multiple diagnostic tools with highly variable yields,” co-author Victoria Gomez, MD, associate professor of medicine and director of bariatric endoscopy at Mayo Clinic, Jacksonville, Fla., said in an interview.
“Providers caring for these patients must be up to date with the most current evidence so that they can make the safest and most well-informed decisions for their patients,” she said. “These include considerations such as limiting the use of anesthesia, using tests that will result in the highest diagnostic yield, and providing effective therapies to decompress biliary obstruction.”
Future questions
Additional research is needed in several areas to strengthen recommendations and advance the field, the study authors write.
“Biliary strictures without an associated mass are a diagnostic challenge, and there are exciting opportunities to understand how new technologies, such as artificial intelligence, can be used to improve our assessment,” co-author Anna Tavakkoli, MD, assistant professor of internal medicine in digestive and liver diseases at the University of Texas Southwestern Medical Center, Dallas, said in an interview.
“Also, we highlighted several controversies in the drainage of perihilar strictures, including whether to use ERCP versus percutaneous drainage, whether metallic or plastic stents are better, and what the optimal stent placement should be,” she said. “Future multicenter studies are needed to address these key controversies.”
Although fully covered SEMS placement remains effective for benign biliary strictures, multiple plastic stents may be a better alternative in some cases. Such cases include those in which the stricture is close to the hilum, those in which the gallbladder is intact and in which crossing the cystic duct orifice cannot be avoided, those in which a fully covered SEMS has previously migrated or was not well tolerated, and those in which stricture has recurred after removal of a fully covered SEMS.
‘Comprehensive list’
“Overall, the authors have done a commendable job putting together a comprehensive list of recommendations that will invariably alter the practice of many therapeutic endoscopists for the diagnosis and management of biliary strictures,” Matthew Fasullo, DO, an advanced endoscopy and gastroenterology fellow at New York University Medical Center, told this news organization.
Dr. Fasullo, who wasn’t involved with the guideline, has published on advances in pathophysiology, diagnosis, and treatment for post-transplant biliary complications.
“The fact that ... cholangioscopy-directed biopsies after an initial negative evaluation via ERCP reveal malignancy in 54% of cases underscores the need for best practice guidelines and supports advancements in diagnostics to confidently rule in or out cancer,” he said.
“The movement toward multimodality sampling at the time of initial evaluation with a combination of brushing, fluoroscopy-directed biopsies, cholangioscopy-directed biopsies, and fluorescence in situ hybridization should become universally adopted in those with an ambiguous diagnosis,” he added. “As technology continues to improve, next-generation sequencing will prove to be an invaluable adjunct to the current pathological evaluation.”
The authors received no financial support for the guideline. One author has a consultant role for Takeda Pharmaceuticals and is an advisory board member role for Advarra. The other authors and Dr. Fasullo have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Nearly one in three patients with IBD affected by skin lesions
People with inflammatory bowel disease (IBD) commonly develop skin lesions linked to their condition, but until now few researchers looked at how common they are.
, according to the prospective, single-center study.
“Skin lesions in IBD patients are much more prevalent than it is generally accepted. The lesions may be related to the pathogenesis of IBD, but it is very important to know that the modern biological therapies may also cause skin lesions,” said senior study author Laimas Jonaitis, MD, PhD, professor in the department of gastroenterology at Lithuanian University of Health Sciences in Kaunas.
“If the gastroenterologist is experienced and has enough competence, he or she may establish the diagnosis, but in all other cases it is wise and advisable to refer the patient to the dermatologist,” Dr. Jonaitis said. A referral should include the history and full treatment for IBD.
The results were presented as a poster at the annual congress of the European Crohn’s and Colitis Organisation, held in Copenhagen and virtually.
Dr. Jonaitis and colleagues conducted a literature analysis to determine the prevalence of extra-abdominal manifestations of IBD. The lack of published data prompted them to survey 152 consecutive patients with IBD receiving outpatient treatment at their institution. The patients completed questionnaires from January to October 2022 about any cutaneous lesions.
The mean age of patients was 42 years, and 58% were men. A majority, 72%, had ulcerative colitis, and 28% had Crohn’s disease.
Prevalence of skin lesions
A total of 43% of participants reported skin lesions, but only 30% of patients had lesions considered related to IBD or IBD therapy due to their emergence after the patient’s IBD diagnosis.
By IBD diagnosis, 29% of patients with ulcerative colitis and 33% of patients with Crohn’s disease had lesions related to their condition. The difference in skin lesion prevalence between the two groups was not significant (P > .05), the researchers noted.
The team further investigated the types of skin lesions deemed to be associated with IBD or IBD therapy.
Overall, they found psoriasis in nine patients, eczema in nine, erythema nodosum in six, pyoderma gangrenosum in five, allergic rash in four, and vitiligo in two. They found acne, epidermolysis bullosa acquisita, and hemorrhagic vasculitis in one patient each.
Specifically, among patients with ulcerative colitis, skin lesions were reported in 8 of 27 with left-sided colitis, 2 of 15 with ulcerative colitis proctitis, and 22 of 67 patients with pancolitis. The difference between the groups of proctitis and pancolitis was significant (P = .03).
Within the group with Crohn’s disease, skin lesions were reported in 3 of 15 patients with ileitis, 4 of 10 with colitis, and 7 of 17 with ileocolitis. The difference among these groups was not significant (P > .05).
The most common skin lesions observed in Crohn’s disease were erythema nodosum and eczema, and in ulcerative colitis, psoriasis and eczema, the researchers reported.
They also noted that the cutaneous lesions were significantly more prevalent in extensive ulcerative colitis compared with distal disease.
Skin lesions add to patient misery
“Skin lesions are considered a burden to patients with IBD and add to their suffering,” said Sara Mesilhy, MBBS, a gastroenterologist with the Royal College of Physicians in the United Kingdom, who was not affiliated with the research.
The severity and location of the disease appears to play a role because researchers found extensive ulcerative colitis may carry a higher risk for the development of skin lesions, Dr. Mesilhy noted.
The first step when facing skin lesions is to control the disease activity via the best treatment option, Dr. Mesilhy suggested.
The study was independently supported. Dr. Jonaitis and Dr. Mesilhy have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
People with inflammatory bowel disease (IBD) commonly develop skin lesions linked to their condition, but until now few researchers looked at how common they are.
, according to the prospective, single-center study.
“Skin lesions in IBD patients are much more prevalent than it is generally accepted. The lesions may be related to the pathogenesis of IBD, but it is very important to know that the modern biological therapies may also cause skin lesions,” said senior study author Laimas Jonaitis, MD, PhD, professor in the department of gastroenterology at Lithuanian University of Health Sciences in Kaunas.
“If the gastroenterologist is experienced and has enough competence, he or she may establish the diagnosis, but in all other cases it is wise and advisable to refer the patient to the dermatologist,” Dr. Jonaitis said. A referral should include the history and full treatment for IBD.
The results were presented as a poster at the annual congress of the European Crohn’s and Colitis Organisation, held in Copenhagen and virtually.
Dr. Jonaitis and colleagues conducted a literature analysis to determine the prevalence of extra-abdominal manifestations of IBD. The lack of published data prompted them to survey 152 consecutive patients with IBD receiving outpatient treatment at their institution. The patients completed questionnaires from January to October 2022 about any cutaneous lesions.
The mean age of patients was 42 years, and 58% were men. A majority, 72%, had ulcerative colitis, and 28% had Crohn’s disease.
Prevalence of skin lesions
A total of 43% of participants reported skin lesions, but only 30% of patients had lesions considered related to IBD or IBD therapy due to their emergence after the patient’s IBD diagnosis.
By IBD diagnosis, 29% of patients with ulcerative colitis and 33% of patients with Crohn’s disease had lesions related to their condition. The difference in skin lesion prevalence between the two groups was not significant (P > .05), the researchers noted.
The team further investigated the types of skin lesions deemed to be associated with IBD or IBD therapy.
Overall, they found psoriasis in nine patients, eczema in nine, erythema nodosum in six, pyoderma gangrenosum in five, allergic rash in four, and vitiligo in two. They found acne, epidermolysis bullosa acquisita, and hemorrhagic vasculitis in one patient each.
Specifically, among patients with ulcerative colitis, skin lesions were reported in 8 of 27 with left-sided colitis, 2 of 15 with ulcerative colitis proctitis, and 22 of 67 patients with pancolitis. The difference between the groups of proctitis and pancolitis was significant (P = .03).
Within the group with Crohn’s disease, skin lesions were reported in 3 of 15 patients with ileitis, 4 of 10 with colitis, and 7 of 17 with ileocolitis. The difference among these groups was not significant (P > .05).
The most common skin lesions observed in Crohn’s disease were erythema nodosum and eczema, and in ulcerative colitis, psoriasis and eczema, the researchers reported.
They also noted that the cutaneous lesions were significantly more prevalent in extensive ulcerative colitis compared with distal disease.
Skin lesions add to patient misery
“Skin lesions are considered a burden to patients with IBD and add to their suffering,” said Sara Mesilhy, MBBS, a gastroenterologist with the Royal College of Physicians in the United Kingdom, who was not affiliated with the research.
The severity and location of the disease appears to play a role because researchers found extensive ulcerative colitis may carry a higher risk for the development of skin lesions, Dr. Mesilhy noted.
The first step when facing skin lesions is to control the disease activity via the best treatment option, Dr. Mesilhy suggested.
The study was independently supported. Dr. Jonaitis and Dr. Mesilhy have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
People with inflammatory bowel disease (IBD) commonly develop skin lesions linked to their condition, but until now few researchers looked at how common they are.
, according to the prospective, single-center study.
“Skin lesions in IBD patients are much more prevalent than it is generally accepted. The lesions may be related to the pathogenesis of IBD, but it is very important to know that the modern biological therapies may also cause skin lesions,” said senior study author Laimas Jonaitis, MD, PhD, professor in the department of gastroenterology at Lithuanian University of Health Sciences in Kaunas.
“If the gastroenterologist is experienced and has enough competence, he or she may establish the diagnosis, but in all other cases it is wise and advisable to refer the patient to the dermatologist,” Dr. Jonaitis said. A referral should include the history and full treatment for IBD.
The results were presented as a poster at the annual congress of the European Crohn’s and Colitis Organisation, held in Copenhagen and virtually.
Dr. Jonaitis and colleagues conducted a literature analysis to determine the prevalence of extra-abdominal manifestations of IBD. The lack of published data prompted them to survey 152 consecutive patients with IBD receiving outpatient treatment at their institution. The patients completed questionnaires from January to October 2022 about any cutaneous lesions.
The mean age of patients was 42 years, and 58% were men. A majority, 72%, had ulcerative colitis, and 28% had Crohn’s disease.
Prevalence of skin lesions
A total of 43% of participants reported skin lesions, but only 30% of patients had lesions considered related to IBD or IBD therapy due to their emergence after the patient’s IBD diagnosis.
By IBD diagnosis, 29% of patients with ulcerative colitis and 33% of patients with Crohn’s disease had lesions related to their condition. The difference in skin lesion prevalence between the two groups was not significant (P > .05), the researchers noted.
The team further investigated the types of skin lesions deemed to be associated with IBD or IBD therapy.
Overall, they found psoriasis in nine patients, eczema in nine, erythema nodosum in six, pyoderma gangrenosum in five, allergic rash in four, and vitiligo in two. They found acne, epidermolysis bullosa acquisita, and hemorrhagic vasculitis in one patient each.
Specifically, among patients with ulcerative colitis, skin lesions were reported in 8 of 27 with left-sided colitis, 2 of 15 with ulcerative colitis proctitis, and 22 of 67 patients with pancolitis. The difference between the groups of proctitis and pancolitis was significant (P = .03).
Within the group with Crohn’s disease, skin lesions were reported in 3 of 15 patients with ileitis, 4 of 10 with colitis, and 7 of 17 with ileocolitis. The difference among these groups was not significant (P > .05).
The most common skin lesions observed in Crohn’s disease were erythema nodosum and eczema, and in ulcerative colitis, psoriasis and eczema, the researchers reported.
They also noted that the cutaneous lesions were significantly more prevalent in extensive ulcerative colitis compared with distal disease.
Skin lesions add to patient misery
“Skin lesions are considered a burden to patients with IBD and add to their suffering,” said Sara Mesilhy, MBBS, a gastroenterologist with the Royal College of Physicians in the United Kingdom, who was not affiliated with the research.
The severity and location of the disease appears to play a role because researchers found extensive ulcerative colitis may carry a higher risk for the development of skin lesions, Dr. Mesilhy noted.
The first step when facing skin lesions is to control the disease activity via the best treatment option, Dr. Mesilhy suggested.
The study was independently supported. Dr. Jonaitis and Dr. Mesilhy have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ECCO 2023
New data on IV ketamine for resistant depression in the elderly
NEW ORLEANS –
“These were patients with depression who had not responded even to intensive therapies or procedures, and we found that after a 6-week ketamine infusion regimen, there was no difference in the response to the treatment between the treatment-resistant geriatric and nongeriatric patients,” study investigator Jonathan Kim, of Emory University, Atlanta, the first author of one of two studies presented as part of the American Association for Geriatric Psychiatry annual meeting, said in an interview.
The findings are important because research on the effects of IV ketamine have not been well documented in geriatric patients, who have high rates of depression and TRD.
“There is a lack of data on IV ketamine in older adults with treatment-resistant depression, and there are some safety and tolerability concerns which may lead some older adults and their clinicians to be reluctant to pursue IV ketamine treatment,” study coinvestigator Hanadi Ajam Oughli, MD, a health sciences assistant clinical professor in the department of psychiatry and biobehavioral sciences, University of California, Los Angeles, told this news organization.
Nasal vs. IV administration
Ketamine has traditionally been used as an anesthetic that blocks N-methyl-D-aspartate (NMDA) glutamate receptors, Dr. Oughli and colleagues note.
In the treatment of TRD, an infusion of 0.5 mg/kg is typically administered over 40 minutes, producing a rapid antidepressant response. Recent research shows the drug reduces suicidality and improves mood and quality of life.
A more recent intranasal formulation of ketamine, esketamine, was approved by the U.S. Food and Drug Administration for TRD in 2019, and some experts questioned its path to approval. In addition, the drug’s high cost and poor bioavailability in comparison with IV ketamine remains an issue, said Dr. Oughli.
In the previous TRANSFORM-3 study, a placebo-controlled randomized trial, there was no difference between esketamine, used in conjunction with an antidepressant, and placebo for geriatric patients.
To better understand the effects of IV ketamine in this patient population, Mr. Kim’s team conducted a retrospective chart review of 91 older patients with TRD who received IV ketamine treatment between October 2016 and August 2022.
Patients were divided into two groups – those older than 60 years (n = 36; 44% women; mean age, 68.86) and those younger than 60 (n = 55; 49% women; mean age, 41.05). Participants in each age group received six ketamine infusions over 6 weeks.
Results showed that with regard to depression severity, as assessed using Beck Depression Inventory (BDI-II) scores, 27.8% of patients in the geriatric group had a 50% or greater improvement, vs. 25.4% of those younger than 60.
The average BDI-II scores represented a significant improvement for both groups (P < .01), and the difference in scores between the groups was not statistically significant (P = .973).
“It is important to note that our study was conducted in a real-world clinical setting with a treatment-resistant population; other clinical studies may not have such sick patients in their trials. Additional studies are therefore warranted to establish further treatment guidelines in this area,” Mr. Kim said.
Open-label trial results
In the second study, Dr. Oughli and colleagues evaluated additional key outcomes in geriatric patients treated with IV ketamine as part of a larger open-label late-life trial on TRD.
The secondary analysis of the trial focused on 23 patients (mean age, 71.5 years) who had been initially treated with twice-a-week IV ketamine for 4 weeks.
After the first 4 weeks, patients who had experienced a partial response received an additional 4 weeks of once-weekly IV ketamine.
Overall, 48% of participants achieved a response, and 24% achieved remission of depressive symptoms following the first 4 weeks of twice-weekly treatment. This effect was maintained during the continuation phase of the study.
These findings are consistent with research in younger adults and demonstrate that twice-weekly infusions yield a more sustained antidepressant response than once-weekly infusions, the authors note.
The analysis also showed important increases in psychological well-being scores on the Scale for Suicidal Ideation, improved sleep quality as measured by the Pittsburgh Sleep Quality Index, and overall psychological well-being as shown on the NIH Toolbox Positive Affect on happiness/contentment and the NIH Toolbox General Life Satisfaction scales.
In a previous analysis, published in The American Journal of Geriatric Psychiatry, the researchers also evaluated cognitive function using the NIH Cognitive Battery, which showed that geriatric patients with TRD had significant improvements in a composite of executive functioning and fluid cognition during the 4-week acute treatment period of twice-weekly IV ketamine infusions (Cohen’s d = 0.61) and that those improvements were sustained in the continuation phase of once-weekly infusions for 4 more weeks.
Those results are consistent with ketamine’s known potential procognitive effects in TRD, due to a putative antidepressant mechanism that rescues prefrontal circuit dysfunction through synaptogenesis, the researchers note.
Dr. Oughli said that in both analyses, patients tolerated ketamine well, and there were no serious adverse events.
“Adverse events, including hypertension, dissociated effects, and cravings, were rare and did not prevent the continued use of IV ketamine by older adults. We were able to use clonidine to help manage blood pressure changes seen during the infusions,” she noted.
“These findings are very promising and will need to be confirmed and extended in a larger randomized controlled trial.”
Unsettling for some older patients
George T. Grossberg, MD, director, geriatric psychiatry, Saint Louis University, noted that in his experience, IV ketamine treatment can be unsettling for some older geriatric patients, such as those in their 80s.
“Particularly with some of my older patients, the kind of psychotomimetic properties of ketamine and the out-of-body experiences [with the initial treatment] can be frightening,” he said. “They may be willing to try, but I’ve had more than one patient quit after one treatment because they became so frightened.”
However, the dire nature of TRD and failure to respond to multiple medications and combinations and other strategies may prompt patients to try ketamine as a measure with at least some potential, he noted.
“But there is a high bar for acceptance, especially on the part of older adults and their families, more than for younger people,” he said.
The investigators have disclosed no relevant financial relationships. Dr. Grossberg has received consulting fees from Acadia, Avanir, Biogen, BioXcel, Genentech, Karuna, Lundbeck, Otsuka, Roche, and Takeda.
A version of this article first appeared on Medscape.com.
NEW ORLEANS –
“These were patients with depression who had not responded even to intensive therapies or procedures, and we found that after a 6-week ketamine infusion regimen, there was no difference in the response to the treatment between the treatment-resistant geriatric and nongeriatric patients,” study investigator Jonathan Kim, of Emory University, Atlanta, the first author of one of two studies presented as part of the American Association for Geriatric Psychiatry annual meeting, said in an interview.
The findings are important because research on the effects of IV ketamine have not been well documented in geriatric patients, who have high rates of depression and TRD.
“There is a lack of data on IV ketamine in older adults with treatment-resistant depression, and there are some safety and tolerability concerns which may lead some older adults and their clinicians to be reluctant to pursue IV ketamine treatment,” study coinvestigator Hanadi Ajam Oughli, MD, a health sciences assistant clinical professor in the department of psychiatry and biobehavioral sciences, University of California, Los Angeles, told this news organization.
Nasal vs. IV administration
Ketamine has traditionally been used as an anesthetic that blocks N-methyl-D-aspartate (NMDA) glutamate receptors, Dr. Oughli and colleagues note.
In the treatment of TRD, an infusion of 0.5 mg/kg is typically administered over 40 minutes, producing a rapid antidepressant response. Recent research shows the drug reduces suicidality and improves mood and quality of life.
A more recent intranasal formulation of ketamine, esketamine, was approved by the U.S. Food and Drug Administration for TRD in 2019, and some experts questioned its path to approval. In addition, the drug’s high cost and poor bioavailability in comparison with IV ketamine remains an issue, said Dr. Oughli.
In the previous TRANSFORM-3 study, a placebo-controlled randomized trial, there was no difference between esketamine, used in conjunction with an antidepressant, and placebo for geriatric patients.
To better understand the effects of IV ketamine in this patient population, Mr. Kim’s team conducted a retrospective chart review of 91 older patients with TRD who received IV ketamine treatment between October 2016 and August 2022.
Patients were divided into two groups – those older than 60 years (n = 36; 44% women; mean age, 68.86) and those younger than 60 (n = 55; 49% women; mean age, 41.05). Participants in each age group received six ketamine infusions over 6 weeks.
Results showed that with regard to depression severity, as assessed using Beck Depression Inventory (BDI-II) scores, 27.8% of patients in the geriatric group had a 50% or greater improvement, vs. 25.4% of those younger than 60.
The average BDI-II scores represented a significant improvement for both groups (P < .01), and the difference in scores between the groups was not statistically significant (P = .973).
“It is important to note that our study was conducted in a real-world clinical setting with a treatment-resistant population; other clinical studies may not have such sick patients in their trials. Additional studies are therefore warranted to establish further treatment guidelines in this area,” Mr. Kim said.
Open-label trial results
In the second study, Dr. Oughli and colleagues evaluated additional key outcomes in geriatric patients treated with IV ketamine as part of a larger open-label late-life trial on TRD.
The secondary analysis of the trial focused on 23 patients (mean age, 71.5 years) who had been initially treated with twice-a-week IV ketamine for 4 weeks.
After the first 4 weeks, patients who had experienced a partial response received an additional 4 weeks of once-weekly IV ketamine.
Overall, 48% of participants achieved a response, and 24% achieved remission of depressive symptoms following the first 4 weeks of twice-weekly treatment. This effect was maintained during the continuation phase of the study.
These findings are consistent with research in younger adults and demonstrate that twice-weekly infusions yield a more sustained antidepressant response than once-weekly infusions, the authors note.
The analysis also showed important increases in psychological well-being scores on the Scale for Suicidal Ideation, improved sleep quality as measured by the Pittsburgh Sleep Quality Index, and overall psychological well-being as shown on the NIH Toolbox Positive Affect on happiness/contentment and the NIH Toolbox General Life Satisfaction scales.
In a previous analysis, published in The American Journal of Geriatric Psychiatry, the researchers also evaluated cognitive function using the NIH Cognitive Battery, which showed that geriatric patients with TRD had significant improvements in a composite of executive functioning and fluid cognition during the 4-week acute treatment period of twice-weekly IV ketamine infusions (Cohen’s d = 0.61) and that those improvements were sustained in the continuation phase of once-weekly infusions for 4 more weeks.
Those results are consistent with ketamine’s known potential procognitive effects in TRD, due to a putative antidepressant mechanism that rescues prefrontal circuit dysfunction through synaptogenesis, the researchers note.
Dr. Oughli said that in both analyses, patients tolerated ketamine well, and there were no serious adverse events.
“Adverse events, including hypertension, dissociated effects, and cravings, were rare and did not prevent the continued use of IV ketamine by older adults. We were able to use clonidine to help manage blood pressure changes seen during the infusions,” she noted.
“These findings are very promising and will need to be confirmed and extended in a larger randomized controlled trial.”
Unsettling for some older patients
George T. Grossberg, MD, director, geriatric psychiatry, Saint Louis University, noted that in his experience, IV ketamine treatment can be unsettling for some older geriatric patients, such as those in their 80s.
“Particularly with some of my older patients, the kind of psychotomimetic properties of ketamine and the out-of-body experiences [with the initial treatment] can be frightening,” he said. “They may be willing to try, but I’ve had more than one patient quit after one treatment because they became so frightened.”
However, the dire nature of TRD and failure to respond to multiple medications and combinations and other strategies may prompt patients to try ketamine as a measure with at least some potential, he noted.
“But there is a high bar for acceptance, especially on the part of older adults and their families, more than for younger people,” he said.
The investigators have disclosed no relevant financial relationships. Dr. Grossberg has received consulting fees from Acadia, Avanir, Biogen, BioXcel, Genentech, Karuna, Lundbeck, Otsuka, Roche, and Takeda.
A version of this article first appeared on Medscape.com.
NEW ORLEANS –
“These were patients with depression who had not responded even to intensive therapies or procedures, and we found that after a 6-week ketamine infusion regimen, there was no difference in the response to the treatment between the treatment-resistant geriatric and nongeriatric patients,” study investigator Jonathan Kim, of Emory University, Atlanta, the first author of one of two studies presented as part of the American Association for Geriatric Psychiatry annual meeting, said in an interview.
The findings are important because research on the effects of IV ketamine have not been well documented in geriatric patients, who have high rates of depression and TRD.
“There is a lack of data on IV ketamine in older adults with treatment-resistant depression, and there are some safety and tolerability concerns which may lead some older adults and their clinicians to be reluctant to pursue IV ketamine treatment,” study coinvestigator Hanadi Ajam Oughli, MD, a health sciences assistant clinical professor in the department of psychiatry and biobehavioral sciences, University of California, Los Angeles, told this news organization.
Nasal vs. IV administration
Ketamine has traditionally been used as an anesthetic that blocks N-methyl-D-aspartate (NMDA) glutamate receptors, Dr. Oughli and colleagues note.
In the treatment of TRD, an infusion of 0.5 mg/kg is typically administered over 40 minutes, producing a rapid antidepressant response. Recent research shows the drug reduces suicidality and improves mood and quality of life.
A more recent intranasal formulation of ketamine, esketamine, was approved by the U.S. Food and Drug Administration for TRD in 2019, and some experts questioned its path to approval. In addition, the drug’s high cost and poor bioavailability in comparison with IV ketamine remains an issue, said Dr. Oughli.
In the previous TRANSFORM-3 study, a placebo-controlled randomized trial, there was no difference between esketamine, used in conjunction with an antidepressant, and placebo for geriatric patients.
To better understand the effects of IV ketamine in this patient population, Mr. Kim’s team conducted a retrospective chart review of 91 older patients with TRD who received IV ketamine treatment between October 2016 and August 2022.
Patients were divided into two groups – those older than 60 years (n = 36; 44% women; mean age, 68.86) and those younger than 60 (n = 55; 49% women; mean age, 41.05). Participants in each age group received six ketamine infusions over 6 weeks.
Results showed that with regard to depression severity, as assessed using Beck Depression Inventory (BDI-II) scores, 27.8% of patients in the geriatric group had a 50% or greater improvement, vs. 25.4% of those younger than 60.
The average BDI-II scores represented a significant improvement for both groups (P < .01), and the difference in scores between the groups was not statistically significant (P = .973).
“It is important to note that our study was conducted in a real-world clinical setting with a treatment-resistant population; other clinical studies may not have such sick patients in their trials. Additional studies are therefore warranted to establish further treatment guidelines in this area,” Mr. Kim said.
Open-label trial results
In the second study, Dr. Oughli and colleagues evaluated additional key outcomes in geriatric patients treated with IV ketamine as part of a larger open-label late-life trial on TRD.
The secondary analysis of the trial focused on 23 patients (mean age, 71.5 years) who had been initially treated with twice-a-week IV ketamine for 4 weeks.
After the first 4 weeks, patients who had experienced a partial response received an additional 4 weeks of once-weekly IV ketamine.
Overall, 48% of participants achieved a response, and 24% achieved remission of depressive symptoms following the first 4 weeks of twice-weekly treatment. This effect was maintained during the continuation phase of the study.
These findings are consistent with research in younger adults and demonstrate that twice-weekly infusions yield a more sustained antidepressant response than once-weekly infusions, the authors note.
The analysis also showed important increases in psychological well-being scores on the Scale for Suicidal Ideation, improved sleep quality as measured by the Pittsburgh Sleep Quality Index, and overall psychological well-being as shown on the NIH Toolbox Positive Affect on happiness/contentment and the NIH Toolbox General Life Satisfaction scales.
In a previous analysis, published in The American Journal of Geriatric Psychiatry, the researchers also evaluated cognitive function using the NIH Cognitive Battery, which showed that geriatric patients with TRD had significant improvements in a composite of executive functioning and fluid cognition during the 4-week acute treatment period of twice-weekly IV ketamine infusions (Cohen’s d = 0.61) and that those improvements were sustained in the continuation phase of once-weekly infusions for 4 more weeks.
Those results are consistent with ketamine’s known potential procognitive effects in TRD, due to a putative antidepressant mechanism that rescues prefrontal circuit dysfunction through synaptogenesis, the researchers note.
Dr. Oughli said that in both analyses, patients tolerated ketamine well, and there were no serious adverse events.
“Adverse events, including hypertension, dissociated effects, and cravings, were rare and did not prevent the continued use of IV ketamine by older adults. We were able to use clonidine to help manage blood pressure changes seen during the infusions,” she noted.
“These findings are very promising and will need to be confirmed and extended in a larger randomized controlled trial.”
Unsettling for some older patients
George T. Grossberg, MD, director, geriatric psychiatry, Saint Louis University, noted that in his experience, IV ketamine treatment can be unsettling for some older geriatric patients, such as those in their 80s.
“Particularly with some of my older patients, the kind of psychotomimetic properties of ketamine and the out-of-body experiences [with the initial treatment] can be frightening,” he said. “They may be willing to try, but I’ve had more than one patient quit after one treatment because they became so frightened.”
However, the dire nature of TRD and failure to respond to multiple medications and combinations and other strategies may prompt patients to try ketamine as a measure with at least some potential, he noted.
“But there is a high bar for acceptance, especially on the part of older adults and their families, more than for younger people,” he said.
The investigators have disclosed no relevant financial relationships. Dr. Grossberg has received consulting fees from Acadia, Avanir, Biogen, BioXcel, Genentech, Karuna, Lundbeck, Otsuka, Roche, and Takeda.
A version of this article first appeared on Medscape.com.
AT AAGP 2023
COVID raises risk for long-term GI complications
, a large new study indicates.
The researchers estimate that, so far, SARS-CoV-2 infections have contributed to more than 6 million new cases of GI disorders in the United States and 42 million new cases worldwide.
The diagnoses more common among patients who’ve had COVID ranged from stomach upset to acute pancreatitis, say the researchers, led by Evan Xu, a data analyst at the Clinical Epidemiology Center, Research and Development Service, VA St. Louis Health Care System.
Signs and symptoms of GI problems, such as constipation and diarrhea, also were more common among patients who had had the virus, the study found.
“Altogether, our results show that people with SARS-CoV-2 infection are at increased risk of gastrointestinal disorders in the post-acute phase of COVID-19,” the researchers write. “Post-COVID care should involve attention to gastrointestinal health and disease.”
The results were published online in Nature Communications.
Disease risks jump
The researchers used data from the U.S. Department of Veterans Affairs national health care databases to identify 154,068 people with confirmed COVID-19 from March 1, 2020, through Jan. 15, 2021. They used statistical modeling to compare those patients with 5.6 million patients with similar characteristics who had not been infected during the same period and an historical control group of 5.9 million patients from March 1, 2018, to Dec. 31, 2019, before the virus began to spread across the globe.
The study included hospitalized and nonhospitalized COVID patients. The majority of the study population was male, but the study included almost 1.2 million female patients.
Compared with control persons, post-COVID patients’ increased risk of a GI diagnosis and the excess disease burden at 1 year, respectively, were as follows.
- 102% for cholangitis; 0.22 per 1,000 persons
- 62% for peptic ulcer disease; 1.57 per 1,000 persons
- 54% for irritable bowel syndrome; 0.44 per 1,000 persons
- 47% for acute gastritis; 0.47 per 1,000 persons
- 46% for acute pancreatitis; 0.6 per 1,000 persons
- 36% for functional dyspepsia; 0.63 per 1,000 persons
- 35% for gastroesophageal reflux disease; 15.5 per 1,000 persons
Patients who’d had the virus were also at higher risk for GI symptoms than their COVID-free peers. Their risk was 60% higher for constipation, 58% for diarrhea, 52% for vomiting, 46% for bloating, and 44% for abdominal pain, the investigators found.
The risk of developing GI symptoms increased with COVID-19 severity and was highest for those who received intensive care because of the virus, the researchers note.
Subgroup analyses found that the risks of composite gastrointestinal outcome were evident in all subgroups based on age, race, sex, obesity, smoking, cardiovascular disease, chronic kidney disease, diabetes, hyperlipidemia, and hypertension, the authors write.
Disease burden rises
The increased numbers of GI patients with prior SARS-CoV-2 infection are altering the burden on the health care system, senior author Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University, St. Louis, said in an interview.
The shift may be pronounced in primary care, where GI concerns should be seen as a trigger for questions about prior SARS-CoV-2 infection, Dr. Al-Aly said.
Patients may encounter longer wait times at GI clinics or may give up on trying to schedule appointments if waits become too long, he said. They may also present to emergency departments if they can’t get an outpatient appointment, he added.
Simon C. Mathews, MD, assistant professor of medicine, division of gastroenterology, Johns Hopkins Medicine, Baltimore, told this news organization that he’s seeing increased wait times since COVID emerged.
“We know that the pandemic impacted patients’ ability and willingness to seek GI care. There continues to be a long backlog for patients who are only now getting reconnected to care. As a result, our clinics are busier than ever, and our wait times for appointments are unfortunately longer than we would like,” said Dr. Mathews, who was not involved in the research.
Abdominal pain, bloating, diarrhea, and constipation continue to be among the most common symptoms Dr. Mathews sees in clinic, he said.
Kyle Staller, MD, a Massachusetts General Brigham gastroenterologist, said in an interview that it’s important to distinguish symptoms from eventual diagnoses, which lag behind.
“Are patients attributing their symptoms to COVID, or is COVID itself creating a background of inflammation or changes in the nerves that are making these symptoms more common? My suspicion is a little bit of both,” said Dr. Staller, who is director of the Gastrointestinal Motility Laboratory at Mass General, Boston.
Although his clinic is seeing patients with the GI signs and symptoms listed in the article, “we’re not seeing as much of some of the diagnoses, like peptic ulcer disease and pancreatitis,” he said. “I wonder if those may be related to some of the consequences of being critically ill in general, rather than COVID specifically. Those diagnoses I would be more skeptical about.”
Duration of symptoms unclear
It’s hard to tell patients how long their GI symptoms might last after COVID, given the relatively short time researchers have had to study the virus, said Dr. Staller, who was not involved in the research.
The symptoms he’s seeing in patients after COVID mimic those of postinfectious IBS, which literature says could last for months or years, Dr. Staller said. “But they should improve over time,” he added.
Senior author Dr. Al-Aly agreed that the duration of post-COVID GI symptoms is unclear.
“What I can tell you is that even people who got SARS-CoV-2 infection from March 2020 are still coming back for GI problems,” he said.
Unlike other symptoms of long COVID, such as brain fog, gastroenterologists fortunately know how to treat the GI disorders that evolve from SARS-CoV-2 infection, said Dr. Al-Aly, who has studied the long-term effects of the virus on the brain, kidneys, heart, and other organs.
All health care providers “need to be thinking about COVID as a risk factor for all these diseases” and should ask patients about SARS-CoV-2 infection when they take their histories, he said.
The authors, Dr. Staller, and Dr. Mathews report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a large new study indicates.
The researchers estimate that, so far, SARS-CoV-2 infections have contributed to more than 6 million new cases of GI disorders in the United States and 42 million new cases worldwide.
The diagnoses more common among patients who’ve had COVID ranged from stomach upset to acute pancreatitis, say the researchers, led by Evan Xu, a data analyst at the Clinical Epidemiology Center, Research and Development Service, VA St. Louis Health Care System.
Signs and symptoms of GI problems, such as constipation and diarrhea, also were more common among patients who had had the virus, the study found.
“Altogether, our results show that people with SARS-CoV-2 infection are at increased risk of gastrointestinal disorders in the post-acute phase of COVID-19,” the researchers write. “Post-COVID care should involve attention to gastrointestinal health and disease.”
The results were published online in Nature Communications.
Disease risks jump
The researchers used data from the U.S. Department of Veterans Affairs national health care databases to identify 154,068 people with confirmed COVID-19 from March 1, 2020, through Jan. 15, 2021. They used statistical modeling to compare those patients with 5.6 million patients with similar characteristics who had not been infected during the same period and an historical control group of 5.9 million patients from March 1, 2018, to Dec. 31, 2019, before the virus began to spread across the globe.
The study included hospitalized and nonhospitalized COVID patients. The majority of the study population was male, but the study included almost 1.2 million female patients.
Compared with control persons, post-COVID patients’ increased risk of a GI diagnosis and the excess disease burden at 1 year, respectively, were as follows.
- 102% for cholangitis; 0.22 per 1,000 persons
- 62% for peptic ulcer disease; 1.57 per 1,000 persons
- 54% for irritable bowel syndrome; 0.44 per 1,000 persons
- 47% for acute gastritis; 0.47 per 1,000 persons
- 46% for acute pancreatitis; 0.6 per 1,000 persons
- 36% for functional dyspepsia; 0.63 per 1,000 persons
- 35% for gastroesophageal reflux disease; 15.5 per 1,000 persons
Patients who’d had the virus were also at higher risk for GI symptoms than their COVID-free peers. Their risk was 60% higher for constipation, 58% for diarrhea, 52% for vomiting, 46% for bloating, and 44% for abdominal pain, the investigators found.
The risk of developing GI symptoms increased with COVID-19 severity and was highest for those who received intensive care because of the virus, the researchers note.
Subgroup analyses found that the risks of composite gastrointestinal outcome were evident in all subgroups based on age, race, sex, obesity, smoking, cardiovascular disease, chronic kidney disease, diabetes, hyperlipidemia, and hypertension, the authors write.
Disease burden rises
The increased numbers of GI patients with prior SARS-CoV-2 infection are altering the burden on the health care system, senior author Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University, St. Louis, said in an interview.
The shift may be pronounced in primary care, where GI concerns should be seen as a trigger for questions about prior SARS-CoV-2 infection, Dr. Al-Aly said.
Patients may encounter longer wait times at GI clinics or may give up on trying to schedule appointments if waits become too long, he said. They may also present to emergency departments if they can’t get an outpatient appointment, he added.
Simon C. Mathews, MD, assistant professor of medicine, division of gastroenterology, Johns Hopkins Medicine, Baltimore, told this news organization that he’s seeing increased wait times since COVID emerged.
“We know that the pandemic impacted patients’ ability and willingness to seek GI care. There continues to be a long backlog for patients who are only now getting reconnected to care. As a result, our clinics are busier than ever, and our wait times for appointments are unfortunately longer than we would like,” said Dr. Mathews, who was not involved in the research.
Abdominal pain, bloating, diarrhea, and constipation continue to be among the most common symptoms Dr. Mathews sees in clinic, he said.
Kyle Staller, MD, a Massachusetts General Brigham gastroenterologist, said in an interview that it’s important to distinguish symptoms from eventual diagnoses, which lag behind.
“Are patients attributing their symptoms to COVID, or is COVID itself creating a background of inflammation or changes in the nerves that are making these symptoms more common? My suspicion is a little bit of both,” said Dr. Staller, who is director of the Gastrointestinal Motility Laboratory at Mass General, Boston.
Although his clinic is seeing patients with the GI signs and symptoms listed in the article, “we’re not seeing as much of some of the diagnoses, like peptic ulcer disease and pancreatitis,” he said. “I wonder if those may be related to some of the consequences of being critically ill in general, rather than COVID specifically. Those diagnoses I would be more skeptical about.”
Duration of symptoms unclear
It’s hard to tell patients how long their GI symptoms might last after COVID, given the relatively short time researchers have had to study the virus, said Dr. Staller, who was not involved in the research.
The symptoms he’s seeing in patients after COVID mimic those of postinfectious IBS, which literature says could last for months or years, Dr. Staller said. “But they should improve over time,” he added.
Senior author Dr. Al-Aly agreed that the duration of post-COVID GI symptoms is unclear.
“What I can tell you is that even people who got SARS-CoV-2 infection from March 2020 are still coming back for GI problems,” he said.
Unlike other symptoms of long COVID, such as brain fog, gastroenterologists fortunately know how to treat the GI disorders that evolve from SARS-CoV-2 infection, said Dr. Al-Aly, who has studied the long-term effects of the virus on the brain, kidneys, heart, and other organs.
All health care providers “need to be thinking about COVID as a risk factor for all these diseases” and should ask patients about SARS-CoV-2 infection when they take their histories, he said.
The authors, Dr. Staller, and Dr. Mathews report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a large new study indicates.
The researchers estimate that, so far, SARS-CoV-2 infections have contributed to more than 6 million new cases of GI disorders in the United States and 42 million new cases worldwide.
The diagnoses more common among patients who’ve had COVID ranged from stomach upset to acute pancreatitis, say the researchers, led by Evan Xu, a data analyst at the Clinical Epidemiology Center, Research and Development Service, VA St. Louis Health Care System.
Signs and symptoms of GI problems, such as constipation and diarrhea, also were more common among patients who had had the virus, the study found.
“Altogether, our results show that people with SARS-CoV-2 infection are at increased risk of gastrointestinal disorders in the post-acute phase of COVID-19,” the researchers write. “Post-COVID care should involve attention to gastrointestinal health and disease.”
The results were published online in Nature Communications.
Disease risks jump
The researchers used data from the U.S. Department of Veterans Affairs national health care databases to identify 154,068 people with confirmed COVID-19 from March 1, 2020, through Jan. 15, 2021. They used statistical modeling to compare those patients with 5.6 million patients with similar characteristics who had not been infected during the same period and an historical control group of 5.9 million patients from March 1, 2018, to Dec. 31, 2019, before the virus began to spread across the globe.
The study included hospitalized and nonhospitalized COVID patients. The majority of the study population was male, but the study included almost 1.2 million female patients.
Compared with control persons, post-COVID patients’ increased risk of a GI diagnosis and the excess disease burden at 1 year, respectively, were as follows.
- 102% for cholangitis; 0.22 per 1,000 persons
- 62% for peptic ulcer disease; 1.57 per 1,000 persons
- 54% for irritable bowel syndrome; 0.44 per 1,000 persons
- 47% for acute gastritis; 0.47 per 1,000 persons
- 46% for acute pancreatitis; 0.6 per 1,000 persons
- 36% for functional dyspepsia; 0.63 per 1,000 persons
- 35% for gastroesophageal reflux disease; 15.5 per 1,000 persons
Patients who’d had the virus were also at higher risk for GI symptoms than their COVID-free peers. Their risk was 60% higher for constipation, 58% for diarrhea, 52% for vomiting, 46% for bloating, and 44% for abdominal pain, the investigators found.
The risk of developing GI symptoms increased with COVID-19 severity and was highest for those who received intensive care because of the virus, the researchers note.
Subgroup analyses found that the risks of composite gastrointestinal outcome were evident in all subgroups based on age, race, sex, obesity, smoking, cardiovascular disease, chronic kidney disease, diabetes, hyperlipidemia, and hypertension, the authors write.
Disease burden rises
The increased numbers of GI patients with prior SARS-CoV-2 infection are altering the burden on the health care system, senior author Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University, St. Louis, said in an interview.
The shift may be pronounced in primary care, where GI concerns should be seen as a trigger for questions about prior SARS-CoV-2 infection, Dr. Al-Aly said.
Patients may encounter longer wait times at GI clinics or may give up on trying to schedule appointments if waits become too long, he said. They may also present to emergency departments if they can’t get an outpatient appointment, he added.
Simon C. Mathews, MD, assistant professor of medicine, division of gastroenterology, Johns Hopkins Medicine, Baltimore, told this news organization that he’s seeing increased wait times since COVID emerged.
“We know that the pandemic impacted patients’ ability and willingness to seek GI care. There continues to be a long backlog for patients who are only now getting reconnected to care. As a result, our clinics are busier than ever, and our wait times for appointments are unfortunately longer than we would like,” said Dr. Mathews, who was not involved in the research.
Abdominal pain, bloating, diarrhea, and constipation continue to be among the most common symptoms Dr. Mathews sees in clinic, he said.
Kyle Staller, MD, a Massachusetts General Brigham gastroenterologist, said in an interview that it’s important to distinguish symptoms from eventual diagnoses, which lag behind.
“Are patients attributing their symptoms to COVID, or is COVID itself creating a background of inflammation or changes in the nerves that are making these symptoms more common? My suspicion is a little bit of both,” said Dr. Staller, who is director of the Gastrointestinal Motility Laboratory at Mass General, Boston.
Although his clinic is seeing patients with the GI signs and symptoms listed in the article, “we’re not seeing as much of some of the diagnoses, like peptic ulcer disease and pancreatitis,” he said. “I wonder if those may be related to some of the consequences of being critically ill in general, rather than COVID specifically. Those diagnoses I would be more skeptical about.”
Duration of symptoms unclear
It’s hard to tell patients how long their GI symptoms might last after COVID, given the relatively short time researchers have had to study the virus, said Dr. Staller, who was not involved in the research.
The symptoms he’s seeing in patients after COVID mimic those of postinfectious IBS, which literature says could last for months or years, Dr. Staller said. “But they should improve over time,” he added.
Senior author Dr. Al-Aly agreed that the duration of post-COVID GI symptoms is unclear.
“What I can tell you is that even people who got SARS-CoV-2 infection from March 2020 are still coming back for GI problems,” he said.
Unlike other symptoms of long COVID, such as brain fog, gastroenterologists fortunately know how to treat the GI disorders that evolve from SARS-CoV-2 infection, said Dr. Al-Aly, who has studied the long-term effects of the virus on the brain, kidneys, heart, and other organs.
All health care providers “need to be thinking about COVID as a risk factor for all these diseases” and should ask patients about SARS-CoV-2 infection when they take their histories, he said.
The authors, Dr. Staller, and Dr. Mathews report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE COMMUNICATIONS
Add-on antipsychotic beats switching meds in older adults with resistant depression
NEW ORLEANS –
“We found that adding aripiprazole led to higher rates of depression remission and greater improvements in psychological well-being – which means how positive and satisfied patients felt – and this is good news,” study investigator Eric J. Lenze, MD, of the department of psychiatry, Washington University, St. Louis, said in a press statement.
“However, even that approach helped only about 30% of people in the study with treatment-resistant depression, underscoring the need to find and develop more effective treatments that can help more people,” he added.
The findings were presented here as part of the American Association for Geriatric Psychiatry annual meeting, and published concurrently in the New England Journal of Medicine.
Need for safe treatment options
Treatment-resistant depression is common in older patients, but switching medications or adding other agents can be challenging. With higher rates of comorbidity and polypharmacy, treatment decisions in this patient population are more complex compared with those involving younger patients.
To compare the benefits of augmentation vs. drug-switching strategies, the researchers conducted a multicenter, two-step trial involving 619 patients with an average baseline age of 69 who had failed to respond to two courses of selective serotonin reuptake inhibitors (SSRIs).
Patients were randomly assigned to one of three groups. These included augmentation of existing antidepressant medication with either aripiprazole (n = 211) or the dopamine and norepinephrine–reuptake inhibitor bupropion (Wellbutrin, Zyban) (n = 206), or to taper off of their current antidepressant and switch to bupropion (n = 202).
After 10 weeks, patients’ psychological well-being was assessed via the National Institutes of Health Toolbox Positive Affect and General Life Satisfaction subscales. The researchers found patients in the aripiprazole and bupropion add-on groups improved by 4.83 points and 4.33 points, respectively. The bupropion switch group had a change of 2.04 points.
The difference between the aripiprazole augmentation group and the switch to bupropion group was significant (difference 2.79 points; P = .014). Other between-group differences were not significantly different.
Remission rates were similar in the aripiprazole and bupropion groups at 28.9% and 28.2%, respectively. The remission rate in the bupropion switch group was 19.3%.
The study results showed patients who received adjunctive bupropion had the highest fall rate at 0.55 falls per patient, vs. 0.33 falls per patient in the aripiprazole group, suggesting that among the three treatment options, adjunctive aripiprazole may be the best choice because of its superior efficacy and lower fall risk.
A total of 248 patients enrolled in the study showed no improvement and were further randomly assigned to receive adjunctive lithium (n = 127) or switch from current therapy to nortriptyline (n = 121).
Well-being scores in the lithium group improved by 3.17 points and 2.18 points in the nortriptyline group. Remission occurred in 18.9% of patients in the lithium group and 21.5% in the nortriptyline group. Fall rates were similar among the two groups.
Overall, “this large, randomized study demonstrated that adding aripiprazole was a superior option for older adults with treatment-resistant depression,” Dr. Lenze told this news organization.
“Since neither lithium nor nortriptyline were promising against treatment-resistant depression in older adults, those medications are unlikely to be helpful in most cases,” he added.
Practice changing?
In an accompanying editorial, Gemma Lewis, PhD, and Glyn Lewis, PhD, division of psychiatry, University of College London, noted the findings “support aripiprazole augmentation as a strategy for treatment-resistant depression in older persons, largely because of the lower risk of falls than with bupropion augmentation.”
However, “in clinical practice, [it] would be important to tailor treatment in light of potential adverse effects and the preferences of the patient,” they added.
Akathisia, for instance, is a common side effect of aripiprazole, shown in one recent trial to affect 11% of the patients. In addition, weight gain, though typically lower than seen with other antipsychotics, is a consideration with aripiprazole.
With respect to fall risk, they noted that bupropion was largely used in relatively high doses of 300 mg and 450 mg, despite some recent research showing little clinical benefit from increasing antidepressant doses above minimum recommendations.
“It is possible that smaller doses of bupropion than those used in the current trial would retain effectiveness while minimizing adverse effects such as falls,” the editorialists noted.
Commenting on the study, Jennifer R. Gatchel, MD, PhD, assistant psychiatrist at Massachusetts General Hospital/McLean Hospital and assistant professor of psychiatry at Harvard Medical School, Boston, said the findings have high clinical significance in the treatment of geriatric depression.
“These results are of great impact for clinicians managing older adults with treatment-resistant depression. They provide some of the first evidence of safety and efficacy of augmentation with aripiprazole as a strategy in clinical management of older adults who fail to initially respond to treatment,” said Dr. Gatchel, who was not associated with this research.
“Of particular significance, efficacy here is based on patient-centered outcomes and psychological well-being as a primary effectiveness outcome, which could translate into strengthened physician-patient alliance.”
While adjunctive aripiprazole is not necessarily a first-line strategy when older adults fail to respond to antidepressants, there is a lack of data on the risks and benefits of any other antipsychotic medications, she noted.
“Thus, this is evidence that will impact clinical practice and hopefully contribute to reduced societal burden of depression in older adults and the morbidity and mortality associated with it,” Dr. Gatchel said.
The study received support from a Patient-Centered Outcomes Research Institute (PCORI) Award (TRD-1511-33321). Dr. Lenze received additional support from the Taylor Family Institute for Innovative Psychiatric Research at Washington University School of Medicine, as well as the Washington University Institute of Clinical and Translational Sciences grant (UL1TR002345) from the National Center for Advancing Translational Sciences of the National Institutes of Health. Dr. Gatchel reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW ORLEANS –
“We found that adding aripiprazole led to higher rates of depression remission and greater improvements in psychological well-being – which means how positive and satisfied patients felt – and this is good news,” study investigator Eric J. Lenze, MD, of the department of psychiatry, Washington University, St. Louis, said in a press statement.
“However, even that approach helped only about 30% of people in the study with treatment-resistant depression, underscoring the need to find and develop more effective treatments that can help more people,” he added.
The findings were presented here as part of the American Association for Geriatric Psychiatry annual meeting, and published concurrently in the New England Journal of Medicine.
Need for safe treatment options
Treatment-resistant depression is common in older patients, but switching medications or adding other agents can be challenging. With higher rates of comorbidity and polypharmacy, treatment decisions in this patient population are more complex compared with those involving younger patients.
To compare the benefits of augmentation vs. drug-switching strategies, the researchers conducted a multicenter, two-step trial involving 619 patients with an average baseline age of 69 who had failed to respond to two courses of selective serotonin reuptake inhibitors (SSRIs).
Patients were randomly assigned to one of three groups. These included augmentation of existing antidepressant medication with either aripiprazole (n = 211) or the dopamine and norepinephrine–reuptake inhibitor bupropion (Wellbutrin, Zyban) (n = 206), or to taper off of their current antidepressant and switch to bupropion (n = 202).
After 10 weeks, patients’ psychological well-being was assessed via the National Institutes of Health Toolbox Positive Affect and General Life Satisfaction subscales. The researchers found patients in the aripiprazole and bupropion add-on groups improved by 4.83 points and 4.33 points, respectively. The bupropion switch group had a change of 2.04 points.
The difference between the aripiprazole augmentation group and the switch to bupropion group was significant (difference 2.79 points; P = .014). Other between-group differences were not significantly different.
Remission rates were similar in the aripiprazole and bupropion groups at 28.9% and 28.2%, respectively. The remission rate in the bupropion switch group was 19.3%.
The study results showed patients who received adjunctive bupropion had the highest fall rate at 0.55 falls per patient, vs. 0.33 falls per patient in the aripiprazole group, suggesting that among the three treatment options, adjunctive aripiprazole may be the best choice because of its superior efficacy and lower fall risk.
A total of 248 patients enrolled in the study showed no improvement and were further randomly assigned to receive adjunctive lithium (n = 127) or switch from current therapy to nortriptyline (n = 121).
Well-being scores in the lithium group improved by 3.17 points and 2.18 points in the nortriptyline group. Remission occurred in 18.9% of patients in the lithium group and 21.5% in the nortriptyline group. Fall rates were similar among the two groups.
Overall, “this large, randomized study demonstrated that adding aripiprazole was a superior option for older adults with treatment-resistant depression,” Dr. Lenze told this news organization.
“Since neither lithium nor nortriptyline were promising against treatment-resistant depression in older adults, those medications are unlikely to be helpful in most cases,” he added.
Practice changing?
In an accompanying editorial, Gemma Lewis, PhD, and Glyn Lewis, PhD, division of psychiatry, University of College London, noted the findings “support aripiprazole augmentation as a strategy for treatment-resistant depression in older persons, largely because of the lower risk of falls than with bupropion augmentation.”
However, “in clinical practice, [it] would be important to tailor treatment in light of potential adverse effects and the preferences of the patient,” they added.
Akathisia, for instance, is a common side effect of aripiprazole, shown in one recent trial to affect 11% of the patients. In addition, weight gain, though typically lower than seen with other antipsychotics, is a consideration with aripiprazole.
With respect to fall risk, they noted that bupropion was largely used in relatively high doses of 300 mg and 450 mg, despite some recent research showing little clinical benefit from increasing antidepressant doses above minimum recommendations.
“It is possible that smaller doses of bupropion than those used in the current trial would retain effectiveness while minimizing adverse effects such as falls,” the editorialists noted.
Commenting on the study, Jennifer R. Gatchel, MD, PhD, assistant psychiatrist at Massachusetts General Hospital/McLean Hospital and assistant professor of psychiatry at Harvard Medical School, Boston, said the findings have high clinical significance in the treatment of geriatric depression.
“These results are of great impact for clinicians managing older adults with treatment-resistant depression. They provide some of the first evidence of safety and efficacy of augmentation with aripiprazole as a strategy in clinical management of older adults who fail to initially respond to treatment,” said Dr. Gatchel, who was not associated with this research.
“Of particular significance, efficacy here is based on patient-centered outcomes and psychological well-being as a primary effectiveness outcome, which could translate into strengthened physician-patient alliance.”
While adjunctive aripiprazole is not necessarily a first-line strategy when older adults fail to respond to antidepressants, there is a lack of data on the risks and benefits of any other antipsychotic medications, she noted.
“Thus, this is evidence that will impact clinical practice and hopefully contribute to reduced societal burden of depression in older adults and the morbidity and mortality associated with it,” Dr. Gatchel said.
The study received support from a Patient-Centered Outcomes Research Institute (PCORI) Award (TRD-1511-33321). Dr. Lenze received additional support from the Taylor Family Institute for Innovative Psychiatric Research at Washington University School of Medicine, as well as the Washington University Institute of Clinical and Translational Sciences grant (UL1TR002345) from the National Center for Advancing Translational Sciences of the National Institutes of Health. Dr. Gatchel reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW ORLEANS –
“We found that adding aripiprazole led to higher rates of depression remission and greater improvements in psychological well-being – which means how positive and satisfied patients felt – and this is good news,” study investigator Eric J. Lenze, MD, of the department of psychiatry, Washington University, St. Louis, said in a press statement.
“However, even that approach helped only about 30% of people in the study with treatment-resistant depression, underscoring the need to find and develop more effective treatments that can help more people,” he added.
The findings were presented here as part of the American Association for Geriatric Psychiatry annual meeting, and published concurrently in the New England Journal of Medicine.
Need for safe treatment options
Treatment-resistant depression is common in older patients, but switching medications or adding other agents can be challenging. With higher rates of comorbidity and polypharmacy, treatment decisions in this patient population are more complex compared with those involving younger patients.
To compare the benefits of augmentation vs. drug-switching strategies, the researchers conducted a multicenter, two-step trial involving 619 patients with an average baseline age of 69 who had failed to respond to two courses of selective serotonin reuptake inhibitors (SSRIs).
Patients were randomly assigned to one of three groups. These included augmentation of existing antidepressant medication with either aripiprazole (n = 211) or the dopamine and norepinephrine–reuptake inhibitor bupropion (Wellbutrin, Zyban) (n = 206), or to taper off of their current antidepressant and switch to bupropion (n = 202).
After 10 weeks, patients’ psychological well-being was assessed via the National Institutes of Health Toolbox Positive Affect and General Life Satisfaction subscales. The researchers found patients in the aripiprazole and bupropion add-on groups improved by 4.83 points and 4.33 points, respectively. The bupropion switch group had a change of 2.04 points.
The difference between the aripiprazole augmentation group and the switch to bupropion group was significant (difference 2.79 points; P = .014). Other between-group differences were not significantly different.
Remission rates were similar in the aripiprazole and bupropion groups at 28.9% and 28.2%, respectively. The remission rate in the bupropion switch group was 19.3%.
The study results showed patients who received adjunctive bupropion had the highest fall rate at 0.55 falls per patient, vs. 0.33 falls per patient in the aripiprazole group, suggesting that among the three treatment options, adjunctive aripiprazole may be the best choice because of its superior efficacy and lower fall risk.
A total of 248 patients enrolled in the study showed no improvement and were further randomly assigned to receive adjunctive lithium (n = 127) or switch from current therapy to nortriptyline (n = 121).
Well-being scores in the lithium group improved by 3.17 points and 2.18 points in the nortriptyline group. Remission occurred in 18.9% of patients in the lithium group and 21.5% in the nortriptyline group. Fall rates were similar among the two groups.
Overall, “this large, randomized study demonstrated that adding aripiprazole was a superior option for older adults with treatment-resistant depression,” Dr. Lenze told this news organization.
“Since neither lithium nor nortriptyline were promising against treatment-resistant depression in older adults, those medications are unlikely to be helpful in most cases,” he added.
Practice changing?
In an accompanying editorial, Gemma Lewis, PhD, and Glyn Lewis, PhD, division of psychiatry, University of College London, noted the findings “support aripiprazole augmentation as a strategy for treatment-resistant depression in older persons, largely because of the lower risk of falls than with bupropion augmentation.”
However, “in clinical practice, [it] would be important to tailor treatment in light of potential adverse effects and the preferences of the patient,” they added.
Akathisia, for instance, is a common side effect of aripiprazole, shown in one recent trial to affect 11% of the patients. In addition, weight gain, though typically lower than seen with other antipsychotics, is a consideration with aripiprazole.
With respect to fall risk, they noted that bupropion was largely used in relatively high doses of 300 mg and 450 mg, despite some recent research showing little clinical benefit from increasing antidepressant doses above minimum recommendations.
“It is possible that smaller doses of bupropion than those used in the current trial would retain effectiveness while minimizing adverse effects such as falls,” the editorialists noted.
Commenting on the study, Jennifer R. Gatchel, MD, PhD, assistant psychiatrist at Massachusetts General Hospital/McLean Hospital and assistant professor of psychiatry at Harvard Medical School, Boston, said the findings have high clinical significance in the treatment of geriatric depression.
“These results are of great impact for clinicians managing older adults with treatment-resistant depression. They provide some of the first evidence of safety and efficacy of augmentation with aripiprazole as a strategy in clinical management of older adults who fail to initially respond to treatment,” said Dr. Gatchel, who was not associated with this research.
“Of particular significance, efficacy here is based on patient-centered outcomes and psychological well-being as a primary effectiveness outcome, which could translate into strengthened physician-patient alliance.”
While adjunctive aripiprazole is not necessarily a first-line strategy when older adults fail to respond to antidepressants, there is a lack of data on the risks and benefits of any other antipsychotic medications, she noted.
“Thus, this is evidence that will impact clinical practice and hopefully contribute to reduced societal burden of depression in older adults and the morbidity and mortality associated with it,” Dr. Gatchel said.
The study received support from a Patient-Centered Outcomes Research Institute (PCORI) Award (TRD-1511-33321). Dr. Lenze received additional support from the Taylor Family Institute for Innovative Psychiatric Research at Washington University School of Medicine, as well as the Washington University Institute of Clinical and Translational Sciences grant (UL1TR002345) from the National Center for Advancing Translational Sciences of the National Institutes of Health. Dr. Gatchel reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT AAGP 2023









