User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
New ABIM fees to stay listed as ‘board certified’ irk physicians
Abdul Moiz Hafiz, MD, was flabbergasted when he received a phone call from his institution’s credentialing office telling him that he was not certified for interventional cardiology – even though he had passed that exam in 2016.
Dr. Hafiz, who directs the Advanced Structural Heart Disease Program at Southern Illinois University, phoned the American Board of Internal Medicine (ABIM), where he learned that to restore his credentials, he would need to pay $1,225 in maintenance of certification (MOC) fees.
Like Dr. Hafiz,
Even doctors who are participating in mandatory continuing education outside the ABIM’s auspices are finding themselves listed as “not certified.” Some physicians learned of the policy change only after applying for hospital privileges or for jobs that require ABIM certification.
Now that increasing numbers of physicians are employed by hospitals and health care organizations that require ABIM certification, many doctors have no option but to pony up the fees if they want to continue to practice medicine.
“We have no say in the matter,” said Dr. Hafiz, “and there’s no appeal process.”
The change affects nearly 330,000 physicians. Responses to the policy on Twitter included accusations of extortion and denunciations of the ABIM’s “money grab policies.”
Sunil Rao, MD, director of interventional cardiology at NYU Langone Health and president of the Society for Cardiovascular Angiography and Interventions (SCAI), has heard from many SCAI members who had experiences similar to Dr. Hafiz’s. While Dr. Rao describes some of the Twitter outrage as “emotional,” he does acknowledge that the ABIM’s moves appear to be financially motivated.
“The issue here was that as soon as they paid the fee, all of a sudden, ABIM flipped the switch and said they were certified,” he said. “It certainly sounds like a purely financial kind of structure.”
Richard Baron, MD, president and CEO of the ABIM, said doctors are misunderstanding the policy change.
“No doctor loses certification solely for failure to pay fees,” Dr. Baron told this news organization. “What caused them to be reported as not certified was that we didn’t have evidence that they had met program requirements. They could say, ‘But I did meet program requirements, you just didn’t know it.’ To which our answer would be, for us to know it, we have to process them. And our policy is that we don’t process them unless you are current on your fees.”
This is not the first time ABIM policies have alienated physicians.
Last year, the ABIM raised its MOC fees from $165 to $220. That also prompted a wave of outrage. Other grievances go further back. At one time, being board certified was a lifetime credential. However, in 1990 the ABIM made periodic recertification mandatory.
The process, which came to be known as “maintenance of certification,” had to be completed every 10 years, and fees were charged for each certification. At that point, said Dr. Baron, the relationship between the ABIM and physicians changed from a one-time interaction to a career-long relationship. He advises doctors to check in periodically on their portal page at the ABIM or download the app so they will always know their status.
Many physicians would prefer not to be bound to a lifetime relationship with the ABIM. There is an alternative licensing board, the National Board of Physicians and Surgeons (NBPAS), but it is accepted by only a limited number of hospitals.
“Until the NBPAS gains wide recognition,” said Dr. Hafiz, “the ABIM is going to continue to have basically a monopoly over the market.”
The value of MOC itself has been called into question. “There are no direct data supporting the value of the MOC process in either improving care, making patient care safer, or making patient care higher quality,” said Dr. Rao. This feeds frustration in a clinical community already dealing with onerous training requirements and expensive board certification exams and adds to the perception that it is a purely financial transaction, he said. (Studies examining whether the MOC system improves patient care have shown mixed results.)
The true value of the ABIM to physicians, Dr. Baron contends, is that the organization is an independent third party that differentiates those doctors from people who don’t have their skills, training, and expertise. “In these days, where anyone can be an ‘expert’ on the Internet, that’s more valuable than ever before,” he said.
A version of this article first appeared on Medscape.com.
Abdul Moiz Hafiz, MD, was flabbergasted when he received a phone call from his institution’s credentialing office telling him that he was not certified for interventional cardiology – even though he had passed that exam in 2016.
Dr. Hafiz, who directs the Advanced Structural Heart Disease Program at Southern Illinois University, phoned the American Board of Internal Medicine (ABIM), where he learned that to restore his credentials, he would need to pay $1,225 in maintenance of certification (MOC) fees.
Like Dr. Hafiz,
Even doctors who are participating in mandatory continuing education outside the ABIM’s auspices are finding themselves listed as “not certified.” Some physicians learned of the policy change only after applying for hospital privileges or for jobs that require ABIM certification.
Now that increasing numbers of physicians are employed by hospitals and health care organizations that require ABIM certification, many doctors have no option but to pony up the fees if they want to continue to practice medicine.
“We have no say in the matter,” said Dr. Hafiz, “and there’s no appeal process.”
The change affects nearly 330,000 physicians. Responses to the policy on Twitter included accusations of extortion and denunciations of the ABIM’s “money grab policies.”
Sunil Rao, MD, director of interventional cardiology at NYU Langone Health and president of the Society for Cardiovascular Angiography and Interventions (SCAI), has heard from many SCAI members who had experiences similar to Dr. Hafiz’s. While Dr. Rao describes some of the Twitter outrage as “emotional,” he does acknowledge that the ABIM’s moves appear to be financially motivated.
“The issue here was that as soon as they paid the fee, all of a sudden, ABIM flipped the switch and said they were certified,” he said. “It certainly sounds like a purely financial kind of structure.”
Richard Baron, MD, president and CEO of the ABIM, said doctors are misunderstanding the policy change.
“No doctor loses certification solely for failure to pay fees,” Dr. Baron told this news organization. “What caused them to be reported as not certified was that we didn’t have evidence that they had met program requirements. They could say, ‘But I did meet program requirements, you just didn’t know it.’ To which our answer would be, for us to know it, we have to process them. And our policy is that we don’t process them unless you are current on your fees.”
This is not the first time ABIM policies have alienated physicians.
Last year, the ABIM raised its MOC fees from $165 to $220. That also prompted a wave of outrage. Other grievances go further back. At one time, being board certified was a lifetime credential. However, in 1990 the ABIM made periodic recertification mandatory.
The process, which came to be known as “maintenance of certification,” had to be completed every 10 years, and fees were charged for each certification. At that point, said Dr. Baron, the relationship between the ABIM and physicians changed from a one-time interaction to a career-long relationship. He advises doctors to check in periodically on their portal page at the ABIM or download the app so they will always know their status.
Many physicians would prefer not to be bound to a lifetime relationship with the ABIM. There is an alternative licensing board, the National Board of Physicians and Surgeons (NBPAS), but it is accepted by only a limited number of hospitals.
“Until the NBPAS gains wide recognition,” said Dr. Hafiz, “the ABIM is going to continue to have basically a monopoly over the market.”
The value of MOC itself has been called into question. “There are no direct data supporting the value of the MOC process in either improving care, making patient care safer, or making patient care higher quality,” said Dr. Rao. This feeds frustration in a clinical community already dealing with onerous training requirements and expensive board certification exams and adds to the perception that it is a purely financial transaction, he said. (Studies examining whether the MOC system improves patient care have shown mixed results.)
The true value of the ABIM to physicians, Dr. Baron contends, is that the organization is an independent third party that differentiates those doctors from people who don’t have their skills, training, and expertise. “In these days, where anyone can be an ‘expert’ on the Internet, that’s more valuable than ever before,” he said.
A version of this article first appeared on Medscape.com.
Abdul Moiz Hafiz, MD, was flabbergasted when he received a phone call from his institution’s credentialing office telling him that he was not certified for interventional cardiology – even though he had passed that exam in 2016.
Dr. Hafiz, who directs the Advanced Structural Heart Disease Program at Southern Illinois University, phoned the American Board of Internal Medicine (ABIM), where he learned that to restore his credentials, he would need to pay $1,225 in maintenance of certification (MOC) fees.
Like Dr. Hafiz,
Even doctors who are participating in mandatory continuing education outside the ABIM’s auspices are finding themselves listed as “not certified.” Some physicians learned of the policy change only after applying for hospital privileges or for jobs that require ABIM certification.
Now that increasing numbers of physicians are employed by hospitals and health care organizations that require ABIM certification, many doctors have no option but to pony up the fees if they want to continue to practice medicine.
“We have no say in the matter,” said Dr. Hafiz, “and there’s no appeal process.”
The change affects nearly 330,000 physicians. Responses to the policy on Twitter included accusations of extortion and denunciations of the ABIM’s “money grab policies.”
Sunil Rao, MD, director of interventional cardiology at NYU Langone Health and president of the Society for Cardiovascular Angiography and Interventions (SCAI), has heard from many SCAI members who had experiences similar to Dr. Hafiz’s. While Dr. Rao describes some of the Twitter outrage as “emotional,” he does acknowledge that the ABIM’s moves appear to be financially motivated.
“The issue here was that as soon as they paid the fee, all of a sudden, ABIM flipped the switch and said they were certified,” he said. “It certainly sounds like a purely financial kind of structure.”
Richard Baron, MD, president and CEO of the ABIM, said doctors are misunderstanding the policy change.
“No doctor loses certification solely for failure to pay fees,” Dr. Baron told this news organization. “What caused them to be reported as not certified was that we didn’t have evidence that they had met program requirements. They could say, ‘But I did meet program requirements, you just didn’t know it.’ To which our answer would be, for us to know it, we have to process them. And our policy is that we don’t process them unless you are current on your fees.”
This is not the first time ABIM policies have alienated physicians.
Last year, the ABIM raised its MOC fees from $165 to $220. That also prompted a wave of outrage. Other grievances go further back. At one time, being board certified was a lifetime credential. However, in 1990 the ABIM made periodic recertification mandatory.
The process, which came to be known as “maintenance of certification,” had to be completed every 10 years, and fees were charged for each certification. At that point, said Dr. Baron, the relationship between the ABIM and physicians changed from a one-time interaction to a career-long relationship. He advises doctors to check in periodically on their portal page at the ABIM or download the app so they will always know their status.
Many physicians would prefer not to be bound to a lifetime relationship with the ABIM. There is an alternative licensing board, the National Board of Physicians and Surgeons (NBPAS), but it is accepted by only a limited number of hospitals.
“Until the NBPAS gains wide recognition,” said Dr. Hafiz, “the ABIM is going to continue to have basically a monopoly over the market.”
The value of MOC itself has been called into question. “There are no direct data supporting the value of the MOC process in either improving care, making patient care safer, or making patient care higher quality,” said Dr. Rao. This feeds frustration in a clinical community already dealing with onerous training requirements and expensive board certification exams and adds to the perception that it is a purely financial transaction, he said. (Studies examining whether the MOC system improves patient care have shown mixed results.)
The true value of the ABIM to physicians, Dr. Baron contends, is that the organization is an independent third party that differentiates those doctors from people who don’t have their skills, training, and expertise. “In these days, where anyone can be an ‘expert’ on the Internet, that’s more valuable than ever before,” he said.
A version of this article first appeared on Medscape.com.
BMI is a flawed measure of obesity. What are alternatives?
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
Meta-analysis examines cancer risk concern for JAK inhibitors
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
AT BSR 2023
Registry data ‘reassure’ on biologics’ heart attack risk in rheumatoid arthritis
MANCHESTER, ENGLAND – Rheumatoid arthritis patients are no more likely to have a heart attack if they are treated with an interleukin-6 inhibitor (IL-6i) than if they are treated with a tumor necrosis factor inhibitor (TNFi), according to data presented at the British Society for Rheumatology annual meeting.
Results of a large analysis from the long-running British Society for Rheumatology Biologics Register–Rheumatoid Arthritis (BSRBR-RA) found no statistical difference in the rate of myocardial infarction (MI), considering treatment in almost 21,000 patients. The overall propensity-score adjusted hazard ratio for MI risk comparing TNFi and IL-6i was 0.77, but the 95% confidence interval crossed the line for statistical significance.
“This result reassures patients and clinical teams about the long-term treatment effects on myocardial infarction in a real-world setting,” said Tian Zixing, a PhD student at the University of Manchester (England).
“Patients with rheumatoid arthritis have an increased risk of myocardial infarction, compared to the general population,” Ms. Tian explained. However, this risk has been “considerably improved” with biologic treatment of rheumatoid arthritis, notably with the TNFi drugs vs. nonbiologic disease-modifying antirheumatic drugs.
The reasoning behind the current analysis was to see if there was any risk associated with IL-6i, as these drugs have been noted to increase low-density cholesterol levels, which in turn can raise the risk for MI.
The study population consisted of all patients registered in the BSRBR-RA over the past 20 years who had started treatment with one of the many TNFi drugs available in the UK – adalimumab (Humira and biosimilars), etanercept (Enbrel), infliximab (Remicade and biosimilars), certolizumab pegol (Cimzia), and golimumab (Simponi) – or the two available drugs that target the effects of IL-6 – tocilizumab (RoActemra, but Actemra in the U.S.), which targets IL-6 itself, and sarilumab (Kevzara), which targets the IL-6 receptor.
Clinical follow-up forms, death certificates, and patient reports confirmed by the clinical team were used to identify patients who experienced a MI, but only MIs that occurred while on treatment were counted.
More than 30,000 lines of therapy in 20,898 patients were recorded. Ms. Tian noted that most (> 90%) patients had been treated with a TNFi across all lines of therapy.
“It is very important to consider the treatment sequence,” she said. “Most patients start first-line treatment with a TNF inhibitor, with only a few patients starting an IL-6 inhibitor,” she noted. “IL-6 inhibitors are more commonly used in the later stages of disease, when more cardiovascular risk factors have accumulated.”
Thus, to ensure that the MI risk was fairly evaluated, the statistical analyses compared TNFi and IL-6i according to the line of treatment. “That means only patients on their first-line treatment will be compared to each other, and only those on their second-line treatment will be compared to each other, and so on,” Ms. Tian explained.
Baseline characteristics were broadly similar for patients treated with TNFi and IL-6i drugs, except for hyperlipidemia, which was higher in patients treated with an IL-6i. Nevertheless, there was no suggestion of any difference in the MI rates after adjustment for cardiovascular risk factors.
There are a lot of strengths to these data, but of course the possibilities of residual confounding and confounding by indication exist, Ms. Tian said. There were also missing data that had to be imputed.
“There has been quite a bit around interleukin-1 blockers being cardiovascular protective,” observed Kenneth Baker, MBChB, PhD, who chaired the RA oral abstracts session during which Ms. Tian presented the findings.
“IL-6 is quite good at suppressing CRP [C-reactive protein],” added Dr. Baker, a senior clinical research fellow at Newcastle University and honorary consultant rheumatologist at Freeman Hospital, both in Newcastle upon Tyne, England.
“You’ve hypothesized or extrapolated that the differences in the lipid levels may not be relevant,” he said to Ms. Tian, “but do you think there might be an extra element going on here?” Maybe IL-6i drugs such as tocilizumab are better at suppressing inflammation, and that counterbalances the effects on lipids, he suggested.
Ms. Tian and Dr. Baker disclosed no relevant financial relationships. The BSRBR-RA is managed by the University of Manchester on behalf of the British Society for Rheumatology. The registry is supported by funding from multiple pharmaceutical companies, including AbbVie, Amgen, Celltrion Healthcare, Eli Lilly, Galapagos, Pfizer, Samsung Bioepis, and Sanofi, and in the past Hospira, Merck Sharp & Dohme, Roche, Sandoz, SOBI, and UCB.
A version of this article originally appeared on Medscape.com.
MANCHESTER, ENGLAND – Rheumatoid arthritis patients are no more likely to have a heart attack if they are treated with an interleukin-6 inhibitor (IL-6i) than if they are treated with a tumor necrosis factor inhibitor (TNFi), according to data presented at the British Society for Rheumatology annual meeting.
Results of a large analysis from the long-running British Society for Rheumatology Biologics Register–Rheumatoid Arthritis (BSRBR-RA) found no statistical difference in the rate of myocardial infarction (MI), considering treatment in almost 21,000 patients. The overall propensity-score adjusted hazard ratio for MI risk comparing TNFi and IL-6i was 0.77, but the 95% confidence interval crossed the line for statistical significance.
“This result reassures patients and clinical teams about the long-term treatment effects on myocardial infarction in a real-world setting,” said Tian Zixing, a PhD student at the University of Manchester (England).
“Patients with rheumatoid arthritis have an increased risk of myocardial infarction, compared to the general population,” Ms. Tian explained. However, this risk has been “considerably improved” with biologic treatment of rheumatoid arthritis, notably with the TNFi drugs vs. nonbiologic disease-modifying antirheumatic drugs.
The reasoning behind the current analysis was to see if there was any risk associated with IL-6i, as these drugs have been noted to increase low-density cholesterol levels, which in turn can raise the risk for MI.
The study population consisted of all patients registered in the BSRBR-RA over the past 20 years who had started treatment with one of the many TNFi drugs available in the UK – adalimumab (Humira and biosimilars), etanercept (Enbrel), infliximab (Remicade and biosimilars), certolizumab pegol (Cimzia), and golimumab (Simponi) – or the two available drugs that target the effects of IL-6 – tocilizumab (RoActemra, but Actemra in the U.S.), which targets IL-6 itself, and sarilumab (Kevzara), which targets the IL-6 receptor.
Clinical follow-up forms, death certificates, and patient reports confirmed by the clinical team were used to identify patients who experienced a MI, but only MIs that occurred while on treatment were counted.
More than 30,000 lines of therapy in 20,898 patients were recorded. Ms. Tian noted that most (> 90%) patients had been treated with a TNFi across all lines of therapy.
“It is very important to consider the treatment sequence,” she said. “Most patients start first-line treatment with a TNF inhibitor, with only a few patients starting an IL-6 inhibitor,” she noted. “IL-6 inhibitors are more commonly used in the later stages of disease, when more cardiovascular risk factors have accumulated.”
Thus, to ensure that the MI risk was fairly evaluated, the statistical analyses compared TNFi and IL-6i according to the line of treatment. “That means only patients on their first-line treatment will be compared to each other, and only those on their second-line treatment will be compared to each other, and so on,” Ms. Tian explained.
Baseline characteristics were broadly similar for patients treated with TNFi and IL-6i drugs, except for hyperlipidemia, which was higher in patients treated with an IL-6i. Nevertheless, there was no suggestion of any difference in the MI rates after adjustment for cardiovascular risk factors.
There are a lot of strengths to these data, but of course the possibilities of residual confounding and confounding by indication exist, Ms. Tian said. There were also missing data that had to be imputed.
“There has been quite a bit around interleukin-1 blockers being cardiovascular protective,” observed Kenneth Baker, MBChB, PhD, who chaired the RA oral abstracts session during which Ms. Tian presented the findings.
“IL-6 is quite good at suppressing CRP [C-reactive protein],” added Dr. Baker, a senior clinical research fellow at Newcastle University and honorary consultant rheumatologist at Freeman Hospital, both in Newcastle upon Tyne, England.
“You’ve hypothesized or extrapolated that the differences in the lipid levels may not be relevant,” he said to Ms. Tian, “but do you think there might be an extra element going on here?” Maybe IL-6i drugs such as tocilizumab are better at suppressing inflammation, and that counterbalances the effects on lipids, he suggested.
Ms. Tian and Dr. Baker disclosed no relevant financial relationships. The BSRBR-RA is managed by the University of Manchester on behalf of the British Society for Rheumatology. The registry is supported by funding from multiple pharmaceutical companies, including AbbVie, Amgen, Celltrion Healthcare, Eli Lilly, Galapagos, Pfizer, Samsung Bioepis, and Sanofi, and in the past Hospira, Merck Sharp & Dohme, Roche, Sandoz, SOBI, and UCB.
A version of this article originally appeared on Medscape.com.
MANCHESTER, ENGLAND – Rheumatoid arthritis patients are no more likely to have a heart attack if they are treated with an interleukin-6 inhibitor (IL-6i) than if they are treated with a tumor necrosis factor inhibitor (TNFi), according to data presented at the British Society for Rheumatology annual meeting.
Results of a large analysis from the long-running British Society for Rheumatology Biologics Register–Rheumatoid Arthritis (BSRBR-RA) found no statistical difference in the rate of myocardial infarction (MI), considering treatment in almost 21,000 patients. The overall propensity-score adjusted hazard ratio for MI risk comparing TNFi and IL-6i was 0.77, but the 95% confidence interval crossed the line for statistical significance.
“This result reassures patients and clinical teams about the long-term treatment effects on myocardial infarction in a real-world setting,” said Tian Zixing, a PhD student at the University of Manchester (England).
“Patients with rheumatoid arthritis have an increased risk of myocardial infarction, compared to the general population,” Ms. Tian explained. However, this risk has been “considerably improved” with biologic treatment of rheumatoid arthritis, notably with the TNFi drugs vs. nonbiologic disease-modifying antirheumatic drugs.
The reasoning behind the current analysis was to see if there was any risk associated with IL-6i, as these drugs have been noted to increase low-density cholesterol levels, which in turn can raise the risk for MI.
The study population consisted of all patients registered in the BSRBR-RA over the past 20 years who had started treatment with one of the many TNFi drugs available in the UK – adalimumab (Humira and biosimilars), etanercept (Enbrel), infliximab (Remicade and biosimilars), certolizumab pegol (Cimzia), and golimumab (Simponi) – or the two available drugs that target the effects of IL-6 – tocilizumab (RoActemra, but Actemra in the U.S.), which targets IL-6 itself, and sarilumab (Kevzara), which targets the IL-6 receptor.
Clinical follow-up forms, death certificates, and patient reports confirmed by the clinical team were used to identify patients who experienced a MI, but only MIs that occurred while on treatment were counted.
More than 30,000 lines of therapy in 20,898 patients were recorded. Ms. Tian noted that most (> 90%) patients had been treated with a TNFi across all lines of therapy.
“It is very important to consider the treatment sequence,” she said. “Most patients start first-line treatment with a TNF inhibitor, with only a few patients starting an IL-6 inhibitor,” she noted. “IL-6 inhibitors are more commonly used in the later stages of disease, when more cardiovascular risk factors have accumulated.”
Thus, to ensure that the MI risk was fairly evaluated, the statistical analyses compared TNFi and IL-6i according to the line of treatment. “That means only patients on their first-line treatment will be compared to each other, and only those on their second-line treatment will be compared to each other, and so on,” Ms. Tian explained.
Baseline characteristics were broadly similar for patients treated with TNFi and IL-6i drugs, except for hyperlipidemia, which was higher in patients treated with an IL-6i. Nevertheless, there was no suggestion of any difference in the MI rates after adjustment for cardiovascular risk factors.
There are a lot of strengths to these data, but of course the possibilities of residual confounding and confounding by indication exist, Ms. Tian said. There were also missing data that had to be imputed.
“There has been quite a bit around interleukin-1 blockers being cardiovascular protective,” observed Kenneth Baker, MBChB, PhD, who chaired the RA oral abstracts session during which Ms. Tian presented the findings.
“IL-6 is quite good at suppressing CRP [C-reactive protein],” added Dr. Baker, a senior clinical research fellow at Newcastle University and honorary consultant rheumatologist at Freeman Hospital, both in Newcastle upon Tyne, England.
“You’ve hypothesized or extrapolated that the differences in the lipid levels may not be relevant,” he said to Ms. Tian, “but do you think there might be an extra element going on here?” Maybe IL-6i drugs such as tocilizumab are better at suppressing inflammation, and that counterbalances the effects on lipids, he suggested.
Ms. Tian and Dr. Baker disclosed no relevant financial relationships. The BSRBR-RA is managed by the University of Manchester on behalf of the British Society for Rheumatology. The registry is supported by funding from multiple pharmaceutical companies, including AbbVie, Amgen, Celltrion Healthcare, Eli Lilly, Galapagos, Pfizer, Samsung Bioepis, and Sanofi, and in the past Hospira, Merck Sharp & Dohme, Roche, Sandoz, SOBI, and UCB.
A version of this article originally appeared on Medscape.com.
AT BSR 2023
Proposed Medicare bill would raise docs’ pay with inflation
Introduced by four physician U.S. House representatives, HR 2474 would link Medicare fee schedule updates to the Medicare Economic Index, a measure of inflation related to physicians’ practice costs and wages.
That’s a long-sought goal of the American Medical Association, which is leading 120 state medical societies and medical specialty groups in championing the bill.
The legislation is essential to enabling physician practices to better absorb payment distributions triggered by budget neutrality rules, performance adjustments, and periods of high inflation, the groups wrote in a joint letter sent to the bill’s sponsors. The sponsors say they hope the legislation will improve access to care, as low reimbursements cause some physicians to limit their number of Medicare patients.
Physicians groups for years have urged federal lawmakers to scrap short-term fixes staving off Medicare pay cuts in favor of permanent reforms. Unlike nearly all other Medicare clinicians including hospitals, physicians’ Medicare payment updates aren’t currently tied to inflation.
Adjusted for inflation, Medicare payments to physicians have declined 26% between 2001 and 2023, including a 2% payment reduction in 2023, according to the AMA. Small and rural physician practices have been disproportionately affected by these reductions, as have doctors treating low-income or uninsured patients, the AMA said.
Last month, an influential federal advisory panel recommended permanently tying Medicare physician pay increases to inflation. Clinicians’ cost of providing services, measured by the Medicare Economic Index, rose by 2.6% in 2021 and are estimated to have risen 4.7% in 2022, significantly more than in recent years, the Medicare Payment Advisory Commission said.
A version of this article originally appeared on Medscape.com.
Introduced by four physician U.S. House representatives, HR 2474 would link Medicare fee schedule updates to the Medicare Economic Index, a measure of inflation related to physicians’ practice costs and wages.
That’s a long-sought goal of the American Medical Association, which is leading 120 state medical societies and medical specialty groups in championing the bill.
The legislation is essential to enabling physician practices to better absorb payment distributions triggered by budget neutrality rules, performance adjustments, and periods of high inflation, the groups wrote in a joint letter sent to the bill’s sponsors. The sponsors say they hope the legislation will improve access to care, as low reimbursements cause some physicians to limit their number of Medicare patients.
Physicians groups for years have urged federal lawmakers to scrap short-term fixes staving off Medicare pay cuts in favor of permanent reforms. Unlike nearly all other Medicare clinicians including hospitals, physicians’ Medicare payment updates aren’t currently tied to inflation.
Adjusted for inflation, Medicare payments to physicians have declined 26% between 2001 and 2023, including a 2% payment reduction in 2023, according to the AMA. Small and rural physician practices have been disproportionately affected by these reductions, as have doctors treating low-income or uninsured patients, the AMA said.
Last month, an influential federal advisory panel recommended permanently tying Medicare physician pay increases to inflation. Clinicians’ cost of providing services, measured by the Medicare Economic Index, rose by 2.6% in 2021 and are estimated to have risen 4.7% in 2022, significantly more than in recent years, the Medicare Payment Advisory Commission said.
A version of this article originally appeared on Medscape.com.
Introduced by four physician U.S. House representatives, HR 2474 would link Medicare fee schedule updates to the Medicare Economic Index, a measure of inflation related to physicians’ practice costs and wages.
That’s a long-sought goal of the American Medical Association, which is leading 120 state medical societies and medical specialty groups in championing the bill.
The legislation is essential to enabling physician practices to better absorb payment distributions triggered by budget neutrality rules, performance adjustments, and periods of high inflation, the groups wrote in a joint letter sent to the bill’s sponsors. The sponsors say they hope the legislation will improve access to care, as low reimbursements cause some physicians to limit their number of Medicare patients.
Physicians groups for years have urged federal lawmakers to scrap short-term fixes staving off Medicare pay cuts in favor of permanent reforms. Unlike nearly all other Medicare clinicians including hospitals, physicians’ Medicare payment updates aren’t currently tied to inflation.
Adjusted for inflation, Medicare payments to physicians have declined 26% between 2001 and 2023, including a 2% payment reduction in 2023, according to the AMA. Small and rural physician practices have been disproportionately affected by these reductions, as have doctors treating low-income or uninsured patients, the AMA said.
Last month, an influential federal advisory panel recommended permanently tying Medicare physician pay increases to inflation. Clinicians’ cost of providing services, measured by the Medicare Economic Index, rose by 2.6% in 2021 and are estimated to have risen 4.7% in 2022, significantly more than in recent years, the Medicare Payment Advisory Commission said.
A version of this article originally appeared on Medscape.com.
Surgeons, intensivists earn more than do colleagues from private insurance
General and orthopedic surgeons and intensivists earn the highest net reimbursements from private U.S. insurers, a new report estimates.
On average in 2021, they were paid $5.8 million, $4.9 million, and $3.3 million, respectively, according to figures compiled by AMN Healthcare, a Dallas-based health staffing company.
None of 15 other physician specialties topped $3 million in net reimbursement on average, and three – dermatology, pediatrics, and family medicine – didn’t reach $1 million.
The report doesn’t include data about reimbursement from Medicare and Medicaid, and its numbers assume that 50% of insurance claims are denied. Denial rates differ from practice to practice.
Still, the findings offer a “benchmark tool” to help clinicians understand how they rank against their peers, Linda Murphy, president of AMN Healthcare’s Revenue Cycle Solutions division, said in an interview.
This is the first year that the company has calculated physician reimbursement levels by using claim and clearinghouse data, Ms. Murphy said. Previously, a division of the firm compiled data by surveying chief financial officers from hospitals.
The report’s estimate that insurers deny 50% of claims is “conservative,” Ms. Murphy said. Miscoding is a significant factor behind that number.
The estimated 2021 net private insurance reimbursements by specialty for direct services, assuming a 50% denial rate:
- Anesthesiology: $1,665,510
- Cardiology: $1,703,013
- Critical Care (intensivist): $3,338,656
- Dermatology: $729,107
- Family medicine: $697,094
- Gastroenterology: $2,765,110
- Internal medicine: $1,297,200
- Neurology: $1,390,181
- Obstetrician/gynecology: $1,880,888
- Otolaryngology: $2,095,277
- Pediatrics: $661,552
- Psychiatry: $1,348,730
- Pulmonology: $1,561,617
- Radiology: $1,015,750
- Rheumatology: $1,705,140
- General surgery: $5,834,508
- Orthopedic surgery: $4,904,757
- Urology: $2,943,381
Among 18 physician specialties overall, the report estimated that the average net reimbursement in 2021 was $1.9 million.
The report also estimated that the net reimbursement amounts at $875,140 for certified registered nurse anesthetists and $388,696 for nurse practitioners.
Surprisingly, Ms. Murphy said, there’s “a really large swing” among reimbursement levels for individual specialties. The quartile of cardiologists with the lowest level of reimbursement, for example, submitted $2.1 million in claims in 2021, netting about $1 million at a 50% denial rate versus the $7.3 million made by those in the highest quartile, netting about $3.6 million.
The gap seems to be due to regional variations, she said, adding that a rural cardiologist will have different billing practices than does one practicing in New York City.
The quartile of general surgeons with the highest reimbursement levels billed for $21.1 million on average in 2021, making about $10.5 million at a 50% denial rate. The lowest quartile billed for $5.5 million, making about $2.7 million at a 50% denial rate.
The report noted that primary care physicians – that is, family medicine, internal medicine, and pediatrics specialists – have much lower levels of reimbursement, compared with most other specialties. But the work of primary care physicians “may lead to considerable ‘downstream revenue’ through the hospital admissions, tests and treatment they order.”
A previous analysis by a division of AMN Healthcare found that primary care physicians, on average, generate $2,113,273 a year in net annual revenue for their affiliated hospitals, nearing the $2,446,429 in net annual hospital revenue generated by specialists.
AMN Healthcare is preparing another report that will examine Medicare reimbursements, Ms. Murphy said. According to the new report, payments by nonprivate insurers amount to about one-third of the total amount of reimbursement by commercial insurers.
A version of this article originally appeared on Medscape.com.
General and orthopedic surgeons and intensivists earn the highest net reimbursements from private U.S. insurers, a new report estimates.
On average in 2021, they were paid $5.8 million, $4.9 million, and $3.3 million, respectively, according to figures compiled by AMN Healthcare, a Dallas-based health staffing company.
None of 15 other physician specialties topped $3 million in net reimbursement on average, and three – dermatology, pediatrics, and family medicine – didn’t reach $1 million.
The report doesn’t include data about reimbursement from Medicare and Medicaid, and its numbers assume that 50% of insurance claims are denied. Denial rates differ from practice to practice.
Still, the findings offer a “benchmark tool” to help clinicians understand how they rank against their peers, Linda Murphy, president of AMN Healthcare’s Revenue Cycle Solutions division, said in an interview.
This is the first year that the company has calculated physician reimbursement levels by using claim and clearinghouse data, Ms. Murphy said. Previously, a division of the firm compiled data by surveying chief financial officers from hospitals.
The report’s estimate that insurers deny 50% of claims is “conservative,” Ms. Murphy said. Miscoding is a significant factor behind that number.
The estimated 2021 net private insurance reimbursements by specialty for direct services, assuming a 50% denial rate:
- Anesthesiology: $1,665,510
- Cardiology: $1,703,013
- Critical Care (intensivist): $3,338,656
- Dermatology: $729,107
- Family medicine: $697,094
- Gastroenterology: $2,765,110
- Internal medicine: $1,297,200
- Neurology: $1,390,181
- Obstetrician/gynecology: $1,880,888
- Otolaryngology: $2,095,277
- Pediatrics: $661,552
- Psychiatry: $1,348,730
- Pulmonology: $1,561,617
- Radiology: $1,015,750
- Rheumatology: $1,705,140
- General surgery: $5,834,508
- Orthopedic surgery: $4,904,757
- Urology: $2,943,381
Among 18 physician specialties overall, the report estimated that the average net reimbursement in 2021 was $1.9 million.
The report also estimated that the net reimbursement amounts at $875,140 for certified registered nurse anesthetists and $388,696 for nurse practitioners.
Surprisingly, Ms. Murphy said, there’s “a really large swing” among reimbursement levels for individual specialties. The quartile of cardiologists with the lowest level of reimbursement, for example, submitted $2.1 million in claims in 2021, netting about $1 million at a 50% denial rate versus the $7.3 million made by those in the highest quartile, netting about $3.6 million.
The gap seems to be due to regional variations, she said, adding that a rural cardiologist will have different billing practices than does one practicing in New York City.
The quartile of general surgeons with the highest reimbursement levels billed for $21.1 million on average in 2021, making about $10.5 million at a 50% denial rate. The lowest quartile billed for $5.5 million, making about $2.7 million at a 50% denial rate.
The report noted that primary care physicians – that is, family medicine, internal medicine, and pediatrics specialists – have much lower levels of reimbursement, compared with most other specialties. But the work of primary care physicians “may lead to considerable ‘downstream revenue’ through the hospital admissions, tests and treatment they order.”
A previous analysis by a division of AMN Healthcare found that primary care physicians, on average, generate $2,113,273 a year in net annual revenue for their affiliated hospitals, nearing the $2,446,429 in net annual hospital revenue generated by specialists.
AMN Healthcare is preparing another report that will examine Medicare reimbursements, Ms. Murphy said. According to the new report, payments by nonprivate insurers amount to about one-third of the total amount of reimbursement by commercial insurers.
A version of this article originally appeared on Medscape.com.
General and orthopedic surgeons and intensivists earn the highest net reimbursements from private U.S. insurers, a new report estimates.
On average in 2021, they were paid $5.8 million, $4.9 million, and $3.3 million, respectively, according to figures compiled by AMN Healthcare, a Dallas-based health staffing company.
None of 15 other physician specialties topped $3 million in net reimbursement on average, and three – dermatology, pediatrics, and family medicine – didn’t reach $1 million.
The report doesn’t include data about reimbursement from Medicare and Medicaid, and its numbers assume that 50% of insurance claims are denied. Denial rates differ from practice to practice.
Still, the findings offer a “benchmark tool” to help clinicians understand how they rank against their peers, Linda Murphy, president of AMN Healthcare’s Revenue Cycle Solutions division, said in an interview.
This is the first year that the company has calculated physician reimbursement levels by using claim and clearinghouse data, Ms. Murphy said. Previously, a division of the firm compiled data by surveying chief financial officers from hospitals.
The report’s estimate that insurers deny 50% of claims is “conservative,” Ms. Murphy said. Miscoding is a significant factor behind that number.
The estimated 2021 net private insurance reimbursements by specialty for direct services, assuming a 50% denial rate:
- Anesthesiology: $1,665,510
- Cardiology: $1,703,013
- Critical Care (intensivist): $3,338,656
- Dermatology: $729,107
- Family medicine: $697,094
- Gastroenterology: $2,765,110
- Internal medicine: $1,297,200
- Neurology: $1,390,181
- Obstetrician/gynecology: $1,880,888
- Otolaryngology: $2,095,277
- Pediatrics: $661,552
- Psychiatry: $1,348,730
- Pulmonology: $1,561,617
- Radiology: $1,015,750
- Rheumatology: $1,705,140
- General surgery: $5,834,508
- Orthopedic surgery: $4,904,757
- Urology: $2,943,381
Among 18 physician specialties overall, the report estimated that the average net reimbursement in 2021 was $1.9 million.
The report also estimated that the net reimbursement amounts at $875,140 for certified registered nurse anesthetists and $388,696 for nurse practitioners.
Surprisingly, Ms. Murphy said, there’s “a really large swing” among reimbursement levels for individual specialties. The quartile of cardiologists with the lowest level of reimbursement, for example, submitted $2.1 million in claims in 2021, netting about $1 million at a 50% denial rate versus the $7.3 million made by those in the highest quartile, netting about $3.6 million.
The gap seems to be due to regional variations, she said, adding that a rural cardiologist will have different billing practices than does one practicing in New York City.
The quartile of general surgeons with the highest reimbursement levels billed for $21.1 million on average in 2021, making about $10.5 million at a 50% denial rate. The lowest quartile billed for $5.5 million, making about $2.7 million at a 50% denial rate.
The report noted that primary care physicians – that is, family medicine, internal medicine, and pediatrics specialists – have much lower levels of reimbursement, compared with most other specialties. But the work of primary care physicians “may lead to considerable ‘downstream revenue’ through the hospital admissions, tests and treatment they order.”
A previous analysis by a division of AMN Healthcare found that primary care physicians, on average, generate $2,113,273 a year in net annual revenue for their affiliated hospitals, nearing the $2,446,429 in net annual hospital revenue generated by specialists.
AMN Healthcare is preparing another report that will examine Medicare reimbursements, Ms. Murphy said. According to the new report, payments by nonprivate insurers amount to about one-third of the total amount of reimbursement by commercial insurers.
A version of this article originally appeared on Medscape.com.
Long COVID mobile monitoring study hunts for answers
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
NPF provides guidance for virtual psoriasis visits
.
The success of telemedicine in managing chronic inflammatory skin conditions including psoriasis during the COVID-19 pandemic “highlighted that teledermatology can be used beyond the context of a global health crisis to provide continuity of care and improve access to health care more broadly,” the task force wrote in a paper published online in JAAD International.
Co–senior author George Han, MD, PhD, said in an interview that the impetus for the guidelines came from NPF patient advocates, who realized that the organization needed something to take to payers and governmental agencies to advocate for better access to dermatologic care. He is associate professor of dermatology and director of teledermatology at the Hofstra/Northwell department of dermatology, Hyde Park, New York.
“We realized that, in many places around the country, people don’t have access to dermatology.” In upstate New York, said Dr. Han, his anecdotal research has revealed wait times of 6 months or more.
As a guiding principle, the authors pronounce teledermatology “a reasonable alternative for providing long-term management of patients with psoriasis.” Research shows that nearly all dermatologists used teledermatology during the pandemic, the authors noted, and that well-run programs improve Psoriasis Area and Severity Index (PASI) scores and other measures on par with in-person care. Telemedicine may be especially useful for initial visits, they added, particularly when distance, patient incapacity, and circumstances prevent face-to-face evaluation.
Additional position statements emphasize that teledermatology should support rather than supplant in-person visits, and that this balance may be particularly important in cases involving psoriatic arthritis (PsA). “Even though we can’t do a physical exam and palpate some of those joints in person,” said Dr. Han, “tools have been developed that, through a series of questions the patient can answer, can guide you towards whether there is a high index of suspicion for psoriatic arthritis.” Such patients require in-person evaluation with urgency, he said, because delays in PsA diagnosis and treatment can lead to irreversible joint damage and significant functional impairment.
Another motivation for producing the guidelines, said Dr. Han, was that, even when underserved patients get a dermatology appointment, some providers may not have all the latest tools or medicines available for treating psoriasis. In such cases, telemedicine may allow dermatologists specializing in psoriasis care to extend their reach in comanaging patients with primary care physicians and community dermatologists.
Before the appointment, guidelines suggest determining what form of teledermatology will best suit each patient. Authors recommended gauging patients’ savviness with computers and cameras, and counseling patients regarding available virtual evaluation tools – such as live video visits, store-and-forward photo strategies, and assessment-tool training videos.
A subsequent guideline underscores the importance of continuously improving technology to support expeditious image capture and workflows that emulate in-person practice. Dr. Han explained, “we wanted to make sure that on the back end there’s adequate support such that – if through teledermatology, we determine that the patient should get, say, a systemic treatment – the patient is able to get the appropriate lab tests, get the medicine, and know how to inject it.”
Regarding reimbursement, Dr. Han said that policies varied prepandemic, but many commercial insurers covered telemedicine at a rate 20% lower than the in-person rate. During the pandemic, he said, insurers shifted to provide the higher rate for telemedicine, consistent with policies adopted by the Centers for Medicare & Medicaid Services.
“There are differences in coverage and reimbursement from plan to plan,” Dr. Han added. “And even within the same plan, there are carve-outs so that some plans don’t allow certain services. The big picture is that for the most part these services are covered at a level comparable to an in-person visit at present.”
With the Department of Health & Human Services’ public health emergency declaration expiring in May, he said, physicians have worried that some of the allowances made by CMS – such as lifting requirements that Medicare patients in rural areas be seen at care sites – will expire. “It seems that some of those limitations have been addressed, and those allowances are going to be extended until Congress is able to pass something that gives us durable access to telemedicine care. We think that based on the current environment telemedicine is here to stay.”
The study was funded by the NPF. Dr. Han has been an investigator, adviser, speaker, or researcher for AbbVie, Amgen, Apogee Therapeutics, Arcutis, Athenex, Bausch Health, Beiersdorf, Boehringer Ingelheim, Bond Avillion, Bristol Myers Squibb, Celgene, CeraVe, Dermavant, DermTech, Eli Lilly, EPI Health, Janssen Pharmaceuticals, LEO Pharma, L’Oreal, MC2 Therapeutics, Novartis, Ortho Dermatologics, PellePharm, Pfizer, Regeneron Pharmaceuticals, Sanofi Genzyme, SUN Pharmaceuticals, and UCB.
.
The success of telemedicine in managing chronic inflammatory skin conditions including psoriasis during the COVID-19 pandemic “highlighted that teledermatology can be used beyond the context of a global health crisis to provide continuity of care and improve access to health care more broadly,” the task force wrote in a paper published online in JAAD International.
Co–senior author George Han, MD, PhD, said in an interview that the impetus for the guidelines came from NPF patient advocates, who realized that the organization needed something to take to payers and governmental agencies to advocate for better access to dermatologic care. He is associate professor of dermatology and director of teledermatology at the Hofstra/Northwell department of dermatology, Hyde Park, New York.
“We realized that, in many places around the country, people don’t have access to dermatology.” In upstate New York, said Dr. Han, his anecdotal research has revealed wait times of 6 months or more.
As a guiding principle, the authors pronounce teledermatology “a reasonable alternative for providing long-term management of patients with psoriasis.” Research shows that nearly all dermatologists used teledermatology during the pandemic, the authors noted, and that well-run programs improve Psoriasis Area and Severity Index (PASI) scores and other measures on par with in-person care. Telemedicine may be especially useful for initial visits, they added, particularly when distance, patient incapacity, and circumstances prevent face-to-face evaluation.
Additional position statements emphasize that teledermatology should support rather than supplant in-person visits, and that this balance may be particularly important in cases involving psoriatic arthritis (PsA). “Even though we can’t do a physical exam and palpate some of those joints in person,” said Dr. Han, “tools have been developed that, through a series of questions the patient can answer, can guide you towards whether there is a high index of suspicion for psoriatic arthritis.” Such patients require in-person evaluation with urgency, he said, because delays in PsA diagnosis and treatment can lead to irreversible joint damage and significant functional impairment.
Another motivation for producing the guidelines, said Dr. Han, was that, even when underserved patients get a dermatology appointment, some providers may not have all the latest tools or medicines available for treating psoriasis. In such cases, telemedicine may allow dermatologists specializing in psoriasis care to extend their reach in comanaging patients with primary care physicians and community dermatologists.
Before the appointment, guidelines suggest determining what form of teledermatology will best suit each patient. Authors recommended gauging patients’ savviness with computers and cameras, and counseling patients regarding available virtual evaluation tools – such as live video visits, store-and-forward photo strategies, and assessment-tool training videos.
A subsequent guideline underscores the importance of continuously improving technology to support expeditious image capture and workflows that emulate in-person practice. Dr. Han explained, “we wanted to make sure that on the back end there’s adequate support such that – if through teledermatology, we determine that the patient should get, say, a systemic treatment – the patient is able to get the appropriate lab tests, get the medicine, and know how to inject it.”
Regarding reimbursement, Dr. Han said that policies varied prepandemic, but many commercial insurers covered telemedicine at a rate 20% lower than the in-person rate. During the pandemic, he said, insurers shifted to provide the higher rate for telemedicine, consistent with policies adopted by the Centers for Medicare & Medicaid Services.
“There are differences in coverage and reimbursement from plan to plan,” Dr. Han added. “And even within the same plan, there are carve-outs so that some plans don’t allow certain services. The big picture is that for the most part these services are covered at a level comparable to an in-person visit at present.”
With the Department of Health & Human Services’ public health emergency declaration expiring in May, he said, physicians have worried that some of the allowances made by CMS – such as lifting requirements that Medicare patients in rural areas be seen at care sites – will expire. “It seems that some of those limitations have been addressed, and those allowances are going to be extended until Congress is able to pass something that gives us durable access to telemedicine care. We think that based on the current environment telemedicine is here to stay.”
The study was funded by the NPF. Dr. Han has been an investigator, adviser, speaker, or researcher for AbbVie, Amgen, Apogee Therapeutics, Arcutis, Athenex, Bausch Health, Beiersdorf, Boehringer Ingelheim, Bond Avillion, Bristol Myers Squibb, Celgene, CeraVe, Dermavant, DermTech, Eli Lilly, EPI Health, Janssen Pharmaceuticals, LEO Pharma, L’Oreal, MC2 Therapeutics, Novartis, Ortho Dermatologics, PellePharm, Pfizer, Regeneron Pharmaceuticals, Sanofi Genzyme, SUN Pharmaceuticals, and UCB.
.
The success of telemedicine in managing chronic inflammatory skin conditions including psoriasis during the COVID-19 pandemic “highlighted that teledermatology can be used beyond the context of a global health crisis to provide continuity of care and improve access to health care more broadly,” the task force wrote in a paper published online in JAAD International.
Co–senior author George Han, MD, PhD, said in an interview that the impetus for the guidelines came from NPF patient advocates, who realized that the organization needed something to take to payers and governmental agencies to advocate for better access to dermatologic care. He is associate professor of dermatology and director of teledermatology at the Hofstra/Northwell department of dermatology, Hyde Park, New York.
“We realized that, in many places around the country, people don’t have access to dermatology.” In upstate New York, said Dr. Han, his anecdotal research has revealed wait times of 6 months or more.
As a guiding principle, the authors pronounce teledermatology “a reasonable alternative for providing long-term management of patients with psoriasis.” Research shows that nearly all dermatologists used teledermatology during the pandemic, the authors noted, and that well-run programs improve Psoriasis Area and Severity Index (PASI) scores and other measures on par with in-person care. Telemedicine may be especially useful for initial visits, they added, particularly when distance, patient incapacity, and circumstances prevent face-to-face evaluation.
Additional position statements emphasize that teledermatology should support rather than supplant in-person visits, and that this balance may be particularly important in cases involving psoriatic arthritis (PsA). “Even though we can’t do a physical exam and palpate some of those joints in person,” said Dr. Han, “tools have been developed that, through a series of questions the patient can answer, can guide you towards whether there is a high index of suspicion for psoriatic arthritis.” Such patients require in-person evaluation with urgency, he said, because delays in PsA diagnosis and treatment can lead to irreversible joint damage and significant functional impairment.
Another motivation for producing the guidelines, said Dr. Han, was that, even when underserved patients get a dermatology appointment, some providers may not have all the latest tools or medicines available for treating psoriasis. In such cases, telemedicine may allow dermatologists specializing in psoriasis care to extend their reach in comanaging patients with primary care physicians and community dermatologists.
Before the appointment, guidelines suggest determining what form of teledermatology will best suit each patient. Authors recommended gauging patients’ savviness with computers and cameras, and counseling patients regarding available virtual evaluation tools – such as live video visits, store-and-forward photo strategies, and assessment-tool training videos.
A subsequent guideline underscores the importance of continuously improving technology to support expeditious image capture and workflows that emulate in-person practice. Dr. Han explained, “we wanted to make sure that on the back end there’s adequate support such that – if through teledermatology, we determine that the patient should get, say, a systemic treatment – the patient is able to get the appropriate lab tests, get the medicine, and know how to inject it.”
Regarding reimbursement, Dr. Han said that policies varied prepandemic, but many commercial insurers covered telemedicine at a rate 20% lower than the in-person rate. During the pandemic, he said, insurers shifted to provide the higher rate for telemedicine, consistent with policies adopted by the Centers for Medicare & Medicaid Services.
“There are differences in coverage and reimbursement from plan to plan,” Dr. Han added. “And even within the same plan, there are carve-outs so that some plans don’t allow certain services. The big picture is that for the most part these services are covered at a level comparable to an in-person visit at present.”
With the Department of Health & Human Services’ public health emergency declaration expiring in May, he said, physicians have worried that some of the allowances made by CMS – such as lifting requirements that Medicare patients in rural areas be seen at care sites – will expire. “It seems that some of those limitations have been addressed, and those allowances are going to be extended until Congress is able to pass something that gives us durable access to telemedicine care. We think that based on the current environment telemedicine is here to stay.”
The study was funded by the NPF. Dr. Han has been an investigator, adviser, speaker, or researcher for AbbVie, Amgen, Apogee Therapeutics, Arcutis, Athenex, Bausch Health, Beiersdorf, Boehringer Ingelheim, Bond Avillion, Bristol Myers Squibb, Celgene, CeraVe, Dermavant, DermTech, Eli Lilly, EPI Health, Janssen Pharmaceuticals, LEO Pharma, L’Oreal, MC2 Therapeutics, Novartis, Ortho Dermatologics, PellePharm, Pfizer, Regeneron Pharmaceuticals, Sanofi Genzyme, SUN Pharmaceuticals, and UCB.
FROM JAAD INTERNATIONAL
Bone-bashing effects of air pollution becoming clearer
We have long recognized that our environment has a significant impact on our general health. Air pollution is known to contribute to respiratory conditions, poor cardiovascular outcomes, and certain kinds of cancer.
It’s increasingly important to identify factors that might contribute to suboptimal bone density and associated fracture risk in the population as a whole, and particularly in older adults. Aging is associated with a higher risk for osteoporosis and fractures, with their attendant morbidity, but individuals differ in their extent of bone loss and risk for fractures.
Known factors affecting bone health include genetics, age, sex, nutrition, physical activity, and hormonal factors. Certain medications, diseases, and lifestyle choices – such as smoking and alcohol intake – can also have deleterious effects on bone.
More recently, researchers have started examining the impact of air pollution on bone health.
As we know, the degree of pollution varies greatly from one region to another and can potentially significantly affect life in many parts of the world. In fact, the World Health Organization indicates that 99% of the world’s population breathes air exceeding the WHO guideline limits for pollutants.
Air pollutants include particulate matter (PM) as well as gases, such as nitric oxide, nitrogen dioxide, ammonia, carbon monoxide, sulfur dioxide, ozone, and certain volatile organic compounds. Particulate pollutants include a variety of substances produced from mostly human activities (such as vehicle emissions, biofuel combustion, mining, agriculture, and manufacturing, and also forest fires). They are classified not by their composition, but by their size (for example, PM1.0, PM2.5, and PM10 indicate PM with a diameter < 1.0, 2.5, and 10 microns, respectively). The finer the particle, the more likely it is to cross into the systemic circulation from the respiratory tract, with the potential to induce oxidative, inflammatory, and other changes in the body.
Many studies report that air pollution is a risk factor for osteoporosis. Some have found associations of lower bone density, osteoporosis, and fracture risk with higher concentrations of PM1.0, PM2.5, or PM10, even after controlling for other factors that could affect bone health. Some researchers have reported that although they didn’t find a significant association between PM and bone health, they did find an association between distance from the freeway and bone health – thus, exposure to polycyclic aromatic hydrocarbons and black carbon from vehicle emissions needs to be studied as a contributor to fracture risk.
Importantly, a prospective, observational study from the Women’s Health Initiative (which included more than 9,000 ethnically diverse women from three sites in the United States) reported a significant negative impact of PM10, nitric oxide, nitrogen dioxide, and sulfur dioxide over 1, 3, and 5 years on bone density at multiple sites, and particularly at the lumbar spine, in both cross-sectional and longitudinal analyses after controlling for demographic and socioeconomic factors. This study reported that nitrogen dioxide exposure may be a key determinant of bone density at the lumbar spine and in the whole body. Similarly, other studies have reported associations between atmospheric nitrogen dioxide or sulfur dioxide and risk for osteoporotic fractures.
Why the impact on bones?
The potential negative impact of pollution on bone has been attributed to many factors. PM induces systemic inflammation and an increase in cytokines that stimulate bone cells (osteoclasts) that cause bone loss. Other pollutants (gases and metal compounds) can cause oxidative damage to bone cells, whereas others act as endocrine disrupters and affect the functioning of these cells.
Pollution might also affect the synthesis and metabolism of vitamin D, which is necessary for absorption of calcium from the gut. High rates of pollution can reduce the amount of ultraviolet radiation reaching the earth which is important because certain wavelengths of ultraviolet radiation are necessary for vitamin D synthesis in our skin. Reduced vitamin D synthesis in skin can lead to poorly mineralized bone unless there is sufficient intake of vitamin D in diet or as supplements. Also, the conversion of vitamin D to its active form happens in the kidneys, and PM can be harmful to renal function. PM is also believed to cause increased breakdown of vitamin D into its inactive form.
Conversely, some studies have reported no association between pollution and bone density or osteoporosis risk, and two meta-analyses indicated that the association between the two is inconsistent. Some factors explaining variances in results include the number of individuals included in the study (larger studies are generally considered to be more reproducible), the fact that most studies are cross-sectional and not prospective, many do not control for other factors that might be deleterious to bone, and prediction models for the extent of PM or other exposure may not be completely accurate.
However, another recent meta-analysis reported an increased risk for lower total-body bone density and hip fracture after exposure to air pollution, particularly PM2.5 and nitrogen dioxide, but not to PM10, nitric oxide, or ozone. More studies are needed to confirm, or refute, the association between air pollution and impaired bone health. But accumulating evidence suggests that air pollution very likely has a deleterious effect on bone.
When feasible, it’s important to avoid living or working in areas with poor air quality and high pollution rates. However, this isn’t always possible based on one’s occupation, geography, circumstances, or economic status. Therefore, attention to a cleaner environment is critical at both the individual and the macro level.
As an example of the latter, the city of London extended its ultralow emission zone (ULEZ) farther out of the city in October 2021, and a further expansion is planned to include all of the city’s boroughs in August 2023.
We can do our bit by driving less and walking, biking, or using public transportation more often. We can also turn off the car engine when it’s not running, maintain our vehicles, switch to electric or hand-powered yard equipment, and not burn household garbage and limit backyard fires. We can also switch from gas to solar energy or wind, use efficient appliances and heating, and avoid unnecessary energy use. And we can choose sustainable products when possible.
For optimal bone health, we should remind patients to eat a healthy diet with the requisite amount of protein, calcium, and vitamin D. Vitamin D and calcium supplementation may be necessary for people whose intake of dairy and dairy products is low. Other important strategies to optimize bone health include engaging in healthy physical activity; avoiding smoking or excessive alcohol intake; and treating underlying gastrointestinal, endocrine, or other conditions that can reduce bone density.
Madhusmita Misra, MD, MPH, is the chief of the division of pediatric endocrinology, Mass General for Children; the associate director of the Harvard Catalyst Translation and Clinical Research Center; and the director of the Pediatric Endocrine-Sports Endocrine-Neuroendocrine Lab, Mass General Hospital, Boston.
A version of this article first appeared on Medscape.com.
We have long recognized that our environment has a significant impact on our general health. Air pollution is known to contribute to respiratory conditions, poor cardiovascular outcomes, and certain kinds of cancer.
It’s increasingly important to identify factors that might contribute to suboptimal bone density and associated fracture risk in the population as a whole, and particularly in older adults. Aging is associated with a higher risk for osteoporosis and fractures, with their attendant morbidity, but individuals differ in their extent of bone loss and risk for fractures.
Known factors affecting bone health include genetics, age, sex, nutrition, physical activity, and hormonal factors. Certain medications, diseases, and lifestyle choices – such as smoking and alcohol intake – can also have deleterious effects on bone.
More recently, researchers have started examining the impact of air pollution on bone health.
As we know, the degree of pollution varies greatly from one region to another and can potentially significantly affect life in many parts of the world. In fact, the World Health Organization indicates that 99% of the world’s population breathes air exceeding the WHO guideline limits for pollutants.
Air pollutants include particulate matter (PM) as well as gases, such as nitric oxide, nitrogen dioxide, ammonia, carbon monoxide, sulfur dioxide, ozone, and certain volatile organic compounds. Particulate pollutants include a variety of substances produced from mostly human activities (such as vehicle emissions, biofuel combustion, mining, agriculture, and manufacturing, and also forest fires). They are classified not by their composition, but by their size (for example, PM1.0, PM2.5, and PM10 indicate PM with a diameter < 1.0, 2.5, and 10 microns, respectively). The finer the particle, the more likely it is to cross into the systemic circulation from the respiratory tract, with the potential to induce oxidative, inflammatory, and other changes in the body.
Many studies report that air pollution is a risk factor for osteoporosis. Some have found associations of lower bone density, osteoporosis, and fracture risk with higher concentrations of PM1.0, PM2.5, or PM10, even after controlling for other factors that could affect bone health. Some researchers have reported that although they didn’t find a significant association between PM and bone health, they did find an association between distance from the freeway and bone health – thus, exposure to polycyclic aromatic hydrocarbons and black carbon from vehicle emissions needs to be studied as a contributor to fracture risk.
Importantly, a prospective, observational study from the Women’s Health Initiative (which included more than 9,000 ethnically diverse women from three sites in the United States) reported a significant negative impact of PM10, nitric oxide, nitrogen dioxide, and sulfur dioxide over 1, 3, and 5 years on bone density at multiple sites, and particularly at the lumbar spine, in both cross-sectional and longitudinal analyses after controlling for demographic and socioeconomic factors. This study reported that nitrogen dioxide exposure may be a key determinant of bone density at the lumbar spine and in the whole body. Similarly, other studies have reported associations between atmospheric nitrogen dioxide or sulfur dioxide and risk for osteoporotic fractures.
Why the impact on bones?
The potential negative impact of pollution on bone has been attributed to many factors. PM induces systemic inflammation and an increase in cytokines that stimulate bone cells (osteoclasts) that cause bone loss. Other pollutants (gases and metal compounds) can cause oxidative damage to bone cells, whereas others act as endocrine disrupters and affect the functioning of these cells.
Pollution might also affect the synthesis and metabolism of vitamin D, which is necessary for absorption of calcium from the gut. High rates of pollution can reduce the amount of ultraviolet radiation reaching the earth which is important because certain wavelengths of ultraviolet radiation are necessary for vitamin D synthesis in our skin. Reduced vitamin D synthesis in skin can lead to poorly mineralized bone unless there is sufficient intake of vitamin D in diet or as supplements. Also, the conversion of vitamin D to its active form happens in the kidneys, and PM can be harmful to renal function. PM is also believed to cause increased breakdown of vitamin D into its inactive form.
Conversely, some studies have reported no association between pollution and bone density or osteoporosis risk, and two meta-analyses indicated that the association between the two is inconsistent. Some factors explaining variances in results include the number of individuals included in the study (larger studies are generally considered to be more reproducible), the fact that most studies are cross-sectional and not prospective, many do not control for other factors that might be deleterious to bone, and prediction models for the extent of PM or other exposure may not be completely accurate.
However, another recent meta-analysis reported an increased risk for lower total-body bone density and hip fracture after exposure to air pollution, particularly PM2.5 and nitrogen dioxide, but not to PM10, nitric oxide, or ozone. More studies are needed to confirm, or refute, the association between air pollution and impaired bone health. But accumulating evidence suggests that air pollution very likely has a deleterious effect on bone.
When feasible, it’s important to avoid living or working in areas with poor air quality and high pollution rates. However, this isn’t always possible based on one’s occupation, geography, circumstances, or economic status. Therefore, attention to a cleaner environment is critical at both the individual and the macro level.
As an example of the latter, the city of London extended its ultralow emission zone (ULEZ) farther out of the city in October 2021, and a further expansion is planned to include all of the city’s boroughs in August 2023.
We can do our bit by driving less and walking, biking, or using public transportation more often. We can also turn off the car engine when it’s not running, maintain our vehicles, switch to electric or hand-powered yard equipment, and not burn household garbage and limit backyard fires. We can also switch from gas to solar energy or wind, use efficient appliances and heating, and avoid unnecessary energy use. And we can choose sustainable products when possible.
For optimal bone health, we should remind patients to eat a healthy diet with the requisite amount of protein, calcium, and vitamin D. Vitamin D and calcium supplementation may be necessary for people whose intake of dairy and dairy products is low. Other important strategies to optimize bone health include engaging in healthy physical activity; avoiding smoking or excessive alcohol intake; and treating underlying gastrointestinal, endocrine, or other conditions that can reduce bone density.
Madhusmita Misra, MD, MPH, is the chief of the division of pediatric endocrinology, Mass General for Children; the associate director of the Harvard Catalyst Translation and Clinical Research Center; and the director of the Pediatric Endocrine-Sports Endocrine-Neuroendocrine Lab, Mass General Hospital, Boston.
A version of this article first appeared on Medscape.com.
We have long recognized that our environment has a significant impact on our general health. Air pollution is known to contribute to respiratory conditions, poor cardiovascular outcomes, and certain kinds of cancer.
It’s increasingly important to identify factors that might contribute to suboptimal bone density and associated fracture risk in the population as a whole, and particularly in older adults. Aging is associated with a higher risk for osteoporosis and fractures, with their attendant morbidity, but individuals differ in their extent of bone loss and risk for fractures.
Known factors affecting bone health include genetics, age, sex, nutrition, physical activity, and hormonal factors. Certain medications, diseases, and lifestyle choices – such as smoking and alcohol intake – can also have deleterious effects on bone.
More recently, researchers have started examining the impact of air pollution on bone health.
As we know, the degree of pollution varies greatly from one region to another and can potentially significantly affect life in many parts of the world. In fact, the World Health Organization indicates that 99% of the world’s population breathes air exceeding the WHO guideline limits for pollutants.
Air pollutants include particulate matter (PM) as well as gases, such as nitric oxide, nitrogen dioxide, ammonia, carbon monoxide, sulfur dioxide, ozone, and certain volatile organic compounds. Particulate pollutants include a variety of substances produced from mostly human activities (such as vehicle emissions, biofuel combustion, mining, agriculture, and manufacturing, and also forest fires). They are classified not by their composition, but by their size (for example, PM1.0, PM2.5, and PM10 indicate PM with a diameter < 1.0, 2.5, and 10 microns, respectively). The finer the particle, the more likely it is to cross into the systemic circulation from the respiratory tract, with the potential to induce oxidative, inflammatory, and other changes in the body.
Many studies report that air pollution is a risk factor for osteoporosis. Some have found associations of lower bone density, osteoporosis, and fracture risk with higher concentrations of PM1.0, PM2.5, or PM10, even after controlling for other factors that could affect bone health. Some researchers have reported that although they didn’t find a significant association between PM and bone health, they did find an association between distance from the freeway and bone health – thus, exposure to polycyclic aromatic hydrocarbons and black carbon from vehicle emissions needs to be studied as a contributor to fracture risk.
Importantly, a prospective, observational study from the Women’s Health Initiative (which included more than 9,000 ethnically diverse women from three sites in the United States) reported a significant negative impact of PM10, nitric oxide, nitrogen dioxide, and sulfur dioxide over 1, 3, and 5 years on bone density at multiple sites, and particularly at the lumbar spine, in both cross-sectional and longitudinal analyses after controlling for demographic and socioeconomic factors. This study reported that nitrogen dioxide exposure may be a key determinant of bone density at the lumbar spine and in the whole body. Similarly, other studies have reported associations between atmospheric nitrogen dioxide or sulfur dioxide and risk for osteoporotic fractures.
Why the impact on bones?
The potential negative impact of pollution on bone has been attributed to many factors. PM induces systemic inflammation and an increase in cytokines that stimulate bone cells (osteoclasts) that cause bone loss. Other pollutants (gases and metal compounds) can cause oxidative damage to bone cells, whereas others act as endocrine disrupters and affect the functioning of these cells.
Pollution might also affect the synthesis and metabolism of vitamin D, which is necessary for absorption of calcium from the gut. High rates of pollution can reduce the amount of ultraviolet radiation reaching the earth which is important because certain wavelengths of ultraviolet radiation are necessary for vitamin D synthesis in our skin. Reduced vitamin D synthesis in skin can lead to poorly mineralized bone unless there is sufficient intake of vitamin D in diet or as supplements. Also, the conversion of vitamin D to its active form happens in the kidneys, and PM can be harmful to renal function. PM is also believed to cause increased breakdown of vitamin D into its inactive form.
Conversely, some studies have reported no association between pollution and bone density or osteoporosis risk, and two meta-analyses indicated that the association between the two is inconsistent. Some factors explaining variances in results include the number of individuals included in the study (larger studies are generally considered to be more reproducible), the fact that most studies are cross-sectional and not prospective, many do not control for other factors that might be deleterious to bone, and prediction models for the extent of PM or other exposure may not be completely accurate.
However, another recent meta-analysis reported an increased risk for lower total-body bone density and hip fracture after exposure to air pollution, particularly PM2.5 and nitrogen dioxide, but not to PM10, nitric oxide, or ozone. More studies are needed to confirm, or refute, the association between air pollution and impaired bone health. But accumulating evidence suggests that air pollution very likely has a deleterious effect on bone.
When feasible, it’s important to avoid living or working in areas with poor air quality and high pollution rates. However, this isn’t always possible based on one’s occupation, geography, circumstances, or economic status. Therefore, attention to a cleaner environment is critical at both the individual and the macro level.
As an example of the latter, the city of London extended its ultralow emission zone (ULEZ) farther out of the city in October 2021, and a further expansion is planned to include all of the city’s boroughs in August 2023.
We can do our bit by driving less and walking, biking, or using public transportation more often. We can also turn off the car engine when it’s not running, maintain our vehicles, switch to electric or hand-powered yard equipment, and not burn household garbage and limit backyard fires. We can also switch from gas to solar energy or wind, use efficient appliances and heating, and avoid unnecessary energy use. And we can choose sustainable products when possible.
For optimal bone health, we should remind patients to eat a healthy diet with the requisite amount of protein, calcium, and vitamin D. Vitamin D and calcium supplementation may be necessary for people whose intake of dairy and dairy products is low. Other important strategies to optimize bone health include engaging in healthy physical activity; avoiding smoking or excessive alcohol intake; and treating underlying gastrointestinal, endocrine, or other conditions that can reduce bone density.
Madhusmita Misra, MD, MPH, is the chief of the division of pediatric endocrinology, Mass General for Children; the associate director of the Harvard Catalyst Translation and Clinical Research Center; and the director of the Pediatric Endocrine-Sports Endocrine-Neuroendocrine Lab, Mass General Hospital, Boston.
A version of this article first appeared on Medscape.com.
Infographic: Is your compensation rising as fast as your peers?
Did doctors’ salaries continue their zesty postpandemic rise in 2022? Are female physicians making pay gains versus their male counterparts that spark optimism for the future?
reveals which medical specialties pay better than others, and evaluates the current gender pay gap in medicine. If you’re interested in delving deeper into the data, check out Your Income vs. Your Peers’: Physician Compensation Report 2023.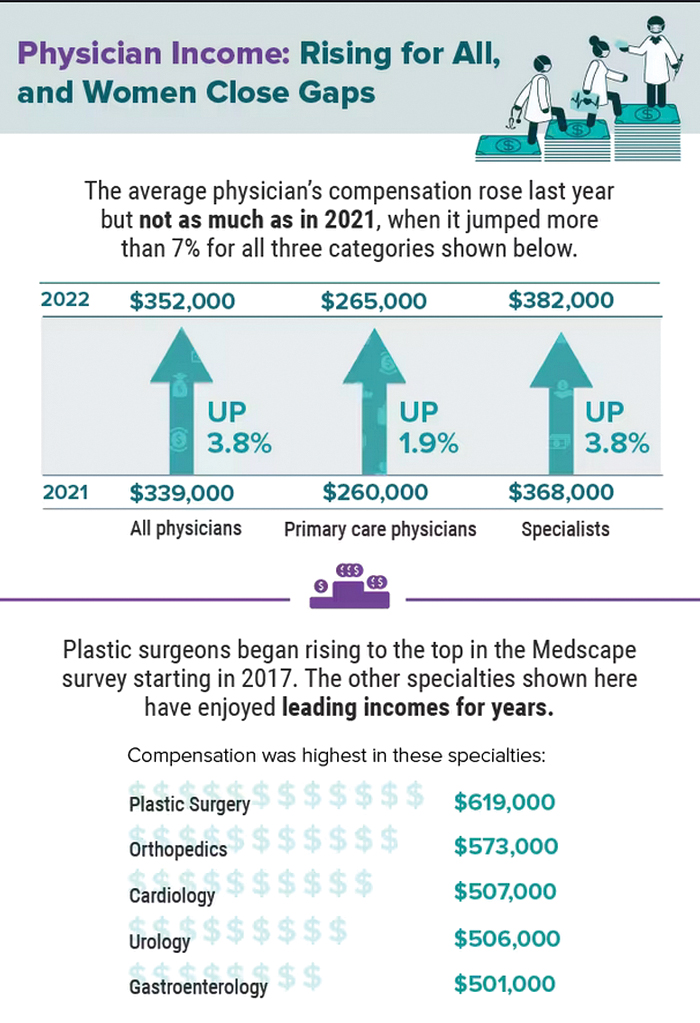
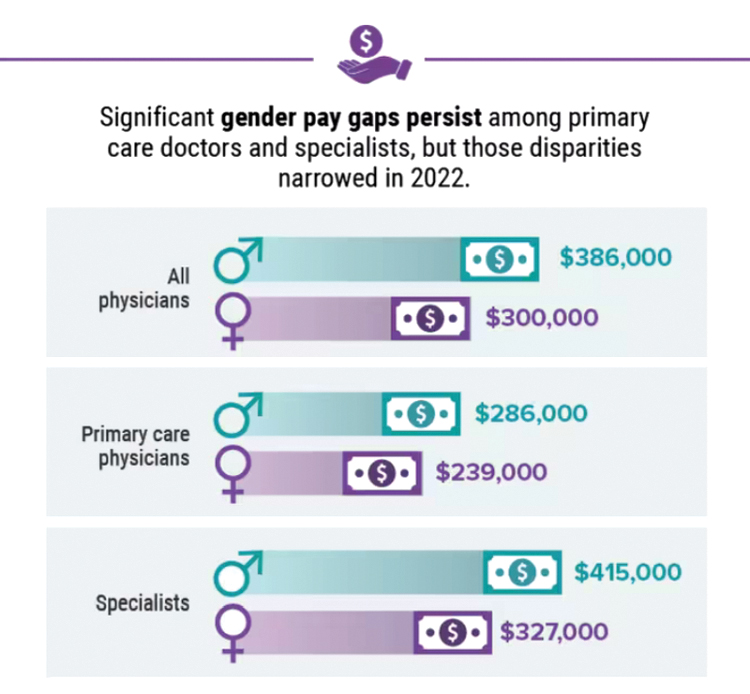
A version of this article first appeared on Medscape.com.
Did doctors’ salaries continue their zesty postpandemic rise in 2022? Are female physicians making pay gains versus their male counterparts that spark optimism for the future?
reveals which medical specialties pay better than others, and evaluates the current gender pay gap in medicine. If you’re interested in delving deeper into the data, check out Your Income vs. Your Peers’: Physician Compensation Report 2023.

A version of this article first appeared on Medscape.com.
Did doctors’ salaries continue their zesty postpandemic rise in 2022? Are female physicians making pay gains versus their male counterparts that spark optimism for the future?
reveals which medical specialties pay better than others, and evaluates the current gender pay gap in medicine. If you’re interested in delving deeper into the data, check out Your Income vs. Your Peers’: Physician Compensation Report 2023.

A version of this article first appeared on Medscape.com.









