User login
Addressing malnutrition and improving performance
Stakeholders develop a malnutrition Toolkit
Hospitalists are key players in improving hospital performance, but they may be overlooking a leading cause of morbidity and mortality, especially among older adults.
Research suggests that at the time of hospital admission, some 20%-50% of all patients are at risk for malnutrition or are malnourished, but only 7% of those patients are diagnosed during their stay, according to research cited in an abstract presented at HM17.1
“Because individuals who are malnourished lack sufficient nutrients to promote healing and rehabilitation, and are at increased risk of medical complications, it can have a serious impact on patient safety indicators, such as rates of pressure ulcers, wound healing, and risk of falls,” said lead author Eleanor Fitall of Avalere Health. “Early identification and subsequent treatment of these patients is the best way to prevent this risk.”
To address the issue, Avalere Health and the Academy of Nutrition and Dietetics established the Malnutrition Quality Improvement Initiative (MQii), a multi-stakeholder effort to identify tools to support hospital-based care teams in improving malnutrition care quality. They developed a malnutrition Toolkit, which was piloted in 2016 and was shown to effectively improve malnutrition care.
“Since the poster presentation in May, we have successfully implemented the Toolkit at 50 hospitals via a multi-hospital Learning Collaborative,” Ms. Fitall said. They are now recruiting hospitals and health systems to participate in an expanded Learning Collaborative. Interested sites should contact the MQii team at [email protected].
“By supporting efforts to improve malnutrition care in the inpatient setting, hospitalists can help reduce the incidence of these problems as well as decrease rates of readmissions and reduce patient lengths of stay,” Ms. Fitall said. “Hospitalists are critical to addressing malnutrition care gaps in the hospital. Dietitians that have undertaken malnutrition quality improvement projects using the MQii Toolkit have found that they are most successful when hospitalists are actively engaged in the team, particularly when looking to improve the rate of malnutrition diagnosis. Hospitalists are ideally positioned to champion these efforts.”
Support for MQii was provided by Abbott, she said.
Reference
1. Fitall E, Bruno M, Jones K, Lynch J, Silver H, Godamunne K, Valladares A, Mitchell K. Malnutrition Care: “Low Hanging Fruit” for Hospitalist Clinical Performance Improvement [abstract]. J Hosp Med. 2017;12(suppl 2).
Stakeholders develop a malnutrition Toolkit
Stakeholders develop a malnutrition Toolkit
Hospitalists are key players in improving hospital performance, but they may be overlooking a leading cause of morbidity and mortality, especially among older adults.
Research suggests that at the time of hospital admission, some 20%-50% of all patients are at risk for malnutrition or are malnourished, but only 7% of those patients are diagnosed during their stay, according to research cited in an abstract presented at HM17.1
“Because individuals who are malnourished lack sufficient nutrients to promote healing and rehabilitation, and are at increased risk of medical complications, it can have a serious impact on patient safety indicators, such as rates of pressure ulcers, wound healing, and risk of falls,” said lead author Eleanor Fitall of Avalere Health. “Early identification and subsequent treatment of these patients is the best way to prevent this risk.”
To address the issue, Avalere Health and the Academy of Nutrition and Dietetics established the Malnutrition Quality Improvement Initiative (MQii), a multi-stakeholder effort to identify tools to support hospital-based care teams in improving malnutrition care quality. They developed a malnutrition Toolkit, which was piloted in 2016 and was shown to effectively improve malnutrition care.
“Since the poster presentation in May, we have successfully implemented the Toolkit at 50 hospitals via a multi-hospital Learning Collaborative,” Ms. Fitall said. They are now recruiting hospitals and health systems to participate in an expanded Learning Collaborative. Interested sites should contact the MQii team at [email protected].
“By supporting efforts to improve malnutrition care in the inpatient setting, hospitalists can help reduce the incidence of these problems as well as decrease rates of readmissions and reduce patient lengths of stay,” Ms. Fitall said. “Hospitalists are critical to addressing malnutrition care gaps in the hospital. Dietitians that have undertaken malnutrition quality improvement projects using the MQii Toolkit have found that they are most successful when hospitalists are actively engaged in the team, particularly when looking to improve the rate of malnutrition diagnosis. Hospitalists are ideally positioned to champion these efforts.”
Support for MQii was provided by Abbott, she said.
Reference
1. Fitall E, Bruno M, Jones K, Lynch J, Silver H, Godamunne K, Valladares A, Mitchell K. Malnutrition Care: “Low Hanging Fruit” for Hospitalist Clinical Performance Improvement [abstract]. J Hosp Med. 2017;12(suppl 2).
Hospitalists are key players in improving hospital performance, but they may be overlooking a leading cause of morbidity and mortality, especially among older adults.
Research suggests that at the time of hospital admission, some 20%-50% of all patients are at risk for malnutrition or are malnourished, but only 7% of those patients are diagnosed during their stay, according to research cited in an abstract presented at HM17.1
“Because individuals who are malnourished lack sufficient nutrients to promote healing and rehabilitation, and are at increased risk of medical complications, it can have a serious impact on patient safety indicators, such as rates of pressure ulcers, wound healing, and risk of falls,” said lead author Eleanor Fitall of Avalere Health. “Early identification and subsequent treatment of these patients is the best way to prevent this risk.”
To address the issue, Avalere Health and the Academy of Nutrition and Dietetics established the Malnutrition Quality Improvement Initiative (MQii), a multi-stakeholder effort to identify tools to support hospital-based care teams in improving malnutrition care quality. They developed a malnutrition Toolkit, which was piloted in 2016 and was shown to effectively improve malnutrition care.
“Since the poster presentation in May, we have successfully implemented the Toolkit at 50 hospitals via a multi-hospital Learning Collaborative,” Ms. Fitall said. They are now recruiting hospitals and health systems to participate in an expanded Learning Collaborative. Interested sites should contact the MQii team at [email protected].
“By supporting efforts to improve malnutrition care in the inpatient setting, hospitalists can help reduce the incidence of these problems as well as decrease rates of readmissions and reduce patient lengths of stay,” Ms. Fitall said. “Hospitalists are critical to addressing malnutrition care gaps in the hospital. Dietitians that have undertaken malnutrition quality improvement projects using the MQii Toolkit have found that they are most successful when hospitalists are actively engaged in the team, particularly when looking to improve the rate of malnutrition diagnosis. Hospitalists are ideally positioned to champion these efforts.”
Support for MQii was provided by Abbott, she said.
Reference
1. Fitall E, Bruno M, Jones K, Lynch J, Silver H, Godamunne K, Valladares A, Mitchell K. Malnutrition Care: “Low Hanging Fruit” for Hospitalist Clinical Performance Improvement [abstract]. J Hosp Med. 2017;12(suppl 2).
Quick Byte: PrEP advances
There are recent advances in preexposure prophylaxis, or PrEP, as a promising prevention option for HIV, according to a recent study.1
Reference
1. Desai M, Field N, Grant R, McCormack S. “Recent advances in pre-exposure prophylaxis for HIV.” BMJ. 2017;359:j5011.
There are recent advances in preexposure prophylaxis, or PrEP, as a promising prevention option for HIV, according to a recent study.1
Reference
1. Desai M, Field N, Grant R, McCormack S. “Recent advances in pre-exposure prophylaxis for HIV.” BMJ. 2017;359:j5011.
There are recent advances in preexposure prophylaxis, or PrEP, as a promising prevention option for HIV, according to a recent study.1
Reference
1. Desai M, Field N, Grant R, McCormack S. “Recent advances in pre-exposure prophylaxis for HIV.” BMJ. 2017;359:j5011.
Biomarker duo rapidly identifies serious bacterial infections
MALMO, SWEDEN – The combination of serum procalcitonin and C-reactive protein levels upon admission to a pediatric ICU displayed high utility for early diagnosis of serious bacterial infection in critically ill children in a large prospective observational study presented at the annual meeting of the European Society for Paediatric Infectious Diseases.
This winning combination significantly outperformed neutrophil gelatinase-associated lipocalin, activated partial thromboplastin time, and resistin, both individually and in various combinations, for the vital task of making a rapid distinction between infectious and noninfectious causes of pediatric systemic inflammatory response syndrome, reported Enitan D. Carrol, MD, professor of pediatric infection at the University of Liverpool (England).
“One of the clinical dilemmas we face in intensive care is being able to differentiate between infectious and noninfectious causes of systemic inflammatory response syndrome. This is important because we need to identify which children have life-threatening infections so that we can promptly initiate antimicrobial therapy,” she explained.
One in four deaths in pediatric ICUs are infection related, Dr. Carrol noted.
“There is an urgent need for infection markers which, firstly, change early in the course of bacterial infection, secondly, correlate with real-time clinical progression, and thirdly, have a rapid turn-around time to allow effective clinical decision making,” she observed.
The combination of procalcitonin and C-reactive protein (CRP) levels measured at admission fits the bill, Dr. Carrol continued. Of the five biomarkers evaluated in her study – all backed by some supporting evidence of efficacy in earlier studies – the top two individual performers in terms of negative predictive value (NPV) were a CRP less than 4.2 mg/dL with a negative NPV of 99%, and a procalcitonin less than 1.52 ng/mL with an NPV of 96%. The positive predictive value of each of the biomarkers was 37%. The sensitivity and specificity of procalcitonin for diagnosis of serious bacterial infection were 78% and 80%, respectively. For CRP, the figures were 93% and 76%.
The combination of procalcitonin and CRP outperformed a multitude of other two-, three-, and four-biomarker combinations tested, with an area under the curve of 93% for combined sensitivity and specificity.
The study included 657 children admitted to the pediatric ICU at Alder Hey Children’s Hospital in Liverpool with systemic inflammatory response syndrome. All had blood samples measured for the five biomarkers on days 1-7. Clinicians were blinded as to the biomarker results. Ninety-two (14%) patients were ultimately found to have a serious bacterial infection – essentially, bacterial meningitis or septic shock – and 565 (86%) had a nonbacterial etiology.
The 28-day mortality rate was 9% in the group with serious bacterial infection, significantly higher than the 2% rate in the group with other causes of their systemic inflammatory response syndrome.
Longitudinal trends in procalcitonin and CRP as evidenced in the study can be used in clinical decision making, according to Dr. Carrol. Mean values of procalcitonin plummeted by 80% from day 1 to day 5 in response to antimicrobial therapy in the group with serious bacterial infections. In contrast, CRP levels rose sharply from day 1 to a peak on day 2, then fell, although the 50% drop from day 2 to day 5 in response to antimicrobial therapy wasn’t as pronounced as the change in procalcitonin.
“There is an additive benefit for both biomarkers compared with CRP alone. The problem with CRP on admission, as I’ve demonstrated in this study, is it often hasn’t risen yet early after admission. So although it gave the best area under the curve of any of the biomarkers, I think that combined with procalcitonin you get a much better descriminator,” Dr. Carrol said.
The median duration of ICU stay in the patients with serious bacterial infection at admission was 5 days, compared with 3 days when the cause of systemic inflammatory response syndrome lay elsewhere. Their median duration of ventilation was significantly longer, too: 4 days versus 2 in children without a serious bacterial infection.
Stepwise logistic regression analysis pinpointed several clinical variables as being associated with prolonged ICU stay.
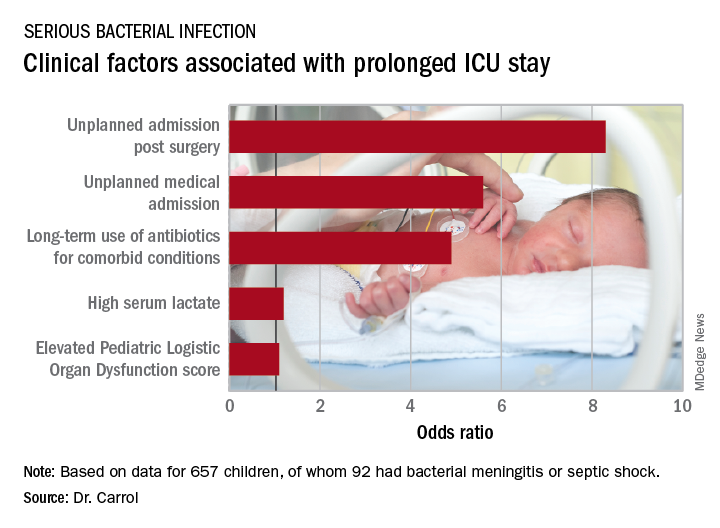
In addition, initiation of antibiotic therapy prior to admission to the pediatric ICU was associated with a 50% reduction in the likelihood of a prolonged ICU stay. “This reflects the fact that early antibiotics give you a better prognosis if you have sepsis,” according to Dr. Carrol.
She and her coinvestigators now have embarked on a multicenter U.K. study looking at the impact of procalcitonin to guide duration of antimicrobial therapy in critically ill children.
The Alder Hey study was funded by the U.K. National Institute for Health Research. Dr. Carrol reported having no financial conflicts. Although she serves as a consultant to several health care companies, all remuneration goes directly to the University of Liverpool.
MALMO, SWEDEN – The combination of serum procalcitonin and C-reactive protein levels upon admission to a pediatric ICU displayed high utility for early diagnosis of serious bacterial infection in critically ill children in a large prospective observational study presented at the annual meeting of the European Society for Paediatric Infectious Diseases.
This winning combination significantly outperformed neutrophil gelatinase-associated lipocalin, activated partial thromboplastin time, and resistin, both individually and in various combinations, for the vital task of making a rapid distinction between infectious and noninfectious causes of pediatric systemic inflammatory response syndrome, reported Enitan D. Carrol, MD, professor of pediatric infection at the University of Liverpool (England).
“One of the clinical dilemmas we face in intensive care is being able to differentiate between infectious and noninfectious causes of systemic inflammatory response syndrome. This is important because we need to identify which children have life-threatening infections so that we can promptly initiate antimicrobial therapy,” she explained.
One in four deaths in pediatric ICUs are infection related, Dr. Carrol noted.
“There is an urgent need for infection markers which, firstly, change early in the course of bacterial infection, secondly, correlate with real-time clinical progression, and thirdly, have a rapid turn-around time to allow effective clinical decision making,” she observed.
The combination of procalcitonin and C-reactive protein (CRP) levels measured at admission fits the bill, Dr. Carrol continued. Of the five biomarkers evaluated in her study – all backed by some supporting evidence of efficacy in earlier studies – the top two individual performers in terms of negative predictive value (NPV) were a CRP less than 4.2 mg/dL with a negative NPV of 99%, and a procalcitonin less than 1.52 ng/mL with an NPV of 96%. The positive predictive value of each of the biomarkers was 37%. The sensitivity and specificity of procalcitonin for diagnosis of serious bacterial infection were 78% and 80%, respectively. For CRP, the figures were 93% and 76%.
The combination of procalcitonin and CRP outperformed a multitude of other two-, three-, and four-biomarker combinations tested, with an area under the curve of 93% for combined sensitivity and specificity.
The study included 657 children admitted to the pediatric ICU at Alder Hey Children’s Hospital in Liverpool with systemic inflammatory response syndrome. All had blood samples measured for the five biomarkers on days 1-7. Clinicians were blinded as to the biomarker results. Ninety-two (14%) patients were ultimately found to have a serious bacterial infection – essentially, bacterial meningitis or septic shock – and 565 (86%) had a nonbacterial etiology.
The 28-day mortality rate was 9% in the group with serious bacterial infection, significantly higher than the 2% rate in the group with other causes of their systemic inflammatory response syndrome.
Longitudinal trends in procalcitonin and CRP as evidenced in the study can be used in clinical decision making, according to Dr. Carrol. Mean values of procalcitonin plummeted by 80% from day 1 to day 5 in response to antimicrobial therapy in the group with serious bacterial infections. In contrast, CRP levels rose sharply from day 1 to a peak on day 2, then fell, although the 50% drop from day 2 to day 5 in response to antimicrobial therapy wasn’t as pronounced as the change in procalcitonin.
“There is an additive benefit for both biomarkers compared with CRP alone. The problem with CRP on admission, as I’ve demonstrated in this study, is it often hasn’t risen yet early after admission. So although it gave the best area under the curve of any of the biomarkers, I think that combined with procalcitonin you get a much better descriminator,” Dr. Carrol said.
The median duration of ICU stay in the patients with serious bacterial infection at admission was 5 days, compared with 3 days when the cause of systemic inflammatory response syndrome lay elsewhere. Their median duration of ventilation was significantly longer, too: 4 days versus 2 in children without a serious bacterial infection.
Stepwise logistic regression analysis pinpointed several clinical variables as being associated with prolonged ICU stay.

In addition, initiation of antibiotic therapy prior to admission to the pediatric ICU was associated with a 50% reduction in the likelihood of a prolonged ICU stay. “This reflects the fact that early antibiotics give you a better prognosis if you have sepsis,” according to Dr. Carrol.
She and her coinvestigators now have embarked on a multicenter U.K. study looking at the impact of procalcitonin to guide duration of antimicrobial therapy in critically ill children.
The Alder Hey study was funded by the U.K. National Institute for Health Research. Dr. Carrol reported having no financial conflicts. Although she serves as a consultant to several health care companies, all remuneration goes directly to the University of Liverpool.
MALMO, SWEDEN – The combination of serum procalcitonin and C-reactive protein levels upon admission to a pediatric ICU displayed high utility for early diagnosis of serious bacterial infection in critically ill children in a large prospective observational study presented at the annual meeting of the European Society for Paediatric Infectious Diseases.
This winning combination significantly outperformed neutrophil gelatinase-associated lipocalin, activated partial thromboplastin time, and resistin, both individually and in various combinations, for the vital task of making a rapid distinction between infectious and noninfectious causes of pediatric systemic inflammatory response syndrome, reported Enitan D. Carrol, MD, professor of pediatric infection at the University of Liverpool (England).
“One of the clinical dilemmas we face in intensive care is being able to differentiate between infectious and noninfectious causes of systemic inflammatory response syndrome. This is important because we need to identify which children have life-threatening infections so that we can promptly initiate antimicrobial therapy,” she explained.
One in four deaths in pediatric ICUs are infection related, Dr. Carrol noted.
“There is an urgent need for infection markers which, firstly, change early in the course of bacterial infection, secondly, correlate with real-time clinical progression, and thirdly, have a rapid turn-around time to allow effective clinical decision making,” she observed.
The combination of procalcitonin and C-reactive protein (CRP) levels measured at admission fits the bill, Dr. Carrol continued. Of the five biomarkers evaluated in her study – all backed by some supporting evidence of efficacy in earlier studies – the top two individual performers in terms of negative predictive value (NPV) were a CRP less than 4.2 mg/dL with a negative NPV of 99%, and a procalcitonin less than 1.52 ng/mL with an NPV of 96%. The positive predictive value of each of the biomarkers was 37%. The sensitivity and specificity of procalcitonin for diagnosis of serious bacterial infection were 78% and 80%, respectively. For CRP, the figures were 93% and 76%.
The combination of procalcitonin and CRP outperformed a multitude of other two-, three-, and four-biomarker combinations tested, with an area under the curve of 93% for combined sensitivity and specificity.
The study included 657 children admitted to the pediatric ICU at Alder Hey Children’s Hospital in Liverpool with systemic inflammatory response syndrome. All had blood samples measured for the five biomarkers on days 1-7. Clinicians were blinded as to the biomarker results. Ninety-two (14%) patients were ultimately found to have a serious bacterial infection – essentially, bacterial meningitis or septic shock – and 565 (86%) had a nonbacterial etiology.
The 28-day mortality rate was 9% in the group with serious bacterial infection, significantly higher than the 2% rate in the group with other causes of their systemic inflammatory response syndrome.
Longitudinal trends in procalcitonin and CRP as evidenced in the study can be used in clinical decision making, according to Dr. Carrol. Mean values of procalcitonin plummeted by 80% from day 1 to day 5 in response to antimicrobial therapy in the group with serious bacterial infections. In contrast, CRP levels rose sharply from day 1 to a peak on day 2, then fell, although the 50% drop from day 2 to day 5 in response to antimicrobial therapy wasn’t as pronounced as the change in procalcitonin.
“There is an additive benefit for both biomarkers compared with CRP alone. The problem with CRP on admission, as I’ve demonstrated in this study, is it often hasn’t risen yet early after admission. So although it gave the best area under the curve of any of the biomarkers, I think that combined with procalcitonin you get a much better descriminator,” Dr. Carrol said.
The median duration of ICU stay in the patients with serious bacterial infection at admission was 5 days, compared with 3 days when the cause of systemic inflammatory response syndrome lay elsewhere. Their median duration of ventilation was significantly longer, too: 4 days versus 2 in children without a serious bacterial infection.
Stepwise logistic regression analysis pinpointed several clinical variables as being associated with prolonged ICU stay.

In addition, initiation of antibiotic therapy prior to admission to the pediatric ICU was associated with a 50% reduction in the likelihood of a prolonged ICU stay. “This reflects the fact that early antibiotics give you a better prognosis if you have sepsis,” according to Dr. Carrol.
She and her coinvestigators now have embarked on a multicenter U.K. study looking at the impact of procalcitonin to guide duration of antimicrobial therapy in critically ill children.
The Alder Hey study was funded by the U.K. National Institute for Health Research. Dr. Carrol reported having no financial conflicts. Although she serves as a consultant to several health care companies, all remuneration goes directly to the University of Liverpool.
REPORTING FROM ESPID 2018
Key clinical point:
Major finding: The area under the curve combining sensitivity and specificity was 93%.
Study details: This was a prospective, observational, single-center, clinician-blinded study of 657 patients admitted to a pediatric ICU with symptoms of systemic inflammatory response syndrome.
Disclosures: The study was funded by the U.K. National Institute for Health Research. The presenter reported having no relevant financial conflicts. Although she serves as a consultant to several health care companies, all remuneration goes directly to the University of Liverpool.
MI risk prediction after noncardiac surgery simplified
ORLANDO – The risk of perioperative MI or death associated with noncardiac surgery is vanishingly low in patients free of diabetes, hypertension, and smoking, Tanya Wilcox, MD, reported at the annual meeting of the American College of Cardiology.
How small is the risk? A mere 1 in 1,000, according to her analysis of more than 3.8 million major noncardiac surgeries in the American College of Surgeons National Surgical Quality Improvement Program database for 2009-2015, according to Dr. Wilcox of New York University.
Physicians are frequently asked by surgeons to clear patients for noncardiac surgery in terms of cardiovascular risk. Because current risk scores are complex, aren’t amenable to rapid bedside calculations, and may entail cardiac stress testing, Dr. Wilcox decided it was worth assessing the impact of three straightforward cardiovascular risk factors – current smoking and treatment for hypertension or diabetes – on 30-day postoperative MI-free survival. For this purpose she turned to the National Surgical Quality Improvement Program database, a validated, risk-adjusted, outcomes-based program to measure and improve the quality of surgical care utilizing data from 250 U.S. surgical centers.
Of the 3,817,113 patients who underwent major noncardiac surgery, 1,586,020 (42%) of them had none of the three cardiovascular risk factors of interest, 1,541,846 (40%) had one, 643,424 (17%) had two, and 45,823, or 1.2%, had all three. The patients’ mean age was 57, 75% were white, and 57% were women. About half of all patients underwent various operations within the realm of general surgery; next most frequent were orthopedic procedures, accounting for 18% of total noncardiac surgery. Of note, only 23% of patients with zero risk factors were American Society of Anesthesiologists Class 3-5, compared with 51% of those with one cardiovascular risk factor, 76% with two, and 71% with all three.
The incidence of acute MI or death within 30 days of noncardiac surgery climbed in stepwise fashion according to a patient’s risk factor burden. In a multivariate analysis adjusted for age, race, and gender, patients with any one of the cardiovascular risk factors had a 30-day risk of acute MI or death that was 1.52 times greater than those with no risk factors, patients with two risk factors were at 2.4-fold increased risk, and those with all three were at 3.63-fold greater risk than those with none. The degree of increased risk associated with any single risk factor ranged from 1.47-fold for hypertension to 1.94-fold for smoking.
“Further study is needed to determine whether aggressive risk factor modifications in the form of blood pressure control, glycemic control, and smoking cessation could reduce the incidence of postoperative MI,” Dr. Wilcox observed.
She reported having no financial conflicts regarding her study.
ORLANDO – The risk of perioperative MI or death associated with noncardiac surgery is vanishingly low in patients free of diabetes, hypertension, and smoking, Tanya Wilcox, MD, reported at the annual meeting of the American College of Cardiology.
How small is the risk? A mere 1 in 1,000, according to her analysis of more than 3.8 million major noncardiac surgeries in the American College of Surgeons National Surgical Quality Improvement Program database for 2009-2015, according to Dr. Wilcox of New York University.
Physicians are frequently asked by surgeons to clear patients for noncardiac surgery in terms of cardiovascular risk. Because current risk scores are complex, aren’t amenable to rapid bedside calculations, and may entail cardiac stress testing, Dr. Wilcox decided it was worth assessing the impact of three straightforward cardiovascular risk factors – current smoking and treatment for hypertension or diabetes – on 30-day postoperative MI-free survival. For this purpose she turned to the National Surgical Quality Improvement Program database, a validated, risk-adjusted, outcomes-based program to measure and improve the quality of surgical care utilizing data from 250 U.S. surgical centers.
Of the 3,817,113 patients who underwent major noncardiac surgery, 1,586,020 (42%) of them had none of the three cardiovascular risk factors of interest, 1,541,846 (40%) had one, 643,424 (17%) had two, and 45,823, or 1.2%, had all three. The patients’ mean age was 57, 75% were white, and 57% were women. About half of all patients underwent various operations within the realm of general surgery; next most frequent were orthopedic procedures, accounting for 18% of total noncardiac surgery. Of note, only 23% of patients with zero risk factors were American Society of Anesthesiologists Class 3-5, compared with 51% of those with one cardiovascular risk factor, 76% with two, and 71% with all three.
The incidence of acute MI or death within 30 days of noncardiac surgery climbed in stepwise fashion according to a patient’s risk factor burden. In a multivariate analysis adjusted for age, race, and gender, patients with any one of the cardiovascular risk factors had a 30-day risk of acute MI or death that was 1.52 times greater than those with no risk factors, patients with two risk factors were at 2.4-fold increased risk, and those with all three were at 3.63-fold greater risk than those with none. The degree of increased risk associated with any single risk factor ranged from 1.47-fold for hypertension to 1.94-fold for smoking.
“Further study is needed to determine whether aggressive risk factor modifications in the form of blood pressure control, glycemic control, and smoking cessation could reduce the incidence of postoperative MI,” Dr. Wilcox observed.
She reported having no financial conflicts regarding her study.
ORLANDO – The risk of perioperative MI or death associated with noncardiac surgery is vanishingly low in patients free of diabetes, hypertension, and smoking, Tanya Wilcox, MD, reported at the annual meeting of the American College of Cardiology.
How small is the risk? A mere 1 in 1,000, according to her analysis of more than 3.8 million major noncardiac surgeries in the American College of Surgeons National Surgical Quality Improvement Program database for 2009-2015, according to Dr. Wilcox of New York University.
Physicians are frequently asked by surgeons to clear patients for noncardiac surgery in terms of cardiovascular risk. Because current risk scores are complex, aren’t amenable to rapid bedside calculations, and may entail cardiac stress testing, Dr. Wilcox decided it was worth assessing the impact of three straightforward cardiovascular risk factors – current smoking and treatment for hypertension or diabetes – on 30-day postoperative MI-free survival. For this purpose she turned to the National Surgical Quality Improvement Program database, a validated, risk-adjusted, outcomes-based program to measure and improve the quality of surgical care utilizing data from 250 U.S. surgical centers.
Of the 3,817,113 patients who underwent major noncardiac surgery, 1,586,020 (42%) of them had none of the three cardiovascular risk factors of interest, 1,541,846 (40%) had one, 643,424 (17%) had two, and 45,823, or 1.2%, had all three. The patients’ mean age was 57, 75% were white, and 57% were women. About half of all patients underwent various operations within the realm of general surgery; next most frequent were orthopedic procedures, accounting for 18% of total noncardiac surgery. Of note, only 23% of patients with zero risk factors were American Society of Anesthesiologists Class 3-5, compared with 51% of those with one cardiovascular risk factor, 76% with two, and 71% with all three.
The incidence of acute MI or death within 30 days of noncardiac surgery climbed in stepwise fashion according to a patient’s risk factor burden. In a multivariate analysis adjusted for age, race, and gender, patients with any one of the cardiovascular risk factors had a 30-day risk of acute MI or death that was 1.52 times greater than those with no risk factors, patients with two risk factors were at 2.4-fold increased risk, and those with all three were at 3.63-fold greater risk than those with none. The degree of increased risk associated with any single risk factor ranged from 1.47-fold for hypertension to 1.94-fold for smoking.
“Further study is needed to determine whether aggressive risk factor modifications in the form of blood pressure control, glycemic control, and smoking cessation could reduce the incidence of postoperative MI,” Dr. Wilcox observed.
She reported having no financial conflicts regarding her study.
REPORTING FROM ACC 2018
Key clinical point: Noncardiac surgery patients can breathe easier regarding perioperative cardiovascular risk provided they don’t smoke and aren’t hypertensive or diabetic.
Major finding: .
Study details: This was a retrospective analysis of more than 3.8 million noncardiac surgeries contained in the American College of Surgeons National Surgical Quality Improvement Program database for 2009-2015.
Disclosures: The study presenter reported having no financial conflicts.
Creating a digital pill
Technology battles medication noncompliance
Hospitalists and other physicians have long struggled with medication noncompliance, which can lead to sicker patients and higher rates of readmittance, and costs some $100-$289 billion a year.
There is a growing field of digital devices being developed to address this problem. The Food and Drug Administration has just approved the newest one: a medication with a sensor embedded that can tell doctors if, and when, patients take their medicine, according to an article in the New York Times.1 It’s expected to become available in 2018.
The digital medication is a version of the antipsychotic Abilify. Patients who agree to take it will sign consent forms allowing their doctors (and up to four other people) to receive electronic data showing the date and time pills are ingested.
The sensor, created by Proteus Digital Health, contains copper, magnesium, and silicon, all said to be safe ingredients found in foods. The electrical signal is created when stomach fluids contact the sensor; a patch worn on the rib cage detects that signal and sends the message.
Other companies are joining the race to create digital medication technologies; these are being tested in medications for patients with conditions including heart disease, diabetes, and HIV infection. Some researchers predict the technology might have applications for monitoring the opioid intake of postsurgical patients or patients in medication clinical trials.
Reference
1. Belluck P. “First Digital Pill Approved to Worries About Biomedical ‘Big Brother.’ ” New York Times. Nov 13, 2017.
Technology battles medication noncompliance
Technology battles medication noncompliance
Hospitalists and other physicians have long struggled with medication noncompliance, which can lead to sicker patients and higher rates of readmittance, and costs some $100-$289 billion a year.
There is a growing field of digital devices being developed to address this problem. The Food and Drug Administration has just approved the newest one: a medication with a sensor embedded that can tell doctors if, and when, patients take their medicine, according to an article in the New York Times.1 It’s expected to become available in 2018.
The digital medication is a version of the antipsychotic Abilify. Patients who agree to take it will sign consent forms allowing their doctors (and up to four other people) to receive electronic data showing the date and time pills are ingested.
The sensor, created by Proteus Digital Health, contains copper, magnesium, and silicon, all said to be safe ingredients found in foods. The electrical signal is created when stomach fluids contact the sensor; a patch worn on the rib cage detects that signal and sends the message.
Other companies are joining the race to create digital medication technologies; these are being tested in medications for patients with conditions including heart disease, diabetes, and HIV infection. Some researchers predict the technology might have applications for monitoring the opioid intake of postsurgical patients or patients in medication clinical trials.
Reference
1. Belluck P. “First Digital Pill Approved to Worries About Biomedical ‘Big Brother.’ ” New York Times. Nov 13, 2017.
Hospitalists and other physicians have long struggled with medication noncompliance, which can lead to sicker patients and higher rates of readmittance, and costs some $100-$289 billion a year.
There is a growing field of digital devices being developed to address this problem. The Food and Drug Administration has just approved the newest one: a medication with a sensor embedded that can tell doctors if, and when, patients take their medicine, according to an article in the New York Times.1 It’s expected to become available in 2018.
The digital medication is a version of the antipsychotic Abilify. Patients who agree to take it will sign consent forms allowing their doctors (and up to four other people) to receive electronic data showing the date and time pills are ingested.
The sensor, created by Proteus Digital Health, contains copper, magnesium, and silicon, all said to be safe ingredients found in foods. The electrical signal is created when stomach fluids contact the sensor; a patch worn on the rib cage detects that signal and sends the message.
Other companies are joining the race to create digital medication technologies; these are being tested in medications for patients with conditions including heart disease, diabetes, and HIV infection. Some researchers predict the technology might have applications for monitoring the opioid intake of postsurgical patients or patients in medication clinical trials.
Reference
1. Belluck P. “First Digital Pill Approved to Worries About Biomedical ‘Big Brother.’ ” New York Times. Nov 13, 2017.
Galectin-3: A new post-MI prognostic biomarker?
ORLANDO – An elevated circulating galactin-3 level after an acute MI is a potent long-term predictor of both heart failure and mortality, independent of known prognostic markers, Rabea Asleh, MD, PhD, reported at the annual meeting of the American College of Cardiology.
“These findings suggest that galectin-3 measurement may have a role in the risk stratification of patients presenting with MI,” according to Dr. Asleh, an Israeli cardiologist doing a fellowship in advanced heart failure and transplant cardiology at the Mayo Clinic in Rochester, Minn.
“The changing clinical presentation of MI necessitates evolution in our approach to risk stratification,” he explained. “Over the last 2 decades we’ve observed a change in the epidemiology of MI, with more patients developing non-ST-elevation MI compared to STEMI. They present at an older age and develop heart failure with preserved ejection fraction more than heart failure with reduced ejection fraction.”
He presented a prospective population-based community cohort study of 1,401 Olmsted County, Minn., residents who had a validated MI during 2002-2012. Their mean age was 67 years, 61% were men, and 79% presented with non-STEMI. During a mean follow-up of 5.3 years, 389 of the participants developed heart failure and 512 patients died.
Galectin-3 was measured a median of 2 days post MI. The median level was 18.4 ng/mL. Patients were divided into tertiles based upon their galactin-3 measurement: Tertile 1 required a post-MI galectin-3 level below 15.2 ng/mL; tertile 2 had a level of 15.2-22.6 ng/mL; and the top tertile was for individuals with a galectin-3 above 22.6 ng/mL.
Of note, patients with a higher galectin-3 level were older, had a higher prevalence of diabetes, hypertension, hyperlipidemia, anterior MI, a higher Killip class, a higher Charlson comorbidity score, and a lower peak troponin T level. They also had a lower estimated glomerular filtration rate; indeed, the median eGFR in the top tertile for galactin-3 was 48 mL/min per 1.73 m2, compared with 68 mL/min in the lowest galectin-3 tertile. Women accounted for 27% of patients in tertile 1, 41% in tertile 2, and fully half of those in tertile 3.
In an unadjusted analysis, the risk of mortality during follow-up was sixfold greater for patients in galectin-3 tertile 3 than in tertile 1; the risk of heart failure was increased 5.5-fold.
More meaningfully, in a Cox multivariate analysis extensively adjusted for age, gender, comorbidities, malignancy, standard cardiovascular risk factors, MI characteristics, eGFR, Killip class, cardiac troponin T, and other potential confounders, patients in galectin-3 tertile 2 had a 1.6-fold increased risk of death and a 1.62-fold increased likelihood of heart failure during follow-up, compared with subjects in tertile 1, Dr. Asleh noted.
Patients in tertile 3 had a 2.4-fold increased risk of death and were at 2.1 times greater risk of heart failure than those in tertile 1. The degree of risk for heart failure associated with elevated galactin-3 was virtually identical for heart failure with preserved as compared with reduced ejection fraction, he added.
Session cochair L. Kristin Newby, MD, of Duke University, Durham, N.C., noted that the Mayo study did not adjust for brain natriuretic peptide (BNP) or N-terminal pro hormone BNP (NT-proBNP), both of which are known to be strong predictors of both heart failure and mortality after acute MI. Doesn’t their absence weaken the strength of galactin-3’s prognostic power as demonstrated in the study? she asked.
Dr. Asleh replied that those biomarkers weren’t collected in this study, which began in 2002.
“What I can tell you is, other studies show there is only a weak correlation between galactin-3 and NT-proBNP post MI. Some studies have even shown an inverse correlation,” he said. “The pathophysiological explanation is that galactin-3 is more implicated in fibrosis before the stage of development of left ventricular loading and stretching of the myocardium. So galactin-3 may be implicated in LV fibrosis leading to heart failure before the NT-proBNP comes into play.”
Also, he cited a study by other investigators conducted in patients with a left ventricular assist device for advanced heart failure. Upon device-induced left ventricular unloading the patients’ NT-proBNP levels dropped significantly while their galactin-3 remained high and unchanged. This suggests the two biomarkers are implicated in different disease pathways.
Both animal and human studies indicate galactin-3 is involved specifically in fibrosis, as opposed to, say, C-reactive protein, a well established marker of systemic inflammation, the cardiologist added.
Dr. Asleh reported having no financial conflicts of interest regarding his study, which was supported by the National Institutes of Health.
ORLANDO – An elevated circulating galactin-3 level after an acute MI is a potent long-term predictor of both heart failure and mortality, independent of known prognostic markers, Rabea Asleh, MD, PhD, reported at the annual meeting of the American College of Cardiology.
“These findings suggest that galectin-3 measurement may have a role in the risk stratification of patients presenting with MI,” according to Dr. Asleh, an Israeli cardiologist doing a fellowship in advanced heart failure and transplant cardiology at the Mayo Clinic in Rochester, Minn.
“The changing clinical presentation of MI necessitates evolution in our approach to risk stratification,” he explained. “Over the last 2 decades we’ve observed a change in the epidemiology of MI, with more patients developing non-ST-elevation MI compared to STEMI. They present at an older age and develop heart failure with preserved ejection fraction more than heart failure with reduced ejection fraction.”
He presented a prospective population-based community cohort study of 1,401 Olmsted County, Minn., residents who had a validated MI during 2002-2012. Their mean age was 67 years, 61% were men, and 79% presented with non-STEMI. During a mean follow-up of 5.3 years, 389 of the participants developed heart failure and 512 patients died.
Galectin-3 was measured a median of 2 days post MI. The median level was 18.4 ng/mL. Patients were divided into tertiles based upon their galactin-3 measurement: Tertile 1 required a post-MI galectin-3 level below 15.2 ng/mL; tertile 2 had a level of 15.2-22.6 ng/mL; and the top tertile was for individuals with a galectin-3 above 22.6 ng/mL.
Of note, patients with a higher galectin-3 level were older, had a higher prevalence of diabetes, hypertension, hyperlipidemia, anterior MI, a higher Killip class, a higher Charlson comorbidity score, and a lower peak troponin T level. They also had a lower estimated glomerular filtration rate; indeed, the median eGFR in the top tertile for galactin-3 was 48 mL/min per 1.73 m2, compared with 68 mL/min in the lowest galectin-3 tertile. Women accounted for 27% of patients in tertile 1, 41% in tertile 2, and fully half of those in tertile 3.
In an unadjusted analysis, the risk of mortality during follow-up was sixfold greater for patients in galectin-3 tertile 3 than in tertile 1; the risk of heart failure was increased 5.5-fold.
More meaningfully, in a Cox multivariate analysis extensively adjusted for age, gender, comorbidities, malignancy, standard cardiovascular risk factors, MI characteristics, eGFR, Killip class, cardiac troponin T, and other potential confounders, patients in galectin-3 tertile 2 had a 1.6-fold increased risk of death and a 1.62-fold increased likelihood of heart failure during follow-up, compared with subjects in tertile 1, Dr. Asleh noted.
Patients in tertile 3 had a 2.4-fold increased risk of death and were at 2.1 times greater risk of heart failure than those in tertile 1. The degree of risk for heart failure associated with elevated galactin-3 was virtually identical for heart failure with preserved as compared with reduced ejection fraction, he added.
Session cochair L. Kristin Newby, MD, of Duke University, Durham, N.C., noted that the Mayo study did not adjust for brain natriuretic peptide (BNP) or N-terminal pro hormone BNP (NT-proBNP), both of which are known to be strong predictors of both heart failure and mortality after acute MI. Doesn’t their absence weaken the strength of galactin-3’s prognostic power as demonstrated in the study? she asked.
Dr. Asleh replied that those biomarkers weren’t collected in this study, which began in 2002.
“What I can tell you is, other studies show there is only a weak correlation between galactin-3 and NT-proBNP post MI. Some studies have even shown an inverse correlation,” he said. “The pathophysiological explanation is that galactin-3 is more implicated in fibrosis before the stage of development of left ventricular loading and stretching of the myocardium. So galactin-3 may be implicated in LV fibrosis leading to heart failure before the NT-proBNP comes into play.”
Also, he cited a study by other investigators conducted in patients with a left ventricular assist device for advanced heart failure. Upon device-induced left ventricular unloading the patients’ NT-proBNP levels dropped significantly while their galactin-3 remained high and unchanged. This suggests the two biomarkers are implicated in different disease pathways.
Both animal and human studies indicate galactin-3 is involved specifically in fibrosis, as opposed to, say, C-reactive protein, a well established marker of systemic inflammation, the cardiologist added.
Dr. Asleh reported having no financial conflicts of interest regarding his study, which was supported by the National Institutes of Health.
ORLANDO – An elevated circulating galactin-3 level after an acute MI is a potent long-term predictor of both heart failure and mortality, independent of known prognostic markers, Rabea Asleh, MD, PhD, reported at the annual meeting of the American College of Cardiology.
“These findings suggest that galectin-3 measurement may have a role in the risk stratification of patients presenting with MI,” according to Dr. Asleh, an Israeli cardiologist doing a fellowship in advanced heart failure and transplant cardiology at the Mayo Clinic in Rochester, Minn.
“The changing clinical presentation of MI necessitates evolution in our approach to risk stratification,” he explained. “Over the last 2 decades we’ve observed a change in the epidemiology of MI, with more patients developing non-ST-elevation MI compared to STEMI. They present at an older age and develop heart failure with preserved ejection fraction more than heart failure with reduced ejection fraction.”
He presented a prospective population-based community cohort study of 1,401 Olmsted County, Minn., residents who had a validated MI during 2002-2012. Their mean age was 67 years, 61% were men, and 79% presented with non-STEMI. During a mean follow-up of 5.3 years, 389 of the participants developed heart failure and 512 patients died.
Galectin-3 was measured a median of 2 days post MI. The median level was 18.4 ng/mL. Patients were divided into tertiles based upon their galactin-3 measurement: Tertile 1 required a post-MI galectin-3 level below 15.2 ng/mL; tertile 2 had a level of 15.2-22.6 ng/mL; and the top tertile was for individuals with a galectin-3 above 22.6 ng/mL.
Of note, patients with a higher galectin-3 level were older, had a higher prevalence of diabetes, hypertension, hyperlipidemia, anterior MI, a higher Killip class, a higher Charlson comorbidity score, and a lower peak troponin T level. They also had a lower estimated glomerular filtration rate; indeed, the median eGFR in the top tertile for galactin-3 was 48 mL/min per 1.73 m2, compared with 68 mL/min in the lowest galectin-3 tertile. Women accounted for 27% of patients in tertile 1, 41% in tertile 2, and fully half of those in tertile 3.
In an unadjusted analysis, the risk of mortality during follow-up was sixfold greater for patients in galectin-3 tertile 3 than in tertile 1; the risk of heart failure was increased 5.5-fold.
More meaningfully, in a Cox multivariate analysis extensively adjusted for age, gender, comorbidities, malignancy, standard cardiovascular risk factors, MI characteristics, eGFR, Killip class, cardiac troponin T, and other potential confounders, patients in galectin-3 tertile 2 had a 1.6-fold increased risk of death and a 1.62-fold increased likelihood of heart failure during follow-up, compared with subjects in tertile 1, Dr. Asleh noted.
Patients in tertile 3 had a 2.4-fold increased risk of death and were at 2.1 times greater risk of heart failure than those in tertile 1. The degree of risk for heart failure associated with elevated galactin-3 was virtually identical for heart failure with preserved as compared with reduced ejection fraction, he added.
Session cochair L. Kristin Newby, MD, of Duke University, Durham, N.C., noted that the Mayo study did not adjust for brain natriuretic peptide (BNP) or N-terminal pro hormone BNP (NT-proBNP), both of which are known to be strong predictors of both heart failure and mortality after acute MI. Doesn’t their absence weaken the strength of galactin-3’s prognostic power as demonstrated in the study? she asked.
Dr. Asleh replied that those biomarkers weren’t collected in this study, which began in 2002.
“What I can tell you is, other studies show there is only a weak correlation between galactin-3 and NT-proBNP post MI. Some studies have even shown an inverse correlation,” he said. “The pathophysiological explanation is that galactin-3 is more implicated in fibrosis before the stage of development of left ventricular loading and stretching of the myocardium. So galactin-3 may be implicated in LV fibrosis leading to heart failure before the NT-proBNP comes into play.”
Also, he cited a study by other investigators conducted in patients with a left ventricular assist device for advanced heart failure. Upon device-induced left ventricular unloading the patients’ NT-proBNP levels dropped significantly while their galactin-3 remained high and unchanged. This suggests the two biomarkers are implicated in different disease pathways.
Both animal and human studies indicate galactin-3 is involved specifically in fibrosis, as opposed to, say, C-reactive protein, a well established marker of systemic inflammation, the cardiologist added.
Dr. Asleh reported having no financial conflicts of interest regarding his study, which was supported by the National Institutes of Health.
REPORTING FROM ACC 18
Key clinical point: Galectin-3 level post-MI is a potent long-term predictor of both heart failure and mortality independent of known prognostic markers.
Major finding: Post-MI patients in the top tertile of circulating galectin-3 were at an adjusted 2.4-fold increased mortality risk and a 2.05-fold greater risk of developing heart failure compared with those in the lowest tertile.
Study details: This prospective population-based cohort study included 1,401 MI patients followed for a mean of 5.3 years.
Disclosures: The National Institutes of Health supported the study. The presenter reported having no financial conflicts of interest.
A call for ‘changing the social norms’ on naloxone
SAN DIEGO – Among individuals with a history of injection drug use, more than one-third reported never or sometimes carrying naloxone, while just one in four reported carrying with it them at all times.
Those are key findings from a survey that set out to examine gaps in the naloxone cascade in a sample of people who inject drugs.
“In order to save a life, you have to have the naloxone with you at all times,” lead study author Karin E. Tobin, PhD, said in an interview at the annual meeting of the College on Problems of Drug Dependence.
While emerging research demonstrates the positive impact of opioid overdose education and community naloxone distribution programs to reduce opioid-related overdose deaths, opiate overdose continues to be a major cause of mortality, said Dr. Tobin, who is affiliated with the department of health behavior and society at Johns Hopkins University, Baltimore. “We’ve made a lot of progress in convincing people that naloxone is not addictive, and that it’s not going to cause any harm,” she said. “Now, drug users aren’t afraid to ask for it. Still, we wondered: If everyone knows about naloxone and no one is embarrassed to talk about it, why are people still dying [from opioid overdoses] in Baltimore?”
She and her associates conducted a cross-sectional survey of 353 individuals aged 18 and older in Baltimore who self-reported a lifetime history of injection drug use. The data came from a baseline survey that was conducted as part of a randomized, controlled trial testing the efficacy of a behavioral intervention focused on the Hepatitis C cascade. Individuals were asked to answer questions related to the five steps of the naloxone cascade: awareness (have you ever heard about naloxone?), access (have you ever received naloxone?), training (have you ever been trained to use naloxone?), use (have you ever used naloxone during an opiate overdose?), and possession (how often do you carry naloxone?)
More than half of the survey respondents (65%) were male; mean age was 47 years. For the previous 6 months, more than half of the sample reported the use of crack (64%), heroin (74%), and other injectable drugs (57%), while 90% reported having ever witnessed an overdose – 59% in the prior year alone. Dr. Tobin and her associates found that 90% of respondents had heard about naloxone, 69% had received it, and 60% had been trained to use it. In addition, 37% reported never carrying naloxone, 38% sometimes carried it, 33% said they had used naloxone at some point, and 25% said they always carried it with them.
On multinomial regression analysis, the researchers found that carrying naloxone often or always was significantly associated with the following variables: female sex (odds ratio, 2.77), having ever witnessed an overdose (OR, 1.84), having injected in the past 12 months (OR, 1.75), and having ever used naloxone during an overdose (OR, 4.33). The latter finding is especially important, “because it means that we just have to let people practice using it,” said Dr. Tobin, who noted that more research is needed to understand reasons why injection drug users do not always carry naloxone. “We need to start changing the social norms about carrying naloxone. You never know when it will be useful.”
The National Institute on Drug Abuse supported the study. Dr. Tobin reported having no financial disclosures.
SAN DIEGO – Among individuals with a history of injection drug use, more than one-third reported never or sometimes carrying naloxone, while just one in four reported carrying with it them at all times.
Those are key findings from a survey that set out to examine gaps in the naloxone cascade in a sample of people who inject drugs.
“In order to save a life, you have to have the naloxone with you at all times,” lead study author Karin E. Tobin, PhD, said in an interview at the annual meeting of the College on Problems of Drug Dependence.
While emerging research demonstrates the positive impact of opioid overdose education and community naloxone distribution programs to reduce opioid-related overdose deaths, opiate overdose continues to be a major cause of mortality, said Dr. Tobin, who is affiliated with the department of health behavior and society at Johns Hopkins University, Baltimore. “We’ve made a lot of progress in convincing people that naloxone is not addictive, and that it’s not going to cause any harm,” she said. “Now, drug users aren’t afraid to ask for it. Still, we wondered: If everyone knows about naloxone and no one is embarrassed to talk about it, why are people still dying [from opioid overdoses] in Baltimore?”
She and her associates conducted a cross-sectional survey of 353 individuals aged 18 and older in Baltimore who self-reported a lifetime history of injection drug use. The data came from a baseline survey that was conducted as part of a randomized, controlled trial testing the efficacy of a behavioral intervention focused on the Hepatitis C cascade. Individuals were asked to answer questions related to the five steps of the naloxone cascade: awareness (have you ever heard about naloxone?), access (have you ever received naloxone?), training (have you ever been trained to use naloxone?), use (have you ever used naloxone during an opiate overdose?), and possession (how often do you carry naloxone?)
More than half of the survey respondents (65%) were male; mean age was 47 years. For the previous 6 months, more than half of the sample reported the use of crack (64%), heroin (74%), and other injectable drugs (57%), while 90% reported having ever witnessed an overdose – 59% in the prior year alone. Dr. Tobin and her associates found that 90% of respondents had heard about naloxone, 69% had received it, and 60% had been trained to use it. In addition, 37% reported never carrying naloxone, 38% sometimes carried it, 33% said they had used naloxone at some point, and 25% said they always carried it with them.
On multinomial regression analysis, the researchers found that carrying naloxone often or always was significantly associated with the following variables: female sex (odds ratio, 2.77), having ever witnessed an overdose (OR, 1.84), having injected in the past 12 months (OR, 1.75), and having ever used naloxone during an overdose (OR, 4.33). The latter finding is especially important, “because it means that we just have to let people practice using it,” said Dr. Tobin, who noted that more research is needed to understand reasons why injection drug users do not always carry naloxone. “We need to start changing the social norms about carrying naloxone. You never know when it will be useful.”
The National Institute on Drug Abuse supported the study. Dr. Tobin reported having no financial disclosures.
SAN DIEGO – Among individuals with a history of injection drug use, more than one-third reported never or sometimes carrying naloxone, while just one in four reported carrying with it them at all times.
Those are key findings from a survey that set out to examine gaps in the naloxone cascade in a sample of people who inject drugs.
“In order to save a life, you have to have the naloxone with you at all times,” lead study author Karin E. Tobin, PhD, said in an interview at the annual meeting of the College on Problems of Drug Dependence.
While emerging research demonstrates the positive impact of opioid overdose education and community naloxone distribution programs to reduce opioid-related overdose deaths, opiate overdose continues to be a major cause of mortality, said Dr. Tobin, who is affiliated with the department of health behavior and society at Johns Hopkins University, Baltimore. “We’ve made a lot of progress in convincing people that naloxone is not addictive, and that it’s not going to cause any harm,” she said. “Now, drug users aren’t afraid to ask for it. Still, we wondered: If everyone knows about naloxone and no one is embarrassed to talk about it, why are people still dying [from opioid overdoses] in Baltimore?”
She and her associates conducted a cross-sectional survey of 353 individuals aged 18 and older in Baltimore who self-reported a lifetime history of injection drug use. The data came from a baseline survey that was conducted as part of a randomized, controlled trial testing the efficacy of a behavioral intervention focused on the Hepatitis C cascade. Individuals were asked to answer questions related to the five steps of the naloxone cascade: awareness (have you ever heard about naloxone?), access (have you ever received naloxone?), training (have you ever been trained to use naloxone?), use (have you ever used naloxone during an opiate overdose?), and possession (how often do you carry naloxone?)
More than half of the survey respondents (65%) were male; mean age was 47 years. For the previous 6 months, more than half of the sample reported the use of crack (64%), heroin (74%), and other injectable drugs (57%), while 90% reported having ever witnessed an overdose – 59% in the prior year alone. Dr. Tobin and her associates found that 90% of respondents had heard about naloxone, 69% had received it, and 60% had been trained to use it. In addition, 37% reported never carrying naloxone, 38% sometimes carried it, 33% said they had used naloxone at some point, and 25% said they always carried it with them.
On multinomial regression analysis, the researchers found that carrying naloxone often or always was significantly associated with the following variables: female sex (odds ratio, 2.77), having ever witnessed an overdose (OR, 1.84), having injected in the past 12 months (OR, 1.75), and having ever used naloxone during an overdose (OR, 4.33). The latter finding is especially important, “because it means that we just have to let people practice using it,” said Dr. Tobin, who noted that more research is needed to understand reasons why injection drug users do not always carry naloxone. “We need to start changing the social norms about carrying naloxone. You never know when it will be useful.”
The National Institute on Drug Abuse supported the study. Dr. Tobin reported having no financial disclosures.
AT CPDD 2018
Key clinical point: Increasing possession of naloxone among people who use opiates is essential.
Major finding:. Carrying naloxone often or always was significantly associated with several variables, including female sex (odds ratio, 2.77) and having ever used naloxone during an overdose (OR, 4.33).
Study details: A cross-sectional survey of 353 individuals aged 18 and older in Baltimore who self-reported a lifetime history of injection drug use.
Disclosures: The National Institute on Drug Abuse supported the study. Dr. Tobin reported having no financial disclosures.
Do free meals to physicians affect opioid prescribing?
SAN DIEGO – Physicians who receive gifts and free meals from opioid manufacturers prescribe more opioids than do their counterparts, a new study suggests.
A sampling of doctors who reported marketing payments or gifts prescribed more of the drugs the following year even as their colleagues prescribed fewer. Researchers also found signs of a dose-effect relationship between more free meals received and more opioid medications prescribed.
The findings, presented at the annual meeting of the College on Problems of Drug Dependence and recently published, do not prove a link between free meals and the massive, deadly opioid epidemic. And the purpose of pharmaceutical marketing, of course, is to persuade physicians to prescribe medications, the researchers noted. The report was published in JAMA Internal Medicine.
Still, in light of the opioid epidemic, “there’s a national effort to reduce overprescribing. Our database suggests that the pharmaceutical industry may be a counterforce,” lead author and pediatrician Scott E. Hadland, MD, MPH, of Boston University, said in an interview.
The findings suggest “it doesn’t take much money to get doctors to potentially prescribe more opioids,” he added.
According to data from the Centers for Disease Control and Prevention, a record number of people – 52,404 – died from drug overdoses in 2015, and nearly 13,000 of the deaths were attributable to prescription drugs (natural or semi-synthetic). An estimated 12.5 million people aged 12 and older in 2015, meanwhile, recently had misused prescription pain relievers.
For the new study, Dr. Hadland and his colleagues sought to understand whether opioid marketing in 2014 influenced prescribing in 2015.
The researchers retrospectively tracked 369,139 physicians in a Medicare Part D database who prescribed opioids in 2015 and found that 7% reported receiving opioid marketing – speaking fees ($6.2 million), meals ($1.8 million), travel ($731,000), consulting fees ($290,000), and education ($80,000).
Overall, received marketing.
“The effect is very subtle,” said Dr. Hadland, an addiction medicine specialist at the university. “Nine percent does not seem like a large number, but when you’re talking about hundreds of thousands of physicians, that’s a large number of opioids being prescribed.”
The study takes only Medicare Part D opioid prescriptions into account, and includes only about 42% of the active national physician workforce, he noted.
The researchers linked rising numbers of meals received in 2014 per physician – from 1 to more than 10 – to a steady increase in the number of opioid claims per physician. For example, physicians who received 1 meal made about 150 opioid claims, while those who received more than 10 made more than 700 claims.
As for physician motivations, Dr. Hadland said, he doesn’t believe “this is intentional for most physicians. If you asked the vast majority of physicians in our study, ‘Do you believe marketing is influencing your prescribing?’ most would say no.”
But the findings, he said, still raise questions.
Going forward, researchers plan to study the effect of opioid marketing on public health, he added.
Dr. Hadland reports funding from the National Institute on Drug Abuse, Society for Adolescent Health and Medicine, Thrasher Research Fund, and Academic Pediatric Association. Another author reports funding from NIDA. No additional relevant disclosures were reported.
SOURCE: Hadland SE et al. JAMA Intern Med. 2018 Jun 1;178(6):861-3.
SAN DIEGO – Physicians who receive gifts and free meals from opioid manufacturers prescribe more opioids than do their counterparts, a new study suggests.
A sampling of doctors who reported marketing payments or gifts prescribed more of the drugs the following year even as their colleagues prescribed fewer. Researchers also found signs of a dose-effect relationship between more free meals received and more opioid medications prescribed.
The findings, presented at the annual meeting of the College on Problems of Drug Dependence and recently published, do not prove a link between free meals and the massive, deadly opioid epidemic. And the purpose of pharmaceutical marketing, of course, is to persuade physicians to prescribe medications, the researchers noted. The report was published in JAMA Internal Medicine.
Still, in light of the opioid epidemic, “there’s a national effort to reduce overprescribing. Our database suggests that the pharmaceutical industry may be a counterforce,” lead author and pediatrician Scott E. Hadland, MD, MPH, of Boston University, said in an interview.
The findings suggest “it doesn’t take much money to get doctors to potentially prescribe more opioids,” he added.
According to data from the Centers for Disease Control and Prevention, a record number of people – 52,404 – died from drug overdoses in 2015, and nearly 13,000 of the deaths were attributable to prescription drugs (natural or semi-synthetic). An estimated 12.5 million people aged 12 and older in 2015, meanwhile, recently had misused prescription pain relievers.
For the new study, Dr. Hadland and his colleagues sought to understand whether opioid marketing in 2014 influenced prescribing in 2015.
The researchers retrospectively tracked 369,139 physicians in a Medicare Part D database who prescribed opioids in 2015 and found that 7% reported receiving opioid marketing – speaking fees ($6.2 million), meals ($1.8 million), travel ($731,000), consulting fees ($290,000), and education ($80,000).
Overall, received marketing.
“The effect is very subtle,” said Dr. Hadland, an addiction medicine specialist at the university. “Nine percent does not seem like a large number, but when you’re talking about hundreds of thousands of physicians, that’s a large number of opioids being prescribed.”
The study takes only Medicare Part D opioid prescriptions into account, and includes only about 42% of the active national physician workforce, he noted.
The researchers linked rising numbers of meals received in 2014 per physician – from 1 to more than 10 – to a steady increase in the number of opioid claims per physician. For example, physicians who received 1 meal made about 150 opioid claims, while those who received more than 10 made more than 700 claims.
As for physician motivations, Dr. Hadland said, he doesn’t believe “this is intentional for most physicians. If you asked the vast majority of physicians in our study, ‘Do you believe marketing is influencing your prescribing?’ most would say no.”
But the findings, he said, still raise questions.
Going forward, researchers plan to study the effect of opioid marketing on public health, he added.
Dr. Hadland reports funding from the National Institute on Drug Abuse, Society for Adolescent Health and Medicine, Thrasher Research Fund, and Academic Pediatric Association. Another author reports funding from NIDA. No additional relevant disclosures were reported.
SOURCE: Hadland SE et al. JAMA Intern Med. 2018 Jun 1;178(6):861-3.
SAN DIEGO – Physicians who receive gifts and free meals from opioid manufacturers prescribe more opioids than do their counterparts, a new study suggests.
A sampling of doctors who reported marketing payments or gifts prescribed more of the drugs the following year even as their colleagues prescribed fewer. Researchers also found signs of a dose-effect relationship between more free meals received and more opioid medications prescribed.
The findings, presented at the annual meeting of the College on Problems of Drug Dependence and recently published, do not prove a link between free meals and the massive, deadly opioid epidemic. And the purpose of pharmaceutical marketing, of course, is to persuade physicians to prescribe medications, the researchers noted. The report was published in JAMA Internal Medicine.
Still, in light of the opioid epidemic, “there’s a national effort to reduce overprescribing. Our database suggests that the pharmaceutical industry may be a counterforce,” lead author and pediatrician Scott E. Hadland, MD, MPH, of Boston University, said in an interview.
The findings suggest “it doesn’t take much money to get doctors to potentially prescribe more opioids,” he added.
According to data from the Centers for Disease Control and Prevention, a record number of people – 52,404 – died from drug overdoses in 2015, and nearly 13,000 of the deaths were attributable to prescription drugs (natural or semi-synthetic). An estimated 12.5 million people aged 12 and older in 2015, meanwhile, recently had misused prescription pain relievers.
For the new study, Dr. Hadland and his colleagues sought to understand whether opioid marketing in 2014 influenced prescribing in 2015.
The researchers retrospectively tracked 369,139 physicians in a Medicare Part D database who prescribed opioids in 2015 and found that 7% reported receiving opioid marketing – speaking fees ($6.2 million), meals ($1.8 million), travel ($731,000), consulting fees ($290,000), and education ($80,000).
Overall, received marketing.
“The effect is very subtle,” said Dr. Hadland, an addiction medicine specialist at the university. “Nine percent does not seem like a large number, but when you’re talking about hundreds of thousands of physicians, that’s a large number of opioids being prescribed.”
The study takes only Medicare Part D opioid prescriptions into account, and includes only about 42% of the active national physician workforce, he noted.
The researchers linked rising numbers of meals received in 2014 per physician – from 1 to more than 10 – to a steady increase in the number of opioid claims per physician. For example, physicians who received 1 meal made about 150 opioid claims, while those who received more than 10 made more than 700 claims.
As for physician motivations, Dr. Hadland said, he doesn’t believe “this is intentional for most physicians. If you asked the vast majority of physicians in our study, ‘Do you believe marketing is influencing your prescribing?’ most would say no.”
But the findings, he said, still raise questions.
Going forward, researchers plan to study the effect of opioid marketing on public health, he added.
Dr. Hadland reports funding from the National Institute on Drug Abuse, Society for Adolescent Health and Medicine, Thrasher Research Fund, and Academic Pediatric Association. Another author reports funding from NIDA. No additional relevant disclosures were reported.
SOURCE: Hadland SE et al. JAMA Intern Med. 2018 Jun 1;178(6):861-3.
REPORTING FROM CPDD 2018
Key clinical point: Opioid marketing appears to affect physician prescribing practices.
Major finding: Physicians who received opioid marketing payments and meals in 2014 prescribed an adjusted 9% more opioids in 2015 than did their opioid-prescribing colleagues.
Study details: Retrospective 2014-2015 analysis of 369,139 opioid-prescribing physicians in a Medicare Part D database.
Disclosures: Dr. Hadland reported funding from the National Institute on Drug Abuse, Society for Adolescent Health and Medicine, Thrasher Research Fund, and Academic Pediatric Association. Another author reported funding from NIDA. No additional relevant disclosures were reported.
Source: Hadland SE et al. JAMA Intern Med. 2018 Jun 1;178(6):861-3.
Implementing a health literacy assessment
Limited health literacy results in poor outcomes.
Hospitalists regularly treat patients with limited health literacy, and in many cases, the hospitalist may not even be aware of it. “Patients are unlikely to know or, more importantly, disclose their limited health literacy status,” according to a recent study.1 But hospitalists certainly see its effects: Limited health literacy often results in poor outcomes and high rates of readmittance.
“We know patients with limited health literacy are common and that they have poor health outcomes,” said study coauthor Robert Leverence, MD. “We also know there are ways to mitigate those outcomes. For that reason, we believe screening is important. In our study, we showed such routine screening is feasible in a large teaching hospital.”
The study describes the implementation of a hospital-wide routine health literacy assessment at an academic medical center initiated by nurses and applied to all adult inpatients. “We incorporated the health literacy screen and care plan into our electronic health record,” the authors wrote. “When a patient screens positive for limited health literacy, two automated responses are triggered: a one-time alert on chart entry for all users ... and a nursing care plan containing relevant educational recommendations.”
“To me it is a cringe-worthy event to give a 10-page AVS to a patient who can’t read,” Dr. Leverence added. “Health literacy screening allows us to tailor the discharge process to meet the needs of the individual patient. Once these patients are identified, then appropriate efforts can be efficiently deployed.”
Those efforts might include, at discharge, offering easy-to-read materials and teach-back, and having a caregiver in the room and a pharmacist performing bedside medication education.
Reference
1. Warring C, Pinkney J, Delvo-Favre E, et al. “Implementation of a Routine Health Literacy Assessment at an Academic Medical Center.” J Healthc Qual. doi: 10.1097/JHQ.0000000000000116
Limited health literacy results in poor outcomes.
Limited health literacy results in poor outcomes.
Hospitalists regularly treat patients with limited health literacy, and in many cases, the hospitalist may not even be aware of it. “Patients are unlikely to know or, more importantly, disclose their limited health literacy status,” according to a recent study.1 But hospitalists certainly see its effects: Limited health literacy often results in poor outcomes and high rates of readmittance.
“We know patients with limited health literacy are common and that they have poor health outcomes,” said study coauthor Robert Leverence, MD. “We also know there are ways to mitigate those outcomes. For that reason, we believe screening is important. In our study, we showed such routine screening is feasible in a large teaching hospital.”
The study describes the implementation of a hospital-wide routine health literacy assessment at an academic medical center initiated by nurses and applied to all adult inpatients. “We incorporated the health literacy screen and care plan into our electronic health record,” the authors wrote. “When a patient screens positive for limited health literacy, two automated responses are triggered: a one-time alert on chart entry for all users ... and a nursing care plan containing relevant educational recommendations.”
“To me it is a cringe-worthy event to give a 10-page AVS to a patient who can’t read,” Dr. Leverence added. “Health literacy screening allows us to tailor the discharge process to meet the needs of the individual patient. Once these patients are identified, then appropriate efforts can be efficiently deployed.”
Those efforts might include, at discharge, offering easy-to-read materials and teach-back, and having a caregiver in the room and a pharmacist performing bedside medication education.
Reference
1. Warring C, Pinkney J, Delvo-Favre E, et al. “Implementation of a Routine Health Literacy Assessment at an Academic Medical Center.” J Healthc Qual. doi: 10.1097/JHQ.0000000000000116
Hospitalists regularly treat patients with limited health literacy, and in many cases, the hospitalist may not even be aware of it. “Patients are unlikely to know or, more importantly, disclose their limited health literacy status,” according to a recent study.1 But hospitalists certainly see its effects: Limited health literacy often results in poor outcomes and high rates of readmittance.
“We know patients with limited health literacy are common and that they have poor health outcomes,” said study coauthor Robert Leverence, MD. “We also know there are ways to mitigate those outcomes. For that reason, we believe screening is important. In our study, we showed such routine screening is feasible in a large teaching hospital.”
The study describes the implementation of a hospital-wide routine health literacy assessment at an academic medical center initiated by nurses and applied to all adult inpatients. “We incorporated the health literacy screen and care plan into our electronic health record,” the authors wrote. “When a patient screens positive for limited health literacy, two automated responses are triggered: a one-time alert on chart entry for all users ... and a nursing care plan containing relevant educational recommendations.”
“To me it is a cringe-worthy event to give a 10-page AVS to a patient who can’t read,” Dr. Leverence added. “Health literacy screening allows us to tailor the discharge process to meet the needs of the individual patient. Once these patients are identified, then appropriate efforts can be efficiently deployed.”
Those efforts might include, at discharge, offering easy-to-read materials and teach-back, and having a caregiver in the room and a pharmacist performing bedside medication education.
Reference
1. Warring C, Pinkney J, Delvo-Favre E, et al. “Implementation of a Routine Health Literacy Assessment at an Academic Medical Center.” J Healthc Qual. doi: 10.1097/JHQ.0000000000000116
A patient portal for the inpatient experience
Hospitalists, nurses most impacted.
Hospitalists see patients at their most fragile – and, as a result, they have a unique opportunity to affect their health going forward.
“These moments can transform the way patients see their health and their behaviors, and any opportunity to position patients as empowered to influence their experience is one that can not be squandered,” said Timothy Huerta, PhD, MS, lead author on a study of patient portals and tablets during inpatient care.1 “In that context, hospitals have the opportunity to set expectations for engagement that can be influenced by technology. Patient portals, positioned within the inpatient setting, offer a platform to engage, empower, and educate.”
His experience – at the first and largest academic medical center to provide this technology across the entire hospital system – offers the first insight into the demands that such a process shift requires, he said. The researchers ran a 90-day pilot program giving tablets to 179 patients; subsequently, the health system committed to providing tablets for accessing inpatient portals in all seven of its hospitals. “Adopting this technology is not easy, and we continue to explore how we can use it more effectively. Our hope is that our experience can make the journey of others easier.”
Providing the technology is a necessary but insufficient component of implementation, he added. “This is not like the movie ‘Field of Dreams’ – if you build it they will come. It requires leaders to see the value proposition, champions throughout the organization to make a reality where the technology matters to the provision of care, and clinicians to see the tool as a means to a greater good.”
In hospitals, nursing staff and hospitalists are likely to be most impacted by the addition of these tools. “They will require choices – for example who will respond on what timeline to patient communication when using these tools – which requires collaboration across the institution.”
Reference
1. Huerta T, McAlearney AS, Rizer MK. “Introducing a Patient Portal and Electronic Tablets to Inpatient Care.” Ann Intern Med. 2017;167(11):816-7.
Hospitalists, nurses most impacted.
Hospitalists, nurses most impacted.
Hospitalists see patients at their most fragile – and, as a result, they have a unique opportunity to affect their health going forward.
“These moments can transform the way patients see their health and their behaviors, and any opportunity to position patients as empowered to influence their experience is one that can not be squandered,” said Timothy Huerta, PhD, MS, lead author on a study of patient portals and tablets during inpatient care.1 “In that context, hospitals have the opportunity to set expectations for engagement that can be influenced by technology. Patient portals, positioned within the inpatient setting, offer a platform to engage, empower, and educate.”
His experience – at the first and largest academic medical center to provide this technology across the entire hospital system – offers the first insight into the demands that such a process shift requires, he said. The researchers ran a 90-day pilot program giving tablets to 179 patients; subsequently, the health system committed to providing tablets for accessing inpatient portals in all seven of its hospitals. “Adopting this technology is not easy, and we continue to explore how we can use it more effectively. Our hope is that our experience can make the journey of others easier.”
Providing the technology is a necessary but insufficient component of implementation, he added. “This is not like the movie ‘Field of Dreams’ – if you build it they will come. It requires leaders to see the value proposition, champions throughout the organization to make a reality where the technology matters to the provision of care, and clinicians to see the tool as a means to a greater good.”
In hospitals, nursing staff and hospitalists are likely to be most impacted by the addition of these tools. “They will require choices – for example who will respond on what timeline to patient communication when using these tools – which requires collaboration across the institution.”
Reference
1. Huerta T, McAlearney AS, Rizer MK. “Introducing a Patient Portal and Electronic Tablets to Inpatient Care.” Ann Intern Med. 2017;167(11):816-7.
Hospitalists see patients at their most fragile – and, as a result, they have a unique opportunity to affect their health going forward.
“These moments can transform the way patients see their health and their behaviors, and any opportunity to position patients as empowered to influence their experience is one that can not be squandered,” said Timothy Huerta, PhD, MS, lead author on a study of patient portals and tablets during inpatient care.1 “In that context, hospitals have the opportunity to set expectations for engagement that can be influenced by technology. Patient portals, positioned within the inpatient setting, offer a platform to engage, empower, and educate.”
His experience – at the first and largest academic medical center to provide this technology across the entire hospital system – offers the first insight into the demands that such a process shift requires, he said. The researchers ran a 90-day pilot program giving tablets to 179 patients; subsequently, the health system committed to providing tablets for accessing inpatient portals in all seven of its hospitals. “Adopting this technology is not easy, and we continue to explore how we can use it more effectively. Our hope is that our experience can make the journey of others easier.”
Providing the technology is a necessary but insufficient component of implementation, he added. “This is not like the movie ‘Field of Dreams’ – if you build it they will come. It requires leaders to see the value proposition, champions throughout the organization to make a reality where the technology matters to the provision of care, and clinicians to see the tool as a means to a greater good.”
In hospitals, nursing staff and hospitalists are likely to be most impacted by the addition of these tools. “They will require choices – for example who will respond on what timeline to patient communication when using these tools – which requires collaboration across the institution.”
Reference
1. Huerta T, McAlearney AS, Rizer MK. “Introducing a Patient Portal and Electronic Tablets to Inpatient Care.” Ann Intern Med. 2017;167(11):816-7.