User login
Diagnosis and Management of Immunoglobulin Light Chain Amyloidosis
The term amyloidosis refers to a fascinating group of disorders that share a common pathogenesis of extracellular deposition of amyloid material. Fundamentally, it is a disorder of the secondary structure of select proteins whereby the amyloidogenic proteins are misfolded into a β-pleated sheet configuration, resulting in the formation of insoluble extracellular amyloid fibrils. The amyloid fibrils appear as amorphous eosinophilic material when hematoxylin and eosin–stained tissue is examined under light microscope. Electron microscopy reveals remarkable similarity between the amyloid fibrils derived from different precursor proteins in that they range from 7.5 to 10 nm in diameter. This ultrastructural similarity is the underlying basis for the characteristic red-green birefringence with Congo red staining observed under polarized microscopy, the pathological hallmark of the disease.
To read the full article in PDF:
The term amyloidosis refers to a fascinating group of disorders that share a common pathogenesis of extracellular deposition of amyloid material. Fundamentally, it is a disorder of the secondary structure of select proteins whereby the amyloidogenic proteins are misfolded into a β-pleated sheet configuration, resulting in the formation of insoluble extracellular amyloid fibrils. The amyloid fibrils appear as amorphous eosinophilic material when hematoxylin and eosin–stained tissue is examined under light microscope. Electron microscopy reveals remarkable similarity between the amyloid fibrils derived from different precursor proteins in that they range from 7.5 to 10 nm in diameter. This ultrastructural similarity is the underlying basis for the characteristic red-green birefringence with Congo red staining observed under polarized microscopy, the pathological hallmark of the disease.
To read the full article in PDF:
The term amyloidosis refers to a fascinating group of disorders that share a common pathogenesis of extracellular deposition of amyloid material. Fundamentally, it is a disorder of the secondary structure of select proteins whereby the amyloidogenic proteins are misfolded into a β-pleated sheet configuration, resulting in the formation of insoluble extracellular amyloid fibrils. The amyloid fibrils appear as amorphous eosinophilic material when hematoxylin and eosin–stained tissue is examined under light microscope. Electron microscopy reveals remarkable similarity between the amyloid fibrils derived from different precursor proteins in that they range from 7.5 to 10 nm in diameter. This ultrastructural similarity is the underlying basis for the characteristic red-green birefringence with Congo red staining observed under polarized microscopy, the pathological hallmark of the disease.
To read the full article in PDF:
Takotsubo Cardiomyopathy: A Clinical Overview
Takotsubo cardiomyopathy (TTC) is characterized by transient wall motion abnormalities of the left ventricle (LV), resulting in apical ballooning. Despite sporadic reports noting right ventricular (RV) involvement, research to date has mainly focused on LV pathology. However, Elesber and colleagues, the first group to systematically evaluate RV involvement in TTC, found RV dysfunction in eight of 25 patients.1
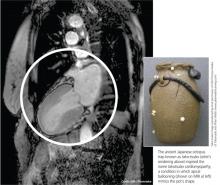
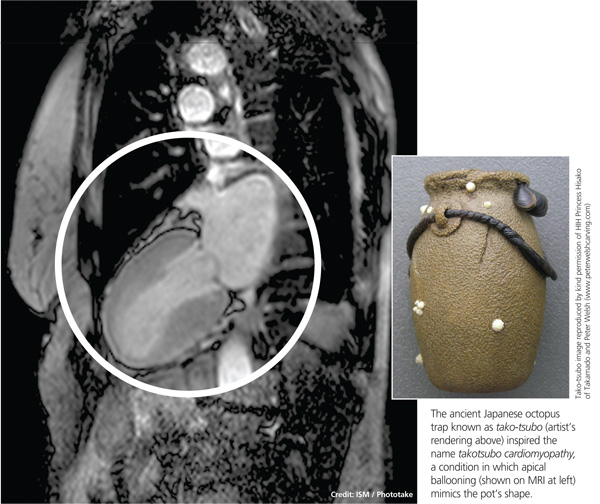
The condition is known by many different names: stress-induced cardiomyopathy, broken heart syndrome, and ampulla cardiomyopathy. The term takotsubo cardiomyopathy is derived from the appearance of the mid-ventricle and apex of the heart on echocardiography or catheterization during systole; this apical ballooning resembles a spherical bottle with a narrow neck, not unlike the ancient Japanese octopus trap called tako-tsubo (see image). First described in Japanese articles in 1991 by Dote and colleagues, TTC is not typically associated with coronary artery stenosis on angiography.2
TTC is diagnosed in approximately 1% to 2% of patients who present with signs and symptoms similar to those of acute myocardial infarction (AMI).3 The majority of affected patients are postmenopausal women, with two studies showing an 89% to 90% female predominance and mean age at presentation ranging from 58 to 77 and 58 to 75 years, respectively.4,5
The pathogenesis of TTC has been postulated to include multivessel epicardial spasm, catecholamine-mediated myocardial damage, microvascular coronary spasm or dysfunction, and neurogenic myocardial stunning.6 TTC is often not recognized on initial presentation, as it mimics AMI with ST elevation. Providers should maintain a high clinical suspicion for this transient clinical condition, which is increasingly recognized in various populations.4
Etiology and Pathophysiology
The etiology of TTC remains unclear, but a physiologic or emotional stressor usually precedes the onset of symptoms.7 It is hypothesized that a catecholamine surge in response to a stressor causes myocardial stunning through an uncertain mechanism.
Catecholamine-induced myocardial stunning due to various stressors has been documented through measurement of plasma levels of these hormones in more than 70% of patients with TTC.3 Myocardial scintillography with 123I-metaiodobenzylguanidine (MIBG) in these patients demonstrated a decreased uptake of a radiotracer in several segments of the LV, emphasizing a severe adrenalin secretion production due to stress. Considerable individual differences in MIBG uptake in patients with TTC may reflect variable responses to adrenergic stimulation, due to differences in genetic inheritance of adrenalin synthesis, functions, storage, and elimination.8
Additionally, Lyon and colleagues demonstrated a higher density of beta-adrenergic receptors in the apical region in these patients.9 They hypothesized that excessive levels of circulating catecholamines significantly alter the apical heart muscle cells, decreasing the force or energy of the muscle contraction. Conversely, Dandel and colleagues suggest that the akinetic appearance of this region can be related to high systolic apical circumferential wall stress.10
A recent literature review of 42 articles reported a possible link between TTC and drugs that overstimulate the sympathetic system.11 Consequently, clinicians should maintain a high suspicion for drug-induced TTC in patients who present with symptoms consistent with TTC but who have not experienced particularly stressful events immediately prior to onset.
The reason TTC predominately affects postmenopausal women remains unclear, but a link with reduced levels of estrogen and effects on the microvascular system has been proposed.5 One study reports a patient with TTC who carried a mutation of gene FMR1 (alleles with sizes between 40 and 55 triplet permutations); the researchers recommend further study of the role of cardiac genes in the acute phase of TTC.12 A familial apical ballooning syndrome was reported in a mother and daughter in another study, suggesting that certain women may be genetically predisposed to TTC; this may explain why only a minority of postmenopausal women are susceptible.13 However, there is no known association with single nucleotide polymorphisms for adrenergic receptors (based on a study that compared women with TTC to a control group).14
Patient Presentation
Chest pain is one of the most concerning symptoms for patients and primary care providers. The priority for a patient presenting with chest pain is to exclude catastrophic or life-threatening causes. Therefore, primary care providers are encouraged to refer patients to the nearest emergency department for appropriate diagnostic evaluation.
Precipitating events are typically severe emotional or physiologic stressors, but the absence of such a stressor does not exclude the diagnosis. Examples of emotional stressors include learning of the death of a loved one, a personal financial blow, legal problems, natural disasters, and motor vehicle collisions.4 Physiologic stressors, such as a severe medical illness, worsening chronic disease, or a noncardiac surgical procedure, can also trigger an episode of TTC.15
Patients present with chest pain and dyspnea that resemble the symptoms of AMI.4 Most patients will report chest pain at rest, although some may have dyspnea as their sole presenting complaint. They may also report palpitations, nausea, and vomiting. Patients who present with TTC typically have few traditional cardiac risk factors, such as hypertension, hyperlipidemia, diabetes, smoking, or a family history of cardiovascular disease.3
Although hemodynamic stability is the norm, a small number of patients may demonstrate hypotension due to reduced stroke volume and LV outflow tract obstruction and require hemodynamic support during the acute phase. Mild-to-moderate congestive heart failure commonly accompanies the condition.16 Rarely, patients will report an episode of syncope or an out-of-hospital cardiac arrest.17 Rare complications of TTC are cardiogenic shock and formation of a thrombus at the LV apex due to apical ballooning akinesis.17,18
Diagnostic Studies
Electrocardiography (ECG) findings may demonstrate ST-segment elevation, although this occurs in only one-half of patients with TTC.17 The elevation is typically noted in the precordial leads,16 but the ECG can be normal or show nonspecific T-wave abnormalities.17 In patients with ST-segment elevation, the severity of ventricular dysfunction or prognosis does not correlate with the ECG changes noted on presentation.19 The ST-segment elevation is followed by T-wave inversions and QT prolongation.16 Torsades de pointes and QT prolongation in patients with TTC has been reported.20
Laboratory studies demonstrate mildly elevated cardiac troponin and brain natriuretic peptide levels.3,21,22 LV wall-motion abnormalities and a severely depressed ejection fraction have been noted on transthoracic echocardiography.23 Several case reports utilized cardiac magnetic resonance imaging in patients with TTC, resulting in findings of wall motion abnormalities, myocardial edema, and hyperenhancement on contrast-enhanced imaging.24,25
Diagnosis
Although universally accepted diagnostic criteria are currently unavailable,23 Bybee and colleagues have proposed criteria for the diagnosis of TTC.6 All four of the following criteria must be met:
• Transient hypokinesis, akinesis, or dyskinesis of the LV apical and midventricular segments with regional wall-motion abnormalities encompassing more than a single epicardial vascular distribution.
• Absence of obstructive coronary disease or angiographic evidence of acute plaque rupture.
• New ECG abnormalities (either ST-segment or T-wave inversion) or modest elevation of cardiac troponin levels.
• Absence of recent significant head trauma, intracranial bleeding, pheochromocytoma, obstructive epicardial coronary artery disease, myocarditis, and hypertrophic cardiomyopathy.
The diagnosis of TTC is often made in an emergency setting because of the presenting complaint of chest pain. Typically, patients with TTC have no associated significant atherosclerotic luminal narrowing7 despite the presence of transient apical and left midventricular systolic dysfunction.
Treatment and Management
Optimal management of TTC has yet to be established, but the general approach is supportive and conservative. Because the presenting chest pain is indistinguishable from AMI, initial management should focus on preventing ischemia. Continuous ECG monitoring and administration of nitrates, morphine for pain control, aspirin, IV heparin, and beta-blockers are recommended.26
Once the diagnosis of TTC is confirmed, if there is no coexisting coronary atherosclerosis, aspirin therapy can be discontinued.17 Treatment with beta-blockers (in hemodynamically stable patients) and ACE inhibitors (in the absence of outflow tract obstruction) is usually recommended, although randomized trials have not been conducted.27
Beta-blockers, which may inhibit the release of catecholamines, could be beneficial since they are hypothesized to mediate in TTC. Additionally, beta-blockers work to reduce LV outflow tract obstruction through basal segment hypercontractility.3 For patients with associated congestive heart failure, diuretics may be effective. For significant hypotension, phenylephrine helps to increase afterload and LV cavity size; note that inotropes are contraindicated in this situation. In the rare occurrence of LV thrombus, anticoagulation is recommended.16,18 Long-term administration of beta-blockers is recommended to reduce the likelihood of TTC recurrence.17
Prognosis
In the absence of underlying comorbid conditions, the prognosis in TTC is generally good. Cardiovascular symptoms—systolic dysfunction and regional wall-motion abnormality—usually resolve completely within days to one month; an alternative diagnosis should be considered if the cardiomyopathy does not resolve after this time. Close follow-up with a cardiologist, usually with serial echocardiograms, in the weeks after diagnosis is recommended to ensure complete resolution. At six-week follow-up, the ECG usually demonstrates complete resolution, although T-wave inversion may persist.17
Inpatient mortality rates associated with TTC range from 0% to 8%.18,28 The recurrence rate has been reported as less than 10%, but additional studies are needed to track recurrence, in addition to the longitudinal effects of this condition.29 Left-sided heart failure, with or without pulmonary edema, is the most common complication associated with TTC. Others are LV mural clot, systemic or pulmonic embolic events, mitral valve regurgitation, and ventricular arrhythmias.3
Conclusion
TTC is an entity of acute heart failure that can mimic AMI. It should be considered in symptomatic postmenopausal women with a normal heart and no history of cardiovascular disease. Providers should include TTC in the differential diagnosis, especially when patients present with acute chest pain after a stressful incident. While short-term management may suffice, providers should follow these patients over time to identify the potential for long-term impact and possible causes of this condition.
References
1. Elesber AA, Prasad A, Bybee KA, et al. Transient cardiac apical ballooning syndrome: prevalence and clinical implications of right ventricular involvement. J Am Coll Cardiol. 2006;47:1082-1083.
2. Dote K, Sato H, Tateishi H, et al. Myocardial stunning due to simultaneous multivessel coronary spasms: a view of 5 cases. J Cardiol. 1991;24:471-476.
3. Pilgrim TM, Wyss TR. Takotsubo cardiomyopathy or transient left ventricular apical ballooning syndrome: a systematic review. Int J Cardiol. 2008;124:283-292.
4. Gianni M, Dentali F, Grandi AM, et al. Apical ballooning syndrome or takotsubo cardiomyopathy; a systematic review. Eur Heart J. 2006;27: 1523-1529.
5. Prasad A, Lerman A, Rihal CS. Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): a mimic of acute myocardial infarction. Am Heart J. 2008;155:408-417.
6. Bybee KA, Prasad A, Barsness GW. Clinical characteristics and thrombolysis in myocardial infarction frame counts in women with transient left ventricular apical ballooning syndrome. Am J Cardiol. 2004; 94:343-346.
7. Sharkey SW, Lesser JR, Zenovich AG, et al. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation. 2005;111:472-479.
8. Soares-Filho GL, Felix RC, Azevedo JC, et al. Broken heart or takotsubo syndrome: support for the neurohumoral hypothesis of stress cardiomyopathy. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:
247-249.
9. Lyon AR, Rees PS, Prasad S, Poole-Wilson PA, Harding SE. Stress (Takotsubo) cardiomyopathy—a novel pathophysiological hypothesis to explain catecholamine-induced acute myocardial stunning. Nat Clin Pract Cardiovasc Med. 2008;5:22-29.
10. Dandel M, Lehmkuhl H, Knosalla C, Hetzer R. Left ventricular wall motion abnormality and myocardial dysfunction in stress cardiomyopathy: new pathophysiological aspects suggested by echocardiography. Int J Cardiol. 2009;135:e40-43.
11. Amariles P. A comprehensive literature search: drugs as possible triggers of Takotsubo cardiomyopathy. Curr Clin Pharmacol. 2011;6:1-11.
12. Kleinfeldt T, Schneider H, Akin I, et al. Detection of FMR1-gene in Takotsubo cardiomyopathy: a new piece of the puzzle. Int J Cardiol. 2009;137:e81-83.
13. Kumar G, Holmes DR, Prasad A. “Familial” apical ballooning syndrome (Takotsubo cardiomyopathy). Int J Cardiol. 2010;144:444-445.
14. Sharkey SW, Maron BJ, Nelson P, et al. Adrenergic receptor polymorphisms in patients with stress (tako-tsubo) cardiomyopathy. J Cardiol. 2009;53:53-57.
15. Le Ven F, Pennec PY, Timsit S, Blanc JJ. Takotsubo syndrome associated with seizures: an underestimated cause of sudden death in epilepsy? Int J Cardiol. 2011;146:475-479.
16. Bybee KA, Kara T, Prasad A, et al. Systematic review: transient left ventricular apical ballooning: a syndrome that mimics ST-segment elevation myocardial infarction. Ann Intern Med. 2004;141:858-865.
17. Prasad A. Apical ballooning syndrome: an important differential diagnosis of acute myocardial infarction. Circulation, 2007;111:e56-e59.
18. Lee PH, Song JK, Sun BJ, et al. Outcomes of patients with stress-induced cardiomyopathy diagnosed by echocardiography in a tertiary referral hospital. J Am Soc Echocardiogr. 2010;23:766-771.
19. Dib C, Asirvatham S, Elesber A, et al. Clinical correlates and prognostic significance of electrocardiographic abnormalities in apical ballooning syndrome (Takotsubo/stress-induced cardiomyopathy). Am Heart J. 2009;157:933-938.
20. Denney SD, Lakkireddy DR, Khan IA. Long QT syndrome and torsade de pointes in transient left ventricular apical ballooning syndrome. Int J Cardio. 2005;100:499-501.
21. Song BG, Park SJ, Noh HJ, et al. Clinical characteristics, and laboratory and echocardiographic findings in takotsubo cardiomyopathy presenting as cardiogenic shock. J Crit Care. 2010;25:329-335.
22. Primetshofer D, Agladze R, Kratzer H, et al. Tako-Tsubo syndrome: an important differential diagnosis in patients with acute chest pain. Wien Klin Wochenschr. 2010;122:37-44.
23. Ando G, Trio O, Gregorio C. Transient left ventricular apical ballooning syndrome and cardiac dysfunction after subarachnoid hemorrhage: similar clinical entities? Open Emerg Med J. 2009;2:8-10.
24. Caudron J, Rey N, Dacher JN. Midventricular Takotsubo cardiomyopathy associated with ventricular fibrillation during general anaesthesia in 34-year-old woman: insight from cardiac computed tomography and magnetic resonance imaging. Arch Cardiovasc Dis. 2012;105:329-331.
25. Matsumoto H, Matsuda T, Miyamoto K. Early enhancement on contrast-enhanced cardiovascular magnetic resonance imaging in takotsubo cardiomyopathy: two cases. Int J Cardiol. 2012;155:e54-e56.
26. Van de Werf F, Bas J, Betriu A, et al; ESC Committee for Practice Guidelines. Management of acute myocardial infarction in patients presenting with persistent ST-segment elevation: the Task Force on the Management of ST-segment Elevation Acute Myocardial Infarction of the European Society of Cardiology. Eur Heart J. 2008;29:2909-2945.
27. Tomich EB, Luerssen E, Kang CS. Takotsubo cardiomyopathy. http://emedicine.medscape.com/article/1513631-overview. Accessed August 14, 2013.
28. Silva C, Goncalves A, Almedia R, et al. Transient left ventricular ballooning syndrome. Eur J Intern Med. 2009;20:454-456.
29. Elesber AA, Prasad A, Lennon RJ, et al. Four-year recurrence rate and prognosis of the apical ballooning syndrome. J Am Coll Cardiol. 2007;50:448-452.
Takotsubo cardiomyopathy (TTC) is characterized by transient wall motion abnormalities of the left ventricle (LV), resulting in apical ballooning. Despite sporadic reports noting right ventricular (RV) involvement, research to date has mainly focused on LV pathology. However, Elesber and colleagues, the first group to systematically evaluate RV involvement in TTC, found RV dysfunction in eight of 25 patients.1
The condition is known by many different names: stress-induced cardiomyopathy, broken heart syndrome, and ampulla cardiomyopathy. The term takotsubo cardiomyopathy is derived from the appearance of the mid-ventricle and apex of the heart on echocardiography or catheterization during systole; this apical ballooning resembles a spherical bottle with a narrow neck, not unlike the ancient Japanese octopus trap called tako-tsubo (see image). First described in Japanese articles in 1991 by Dote and colleagues, TTC is not typically associated with coronary artery stenosis on angiography.2
TTC is diagnosed in approximately 1% to 2% of patients who present with signs and symptoms similar to those of acute myocardial infarction (AMI).3 The majority of affected patients are postmenopausal women, with two studies showing an 89% to 90% female predominance and mean age at presentation ranging from 58 to 77 and 58 to 75 years, respectively.4,5
The pathogenesis of TTC has been postulated to include multivessel epicardial spasm, catecholamine-mediated myocardial damage, microvascular coronary spasm or dysfunction, and neurogenic myocardial stunning.6 TTC is often not recognized on initial presentation, as it mimics AMI with ST elevation. Providers should maintain a high clinical suspicion for this transient clinical condition, which is increasingly recognized in various populations.4
Etiology and Pathophysiology
The etiology of TTC remains unclear, but a physiologic or emotional stressor usually precedes the onset of symptoms.7 It is hypothesized that a catecholamine surge in response to a stressor causes myocardial stunning through an uncertain mechanism.
Catecholamine-induced myocardial stunning due to various stressors has been documented through measurement of plasma levels of these hormones in more than 70% of patients with TTC.3 Myocardial scintillography with 123I-metaiodobenzylguanidine (MIBG) in these patients demonstrated a decreased uptake of a radiotracer in several segments of the LV, emphasizing a severe adrenalin secretion production due to stress. Considerable individual differences in MIBG uptake in patients with TTC may reflect variable responses to adrenergic stimulation, due to differences in genetic inheritance of adrenalin synthesis, functions, storage, and elimination.8
Additionally, Lyon and colleagues demonstrated a higher density of beta-adrenergic receptors in the apical region in these patients.9 They hypothesized that excessive levels of circulating catecholamines significantly alter the apical heart muscle cells, decreasing the force or energy of the muscle contraction. Conversely, Dandel and colleagues suggest that the akinetic appearance of this region can be related to high systolic apical circumferential wall stress.10
A recent literature review of 42 articles reported a possible link between TTC and drugs that overstimulate the sympathetic system.11 Consequently, clinicians should maintain a high suspicion for drug-induced TTC in patients who present with symptoms consistent with TTC but who have not experienced particularly stressful events immediately prior to onset.
The reason TTC predominately affects postmenopausal women remains unclear, but a link with reduced levels of estrogen and effects on the microvascular system has been proposed.5 One study reports a patient with TTC who carried a mutation of gene FMR1 (alleles with sizes between 40 and 55 triplet permutations); the researchers recommend further study of the role of cardiac genes in the acute phase of TTC.12 A familial apical ballooning syndrome was reported in a mother and daughter in another study, suggesting that certain women may be genetically predisposed to TTC; this may explain why only a minority of postmenopausal women are susceptible.13 However, there is no known association with single nucleotide polymorphisms for adrenergic receptors (based on a study that compared women with TTC to a control group).14
Patient Presentation
Chest pain is one of the most concerning symptoms for patients and primary care providers. The priority for a patient presenting with chest pain is to exclude catastrophic or life-threatening causes. Therefore, primary care providers are encouraged to refer patients to the nearest emergency department for appropriate diagnostic evaluation.
Precipitating events are typically severe emotional or physiologic stressors, but the absence of such a stressor does not exclude the diagnosis. Examples of emotional stressors include learning of the death of a loved one, a personal financial blow, legal problems, natural disasters, and motor vehicle collisions.4 Physiologic stressors, such as a severe medical illness, worsening chronic disease, or a noncardiac surgical procedure, can also trigger an episode of TTC.15
Patients present with chest pain and dyspnea that resemble the symptoms of AMI.4 Most patients will report chest pain at rest, although some may have dyspnea as their sole presenting complaint. They may also report palpitations, nausea, and vomiting. Patients who present with TTC typically have few traditional cardiac risk factors, such as hypertension, hyperlipidemia, diabetes, smoking, or a family history of cardiovascular disease.3
Although hemodynamic stability is the norm, a small number of patients may demonstrate hypotension due to reduced stroke volume and LV outflow tract obstruction and require hemodynamic support during the acute phase. Mild-to-moderate congestive heart failure commonly accompanies the condition.16 Rarely, patients will report an episode of syncope or an out-of-hospital cardiac arrest.17 Rare complications of TTC are cardiogenic shock and formation of a thrombus at the LV apex due to apical ballooning akinesis.17,18
Diagnostic Studies
Electrocardiography (ECG) findings may demonstrate ST-segment elevation, although this occurs in only one-half of patients with TTC.17 The elevation is typically noted in the precordial leads,16 but the ECG can be normal or show nonspecific T-wave abnormalities.17 In patients with ST-segment elevation, the severity of ventricular dysfunction or prognosis does not correlate with the ECG changes noted on presentation.19 The ST-segment elevation is followed by T-wave inversions and QT prolongation.16 Torsades de pointes and QT prolongation in patients with TTC has been reported.20
Laboratory studies demonstrate mildly elevated cardiac troponin and brain natriuretic peptide levels.3,21,22 LV wall-motion abnormalities and a severely depressed ejection fraction have been noted on transthoracic echocardiography.23 Several case reports utilized cardiac magnetic resonance imaging in patients with TTC, resulting in findings of wall motion abnormalities, myocardial edema, and hyperenhancement on contrast-enhanced imaging.24,25
Diagnosis
Although universally accepted diagnostic criteria are currently unavailable,23 Bybee and colleagues have proposed criteria for the diagnosis of TTC.6 All four of the following criteria must be met:
• Transient hypokinesis, akinesis, or dyskinesis of the LV apical and midventricular segments with regional wall-motion abnormalities encompassing more than a single epicardial vascular distribution.
• Absence of obstructive coronary disease or angiographic evidence of acute plaque rupture.
• New ECG abnormalities (either ST-segment or T-wave inversion) or modest elevation of cardiac troponin levels.
• Absence of recent significant head trauma, intracranial bleeding, pheochromocytoma, obstructive epicardial coronary artery disease, myocarditis, and hypertrophic cardiomyopathy.
The diagnosis of TTC is often made in an emergency setting because of the presenting complaint of chest pain. Typically, patients with TTC have no associated significant atherosclerotic luminal narrowing7 despite the presence of transient apical and left midventricular systolic dysfunction.
Treatment and Management
Optimal management of TTC has yet to be established, but the general approach is supportive and conservative. Because the presenting chest pain is indistinguishable from AMI, initial management should focus on preventing ischemia. Continuous ECG monitoring and administration of nitrates, morphine for pain control, aspirin, IV heparin, and beta-blockers are recommended.26
Once the diagnosis of TTC is confirmed, if there is no coexisting coronary atherosclerosis, aspirin therapy can be discontinued.17 Treatment with beta-blockers (in hemodynamically stable patients) and ACE inhibitors (in the absence of outflow tract obstruction) is usually recommended, although randomized trials have not been conducted.27
Beta-blockers, which may inhibit the release of catecholamines, could be beneficial since they are hypothesized to mediate in TTC. Additionally, beta-blockers work to reduce LV outflow tract obstruction through basal segment hypercontractility.3 For patients with associated congestive heart failure, diuretics may be effective. For significant hypotension, phenylephrine helps to increase afterload and LV cavity size; note that inotropes are contraindicated in this situation. In the rare occurrence of LV thrombus, anticoagulation is recommended.16,18 Long-term administration of beta-blockers is recommended to reduce the likelihood of TTC recurrence.17
Prognosis
In the absence of underlying comorbid conditions, the prognosis in TTC is generally good. Cardiovascular symptoms—systolic dysfunction and regional wall-motion abnormality—usually resolve completely within days to one month; an alternative diagnosis should be considered if the cardiomyopathy does not resolve after this time. Close follow-up with a cardiologist, usually with serial echocardiograms, in the weeks after diagnosis is recommended to ensure complete resolution. At six-week follow-up, the ECG usually demonstrates complete resolution, although T-wave inversion may persist.17
Inpatient mortality rates associated with TTC range from 0% to 8%.18,28 The recurrence rate has been reported as less than 10%, but additional studies are needed to track recurrence, in addition to the longitudinal effects of this condition.29 Left-sided heart failure, with or without pulmonary edema, is the most common complication associated with TTC. Others are LV mural clot, systemic or pulmonic embolic events, mitral valve regurgitation, and ventricular arrhythmias.3
Conclusion
TTC is an entity of acute heart failure that can mimic AMI. It should be considered in symptomatic postmenopausal women with a normal heart and no history of cardiovascular disease. Providers should include TTC in the differential diagnosis, especially when patients present with acute chest pain after a stressful incident. While short-term management may suffice, providers should follow these patients over time to identify the potential for long-term impact and possible causes of this condition.
References
1. Elesber AA, Prasad A, Bybee KA, et al. Transient cardiac apical ballooning syndrome: prevalence and clinical implications of right ventricular involvement. J Am Coll Cardiol. 2006;47:1082-1083.
2. Dote K, Sato H, Tateishi H, et al. Myocardial stunning due to simultaneous multivessel coronary spasms: a view of 5 cases. J Cardiol. 1991;24:471-476.
3. Pilgrim TM, Wyss TR. Takotsubo cardiomyopathy or transient left ventricular apical ballooning syndrome: a systematic review. Int J Cardiol. 2008;124:283-292.
4. Gianni M, Dentali F, Grandi AM, et al. Apical ballooning syndrome or takotsubo cardiomyopathy; a systematic review. Eur Heart J. 2006;27: 1523-1529.
5. Prasad A, Lerman A, Rihal CS. Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): a mimic of acute myocardial infarction. Am Heart J. 2008;155:408-417.
6. Bybee KA, Prasad A, Barsness GW. Clinical characteristics and thrombolysis in myocardial infarction frame counts in women with transient left ventricular apical ballooning syndrome. Am J Cardiol. 2004; 94:343-346.
7. Sharkey SW, Lesser JR, Zenovich AG, et al. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation. 2005;111:472-479.
8. Soares-Filho GL, Felix RC, Azevedo JC, et al. Broken heart or takotsubo syndrome: support for the neurohumoral hypothesis of stress cardiomyopathy. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:
247-249.
9. Lyon AR, Rees PS, Prasad S, Poole-Wilson PA, Harding SE. Stress (Takotsubo) cardiomyopathy—a novel pathophysiological hypothesis to explain catecholamine-induced acute myocardial stunning. Nat Clin Pract Cardiovasc Med. 2008;5:22-29.
10. Dandel M, Lehmkuhl H, Knosalla C, Hetzer R. Left ventricular wall motion abnormality and myocardial dysfunction in stress cardiomyopathy: new pathophysiological aspects suggested by echocardiography. Int J Cardiol. 2009;135:e40-43.
11. Amariles P. A comprehensive literature search: drugs as possible triggers of Takotsubo cardiomyopathy. Curr Clin Pharmacol. 2011;6:1-11.
12. Kleinfeldt T, Schneider H, Akin I, et al. Detection of FMR1-gene in Takotsubo cardiomyopathy: a new piece of the puzzle. Int J Cardiol. 2009;137:e81-83.
13. Kumar G, Holmes DR, Prasad A. “Familial” apical ballooning syndrome (Takotsubo cardiomyopathy). Int J Cardiol. 2010;144:444-445.
14. Sharkey SW, Maron BJ, Nelson P, et al. Adrenergic receptor polymorphisms in patients with stress (tako-tsubo) cardiomyopathy. J Cardiol. 2009;53:53-57.
15. Le Ven F, Pennec PY, Timsit S, Blanc JJ. Takotsubo syndrome associated with seizures: an underestimated cause of sudden death in epilepsy? Int J Cardiol. 2011;146:475-479.
16. Bybee KA, Kara T, Prasad A, et al. Systematic review: transient left ventricular apical ballooning: a syndrome that mimics ST-segment elevation myocardial infarction. Ann Intern Med. 2004;141:858-865.
17. Prasad A. Apical ballooning syndrome: an important differential diagnosis of acute myocardial infarction. Circulation, 2007;111:e56-e59.
18. Lee PH, Song JK, Sun BJ, et al. Outcomes of patients with stress-induced cardiomyopathy diagnosed by echocardiography in a tertiary referral hospital. J Am Soc Echocardiogr. 2010;23:766-771.
19. Dib C, Asirvatham S, Elesber A, et al. Clinical correlates and prognostic significance of electrocardiographic abnormalities in apical ballooning syndrome (Takotsubo/stress-induced cardiomyopathy). Am Heart J. 2009;157:933-938.
20. Denney SD, Lakkireddy DR, Khan IA. Long QT syndrome and torsade de pointes in transient left ventricular apical ballooning syndrome. Int J Cardio. 2005;100:499-501.
21. Song BG, Park SJ, Noh HJ, et al. Clinical characteristics, and laboratory and echocardiographic findings in takotsubo cardiomyopathy presenting as cardiogenic shock. J Crit Care. 2010;25:329-335.
22. Primetshofer D, Agladze R, Kratzer H, et al. Tako-Tsubo syndrome: an important differential diagnosis in patients with acute chest pain. Wien Klin Wochenschr. 2010;122:37-44.
23. Ando G, Trio O, Gregorio C. Transient left ventricular apical ballooning syndrome and cardiac dysfunction after subarachnoid hemorrhage: similar clinical entities? Open Emerg Med J. 2009;2:8-10.
24. Caudron J, Rey N, Dacher JN. Midventricular Takotsubo cardiomyopathy associated with ventricular fibrillation during general anaesthesia in 34-year-old woman: insight from cardiac computed tomography and magnetic resonance imaging. Arch Cardiovasc Dis. 2012;105:329-331.
25. Matsumoto H, Matsuda T, Miyamoto K. Early enhancement on contrast-enhanced cardiovascular magnetic resonance imaging in takotsubo cardiomyopathy: two cases. Int J Cardiol. 2012;155:e54-e56.
26. Van de Werf F, Bas J, Betriu A, et al; ESC Committee for Practice Guidelines. Management of acute myocardial infarction in patients presenting with persistent ST-segment elevation: the Task Force on the Management of ST-segment Elevation Acute Myocardial Infarction of the European Society of Cardiology. Eur Heart J. 2008;29:2909-2945.
27. Tomich EB, Luerssen E, Kang CS. Takotsubo cardiomyopathy. http://emedicine.medscape.com/article/1513631-overview. Accessed August 14, 2013.
28. Silva C, Goncalves A, Almedia R, et al. Transient left ventricular ballooning syndrome. Eur J Intern Med. 2009;20:454-456.
29. Elesber AA, Prasad A, Lennon RJ, et al. Four-year recurrence rate and prognosis of the apical ballooning syndrome. J Am Coll Cardiol. 2007;50:448-452.
Takotsubo cardiomyopathy (TTC) is characterized by transient wall motion abnormalities of the left ventricle (LV), resulting in apical ballooning. Despite sporadic reports noting right ventricular (RV) involvement, research to date has mainly focused on LV pathology. However, Elesber and colleagues, the first group to systematically evaluate RV involvement in TTC, found RV dysfunction in eight of 25 patients.1
The condition is known by many different names: stress-induced cardiomyopathy, broken heart syndrome, and ampulla cardiomyopathy. The term takotsubo cardiomyopathy is derived from the appearance of the mid-ventricle and apex of the heart on echocardiography or catheterization during systole; this apical ballooning resembles a spherical bottle with a narrow neck, not unlike the ancient Japanese octopus trap called tako-tsubo (see image). First described in Japanese articles in 1991 by Dote and colleagues, TTC is not typically associated with coronary artery stenosis on angiography.2
TTC is diagnosed in approximately 1% to 2% of patients who present with signs and symptoms similar to those of acute myocardial infarction (AMI).3 The majority of affected patients are postmenopausal women, with two studies showing an 89% to 90% female predominance and mean age at presentation ranging from 58 to 77 and 58 to 75 years, respectively.4,5
The pathogenesis of TTC has been postulated to include multivessel epicardial spasm, catecholamine-mediated myocardial damage, microvascular coronary spasm or dysfunction, and neurogenic myocardial stunning.6 TTC is often not recognized on initial presentation, as it mimics AMI with ST elevation. Providers should maintain a high clinical suspicion for this transient clinical condition, which is increasingly recognized in various populations.4
Etiology and Pathophysiology
The etiology of TTC remains unclear, but a physiologic or emotional stressor usually precedes the onset of symptoms.7 It is hypothesized that a catecholamine surge in response to a stressor causes myocardial stunning through an uncertain mechanism.
Catecholamine-induced myocardial stunning due to various stressors has been documented through measurement of plasma levels of these hormones in more than 70% of patients with TTC.3 Myocardial scintillography with 123I-metaiodobenzylguanidine (MIBG) in these patients demonstrated a decreased uptake of a radiotracer in several segments of the LV, emphasizing a severe adrenalin secretion production due to stress. Considerable individual differences in MIBG uptake in patients with TTC may reflect variable responses to adrenergic stimulation, due to differences in genetic inheritance of adrenalin synthesis, functions, storage, and elimination.8
Additionally, Lyon and colleagues demonstrated a higher density of beta-adrenergic receptors in the apical region in these patients.9 They hypothesized that excessive levels of circulating catecholamines significantly alter the apical heart muscle cells, decreasing the force or energy of the muscle contraction. Conversely, Dandel and colleagues suggest that the akinetic appearance of this region can be related to high systolic apical circumferential wall stress.10
A recent literature review of 42 articles reported a possible link between TTC and drugs that overstimulate the sympathetic system.11 Consequently, clinicians should maintain a high suspicion for drug-induced TTC in patients who present with symptoms consistent with TTC but who have not experienced particularly stressful events immediately prior to onset.
The reason TTC predominately affects postmenopausal women remains unclear, but a link with reduced levels of estrogen and effects on the microvascular system has been proposed.5 One study reports a patient with TTC who carried a mutation of gene FMR1 (alleles with sizes between 40 and 55 triplet permutations); the researchers recommend further study of the role of cardiac genes in the acute phase of TTC.12 A familial apical ballooning syndrome was reported in a mother and daughter in another study, suggesting that certain women may be genetically predisposed to TTC; this may explain why only a minority of postmenopausal women are susceptible.13 However, there is no known association with single nucleotide polymorphisms for adrenergic receptors (based on a study that compared women with TTC to a control group).14
Patient Presentation
Chest pain is one of the most concerning symptoms for patients and primary care providers. The priority for a patient presenting with chest pain is to exclude catastrophic or life-threatening causes. Therefore, primary care providers are encouraged to refer patients to the nearest emergency department for appropriate diagnostic evaluation.
Precipitating events are typically severe emotional or physiologic stressors, but the absence of such a stressor does not exclude the diagnosis. Examples of emotional stressors include learning of the death of a loved one, a personal financial blow, legal problems, natural disasters, and motor vehicle collisions.4 Physiologic stressors, such as a severe medical illness, worsening chronic disease, or a noncardiac surgical procedure, can also trigger an episode of TTC.15
Patients present with chest pain and dyspnea that resemble the symptoms of AMI.4 Most patients will report chest pain at rest, although some may have dyspnea as their sole presenting complaint. They may also report palpitations, nausea, and vomiting. Patients who present with TTC typically have few traditional cardiac risk factors, such as hypertension, hyperlipidemia, diabetes, smoking, or a family history of cardiovascular disease.3
Although hemodynamic stability is the norm, a small number of patients may demonstrate hypotension due to reduced stroke volume and LV outflow tract obstruction and require hemodynamic support during the acute phase. Mild-to-moderate congestive heart failure commonly accompanies the condition.16 Rarely, patients will report an episode of syncope or an out-of-hospital cardiac arrest.17 Rare complications of TTC are cardiogenic shock and formation of a thrombus at the LV apex due to apical ballooning akinesis.17,18
Diagnostic Studies
Electrocardiography (ECG) findings may demonstrate ST-segment elevation, although this occurs in only one-half of patients with TTC.17 The elevation is typically noted in the precordial leads,16 but the ECG can be normal or show nonspecific T-wave abnormalities.17 In patients with ST-segment elevation, the severity of ventricular dysfunction or prognosis does not correlate with the ECG changes noted on presentation.19 The ST-segment elevation is followed by T-wave inversions and QT prolongation.16 Torsades de pointes and QT prolongation in patients with TTC has been reported.20
Laboratory studies demonstrate mildly elevated cardiac troponin and brain natriuretic peptide levels.3,21,22 LV wall-motion abnormalities and a severely depressed ejection fraction have been noted on transthoracic echocardiography.23 Several case reports utilized cardiac magnetic resonance imaging in patients with TTC, resulting in findings of wall motion abnormalities, myocardial edema, and hyperenhancement on contrast-enhanced imaging.24,25
Diagnosis
Although universally accepted diagnostic criteria are currently unavailable,23 Bybee and colleagues have proposed criteria for the diagnosis of TTC.6 All four of the following criteria must be met:
• Transient hypokinesis, akinesis, or dyskinesis of the LV apical and midventricular segments with regional wall-motion abnormalities encompassing more than a single epicardial vascular distribution.
• Absence of obstructive coronary disease or angiographic evidence of acute plaque rupture.
• New ECG abnormalities (either ST-segment or T-wave inversion) or modest elevation of cardiac troponin levels.
• Absence of recent significant head trauma, intracranial bleeding, pheochromocytoma, obstructive epicardial coronary artery disease, myocarditis, and hypertrophic cardiomyopathy.
The diagnosis of TTC is often made in an emergency setting because of the presenting complaint of chest pain. Typically, patients with TTC have no associated significant atherosclerotic luminal narrowing7 despite the presence of transient apical and left midventricular systolic dysfunction.
Treatment and Management
Optimal management of TTC has yet to be established, but the general approach is supportive and conservative. Because the presenting chest pain is indistinguishable from AMI, initial management should focus on preventing ischemia. Continuous ECG monitoring and administration of nitrates, morphine for pain control, aspirin, IV heparin, and beta-blockers are recommended.26
Once the diagnosis of TTC is confirmed, if there is no coexisting coronary atherosclerosis, aspirin therapy can be discontinued.17 Treatment with beta-blockers (in hemodynamically stable patients) and ACE inhibitors (in the absence of outflow tract obstruction) is usually recommended, although randomized trials have not been conducted.27
Beta-blockers, which may inhibit the release of catecholamines, could be beneficial since they are hypothesized to mediate in TTC. Additionally, beta-blockers work to reduce LV outflow tract obstruction through basal segment hypercontractility.3 For patients with associated congestive heart failure, diuretics may be effective. For significant hypotension, phenylephrine helps to increase afterload and LV cavity size; note that inotropes are contraindicated in this situation. In the rare occurrence of LV thrombus, anticoagulation is recommended.16,18 Long-term administration of beta-blockers is recommended to reduce the likelihood of TTC recurrence.17
Prognosis
In the absence of underlying comorbid conditions, the prognosis in TTC is generally good. Cardiovascular symptoms—systolic dysfunction and regional wall-motion abnormality—usually resolve completely within days to one month; an alternative diagnosis should be considered if the cardiomyopathy does not resolve after this time. Close follow-up with a cardiologist, usually with serial echocardiograms, in the weeks after diagnosis is recommended to ensure complete resolution. At six-week follow-up, the ECG usually demonstrates complete resolution, although T-wave inversion may persist.17
Inpatient mortality rates associated with TTC range from 0% to 8%.18,28 The recurrence rate has been reported as less than 10%, but additional studies are needed to track recurrence, in addition to the longitudinal effects of this condition.29 Left-sided heart failure, with or without pulmonary edema, is the most common complication associated with TTC. Others are LV mural clot, systemic or pulmonic embolic events, mitral valve regurgitation, and ventricular arrhythmias.3
Conclusion
TTC is an entity of acute heart failure that can mimic AMI. It should be considered in symptomatic postmenopausal women with a normal heart and no history of cardiovascular disease. Providers should include TTC in the differential diagnosis, especially when patients present with acute chest pain after a stressful incident. While short-term management may suffice, providers should follow these patients over time to identify the potential for long-term impact and possible causes of this condition.
References
1. Elesber AA, Prasad A, Bybee KA, et al. Transient cardiac apical ballooning syndrome: prevalence and clinical implications of right ventricular involvement. J Am Coll Cardiol. 2006;47:1082-1083.
2. Dote K, Sato H, Tateishi H, et al. Myocardial stunning due to simultaneous multivessel coronary spasms: a view of 5 cases. J Cardiol. 1991;24:471-476.
3. Pilgrim TM, Wyss TR. Takotsubo cardiomyopathy or transient left ventricular apical ballooning syndrome: a systematic review. Int J Cardiol. 2008;124:283-292.
4. Gianni M, Dentali F, Grandi AM, et al. Apical ballooning syndrome or takotsubo cardiomyopathy; a systematic review. Eur Heart J. 2006;27: 1523-1529.
5. Prasad A, Lerman A, Rihal CS. Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): a mimic of acute myocardial infarction. Am Heart J. 2008;155:408-417.
6. Bybee KA, Prasad A, Barsness GW. Clinical characteristics and thrombolysis in myocardial infarction frame counts in women with transient left ventricular apical ballooning syndrome. Am J Cardiol. 2004; 94:343-346.
7. Sharkey SW, Lesser JR, Zenovich AG, et al. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation. 2005;111:472-479.
8. Soares-Filho GL, Felix RC, Azevedo JC, et al. Broken heart or takotsubo syndrome: support for the neurohumoral hypothesis of stress cardiomyopathy. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:
247-249.
9. Lyon AR, Rees PS, Prasad S, Poole-Wilson PA, Harding SE. Stress (Takotsubo) cardiomyopathy—a novel pathophysiological hypothesis to explain catecholamine-induced acute myocardial stunning. Nat Clin Pract Cardiovasc Med. 2008;5:22-29.
10. Dandel M, Lehmkuhl H, Knosalla C, Hetzer R. Left ventricular wall motion abnormality and myocardial dysfunction in stress cardiomyopathy: new pathophysiological aspects suggested by echocardiography. Int J Cardiol. 2009;135:e40-43.
11. Amariles P. A comprehensive literature search: drugs as possible triggers of Takotsubo cardiomyopathy. Curr Clin Pharmacol. 2011;6:1-11.
12. Kleinfeldt T, Schneider H, Akin I, et al. Detection of FMR1-gene in Takotsubo cardiomyopathy: a new piece of the puzzle. Int J Cardiol. 2009;137:e81-83.
13. Kumar G, Holmes DR, Prasad A. “Familial” apical ballooning syndrome (Takotsubo cardiomyopathy). Int J Cardiol. 2010;144:444-445.
14. Sharkey SW, Maron BJ, Nelson P, et al. Adrenergic receptor polymorphisms in patients with stress (tako-tsubo) cardiomyopathy. J Cardiol. 2009;53:53-57.
15. Le Ven F, Pennec PY, Timsit S, Blanc JJ. Takotsubo syndrome associated with seizures: an underestimated cause of sudden death in epilepsy? Int J Cardiol. 2011;146:475-479.
16. Bybee KA, Kara T, Prasad A, et al. Systematic review: transient left ventricular apical ballooning: a syndrome that mimics ST-segment elevation myocardial infarction. Ann Intern Med. 2004;141:858-865.
17. Prasad A. Apical ballooning syndrome: an important differential diagnosis of acute myocardial infarction. Circulation, 2007;111:e56-e59.
18. Lee PH, Song JK, Sun BJ, et al. Outcomes of patients with stress-induced cardiomyopathy diagnosed by echocardiography in a tertiary referral hospital. J Am Soc Echocardiogr. 2010;23:766-771.
19. Dib C, Asirvatham S, Elesber A, et al. Clinical correlates and prognostic significance of electrocardiographic abnormalities in apical ballooning syndrome (Takotsubo/stress-induced cardiomyopathy). Am Heart J. 2009;157:933-938.
20. Denney SD, Lakkireddy DR, Khan IA. Long QT syndrome and torsade de pointes in transient left ventricular apical ballooning syndrome. Int J Cardio. 2005;100:499-501.
21. Song BG, Park SJ, Noh HJ, et al. Clinical characteristics, and laboratory and echocardiographic findings in takotsubo cardiomyopathy presenting as cardiogenic shock. J Crit Care. 2010;25:329-335.
22. Primetshofer D, Agladze R, Kratzer H, et al. Tako-Tsubo syndrome: an important differential diagnosis in patients with acute chest pain. Wien Klin Wochenschr. 2010;122:37-44.
23. Ando G, Trio O, Gregorio C. Transient left ventricular apical ballooning syndrome and cardiac dysfunction after subarachnoid hemorrhage: similar clinical entities? Open Emerg Med J. 2009;2:8-10.
24. Caudron J, Rey N, Dacher JN. Midventricular Takotsubo cardiomyopathy associated with ventricular fibrillation during general anaesthesia in 34-year-old woman: insight from cardiac computed tomography and magnetic resonance imaging. Arch Cardiovasc Dis. 2012;105:329-331.
25. Matsumoto H, Matsuda T, Miyamoto K. Early enhancement on contrast-enhanced cardiovascular magnetic resonance imaging in takotsubo cardiomyopathy: two cases. Int J Cardiol. 2012;155:e54-e56.
26. Van de Werf F, Bas J, Betriu A, et al; ESC Committee for Practice Guidelines. Management of acute myocardial infarction in patients presenting with persistent ST-segment elevation: the Task Force on the Management of ST-segment Elevation Acute Myocardial Infarction of the European Society of Cardiology. Eur Heart J. 2008;29:2909-2945.
27. Tomich EB, Luerssen E, Kang CS. Takotsubo cardiomyopathy. http://emedicine.medscape.com/article/1513631-overview. Accessed August 14, 2013.
28. Silva C, Goncalves A, Almedia R, et al. Transient left ventricular ballooning syndrome. Eur J Intern Med. 2009;20:454-456.
29. Elesber AA, Prasad A, Lennon RJ, et al. Four-year recurrence rate and prognosis of the apical ballooning syndrome. J Am Coll Cardiol. 2007;50:448-452.
The push is on for universal influenza vaccines
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
EXPERT OPINION FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
Adaptability and Resiliency of Military Families During Reunification: Results of a Longitudinal Study
A Veterans Health Administration Imperative: Recommendations for Detecting Anxiety in Older Adults
Competing Cardiovascular Outcomes
When to Worry About Incidental Renal and Adrenal Masses
› Use computed tomography studies and the Bosniak classification system to
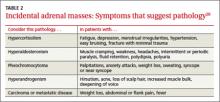
guide management of renal cystic masses. A
› Perform laboratory tests for hypercortisolism, hyperaldosteronism, and hypersecretion of catecholamines (pheochromocytoma) on any patient with an incidental adrenal mass, regardless of signs or symptoms. C
› Refer patients with adrenal masses >4 cm for surgical evaluation. Refer any individual who has a history of malignancy and an adrenal mass for oncologic evaluation. B
Strength of recommendation (SOR)
A. Good-quality patient-oriented evidence
B. Inconsistent or limited-quality patient-oriented evidence
C. Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Jane C, a 76-year-old patient, reports lower abdominal discomfort and increased bowel movements. Her left lower quadrant is tender to palpation, without signs of a surgical abdomen, and vital signs are normal. Laboratory studies are also normal, except for mild anemia and a positive fecal occult blood test. Abdominal and pelvic computed tomography (CT), with and without contrast, are negative for acute pathology, but a 1.7-cm lesion is found in the upper pole of the left kidney. What is your next step?
Renal or adrenal masses may be discovered during imaging studies for complaints unrelated to the kidneys or adrenals. Detection of incidentalomas has increased dramatically, keeping pace with the growing use of ultrasonography, CT, and magnetic resonance imaging (MRI) for abdominal, chest, and back complaints.1
Family physicians can evaluate most of these masses and determine the need for referral by using clinical judgment, appropriate imaging studies, and screening laboratory tests. In the pages that follow, we present a systematic approach for evaluating these incidentalomas and determining when consultation or referral is needed.
Incidental renal masses are common
Lesions are commonly found in normal kidneys, and the incidence increases with age. Approximately one-third of individuals age 50 and older will have at least one renal cyst on CT.2
Most incidental renal masses are benign cysts requiring no further evaluation. Other possibilities include indeterminate or malignant cysts or solid masses, which may be malignant or benign. Inflammatory renal lesions from infection, infarction, or trauma also occur, but these tend to be symptomatic and are rarely found incidentally.
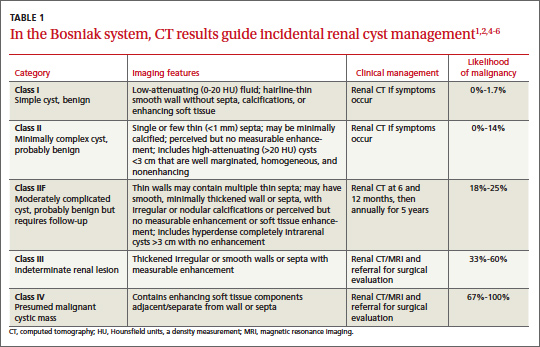
Classification of renal cysts—not based on size
Cysts are the most common adult renal masses. Typically they are unilocular and located in the renal cortex, frequently extending to the renal surface.3 Renal function is usually preserved, regardless of the cyst’s location or size. Careful examination of adjacent tissue is essential, as secondary cysts may form when solid tumors obstruct tubules of normal parenchyma. Cystic lesions containing enhancing soft tissue unattached to the wall or septa likely are malignant.4
The Bosniak classification system, with 5 classes based on CT characteristics
(TABLE 1), is a useful guide for managing renal cystic lesions.4 Size is not an important feature in the Bosniak system; small cysts may be malignant and larger ones benign. Small cysts may grow into larger benign lesions, occasionally causing flank or abdominal pain, palpable masses, or hematuria.
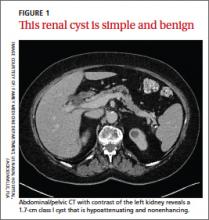
Simple cysts. Renal cysts that meet Bosniak class I criteria can be confidently labeled benign and need no further evaluation (FIGURE 1). Simple renal cysts on CT have homogenous low-attenuating fluid and thin nonenhancing walls without septa.4
On ultrasound, simple renal cysts show spherical or ovoid shape without internal echoes, a thin smooth wall separate from the surrounding parenchyma, and posterior wall enhancement caused by increased transmission through the water-filled cyst. The likelihood of malignancy is extremely low in a renal cyst that meets these criteria, which have a reported accuracy of 98% to 100%.3 Thus, no further evaluation is required if an obviously benign simple cyst is first noted on an adequate ultrasound. Inadequate ultrasound visualization or evidence of calcifications, septa, or multiple chambers calls for prompt renal CT.
CASE The mass on Ms. C’s left kidney is hypoattenuating and nonenhancing on CT. It meets Bosniak criteria for a benign simple cyst (class I) and requires no further evaluation or follow-up. Colonoscopy detects multiple colonic polyps that are removed, and the patient does well.
Mildly complicated cysts. Less diagnostic certainty characterizes cysts with mild abnormalities that keep them from being labeled as simple. Bosniak classes II and IIF describe mildly abnormal renal cysts. Class II cysts can be dismissed, whereas class IIF cysts require follow-up.
Class II cysts may contain a few hairline septa, fine calcium deposits in walls or septa, or an unmeasurable enhancement of the walls. A hyperattenuating but nonenhancing fluid also is described as category II. Small homogeneous cysts <3 cm, without enhancement but hyperattenuated, are reliably considered benign and need not be evaluated.2,7
Class IIF cysts may have multiple hairline-thin septa with unmeasurable enhancement or minimal smooth thickening or irregular/nodular calcifications of wall or septa without enhancing soft tissue components. Hyperattenuating cystic lesions >3 cm and intrarenal “noncortical” cysts are included in this category. Class IIF cysts require follow-up at 6 months with CT or MRI, then annually for at least 5 years.8
Obviously complicated cysts. Bosniak class III is indeterminate—neither benign nor clearly malignant. Class III cysts may have thickened borders or septa with measurable enhancement, or they may be multilocular, hemorrhagic, or infected. In 5 case series, 29 of 57 class III lesions proved to be malignant.5 MRI may characterize these lesions more definitively than CT prior to urologic referral.
Malignant cysts. Bosniak class IV renal lesions are clearly malignant, with large heterogeneous cysts or necrotic components, shaggy thickened walls, or enhancing soft tissue components separate from the wall or septa. Their unequivocal appearance results from solid tumor necrosis and liquefaction. Diagnosis is straightforward, and excision is indicated.2
A closer look at solid renal masses
Solid renal masses usually consist of enhancing tissue with little or no fluid. The goal of evaluation is to exclude malignancies, such as renal cell cancer, lymphomas, sarcomas, or metastasis. Benign solid masses include renal adenomas, angiomyolipomas, and oncocytomas, among others.
Several lesions can be diagnosed by appearance or symptoms:
Angiomyolipomas are recognized by their fat content within a noncalcified mass. Unenhanced CT usually is sufficient for diagnosis, unless the mass is very small or has atypical features.9
Vascular lesions can be identified because they enhance to the same degree as the vasculature. With the exception of inflammatory or vascular abnormalities, all enhancing lesions that do not contain fat should be presumed to be malignant.
In patients with a known extrarenal primary malignancy, 50% to 85% of incidental solid renal masses will represent metastatic disease.10 Percutaneous biopsy may be warranted to differentiate metastatic lesions from a secondary, primary (ie, renal cell carcinoma), or benign process.11
A study of 2770 solid renal mass excisions revealed that 12.8% were benign, with a direct relationship between malignancy and size. Masses <1 cm were benign 44% of the time.12 Early identification of small renal carcinomas may improve survival rates. Although renal cell carcinomas <3 cm in diameter have low metastatic potential, a solid, nonfat-containing mass should be evaluated for aggressive nephron-sparing surgery.6,13
Incidental adrenal masses occur infrequently
Adrenal incidentalomas are defined as radiographically identified masses >1 cm in diameter.14 They are much less common than their renal counterparts, with a reported prevalence of 0.35% to 5% on CT.15 Because the adrenal glands are hormonally active and receive substantial blood flow, metastatic, hormonally active, and nonfunctional causes for adrenal masses need to be considered.16
Adrenal pathology
Adrenal masses may be characterized by increased or normal adrenal function. Hyperfunctioning syndromes include hypercortisolism, hyperaldosteronism, adrenogenital hypersecretion of adrenocortical origin, and pheochromocytomas of the medulla. Symptom evaluation of these syndromes is important, but not sufficient to rule out a hyperfunctioning syndrome.
In a retrospective review of inapparent adrenal masses, ≤13% of pheochromocytomas were clinically silent.17 Therefore, laboratory testing is necessary for an incidental adrenal mass.
Nonfunctional lesions include adenomas, metastases, cysts, myelolipomas, hemorrhage, and adrenal carcinomas. These masses require evaluation for the possibility of cancer, the most common of which is metastasis. In patients with an extra-adrenal malignancy, the likelihood of malignancy in an incidental adrenal mass is at least 50%.18 An adrenal mass representing metastasis of a previously unrecognized cancer is exceedingly rare.19
Primary adrenal carcinoma is also rare, with an estimated incidence of 2 cases per one million in the general population. For patients with adrenal masses, the prevalence of carcinoma increases with lesion size (2% for tumors <4 cm, 6% for tumors 4-6 cm, and 25% for tumors >6 cm in diameter). 17 For this reason, tumors >4 cm in diameter are usually surgically resected in patients with no previous cancer history, unless radiologic criteria demonstrate clearly benign characteristics.
Although adrenal carcinomas are considered nonfunctioning, some evidence suggests they produce low levels of cortisol that may be associated with clinical features of metabolic syndrome.20
CT is first choice for adrenal mass evaluation
Dedicated adrenal CT with both unenhanced and delayed contrast-enhanced images is the most reliable study to evaluate an adrenal mass, according to the American College of Radiology. Consider another study only in patients with contrast allergy, renal compromise, or cancer history.21
Unenhanced CT can diagnose the approximately 70% of adenomas that are small, well-defined round masses with homogenous low-density lipid deposition.22 Delayed contrast enhancement can characterize most of the remaining 30%.23 Unenhanced CT with attenuation values of <10 Hounsfield units (HU) can diagnose adenomas with 71% specificity and 98% sensitivity,24 and can often diagnose simple cysts and myelolipomas, as well.
Other imaging options. MRI is an alternative to CT for patients with contraindications for contrast or radiation exposure. MRI provides less spatial resolution than CT, but chemical shift imaging can measure cytoplasmic lipid content similar to unenhanced CT. A small study found chemical shift MRI more reliable than unenhanced CT, but less reliable than CT with delayed contrast enhancement.25
Positron emission tomography (PET) is useful to noninvasively evaluate biochemical and physiologic processes. PET-CT incorporates unenhanced CT density measurements to improve PET accuracy. In a patient with a history of cancer, PET-CT has a sensitivity of 93% to 100% and a specificity of 95% in differentiating benign from malignant adrenal tumors.26
When to order a biopsy
The need for biopsy has decreased as imaging has improved, but biopsy is required whenever diagnostic imaging fails to differentiatea lesion as benign or malignant. CT guided biopsy provides diagnostic accuracy of 85% to 95%.27 Complications such as pneumothorax, hemorrhage, and bacteremia occur in 3% to 9% of biopsies. Before any adrenal biopsy, measure plasma-free metanephrines to exclude undiagnosed pheochromocytoma, which could precipitate a hypertensive crisis if untreated.22
These 3 laboratory screening tests are critical
Family physicians can perform the initial biochemical evaluation of an adrenal incidentaloma. Guidance is available from the National Institutes of Health (NIH)28 and the American Academy of Clinical Endocrinologists (AACE) (FIGURE 2).29
Regardless of signs or symptoms, perform screening laboratory tests for 3 types of adrenal hyperfunction: hypercortisolism, hyperaldosteronism, and hypersecretion of catecholamines (pheochromocytoma). Screening tests are not recommended for androgen hypersecretion, which is extremely rare and causes recognizable symptoms such as hirsutism (Table 2).29
Hypercortisolism occurs in approximately 5% of adrenal incidentalomas.30 An overnight dexamethasone suppression test (DST) is most reliable for screening, with sensitivity >95% for Cushing syndrome.31 The patient takes a 1-mg dose of oral dexamethasone at 11 pm, and a fasting plasma cortisol sample is drawn the next day at 8 am.
Dexamethasone binds to glucocorticoid receptors in the pituitary gland, suppressing adrenocorticotropic hormone secretion. Cortisol will be depressed the next morning unless the adrenal mass produces cortisol autonomously. Patients with a DST >5 mcg/dL—highly suggestive of Cushing syndrome—require further evaluation, and we suggest referral to an endocrinologist.
Hyperaldosteronism is seen in 1% to 2% of adrenal incidentalomas.32 The aldosterone- to-renin ratio (ARR) is recommended as a screening test for hyperaldosteronism, with an ARR >20 requiring further testing.33 Medications that may affect the ARR include beta-blockers, spironolactone, clonidine, diuretics, angiotensin-converting enzyme inhibitors, and angiotensin receptor blockers.29
Refer a patient with evidence of hyperaldosteronism to an endocrinologist and a surgeon with experience in managing these lesions. If the ARR test result suggests an aldosterone excess, a salt-loading test is used to verify failure of aldosterone suppression. Adrenal venous sampling is often performed prior to surgical removal to confirm that an incidentaloma is the source of hyperaldosteronism.
Pheochromocytoma. Approximately 5% of incidental adrenal lesions are pheochromocytomas.30 Many patients with these epinephrine/norepinephrine secreting tumors do not show the classic symptom triad of headache, palpitations, and diaphoresis, and approximately half have normal blood pressure.34
Identifying a pheochromocytoma is important in any patient requiring surgery or biopsy, as surgical manipulation can cause a potentially fatal intraoperative catecholamine surge. Presurgical medical management can mitigate this reaction.
A plasma-free metanephrines test, which has 95% sensitivity, is the most reliable test for pheochromocytoma.35 Medications, including tricyclic antidepressants, decongestants, amphetamines, reserpine, and phenoxybenzamine, can cause falsepositive results.29 Confirm a positive plasma-free metanephrines test with a 24-hour fractionated urine metanephrines test, and refer the patient to an endocrinologist.
Managing adrenal incidentalomas
Refer all patients with adrenal masses >4 cm for surgical evaluation because of the risk of malignancy; all patients who have a history of malignancy and an adrenal mass of any size require a referral to an oncologist. Perform the AACE-recommended 3-element biochemical workup for all masses, with the exception of definitively diagnosed cysts or myelolipomas.
Refer to an endocrinologist all patients with abnormal screening laboratory results, regardless of adrenal mass size, as well as patients with concerning clinical findings. Initiate cardiovascular, diabetes, and bone density evaluation and management for metabolic syndrome.20
Monitoring after a negative workup
Little evidence exists to guide monitoring of small adrenal incidentalomas (<4 cm) with a negative workup. The 2002 NIH report recommended annual radiologic follow-up for 5 years,28 whereas the 2009 AACE guidelines recommend radiographic follow-up at 3 to 6 months, then at one and 2 years.29
Evidence indicates that 14% of lesions will enlarge in 2 years, although the clinical significance of enlargement is unknown. Some authors argue against CT monitoring because the risk of adrenal mass progression is similar to the malignancy risk posed by 3 years of radiation exposure with CT.20
Some guidelines recommend repeat biochemical screening every 3 to 4 years.28,29 AACE guidelines quote a 47% rate of progression over 3 years, but most adrenal masses progress to subclinical Cushing syndrome— a condition of uncertain significance. Subclinical Cushing’s has not been reported to progress to the overt syndrome, and new catecholamine or aldosterone secretion is rare.
Many endocrinologists reduce the frequency of follow-up, depending on the type of adrenal mass (cyst or solid) and its size. AACE suggests CT for adenomas one to 4 cm at 12 months. AACE and NIH recommend hormonal evaluation annually for 4 years. Adrenal cysts or myelolipoma in patients without cancer need no follow-up.29
CORRESPONDENCE
James C. Higgins, DO, CAPT, MC, USN, Ret., Naval Hospital Jacksonville, Family Medicine Department, 2080 Child Street, Box 1000, Jacksonville, FL 32214;
[email protected]
1. Berland LL, Silverman SG, Gore RM, et al. Managing incidental findings on abdominal CT: white paper of the ACR incidental findings committee. J Am Coll Radiol. 2010;7:754-773.
2. Silverman S, Israel G, Herts B, et al. Management of the incidental renal mass. Radiology. 2008;249:16-31.
3. Curry NS, Bissada NK. Radiologic evaluation of small and indeterminate renal masses. Urol Clin North Am. 1997;24:493-505.
4. Bosniak MA. The current radiological approach to renal cysts. Radiology. 1986;158:1-10.
5. Harisinghani M, Maher M, Gervais D, et al. Incidence of malignancy in complex cystic renal masses (Bosniak category III): should imaging guided biopsy precede surgery? AJR Am J Roentgenol. 2003;180:755-758.
6. Remzi M, Ozsoy M, Klingler HC. Are small renal tumors harmless? Analysis of histopathological features according to tumors less than 4 cm in diameter. J Urol. 2006;176:896-899.
7. Jonisch AI, Rubinowitz A, Mutalik P, et al. Can high attenuation renal cysts be differentiated from renal cell carcinoma at unenhanced computed tomography? Radiology. 2007;243:445-450.
8. Israel GM, Bosniak MA. Follow-up CT of moderately complex cystic lesions of the kidney. AJR Am J Roentgenol. 2003;181: 627-633.
9. Bosniak MA, Megibow AJ, Hulnick DH, et al. CT diagnosis of renal angiomyolipoma: the importance of detecting small amounts of fat. AJR Am J Roentengol. 1988;151:497-501.
10. Mitnick JS, Bosniak MA, Rothberg M, et al. Metastatic neoplasm to the kidney studied by computed tomography and sonogram. J Comput Assist Tomogr. 1985;9:43-49.
11. Rybicki FJ, Shu KM, Cibas ES, et al. Percutaneous biopsy of renal masses: sensitivity and negative predictive value stratified by clinical setting and size of masses. AJR Am J Roentgenol. 2003;180:1281-1287.
12. Frank I, Blure MI, Cheville JC, et al. Solid renal tumors: an analysis of pathological features related to tumor size. J Urol. 2003;170:2217-2220.
13. Hollingsworth JM, Miller DC, Daignault S, et al. Rising incidence of small renal masses: a need to reassess treatment effect. J Natl Cancer Inst. 2006;98:1331-1334.
14. Geelhoed GW, Spiegel CT. “Incidental” adrenal cyst: a correctable lesion possibly associated with hypertension. South Med J. 1981;74:626-630.
15. Davenport C, Liew A, Doherty B, et al. The prevalence of adrenal incidentaloma in routine clinical practice. Endocrine. 2011;40: 80-83.
16. Cook DM, Loriaux LD. The incidental adrenal mass. Am J Med. 1996;101:88 94.
17. Mansmann G, Lau J, Balk E, et al. The clinically inapparent adrenal mass: update in diagnosis and management. Endocr Rev. 2004;25:309-340.
18. Androulakis II, Kaltsas G, Piatitis G, et al. The clinical significance of adrenal incidentalomas. Eur J Clin Invest. 2011;41: 552-560.
19. Lee JE, Evans DB, Hickey RC, et al. Unknown primary cancer presenting as an adrenal mass: frequency and implications for diagnostic evaluation of adrenal incidentalomas. Surgery. 1998;124:1115-1122.
20. Aron D, Terzolo M, Cawood TJ. Adrenal incidentalomas. Best Pract Res Clin Endocrinol Metab. 2012;26:69-82.
21. ACR appropriateness criteria: incidentally discovered adrenal mass. American College of Radiology. Available at: http://www.acr.org/~/media/ACR/Documents/AppCriteria/Diagnostic/IncidentallyDiscoveredAdrenalMass.pdf. Accessed November 20, 2012.
22. Song JH, Mayo-Smith WW. Incidentally discovered adrenal mass. Radiol Clin North Am. 2011;49:361-368.
23. Korobkin M, Brodeur FJ, Francis IR, et al. CT time-attenuation washout curves of adrenal adenomas and nonadenomas. AJR Am J Roentgenol. 1998;170:747-752.
24. Boland GW, Lee MJ, Gazelle GS, et al. Characterization of adrenal masses using unenhanced CT: an analysis of the CT literature. AJR Am J Roentgenol. 1998;171:201-204.
25. Park BK, Kim CK, Kim B, et al. Chemical shift MR imaging of hyperattenuating (>10 HU) adrenal masses: does it still have a role? Radiology. 2004;231:711-716.
26. Boland GW, Blake MA, Holakere NS, et al. PET/CT for the characterization of adrenal masses in patients with cancer: qualitative vs quantitative accuracy in 150 consecutive patients. AJR Am J Roentgenol. 2009;192:956-962.
27. Paulsen SD, Nghiem HV, Korobkin M, et al. Changing role of imaging- guided percutaneous biopsy of adrenal masses: evaluation of 50 adrenal biopsies. AJR Am J Roentgenol. 2004;182:1033-1037
28. Grumbach MM, Biller BMK, Braunstein GD, et al. Management of the clinically inapparent adrenal mass (“incidentalomas”). Ann Intern Med. 2003;138:424-429.
29. Zeiger MA, Thompson GB, Quan-Yang D, et al. American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons medical guidelines for the management of adrenal incidentalomas. Endocr Pract. 2009;15(suppl 1):1-20.
30. Young WF. The incidentally discovered adrenal mass. N Engl J Med. 2007; 356:601-610.
31. Deutschbein T, Unger N, Hinrichs J, et al. Late-night and lowdose dexamethasone-suppressed cortisol in saliva and serum for the diagnosis of cortisol-secreting adrenal adenomas. Eur J Endocrinol. 2009;161:747-753.
32. Bernini G, Moretti A, Gianfranco A, et al. Primary aldosteronism in normokalemic patients with adrenal incidentalomas. Eur J Endocrinol. 2002;146:523-529.
33. Montori VM, Young WF Jr. Use of plasma aldosterone concentration-to-plasma renin activity ratio as a screening test for primary aldosteronism: a systematic review of the literature. Endocrinol Metab Clin North Am. 2002;31:619-632.
34. Motta-Ramirez GA, Remer EM, Herts BR, et al. Comparison of CT findings in symptomatic and incidentally discovered pheochromocytomas. AJR Am J Roentgenol. 2005;185:684-688.
35. Pacak K, Eisenhofer G, Grossman A. The incidentally discovered adrenal mass. N Engl J Med. 2007;356:2005.
› Use computed tomography studies and the Bosniak classification system to
guide management of renal cystic masses. A
› Perform laboratory tests for hypercortisolism, hyperaldosteronism, and hypersecretion of catecholamines (pheochromocytoma) on any patient with an incidental adrenal mass, regardless of signs or symptoms. C
› Refer patients with adrenal masses >4 cm for surgical evaluation. Refer any individual who has a history of malignancy and an adrenal mass for oncologic evaluation. B
Strength of recommendation (SOR)
A. Good-quality patient-oriented evidence
B. Inconsistent or limited-quality patient-oriented evidence
C. Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Jane C, a 76-year-old patient, reports lower abdominal discomfort and increased bowel movements. Her left lower quadrant is tender to palpation, without signs of a surgical abdomen, and vital signs are normal. Laboratory studies are also normal, except for mild anemia and a positive fecal occult blood test. Abdominal and pelvic computed tomography (CT), with and without contrast, are negative for acute pathology, but a 1.7-cm lesion is found in the upper pole of the left kidney. What is your next step?
Renal or adrenal masses may be discovered during imaging studies for complaints unrelated to the kidneys or adrenals. Detection of incidentalomas has increased dramatically, keeping pace with the growing use of ultrasonography, CT, and magnetic resonance imaging (MRI) for abdominal, chest, and back complaints.1
Family physicians can evaluate most of these masses and determine the need for referral by using clinical judgment, appropriate imaging studies, and screening laboratory tests. In the pages that follow, we present a systematic approach for evaluating these incidentalomas and determining when consultation or referral is needed.
Incidental renal masses are common
Lesions are commonly found in normal kidneys, and the incidence increases with age. Approximately one-third of individuals age 50 and older will have at least one renal cyst on CT.2
Most incidental renal masses are benign cysts requiring no further evaluation. Other possibilities include indeterminate or malignant cysts or solid masses, which may be malignant or benign. Inflammatory renal lesions from infection, infarction, or trauma also occur, but these tend to be symptomatic and are rarely found incidentally.

Classification of renal cysts—not based on size
Cysts are the most common adult renal masses. Typically they are unilocular and located in the renal cortex, frequently extending to the renal surface.3 Renal function is usually preserved, regardless of the cyst’s location or size. Careful examination of adjacent tissue is essential, as secondary cysts may form when solid tumors obstruct tubules of normal parenchyma. Cystic lesions containing enhancing soft tissue unattached to the wall or septa likely are malignant.4
The Bosniak classification system, with 5 classes based on CT characteristics
(TABLE 1), is a useful guide for managing renal cystic lesions.4 Size is not an important feature in the Bosniak system; small cysts may be malignant and larger ones benign. Small cysts may grow into larger benign lesions, occasionally causing flank or abdominal pain, palpable masses, or hematuria.
Simple cysts. Renal cysts that meet Bosniak class I criteria can be confidently labeled benign and need no further evaluation (FIGURE 1). Simple renal cysts on CT have homogenous low-attenuating fluid and thin nonenhancing walls without septa.4
On ultrasound, simple renal cysts show spherical or ovoid shape without internal echoes, a thin smooth wall separate from the surrounding parenchyma, and posterior wall enhancement caused by increased transmission through the water-filled cyst. The likelihood of malignancy is extremely low in a renal cyst that meets these criteria, which have a reported accuracy of 98% to 100%.3 Thus, no further evaluation is required if an obviously benign simple cyst is first noted on an adequate ultrasound. Inadequate ultrasound visualization or evidence of calcifications, septa, or multiple chambers calls for prompt renal CT.
CASE The mass on Ms. C’s left kidney is hypoattenuating and nonenhancing on CT. It meets Bosniak criteria for a benign simple cyst (class I) and requires no further evaluation or follow-up. Colonoscopy detects multiple colonic polyps that are removed, and the patient does well.
Mildly complicated cysts. Less diagnostic certainty characterizes cysts with mild abnormalities that keep them from being labeled as simple. Bosniak classes II and IIF describe mildly abnormal renal cysts. Class II cysts can be dismissed, whereas class IIF cysts require follow-up.
Class II cysts may contain a few hairline septa, fine calcium deposits in walls or septa, or an unmeasurable enhancement of the walls. A hyperattenuating but nonenhancing fluid also is described as category II. Small homogeneous cysts <3 cm, without enhancement but hyperattenuated, are reliably considered benign and need not be evaluated.2,7
Class IIF cysts may have multiple hairline-thin septa with unmeasurable enhancement or minimal smooth thickening or irregular/nodular calcifications of wall or septa without enhancing soft tissue components. Hyperattenuating cystic lesions >3 cm and intrarenal “noncortical” cysts are included in this category. Class IIF cysts require follow-up at 6 months with CT or MRI, then annually for at least 5 years.8
Obviously complicated cysts. Bosniak class III is indeterminate—neither benign nor clearly malignant. Class III cysts may have thickened borders or septa with measurable enhancement, or they may be multilocular, hemorrhagic, or infected. In 5 case series, 29 of 57 class III lesions proved to be malignant.5 MRI may characterize these lesions more definitively than CT prior to urologic referral.
Malignant cysts. Bosniak class IV renal lesions are clearly malignant, with large heterogeneous cysts or necrotic components, shaggy thickened walls, or enhancing soft tissue components separate from the wall or septa. Their unequivocal appearance results from solid tumor necrosis and liquefaction. Diagnosis is straightforward, and excision is indicated.2
A closer look at solid renal masses
Solid renal masses usually consist of enhancing tissue with little or no fluid. The goal of evaluation is to exclude malignancies, such as renal cell cancer, lymphomas, sarcomas, or metastasis. Benign solid masses include renal adenomas, angiomyolipomas, and oncocytomas, among others.
Several lesions can be diagnosed by appearance or symptoms:
Angiomyolipomas are recognized by their fat content within a noncalcified mass. Unenhanced CT usually is sufficient for diagnosis, unless the mass is very small or has atypical features.9
Vascular lesions can be identified because they enhance to the same degree as the vasculature. With the exception of inflammatory or vascular abnormalities, all enhancing lesions that do not contain fat should be presumed to be malignant.
In patients with a known extrarenal primary malignancy, 50% to 85% of incidental solid renal masses will represent metastatic disease.10 Percutaneous biopsy may be warranted to differentiate metastatic lesions from a secondary, primary (ie, renal cell carcinoma), or benign process.11
A study of 2770 solid renal mass excisions revealed that 12.8% were benign, with a direct relationship between malignancy and size. Masses <1 cm were benign 44% of the time.12 Early identification of small renal carcinomas may improve survival rates. Although renal cell carcinomas <3 cm in diameter have low metastatic potential, a solid, nonfat-containing mass should be evaluated for aggressive nephron-sparing surgery.6,13
Incidental adrenal masses occur infrequently
Adrenal incidentalomas are defined as radiographically identified masses >1 cm in diameter.14 They are much less common than their renal counterparts, with a reported prevalence of 0.35% to 5% on CT.15 Because the adrenal glands are hormonally active and receive substantial blood flow, metastatic, hormonally active, and nonfunctional causes for adrenal masses need to be considered.16
Adrenal pathology
Adrenal masses may be characterized by increased or normal adrenal function. Hyperfunctioning syndromes include hypercortisolism, hyperaldosteronism, adrenogenital hypersecretion of adrenocortical origin, and pheochromocytomas of the medulla. Symptom evaluation of these syndromes is important, but not sufficient to rule out a hyperfunctioning syndrome.
In a retrospective review of inapparent adrenal masses, ≤13% of pheochromocytomas were clinically silent.17 Therefore, laboratory testing is necessary for an incidental adrenal mass.
Nonfunctional lesions include adenomas, metastases, cysts, myelolipomas, hemorrhage, and adrenal carcinomas. These masses require evaluation for the possibility of cancer, the most common of which is metastasis. In patients with an extra-adrenal malignancy, the likelihood of malignancy in an incidental adrenal mass is at least 50%.18 An adrenal mass representing metastasis of a previously unrecognized cancer is exceedingly rare.19
Primary adrenal carcinoma is also rare, with an estimated incidence of 2 cases per one million in the general population. For patients with adrenal masses, the prevalence of carcinoma increases with lesion size (2% for tumors <4 cm, 6% for tumors 4-6 cm, and 25% for tumors >6 cm in diameter). 17 For this reason, tumors >4 cm in diameter are usually surgically resected in patients with no previous cancer history, unless radiologic criteria demonstrate clearly benign characteristics.
Although adrenal carcinomas are considered nonfunctioning, some evidence suggests they produce low levels of cortisol that may be associated with clinical features of metabolic syndrome.20
CT is first choice for adrenal mass evaluation
Dedicated adrenal CT with both unenhanced and delayed contrast-enhanced images is the most reliable study to evaluate an adrenal mass, according to the American College of Radiology. Consider another study only in patients with contrast allergy, renal compromise, or cancer history.21
Unenhanced CT can diagnose the approximately 70% of adenomas that are small, well-defined round masses with homogenous low-density lipid deposition.22 Delayed contrast enhancement can characterize most of the remaining 30%.23 Unenhanced CT with attenuation values of <10 Hounsfield units (HU) can diagnose adenomas with 71% specificity and 98% sensitivity,24 and can often diagnose simple cysts and myelolipomas, as well.
Other imaging options. MRI is an alternative to CT for patients with contraindications for contrast or radiation exposure. MRI provides less spatial resolution than CT, but chemical shift imaging can measure cytoplasmic lipid content similar to unenhanced CT. A small study found chemical shift MRI more reliable than unenhanced CT, but less reliable than CT with delayed contrast enhancement.25
Positron emission tomography (PET) is useful to noninvasively evaluate biochemical and physiologic processes. PET-CT incorporates unenhanced CT density measurements to improve PET accuracy. In a patient with a history of cancer, PET-CT has a sensitivity of 93% to 100% and a specificity of 95% in differentiating benign from malignant adrenal tumors.26
When to order a biopsy
The need for biopsy has decreased as imaging has improved, but biopsy is required whenever diagnostic imaging fails to differentiatea lesion as benign or malignant. CT guided biopsy provides diagnostic accuracy of 85% to 95%.27 Complications such as pneumothorax, hemorrhage, and bacteremia occur in 3% to 9% of biopsies. Before any adrenal biopsy, measure plasma-free metanephrines to exclude undiagnosed pheochromocytoma, which could precipitate a hypertensive crisis if untreated.22
These 3 laboratory screening tests are critical
Family physicians can perform the initial biochemical evaluation of an adrenal incidentaloma. Guidance is available from the National Institutes of Health (NIH)28 and the American Academy of Clinical Endocrinologists (AACE) (FIGURE 2).29
Regardless of signs or symptoms, perform screening laboratory tests for 3 types of adrenal hyperfunction: hypercortisolism, hyperaldosteronism, and hypersecretion of catecholamines (pheochromocytoma). Screening tests are not recommended for androgen hypersecretion, which is extremely rare and causes recognizable symptoms such as hirsutism (Table 2).29
Hypercortisolism occurs in approximately 5% of adrenal incidentalomas.30 An overnight dexamethasone suppression test (DST) is most reliable for screening, with sensitivity >95% for Cushing syndrome.31 The patient takes a 1-mg dose of oral dexamethasone at 11 pm, and a fasting plasma cortisol sample is drawn the next day at 8 am.
Dexamethasone binds to glucocorticoid receptors in the pituitary gland, suppressing adrenocorticotropic hormone secretion. Cortisol will be depressed the next morning unless the adrenal mass produces cortisol autonomously. Patients with a DST >5 mcg/dL—highly suggestive of Cushing syndrome—require further evaluation, and we suggest referral to an endocrinologist.
Hyperaldosteronism is seen in 1% to 2% of adrenal incidentalomas.32 The aldosterone- to-renin ratio (ARR) is recommended as a screening test for hyperaldosteronism, with an ARR >20 requiring further testing.33 Medications that may affect the ARR include beta-blockers, spironolactone, clonidine, diuretics, angiotensin-converting enzyme inhibitors, and angiotensin receptor blockers.29
Refer a patient with evidence of hyperaldosteronism to an endocrinologist and a surgeon with experience in managing these lesions. If the ARR test result suggests an aldosterone excess, a salt-loading test is used to verify failure of aldosterone suppression. Adrenal venous sampling is often performed prior to surgical removal to confirm that an incidentaloma is the source of hyperaldosteronism.
Pheochromocytoma. Approximately 5% of incidental adrenal lesions are pheochromocytomas.30 Many patients with these epinephrine/norepinephrine secreting tumors do not show the classic symptom triad of headache, palpitations, and diaphoresis, and approximately half have normal blood pressure.34
Identifying a pheochromocytoma is important in any patient requiring surgery or biopsy, as surgical manipulation can cause a potentially fatal intraoperative catecholamine surge. Presurgical medical management can mitigate this reaction.
A plasma-free metanephrines test, which has 95% sensitivity, is the most reliable test for pheochromocytoma.35 Medications, including tricyclic antidepressants, decongestants, amphetamines, reserpine, and phenoxybenzamine, can cause falsepositive results.29 Confirm a positive plasma-free metanephrines test with a 24-hour fractionated urine metanephrines test, and refer the patient to an endocrinologist.
Managing adrenal incidentalomas
Refer all patients with adrenal masses >4 cm for surgical evaluation because of the risk of malignancy; all patients who have a history of malignancy and an adrenal mass of any size require a referral to an oncologist. Perform the AACE-recommended 3-element biochemical workup for all masses, with the exception of definitively diagnosed cysts or myelolipomas.
Refer to an endocrinologist all patients with abnormal screening laboratory results, regardless of adrenal mass size, as well as patients with concerning clinical findings. Initiate cardiovascular, diabetes, and bone density evaluation and management for metabolic syndrome.20
Monitoring after a negative workup
Little evidence exists to guide monitoring of small adrenal incidentalomas (<4 cm) with a negative workup. The 2002 NIH report recommended annual radiologic follow-up for 5 years,28 whereas the 2009 AACE guidelines recommend radiographic follow-up at 3 to 6 months, then at one and 2 years.29
Evidence indicates that 14% of lesions will enlarge in 2 years, although the clinical significance of enlargement is unknown. Some authors argue against CT monitoring because the risk of adrenal mass progression is similar to the malignancy risk posed by 3 years of radiation exposure with CT.20
Some guidelines recommend repeat biochemical screening every 3 to 4 years.28,29 AACE guidelines quote a 47% rate of progression over 3 years, but most adrenal masses progress to subclinical Cushing syndrome— a condition of uncertain significance. Subclinical Cushing’s has not been reported to progress to the overt syndrome, and new catecholamine or aldosterone secretion is rare.
Many endocrinologists reduce the frequency of follow-up, depending on the type of adrenal mass (cyst or solid) and its size. AACE suggests CT for adenomas one to 4 cm at 12 months. AACE and NIH recommend hormonal evaluation annually for 4 years. Adrenal cysts or myelolipoma in patients without cancer need no follow-up.29
CORRESPONDENCE
James C. Higgins, DO, CAPT, MC, USN, Ret., Naval Hospital Jacksonville, Family Medicine Department, 2080 Child Street, Box 1000, Jacksonville, FL 32214;
[email protected]
› Use computed tomography studies and the Bosniak classification system to
guide management of renal cystic masses. A
› Perform laboratory tests for hypercortisolism, hyperaldosteronism, and hypersecretion of catecholamines (pheochromocytoma) on any patient with an incidental adrenal mass, regardless of signs or symptoms. C
› Refer patients with adrenal masses >4 cm for surgical evaluation. Refer any individual who has a history of malignancy and an adrenal mass for oncologic evaluation. B
Strength of recommendation (SOR)
A. Good-quality patient-oriented evidence
B. Inconsistent or limited-quality patient-oriented evidence
C. Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Jane C, a 76-year-old patient, reports lower abdominal discomfort and increased bowel movements. Her left lower quadrant is tender to palpation, without signs of a surgical abdomen, and vital signs are normal. Laboratory studies are also normal, except for mild anemia and a positive fecal occult blood test. Abdominal and pelvic computed tomography (CT), with and without contrast, are negative for acute pathology, but a 1.7-cm lesion is found in the upper pole of the left kidney. What is your next step?
Renal or adrenal masses may be discovered during imaging studies for complaints unrelated to the kidneys or adrenals. Detection of incidentalomas has increased dramatically, keeping pace with the growing use of ultrasonography, CT, and magnetic resonance imaging (MRI) for abdominal, chest, and back complaints.1
Family physicians can evaluate most of these masses and determine the need for referral by using clinical judgment, appropriate imaging studies, and screening laboratory tests. In the pages that follow, we present a systematic approach for evaluating these incidentalomas and determining when consultation or referral is needed.
Incidental renal masses are common
Lesions are commonly found in normal kidneys, and the incidence increases with age. Approximately one-third of individuals age 50 and older will have at least one renal cyst on CT.2
Most incidental renal masses are benign cysts requiring no further evaluation. Other possibilities include indeterminate or malignant cysts or solid masses, which may be malignant or benign. Inflammatory renal lesions from infection, infarction, or trauma also occur, but these tend to be symptomatic and are rarely found incidentally.

Classification of renal cysts—not based on size
Cysts are the most common adult renal masses. Typically they are unilocular and located in the renal cortex, frequently extending to the renal surface.3 Renal function is usually preserved, regardless of the cyst’s location or size. Careful examination of adjacent tissue is essential, as secondary cysts may form when solid tumors obstruct tubules of normal parenchyma. Cystic lesions containing enhancing soft tissue unattached to the wall or septa likely are malignant.4
The Bosniak classification system, with 5 classes based on CT characteristics
(TABLE 1), is a useful guide for managing renal cystic lesions.4 Size is not an important feature in the Bosniak system; small cysts may be malignant and larger ones benign. Small cysts may grow into larger benign lesions, occasionally causing flank or abdominal pain, palpable masses, or hematuria.
Simple cysts. Renal cysts that meet Bosniak class I criteria can be confidently labeled benign and need no further evaluation (FIGURE 1). Simple renal cysts on CT have homogenous low-attenuating fluid and thin nonenhancing walls without septa.4
On ultrasound, simple renal cysts show spherical or ovoid shape without internal echoes, a thin smooth wall separate from the surrounding parenchyma, and posterior wall enhancement caused by increased transmission through the water-filled cyst. The likelihood of malignancy is extremely low in a renal cyst that meets these criteria, which have a reported accuracy of 98% to 100%.3 Thus, no further evaluation is required if an obviously benign simple cyst is first noted on an adequate ultrasound. Inadequate ultrasound visualization or evidence of calcifications, septa, or multiple chambers calls for prompt renal CT.
CASE The mass on Ms. C’s left kidney is hypoattenuating and nonenhancing on CT. It meets Bosniak criteria for a benign simple cyst (class I) and requires no further evaluation or follow-up. Colonoscopy detects multiple colonic polyps that are removed, and the patient does well.
Mildly complicated cysts. Less diagnostic certainty characterizes cysts with mild abnormalities that keep them from being labeled as simple. Bosniak classes II and IIF describe mildly abnormal renal cysts. Class II cysts can be dismissed, whereas class IIF cysts require follow-up.
Class II cysts may contain a few hairline septa, fine calcium deposits in walls or septa, or an unmeasurable enhancement of the walls. A hyperattenuating but nonenhancing fluid also is described as category II. Small homogeneous cysts <3 cm, without enhancement but hyperattenuated, are reliably considered benign and need not be evaluated.2,7
Class IIF cysts may have multiple hairline-thin septa with unmeasurable enhancement or minimal smooth thickening or irregular/nodular calcifications of wall or septa without enhancing soft tissue components. Hyperattenuating cystic lesions >3 cm and intrarenal “noncortical” cysts are included in this category. Class IIF cysts require follow-up at 6 months with CT or MRI, then annually for at least 5 years.8
Obviously complicated cysts. Bosniak class III is indeterminate—neither benign nor clearly malignant. Class III cysts may have thickened borders or septa with measurable enhancement, or they may be multilocular, hemorrhagic, or infected. In 5 case series, 29 of 57 class III lesions proved to be malignant.5 MRI may characterize these lesions more definitively than CT prior to urologic referral.
Malignant cysts. Bosniak class IV renal lesions are clearly malignant, with large heterogeneous cysts or necrotic components, shaggy thickened walls, or enhancing soft tissue components separate from the wall or septa. Their unequivocal appearance results from solid tumor necrosis and liquefaction. Diagnosis is straightforward, and excision is indicated.2
A closer look at solid renal masses
Solid renal masses usually consist of enhancing tissue with little or no fluid. The goal of evaluation is to exclude malignancies, such as renal cell cancer, lymphomas, sarcomas, or metastasis. Benign solid masses include renal adenomas, angiomyolipomas, and oncocytomas, among others.
Several lesions can be diagnosed by appearance or symptoms:
Angiomyolipomas are recognized by their fat content within a noncalcified mass. Unenhanced CT usually is sufficient for diagnosis, unless the mass is very small or has atypical features.9
Vascular lesions can be identified because they enhance to the same degree as the vasculature. With the exception of inflammatory or vascular abnormalities, all enhancing lesions that do not contain fat should be presumed to be malignant.
In patients with a known extrarenal primary malignancy, 50% to 85% of incidental solid renal masses will represent metastatic disease.10 Percutaneous biopsy may be warranted to differentiate metastatic lesions from a secondary, primary (ie, renal cell carcinoma), or benign process.11
A study of 2770 solid renal mass excisions revealed that 12.8% were benign, with a direct relationship between malignancy and size. Masses <1 cm were benign 44% of the time.12 Early identification of small renal carcinomas may improve survival rates. Although renal cell carcinomas <3 cm in diameter have low metastatic potential, a solid, nonfat-containing mass should be evaluated for aggressive nephron-sparing surgery.6,13
Incidental adrenal masses occur infrequently
Adrenal incidentalomas are defined as radiographically identified masses >1 cm in diameter.14 They are much less common than their renal counterparts, with a reported prevalence of 0.35% to 5% on CT.15 Because the adrenal glands are hormonally active and receive substantial blood flow, metastatic, hormonally active, and nonfunctional causes for adrenal masses need to be considered.16
Adrenal pathology
Adrenal masses may be characterized by increased or normal adrenal function. Hyperfunctioning syndromes include hypercortisolism, hyperaldosteronism, adrenogenital hypersecretion of adrenocortical origin, and pheochromocytomas of the medulla. Symptom evaluation of these syndromes is important, but not sufficient to rule out a hyperfunctioning syndrome.
In a retrospective review of inapparent adrenal masses, ≤13% of pheochromocytomas were clinically silent.17 Therefore, laboratory testing is necessary for an incidental adrenal mass.
Nonfunctional lesions include adenomas, metastases, cysts, myelolipomas, hemorrhage, and adrenal carcinomas. These masses require evaluation for the possibility of cancer, the most common of which is metastasis. In patients with an extra-adrenal malignancy, the likelihood of malignancy in an incidental adrenal mass is at least 50%.18 An adrenal mass representing metastasis of a previously unrecognized cancer is exceedingly rare.19
Primary adrenal carcinoma is also rare, with an estimated incidence of 2 cases per one million in the general population. For patients with adrenal masses, the prevalence of carcinoma increases with lesion size (2% for tumors <4 cm, 6% for tumors 4-6 cm, and 25% for tumors >6 cm in diameter). 17 For this reason, tumors >4 cm in diameter are usually surgically resected in patients with no previous cancer history, unless radiologic criteria demonstrate clearly benign characteristics.
Although adrenal carcinomas are considered nonfunctioning, some evidence suggests they produce low levels of cortisol that may be associated with clinical features of metabolic syndrome.20
CT is first choice for adrenal mass evaluation
Dedicated adrenal CT with both unenhanced and delayed contrast-enhanced images is the most reliable study to evaluate an adrenal mass, according to the American College of Radiology. Consider another study only in patients with contrast allergy, renal compromise, or cancer history.21
Unenhanced CT can diagnose the approximately 70% of adenomas that are small, well-defined round masses with homogenous low-density lipid deposition.22 Delayed contrast enhancement can characterize most of the remaining 30%.23 Unenhanced CT with attenuation values of <10 Hounsfield units (HU) can diagnose adenomas with 71% specificity and 98% sensitivity,24 and can often diagnose simple cysts and myelolipomas, as well.
Other imaging options. MRI is an alternative to CT for patients with contraindications for contrast or radiation exposure. MRI provides less spatial resolution than CT, but chemical shift imaging can measure cytoplasmic lipid content similar to unenhanced CT. A small study found chemical shift MRI more reliable than unenhanced CT, but less reliable than CT with delayed contrast enhancement.25
Positron emission tomography (PET) is useful to noninvasively evaluate biochemical and physiologic processes. PET-CT incorporates unenhanced CT density measurements to improve PET accuracy. In a patient with a history of cancer, PET-CT has a sensitivity of 93% to 100% and a specificity of 95% in differentiating benign from malignant adrenal tumors.26
When to order a biopsy
The need for biopsy has decreased as imaging has improved, but biopsy is required whenever diagnostic imaging fails to differentiatea lesion as benign or malignant. CT guided biopsy provides diagnostic accuracy of 85% to 95%.27 Complications such as pneumothorax, hemorrhage, and bacteremia occur in 3% to 9% of biopsies. Before any adrenal biopsy, measure plasma-free metanephrines to exclude undiagnosed pheochromocytoma, which could precipitate a hypertensive crisis if untreated.22
These 3 laboratory screening tests are critical
Family physicians can perform the initial biochemical evaluation of an adrenal incidentaloma. Guidance is available from the National Institutes of Health (NIH)28 and the American Academy of Clinical Endocrinologists (AACE) (FIGURE 2).29
Regardless of signs or symptoms, perform screening laboratory tests for 3 types of adrenal hyperfunction: hypercortisolism, hyperaldosteronism, and hypersecretion of catecholamines (pheochromocytoma). Screening tests are not recommended for androgen hypersecretion, which is extremely rare and causes recognizable symptoms such as hirsutism (Table 2).29
Hypercortisolism occurs in approximately 5% of adrenal incidentalomas.30 An overnight dexamethasone suppression test (DST) is most reliable for screening, with sensitivity >95% for Cushing syndrome.31 The patient takes a 1-mg dose of oral dexamethasone at 11 pm, and a fasting plasma cortisol sample is drawn the next day at 8 am.
Dexamethasone binds to glucocorticoid receptors in the pituitary gland, suppressing adrenocorticotropic hormone secretion. Cortisol will be depressed the next morning unless the adrenal mass produces cortisol autonomously. Patients with a DST >5 mcg/dL—highly suggestive of Cushing syndrome—require further evaluation, and we suggest referral to an endocrinologist.
Hyperaldosteronism is seen in 1% to 2% of adrenal incidentalomas.32 The aldosterone- to-renin ratio (ARR) is recommended as a screening test for hyperaldosteronism, with an ARR >20 requiring further testing.33 Medications that may affect the ARR include beta-blockers, spironolactone, clonidine, diuretics, angiotensin-converting enzyme inhibitors, and angiotensin receptor blockers.29
Refer a patient with evidence of hyperaldosteronism to an endocrinologist and a surgeon with experience in managing these lesions. If the ARR test result suggests an aldosterone excess, a salt-loading test is used to verify failure of aldosterone suppression. Adrenal venous sampling is often performed prior to surgical removal to confirm that an incidentaloma is the source of hyperaldosteronism.
Pheochromocytoma. Approximately 5% of incidental adrenal lesions are pheochromocytomas.30 Many patients with these epinephrine/norepinephrine secreting tumors do not show the classic symptom triad of headache, palpitations, and diaphoresis, and approximately half have normal blood pressure.34
Identifying a pheochromocytoma is important in any patient requiring surgery or biopsy, as surgical manipulation can cause a potentially fatal intraoperative catecholamine surge. Presurgical medical management can mitigate this reaction.
A plasma-free metanephrines test, which has 95% sensitivity, is the most reliable test for pheochromocytoma.35 Medications, including tricyclic antidepressants, decongestants, amphetamines, reserpine, and phenoxybenzamine, can cause falsepositive results.29 Confirm a positive plasma-free metanephrines test with a 24-hour fractionated urine metanephrines test, and refer the patient to an endocrinologist.
Managing adrenal incidentalomas
Refer all patients with adrenal masses >4 cm for surgical evaluation because of the risk of malignancy; all patients who have a history of malignancy and an adrenal mass of any size require a referral to an oncologist. Perform the AACE-recommended 3-element biochemical workup for all masses, with the exception of definitively diagnosed cysts or myelolipomas.
Refer to an endocrinologist all patients with abnormal screening laboratory results, regardless of adrenal mass size, as well as patients with concerning clinical findings. Initiate cardiovascular, diabetes, and bone density evaluation and management for metabolic syndrome.20
Monitoring after a negative workup
Little evidence exists to guide monitoring of small adrenal incidentalomas (<4 cm) with a negative workup. The 2002 NIH report recommended annual radiologic follow-up for 5 years,28 whereas the 2009 AACE guidelines recommend radiographic follow-up at 3 to 6 months, then at one and 2 years.29
Evidence indicates that 14% of lesions will enlarge in 2 years, although the clinical significance of enlargement is unknown. Some authors argue against CT monitoring because the risk of adrenal mass progression is similar to the malignancy risk posed by 3 years of radiation exposure with CT.20
Some guidelines recommend repeat biochemical screening every 3 to 4 years.28,29 AACE guidelines quote a 47% rate of progression over 3 years, but most adrenal masses progress to subclinical Cushing syndrome— a condition of uncertain significance. Subclinical Cushing’s has not been reported to progress to the overt syndrome, and new catecholamine or aldosterone secretion is rare.
Many endocrinologists reduce the frequency of follow-up, depending on the type of adrenal mass (cyst or solid) and its size. AACE suggests CT for adenomas one to 4 cm at 12 months. AACE and NIH recommend hormonal evaluation annually for 4 years. Adrenal cysts or myelolipoma in patients without cancer need no follow-up.29
CORRESPONDENCE
James C. Higgins, DO, CAPT, MC, USN, Ret., Naval Hospital Jacksonville, Family Medicine Department, 2080 Child Street, Box 1000, Jacksonville, FL 32214;
[email protected]
1. Berland LL, Silverman SG, Gore RM, et al. Managing incidental findings on abdominal CT: white paper of the ACR incidental findings committee. J Am Coll Radiol. 2010;7:754-773.
2. Silverman S, Israel G, Herts B, et al. Management of the incidental renal mass. Radiology. 2008;249:16-31.
3. Curry NS, Bissada NK. Radiologic evaluation of small and indeterminate renal masses. Urol Clin North Am. 1997;24:493-505.
4. Bosniak MA. The current radiological approach to renal cysts. Radiology. 1986;158:1-10.
5. Harisinghani M, Maher M, Gervais D, et al. Incidence of malignancy in complex cystic renal masses (Bosniak category III): should imaging guided biopsy precede surgery? AJR Am J Roentgenol. 2003;180:755-758.
6. Remzi M, Ozsoy M, Klingler HC. Are small renal tumors harmless? Analysis of histopathological features according to tumors less than 4 cm in diameter. J Urol. 2006;176:896-899.
7. Jonisch AI, Rubinowitz A, Mutalik P, et al. Can high attenuation renal cysts be differentiated from renal cell carcinoma at unenhanced computed tomography? Radiology. 2007;243:445-450.
8. Israel GM, Bosniak MA. Follow-up CT of moderately complex cystic lesions of the kidney. AJR Am J Roentgenol. 2003;181: 627-633.
9. Bosniak MA, Megibow AJ, Hulnick DH, et al. CT diagnosis of renal angiomyolipoma: the importance of detecting small amounts of fat. AJR Am J Roentengol. 1988;151:497-501.
10. Mitnick JS, Bosniak MA, Rothberg M, et al. Metastatic neoplasm to the kidney studied by computed tomography and sonogram. J Comput Assist Tomogr. 1985;9:43-49.
11. Rybicki FJ, Shu KM, Cibas ES, et al. Percutaneous biopsy of renal masses: sensitivity and negative predictive value stratified by clinical setting and size of masses. AJR Am J Roentgenol. 2003;180:1281-1287.
12. Frank I, Blure MI, Cheville JC, et al. Solid renal tumors: an analysis of pathological features related to tumor size. J Urol. 2003;170:2217-2220.
13. Hollingsworth JM, Miller DC, Daignault S, et al. Rising incidence of small renal masses: a need to reassess treatment effect. J Natl Cancer Inst. 2006;98:1331-1334.
14. Geelhoed GW, Spiegel CT. “Incidental” adrenal cyst: a correctable lesion possibly associated with hypertension. South Med J. 1981;74:626-630.
15. Davenport C, Liew A, Doherty B, et al. The prevalence of adrenal incidentaloma in routine clinical practice. Endocrine. 2011;40: 80-83.
16. Cook DM, Loriaux LD. The incidental adrenal mass. Am J Med. 1996;101:88 94.
17. Mansmann G, Lau J, Balk E, et al. The clinically inapparent adrenal mass: update in diagnosis and management. Endocr Rev. 2004;25:309-340.
18. Androulakis II, Kaltsas G, Piatitis G, et al. The clinical significance of adrenal incidentalomas. Eur J Clin Invest. 2011;41: 552-560.
19. Lee JE, Evans DB, Hickey RC, et al. Unknown primary cancer presenting as an adrenal mass: frequency and implications for diagnostic evaluation of adrenal incidentalomas. Surgery. 1998;124:1115-1122.
20. Aron D, Terzolo M, Cawood TJ. Adrenal incidentalomas. Best Pract Res Clin Endocrinol Metab. 2012;26:69-82.
21. ACR appropriateness criteria: incidentally discovered adrenal mass. American College of Radiology. Available at: http://www.acr.org/~/media/ACR/Documents/AppCriteria/Diagnostic/IncidentallyDiscoveredAdrenalMass.pdf. Accessed November 20, 2012.
22. Song JH, Mayo-Smith WW. Incidentally discovered adrenal mass. Radiol Clin North Am. 2011;49:361-368.
23. Korobkin M, Brodeur FJ, Francis IR, et al. CT time-attenuation washout curves of adrenal adenomas and nonadenomas. AJR Am J Roentgenol. 1998;170:747-752.
24. Boland GW, Lee MJ, Gazelle GS, et al. Characterization of adrenal masses using unenhanced CT: an analysis of the CT literature. AJR Am J Roentgenol. 1998;171:201-204.
25. Park BK, Kim CK, Kim B, et al. Chemical shift MR imaging of hyperattenuating (>10 HU) adrenal masses: does it still have a role? Radiology. 2004;231:711-716.
26. Boland GW, Blake MA, Holakere NS, et al. PET/CT for the characterization of adrenal masses in patients with cancer: qualitative vs quantitative accuracy in 150 consecutive patients. AJR Am J Roentgenol. 2009;192:956-962.
27. Paulsen SD, Nghiem HV, Korobkin M, et al. Changing role of imaging- guided percutaneous biopsy of adrenal masses: evaluation of 50 adrenal biopsies. AJR Am J Roentgenol. 2004;182:1033-1037
28. Grumbach MM, Biller BMK, Braunstein GD, et al. Management of the clinically inapparent adrenal mass (“incidentalomas”). Ann Intern Med. 2003;138:424-429.
29. Zeiger MA, Thompson GB, Quan-Yang D, et al. American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons medical guidelines for the management of adrenal incidentalomas. Endocr Pract. 2009;15(suppl 1):1-20.
30. Young WF. The incidentally discovered adrenal mass. N Engl J Med. 2007; 356:601-610.
31. Deutschbein T, Unger N, Hinrichs J, et al. Late-night and lowdose dexamethasone-suppressed cortisol in saliva and serum for the diagnosis of cortisol-secreting adrenal adenomas. Eur J Endocrinol. 2009;161:747-753.
32. Bernini G, Moretti A, Gianfranco A, et al. Primary aldosteronism in normokalemic patients with adrenal incidentalomas. Eur J Endocrinol. 2002;146:523-529.
33. Montori VM, Young WF Jr. Use of plasma aldosterone concentration-to-plasma renin activity ratio as a screening test for primary aldosteronism: a systematic review of the literature. Endocrinol Metab Clin North Am. 2002;31:619-632.
34. Motta-Ramirez GA, Remer EM, Herts BR, et al. Comparison of CT findings in symptomatic and incidentally discovered pheochromocytomas. AJR Am J Roentgenol. 2005;185:684-688.
35. Pacak K, Eisenhofer G, Grossman A. The incidentally discovered adrenal mass. N Engl J Med. 2007;356:2005.
1. Berland LL, Silverman SG, Gore RM, et al. Managing incidental findings on abdominal CT: white paper of the ACR incidental findings committee. J Am Coll Radiol. 2010;7:754-773.
2. Silverman S, Israel G, Herts B, et al. Management of the incidental renal mass. Radiology. 2008;249:16-31.
3. Curry NS, Bissada NK. Radiologic evaluation of small and indeterminate renal masses. Urol Clin North Am. 1997;24:493-505.
4. Bosniak MA. The current radiological approach to renal cysts. Radiology. 1986;158:1-10.
5. Harisinghani M, Maher M, Gervais D, et al. Incidence of malignancy in complex cystic renal masses (Bosniak category III): should imaging guided biopsy precede surgery? AJR Am J Roentgenol. 2003;180:755-758.
6. Remzi M, Ozsoy M, Klingler HC. Are small renal tumors harmless? Analysis of histopathological features according to tumors less than 4 cm in diameter. J Urol. 2006;176:896-899.
7. Jonisch AI, Rubinowitz A, Mutalik P, et al. Can high attenuation renal cysts be differentiated from renal cell carcinoma at unenhanced computed tomography? Radiology. 2007;243:445-450.
8. Israel GM, Bosniak MA. Follow-up CT of moderately complex cystic lesions of the kidney. AJR Am J Roentgenol. 2003;181: 627-633.
9. Bosniak MA, Megibow AJ, Hulnick DH, et al. CT diagnosis of renal angiomyolipoma: the importance of detecting small amounts of fat. AJR Am J Roentengol. 1988;151:497-501.
10. Mitnick JS, Bosniak MA, Rothberg M, et al. Metastatic neoplasm to the kidney studied by computed tomography and sonogram. J Comput Assist Tomogr. 1985;9:43-49.
11. Rybicki FJ, Shu KM, Cibas ES, et al. Percutaneous biopsy of renal masses: sensitivity and negative predictive value stratified by clinical setting and size of masses. AJR Am J Roentgenol. 2003;180:1281-1287.
12. Frank I, Blure MI, Cheville JC, et al. Solid renal tumors: an analysis of pathological features related to tumor size. J Urol. 2003;170:2217-2220.
13. Hollingsworth JM, Miller DC, Daignault S, et al. Rising incidence of small renal masses: a need to reassess treatment effect. J Natl Cancer Inst. 2006;98:1331-1334.
14. Geelhoed GW, Spiegel CT. “Incidental” adrenal cyst: a correctable lesion possibly associated with hypertension. South Med J. 1981;74:626-630.
15. Davenport C, Liew A, Doherty B, et al. The prevalence of adrenal incidentaloma in routine clinical practice. Endocrine. 2011;40: 80-83.
16. Cook DM, Loriaux LD. The incidental adrenal mass. Am J Med. 1996;101:88 94.
17. Mansmann G, Lau J, Balk E, et al. The clinically inapparent adrenal mass: update in diagnosis and management. Endocr Rev. 2004;25:309-340.
18. Androulakis II, Kaltsas G, Piatitis G, et al. The clinical significance of adrenal incidentalomas. Eur J Clin Invest. 2011;41: 552-560.
19. Lee JE, Evans DB, Hickey RC, et al. Unknown primary cancer presenting as an adrenal mass: frequency and implications for diagnostic evaluation of adrenal incidentalomas. Surgery. 1998;124:1115-1122.
20. Aron D, Terzolo M, Cawood TJ. Adrenal incidentalomas. Best Pract Res Clin Endocrinol Metab. 2012;26:69-82.
21. ACR appropriateness criteria: incidentally discovered adrenal mass. American College of Radiology. Available at: http://www.acr.org/~/media/ACR/Documents/AppCriteria/Diagnostic/IncidentallyDiscoveredAdrenalMass.pdf. Accessed November 20, 2012.
22. Song JH, Mayo-Smith WW. Incidentally discovered adrenal mass. Radiol Clin North Am. 2011;49:361-368.
23. Korobkin M, Brodeur FJ, Francis IR, et al. CT time-attenuation washout curves of adrenal adenomas and nonadenomas. AJR Am J Roentgenol. 1998;170:747-752.
24. Boland GW, Lee MJ, Gazelle GS, et al. Characterization of adrenal masses using unenhanced CT: an analysis of the CT literature. AJR Am J Roentgenol. 1998;171:201-204.
25. Park BK, Kim CK, Kim B, et al. Chemical shift MR imaging of hyperattenuating (>10 HU) adrenal masses: does it still have a role? Radiology. 2004;231:711-716.
26. Boland GW, Blake MA, Holakere NS, et al. PET/CT for the characterization of adrenal masses in patients with cancer: qualitative vs quantitative accuracy in 150 consecutive patients. AJR Am J Roentgenol. 2009;192:956-962.
27. Paulsen SD, Nghiem HV, Korobkin M, et al. Changing role of imaging- guided percutaneous biopsy of adrenal masses: evaluation of 50 adrenal biopsies. AJR Am J Roentgenol. 2004;182:1033-1037
28. Grumbach MM, Biller BMK, Braunstein GD, et al. Management of the clinically inapparent adrenal mass (“incidentalomas”). Ann Intern Med. 2003;138:424-429.
29. Zeiger MA, Thompson GB, Quan-Yang D, et al. American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons medical guidelines for the management of adrenal incidentalomas. Endocr Pract. 2009;15(suppl 1):1-20.
30. Young WF. The incidentally discovered adrenal mass. N Engl J Med. 2007; 356:601-610.
31. Deutschbein T, Unger N, Hinrichs J, et al. Late-night and lowdose dexamethasone-suppressed cortisol in saliva and serum for the diagnosis of cortisol-secreting adrenal adenomas. Eur J Endocrinol. 2009;161:747-753.
32. Bernini G, Moretti A, Gianfranco A, et al. Primary aldosteronism in normokalemic patients with adrenal incidentalomas. Eur J Endocrinol. 2002;146:523-529.
33. Montori VM, Young WF Jr. Use of plasma aldosterone concentration-to-plasma renin activity ratio as a screening test for primary aldosteronism: a systematic review of the literature. Endocrinol Metab Clin North Am. 2002;31:619-632.
34. Motta-Ramirez GA, Remer EM, Herts BR, et al. Comparison of CT findings in symptomatic and incidentally discovered pheochromocytomas. AJR Am J Roentgenol. 2005;185:684-688.
35. Pacak K, Eisenhofer G, Grossman A. The incidentally discovered adrenal mass. N Engl J Med. 2007;356:2005.
Plantar Fasciitis: How Best to Treat?
› Use plantar fascia specific stretching to decrease pain in patients with plantar fasciitis. A
› Consider recommending prefabricated orthoses, including night splints, to decrease pain. A
› Consider using extracorporeal shock wave therapy for plantar fascial pain. A
Strength of recommendation (SOR)
A. Good-quality patient-oriented evidence
B. Inconsistent or limited-quality patient-oriented evidence
C. Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE A 43-year-old obese woman seeks advice for left heel pain she has had for 2 months. Before the onset of pain, her activity level had increased as part of a weight loss program. Her pain is at its worst in the morning, with her first few steps; it decreases with continued walking and intensifies again after being on her feet all day. There is no history of trauma, and she reports no paresthesias or radiation of the pain. Her medical history is otherwise unremarkable. She has used ibuprofen sparingly, with limited relief.
If you were this patient’s physician, how would you proceed with her care?
Plantar fasciitis (PF) is a common cause of heel pain that affects up to 10% of the US population and accounts for approximately 600,000 outpatient visits annually.1 The plantar fascia is a dense, fibrous membrane spanning the length of the foot. It originates at the medial calcaneal tubercle, attaches to the phalanges, and provides stability and arch support to the foot. The etiology of PF is unknown, but predisposing factors include overtraining, obesity, pes planus, decreased ankle dorsiflexion, and inappropriate footwear.2 Limited dorsiflexion due to tightness of the Achilles tendon strains the plantar fascia and can lead to PF. Histology shows minimal inflammatory changes, and some experts advocate the term plantar fasciosis to counter the misperception that it is primarily an inflammatory condition.3
A patient’s history and physical exam findings are the basis for confirming or dismissing a diagnosis of PF. Radiologic studies, used judiciously, can rule out important alternative diagnoses that should not be overlooked. Multiple treatment options range from conservative to surgical interventions, although studies of the effectiveness of each modality have had conflicting results. Clinical practice guidelines generally advocate a stepwise approach to treatment.
Diagnosis
The differential diagnosis of PF (TABLE) includes significant disorders such as calcaneal stress fracture, entrapment neuropathies (eg, tarsal tunnel syndrome), calcaneal tumor, Paget’s disease, and systemic arthritidies.4,5
What to look for in the history and physical exam
Severe heel pain upon initial weight bearing in the morning or after prolonged periods of inactivity is pathognomonic for PF.2 Initially the pain presents diffusely, but over time it localizes to the area of the medial calcaneal tubercle. Pain typically subsides with activity but may return with prolonged weight bearing, as it did with the patient in the opening case.
Test range of motion of the foot and ankle. Although this is not needed for diagnosing PF, some patients will exhibit limited ankle dorsiflexion, a predisposing factor for PF.4,6 Look for heel pad swelling, inflammation, or atrophy, and palpate the heel, plantar fascia, and calcaneal tubercle. Lastly, evaluate for gait abnormalities and the presence of sensory deficits or hypesthesias.4
The most common exam finding in PF is pain at the medial calcaneal tubercle, which may be exacerbated with passive ankle dorsiflexion or first digit extension.2,4 If paresthesias occur with percussion inferior to the medial malleolus, suspect possible nerve entrapment or tarsal tunnel syndrome. Tenderness with heel compression (squeeze test) may indicate a calcaneal fracture or apophysitis.
Imaging is useful to rule out alternative disorders
Radiologic studies generally do not contribute to the diagnosis or management of PF, but they can assist in ruling out alternative causes of heel pain or in reevaluation if symptoms of PF persist after 3 to 6 months of treatment.
Plain films lack the sensitivity to detect plantar fasciitis. While a plantar calcaneal spur is often seen on radiography, it does not confirm the diagnosis, correlate with severity of symptoms, or predict prognosis.4 Despite this deficiency, plain radiography remains the initial choice of imaging modalities, particularly to rule out other conditions.
Ultrasound accurately diagnoses plantar fasciitis. Plantar fascia thickness of more than 4.0 mm is diagnostic of PF.7 Additionally, a decrease in plantar fascia thickness correlates with a decrease in pain levels, and thus ultrasound can aid in monitoring treatment progress.8 If results of plain films and ultrasound are inconclusive and clinical concern for an alternative diagnosis warrants additional expense, consider arranging for magnetic resonance imaging.9
Noninvasive treatments
Conservative therapies remain the preferred approach to treating PF, successfully managing 85% to 90% of cases.10,11 A 2010 clinical practice guideline from the American College of Foot and Ankle Surgeons recommends conservative treatments such as nonsteroidal inflammatory drugs (NSAIDs), stretching, and prefabricated orthotics for the initial management of plantar heel pain.4 Emphasize to patients that it may take 6 to 12 months for symptoms to resolve.4
Stretching and trigger-point manual therapy are effective
The traditional primary treatment modality for PF has been early initiation of an Achilles-soleus (heel-cord) muscle–stretching program. However, studies have shown that plantar fascia–specific stretching (PFSS) (FIGURE) significantly diminishes or eliminates heel pain when compared with traditional stretching movements, and is useful in treating chronic recalcitrant heel pain.12,13 PFSS has also yielded results superior to low-dose shock wave therapy.14
In a 2011 study, adding myofascial trigger-point manual therapy to a PFSS routine improved self-reported physical function and pain vs stretching alone.15 This manual therapy technique is specialized and should be administered only by trained physical therapists. Data are limited and mixed regarding the effectiveness of deep tissue massage, iontophoresis, or eccentric stretching of the plantar fascia to alleviate plantar fascial pain. Support for therapies such as rest, ice, heat, and massage has largely been anecdotal.
NSAIDs for PF lack good evidence
Nonsteroidal anti-inflammatory drugs (NSAIDs) are often prescribed to treat PF, despite a lack of evidence supporting their use. A small randomized, placebo-controlled double-blind study established a trend toward improvement in pain and disability scores with the use of NSAIDs. However, no statistically significant difference was noted in the measures between the NSAID and placebo groups at 1, 2, and 6 months.16 We found no studies that demonstrate a significant reduction in pain or improvement in function with the use of NSAIDs alone.
Although NSAIDs carry their own risks, they may work for some patients. And studies showing a lack of significant pain reduction may have been underpowered. If patients are willing to accept the risks of NSAID use, it would be reasonable to prescribe a therapeutic trial.
Orthotics and night splints can help, depending on comfort and compliance
Foot orthotics help prevent overpronation and attenuate tensile forces on the plantar fascia. A 2009 meta-analysis confirmed that both prefabricated and custom-made foot orthotics can decrease pain.17 One prospective study showed that 95% of patients had improvement in PF symptoms after 8 weeks of treatment with prefabricated orthotics.18 A Cochrane review found no difference in pain reduction between custom and prefabricated foot orthotics.19 A recent study demonstrated that rocker sole shoes—a type of therapeutic footwear with a more rounded outsole contour—combined with custom orthotics significantly reduced pain during walking compared with either modality alone.20 More research needs to be conducted into the use of rocker sole shoes before recommending them to PF patients.
Night splints help keep the foot and ankle in a neutral position, or slightly dorsiflexed, while patients sleep. Several studies have shown a reduction in pain with the use of night splints alone.17,21,22 Patient comfort and compliance tend to be the limiting factors in their use. Anterior splints are better tolerated than posterior splints.23
Shock wave therapy has better long-term results than steroid injections
Shock waves used to treat PF are thought to invoke extracellular responses that cause neovascularization and induce tissue repair and regeneration. A 2012 review article concluded that most research confirms that extracorporeal shock wave therapy (ESWT) reduces PF pain and improves function in 34% to 88% of cases.24 ESWT is comparable to surgical plantar fasciotomy without the operative risks, and yields better long-term effects in recalcitrant PF compared with corticosteroid injections (CSI).24 Many studies are underway to validate the effectiveness of ESWT. Currently, expense or lack of availability limits its use in some communities.
Invasive treatments
Corticosteroid injections may be used for more than just refractory pain
CSI have historically been reserved for recalcitrant heel pain. However, one systematic review cites evidence in support of CSI for the short-term management of plantar fascia pain.25 Compared with placebo, CSI reduces pain at both 6 and 12 weeks and decreases plantar fascia thickness.26 Additionally, the American College of Foot and Ankle Surgeons lists CSI as an acceptable first-line treatment for PF.4
The most common complication of CSI is postinjection pain. Other complications, such as fat pad atrophy, rarely occur.27 While the evidence is limited, CSI may be part of an initial approach to treating PF in addition to heel-cord or plantar fascia-specific stretching, particularly for patients who desire an expedited return to normal activity.
Platelet-rich plasma therapy holds promise
Platelet-rich plasma (PRP) has been gaining popularity as a treatment for PF pain. PRP is a component of whole blood that is centrifuged to a concentrated state, treated with an activating agent, and injected into the affected area. Theoretically, injected PRP increases the release of reparative growth factors, enhancing the healing process.28 PRP has been shown to be as effective in reducing pain scores as CSI at 3 weeks and 6 months.29 PRP also decreases plantar fascia thickness and improves pain scores and functional ability.30
To date, no trials have compared PRP with placebo injections. Postprocedural pain is the most common risk with PRP. While limited evidence exists, PRP seems to be a relatively safe and effective therapeutic alternative for treating chronic PF.
Surgery only when conservative measures fail
Reserve surgery for those who have not responded adequately after 6 to 12 months of conservative therapy.5 Endoscopic plantar fascia release is superior to traditional open surgery.31 Heel spur resection is no longer routinely practiced. Patients undergoing surgery should expect a return to normal activity in approximately 2 to 3 months, and up to 35% of patients may continue to have symptoms after surgical intervention.2,31
Treatment options in perspective
Treat conservatively at first. Stretching the plantar fascia and heel cord, using prefabricated orthotics, and wearing night splints are backed by firm clinical evidence of benefit. Acute treatment of PF may also include CSI, especially for patients who are athletic or otherwise active and wish to return to full function as soon as possible, and are willing to accept the risks associated with CSI.
ESWT improves pain and function scores and may also relieve pain in patients with recalcitrant PF pain. PRP has limited but promising evidence for patients with chronic PF pain. Surgical intervention remains the last line of therapy and is not always effective at reducing pain.
CASE You prescribe a conservative treatment program of plantar fascia–specific stretches and prefabricated orthoses for the patient in the opening scenario. At one month, her pain drops by 30%. At 6 months, her pain disappears, and she resumes a daily aerobic exercise program to assist in weight loss.
CORRESPONDENCE
Carlton J. Covey, MD, Nellis Family Medicine Residency, 99MDOS/SGOF, 4700 Las Vegas Boulevard N, Las Vegas, NV 89191;
[email protected]
1. Riddle DL, Schappert SM. Volume of ambulatory care visits and patterns of care for patients diagnosed with plantar fasciitis: a national study of medical doctors. Foot Ankle Int. 2004;25: 303-310.
2. Glazer JL. An approach to the diagnosis and treatment of plantar fasciitis. Phys Sportsmed. 2009;37:74-79.
3. Lemont H, Ammirati KM, Usen N. Plantar fasciitis: a degenerative process (fasciosis) without inflammation. J Am Podiatr Med Assoc. 2003;93:234-237.
4. Thomas JL, Christensen JC, Kravitz SR, et al. The diagnosis and treatment of heal pain: a clinical practice guideline – revision 2010. J Foot Ankle Surg. 2010;49(suppl):S1-S19.
5. Neufeld SK, Cerrato R. Plantar fasciitis: evaluation and treatment. J Am Acad Orthop Surg. 2008;16:338-346.
6. Singh D, Angel J, Bentley G, et al. Fortnightly review: plantar fasciitis. BMJ. 1997;315:172-175.
7. McMillan AM, Landorf KB, Barrett JT, et al. Diagnostic imaging for chronic plantar heel pain: a systematic review and metaanalysis. J Foot Ankle Res. 2009;2:32.
8. Mahowald S, Legge BS, Grady JF. The correlation between plantar fascia thickness and symptoms of plantar fasciitis. J Am Podiatr Med Assoc. 2011;101:385-389.
9. American College of Radiology. ACR appropriateness criteria. Chronic foot pain. Available at: http://www.acr.org/~/media/ACR/Documents/AppCriteria/Diagnostic/ChronicFootPain.pdf. Accessed November 11, 2012.
10. Gill LH. Plantar fasciitis: diagnosis and conservative treatment. J Am Acad Orthop Surg. 1997;5:109-117.
11. Martin RL, Irrgang JJ, Conti SF. Outcome study of subjects with insertional plantar fasciitis. Foot Ankle Int. 1998;19:803-811.
12. DiGiovanni BF, Nawoczenski DA, Malay DP, et al. Plantar fasciaspecific stretching exercise improves outcomes in patients with chronic plantar fasciitis: a prospective clinic trial with two-year follow up. J Bone Joint Surg Am. 2006;88:1775-1781.
13. Sweeting D, Parish B, Hooper L, et al. The effectiveness of manual stretching in the treatment of plantar heel pain: a systemic review. J Foot Ankle Res. 2011;4:1-13.
14. Rompe JD, Cacchio A, Lowell W, et al. Plantar fascia-specific stretching versus radial shock-wave therapy as initial treatment of plantar fasciopathy. J Bone Joint Surg Am. 2010;92:2514-2522.
15. Renan-Ordine R, Alburquerque-Sendin F, Rodriques De Souza DP, et al. Effectiveness of myofascial trigger point manual therapy combined with a self stretching protocol for the management of plantar heel pain: a randomized controlled trial. J Orthop Sports Phys Ther. 2011;41:43-50.
16. Donley BG, Moore T, Sferra J, et al. The efficacy of oral nonsteroidal anti-inflammatory medication (NSAID) in the treatment of plantar fasciitis: a randomized, prospective, placebo-controlled study. Foot Ankle Int. 2007;28:20-23.
17. Lee SY, McKeon P, Hertel J. Does the use of orthoses improve selfreported pain and function measures in patients with plantar fasciitis? A meta-analysis. Phys Ther Sport. 2009;10:12-18.
18. Pfeffer G, Bacchetti P, Deland J, et al. Comparison of custom and prefabricated orthoses in the initial treatment of proximal plantar fasciitis. Foot Ankle Int. 1999;20:214-221.
19. Hawke F, Burns J, Radford JA, et al. Custom-made foot orthoses for the treatment of foot pain. Cochrane Database Syst Rev. 2008;(3):CD006801.
20. Fong DT, Pang KY, Chung MM, et al. Evaluation of combined prescription of rocker sole shoes and custom-made foot orthoses for the treatment of plantar fasciitis. Clin Biomech. 2012;27: 1072-1077.
21. Berlet GC, Anderson RB, Davis H. A prospective trial of night splinting in the treatment of recalcitrant plantar fasciitis: the Ankle Dorsiflexion Dynasplint. Orthopedics. 2002;25: 1273-1275.
22. Roos E, Engstrom M, Soderberg B. Foot orthoses for the treatment of plantar fasciitis. Foot Ankle Int. 2006;27:606-611.
23. Goff JD, Crawford R. Diagnosis and treatment of plantar fasciitis. Am Fam Physician. 2011;84:676-682.
24. Wang CJ. Extracorporeal shockwave therapy in musculoskeletal disorders. J Orthop Surg Res. 2012;7:11.
25. Landorf KB, Menz HB. Plantar heel pain and fasciitis. Clin Evid (Online). 2008;2008:1111.
26. Ball EM, McKeeman HM, Patterson C, et al. Steroid injection for inferior heel pain: a randomized controlled trial. Ann Rheum Dis. 2013;72:996-1002.
27. Uden H, Boesch E, Kumar S. Plantar fasciitis – to jab or support? A systematic review of the current best evidence. J Multidiscip Healthc. 2011;4:155-164.
28. Shetty VD. Platelet-rich plasma: a ‘feeling’ and ‘hope’ ailing athletes. Br J Sports Med. 2010;44(suppl 1):i1-i82.
29. Aksahin E, Dogruyol D, Yüksel HY, et al. The comparison of the effect of corticosteroids and platelet-rich plasma (PRP) for the treatment of plantar fasciitis. Arch Orthop Trauma Surg. 2012;132:781-785.
30. Ragab EM, Othman AM. Platelets rich plasma for treatment of chronic plantar fasciitis. Arch Orthop Trauma Surg. 2012;132:1065-1070.
31. Saxena A. Uniportal endoscopic plantar fasciotomy: a prospective study on athletic patients. Foot Ankle Int. 2004;25:882-889.
› Use plantar fascia specific stretching to decrease pain in patients with plantar fasciitis. A
› Consider recommending prefabricated orthoses, including night splints, to decrease pain. A
› Consider using extracorporeal shock wave therapy for plantar fascial pain. A
Strength of recommendation (SOR)
A. Good-quality patient-oriented evidence
B. Inconsistent or limited-quality patient-oriented evidence
C. Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE A 43-year-old obese woman seeks advice for left heel pain she has had for 2 months. Before the onset of pain, her activity level had increased as part of a weight loss program. Her pain is at its worst in the morning, with her first few steps; it decreases with continued walking and intensifies again after being on her feet all day. There is no history of trauma, and she reports no paresthesias or radiation of the pain. Her medical history is otherwise unremarkable. She has used ibuprofen sparingly, with limited relief.
If you were this patient’s physician, how would you proceed with her care?
Plantar fasciitis (PF) is a common cause of heel pain that affects up to 10% of the US population and accounts for approximately 600,000 outpatient visits annually.1 The plantar fascia is a dense, fibrous membrane spanning the length of the foot. It originates at the medial calcaneal tubercle, attaches to the phalanges, and provides stability and arch support to the foot. The etiology of PF is unknown, but predisposing factors include overtraining, obesity, pes planus, decreased ankle dorsiflexion, and inappropriate footwear.2 Limited dorsiflexion due to tightness of the Achilles tendon strains the plantar fascia and can lead to PF. Histology shows minimal inflammatory changes, and some experts advocate the term plantar fasciosis to counter the misperception that it is primarily an inflammatory condition.3
A patient’s history and physical exam findings are the basis for confirming or dismissing a diagnosis of PF. Radiologic studies, used judiciously, can rule out important alternative diagnoses that should not be overlooked. Multiple treatment options range from conservative to surgical interventions, although studies of the effectiveness of each modality have had conflicting results. Clinical practice guidelines generally advocate a stepwise approach to treatment.
Diagnosis
The differential diagnosis of PF (TABLE) includes significant disorders such as calcaneal stress fracture, entrapment neuropathies (eg, tarsal tunnel syndrome), calcaneal tumor, Paget’s disease, and systemic arthritidies.4,5
What to look for in the history and physical exam
Severe heel pain upon initial weight bearing in the morning or after prolonged periods of inactivity is pathognomonic for PF.2 Initially the pain presents diffusely, but over time it localizes to the area of the medial calcaneal tubercle. Pain typically subsides with activity but may return with prolonged weight bearing, as it did with the patient in the opening case.
Test range of motion of the foot and ankle. Although this is not needed for diagnosing PF, some patients will exhibit limited ankle dorsiflexion, a predisposing factor for PF.4,6 Look for heel pad swelling, inflammation, or atrophy, and palpate the heel, plantar fascia, and calcaneal tubercle. Lastly, evaluate for gait abnormalities and the presence of sensory deficits or hypesthesias.4
The most common exam finding in PF is pain at the medial calcaneal tubercle, which may be exacerbated with passive ankle dorsiflexion or first digit extension.2,4 If paresthesias occur with percussion inferior to the medial malleolus, suspect possible nerve entrapment or tarsal tunnel syndrome. Tenderness with heel compression (squeeze test) may indicate a calcaneal fracture or apophysitis.
Imaging is useful to rule out alternative disorders
Radiologic studies generally do not contribute to the diagnosis or management of PF, but they can assist in ruling out alternative causes of heel pain or in reevaluation if symptoms of PF persist after 3 to 6 months of treatment.
Plain films lack the sensitivity to detect plantar fasciitis. While a plantar calcaneal spur is often seen on radiography, it does not confirm the diagnosis, correlate with severity of symptoms, or predict prognosis.4 Despite this deficiency, plain radiography remains the initial choice of imaging modalities, particularly to rule out other conditions.
Ultrasound accurately diagnoses plantar fasciitis. Plantar fascia thickness of more than 4.0 mm is diagnostic of PF.7 Additionally, a decrease in plantar fascia thickness correlates with a decrease in pain levels, and thus ultrasound can aid in monitoring treatment progress.8 If results of plain films and ultrasound are inconclusive and clinical concern for an alternative diagnosis warrants additional expense, consider arranging for magnetic resonance imaging.9
Noninvasive treatments
Conservative therapies remain the preferred approach to treating PF, successfully managing 85% to 90% of cases.10,11 A 2010 clinical practice guideline from the American College of Foot and Ankle Surgeons recommends conservative treatments such as nonsteroidal inflammatory drugs (NSAIDs), stretching, and prefabricated orthotics for the initial management of plantar heel pain.4 Emphasize to patients that it may take 6 to 12 months for symptoms to resolve.4
Stretching and trigger-point manual therapy are effective
The traditional primary treatment modality for PF has been early initiation of an Achilles-soleus (heel-cord) muscle–stretching program. However, studies have shown that plantar fascia–specific stretching (PFSS) (FIGURE) significantly diminishes or eliminates heel pain when compared with traditional stretching movements, and is useful in treating chronic recalcitrant heel pain.12,13 PFSS has also yielded results superior to low-dose shock wave therapy.14
In a 2011 study, adding myofascial trigger-point manual therapy to a PFSS routine improved self-reported physical function and pain vs stretching alone.15 This manual therapy technique is specialized and should be administered only by trained physical therapists. Data are limited and mixed regarding the effectiveness of deep tissue massage, iontophoresis, or eccentric stretching of the plantar fascia to alleviate plantar fascial pain. Support for therapies such as rest, ice, heat, and massage has largely been anecdotal.
NSAIDs for PF lack good evidence
Nonsteroidal anti-inflammatory drugs (NSAIDs) are often prescribed to treat PF, despite a lack of evidence supporting their use. A small randomized, placebo-controlled double-blind study established a trend toward improvement in pain and disability scores with the use of NSAIDs. However, no statistically significant difference was noted in the measures between the NSAID and placebo groups at 1, 2, and 6 months.16 We found no studies that demonstrate a significant reduction in pain or improvement in function with the use of NSAIDs alone.
Although NSAIDs carry their own risks, they may work for some patients. And studies showing a lack of significant pain reduction may have been underpowered. If patients are willing to accept the risks of NSAID use, it would be reasonable to prescribe a therapeutic trial.
Orthotics and night splints can help, depending on comfort and compliance
Foot orthotics help prevent overpronation and attenuate tensile forces on the plantar fascia. A 2009 meta-analysis confirmed that both prefabricated and custom-made foot orthotics can decrease pain.17 One prospective study showed that 95% of patients had improvement in PF symptoms after 8 weeks of treatment with prefabricated orthotics.18 A Cochrane review found no difference in pain reduction between custom and prefabricated foot orthotics.19 A recent study demonstrated that rocker sole shoes—a type of therapeutic footwear with a more rounded outsole contour—combined with custom orthotics significantly reduced pain during walking compared with either modality alone.20 More research needs to be conducted into the use of rocker sole shoes before recommending them to PF patients.
Night splints help keep the foot and ankle in a neutral position, or slightly dorsiflexed, while patients sleep. Several studies have shown a reduction in pain with the use of night splints alone.17,21,22 Patient comfort and compliance tend to be the limiting factors in their use. Anterior splints are better tolerated than posterior splints.23
Shock wave therapy has better long-term results than steroid injections
Shock waves used to treat PF are thought to invoke extracellular responses that cause neovascularization and induce tissue repair and regeneration. A 2012 review article concluded that most research confirms that extracorporeal shock wave therapy (ESWT) reduces PF pain and improves function in 34% to 88% of cases.24 ESWT is comparable to surgical plantar fasciotomy without the operative risks, and yields better long-term effects in recalcitrant PF compared with corticosteroid injections (CSI).24 Many studies are underway to validate the effectiveness of ESWT. Currently, expense or lack of availability limits its use in some communities.
Invasive treatments
Corticosteroid injections may be used for more than just refractory pain
CSI have historically been reserved for recalcitrant heel pain. However, one systematic review cites evidence in support of CSI for the short-term management of plantar fascia pain.25 Compared with placebo, CSI reduces pain at both 6 and 12 weeks and decreases plantar fascia thickness.26 Additionally, the American College of Foot and Ankle Surgeons lists CSI as an acceptable first-line treatment for PF.4
The most common complication of CSI is postinjection pain. Other complications, such as fat pad atrophy, rarely occur.27 While the evidence is limited, CSI may be part of an initial approach to treating PF in addition to heel-cord or plantar fascia-specific stretching, particularly for patients who desire an expedited return to normal activity.
Platelet-rich plasma therapy holds promise
Platelet-rich plasma (PRP) has been gaining popularity as a treatment for PF pain. PRP is a component of whole blood that is centrifuged to a concentrated state, treated with an activating agent, and injected into the affected area. Theoretically, injected PRP increases the release of reparative growth factors, enhancing the healing process.28 PRP has been shown to be as effective in reducing pain scores as CSI at 3 weeks and 6 months.29 PRP also decreases plantar fascia thickness and improves pain scores and functional ability.30
To date, no trials have compared PRP with placebo injections. Postprocedural pain is the most common risk with PRP. While limited evidence exists, PRP seems to be a relatively safe and effective therapeutic alternative for treating chronic PF.
Surgery only when conservative measures fail
Reserve surgery for those who have not responded adequately after 6 to 12 months of conservative therapy.5 Endoscopic plantar fascia release is superior to traditional open surgery.31 Heel spur resection is no longer routinely practiced. Patients undergoing surgery should expect a return to normal activity in approximately 2 to 3 months, and up to 35% of patients may continue to have symptoms after surgical intervention.2,31
Treatment options in perspective
Treat conservatively at first. Stretching the plantar fascia and heel cord, using prefabricated orthotics, and wearing night splints are backed by firm clinical evidence of benefit. Acute treatment of PF may also include CSI, especially for patients who are athletic or otherwise active and wish to return to full function as soon as possible, and are willing to accept the risks associated with CSI.
ESWT improves pain and function scores and may also relieve pain in patients with recalcitrant PF pain. PRP has limited but promising evidence for patients with chronic PF pain. Surgical intervention remains the last line of therapy and is not always effective at reducing pain.
CASE You prescribe a conservative treatment program of plantar fascia–specific stretches and prefabricated orthoses for the patient in the opening scenario. At one month, her pain drops by 30%. At 6 months, her pain disappears, and she resumes a daily aerobic exercise program to assist in weight loss.
CORRESPONDENCE
Carlton J. Covey, MD, Nellis Family Medicine Residency, 99MDOS/SGOF, 4700 Las Vegas Boulevard N, Las Vegas, NV 89191;
[email protected]
› Use plantar fascia specific stretching to decrease pain in patients with plantar fasciitis. A
› Consider recommending prefabricated orthoses, including night splints, to decrease pain. A
› Consider using extracorporeal shock wave therapy for plantar fascial pain. A
Strength of recommendation (SOR)
A. Good-quality patient-oriented evidence
B. Inconsistent or limited-quality patient-oriented evidence
C. Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE A 43-year-old obese woman seeks advice for left heel pain she has had for 2 months. Before the onset of pain, her activity level had increased as part of a weight loss program. Her pain is at its worst in the morning, with her first few steps; it decreases with continued walking and intensifies again after being on her feet all day. There is no history of trauma, and she reports no paresthesias or radiation of the pain. Her medical history is otherwise unremarkable. She has used ibuprofen sparingly, with limited relief.
If you were this patient’s physician, how would you proceed with her care?
Plantar fasciitis (PF) is a common cause of heel pain that affects up to 10% of the US population and accounts for approximately 600,000 outpatient visits annually.1 The plantar fascia is a dense, fibrous membrane spanning the length of the foot. It originates at the medial calcaneal tubercle, attaches to the phalanges, and provides stability and arch support to the foot. The etiology of PF is unknown, but predisposing factors include overtraining, obesity, pes planus, decreased ankle dorsiflexion, and inappropriate footwear.2 Limited dorsiflexion due to tightness of the Achilles tendon strains the plantar fascia and can lead to PF. Histology shows minimal inflammatory changes, and some experts advocate the term plantar fasciosis to counter the misperception that it is primarily an inflammatory condition.3
A patient’s history and physical exam findings are the basis for confirming or dismissing a diagnosis of PF. Radiologic studies, used judiciously, can rule out important alternative diagnoses that should not be overlooked. Multiple treatment options range from conservative to surgical interventions, although studies of the effectiveness of each modality have had conflicting results. Clinical practice guidelines generally advocate a stepwise approach to treatment.
Diagnosis
The differential diagnosis of PF (TABLE) includes significant disorders such as calcaneal stress fracture, entrapment neuropathies (eg, tarsal tunnel syndrome), calcaneal tumor, Paget’s disease, and systemic arthritidies.4,5
What to look for in the history and physical exam
Severe heel pain upon initial weight bearing in the morning or after prolonged periods of inactivity is pathognomonic for PF.2 Initially the pain presents diffusely, but over time it localizes to the area of the medial calcaneal tubercle. Pain typically subsides with activity but may return with prolonged weight bearing, as it did with the patient in the opening case.
Test range of motion of the foot and ankle. Although this is not needed for diagnosing PF, some patients will exhibit limited ankle dorsiflexion, a predisposing factor for PF.4,6 Look for heel pad swelling, inflammation, or atrophy, and palpate the heel, plantar fascia, and calcaneal tubercle. Lastly, evaluate for gait abnormalities and the presence of sensory deficits or hypesthesias.4
The most common exam finding in PF is pain at the medial calcaneal tubercle, which may be exacerbated with passive ankle dorsiflexion or first digit extension.2,4 If paresthesias occur with percussion inferior to the medial malleolus, suspect possible nerve entrapment or tarsal tunnel syndrome. Tenderness with heel compression (squeeze test) may indicate a calcaneal fracture or apophysitis.
Imaging is useful to rule out alternative disorders
Radiologic studies generally do not contribute to the diagnosis or management of PF, but they can assist in ruling out alternative causes of heel pain or in reevaluation if symptoms of PF persist after 3 to 6 months of treatment.
Plain films lack the sensitivity to detect plantar fasciitis. While a plantar calcaneal spur is often seen on radiography, it does not confirm the diagnosis, correlate with severity of symptoms, or predict prognosis.4 Despite this deficiency, plain radiography remains the initial choice of imaging modalities, particularly to rule out other conditions.
Ultrasound accurately diagnoses plantar fasciitis. Plantar fascia thickness of more than 4.0 mm is diagnostic of PF.7 Additionally, a decrease in plantar fascia thickness correlates with a decrease in pain levels, and thus ultrasound can aid in monitoring treatment progress.8 If results of plain films and ultrasound are inconclusive and clinical concern for an alternative diagnosis warrants additional expense, consider arranging for magnetic resonance imaging.9
Noninvasive treatments
Conservative therapies remain the preferred approach to treating PF, successfully managing 85% to 90% of cases.10,11 A 2010 clinical practice guideline from the American College of Foot and Ankle Surgeons recommends conservative treatments such as nonsteroidal inflammatory drugs (NSAIDs), stretching, and prefabricated orthotics for the initial management of plantar heel pain.4 Emphasize to patients that it may take 6 to 12 months for symptoms to resolve.4
Stretching and trigger-point manual therapy are effective
The traditional primary treatment modality for PF has been early initiation of an Achilles-soleus (heel-cord) muscle–stretching program. However, studies have shown that plantar fascia–specific stretching (PFSS) (FIGURE) significantly diminishes or eliminates heel pain when compared with traditional stretching movements, and is useful in treating chronic recalcitrant heel pain.12,13 PFSS has also yielded results superior to low-dose shock wave therapy.14
In a 2011 study, adding myofascial trigger-point manual therapy to a PFSS routine improved self-reported physical function and pain vs stretching alone.15 This manual therapy technique is specialized and should be administered only by trained physical therapists. Data are limited and mixed regarding the effectiveness of deep tissue massage, iontophoresis, or eccentric stretching of the plantar fascia to alleviate plantar fascial pain. Support for therapies such as rest, ice, heat, and massage has largely been anecdotal.
NSAIDs for PF lack good evidence
Nonsteroidal anti-inflammatory drugs (NSAIDs) are often prescribed to treat PF, despite a lack of evidence supporting their use. A small randomized, placebo-controlled double-blind study established a trend toward improvement in pain and disability scores with the use of NSAIDs. However, no statistically significant difference was noted in the measures between the NSAID and placebo groups at 1, 2, and 6 months.16 We found no studies that demonstrate a significant reduction in pain or improvement in function with the use of NSAIDs alone.
Although NSAIDs carry their own risks, they may work for some patients. And studies showing a lack of significant pain reduction may have been underpowered. If patients are willing to accept the risks of NSAID use, it would be reasonable to prescribe a therapeutic trial.
Orthotics and night splints can help, depending on comfort and compliance
Foot orthotics help prevent overpronation and attenuate tensile forces on the plantar fascia. A 2009 meta-analysis confirmed that both prefabricated and custom-made foot orthotics can decrease pain.17 One prospective study showed that 95% of patients had improvement in PF symptoms after 8 weeks of treatment with prefabricated orthotics.18 A Cochrane review found no difference in pain reduction between custom and prefabricated foot orthotics.19 A recent study demonstrated that rocker sole shoes—a type of therapeutic footwear with a more rounded outsole contour—combined with custom orthotics significantly reduced pain during walking compared with either modality alone.20 More research needs to be conducted into the use of rocker sole shoes before recommending them to PF patients.
Night splints help keep the foot and ankle in a neutral position, or slightly dorsiflexed, while patients sleep. Several studies have shown a reduction in pain with the use of night splints alone.17,21,22 Patient comfort and compliance tend to be the limiting factors in their use. Anterior splints are better tolerated than posterior splints.23
Shock wave therapy has better long-term results than steroid injections
Shock waves used to treat PF are thought to invoke extracellular responses that cause neovascularization and induce tissue repair and regeneration. A 2012 review article concluded that most research confirms that extracorporeal shock wave therapy (ESWT) reduces PF pain and improves function in 34% to 88% of cases.24 ESWT is comparable to surgical plantar fasciotomy without the operative risks, and yields better long-term effects in recalcitrant PF compared with corticosteroid injections (CSI).24 Many studies are underway to validate the effectiveness of ESWT. Currently, expense or lack of availability limits its use in some communities.
Invasive treatments
Corticosteroid injections may be used for more than just refractory pain
CSI have historically been reserved for recalcitrant heel pain. However, one systematic review cites evidence in support of CSI for the short-term management of plantar fascia pain.25 Compared with placebo, CSI reduces pain at both 6 and 12 weeks and decreases plantar fascia thickness.26 Additionally, the American College of Foot and Ankle Surgeons lists CSI as an acceptable first-line treatment for PF.4
The most common complication of CSI is postinjection pain. Other complications, such as fat pad atrophy, rarely occur.27 While the evidence is limited, CSI may be part of an initial approach to treating PF in addition to heel-cord or plantar fascia-specific stretching, particularly for patients who desire an expedited return to normal activity.
Platelet-rich plasma therapy holds promise
Platelet-rich plasma (PRP) has been gaining popularity as a treatment for PF pain. PRP is a component of whole blood that is centrifuged to a concentrated state, treated with an activating agent, and injected into the affected area. Theoretically, injected PRP increases the release of reparative growth factors, enhancing the healing process.28 PRP has been shown to be as effective in reducing pain scores as CSI at 3 weeks and 6 months.29 PRP also decreases plantar fascia thickness and improves pain scores and functional ability.30
To date, no trials have compared PRP with placebo injections. Postprocedural pain is the most common risk with PRP. While limited evidence exists, PRP seems to be a relatively safe and effective therapeutic alternative for treating chronic PF.
Surgery only when conservative measures fail
Reserve surgery for those who have not responded adequately after 6 to 12 months of conservative therapy.5 Endoscopic plantar fascia release is superior to traditional open surgery.31 Heel spur resection is no longer routinely practiced. Patients undergoing surgery should expect a return to normal activity in approximately 2 to 3 months, and up to 35% of patients may continue to have symptoms after surgical intervention.2,31
Treatment options in perspective
Treat conservatively at first. Stretching the plantar fascia and heel cord, using prefabricated orthotics, and wearing night splints are backed by firm clinical evidence of benefit. Acute treatment of PF may also include CSI, especially for patients who are athletic or otherwise active and wish to return to full function as soon as possible, and are willing to accept the risks associated with CSI.
ESWT improves pain and function scores and may also relieve pain in patients with recalcitrant PF pain. PRP has limited but promising evidence for patients with chronic PF pain. Surgical intervention remains the last line of therapy and is not always effective at reducing pain.
CASE You prescribe a conservative treatment program of plantar fascia–specific stretches and prefabricated orthoses for the patient in the opening scenario. At one month, her pain drops by 30%. At 6 months, her pain disappears, and she resumes a daily aerobic exercise program to assist in weight loss.
CORRESPONDENCE
Carlton J. Covey, MD, Nellis Family Medicine Residency, 99MDOS/SGOF, 4700 Las Vegas Boulevard N, Las Vegas, NV 89191;
[email protected]
1. Riddle DL, Schappert SM. Volume of ambulatory care visits and patterns of care for patients diagnosed with plantar fasciitis: a national study of medical doctors. Foot Ankle Int. 2004;25: 303-310.
2. Glazer JL. An approach to the diagnosis and treatment of plantar fasciitis. Phys Sportsmed. 2009;37:74-79.
3. Lemont H, Ammirati KM, Usen N. Plantar fasciitis: a degenerative process (fasciosis) without inflammation. J Am Podiatr Med Assoc. 2003;93:234-237.
4. Thomas JL, Christensen JC, Kravitz SR, et al. The diagnosis and treatment of heal pain: a clinical practice guideline – revision 2010. J Foot Ankle Surg. 2010;49(suppl):S1-S19.
5. Neufeld SK, Cerrato R. Plantar fasciitis: evaluation and treatment. J Am Acad Orthop Surg. 2008;16:338-346.
6. Singh D, Angel J, Bentley G, et al. Fortnightly review: plantar fasciitis. BMJ. 1997;315:172-175.
7. McMillan AM, Landorf KB, Barrett JT, et al. Diagnostic imaging for chronic plantar heel pain: a systematic review and metaanalysis. J Foot Ankle Res. 2009;2:32.
8. Mahowald S, Legge BS, Grady JF. The correlation between plantar fascia thickness and symptoms of plantar fasciitis. J Am Podiatr Med Assoc. 2011;101:385-389.
9. American College of Radiology. ACR appropriateness criteria. Chronic foot pain. Available at: http://www.acr.org/~/media/ACR/Documents/AppCriteria/Diagnostic/ChronicFootPain.pdf. Accessed November 11, 2012.
10. Gill LH. Plantar fasciitis: diagnosis and conservative treatment. J Am Acad Orthop Surg. 1997;5:109-117.
11. Martin RL, Irrgang JJ, Conti SF. Outcome study of subjects with insertional plantar fasciitis. Foot Ankle Int. 1998;19:803-811.
12. DiGiovanni BF, Nawoczenski DA, Malay DP, et al. Plantar fasciaspecific stretching exercise improves outcomes in patients with chronic plantar fasciitis: a prospective clinic trial with two-year follow up. J Bone Joint Surg Am. 2006;88:1775-1781.
13. Sweeting D, Parish B, Hooper L, et al. The effectiveness of manual stretching in the treatment of plantar heel pain: a systemic review. J Foot Ankle Res. 2011;4:1-13.
14. Rompe JD, Cacchio A, Lowell W, et al. Plantar fascia-specific stretching versus radial shock-wave therapy as initial treatment of plantar fasciopathy. J Bone Joint Surg Am. 2010;92:2514-2522.
15. Renan-Ordine R, Alburquerque-Sendin F, Rodriques De Souza DP, et al. Effectiveness of myofascial trigger point manual therapy combined with a self stretching protocol for the management of plantar heel pain: a randomized controlled trial. J Orthop Sports Phys Ther. 2011;41:43-50.
16. Donley BG, Moore T, Sferra J, et al. The efficacy of oral nonsteroidal anti-inflammatory medication (NSAID) in the treatment of plantar fasciitis: a randomized, prospective, placebo-controlled study. Foot Ankle Int. 2007;28:20-23.
17. Lee SY, McKeon P, Hertel J. Does the use of orthoses improve selfreported pain and function measures in patients with plantar fasciitis? A meta-analysis. Phys Ther Sport. 2009;10:12-18.
18. Pfeffer G, Bacchetti P, Deland J, et al. Comparison of custom and prefabricated orthoses in the initial treatment of proximal plantar fasciitis. Foot Ankle Int. 1999;20:214-221.
19. Hawke F, Burns J, Radford JA, et al. Custom-made foot orthoses for the treatment of foot pain. Cochrane Database Syst Rev. 2008;(3):CD006801.
20. Fong DT, Pang KY, Chung MM, et al. Evaluation of combined prescription of rocker sole shoes and custom-made foot orthoses for the treatment of plantar fasciitis. Clin Biomech. 2012;27: 1072-1077.
21. Berlet GC, Anderson RB, Davis H. A prospective trial of night splinting in the treatment of recalcitrant plantar fasciitis: the Ankle Dorsiflexion Dynasplint. Orthopedics. 2002;25: 1273-1275.
22. Roos E, Engstrom M, Soderberg B. Foot orthoses for the treatment of plantar fasciitis. Foot Ankle Int. 2006;27:606-611.
23. Goff JD, Crawford R. Diagnosis and treatment of plantar fasciitis. Am Fam Physician. 2011;84:676-682.
24. Wang CJ. Extracorporeal shockwave therapy in musculoskeletal disorders. J Orthop Surg Res. 2012;7:11.
25. Landorf KB, Menz HB. Plantar heel pain and fasciitis. Clin Evid (Online). 2008;2008:1111.
26. Ball EM, McKeeman HM, Patterson C, et al. Steroid injection for inferior heel pain: a randomized controlled trial. Ann Rheum Dis. 2013;72:996-1002.
27. Uden H, Boesch E, Kumar S. Plantar fasciitis – to jab or support? A systematic review of the current best evidence. J Multidiscip Healthc. 2011;4:155-164.
28. Shetty VD. Platelet-rich plasma: a ‘feeling’ and ‘hope’ ailing athletes. Br J Sports Med. 2010;44(suppl 1):i1-i82.
29. Aksahin E, Dogruyol D, Yüksel HY, et al. The comparison of the effect of corticosteroids and platelet-rich plasma (PRP) for the treatment of plantar fasciitis. Arch Orthop Trauma Surg. 2012;132:781-785.
30. Ragab EM, Othman AM. Platelets rich plasma for treatment of chronic plantar fasciitis. Arch Orthop Trauma Surg. 2012;132:1065-1070.
31. Saxena A. Uniportal endoscopic plantar fasciotomy: a prospective study on athletic patients. Foot Ankle Int. 2004;25:882-889.
1. Riddle DL, Schappert SM. Volume of ambulatory care visits and patterns of care for patients diagnosed with plantar fasciitis: a national study of medical doctors. Foot Ankle Int. 2004;25: 303-310.
2. Glazer JL. An approach to the diagnosis and treatment of plantar fasciitis. Phys Sportsmed. 2009;37:74-79.
3. Lemont H, Ammirati KM, Usen N. Plantar fasciitis: a degenerative process (fasciosis) without inflammation. J Am Podiatr Med Assoc. 2003;93:234-237.
4. Thomas JL, Christensen JC, Kravitz SR, et al. The diagnosis and treatment of heal pain: a clinical practice guideline – revision 2010. J Foot Ankle Surg. 2010;49(suppl):S1-S19.
5. Neufeld SK, Cerrato R. Plantar fasciitis: evaluation and treatment. J Am Acad Orthop Surg. 2008;16:338-346.
6. Singh D, Angel J, Bentley G, et al. Fortnightly review: plantar fasciitis. BMJ. 1997;315:172-175.
7. McMillan AM, Landorf KB, Barrett JT, et al. Diagnostic imaging for chronic plantar heel pain: a systematic review and metaanalysis. J Foot Ankle Res. 2009;2:32.
8. Mahowald S, Legge BS, Grady JF. The correlation between plantar fascia thickness and symptoms of plantar fasciitis. J Am Podiatr Med Assoc. 2011;101:385-389.
9. American College of Radiology. ACR appropriateness criteria. Chronic foot pain. Available at: http://www.acr.org/~/media/ACR/Documents/AppCriteria/Diagnostic/ChronicFootPain.pdf. Accessed November 11, 2012.
10. Gill LH. Plantar fasciitis: diagnosis and conservative treatment. J Am Acad Orthop Surg. 1997;5:109-117.
11. Martin RL, Irrgang JJ, Conti SF. Outcome study of subjects with insertional plantar fasciitis. Foot Ankle Int. 1998;19:803-811.
12. DiGiovanni BF, Nawoczenski DA, Malay DP, et al. Plantar fasciaspecific stretching exercise improves outcomes in patients with chronic plantar fasciitis: a prospective clinic trial with two-year follow up. J Bone Joint Surg Am. 2006;88:1775-1781.
13. Sweeting D, Parish B, Hooper L, et al. The effectiveness of manual stretching in the treatment of plantar heel pain: a systemic review. J Foot Ankle Res. 2011;4:1-13.
14. Rompe JD, Cacchio A, Lowell W, et al. Plantar fascia-specific stretching versus radial shock-wave therapy as initial treatment of plantar fasciopathy. J Bone Joint Surg Am. 2010;92:2514-2522.
15. Renan-Ordine R, Alburquerque-Sendin F, Rodriques De Souza DP, et al. Effectiveness of myofascial trigger point manual therapy combined with a self stretching protocol for the management of plantar heel pain: a randomized controlled trial. J Orthop Sports Phys Ther. 2011;41:43-50.
16. Donley BG, Moore T, Sferra J, et al. The efficacy of oral nonsteroidal anti-inflammatory medication (NSAID) in the treatment of plantar fasciitis: a randomized, prospective, placebo-controlled study. Foot Ankle Int. 2007;28:20-23.
17. Lee SY, McKeon P, Hertel J. Does the use of orthoses improve selfreported pain and function measures in patients with plantar fasciitis? A meta-analysis. Phys Ther Sport. 2009;10:12-18.
18. Pfeffer G, Bacchetti P, Deland J, et al. Comparison of custom and prefabricated orthoses in the initial treatment of proximal plantar fasciitis. Foot Ankle Int. 1999;20:214-221.
19. Hawke F, Burns J, Radford JA, et al. Custom-made foot orthoses for the treatment of foot pain. Cochrane Database Syst Rev. 2008;(3):CD006801.
20. Fong DT, Pang KY, Chung MM, et al. Evaluation of combined prescription of rocker sole shoes and custom-made foot orthoses for the treatment of plantar fasciitis. Clin Biomech. 2012;27: 1072-1077.
21. Berlet GC, Anderson RB, Davis H. A prospective trial of night splinting in the treatment of recalcitrant plantar fasciitis: the Ankle Dorsiflexion Dynasplint. Orthopedics. 2002;25: 1273-1275.
22. Roos E, Engstrom M, Soderberg B. Foot orthoses for the treatment of plantar fasciitis. Foot Ankle Int. 2006;27:606-611.
23. Goff JD, Crawford R. Diagnosis and treatment of plantar fasciitis. Am Fam Physician. 2011;84:676-682.
24. Wang CJ. Extracorporeal shockwave therapy in musculoskeletal disorders. J Orthop Surg Res. 2012;7:11.
25. Landorf KB, Menz HB. Plantar heel pain and fasciitis. Clin Evid (Online). 2008;2008:1111.
26. Ball EM, McKeeman HM, Patterson C, et al. Steroid injection for inferior heel pain: a randomized controlled trial. Ann Rheum Dis. 2013;72:996-1002.
27. Uden H, Boesch E, Kumar S. Plantar fasciitis – to jab or support? A systematic review of the current best evidence. J Multidiscip Healthc. 2011;4:155-164.
28. Shetty VD. Platelet-rich plasma: a ‘feeling’ and ‘hope’ ailing athletes. Br J Sports Med. 2010;44(suppl 1):i1-i82.
29. Aksahin E, Dogruyol D, Yüksel HY, et al. The comparison of the effect of corticosteroids and platelet-rich plasma (PRP) for the treatment of plantar fasciitis. Arch Orthop Trauma Surg. 2012;132:781-785.
30. Ragab EM, Othman AM. Platelets rich plasma for treatment of chronic plantar fasciitis. Arch Orthop Trauma Surg. 2012;132:1065-1070.
31. Saxena A. Uniportal endoscopic plantar fasciotomy: a prospective study on athletic patients. Foot Ankle Int. 2004;25:882-889.
The Pros and Cons of Using Larger Femoral Heads in Total Hip Arthroplasty
Colorectal cancer risk increased with bariatric surgery
The risk of colorectal cancer was significantly increased among people who had undergone obesity surgery in a retrospective cohort study of more than 77,000 obese patients enrolled in a Swedish registry.
The increased risk for colorectal cancer was associated with all three bariatric procedures – vertical banded gastroplasty, adjustable gastric banding, and Roux-en-Y gastric bypass – and increased further over time, reported Dr. Maryam Derogar, of the Karolinska Institutet, Stockholm, and her associates. No such pattern over time was seen among the obese patients who did not have surgery.
"Our data suggest that increased colorectal cancer risk may be a long-term consequence of such surgery," they concluded. If the association is confirmed, they added, "it should stimulate research addressing colonoscopic evaluation of the incidence of colorectal adenomatous polyps after obesity surgery with a view to defining an optimum colonoscopy surveillance strategy for the increasing number of patients who undergo obesity surgery. The study was published online in the Annals of Surgery (2013 [doi:10.1097/SLA.0b013e318288463a]).
To address their "unexpected" finding in an earlier study of an apparent increase in the risk of colorectal cancer after obesity surgery, but no increase in the risk of other cancers related to obesity, they conducted a retrospective cohort study using national registry data between 1980 and 2009, of 15,095 obese patients who had undergone obesity surgery and 62,016 patients who had been diagnosed with obesity but did not undergo surgery. They calculated the colorectal cancer risk using the standardized incidence ratio (SIR), the observed number of cases divided by the number of expected cases in that group.
Over a median of 10 years, there were 70 colorectal cancers in the obesity surgery group; and over a median of 7 years, 373 among those who had no surgery. The SIR for colorectal cancer among those who had surgery was 1.60, which was statistically significant. Among those who had no surgery, there was a small, insignificant increase in risk group (a SIR of 1.26). In the surgery group, the risk increased over time in men and women, up to a twofold increased risk among those patients followed for at least 10 years, a pattern than was not observed in the obese patients who had no surgery.
The "substantial increase in colorectal cancer risk, above that associated with excess body weight alone, more than 10 years after surgery is compatible with the long natural history of colorectal carcinogenesis from normal mucosa to a malignant colorectal cancer," the authors wrote. Why the risk was increased is not clear, but one possible explanation could be that the malabsorption effects of the gastric bypass procedure results in local mucosal changes, the authors speculated. Previously, they had identified rectal mucosal hyperproliferation in patients who had undergone obesity surgery, present at least 3 years after the procedure, a finding that was "associated with increased mucosal expression of the protumorigenic cytokine macrophage migration inhibitory factor," they wrote.
The study’s strengths included the size of the sample, long follow-up, and the validity of Swedish national registry data, while the limitations included the retrospective design and the lack of data on body weight over time.
As in the United States and other countries, obesity has been increasing in Sweden, with a corresponding increase in bariatric surgery. Over the last 20 years, the prevalence of obesity in Sweden has doubled, and the annual number of obesity operations performed has increased from 1,500 in 2006 to almost 4,000 in 2009, according to the authors.
The study was supported by the Swedish Research Council. The authors had no conflicts of interest to declare.
The risk of colorectal cancer was significantly increased among people who had undergone obesity surgery in a retrospective cohort study of more than 77,000 obese patients enrolled in a Swedish registry.
The increased risk for colorectal cancer was associated with all three bariatric procedures – vertical banded gastroplasty, adjustable gastric banding, and Roux-en-Y gastric bypass – and increased further over time, reported Dr. Maryam Derogar, of the Karolinska Institutet, Stockholm, and her associates. No such pattern over time was seen among the obese patients who did not have surgery.
"Our data suggest that increased colorectal cancer risk may be a long-term consequence of such surgery," they concluded. If the association is confirmed, they added, "it should stimulate research addressing colonoscopic evaluation of the incidence of colorectal adenomatous polyps after obesity surgery with a view to defining an optimum colonoscopy surveillance strategy for the increasing number of patients who undergo obesity surgery. The study was published online in the Annals of Surgery (2013 [doi:10.1097/SLA.0b013e318288463a]).
To address their "unexpected" finding in an earlier study of an apparent increase in the risk of colorectal cancer after obesity surgery, but no increase in the risk of other cancers related to obesity, they conducted a retrospective cohort study using national registry data between 1980 and 2009, of 15,095 obese patients who had undergone obesity surgery and 62,016 patients who had been diagnosed with obesity but did not undergo surgery. They calculated the colorectal cancer risk using the standardized incidence ratio (SIR), the observed number of cases divided by the number of expected cases in that group.
Over a median of 10 years, there were 70 colorectal cancers in the obesity surgery group; and over a median of 7 years, 373 among those who had no surgery. The SIR for colorectal cancer among those who had surgery was 1.60, which was statistically significant. Among those who had no surgery, there was a small, insignificant increase in risk group (a SIR of 1.26). In the surgery group, the risk increased over time in men and women, up to a twofold increased risk among those patients followed for at least 10 years, a pattern than was not observed in the obese patients who had no surgery.
The "substantial increase in colorectal cancer risk, above that associated with excess body weight alone, more than 10 years after surgery is compatible with the long natural history of colorectal carcinogenesis from normal mucosa to a malignant colorectal cancer," the authors wrote. Why the risk was increased is not clear, but one possible explanation could be that the malabsorption effects of the gastric bypass procedure results in local mucosal changes, the authors speculated. Previously, they had identified rectal mucosal hyperproliferation in patients who had undergone obesity surgery, present at least 3 years after the procedure, a finding that was "associated with increased mucosal expression of the protumorigenic cytokine macrophage migration inhibitory factor," they wrote.
The study’s strengths included the size of the sample, long follow-up, and the validity of Swedish national registry data, while the limitations included the retrospective design and the lack of data on body weight over time.
As in the United States and other countries, obesity has been increasing in Sweden, with a corresponding increase in bariatric surgery. Over the last 20 years, the prevalence of obesity in Sweden has doubled, and the annual number of obesity operations performed has increased from 1,500 in 2006 to almost 4,000 in 2009, according to the authors.
The study was supported by the Swedish Research Council. The authors had no conflicts of interest to declare.
The risk of colorectal cancer was significantly increased among people who had undergone obesity surgery in a retrospective cohort study of more than 77,000 obese patients enrolled in a Swedish registry.
The increased risk for colorectal cancer was associated with all three bariatric procedures – vertical banded gastroplasty, adjustable gastric banding, and Roux-en-Y gastric bypass – and increased further over time, reported Dr. Maryam Derogar, of the Karolinska Institutet, Stockholm, and her associates. No such pattern over time was seen among the obese patients who did not have surgery.
"Our data suggest that increased colorectal cancer risk may be a long-term consequence of such surgery," they concluded. If the association is confirmed, they added, "it should stimulate research addressing colonoscopic evaluation of the incidence of colorectal adenomatous polyps after obesity surgery with a view to defining an optimum colonoscopy surveillance strategy for the increasing number of patients who undergo obesity surgery. The study was published online in the Annals of Surgery (2013 [doi:10.1097/SLA.0b013e318288463a]).
To address their "unexpected" finding in an earlier study of an apparent increase in the risk of colorectal cancer after obesity surgery, but no increase in the risk of other cancers related to obesity, they conducted a retrospective cohort study using national registry data between 1980 and 2009, of 15,095 obese patients who had undergone obesity surgery and 62,016 patients who had been diagnosed with obesity but did not undergo surgery. They calculated the colorectal cancer risk using the standardized incidence ratio (SIR), the observed number of cases divided by the number of expected cases in that group.
Over a median of 10 years, there were 70 colorectal cancers in the obesity surgery group; and over a median of 7 years, 373 among those who had no surgery. The SIR for colorectal cancer among those who had surgery was 1.60, which was statistically significant. Among those who had no surgery, there was a small, insignificant increase in risk group (a SIR of 1.26). In the surgery group, the risk increased over time in men and women, up to a twofold increased risk among those patients followed for at least 10 years, a pattern than was not observed in the obese patients who had no surgery.
The "substantial increase in colorectal cancer risk, above that associated with excess body weight alone, more than 10 years after surgery is compatible with the long natural history of colorectal carcinogenesis from normal mucosa to a malignant colorectal cancer," the authors wrote. Why the risk was increased is not clear, but one possible explanation could be that the malabsorption effects of the gastric bypass procedure results in local mucosal changes, the authors speculated. Previously, they had identified rectal mucosal hyperproliferation in patients who had undergone obesity surgery, present at least 3 years after the procedure, a finding that was "associated with increased mucosal expression of the protumorigenic cytokine macrophage migration inhibitory factor," they wrote.
The study’s strengths included the size of the sample, long follow-up, and the validity of Swedish national registry data, while the limitations included the retrospective design and the lack of data on body weight over time.
As in the United States and other countries, obesity has been increasing in Sweden, with a corresponding increase in bariatric surgery. Over the last 20 years, the prevalence of obesity in Sweden has doubled, and the annual number of obesity operations performed has increased from 1,500 in 2006 to almost 4,000 in 2009, according to the authors.
The study was supported by the Swedish Research Council. The authors had no conflicts of interest to declare.
FROM THE ANNALS OF SURGERY