User login
Evaluation of the Limping Child
Pelvic Fractures-Preventing Complications
Common Facial Injuries
Pigmented Villonodular Synovitis
Should you worry that patients will use the Web to grade you?
CASE: Accusatory comments from a nameless source
A small-town ObGyn discovers an online surprise: a claim by an unidentified patient that he caused the stillbirth of her infant. Convinced that the allegation is false, he strives to uncover the patient’s identity and counter the charges. Only after months of litigation is he successful. The patient in question was particularly noncompliant with prenatal care: She continued to consume alcohol and smoke throughout the pregnancy.
Could this kind of smear on a reputation happen to you?
Professionals have long gone to extreme measures to protect their reputation. In the early days of the Republic, gentlemen defended their honor with violence. Remember Alexander Hamilton? After his allegedly defamatory comments about longtime rival Aaron Burr appeared in a New York newspaper, Burr killed Hamilton in a duel.
We are fortunate that an attack on our professional reputation no longer needs to be addressed with violence—but we have other challenges to overcome. Scandalous comments can now be posted instantly, worldwide. Such comments can be uploaded anonymously by anyone who has a computer and a vendetta. Under Section 230 of the Federal Communications Decency Act, Web sites that serve as platforms for blogs enjoy complete immunity—even when those blogs are malicious.
With a mouse-click, a person can anonymously tarnish a reputation that took decades to build. Such musings include “He is a butcher who should be in jail” or “He’s a rotten doctor and a thief.” Hosting Web sites have no legal responsibility for content. Even if a physician can determine the blogger’s identity—itself a challenge—the doctor must hold his tongue because of privacy laws.
The Web sites that purport to “rate” physicians are hardly repositories of substantive information on which to base life-and-death decisions. Some use the sophisticated metrics of “smiley faces” or “frowny faces” to characterize doctors. The great majority of sites admit that they cannot confirm that the poster is even a patient.
One thing is evident: Most of these sites exist to make money. Observe the ads for erectile dysfunction drugs that straddle the posts. The more salacious the posts, the more eyeballs and dollars for the owners of the site and the sponsors of the ads.
Is there a solution that can protect physicians?
In response to this worsening problem, we developed an agreement for physicians to use with patients to prohibit online rating of the physician without the doctor’s assent. The document was unveiled in 2007 as a bilateral privacy agreement, but it has evolved to encourage ratings on sites that embrace minimum standards of fairness and balance. This is accomplished by having the patient transfer copyright to commentary to the treating physician. If a post is fraudulent or defamatory, the doctor has a tool to enable its removal.*
The initial agreement received much attention—and some criticism. Opponents refer to it as a gag order that impedes free speech. “What are they trying to hide?,” goes the insinuation. “Patients should have a right to know what kind of care they can expect to receive from a doctor,” plead others.
We agree: Patients have a need and a right to know what kind of care they can expect from a physician. But we reject the premise that anonymous, undocumented comments posted on a Web site whose owners are unaccountable for what is said produce this information. We do believe that patients are entirely qualified to provide impressions, but such Web sites are being used as surrogate, supposedly objective measures of quality of care. In that role, the sites have failed.
A physician is not a roofer
Selecting a physician is different from hiring a roofer. First, some measure of the success of medicine depends on the patient. Adherent patients fare better than nonadherent ones. A roofer asks only that the customer pay for services provided. If that roofer asked the customer to pay and, in addition, layer the caulk, the comparison might work, but that’s not the case.
Second, health care is rarely provided by one source. A patient’s care is more often collaborative than the product of an individual. Think how many doctors are involved in treating a patient hospitalized for a few days.
Third, more frightening is that many hospitalized patients cannot even identify their physicians. In a survey released earlier this year, three quarters of patients were unable to name anyone in charge of their care. Worse, of those who provided at least one name, 60% gave an incorrect answer. The survey included more than 2,800 patients.1
Last, it is difficult for a patient to evaluate a physician’s judgment and technical prowess objectively. Patients can offer useful subjective information about many aspects of a physician’s skills, such as ability to communicate, but other areas, such as deep technical skills, are less than clear. With the roofer, it’s easy: Either the roof leaks or it doesn’t. Evaluating the details of a gynecologic oncology procedure—well, that’s another matter.
Until 2008, Angie’s List (www.angieslist.com) was known primarily for its reviews of plumbers, handymen, and other contractors. But in March 2008, the Web site rolled out 40 new categories of service providers—all of them related to health care.
The result: 10,000 physician reviews in the first month.
“We just saw a tremendous amount of interest,” says Mike Rutz, Vice President of Angie’s List Health—so much interest that the company increased the number of health-care categories to 150 shortly thereafter, including one for ObGyn care.
Angie’s List is a pay-to-use service with roughly 1,000,000 members in the United States. It differs from other physician-rating sites in other respects as well: Reviews are not anonymous, and any physician who receives a negative review is given the opportunity to respond. In fact, a physician can sign up, free of charge, to be notified when a review goes up on the site. Although the member’s name is not posted on the individual review, it is recorded so that any disputes can be clarified.
“We do have some doctors who say, ‘These people are not my patients,’” Rutz reports. When this happens, Angie’s List can consult its database and resolve the issue definitively.
How is the physician rated?
A patient gives her physician a grade, ranging from A to F, in eight categories:
- availability
- office environment
- punctuality
- staff friendliness
- bedside manner
- communication
- effectiveness of treatment
- billing and administration.
The eight scores are averaged to yield an overall grade.
When a member seeks information on health-care providers in her locality, she sees only the overall grade at first. She clicks through this screen to view the full report.
Rutz believes that most patients have the expertise to judge effectiveness of treatment. “They absolutely know whether the treatment was effective,” he says. He does concede that “the patient is the most important participant in her health care” and does have an impact on the success or failure of treatment. Angie’s List has no plans to remove effectiveness of treatment from the rating categories.
Physicians can encourage positive reviews
Physicians aren’t penalized for encouraging patients to give them a positive review on Angie’s List—in fact, that strategy is encouraged.
“More information is better,” says Rutz. “The folks that are encouraging reviews are usually the folks providing the best service.”
There is an added bonus to positive reviews: Providers who have the best grades and the most reports rise to the top of the list, gaining prominence on the site.
At the same time, there are mechanisms in place to prevent a health-care or other service provider from “gaming the system,” Rutz adds. It is not acceptable for a physician or a member of his or her staff or family to file a review.
So is Angie’s List good news for physicians?
Jeffrey Segal, MD, does not think so.
The problem is that the site requires paid membership, Dr. Segal, founder of Medical Justice Services in Greensboro, NC, says.
“Because of that, [Angie’s List] will never really accumulate more than a handful of reviews on any given doctor; particularly given the number of free sites. And since the average doctor sees over 1,000 patients a year, we do not believe three or four reviews can ever mean anything substantive.”—JANELLE YATES, SENIOR EDITOR
How rating sites need to change
You may conclude that we oppose online rating of physicians. We do not. We understand the desire to know as much as possible about a provider’s abilities. We merely appeal to online rating sites to adhere to minimum standards that promote a responsible system for both physicians and patients.
Here are our recommendations:
- Ensure that every potential rater who claims to be a patient of a particular physician really is one.
- Require a meaningful number of raters. Given that the average physician sees 1,000 to 2,000 patients in a year, ratings that involve only two or three patients are meaningless. Forty or 50 ratings are another story. Comments could be collected until a threshold number is reached. They could then be published.
- Require patients to stick to areas in which they are expert—namely, subjective impressions. A physician’s personality, staff, and communication skills are fair game. Skill at performing oophorectomy—out of bounds. However, should a patient wish to have the physician’s technical skills reviewed by a trained professional, all the better.
For example, to deputize the patient as a partner in preventing spread of infection and wrong-site surgery, a survey can ask: “Do you recall if the doctor washed her hands before she examined you?” (The answer is informative only if the office asks the patient, upfront, to make this observation.) Another question: “Did the doctor ask you to mark your name on the affected limb before surgery?”
The feedback loop will either confirm great performance or serve as a strong motivator for fast improvement. Both physicians and patients must participate to make the health-care system better.
Even when reviews are positive, the experience of being rated on the Web is a little disconcerting.
Why?
Most reviews are anonymous, and few, if any, patients have the background to fully understand why a particular treatment succeeded—or did not. That’s the view, at least, of two ObGyns interviewed—both of whom serve on the Board of Editors of OBG Management.
Steven R. Goldstein, MD, has been glowingly reviewed on Angie’s List, but still finds the value of online rating dubious. Dr. Goldstein is professor of obstetrics and gynecology at New York University School of Medicine and director of gynecologic ultrasound and co-director of bone densitometry at New York University Medical Center in New York City.
“There’s nothing scientific or objective about it,” he says, particularly when it comes to assessment of complex medical treatment. “Patients can say how they felt about the person,” he adds, but their ability to evaluate the scientific and technical components of treatment is limited.
Barbara S. Levy, MD, agrees. She is medical director of the Women’s Health Center for the Franciscan Health System in Federal Way, Washington.
“I think people can rate their experience,” she says. “I don’t think they can judge training and surgical skill.” Dr. Levy has been rated positively on several Web sites.
Neither physician had read any reviews of their services. Both are “kind of” aware that online rating is increasingly common.
“It’s happening in everything,” Dr. Levy says.
Is there reason to worry about a poor rating?
Dr. Levy believes that anonymous reviews should be prohibited. “Not that a site has to publish the names of reviewers,” she says, “only that whatever agency is allowing them to do the reviewing should be collecting their information to make sure they are legitimate. It’s just due diligence.”
Dr. Levy does believe that patients should have the right to review their experience with physicians. As for what to do about an unfair review, “I think it’s only fair for the doctor to be able to rebut it,” she says.
Dr. Goldstein has a slightly different take on the matter.
“I don’t really have the time or energy to devote to this issue,” he says. “I like to think that patients who come to me do so because of other patients, whom they know fairly well, or other physicians.” If a patient sees one negative Web review and chooses not to use a doctor on that basis, “there isn’t much I can do about it,” he says.
“The most important part of this issue”—online rating of physicians—“is that it is totally unsubstantiated.”—JANELLE YATES, SENIOR EDITOR
What we are doing now
Our company is working with rating sites that share our understanding of the problem and endorse our proposed solutions. Bad information can be worse than no information, we argue! If health care is to be improved, patients and physicians need high-quality information about providers and health-care systems. By injecting fairness and accountability into online ratings, we will all benefit.
CASE RESOLVED
By the time the ObGyn is able to have the woman’s comments removed from the Web site, he lived for more than a year with the innuendo made possible by the Internet.
1. Arora V, Gangireddy S, Mehhrotra A, Ginde R, Tormey M, Meltzer D. Ability of hospitalized patients to identify their inhospital physicians. Arch Intern Med. 2009;169:199-201.
CASE: Accusatory comments from a nameless source
A small-town ObGyn discovers an online surprise: a claim by an unidentified patient that he caused the stillbirth of her infant. Convinced that the allegation is false, he strives to uncover the patient’s identity and counter the charges. Only after months of litigation is he successful. The patient in question was particularly noncompliant with prenatal care: She continued to consume alcohol and smoke throughout the pregnancy.
Could this kind of smear on a reputation happen to you?
Professionals have long gone to extreme measures to protect their reputation. In the early days of the Republic, gentlemen defended their honor with violence. Remember Alexander Hamilton? After his allegedly defamatory comments about longtime rival Aaron Burr appeared in a New York newspaper, Burr killed Hamilton in a duel.
We are fortunate that an attack on our professional reputation no longer needs to be addressed with violence—but we have other challenges to overcome. Scandalous comments can now be posted instantly, worldwide. Such comments can be uploaded anonymously by anyone who has a computer and a vendetta. Under Section 230 of the Federal Communications Decency Act, Web sites that serve as platforms for blogs enjoy complete immunity—even when those blogs are malicious.
With a mouse-click, a person can anonymously tarnish a reputation that took decades to build. Such musings include “He is a butcher who should be in jail” or “He’s a rotten doctor and a thief.” Hosting Web sites have no legal responsibility for content. Even if a physician can determine the blogger’s identity—itself a challenge—the doctor must hold his tongue because of privacy laws.
The Web sites that purport to “rate” physicians are hardly repositories of substantive information on which to base life-and-death decisions. Some use the sophisticated metrics of “smiley faces” or “frowny faces” to characterize doctors. The great majority of sites admit that they cannot confirm that the poster is even a patient.
One thing is evident: Most of these sites exist to make money. Observe the ads for erectile dysfunction drugs that straddle the posts. The more salacious the posts, the more eyeballs and dollars for the owners of the site and the sponsors of the ads.
Is there a solution that can protect physicians?
In response to this worsening problem, we developed an agreement for physicians to use with patients to prohibit online rating of the physician without the doctor’s assent. The document was unveiled in 2007 as a bilateral privacy agreement, but it has evolved to encourage ratings on sites that embrace minimum standards of fairness and balance. This is accomplished by having the patient transfer copyright to commentary to the treating physician. If a post is fraudulent or defamatory, the doctor has a tool to enable its removal.*
The initial agreement received much attention—and some criticism. Opponents refer to it as a gag order that impedes free speech. “What are they trying to hide?,” goes the insinuation. “Patients should have a right to know what kind of care they can expect to receive from a doctor,” plead others.
We agree: Patients have a need and a right to know what kind of care they can expect from a physician. But we reject the premise that anonymous, undocumented comments posted on a Web site whose owners are unaccountable for what is said produce this information. We do believe that patients are entirely qualified to provide impressions, but such Web sites are being used as surrogate, supposedly objective measures of quality of care. In that role, the sites have failed.
A physician is not a roofer
Selecting a physician is different from hiring a roofer. First, some measure of the success of medicine depends on the patient. Adherent patients fare better than nonadherent ones. A roofer asks only that the customer pay for services provided. If that roofer asked the customer to pay and, in addition, layer the caulk, the comparison might work, but that’s not the case.
Second, health care is rarely provided by one source. A patient’s care is more often collaborative than the product of an individual. Think how many doctors are involved in treating a patient hospitalized for a few days.
Third, more frightening is that many hospitalized patients cannot even identify their physicians. In a survey released earlier this year, three quarters of patients were unable to name anyone in charge of their care. Worse, of those who provided at least one name, 60% gave an incorrect answer. The survey included more than 2,800 patients.1
Last, it is difficult for a patient to evaluate a physician’s judgment and technical prowess objectively. Patients can offer useful subjective information about many aspects of a physician’s skills, such as ability to communicate, but other areas, such as deep technical skills, are less than clear. With the roofer, it’s easy: Either the roof leaks or it doesn’t. Evaluating the details of a gynecologic oncology procedure—well, that’s another matter.
Until 2008, Angie’s List (www.angieslist.com) was known primarily for its reviews of plumbers, handymen, and other contractors. But in March 2008, the Web site rolled out 40 new categories of service providers—all of them related to health care.
The result: 10,000 physician reviews in the first month.
“We just saw a tremendous amount of interest,” says Mike Rutz, Vice President of Angie’s List Health—so much interest that the company increased the number of health-care categories to 150 shortly thereafter, including one for ObGyn care.
Angie’s List is a pay-to-use service with roughly 1,000,000 members in the United States. It differs from other physician-rating sites in other respects as well: Reviews are not anonymous, and any physician who receives a negative review is given the opportunity to respond. In fact, a physician can sign up, free of charge, to be notified when a review goes up on the site. Although the member’s name is not posted on the individual review, it is recorded so that any disputes can be clarified.
“We do have some doctors who say, ‘These people are not my patients,’” Rutz reports. When this happens, Angie’s List can consult its database and resolve the issue definitively.
How is the physician rated?
A patient gives her physician a grade, ranging from A to F, in eight categories:
- availability
- office environment
- punctuality
- staff friendliness
- bedside manner
- communication
- effectiveness of treatment
- billing and administration.
The eight scores are averaged to yield an overall grade.
When a member seeks information on health-care providers in her locality, she sees only the overall grade at first. She clicks through this screen to view the full report.
Rutz believes that most patients have the expertise to judge effectiveness of treatment. “They absolutely know whether the treatment was effective,” he says. He does concede that “the patient is the most important participant in her health care” and does have an impact on the success or failure of treatment. Angie’s List has no plans to remove effectiveness of treatment from the rating categories.
Physicians can encourage positive reviews
Physicians aren’t penalized for encouraging patients to give them a positive review on Angie’s List—in fact, that strategy is encouraged.
“More information is better,” says Rutz. “The folks that are encouraging reviews are usually the folks providing the best service.”
There is an added bonus to positive reviews: Providers who have the best grades and the most reports rise to the top of the list, gaining prominence on the site.
At the same time, there are mechanisms in place to prevent a health-care or other service provider from “gaming the system,” Rutz adds. It is not acceptable for a physician or a member of his or her staff or family to file a review.
So is Angie’s List good news for physicians?
Jeffrey Segal, MD, does not think so.
The problem is that the site requires paid membership, Dr. Segal, founder of Medical Justice Services in Greensboro, NC, says.
“Because of that, [Angie’s List] will never really accumulate more than a handful of reviews on any given doctor; particularly given the number of free sites. And since the average doctor sees over 1,000 patients a year, we do not believe three or four reviews can ever mean anything substantive.”—JANELLE YATES, SENIOR EDITOR
How rating sites need to change
You may conclude that we oppose online rating of physicians. We do not. We understand the desire to know as much as possible about a provider’s abilities. We merely appeal to online rating sites to adhere to minimum standards that promote a responsible system for both physicians and patients.
Here are our recommendations:
- Ensure that every potential rater who claims to be a patient of a particular physician really is one.
- Require a meaningful number of raters. Given that the average physician sees 1,000 to 2,000 patients in a year, ratings that involve only two or three patients are meaningless. Forty or 50 ratings are another story. Comments could be collected until a threshold number is reached. They could then be published.
- Require patients to stick to areas in which they are expert—namely, subjective impressions. A physician’s personality, staff, and communication skills are fair game. Skill at performing oophorectomy—out of bounds. However, should a patient wish to have the physician’s technical skills reviewed by a trained professional, all the better.
For example, to deputize the patient as a partner in preventing spread of infection and wrong-site surgery, a survey can ask: “Do you recall if the doctor washed her hands before she examined you?” (The answer is informative only if the office asks the patient, upfront, to make this observation.) Another question: “Did the doctor ask you to mark your name on the affected limb before surgery?”
The feedback loop will either confirm great performance or serve as a strong motivator for fast improvement. Both physicians and patients must participate to make the health-care system better.
Even when reviews are positive, the experience of being rated on the Web is a little disconcerting.
Why?
Most reviews are anonymous, and few, if any, patients have the background to fully understand why a particular treatment succeeded—or did not. That’s the view, at least, of two ObGyns interviewed—both of whom serve on the Board of Editors of OBG Management.
Steven R. Goldstein, MD, has been glowingly reviewed on Angie’s List, but still finds the value of online rating dubious. Dr. Goldstein is professor of obstetrics and gynecology at New York University School of Medicine and director of gynecologic ultrasound and co-director of bone densitometry at New York University Medical Center in New York City.
“There’s nothing scientific or objective about it,” he says, particularly when it comes to assessment of complex medical treatment. “Patients can say how they felt about the person,” he adds, but their ability to evaluate the scientific and technical components of treatment is limited.
Barbara S. Levy, MD, agrees. She is medical director of the Women’s Health Center for the Franciscan Health System in Federal Way, Washington.
“I think people can rate their experience,” she says. “I don’t think they can judge training and surgical skill.” Dr. Levy has been rated positively on several Web sites.
Neither physician had read any reviews of their services. Both are “kind of” aware that online rating is increasingly common.
“It’s happening in everything,” Dr. Levy says.
Is there reason to worry about a poor rating?
Dr. Levy believes that anonymous reviews should be prohibited. “Not that a site has to publish the names of reviewers,” she says, “only that whatever agency is allowing them to do the reviewing should be collecting their information to make sure they are legitimate. It’s just due diligence.”
Dr. Levy does believe that patients should have the right to review their experience with physicians. As for what to do about an unfair review, “I think it’s only fair for the doctor to be able to rebut it,” she says.
Dr. Goldstein has a slightly different take on the matter.
“I don’t really have the time or energy to devote to this issue,” he says. “I like to think that patients who come to me do so because of other patients, whom they know fairly well, or other physicians.” If a patient sees one negative Web review and chooses not to use a doctor on that basis, “there isn’t much I can do about it,” he says.
“The most important part of this issue”—online rating of physicians—“is that it is totally unsubstantiated.”—JANELLE YATES, SENIOR EDITOR
What we are doing now
Our company is working with rating sites that share our understanding of the problem and endorse our proposed solutions. Bad information can be worse than no information, we argue! If health care is to be improved, patients and physicians need high-quality information about providers and health-care systems. By injecting fairness and accountability into online ratings, we will all benefit.
CASE RESOLVED
By the time the ObGyn is able to have the woman’s comments removed from the Web site, he lived for more than a year with the innuendo made possible by the Internet.
CASE: Accusatory comments from a nameless source
A small-town ObGyn discovers an online surprise: a claim by an unidentified patient that he caused the stillbirth of her infant. Convinced that the allegation is false, he strives to uncover the patient’s identity and counter the charges. Only after months of litigation is he successful. The patient in question was particularly noncompliant with prenatal care: She continued to consume alcohol and smoke throughout the pregnancy.
Could this kind of smear on a reputation happen to you?
Professionals have long gone to extreme measures to protect their reputation. In the early days of the Republic, gentlemen defended their honor with violence. Remember Alexander Hamilton? After his allegedly defamatory comments about longtime rival Aaron Burr appeared in a New York newspaper, Burr killed Hamilton in a duel.
We are fortunate that an attack on our professional reputation no longer needs to be addressed with violence—but we have other challenges to overcome. Scandalous comments can now be posted instantly, worldwide. Such comments can be uploaded anonymously by anyone who has a computer and a vendetta. Under Section 230 of the Federal Communications Decency Act, Web sites that serve as platforms for blogs enjoy complete immunity—even when those blogs are malicious.
With a mouse-click, a person can anonymously tarnish a reputation that took decades to build. Such musings include “He is a butcher who should be in jail” or “He’s a rotten doctor and a thief.” Hosting Web sites have no legal responsibility for content. Even if a physician can determine the blogger’s identity—itself a challenge—the doctor must hold his tongue because of privacy laws.
The Web sites that purport to “rate” physicians are hardly repositories of substantive information on which to base life-and-death decisions. Some use the sophisticated metrics of “smiley faces” or “frowny faces” to characterize doctors. The great majority of sites admit that they cannot confirm that the poster is even a patient.
One thing is evident: Most of these sites exist to make money. Observe the ads for erectile dysfunction drugs that straddle the posts. The more salacious the posts, the more eyeballs and dollars for the owners of the site and the sponsors of the ads.
Is there a solution that can protect physicians?
In response to this worsening problem, we developed an agreement for physicians to use with patients to prohibit online rating of the physician without the doctor’s assent. The document was unveiled in 2007 as a bilateral privacy agreement, but it has evolved to encourage ratings on sites that embrace minimum standards of fairness and balance. This is accomplished by having the patient transfer copyright to commentary to the treating physician. If a post is fraudulent or defamatory, the doctor has a tool to enable its removal.*
The initial agreement received much attention—and some criticism. Opponents refer to it as a gag order that impedes free speech. “What are they trying to hide?,” goes the insinuation. “Patients should have a right to know what kind of care they can expect to receive from a doctor,” plead others.
We agree: Patients have a need and a right to know what kind of care they can expect from a physician. But we reject the premise that anonymous, undocumented comments posted on a Web site whose owners are unaccountable for what is said produce this information. We do believe that patients are entirely qualified to provide impressions, but such Web sites are being used as surrogate, supposedly objective measures of quality of care. In that role, the sites have failed.
A physician is not a roofer
Selecting a physician is different from hiring a roofer. First, some measure of the success of medicine depends on the patient. Adherent patients fare better than nonadherent ones. A roofer asks only that the customer pay for services provided. If that roofer asked the customer to pay and, in addition, layer the caulk, the comparison might work, but that’s not the case.
Second, health care is rarely provided by one source. A patient’s care is more often collaborative than the product of an individual. Think how many doctors are involved in treating a patient hospitalized for a few days.
Third, more frightening is that many hospitalized patients cannot even identify their physicians. In a survey released earlier this year, three quarters of patients were unable to name anyone in charge of their care. Worse, of those who provided at least one name, 60% gave an incorrect answer. The survey included more than 2,800 patients.1
Last, it is difficult for a patient to evaluate a physician’s judgment and technical prowess objectively. Patients can offer useful subjective information about many aspects of a physician’s skills, such as ability to communicate, but other areas, such as deep technical skills, are less than clear. With the roofer, it’s easy: Either the roof leaks or it doesn’t. Evaluating the details of a gynecologic oncology procedure—well, that’s another matter.
Until 2008, Angie’s List (www.angieslist.com) was known primarily for its reviews of plumbers, handymen, and other contractors. But in March 2008, the Web site rolled out 40 new categories of service providers—all of them related to health care.
The result: 10,000 physician reviews in the first month.
“We just saw a tremendous amount of interest,” says Mike Rutz, Vice President of Angie’s List Health—so much interest that the company increased the number of health-care categories to 150 shortly thereafter, including one for ObGyn care.
Angie’s List is a pay-to-use service with roughly 1,000,000 members in the United States. It differs from other physician-rating sites in other respects as well: Reviews are not anonymous, and any physician who receives a negative review is given the opportunity to respond. In fact, a physician can sign up, free of charge, to be notified when a review goes up on the site. Although the member’s name is not posted on the individual review, it is recorded so that any disputes can be clarified.
“We do have some doctors who say, ‘These people are not my patients,’” Rutz reports. When this happens, Angie’s List can consult its database and resolve the issue definitively.
How is the physician rated?
A patient gives her physician a grade, ranging from A to F, in eight categories:
- availability
- office environment
- punctuality
- staff friendliness
- bedside manner
- communication
- effectiveness of treatment
- billing and administration.
The eight scores are averaged to yield an overall grade.
When a member seeks information on health-care providers in her locality, she sees only the overall grade at first. She clicks through this screen to view the full report.
Rutz believes that most patients have the expertise to judge effectiveness of treatment. “They absolutely know whether the treatment was effective,” he says. He does concede that “the patient is the most important participant in her health care” and does have an impact on the success or failure of treatment. Angie’s List has no plans to remove effectiveness of treatment from the rating categories.
Physicians can encourage positive reviews
Physicians aren’t penalized for encouraging patients to give them a positive review on Angie’s List—in fact, that strategy is encouraged.
“More information is better,” says Rutz. “The folks that are encouraging reviews are usually the folks providing the best service.”
There is an added bonus to positive reviews: Providers who have the best grades and the most reports rise to the top of the list, gaining prominence on the site.
At the same time, there are mechanisms in place to prevent a health-care or other service provider from “gaming the system,” Rutz adds. It is not acceptable for a physician or a member of his or her staff or family to file a review.
So is Angie’s List good news for physicians?
Jeffrey Segal, MD, does not think so.
The problem is that the site requires paid membership, Dr. Segal, founder of Medical Justice Services in Greensboro, NC, says.
“Because of that, [Angie’s List] will never really accumulate more than a handful of reviews on any given doctor; particularly given the number of free sites. And since the average doctor sees over 1,000 patients a year, we do not believe three or four reviews can ever mean anything substantive.”—JANELLE YATES, SENIOR EDITOR
How rating sites need to change
You may conclude that we oppose online rating of physicians. We do not. We understand the desire to know as much as possible about a provider’s abilities. We merely appeal to online rating sites to adhere to minimum standards that promote a responsible system for both physicians and patients.
Here are our recommendations:
- Ensure that every potential rater who claims to be a patient of a particular physician really is one.
- Require a meaningful number of raters. Given that the average physician sees 1,000 to 2,000 patients in a year, ratings that involve only two or three patients are meaningless. Forty or 50 ratings are another story. Comments could be collected until a threshold number is reached. They could then be published.
- Require patients to stick to areas in which they are expert—namely, subjective impressions. A physician’s personality, staff, and communication skills are fair game. Skill at performing oophorectomy—out of bounds. However, should a patient wish to have the physician’s technical skills reviewed by a trained professional, all the better.
For example, to deputize the patient as a partner in preventing spread of infection and wrong-site surgery, a survey can ask: “Do you recall if the doctor washed her hands before she examined you?” (The answer is informative only if the office asks the patient, upfront, to make this observation.) Another question: “Did the doctor ask you to mark your name on the affected limb before surgery?”
The feedback loop will either confirm great performance or serve as a strong motivator for fast improvement. Both physicians and patients must participate to make the health-care system better.
Even when reviews are positive, the experience of being rated on the Web is a little disconcerting.
Why?
Most reviews are anonymous, and few, if any, patients have the background to fully understand why a particular treatment succeeded—or did not. That’s the view, at least, of two ObGyns interviewed—both of whom serve on the Board of Editors of OBG Management.
Steven R. Goldstein, MD, has been glowingly reviewed on Angie’s List, but still finds the value of online rating dubious. Dr. Goldstein is professor of obstetrics and gynecology at New York University School of Medicine and director of gynecologic ultrasound and co-director of bone densitometry at New York University Medical Center in New York City.
“There’s nothing scientific or objective about it,” he says, particularly when it comes to assessment of complex medical treatment. “Patients can say how they felt about the person,” he adds, but their ability to evaluate the scientific and technical components of treatment is limited.
Barbara S. Levy, MD, agrees. She is medical director of the Women’s Health Center for the Franciscan Health System in Federal Way, Washington.
“I think people can rate their experience,” she says. “I don’t think they can judge training and surgical skill.” Dr. Levy has been rated positively on several Web sites.
Neither physician had read any reviews of their services. Both are “kind of” aware that online rating is increasingly common.
“It’s happening in everything,” Dr. Levy says.
Is there reason to worry about a poor rating?
Dr. Levy believes that anonymous reviews should be prohibited. “Not that a site has to publish the names of reviewers,” she says, “only that whatever agency is allowing them to do the reviewing should be collecting their information to make sure they are legitimate. It’s just due diligence.”
Dr. Levy does believe that patients should have the right to review their experience with physicians. As for what to do about an unfair review, “I think it’s only fair for the doctor to be able to rebut it,” she says.
Dr. Goldstein has a slightly different take on the matter.
“I don’t really have the time or energy to devote to this issue,” he says. “I like to think that patients who come to me do so because of other patients, whom they know fairly well, or other physicians.” If a patient sees one negative Web review and chooses not to use a doctor on that basis, “there isn’t much I can do about it,” he says.
“The most important part of this issue”—online rating of physicians—“is that it is totally unsubstantiated.”—JANELLE YATES, SENIOR EDITOR
What we are doing now
Our company is working with rating sites that share our understanding of the problem and endorse our proposed solutions. Bad information can be worse than no information, we argue! If health care is to be improved, patients and physicians need high-quality information about providers and health-care systems. By injecting fairness and accountability into online ratings, we will all benefit.
CASE RESOLVED
By the time the ObGyn is able to have the woman’s comments removed from the Web site, he lived for more than a year with the innuendo made possible by the Internet.
1. Arora V, Gangireddy S, Mehhrotra A, Ginde R, Tormey M, Meltzer D. Ability of hospitalized patients to identify their inhospital physicians. Arch Intern Med. 2009;169:199-201.
1. Arora V, Gangireddy S, Mehhrotra A, Ginde R, Tormey M, Meltzer D. Ability of hospitalized patients to identify their inhospital physicians. Arch Intern Med. 2009;169:199-201.
UPDATE ON: URINARY INCONTINENCE
The authors report no financial relationships relevant to this article.
Four recent studies enhance our understanding of the benefits, efficacy, and risks of the following interventions in women who have urinary incontinence (UI):
- weight loss. Women who were randomized to intensive weight loss reduced the total number of UI episodes in a week by 47.4%—compared with 28.1% in the group randomized to a structured educational program.
- midurethral slings. Treatment of stress UI arising from intrinsic sphincter deficiency was more successful in women randomized to tension-free vaginal tape (TVT) than in women assigned to transobturator tape (TOT). In the first group, urodynamically confirmed stress UI was present in 21% of subjects after treatment, compared with 45% in the TOT group.
- botulinum toxin type A (Botox) injection. Women who had refractory urge incontinence were likely to improve significantly after injection of Botox. Sixty percent of women treated with Botox reported a reduction in incontinence after treatment, with a median response of 373 days, compared with 62 days in the placebo group.
- sacral neuromodulation. The mean number of urge incontinence episodes decreased from 9.6 to 3.9, and the mean number of voids per day decreased from 19.3 to 14.8 in a 5-year follow-up study.
Despite the promise of these findings, all of the studies had limitations, and several identified risks associated with the intervention. These limitations and risks are detailed in the articles that follow.
Urinary incontinence is no small problem. It affects more than 13 million women in the United States alone, and costs more than $20 billion annually in direct health-care costs.1
Despite the high prevalence of urinary incontinence, women are often reluctant to discuss symptoms with their physician. As a result, the condition remains undiagnosed or undertreated in many women.2
The most common types of urinary incontinence include:
- stress incontinence – leakage upon effort, exertion, or increased abdominal pressure
- urge incontinence – leakage accompanied by, or immediately preceded by, urgency
- mixed incontinence – leakage with urgency as well as effort, exertion, or increased abdominal pressure.
Another common problem is overactive bladder syndrome, which involves urgency with or without leakage, and usually increased frequency and nocturia as well.3
Although the midurethral sling revolutionized the treatment of stress urinary incontinence, most women who have incontinence experience mixed symptoms, making it a more challenging condition for the general-practice ObGyn to treat successfully. Furthermore, traditional therapies such as behavior modification, pelvic floor exercise, and medication have had only modest success in certain patient populations.
Weight loss can reduce urinary incontinence
in overweight and obese women
Subak LL, Wing R, Smith West D, et al. Weight loss to treat urinary incontinence in overweight and obese women. N Engl J Med. 2009;360:481–490.
Obesity increases intra-abdominal pressure, thereby exerting added force on the bladder, urethra, and pelvic floor and potentially exacerbating urinary incontinence (UI). It has been hypothesized that weight reduction reduces these forces and improves incontinence.
This randomized, clinical trial of 338 women showed that weight loss does have an effect on UI. Investigators compared an intensive 6-month weight loss program—designed to prompt a weight loss of 7% to 9% of body weight—with a structured educational program. The primary outcome was the percentage of change in the number of UI episodes reported in a 7-day voiding diary at 6 months.
After 6 months and a mean weight loss of 8% of baseline body weight, the women in the intensive weight loss group experienced a mean decrease of 47.4% in the total number of UI episodes in a week. Compare this with a mean weight loss of 1.6% (P<.001) and a mean decrease of 28.1% in UI episodes in the control group (P=.01). The reduction in the total number of UI episodes was primarily attributed to a reduction of 57.6% in stress-induced UI in the intensive weight loss group, compared with a reduction of 32.7% in the control group (P=.02).
Women in the weight loss program also perceived incontinence to be less of a problem and reported greater satisfaction with the change in their incontinence at 6 months than did women in the control group (P<.001).
Details of the trial
Women were eligible to participate if they reported 10 or more episodes of UI over 7 days, were at least 30 years old, and had a body mass index (BMI) of 25 to 50 at baseline (normal is 19 to 24). In addition, they had to agree not to initiate any new treatments for UI or weight loss during the 6-month study period.
Subjects were randomized in a 2:1 ratio, with 226 women assigned to the intensive weight loss program and 112 assigned to the structured educational program. Baseline characteristics were similar in both groups, with a mean age of 53±11 years, mean BMI of 36±6 kg/m3, and total mean number of UI episodes of 24±18 per week.
Strengths and limitations
Strengths of this study include the large and varied study population. One important limitation, however, is the fact that the primary outcome measure was based on self-reported UI episodes. Because participants were not blinded to their treatment assignment, bias in self-reporting may have been present. In addition, subjects were selected because of their potential to adhere to the rigorous study protocol.
As the prevalence of obesity reaches pandemic level, it’s imperative that medical science continue to develop novel methods with which we can help patients achieve ideal body weight.
The findings of this study contribute to the growing body of medical literature—across specialties—demonstrating that weight loss can significantly improve the general health of patients, and that it should be part of the first-line treatment for overweight and obese women who complain of UI.
TVT is more effective than TOT
for intrinsic sphincter deficiency
Schierlitz L, Dwyer PL, Rosamilia A, et al. Effectiveness of tension-free vaginal tape compared with transobturator tape in women with stress urinary incontinence and intrinsic sphincter deficiency: a randomized controlled trial. Obstet Gynecol. 2008;112:1253–1261.
Many experts consider intrinsic sphincter deficiency (ISD) to be a severe form of stress incontinence. Earlier studies suggested that women who had stress incontinence complicated by ISD had a lower success rate after certain surgical procedures than did women who had stress incontinence alone.
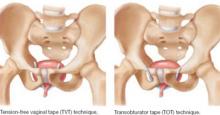
This randomized trial compared tension-free vaginal tape (TVT) with transobturator tape (TOT) in the treatment of stress incontinence with ISD ( FIGURE 1 ). The primary outcome measure was the presence of urodynamically confirmed stress incontinence 6 months after surgery.
At that 6-month mark, stress incontinence was present in 14 of 67 subjects (21%) in the TVT group, compared with 32 of 71 subjects (45%) in the TOT group (P=.004). Nine patients in the TOT group requested a repeat surgical procedure, compared with none in the TVT group.
Although the study was not powered to detect a difference in postoperative complications, there were six bladder perforations in the TVT group and none in the TOT group.
FIGURE 1 TVT and TOT trace different routes
Details of the trial
Women were selected for the trial on the basis of urodynamic parameters and recruited from two academic centers. ISD was defined as maximal urethral closure pressure below 20 cm H2O or Valsalva leak-point pressure less than 60 cm H2O. Subjects in the two groups had similar baseline characteristics.
Surgeons were required to have independently performed at least 15 surgical midurethral sling procedures before the study began.
In addition to placement of TVT or TOT, approximately one third of subjects underwent concomitant prolapse surgery. Postoperatively, subjects were assessed at 6 weeks, 6 months, and 12 months, with repeat urodynamic testing performed at 6 months. Analysis of data was based on intention to treat.
Only 138 of 164 women completed repeat urodynamic testing 6 months after surgery. Seventeen women declined testing, claiming to be “cured,” and nine women withdrew from the study or were lost to follow-up.
Short follow-up was a limitation
The definition of treatment failure as persistent, urodynamically confirmed stress incontinence was another shortcoming of the trial. In addition, the 17 subjects who declined repeat postoperative testing were classified as “cured,” potentially biasing the results.
TVT may be the preferred surgical option for women who have urodynamically confirmed stress incontinence complicated by intrinsic sphincter deficiency (ISD)—on the basis of the data gathered by these researchers. Longer follow-up is needed, however, to determine the long-term, clinical efficacy of midurethral slings in women who have ISD.
Accumulation of more data from future studies will better equip ObGyns to customize surgical treatment options to individual clinical parameters and reduce the risk of surgical failure.
Is Botox a panacea for refractory urge incontinence?
Brubaker L, Richter HE, Visco A, et al: Pelvic Floor Disorders Network. Refractory idiopathic urge urinary incontinence and botulinum A injection. J Urol. 2008;180:217–222.
Women who fail medical management of urge incontinence have few other options. This multicenter, randomized, double-blind, placebo-controlled trial suggests that there may one day be an effective alternative. Investigators examined the safety and efficacy of botulinum toxin type A (Botox) for the treatment of refractory idiopathic urge incontinence in 43 women—28 of them randomized to injection of 200 U of Botox and 15 to placebo.
Sixty percent of subjects in the Botox arm reported an improvement in symptoms, with a median response of 373 days, compared with 62 days in the placebo arm (P<.0001). Moreover, in the Botox arm, women perceived greater improvement in symptom control and a decrease in the number of self-reported incontinence episodes, compared with the placebo group (P<.0001).
However, 12 of 28 patients (43%) who received Botox developed elevated postvoid residuals (i.e., retention of more than 200 mL of urine), and enrollment was halted for this reason. Median time to initiate intermittent self-catheterization was 30 days, and intermittent self-catheterization lasted a median of 60 days. Nine of the 12 subjects who required self-catheterization developed a urinary tract infection.
Details of the trial
This study was conducted by the Pelvic Floor Disorders Network and sponsored by the National Institute of Child Health and Human Development. To be eligible for the trial, women had to have been diagnosed with refractory urge incontinence, which was defined as persistent symptoms after failing at least two first-line therapies such as anticholinergic medications and behavioral therapy. Also required was documented evidence of detrusor overactivity on urodynamic studies or at least six episodes of urge-related incontinence in a 3-day bladder diary. Investigators determined that a sample size of 210 subjects was needed to test a 50% efficacy rate for Botox, compared with 30% for placebo, at 6 months.
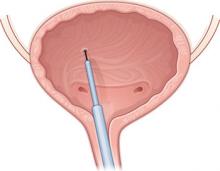
Baseline characteristics were similar between groups. A blinded physician used a cystoscope to inject the agents into the detrusor muscle over the posterior bladder wall ( FIGURE 2 ). Subjects also received an antibiotic before the procedure and for 3 additional days.
The primary outcome measure was treatment failure, defined as the return of symptoms measured at least 2 months after Botox injection or any change in medical therapy.
Enrollment was halted after interval analysis revealed a significantly higher rate of voiding dysfunction in the Botox arm, necessitating intermittent self-catheterization and associated urinary tract infections.
FIGURE 2 Intradetrusor Botox injection
In the trial by Brubaker and colleagues, 200 U of Botox was injected into the detrusor muscle on the posterior bladder wall to treat refractory urge incontinence in 28 women—60% of whom reported improvement.
Is a postvoid residual clinically significant?
Some experts questioned the clinical significance of the elevated postvoid residual reported in this trial, arguing that it was temporary and that many patients were asymptomatic.
Further study is needed to determine the optimal dosing of Botox and a management strategy for postprocedure voiding dysfunction.
When one of your patients considers cystoscopic intradetrusor Botox therapy for refractory urge incontinence, you should:
- counsel her extensively that its use here is not FDA-approved
- caution her that she may need to perform intermittent self-catheterization after injection
- advise her that the procedure and the medication are likely not covered by her health insurance and will be out-of-pocket expenses.
Much-needed and much-anticipated clinical trials are under way with the aim of obtaining FDA approval of Botox for urge incontinence.
At the Cleveland Clinic, we currently offer Botox for refractory urge incontinence.
InterStim therapy may become the gold standard for refractory overactive bladder syndrome
van Kerrebroeck PE, van Voskuilen AC, Heesakkers JP, et al. Results of sacral neuromodulation therapy for urinary voiding dysfunction: outcomes of a prospective, worldwide clinical study. J Urol. 2007;178:2029–2034.
Sacral nerve stimulation has been approved for use in patients with refractory voiding dysfunction since 1997. This prospective, worldwide, follow-up study sought to determine the long-term efficacy and safety of sacral neuromodulation for the treatment of refractory urgency, frequency, urge incontinence, and nonobstructive urinary retention.
After 5 years of follow-up, 68% of subjects who had urge incontinence, 56% who had urgency and frequency, and 71% who had nonobstructive urinary retention reported a degree of improvement of 50% or more in their symptoms (relative to baseline). The mean number of urge incontinence episodes decreased from 9.6 to 3.9, and mean voids per day decreased from 19.3 to 14.8 at 5 years (P<.001).
Details of the trial
Eligible patients were previously enrolled in a randomized, clinical trial investigating the efficacy of InterStim. Of the 23 sites that participated in the original study, only 17 elected to participate in the follow-up trial.
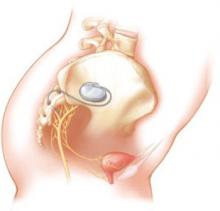
One hundred fifty-two subjects were enrolled—129 of them crossing over from the original study and 23 newly recruited and implanted with the InterStim device ( FIGURE 3 ). Investigators reviewed data from follow-up visits, self-reported symptoms obtained from voiding diaries collected annually for 5 years, and descriptive summaries of adverse events related to the sacral nerve stimulator.
One-year data were available on 138 subjects, and 5-year data were available on 105. Of the 47 participants who did not participate in 5-year follow-up, 16 had the InterStim device removed.
FIGURE 3 Aim of InterStim therapy is to regulate brain–bladder signals
The InterStim device sends a mild electrical impulse through a lead to the sacral nerves to influence the bladder and surrounding muscles. Sacral nerve stimulation helps regulate interaction between the brain and bladder and reduce voiding dysfunction.
Additional surgery was performed in half of patients
who had an adverse event
No life-threatening or irreversible adverse events were reported during the 5 years of follow-up. However, 221 adverse events occurred, of which 110 required a minor surgical procedure—in some cases, more than one. The most commonly reported adverse event was new pain or pain at the implantation site (46.1%). The most common surgical procedure was device exchange (23.7%).
Because many patients who suffer from overactive bladder syndrome fail to respond to conservative therapies, such as behavior modification and pharmacotherapy, novel alternatives are needed. This study:
- contributes to our understanding of sacral nerve stimulation
- provides much needed data on the long-term safety and efficacy of InterStim therapy
- proposes that InterStim therapy is safe and efficacious for women who have refractory overactive bladder syndrome
- suggests that it may become the gold standard for treatment of this condition.
At the Cleveland Clinic, we offer sacral nerve stimulation for refractory overactive bladder and nonobstructive urinary retention.
1. Hu TW, Wagner TH, Bentkover JD, Leblanc K, Zhou SZ, Hunt T. Costs of urinary incontinence and overactive bladder in the United States: a comparative study. Urology. 2004;63:461-465.
2. Mardon RE, Halim S, Pawlson LG, Haffer SC. Management of urinary incontinence in Medicare managed care beneficiaries: results from the 2004 Medicare Health Outcomes Survey. Arch Intern Med. 2006;166:1128-1133.
3. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167-178.
The authors report no financial relationships relevant to this article.
Four recent studies enhance our understanding of the benefits, efficacy, and risks of the following interventions in women who have urinary incontinence (UI):
- weight loss. Women who were randomized to intensive weight loss reduced the total number of UI episodes in a week by 47.4%—compared with 28.1% in the group randomized to a structured educational program.
- midurethral slings. Treatment of stress UI arising from intrinsic sphincter deficiency was more successful in women randomized to tension-free vaginal tape (TVT) than in women assigned to transobturator tape (TOT). In the first group, urodynamically confirmed stress UI was present in 21% of subjects after treatment, compared with 45% in the TOT group.
- botulinum toxin type A (Botox) injection. Women who had refractory urge incontinence were likely to improve significantly after injection of Botox. Sixty percent of women treated with Botox reported a reduction in incontinence after treatment, with a median response of 373 days, compared with 62 days in the placebo group.
- sacral neuromodulation. The mean number of urge incontinence episodes decreased from 9.6 to 3.9, and the mean number of voids per day decreased from 19.3 to 14.8 in a 5-year follow-up study.
Despite the promise of these findings, all of the studies had limitations, and several identified risks associated with the intervention. These limitations and risks are detailed in the articles that follow.
Urinary incontinence is no small problem. It affects more than 13 million women in the United States alone, and costs more than $20 billion annually in direct health-care costs.1
Despite the high prevalence of urinary incontinence, women are often reluctant to discuss symptoms with their physician. As a result, the condition remains undiagnosed or undertreated in many women.2
The most common types of urinary incontinence include:
- stress incontinence – leakage upon effort, exertion, or increased abdominal pressure
- urge incontinence – leakage accompanied by, or immediately preceded by, urgency
- mixed incontinence – leakage with urgency as well as effort, exertion, or increased abdominal pressure.
Another common problem is overactive bladder syndrome, which involves urgency with or without leakage, and usually increased frequency and nocturia as well.3
Although the midurethral sling revolutionized the treatment of stress urinary incontinence, most women who have incontinence experience mixed symptoms, making it a more challenging condition for the general-practice ObGyn to treat successfully. Furthermore, traditional therapies such as behavior modification, pelvic floor exercise, and medication have had only modest success in certain patient populations.
Weight loss can reduce urinary incontinence
in overweight and obese women
Subak LL, Wing R, Smith West D, et al. Weight loss to treat urinary incontinence in overweight and obese women. N Engl J Med. 2009;360:481–490.
Obesity increases intra-abdominal pressure, thereby exerting added force on the bladder, urethra, and pelvic floor and potentially exacerbating urinary incontinence (UI). It has been hypothesized that weight reduction reduces these forces and improves incontinence.
This randomized, clinical trial of 338 women showed that weight loss does have an effect on UI. Investigators compared an intensive 6-month weight loss program—designed to prompt a weight loss of 7% to 9% of body weight—with a structured educational program. The primary outcome was the percentage of change in the number of UI episodes reported in a 7-day voiding diary at 6 months.
After 6 months and a mean weight loss of 8% of baseline body weight, the women in the intensive weight loss group experienced a mean decrease of 47.4% in the total number of UI episodes in a week. Compare this with a mean weight loss of 1.6% (P<.001) and a mean decrease of 28.1% in UI episodes in the control group (P=.01). The reduction in the total number of UI episodes was primarily attributed to a reduction of 57.6% in stress-induced UI in the intensive weight loss group, compared with a reduction of 32.7% in the control group (P=.02).
Women in the weight loss program also perceived incontinence to be less of a problem and reported greater satisfaction with the change in their incontinence at 6 months than did women in the control group (P<.001).
Details of the trial
Women were eligible to participate if they reported 10 or more episodes of UI over 7 days, were at least 30 years old, and had a body mass index (BMI) of 25 to 50 at baseline (normal is 19 to 24). In addition, they had to agree not to initiate any new treatments for UI or weight loss during the 6-month study period.
Subjects were randomized in a 2:1 ratio, with 226 women assigned to the intensive weight loss program and 112 assigned to the structured educational program. Baseline characteristics were similar in both groups, with a mean age of 53±11 years, mean BMI of 36±6 kg/m3, and total mean number of UI episodes of 24±18 per week.
Strengths and limitations
Strengths of this study include the large and varied study population. One important limitation, however, is the fact that the primary outcome measure was based on self-reported UI episodes. Because participants were not blinded to their treatment assignment, bias in self-reporting may have been present. In addition, subjects were selected because of their potential to adhere to the rigorous study protocol.
As the prevalence of obesity reaches pandemic level, it’s imperative that medical science continue to develop novel methods with which we can help patients achieve ideal body weight.
The findings of this study contribute to the growing body of medical literature—across specialties—demonstrating that weight loss can significantly improve the general health of patients, and that it should be part of the first-line treatment for overweight and obese women who complain of UI.
TVT is more effective than TOT
for intrinsic sphincter deficiency
Schierlitz L, Dwyer PL, Rosamilia A, et al. Effectiveness of tension-free vaginal tape compared with transobturator tape in women with stress urinary incontinence and intrinsic sphincter deficiency: a randomized controlled trial. Obstet Gynecol. 2008;112:1253–1261.
Many experts consider intrinsic sphincter deficiency (ISD) to be a severe form of stress incontinence. Earlier studies suggested that women who had stress incontinence complicated by ISD had a lower success rate after certain surgical procedures than did women who had stress incontinence alone.
This randomized trial compared tension-free vaginal tape (TVT) with transobturator tape (TOT) in the treatment of stress incontinence with ISD ( FIGURE 1 ). The primary outcome measure was the presence of urodynamically confirmed stress incontinence 6 months after surgery.
At that 6-month mark, stress incontinence was present in 14 of 67 subjects (21%) in the TVT group, compared with 32 of 71 subjects (45%) in the TOT group (P=.004). Nine patients in the TOT group requested a repeat surgical procedure, compared with none in the TVT group.
Although the study was not powered to detect a difference in postoperative complications, there were six bladder perforations in the TVT group and none in the TOT group.
FIGURE 1 TVT and TOT trace different routes
Details of the trial
Women were selected for the trial on the basis of urodynamic parameters and recruited from two academic centers. ISD was defined as maximal urethral closure pressure below 20 cm H2O or Valsalva leak-point pressure less than 60 cm H2O. Subjects in the two groups had similar baseline characteristics.
Surgeons were required to have independently performed at least 15 surgical midurethral sling procedures before the study began.
In addition to placement of TVT or TOT, approximately one third of subjects underwent concomitant prolapse surgery. Postoperatively, subjects were assessed at 6 weeks, 6 months, and 12 months, with repeat urodynamic testing performed at 6 months. Analysis of data was based on intention to treat.
Only 138 of 164 women completed repeat urodynamic testing 6 months after surgery. Seventeen women declined testing, claiming to be “cured,” and nine women withdrew from the study or were lost to follow-up.
Short follow-up was a limitation
The definition of treatment failure as persistent, urodynamically confirmed stress incontinence was another shortcoming of the trial. In addition, the 17 subjects who declined repeat postoperative testing were classified as “cured,” potentially biasing the results.
TVT may be the preferred surgical option for women who have urodynamically confirmed stress incontinence complicated by intrinsic sphincter deficiency (ISD)—on the basis of the data gathered by these researchers. Longer follow-up is needed, however, to determine the long-term, clinical efficacy of midurethral slings in women who have ISD.
Accumulation of more data from future studies will better equip ObGyns to customize surgical treatment options to individual clinical parameters and reduce the risk of surgical failure.
Is Botox a panacea for refractory urge incontinence?
Brubaker L, Richter HE, Visco A, et al: Pelvic Floor Disorders Network. Refractory idiopathic urge urinary incontinence and botulinum A injection. J Urol. 2008;180:217–222.
Women who fail medical management of urge incontinence have few other options. This multicenter, randomized, double-blind, placebo-controlled trial suggests that there may one day be an effective alternative. Investigators examined the safety and efficacy of botulinum toxin type A (Botox) for the treatment of refractory idiopathic urge incontinence in 43 women—28 of them randomized to injection of 200 U of Botox and 15 to placebo.
Sixty percent of subjects in the Botox arm reported an improvement in symptoms, with a median response of 373 days, compared with 62 days in the placebo arm (P<.0001). Moreover, in the Botox arm, women perceived greater improvement in symptom control and a decrease in the number of self-reported incontinence episodes, compared with the placebo group (P<.0001).
However, 12 of 28 patients (43%) who received Botox developed elevated postvoid residuals (i.e., retention of more than 200 mL of urine), and enrollment was halted for this reason. Median time to initiate intermittent self-catheterization was 30 days, and intermittent self-catheterization lasted a median of 60 days. Nine of the 12 subjects who required self-catheterization developed a urinary tract infection.
Details of the trial
This study was conducted by the Pelvic Floor Disorders Network and sponsored by the National Institute of Child Health and Human Development. To be eligible for the trial, women had to have been diagnosed with refractory urge incontinence, which was defined as persistent symptoms after failing at least two first-line therapies such as anticholinergic medications and behavioral therapy. Also required was documented evidence of detrusor overactivity on urodynamic studies or at least six episodes of urge-related incontinence in a 3-day bladder diary. Investigators determined that a sample size of 210 subjects was needed to test a 50% efficacy rate for Botox, compared with 30% for placebo, at 6 months.
Baseline characteristics were similar between groups. A blinded physician used a cystoscope to inject the agents into the detrusor muscle over the posterior bladder wall ( FIGURE 2 ). Subjects also received an antibiotic before the procedure and for 3 additional days.
The primary outcome measure was treatment failure, defined as the return of symptoms measured at least 2 months after Botox injection or any change in medical therapy.
Enrollment was halted after interval analysis revealed a significantly higher rate of voiding dysfunction in the Botox arm, necessitating intermittent self-catheterization and associated urinary tract infections.
FIGURE 2 Intradetrusor Botox injection
In the trial by Brubaker and colleagues, 200 U of Botox was injected into the detrusor muscle on the posterior bladder wall to treat refractory urge incontinence in 28 women—60% of whom reported improvement.
Is a postvoid residual clinically significant?
Some experts questioned the clinical significance of the elevated postvoid residual reported in this trial, arguing that it was temporary and that many patients were asymptomatic.
Further study is needed to determine the optimal dosing of Botox and a management strategy for postprocedure voiding dysfunction.
When one of your patients considers cystoscopic intradetrusor Botox therapy for refractory urge incontinence, you should:
- counsel her extensively that its use here is not FDA-approved
- caution her that she may need to perform intermittent self-catheterization after injection
- advise her that the procedure and the medication are likely not covered by her health insurance and will be out-of-pocket expenses.
Much-needed and much-anticipated clinical trials are under way with the aim of obtaining FDA approval of Botox for urge incontinence.
At the Cleveland Clinic, we currently offer Botox for refractory urge incontinence.
InterStim therapy may become the gold standard for refractory overactive bladder syndrome
van Kerrebroeck PE, van Voskuilen AC, Heesakkers JP, et al. Results of sacral neuromodulation therapy for urinary voiding dysfunction: outcomes of a prospective, worldwide clinical study. J Urol. 2007;178:2029–2034.
Sacral nerve stimulation has been approved for use in patients with refractory voiding dysfunction since 1997. This prospective, worldwide, follow-up study sought to determine the long-term efficacy and safety of sacral neuromodulation for the treatment of refractory urgency, frequency, urge incontinence, and nonobstructive urinary retention.
After 5 years of follow-up, 68% of subjects who had urge incontinence, 56% who had urgency and frequency, and 71% who had nonobstructive urinary retention reported a degree of improvement of 50% or more in their symptoms (relative to baseline). The mean number of urge incontinence episodes decreased from 9.6 to 3.9, and mean voids per day decreased from 19.3 to 14.8 at 5 years (P<.001).
Details of the trial
Eligible patients were previously enrolled in a randomized, clinical trial investigating the efficacy of InterStim. Of the 23 sites that participated in the original study, only 17 elected to participate in the follow-up trial.
One hundred fifty-two subjects were enrolled—129 of them crossing over from the original study and 23 newly recruited and implanted with the InterStim device ( FIGURE 3 ). Investigators reviewed data from follow-up visits, self-reported symptoms obtained from voiding diaries collected annually for 5 years, and descriptive summaries of adverse events related to the sacral nerve stimulator.
One-year data were available on 138 subjects, and 5-year data were available on 105. Of the 47 participants who did not participate in 5-year follow-up, 16 had the InterStim device removed.
FIGURE 3 Aim of InterStim therapy is to regulate brain–bladder signals
The InterStim device sends a mild electrical impulse through a lead to the sacral nerves to influence the bladder and surrounding muscles. Sacral nerve stimulation helps regulate interaction between the brain and bladder and reduce voiding dysfunction.
Additional surgery was performed in half of patients
who had an adverse event
No life-threatening or irreversible adverse events were reported during the 5 years of follow-up. However, 221 adverse events occurred, of which 110 required a minor surgical procedure—in some cases, more than one. The most commonly reported adverse event was new pain or pain at the implantation site (46.1%). The most common surgical procedure was device exchange (23.7%).
Because many patients who suffer from overactive bladder syndrome fail to respond to conservative therapies, such as behavior modification and pharmacotherapy, novel alternatives are needed. This study:
- contributes to our understanding of sacral nerve stimulation
- provides much needed data on the long-term safety and efficacy of InterStim therapy
- proposes that InterStim therapy is safe and efficacious for women who have refractory overactive bladder syndrome
- suggests that it may become the gold standard for treatment of this condition.
At the Cleveland Clinic, we offer sacral nerve stimulation for refractory overactive bladder and nonobstructive urinary retention.
The authors report no financial relationships relevant to this article.
Four recent studies enhance our understanding of the benefits, efficacy, and risks of the following interventions in women who have urinary incontinence (UI):
- weight loss. Women who were randomized to intensive weight loss reduced the total number of UI episodes in a week by 47.4%—compared with 28.1% in the group randomized to a structured educational program.
- midurethral slings. Treatment of stress UI arising from intrinsic sphincter deficiency was more successful in women randomized to tension-free vaginal tape (TVT) than in women assigned to transobturator tape (TOT). In the first group, urodynamically confirmed stress UI was present in 21% of subjects after treatment, compared with 45% in the TOT group.
- botulinum toxin type A (Botox) injection. Women who had refractory urge incontinence were likely to improve significantly after injection of Botox. Sixty percent of women treated with Botox reported a reduction in incontinence after treatment, with a median response of 373 days, compared with 62 days in the placebo group.
- sacral neuromodulation. The mean number of urge incontinence episodes decreased from 9.6 to 3.9, and the mean number of voids per day decreased from 19.3 to 14.8 in a 5-year follow-up study.
Despite the promise of these findings, all of the studies had limitations, and several identified risks associated with the intervention. These limitations and risks are detailed in the articles that follow.
Urinary incontinence is no small problem. It affects more than 13 million women in the United States alone, and costs more than $20 billion annually in direct health-care costs.1
Despite the high prevalence of urinary incontinence, women are often reluctant to discuss symptoms with their physician. As a result, the condition remains undiagnosed or undertreated in many women.2
The most common types of urinary incontinence include:
- stress incontinence – leakage upon effort, exertion, or increased abdominal pressure
- urge incontinence – leakage accompanied by, or immediately preceded by, urgency
- mixed incontinence – leakage with urgency as well as effort, exertion, or increased abdominal pressure.
Another common problem is overactive bladder syndrome, which involves urgency with or without leakage, and usually increased frequency and nocturia as well.3
Although the midurethral sling revolutionized the treatment of stress urinary incontinence, most women who have incontinence experience mixed symptoms, making it a more challenging condition for the general-practice ObGyn to treat successfully. Furthermore, traditional therapies such as behavior modification, pelvic floor exercise, and medication have had only modest success in certain patient populations.
Weight loss can reduce urinary incontinence
in overweight and obese women
Subak LL, Wing R, Smith West D, et al. Weight loss to treat urinary incontinence in overweight and obese women. N Engl J Med. 2009;360:481–490.
Obesity increases intra-abdominal pressure, thereby exerting added force on the bladder, urethra, and pelvic floor and potentially exacerbating urinary incontinence (UI). It has been hypothesized that weight reduction reduces these forces and improves incontinence.
This randomized, clinical trial of 338 women showed that weight loss does have an effect on UI. Investigators compared an intensive 6-month weight loss program—designed to prompt a weight loss of 7% to 9% of body weight—with a structured educational program. The primary outcome was the percentage of change in the number of UI episodes reported in a 7-day voiding diary at 6 months.
After 6 months and a mean weight loss of 8% of baseline body weight, the women in the intensive weight loss group experienced a mean decrease of 47.4% in the total number of UI episodes in a week. Compare this with a mean weight loss of 1.6% (P<.001) and a mean decrease of 28.1% in UI episodes in the control group (P=.01). The reduction in the total number of UI episodes was primarily attributed to a reduction of 57.6% in stress-induced UI in the intensive weight loss group, compared with a reduction of 32.7% in the control group (P=.02).
Women in the weight loss program also perceived incontinence to be less of a problem and reported greater satisfaction with the change in their incontinence at 6 months than did women in the control group (P<.001).
Details of the trial
Women were eligible to participate if they reported 10 or more episodes of UI over 7 days, were at least 30 years old, and had a body mass index (BMI) of 25 to 50 at baseline (normal is 19 to 24). In addition, they had to agree not to initiate any new treatments for UI or weight loss during the 6-month study period.
Subjects were randomized in a 2:1 ratio, with 226 women assigned to the intensive weight loss program and 112 assigned to the structured educational program. Baseline characteristics were similar in both groups, with a mean age of 53±11 years, mean BMI of 36±6 kg/m3, and total mean number of UI episodes of 24±18 per week.
Strengths and limitations
Strengths of this study include the large and varied study population. One important limitation, however, is the fact that the primary outcome measure was based on self-reported UI episodes. Because participants were not blinded to their treatment assignment, bias in self-reporting may have been present. In addition, subjects were selected because of their potential to adhere to the rigorous study protocol.
As the prevalence of obesity reaches pandemic level, it’s imperative that medical science continue to develop novel methods with which we can help patients achieve ideal body weight.
The findings of this study contribute to the growing body of medical literature—across specialties—demonstrating that weight loss can significantly improve the general health of patients, and that it should be part of the first-line treatment for overweight and obese women who complain of UI.
TVT is more effective than TOT
for intrinsic sphincter deficiency
Schierlitz L, Dwyer PL, Rosamilia A, et al. Effectiveness of tension-free vaginal tape compared with transobturator tape in women with stress urinary incontinence and intrinsic sphincter deficiency: a randomized controlled trial. Obstet Gynecol. 2008;112:1253–1261.
Many experts consider intrinsic sphincter deficiency (ISD) to be a severe form of stress incontinence. Earlier studies suggested that women who had stress incontinence complicated by ISD had a lower success rate after certain surgical procedures than did women who had stress incontinence alone.
This randomized trial compared tension-free vaginal tape (TVT) with transobturator tape (TOT) in the treatment of stress incontinence with ISD ( FIGURE 1 ). The primary outcome measure was the presence of urodynamically confirmed stress incontinence 6 months after surgery.
At that 6-month mark, stress incontinence was present in 14 of 67 subjects (21%) in the TVT group, compared with 32 of 71 subjects (45%) in the TOT group (P=.004). Nine patients in the TOT group requested a repeat surgical procedure, compared with none in the TVT group.
Although the study was not powered to detect a difference in postoperative complications, there were six bladder perforations in the TVT group and none in the TOT group.
FIGURE 1 TVT and TOT trace different routes
Details of the trial
Women were selected for the trial on the basis of urodynamic parameters and recruited from two academic centers. ISD was defined as maximal urethral closure pressure below 20 cm H2O or Valsalva leak-point pressure less than 60 cm H2O. Subjects in the two groups had similar baseline characteristics.
Surgeons were required to have independently performed at least 15 surgical midurethral sling procedures before the study began.
In addition to placement of TVT or TOT, approximately one third of subjects underwent concomitant prolapse surgery. Postoperatively, subjects were assessed at 6 weeks, 6 months, and 12 months, with repeat urodynamic testing performed at 6 months. Analysis of data was based on intention to treat.
Only 138 of 164 women completed repeat urodynamic testing 6 months after surgery. Seventeen women declined testing, claiming to be “cured,” and nine women withdrew from the study or were lost to follow-up.
Short follow-up was a limitation
The definition of treatment failure as persistent, urodynamically confirmed stress incontinence was another shortcoming of the trial. In addition, the 17 subjects who declined repeat postoperative testing were classified as “cured,” potentially biasing the results.
TVT may be the preferred surgical option for women who have urodynamically confirmed stress incontinence complicated by intrinsic sphincter deficiency (ISD)—on the basis of the data gathered by these researchers. Longer follow-up is needed, however, to determine the long-term, clinical efficacy of midurethral slings in women who have ISD.
Accumulation of more data from future studies will better equip ObGyns to customize surgical treatment options to individual clinical parameters and reduce the risk of surgical failure.
Is Botox a panacea for refractory urge incontinence?
Brubaker L, Richter HE, Visco A, et al: Pelvic Floor Disorders Network. Refractory idiopathic urge urinary incontinence and botulinum A injection. J Urol. 2008;180:217–222.
Women who fail medical management of urge incontinence have few other options. This multicenter, randomized, double-blind, placebo-controlled trial suggests that there may one day be an effective alternative. Investigators examined the safety and efficacy of botulinum toxin type A (Botox) for the treatment of refractory idiopathic urge incontinence in 43 women—28 of them randomized to injection of 200 U of Botox and 15 to placebo.
Sixty percent of subjects in the Botox arm reported an improvement in symptoms, with a median response of 373 days, compared with 62 days in the placebo arm (P<.0001). Moreover, in the Botox arm, women perceived greater improvement in symptom control and a decrease in the number of self-reported incontinence episodes, compared with the placebo group (P<.0001).
However, 12 of 28 patients (43%) who received Botox developed elevated postvoid residuals (i.e., retention of more than 200 mL of urine), and enrollment was halted for this reason. Median time to initiate intermittent self-catheterization was 30 days, and intermittent self-catheterization lasted a median of 60 days. Nine of the 12 subjects who required self-catheterization developed a urinary tract infection.
Details of the trial
This study was conducted by the Pelvic Floor Disorders Network and sponsored by the National Institute of Child Health and Human Development. To be eligible for the trial, women had to have been diagnosed with refractory urge incontinence, which was defined as persistent symptoms after failing at least two first-line therapies such as anticholinergic medications and behavioral therapy. Also required was documented evidence of detrusor overactivity on urodynamic studies or at least six episodes of urge-related incontinence in a 3-day bladder diary. Investigators determined that a sample size of 210 subjects was needed to test a 50% efficacy rate for Botox, compared with 30% for placebo, at 6 months.
Baseline characteristics were similar between groups. A blinded physician used a cystoscope to inject the agents into the detrusor muscle over the posterior bladder wall ( FIGURE 2 ). Subjects also received an antibiotic before the procedure and for 3 additional days.
The primary outcome measure was treatment failure, defined as the return of symptoms measured at least 2 months after Botox injection or any change in medical therapy.
Enrollment was halted after interval analysis revealed a significantly higher rate of voiding dysfunction in the Botox arm, necessitating intermittent self-catheterization and associated urinary tract infections.
FIGURE 2 Intradetrusor Botox injection
In the trial by Brubaker and colleagues, 200 U of Botox was injected into the detrusor muscle on the posterior bladder wall to treat refractory urge incontinence in 28 women—60% of whom reported improvement.
Is a postvoid residual clinically significant?
Some experts questioned the clinical significance of the elevated postvoid residual reported in this trial, arguing that it was temporary and that many patients were asymptomatic.
Further study is needed to determine the optimal dosing of Botox and a management strategy for postprocedure voiding dysfunction.
When one of your patients considers cystoscopic intradetrusor Botox therapy for refractory urge incontinence, you should:
- counsel her extensively that its use here is not FDA-approved
- caution her that she may need to perform intermittent self-catheterization after injection
- advise her that the procedure and the medication are likely not covered by her health insurance and will be out-of-pocket expenses.
Much-needed and much-anticipated clinical trials are under way with the aim of obtaining FDA approval of Botox for urge incontinence.
At the Cleveland Clinic, we currently offer Botox for refractory urge incontinence.
InterStim therapy may become the gold standard for refractory overactive bladder syndrome
van Kerrebroeck PE, van Voskuilen AC, Heesakkers JP, et al. Results of sacral neuromodulation therapy for urinary voiding dysfunction: outcomes of a prospective, worldwide clinical study. J Urol. 2007;178:2029–2034.
Sacral nerve stimulation has been approved for use in patients with refractory voiding dysfunction since 1997. This prospective, worldwide, follow-up study sought to determine the long-term efficacy and safety of sacral neuromodulation for the treatment of refractory urgency, frequency, urge incontinence, and nonobstructive urinary retention.
After 5 years of follow-up, 68% of subjects who had urge incontinence, 56% who had urgency and frequency, and 71% who had nonobstructive urinary retention reported a degree of improvement of 50% or more in their symptoms (relative to baseline). The mean number of urge incontinence episodes decreased from 9.6 to 3.9, and mean voids per day decreased from 19.3 to 14.8 at 5 years (P<.001).
Details of the trial
Eligible patients were previously enrolled in a randomized, clinical trial investigating the efficacy of InterStim. Of the 23 sites that participated in the original study, only 17 elected to participate in the follow-up trial.
One hundred fifty-two subjects were enrolled—129 of them crossing over from the original study and 23 newly recruited and implanted with the InterStim device ( FIGURE 3 ). Investigators reviewed data from follow-up visits, self-reported symptoms obtained from voiding diaries collected annually for 5 years, and descriptive summaries of adverse events related to the sacral nerve stimulator.
One-year data were available on 138 subjects, and 5-year data were available on 105. Of the 47 participants who did not participate in 5-year follow-up, 16 had the InterStim device removed.
FIGURE 3 Aim of InterStim therapy is to regulate brain–bladder signals
The InterStim device sends a mild electrical impulse through a lead to the sacral nerves to influence the bladder and surrounding muscles. Sacral nerve stimulation helps regulate interaction between the brain and bladder and reduce voiding dysfunction.
Additional surgery was performed in half of patients
who had an adverse event
No life-threatening or irreversible adverse events were reported during the 5 years of follow-up. However, 221 adverse events occurred, of which 110 required a minor surgical procedure—in some cases, more than one. The most commonly reported adverse event was new pain or pain at the implantation site (46.1%). The most common surgical procedure was device exchange (23.7%).
Because many patients who suffer from overactive bladder syndrome fail to respond to conservative therapies, such as behavior modification and pharmacotherapy, novel alternatives are needed. This study:
- contributes to our understanding of sacral nerve stimulation
- provides much needed data on the long-term safety and efficacy of InterStim therapy
- proposes that InterStim therapy is safe and efficacious for women who have refractory overactive bladder syndrome
- suggests that it may become the gold standard for treatment of this condition.
At the Cleveland Clinic, we offer sacral nerve stimulation for refractory overactive bladder and nonobstructive urinary retention.
1. Hu TW, Wagner TH, Bentkover JD, Leblanc K, Zhou SZ, Hunt T. Costs of urinary incontinence and overactive bladder in the United States: a comparative study. Urology. 2004;63:461-465.
2. Mardon RE, Halim S, Pawlson LG, Haffer SC. Management of urinary incontinence in Medicare managed care beneficiaries: results from the 2004 Medicare Health Outcomes Survey. Arch Intern Med. 2006;166:1128-1133.
3. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167-178.
1. Hu TW, Wagner TH, Bentkover JD, Leblanc K, Zhou SZ, Hunt T. Costs of urinary incontinence and overactive bladder in the United States: a comparative study. Urology. 2004;63:461-465.
2. Mardon RE, Halim S, Pawlson LG, Haffer SC. Management of urinary incontinence in Medicare managed care beneficiaries: results from the 2004 Medicare Health Outcomes Survey. Arch Intern Med. 2006;166:1128-1133.
3. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167-178.
A guide to lotions and potions for treating vaginal atrophy
The authors would like to acknowledge Lauren Melcher, MD, an ObGyn resident at Washington Hospital Center, who contributed to this article.
CASE: New-onset dyspareunia in a menopausal patient
J. B., 53 years old, has been menopausal for 2 years. Several months after her annual examination, she schedules another appointment to discuss a worsening complaint: dyspareunia. She says she never had the problem until she reached menopause, and reports that it has become so severe that she has started avoiding sexual intercourse altogether. Even when she avoids intercourse, however, she is bothered by vaginal itching and burning.
What can you offer to her?
Various hormonal and nonhormonal products are available to relieve the frequent complaint, in menopausal women, of symptoms of vaginal atrophy: vaginal dryness, itching, burning, and dyspareunia.1-3 The array of products isn’t really surprising: As women advance through menopause, their complaints of vaginal dryness increase fivefold.4
Systemic and local estrogen therapies reverse some atrophic changes and alleviate symptoms.5 After menopause, local vaginal estrogen formulations are recommended as first-line treatment for women who experience moderate or severe symptoms of vaginal atrophy.3 Formulations such as the vaginal ring, vaginal tablet, and transdermal gels and sprays are increasingly popular.
In this article, we describe these and other products, including nonhormonal lubricants and moisturizers, to relieve:
- the range of symptoms of vaginal atrophy in menopausal women
- isolated vaginal dryness in premenopausal women.
Is hormonal therapy always necessary?
When a postmenopausal woman complains of chronic vaginal dryness, and the exam is consistent with vaginal atrophy, the recommended treatment is local vaginal estrogen. If she complains of vaginal dryness during sexual intercourse only, a vaginal lubricant is a suitable option.
When a premenopausal woman complains of vaginal dryness, a vaginal moisturizer is the best long-term treatment option. However, a vaginal lubricant is recommended for intermittent dryness during intercourse or dyspareunia.
Local estrogens avoid many risks of systemic therapy
Topical estrogen preparations are available as vaginal creams, tablets, and rings, and as transdermal lotions, gels, and patches (TABLE 1). Local preparations are preferred to systemic therapy for the treatment of atrophy because they bypass the gastrointestinal tract, undergo less conversion in the liver, and improve local tissue with minimal elevation of the serum estradiol level.1,3
The vaginal ring (Estring) delivers the lowest systemic estradiol level—approximately 5 to 10 μg of estradiol daily. Femring delivers more estradiol daily and requires the addition of progesterone in women who have an intact uterus.
Studies suggest that patients favor the estradiol-releasing vaginal ring because of its ease of use, comfort, and effectiveness, compared with vaginal estrogen cream.2,5
Local estrogen formulations were compared and reviewed in a systematic Cochrane meta-analysis of 19 trials that included 4,162 women.5 Vaginal cream, tablets, and rings were all equally effective in treating symptoms of atrophy. One trial found that cream (conjugated equine estrogen) increased the risk of uterine bleeding, breast pain, and perineal pain, compared with vaginal tablets.
Newer estrogen formulations include topical and transdermal patches, gels, lotions, and sprays (TABLE 1), all of which are systemic. They are effective in the treatment of vasomotor symptoms and vaginal atrophy.
TABLE 1
Topical estrogen formulations—a rundown of local and systemic options
| Product | Dosing | Administration | Source of active ingredient |
|---|---|---|---|
| Absorbed locally | |||
| VAGINAL TABLET | |||
| Vagifem | 25 μg of estradiol | One tablet intravaginally daily for 2 weeks; then, twice weekly | Synthesized from soy |
| VAGINAL CREAM | |||
| Premarin | 0.5 g (0.625 mg/g of conjugated estrogen) | Insert 0.5 g daily for 3 weeks; then, twice weekly (Note: Dosage can be increased to 2 g daily but this may require progesterone supplementation) | Urine of pregnant mares |
| Estrace | 0.1 mg of estradiol/g of cream | Insert 0.5 g daily for 1 or 2 weeks; then, twice weekly | Synthesized from soy and yams |
| VAGINAL RING | |||
| Estring | 2 mg (delivers 6–9 μg of estradiol daily) | Insert 1 ring intravaginally for 3 months | Synthesized from Mexican yams |
| Absorbed systemically | |||
| VAGINAL RING | |||
| Femring | Delivers 0.05 mg–0.1 mg of estradiol daily | Insert 1 ring intravaginally for 3 months | Synthesized from soy |
| ESTROGEN PATCH | |||
| Estraderm | Delivers 0.05 mg or 0.1 mg of estradiol daily | Apply patch twice weekly | Synthesized from Mexican yams |
| Estradiol (generic) | Delivers 0.05 mg or 0.1 mg of estradiol daily | ||
| Esclim | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | ||
| Vivelle, Vivelle-Dot | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | Synthesized from Mexican yams | |
| Climara | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.06 mg, 0.075 mg, or 0.1 mg of estradiol daily | Synthesized from soy | |
| Alora | Delivers 0.025 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | ||
| Menostar | Delivers 0.014 mg of estradiol daily | Apply patch once weekly (Note: Indicated only for prevention of osteoporosis) | |
| CombiPatch | Delivers 0.05 mg or 0.14 mg daily of estradiol plus 0.05 mg or 0.25 mg daily of norethindrone | Apply patch twice weekly | Synthesized from soy (estradiol) and Mexican yams (norethrindrone) |
| ESTROGEN LOTION, GEL | |||
| Estrasorb (lotion) | Content of two pouches delivers 0.05 mg daily of estradiol | Apply one packet to each leg daily | Synthesized from soy |
| EstroGel (gel) | 1.25 g (0.75 mg of estradiol) | Apply one pump to arm once daily | |
| Divigel (gel) | 0.25 g, 0.5 g, or 1 g of 0.1% estradiol | Apply one packet to upper thigh daily | |
| Elestrin (gel) | 0.87 g (0.52 mg of estradiol) | Apply one pump to arm once daily | |
| ESTROGEN SPRAY | |||
| Evamist | 1.53 mg of estradiol in each spray | Apply 1-3 sprays to forearm daily | |
| Source: Cirigliano M. Bioidentical hormone therapy: a review of the evidence. J Womens Health (Larchmt). 2007;16:600–631. | |||
When to add a progestin
A progestin is recommended in addition to a systemic estrogen formulation in women who have a uterus. For low-dose, local vaginal estrogen formulations, a progestin is usually not needed.3 However, when the treatment is vaginal cream, consider progestin supplementation when the dosage exceeds 0.5 g twice weekly for an extended time (>1 year).
The serum estrogen level with local vaginal treatment is dose-dependent, and the long-term endometrial effects of vaginal estrogens are unknown. If vaginal bleeding develops, a workup is indicated and may necessitate imaging of the endometrial echo or endometrial sampling to rule out hyperplasia, neoplasia, and cancer.
Counsel the patient about any risks
If you prescribe transdermal or oral estrogen for a patient, be sure to counsel her about the risks of systemic therapy described in the Women’s Health Initiative.6
Consider the patient’s preference
Local estrogen treatment is recommended over systemic therapy for vaginal atrophy, but patient preference should also be considered. Some women may prefer the ring or tablet to minimize excess vaginal discharge, while others may prefer a cream because of its soothing effects. Always individualize management!
Lubricants and moisturizers
Insufficient lubrication during intercourse is a common complaint among both premenopausal and postmenopausal women: As many as 60% of women report intermittent episodes of insufficient lubrication.7
Many women and their partners use a vaginal lubricant to assist with sexual relations and to self-treat for pain. A wide variety of nonhormonal products are available—many of them advertised at pharmacies and in the media—despite little published scientific evaluation. Because gynecologists routinely counsel patients on sensitive matters, including sexual practices, you may find it valuable—with appropriate candidates—to open a line of questioning about difficulties with intercourse and resulting attempts to self-medicate using over-the-counter products.
What are the indications?
A vaginal lubricant is a solution used locally, and as a temporary measure, to moisten the vaginal epithelium to facilitate a medical examination or sexual intercourse.2 Because it has a short duration, it must be applied at the time of intercourse. Lubricants can be categorized as water-, silicone-, and oil-based. Each formulation may affect the local inflammatory response, viability of sperm, and condom integrity.
A vaginal moisturizer is a gel or cream used regularly to maintain hydration of the vaginal epithelium for long-term relief of vaginal dryness.2
Both lubricants and moisturizers have many indications for both medical and personal use. Personal lubricants can be used for assistance during sexual activity, such as intercourse, masturbation, or use of sex toys. These products reduce friction and are thought to enhance pleasure in women who suffer from vaginal dryness. However, we lack sufficient data to confirm that lubricants can improve sexual dysfunction and vaginal atrophy. In general, these products are affordable, readily available, and may be helpful in the treatment of sexual dysfunction and vaginal dryness.
See TABLE 2 for a list of personal lubricants and vaginal moisturizers.
TABLE 2
Vaginal moisturizers and lubricants are plentiful and diverse
| Product (Manufacturer) | Ingredients | Notes |
|---|---|---|
| MOISTURIZERS | ||
| Replens (Columbia Laboratories) | Water, carbomer, polycarbophil, paraffin, hydrogenated palm oil, glyceride, sorbic acid, and sodium hydroxide | Should be used 3 times weekly |
| Moist Again (Lake Consumer Products) | Water, carbomer, aloe, citric acid, chlorhexidine deglutinate, sodium benzoate, potassium sorbate, diazolidinyl urea, and sorbic acid | Safe to use with a latex condom; no data on effects on sperm motility |
| Vagisil Feminine Moisturizer (Combe) | Water, glycerin, propylene glycol, poloxamer 407, methylparaben, polyquaternium-32, propylparaben, chamomile, and aloe | |
| Feminease (Parnell Pharmaceuticals) | Water, mineral oil, glycerin, yerba santa, cetyl alcohol, and methyl paraben | Yerba santa (Eriodictyon spp), a plant native to the Pacific Northwest, is used as a moisturizer in place of aloe |
| K-Y Long Lasting Moisturizer (McNeil) | Purified water, glycerin, mineral oil, calcium/sodium PVM/MA copolymer, PVM/MA decadiene crosspolymer, hydrogenated palm glyceride, methylparaben, benzoic acid, tocopherol acetate, and sodium hydroxide | |
| K-Y Silk-E (McNeil) | Water, propylene glycol, sorbitol, polysorbate 60, hydroxyethylcellulose, benzoic acid, methylparaben, tocopherol, and aloe | |
| LUBRICANTS | ||
| Water-based | ||
| Slippery Stuff (Wallace-O’Farrell) | Water, polyoxyethylene, methylparaben, propylene glycol, isopropynol | |
| Astroglide (BioFilm) | Water, glycerin, methylparaben, propylparaben, polypropylene glycol, polyquaternium, hydroxyethylcellulose, and sodium benzoate | Also sold in a glycerin-free and paraben-free formulation |
| K-Y Jelly (McNeil) | Water, glycerin, hydroxyethylcellulose, parabens, and chlorhexidine | |
| Summer’s Eve Lubricant (C.B. Fleet) | Water, propylene glycol, methylcellulose, xanthan gum, sodium lactate, methylparaben, lactic acid, dextrose, sodium chloride, edatate disodium, pectin, and propylparaben | |
| FemGlide (WalMed) | Water, polyoxyethylene, methylparaben, and sodium carbomer | |
| Pre-Seed (INGfertility) | Water, hydroxyethylcellulose, arabinogalactan, paraben, and Pluronic copolymers | Promoted to women and their partners who are trying to conceive |
| Silicone-based | ||
| ID Millennium (Westridge Laboratories) | Cyclomethicone, dimethicone, and dimethiconol | Less drying than other lubricants |
| Pjur | Cyclopentasiloxane, dimethicone, and dimethiconol | Compatible with a condom |
| Pink | Dimethicone, vitamin E, aloe vera, dimethiconol, and cyclomethicone | |
| K-Y Liquibeads (McNeil) | Dimethicone, gelatin, glycerin, and dimethiconol | Active ingredients are contained in so-called ovules that release lubricant over several days |
| Oil-based | ||
| Élégance Women’s Lubricant | Natural oils | Does not contain alcohol, glycerin, or parabens; is incompatible with a condom; helpful for women who have vulvodynia or vestibulitis |
What to offer when estrogen is not an option
Some women may want to avoid hormonal treatment, or have a contraindication to it, such as estrogen-receptor–positive breast cancer.8 In premenopausal women, vaginal atrophy can occur with lactation or postpartum hormonal changes, or may result from the use of anti-estrogenic agents for breast cancer. Other candidates for nonhormonal therapy are women who have chronic vulvar pain syndromes. In these women, vaginal lubricants can be especially useful.
Although they are less effective than estrogen, vaginal moisturizers, such as Replens, have been shown to reverse symptoms of vaginal atrophy and decrease discomfort during intercourse.9
The therapeutic options for vaginal atrophy are likely to broaden in the near future. Ospemifene (Ophena), a selective estrogen receptor modulator (SERM) under development for the treatment of vaginal atrophy, has reached the end of Phase-3 clinical study, with positive efficacy results. A long-term safety study of the orally administered SERM has also been completed, reports QuatRx, the drug’s manufacturer. The company expects to file a New Drug Application with the US Food and Drug Administration early next year.
Phase-3 trials documented significant improvement in dryness, dyspareunia, and other endpoints
The first Phase-3 study of Ophena was announced by QuatRx in January 2008 and presented at the 90th annual meeting of the Endocrine Society. Women who were treated with 60 mg daily of Ophena experienced statistically significant improvement in vaginal dryness, dyspareunia, and the proportion of parabasal and superficial cells in the epithelium of vaginal walls. The vaginal pH level also declined. The drug did not cause hot flushes among users.
The second Phase-3 study was a randomized, double-blind, placebo-controlled study of 919 women who had vulvovaginal atrophy. It was conducted at 116 sites in the United States. Among the cohort of 605 women who identified dyspareunia as their most bothersome symptom, positive efficacy results were achieved in all four primary endpoints, including:
- a decrease in parabasal cells
- an increase in superficial cells
- a decrease in the vaginal pH level
- improvement in dyspareunia.
The trial demonstrated statistically significant improvement from baseline to week 12 in all four endpoints, compared with placebo (P.0001>
All women were supplied with a nonhormonal vaginal lubricant to be used as needed during the treatment period; the study found efficacy above and beyond usage of this lubricant, according to a press release from QuatRx.
Is the benefit worth the risk?
Some have questioned whether a systemic drug is overkill for a complaint like postmenopausal vaginal atrophy.14 Because Ophena is a SERM, it is likely to carry a cardiovascular risk profile similar to that of other drugs in its class. For example, in a large randomized trial, raloxifene (Evista) failed to reduce coronary artery disease and significantly increased the incidence of fatal stroke and venous thromboembolism.15 When local estrogen formulations that do not carry such risks are already available, some experts question the advisability of developing another systemic agent.
Another question: Is it realistic to expect the patient to take a drug every day when her chief complaint is postmenopausal dyspareunia and she is likely to have intercourse only once or twice a week?
These questions probably won’t be addressed until the drug enters the market—and physicians and their patients will be the ones providing the answers.
Specialty lubricants are unproven
In contrast to products designed to treat vaginal dryness and atrophy, some lubricants are marketed specifically for sexual enhancement. Warming lubricants cause a heating sensation on the skin and usually contain menthol, L-arginine, or capsaicin. Natural and artificial flavors are used to manufacture flavored lubricants.
None of these products have been scientifically proven to enhance sexual function.
Oil-based lubricants may impede condom integrity
It is estimated that 40% of couples who use condoms also use a lubricant to assist with intercourse.10 The integrity of latex condoms has been shown to deteriorate with the use of an oil-based lubricant or petrolatum. One study, in which the mean burst time of condoms was assessed during pressurized air inflation, showed a significant reduction in that time when vaginal lubricants that contained mineral or vegetable oil were used.11
Oil-based lubricants also have been shown to increase the slippage rate, with a trend toward increased breakage.10
Water-based lubricants may slightly increase slippage, but they reduce breakage.
Women should avoid oil-based lubricants when their partner uses a condom.
Some lubricants affect sperm quality
Choosing a vaginal lubricant can be of particular concern to a woman who is being treated for infertility. Lubricants may affect the integrity and function of sperm, even if they do not contain spermicide. Noncommercial products, such as glycerin, olive oil, vegetable oil, and, even, saliva have been associated with a loss of sperm function.12
A recent study found that Replens and Astroglide cause a dramatic decrease in sperm motility. FemGlide causes less of a decrease—but still a significant one.12 The nonphysiologic osmolality and pH of these products may be the cause of such sperm damage. Pre-Seed, which has a more physiologic pH level and isotonic quality, was found to cause minimal harm to sperm motility and chromatin quality.12
Avoid propylene glycol in women who have vulvodynia
Vaginal lubricants and moisturizers are also used in the treatment of vulvodynia or chronic vulvar pain syndromes. According to an ACOG Committee Opinion, topical application of preservative-free solutions, such as vegetable oil or plain petrolatum, is recommended to hold moisture within the tissues and provide a protective barrier.13 Adequate lubrication is also recommended during intercourse.
Products that contain propylene glycol, or alcohol, may act as an irritant in women who experience local pain and heightened vaginal sensitivity. For that reason, such products should be avoided in this population.
CASE RESOLVED
Careful examination reveals urogenital atrophy with absence of any fungal or bacterial infection of the vulva or vagina. The patient chooses to use a water-based lubricant during sexual relations and begins using intravaginal estradiol tablets (other options include the vaginal ring or cream). Symptoms of dyspareunia disappear almost immediately, and vaginal burning improves after 6 weeks.
1. Al-Baghdadi O, Ewies AA. Topical estrogen therapy in the management of postmenopausal vaginal atrophy: an up-to-date overview. Climacteric. 2009;12:91-105.
2. Willhite LA, O’Connell MB. Urogenital atrophy: prevention and treatment. Pharmacotherapy. 2001;21:464-480.
3. Archer DF. Efficacy and tolerability of local estrogen therapy for urogenital atrophy. Menopause. 2009;17:1-10.
4. Dennerstein L, Dudley EC, Hopper JL, Guthrie JR, Burger HG. A prospective population-based study of menopausal symptoms. Obstet Gynecol. 2000;96:351-358.
5. Suckling J, Lethaby A, Kennedy R. Local oestrogen for vaginal atrophy in postmenopausal women. Cochrane Database Syst Rev. 2006;(4):CD001500.-
6. Anderson GL, Limacher M, Assaf AR, et al. Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial. JAMA. 2004;291:1701-1712.
7. Oberg K, Fugl-Meyer AR, Fugl-Meyer KS. On categorization and quantification of women’s sexual dysfunctions: an epidemiological approach. Int J Impot Res. 2004;16:261-269.
8. Loprinzi CL, Wolf SL, Barton DL, Laack NN. Symptom management in premenopausal patients with breast cancer. Lancet Oncol. 2008;9:993-1001.
9. Bygdeman M, Swahn ML. Replens versus dienoestrol cream in the symptomatic treatment of vaginal atrophy in postmenopausal women. Maturitas. 1996;23:259-263.
10. Steiner M, Piedrahita C, Glover L, Joanis C, Spruyt A, Foldesy R. The impact of lubricants on latex condoms during vaginal intercourse. Int J STD AIDS. 1994;5:29-36.
11. Rosen AD, Rosen T. Study of condom integrity after brief exposure to over-the-counter vaginal preparations. South Med J. 1999;92:305-307.
12. Agarwal A, Deepinder F, Cocuzza M, Short RA, Evenson DP. Effect of vaginal lubricants on sperm motility and chromatin integrity: a prospective comparative study. Fertil Steril. 2008;89:375-379.
13. ACOG Committee Opinion #345: Vulvodynia. October 2006. ACOG Committee on Gynecologic Practice. Obstet Gynecol. 2006;108:1049-1052.
14. Ophena is a me-too drug with an impractical mode of administration. Gerson Lehrman Group. Jan. 14, 2008. Available at: http://www.glgroup.com/News/Ophena-is-a-Me-Too-Drug-with-an-Impractical-Mode-of-Administration-20651.html. Accessed Nov. 11, 2009.
15. de Villiers TJ. Clinical issues regarding cardiovascular disease and selective estrogen receptor modulators in postmenopausal women. Climacteric. 2009;12 Suppl 1:108-111.
The authors would like to acknowledge Lauren Melcher, MD, an ObGyn resident at Washington Hospital Center, who contributed to this article.
CASE: New-onset dyspareunia in a menopausal patient
J. B., 53 years old, has been menopausal for 2 years. Several months after her annual examination, she schedules another appointment to discuss a worsening complaint: dyspareunia. She says she never had the problem until she reached menopause, and reports that it has become so severe that she has started avoiding sexual intercourse altogether. Even when she avoids intercourse, however, she is bothered by vaginal itching and burning.
What can you offer to her?
Various hormonal and nonhormonal products are available to relieve the frequent complaint, in menopausal women, of symptoms of vaginal atrophy: vaginal dryness, itching, burning, and dyspareunia.1-3 The array of products isn’t really surprising: As women advance through menopause, their complaints of vaginal dryness increase fivefold.4
Systemic and local estrogen therapies reverse some atrophic changes and alleviate symptoms.5 After menopause, local vaginal estrogen formulations are recommended as first-line treatment for women who experience moderate or severe symptoms of vaginal atrophy.3 Formulations such as the vaginal ring, vaginal tablet, and transdermal gels and sprays are increasingly popular.
In this article, we describe these and other products, including nonhormonal lubricants and moisturizers, to relieve:
- the range of symptoms of vaginal atrophy in menopausal women
- isolated vaginal dryness in premenopausal women.
Is hormonal therapy always necessary?
When a postmenopausal woman complains of chronic vaginal dryness, and the exam is consistent with vaginal atrophy, the recommended treatment is local vaginal estrogen. If she complains of vaginal dryness during sexual intercourse only, a vaginal lubricant is a suitable option.
When a premenopausal woman complains of vaginal dryness, a vaginal moisturizer is the best long-term treatment option. However, a vaginal lubricant is recommended for intermittent dryness during intercourse or dyspareunia.
Local estrogens avoid many risks of systemic therapy
Topical estrogen preparations are available as vaginal creams, tablets, and rings, and as transdermal lotions, gels, and patches (TABLE 1). Local preparations are preferred to systemic therapy for the treatment of atrophy because they bypass the gastrointestinal tract, undergo less conversion in the liver, and improve local tissue with minimal elevation of the serum estradiol level.1,3
The vaginal ring (Estring) delivers the lowest systemic estradiol level—approximately 5 to 10 μg of estradiol daily. Femring delivers more estradiol daily and requires the addition of progesterone in women who have an intact uterus.
Studies suggest that patients favor the estradiol-releasing vaginal ring because of its ease of use, comfort, and effectiveness, compared with vaginal estrogen cream.2,5
Local estrogen formulations were compared and reviewed in a systematic Cochrane meta-analysis of 19 trials that included 4,162 women.5 Vaginal cream, tablets, and rings were all equally effective in treating symptoms of atrophy. One trial found that cream (conjugated equine estrogen) increased the risk of uterine bleeding, breast pain, and perineal pain, compared with vaginal tablets.
Newer estrogen formulations include topical and transdermal patches, gels, lotions, and sprays (TABLE 1), all of which are systemic. They are effective in the treatment of vasomotor symptoms and vaginal atrophy.
TABLE 1
Topical estrogen formulations—a rundown of local and systemic options
| Product | Dosing | Administration | Source of active ingredient |
|---|---|---|---|
| Absorbed locally | |||
| VAGINAL TABLET | |||
| Vagifem | 25 μg of estradiol | One tablet intravaginally daily for 2 weeks; then, twice weekly | Synthesized from soy |
| VAGINAL CREAM | |||
| Premarin | 0.5 g (0.625 mg/g of conjugated estrogen) | Insert 0.5 g daily for 3 weeks; then, twice weekly (Note: Dosage can be increased to 2 g daily but this may require progesterone supplementation) | Urine of pregnant mares |
| Estrace | 0.1 mg of estradiol/g of cream | Insert 0.5 g daily for 1 or 2 weeks; then, twice weekly | Synthesized from soy and yams |
| VAGINAL RING | |||
| Estring | 2 mg (delivers 6–9 μg of estradiol daily) | Insert 1 ring intravaginally for 3 months | Synthesized from Mexican yams |
| Absorbed systemically | |||
| VAGINAL RING | |||
| Femring | Delivers 0.05 mg–0.1 mg of estradiol daily | Insert 1 ring intravaginally for 3 months | Synthesized from soy |
| ESTROGEN PATCH | |||
| Estraderm | Delivers 0.05 mg or 0.1 mg of estradiol daily | Apply patch twice weekly | Synthesized from Mexican yams |
| Estradiol (generic) | Delivers 0.05 mg or 0.1 mg of estradiol daily | ||
| Esclim | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | ||
| Vivelle, Vivelle-Dot | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | Synthesized from Mexican yams | |
| Climara | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.06 mg, 0.075 mg, or 0.1 mg of estradiol daily | Synthesized from soy | |
| Alora | Delivers 0.025 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | ||
| Menostar | Delivers 0.014 mg of estradiol daily | Apply patch once weekly (Note: Indicated only for prevention of osteoporosis) | |
| CombiPatch | Delivers 0.05 mg or 0.14 mg daily of estradiol plus 0.05 mg or 0.25 mg daily of norethindrone | Apply patch twice weekly | Synthesized from soy (estradiol) and Mexican yams (norethrindrone) |
| ESTROGEN LOTION, GEL | |||
| Estrasorb (lotion) | Content of two pouches delivers 0.05 mg daily of estradiol | Apply one packet to each leg daily | Synthesized from soy |
| EstroGel (gel) | 1.25 g (0.75 mg of estradiol) | Apply one pump to arm once daily | |
| Divigel (gel) | 0.25 g, 0.5 g, or 1 g of 0.1% estradiol | Apply one packet to upper thigh daily | |
| Elestrin (gel) | 0.87 g (0.52 mg of estradiol) | Apply one pump to arm once daily | |
| ESTROGEN SPRAY | |||
| Evamist | 1.53 mg of estradiol in each spray | Apply 1-3 sprays to forearm daily | |
| Source: Cirigliano M. Bioidentical hormone therapy: a review of the evidence. J Womens Health (Larchmt). 2007;16:600–631. | |||
When to add a progestin
A progestin is recommended in addition to a systemic estrogen formulation in women who have a uterus. For low-dose, local vaginal estrogen formulations, a progestin is usually not needed.3 However, when the treatment is vaginal cream, consider progestin supplementation when the dosage exceeds 0.5 g twice weekly for an extended time (>1 year).
The serum estrogen level with local vaginal treatment is dose-dependent, and the long-term endometrial effects of vaginal estrogens are unknown. If vaginal bleeding develops, a workup is indicated and may necessitate imaging of the endometrial echo or endometrial sampling to rule out hyperplasia, neoplasia, and cancer.
Counsel the patient about any risks
If you prescribe transdermal or oral estrogen for a patient, be sure to counsel her about the risks of systemic therapy described in the Women’s Health Initiative.6
Consider the patient’s preference
Local estrogen treatment is recommended over systemic therapy for vaginal atrophy, but patient preference should also be considered. Some women may prefer the ring or tablet to minimize excess vaginal discharge, while others may prefer a cream because of its soothing effects. Always individualize management!
Lubricants and moisturizers
Insufficient lubrication during intercourse is a common complaint among both premenopausal and postmenopausal women: As many as 60% of women report intermittent episodes of insufficient lubrication.7
Many women and their partners use a vaginal lubricant to assist with sexual relations and to self-treat for pain. A wide variety of nonhormonal products are available—many of them advertised at pharmacies and in the media—despite little published scientific evaluation. Because gynecologists routinely counsel patients on sensitive matters, including sexual practices, you may find it valuable—with appropriate candidates—to open a line of questioning about difficulties with intercourse and resulting attempts to self-medicate using over-the-counter products.
What are the indications?
A vaginal lubricant is a solution used locally, and as a temporary measure, to moisten the vaginal epithelium to facilitate a medical examination or sexual intercourse.2 Because it has a short duration, it must be applied at the time of intercourse. Lubricants can be categorized as water-, silicone-, and oil-based. Each formulation may affect the local inflammatory response, viability of sperm, and condom integrity.
A vaginal moisturizer is a gel or cream used regularly to maintain hydration of the vaginal epithelium for long-term relief of vaginal dryness.2
Both lubricants and moisturizers have many indications for both medical and personal use. Personal lubricants can be used for assistance during sexual activity, such as intercourse, masturbation, or use of sex toys. These products reduce friction and are thought to enhance pleasure in women who suffer from vaginal dryness. However, we lack sufficient data to confirm that lubricants can improve sexual dysfunction and vaginal atrophy. In general, these products are affordable, readily available, and may be helpful in the treatment of sexual dysfunction and vaginal dryness.
See TABLE 2 for a list of personal lubricants and vaginal moisturizers.
TABLE 2
Vaginal moisturizers and lubricants are plentiful and diverse
| Product (Manufacturer) | Ingredients | Notes |
|---|---|---|
| MOISTURIZERS | ||
| Replens (Columbia Laboratories) | Water, carbomer, polycarbophil, paraffin, hydrogenated palm oil, glyceride, sorbic acid, and sodium hydroxide | Should be used 3 times weekly |
| Moist Again (Lake Consumer Products) | Water, carbomer, aloe, citric acid, chlorhexidine deglutinate, sodium benzoate, potassium sorbate, diazolidinyl urea, and sorbic acid | Safe to use with a latex condom; no data on effects on sperm motility |
| Vagisil Feminine Moisturizer (Combe) | Water, glycerin, propylene glycol, poloxamer 407, methylparaben, polyquaternium-32, propylparaben, chamomile, and aloe | |
| Feminease (Parnell Pharmaceuticals) | Water, mineral oil, glycerin, yerba santa, cetyl alcohol, and methyl paraben | Yerba santa (Eriodictyon spp), a plant native to the Pacific Northwest, is used as a moisturizer in place of aloe |
| K-Y Long Lasting Moisturizer (McNeil) | Purified water, glycerin, mineral oil, calcium/sodium PVM/MA copolymer, PVM/MA decadiene crosspolymer, hydrogenated palm glyceride, methylparaben, benzoic acid, tocopherol acetate, and sodium hydroxide | |
| K-Y Silk-E (McNeil) | Water, propylene glycol, sorbitol, polysorbate 60, hydroxyethylcellulose, benzoic acid, methylparaben, tocopherol, and aloe | |
| LUBRICANTS | ||
| Water-based | ||
| Slippery Stuff (Wallace-O’Farrell) | Water, polyoxyethylene, methylparaben, propylene glycol, isopropynol | |
| Astroglide (BioFilm) | Water, glycerin, methylparaben, propylparaben, polypropylene glycol, polyquaternium, hydroxyethylcellulose, and sodium benzoate | Also sold in a glycerin-free and paraben-free formulation |
| K-Y Jelly (McNeil) | Water, glycerin, hydroxyethylcellulose, parabens, and chlorhexidine | |
| Summer’s Eve Lubricant (C.B. Fleet) | Water, propylene glycol, methylcellulose, xanthan gum, sodium lactate, methylparaben, lactic acid, dextrose, sodium chloride, edatate disodium, pectin, and propylparaben | |
| FemGlide (WalMed) | Water, polyoxyethylene, methylparaben, and sodium carbomer | |
| Pre-Seed (INGfertility) | Water, hydroxyethylcellulose, arabinogalactan, paraben, and Pluronic copolymers | Promoted to women and their partners who are trying to conceive |
| Silicone-based | ||
| ID Millennium (Westridge Laboratories) | Cyclomethicone, dimethicone, and dimethiconol | Less drying than other lubricants |
| Pjur | Cyclopentasiloxane, dimethicone, and dimethiconol | Compatible with a condom |
| Pink | Dimethicone, vitamin E, aloe vera, dimethiconol, and cyclomethicone | |
| K-Y Liquibeads (McNeil) | Dimethicone, gelatin, glycerin, and dimethiconol | Active ingredients are contained in so-called ovules that release lubricant over several days |
| Oil-based | ||
| Élégance Women’s Lubricant | Natural oils | Does not contain alcohol, glycerin, or parabens; is incompatible with a condom; helpful for women who have vulvodynia or vestibulitis |
What to offer when estrogen is not an option
Some women may want to avoid hormonal treatment, or have a contraindication to it, such as estrogen-receptor–positive breast cancer.8 In premenopausal women, vaginal atrophy can occur with lactation or postpartum hormonal changes, or may result from the use of anti-estrogenic agents for breast cancer. Other candidates for nonhormonal therapy are women who have chronic vulvar pain syndromes. In these women, vaginal lubricants can be especially useful.
Although they are less effective than estrogen, vaginal moisturizers, such as Replens, have been shown to reverse symptoms of vaginal atrophy and decrease discomfort during intercourse.9
The therapeutic options for vaginal atrophy are likely to broaden in the near future. Ospemifene (Ophena), a selective estrogen receptor modulator (SERM) under development for the treatment of vaginal atrophy, has reached the end of Phase-3 clinical study, with positive efficacy results. A long-term safety study of the orally administered SERM has also been completed, reports QuatRx, the drug’s manufacturer. The company expects to file a New Drug Application with the US Food and Drug Administration early next year.
Phase-3 trials documented significant improvement in dryness, dyspareunia, and other endpoints
The first Phase-3 study of Ophena was announced by QuatRx in January 2008 and presented at the 90th annual meeting of the Endocrine Society. Women who were treated with 60 mg daily of Ophena experienced statistically significant improvement in vaginal dryness, dyspareunia, and the proportion of parabasal and superficial cells in the epithelium of vaginal walls. The vaginal pH level also declined. The drug did not cause hot flushes among users.
The second Phase-3 study was a randomized, double-blind, placebo-controlled study of 919 women who had vulvovaginal atrophy. It was conducted at 116 sites in the United States. Among the cohort of 605 women who identified dyspareunia as their most bothersome symptom, positive efficacy results were achieved in all four primary endpoints, including:
- a decrease in parabasal cells
- an increase in superficial cells
- a decrease in the vaginal pH level
- improvement in dyspareunia.
The trial demonstrated statistically significant improvement from baseline to week 12 in all four endpoints, compared with placebo (P.0001>
All women were supplied with a nonhormonal vaginal lubricant to be used as needed during the treatment period; the study found efficacy above and beyond usage of this lubricant, according to a press release from QuatRx.
Is the benefit worth the risk?
Some have questioned whether a systemic drug is overkill for a complaint like postmenopausal vaginal atrophy.14 Because Ophena is a SERM, it is likely to carry a cardiovascular risk profile similar to that of other drugs in its class. For example, in a large randomized trial, raloxifene (Evista) failed to reduce coronary artery disease and significantly increased the incidence of fatal stroke and venous thromboembolism.15 When local estrogen formulations that do not carry such risks are already available, some experts question the advisability of developing another systemic agent.
Another question: Is it realistic to expect the patient to take a drug every day when her chief complaint is postmenopausal dyspareunia and she is likely to have intercourse only once or twice a week?
These questions probably won’t be addressed until the drug enters the market—and physicians and their patients will be the ones providing the answers.
Specialty lubricants are unproven
In contrast to products designed to treat vaginal dryness and atrophy, some lubricants are marketed specifically for sexual enhancement. Warming lubricants cause a heating sensation on the skin and usually contain menthol, L-arginine, or capsaicin. Natural and artificial flavors are used to manufacture flavored lubricants.
None of these products have been scientifically proven to enhance sexual function.
Oil-based lubricants may impede condom integrity
It is estimated that 40% of couples who use condoms also use a lubricant to assist with intercourse.10 The integrity of latex condoms has been shown to deteriorate with the use of an oil-based lubricant or petrolatum. One study, in which the mean burst time of condoms was assessed during pressurized air inflation, showed a significant reduction in that time when vaginal lubricants that contained mineral or vegetable oil were used.11
Oil-based lubricants also have been shown to increase the slippage rate, with a trend toward increased breakage.10
Water-based lubricants may slightly increase slippage, but they reduce breakage.
Women should avoid oil-based lubricants when their partner uses a condom.
Some lubricants affect sperm quality
Choosing a vaginal lubricant can be of particular concern to a woman who is being treated for infertility. Lubricants may affect the integrity and function of sperm, even if they do not contain spermicide. Noncommercial products, such as glycerin, olive oil, vegetable oil, and, even, saliva have been associated with a loss of sperm function.12
A recent study found that Replens and Astroglide cause a dramatic decrease in sperm motility. FemGlide causes less of a decrease—but still a significant one.12 The nonphysiologic osmolality and pH of these products may be the cause of such sperm damage. Pre-Seed, which has a more physiologic pH level and isotonic quality, was found to cause minimal harm to sperm motility and chromatin quality.12
Avoid propylene glycol in women who have vulvodynia
Vaginal lubricants and moisturizers are also used in the treatment of vulvodynia or chronic vulvar pain syndromes. According to an ACOG Committee Opinion, topical application of preservative-free solutions, such as vegetable oil or plain petrolatum, is recommended to hold moisture within the tissues and provide a protective barrier.13 Adequate lubrication is also recommended during intercourse.
Products that contain propylene glycol, or alcohol, may act as an irritant in women who experience local pain and heightened vaginal sensitivity. For that reason, such products should be avoided in this population.
CASE RESOLVED
Careful examination reveals urogenital atrophy with absence of any fungal or bacterial infection of the vulva or vagina. The patient chooses to use a water-based lubricant during sexual relations and begins using intravaginal estradiol tablets (other options include the vaginal ring or cream). Symptoms of dyspareunia disappear almost immediately, and vaginal burning improves after 6 weeks.
The authors would like to acknowledge Lauren Melcher, MD, an ObGyn resident at Washington Hospital Center, who contributed to this article.
CASE: New-onset dyspareunia in a menopausal patient
J. B., 53 years old, has been menopausal for 2 years. Several months after her annual examination, she schedules another appointment to discuss a worsening complaint: dyspareunia. She says she never had the problem until she reached menopause, and reports that it has become so severe that she has started avoiding sexual intercourse altogether. Even when she avoids intercourse, however, she is bothered by vaginal itching and burning.
What can you offer to her?
Various hormonal and nonhormonal products are available to relieve the frequent complaint, in menopausal women, of symptoms of vaginal atrophy: vaginal dryness, itching, burning, and dyspareunia.1-3 The array of products isn’t really surprising: As women advance through menopause, their complaints of vaginal dryness increase fivefold.4
Systemic and local estrogen therapies reverse some atrophic changes and alleviate symptoms.5 After menopause, local vaginal estrogen formulations are recommended as first-line treatment for women who experience moderate or severe symptoms of vaginal atrophy.3 Formulations such as the vaginal ring, vaginal tablet, and transdermal gels and sprays are increasingly popular.
In this article, we describe these and other products, including nonhormonal lubricants and moisturizers, to relieve:
- the range of symptoms of vaginal atrophy in menopausal women
- isolated vaginal dryness in premenopausal women.
Is hormonal therapy always necessary?
When a postmenopausal woman complains of chronic vaginal dryness, and the exam is consistent with vaginal atrophy, the recommended treatment is local vaginal estrogen. If she complains of vaginal dryness during sexual intercourse only, a vaginal lubricant is a suitable option.
When a premenopausal woman complains of vaginal dryness, a vaginal moisturizer is the best long-term treatment option. However, a vaginal lubricant is recommended for intermittent dryness during intercourse or dyspareunia.
Local estrogens avoid many risks of systemic therapy
Topical estrogen preparations are available as vaginal creams, tablets, and rings, and as transdermal lotions, gels, and patches (TABLE 1). Local preparations are preferred to systemic therapy for the treatment of atrophy because they bypass the gastrointestinal tract, undergo less conversion in the liver, and improve local tissue with minimal elevation of the serum estradiol level.1,3
The vaginal ring (Estring) delivers the lowest systemic estradiol level—approximately 5 to 10 μg of estradiol daily. Femring delivers more estradiol daily and requires the addition of progesterone in women who have an intact uterus.
Studies suggest that patients favor the estradiol-releasing vaginal ring because of its ease of use, comfort, and effectiveness, compared with vaginal estrogen cream.2,5
Local estrogen formulations were compared and reviewed in a systematic Cochrane meta-analysis of 19 trials that included 4,162 women.5 Vaginal cream, tablets, and rings were all equally effective in treating symptoms of atrophy. One trial found that cream (conjugated equine estrogen) increased the risk of uterine bleeding, breast pain, and perineal pain, compared with vaginal tablets.
Newer estrogen formulations include topical and transdermal patches, gels, lotions, and sprays (TABLE 1), all of which are systemic. They are effective in the treatment of vasomotor symptoms and vaginal atrophy.
TABLE 1
Topical estrogen formulations—a rundown of local and systemic options
| Product | Dosing | Administration | Source of active ingredient |
|---|---|---|---|
| Absorbed locally | |||
| VAGINAL TABLET | |||
| Vagifem | 25 μg of estradiol | One tablet intravaginally daily for 2 weeks; then, twice weekly | Synthesized from soy |
| VAGINAL CREAM | |||
| Premarin | 0.5 g (0.625 mg/g of conjugated estrogen) | Insert 0.5 g daily for 3 weeks; then, twice weekly (Note: Dosage can be increased to 2 g daily but this may require progesterone supplementation) | Urine of pregnant mares |
| Estrace | 0.1 mg of estradiol/g of cream | Insert 0.5 g daily for 1 or 2 weeks; then, twice weekly | Synthesized from soy and yams |
| VAGINAL RING | |||
| Estring | 2 mg (delivers 6–9 μg of estradiol daily) | Insert 1 ring intravaginally for 3 months | Synthesized from Mexican yams |
| Absorbed systemically | |||
| VAGINAL RING | |||
| Femring | Delivers 0.05 mg–0.1 mg of estradiol daily | Insert 1 ring intravaginally for 3 months | Synthesized from soy |
| ESTROGEN PATCH | |||
| Estraderm | Delivers 0.05 mg or 0.1 mg of estradiol daily | Apply patch twice weekly | Synthesized from Mexican yams |
| Estradiol (generic) | Delivers 0.05 mg or 0.1 mg of estradiol daily | ||
| Esclim | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | ||
| Vivelle, Vivelle-Dot | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | Synthesized from Mexican yams | |
| Climara | Delivers 0.025 mg, 0.0375 mg, 0.05 mg, 0.06 mg, 0.075 mg, or 0.1 mg of estradiol daily | Synthesized from soy | |
| Alora | Delivers 0.025 mg, 0.05 mg, 0.075 mg, or 0.1 mg of estradiol daily | ||
| Menostar | Delivers 0.014 mg of estradiol daily | Apply patch once weekly (Note: Indicated only for prevention of osteoporosis) | |
| CombiPatch | Delivers 0.05 mg or 0.14 mg daily of estradiol plus 0.05 mg or 0.25 mg daily of norethindrone | Apply patch twice weekly | Synthesized from soy (estradiol) and Mexican yams (norethrindrone) |
| ESTROGEN LOTION, GEL | |||
| Estrasorb (lotion) | Content of two pouches delivers 0.05 mg daily of estradiol | Apply one packet to each leg daily | Synthesized from soy |
| EstroGel (gel) | 1.25 g (0.75 mg of estradiol) | Apply one pump to arm once daily | |
| Divigel (gel) | 0.25 g, 0.5 g, or 1 g of 0.1% estradiol | Apply one packet to upper thigh daily | |
| Elestrin (gel) | 0.87 g (0.52 mg of estradiol) | Apply one pump to arm once daily | |
| ESTROGEN SPRAY | |||
| Evamist | 1.53 mg of estradiol in each spray | Apply 1-3 sprays to forearm daily | |
| Source: Cirigliano M. Bioidentical hormone therapy: a review of the evidence. J Womens Health (Larchmt). 2007;16:600–631. | |||
When to add a progestin
A progestin is recommended in addition to a systemic estrogen formulation in women who have a uterus. For low-dose, local vaginal estrogen formulations, a progestin is usually not needed.3 However, when the treatment is vaginal cream, consider progestin supplementation when the dosage exceeds 0.5 g twice weekly for an extended time (>1 year).
The serum estrogen level with local vaginal treatment is dose-dependent, and the long-term endometrial effects of vaginal estrogens are unknown. If vaginal bleeding develops, a workup is indicated and may necessitate imaging of the endometrial echo or endometrial sampling to rule out hyperplasia, neoplasia, and cancer.
Counsel the patient about any risks
If you prescribe transdermal or oral estrogen for a patient, be sure to counsel her about the risks of systemic therapy described in the Women’s Health Initiative.6
Consider the patient’s preference
Local estrogen treatment is recommended over systemic therapy for vaginal atrophy, but patient preference should also be considered. Some women may prefer the ring or tablet to minimize excess vaginal discharge, while others may prefer a cream because of its soothing effects. Always individualize management!
Lubricants and moisturizers
Insufficient lubrication during intercourse is a common complaint among both premenopausal and postmenopausal women: As many as 60% of women report intermittent episodes of insufficient lubrication.7
Many women and their partners use a vaginal lubricant to assist with sexual relations and to self-treat for pain. A wide variety of nonhormonal products are available—many of them advertised at pharmacies and in the media—despite little published scientific evaluation. Because gynecologists routinely counsel patients on sensitive matters, including sexual practices, you may find it valuable—with appropriate candidates—to open a line of questioning about difficulties with intercourse and resulting attempts to self-medicate using over-the-counter products.
What are the indications?
A vaginal lubricant is a solution used locally, and as a temporary measure, to moisten the vaginal epithelium to facilitate a medical examination or sexual intercourse.2 Because it has a short duration, it must be applied at the time of intercourse. Lubricants can be categorized as water-, silicone-, and oil-based. Each formulation may affect the local inflammatory response, viability of sperm, and condom integrity.
A vaginal moisturizer is a gel or cream used regularly to maintain hydration of the vaginal epithelium for long-term relief of vaginal dryness.2
Both lubricants and moisturizers have many indications for both medical and personal use. Personal lubricants can be used for assistance during sexual activity, such as intercourse, masturbation, or use of sex toys. These products reduce friction and are thought to enhance pleasure in women who suffer from vaginal dryness. However, we lack sufficient data to confirm that lubricants can improve sexual dysfunction and vaginal atrophy. In general, these products are affordable, readily available, and may be helpful in the treatment of sexual dysfunction and vaginal dryness.
See TABLE 2 for a list of personal lubricants and vaginal moisturizers.
TABLE 2
Vaginal moisturizers and lubricants are plentiful and diverse
| Product (Manufacturer) | Ingredients | Notes |
|---|---|---|
| MOISTURIZERS | ||
| Replens (Columbia Laboratories) | Water, carbomer, polycarbophil, paraffin, hydrogenated palm oil, glyceride, sorbic acid, and sodium hydroxide | Should be used 3 times weekly |
| Moist Again (Lake Consumer Products) | Water, carbomer, aloe, citric acid, chlorhexidine deglutinate, sodium benzoate, potassium sorbate, diazolidinyl urea, and sorbic acid | Safe to use with a latex condom; no data on effects on sperm motility |
| Vagisil Feminine Moisturizer (Combe) | Water, glycerin, propylene glycol, poloxamer 407, methylparaben, polyquaternium-32, propylparaben, chamomile, and aloe | |
| Feminease (Parnell Pharmaceuticals) | Water, mineral oil, glycerin, yerba santa, cetyl alcohol, and methyl paraben | Yerba santa (Eriodictyon spp), a plant native to the Pacific Northwest, is used as a moisturizer in place of aloe |
| K-Y Long Lasting Moisturizer (McNeil) | Purified water, glycerin, mineral oil, calcium/sodium PVM/MA copolymer, PVM/MA decadiene crosspolymer, hydrogenated palm glyceride, methylparaben, benzoic acid, tocopherol acetate, and sodium hydroxide | |
| K-Y Silk-E (McNeil) | Water, propylene glycol, sorbitol, polysorbate 60, hydroxyethylcellulose, benzoic acid, methylparaben, tocopherol, and aloe | |
| LUBRICANTS | ||
| Water-based | ||
| Slippery Stuff (Wallace-O’Farrell) | Water, polyoxyethylene, methylparaben, propylene glycol, isopropynol | |
| Astroglide (BioFilm) | Water, glycerin, methylparaben, propylparaben, polypropylene glycol, polyquaternium, hydroxyethylcellulose, and sodium benzoate | Also sold in a glycerin-free and paraben-free formulation |
| K-Y Jelly (McNeil) | Water, glycerin, hydroxyethylcellulose, parabens, and chlorhexidine | |
| Summer’s Eve Lubricant (C.B. Fleet) | Water, propylene glycol, methylcellulose, xanthan gum, sodium lactate, methylparaben, lactic acid, dextrose, sodium chloride, edatate disodium, pectin, and propylparaben | |
| FemGlide (WalMed) | Water, polyoxyethylene, methylparaben, and sodium carbomer | |
| Pre-Seed (INGfertility) | Water, hydroxyethylcellulose, arabinogalactan, paraben, and Pluronic copolymers | Promoted to women and their partners who are trying to conceive |
| Silicone-based | ||
| ID Millennium (Westridge Laboratories) | Cyclomethicone, dimethicone, and dimethiconol | Less drying than other lubricants |
| Pjur | Cyclopentasiloxane, dimethicone, and dimethiconol | Compatible with a condom |
| Pink | Dimethicone, vitamin E, aloe vera, dimethiconol, and cyclomethicone | |
| K-Y Liquibeads (McNeil) | Dimethicone, gelatin, glycerin, and dimethiconol | Active ingredients are contained in so-called ovules that release lubricant over several days |
| Oil-based | ||
| Élégance Women’s Lubricant | Natural oils | Does not contain alcohol, glycerin, or parabens; is incompatible with a condom; helpful for women who have vulvodynia or vestibulitis |
What to offer when estrogen is not an option
Some women may want to avoid hormonal treatment, or have a contraindication to it, such as estrogen-receptor–positive breast cancer.8 In premenopausal women, vaginal atrophy can occur with lactation or postpartum hormonal changes, or may result from the use of anti-estrogenic agents for breast cancer. Other candidates for nonhormonal therapy are women who have chronic vulvar pain syndromes. In these women, vaginal lubricants can be especially useful.
Although they are less effective than estrogen, vaginal moisturizers, such as Replens, have been shown to reverse symptoms of vaginal atrophy and decrease discomfort during intercourse.9
The therapeutic options for vaginal atrophy are likely to broaden in the near future. Ospemifene (Ophena), a selective estrogen receptor modulator (SERM) under development for the treatment of vaginal atrophy, has reached the end of Phase-3 clinical study, with positive efficacy results. A long-term safety study of the orally administered SERM has also been completed, reports QuatRx, the drug’s manufacturer. The company expects to file a New Drug Application with the US Food and Drug Administration early next year.
Phase-3 trials documented significant improvement in dryness, dyspareunia, and other endpoints
The first Phase-3 study of Ophena was announced by QuatRx in January 2008 and presented at the 90th annual meeting of the Endocrine Society. Women who were treated with 60 mg daily of Ophena experienced statistically significant improvement in vaginal dryness, dyspareunia, and the proportion of parabasal and superficial cells in the epithelium of vaginal walls. The vaginal pH level also declined. The drug did not cause hot flushes among users.
The second Phase-3 study was a randomized, double-blind, placebo-controlled study of 919 women who had vulvovaginal atrophy. It was conducted at 116 sites in the United States. Among the cohort of 605 women who identified dyspareunia as their most bothersome symptom, positive efficacy results were achieved in all four primary endpoints, including:
- a decrease in parabasal cells
- an increase in superficial cells
- a decrease in the vaginal pH level
- improvement in dyspareunia.
The trial demonstrated statistically significant improvement from baseline to week 12 in all four endpoints, compared with placebo (P.0001>
All women were supplied with a nonhormonal vaginal lubricant to be used as needed during the treatment period; the study found efficacy above and beyond usage of this lubricant, according to a press release from QuatRx.
Is the benefit worth the risk?
Some have questioned whether a systemic drug is overkill for a complaint like postmenopausal vaginal atrophy.14 Because Ophena is a SERM, it is likely to carry a cardiovascular risk profile similar to that of other drugs in its class. For example, in a large randomized trial, raloxifene (Evista) failed to reduce coronary artery disease and significantly increased the incidence of fatal stroke and venous thromboembolism.15 When local estrogen formulations that do not carry such risks are already available, some experts question the advisability of developing another systemic agent.
Another question: Is it realistic to expect the patient to take a drug every day when her chief complaint is postmenopausal dyspareunia and she is likely to have intercourse only once or twice a week?
These questions probably won’t be addressed until the drug enters the market—and physicians and their patients will be the ones providing the answers.
Specialty lubricants are unproven
In contrast to products designed to treat vaginal dryness and atrophy, some lubricants are marketed specifically for sexual enhancement. Warming lubricants cause a heating sensation on the skin and usually contain menthol, L-arginine, or capsaicin. Natural and artificial flavors are used to manufacture flavored lubricants.
None of these products have been scientifically proven to enhance sexual function.
Oil-based lubricants may impede condom integrity
It is estimated that 40% of couples who use condoms also use a lubricant to assist with intercourse.10 The integrity of latex condoms has been shown to deteriorate with the use of an oil-based lubricant or petrolatum. One study, in which the mean burst time of condoms was assessed during pressurized air inflation, showed a significant reduction in that time when vaginal lubricants that contained mineral or vegetable oil were used.11
Oil-based lubricants also have been shown to increase the slippage rate, with a trend toward increased breakage.10
Water-based lubricants may slightly increase slippage, but they reduce breakage.
Women should avoid oil-based lubricants when their partner uses a condom.
Some lubricants affect sperm quality
Choosing a vaginal lubricant can be of particular concern to a woman who is being treated for infertility. Lubricants may affect the integrity and function of sperm, even if they do not contain spermicide. Noncommercial products, such as glycerin, olive oil, vegetable oil, and, even, saliva have been associated with a loss of sperm function.12
A recent study found that Replens and Astroglide cause a dramatic decrease in sperm motility. FemGlide causes less of a decrease—but still a significant one.12 The nonphysiologic osmolality and pH of these products may be the cause of such sperm damage. Pre-Seed, which has a more physiologic pH level and isotonic quality, was found to cause minimal harm to sperm motility and chromatin quality.12
Avoid propylene glycol in women who have vulvodynia
Vaginal lubricants and moisturizers are also used in the treatment of vulvodynia or chronic vulvar pain syndromes. According to an ACOG Committee Opinion, topical application of preservative-free solutions, such as vegetable oil or plain petrolatum, is recommended to hold moisture within the tissues and provide a protective barrier.13 Adequate lubrication is also recommended during intercourse.
Products that contain propylene glycol, or alcohol, may act as an irritant in women who experience local pain and heightened vaginal sensitivity. For that reason, such products should be avoided in this population.
CASE RESOLVED
Careful examination reveals urogenital atrophy with absence of any fungal or bacterial infection of the vulva or vagina. The patient chooses to use a water-based lubricant during sexual relations and begins using intravaginal estradiol tablets (other options include the vaginal ring or cream). Symptoms of dyspareunia disappear almost immediately, and vaginal burning improves after 6 weeks.
1. Al-Baghdadi O, Ewies AA. Topical estrogen therapy in the management of postmenopausal vaginal atrophy: an up-to-date overview. Climacteric. 2009;12:91-105.
2. Willhite LA, O’Connell MB. Urogenital atrophy: prevention and treatment. Pharmacotherapy. 2001;21:464-480.
3. Archer DF. Efficacy and tolerability of local estrogen therapy for urogenital atrophy. Menopause. 2009;17:1-10.
4. Dennerstein L, Dudley EC, Hopper JL, Guthrie JR, Burger HG. A prospective population-based study of menopausal symptoms. Obstet Gynecol. 2000;96:351-358.
5. Suckling J, Lethaby A, Kennedy R. Local oestrogen for vaginal atrophy in postmenopausal women. Cochrane Database Syst Rev. 2006;(4):CD001500.-
6. Anderson GL, Limacher M, Assaf AR, et al. Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial. JAMA. 2004;291:1701-1712.
7. Oberg K, Fugl-Meyer AR, Fugl-Meyer KS. On categorization and quantification of women’s sexual dysfunctions: an epidemiological approach. Int J Impot Res. 2004;16:261-269.
8. Loprinzi CL, Wolf SL, Barton DL, Laack NN. Symptom management in premenopausal patients with breast cancer. Lancet Oncol. 2008;9:993-1001.
9. Bygdeman M, Swahn ML. Replens versus dienoestrol cream in the symptomatic treatment of vaginal atrophy in postmenopausal women. Maturitas. 1996;23:259-263.
10. Steiner M, Piedrahita C, Glover L, Joanis C, Spruyt A, Foldesy R. The impact of lubricants on latex condoms during vaginal intercourse. Int J STD AIDS. 1994;5:29-36.
11. Rosen AD, Rosen T. Study of condom integrity after brief exposure to over-the-counter vaginal preparations. South Med J. 1999;92:305-307.
12. Agarwal A, Deepinder F, Cocuzza M, Short RA, Evenson DP. Effect of vaginal lubricants on sperm motility and chromatin integrity: a prospective comparative study. Fertil Steril. 2008;89:375-379.
13. ACOG Committee Opinion #345: Vulvodynia. October 2006. ACOG Committee on Gynecologic Practice. Obstet Gynecol. 2006;108:1049-1052.
14. Ophena is a me-too drug with an impractical mode of administration. Gerson Lehrman Group. Jan. 14, 2008. Available at: http://www.glgroup.com/News/Ophena-is-a-Me-Too-Drug-with-an-Impractical-Mode-of-Administration-20651.html. Accessed Nov. 11, 2009.
15. de Villiers TJ. Clinical issues regarding cardiovascular disease and selective estrogen receptor modulators in postmenopausal women. Climacteric. 2009;12 Suppl 1:108-111.
1. Al-Baghdadi O, Ewies AA. Topical estrogen therapy in the management of postmenopausal vaginal atrophy: an up-to-date overview. Climacteric. 2009;12:91-105.
2. Willhite LA, O’Connell MB. Urogenital atrophy: prevention and treatment. Pharmacotherapy. 2001;21:464-480.
3. Archer DF. Efficacy and tolerability of local estrogen therapy for urogenital atrophy. Menopause. 2009;17:1-10.
4. Dennerstein L, Dudley EC, Hopper JL, Guthrie JR, Burger HG. A prospective population-based study of menopausal symptoms. Obstet Gynecol. 2000;96:351-358.
5. Suckling J, Lethaby A, Kennedy R. Local oestrogen for vaginal atrophy in postmenopausal women. Cochrane Database Syst Rev. 2006;(4):CD001500.-
6. Anderson GL, Limacher M, Assaf AR, et al. Women’s Health Initiative Steering Committee. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial. JAMA. 2004;291:1701-1712.
7. Oberg K, Fugl-Meyer AR, Fugl-Meyer KS. On categorization and quantification of women’s sexual dysfunctions: an epidemiological approach. Int J Impot Res. 2004;16:261-269.
8. Loprinzi CL, Wolf SL, Barton DL, Laack NN. Symptom management in premenopausal patients with breast cancer. Lancet Oncol. 2008;9:993-1001.
9. Bygdeman M, Swahn ML. Replens versus dienoestrol cream in the symptomatic treatment of vaginal atrophy in postmenopausal women. Maturitas. 1996;23:259-263.
10. Steiner M, Piedrahita C, Glover L, Joanis C, Spruyt A, Foldesy R. The impact of lubricants on latex condoms during vaginal intercourse. Int J STD AIDS. 1994;5:29-36.
11. Rosen AD, Rosen T. Study of condom integrity after brief exposure to over-the-counter vaginal preparations. South Med J. 1999;92:305-307.
12. Agarwal A, Deepinder F, Cocuzza M, Short RA, Evenson DP. Effect of vaginal lubricants on sperm motility and chromatin integrity: a prospective comparative study. Fertil Steril. 2008;89:375-379.
13. ACOG Committee Opinion #345: Vulvodynia. October 2006. ACOG Committee on Gynecologic Practice. Obstet Gynecol. 2006;108:1049-1052.
14. Ophena is a me-too drug with an impractical mode of administration. Gerson Lehrman Group. Jan. 14, 2008. Available at: http://www.glgroup.com/News/Ophena-is-a-Me-Too-Drug-with-an-Impractical-Mode-of-Administration-20651.html. Accessed Nov. 11, 2009.
15. de Villiers TJ. Clinical issues regarding cardiovascular disease and selective estrogen receptor modulators in postmenopausal women. Climacteric. 2009;12 Suppl 1:108-111.