User login
Evidence builds linking anticoagulation to COVID-19 survival
, a large study from the epicenter of the U.S. outbreak suggests.
Among nearly 3,000 patients with COVID-19 admitted to New York City’s Mount Sinai Health System beginning in mid-March, median survival increased from 14 days to 21 days with the addition of anticoagulation.
The results were particularly striking among sicker patients who required mechanical ventilation, in whom in-hospital mortality fell from 62.7% to 29.1% and median survival jumped from 9 days to 21 days.
Interestingly, the association with anticoagulation and improved survival remained even after adjusting for mechanical ventilation, the authors reported May 6 in the Journal of the American College of Cardiology.
“It’s important for the community to know, first of all, how this should be approached and, second, it’s really opening a door to a new reality,” senior corresponding author Valentin Fuster, MD, PhD, director of Mount Sinai’s Zena and Michael A. Wiener Cardiovascular Institute and JACC editor-in-chief.
“I can tell you any family of mine who will have this disease absolutely will be on antithrombotic therapy and, actually, so are all of the patients at Mount Sinai now,” he said in an interview. COVID-19 is thought to promote thrombosis but the exact role of anticoagulation in the management of COVID-19 and optimal regimen are unknown.
In late March, the International Society on Thrombosis and Haemostasis recommended that all hospitalized COVID-19 patients, even those not in the ICU, should receive prophylactic-dose low-molecular-weight heparin (LMWH), unless they have contraindications.
Last month, international consensus-based recommendations were published for the diagnosis and management of thrombotic disease in patients with COVID-19.
In early March, however, data were scare and only a minimal number of patients were receiving anticoagulants at Mount Sinai.
“But after a few weeks, we reached an intuitive feeling that anticoagulation was of benefit and, at the same time, the literature was beginning to say clots were important in this disease,” Dr. Fuster said. “So we took a very straightforward approach and set up a policy in our institution that all COVID-19 patients should be on antithrombotic therapy. It was a decision made without data, but it was a feeling.”
For the present study, the researchers examined mortality and bleeding among 2,773 patients hospitalized at Mount Sinai with confirmed COVID-19 between March 14 and April 11.
Of these, 786 (28%) received systemic anticoagulation including subcutaneous heparin, LMWH, fractionated heparin, and the novel oral anticoagulants apixaban and dabigatran, for a median of 3 days (range, 2-7 days). Tissue plasminogen activator was also used in some ICU cases.
Major bleeding was defined as hemoglobin less than 7 g/dL and any red blood cell transfusion; at least two units of red blood cell transfusion within 48 hours; or a diagnosis code for major bleeding, notably including intracranial hemorrhage.
Patients treated with anticoagulation were more likely to require invasive mechanical ventilation (29.8% vs. 8.1%) and to have significantly increased prothrombin time, activated partial thromboplastin time, lactate dehydrogenase, ferritin, C-reactive protein, and d-dimer values. In-hospital mortality was 22.5% with anticoagulation and 22.8% without anticoagulation (median survival, 14 days vs. 21 days).
In multivariate analysis, longer anticoagulation duration was associated with a 14% lower adjusted risk of in-hospital death (hazard ratio, 0.86 per day; 95% confidence interval, 0.82-0.89; P < .001).
The model adjusted for several potential confounders such as age, ethnicity, body mass index, and prehospital anticoagulation use. To adjust for differential length of stay and anticoagulation initiation, anticoagulation duration was used as a covariate and intubation was treated as a time-dependent variable.
Bleeding events were similar in patients treated with and without anticoagulation (3% vs. 1.9%; P = .2) but were more common among the 375 intubated patients than among nonintubated patients (7.5% vs. 1.35%; P value not given). “The most important thing was there was no increase in bleeding,” said Dr. Fuster.
Additional support for a possible survival benefit was published April 27 and included 449 patients with severe COVID-19 treated with heparin (mostly LMWH) for at least 7 days in Hunan, China. Overall, 28-day mortality was similar between heparin users and nonusers (30.3% vs. 29.7%) but was significantly lower among heparin users who had a Sepsis-Induced Coagulopathy score of at least 4 (40% vs. 64.2%; P = .02) or d-dimer greater than sixfold the upper limit of normal (32.8% vs. 52.4%; P = .01).
In multivariate analysis, d-dimer, prothrombin time, and age were positively correlated with 28-day mortality, and platelet count was negatively correlated with 28-day mortality.
Victor F. Tapson, MD, who directs the pulmonary embolism response team at Cedars-Sinai Medical Center in Los Angeles and was not involved with the study, said, “The Chinese data were not enough for me to anticoagulate patients therapeutically” but the Mount Sinai data strengthen the case.
“They’re wise to call this a ‘suggestion of improved outcomes,’ but it’s pretty compelling that those patients who were on anticoagulation had improved survival after adjusting for mechanical ventilation,” he said in an interview. “These are sicker patients and sicker patients may get anticoagulated more, but they may bleed more. The bleed risks were a little different but they didn’t seem too concerning.”
“I think this helps move us forward some that we should consider anticoagulating with therapeutic anticoagulation certain patients that meet certain criteria,” Dr. Tapson said. “An easy example is a patient who comes to the hospital, has active cancer and is on a DOAC [direct oral anticoagulant], and comes up with COVID.”
At the same time, some clinicians want to increase prophylactic anticoagulation “using enoxaparin 40 mg once a day and maybe go to twice a day – not quite therapeutic doses but increased prophylaxis,” he observed. Anticoagulation was given at “relatively low doses” in the Mount Sinai study but that is evolving in light of the reassuring bleeding data, Dr. Fuster said. They now have three enoxaparin regimens and, for example, give patients who don’t require intensive care enoxaparin 30 mg twice a day, up from 40 mg a day initially.
Patients are also stratified by factors such as renal failure and obesity, creating an intermediate group between those not initially needing intensive care and ICU cases.
In the coming weeks, the researchers will evaluate anticoagulation regimens and a broader array of outcomes among 5,000 patients, two-thirds of whom received anticoagulation after Mount Sinai enacted its anticoagulation policy. “We’re now going to look at the difference between all these [regimens],” Dr. Fuster said. “My personal feeling and, for feasibility issues, I hope the winner is subcutaneous heparin.”
Three randomized trials are also planned. “Three questions we really want to ask are: what to give in the hospital, what to give those who go home after the hospital, and what to give those who are not hospitalized,” he said.
The work was supported by U54 TR001433-05, National Center for Advancing Translational Sciences, National Institutes of Health. Dr. Fuster has disclosed no relevant financial relationships. Dr. Tapson reported consulting and clinical trial work for BMS, Janssen, Daiichi Medical, ECOS/BTG, Inari, and Penumbra.
A version of this article originally appeared on Medscape.com.
, a large study from the epicenter of the U.S. outbreak suggests.
Among nearly 3,000 patients with COVID-19 admitted to New York City’s Mount Sinai Health System beginning in mid-March, median survival increased from 14 days to 21 days with the addition of anticoagulation.
The results were particularly striking among sicker patients who required mechanical ventilation, in whom in-hospital mortality fell from 62.7% to 29.1% and median survival jumped from 9 days to 21 days.
Interestingly, the association with anticoagulation and improved survival remained even after adjusting for mechanical ventilation, the authors reported May 6 in the Journal of the American College of Cardiology.
“It’s important for the community to know, first of all, how this should be approached and, second, it’s really opening a door to a new reality,” senior corresponding author Valentin Fuster, MD, PhD, director of Mount Sinai’s Zena and Michael A. Wiener Cardiovascular Institute and JACC editor-in-chief.
“I can tell you any family of mine who will have this disease absolutely will be on antithrombotic therapy and, actually, so are all of the patients at Mount Sinai now,” he said in an interview. COVID-19 is thought to promote thrombosis but the exact role of anticoagulation in the management of COVID-19 and optimal regimen are unknown.
In late March, the International Society on Thrombosis and Haemostasis recommended that all hospitalized COVID-19 patients, even those not in the ICU, should receive prophylactic-dose low-molecular-weight heparin (LMWH), unless they have contraindications.
Last month, international consensus-based recommendations were published for the diagnosis and management of thrombotic disease in patients with COVID-19.
In early March, however, data were scare and only a minimal number of patients were receiving anticoagulants at Mount Sinai.
“But after a few weeks, we reached an intuitive feeling that anticoagulation was of benefit and, at the same time, the literature was beginning to say clots were important in this disease,” Dr. Fuster said. “So we took a very straightforward approach and set up a policy in our institution that all COVID-19 patients should be on antithrombotic therapy. It was a decision made without data, but it was a feeling.”
For the present study, the researchers examined mortality and bleeding among 2,773 patients hospitalized at Mount Sinai with confirmed COVID-19 between March 14 and April 11.
Of these, 786 (28%) received systemic anticoagulation including subcutaneous heparin, LMWH, fractionated heparin, and the novel oral anticoagulants apixaban and dabigatran, for a median of 3 days (range, 2-7 days). Tissue plasminogen activator was also used in some ICU cases.
Major bleeding was defined as hemoglobin less than 7 g/dL and any red blood cell transfusion; at least two units of red blood cell transfusion within 48 hours; or a diagnosis code for major bleeding, notably including intracranial hemorrhage.
Patients treated with anticoagulation were more likely to require invasive mechanical ventilation (29.8% vs. 8.1%) and to have significantly increased prothrombin time, activated partial thromboplastin time, lactate dehydrogenase, ferritin, C-reactive protein, and d-dimer values. In-hospital mortality was 22.5% with anticoagulation and 22.8% without anticoagulation (median survival, 14 days vs. 21 days).
In multivariate analysis, longer anticoagulation duration was associated with a 14% lower adjusted risk of in-hospital death (hazard ratio, 0.86 per day; 95% confidence interval, 0.82-0.89; P < .001).
The model adjusted for several potential confounders such as age, ethnicity, body mass index, and prehospital anticoagulation use. To adjust for differential length of stay and anticoagulation initiation, anticoagulation duration was used as a covariate and intubation was treated as a time-dependent variable.
Bleeding events were similar in patients treated with and without anticoagulation (3% vs. 1.9%; P = .2) but were more common among the 375 intubated patients than among nonintubated patients (7.5% vs. 1.35%; P value not given). “The most important thing was there was no increase in bleeding,” said Dr. Fuster.
Additional support for a possible survival benefit was published April 27 and included 449 patients with severe COVID-19 treated with heparin (mostly LMWH) for at least 7 days in Hunan, China. Overall, 28-day mortality was similar between heparin users and nonusers (30.3% vs. 29.7%) but was significantly lower among heparin users who had a Sepsis-Induced Coagulopathy score of at least 4 (40% vs. 64.2%; P = .02) or d-dimer greater than sixfold the upper limit of normal (32.8% vs. 52.4%; P = .01).
In multivariate analysis, d-dimer, prothrombin time, and age were positively correlated with 28-day mortality, and platelet count was negatively correlated with 28-day mortality.
Victor F. Tapson, MD, who directs the pulmonary embolism response team at Cedars-Sinai Medical Center in Los Angeles and was not involved with the study, said, “The Chinese data were not enough for me to anticoagulate patients therapeutically” but the Mount Sinai data strengthen the case.
“They’re wise to call this a ‘suggestion of improved outcomes,’ but it’s pretty compelling that those patients who were on anticoagulation had improved survival after adjusting for mechanical ventilation,” he said in an interview. “These are sicker patients and sicker patients may get anticoagulated more, but they may bleed more. The bleed risks were a little different but they didn’t seem too concerning.”
“I think this helps move us forward some that we should consider anticoagulating with therapeutic anticoagulation certain patients that meet certain criteria,” Dr. Tapson said. “An easy example is a patient who comes to the hospital, has active cancer and is on a DOAC [direct oral anticoagulant], and comes up with COVID.”
At the same time, some clinicians want to increase prophylactic anticoagulation “using enoxaparin 40 mg once a day and maybe go to twice a day – not quite therapeutic doses but increased prophylaxis,” he observed. Anticoagulation was given at “relatively low doses” in the Mount Sinai study but that is evolving in light of the reassuring bleeding data, Dr. Fuster said. They now have three enoxaparin regimens and, for example, give patients who don’t require intensive care enoxaparin 30 mg twice a day, up from 40 mg a day initially.
Patients are also stratified by factors such as renal failure and obesity, creating an intermediate group between those not initially needing intensive care and ICU cases.
In the coming weeks, the researchers will evaluate anticoagulation regimens and a broader array of outcomes among 5,000 patients, two-thirds of whom received anticoagulation after Mount Sinai enacted its anticoagulation policy. “We’re now going to look at the difference between all these [regimens],” Dr. Fuster said. “My personal feeling and, for feasibility issues, I hope the winner is subcutaneous heparin.”
Three randomized trials are also planned. “Three questions we really want to ask are: what to give in the hospital, what to give those who go home after the hospital, and what to give those who are not hospitalized,” he said.
The work was supported by U54 TR001433-05, National Center for Advancing Translational Sciences, National Institutes of Health. Dr. Fuster has disclosed no relevant financial relationships. Dr. Tapson reported consulting and clinical trial work for BMS, Janssen, Daiichi Medical, ECOS/BTG, Inari, and Penumbra.
A version of this article originally appeared on Medscape.com.
, a large study from the epicenter of the U.S. outbreak suggests.
Among nearly 3,000 patients with COVID-19 admitted to New York City’s Mount Sinai Health System beginning in mid-March, median survival increased from 14 days to 21 days with the addition of anticoagulation.
The results were particularly striking among sicker patients who required mechanical ventilation, in whom in-hospital mortality fell from 62.7% to 29.1% and median survival jumped from 9 days to 21 days.
Interestingly, the association with anticoagulation and improved survival remained even after adjusting for mechanical ventilation, the authors reported May 6 in the Journal of the American College of Cardiology.
“It’s important for the community to know, first of all, how this should be approached and, second, it’s really opening a door to a new reality,” senior corresponding author Valentin Fuster, MD, PhD, director of Mount Sinai’s Zena and Michael A. Wiener Cardiovascular Institute and JACC editor-in-chief.
“I can tell you any family of mine who will have this disease absolutely will be on antithrombotic therapy and, actually, so are all of the patients at Mount Sinai now,” he said in an interview. COVID-19 is thought to promote thrombosis but the exact role of anticoagulation in the management of COVID-19 and optimal regimen are unknown.
In late March, the International Society on Thrombosis and Haemostasis recommended that all hospitalized COVID-19 patients, even those not in the ICU, should receive prophylactic-dose low-molecular-weight heparin (LMWH), unless they have contraindications.
Last month, international consensus-based recommendations were published for the diagnosis and management of thrombotic disease in patients with COVID-19.
In early March, however, data were scare and only a minimal number of patients were receiving anticoagulants at Mount Sinai.
“But after a few weeks, we reached an intuitive feeling that anticoagulation was of benefit and, at the same time, the literature was beginning to say clots were important in this disease,” Dr. Fuster said. “So we took a very straightforward approach and set up a policy in our institution that all COVID-19 patients should be on antithrombotic therapy. It was a decision made without data, but it was a feeling.”
For the present study, the researchers examined mortality and bleeding among 2,773 patients hospitalized at Mount Sinai with confirmed COVID-19 between March 14 and April 11.
Of these, 786 (28%) received systemic anticoagulation including subcutaneous heparin, LMWH, fractionated heparin, and the novel oral anticoagulants apixaban and dabigatran, for a median of 3 days (range, 2-7 days). Tissue plasminogen activator was also used in some ICU cases.
Major bleeding was defined as hemoglobin less than 7 g/dL and any red blood cell transfusion; at least two units of red blood cell transfusion within 48 hours; or a diagnosis code for major bleeding, notably including intracranial hemorrhage.
Patients treated with anticoagulation were more likely to require invasive mechanical ventilation (29.8% vs. 8.1%) and to have significantly increased prothrombin time, activated partial thromboplastin time, lactate dehydrogenase, ferritin, C-reactive protein, and d-dimer values. In-hospital mortality was 22.5% with anticoagulation and 22.8% without anticoagulation (median survival, 14 days vs. 21 days).
In multivariate analysis, longer anticoagulation duration was associated with a 14% lower adjusted risk of in-hospital death (hazard ratio, 0.86 per day; 95% confidence interval, 0.82-0.89; P < .001).
The model adjusted for several potential confounders such as age, ethnicity, body mass index, and prehospital anticoagulation use. To adjust for differential length of stay and anticoagulation initiation, anticoagulation duration was used as a covariate and intubation was treated as a time-dependent variable.
Bleeding events were similar in patients treated with and without anticoagulation (3% vs. 1.9%; P = .2) but were more common among the 375 intubated patients than among nonintubated patients (7.5% vs. 1.35%; P value not given). “The most important thing was there was no increase in bleeding,” said Dr. Fuster.
Additional support for a possible survival benefit was published April 27 and included 449 patients with severe COVID-19 treated with heparin (mostly LMWH) for at least 7 days in Hunan, China. Overall, 28-day mortality was similar between heparin users and nonusers (30.3% vs. 29.7%) but was significantly lower among heparin users who had a Sepsis-Induced Coagulopathy score of at least 4 (40% vs. 64.2%; P = .02) or d-dimer greater than sixfold the upper limit of normal (32.8% vs. 52.4%; P = .01).
In multivariate analysis, d-dimer, prothrombin time, and age were positively correlated with 28-day mortality, and platelet count was negatively correlated with 28-day mortality.
Victor F. Tapson, MD, who directs the pulmonary embolism response team at Cedars-Sinai Medical Center in Los Angeles and was not involved with the study, said, “The Chinese data were not enough for me to anticoagulate patients therapeutically” but the Mount Sinai data strengthen the case.
“They’re wise to call this a ‘suggestion of improved outcomes,’ but it’s pretty compelling that those patients who were on anticoagulation had improved survival after adjusting for mechanical ventilation,” he said in an interview. “These are sicker patients and sicker patients may get anticoagulated more, but they may bleed more. The bleed risks were a little different but they didn’t seem too concerning.”
“I think this helps move us forward some that we should consider anticoagulating with therapeutic anticoagulation certain patients that meet certain criteria,” Dr. Tapson said. “An easy example is a patient who comes to the hospital, has active cancer and is on a DOAC [direct oral anticoagulant], and comes up with COVID.”
At the same time, some clinicians want to increase prophylactic anticoagulation “using enoxaparin 40 mg once a day and maybe go to twice a day – not quite therapeutic doses but increased prophylaxis,” he observed. Anticoagulation was given at “relatively low doses” in the Mount Sinai study but that is evolving in light of the reassuring bleeding data, Dr. Fuster said. They now have three enoxaparin regimens and, for example, give patients who don’t require intensive care enoxaparin 30 mg twice a day, up from 40 mg a day initially.
Patients are also stratified by factors such as renal failure and obesity, creating an intermediate group between those not initially needing intensive care and ICU cases.
In the coming weeks, the researchers will evaluate anticoagulation regimens and a broader array of outcomes among 5,000 patients, two-thirds of whom received anticoagulation after Mount Sinai enacted its anticoagulation policy. “We’re now going to look at the difference between all these [regimens],” Dr. Fuster said. “My personal feeling and, for feasibility issues, I hope the winner is subcutaneous heparin.”
Three randomized trials are also planned. “Three questions we really want to ask are: what to give in the hospital, what to give those who go home after the hospital, and what to give those who are not hospitalized,” he said.
The work was supported by U54 TR001433-05, National Center for Advancing Translational Sciences, National Institutes of Health. Dr. Fuster has disclosed no relevant financial relationships. Dr. Tapson reported consulting and clinical trial work for BMS, Janssen, Daiichi Medical, ECOS/BTG, Inari, and Penumbra.
A version of this article originally appeared on Medscape.com.
Coronary CT angiography gives superior MI risk prediction
In patients with stable chest pain, the burden of low-attenuation noncalcified plaque on coronary CT angiography is a better predictor of future myocardial infarction risk than a cardiovascular risk score, an Agatson coronary artery calcium score, or angiographic severity of coronary stenoses, Michelle C. Williams, MBChB, PhD, reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
These findings from a post hoc analysis of the large multicenter SCOT-HEART trial challenge current concepts regarding the supposed superiority of the classic tools for MI risk prediction, noted Dr. Williams, a senior clinical research fellow at the University of Edinburgh.
Indeed, it’s likely that the current established predictors of risk – that is, coronary artery calcium, severity of stenosis, and cardiovascular risk score – are associated with clinical events only indirectly through their correlation with low-attenuated calcified plaque burden, which is the real driver of future MI, she continued.
Histologically, low-attenuated noncalcified plaque on coronary CT angiography (CCTA) is defined by a thin fibrous cap, a large, inflamed, lipid-rich necrotic core, and microcalcification. Previously, Dr. Williams and her coinvestigators demonstrated that visual identification of this unstable plaque subtype is of benefit in predicting future risk of MI (J Am Coll Cardiol. 2019 Jan 29;73[3]:291-301).
But visual identification of plaque subtypes is a crude and laborious process. In her current study, she and her coworkers have taken things a giant step further, using commercially available CCTA software to semiautomatically quantify the burden of this highest-risk plaque subtype as well as all the other subtypes.
This post hoc analysis of the previously reported main SCOT-HEART trial (N Engl J Med. 2018 Sep 6;379[10]:924-933) included 1,769 patients with stable chest pain randomized to standard care with or without CCTA guidance and followed for a median of 4.7 years, during which 41 patients had a fatal or nonfatal MI. At enrollment, 37% of participants had normal coronary arteries, 38% had nonobstructive coronary artery disease (CAD), and the remainder had obstructive CAD.
In a multivariate analysis, low-attenuation noncalcified plaque burden was the strongest predictor of future MI, with an adjusted hazard ratio of 1.6 per doubling. This metric was strongly correlated with coronary artery calcium score, underscoring the limited value of doing noncontrast CT in order to determine a coronary artery calcium score when CCTA is performed.
Low-attenuation plaque burden correlated very strongly with angiographic severity of stenosis, and only weakly with cardiovascular risk score, perhaps explaining the poor prognostic performance of cardiovascular risk scores in SCOT-HEART and other studies, according to Dr. Williams.
Patients with a low-attenuation noncalcified plaque burden greater than 4% in their coronary tree were 4.7 times more likely to have a subsequent MI than were those with a lesser burden. The predictive power was even greater in patients with nonobstructive CAD, where a low-attenuation noncalcified plaque burden in excess of 4% conferred a 6.6-fold greater likelihood of fatal or nonfatal MI, she observed.
Two things need to happen before measurement of low-attenuation noncalcified plaque via CCTA to predict MI risk is ready to be adopted in routine clinical practice, according to Dr. Williams. These SCOT-HEART results need to be validated in other cohorts, a process now underway in the SCOT-HEART 2 trial and other studies. Also, improved software incorporating machine learning is needed in order to speed up the semiautomated analysis of plaque subtypes, which now takes 20-30 minutes.
Dr. Williams reported having no financial conflicts regarding her study, funded by the National Health Service.
In conjunction with her virtual presentation at ACC 2020, the SCOT-HEART study results were published online (Circulation. 2020 Mar 16. doi: 10.1161/CIRCULATIONAHA.119.044720. [Epub ahead of print]).
SOURCE: Williams MC et al. ACC 2020, Abstract 909-06.
In patients with stable chest pain, the burden of low-attenuation noncalcified plaque on coronary CT angiography is a better predictor of future myocardial infarction risk than a cardiovascular risk score, an Agatson coronary artery calcium score, or angiographic severity of coronary stenoses, Michelle C. Williams, MBChB, PhD, reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
These findings from a post hoc analysis of the large multicenter SCOT-HEART trial challenge current concepts regarding the supposed superiority of the classic tools for MI risk prediction, noted Dr. Williams, a senior clinical research fellow at the University of Edinburgh.
Indeed, it’s likely that the current established predictors of risk – that is, coronary artery calcium, severity of stenosis, and cardiovascular risk score – are associated with clinical events only indirectly through their correlation with low-attenuated calcified plaque burden, which is the real driver of future MI, she continued.
Histologically, low-attenuated noncalcified plaque on coronary CT angiography (CCTA) is defined by a thin fibrous cap, a large, inflamed, lipid-rich necrotic core, and microcalcification. Previously, Dr. Williams and her coinvestigators demonstrated that visual identification of this unstable plaque subtype is of benefit in predicting future risk of MI (J Am Coll Cardiol. 2019 Jan 29;73[3]:291-301).
But visual identification of plaque subtypes is a crude and laborious process. In her current study, she and her coworkers have taken things a giant step further, using commercially available CCTA software to semiautomatically quantify the burden of this highest-risk plaque subtype as well as all the other subtypes.
This post hoc analysis of the previously reported main SCOT-HEART trial (N Engl J Med. 2018 Sep 6;379[10]:924-933) included 1,769 patients with stable chest pain randomized to standard care with or without CCTA guidance and followed for a median of 4.7 years, during which 41 patients had a fatal or nonfatal MI. At enrollment, 37% of participants had normal coronary arteries, 38% had nonobstructive coronary artery disease (CAD), and the remainder had obstructive CAD.
In a multivariate analysis, low-attenuation noncalcified plaque burden was the strongest predictor of future MI, with an adjusted hazard ratio of 1.6 per doubling. This metric was strongly correlated with coronary artery calcium score, underscoring the limited value of doing noncontrast CT in order to determine a coronary artery calcium score when CCTA is performed.
Low-attenuation plaque burden correlated very strongly with angiographic severity of stenosis, and only weakly with cardiovascular risk score, perhaps explaining the poor prognostic performance of cardiovascular risk scores in SCOT-HEART and other studies, according to Dr. Williams.
Patients with a low-attenuation noncalcified plaque burden greater than 4% in their coronary tree were 4.7 times more likely to have a subsequent MI than were those with a lesser burden. The predictive power was even greater in patients with nonobstructive CAD, where a low-attenuation noncalcified plaque burden in excess of 4% conferred a 6.6-fold greater likelihood of fatal or nonfatal MI, she observed.
Two things need to happen before measurement of low-attenuation noncalcified plaque via CCTA to predict MI risk is ready to be adopted in routine clinical practice, according to Dr. Williams. These SCOT-HEART results need to be validated in other cohorts, a process now underway in the SCOT-HEART 2 trial and other studies. Also, improved software incorporating machine learning is needed in order to speed up the semiautomated analysis of plaque subtypes, which now takes 20-30 minutes.
Dr. Williams reported having no financial conflicts regarding her study, funded by the National Health Service.
In conjunction with her virtual presentation at ACC 2020, the SCOT-HEART study results were published online (Circulation. 2020 Mar 16. doi: 10.1161/CIRCULATIONAHA.119.044720. [Epub ahead of print]).
SOURCE: Williams MC et al. ACC 2020, Abstract 909-06.
In patients with stable chest pain, the burden of low-attenuation noncalcified plaque on coronary CT angiography is a better predictor of future myocardial infarction risk than a cardiovascular risk score, an Agatson coronary artery calcium score, or angiographic severity of coronary stenoses, Michelle C. Williams, MBChB, PhD, reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation. The meeting was conducted online after its cancellation because of the COVID-19 pandemic.
These findings from a post hoc analysis of the large multicenter SCOT-HEART trial challenge current concepts regarding the supposed superiority of the classic tools for MI risk prediction, noted Dr. Williams, a senior clinical research fellow at the University of Edinburgh.
Indeed, it’s likely that the current established predictors of risk – that is, coronary artery calcium, severity of stenosis, and cardiovascular risk score – are associated with clinical events only indirectly through their correlation with low-attenuated calcified plaque burden, which is the real driver of future MI, she continued.
Histologically, low-attenuated noncalcified plaque on coronary CT angiography (CCTA) is defined by a thin fibrous cap, a large, inflamed, lipid-rich necrotic core, and microcalcification. Previously, Dr. Williams and her coinvestigators demonstrated that visual identification of this unstable plaque subtype is of benefit in predicting future risk of MI (J Am Coll Cardiol. 2019 Jan 29;73[3]:291-301).
But visual identification of plaque subtypes is a crude and laborious process. In her current study, she and her coworkers have taken things a giant step further, using commercially available CCTA software to semiautomatically quantify the burden of this highest-risk plaque subtype as well as all the other subtypes.
This post hoc analysis of the previously reported main SCOT-HEART trial (N Engl J Med. 2018 Sep 6;379[10]:924-933) included 1,769 patients with stable chest pain randomized to standard care with or without CCTA guidance and followed for a median of 4.7 years, during which 41 patients had a fatal or nonfatal MI. At enrollment, 37% of participants had normal coronary arteries, 38% had nonobstructive coronary artery disease (CAD), and the remainder had obstructive CAD.
In a multivariate analysis, low-attenuation noncalcified plaque burden was the strongest predictor of future MI, with an adjusted hazard ratio of 1.6 per doubling. This metric was strongly correlated with coronary artery calcium score, underscoring the limited value of doing noncontrast CT in order to determine a coronary artery calcium score when CCTA is performed.
Low-attenuation plaque burden correlated very strongly with angiographic severity of stenosis, and only weakly with cardiovascular risk score, perhaps explaining the poor prognostic performance of cardiovascular risk scores in SCOT-HEART and other studies, according to Dr. Williams.
Patients with a low-attenuation noncalcified plaque burden greater than 4% in their coronary tree were 4.7 times more likely to have a subsequent MI than were those with a lesser burden. The predictive power was even greater in patients with nonobstructive CAD, where a low-attenuation noncalcified plaque burden in excess of 4% conferred a 6.6-fold greater likelihood of fatal or nonfatal MI, she observed.
Two things need to happen before measurement of low-attenuation noncalcified plaque via CCTA to predict MI risk is ready to be adopted in routine clinical practice, according to Dr. Williams. These SCOT-HEART results need to be validated in other cohorts, a process now underway in the SCOT-HEART 2 trial and other studies. Also, improved software incorporating machine learning is needed in order to speed up the semiautomated analysis of plaque subtypes, which now takes 20-30 minutes.
Dr. Williams reported having no financial conflicts regarding her study, funded by the National Health Service.
In conjunction with her virtual presentation at ACC 2020, the SCOT-HEART study results were published online (Circulation. 2020 Mar 16. doi: 10.1161/CIRCULATIONAHA.119.044720. [Epub ahead of print]).
SOURCE: Williams MC et al. ACC 2020, Abstract 909-06.
FROM ACC 2020
U.S. ‘deaths of despair’ from COVID-19 could top 75,000, experts warn
The number of “deaths of despair” could be even higher if the country fails to take bold action to address the mental health toll of unemployment, isolation, and uncertainty, according to the report from the Well Being Trust (WBT) and the Robert Graham Center for Policy Studies in Family Medicine and Primary Care.
“If nothing happens and nothing improves – ie, the worst-case scenario – we could be looking at an additional 150,000 people who died who didn’t have to,” Benjamin Miller, PsyD, WBT chief strategy officer, told Medscape Medical News.
“We can prevent these deaths. We know how and have a bevy of evidence-based solutions. We lack the resources to really stand this up in a way that can most positively impact communities,” Miller added.
Slow recovery, quick recovery scenarios
For the analysis, Miller and colleagues combined information on the number of deaths from suicide, alcohol, and drugs from 2018 as a baseline (n = 181,686). They projected levels of unemployment from 2020 to 2029 and then used economic modeling to estimate the additional annual number of deaths.
Across nine different scenarios, the number of additional deaths of despair range from 27,644 (quick recovery, smallest impact of unemployment on suicide, alcohol-, and drug-related deaths) to 154,037 (slow recovery, greatest impact of unemployment on these deaths), with 75,000 being the most likely.
The report offers several policy solutions to prevent a surge in “avoidable” deaths. They include finding ways to ameliorate the effects of unemployment and provide meaningful work to those who are out of work. Making access to care easier and fully integrating mental health and addiction care into primary and clinical care as well as community settings are also essential.
These solutions should also serve to prevent drug and alcohol misuse and suicide in normal times, the researchers say.
Miller believes it’s time for the federal government to fully support a framework of excellence in mental health and well-being and to invest in mental health now.
“In the short term, we need at least $48 billion to keep the lights on in the current system,” he said.
“This is because 92.6% of mental health organizations have had to reduce their operations in some capacity, 61.8% have had to completely close at least one program, and 31.0% have had to turn away patients. This scenario is not optimal for people who will need a system to help them right now during a crisis,” he added.
In the long term, $150 billion is needed for a “massive structural redesign” of the US mental health system, Miller said.
“This means bringing mental health fully into all facets of our healthcare system, of our community. It will take robust investment in creating new mechanisms for care ― those that are team-based, create a new type of workforce to deliver that care, and one that is seamless across clinical and community settings,” said Miller.
A version of this article first appeared on Medscape.com.
The number of “deaths of despair” could be even higher if the country fails to take bold action to address the mental health toll of unemployment, isolation, and uncertainty, according to the report from the Well Being Trust (WBT) and the Robert Graham Center for Policy Studies in Family Medicine and Primary Care.
“If nothing happens and nothing improves – ie, the worst-case scenario – we could be looking at an additional 150,000 people who died who didn’t have to,” Benjamin Miller, PsyD, WBT chief strategy officer, told Medscape Medical News.
“We can prevent these deaths. We know how and have a bevy of evidence-based solutions. We lack the resources to really stand this up in a way that can most positively impact communities,” Miller added.
Slow recovery, quick recovery scenarios
For the analysis, Miller and colleagues combined information on the number of deaths from suicide, alcohol, and drugs from 2018 as a baseline (n = 181,686). They projected levels of unemployment from 2020 to 2029 and then used economic modeling to estimate the additional annual number of deaths.
Across nine different scenarios, the number of additional deaths of despair range from 27,644 (quick recovery, smallest impact of unemployment on suicide, alcohol-, and drug-related deaths) to 154,037 (slow recovery, greatest impact of unemployment on these deaths), with 75,000 being the most likely.
The report offers several policy solutions to prevent a surge in “avoidable” deaths. They include finding ways to ameliorate the effects of unemployment and provide meaningful work to those who are out of work. Making access to care easier and fully integrating mental health and addiction care into primary and clinical care as well as community settings are also essential.
These solutions should also serve to prevent drug and alcohol misuse and suicide in normal times, the researchers say.
Miller believes it’s time for the federal government to fully support a framework of excellence in mental health and well-being and to invest in mental health now.
“In the short term, we need at least $48 billion to keep the lights on in the current system,” he said.
“This is because 92.6% of mental health organizations have had to reduce their operations in some capacity, 61.8% have had to completely close at least one program, and 31.0% have had to turn away patients. This scenario is not optimal for people who will need a system to help them right now during a crisis,” he added.
In the long term, $150 billion is needed for a “massive structural redesign” of the US mental health system, Miller said.
“This means bringing mental health fully into all facets of our healthcare system, of our community. It will take robust investment in creating new mechanisms for care ― those that are team-based, create a new type of workforce to deliver that care, and one that is seamless across clinical and community settings,” said Miller.
A version of this article first appeared on Medscape.com.
The number of “deaths of despair” could be even higher if the country fails to take bold action to address the mental health toll of unemployment, isolation, and uncertainty, according to the report from the Well Being Trust (WBT) and the Robert Graham Center for Policy Studies in Family Medicine and Primary Care.
“If nothing happens and nothing improves – ie, the worst-case scenario – we could be looking at an additional 150,000 people who died who didn’t have to,” Benjamin Miller, PsyD, WBT chief strategy officer, told Medscape Medical News.
“We can prevent these deaths. We know how and have a bevy of evidence-based solutions. We lack the resources to really stand this up in a way that can most positively impact communities,” Miller added.
Slow recovery, quick recovery scenarios
For the analysis, Miller and colleagues combined information on the number of deaths from suicide, alcohol, and drugs from 2018 as a baseline (n = 181,686). They projected levels of unemployment from 2020 to 2029 and then used economic modeling to estimate the additional annual number of deaths.
Across nine different scenarios, the number of additional deaths of despair range from 27,644 (quick recovery, smallest impact of unemployment on suicide, alcohol-, and drug-related deaths) to 154,037 (slow recovery, greatest impact of unemployment on these deaths), with 75,000 being the most likely.
The report offers several policy solutions to prevent a surge in “avoidable” deaths. They include finding ways to ameliorate the effects of unemployment and provide meaningful work to those who are out of work. Making access to care easier and fully integrating mental health and addiction care into primary and clinical care as well as community settings are also essential.
These solutions should also serve to prevent drug and alcohol misuse and suicide in normal times, the researchers say.
Miller believes it’s time for the federal government to fully support a framework of excellence in mental health and well-being and to invest in mental health now.
“In the short term, we need at least $48 billion to keep the lights on in the current system,” he said.
“This is because 92.6% of mental health organizations have had to reduce their operations in some capacity, 61.8% have had to completely close at least one program, and 31.0% have had to turn away patients. This scenario is not optimal for people who will need a system to help them right now during a crisis,” he added.
In the long term, $150 billion is needed for a “massive structural redesign” of the US mental health system, Miller said.
“This means bringing mental health fully into all facets of our healthcare system, of our community. It will take robust investment in creating new mechanisms for care ― those that are team-based, create a new type of workforce to deliver that care, and one that is seamless across clinical and community settings,” said Miller.
A version of this article first appeared on Medscape.com.
Andexanet alfa reverses factor Xa inhibitors
Background: Factor Xa inhibitors have become increasingly popular in the treatment and prevention of thrombotic events, but the lack of specific reversal agents in the event of life-threatening or uncontrolled bleeding may limit their use. Andexanet alfa is a new Food and Drug Administration–approved reversal agent which rapidly reduces anti–factor Xa activity, thereby reversing the anticoagulation effects of factor Xa inhibitors.
Study design: A prospective, open-label, single-group cohort study.
Setting: An industry-sponsored, multicenter study.
Synopsis: The study evaluated 352 adult patients who had acute major bleeding (such as intracranial hemorrhage [64%] or GI bleeding [26%] within 18 hours after administration of a factor Xa inhibitor, including apixaban, rivaroxaban, or edoxaban). Efficacy was assessed in 254 patients who met criteria for severe bleeding and elevated baseline anti–factor Xa activity. Patients were administered a bolus dose of andexanet alfa followed by a 2-hour infusion. The median anti–factor Xa activity reduced by 92% each among patients receiving apixaban or rivaroxaban. The majority (82%) of evaluable patients achieved excellent or good hemostasis at 12 hours after andexanet alfa administration, which compares favorably with the hemostatic efficacy of 72% observed with prothrombin complex concentrate used to reverse anticoagulation in patients treated with vitamin K antagonists. Of patients in the study, 10% experienced a thrombotic event during the 30-day follow-up period, and 14% died.
Limitations of the study include lack of a control group and absence of a significant relationship between a reduction in anti–factor Xa activity and hemostasis. The sponsor is planning to conduct a randomized trial with FDA guidance in the near future.
Bottom line: Andexanet alfa is an FDA-approved agent and appears effective in achieving hemostasis in patients with a factor Xa inhibitor–associated major acute bleeding.
Citation: Connolly SJ et al. Full study report of andexanet alfa for bleeding associated with factor Xa inhibitors. N Eng J Med. 2019 Feb 7. doi: 10.1056/NEJMoa1814051.
Dr. Vedamurthy is a hospitalist at Massachusetts General Hospital.
Background: Factor Xa inhibitors have become increasingly popular in the treatment and prevention of thrombotic events, but the lack of specific reversal agents in the event of life-threatening or uncontrolled bleeding may limit their use. Andexanet alfa is a new Food and Drug Administration–approved reversal agent which rapidly reduces anti–factor Xa activity, thereby reversing the anticoagulation effects of factor Xa inhibitors.
Study design: A prospective, open-label, single-group cohort study.
Setting: An industry-sponsored, multicenter study.
Synopsis: The study evaluated 352 adult patients who had acute major bleeding (such as intracranial hemorrhage [64%] or GI bleeding [26%] within 18 hours after administration of a factor Xa inhibitor, including apixaban, rivaroxaban, or edoxaban). Efficacy was assessed in 254 patients who met criteria for severe bleeding and elevated baseline anti–factor Xa activity. Patients were administered a bolus dose of andexanet alfa followed by a 2-hour infusion. The median anti–factor Xa activity reduced by 92% each among patients receiving apixaban or rivaroxaban. The majority (82%) of evaluable patients achieved excellent or good hemostasis at 12 hours after andexanet alfa administration, which compares favorably with the hemostatic efficacy of 72% observed with prothrombin complex concentrate used to reverse anticoagulation in patients treated with vitamin K antagonists. Of patients in the study, 10% experienced a thrombotic event during the 30-day follow-up period, and 14% died.
Limitations of the study include lack of a control group and absence of a significant relationship between a reduction in anti–factor Xa activity and hemostasis. The sponsor is planning to conduct a randomized trial with FDA guidance in the near future.
Bottom line: Andexanet alfa is an FDA-approved agent and appears effective in achieving hemostasis in patients with a factor Xa inhibitor–associated major acute bleeding.
Citation: Connolly SJ et al. Full study report of andexanet alfa for bleeding associated with factor Xa inhibitors. N Eng J Med. 2019 Feb 7. doi: 10.1056/NEJMoa1814051.
Dr. Vedamurthy is a hospitalist at Massachusetts General Hospital.
Background: Factor Xa inhibitors have become increasingly popular in the treatment and prevention of thrombotic events, but the lack of specific reversal agents in the event of life-threatening or uncontrolled bleeding may limit their use. Andexanet alfa is a new Food and Drug Administration–approved reversal agent which rapidly reduces anti–factor Xa activity, thereby reversing the anticoagulation effects of factor Xa inhibitors.
Study design: A prospective, open-label, single-group cohort study.
Setting: An industry-sponsored, multicenter study.
Synopsis: The study evaluated 352 adult patients who had acute major bleeding (such as intracranial hemorrhage [64%] or GI bleeding [26%] within 18 hours after administration of a factor Xa inhibitor, including apixaban, rivaroxaban, or edoxaban). Efficacy was assessed in 254 patients who met criteria for severe bleeding and elevated baseline anti–factor Xa activity. Patients were administered a bolus dose of andexanet alfa followed by a 2-hour infusion. The median anti–factor Xa activity reduced by 92% each among patients receiving apixaban or rivaroxaban. The majority (82%) of evaluable patients achieved excellent or good hemostasis at 12 hours after andexanet alfa administration, which compares favorably with the hemostatic efficacy of 72% observed with prothrombin complex concentrate used to reverse anticoagulation in patients treated with vitamin K antagonists. Of patients in the study, 10% experienced a thrombotic event during the 30-day follow-up period, and 14% died.
Limitations of the study include lack of a control group and absence of a significant relationship between a reduction in anti–factor Xa activity and hemostasis. The sponsor is planning to conduct a randomized trial with FDA guidance in the near future.
Bottom line: Andexanet alfa is an FDA-approved agent and appears effective in achieving hemostasis in patients with a factor Xa inhibitor–associated major acute bleeding.
Citation: Connolly SJ et al. Full study report of andexanet alfa for bleeding associated with factor Xa inhibitors. N Eng J Med. 2019 Feb 7. doi: 10.1056/NEJMoa1814051.
Dr. Vedamurthy is a hospitalist at Massachusetts General Hospital.
Novel inflammatory syndrome in children possibly linked to COVID-19
according to reports from National Health Service England, The Lancet, and the New York City health department.
Fifteen children in New York City hospitals have presented with the condition, provisionally called pediatric multisystem inflammatory syndrome, between April 17 and May 1, according to a health alert from New York City health department deputy commissioner Demetre C. Daskalakis, MD, MPH, on May 4. On May 5, the New York state department of health released a health advisory that 64 suspected cases had been reported in children in New York state hospitals, including New York City.
The New York City reports follow a case study published April 7 in Hospital Pediatrics about the presentation. There also was a statement from the U.K.’s Paediatric Intensive Care Society (PICS) on April 27 that noted “blood parameters consistent with severe COVID-19 in children” as well as abdominal pain, gastrointestinal symptoms, and cardiac inflammation.
“Whilst it is too early to say with confidence, features appear to include high CRP [C-reactive protein], high [erythrocyte sedimentation rate] and high ferritin,” the PICS release stated. The cardiac inflammation consists of “myocarditis with raised troponin and [prohormone brain natriuretic peptide],” according to the PICS statement. “Some have an appearance of their coronary arteries in keeping with Kawasaki disease.”
The initial 15 New York City patients reportedly all had “subjective or measured fever, and more than half reported rash, abdominal pain, vomiting, or diarrhea,” but fewer than half had respiratory symptoms.
The case study described a 6-month-old infant who was admitted and diagnosed with classic Kawasaki disease, who also tested positive for COVID-19 with fever and mild respiratory symptoms, reported Veena G. Jones, MD, a pediatric hospitalist in Palo Alto, Calif., and associates.
While many of the U.K. children presenting with the symptoms had a positive polymerase chain reaction tests for infection from SARS-CoV-2, some also had a negative test. Polymerase chain reaction testing in New York City was positive for 4 children and negative for 11 children, but 6 of the those who tested negative had positive serology tests, potentially pointing to postinfection sequelae.
At press time, more cases were reported from the United Kingdom in The Lancet. In London, eight children with hyperinflammatory shock, showing features similar to atypical Kawasaki disease, Kawasaki disease shock syndrome, or toxic shock syndrome, presented within 10 days to Evelina London Children’s Hospital Paediatric ICU, Shelley Riphagen, MBChB, and colleagues revealed.
Clinically, their presentations were similar, with persistent fever, rash, conjunctivitis, peripheral edema, extremity pain, and gastrointestinal symptoms. They all developed warm vasoplegic shock that did not respond to volume resuscitation; noradrenaline and milrinone were administered for hemodynamic support. Seven of the children needed mechanical ventilation for cardiovascular stabilization, although most of them had no significant respiratory involvement.
Of note was development of small pleural, pericardial, and ascitic effusion – “suggestive of a diffuse inflammatory process,” Dr. Riphagen and associates wrote. None of the children initially was positive for SARS-CoV-2; laboratory evidence of infection or inflammation included “elevated concentrations of CRP, procalcitonin, ferritin, triglycerides or d-dimers.”
“A common echocardiographic finding was echobright coronary vessels,” they wrote. “One child developed arrhythmia with refractory shock, requiring extracorporeal life support, and died from a large cerebrovascular infarct.”
As the article went to press, the doctors in that same ICU had seen more than 20 children with similar clinical presentations, Dr. Riphagen and associates reported, and the first 10 tested positive for SARS-CoV-2 antibody, including the 8 described above.
“Most of the children appear to have antibodies to the novel coronavirus, even when they do not have virus detectable in their nose,” said Audrey John, MD, PhD, chief of the division of pediatric infectious diseases at Children’s Hospital of Philadelphia, where clinicians have seen several cases similar to those described by NHS England and the New York City health department. “This suggests that these symptoms are ‘postinfectious,’ likely due to an abnormal immune response that happens after viral infection.”
She noted at the time of her interview, however, that fewer than 100 U.S. pediatric cases appear to have been reported.
“While our understanding is evolving, given the scope of the COVID-19 pandemic, this suggests that this kind of severe disease in children is very rare indeed,” Dr. John said. “Because this syndrome is so newly described, we have to continue to be cautious in attributing this syndrome to COVID-19, as there are many other diseases that look quite similar.”
She advised clinicians to be “wary of attributing fever/rash/shock to this syndrome, as the differential is broad, and we do not want to fail to recognize and treat true toxic shock or tick-borne disease.”
Dawn Nolt, MD, MPH, an associate professor of pediatrics in infectious diseases at Oregon Health & Science University’s Doernbecher Children’s Hospital, Portland, also underscored the need to avoid drawing conclusions too quickly.
“At this time, there is no causality established between SARS-COV-2 and these inflammatory syndromes other than a temporal association,” said Dr. Nolt, whose hospital has not yet seen any of these cases. “If there is a link, then the symptoms may be from a ‘direct hit’ of the virus on tissues, or from an overly exuberant immune response.”
None of the initial 15 New York City children died, although 5 needed mechanical ventilation and over half needed blood pressure support. The one child in London died from a large cerebrovascular infarct.
If the cases are connected to COVID-19, one explanation for the presentation may be related to the leading hypothesis “that SARS-CoV-2 may stimulate the immune system in such a way to promote vasculitis,” Dr. Nolt said in an interview.
“It is unusual that this particular constellation was not reported from the known pediatric cases out of China, where the COVID-19 pandemic originated,” Dr. Nolt said. “If there is a link between SARS-CoV-2 and these inflammatory syndromes, this may have resulted from genetic/host differences, changes in the SARS-CoV-2 virus, or other factors yet to be determined.”
The New York City bulletin recommended that clinicians immediately refer children presenting with the described symptoms to a specialist in pediatric infectious disease, rheumatology, or critical care.
“Early diagnosis and treatment of patients meeting full or partial criteria for Kawasaki disease is critical to preventing end-organ damage and other long-term complications,” the bulletin stated. It recommended aspirin and intravenous immunoglobulin for those who met Kawasaki criteria.
Dr. John said that children with the presentation appear to be responding well to intravenous immunoglobulin and/or steroids. She further emphasized that virtually all pediatric patients recover from COVID-19.
“Physicians should advise families to bring their children and teens back in for evaluation if they develop new fever, rash, or abdominal pain and diarrhea,” Dr. John said. “Families should not be afraid to seek care when their kids are sick. Our pediatric hospitals and EDs are open for business and working hard to protect staff and patients.”
A Kawasaki syndrome diagnosis requires at least 5 days of a fever at 101-104° F or higher along with four of the following five symptoms: rash over the torso; redness and swelling on palms and soles of the feet with later skin peeling; bloodshot, light-sensitive eyes; swollen lymph glands in the neck; and irritation and inflammation of the mouth, lips and throat, sometimes with “strawberry” tongue, according to the American Heart Association.
A press release from the AHA noted that Kawasaki disease is the most common cause of acquired heart disease in developed countries, but the condition remains rare.
Kawasaki disease’s etiology is unknown, but “some evidence suggests an infectious trigger, with winter-spring seasonality of the disease,” wrote the case study authors, noting that past research has linked Kawasaki disease with previous or concurrent infections of rhinovirus/enterovirus, parainfluenza, respiratory syncytial virus, influenza, adenovirus, and the four common human coronavirus strains.
“We have to remember that our experience with this pandemic is less than 12 months,” Dr. Nolt said. “We are still accumulating information, and any additional manifestations, particularly severe ones, adds to our ability to more quickly detect and treat children.”
Dr. Nolt and Dr. John had no disclosures.
SOURCES: Jones VG et al. Hosp Pediatr. 2020 Apr 7. doi: 10.1542/hpeds.2020-0123; Riphagen S et al. Lancet. 2020 May 6. doi: 10.1016/S0140-6736(20)31094-1.
according to reports from National Health Service England, The Lancet, and the New York City health department.
Fifteen children in New York City hospitals have presented with the condition, provisionally called pediatric multisystem inflammatory syndrome, between April 17 and May 1, according to a health alert from New York City health department deputy commissioner Demetre C. Daskalakis, MD, MPH, on May 4. On May 5, the New York state department of health released a health advisory that 64 suspected cases had been reported in children in New York state hospitals, including New York City.
The New York City reports follow a case study published April 7 in Hospital Pediatrics about the presentation. There also was a statement from the U.K.’s Paediatric Intensive Care Society (PICS) on April 27 that noted “blood parameters consistent with severe COVID-19 in children” as well as abdominal pain, gastrointestinal symptoms, and cardiac inflammation.
“Whilst it is too early to say with confidence, features appear to include high CRP [C-reactive protein], high [erythrocyte sedimentation rate] and high ferritin,” the PICS release stated. The cardiac inflammation consists of “myocarditis with raised troponin and [prohormone brain natriuretic peptide],” according to the PICS statement. “Some have an appearance of their coronary arteries in keeping with Kawasaki disease.”
The initial 15 New York City patients reportedly all had “subjective or measured fever, and more than half reported rash, abdominal pain, vomiting, or diarrhea,” but fewer than half had respiratory symptoms.
The case study described a 6-month-old infant who was admitted and diagnosed with classic Kawasaki disease, who also tested positive for COVID-19 with fever and mild respiratory symptoms, reported Veena G. Jones, MD, a pediatric hospitalist in Palo Alto, Calif., and associates.
While many of the U.K. children presenting with the symptoms had a positive polymerase chain reaction tests for infection from SARS-CoV-2, some also had a negative test. Polymerase chain reaction testing in New York City was positive for 4 children and negative for 11 children, but 6 of the those who tested negative had positive serology tests, potentially pointing to postinfection sequelae.
At press time, more cases were reported from the United Kingdom in The Lancet. In London, eight children with hyperinflammatory shock, showing features similar to atypical Kawasaki disease, Kawasaki disease shock syndrome, or toxic shock syndrome, presented within 10 days to Evelina London Children’s Hospital Paediatric ICU, Shelley Riphagen, MBChB, and colleagues revealed.
Clinically, their presentations were similar, with persistent fever, rash, conjunctivitis, peripheral edema, extremity pain, and gastrointestinal symptoms. They all developed warm vasoplegic shock that did not respond to volume resuscitation; noradrenaline and milrinone were administered for hemodynamic support. Seven of the children needed mechanical ventilation for cardiovascular stabilization, although most of them had no significant respiratory involvement.
Of note was development of small pleural, pericardial, and ascitic effusion – “suggestive of a diffuse inflammatory process,” Dr. Riphagen and associates wrote. None of the children initially was positive for SARS-CoV-2; laboratory evidence of infection or inflammation included “elevated concentrations of CRP, procalcitonin, ferritin, triglycerides or d-dimers.”
“A common echocardiographic finding was echobright coronary vessels,” they wrote. “One child developed arrhythmia with refractory shock, requiring extracorporeal life support, and died from a large cerebrovascular infarct.”
As the article went to press, the doctors in that same ICU had seen more than 20 children with similar clinical presentations, Dr. Riphagen and associates reported, and the first 10 tested positive for SARS-CoV-2 antibody, including the 8 described above.
“Most of the children appear to have antibodies to the novel coronavirus, even when they do not have virus detectable in their nose,” said Audrey John, MD, PhD, chief of the division of pediatric infectious diseases at Children’s Hospital of Philadelphia, where clinicians have seen several cases similar to those described by NHS England and the New York City health department. “This suggests that these symptoms are ‘postinfectious,’ likely due to an abnormal immune response that happens after viral infection.”
She noted at the time of her interview, however, that fewer than 100 U.S. pediatric cases appear to have been reported.
“While our understanding is evolving, given the scope of the COVID-19 pandemic, this suggests that this kind of severe disease in children is very rare indeed,” Dr. John said. “Because this syndrome is so newly described, we have to continue to be cautious in attributing this syndrome to COVID-19, as there are many other diseases that look quite similar.”
She advised clinicians to be “wary of attributing fever/rash/shock to this syndrome, as the differential is broad, and we do not want to fail to recognize and treat true toxic shock or tick-borne disease.”
Dawn Nolt, MD, MPH, an associate professor of pediatrics in infectious diseases at Oregon Health & Science University’s Doernbecher Children’s Hospital, Portland, also underscored the need to avoid drawing conclusions too quickly.
“At this time, there is no causality established between SARS-COV-2 and these inflammatory syndromes other than a temporal association,” said Dr. Nolt, whose hospital has not yet seen any of these cases. “If there is a link, then the symptoms may be from a ‘direct hit’ of the virus on tissues, or from an overly exuberant immune response.”
None of the initial 15 New York City children died, although 5 needed mechanical ventilation and over half needed blood pressure support. The one child in London died from a large cerebrovascular infarct.
If the cases are connected to COVID-19, one explanation for the presentation may be related to the leading hypothesis “that SARS-CoV-2 may stimulate the immune system in such a way to promote vasculitis,” Dr. Nolt said in an interview.
“It is unusual that this particular constellation was not reported from the known pediatric cases out of China, where the COVID-19 pandemic originated,” Dr. Nolt said. “If there is a link between SARS-CoV-2 and these inflammatory syndromes, this may have resulted from genetic/host differences, changes in the SARS-CoV-2 virus, or other factors yet to be determined.”
The New York City bulletin recommended that clinicians immediately refer children presenting with the described symptoms to a specialist in pediatric infectious disease, rheumatology, or critical care.
“Early diagnosis and treatment of patients meeting full or partial criteria for Kawasaki disease is critical to preventing end-organ damage and other long-term complications,” the bulletin stated. It recommended aspirin and intravenous immunoglobulin for those who met Kawasaki criteria.
Dr. John said that children with the presentation appear to be responding well to intravenous immunoglobulin and/or steroids. She further emphasized that virtually all pediatric patients recover from COVID-19.
“Physicians should advise families to bring their children and teens back in for evaluation if they develop new fever, rash, or abdominal pain and diarrhea,” Dr. John said. “Families should not be afraid to seek care when their kids are sick. Our pediatric hospitals and EDs are open for business and working hard to protect staff and patients.”
A Kawasaki syndrome diagnosis requires at least 5 days of a fever at 101-104° F or higher along with four of the following five symptoms: rash over the torso; redness and swelling on palms and soles of the feet with later skin peeling; bloodshot, light-sensitive eyes; swollen lymph glands in the neck; and irritation and inflammation of the mouth, lips and throat, sometimes with “strawberry” tongue, according to the American Heart Association.
A press release from the AHA noted that Kawasaki disease is the most common cause of acquired heart disease in developed countries, but the condition remains rare.
Kawasaki disease’s etiology is unknown, but “some evidence suggests an infectious trigger, with winter-spring seasonality of the disease,” wrote the case study authors, noting that past research has linked Kawasaki disease with previous or concurrent infections of rhinovirus/enterovirus, parainfluenza, respiratory syncytial virus, influenza, adenovirus, and the four common human coronavirus strains.
“We have to remember that our experience with this pandemic is less than 12 months,” Dr. Nolt said. “We are still accumulating information, and any additional manifestations, particularly severe ones, adds to our ability to more quickly detect and treat children.”
Dr. Nolt and Dr. John had no disclosures.
SOURCES: Jones VG et al. Hosp Pediatr. 2020 Apr 7. doi: 10.1542/hpeds.2020-0123; Riphagen S et al. Lancet. 2020 May 6. doi: 10.1016/S0140-6736(20)31094-1.
according to reports from National Health Service England, The Lancet, and the New York City health department.
Fifteen children in New York City hospitals have presented with the condition, provisionally called pediatric multisystem inflammatory syndrome, between April 17 and May 1, according to a health alert from New York City health department deputy commissioner Demetre C. Daskalakis, MD, MPH, on May 4. On May 5, the New York state department of health released a health advisory that 64 suspected cases had been reported in children in New York state hospitals, including New York City.
The New York City reports follow a case study published April 7 in Hospital Pediatrics about the presentation. There also was a statement from the U.K.’s Paediatric Intensive Care Society (PICS) on April 27 that noted “blood parameters consistent with severe COVID-19 in children” as well as abdominal pain, gastrointestinal symptoms, and cardiac inflammation.
“Whilst it is too early to say with confidence, features appear to include high CRP [C-reactive protein], high [erythrocyte sedimentation rate] and high ferritin,” the PICS release stated. The cardiac inflammation consists of “myocarditis with raised troponin and [prohormone brain natriuretic peptide],” according to the PICS statement. “Some have an appearance of their coronary arteries in keeping with Kawasaki disease.”
The initial 15 New York City patients reportedly all had “subjective or measured fever, and more than half reported rash, abdominal pain, vomiting, or diarrhea,” but fewer than half had respiratory symptoms.
The case study described a 6-month-old infant who was admitted and diagnosed with classic Kawasaki disease, who also tested positive for COVID-19 with fever and mild respiratory symptoms, reported Veena G. Jones, MD, a pediatric hospitalist in Palo Alto, Calif., and associates.
While many of the U.K. children presenting with the symptoms had a positive polymerase chain reaction tests for infection from SARS-CoV-2, some also had a negative test. Polymerase chain reaction testing in New York City was positive for 4 children and negative for 11 children, but 6 of the those who tested negative had positive serology tests, potentially pointing to postinfection sequelae.
At press time, more cases were reported from the United Kingdom in The Lancet. In London, eight children with hyperinflammatory shock, showing features similar to atypical Kawasaki disease, Kawasaki disease shock syndrome, or toxic shock syndrome, presented within 10 days to Evelina London Children’s Hospital Paediatric ICU, Shelley Riphagen, MBChB, and colleagues revealed.
Clinically, their presentations were similar, with persistent fever, rash, conjunctivitis, peripheral edema, extremity pain, and gastrointestinal symptoms. They all developed warm vasoplegic shock that did not respond to volume resuscitation; noradrenaline and milrinone were administered for hemodynamic support. Seven of the children needed mechanical ventilation for cardiovascular stabilization, although most of them had no significant respiratory involvement.
Of note was development of small pleural, pericardial, and ascitic effusion – “suggestive of a diffuse inflammatory process,” Dr. Riphagen and associates wrote. None of the children initially was positive for SARS-CoV-2; laboratory evidence of infection or inflammation included “elevated concentrations of CRP, procalcitonin, ferritin, triglycerides or d-dimers.”
“A common echocardiographic finding was echobright coronary vessels,” they wrote. “One child developed arrhythmia with refractory shock, requiring extracorporeal life support, and died from a large cerebrovascular infarct.”
As the article went to press, the doctors in that same ICU had seen more than 20 children with similar clinical presentations, Dr. Riphagen and associates reported, and the first 10 tested positive for SARS-CoV-2 antibody, including the 8 described above.
“Most of the children appear to have antibodies to the novel coronavirus, even when they do not have virus detectable in their nose,” said Audrey John, MD, PhD, chief of the division of pediatric infectious diseases at Children’s Hospital of Philadelphia, where clinicians have seen several cases similar to those described by NHS England and the New York City health department. “This suggests that these symptoms are ‘postinfectious,’ likely due to an abnormal immune response that happens after viral infection.”
She noted at the time of her interview, however, that fewer than 100 U.S. pediatric cases appear to have been reported.
“While our understanding is evolving, given the scope of the COVID-19 pandemic, this suggests that this kind of severe disease in children is very rare indeed,” Dr. John said. “Because this syndrome is so newly described, we have to continue to be cautious in attributing this syndrome to COVID-19, as there are many other diseases that look quite similar.”
She advised clinicians to be “wary of attributing fever/rash/shock to this syndrome, as the differential is broad, and we do not want to fail to recognize and treat true toxic shock or tick-borne disease.”
Dawn Nolt, MD, MPH, an associate professor of pediatrics in infectious diseases at Oregon Health & Science University’s Doernbecher Children’s Hospital, Portland, also underscored the need to avoid drawing conclusions too quickly.
“At this time, there is no causality established between SARS-COV-2 and these inflammatory syndromes other than a temporal association,” said Dr. Nolt, whose hospital has not yet seen any of these cases. “If there is a link, then the symptoms may be from a ‘direct hit’ of the virus on tissues, or from an overly exuberant immune response.”
None of the initial 15 New York City children died, although 5 needed mechanical ventilation and over half needed blood pressure support. The one child in London died from a large cerebrovascular infarct.
If the cases are connected to COVID-19, one explanation for the presentation may be related to the leading hypothesis “that SARS-CoV-2 may stimulate the immune system in such a way to promote vasculitis,” Dr. Nolt said in an interview.
“It is unusual that this particular constellation was not reported from the known pediatric cases out of China, where the COVID-19 pandemic originated,” Dr. Nolt said. “If there is a link between SARS-CoV-2 and these inflammatory syndromes, this may have resulted from genetic/host differences, changes in the SARS-CoV-2 virus, or other factors yet to be determined.”
The New York City bulletin recommended that clinicians immediately refer children presenting with the described symptoms to a specialist in pediatric infectious disease, rheumatology, or critical care.
“Early diagnosis and treatment of patients meeting full or partial criteria for Kawasaki disease is critical to preventing end-organ damage and other long-term complications,” the bulletin stated. It recommended aspirin and intravenous immunoglobulin for those who met Kawasaki criteria.
Dr. John said that children with the presentation appear to be responding well to intravenous immunoglobulin and/or steroids. She further emphasized that virtually all pediatric patients recover from COVID-19.
“Physicians should advise families to bring their children and teens back in for evaluation if they develop new fever, rash, or abdominal pain and diarrhea,” Dr. John said. “Families should not be afraid to seek care when their kids are sick. Our pediatric hospitals and EDs are open for business and working hard to protect staff and patients.”
A Kawasaki syndrome diagnosis requires at least 5 days of a fever at 101-104° F or higher along with four of the following five symptoms: rash over the torso; redness and swelling on palms and soles of the feet with later skin peeling; bloodshot, light-sensitive eyes; swollen lymph glands in the neck; and irritation and inflammation of the mouth, lips and throat, sometimes with “strawberry” tongue, according to the American Heart Association.
A press release from the AHA noted that Kawasaki disease is the most common cause of acquired heart disease in developed countries, but the condition remains rare.
Kawasaki disease’s etiology is unknown, but “some evidence suggests an infectious trigger, with winter-spring seasonality of the disease,” wrote the case study authors, noting that past research has linked Kawasaki disease with previous or concurrent infections of rhinovirus/enterovirus, parainfluenza, respiratory syncytial virus, influenza, adenovirus, and the four common human coronavirus strains.
“We have to remember that our experience with this pandemic is less than 12 months,” Dr. Nolt said. “We are still accumulating information, and any additional manifestations, particularly severe ones, adds to our ability to more quickly detect and treat children.”
Dr. Nolt and Dr. John had no disclosures.
SOURCES: Jones VG et al. Hosp Pediatr. 2020 Apr 7. doi: 10.1542/hpeds.2020-0123; Riphagen S et al. Lancet. 2020 May 6. doi: 10.1016/S0140-6736(20)31094-1.
Androgens may explain male vulnerability to COVID-19
As the COVID-19 pandemic has swept across the world, a striking difference has been seen between the sexes. But why are men so much more susceptible to severe outcomes from COVID-19 than women?
Suspicions naturally turn to the sex hormones, and there have been suggestions that estrogen may be protective against COVID-19 in females and/or that androgens worsen COVID-19 outcomes in males.
New data supporting the androgen theory come from a study in Italy.
These researchers found that patients with prostate cancer being treated with androgen deprivation therapy (ADT) were less likely to become infected with COVID-19 and die from the disease than other groups, including other patients with cancer.
The findings suggest that androgens somehow make the virus more virulent and that this exacerbates the severity of disease in men, they say. They also speculate that ADT may be protective against COVID-19.
The study was published online May 7 in Annals of Oncology.
The team analyzed data from 68 hospitals in the Veneto region, one of the areas in Italy most severely affected by the COVID-19 pandemic.
They found data on 9280 patients with laboratory-confirmed SARS-CoV-2 infection — of whom 4532 were males.
Women in the region were actually slightly more likely to be infected with COVID-19 than men, 56% vs 44%, the researchers point out.
However, men were more prone to develop more severe forms of the disease: 60% of men vs 40% of women required hospitalization, rising to 78% of men vs 22% of women who required intensive care. Also, more men died than women (62% vs 38%).
The team then turned their focus onto patients with cancer.
Of the entire male population of Veneto, those with cancer had an almost twofold higher risk of becoming infected with COVID-19 than men without cancer (P < .0001).
However, when the team looked specifically at men with prostate cancer in the region, they found “strikingly, only 4 out of 5273 patients receiving ADT developed SARS-CoV-2 infection and none of these patients died.”
This compared to 37,161 men with prostate cancer who were not receiving ADT, among whom 114 men developed COVID-19 and 18 died.
Among another 79,661 patients in the Veneto region with cancer other than prostate cancer, 312 developed COVID-19 and 57 died.
“This is the first paper to suggest a link between ADT and COVID-19,” commented lead author Andrea Alimonti, MD, PhD, Università della Svizzera Italiana in Lugano, Switzerland.
“Patients with prostate cancer receiving ADT had a significant fourfold reduced risk of COVID-19 infections compared to patients who did not receive ADT. An even greater difference (fivefold reduction in risk) was found when we compared prostate cancer patients receiving ADT to patients with any other type of cancer,” he said.
The finding raises “the hypothesis that androgen levels can facilitate coronavirus infections and increase the severity of symptoms, as has been seen in male patients,” he said.
“These data are very interesting and raise a fascinating hypothesis,” said Richard Martin, PhD, professor of clinical epidemiology at the University of Bristol, UK, commenting about the study. “But they do need independent validation in other large population-wide datasets...with appropriate statistical analysis including adjustment for important risk factors for SARS-CoV-2.”
He noted that the Italian study results were not adjusted for potential confounders, for example, age, body mass index, and cardiometabolic comorbidities, that are strong risk factors for SARS-CoV-2. In addition, men taking ADT may have been more likely to self-isolate and so be at reduced risk of getting the infection, he suggested.
How Do Androgens Interact With the Virus?
Alimonti and colleagues offer a mechanistic explanation of how androgens interact with the virus.
Coronavirus gains entry into the human cell by binding its viral spike (S) proteins to ACE2 and on S protein priming by TMPRSS2. TMPRSS2 is a member of a family of proteins called type II transmembrane serine proteases, which are involved in a number of processes including cancer and viral infections, they explain.
“Intriguingly, TMPRSS2 is an androgen-regulated gene that is upregulated in prostate cancer where it supports tumor progression,” they point out.
There is also evidence that the same androgen receptor regulates TMPRSS2 expression in nonprostatic tissues, including the lungs.
“[This] may explain the increased susceptibility of men to develop SARS-CoV-2 severe infections when compared to women,” the authors speculate.
Because ADT is known to decrease TMPRSS2 levels, they suggest that androgen receptor antagonists “could be used to block or decrease the severity of SARS-CoV-2 infection in male patients.”
They go even further and suggest that men without prostate cancer at high risk for COVID-19 could take ADT to prevent infection.
For men who do become infected with COVID-19, ADT might also help reduce symptom severity, they add.
Given that the effects of androgen receptor antagonists are reversible, “they could be used transiently (eg, 1 month) in patients affected by SARS-CoV-2, thereby reducing the risk of side effects due to long-term administration,” the authors suggest.
Another Theory: Is Estrogen Protective?
Another theory to explain the male/female difference for severe COVID-19 is that the female hormone estrogen may be protective.
“People have to stop putting estrogen in that ‘female hormone box’ because it’s a molecule that we all use as humans, it’s just not women,” Sharon Nachman, MD, told Medscape Medical News.
“Looking at estrogen as having potentially important immune effects is part of thinking outside the box,” she said.
Nachman is associate dean for research at the Renaissance School of Medicine, Stony Brook University in New York, and is working together with Antonios Gasparis, MD, professor of surgery at the same center.
They are exploring the use of a transdermal estrogen patch in patients with COVID-19 in a randomized trial with a placebo-controlled arm. They are recruiting patients who present to their emergency department with signs and symptoms of COVID-19, and enroll them into the trial if they are interested.
“We are testing everyone as well, but we are starting patients on the medication at the time of entry as opposed to waiting until we have a test result back,” Nachman explained.
The primary objective of the study is to evaluate whether the transdermal patch, applied to the skin for 7 days, might reduce the need for intubation in men and women infected with COVID-19 versus standard of care.
The product is the same single-use transdermal estradiol patch (Climara, 25 cm2, Bayer) prescribed for postmenopausal women and will be used at the same dose, which is known to be safe.
After the patch is removed, patients will be carefully tracked for symptoms over the next 45 days to see if the patch reduced symptom severity, and if so, in which patients.
Nachman would have preferred to enroll patients before they had overt symptoms, but this simply isn’t possible in a medical center where symptomatic patients present, she told Medscape Medical News.
However, she does know that even at their own medical center, the odds are stacked against male COVID-19 patients — and something is needed to mitigate its severity in this patient group.
As they were developing the protocol for the current study, the team decided to see who was in their ICU during a single study day.
The answer: mostly males. Intubation and death rates in men in their ICU for that single day was approximately 80% compared with only 20% among women.
“We have a new horrific pathogen that is pandemic and we’re all probably going to get it, it’s just a question of when and how sick we’ll be from it,” Nachman said.
Alimonti and coauthors have reported no relevant financial relationships, as did Goulder and Nachman.
This article first appeared on Medscape.com.
As the COVID-19 pandemic has swept across the world, a striking difference has been seen between the sexes. But why are men so much more susceptible to severe outcomes from COVID-19 than women?
Suspicions naturally turn to the sex hormones, and there have been suggestions that estrogen may be protective against COVID-19 in females and/or that androgens worsen COVID-19 outcomes in males.
New data supporting the androgen theory come from a study in Italy.
These researchers found that patients with prostate cancer being treated with androgen deprivation therapy (ADT) were less likely to become infected with COVID-19 and die from the disease than other groups, including other patients with cancer.
The findings suggest that androgens somehow make the virus more virulent and that this exacerbates the severity of disease in men, they say. They also speculate that ADT may be protective against COVID-19.
The study was published online May 7 in Annals of Oncology.
The team analyzed data from 68 hospitals in the Veneto region, one of the areas in Italy most severely affected by the COVID-19 pandemic.
They found data on 9280 patients with laboratory-confirmed SARS-CoV-2 infection — of whom 4532 were males.
Women in the region were actually slightly more likely to be infected with COVID-19 than men, 56% vs 44%, the researchers point out.
However, men were more prone to develop more severe forms of the disease: 60% of men vs 40% of women required hospitalization, rising to 78% of men vs 22% of women who required intensive care. Also, more men died than women (62% vs 38%).
The team then turned their focus onto patients with cancer.
Of the entire male population of Veneto, those with cancer had an almost twofold higher risk of becoming infected with COVID-19 than men without cancer (P < .0001).
However, when the team looked specifically at men with prostate cancer in the region, they found “strikingly, only 4 out of 5273 patients receiving ADT developed SARS-CoV-2 infection and none of these patients died.”
This compared to 37,161 men with prostate cancer who were not receiving ADT, among whom 114 men developed COVID-19 and 18 died.
Among another 79,661 patients in the Veneto region with cancer other than prostate cancer, 312 developed COVID-19 and 57 died.
“This is the first paper to suggest a link between ADT and COVID-19,” commented lead author Andrea Alimonti, MD, PhD, Università della Svizzera Italiana in Lugano, Switzerland.
“Patients with prostate cancer receiving ADT had a significant fourfold reduced risk of COVID-19 infections compared to patients who did not receive ADT. An even greater difference (fivefold reduction in risk) was found when we compared prostate cancer patients receiving ADT to patients with any other type of cancer,” he said.
The finding raises “the hypothesis that androgen levels can facilitate coronavirus infections and increase the severity of symptoms, as has been seen in male patients,” he said.
“These data are very interesting and raise a fascinating hypothesis,” said Richard Martin, PhD, professor of clinical epidemiology at the University of Bristol, UK, commenting about the study. “But they do need independent validation in other large population-wide datasets...with appropriate statistical analysis including adjustment for important risk factors for SARS-CoV-2.”
He noted that the Italian study results were not adjusted for potential confounders, for example, age, body mass index, and cardiometabolic comorbidities, that are strong risk factors for SARS-CoV-2. In addition, men taking ADT may have been more likely to self-isolate and so be at reduced risk of getting the infection, he suggested.
How Do Androgens Interact With the Virus?
Alimonti and colleagues offer a mechanistic explanation of how androgens interact with the virus.
Coronavirus gains entry into the human cell by binding its viral spike (S) proteins to ACE2 and on S protein priming by TMPRSS2. TMPRSS2 is a member of a family of proteins called type II transmembrane serine proteases, which are involved in a number of processes including cancer and viral infections, they explain.
“Intriguingly, TMPRSS2 is an androgen-regulated gene that is upregulated in prostate cancer where it supports tumor progression,” they point out.
There is also evidence that the same androgen receptor regulates TMPRSS2 expression in nonprostatic tissues, including the lungs.
“[This] may explain the increased susceptibility of men to develop SARS-CoV-2 severe infections when compared to women,” the authors speculate.
Because ADT is known to decrease TMPRSS2 levels, they suggest that androgen receptor antagonists “could be used to block or decrease the severity of SARS-CoV-2 infection in male patients.”
They go even further and suggest that men without prostate cancer at high risk for COVID-19 could take ADT to prevent infection.
For men who do become infected with COVID-19, ADT might also help reduce symptom severity, they add.
Given that the effects of androgen receptor antagonists are reversible, “they could be used transiently (eg, 1 month) in patients affected by SARS-CoV-2, thereby reducing the risk of side effects due to long-term administration,” the authors suggest.
Another Theory: Is Estrogen Protective?
Another theory to explain the male/female difference for severe COVID-19 is that the female hormone estrogen may be protective.
“People have to stop putting estrogen in that ‘female hormone box’ because it’s a molecule that we all use as humans, it’s just not women,” Sharon Nachman, MD, told Medscape Medical News.
“Looking at estrogen as having potentially important immune effects is part of thinking outside the box,” she said.
Nachman is associate dean for research at the Renaissance School of Medicine, Stony Brook University in New York, and is working together with Antonios Gasparis, MD, professor of surgery at the same center.
They are exploring the use of a transdermal estrogen patch in patients with COVID-19 in a randomized trial with a placebo-controlled arm. They are recruiting patients who present to their emergency department with signs and symptoms of COVID-19, and enroll them into the trial if they are interested.
“We are testing everyone as well, but we are starting patients on the medication at the time of entry as opposed to waiting until we have a test result back,” Nachman explained.
The primary objective of the study is to evaluate whether the transdermal patch, applied to the skin for 7 days, might reduce the need for intubation in men and women infected with COVID-19 versus standard of care.
The product is the same single-use transdermal estradiol patch (Climara, 25 cm2, Bayer) prescribed for postmenopausal women and will be used at the same dose, which is known to be safe.
After the patch is removed, patients will be carefully tracked for symptoms over the next 45 days to see if the patch reduced symptom severity, and if so, in which patients.
Nachman would have preferred to enroll patients before they had overt symptoms, but this simply isn’t possible in a medical center where symptomatic patients present, she told Medscape Medical News.
However, she does know that even at their own medical center, the odds are stacked against male COVID-19 patients — and something is needed to mitigate its severity in this patient group.
As they were developing the protocol for the current study, the team decided to see who was in their ICU during a single study day.
The answer: mostly males. Intubation and death rates in men in their ICU for that single day was approximately 80% compared with only 20% among women.
“We have a new horrific pathogen that is pandemic and we’re all probably going to get it, it’s just a question of when and how sick we’ll be from it,” Nachman said.
Alimonti and coauthors have reported no relevant financial relationships, as did Goulder and Nachman.
This article first appeared on Medscape.com.
As the COVID-19 pandemic has swept across the world, a striking difference has been seen between the sexes. But why are men so much more susceptible to severe outcomes from COVID-19 than women?
Suspicions naturally turn to the sex hormones, and there have been suggestions that estrogen may be protective against COVID-19 in females and/or that androgens worsen COVID-19 outcomes in males.
New data supporting the androgen theory come from a study in Italy.
These researchers found that patients with prostate cancer being treated with androgen deprivation therapy (ADT) were less likely to become infected with COVID-19 and die from the disease than other groups, including other patients with cancer.
The findings suggest that androgens somehow make the virus more virulent and that this exacerbates the severity of disease in men, they say. They also speculate that ADT may be protective against COVID-19.
The study was published online May 7 in Annals of Oncology.
The team analyzed data from 68 hospitals in the Veneto region, one of the areas in Italy most severely affected by the COVID-19 pandemic.
They found data on 9280 patients with laboratory-confirmed SARS-CoV-2 infection — of whom 4532 were males.
Women in the region were actually slightly more likely to be infected with COVID-19 than men, 56% vs 44%, the researchers point out.
However, men were more prone to develop more severe forms of the disease: 60% of men vs 40% of women required hospitalization, rising to 78% of men vs 22% of women who required intensive care. Also, more men died than women (62% vs 38%).
The team then turned their focus onto patients with cancer.
Of the entire male population of Veneto, those with cancer had an almost twofold higher risk of becoming infected with COVID-19 than men without cancer (P < .0001).
However, when the team looked specifically at men with prostate cancer in the region, they found “strikingly, only 4 out of 5273 patients receiving ADT developed SARS-CoV-2 infection and none of these patients died.”
This compared to 37,161 men with prostate cancer who were not receiving ADT, among whom 114 men developed COVID-19 and 18 died.
Among another 79,661 patients in the Veneto region with cancer other than prostate cancer, 312 developed COVID-19 and 57 died.
“This is the first paper to suggest a link between ADT and COVID-19,” commented lead author Andrea Alimonti, MD, PhD, Università della Svizzera Italiana in Lugano, Switzerland.
“Patients with prostate cancer receiving ADT had a significant fourfold reduced risk of COVID-19 infections compared to patients who did not receive ADT. An even greater difference (fivefold reduction in risk) was found when we compared prostate cancer patients receiving ADT to patients with any other type of cancer,” he said.
The finding raises “the hypothesis that androgen levels can facilitate coronavirus infections and increase the severity of symptoms, as has been seen in male patients,” he said.
“These data are very interesting and raise a fascinating hypothesis,” said Richard Martin, PhD, professor of clinical epidemiology at the University of Bristol, UK, commenting about the study. “But they do need independent validation in other large population-wide datasets...with appropriate statistical analysis including adjustment for important risk factors for SARS-CoV-2.”
He noted that the Italian study results were not adjusted for potential confounders, for example, age, body mass index, and cardiometabolic comorbidities, that are strong risk factors for SARS-CoV-2. In addition, men taking ADT may have been more likely to self-isolate and so be at reduced risk of getting the infection, he suggested.
How Do Androgens Interact With the Virus?
Alimonti and colleagues offer a mechanistic explanation of how androgens interact with the virus.
Coronavirus gains entry into the human cell by binding its viral spike (S) proteins to ACE2 and on S protein priming by TMPRSS2. TMPRSS2 is a member of a family of proteins called type II transmembrane serine proteases, which are involved in a number of processes including cancer and viral infections, they explain.
“Intriguingly, TMPRSS2 is an androgen-regulated gene that is upregulated in prostate cancer where it supports tumor progression,” they point out.
There is also evidence that the same androgen receptor regulates TMPRSS2 expression in nonprostatic tissues, including the lungs.
“[This] may explain the increased susceptibility of men to develop SARS-CoV-2 severe infections when compared to women,” the authors speculate.
Because ADT is known to decrease TMPRSS2 levels, they suggest that androgen receptor antagonists “could be used to block or decrease the severity of SARS-CoV-2 infection in male patients.”
They go even further and suggest that men without prostate cancer at high risk for COVID-19 could take ADT to prevent infection.
For men who do become infected with COVID-19, ADT might also help reduce symptom severity, they add.
Given that the effects of androgen receptor antagonists are reversible, “they could be used transiently (eg, 1 month) in patients affected by SARS-CoV-2, thereby reducing the risk of side effects due to long-term administration,” the authors suggest.
Another Theory: Is Estrogen Protective?
Another theory to explain the male/female difference for severe COVID-19 is that the female hormone estrogen may be protective.
“People have to stop putting estrogen in that ‘female hormone box’ because it’s a molecule that we all use as humans, it’s just not women,” Sharon Nachman, MD, told Medscape Medical News.
“Looking at estrogen as having potentially important immune effects is part of thinking outside the box,” she said.
Nachman is associate dean for research at the Renaissance School of Medicine, Stony Brook University in New York, and is working together with Antonios Gasparis, MD, professor of surgery at the same center.
They are exploring the use of a transdermal estrogen patch in patients with COVID-19 in a randomized trial with a placebo-controlled arm. They are recruiting patients who present to their emergency department with signs and symptoms of COVID-19, and enroll them into the trial if they are interested.
“We are testing everyone as well, but we are starting patients on the medication at the time of entry as opposed to waiting until we have a test result back,” Nachman explained.
The primary objective of the study is to evaluate whether the transdermal patch, applied to the skin for 7 days, might reduce the need for intubation in men and women infected with COVID-19 versus standard of care.
The product is the same single-use transdermal estradiol patch (Climara, 25 cm2, Bayer) prescribed for postmenopausal women and will be used at the same dose, which is known to be safe.
After the patch is removed, patients will be carefully tracked for symptoms over the next 45 days to see if the patch reduced symptom severity, and if so, in which patients.
Nachman would have preferred to enroll patients before they had overt symptoms, but this simply isn’t possible in a medical center where symptomatic patients present, she told Medscape Medical News.
However, she does know that even at their own medical center, the odds are stacked against male COVID-19 patients — and something is needed to mitigate its severity in this patient group.
As they were developing the protocol for the current study, the team decided to see who was in their ICU during a single study day.
The answer: mostly males. Intubation and death rates in men in their ICU for that single day was approximately 80% compared with only 20% among women.
“We have a new horrific pathogen that is pandemic and we’re all probably going to get it, it’s just a question of when and how sick we’ll be from it,” Nachman said.
Alimonti and coauthors have reported no relevant financial relationships, as did Goulder and Nachman.
This article first appeared on Medscape.com.
Pandemic-related stress rising among ICU clinicians
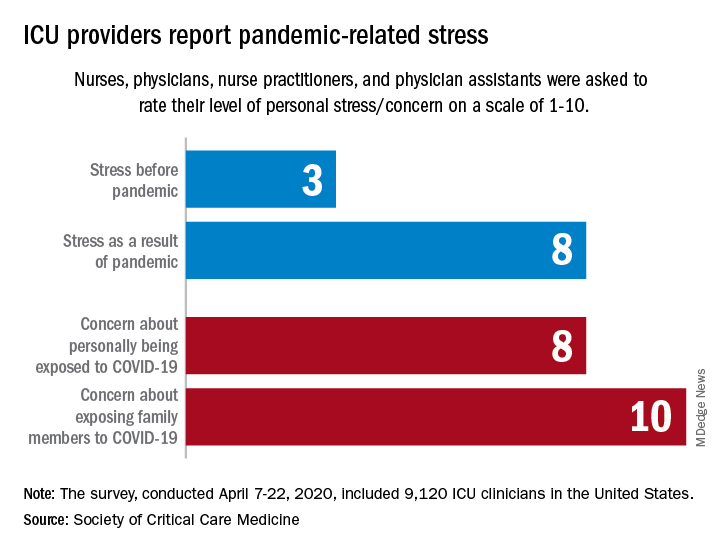
They are worried about getting infected, and they are even more worried about infecting family members, according to the Society for Critical Care Medicine, which surveyed members of four professional organizations – the American Association of Critical-Care Nurses, American College of Chest Physicians, American Thoracic Society, and the SCCM – April 7-22, 2020.
Four items in the survey assessed respondents’ level of stress or concern on a scale of 1-10:
- Personal stress before the COVID-19 pandemic.
- Personal stress as a result of COVID-19 pandemic.
- Concern about personally being exposed to COVID-19.
- Concern about exposing family members to COVID-19.
Personal stress rose from a median of 3 before the pandemic to a current 8, a level that was equaled by personal concerns about being exposed and surpassed (10) by concerns about exposing family members, the SCCM reported in a blog post.
Most of the respondents “are taking special measures to limit the potential spread of the virus to their loved ones, including implementing a decontamination routine before interacting with families,” the SCCM wrote.
The most common strategy, employed by 72% of ICU clinicians, is changing clothes before/after work. Showering before joining family was mentioned by 64% of providers, followed by limiting contact until decontamination (57%) and using hand sanitizer before entering home (51%), the SCCM said.
More extreme measures included self-isolating within their homes (16%) and staying in alternative housing away from their families (12%), the SCCM said, based on data for 9,120 clinicians in the United States.
Most of the respondents (88%) reported having cared for a patient with confirmed or presumed COVID-19. Nurses made up the majority (91%) of the sample, which also included nurse practitioners and physician assistants (4.5%) and physicians (2.9%), as well as smaller numbers of respiratory therapists, pharmacists, and emergency medicine flight personnel.
The results of the survey “underline the personal sacrifices of critical care clinicians during the COVID-19 response and suggest the need to help them proactively manage stress,” the SCCM wrote.

They are worried about getting infected, and they are even more worried about infecting family members, according to the Society for Critical Care Medicine, which surveyed members of four professional organizations – the American Association of Critical-Care Nurses, American College of Chest Physicians, American Thoracic Society, and the SCCM – April 7-22, 2020.
Four items in the survey assessed respondents’ level of stress or concern on a scale of 1-10:
- Personal stress before the COVID-19 pandemic.
- Personal stress as a result of COVID-19 pandemic.
- Concern about personally being exposed to COVID-19.
- Concern about exposing family members to COVID-19.
Personal stress rose from a median of 3 before the pandemic to a current 8, a level that was equaled by personal concerns about being exposed and surpassed (10) by concerns about exposing family members, the SCCM reported in a blog post.
Most of the respondents “are taking special measures to limit the potential spread of the virus to their loved ones, including implementing a decontamination routine before interacting with families,” the SCCM wrote.
The most common strategy, employed by 72% of ICU clinicians, is changing clothes before/after work. Showering before joining family was mentioned by 64% of providers, followed by limiting contact until decontamination (57%) and using hand sanitizer before entering home (51%), the SCCM said.
More extreme measures included self-isolating within their homes (16%) and staying in alternative housing away from their families (12%), the SCCM said, based on data for 9,120 clinicians in the United States.
Most of the respondents (88%) reported having cared for a patient with confirmed or presumed COVID-19. Nurses made up the majority (91%) of the sample, which also included nurse practitioners and physician assistants (4.5%) and physicians (2.9%), as well as smaller numbers of respiratory therapists, pharmacists, and emergency medicine flight personnel.
The results of the survey “underline the personal sacrifices of critical care clinicians during the COVID-19 response and suggest the need to help them proactively manage stress,” the SCCM wrote.

They are worried about getting infected, and they are even more worried about infecting family members, according to the Society for Critical Care Medicine, which surveyed members of four professional organizations – the American Association of Critical-Care Nurses, American College of Chest Physicians, American Thoracic Society, and the SCCM – April 7-22, 2020.
Four items in the survey assessed respondents’ level of stress or concern on a scale of 1-10:
- Personal stress before the COVID-19 pandemic.
- Personal stress as a result of COVID-19 pandemic.
- Concern about personally being exposed to COVID-19.
- Concern about exposing family members to COVID-19.
Personal stress rose from a median of 3 before the pandemic to a current 8, a level that was equaled by personal concerns about being exposed and surpassed (10) by concerns about exposing family members, the SCCM reported in a blog post.
Most of the respondents “are taking special measures to limit the potential spread of the virus to their loved ones, including implementing a decontamination routine before interacting with families,” the SCCM wrote.
The most common strategy, employed by 72% of ICU clinicians, is changing clothes before/after work. Showering before joining family was mentioned by 64% of providers, followed by limiting contact until decontamination (57%) and using hand sanitizer before entering home (51%), the SCCM said.
More extreme measures included self-isolating within their homes (16%) and staying in alternative housing away from their families (12%), the SCCM said, based on data for 9,120 clinicians in the United States.
Most of the respondents (88%) reported having cared for a patient with confirmed or presumed COVID-19. Nurses made up the majority (91%) of the sample, which also included nurse practitioners and physician assistants (4.5%) and physicians (2.9%), as well as smaller numbers of respiratory therapists, pharmacists, and emergency medicine flight personnel.
The results of the survey “underline the personal sacrifices of critical care clinicians during the COVID-19 response and suggest the need to help them proactively manage stress,” the SCCM wrote.
More guidance on inpatient management of blood glucose in COVID-19
“The glycemic management of many COVID-19–positive patients with diabetes is proving extremely complex, with huge fluctuations in glucose control and the need for very high doses of insulin,” says Diabetes UK’s National Diabetes Inpatient COVID Response Team.
“Intravenous infusion pumps, also required for inotropes, are at a premium and there may be the need to consider the use of subcutaneous or intramuscular insulin protocols,” they note.
Updated as of April 29, all of the information of the National Diabetes Inpatient COVID Response Team is available on the Diabetes UK website.
The new inpatient management graphic adds more detail to the previous “front-door” guidance, as reported by Medscape Medical News.
The document stressed that, as well as identifying patients with known diabetes, it is imperative that all newly admitted patients with COVID-19 are evaluated for diabetes, as the infection is known to cause new-onset diabetes.
Subcutaneous insulin dosing
The new graphic gives extensive details on subcutaneous insulin dosing in place of variable rate intravenous insulin when infusion pumps are not available, and when the patient has a glucose level above 12 mmol/L (216 mg/dL) but does not have DKA or hyperosmolar hyperglycemic state.
However, the advice is not intended for people with COVID-19 causing severe insulin resistance in the intensive care unit.
The other new guidance graphic on managing DKA or hyperosmolar state in people with COVID-19 using subcutaneous insulin is also intended for situations where intravenous infusion isn’t available.
Seek help from specialist diabetes team when needed
This is not to be used for mixed DKA/hyperosmolar state or for patients who are pregnant, have severe metabolic derangement, other significant comorbidity, or impaired consciousness, however.
For those situations, the advice is to seek help from a specialist diabetes team, says Diabetes UK.
Specialist teams will be available to answer diabetes queries, both by signposting to relevant existing local documents and also by providing patient-specific advice.
Indeed, NHS England recommends that such a team be available in every hospital, with a lead consultant designated each day to co-ordinate these services who must be free of other clinical duties when doing so. The role involves co-ordination of the whole service from the emergency department through to liaison with other specialties and managers.
Also newly updated is a page with extensive information for patients, including advice for staying at home, medication use, self-isolating, shielding, hospital and doctor appointments, need for urgent medical advice, and going to the hospital.
It also covers how coronavirus can affect people with diabetes, children and school, pregnancy, work situations, and tips for picking up prescriptions.
Another, shorter document with COVID-19 advice for patients has been posted by the JDRF and Beyond Type 1 Alliance.
It has also been endorsed by the American Diabetes Association, Harvard Medical School, and International Society for Pediatric and Adolescent Diabetes, in partnership with many other professional organizations, including the International Diabetes Federation, American Association of Clinical Endocrinologists, and Association of Diabetes Care & Education Specialists.
The shorter document covers topics such as personal hygiene, distancing, diabetes management, and seeking treatment, as well as links to other resources on what to do when health insurance is lost and legal rights.
This article first appeared on Medscape.com.
“The glycemic management of many COVID-19–positive patients with diabetes is proving extremely complex, with huge fluctuations in glucose control and the need for very high doses of insulin,” says Diabetes UK’s National Diabetes Inpatient COVID Response Team.
“Intravenous infusion pumps, also required for inotropes, are at a premium and there may be the need to consider the use of subcutaneous or intramuscular insulin protocols,” they note.
Updated as of April 29, all of the information of the National Diabetes Inpatient COVID Response Team is available on the Diabetes UK website.
The new inpatient management graphic adds more detail to the previous “front-door” guidance, as reported by Medscape Medical News.
The document stressed that, as well as identifying patients with known diabetes, it is imperative that all newly admitted patients with COVID-19 are evaluated for diabetes, as the infection is known to cause new-onset diabetes.
Subcutaneous insulin dosing
The new graphic gives extensive details on subcutaneous insulin dosing in place of variable rate intravenous insulin when infusion pumps are not available, and when the patient has a glucose level above 12 mmol/L (216 mg/dL) but does not have DKA or hyperosmolar hyperglycemic state.
However, the advice is not intended for people with COVID-19 causing severe insulin resistance in the intensive care unit.
The other new guidance graphic on managing DKA or hyperosmolar state in people with COVID-19 using subcutaneous insulin is also intended for situations where intravenous infusion isn’t available.
Seek help from specialist diabetes team when needed
This is not to be used for mixed DKA/hyperosmolar state or for patients who are pregnant, have severe metabolic derangement, other significant comorbidity, or impaired consciousness, however.
For those situations, the advice is to seek help from a specialist diabetes team, says Diabetes UK.
Specialist teams will be available to answer diabetes queries, both by signposting to relevant existing local documents and also by providing patient-specific advice.
Indeed, NHS England recommends that such a team be available in every hospital, with a lead consultant designated each day to co-ordinate these services who must be free of other clinical duties when doing so. The role involves co-ordination of the whole service from the emergency department through to liaison with other specialties and managers.
Also newly updated is a page with extensive information for patients, including advice for staying at home, medication use, self-isolating, shielding, hospital and doctor appointments, need for urgent medical advice, and going to the hospital.
It also covers how coronavirus can affect people with diabetes, children and school, pregnancy, work situations, and tips for picking up prescriptions.
Another, shorter document with COVID-19 advice for patients has been posted by the JDRF and Beyond Type 1 Alliance.
It has also been endorsed by the American Diabetes Association, Harvard Medical School, and International Society for Pediatric and Adolescent Diabetes, in partnership with many other professional organizations, including the International Diabetes Federation, American Association of Clinical Endocrinologists, and Association of Diabetes Care & Education Specialists.
The shorter document covers topics such as personal hygiene, distancing, diabetes management, and seeking treatment, as well as links to other resources on what to do when health insurance is lost and legal rights.
This article first appeared on Medscape.com.
“The glycemic management of many COVID-19–positive patients with diabetes is proving extremely complex, with huge fluctuations in glucose control and the need for very high doses of insulin,” says Diabetes UK’s National Diabetes Inpatient COVID Response Team.
“Intravenous infusion pumps, also required for inotropes, are at a premium and there may be the need to consider the use of subcutaneous or intramuscular insulin protocols,” they note.
Updated as of April 29, all of the information of the National Diabetes Inpatient COVID Response Team is available on the Diabetes UK website.
The new inpatient management graphic adds more detail to the previous “front-door” guidance, as reported by Medscape Medical News.
The document stressed that, as well as identifying patients with known diabetes, it is imperative that all newly admitted patients with COVID-19 are evaluated for diabetes, as the infection is known to cause new-onset diabetes.
Subcutaneous insulin dosing
The new graphic gives extensive details on subcutaneous insulin dosing in place of variable rate intravenous insulin when infusion pumps are not available, and when the patient has a glucose level above 12 mmol/L (216 mg/dL) but does not have DKA or hyperosmolar hyperglycemic state.
However, the advice is not intended for people with COVID-19 causing severe insulin resistance in the intensive care unit.
The other new guidance graphic on managing DKA or hyperosmolar state in people with COVID-19 using subcutaneous insulin is also intended for situations where intravenous infusion isn’t available.
Seek help from specialist diabetes team when needed
This is not to be used for mixed DKA/hyperosmolar state or for patients who are pregnant, have severe metabolic derangement, other significant comorbidity, or impaired consciousness, however.
For those situations, the advice is to seek help from a specialist diabetes team, says Diabetes UK.
Specialist teams will be available to answer diabetes queries, both by signposting to relevant existing local documents and also by providing patient-specific advice.
Indeed, NHS England recommends that such a team be available in every hospital, with a lead consultant designated each day to co-ordinate these services who must be free of other clinical duties when doing so. The role involves co-ordination of the whole service from the emergency department through to liaison with other specialties and managers.
Also newly updated is a page with extensive information for patients, including advice for staying at home, medication use, self-isolating, shielding, hospital and doctor appointments, need for urgent medical advice, and going to the hospital.
It also covers how coronavirus can affect people with diabetes, children and school, pregnancy, work situations, and tips for picking up prescriptions.
Another, shorter document with COVID-19 advice for patients has been posted by the JDRF and Beyond Type 1 Alliance.
It has also been endorsed by the American Diabetes Association, Harvard Medical School, and International Society for Pediatric and Adolescent Diabetes, in partnership with many other professional organizations, including the International Diabetes Federation, American Association of Clinical Endocrinologists, and Association of Diabetes Care & Education Specialists.
The shorter document covers topics such as personal hygiene, distancing, diabetes management, and seeking treatment, as well as links to other resources on what to do when health insurance is lost and legal rights.
This article first appeared on Medscape.com.
Results from 11 AHA-funded COVID-19 studies expected within months
Work is set to start in June, with findings reported in as few as 6 months. The Cleveland Clinic will coordinate the efforts, collecting and disseminating the findings.
There were more than 750 research proposals in less than a month after the association announced its COVID-19 and Its Cardiovascular Impact Rapid Response Grant initiative.
“We were just blown away and so impressed to see this level of interest and commitment from the teams submitting such thorough proposals so quickly,” AHA President Robert Harrington, MD, chair of the department of medicine at Stanford (Calif.) University, said in a press statement. “There’s so much we don’t know about this unique coronavirus, and we continue to see emerging complications affecting both heart and brain health for which we desperately need answers and we need them quickly.”
The projects include the following:
- A Comprehensive Assessment of Arterial and Venous Thrombotic Complications in Patients with COVID-19, led by Columbia University, New York City.
- Repurposing Drugs for Treatment of Cardiomyopathy Caused by Coronavirus-2 (SARS-CoV-2), led by Brigham and Women’s Hospital and Harvard Medical School, Boston.
- Risk of Severe Morbidity and Mortality of Coronavirus Disease 2019 (COVID-19) Among Patients Taking Antihypertensive Medications, led by Kaiser Permanente Southern California.
- Deep Learning Using Chest Radiographs to Predict COVID-19 Cardiopulmonary Risk, led by Massachusetts General Hospital, Boston.
- Cardiovascular Outcomes and Biomarker Titrated Corticosteroid Dosing for SARS COV-2 (COVID-19): A Randomized Controlled Trial, led by the Mayo Clinic, Rochester Minn.
- Outcomes for Patients With Hypertension, Diabetes, and Heart Disease in the Coronavirus Pandemic: Impact of Angiotensin Converting Enzyme Inhibitors and Angiotensin Receptor Blockers Treatment, led by Stanford University.
- Rapid COVID-19-on-A-Chip to Screen Competitive Targets for SARS-CoV-2 Spike Binding Sites, led by University of California, Los Angeles.
- COVID-19 Infection, African American Women and Cardiovascular Health, led by University of California, San Francisco.
- Myocardial Virus and Gene Expression in SARS CoV-2 Positive Patients with Clinically Important Myocardial Dysfunction, led by the University of Colorado, Aurora.
- The Role of the Platelet in Mediating Cardiovascular Disease in SARS-CoV-2 Infection, led by the University of Massachusetts, Worcester.
- Harnessing Glycomics to Understand Myocardial Injury in COVID-19, led by the University of Nebraska Medical Center, Omaha.
The AHA also awarded $800,000 for short-term projects to members of its new Health Technologies & Innovation Strategically Focused Research Network.
Cincinnati Children’s Hospital will assess the use of ejection fraction to triage COVID-19 patients; Johns Hopkins University, Baltimore, will assess smartphones for “virtual check-in” for stroke symptoms; Stanford will assess digital tracking of COVID-19 patients with cardiovascular complications; and the University of Michigan, Ann Arbor, will assess a system to track physiological and cardiovascular consequences of the infection.
Work is set to start in June, with findings reported in as few as 6 months. The Cleveland Clinic will coordinate the efforts, collecting and disseminating the findings.
There were more than 750 research proposals in less than a month after the association announced its COVID-19 and Its Cardiovascular Impact Rapid Response Grant initiative.
“We were just blown away and so impressed to see this level of interest and commitment from the teams submitting such thorough proposals so quickly,” AHA President Robert Harrington, MD, chair of the department of medicine at Stanford (Calif.) University, said in a press statement. “There’s so much we don’t know about this unique coronavirus, and we continue to see emerging complications affecting both heart and brain health for which we desperately need answers and we need them quickly.”
The projects include the following:
- A Comprehensive Assessment of Arterial and Venous Thrombotic Complications in Patients with COVID-19, led by Columbia University, New York City.
- Repurposing Drugs for Treatment of Cardiomyopathy Caused by Coronavirus-2 (SARS-CoV-2), led by Brigham and Women’s Hospital and Harvard Medical School, Boston.
- Risk of Severe Morbidity and Mortality of Coronavirus Disease 2019 (COVID-19) Among Patients Taking Antihypertensive Medications, led by Kaiser Permanente Southern California.
- Deep Learning Using Chest Radiographs to Predict COVID-19 Cardiopulmonary Risk, led by Massachusetts General Hospital, Boston.
- Cardiovascular Outcomes and Biomarker Titrated Corticosteroid Dosing for SARS COV-2 (COVID-19): A Randomized Controlled Trial, led by the Mayo Clinic, Rochester Minn.
- Outcomes for Patients With Hypertension, Diabetes, and Heart Disease in the Coronavirus Pandemic: Impact of Angiotensin Converting Enzyme Inhibitors and Angiotensin Receptor Blockers Treatment, led by Stanford University.
- Rapid COVID-19-on-A-Chip to Screen Competitive Targets for SARS-CoV-2 Spike Binding Sites, led by University of California, Los Angeles.
- COVID-19 Infection, African American Women and Cardiovascular Health, led by University of California, San Francisco.
- Myocardial Virus and Gene Expression in SARS CoV-2 Positive Patients with Clinically Important Myocardial Dysfunction, led by the University of Colorado, Aurora.
- The Role of the Platelet in Mediating Cardiovascular Disease in SARS-CoV-2 Infection, led by the University of Massachusetts, Worcester.
- Harnessing Glycomics to Understand Myocardial Injury in COVID-19, led by the University of Nebraska Medical Center, Omaha.
The AHA also awarded $800,000 for short-term projects to members of its new Health Technologies & Innovation Strategically Focused Research Network.
Cincinnati Children’s Hospital will assess the use of ejection fraction to triage COVID-19 patients; Johns Hopkins University, Baltimore, will assess smartphones for “virtual check-in” for stroke symptoms; Stanford will assess digital tracking of COVID-19 patients with cardiovascular complications; and the University of Michigan, Ann Arbor, will assess a system to track physiological and cardiovascular consequences of the infection.
Work is set to start in June, with findings reported in as few as 6 months. The Cleveland Clinic will coordinate the efforts, collecting and disseminating the findings.
There were more than 750 research proposals in less than a month after the association announced its COVID-19 and Its Cardiovascular Impact Rapid Response Grant initiative.
“We were just blown away and so impressed to see this level of interest and commitment from the teams submitting such thorough proposals so quickly,” AHA President Robert Harrington, MD, chair of the department of medicine at Stanford (Calif.) University, said in a press statement. “There’s so much we don’t know about this unique coronavirus, and we continue to see emerging complications affecting both heart and brain health for which we desperately need answers and we need them quickly.”
The projects include the following:
- A Comprehensive Assessment of Arterial and Venous Thrombotic Complications in Patients with COVID-19, led by Columbia University, New York City.
- Repurposing Drugs for Treatment of Cardiomyopathy Caused by Coronavirus-2 (SARS-CoV-2), led by Brigham and Women’s Hospital and Harvard Medical School, Boston.
- Risk of Severe Morbidity and Mortality of Coronavirus Disease 2019 (COVID-19) Among Patients Taking Antihypertensive Medications, led by Kaiser Permanente Southern California.
- Deep Learning Using Chest Radiographs to Predict COVID-19 Cardiopulmonary Risk, led by Massachusetts General Hospital, Boston.
- Cardiovascular Outcomes and Biomarker Titrated Corticosteroid Dosing for SARS COV-2 (COVID-19): A Randomized Controlled Trial, led by the Mayo Clinic, Rochester Minn.
- Outcomes for Patients With Hypertension, Diabetes, and Heart Disease in the Coronavirus Pandemic: Impact of Angiotensin Converting Enzyme Inhibitors and Angiotensin Receptor Blockers Treatment, led by Stanford University.
- Rapid COVID-19-on-A-Chip to Screen Competitive Targets for SARS-CoV-2 Spike Binding Sites, led by University of California, Los Angeles.
- COVID-19 Infection, African American Women and Cardiovascular Health, led by University of California, San Francisco.
- Myocardial Virus and Gene Expression in SARS CoV-2 Positive Patients with Clinically Important Myocardial Dysfunction, led by the University of Colorado, Aurora.
- The Role of the Platelet in Mediating Cardiovascular Disease in SARS-CoV-2 Infection, led by the University of Massachusetts, Worcester.
- Harnessing Glycomics to Understand Myocardial Injury in COVID-19, led by the University of Nebraska Medical Center, Omaha.
The AHA also awarded $800,000 for short-term projects to members of its new Health Technologies & Innovation Strategically Focused Research Network.
Cincinnati Children’s Hospital will assess the use of ejection fraction to triage COVID-19 patients; Johns Hopkins University, Baltimore, will assess smartphones for “virtual check-in” for stroke symptoms; Stanford will assess digital tracking of COVID-19 patients with cardiovascular complications; and the University of Michigan, Ann Arbor, will assess a system to track physiological and cardiovascular consequences of the infection.
COVID-19–associated coagulopathy
Coronavirus disease 2019 (COVID-19) is a viral illness caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), currently causing a pandemic affecting many countries around the world, beginning in December 2019 and spreading rapidly on a global scale since. Globally, its burden has been increasing rapidly, with more than 1.2 million people testing positive for the illness and 123,000 people losing their lives, as per April 15th’s WHO COVID-19 Situation Report.1 These numbers are increasing with each passing day. Clinically, SARS-CoV-2 has a highly variable course, ranging from mild disease manifested as a self-limited illness (seen in younger and healthier patients) to severe pneumonia/ARDS and multiorgan failure with intravascular coagulopathy.2
In this article, we intend to investigate and establish a comprehensive review of COVID-19–associated coagulopathy mechanisms, laboratory findings, and current management guidelines put forth by various societies globally.
Mechanism of coagulopathy
COVID-19–associated coagulopathy has been shown to predispose to both arterial and venous thrombosis through excessive inflammation and hypoxia, leading to activation of the coagulation cascade and consumption of coagulation factors, resulting in microvascular thrombosis.3 Though the exact pathophysiology for the activation of this cascade is not known, the proposed mechanism has been: endothelial damage triggering platelet activation within the lung, leading to aggregation, thrombosis, and consumption of platelets in the lung.2,5,6
Fox et al. noted similar coagulopathy findings of four deceased COVID-19 patients. Autopsy results concluded that the dominant process was diffuse alveolar damage, notable CD4+ aggregates around thrombosed small vessels, significant associated hemorrhage, and thrombotic microangiopathy restricted to the lungs. The proposed mechanism was the activation of megakaryocytes, possibly native to the lung, with platelet aggregation, formation of platelet-rich clots, and fibrin deposition playing a major role.4
It has been noted that diabetic patients are at an increased risk of vascular events and hypercoagulability with COVID-19.7 COVID-19 can also cause livedo reticularis and acrocyanosis because of the microthrombosis in the cutaneous vasculature secondary to underlying coagulopathy, as reported in a case report of two U.S. patients with COVID-19.8
Clinical and laboratory abnormalities
A recent study reported from Netherlands by Klok et al. analyzed 184 ICU patients with COVID-19 pneumonia and concluded that the cumulative incidence of acute pulmonary embolism (PE), deep vein thrombosis (DVT), ischemic stroke, MI, or systemic arterial embolism was 31% (95% confidence interval, 20%-41%). PE was the most frequent thrombotic complication and was noted in 81% of patients. Coagulopathy, defined as spontaneous prolongation of prothrombin time (PT) > 3s or activated partial thromboplastin time (aPTT) > 5s, was reported as an independent predictor of thrombotic complications.3
Hematologic abnormalities that were noted in COVID-19 coagulopathy include: decreased platelet counts, decreased fibrinogen levels, elevated PT/INR, elevated partial thromboplastin time (PTT), and elevated d-dimer.9,10 In a retrospective analysis9 by Tang et al., 71.4% of nonsurvivors and 0.6% of survivors had met the criteria of disseminated intravascular coagulation (DIC) during their hospital stay. Nonsurvivors of COVID-19 had statistically significant elevation of d-dimer levels, FDP levels, PT, and aPTT, when compared to survivors (P < .05). The overall mortality in this study was reported as 11.5%.9 In addition, elevated d-dimer, fibrin and fibrinogen degradation product (FDP) levels and longer PT and aPTT were associated with poor prognosis.
Thus, d-dimer, PT, and platelet count should be measured in all patients who present with COVID-19 infection. We can also suggest that in patients with markedly elevated d-dimer (three- to fourfold increase), admission to hospital should be considered even in the absence of severe clinical symptoms.11
COVID-19 coagulopathy management
In a retrospective study9 of 449 patients with severe COVID-19 from Wuhan, China, by Tang et al., 99 patients mainly received low-weight molecular heparin (LMWH) for 7 days or longer. No difference in 28-day mortality was noted between heparin users and nonusers (30.3% vs. 29.7%; P = .910). A lower 28-day mortality rate was noted in heparin patients with sepsis-induced coagulopathy score of ≥4.0 (40.0% vs. 64.2%; P = .029) or a d-dimer level greater than sixfold of upper limit of normal, compared with nonusers of heparin.12
Another small study of seven COVID-19 patients with acroischemia in China demonstrated that administering LMWH was successful at decreasing the d-dimer and fibrinogen degradation product levels but noted no significant improvement in clinical symptoms.13
Recently, the International Society of Thrombosis and Hemostasis and American Society of Hematology published recommendations and guidelines regarding the recognition and management of coagulopathy in COVID-19.11 Prophylactic anticoagulation therapy with LMWH was recommended in all hospitalized patients with COVID-19, provided there was an absence of any contraindications (active bleeding, platelet count less than 25 x 109/L and fibrinogen less than 0.5 g/dL). Anticoagulation with LMWH was associated with better prognosis in severe COVID-19 patients and in COVID-19 patients with markedly elevated d-dimer, as it also has anti-inflammatory effects.12 This anti-inflammatory property of heparin has been documented in previous studies but the underlying mechanism is unknown and more research is required.14,15
Despite coagulopathy being noticed with cases of COVID-19, bleeding has been a rare finding in COVID-19 infections. If bleeding is noted, recommendations were made to keep platelet levels greater than 50 x109/L, fibrinogen less than 2.0 g/L, and INR [international normalized ratio] greater than 1.5.11 Mechanical thromboprophylaxis should be used when pharmacologic thromboprophylaxis is contraindicated.16
COVID-19 patients with new diagnoses of venous thromboembolism (VTE) or atrial fibrillation should be prescribed therapeutic anticoagulation. Patients who are already on anticoagulation for VTE or atrial fibrillation should continue their therapy unless the platelet count is less than 30-50x109/L or if the fibrinogen is less than 1.0 g/L.16
Conclusion
Coagulopathies associated with COVID-19 infections have been documented in several studies around the world, and it has been shown to be fatal in some cases. Despite documentation, the mechanism behind this coagulopathy is not well understood. Because of the potentially lethal complications associated with coagulopathies, early recognition and anticoagulation is imperative to improve clinical outcomes. These results are very preliminary: More studies are required to understand the role of anticoagulation and its effect on the morbidity and mortality associated with COVID-19–associated coagulopathy.
Dr. Yeruva is a board-certified hematologist/medical oncologist with WellSpan Health and clinical assistant professor of internal medicine, Penn State University, Hershey. Mr. Henderson is a third-year graduate-entry medical student at the Royal College of Surgeons in Ireland with interests in family medicine, dermatology, and tropical diseases. Dr. Al-Tawfiq is a consultant of internal medicine & infectious diseases, and the director of quality at Johns Hopkins Aramco Healthcare in Dhahran, Saudi Arabia, an adjunct associate professor of infectious diseases, molecular medicine and clinical pharmacology at Johns Hopkins University School of Medicine, and adjunct associate professor at Indiana University School of Medicine, Indianapolis. Dr. Tirupathi is the medical director of Keystone Infectious Diseases/HIV in Chambersburg, Pa., and currently chair of infection prevention at Wellspan Chambersburg and Waynesboro (Pa.) Hospitals. He also is the lead physician for antibiotic stewardship at these hospitals.
References
1. World Health Organization. Coronavirus disease (COVID-2019) situation reports.
2. Lippi G et al. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin Chim Acta. 2020 Mar 13. 506:145-8. doi: 10.1016/j.cca.2020.03.022.
3. Klok FA et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Throm Res. 2020;18(4):844-7. doi: 10.1016/j.thromres.2020.04.013.
4. Fox S et al. Pulmonary and cardiac pathology in Covid-19: The first autopsy series from New Orleans. MedRxiv. 2020 Apr 10. doi: 10.1101/2020.04.06.20050575.
5. Yang M et al. Thrombocytopenia in patients with severe acute respiratory syndrome (review). Hematology 2013 Sep 4. doi: 10.1080/1024533040002617.
6. Giannis D et al. Coagulation disorders in coronavirus infected patients: COVID-19, SARS-CoV-1, MERS-CoV and lessons from the past. J Clin Virol. 2020 June. doi: 10.1016/j.jcv.2020.104362.
7. Guo W et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020 Mar 31. doi: 10.1002/dmrr.3319.
8. Manalo IF et al. A dermatologic manifestation of COVID-19: Transient livedo reticularis. J Am Acad Dermat. 2020 Apr. doi: 10.1016/j.jaad.2020.04.018.
9. Tang N et al. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020 Feb 19. doi: 10.1111/jth.14768, 18: 844-847.
10. Huang C et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020 Jan 24. doi: 10.1016/S0140-6736(20)30183-5.
11. Thachil J et al. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J Thromb Haemost. 2020 Mar 25. doi: 10.1111/JTH.14810.
12. Tang N et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020 Mar 27. doi: 10.1111/JTH.14817.
13. Zhang Y et al. Clinical and coagulation characteristics of 7 patients with critical COVID-2019 pneumonia and acro-ischemia. Zhonghua Xue Ye Xue Za Zhi. 2020 Mar 28. doi: 10.3760/cma.j.issn.0253-2727.2020.0006.
14. Poterucha TJ et al. More than an anticoagulant: Do heparins have direct anti-inflammatory effects? Thromb Haemost. 2017. doi: 10.1160/TH16-08-0620.
15. Mousavi S et al. Anti-inflammatory effects of heparin and its derivatives: A systematic review. Adv Pharmacol Pharm Sci. 2015 May 12. doi: 10.1155/2015/507151.
16. Kreuziger L et al. COVID-19 and VTE/anticoagulation: Frequently asked questions. American Society of Hematology. 2020 Apr 17.
Coronavirus disease 2019 (COVID-19) is a viral illness caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), currently causing a pandemic affecting many countries around the world, beginning in December 2019 and spreading rapidly on a global scale since. Globally, its burden has been increasing rapidly, with more than 1.2 million people testing positive for the illness and 123,000 people losing their lives, as per April 15th’s WHO COVID-19 Situation Report.1 These numbers are increasing with each passing day. Clinically, SARS-CoV-2 has a highly variable course, ranging from mild disease manifested as a self-limited illness (seen in younger and healthier patients) to severe pneumonia/ARDS and multiorgan failure with intravascular coagulopathy.2
In this article, we intend to investigate and establish a comprehensive review of COVID-19–associated coagulopathy mechanisms, laboratory findings, and current management guidelines put forth by various societies globally.
Mechanism of coagulopathy
COVID-19–associated coagulopathy has been shown to predispose to both arterial and venous thrombosis through excessive inflammation and hypoxia, leading to activation of the coagulation cascade and consumption of coagulation factors, resulting in microvascular thrombosis.3 Though the exact pathophysiology for the activation of this cascade is not known, the proposed mechanism has been: endothelial damage triggering platelet activation within the lung, leading to aggregation, thrombosis, and consumption of platelets in the lung.2,5,6
Fox et al. noted similar coagulopathy findings of four deceased COVID-19 patients. Autopsy results concluded that the dominant process was diffuse alveolar damage, notable CD4+ aggregates around thrombosed small vessels, significant associated hemorrhage, and thrombotic microangiopathy restricted to the lungs. The proposed mechanism was the activation of megakaryocytes, possibly native to the lung, with platelet aggregation, formation of platelet-rich clots, and fibrin deposition playing a major role.4
It has been noted that diabetic patients are at an increased risk of vascular events and hypercoagulability with COVID-19.7 COVID-19 can also cause livedo reticularis and acrocyanosis because of the microthrombosis in the cutaneous vasculature secondary to underlying coagulopathy, as reported in a case report of two U.S. patients with COVID-19.8
Clinical and laboratory abnormalities
A recent study reported from Netherlands by Klok et al. analyzed 184 ICU patients with COVID-19 pneumonia and concluded that the cumulative incidence of acute pulmonary embolism (PE), deep vein thrombosis (DVT), ischemic stroke, MI, or systemic arterial embolism was 31% (95% confidence interval, 20%-41%). PE was the most frequent thrombotic complication and was noted in 81% of patients. Coagulopathy, defined as spontaneous prolongation of prothrombin time (PT) > 3s or activated partial thromboplastin time (aPTT) > 5s, was reported as an independent predictor of thrombotic complications.3
Hematologic abnormalities that were noted in COVID-19 coagulopathy include: decreased platelet counts, decreased fibrinogen levels, elevated PT/INR, elevated partial thromboplastin time (PTT), and elevated d-dimer.9,10 In a retrospective analysis9 by Tang et al., 71.4% of nonsurvivors and 0.6% of survivors had met the criteria of disseminated intravascular coagulation (DIC) during their hospital stay. Nonsurvivors of COVID-19 had statistically significant elevation of d-dimer levels, FDP levels, PT, and aPTT, when compared to survivors (P < .05). The overall mortality in this study was reported as 11.5%.9 In addition, elevated d-dimer, fibrin and fibrinogen degradation product (FDP) levels and longer PT and aPTT were associated with poor prognosis.
Thus, d-dimer, PT, and platelet count should be measured in all patients who present with COVID-19 infection. We can also suggest that in patients with markedly elevated d-dimer (three- to fourfold increase), admission to hospital should be considered even in the absence of severe clinical symptoms.11
COVID-19 coagulopathy management
In a retrospective study9 of 449 patients with severe COVID-19 from Wuhan, China, by Tang et al., 99 patients mainly received low-weight molecular heparin (LMWH) for 7 days or longer. No difference in 28-day mortality was noted between heparin users and nonusers (30.3% vs. 29.7%; P = .910). A lower 28-day mortality rate was noted in heparin patients with sepsis-induced coagulopathy score of ≥4.0 (40.0% vs. 64.2%; P = .029) or a d-dimer level greater than sixfold of upper limit of normal, compared with nonusers of heparin.12
Another small study of seven COVID-19 patients with acroischemia in China demonstrated that administering LMWH was successful at decreasing the d-dimer and fibrinogen degradation product levels but noted no significant improvement in clinical symptoms.13
Recently, the International Society of Thrombosis and Hemostasis and American Society of Hematology published recommendations and guidelines regarding the recognition and management of coagulopathy in COVID-19.11 Prophylactic anticoagulation therapy with LMWH was recommended in all hospitalized patients with COVID-19, provided there was an absence of any contraindications (active bleeding, platelet count less than 25 x 109/L and fibrinogen less than 0.5 g/dL). Anticoagulation with LMWH was associated with better prognosis in severe COVID-19 patients and in COVID-19 patients with markedly elevated d-dimer, as it also has anti-inflammatory effects.12 This anti-inflammatory property of heparin has been documented in previous studies but the underlying mechanism is unknown and more research is required.14,15
Despite coagulopathy being noticed with cases of COVID-19, bleeding has been a rare finding in COVID-19 infections. If bleeding is noted, recommendations were made to keep platelet levels greater than 50 x109/L, fibrinogen less than 2.0 g/L, and INR [international normalized ratio] greater than 1.5.11 Mechanical thromboprophylaxis should be used when pharmacologic thromboprophylaxis is contraindicated.16
COVID-19 patients with new diagnoses of venous thromboembolism (VTE) or atrial fibrillation should be prescribed therapeutic anticoagulation. Patients who are already on anticoagulation for VTE or atrial fibrillation should continue their therapy unless the platelet count is less than 30-50x109/L or if the fibrinogen is less than 1.0 g/L.16
Conclusion
Coagulopathies associated with COVID-19 infections have been documented in several studies around the world, and it has been shown to be fatal in some cases. Despite documentation, the mechanism behind this coagulopathy is not well understood. Because of the potentially lethal complications associated with coagulopathies, early recognition and anticoagulation is imperative to improve clinical outcomes. These results are very preliminary: More studies are required to understand the role of anticoagulation and its effect on the morbidity and mortality associated with COVID-19–associated coagulopathy.
Dr. Yeruva is a board-certified hematologist/medical oncologist with WellSpan Health and clinical assistant professor of internal medicine, Penn State University, Hershey. Mr. Henderson is a third-year graduate-entry medical student at the Royal College of Surgeons in Ireland with interests in family medicine, dermatology, and tropical diseases. Dr. Al-Tawfiq is a consultant of internal medicine & infectious diseases, and the director of quality at Johns Hopkins Aramco Healthcare in Dhahran, Saudi Arabia, an adjunct associate professor of infectious diseases, molecular medicine and clinical pharmacology at Johns Hopkins University School of Medicine, and adjunct associate professor at Indiana University School of Medicine, Indianapolis. Dr. Tirupathi is the medical director of Keystone Infectious Diseases/HIV in Chambersburg, Pa., and currently chair of infection prevention at Wellspan Chambersburg and Waynesboro (Pa.) Hospitals. He also is the lead physician for antibiotic stewardship at these hospitals.
References
1. World Health Organization. Coronavirus disease (COVID-2019) situation reports.
2. Lippi G et al. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin Chim Acta. 2020 Mar 13. 506:145-8. doi: 10.1016/j.cca.2020.03.022.
3. Klok FA et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Throm Res. 2020;18(4):844-7. doi: 10.1016/j.thromres.2020.04.013.
4. Fox S et al. Pulmonary and cardiac pathology in Covid-19: The first autopsy series from New Orleans. MedRxiv. 2020 Apr 10. doi: 10.1101/2020.04.06.20050575.
5. Yang M et al. Thrombocytopenia in patients with severe acute respiratory syndrome (review). Hematology 2013 Sep 4. doi: 10.1080/1024533040002617.
6. Giannis D et al. Coagulation disorders in coronavirus infected patients: COVID-19, SARS-CoV-1, MERS-CoV and lessons from the past. J Clin Virol. 2020 June. doi: 10.1016/j.jcv.2020.104362.
7. Guo W et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020 Mar 31. doi: 10.1002/dmrr.3319.
8. Manalo IF et al. A dermatologic manifestation of COVID-19: Transient livedo reticularis. J Am Acad Dermat. 2020 Apr. doi: 10.1016/j.jaad.2020.04.018.
9. Tang N et al. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020 Feb 19. doi: 10.1111/jth.14768, 18: 844-847.
10. Huang C et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020 Jan 24. doi: 10.1016/S0140-6736(20)30183-5.
11. Thachil J et al. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J Thromb Haemost. 2020 Mar 25. doi: 10.1111/JTH.14810.
12. Tang N et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020 Mar 27. doi: 10.1111/JTH.14817.
13. Zhang Y et al. Clinical and coagulation characteristics of 7 patients with critical COVID-2019 pneumonia and acro-ischemia. Zhonghua Xue Ye Xue Za Zhi. 2020 Mar 28. doi: 10.3760/cma.j.issn.0253-2727.2020.0006.
14. Poterucha TJ et al. More than an anticoagulant: Do heparins have direct anti-inflammatory effects? Thromb Haemost. 2017. doi: 10.1160/TH16-08-0620.
15. Mousavi S et al. Anti-inflammatory effects of heparin and its derivatives: A systematic review. Adv Pharmacol Pharm Sci. 2015 May 12. doi: 10.1155/2015/507151.
16. Kreuziger L et al. COVID-19 and VTE/anticoagulation: Frequently asked questions. American Society of Hematology. 2020 Apr 17.
Coronavirus disease 2019 (COVID-19) is a viral illness caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), currently causing a pandemic affecting many countries around the world, beginning in December 2019 and spreading rapidly on a global scale since. Globally, its burden has been increasing rapidly, with more than 1.2 million people testing positive for the illness and 123,000 people losing their lives, as per April 15th’s WHO COVID-19 Situation Report.1 These numbers are increasing with each passing day. Clinically, SARS-CoV-2 has a highly variable course, ranging from mild disease manifested as a self-limited illness (seen in younger and healthier patients) to severe pneumonia/ARDS and multiorgan failure with intravascular coagulopathy.2
In this article, we intend to investigate and establish a comprehensive review of COVID-19–associated coagulopathy mechanisms, laboratory findings, and current management guidelines put forth by various societies globally.
Mechanism of coagulopathy
COVID-19–associated coagulopathy has been shown to predispose to both arterial and venous thrombosis through excessive inflammation and hypoxia, leading to activation of the coagulation cascade and consumption of coagulation factors, resulting in microvascular thrombosis.3 Though the exact pathophysiology for the activation of this cascade is not known, the proposed mechanism has been: endothelial damage triggering platelet activation within the lung, leading to aggregation, thrombosis, and consumption of platelets in the lung.2,5,6
Fox et al. noted similar coagulopathy findings of four deceased COVID-19 patients. Autopsy results concluded that the dominant process was diffuse alveolar damage, notable CD4+ aggregates around thrombosed small vessels, significant associated hemorrhage, and thrombotic microangiopathy restricted to the lungs. The proposed mechanism was the activation of megakaryocytes, possibly native to the lung, with platelet aggregation, formation of platelet-rich clots, and fibrin deposition playing a major role.4
It has been noted that diabetic patients are at an increased risk of vascular events and hypercoagulability with COVID-19.7 COVID-19 can also cause livedo reticularis and acrocyanosis because of the microthrombosis in the cutaneous vasculature secondary to underlying coagulopathy, as reported in a case report of two U.S. patients with COVID-19.8
Clinical and laboratory abnormalities
A recent study reported from Netherlands by Klok et al. analyzed 184 ICU patients with COVID-19 pneumonia and concluded that the cumulative incidence of acute pulmonary embolism (PE), deep vein thrombosis (DVT), ischemic stroke, MI, or systemic arterial embolism was 31% (95% confidence interval, 20%-41%). PE was the most frequent thrombotic complication and was noted in 81% of patients. Coagulopathy, defined as spontaneous prolongation of prothrombin time (PT) > 3s or activated partial thromboplastin time (aPTT) > 5s, was reported as an independent predictor of thrombotic complications.3
Hematologic abnormalities that were noted in COVID-19 coagulopathy include: decreased platelet counts, decreased fibrinogen levels, elevated PT/INR, elevated partial thromboplastin time (PTT), and elevated d-dimer.9,10 In a retrospective analysis9 by Tang et al., 71.4% of nonsurvivors and 0.6% of survivors had met the criteria of disseminated intravascular coagulation (DIC) during their hospital stay. Nonsurvivors of COVID-19 had statistically significant elevation of d-dimer levels, FDP levels, PT, and aPTT, when compared to survivors (P < .05). The overall mortality in this study was reported as 11.5%.9 In addition, elevated d-dimer, fibrin and fibrinogen degradation product (FDP) levels and longer PT and aPTT were associated with poor prognosis.
Thus, d-dimer, PT, and platelet count should be measured in all patients who present with COVID-19 infection. We can also suggest that in patients with markedly elevated d-dimer (three- to fourfold increase), admission to hospital should be considered even in the absence of severe clinical symptoms.11
COVID-19 coagulopathy management
In a retrospective study9 of 449 patients with severe COVID-19 from Wuhan, China, by Tang et al., 99 patients mainly received low-weight molecular heparin (LMWH) for 7 days or longer. No difference in 28-day mortality was noted between heparin users and nonusers (30.3% vs. 29.7%; P = .910). A lower 28-day mortality rate was noted in heparin patients with sepsis-induced coagulopathy score of ≥4.0 (40.0% vs. 64.2%; P = .029) or a d-dimer level greater than sixfold of upper limit of normal, compared with nonusers of heparin.12
Another small study of seven COVID-19 patients with acroischemia in China demonstrated that administering LMWH was successful at decreasing the d-dimer and fibrinogen degradation product levels but noted no significant improvement in clinical symptoms.13
Recently, the International Society of Thrombosis and Hemostasis and American Society of Hematology published recommendations and guidelines regarding the recognition and management of coagulopathy in COVID-19.11 Prophylactic anticoagulation therapy with LMWH was recommended in all hospitalized patients with COVID-19, provided there was an absence of any contraindications (active bleeding, platelet count less than 25 x 109/L and fibrinogen less than 0.5 g/dL). Anticoagulation with LMWH was associated with better prognosis in severe COVID-19 patients and in COVID-19 patients with markedly elevated d-dimer, as it also has anti-inflammatory effects.12 This anti-inflammatory property of heparin has been documented in previous studies but the underlying mechanism is unknown and more research is required.14,15
Despite coagulopathy being noticed with cases of COVID-19, bleeding has been a rare finding in COVID-19 infections. If bleeding is noted, recommendations were made to keep platelet levels greater than 50 x109/L, fibrinogen less than 2.0 g/L, and INR [international normalized ratio] greater than 1.5.11 Mechanical thromboprophylaxis should be used when pharmacologic thromboprophylaxis is contraindicated.16
COVID-19 patients with new diagnoses of venous thromboembolism (VTE) or atrial fibrillation should be prescribed therapeutic anticoagulation. Patients who are already on anticoagulation for VTE or atrial fibrillation should continue their therapy unless the platelet count is less than 30-50x109/L or if the fibrinogen is less than 1.0 g/L.16
Conclusion
Coagulopathies associated with COVID-19 infections have been documented in several studies around the world, and it has been shown to be fatal in some cases. Despite documentation, the mechanism behind this coagulopathy is not well understood. Because of the potentially lethal complications associated with coagulopathies, early recognition and anticoagulation is imperative to improve clinical outcomes. These results are very preliminary: More studies are required to understand the role of anticoagulation and its effect on the morbidity and mortality associated with COVID-19–associated coagulopathy.
Dr. Yeruva is a board-certified hematologist/medical oncologist with WellSpan Health and clinical assistant professor of internal medicine, Penn State University, Hershey. Mr. Henderson is a third-year graduate-entry medical student at the Royal College of Surgeons in Ireland with interests in family medicine, dermatology, and tropical diseases. Dr. Al-Tawfiq is a consultant of internal medicine & infectious diseases, and the director of quality at Johns Hopkins Aramco Healthcare in Dhahran, Saudi Arabia, an adjunct associate professor of infectious diseases, molecular medicine and clinical pharmacology at Johns Hopkins University School of Medicine, and adjunct associate professor at Indiana University School of Medicine, Indianapolis. Dr. Tirupathi is the medical director of Keystone Infectious Diseases/HIV in Chambersburg, Pa., and currently chair of infection prevention at Wellspan Chambersburg and Waynesboro (Pa.) Hospitals. He also is the lead physician for antibiotic stewardship at these hospitals.
References
1. World Health Organization. Coronavirus disease (COVID-2019) situation reports.
2. Lippi G et al. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin Chim Acta. 2020 Mar 13. 506:145-8. doi: 10.1016/j.cca.2020.03.022.
3. Klok FA et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Throm Res. 2020;18(4):844-7. doi: 10.1016/j.thromres.2020.04.013.
4. Fox S et al. Pulmonary and cardiac pathology in Covid-19: The first autopsy series from New Orleans. MedRxiv. 2020 Apr 10. doi: 10.1101/2020.04.06.20050575.
5. Yang M et al. Thrombocytopenia in patients with severe acute respiratory syndrome (review). Hematology 2013 Sep 4. doi: 10.1080/1024533040002617.
6. Giannis D et al. Coagulation disorders in coronavirus infected patients: COVID-19, SARS-CoV-1, MERS-CoV and lessons from the past. J Clin Virol. 2020 June. doi: 10.1016/j.jcv.2020.104362.
7. Guo W et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020 Mar 31. doi: 10.1002/dmrr.3319.
8. Manalo IF et al. A dermatologic manifestation of COVID-19: Transient livedo reticularis. J Am Acad Dermat. 2020 Apr. doi: 10.1016/j.jaad.2020.04.018.
9. Tang N et al. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020 Feb 19. doi: 10.1111/jth.14768, 18: 844-847.
10. Huang C et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020 Jan 24. doi: 10.1016/S0140-6736(20)30183-5.
11. Thachil J et al. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J Thromb Haemost. 2020 Mar 25. doi: 10.1111/JTH.14810.
12. Tang N et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020 Mar 27. doi: 10.1111/JTH.14817.
13. Zhang Y et al. Clinical and coagulation characteristics of 7 patients with critical COVID-2019 pneumonia and acro-ischemia. Zhonghua Xue Ye Xue Za Zhi. 2020 Mar 28. doi: 10.3760/cma.j.issn.0253-2727.2020.0006.
14. Poterucha TJ et al. More than an anticoagulant: Do heparins have direct anti-inflammatory effects? Thromb Haemost. 2017. doi: 10.1160/TH16-08-0620.
15. Mousavi S et al. Anti-inflammatory effects of heparin and its derivatives: A systematic review. Adv Pharmacol Pharm Sci. 2015 May 12. doi: 10.1155/2015/507151.
16. Kreuziger L et al. COVID-19 and VTE/anticoagulation: Frequently asked questions. American Society of Hematology. 2020 Apr 17.